User login
COVID-19 in pregnant women and the impact on newborns
Clinical question: How does infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in pregnant mothers affect their newborns?
Background: A novel coronavirus, now named SARS-CoV-2 by the World Health Organization (previously referred to as 2019-nCoV), is currently causing a worldwide pandemic. It is believed to have originated in Hubei province, China, but is now rapidly spreading in other countries. Although its effects are most severe in the elderly, SARS-CoV-2 has been infecting younger patients, including pregnant women. The effect of COVID-19, the disease caused by SARS-CoV-2, in pregnant women on their newborn children, is unknown, as is the nature of perinatal transmission of SARS-CoV-2.
Study design: Retrospective analysis.
Setting: Five hospitals in Hubei province, China.
Synopsis: Researchers retrospectively analyzed the clinical features and outcomes of 10 neonates (including two twins) born to nine mothers with confirmed SARS-CoV-2 infection in five hospitals in Hubei province, China, during Jan. 20–Feb. 5, 2020. The mothers were, on average, 30 years of age, but their prior state of health was not described. SARS-CoV-2 infection was confirmed in eight mothers by SARS-CoV-2 nucleic acid testing (NAT). The twins’ mother was diagnosed with COVID-19 based on chest CT scan showing viral interstitial pneumonia with other causes of fever and lung infection being “excluded,” despite a negative SARS-CoV-2 NAT test.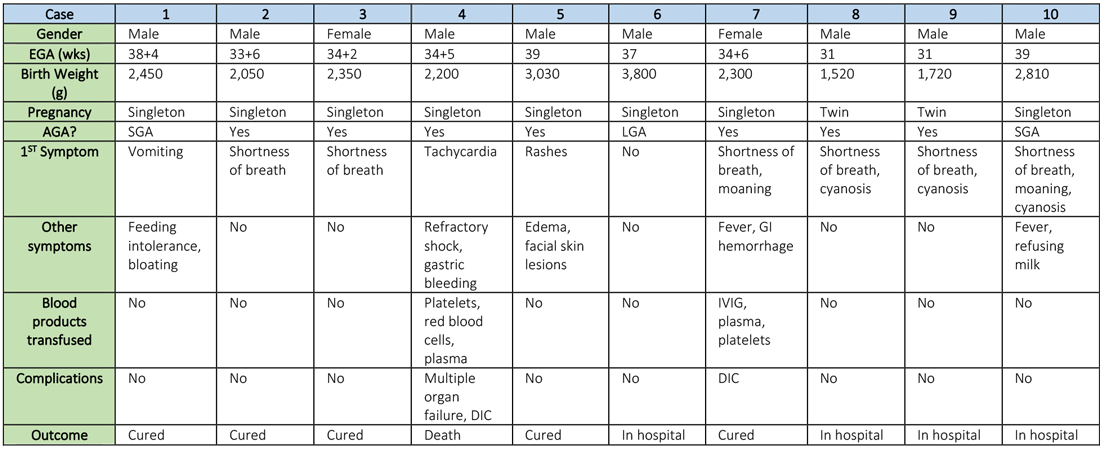
Symptoms occurred in the following:
- Before delivery in four mothers, three of whom were treated with oseltamivir (Tamiflu) after delivery.
- On the day of delivery in two mothers, one of whom was treated with oseltamivir and nebulized inhaled interferon after delivery.
- After delivery in three mothers.
Seven mothers delivered by cesarean section and two by vaginal delivery. Prenatal complications included intrauterine distress in six mothers, premature rupture of membranes in three (5-7 hours before onset of true labor), abnormal amniotic fluid in two, “abnormal” umbilical cord in two, and placenta previa in one.
The neonates born to these mothers included two females and eight males; four were full-term and six were premature (degree of prematurity not described). Symptoms first observed in these newborns included shortness of breath (six), fevers (two), tachycardia (one), and vomiting, feeding intolerance, “bloating,” refusing milk, and “gastric bleeding.” Chest radiographs were abnormal in seven newborns, including evidence of “infection” (four), neonatal respiratory distress syndrome (two), and pneumothorax (one). Two cases were described in detail:
- A neonate delivered at 34+5/7 weeks gestational age, was admitted due to shortness of breath and “moaning.” Eight days later, the neonate developed refractory shock, multiple organ failure, disseminated intravascular coagulation requiring transfusions of platelets, red blood cells, and plasma. He died on the ninth day.
- A neonate delivered at 34+6 weeks gestational age and was admitted 25 minutes after delivery due to shortness of breath and “moaning.” He required 2 days of noninvasive support/oxygen therapy and was observed to later develop “oxygen fluctuations” and thrombocytopenia at 3 days of life. The neonate was treated with “respiratory support,” intravenous immunoglobulin, transfusions of platelets and plasma, hydrocortisone (5 mg/kg per day for 6 days), low-dose heparin (2 units/kg per hr for 6 days), and low molecular weight heparin (2 units/kg per hr for 6 days). He was described to be “cured” 15 days later.
All nine neonates underwent pharyngeal swabs for SARS-CoV-2 NAT, and all were negative.
Bottom line: Although data are currently very limited, neonates born to mothers with COVID-19 appear to be at risk for adverse outcomes, including fetal distress, respiratory distress, thrombocytopenia associated with abnormal liver function, and death. There was no evidence of vertical transmission in this study.
Citation: Zhu H et al. Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Transl Pediatr. 2020 Feb;9(1):51-60.
Dr. Chang is chief of pediatric hospital medicine at Baystate Children’s Hospital in Springfield, Mass., and associate professor of pediatrics at the University of Massachusetts, also in Springfield.
Clinical question: How does infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in pregnant mothers affect their newborns?
Background: A novel coronavirus, now named SARS-CoV-2 by the World Health Organization (previously referred to as 2019-nCoV), is currently causing a worldwide pandemic. It is believed to have originated in Hubei province, China, but is now rapidly spreading in other countries. Although its effects are most severe in the elderly, SARS-CoV-2 has been infecting younger patients, including pregnant women. The effect of COVID-19, the disease caused by SARS-CoV-2, in pregnant women on their newborn children, is unknown, as is the nature of perinatal transmission of SARS-CoV-2.
Study design: Retrospective analysis.
Setting: Five hospitals in Hubei province, China.
Synopsis: Researchers retrospectively analyzed the clinical features and outcomes of 10 neonates (including two twins) born to nine mothers with confirmed SARS-CoV-2 infection in five hospitals in Hubei province, China, during Jan. 20–Feb. 5, 2020. The mothers were, on average, 30 years of age, but their prior state of health was not described. SARS-CoV-2 infection was confirmed in eight mothers by SARS-CoV-2 nucleic acid testing (NAT). The twins’ mother was diagnosed with COVID-19 based on chest CT scan showing viral interstitial pneumonia with other causes of fever and lung infection being “excluded,” despite a negative SARS-CoV-2 NAT test.
Symptoms occurred in the following:
- Before delivery in four mothers, three of whom were treated with oseltamivir (Tamiflu) after delivery.
- On the day of delivery in two mothers, one of whom was treated with oseltamivir and nebulized inhaled interferon after delivery.
- After delivery in three mothers.
Seven mothers delivered by cesarean section and two by vaginal delivery. Prenatal complications included intrauterine distress in six mothers, premature rupture of membranes in three (5-7 hours before onset of true labor), abnormal amniotic fluid in two, “abnormal” umbilical cord in two, and placenta previa in one.
The neonates born to these mothers included two females and eight males; four were full-term and six were premature (degree of prematurity not described). Symptoms first observed in these newborns included shortness of breath (six), fevers (two), tachycardia (one), and vomiting, feeding intolerance, “bloating,” refusing milk, and “gastric bleeding.” Chest radiographs were abnormal in seven newborns, including evidence of “infection” (four), neonatal respiratory distress syndrome (two), and pneumothorax (one). Two cases were described in detail:
- A neonate delivered at 34+5/7 weeks gestational age, was admitted due to shortness of breath and “moaning.” Eight days later, the neonate developed refractory shock, multiple organ failure, disseminated intravascular coagulation requiring transfusions of platelets, red blood cells, and plasma. He died on the ninth day.
- A neonate delivered at 34+6 weeks gestational age and was admitted 25 minutes after delivery due to shortness of breath and “moaning.” He required 2 days of noninvasive support/oxygen therapy and was observed to later develop “oxygen fluctuations” and thrombocytopenia at 3 days of life. The neonate was treated with “respiratory support,” intravenous immunoglobulin, transfusions of platelets and plasma, hydrocortisone (5 mg/kg per day for 6 days), low-dose heparin (2 units/kg per hr for 6 days), and low molecular weight heparin (2 units/kg per hr for 6 days). He was described to be “cured” 15 days later.
All nine neonates underwent pharyngeal swabs for SARS-CoV-2 NAT, and all were negative.
Bottom line: Although data are currently very limited, neonates born to mothers with COVID-19 appear to be at risk for adverse outcomes, including fetal distress, respiratory distress, thrombocytopenia associated with abnormal liver function, and death. There was no evidence of vertical transmission in this study.
Citation: Zhu H et al. Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Transl Pediatr. 2020 Feb;9(1):51-60.
Dr. Chang is chief of pediatric hospital medicine at Baystate Children’s Hospital in Springfield, Mass., and associate professor of pediatrics at the University of Massachusetts, also in Springfield.
Clinical question: How does infection with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) in pregnant mothers affect their newborns?
Background: A novel coronavirus, now named SARS-CoV-2 by the World Health Organization (previously referred to as 2019-nCoV), is currently causing a worldwide pandemic. It is believed to have originated in Hubei province, China, but is now rapidly spreading in other countries. Although its effects are most severe in the elderly, SARS-CoV-2 has been infecting younger patients, including pregnant women. The effect of COVID-19, the disease caused by SARS-CoV-2, in pregnant women on their newborn children, is unknown, as is the nature of perinatal transmission of SARS-CoV-2.
Study design: Retrospective analysis.
Setting: Five hospitals in Hubei province, China.
Synopsis: Researchers retrospectively analyzed the clinical features and outcomes of 10 neonates (including two twins) born to nine mothers with confirmed SARS-CoV-2 infection in five hospitals in Hubei province, China, during Jan. 20–Feb. 5, 2020. The mothers were, on average, 30 years of age, but their prior state of health was not described. SARS-CoV-2 infection was confirmed in eight mothers by SARS-CoV-2 nucleic acid testing (NAT). The twins’ mother was diagnosed with COVID-19 based on chest CT scan showing viral interstitial pneumonia with other causes of fever and lung infection being “excluded,” despite a negative SARS-CoV-2 NAT test.
Symptoms occurred in the following:
- Before delivery in four mothers, three of whom were treated with oseltamivir (Tamiflu) after delivery.
- On the day of delivery in two mothers, one of whom was treated with oseltamivir and nebulized inhaled interferon after delivery.
- After delivery in three mothers.
Seven mothers delivered by cesarean section and two by vaginal delivery. Prenatal complications included intrauterine distress in six mothers, premature rupture of membranes in three (5-7 hours before onset of true labor), abnormal amniotic fluid in two, “abnormal” umbilical cord in two, and placenta previa in one.
The neonates born to these mothers included two females and eight males; four were full-term and six were premature (degree of prematurity not described). Symptoms first observed in these newborns included shortness of breath (six), fevers (two), tachycardia (one), and vomiting, feeding intolerance, “bloating,” refusing milk, and “gastric bleeding.” Chest radiographs were abnormal in seven newborns, including evidence of “infection” (four), neonatal respiratory distress syndrome (two), and pneumothorax (one). Two cases were described in detail:
- A neonate delivered at 34+5/7 weeks gestational age, was admitted due to shortness of breath and “moaning.” Eight days later, the neonate developed refractory shock, multiple organ failure, disseminated intravascular coagulation requiring transfusions of platelets, red blood cells, and plasma. He died on the ninth day.
- A neonate delivered at 34+6 weeks gestational age and was admitted 25 minutes after delivery due to shortness of breath and “moaning.” He required 2 days of noninvasive support/oxygen therapy and was observed to later develop “oxygen fluctuations” and thrombocytopenia at 3 days of life. The neonate was treated with “respiratory support,” intravenous immunoglobulin, transfusions of platelets and plasma, hydrocortisone (5 mg/kg per day for 6 days), low-dose heparin (2 units/kg per hr for 6 days), and low molecular weight heparin (2 units/kg per hr for 6 days). He was described to be “cured” 15 days later.
All nine neonates underwent pharyngeal swabs for SARS-CoV-2 NAT, and all were negative.
Bottom line: Although data are currently very limited, neonates born to mothers with COVID-19 appear to be at risk for adverse outcomes, including fetal distress, respiratory distress, thrombocytopenia associated with abnormal liver function, and death. There was no evidence of vertical transmission in this study.
Citation: Zhu H et al. Clinical analysis of 10 neonates born to mothers with 2019-nCoV pneumonia. Transl Pediatr. 2020 Feb;9(1):51-60.
Dr. Chang is chief of pediatric hospital medicine at Baystate Children’s Hospital in Springfield, Mass., and associate professor of pediatrics at the University of Massachusetts, also in Springfield.
COVID-19: U.S. cardiology groups reaffirm continued use of RAAS-active drugs
Controversy continued over the potential effect of drugs that interfere with the renin-angiotensin-aldosterone system via the angiotensin-converting enzymes (ACE) may have on exacerbating infection with the SARS-CoV-2 virus that causes COVID-19.
A joint statement from the American Heart Association, American College of Cardiology, and the Heart Failure Society of America on March 17 gave full, unqualified support to maintaining patients on drugs that work this way, specifically the ACE inhibitors and angiotensin-receptor blockers (ARBs), which together form a long-standing cornerstone of treatment for hypertension, heart failure, and ischemic heart disease.
The three societies “recommend continuation” of ACE inhibitors or ARBs “for all patients already prescribed.” The statement went on to say that patients already diagnosed with a COVID-19 infection “should be fully evaluated before adding or removing any treatments, and any changes to their treatment should be based on the latest scientific evidence and shared decision making with their physician and health care team.”
“We understand the concern – as it has become clear that people with cardiovascular disease are at much higher risk of serious complications including death from COVID-19. However, we have reviewed the latest research – the evidence does not confirm the need to discontinue ACE inhibitors or ARBs, and we strongly recommend all physicians to consider the individual needs of each patient before making any changes to ACE-inhibitor or ARB treatment regimens,” said Robert A. Harrington, MD, president of the American Heart Association and professor and chair of medicine at Stanford (Calif.) University, in the statement.
“There are no experimental or clinical data demonstrating beneficial or adverse outcomes among COVID-19 patients using ACE-inhibitor or ARB medications,” added Richard J. Kovacs, MD, president of the American College of Cardiology and professor of cardiology at Indiana University in Indianapolis.
The “latest research” referred to in the statement likely focuses on a report that had appeared less than a week earlier in a British journal that hypothesized a possible increase in the susceptibility of human epithelial cells of the lungs, intestine, kidneys, and blood vessels exposed to these or certain other drugs, like the thiazolidinedione oral diabetes drugs or ibuprofen, because they cause up-regulation of the ACE2 protein in cell membranes, and ACE2 is the primary cell-surface receptor that allows the SARS-CoV-2 virus to enter.
“We therefore hypothesize that diabetes and hypertension treatment with ACE2-stimulating drugs increases the risk of developing severe and fatal COVID-19,” wrote Michael Roth, MD, and his associates in their recent article (Lancet Resp Med. 2020 Mar 11. doi: 10.1016/S2213-2600[20]30116-8). While the potential clinical impact of an increase in the number of ACE2 molecules in a cell’s surface membrane remains uninvestigated, the risk this phenomenon poses should mean that patients taking these drugs should receive heightened monitoring for COVID-19 disease, suggested Dr. Roth, a professor of biomedicine who specializes in studying inflammatory lung diseases including asthma, and associates.
However, others who have considered the impact that ACE inhibitors and ARBs might have on ACE2 and COVID-19 infections have noted that the picture is not simple. “Higher ACE2 expression following chronically medicating SARS‐CoV‐2 infected patients with AT1R [angiotensin receptor 1] blockers, while seemingly paradoxical, may protect them against acute lung injury rather than putting them at higher risk to develop SARS. This may be accounted for by two complementary mechanisms: blocking the excessive angiotensin‐mediated AT1R activation caused by the viral infection, as well as up-regulating ACE2, thereby reducing angiotensin production by ACE and increasing the production” of a vasodilating form of angiotensin, wrote David Gurwitz, PhD, in a recently published editorial (Drug Dev Res. 2020 Mar 4. doi: 10.1002/ddr.21656). A data-mining approach may allow researchers to determine whether patients who received drugs that interfere with angiotensin 1 function prior to being diagnosed with a COVID-19 infection had a better disease outcome, suggested Dr. Gurwitz, a molecular geneticist at Tel Aviv University in Jerusalem.
The statement from the three U.S. cardiology societies came a few days following a similar statement of support for ongoing use of ACE inhibitors and ARBs from the European Society of Cardiology’s Council on Hypertension.
Dr. Harrington, Dr. Kovacs, Dr. Roth, and Dr. Gurwitz had no relevant disclosures.
Controversy continued over the potential effect of drugs that interfere with the renin-angiotensin-aldosterone system via the angiotensin-converting enzymes (ACE) may have on exacerbating infection with the SARS-CoV-2 virus that causes COVID-19.
A joint statement from the American Heart Association, American College of Cardiology, and the Heart Failure Society of America on March 17 gave full, unqualified support to maintaining patients on drugs that work this way, specifically the ACE inhibitors and angiotensin-receptor blockers (ARBs), which together form a long-standing cornerstone of treatment for hypertension, heart failure, and ischemic heart disease.
The three societies “recommend continuation” of ACE inhibitors or ARBs “for all patients already prescribed.” The statement went on to say that patients already diagnosed with a COVID-19 infection “should be fully evaluated before adding or removing any treatments, and any changes to their treatment should be based on the latest scientific evidence and shared decision making with their physician and health care team.”
“We understand the concern – as it has become clear that people with cardiovascular disease are at much higher risk of serious complications including death from COVID-19. However, we have reviewed the latest research – the evidence does not confirm the need to discontinue ACE inhibitors or ARBs, and we strongly recommend all physicians to consider the individual needs of each patient before making any changes to ACE-inhibitor or ARB treatment regimens,” said Robert A. Harrington, MD, president of the American Heart Association and professor and chair of medicine at Stanford (Calif.) University, in the statement.
“There are no experimental or clinical data demonstrating beneficial or adverse outcomes among COVID-19 patients using ACE-inhibitor or ARB medications,” added Richard J. Kovacs, MD, president of the American College of Cardiology and professor of cardiology at Indiana University in Indianapolis.
The “latest research” referred to in the statement likely focuses on a report that had appeared less than a week earlier in a British journal that hypothesized a possible increase in the susceptibility of human epithelial cells of the lungs, intestine, kidneys, and blood vessels exposed to these or certain other drugs, like the thiazolidinedione oral diabetes drugs or ibuprofen, because they cause up-regulation of the ACE2 protein in cell membranes, and ACE2 is the primary cell-surface receptor that allows the SARS-CoV-2 virus to enter.
“We therefore hypothesize that diabetes and hypertension treatment with ACE2-stimulating drugs increases the risk of developing severe and fatal COVID-19,” wrote Michael Roth, MD, and his associates in their recent article (Lancet Resp Med. 2020 Mar 11. doi: 10.1016/S2213-2600[20]30116-8). While the potential clinical impact of an increase in the number of ACE2 molecules in a cell’s surface membrane remains uninvestigated, the risk this phenomenon poses should mean that patients taking these drugs should receive heightened monitoring for COVID-19 disease, suggested Dr. Roth, a professor of biomedicine who specializes in studying inflammatory lung diseases including asthma, and associates.
However, others who have considered the impact that ACE inhibitors and ARBs might have on ACE2 and COVID-19 infections have noted that the picture is not simple. “Higher ACE2 expression following chronically medicating SARS‐CoV‐2 infected patients with AT1R [angiotensin receptor 1] blockers, while seemingly paradoxical, may protect them against acute lung injury rather than putting them at higher risk to develop SARS. This may be accounted for by two complementary mechanisms: blocking the excessive angiotensin‐mediated AT1R activation caused by the viral infection, as well as up-regulating ACE2, thereby reducing angiotensin production by ACE and increasing the production” of a vasodilating form of angiotensin, wrote David Gurwitz, PhD, in a recently published editorial (Drug Dev Res. 2020 Mar 4. doi: 10.1002/ddr.21656). A data-mining approach may allow researchers to determine whether patients who received drugs that interfere with angiotensin 1 function prior to being diagnosed with a COVID-19 infection had a better disease outcome, suggested Dr. Gurwitz, a molecular geneticist at Tel Aviv University in Jerusalem.
The statement from the three U.S. cardiology societies came a few days following a similar statement of support for ongoing use of ACE inhibitors and ARBs from the European Society of Cardiology’s Council on Hypertension.
Dr. Harrington, Dr. Kovacs, Dr. Roth, and Dr. Gurwitz had no relevant disclosures.
Controversy continued over the potential effect of drugs that interfere with the renin-angiotensin-aldosterone system via the angiotensin-converting enzymes (ACE) may have on exacerbating infection with the SARS-CoV-2 virus that causes COVID-19.
A joint statement from the American Heart Association, American College of Cardiology, and the Heart Failure Society of America on March 17 gave full, unqualified support to maintaining patients on drugs that work this way, specifically the ACE inhibitors and angiotensin-receptor blockers (ARBs), which together form a long-standing cornerstone of treatment for hypertension, heart failure, and ischemic heart disease.
The three societies “recommend continuation” of ACE inhibitors or ARBs “for all patients already prescribed.” The statement went on to say that patients already diagnosed with a COVID-19 infection “should be fully evaluated before adding or removing any treatments, and any changes to their treatment should be based on the latest scientific evidence and shared decision making with their physician and health care team.”
“We understand the concern – as it has become clear that people with cardiovascular disease are at much higher risk of serious complications including death from COVID-19. However, we have reviewed the latest research – the evidence does not confirm the need to discontinue ACE inhibitors or ARBs, and we strongly recommend all physicians to consider the individual needs of each patient before making any changes to ACE-inhibitor or ARB treatment regimens,” said Robert A. Harrington, MD, president of the American Heart Association and professor and chair of medicine at Stanford (Calif.) University, in the statement.
“There are no experimental or clinical data demonstrating beneficial or adverse outcomes among COVID-19 patients using ACE-inhibitor or ARB medications,” added Richard J. Kovacs, MD, president of the American College of Cardiology and professor of cardiology at Indiana University in Indianapolis.
The “latest research” referred to in the statement likely focuses on a report that had appeared less than a week earlier in a British journal that hypothesized a possible increase in the susceptibility of human epithelial cells of the lungs, intestine, kidneys, and blood vessels exposed to these or certain other drugs, like the thiazolidinedione oral diabetes drugs or ibuprofen, because they cause up-regulation of the ACE2 protein in cell membranes, and ACE2 is the primary cell-surface receptor that allows the SARS-CoV-2 virus to enter.
“We therefore hypothesize that diabetes and hypertension treatment with ACE2-stimulating drugs increases the risk of developing severe and fatal COVID-19,” wrote Michael Roth, MD, and his associates in their recent article (Lancet Resp Med. 2020 Mar 11. doi: 10.1016/S2213-2600[20]30116-8). While the potential clinical impact of an increase in the number of ACE2 molecules in a cell’s surface membrane remains uninvestigated, the risk this phenomenon poses should mean that patients taking these drugs should receive heightened monitoring for COVID-19 disease, suggested Dr. Roth, a professor of biomedicine who specializes in studying inflammatory lung diseases including asthma, and associates.
However, others who have considered the impact that ACE inhibitors and ARBs might have on ACE2 and COVID-19 infections have noted that the picture is not simple. “Higher ACE2 expression following chronically medicating SARS‐CoV‐2 infected patients with AT1R [angiotensin receptor 1] blockers, while seemingly paradoxical, may protect them against acute lung injury rather than putting them at higher risk to develop SARS. This may be accounted for by two complementary mechanisms: blocking the excessive angiotensin‐mediated AT1R activation caused by the viral infection, as well as up-regulating ACE2, thereby reducing angiotensin production by ACE and increasing the production” of a vasodilating form of angiotensin, wrote David Gurwitz, PhD, in a recently published editorial (Drug Dev Res. 2020 Mar 4. doi: 10.1002/ddr.21656). A data-mining approach may allow researchers to determine whether patients who received drugs that interfere with angiotensin 1 function prior to being diagnosed with a COVID-19 infection had a better disease outcome, suggested Dr. Gurwitz, a molecular geneticist at Tel Aviv University in Jerusalem.
The statement from the three U.S. cardiology societies came a few days following a similar statement of support for ongoing use of ACE inhibitors and ARBs from the European Society of Cardiology’s Council on Hypertension.
Dr. Harrington, Dr. Kovacs, Dr. Roth, and Dr. Gurwitz had no relevant disclosures.
COVID-19 guidance for children’s health care providers
We are in uncharted waters with national and local states of emergency, schools and most activities being shut down, and rapidly evolving strategies on managing the COVID-19 outbreak. Everyone’s anxiety is appropriately high. As health care providers for children, you are facing changes in your personal life at home and in practice, likely including setting up televisits, trying to assess which patients to see, managing staffing challenges, and facing potential cash flow issues as expenses continue but revenue may fall short. And, of course, you will address a host of novel questions and concerns from the families you care for.
Your top priorities are to stay calm while offering clear recommendations on testing, quarantine, and treatment with guidance from our federal and local public health agencies. By providing clear guidance on the medical issues, you will offer substantial reassurance to families. But even with a medical plan in place, this remains a confusing and anxiety-provoking moment, one without much precedent in most people’s lives or in our national experience. Our aim is to complement that guidance by offering you some principles to help families manage the stress and anxiety that the disruptions and uncertainties that this public health emergency has created.
Offer clear, open, regular, and child-centered communication
If you have an email mailing list of your parents, you may want to summarize information you are gathering with a note they can expect at a specified time each day. You could request them to email you questions that then can be included as an FAQ (frequently asked questions).
Most children will have noticed people wearing face masks, or dramatic scenes on the news with hospital workers in full protective gear, breathlessly reporting growing numbers of the infected and the deceased. At a minimum, they are being commanded to wash hands and to not touch their faces (which is challenging enough for adults!), and are probably overhearing conversations about quarantines and contagion as well as family concerns about jobs and family finances. Many children are managing extended school closures and some are even managing the quarantine or serious illness of a loved one. When children overhear frightening news from distressed adults, they are going to become anxious and afraid themselves. Parents should remember to find out what their children have seen, heard, or understood about what is going on, and they should correct misinformation or misunderstandings with clear explanations. They also should find out what their children are curious about. “What has you wondering about that?” is a great response when children have questions, in order to make sure you get at any underlying worry.
It is fine to not have an answer to every question. It is difficult to offer clear explanations about something that we don’t yet fully understand, and it is fine to acknowledge what we don’t know. “That’s a great question. Let’s look together at the CDC [Centers for Disease Control and Prevention] website.” Offering to look for answers or information together can be a powerful way to model how to handle uncertainty. And always couch answers with appropriate (not false) reassurance: “Children and young adults appear to be very safe from this illness, but we want to take care to protect those that are older or already sick.”
Remember most children set their anxiety level based on their parent’s anxiety, and part of being child centered in your communication includes offering information in an age-appropriate manner. Preschool-aged children (up to 5 years) still have magical thinking. They are prone to finding masks and gowns scary and to assume that school stopping may be because they did something wrong. Tell them about the new illness, and about the doctors and officials working hard to keep people safe. Reassure them about all of the adults working hard together to understand the illness and take care of people who are sick. Their sense of time is less logical, so you may have to tell them more than once. Reassure them that children do not get very sick from this illness, but they can carry and spread it, like having paint on their hands, so they need to wash their hands often to take good care of other people.
School-age children (aged roughly 5-12 years) are better equipped cognitively to understand the seriousness of this outbreak. They are built to master new situations, but are prone to anxiety as they don’t yet have the emotional maturity to tolerate uncertainty or unfairness. Explain what is known without euphemisms, be truly curious about what their questions are, and look for answers together. Often what they need is to see you being calm in the face of uncertainty, bearing the strong feelings that may come, and preserving curiosity and compassion for others.
Adolescents also will need all of this support, and can be curious about more abstract implications (political, ethical, financial). Do not be surprised when they ask sophisticated questions, but still are focused on the personal disruptions or sacrifices (a canceled dance or sports meet, concerns about academic performance). Adolescence is a time of intense preoccupation with their emerging identity and relationships; it is normal for them to experience events in a way that may seem selfish, especially if it disrupts their time with friends. Remind parents to offer compassion and validation, while acknowledging that shared sacrifice and discomfort are a part of every individual’s experience when a society must respond to such a large challenge.
Be mindful of children’s vulnerabilities
Being child centered goes beyond thinking about their age and developmental stage. Parents are the experts on their children and will know about any particular vulnerabilities to the stresses of this serious outbreak. Children who are prone to anxiety or suffer from anxiety disorders may be more prone to silent worry. It is especially important to check in with them often, find out what they know and what they are worried about, and remind them to “never worry alone.” It also is important to continue with any recommended treatment, avoiding accommodation of their anxieties, except when it is required by public health protocols (i.e., staying home from school). Children with developmental disabilities may require additional support to change behaviors (hand washing) and may be more sensitive to changes in routine. And children with learning disabilities or special services in school may require additional support or structure during a prolonged period at home.
Preserve routines and structure
Routines and predictability are important to the sense of stability and well-being of most children (and adults). While disruptions are unavoidable, preserve what routines you can, and establish some new ones. For children who are out of school for several weeks, set up a consistent home routine, with a similar wake-up and bedtime, and a “school schedule.” There may be academic activities like reading or work sheets. If the parents’ work is disrupted, they can homeschool, shoring up weak academic areas or enhancing areas of interest. Be sure to preserve time for physical activity and social connections within this new framework. Social time does not require physical proximity, and can happen by screen or phone. Physical activity should be outside if at all possible. Predictability, preserved expectations (academic and otherwise), physical exercise, social connection, and consistent sleep will go a long way in protecting everyone’s ability to manage the disruptions of this epidemic.
Find opportunity in the disruption
Many families have been on a treadmill of work, school, and activities that have left little unscheduled time or spontaneity. Recommend looking at this disruption as a rare opportunity to slow down, spend time together, listen, learn more about one another, and even to have fun. Families could play board games, card games, watch movies together, or even read aloud. They might discover it is the time to try new hobbies (knitting, learning a new language or instrument), or to teach each other new skills. You might learn something new, or something new about your children. You also will offer a model of finding the opportunity in adversity, and even offer them some wonderful memories from a difficult time.
Take care of the vulnerable and ease others’ hardships
Without a doubt, this will be a difficult time for many people, medically, financially, and emotionally. One powerful strategy to build resilience in our children and strengthen our communities is to think with children about ways to help those who are most at risk or burdened by this challenge. Perhaps they want to make cards or FaceTime calls to older relatives who may be otherwise isolated. They may want to consider ways to support the work of first responders, even just with appreciation. They may want to reach out to elderly neighbors and offer to get groceries or other needed supplies for them. Balancing appropriate self-care with a focus on the needs of those who are more vulnerable or burdened than ourselves is a powerful way to show our children how communities pull together in a challenging time; enhance their feeling of connectedness; and build resilience in them, in our families, and in our communities.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected]
We are in uncharted waters with national and local states of emergency, schools and most activities being shut down, and rapidly evolving strategies on managing the COVID-19 outbreak. Everyone’s anxiety is appropriately high. As health care providers for children, you are facing changes in your personal life at home and in practice, likely including setting up televisits, trying to assess which patients to see, managing staffing challenges, and facing potential cash flow issues as expenses continue but revenue may fall short. And, of course, you will address a host of novel questions and concerns from the families you care for.
Your top priorities are to stay calm while offering clear recommendations on testing, quarantine, and treatment with guidance from our federal and local public health agencies. By providing clear guidance on the medical issues, you will offer substantial reassurance to families. But even with a medical plan in place, this remains a confusing and anxiety-provoking moment, one without much precedent in most people’s lives or in our national experience. Our aim is to complement that guidance by offering you some principles to help families manage the stress and anxiety that the disruptions and uncertainties that this public health emergency has created.
Offer clear, open, regular, and child-centered communication
If you have an email mailing list of your parents, you may want to summarize information you are gathering with a note they can expect at a specified time each day. You could request them to email you questions that then can be included as an FAQ (frequently asked questions).
Most children will have noticed people wearing face masks, or dramatic scenes on the news with hospital workers in full protective gear, breathlessly reporting growing numbers of the infected and the deceased. At a minimum, they are being commanded to wash hands and to not touch their faces (which is challenging enough for adults!), and are probably overhearing conversations about quarantines and contagion as well as family concerns about jobs and family finances. Many children are managing extended school closures and some are even managing the quarantine or serious illness of a loved one. When children overhear frightening news from distressed adults, they are going to become anxious and afraid themselves. Parents should remember to find out what their children have seen, heard, or understood about what is going on, and they should correct misinformation or misunderstandings with clear explanations. They also should find out what their children are curious about. “What has you wondering about that?” is a great response when children have questions, in order to make sure you get at any underlying worry.
It is fine to not have an answer to every question. It is difficult to offer clear explanations about something that we don’t yet fully understand, and it is fine to acknowledge what we don’t know. “That’s a great question. Let’s look together at the CDC [Centers for Disease Control and Prevention] website.” Offering to look for answers or information together can be a powerful way to model how to handle uncertainty. And always couch answers with appropriate (not false) reassurance: “Children and young adults appear to be very safe from this illness, but we want to take care to protect those that are older or already sick.”
Remember most children set their anxiety level based on their parent’s anxiety, and part of being child centered in your communication includes offering information in an age-appropriate manner. Preschool-aged children (up to 5 years) still have magical thinking. They are prone to finding masks and gowns scary and to assume that school stopping may be because they did something wrong. Tell them about the new illness, and about the doctors and officials working hard to keep people safe. Reassure them about all of the adults working hard together to understand the illness and take care of people who are sick. Their sense of time is less logical, so you may have to tell them more than once. Reassure them that children do not get very sick from this illness, but they can carry and spread it, like having paint on their hands, so they need to wash their hands often to take good care of other people.
School-age children (aged roughly 5-12 years) are better equipped cognitively to understand the seriousness of this outbreak. They are built to master new situations, but are prone to anxiety as they don’t yet have the emotional maturity to tolerate uncertainty or unfairness. Explain what is known without euphemisms, be truly curious about what their questions are, and look for answers together. Often what they need is to see you being calm in the face of uncertainty, bearing the strong feelings that may come, and preserving curiosity and compassion for others.
Adolescents also will need all of this support, and can be curious about more abstract implications (political, ethical, financial). Do not be surprised when they ask sophisticated questions, but still are focused on the personal disruptions or sacrifices (a canceled dance or sports meet, concerns about academic performance). Adolescence is a time of intense preoccupation with their emerging identity and relationships; it is normal for them to experience events in a way that may seem selfish, especially if it disrupts their time with friends. Remind parents to offer compassion and validation, while acknowledging that shared sacrifice and discomfort are a part of every individual’s experience when a society must respond to such a large challenge.
Be mindful of children’s vulnerabilities
Being child centered goes beyond thinking about their age and developmental stage. Parents are the experts on their children and will know about any particular vulnerabilities to the stresses of this serious outbreak. Children who are prone to anxiety or suffer from anxiety disorders may be more prone to silent worry. It is especially important to check in with them often, find out what they know and what they are worried about, and remind them to “never worry alone.” It also is important to continue with any recommended treatment, avoiding accommodation of their anxieties, except when it is required by public health protocols (i.e., staying home from school). Children with developmental disabilities may require additional support to change behaviors (hand washing) and may be more sensitive to changes in routine. And children with learning disabilities or special services in school may require additional support or structure during a prolonged period at home.
Preserve routines and structure
Routines and predictability are important to the sense of stability and well-being of most children (and adults). While disruptions are unavoidable, preserve what routines you can, and establish some new ones. For children who are out of school for several weeks, set up a consistent home routine, with a similar wake-up and bedtime, and a “school schedule.” There may be academic activities like reading or work sheets. If the parents’ work is disrupted, they can homeschool, shoring up weak academic areas or enhancing areas of interest. Be sure to preserve time for physical activity and social connections within this new framework. Social time does not require physical proximity, and can happen by screen or phone. Physical activity should be outside if at all possible. Predictability, preserved expectations (academic and otherwise), physical exercise, social connection, and consistent sleep will go a long way in protecting everyone’s ability to manage the disruptions of this epidemic.
Find opportunity in the disruption
Many families have been on a treadmill of work, school, and activities that have left little unscheduled time or spontaneity. Recommend looking at this disruption as a rare opportunity to slow down, spend time together, listen, learn more about one another, and even to have fun. Families could play board games, card games, watch movies together, or even read aloud. They might discover it is the time to try new hobbies (knitting, learning a new language or instrument), or to teach each other new skills. You might learn something new, or something new about your children. You also will offer a model of finding the opportunity in adversity, and even offer them some wonderful memories from a difficult time.
Take care of the vulnerable and ease others’ hardships
Without a doubt, this will be a difficult time for many people, medically, financially, and emotionally. One powerful strategy to build resilience in our children and strengthen our communities is to think with children about ways to help those who are most at risk or burdened by this challenge. Perhaps they want to make cards or FaceTime calls to older relatives who may be otherwise isolated. They may want to consider ways to support the work of first responders, even just with appreciation. They may want to reach out to elderly neighbors and offer to get groceries or other needed supplies for them. Balancing appropriate self-care with a focus on the needs of those who are more vulnerable or burdened than ourselves is a powerful way to show our children how communities pull together in a challenging time; enhance their feeling of connectedness; and build resilience in them, in our families, and in our communities.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected]
We are in uncharted waters with national and local states of emergency, schools and most activities being shut down, and rapidly evolving strategies on managing the COVID-19 outbreak. Everyone’s anxiety is appropriately high. As health care providers for children, you are facing changes in your personal life at home and in practice, likely including setting up televisits, trying to assess which patients to see, managing staffing challenges, and facing potential cash flow issues as expenses continue but revenue may fall short. And, of course, you will address a host of novel questions and concerns from the families you care for.
Your top priorities are to stay calm while offering clear recommendations on testing, quarantine, and treatment with guidance from our federal and local public health agencies. By providing clear guidance on the medical issues, you will offer substantial reassurance to families. But even with a medical plan in place, this remains a confusing and anxiety-provoking moment, one without much precedent in most people’s lives or in our national experience. Our aim is to complement that guidance by offering you some principles to help families manage the stress and anxiety that the disruptions and uncertainties that this public health emergency has created.
Offer clear, open, regular, and child-centered communication
If you have an email mailing list of your parents, you may want to summarize information you are gathering with a note they can expect at a specified time each day. You could request them to email you questions that then can be included as an FAQ (frequently asked questions).
Most children will have noticed people wearing face masks, or dramatic scenes on the news with hospital workers in full protective gear, breathlessly reporting growing numbers of the infected and the deceased. At a minimum, they are being commanded to wash hands and to not touch their faces (which is challenging enough for adults!), and are probably overhearing conversations about quarantines and contagion as well as family concerns about jobs and family finances. Many children are managing extended school closures and some are even managing the quarantine or serious illness of a loved one. When children overhear frightening news from distressed adults, they are going to become anxious and afraid themselves. Parents should remember to find out what their children have seen, heard, or understood about what is going on, and they should correct misinformation or misunderstandings with clear explanations. They also should find out what their children are curious about. “What has you wondering about that?” is a great response when children have questions, in order to make sure you get at any underlying worry.
It is fine to not have an answer to every question. It is difficult to offer clear explanations about something that we don’t yet fully understand, and it is fine to acknowledge what we don’t know. “That’s a great question. Let’s look together at the CDC [Centers for Disease Control and Prevention] website.” Offering to look for answers or information together can be a powerful way to model how to handle uncertainty. And always couch answers with appropriate (not false) reassurance: “Children and young adults appear to be very safe from this illness, but we want to take care to protect those that are older or already sick.”
Remember most children set their anxiety level based on their parent’s anxiety, and part of being child centered in your communication includes offering information in an age-appropriate manner. Preschool-aged children (up to 5 years) still have magical thinking. They are prone to finding masks and gowns scary and to assume that school stopping may be because they did something wrong. Tell them about the new illness, and about the doctors and officials working hard to keep people safe. Reassure them about all of the adults working hard together to understand the illness and take care of people who are sick. Their sense of time is less logical, so you may have to tell them more than once. Reassure them that children do not get very sick from this illness, but they can carry and spread it, like having paint on their hands, so they need to wash their hands often to take good care of other people.
School-age children (aged roughly 5-12 years) are better equipped cognitively to understand the seriousness of this outbreak. They are built to master new situations, but are prone to anxiety as they don’t yet have the emotional maturity to tolerate uncertainty or unfairness. Explain what is known without euphemisms, be truly curious about what their questions are, and look for answers together. Often what they need is to see you being calm in the face of uncertainty, bearing the strong feelings that may come, and preserving curiosity and compassion for others.
Adolescents also will need all of this support, and can be curious about more abstract implications (political, ethical, financial). Do not be surprised when they ask sophisticated questions, but still are focused on the personal disruptions or sacrifices (a canceled dance or sports meet, concerns about academic performance). Adolescence is a time of intense preoccupation with their emerging identity and relationships; it is normal for them to experience events in a way that may seem selfish, especially if it disrupts their time with friends. Remind parents to offer compassion and validation, while acknowledging that shared sacrifice and discomfort are a part of every individual’s experience when a society must respond to such a large challenge.
Be mindful of children’s vulnerabilities
Being child centered goes beyond thinking about their age and developmental stage. Parents are the experts on their children and will know about any particular vulnerabilities to the stresses of this serious outbreak. Children who are prone to anxiety or suffer from anxiety disorders may be more prone to silent worry. It is especially important to check in with them often, find out what they know and what they are worried about, and remind them to “never worry alone.” It also is important to continue with any recommended treatment, avoiding accommodation of their anxieties, except when it is required by public health protocols (i.e., staying home from school). Children with developmental disabilities may require additional support to change behaviors (hand washing) and may be more sensitive to changes in routine. And children with learning disabilities or special services in school may require additional support or structure during a prolonged period at home.
Preserve routines and structure
Routines and predictability are important to the sense of stability and well-being of most children (and adults). While disruptions are unavoidable, preserve what routines you can, and establish some new ones. For children who are out of school for several weeks, set up a consistent home routine, with a similar wake-up and bedtime, and a “school schedule.” There may be academic activities like reading or work sheets. If the parents’ work is disrupted, they can homeschool, shoring up weak academic areas or enhancing areas of interest. Be sure to preserve time for physical activity and social connections within this new framework. Social time does not require physical proximity, and can happen by screen or phone. Physical activity should be outside if at all possible. Predictability, preserved expectations (academic and otherwise), physical exercise, social connection, and consistent sleep will go a long way in protecting everyone’s ability to manage the disruptions of this epidemic.
Find opportunity in the disruption
Many families have been on a treadmill of work, school, and activities that have left little unscheduled time or spontaneity. Recommend looking at this disruption as a rare opportunity to slow down, spend time together, listen, learn more about one another, and even to have fun. Families could play board games, card games, watch movies together, or even read aloud. They might discover it is the time to try new hobbies (knitting, learning a new language or instrument), or to teach each other new skills. You might learn something new, or something new about your children. You also will offer a model of finding the opportunity in adversity, and even offer them some wonderful memories from a difficult time.
Take care of the vulnerable and ease others’ hardships
Without a doubt, this will be a difficult time for many people, medically, financially, and emotionally. One powerful strategy to build resilience in our children and strengthen our communities is to think with children about ways to help those who are most at risk or burdened by this challenge. Perhaps they want to make cards or FaceTime calls to older relatives who may be otherwise isolated. They may want to consider ways to support the work of first responders, even just with appreciation. They may want to reach out to elderly neighbors and offer to get groceries or other needed supplies for them. Balancing appropriate self-care with a focus on the needs of those who are more vulnerable or burdened than ourselves is a powerful way to show our children how communities pull together in a challenging time; enhance their feeling of connectedness; and build resilience in them, in our families, and in our communities.
Dr. Swick is physician in chief at Ohana, Center for Child and Adolescent Behavioral Health, Community Hospital of the Monterey (Calif.) Peninsula. Dr. Jellinek is professor emeritus of psychiatry and pediatrics, Harvard Medical School, Boston. Email them at [email protected]
FDA advises stopping SGLT2 inhibitor treatment prior to surgery
The new changes affect canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin, and were made because surgery may put patients being treated with SGLT2 inhibitors at a higher risk of ketoacidosis. Canagliflozin, dapagliflozin, and empagliflozin should be discontinued 3 days before scheduled surgery, and ertugliflozin should be stopped at least 4 days before, the agency noted in a press release. Blood glucose should be monitored after drug discontinuation and appropriately managed before surgery.
“The SGLT2 inhibitor may be restarted once the patient’s oral intake is back to baseline and any other risk factors for ketoacidosis are resolved,” the agency added.
SGLT2 inhibitors lower blood sugar by causing the kidney to remove sugar from the body through urine. Side effects for the drugs vary, but include urinary tract infections and genital mycotic infection. Patients with severe renal impairment or end-stage renal disease, who are on dialysis treatment, or who have a known hypersensitivity to the medication should not take SGLT2 inhibitors, the FDA said.
The new changes affect canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin, and were made because surgery may put patients being treated with SGLT2 inhibitors at a higher risk of ketoacidosis. Canagliflozin, dapagliflozin, and empagliflozin should be discontinued 3 days before scheduled surgery, and ertugliflozin should be stopped at least 4 days before, the agency noted in a press release. Blood glucose should be monitored after drug discontinuation and appropriately managed before surgery.
“The SGLT2 inhibitor may be restarted once the patient’s oral intake is back to baseline and any other risk factors for ketoacidosis are resolved,” the agency added.
SGLT2 inhibitors lower blood sugar by causing the kidney to remove sugar from the body through urine. Side effects for the drugs vary, but include urinary tract infections and genital mycotic infection. Patients with severe renal impairment or end-stage renal disease, who are on dialysis treatment, or who have a known hypersensitivity to the medication should not take SGLT2 inhibitors, the FDA said.
The new changes affect canagliflozin, dapagliflozin, empagliflozin, and ertugliflozin, and were made because surgery may put patients being treated with SGLT2 inhibitors at a higher risk of ketoacidosis. Canagliflozin, dapagliflozin, and empagliflozin should be discontinued 3 days before scheduled surgery, and ertugliflozin should be stopped at least 4 days before, the agency noted in a press release. Blood glucose should be monitored after drug discontinuation and appropriately managed before surgery.
“The SGLT2 inhibitor may be restarted once the patient’s oral intake is back to baseline and any other risk factors for ketoacidosis are resolved,” the agency added.
SGLT2 inhibitors lower blood sugar by causing the kidney to remove sugar from the body through urine. Side effects for the drugs vary, but include urinary tract infections and genital mycotic infection. Patients with severe renal impairment or end-stage renal disease, who are on dialysis treatment, or who have a known hypersensitivity to the medication should not take SGLT2 inhibitors, the FDA said.
PARAGON-HF: Optimal systolic pressure in HFpEF is 120-129 mm Hg
A target systolic blood pressure (SBP) of 120-129 mm Hg in patients with heart failure with preserved ejection fraction proved to be the sweet spot with the lowest rates of major adverse cardiovascular and renal events in a new analysis from the landmark PARAGON-HF trial.
This finding from the largest-ever randomized, controlled study in heart failure with preserved ejection fraction (HFpEF) strengthens support for current U.S. joint hypertension guidelines, which call for a target SBP less than 130 mm Hg in patients with HFpEF (J Am Coll Cardiol. 2017 Aug 8;70[6]:776-803), a recommendation based upon weak evidence until now. That’s because the SPRINT trial, the major impetus for adoption of intensive blood pressure control in the current guidelines, excluded patients with symptomatic HF, Scott D. Solomon, MD, and coinvestigators noted in their new analysis. The study was published in the Journal of the American College of Cardiology and had been planned for presentation during the joint scientific sessions of the American College of Cardiology and the World Heart Federation. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
The new analysis from PARAGON-HF (Prospective Comparison of ARNI with ARB Global Outcomes in HFpEF) also ruled out the SBP-lowering effect of sacubitril/valsartan (Entresto) as the explanation for the combination drug’s demonstrated beneficial impact on outcomes in the subgroup with an SBP of 120-129 mm Hg. That wasn’t actually a surprise. Indeed, the new study had two hypotheses: one, that the relationship between SBP and cardiovascular and renal outcomes in HFpEF would follow a J-shaped curve, and two, that sacubitril/valsartan’s blood pressure–lowering effect would not account for the drug’s outcome benefits in the subset of HFpEF patients with an SBP in the sweet spot of 120-129 mm Hg. Both hypotheses were borne out, noted Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, both in Boston.
“These data strongly support that additional mechanisms other than blood pressure–lowering account for the benefit. But this is not surprising. The same can be said for most of the therapies that work in heart failure,” he said in an interview.
Take, for example, spironolactone. In TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist), another major trial in which Dr. Solomon played a leadership role, the beneficial effect of spironolactone on clinical outcomes also proved unrelated to the drug’s blood pressure–lowering effect.
Other known effects of sacubitril/valsartan, a novel angiotensin receptor–neprilysin inhibitor, or ARNI, might in theory account for the observed clinical benefits in ARNI-treated patients with an on-treatment SBP of 120-129 mm Hg in PARAGON-HF. These include improved left atrial remodeling, an increase in natriuretic peptides, and improved myocardial relaxation. However, the current lack of understanding of the basic mechanistic processes underlying the varied clinical expressions of HFpEF is a major factor contributing to the lack of any proven-effective therapy for this extremely common and costly disorder, according to Dr. Solomon and coinvestigators.
In contrast to HFpEF, for which to date there is no proven treatment, heart failure with reduced ejection fraction sacubitril/valsartan has a class I recommendation on the strength of its performance in significantly reducing cardiovascular deaths and heart failure hospitalizations in the PARADIGM-HF trial (N Engl J Med. 2014 Sep 11;371:993-1004).
PARAGON-HF included 4,822 patients with symptomatic HFpEF who were randomized to sacubitril/valsartan at 97/103 mg b.i.d. or valsartan at 160 mg b.i.d. As previously reported (N Engl J Med. 2019 Oct 24;381[17]:1609-20), at an average follow-up of 35 months, the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – occurred at a rate of 12.8 events per 100 patient-years in the sacubitril/valsartan group and 14.6 per 100 patient-years in the valsartan arm, for a 13% relative risk reduction that narrowly missed statistical significance (P = .059).
However, sacubitril/valsartan showed significant benefit on some prespecified secondary endpoints, including worsening renal function, change in New York Heart Association class, and quality of life. Women, who notably accounted for 52% of study participants, appeared to benefit from sacubitril/valsartan more than men as evidenced by their 27% relative risk reduction in the primary endpoint. Also, in the roughly half of PARAGON-HF participants with a baseline left ventricular ejection fraction of 45%-57%, treatment with sacubitril/valsartan resulted in a statistically significant 22% relative risk reduction in the primary endpoint, compared with valsartan alone.
SBP and cardiovascular outcomes in HFpEF
In the new analysis, Dr. Solomon and coworkers examined outcomes based on baseline and mean achieved SBP quartiles regardless of treatment arm. In an unadjusted analysis, the primary composite endpoint occurred at a rate of 15.2 events/100 patient-years in HFpEF patients with an achieved SBP below 120 mm Hg, 11.4/100 patient-years at 120-129 mm Hg, 12.2/100 patient-years at 130-139 mm Hg, and 15.6/100 patient-years at 140 mm Hg or more. Further, in a multivariate regression analysis extensively adjusted for atrial fibrillation, sex, race, and numerous other potential confounders, the group with an achieved SBP of 120-129 mm Hg continued to fare best. The adjusted risks for the primary endpoint were 11% and 21% higher in patients in the first and third quartiles of achieved SBP, compared with those at 120-129 mm Hg, although neither trend reached statistical significance. But patients in the top quartile, with an achieved SBP of 140 mm Hg or more, had a highly significant 56% increase in risk, compared with patients in the second-lowest SBP quartile.
Change in blood pressure from baseline to week 48 had no impact on quality of life or high-sensitivity troponin T. However, each 10–mm Hg lowering of SBP was associated with a modest 2.1% reduction in log-transformed N-terminal of the prohormone brain natriuretic peptide.
Sacubitril/valsartan reduced SBP by an average of 5.2 mm Hg more than valsartan alone at 4 weeks regardless of baseline SBP. And the combo drug had a significantly greater SBP-lowering effect in women than men, by a margin of 6.3 mm Hg versus 4.0 mm Hg. But a Cox regression analysis showed that in women, as in the study population as a whole, sacubitril/valsartan’s SBP-lowering effects didn’t account for the drug’s impact on outcomes.
In an editorial accompanying publication of the new PARAGON-HF blood pressure analysis (J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.024), Hector O. Ventura, MD, and colleagues at the Ochsner Clinic in New Orleans observed that the study results “lend some credence to the prognostic relationship of blood pressure in HFpEF, but whether they should serve as a therapeutic target or are merely a prognostic surrogate determined by other pathogenic factors, such as vascular ventricular uncoupling or aortic stiffness on one hand when blood pressure is greater than 140 mm Hg, or a reduced cardiac performance indicated by reduced blood pressure to less than 120 mm Hg, remains uncertain.”
“What is certain, however, is that the relationship and contributions of hypertension in manifest HFpEF are complex, multifactorial and likely go well beyond a simplistic framework of hemodynamic influences,” they added.
Dr. Solomon has received research grants from and serves as a consultant to Novartis, which funded PARAGON-HF, and has similar financial relationships with more than a dozen other pharmaceutical companies. Dr. Ventura reported having no relevant financial interests.
SOURCE: Solomon SD et al. J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.009.
A target systolic blood pressure (SBP) of 120-129 mm Hg in patients with heart failure with preserved ejection fraction proved to be the sweet spot with the lowest rates of major adverse cardiovascular and renal events in a new analysis from the landmark PARAGON-HF trial.
This finding from the largest-ever randomized, controlled study in heart failure with preserved ejection fraction (HFpEF) strengthens support for current U.S. joint hypertension guidelines, which call for a target SBP less than 130 mm Hg in patients with HFpEF (J Am Coll Cardiol. 2017 Aug 8;70[6]:776-803), a recommendation based upon weak evidence until now. That’s because the SPRINT trial, the major impetus for adoption of intensive blood pressure control in the current guidelines, excluded patients with symptomatic HF, Scott D. Solomon, MD, and coinvestigators noted in their new analysis. The study was published in the Journal of the American College of Cardiology and had been planned for presentation during the joint scientific sessions of the American College of Cardiology and the World Heart Federation. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
The new analysis from PARAGON-HF (Prospective Comparison of ARNI with ARB Global Outcomes in HFpEF) also ruled out the SBP-lowering effect of sacubitril/valsartan (Entresto) as the explanation for the combination drug’s demonstrated beneficial impact on outcomes in the subgroup with an SBP of 120-129 mm Hg. That wasn’t actually a surprise. Indeed, the new study had two hypotheses: one, that the relationship between SBP and cardiovascular and renal outcomes in HFpEF would follow a J-shaped curve, and two, that sacubitril/valsartan’s blood pressure–lowering effect would not account for the drug’s outcome benefits in the subset of HFpEF patients with an SBP in the sweet spot of 120-129 mm Hg. Both hypotheses were borne out, noted Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, both in Boston.
“These data strongly support that additional mechanisms other than blood pressure–lowering account for the benefit. But this is not surprising. The same can be said for most of the therapies that work in heart failure,” he said in an interview.
Take, for example, spironolactone. In TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist), another major trial in which Dr. Solomon played a leadership role, the beneficial effect of spironolactone on clinical outcomes also proved unrelated to the drug’s blood pressure–lowering effect.
Other known effects of sacubitril/valsartan, a novel angiotensin receptor–neprilysin inhibitor, or ARNI, might in theory account for the observed clinical benefits in ARNI-treated patients with an on-treatment SBP of 120-129 mm Hg in PARAGON-HF. These include improved left atrial remodeling, an increase in natriuretic peptides, and improved myocardial relaxation. However, the current lack of understanding of the basic mechanistic processes underlying the varied clinical expressions of HFpEF is a major factor contributing to the lack of any proven-effective therapy for this extremely common and costly disorder, according to Dr. Solomon and coinvestigators.
In contrast to HFpEF, for which to date there is no proven treatment, heart failure with reduced ejection fraction sacubitril/valsartan has a class I recommendation on the strength of its performance in significantly reducing cardiovascular deaths and heart failure hospitalizations in the PARADIGM-HF trial (N Engl J Med. 2014 Sep 11;371:993-1004).
PARAGON-HF included 4,822 patients with symptomatic HFpEF who were randomized to sacubitril/valsartan at 97/103 mg b.i.d. or valsartan at 160 mg b.i.d. As previously reported (N Engl J Med. 2019 Oct 24;381[17]:1609-20), at an average follow-up of 35 months, the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – occurred at a rate of 12.8 events per 100 patient-years in the sacubitril/valsartan group and 14.6 per 100 patient-years in the valsartan arm, for a 13% relative risk reduction that narrowly missed statistical significance (P = .059).
However, sacubitril/valsartan showed significant benefit on some prespecified secondary endpoints, including worsening renal function, change in New York Heart Association class, and quality of life. Women, who notably accounted for 52% of study participants, appeared to benefit from sacubitril/valsartan more than men as evidenced by their 27% relative risk reduction in the primary endpoint. Also, in the roughly half of PARAGON-HF participants with a baseline left ventricular ejection fraction of 45%-57%, treatment with sacubitril/valsartan resulted in a statistically significant 22% relative risk reduction in the primary endpoint, compared with valsartan alone.
SBP and cardiovascular outcomes in HFpEF
In the new analysis, Dr. Solomon and coworkers examined outcomes based on baseline and mean achieved SBP quartiles regardless of treatment arm. In an unadjusted analysis, the primary composite endpoint occurred at a rate of 15.2 events/100 patient-years in HFpEF patients with an achieved SBP below 120 mm Hg, 11.4/100 patient-years at 120-129 mm Hg, 12.2/100 patient-years at 130-139 mm Hg, and 15.6/100 patient-years at 140 mm Hg or more. Further, in a multivariate regression analysis extensively adjusted for atrial fibrillation, sex, race, and numerous other potential confounders, the group with an achieved SBP of 120-129 mm Hg continued to fare best. The adjusted risks for the primary endpoint were 11% and 21% higher in patients in the first and third quartiles of achieved SBP, compared with those at 120-129 mm Hg, although neither trend reached statistical significance. But patients in the top quartile, with an achieved SBP of 140 mm Hg or more, had a highly significant 56% increase in risk, compared with patients in the second-lowest SBP quartile.
Change in blood pressure from baseline to week 48 had no impact on quality of life or high-sensitivity troponin T. However, each 10–mm Hg lowering of SBP was associated with a modest 2.1% reduction in log-transformed N-terminal of the prohormone brain natriuretic peptide.
Sacubitril/valsartan reduced SBP by an average of 5.2 mm Hg more than valsartan alone at 4 weeks regardless of baseline SBP. And the combo drug had a significantly greater SBP-lowering effect in women than men, by a margin of 6.3 mm Hg versus 4.0 mm Hg. But a Cox regression analysis showed that in women, as in the study population as a whole, sacubitril/valsartan’s SBP-lowering effects didn’t account for the drug’s impact on outcomes.
In an editorial accompanying publication of the new PARAGON-HF blood pressure analysis (J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.024), Hector O. Ventura, MD, and colleagues at the Ochsner Clinic in New Orleans observed that the study results “lend some credence to the prognostic relationship of blood pressure in HFpEF, but whether they should serve as a therapeutic target or are merely a prognostic surrogate determined by other pathogenic factors, such as vascular ventricular uncoupling or aortic stiffness on one hand when blood pressure is greater than 140 mm Hg, or a reduced cardiac performance indicated by reduced blood pressure to less than 120 mm Hg, remains uncertain.”
“What is certain, however, is that the relationship and contributions of hypertension in manifest HFpEF are complex, multifactorial and likely go well beyond a simplistic framework of hemodynamic influences,” they added.
Dr. Solomon has received research grants from and serves as a consultant to Novartis, which funded PARAGON-HF, and has similar financial relationships with more than a dozen other pharmaceutical companies. Dr. Ventura reported having no relevant financial interests.
SOURCE: Solomon SD et al. J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.009.
A target systolic blood pressure (SBP) of 120-129 mm Hg in patients with heart failure with preserved ejection fraction proved to be the sweet spot with the lowest rates of major adverse cardiovascular and renal events in a new analysis from the landmark PARAGON-HF trial.
This finding from the largest-ever randomized, controlled study in heart failure with preserved ejection fraction (HFpEF) strengthens support for current U.S. joint hypertension guidelines, which call for a target SBP less than 130 mm Hg in patients with HFpEF (J Am Coll Cardiol. 2017 Aug 8;70[6]:776-803), a recommendation based upon weak evidence until now. That’s because the SPRINT trial, the major impetus for adoption of intensive blood pressure control in the current guidelines, excluded patients with symptomatic HF, Scott D. Solomon, MD, and coinvestigators noted in their new analysis. The study was published in the Journal of the American College of Cardiology and had been planned for presentation during the joint scientific sessions of the American College of Cardiology and the World Heart Federation. ACC organizers chose to present parts of the meeting virtually after COVID-19 concerns caused them to cancel the meeting.
The new analysis from PARAGON-HF (Prospective Comparison of ARNI with ARB Global Outcomes in HFpEF) also ruled out the SBP-lowering effect of sacubitril/valsartan (Entresto) as the explanation for the combination drug’s demonstrated beneficial impact on outcomes in the subgroup with an SBP of 120-129 mm Hg. That wasn’t actually a surprise. Indeed, the new study had two hypotheses: one, that the relationship between SBP and cardiovascular and renal outcomes in HFpEF would follow a J-shaped curve, and two, that sacubitril/valsartan’s blood pressure–lowering effect would not account for the drug’s outcome benefits in the subset of HFpEF patients with an SBP in the sweet spot of 120-129 mm Hg. Both hypotheses were borne out, noted Dr. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, both in Boston.
“These data strongly support that additional mechanisms other than blood pressure–lowering account for the benefit. But this is not surprising. The same can be said for most of the therapies that work in heart failure,” he said in an interview.
Take, for example, spironolactone. In TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist), another major trial in which Dr. Solomon played a leadership role, the beneficial effect of spironolactone on clinical outcomes also proved unrelated to the drug’s blood pressure–lowering effect.
Other known effects of sacubitril/valsartan, a novel angiotensin receptor–neprilysin inhibitor, or ARNI, might in theory account for the observed clinical benefits in ARNI-treated patients with an on-treatment SBP of 120-129 mm Hg in PARAGON-HF. These include improved left atrial remodeling, an increase in natriuretic peptides, and improved myocardial relaxation. However, the current lack of understanding of the basic mechanistic processes underlying the varied clinical expressions of HFpEF is a major factor contributing to the lack of any proven-effective therapy for this extremely common and costly disorder, according to Dr. Solomon and coinvestigators.
In contrast to HFpEF, for which to date there is no proven treatment, heart failure with reduced ejection fraction sacubitril/valsartan has a class I recommendation on the strength of its performance in significantly reducing cardiovascular deaths and heart failure hospitalizations in the PARADIGM-HF trial (N Engl J Med. 2014 Sep 11;371:993-1004).
PARAGON-HF included 4,822 patients with symptomatic HFpEF who were randomized to sacubitril/valsartan at 97/103 mg b.i.d. or valsartan at 160 mg b.i.d. As previously reported (N Engl J Med. 2019 Oct 24;381[17]:1609-20), at an average follow-up of 35 months, the primary outcome – a composite of total hospitalizations for heart failure and cardiovascular death – occurred at a rate of 12.8 events per 100 patient-years in the sacubitril/valsartan group and 14.6 per 100 patient-years in the valsartan arm, for a 13% relative risk reduction that narrowly missed statistical significance (P = .059).
However, sacubitril/valsartan showed significant benefit on some prespecified secondary endpoints, including worsening renal function, change in New York Heart Association class, and quality of life. Women, who notably accounted for 52% of study participants, appeared to benefit from sacubitril/valsartan more than men as evidenced by their 27% relative risk reduction in the primary endpoint. Also, in the roughly half of PARAGON-HF participants with a baseline left ventricular ejection fraction of 45%-57%, treatment with sacubitril/valsartan resulted in a statistically significant 22% relative risk reduction in the primary endpoint, compared with valsartan alone.
SBP and cardiovascular outcomes in HFpEF
In the new analysis, Dr. Solomon and coworkers examined outcomes based on baseline and mean achieved SBP quartiles regardless of treatment arm. In an unadjusted analysis, the primary composite endpoint occurred at a rate of 15.2 events/100 patient-years in HFpEF patients with an achieved SBP below 120 mm Hg, 11.4/100 patient-years at 120-129 mm Hg, 12.2/100 patient-years at 130-139 mm Hg, and 15.6/100 patient-years at 140 mm Hg or more. Further, in a multivariate regression analysis extensively adjusted for atrial fibrillation, sex, race, and numerous other potential confounders, the group with an achieved SBP of 120-129 mm Hg continued to fare best. The adjusted risks for the primary endpoint were 11% and 21% higher in patients in the first and third quartiles of achieved SBP, compared with those at 120-129 mm Hg, although neither trend reached statistical significance. But patients in the top quartile, with an achieved SBP of 140 mm Hg or more, had a highly significant 56% increase in risk, compared with patients in the second-lowest SBP quartile.
Change in blood pressure from baseline to week 48 had no impact on quality of life or high-sensitivity troponin T. However, each 10–mm Hg lowering of SBP was associated with a modest 2.1% reduction in log-transformed N-terminal of the prohormone brain natriuretic peptide.
Sacubitril/valsartan reduced SBP by an average of 5.2 mm Hg more than valsartan alone at 4 weeks regardless of baseline SBP. And the combo drug had a significantly greater SBP-lowering effect in women than men, by a margin of 6.3 mm Hg versus 4.0 mm Hg. But a Cox regression analysis showed that in women, as in the study population as a whole, sacubitril/valsartan’s SBP-lowering effects didn’t account for the drug’s impact on outcomes.
In an editorial accompanying publication of the new PARAGON-HF blood pressure analysis (J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.024), Hector O. Ventura, MD, and colleagues at the Ochsner Clinic in New Orleans observed that the study results “lend some credence to the prognostic relationship of blood pressure in HFpEF, but whether they should serve as a therapeutic target or are merely a prognostic surrogate determined by other pathogenic factors, such as vascular ventricular uncoupling or aortic stiffness on one hand when blood pressure is greater than 140 mm Hg, or a reduced cardiac performance indicated by reduced blood pressure to less than 120 mm Hg, remains uncertain.”
“What is certain, however, is that the relationship and contributions of hypertension in manifest HFpEF are complex, multifactorial and likely go well beyond a simplistic framework of hemodynamic influences,” they added.
Dr. Solomon has received research grants from and serves as a consultant to Novartis, which funded PARAGON-HF, and has similar financial relationships with more than a dozen other pharmaceutical companies. Dr. Ventura reported having no relevant financial interests.
SOURCE: Solomon SD et al. J Am Coll Cardiol. 2020 Mar 16. doi: 10.1016/j.jacc.2020.02.009.
FROM ACC 2020
COVID-19 in pediatric patients: What the hospitalist needs to know
Coronavirus disease (COVID-19) was declared a pandemic by the World Health Organization on March 11. This rapidly spreading disease is caused by the novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The infection has spread to more than 140 countries, including the United States. As of March 16, more than 170,400 people had tested positive for SARS-CoV-2 and more than 6,619 people have died across the globe.
The number of new COVID-19 cases appears to be decreasing in China, but the number of cases are rapidly increasing worldwide. Based on available data, primarily from China, children (aged 0-19 years) account for only about 2% of all cases. Despite the probable low virulence and incidence of infection in children, they could act as potential vectors and transmit infection to more vulnerable populations. As of March 16, approximately 3,823 cases and more than 67 deaths had been reported in the United States with few pediatric patients testing positive for the disease.
SARS-CoV2 transmission mainly occurs via respiratory route through close contact with infected individuals and through fomites. The incubation period ranges from 2-14 days with an average of about 5 days. Adult patients present with cough and fever, which may progress to lower respiratory tract symptoms, including shortness of breath. Approximately 10% of all patients develop severe disease and acute respiratory distress syndrome (ARDS), requiring mechanical ventilation.
COVID-19 carries a mortality rate of up to 3%, but has been significantly higher in the elderly population, and those with chronic health conditions. Available data so far shows that children are at lower risk and the severity of the disease has been milder compared to adults. The reasons for this are not clear at this time. As of March 16, there were no reported COVID-19 related deaths in children under age 9 years.
The pediatric population: Disease patterns and transmission
The epidemiology and spectrum of disease for COVID-19 is poorly understood in pediatrics because of the low number of reported pediatric cases and limited data available from these patients. Small numbers of reported cases in children has led some to believe that children are relatively immune to the infection by SARS-CoV-2. However, Oifang et al. found that children are equally as likely as adults to be infected.1
Liu et al. found that of 366 children admitted to a hospital in Wuhan with respiratory infections in January 2020, 1.6% (six patients) cases were positive for SARS-CoV-2.2 These six children were aged 1-7 years and had all been previously healthy; all six presented with cough and fever of 102.2° F or greater. Four of the children also had vomiting. Laboratory findings were notable for lymphopenia (six of six), leukopenia (four of six), and neutropenia (3/6) with mild to moderate elevation in C-reactive protein (6.8-58.8 mg/L). Five of six children had chest CT scans. One child’s CT scan showed “bilateral ground-glass opacities” (similar to what is reported in adults), three showed “bilateral patchy shadows,” and one was normal. One child (aged 3 years) was admitted to the ICU. All of the children were treated with supportive measures, empiric antibiotics, and antivirals (six of six received oseltamivir and four of six received ribavirin). All six children recovered completely and their median hospital stay was 7.5 days with a range of 5-13 days.
Xia et al. reviewed 20 children (aged 1 day to 14 years) admitted to a hospital in Wuhan during Jan. 23–Feb. 8.3 The study reported that fever and cough were the most common presenting symptoms (approximately 65%). Less common symptoms included rhinorrhea (15%), diarrhea (15%), vomiting (10%), and sore throat (5%). WBC count was normal in majority of children (70%) with leukopenia in 20% and leukocytosis in 10%. Lymphopenia was noted to be 35%. Elevated procalcitonin was noted in 80% of children, although the degree of elevation is unclear. In this study, 8 of 20 children were coinfected with other respiratory pathogens such as influenza, respiratory syncytial virus, mycoplasma, and cytomegalovirus. All children had chest CT scans. Ten of 20 children had bilateral pulmonary lesions, 6 of 20 had unilateral pulmonary lesions, 12 of 20 had ground-glass opacities and 10 of 20 had lung consolidations with halo signs.
Wei et al., retrospective chart review of nine infants admitted for COVID-19 found that all nine had at least one infected family member.4 This study reported that seven of nine were female infants, four of nine had fever, two had mild upper respiratory infection symptoms, and one had no symptoms. The study did report that two infants did not have any information available related to symptoms. None of the infants developed severe symptoms or required ICU admission.
The youngest patient to be diagnosed with COVID-19 was a newborn of less than 24 hours old from England, whose mother also tested positive for SARS-CoV-2. However, Chen et al. found no evidence of vertical transmission of the virus from infected pregnant women to their newborns.5
Although the risk of infection in children has been reported to be low, the infection has been shown to be particularly severe in adults with compromised immune systems and chronic health conditions. Thus immunocompromised children and those with chronic health conditions are thought to be at a higher risk for contracting the infection, with the probability for increased morbidity and mortality. Some of these risk groups include premature infants, young infants, immunocompromised children, and children with chronic health conditions like asthma, diabetes, and others. It is essential that caregivers, healthy siblings, and other family members are protected from contracting the infection in order to protect these vulnerable children. Given the high infectivity of SARS-CoV-2, the implications of infected children attending schools and daycares may be far reaching if there is delayed identification of the infection. For these reasons, it is important to closely monitor and promptly test children living with infected adults to prevent the spread. It may become necessary to close schools to mitigate transmission.
Schools and daycares should work with their local health departments and physicians in case of infected individuals in their community. In China, authorities closed schools and allowed students to receive virtual education from home, which may be a reasonable choice depending on resources.
Current challenges
Given the aggressive transmission of COVID-19, these numbers seem to be increasing exponentially with a significant impact on the life of the entire country. Therefore, we must focus on containing the spread and mitigating the transmission with a multimodality approach.
Some of the initial challenges faced by physicians in the United States were related to difficulty in access to testing in persons under investigation (PUI), which in turn resulted in a delay in diagnosis and infection control. At this time, the need is to increase surge testing capabilities across the country through a variety of innovative approaches including public-private partnerships with commercial labs through Emergency Use Authorization (EUA) issued by the Centers for Disease Control and Prevention and the Department of Health and Human Services. To minimize exposure to health care professionals, telemedicine and telehealth capabilities should be exploited. This will minimize the exposure to infected patients and reduce the need for already limited personal protective equipment (PPE). As the number of cases rise, hospitals should expect and prepare for a surge in COVID-19–related hospitalizations and health care utilization.
Conclusion
Various theories are being proposed as to why children are not experiencing severe disease with COVID-19. Children may have cross-protective immunity from infection with other coronaviruses. Children may not have the same exposures from work, travel, and caregiving that adults experience as they are typically exposed by someone in their home. At this time, not enough is known about clinical presentations in children as the situation continues to evolve across the globe.
Respiratory infections in children pose unique infection control challenges with respect to compliant hand hygiene, cough etiquette, and the use of PPE when indicated. There is also concern for persistent fecal shedding of virus in infected pediatric patients, which could be another mode of transmission.6 Children could, however, be very efficient vectors of COVID-19, similar to flu, and potentially spread the pathogen to very vulnerable populations leading to high morbidity and mortality. School closures are an effective social distancing measure needed to flatten the curve and avoid overwhelming the health care structure of the United States.
Dr. Konanki is a board-certified pediatrician doing inpatient work at Wellspan Chambersburg Hospital and outpatient work at Keystone Pediatrics in Chambersburg, Pa. He also serves as the physician member of the hospital’s Code Blue Jr. committee and as a member of Quality Metrics committee at Keystone Health. Dr. Tirupathi is the medical director of Keystone Infectious Diseases/HIV in Chambersburg, Pa., and currently chair of infection prevention at Wellspan Chambersburg and Waynesboro (Pa.) Hospitals. He also is the lead physician for antibiotic stewardship at these hospitals. Dr. Palabindala is hospital medicine division chief at the University of Mississippi Medical Center, Jackson.
References
1. Bi Q et al. Epidemiology and transmission of COVID-19 in Shenzhen China: Analysis of 391 cases and 1,286 of their close contacts. medRxiv 2020.03.03.20028423.
2. Liu W et al. Detection of Covid-19 in children in early January 2020 in Wuhan, China. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
3. Xia W et al. Clinical and CT features in pediatric patients with COVID‐19 infection: Different points from adults. Pediatr Pulmonol. 2020 Mar 5. doi: 10.1002/ppul.24718.
4. Wei M et al. Novel Coronavirus infection in hospitalized infants under 1 year of age in China. JAMA. 2020 Feb. 14. doi: 10.1001/jama.2020.2131.
5. Huijun C et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: A retrospective review of medical records. Lancet. 2020 Mar 7 395;10226:809-15.
6. Xu Y et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat Med. 2020 Mar 13. doi. org/10.1038/s41591-020-0817-4.
Coronavirus disease (COVID-19) was declared a pandemic by the World Health Organization on March 11. This rapidly spreading disease is caused by the novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The infection has spread to more than 140 countries, including the United States. As of March 16, more than 170,400 people had tested positive for SARS-CoV-2 and more than 6,619 people have died across the globe.
The number of new COVID-19 cases appears to be decreasing in China, but the number of cases are rapidly increasing worldwide. Based on available data, primarily from China, children (aged 0-19 years) account for only about 2% of all cases. Despite the probable low virulence and incidence of infection in children, they could act as potential vectors and transmit infection to more vulnerable populations. As of March 16, approximately 3,823 cases and more than 67 deaths had been reported in the United States with few pediatric patients testing positive for the disease.
SARS-CoV2 transmission mainly occurs via respiratory route through close contact with infected individuals and through fomites. The incubation period ranges from 2-14 days with an average of about 5 days. Adult patients present with cough and fever, which may progress to lower respiratory tract symptoms, including shortness of breath. Approximately 10% of all patients develop severe disease and acute respiratory distress syndrome (ARDS), requiring mechanical ventilation.
COVID-19 carries a mortality rate of up to 3%, but has been significantly higher in the elderly population, and those with chronic health conditions. Available data so far shows that children are at lower risk and the severity of the disease has been milder compared to adults. The reasons for this are not clear at this time. As of March 16, there were no reported COVID-19 related deaths in children under age 9 years.
The pediatric population: Disease patterns and transmission
The epidemiology and spectrum of disease for COVID-19 is poorly understood in pediatrics because of the low number of reported pediatric cases and limited data available from these patients. Small numbers of reported cases in children has led some to believe that children are relatively immune to the infection by SARS-CoV-2. However, Oifang et al. found that children are equally as likely as adults to be infected.1
Liu et al. found that of 366 children admitted to a hospital in Wuhan with respiratory infections in January 2020, 1.6% (six patients) cases were positive for SARS-CoV-2.2 These six children were aged 1-7 years and had all been previously healthy; all six presented with cough and fever of 102.2° F or greater. Four of the children also had vomiting. Laboratory findings were notable for lymphopenia (six of six), leukopenia (four of six), and neutropenia (3/6) with mild to moderate elevation in C-reactive protein (6.8-58.8 mg/L). Five of six children had chest CT scans. One child’s CT scan showed “bilateral ground-glass opacities” (similar to what is reported in adults), three showed “bilateral patchy shadows,” and one was normal. One child (aged 3 years) was admitted to the ICU. All of the children were treated with supportive measures, empiric antibiotics, and antivirals (six of six received oseltamivir and four of six received ribavirin). All six children recovered completely and their median hospital stay was 7.5 days with a range of 5-13 days.
Xia et al. reviewed 20 children (aged 1 day to 14 years) admitted to a hospital in Wuhan during Jan. 23–Feb. 8.3 The study reported that fever and cough were the most common presenting symptoms (approximately 65%). Less common symptoms included rhinorrhea (15%), diarrhea (15%), vomiting (10%), and sore throat (5%). WBC count was normal in majority of children (70%) with leukopenia in 20% and leukocytosis in 10%. Lymphopenia was noted to be 35%. Elevated procalcitonin was noted in 80% of children, although the degree of elevation is unclear. In this study, 8 of 20 children were coinfected with other respiratory pathogens such as influenza, respiratory syncytial virus, mycoplasma, and cytomegalovirus. All children had chest CT scans. Ten of 20 children had bilateral pulmonary lesions, 6 of 20 had unilateral pulmonary lesions, 12 of 20 had ground-glass opacities and 10 of 20 had lung consolidations with halo signs.
Wei et al., retrospective chart review of nine infants admitted for COVID-19 found that all nine had at least one infected family member.4 This study reported that seven of nine were female infants, four of nine had fever, two had mild upper respiratory infection symptoms, and one had no symptoms. The study did report that two infants did not have any information available related to symptoms. None of the infants developed severe symptoms or required ICU admission.
The youngest patient to be diagnosed with COVID-19 was a newborn of less than 24 hours old from England, whose mother also tested positive for SARS-CoV-2. However, Chen et al. found no evidence of vertical transmission of the virus from infected pregnant women to their newborns.5
Although the risk of infection in children has been reported to be low, the infection has been shown to be particularly severe in adults with compromised immune systems and chronic health conditions. Thus immunocompromised children and those with chronic health conditions are thought to be at a higher risk for contracting the infection, with the probability for increased morbidity and mortality. Some of these risk groups include premature infants, young infants, immunocompromised children, and children with chronic health conditions like asthma, diabetes, and others. It is essential that caregivers, healthy siblings, and other family members are protected from contracting the infection in order to protect these vulnerable children. Given the high infectivity of SARS-CoV-2, the implications of infected children attending schools and daycares may be far reaching if there is delayed identification of the infection. For these reasons, it is important to closely monitor and promptly test children living with infected adults to prevent the spread. It may become necessary to close schools to mitigate transmission.
Schools and daycares should work with their local health departments and physicians in case of infected individuals in their community. In China, authorities closed schools and allowed students to receive virtual education from home, which may be a reasonable choice depending on resources.
Current challenges
Given the aggressive transmission of COVID-19, these numbers seem to be increasing exponentially with a significant impact on the life of the entire country. Therefore, we must focus on containing the spread and mitigating the transmission with a multimodality approach.
Some of the initial challenges faced by physicians in the United States were related to difficulty in access to testing in persons under investigation (PUI), which in turn resulted in a delay in diagnosis and infection control. At this time, the need is to increase surge testing capabilities across the country through a variety of innovative approaches including public-private partnerships with commercial labs through Emergency Use Authorization (EUA) issued by the Centers for Disease Control and Prevention and the Department of Health and Human Services. To minimize exposure to health care professionals, telemedicine and telehealth capabilities should be exploited. This will minimize the exposure to infected patients and reduce the need for already limited personal protective equipment (PPE). As the number of cases rise, hospitals should expect and prepare for a surge in COVID-19–related hospitalizations and health care utilization.
Conclusion
Various theories are being proposed as to why children are not experiencing severe disease with COVID-19. Children may have cross-protective immunity from infection with other coronaviruses. Children may not have the same exposures from work, travel, and caregiving that adults experience as they are typically exposed by someone in their home. At this time, not enough is known about clinical presentations in children as the situation continues to evolve across the globe.
Respiratory infections in children pose unique infection control challenges with respect to compliant hand hygiene, cough etiquette, and the use of PPE when indicated. There is also concern for persistent fecal shedding of virus in infected pediatric patients, which could be another mode of transmission.6 Children could, however, be very efficient vectors of COVID-19, similar to flu, and potentially spread the pathogen to very vulnerable populations leading to high morbidity and mortality. School closures are an effective social distancing measure needed to flatten the curve and avoid overwhelming the health care structure of the United States.
Dr. Konanki is a board-certified pediatrician doing inpatient work at Wellspan Chambersburg Hospital and outpatient work at Keystone Pediatrics in Chambersburg, Pa. He also serves as the physician member of the hospital’s Code Blue Jr. committee and as a member of Quality Metrics committee at Keystone Health. Dr. Tirupathi is the medical director of Keystone Infectious Diseases/HIV in Chambersburg, Pa., and currently chair of infection prevention at Wellspan Chambersburg and Waynesboro (Pa.) Hospitals. He also is the lead physician for antibiotic stewardship at these hospitals. Dr. Palabindala is hospital medicine division chief at the University of Mississippi Medical Center, Jackson.
References
1. Bi Q et al. Epidemiology and transmission of COVID-19 in Shenzhen China: Analysis of 391 cases and 1,286 of their close contacts. medRxiv 2020.03.03.20028423.
2. Liu W et al. Detection of Covid-19 in children in early January 2020 in Wuhan, China. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
3. Xia W et al. Clinical and CT features in pediatric patients with COVID‐19 infection: Different points from adults. Pediatr Pulmonol. 2020 Mar 5. doi: 10.1002/ppul.24718.
4. Wei M et al. Novel Coronavirus infection in hospitalized infants under 1 year of age in China. JAMA. 2020 Feb. 14. doi: 10.1001/jama.2020.2131.
5. Huijun C et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: A retrospective review of medical records. Lancet. 2020 Mar 7 395;10226:809-15.
6. Xu Y et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat Med. 2020 Mar 13. doi. org/10.1038/s41591-020-0817-4.
Coronavirus disease (COVID-19) was declared a pandemic by the World Health Organization on March 11. This rapidly spreading disease is caused by the novel coronavirus, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). The infection has spread to more than 140 countries, including the United States. As of March 16, more than 170,400 people had tested positive for SARS-CoV-2 and more than 6,619 people have died across the globe.
The number of new COVID-19 cases appears to be decreasing in China, but the number of cases are rapidly increasing worldwide. Based on available data, primarily from China, children (aged 0-19 years) account for only about 2% of all cases. Despite the probable low virulence and incidence of infection in children, they could act as potential vectors and transmit infection to more vulnerable populations. As of March 16, approximately 3,823 cases and more than 67 deaths had been reported in the United States with few pediatric patients testing positive for the disease.
SARS-CoV2 transmission mainly occurs via respiratory route through close contact with infected individuals and through fomites. The incubation period ranges from 2-14 days with an average of about 5 days. Adult patients present with cough and fever, which may progress to lower respiratory tract symptoms, including shortness of breath. Approximately 10% of all patients develop severe disease and acute respiratory distress syndrome (ARDS), requiring mechanical ventilation.
COVID-19 carries a mortality rate of up to 3%, but has been significantly higher in the elderly population, and those with chronic health conditions. Available data so far shows that children are at lower risk and the severity of the disease has been milder compared to adults. The reasons for this are not clear at this time. As of March 16, there were no reported COVID-19 related deaths in children under age 9 years.
The pediatric population: Disease patterns and transmission
The epidemiology and spectrum of disease for COVID-19 is poorly understood in pediatrics because of the low number of reported pediatric cases and limited data available from these patients. Small numbers of reported cases in children has led some to believe that children are relatively immune to the infection by SARS-CoV-2. However, Oifang et al. found that children are equally as likely as adults to be infected.1
Liu et al. found that of 366 children admitted to a hospital in Wuhan with respiratory infections in January 2020, 1.6% (six patients) cases were positive for SARS-CoV-2.2 These six children were aged 1-7 years and had all been previously healthy; all six presented with cough and fever of 102.2° F or greater. Four of the children also had vomiting. Laboratory findings were notable for lymphopenia (six of six), leukopenia (four of six), and neutropenia (3/6) with mild to moderate elevation in C-reactive protein (6.8-58.8 mg/L). Five of six children had chest CT scans. One child’s CT scan showed “bilateral ground-glass opacities” (similar to what is reported in adults), three showed “bilateral patchy shadows,” and one was normal. One child (aged 3 years) was admitted to the ICU. All of the children were treated with supportive measures, empiric antibiotics, and antivirals (six of six received oseltamivir and four of six received ribavirin). All six children recovered completely and their median hospital stay was 7.5 days with a range of 5-13 days.
Xia et al. reviewed 20 children (aged 1 day to 14 years) admitted to a hospital in Wuhan during Jan. 23–Feb. 8.3 The study reported that fever and cough were the most common presenting symptoms (approximately 65%). Less common symptoms included rhinorrhea (15%), diarrhea (15%), vomiting (10%), and sore throat (5%). WBC count was normal in majority of children (70%) with leukopenia in 20% and leukocytosis in 10%. Lymphopenia was noted to be 35%. Elevated procalcitonin was noted in 80% of children, although the degree of elevation is unclear. In this study, 8 of 20 children were coinfected with other respiratory pathogens such as influenza, respiratory syncytial virus, mycoplasma, and cytomegalovirus. All children had chest CT scans. Ten of 20 children had bilateral pulmonary lesions, 6 of 20 had unilateral pulmonary lesions, 12 of 20 had ground-glass opacities and 10 of 20 had lung consolidations with halo signs.
Wei et al., retrospective chart review of nine infants admitted for COVID-19 found that all nine had at least one infected family member.4 This study reported that seven of nine were female infants, four of nine had fever, two had mild upper respiratory infection symptoms, and one had no symptoms. The study did report that two infants did not have any information available related to symptoms. None of the infants developed severe symptoms or required ICU admission.
The youngest patient to be diagnosed with COVID-19 was a newborn of less than 24 hours old from England, whose mother also tested positive for SARS-CoV-2. However, Chen et al. found no evidence of vertical transmission of the virus from infected pregnant women to their newborns.5
Although the risk of infection in children has been reported to be low, the infection has been shown to be particularly severe in adults with compromised immune systems and chronic health conditions. Thus immunocompromised children and those with chronic health conditions are thought to be at a higher risk for contracting the infection, with the probability for increased morbidity and mortality. Some of these risk groups include premature infants, young infants, immunocompromised children, and children with chronic health conditions like asthma, diabetes, and others. It is essential that caregivers, healthy siblings, and other family members are protected from contracting the infection in order to protect these vulnerable children. Given the high infectivity of SARS-CoV-2, the implications of infected children attending schools and daycares may be far reaching if there is delayed identification of the infection. For these reasons, it is important to closely monitor and promptly test children living with infected adults to prevent the spread. It may become necessary to close schools to mitigate transmission.
Schools and daycares should work with their local health departments and physicians in case of infected individuals in their community. In China, authorities closed schools and allowed students to receive virtual education from home, which may be a reasonable choice depending on resources.
Current challenges
Given the aggressive transmission of COVID-19, these numbers seem to be increasing exponentially with a significant impact on the life of the entire country. Therefore, we must focus on containing the spread and mitigating the transmission with a multimodality approach.
Some of the initial challenges faced by physicians in the United States were related to difficulty in access to testing in persons under investigation (PUI), which in turn resulted in a delay in diagnosis and infection control. At this time, the need is to increase surge testing capabilities across the country through a variety of innovative approaches including public-private partnerships with commercial labs through Emergency Use Authorization (EUA) issued by the Centers for Disease Control and Prevention and the Department of Health and Human Services. To minimize exposure to health care professionals, telemedicine and telehealth capabilities should be exploited. This will minimize the exposure to infected patients and reduce the need for already limited personal protective equipment (PPE). As the number of cases rise, hospitals should expect and prepare for a surge in COVID-19–related hospitalizations and health care utilization.
Conclusion
Various theories are being proposed as to why children are not experiencing severe disease with COVID-19. Children may have cross-protective immunity from infection with other coronaviruses. Children may not have the same exposures from work, travel, and caregiving that adults experience as they are typically exposed by someone in their home. At this time, not enough is known about clinical presentations in children as the situation continues to evolve across the globe.
Respiratory infections in children pose unique infection control challenges with respect to compliant hand hygiene, cough etiquette, and the use of PPE when indicated. There is also concern for persistent fecal shedding of virus in infected pediatric patients, which could be another mode of transmission.6 Children could, however, be very efficient vectors of COVID-19, similar to flu, and potentially spread the pathogen to very vulnerable populations leading to high morbidity and mortality. School closures are an effective social distancing measure needed to flatten the curve and avoid overwhelming the health care structure of the United States.
Dr. Konanki is a board-certified pediatrician doing inpatient work at Wellspan Chambersburg Hospital and outpatient work at Keystone Pediatrics in Chambersburg, Pa. He also serves as the physician member of the hospital’s Code Blue Jr. committee and as a member of Quality Metrics committee at Keystone Health. Dr. Tirupathi is the medical director of Keystone Infectious Diseases/HIV in Chambersburg, Pa., and currently chair of infection prevention at Wellspan Chambersburg and Waynesboro (Pa.) Hospitals. He also is the lead physician for antibiotic stewardship at these hospitals. Dr. Palabindala is hospital medicine division chief at the University of Mississippi Medical Center, Jackson.
References
1. Bi Q et al. Epidemiology and transmission of COVID-19 in Shenzhen China: Analysis of 391 cases and 1,286 of their close contacts. medRxiv 2020.03.03.20028423.
2. Liu W et al. Detection of Covid-19 in children in early January 2020 in Wuhan, China. N Engl J Med. 2020 Mar 12. doi: 10.1056/NEJMc2003717.
3. Xia W et al. Clinical and CT features in pediatric patients with COVID‐19 infection: Different points from adults. Pediatr Pulmonol. 2020 Mar 5. doi: 10.1002/ppul.24718.
4. Wei M et al. Novel Coronavirus infection in hospitalized infants under 1 year of age in China. JAMA. 2020 Feb. 14. doi: 10.1001/jama.2020.2131.
5. Huijun C et al. Clinical characteristics and intrauterine vertical transmission potential of COVID-19 infection in nine pregnant women: A retrospective review of medical records. Lancet. 2020 Mar 7 395;10226:809-15.
6. Xu Y et al. Characteristics of pediatric SARS-CoV-2 infection and potential evidence for persistent fecal viral shedding. Nat Med. 2020 Mar 13. doi. org/10.1038/s41591-020-0817-4.
Coronavirus stays in aerosols for hours, on surfaces for days
according to a new study.
The data indicate that the stability of the new virus is similar to that of SARS-CoV-1, which caused the SARS epidemic, researchers report in an article published on the medRxivpreprint server. (The posted article has been submitted for journal publication but has not been peer reviewed.)
Transmission of SARS-CoV-2, which causes COVID-19, has quickly outstripped the pace of the 2003 SARS epidemic. “Superspread” of the earlier disease arose from infection during medical procedures, in which a single infected individual seeded many secondary cases. In contrast, the novel coronavirus appears to be spread more through human-to-human transmission in a variety of settings.
However, it’s not yet known the extent to which asymptomatic or presymptomatic individuals spread the new virus through daily routine.
To investigate how long SARS-CoV-2 remains infective in the environment, Neeltje van Doremalen, PhD, of the Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, in Hamilton, Montana, and colleagues conducted simulation experiments in which they compared the viability of SARS-CoV-2 with that of SARS-CoV-1 in aerosols and on surfaces.
Among patients infected with SARS-CoV-2, viral loads in the upper respiratory tract are high; as a consequence, respiratory secretion in the form of aerosols (<5 μm) or droplets (>5 mcm) is likely, the authors note.
van Doremalen and colleagues used nebulizers to generate aerosols. Samples of SARS-CoV-1 and SARS-CoV-2 were collecting at 0, 30, 60, 120, and 180 minutes on a gelatin filter. The researchers then tested the infectivity of the viruses on Vero cells grown in culture.
They found that SARS-CoV-2 was largely stable through the full 180-minute test, with only a slight decline at 3 hours. This time course is similar to that of SARS-CoV-1; both viruses have a median half-life in aerosols of 2.7 hours (range, 1.65 hr for SARS-CoV-1, vs 7.24 hr for SARS-CoV-2).
The researchers then tested the viruses on a variety of surfaces for up to 7 days, using humidity values and temperatures designed to mimic “a variety of household and hospital situations.” The volumes of viral exposures that the team used were consistent with amounts found in the human upper and lower respiratory tracts.
For example, they applied 50 mcL of virus-containing solution to a piece of cardboard and then swabbed the surface, at different times, with an additional 1 mcL of medium. Each surface assay was replicated three times.
The novel coronavirus was most stable on plastic and stainless steel, with some virus remaining viable up to 72 hours. However, by that time the viral load had fallen by about three orders of magnitude, indicating exponential decay. This profile was remarkably similar to that of SARS-CoV-1, according to the authors.
However, the two viruses differed in staying power on copper and cardboard. No viable SARS-CoV-2 was detectable on copper after 4 hours or on cardboard after 24 hours. In contrast, SARS-CoV-1 was not viable beyond 8 hours for either copper or cardboard.
“Taken together, our results indicate that aerosol and fomite transmission of HCoV-19 [SARS-CoV-2] are plausible, as the virus can remain viable in aerosols for multiple hours and on surfaces up to days,” the authors conclude.
Andrew Pekosz, PhD, codirector of the Center of Excellence in Influenza Research and Surveillance and director of the Center for Emerging Viruses and Infectious Diseases at the Johns Hopkins Center for Global Health, Baltimore, Maryland, applauds the real-world value of the experiments.
“The PCR [polymerase chain reaction] test used [in other studies] to detect SARS-CoV-2 just detects the virus genome. It doesn’t tell you if the virus was still infectious, or ‘viable.’ That’s why this study is interesting,” Pekosz said. “It focuses on infectious virus, which is the virus that has the potential to transmit and infect another person. What we don’t know yet is how much infectious (viable) virus is needed to initiate infection in another person.”
He suggests that further investigations evaluate other types of environmental surfaces, including lacquered wood that is made into desks and ceramic tiles found in bathrooms and kitchens.
One limitation of the study is that the data for experiments on cardboard were more variable than the data for other surfaces tested.
The investigators and Pekosz have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
according to a new study.

The data indicate that the stability of the new virus is similar to that of SARS-CoV-1, which caused the SARS epidemic, researchers report in an article published on the medRxivpreprint server. (The posted article has been submitted for journal publication but has not been peer reviewed.)
Transmission of SARS-CoV-2, which causes COVID-19, has quickly outstripped the pace of the 2003 SARS epidemic. “Superspread” of the earlier disease arose from infection during medical procedures, in which a single infected individual seeded many secondary cases. In contrast, the novel coronavirus appears to be spread more through human-to-human transmission in a variety of settings.
However, it’s not yet known the extent to which asymptomatic or presymptomatic individuals spread the new virus through daily routine.
To investigate how long SARS-CoV-2 remains infective in the environment, Neeltje van Doremalen, PhD, of the Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, in Hamilton, Montana, and colleagues conducted simulation experiments in which they compared the viability of SARS-CoV-2 with that of SARS-CoV-1 in aerosols and on surfaces.
Among patients infected with SARS-CoV-2, viral loads in the upper respiratory tract are high; as a consequence, respiratory secretion in the form of aerosols (<5 μm) or droplets (>5 mcm) is likely, the authors note.
van Doremalen and colleagues used nebulizers to generate aerosols. Samples of SARS-CoV-1 and SARS-CoV-2 were collecting at 0, 30, 60, 120, and 180 minutes on a gelatin filter. The researchers then tested the infectivity of the viruses on Vero cells grown in culture.
They found that SARS-CoV-2 was largely stable through the full 180-minute test, with only a slight decline at 3 hours. This time course is similar to that of SARS-CoV-1; both viruses have a median half-life in aerosols of 2.7 hours (range, 1.65 hr for SARS-CoV-1, vs 7.24 hr for SARS-CoV-2).
The researchers then tested the viruses on a variety of surfaces for up to 7 days, using humidity values and temperatures designed to mimic “a variety of household and hospital situations.” The volumes of viral exposures that the team used were consistent with amounts found in the human upper and lower respiratory tracts.
For example, they applied 50 mcL of virus-containing solution to a piece of cardboard and then swabbed the surface, at different times, with an additional 1 mcL of medium. Each surface assay was replicated three times.
The novel coronavirus was most stable on plastic and stainless steel, with some virus remaining viable up to 72 hours. However, by that time the viral load had fallen by about three orders of magnitude, indicating exponential decay. This profile was remarkably similar to that of SARS-CoV-1, according to the authors.
However, the two viruses differed in staying power on copper and cardboard. No viable SARS-CoV-2 was detectable on copper after 4 hours or on cardboard after 24 hours. In contrast, SARS-CoV-1 was not viable beyond 8 hours for either copper or cardboard.
“Taken together, our results indicate that aerosol and fomite transmission of HCoV-19 [SARS-CoV-2] are plausible, as the virus can remain viable in aerosols for multiple hours and on surfaces up to days,” the authors conclude.
Andrew Pekosz, PhD, codirector of the Center of Excellence in Influenza Research and Surveillance and director of the Center for Emerging Viruses and Infectious Diseases at the Johns Hopkins Center for Global Health, Baltimore, Maryland, applauds the real-world value of the experiments.
“The PCR [polymerase chain reaction] test used [in other studies] to detect SARS-CoV-2 just detects the virus genome. It doesn’t tell you if the virus was still infectious, or ‘viable.’ That’s why this study is interesting,” Pekosz said. “It focuses on infectious virus, which is the virus that has the potential to transmit and infect another person. What we don’t know yet is how much infectious (viable) virus is needed to initiate infection in another person.”
He suggests that further investigations evaluate other types of environmental surfaces, including lacquered wood that is made into desks and ceramic tiles found in bathrooms and kitchens.
One limitation of the study is that the data for experiments on cardboard were more variable than the data for other surfaces tested.
The investigators and Pekosz have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
according to a new study.

The data indicate that the stability of the new virus is similar to that of SARS-CoV-1, which caused the SARS epidemic, researchers report in an article published on the medRxivpreprint server. (The posted article has been submitted for journal publication but has not been peer reviewed.)
Transmission of SARS-CoV-2, which causes COVID-19, has quickly outstripped the pace of the 2003 SARS epidemic. “Superspread” of the earlier disease arose from infection during medical procedures, in which a single infected individual seeded many secondary cases. In contrast, the novel coronavirus appears to be spread more through human-to-human transmission in a variety of settings.
However, it’s not yet known the extent to which asymptomatic or presymptomatic individuals spread the new virus through daily routine.
To investigate how long SARS-CoV-2 remains infective in the environment, Neeltje van Doremalen, PhD, of the Laboratory of Virology, Division of Intramural Research, National Institute of Allergy and Infectious Diseases, in Hamilton, Montana, and colleagues conducted simulation experiments in which they compared the viability of SARS-CoV-2 with that of SARS-CoV-1 in aerosols and on surfaces.
Among patients infected with SARS-CoV-2, viral loads in the upper respiratory tract are high; as a consequence, respiratory secretion in the form of aerosols (<5 μm) or droplets (>5 mcm) is likely, the authors note.
van Doremalen and colleagues used nebulizers to generate aerosols. Samples of SARS-CoV-1 and SARS-CoV-2 were collecting at 0, 30, 60, 120, and 180 minutes on a gelatin filter. The researchers then tested the infectivity of the viruses on Vero cells grown in culture.
They found that SARS-CoV-2 was largely stable through the full 180-minute test, with only a slight decline at 3 hours. This time course is similar to that of SARS-CoV-1; both viruses have a median half-life in aerosols of 2.7 hours (range, 1.65 hr for SARS-CoV-1, vs 7.24 hr for SARS-CoV-2).
The researchers then tested the viruses on a variety of surfaces for up to 7 days, using humidity values and temperatures designed to mimic “a variety of household and hospital situations.” The volumes of viral exposures that the team used were consistent with amounts found in the human upper and lower respiratory tracts.
For example, they applied 50 mcL of virus-containing solution to a piece of cardboard and then swabbed the surface, at different times, with an additional 1 mcL of medium. Each surface assay was replicated three times.
The novel coronavirus was most stable on plastic and stainless steel, with some virus remaining viable up to 72 hours. However, by that time the viral load had fallen by about three orders of magnitude, indicating exponential decay. This profile was remarkably similar to that of SARS-CoV-1, according to the authors.
However, the two viruses differed in staying power on copper and cardboard. No viable SARS-CoV-2 was detectable on copper after 4 hours or on cardboard after 24 hours. In contrast, SARS-CoV-1 was not viable beyond 8 hours for either copper or cardboard.
“Taken together, our results indicate that aerosol and fomite transmission of HCoV-19 [SARS-CoV-2] are plausible, as the virus can remain viable in aerosols for multiple hours and on surfaces up to days,” the authors conclude.
Andrew Pekosz, PhD, codirector of the Center of Excellence in Influenza Research and Surveillance and director of the Center for Emerging Viruses and Infectious Diseases at the Johns Hopkins Center for Global Health, Baltimore, Maryland, applauds the real-world value of the experiments.
“The PCR [polymerase chain reaction] test used [in other studies] to detect SARS-CoV-2 just detects the virus genome. It doesn’t tell you if the virus was still infectious, or ‘viable.’ That’s why this study is interesting,” Pekosz said. “It focuses on infectious virus, which is the virus that has the potential to transmit and infect another person. What we don’t know yet is how much infectious (viable) virus is needed to initiate infection in another person.”
He suggests that further investigations evaluate other types of environmental surfaces, including lacquered wood that is made into desks and ceramic tiles found in bathrooms and kitchens.
One limitation of the study is that the data for experiments on cardboard were more variable than the data for other surfaces tested.
The investigators and Pekosz have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Potential GI manifestation, transmission of coronavirus
The novel coronavirus (2019-nCoV) shows evidence of causing gastrointestinal symptoms and has the potential to be transmitted by the fecal-oral route, according to a new report from physicians at Shanghai Jiao Tong University, published online (Gastroenterology. 2020 March 3. doi: 10.1053/j.gastro.2020.02.054).
The virus’s respiratory symptoms are well documented and suggest primary transmission by droplet or contact, while other symptoms such as diarrhea, nausea, vomiting, and abdominal discomfort are less common and appear to vary between populations. The SARS coronavirus showed up in stool, even sometimes in patients discharged from the hospital. In a study of hospitalized patients in Wuhan, China, 10.1% of coronavirus patients had diarrhea and nausea in the 1-2 days before onset of fever and dyspnea. The first U.S. patient to be diagnosed had a 2-day history of nausea and vomiting, and had a loose bowel movement on the second day in the hospital. Clinicians later confirmed the presence of viral RNA in both the patient’s stool and airway.
The authors say that researchers in China have isolated viral RNA from the stool of two patients (unpublished), and it has been found in saliva, suggesting the possibility of the salivary gland as an infection or transmission route.
The authors maintain that previous studies likely overlooked or neglected patients who had mild intestinal symptoms. “Many efforts should be made to be alert on the initial digestive symptoms of COVID-19 for early detection, early diagnosis, early isolation and early intervention,” the authors wrote.
Like other coronaviruses, it appears that 2019-nCoV infects cells through an interaction between viral transmembrane spike glycoprotein (S-protein) receptor-binding domain, and the cell receptors angiotensin-converting enzyme 2 (ACE-2) and host cellular transmembrane serine protease (TMPRSS). Transcriptome analysis has shown that human lung AT2 cells express ACE-2 and TMPRSS, but esophagus upper and stratified epithelial cells also express both factors, as do stratified epithelial cells and absorptive enterocytes in the ileum and colon.
The researchers call for investigation into ACE-2 fusion proteins and TMPRSS inhibitors for diagnosis, prophylaxis, or treatment of COVID-19.
The authors also noted that COVID-19 has been linked to mild to moderate liver injury as revealed by elevated aminotransferases, hypoproteinemia and prothrombin time prolongation. This also has precedent in that the SARS coronavirus can infect the liver, and biopsies revealed mitoses and apoptosis, along with other abnormalities. SARS-associated hepatitis may be the result of viral hepatitis, immune overreaction, or a secondary effect of antiviral medications or other drugs. Little is known to date about the ability of 2019-nCoV to infect the liver, but single-cell RNA sequencing data from two distinct cohorts showed more ACE-2 expression in cholangiocytes (59.7%) than hepatocytes (2.6%), which indicates that the virus might directly affect intrahepatic bile ducts.
The authors had no sources of funding or financial conflicts.
SOURCE: GU J et al. Gastroenterology. 2020 March 3. doi: 10.1053/j.gastro.2020.02.054.
*This story was updated on 4/10.2020.
The novel coronavirus (2019-nCoV) shows evidence of causing gastrointestinal symptoms and has the potential to be transmitted by the fecal-oral route, according to a new report from physicians at Shanghai Jiao Tong University, published online (Gastroenterology. 2020 March 3. doi: 10.1053/j.gastro.2020.02.054).
The virus’s respiratory symptoms are well documented and suggest primary transmission by droplet or contact, while other symptoms such as diarrhea, nausea, vomiting, and abdominal discomfort are less common and appear to vary between populations. The SARS coronavirus showed up in stool, even sometimes in patients discharged from the hospital. In a study of hospitalized patients in Wuhan, China, 10.1% of coronavirus patients had diarrhea and nausea in the 1-2 days before onset of fever and dyspnea. The first U.S. patient to be diagnosed had a 2-day history of nausea and vomiting, and had a loose bowel movement on the second day in the hospital. Clinicians later confirmed the presence of viral RNA in both the patient’s stool and airway.
The authors say that researchers in China have isolated viral RNA from the stool of two patients (unpublished), and it has been found in saliva, suggesting the possibility of the salivary gland as an infection or transmission route.
The authors maintain that previous studies likely overlooked or neglected patients who had mild intestinal symptoms. “Many efforts should be made to be alert on the initial digestive symptoms of COVID-19 for early detection, early diagnosis, early isolation and early intervention,” the authors wrote.
Like other coronaviruses, it appears that 2019-nCoV infects cells through an interaction between viral transmembrane spike glycoprotein (S-protein) receptor-binding domain, and the cell receptors angiotensin-converting enzyme 2 (ACE-2) and host cellular transmembrane serine protease (TMPRSS). Transcriptome analysis has shown that human lung AT2 cells express ACE-2 and TMPRSS, but esophagus upper and stratified epithelial cells also express both factors, as do stratified epithelial cells and absorptive enterocytes in the ileum and colon.
The researchers call for investigation into ACE-2 fusion proteins and TMPRSS inhibitors for diagnosis, prophylaxis, or treatment of COVID-19.
The authors also noted that COVID-19 has been linked to mild to moderate liver injury as revealed by elevated aminotransferases, hypoproteinemia and prothrombin time prolongation. This also has precedent in that the SARS coronavirus can infect the liver, and biopsies revealed mitoses and apoptosis, along with other abnormalities. SARS-associated hepatitis may be the result of viral hepatitis, immune overreaction, or a secondary effect of antiviral medications or other drugs. Little is known to date about the ability of 2019-nCoV to infect the liver, but single-cell RNA sequencing data from two distinct cohorts showed more ACE-2 expression in cholangiocytes (59.7%) than hepatocytes (2.6%), which indicates that the virus might directly affect intrahepatic bile ducts.
The authors had no sources of funding or financial conflicts.
SOURCE: GU J et al. Gastroenterology. 2020 March 3. doi: 10.1053/j.gastro.2020.02.054.
*This story was updated on 4/10.2020.
The novel coronavirus (2019-nCoV) shows evidence of causing gastrointestinal symptoms and has the potential to be transmitted by the fecal-oral route, according to a new report from physicians at Shanghai Jiao Tong University, published online (Gastroenterology. 2020 March 3. doi: 10.1053/j.gastro.2020.02.054).
The virus’s respiratory symptoms are well documented and suggest primary transmission by droplet or contact, while other symptoms such as diarrhea, nausea, vomiting, and abdominal discomfort are less common and appear to vary between populations. The SARS coronavirus showed up in stool, even sometimes in patients discharged from the hospital. In a study of hospitalized patients in Wuhan, China, 10.1% of coronavirus patients had diarrhea and nausea in the 1-2 days before onset of fever and dyspnea. The first U.S. patient to be diagnosed had a 2-day history of nausea and vomiting, and had a loose bowel movement on the second day in the hospital. Clinicians later confirmed the presence of viral RNA in both the patient’s stool and airway.
The authors say that researchers in China have isolated viral RNA from the stool of two patients (unpublished), and it has been found in saliva, suggesting the possibility of the salivary gland as an infection or transmission route.
The authors maintain that previous studies likely overlooked or neglected patients who had mild intestinal symptoms. “Many efforts should be made to be alert on the initial digestive symptoms of COVID-19 for early detection, early diagnosis, early isolation and early intervention,” the authors wrote.
Like other coronaviruses, it appears that 2019-nCoV infects cells through an interaction between viral transmembrane spike glycoprotein (S-protein) receptor-binding domain, and the cell receptors angiotensin-converting enzyme 2 (ACE-2) and host cellular transmembrane serine protease (TMPRSS). Transcriptome analysis has shown that human lung AT2 cells express ACE-2 and TMPRSS, but esophagus upper and stratified epithelial cells also express both factors, as do stratified epithelial cells and absorptive enterocytes in the ileum and colon.
The researchers call for investigation into ACE-2 fusion proteins and TMPRSS inhibitors for diagnosis, prophylaxis, or treatment of COVID-19.
The authors also noted that COVID-19 has been linked to mild to moderate liver injury as revealed by elevated aminotransferases, hypoproteinemia and prothrombin time prolongation. This also has precedent in that the SARS coronavirus can infect the liver, and biopsies revealed mitoses and apoptosis, along with other abnormalities. SARS-associated hepatitis may be the result of viral hepatitis, immune overreaction, or a secondary effect of antiviral medications or other drugs. Little is known to date about the ability of 2019-nCoV to infect the liver, but single-cell RNA sequencing data from two distinct cohorts showed more ACE-2 expression in cholangiocytes (59.7%) than hepatocytes (2.6%), which indicates that the virus might directly affect intrahepatic bile ducts.
The authors had no sources of funding or financial conflicts.
SOURCE: GU J et al. Gastroenterology. 2020 March 3. doi: 10.1053/j.gastro.2020.02.054.
*This story was updated on 4/10.2020.
FROM GASTROENTEROLOGY
ICH survival lags in the community setting
LOS ANGELES – Although recent findings from circumscribed patient populations enrolled in intervention studies have shown improved survival rates in patients with a recent intracerebral hemorrhagic stroke, data from a large, observational study in the Netherlands suggested a much darker real-world picture, with a 6-month mortality of 64% identified in a total cohort of nearly 15,000 people followed prospectively starting in 1990.
In striking contrast to the survival pattern over time of patients in the same Dutch study who had a first acute ischemic stroke, which showed a statistically significant and meaningful cut in mortality for ischemic stroke patients during the 25-year period examined, survival rates for patients during the first months following a first intracerebral hemorrhage (ICH) stayed flat during 1991-2015, Reem Waziry, MD, said at the International Stroke Conference sponsored by the American Heart Association.
“The promising treatment advances [applied to patients] in the recent ICH trials may not be reflected in community-based treatment,” suggested Dr. Waziry, a research and teaching fellow in clinical epidemiology at the Harvard School of Public Health in Boston.
The data she reported came from the Rotterdam Study, which followed unselected, older people in the Rotterdam community with no stroke history, and during 25 years of monitoring identified 162 incident ICH strokes and 988 acute ischemic strokes. Concurrently with Dr. Waziry’s talk at the conference, the data she reported were published in Stroke. The data she reported also showed that, during the 25 years studied, mortality at 3 years following a first ICH stroke rose to 73% on average.
During her talk, Dr. Waziry also presented an unpublished comparison of the 64% 6-month mortality in the Rotterdam Study with the 3- to 6-month mortality reported in the control arms of four recent, randomized intervention trials, including the MISTIE III trial. Among the four randomized trials Dr. Waziry selected to make this post-hoc comparison, the study with the highest mortality among control patients was MISTIE III, which showed about 25% mortality after 6 months. In contrast, the 19% 6-month mortality among ischemic stroke patients in the Rotterdam Study was roughly similar to the mortality seem in the control arms of some recent studies of interventions for patients with acute ischemic stroke.
The Rotterdam Study receives no commercial funding. Dr. Waziry had no disclosures.
SOURCE: Waziry R et al. ISC 2020, Abstract LB14.
LOS ANGELES – Although recent findings from circumscribed patient populations enrolled in intervention studies have shown improved survival rates in patients with a recent intracerebral hemorrhagic stroke, data from a large, observational study in the Netherlands suggested a much darker real-world picture, with a 6-month mortality of 64% identified in a total cohort of nearly 15,000 people followed prospectively starting in 1990.
In striking contrast to the survival pattern over time of patients in the same Dutch study who had a first acute ischemic stroke, which showed a statistically significant and meaningful cut in mortality for ischemic stroke patients during the 25-year period examined, survival rates for patients during the first months following a first intracerebral hemorrhage (ICH) stayed flat during 1991-2015, Reem Waziry, MD, said at the International Stroke Conference sponsored by the American Heart Association.
“The promising treatment advances [applied to patients] in the recent ICH trials may not be reflected in community-based treatment,” suggested Dr. Waziry, a research and teaching fellow in clinical epidemiology at the Harvard School of Public Health in Boston.
The data she reported came from the Rotterdam Study, which followed unselected, older people in the Rotterdam community with no stroke history, and during 25 years of monitoring identified 162 incident ICH strokes and 988 acute ischemic strokes. Concurrently with Dr. Waziry’s talk at the conference, the data she reported were published in Stroke. The data she reported also showed that, during the 25 years studied, mortality at 3 years following a first ICH stroke rose to 73% on average.
During her talk, Dr. Waziry also presented an unpublished comparison of the 64% 6-month mortality in the Rotterdam Study with the 3- to 6-month mortality reported in the control arms of four recent, randomized intervention trials, including the MISTIE III trial. Among the four randomized trials Dr. Waziry selected to make this post-hoc comparison, the study with the highest mortality among control patients was MISTIE III, which showed about 25% mortality after 6 months. In contrast, the 19% 6-month mortality among ischemic stroke patients in the Rotterdam Study was roughly similar to the mortality seem in the control arms of some recent studies of interventions for patients with acute ischemic stroke.
The Rotterdam Study receives no commercial funding. Dr. Waziry had no disclosures.
SOURCE: Waziry R et al. ISC 2020, Abstract LB14.
LOS ANGELES – Although recent findings from circumscribed patient populations enrolled in intervention studies have shown improved survival rates in patients with a recent intracerebral hemorrhagic stroke, data from a large, observational study in the Netherlands suggested a much darker real-world picture, with a 6-month mortality of 64% identified in a total cohort of nearly 15,000 people followed prospectively starting in 1990.
In striking contrast to the survival pattern over time of patients in the same Dutch study who had a first acute ischemic stroke, which showed a statistically significant and meaningful cut in mortality for ischemic stroke patients during the 25-year period examined, survival rates for patients during the first months following a first intracerebral hemorrhage (ICH) stayed flat during 1991-2015, Reem Waziry, MD, said at the International Stroke Conference sponsored by the American Heart Association.
“The promising treatment advances [applied to patients] in the recent ICH trials may not be reflected in community-based treatment,” suggested Dr. Waziry, a research and teaching fellow in clinical epidemiology at the Harvard School of Public Health in Boston.
The data she reported came from the Rotterdam Study, which followed unselected, older people in the Rotterdam community with no stroke history, and during 25 years of monitoring identified 162 incident ICH strokes and 988 acute ischemic strokes. Concurrently with Dr. Waziry’s talk at the conference, the data she reported were published in Stroke. The data she reported also showed that, during the 25 years studied, mortality at 3 years following a first ICH stroke rose to 73% on average.
During her talk, Dr. Waziry also presented an unpublished comparison of the 64% 6-month mortality in the Rotterdam Study with the 3- to 6-month mortality reported in the control arms of four recent, randomized intervention trials, including the MISTIE III trial. Among the four randomized trials Dr. Waziry selected to make this post-hoc comparison, the study with the highest mortality among control patients was MISTIE III, which showed about 25% mortality after 6 months. In contrast, the 19% 6-month mortality among ischemic stroke patients in the Rotterdam Study was roughly similar to the mortality seem in the control arms of some recent studies of interventions for patients with acute ischemic stroke.
The Rotterdam Study receives no commercial funding. Dr. Waziry had no disclosures.
SOURCE: Waziry R et al. ISC 2020, Abstract LB14.
REPORTING FROM ISC 2020
Get With the Guidelines – Stroke targets ICH
The Get With the Guidelines – Stroke program is finally turning its attention to hemorrhagic strokes after having spurred improved patient management performance from participating U.S. stroke centers since its start in 2003 with a focus on acute ischemic stroke.
The advisers who craft policy for Get With the Guidelines – Stroke (GWTG–S) are planning to launch a pilot program later in 2020 that will initiate data monitoring and quality improvement aimed at optimizing care for patients following an intracerebral hemorrhage (ICH) starting at 15 U.S. stroke centers, with announcement of these 15 participating centers expected later in 2020. The program will start by targeting nine specific, evidence-based, key aspects of the acute management of ICH patients, said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn, and a volunteer expert who is part of the team developing the ICH initiative.
According to Dr. Sheth, the nine imperatives of acute ICH care that the program plans to monitor at participating centers are:
- Obtain a baseline severity score.
- Identify etiology as spontaneous or treatment related.
- Perform coagulopathy reversal or anticoagulant reversal.
- Administer venous thromboembolism prophylaxis.
- Apply dysphagia screening within 24 hours, and delay oral intake until patient passes dysphagia screen.
- Provide patient management in a multidisciplinary stroke or ICU unit.
- Prescribe appropriate blood pressure treatment at discharge.
- Perform assessment for rehabilitation.
- Avoid prescribing corticosteroids and other contraindicated drugs.
GWTG–S is adopting these metrics for assessing the acute care of ICH patients based largely on the recommendations of an expert 2018 panel organized by the American Heart Association and American Stroke. Association that proposed a set of performance measures for the care of ICH patients. This set of performance measures served as the primary basis for designing the new GWTG–S program, along with considerations of feasibility for collecting data on these measures, Dr. Sheth said in an interview. “We hope to make it easy” for centers to collect the data needed to participate.
The existing GWTG–S program is now 17-years old, and has spread to nearly 2,400 U.S. stroke centers as of early 2020, but the time has come to broaden its reach to patients with ICH and the programs that treat these patients, Dr. Sheth said. After years of nihilism about the prospects for patients following an ICH stroke, survival rates have increased, presenting “an opportunity to optimize care, for quality improvement,” he explained. “It’s a huge shift.” ICH patients “do better than we used to think.”
The Get With the Guidelines – Stroke program is finally turning its attention to hemorrhagic strokes after having spurred improved patient management performance from participating U.S. stroke centers since its start in 2003 with a focus on acute ischemic stroke.
The advisers who craft policy for Get With the Guidelines – Stroke (GWTG–S) are planning to launch a pilot program later in 2020 that will initiate data monitoring and quality improvement aimed at optimizing care for patients following an intracerebral hemorrhage (ICH) starting at 15 U.S. stroke centers, with announcement of these 15 participating centers expected later in 2020. The program will start by targeting nine specific, evidence-based, key aspects of the acute management of ICH patients, said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn, and a volunteer expert who is part of the team developing the ICH initiative.
According to Dr. Sheth, the nine imperatives of acute ICH care that the program plans to monitor at participating centers are:
- Obtain a baseline severity score.
- Identify etiology as spontaneous or treatment related.
- Perform coagulopathy reversal or anticoagulant reversal.
- Administer venous thromboembolism prophylaxis.
- Apply dysphagia screening within 24 hours, and delay oral intake until patient passes dysphagia screen.
- Provide patient management in a multidisciplinary stroke or ICU unit.
- Prescribe appropriate blood pressure treatment at discharge.
- Perform assessment for rehabilitation.
- Avoid prescribing corticosteroids and other contraindicated drugs.
GWTG–S is adopting these metrics for assessing the acute care of ICH patients based largely on the recommendations of an expert 2018 panel organized by the American Heart Association and American Stroke. Association that proposed a set of performance measures for the care of ICH patients. This set of performance measures served as the primary basis for designing the new GWTG–S program, along with considerations of feasibility for collecting data on these measures, Dr. Sheth said in an interview. “We hope to make it easy” for centers to collect the data needed to participate.
The existing GWTG–S program is now 17-years old, and has spread to nearly 2,400 U.S. stroke centers as of early 2020, but the time has come to broaden its reach to patients with ICH and the programs that treat these patients, Dr. Sheth said. After years of nihilism about the prospects for patients following an ICH stroke, survival rates have increased, presenting “an opportunity to optimize care, for quality improvement,” he explained. “It’s a huge shift.” ICH patients “do better than we used to think.”
The Get With the Guidelines – Stroke program is finally turning its attention to hemorrhagic strokes after having spurred improved patient management performance from participating U.S. stroke centers since its start in 2003 with a focus on acute ischemic stroke.
The advisers who craft policy for Get With the Guidelines – Stroke (GWTG–S) are planning to launch a pilot program later in 2020 that will initiate data monitoring and quality improvement aimed at optimizing care for patients following an intracerebral hemorrhage (ICH) starting at 15 U.S. stroke centers, with announcement of these 15 participating centers expected later in 2020. The program will start by targeting nine specific, evidence-based, key aspects of the acute management of ICH patients, said Kevin N. Sheth, MD, professor of neurology and neurosurgery, and chief of neurocritical care and emergency neurology at Yale University in New Haven, Conn, and a volunteer expert who is part of the team developing the ICH initiative.
According to Dr. Sheth, the nine imperatives of acute ICH care that the program plans to monitor at participating centers are:
- Obtain a baseline severity score.
- Identify etiology as spontaneous or treatment related.
- Perform coagulopathy reversal or anticoagulant reversal.
- Administer venous thromboembolism prophylaxis.
- Apply dysphagia screening within 24 hours, and delay oral intake until patient passes dysphagia screen.
- Provide patient management in a multidisciplinary stroke or ICU unit.
- Prescribe appropriate blood pressure treatment at discharge.
- Perform assessment for rehabilitation.
- Avoid prescribing corticosteroids and other contraindicated drugs.
GWTG–S is adopting these metrics for assessing the acute care of ICH patients based largely on the recommendations of an expert 2018 panel organized by the American Heart Association and American Stroke. Association that proposed a set of performance measures for the care of ICH patients. This set of performance measures served as the primary basis for designing the new GWTG–S program, along with considerations of feasibility for collecting data on these measures, Dr. Sheth said in an interview. “We hope to make it easy” for centers to collect the data needed to participate.
The existing GWTG–S program is now 17-years old, and has spread to nearly 2,400 U.S. stroke centers as of early 2020, but the time has come to broaden its reach to patients with ICH and the programs that treat these patients, Dr. Sheth said. After years of nihilism about the prospects for patients following an ICH stroke, survival rates have increased, presenting “an opportunity to optimize care, for quality improvement,” he explained. “It’s a huge shift.” ICH patients “do better than we used to think.”


















