User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
COVID-19 plans put to test as firefighters crowd camps for peak wildfire season
Jon Paul was leery entering his first wildfire camp of the year late last month to fight three lightning-caused fires scorching parts of a Northern California forest that hadn’t burned in 40 years.

The 54-year-old engine captain from southern Oregon knew from experience that these crowded, grimy camps can be breeding grounds for norovirus and a respiratory illness that firefighters call the “camp crud” in a normal year. He wondered what the coronavirus would do in the tent cities where hundreds of men and women eat, sleep, wash, and spend their downtime between shifts.
Mr. Paul thought about his immunocompromised wife and his 84-year-old mother back home. Then he joined the approximately 1,300 people spread across the Modoc National Forest who would provide a major test for the COVID-prevention measures that had been developed for wildland firefighters.
“We’re still first responders and we have that responsibility to go and deal with these emergencies,” he said in a recent interview. “I don’t scare easy, but I’m very wary and concerned about my surroundings. I’m still going to work and do my job.”
Mr. Paul is one of thousands of firefighters from across the United States battling dozens of wildfires burning throughout the West. It’s an inherently dangerous job that now carries the additional risk of COVID-19 transmission. Any outbreak that ripples through a camp could easily sideline crews and spread the virus across multiple fires – and back to communities across the country – as personnel transfer in and out of “hot zones” and return home.
Though most firefighters are young and fit, some will inevitably fall ill in these remote makeshift communities of shared showers and portable toilets, where medical care can be limited. The pollutants in the smoke they breathe daily also make them more susceptible to COVID-19 and can worsen the effects of the disease, according to the Centers for Disease Control and Prevention.
Also, one suspected or positive case in a camp will mean many other firefighters will need to be quarantined, unable to work. The worst-case scenario is that multiple outbreaks could hamstring the nation’s ability to respond as wildfire season peaks in August, the hottest and driest month of the year in the western United States.
The number of acres burned so far this year is below the 10-year average, but the fire outlook for August is above average in nine states, according to the National Interagency Fire Center. Twenty-two large fires were ignited on Monday alone after lightning storms passed through the Northwest.
A study published this month by researchers at Colorado State University and the U.S. Forest Service’s Rocky Mountain Research Station concluded that COVID outbreaks “could be a serious threat to the firefighting mission” and urged vigilant social distancing and screening measures in the camps.
“If simultaneous fires incurred outbreaks, the entire wildland response system could be stressed substantially, with a large portion of the workforce quarantined,” the study’s authors wrote.
This spring, the National Wildfire Coordinating Group’s Fire Management Board wrote – and has since been updating – protocols to prevent the spread of COVID-19 in fire camps, based on CDC guidelines. Though they can be adapted by managers at different fires and even by individual team, they center on some key recommendations, including the following:
- Firefighters should be screened for fever and other COVID symptoms when they arrive at camp.
- Every crew should insulate itself as a “module of one” for the fire season and limit interactions with other crews.
- Firefighters should maintain social distancing and wear face coverings when social distancing isn’t possible. Smaller satellite camps, known as spike camps, can be built to ensure enough space.
- Shared areas should be regularly cleaned and disinfected, and sharing tools and radios should be minimized.
The guidance does not include routine testing of newly arrived firefighters – a practice used for athletes at training camps and students returning to college campuses.
The Fire Management Board’s Wildland Fire Medical and Public Health Advisory Team wrote in a July 2 memo that it “does not recommend utilizing universal COVID-19 laboratory testing as a standalone risk mitigation or screening measure among wildland firefighters.” Rather, the group recommends testing an individual and directly exposed coworkers, saying that approach is in line with CDC guidance.
The lack of testing capacity and long turnaround times are factors, according to Forest Service spokesperson Dan Hottle.
The exception is Alaska, where firefighters are tested upon arrival at the airport and are quarantined in a hotel while awaiting results, which come within 24 hours, Mr. Hottle said.
Fire crews responding to early-season fires in the spring had some problems adjusting to the new protocols, according to assessments written by fire leaders and compiled by the Wildland Fire Lessons Learned Center.
Shawn Faiella, superintendent of the interagency “hotshot crew” – so named because they work the most challenging or “hottest” parts of wildfires – based at Montana’s Lolo National Forest, questioned the need to wear masks inside vehicles and the safety of bringing extra vehicles to space out firefighters traveling to a blaze. Parking extra vehicles at the scene of a fire is difficult in tight dirt roads – and would be dangerous if evacuations are necessary, he wrote.
“It’s damn tough to take these practices to the fire line,” Mr. Faiella wrote after his team responded to a 40-acre Montana fire in April.
One recommendation that fire managers say has been particularly effective is the “module of one” concept requiring crews to eat and sleep together in isolation for the entire fire season.
“Whoever came up with it, it is working,” said Mike Goicoechea, the Montana-based incident commander for the Forest Service’s Northern Region Type 1 team, which manages the nation’s largest and most complex wildfires and natural disasters. “Somebody may test positive, and you end up having to take that module out of service for 14 days. But the nice part is you’re not taking out a whole camp. ... It’s just that module.”
The total number of positive COVID cases among wildland firefighters among the various federal, state, local, and tribal agencies is not being tracked. Each fire agency has its own system for tracking and reporting COVID-19, said Jessica Gardetto, a spokesperson for the Bureau of Land Management (BLM) and the National Interagency Fire Center in Idaho.
The largest wildland firefighting agency is the Department of Agriculture’s Forest Service, with 10,000 firefighters. Another major agency is the Department of the Interior, which BLM is part of and which had more than 3,500 full-time fire employees last year. As of the first week of August, 111 Forest Service firefighters and 40 BLM firefighters (who work underneath the broader Interior Department agency) had tested positive for COVID-19, according to officials for the respective agencies.
“Considering we’ve now been experiencing fire activity for several months, this number is surprisingly low if you think about the thousands of fire personnel who’ve been suppressing wildfires this summer,” Ms. Gardetto said.
Mr. Goicoechea and his Montana team traveled north of Tucson, Arizona, on June 22 to manage a rapidly spreading fire in the Santa Catalina Mountains that required 1,200 responders at its peak. Within 2 days of the team’s arrival, his managers were overwhelmed by calls from firefighters worried or with questions about preventing the spread of COVID-19 or carrying the virus home to their families.
In an unusual move, Mr. Goicoechea called upon Montana physician – and former National Park Service ranger with wildfire experience – Harry Sibold, MD, to join the team. Physicians are rarely, if ever, part of a wildfire camp’s medical team, Mr. Goicoechea said.
Dr. Sibold gave regular coronavirus updates during morning briefings, consulted with local health officials, soothed firefighters worried about bringing the virus home to their families, and advised fire managers on how to handle scenarios that might come up.
But Dr. Sibold said he wasn’t optimistic at the beginning about keeping the coronavirus in check in a large camp in Pima County, which has the second-highest number of confirmed cases in Arizona, at the time a national COVID-19 hot spot. “I quite firmly expected that we might have two or three outbreaks,” he said.
There were no positive cases during the team’s 2-week deployment, just three or four cases in which a firefighter showed symptoms but tested negative for the virus. After the Montana team returned home, nine firefighters at the Arizona fire from other units tested positive, Mr. Goicoechea said. Contact tracers notified the Montana team, some of whom were tested. All tests returned negative.
“I can’t say enough about having that doctor to help,” Mr. Goicoechea said, suggesting other teams might consider doing the same. “We’re not the experts in a pandemic. We’re the experts with fire.”
That early success will be tested as the number of fires increases across the West, along with the number of firefighters responding to them. There were more than 15,000 firefighters and support personnel assigned to fires across the nation as of mid-August, and the success of those COVID-19 prevention protocols depend largely on them.
Mr. Paul, the Oregon firefighter, said that the guidelines were followed closely in camp, but less so out on the fire line. It also appeared to him that younger firefighters were less likely to follow the masking and social-distancing rules than the veterans like him. That worried him as he realized it wouldn’t take much to spark an outbreak that could sideline crews and cripple the ability to respond to a fire.
“We’re outside, so it definitely helps with mitigation and makes it simpler to social distance,” Mr. Paul said. “But I think if there’s a mistake made, it could happen.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Jon Paul was leery entering his first wildfire camp of the year late last month to fight three lightning-caused fires scorching parts of a Northern California forest that hadn’t burned in 40 years.

The 54-year-old engine captain from southern Oregon knew from experience that these crowded, grimy camps can be breeding grounds for norovirus and a respiratory illness that firefighters call the “camp crud” in a normal year. He wondered what the coronavirus would do in the tent cities where hundreds of men and women eat, sleep, wash, and spend their downtime between shifts.
Mr. Paul thought about his immunocompromised wife and his 84-year-old mother back home. Then he joined the approximately 1,300 people spread across the Modoc National Forest who would provide a major test for the COVID-prevention measures that had been developed for wildland firefighters.
“We’re still first responders and we have that responsibility to go and deal with these emergencies,” he said in a recent interview. “I don’t scare easy, but I’m very wary and concerned about my surroundings. I’m still going to work and do my job.”
Mr. Paul is one of thousands of firefighters from across the United States battling dozens of wildfires burning throughout the West. It’s an inherently dangerous job that now carries the additional risk of COVID-19 transmission. Any outbreak that ripples through a camp could easily sideline crews and spread the virus across multiple fires – and back to communities across the country – as personnel transfer in and out of “hot zones” and return home.
Though most firefighters are young and fit, some will inevitably fall ill in these remote makeshift communities of shared showers and portable toilets, where medical care can be limited. The pollutants in the smoke they breathe daily also make them more susceptible to COVID-19 and can worsen the effects of the disease, according to the Centers for Disease Control and Prevention.
Also, one suspected or positive case in a camp will mean many other firefighters will need to be quarantined, unable to work. The worst-case scenario is that multiple outbreaks could hamstring the nation’s ability to respond as wildfire season peaks in August, the hottest and driest month of the year in the western United States.
The number of acres burned so far this year is below the 10-year average, but the fire outlook for August is above average in nine states, according to the National Interagency Fire Center. Twenty-two large fires were ignited on Monday alone after lightning storms passed through the Northwest.
A study published this month by researchers at Colorado State University and the U.S. Forest Service’s Rocky Mountain Research Station concluded that COVID outbreaks “could be a serious threat to the firefighting mission” and urged vigilant social distancing and screening measures in the camps.
“If simultaneous fires incurred outbreaks, the entire wildland response system could be stressed substantially, with a large portion of the workforce quarantined,” the study’s authors wrote.
This spring, the National Wildfire Coordinating Group’s Fire Management Board wrote – and has since been updating – protocols to prevent the spread of COVID-19 in fire camps, based on CDC guidelines. Though they can be adapted by managers at different fires and even by individual team, they center on some key recommendations, including the following:
- Firefighters should be screened for fever and other COVID symptoms when they arrive at camp.
- Every crew should insulate itself as a “module of one” for the fire season and limit interactions with other crews.
- Firefighters should maintain social distancing and wear face coverings when social distancing isn’t possible. Smaller satellite camps, known as spike camps, can be built to ensure enough space.
- Shared areas should be regularly cleaned and disinfected, and sharing tools and radios should be minimized.
The guidance does not include routine testing of newly arrived firefighters – a practice used for athletes at training camps and students returning to college campuses.
The Fire Management Board’s Wildland Fire Medical and Public Health Advisory Team wrote in a July 2 memo that it “does not recommend utilizing universal COVID-19 laboratory testing as a standalone risk mitigation or screening measure among wildland firefighters.” Rather, the group recommends testing an individual and directly exposed coworkers, saying that approach is in line with CDC guidance.
The lack of testing capacity and long turnaround times are factors, according to Forest Service spokesperson Dan Hottle.
The exception is Alaska, where firefighters are tested upon arrival at the airport and are quarantined in a hotel while awaiting results, which come within 24 hours, Mr. Hottle said.
Fire crews responding to early-season fires in the spring had some problems adjusting to the new protocols, according to assessments written by fire leaders and compiled by the Wildland Fire Lessons Learned Center.
Shawn Faiella, superintendent of the interagency “hotshot crew” – so named because they work the most challenging or “hottest” parts of wildfires – based at Montana’s Lolo National Forest, questioned the need to wear masks inside vehicles and the safety of bringing extra vehicles to space out firefighters traveling to a blaze. Parking extra vehicles at the scene of a fire is difficult in tight dirt roads – and would be dangerous if evacuations are necessary, he wrote.
“It’s damn tough to take these practices to the fire line,” Mr. Faiella wrote after his team responded to a 40-acre Montana fire in April.
One recommendation that fire managers say has been particularly effective is the “module of one” concept requiring crews to eat and sleep together in isolation for the entire fire season.
“Whoever came up with it, it is working,” said Mike Goicoechea, the Montana-based incident commander for the Forest Service’s Northern Region Type 1 team, which manages the nation’s largest and most complex wildfires and natural disasters. “Somebody may test positive, and you end up having to take that module out of service for 14 days. But the nice part is you’re not taking out a whole camp. ... It’s just that module.”
The total number of positive COVID cases among wildland firefighters among the various federal, state, local, and tribal agencies is not being tracked. Each fire agency has its own system for tracking and reporting COVID-19, said Jessica Gardetto, a spokesperson for the Bureau of Land Management (BLM) and the National Interagency Fire Center in Idaho.
The largest wildland firefighting agency is the Department of Agriculture’s Forest Service, with 10,000 firefighters. Another major agency is the Department of the Interior, which BLM is part of and which had more than 3,500 full-time fire employees last year. As of the first week of August, 111 Forest Service firefighters and 40 BLM firefighters (who work underneath the broader Interior Department agency) had tested positive for COVID-19, according to officials for the respective agencies.
“Considering we’ve now been experiencing fire activity for several months, this number is surprisingly low if you think about the thousands of fire personnel who’ve been suppressing wildfires this summer,” Ms. Gardetto said.
Mr. Goicoechea and his Montana team traveled north of Tucson, Arizona, on June 22 to manage a rapidly spreading fire in the Santa Catalina Mountains that required 1,200 responders at its peak. Within 2 days of the team’s arrival, his managers were overwhelmed by calls from firefighters worried or with questions about preventing the spread of COVID-19 or carrying the virus home to their families.
In an unusual move, Mr. Goicoechea called upon Montana physician – and former National Park Service ranger with wildfire experience – Harry Sibold, MD, to join the team. Physicians are rarely, if ever, part of a wildfire camp’s medical team, Mr. Goicoechea said.
Dr. Sibold gave regular coronavirus updates during morning briefings, consulted with local health officials, soothed firefighters worried about bringing the virus home to their families, and advised fire managers on how to handle scenarios that might come up.
But Dr. Sibold said he wasn’t optimistic at the beginning about keeping the coronavirus in check in a large camp in Pima County, which has the second-highest number of confirmed cases in Arizona, at the time a national COVID-19 hot spot. “I quite firmly expected that we might have two or three outbreaks,” he said.
There were no positive cases during the team’s 2-week deployment, just three or four cases in which a firefighter showed symptoms but tested negative for the virus. After the Montana team returned home, nine firefighters at the Arizona fire from other units tested positive, Mr. Goicoechea said. Contact tracers notified the Montana team, some of whom were tested. All tests returned negative.
“I can’t say enough about having that doctor to help,” Mr. Goicoechea said, suggesting other teams might consider doing the same. “We’re not the experts in a pandemic. We’re the experts with fire.”
That early success will be tested as the number of fires increases across the West, along with the number of firefighters responding to them. There were more than 15,000 firefighters and support personnel assigned to fires across the nation as of mid-August, and the success of those COVID-19 prevention protocols depend largely on them.
Mr. Paul, the Oregon firefighter, said that the guidelines were followed closely in camp, but less so out on the fire line. It also appeared to him that younger firefighters were less likely to follow the masking and social-distancing rules than the veterans like him. That worried him as he realized it wouldn’t take much to spark an outbreak that could sideline crews and cripple the ability to respond to a fire.
“We’re outside, so it definitely helps with mitigation and makes it simpler to social distance,” Mr. Paul said. “But I think if there’s a mistake made, it could happen.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Jon Paul was leery entering his first wildfire camp of the year late last month to fight three lightning-caused fires scorching parts of a Northern California forest that hadn’t burned in 40 years.

The 54-year-old engine captain from southern Oregon knew from experience that these crowded, grimy camps can be breeding grounds for norovirus and a respiratory illness that firefighters call the “camp crud” in a normal year. He wondered what the coronavirus would do in the tent cities where hundreds of men and women eat, sleep, wash, and spend their downtime between shifts.
Mr. Paul thought about his immunocompromised wife and his 84-year-old mother back home. Then he joined the approximately 1,300 people spread across the Modoc National Forest who would provide a major test for the COVID-prevention measures that had been developed for wildland firefighters.
“We’re still first responders and we have that responsibility to go and deal with these emergencies,” he said in a recent interview. “I don’t scare easy, but I’m very wary and concerned about my surroundings. I’m still going to work and do my job.”
Mr. Paul is one of thousands of firefighters from across the United States battling dozens of wildfires burning throughout the West. It’s an inherently dangerous job that now carries the additional risk of COVID-19 transmission. Any outbreak that ripples through a camp could easily sideline crews and spread the virus across multiple fires – and back to communities across the country – as personnel transfer in and out of “hot zones” and return home.
Though most firefighters are young and fit, some will inevitably fall ill in these remote makeshift communities of shared showers and portable toilets, where medical care can be limited. The pollutants in the smoke they breathe daily also make them more susceptible to COVID-19 and can worsen the effects of the disease, according to the Centers for Disease Control and Prevention.
Also, one suspected or positive case in a camp will mean many other firefighters will need to be quarantined, unable to work. The worst-case scenario is that multiple outbreaks could hamstring the nation’s ability to respond as wildfire season peaks in August, the hottest and driest month of the year in the western United States.
The number of acres burned so far this year is below the 10-year average, but the fire outlook for August is above average in nine states, according to the National Interagency Fire Center. Twenty-two large fires were ignited on Monday alone after lightning storms passed through the Northwest.
A study published this month by researchers at Colorado State University and the U.S. Forest Service’s Rocky Mountain Research Station concluded that COVID outbreaks “could be a serious threat to the firefighting mission” and urged vigilant social distancing and screening measures in the camps.
“If simultaneous fires incurred outbreaks, the entire wildland response system could be stressed substantially, with a large portion of the workforce quarantined,” the study’s authors wrote.
This spring, the National Wildfire Coordinating Group’s Fire Management Board wrote – and has since been updating – protocols to prevent the spread of COVID-19 in fire camps, based on CDC guidelines. Though they can be adapted by managers at different fires and even by individual team, they center on some key recommendations, including the following:
- Firefighters should be screened for fever and other COVID symptoms when they arrive at camp.
- Every crew should insulate itself as a “module of one” for the fire season and limit interactions with other crews.
- Firefighters should maintain social distancing and wear face coverings when social distancing isn’t possible. Smaller satellite camps, known as spike camps, can be built to ensure enough space.
- Shared areas should be regularly cleaned and disinfected, and sharing tools and radios should be minimized.
The guidance does not include routine testing of newly arrived firefighters – a practice used for athletes at training camps and students returning to college campuses.
The Fire Management Board’s Wildland Fire Medical and Public Health Advisory Team wrote in a July 2 memo that it “does not recommend utilizing universal COVID-19 laboratory testing as a standalone risk mitigation or screening measure among wildland firefighters.” Rather, the group recommends testing an individual and directly exposed coworkers, saying that approach is in line with CDC guidance.
The lack of testing capacity and long turnaround times are factors, according to Forest Service spokesperson Dan Hottle.
The exception is Alaska, where firefighters are tested upon arrival at the airport and are quarantined in a hotel while awaiting results, which come within 24 hours, Mr. Hottle said.
Fire crews responding to early-season fires in the spring had some problems adjusting to the new protocols, according to assessments written by fire leaders and compiled by the Wildland Fire Lessons Learned Center.
Shawn Faiella, superintendent of the interagency “hotshot crew” – so named because they work the most challenging or “hottest” parts of wildfires – based at Montana’s Lolo National Forest, questioned the need to wear masks inside vehicles and the safety of bringing extra vehicles to space out firefighters traveling to a blaze. Parking extra vehicles at the scene of a fire is difficult in tight dirt roads – and would be dangerous if evacuations are necessary, he wrote.
“It’s damn tough to take these practices to the fire line,” Mr. Faiella wrote after his team responded to a 40-acre Montana fire in April.
One recommendation that fire managers say has been particularly effective is the “module of one” concept requiring crews to eat and sleep together in isolation for the entire fire season.
“Whoever came up with it, it is working,” said Mike Goicoechea, the Montana-based incident commander for the Forest Service’s Northern Region Type 1 team, which manages the nation’s largest and most complex wildfires and natural disasters. “Somebody may test positive, and you end up having to take that module out of service for 14 days. But the nice part is you’re not taking out a whole camp. ... It’s just that module.”
The total number of positive COVID cases among wildland firefighters among the various federal, state, local, and tribal agencies is not being tracked. Each fire agency has its own system for tracking and reporting COVID-19, said Jessica Gardetto, a spokesperson for the Bureau of Land Management (BLM) and the National Interagency Fire Center in Idaho.
The largest wildland firefighting agency is the Department of Agriculture’s Forest Service, with 10,000 firefighters. Another major agency is the Department of the Interior, which BLM is part of and which had more than 3,500 full-time fire employees last year. As of the first week of August, 111 Forest Service firefighters and 40 BLM firefighters (who work underneath the broader Interior Department agency) had tested positive for COVID-19, according to officials for the respective agencies.
“Considering we’ve now been experiencing fire activity for several months, this number is surprisingly low if you think about the thousands of fire personnel who’ve been suppressing wildfires this summer,” Ms. Gardetto said.
Mr. Goicoechea and his Montana team traveled north of Tucson, Arizona, on June 22 to manage a rapidly spreading fire in the Santa Catalina Mountains that required 1,200 responders at its peak. Within 2 days of the team’s arrival, his managers were overwhelmed by calls from firefighters worried or with questions about preventing the spread of COVID-19 or carrying the virus home to their families.
In an unusual move, Mr. Goicoechea called upon Montana physician – and former National Park Service ranger with wildfire experience – Harry Sibold, MD, to join the team. Physicians are rarely, if ever, part of a wildfire camp’s medical team, Mr. Goicoechea said.
Dr. Sibold gave regular coronavirus updates during morning briefings, consulted with local health officials, soothed firefighters worried about bringing the virus home to their families, and advised fire managers on how to handle scenarios that might come up.
But Dr. Sibold said he wasn’t optimistic at the beginning about keeping the coronavirus in check in a large camp in Pima County, which has the second-highest number of confirmed cases in Arizona, at the time a national COVID-19 hot spot. “I quite firmly expected that we might have two or three outbreaks,” he said.
There were no positive cases during the team’s 2-week deployment, just three or four cases in which a firefighter showed symptoms but tested negative for the virus. After the Montana team returned home, nine firefighters at the Arizona fire from other units tested positive, Mr. Goicoechea said. Contact tracers notified the Montana team, some of whom were tested. All tests returned negative.
“I can’t say enough about having that doctor to help,” Mr. Goicoechea said, suggesting other teams might consider doing the same. “We’re not the experts in a pandemic. We’re the experts with fire.”
That early success will be tested as the number of fires increases across the West, along with the number of firefighters responding to them. There were more than 15,000 firefighters and support personnel assigned to fires across the nation as of mid-August, and the success of those COVID-19 prevention protocols depend largely on them.
Mr. Paul, the Oregon firefighter, said that the guidelines were followed closely in camp, but less so out on the fire line. It also appeared to him that younger firefighters were less likely to follow the masking and social-distancing rules than the veterans like him. That worried him as he realized it wouldn’t take much to spark an outbreak that could sideline crews and cripple the ability to respond to a fire.
“We’re outside, so it definitely helps with mitigation and makes it simpler to social distance,” Mr. Paul said. “But I think if there’s a mistake made, it could happen.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
Treatment for a tobacco-dependent adult
Applying American Thoracic Society’s new clinical practice guideline
Complications from tobacco use are the most common preventable cause of death, disability, and disease in the United States. Tobacco use causes 480,000 premature deaths every year. In pregnancy, tobacco use causes complications such as premature birth, intrauterine growth restriction, and placental abruption. In the perinatal period, it is associated with sudden infant death syndrome. While cigarette smoking is decreasing in adolescents, e-cigarette use in on the rise. Approximately 1,600 children aged 12-17 smoke their first cigarette every day and it is estimated that 5.6 million children and adolescents will die of a tobacco use–related death.1 For these reasons it is important to address tobacco use and cessation with patients whenever it is possible.
Case
A forty-five-year-old male who rarely comes to the office is here today for a physical exam at the urging of his partner. He has been smoking a pack a day since age 17. You have tried at past visits to discuss quitting, but he had been in the precontemplative stage and had been unwilling to consider any change. This visit, however, he is ready to try to quit. What can you offer him?
Core recommendations from ATS guidelines
This patient can be offered varenicline plus nicotine replacement therapy rather than nicotine replacement therapy, bupropion, e-cigarettes, or varenicline alone. His course of therapy should extend beyond 12 weeks instead of the standard 6- to 12-week therapy. Alternatively, he could be offered varenicline alone, rather than nicotine replacement.2
A change from previous guidelines
What makes this recommendation so interesting and new is the emphasis it places on varenicline. The United States Preventive Services Task Force released a recommendation statement in 2015 that stressed a combination of pharmacological and behavioral interventions. It discussed nicotine replacement therapy, bupropion, and varenicline, but did not recommend any one over any of the others.3 The new recommendation from the American Thoracic Society favors varenicline over other pharmacologic interventions. It is based on an independent systematic review of the literature that showed higher rates of tobacco use abstinence at the 6-month follow-up with varenicline alone versus nicotine replacement therapy alone, bupropion alone, or e-cigarette use only.
A review of 14 randomized controlled trials showed that varenicline improves abstinence rates during treatment by approximately 40% compared with nicotine replacement, and by 20% at the end of 6 months of treatment. The review found that varenicline plus nicotine replacement therapy is more effective than varenicline alone. In this comparison, based on three trials, there was a 36% higher abstinence rate at 6 months using varenicline plus nicotine replacement. When varenicline use was compared with use of a nicotine patch, bupropion, or e-cigarettes, there was a reduction in serious adverse events – changes in mood, suicidal ideation, and neurological side effects such as seizures.2 Clinicians may remember a black box warning on the varenicline label citing neuropsychiatric effects and it is important to note that the Food and Drug Administration removed this boxed warning in 2016.4
Opinion
This recommendation represents an important, evidence-based change from previous guidelines. It presents the opportunity for better outcomes, but will likely take a while to filter into practice, as clinicians need to become more comfortable with the use of varenicline and insurance supports the cost of varenicline.
The average cost of varenicline for 12 weeks is between $1,220 and $1,584. For comparison, nicotine replacement therapy costs $170 to $240 for the same number of weeks. To put those costs in perspective, the 12-week cost of cigarettes for a two-pack-a-day smoker is approximately $1,000.
For some patients, the motivation to quit smoking comes from the realization of how much they are spending on cigarettes each month. That said, if a patient does not have insurance or their insurance does not cover the cost of varenicline, nicotine replacement therapy might be more appealing. It should be noted that better abstinence rates have been seen in patients taking varenicline plus nicotine replacement therapy versus varenicline alone.
Suggested treatment
Based on a systematic review of randomized controlled trials, the American Thoracic Society’s guideline on pharmacological treatment in tobacco-dependent adults concludes that varenicline plus nicotine patch is the preferred pharmacological treatment for tobacco cessation when compared with varenicline alone, bupropion alone, nicotine replacement therapy alone, and e-cigarettes alone. If the patient does not want to start two medicines at once, then varenicline alone would be the preferred choice.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. Dr. Sprogell is a third-year resident in the family medicine residency program at Abington Jefferson Health. They have no conflicts related to the content of this piece. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. U.S. Preventive Services Task Force. Primary care interventions for prevention and cessation of tobacco use in children and adolescents: U.S. Preventive Services Task Force Recommendation Statement. JAMA.2020;323(16):1590-8. doi: 10.1001/jama.2020.4679.
2. Leone FT et al. Initiating pharmacologic treatment in tobacco-dependent adults: An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;202(2):e5–e31.
3. Tobacco smoking cessation in adults, including pregnant women: Behavioral and pharmacotherapy interventions. U.S. Preventive Services Task Force 2015 Sep 21.
4. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. 2016 Dec. 16.
Applying American Thoracic Society’s new clinical practice guideline
Applying American Thoracic Society’s new clinical practice guideline
Complications from tobacco use are the most common preventable cause of death, disability, and disease in the United States. Tobacco use causes 480,000 premature deaths every year. In pregnancy, tobacco use causes complications such as premature birth, intrauterine growth restriction, and placental abruption. In the perinatal period, it is associated with sudden infant death syndrome. While cigarette smoking is decreasing in adolescents, e-cigarette use in on the rise. Approximately 1,600 children aged 12-17 smoke their first cigarette every day and it is estimated that 5.6 million children and adolescents will die of a tobacco use–related death.1 For these reasons it is important to address tobacco use and cessation with patients whenever it is possible.
Case
A forty-five-year-old male who rarely comes to the office is here today for a physical exam at the urging of his partner. He has been smoking a pack a day since age 17. You have tried at past visits to discuss quitting, but he had been in the precontemplative stage and had been unwilling to consider any change. This visit, however, he is ready to try to quit. What can you offer him?
Core recommendations from ATS guidelines
This patient can be offered varenicline plus nicotine replacement therapy rather than nicotine replacement therapy, bupropion, e-cigarettes, or varenicline alone. His course of therapy should extend beyond 12 weeks instead of the standard 6- to 12-week therapy. Alternatively, he could be offered varenicline alone, rather than nicotine replacement.2
A change from previous guidelines
What makes this recommendation so interesting and new is the emphasis it places on varenicline. The United States Preventive Services Task Force released a recommendation statement in 2015 that stressed a combination of pharmacological and behavioral interventions. It discussed nicotine replacement therapy, bupropion, and varenicline, but did not recommend any one over any of the others.3 The new recommendation from the American Thoracic Society favors varenicline over other pharmacologic interventions. It is based on an independent systematic review of the literature that showed higher rates of tobacco use abstinence at the 6-month follow-up with varenicline alone versus nicotine replacement therapy alone, bupropion alone, or e-cigarette use only.
A review of 14 randomized controlled trials showed that varenicline improves abstinence rates during treatment by approximately 40% compared with nicotine replacement, and by 20% at the end of 6 months of treatment. The review found that varenicline plus nicotine replacement therapy is more effective than varenicline alone. In this comparison, based on three trials, there was a 36% higher abstinence rate at 6 months using varenicline plus nicotine replacement. When varenicline use was compared with use of a nicotine patch, bupropion, or e-cigarettes, there was a reduction in serious adverse events – changes in mood, suicidal ideation, and neurological side effects such as seizures.2 Clinicians may remember a black box warning on the varenicline label citing neuropsychiatric effects and it is important to note that the Food and Drug Administration removed this boxed warning in 2016.4
Opinion
This recommendation represents an important, evidence-based change from previous guidelines. It presents the opportunity for better outcomes, but will likely take a while to filter into practice, as clinicians need to become more comfortable with the use of varenicline and insurance supports the cost of varenicline.
The average cost of varenicline for 12 weeks is between $1,220 and $1,584. For comparison, nicotine replacement therapy costs $170 to $240 for the same number of weeks. To put those costs in perspective, the 12-week cost of cigarettes for a two-pack-a-day smoker is approximately $1,000.
For some patients, the motivation to quit smoking comes from the realization of how much they are spending on cigarettes each month. That said, if a patient does not have insurance or their insurance does not cover the cost of varenicline, nicotine replacement therapy might be more appealing. It should be noted that better abstinence rates have been seen in patients taking varenicline plus nicotine replacement therapy versus varenicline alone.
Suggested treatment
Based on a systematic review of randomized controlled trials, the American Thoracic Society’s guideline on pharmacological treatment in tobacco-dependent adults concludes that varenicline plus nicotine patch is the preferred pharmacological treatment for tobacco cessation when compared with varenicline alone, bupropion alone, nicotine replacement therapy alone, and e-cigarettes alone. If the patient does not want to start two medicines at once, then varenicline alone would be the preferred choice.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. Dr. Sprogell is a third-year resident in the family medicine residency program at Abington Jefferson Health. They have no conflicts related to the content of this piece. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. U.S. Preventive Services Task Force. Primary care interventions for prevention and cessation of tobacco use in children and adolescents: U.S. Preventive Services Task Force Recommendation Statement. JAMA.2020;323(16):1590-8. doi: 10.1001/jama.2020.4679.
2. Leone FT et al. Initiating pharmacologic treatment in tobacco-dependent adults: An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;202(2):e5–e31.
3. Tobacco smoking cessation in adults, including pregnant women: Behavioral and pharmacotherapy interventions. U.S. Preventive Services Task Force 2015 Sep 21.
4. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. 2016 Dec. 16.
Complications from tobacco use are the most common preventable cause of death, disability, and disease in the United States. Tobacco use causes 480,000 premature deaths every year. In pregnancy, tobacco use causes complications such as premature birth, intrauterine growth restriction, and placental abruption. In the perinatal period, it is associated with sudden infant death syndrome. While cigarette smoking is decreasing in adolescents, e-cigarette use in on the rise. Approximately 1,600 children aged 12-17 smoke their first cigarette every day and it is estimated that 5.6 million children and adolescents will die of a tobacco use–related death.1 For these reasons it is important to address tobacco use and cessation with patients whenever it is possible.
Case
A forty-five-year-old male who rarely comes to the office is here today for a physical exam at the urging of his partner. He has been smoking a pack a day since age 17. You have tried at past visits to discuss quitting, but he had been in the precontemplative stage and had been unwilling to consider any change. This visit, however, he is ready to try to quit. What can you offer him?
Core recommendations from ATS guidelines
This patient can be offered varenicline plus nicotine replacement therapy rather than nicotine replacement therapy, bupropion, e-cigarettes, or varenicline alone. His course of therapy should extend beyond 12 weeks instead of the standard 6- to 12-week therapy. Alternatively, he could be offered varenicline alone, rather than nicotine replacement.2
A change from previous guidelines
What makes this recommendation so interesting and new is the emphasis it places on varenicline. The United States Preventive Services Task Force released a recommendation statement in 2015 that stressed a combination of pharmacological and behavioral interventions. It discussed nicotine replacement therapy, bupropion, and varenicline, but did not recommend any one over any of the others.3 The new recommendation from the American Thoracic Society favors varenicline over other pharmacologic interventions. It is based on an independent systematic review of the literature that showed higher rates of tobacco use abstinence at the 6-month follow-up with varenicline alone versus nicotine replacement therapy alone, bupropion alone, or e-cigarette use only.
A review of 14 randomized controlled trials showed that varenicline improves abstinence rates during treatment by approximately 40% compared with nicotine replacement, and by 20% at the end of 6 months of treatment. The review found that varenicline plus nicotine replacement therapy is more effective than varenicline alone. In this comparison, based on three trials, there was a 36% higher abstinence rate at 6 months using varenicline plus nicotine replacement. When varenicline use was compared with use of a nicotine patch, bupropion, or e-cigarettes, there was a reduction in serious adverse events – changes in mood, suicidal ideation, and neurological side effects such as seizures.2 Clinicians may remember a black box warning on the varenicline label citing neuropsychiatric effects and it is important to note that the Food and Drug Administration removed this boxed warning in 2016.4
Opinion
This recommendation represents an important, evidence-based change from previous guidelines. It presents the opportunity for better outcomes, but will likely take a while to filter into practice, as clinicians need to become more comfortable with the use of varenicline and insurance supports the cost of varenicline.
The average cost of varenicline for 12 weeks is between $1,220 and $1,584. For comparison, nicotine replacement therapy costs $170 to $240 for the same number of weeks. To put those costs in perspective, the 12-week cost of cigarettes for a two-pack-a-day smoker is approximately $1,000.
For some patients, the motivation to quit smoking comes from the realization of how much they are spending on cigarettes each month. That said, if a patient does not have insurance or their insurance does not cover the cost of varenicline, nicotine replacement therapy might be more appealing. It should be noted that better abstinence rates have been seen in patients taking varenicline plus nicotine replacement therapy versus varenicline alone.
Suggested treatment
Based on a systematic review of randomized controlled trials, the American Thoracic Society’s guideline on pharmacological treatment in tobacco-dependent adults concludes that varenicline plus nicotine patch is the preferred pharmacological treatment for tobacco cessation when compared with varenicline alone, bupropion alone, nicotine replacement therapy alone, and e-cigarettes alone. If the patient does not want to start two medicines at once, then varenicline alone would be the preferred choice.
Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. Dr. Sprogell is a third-year resident in the family medicine residency program at Abington Jefferson Health. They have no conflicts related to the content of this piece. For questions or comments, feel free to contact Dr. Skolnik on Twitter @NeilSkolnik.
References
1. U.S. Preventive Services Task Force. Primary care interventions for prevention and cessation of tobacco use in children and adolescents: U.S. Preventive Services Task Force Recommendation Statement. JAMA.2020;323(16):1590-8. doi: 10.1001/jama.2020.4679.
2. Leone FT et al. Initiating pharmacologic treatment in tobacco-dependent adults: An official American Thoracic Society Clinical Practice Guideline. Am J Respir Crit Care Med. 2020;202(2):e5–e31.
3. Tobacco smoking cessation in adults, including pregnant women: Behavioral and pharmacotherapy interventions. U.S. Preventive Services Task Force 2015 Sep 21.
4. FDA Drug Safety Communication: FDA revises description of mental health side effects of the stop-smoking medicines Chantix (varenicline) and Zyban (bupropion) to reflect clinical trial findings. 2016 Dec. 16.
COVID-19: Optimizing therapeutic strategies for children, adolescents with ADHD
Recently, the Yakima Health District (YHD), in collaboration with the Washington State Department of Health, issued dramatic revisions to its educational curriculum, opting for exclusively remote learning as an important next step in COVID-19 containment measures.
The newly implemented “enhanced” distance-learning paradigm has garnered considerable national attention. Even more noteworthy is how YHD addressed those with language barriers and learning differences such as ADHD as a “priority group”; these individuals are exempt from the newly implemented measures, and small instructional groups of no more than five “at-risk” students will be directly supervised by specialized educators.1,2 To overcome these new unprecedented challenges from the coronavirus pandemic, especially from the perspective of distance education and mental health for susceptible groups such as those with ADHD, it is of utmost importance to explore various programs of interest, as well as the targeted therapies being considered during this crisis.
From a therapeutic standpoint, individuals with learning differences are more likely to play catch-up with their age-matched peers. This puts them at significant risk for developmental delays with symptoms manifesting as disruptive behavioral issues. This is why ongoing parental guidance, coupled with a paradoxically stimulating environment, is critical for children and adolescents with ADHD.3 Accumulating evidence, based on a myriad of studies, demonstrates that childhood treatment with ADHD stimulants reduces the incidence of future substance use, as well as that of other negative outcomes.4,5
Therapeutic strategies that work
“The new normal” has forced unique challenges on clinicians for mitigating distress by novel means of health care delivery. Given the paucity of research exploring the interactions of individuals with ADHD within the context of COVID-19, Take for example, the suggested guidelines from the European ADHD Guidelines Group (EAGG) – such as the following:
- Telecommunications in general, and telepsychiatry in particular, should function as the primary mode of health care delivery to fulfill societal standards of physical distancing.
- Children and adolescents with ADHD should be designated as a “priority group” with respect to monitoring initiatives by educators in a school setting, be it virtual or otherwise.
- Implementation of behavioral strategies by parent or guardian to address psychological well-being and reduce the presence of comorbid behavioral conditions (such as oppositional defiant disorder).
In addition to the aforementioned guidance, EAGG maintains that individuals with ADHD may be initiated on medications after the completion of a baseline examination; if the patients in question are already on a treatment regimen, they should proceed with it as indicated. Interruptions to therapy are not ideal because patients are then subjected to health-related stressors of COVID-19. Reasonable regulations concerning access to medications, without unnecessary delays, undoubtedly will facilitate patient needs, allowing for a smooth transition in day-to-day activities. The family, as a cohesive unit, may benefit from reeducation because it contributes toward the therapeutic process. Neurofeedback, coping skills, and cognitive restructuring training are potential modalities that can augment medications.
Although it may seem counterintuitive, parents or caregivers should resist the urge to increase the medication dose during an outbreak with the intended goal of diminishing the psychosocial burden of ADHD symptomatology. Likewise, unless indicated by a specialist, antipsychotics and/or hypnotics should not be introduced for addressing behavioral dysregulation (such as agitation) during the confinement period.
Historically, numerous clinicians have suggested that patients undergo a routine cardiovascular examination and EKG before being prescribed psychostimulants (the rationale for this recommendation is that sympathomimetics unduly affect blood pressure and heart rate).6,7 However, the American Academy of Pediatrics (AAP) and the American Heart Association (AHA) eventually amended their previous stance by releasing a joint statement in which they deemed a baseline EKG necessary only in ADHD patients with preexisting cardiac risk. For all other patients, the use of EKGs was entirely contingent on physician discretion. However, given the nature of safety precautions for COVID-19, it is prudent to discourage or delay in-person cardiovascular examination/monitoring protocols altogether, especially in those patients without known heart conditions.
Another area of concern is sleep dysfunction, which might exist as an untoward effect of ADHD medication intake or because of the presence of COVID-19 psychosocial stressors. However, clinicians advise that unnecessary psychopharmacology (such as hypnotics or melatonin) be avoided. Instead, conservative lifestyle measures should be enacted, emphasizing the role of proper sleep hygiene in maintaining optimal behavioral health. Despite setbacks to in-person appointments, patients are expected to continue their pharmacotherapy with “parent-focused” ADHD interventions taking a primary role in facilitating compliance through remote monitoring.
ADMiRE, a tertiary-level, dedicated ADHD intervention program from South Dublin, Ireland, has identified several roadblocks with respect to streamlining health care for individuals with ADHD during the confinement period. The proposed resolution to these issues, some of which are derived from EAGG guidelines, might have universal applications elsewhere, thereby facilitating the development of therapeutic services of interest. ADMiRE has noted a correspondence between the guidelines established by EAGG and that of the Canadian ADHD Resource Alliance (CADDRA), including minimal in-person interactions (in favor of virtual teleconferencing) and a cardiovascular screen can be performed in lieu of baseline cardiac auscultation. Moreover, in the event that the patient is a low cardiac risk candidate for ADHD treatment, monitoring protocols may be continued from a home setting. However, if a physical examination is indicated, CADDRA recommends the use of precautionary PPE before commencing ADHD pharmacotherapy.
One of the most significant hurdles is that of school closures because teacher feedback for baseline behavior was traditionally instrumental in dictating patient medical management (for example, for titration schedule). It is expected that, for the time being, this role will be supplanted by parental reports. As well as disclosing information on behavioral dysregulation, family members should be trained to relay critical information about the development of stimulant-induced cardiovascular symptoms – namely, dyspnea, chest pain, and/or palpitations. Furthermore, as primary caregivers, parents should harbor a certain degree of emotional sensitivity because their mood state may influence the child’s overall behavioral course in terms of symptom exacerbation.8
Toward adopting an integrated model for care
Developing an effective assessment plan for patients with ADHD often proves to be a challenging task for clinicians, perhaps even more so in environments that enforce social distancing and limited physical contact by default. As a neurodevelopmental disorder from childhood, the symptoms (including inattention, hyperactivity, and/or impulsivity) of ADHD do not arise in a vacuum – comorbid conditions include mood and anxiety disorders, which are complicated further by a background risk for substance use and self-medicating tendencies.9 Unfortunately, the pandemic has limited the breadth of non-COVID doctors visits, which hinders the overall diagnostic and monitoring process for identifiable comorbid conditions, such as autism spectrum disorder, intellectual disability, oppositional defiant and conduct disorders, and so on.10 Since ADHD symptoms cannot be treated by pharmacotherapy or behavioral interventions alone, our team advocates that families provide additional emotional support and continuous encouragement during these uncertain times.
ADHD and the self-medication hypothesis
The Khantzian self-medication hypothesis posits that a drug seeker may subconsciously gravitate toward a particular agent only to discover a sense of relief concerning inner turmoil or restlessness after use. Observations support the notion that individuals with undiagnosed ADHD have sought cocaine or even recreational designer drugs (such as methylenedioxypyrovalerone, or “bath salts”).11 Given the similar mechanism of action between cocaine, methylenedioxypyrovalerone, and prescribed psychostimulants such as methylphenidate, the results are hardly surprising because these agents all work on the brain’s “reward center” (for example, the nucleus accumbens) by invoking dopamine release. Aside from the aforementioned self-medication hypothesis, “downers” such as Xanax recently have experienced a prescription spike during the outbreak. While there isn’t an immediate cause for concern of Xanax abuse in ADHD individuals, the potential for addiction is certainly real, especially when taking into account comorbid anxiety disorder or sleep dysfunction.
Because of limited resources and precautionary guidelines, clinicians are at a considerable disadvantage in terms of formulating a comprehensive diagnostic and treatment plan for children and adolescents with ADHD. This situation is further compounded by the recent closure of schools and the lack of feedback with respect to baseline behavior from teachers and specialized educators. This is why it is imperative for primary caregivers to closely monitor children with ADHD for developing changes in behavioral patterns (for example, mood or anxiety issues and drug-seeking or disruptive behavior) and work with health care professionals.
References
1. “Distance learning strongly recommended for all Yakima county schools.” NBC Right Now. 2020 Aug 5.
2. Retka J. “Enhanced” remote learning in Yakima county schools? What that means for students this fall. Yakima Herald-Republic. 2020 Aug 8.
3. Armstrong T. “To empower! Not Control! A holistic approach to ADHD.” American Institute for Learning and Development. 1998.
4. J Child Psychol Psychiatry. 2014 Aug;55(8):878-85.
5. Ir J Psychol Med. 2020 May 21:1-22.
6. Lancet Child Adolesc Health. 2020 Jun;4(6):412-4.
7. O’Keefe L. AAP News. 2008 Jun;29(6):1.
8. Asian J Psychiatr. 2020 Jun;51:102077.
9. Current Psychiatry. 2015 Dec;14(12):e3-4.
10. Encephale. 2020 Jun 7;46(3S):S85-92.
11. Current Psychiatry. 2014 Dec; 3(12): e3-4.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation (IMCHF), Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry is the chief scientific officer and head of the department of mental health and clini-cal research at the IMCHF and is Mr. Choudhry’s father. He has no disclosures.
Recently, the Yakima Health District (YHD), in collaboration with the Washington State Department of Health, issued dramatic revisions to its educational curriculum, opting for exclusively remote learning as an important next step in COVID-19 containment measures.
The newly implemented “enhanced” distance-learning paradigm has garnered considerable national attention. Even more noteworthy is how YHD addressed those with language barriers and learning differences such as ADHD as a “priority group”; these individuals are exempt from the newly implemented measures, and small instructional groups of no more than five “at-risk” students will be directly supervised by specialized educators.1,2 To overcome these new unprecedented challenges from the coronavirus pandemic, especially from the perspective of distance education and mental health for susceptible groups such as those with ADHD, it is of utmost importance to explore various programs of interest, as well as the targeted therapies being considered during this crisis.
From a therapeutic standpoint, individuals with learning differences are more likely to play catch-up with their age-matched peers. This puts them at significant risk for developmental delays with symptoms manifesting as disruptive behavioral issues. This is why ongoing parental guidance, coupled with a paradoxically stimulating environment, is critical for children and adolescents with ADHD.3 Accumulating evidence, based on a myriad of studies, demonstrates that childhood treatment with ADHD stimulants reduces the incidence of future substance use, as well as that of other negative outcomes.4,5
Therapeutic strategies that work
“The new normal” has forced unique challenges on clinicians for mitigating distress by novel means of health care delivery. Given the paucity of research exploring the interactions of individuals with ADHD within the context of COVID-19, Take for example, the suggested guidelines from the European ADHD Guidelines Group (EAGG) – such as the following:
- Telecommunications in general, and telepsychiatry in particular, should function as the primary mode of health care delivery to fulfill societal standards of physical distancing.
- Children and adolescents with ADHD should be designated as a “priority group” with respect to monitoring initiatives by educators in a school setting, be it virtual or otherwise.
- Implementation of behavioral strategies by parent or guardian to address psychological well-being and reduce the presence of comorbid behavioral conditions (such as oppositional defiant disorder).
In addition to the aforementioned guidance, EAGG maintains that individuals with ADHD may be initiated on medications after the completion of a baseline examination; if the patients in question are already on a treatment regimen, they should proceed with it as indicated. Interruptions to therapy are not ideal because patients are then subjected to health-related stressors of COVID-19. Reasonable regulations concerning access to medications, without unnecessary delays, undoubtedly will facilitate patient needs, allowing for a smooth transition in day-to-day activities. The family, as a cohesive unit, may benefit from reeducation because it contributes toward the therapeutic process. Neurofeedback, coping skills, and cognitive restructuring training are potential modalities that can augment medications.
Although it may seem counterintuitive, parents or caregivers should resist the urge to increase the medication dose during an outbreak with the intended goal of diminishing the psychosocial burden of ADHD symptomatology. Likewise, unless indicated by a specialist, antipsychotics and/or hypnotics should not be introduced for addressing behavioral dysregulation (such as agitation) during the confinement period.
Historically, numerous clinicians have suggested that patients undergo a routine cardiovascular examination and EKG before being prescribed psychostimulants (the rationale for this recommendation is that sympathomimetics unduly affect blood pressure and heart rate).6,7 However, the American Academy of Pediatrics (AAP) and the American Heart Association (AHA) eventually amended their previous stance by releasing a joint statement in which they deemed a baseline EKG necessary only in ADHD patients with preexisting cardiac risk. For all other patients, the use of EKGs was entirely contingent on physician discretion. However, given the nature of safety precautions for COVID-19, it is prudent to discourage or delay in-person cardiovascular examination/monitoring protocols altogether, especially in those patients without known heart conditions.
Another area of concern is sleep dysfunction, which might exist as an untoward effect of ADHD medication intake or because of the presence of COVID-19 psychosocial stressors. However, clinicians advise that unnecessary psychopharmacology (such as hypnotics or melatonin) be avoided. Instead, conservative lifestyle measures should be enacted, emphasizing the role of proper sleep hygiene in maintaining optimal behavioral health. Despite setbacks to in-person appointments, patients are expected to continue their pharmacotherapy with “parent-focused” ADHD interventions taking a primary role in facilitating compliance through remote monitoring.
ADMiRE, a tertiary-level, dedicated ADHD intervention program from South Dublin, Ireland, has identified several roadblocks with respect to streamlining health care for individuals with ADHD during the confinement period. The proposed resolution to these issues, some of which are derived from EAGG guidelines, might have universal applications elsewhere, thereby facilitating the development of therapeutic services of interest. ADMiRE has noted a correspondence between the guidelines established by EAGG and that of the Canadian ADHD Resource Alliance (CADDRA), including minimal in-person interactions (in favor of virtual teleconferencing) and a cardiovascular screen can be performed in lieu of baseline cardiac auscultation. Moreover, in the event that the patient is a low cardiac risk candidate for ADHD treatment, monitoring protocols may be continued from a home setting. However, if a physical examination is indicated, CADDRA recommends the use of precautionary PPE before commencing ADHD pharmacotherapy.
One of the most significant hurdles is that of school closures because teacher feedback for baseline behavior was traditionally instrumental in dictating patient medical management (for example, for titration schedule). It is expected that, for the time being, this role will be supplanted by parental reports. As well as disclosing information on behavioral dysregulation, family members should be trained to relay critical information about the development of stimulant-induced cardiovascular symptoms – namely, dyspnea, chest pain, and/or palpitations. Furthermore, as primary caregivers, parents should harbor a certain degree of emotional sensitivity because their mood state may influence the child’s overall behavioral course in terms of symptom exacerbation.8
Toward adopting an integrated model for care
Developing an effective assessment plan for patients with ADHD often proves to be a challenging task for clinicians, perhaps even more so in environments that enforce social distancing and limited physical contact by default. As a neurodevelopmental disorder from childhood, the symptoms (including inattention, hyperactivity, and/or impulsivity) of ADHD do not arise in a vacuum – comorbid conditions include mood and anxiety disorders, which are complicated further by a background risk for substance use and self-medicating tendencies.9 Unfortunately, the pandemic has limited the breadth of non-COVID doctors visits, which hinders the overall diagnostic and monitoring process for identifiable comorbid conditions, such as autism spectrum disorder, intellectual disability, oppositional defiant and conduct disorders, and so on.10 Since ADHD symptoms cannot be treated by pharmacotherapy or behavioral interventions alone, our team advocates that families provide additional emotional support and continuous encouragement during these uncertain times.
ADHD and the self-medication hypothesis
The Khantzian self-medication hypothesis posits that a drug seeker may subconsciously gravitate toward a particular agent only to discover a sense of relief concerning inner turmoil or restlessness after use. Observations support the notion that individuals with undiagnosed ADHD have sought cocaine or even recreational designer drugs (such as methylenedioxypyrovalerone, or “bath salts”).11 Given the similar mechanism of action between cocaine, methylenedioxypyrovalerone, and prescribed psychostimulants such as methylphenidate, the results are hardly surprising because these agents all work on the brain’s “reward center” (for example, the nucleus accumbens) by invoking dopamine release. Aside from the aforementioned self-medication hypothesis, “downers” such as Xanax recently have experienced a prescription spike during the outbreak. While there isn’t an immediate cause for concern of Xanax abuse in ADHD individuals, the potential for addiction is certainly real, especially when taking into account comorbid anxiety disorder or sleep dysfunction.
Because of limited resources and precautionary guidelines, clinicians are at a considerable disadvantage in terms of formulating a comprehensive diagnostic and treatment plan for children and adolescents with ADHD. This situation is further compounded by the recent closure of schools and the lack of feedback with respect to baseline behavior from teachers and specialized educators. This is why it is imperative for primary caregivers to closely monitor children with ADHD for developing changes in behavioral patterns (for example, mood or anxiety issues and drug-seeking or disruptive behavior) and work with health care professionals.
References
1. “Distance learning strongly recommended for all Yakima county schools.” NBC Right Now. 2020 Aug 5.
2. Retka J. “Enhanced” remote learning in Yakima county schools? What that means for students this fall. Yakima Herald-Republic. 2020 Aug 8.
3. Armstrong T. “To empower! Not Control! A holistic approach to ADHD.” American Institute for Learning and Development. 1998.
4. J Child Psychol Psychiatry. 2014 Aug;55(8):878-85.
5. Ir J Psychol Med. 2020 May 21:1-22.
6. Lancet Child Adolesc Health. 2020 Jun;4(6):412-4.
7. O’Keefe L. AAP News. 2008 Jun;29(6):1.
8. Asian J Psychiatr. 2020 Jun;51:102077.
9. Current Psychiatry. 2015 Dec;14(12):e3-4.
10. Encephale. 2020 Jun 7;46(3S):S85-92.
11. Current Psychiatry. 2014 Dec; 3(12): e3-4.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation (IMCHF), Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry is the chief scientific officer and head of the department of mental health and clini-cal research at the IMCHF and is Mr. Choudhry’s father. He has no disclosures.
Recently, the Yakima Health District (YHD), in collaboration with the Washington State Department of Health, issued dramatic revisions to its educational curriculum, opting for exclusively remote learning as an important next step in COVID-19 containment measures.
The newly implemented “enhanced” distance-learning paradigm has garnered considerable national attention. Even more noteworthy is how YHD addressed those with language barriers and learning differences such as ADHD as a “priority group”; these individuals are exempt from the newly implemented measures, and small instructional groups of no more than five “at-risk” students will be directly supervised by specialized educators.1,2 To overcome these new unprecedented challenges from the coronavirus pandemic, especially from the perspective of distance education and mental health for susceptible groups such as those with ADHD, it is of utmost importance to explore various programs of interest, as well as the targeted therapies being considered during this crisis.
From a therapeutic standpoint, individuals with learning differences are more likely to play catch-up with their age-matched peers. This puts them at significant risk for developmental delays with symptoms manifesting as disruptive behavioral issues. This is why ongoing parental guidance, coupled with a paradoxically stimulating environment, is critical for children and adolescents with ADHD.3 Accumulating evidence, based on a myriad of studies, demonstrates that childhood treatment with ADHD stimulants reduces the incidence of future substance use, as well as that of other negative outcomes.4,5
Therapeutic strategies that work
“The new normal” has forced unique challenges on clinicians for mitigating distress by novel means of health care delivery. Given the paucity of research exploring the interactions of individuals with ADHD within the context of COVID-19, Take for example, the suggested guidelines from the European ADHD Guidelines Group (EAGG) – such as the following:
- Telecommunications in general, and telepsychiatry in particular, should function as the primary mode of health care delivery to fulfill societal standards of physical distancing.
- Children and adolescents with ADHD should be designated as a “priority group” with respect to monitoring initiatives by educators in a school setting, be it virtual or otherwise.
- Implementation of behavioral strategies by parent or guardian to address psychological well-being and reduce the presence of comorbid behavioral conditions (such as oppositional defiant disorder).
In addition to the aforementioned guidance, EAGG maintains that individuals with ADHD may be initiated on medications after the completion of a baseline examination; if the patients in question are already on a treatment regimen, they should proceed with it as indicated. Interruptions to therapy are not ideal because patients are then subjected to health-related stressors of COVID-19. Reasonable regulations concerning access to medications, without unnecessary delays, undoubtedly will facilitate patient needs, allowing for a smooth transition in day-to-day activities. The family, as a cohesive unit, may benefit from reeducation because it contributes toward the therapeutic process. Neurofeedback, coping skills, and cognitive restructuring training are potential modalities that can augment medications.
Although it may seem counterintuitive, parents or caregivers should resist the urge to increase the medication dose during an outbreak with the intended goal of diminishing the psychosocial burden of ADHD symptomatology. Likewise, unless indicated by a specialist, antipsychotics and/or hypnotics should not be introduced for addressing behavioral dysregulation (such as agitation) during the confinement period.
Historically, numerous clinicians have suggested that patients undergo a routine cardiovascular examination and EKG before being prescribed psychostimulants (the rationale for this recommendation is that sympathomimetics unduly affect blood pressure and heart rate).6,7 However, the American Academy of Pediatrics (AAP) and the American Heart Association (AHA) eventually amended their previous stance by releasing a joint statement in which they deemed a baseline EKG necessary only in ADHD patients with preexisting cardiac risk. For all other patients, the use of EKGs was entirely contingent on physician discretion. However, given the nature of safety precautions for COVID-19, it is prudent to discourage or delay in-person cardiovascular examination/monitoring protocols altogether, especially in those patients without known heart conditions.
Another area of concern is sleep dysfunction, which might exist as an untoward effect of ADHD medication intake or because of the presence of COVID-19 psychosocial stressors. However, clinicians advise that unnecessary psychopharmacology (such as hypnotics or melatonin) be avoided. Instead, conservative lifestyle measures should be enacted, emphasizing the role of proper sleep hygiene in maintaining optimal behavioral health. Despite setbacks to in-person appointments, patients are expected to continue their pharmacotherapy with “parent-focused” ADHD interventions taking a primary role in facilitating compliance through remote monitoring.
ADMiRE, a tertiary-level, dedicated ADHD intervention program from South Dublin, Ireland, has identified several roadblocks with respect to streamlining health care for individuals with ADHD during the confinement period. The proposed resolution to these issues, some of which are derived from EAGG guidelines, might have universal applications elsewhere, thereby facilitating the development of therapeutic services of interest. ADMiRE has noted a correspondence between the guidelines established by EAGG and that of the Canadian ADHD Resource Alliance (CADDRA), including minimal in-person interactions (in favor of virtual teleconferencing) and a cardiovascular screen can be performed in lieu of baseline cardiac auscultation. Moreover, in the event that the patient is a low cardiac risk candidate for ADHD treatment, monitoring protocols may be continued from a home setting. However, if a physical examination is indicated, CADDRA recommends the use of precautionary PPE before commencing ADHD pharmacotherapy.
One of the most significant hurdles is that of school closures because teacher feedback for baseline behavior was traditionally instrumental in dictating patient medical management (for example, for titration schedule). It is expected that, for the time being, this role will be supplanted by parental reports. As well as disclosing information on behavioral dysregulation, family members should be trained to relay critical information about the development of stimulant-induced cardiovascular symptoms – namely, dyspnea, chest pain, and/or palpitations. Furthermore, as primary caregivers, parents should harbor a certain degree of emotional sensitivity because their mood state may influence the child’s overall behavioral course in terms of symptom exacerbation.8
Toward adopting an integrated model for care
Developing an effective assessment plan for patients with ADHD often proves to be a challenging task for clinicians, perhaps even more so in environments that enforce social distancing and limited physical contact by default. As a neurodevelopmental disorder from childhood, the symptoms (including inattention, hyperactivity, and/or impulsivity) of ADHD do not arise in a vacuum – comorbid conditions include mood and anxiety disorders, which are complicated further by a background risk for substance use and self-medicating tendencies.9 Unfortunately, the pandemic has limited the breadth of non-COVID doctors visits, which hinders the overall diagnostic and monitoring process for identifiable comorbid conditions, such as autism spectrum disorder, intellectual disability, oppositional defiant and conduct disorders, and so on.10 Since ADHD symptoms cannot be treated by pharmacotherapy or behavioral interventions alone, our team advocates that families provide additional emotional support and continuous encouragement during these uncertain times.
ADHD and the self-medication hypothesis
The Khantzian self-medication hypothesis posits that a drug seeker may subconsciously gravitate toward a particular agent only to discover a sense of relief concerning inner turmoil or restlessness after use. Observations support the notion that individuals with undiagnosed ADHD have sought cocaine or even recreational designer drugs (such as methylenedioxypyrovalerone, or “bath salts”).11 Given the similar mechanism of action between cocaine, methylenedioxypyrovalerone, and prescribed psychostimulants such as methylphenidate, the results are hardly surprising because these agents all work on the brain’s “reward center” (for example, the nucleus accumbens) by invoking dopamine release. Aside from the aforementioned self-medication hypothesis, “downers” such as Xanax recently have experienced a prescription spike during the outbreak. While there isn’t an immediate cause for concern of Xanax abuse in ADHD individuals, the potential for addiction is certainly real, especially when taking into account comorbid anxiety disorder or sleep dysfunction.
Because of limited resources and precautionary guidelines, clinicians are at a considerable disadvantage in terms of formulating a comprehensive diagnostic and treatment plan for children and adolescents with ADHD. This situation is further compounded by the recent closure of schools and the lack of feedback with respect to baseline behavior from teachers and specialized educators. This is why it is imperative for primary caregivers to closely monitor children with ADHD for developing changes in behavioral patterns (for example, mood or anxiety issues and drug-seeking or disruptive behavior) and work with health care professionals.
References
1. “Distance learning strongly recommended for all Yakima county schools.” NBC Right Now. 2020 Aug 5.
2. Retka J. “Enhanced” remote learning in Yakima county schools? What that means for students this fall. Yakima Herald-Republic. 2020 Aug 8.
3. Armstrong T. “To empower! Not Control! A holistic approach to ADHD.” American Institute for Learning and Development. 1998.
4. J Child Psychol Psychiatry. 2014 Aug;55(8):878-85.
5. Ir J Psychol Med. 2020 May 21:1-22.
6. Lancet Child Adolesc Health. 2020 Jun;4(6):412-4.
7. O’Keefe L. AAP News. 2008 Jun;29(6):1.
8. Asian J Psychiatr. 2020 Jun;51:102077.
9. Current Psychiatry. 2015 Dec;14(12):e3-4.
10. Encephale. 2020 Jun 7;46(3S):S85-92.
11. Current Psychiatry. 2014 Dec; 3(12): e3-4.
Dr. Islam is a medical adviser for the International Maternal and Child Health Foundation (IMCHF), Montreal, and is based in New York. He also is a postdoctoral fellow, psychopharmacologist, and a board-certified medical affairs specialist. Dr. Islam disclosed no relevant financial relationships. Zaid Ulhaq Choudhry is a research assistant at the IMCHF. He has no disclosures. Dr. Zia Choudhry is the chief scientific officer and head of the department of mental health and clini-cal research at the IMCHF and is Mr. Choudhry’s father. He has no disclosures.
Hospitalists confront administrative, financial challenges of COVID-19 crisis
Hospitalists nationwide have put in longer hours, played new clinical roles, and stretched beyond their medical specialty and comfort level to meet their hospital’s COVID-19 care demands. Can they expect some kind of financial recognition – perhaps in the form of “hazard pay” for going above and beyond – even though their institutions are experiencing negative financial fallout from the crisis?
Hospitals in regions experiencing a COVID-19 surge have limited elective procedures, discouraged non–COVID-19 admissions, and essentially entered crisis management mode. Other facilities in less hard-hit communities are also standing by, with reduced hospital census, smaller caseloads and less work to do, while trying to prepare their bottom lines for lower demand.
“This crisis has put most hospitals in financial jeopardy and that is likely to trickle down to all employees – including hospitalists,” said Ron Greeno, MD, FCCP, MHM, a past president of SHM and the society’s current senior advisor for government affairs. “But it’s not like hospitals could or would forgo an effective hospitalist program today. Hospitalists will be important players in defining the hospital’s future direction post crisis.”
That doesn’t mean tighter financials, caps on annual salary increases, or higher productivity expectations won’t be part of future conversations between hospital administrators and their hospitalists, Dr. Greeno said. Administrators are starting to look ahead to the post–COVID-19 era even as numbers of cases and rates of growth continue to rise in various regions, and Dr. Greeno sees a lot of uncertainty ahead.
Even prior to the crisis, he noted, hospital margins had been falling, while the cost of labor, including hospitalist labor, was going up. That was pointing toward an inevitable collision, which has only intensified with the new financial crisis facing hospitals – created by SARS-CoV-2 and by policies such as shutting down elective surgeries in anticipation of a COVID-19 patient surge that, for some institutions, may never come.
Brian Harte, MD, MHM, president of Cleveland Clinic Akron General and a past president of SHM, said that the Cleveland Clinic system has been planning since January its response to the coming crisis. “Governor Mike DeWine and the state Department of Health led the way in flattening the curve in Ohio. We engaged our hospitalists in brainstorming solutions. They have been excellent partners,” he said.
Approaching the crisis with a sense of urgency from the outset, the Cleveland Clinic built a COVID-19 surge team and incident command structure, with nursing, infectious diseases, critical care and hospital medicine represented. “We used that time to get ready for what was coming. We worked on streamlining consultant work flows.”
But utilization numbers are off in almost every service line, Dr. Harte said. “It has forced us to look at things we’ve always talked about, including greater use of telemedicine and exploring other ways of caring for patients, such as increased use of evening hours.”
Cleveland Clinic contracts with Sound Physicians of Tacoma, Wash., for its hospitalist coverage. “We have an excellent working relationship with Sound at the local, regional, and national levels, with common goals for quality and utilization. We tried to involve our hospitalists as early as possible in planning. We needed them to step in and role model and lead the way,” Dr. Harte said, for everybody’s anxiety levels.
“We’re still in the process of understanding the long-term financial impact of the epidemic,” Dr. Harte added. “But at this point I see no reason to think our relationship with our hospitalists needs to change. We’re the stewards of long-term finances. We’ll need to keep a close eye on this. But we’re committed to working through this together.”
Hazard pay for frontline health care workers was included in the COVID-19 relief package assembled in mid-May by Democrats in the House of Representatives. The $3 trillion HEROES Act includes $200 billion to award hazard pay to essential workers, including those in the health field, but Senate Majority Leader Mitch McConnell (R-Ky.) declared the legislation “dead on arrival” in the Senate.
Supplementary hazard payments made by hospitals to their hospitalists as a reward for sacrifices they made in the crisis is an interesting question, Dr. Greeno noted, and it’s definitely on the table at some hospitals. “But I think it is going to be a tough ask in these times.”
Dr. Harte said he has not offered nor been asked about hazard pay for hospitalists. Cleveland Clinic Akron General made a strategic decision that hazard pay was not going to be part of its response to the pandemic. Other hospital administrators interviewed for this article concur.
Hospitals respond to the fiscal crisis
Hospitals in other parts of the country also report significant fiscal fallout from the COVID-19 crisis, with predictions that 100 or more hospitals may be forced to close. Jeff Dye, president of the New Mexico Hospital Association, told the Albuquerque Journal on May 1 that hospitals in his state have been squeezed on all sides by increased costs, patients delaying routine care, and public health orders restricting elective surgeries. New Mexico hospitals, especially in rural areas, face incredible financial strain.
The University of Virginia Medical Center, Charlottesville, recently announced 20% reductions in total compensation for its providers through July 31, along with suspension of retirement contributions. Those changes won’t affect team members caring for COVID-19 patients. And the Spectrum Health Medical Group of 15 hospitals in western Michigan, according to Michigan Public Radio, told its doctors they either needed to sign “contract addendums” giving the system more control over their hours – or face a 25% pay cut, or worse.
Cheyenne (Wyo.) Regional Medical Center issued a statement April 24 that it expected losses of $10 million for the month of April. “CRMC, like every other hospital in Wyoming, is certainly feeling the financial impact that COVID-19 is having,” CEO Tim Thornell told the Cowboy State Daily on April 24. That includes a 30% reduction in inpatient care and 50% reduction in outpatient care, while the hospital has only had a handful of COVID-19 patients at any time. Capital projects are now on hold, overtime is limited, and a hiring freeze is in effect.
“We’re certainly prepared for a larger surge, which hasn’t come yet,” Mr. Thornell said in an interview. CRMC’s ICU was split to create a nine-bed dedicated COVID-19 unit. Intensivists see most of the critical care patients, while the hospital’s 15 directly-employed hospitalists are treating all of the non-ICU COVID-19 patients. “Among themselves, the hospitalists volunteered who would work on the unit. We’ve been fortunate enough to have enough volunteers and enough PPE [personal protective equipment],” he said.
Preparing for the COVID-19 pandemic has strengthened the medical center’s relationship with its hospitalists, Mr. Thornell explained. “Hospitalists are key to our operations, involved in so much that happens here. We’re trying to staff to volume with decreased utilization. We’ve scaled back, which only makes fiscal sense. Now, how do we reinfuse patients back into the mix? Our hospitalists are paid by the number of shifts, and as you distribute shift reductions over 15 providers, it shouldn’t be an intolerable burden.” But two open hospitalist positions have not been filled, he noted.
CRMC is trying to approach these changes with a Lean perspective, Mr. Thornell said. “We had already adopted a Lean program, but this has been a chance to go through a life-altering circumstance using the tools of Lean planning and applying them instantaneously.”
Providers step up
At Emory Healthcare in Atlanta, a major center for COVID-19 cases, communication has been essential in the crisis, said Bryce Gartland, MD, SFHM, Emory’s hospital group president and cochief of clinical operations. “Our group was prepared for a significant influx of patients. Like every other institution, we made the decision to postpone elective care, with a resulting plummet in volume,” he said.
As COVID-19 patients entered the Emory system, frontline hospitalists stepped up to care for those patients. “We’ve had ample providers in terms of clinical care. We guaranteed our physicians’ base compensation. They have flexed teams up and down as needed.” Advanced practice professionals also stepped up to bridge gaps.
With regard to the return of volumes of non–COVID-19 patients, the jury’s still out, Dr. Gartland said. “None of us has a crystal ball, and there are tremendous variables and decision points that will have significant impact. We have started to see numbers of time-sensitive and essential cases increase as of the first week of May.”
What lies ahead will likely include some rightsizing to future volumes. On top of that, the broader economic pressures on hospitals from high rates of unemployment, uninsured patients, bad debt, and charity care will push health care systems to significantly address costs and infrastructure, he said. “We’re still early in planning, and striving to maintain flexibility and nimbleness, given the uncertainties to this early understanding of our new normal. No hospital is immune from the financial impact. We’ll see and hear about more of these conversations in the months ahead.”
But the experience has also generated some positives, Dr. Gartland noted. “Things like telehealth, which we’ve been talking about for years but previously faced barriers to widespread adoption.” Now with COVID-19, the federal government issued waivers, and barriers – both internal and external – came down. “With telehealth, what will the role and deployment of hospitalists look like in this new model? How will traditional productivity expectations change, or the numbers and types of providers? This will make the relationship and partnership between hospitalist groups and hospital administrators ever more important as we consider the evolution toward new care models.”
Dr. Gartland said that “one of the great things about hospital medicine as a field is its flexibility and adaptability. Where there have been gaps, hospitalists were quick to step in. As long as hospital medicine continues to embrace those kinds of behaviors, it will be successful.” But if the conversation with hospitals is just about money, it will be harder, he acknowledged. “Where there is this kind of disruption in our usual way of doing things, there are also tremendous opportunities for care model innovation. I would encourage hospitalist groups to try to be true value partners.”
Command center mode
Like other physicians in hospital C-suites, Chad Whelan MD, FACP, SFHM, chief executive officer of Banner–University Medicine in Tucson, Ariz., led his two hospitals into command center mode when the crisis hit, planning for a surge of COVID-19 cases that could overwhelm hospital capacity.
“In terms of our hospitalists, we leaned in to them hard in the beginning, preparing them to supervise other physicians who came in to help if needed,” he said. “Our [non–COVID-19] census is down, revenues are down, and the implications are enormous – like nothing we’ve ever seen before.”
“We’re fortunate that we’re part of the Banner health system. We made a decision that we would essentially keep our physicians financially protected through this crisis,” Dr. Whelan said. “In return, we called on them to step up and be on the front lines and to put in enormous hours for planning. We asked them to consider: How could you contribute if the surge comes?”
He affirmed that hospital medicine has been a major part of his medical center’s planning and implementation. “I’ve been overwhelmed by the degree to which the entire delivery team has rallied around the pandemic, with everybody saying they want to keep people safe and be part of the solution. We have always had hospitalist leaders at the table as we’ve planned our response and as decisions were made,” said Dr. Whelan, a practicing hospitalist and teaching service attending since 2000 until he assumed his current executive position in Arizona 18 months ago.
“While we have kept people whole during the immediate crisis, we have acknowledged that we don’t know what our recovery will look like. What if [non–COVID-19] volume doesn’t return? That keeps me awake at night,” he said. “I have talked to our physician leadership in hospital medicine and more broadly. We need to ask ourselves many questions, including: do we have the right levels of staffing? Is this the time to consider alternate models of staffing, for example, advanced practice providers? And does the compensation plan need adjustments?”
Dr. Whelan thinks that the COVID-19 crisis is an opportunity for hospital medicine to more rapidly explore different models and to ask what additional value hospitalists can bring to the care model. “For example, what would it mean to redefine the hospitalist’s scope of practice as an acute medicine specialist, not defined by the hospital’s four walls?” he noted.
“One of the reasons our smaller hospital reached capacity with COVID-19 patients was the skilled nursing facility located a few hundred feet away that turned into a hot spot. If we had imported the hospital medicine model virtually into that SNF early on, could there have been a different scenario? Have we thought through what that would have even looked like?” Dr. Whelan asked.
He challenges the hospital medicine field, once it gets to the other side of this crisis, to not fall back on old way of doing things. “Instead, let’s use this time to create a better model today,” he said. “That’s what we’re trying to do at a system level at Banner, with our hospital medicine groups partnering with the hospital. I want to see our hospitalists create and thrive in that new model.”
Hospitalists nationwide have put in longer hours, played new clinical roles, and stretched beyond their medical specialty and comfort level to meet their hospital’s COVID-19 care demands. Can they expect some kind of financial recognition – perhaps in the form of “hazard pay” for going above and beyond – even though their institutions are experiencing negative financial fallout from the crisis?
Hospitals in regions experiencing a COVID-19 surge have limited elective procedures, discouraged non–COVID-19 admissions, and essentially entered crisis management mode. Other facilities in less hard-hit communities are also standing by, with reduced hospital census, smaller caseloads and less work to do, while trying to prepare their bottom lines for lower demand.
“This crisis has put most hospitals in financial jeopardy and that is likely to trickle down to all employees – including hospitalists,” said Ron Greeno, MD, FCCP, MHM, a past president of SHM and the society’s current senior advisor for government affairs. “But it’s not like hospitals could or would forgo an effective hospitalist program today. Hospitalists will be important players in defining the hospital’s future direction post crisis.”
That doesn’t mean tighter financials, caps on annual salary increases, or higher productivity expectations won’t be part of future conversations between hospital administrators and their hospitalists, Dr. Greeno said. Administrators are starting to look ahead to the post–COVID-19 era even as numbers of cases and rates of growth continue to rise in various regions, and Dr. Greeno sees a lot of uncertainty ahead.
Even prior to the crisis, he noted, hospital margins had been falling, while the cost of labor, including hospitalist labor, was going up. That was pointing toward an inevitable collision, which has only intensified with the new financial crisis facing hospitals – created by SARS-CoV-2 and by policies such as shutting down elective surgeries in anticipation of a COVID-19 patient surge that, for some institutions, may never come.
Brian Harte, MD, MHM, president of Cleveland Clinic Akron General and a past president of SHM, said that the Cleveland Clinic system has been planning since January its response to the coming crisis. “Governor Mike DeWine and the state Department of Health led the way in flattening the curve in Ohio. We engaged our hospitalists in brainstorming solutions. They have been excellent partners,” he said.
Approaching the crisis with a sense of urgency from the outset, the Cleveland Clinic built a COVID-19 surge team and incident command structure, with nursing, infectious diseases, critical care and hospital medicine represented. “We used that time to get ready for what was coming. We worked on streamlining consultant work flows.”
But utilization numbers are off in almost every service line, Dr. Harte said. “It has forced us to look at things we’ve always talked about, including greater use of telemedicine and exploring other ways of caring for patients, such as increased use of evening hours.”
Cleveland Clinic contracts with Sound Physicians of Tacoma, Wash., for its hospitalist coverage. “We have an excellent working relationship with Sound at the local, regional, and national levels, with common goals for quality and utilization. We tried to involve our hospitalists as early as possible in planning. We needed them to step in and role model and lead the way,” Dr. Harte said, for everybody’s anxiety levels.
“We’re still in the process of understanding the long-term financial impact of the epidemic,” Dr. Harte added. “But at this point I see no reason to think our relationship with our hospitalists needs to change. We’re the stewards of long-term finances. We’ll need to keep a close eye on this. But we’re committed to working through this together.”
Hazard pay for frontline health care workers was included in the COVID-19 relief package assembled in mid-May by Democrats in the House of Representatives. The $3 trillion HEROES Act includes $200 billion to award hazard pay to essential workers, including those in the health field, but Senate Majority Leader Mitch McConnell (R-Ky.) declared the legislation “dead on arrival” in the Senate.
Supplementary hazard payments made by hospitals to their hospitalists as a reward for sacrifices they made in the crisis is an interesting question, Dr. Greeno noted, and it’s definitely on the table at some hospitals. “But I think it is going to be a tough ask in these times.”
Dr. Harte said he has not offered nor been asked about hazard pay for hospitalists. Cleveland Clinic Akron General made a strategic decision that hazard pay was not going to be part of its response to the pandemic. Other hospital administrators interviewed for this article concur.
Hospitals respond to the fiscal crisis
Hospitals in other parts of the country also report significant fiscal fallout from the COVID-19 crisis, with predictions that 100 or more hospitals may be forced to close. Jeff Dye, president of the New Mexico Hospital Association, told the Albuquerque Journal on May 1 that hospitals in his state have been squeezed on all sides by increased costs, patients delaying routine care, and public health orders restricting elective surgeries. New Mexico hospitals, especially in rural areas, face incredible financial strain.
The University of Virginia Medical Center, Charlottesville, recently announced 20% reductions in total compensation for its providers through July 31, along with suspension of retirement contributions. Those changes won’t affect team members caring for COVID-19 patients. And the Spectrum Health Medical Group of 15 hospitals in western Michigan, according to Michigan Public Radio, told its doctors they either needed to sign “contract addendums” giving the system more control over their hours – or face a 25% pay cut, or worse.
Cheyenne (Wyo.) Regional Medical Center issued a statement April 24 that it expected losses of $10 million for the month of April. “CRMC, like every other hospital in Wyoming, is certainly feeling the financial impact that COVID-19 is having,” CEO Tim Thornell told the Cowboy State Daily on April 24. That includes a 30% reduction in inpatient care and 50% reduction in outpatient care, while the hospital has only had a handful of COVID-19 patients at any time. Capital projects are now on hold, overtime is limited, and a hiring freeze is in effect.
“We’re certainly prepared for a larger surge, which hasn’t come yet,” Mr. Thornell said in an interview. CRMC’s ICU was split to create a nine-bed dedicated COVID-19 unit. Intensivists see most of the critical care patients, while the hospital’s 15 directly-employed hospitalists are treating all of the non-ICU COVID-19 patients. “Among themselves, the hospitalists volunteered who would work on the unit. We’ve been fortunate enough to have enough volunteers and enough PPE [personal protective equipment],” he said.
Preparing for the COVID-19 pandemic has strengthened the medical center’s relationship with its hospitalists, Mr. Thornell explained. “Hospitalists are key to our operations, involved in so much that happens here. We’re trying to staff to volume with decreased utilization. We’ve scaled back, which only makes fiscal sense. Now, how do we reinfuse patients back into the mix? Our hospitalists are paid by the number of shifts, and as you distribute shift reductions over 15 providers, it shouldn’t be an intolerable burden.” But two open hospitalist positions have not been filled, he noted.
CRMC is trying to approach these changes with a Lean perspective, Mr. Thornell said. “We had already adopted a Lean program, but this has been a chance to go through a life-altering circumstance using the tools of Lean planning and applying them instantaneously.”
Providers step up
At Emory Healthcare in Atlanta, a major center for COVID-19 cases, communication has been essential in the crisis, said Bryce Gartland, MD, SFHM, Emory’s hospital group president and cochief of clinical operations. “Our group was prepared for a significant influx of patients. Like every other institution, we made the decision to postpone elective care, with a resulting plummet in volume,” he said.
As COVID-19 patients entered the Emory system, frontline hospitalists stepped up to care for those patients. “We’ve had ample providers in terms of clinical care. We guaranteed our physicians’ base compensation. They have flexed teams up and down as needed.” Advanced practice professionals also stepped up to bridge gaps.
With regard to the return of volumes of non–COVID-19 patients, the jury’s still out, Dr. Gartland said. “None of us has a crystal ball, and there are tremendous variables and decision points that will have significant impact. We have started to see numbers of time-sensitive and essential cases increase as of the first week of May.”
What lies ahead will likely include some rightsizing to future volumes. On top of that, the broader economic pressures on hospitals from high rates of unemployment, uninsured patients, bad debt, and charity care will push health care systems to significantly address costs and infrastructure, he said. “We’re still early in planning, and striving to maintain flexibility and nimbleness, given the uncertainties to this early understanding of our new normal. No hospital is immune from the financial impact. We’ll see and hear about more of these conversations in the months ahead.”
But the experience has also generated some positives, Dr. Gartland noted. “Things like telehealth, which we’ve been talking about for years but previously faced barriers to widespread adoption.” Now with COVID-19, the federal government issued waivers, and barriers – both internal and external – came down. “With telehealth, what will the role and deployment of hospitalists look like in this new model? How will traditional productivity expectations change, or the numbers and types of providers? This will make the relationship and partnership between hospitalist groups and hospital administrators ever more important as we consider the evolution toward new care models.”
Dr. Gartland said that “one of the great things about hospital medicine as a field is its flexibility and adaptability. Where there have been gaps, hospitalists were quick to step in. As long as hospital medicine continues to embrace those kinds of behaviors, it will be successful.” But if the conversation with hospitals is just about money, it will be harder, he acknowledged. “Where there is this kind of disruption in our usual way of doing things, there are also tremendous opportunities for care model innovation. I would encourage hospitalist groups to try to be true value partners.”
Command center mode
Like other physicians in hospital C-suites, Chad Whelan MD, FACP, SFHM, chief executive officer of Banner–University Medicine in Tucson, Ariz., led his two hospitals into command center mode when the crisis hit, planning for a surge of COVID-19 cases that could overwhelm hospital capacity.
“In terms of our hospitalists, we leaned in to them hard in the beginning, preparing them to supervise other physicians who came in to help if needed,” he said. “Our [non–COVID-19] census is down, revenues are down, and the implications are enormous – like nothing we’ve ever seen before.”
“We’re fortunate that we’re part of the Banner health system. We made a decision that we would essentially keep our physicians financially protected through this crisis,” Dr. Whelan said. “In return, we called on them to step up and be on the front lines and to put in enormous hours for planning. We asked them to consider: How could you contribute if the surge comes?”
He affirmed that hospital medicine has been a major part of his medical center’s planning and implementation. “I’ve been overwhelmed by the degree to which the entire delivery team has rallied around the pandemic, with everybody saying they want to keep people safe and be part of the solution. We have always had hospitalist leaders at the table as we’ve planned our response and as decisions were made,” said Dr. Whelan, a practicing hospitalist and teaching service attending since 2000 until he assumed his current executive position in Arizona 18 months ago.
“While we have kept people whole during the immediate crisis, we have acknowledged that we don’t know what our recovery will look like. What if [non–COVID-19] volume doesn’t return? That keeps me awake at night,” he said. “I have talked to our physician leadership in hospital medicine and more broadly. We need to ask ourselves many questions, including: do we have the right levels of staffing? Is this the time to consider alternate models of staffing, for example, advanced practice providers? And does the compensation plan need adjustments?”
Dr. Whelan thinks that the COVID-19 crisis is an opportunity for hospital medicine to more rapidly explore different models and to ask what additional value hospitalists can bring to the care model. “For example, what would it mean to redefine the hospitalist’s scope of practice as an acute medicine specialist, not defined by the hospital’s four walls?” he noted.
“One of the reasons our smaller hospital reached capacity with COVID-19 patients was the skilled nursing facility located a few hundred feet away that turned into a hot spot. If we had imported the hospital medicine model virtually into that SNF early on, could there have been a different scenario? Have we thought through what that would have even looked like?” Dr. Whelan asked.
He challenges the hospital medicine field, once it gets to the other side of this crisis, to not fall back on old way of doing things. “Instead, let’s use this time to create a better model today,” he said. “That’s what we’re trying to do at a system level at Banner, with our hospital medicine groups partnering with the hospital. I want to see our hospitalists create and thrive in that new model.”
Hospitalists nationwide have put in longer hours, played new clinical roles, and stretched beyond their medical specialty and comfort level to meet their hospital’s COVID-19 care demands. Can they expect some kind of financial recognition – perhaps in the form of “hazard pay” for going above and beyond – even though their institutions are experiencing negative financial fallout from the crisis?
Hospitals in regions experiencing a COVID-19 surge have limited elective procedures, discouraged non–COVID-19 admissions, and essentially entered crisis management mode. Other facilities in less hard-hit communities are also standing by, with reduced hospital census, smaller caseloads and less work to do, while trying to prepare their bottom lines for lower demand.
“This crisis has put most hospitals in financial jeopardy and that is likely to trickle down to all employees – including hospitalists,” said Ron Greeno, MD, FCCP, MHM, a past president of SHM and the society’s current senior advisor for government affairs. “But it’s not like hospitals could or would forgo an effective hospitalist program today. Hospitalists will be important players in defining the hospital’s future direction post crisis.”
That doesn’t mean tighter financials, caps on annual salary increases, or higher productivity expectations won’t be part of future conversations between hospital administrators and their hospitalists, Dr. Greeno said. Administrators are starting to look ahead to the post–COVID-19 era even as numbers of cases and rates of growth continue to rise in various regions, and Dr. Greeno sees a lot of uncertainty ahead.
Even prior to the crisis, he noted, hospital margins had been falling, while the cost of labor, including hospitalist labor, was going up. That was pointing toward an inevitable collision, which has only intensified with the new financial crisis facing hospitals – created by SARS-CoV-2 and by policies such as shutting down elective surgeries in anticipation of a COVID-19 patient surge that, for some institutions, may never come.
Brian Harte, MD, MHM, president of Cleveland Clinic Akron General and a past president of SHM, said that the Cleveland Clinic system has been planning since January its response to the coming crisis. “Governor Mike DeWine and the state Department of Health led the way in flattening the curve in Ohio. We engaged our hospitalists in brainstorming solutions. They have been excellent partners,” he said.
Approaching the crisis with a sense of urgency from the outset, the Cleveland Clinic built a COVID-19 surge team and incident command structure, with nursing, infectious diseases, critical care and hospital medicine represented. “We used that time to get ready for what was coming. We worked on streamlining consultant work flows.”
But utilization numbers are off in almost every service line, Dr. Harte said. “It has forced us to look at things we’ve always talked about, including greater use of telemedicine and exploring other ways of caring for patients, such as increased use of evening hours.”
Cleveland Clinic contracts with Sound Physicians of Tacoma, Wash., for its hospitalist coverage. “We have an excellent working relationship with Sound at the local, regional, and national levels, with common goals for quality and utilization. We tried to involve our hospitalists as early as possible in planning. We needed them to step in and role model and lead the way,” Dr. Harte said, for everybody’s anxiety levels.
“We’re still in the process of understanding the long-term financial impact of the epidemic,” Dr. Harte added. “But at this point I see no reason to think our relationship with our hospitalists needs to change. We’re the stewards of long-term finances. We’ll need to keep a close eye on this. But we’re committed to working through this together.”
Hazard pay for frontline health care workers was included in the COVID-19 relief package assembled in mid-May by Democrats in the House of Representatives. The $3 trillion HEROES Act includes $200 billion to award hazard pay to essential workers, including those in the health field, but Senate Majority Leader Mitch McConnell (R-Ky.) declared the legislation “dead on arrival” in the Senate.
Supplementary hazard payments made by hospitals to their hospitalists as a reward for sacrifices they made in the crisis is an interesting question, Dr. Greeno noted, and it’s definitely on the table at some hospitals. “But I think it is going to be a tough ask in these times.”
Dr. Harte said he has not offered nor been asked about hazard pay for hospitalists. Cleveland Clinic Akron General made a strategic decision that hazard pay was not going to be part of its response to the pandemic. Other hospital administrators interviewed for this article concur.
Hospitals respond to the fiscal crisis
Hospitals in other parts of the country also report significant fiscal fallout from the COVID-19 crisis, with predictions that 100 or more hospitals may be forced to close. Jeff Dye, president of the New Mexico Hospital Association, told the Albuquerque Journal on May 1 that hospitals in his state have been squeezed on all sides by increased costs, patients delaying routine care, and public health orders restricting elective surgeries. New Mexico hospitals, especially in rural areas, face incredible financial strain.
The University of Virginia Medical Center, Charlottesville, recently announced 20% reductions in total compensation for its providers through July 31, along with suspension of retirement contributions. Those changes won’t affect team members caring for COVID-19 patients. And the Spectrum Health Medical Group of 15 hospitals in western Michigan, according to Michigan Public Radio, told its doctors they either needed to sign “contract addendums” giving the system more control over their hours – or face a 25% pay cut, or worse.
Cheyenne (Wyo.) Regional Medical Center issued a statement April 24 that it expected losses of $10 million for the month of April. “CRMC, like every other hospital in Wyoming, is certainly feeling the financial impact that COVID-19 is having,” CEO Tim Thornell told the Cowboy State Daily on April 24. That includes a 30% reduction in inpatient care and 50% reduction in outpatient care, while the hospital has only had a handful of COVID-19 patients at any time. Capital projects are now on hold, overtime is limited, and a hiring freeze is in effect.
“We’re certainly prepared for a larger surge, which hasn’t come yet,” Mr. Thornell said in an interview. CRMC’s ICU was split to create a nine-bed dedicated COVID-19 unit. Intensivists see most of the critical care patients, while the hospital’s 15 directly-employed hospitalists are treating all of the non-ICU COVID-19 patients. “Among themselves, the hospitalists volunteered who would work on the unit. We’ve been fortunate enough to have enough volunteers and enough PPE [personal protective equipment],” he said.
Preparing for the COVID-19 pandemic has strengthened the medical center’s relationship with its hospitalists, Mr. Thornell explained. “Hospitalists are key to our operations, involved in so much that happens here. We’re trying to staff to volume with decreased utilization. We’ve scaled back, which only makes fiscal sense. Now, how do we reinfuse patients back into the mix? Our hospitalists are paid by the number of shifts, and as you distribute shift reductions over 15 providers, it shouldn’t be an intolerable burden.” But two open hospitalist positions have not been filled, he noted.
CRMC is trying to approach these changes with a Lean perspective, Mr. Thornell said. “We had already adopted a Lean program, but this has been a chance to go through a life-altering circumstance using the tools of Lean planning and applying them instantaneously.”
Providers step up
At Emory Healthcare in Atlanta, a major center for COVID-19 cases, communication has been essential in the crisis, said Bryce Gartland, MD, SFHM, Emory’s hospital group president and cochief of clinical operations. “Our group was prepared for a significant influx of patients. Like every other institution, we made the decision to postpone elective care, with a resulting plummet in volume,” he said.
As COVID-19 patients entered the Emory system, frontline hospitalists stepped up to care for those patients. “We’ve had ample providers in terms of clinical care. We guaranteed our physicians’ base compensation. They have flexed teams up and down as needed.” Advanced practice professionals also stepped up to bridge gaps.
With regard to the return of volumes of non–COVID-19 patients, the jury’s still out, Dr. Gartland said. “None of us has a crystal ball, and there are tremendous variables and decision points that will have significant impact. We have started to see numbers of time-sensitive and essential cases increase as of the first week of May.”
What lies ahead will likely include some rightsizing to future volumes. On top of that, the broader economic pressures on hospitals from high rates of unemployment, uninsured patients, bad debt, and charity care will push health care systems to significantly address costs and infrastructure, he said. “We’re still early in planning, and striving to maintain flexibility and nimbleness, given the uncertainties to this early understanding of our new normal. No hospital is immune from the financial impact. We’ll see and hear about more of these conversations in the months ahead.”
But the experience has also generated some positives, Dr. Gartland noted. “Things like telehealth, which we’ve been talking about for years but previously faced barriers to widespread adoption.” Now with COVID-19, the federal government issued waivers, and barriers – both internal and external – came down. “With telehealth, what will the role and deployment of hospitalists look like in this new model? How will traditional productivity expectations change, or the numbers and types of providers? This will make the relationship and partnership between hospitalist groups and hospital administrators ever more important as we consider the evolution toward new care models.”
Dr. Gartland said that “one of the great things about hospital medicine as a field is its flexibility and adaptability. Where there have been gaps, hospitalists were quick to step in. As long as hospital medicine continues to embrace those kinds of behaviors, it will be successful.” But if the conversation with hospitals is just about money, it will be harder, he acknowledged. “Where there is this kind of disruption in our usual way of doing things, there are also tremendous opportunities for care model innovation. I would encourage hospitalist groups to try to be true value partners.”
Command center mode
Like other physicians in hospital C-suites, Chad Whelan MD, FACP, SFHM, chief executive officer of Banner–University Medicine in Tucson, Ariz., led his two hospitals into command center mode when the crisis hit, planning for a surge of COVID-19 cases that could overwhelm hospital capacity.
“In terms of our hospitalists, we leaned in to them hard in the beginning, preparing them to supervise other physicians who came in to help if needed,” he said. “Our [non–COVID-19] census is down, revenues are down, and the implications are enormous – like nothing we’ve ever seen before.”
“We’re fortunate that we’re part of the Banner health system. We made a decision that we would essentially keep our physicians financially protected through this crisis,” Dr. Whelan said. “In return, we called on them to step up and be on the front lines and to put in enormous hours for planning. We asked them to consider: How could you contribute if the surge comes?”
He affirmed that hospital medicine has been a major part of his medical center’s planning and implementation. “I’ve been overwhelmed by the degree to which the entire delivery team has rallied around the pandemic, with everybody saying they want to keep people safe and be part of the solution. We have always had hospitalist leaders at the table as we’ve planned our response and as decisions were made,” said Dr. Whelan, a practicing hospitalist and teaching service attending since 2000 until he assumed his current executive position in Arizona 18 months ago.
“While we have kept people whole during the immediate crisis, we have acknowledged that we don’t know what our recovery will look like. What if [non–COVID-19] volume doesn’t return? That keeps me awake at night,” he said. “I have talked to our physician leadership in hospital medicine and more broadly. We need to ask ourselves many questions, including: do we have the right levels of staffing? Is this the time to consider alternate models of staffing, for example, advanced practice providers? And does the compensation plan need adjustments?”
Dr. Whelan thinks that the COVID-19 crisis is an opportunity for hospital medicine to more rapidly explore different models and to ask what additional value hospitalists can bring to the care model. “For example, what would it mean to redefine the hospitalist’s scope of practice as an acute medicine specialist, not defined by the hospital’s four walls?” he noted.
“One of the reasons our smaller hospital reached capacity with COVID-19 patients was the skilled nursing facility located a few hundred feet away that turned into a hot spot. If we had imported the hospital medicine model virtually into that SNF early on, could there have been a different scenario? Have we thought through what that would have even looked like?” Dr. Whelan asked.
He challenges the hospital medicine field, once it gets to the other side of this crisis, to not fall back on old way of doing things. “Instead, let’s use this time to create a better model today,” he said. “That’s what we’re trying to do at a system level at Banner, with our hospital medicine groups partnering with the hospital. I want to see our hospitalists create and thrive in that new model.”
Oleander extract for COVID-19? That’s a hard ‘no’ experts say
“Though renowned for its beauty and use in landscaping, this Mediterranean shrub is responsible for cases of accidental poisoning across the globe. All parts of the plant are poisonous,” Cassandra Quave, PhD, ethnobotanist and herbarium curator at Emory University, Atlanta, cautioned in an article in The Conversation, an independent, not-for-profit publication.
Oleandrin has properties similar to digoxin; the onset of toxicity occurs several hours after consumption.
The first symptoms of oleandrin poisoning may be gastrointestinal, such as nausea, vomiting, abdominal pain, diarrhea (which may contain blood), and loss of appetite.
After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Xanthopsia (yellow vision), a burning sensation in the eyes, paralysis of the gastrointestinal tract, and respiratory symptoms may also occur.
Oleandrin poisoning may affect the central nervous system, as evidenced by drowsiness, tremors, seizures, collapse, and coma leading to death. When applied to the skin, oleander sap can cause skin irritations and allergic reactions characterized by dermatitis.
Diagnosis of oleandrin poisoning is mainly made on the basis of a description of the plant, how much of it was ingested, how much time has elapsed since ingestion, and symptoms. Confirmation of oleandrin in blood involves fluorescence polarization immunoassay, digoxin immunoassay, or liquid chromatography-electrospray tandem mass spectrometry.
Neither oleander nor oleandrin is approved by regulatory agencies as a prescription drug or dietary supplement.
In vitro study
Oleandrin for COVID-19 made headlines after President Trump met in the Oval Office with Andrew Whitney, vice chairman and director of Phoenix Biotechnology, along with Housing and Urban Development Secretary Ben Carson, MD, and MyPillow founder/CEO Mike Lindell, a strong supporter of Trump and an investor in the biotech company, to learn about oleandrin, which Whitney called a “cure” for COVID-19, Axios reported.
In an in vitro study, researchers from Phoenix Biotechnology and the University of Texas Medical Branch, Galveston, tested oleandrin against SARS-CoV-2 in cultured Vero cells.
“When administered both before and after virus infection, nanogram doses of oleandrin significantly inhibited replication by 45 to 3000-fold,” the researchers said in an article posted on bioRxiv, a free online archive and distribution service for unpublished preprints in the life sciences. The study has not been peer reviewed.
On the basis of these in vitro findings, the researchers said the plant extract has “potential to prevent disease and virus spread in persons recently exposed to SARS-CoV-2, as well as to prevent severe disease in persons at high risk.”
But it’s a far cry from test tube to human, one expert cautioned.
“This is an understatement: Care must be taken when inferring potential therapeutic benefits from in vitro antiviral effects,” Harlan Krumholz, MD, cardiologist and director, Yale New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, Connecticut, told Medscape Medical News.
“There is a chasm between a single in vitro study and any use in humans outside of a protocol. People should be cautioned about that distance and the need [to] avoid such remedies unless part of a credible research project,” said Krumholz.
Yet Lindell told Axios that, in the Oval Office meeting, Trump expressed enthusiasm for the Food and Drug Administration to allow oleandrin to be marketed as a dietary supplement or approved for COVID-19.
“This is really just nonsense and a distraction,” Jonathan Reiner, MD, of George Washington University Medical Center, Washington, DC, said on CNN.
This article first appeared on Medscape.com.
“Though renowned for its beauty and use in landscaping, this Mediterranean shrub is responsible for cases of accidental poisoning across the globe. All parts of the plant are poisonous,” Cassandra Quave, PhD, ethnobotanist and herbarium curator at Emory University, Atlanta, cautioned in an article in The Conversation, an independent, not-for-profit publication.
Oleandrin has properties similar to digoxin; the onset of toxicity occurs several hours after consumption.
The first symptoms of oleandrin poisoning may be gastrointestinal, such as nausea, vomiting, abdominal pain, diarrhea (which may contain blood), and loss of appetite.
After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Xanthopsia (yellow vision), a burning sensation in the eyes, paralysis of the gastrointestinal tract, and respiratory symptoms may also occur.
Oleandrin poisoning may affect the central nervous system, as evidenced by drowsiness, tremors, seizures, collapse, and coma leading to death. When applied to the skin, oleander sap can cause skin irritations and allergic reactions characterized by dermatitis.
Diagnosis of oleandrin poisoning is mainly made on the basis of a description of the plant, how much of it was ingested, how much time has elapsed since ingestion, and symptoms. Confirmation of oleandrin in blood involves fluorescence polarization immunoassay, digoxin immunoassay, or liquid chromatography-electrospray tandem mass spectrometry.
Neither oleander nor oleandrin is approved by regulatory agencies as a prescription drug or dietary supplement.
In vitro study
Oleandrin for COVID-19 made headlines after President Trump met in the Oval Office with Andrew Whitney, vice chairman and director of Phoenix Biotechnology, along with Housing and Urban Development Secretary Ben Carson, MD, and MyPillow founder/CEO Mike Lindell, a strong supporter of Trump and an investor in the biotech company, to learn about oleandrin, which Whitney called a “cure” for COVID-19, Axios reported.
In an in vitro study, researchers from Phoenix Biotechnology and the University of Texas Medical Branch, Galveston, tested oleandrin against SARS-CoV-2 in cultured Vero cells.
“When administered both before and after virus infection, nanogram doses of oleandrin significantly inhibited replication by 45 to 3000-fold,” the researchers said in an article posted on bioRxiv, a free online archive and distribution service for unpublished preprints in the life sciences. The study has not been peer reviewed.
On the basis of these in vitro findings, the researchers said the plant extract has “potential to prevent disease and virus spread in persons recently exposed to SARS-CoV-2, as well as to prevent severe disease in persons at high risk.”
But it’s a far cry from test tube to human, one expert cautioned.
“This is an understatement: Care must be taken when inferring potential therapeutic benefits from in vitro antiviral effects,” Harlan Krumholz, MD, cardiologist and director, Yale New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, Connecticut, told Medscape Medical News.
“There is a chasm between a single in vitro study and any use in humans outside of a protocol. People should be cautioned about that distance and the need [to] avoid such remedies unless part of a credible research project,” said Krumholz.
Yet Lindell told Axios that, in the Oval Office meeting, Trump expressed enthusiasm for the Food and Drug Administration to allow oleandrin to be marketed as a dietary supplement or approved for COVID-19.
“This is really just nonsense and a distraction,” Jonathan Reiner, MD, of George Washington University Medical Center, Washington, DC, said on CNN.
This article first appeared on Medscape.com.
“Though renowned for its beauty and use in landscaping, this Mediterranean shrub is responsible for cases of accidental poisoning across the globe. All parts of the plant are poisonous,” Cassandra Quave, PhD, ethnobotanist and herbarium curator at Emory University, Atlanta, cautioned in an article in The Conversation, an independent, not-for-profit publication.
Oleandrin has properties similar to digoxin; the onset of toxicity occurs several hours after consumption.
The first symptoms of oleandrin poisoning may be gastrointestinal, such as nausea, vomiting, abdominal pain, diarrhea (which may contain blood), and loss of appetite.
After these first symptoms, the heart may be affected by tachyarrhythmia, bradyarrhythmia, premature ventricular contractions, or atrioventricular blockage. Xanthopsia (yellow vision), a burning sensation in the eyes, paralysis of the gastrointestinal tract, and respiratory symptoms may also occur.
Oleandrin poisoning may affect the central nervous system, as evidenced by drowsiness, tremors, seizures, collapse, and coma leading to death. When applied to the skin, oleander sap can cause skin irritations and allergic reactions characterized by dermatitis.
Diagnosis of oleandrin poisoning is mainly made on the basis of a description of the plant, how much of it was ingested, how much time has elapsed since ingestion, and symptoms. Confirmation of oleandrin in blood involves fluorescence polarization immunoassay, digoxin immunoassay, or liquid chromatography-electrospray tandem mass spectrometry.
Neither oleander nor oleandrin is approved by regulatory agencies as a prescription drug or dietary supplement.
In vitro study
Oleandrin for COVID-19 made headlines after President Trump met in the Oval Office with Andrew Whitney, vice chairman and director of Phoenix Biotechnology, along with Housing and Urban Development Secretary Ben Carson, MD, and MyPillow founder/CEO Mike Lindell, a strong supporter of Trump and an investor in the biotech company, to learn about oleandrin, which Whitney called a “cure” for COVID-19, Axios reported.
In an in vitro study, researchers from Phoenix Biotechnology and the University of Texas Medical Branch, Galveston, tested oleandrin against SARS-CoV-2 in cultured Vero cells.
“When administered both before and after virus infection, nanogram doses of oleandrin significantly inhibited replication by 45 to 3000-fold,” the researchers said in an article posted on bioRxiv, a free online archive and distribution service for unpublished preprints in the life sciences. The study has not been peer reviewed.
On the basis of these in vitro findings, the researchers said the plant extract has “potential to prevent disease and virus spread in persons recently exposed to SARS-CoV-2, as well as to prevent severe disease in persons at high risk.”
But it’s a far cry from test tube to human, one expert cautioned.
“This is an understatement: Care must be taken when inferring potential therapeutic benefits from in vitro antiviral effects,” Harlan Krumholz, MD, cardiologist and director, Yale New Haven Hospital Center for Outcomes Research and Evaluation, New Haven, Connecticut, told Medscape Medical News.
“There is a chasm between a single in vitro study and any use in humans outside of a protocol. People should be cautioned about that distance and the need [to] avoid such remedies unless part of a credible research project,” said Krumholz.
Yet Lindell told Axios that, in the Oval Office meeting, Trump expressed enthusiasm for the Food and Drug Administration to allow oleandrin to be marketed as a dietary supplement or approved for COVID-19.
“This is really just nonsense and a distraction,” Jonathan Reiner, MD, of George Washington University Medical Center, Washington, DC, said on CNN.
This article first appeared on Medscape.com.
Canakinumab (Ilaris) tapering tested in systemic JIA trial
A trial that tested two ways of tapering canakinumab (Ilaris) monotherapy in children with systemic juvenile idiopathic arthritis (sJIA) showed that both approaches might be feasible, but the lack of a control arm means that more data are needed before putting either into routine clinical practice.
Pierre Quartier, MD, and associates reported in Arthritis & Rheumatology. That’s with the proviso that children are taking canakinumab at the recommended dose of 4 mg/kg every 4 weeks and achieve clinical remission before any tapering is started.
The researchers found that 70%-80% of children who were in complete clinical remission (CR) maintained this for 6 months after the first dose-tapering step had been taken. However, at least one-fifth of study participants experienced a disease flare during treatment withdrawal, and only one-third were able to discontinue treatment altogether, which suggests continued treatment is needed.
“The results are a step in the right direction,” commented Athimaleipet Ramanan, MBBS, a consultant pediatric rheumatologist who was not involved in the study. They “offer the first vision of whether we can actually taper a medication” in sJIA because “until now we have not had any evidence for this,” he added.
“What we really need to know, which the study doesn’t tell you, is: Is decreasing the dose better than stopping?” said Dr. Ramanan, of the Bristol (England) Royal Hospital for Children.
Another thing that is important to know is: Does reducing the dose lead to the development of anti-drug antibodies? he said.
“There were some concerns that, when you give less of a monoclonal antibody, you might get more neutralizing anti-drug antibodies or you might make a drug more immunogenic,” he said. These data perhaps suggest that this isn’t the case because only one child on one occasion had detectable non-neutralizing anti-drug antibodies during the entire study, and that was 11 weeks after the last dose of canakinumab had been given.
Study results and design
Canakinumab is a monoclonal antibody that inhibits interleukin-1 (IL-1) that has been approved in the United States and Europe for the treatment of sJIA since 2013. Reducing a child’s exposure to canakinumab once their disease is under control is an attractive proposition given the treat-to-target approach used increasingly throughout modern rheumatologic practice. It could also help reduce the cost of what is an expensive treatment, compared with other available options for sJIA such as the interleukin-6 inhibitor tocilizumab (Actemra) or another IL-1 inhibitor anakinra (Kineret), which is not FDA-approved for use in sJIA and requires weekly injections.
“We don’t want to the disease to reappear, to flare, but we also don’t want to overuse treatments in these patients,” explained Dr. Quartier of Hôpital Necker-Enfants Malades, Assistance Publique-Hôpitaux de Paris in an interview.
The study he coconducted, given the acronym B-SPECIFIC-4 Patients, was an open-label study that consisted of two parts. In part 1, 182 children were given subcutaneous canakinumab 4 mg/kg every 4 weeks. In part 2, 76 children who were in complete CR after canakinumab treatment were randomized to one of two tapering strategies. In one arm, the dose of canakinumab was reduced from 4 mg/kg to 2 mg/kg given every 4 weeks before eventually discontinuing treatment, and in other arm the duration between dosing was increased by 4-week intervals, from 4 to 8 weeks, then 12 weeks, then discontinuation.
If children were taking glucocorticoids or methotrexate, the treating physicians were encouraged to stop these medications if possible, with 34%-39% and 42%-59% of children, respectively, being able to do so. The rate depended on whether children had received canakinumab before entering the study because some had been recruited from a long-term extension study while others were naive to the biologic; all had inactive disease at study entry.
This was the first study to evaluate the effectiveness of canakinumab in enabling the discontinuation of methotrexate, but the primary objective was to assess whether more than 40% of randomized patients in either discontinuation arm remained in CR for 24 weeks after the first step of discontinuation. This was achieved in 71% of children who were in the dose-reduction arm and in 84% of children in the dose-prolongation arm (P ≤ .0001 for each arm vs. 40%).
Prior exposure to canakinumab did not seem to affect the maintenance of CR, but it was also found that more children who maintained CR at their second but not their first attempt at tapering were in CR than those who were still in CR at the first step (76% and 89% in the dose-reduction and dose-prolongation arms, respectively).
Among the two dosing regimens, failure occurred in 18% of children in the dose-reduction arm at the first step, 10% at the second, and 8% at the third, whereas 2.7% of children in the dose-prolongation arm experienced regimen failure at the first step, 6.1% at the second, and 15% at the third.
No substantial difference between the two tapering approaches was observed because the study was not powered to look at this. There was also no control arm, such as a group continuing treatment while the other groups tapered, or as Dr. Ramanan had pointed out, stopping canakinumab altogether.
“As long as the treatment was continued, even at very low dosage, most patients remained in inactive disease,” Dr. Quartier said. He added, however, that only a minority of patients could stop treatment completely. Treatment should not be stopped abruptly, he advised, because this was associated with a substantial number of disease flares that needed treatment to be reinstated.
These findings suggest that “a certain level of sustained inhibition of the IL-1 pathway seems important for the maintenance of CR in most sJIA patients,” Dr. Quartier and coinvestigators wrote in their article.
They added: “We believe that these results are relevant for clinical practice, particularly for designing personalized tapering strategies that can allow an adequate control of disease while minimizing the side effects of certain medications, notably glucocorticoids.”
The study was funded by Novartis in collaboration with the Pediatric Rheumatology International Trials Organization and the Pediatric Rheumatology Collaborative Study Group. Dr. Quartier was an investigator for the trial and has received research and consultancy fees from Novartis, among other pharmaceutical companies. Two of his coauthors are employees of Novartis. Dr. Ramanan has acted as an investigator in prior canakinumab trials and has received consultancy fees from Novartis and multiple other companies.
SOURCE: Quartier P et al. Arthritis Rheumatol. 2020 Aug 11. doi: 10.1002/art.41488.
A trial that tested two ways of tapering canakinumab (Ilaris) monotherapy in children with systemic juvenile idiopathic arthritis (sJIA) showed that both approaches might be feasible, but the lack of a control arm means that more data are needed before putting either into routine clinical practice.
Pierre Quartier, MD, and associates reported in Arthritis & Rheumatology. That’s with the proviso that children are taking canakinumab at the recommended dose of 4 mg/kg every 4 weeks and achieve clinical remission before any tapering is started.
The researchers found that 70%-80% of children who were in complete clinical remission (CR) maintained this for 6 months after the first dose-tapering step had been taken. However, at least one-fifth of study participants experienced a disease flare during treatment withdrawal, and only one-third were able to discontinue treatment altogether, which suggests continued treatment is needed.
“The results are a step in the right direction,” commented Athimaleipet Ramanan, MBBS, a consultant pediatric rheumatologist who was not involved in the study. They “offer the first vision of whether we can actually taper a medication” in sJIA because “until now we have not had any evidence for this,” he added.
“What we really need to know, which the study doesn’t tell you, is: Is decreasing the dose better than stopping?” said Dr. Ramanan, of the Bristol (England) Royal Hospital for Children.
Another thing that is important to know is: Does reducing the dose lead to the development of anti-drug antibodies? he said.
“There were some concerns that, when you give less of a monoclonal antibody, you might get more neutralizing anti-drug antibodies or you might make a drug more immunogenic,” he said. These data perhaps suggest that this isn’t the case because only one child on one occasion had detectable non-neutralizing anti-drug antibodies during the entire study, and that was 11 weeks after the last dose of canakinumab had been given.
Study results and design
Canakinumab is a monoclonal antibody that inhibits interleukin-1 (IL-1) that has been approved in the United States and Europe for the treatment of sJIA since 2013. Reducing a child’s exposure to canakinumab once their disease is under control is an attractive proposition given the treat-to-target approach used increasingly throughout modern rheumatologic practice. It could also help reduce the cost of what is an expensive treatment, compared with other available options for sJIA such as the interleukin-6 inhibitor tocilizumab (Actemra) or another IL-1 inhibitor anakinra (Kineret), which is not FDA-approved for use in sJIA and requires weekly injections.
“We don’t want to the disease to reappear, to flare, but we also don’t want to overuse treatments in these patients,” explained Dr. Quartier of Hôpital Necker-Enfants Malades, Assistance Publique-Hôpitaux de Paris in an interview.
The study he coconducted, given the acronym B-SPECIFIC-4 Patients, was an open-label study that consisted of two parts. In part 1, 182 children were given subcutaneous canakinumab 4 mg/kg every 4 weeks. In part 2, 76 children who were in complete CR after canakinumab treatment were randomized to one of two tapering strategies. In one arm, the dose of canakinumab was reduced from 4 mg/kg to 2 mg/kg given every 4 weeks before eventually discontinuing treatment, and in other arm the duration between dosing was increased by 4-week intervals, from 4 to 8 weeks, then 12 weeks, then discontinuation.
If children were taking glucocorticoids or methotrexate, the treating physicians were encouraged to stop these medications if possible, with 34%-39% and 42%-59% of children, respectively, being able to do so. The rate depended on whether children had received canakinumab before entering the study because some had been recruited from a long-term extension study while others were naive to the biologic; all had inactive disease at study entry.
This was the first study to evaluate the effectiveness of canakinumab in enabling the discontinuation of methotrexate, but the primary objective was to assess whether more than 40% of randomized patients in either discontinuation arm remained in CR for 24 weeks after the first step of discontinuation. This was achieved in 71% of children who were in the dose-reduction arm and in 84% of children in the dose-prolongation arm (P ≤ .0001 for each arm vs. 40%).
Prior exposure to canakinumab did not seem to affect the maintenance of CR, but it was also found that more children who maintained CR at their second but not their first attempt at tapering were in CR than those who were still in CR at the first step (76% and 89% in the dose-reduction and dose-prolongation arms, respectively).
Among the two dosing regimens, failure occurred in 18% of children in the dose-reduction arm at the first step, 10% at the second, and 8% at the third, whereas 2.7% of children in the dose-prolongation arm experienced regimen failure at the first step, 6.1% at the second, and 15% at the third.
No substantial difference between the two tapering approaches was observed because the study was not powered to look at this. There was also no control arm, such as a group continuing treatment while the other groups tapered, or as Dr. Ramanan had pointed out, stopping canakinumab altogether.
“As long as the treatment was continued, even at very low dosage, most patients remained in inactive disease,” Dr. Quartier said. He added, however, that only a minority of patients could stop treatment completely. Treatment should not be stopped abruptly, he advised, because this was associated with a substantial number of disease flares that needed treatment to be reinstated.
These findings suggest that “a certain level of sustained inhibition of the IL-1 pathway seems important for the maintenance of CR in most sJIA patients,” Dr. Quartier and coinvestigators wrote in their article.
They added: “We believe that these results are relevant for clinical practice, particularly for designing personalized tapering strategies that can allow an adequate control of disease while minimizing the side effects of certain medications, notably glucocorticoids.”
The study was funded by Novartis in collaboration with the Pediatric Rheumatology International Trials Organization and the Pediatric Rheumatology Collaborative Study Group. Dr. Quartier was an investigator for the trial and has received research and consultancy fees from Novartis, among other pharmaceutical companies. Two of his coauthors are employees of Novartis. Dr. Ramanan has acted as an investigator in prior canakinumab trials and has received consultancy fees from Novartis and multiple other companies.
SOURCE: Quartier P et al. Arthritis Rheumatol. 2020 Aug 11. doi: 10.1002/art.41488.
A trial that tested two ways of tapering canakinumab (Ilaris) monotherapy in children with systemic juvenile idiopathic arthritis (sJIA) showed that both approaches might be feasible, but the lack of a control arm means that more data are needed before putting either into routine clinical practice.
Pierre Quartier, MD, and associates reported in Arthritis & Rheumatology. That’s with the proviso that children are taking canakinumab at the recommended dose of 4 mg/kg every 4 weeks and achieve clinical remission before any tapering is started.
The researchers found that 70%-80% of children who were in complete clinical remission (CR) maintained this for 6 months after the first dose-tapering step had been taken. However, at least one-fifth of study participants experienced a disease flare during treatment withdrawal, and only one-third were able to discontinue treatment altogether, which suggests continued treatment is needed.
“The results are a step in the right direction,” commented Athimaleipet Ramanan, MBBS, a consultant pediatric rheumatologist who was not involved in the study. They “offer the first vision of whether we can actually taper a medication” in sJIA because “until now we have not had any evidence for this,” he added.
“What we really need to know, which the study doesn’t tell you, is: Is decreasing the dose better than stopping?” said Dr. Ramanan, of the Bristol (England) Royal Hospital for Children.
Another thing that is important to know is: Does reducing the dose lead to the development of anti-drug antibodies? he said.
“There were some concerns that, when you give less of a monoclonal antibody, you might get more neutralizing anti-drug antibodies or you might make a drug more immunogenic,” he said. These data perhaps suggest that this isn’t the case because only one child on one occasion had detectable non-neutralizing anti-drug antibodies during the entire study, and that was 11 weeks after the last dose of canakinumab had been given.
Study results and design
Canakinumab is a monoclonal antibody that inhibits interleukin-1 (IL-1) that has been approved in the United States and Europe for the treatment of sJIA since 2013. Reducing a child’s exposure to canakinumab once their disease is under control is an attractive proposition given the treat-to-target approach used increasingly throughout modern rheumatologic practice. It could also help reduce the cost of what is an expensive treatment, compared with other available options for sJIA such as the interleukin-6 inhibitor tocilizumab (Actemra) or another IL-1 inhibitor anakinra (Kineret), which is not FDA-approved for use in sJIA and requires weekly injections.
“We don’t want to the disease to reappear, to flare, but we also don’t want to overuse treatments in these patients,” explained Dr. Quartier of Hôpital Necker-Enfants Malades, Assistance Publique-Hôpitaux de Paris in an interview.
The study he coconducted, given the acronym B-SPECIFIC-4 Patients, was an open-label study that consisted of two parts. In part 1, 182 children were given subcutaneous canakinumab 4 mg/kg every 4 weeks. In part 2, 76 children who were in complete CR after canakinumab treatment were randomized to one of two tapering strategies. In one arm, the dose of canakinumab was reduced from 4 mg/kg to 2 mg/kg given every 4 weeks before eventually discontinuing treatment, and in other arm the duration between dosing was increased by 4-week intervals, from 4 to 8 weeks, then 12 weeks, then discontinuation.
If children were taking glucocorticoids or methotrexate, the treating physicians were encouraged to stop these medications if possible, with 34%-39% and 42%-59% of children, respectively, being able to do so. The rate depended on whether children had received canakinumab before entering the study because some had been recruited from a long-term extension study while others were naive to the biologic; all had inactive disease at study entry.
This was the first study to evaluate the effectiveness of canakinumab in enabling the discontinuation of methotrexate, but the primary objective was to assess whether more than 40% of randomized patients in either discontinuation arm remained in CR for 24 weeks after the first step of discontinuation. This was achieved in 71% of children who were in the dose-reduction arm and in 84% of children in the dose-prolongation arm (P ≤ .0001 for each arm vs. 40%).
Prior exposure to canakinumab did not seem to affect the maintenance of CR, but it was also found that more children who maintained CR at their second but not their first attempt at tapering were in CR than those who were still in CR at the first step (76% and 89% in the dose-reduction and dose-prolongation arms, respectively).
Among the two dosing regimens, failure occurred in 18% of children in the dose-reduction arm at the first step, 10% at the second, and 8% at the third, whereas 2.7% of children in the dose-prolongation arm experienced regimen failure at the first step, 6.1% at the second, and 15% at the third.
No substantial difference between the two tapering approaches was observed because the study was not powered to look at this. There was also no control arm, such as a group continuing treatment while the other groups tapered, or as Dr. Ramanan had pointed out, stopping canakinumab altogether.
“As long as the treatment was continued, even at very low dosage, most patients remained in inactive disease,” Dr. Quartier said. He added, however, that only a minority of patients could stop treatment completely. Treatment should not be stopped abruptly, he advised, because this was associated with a substantial number of disease flares that needed treatment to be reinstated.
These findings suggest that “a certain level of sustained inhibition of the IL-1 pathway seems important for the maintenance of CR in most sJIA patients,” Dr. Quartier and coinvestigators wrote in their article.
They added: “We believe that these results are relevant for clinical practice, particularly for designing personalized tapering strategies that can allow an adequate control of disease while minimizing the side effects of certain medications, notably glucocorticoids.”
The study was funded by Novartis in collaboration with the Pediatric Rheumatology International Trials Organization and the Pediatric Rheumatology Collaborative Study Group. Dr. Quartier was an investigator for the trial and has received research and consultancy fees from Novartis, among other pharmaceutical companies. Two of his coauthors are employees of Novartis. Dr. Ramanan has acted as an investigator in prior canakinumab trials and has received consultancy fees from Novartis and multiple other companies.
SOURCE: Quartier P et al. Arthritis Rheumatol. 2020 Aug 11. doi: 10.1002/art.41488.
FROM ARTHRITIS & RHEUMATOLOGY
Maternal depression may derail children’s school readiness
based on data from a cohort study of more than 50,000 children.
Previous research supports a link between maternal depression and poor cognitive development in children, but the effect of children’s age when exposed to depression on their development has not been well studied, wrote Elizabeth Wall-Wieler, PhD, of Stanford (Calif.) University, and colleagues.
In a population-based cohort study published in Pediatrics, the researchers examined data from 52,103 children born in Manitoba, Canada who completed the Early Development Instrument (EDI) between 2005 and 2016. The EDI is a 103-item questionnaire given to kindergarteners in a classroom setting during the second half of the school year. The EDI is designed to assess five domains: physical health and well-being, social competence, emotional maturity, language and cognitive development, and communication skills and general knowledge. The researchers assessed maternal depression using a combination of physician visits, hospitalizations, and pharmaceutical data; 19% of the children had a mother diagnosed with depression during the study.
Difficulties emerged in social functioning, emotion regulation
Children whose mothers had a diagnosis of depression before they reached age 5 years had a 1.17 times increased risk of having a problem in one or more of the developmental domains at school entry, compared with those whose mothers had no such diagnosis. Overall, exposure to maternal depression was significantly associated with problems in the areas of social competence (adjusted risk ratio 1.28), physical health and well-being (aRR 1.28), and emotional maturity (aRR 1.27) in kindergarten. For these three domains, children exposed to maternal depression between the ages of 4 and 5 years showed the greatest risk of developmental vulnerability, the researchers said.
“It is noteworthy that these difficulties were reported by the children’s teachers, avoiding negative biases inherent in having mothers with depression serve as informants,” Dr. Wall-Wieler and associates wrote.
The study findings were limited by several factors related to the observational design, including incomplete data on maternal depression prevalence and severity, the researchers said. In addition, the study did not account for confounding variables related to parenting style and information on the presence and psychiatric profiles of fathers.
The results support previous studies by identifying the impact of exposure to maternal depression on particular developmental domains, the researchers concluded. However, more research is needed to extend their findings.
“In particular, investigators should work to elucidate the mechanisms that underlie this intergenerational transmission to young children of mothers with depression at risk for social and emotional difficulties, focusing on aspects of the caregiving environments and behaviors to which these children are exposed,” Dr. Wall-Wieler and associates concluded.
Approximately 70% of the children exposed to maternal depression did not test as vulnerable in any of the developmental domains. Factors that promoted resilience in the children exposed to maternal depression who did not experience developmental vulnerability should be identified, they noted.
Maternal depression is a public health crisis
“The widespread prevalence of maternal depression in the United States and worldwide, with estimates ranging from 3% to 60%, constitutes a public health crisis, particularly one affecting low-income women and their children,” Stephanie Klees Goeglein, MD, and Yvette E. Yatchmink, MD, PhD, of Brown University, Providence, R.I., wrote in an accompanying editorial (Pediatrics. 2020 Aug 17. doi: 10.1542/peds.2020-010413). The study adds to the research and understanding of the negative impact of maternal depression on child development, but also raises questions because some key confounders were not addressed, mainly “the presence of prenatal depression, severity and chronicity of maternal depression, comorbidities, and parenting practices including the involvement of fathers or partners,” they noted.
Don’t discount the impact of prenatal depression, the editorialists stressed. “Surprising to some, prenatal depression is more common than postpartum depression and may be a major, unexplored contributor to the developmental vulnerability of the children in the sample,” Dr. Goeglein and Dr. Yatchmink said.
However, the fact that approximately 70% of the children exposed to maternal depression did not have developmental vulnerability in any domain is encouraging, and speaks to the resiliency of children to overcome adverse conditions, they added.
Pediatricians can play a role in reducing maternal depression by screening mothers starting at a prenatal visit and continuing through post partum, infancy, and beyond. In addition, actions to improve access to mental health care for mothers and to develop culturally-sensitive positive parenting programs may help reduce the effects of depression on mothers and children, Dr. Goeglein and Dr. Yatchmink emphasized.
Empower depressed mothers to seek assistance
The results were not surprising, but a study that “validates that which we feel we know is clinically significant,” Lillian M. Beard, MD, of Children’s National in Washington, said in an interview.
“This study restates why it is so important for us to evaluate and recognize maternal depression,” she said. “As a busy pediatrician, once I recognize a problem, I struggle with how to provide viable professional and valuable community resources for these mothers.
“My take-home message from the study is that we must screen for maternal depression. When we suspect maternal depression, it is usually present,” Dr. Beard said. “While we cannot change all the factors that contribute to this condition, to help the children we serve to be physically and emotionally sound, we must help empower their mothers to seek assistance, when needed, to be emotionally healthy,” she emphasized.
“More research is needed to recognize the efficacy and value of community-based assistance for maternal and family depression,” said Dr. Beard, who was not involved with the study and was asked to comment on the findings.
The study was supported by the Canadian Institutes of Health Research, the National Institute of Mental Health, and the National Institutes of Health. The researchers and editorialists had no financial conflicts to disclose. Dr. Beard, a member of the Pediatric News editorial advisory board, had no relevant financial disclosures.
SOURCE: Wall-Wieler E et al. Pediatrics. 2020 Aug 17. doi: 10.1542/peds.2020-0794.
based on data from a cohort study of more than 50,000 children.
Previous research supports a link between maternal depression and poor cognitive development in children, but the effect of children’s age when exposed to depression on their development has not been well studied, wrote Elizabeth Wall-Wieler, PhD, of Stanford (Calif.) University, and colleagues.
In a population-based cohort study published in Pediatrics, the researchers examined data from 52,103 children born in Manitoba, Canada who completed the Early Development Instrument (EDI) between 2005 and 2016. The EDI is a 103-item questionnaire given to kindergarteners in a classroom setting during the second half of the school year. The EDI is designed to assess five domains: physical health and well-being, social competence, emotional maturity, language and cognitive development, and communication skills and general knowledge. The researchers assessed maternal depression using a combination of physician visits, hospitalizations, and pharmaceutical data; 19% of the children had a mother diagnosed with depression during the study.
Difficulties emerged in social functioning, emotion regulation
Children whose mothers had a diagnosis of depression before they reached age 5 years had a 1.17 times increased risk of having a problem in one or more of the developmental domains at school entry, compared with those whose mothers had no such diagnosis. Overall, exposure to maternal depression was significantly associated with problems in the areas of social competence (adjusted risk ratio 1.28), physical health and well-being (aRR 1.28), and emotional maturity (aRR 1.27) in kindergarten. For these three domains, children exposed to maternal depression between the ages of 4 and 5 years showed the greatest risk of developmental vulnerability, the researchers said.
“It is noteworthy that these difficulties were reported by the children’s teachers, avoiding negative biases inherent in having mothers with depression serve as informants,” Dr. Wall-Wieler and associates wrote.
The study findings were limited by several factors related to the observational design, including incomplete data on maternal depression prevalence and severity, the researchers said. In addition, the study did not account for confounding variables related to parenting style and information on the presence and psychiatric profiles of fathers.
The results support previous studies by identifying the impact of exposure to maternal depression on particular developmental domains, the researchers concluded. However, more research is needed to extend their findings.
“In particular, investigators should work to elucidate the mechanisms that underlie this intergenerational transmission to young children of mothers with depression at risk for social and emotional difficulties, focusing on aspects of the caregiving environments and behaviors to which these children are exposed,” Dr. Wall-Wieler and associates concluded.
Approximately 70% of the children exposed to maternal depression did not test as vulnerable in any of the developmental domains. Factors that promoted resilience in the children exposed to maternal depression who did not experience developmental vulnerability should be identified, they noted.
Maternal depression is a public health crisis
“The widespread prevalence of maternal depression in the United States and worldwide, with estimates ranging from 3% to 60%, constitutes a public health crisis, particularly one affecting low-income women and their children,” Stephanie Klees Goeglein, MD, and Yvette E. Yatchmink, MD, PhD, of Brown University, Providence, R.I., wrote in an accompanying editorial (Pediatrics. 2020 Aug 17. doi: 10.1542/peds.2020-010413). The study adds to the research and understanding of the negative impact of maternal depression on child development, but also raises questions because some key confounders were not addressed, mainly “the presence of prenatal depression, severity and chronicity of maternal depression, comorbidities, and parenting practices including the involvement of fathers or partners,” they noted.
Don’t discount the impact of prenatal depression, the editorialists stressed. “Surprising to some, prenatal depression is more common than postpartum depression and may be a major, unexplored contributor to the developmental vulnerability of the children in the sample,” Dr. Goeglein and Dr. Yatchmink said.
However, the fact that approximately 70% of the children exposed to maternal depression did not have developmental vulnerability in any domain is encouraging, and speaks to the resiliency of children to overcome adverse conditions, they added.
Pediatricians can play a role in reducing maternal depression by screening mothers starting at a prenatal visit and continuing through post partum, infancy, and beyond. In addition, actions to improve access to mental health care for mothers and to develop culturally-sensitive positive parenting programs may help reduce the effects of depression on mothers and children, Dr. Goeglein and Dr. Yatchmink emphasized.
Empower depressed mothers to seek assistance
The results were not surprising, but a study that “validates that which we feel we know is clinically significant,” Lillian M. Beard, MD, of Children’s National in Washington, said in an interview.
“This study restates why it is so important for us to evaluate and recognize maternal depression,” she said. “As a busy pediatrician, once I recognize a problem, I struggle with how to provide viable professional and valuable community resources for these mothers.
“My take-home message from the study is that we must screen for maternal depression. When we suspect maternal depression, it is usually present,” Dr. Beard said. “While we cannot change all the factors that contribute to this condition, to help the children we serve to be physically and emotionally sound, we must help empower their mothers to seek assistance, when needed, to be emotionally healthy,” she emphasized.
“More research is needed to recognize the efficacy and value of community-based assistance for maternal and family depression,” said Dr. Beard, who was not involved with the study and was asked to comment on the findings.
The study was supported by the Canadian Institutes of Health Research, the National Institute of Mental Health, and the National Institutes of Health. The researchers and editorialists had no financial conflicts to disclose. Dr. Beard, a member of the Pediatric News editorial advisory board, had no relevant financial disclosures.
SOURCE: Wall-Wieler E et al. Pediatrics. 2020 Aug 17. doi: 10.1542/peds.2020-0794.
based on data from a cohort study of more than 50,000 children.
Previous research supports a link between maternal depression and poor cognitive development in children, but the effect of children’s age when exposed to depression on their development has not been well studied, wrote Elizabeth Wall-Wieler, PhD, of Stanford (Calif.) University, and colleagues.
In a population-based cohort study published in Pediatrics, the researchers examined data from 52,103 children born in Manitoba, Canada who completed the Early Development Instrument (EDI) between 2005 and 2016. The EDI is a 103-item questionnaire given to kindergarteners in a classroom setting during the second half of the school year. The EDI is designed to assess five domains: physical health and well-being, social competence, emotional maturity, language and cognitive development, and communication skills and general knowledge. The researchers assessed maternal depression using a combination of physician visits, hospitalizations, and pharmaceutical data; 19% of the children had a mother diagnosed with depression during the study.
Difficulties emerged in social functioning, emotion regulation
Children whose mothers had a diagnosis of depression before they reached age 5 years had a 1.17 times increased risk of having a problem in one or more of the developmental domains at school entry, compared with those whose mothers had no such diagnosis. Overall, exposure to maternal depression was significantly associated with problems in the areas of social competence (adjusted risk ratio 1.28), physical health and well-being (aRR 1.28), and emotional maturity (aRR 1.27) in kindergarten. For these three domains, children exposed to maternal depression between the ages of 4 and 5 years showed the greatest risk of developmental vulnerability, the researchers said.
“It is noteworthy that these difficulties were reported by the children’s teachers, avoiding negative biases inherent in having mothers with depression serve as informants,” Dr. Wall-Wieler and associates wrote.
The study findings were limited by several factors related to the observational design, including incomplete data on maternal depression prevalence and severity, the researchers said. In addition, the study did not account for confounding variables related to parenting style and information on the presence and psychiatric profiles of fathers.
The results support previous studies by identifying the impact of exposure to maternal depression on particular developmental domains, the researchers concluded. However, more research is needed to extend their findings.
“In particular, investigators should work to elucidate the mechanisms that underlie this intergenerational transmission to young children of mothers with depression at risk for social and emotional difficulties, focusing on aspects of the caregiving environments and behaviors to which these children are exposed,” Dr. Wall-Wieler and associates concluded.
Approximately 70% of the children exposed to maternal depression did not test as vulnerable in any of the developmental domains. Factors that promoted resilience in the children exposed to maternal depression who did not experience developmental vulnerability should be identified, they noted.
Maternal depression is a public health crisis
“The widespread prevalence of maternal depression in the United States and worldwide, with estimates ranging from 3% to 60%, constitutes a public health crisis, particularly one affecting low-income women and their children,” Stephanie Klees Goeglein, MD, and Yvette E. Yatchmink, MD, PhD, of Brown University, Providence, R.I., wrote in an accompanying editorial (Pediatrics. 2020 Aug 17. doi: 10.1542/peds.2020-010413). The study adds to the research and understanding of the negative impact of maternal depression on child development, but also raises questions because some key confounders were not addressed, mainly “the presence of prenatal depression, severity and chronicity of maternal depression, comorbidities, and parenting practices including the involvement of fathers or partners,” they noted.
Don’t discount the impact of prenatal depression, the editorialists stressed. “Surprising to some, prenatal depression is more common than postpartum depression and may be a major, unexplored contributor to the developmental vulnerability of the children in the sample,” Dr. Goeglein and Dr. Yatchmink said.
However, the fact that approximately 70% of the children exposed to maternal depression did not have developmental vulnerability in any domain is encouraging, and speaks to the resiliency of children to overcome adverse conditions, they added.
Pediatricians can play a role in reducing maternal depression by screening mothers starting at a prenatal visit and continuing through post partum, infancy, and beyond. In addition, actions to improve access to mental health care for mothers and to develop culturally-sensitive positive parenting programs may help reduce the effects of depression on mothers and children, Dr. Goeglein and Dr. Yatchmink emphasized.
Empower depressed mothers to seek assistance
The results were not surprising, but a study that “validates that which we feel we know is clinically significant,” Lillian M. Beard, MD, of Children’s National in Washington, said in an interview.
“This study restates why it is so important for us to evaluate and recognize maternal depression,” she said. “As a busy pediatrician, once I recognize a problem, I struggle with how to provide viable professional and valuable community resources for these mothers.
“My take-home message from the study is that we must screen for maternal depression. When we suspect maternal depression, it is usually present,” Dr. Beard said. “While we cannot change all the factors that contribute to this condition, to help the children we serve to be physically and emotionally sound, we must help empower their mothers to seek assistance, when needed, to be emotionally healthy,” she emphasized.
“More research is needed to recognize the efficacy and value of community-based assistance for maternal and family depression,” said Dr. Beard, who was not involved with the study and was asked to comment on the findings.
The study was supported by the Canadian Institutes of Health Research, the National Institute of Mental Health, and the National Institutes of Health. The researchers and editorialists had no financial conflicts to disclose. Dr. Beard, a member of the Pediatric News editorial advisory board, had no relevant financial disclosures.
SOURCE: Wall-Wieler E et al. Pediatrics. 2020 Aug 17. doi: 10.1542/peds.2020-0794.
FROM PEDIATRICS
Anxiety disorders begin earlier in life, differ by gender
Anxiety disorders start very early in life and may manifest themselves first as other conditions like social anxiety disorder, according to Jeffrey R. Strawn, MD.
An adolescent presenting to a mental health clinician with anxiety at 16 years old, for example has likely struggled with her anxiety for years before visiting a clinic. “That child may have been someone who had separation anxiety earlier in life and who as, even an infant, had behavioral inhibitions, that reluctance or timidness to explore new things, that tendency to retreat from novel stimuli,” Dr. Strawn, associate professor of psychiatry, pediatrics and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists. “Anxiety disorders are enduring and persistent, and they begin very early in life.”
Social anxiety disorder is one of the first anxiety disorders that appear in childhood or adolescents, which rises during puberty and during a time in a child’s life when they are dealing with new social pressures and challenges, such as graduating from elementary to middle school, Dr. Strawn noted. Generalized anxiety disorder is usually the next to emerge, followed by panic disorder. On the other hand, agoraphobia, another anxiety disorder that begins in childhood, “often represents behavioral avoidance as opposed to agoraphobia as we classically think about it as adult psychiatrists.”
Onset of anxiety disorders also differ by gender. “In terms of the emergence of these anxiety disorders, another thing that’s important to know is that the onset seems to be a bit different with regard to girls and boys. We see that break there emerging really around the time of puberty or as people are moving into late puberty, at least for girls,” Dr. Strawn said at the meeting presented by Global Academy for Medical Education. .
A shift occurs in amygdala prefrontal circuitry as children age, Dr. Strawn explained. Younger children do not have the ability to modulate the amygdala with their prefrontal cortex, but this amygdala–medial prefrontal cortex functional connectivity will change as children grow. A study by Dylan G. Gee, PhD, and colleagues found positive amygdala–medial prefrontal cortex functional connectivity at younger than 10 years old, and a “steady decline in amygdala activity” from 10-13 years to adulthood at 22 years old (J Neurosci. 2013 Mar 6;33[10]:4584-93).
“In essence, what we’re seeing is that there’s improvement or more effectiveness in terms of that connection between the prefrontal cortex, the amygdala, and that ability to amplify the brake to the amygdala,” Dr. Strawn said.
SSRIs, SNRIs for pediatric patients
Selective serotonin reuptake inhibitors can be effective for pediatric patients with anxiety disorders. Results from the Child/Adolescent Anxiety Multimodal Study (CAMS) show that patients with generalized separation or social anxiety disorder treated with sertraline or cognitive-behavioral therapy (CBT) for 3 months responded better to treatment than placebo. A combination of sertraline and CBT performing best, compared with either intervention alone (N Engl J Med. 2008;359:2753-66).
When examining treatment response in 76 patients from CAMS, the researchers saw improvement at 4 weeks from baseline in patients with anxiety symptoms receiving CBT, but no significant change in improvement after 4 weeks up to 12 weeks (J Child Adolesc Psychopharm. 2017 Aug 1. doi: 10.1089/cap.2016.0198).
“What that actually means is that your improvement at week 4 is better than your improvement at baseline, and your improvement at week 8 is greater than your improvement at week 4. Similarly, in your improvement, week 12 is greater than your improvement at week 8,” Dr. Strawn said.
However, “that’s not the case for aggressively titrated sertraline,” which had no statistically significant difference in improvement at 8 weeks and 12 weeks, he explained. “What this actually means is that, if I have not had improvement by week 8, there is a three-to-one odds against improvement over those next 4 weeks. The take-home message here is really that an adequate trial for an SSRI in pediatric anxiety disorders is probably about 8 weeks – not 12, not longer.”
Serotonin norepinephrine reuptake inhibitors (SNRIs) are also effective in pediatric patients with anxiety disorders.
“Both SNRIs as well as SSRIs have certainly demonstrated efficacy in terms of treating pediatric patients with anxiety, but there is a very important difference here with regard to the trajectory of improvement and also the magnitude of improvement,” Dr. Strawn said. SNRIs like atomoxetine, duloxetine, or venlafaxine “do not improve as rapidly and do not improve to the same extent as kids who are treated with an SSRI.”
Dose is another factor that affects symptom improvement in patients with pediatric anxiety disorders. In a 2018 meta-analysis, Dr. Strawn and colleagues found that patients treated with a higher dose of SSRIs demonstrated more rapid improvement at 2 weeks, compared with patients who received SNRIs (P = .002), but there was no significant difference in overall response trajectory (J Am Acad Child Adolesc Psychiatry. 2018 Apr;57[4]:235-44.E2).
Response to SSRIs can depend a patient’s genotype, Dr. Strawn said. The serotonin transporter promotor polymorphism has received “considerable attention in adults with depressive disorders primarily” but also might play a role in anxiety disorder response in pediatric patients. One study presented by his group at the 2019 annual meeting of the American Academy of Child & Adolescent Psychiatry showed that patients with a short-short copy of the serotonin transporter promoter polymorphism instead of a long copy had “shallower and less improvement over the course of treatment” when taking escitalopram.
“This is something that doesn’t necessarily compel us to use an SNRI over an SSRI, but it’s something that does give us some important information in terms of the trajectory of improvement,” he said.
When it comes to side effects of SNRIs and SSRIs, the profile is “pretty consistent with what we know to be the side effect profile in adults with depressive and anxiety disorders,” Dr. Strawn noted. “SNRIs tend to be a little bit better tolerated, both in terms of adverse event–related discontinuation and also in terms of their likelihood of producing activation.”
Patient and caregiver expectations can further affect response to treatment. “I think this has implications in terms of how we actively manage expectations and discussions about the evidence for interventions with our patients in the clinic.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Edgemont Pharmaceuticals, Eli Lilly, Forest Research Laboratories, Lundbeck, the National Institutes of Health, Neuronetics, and Shire. He also reported receiving royalties from Springer Publishing, and is a consultant for and receives material support from Assurex/Genesight.
Anxiety disorders start very early in life and may manifest themselves first as other conditions like social anxiety disorder, according to Jeffrey R. Strawn, MD.
An adolescent presenting to a mental health clinician with anxiety at 16 years old, for example has likely struggled with her anxiety for years before visiting a clinic. “That child may have been someone who had separation anxiety earlier in life and who as, even an infant, had behavioral inhibitions, that reluctance or timidness to explore new things, that tendency to retreat from novel stimuli,” Dr. Strawn, associate professor of psychiatry, pediatrics and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists. “Anxiety disorders are enduring and persistent, and they begin very early in life.”
Social anxiety disorder is one of the first anxiety disorders that appear in childhood or adolescents, which rises during puberty and during a time in a child’s life when they are dealing with new social pressures and challenges, such as graduating from elementary to middle school, Dr. Strawn noted. Generalized anxiety disorder is usually the next to emerge, followed by panic disorder. On the other hand, agoraphobia, another anxiety disorder that begins in childhood, “often represents behavioral avoidance as opposed to agoraphobia as we classically think about it as adult psychiatrists.”
Onset of anxiety disorders also differ by gender. “In terms of the emergence of these anxiety disorders, another thing that’s important to know is that the onset seems to be a bit different with regard to girls and boys. We see that break there emerging really around the time of puberty or as people are moving into late puberty, at least for girls,” Dr. Strawn said at the meeting presented by Global Academy for Medical Education. .
A shift occurs in amygdala prefrontal circuitry as children age, Dr. Strawn explained. Younger children do not have the ability to modulate the amygdala with their prefrontal cortex, but this amygdala–medial prefrontal cortex functional connectivity will change as children grow. A study by Dylan G. Gee, PhD, and colleagues found positive amygdala–medial prefrontal cortex functional connectivity at younger than 10 years old, and a “steady decline in amygdala activity” from 10-13 years to adulthood at 22 years old (J Neurosci. 2013 Mar 6;33[10]:4584-93).
“In essence, what we’re seeing is that there’s improvement or more effectiveness in terms of that connection between the prefrontal cortex, the amygdala, and that ability to amplify the brake to the amygdala,” Dr. Strawn said.
SSRIs, SNRIs for pediatric patients
Selective serotonin reuptake inhibitors can be effective for pediatric patients with anxiety disorders. Results from the Child/Adolescent Anxiety Multimodal Study (CAMS) show that patients with generalized separation or social anxiety disorder treated with sertraline or cognitive-behavioral therapy (CBT) for 3 months responded better to treatment than placebo. A combination of sertraline and CBT performing best, compared with either intervention alone (N Engl J Med. 2008;359:2753-66).
When examining treatment response in 76 patients from CAMS, the researchers saw improvement at 4 weeks from baseline in patients with anxiety symptoms receiving CBT, but no significant change in improvement after 4 weeks up to 12 weeks (J Child Adolesc Psychopharm. 2017 Aug 1. doi: 10.1089/cap.2016.0198).
“What that actually means is that your improvement at week 4 is better than your improvement at baseline, and your improvement at week 8 is greater than your improvement at week 4. Similarly, in your improvement, week 12 is greater than your improvement at week 8,” Dr. Strawn said.
However, “that’s not the case for aggressively titrated sertraline,” which had no statistically significant difference in improvement at 8 weeks and 12 weeks, he explained. “What this actually means is that, if I have not had improvement by week 8, there is a three-to-one odds against improvement over those next 4 weeks. The take-home message here is really that an adequate trial for an SSRI in pediatric anxiety disorders is probably about 8 weeks – not 12, not longer.”
Serotonin norepinephrine reuptake inhibitors (SNRIs) are also effective in pediatric patients with anxiety disorders.
“Both SNRIs as well as SSRIs have certainly demonstrated efficacy in terms of treating pediatric patients with anxiety, but there is a very important difference here with regard to the trajectory of improvement and also the magnitude of improvement,” Dr. Strawn said. SNRIs like atomoxetine, duloxetine, or venlafaxine “do not improve as rapidly and do not improve to the same extent as kids who are treated with an SSRI.”
Dose is another factor that affects symptom improvement in patients with pediatric anxiety disorders. In a 2018 meta-analysis, Dr. Strawn and colleagues found that patients treated with a higher dose of SSRIs demonstrated more rapid improvement at 2 weeks, compared with patients who received SNRIs (P = .002), but there was no significant difference in overall response trajectory (J Am Acad Child Adolesc Psychiatry. 2018 Apr;57[4]:235-44.E2).
Response to SSRIs can depend a patient’s genotype, Dr. Strawn said. The serotonin transporter promotor polymorphism has received “considerable attention in adults with depressive disorders primarily” but also might play a role in anxiety disorder response in pediatric patients. One study presented by his group at the 2019 annual meeting of the American Academy of Child & Adolescent Psychiatry showed that patients with a short-short copy of the serotonin transporter promoter polymorphism instead of a long copy had “shallower and less improvement over the course of treatment” when taking escitalopram.
“This is something that doesn’t necessarily compel us to use an SNRI over an SSRI, but it’s something that does give us some important information in terms of the trajectory of improvement,” he said.
When it comes to side effects of SNRIs and SSRIs, the profile is “pretty consistent with what we know to be the side effect profile in adults with depressive and anxiety disorders,” Dr. Strawn noted. “SNRIs tend to be a little bit better tolerated, both in terms of adverse event–related discontinuation and also in terms of their likelihood of producing activation.”
Patient and caregiver expectations can further affect response to treatment. “I think this has implications in terms of how we actively manage expectations and discussions about the evidence for interventions with our patients in the clinic.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Edgemont Pharmaceuticals, Eli Lilly, Forest Research Laboratories, Lundbeck, the National Institutes of Health, Neuronetics, and Shire. He also reported receiving royalties from Springer Publishing, and is a consultant for and receives material support from Assurex/Genesight.
Anxiety disorders start very early in life and may manifest themselves first as other conditions like social anxiety disorder, according to Jeffrey R. Strawn, MD.
An adolescent presenting to a mental health clinician with anxiety at 16 years old, for example has likely struggled with her anxiety for years before visiting a clinic. “That child may have been someone who had separation anxiety earlier in life and who as, even an infant, had behavioral inhibitions, that reluctance or timidness to explore new things, that tendency to retreat from novel stimuli,” Dr. Strawn, associate professor of psychiatry, pediatrics and clinical pharmacology at Cincinnati Children’s Hospital Medical Center, said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists. “Anxiety disorders are enduring and persistent, and they begin very early in life.”
Social anxiety disorder is one of the first anxiety disorders that appear in childhood or adolescents, which rises during puberty and during a time in a child’s life when they are dealing with new social pressures and challenges, such as graduating from elementary to middle school, Dr. Strawn noted. Generalized anxiety disorder is usually the next to emerge, followed by panic disorder. On the other hand, agoraphobia, another anxiety disorder that begins in childhood, “often represents behavioral avoidance as opposed to agoraphobia as we classically think about it as adult psychiatrists.”
Onset of anxiety disorders also differ by gender. “In terms of the emergence of these anxiety disorders, another thing that’s important to know is that the onset seems to be a bit different with regard to girls and boys. We see that break there emerging really around the time of puberty or as people are moving into late puberty, at least for girls,” Dr. Strawn said at the meeting presented by Global Academy for Medical Education. .
A shift occurs in amygdala prefrontal circuitry as children age, Dr. Strawn explained. Younger children do not have the ability to modulate the amygdala with their prefrontal cortex, but this amygdala–medial prefrontal cortex functional connectivity will change as children grow. A study by Dylan G. Gee, PhD, and colleagues found positive amygdala–medial prefrontal cortex functional connectivity at younger than 10 years old, and a “steady decline in amygdala activity” from 10-13 years to adulthood at 22 years old (J Neurosci. 2013 Mar 6;33[10]:4584-93).
“In essence, what we’re seeing is that there’s improvement or more effectiveness in terms of that connection between the prefrontal cortex, the amygdala, and that ability to amplify the brake to the amygdala,” Dr. Strawn said.
SSRIs, SNRIs for pediatric patients
Selective serotonin reuptake inhibitors can be effective for pediatric patients with anxiety disorders. Results from the Child/Adolescent Anxiety Multimodal Study (CAMS) show that patients with generalized separation or social anxiety disorder treated with sertraline or cognitive-behavioral therapy (CBT) for 3 months responded better to treatment than placebo. A combination of sertraline and CBT performing best, compared with either intervention alone (N Engl J Med. 2008;359:2753-66).
When examining treatment response in 76 patients from CAMS, the researchers saw improvement at 4 weeks from baseline in patients with anxiety symptoms receiving CBT, but no significant change in improvement after 4 weeks up to 12 weeks (J Child Adolesc Psychopharm. 2017 Aug 1. doi: 10.1089/cap.2016.0198).
“What that actually means is that your improvement at week 4 is better than your improvement at baseline, and your improvement at week 8 is greater than your improvement at week 4. Similarly, in your improvement, week 12 is greater than your improvement at week 8,” Dr. Strawn said.
However, “that’s not the case for aggressively titrated sertraline,” which had no statistically significant difference in improvement at 8 weeks and 12 weeks, he explained. “What this actually means is that, if I have not had improvement by week 8, there is a three-to-one odds against improvement over those next 4 weeks. The take-home message here is really that an adequate trial for an SSRI in pediatric anxiety disorders is probably about 8 weeks – not 12, not longer.”
Serotonin norepinephrine reuptake inhibitors (SNRIs) are also effective in pediatric patients with anxiety disorders.
“Both SNRIs as well as SSRIs have certainly demonstrated efficacy in terms of treating pediatric patients with anxiety, but there is a very important difference here with regard to the trajectory of improvement and also the magnitude of improvement,” Dr. Strawn said. SNRIs like atomoxetine, duloxetine, or venlafaxine “do not improve as rapidly and do not improve to the same extent as kids who are treated with an SSRI.”
Dose is another factor that affects symptom improvement in patients with pediatric anxiety disorders. In a 2018 meta-analysis, Dr. Strawn and colleagues found that patients treated with a higher dose of SSRIs demonstrated more rapid improvement at 2 weeks, compared with patients who received SNRIs (P = .002), but there was no significant difference in overall response trajectory (J Am Acad Child Adolesc Psychiatry. 2018 Apr;57[4]:235-44.E2).
Response to SSRIs can depend a patient’s genotype, Dr. Strawn said. The serotonin transporter promotor polymorphism has received “considerable attention in adults with depressive disorders primarily” but also might play a role in anxiety disorder response in pediatric patients. One study presented by his group at the 2019 annual meeting of the American Academy of Child & Adolescent Psychiatry showed that patients with a short-short copy of the serotonin transporter promoter polymorphism instead of a long copy had “shallower and less improvement over the course of treatment” when taking escitalopram.
“This is something that doesn’t necessarily compel us to use an SNRI over an SSRI, but it’s something that does give us some important information in terms of the trajectory of improvement,” he said.
When it comes to side effects of SNRIs and SSRIs, the profile is “pretty consistent with what we know to be the side effect profile in adults with depressive and anxiety disorders,” Dr. Strawn noted. “SNRIs tend to be a little bit better tolerated, both in terms of adverse event–related discontinuation and also in terms of their likelihood of producing activation.”
Patient and caregiver expectations can further affect response to treatment. “I think this has implications in terms of how we actively manage expectations and discussions about the evidence for interventions with our patients in the clinic.”
Global Academy and this news organization are owned by the same parent company. Dr. Strawn reported receiving research support from Edgemont Pharmaceuticals, Eli Lilly, Forest Research Laboratories, Lundbeck, the National Institutes of Health, Neuronetics, and Shire. He also reported receiving royalties from Springer Publishing, and is a consultant for and receives material support from Assurex/Genesight.
FROM Focus on Neuropsychiatry 2020
A 4-year-old with a lesion on her cheek, which grew and became firmer over two months
The patient was diagnosed with idiopathic facial aseptic granuloma (IFAG) based on the clinical findings, as well as the associated history of chalazia and erythematous papules seen in childhood rosacea.
She was treated with several months of azithromycin, sulfur wash, and metronidazole cream with improvement of some of the smaller lesions but no change on the larger nodules. Later she was treated with oral and topical ivermectin with no improvement. Some of the nodules slowly resolved except for the larger lesion on the right cheek. She was later treated with a 6-week course of clarithromycin with partial improvement of the nodule. The lesion resolved after 2 months of stopping clarithromycin.
IFAG is a rare condition seen in prepubescent children. The etiology of this condition is not well understood and is thought to be on the spectrum of childhood rosacea.1 From several recent reports, IFAG usually is seen in children with associated conditions including chalazia, conjunctivitis, blepharitis, and telangiectasias, which can be seen in patients with rosacea. These associated findings suggest the possibility of IFAG being a form of granulomatous rosacea in children.
This condition presents in childhood between the ages of 8 months and 13 years. Most of the cases occur in toddlers, and girls appear to be more affected than boys. The lesions appear as pink, rubbery, nontender, nonfluctuant nodules on the cheeks, which can be single or multiple. A large prospective study in 30 children demonstrated that more 70% of the lesions cultured were negative for bacteria. Histologic analysis of some of the lesions showed a chronic dermal lymphohistiocytic granulomatous perifollicular infiltrate with numerous foreign body–type giant cells.2
The differential diagnosis of these lesions should include infectious pyodermas such as mycobacterial infections, cutaneous leishmaniasis, and botryomycosis; deep fungal infections such as sporotrichosis, coccidioidomycosis, and cryptococcosis; childhood nodulocystic acne; pilomatrixoma; epidermoid cyst; vascular tumors or malformations; and leukemia cutis.3
The diagnosis is usually clinical but in atypical cases a skin biopsy with tissue cultures should be performed. The decision to biopsy these lesions will need to be done in a one by one basis, as a biopsy may leave scaring on the area affected.
It has been postulated that a color Doppler ultrasound of the lesion may be a helpful ancillary study. Echographic findings show a well demarcated solid-cystic, hypoechoic dermal lesion, the largest axis of which lies parallel to the skin surface. The lesion lacks calcium deposits. Other findings include increased echogenicity of the underlaying hypodermis. The findings may vary depending on the stage of the lesion.4
The course of the condition may last on average months to years. Some lesions resolve spontaneously and others may respond to courses of oral antibiotics such as clarithromycin, azithromycin, or ivermectin. In our patient, several lesions improved with oral antibiotics, but the larger lesions were more persistent and resolved after a year.
The lesions usually resolve without scarring. In those patients with associated rosacea, maintenance topical treatments may be warranted and also may need follow-up with ophthalmology because they tend to commonly have ocular rosacea as well.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Pediatr Dermatol. 2013 Jan-Feb;30(1):109-11.
2. Br J Dermatol. 2007 Apr;156(4):705-8.
3. Pediatr Dermatol. 2018 Jul;35(4):490-3.
4. Actas Dermosifiliogr. 2019 Oct;110(8):637-41.
The patient was diagnosed with idiopathic facial aseptic granuloma (IFAG) based on the clinical findings, as well as the associated history of chalazia and erythematous papules seen in childhood rosacea.
She was treated with several months of azithromycin, sulfur wash, and metronidazole cream with improvement of some of the smaller lesions but no change on the larger nodules. Later she was treated with oral and topical ivermectin with no improvement. Some of the nodules slowly resolved except for the larger lesion on the right cheek. She was later treated with a 6-week course of clarithromycin with partial improvement of the nodule. The lesion resolved after 2 months of stopping clarithromycin.
IFAG is a rare condition seen in prepubescent children. The etiology of this condition is not well understood and is thought to be on the spectrum of childhood rosacea.1 From several recent reports, IFAG usually is seen in children with associated conditions including chalazia, conjunctivitis, blepharitis, and telangiectasias, which can be seen in patients with rosacea. These associated findings suggest the possibility of IFAG being a form of granulomatous rosacea in children.
This condition presents in childhood between the ages of 8 months and 13 years. Most of the cases occur in toddlers, and girls appear to be more affected than boys. The lesions appear as pink, rubbery, nontender, nonfluctuant nodules on the cheeks, which can be single or multiple. A large prospective study in 30 children demonstrated that more 70% of the lesions cultured were negative for bacteria. Histologic analysis of some of the lesions showed a chronic dermal lymphohistiocytic granulomatous perifollicular infiltrate with numerous foreign body–type giant cells.2
The differential diagnosis of these lesions should include infectious pyodermas such as mycobacterial infections, cutaneous leishmaniasis, and botryomycosis; deep fungal infections such as sporotrichosis, coccidioidomycosis, and cryptococcosis; childhood nodulocystic acne; pilomatrixoma; epidermoid cyst; vascular tumors or malformations; and leukemia cutis.3
The diagnosis is usually clinical but in atypical cases a skin biopsy with tissue cultures should be performed. The decision to biopsy these lesions will need to be done in a one by one basis, as a biopsy may leave scaring on the area affected.
It has been postulated that a color Doppler ultrasound of the lesion may be a helpful ancillary study. Echographic findings show a well demarcated solid-cystic, hypoechoic dermal lesion, the largest axis of which lies parallel to the skin surface. The lesion lacks calcium deposits. Other findings include increased echogenicity of the underlaying hypodermis. The findings may vary depending on the stage of the lesion.4
The course of the condition may last on average months to years. Some lesions resolve spontaneously and others may respond to courses of oral antibiotics such as clarithromycin, azithromycin, or ivermectin. In our patient, several lesions improved with oral antibiotics, but the larger lesions were more persistent and resolved after a year.
The lesions usually resolve without scarring. In those patients with associated rosacea, maintenance topical treatments may be warranted and also may need follow-up with ophthalmology because they tend to commonly have ocular rosacea as well.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Pediatr Dermatol. 2013 Jan-Feb;30(1):109-11.
2. Br J Dermatol. 2007 Apr;156(4):705-8.
3. Pediatr Dermatol. 2018 Jul;35(4):490-3.
4. Actas Dermosifiliogr. 2019 Oct;110(8):637-41.
The patient was diagnosed with idiopathic facial aseptic granuloma (IFAG) based on the clinical findings, as well as the associated history of chalazia and erythematous papules seen in childhood rosacea.
She was treated with several months of azithromycin, sulfur wash, and metronidazole cream with improvement of some of the smaller lesions but no change on the larger nodules. Later she was treated with oral and topical ivermectin with no improvement. Some of the nodules slowly resolved except for the larger lesion on the right cheek. She was later treated with a 6-week course of clarithromycin with partial improvement of the nodule. The lesion resolved after 2 months of stopping clarithromycin.
IFAG is a rare condition seen in prepubescent children. The etiology of this condition is not well understood and is thought to be on the spectrum of childhood rosacea.1 From several recent reports, IFAG usually is seen in children with associated conditions including chalazia, conjunctivitis, blepharitis, and telangiectasias, which can be seen in patients with rosacea. These associated findings suggest the possibility of IFAG being a form of granulomatous rosacea in children.
This condition presents in childhood between the ages of 8 months and 13 years. Most of the cases occur in toddlers, and girls appear to be more affected than boys. The lesions appear as pink, rubbery, nontender, nonfluctuant nodules on the cheeks, which can be single or multiple. A large prospective study in 30 children demonstrated that more 70% of the lesions cultured were negative for bacteria. Histologic analysis of some of the lesions showed a chronic dermal lymphohistiocytic granulomatous perifollicular infiltrate with numerous foreign body–type giant cells.2
The differential diagnosis of these lesions should include infectious pyodermas such as mycobacterial infections, cutaneous leishmaniasis, and botryomycosis; deep fungal infections such as sporotrichosis, coccidioidomycosis, and cryptococcosis; childhood nodulocystic acne; pilomatrixoma; epidermoid cyst; vascular tumors or malformations; and leukemia cutis.3
The diagnosis is usually clinical but in atypical cases a skin biopsy with tissue cultures should be performed. The decision to biopsy these lesions will need to be done in a one by one basis, as a biopsy may leave scaring on the area affected.
It has been postulated that a color Doppler ultrasound of the lesion may be a helpful ancillary study. Echographic findings show a well demarcated solid-cystic, hypoechoic dermal lesion, the largest axis of which lies parallel to the skin surface. The lesion lacks calcium deposits. Other findings include increased echogenicity of the underlaying hypodermis. The findings may vary depending on the stage of the lesion.4
The course of the condition may last on average months to years. Some lesions resolve spontaneously and others may respond to courses of oral antibiotics such as clarithromycin, azithromycin, or ivermectin. In our patient, several lesions improved with oral antibiotics, but the larger lesions were more persistent and resolved after a year.
The lesions usually resolve without scarring. In those patients with associated rosacea, maintenance topical treatments may be warranted and also may need follow-up with ophthalmology because they tend to commonly have ocular rosacea as well.
Dr. Matiz is a pediatric dermatologist at Southern California Permanente Medical Group, San Diego. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Pediatr Dermatol. 2013 Jan-Feb;30(1):109-11.
2. Br J Dermatol. 2007 Apr;156(4):705-8.
3. Pediatr Dermatol. 2018 Jul;35(4):490-3.
4. Actas Dermosifiliogr. 2019 Oct;110(8):637-41.
A 4-year-old female is brought to our pediatric dermatology clinic for evaluation of a persistent lesion on the cheek.
The mother of the child reports that the lesion started as a small "bug bite" and then started growing and getting firmer for the past 2 months. The girl has developed other smaller red, pimple-like lesions on the cheeks and one of them is starting to increase in size.
She denies any tenderness on the area or any purulent discharge. She has had no fevers, chills, weight loss, nose bleeds, fatigue, or any other symptoms. The mother has not noted any changes on the child's body odor, any rapid growth, or hair on her axillary or pubic area. She was treated with three different courses of oral antibiotics including cephalexin, trimethoprim/sulfamethoxazole, and clindamycin, as well as topical mupirocin, with no improvement.
Her past medical history is significant for several episodes of eyelid cysts that were treated with warm compresses and topical erythromycin ointment. The family history is significant for the father having severe acne as a teenager. She has two cats, she has not traveled, and she has an older sister who has no lesions.
On physical examination she is a lovely 4-year-old female in no acute distress. Her height is on the 70th percentile and weight on the 40th percentile for her age. Her blood pressure is 95/84 with a heart rate of 96. On skin examination she has several pink macules and papules on her bilateral cheeks. On the left cheek there are two pink nodules: One is 1 cm, and the other is 7 mm. The nodules are not tender. There is no warmth, fluctuance, or discharge from the lesions.
She has no cervical lymphadenopathy. She has no axillary or pubic hair. She is Tanner stage I.
COVID-19 child case count now over 400,000
according to a new report from the American Academy of Pediatrics and the Children’s Hospital Association.
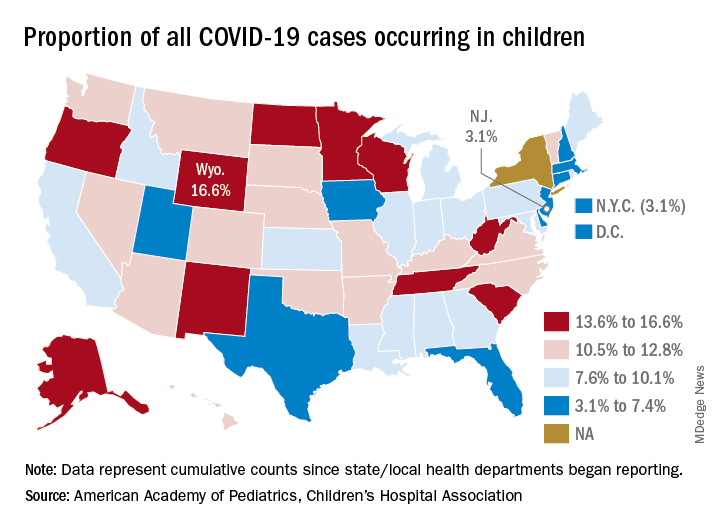
The 406,000 children who have tested positive for COVID-19 represent 9.1% of all cases reported so far by 49 states (New York does not provide age distribution), New York City, the District of Columbia, Puerto Rico, and Guam. Since the proportion of child cases also was 9.1% on Aug. 6, the most recent week is the first without an increase since tracking began in mid-April, the report shows.
State-level data show that Wyoming has the highest percentage of child cases (16.6%) after Alabama changed its “definition of child case from 0-24 to 0-17 years, resulting in a downward revision of cumulative child cases,” the AAP and the CHA said. Alabama’s proportion of such cases dropped from 22.5% to 9.0%.
New Jersey had the lowest rate (3.1%) again this week, along with New York City, but both were up slightly from the week before, when New Jersey was at 2.9% and N.Y.C. was 3.0%. The only states, other than Alabama, that saw declines over the last week were Arkansas, Massachusetts, Mississippi, South Dakota, Texas, and West Virginia. Texas, however, has reported age for only 8% of its confirmed cases, the report noted.
The overall rate of child COVID-19 cases as of Aug. 13 was 538 per 100,000 children, up from 500.7 per 100,000 a week earlier. Arizona was again highest among the states with a rate of 1,254 per 100,000 (up from 1,206) and Vermont was lowest at 121, although Puerto Rico (114) and Guam (88) were lower still, the AAP/CHA data indicate.
For the nine states that report testing information for children, Arizona has the highest positivity rate at 18.3% and West Virginia has the lowest at 3.6%. Data on hospitalizations – available from 21 states and N.Y.C. – show that 3,849 children have been admitted, with rates varying from 0.2% of children in Hawaii to 8.8% in the Big Apple, according to the report.
More specific information on child cases, such as symptoms or underlying conditions, is not being provided by states at this time, the AAP and CHA pointed out.
according to a new report from the American Academy of Pediatrics and the Children’s Hospital Association.

The 406,000 children who have tested positive for COVID-19 represent 9.1% of all cases reported so far by 49 states (New York does not provide age distribution), New York City, the District of Columbia, Puerto Rico, and Guam. Since the proportion of child cases also was 9.1% on Aug. 6, the most recent week is the first without an increase since tracking began in mid-April, the report shows.
State-level data show that Wyoming has the highest percentage of child cases (16.6%) after Alabama changed its “definition of child case from 0-24 to 0-17 years, resulting in a downward revision of cumulative child cases,” the AAP and the CHA said. Alabama’s proportion of such cases dropped from 22.5% to 9.0%.
New Jersey had the lowest rate (3.1%) again this week, along with New York City, but both were up slightly from the week before, when New Jersey was at 2.9% and N.Y.C. was 3.0%. The only states, other than Alabama, that saw declines over the last week were Arkansas, Massachusetts, Mississippi, South Dakota, Texas, and West Virginia. Texas, however, has reported age for only 8% of its confirmed cases, the report noted.
The overall rate of child COVID-19 cases as of Aug. 13 was 538 per 100,000 children, up from 500.7 per 100,000 a week earlier. Arizona was again highest among the states with a rate of 1,254 per 100,000 (up from 1,206) and Vermont was lowest at 121, although Puerto Rico (114) and Guam (88) were lower still, the AAP/CHA data indicate.
For the nine states that report testing information for children, Arizona has the highest positivity rate at 18.3% and West Virginia has the lowest at 3.6%. Data on hospitalizations – available from 21 states and N.Y.C. – show that 3,849 children have been admitted, with rates varying from 0.2% of children in Hawaii to 8.8% in the Big Apple, according to the report.
More specific information on child cases, such as symptoms or underlying conditions, is not being provided by states at this time, the AAP and CHA pointed out.
according to a new report from the American Academy of Pediatrics and the Children’s Hospital Association.

The 406,000 children who have tested positive for COVID-19 represent 9.1% of all cases reported so far by 49 states (New York does not provide age distribution), New York City, the District of Columbia, Puerto Rico, and Guam. Since the proportion of child cases also was 9.1% on Aug. 6, the most recent week is the first without an increase since tracking began in mid-April, the report shows.
State-level data show that Wyoming has the highest percentage of child cases (16.6%) after Alabama changed its “definition of child case from 0-24 to 0-17 years, resulting in a downward revision of cumulative child cases,” the AAP and the CHA said. Alabama’s proportion of such cases dropped from 22.5% to 9.0%.
New Jersey had the lowest rate (3.1%) again this week, along with New York City, but both were up slightly from the week before, when New Jersey was at 2.9% and N.Y.C. was 3.0%. The only states, other than Alabama, that saw declines over the last week were Arkansas, Massachusetts, Mississippi, South Dakota, Texas, and West Virginia. Texas, however, has reported age for only 8% of its confirmed cases, the report noted.
The overall rate of child COVID-19 cases as of Aug. 13 was 538 per 100,000 children, up from 500.7 per 100,000 a week earlier. Arizona was again highest among the states with a rate of 1,254 per 100,000 (up from 1,206) and Vermont was lowest at 121, although Puerto Rico (114) and Guam (88) were lower still, the AAP/CHA data indicate.
For the nine states that report testing information for children, Arizona has the highest positivity rate at 18.3% and West Virginia has the lowest at 3.6%. Data on hospitalizations – available from 21 states and N.Y.C. – show that 3,849 children have been admitted, with rates varying from 0.2% of children in Hawaii to 8.8% in the Big Apple, according to the report.
More specific information on child cases, such as symptoms or underlying conditions, is not being provided by states at this time, the AAP and CHA pointed out.