User login
Hand Rejuvenation With Calcium Hydroxylapatite
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Local Anesthetics in Cosmetic Dermatology
Local anesthesia is a central component of successful interventions in cosmetic dermatology. The number of anesthetic medications and administration techniques has grown in recent years as outpatient cosmetic procedures continue to expand. Pain is a common barrier to cosmetic procedures, and alleviating the fear of painful interventions is critical to patient satisfaction and future visits. To accommodate a multitude of cosmetic interventions, it is important for clinicians to be well versed in applications of topical and regional anesthesia. In this article, we review pain management strategies for use in cosmetic practice.
Local Anesthetics
The sensation of pain is carried to the central nervous system by unmyelinated C nerve fibers. Local anesthetics (LAs) act by blocking fast voltage-gated sodium channels in the cell membrane of the nerve, thereby inhibiting downstream propagation of an action potential and the transmission of painful stimuli.1 The chemical structure of LAs is fundamental to their mechanism of action and metabolism. Local anesthetics contain a lipophilic aromatic group, an intermediate chain, and a hydrophilic amine group. Broadly, agents are classified as amides or esters depending on the chemical group attached to the intermediate chain.2 Amides (eg, lidocaine, bupivacaine, articaine, mepivacaine, prilocaine, levobupivacaine) are metabolized by the hepatic system; esters (eg, procaine, proparacaine, benzocaine, chlorprocaine, tetracaine, cocaine) are metabolized by plasma cholinesterase, which produces para-aminobenzoic acid, a potentially dangerous metabolite that has been implicated in allergic reactions.3
Lidocaine is the most prevalent LA used in dermatology practices. Importantly, lidocaine is a class IB antiarrhythmic agent used in cardiology to treat ventricular arrhythmias.4 As an anesthetic, a maximum dose of 4.5 mg/kg can be administered, increasing to 7.0 mg/kg when mixed with epinephrine; with higher doses, there is a risk for central nervous system and cardiovascular toxicity.5 Initial symptoms of lidocaine toxicity include dizziness, tinnitus, circumoral paresthesia, blurred vision, and a metallic taste in the mouth.6 Systemic absorption of topical anesthetics is heightened across mucosal membranes, and care should be taken when applying over large surface areas.
Allergic reactions to LAs may be local or less frequently systemic. It is important to note that LAs tend to show cross-reactivity within their class rather than across different classes.7 Reactions can be classified as type I or type IV. Type I (IgE-mediated) reactions evolve in minutes to hours, affecting the skin and possibly leading to respiratory and circulatory collapse. Delayed reactions to LAs have increased in recent years, with type IV contact allergy most frequently found in connection with benzocaine and lidocaine.8
Topical Anesthesia
Topical anesthetics are effective and easy to use and are particularly valuable in patients with needle phobia. In certain cases, these medications may be applied by the patient prior to arrival, thereby reducing visit time. Topical agents act on nerve fibers running through the dermis; therefore, efficacy is dependent on successful penetration through the stratum corneum and viable epidermis. To enhance absorption, agents may be applied under an occlusive dressing.
Topical anesthetics are most commonly used for injectable fillers, ablative and nonablative laser resurfacing, laser hair removal, and tattoo removal. The eutectic mixture of 2.5% lidocaine and 2.5% prilocaine as well as topical 4% or 5% lidocaine are the most commonly used US Food and Drug Administration–approved products for topical anesthesia. In addition, several compounded pharmacy products are available.
After 60 minutes of application of the eutectic mixture of 2.5% lidocaine and 2.5% prilocaine, a 3-mm depth of analgesia is reached, and after 120 minutes, a 4.5-mm depth is reached.9 It elicits a biphasic vascular response of vasoconstriction and blanching followed by vasodilation and erythema.10 Most adverse events are mild and transient, but allergic contact dermatitis and contact urticaria have been reported.11-13 In older children and adults, the maximum application area is 200 cm2, with a maximum dose of 20 g used for no longer than 4 hours.
The 4% or 5% lidocaine cream uses a liposomal delivery system, which is designed to improve cutaneous penetration and has been shown to provide longer durations of anesthesia than nonliposomal lidocaine preparations.14 Application should be performed 30 to 60 minutes prior to a procedure. In a study comparing the eutectic mixture of 2.5% lidocaine and 2.5% prilocaine versus lidocaine cream 5% for pain control during laser hair removal with a 1064-nm Nd:YAG laser, no significant differences were found.15 The maximum application area is 100 cm2 in children weighing less than 20 kg. A study of healthy adults demonstrated safety with the use of 30 to 60 g of occluded liposomal lidocaine cream 4%.16
In addition to US Food and Drug Administration–approved products, several compounded pharmacy products are available for topical anesthesia. These formulations include benzocaine-lidocaine-tetracaine gel, tetracaine-adrenaline-cocaine solution, and lidocaine-epinephrine-tetracaine solution. A triple-anesthetic gel, benzocaine-lidocaine-tetracaine is widely used in cosmetic practice. The product has been shown to provide adequate anesthesia for laser resurfacing after 20 minutes without occlusion.17 Of note, compounded anesthetics lack standardization, and different pharmacies may follow their own individual protocols.
Regional Anesthesia
Regional nerve blockade is a useful option for more widespread or complex interventions. Using regional nerve blockade, effective analgesia can be delivered to a target area while avoiding the toxicity and pain associated with numerous anesthetic infiltrations. In addition, there is no distortion of the tissue architecture, allowing for improved visual evaluation during the procedure. Recently, hyaluronic acid fillers have been compounded with lidocaine as a means of reducing procedural pain.
Blocks for Dermal Fillers
Forehead
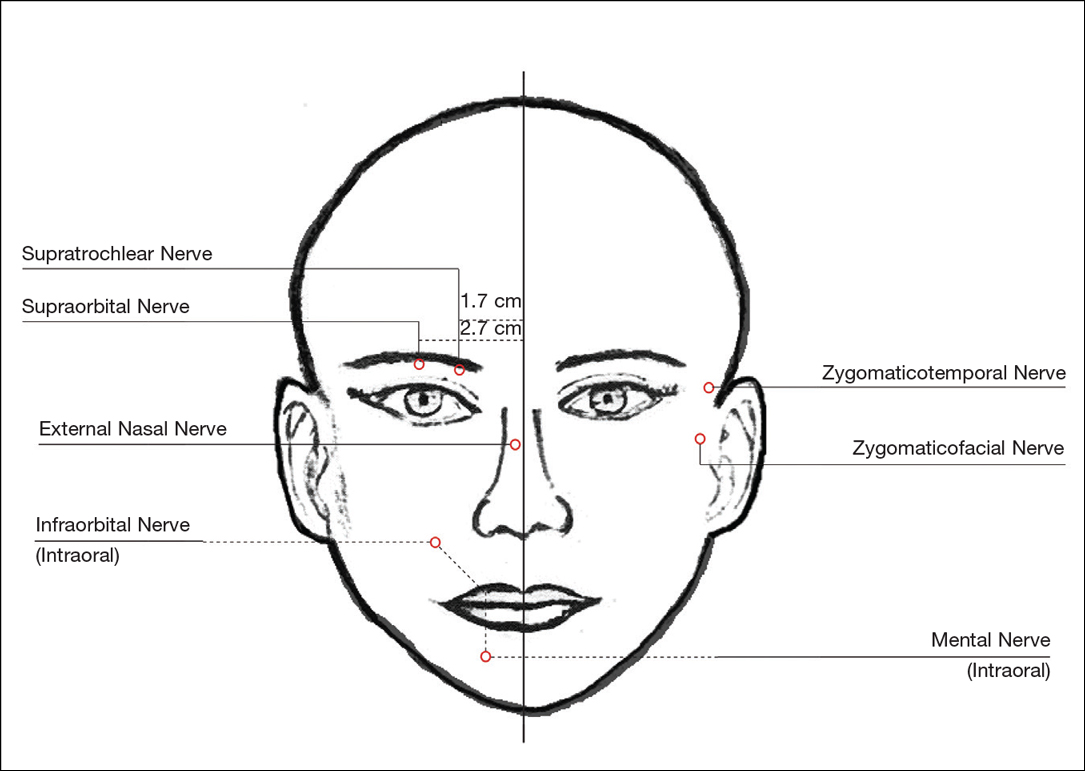
For dermal filler injections of the glabellar and frontalis lines, anesthesia of the forehead may be desired. The supraorbital and supratrochlear nerves supply this area. The supraorbital nerve can be injected at the supraorbital notch, which is measured roughly 2.7 cm from the glabella. The orbital rim should be palpated with the nondominant hand, and 1 to 2 mL of anesthetic should be injected just below the rim (Figure 1). The supratrochlear nerve is located roughly 1.7 cm from the midline and can be similarly injected under the orbital rim with 1 to 2 mL of anesthetic (Figure 1).
Lateral Temple Region
Anesthesia of the zygomaticotemporal nerve can be used to reduce pain from dermal filler injections of the lateral canthal and temporal areas. The nerve is identified by first palpating the zygomaticofrontal suture. A long needle is then inserted posteriorly, immediately behind the concave surface of the lateral orbital rim, and 1 to 2 mL of anesthetic is injected (Figure 1).
Malar Region
Blockade of the zygomaticofacial nerve is commonly performed in conjunction with the zygomaticotemporal nerve and provides anesthesia to the malar region for cheek augmentation procedures. To identify the target area, the junction of the lateral and inferior orbital rim should be palpated. With the needle placed just lateral to this point, 1 to 2 mL of anesthetic is injected (Figure 1).
Blocks for Perioral Fillers
Upper Lips/Nasolabial Folds
Bilateral blockade of the infraorbital nerves provides anesthesia to the upper lip and nasolabial folds prior to filler injections. The infraorbital nerve can be targeted via an intraoral route where it exits the maxilla at the infraorbital foramen. The nerve is anesthetized by palpating the infraorbital ridge and injecting 3 to 5 mL of anesthetic roughly 1 cm below this point on the vertical axis of the midpupillary line (Figure 1). The external nasal nerve, thought to be a branch of cranial nerve V, also may be targeted if there is inadequate anesthesia from the infraorbital block. This nerve is reached by injecting at the osseocartilaginous junction of the nasal bones (Figure 1).
Lower Lips
Blockade of the mental nerve provides anesthesia to the lower lips for augmentation procedures. The mental nerve can be targeted on each side at the mental foramen, which is located below the root of the lower second premolar. Aiming roughly 1 cm below the gumline, 3 to 5 mL of anesthetic is injected intraorally (Figure 1). A transcutaneous approach toward the same target also is possible, though this technique risks visible bruising. Alternatively, the upper or lower lips can be anesthetized using 4 to 5 submucosal injections at evenly spaced intervals between the canine teeth.18
Blocks for Palmoplantar Hyperhidrosis
The treatment of palmoplantar hyperhidrosis benefits from regional blocks. Botulinum toxin has been well established as an effective therapy for the condition.19-21 Given the sensitivity of palmoplantar sites, it is valuable to achieve effective analgesia of the region prior to dermal injections of botulinum toxin.
Wrists
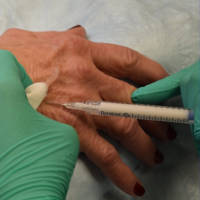
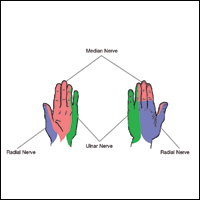
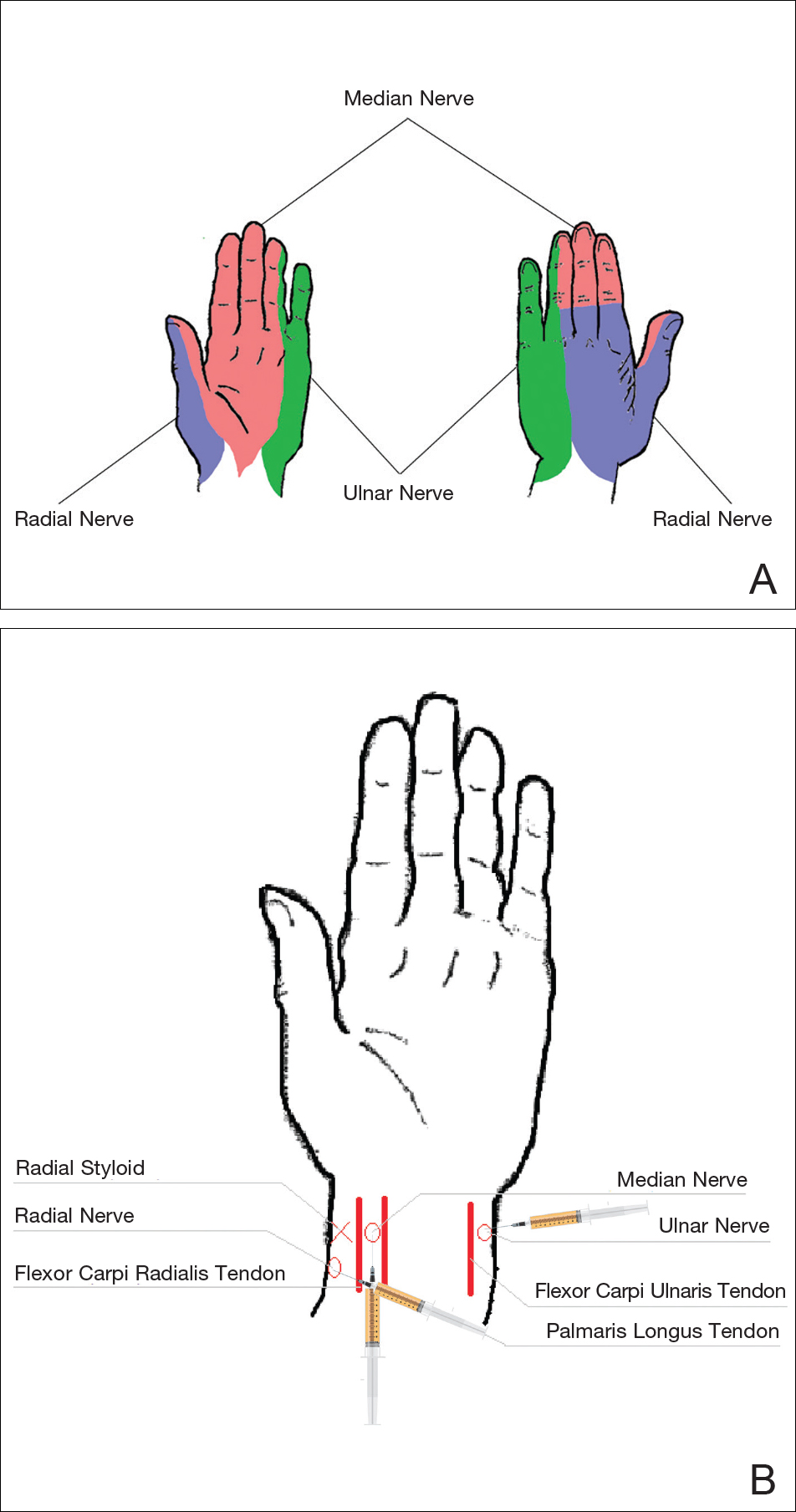
Sensory innervation of the palm is provided by the median, ulnar, and radial nerves (Figure 2A).
The ulnar nerve is anesthetized between the ulnar artery and the flexor carpi ulnaris muscle. The artery is identified by palpation, and special care should be taken to avoid intra-arterial injection. The needle is directed toward the radial styloid, and 3 to 5 mL of anesthetic is injected roughly 1 cm proximal to the wrist crease (Figure 2B).
Anesthesia of the radial nerve can be considered a field block given the numerous small branches that supply the hand. These branches are reached by injecting anesthetic roughly 2 to 3 cm proximal to the radial styloid with the needle aimed medially and extending the injection dorsally (Figure 2B). A total of 4 to 6 mL of anesthetic is used.
Ankles
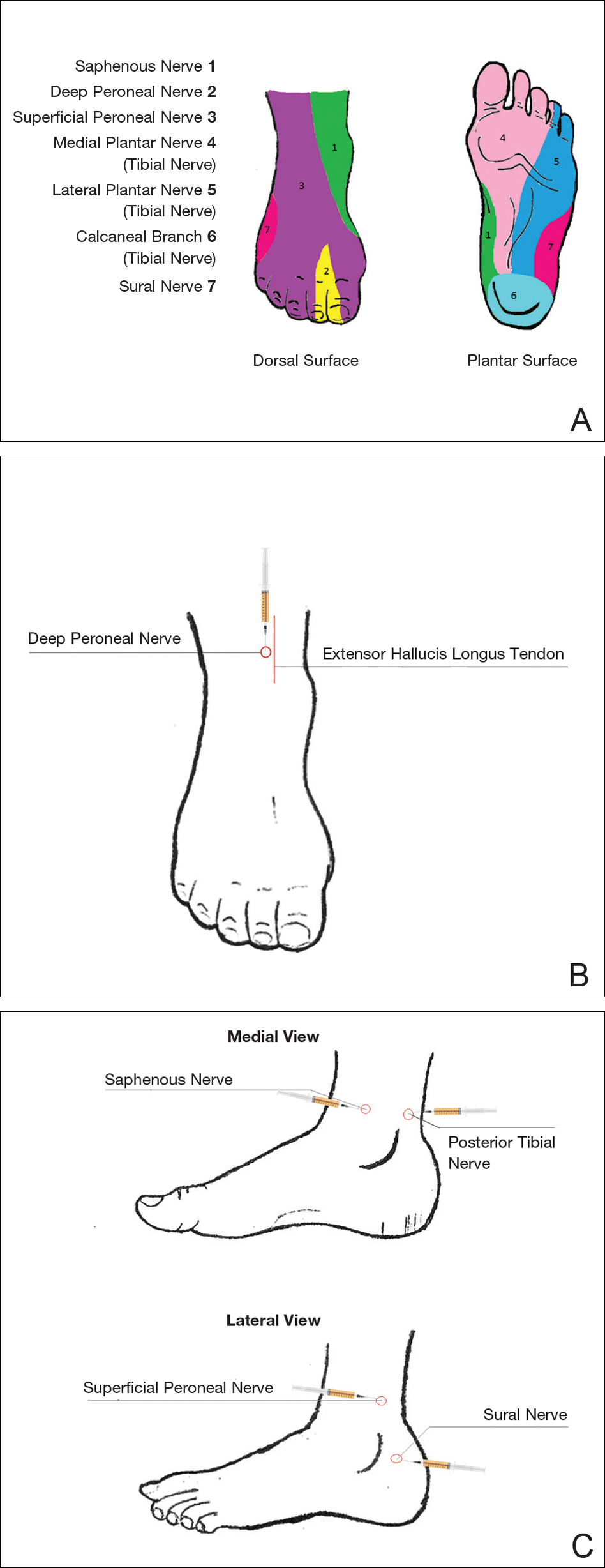
An ankle block provides anesthesia to the dorsal and plantar surfaces of the foot.22 The region is supplied by the superficial peroneal nerve, deep peroneal nerve, sural nerve, saphenous nerve, and branches of the posterior tibial nerve (Figure 3A).
To anesthetize the deep peroneal nerve, the extensor hallucis longus tendon is first identified on the anterior surface of the ankle through dorsiflexion of the toes; the dorsalis pedis artery runs in close proximity. The injection should be placed lateral to the tendon and artery (Figure 3B). The needle should be inserted until bone is reached, withdrawn slightly, and then 3 to 5 mL of anesthetic should be injected. To block the saphenous nerve, the needle can then be directed superficially toward the medial malleolus, and 3 to 5 mL should be injected in a subcutaneous wheal (Figure 3C). To block the superficial peroneal nerve, the needle should then be directed toward the lateral malleolus, and 3 to 5 mL should be injected in a subcutaneous wheal (Figure 3C).
The posterior tibial nerve is located posterior to the medial malleolus. The dorsalis pedis artery can be palpated near this location. The needle should be inserted posterior to the artery, extending until bone is reached (Figure 3C). The needle is then withdrawn slightly, and 3 to 5 mL of anesthetic is injected. Finally, the sural nerve is anesthetized between the Achilles tendon and the lateral malleolus, using 5 mL of anesthetic to raise a subcutaneous wheal (Figure 3C).
Conclusion
Proper pain management is integral to ensuring a positive experience for cosmetic patients. Enhanced knowledge of local anesthetic techniques allows the clinician to provide for a variety of procedural indications and patient preferences. As anesthetic strategies are continually evolving, it is important for practitioners to remain informed of these developments.
- Scholz A. Mechanisms of (local) anaesthetics on voltage-gated sodium and other ion channels. Br J Anaesth. 2002;89:52-61.
- Auletta MJ. Local anesthesia for dermatologic surgery. Semin Dermatol. 1994;13:35-42.
- Park KK, Sharon VR. A review of local anesthetics: minimizing risk and side effects in cutaneous surgery. Dermatol Surg. 2017;43:173-187.
- Reiz S, Nath S. Cardiotoxicity of local anaesthetic agents. Br J Anaesth. 1986;58:736-746.
- Klein JA, Kassarjdian N. Lidocaine toxicity with tumescent liposuction. a case report of probable drug interactions. Dermatol Surg. 1997;23:1169-1174.
- Minkis K, Whittington A, Alam M. Dermatologic surgery emergencies: complications caused by systemic reactions, high-energy systems, and trauma. J Am Acad Dermatol. 2016;75:265-284.
- Morais-Almeida M, Gaspar A, Marinho S, et al. Allergy to local anesthetics of the amide group with tolerance to procaine. Allergy. 2003;58:827-828.
- To D, Kossintseva I, de Gannes G. Lidocaine contact allergy is becoming more prevalent. Dermatol Surg. 2014;40:1367-1372.
- Wahlgren CF, Quiding H. Depth of cutaneous analgesia after application of a eutectic mixture of the local anesthetics lidocaine and prilocaine (EMLA cream). J Am Acad Dermatol. 2000;42:584-588.
- Bjerring P, Andersen PH, Arendt-Nielsen L. Vascular response of human skin after analgesia with EMLA cream. Br J Anaesth. 1989;63:655-660.
- Ismail F, Goldsmith PC. EMLA cream-induced allergic contact dermatitis in a child with thalassaemia major. Contact Dermatitis. 2005;52:111.
- Thakur BK, Murali MR. EMLA cream-induced allergic contact dermatitis: a role for prilocaine as an immunogen. J Allergy Clin Immunol. 1995;95:776-778.
- Waton J, Boulanger A, Trechot PH, et al. Contact urticaria from EMLA cream. Contact Dermatitis. 2004;51:284-287.
- Bucalo BD, Mirikitani EJ, Moy RL. Comparison of skin anesthetic effect of liposomal lidocaine, nonliposomal lidocaine, and EMLA using 30-minute application time. Dermatol Surg. 1998;24:537-541.
- Guardiano RA, Norwood CW. Direct comparison of EMLA versus lidocaine for pain control in Nd:YAG 1,064 nm laser hair removal. Dermatol Surg. 2005;31:396-398.
- Nestor MS. Safety of occluded 4% liposomal lidocaine cream. J Drugs Dermatol. 2006;5:618-620.
- Oni G, Rasko Y, Kenkel J. Topical lidocaine enhanced by laser pretreatment: a safe and effective method of analgesia for facial rejuvenation. Aesthet Surg J. 2013;33:854-861.
- Niamtu J 3rd. Simple technique for lip and nasolabial fold anesthesia for injectable fillers. Dermatol Surg. 2005;31:1330-1332.
- Naumann M, Flachenecker P, Brocker EB, et al. Botulinum toxin for palmar hyperhidrosis. Lancet. 1997;349:252.
- Naumann M, Hofmann U, Bergmann I, et al. Focal hyperhidrosis: effective treatment with intracutaneous botulinum toxin. Arch Dermatol. 1998;134:301-304.
- Shelley WB, Talanin NY, Shelley ED. Botulinum toxin therapy for palmar hyperhidrosis. J Am Acad Dermatol. 1998;38(2, pt 1):227-229.
- Davies T, Karanovic S, Shergill B. Essential regional nerve blocks for the dermatologist: part 2. Clin Exp Dermatol. 2014;39:861-867.
Local anesthesia is a central component of successful interventions in cosmetic dermatology. The number of anesthetic medications and administration techniques has grown in recent years as outpatient cosmetic procedures continue to expand. Pain is a common barrier to cosmetic procedures, and alleviating the fear of painful interventions is critical to patient satisfaction and future visits. To accommodate a multitude of cosmetic interventions, it is important for clinicians to be well versed in applications of topical and regional anesthesia. In this article, we review pain management strategies for use in cosmetic practice.
Local Anesthetics
The sensation of pain is carried to the central nervous system by unmyelinated C nerve fibers. Local anesthetics (LAs) act by blocking fast voltage-gated sodium channels in the cell membrane of the nerve, thereby inhibiting downstream propagation of an action potential and the transmission of painful stimuli.1 The chemical structure of LAs is fundamental to their mechanism of action and metabolism. Local anesthetics contain a lipophilic aromatic group, an intermediate chain, and a hydrophilic amine group. Broadly, agents are classified as amides or esters depending on the chemical group attached to the intermediate chain.2 Amides (eg, lidocaine, bupivacaine, articaine, mepivacaine, prilocaine, levobupivacaine) are metabolized by the hepatic system; esters (eg, procaine, proparacaine, benzocaine, chlorprocaine, tetracaine, cocaine) are metabolized by plasma cholinesterase, which produces para-aminobenzoic acid, a potentially dangerous metabolite that has been implicated in allergic reactions.3
Lidocaine is the most prevalent LA used in dermatology practices. Importantly, lidocaine is a class IB antiarrhythmic agent used in cardiology to treat ventricular arrhythmias.4 As an anesthetic, a maximum dose of 4.5 mg/kg can be administered, increasing to 7.0 mg/kg when mixed with epinephrine; with higher doses, there is a risk for central nervous system and cardiovascular toxicity.5 Initial symptoms of lidocaine toxicity include dizziness, tinnitus, circumoral paresthesia, blurred vision, and a metallic taste in the mouth.6 Systemic absorption of topical anesthetics is heightened across mucosal membranes, and care should be taken when applying over large surface areas.
Allergic reactions to LAs may be local or less frequently systemic. It is important to note that LAs tend to show cross-reactivity within their class rather than across different classes.7 Reactions can be classified as type I or type IV. Type I (IgE-mediated) reactions evolve in minutes to hours, affecting the skin and possibly leading to respiratory and circulatory collapse. Delayed reactions to LAs have increased in recent years, with type IV contact allergy most frequently found in connection with benzocaine and lidocaine.8
Topical Anesthesia
Topical anesthetics are effective and easy to use and are particularly valuable in patients with needle phobia. In certain cases, these medications may be applied by the patient prior to arrival, thereby reducing visit time. Topical agents act on nerve fibers running through the dermis; therefore, efficacy is dependent on successful penetration through the stratum corneum and viable epidermis. To enhance absorption, agents may be applied under an occlusive dressing.
Topical anesthetics are most commonly used for injectable fillers, ablative and nonablative laser resurfacing, laser hair removal, and tattoo removal. The eutectic mixture of 2.5% lidocaine and 2.5% prilocaine as well as topical 4% or 5% lidocaine are the most commonly used US Food and Drug Administration–approved products for topical anesthesia. In addition, several compounded pharmacy products are available.
After 60 minutes of application of the eutectic mixture of 2.5% lidocaine and 2.5% prilocaine, a 3-mm depth of analgesia is reached, and after 120 minutes, a 4.5-mm depth is reached.9 It elicits a biphasic vascular response of vasoconstriction and blanching followed by vasodilation and erythema.10 Most adverse events are mild and transient, but allergic contact dermatitis and contact urticaria have been reported.11-13 In older children and adults, the maximum application area is 200 cm2, with a maximum dose of 20 g used for no longer than 4 hours.
The 4% or 5% lidocaine cream uses a liposomal delivery system, which is designed to improve cutaneous penetration and has been shown to provide longer durations of anesthesia than nonliposomal lidocaine preparations.14 Application should be performed 30 to 60 minutes prior to a procedure. In a study comparing the eutectic mixture of 2.5% lidocaine and 2.5% prilocaine versus lidocaine cream 5% for pain control during laser hair removal with a 1064-nm Nd:YAG laser, no significant differences were found.15 The maximum application area is 100 cm2 in children weighing less than 20 kg. A study of healthy adults demonstrated safety with the use of 30 to 60 g of occluded liposomal lidocaine cream 4%.16
In addition to US Food and Drug Administration–approved products, several compounded pharmacy products are available for topical anesthesia. These formulations include benzocaine-lidocaine-tetracaine gel, tetracaine-adrenaline-cocaine solution, and lidocaine-epinephrine-tetracaine solution. A triple-anesthetic gel, benzocaine-lidocaine-tetracaine is widely used in cosmetic practice. The product has been shown to provide adequate anesthesia for laser resurfacing after 20 minutes without occlusion.17 Of note, compounded anesthetics lack standardization, and different pharmacies may follow their own individual protocols.
Regional Anesthesia
Regional nerve blockade is a useful option for more widespread or complex interventions. Using regional nerve blockade, effective analgesia can be delivered to a target area while avoiding the toxicity and pain associated with numerous anesthetic infiltrations. In addition, there is no distortion of the tissue architecture, allowing for improved visual evaluation during the procedure. Recently, hyaluronic acid fillers have been compounded with lidocaine as a means of reducing procedural pain.
Blocks for Dermal Fillers
Forehead
For dermal filler injections of the glabellar and frontalis lines, anesthesia of the forehead may be desired. The supraorbital and supratrochlear nerves supply this area. The supraorbital nerve can be injected at the supraorbital notch, which is measured roughly 2.7 cm from the glabella. The orbital rim should be palpated with the nondominant hand, and 1 to 2 mL of anesthetic should be injected just below the rim (Figure 1). The supratrochlear nerve is located roughly 1.7 cm from the midline and can be similarly injected under the orbital rim with 1 to 2 mL of anesthetic (Figure 1).
Lateral Temple Region
Anesthesia of the zygomaticotemporal nerve can be used to reduce pain from dermal filler injections of the lateral canthal and temporal areas. The nerve is identified by first palpating the zygomaticofrontal suture. A long needle is then inserted posteriorly, immediately behind the concave surface of the lateral orbital rim, and 1 to 2 mL of anesthetic is injected (Figure 1).
Malar Region
Blockade of the zygomaticofacial nerve is commonly performed in conjunction with the zygomaticotemporal nerve and provides anesthesia to the malar region for cheek augmentation procedures. To identify the target area, the junction of the lateral and inferior orbital rim should be palpated. With the needle placed just lateral to this point, 1 to 2 mL of anesthetic is injected (Figure 1).

Blocks for Perioral Fillers
Upper Lips/Nasolabial Folds
Bilateral blockade of the infraorbital nerves provides anesthesia to the upper lip and nasolabial folds prior to filler injections. The infraorbital nerve can be targeted via an intraoral route where it exits the maxilla at the infraorbital foramen. The nerve is anesthetized by palpating the infraorbital ridge and injecting 3 to 5 mL of anesthetic roughly 1 cm below this point on the vertical axis of the midpupillary line (Figure 1). The external nasal nerve, thought to be a branch of cranial nerve V, also may be targeted if there is inadequate anesthesia from the infraorbital block. This nerve is reached by injecting at the osseocartilaginous junction of the nasal bones (Figure 1).
Lower Lips
Blockade of the mental nerve provides anesthesia to the lower lips for augmentation procedures. The mental nerve can be targeted on each side at the mental foramen, which is located below the root of the lower second premolar. Aiming roughly 1 cm below the gumline, 3 to 5 mL of anesthetic is injected intraorally (Figure 1). A transcutaneous approach toward the same target also is possible, though this technique risks visible bruising. Alternatively, the upper or lower lips can be anesthetized using 4 to 5 submucosal injections at evenly spaced intervals between the canine teeth.18
Blocks for Palmoplantar Hyperhidrosis
The treatment of palmoplantar hyperhidrosis benefits from regional blocks. Botulinum toxin has been well established as an effective therapy for the condition.19-21 Given the sensitivity of palmoplantar sites, it is valuable to achieve effective analgesia of the region prior to dermal injections of botulinum toxin.
Wrists
Sensory innervation of the palm is provided by the median, ulnar, and radial nerves (Figure 2A).
The ulnar nerve is anesthetized between the ulnar artery and the flexor carpi ulnaris muscle. The artery is identified by palpation, and special care should be taken to avoid intra-arterial injection. The needle is directed toward the radial styloid, and 3 to 5 mL of anesthetic is injected roughly 1 cm proximal to the wrist crease (Figure 2B).
Anesthesia of the radial nerve can be considered a field block given the numerous small branches that supply the hand. These branches are reached by injecting anesthetic roughly 2 to 3 cm proximal to the radial styloid with the needle aimed medially and extending the injection dorsally (Figure 2B). A total of 4 to 6 mL of anesthetic is used.

Ankles
An ankle block provides anesthesia to the dorsal and plantar surfaces of the foot.22 The region is supplied by the superficial peroneal nerve, deep peroneal nerve, sural nerve, saphenous nerve, and branches of the posterior tibial nerve (Figure 3A).
To anesthetize the deep peroneal nerve, the extensor hallucis longus tendon is first identified on the anterior surface of the ankle through dorsiflexion of the toes; the dorsalis pedis artery runs in close proximity. The injection should be placed lateral to the tendon and artery (Figure 3B). The needle should be inserted until bone is reached, withdrawn slightly, and then 3 to 5 mL of anesthetic should be injected. To block the saphenous nerve, the needle can then be directed superficially toward the medial malleolus, and 3 to 5 mL should be injected in a subcutaneous wheal (Figure 3C). To block the superficial peroneal nerve, the needle should then be directed toward the lateral malleolus, and 3 to 5 mL should be injected in a subcutaneous wheal (Figure 3C).
The posterior tibial nerve is located posterior to the medial malleolus. The dorsalis pedis artery can be palpated near this location. The needle should be inserted posterior to the artery, extending until bone is reached (Figure 3C). The needle is then withdrawn slightly, and 3 to 5 mL of anesthetic is injected. Finally, the sural nerve is anesthetized between the Achilles tendon and the lateral malleolus, using 5 mL of anesthetic to raise a subcutaneous wheal (Figure 3C).

Conclusion
Proper pain management is integral to ensuring a positive experience for cosmetic patients. Enhanced knowledge of local anesthetic techniques allows the clinician to provide for a variety of procedural indications and patient preferences. As anesthetic strategies are continually evolving, it is important for practitioners to remain informed of these developments.
Local anesthesia is a central component of successful interventions in cosmetic dermatology. The number of anesthetic medications and administration techniques has grown in recent years as outpatient cosmetic procedures continue to expand. Pain is a common barrier to cosmetic procedures, and alleviating the fear of painful interventions is critical to patient satisfaction and future visits. To accommodate a multitude of cosmetic interventions, it is important for clinicians to be well versed in applications of topical and regional anesthesia. In this article, we review pain management strategies for use in cosmetic practice.
Local Anesthetics
The sensation of pain is carried to the central nervous system by unmyelinated C nerve fibers. Local anesthetics (LAs) act by blocking fast voltage-gated sodium channels in the cell membrane of the nerve, thereby inhibiting downstream propagation of an action potential and the transmission of painful stimuli.1 The chemical structure of LAs is fundamental to their mechanism of action and metabolism. Local anesthetics contain a lipophilic aromatic group, an intermediate chain, and a hydrophilic amine group. Broadly, agents are classified as amides or esters depending on the chemical group attached to the intermediate chain.2 Amides (eg, lidocaine, bupivacaine, articaine, mepivacaine, prilocaine, levobupivacaine) are metabolized by the hepatic system; esters (eg, procaine, proparacaine, benzocaine, chlorprocaine, tetracaine, cocaine) are metabolized by plasma cholinesterase, which produces para-aminobenzoic acid, a potentially dangerous metabolite that has been implicated in allergic reactions.3
Lidocaine is the most prevalent LA used in dermatology practices. Importantly, lidocaine is a class IB antiarrhythmic agent used in cardiology to treat ventricular arrhythmias.4 As an anesthetic, a maximum dose of 4.5 mg/kg can be administered, increasing to 7.0 mg/kg when mixed with epinephrine; with higher doses, there is a risk for central nervous system and cardiovascular toxicity.5 Initial symptoms of lidocaine toxicity include dizziness, tinnitus, circumoral paresthesia, blurred vision, and a metallic taste in the mouth.6 Systemic absorption of topical anesthetics is heightened across mucosal membranes, and care should be taken when applying over large surface areas.
Allergic reactions to LAs may be local or less frequently systemic. It is important to note that LAs tend to show cross-reactivity within their class rather than across different classes.7 Reactions can be classified as type I or type IV. Type I (IgE-mediated) reactions evolve in minutes to hours, affecting the skin and possibly leading to respiratory and circulatory collapse. Delayed reactions to LAs have increased in recent years, with type IV contact allergy most frequently found in connection with benzocaine and lidocaine.8
Topical Anesthesia
Topical anesthetics are effective and easy to use and are particularly valuable in patients with needle phobia. In certain cases, these medications may be applied by the patient prior to arrival, thereby reducing visit time. Topical agents act on nerve fibers running through the dermis; therefore, efficacy is dependent on successful penetration through the stratum corneum and viable epidermis. To enhance absorption, agents may be applied under an occlusive dressing.
Topical anesthetics are most commonly used for injectable fillers, ablative and nonablative laser resurfacing, laser hair removal, and tattoo removal. The eutectic mixture of 2.5% lidocaine and 2.5% prilocaine as well as topical 4% or 5% lidocaine are the most commonly used US Food and Drug Administration–approved products for topical anesthesia. In addition, several compounded pharmacy products are available.
After 60 minutes of application of the eutectic mixture of 2.5% lidocaine and 2.5% prilocaine, a 3-mm depth of analgesia is reached, and after 120 minutes, a 4.5-mm depth is reached.9 It elicits a biphasic vascular response of vasoconstriction and blanching followed by vasodilation and erythema.10 Most adverse events are mild and transient, but allergic contact dermatitis and contact urticaria have been reported.11-13 In older children and adults, the maximum application area is 200 cm2, with a maximum dose of 20 g used for no longer than 4 hours.
The 4% or 5% lidocaine cream uses a liposomal delivery system, which is designed to improve cutaneous penetration and has been shown to provide longer durations of anesthesia than nonliposomal lidocaine preparations.14 Application should be performed 30 to 60 minutes prior to a procedure. In a study comparing the eutectic mixture of 2.5% lidocaine and 2.5% prilocaine versus lidocaine cream 5% for pain control during laser hair removal with a 1064-nm Nd:YAG laser, no significant differences were found.15 The maximum application area is 100 cm2 in children weighing less than 20 kg. A study of healthy adults demonstrated safety with the use of 30 to 60 g of occluded liposomal lidocaine cream 4%.16
In addition to US Food and Drug Administration–approved products, several compounded pharmacy products are available for topical anesthesia. These formulations include benzocaine-lidocaine-tetracaine gel, tetracaine-adrenaline-cocaine solution, and lidocaine-epinephrine-tetracaine solution. A triple-anesthetic gel, benzocaine-lidocaine-tetracaine is widely used in cosmetic practice. The product has been shown to provide adequate anesthesia for laser resurfacing after 20 minutes without occlusion.17 Of note, compounded anesthetics lack standardization, and different pharmacies may follow their own individual protocols.
Regional Anesthesia
Regional nerve blockade is a useful option for more widespread or complex interventions. Using regional nerve blockade, effective analgesia can be delivered to a target area while avoiding the toxicity and pain associated with numerous anesthetic infiltrations. In addition, there is no distortion of the tissue architecture, allowing for improved visual evaluation during the procedure. Recently, hyaluronic acid fillers have been compounded with lidocaine as a means of reducing procedural pain.
Blocks for Dermal Fillers
Forehead
For dermal filler injections of the glabellar and frontalis lines, anesthesia of the forehead may be desired. The supraorbital and supratrochlear nerves supply this area. The supraorbital nerve can be injected at the supraorbital notch, which is measured roughly 2.7 cm from the glabella. The orbital rim should be palpated with the nondominant hand, and 1 to 2 mL of anesthetic should be injected just below the rim (Figure 1). The supratrochlear nerve is located roughly 1.7 cm from the midline and can be similarly injected under the orbital rim with 1 to 2 mL of anesthetic (Figure 1).
Lateral Temple Region
Anesthesia of the zygomaticotemporal nerve can be used to reduce pain from dermal filler injections of the lateral canthal and temporal areas. The nerve is identified by first palpating the zygomaticofrontal suture. A long needle is then inserted posteriorly, immediately behind the concave surface of the lateral orbital rim, and 1 to 2 mL of anesthetic is injected (Figure 1).
Malar Region
Blockade of the zygomaticofacial nerve is commonly performed in conjunction with the zygomaticotemporal nerve and provides anesthesia to the malar region for cheek augmentation procedures. To identify the target area, the junction of the lateral and inferior orbital rim should be palpated. With the needle placed just lateral to this point, 1 to 2 mL of anesthetic is injected (Figure 1).

Blocks for Perioral Fillers
Upper Lips/Nasolabial Folds
Bilateral blockade of the infraorbital nerves provides anesthesia to the upper lip and nasolabial folds prior to filler injections. The infraorbital nerve can be targeted via an intraoral route where it exits the maxilla at the infraorbital foramen. The nerve is anesthetized by palpating the infraorbital ridge and injecting 3 to 5 mL of anesthetic roughly 1 cm below this point on the vertical axis of the midpupillary line (Figure 1). The external nasal nerve, thought to be a branch of cranial nerve V, also may be targeted if there is inadequate anesthesia from the infraorbital block. This nerve is reached by injecting at the osseocartilaginous junction of the nasal bones (Figure 1).
Lower Lips
Blockade of the mental nerve provides anesthesia to the lower lips for augmentation procedures. The mental nerve can be targeted on each side at the mental foramen, which is located below the root of the lower second premolar. Aiming roughly 1 cm below the gumline, 3 to 5 mL of anesthetic is injected intraorally (Figure 1). A transcutaneous approach toward the same target also is possible, though this technique risks visible bruising. Alternatively, the upper or lower lips can be anesthetized using 4 to 5 submucosal injections at evenly spaced intervals between the canine teeth.18
Blocks for Palmoplantar Hyperhidrosis
The treatment of palmoplantar hyperhidrosis benefits from regional blocks. Botulinum toxin has been well established as an effective therapy for the condition.19-21 Given the sensitivity of palmoplantar sites, it is valuable to achieve effective analgesia of the region prior to dermal injections of botulinum toxin.
Wrists
Sensory innervation of the palm is provided by the median, ulnar, and radial nerves (Figure 2A).
The ulnar nerve is anesthetized between the ulnar artery and the flexor carpi ulnaris muscle. The artery is identified by palpation, and special care should be taken to avoid intra-arterial injection. The needle is directed toward the radial styloid, and 3 to 5 mL of anesthetic is injected roughly 1 cm proximal to the wrist crease (Figure 2B).
Anesthesia of the radial nerve can be considered a field block given the numerous small branches that supply the hand. These branches are reached by injecting anesthetic roughly 2 to 3 cm proximal to the radial styloid with the needle aimed medially and extending the injection dorsally (Figure 2B). A total of 4 to 6 mL of anesthetic is used.

Ankles
An ankle block provides anesthesia to the dorsal and plantar surfaces of the foot.22 The region is supplied by the superficial peroneal nerve, deep peroneal nerve, sural nerve, saphenous nerve, and branches of the posterior tibial nerve (Figure 3A).
To anesthetize the deep peroneal nerve, the extensor hallucis longus tendon is first identified on the anterior surface of the ankle through dorsiflexion of the toes; the dorsalis pedis artery runs in close proximity. The injection should be placed lateral to the tendon and artery (Figure 3B). The needle should be inserted until bone is reached, withdrawn slightly, and then 3 to 5 mL of anesthetic should be injected. To block the saphenous nerve, the needle can then be directed superficially toward the medial malleolus, and 3 to 5 mL should be injected in a subcutaneous wheal (Figure 3C). To block the superficial peroneal nerve, the needle should then be directed toward the lateral malleolus, and 3 to 5 mL should be injected in a subcutaneous wheal (Figure 3C).
The posterior tibial nerve is located posterior to the medial malleolus. The dorsalis pedis artery can be palpated near this location. The needle should be inserted posterior to the artery, extending until bone is reached (Figure 3C). The needle is then withdrawn slightly, and 3 to 5 mL of anesthetic is injected. Finally, the sural nerve is anesthetized between the Achilles tendon and the lateral malleolus, using 5 mL of anesthetic to raise a subcutaneous wheal (Figure 3C).

Conclusion
Proper pain management is integral to ensuring a positive experience for cosmetic patients. Enhanced knowledge of local anesthetic techniques allows the clinician to provide for a variety of procedural indications and patient preferences. As anesthetic strategies are continually evolving, it is important for practitioners to remain informed of these developments.
- Scholz A. Mechanisms of (local) anaesthetics on voltage-gated sodium and other ion channels. Br J Anaesth. 2002;89:52-61.
- Auletta MJ. Local anesthesia for dermatologic surgery. Semin Dermatol. 1994;13:35-42.
- Park KK, Sharon VR. A review of local anesthetics: minimizing risk and side effects in cutaneous surgery. Dermatol Surg. 2017;43:173-187.
- Reiz S, Nath S. Cardiotoxicity of local anaesthetic agents. Br J Anaesth. 1986;58:736-746.
- Klein JA, Kassarjdian N. Lidocaine toxicity with tumescent liposuction. a case report of probable drug interactions. Dermatol Surg. 1997;23:1169-1174.
- Minkis K, Whittington A, Alam M. Dermatologic surgery emergencies: complications caused by systemic reactions, high-energy systems, and trauma. J Am Acad Dermatol. 2016;75:265-284.
- Morais-Almeida M, Gaspar A, Marinho S, et al. Allergy to local anesthetics of the amide group with tolerance to procaine. Allergy. 2003;58:827-828.
- To D, Kossintseva I, de Gannes G. Lidocaine contact allergy is becoming more prevalent. Dermatol Surg. 2014;40:1367-1372.
- Wahlgren CF, Quiding H. Depth of cutaneous analgesia after application of a eutectic mixture of the local anesthetics lidocaine and prilocaine (EMLA cream). J Am Acad Dermatol. 2000;42:584-588.
- Bjerring P, Andersen PH, Arendt-Nielsen L. Vascular response of human skin after analgesia with EMLA cream. Br J Anaesth. 1989;63:655-660.
- Ismail F, Goldsmith PC. EMLA cream-induced allergic contact dermatitis in a child with thalassaemia major. Contact Dermatitis. 2005;52:111.
- Thakur BK, Murali MR. EMLA cream-induced allergic contact dermatitis: a role for prilocaine as an immunogen. J Allergy Clin Immunol. 1995;95:776-778.
- Waton J, Boulanger A, Trechot PH, et al. Contact urticaria from EMLA cream. Contact Dermatitis. 2004;51:284-287.
- Bucalo BD, Mirikitani EJ, Moy RL. Comparison of skin anesthetic effect of liposomal lidocaine, nonliposomal lidocaine, and EMLA using 30-minute application time. Dermatol Surg. 1998;24:537-541.
- Guardiano RA, Norwood CW. Direct comparison of EMLA versus lidocaine for pain control in Nd:YAG 1,064 nm laser hair removal. Dermatol Surg. 2005;31:396-398.
- Nestor MS. Safety of occluded 4% liposomal lidocaine cream. J Drugs Dermatol. 2006;5:618-620.
- Oni G, Rasko Y, Kenkel J. Topical lidocaine enhanced by laser pretreatment: a safe and effective method of analgesia for facial rejuvenation. Aesthet Surg J. 2013;33:854-861.
- Niamtu J 3rd. Simple technique for lip and nasolabial fold anesthesia for injectable fillers. Dermatol Surg. 2005;31:1330-1332.
- Naumann M, Flachenecker P, Brocker EB, et al. Botulinum toxin for palmar hyperhidrosis. Lancet. 1997;349:252.
- Naumann M, Hofmann U, Bergmann I, et al. Focal hyperhidrosis: effective treatment with intracutaneous botulinum toxin. Arch Dermatol. 1998;134:301-304.
- Shelley WB, Talanin NY, Shelley ED. Botulinum toxin therapy for palmar hyperhidrosis. J Am Acad Dermatol. 1998;38(2, pt 1):227-229.
- Davies T, Karanovic S, Shergill B. Essential regional nerve blocks for the dermatologist: part 2. Clin Exp Dermatol. 2014;39:861-867.
- Scholz A. Mechanisms of (local) anaesthetics on voltage-gated sodium and other ion channels. Br J Anaesth. 2002;89:52-61.
- Auletta MJ. Local anesthesia for dermatologic surgery. Semin Dermatol. 1994;13:35-42.
- Park KK, Sharon VR. A review of local anesthetics: minimizing risk and side effects in cutaneous surgery. Dermatol Surg. 2017;43:173-187.
- Reiz S, Nath S. Cardiotoxicity of local anaesthetic agents. Br J Anaesth. 1986;58:736-746.
- Klein JA, Kassarjdian N. Lidocaine toxicity with tumescent liposuction. a case report of probable drug interactions. Dermatol Surg. 1997;23:1169-1174.
- Minkis K, Whittington A, Alam M. Dermatologic surgery emergencies: complications caused by systemic reactions, high-energy systems, and trauma. J Am Acad Dermatol. 2016;75:265-284.
- Morais-Almeida M, Gaspar A, Marinho S, et al. Allergy to local anesthetics of the amide group with tolerance to procaine. Allergy. 2003;58:827-828.
- To D, Kossintseva I, de Gannes G. Lidocaine contact allergy is becoming more prevalent. Dermatol Surg. 2014;40:1367-1372.
- Wahlgren CF, Quiding H. Depth of cutaneous analgesia after application of a eutectic mixture of the local anesthetics lidocaine and prilocaine (EMLA cream). J Am Acad Dermatol. 2000;42:584-588.
- Bjerring P, Andersen PH, Arendt-Nielsen L. Vascular response of human skin after analgesia with EMLA cream. Br J Anaesth. 1989;63:655-660.
- Ismail F, Goldsmith PC. EMLA cream-induced allergic contact dermatitis in a child with thalassaemia major. Contact Dermatitis. 2005;52:111.
- Thakur BK, Murali MR. EMLA cream-induced allergic contact dermatitis: a role for prilocaine as an immunogen. J Allergy Clin Immunol. 1995;95:776-778.
- Waton J, Boulanger A, Trechot PH, et al. Contact urticaria from EMLA cream. Contact Dermatitis. 2004;51:284-287.
- Bucalo BD, Mirikitani EJ, Moy RL. Comparison of skin anesthetic effect of liposomal lidocaine, nonliposomal lidocaine, and EMLA using 30-minute application time. Dermatol Surg. 1998;24:537-541.
- Guardiano RA, Norwood CW. Direct comparison of EMLA versus lidocaine for pain control in Nd:YAG 1,064 nm laser hair removal. Dermatol Surg. 2005;31:396-398.
- Nestor MS. Safety of occluded 4% liposomal lidocaine cream. J Drugs Dermatol. 2006;5:618-620.
- Oni G, Rasko Y, Kenkel J. Topical lidocaine enhanced by laser pretreatment: a safe and effective method of analgesia for facial rejuvenation. Aesthet Surg J. 2013;33:854-861.
- Niamtu J 3rd. Simple technique for lip and nasolabial fold anesthesia for injectable fillers. Dermatol Surg. 2005;31:1330-1332.
- Naumann M, Flachenecker P, Brocker EB, et al. Botulinum toxin for palmar hyperhidrosis. Lancet. 1997;349:252.
- Naumann M, Hofmann U, Bergmann I, et al. Focal hyperhidrosis: effective treatment with intracutaneous botulinum toxin. Arch Dermatol. 1998;134:301-304.
- Shelley WB, Talanin NY, Shelley ED. Botulinum toxin therapy for palmar hyperhidrosis. J Am Acad Dermatol. 1998;38(2, pt 1):227-229.
- Davies T, Karanovic S, Shergill B. Essential regional nerve blocks for the dermatologist: part 2. Clin Exp Dermatol. 2014;39:861-867.
Practice Points
- The proper delivery of local anesthesia is integral to successful cosmetic interventions.
- Regional nerve blocks can provide effective analgesia while reducing the number of injections and preserving the architecture of the cosmetic field.
In Vivo Reflectance Confocal Microscopy
Reflectance confocal microscopy (RCM) imaging received Category I Current Procedural Terminology (CPT) codes by the Centers for Medicare & Medicaid Services in January 2016 and can now be submitted to insurance companies with reimbursement comparable to a skin biopsy or a global skin pathology service.1 This fairly new technology is a US Food and Drug Administration–cleared noninvasive imaging modality that provides high-resolution in vivo cellular images of the skin. It has been shown to be efficacious in differentiating benign and malignant skin lesions, increasing diagnostic accuracy, and reducing the number of unnecessary skin biopsies that are performed. In addition to skin cancer diagnosis, RCM imaging also can help guide management of malignant lesions by detecting lateral margins prior to surgery as well as monitoring the lesion over time for treatment efficacy or recurrence. The potential impact of RCM imaging is tremendous, and reimbursement may lead to increased use in clinical practice to the benefit of our patients. Herein, we present a brief review of RCM imaging and reimbursement as well as the benefits and limitations of this new technology for dermatologists.
Reflectance Confocal Microscopy
In vivo RCM allows us to visualize the epidermis in real time on a cellular level down to the papillary dermis at a high resolution (×30) comparable to histologic examination. With optical sections 3- to 5-µm thick and a lateral resolution of 0.5 to 1.0 µm, RCM produces a stack of 500×500-µm2 images up to a depth of approximately 200 µm.2,3 At any chosen depth, these smaller images are stitched together with sophisticated software into a block, or mosaic, increasing the field of view to up to 8×8 mm2. Imaging is performed in en face planes oriented parallel to the skin surface, similar to dermoscopy.
Current CPT Guidelines and Reimbursement
The CPT codes for RCM imaging provide reimbursement on a per-lesion basis and are similar to those used for skin biopsy and pathology (Table).1 Codes 96931 through 96933 are used for imaging of a single lesion on a patient. The first code—96931—is used when image acquisition, interpretation, and report creation are carried out by a single clinician. The next 2 codes are used when one clinician acquires the image—96932—comparable to the technical component of a pathology code, while another reads it and creates the report—96933—similar to a dermatopathologist billing for the professional component of a pathology report. For patients presenting with multiple lesions, the next 3 codes—96934, 96935, and 96936—are used in conjunction with the applicable first code for each additional lesion with similar global, technical, and professional components. Because these codes are not in the radiology or pathology sections of CPT, a single code cannot be used with modifier -TC (technical component) and modifier -26, as they are in those sections.
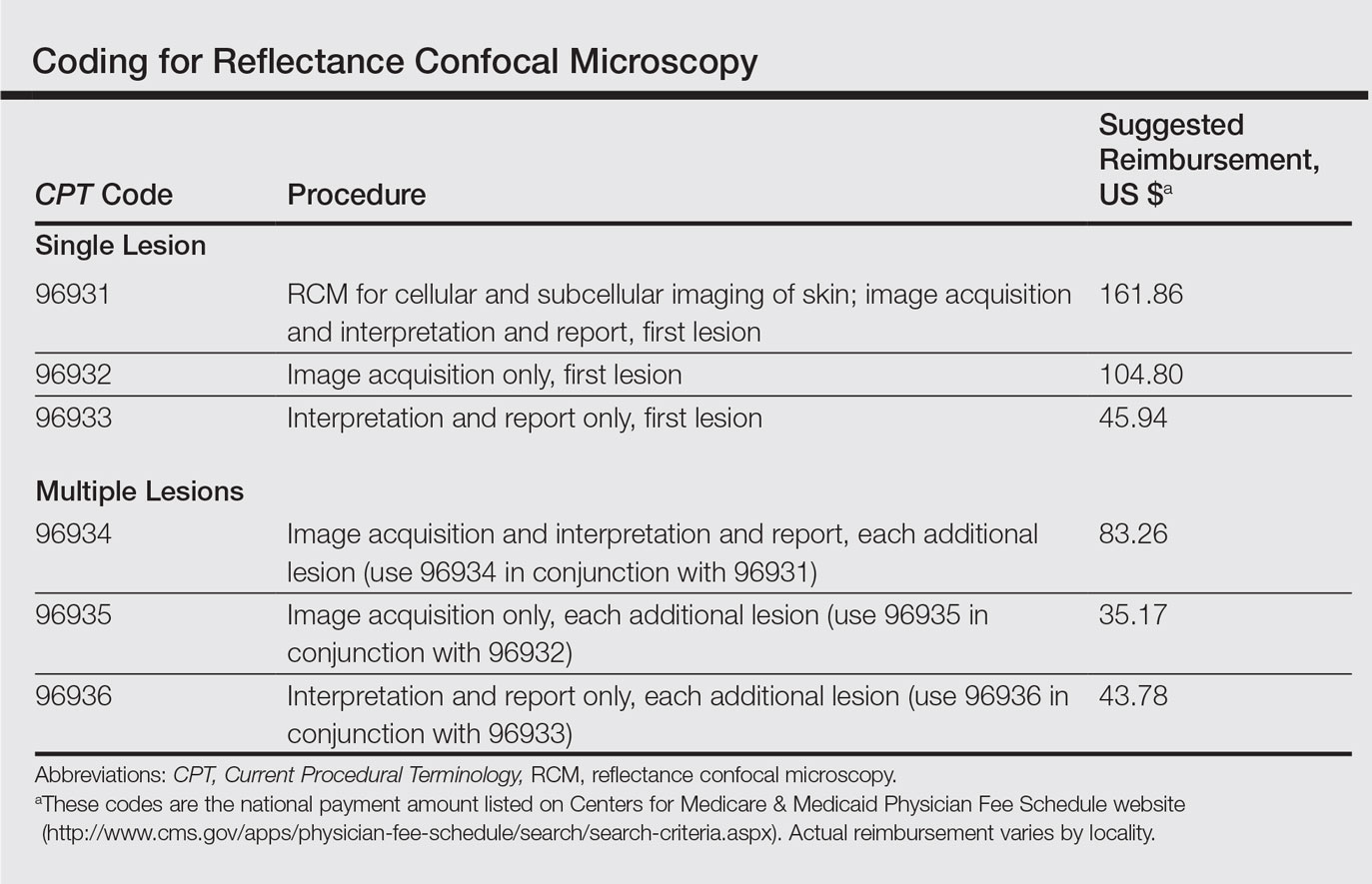
The wide-probe VivaScope 1500 (Caliber I.D., Inc) currently is the only confocal device that can be reported with a CPT code and routinely reimbursed. The handheld VivaScope 3000 (Caliber I.D., Inc) can only view a small stack and does not have the ability to acquire a full mosaic image; it is not covered by these codes.
Images can be viewed as a stack captured at the same horizontal position but at sequential depths or as a mosaic, which has a larger field of view but is limited to a single plane. To appropriately assess a lesion, clinicians must obtain a mosaic that needs to be assessed at multiple layers for a diagnosis to be made because it is a cross-section view.
Diagnosis
Studies have demonstrated the usefulness of RCM imaging in the diagnosis of a wide range of skin diseases, including melanoma and nonmelanoma skin cancers, infectious diseases, and inflammatory and autoimmune conditions, as well as wound healing and skin aging. Reflectance confocal microscopy imaging is not limited to the skin; it can be used to evaluate the hair, nails, oral mucosa, and other organs.
According to several studies, RCM imaging notably increases the diagnostic accuracy and detection rate of skin cancers over clinical and dermoscopic examination alone and therefore can act as an aid in differentiating lesions that are benign versus those that are suspicious and should be biopsied.
Reflectance confocal microscopy has been shown to have a mean sensitivity of 94% (range, 92%–96%) and specificity of 83% (range, 81%–84%) for all types of skin cancer when used with dermoscopy.4 In particular, for melanocytic lesions that are ambiguous on dermoscopy, RCM used in addition to dermoscopy increases the mean sensitivity and specificity for melanoma diagnosis to 93% (range, 89%–96%) and 76% (range, 68%–83%), respectively.5 Although these reported sensitivities are comparable to dermoscopy, the specificity is superior, especially for detecting hypomelanotic and amelanotic melanomas, which often lack specific features on dermoscopy.6-8
The combination of RCM with dermoscopy has reduced the number of unnecessary excisions of benign nevi by more than 50% when compared to dermoscopy alone.9 One study showed that the number needed to treat (ie, excise) a melanoma decreased from 14.6 with dermoscopy alone to 6.8 when guided by dermoscopy and RCM imaging.9 In a similar study, the number needed to treat dropped from 19.41 with dermoscopy alone to 6.25 with dermoscopy and RCM.10
These studies were not looking to evaluate RCM as a replacement test but rather as an add-on test to dermoscopy. Reflectance confocal microscopy imaging takes longer than dermoscopy for each lesion; therefore, RCM should only be used as an adjunctive tool to dermoscopy and not as an initial screening test. Consequentially, a dermatologist skilled in dermoscopy is essential in deciding which lesions would be appropriate for subsequent RCM imaging.
In Vivo Margin Mapping as an Adjunct to Surgery
Oftentimes, tumor margins are poorly defined and can be difficult to map clinically and dermoscopically. Studies have demonstrated the use of RCM in delineation of surgical margins prior to surgery or excisional biopsies.11,12 Alternatively, when complete removal at biopsy would be impractical (eg, for extremely large lesions or lesions located in cosmetically sensitive areas such as the face), RCM can be used to pick the best site for an appropriate biopsy, which decreases the chance of sampling error due to skip lesions and increases histologic accuracy.
Nonsurgical Treatment Monitoring
One advantage of RCM over conventional histology is that RCM imaging leaves the tissue intact, allowing dynamic changes to be studied over time, which is useful for monitoring nonmelanoma skin cancers and lentigo maligna being treated with noninvasive therapeutic modalities.13 If not as a definitive treatment, RCM can act as an adjunct for surgery by monitoring reduction in lesion size prior to Mohs micrographic surgery, thereby decreasing the resulting surgical defect.14
Limitations
Imaging Depth
Although RCM is a revolutionary device in the field of dermatology, it has several limitations. With a maximal imaging depth of 350 µm, the imaging resolution decreases substantially with depth, limiting accurate interpretation to 200 µm. Reflectance confocal microscopy can only image the superficial portion of a lesion; therefore, deep tumor margins cannot be assessed. Hypertrophic or hyperkeratotic lesions, including lesions on the palms and soles, also are unable to be imaged with RCM. This limitation in depth penetration makes treatment monitoring impossible for invasive lesions that extend into the dermal layer.
Difficult-to-Reach Areas
Another limitation is the difficulty imaging areas such as the ocular canthi, nasal alae, or helices of the ear due to the wide probe size on the VivaScope 1500. The advent of the smaller handheld VivaScope 3000 device allows for improved imaging of concave services and difficult lesions at the risk of less accurate imaging, low field of view, and no reimbursement at present.
False-Positive Results
Although RCM has been shown to be helpful in reducing unnecessary biopsies, there still is the issue of false-positives on imaging. False-positives most commonly occur in nevi with severe atypia or when Langerhans cells are present that cannot always be differentiated from melanocytic cells.3,15,16 One prospective study found 7 false-positive results from 63 sites using RCM for the diagnosis of lentigo malignas.16 False-negatives can occur in the presence of inflammatory infiltrates and scar tissue that can hide cellular morphology or in sampling errors due to skip lesions.3,16
Time Efficiency
The time required for acquisition of RCM mosaics and stacks followed by reading and interpretation can be substantial depending on the size and complexity of the lesion, which is a major limitation for use of RCM in busy dermatology practices; therefore, RCM should be reserved for lesions selected to undergo biopsy that are clinically equivocal for malignancy prior to RCM examination.17 It would not be cost-effective or time effective to evaluate lesions that either clinically or dermoscopically have a high probability of malignancy; however, patients and physicians may opt for increased specificity at the expense of time, particularly when a lesion is located on a cosmetically sensitive area, as patients can avoid initial histologic biopsy and gain the cosmetic benefit of going straight to surgery versus obtaining an initial diagnostic biopsy.
Cost
Lastly, the high cost involved in purchasing an RCM device and the training involved to use and interpret RCM images currently limits RCM to large academic centers. Reimbursement may make more widespread use feasible. In any event, RCM imaging should be part of the curriculum for both dermatology and pathology trainees.
Future Directions
In vivo RCM is a noninvasive imaging modality that allows for real-time evaluation of the skin. Used in conjunction with dermoscopy, RCM can substantially improve diagnostic accuracy and reduce the number of unnecessary biopsies. Now that RCM has finally gained foundational CPT codes and insurance reimbursement, there may be a growing demand for clinicians to incorporate this technology into their clinical practice.
- Current Procedural Terminology 2017, Professional Edition. Chicago IL: American Medical Association; 2016.
- Que SK, Fraga-Braghiroli N, Grant-Kels JM, et al. Through the looking glass: basics and principles of reflectance confocal microscopy [published online June 4, 2015]. J Am Acad Dermatol. 2015;73:276-284.
- Rajadhyaksha M, Marghoob A, Rossi A, et al. Reflectance confocal microscopy of skin in vivo: from bench to bedside [published online October 27, 2016]. Lasers Surg Med. 2017;49:7-19.
- Xiong YD, Ma S, Li X, et al. A meta-analysis of reflectance confocal microscopy for the diagnosis of malignant skin tumours. J Eur Acad Dermatol Venereol. 2016;30:1295-1302.
- Stevenson AD, Mickan S, Mallett S, et al. Systematic review of diagnostic accuracy of reflectance confocal microscopy for melanoma diagnosis in patients with clinically equivocal skin lesions. Dermatol Pract Concept. 2013;3:19-27.
- Busam KJ, Hester K, Charles C, et al. Detection of clinically amelanotic malignant melanoma and assessment of its margins by in vivo confocal scanning laser microscopy. Arch Dermatol. 2001;137:923-929.
- Losi A, Longo C, Cesinaro AM, et al. Hyporeflective pagetoid cells: a new clue for amelanotic melanoma diagnosis by reflectance confocal microscopy. Br J Dermatol. 2014;171:48-54.
- Guitera P, Menzies SQ, Argenziano G, et al. Dermoscopy and in vivo confocal microscopy are complementary techniques for the diagnosis of difficult amelanotic and light-coloured skin lesions [published online October 12, 2016]. Br J Dermatol. 2016;175:1311-1319.
- Pellacani G, Pepe P, Casari A, et al. Reflectance confocal microscopy as a second-level examination in skin oncology improves diagnostic accuracy and saves unnecessary excisions: a longitudinal prospective study. Br J Dermatol. 2014;171:1044-1051.
- Pellacani G, Witkowski A, Cesinaro AM, et al. Cost-benefit of reflectance confocal microscopy in the diagnostic performance of melanoma. J Eur Acad Dermatol Venereol. 2016;30:413-419.
- Champin J, Perrot JL, Cinotti E, et al. In vivo reflectance confocal microscopy to optimize the spaghetti technique for defining surgical margins of lentigo maligna. Dermatol Surg. 2014;40:247-256.
- Hibler BP, Cordova M, Wong RJ, et al. Intraoperative real-time reflectance confocal microscopy for guiding surgical margins of lentigo maligna melanoma. Dermatol Surg. 2015;41:980-983.
- Ulrich M, Lange-Asschenfeldt S, Gonzalez S. The use of reflectance confocal microscopy for monitoring response to therapy of skin malignancies. Dermatol Pract Concept. 2012;2:202a10.
- Torres A, Niemeyer A, Berkes B, et al. 5% imiquimod cream and reflectance-mode confocal microscopy as adjunct modalities to Mohs micrographic surgery for treatment of basal cell carcinoma. Dermatol Surg. 2004;30(12, pt 1):1462-1469.
- Hashemi P, Pulitzer MP, Scope A, et al. Langerhans cells and melanocytes share similar morphologic features under in vivo reflectance confocal microscopy: a challenge for melanoma diagnosis. J Am Acad Dermatol. 2012;66:452-462.
- Menge TD, Hibler BP, Cordova MA, et al. Concordance of handheld reflectance confocal microscopy (RCM) with histopathology in the diagnosis of lentigo maligna (LM): a prospective study. J Am Acad Dermatol. 2016;74:1114-1120.
- Borsari S, Pampena R, Lallas A, et al. Clinical indications for use of reflectance confocal microscopy for skin cancer diagnosis. JAMA Dermatol. 2016;152:1093-1098.
Reflectance confocal microscopy (RCM) imaging received Category I Current Procedural Terminology (CPT) codes by the Centers for Medicare & Medicaid Services in January 2016 and can now be submitted to insurance companies with reimbursement comparable to a skin biopsy or a global skin pathology service.1 This fairly new technology is a US Food and Drug Administration–cleared noninvasive imaging modality that provides high-resolution in vivo cellular images of the skin. It has been shown to be efficacious in differentiating benign and malignant skin lesions, increasing diagnostic accuracy, and reducing the number of unnecessary skin biopsies that are performed. In addition to skin cancer diagnosis, RCM imaging also can help guide management of malignant lesions by detecting lateral margins prior to surgery as well as monitoring the lesion over time for treatment efficacy or recurrence. The potential impact of RCM imaging is tremendous, and reimbursement may lead to increased use in clinical practice to the benefit of our patients. Herein, we present a brief review of RCM imaging and reimbursement as well as the benefits and limitations of this new technology for dermatologists.
Reflectance Confocal Microscopy
In vivo RCM allows us to visualize the epidermis in real time on a cellular level down to the papillary dermis at a high resolution (×30) comparable to histologic examination. With optical sections 3- to 5-µm thick and a lateral resolution of 0.5 to 1.0 µm, RCM produces a stack of 500×500-µm2 images up to a depth of approximately 200 µm.2,3 At any chosen depth, these smaller images are stitched together with sophisticated software into a block, or mosaic, increasing the field of view to up to 8×8 mm2. Imaging is performed in en face planes oriented parallel to the skin surface, similar to dermoscopy.
Current CPT Guidelines and Reimbursement
The CPT codes for RCM imaging provide reimbursement on a per-lesion basis and are similar to those used for skin biopsy and pathology (Table).1 Codes 96931 through 96933 are used for imaging of a single lesion on a patient. The first code—96931—is used when image acquisition, interpretation, and report creation are carried out by a single clinician. The next 2 codes are used when one clinician acquires the image—96932—comparable to the technical component of a pathology code, while another reads it and creates the report—96933—similar to a dermatopathologist billing for the professional component of a pathology report. For patients presenting with multiple lesions, the next 3 codes—96934, 96935, and 96936—are used in conjunction with the applicable first code for each additional lesion with similar global, technical, and professional components. Because these codes are not in the radiology or pathology sections of CPT, a single code cannot be used with modifier -TC (technical component) and modifier -26, as they are in those sections.

The wide-probe VivaScope 1500 (Caliber I.D., Inc) currently is the only confocal device that can be reported with a CPT code and routinely reimbursed. The handheld VivaScope 3000 (Caliber I.D., Inc) can only view a small stack and does not have the ability to acquire a full mosaic image; it is not covered by these codes.
Images can be viewed as a stack captured at the same horizontal position but at sequential depths or as a mosaic, which has a larger field of view but is limited to a single plane. To appropriately assess a lesion, clinicians must obtain a mosaic that needs to be assessed at multiple layers for a diagnosis to be made because it is a cross-section view.
Diagnosis
Studies have demonstrated the usefulness of RCM imaging in the diagnosis of a wide range of skin diseases, including melanoma and nonmelanoma skin cancers, infectious diseases, and inflammatory and autoimmune conditions, as well as wound healing and skin aging. Reflectance confocal microscopy imaging is not limited to the skin; it can be used to evaluate the hair, nails, oral mucosa, and other organs.
According to several studies, RCM imaging notably increases the diagnostic accuracy and detection rate of skin cancers over clinical and dermoscopic examination alone and therefore can act as an aid in differentiating lesions that are benign versus those that are suspicious and should be biopsied.
Reflectance confocal microscopy has been shown to have a mean sensitivity of 94% (range, 92%–96%) and specificity of 83% (range, 81%–84%) for all types of skin cancer when used with dermoscopy.4 In particular, for melanocytic lesions that are ambiguous on dermoscopy, RCM used in addition to dermoscopy increases the mean sensitivity and specificity for melanoma diagnosis to 93% (range, 89%–96%) and 76% (range, 68%–83%), respectively.5 Although these reported sensitivities are comparable to dermoscopy, the specificity is superior, especially for detecting hypomelanotic and amelanotic melanomas, which often lack specific features on dermoscopy.6-8
The combination of RCM with dermoscopy has reduced the number of unnecessary excisions of benign nevi by more than 50% when compared to dermoscopy alone.9 One study showed that the number needed to treat (ie, excise) a melanoma decreased from 14.6 with dermoscopy alone to 6.8 when guided by dermoscopy and RCM imaging.9 In a similar study, the number needed to treat dropped from 19.41 with dermoscopy alone to 6.25 with dermoscopy and RCM.10
These studies were not looking to evaluate RCM as a replacement test but rather as an add-on test to dermoscopy. Reflectance confocal microscopy imaging takes longer than dermoscopy for each lesion; therefore, RCM should only be used as an adjunctive tool to dermoscopy and not as an initial screening test. Consequentially, a dermatologist skilled in dermoscopy is essential in deciding which lesions would be appropriate for subsequent RCM imaging.
In Vivo Margin Mapping as an Adjunct to Surgery
Oftentimes, tumor margins are poorly defined and can be difficult to map clinically and dermoscopically. Studies have demonstrated the use of RCM in delineation of surgical margins prior to surgery or excisional biopsies.11,12 Alternatively, when complete removal at biopsy would be impractical (eg, for extremely large lesions or lesions located in cosmetically sensitive areas such as the face), RCM can be used to pick the best site for an appropriate biopsy, which decreases the chance of sampling error due to skip lesions and increases histologic accuracy.
Nonsurgical Treatment Monitoring
One advantage of RCM over conventional histology is that RCM imaging leaves the tissue intact, allowing dynamic changes to be studied over time, which is useful for monitoring nonmelanoma skin cancers and lentigo maligna being treated with noninvasive therapeutic modalities.13 If not as a definitive treatment, RCM can act as an adjunct for surgery by monitoring reduction in lesion size prior to Mohs micrographic surgery, thereby decreasing the resulting surgical defect.14
Limitations
Imaging Depth
Although RCM is a revolutionary device in the field of dermatology, it has several limitations. With a maximal imaging depth of 350 µm, the imaging resolution decreases substantially with depth, limiting accurate interpretation to 200 µm. Reflectance confocal microscopy can only image the superficial portion of a lesion; therefore, deep tumor margins cannot be assessed. Hypertrophic or hyperkeratotic lesions, including lesions on the palms and soles, also are unable to be imaged with RCM. This limitation in depth penetration makes treatment monitoring impossible for invasive lesions that extend into the dermal layer.
Difficult-to-Reach Areas
Another limitation is the difficulty imaging areas such as the ocular canthi, nasal alae, or helices of the ear due to the wide probe size on the VivaScope 1500. The advent of the smaller handheld VivaScope 3000 device allows for improved imaging of concave services and difficult lesions at the risk of less accurate imaging, low field of view, and no reimbursement at present.
False-Positive Results
Although RCM has been shown to be helpful in reducing unnecessary biopsies, there still is the issue of false-positives on imaging. False-positives most commonly occur in nevi with severe atypia or when Langerhans cells are present that cannot always be differentiated from melanocytic cells.3,15,16 One prospective study found 7 false-positive results from 63 sites using RCM for the diagnosis of lentigo malignas.16 False-negatives can occur in the presence of inflammatory infiltrates and scar tissue that can hide cellular morphology or in sampling errors due to skip lesions.3,16
Time Efficiency
The time required for acquisition of RCM mosaics and stacks followed by reading and interpretation can be substantial depending on the size and complexity of the lesion, which is a major limitation for use of RCM in busy dermatology practices; therefore, RCM should be reserved for lesions selected to undergo biopsy that are clinically equivocal for malignancy prior to RCM examination.17 It would not be cost-effective or time effective to evaluate lesions that either clinically or dermoscopically have a high probability of malignancy; however, patients and physicians may opt for increased specificity at the expense of time, particularly when a lesion is located on a cosmetically sensitive area, as patients can avoid initial histologic biopsy and gain the cosmetic benefit of going straight to surgery versus obtaining an initial diagnostic biopsy.
Cost
Lastly, the high cost involved in purchasing an RCM device and the training involved to use and interpret RCM images currently limits RCM to large academic centers. Reimbursement may make more widespread use feasible. In any event, RCM imaging should be part of the curriculum for both dermatology and pathology trainees.
Future Directions
In vivo RCM is a noninvasive imaging modality that allows for real-time evaluation of the skin. Used in conjunction with dermoscopy, RCM can substantially improve diagnostic accuracy and reduce the number of unnecessary biopsies. Now that RCM has finally gained foundational CPT codes and insurance reimbursement, there may be a growing demand for clinicians to incorporate this technology into their clinical practice.
Reflectance confocal microscopy (RCM) imaging received Category I Current Procedural Terminology (CPT) codes by the Centers for Medicare & Medicaid Services in January 2016 and can now be submitted to insurance companies with reimbursement comparable to a skin biopsy or a global skin pathology service.1 This fairly new technology is a US Food and Drug Administration–cleared noninvasive imaging modality that provides high-resolution in vivo cellular images of the skin. It has been shown to be efficacious in differentiating benign and malignant skin lesions, increasing diagnostic accuracy, and reducing the number of unnecessary skin biopsies that are performed. In addition to skin cancer diagnosis, RCM imaging also can help guide management of malignant lesions by detecting lateral margins prior to surgery as well as monitoring the lesion over time for treatment efficacy or recurrence. The potential impact of RCM imaging is tremendous, and reimbursement may lead to increased use in clinical practice to the benefit of our patients. Herein, we present a brief review of RCM imaging and reimbursement as well as the benefits and limitations of this new technology for dermatologists.
Reflectance Confocal Microscopy
In vivo RCM allows us to visualize the epidermis in real time on a cellular level down to the papillary dermis at a high resolution (×30) comparable to histologic examination. With optical sections 3- to 5-µm thick and a lateral resolution of 0.5 to 1.0 µm, RCM produces a stack of 500×500-µm2 images up to a depth of approximately 200 µm.2,3 At any chosen depth, these smaller images are stitched together with sophisticated software into a block, or mosaic, increasing the field of view to up to 8×8 mm2. Imaging is performed in en face planes oriented parallel to the skin surface, similar to dermoscopy.
Current CPT Guidelines and Reimbursement
The CPT codes for RCM imaging provide reimbursement on a per-lesion basis and are similar to those used for skin biopsy and pathology (Table).1 Codes 96931 through 96933 are used for imaging of a single lesion on a patient. The first code—96931—is used when image acquisition, interpretation, and report creation are carried out by a single clinician. The next 2 codes are used when one clinician acquires the image—96932—comparable to the technical component of a pathology code, while another reads it and creates the report—96933—similar to a dermatopathologist billing for the professional component of a pathology report. For patients presenting with multiple lesions, the next 3 codes—96934, 96935, and 96936—are used in conjunction with the applicable first code for each additional lesion with similar global, technical, and professional components. Because these codes are not in the radiology or pathology sections of CPT, a single code cannot be used with modifier -TC (technical component) and modifier -26, as they are in those sections.

The wide-probe VivaScope 1500 (Caliber I.D., Inc) currently is the only confocal device that can be reported with a CPT code and routinely reimbursed. The handheld VivaScope 3000 (Caliber I.D., Inc) can only view a small stack and does not have the ability to acquire a full mosaic image; it is not covered by these codes.
Images can be viewed as a stack captured at the same horizontal position but at sequential depths or as a mosaic, which has a larger field of view but is limited to a single plane. To appropriately assess a lesion, clinicians must obtain a mosaic that needs to be assessed at multiple layers for a diagnosis to be made because it is a cross-section view.
Diagnosis
Studies have demonstrated the usefulness of RCM imaging in the diagnosis of a wide range of skin diseases, including melanoma and nonmelanoma skin cancers, infectious diseases, and inflammatory and autoimmune conditions, as well as wound healing and skin aging. Reflectance confocal microscopy imaging is not limited to the skin; it can be used to evaluate the hair, nails, oral mucosa, and other organs.
According to several studies, RCM imaging notably increases the diagnostic accuracy and detection rate of skin cancers over clinical and dermoscopic examination alone and therefore can act as an aid in differentiating lesions that are benign versus those that are suspicious and should be biopsied.
Reflectance confocal microscopy has been shown to have a mean sensitivity of 94% (range, 92%–96%) and specificity of 83% (range, 81%–84%) for all types of skin cancer when used with dermoscopy.4 In particular, for melanocytic lesions that are ambiguous on dermoscopy, RCM used in addition to dermoscopy increases the mean sensitivity and specificity for melanoma diagnosis to 93% (range, 89%–96%) and 76% (range, 68%–83%), respectively.5 Although these reported sensitivities are comparable to dermoscopy, the specificity is superior, especially for detecting hypomelanotic and amelanotic melanomas, which often lack specific features on dermoscopy.6-8
The combination of RCM with dermoscopy has reduced the number of unnecessary excisions of benign nevi by more than 50% when compared to dermoscopy alone.9 One study showed that the number needed to treat (ie, excise) a melanoma decreased from 14.6 with dermoscopy alone to 6.8 when guided by dermoscopy and RCM imaging.9 In a similar study, the number needed to treat dropped from 19.41 with dermoscopy alone to 6.25 with dermoscopy and RCM.10
These studies were not looking to evaluate RCM as a replacement test but rather as an add-on test to dermoscopy. Reflectance confocal microscopy imaging takes longer than dermoscopy for each lesion; therefore, RCM should only be used as an adjunctive tool to dermoscopy and not as an initial screening test. Consequentially, a dermatologist skilled in dermoscopy is essential in deciding which lesions would be appropriate for subsequent RCM imaging.
In Vivo Margin Mapping as an Adjunct to Surgery
Oftentimes, tumor margins are poorly defined and can be difficult to map clinically and dermoscopically. Studies have demonstrated the use of RCM in delineation of surgical margins prior to surgery or excisional biopsies.11,12 Alternatively, when complete removal at biopsy would be impractical (eg, for extremely large lesions or lesions located in cosmetically sensitive areas such as the face), RCM can be used to pick the best site for an appropriate biopsy, which decreases the chance of sampling error due to skip lesions and increases histologic accuracy.
Nonsurgical Treatment Monitoring
One advantage of RCM over conventional histology is that RCM imaging leaves the tissue intact, allowing dynamic changes to be studied over time, which is useful for monitoring nonmelanoma skin cancers and lentigo maligna being treated with noninvasive therapeutic modalities.13 If not as a definitive treatment, RCM can act as an adjunct for surgery by monitoring reduction in lesion size prior to Mohs micrographic surgery, thereby decreasing the resulting surgical defect.14
Limitations
Imaging Depth
Although RCM is a revolutionary device in the field of dermatology, it has several limitations. With a maximal imaging depth of 350 µm, the imaging resolution decreases substantially with depth, limiting accurate interpretation to 200 µm. Reflectance confocal microscopy can only image the superficial portion of a lesion; therefore, deep tumor margins cannot be assessed. Hypertrophic or hyperkeratotic lesions, including lesions on the palms and soles, also are unable to be imaged with RCM. This limitation in depth penetration makes treatment monitoring impossible for invasive lesions that extend into the dermal layer.
Difficult-to-Reach Areas
Another limitation is the difficulty imaging areas such as the ocular canthi, nasal alae, or helices of the ear due to the wide probe size on the VivaScope 1500. The advent of the smaller handheld VivaScope 3000 device allows for improved imaging of concave services and difficult lesions at the risk of less accurate imaging, low field of view, and no reimbursement at present.
False-Positive Results
Although RCM has been shown to be helpful in reducing unnecessary biopsies, there still is the issue of false-positives on imaging. False-positives most commonly occur in nevi with severe atypia or when Langerhans cells are present that cannot always be differentiated from melanocytic cells.3,15,16 One prospective study found 7 false-positive results from 63 sites using RCM for the diagnosis of lentigo malignas.16 False-negatives can occur in the presence of inflammatory infiltrates and scar tissue that can hide cellular morphology or in sampling errors due to skip lesions.3,16
Time Efficiency
The time required for acquisition of RCM mosaics and stacks followed by reading and interpretation can be substantial depending on the size and complexity of the lesion, which is a major limitation for use of RCM in busy dermatology practices; therefore, RCM should be reserved for lesions selected to undergo biopsy that are clinically equivocal for malignancy prior to RCM examination.17 It would not be cost-effective or time effective to evaluate lesions that either clinically or dermoscopically have a high probability of malignancy; however, patients and physicians may opt for increased specificity at the expense of time, particularly when a lesion is located on a cosmetically sensitive area, as patients can avoid initial histologic biopsy and gain the cosmetic benefit of going straight to surgery versus obtaining an initial diagnostic biopsy.
Cost
Lastly, the high cost involved in purchasing an RCM device and the training involved to use and interpret RCM images currently limits RCM to large academic centers. Reimbursement may make more widespread use feasible. In any event, RCM imaging should be part of the curriculum for both dermatology and pathology trainees.
Future Directions
In vivo RCM is a noninvasive imaging modality that allows for real-time evaluation of the skin. Used in conjunction with dermoscopy, RCM can substantially improve diagnostic accuracy and reduce the number of unnecessary biopsies. Now that RCM has finally gained foundational CPT codes and insurance reimbursement, there may be a growing demand for clinicians to incorporate this technology into their clinical practice.
- Current Procedural Terminology 2017, Professional Edition. Chicago IL: American Medical Association; 2016.
- Que SK, Fraga-Braghiroli N, Grant-Kels JM, et al. Through the looking glass: basics and principles of reflectance confocal microscopy [published online June 4, 2015]. J Am Acad Dermatol. 2015;73:276-284.
- Rajadhyaksha M, Marghoob A, Rossi A, et al. Reflectance confocal microscopy of skin in vivo: from bench to bedside [published online October 27, 2016]. Lasers Surg Med. 2017;49:7-19.
- Xiong YD, Ma S, Li X, et al. A meta-analysis of reflectance confocal microscopy for the diagnosis of malignant skin tumours. J Eur Acad Dermatol Venereol. 2016;30:1295-1302.
- Stevenson AD, Mickan S, Mallett S, et al. Systematic review of diagnostic accuracy of reflectance confocal microscopy for melanoma diagnosis in patients with clinically equivocal skin lesions. Dermatol Pract Concept. 2013;3:19-27.
- Busam KJ, Hester K, Charles C, et al. Detection of clinically amelanotic malignant melanoma and assessment of its margins by in vivo confocal scanning laser microscopy. Arch Dermatol. 2001;137:923-929.
- Losi A, Longo C, Cesinaro AM, et al. Hyporeflective pagetoid cells: a new clue for amelanotic melanoma diagnosis by reflectance confocal microscopy. Br J Dermatol. 2014;171:48-54.
- Guitera P, Menzies SQ, Argenziano G, et al. Dermoscopy and in vivo confocal microscopy are complementary techniques for the diagnosis of difficult amelanotic and light-coloured skin lesions [published online October 12, 2016]. Br J Dermatol. 2016;175:1311-1319.
- Pellacani G, Pepe P, Casari A, et al. Reflectance confocal microscopy as a second-level examination in skin oncology improves diagnostic accuracy and saves unnecessary excisions: a longitudinal prospective study. Br J Dermatol. 2014;171:1044-1051.
- Pellacani G, Witkowski A, Cesinaro AM, et al. Cost-benefit of reflectance confocal microscopy in the diagnostic performance of melanoma. J Eur Acad Dermatol Venereol. 2016;30:413-419.
- Champin J, Perrot JL, Cinotti E, et al. In vivo reflectance confocal microscopy to optimize the spaghetti technique for defining surgical margins of lentigo maligna. Dermatol Surg. 2014;40:247-256.
- Hibler BP, Cordova M, Wong RJ, et al. Intraoperative real-time reflectance confocal microscopy for guiding surgical margins of lentigo maligna melanoma. Dermatol Surg. 2015;41:980-983.
- Ulrich M, Lange-Asschenfeldt S, Gonzalez S. The use of reflectance confocal microscopy for monitoring response to therapy of skin malignancies. Dermatol Pract Concept. 2012;2:202a10.
- Torres A, Niemeyer A, Berkes B, et al. 5% imiquimod cream and reflectance-mode confocal microscopy as adjunct modalities to Mohs micrographic surgery for treatment of basal cell carcinoma. Dermatol Surg. 2004;30(12, pt 1):1462-1469.
- Hashemi P, Pulitzer MP, Scope A, et al. Langerhans cells and melanocytes share similar morphologic features under in vivo reflectance confocal microscopy: a challenge for melanoma diagnosis. J Am Acad Dermatol. 2012;66:452-462.
- Menge TD, Hibler BP, Cordova MA, et al. Concordance of handheld reflectance confocal microscopy (RCM) with histopathology in the diagnosis of lentigo maligna (LM): a prospective study. J Am Acad Dermatol. 2016;74:1114-1120.
- Borsari S, Pampena R, Lallas A, et al. Clinical indications for use of reflectance confocal microscopy for skin cancer diagnosis. JAMA Dermatol. 2016;152:1093-1098.
- Current Procedural Terminology 2017, Professional Edition. Chicago IL: American Medical Association; 2016.
- Que SK, Fraga-Braghiroli N, Grant-Kels JM, et al. Through the looking glass: basics and principles of reflectance confocal microscopy [published online June 4, 2015]. J Am Acad Dermatol. 2015;73:276-284.
- Rajadhyaksha M, Marghoob A, Rossi A, et al. Reflectance confocal microscopy of skin in vivo: from bench to bedside [published online October 27, 2016]. Lasers Surg Med. 2017;49:7-19.
- Xiong YD, Ma S, Li X, et al. A meta-analysis of reflectance confocal microscopy for the diagnosis of malignant skin tumours. J Eur Acad Dermatol Venereol. 2016;30:1295-1302.
- Stevenson AD, Mickan S, Mallett S, et al. Systematic review of diagnostic accuracy of reflectance confocal microscopy for melanoma diagnosis in patients with clinically equivocal skin lesions. Dermatol Pract Concept. 2013;3:19-27.
- Busam KJ, Hester K, Charles C, et al. Detection of clinically amelanotic malignant melanoma and assessment of its margins by in vivo confocal scanning laser microscopy. Arch Dermatol. 2001;137:923-929.
- Losi A, Longo C, Cesinaro AM, et al. Hyporeflective pagetoid cells: a new clue for amelanotic melanoma diagnosis by reflectance confocal microscopy. Br J Dermatol. 2014;171:48-54.
- Guitera P, Menzies SQ, Argenziano G, et al. Dermoscopy and in vivo confocal microscopy are complementary techniques for the diagnosis of difficult amelanotic and light-coloured skin lesions [published online October 12, 2016]. Br J Dermatol. 2016;175:1311-1319.
- Pellacani G, Pepe P, Casari A, et al. Reflectance confocal microscopy as a second-level examination in skin oncology improves diagnostic accuracy and saves unnecessary excisions: a longitudinal prospective study. Br J Dermatol. 2014;171:1044-1051.
- Pellacani G, Witkowski A, Cesinaro AM, et al. Cost-benefit of reflectance confocal microscopy in the diagnostic performance of melanoma. J Eur Acad Dermatol Venereol. 2016;30:413-419.
- Champin J, Perrot JL, Cinotti E, et al. In vivo reflectance confocal microscopy to optimize the spaghetti technique for defining surgical margins of lentigo maligna. Dermatol Surg. 2014;40:247-256.
- Hibler BP, Cordova M, Wong RJ, et al. Intraoperative real-time reflectance confocal microscopy for guiding surgical margins of lentigo maligna melanoma. Dermatol Surg. 2015;41:980-983.
- Ulrich M, Lange-Asschenfeldt S, Gonzalez S. The use of reflectance confocal microscopy for monitoring response to therapy of skin malignancies. Dermatol Pract Concept. 2012;2:202a10.
- Torres A, Niemeyer A, Berkes B, et al. 5% imiquimod cream and reflectance-mode confocal microscopy as adjunct modalities to Mohs micrographic surgery for treatment of basal cell carcinoma. Dermatol Surg. 2004;30(12, pt 1):1462-1469.
- Hashemi P, Pulitzer MP, Scope A, et al. Langerhans cells and melanocytes share similar morphologic features under in vivo reflectance confocal microscopy: a challenge for melanoma diagnosis. J Am Acad Dermatol. 2012;66:452-462.
- Menge TD, Hibler BP, Cordova MA, et al. Concordance of handheld reflectance confocal microscopy (RCM) with histopathology in the diagnosis of lentigo maligna (LM): a prospective study. J Am Acad Dermatol. 2016;74:1114-1120.
- Borsari S, Pampena R, Lallas A, et al. Clinical indications for use of reflectance confocal microscopy for skin cancer diagnosis. JAMA Dermatol. 2016;152:1093-1098.
Practice Points
- Reflectance confocal microscopy (RCM) recently received Category I Current Procedural Terminology codes for reimbursement comparable to a skin biopsy.
- When used in combination with dermoscopy, RCM has been shown to increase diagnostic accuracy of skin cancer.
- Reflectance confocal microscopy also is useful in surgical treatment planning and monitoring nonsurgical treatments over time.
- Limitations of RCM imaging include low imaging depth, difficulty in imaging certain areas of the skin, learning curve for interpreting these images, and the cost of equipment.
Cutaneous laser surgery: Basic caution isn’t enough to prevent lawsuits
SAN DIEGO – Injuries and lawsuits related to laser cosmetic surgery are increasing and potential legal threats are not always easy to predict, according to two dermatologists who spoke at the annual meeting of the American Society for Laser Medicine and Surgery (ASLMS).
A laser procedure could go smoothly, for example, but the patient might be able to successfully sue if he or she is allowed to drive home after receiving a sedative. Or a physician might get sued because his or her nurse set a laser at the wrong setting and singed a patient.
The risk of a lawsuit is high, H. Ray Jalian, MD, a dermatologist in Los Angeles, said at the meeting. “The reality is that we’re all at some point going to face this.”
The most common procedure litigated was laser hair removal, making up almost 40% of the cases, which is not an indication that this particular procedure is dangerous, Dr. Jalian said. “It’s quite safe, and the complication rate is quite low,” but more of these procedures are being done, he noted. Rejuvenation procedures followed, accounting for 25% of cases.
The alleged injuries sustained from laser surgery included burns (47%), scars (39%), and pigmentation problems (24%). Deaths occurred in just over 2% of the cases. In the study, almost a third of plaintiffs alleged that they were not provided informed consent. Plaintiffs also alleged fraud (9%) and assault/battery (5%), and a family member occasionally sued for loss of consortium (8% of cases). The specialty with the largest percentage of the cases was plastic surgery (26%), followed by dermatology (21%).
Dr. Jalian and his copresenter, Mathew Avram, MD, JD, director of the Dermatology Laser & Cosmetic Center, and director of dermatologic surgery at Massachusetts General Hospital, Boston, offered these lessons about the legal risks associated with laser procedures:
• You may have a duty to protect your patient from bad choices.
Physicians aren’t expected to keep patients from making certain bad decisions such as sunbathing after a traditional resurfacing procedure, said Dr. Avram, of the department of dermatology, Harvard Medical School, Boston, and the ASLMS president. But in some cases, he said, the law may expect the physician to step in to prevent harm. For example, he said, a patient who has undergone a fractional ablative laser procedure and has received a sedative should not be allowed to drive home.
• You may get sued even if your employee is at fault.
The 2013 study found physicians were often sued even when they did not perform the laser procedure in question. Nonphysicians such as physician assistants and nurses often perform laser operations, and many states allow them to do so. “Nonphysicians were less likely to be sued even if they were the operators,” Dr. Jalian said. In the study, almost 38% of the 174 analyzed cases involved nonphysician operators, but they were sued in just 26% of the cases. In 33 of the 174 cases in the study, plaintiffs alleged failure to properly hire, train, or supervise staff.
He recommended looking at state laws, which differ greatly in their regulations – or lack of them – regarding the operation of medical lasers. In some cases, physicians must supervise laser use, he said. “But what are the requirements? Can you be available by phone down the street or in the Caribbean?”
Dr. Jalian, Dr. Avram, and a colleague followed up the 2013 study with another study that tracked 175 legal cases from 1999 to 2012 involving alleged injuries from cutaneous laser surgery. During this time period, 75 (43%) involved a nonphysician operating a laser, increasing from 36% in 2008 to 78% in 2012.
In almost two-thirds of cases, the procedures in question were done by nonphysicians outside a “traditional medical setting” such as a salon or spa (JAMA Dermatol. 2014 Apr;150[4]:407-11).
• Delayed side effects could mean delayed lawsuits.
According to Dr. Avram, statutes of limitations – the length of time in which a patient can file a lawsuit – typically last for 2-3 years in malpractice cases. But he said that the period begins when the physician is alleged to have made a mistake or when the patient becomes aware of – or should reasonably be aware of – an injury. Therefore, physicians could face legal trouble over delayed hypopigmentation that appears 6 months after a laser resurfacing treatment, or granulomas that appear years after a filler treatment, he said.
• A signed form is not a cure-all.
It is wise to make patients sign an extensive informed consent form, but this will not protect a physician against a claim of negligence, Dr. Avram said. And the reverse is also true: If a patient did not sign a proper consent form, he or she could still sue even if the procedure went perfectly, he noted.
• Your instincts are worth trusting.
When it comes to lawsuit prevention, Dr. Avram said, “by far the most important thing you can do happens within a minute of when you see the patient. Assess and trust your own intuition and your staff’s intuition. For elective, cosmetic treatments, don’t be afraid to say no. There’s no legal obligation to perform a cosmetic treatment on a patient.”
If you do choose to treat a patient, he advised, be open about the procedure and “maybe even tell them some of the tougher, worse-case scenarios.” If a procedure goes poorly, he said, consider how to fix it. “Many complications can be significantly improved or cleared with timely and appropriate intervention,” he said.
In some cases, refunding the patient’s money can be considered, with the patient signing a release, he said. “Document that you are refunding the money in order to preserve the doctor-patient relationship, not to avoid negligence.”
Dr. Jalian and Dr. Avram reported no relevant disclosures.
SAN DIEGO – Injuries and lawsuits related to laser cosmetic surgery are increasing and potential legal threats are not always easy to predict, according to two dermatologists who spoke at the annual meeting of the American Society for Laser Medicine and Surgery (ASLMS).
A laser procedure could go smoothly, for example, but the patient might be able to successfully sue if he or she is allowed to drive home after receiving a sedative. Or a physician might get sued because his or her nurse set a laser at the wrong setting and singed a patient.
The risk of a lawsuit is high, H. Ray Jalian, MD, a dermatologist in Los Angeles, said at the meeting. “The reality is that we’re all at some point going to face this.”
The most common procedure litigated was laser hair removal, making up almost 40% of the cases, which is not an indication that this particular procedure is dangerous, Dr. Jalian said. “It’s quite safe, and the complication rate is quite low,” but more of these procedures are being done, he noted. Rejuvenation procedures followed, accounting for 25% of cases.
The alleged injuries sustained from laser surgery included burns (47%), scars (39%), and pigmentation problems (24%). Deaths occurred in just over 2% of the cases. In the study, almost a third of plaintiffs alleged that they were not provided informed consent. Plaintiffs also alleged fraud (9%) and assault/battery (5%), and a family member occasionally sued for loss of consortium (8% of cases). The specialty with the largest percentage of the cases was plastic surgery (26%), followed by dermatology (21%).
Dr. Jalian and his copresenter, Mathew Avram, MD, JD, director of the Dermatology Laser & Cosmetic Center, and director of dermatologic surgery at Massachusetts General Hospital, Boston, offered these lessons about the legal risks associated with laser procedures:
• You may have a duty to protect your patient from bad choices.
Physicians aren’t expected to keep patients from making certain bad decisions such as sunbathing after a traditional resurfacing procedure, said Dr. Avram, of the department of dermatology, Harvard Medical School, Boston, and the ASLMS president. But in some cases, he said, the law may expect the physician to step in to prevent harm. For example, he said, a patient who has undergone a fractional ablative laser procedure and has received a sedative should not be allowed to drive home.
• You may get sued even if your employee is at fault.
The 2013 study found physicians were often sued even when they did not perform the laser procedure in question. Nonphysicians such as physician assistants and nurses often perform laser operations, and many states allow them to do so. “Nonphysicians were less likely to be sued even if they were the operators,” Dr. Jalian said. In the study, almost 38% of the 174 analyzed cases involved nonphysician operators, but they were sued in just 26% of the cases. In 33 of the 174 cases in the study, plaintiffs alleged failure to properly hire, train, or supervise staff.
He recommended looking at state laws, which differ greatly in their regulations – or lack of them – regarding the operation of medical lasers. In some cases, physicians must supervise laser use, he said. “But what are the requirements? Can you be available by phone down the street or in the Caribbean?”
Dr. Jalian, Dr. Avram, and a colleague followed up the 2013 study with another study that tracked 175 legal cases from 1999 to 2012 involving alleged injuries from cutaneous laser surgery. During this time period, 75 (43%) involved a nonphysician operating a laser, increasing from 36% in 2008 to 78% in 2012.
In almost two-thirds of cases, the procedures in question were done by nonphysicians outside a “traditional medical setting” such as a salon or spa (JAMA Dermatol. 2014 Apr;150[4]:407-11).
• Delayed side effects could mean delayed lawsuits.
According to Dr. Avram, statutes of limitations – the length of time in which a patient can file a lawsuit – typically last for 2-3 years in malpractice cases. But he said that the period begins when the physician is alleged to have made a mistake or when the patient becomes aware of – or should reasonably be aware of – an injury. Therefore, physicians could face legal trouble over delayed hypopigmentation that appears 6 months after a laser resurfacing treatment, or granulomas that appear years after a filler treatment, he said.
• A signed form is not a cure-all.
It is wise to make patients sign an extensive informed consent form, but this will not protect a physician against a claim of negligence, Dr. Avram said. And the reverse is also true: If a patient did not sign a proper consent form, he or she could still sue even if the procedure went perfectly, he noted.
• Your instincts are worth trusting.
When it comes to lawsuit prevention, Dr. Avram said, “by far the most important thing you can do happens within a minute of when you see the patient. Assess and trust your own intuition and your staff’s intuition. For elective, cosmetic treatments, don’t be afraid to say no. There’s no legal obligation to perform a cosmetic treatment on a patient.”
If you do choose to treat a patient, he advised, be open about the procedure and “maybe even tell them some of the tougher, worse-case scenarios.” If a procedure goes poorly, he said, consider how to fix it. “Many complications can be significantly improved or cleared with timely and appropriate intervention,” he said.
In some cases, refunding the patient’s money can be considered, with the patient signing a release, he said. “Document that you are refunding the money in order to preserve the doctor-patient relationship, not to avoid negligence.”
Dr. Jalian and Dr. Avram reported no relevant disclosures.
SAN DIEGO – Injuries and lawsuits related to laser cosmetic surgery are increasing and potential legal threats are not always easy to predict, according to two dermatologists who spoke at the annual meeting of the American Society for Laser Medicine and Surgery (ASLMS).
A laser procedure could go smoothly, for example, but the patient might be able to successfully sue if he or she is allowed to drive home after receiving a sedative. Or a physician might get sued because his or her nurse set a laser at the wrong setting and singed a patient.
The risk of a lawsuit is high, H. Ray Jalian, MD, a dermatologist in Los Angeles, said at the meeting. “The reality is that we’re all at some point going to face this.”
The most common procedure litigated was laser hair removal, making up almost 40% of the cases, which is not an indication that this particular procedure is dangerous, Dr. Jalian said. “It’s quite safe, and the complication rate is quite low,” but more of these procedures are being done, he noted. Rejuvenation procedures followed, accounting for 25% of cases.
The alleged injuries sustained from laser surgery included burns (47%), scars (39%), and pigmentation problems (24%). Deaths occurred in just over 2% of the cases. In the study, almost a third of plaintiffs alleged that they were not provided informed consent. Plaintiffs also alleged fraud (9%) and assault/battery (5%), and a family member occasionally sued for loss of consortium (8% of cases). The specialty with the largest percentage of the cases was plastic surgery (26%), followed by dermatology (21%).
Dr. Jalian and his copresenter, Mathew Avram, MD, JD, director of the Dermatology Laser & Cosmetic Center, and director of dermatologic surgery at Massachusetts General Hospital, Boston, offered these lessons about the legal risks associated with laser procedures:
• You may have a duty to protect your patient from bad choices.
Physicians aren’t expected to keep patients from making certain bad decisions such as sunbathing after a traditional resurfacing procedure, said Dr. Avram, of the department of dermatology, Harvard Medical School, Boston, and the ASLMS president. But in some cases, he said, the law may expect the physician to step in to prevent harm. For example, he said, a patient who has undergone a fractional ablative laser procedure and has received a sedative should not be allowed to drive home.
• You may get sued even if your employee is at fault.
The 2013 study found physicians were often sued even when they did not perform the laser procedure in question. Nonphysicians such as physician assistants and nurses often perform laser operations, and many states allow them to do so. “Nonphysicians were less likely to be sued even if they were the operators,” Dr. Jalian said. In the study, almost 38% of the 174 analyzed cases involved nonphysician operators, but they were sued in just 26% of the cases. In 33 of the 174 cases in the study, plaintiffs alleged failure to properly hire, train, or supervise staff.
He recommended looking at state laws, which differ greatly in their regulations – or lack of them – regarding the operation of medical lasers. In some cases, physicians must supervise laser use, he said. “But what are the requirements? Can you be available by phone down the street or in the Caribbean?”
Dr. Jalian, Dr. Avram, and a colleague followed up the 2013 study with another study that tracked 175 legal cases from 1999 to 2012 involving alleged injuries from cutaneous laser surgery. During this time period, 75 (43%) involved a nonphysician operating a laser, increasing from 36% in 2008 to 78% in 2012.
In almost two-thirds of cases, the procedures in question were done by nonphysicians outside a “traditional medical setting” such as a salon or spa (JAMA Dermatol. 2014 Apr;150[4]:407-11).
• Delayed side effects could mean delayed lawsuits.
According to Dr. Avram, statutes of limitations – the length of time in which a patient can file a lawsuit – typically last for 2-3 years in malpractice cases. But he said that the period begins when the physician is alleged to have made a mistake or when the patient becomes aware of – or should reasonably be aware of – an injury. Therefore, physicians could face legal trouble over delayed hypopigmentation that appears 6 months after a laser resurfacing treatment, or granulomas that appear years after a filler treatment, he said.
• A signed form is not a cure-all.
It is wise to make patients sign an extensive informed consent form, but this will not protect a physician against a claim of negligence, Dr. Avram said. And the reverse is also true: If a patient did not sign a proper consent form, he or she could still sue even if the procedure went perfectly, he noted.
• Your instincts are worth trusting.
When it comes to lawsuit prevention, Dr. Avram said, “by far the most important thing you can do happens within a minute of when you see the patient. Assess and trust your own intuition and your staff’s intuition. For elective, cosmetic treatments, don’t be afraid to say no. There’s no legal obligation to perform a cosmetic treatment on a patient.”
If you do choose to treat a patient, he advised, be open about the procedure and “maybe even tell them some of the tougher, worse-case scenarios.” If a procedure goes poorly, he said, consider how to fix it. “Many complications can be significantly improved or cleared with timely and appropriate intervention,” he said.
In some cases, refunding the patient’s money can be considered, with the patient signing a release, he said. “Document that you are refunding the money in order to preserve the doctor-patient relationship, not to avoid negligence.”
Dr. Jalian and Dr. Avram reported no relevant disclosures.
AT LASER 2017
Minimally invasive cosmetic procedures more popular than ever
The total number of minimally invasive cosmetic procedures topped 15 million for the first time in 2016, with onabotulinumtoxinA injections leading the way, according to the American Society of Plastic Surgeons.
An estimated 15.4 million minimally invasive cosmetic procedures were performed last year, with onabotulinumtoxinA making up over 45% – approximately 7.1 million anatomic sites injected – of that total, ASPS data show.
Next in popularity was injection of soft tissue fillers, with just over 2.6 million procedures – almost 17% of the procedures performed in 2016 – followed by chemical peels with almost 1.4 million procedures, laser hair removal with 1.1 million procedures, and microdermabrasion at 775,000, the ASPS reported.
The total number of minimally invasive procedures was up 3% from the 14.96 million performed in 2015, while onabotulinumtoxinA was up 4%, soft tissue fillers were up 2%, chemical peels were up 4%, laser hair removal was down 1%, and microdermabrasion was down 3%. Larger changes were seen among some less common procedures: injection lipolysis was up 18% (55,660 procedures in 2016), fat injection was up 13% (79,208 procedures in 2016), and acellular dermal matrix procedures were down 18% (7,809 in 2016), according to the ASPS Tracking Operations and Outcomes for Plastic Surgeons database and an annual survey of board-certified dermatologists, otolaryngologists, and plastic surgeons (final sample = 703).
The total number of minimally invasive cosmetic procedures topped 15 million for the first time in 2016, with onabotulinumtoxinA injections leading the way, according to the American Society of Plastic Surgeons.
An estimated 15.4 million minimally invasive cosmetic procedures were performed last year, with onabotulinumtoxinA making up over 45% – approximately 7.1 million anatomic sites injected – of that total, ASPS data show.
Next in popularity was injection of soft tissue fillers, with just over 2.6 million procedures – almost 17% of the procedures performed in 2016 – followed by chemical peels with almost 1.4 million procedures, laser hair removal with 1.1 million procedures, and microdermabrasion at 775,000, the ASPS reported.
The total number of minimally invasive procedures was up 3% from the 14.96 million performed in 2015, while onabotulinumtoxinA was up 4%, soft tissue fillers were up 2%, chemical peels were up 4%, laser hair removal was down 1%, and microdermabrasion was down 3%. Larger changes were seen among some less common procedures: injection lipolysis was up 18% (55,660 procedures in 2016), fat injection was up 13% (79,208 procedures in 2016), and acellular dermal matrix procedures were down 18% (7,809 in 2016), according to the ASPS Tracking Operations and Outcomes for Plastic Surgeons database and an annual survey of board-certified dermatologists, otolaryngologists, and plastic surgeons (final sample = 703).
The total number of minimally invasive cosmetic procedures topped 15 million for the first time in 2016, with onabotulinumtoxinA injections leading the way, according to the American Society of Plastic Surgeons.
An estimated 15.4 million minimally invasive cosmetic procedures were performed last year, with onabotulinumtoxinA making up over 45% – approximately 7.1 million anatomic sites injected – of that total, ASPS data show.
Next in popularity was injection of soft tissue fillers, with just over 2.6 million procedures – almost 17% of the procedures performed in 2016 – followed by chemical peels with almost 1.4 million procedures, laser hair removal with 1.1 million procedures, and microdermabrasion at 775,000, the ASPS reported.
The total number of minimally invasive procedures was up 3% from the 14.96 million performed in 2015, while onabotulinumtoxinA was up 4%, soft tissue fillers were up 2%, chemical peels were up 4%, laser hair removal was down 1%, and microdermabrasion was down 3%. Larger changes were seen among some less common procedures: injection lipolysis was up 18% (55,660 procedures in 2016), fat injection was up 13% (79,208 procedures in 2016), and acellular dermal matrix procedures were down 18% (7,809 in 2016), according to the ASPS Tracking Operations and Outcomes for Plastic Surgeons database and an annual survey of board-certified dermatologists, otolaryngologists, and plastic surgeons (final sample = 703).
Cosmetic Corner: Dermatologists Weigh in on Face Scrubs
To improve patient care and outcomes, leading dermatologists offered their recommendations on face scrubs. Consideration must be given to:
- Crystal Peel Microdermabrasion Exfoliating Face Crème
Formulary for Physicians, Inc
“This product is a highly effective facial scrub for patients with thick skin. Its exfoliating ingredient is corundum, another name for aluminum oxide, the crystal used by most microabrasion machines.”— Mark G. Rubin, MD, Beverly Hills, California
- Facial Fuel Energizing Scrub
Kiehl’s
Recommended by Gary Goldenberg, MD, New York, New York
- Olay Regenerist Regenerating Cream Cleanser
Procter & Gamble
“Oxygenated beads in the creamy formula help to gently exfoliate the skin without overdrying and stripping the skin’s outer layer, leaving the skin soft and fresh.”—Jeannette Graf, MD, New York, New York
- PRESCRIBEDsolutions: Starting Up/Face, Surface Improvement
Biopelle, Inc
“I use Starting Up/Face as my daily cleanser, as it contains salicylic acid and helps improve the overall texture plus minimize bumps from shaving, and Surface Improvement about every other day on my face in the shower.”—Joel L. Cohen, MD, Greenwood Village, Colorado
- St. Ives Apricot Blemish Control Scrub
Unilever
“It exfoliates and has salicylic acid. After exfoliating, I recommend allowing it to sit on the skin for 5 minutes before washing off.”—Anthony M. Rossi, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, cleansing devices, and redness-reducing products will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on face scrubs. Consideration must be given to:
- Crystal Peel Microdermabrasion Exfoliating Face Crème
Formulary for Physicians, Inc
“This product is a highly effective facial scrub for patients with thick skin. Its exfoliating ingredient is corundum, another name for aluminum oxide, the crystal used by most microabrasion machines.”— Mark G. Rubin, MD, Beverly Hills, California
- Facial Fuel Energizing Scrub
Kiehl’s
Recommended by Gary Goldenberg, MD, New York, New York
- Olay Regenerist Regenerating Cream Cleanser
Procter & Gamble
“Oxygenated beads in the creamy formula help to gently exfoliate the skin without overdrying and stripping the skin’s outer layer, leaving the skin soft and fresh.”—Jeannette Graf, MD, New York, New York
- PRESCRIBEDsolutions: Starting Up/Face, Surface Improvement
Biopelle, Inc
“I use Starting Up/Face as my daily cleanser, as it contains salicylic acid and helps improve the overall texture plus minimize bumps from shaving, and Surface Improvement about every other day on my face in the shower.”—Joel L. Cohen, MD, Greenwood Village, Colorado
- St. Ives Apricot Blemish Control Scrub
Unilever
“It exfoliates and has salicylic acid. After exfoliating, I recommend allowing it to sit on the skin for 5 minutes before washing off.”—Anthony M. Rossi, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, cleansing devices, and redness-reducing products will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on face scrubs. Consideration must be given to:
- Crystal Peel Microdermabrasion Exfoliating Face Crème
Formulary for Physicians, Inc
“This product is a highly effective facial scrub for patients with thick skin. Its exfoliating ingredient is corundum, another name for aluminum oxide, the crystal used by most microabrasion machines.”— Mark G. Rubin, MD, Beverly Hills, California
- Facial Fuel Energizing Scrub
Kiehl’s
Recommended by Gary Goldenberg, MD, New York, New York
- Olay Regenerist Regenerating Cream Cleanser
Procter & Gamble
“Oxygenated beads in the creamy formula help to gently exfoliate the skin without overdrying and stripping the skin’s outer layer, leaving the skin soft and fresh.”—Jeannette Graf, MD, New York, New York
- PRESCRIBEDsolutions: Starting Up/Face, Surface Improvement
Biopelle, Inc
“I use Starting Up/Face as my daily cleanser, as it contains salicylic acid and helps improve the overall texture plus minimize bumps from shaving, and Surface Improvement about every other day on my face in the shower.”—Joel L. Cohen, MD, Greenwood Village, Colorado
- St. Ives Apricot Blemish Control Scrub
Unilever
“It exfoliates and has salicylic acid. After exfoliating, I recommend allowing it to sit on the skin for 5 minutes before washing off.”—Anthony M. Rossi, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete’s foot treatments, cleansing devices, and redness-reducing products will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Gray hair
Besides skin wrinkling, volume shifts, and photoaging, graying hair can also be a telltale sign of aging. While it was recently a fashionable trend for younger persons to dye their hair white or gray, graying hair can make a younger person appear older, even in those with naturally premature graying of the hair.
In a study recently published in Genes & Development, researchers at the University of Texas Southwestern Medical Center, Dallas, identified hair shaft progenitors in the matrix that are specific to the hair shaft and not to follicular epithelial cells.1 These hair shaft progenitors express transcription factor KROX20, which expresses stem cell growth factor necessary for hair pigmentation by maintenance of differentiated melanocytes. When KROX20+ is depleted, hair growth is halted and hair turns gray, proving its important role in both hair growth and graying pathways.
Other mechanisms for hair graying include oxidative stress to the hair, at the level of the melanocyte stem cell or at the end-stage of the hair melanocyte, resulting in follicular melanocyte death. With aging and certain genetic mutations (such as that seen in Chediak-Higashi syndrome), reduction of catalase and sometimes downregulation of antioxidant proteins such as BCL-2 and TRP-2 are reduced, resulting in higher reactive oxygen species (ROS) that lead to bulbar melanocyte malfunction and death.
Last year, for the first time, researchers at University College of London identified a gene involved in gray hair, the interferon regulatory factor 4 gene (IRF4).2 The IRF4 gene is involved in regulating production and storage of melanin.
Besides photoprotection and vitamin antioxidants as a preventive measure, therapies that have been developed to target the reduction of ROS in hair have been largely unsatisfactory in treating gray hair. Most people either allow their hair to gray or dye their hair, which can be time consuming and costly and is required on a more frequent basis over time – not to mention the distress related to allergic contact dermatitis caused by some components of some hair dyes, including paraphenylenediamine, which we sometimes see in our profession.
Knowledge of KROX20+, the IRF4 gene, and other pathways involved may be useful in developing novel treatments to prevent or treat graying hair. Information regarding the use of platelet rich plasma (PRP) for hair growth is increasingly being published in the literature. While some physicians purport seeing a reversal in graying with scalp PRP injections, the majority say the results are not universal.
Currently, there are no published studies evaluating the effects of PRP on gray hair. Perhaps providing stem cell factors via injections of PRP or other growth factors may aid not only in hair regrowth but in preserving pigmentation and repigmentation.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References:
1. Genes Dev. 2017 May 2. doi: 10.1101/gad.298703.117.
2. Nat Commun. 2016 Mar 1;7:10815.
Besides skin wrinkling, volume shifts, and photoaging, graying hair can also be a telltale sign of aging. While it was recently a fashionable trend for younger persons to dye their hair white or gray, graying hair can make a younger person appear older, even in those with naturally premature graying of the hair.
In a study recently published in Genes & Development, researchers at the University of Texas Southwestern Medical Center, Dallas, identified hair shaft progenitors in the matrix that are specific to the hair shaft and not to follicular epithelial cells.1 These hair shaft progenitors express transcription factor KROX20, which expresses stem cell growth factor necessary for hair pigmentation by maintenance of differentiated melanocytes. When KROX20+ is depleted, hair growth is halted and hair turns gray, proving its important role in both hair growth and graying pathways.
Other mechanisms for hair graying include oxidative stress to the hair, at the level of the melanocyte stem cell or at the end-stage of the hair melanocyte, resulting in follicular melanocyte death. With aging and certain genetic mutations (such as that seen in Chediak-Higashi syndrome), reduction of catalase and sometimes downregulation of antioxidant proteins such as BCL-2 and TRP-2 are reduced, resulting in higher reactive oxygen species (ROS) that lead to bulbar melanocyte malfunction and death.
Last year, for the first time, researchers at University College of London identified a gene involved in gray hair, the interferon regulatory factor 4 gene (IRF4).2 The IRF4 gene is involved in regulating production and storage of melanin.
Besides photoprotection and vitamin antioxidants as a preventive measure, therapies that have been developed to target the reduction of ROS in hair have been largely unsatisfactory in treating gray hair. Most people either allow their hair to gray or dye their hair, which can be time consuming and costly and is required on a more frequent basis over time – not to mention the distress related to allergic contact dermatitis caused by some components of some hair dyes, including paraphenylenediamine, which we sometimes see in our profession.
Knowledge of KROX20+, the IRF4 gene, and other pathways involved may be useful in developing novel treatments to prevent or treat graying hair. Information regarding the use of platelet rich plasma (PRP) for hair growth is increasingly being published in the literature. While some physicians purport seeing a reversal in graying with scalp PRP injections, the majority say the results are not universal.
Currently, there are no published studies evaluating the effects of PRP on gray hair. Perhaps providing stem cell factors via injections of PRP or other growth factors may aid not only in hair regrowth but in preserving pigmentation and repigmentation.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References:
1. Genes Dev. 2017 May 2. doi: 10.1101/gad.298703.117.
2. Nat Commun. 2016 Mar 1;7:10815.
Besides skin wrinkling, volume shifts, and photoaging, graying hair can also be a telltale sign of aging. While it was recently a fashionable trend for younger persons to dye their hair white or gray, graying hair can make a younger person appear older, even in those with naturally premature graying of the hair.
In a study recently published in Genes & Development, researchers at the University of Texas Southwestern Medical Center, Dallas, identified hair shaft progenitors in the matrix that are specific to the hair shaft and not to follicular epithelial cells.1 These hair shaft progenitors express transcription factor KROX20, which expresses stem cell growth factor necessary for hair pigmentation by maintenance of differentiated melanocytes. When KROX20+ is depleted, hair growth is halted and hair turns gray, proving its important role in both hair growth and graying pathways.
Other mechanisms for hair graying include oxidative stress to the hair, at the level of the melanocyte stem cell or at the end-stage of the hair melanocyte, resulting in follicular melanocyte death. With aging and certain genetic mutations (such as that seen in Chediak-Higashi syndrome), reduction of catalase and sometimes downregulation of antioxidant proteins such as BCL-2 and TRP-2 are reduced, resulting in higher reactive oxygen species (ROS) that lead to bulbar melanocyte malfunction and death.
Last year, for the first time, researchers at University College of London identified a gene involved in gray hair, the interferon regulatory factor 4 gene (IRF4).2 The IRF4 gene is involved in regulating production and storage of melanin.
Besides photoprotection and vitamin antioxidants as a preventive measure, therapies that have been developed to target the reduction of ROS in hair have been largely unsatisfactory in treating gray hair. Most people either allow their hair to gray or dye their hair, which can be time consuming and costly and is required on a more frequent basis over time – not to mention the distress related to allergic contact dermatitis caused by some components of some hair dyes, including paraphenylenediamine, which we sometimes see in our profession.
Knowledge of KROX20+, the IRF4 gene, and other pathways involved may be useful in developing novel treatments to prevent or treat graying hair. Information regarding the use of platelet rich plasma (PRP) for hair growth is increasingly being published in the literature. While some physicians purport seeing a reversal in graying with scalp PRP injections, the majority say the results are not universal.
Currently, there are no published studies evaluating the effects of PRP on gray hair. Perhaps providing stem cell factors via injections of PRP or other growth factors may aid not only in hair regrowth but in preserving pigmentation and repigmentation.
Dr. Wesley and Dr. Talakoub are cocontributors to this column. Dr. Wesley practices dermatology in Beverly Hills, Calif. Dr. Talakoub is in private practice in McLean, Va. This month’s column is by Dr. Wesley. Write to them at [email protected]. They had no relevant disclosures.
References:
1. Genes Dev. 2017 May 2. doi: 10.1101/gad.298703.117.
2. Nat Commun. 2016 Mar 1;7:10815.
RF, IPL score highest for facial rejuvenation in review
SAN DIEGO – A review of existing research found that newer technologies are safer and more effective at skin rejuvenation than older ones, with two types – intense pulsed laser (IPL) and radiofrequency (RF) – at or near the head of the pack on both fronts.
In addition, 10 types of treatments, including multiple laser technologies, scored higher than facial peels on safety, although the peels beat almost all comers in terms of efficacy, the study’s lead author, Caerwyn Ash, PhD, said in an interview after presenting the results at the annual meeting of the American Society for Laser Medicine and Surgery.
The review is unique because it maps the various treatments on two axes – safety and efficacy – in a chart. Viewers gain an instant perspective on how the individual treatments fare when stacked up against each other. “The study gives credence to the new technologies,” said Dr. Ash, associate professor of medical devices at the University of Wales Trinity Saint David, Swansea. “We’re moving toward safer and more efficacious results.”
He and his colleagues analyzed more than 500 studies published since 1985, which evaluated 15 different types of technology used for facial rejuvenation. They reviewed at least 35 clinical studies per platform. They also reported on the safety and efficacy of facial peels.
The technologies examined included RF, light emitting diode (LED), pulsed dyed laser (PDL), frequency doubled 532-nm potassium-titanyl-phosphate neodymium:YAG laser (which had a low number of clinical studies), 980-nm diode laser, Q-switched Nd:YAG laser, 1320-nm Nd:YAG laser, 1540-nm Erbium:glass laser, 1450-nm diode laser, IPL, long pulsed Nd:YAG laser, plasma, and copper bromide laser.
Another technology reviewed was fractional selective photothermolysis, but studies of this modality were difficult to compare, according to the authors. Also included was the carbon dioxide laser, which the authors described as being hampered by long healing times and discomfort that requires anesthesia.
The review notes that comparisons between studies of the different treatments in the review were limited by such factors as varied beam and tip sizes (even within individual studies), various pulse durations, and different treatment times and intervals.
The researchers mapped the treatments in a single chart on two axes, safety and efficacy.
Three treatments scored the best in terms of efficacy: RF, IPL and chemical peels. However, while RF and IPL scored near the top of all treatments on the safety scale, chemical peels pulled up the rear.
The treatment strategies on the safety scale, ranked from most safe to least safe, were LED, IPL, diode laser, RF, argon laser, PDL and carbon dioxide laser, Nd:YAG, ablative Erbium, plasma, and chemical peel.
Radiofrequency topped the efficacy scale, followed closely by chemical peel and IPL. Following them, from most efficacious to least efficacious, were carbon dioxide laser, ablative Erbium, plasma treatments, and PDL, Nd:YAG and argon laser, diode laser, and LED.
The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
SAN DIEGO – A review of existing research found that newer technologies are safer and more effective at skin rejuvenation than older ones, with two types – intense pulsed laser (IPL) and radiofrequency (RF) – at or near the head of the pack on both fronts.
In addition, 10 types of treatments, including multiple laser technologies, scored higher than facial peels on safety, although the peels beat almost all comers in terms of efficacy, the study’s lead author, Caerwyn Ash, PhD, said in an interview after presenting the results at the annual meeting of the American Society for Laser Medicine and Surgery.
The review is unique because it maps the various treatments on two axes – safety and efficacy – in a chart. Viewers gain an instant perspective on how the individual treatments fare when stacked up against each other. “The study gives credence to the new technologies,” said Dr. Ash, associate professor of medical devices at the University of Wales Trinity Saint David, Swansea. “We’re moving toward safer and more efficacious results.”
He and his colleagues analyzed more than 500 studies published since 1985, which evaluated 15 different types of technology used for facial rejuvenation. They reviewed at least 35 clinical studies per platform. They also reported on the safety and efficacy of facial peels.
The technologies examined included RF, light emitting diode (LED), pulsed dyed laser (PDL), frequency doubled 532-nm potassium-titanyl-phosphate neodymium:YAG laser (which had a low number of clinical studies), 980-nm diode laser, Q-switched Nd:YAG laser, 1320-nm Nd:YAG laser, 1540-nm Erbium:glass laser, 1450-nm diode laser, IPL, long pulsed Nd:YAG laser, plasma, and copper bromide laser.
Another technology reviewed was fractional selective photothermolysis, but studies of this modality were difficult to compare, according to the authors. Also included was the carbon dioxide laser, which the authors described as being hampered by long healing times and discomfort that requires anesthesia.
The review notes that comparisons between studies of the different treatments in the review were limited by such factors as varied beam and tip sizes (even within individual studies), various pulse durations, and different treatment times and intervals.
The researchers mapped the treatments in a single chart on two axes, safety and efficacy.
Three treatments scored the best in terms of efficacy: RF, IPL and chemical peels. However, while RF and IPL scored near the top of all treatments on the safety scale, chemical peels pulled up the rear.
The treatment strategies on the safety scale, ranked from most safe to least safe, were LED, IPL, diode laser, RF, argon laser, PDL and carbon dioxide laser, Nd:YAG, ablative Erbium, plasma, and chemical peel.
Radiofrequency topped the efficacy scale, followed closely by chemical peel and IPL. Following them, from most efficacious to least efficacious, were carbon dioxide laser, ablative Erbium, plasma treatments, and PDL, Nd:YAG and argon laser, diode laser, and LED.
The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
SAN DIEGO – A review of existing research found that newer technologies are safer and more effective at skin rejuvenation than older ones, with two types – intense pulsed laser (IPL) and radiofrequency (RF) – at or near the head of the pack on both fronts.
In addition, 10 types of treatments, including multiple laser technologies, scored higher than facial peels on safety, although the peels beat almost all comers in terms of efficacy, the study’s lead author, Caerwyn Ash, PhD, said in an interview after presenting the results at the annual meeting of the American Society for Laser Medicine and Surgery.
The review is unique because it maps the various treatments on two axes – safety and efficacy – in a chart. Viewers gain an instant perspective on how the individual treatments fare when stacked up against each other. “The study gives credence to the new technologies,” said Dr. Ash, associate professor of medical devices at the University of Wales Trinity Saint David, Swansea. “We’re moving toward safer and more efficacious results.”
He and his colleagues analyzed more than 500 studies published since 1985, which evaluated 15 different types of technology used for facial rejuvenation. They reviewed at least 35 clinical studies per platform. They also reported on the safety and efficacy of facial peels.
The technologies examined included RF, light emitting diode (LED), pulsed dyed laser (PDL), frequency doubled 532-nm potassium-titanyl-phosphate neodymium:YAG laser (which had a low number of clinical studies), 980-nm diode laser, Q-switched Nd:YAG laser, 1320-nm Nd:YAG laser, 1540-nm Erbium:glass laser, 1450-nm diode laser, IPL, long pulsed Nd:YAG laser, plasma, and copper bromide laser.
Another technology reviewed was fractional selective photothermolysis, but studies of this modality were difficult to compare, according to the authors. Also included was the carbon dioxide laser, which the authors described as being hampered by long healing times and discomfort that requires anesthesia.
The review notes that comparisons between studies of the different treatments in the review were limited by such factors as varied beam and tip sizes (even within individual studies), various pulse durations, and different treatment times and intervals.
The researchers mapped the treatments in a single chart on two axes, safety and efficacy.
Three treatments scored the best in terms of efficacy: RF, IPL and chemical peels. However, while RF and IPL scored near the top of all treatments on the safety scale, chemical peels pulled up the rear.
The treatment strategies on the safety scale, ranked from most safe to least safe, were LED, IPL, diode laser, RF, argon laser, PDL and carbon dioxide laser, Nd:YAG, ablative Erbium, plasma, and chemical peel.
Radiofrequency topped the efficacy scale, followed closely by chemical peel and IPL. Following them, from most efficacious to least efficacious, were carbon dioxide laser, ablative Erbium, plasma treatments, and PDL, Nd:YAG and argon laser, diode laser, and LED.
The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
AT LASER 2017
Key clinical point:
Major finding: IPL and RF were among the modalities that were safer and more effective at skin rejuvenation than were older ones.
Data source: An analysis of more than 500 studies published since 1985 evaluating of 15 different types of technology used for facial rejuvenation, reviewing at least 35 clinical studies per platform.
Disclosures: The study was funded by the University of Wales Trinity Saint David. The authors had no disclosures.
Lasers may be effective for treating xanthelasma
SAN DIEGO – Laser treatments may be effective for xanthelasma palpebrarum lesions, based on a systematic review of existing studies, although the research is limited.
“The number of cases we looked at was relatively small, so you can’t come up with any definite conclusions,” said review coauthor Christopher J. Huerter, MD, head of the division of dermatology at Creighton University, Omaha. “But it’s promising since the lasers we examined all work with some efficacy, with the CO2 and Er:YAG [erbium:YAG] lasers probably having the best results.”
Xanthelasma lesions appear as small yellowish plaques on the eyelids. “About half the people who have it have some blood lipid abnormality,” Dr. Huerter said in an interview. “If a person has it, it’s worthwhile to do a cholesterol screen or a lipid profile.”
Treatment with trichloroacetic acid is one option, although it was more common before lasers began to be used. In addition, “surgical incision can be very effective,” he said, although the review notes that it can create undesirable scarring.
Researchers have studied laser treatment for xanthelasma for at least 30 years. Dr. Huerter and his colleagues examined 21 studies published since 1987, with the following lasers: CO2 laser (three studies), argon laser (one study), Er:YAG laser (four studies), ultrapulse CO2 laser (five studies), 1,450-nm diode laser (one study), pulsed dye laser (PDL, two studies), superpulsed or fractional CO2 laser (one study), and Q-switched neodymium:YAG laser (three studies). An additional study examined both argon and Er:YAG lasers.
The number of treated patients in the studies ranged from 1 to 50, and the number of treated lesions ranged from 1 to 76. (Patients often had more than one lesion.) “It would be nice to have bigger studies and bigger numbers,” Dr. Huerter said at the annual meeting of the American Society for Laser Medicine and Surgery.
Although the studies were limited by small cohorts, short follow-up, and lack of comparison groups, the findings did reveal signs of effectiveness: Clearance rates were 100% in CO2, argon, and PDL cases and about 85% with Er:YAG lasers. Clearance rates were lower with Nd:YAG (about 55%) and 1,450-nm diode (about 48%) lasers.
Edema was reported in all PDL cases and erythema in almost 20% of CO2 cases. Dyspigmentation was most common – at about 30% – in Er:YAG and 1,450-nm diode cases. Visible scars were reported in more than 5% of Er:YAG cases.
The review concluded that “sufficient evidence is available to suggest laser therapies to be a cosmetically excellent treatment option for xanthelasma , particularly applicable in patients who are not good candidates for surgical excision,” he said.
As for advice to dermatologists, Dr. Huerter pointed to the positive results for CO2 and Er:YAG lasers. He said PDL lasers could also be used. As for argon lasers, he noted that it’s not as likely for dermatologists to have them on hand, he said.
In regard to choosing which xanthelasma lesions to treat with laser, he said thicker ones may not be as amenable. “But if you do laser treatment and don’t get the results you want, you can always excise.”
No funding for the study was reported. Dr. Huerter reported no disclosures.
SAN DIEGO – Laser treatments may be effective for xanthelasma palpebrarum lesions, based on a systematic review of existing studies, although the research is limited.
“The number of cases we looked at was relatively small, so you can’t come up with any definite conclusions,” said review coauthor Christopher J. Huerter, MD, head of the division of dermatology at Creighton University, Omaha. “But it’s promising since the lasers we examined all work with some efficacy, with the CO2 and Er:YAG [erbium:YAG] lasers probably having the best results.”
Xanthelasma lesions appear as small yellowish plaques on the eyelids. “About half the people who have it have some blood lipid abnormality,” Dr. Huerter said in an interview. “If a person has it, it’s worthwhile to do a cholesterol screen or a lipid profile.”
Treatment with trichloroacetic acid is one option, although it was more common before lasers began to be used. In addition, “surgical incision can be very effective,” he said, although the review notes that it can create undesirable scarring.
Researchers have studied laser treatment for xanthelasma for at least 30 years. Dr. Huerter and his colleagues examined 21 studies published since 1987, with the following lasers: CO2 laser (three studies), argon laser (one study), Er:YAG laser (four studies), ultrapulse CO2 laser (five studies), 1,450-nm diode laser (one study), pulsed dye laser (PDL, two studies), superpulsed or fractional CO2 laser (one study), and Q-switched neodymium:YAG laser (three studies). An additional study examined both argon and Er:YAG lasers.
The number of treated patients in the studies ranged from 1 to 50, and the number of treated lesions ranged from 1 to 76. (Patients often had more than one lesion.) “It would be nice to have bigger studies and bigger numbers,” Dr. Huerter said at the annual meeting of the American Society for Laser Medicine and Surgery.
Although the studies were limited by small cohorts, short follow-up, and lack of comparison groups, the findings did reveal signs of effectiveness: Clearance rates were 100% in CO2, argon, and PDL cases and about 85% with Er:YAG lasers. Clearance rates were lower with Nd:YAG (about 55%) and 1,450-nm diode (about 48%) lasers.
Edema was reported in all PDL cases and erythema in almost 20% of CO2 cases. Dyspigmentation was most common – at about 30% – in Er:YAG and 1,450-nm diode cases. Visible scars were reported in more than 5% of Er:YAG cases.
The review concluded that “sufficient evidence is available to suggest laser therapies to be a cosmetically excellent treatment option for xanthelasma , particularly applicable in patients who are not good candidates for surgical excision,” he said.
As for advice to dermatologists, Dr. Huerter pointed to the positive results for CO2 and Er:YAG lasers. He said PDL lasers could also be used. As for argon lasers, he noted that it’s not as likely for dermatologists to have them on hand, he said.
In regard to choosing which xanthelasma lesions to treat with laser, he said thicker ones may not be as amenable. “But if you do laser treatment and don’t get the results you want, you can always excise.”
No funding for the study was reported. Dr. Huerter reported no disclosures.
SAN DIEGO – Laser treatments may be effective for xanthelasma palpebrarum lesions, based on a systematic review of existing studies, although the research is limited.
“The number of cases we looked at was relatively small, so you can’t come up with any definite conclusions,” said review coauthor Christopher J. Huerter, MD, head of the division of dermatology at Creighton University, Omaha. “But it’s promising since the lasers we examined all work with some efficacy, with the CO2 and Er:YAG [erbium:YAG] lasers probably having the best results.”
Xanthelasma lesions appear as small yellowish plaques on the eyelids. “About half the people who have it have some blood lipid abnormality,” Dr. Huerter said in an interview. “If a person has it, it’s worthwhile to do a cholesterol screen or a lipid profile.”
Treatment with trichloroacetic acid is one option, although it was more common before lasers began to be used. In addition, “surgical incision can be very effective,” he said, although the review notes that it can create undesirable scarring.
Researchers have studied laser treatment for xanthelasma for at least 30 years. Dr. Huerter and his colleagues examined 21 studies published since 1987, with the following lasers: CO2 laser (three studies), argon laser (one study), Er:YAG laser (four studies), ultrapulse CO2 laser (five studies), 1,450-nm diode laser (one study), pulsed dye laser (PDL, two studies), superpulsed or fractional CO2 laser (one study), and Q-switched neodymium:YAG laser (three studies). An additional study examined both argon and Er:YAG lasers.
The number of treated patients in the studies ranged from 1 to 50, and the number of treated lesions ranged from 1 to 76. (Patients often had more than one lesion.) “It would be nice to have bigger studies and bigger numbers,” Dr. Huerter said at the annual meeting of the American Society for Laser Medicine and Surgery.
Although the studies were limited by small cohorts, short follow-up, and lack of comparison groups, the findings did reveal signs of effectiveness: Clearance rates were 100% in CO2, argon, and PDL cases and about 85% with Er:YAG lasers. Clearance rates were lower with Nd:YAG (about 55%) and 1,450-nm diode (about 48%) lasers.
Edema was reported in all PDL cases and erythema in almost 20% of CO2 cases. Dyspigmentation was most common – at about 30% – in Er:YAG and 1,450-nm diode cases. Visible scars were reported in more than 5% of Er:YAG cases.
The review concluded that “sufficient evidence is available to suggest laser therapies to be a cosmetically excellent treatment option for xanthelasma , particularly applicable in patients who are not good candidates for surgical excision,” he said.
As for advice to dermatologists, Dr. Huerter pointed to the positive results for CO2 and Er:YAG lasers. He said PDL lasers could also be used. As for argon lasers, he noted that it’s not as likely for dermatologists to have them on hand, he said.
In regard to choosing which xanthelasma lesions to treat with laser, he said thicker ones may not be as amenable. “But if you do laser treatment and don’t get the results you want, you can always excise.”
No funding for the study was reported. Dr. Huerter reported no disclosures.
Key clinical point:
Major finding: Clearance rates of about 85%-100% were reported for xanthelasma treatment with CO2, argon, pulsed dye, and Er:YAG laser treatments.
Data source: A systematic review of 21 studies evaluating different laser treatments for xanthelasma.
Disclosures: No funding was reported. Dr. Huerter reported no disclosures.
Cosmetic Corner: Dermatologists Weigh in on Products for Sensitive Skin
To improve patient care and outcomes, leading dermatologists offered their recommendations on products for sensitive skin. Consideration must be given to:
- Avène Cicalfate Restorative Skin Cream
Pierre Fabre Dermo-Cosmetique USA
“Sucralfate for speeding up skin repair and the soothing thermal spring waters found in this product make it perfect postprocedure for immediately cooling and calming the skin.”—Jeannette Graf, MD, New York, New York
- Cetaphil RestoraDerm Eczema Calming Body Moisturizer
Galderma Laboratories, LP
“This product is formulated for atopic skin. I personally use it on my face as a moisturizer during the cold New York City winter.”—Anthony M. Rossi, MD, New York, New York
- Vanicream
Pharmaceutical Specialties, Inc
“I recommend Vanicream brand products to patients with sensitive skin or eczema. These products are fragrance free and have minimal ingredients.”— Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete's foot treatments, cleansing devices, redness-reducing products, and face scrubs will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on products for sensitive skin. Consideration must be given to:
- Avène Cicalfate Restorative Skin Cream
Pierre Fabre Dermo-Cosmetique USA
“Sucralfate for speeding up skin repair and the soothing thermal spring waters found in this product make it perfect postprocedure for immediately cooling and calming the skin.”—Jeannette Graf, MD, New York, New York
- Cetaphil RestoraDerm Eczema Calming Body Moisturizer
Galderma Laboratories, LP
“This product is formulated for atopic skin. I personally use it on my face as a moisturizer during the cold New York City winter.”—Anthony M. Rossi, MD, New York, New York
- Vanicream
Pharmaceutical Specialties, Inc
“I recommend Vanicream brand products to patients with sensitive skin or eczema. These products are fragrance free and have minimal ingredients.”— Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete's foot treatments, cleansing devices, redness-reducing products, and face scrubs will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on products for sensitive skin. Consideration must be given to:
- Avène Cicalfate Restorative Skin Cream
Pierre Fabre Dermo-Cosmetique USA
“Sucralfate for speeding up skin repair and the soothing thermal spring waters found in this product make it perfect postprocedure for immediately cooling and calming the skin.”—Jeannette Graf, MD, New York, New York
- Cetaphil RestoraDerm Eczema Calming Body Moisturizer
Galderma Laboratories, LP
“This product is formulated for atopic skin. I personally use it on my face as a moisturizer during the cold New York City winter.”—Anthony M. Rossi, MD, New York, New York
- Vanicream
Pharmaceutical Specialties, Inc
“I recommend Vanicream brand products to patients with sensitive skin or eczema. These products are fragrance free and have minimal ingredients.”— Gary Goldenberg, MD, New York, New York
Cutis invites readers to send us their recommendations. Athlete's foot treatments, cleansing devices, redness-reducing products, and face scrubs will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to the Editorial Office.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.