User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
COPD patients more likely to be unemployed, less physically active
Current smoking and lack of physical activity among U.S. patients with chronic obstructive pulmonary disease were both linked to limitations on physical activity and inability to work, a new government report showed.
Given those associations, health care professionals should advise COPD patients to participate in pulmonary rehabilitation and a personalized exercise regimen, the Centers for Disease Prevention and Control said.
In a new report, “Employment and Activity Limitations Among Adults with Chronic Obstructive Pulmonary Disease – United States, 2013,” CDC researchers analyzed data from the 2013 Behavioral Risk Factor Surveillance System, a nationwide, self-reported telephone survey. As of 2013, 15.7 million U.S. adults (6.4%) were diagnosed with COPD by a physician.
Adults who reported having COPD were more likely than adults without the disease to report having difficulty walking or climbing stairs (38.4% vs. 11.3%), being unable to work (24.3% vs. 5.3%), and having activity limitation because of health problems (49.6% vs. 16.9%). COPD-related medical costs were estimated at $32 billion in the United States in 2010, and COPD generated an additional $4 billion in absenteeism costs.
The report also noted that smoking was associated with worse symptoms among COPD patients, although more than one-third (38.0%) of adults with COPD were current smokers. COPD was more common among current smokers (14.3%) than former smokers (7.0%) or people who never smoked (2.8%).
Among adults with COPD, nonsmokers who also reported being physically active were least likely to report all of the activity limitations, while those not physically active, regardless of smoking status, were most likely to report the activity limitations.
Read the full article here: MMWR Morb. Mortal. Wkly Rep. 2015;64:289-95.
Current smoking and lack of physical activity among U.S. patients with chronic obstructive pulmonary disease were both linked to limitations on physical activity and inability to work, a new government report showed.
Given those associations, health care professionals should advise COPD patients to participate in pulmonary rehabilitation and a personalized exercise regimen, the Centers for Disease Prevention and Control said.
In a new report, “Employment and Activity Limitations Among Adults with Chronic Obstructive Pulmonary Disease – United States, 2013,” CDC researchers analyzed data from the 2013 Behavioral Risk Factor Surveillance System, a nationwide, self-reported telephone survey. As of 2013, 15.7 million U.S. adults (6.4%) were diagnosed with COPD by a physician.
Adults who reported having COPD were more likely than adults without the disease to report having difficulty walking or climbing stairs (38.4% vs. 11.3%), being unable to work (24.3% vs. 5.3%), and having activity limitation because of health problems (49.6% vs. 16.9%). COPD-related medical costs were estimated at $32 billion in the United States in 2010, and COPD generated an additional $4 billion in absenteeism costs.
The report also noted that smoking was associated with worse symptoms among COPD patients, although more than one-third (38.0%) of adults with COPD were current smokers. COPD was more common among current smokers (14.3%) than former smokers (7.0%) or people who never smoked (2.8%).
Among adults with COPD, nonsmokers who also reported being physically active were least likely to report all of the activity limitations, while those not physically active, regardless of smoking status, were most likely to report the activity limitations.
Read the full article here: MMWR Morb. Mortal. Wkly Rep. 2015;64:289-95.
Current smoking and lack of physical activity among U.S. patients with chronic obstructive pulmonary disease were both linked to limitations on physical activity and inability to work, a new government report showed.
Given those associations, health care professionals should advise COPD patients to participate in pulmonary rehabilitation and a personalized exercise regimen, the Centers for Disease Prevention and Control said.
In a new report, “Employment and Activity Limitations Among Adults with Chronic Obstructive Pulmonary Disease – United States, 2013,” CDC researchers analyzed data from the 2013 Behavioral Risk Factor Surveillance System, a nationwide, self-reported telephone survey. As of 2013, 15.7 million U.S. adults (6.4%) were diagnosed with COPD by a physician.
Adults who reported having COPD were more likely than adults without the disease to report having difficulty walking or climbing stairs (38.4% vs. 11.3%), being unable to work (24.3% vs. 5.3%), and having activity limitation because of health problems (49.6% vs. 16.9%). COPD-related medical costs were estimated at $32 billion in the United States in 2010, and COPD generated an additional $4 billion in absenteeism costs.
The report also noted that smoking was associated with worse symptoms among COPD patients, although more than one-third (38.0%) of adults with COPD were current smokers. COPD was more common among current smokers (14.3%) than former smokers (7.0%) or people who never smoked (2.8%).
Among adults with COPD, nonsmokers who also reported being physically active were least likely to report all of the activity limitations, while those not physically active, regardless of smoking status, were most likely to report the activity limitations.
Read the full article here: MMWR Morb. Mortal. Wkly Rep. 2015;64:289-95.
Consider cephalosporin a safe alternative for patients with penicillin allergy
HOUSTON– Given the low incidence of adverse drug reactions to cephalosporin antibiotics among nearly a million California health plan patients, patients with a history of penicillin allergy can safely be given cephalosporins, according to Dr. Eric M. Macy.
The recommendation is based on the findings of a retrospective, population-based analysis of the records of 949,323 Kaiser Permanente Southern California health plan members, which was presented by Dr. Macy, of the Kaiser Permanente Medical Center in San Diego, at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Dr. Macy and his colleagues examined the records of 622,456 health plan members who were given 901,908 regimens of oral cephalosporins and 326,867 members given 487,630 parenteral cephalosporin regimens between Jan. 1, 2010, and Dec. 31, 2012.
Clostridium difficile infection within 90 days, nephropathy within 30 days, and all-cause death within 1 day were the most common cephalosporin-associated adverse drug reactions (ADRs) reported by physicians, with rates of 0.91%, 0.15%, and 0.10%, respectively. Cephalosporin-associated anaphylaxis was documented by physicians a total of five times in oral patients and eight times in parenteral patients (P = .0761), while only three serious cutaneous adverse reactions (SCARs) were recorded. All of those SCARS were attributable to other antibiotics taken at the same time as the cephalosporins, according to Dr. Macy.
Patients who reported developing new cephalosporin allergies over the course of the study period were significantly more likely to be female than male: 0.56% vs. 0.43% (P < .0001). And patients with a history of penicillin allergy were more likely to report a new cephalosporin allergy within 30 days than patients with no drug allergy, another cephalosporin allergy, or a non–beta-lactam allergy.
The numbers are smaller than those reported recently in the American Journal of Medicine (2009;122: 778.e1–7), Dr. Macy noted, “because there it was new allergy reports in the year after any cephalosporin exposure, as opposed to our new report, which looks at reactions within 30 days of exposure.”
Dr. Macy disclosed that he has received research support from ALK and BioMarin.
HOUSTON– Given the low incidence of adverse drug reactions to cephalosporin antibiotics among nearly a million California health plan patients, patients with a history of penicillin allergy can safely be given cephalosporins, according to Dr. Eric M. Macy.
The recommendation is based on the findings of a retrospective, population-based analysis of the records of 949,323 Kaiser Permanente Southern California health plan members, which was presented by Dr. Macy, of the Kaiser Permanente Medical Center in San Diego, at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Dr. Macy and his colleagues examined the records of 622,456 health plan members who were given 901,908 regimens of oral cephalosporins and 326,867 members given 487,630 parenteral cephalosporin regimens between Jan. 1, 2010, and Dec. 31, 2012.
Clostridium difficile infection within 90 days, nephropathy within 30 days, and all-cause death within 1 day were the most common cephalosporin-associated adverse drug reactions (ADRs) reported by physicians, with rates of 0.91%, 0.15%, and 0.10%, respectively. Cephalosporin-associated anaphylaxis was documented by physicians a total of five times in oral patients and eight times in parenteral patients (P = .0761), while only three serious cutaneous adverse reactions (SCARs) were recorded. All of those SCARS were attributable to other antibiotics taken at the same time as the cephalosporins, according to Dr. Macy.
Patients who reported developing new cephalosporin allergies over the course of the study period were significantly more likely to be female than male: 0.56% vs. 0.43% (P < .0001). And patients with a history of penicillin allergy were more likely to report a new cephalosporin allergy within 30 days than patients with no drug allergy, another cephalosporin allergy, or a non–beta-lactam allergy.
The numbers are smaller than those reported recently in the American Journal of Medicine (2009;122: 778.e1–7), Dr. Macy noted, “because there it was new allergy reports in the year after any cephalosporin exposure, as opposed to our new report, which looks at reactions within 30 days of exposure.”
Dr. Macy disclosed that he has received research support from ALK and BioMarin.
HOUSTON– Given the low incidence of adverse drug reactions to cephalosporin antibiotics among nearly a million California health plan patients, patients with a history of penicillin allergy can safely be given cephalosporins, according to Dr. Eric M. Macy.
The recommendation is based on the findings of a retrospective, population-based analysis of the records of 949,323 Kaiser Permanente Southern California health plan members, which was presented by Dr. Macy, of the Kaiser Permanente Medical Center in San Diego, at the annual meeting of the American Academy of Allergy, Asthma, and Immunology.
Dr. Macy and his colleagues examined the records of 622,456 health plan members who were given 901,908 regimens of oral cephalosporins and 326,867 members given 487,630 parenteral cephalosporin regimens between Jan. 1, 2010, and Dec. 31, 2012.
Clostridium difficile infection within 90 days, nephropathy within 30 days, and all-cause death within 1 day were the most common cephalosporin-associated adverse drug reactions (ADRs) reported by physicians, with rates of 0.91%, 0.15%, and 0.10%, respectively. Cephalosporin-associated anaphylaxis was documented by physicians a total of five times in oral patients and eight times in parenteral patients (P = .0761), while only three serious cutaneous adverse reactions (SCARs) were recorded. All of those SCARS were attributable to other antibiotics taken at the same time as the cephalosporins, according to Dr. Macy.
Patients who reported developing new cephalosporin allergies over the course of the study period were significantly more likely to be female than male: 0.56% vs. 0.43% (P < .0001). And patients with a history of penicillin allergy were more likely to report a new cephalosporin allergy within 30 days than patients with no drug allergy, another cephalosporin allergy, or a non–beta-lactam allergy.
The numbers are smaller than those reported recently in the American Journal of Medicine (2009;122: 778.e1–7), Dr. Macy noted, “because there it was new allergy reports in the year after any cephalosporin exposure, as opposed to our new report, which looks at reactions within 30 days of exposure.”
Dr. Macy disclosed that he has received research support from ALK and BioMarin.
AT THE 2015 AAAAI ANNUAL MEETING
Key clinical point: Given the low risk of adverse drug reactions with cephalosporins, patients with a history of penicillin allergy can safely take cephalosporins.
Major finding: The most frequent ADRs in patients taking either oral or parenteral cephalosporins were Clostridium difficile infection within 90 days (0.91%), nephropathy within 30 days (0.15%), and all-cause death within 1 day (0.10%).
Data source: Retrospective, population-based analysis of 949,323 Kaiser Permanente Southern California health plan members from 2010 to 2012.
Disclosures: Dr. Macy disclosed receiving research support from ALK and BioMarin.
Ebola vaccines pass phase II trial testing in Liberia
Two experimental Ebola vaccines seem to be safe after a phase II clinical trial undertaken in Liberia, according to a press release from the National Institutes of Health.
The Partnership for Research on Ebola Vaccines in Liberia (PREVAIL) trial began in early February 2015 in Monrovia to test two vaccines: cAd3-EBOZ and VSV-ZEBOV. While the initial trial goal has been met, phase II trials are continuing at another Monrovia hospital to boost the number of participants from 600 to 1,500.
While the study sponsor, the National Institute of Allergy and Infectious Diseases, was initially planning to conduct the phase III trial portion in Liberia, the country has seen only one case of Ebola since mid-February. The NIAID has determined that phase III studies could be moved to other countries, and discussions are underway to explore that possibility.
“We must move forward to adapt and expand the study so that ultimately we can determine whether these experimental vaccines can protect against Ebola virus disease and therefore be used in future Ebola outbreaks,” Dr. Anthony S. Fauci, director of the NIAID, said in the press release.
Read the full press release on the NIH website.
Two experimental Ebola vaccines seem to be safe after a phase II clinical trial undertaken in Liberia, according to a press release from the National Institutes of Health.
The Partnership for Research on Ebola Vaccines in Liberia (PREVAIL) trial began in early February 2015 in Monrovia to test two vaccines: cAd3-EBOZ and VSV-ZEBOV. While the initial trial goal has been met, phase II trials are continuing at another Monrovia hospital to boost the number of participants from 600 to 1,500.
While the study sponsor, the National Institute of Allergy and Infectious Diseases, was initially planning to conduct the phase III trial portion in Liberia, the country has seen only one case of Ebola since mid-February. The NIAID has determined that phase III studies could be moved to other countries, and discussions are underway to explore that possibility.
“We must move forward to adapt and expand the study so that ultimately we can determine whether these experimental vaccines can protect against Ebola virus disease and therefore be used in future Ebola outbreaks,” Dr. Anthony S. Fauci, director of the NIAID, said in the press release.
Read the full press release on the NIH website.
Two experimental Ebola vaccines seem to be safe after a phase II clinical trial undertaken in Liberia, according to a press release from the National Institutes of Health.
The Partnership for Research on Ebola Vaccines in Liberia (PREVAIL) trial began in early February 2015 in Monrovia to test two vaccines: cAd3-EBOZ and VSV-ZEBOV. While the initial trial goal has been met, phase II trials are continuing at another Monrovia hospital to boost the number of participants from 600 to 1,500.
While the study sponsor, the National Institute of Allergy and Infectious Diseases, was initially planning to conduct the phase III trial portion in Liberia, the country has seen only one case of Ebola since mid-February. The NIAID has determined that phase III studies could be moved to other countries, and discussions are underway to explore that possibility.
“We must move forward to adapt and expand the study so that ultimately we can determine whether these experimental vaccines can protect against Ebola virus disease and therefore be used in future Ebola outbreaks,” Dr. Anthony S. Fauci, director of the NIAID, said in the press release.
Read the full press release on the NIH website.
Catheter treatment for PAH outperforms medication
SAN DIEGO – Percutaneous pulmonary artery denervation for the treatment of pulmonary arterial hypertension safely resulted in significantly greater improvement in functional capacity and hemodynamics compared with medication, in a controlled before-and-after study.
A particularly noteworthy secondary finding in the study was that rehospitalizations during the first 6 months after pulmonary artery denervation (PADN) occurred just one-third as frequently as in the 6-month preprocedural period on standard medications, Dr. Shao-Liang Chen said at the annual meeting of the American College of Cardiology.
He and his coinvestigators, including Dr. Gregg W. Stone of Columbia University in New York, developed a percutaneous catheter-based method of destroying the pulmonary baroreceptor structure located at the bifurcation area of the middle pulmonary artery. Along the way, they redefined the understanding of the pathogenesis of pulmonary artery hypertension (PAH) by demonstrating that local sympathetic nerve activity plays a pivotal role in modulating the elevations of mean pulmonary artery pressure (mPAP) and pulmonary vascular resistance (PVR), which are the disease hallmarks.
Dr. Chen and coinvestigators previously reported the first-in-man study of PADN, which demonstrated safety and short-term efficacy (J. Am. Coll. Cardiol. 2013;62:1092-100). At ACC 15, Dr. Chen presented the findings of the new PADN-2 study, which expands upon the first study by including more patients and longer and more comprehensive follow-up.
The study comprised 28 patients with PAH, including 11 with idiopathic PAH and 8 with pulmonary hypertension caused by left ventricular disease. All of them underwent medication washout followed by right heart catheterization and echocardiography for baseline off-drug hemodynamic measurements as well as a 6-minute walk distance test of their functional capacity. Then they went back on medications for 6 months, after which they underwent repeat testing. Then their medications were discontinued and they underwent PADN. Six months after the procedure, still off medications, they were retested once again.
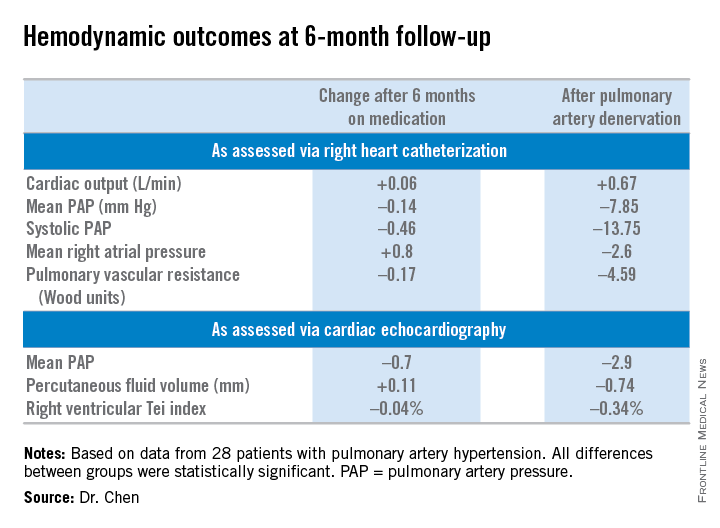
The primary study endpoint was change in 6-minute walk distance. After 6 months of medication it improved from 361 to 373 meters, a modest 3.9% gain over off-drug baseline. In contrast, 6-minute walk distance grew from 358 to 423 meters 6 months after PADN, a clinically important 23.9% improvement, reported Dr. Chen, a cardiologist at First Hospital of Nanjing (China) Medical University.
Multiple secondary hemodynamic endpoints also showed significantly greater improvement with PADN than medical therapy.
Twelve predefined clinical events – mostly involving worsening PAH – occurred during medical management, compared with three in the 6 months following PADN.
In addition, there were 12 hospitalizations during the 6 months on medical management compared with only 4 after the same patients underwent PADN. Health care costs averaged $35,000 per patient during the 6-month study period on medication compared with $6,000 per patient in the first 6 months after PADN.
There were no deaths, aneurysms, access site hematomas, or thrombotic events during either study period.
Further randomized, controlled trials are planned to explore the possibility that the benefits seen in the PADN-2 trial will result in reduced mortality in patients with PAH, according to Dr. Chen.
The PADN-2 trial was sponsored by Nanjing Medical University. Dr. Chen reported serving as a consultant to MicroPort.
SAN DIEGO – Percutaneous pulmonary artery denervation for the treatment of pulmonary arterial hypertension safely resulted in significantly greater improvement in functional capacity and hemodynamics compared with medication, in a controlled before-and-after study.
A particularly noteworthy secondary finding in the study was that rehospitalizations during the first 6 months after pulmonary artery denervation (PADN) occurred just one-third as frequently as in the 6-month preprocedural period on standard medications, Dr. Shao-Liang Chen said at the annual meeting of the American College of Cardiology.

He and his coinvestigators, including Dr. Gregg W. Stone of Columbia University in New York, developed a percutaneous catheter-based method of destroying the pulmonary baroreceptor structure located at the bifurcation area of the middle pulmonary artery. Along the way, they redefined the understanding of the pathogenesis of pulmonary artery hypertension (PAH) by demonstrating that local sympathetic nerve activity plays a pivotal role in modulating the elevations of mean pulmonary artery pressure (mPAP) and pulmonary vascular resistance (PVR), which are the disease hallmarks.
Dr. Chen and coinvestigators previously reported the first-in-man study of PADN, which demonstrated safety and short-term efficacy (J. Am. Coll. Cardiol. 2013;62:1092-100). At ACC 15, Dr. Chen presented the findings of the new PADN-2 study, which expands upon the first study by including more patients and longer and more comprehensive follow-up.
The study comprised 28 patients with PAH, including 11 with idiopathic PAH and 8 with pulmonary hypertension caused by left ventricular disease. All of them underwent medication washout followed by right heart catheterization and echocardiography for baseline off-drug hemodynamic measurements as well as a 6-minute walk distance test of their functional capacity. Then they went back on medications for 6 months, after which they underwent repeat testing. Then their medications were discontinued and they underwent PADN. Six months after the procedure, still off medications, they were retested once again.
The primary study endpoint was change in 6-minute walk distance. After 6 months of medication it improved from 361 to 373 meters, a modest 3.9% gain over off-drug baseline. In contrast, 6-minute walk distance grew from 358 to 423 meters 6 months after PADN, a clinically important 23.9% improvement, reported Dr. Chen, a cardiologist at First Hospital of Nanjing (China) Medical University.
Multiple secondary hemodynamic endpoints also showed significantly greater improvement with PADN than medical therapy.
Twelve predefined clinical events – mostly involving worsening PAH – occurred during medical management, compared with three in the 6 months following PADN.
In addition, there were 12 hospitalizations during the 6 months on medical management compared with only 4 after the same patients underwent PADN. Health care costs averaged $35,000 per patient during the 6-month study period on medication compared with $6,000 per patient in the first 6 months after PADN.
There were no deaths, aneurysms, access site hematomas, or thrombotic events during either study period.
Further randomized, controlled trials are planned to explore the possibility that the benefits seen in the PADN-2 trial will result in reduced mortality in patients with PAH, according to Dr. Chen.
The PADN-2 trial was sponsored by Nanjing Medical University. Dr. Chen reported serving as a consultant to MicroPort.
SAN DIEGO – Percutaneous pulmonary artery denervation for the treatment of pulmonary arterial hypertension safely resulted in significantly greater improvement in functional capacity and hemodynamics compared with medication, in a controlled before-and-after study.
A particularly noteworthy secondary finding in the study was that rehospitalizations during the first 6 months after pulmonary artery denervation (PADN) occurred just one-third as frequently as in the 6-month preprocedural period on standard medications, Dr. Shao-Liang Chen said at the annual meeting of the American College of Cardiology.

He and his coinvestigators, including Dr. Gregg W. Stone of Columbia University in New York, developed a percutaneous catheter-based method of destroying the pulmonary baroreceptor structure located at the bifurcation area of the middle pulmonary artery. Along the way, they redefined the understanding of the pathogenesis of pulmonary artery hypertension (PAH) by demonstrating that local sympathetic nerve activity plays a pivotal role in modulating the elevations of mean pulmonary artery pressure (mPAP) and pulmonary vascular resistance (PVR), which are the disease hallmarks.
Dr. Chen and coinvestigators previously reported the first-in-man study of PADN, which demonstrated safety and short-term efficacy (J. Am. Coll. Cardiol. 2013;62:1092-100). At ACC 15, Dr. Chen presented the findings of the new PADN-2 study, which expands upon the first study by including more patients and longer and more comprehensive follow-up.
The study comprised 28 patients with PAH, including 11 with idiopathic PAH and 8 with pulmonary hypertension caused by left ventricular disease. All of them underwent medication washout followed by right heart catheterization and echocardiography for baseline off-drug hemodynamic measurements as well as a 6-minute walk distance test of their functional capacity. Then they went back on medications for 6 months, after which they underwent repeat testing. Then their medications were discontinued and they underwent PADN. Six months after the procedure, still off medications, they were retested once again.
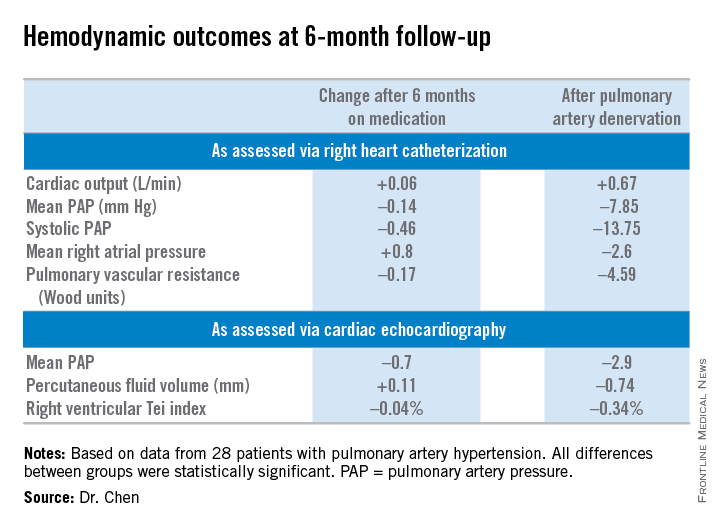
The primary study endpoint was change in 6-minute walk distance. After 6 months of medication it improved from 361 to 373 meters, a modest 3.9% gain over off-drug baseline. In contrast, 6-minute walk distance grew from 358 to 423 meters 6 months after PADN, a clinically important 23.9% improvement, reported Dr. Chen, a cardiologist at First Hospital of Nanjing (China) Medical University.
Multiple secondary hemodynamic endpoints also showed significantly greater improvement with PADN than medical therapy.
Twelve predefined clinical events – mostly involving worsening PAH – occurred during medical management, compared with three in the 6 months following PADN.
In addition, there were 12 hospitalizations during the 6 months on medical management compared with only 4 after the same patients underwent PADN. Health care costs averaged $35,000 per patient during the 6-month study period on medication compared with $6,000 per patient in the first 6 months after PADN.
There were no deaths, aneurysms, access site hematomas, or thrombotic events during either study period.
Further randomized, controlled trials are planned to explore the possibility that the benefits seen in the PADN-2 trial will result in reduced mortality in patients with PAH, according to Dr. Chen.
The PADN-2 trial was sponsored by Nanjing Medical University. Dr. Chen reported serving as a consultant to MicroPort.
AT ACC 15
Key clinical point: Pulmonary artery denervation for PAH showed promise in a small study.
Major finding: Six-minute walk distance improved by 3.9% after 6 months of medication, compared with 23.9% in testing done 6 months after percutaneous pulmonary artery denervation.
Data source: The PADN-2 trial, a prospective before-and-after study of 28 patients with pulmonary arterial hypertension.
Disclosures: Nanjing Medical University funded the study. The presenter reported serving as a consultant to MicroPort.
Untreated OSA upped risk of need for repeat revascularization after PCI
Patients with untreated, moderate to severe obstructive sleep apnea who underwent percutaneous coronary intervention were more than twice as likely to undergo repeat revascularization during the next 4.8 years, compared with patients on continuous positive airway pressure (CPAP), researchers reported in the March issue of Chest.
The first-in-kind finding “provides new evidence that untreated moderate-severe OSA [obstructive sleep apnea] is an independent risk factor for repeat revascularization after PCI [percutaneous coronary intervention] and that CPAP can reduce this risk.,” said Dr. Xiaofan Wu at the Beijing Anzhen Hospital at Capital Medical University in Beijing and her associates. “Interestingly, the data show that untreated mild OSA was not associated with an increased risk of repeat revascularization, suggesting a dose-effect relationship between OSA severity and risk of complications after PCI.”
Untreated OSA has been linked with many cardiovascular problems. Patients have high levels of sympathetic excitation, oxidative stress, inflammatory mediators, endothelial dysfunction, and attenuated endothelial repair, which all can promote atherogenesis, hypertension, arrhythmogenesis, and cardiac death, the researchers noted. But the effect of untreated OSA on PCI outcomes was not well understood, they said (Chest 2015;147:708-18).
Their study retrospectively followed 390 patients with OSA who had undergone PCI. The cohorts included 128 patients with moderate to severe OSA that was successfully treated with CPAP, 167 patients with untreated, moderate to severe OSA, and 95 patients with untreated mild OSA. The investigators used subjective patient reports to assess adherence to CPAP. In all, 83.6% of treated patients had used CPAP for at least 6 months, and the rest had used CPAP for 3-6 months, they said.
Over a median follow-up of 4.8 years, 25.1% of patients with untreated, moderate to severe OSA underwent repeat revascularization, compared with 14.1% of patients on CPAP for similarly severe OSA (P = .019), the investigators reported. In the adjusted analysis, untreated patients had more than double the likelihood of repeat revascularization during the follow-up period (hazard ratio, 2.13; 95% confidence interval, 1.19-3.81; P = .011).
Mortality and rates of major adverse cardiac and cerebrovascular events were similar among the groups, said the researchers. “Although untreated moderate to severe OSA was not associated with an increased risk of death in this cohort, we believe that timely diagnosis and treatment in patients undergoing PCI can serve as a clinically relevant method of secondary prevention to decrease the risk of repeat revascularization,” they said.
The work was funded by the Program for New Century Excellent Talents in University from the Ministry of Education of China, National Natural Science Foundation of China, the Capital Health Research and Development Fund of China, and National Institutes of Health. One coauthor reported relationships with Koninklijke Philips NV and Philips/Respironics. The other authors declared no relevant conflicts of interest.
Patients with untreated, moderate to severe obstructive sleep apnea who underwent percutaneous coronary intervention were more than twice as likely to undergo repeat revascularization during the next 4.8 years, compared with patients on continuous positive airway pressure (CPAP), researchers reported in the March issue of Chest.
The first-in-kind finding “provides new evidence that untreated moderate-severe OSA [obstructive sleep apnea] is an independent risk factor for repeat revascularization after PCI [percutaneous coronary intervention] and that CPAP can reduce this risk.,” said Dr. Xiaofan Wu at the Beijing Anzhen Hospital at Capital Medical University in Beijing and her associates. “Interestingly, the data show that untreated mild OSA was not associated with an increased risk of repeat revascularization, suggesting a dose-effect relationship between OSA severity and risk of complications after PCI.”
Untreated OSA has been linked with many cardiovascular problems. Patients have high levels of sympathetic excitation, oxidative stress, inflammatory mediators, endothelial dysfunction, and attenuated endothelial repair, which all can promote atherogenesis, hypertension, arrhythmogenesis, and cardiac death, the researchers noted. But the effect of untreated OSA on PCI outcomes was not well understood, they said (Chest 2015;147:708-18).
Their study retrospectively followed 390 patients with OSA who had undergone PCI. The cohorts included 128 patients with moderate to severe OSA that was successfully treated with CPAP, 167 patients with untreated, moderate to severe OSA, and 95 patients with untreated mild OSA. The investigators used subjective patient reports to assess adherence to CPAP. In all, 83.6% of treated patients had used CPAP for at least 6 months, and the rest had used CPAP for 3-6 months, they said.
Over a median follow-up of 4.8 years, 25.1% of patients with untreated, moderate to severe OSA underwent repeat revascularization, compared with 14.1% of patients on CPAP for similarly severe OSA (P = .019), the investigators reported. In the adjusted analysis, untreated patients had more than double the likelihood of repeat revascularization during the follow-up period (hazard ratio, 2.13; 95% confidence interval, 1.19-3.81; P = .011).
Mortality and rates of major adverse cardiac and cerebrovascular events were similar among the groups, said the researchers. “Although untreated moderate to severe OSA was not associated with an increased risk of death in this cohort, we believe that timely diagnosis and treatment in patients undergoing PCI can serve as a clinically relevant method of secondary prevention to decrease the risk of repeat revascularization,” they said.
The work was funded by the Program for New Century Excellent Talents in University from the Ministry of Education of China, National Natural Science Foundation of China, the Capital Health Research and Development Fund of China, and National Institutes of Health. One coauthor reported relationships with Koninklijke Philips NV and Philips/Respironics. The other authors declared no relevant conflicts of interest.
Patients with untreated, moderate to severe obstructive sleep apnea who underwent percutaneous coronary intervention were more than twice as likely to undergo repeat revascularization during the next 4.8 years, compared with patients on continuous positive airway pressure (CPAP), researchers reported in the March issue of Chest.
The first-in-kind finding “provides new evidence that untreated moderate-severe OSA [obstructive sleep apnea] is an independent risk factor for repeat revascularization after PCI [percutaneous coronary intervention] and that CPAP can reduce this risk.,” said Dr. Xiaofan Wu at the Beijing Anzhen Hospital at Capital Medical University in Beijing and her associates. “Interestingly, the data show that untreated mild OSA was not associated with an increased risk of repeat revascularization, suggesting a dose-effect relationship between OSA severity and risk of complications after PCI.”
Untreated OSA has been linked with many cardiovascular problems. Patients have high levels of sympathetic excitation, oxidative stress, inflammatory mediators, endothelial dysfunction, and attenuated endothelial repair, which all can promote atherogenesis, hypertension, arrhythmogenesis, and cardiac death, the researchers noted. But the effect of untreated OSA on PCI outcomes was not well understood, they said (Chest 2015;147:708-18).
Their study retrospectively followed 390 patients with OSA who had undergone PCI. The cohorts included 128 patients with moderate to severe OSA that was successfully treated with CPAP, 167 patients with untreated, moderate to severe OSA, and 95 patients with untreated mild OSA. The investigators used subjective patient reports to assess adherence to CPAP. In all, 83.6% of treated patients had used CPAP for at least 6 months, and the rest had used CPAP for 3-6 months, they said.
Over a median follow-up of 4.8 years, 25.1% of patients with untreated, moderate to severe OSA underwent repeat revascularization, compared with 14.1% of patients on CPAP for similarly severe OSA (P = .019), the investigators reported. In the adjusted analysis, untreated patients had more than double the likelihood of repeat revascularization during the follow-up period (hazard ratio, 2.13; 95% confidence interval, 1.19-3.81; P = .011).
Mortality and rates of major adverse cardiac and cerebrovascular events were similar among the groups, said the researchers. “Although untreated moderate to severe OSA was not associated with an increased risk of death in this cohort, we believe that timely diagnosis and treatment in patients undergoing PCI can serve as a clinically relevant method of secondary prevention to decrease the risk of repeat revascularization,” they said.
The work was funded by the Program for New Century Excellent Talents in University from the Ministry of Education of China, National Natural Science Foundation of China, the Capital Health Research and Development Fund of China, and National Institutes of Health. One coauthor reported relationships with Koninklijke Philips NV and Philips/Respironics. The other authors declared no relevant conflicts of interest.
Key clinical point: Untreated, moderate to severe sleep apnea significantly increased the risk of repeat revascularization after percutaneous coronary intervention.
Major finding: Untreated patients had more than double the likelihood of repeat revascularization during the follow-up period (HR, 2.13; 95% CI, 1.19-3.81; P = .011).
Data source: Retrospective observational study of 390 adults with OSA who had undergone PCI.
Disclosures: The work was supported by the Program for New Century Excellent Talents in University from the Ministry of Education of China, National Natural Science Foundation of China, the Capital Health Research and Development Fund of China, and National Institutes of Health. One coauthor reported relationships with Koninklijke Philips NV and Philips/Respironics. The other authors declared no relevant conflicts of interest.
Automated scoring from home sleep test monitors misclassified OSA severity
Automated scoring data from two type-3 home sleep test monitors misclassified the severity of obstructive sleep apnea, especially in patients with mild to moderate disease, researchers reported in the March issue of Chest.
“Agreement between automated and manual scoring of home sleep tests varies as a function of the portable device and definition of disordered breathing event used,” said Dr. Nisha Aurora and her associates at the Johns Hopkins University in Baltimore. “Although modest agreement exists between automated and manual scoring, input by a sleep specialist or a certified polysomnologist in reviewing home sleep study results may help to improve diagnostic accuracy and classification of OSA [obstructive sleep apnea], particularly if there is mild disease.”
About 10%-15% of the general population has OSA, and home sleep testing has become an important part of ambulatory care for the condition. The American Academy of Sleep Medicine recommends that raw data from home sleep test devices be reviewed by either a board-certified sleep specialist or a clinician who fulfills eligibility criteria for the sleep medicine certification examination. Few studies, however, have actually compared automated and manual scoring data from these devices, the researchers said (Chest 2015;147:719-27).
For their analysis, Dr. Aurora and her associates evaluated automated and manual scoring data for 200 patients without a previous OSA diagnosis who used one of two home sleep test monitors: the ApneaLink Plus monitor from ResMed or the Embletta device from Embla Systems. To assess the apnea-hypopnea index (AHI), the researchers defined disordered-breathing events based on both the >3% and >4% thresholds for oxygen desaturation.
Automated scoring consistently underscored disordered-breathing events, regardless of which threshold was used to define AHI, the researchers found. For the ApneaLink Plus monitor, the average difference in AHI between manual and automated scoring was 6.1 events per hour (95% confidence interval, 4.9-7.3) for the 3% threshold and 4.6 events per hour (95% CI, 3.5-5.6) for the 4% threshold, they reported. For the Embletta monitor, manual and automated scoring varied by an average of 5.3 (95% CI, 3.2-7.3) and 8.4 (95% CI, 7.2-9.6) events per hour, respectively.
The difference between automated and manual scoring narrowed substantially when considering oxygen desaturation index instead of AHI, the researchers reported.
The investigators then used AHI to assess OSA severity. For the ApneaLink Plus monitor, relying on automated readings led to a <10% rate of clinically significant misclassification of OSA severity, regardless which oxygen desaturation threshold was used. For the Embletta device, rates of clinically significant misclassification were 29% and 41% for the 3% and 4% thresholds, respectively. “As expected, disease misclassification was most notable for subjects with mild disease irrespective of the device used,” they wrote.
The National Institutes of Health funded the work. One coauthor reported relationships with ResMed and Koninklijke Philips NV (Respironics). The other authors declared no relevant conflicts of interest.
Automated scoring data from two type-3 home sleep test monitors misclassified the severity of obstructive sleep apnea, especially in patients with mild to moderate disease, researchers reported in the March issue of Chest.
“Agreement between automated and manual scoring of home sleep tests varies as a function of the portable device and definition of disordered breathing event used,” said Dr. Nisha Aurora and her associates at the Johns Hopkins University in Baltimore. “Although modest agreement exists between automated and manual scoring, input by a sleep specialist or a certified polysomnologist in reviewing home sleep study results may help to improve diagnostic accuracy and classification of OSA [obstructive sleep apnea], particularly if there is mild disease.”
About 10%-15% of the general population has OSA, and home sleep testing has become an important part of ambulatory care for the condition. The American Academy of Sleep Medicine recommends that raw data from home sleep test devices be reviewed by either a board-certified sleep specialist or a clinician who fulfills eligibility criteria for the sleep medicine certification examination. Few studies, however, have actually compared automated and manual scoring data from these devices, the researchers said (Chest 2015;147:719-27).
For their analysis, Dr. Aurora and her associates evaluated automated and manual scoring data for 200 patients without a previous OSA diagnosis who used one of two home sleep test monitors: the ApneaLink Plus monitor from ResMed or the Embletta device from Embla Systems. To assess the apnea-hypopnea index (AHI), the researchers defined disordered-breathing events based on both the >3% and >4% thresholds for oxygen desaturation.
Automated scoring consistently underscored disordered-breathing events, regardless of which threshold was used to define AHI, the researchers found. For the ApneaLink Plus monitor, the average difference in AHI between manual and automated scoring was 6.1 events per hour (95% confidence interval, 4.9-7.3) for the 3% threshold and 4.6 events per hour (95% CI, 3.5-5.6) for the 4% threshold, they reported. For the Embletta monitor, manual and automated scoring varied by an average of 5.3 (95% CI, 3.2-7.3) and 8.4 (95% CI, 7.2-9.6) events per hour, respectively.
The difference between automated and manual scoring narrowed substantially when considering oxygen desaturation index instead of AHI, the researchers reported.
The investigators then used AHI to assess OSA severity. For the ApneaLink Plus monitor, relying on automated readings led to a <10% rate of clinically significant misclassification of OSA severity, regardless which oxygen desaturation threshold was used. For the Embletta device, rates of clinically significant misclassification were 29% and 41% for the 3% and 4% thresholds, respectively. “As expected, disease misclassification was most notable for subjects with mild disease irrespective of the device used,” they wrote.
The National Institutes of Health funded the work. One coauthor reported relationships with ResMed and Koninklijke Philips NV (Respironics). The other authors declared no relevant conflicts of interest.
Automated scoring data from two type-3 home sleep test monitors misclassified the severity of obstructive sleep apnea, especially in patients with mild to moderate disease, researchers reported in the March issue of Chest.
“Agreement between automated and manual scoring of home sleep tests varies as a function of the portable device and definition of disordered breathing event used,” said Dr. Nisha Aurora and her associates at the Johns Hopkins University in Baltimore. “Although modest agreement exists between automated and manual scoring, input by a sleep specialist or a certified polysomnologist in reviewing home sleep study results may help to improve diagnostic accuracy and classification of OSA [obstructive sleep apnea], particularly if there is mild disease.”
About 10%-15% of the general population has OSA, and home sleep testing has become an important part of ambulatory care for the condition. The American Academy of Sleep Medicine recommends that raw data from home sleep test devices be reviewed by either a board-certified sleep specialist or a clinician who fulfills eligibility criteria for the sleep medicine certification examination. Few studies, however, have actually compared automated and manual scoring data from these devices, the researchers said (Chest 2015;147:719-27).
For their analysis, Dr. Aurora and her associates evaluated automated and manual scoring data for 200 patients without a previous OSA diagnosis who used one of two home sleep test monitors: the ApneaLink Plus monitor from ResMed or the Embletta device from Embla Systems. To assess the apnea-hypopnea index (AHI), the researchers defined disordered-breathing events based on both the >3% and >4% thresholds for oxygen desaturation.
Automated scoring consistently underscored disordered-breathing events, regardless of which threshold was used to define AHI, the researchers found. For the ApneaLink Plus monitor, the average difference in AHI between manual and automated scoring was 6.1 events per hour (95% confidence interval, 4.9-7.3) for the 3% threshold and 4.6 events per hour (95% CI, 3.5-5.6) for the 4% threshold, they reported. For the Embletta monitor, manual and automated scoring varied by an average of 5.3 (95% CI, 3.2-7.3) and 8.4 (95% CI, 7.2-9.6) events per hour, respectively.
The difference between automated and manual scoring narrowed substantially when considering oxygen desaturation index instead of AHI, the researchers reported.
The investigators then used AHI to assess OSA severity. For the ApneaLink Plus monitor, relying on automated readings led to a <10% rate of clinically significant misclassification of OSA severity, regardless which oxygen desaturation threshold was used. For the Embletta device, rates of clinically significant misclassification were 29% and 41% for the 3% and 4% thresholds, respectively. “As expected, disease misclassification was most notable for subjects with mild disease irrespective of the device used,” they wrote.
The National Institutes of Health funded the work. One coauthor reported relationships with ResMed and Koninklijke Philips NV (Respironics). The other authors declared no relevant conflicts of interest.
FROM CHEST
Key clinical point: Automated scoring data from two type-3 home sleep test monitors misclassified the severity of obstructive sleep apnea.
Major finding: Automated scoring consistently underscored disordered-breathing events, compared with manual scoring, for both monitors, regardless of whether the apnea-hypopnea index was defined based on the >3% or >4% oxygen desaturation threshold.
Data source: Evaluation of the ApneaLink Plus (ResMed) and Embletta (Embla Systems) type-3 devices in 200 subjects who had not been previously diagnosed with OSA.
Disclosures: The National Institutes of Health funded the work. One coauthor reported relationships with ResMed and Koninklijke Philips NV (Respironics). The other authors declared no relevant conflicts of interest.
FDA approves Anthrasil to treat inhalational anthrax
The Food and Drug Administration has approved Anthrasil, Anthrax Immune Globulin Intravenous (Human), for treatment of inhalational anthrax when used with appropriate antibacterial drugs.
Inhalational anthrax is caused by breathing in Bacillus anthracis spores, which can occur after exposure to infected animals or contaminated animal products, or as a result of an intentional release of spores. In a statement, Dr. Karen Midthun – director of the FDA’s Center for Biologics Evaluation and Research – explained that Anthrasil “will be stored in U.S. Strategic National Stockpile to facilitate its availability in response to an anthrax emergency.”
Anthrasil was purchased by the U.S. Department of Health & Human Services’ Biomedical Advanced Research and Development Authority (BARDA) in 2011, but because it was not approved, its use prior to FDA approval would have required an emergency use authorization from the FDA.
The efficacy of Anthrasil was studied in animals because it was not feasible or ethical to conduct adequately controlled efficacy studies in humans, the FDA said. Monkeys and rabbits were exposed to Bacillus anthracis spores, and subsequently given either Anthrasil or a placebo. The survival rate for monkeys given Anthrasil was between 36% and 70%, with a trend toward increased survival at higher doses of Anthrasil. None of the monkeys given placebo survived. Rabbits had a 26% survival rate when given the drug, compared to 2% of those given placebo. A separate study exposed rabbits to Bacillus anthracis and treated them with either antibiotics or a combination of antibiotics and Anthrasil; survival rates were 71% for those treated with the combination and 25% for those treated with antibiotics only.
Safety was tested in 74 healthy human volunteers and the most commonly reported side effects were headache, back pain, nausea, and pain and swelling at the infusion site.
Anthrasil is manufactured by Cangene Corporation, based in Winnipeg, Canada, which developed the drug in collaboration with BARDA.
The Food and Drug Administration has approved Anthrasil, Anthrax Immune Globulin Intravenous (Human), for treatment of inhalational anthrax when used with appropriate antibacterial drugs.
Inhalational anthrax is caused by breathing in Bacillus anthracis spores, which can occur after exposure to infected animals or contaminated animal products, or as a result of an intentional release of spores. In a statement, Dr. Karen Midthun – director of the FDA’s Center for Biologics Evaluation and Research – explained that Anthrasil “will be stored in U.S. Strategic National Stockpile to facilitate its availability in response to an anthrax emergency.”
Anthrasil was purchased by the U.S. Department of Health & Human Services’ Biomedical Advanced Research and Development Authority (BARDA) in 2011, but because it was not approved, its use prior to FDA approval would have required an emergency use authorization from the FDA.
The efficacy of Anthrasil was studied in animals because it was not feasible or ethical to conduct adequately controlled efficacy studies in humans, the FDA said. Monkeys and rabbits were exposed to Bacillus anthracis spores, and subsequently given either Anthrasil or a placebo. The survival rate for monkeys given Anthrasil was between 36% and 70%, with a trend toward increased survival at higher doses of Anthrasil. None of the monkeys given placebo survived. Rabbits had a 26% survival rate when given the drug, compared to 2% of those given placebo. A separate study exposed rabbits to Bacillus anthracis and treated them with either antibiotics or a combination of antibiotics and Anthrasil; survival rates were 71% for those treated with the combination and 25% for those treated with antibiotics only.
Safety was tested in 74 healthy human volunteers and the most commonly reported side effects were headache, back pain, nausea, and pain and swelling at the infusion site.
Anthrasil is manufactured by Cangene Corporation, based in Winnipeg, Canada, which developed the drug in collaboration with BARDA.
The Food and Drug Administration has approved Anthrasil, Anthrax Immune Globulin Intravenous (Human), for treatment of inhalational anthrax when used with appropriate antibacterial drugs.
Inhalational anthrax is caused by breathing in Bacillus anthracis spores, which can occur after exposure to infected animals or contaminated animal products, or as a result of an intentional release of spores. In a statement, Dr. Karen Midthun – director of the FDA’s Center for Biologics Evaluation and Research – explained that Anthrasil “will be stored in U.S. Strategic National Stockpile to facilitate its availability in response to an anthrax emergency.”
Anthrasil was purchased by the U.S. Department of Health & Human Services’ Biomedical Advanced Research and Development Authority (BARDA) in 2011, but because it was not approved, its use prior to FDA approval would have required an emergency use authorization from the FDA.
The efficacy of Anthrasil was studied in animals because it was not feasible or ethical to conduct adequately controlled efficacy studies in humans, the FDA said. Monkeys and rabbits were exposed to Bacillus anthracis spores, and subsequently given either Anthrasil or a placebo. The survival rate for monkeys given Anthrasil was between 36% and 70%, with a trend toward increased survival at higher doses of Anthrasil. None of the monkeys given placebo survived. Rabbits had a 26% survival rate when given the drug, compared to 2% of those given placebo. A separate study exposed rabbits to Bacillus anthracis and treated them with either antibiotics or a combination of antibiotics and Anthrasil; survival rates were 71% for those treated with the combination and 25% for those treated with antibiotics only.
Safety was tested in 74 healthy human volunteers and the most commonly reported side effects were headache, back pain, nausea, and pain and swelling at the infusion site.
Anthrasil is manufactured by Cangene Corporation, based in Winnipeg, Canada, which developed the drug in collaboration with BARDA.
Ebola vaccine based on 2014 strain safe for human use
A novel Ebola vaccine based on the glycoprotein of the 2014 strain instead of the 1976 Zaire virus is safe and immunogenic, according to Feng-Cai Zhu and associates.
One hundred twenty participants were separated into three groups: One group received placebo, one group received low-dose vaccine, and one group received high-dose vaccine. Glycoprotein-specific antibodies were significantly higher in the low-dose and high-dose groups at both 14 and 28 days, compared with placebo. About two-thirds of participants reported at least one adverse reaction, with the most common being mild pain around the injection site, which was reported by 42% of patients (Lancet 2015 March 24 [doi:10.1016/S0140-6736(15)60553-0]).
“The mutation of Ebola virus might compromise the immunogenicity of the Ebola vaccines based on the 1976 Zaire strain; therefore, we expected that the vaccine based on the 2014 Guinea strain could be more efficient and specific to protect against the presently epidemic Ebola disease. However, more evidence about protectiveness is needed to establish whether the vaccine based on the 2014 strain is really more beneficial than that based on the 1976 Zaire strain,” the researchers noted. They are planning “to assess the persistence of the specific immune response by following up the vaccine recipients of this study” beyond the 28 days.
To read the full study in the Lancet, click here.
A novel Ebola vaccine based on the glycoprotein of the 2014 strain instead of the 1976 Zaire virus is safe and immunogenic, according to Feng-Cai Zhu and associates.
One hundred twenty participants were separated into three groups: One group received placebo, one group received low-dose vaccine, and one group received high-dose vaccine. Glycoprotein-specific antibodies were significantly higher in the low-dose and high-dose groups at both 14 and 28 days, compared with placebo. About two-thirds of participants reported at least one adverse reaction, with the most common being mild pain around the injection site, which was reported by 42% of patients (Lancet 2015 March 24 [doi:10.1016/S0140-6736(15)60553-0]).
“The mutation of Ebola virus might compromise the immunogenicity of the Ebola vaccines based on the 1976 Zaire strain; therefore, we expected that the vaccine based on the 2014 Guinea strain could be more efficient and specific to protect against the presently epidemic Ebola disease. However, more evidence about protectiveness is needed to establish whether the vaccine based on the 2014 strain is really more beneficial than that based on the 1976 Zaire strain,” the researchers noted. They are planning “to assess the persistence of the specific immune response by following up the vaccine recipients of this study” beyond the 28 days.
To read the full study in the Lancet, click here.
A novel Ebola vaccine based on the glycoprotein of the 2014 strain instead of the 1976 Zaire virus is safe and immunogenic, according to Feng-Cai Zhu and associates.
One hundred twenty participants were separated into three groups: One group received placebo, one group received low-dose vaccine, and one group received high-dose vaccine. Glycoprotein-specific antibodies were significantly higher in the low-dose and high-dose groups at both 14 and 28 days, compared with placebo. About two-thirds of participants reported at least one adverse reaction, with the most common being mild pain around the injection site, which was reported by 42% of patients (Lancet 2015 March 24 [doi:10.1016/S0140-6736(15)60553-0]).
“The mutation of Ebola virus might compromise the immunogenicity of the Ebola vaccines based on the 1976 Zaire strain; therefore, we expected that the vaccine based on the 2014 Guinea strain could be more efficient and specific to protect against the presently epidemic Ebola disease. However, more evidence about protectiveness is needed to establish whether the vaccine based on the 2014 strain is really more beneficial than that based on the 1976 Zaire strain,” the researchers noted. They are planning “to assess the persistence of the specific immune response by following up the vaccine recipients of this study” beyond the 28 days.
To read the full study in the Lancet, click here.
Recurrent depression worsens odds of smoking cessation
People with recurrent depression are much less likely to be able to effectively quit smoking, according to a study conducted by Laurie Zawertailo, Ph.D., and her associates.
Participants were interviewed 6 months after receiving an intervention. In a secondary analysis of data, Dr. Zawertailo and her colleagues found that those with recurrent depression reported trying to quit six or more times in the past. When variables known to be tied to quit success such as age and socioeconomic indicators were entered into a regression model, the investigators found that recurrent depression was the only factor that remained a significant predictor of abstinence at 6 months. Depression diagnoses were self-reported and not confirmed by validated scales or interviews.
“These findings indicate that depression status should be assessed prior to initiating treatment for smoking cessation and treatment should be tailored to provide additional support through counseling and/or pharmacotherapy,” the investigators concluded.
Find the full study in Psychiatry Research (doi:10.1016/j.psychres.2014.12.027).
People with recurrent depression are much less likely to be able to effectively quit smoking, according to a study conducted by Laurie Zawertailo, Ph.D., and her associates.
Participants were interviewed 6 months after receiving an intervention. In a secondary analysis of data, Dr. Zawertailo and her colleagues found that those with recurrent depression reported trying to quit six or more times in the past. When variables known to be tied to quit success such as age and socioeconomic indicators were entered into a regression model, the investigators found that recurrent depression was the only factor that remained a significant predictor of abstinence at 6 months. Depression diagnoses were self-reported and not confirmed by validated scales or interviews.
“These findings indicate that depression status should be assessed prior to initiating treatment for smoking cessation and treatment should be tailored to provide additional support through counseling and/or pharmacotherapy,” the investigators concluded.
Find the full study in Psychiatry Research (doi:10.1016/j.psychres.2014.12.027).
People with recurrent depression are much less likely to be able to effectively quit smoking, according to a study conducted by Laurie Zawertailo, Ph.D., and her associates.
Participants were interviewed 6 months after receiving an intervention. In a secondary analysis of data, Dr. Zawertailo and her colleagues found that those with recurrent depression reported trying to quit six or more times in the past. When variables known to be tied to quit success such as age and socioeconomic indicators were entered into a regression model, the investigators found that recurrent depression was the only factor that remained a significant predictor of abstinence at 6 months. Depression diagnoses were self-reported and not confirmed by validated scales or interviews.
“These findings indicate that depression status should be assessed prior to initiating treatment for smoking cessation and treatment should be tailored to provide additional support through counseling and/or pharmacotherapy,” the investigators concluded.
Find the full study in Psychiatry Research (doi:10.1016/j.psychres.2014.12.027).
Outcomes worse for lung cancer patients with VTE
Hospitalized lung cancer patients who had venous thromboembolisms had significantly worse outcomes than did other patients, according to a study published in Lung Cancer.
Of the more than 570,000 included lung cancer patient hospitalizations, just over 3.6% also presented with venous thromboembolisms (VTE). Patients with VTE had a longer length of stay (7.15 days vs. 6.05 days), higher inpatient mortality (10.03% vs. 8.69%), greater hospital costs ($43,800 vs. $37,800), and were more likely to have moderate to severe disability after being discharged (55% vs. 49%) than patients who did not have VTE. Blacks were also at a higher risk, comprising 13.07% of the VTE group but only 10.66% of the non-VTE group, reported Dr. Conor Steuer and his associates at Emory University, Atlanta.
The study results “highlight the need for prospective studies to clarify the impact of VTE complications in lung cancer patients and identify effective interventions to improve the poorer outcome observed in this population,” Dr. Steuer and his associates concluded.
Find the full study in Lung Cancer (doi: 10.1016/j.lungcan.2015.01.022).
Hospitalized lung cancer patients who had venous thromboembolisms had significantly worse outcomes than did other patients, according to a study published in Lung Cancer.
Of the more than 570,000 included lung cancer patient hospitalizations, just over 3.6% also presented with venous thromboembolisms (VTE). Patients with VTE had a longer length of stay (7.15 days vs. 6.05 days), higher inpatient mortality (10.03% vs. 8.69%), greater hospital costs ($43,800 vs. $37,800), and were more likely to have moderate to severe disability after being discharged (55% vs. 49%) than patients who did not have VTE. Blacks were also at a higher risk, comprising 13.07% of the VTE group but only 10.66% of the non-VTE group, reported Dr. Conor Steuer and his associates at Emory University, Atlanta.
The study results “highlight the need for prospective studies to clarify the impact of VTE complications in lung cancer patients and identify effective interventions to improve the poorer outcome observed in this population,” Dr. Steuer and his associates concluded.
Find the full study in Lung Cancer (doi: 10.1016/j.lungcan.2015.01.022).
Hospitalized lung cancer patients who had venous thromboembolisms had significantly worse outcomes than did other patients, according to a study published in Lung Cancer.
Of the more than 570,000 included lung cancer patient hospitalizations, just over 3.6% also presented with venous thromboembolisms (VTE). Patients with VTE had a longer length of stay (7.15 days vs. 6.05 days), higher inpatient mortality (10.03% vs. 8.69%), greater hospital costs ($43,800 vs. $37,800), and were more likely to have moderate to severe disability after being discharged (55% vs. 49%) than patients who did not have VTE. Blacks were also at a higher risk, comprising 13.07% of the VTE group but only 10.66% of the non-VTE group, reported Dr. Conor Steuer and his associates at Emory University, Atlanta.
The study results “highlight the need for prospective studies to clarify the impact of VTE complications in lung cancer patients and identify effective interventions to improve the poorer outcome observed in this population,” Dr. Steuer and his associates concluded.
Find the full study in Lung Cancer (doi: 10.1016/j.lungcan.2015.01.022).