User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
Powered by CHEST Physician, Clinician Reviews, MDedge Family Medicine, Internal Medicine News, and The Journal of Clinical Outcomes Management.
FDA axes asthma drugs’ boxed warning
The Food and Drug Administration has eliminated the boxed warning for risk of asthma-related death from the labels of products containing both an inhaled corticosteroid (ICS) and a long-acting beta agonist (LABA), the agency announced.
In 2011, the FDA required companies manufacturing fixed-dose LABA-ICS combination products to conduct 26-week clinical safety trials to evaluate the risks of serious adverse asthma-related events in patients treated with these drugs. Specifically, the companies had to compare the follows the FDA’s review of these trials, which found that treating asthma with LABAs in combination with ICS did not result in patients experiencing significantly more serious asthma-related side effects and asthma-related deaths, compared with those being treated with an ICS alone, according to the FDA announcement. “Results of subgroup analyses for gender, adolescents 12-18 years, and African Americans are consistent with the primary endpoint results,” the statement added.
“These trials showed that LABAs, when used with ICS, did not significantly increase the risk of asthma-related hospitalizations, the need to insert a breathing tube known as intubation, or asthma-related deaths, compared to ICS alone,” the FDA said in the statement.
The trials also demonstrated that using the combination reduced asthma exacerbations, compared with using ICS alone, and that most of the exacerbations “were those that required at least 3 days of systemic corticosteroids” – information that is being added the product labels, according to the FDA.
The products that will no longer carry this boxed warning in their labels include AstraZeneca’s budesonide/formoterol fumarate dihydrate (Symbicort) and GlaxoSmithKline’s fluticasone furoate/vilanterol (Breo Ellipta) and fluticasone propionate/salmeterol (Advair Diskus and Advair HFA).
The FDA also approved updates to the Warnings and Precautions section of labeling for the ICS/LABA class, which now includes a description of the four trials. Information on the efficacy of the drugs, found in the trials, has been added to the Clinical Studies section of the labels as well.
In a related safety announcement, the FDA stated the following: “Using LABAs alone to treat asthma without an ICS to treat lung inflammation is associated with an increased risk of asthma-related death. Therefore, the Boxed Warning stating this will remain in the labels of all single-ingredient LABA medicines, which are approved to treat asthma, chronic obstructive pulmonary disease (COPD), and wheezing caused by exercise. The labels of medicines that contain both an ICS and LABA also retain a Warning and Precaution related to the increased risk of asthma-related death when LABAs are used without an ICS to treat asthma.
The Food and Drug Administration has eliminated the boxed warning for risk of asthma-related death from the labels of products containing both an inhaled corticosteroid (ICS) and a long-acting beta agonist (LABA), the agency announced.
In 2011, the FDA required companies manufacturing fixed-dose LABA-ICS combination products to conduct 26-week clinical safety trials to evaluate the risks of serious adverse asthma-related events in patients treated with these drugs. Specifically, the companies had to compare the follows the FDA’s review of these trials, which found that treating asthma with LABAs in combination with ICS did not result in patients experiencing significantly more serious asthma-related side effects and asthma-related deaths, compared with those being treated with an ICS alone, according to the FDA announcement. “Results of subgroup analyses for gender, adolescents 12-18 years, and African Americans are consistent with the primary endpoint results,” the statement added.
“These trials showed that LABAs, when used with ICS, did not significantly increase the risk of asthma-related hospitalizations, the need to insert a breathing tube known as intubation, or asthma-related deaths, compared to ICS alone,” the FDA said in the statement.
The trials also demonstrated that using the combination reduced asthma exacerbations, compared with using ICS alone, and that most of the exacerbations “were those that required at least 3 days of systemic corticosteroids” – information that is being added the product labels, according to the FDA.
The products that will no longer carry this boxed warning in their labels include AstraZeneca’s budesonide/formoterol fumarate dihydrate (Symbicort) and GlaxoSmithKline’s fluticasone furoate/vilanterol (Breo Ellipta) and fluticasone propionate/salmeterol (Advair Diskus and Advair HFA).
The FDA also approved updates to the Warnings and Precautions section of labeling for the ICS/LABA class, which now includes a description of the four trials. Information on the efficacy of the drugs, found in the trials, has been added to the Clinical Studies section of the labels as well.
In a related safety announcement, the FDA stated the following: “Using LABAs alone to treat asthma without an ICS to treat lung inflammation is associated with an increased risk of asthma-related death. Therefore, the Boxed Warning stating this will remain in the labels of all single-ingredient LABA medicines, which are approved to treat asthma, chronic obstructive pulmonary disease (COPD), and wheezing caused by exercise. The labels of medicines that contain both an ICS and LABA also retain a Warning and Precaution related to the increased risk of asthma-related death when LABAs are used without an ICS to treat asthma.
The Food and Drug Administration has eliminated the boxed warning for risk of asthma-related death from the labels of products containing both an inhaled corticosteroid (ICS) and a long-acting beta agonist (LABA), the agency announced.
In 2011, the FDA required companies manufacturing fixed-dose LABA-ICS combination products to conduct 26-week clinical safety trials to evaluate the risks of serious adverse asthma-related events in patients treated with these drugs. Specifically, the companies had to compare the follows the FDA’s review of these trials, which found that treating asthma with LABAs in combination with ICS did not result in patients experiencing significantly more serious asthma-related side effects and asthma-related deaths, compared with those being treated with an ICS alone, according to the FDA announcement. “Results of subgroup analyses for gender, adolescents 12-18 years, and African Americans are consistent with the primary endpoint results,” the statement added.
“These trials showed that LABAs, when used with ICS, did not significantly increase the risk of asthma-related hospitalizations, the need to insert a breathing tube known as intubation, or asthma-related deaths, compared to ICS alone,” the FDA said in the statement.
The trials also demonstrated that using the combination reduced asthma exacerbations, compared with using ICS alone, and that most of the exacerbations “were those that required at least 3 days of systemic corticosteroids” – information that is being added the product labels, according to the FDA.
The products that will no longer carry this boxed warning in their labels include AstraZeneca’s budesonide/formoterol fumarate dihydrate (Symbicort) and GlaxoSmithKline’s fluticasone furoate/vilanterol (Breo Ellipta) and fluticasone propionate/salmeterol (Advair Diskus and Advair HFA).
The FDA also approved updates to the Warnings and Precautions section of labeling for the ICS/LABA class, which now includes a description of the four trials. Information on the efficacy of the drugs, found in the trials, has been added to the Clinical Studies section of the labels as well.
In a related safety announcement, the FDA stated the following: “Using LABAs alone to treat asthma without an ICS to treat lung inflammation is associated with an increased risk of asthma-related death. Therefore, the Boxed Warning stating this will remain in the labels of all single-ingredient LABA medicines, which are approved to treat asthma, chronic obstructive pulmonary disease (COPD), and wheezing caused by exercise. The labels of medicines that contain both an ICS and LABA also retain a Warning and Precaution related to the increased risk of asthma-related death when LABAs are used without an ICS to treat asthma.
Differentiate Acute Bronchitis and Community Acquired Pneumonia
Nebulized LAMA for COPD approved
The Food and Drug Administration has given the nod to the first nebulized long-acting muscarinic antagonist (LAMA) treatment for chronic obstructive pulmonary disease (COPD) in the United States.
Glycopyrrolate (Lonhala Magnair) utilizes the eFlow technology system, developed by Pari Pharma. This nebulizing system is portable, virtually silent, and delivers the drug in 2-3 minutes, according to a statement from Sunovion Pharmaceuticals.
The approval of glycopyrrolate is based on the results of the GOLDEN (Glycopyrrolate for Obstructive Lung Disease via Electronic Nebulizer) trials. The GOLDEN program comprised the GOLDEN-3 and GOLDEN-4 trials, both of which were phase 3, 12-week, randomized, double-blind, placebo-controlled, parallel-group, multicenter safety and efficacy trials, which compared adult glycopyrrolate patients to a placebo group with moderate to severe COPD. At 12 weeks, patients receiving treatment with glycopyrrolate showed clinical and statistically significant improvements in their baseline forced expiratory volume second (FEV1), compared with placebo.
GOLDEN-5, an additional study, followed the same criteria as previous studies, but increased its length to 48 weeks to evaluate the long-term safety and patient tolerability of glycopyrrolate. It also compared treatment of COPD with glycopyrrolate to treatment of COPD with the previously approved LAMA Spiriva (tiotropium bromide), delivered by the Handihaler device. Glycopyrrolate was well tolerated, and the overall treatment emergence of adverse events for glycopyrrolate and tiotropium bromide were similar.
Sunovion expects glycopyrrolate to be available in U.S. pharmacies in early 2018, according to the statement.
The Food and Drug Administration has given the nod to the first nebulized long-acting muscarinic antagonist (LAMA) treatment for chronic obstructive pulmonary disease (COPD) in the United States.
Glycopyrrolate (Lonhala Magnair) utilizes the eFlow technology system, developed by Pari Pharma. This nebulizing system is portable, virtually silent, and delivers the drug in 2-3 minutes, according to a statement from Sunovion Pharmaceuticals.
The approval of glycopyrrolate is based on the results of the GOLDEN (Glycopyrrolate for Obstructive Lung Disease via Electronic Nebulizer) trials. The GOLDEN program comprised the GOLDEN-3 and GOLDEN-4 trials, both of which were phase 3, 12-week, randomized, double-blind, placebo-controlled, parallel-group, multicenter safety and efficacy trials, which compared adult glycopyrrolate patients to a placebo group with moderate to severe COPD. At 12 weeks, patients receiving treatment with glycopyrrolate showed clinical and statistically significant improvements in their baseline forced expiratory volume second (FEV1), compared with placebo.
GOLDEN-5, an additional study, followed the same criteria as previous studies, but increased its length to 48 weeks to evaluate the long-term safety and patient tolerability of glycopyrrolate. It also compared treatment of COPD with glycopyrrolate to treatment of COPD with the previously approved LAMA Spiriva (tiotropium bromide), delivered by the Handihaler device. Glycopyrrolate was well tolerated, and the overall treatment emergence of adverse events for glycopyrrolate and tiotropium bromide were similar.
Sunovion expects glycopyrrolate to be available in U.S. pharmacies in early 2018, according to the statement.
The Food and Drug Administration has given the nod to the first nebulized long-acting muscarinic antagonist (LAMA) treatment for chronic obstructive pulmonary disease (COPD) in the United States.
Glycopyrrolate (Lonhala Magnair) utilizes the eFlow technology system, developed by Pari Pharma. This nebulizing system is portable, virtually silent, and delivers the drug in 2-3 minutes, according to a statement from Sunovion Pharmaceuticals.
The approval of glycopyrrolate is based on the results of the GOLDEN (Glycopyrrolate for Obstructive Lung Disease via Electronic Nebulizer) trials. The GOLDEN program comprised the GOLDEN-3 and GOLDEN-4 trials, both of which were phase 3, 12-week, randomized, double-blind, placebo-controlled, parallel-group, multicenter safety and efficacy trials, which compared adult glycopyrrolate patients to a placebo group with moderate to severe COPD. At 12 weeks, patients receiving treatment with glycopyrrolate showed clinical and statistically significant improvements in their baseline forced expiratory volume second (FEV1), compared with placebo.
GOLDEN-5, an additional study, followed the same criteria as previous studies, but increased its length to 48 weeks to evaluate the long-term safety and patient tolerability of glycopyrrolate. It also compared treatment of COPD with glycopyrrolate to treatment of COPD with the previously approved LAMA Spiriva (tiotropium bromide), delivered by the Handihaler device. Glycopyrrolate was well tolerated, and the overall treatment emergence of adverse events for glycopyrrolate and tiotropium bromide were similar.
Sunovion expects glycopyrrolate to be available in U.S. pharmacies in early 2018, according to the statement.
PCVs reduced CAP hospitalizations in young children but not other age groups
, but there was no clear impact apparent in other age groups, reported Annemarie van Deursen, MD, of the University Medical Centre (the Netherlands) Utrecht, and her associates.
In the Netherlands, the 7-valent pneumococcal conjugate vaccine (PCV7) was added to the national infant immunization program in 2006; in 2011, PCV7 was replaced by the 10-valent vaccine (PCV10). The investigators undertook a population-based retrospective study during 1999-2014 on all-cause CAP hospitalizations in all ages, identifying 155,994 CAP hospitalizations.
In children aged 0-6 months, the CAP hospitalization rate ratio (RR) was significant from 2012 onward, with an overall post-PCV RR of 0.62 and a RR of 0.19 at the end of the study period in December 2014. In children aged 6 months-1 year, the RR was statistically significant directly after the introduction of PCV, with an overall post-PCV RR of 0.67 and a RR of 0.47 in December 2014, the investigators wrote.
In none of the other age groups did the overall post-PCV hospitalization RR reach statistical significance.
The association of reductions in CAP hospitalizations in children up to 2 years with the introduction of PCV7 “supports the interpretation for a direct causal effect of PCV7, in line with IPD [invasive pneumococcal disease] results that showed a sustained overall IPD reduction in children,” the investigators said. “Furthermore, [during] each subsequent year of the post-PCV period, the reduction in CAP hospitalization rates increased in line with progressive vaccine-type–IPD reduction in the population and limited replacement by nonvaccine type in childhood IPD.”
Read more in Vaccine (2017 Nov 13. doi: 10.1016/j.vaccine.2017.10.090).
, but there was no clear impact apparent in other age groups, reported Annemarie van Deursen, MD, of the University Medical Centre (the Netherlands) Utrecht, and her associates.
In the Netherlands, the 7-valent pneumococcal conjugate vaccine (PCV7) was added to the national infant immunization program in 2006; in 2011, PCV7 was replaced by the 10-valent vaccine (PCV10). The investigators undertook a population-based retrospective study during 1999-2014 on all-cause CAP hospitalizations in all ages, identifying 155,994 CAP hospitalizations.
In children aged 0-6 months, the CAP hospitalization rate ratio (RR) was significant from 2012 onward, with an overall post-PCV RR of 0.62 and a RR of 0.19 at the end of the study period in December 2014. In children aged 6 months-1 year, the RR was statistically significant directly after the introduction of PCV, with an overall post-PCV RR of 0.67 and a RR of 0.47 in December 2014, the investigators wrote.
In none of the other age groups did the overall post-PCV hospitalization RR reach statistical significance.
The association of reductions in CAP hospitalizations in children up to 2 years with the introduction of PCV7 “supports the interpretation for a direct causal effect of PCV7, in line with IPD [invasive pneumococcal disease] results that showed a sustained overall IPD reduction in children,” the investigators said. “Furthermore, [during] each subsequent year of the post-PCV period, the reduction in CAP hospitalization rates increased in line with progressive vaccine-type–IPD reduction in the population and limited replacement by nonvaccine type in childhood IPD.”
Read more in Vaccine (2017 Nov 13. doi: 10.1016/j.vaccine.2017.10.090).
, but there was no clear impact apparent in other age groups, reported Annemarie van Deursen, MD, of the University Medical Centre (the Netherlands) Utrecht, and her associates.
In the Netherlands, the 7-valent pneumococcal conjugate vaccine (PCV7) was added to the national infant immunization program in 2006; in 2011, PCV7 was replaced by the 10-valent vaccine (PCV10). The investigators undertook a population-based retrospective study during 1999-2014 on all-cause CAP hospitalizations in all ages, identifying 155,994 CAP hospitalizations.
In children aged 0-6 months, the CAP hospitalization rate ratio (RR) was significant from 2012 onward, with an overall post-PCV RR of 0.62 and a RR of 0.19 at the end of the study period in December 2014. In children aged 6 months-1 year, the RR was statistically significant directly after the introduction of PCV, with an overall post-PCV RR of 0.67 and a RR of 0.47 in December 2014, the investigators wrote.
In none of the other age groups did the overall post-PCV hospitalization RR reach statistical significance.
The association of reductions in CAP hospitalizations in children up to 2 years with the introduction of PCV7 “supports the interpretation for a direct causal effect of PCV7, in line with IPD [invasive pneumococcal disease] results that showed a sustained overall IPD reduction in children,” the investigators said. “Furthermore, [during] each subsequent year of the post-PCV period, the reduction in CAP hospitalization rates increased in line with progressive vaccine-type–IPD reduction in the population and limited replacement by nonvaccine type in childhood IPD.”
Read more in Vaccine (2017 Nov 13. doi: 10.1016/j.vaccine.2017.10.090).
FROM VACCINE
Flu vaccine did not protect children with acute leukemia
said April Sykes of St. Jude Children’s Research Hospital in Carmel, Ind., and her associates.
Patients aged 1-21 years being treated for acute leukemia during three successive influenza seasons (2011-2012, 2012-2013, and 2013-2014) were identified by a retrospective review of EHRs; of those patients, 354 (71%) patients received TIV, and 98 (20%) received a booster dose of flu vaccine.
Also, whether the children and youth received one or two doses of flu vaccine made no difference in the rates of influenza (0.60 vs. 1.02; P = .107), the investigators reported.
These data suggest “that influenza vaccine may be ineffective in children receiving therapy for acute leukemia and that routine administration of TIV may not reflect high-value care,” the researchers said. “Until more immunogenic and protective vaccines are developed, efforts to prevent influenza in high-risk populations should focus on more general strategies, such as avoiding ill persons and practicing good respiratory hygiene in households and health care facilities.”
Read more in the Journal of Pediatrics (2017 Nov 21. doi: 10.1016/j.jpeds.2017.08.071).
said April Sykes of St. Jude Children’s Research Hospital in Carmel, Ind., and her associates.
Patients aged 1-21 years being treated for acute leukemia during three successive influenza seasons (2011-2012, 2012-2013, and 2013-2014) were identified by a retrospective review of EHRs; of those patients, 354 (71%) patients received TIV, and 98 (20%) received a booster dose of flu vaccine.
Also, whether the children and youth received one or two doses of flu vaccine made no difference in the rates of influenza (0.60 vs. 1.02; P = .107), the investigators reported.
These data suggest “that influenza vaccine may be ineffective in children receiving therapy for acute leukemia and that routine administration of TIV may not reflect high-value care,” the researchers said. “Until more immunogenic and protective vaccines are developed, efforts to prevent influenza in high-risk populations should focus on more general strategies, such as avoiding ill persons and practicing good respiratory hygiene in households and health care facilities.”
Read more in the Journal of Pediatrics (2017 Nov 21. doi: 10.1016/j.jpeds.2017.08.071).
said April Sykes of St. Jude Children’s Research Hospital in Carmel, Ind., and her associates.
Patients aged 1-21 years being treated for acute leukemia during three successive influenza seasons (2011-2012, 2012-2013, and 2013-2014) were identified by a retrospective review of EHRs; of those patients, 354 (71%) patients received TIV, and 98 (20%) received a booster dose of flu vaccine.
Also, whether the children and youth received one or two doses of flu vaccine made no difference in the rates of influenza (0.60 vs. 1.02; P = .107), the investigators reported.
These data suggest “that influenza vaccine may be ineffective in children receiving therapy for acute leukemia and that routine administration of TIV may not reflect high-value care,” the researchers said. “Until more immunogenic and protective vaccines are developed, efforts to prevent influenza in high-risk populations should focus on more general strategies, such as avoiding ill persons and practicing good respiratory hygiene in households and health care facilities.”
Read more in the Journal of Pediatrics (2017 Nov 21. doi: 10.1016/j.jpeds.2017.08.071).
FROM THE JOURNAL OF PEDIATRICS
Hyperbaric oxygen may cut CO deaths
In patients with carbon monoxide poisoning, hyperbaric oxygen therapy was associated with a lower rate of mortality, according to results of a recent retrospective study.
The mortality reduction was particularly evident among patients under 20 years of age and in patients with acute respiratory failure, authors of the study said in a report published in Chest (2017 Nov. doi: 10.1016/j.chest.2017.03.049).
“The results provide important references for decision making in the treatment of carbon monoxide poisoning,” Chien-Cheng Huang, MD, department of emergency medicine, Chi-Mei Medical Center, Tainan, Taiwan, and colleagues said in their report.
While hyperbaric oxygen has been suggested for severe carbon monoxide poisoning, 100% normobaric oxygen is considered standard treatment, according to Dr. Huang and colleagues.
“There has been no consensus about whether hyperbaric oxygen therapy is better than 100% normobaric oxygen alone, or the number of sessions of hyperbaric oxygen therapy that are necessary regarding mortality and morbidity,” they wrote.
In a Taiwanese nationwide poisoning database, Dr. Huang and colleagues identified 25,737 patients diagnosed with carbon monoxide poisoning between 1999 and 2012. Of those patients, 7,278 had hyperbaric oxygen therapy.
After researchers adjusted for variables including age, sex, and underlying comorbidities, the mortality rate was lower in patients who underwent hyperbaric oxygen therapy, compared with those who did not (adjusted hazard ratio, 0.74; 95% confidence interval, 0.67-0.81), data show.
The reduction in mortality was especially notable in patients younger than age 20 years (adjusted HR, 0.45; 95% CI, 0.26-0.80), according to the researchers.
A similarly greater magnitude of mortality benefit also was found for patients who had acute respiratory failure, “which supports acute respiratory failure being an indication for hyperbaric oxygen therapy,” investigators wrote. “Further studies are warranted to clarify this issue.”
The number of hyperbaric oxygen therapy sessions appeared to make a difference in mortality. Patients who received two or more sessions had a lower rate of mortality than did those who had only one session, according to the report.
Predictors of mortality, described in more detail in the published report, included older age, diabetes, alcoholism, and suicide attempts, among other factors.
“In addition to considering hyperbaric oxygen therapy for reducing mortality, control of other concomitant mortality predictors is necessary,” the authors concluded based on their results.
Accidental deaths from carbon monoxide poisoning also are a major issue in the United States, where each year, there are an estimated 1,000-2,000 cases, according to the authors. Additionally, accidental carbon monoxide poisoning has “increased greatly in the past 10 years,” they said in the report.
Other studies have shown that, compared with normobaric oxygen, hyperbaric oxygen therapy did not reduce neurologic complications, the authors noted. Even so, that fact “does not suggest that hyperbaric oxygen therapy is not beneficial regarding mortality,” they wrote. “In fact, it is possible that reducing mortality may increase morbidities such as neurologic sequelae.”
Dr. Huang and coauthors reported no conflicts of interest related to the study, which was supported by Chi-Mei Medical Center in Taiwan.
Use of hyperbaric oxygen therapy to treat carbon monoxide poisioning has been “controversial since its inception” since early promoters “tended to place hyperbaric treatment ahead of strong supporting data,” wrote Clayton T. Cowl, MD, FCCP, in an editorial regarding the study by Dr. Huang and colleagues.
These data are compelling because they come from what is believed to be the first large-scale study that specifically examines mortality as an endpoint in an entire nation, as opposed to smaller cohorts in single centers or even multiple institutions, he said in his editorial.
“Have we reached the point of clearly establishing that delivery of pure oxygen in a high-pressure environment is more effective in treating patients who have carbon monoxide poisoning than is normobaric supplemental oxygen alone? Probably not,” Dr. Cowl wrote.
“The retrospective database study by Huang et al, despite its large size and interesting findings, remains distant from the ideal of a large blinded multicenter randomized controlled trial using a standardized protocol to compare normobaric supplemental oxygenation with hyperbaric oxygen therapy delivery for this cohort,” he explained. “However, its size, scale, and findings add credibility to the mounting data supporting HBOT for this indication.”
Clayton T. Cowl, MD, FCCP, is with the division of preventive, occupational, and aerospace medicine and the division of pulmonary and critical care medicine, Mayo Clinic. His comments came from an editorial in Chest (doi: 10.1016/j.chest.2017.07.022) He declared no financial or nonfinancial disclosures related to the editorial.
Use of hyperbaric oxygen therapy to treat carbon monoxide poisioning has been “controversial since its inception” since early promoters “tended to place hyperbaric treatment ahead of strong supporting data,” wrote Clayton T. Cowl, MD, FCCP, in an editorial regarding the study by Dr. Huang and colleagues.
These data are compelling because they come from what is believed to be the first large-scale study that specifically examines mortality as an endpoint in an entire nation, as opposed to smaller cohorts in single centers or even multiple institutions, he said in his editorial.
“Have we reached the point of clearly establishing that delivery of pure oxygen in a high-pressure environment is more effective in treating patients who have carbon monoxide poisoning than is normobaric supplemental oxygen alone? Probably not,” Dr. Cowl wrote.
“The retrospective database study by Huang et al, despite its large size and interesting findings, remains distant from the ideal of a large blinded multicenter randomized controlled trial using a standardized protocol to compare normobaric supplemental oxygenation with hyperbaric oxygen therapy delivery for this cohort,” he explained. “However, its size, scale, and findings add credibility to the mounting data supporting HBOT for this indication.”
Clayton T. Cowl, MD, FCCP, is with the division of preventive, occupational, and aerospace medicine and the division of pulmonary and critical care medicine, Mayo Clinic. His comments came from an editorial in Chest (doi: 10.1016/j.chest.2017.07.022) He declared no financial or nonfinancial disclosures related to the editorial.
Use of hyperbaric oxygen therapy to treat carbon monoxide poisioning has been “controversial since its inception” since early promoters “tended to place hyperbaric treatment ahead of strong supporting data,” wrote Clayton T. Cowl, MD, FCCP, in an editorial regarding the study by Dr. Huang and colleagues.
These data are compelling because they come from what is believed to be the first large-scale study that specifically examines mortality as an endpoint in an entire nation, as opposed to smaller cohorts in single centers or even multiple institutions, he said in his editorial.
“Have we reached the point of clearly establishing that delivery of pure oxygen in a high-pressure environment is more effective in treating patients who have carbon monoxide poisoning than is normobaric supplemental oxygen alone? Probably not,” Dr. Cowl wrote.
“The retrospective database study by Huang et al, despite its large size and interesting findings, remains distant from the ideal of a large blinded multicenter randomized controlled trial using a standardized protocol to compare normobaric supplemental oxygenation with hyperbaric oxygen therapy delivery for this cohort,” he explained. “However, its size, scale, and findings add credibility to the mounting data supporting HBOT for this indication.”
Clayton T. Cowl, MD, FCCP, is with the division of preventive, occupational, and aerospace medicine and the division of pulmonary and critical care medicine, Mayo Clinic. His comments came from an editorial in Chest (doi: 10.1016/j.chest.2017.07.022) He declared no financial or nonfinancial disclosures related to the editorial.
In patients with carbon monoxide poisoning, hyperbaric oxygen therapy was associated with a lower rate of mortality, according to results of a recent retrospective study.
The mortality reduction was particularly evident among patients under 20 years of age and in patients with acute respiratory failure, authors of the study said in a report published in Chest (2017 Nov. doi: 10.1016/j.chest.2017.03.049).
“The results provide important references for decision making in the treatment of carbon monoxide poisoning,” Chien-Cheng Huang, MD, department of emergency medicine, Chi-Mei Medical Center, Tainan, Taiwan, and colleagues said in their report.
While hyperbaric oxygen has been suggested for severe carbon monoxide poisoning, 100% normobaric oxygen is considered standard treatment, according to Dr. Huang and colleagues.
“There has been no consensus about whether hyperbaric oxygen therapy is better than 100% normobaric oxygen alone, or the number of sessions of hyperbaric oxygen therapy that are necessary regarding mortality and morbidity,” they wrote.
In a Taiwanese nationwide poisoning database, Dr. Huang and colleagues identified 25,737 patients diagnosed with carbon monoxide poisoning between 1999 and 2012. Of those patients, 7,278 had hyperbaric oxygen therapy.
After researchers adjusted for variables including age, sex, and underlying comorbidities, the mortality rate was lower in patients who underwent hyperbaric oxygen therapy, compared with those who did not (adjusted hazard ratio, 0.74; 95% confidence interval, 0.67-0.81), data show.
The reduction in mortality was especially notable in patients younger than age 20 years (adjusted HR, 0.45; 95% CI, 0.26-0.80), according to the researchers.
A similarly greater magnitude of mortality benefit also was found for patients who had acute respiratory failure, “which supports acute respiratory failure being an indication for hyperbaric oxygen therapy,” investigators wrote. “Further studies are warranted to clarify this issue.”
The number of hyperbaric oxygen therapy sessions appeared to make a difference in mortality. Patients who received two or more sessions had a lower rate of mortality than did those who had only one session, according to the report.
Predictors of mortality, described in more detail in the published report, included older age, diabetes, alcoholism, and suicide attempts, among other factors.
“In addition to considering hyperbaric oxygen therapy for reducing mortality, control of other concomitant mortality predictors is necessary,” the authors concluded based on their results.
Accidental deaths from carbon monoxide poisoning also are a major issue in the United States, where each year, there are an estimated 1,000-2,000 cases, according to the authors. Additionally, accidental carbon monoxide poisoning has “increased greatly in the past 10 years,” they said in the report.
Other studies have shown that, compared with normobaric oxygen, hyperbaric oxygen therapy did not reduce neurologic complications, the authors noted. Even so, that fact “does not suggest that hyperbaric oxygen therapy is not beneficial regarding mortality,” they wrote. “In fact, it is possible that reducing mortality may increase morbidities such as neurologic sequelae.”
Dr. Huang and coauthors reported no conflicts of interest related to the study, which was supported by Chi-Mei Medical Center in Taiwan.
In patients with carbon monoxide poisoning, hyperbaric oxygen therapy was associated with a lower rate of mortality, according to results of a recent retrospective study.
The mortality reduction was particularly evident among patients under 20 years of age and in patients with acute respiratory failure, authors of the study said in a report published in Chest (2017 Nov. doi: 10.1016/j.chest.2017.03.049).
“The results provide important references for decision making in the treatment of carbon monoxide poisoning,” Chien-Cheng Huang, MD, department of emergency medicine, Chi-Mei Medical Center, Tainan, Taiwan, and colleagues said in their report.
While hyperbaric oxygen has been suggested for severe carbon monoxide poisoning, 100% normobaric oxygen is considered standard treatment, according to Dr. Huang and colleagues.
“There has been no consensus about whether hyperbaric oxygen therapy is better than 100% normobaric oxygen alone, or the number of sessions of hyperbaric oxygen therapy that are necessary regarding mortality and morbidity,” they wrote.
In a Taiwanese nationwide poisoning database, Dr. Huang and colleagues identified 25,737 patients diagnosed with carbon monoxide poisoning between 1999 and 2012. Of those patients, 7,278 had hyperbaric oxygen therapy.
After researchers adjusted for variables including age, sex, and underlying comorbidities, the mortality rate was lower in patients who underwent hyperbaric oxygen therapy, compared with those who did not (adjusted hazard ratio, 0.74; 95% confidence interval, 0.67-0.81), data show.
The reduction in mortality was especially notable in patients younger than age 20 years (adjusted HR, 0.45; 95% CI, 0.26-0.80), according to the researchers.
A similarly greater magnitude of mortality benefit also was found for patients who had acute respiratory failure, “which supports acute respiratory failure being an indication for hyperbaric oxygen therapy,” investigators wrote. “Further studies are warranted to clarify this issue.”
The number of hyperbaric oxygen therapy sessions appeared to make a difference in mortality. Patients who received two or more sessions had a lower rate of mortality than did those who had only one session, according to the report.
Predictors of mortality, described in more detail in the published report, included older age, diabetes, alcoholism, and suicide attempts, among other factors.
“In addition to considering hyperbaric oxygen therapy for reducing mortality, control of other concomitant mortality predictors is necessary,” the authors concluded based on their results.
Accidental deaths from carbon monoxide poisoning also are a major issue in the United States, where each year, there are an estimated 1,000-2,000 cases, according to the authors. Additionally, accidental carbon monoxide poisoning has “increased greatly in the past 10 years,” they said in the report.
Other studies have shown that, compared with normobaric oxygen, hyperbaric oxygen therapy did not reduce neurologic complications, the authors noted. Even so, that fact “does not suggest that hyperbaric oxygen therapy is not beneficial regarding mortality,” they wrote. “In fact, it is possible that reducing mortality may increase morbidities such as neurologic sequelae.”
Dr. Huang and coauthors reported no conflicts of interest related to the study, which was supported by Chi-Mei Medical Center in Taiwan.
FROM CHEST
Key clinical point: In patients with carbon monoxide poisoning, hyperbaric oxygen therapy was associated with a lower rate of mortality.
Major finding: The mortality rate was lower in patients who received hyperbaric oxygen therapy, compared with those who did not (adjusted HR, 0.74; 95% CI, 0.67-0.81).
Data source: A retrospective nationwide population-based study from Taiwan including 25,737 individuals diagnosed with carbon monoxide poisoning during 1999-2012.
Disclosures: Chi-Mei Medical Center in Taiwan supported the study. The authors reported no conflicts of interest.
Health disparities in rural America: Chronic conditions
Among rural adults, multiple chronic health conditions are most common in non-Hispanic blacks and American Indians/Alaska Natives (AI/ANs) and least common among Asians and Native Hawaiians/other Pacific Islanders (NHOPIs), according to the Centers for Disease Control and Prevention.
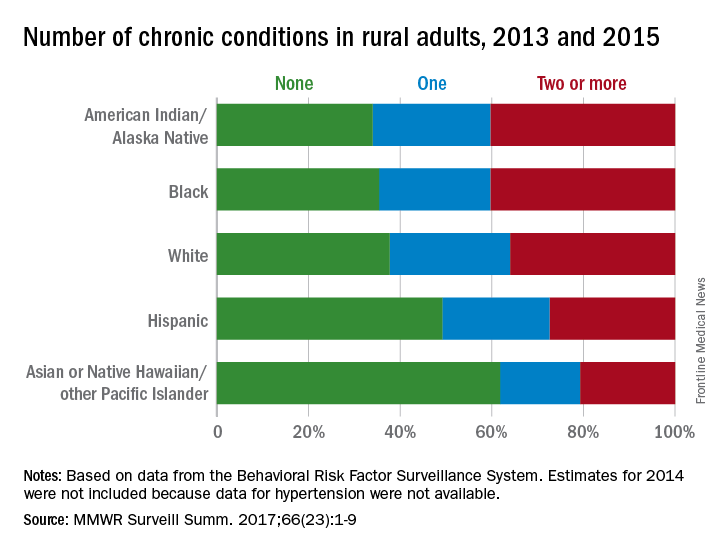
The order was reversed for adults reporting no chronic conditions: Asians and NHOPIs at 61.8%, Hispanics at 49.2%, whites at 37.8%, blacks at 35.4%, and AI/ANs at 34.0%, the researchers said.
For the chronic health conditions included separately in the report, blacks had the highest rate (45.9%) and Asians and NHOPIs had the lowest rate (15.5%) of obesity; AI/ANs were most likely (23.2%) and Asians and NHOPIs were least likely (5.8%) to report depressive disorder. Other conditions considered in the estimates were myocardial infarction; coronary heart disease; stroke; hypertension; asthma; skin cancer; other types of cancer; chronic obstructive pulmonary disease; kidney disease; some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia; and diabetes. Estimates for 2014 were not included because data for hypertension were not available, the investigators noted.
Of the 3,143 counties categorized by the National Center for Health Statistics’ Urban-Rural Classification Scheme for Counties, a total of 1,325 were considered rural and included 6.1% of the U.S. population, they said.
Among rural adults, multiple chronic health conditions are most common in non-Hispanic blacks and American Indians/Alaska Natives (AI/ANs) and least common among Asians and Native Hawaiians/other Pacific Islanders (NHOPIs), according to the Centers for Disease Control and Prevention.

The order was reversed for adults reporting no chronic conditions: Asians and NHOPIs at 61.8%, Hispanics at 49.2%, whites at 37.8%, blacks at 35.4%, and AI/ANs at 34.0%, the researchers said.
For the chronic health conditions included separately in the report, blacks had the highest rate (45.9%) and Asians and NHOPIs had the lowest rate (15.5%) of obesity; AI/ANs were most likely (23.2%) and Asians and NHOPIs were least likely (5.8%) to report depressive disorder. Other conditions considered in the estimates were myocardial infarction; coronary heart disease; stroke; hypertension; asthma; skin cancer; other types of cancer; chronic obstructive pulmonary disease; kidney disease; some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia; and diabetes. Estimates for 2014 were not included because data for hypertension were not available, the investigators noted.
Of the 3,143 counties categorized by the National Center for Health Statistics’ Urban-Rural Classification Scheme for Counties, a total of 1,325 were considered rural and included 6.1% of the U.S. population, they said.
Among rural adults, multiple chronic health conditions are most common in non-Hispanic blacks and American Indians/Alaska Natives (AI/ANs) and least common among Asians and Native Hawaiians/other Pacific Islanders (NHOPIs), according to the Centers for Disease Control and Prevention.

The order was reversed for adults reporting no chronic conditions: Asians and NHOPIs at 61.8%, Hispanics at 49.2%, whites at 37.8%, blacks at 35.4%, and AI/ANs at 34.0%, the researchers said.
For the chronic health conditions included separately in the report, blacks had the highest rate (45.9%) and Asians and NHOPIs had the lowest rate (15.5%) of obesity; AI/ANs were most likely (23.2%) and Asians and NHOPIs were least likely (5.8%) to report depressive disorder. Other conditions considered in the estimates were myocardial infarction; coronary heart disease; stroke; hypertension; asthma; skin cancer; other types of cancer; chronic obstructive pulmonary disease; kidney disease; some form of arthritis, rheumatoid arthritis, gout, lupus, or fibromyalgia; and diabetes. Estimates for 2014 were not included because data for hypertension were not available, the investigators noted.
Of the 3,143 counties categorized by the National Center for Health Statistics’ Urban-Rural Classification Scheme for Counties, a total of 1,325 were considered rural and included 6.1% of the U.S. population, they said.
FROM MMWR SURVEILLANCE SUMMARIES
Acute kidney injury linked with doubled inpatient VTEs
TORONTO – Hospitalized patients with acute kidney injury had more than double the inpatient rate of venous thromboembolism as had patients without acute kidney injury in a prospective, observational study of more than 6,000 hospitalized U.S. soldiers.
He offered four possible mechanisms to explain a link between AKI and VTE:
- Patients with AKI are in a hypercoagulable state.
- AKI alters the pharmacodynamics or pharmacokinetics of VTE prophylactic treatments.
- AKI is a marker of an illness that causes VTE.
- VTE leads to an increased rate of AKI rather than the other way around.
Dr. McMahon’s analysis also revealed that two other clinical conditions that are generally believed to raise VTE risk – obesity and impaired overall renal function identified with stagnant measures – did not correspond with a significantly elevated VTE rate in this study.
The data came from 6,552 adults hospitalized for at least 2 days at Walter Reed between September 2009 and March 2011. The study excluded patients with VTE at the time of admission and also those who had been treated with an anticoagulant at the time of admission. The patients averaged 55 years of age and were hospitalized for a median of 4 days. About 22% of patients received VTE prophylaxis with unfractionated heparin, about 41% received prophylaxis with low-molecular-weight heparin, and about 39% received no VTE prophylaxis (percentages total 102% because of rounding).
About 16% of the patients had been diagnosed with AKI at the time of admission, and an additional 8% developed AKI while hospitalized, defined as an increase in serum creatinine during hospitalization of at least 50% above baseline levels or an increase of more than 0.3 mg/dL above the level at time of admission. During hospitalization, 160 patients (2%) developed a new onset VTE.
In an analysis that adjusted for baseline differences in type of surgery, body mass index, sex, age, and prior hospitalizations during the prior 90 days, the results showed that patients with preexisting or new onset AKI had a 2.2-fold higher rate of VTE, compared with patients without AKI, and this difference was statistically significant, Dr. McMahon reported.
The analysis also showed a significant 62% relatively higher rate of VTE among soldiers hospitalized for a deployment-related event, as well as a significant 63% relatively lower VTE rate among patients not receiving medical prophylaxis, compared with patients receiving an anticoagulant. Dr. McMahon suggested that this lower rate of VTEs among patients not on prophylaxis reflected success in identifying which patients had an increased risk for VTE and hence received prophylaxis.
[email protected]
On Twitter @mitchelzoler
TORONTO – Hospitalized patients with acute kidney injury had more than double the inpatient rate of venous thromboembolism as had patients without acute kidney injury in a prospective, observational study of more than 6,000 hospitalized U.S. soldiers.
He offered four possible mechanisms to explain a link between AKI and VTE:
- Patients with AKI are in a hypercoagulable state.
- AKI alters the pharmacodynamics or pharmacokinetics of VTE prophylactic treatments.
- AKI is a marker of an illness that causes VTE.
- VTE leads to an increased rate of AKI rather than the other way around.
Dr. McMahon’s analysis also revealed that two other clinical conditions that are generally believed to raise VTE risk – obesity and impaired overall renal function identified with stagnant measures – did not correspond with a significantly elevated VTE rate in this study.
The data came from 6,552 adults hospitalized for at least 2 days at Walter Reed between September 2009 and March 2011. The study excluded patients with VTE at the time of admission and also those who had been treated with an anticoagulant at the time of admission. The patients averaged 55 years of age and were hospitalized for a median of 4 days. About 22% of patients received VTE prophylaxis with unfractionated heparin, about 41% received prophylaxis with low-molecular-weight heparin, and about 39% received no VTE prophylaxis (percentages total 102% because of rounding).
About 16% of the patients had been diagnosed with AKI at the time of admission, and an additional 8% developed AKI while hospitalized, defined as an increase in serum creatinine during hospitalization of at least 50% above baseline levels or an increase of more than 0.3 mg/dL above the level at time of admission. During hospitalization, 160 patients (2%) developed a new onset VTE.
In an analysis that adjusted for baseline differences in type of surgery, body mass index, sex, age, and prior hospitalizations during the prior 90 days, the results showed that patients with preexisting or new onset AKI had a 2.2-fold higher rate of VTE, compared with patients without AKI, and this difference was statistically significant, Dr. McMahon reported.
The analysis also showed a significant 62% relatively higher rate of VTE among soldiers hospitalized for a deployment-related event, as well as a significant 63% relatively lower VTE rate among patients not receiving medical prophylaxis, compared with patients receiving an anticoagulant. Dr. McMahon suggested that this lower rate of VTEs among patients not on prophylaxis reflected success in identifying which patients had an increased risk for VTE and hence received prophylaxis.
[email protected]
On Twitter @mitchelzoler
TORONTO – Hospitalized patients with acute kidney injury had more than double the inpatient rate of venous thromboembolism as had patients without acute kidney injury in a prospective, observational study of more than 6,000 hospitalized U.S. soldiers.
He offered four possible mechanisms to explain a link between AKI and VTE:
- Patients with AKI are in a hypercoagulable state.
- AKI alters the pharmacodynamics or pharmacokinetics of VTE prophylactic treatments.
- AKI is a marker of an illness that causes VTE.
- VTE leads to an increased rate of AKI rather than the other way around.
Dr. McMahon’s analysis also revealed that two other clinical conditions that are generally believed to raise VTE risk – obesity and impaired overall renal function identified with stagnant measures – did not correspond with a significantly elevated VTE rate in this study.
The data came from 6,552 adults hospitalized for at least 2 days at Walter Reed between September 2009 and March 2011. The study excluded patients with VTE at the time of admission and also those who had been treated with an anticoagulant at the time of admission. The patients averaged 55 years of age and were hospitalized for a median of 4 days. About 22% of patients received VTE prophylaxis with unfractionated heparin, about 41% received prophylaxis with low-molecular-weight heparin, and about 39% received no VTE prophylaxis (percentages total 102% because of rounding).
About 16% of the patients had been diagnosed with AKI at the time of admission, and an additional 8% developed AKI while hospitalized, defined as an increase in serum creatinine during hospitalization of at least 50% above baseline levels or an increase of more than 0.3 mg/dL above the level at time of admission. During hospitalization, 160 patients (2%) developed a new onset VTE.
In an analysis that adjusted for baseline differences in type of surgery, body mass index, sex, age, and prior hospitalizations during the prior 90 days, the results showed that patients with preexisting or new onset AKI had a 2.2-fold higher rate of VTE, compared with patients without AKI, and this difference was statistically significant, Dr. McMahon reported.
The analysis also showed a significant 62% relatively higher rate of VTE among soldiers hospitalized for a deployment-related event, as well as a significant 63% relatively lower VTE rate among patients not receiving medical prophylaxis, compared with patients receiving an anticoagulant. Dr. McMahon suggested that this lower rate of VTEs among patients not on prophylaxis reflected success in identifying which patients had an increased risk for VTE and hence received prophylaxis.
[email protected]
On Twitter @mitchelzoler
AT CHEST 2017
Key clinical point:
Major finding: Inpatients with AKI had an adjusted 2.2-fold higher rate of VTE, compared with other inpatients.
Data source: Prospective, observational data from 6,552 inpatients at a single U.S. military hospital.
Disclosures: Dr. McMahon had no disclosures.
Attack on asthma: Scrubbing homes of allergens may tame disease and its costs
After years of studying the causes of asthma, a pediatrician-turned-public health sleuth thinks there’s a way to substantially reduce its impact.
But the approach faces a big hurdle: getting someone to pay for it, said Elizabeth C. Matsui, MD, a professor at Johns Hopkins University in Baltimore.
Dr. Matsui, who suffered from asthma as a child, has spent much of her career studying the link between poor housing and asthma in low-income neighborhoods. In particular, she’s looked at the effects of mouse allergens, typically found in high concentrations in urban homes.
Dr. Matsui cited a 2004 study in the New England Journal of Medicine that described measures to reduce home allergen levels and concluded that they were linked to reductions in asthma symptoms.
That research “was highly successful and impactful,” but the approach wasn’t widely adopted, according to Dr. Matsui.
“So here we have this trial that was published more than 10 years ago that shows [indoor allergen control] works,” said Dr. Matsui, who did not participate in the study. “But the families who need it most can’t afford to do these things, don’t have control oftentimes over their home environment, and insurance or other payers don’t cover these things.”
Dr. Matsui has proposed new incentives for hospitals to provide home intervention, including Medicaid waivers. But, she said, scientists can’t use research money for these programs. “Delivery of community health care programs would require a different type of funding.”
As a result, doctors and scientists doubted if a plan to control home allergens would scale up, and insurers questioned whether benefits to their bottom line would justify the added cost.
“We have this enormous public health problem in that there are housing conditions that directly affect allergen exposure in this population of kids,” Dr. Matsui said. “We have dedicated individuals and groups who are trying to solve the problem. But we don’t have a system that is able to solve the problem.”
A 2017 study by Dr. Matsui, published in JAMA, suggests that even without intensive professional cleaning services, families that receive some training can substantially reduce home allergens on their own.
That finding suggests health agencies should routinely offer to educate asthma-affected families in home allergen control. “There’s potentially a large benefit,” Dr. Matsui said.
In a separate study, Dr. Matsui’s group is following 200 Baltimore children to see if those in homes scrubbed of allergens need fewer treatments with rescue inhalers. If they do, that could give health insurers an incentive to pay for the approach.
There’s another incentive: Clearing the air in a child’s home may be critical in cases where medications alone don’t work. “We continue to see a lot of kids that, despite being on medication, don’t have well-controlled asthma,” Dr. Matsui said.
Asthma drugs can also have serious side effects, she said, especially at higher doses, and may suppress symptoms without halting lung damage.
Dr. Matsui’s work on asthma began while working as a pediatrician at Baltimore’s Franklin Square Hospital in 1998. As part of her job, she spent a half-day each week in a school health clinic in a low-income area.
Dr. Matsui was struck by the number of kids she saw with severe asthma, she said, and set up a home health visit program to help them. But she wasn’t certain the program was working, so she consulted with experts at Hopkins.
In 2004, she earned a master’s from the Johns Hopkins School of Public Health. Today, she is one of the nation’s leading asthma researchers.
Dr. Matsui said her career was shaped by her own struggle with childhood asthma. “I think that that probably played a role, consciously or unconsciously,” she said.
KHN’s coverage of health disparities in east Baltimore is supported by The Annie E. Casey Foundation. Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
Environment is certainly a factor in asthma control. We are lucky enough to have a network in our county that will take pediatric asthma cases to help with asthma education. The nurse will even accompany the child and parent to their outpatient visits to help advocate for help with their asthma management and go into the home to see if there is a cockroach problem or a dusty environment, for example. They will also help families learn how to better organize the child’s bedroom so there is less dust! Some Medicaid HMOs will pay for this care management plan but not all of the insurances in our area.
Environment is certainly a factor in asthma control. We are lucky enough to have a network in our county that will take pediatric asthma cases to help with asthma education. The nurse will even accompany the child and parent to their outpatient visits to help advocate for help with their asthma management and go into the home to see if there is a cockroach problem or a dusty environment, for example. They will also help families learn how to better organize the child’s bedroom so there is less dust! Some Medicaid HMOs will pay for this care management plan but not all of the insurances in our area.
Environment is certainly a factor in asthma control. We are lucky enough to have a network in our county that will take pediatric asthma cases to help with asthma education. The nurse will even accompany the child and parent to their outpatient visits to help advocate for help with their asthma management and go into the home to see if there is a cockroach problem or a dusty environment, for example. They will also help families learn how to better organize the child’s bedroom so there is less dust! Some Medicaid HMOs will pay for this care management plan but not all of the insurances in our area.
After years of studying the causes of asthma, a pediatrician-turned-public health sleuth thinks there’s a way to substantially reduce its impact.
But the approach faces a big hurdle: getting someone to pay for it, said Elizabeth C. Matsui, MD, a professor at Johns Hopkins University in Baltimore.
Dr. Matsui, who suffered from asthma as a child, has spent much of her career studying the link between poor housing and asthma in low-income neighborhoods. In particular, she’s looked at the effects of mouse allergens, typically found in high concentrations in urban homes.
Dr. Matsui cited a 2004 study in the New England Journal of Medicine that described measures to reduce home allergen levels and concluded that they were linked to reductions in asthma symptoms.
That research “was highly successful and impactful,” but the approach wasn’t widely adopted, according to Dr. Matsui.
“So here we have this trial that was published more than 10 years ago that shows [indoor allergen control] works,” said Dr. Matsui, who did not participate in the study. “But the families who need it most can’t afford to do these things, don’t have control oftentimes over their home environment, and insurance or other payers don’t cover these things.”
Dr. Matsui has proposed new incentives for hospitals to provide home intervention, including Medicaid waivers. But, she said, scientists can’t use research money for these programs. “Delivery of community health care programs would require a different type of funding.”
As a result, doctors and scientists doubted if a plan to control home allergens would scale up, and insurers questioned whether benefits to their bottom line would justify the added cost.
“We have this enormous public health problem in that there are housing conditions that directly affect allergen exposure in this population of kids,” Dr. Matsui said. “We have dedicated individuals and groups who are trying to solve the problem. But we don’t have a system that is able to solve the problem.”
A 2017 study by Dr. Matsui, published in JAMA, suggests that even without intensive professional cleaning services, families that receive some training can substantially reduce home allergens on their own.
That finding suggests health agencies should routinely offer to educate asthma-affected families in home allergen control. “There’s potentially a large benefit,” Dr. Matsui said.
In a separate study, Dr. Matsui’s group is following 200 Baltimore children to see if those in homes scrubbed of allergens need fewer treatments with rescue inhalers. If they do, that could give health insurers an incentive to pay for the approach.
There’s another incentive: Clearing the air in a child’s home may be critical in cases where medications alone don’t work. “We continue to see a lot of kids that, despite being on medication, don’t have well-controlled asthma,” Dr. Matsui said.
Asthma drugs can also have serious side effects, she said, especially at higher doses, and may suppress symptoms without halting lung damage.
Dr. Matsui’s work on asthma began while working as a pediatrician at Baltimore’s Franklin Square Hospital in 1998. As part of her job, she spent a half-day each week in a school health clinic in a low-income area.
Dr. Matsui was struck by the number of kids she saw with severe asthma, she said, and set up a home health visit program to help them. But she wasn’t certain the program was working, so she consulted with experts at Hopkins.
In 2004, she earned a master’s from the Johns Hopkins School of Public Health. Today, she is one of the nation’s leading asthma researchers.
Dr. Matsui said her career was shaped by her own struggle with childhood asthma. “I think that that probably played a role, consciously or unconsciously,” she said.
KHN’s coverage of health disparities in east Baltimore is supported by The Annie E. Casey Foundation. Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
After years of studying the causes of asthma, a pediatrician-turned-public health sleuth thinks there’s a way to substantially reduce its impact.
But the approach faces a big hurdle: getting someone to pay for it, said Elizabeth C. Matsui, MD, a professor at Johns Hopkins University in Baltimore.
Dr. Matsui, who suffered from asthma as a child, has spent much of her career studying the link between poor housing and asthma in low-income neighborhoods. In particular, she’s looked at the effects of mouse allergens, typically found in high concentrations in urban homes.
Dr. Matsui cited a 2004 study in the New England Journal of Medicine that described measures to reduce home allergen levels and concluded that they were linked to reductions in asthma symptoms.
That research “was highly successful and impactful,” but the approach wasn’t widely adopted, according to Dr. Matsui.
“So here we have this trial that was published more than 10 years ago that shows [indoor allergen control] works,” said Dr. Matsui, who did not participate in the study. “But the families who need it most can’t afford to do these things, don’t have control oftentimes over their home environment, and insurance or other payers don’t cover these things.”
Dr. Matsui has proposed new incentives for hospitals to provide home intervention, including Medicaid waivers. But, she said, scientists can’t use research money for these programs. “Delivery of community health care programs would require a different type of funding.”
As a result, doctors and scientists doubted if a plan to control home allergens would scale up, and insurers questioned whether benefits to their bottom line would justify the added cost.
“We have this enormous public health problem in that there are housing conditions that directly affect allergen exposure in this population of kids,” Dr. Matsui said. “We have dedicated individuals and groups who are trying to solve the problem. But we don’t have a system that is able to solve the problem.”
A 2017 study by Dr. Matsui, published in JAMA, suggests that even without intensive professional cleaning services, families that receive some training can substantially reduce home allergens on their own.
That finding suggests health agencies should routinely offer to educate asthma-affected families in home allergen control. “There’s potentially a large benefit,” Dr. Matsui said.
In a separate study, Dr. Matsui’s group is following 200 Baltimore children to see if those in homes scrubbed of allergens need fewer treatments with rescue inhalers. If they do, that could give health insurers an incentive to pay for the approach.
There’s another incentive: Clearing the air in a child’s home may be critical in cases where medications alone don’t work. “We continue to see a lot of kids that, despite being on medication, don’t have well-controlled asthma,” Dr. Matsui said.
Asthma drugs can also have serious side effects, she said, especially at higher doses, and may suppress symptoms without halting lung damage.
Dr. Matsui’s work on asthma began while working as a pediatrician at Baltimore’s Franklin Square Hospital in 1998. As part of her job, she spent a half-day each week in a school health clinic in a low-income area.
Dr. Matsui was struck by the number of kids she saw with severe asthma, she said, and set up a home health visit program to help them. But she wasn’t certain the program was working, so she consulted with experts at Hopkins.
In 2004, she earned a master’s from the Johns Hopkins School of Public Health. Today, she is one of the nation’s leading asthma researchers.
Dr. Matsui said her career was shaped by her own struggle with childhood asthma. “I think that that probably played a role, consciously or unconsciously,” she said.
KHN’s coverage of health disparities in east Baltimore is supported by The Annie E. Casey Foundation. Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
More states allowing pharmacists to administer vaccines to younger patients
.
Since the 1990s, states have made laws to increase access to immunization services by giving pharmacists authority to give vaccines, said Cason D. Schmit, JD, of Texas A&M University at College Station, and Matthew S. Penn, JD, director of the Public Health Law Program at the Centers for Disease Control and Prevention, Atlanta. This has the advantage of pharmacies being open longer hours than most physicians’ offices, in addition to the opportunities for immunizing people in rural locations as well as those people without a regular physician.
Yet barriers to pharmacists’ providing immunization services remain because of some state laws. Laws in nine states that prevent pharmacists from vaccinating patients younger than age 18 years keep pharmacists from administering any of the vaccines listed in the 2016 Advisory Committee of Immunization Practices child immunization schedule. The two states in which pharmacists can vaccinate patients as young as 14 years allow the pharmacists to administer only the recommended booster for meningococcal vaccine and annual influenza vaccines for children. And the 15 states with minimum patient age restrictions for 7- to 12-year-oldss allow pharmacists to administer only the four vaccines ACIP recommends on the 2016 schedule for children (meningococcal, Tdap, human papillomavirus, and annual influenza vaccines).
Read more in the Journal of the American Pharmacists Association (2017 Nov-Dec;57[6]:661-9).
.
Since the 1990s, states have made laws to increase access to immunization services by giving pharmacists authority to give vaccines, said Cason D. Schmit, JD, of Texas A&M University at College Station, and Matthew S. Penn, JD, director of the Public Health Law Program at the Centers for Disease Control and Prevention, Atlanta. This has the advantage of pharmacies being open longer hours than most physicians’ offices, in addition to the opportunities for immunizing people in rural locations as well as those people without a regular physician.
Yet barriers to pharmacists’ providing immunization services remain because of some state laws. Laws in nine states that prevent pharmacists from vaccinating patients younger than age 18 years keep pharmacists from administering any of the vaccines listed in the 2016 Advisory Committee of Immunization Practices child immunization schedule. The two states in which pharmacists can vaccinate patients as young as 14 years allow the pharmacists to administer only the recommended booster for meningococcal vaccine and annual influenza vaccines for children. And the 15 states with minimum patient age restrictions for 7- to 12-year-oldss allow pharmacists to administer only the four vaccines ACIP recommends on the 2016 schedule for children (meningococcal, Tdap, human papillomavirus, and annual influenza vaccines).
Read more in the Journal of the American Pharmacists Association (2017 Nov-Dec;57[6]:661-9).
.
Since the 1990s, states have made laws to increase access to immunization services by giving pharmacists authority to give vaccines, said Cason D. Schmit, JD, of Texas A&M University at College Station, and Matthew S. Penn, JD, director of the Public Health Law Program at the Centers for Disease Control and Prevention, Atlanta. This has the advantage of pharmacies being open longer hours than most physicians’ offices, in addition to the opportunities for immunizing people in rural locations as well as those people without a regular physician.
Yet barriers to pharmacists’ providing immunization services remain because of some state laws. Laws in nine states that prevent pharmacists from vaccinating patients younger than age 18 years keep pharmacists from administering any of the vaccines listed in the 2016 Advisory Committee of Immunization Practices child immunization schedule. The two states in which pharmacists can vaccinate patients as young as 14 years allow the pharmacists to administer only the recommended booster for meningococcal vaccine and annual influenza vaccines for children. And the 15 states with minimum patient age restrictions for 7- to 12-year-oldss allow pharmacists to administer only the four vaccines ACIP recommends on the 2016 schedule for children (meningococcal, Tdap, human papillomavirus, and annual influenza vaccines).
Read more in the Journal of the American Pharmacists Association (2017 Nov-Dec;57[6]:661-9).
FROM THE JOURNAL OF THE AMERICAN PHARMACISTS ASSOCIATION