User login
Microneedling Therapy With and Without Platelet-Rich Plasma
Microneedling therapy, also known as collagen induction therapy or percutaneous collagen induction, is an increasingly popular treatment modality for skin rejuvenation. The approach employs small needles to puncture the skin and stimulate local collagen production in a minimally invasive manner. Recently, clinicians have incorporated the use of platelet-rich plasma (PRP) with the aim of augmenting cosmetic outcomes. In this article, we examine the utility of this approach by reviewing comparison studies of microneedling therapy with and without the application of PRP.
Dr. Gary Goldenberg demonstrates microneedling with platelet-rich plasma in a procedural video available here.
Microneedling Therapy
The use of microneedling first gained attention in the 1990s. Initially, Camirand and Doucet1 described tattooing without pigment for the treatment of achromatic and hypertrophic scars. Fernandes2 evolved this concept and developed a drum-shaped device with fine protruding needles to puncture the skin. Microneedling devices have expanded in recent years and now include both cord- and battery-powered pens and rollers, with needles ranging in length from 0.25 to 3.0 mm.
Treatment with microneedling promotes skin rejuvenation by creating small puncture wounds in the epidermis and dermis. This injury triggers the wound healing cascade and alters the modulation of growth factors to promote regenerative effects.3,4 Following microneedling therapy, increases occur in elastic fiber formation, collagen deposition, and dermal thickness (Figure).5 Of interesting histologic note, collagen is deposited in the normal lattice pattern following this treatment rather than in the parallel bundles typical of scars.6 Microneedling preserves the overall integrity of the epidermal layers and basement membrane, allowing the epidermis to heal without abnormality, verified on histology by a normal stratum corneum, enhanced stratum granulosum, and normal rete ridges.7
Microneedling has demonstrated several uses beyond general skin rejuvenation. In patients with atrophic acne scars, therapy can lead to improved scar appearance, skin texture, and patient satisfaction.8,9 Hypertrophic and dyspigmented burn scars on the body, face, arms, and legs have shown to be receptive to repeated treatments.10 Microneedling also has shown promise in treating androgenic alopecia, increasing hair regrowth in patients who previously showed poor response to conventional therapy with minoxidil and finasteride.11,12
Platelet-Rich Plasma
Platelet-rich plasma is developed by enriching blood with an autologous concentration of platelets. The preparation of PRP begins with whole blood, commonly obtained peripherally by venipuncture. Samples undergo centrifugation to allow separation of the blood into 3 layers: platelet-poor plasma, PRP, and erythrocytes.13 The typical platelet count of whole blood is approximately 200,000/µL; PRP aims to prepare a platelet count of at least 1,000,000/µL in a 5-mL volume.14
An attractive component of PRP is its high concentration of growth factors, including platelet-derived growth factor, transforming growth factor, vascular endothelial growth factor, and epithelial growth factor.15 Because of the regenerative effects of these proteins, PRP has been investigated as a modality to augment wound healing in a variety of clinical areas, such as maxillofacial surgery, orthopedics, cardiovascular surgery, and treatment of soft tissue ulcers.16
Combination Use of Microneedling and PRP
Several studies have compared the effects of microneedling with and without the application of PRP (Table).17-20 In an animal model, Akcal et al17 examined the effects of microneedling and PRP on skin flap survival. Eight rats were randomly divided into 5 groups: sham, control, microneedling alone, microneedling plus PRP, and microneedling plus platelet-poor plasma. Treatments were applied to skin flaps after 4 hours of induced ischemia. The surviving flap area was measured, with results demonstrating significantly higher viable areas in the microneedling plus PRP group relative to all other groups (P<.01). On histologic examination, the microneedling plus PRP group showed well-organized epidermal layers and a dermal integrity that matched the dermis of the sham group.17
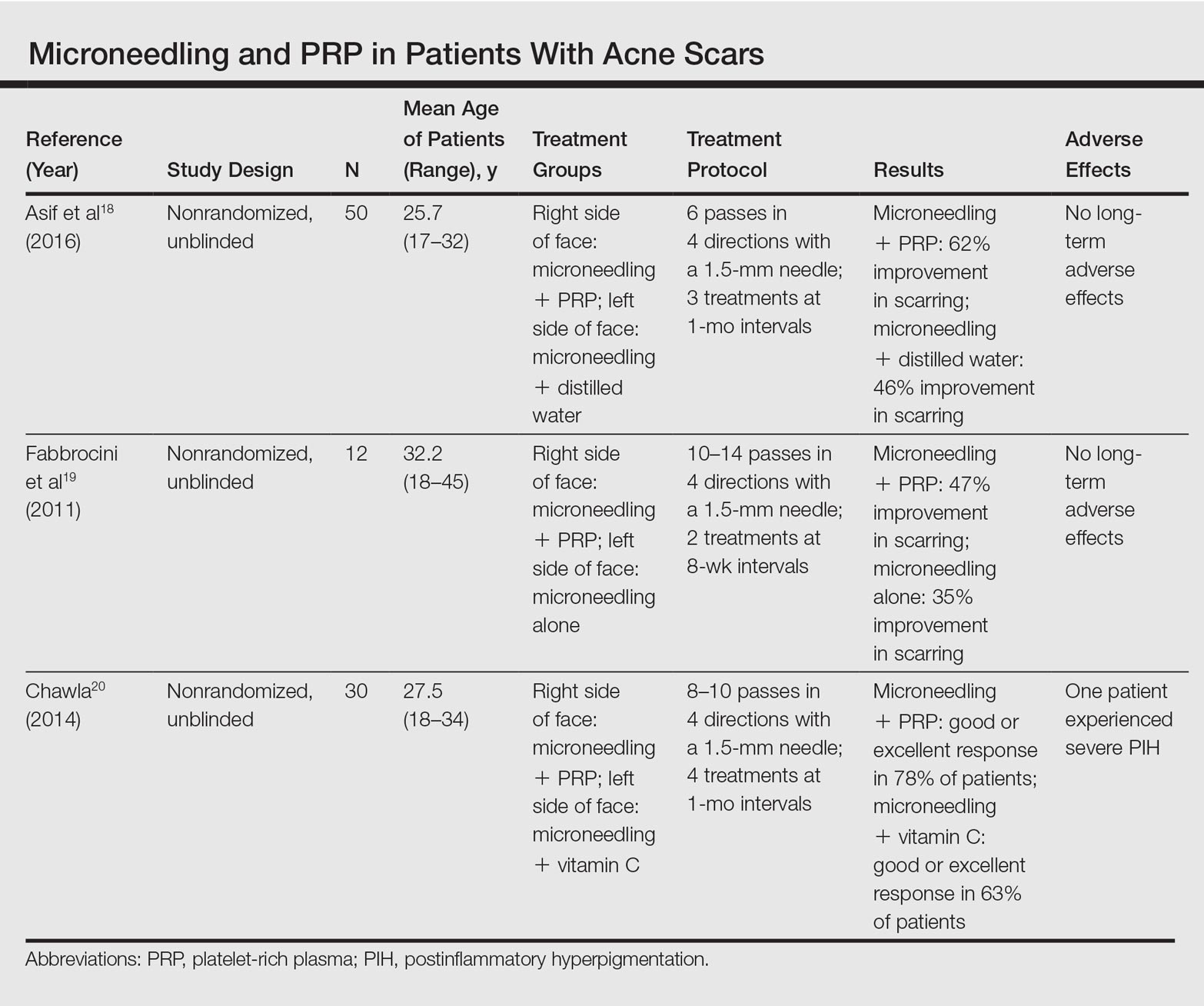
Asif et al18 performed a split-face comparison study of 50 patients with atrophic acne scars. On the right side, microneedling was performed followed by intradermal injections and topical application of PRP. On the left side, microneedling was performed followed by intradermal injections of distilled water. The study included 3 treatment sessions with 1 month between each session. Scars were assessed using the Goodman and Baron scale,21 which is designed to grade the morphology of postacne scarring. Scars on the right side improved by 62.2% and scars on the left side improved by 45.8%; prior to treatment, both sides demonstrated similar severity scores, but final severity scores were significantly reduced in the microneedling plus PRP group relative to the microneedling plus distilled water group (P<.00001). No residual side effects from treatment were reported.18
Examining the degree of improvement more carefully, microneedling plus PRP yielded excellent improvement in 40% (20/50) of patients and good improvement in 60% (30/50).18 Microneedling plus distilled water led to excellent improvement in 10% (5/50) and good improvement in 84% (42/50). Given that microneedling plus distilled water still provided good to excellent results in 94% of patients, the addition of PRP was helpful though not necessary in achieving meaningful benefit.18
In another split-face study, Fabbrocini et al19 evaluated 12 adult patients with acne scars. The right side of the face received microneedling plus PRP, while the left side received microneedling alone. Two treatments were performed 8 weeks apart. Severity scores (0=no lesions; 10=maximum severity) were used to assess patient outcomes throughout the study. Acne scars improved on both sides of the face following the treatment period, but the reduction in scar severity with microneedling plus PRP (3.5 points) was significantly greater than with microneedling alone (2.6 points)(P<.05). Patients tended to experience2 to 3 days of mild swelling and erythema after treatment regardless of PRP addition. With only 12 patients, the study was limited by a small sample size. The 10-point grading system differed from the Goodman and Baron scale in that it lacked corresponding qualitative markers, likely decreasing reproducibility.19
Chawla20 compared the effectiveness of combination therapy with microneedling plus PRP versus microneedling and vitamin C application. In a split-face study of 30 patients with atrophic acne scars, the right side of the face was treated with microneedling plus PRP and the left side was treated with microneedling plus vitamin C. Four sessions were performed with an interval of 1 month in between treatments. The Goodman and Baron Scale was used to assess treatment efficacy. Overall, both treatments led to improved outcomes, but in categorizing patients who demonstrated poor responses, a significantly larger percentage existed in the microneedling plus vitamin C group (37% [10/27]) versus the microneedling plus PRP group (22% [6/27])(P=.021). Additionally, aggregate patient satisfaction scores were higher with microneedling plus PRP relative to microneedling plus vitamin C (P=.01). Of note, assessments of improvement were performed by the treating physician and patient satisfaction reports were completed with knowledge of the therapies and cost factor, which may have influenced results.20
Conclusion
Microneedling therapy continues to evolve with a range of applications now emerging in dermatology. As PRP has gained popularity, there has been increased interest in its utilization to amplify the regenerative effects of microneedling. Although the number of direct comparisons examining microneedling with and without PRP is limited, the available evidence indicates that the addition of PRP may improve cosmetic outcomes. These results have been demonstrated primarily in the management of acne scars, but favorable effects may extend to other indications. Continued study is warranted to further quantify the degree of these benefits and to elucidate optimal treatment schedules.
In addition, it is important to consider a cost-benefit analysis of PRP. The price of PRP varies depending on the clinical site but in certain cases may double the cost of a microneedling treatment session. Although studies have demonstrated a statistically significant benefit to PRP, the clinical significance of this supplementary treatment must be weighed against the increased expense. A discussion should take place with the consideration that microneedling alone can provide a satisfactory result for some patients.
- Camirand A, Doucet J. Needle dermabrasion. Aesthetic Plast Surg. 1997;21:48-51.
- Fernandes D. Percutaneous collagen induction: an alternative to laser resurfacing. Aesthet Surg J. 2002;22:307-309.
- Fabbrocini G, Fardella N, Monfrecola A, et al. Acne scarring treatment using skin needling. Clin Exp Dermatol. 2009;34:874-879.
- Zeitter S, Sikora Z, Jahn S, et al. Microneedling: matching the results of medical needling and repetitive treatments to maximize potential for skin regeneration [published online February 7, 2014]. Burns. 2014;40:966-973.
- Schwarz M, Laaff H. A prospective controlled assessment of microneedling with the Dermaroller device. Plast Reconstr Surg. 2011;127:E146-E148.
- Fernandes D, Signorini M. Combating photoaging with percutaneous collagen induction. Clin Dermatol. 2008;26:192-199.
- Aust MC, Fernandes D, Kolokythas P, et al. Percutaneous collagen induction therapy: an alternative treatment for scars, wrinkles, and skin laxity. Plast Reconstr Surg. 2008;121:1421-1429.
- El-Domyati M, Barakat M, Awad S, et al. Microneedling therapy for atrophic acne scars: an objective evaluation. J Clin Aesthet Dermatol. 2015;8:36-42.
- Leheta T, El Tawdy A, Abdel Hay R, et al. Percutaneous collagen induction versus full-concentration trichloroacetic acid in the treatment of atrophic acne scars. Dermatol Surg. 2011;37:207-216.
- Aust MC, Knobloch K, Reimers K, et al. Percutaneous collagen induction therapy: an alternative treatment for burn scars. Burns. 2010;36:836-843.
- Dhurat R, Mathapati S. Response to microneedling treatment in men with androgenetic alopecia who failed to respond to conventional therapy. Indian J Dermatol. 2015;60:260-263.
- Dhurat R, Sukesh M, Avhad G, et al. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5:6-11.
- Wang HL, Avila G. Platelet rich plasma: myth or reality? Eur J Dent. 2007;1:192-194.
- Marx RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10:225-228.
- Lubkowska A, Dolegowska B, Banfi G. Growth factor content in PRP and their applicability in medicine. J Biol Regul Homeost Agents. 2012;26(2 suppl 1):3S-22S.
- Pietrzak WS, Eppley BL. Platelet rich plasma: biology and new technology. J Craniofac Surg. 2005;16:1043-1054.
- Akcal A, Savas SA, Gorgulu T, et al. The effect of platelete rich plasma combined with microneedling on full venous outflow compromise in a rat skin flap model. Plast Reconstr Surg. 2015;136(4 suppl):71-72.
- Asif M, Kanodia S, Singh K. Combined autologous platelet-rich plasma with microneedling verses microneedling with distilled water in the treatment of atrophic acne scars: a concurrent split-face study [published online January 8, 2016]. J Cosmet Dermatol. 2016;15:434-443.
- Fabbrocini G, De Vita V, Pastore F, et al. Combined use of skin needling and platelet-rich plasma in acne scarring treatment. Cosmet Dermatol. 2011;24:177-183.
- Chawla S. Split face comparative study of microneedling with PRP versus microneedling with vitamin C in treating atrophic post acne scars. J Cutan Aesthet Surg. 2014;7:209-212.
- Goodman GJ, Baron JA. Postacne scarring: a qualitative global scarring grading system. Dermatol Surg. 2006;32:1458-1466.
Microneedling therapy, also known as collagen induction therapy or percutaneous collagen induction, is an increasingly popular treatment modality for skin rejuvenation. The approach employs small needles to puncture the skin and stimulate local collagen production in a minimally invasive manner. Recently, clinicians have incorporated the use of platelet-rich plasma (PRP) with the aim of augmenting cosmetic outcomes. In this article, we examine the utility of this approach by reviewing comparison studies of microneedling therapy with and without the application of PRP.
Dr. Gary Goldenberg demonstrates microneedling with platelet-rich plasma in a procedural video available here.
Microneedling Therapy
The use of microneedling first gained attention in the 1990s. Initially, Camirand and Doucet1 described tattooing without pigment for the treatment of achromatic and hypertrophic scars. Fernandes2 evolved this concept and developed a drum-shaped device with fine protruding needles to puncture the skin. Microneedling devices have expanded in recent years and now include both cord- and battery-powered pens and rollers, with needles ranging in length from 0.25 to 3.0 mm.
Treatment with microneedling promotes skin rejuvenation by creating small puncture wounds in the epidermis and dermis. This injury triggers the wound healing cascade and alters the modulation of growth factors to promote regenerative effects.3,4 Following microneedling therapy, increases occur in elastic fiber formation, collagen deposition, and dermal thickness (Figure).5 Of interesting histologic note, collagen is deposited in the normal lattice pattern following this treatment rather than in the parallel bundles typical of scars.6 Microneedling preserves the overall integrity of the epidermal layers and basement membrane, allowing the epidermis to heal without abnormality, verified on histology by a normal stratum corneum, enhanced stratum granulosum, and normal rete ridges.7

Microneedling has demonstrated several uses beyond general skin rejuvenation. In patients with atrophic acne scars, therapy can lead to improved scar appearance, skin texture, and patient satisfaction.8,9 Hypertrophic and dyspigmented burn scars on the body, face, arms, and legs have shown to be receptive to repeated treatments.10 Microneedling also has shown promise in treating androgenic alopecia, increasing hair regrowth in patients who previously showed poor response to conventional therapy with minoxidil and finasteride.11,12
Platelet-Rich Plasma
Platelet-rich plasma is developed by enriching blood with an autologous concentration of platelets. The preparation of PRP begins with whole blood, commonly obtained peripherally by venipuncture. Samples undergo centrifugation to allow separation of the blood into 3 layers: platelet-poor plasma, PRP, and erythrocytes.13 The typical platelet count of whole blood is approximately 200,000/µL; PRP aims to prepare a platelet count of at least 1,000,000/µL in a 5-mL volume.14
An attractive component of PRP is its high concentration of growth factors, including platelet-derived growth factor, transforming growth factor, vascular endothelial growth factor, and epithelial growth factor.15 Because of the regenerative effects of these proteins, PRP has been investigated as a modality to augment wound healing in a variety of clinical areas, such as maxillofacial surgery, orthopedics, cardiovascular surgery, and treatment of soft tissue ulcers.16
Combination Use of Microneedling and PRP
Several studies have compared the effects of microneedling with and without the application of PRP (Table).17-20 In an animal model, Akcal et al17 examined the effects of microneedling and PRP on skin flap survival. Eight rats were randomly divided into 5 groups: sham, control, microneedling alone, microneedling plus PRP, and microneedling plus platelet-poor plasma. Treatments were applied to skin flaps after 4 hours of induced ischemia. The surviving flap area was measured, with results demonstrating significantly higher viable areas in the microneedling plus PRP group relative to all other groups (P<.01). On histologic examination, the microneedling plus PRP group showed well-organized epidermal layers and a dermal integrity that matched the dermis of the sham group.17

Asif et al18 performed a split-face comparison study of 50 patients with atrophic acne scars. On the right side, microneedling was performed followed by intradermal injections and topical application of PRP. On the left side, microneedling was performed followed by intradermal injections of distilled water. The study included 3 treatment sessions with 1 month between each session. Scars were assessed using the Goodman and Baron scale,21 which is designed to grade the morphology of postacne scarring. Scars on the right side improved by 62.2% and scars on the left side improved by 45.8%; prior to treatment, both sides demonstrated similar severity scores, but final severity scores were significantly reduced in the microneedling plus PRP group relative to the microneedling plus distilled water group (P<.00001). No residual side effects from treatment were reported.18
Examining the degree of improvement more carefully, microneedling plus PRP yielded excellent improvement in 40% (20/50) of patients and good improvement in 60% (30/50).18 Microneedling plus distilled water led to excellent improvement in 10% (5/50) and good improvement in 84% (42/50). Given that microneedling plus distilled water still provided good to excellent results in 94% of patients, the addition of PRP was helpful though not necessary in achieving meaningful benefit.18
In another split-face study, Fabbrocini et al19 evaluated 12 adult patients with acne scars. The right side of the face received microneedling plus PRP, while the left side received microneedling alone. Two treatments were performed 8 weeks apart. Severity scores (0=no lesions; 10=maximum severity) were used to assess patient outcomes throughout the study. Acne scars improved on both sides of the face following the treatment period, but the reduction in scar severity with microneedling plus PRP (3.5 points) was significantly greater than with microneedling alone (2.6 points)(P<.05). Patients tended to experience2 to 3 days of mild swelling and erythema after treatment regardless of PRP addition. With only 12 patients, the study was limited by a small sample size. The 10-point grading system differed from the Goodman and Baron scale in that it lacked corresponding qualitative markers, likely decreasing reproducibility.19
Chawla20 compared the effectiveness of combination therapy with microneedling plus PRP versus microneedling and vitamin C application. In a split-face study of 30 patients with atrophic acne scars, the right side of the face was treated with microneedling plus PRP and the left side was treated with microneedling plus vitamin C. Four sessions were performed with an interval of 1 month in between treatments. The Goodman and Baron Scale was used to assess treatment efficacy. Overall, both treatments led to improved outcomes, but in categorizing patients who demonstrated poor responses, a significantly larger percentage existed in the microneedling plus vitamin C group (37% [10/27]) versus the microneedling plus PRP group (22% [6/27])(P=.021). Additionally, aggregate patient satisfaction scores were higher with microneedling plus PRP relative to microneedling plus vitamin C (P=.01). Of note, assessments of improvement were performed by the treating physician and patient satisfaction reports were completed with knowledge of the therapies and cost factor, which may have influenced results.20
Conclusion
Microneedling therapy continues to evolve with a range of applications now emerging in dermatology. As PRP has gained popularity, there has been increased interest in its utilization to amplify the regenerative effects of microneedling. Although the number of direct comparisons examining microneedling with and without PRP is limited, the available evidence indicates that the addition of PRP may improve cosmetic outcomes. These results have been demonstrated primarily in the management of acne scars, but favorable effects may extend to other indications. Continued study is warranted to further quantify the degree of these benefits and to elucidate optimal treatment schedules.
In addition, it is important to consider a cost-benefit analysis of PRP. The price of PRP varies depending on the clinical site but in certain cases may double the cost of a microneedling treatment session. Although studies have demonstrated a statistically significant benefit to PRP, the clinical significance of this supplementary treatment must be weighed against the increased expense. A discussion should take place with the consideration that microneedling alone can provide a satisfactory result for some patients.
Microneedling therapy, also known as collagen induction therapy or percutaneous collagen induction, is an increasingly popular treatment modality for skin rejuvenation. The approach employs small needles to puncture the skin and stimulate local collagen production in a minimally invasive manner. Recently, clinicians have incorporated the use of platelet-rich plasma (PRP) with the aim of augmenting cosmetic outcomes. In this article, we examine the utility of this approach by reviewing comparison studies of microneedling therapy with and without the application of PRP.
Dr. Gary Goldenberg demonstrates microneedling with platelet-rich plasma in a procedural video available here.
Microneedling Therapy
The use of microneedling first gained attention in the 1990s. Initially, Camirand and Doucet1 described tattooing without pigment for the treatment of achromatic and hypertrophic scars. Fernandes2 evolved this concept and developed a drum-shaped device with fine protruding needles to puncture the skin. Microneedling devices have expanded in recent years and now include both cord- and battery-powered pens and rollers, with needles ranging in length from 0.25 to 3.0 mm.
Treatment with microneedling promotes skin rejuvenation by creating small puncture wounds in the epidermis and dermis. This injury triggers the wound healing cascade and alters the modulation of growth factors to promote regenerative effects.3,4 Following microneedling therapy, increases occur in elastic fiber formation, collagen deposition, and dermal thickness (Figure).5 Of interesting histologic note, collagen is deposited in the normal lattice pattern following this treatment rather than in the parallel bundles typical of scars.6 Microneedling preserves the overall integrity of the epidermal layers and basement membrane, allowing the epidermis to heal without abnormality, verified on histology by a normal stratum corneum, enhanced stratum granulosum, and normal rete ridges.7

Microneedling has demonstrated several uses beyond general skin rejuvenation. In patients with atrophic acne scars, therapy can lead to improved scar appearance, skin texture, and patient satisfaction.8,9 Hypertrophic and dyspigmented burn scars on the body, face, arms, and legs have shown to be receptive to repeated treatments.10 Microneedling also has shown promise in treating androgenic alopecia, increasing hair regrowth in patients who previously showed poor response to conventional therapy with minoxidil and finasteride.11,12
Platelet-Rich Plasma
Platelet-rich plasma is developed by enriching blood with an autologous concentration of platelets. The preparation of PRP begins with whole blood, commonly obtained peripherally by venipuncture. Samples undergo centrifugation to allow separation of the blood into 3 layers: platelet-poor plasma, PRP, and erythrocytes.13 The typical platelet count of whole blood is approximately 200,000/µL; PRP aims to prepare a platelet count of at least 1,000,000/µL in a 5-mL volume.14
An attractive component of PRP is its high concentration of growth factors, including platelet-derived growth factor, transforming growth factor, vascular endothelial growth factor, and epithelial growth factor.15 Because of the regenerative effects of these proteins, PRP has been investigated as a modality to augment wound healing in a variety of clinical areas, such as maxillofacial surgery, orthopedics, cardiovascular surgery, and treatment of soft tissue ulcers.16
Combination Use of Microneedling and PRP
Several studies have compared the effects of microneedling with and without the application of PRP (Table).17-20 In an animal model, Akcal et al17 examined the effects of microneedling and PRP on skin flap survival. Eight rats were randomly divided into 5 groups: sham, control, microneedling alone, microneedling plus PRP, and microneedling plus platelet-poor plasma. Treatments were applied to skin flaps after 4 hours of induced ischemia. The surviving flap area was measured, with results demonstrating significantly higher viable areas in the microneedling plus PRP group relative to all other groups (P<.01). On histologic examination, the microneedling plus PRP group showed well-organized epidermal layers and a dermal integrity that matched the dermis of the sham group.17

Asif et al18 performed a split-face comparison study of 50 patients with atrophic acne scars. On the right side, microneedling was performed followed by intradermal injections and topical application of PRP. On the left side, microneedling was performed followed by intradermal injections of distilled water. The study included 3 treatment sessions with 1 month between each session. Scars were assessed using the Goodman and Baron scale,21 which is designed to grade the morphology of postacne scarring. Scars on the right side improved by 62.2% and scars on the left side improved by 45.8%; prior to treatment, both sides demonstrated similar severity scores, but final severity scores were significantly reduced in the microneedling plus PRP group relative to the microneedling plus distilled water group (P<.00001). No residual side effects from treatment were reported.18
Examining the degree of improvement more carefully, microneedling plus PRP yielded excellent improvement in 40% (20/50) of patients and good improvement in 60% (30/50).18 Microneedling plus distilled water led to excellent improvement in 10% (5/50) and good improvement in 84% (42/50). Given that microneedling plus distilled water still provided good to excellent results in 94% of patients, the addition of PRP was helpful though not necessary in achieving meaningful benefit.18
In another split-face study, Fabbrocini et al19 evaluated 12 adult patients with acne scars. The right side of the face received microneedling plus PRP, while the left side received microneedling alone. Two treatments were performed 8 weeks apart. Severity scores (0=no lesions; 10=maximum severity) were used to assess patient outcomes throughout the study. Acne scars improved on both sides of the face following the treatment period, but the reduction in scar severity with microneedling plus PRP (3.5 points) was significantly greater than with microneedling alone (2.6 points)(P<.05). Patients tended to experience2 to 3 days of mild swelling and erythema after treatment regardless of PRP addition. With only 12 patients, the study was limited by a small sample size. The 10-point grading system differed from the Goodman and Baron scale in that it lacked corresponding qualitative markers, likely decreasing reproducibility.19
Chawla20 compared the effectiveness of combination therapy with microneedling plus PRP versus microneedling and vitamin C application. In a split-face study of 30 patients with atrophic acne scars, the right side of the face was treated with microneedling plus PRP and the left side was treated with microneedling plus vitamin C. Four sessions were performed with an interval of 1 month in between treatments. The Goodman and Baron Scale was used to assess treatment efficacy. Overall, both treatments led to improved outcomes, but in categorizing patients who demonstrated poor responses, a significantly larger percentage existed in the microneedling plus vitamin C group (37% [10/27]) versus the microneedling plus PRP group (22% [6/27])(P=.021). Additionally, aggregate patient satisfaction scores were higher with microneedling plus PRP relative to microneedling plus vitamin C (P=.01). Of note, assessments of improvement were performed by the treating physician and patient satisfaction reports were completed with knowledge of the therapies and cost factor, which may have influenced results.20
Conclusion
Microneedling therapy continues to evolve with a range of applications now emerging in dermatology. As PRP has gained popularity, there has been increased interest in its utilization to amplify the regenerative effects of microneedling. Although the number of direct comparisons examining microneedling with and without PRP is limited, the available evidence indicates that the addition of PRP may improve cosmetic outcomes. These results have been demonstrated primarily in the management of acne scars, but favorable effects may extend to other indications. Continued study is warranted to further quantify the degree of these benefits and to elucidate optimal treatment schedules.
In addition, it is important to consider a cost-benefit analysis of PRP. The price of PRP varies depending on the clinical site but in certain cases may double the cost of a microneedling treatment session. Although studies have demonstrated a statistically significant benefit to PRP, the clinical significance of this supplementary treatment must be weighed against the increased expense. A discussion should take place with the consideration that microneedling alone can provide a satisfactory result for some patients.
- Camirand A, Doucet J. Needle dermabrasion. Aesthetic Plast Surg. 1997;21:48-51.
- Fernandes D. Percutaneous collagen induction: an alternative to laser resurfacing. Aesthet Surg J. 2002;22:307-309.
- Fabbrocini G, Fardella N, Monfrecola A, et al. Acne scarring treatment using skin needling. Clin Exp Dermatol. 2009;34:874-879.
- Zeitter S, Sikora Z, Jahn S, et al. Microneedling: matching the results of medical needling and repetitive treatments to maximize potential for skin regeneration [published online February 7, 2014]. Burns. 2014;40:966-973.
- Schwarz M, Laaff H. A prospective controlled assessment of microneedling with the Dermaroller device. Plast Reconstr Surg. 2011;127:E146-E148.
- Fernandes D, Signorini M. Combating photoaging with percutaneous collagen induction. Clin Dermatol. 2008;26:192-199.
- Aust MC, Fernandes D, Kolokythas P, et al. Percutaneous collagen induction therapy: an alternative treatment for scars, wrinkles, and skin laxity. Plast Reconstr Surg. 2008;121:1421-1429.
- El-Domyati M, Barakat M, Awad S, et al. Microneedling therapy for atrophic acne scars: an objective evaluation. J Clin Aesthet Dermatol. 2015;8:36-42.
- Leheta T, El Tawdy A, Abdel Hay R, et al. Percutaneous collagen induction versus full-concentration trichloroacetic acid in the treatment of atrophic acne scars. Dermatol Surg. 2011;37:207-216.
- Aust MC, Knobloch K, Reimers K, et al. Percutaneous collagen induction therapy: an alternative treatment for burn scars. Burns. 2010;36:836-843.
- Dhurat R, Mathapati S. Response to microneedling treatment in men with androgenetic alopecia who failed to respond to conventional therapy. Indian J Dermatol. 2015;60:260-263.
- Dhurat R, Sukesh M, Avhad G, et al. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5:6-11.
- Wang HL, Avila G. Platelet rich plasma: myth or reality? Eur J Dent. 2007;1:192-194.
- Marx RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10:225-228.
- Lubkowska A, Dolegowska B, Banfi G. Growth factor content in PRP and their applicability in medicine. J Biol Regul Homeost Agents. 2012;26(2 suppl 1):3S-22S.
- Pietrzak WS, Eppley BL. Platelet rich plasma: biology and new technology. J Craniofac Surg. 2005;16:1043-1054.
- Akcal A, Savas SA, Gorgulu T, et al. The effect of platelete rich plasma combined with microneedling on full venous outflow compromise in a rat skin flap model. Plast Reconstr Surg. 2015;136(4 suppl):71-72.
- Asif M, Kanodia S, Singh K. Combined autologous platelet-rich plasma with microneedling verses microneedling with distilled water in the treatment of atrophic acne scars: a concurrent split-face study [published online January 8, 2016]. J Cosmet Dermatol. 2016;15:434-443.
- Fabbrocini G, De Vita V, Pastore F, et al. Combined use of skin needling and platelet-rich plasma in acne scarring treatment. Cosmet Dermatol. 2011;24:177-183.
- Chawla S. Split face comparative study of microneedling with PRP versus microneedling with vitamin C in treating atrophic post acne scars. J Cutan Aesthet Surg. 2014;7:209-212.
- Goodman GJ, Baron JA. Postacne scarring: a qualitative global scarring grading system. Dermatol Surg. 2006;32:1458-1466.
- Camirand A, Doucet J. Needle dermabrasion. Aesthetic Plast Surg. 1997;21:48-51.
- Fernandes D. Percutaneous collagen induction: an alternative to laser resurfacing. Aesthet Surg J. 2002;22:307-309.
- Fabbrocini G, Fardella N, Monfrecola A, et al. Acne scarring treatment using skin needling. Clin Exp Dermatol. 2009;34:874-879.
- Zeitter S, Sikora Z, Jahn S, et al. Microneedling: matching the results of medical needling and repetitive treatments to maximize potential for skin regeneration [published online February 7, 2014]. Burns. 2014;40:966-973.
- Schwarz M, Laaff H. A prospective controlled assessment of microneedling with the Dermaroller device. Plast Reconstr Surg. 2011;127:E146-E148.
- Fernandes D, Signorini M. Combating photoaging with percutaneous collagen induction. Clin Dermatol. 2008;26:192-199.
- Aust MC, Fernandes D, Kolokythas P, et al. Percutaneous collagen induction therapy: an alternative treatment for scars, wrinkles, and skin laxity. Plast Reconstr Surg. 2008;121:1421-1429.
- El-Domyati M, Barakat M, Awad S, et al. Microneedling therapy for atrophic acne scars: an objective evaluation. J Clin Aesthet Dermatol. 2015;8:36-42.
- Leheta T, El Tawdy A, Abdel Hay R, et al. Percutaneous collagen induction versus full-concentration trichloroacetic acid in the treatment of atrophic acne scars. Dermatol Surg. 2011;37:207-216.
- Aust MC, Knobloch K, Reimers K, et al. Percutaneous collagen induction therapy: an alternative treatment for burn scars. Burns. 2010;36:836-843.
- Dhurat R, Mathapati S. Response to microneedling treatment in men with androgenetic alopecia who failed to respond to conventional therapy. Indian J Dermatol. 2015;60:260-263.
- Dhurat R, Sukesh M, Avhad G, et al. A randomized evaluator blinded study of effect of microneedling in androgenetic alopecia: a pilot study. Int J Trichology. 2013;5:6-11.
- Wang HL, Avila G. Platelet rich plasma: myth or reality? Eur J Dent. 2007;1:192-194.
- Marx RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10:225-228.
- Lubkowska A, Dolegowska B, Banfi G. Growth factor content in PRP and their applicability in medicine. J Biol Regul Homeost Agents. 2012;26(2 suppl 1):3S-22S.
- Pietrzak WS, Eppley BL. Platelet rich plasma: biology and new technology. J Craniofac Surg. 2005;16:1043-1054.
- Akcal A, Savas SA, Gorgulu T, et al. The effect of platelete rich plasma combined with microneedling on full venous outflow compromise in a rat skin flap model. Plast Reconstr Surg. 2015;136(4 suppl):71-72.
- Asif M, Kanodia S, Singh K. Combined autologous platelet-rich plasma with microneedling verses microneedling with distilled water in the treatment of atrophic acne scars: a concurrent split-face study [published online January 8, 2016]. J Cosmet Dermatol. 2016;15:434-443.
- Fabbrocini G, De Vita V, Pastore F, et al. Combined use of skin needling and platelet-rich plasma in acne scarring treatment. Cosmet Dermatol. 2011;24:177-183.
- Chawla S. Split face comparative study of microneedling with PRP versus microneedling with vitamin C in treating atrophic post acne scars. J Cutan Aesthet Surg. 2014;7:209-212.
- Goodman GJ, Baron JA. Postacne scarring: a qualitative global scarring grading system. Dermatol Surg. 2006;32:1458-1466.
Practice Points
- Microneedling is an effective therapy for skin rejuvenation.
- Preliminary evidence indicates that the addition of platelet-rich plasma to microneedling improves cosmetic outcomes.
Cosmetic Corner: Dermatologists Weigh in on Products for Dry Cuticles
To improve patient care and outcomes, leading dermatologists offered their recommendations on dry cuticle products. Consideration must be given to:
- Aquaphor Healing Ointment
Beiersdorf Inc.
“Using this product several times daily works great.”—Gary Goldenberg, MD, New York, New York
- Elon Lanolin-Rich Nail Conditioner
Dartmouth Pharmaceuticals
“Dry cuticles often are accompanied by splitting, cracking, and peeling of the nails. I have found that with regular use of this product, the condition of the nail as well as the cuticle can improve dramatically, leading to smoother, stronger cuticles and nails.”—Jeannette Graf, MD, New York, New York
- Petrolatum or Olive Oil
Manufacturers vary
“Apply petrolatum or olive oil to the fingertips after soaking for 5 to 10 minutes in lukewarm water, then wear nitrile gloves for an hour. Patients should then wipe off the excess and put on cotton gloves overnight.”—Larisa Ravitskiy, MD, Gahanna, Ohio
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, as well as products for dry cuticles, hyperhidrosis, and sensitive skin will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
To improve patient care and outcomes, leading dermatologists offered their recommendations on dry cuticle products. Consideration must be given to:
- Aquaphor Healing Ointment
Beiersdorf Inc.
“Using this product several times daily works great.”—Gary Goldenberg, MD, New York, New York
- Elon Lanolin-Rich Nail Conditioner
Dartmouth Pharmaceuticals
“Dry cuticles often are accompanied by splitting, cracking, and peeling of the nails. I have found that with regular use of this product, the condition of the nail as well as the cuticle can improve dramatically, leading to smoother, stronger cuticles and nails.”—Jeannette Graf, MD, New York, New York
- Petrolatum or Olive Oil
Manufacturers vary
“Apply petrolatum or olive oil to the fingertips after soaking for 5 to 10 minutes in lukewarm water, then wear nitrile gloves for an hour. Patients should then wipe off the excess and put on cotton gloves overnight.”—Larisa Ravitskiy, MD, Gahanna, Ohio
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, as well as products for dry cuticles, hyperhidrosis, and sensitive skin will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
To improve patient care and outcomes, leading dermatologists offered their recommendations on dry cuticle products. Consideration must be given to:
- Aquaphor Healing Ointment
Beiersdorf Inc.
“Using this product several times daily works great.”—Gary Goldenberg, MD, New York, New York
- Elon Lanolin-Rich Nail Conditioner
Dartmouth Pharmaceuticals
“Dry cuticles often are accompanied by splitting, cracking, and peeling of the nails. I have found that with regular use of this product, the condition of the nail as well as the cuticle can improve dramatically, leading to smoother, stronger cuticles and nails.”—Jeannette Graf, MD, New York, New York
- Petrolatum or Olive Oil
Manufacturers vary
“Apply petrolatum or olive oil to the fingertips after soaking for 5 to 10 minutes in lukewarm water, then wear nitrile gloves for an hour. Patients should then wipe off the excess and put on cotton gloves overnight.”—Larisa Ravitskiy, MD, Gahanna, Ohio
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, as well as products for dry cuticles, hyperhidrosis, and sensitive skin will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
[polldaddy:9711250]
VIDEO: Picowave laser uses are expanding beyond tattoo removal
ORLANDO – The applications for picowavelength lasers are expanding, with emerging data on their uses for cosmetic indications other than tattoo removal, according to Anne Chapas, MD, who is in private practice in New York.
First introduced and cleared by the Food and Drug Administration for tattoo removal, “picowave devices ... are now being studied in multiple different cosmetic conditions, including their use in acne scars, fine lines and wrinkles, and melasma,” Dr. Chapas said in a video interview at the annual meeting of the American Academy of Dermatology.
The No. 1 thing dermatologists need to know is that these types of lasers are delivering energy extremely quickly,” at 1,000 times faster than nanosecond lasers, she said. Another difference between the two is that “the laser tissue interaction between the two types of devices is completely different.”
In the interview, she highlighted other important points about picowave lasers, including less downtime after treatment.
At the meeting, Dr. Chapas spoke during a session entitled “the Science Behind New Devices in Dermatology.”
Her disclosures include serving as a consultant and investigator for Syneron and Candela.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ORLANDO – The applications for picowavelength lasers are expanding, with emerging data on their uses for cosmetic indications other than tattoo removal, according to Anne Chapas, MD, who is in private practice in New York.
First introduced and cleared by the Food and Drug Administration for tattoo removal, “picowave devices ... are now being studied in multiple different cosmetic conditions, including their use in acne scars, fine lines and wrinkles, and melasma,” Dr. Chapas said in a video interview at the annual meeting of the American Academy of Dermatology.
The No. 1 thing dermatologists need to know is that these types of lasers are delivering energy extremely quickly,” at 1,000 times faster than nanosecond lasers, she said. Another difference between the two is that “the laser tissue interaction between the two types of devices is completely different.”
In the interview, she highlighted other important points about picowave lasers, including less downtime after treatment.
At the meeting, Dr. Chapas spoke during a session entitled “the Science Behind New Devices in Dermatology.”
Her disclosures include serving as a consultant and investigator for Syneron and Candela.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
ORLANDO – The applications for picowavelength lasers are expanding, with emerging data on their uses for cosmetic indications other than tattoo removal, according to Anne Chapas, MD, who is in private practice in New York.
First introduced and cleared by the Food and Drug Administration for tattoo removal, “picowave devices ... are now being studied in multiple different cosmetic conditions, including their use in acne scars, fine lines and wrinkles, and melasma,” Dr. Chapas said in a video interview at the annual meeting of the American Academy of Dermatology.
The No. 1 thing dermatologists need to know is that these types of lasers are delivering energy extremely quickly,” at 1,000 times faster than nanosecond lasers, she said. Another difference between the two is that “the laser tissue interaction between the two types of devices is completely different.”
In the interview, she highlighted other important points about picowave lasers, including less downtime after treatment.
At the meeting, Dr. Chapas spoke during a session entitled “the Science Behind New Devices in Dermatology.”
Her disclosures include serving as a consultant and investigator for Syneron and Candela.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT AAD 17
Lasers for Latino skin – A balance of gentleness and strength
ORLANDO – With its unique tendency to develop postinflammatory hyperpigmentation (PIH), Latino skin needs a gentle touch from powerful lasers, according to Eduardo Weiss, MD.
“It’s often best to use a lower power, even though you trade off some efficacy for safety,” said Dr. Weiss, a dermatologist in Miami. “In general, it’s better to go with less aggressive treatment and more sessions, than to risk getting too aggressive and having a bad outcome.”
Dr. Weiss stressed that there are no “one size fits all” recommendations about laser treatment in typical Latino skin, because there’s no such thing as typical Latino skin. The group comprises all Fitzpatrick phototypes. But, in general, he said, the darker the skin, the greater the chance of an acute laser-induced burn, postinflammatory hyperpigmentation, and scarring.
Although general skin tone can provide a good first guess about the potential for hyperreaction to lasers, Dr. Weiss bolsters his judgments with a very simple – but effective – screen: palmar and digital crease pigmentation. First suggested by Hector G. Leal-Silva, MD, of the Institute of Dermatology and Cosmetic Surgery, Monterrey, Mexico, the screen divides patients into four groups, depending on the concentration of pigment present in palmar creases. A score of 0 means no pigment is visible, and the risk of PIH is negligible; a score of 3 means the creases are highly pigmented, and that the risk of PIH is very high.
But, with some adjustments in delivery – including using longer wavelengths and pulse duration, lower fluence and density, and smaller spot sizes – lasers can be used safely and effectively in these at-risk patients, Dr. Weiss said.
Safe treatment starts with pretreatment. It’s best to avoid laser procedures during the summer, when skin is at its darkest. Dr. Weiss also recommends a 6-week regimen aimed at lightening the area to be treated. This can include:
- Sun avoidance and the regular use of a high SPF sunscreen.
- Hydroquinone 4%-8%.
- A “Miami peel,” which is a modified Jessner’s peel with kojic acid and hydroquinone.
- Kligman’s formula of hydroquinone, tretinoin, and a corticosteroid.
- Heliocare, an oral extract of the Polypodium leucotomos fern.
About a month before the procedure, he performs a test spot with the intended laser and its planned settings, in the preauricular area. Any PIH will be obvious within 2-4 weeks. He also carefully screens patients or any photosensitizing condition, like lupus or herpes simplex, or a history of any photosensitizing drugs, such as tetracycline.
Dr. Weiss made specific suggestions for laser treatment of some common Latino skin issues:
Pigmented lesions
The high density of epidermal melanin in Fitzpatrick types IV-VI acts as a competitive chromophore against hemoglobin and oxyhemoglobin. This makes it quite challenging to treat vascular lesions such as port wine stains and telangiectasias, he said. The pulsed dye laser is a good choice, with wavelengths of 585-590 nm especially effective. “The longer 595-nm wavelength allows for a slightly deeper penetration,” Dr. Weiss said. “However, the absorption coefficient of oxyhemoglobin is three times higher at 585 nm than [at] 590 nm.”
Longer pulses are generally safer in dark-skinned patients, he noted. “It will be much less effective than the 585, but for darker-skinned patients, we must sacrifice a little efficacy for safety.”
For rosacea and telangiectasias, Dr. Weiss suggests a 515-nm intense pulsed light with pulse duration of 12-15/ms for these lesions, or pigmentation, he also uses 540 nm or 500-600 nm with pulse duration of 12-15/ms.
Melasma, a very common condition in dark-skinned Latinos, is also “one of the most difficult and frustrating conditions to manage,” he pointed out. He turns to a laser only when the case is resistant to more conservative treatment, which typically includes Kligman’s formula, sunscreen, light peels, and azelaic or kojic acid.
“Lasers are still controversial and, in my opinion, a last resort for melasma. I wouldn’t start unless everything else fails. I reserve them for the deep nonresponding melasmas.”
The Q-switched Nd:YAG is the most widely used for melasma. The fluence used is less than 5 J/cm2, with a 6-mm spot size and a frequency of 10 Hz. The number of treatment sessions varies from 5 to 10 at 1-week intervals, Dr. Weiss said. “Keep in mind that rebound hyperpigmentation could be due to the multiple subthreshold exposures that can stimulate melanogenesis in some areas.”
Skin rejuvenation
Ablative lasers – long the gold standard for skin rejuvenation in those with light skin – can be problematic for darker-skinned patients, Dr. Weiss said.
“These lasers, like the CO2 and Erbium:YAG, can cause several unwanted side effects in Latino skin.” These can include hyperpigmentation, which occurs in 50% of Fitzpatrick III or higher phototypes; erythema that can last for months; and delayed-onset hypopigmentation.
“I think better options for our darker-skinned patients are nonablative infrared, microneedling, and radiofrequency devices,” Dr. Weiss said. “There are, however, newer microablative resurfacing lasers. Fractional CO2, fractional Erbium, and the 2,790-nm yttrium scandium gallium garnet, offer a safer modality with which to treat skin types IV and above. Compared with the older-generation resurfacing lasers, the microablative lasers minimize the amount and duration of erythema and edema, which can last just 3-4 days.”
Dr. Weiss had no relevant financial disclosures.
ORLANDO – With its unique tendency to develop postinflammatory hyperpigmentation (PIH), Latino skin needs a gentle touch from powerful lasers, according to Eduardo Weiss, MD.
“It’s often best to use a lower power, even though you trade off some efficacy for safety,” said Dr. Weiss, a dermatologist in Miami. “In general, it’s better to go with less aggressive treatment and more sessions, than to risk getting too aggressive and having a bad outcome.”
Dr. Weiss stressed that there are no “one size fits all” recommendations about laser treatment in typical Latino skin, because there’s no such thing as typical Latino skin. The group comprises all Fitzpatrick phototypes. But, in general, he said, the darker the skin, the greater the chance of an acute laser-induced burn, postinflammatory hyperpigmentation, and scarring.
Although general skin tone can provide a good first guess about the potential for hyperreaction to lasers, Dr. Weiss bolsters his judgments with a very simple – but effective – screen: palmar and digital crease pigmentation. First suggested by Hector G. Leal-Silva, MD, of the Institute of Dermatology and Cosmetic Surgery, Monterrey, Mexico, the screen divides patients into four groups, depending on the concentration of pigment present in palmar creases. A score of 0 means no pigment is visible, and the risk of PIH is negligible; a score of 3 means the creases are highly pigmented, and that the risk of PIH is very high.
But, with some adjustments in delivery – including using longer wavelengths and pulse duration, lower fluence and density, and smaller spot sizes – lasers can be used safely and effectively in these at-risk patients, Dr. Weiss said.
Safe treatment starts with pretreatment. It’s best to avoid laser procedures during the summer, when skin is at its darkest. Dr. Weiss also recommends a 6-week regimen aimed at lightening the area to be treated. This can include:
- Sun avoidance and the regular use of a high SPF sunscreen.
- Hydroquinone 4%-8%.
- A “Miami peel,” which is a modified Jessner’s peel with kojic acid and hydroquinone.
- Kligman’s formula of hydroquinone, tretinoin, and a corticosteroid.
- Heliocare, an oral extract of the Polypodium leucotomos fern.
About a month before the procedure, he performs a test spot with the intended laser and its planned settings, in the preauricular area. Any PIH will be obvious within 2-4 weeks. He also carefully screens patients or any photosensitizing condition, like lupus or herpes simplex, or a history of any photosensitizing drugs, such as tetracycline.
Dr. Weiss made specific suggestions for laser treatment of some common Latino skin issues:
Pigmented lesions
The high density of epidermal melanin in Fitzpatrick types IV-VI acts as a competitive chromophore against hemoglobin and oxyhemoglobin. This makes it quite challenging to treat vascular lesions such as port wine stains and telangiectasias, he said. The pulsed dye laser is a good choice, with wavelengths of 585-590 nm especially effective. “The longer 595-nm wavelength allows for a slightly deeper penetration,” Dr. Weiss said. “However, the absorption coefficient of oxyhemoglobin is three times higher at 585 nm than [at] 590 nm.”
Longer pulses are generally safer in dark-skinned patients, he noted. “It will be much less effective than the 585, but for darker-skinned patients, we must sacrifice a little efficacy for safety.”
For rosacea and telangiectasias, Dr. Weiss suggests a 515-nm intense pulsed light with pulse duration of 12-15/ms for these lesions, or pigmentation, he also uses 540 nm or 500-600 nm with pulse duration of 12-15/ms.
Melasma, a very common condition in dark-skinned Latinos, is also “one of the most difficult and frustrating conditions to manage,” he pointed out. He turns to a laser only when the case is resistant to more conservative treatment, which typically includes Kligman’s formula, sunscreen, light peels, and azelaic or kojic acid.
“Lasers are still controversial and, in my opinion, a last resort for melasma. I wouldn’t start unless everything else fails. I reserve them for the deep nonresponding melasmas.”
The Q-switched Nd:YAG is the most widely used for melasma. The fluence used is less than 5 J/cm2, with a 6-mm spot size and a frequency of 10 Hz. The number of treatment sessions varies from 5 to 10 at 1-week intervals, Dr. Weiss said. “Keep in mind that rebound hyperpigmentation could be due to the multiple subthreshold exposures that can stimulate melanogenesis in some areas.”
Skin rejuvenation
Ablative lasers – long the gold standard for skin rejuvenation in those with light skin – can be problematic for darker-skinned patients, Dr. Weiss said.
“These lasers, like the CO2 and Erbium:YAG, can cause several unwanted side effects in Latino skin.” These can include hyperpigmentation, which occurs in 50% of Fitzpatrick III or higher phototypes; erythema that can last for months; and delayed-onset hypopigmentation.
“I think better options for our darker-skinned patients are nonablative infrared, microneedling, and radiofrequency devices,” Dr. Weiss said. “There are, however, newer microablative resurfacing lasers. Fractional CO2, fractional Erbium, and the 2,790-nm yttrium scandium gallium garnet, offer a safer modality with which to treat skin types IV and above. Compared with the older-generation resurfacing lasers, the microablative lasers minimize the amount and duration of erythema and edema, which can last just 3-4 days.”
Dr. Weiss had no relevant financial disclosures.
ORLANDO – With its unique tendency to develop postinflammatory hyperpigmentation (PIH), Latino skin needs a gentle touch from powerful lasers, according to Eduardo Weiss, MD.
“It’s often best to use a lower power, even though you trade off some efficacy for safety,” said Dr. Weiss, a dermatologist in Miami. “In general, it’s better to go with less aggressive treatment and more sessions, than to risk getting too aggressive and having a bad outcome.”
Dr. Weiss stressed that there are no “one size fits all” recommendations about laser treatment in typical Latino skin, because there’s no such thing as typical Latino skin. The group comprises all Fitzpatrick phototypes. But, in general, he said, the darker the skin, the greater the chance of an acute laser-induced burn, postinflammatory hyperpigmentation, and scarring.
Although general skin tone can provide a good first guess about the potential for hyperreaction to lasers, Dr. Weiss bolsters his judgments with a very simple – but effective – screen: palmar and digital crease pigmentation. First suggested by Hector G. Leal-Silva, MD, of the Institute of Dermatology and Cosmetic Surgery, Monterrey, Mexico, the screen divides patients into four groups, depending on the concentration of pigment present in palmar creases. A score of 0 means no pigment is visible, and the risk of PIH is negligible; a score of 3 means the creases are highly pigmented, and that the risk of PIH is very high.
But, with some adjustments in delivery – including using longer wavelengths and pulse duration, lower fluence and density, and smaller spot sizes – lasers can be used safely and effectively in these at-risk patients, Dr. Weiss said.
Safe treatment starts with pretreatment. It’s best to avoid laser procedures during the summer, when skin is at its darkest. Dr. Weiss also recommends a 6-week regimen aimed at lightening the area to be treated. This can include:
- Sun avoidance and the regular use of a high SPF sunscreen.
- Hydroquinone 4%-8%.
- A “Miami peel,” which is a modified Jessner’s peel with kojic acid and hydroquinone.
- Kligman’s formula of hydroquinone, tretinoin, and a corticosteroid.
- Heliocare, an oral extract of the Polypodium leucotomos fern.
About a month before the procedure, he performs a test spot with the intended laser and its planned settings, in the preauricular area. Any PIH will be obvious within 2-4 weeks. He also carefully screens patients or any photosensitizing condition, like lupus or herpes simplex, or a history of any photosensitizing drugs, such as tetracycline.
Dr. Weiss made specific suggestions for laser treatment of some common Latino skin issues:
Pigmented lesions
The high density of epidermal melanin in Fitzpatrick types IV-VI acts as a competitive chromophore against hemoglobin and oxyhemoglobin. This makes it quite challenging to treat vascular lesions such as port wine stains and telangiectasias, he said. The pulsed dye laser is a good choice, with wavelengths of 585-590 nm especially effective. “The longer 595-nm wavelength allows for a slightly deeper penetration,” Dr. Weiss said. “However, the absorption coefficient of oxyhemoglobin is three times higher at 585 nm than [at] 590 nm.”
Longer pulses are generally safer in dark-skinned patients, he noted. “It will be much less effective than the 585, but for darker-skinned patients, we must sacrifice a little efficacy for safety.”
For rosacea and telangiectasias, Dr. Weiss suggests a 515-nm intense pulsed light with pulse duration of 12-15/ms for these lesions, or pigmentation, he also uses 540 nm or 500-600 nm with pulse duration of 12-15/ms.
Melasma, a very common condition in dark-skinned Latinos, is also “one of the most difficult and frustrating conditions to manage,” he pointed out. He turns to a laser only when the case is resistant to more conservative treatment, which typically includes Kligman’s formula, sunscreen, light peels, and azelaic or kojic acid.
“Lasers are still controversial and, in my opinion, a last resort for melasma. I wouldn’t start unless everything else fails. I reserve them for the deep nonresponding melasmas.”
The Q-switched Nd:YAG is the most widely used for melasma. The fluence used is less than 5 J/cm2, with a 6-mm spot size and a frequency of 10 Hz. The number of treatment sessions varies from 5 to 10 at 1-week intervals, Dr. Weiss said. “Keep in mind that rebound hyperpigmentation could be due to the multiple subthreshold exposures that can stimulate melanogenesis in some areas.”
Skin rejuvenation
Ablative lasers – long the gold standard for skin rejuvenation in those with light skin – can be problematic for darker-skinned patients, Dr. Weiss said.
“These lasers, like the CO2 and Erbium:YAG, can cause several unwanted side effects in Latino skin.” These can include hyperpigmentation, which occurs in 50% of Fitzpatrick III or higher phototypes; erythema that can last for months; and delayed-onset hypopigmentation.
“I think better options for our darker-skinned patients are nonablative infrared, microneedling, and radiofrequency devices,” Dr. Weiss said. “There are, however, newer microablative resurfacing lasers. Fractional CO2, fractional Erbium, and the 2,790-nm yttrium scandium gallium garnet, offer a safer modality with which to treat skin types IV and above. Compared with the older-generation resurfacing lasers, the microablative lasers minimize the amount and duration of erythema and edema, which can last just 3-4 days.”
Dr. Weiss had no relevant financial disclosures.
EXPERT ANALYSIS FROM AAD 17
The power of words in aesthetic procedures and healing patients
The words we choose to use prior to procedures can positively or negatively impact a patient’s experience during a procedure and their decision to have the procedure performed. A practical example would include using the word discomfort instead of pain to describe pain that may be associated with a procedure. The root word of discomfort is comfort, which the mind focuses on and creates less of an anxious state than pain.
Obviously, the need to provide proper and realistic expectations, as well as risks and benefits, is of utmost importance when obtaining informed consent. The words used can put a patient’s mind at ease or cause further anxiety about ideas of needles, scalpels, pain, risk of infection, and bleeding that are part of our everyday procedures.
Judith Thomas, DDS, a dentist in Virginia who is trained in clinical hypnosis, once described the power of the word but. People will often put more emphasis in their minds on what is said after the word but than on what is said before. For example, in a romantic relationship context, saying “I love you, but you drive me crazy” has a different impact than “You drive me crazy, but I love you.” The focus tends to stay on the “I love you” portion more when it is said last, after the “but.”
The same phenomenon can happen when we discuss procedures with our patients. When a medical assistant performs phlebotomy or when we as doctors are about to perform an injection, instead of saying this is going to hurt, another way to phrase it would be “In a moment you may feel something, but it doesn’t have to bother you” or “You may experience some discomfort, but it will resolve quickly.” Something I’ve said for years to patients before surgery is “You may feel a little stinging as the anesthetic goes in, after that you may feel me touching you, but nothing uncomfortable.” I guess I had been intuitively using this technique for years, without knowing the impact of the word “but.” Perhaps now that I am more mindful of it, I will be even more mindful of how I phrase these terms. We, in addition to our nurses and medical assistants, can use these techniques to enhance patient comfort and the patient’s experience.
According to the American Society of Clinical Hypnosis, physicians and dentists used the power of words through hypnosis as anesthesia before the first chemical general anesthetic agent, ether, was used for surgery in the 1840s, followed by chloroform. Prior to this time, British and Scottish physicians John Elliotson, James Esdaile, and James Braid performed over 3,000 procedures and surgeries with clinical hypnosis alone. Some may argue that the ancient Egyptians also used hypnosis for their well-described surgeries, as no other anesthetic has been documented. Moreover, there is evidence of “sleep temples” that the ancient Egyptians used for healing.1
This article is not to suggest that our words should replace anesthesia. Many advances in anesthesia and pain control have been made since the time of chloroform. However, being mindful of our words can aid and assist in our surgical and aesthetic procedures where less anesthesia is used: Patients feel more comfortable, they heal faster, and overall, they have a more positive outcome and pleasant physician-patient experience.2
For patients, the skill of the doctor and the outcome of the procedure are of the utmost importance, but, especially in aesthetic dermatology, where some of our procedures are repeated or performed periodically, the positive impact of the entire experience will entrust them with your care long term.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References
1. Mutter, C.B. (1998). History of Hypnosis. (pp. 10-12) “Hypnotic Induction and Suggestion.” Chicago: American Society of Clinical Hypnosis.
2. Burns. 2010 Aug;36(5):639-46.
The words we choose to use prior to procedures can positively or negatively impact a patient’s experience during a procedure and their decision to have the procedure performed. A practical example would include using the word discomfort instead of pain to describe pain that may be associated with a procedure. The root word of discomfort is comfort, which the mind focuses on and creates less of an anxious state than pain.
Obviously, the need to provide proper and realistic expectations, as well as risks and benefits, is of utmost importance when obtaining informed consent. The words used can put a patient’s mind at ease or cause further anxiety about ideas of needles, scalpels, pain, risk of infection, and bleeding that are part of our everyday procedures.
Judith Thomas, DDS, a dentist in Virginia who is trained in clinical hypnosis, once described the power of the word but. People will often put more emphasis in their minds on what is said after the word but than on what is said before. For example, in a romantic relationship context, saying “I love you, but you drive me crazy” has a different impact than “You drive me crazy, but I love you.” The focus tends to stay on the “I love you” portion more when it is said last, after the “but.”
The same phenomenon can happen when we discuss procedures with our patients. When a medical assistant performs phlebotomy or when we as doctors are about to perform an injection, instead of saying this is going to hurt, another way to phrase it would be “In a moment you may feel something, but it doesn’t have to bother you” or “You may experience some discomfort, but it will resolve quickly.” Something I’ve said for years to patients before surgery is “You may feel a little stinging as the anesthetic goes in, after that you may feel me touching you, but nothing uncomfortable.” I guess I had been intuitively using this technique for years, without knowing the impact of the word “but.” Perhaps now that I am more mindful of it, I will be even more mindful of how I phrase these terms. We, in addition to our nurses and medical assistants, can use these techniques to enhance patient comfort and the patient’s experience.
According to the American Society of Clinical Hypnosis, physicians and dentists used the power of words through hypnosis as anesthesia before the first chemical general anesthetic agent, ether, was used for surgery in the 1840s, followed by chloroform. Prior to this time, British and Scottish physicians John Elliotson, James Esdaile, and James Braid performed over 3,000 procedures and surgeries with clinical hypnosis alone. Some may argue that the ancient Egyptians also used hypnosis for their well-described surgeries, as no other anesthetic has been documented. Moreover, there is evidence of “sleep temples” that the ancient Egyptians used for healing.1
This article is not to suggest that our words should replace anesthesia. Many advances in anesthesia and pain control have been made since the time of chloroform. However, being mindful of our words can aid and assist in our surgical and aesthetic procedures where less anesthesia is used: Patients feel more comfortable, they heal faster, and overall, they have a more positive outcome and pleasant physician-patient experience.2
For patients, the skill of the doctor and the outcome of the procedure are of the utmost importance, but, especially in aesthetic dermatology, where some of our procedures are repeated or performed periodically, the positive impact of the entire experience will entrust them with your care long term.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References
1. Mutter, C.B. (1998). History of Hypnosis. (pp. 10-12) “Hypnotic Induction and Suggestion.” Chicago: American Society of Clinical Hypnosis.
2. Burns. 2010 Aug;36(5):639-46.
The words we choose to use prior to procedures can positively or negatively impact a patient’s experience during a procedure and their decision to have the procedure performed. A practical example would include using the word discomfort instead of pain to describe pain that may be associated with a procedure. The root word of discomfort is comfort, which the mind focuses on and creates less of an anxious state than pain.
Obviously, the need to provide proper and realistic expectations, as well as risks and benefits, is of utmost importance when obtaining informed consent. The words used can put a patient’s mind at ease or cause further anxiety about ideas of needles, scalpels, pain, risk of infection, and bleeding that are part of our everyday procedures.
Judith Thomas, DDS, a dentist in Virginia who is trained in clinical hypnosis, once described the power of the word but. People will often put more emphasis in their minds on what is said after the word but than on what is said before. For example, in a romantic relationship context, saying “I love you, but you drive me crazy” has a different impact than “You drive me crazy, but I love you.” The focus tends to stay on the “I love you” portion more when it is said last, after the “but.”
The same phenomenon can happen when we discuss procedures with our patients. When a medical assistant performs phlebotomy or when we as doctors are about to perform an injection, instead of saying this is going to hurt, another way to phrase it would be “In a moment you may feel something, but it doesn’t have to bother you” or “You may experience some discomfort, but it will resolve quickly.” Something I’ve said for years to patients before surgery is “You may feel a little stinging as the anesthetic goes in, after that you may feel me touching you, but nothing uncomfortable.” I guess I had been intuitively using this technique for years, without knowing the impact of the word “but.” Perhaps now that I am more mindful of it, I will be even more mindful of how I phrase these terms. We, in addition to our nurses and medical assistants, can use these techniques to enhance patient comfort and the patient’s experience.
According to the American Society of Clinical Hypnosis, physicians and dentists used the power of words through hypnosis as anesthesia before the first chemical general anesthetic agent, ether, was used for surgery in the 1840s, followed by chloroform. Prior to this time, British and Scottish physicians John Elliotson, James Esdaile, and James Braid performed over 3,000 procedures and surgeries with clinical hypnosis alone. Some may argue that the ancient Egyptians also used hypnosis for their well-described surgeries, as no other anesthetic has been documented. Moreover, there is evidence of “sleep temples” that the ancient Egyptians used for healing.1
This article is not to suggest that our words should replace anesthesia. Many advances in anesthesia and pain control have been made since the time of chloroform. However, being mindful of our words can aid and assist in our surgical and aesthetic procedures where less anesthesia is used: Patients feel more comfortable, they heal faster, and overall, they have a more positive outcome and pleasant physician-patient experience.2
For patients, the skill of the doctor and the outcome of the procedure are of the utmost importance, but, especially in aesthetic dermatology, where some of our procedures are repeated or performed periodically, the positive impact of the entire experience will entrust them with your care long term.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References
1. Mutter, C.B. (1998). History of Hypnosis. (pp. 10-12) “Hypnotic Induction and Suggestion.” Chicago: American Society of Clinical Hypnosis.
2. Burns. 2010 Aug;36(5):639-46.
Ursolic acid
Ursolic acid (3beta-hydroxy-urs-12-en-28-oic acid) is a pentacyclic triterpenoid found naturally in apples, waxy berries, rosemary, oregano, and several other plants and herbs used in medicine and the diet.1,2 It is known to have significant antioxidant, anti-inflammatory, and antiproliferative properties, and has also been associated with a wider range of biologic activities, including anticancer, antimicrobial, antitumor, antiwrinkle, anti-HIV, cytotoxic, and hepatoprotective.3,4 In addition, ursolic acid is the focus of human clinical trials for potential uses in cancer and skin wrinkles.4 While this triterpenoid is known to suppress tumor formation and viability in various kinds of cancer, including skin cancer, several forms of cancer are resistant to ursolic acid.
Anti-inflammatory activity
In a 2013 study of the antibacterial and anti-inflammatory effects of Syzygium jambos on acne, Sharma et al. found that ursolic acid was one of the constituents of the leaf extracts that contributed to a significant suppression of the release of inflammatory cytokines interleukin (IL)-8 and tumor necrosis factor-alpha.5
In 2010, Yang et al. identified ursolic acid as a key constituent of Acanthopanax koreanum fruit, a popular fruit in Jeju Island, South Korea, extracts of which they found to exhibit significant anti-inflammatory activity and suitability as a topical agent.6
Yasukawa et al. conducted an in vivo two-stage carcinogenesis test in mice in 2009 in which extracts of the branches of Hippophae rhamnoides displayed significant antitumor activity after initiation with 7,12-dimethylbenz[a]anthracene (DMBA) and promotion with 12-O-tetradecanoylphorbol-13-acetate (TPA). Ursolic acid and (-)-epigallocatechin were the constituents found to have the greatest inhibitory effects on TPA-induced inflammation.7
A 2002 study by Chattopadhyay et al. revealed that the ursolic acid present in Mallotus peltatus extract (long used in traditional folk medicine to treat skin infections and intestinal disorders) may partially account for the broad anti-inflammatory and antimicrobial activity of the plant.9
In 1997, Máñez et al. noted that ursolic acid was among two of the four selected natural triterpenoids tested and found to be significantly effective against inflammation in a TPA multiple-dose model of chronic skin inflammation.10
Anticancer activity
In 2015, Cho et al. reported on the inhibitory effects on skin tumor promotion from the topical application of ursolic acid, resveratrol, or the combination of the two prior to TPA treatment on mouse skin. The combination of the two botanical agents yielded the strongest suppression of TPA-induced epidermal hyperproliferation, skin inflammation, inflammatory gene expression, and skin tumor promotion.11
In another study that year buttressing the combination of the two botanical agents, Junco et al. demonstrated that chloroquine could be used to sensitize B16F10 metastatic mouse melanoma to the anticancer activities of ursolic acid and resveratrol. The investigators concluded that the combination of ursolic acid or resveratrol with chloroquine has potential for inclusion in melanoma treatment in humans.12 Previously, Junco et al. observed that the anti–skin cancer effects of ursolic acid are augmented by P-glycoprotein inhibitors, and that ursolic acid and the stilbene resveratrol, a potent antioxidant, work synergistically, although not by blocking P-glycoprotein. The investigators suggested that ursolic acid along with resveratrol and/or P-glycoprotein inhibitors have potential as effective anti–skin cancer regimens.
In 2014, Lee et al. showed that ursolic acid can differentially modulate apoptosis in cutaneous melanoma and retinal pigment epithelial cells exposed to ultraviolet to visible broadband radiation, exhibiting the potential to protect normal cells while sensitizing melanoma cells to the effects of UV radiation.13 These findings supported earlier work by the team showing that pretreatment of human cells derived from a malignant skin melanoma markedly enhanced the sensitivity of melanoma cells to UV radiation, while providing some photoprotection to retinal pigment epithelium.
Also that year, Soica et al. demonstrated, using in vitro tests and in vivo skin cancer models, that the mixture of oleanolic and ursolic acids and in complex with cyclodextrin rendered a synergistic antitumor activity.14
A year earlier, Kowalczyk et al. showed that the combined action of phytochemicals – dietary calcium D-glucarate and topical ursolic acid and resveratrol – was effective in suppressing the initiation (with 7,12-dimethylbenz[a]anthracene [DMBA]) and promotion (with TPA) of skin tumorigenesis in SENCAR mice. Ursolic acid alone or in combination with calcium D-glucarate significantly diminished epidermal hyperplasia when applied during promotion. All of the antipromotion protocols led to significant decreases in cyclooxygenase-2 and interleukin (IL)-6 expression. The researchers concluded that ursolic acid strongly inhibits skin tumor promotion and inflammatory signaling, and warrants attention as a potential preventive agent against skin and other epithelial cancers.15 Kowalczyk et al. had previously found that ursolic acid and other phytochemicals displayed significant in vitro and in vivo antioxidant and antitumorigenic activity, inhibiting murine skin carcinogenesis by blunting tumor initiation and tumor promotion/progression.16
In 2006, beta-ursolic acid isolated from Salvia officinalis was found by Jedinák et al. to be effective in suppressing lung colonization of beta16 mouse melanoma cells in vivo.17
Huang et al. showed in 1994 that extracts of the leaves of Rosmarinus officinalis (rosemary) were effective in suppressing tumor initiation and promotion in a two-stage skin tumorigenesis mouse model. Topically applied ursolic acid isolated from the leaves was found to hinder TPA-induced ear inflammation, ornithine decarboxylase activity, and tumor promotion. The number of tumors per mouse also declined significantly due to the topical application of ursolic acid concurrent with twice weekly application of the tumor-promoter TPA in DMBA-initiated mice.18
Antiaging and other activities
In 2015, Herndon et al. conducted an open-label clinical trial in 37 females (aged 35-60 years) to ascertain the effectiveness of an anti-aging moisturizer containing Astragalus membranaceus root extract, a peptide blend including palmitoyl tripeptide-38, standardized rosemary leaf extract (ursolic acid), tetrahexyldecyl ascorbate (THD ascorbate), and ubiquinone (coenzyme Q10). Subjects were instructed to apply the moisturizer once in the morning and once in the evening, and were assessed at baseline, and after 4, 8, and 12 weeks of twice daily application. Clinical evaluations after 8 weeks revealed a statistically significant improvement in all grading parameters (fine lines and wrinkles, clarity/brightness, visual roughness, tactile roughness, redness, hyperpigmentation, and overall appearance), with even more pronounced improvement at 12 weeks. The product was found to be mild and well tolerated, and digital photography reinforced clinical assessments and self-evaluations.19
Lee et al. reported in 2012 on in vitro results suggesting that ursolic acid was effective as an inhibitor of matrix metalloproteinase (MMP)-1 after UVB exposure and was more effective than retinoic acid.20
Based on studies with hairless mice, Lim et al. found in 2007 that ursolic and oleanolic acids can enhance the recovery of skin barrier function and, via peroxisome proliferator-activated receptor-alpha, spur epidermal keratinocyte differentiation. They concluded that both acids have potential for use as agents to promote epidermal permeability barrier function.21
In 2003, Soo et al. observed that pretreatment with ursolic acid inhibited UVA-induced oxidative stress and activation and expression of MMP-2 in HaCaT human keratinocytes. They concluded that ursolic acid may merit attention for the prevention of UVA-induced photoaging.22
Three years earlier, Yarosh et al. showed that liposomes containing ursolic acid augmented ceramide content in cultured normal human epidermal keratinocytes and collagen content in cultured normal human dermal fibroblasts. Over an 11-day period, clinical tests with the ursolic acid–containing liposome (Merotaine) revealed increases in the ceramide content in human skin.23 Two years later, many of the same researchers duplicated their results. This new study also demonstrated that ursolic acid liposomes raise ceramide levels in normal human epidermal keratinocytes, in contrast to the effects of retinoic acid, earlier shown to reduce such levels. They concluded that ursolic acid liposomes show promise for use alone or in combination to replenish or maintain cutaneous ceramide and collagen levels.24 Notably, ursolic acid is incorporated into topical oils and creams intended to confer rejuvenating effects to the skin.
Conclusion
Ursolic acid is a compelling ingredient. I especially will be interested in the results of ongoing human clinical trials of this triterpenoid for treating cancer and skin wrinkles. As it is, ursolic acid is known to exert significant inhibitory activity against tumor formation and tumor cell viability in the laboratory. Given its wide range of biologic activity, and some promising cutaneous results, there is reason to believe that ursolic acid has the potential to play an increasingly useful role in topical skin care agents and dermatologic practice.
References
1. J Dermatol. 2007 Sep;34(9):625-34.
2. Folia Histochem Cytobiol. 2011;49(4):664-9.
3. J Cosmet Dermatol. 2004 Jan;3(1):26-34.
4. J Enzyme Inhib Med Chem. 2011 Oct;26(5):616-42.
5. BMC Complement Altern Med. 2013 Oct 29;13:292.
6. J Biomed Biotechnol. 2010;2010:715739.
7. Fitoterapia. 2009 Apr;80(3):164-7.
8. Biosci Biotechnol Biochem. 2004 Jan;68(1):85-90.
9. J Ethnopharmacol. 2002 Oct;82(2-3):229-37.
10. Eur J Pharmacol. 1997 Sep 3;334(1):103-5.
11. Cancer Prev Res (Phila). 2015 Sep;8(9):817-25.
12. Melanoma Res. 2015 Apr;25(2):103-12.
13. Apoptosis. 2014 May;19(5):816-28.
14. Molecules. 2014 Apr 17;19(4):4924-40.
15. Int J Oncol. 2013 Sep;43(3):911-8.
16. Carcinogenesis. 2009 Jun;30(6):1008-15.
17. Z Naturforsch C. 2006 Nov-Dec;61(11-12):777-82.
18. Cancer Res. 1994 Feb 1;54(3):701-8.
19. J Drugs Dermatol. 2015 Jul;14(7):699-704.
20. Bioorg Khim. 2012 May-Jun;38(3):374-81.
21. J Dermatol. 2007;34(9):625-34.
22. Eur J Pharmacol. 2003 Aug 29;476(3):173-8.
23. Horm Res. 2000;54(5-6):318-21.
24. Arch Dermatol Res. 2002 Jan;293(11):569-75.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Ursolic acid (3beta-hydroxy-urs-12-en-28-oic acid) is a pentacyclic triterpenoid found naturally in apples, waxy berries, rosemary, oregano, and several other plants and herbs used in medicine and the diet.1,2 It is known to have significant antioxidant, anti-inflammatory, and antiproliferative properties, and has also been associated with a wider range of biologic activities, including anticancer, antimicrobial, antitumor, antiwrinkle, anti-HIV, cytotoxic, and hepatoprotective.3,4 In addition, ursolic acid is the focus of human clinical trials for potential uses in cancer and skin wrinkles.4 While this triterpenoid is known to suppress tumor formation and viability in various kinds of cancer, including skin cancer, several forms of cancer are resistant to ursolic acid.
Anti-inflammatory activity
In a 2013 study of the antibacterial and anti-inflammatory effects of Syzygium jambos on acne, Sharma et al. found that ursolic acid was one of the constituents of the leaf extracts that contributed to a significant suppression of the release of inflammatory cytokines interleukin (IL)-8 and tumor necrosis factor-alpha.5
In 2010, Yang et al. identified ursolic acid as a key constituent of Acanthopanax koreanum fruit, a popular fruit in Jeju Island, South Korea, extracts of which they found to exhibit significant anti-inflammatory activity and suitability as a topical agent.6
Yasukawa et al. conducted an in vivo two-stage carcinogenesis test in mice in 2009 in which extracts of the branches of Hippophae rhamnoides displayed significant antitumor activity after initiation with 7,12-dimethylbenz[a]anthracene (DMBA) and promotion with 12-O-tetradecanoylphorbol-13-acetate (TPA). Ursolic acid and (-)-epigallocatechin were the constituents found to have the greatest inhibitory effects on TPA-induced inflammation.7
A 2002 study by Chattopadhyay et al. revealed that the ursolic acid present in Mallotus peltatus extract (long used in traditional folk medicine to treat skin infections and intestinal disorders) may partially account for the broad anti-inflammatory and antimicrobial activity of the plant.9
In 1997, Máñez et al. noted that ursolic acid was among two of the four selected natural triterpenoids tested and found to be significantly effective against inflammation in a TPA multiple-dose model of chronic skin inflammation.10
Anticancer activity
In 2015, Cho et al. reported on the inhibitory effects on skin tumor promotion from the topical application of ursolic acid, resveratrol, or the combination of the two prior to TPA treatment on mouse skin. The combination of the two botanical agents yielded the strongest suppression of TPA-induced epidermal hyperproliferation, skin inflammation, inflammatory gene expression, and skin tumor promotion.11
In another study that year buttressing the combination of the two botanical agents, Junco et al. demonstrated that chloroquine could be used to sensitize B16F10 metastatic mouse melanoma to the anticancer activities of ursolic acid and resveratrol. The investigators concluded that the combination of ursolic acid or resveratrol with chloroquine has potential for inclusion in melanoma treatment in humans.12 Previously, Junco et al. observed that the anti–skin cancer effects of ursolic acid are augmented by P-glycoprotein inhibitors, and that ursolic acid and the stilbene resveratrol, a potent antioxidant, work synergistically, although not by blocking P-glycoprotein. The investigators suggested that ursolic acid along with resveratrol and/or P-glycoprotein inhibitors have potential as effective anti–skin cancer regimens.
In 2014, Lee et al. showed that ursolic acid can differentially modulate apoptosis in cutaneous melanoma and retinal pigment epithelial cells exposed to ultraviolet to visible broadband radiation, exhibiting the potential to protect normal cells while sensitizing melanoma cells to the effects of UV radiation.13 These findings supported earlier work by the team showing that pretreatment of human cells derived from a malignant skin melanoma markedly enhanced the sensitivity of melanoma cells to UV radiation, while providing some photoprotection to retinal pigment epithelium.
Also that year, Soica et al. demonstrated, using in vitro tests and in vivo skin cancer models, that the mixture of oleanolic and ursolic acids and in complex with cyclodextrin rendered a synergistic antitumor activity.14
A year earlier, Kowalczyk et al. showed that the combined action of phytochemicals – dietary calcium D-glucarate and topical ursolic acid and resveratrol – was effective in suppressing the initiation (with 7,12-dimethylbenz[a]anthracene [DMBA]) and promotion (with TPA) of skin tumorigenesis in SENCAR mice. Ursolic acid alone or in combination with calcium D-glucarate significantly diminished epidermal hyperplasia when applied during promotion. All of the antipromotion protocols led to significant decreases in cyclooxygenase-2 and interleukin (IL)-6 expression. The researchers concluded that ursolic acid strongly inhibits skin tumor promotion and inflammatory signaling, and warrants attention as a potential preventive agent against skin and other epithelial cancers.15 Kowalczyk et al. had previously found that ursolic acid and other phytochemicals displayed significant in vitro and in vivo antioxidant and antitumorigenic activity, inhibiting murine skin carcinogenesis by blunting tumor initiation and tumor promotion/progression.16
In 2006, beta-ursolic acid isolated from Salvia officinalis was found by Jedinák et al. to be effective in suppressing lung colonization of beta16 mouse melanoma cells in vivo.17
Huang et al. showed in 1994 that extracts of the leaves of Rosmarinus officinalis (rosemary) were effective in suppressing tumor initiation and promotion in a two-stage skin tumorigenesis mouse model. Topically applied ursolic acid isolated from the leaves was found to hinder TPA-induced ear inflammation, ornithine decarboxylase activity, and tumor promotion. The number of tumors per mouse also declined significantly due to the topical application of ursolic acid concurrent with twice weekly application of the tumor-promoter TPA in DMBA-initiated mice.18
Antiaging and other activities
In 2015, Herndon et al. conducted an open-label clinical trial in 37 females (aged 35-60 years) to ascertain the effectiveness of an anti-aging moisturizer containing Astragalus membranaceus root extract, a peptide blend including palmitoyl tripeptide-38, standardized rosemary leaf extract (ursolic acid), tetrahexyldecyl ascorbate (THD ascorbate), and ubiquinone (coenzyme Q10). Subjects were instructed to apply the moisturizer once in the morning and once in the evening, and were assessed at baseline, and after 4, 8, and 12 weeks of twice daily application. Clinical evaluations after 8 weeks revealed a statistically significant improvement in all grading parameters (fine lines and wrinkles, clarity/brightness, visual roughness, tactile roughness, redness, hyperpigmentation, and overall appearance), with even more pronounced improvement at 12 weeks. The product was found to be mild and well tolerated, and digital photography reinforced clinical assessments and self-evaluations.19
Lee et al. reported in 2012 on in vitro results suggesting that ursolic acid was effective as an inhibitor of matrix metalloproteinase (MMP)-1 after UVB exposure and was more effective than retinoic acid.20
Based on studies with hairless mice, Lim et al. found in 2007 that ursolic and oleanolic acids can enhance the recovery of skin barrier function and, via peroxisome proliferator-activated receptor-alpha, spur epidermal keratinocyte differentiation. They concluded that both acids have potential for use as agents to promote epidermal permeability barrier function.21
In 2003, Soo et al. observed that pretreatment with ursolic acid inhibited UVA-induced oxidative stress and activation and expression of MMP-2 in HaCaT human keratinocytes. They concluded that ursolic acid may merit attention for the prevention of UVA-induced photoaging.22
Three years earlier, Yarosh et al. showed that liposomes containing ursolic acid augmented ceramide content in cultured normal human epidermal keratinocytes and collagen content in cultured normal human dermal fibroblasts. Over an 11-day period, clinical tests with the ursolic acid–containing liposome (Merotaine) revealed increases in the ceramide content in human skin.23 Two years later, many of the same researchers duplicated their results. This new study also demonstrated that ursolic acid liposomes raise ceramide levels in normal human epidermal keratinocytes, in contrast to the effects of retinoic acid, earlier shown to reduce such levels. They concluded that ursolic acid liposomes show promise for use alone or in combination to replenish or maintain cutaneous ceramide and collagen levels.24 Notably, ursolic acid is incorporated into topical oils and creams intended to confer rejuvenating effects to the skin.
Conclusion
Ursolic acid is a compelling ingredient. I especially will be interested in the results of ongoing human clinical trials of this triterpenoid for treating cancer and skin wrinkles. As it is, ursolic acid is known to exert significant inhibitory activity against tumor formation and tumor cell viability in the laboratory. Given its wide range of biologic activity, and some promising cutaneous results, there is reason to believe that ursolic acid has the potential to play an increasingly useful role in topical skin care agents and dermatologic practice.
References
1. J Dermatol. 2007 Sep;34(9):625-34.
2. Folia Histochem Cytobiol. 2011;49(4):664-9.
3. J Cosmet Dermatol. 2004 Jan;3(1):26-34.
4. J Enzyme Inhib Med Chem. 2011 Oct;26(5):616-42.
5. BMC Complement Altern Med. 2013 Oct 29;13:292.
6. J Biomed Biotechnol. 2010;2010:715739.
7. Fitoterapia. 2009 Apr;80(3):164-7.
8. Biosci Biotechnol Biochem. 2004 Jan;68(1):85-90.
9. J Ethnopharmacol. 2002 Oct;82(2-3):229-37.
10. Eur J Pharmacol. 1997 Sep 3;334(1):103-5.
11. Cancer Prev Res (Phila). 2015 Sep;8(9):817-25.
12. Melanoma Res. 2015 Apr;25(2):103-12.
13. Apoptosis. 2014 May;19(5):816-28.
14. Molecules. 2014 Apr 17;19(4):4924-40.
15. Int J Oncol. 2013 Sep;43(3):911-8.
16. Carcinogenesis. 2009 Jun;30(6):1008-15.
17. Z Naturforsch C. 2006 Nov-Dec;61(11-12):777-82.
18. Cancer Res. 1994 Feb 1;54(3):701-8.
19. J Drugs Dermatol. 2015 Jul;14(7):699-704.
20. Bioorg Khim. 2012 May-Jun;38(3):374-81.
21. J Dermatol. 2007;34(9):625-34.
22. Eur J Pharmacol. 2003 Aug 29;476(3):173-8.
23. Horm Res. 2000;54(5-6):318-21.
24. Arch Dermatol Res. 2002 Jan;293(11):569-75.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Ursolic acid (3beta-hydroxy-urs-12-en-28-oic acid) is a pentacyclic triterpenoid found naturally in apples, waxy berries, rosemary, oregano, and several other plants and herbs used in medicine and the diet.1,2 It is known to have significant antioxidant, anti-inflammatory, and antiproliferative properties, and has also been associated with a wider range of biologic activities, including anticancer, antimicrobial, antitumor, antiwrinkle, anti-HIV, cytotoxic, and hepatoprotective.3,4 In addition, ursolic acid is the focus of human clinical trials for potential uses in cancer and skin wrinkles.4 While this triterpenoid is known to suppress tumor formation and viability in various kinds of cancer, including skin cancer, several forms of cancer are resistant to ursolic acid.
Anti-inflammatory activity
In a 2013 study of the antibacterial and anti-inflammatory effects of Syzygium jambos on acne, Sharma et al. found that ursolic acid was one of the constituents of the leaf extracts that contributed to a significant suppression of the release of inflammatory cytokines interleukin (IL)-8 and tumor necrosis factor-alpha.5
In 2010, Yang et al. identified ursolic acid as a key constituent of Acanthopanax koreanum fruit, a popular fruit in Jeju Island, South Korea, extracts of which they found to exhibit significant anti-inflammatory activity and suitability as a topical agent.6
Yasukawa et al. conducted an in vivo two-stage carcinogenesis test in mice in 2009 in which extracts of the branches of Hippophae rhamnoides displayed significant antitumor activity after initiation with 7,12-dimethylbenz[a]anthracene (DMBA) and promotion with 12-O-tetradecanoylphorbol-13-acetate (TPA). Ursolic acid and (-)-epigallocatechin were the constituents found to have the greatest inhibitory effects on TPA-induced inflammation.7
A 2002 study by Chattopadhyay et al. revealed that the ursolic acid present in Mallotus peltatus extract (long used in traditional folk medicine to treat skin infections and intestinal disorders) may partially account for the broad anti-inflammatory and antimicrobial activity of the plant.9
In 1997, Máñez et al. noted that ursolic acid was among two of the four selected natural triterpenoids tested and found to be significantly effective against inflammation in a TPA multiple-dose model of chronic skin inflammation.10
Anticancer activity
In 2015, Cho et al. reported on the inhibitory effects on skin tumor promotion from the topical application of ursolic acid, resveratrol, or the combination of the two prior to TPA treatment on mouse skin. The combination of the two botanical agents yielded the strongest suppression of TPA-induced epidermal hyperproliferation, skin inflammation, inflammatory gene expression, and skin tumor promotion.11
In another study that year buttressing the combination of the two botanical agents, Junco et al. demonstrated that chloroquine could be used to sensitize B16F10 metastatic mouse melanoma to the anticancer activities of ursolic acid and resveratrol. The investigators concluded that the combination of ursolic acid or resveratrol with chloroquine has potential for inclusion in melanoma treatment in humans.12 Previously, Junco et al. observed that the anti–skin cancer effects of ursolic acid are augmented by P-glycoprotein inhibitors, and that ursolic acid and the stilbene resveratrol, a potent antioxidant, work synergistically, although not by blocking P-glycoprotein. The investigators suggested that ursolic acid along with resveratrol and/or P-glycoprotein inhibitors have potential as effective anti–skin cancer regimens.
In 2014, Lee et al. showed that ursolic acid can differentially modulate apoptosis in cutaneous melanoma and retinal pigment epithelial cells exposed to ultraviolet to visible broadband radiation, exhibiting the potential to protect normal cells while sensitizing melanoma cells to the effects of UV radiation.13 These findings supported earlier work by the team showing that pretreatment of human cells derived from a malignant skin melanoma markedly enhanced the sensitivity of melanoma cells to UV radiation, while providing some photoprotection to retinal pigment epithelium.
Also that year, Soica et al. demonstrated, using in vitro tests and in vivo skin cancer models, that the mixture of oleanolic and ursolic acids and in complex with cyclodextrin rendered a synergistic antitumor activity.14
A year earlier, Kowalczyk et al. showed that the combined action of phytochemicals – dietary calcium D-glucarate and topical ursolic acid and resveratrol – was effective in suppressing the initiation (with 7,12-dimethylbenz[a]anthracene [DMBA]) and promotion (with TPA) of skin tumorigenesis in SENCAR mice. Ursolic acid alone or in combination with calcium D-glucarate significantly diminished epidermal hyperplasia when applied during promotion. All of the antipromotion protocols led to significant decreases in cyclooxygenase-2 and interleukin (IL)-6 expression. The researchers concluded that ursolic acid strongly inhibits skin tumor promotion and inflammatory signaling, and warrants attention as a potential preventive agent against skin and other epithelial cancers.15 Kowalczyk et al. had previously found that ursolic acid and other phytochemicals displayed significant in vitro and in vivo antioxidant and antitumorigenic activity, inhibiting murine skin carcinogenesis by blunting tumor initiation and tumor promotion/progression.16
In 2006, beta-ursolic acid isolated from Salvia officinalis was found by Jedinák et al. to be effective in suppressing lung colonization of beta16 mouse melanoma cells in vivo.17
Huang et al. showed in 1994 that extracts of the leaves of Rosmarinus officinalis (rosemary) were effective in suppressing tumor initiation and promotion in a two-stage skin tumorigenesis mouse model. Topically applied ursolic acid isolated from the leaves was found to hinder TPA-induced ear inflammation, ornithine decarboxylase activity, and tumor promotion. The number of tumors per mouse also declined significantly due to the topical application of ursolic acid concurrent with twice weekly application of the tumor-promoter TPA in DMBA-initiated mice.18
Antiaging and other activities
In 2015, Herndon et al. conducted an open-label clinical trial in 37 females (aged 35-60 years) to ascertain the effectiveness of an anti-aging moisturizer containing Astragalus membranaceus root extract, a peptide blend including palmitoyl tripeptide-38, standardized rosemary leaf extract (ursolic acid), tetrahexyldecyl ascorbate (THD ascorbate), and ubiquinone (coenzyme Q10). Subjects were instructed to apply the moisturizer once in the morning and once in the evening, and were assessed at baseline, and after 4, 8, and 12 weeks of twice daily application. Clinical evaluations after 8 weeks revealed a statistically significant improvement in all grading parameters (fine lines and wrinkles, clarity/brightness, visual roughness, tactile roughness, redness, hyperpigmentation, and overall appearance), with even more pronounced improvement at 12 weeks. The product was found to be mild and well tolerated, and digital photography reinforced clinical assessments and self-evaluations.19
Lee et al. reported in 2012 on in vitro results suggesting that ursolic acid was effective as an inhibitor of matrix metalloproteinase (MMP)-1 after UVB exposure and was more effective than retinoic acid.20
Based on studies with hairless mice, Lim et al. found in 2007 that ursolic and oleanolic acids can enhance the recovery of skin barrier function and, via peroxisome proliferator-activated receptor-alpha, spur epidermal keratinocyte differentiation. They concluded that both acids have potential for use as agents to promote epidermal permeability barrier function.21
In 2003, Soo et al. observed that pretreatment with ursolic acid inhibited UVA-induced oxidative stress and activation and expression of MMP-2 in HaCaT human keratinocytes. They concluded that ursolic acid may merit attention for the prevention of UVA-induced photoaging.22
Three years earlier, Yarosh et al. showed that liposomes containing ursolic acid augmented ceramide content in cultured normal human epidermal keratinocytes and collagen content in cultured normal human dermal fibroblasts. Over an 11-day period, clinical tests with the ursolic acid–containing liposome (Merotaine) revealed increases in the ceramide content in human skin.23 Two years later, many of the same researchers duplicated their results. This new study also demonstrated that ursolic acid liposomes raise ceramide levels in normal human epidermal keratinocytes, in contrast to the effects of retinoic acid, earlier shown to reduce such levels. They concluded that ursolic acid liposomes show promise for use alone or in combination to replenish or maintain cutaneous ceramide and collagen levels.24 Notably, ursolic acid is incorporated into topical oils and creams intended to confer rejuvenating effects to the skin.
Conclusion
Ursolic acid is a compelling ingredient. I especially will be interested in the results of ongoing human clinical trials of this triterpenoid for treating cancer and skin wrinkles. As it is, ursolic acid is known to exert significant inhibitory activity against tumor formation and tumor cell viability in the laboratory. Given its wide range of biologic activity, and some promising cutaneous results, there is reason to believe that ursolic acid has the potential to play an increasingly useful role in topical skin care agents and dermatologic practice.
References
1. J Dermatol. 2007 Sep;34(9):625-34.
2. Folia Histochem Cytobiol. 2011;49(4):664-9.
3. J Cosmet Dermatol. 2004 Jan;3(1):26-34.
4. J Enzyme Inhib Med Chem. 2011 Oct;26(5):616-42.
5. BMC Complement Altern Med. 2013 Oct 29;13:292.
6. J Biomed Biotechnol. 2010;2010:715739.
7. Fitoterapia. 2009 Apr;80(3):164-7.
8. Biosci Biotechnol Biochem. 2004 Jan;68(1):85-90.
9. J Ethnopharmacol. 2002 Oct;82(2-3):229-37.
10. Eur J Pharmacol. 1997 Sep 3;334(1):103-5.
11. Cancer Prev Res (Phila). 2015 Sep;8(9):817-25.
12. Melanoma Res. 2015 Apr;25(2):103-12.
13. Apoptosis. 2014 May;19(5):816-28.
14. Molecules. 2014 Apr 17;19(4):4924-40.
15. Int J Oncol. 2013 Sep;43(3):911-8.
16. Carcinogenesis. 2009 Jun;30(6):1008-15.
17. Z Naturforsch C. 2006 Nov-Dec;61(11-12):777-82.
18. Cancer Res. 1994 Feb 1;54(3):701-8.
19. J Drugs Dermatol. 2015 Jul;14(7):699-704.
20. Bioorg Khim. 2012 May-Jun;38(3):374-81.
21. J Dermatol. 2007;34(9):625-34.
22. Eur J Pharmacol. 2003 Aug 29;476(3):173-8.
23. Horm Res. 2000;54(5-6):318-21.
24. Arch Dermatol Res. 2002 Jan;293(11):569-75.
Dr. Baumann is chief executive officer of the Baumann Cosmetic & Research Institute in the Design District in Miami. She founded the Cosmetic Dermatology Center at the University of Miami in 1997. Dr. Baumann wrote the textbook “Cosmetic Dermatology: Principles and Practice” (New York: McGraw-Hill, 2002), and a book for consumers, “The Skin Type Solution” (New York: Bantam Dell, 2006). Her latest book, “Cosmeceuticals and Cosmetic Ingredients,” was published in November 2014. Dr. Baumann has received funding for clinical grants from Allergan, Aveeno, Avon Products, Evolus, Galderma, GlaxoSmithKline, Kythera Biopharmaceuticals, Mary Kay, Medicis Pharmaceuticals, Neutrogena, Philosophy, Topix Pharmaceuticals, and Unilever. Dr. Baumann also developed and owns the Baumann Skin Type Solution skin typing systems and related products.
Cosmetic Treatments for Skin of Color: Report From the AAD Meeting
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: Consider alopecia ‘camouflage kits’ to boost patients’ self-esteem
ORLANDO – The hair loss encounter – which can be challenging for both physicians and patients – should address the negative psychological effects of hair loss, including ways to camouflage hair loss, advised Adriana N. Schmidt, MD, a dermatologist in Santa Monica, Calif.
Dermatologists may spend so much time on the work-up – reviewing history regarding medication, lab values, and hair care practices – that they do not spend time to simply say to patients, “I want to help you feel better about yourself, and here’s how,” she said in a video interview at the annual meeting of the American Academy of Dermatology.
“What we can do is offer them a way to camouflage the hair loss,” Dr. Schmidt said. She shared tips that include creating a kit to keep in the office filled with lists of reputable hairpiece vendors and tattoo specialists in the community, as well as sample wigs, cosmetic powders, and other items to show to patients during hair loss consultations. She also offers thoughts on working with new hair styles and stylists to help improve the self-esteem of alopecia patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Dr. Schmidt had no relevant disclosures.
[email protected]
On Twitter @whitneymcknight
ORLANDO – The hair loss encounter – which can be challenging for both physicians and patients – should address the negative psychological effects of hair loss, including ways to camouflage hair loss, advised Adriana N. Schmidt, MD, a dermatologist in Santa Monica, Calif.
Dermatologists may spend so much time on the work-up – reviewing history regarding medication, lab values, and hair care practices – that they do not spend time to simply say to patients, “I want to help you feel better about yourself, and here’s how,” she said in a video interview at the annual meeting of the American Academy of Dermatology.
“What we can do is offer them a way to camouflage the hair loss,” Dr. Schmidt said. She shared tips that include creating a kit to keep in the office filled with lists of reputable hairpiece vendors and tattoo specialists in the community, as well as sample wigs, cosmetic powders, and other items to show to patients during hair loss consultations. She also offers thoughts on working with new hair styles and stylists to help improve the self-esteem of alopecia patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Dr. Schmidt had no relevant disclosures.
[email protected]
On Twitter @whitneymcknight
ORLANDO – The hair loss encounter – which can be challenging for both physicians and patients – should address the negative psychological effects of hair loss, including ways to camouflage hair loss, advised Adriana N. Schmidt, MD, a dermatologist in Santa Monica, Calif.
Dermatologists may spend so much time on the work-up – reviewing history regarding medication, lab values, and hair care practices – that they do not spend time to simply say to patients, “I want to help you feel better about yourself, and here’s how,” she said in a video interview at the annual meeting of the American Academy of Dermatology.
“What we can do is offer them a way to camouflage the hair loss,” Dr. Schmidt said. She shared tips that include creating a kit to keep in the office filled with lists of reputable hairpiece vendors and tattoo specialists in the community, as well as sample wigs, cosmetic powders, and other items to show to patients during hair loss consultations. She also offers thoughts on working with new hair styles and stylists to help improve the self-esteem of alopecia patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Dr. Schmidt had no relevant disclosures.
[email protected]
On Twitter @whitneymcknight
AT AAD 2017
VIDEO: Tips, tricks, and pearls for keloid scar steroid injections
ORLANDO – What are the best ways to avoid atrophy when treating keloid scars with steroid injections? When should a keloid be treated with smaller amounts, but with a stronger steroid concentration? What is the most effective way to avoid precipitation in the syringe?
The answers to these questions – along with other tips, tricks, and pearls for treating keloids, both in patients with skin of color and those with white skin – are provided by Temitayo Ogunleye, MD, of the department of dermatology, University of Pennsylvania, Philadelphia, in a video interview at the annual meeting of the American Academy of Dermatology.
Dr. Ogunleye had no relevant disclosures.
[email protected]
On Twitter @whitneymcknight
ORLANDO – What are the best ways to avoid atrophy when treating keloid scars with steroid injections? When should a keloid be treated with smaller amounts, but with a stronger steroid concentration? What is the most effective way to avoid precipitation in the syringe?
The answers to these questions – along with other tips, tricks, and pearls for treating keloids, both in patients with skin of color and those with white skin – are provided by Temitayo Ogunleye, MD, of the department of dermatology, University of Pennsylvania, Philadelphia, in a video interview at the annual meeting of the American Academy of Dermatology.
Dr. Ogunleye had no relevant disclosures.
[email protected]
On Twitter @whitneymcknight
ORLANDO – What are the best ways to avoid atrophy when treating keloid scars with steroid injections? When should a keloid be treated with smaller amounts, but with a stronger steroid concentration? What is the most effective way to avoid precipitation in the syringe?
The answers to these questions – along with other tips, tricks, and pearls for treating keloids, both in patients with skin of color and those with white skin – are provided by Temitayo Ogunleye, MD, of the department of dermatology, University of Pennsylvania, Philadelphia, in a video interview at the annual meeting of the American Academy of Dermatology.
Dr. Ogunleye had no relevant disclosures.
[email protected]
On Twitter @whitneymcknight
AT AAD 17
VIDEO: Grading tools help set alopecia treatment expectations and monitor progress
ORLANDO – Two hair loss clinical grading tools can help physicians and their female androgenetic alopecia patients set medical treatment expectations, and make tracking progress both easier and more accurate, according to the dermatologist who developed the scales.
The five-point clinical grading scale helps physicians with diagnosing female pattern hair loss and grading the severity, Rodney Sinclair, MD, said in a video interview at the annual meeting of the American Academy of Dermatology. “And you can use that clinical grading scale to monitor the response to treatment” and show patients what to expect with treatment, added Dr. Sinclair, professor and chairman, department of dermatology, Epworth Hospital, Melbourne.
He also discussed a validated hair shedding scale, “a really simple and easy test to use,” with six photographs to help patients determine how much hair they are shedding on a daily basis. “Most women don’t know what’s a normal amount of hair to shed,” he said. In the interview, Dr. Sinclair explains more about the two scales, and how they can be used to obtain clinically relevant information to help guide treatment – and shares his tips for how to work with women who are anxious about their hair loss improvement.
Dr. Sinclair owns the copyrights for the Sinclair Severity Scale. He has no other relevant disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
[email protected]
On Twitter @whitneymcknight
ORLANDO – Two hair loss clinical grading tools can help physicians and their female androgenetic alopecia patients set medical treatment expectations, and make tracking progress both easier and more accurate, according to the dermatologist who developed the scales.
The five-point clinical grading scale helps physicians with diagnosing female pattern hair loss and grading the severity, Rodney Sinclair, MD, said in a video interview at the annual meeting of the American Academy of Dermatology. “And you can use that clinical grading scale to monitor the response to treatment” and show patients what to expect with treatment, added Dr. Sinclair, professor and chairman, department of dermatology, Epworth Hospital, Melbourne.
He also discussed a validated hair shedding scale, “a really simple and easy test to use,” with six photographs to help patients determine how much hair they are shedding on a daily basis. “Most women don’t know what’s a normal amount of hair to shed,” he said. In the interview, Dr. Sinclair explains more about the two scales, and how they can be used to obtain clinically relevant information to help guide treatment – and shares his tips for how to work with women who are anxious about their hair loss improvement.
Dr. Sinclair owns the copyrights for the Sinclair Severity Scale. He has no other relevant disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
[email protected]
On Twitter @whitneymcknight
ORLANDO – Two hair loss clinical grading tools can help physicians and their female androgenetic alopecia patients set medical treatment expectations, and make tracking progress both easier and more accurate, according to the dermatologist who developed the scales.
The five-point clinical grading scale helps physicians with diagnosing female pattern hair loss and grading the severity, Rodney Sinclair, MD, said in a video interview at the annual meeting of the American Academy of Dermatology. “And you can use that clinical grading scale to monitor the response to treatment” and show patients what to expect with treatment, added Dr. Sinclair, professor and chairman, department of dermatology, Epworth Hospital, Melbourne.
He also discussed a validated hair shedding scale, “a really simple and easy test to use,” with six photographs to help patients determine how much hair they are shedding on a daily basis. “Most women don’t know what’s a normal amount of hair to shed,” he said. In the interview, Dr. Sinclair explains more about the two scales, and how they can be used to obtain clinically relevant information to help guide treatment – and shares his tips for how to work with women who are anxious about their hair loss improvement.
Dr. Sinclair owns the copyrights for the Sinclair Severity Scale. He has no other relevant disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
[email protected]
On Twitter @whitneymcknight
AT AAD 17