User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
FDA approves clinical trials for cannabinoid drug designed to reduce COVID-19 lung inflammation
The US Food and Drug Administration has approved phase one clinical trials for a synthetic cannabinoid drug designed to treat acute respiratory distress syndrome (ARDS), a life-threatening lung condition which may occur in severe cases of the novel coronavirus, Forbes reported.
ARDS can be triggered by over-creation of cytokines, proteins which tell the body to produce more inflammation, Forbes said.
The drug going to clinical trials, ARDS-003, would “dampen the cytokine release” and prevent development of ARDS, Tetra Bio-Pharma company CEO and chief regulatory officer Guy Chamberland, MD, said in a news release.
Consequences of ARDS include scarring of the lungs and organ injury caused by the decrease in blood to the tissue, the release said.
“The FDA repeatedly stated that they want clinical trials for COVID-19 to begin as soon as possible, as long as they meet regulatory requirements,” the news release said. “The medical community is in urgent need of drugs that can reduce the strength and duration of the severe inflammation. It is anticipated that this type of new drug would favorably impact health care and possibly reduce the negative health outcomes post infection.”
ARDS-003 works by binding to CB2 receptors, one of two main receptors in the endocannabinoid system which modulate inflammation and cytokine activity, Forbes said. CB2 receptors don’t bring on a psychoactive high.
Phase one clinical trials would begin enrolling participants in December to determine if the drug is safe, Chamberland said, according to Forbes.
If phase one is successful, phase two would test the drug on a larger group in the second quarter of 2021 to assess safety and tolerability for people who have COVID-19.
If phase two is successful, the company may seek emergency authorization through the FDA, Chamberland said. Phase three would start at the end of 2021.
Tetra Bio-Pharma says it has already contracted with Dalton Pharma Services to manufacture the active pharmaceutical ingredient (API), HU-308, and the finished drug product ARDS-003.
This article first appeared on Medscape.com.
The US Food and Drug Administration has approved phase one clinical trials for a synthetic cannabinoid drug designed to treat acute respiratory distress syndrome (ARDS), a life-threatening lung condition which may occur in severe cases of the novel coronavirus, Forbes reported.
ARDS can be triggered by over-creation of cytokines, proteins which tell the body to produce more inflammation, Forbes said.
The drug going to clinical trials, ARDS-003, would “dampen the cytokine release” and prevent development of ARDS, Tetra Bio-Pharma company CEO and chief regulatory officer Guy Chamberland, MD, said in a news release.
Consequences of ARDS include scarring of the lungs and organ injury caused by the decrease in blood to the tissue, the release said.
“The FDA repeatedly stated that they want clinical trials for COVID-19 to begin as soon as possible, as long as they meet regulatory requirements,” the news release said. “The medical community is in urgent need of drugs that can reduce the strength and duration of the severe inflammation. It is anticipated that this type of new drug would favorably impact health care and possibly reduce the negative health outcomes post infection.”
ARDS-003 works by binding to CB2 receptors, one of two main receptors in the endocannabinoid system which modulate inflammation and cytokine activity, Forbes said. CB2 receptors don’t bring on a psychoactive high.
Phase one clinical trials would begin enrolling participants in December to determine if the drug is safe, Chamberland said, according to Forbes.
If phase one is successful, phase two would test the drug on a larger group in the second quarter of 2021 to assess safety and tolerability for people who have COVID-19.
If phase two is successful, the company may seek emergency authorization through the FDA, Chamberland said. Phase three would start at the end of 2021.
Tetra Bio-Pharma says it has already contracted with Dalton Pharma Services to manufacture the active pharmaceutical ingredient (API), HU-308, and the finished drug product ARDS-003.
This article first appeared on Medscape.com.
The US Food and Drug Administration has approved phase one clinical trials for a synthetic cannabinoid drug designed to treat acute respiratory distress syndrome (ARDS), a life-threatening lung condition which may occur in severe cases of the novel coronavirus, Forbes reported.
ARDS can be triggered by over-creation of cytokines, proteins which tell the body to produce more inflammation, Forbes said.
The drug going to clinical trials, ARDS-003, would “dampen the cytokine release” and prevent development of ARDS, Tetra Bio-Pharma company CEO and chief regulatory officer Guy Chamberland, MD, said in a news release.
Consequences of ARDS include scarring of the lungs and organ injury caused by the decrease in blood to the tissue, the release said.
“The FDA repeatedly stated that they want clinical trials for COVID-19 to begin as soon as possible, as long as they meet regulatory requirements,” the news release said. “The medical community is in urgent need of drugs that can reduce the strength and duration of the severe inflammation. It is anticipated that this type of new drug would favorably impact health care and possibly reduce the negative health outcomes post infection.”
ARDS-003 works by binding to CB2 receptors, one of two main receptors in the endocannabinoid system which modulate inflammation and cytokine activity, Forbes said. CB2 receptors don’t bring on a psychoactive high.
Phase one clinical trials would begin enrolling participants in December to determine if the drug is safe, Chamberland said, according to Forbes.
If phase one is successful, phase two would test the drug on a larger group in the second quarter of 2021 to assess safety and tolerability for people who have COVID-19.
If phase two is successful, the company may seek emergency authorization through the FDA, Chamberland said. Phase three would start at the end of 2021.
Tetra Bio-Pharma says it has already contracted with Dalton Pharma Services to manufacture the active pharmaceutical ingredient (API), HU-308, and the finished drug product ARDS-003.
This article first appeared on Medscape.com.
Study confirms it’s possible to catch COVID-19 twice
Researchers in Hong Kong say they’ve confirmed that a person can be infected with COVID-19 twice.
The new proof comes from a 33-year-old man in Hong Kong who first caught COVID-19 in March. He was tested for the coronavirus after he developed a cough, sore throat, fever, and a headache for 3 days. He stayed in the hospital until he twice tested negative for the virus in mid-April.
On Aug. 15, the man returned to Hong Kong from a recent trip to Spain and the United Kingdom, areas that have recently seen a resurgence of COVID-19 cases. At the airport, he was screened for COVID-19 with a test that checks saliva for the virus. He tested positive, but this time, had no symptoms. He was taken to the hospital for monitoring. His viral load – the amount of virus he had in his body – went down over time, suggesting that his immune system was taking care of the intrusion on its own.
The special thing about his case is that each time he was hospitalized, doctors sequenced the genome of the virus that infected him. It was slightly different from one infection to the next, suggesting that the virus had mutated – or changed – in the 4 months between his infections. It also proves that it’s possible for this coronavirus to infect the same person twice.
Experts with the World Health Organization responded to the case at a news briefing.
“What we are learning about infection is that people do develop an immune response. What is not completely clear yet is how strong that immune response is and for how long that immune response lasts,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist with the World Health Organization in Geneva, Switzerland.
A study on the man’s case is being prepared for publication in the journal Clinical Infectious Diseases. Experts say the finding shouldn’t cause alarm, but it does have important implications for the development of herd immunity and efforts to come up with vaccines and treatments.
“This appears to be pretty clear-cut evidence of reinfection because of sequencing and isolation of two different viruses,” said Gregory Poland, MD, an expert on vaccine development and immunology at the Mayo Clinic in Rochester, Minn. “The big unknown is how often is this happening,” he said. More studies are needed to learn whether this was a rare case or something that is happening often.
Past experience guides present
Until we know more, Dr. Poland said, the possibility of getting COVID-19 twice shouldn’t make anyone worry.
This also happens with other kinds of coronaviruses – the ones that cause common colds. Those coronaviruses change slightly each year as they circle the globe, which allows them to keep spreading and causing their more run-of-the-mill kind of misery.
It also happens with seasonal flu. It is the reason people have to get vaccinated against the flu year after year, and why the flu vaccine has to change slightly each year in an effort to keep up with the ever-evolving influenza virus.
“We’ve been making flu vaccines for 80 years, and there are clinical trials happening as we speak to find new and better influenza vaccines,” Dr. Poland said.
There has been other evidence the virus that causes COVID-19 can change this way, too. Researchers at Howard Hughes Medical Center, at Rockefeller University in New York, recently used a key piece of the SARS-CoV-2 virus – the genetic instructions for its spike protein – to repeatedly infect human cells. Scientists watched as each new generation of the virus went on to infect a new batch of cells. Over time, as it copied itself, some of the copies changed their genes to allow them to survive after scientists attacked them with neutralizing antibodies. Those antibodies are among the main weapons used by the immune system to recognize and disable a virus.
Though that study is still a preprint, which means it hasn’t yet been reviewed by outside experts, the authors wrote that their findings suggest the virus can change in ways that help it evade our immune system. If true, they wrote in mid-July, it means reinfection is possible, especially in people who have a weak immune response to the virus the first time they encounter it.
Good news
That seems to be true in the case of the man from Hong Kong. When doctors tested his blood to look for antibodies to the virus, they didn’t find any. That could mean that he either had a weak immune response to the virus the first time around, or that the antibodies he made during his first infection diminished over time. But during his second infection, he quickly developed more antibodies, suggesting that the second infection acted a little bit like a booster to fire up his immune system. That’s probably the reason he didn’t have any symptoms the second time, too.
That’s good news, Dr. Poland said. It means our bodies can get better at fighting off the COVID-19 virus and that catching it once means the second time might not be so bad.
But the fact that the virus can change quickly this way does have some impact on the effort to come up with a vaccine that works well.
“I think a potential implication of this is that we will have to give booster doses. The question is how frequently,” Dr. Poland said. That will depend on how fast the virus is changing, and how often reinfection is happening in the real world.
“I’m a little surprised at 4½ months,” Dr. Poland said, referencing the time between the Hong Kong man’s infections. “I’m not surprised by, you know, I got infected last winter and I got infected again this winter,” he said.
It also suggests that immune-based therapies such as convalescent plasma and monoclonal antibodies may be of limited help over time, since the virus might be changing in ways that help it outsmart those treatments.
Convalescent plasma is essentially a concentrated dose of antibodies from people who have recovered from a COVID-19 infection. As the virus changes, the antibodies in that plasma may not work as well for future infections.
Drug companies have learned to harness the power of monoclonal antibodies as powerful treatments against cancer and other diseases. Monoclonal antibodies, which are mass-produced in a lab, mimic the body’s natural defenses against a pathogen. Just like the virus can become resistant to natural immunity, it can change in ways that help it outsmart lab-created treatments. Some drug companies that are developing monoclonal antibodies to fight COVID-19 have already prepared for that possibility by making antibody cocktails that are designed to disable the virus by locking onto it in different places, which may help prevent it from developing resistance to those therapies.
“We have a lot to learn,” Dr. Poland said. “Now that the proof of principle has been established, and I would say it has with this man, and with our knowledge of seasonal coronaviruses, we need to look more aggressively to define how often this occurs.”
A version of this article originally appeared on WebMD.com.
Researchers in Hong Kong say they’ve confirmed that a person can be infected with COVID-19 twice.
The new proof comes from a 33-year-old man in Hong Kong who first caught COVID-19 in March. He was tested for the coronavirus after he developed a cough, sore throat, fever, and a headache for 3 days. He stayed in the hospital until he twice tested negative for the virus in mid-April.
On Aug. 15, the man returned to Hong Kong from a recent trip to Spain and the United Kingdom, areas that have recently seen a resurgence of COVID-19 cases. At the airport, he was screened for COVID-19 with a test that checks saliva for the virus. He tested positive, but this time, had no symptoms. He was taken to the hospital for monitoring. His viral load – the amount of virus he had in his body – went down over time, suggesting that his immune system was taking care of the intrusion on its own.
The special thing about his case is that each time he was hospitalized, doctors sequenced the genome of the virus that infected him. It was slightly different from one infection to the next, suggesting that the virus had mutated – or changed – in the 4 months between his infections. It also proves that it’s possible for this coronavirus to infect the same person twice.
Experts with the World Health Organization responded to the case at a news briefing.
“What we are learning about infection is that people do develop an immune response. What is not completely clear yet is how strong that immune response is and for how long that immune response lasts,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist with the World Health Organization in Geneva, Switzerland.
A study on the man’s case is being prepared for publication in the journal Clinical Infectious Diseases. Experts say the finding shouldn’t cause alarm, but it does have important implications for the development of herd immunity and efforts to come up with vaccines and treatments.
“This appears to be pretty clear-cut evidence of reinfection because of sequencing and isolation of two different viruses,” said Gregory Poland, MD, an expert on vaccine development and immunology at the Mayo Clinic in Rochester, Minn. “The big unknown is how often is this happening,” he said. More studies are needed to learn whether this was a rare case or something that is happening often.
Past experience guides present
Until we know more, Dr. Poland said, the possibility of getting COVID-19 twice shouldn’t make anyone worry.
This also happens with other kinds of coronaviruses – the ones that cause common colds. Those coronaviruses change slightly each year as they circle the globe, which allows them to keep spreading and causing their more run-of-the-mill kind of misery.
It also happens with seasonal flu. It is the reason people have to get vaccinated against the flu year after year, and why the flu vaccine has to change slightly each year in an effort to keep up with the ever-evolving influenza virus.
“We’ve been making flu vaccines for 80 years, and there are clinical trials happening as we speak to find new and better influenza vaccines,” Dr. Poland said.
There has been other evidence the virus that causes COVID-19 can change this way, too. Researchers at Howard Hughes Medical Center, at Rockefeller University in New York, recently used a key piece of the SARS-CoV-2 virus – the genetic instructions for its spike protein – to repeatedly infect human cells. Scientists watched as each new generation of the virus went on to infect a new batch of cells. Over time, as it copied itself, some of the copies changed their genes to allow them to survive after scientists attacked them with neutralizing antibodies. Those antibodies are among the main weapons used by the immune system to recognize and disable a virus.
Though that study is still a preprint, which means it hasn’t yet been reviewed by outside experts, the authors wrote that their findings suggest the virus can change in ways that help it evade our immune system. If true, they wrote in mid-July, it means reinfection is possible, especially in people who have a weak immune response to the virus the first time they encounter it.
Good news
That seems to be true in the case of the man from Hong Kong. When doctors tested his blood to look for antibodies to the virus, they didn’t find any. That could mean that he either had a weak immune response to the virus the first time around, or that the antibodies he made during his first infection diminished over time. But during his second infection, he quickly developed more antibodies, suggesting that the second infection acted a little bit like a booster to fire up his immune system. That’s probably the reason he didn’t have any symptoms the second time, too.
That’s good news, Dr. Poland said. It means our bodies can get better at fighting off the COVID-19 virus and that catching it once means the second time might not be so bad.
But the fact that the virus can change quickly this way does have some impact on the effort to come up with a vaccine that works well.
“I think a potential implication of this is that we will have to give booster doses. The question is how frequently,” Dr. Poland said. That will depend on how fast the virus is changing, and how often reinfection is happening in the real world.
“I’m a little surprised at 4½ months,” Dr. Poland said, referencing the time between the Hong Kong man’s infections. “I’m not surprised by, you know, I got infected last winter and I got infected again this winter,” he said.
It also suggests that immune-based therapies such as convalescent plasma and monoclonal antibodies may be of limited help over time, since the virus might be changing in ways that help it outsmart those treatments.
Convalescent plasma is essentially a concentrated dose of antibodies from people who have recovered from a COVID-19 infection. As the virus changes, the antibodies in that plasma may not work as well for future infections.
Drug companies have learned to harness the power of monoclonal antibodies as powerful treatments against cancer and other diseases. Monoclonal antibodies, which are mass-produced in a lab, mimic the body’s natural defenses against a pathogen. Just like the virus can become resistant to natural immunity, it can change in ways that help it outsmart lab-created treatments. Some drug companies that are developing monoclonal antibodies to fight COVID-19 have already prepared for that possibility by making antibody cocktails that are designed to disable the virus by locking onto it in different places, which may help prevent it from developing resistance to those therapies.
“We have a lot to learn,” Dr. Poland said. “Now that the proof of principle has been established, and I would say it has with this man, and with our knowledge of seasonal coronaviruses, we need to look more aggressively to define how often this occurs.”
A version of this article originally appeared on WebMD.com.
Researchers in Hong Kong say they’ve confirmed that a person can be infected with COVID-19 twice.
The new proof comes from a 33-year-old man in Hong Kong who first caught COVID-19 in March. He was tested for the coronavirus after he developed a cough, sore throat, fever, and a headache for 3 days. He stayed in the hospital until he twice tested negative for the virus in mid-April.
On Aug. 15, the man returned to Hong Kong from a recent trip to Spain and the United Kingdom, areas that have recently seen a resurgence of COVID-19 cases. At the airport, he was screened for COVID-19 with a test that checks saliva for the virus. He tested positive, but this time, had no symptoms. He was taken to the hospital for monitoring. His viral load – the amount of virus he had in his body – went down over time, suggesting that his immune system was taking care of the intrusion on its own.
The special thing about his case is that each time he was hospitalized, doctors sequenced the genome of the virus that infected him. It was slightly different from one infection to the next, suggesting that the virus had mutated – or changed – in the 4 months between his infections. It also proves that it’s possible for this coronavirus to infect the same person twice.
Experts with the World Health Organization responded to the case at a news briefing.
“What we are learning about infection is that people do develop an immune response. What is not completely clear yet is how strong that immune response is and for how long that immune response lasts,” said Maria Van Kerkhove, PhD, an infectious disease epidemiologist with the World Health Organization in Geneva, Switzerland.
A study on the man’s case is being prepared for publication in the journal Clinical Infectious Diseases. Experts say the finding shouldn’t cause alarm, but it does have important implications for the development of herd immunity and efforts to come up with vaccines and treatments.
“This appears to be pretty clear-cut evidence of reinfection because of sequencing and isolation of two different viruses,” said Gregory Poland, MD, an expert on vaccine development and immunology at the Mayo Clinic in Rochester, Minn. “The big unknown is how often is this happening,” he said. More studies are needed to learn whether this was a rare case or something that is happening often.
Past experience guides present
Until we know more, Dr. Poland said, the possibility of getting COVID-19 twice shouldn’t make anyone worry.
This also happens with other kinds of coronaviruses – the ones that cause common colds. Those coronaviruses change slightly each year as they circle the globe, which allows them to keep spreading and causing their more run-of-the-mill kind of misery.
It also happens with seasonal flu. It is the reason people have to get vaccinated against the flu year after year, and why the flu vaccine has to change slightly each year in an effort to keep up with the ever-evolving influenza virus.
“We’ve been making flu vaccines for 80 years, and there are clinical trials happening as we speak to find new and better influenza vaccines,” Dr. Poland said.
There has been other evidence the virus that causes COVID-19 can change this way, too. Researchers at Howard Hughes Medical Center, at Rockefeller University in New York, recently used a key piece of the SARS-CoV-2 virus – the genetic instructions for its spike protein – to repeatedly infect human cells. Scientists watched as each new generation of the virus went on to infect a new batch of cells. Over time, as it copied itself, some of the copies changed their genes to allow them to survive after scientists attacked them with neutralizing antibodies. Those antibodies are among the main weapons used by the immune system to recognize and disable a virus.
Though that study is still a preprint, which means it hasn’t yet been reviewed by outside experts, the authors wrote that their findings suggest the virus can change in ways that help it evade our immune system. If true, they wrote in mid-July, it means reinfection is possible, especially in people who have a weak immune response to the virus the first time they encounter it.
Good news
That seems to be true in the case of the man from Hong Kong. When doctors tested his blood to look for antibodies to the virus, they didn’t find any. That could mean that he either had a weak immune response to the virus the first time around, or that the antibodies he made during his first infection diminished over time. But during his second infection, he quickly developed more antibodies, suggesting that the second infection acted a little bit like a booster to fire up his immune system. That’s probably the reason he didn’t have any symptoms the second time, too.
That’s good news, Dr. Poland said. It means our bodies can get better at fighting off the COVID-19 virus and that catching it once means the second time might not be so bad.
But the fact that the virus can change quickly this way does have some impact on the effort to come up with a vaccine that works well.
“I think a potential implication of this is that we will have to give booster doses. The question is how frequently,” Dr. Poland said. That will depend on how fast the virus is changing, and how often reinfection is happening in the real world.
“I’m a little surprised at 4½ months,” Dr. Poland said, referencing the time between the Hong Kong man’s infections. “I’m not surprised by, you know, I got infected last winter and I got infected again this winter,” he said.
It also suggests that immune-based therapies such as convalescent plasma and monoclonal antibodies may be of limited help over time, since the virus might be changing in ways that help it outsmart those treatments.
Convalescent plasma is essentially a concentrated dose of antibodies from people who have recovered from a COVID-19 infection. As the virus changes, the antibodies in that plasma may not work as well for future infections.
Drug companies have learned to harness the power of monoclonal antibodies as powerful treatments against cancer and other diseases. Monoclonal antibodies, which are mass-produced in a lab, mimic the body’s natural defenses against a pathogen. Just like the virus can become resistant to natural immunity, it can change in ways that help it outsmart lab-created treatments. Some drug companies that are developing monoclonal antibodies to fight COVID-19 have already prepared for that possibility by making antibody cocktails that are designed to disable the virus by locking onto it in different places, which may help prevent it from developing resistance to those therapies.
“We have a lot to learn,” Dr. Poland said. “Now that the proof of principle has been established, and I would say it has with this man, and with our knowledge of seasonal coronaviruses, we need to look more aggressively to define how often this occurs.”
A version of this article originally appeared on WebMD.com.
Research examines links between ‘long COVID’ and ME/CFS
Some patients who had COVID-19 continue to have symptoms weeks to months later, even after they no longer test positive for the virus. In two recent reports – one published in JAMA in July and another published in Morbidity and Mortality Weekly Report in August – chronic fatigue was listed as the top symptom among individuals still feeling unwell beyond 2 weeks after COVID-19 onset.
Although some of the reported persistent symptoms appear specific to SARS-CoV-2 – such as cough, chest pain, and dyspnea – others overlap with the diagnostic criteria for ME/CFS, which is defined by substantial, profound fatigue for at least 6 months, postexertional malaise, unrefreshing sleep, and one or both of orthostatic intolerance and/or cognitive impairment. Although the etiology of ME/CFS is unclear, the condition commonly arises following a viral illness.
At the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis August 21, the opening session was devoted to research documenting the extent to which COVID-19 survivors subsequently meet ME/CFS criteria, and to exploring underlying mechanisms.
“It offers a lot of opportunities for us to study potentially early ME/CFS and how it develops, but in addition, a lot of the research that has been done on ME/CFS may also provide answers for COVID-19,” IACFS/ME vice president Lily Chu, MD, said in an interview.
A hint from the SARS outbreak
This isn’t the first time researchers have seen a possible link between a coronavirus and ME/CFS, Harvey Moldofsky, MD, told attendees. To illustrate that point, Dr. Moldofsky, of the department of psychiatry (emeritus) at the University of Toronto, reviewed data from a previously published case-controlled study, which included 22 health care workers who had been infected in 2003 with SARS-CoV-1 and continued to report chronic fatigue, musculoskeletal pain, and disturbed and unrefreshing sleep with EEG-documented sleep disturbances 1-3 years following the illness. None had been able to return to work by 1 year.
“We’re looking at similar symptoms now” among survivors of COVID-19, Dr. Moldofsky said. “[T]he key issue is that we have no idea of its prevalence. … We need epidemiologic studies.”
Distinguishing ME/CFS from other post–COVID-19 symptoms
Not everyone who has persistent symptoms after COVID-19 will develop ME/CFS, and distinguishing between cases may be important.
Clinically, Dr. Chu said, one way to assess whether a patient with persistent COVID-19 symptoms might be progressing to ME/CFS is to ask him or her specifically about the level of fatigue following physical exertion and the timing of any fatigue. With ME/CFS, postexertional malaise often involves a dramatic exacerbation of symptoms such as fatigue, pain, and cognitive impairment a day or 2 after exertion rather than immediately following it. In contrast, shortness of breath during exertion isn’t typical of ME/CFS.
Objective measures of ME/CFS include low natural killer cell function (the test can be ordered from commercial labs but requires rapid transport of the blood sample), and autonomic dysfunction assessed by a tilt-table test.
While there is currently no cure for ME/CFS, diagnosing it allows for the patient to be taught “pacing” in which the person conserves his or her energy by balancing activity with rest. “That type of behavioral technique is valuable for everyone who suffers from a chronic disease with fatigue. It can help them be more functional,” Dr. Chu said.
If a patient appears to be exhibiting signs of ME/CFS, “don’t wait until they hit the 6-month mark to start helping them manage their symptoms,” she said. “Teaching pacing to COVID-19 patients who have a lot of fatigue isn’t going to harm them. As they get better they’re going to just naturally do more. But if they do have ME/CFS, [pacing] stresses their system less, since the data seem to be pointing to deficiencies in producing energy.”
Will COVID-19 unleash a new wave of ME/CFS patients?
Much of the session at the virtual meeting was devoted to ongoing studies. For example, Leonard Jason, PhD, of the Center for Community Research at DePaul University, Chicago, described a prospective study launched in 2014 that looked at risk factors for developing ME/CFS in college students who contracted infectious mononucleosis as a result of Epstein-Barr virus. Now, his team is also following students from the same cohort who develop COVID-19.
Because the study included collection of baseline biological samples, the results could help reveal predisposing factors associated with long-term illness from either virus.
Another project, funded by the Open Medicine Foundation, will follow patients who are discharged from the ICU following severe COVID-19 illness. Blood, urine, and cerebrospinal fluid will be collected from those with persistent symptoms at 6 months, along with questionnaire data. At 18-24 months, those who continue to report symptoms will undergo more intensive evaluation using genomics, metabolomics, and proteomics.
“We’re taking advantage of this horrible situation, hoping to understand how a serious viral infection might lead to ME/CFS,” said lead investigator Ronald Tompkins, MD, ScD, chief medical officer at the Open Medicine Foundation and a faculty member at Harvard Medical School, Boston. The results, he said, “might give us insight into potential drug targets or biomarkers useful for prevention and treatment strategies.”
Meanwhile, Sadie Whittaker, PhD, head of the Solve ME/CFS initiative, described her organization’s new plan to use their registry to prospectively track the impact of COVID-19 on people with ME/CFS.
She noted that they’ve also teamed up with “long-COVID” communities including Body Politic. “Our goal is to form a coalition to study together or at least harmonize data … and understand what’s going on through the power of bigger sample sizes,” Dr. Whittaker said.
None of the speakers disclosed relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Some patients who had COVID-19 continue to have symptoms weeks to months later, even after they no longer test positive for the virus. In two recent reports – one published in JAMA in July and another published in Morbidity and Mortality Weekly Report in August – chronic fatigue was listed as the top symptom among individuals still feeling unwell beyond 2 weeks after COVID-19 onset.
Although some of the reported persistent symptoms appear specific to SARS-CoV-2 – such as cough, chest pain, and dyspnea – others overlap with the diagnostic criteria for ME/CFS, which is defined by substantial, profound fatigue for at least 6 months, postexertional malaise, unrefreshing sleep, and one or both of orthostatic intolerance and/or cognitive impairment. Although the etiology of ME/CFS is unclear, the condition commonly arises following a viral illness.
At the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis August 21, the opening session was devoted to research documenting the extent to which COVID-19 survivors subsequently meet ME/CFS criteria, and to exploring underlying mechanisms.
“It offers a lot of opportunities for us to study potentially early ME/CFS and how it develops, but in addition, a lot of the research that has been done on ME/CFS may also provide answers for COVID-19,” IACFS/ME vice president Lily Chu, MD, said in an interview.
A hint from the SARS outbreak
This isn’t the first time researchers have seen a possible link between a coronavirus and ME/CFS, Harvey Moldofsky, MD, told attendees. To illustrate that point, Dr. Moldofsky, of the department of psychiatry (emeritus) at the University of Toronto, reviewed data from a previously published case-controlled study, which included 22 health care workers who had been infected in 2003 with SARS-CoV-1 and continued to report chronic fatigue, musculoskeletal pain, and disturbed and unrefreshing sleep with EEG-documented sleep disturbances 1-3 years following the illness. None had been able to return to work by 1 year.
“We’re looking at similar symptoms now” among survivors of COVID-19, Dr. Moldofsky said. “[T]he key issue is that we have no idea of its prevalence. … We need epidemiologic studies.”
Distinguishing ME/CFS from other post–COVID-19 symptoms
Not everyone who has persistent symptoms after COVID-19 will develop ME/CFS, and distinguishing between cases may be important.
Clinically, Dr. Chu said, one way to assess whether a patient with persistent COVID-19 symptoms might be progressing to ME/CFS is to ask him or her specifically about the level of fatigue following physical exertion and the timing of any fatigue. With ME/CFS, postexertional malaise often involves a dramatic exacerbation of symptoms such as fatigue, pain, and cognitive impairment a day or 2 after exertion rather than immediately following it. In contrast, shortness of breath during exertion isn’t typical of ME/CFS.
Objective measures of ME/CFS include low natural killer cell function (the test can be ordered from commercial labs but requires rapid transport of the blood sample), and autonomic dysfunction assessed by a tilt-table test.
While there is currently no cure for ME/CFS, diagnosing it allows for the patient to be taught “pacing” in which the person conserves his or her energy by balancing activity with rest. “That type of behavioral technique is valuable for everyone who suffers from a chronic disease with fatigue. It can help them be more functional,” Dr. Chu said.
If a patient appears to be exhibiting signs of ME/CFS, “don’t wait until they hit the 6-month mark to start helping them manage their symptoms,” she said. “Teaching pacing to COVID-19 patients who have a lot of fatigue isn’t going to harm them. As they get better they’re going to just naturally do more. But if they do have ME/CFS, [pacing] stresses their system less, since the data seem to be pointing to deficiencies in producing energy.”
Will COVID-19 unleash a new wave of ME/CFS patients?
Much of the session at the virtual meeting was devoted to ongoing studies. For example, Leonard Jason, PhD, of the Center for Community Research at DePaul University, Chicago, described a prospective study launched in 2014 that looked at risk factors for developing ME/CFS in college students who contracted infectious mononucleosis as a result of Epstein-Barr virus. Now, his team is also following students from the same cohort who develop COVID-19.
Because the study included collection of baseline biological samples, the results could help reveal predisposing factors associated with long-term illness from either virus.
Another project, funded by the Open Medicine Foundation, will follow patients who are discharged from the ICU following severe COVID-19 illness. Blood, urine, and cerebrospinal fluid will be collected from those with persistent symptoms at 6 months, along with questionnaire data. At 18-24 months, those who continue to report symptoms will undergo more intensive evaluation using genomics, metabolomics, and proteomics.
“We’re taking advantage of this horrible situation, hoping to understand how a serious viral infection might lead to ME/CFS,” said lead investigator Ronald Tompkins, MD, ScD, chief medical officer at the Open Medicine Foundation and a faculty member at Harvard Medical School, Boston. The results, he said, “might give us insight into potential drug targets or biomarkers useful for prevention and treatment strategies.”
Meanwhile, Sadie Whittaker, PhD, head of the Solve ME/CFS initiative, described her organization’s new plan to use their registry to prospectively track the impact of COVID-19 on people with ME/CFS.
She noted that they’ve also teamed up with “long-COVID” communities including Body Politic. “Our goal is to form a coalition to study together or at least harmonize data … and understand what’s going on through the power of bigger sample sizes,” Dr. Whittaker said.
None of the speakers disclosed relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Some patients who had COVID-19 continue to have symptoms weeks to months later, even after they no longer test positive for the virus. In two recent reports – one published in JAMA in July and another published in Morbidity and Mortality Weekly Report in August – chronic fatigue was listed as the top symptom among individuals still feeling unwell beyond 2 weeks after COVID-19 onset.
Although some of the reported persistent symptoms appear specific to SARS-CoV-2 – such as cough, chest pain, and dyspnea – others overlap with the diagnostic criteria for ME/CFS, which is defined by substantial, profound fatigue for at least 6 months, postexertional malaise, unrefreshing sleep, and one or both of orthostatic intolerance and/or cognitive impairment. Although the etiology of ME/CFS is unclear, the condition commonly arises following a viral illness.
At the virtual meeting of the International Association for Chronic Fatigue Syndrome/Myalgic Encephalomyelitis August 21, the opening session was devoted to research documenting the extent to which COVID-19 survivors subsequently meet ME/CFS criteria, and to exploring underlying mechanisms.
“It offers a lot of opportunities for us to study potentially early ME/CFS and how it develops, but in addition, a lot of the research that has been done on ME/CFS may also provide answers for COVID-19,” IACFS/ME vice president Lily Chu, MD, said in an interview.
A hint from the SARS outbreak
This isn’t the first time researchers have seen a possible link between a coronavirus and ME/CFS, Harvey Moldofsky, MD, told attendees. To illustrate that point, Dr. Moldofsky, of the department of psychiatry (emeritus) at the University of Toronto, reviewed data from a previously published case-controlled study, which included 22 health care workers who had been infected in 2003 with SARS-CoV-1 and continued to report chronic fatigue, musculoskeletal pain, and disturbed and unrefreshing sleep with EEG-documented sleep disturbances 1-3 years following the illness. None had been able to return to work by 1 year.
“We’re looking at similar symptoms now” among survivors of COVID-19, Dr. Moldofsky said. “[T]he key issue is that we have no idea of its prevalence. … We need epidemiologic studies.”
Distinguishing ME/CFS from other post–COVID-19 symptoms
Not everyone who has persistent symptoms after COVID-19 will develop ME/CFS, and distinguishing between cases may be important.
Clinically, Dr. Chu said, one way to assess whether a patient with persistent COVID-19 symptoms might be progressing to ME/CFS is to ask him or her specifically about the level of fatigue following physical exertion and the timing of any fatigue. With ME/CFS, postexertional malaise often involves a dramatic exacerbation of symptoms such as fatigue, pain, and cognitive impairment a day or 2 after exertion rather than immediately following it. In contrast, shortness of breath during exertion isn’t typical of ME/CFS.
Objective measures of ME/CFS include low natural killer cell function (the test can be ordered from commercial labs but requires rapid transport of the blood sample), and autonomic dysfunction assessed by a tilt-table test.
While there is currently no cure for ME/CFS, diagnosing it allows for the patient to be taught “pacing” in which the person conserves his or her energy by balancing activity with rest. “That type of behavioral technique is valuable for everyone who suffers from a chronic disease with fatigue. It can help them be more functional,” Dr. Chu said.
If a patient appears to be exhibiting signs of ME/CFS, “don’t wait until they hit the 6-month mark to start helping them manage their symptoms,” she said. “Teaching pacing to COVID-19 patients who have a lot of fatigue isn’t going to harm them. As they get better they’re going to just naturally do more. But if they do have ME/CFS, [pacing] stresses their system less, since the data seem to be pointing to deficiencies in producing energy.”
Will COVID-19 unleash a new wave of ME/CFS patients?
Much of the session at the virtual meeting was devoted to ongoing studies. For example, Leonard Jason, PhD, of the Center for Community Research at DePaul University, Chicago, described a prospective study launched in 2014 that looked at risk factors for developing ME/CFS in college students who contracted infectious mononucleosis as a result of Epstein-Barr virus. Now, his team is also following students from the same cohort who develop COVID-19.
Because the study included collection of baseline biological samples, the results could help reveal predisposing factors associated with long-term illness from either virus.
Another project, funded by the Open Medicine Foundation, will follow patients who are discharged from the ICU following severe COVID-19 illness. Blood, urine, and cerebrospinal fluid will be collected from those with persistent symptoms at 6 months, along with questionnaire data. At 18-24 months, those who continue to report symptoms will undergo more intensive evaluation using genomics, metabolomics, and proteomics.
“We’re taking advantage of this horrible situation, hoping to understand how a serious viral infection might lead to ME/CFS,” said lead investigator Ronald Tompkins, MD, ScD, chief medical officer at the Open Medicine Foundation and a faculty member at Harvard Medical School, Boston. The results, he said, “might give us insight into potential drug targets or biomarkers useful for prevention and treatment strategies.”
Meanwhile, Sadie Whittaker, PhD, head of the Solve ME/CFS initiative, described her organization’s new plan to use their registry to prospectively track the impact of COVID-19 on people with ME/CFS.
She noted that they’ve also teamed up with “long-COVID” communities including Body Politic. “Our goal is to form a coalition to study together or at least harmonize data … and understand what’s going on through the power of bigger sample sizes,” Dr. Whittaker said.
None of the speakers disclosed relevant financial relationships.
A version of this article originally appeared on Medscape.com.
FDA authorizes convalescent plasma for COVID-19
Convalescent plasma contains antibodies from the blood of recovered COVID-19 patients, which can be used to treat people with severe infections. Convalescent plasma has been used to treat patients for other infectious diseases. The authorization allows the plasma to be distributed in the United States and administered by health care providers.
“COVID-19 convalescent plasma is safe and shows promising efficacy,” Stephen Hahn, MD, commissioner of the FDA, said during a press briefing with President Donald Trump.
In April, the FDA approved a program to test convalescent plasma in COVID-19 patients at the Mayo Clinic, followed by other institutions. More than 90,000 patients have enrolled in the program, and 70,000 have received the treatment, Dr. Hahn said.
The data indicate that the plasma can reduce mortality in patients by 35%, particularly if patients are treated within 3 days of being diagnosed. Those who have benefited the most were under age 80 and not on artificial respiration, Alex Azar, the secretary for the Department of Health & Human Services, said during the briefing.
“We dream, in drug development, of something like a 35% mortality reduction,” he said.
But top scientists pushed back against the announcement.
Eric Topol, MD, director of the Scripps Research Translational Institute, professor of molecular medicine, and executive vice president of Scripps Research, said the data the FDA are relying on did not come from the rigorous randomized, double-blind placebo trials that best determine if a treatment is successful.
Still, convalescent plasma is “one more tool added to the arsenal” of combating COVID-19, Mr. Azar said. The FDA will continue to study convalescent plasma as a COVID-19 treatment, Dr. Hahn added.
“We’re waiting for more data. We’re going to continue to gather data,” Dr. Hahn said during the briefing, but the current results meet FDA criteria for issuing an emergency use authorization.
Convalescent plasma “may be effective in lessening the severity or shortening the length of COVID-19 illness in some hospitalized patients,” according to the FDA announcement. Potential side effects include allergic reactions, transfusion-transmitted infections, and transfusion-associated lung injury.
“We’ve seen a great deal of demand for this from doctors around the country,” Dr. Hahn said during the briefing. “The EUA … allows us to continue that and meet that demand.”
Dr. Topol, however, said it appears Trump and the FDA are playing politics with science.
“There’s no evidence to support any survival benefit,” Dr. Topol said on Twitter. “Two days ago [the] FDA’s website stated there was no evidence for an EUA.”
The American Red Cross and other blood centers put out a national call for blood donors in July, especially for patients who have recovered from COVID-19. Mr. Azar and Dr. Hahn emphasized the need for blood donors during the press briefing.
“If you donate plasma, you could save a life,” Mr. Azar said.
The study has not been peer reviewed and did not include a placebo group for comparison, STAT reported.
Last week several health officials warned that the scientific data were too weak to warrant an emergency authorization, the New York Times reported.
A version of this originally appeared on WebMD.com.
Convalescent plasma contains antibodies from the blood of recovered COVID-19 patients, which can be used to treat people with severe infections. Convalescent plasma has been used to treat patients for other infectious diseases. The authorization allows the plasma to be distributed in the United States and administered by health care providers.
“COVID-19 convalescent plasma is safe and shows promising efficacy,” Stephen Hahn, MD, commissioner of the FDA, said during a press briefing with President Donald Trump.
In April, the FDA approved a program to test convalescent plasma in COVID-19 patients at the Mayo Clinic, followed by other institutions. More than 90,000 patients have enrolled in the program, and 70,000 have received the treatment, Dr. Hahn said.
The data indicate that the plasma can reduce mortality in patients by 35%, particularly if patients are treated within 3 days of being diagnosed. Those who have benefited the most were under age 80 and not on artificial respiration, Alex Azar, the secretary for the Department of Health & Human Services, said during the briefing.
“We dream, in drug development, of something like a 35% mortality reduction,” he said.
But top scientists pushed back against the announcement.
Eric Topol, MD, director of the Scripps Research Translational Institute, professor of molecular medicine, and executive vice president of Scripps Research, said the data the FDA are relying on did not come from the rigorous randomized, double-blind placebo trials that best determine if a treatment is successful.
Still, convalescent plasma is “one more tool added to the arsenal” of combating COVID-19, Mr. Azar said. The FDA will continue to study convalescent plasma as a COVID-19 treatment, Dr. Hahn added.
“We’re waiting for more data. We’re going to continue to gather data,” Dr. Hahn said during the briefing, but the current results meet FDA criteria for issuing an emergency use authorization.
Convalescent plasma “may be effective in lessening the severity or shortening the length of COVID-19 illness in some hospitalized patients,” according to the FDA announcement. Potential side effects include allergic reactions, transfusion-transmitted infections, and transfusion-associated lung injury.
“We’ve seen a great deal of demand for this from doctors around the country,” Dr. Hahn said during the briefing. “The EUA … allows us to continue that and meet that demand.”
Dr. Topol, however, said it appears Trump and the FDA are playing politics with science.
“There’s no evidence to support any survival benefit,” Dr. Topol said on Twitter. “Two days ago [the] FDA’s website stated there was no evidence for an EUA.”
The American Red Cross and other blood centers put out a national call for blood donors in July, especially for patients who have recovered from COVID-19. Mr. Azar and Dr. Hahn emphasized the need for blood donors during the press briefing.
“If you donate plasma, you could save a life,” Mr. Azar said.
The study has not been peer reviewed and did not include a placebo group for comparison, STAT reported.
Last week several health officials warned that the scientific data were too weak to warrant an emergency authorization, the New York Times reported.
A version of this originally appeared on WebMD.com.
Convalescent plasma contains antibodies from the blood of recovered COVID-19 patients, which can be used to treat people with severe infections. Convalescent plasma has been used to treat patients for other infectious diseases. The authorization allows the plasma to be distributed in the United States and administered by health care providers.
“COVID-19 convalescent plasma is safe and shows promising efficacy,” Stephen Hahn, MD, commissioner of the FDA, said during a press briefing with President Donald Trump.
In April, the FDA approved a program to test convalescent plasma in COVID-19 patients at the Mayo Clinic, followed by other institutions. More than 90,000 patients have enrolled in the program, and 70,000 have received the treatment, Dr. Hahn said.
The data indicate that the plasma can reduce mortality in patients by 35%, particularly if patients are treated within 3 days of being diagnosed. Those who have benefited the most were under age 80 and not on artificial respiration, Alex Azar, the secretary for the Department of Health & Human Services, said during the briefing.
“We dream, in drug development, of something like a 35% mortality reduction,” he said.
But top scientists pushed back against the announcement.
Eric Topol, MD, director of the Scripps Research Translational Institute, professor of molecular medicine, and executive vice president of Scripps Research, said the data the FDA are relying on did not come from the rigorous randomized, double-blind placebo trials that best determine if a treatment is successful.
Still, convalescent plasma is “one more tool added to the arsenal” of combating COVID-19, Mr. Azar said. The FDA will continue to study convalescent plasma as a COVID-19 treatment, Dr. Hahn added.
“We’re waiting for more data. We’re going to continue to gather data,” Dr. Hahn said during the briefing, but the current results meet FDA criteria for issuing an emergency use authorization.
Convalescent plasma “may be effective in lessening the severity or shortening the length of COVID-19 illness in some hospitalized patients,” according to the FDA announcement. Potential side effects include allergic reactions, transfusion-transmitted infections, and transfusion-associated lung injury.
“We’ve seen a great deal of demand for this from doctors around the country,” Dr. Hahn said during the briefing. “The EUA … allows us to continue that and meet that demand.”
Dr. Topol, however, said it appears Trump and the FDA are playing politics with science.
“There’s no evidence to support any survival benefit,” Dr. Topol said on Twitter. “Two days ago [the] FDA’s website stated there was no evidence for an EUA.”
The American Red Cross and other blood centers put out a national call for blood donors in July, especially for patients who have recovered from COVID-19. Mr. Azar and Dr. Hahn emphasized the need for blood donors during the press briefing.
“If you donate plasma, you could save a life,” Mr. Azar said.
The study has not been peer reviewed and did not include a placebo group for comparison, STAT reported.
Last week several health officials warned that the scientific data were too weak to warrant an emergency authorization, the New York Times reported.
A version of this originally appeared on WebMD.com.
Aggression is influenced by genetic, environmental factors
Aggression in individuals is influenced by genetic and environmental factors, but can be reduced with treatment, according to Emil F. Coccaro, MD.
“It actually is a complex triad of emotion, cognition, and behavior. The emotion is anger, the cognition is hostility, and the behavior is aggression. And they sort of go in that order,” Dr. Coccaro said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
Although aggression can be thought of in a numerous ways, premeditated and impulsive aggression are most relevant to behavioral studies in psychiatry, Dr. Coccaro explained. Premeditated aggression is goal oriented, while impulsive aggression comes from frustration or a response to a threat. Impulsive aggression is “typically social or frustrative in nature, and studies that we’ve done that show that individuals move toward a threat while nonaggressives move away it,” he said. Both types of aggression can be seen in the same individuals at different times.
Aggression also can be considered using a threshold model. Calm individuals, for example, might have a low baseline of aggression and a high threshold before they act out. An aggressive person, on the other hand, has a lower threshold and a higher baseline level. “Their delta to get to the point where they’re going to explode is much shorter, much lower than it is in someone who is healthy,” Dr. Coccaro said.
“What we think is that the threshold to explode is probably regulated by various neurobiological features. The baseline state of aggression also may be related to baseline neurobiological features, but also what’s going on in the environment, because the neurobiological features that send someone to exploding aggression are there all the time,” he explained.
Individuals with secondary aggression are likely to have an underlying condition, such as a primary disease of the brain, systemic or metabolic disorder, or a psychiatric disorder such as schizophrenia. “If someone’s schizophrenic and they’ve got voices telling them to hurt somebody, or delusions that someone’s going to hurt them, that’s not primary aggression, that’s secondary to the psychosis,” Dr. Coccaro noted.
An individual with primary aggression is likely to have intermittent explosive disorder (IED). IED is not a new diagnosis and has been listed in the DSM since the DSM-I as “passive-aggressive personality.” It was relisted in the DSM-II as “explosive personality,” then changed to IED in the DSM-3 as a diagnosis of exclusion that was poorly operationalized, according to Dr. Coccaro. The criteria for IED under the DSM-III did not define the number of recurrent outbursts needed, what they looked like, the time frame, and excluded people who were generally impulsive.
“That’s not really what these people look like and it’s not what impulsive aggression looks like,” he said. Although the DSM-IV removed the exclusion criteria for general impulsivity and aggression, “it was still purely operational.”
The DSM-5 criteria define IED as “verbal and physical aggression without destruction or assault, twice equally on average for 3 months, or three or more episodes of physical destruction/assault over a 1-year period. These individuals have outbursts “grossly out of proportion to provocation,” the aggression is generally impulsive, and it causes stress and impairment with an age of onset at older than 6 years.
“It’s not better accounted for a whole variety of things, but we actually made some of those exclusion criteria a little less stringent,” compared with criteria in the DSM-IV, Dr. Coccaro said. “That’s because it turns out that it doesn’t really matter much of the time what the comorbidity is. If you have this aggressiveness in the absence of those other conditions, it’s IED.”
According to a reanalysis of the National Comorbidity Survey, 11.7% of adolescents displayed aggressiveness within the last year and 17.3% over a lifetime, compared with 5.1% of adults within the last year and 8.0% within a lifetime. Under DSM-5 criteria, 6.4% of adolescents within the last year and 8.9% over a lifetime currently have IED, compared with 2.6% of adults within the last year and 4.0% over a lifetime, but “could go as high” as the percentage of individuals diagnosed with aggressiveness, Dr. Coccaro noted.
“People who are not called IED many times are not called IED because we didn’t have all the information we needed to actually make the diagnosis,” he said.
Individuals with DSM-5 IED can have as many as 30 episodes in 1 year, compared with those who are nonaggressive and are also more likely to damage property. “These are the big episodes, not simply the episodes where people are getting irritable and snapping at people. These are the big ones, where they’re really destroying objects and pushing or hitting people,” Dr. Coccaro said. About one-fourth of individuals with IED hurt victims badly enough that they require medical attention, one-fifth exhibit aggression toward a partner, and one-fourth receive aggression from their own partner.
In terms of comorbidity with other psychiatric disorders, “IEDs don’t have more comorbidity in general than other disorders,” Dr. Coccaro noted. Personality disorders such as paranoid, antisocial, borderline narcissistic, and obsessive-compulsive disorders are more common in individuals with IED. Aggression in these people present differently depending on the personality disorder. “Someone who’s paranoid might blow up at you if you get in their face. For an antisocial, they’ll blow up at you if you’re preventing them from doing what they want to do. Borderlines, you reject them or you abandon them, they’re going to blow up. Narcissists will blow up when you reject. OCD will also blow up when you mess around with their sense of order,” Dr. Coccaro said.
Genetics also play a role in whether a person may have IED. These percentages were consistent, regardless of whether the individual had a comorbid condition, history of alcohol or drug use, or history of suicide, he said. Other factors that influence likelihood of IED are environment, behaviors such as smoking, and conditions such as traumatic brain injury. Experiencing aggression as a child is another factor.
“IED is the categorical expression of impulsive aggression, and it’s far more common than once thought,” Dr. Coccaro said. “And IED is totally unrecognized in its role in societal violence.”
Treatment can suppress, but not cure aggression
Medications used to treat aggression and impulsive aggression include lithium, SSRIs, mood stabilizers, neuroleptics, and beta-blockers. However, the treatments are not a “magic bullet,” Dr. Coccaro noted. “The meds tend to suppress aggressiveness, but not cure it.”
Timing of treatment is also a factor for medication. In studies of patients taking lithium for aggression, for example, “when they gave the drug to people who liked being aggressive, they didn’t like being on these drugs because it made them feel unprotected. It just was at odds with who they thought they were,” Dr. Coccaro said. “The people who took the drug and did well and really liked being on the drug with people who didn’t like that they were aggressive.”
Neurorehabilitation and cognitive-behavioral therapy specific to aggression, called cognitive relaxation and coping skills therapy, are nonpsychotropic approaches to treating aggression. “These therapeutic approaches are working not only to reduce progression, but also to reduce the social information processing problems that aggressive individuals have,” Dr. Coccaro said.
Another approach, known as interpretation bias training, teaches individuals with aggression to judge slightly angry-looking photos of people as not being angry. After 7-14 days of training, aggressive behavior in adolescents has been shown to be reduced. The changes were also visible on functional MRI.
“What they found was that when you treated them, the change in the amygdala went down when you looked at the angry faces and in the left lateral, post training, they became happier,” Dr. Coccaro said.
Global Academy and this news organization are owned by the same parent company. Dr. Coccaro reported serving as a consultant for Avanir, Azevan, and Bracket. He also reported receiving research grants from the National Institute of Mental Health, the National Institute on Alcohol Abuse and Alcoholism, and the Pritzker Pucker Family Foundation.
Aggression in individuals is influenced by genetic and environmental factors, but can be reduced with treatment, according to Emil F. Coccaro, MD.
“It actually is a complex triad of emotion, cognition, and behavior. The emotion is anger, the cognition is hostility, and the behavior is aggression. And they sort of go in that order,” Dr. Coccaro said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
Although aggression can be thought of in a numerous ways, premeditated and impulsive aggression are most relevant to behavioral studies in psychiatry, Dr. Coccaro explained. Premeditated aggression is goal oriented, while impulsive aggression comes from frustration or a response to a threat. Impulsive aggression is “typically social or frustrative in nature, and studies that we’ve done that show that individuals move toward a threat while nonaggressives move away it,” he said. Both types of aggression can be seen in the same individuals at different times.
Aggression also can be considered using a threshold model. Calm individuals, for example, might have a low baseline of aggression and a high threshold before they act out. An aggressive person, on the other hand, has a lower threshold and a higher baseline level. “Their delta to get to the point where they’re going to explode is much shorter, much lower than it is in someone who is healthy,” Dr. Coccaro said.
“What we think is that the threshold to explode is probably regulated by various neurobiological features. The baseline state of aggression also may be related to baseline neurobiological features, but also what’s going on in the environment, because the neurobiological features that send someone to exploding aggression are there all the time,” he explained.
Individuals with secondary aggression are likely to have an underlying condition, such as a primary disease of the brain, systemic or metabolic disorder, or a psychiatric disorder such as schizophrenia. “If someone’s schizophrenic and they’ve got voices telling them to hurt somebody, or delusions that someone’s going to hurt them, that’s not primary aggression, that’s secondary to the psychosis,” Dr. Coccaro noted.
An individual with primary aggression is likely to have intermittent explosive disorder (IED). IED is not a new diagnosis and has been listed in the DSM since the DSM-I as “passive-aggressive personality.” It was relisted in the DSM-II as “explosive personality,” then changed to IED in the DSM-3 as a diagnosis of exclusion that was poorly operationalized, according to Dr. Coccaro. The criteria for IED under the DSM-III did not define the number of recurrent outbursts needed, what they looked like, the time frame, and excluded people who were generally impulsive.
“That’s not really what these people look like and it’s not what impulsive aggression looks like,” he said. Although the DSM-IV removed the exclusion criteria for general impulsivity and aggression, “it was still purely operational.”
The DSM-5 criteria define IED as “verbal and physical aggression without destruction or assault, twice equally on average for 3 months, or three or more episodes of physical destruction/assault over a 1-year period. These individuals have outbursts “grossly out of proportion to provocation,” the aggression is generally impulsive, and it causes stress and impairment with an age of onset at older than 6 years.
“It’s not better accounted for a whole variety of things, but we actually made some of those exclusion criteria a little less stringent,” compared with criteria in the DSM-IV, Dr. Coccaro said. “That’s because it turns out that it doesn’t really matter much of the time what the comorbidity is. If you have this aggressiveness in the absence of those other conditions, it’s IED.”
According to a reanalysis of the National Comorbidity Survey, 11.7% of adolescents displayed aggressiveness within the last year and 17.3% over a lifetime, compared with 5.1% of adults within the last year and 8.0% within a lifetime. Under DSM-5 criteria, 6.4% of adolescents within the last year and 8.9% over a lifetime currently have IED, compared with 2.6% of adults within the last year and 4.0% over a lifetime, but “could go as high” as the percentage of individuals diagnosed with aggressiveness, Dr. Coccaro noted.
“People who are not called IED many times are not called IED because we didn’t have all the information we needed to actually make the diagnosis,” he said.
Individuals with DSM-5 IED can have as many as 30 episodes in 1 year, compared with those who are nonaggressive and are also more likely to damage property. “These are the big episodes, not simply the episodes where people are getting irritable and snapping at people. These are the big ones, where they’re really destroying objects and pushing or hitting people,” Dr. Coccaro said. About one-fourth of individuals with IED hurt victims badly enough that they require medical attention, one-fifth exhibit aggression toward a partner, and one-fourth receive aggression from their own partner.
In terms of comorbidity with other psychiatric disorders, “IEDs don’t have more comorbidity in general than other disorders,” Dr. Coccaro noted. Personality disorders such as paranoid, antisocial, borderline narcissistic, and obsessive-compulsive disorders are more common in individuals with IED. Aggression in these people present differently depending on the personality disorder. “Someone who’s paranoid might blow up at you if you get in their face. For an antisocial, they’ll blow up at you if you’re preventing them from doing what they want to do. Borderlines, you reject them or you abandon them, they’re going to blow up. Narcissists will blow up when you reject. OCD will also blow up when you mess around with their sense of order,” Dr. Coccaro said.
Genetics also play a role in whether a person may have IED. These percentages were consistent, regardless of whether the individual had a comorbid condition, history of alcohol or drug use, or history of suicide, he said. Other factors that influence likelihood of IED are environment, behaviors such as smoking, and conditions such as traumatic brain injury. Experiencing aggression as a child is another factor.
“IED is the categorical expression of impulsive aggression, and it’s far more common than once thought,” Dr. Coccaro said. “And IED is totally unrecognized in its role in societal violence.”
Treatment can suppress, but not cure aggression
Medications used to treat aggression and impulsive aggression include lithium, SSRIs, mood stabilizers, neuroleptics, and beta-blockers. However, the treatments are not a “magic bullet,” Dr. Coccaro noted. “The meds tend to suppress aggressiveness, but not cure it.”
Timing of treatment is also a factor for medication. In studies of patients taking lithium for aggression, for example, “when they gave the drug to people who liked being aggressive, they didn’t like being on these drugs because it made them feel unprotected. It just was at odds with who they thought they were,” Dr. Coccaro said. “The people who took the drug and did well and really liked being on the drug with people who didn’t like that they were aggressive.”
Neurorehabilitation and cognitive-behavioral therapy specific to aggression, called cognitive relaxation and coping skills therapy, are nonpsychotropic approaches to treating aggression. “These therapeutic approaches are working not only to reduce progression, but also to reduce the social information processing problems that aggressive individuals have,” Dr. Coccaro said.
Another approach, known as interpretation bias training, teaches individuals with aggression to judge slightly angry-looking photos of people as not being angry. After 7-14 days of training, aggressive behavior in adolescents has been shown to be reduced. The changes were also visible on functional MRI.
“What they found was that when you treated them, the change in the amygdala went down when you looked at the angry faces and in the left lateral, post training, they became happier,” Dr. Coccaro said.
Global Academy and this news organization are owned by the same parent company. Dr. Coccaro reported serving as a consultant for Avanir, Azevan, and Bracket. He also reported receiving research grants from the National Institute of Mental Health, the National Institute on Alcohol Abuse and Alcoholism, and the Pritzker Pucker Family Foundation.
Aggression in individuals is influenced by genetic and environmental factors, but can be reduced with treatment, according to Emil F. Coccaro, MD.
“It actually is a complex triad of emotion, cognition, and behavior. The emotion is anger, the cognition is hostility, and the behavior is aggression. And they sort of go in that order,” Dr. Coccaro said at Focus on Neuropsychiatry presented by Current Psychiatry and the American Academy of Clinical Psychiatrists.
Although aggression can be thought of in a numerous ways, premeditated and impulsive aggression are most relevant to behavioral studies in psychiatry, Dr. Coccaro explained. Premeditated aggression is goal oriented, while impulsive aggression comes from frustration or a response to a threat. Impulsive aggression is “typically social or frustrative in nature, and studies that we’ve done that show that individuals move toward a threat while nonaggressives move away it,” he said. Both types of aggression can be seen in the same individuals at different times.
Aggression also can be considered using a threshold model. Calm individuals, for example, might have a low baseline of aggression and a high threshold before they act out. An aggressive person, on the other hand, has a lower threshold and a higher baseline level. “Their delta to get to the point where they’re going to explode is much shorter, much lower than it is in someone who is healthy,” Dr. Coccaro said.
“What we think is that the threshold to explode is probably regulated by various neurobiological features. The baseline state of aggression also may be related to baseline neurobiological features, but also what’s going on in the environment, because the neurobiological features that send someone to exploding aggression are there all the time,” he explained.
Individuals with secondary aggression are likely to have an underlying condition, such as a primary disease of the brain, systemic or metabolic disorder, or a psychiatric disorder such as schizophrenia. “If someone’s schizophrenic and they’ve got voices telling them to hurt somebody, or delusions that someone’s going to hurt them, that’s not primary aggression, that’s secondary to the psychosis,” Dr. Coccaro noted.
An individual with primary aggression is likely to have intermittent explosive disorder (IED). IED is not a new diagnosis and has been listed in the DSM since the DSM-I as “passive-aggressive personality.” It was relisted in the DSM-II as “explosive personality,” then changed to IED in the DSM-3 as a diagnosis of exclusion that was poorly operationalized, according to Dr. Coccaro. The criteria for IED under the DSM-III did not define the number of recurrent outbursts needed, what they looked like, the time frame, and excluded people who were generally impulsive.
“That’s not really what these people look like and it’s not what impulsive aggression looks like,” he said. Although the DSM-IV removed the exclusion criteria for general impulsivity and aggression, “it was still purely operational.”
The DSM-5 criteria define IED as “verbal and physical aggression without destruction or assault, twice equally on average for 3 months, or three or more episodes of physical destruction/assault over a 1-year period. These individuals have outbursts “grossly out of proportion to provocation,” the aggression is generally impulsive, and it causes stress and impairment with an age of onset at older than 6 years.
“It’s not better accounted for a whole variety of things, but we actually made some of those exclusion criteria a little less stringent,” compared with criteria in the DSM-IV, Dr. Coccaro said. “That’s because it turns out that it doesn’t really matter much of the time what the comorbidity is. If you have this aggressiveness in the absence of those other conditions, it’s IED.”
According to a reanalysis of the National Comorbidity Survey, 11.7% of adolescents displayed aggressiveness within the last year and 17.3% over a lifetime, compared with 5.1% of adults within the last year and 8.0% within a lifetime. Under DSM-5 criteria, 6.4% of adolescents within the last year and 8.9% over a lifetime currently have IED, compared with 2.6% of adults within the last year and 4.0% over a lifetime, but “could go as high” as the percentage of individuals diagnosed with aggressiveness, Dr. Coccaro noted.
“People who are not called IED many times are not called IED because we didn’t have all the information we needed to actually make the diagnosis,” he said.
Individuals with DSM-5 IED can have as many as 30 episodes in 1 year, compared with those who are nonaggressive and are also more likely to damage property. “These are the big episodes, not simply the episodes where people are getting irritable and snapping at people. These are the big ones, where they’re really destroying objects and pushing or hitting people,” Dr. Coccaro said. About one-fourth of individuals with IED hurt victims badly enough that they require medical attention, one-fifth exhibit aggression toward a partner, and one-fourth receive aggression from their own partner.
In terms of comorbidity with other psychiatric disorders, “IEDs don’t have more comorbidity in general than other disorders,” Dr. Coccaro noted. Personality disorders such as paranoid, antisocial, borderline narcissistic, and obsessive-compulsive disorders are more common in individuals with IED. Aggression in these people present differently depending on the personality disorder. “Someone who’s paranoid might blow up at you if you get in their face. For an antisocial, they’ll blow up at you if you’re preventing them from doing what they want to do. Borderlines, you reject them or you abandon them, they’re going to blow up. Narcissists will blow up when you reject. OCD will also blow up when you mess around with their sense of order,” Dr. Coccaro said.
Genetics also play a role in whether a person may have IED. These percentages were consistent, regardless of whether the individual had a comorbid condition, history of alcohol or drug use, or history of suicide, he said. Other factors that influence likelihood of IED are environment, behaviors such as smoking, and conditions such as traumatic brain injury. Experiencing aggression as a child is another factor.
“IED is the categorical expression of impulsive aggression, and it’s far more common than once thought,” Dr. Coccaro said. “And IED is totally unrecognized in its role in societal violence.”
Treatment can suppress, but not cure aggression
Medications used to treat aggression and impulsive aggression include lithium, SSRIs, mood stabilizers, neuroleptics, and beta-blockers. However, the treatments are not a “magic bullet,” Dr. Coccaro noted. “The meds tend to suppress aggressiveness, but not cure it.”
Timing of treatment is also a factor for medication. In studies of patients taking lithium for aggression, for example, “when they gave the drug to people who liked being aggressive, they didn’t like being on these drugs because it made them feel unprotected. It just was at odds with who they thought they were,” Dr. Coccaro said. “The people who took the drug and did well and really liked being on the drug with people who didn’t like that they were aggressive.”
Neurorehabilitation and cognitive-behavioral therapy specific to aggression, called cognitive relaxation and coping skills therapy, are nonpsychotropic approaches to treating aggression. “These therapeutic approaches are working not only to reduce progression, but also to reduce the social information processing problems that aggressive individuals have,” Dr. Coccaro said.
Another approach, known as interpretation bias training, teaches individuals with aggression to judge slightly angry-looking photos of people as not being angry. After 7-14 days of training, aggressive behavior in adolescents has been shown to be reduced. The changes were also visible on functional MRI.
“What they found was that when you treated them, the change in the amygdala went down when you looked at the angry faces and in the left lateral, post training, they became happier,” Dr. Coccaro said.
Global Academy and this news organization are owned by the same parent company. Dr. Coccaro reported serving as a consultant for Avanir, Azevan, and Bracket. He also reported receiving research grants from the National Institute of Mental Health, the National Institute on Alcohol Abuse and Alcoholism, and the Pritzker Pucker Family Foundation.
FROM FOCUS ON NEUROPSYCHIATRY 2020
‘The pandemic within the pandemic’
The coronavirus has infected millions of Americans and killed over 174,000. But could it be worse? Maybe.
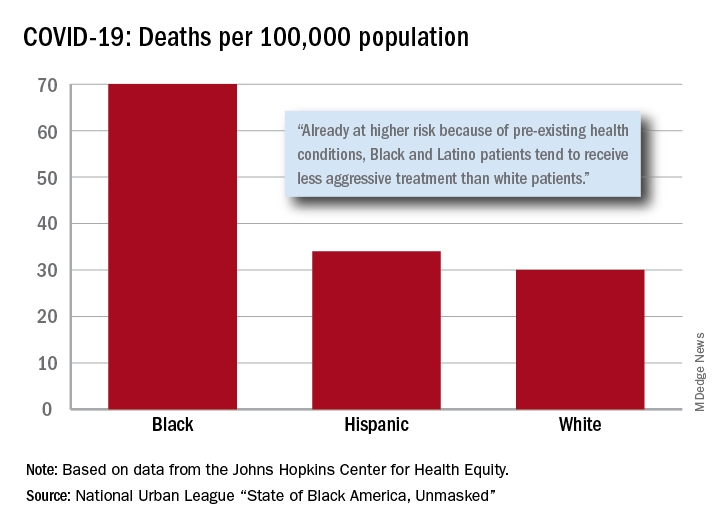
“Racism is the pandemic within the pandemic,” Marc H. Morial, president and CEO of the National Urban League, said in the 2020 “State of Black America, Unmasked” report.
“Black people with COVID-19 symptoms in February and March were less likely to get tested or treated than white patients,” he wrote.
After less testing and less treatment, the next step seems inevitable. The death rate from COVID-19 is 70 per 100,000 population among Black Americans, compared with 30 per 100,000 for Whites and 34 per 100,000 for Hispanics, the league said based on data from the Johns Hopkins Center for Health Equity.
Black and Hispanic patients with COVID-19 are more likely to have preexisting health conditions, but they “tend to receive less aggressive treatment than white patients,” the report noted. The lower death rate among Hispanics may be explained by the Black population’s greater age, although Hispanic Americans have a higher infection rate (73 per 10,000) than Blacks (62 per 10,000) or Whites (23 per 10,000).
Another possible explanation for the differences in infection rates: Blacks and Hispanics are less able to work at home because they “are overrepresented in low-wage jobs that offer the least flexibility and increase their risk of exposure to the coronavirus,” the league said.
Hispanics and Blacks also are more likely to be uninsured than Whites – 19.5% and 11.5%, respectively, vs. 7.5% – so “they tend to delay seeking treatment and are sicker than white patients when they finally do,” the league said. That may account for their much higher COVID-19 hospitalization rates: 213 per 100,000 for Blacks, 205 for Hispanics, and 46 for Whites.
“The silver lining during these dark times is that this pandemic has revealed our shared vulnerability and our interconnectedness. Many people are beginning to see that when others don’t have the opportunity to be healthy, it puts all of us at risk,” Lisa Cooper, MD, James F. Fries Professor of Medicine and Bloomberg Distinguished Professor in Health Equity at Johns Hopkins University, Baltimore, wrote in an essay accompanying the report.
The coronavirus has infected millions of Americans and killed over 174,000. But could it be worse? Maybe.

“Racism is the pandemic within the pandemic,” Marc H. Morial, president and CEO of the National Urban League, said in the 2020 “State of Black America, Unmasked” report.
“Black people with COVID-19 symptoms in February and March were less likely to get tested or treated than white patients,” he wrote.
After less testing and less treatment, the next step seems inevitable. The death rate from COVID-19 is 70 per 100,000 population among Black Americans, compared with 30 per 100,000 for Whites and 34 per 100,000 for Hispanics, the league said based on data from the Johns Hopkins Center for Health Equity.
Black and Hispanic patients with COVID-19 are more likely to have preexisting health conditions, but they “tend to receive less aggressive treatment than white patients,” the report noted. The lower death rate among Hispanics may be explained by the Black population’s greater age, although Hispanic Americans have a higher infection rate (73 per 10,000) than Blacks (62 per 10,000) or Whites (23 per 10,000).
Another possible explanation for the differences in infection rates: Blacks and Hispanics are less able to work at home because they “are overrepresented in low-wage jobs that offer the least flexibility and increase their risk of exposure to the coronavirus,” the league said.
Hispanics and Blacks also are more likely to be uninsured than Whites – 19.5% and 11.5%, respectively, vs. 7.5% – so “they tend to delay seeking treatment and are sicker than white patients when they finally do,” the league said. That may account for their much higher COVID-19 hospitalization rates: 213 per 100,000 for Blacks, 205 for Hispanics, and 46 for Whites.
“The silver lining during these dark times is that this pandemic has revealed our shared vulnerability and our interconnectedness. Many people are beginning to see that when others don’t have the opportunity to be healthy, it puts all of us at risk,” Lisa Cooper, MD, James F. Fries Professor of Medicine and Bloomberg Distinguished Professor in Health Equity at Johns Hopkins University, Baltimore, wrote in an essay accompanying the report.
The coronavirus has infected millions of Americans and killed over 174,000. But could it be worse? Maybe.

“Racism is the pandemic within the pandemic,” Marc H. Morial, president and CEO of the National Urban League, said in the 2020 “State of Black America, Unmasked” report.
“Black people with COVID-19 symptoms in February and March were less likely to get tested or treated than white patients,” he wrote.
After less testing and less treatment, the next step seems inevitable. The death rate from COVID-19 is 70 per 100,000 population among Black Americans, compared with 30 per 100,000 for Whites and 34 per 100,000 for Hispanics, the league said based on data from the Johns Hopkins Center for Health Equity.
Black and Hispanic patients with COVID-19 are more likely to have preexisting health conditions, but they “tend to receive less aggressive treatment than white patients,” the report noted. The lower death rate among Hispanics may be explained by the Black population’s greater age, although Hispanic Americans have a higher infection rate (73 per 10,000) than Blacks (62 per 10,000) or Whites (23 per 10,000).
Another possible explanation for the differences in infection rates: Blacks and Hispanics are less able to work at home because they “are overrepresented in low-wage jobs that offer the least flexibility and increase their risk of exposure to the coronavirus,” the league said.
Hispanics and Blacks also are more likely to be uninsured than Whites – 19.5% and 11.5%, respectively, vs. 7.5% – so “they tend to delay seeking treatment and are sicker than white patients when they finally do,” the league said. That may account for their much higher COVID-19 hospitalization rates: 213 per 100,000 for Blacks, 205 for Hispanics, and 46 for Whites.
“The silver lining during these dark times is that this pandemic has revealed our shared vulnerability and our interconnectedness. Many people are beginning to see that when others don’t have the opportunity to be healthy, it puts all of us at risk,” Lisa Cooper, MD, James F. Fries Professor of Medicine and Bloomberg Distinguished Professor in Health Equity at Johns Hopkins University, Baltimore, wrote in an essay accompanying the report.
Patient visits post COVID-19
Has telemedicine found its footing?
When Alexander Graham Bell invented the telephone, he accomplished something that many telegraph devotees never thought possible: the synchronous, bidirectional transmission of voice over electrical lines.
This was an incredible milestone in the advancement of mankind and enabled true revolutions in commerce, scientific collaboration, and human interaction. But Mr. Bell knew his invention didn’t represent the final advancement in telecommunication; he was quite prescient in imagining a day when individuals could see each other while speaking on the phone.
Many years later, what was once only a dream is now commonplace, and children growing up today can’t imagine a world where apps such as FaceTime and Skype don’t exist. Until recently, however, the medical community has been slow to adopt the idea of video interactions. This has dramatically changed because of the pandemic and the need for social distancing. It appears that telemedicine has found its footing, but whether it will remain popular once patients feel safe going to see their doctors in person again remains to be seen. This month, we’ll examine a few key issues that will determine the future of virtual medical visits.
Collect calling
The pandemic has wrought both human and economic casualties. With fear, job loss, and regulations leading to decreased spending, many large and small businesses have been and will continue to be unable to survive. Companies, including Brooks Brothers, Hertz, Lord and Taylor, GNC, and J.C. Penney, have declared bankruptcy.1 Medical practices and hospitals have taken cuts to their bottom line, and we’ve heard of many physician groups that have had to enact substantial salary cuts or even lay off providers – something previously unheard of. Recent months have demonstrated the health care community’s commitment to put patients first, but we simply cannot survive if we aren’t adequately reimbursed. Traditionally, this has been a significant roadblock toward the widespread adoption of telemedicine.
In most cases, these visits were not reimbursed at all. Thankfully, shortly after the coronavirus hit our shores, Medicare and Medicaid changed their policies, offering equal payment for video and in-person patient encounters. Most private insurers have followed suit, but the commitment to this payment parity appears – thus far – to be temporary. It is unclear that the financial support of telemedicine will continue post COVID-19, and this has many physicians feeling uncomfortable. In the meantime, many patients have come to prefer virtual visits, appreciating the convenience and efficiency.
Physicians don’t always have the same experience. Telemedicine can be technically challenging and take just as much – or sometimes more – time to navigate and document. Unless they are reimbursed equitably, providers will be forced to limit their use of virtual visits or not offer them at all. This leads to another issue: reliability.
‘Can you hear me now?’
Over the past several months, we have had the opportunity to use telemedicine firsthand and have spoken to many other physicians and patients about their experiences with it. The reports are all quite consistent: Most have had generally positive things to say. Still, some common concerns emerge when diving a bit deeper. Most notably are complaints about usability and reliability of the software.
While there are large telemedicine companies that have developed world-class cross-platform products, many in use today are proprietary and EHR dependent. As a result, the quality varies widely. Many EHR vendors were caught completely off guard by the sudden demand for telemedicine and are playing catch-up as they develop their own virtual visit platforms. While these vendor-developed platforms promise tight integration with patient records, some have significant shortcomings in stability when taxed under high utilization, including choppy video and garbled voice. This simply won’t do if telemedicine is to survive. It is incumbent on software developers and health care providers to invest in high-quality, reliable platforms on which to build their virtual visit offerings. This will ensure a more rapid adoption and the “staying power” of the new technology.
Dialing ‘0’ for the operator
Once seen as a “novelty” offered by only a small number of medical providers, virtual visits now represent a significant and ever-increasing percentage of patient encounters. The technology therefore must be easy to use. Given confidentiality and documentation requirements, along with the broad variety of available computing platforms and devices (e.g., PC, Mac, iOS, and Android), the process is often far from problem free. Patients may need help downloading apps, setting up webcams, or registering for the service. Providers may face issues with Internet connectivity or EHR-related delays.
It is critical that help be available to make the connection seamless and the experience a positive one. We are fortunate to work for a health care institution that has made this a priority, dedicating a team of individuals to provide real-time support to patients and clinicians. Small independent practices may not have this luxury, but we would encourage all providers to engage with their telemedicine or EHR vendors to determine what resources are available when problems arise, as they undoubtedly will.
Answering the call
Like the invention of the telephone, the advent of telemedicine is another milestone on the journey toward better communication with our patients, and it appears to be here to stay. Virtual visits won’t completely replace in-person care, nor minimize the benefit of human interaction, but they will continue to play an important role in the care continuum. By addressing the above concerns, we’ll lay a solid foundation for success and create a positive experience for physicians and patients alike.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Reference
1. A running list of companies that have filed for bankruptcy during the coronavirus pandemic. Fortune.
Has telemedicine found its footing?
Has telemedicine found its footing?
When Alexander Graham Bell invented the telephone, he accomplished something that many telegraph devotees never thought possible: the synchronous, bidirectional transmission of voice over electrical lines.
This was an incredible milestone in the advancement of mankind and enabled true revolutions in commerce, scientific collaboration, and human interaction. But Mr. Bell knew his invention didn’t represent the final advancement in telecommunication; he was quite prescient in imagining a day when individuals could see each other while speaking on the phone.
Many years later, what was once only a dream is now commonplace, and children growing up today can’t imagine a world where apps such as FaceTime and Skype don’t exist. Until recently, however, the medical community has been slow to adopt the idea of video interactions. This has dramatically changed because of the pandemic and the need for social distancing. It appears that telemedicine has found its footing, but whether it will remain popular once patients feel safe going to see their doctors in person again remains to be seen. This month, we’ll examine a few key issues that will determine the future of virtual medical visits.
Collect calling
The pandemic has wrought both human and economic casualties. With fear, job loss, and regulations leading to decreased spending, many large and small businesses have been and will continue to be unable to survive. Companies, including Brooks Brothers, Hertz, Lord and Taylor, GNC, and J.C. Penney, have declared bankruptcy.1 Medical practices and hospitals have taken cuts to their bottom line, and we’ve heard of many physician groups that have had to enact substantial salary cuts or even lay off providers – something previously unheard of. Recent months have demonstrated the health care community’s commitment to put patients first, but we simply cannot survive if we aren’t adequately reimbursed. Traditionally, this has been a significant roadblock toward the widespread adoption of telemedicine.
In most cases, these visits were not reimbursed at all. Thankfully, shortly after the coronavirus hit our shores, Medicare and Medicaid changed their policies, offering equal payment for video and in-person patient encounters. Most private insurers have followed suit, but the commitment to this payment parity appears – thus far – to be temporary. It is unclear that the financial support of telemedicine will continue post COVID-19, and this has many physicians feeling uncomfortable. In the meantime, many patients have come to prefer virtual visits, appreciating the convenience and efficiency.
Physicians don’t always have the same experience. Telemedicine can be technically challenging and take just as much – or sometimes more – time to navigate and document. Unless they are reimbursed equitably, providers will be forced to limit their use of virtual visits or not offer them at all. This leads to another issue: reliability.
‘Can you hear me now?’
Over the past several months, we have had the opportunity to use telemedicine firsthand and have spoken to many other physicians and patients about their experiences with it. The reports are all quite consistent: Most have had generally positive things to say. Still, some common concerns emerge when diving a bit deeper. Most notably are complaints about usability and reliability of the software.
While there are large telemedicine companies that have developed world-class cross-platform products, many in use today are proprietary and EHR dependent. As a result, the quality varies widely. Many EHR vendors were caught completely off guard by the sudden demand for telemedicine and are playing catch-up as they develop their own virtual visit platforms. While these vendor-developed platforms promise tight integration with patient records, some have significant shortcomings in stability when taxed under high utilization, including choppy video and garbled voice. This simply won’t do if telemedicine is to survive. It is incumbent on software developers and health care providers to invest in high-quality, reliable platforms on which to build their virtual visit offerings. This will ensure a more rapid adoption and the “staying power” of the new technology.
Dialing ‘0’ for the operator
Once seen as a “novelty” offered by only a small number of medical providers, virtual visits now represent a significant and ever-increasing percentage of patient encounters. The technology therefore must be easy to use. Given confidentiality and documentation requirements, along with the broad variety of available computing platforms and devices (e.g., PC, Mac, iOS, and Android), the process is often far from problem free. Patients may need help downloading apps, setting up webcams, or registering for the service. Providers may face issues with Internet connectivity or EHR-related delays.
It is critical that help be available to make the connection seamless and the experience a positive one. We are fortunate to work for a health care institution that has made this a priority, dedicating a team of individuals to provide real-time support to patients and clinicians. Small independent practices may not have this luxury, but we would encourage all providers to engage with their telemedicine or EHR vendors to determine what resources are available when problems arise, as they undoubtedly will.
Answering the call
Like the invention of the telephone, the advent of telemedicine is another milestone on the journey toward better communication with our patients, and it appears to be here to stay. Virtual visits won’t completely replace in-person care, nor minimize the benefit of human interaction, but they will continue to play an important role in the care continuum. By addressing the above concerns, we’ll lay a solid foundation for success and create a positive experience for physicians and patients alike.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Reference
1. A running list of companies that have filed for bankruptcy during the coronavirus pandemic. Fortune.
When Alexander Graham Bell invented the telephone, he accomplished something that many telegraph devotees never thought possible: the synchronous, bidirectional transmission of voice over electrical lines.
This was an incredible milestone in the advancement of mankind and enabled true revolutions in commerce, scientific collaboration, and human interaction. But Mr. Bell knew his invention didn’t represent the final advancement in telecommunication; he was quite prescient in imagining a day when individuals could see each other while speaking on the phone.
Many years later, what was once only a dream is now commonplace, and children growing up today can’t imagine a world where apps such as FaceTime and Skype don’t exist. Until recently, however, the medical community has been slow to adopt the idea of video interactions. This has dramatically changed because of the pandemic and the need for social distancing. It appears that telemedicine has found its footing, but whether it will remain popular once patients feel safe going to see their doctors in person again remains to be seen. This month, we’ll examine a few key issues that will determine the future of virtual medical visits.
Collect calling
The pandemic has wrought both human and economic casualties. With fear, job loss, and regulations leading to decreased spending, many large and small businesses have been and will continue to be unable to survive. Companies, including Brooks Brothers, Hertz, Lord and Taylor, GNC, and J.C. Penney, have declared bankruptcy.1 Medical practices and hospitals have taken cuts to their bottom line, and we’ve heard of many physician groups that have had to enact substantial salary cuts or even lay off providers – something previously unheard of. Recent months have demonstrated the health care community’s commitment to put patients first, but we simply cannot survive if we aren’t adequately reimbursed. Traditionally, this has been a significant roadblock toward the widespread adoption of telemedicine.
In most cases, these visits were not reimbursed at all. Thankfully, shortly after the coronavirus hit our shores, Medicare and Medicaid changed their policies, offering equal payment for video and in-person patient encounters. Most private insurers have followed suit, but the commitment to this payment parity appears – thus far – to be temporary. It is unclear that the financial support of telemedicine will continue post COVID-19, and this has many physicians feeling uncomfortable. In the meantime, many patients have come to prefer virtual visits, appreciating the convenience and efficiency.
Physicians don’t always have the same experience. Telemedicine can be technically challenging and take just as much – or sometimes more – time to navigate and document. Unless they are reimbursed equitably, providers will be forced to limit their use of virtual visits or not offer them at all. This leads to another issue: reliability.
‘Can you hear me now?’
Over the past several months, we have had the opportunity to use telemedicine firsthand and have spoken to many other physicians and patients about their experiences with it. The reports are all quite consistent: Most have had generally positive things to say. Still, some common concerns emerge when diving a bit deeper. Most notably are complaints about usability and reliability of the software.
While there are large telemedicine companies that have developed world-class cross-platform products, many in use today are proprietary and EHR dependent. As a result, the quality varies widely. Many EHR vendors were caught completely off guard by the sudden demand for telemedicine and are playing catch-up as they develop their own virtual visit platforms. While these vendor-developed platforms promise tight integration with patient records, some have significant shortcomings in stability when taxed under high utilization, including choppy video and garbled voice. This simply won’t do if telemedicine is to survive. It is incumbent on software developers and health care providers to invest in high-quality, reliable platforms on which to build their virtual visit offerings. This will ensure a more rapid adoption and the “staying power” of the new technology.
Dialing ‘0’ for the operator
Once seen as a “novelty” offered by only a small number of medical providers, virtual visits now represent a significant and ever-increasing percentage of patient encounters. The technology therefore must be easy to use. Given confidentiality and documentation requirements, along with the broad variety of available computing platforms and devices (e.g., PC, Mac, iOS, and Android), the process is often far from problem free. Patients may need help downloading apps, setting up webcams, or registering for the service. Providers may face issues with Internet connectivity or EHR-related delays.
It is critical that help be available to make the connection seamless and the experience a positive one. We are fortunate to work for a health care institution that has made this a priority, dedicating a team of individuals to provide real-time support to patients and clinicians. Small independent practices may not have this luxury, but we would encourage all providers to engage with their telemedicine or EHR vendors to determine what resources are available when problems arise, as they undoubtedly will.
Answering the call
Like the invention of the telephone, the advent of telemedicine is another milestone on the journey toward better communication with our patients, and it appears to be here to stay. Virtual visits won’t completely replace in-person care, nor minimize the benefit of human interaction, but they will continue to play an important role in the care continuum. By addressing the above concerns, we’ll lay a solid foundation for success and create a positive experience for physicians and patients alike.
Dr. Notte is a family physician and chief medical officer of Abington (Pa.) Hospital–Jefferson Health. Follow him on Twitter (@doctornotte). Dr. Skolnik is professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, and associate director of the family medicine residency program at Abington (Pa.) Hospital–Jefferson Health. They have no conflicts related to the content of this piece.
Reference
1. A running list of companies that have filed for bankruptcy during the coronavirus pandemic. Fortune.
Swab, spit, stay home? College coronavirus testing plans are all over the map
Yousuf El-Jayyousi, a junior engineering student at the University of Missouri, wanted guidance and reassurance that it would be safe to go back to school for the fall semester. He tuned into a pair of online town halls organized by the university hoping to find that.
He did not.

What he got instead from those town halls last month was encouragement to return to class at the institution affectionately known as Mizzou. The university, in Columbia, would be testing only people with symptoms, and at that point, the university said people who test positive off campus were under no obligation to inform the school.
“It feels like the university doesn’t really care whether we get sick or not,” said El-Jayyousi, who is scheduled for two in-person classes, and lives at home with his parents and 90-year-old grandmother.
He’s seen the studies from researchers at Yale and Harvard that suggest testing needs to be much more widespread. He asked his instructors if he could join lectures remotely once classes begin Monday. One was considering it; the other rejected it.
“It was kind of very dismissive, like ‘so what?’ ” El-Jayyousi said.
But it’s an enormous “so what?” packed with fear and unknowns for Jayyousi and some 20 million other students enrolled in some level of postsecondary education in America, if they are not already online only.
Policies for reentry onto campuses that were abruptly shut in March are all over the map.
Hundreds Undecided
According to the College Crisis Initiative, or C2i, a project of Davidson College that monitors how higher ed is responding to the pandemic, there is nothing resembling a common approach. Of 2,958 institutions it follows, 151 were planning to open fully online, 729 were mostly online and 433 were taking a hybrid approach. Just 75 schools were insisting on students attending fully in person, and 614 were aiming to be primarily in-person. Some 800 others were still deciding, just weeks before instruction was to start.
The decisions often have little correlation with the public health advisories in the region. Mizzou, which is in an area with recent COVID spikes, is holding some in-person instruction and has nearly 7,000 students signed up to live in dorms and other university-owned housing. Harvard, in a region with extremely low rates of viral spread, has opted to go all online and allowed students to defer a year.
The specific circumstances colleges and universities face are as much determined by local fiscal and political dictates as by medicine and epidemiology. It is often unclear who is making the call. So it’s every student for herself to chart these unknown waters, even as students (or their families) have written tuition checks for tens of thousands of dollars and signed leases for campus and off-campus housing.
And the risks – health, educational and financial – boomerang back on individual students: Two weeks after University of North Carolina students, as instructed, returned to the flagship campus in Chapel Hill with the promise of at least some in-person learning, all classes went online. Early outbreaks surged from a few students to more than 130 in a matter of days. Most undergrads have about a week to clear out of their dorms.
“It’s really tough,” said neuroscience major Luke Lawless, 20. “Chapel Hill is an amazing place, and as a senior it’s tough to know that my time’s running out – and the virus only adds to that.”
Location, location, location
C2i’s creator, Davidson education Assistant Professor Chris Marsicano, said the extreme diversity of approaches comes from the sheer diversity of schools, the penchant of many to follow the leads of more prestigious peers, and local politics.
“Some states have very strong and stringent mask requirements. Some have stronger stay-at-home orders. Others are sort of leaving it up to localities. So the confluence of politics, institutional isomorphism – that imitation – and different needs that the institutions have are driving the differences,” Marsicano said.
Location matters a lot, too, Marsicano said, pointing to schools like George Washington University and Boston University in urban settings where the environment is beyond the control of the school, versus a place like the University of the South in remote, rural Sewanee, Tennessee, where 90% of students will return to campus.
“It’s a lot easier to control an outbreak if you are a fairly isolated college campus than if you are in the middle of a city,” Marsicano said.
Student behavior is another wild card, Marsicano said, since even the best plans will fail if college kids “do something stupid, like have a massive frat party without masks.”
“You’ve got student affairs professionals across the country who are screaming at the top of their lungs, ‘We can’t control student behavior when they go off campus’” Marsicano said.
Another factor is a vacuum at the federal level. Although the Department of Education says Secretary Betsy DeVos has held dozens of calls with governors and state education superintendents, there’s no sign of an attempt to offer unified guidance to colleges beyond a webpage that links to relaxed regulatory requirements and anodyne fact sheets from the Centers for Disease Control and Prevention on preventing viral spread.
Even the money that the department notes it has dispensed – $30 billion from Congress’ CARES Act – is weighted toward K-12 schools, with about $13 billion for higher education, including student aid.
The U.S. Senate adjourned last week until Sept. 8, having never taken up a House-passed relief package that included some $30 billion for higher education. A trio of Democratic senators, including Sen. Elizabeth Warren, is calling for national reporting standards on college campuses.
No benchmarks
Campus communities with very different levels of contagion are making opposite calls about in-person learning. Mizzou’s Boone County has seen more than 1,400 confirmed COVID cases after a spike in mid-July. According to the Harvard Global Health Institute’s COVID risk map, Boone has accelerated spread, with 14 infections per day per 100,000 people. The institute advises stay-at-home orders or rigorous testing and tracing at such rates of infection. Two neighboring counties were in the red zone recently, with more than 25 cases per day per 100,000 people. Mizzou has left it up to deans whether classes will meet in person, making a strong argument for face-to-face instruction.
Meanwhile, Columbia University in New York City opted for all online instruction, even though the rate of infection there is a comparatively low 3.8 cases per day per 100,000 people.
Administrators at Mizzou considered and rejected mandatory testing. “All that does is provide one a snapshot of the situation,” University of Missouri system President Mun Choi said in one of the town halls.
Mizzou has an in-house team that will carry out case investigation and contact tracing with the local health department. This week, following questions from the press and pressure from the public, the university announced students will be required to report any positive COVID test to the school.
Who do you test? When?
CDC guidance for higher education suggests there’s not enough data to know whether testing everyone is effective, but some influential researchers, such as those at Harvard and Yale, disagree.
“This virus is subject to silent spreading and asymptomatic spreading, and it’s very hard to play catch-up,” said Yale professor David Paltiel, who studies public health policy. “And so thinking that you can keep your campus safe by simply waiting until students develop symptoms before acting, I think, is a very dangerous game.”
Simulation models conducted by Paltiel and his colleagues show that, of all the factors university administrators can control – including the sensitivity and specificity of COVID-19 tests – the frequency of testing is most important.
He’s “painfully aware” that testing everyone on campus every few days sets a very high bar – logistically, financially, behaviorally – that may be beyond what most schools can reach. But he says the consequences of reopening campuses without those measures are severe, not just for students, but for vulnerable populations among school workers and in the surrounding community.
“You really have to ask yourself whether you have any business reopening if you’re not going to commit to an aggressive program of high-frequency testing,” he said.
The fighting – and testing – Illini
Some institutions that desperately want students to return to campus are backing the goal with a maximal approach to safety and testing.
About a 4-hour drive east along the interstates from Mizzou is the University of Illinois at Urbana-Champaign, whose sports teams are known as the Fighting Illini.
Weeks ago, large white tents with signs reading “Walk-Up COVID-19 Testing” have popped up across campus; there students take a simple saliva test.
“This seems to be a lot easier than sticking a cotton swab up your nose,” graduate student Kristen Muñoz said after collecting a bit of her saliva in a plastic tube and sealing it in a bag labeled “Biohazard.”
In just a few hours, she got back her result: negative.
The school plans to offer free tests to the 50,000 students expected to return this month, as well as some 11,000 faculty and staff members.
“The exciting thing is, because we can test up to 10,000 per day, it allows the scientist to do what’s really the best for trying to protect the community as opposed to having to cut corners, because of the limitations of the testing,” said University of Illinois chemist Martin Burke, who helped develop the campus’s saliva test, which received emergency use authorization from the federal Food and Drug Administration this week.
The test is similar to one designed by Yale and funded by the NBA that cleared the FDA hurdle just before the Illinois test. Both Yale and Illinois hope aggressive testing will allow most undergraduate students to live on campus, even though most classes will be online.
University of Illinois epidemiologist Becky Smith said they are following data that suggest campuses need to test everyone every few days because the virus is not detectable in infected people for 3 or 4 days.
“But about two days after that, your infectiousness peaks,” she said. “So, we have a very small window of time in which to catch people before they have done most of the infection that they’re going to be doing.”
Campus officials accepted Smith’s recommendation that all faculty, staffers and students participating in any on-campus activities be required to get tested twice a week.
Illinois can do that because its test is convenient and not invasive, which spares the campus from using as much personal protective equipment as the more invasive tests require, Burke said. And on-site analysis avoids backlogs at public health and commercial labs.
Muddled in the middle
Most other colleges fall somewhere between the approaches of Mizzou and the University of Illinois, and many of their students still are uncertain how their fall semester will go.
At the University of Southern California, a private campus of about 48,500 students in Los Angeles, officials had hoped to have about 20% of classes in person – but the county government scaled that back, insisting on tougher rules for reopening than the statewide standards.
If students eventually are allowed back, they will have to show a recent coronavirus test result that they obtained on their own, said Dr. Sarah Van Orman, chief health officer of USC Student Health.
They will be asked to do daily health assessments, such as fever checks, and those who have been exposed to the virus or show symptoms will receive a rapid test, with about a 24-hour turnaround through the university medical center’s lab. “We believe it is really important to have very rapid access to those results,” Van Orman said.
At California State University – the nation’s largest 4-year system, with 23 campuses and nearly a half-million students – officials decided back in May to move nearly all its fall courses online.
“The first priority was really the health and safety of all of the campus community,” said Mike Uhlenkamp, spokesperson for the CSU Chancellor’s Office. About 10% of CSU students are expected to attend some in-person classes, such as nursing lab courses, fine art and dance classes, and some graduate classes.
Uhlenkamp said testing protocols are being left up to each campus, though all are required to follow local safety guidelines. And without a medical campus in the system, CSU campuses do not have the same capacity to take charge of their own testing, as the University of Illinois is doing.
For students who know they won’t be on campus this fall, there is regret at lost social experiences, networking and hands-on learning so important to college.
But the certainty also brings relief.
“I don’t think I would want to be indoors with a group of, you know, even just a handful of people, even if we have masks on,” said Haley Gray, a 28-year-old graduate student at the University of California-Berkeley starting the second year of her journalism program.
She knows she won’t have access to Berkeley’s advanced media labs or the collaborative sessions students experience there. And she said she realized the other day she probably won’t just sit around the student lounge and strike up unexpected friendships.
“That’s a pretty big bummer but, you know, I think overall we’re all just doing our best, and given the circumstances, I feel pretty OK about it,” she said.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente. This story is part of a partnership that includes KBIA, Illinois Public Media, Side Effects Public Media, NPR and Kaiser Health News.
Yousuf El-Jayyousi, a junior engineering student at the University of Missouri, wanted guidance and reassurance that it would be safe to go back to school for the fall semester. He tuned into a pair of online town halls organized by the university hoping to find that.
He did not.

What he got instead from those town halls last month was encouragement to return to class at the institution affectionately known as Mizzou. The university, in Columbia, would be testing only people with symptoms, and at that point, the university said people who test positive off campus were under no obligation to inform the school.
“It feels like the university doesn’t really care whether we get sick or not,” said El-Jayyousi, who is scheduled for two in-person classes, and lives at home with his parents and 90-year-old grandmother.
He’s seen the studies from researchers at Yale and Harvard that suggest testing needs to be much more widespread. He asked his instructors if he could join lectures remotely once classes begin Monday. One was considering it; the other rejected it.
“It was kind of very dismissive, like ‘so what?’ ” El-Jayyousi said.
But it’s an enormous “so what?” packed with fear and unknowns for Jayyousi and some 20 million other students enrolled in some level of postsecondary education in America, if they are not already online only.
Policies for reentry onto campuses that were abruptly shut in March are all over the map.
Hundreds Undecided
According to the College Crisis Initiative, or C2i, a project of Davidson College that monitors how higher ed is responding to the pandemic, there is nothing resembling a common approach. Of 2,958 institutions it follows, 151 were planning to open fully online, 729 were mostly online and 433 were taking a hybrid approach. Just 75 schools were insisting on students attending fully in person, and 614 were aiming to be primarily in-person. Some 800 others were still deciding, just weeks before instruction was to start.
The decisions often have little correlation with the public health advisories in the region. Mizzou, which is in an area with recent COVID spikes, is holding some in-person instruction and has nearly 7,000 students signed up to live in dorms and other university-owned housing. Harvard, in a region with extremely low rates of viral spread, has opted to go all online and allowed students to defer a year.
The specific circumstances colleges and universities face are as much determined by local fiscal and political dictates as by medicine and epidemiology. It is often unclear who is making the call. So it’s every student for herself to chart these unknown waters, even as students (or their families) have written tuition checks for tens of thousands of dollars and signed leases for campus and off-campus housing.
And the risks – health, educational and financial – boomerang back on individual students: Two weeks after University of North Carolina students, as instructed, returned to the flagship campus in Chapel Hill with the promise of at least some in-person learning, all classes went online. Early outbreaks surged from a few students to more than 130 in a matter of days. Most undergrads have about a week to clear out of their dorms.
“It’s really tough,” said neuroscience major Luke Lawless, 20. “Chapel Hill is an amazing place, and as a senior it’s tough to know that my time’s running out – and the virus only adds to that.”
Location, location, location
C2i’s creator, Davidson education Assistant Professor Chris Marsicano, said the extreme diversity of approaches comes from the sheer diversity of schools, the penchant of many to follow the leads of more prestigious peers, and local politics.
“Some states have very strong and stringent mask requirements. Some have stronger stay-at-home orders. Others are sort of leaving it up to localities. So the confluence of politics, institutional isomorphism – that imitation – and different needs that the institutions have are driving the differences,” Marsicano said.
Location matters a lot, too, Marsicano said, pointing to schools like George Washington University and Boston University in urban settings where the environment is beyond the control of the school, versus a place like the University of the South in remote, rural Sewanee, Tennessee, where 90% of students will return to campus.
“It’s a lot easier to control an outbreak if you are a fairly isolated college campus than if you are in the middle of a city,” Marsicano said.
Student behavior is another wild card, Marsicano said, since even the best plans will fail if college kids “do something stupid, like have a massive frat party without masks.”
“You’ve got student affairs professionals across the country who are screaming at the top of their lungs, ‘We can’t control student behavior when they go off campus’” Marsicano said.
Another factor is a vacuum at the federal level. Although the Department of Education says Secretary Betsy DeVos has held dozens of calls with governors and state education superintendents, there’s no sign of an attempt to offer unified guidance to colleges beyond a webpage that links to relaxed regulatory requirements and anodyne fact sheets from the Centers for Disease Control and Prevention on preventing viral spread.
Even the money that the department notes it has dispensed – $30 billion from Congress’ CARES Act – is weighted toward K-12 schools, with about $13 billion for higher education, including student aid.
The U.S. Senate adjourned last week until Sept. 8, having never taken up a House-passed relief package that included some $30 billion for higher education. A trio of Democratic senators, including Sen. Elizabeth Warren, is calling for national reporting standards on college campuses.
No benchmarks
Campus communities with very different levels of contagion are making opposite calls about in-person learning. Mizzou’s Boone County has seen more than 1,400 confirmed COVID cases after a spike in mid-July. According to the Harvard Global Health Institute’s COVID risk map, Boone has accelerated spread, with 14 infections per day per 100,000 people. The institute advises stay-at-home orders or rigorous testing and tracing at such rates of infection. Two neighboring counties were in the red zone recently, with more than 25 cases per day per 100,000 people. Mizzou has left it up to deans whether classes will meet in person, making a strong argument for face-to-face instruction.
Meanwhile, Columbia University in New York City opted for all online instruction, even though the rate of infection there is a comparatively low 3.8 cases per day per 100,000 people.
Administrators at Mizzou considered and rejected mandatory testing. “All that does is provide one a snapshot of the situation,” University of Missouri system President Mun Choi said in one of the town halls.
Mizzou has an in-house team that will carry out case investigation and contact tracing with the local health department. This week, following questions from the press and pressure from the public, the university announced students will be required to report any positive COVID test to the school.
Who do you test? When?
CDC guidance for higher education suggests there’s not enough data to know whether testing everyone is effective, but some influential researchers, such as those at Harvard and Yale, disagree.
“This virus is subject to silent spreading and asymptomatic spreading, and it’s very hard to play catch-up,” said Yale professor David Paltiel, who studies public health policy. “And so thinking that you can keep your campus safe by simply waiting until students develop symptoms before acting, I think, is a very dangerous game.”
Simulation models conducted by Paltiel and his colleagues show that, of all the factors university administrators can control – including the sensitivity and specificity of COVID-19 tests – the frequency of testing is most important.
He’s “painfully aware” that testing everyone on campus every few days sets a very high bar – logistically, financially, behaviorally – that may be beyond what most schools can reach. But he says the consequences of reopening campuses without those measures are severe, not just for students, but for vulnerable populations among school workers and in the surrounding community.
“You really have to ask yourself whether you have any business reopening if you’re not going to commit to an aggressive program of high-frequency testing,” he said.
The fighting – and testing – Illini
Some institutions that desperately want students to return to campus are backing the goal with a maximal approach to safety and testing.
About a 4-hour drive east along the interstates from Mizzou is the University of Illinois at Urbana-Champaign, whose sports teams are known as the Fighting Illini.
Weeks ago, large white tents with signs reading “Walk-Up COVID-19 Testing” have popped up across campus; there students take a simple saliva test.
“This seems to be a lot easier than sticking a cotton swab up your nose,” graduate student Kristen Muñoz said after collecting a bit of her saliva in a plastic tube and sealing it in a bag labeled “Biohazard.”
In just a few hours, she got back her result: negative.
The school plans to offer free tests to the 50,000 students expected to return this month, as well as some 11,000 faculty and staff members.
“The exciting thing is, because we can test up to 10,000 per day, it allows the scientist to do what’s really the best for trying to protect the community as opposed to having to cut corners, because of the limitations of the testing,” said University of Illinois chemist Martin Burke, who helped develop the campus’s saliva test, which received emergency use authorization from the federal Food and Drug Administration this week.
The test is similar to one designed by Yale and funded by the NBA that cleared the FDA hurdle just before the Illinois test. Both Yale and Illinois hope aggressive testing will allow most undergraduate students to live on campus, even though most classes will be online.
University of Illinois epidemiologist Becky Smith said they are following data that suggest campuses need to test everyone every few days because the virus is not detectable in infected people for 3 or 4 days.
“But about two days after that, your infectiousness peaks,” she said. “So, we have a very small window of time in which to catch people before they have done most of the infection that they’re going to be doing.”
Campus officials accepted Smith’s recommendation that all faculty, staffers and students participating in any on-campus activities be required to get tested twice a week.
Illinois can do that because its test is convenient and not invasive, which spares the campus from using as much personal protective equipment as the more invasive tests require, Burke said. And on-site analysis avoids backlogs at public health and commercial labs.
Muddled in the middle
Most other colleges fall somewhere between the approaches of Mizzou and the University of Illinois, and many of their students still are uncertain how their fall semester will go.
At the University of Southern California, a private campus of about 48,500 students in Los Angeles, officials had hoped to have about 20% of classes in person – but the county government scaled that back, insisting on tougher rules for reopening than the statewide standards.
If students eventually are allowed back, they will have to show a recent coronavirus test result that they obtained on their own, said Dr. Sarah Van Orman, chief health officer of USC Student Health.
They will be asked to do daily health assessments, such as fever checks, and those who have been exposed to the virus or show symptoms will receive a rapid test, with about a 24-hour turnaround through the university medical center’s lab. “We believe it is really important to have very rapid access to those results,” Van Orman said.
At California State University – the nation’s largest 4-year system, with 23 campuses and nearly a half-million students – officials decided back in May to move nearly all its fall courses online.
“The first priority was really the health and safety of all of the campus community,” said Mike Uhlenkamp, spokesperson for the CSU Chancellor’s Office. About 10% of CSU students are expected to attend some in-person classes, such as nursing lab courses, fine art and dance classes, and some graduate classes.
Uhlenkamp said testing protocols are being left up to each campus, though all are required to follow local safety guidelines. And without a medical campus in the system, CSU campuses do not have the same capacity to take charge of their own testing, as the University of Illinois is doing.
For students who know they won’t be on campus this fall, there is regret at lost social experiences, networking and hands-on learning so important to college.
But the certainty also brings relief.
“I don’t think I would want to be indoors with a group of, you know, even just a handful of people, even if we have masks on,” said Haley Gray, a 28-year-old graduate student at the University of California-Berkeley starting the second year of her journalism program.
She knows she won’t have access to Berkeley’s advanced media labs or the collaborative sessions students experience there. And she said she realized the other day she probably won’t just sit around the student lounge and strike up unexpected friendships.
“That’s a pretty big bummer but, you know, I think overall we’re all just doing our best, and given the circumstances, I feel pretty OK about it,” she said.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente. This story is part of a partnership that includes KBIA, Illinois Public Media, Side Effects Public Media, NPR and Kaiser Health News.
Yousuf El-Jayyousi, a junior engineering student at the University of Missouri, wanted guidance and reassurance that it would be safe to go back to school for the fall semester. He tuned into a pair of online town halls organized by the university hoping to find that.
He did not.

What he got instead from those town halls last month was encouragement to return to class at the institution affectionately known as Mizzou. The university, in Columbia, would be testing only people with symptoms, and at that point, the university said people who test positive off campus were under no obligation to inform the school.
“It feels like the university doesn’t really care whether we get sick or not,” said El-Jayyousi, who is scheduled for two in-person classes, and lives at home with his parents and 90-year-old grandmother.
He’s seen the studies from researchers at Yale and Harvard that suggest testing needs to be much more widespread. He asked his instructors if he could join lectures remotely once classes begin Monday. One was considering it; the other rejected it.
“It was kind of very dismissive, like ‘so what?’ ” El-Jayyousi said.
But it’s an enormous “so what?” packed with fear and unknowns for Jayyousi and some 20 million other students enrolled in some level of postsecondary education in America, if they are not already online only.
Policies for reentry onto campuses that were abruptly shut in March are all over the map.
Hundreds Undecided
According to the College Crisis Initiative, or C2i, a project of Davidson College that monitors how higher ed is responding to the pandemic, there is nothing resembling a common approach. Of 2,958 institutions it follows, 151 were planning to open fully online, 729 were mostly online and 433 were taking a hybrid approach. Just 75 schools were insisting on students attending fully in person, and 614 were aiming to be primarily in-person. Some 800 others were still deciding, just weeks before instruction was to start.
The decisions often have little correlation with the public health advisories in the region. Mizzou, which is in an area with recent COVID spikes, is holding some in-person instruction and has nearly 7,000 students signed up to live in dorms and other university-owned housing. Harvard, in a region with extremely low rates of viral spread, has opted to go all online and allowed students to defer a year.
The specific circumstances colleges and universities face are as much determined by local fiscal and political dictates as by medicine and epidemiology. It is often unclear who is making the call. So it’s every student for herself to chart these unknown waters, even as students (or their families) have written tuition checks for tens of thousands of dollars and signed leases for campus and off-campus housing.
And the risks – health, educational and financial – boomerang back on individual students: Two weeks after University of North Carolina students, as instructed, returned to the flagship campus in Chapel Hill with the promise of at least some in-person learning, all classes went online. Early outbreaks surged from a few students to more than 130 in a matter of days. Most undergrads have about a week to clear out of their dorms.
“It’s really tough,” said neuroscience major Luke Lawless, 20. “Chapel Hill is an amazing place, and as a senior it’s tough to know that my time’s running out – and the virus only adds to that.”
Location, location, location
C2i’s creator, Davidson education Assistant Professor Chris Marsicano, said the extreme diversity of approaches comes from the sheer diversity of schools, the penchant of many to follow the leads of more prestigious peers, and local politics.
“Some states have very strong and stringent mask requirements. Some have stronger stay-at-home orders. Others are sort of leaving it up to localities. So the confluence of politics, institutional isomorphism – that imitation – and different needs that the institutions have are driving the differences,” Marsicano said.
Location matters a lot, too, Marsicano said, pointing to schools like George Washington University and Boston University in urban settings where the environment is beyond the control of the school, versus a place like the University of the South in remote, rural Sewanee, Tennessee, where 90% of students will return to campus.
“It’s a lot easier to control an outbreak if you are a fairly isolated college campus than if you are in the middle of a city,” Marsicano said.
Student behavior is another wild card, Marsicano said, since even the best plans will fail if college kids “do something stupid, like have a massive frat party without masks.”
“You’ve got student affairs professionals across the country who are screaming at the top of their lungs, ‘We can’t control student behavior when they go off campus’” Marsicano said.
Another factor is a vacuum at the federal level. Although the Department of Education says Secretary Betsy DeVos has held dozens of calls with governors and state education superintendents, there’s no sign of an attempt to offer unified guidance to colleges beyond a webpage that links to relaxed regulatory requirements and anodyne fact sheets from the Centers for Disease Control and Prevention on preventing viral spread.
Even the money that the department notes it has dispensed – $30 billion from Congress’ CARES Act – is weighted toward K-12 schools, with about $13 billion for higher education, including student aid.
The U.S. Senate adjourned last week until Sept. 8, having never taken up a House-passed relief package that included some $30 billion for higher education. A trio of Democratic senators, including Sen. Elizabeth Warren, is calling for national reporting standards on college campuses.
No benchmarks
Campus communities with very different levels of contagion are making opposite calls about in-person learning. Mizzou’s Boone County has seen more than 1,400 confirmed COVID cases after a spike in mid-July. According to the Harvard Global Health Institute’s COVID risk map, Boone has accelerated spread, with 14 infections per day per 100,000 people. The institute advises stay-at-home orders or rigorous testing and tracing at such rates of infection. Two neighboring counties were in the red zone recently, with more than 25 cases per day per 100,000 people. Mizzou has left it up to deans whether classes will meet in person, making a strong argument for face-to-face instruction.
Meanwhile, Columbia University in New York City opted for all online instruction, even though the rate of infection there is a comparatively low 3.8 cases per day per 100,000 people.
Administrators at Mizzou considered and rejected mandatory testing. “All that does is provide one a snapshot of the situation,” University of Missouri system President Mun Choi said in one of the town halls.
Mizzou has an in-house team that will carry out case investigation and contact tracing with the local health department. This week, following questions from the press and pressure from the public, the university announced students will be required to report any positive COVID test to the school.
Who do you test? When?
CDC guidance for higher education suggests there’s not enough data to know whether testing everyone is effective, but some influential researchers, such as those at Harvard and Yale, disagree.
“This virus is subject to silent spreading and asymptomatic spreading, and it’s very hard to play catch-up,” said Yale professor David Paltiel, who studies public health policy. “And so thinking that you can keep your campus safe by simply waiting until students develop symptoms before acting, I think, is a very dangerous game.”
Simulation models conducted by Paltiel and his colleagues show that, of all the factors university administrators can control – including the sensitivity and specificity of COVID-19 tests – the frequency of testing is most important.
He’s “painfully aware” that testing everyone on campus every few days sets a very high bar – logistically, financially, behaviorally – that may be beyond what most schools can reach. But he says the consequences of reopening campuses without those measures are severe, not just for students, but for vulnerable populations among school workers and in the surrounding community.
“You really have to ask yourself whether you have any business reopening if you’re not going to commit to an aggressive program of high-frequency testing,” he said.
The fighting – and testing – Illini
Some institutions that desperately want students to return to campus are backing the goal with a maximal approach to safety and testing.
About a 4-hour drive east along the interstates from Mizzou is the University of Illinois at Urbana-Champaign, whose sports teams are known as the Fighting Illini.
Weeks ago, large white tents with signs reading “Walk-Up COVID-19 Testing” have popped up across campus; there students take a simple saliva test.
“This seems to be a lot easier than sticking a cotton swab up your nose,” graduate student Kristen Muñoz said after collecting a bit of her saliva in a plastic tube and sealing it in a bag labeled “Biohazard.”
In just a few hours, she got back her result: negative.
The school plans to offer free tests to the 50,000 students expected to return this month, as well as some 11,000 faculty and staff members.
“The exciting thing is, because we can test up to 10,000 per day, it allows the scientist to do what’s really the best for trying to protect the community as opposed to having to cut corners, because of the limitations of the testing,” said University of Illinois chemist Martin Burke, who helped develop the campus’s saliva test, which received emergency use authorization from the federal Food and Drug Administration this week.
The test is similar to one designed by Yale and funded by the NBA that cleared the FDA hurdle just before the Illinois test. Both Yale and Illinois hope aggressive testing will allow most undergraduate students to live on campus, even though most classes will be online.
University of Illinois epidemiologist Becky Smith said they are following data that suggest campuses need to test everyone every few days because the virus is not detectable in infected people for 3 or 4 days.
“But about two days after that, your infectiousness peaks,” she said. “So, we have a very small window of time in which to catch people before they have done most of the infection that they’re going to be doing.”
Campus officials accepted Smith’s recommendation that all faculty, staffers and students participating in any on-campus activities be required to get tested twice a week.
Illinois can do that because its test is convenient and not invasive, which spares the campus from using as much personal protective equipment as the more invasive tests require, Burke said. And on-site analysis avoids backlogs at public health and commercial labs.
Muddled in the middle
Most other colleges fall somewhere between the approaches of Mizzou and the University of Illinois, and many of their students still are uncertain how their fall semester will go.
At the University of Southern California, a private campus of about 48,500 students in Los Angeles, officials had hoped to have about 20% of classes in person – but the county government scaled that back, insisting on tougher rules for reopening than the statewide standards.
If students eventually are allowed back, they will have to show a recent coronavirus test result that they obtained on their own, said Dr. Sarah Van Orman, chief health officer of USC Student Health.
They will be asked to do daily health assessments, such as fever checks, and those who have been exposed to the virus or show symptoms will receive a rapid test, with about a 24-hour turnaround through the university medical center’s lab. “We believe it is really important to have very rapid access to those results,” Van Orman said.
At California State University – the nation’s largest 4-year system, with 23 campuses and nearly a half-million students – officials decided back in May to move nearly all its fall courses online.
“The first priority was really the health and safety of all of the campus community,” said Mike Uhlenkamp, spokesperson for the CSU Chancellor’s Office. About 10% of CSU students are expected to attend some in-person classes, such as nursing lab courses, fine art and dance classes, and some graduate classes.
Uhlenkamp said testing protocols are being left up to each campus, though all are required to follow local safety guidelines. And without a medical campus in the system, CSU campuses do not have the same capacity to take charge of their own testing, as the University of Illinois is doing.
For students who know they won’t be on campus this fall, there is regret at lost social experiences, networking and hands-on learning so important to college.
But the certainty also brings relief.
“I don’t think I would want to be indoors with a group of, you know, even just a handful of people, even if we have masks on,” said Haley Gray, a 28-year-old graduate student at the University of California-Berkeley starting the second year of her journalism program.
She knows she won’t have access to Berkeley’s advanced media labs or the collaborative sessions students experience there. And she said she realized the other day she probably won’t just sit around the student lounge and strike up unexpected friendships.
“That’s a pretty big bummer but, you know, I think overall we’re all just doing our best, and given the circumstances, I feel pretty OK about it,” she said.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente. This story is part of a partnership that includes KBIA, Illinois Public Media, Side Effects Public Media, NPR and Kaiser Health News.
ADHD and dyslexia may affect evaluation of concussion
, a new study shows.
“Our results suggest kids with certain learning disorders may respond differently to concussion tests, and this needs to be taken into account when advising on recovery times and when they can return to sport,” said lead author Mathew Stokes, MD. Dr. Stokes is assistant professor of pediatrics and neurology/neurotherapeutics at the University of Texas–Southwestern Medical Center, Dallas.
The study was presented at the American Academy of Neurology Sports Concussion Virtual Conference, held online July 31 to Aug. 1.
Learning disorders affected scores
The researchers analyzed data from participants aged 10-18 years who were enrolled in the North Texas Concussion Registry (ConTex). Participants had been diagnosed with a concussion that was sustained within 30 days of enrollment. The researchers investigated whether there were differences between patients who had no history of learning disorders and those with a history of dyslexia and/or ADD/ADHD with regard to results of clinical testing following concussion.
Of the 1,298 individuals in the study, 58 had been diagnosed with dyslexia, 158 had been diagnosed with ADD/ADHD, and 35 had been diagnosed with both conditions. There was no difference in age, time since injury, or history of concussion between those with learning disorders and those without, but there were more male patients in the ADD/ADHD group.
Results showed that in the dyslexia group, mean time was slower (P = .011), and there was an increase in error scores on the King-Devick (KD) test (P = .028). That test assesses eye movements and involves the rapid naming of numbers that are spaced differently. In addition, those with ADD/ADHD had significantly higher impulse control scores (P = .007) on the ImPACT series of tests, which are commonly used in the evaluation of concussion. Participants with both dyslexia and ADHD demonstrated slower KD times (P = .009) and had higher depression scores and anxiety scores.
Dr. Stokes noted that a limiting factor of the study was that baseline scores were not available. “It is possible that kids with ADD have less impulse control even at baseline, and this would need to be taken into account,” he said. “You may perhaps also expect someone with dyslexia to have a worse score on the KD tests, so we need more data on how these scores are affected from baseline in these individuals. But our results show that when evaluating kids pre- or post concussion, it is important to know about learning disorders, as this will affect how we interpret the data.”
At 3-month follow-up, there were no longer significant differences in anxiety and depression scores for those with and those without learning disorders. “This suggests anxiety and depression may well be worse temporarily after concussion for those with ADD/ADHD but gets better with time,” Dr. Stokes said.
Follow-up data were not available for the other cognitive tests.
Are recovery times longer?
Asked whether young people with these learning disorders needed a longer time to recover after concussion, Dr. Stokes said: “That is a million-dollar question. Studies so far on this have shown conflicting results. Our results add to a growing body of literature on this.” He stressed that it is important to include anxiety and depression scores on both baseline and postconcussion tests. “People don’t tend to think of these symptoms as being associated with concussion, but they are actually very prominent in this situation,” he noted. “Our results suggest that individuals with ADHD may be more prone to anxiety and depression, and a blow to the head may tip them more into these symptoms.”
Discussing the study at a virtual press conference as part of the AAN Sports Concussion meeting, the codirector of the meeting, David Dodick, MD, Mayo Clinic, Scottsdale, Ariz., said: “This is a very interesting and important study which suggests there are differences between adolescents with a history of dyslexia/ADHD and those without these conditions in performance in concussion tests. Understanding the differences in these groups will help health care providers in evaluating these athletes and assisting in counseling them and their families with regard to their risk of injury.
“It is important to recognize that athletes with ADHD, whether or not they are on medication, may take longer to recover from a concussion,” Dr. Dodick added. They also exhibit greater reductions in cognitive skills and visual motor speed regarding hand-eye coordination, he said. There is an increase in the severity of symptoms. “Symptoms that exist in both groups tend to more severe in those individuals with ADHD,” he noted.
“Ascertaining the presence or absence of ADHD or dyslexia in those who are participating in sport is important, especially when trying to interpret the results of baseline testing, the results of postinjury testing, decisions on when to return to play, and assessing for individuals and their families the risk of long-term repeat concussions and adverse outcomes,” he concluded.
The other codirector of the AAN meeting, Brian Hainline, MD, chief medical officer of the National Collegiate Athletic Association, added: “It appears that athletes with ADHD may suffer more with concussion and have a longer recovery time. This can inform our decision making and help these individuals to understand that they are at higher risk.”
Dr. Hainline said this raises another important point: “Concussion is not a homogeneous entity. It is a brain injury that can manifest in multiple parts of the brain, and the way the brain is from a premorbid or comorbid point of view can influence the manifestation of concussion as well,” he said. “All these things need to be taken into account.”
Attentional deficit may itself make an individual more susceptible to sustaining an injury in the first place, he said. “All of this is an evolving body of research which is helping clinicians to make better-informed decisions for athletes who may manifest differently.”
A version of this article originally appeared on Medscape.com.
, a new study shows.
“Our results suggest kids with certain learning disorders may respond differently to concussion tests, and this needs to be taken into account when advising on recovery times and when they can return to sport,” said lead author Mathew Stokes, MD. Dr. Stokes is assistant professor of pediatrics and neurology/neurotherapeutics at the University of Texas–Southwestern Medical Center, Dallas.
The study was presented at the American Academy of Neurology Sports Concussion Virtual Conference, held online July 31 to Aug. 1.
Learning disorders affected scores
The researchers analyzed data from participants aged 10-18 years who were enrolled in the North Texas Concussion Registry (ConTex). Participants had been diagnosed with a concussion that was sustained within 30 days of enrollment. The researchers investigated whether there were differences between patients who had no history of learning disorders and those with a history of dyslexia and/or ADD/ADHD with regard to results of clinical testing following concussion.
Of the 1,298 individuals in the study, 58 had been diagnosed with dyslexia, 158 had been diagnosed with ADD/ADHD, and 35 had been diagnosed with both conditions. There was no difference in age, time since injury, or history of concussion between those with learning disorders and those without, but there were more male patients in the ADD/ADHD group.
Results showed that in the dyslexia group, mean time was slower (P = .011), and there was an increase in error scores on the King-Devick (KD) test (P = .028). That test assesses eye movements and involves the rapid naming of numbers that are spaced differently. In addition, those with ADD/ADHD had significantly higher impulse control scores (P = .007) on the ImPACT series of tests, which are commonly used in the evaluation of concussion. Participants with both dyslexia and ADHD demonstrated slower KD times (P = .009) and had higher depression scores and anxiety scores.
Dr. Stokes noted that a limiting factor of the study was that baseline scores were not available. “It is possible that kids with ADD have less impulse control even at baseline, and this would need to be taken into account,” he said. “You may perhaps also expect someone with dyslexia to have a worse score on the KD tests, so we need more data on how these scores are affected from baseline in these individuals. But our results show that when evaluating kids pre- or post concussion, it is important to know about learning disorders, as this will affect how we interpret the data.”
At 3-month follow-up, there were no longer significant differences in anxiety and depression scores for those with and those without learning disorders. “This suggests anxiety and depression may well be worse temporarily after concussion for those with ADD/ADHD but gets better with time,” Dr. Stokes said.
Follow-up data were not available for the other cognitive tests.
Are recovery times longer?
Asked whether young people with these learning disorders needed a longer time to recover after concussion, Dr. Stokes said: “That is a million-dollar question. Studies so far on this have shown conflicting results. Our results add to a growing body of literature on this.” He stressed that it is important to include anxiety and depression scores on both baseline and postconcussion tests. “People don’t tend to think of these symptoms as being associated with concussion, but they are actually very prominent in this situation,” he noted. “Our results suggest that individuals with ADHD may be more prone to anxiety and depression, and a blow to the head may tip them more into these symptoms.”
Discussing the study at a virtual press conference as part of the AAN Sports Concussion meeting, the codirector of the meeting, David Dodick, MD, Mayo Clinic, Scottsdale, Ariz., said: “This is a very interesting and important study which suggests there are differences between adolescents with a history of dyslexia/ADHD and those without these conditions in performance in concussion tests. Understanding the differences in these groups will help health care providers in evaluating these athletes and assisting in counseling them and their families with regard to their risk of injury.
“It is important to recognize that athletes with ADHD, whether or not they are on medication, may take longer to recover from a concussion,” Dr. Dodick added. They also exhibit greater reductions in cognitive skills and visual motor speed regarding hand-eye coordination, he said. There is an increase in the severity of symptoms. “Symptoms that exist in both groups tend to more severe in those individuals with ADHD,” he noted.
“Ascertaining the presence or absence of ADHD or dyslexia in those who are participating in sport is important, especially when trying to interpret the results of baseline testing, the results of postinjury testing, decisions on when to return to play, and assessing for individuals and their families the risk of long-term repeat concussions and adverse outcomes,” he concluded.
The other codirector of the AAN meeting, Brian Hainline, MD, chief medical officer of the National Collegiate Athletic Association, added: “It appears that athletes with ADHD may suffer more with concussion and have a longer recovery time. This can inform our decision making and help these individuals to understand that they are at higher risk.”
Dr. Hainline said this raises another important point: “Concussion is not a homogeneous entity. It is a brain injury that can manifest in multiple parts of the brain, and the way the brain is from a premorbid or comorbid point of view can influence the manifestation of concussion as well,” he said. “All these things need to be taken into account.”
Attentional deficit may itself make an individual more susceptible to sustaining an injury in the first place, he said. “All of this is an evolving body of research which is helping clinicians to make better-informed decisions for athletes who may manifest differently.”
A version of this article originally appeared on Medscape.com.
, a new study shows.
“Our results suggest kids with certain learning disorders may respond differently to concussion tests, and this needs to be taken into account when advising on recovery times and when they can return to sport,” said lead author Mathew Stokes, MD. Dr. Stokes is assistant professor of pediatrics and neurology/neurotherapeutics at the University of Texas–Southwestern Medical Center, Dallas.
The study was presented at the American Academy of Neurology Sports Concussion Virtual Conference, held online July 31 to Aug. 1.
Learning disorders affected scores
The researchers analyzed data from participants aged 10-18 years who were enrolled in the North Texas Concussion Registry (ConTex). Participants had been diagnosed with a concussion that was sustained within 30 days of enrollment. The researchers investigated whether there were differences between patients who had no history of learning disorders and those with a history of dyslexia and/or ADD/ADHD with regard to results of clinical testing following concussion.
Of the 1,298 individuals in the study, 58 had been diagnosed with dyslexia, 158 had been diagnosed with ADD/ADHD, and 35 had been diagnosed with both conditions. There was no difference in age, time since injury, or history of concussion between those with learning disorders and those without, but there were more male patients in the ADD/ADHD group.
Results showed that in the dyslexia group, mean time was slower (P = .011), and there was an increase in error scores on the King-Devick (KD) test (P = .028). That test assesses eye movements and involves the rapid naming of numbers that are spaced differently. In addition, those with ADD/ADHD had significantly higher impulse control scores (P = .007) on the ImPACT series of tests, which are commonly used in the evaluation of concussion. Participants with both dyslexia and ADHD demonstrated slower KD times (P = .009) and had higher depression scores and anxiety scores.
Dr. Stokes noted that a limiting factor of the study was that baseline scores were not available. “It is possible that kids with ADD have less impulse control even at baseline, and this would need to be taken into account,” he said. “You may perhaps also expect someone with dyslexia to have a worse score on the KD tests, so we need more data on how these scores are affected from baseline in these individuals. But our results show that when evaluating kids pre- or post concussion, it is important to know about learning disorders, as this will affect how we interpret the data.”
At 3-month follow-up, there were no longer significant differences in anxiety and depression scores for those with and those without learning disorders. “This suggests anxiety and depression may well be worse temporarily after concussion for those with ADD/ADHD but gets better with time,” Dr. Stokes said.
Follow-up data were not available for the other cognitive tests.
Are recovery times longer?
Asked whether young people with these learning disorders needed a longer time to recover after concussion, Dr. Stokes said: “That is a million-dollar question. Studies so far on this have shown conflicting results. Our results add to a growing body of literature on this.” He stressed that it is important to include anxiety and depression scores on both baseline and postconcussion tests. “People don’t tend to think of these symptoms as being associated with concussion, but they are actually very prominent in this situation,” he noted. “Our results suggest that individuals with ADHD may be more prone to anxiety and depression, and a blow to the head may tip them more into these symptoms.”
Discussing the study at a virtual press conference as part of the AAN Sports Concussion meeting, the codirector of the meeting, David Dodick, MD, Mayo Clinic, Scottsdale, Ariz., said: “This is a very interesting and important study which suggests there are differences between adolescents with a history of dyslexia/ADHD and those without these conditions in performance in concussion tests. Understanding the differences in these groups will help health care providers in evaluating these athletes and assisting in counseling them and their families with regard to their risk of injury.
“It is important to recognize that athletes with ADHD, whether or not they are on medication, may take longer to recover from a concussion,” Dr. Dodick added. They also exhibit greater reductions in cognitive skills and visual motor speed regarding hand-eye coordination, he said. There is an increase in the severity of symptoms. “Symptoms that exist in both groups tend to more severe in those individuals with ADHD,” he noted.
“Ascertaining the presence or absence of ADHD or dyslexia in those who are participating in sport is important, especially when trying to interpret the results of baseline testing, the results of postinjury testing, decisions on when to return to play, and assessing for individuals and their families the risk of long-term repeat concussions and adverse outcomes,” he concluded.
The other codirector of the AAN meeting, Brian Hainline, MD, chief medical officer of the National Collegiate Athletic Association, added: “It appears that athletes with ADHD may suffer more with concussion and have a longer recovery time. This can inform our decision making and help these individuals to understand that they are at higher risk.”
Dr. Hainline said this raises another important point: “Concussion is not a homogeneous entity. It is a brain injury that can manifest in multiple parts of the brain, and the way the brain is from a premorbid or comorbid point of view can influence the manifestation of concussion as well,” he said. “All these things need to be taken into account.”
Attentional deficit may itself make an individual more susceptible to sustaining an injury in the first place, he said. “All of this is an evolving body of research which is helping clinicians to make better-informed decisions for athletes who may manifest differently.”
A version of this article originally appeared on Medscape.com.
From AAN Sports Concussion Conference
NBA star Mason Plumlee on COVID and life inside the Orlando ‘bubble’
Editor’s Note: This transcript from the August 20 episode of the Blood & Cancer podcast has been edited for clarity. Click this link to listen to the full episode.
David Henry, MD: Welcome to this Blood & Cancer podcast. I’m your host, Dr. David Henry. This podcast airs on Thursday morning each week. This interview and others are archived with show notes from our residents at Pennsylvania Hospital at this link.
Each week we interview key opinion leaders involved in various aspects of blood and cancer. Mason was a first round pick in the NBA, a gold medalist for the U.S. men’s national team, and NBA All-Rookie first team honoree. He’s one of the top playmaking forwards in the country, if not the world, in my opinion. In his four-year college career at Duke University, he helped lead the Blue Devils to a National Collegiate Athletic Association (NCAA) championship and twice earned All-America first team academic honors at Duke. So he’s not just a basketball star, but an academic star as well. Mason, thanks so much for taking some time out from the bubble in Florida to talk with us today.
Mason Plumlee: Thanks for having me on. I’m happy to be here.
Henry: Beginning in March, the NBA didn’t know what to do about the COVID pandemic but finally decided to put you professional players in a ‘bubble.’ What did you have to go through to get there? You, your teammates, coaches, trainers, etc. And what’s the ongoing plan to be sure you continue to be safe?
Plumlee: Back to when the season shut down in March, the NBA shut down the practice facilities at the same time. Most people went home. I went back to Indiana. And then, as the idea of this bubble came up and the NBA formalized a plan to start the season again, players started to go back to market. I went back to Denver and was working out there.
About two weeks before we were scheduled to arrive in Orlando, they started testing us every other day. They used the deep nasal swab as well as the throat swab. But they were also taking two to three blood tests in that time period. You needed a certain number of consecutive negative tests before they would allow you to fly on the team plane down to Orlando. So there was an incredible amount of testing in the market. Once you got to Orlando, you went into a 48-hour quarantine. You had to have two negative tests with 48 hours between them before you could leave your hotel room.
Since then, it’s been quite strict down here. And although it’s annoying in a lot of ways, I think it’s one of the reasons our league has been able to pull this off. We’ve had no positive tests within the bubble and we are tested every day. A company called BioReference Laboratories has a setup in one of the meeting rooms here, and it’s like clockwork—we go in, we get our tests. One of my teammates missed a test and they made him stay in his room until he could get another test and get the results, so he missed a game because of that.
Henry: During this bubble time, no one has tested positive—players, coaches, staff?
Plumlee: Correct.
Henry: That’s incredible, and it’s allowed those of us who want to watch the NBA and those of you who are in it professionally to continue the sport. It must be a real nuisance for you and your family and friends, because no one can visit you, right?
Plumlee: Right. There’s no visitation. We had one false positive. It was our media relations person and the actions they took when that positive test came in -- they quarantined him in his room and interviewed everybody he had talked to; they tested anyone who had any interaction with him and those people had to go into quarantine. They’re on top of things down here. In addition to the testing, we each have a pulse oximeter and a thermometer, and we use these to check in everyday on an app. So, they’re getting all the insight they need. After the first round of the playoffs, they’re going to open the bubble to friends and family, but those friends and family will be subject to all the same protocols that we were coming in and once they’re here as well.
Henry: I’m sure you’ve heard about the Broadway star [Nick Cordero] who was healthy and suddenly got sick, lost a leg, and then lost his life. There have been some heart attacks that surprised us. Have your colleagues—players, coaches, etc.—been worried? Or are they thinking, what’s the big deal? Has the sense of how serious this is permeated through this sport?
Plumlee: The NBA is one of the groups that has heightened the understanding and awareness of this by shutting down. I think a lot of people were moving forward as is, and then, when the NBA decided to cancel the season, it let the world know, look, this is to be taken seriously.
Henry: A couple of players did test positive early on.
Plumlee: Exactly. A couple of people tested positive. I think at the outset, the unknown is always scarier. As we’ve learned more about the virus, the guys have become more comfortable. You know, I tested positive back in March. At the time, a loss of taste and smell was not a reported symptom.
Henry: And you had that?
Plumlee: I did have that, but I didn’t know what to think. More research has come out and we have a better understanding of that. I think most of the players are comfortable with the virus. We’re at a time in our lives where we’re healthy, we’re active, and we should be able to fight it off. We know the numbers for our age group. Even still, I think nobody wants to get it. Nobody wants to have to go through it. So why chance it?
Henry: Hats off to you and your sport. Other sports such as Major League Baseball haven’t been quite so successful. Of course, they’re wrestling with the players testing positive, and this has stopped games this season.
I was looking over your background prior to the interview and learned that your mother and father have been involved in the medical arena. Can you tell us about that and how it’s rubbed off on you?
Plumlee: Definitely. My mom is a pharmacist, so I spent a lot of time as a kid going to see her at work. And my dad is general counsel for an orthopedic company. My hometown is Warsaw, Ind. Some people refer to it as the “Orthopedic Capital of the World.” Zimmer Biomet is headquartered there. DePuy Synthes is there. Medtronic has offices there, as well as a lot of cottage businesses that support the orthopedic industry. In my hometown, the rock star was Dane Miller, who founded Biomet. I have no formal education in medicine or health care, but I’ve seen the impact of it. From my parents and some cousins, uncles who are doctors and surgeons, it’s been interesting to see their work and learn about what’s the latest and greatest in health care.
Henry: What’s so nice about you in particular is, with that background of interests from your family and your celebrity and accomplishments in professional basketball, you have used that to explore and promote ways to make progress in health care and help others who are less fortunate. For example, you’re involved in a telehealth platform for all-in-one practice management; affordable telehealth for pediatrics; health benefits for small businesses; prior authorization—if you can help with prior authorization, we will be in the stands for you at every game because it’s the bane of our existence; radiotherapy; and probably from mom’s background, pharmacy benefit management. Pick any of those you’d like to talk about, and tell us about your involvement and how it’s going.
Plumlee: My ticket into the arena is investment. Nobody’s calling me, asking for my expertise. But a lot of these visionary founders need financial support, and that’s where I get involved. Then also, with the celebrity angle from being an athlete, sometimes you can open doors for a start-up founder that they may not be able to open themselves.
I’m happy to speak about any of those companies. I am excited about the relaxed regulation that’s come from the pandemic; not that it’s like the Wild West out here, but I think it has allowed companies to implement solutions or think about problems in a way that they couldn’t before the pandemic. Take the prior authorization play, for example, and a company called Banjo Health, with one of my favorite founders, a guy named Saar Mahna. Medicare mandates that you turn around prior authorizations within three days. This company has an artificial intelligence and machine-learning play on prior authorizations that can deliver on that.
So efficiencies, things that increase access or affordability, better outcomes, those are the things that attract me. I lean on other people for the due diligence. The pediatric play that you referenced is a company called Blueberry Pediatrics. You have a monthly subscription for $15 that can be reimbursed by Medicaid. They send two devices to your home—an otoscope and an oximeter. The company is live in Florida right now, and it’s diverting a ton of emergency room (ER) visits. From home, for $15 a month, a mom has an otoscope and an oximeter, and she can chat or video conference with a pediatrician. There’s no additional fee. So that’s saving everyone time and saving the system money. Those are the kinds of things I’m attracted to.
Henry: You’ve touched on a couple of hot button issues for us. In oncology, unfortunately, most of our patients have pain. I am mystified every time I try to get a narcotic or a strong painkiller for a patient on a Friday night and I’m told it requires prior authorization and they’ll open up again on Monday. Well, that’s insane. These patients need something right away. So if you have a special interest in helping all of us with prior authorization, the artificial intelligence is a no brainer. If this kind of computer algorithm could happen overnight, that would be wonderful.
You mentioned the ER. Many people go to the ER as a default. They don’t know what else to do. In the COVID era, we’re trying to dial that down because we want to be able to see the sickest and have the non-sick get care elsewhere. If this particular person or people don’t know what to do, they go to the ER, it costs money, takes a lot of time, and others who may be sick are diverted from care. Families worry terribly about their children, so a device for mom and access to a pediatrician for $15 a month is another wonderful idea. These are both very interesting. Another company is in the pharmacy benefit management (PBM) space. Anything you could say about how that works?
Plumlee: I can give an overview of how I look at this as an investor in the PBM space. Three companies control about 75% of a multibillion dollar market. Several initiatives have been pursued politically to provide transparent pricing between these PBMs and pharmaceutical companies, and a lot of people are pointing fingers, but ultimately, drug prices just keep going up. Everybody knows it.
A couple of start-up founders are really set on bringing a competitive marketplace back to the pharmacy benefit manager. As an investor, when you see three people controlling a market, and you have small or medium PBMs that depend on aggregators to get competitive pricing with those big three, you get interested. It’s an interesting industry. My feeling is that somebody is going to disrupt it and bring competition back to that space. Ultimately, drug prices will come down because it’s not sustainable. The insurance companies just accommodate whatever the drug pricing is. If the drug prices go up, your premiums go up. I think these new companies will be level-setting.
Henry: In my world of oncology, we’re just a little more than halfway through 2020 and we’ve had five, six, seven new drugs approved. They all will be very expensive. One of the nicer things that’s happening and may help to tamp this down involves biosimilars. When you go to CVS or Rite Aid, you go down the aspirin aisle and see the generics, and they’re identical to the brand name aspirin. Well, these very complex molecules we used to treat cancer are antibodies or proteins, and they’re made in nature’s factories called cells. They’re not identical to the brand name drugs, but they’re called biosimilars. They work exactly the same as the branded drugs with exactly the same safety–our U.S. FDA has done a nice job of vetting that, to be sure. X, Y, Z Company has copied the brand drug after the patent expires. They were hoping for about a 30% discount in price but we’re seeing more like 15%. Nothing’s ever easy. So you make a very good point. This is not sustainable and the competition will be wonderful to tamp down these prices.
Plumlee: My hope is that those biosimilars and generics get placement in these formularies because the formularies are what’s valuable to the drug manufacturers. But they have to accommodate what the Big Three want in the PBM space. To me, making things affordable and accessible is what a lot of these startups are trying to do. And hopefully they will win.
Henry: What have you been going through, in terms of COVID? Have you recovered fully? Have your taste and smell returned, and you’re back to normal?
Plumlee: I’m all good. It caught me off guard but the symptoms weren’t too intense. For me, it was less than a flu, but more than a cold. And I’m all good today.
Henry: We’re so glad and wish you the best of luck.
Dr. Henry is a clinical professor of medicine at the University of Pennsylvania and vice chairman of the department of medicine at Pennsylvania Hospital in Philadelphia and the host of the Blood & Cancer podcast. He has no relevant financial conflicts.
Mr. Plumlee is a board advisor to both Formsense and the Prysm Institute and a board observer with Voiceitt.
Editor’s Note: This transcript from the August 20 episode of the Blood & Cancer podcast has been edited for clarity. Click this link to listen to the full episode.
David Henry, MD: Welcome to this Blood & Cancer podcast. I’m your host, Dr. David Henry. This podcast airs on Thursday morning each week. This interview and others are archived with show notes from our residents at Pennsylvania Hospital at this link.
Each week we interview key opinion leaders involved in various aspects of blood and cancer. Mason was a first round pick in the NBA, a gold medalist for the U.S. men’s national team, and NBA All-Rookie first team honoree. He’s one of the top playmaking forwards in the country, if not the world, in my opinion. In his four-year college career at Duke University, he helped lead the Blue Devils to a National Collegiate Athletic Association (NCAA) championship and twice earned All-America first team academic honors at Duke. So he’s not just a basketball star, but an academic star as well. Mason, thanks so much for taking some time out from the bubble in Florida to talk with us today.
Mason Plumlee: Thanks for having me on. I’m happy to be here.
Henry: Beginning in March, the NBA didn’t know what to do about the COVID pandemic but finally decided to put you professional players in a ‘bubble.’ What did you have to go through to get there? You, your teammates, coaches, trainers, etc. And what’s the ongoing plan to be sure you continue to be safe?
Plumlee: Back to when the season shut down in March, the NBA shut down the practice facilities at the same time. Most people went home. I went back to Indiana. And then, as the idea of this bubble came up and the NBA formalized a plan to start the season again, players started to go back to market. I went back to Denver and was working out there.
About two weeks before we were scheduled to arrive in Orlando, they started testing us every other day. They used the deep nasal swab as well as the throat swab. But they were also taking two to three blood tests in that time period. You needed a certain number of consecutive negative tests before they would allow you to fly on the team plane down to Orlando. So there was an incredible amount of testing in the market. Once you got to Orlando, you went into a 48-hour quarantine. You had to have two negative tests with 48 hours between them before you could leave your hotel room.
Since then, it’s been quite strict down here. And although it’s annoying in a lot of ways, I think it’s one of the reasons our league has been able to pull this off. We’ve had no positive tests within the bubble and we are tested every day. A company called BioReference Laboratories has a setup in one of the meeting rooms here, and it’s like clockwork—we go in, we get our tests. One of my teammates missed a test and they made him stay in his room until he could get another test and get the results, so he missed a game because of that.
Henry: During this bubble time, no one has tested positive—players, coaches, staff?
Plumlee: Correct.
Henry: That’s incredible, and it’s allowed those of us who want to watch the NBA and those of you who are in it professionally to continue the sport. It must be a real nuisance for you and your family and friends, because no one can visit you, right?
Plumlee: Right. There’s no visitation. We had one false positive. It was our media relations person and the actions they took when that positive test came in -- they quarantined him in his room and interviewed everybody he had talked to; they tested anyone who had any interaction with him and those people had to go into quarantine. They’re on top of things down here. In addition to the testing, we each have a pulse oximeter and a thermometer, and we use these to check in everyday on an app. So, they’re getting all the insight they need. After the first round of the playoffs, they’re going to open the bubble to friends and family, but those friends and family will be subject to all the same protocols that we were coming in and once they’re here as well.
Henry: I’m sure you’ve heard about the Broadway star [Nick Cordero] who was healthy and suddenly got sick, lost a leg, and then lost his life. There have been some heart attacks that surprised us. Have your colleagues—players, coaches, etc.—been worried? Or are they thinking, what’s the big deal? Has the sense of how serious this is permeated through this sport?
Plumlee: The NBA is one of the groups that has heightened the understanding and awareness of this by shutting down. I think a lot of people were moving forward as is, and then, when the NBA decided to cancel the season, it let the world know, look, this is to be taken seriously.
Henry: A couple of players did test positive early on.
Plumlee: Exactly. A couple of people tested positive. I think at the outset, the unknown is always scarier. As we’ve learned more about the virus, the guys have become more comfortable. You know, I tested positive back in March. At the time, a loss of taste and smell was not a reported symptom.
Henry: And you had that?
Plumlee: I did have that, but I didn’t know what to think. More research has come out and we have a better understanding of that. I think most of the players are comfortable with the virus. We’re at a time in our lives where we’re healthy, we’re active, and we should be able to fight it off. We know the numbers for our age group. Even still, I think nobody wants to get it. Nobody wants to have to go through it. So why chance it?
Henry: Hats off to you and your sport. Other sports such as Major League Baseball haven’t been quite so successful. Of course, they’re wrestling with the players testing positive, and this has stopped games this season.
I was looking over your background prior to the interview and learned that your mother and father have been involved in the medical arena. Can you tell us about that and how it’s rubbed off on you?
Plumlee: Definitely. My mom is a pharmacist, so I spent a lot of time as a kid going to see her at work. And my dad is general counsel for an orthopedic company. My hometown is Warsaw, Ind. Some people refer to it as the “Orthopedic Capital of the World.” Zimmer Biomet is headquartered there. DePuy Synthes is there. Medtronic has offices there, as well as a lot of cottage businesses that support the orthopedic industry. In my hometown, the rock star was Dane Miller, who founded Biomet. I have no formal education in medicine or health care, but I’ve seen the impact of it. From my parents and some cousins, uncles who are doctors and surgeons, it’s been interesting to see their work and learn about what’s the latest and greatest in health care.
Henry: What’s so nice about you in particular is, with that background of interests from your family and your celebrity and accomplishments in professional basketball, you have used that to explore and promote ways to make progress in health care and help others who are less fortunate. For example, you’re involved in a telehealth platform for all-in-one practice management; affordable telehealth for pediatrics; health benefits for small businesses; prior authorization—if you can help with prior authorization, we will be in the stands for you at every game because it’s the bane of our existence; radiotherapy; and probably from mom’s background, pharmacy benefit management. Pick any of those you’d like to talk about, and tell us about your involvement and how it’s going.
Plumlee: My ticket into the arena is investment. Nobody’s calling me, asking for my expertise. But a lot of these visionary founders need financial support, and that’s where I get involved. Then also, with the celebrity angle from being an athlete, sometimes you can open doors for a start-up founder that they may not be able to open themselves.
I’m happy to speak about any of those companies. I am excited about the relaxed regulation that’s come from the pandemic; not that it’s like the Wild West out here, but I think it has allowed companies to implement solutions or think about problems in a way that they couldn’t before the pandemic. Take the prior authorization play, for example, and a company called Banjo Health, with one of my favorite founders, a guy named Saar Mahna. Medicare mandates that you turn around prior authorizations within three days. This company has an artificial intelligence and machine-learning play on prior authorizations that can deliver on that.
So efficiencies, things that increase access or affordability, better outcomes, those are the things that attract me. I lean on other people for the due diligence. The pediatric play that you referenced is a company called Blueberry Pediatrics. You have a monthly subscription for $15 that can be reimbursed by Medicaid. They send two devices to your home—an otoscope and an oximeter. The company is live in Florida right now, and it’s diverting a ton of emergency room (ER) visits. From home, for $15 a month, a mom has an otoscope and an oximeter, and she can chat or video conference with a pediatrician. There’s no additional fee. So that’s saving everyone time and saving the system money. Those are the kinds of things I’m attracted to.
Henry: You’ve touched on a couple of hot button issues for us. In oncology, unfortunately, most of our patients have pain. I am mystified every time I try to get a narcotic or a strong painkiller for a patient on a Friday night and I’m told it requires prior authorization and they’ll open up again on Monday. Well, that’s insane. These patients need something right away. So if you have a special interest in helping all of us with prior authorization, the artificial intelligence is a no brainer. If this kind of computer algorithm could happen overnight, that would be wonderful.
You mentioned the ER. Many people go to the ER as a default. They don’t know what else to do. In the COVID era, we’re trying to dial that down because we want to be able to see the sickest and have the non-sick get care elsewhere. If this particular person or people don’t know what to do, they go to the ER, it costs money, takes a lot of time, and others who may be sick are diverted from care. Families worry terribly about their children, so a device for mom and access to a pediatrician for $15 a month is another wonderful idea. These are both very interesting. Another company is in the pharmacy benefit management (PBM) space. Anything you could say about how that works?
Plumlee: I can give an overview of how I look at this as an investor in the PBM space. Three companies control about 75% of a multibillion dollar market. Several initiatives have been pursued politically to provide transparent pricing between these PBMs and pharmaceutical companies, and a lot of people are pointing fingers, but ultimately, drug prices just keep going up. Everybody knows it.
A couple of start-up founders are really set on bringing a competitive marketplace back to the pharmacy benefit manager. As an investor, when you see three people controlling a market, and you have small or medium PBMs that depend on aggregators to get competitive pricing with those big three, you get interested. It’s an interesting industry. My feeling is that somebody is going to disrupt it and bring competition back to that space. Ultimately, drug prices will come down because it’s not sustainable. The insurance companies just accommodate whatever the drug pricing is. If the drug prices go up, your premiums go up. I think these new companies will be level-setting.
Henry: In my world of oncology, we’re just a little more than halfway through 2020 and we’ve had five, six, seven new drugs approved. They all will be very expensive. One of the nicer things that’s happening and may help to tamp this down involves biosimilars. When you go to CVS or Rite Aid, you go down the aspirin aisle and see the generics, and they’re identical to the brand name aspirin. Well, these very complex molecules we used to treat cancer are antibodies or proteins, and they’re made in nature’s factories called cells. They’re not identical to the brand name drugs, but they’re called biosimilars. They work exactly the same as the branded drugs with exactly the same safety–our U.S. FDA has done a nice job of vetting that, to be sure. X, Y, Z Company has copied the brand drug after the patent expires. They were hoping for about a 30% discount in price but we’re seeing more like 15%. Nothing’s ever easy. So you make a very good point. This is not sustainable and the competition will be wonderful to tamp down these prices.
Plumlee: My hope is that those biosimilars and generics get placement in these formularies because the formularies are what’s valuable to the drug manufacturers. But they have to accommodate what the Big Three want in the PBM space. To me, making things affordable and accessible is what a lot of these startups are trying to do. And hopefully they will win.
Henry: What have you been going through, in terms of COVID? Have you recovered fully? Have your taste and smell returned, and you’re back to normal?
Plumlee: I’m all good. It caught me off guard but the symptoms weren’t too intense. For me, it was less than a flu, but more than a cold. And I’m all good today.
Henry: We’re so glad and wish you the best of luck.
Dr. Henry is a clinical professor of medicine at the University of Pennsylvania and vice chairman of the department of medicine at Pennsylvania Hospital in Philadelphia and the host of the Blood & Cancer podcast. He has no relevant financial conflicts.
Mr. Plumlee is a board advisor to both Formsense and the Prysm Institute and a board observer with Voiceitt.
Editor’s Note: This transcript from the August 20 episode of the Blood & Cancer podcast has been edited for clarity. Click this link to listen to the full episode.
David Henry, MD: Welcome to this Blood & Cancer podcast. I’m your host, Dr. David Henry. This podcast airs on Thursday morning each week. This interview and others are archived with show notes from our residents at Pennsylvania Hospital at this link.
Each week we interview key opinion leaders involved in various aspects of blood and cancer. Mason was a first round pick in the NBA, a gold medalist for the U.S. men’s national team, and NBA All-Rookie first team honoree. He’s one of the top playmaking forwards in the country, if not the world, in my opinion. In his four-year college career at Duke University, he helped lead the Blue Devils to a National Collegiate Athletic Association (NCAA) championship and twice earned All-America first team academic honors at Duke. So he’s not just a basketball star, but an academic star as well. Mason, thanks so much for taking some time out from the bubble in Florida to talk with us today.
Mason Plumlee: Thanks for having me on. I’m happy to be here.
Henry: Beginning in March, the NBA didn’t know what to do about the COVID pandemic but finally decided to put you professional players in a ‘bubble.’ What did you have to go through to get there? You, your teammates, coaches, trainers, etc. And what’s the ongoing plan to be sure you continue to be safe?
Plumlee: Back to when the season shut down in March, the NBA shut down the practice facilities at the same time. Most people went home. I went back to Indiana. And then, as the idea of this bubble came up and the NBA formalized a plan to start the season again, players started to go back to market. I went back to Denver and was working out there.
About two weeks before we were scheduled to arrive in Orlando, they started testing us every other day. They used the deep nasal swab as well as the throat swab. But they were also taking two to three blood tests in that time period. You needed a certain number of consecutive negative tests before they would allow you to fly on the team plane down to Orlando. So there was an incredible amount of testing in the market. Once you got to Orlando, you went into a 48-hour quarantine. You had to have two negative tests with 48 hours between them before you could leave your hotel room.
Since then, it’s been quite strict down here. And although it’s annoying in a lot of ways, I think it’s one of the reasons our league has been able to pull this off. We’ve had no positive tests within the bubble and we are tested every day. A company called BioReference Laboratories has a setup in one of the meeting rooms here, and it’s like clockwork—we go in, we get our tests. One of my teammates missed a test and they made him stay in his room until he could get another test and get the results, so he missed a game because of that.
Henry: During this bubble time, no one has tested positive—players, coaches, staff?
Plumlee: Correct.
Henry: That’s incredible, and it’s allowed those of us who want to watch the NBA and those of you who are in it professionally to continue the sport. It must be a real nuisance for you and your family and friends, because no one can visit you, right?
Plumlee: Right. There’s no visitation. We had one false positive. It was our media relations person and the actions they took when that positive test came in -- they quarantined him in his room and interviewed everybody he had talked to; they tested anyone who had any interaction with him and those people had to go into quarantine. They’re on top of things down here. In addition to the testing, we each have a pulse oximeter and a thermometer, and we use these to check in everyday on an app. So, they’re getting all the insight they need. After the first round of the playoffs, they’re going to open the bubble to friends and family, but those friends and family will be subject to all the same protocols that we were coming in and once they’re here as well.
Henry: I’m sure you’ve heard about the Broadway star [Nick Cordero] who was healthy and suddenly got sick, lost a leg, and then lost his life. There have been some heart attacks that surprised us. Have your colleagues—players, coaches, etc.—been worried? Or are they thinking, what’s the big deal? Has the sense of how serious this is permeated through this sport?
Plumlee: The NBA is one of the groups that has heightened the understanding and awareness of this by shutting down. I think a lot of people were moving forward as is, and then, when the NBA decided to cancel the season, it let the world know, look, this is to be taken seriously.
Henry: A couple of players did test positive early on.
Plumlee: Exactly. A couple of people tested positive. I think at the outset, the unknown is always scarier. As we’ve learned more about the virus, the guys have become more comfortable. You know, I tested positive back in March. At the time, a loss of taste and smell was not a reported symptom.
Henry: And you had that?
Plumlee: I did have that, but I didn’t know what to think. More research has come out and we have a better understanding of that. I think most of the players are comfortable with the virus. We’re at a time in our lives where we’re healthy, we’re active, and we should be able to fight it off. We know the numbers for our age group. Even still, I think nobody wants to get it. Nobody wants to have to go through it. So why chance it?
Henry: Hats off to you and your sport. Other sports such as Major League Baseball haven’t been quite so successful. Of course, they’re wrestling with the players testing positive, and this has stopped games this season.
I was looking over your background prior to the interview and learned that your mother and father have been involved in the medical arena. Can you tell us about that and how it’s rubbed off on you?
Plumlee: Definitely. My mom is a pharmacist, so I spent a lot of time as a kid going to see her at work. And my dad is general counsel for an orthopedic company. My hometown is Warsaw, Ind. Some people refer to it as the “Orthopedic Capital of the World.” Zimmer Biomet is headquartered there. DePuy Synthes is there. Medtronic has offices there, as well as a lot of cottage businesses that support the orthopedic industry. In my hometown, the rock star was Dane Miller, who founded Biomet. I have no formal education in medicine or health care, but I’ve seen the impact of it. From my parents and some cousins, uncles who are doctors and surgeons, it’s been interesting to see their work and learn about what’s the latest and greatest in health care.
Henry: What’s so nice about you in particular is, with that background of interests from your family and your celebrity and accomplishments in professional basketball, you have used that to explore and promote ways to make progress in health care and help others who are less fortunate. For example, you’re involved in a telehealth platform for all-in-one practice management; affordable telehealth for pediatrics; health benefits for small businesses; prior authorization—if you can help with prior authorization, we will be in the stands for you at every game because it’s the bane of our existence; radiotherapy; and probably from mom’s background, pharmacy benefit management. Pick any of those you’d like to talk about, and tell us about your involvement and how it’s going.
Plumlee: My ticket into the arena is investment. Nobody’s calling me, asking for my expertise. But a lot of these visionary founders need financial support, and that’s where I get involved. Then also, with the celebrity angle from being an athlete, sometimes you can open doors for a start-up founder that they may not be able to open themselves.
I’m happy to speak about any of those companies. I am excited about the relaxed regulation that’s come from the pandemic; not that it’s like the Wild West out here, but I think it has allowed companies to implement solutions or think about problems in a way that they couldn’t before the pandemic. Take the prior authorization play, for example, and a company called Banjo Health, with one of my favorite founders, a guy named Saar Mahna. Medicare mandates that you turn around prior authorizations within three days. This company has an artificial intelligence and machine-learning play on prior authorizations that can deliver on that.
So efficiencies, things that increase access or affordability, better outcomes, those are the things that attract me. I lean on other people for the due diligence. The pediatric play that you referenced is a company called Blueberry Pediatrics. You have a monthly subscription for $15 that can be reimbursed by Medicaid. They send two devices to your home—an otoscope and an oximeter. The company is live in Florida right now, and it’s diverting a ton of emergency room (ER) visits. From home, for $15 a month, a mom has an otoscope and an oximeter, and she can chat or video conference with a pediatrician. There’s no additional fee. So that’s saving everyone time and saving the system money. Those are the kinds of things I’m attracted to.
Henry: You’ve touched on a couple of hot button issues for us. In oncology, unfortunately, most of our patients have pain. I am mystified every time I try to get a narcotic or a strong painkiller for a patient on a Friday night and I’m told it requires prior authorization and they’ll open up again on Monday. Well, that’s insane. These patients need something right away. So if you have a special interest in helping all of us with prior authorization, the artificial intelligence is a no brainer. If this kind of computer algorithm could happen overnight, that would be wonderful.
You mentioned the ER. Many people go to the ER as a default. They don’t know what else to do. In the COVID era, we’re trying to dial that down because we want to be able to see the sickest and have the non-sick get care elsewhere. If this particular person or people don’t know what to do, they go to the ER, it costs money, takes a lot of time, and others who may be sick are diverted from care. Families worry terribly about their children, so a device for mom and access to a pediatrician for $15 a month is another wonderful idea. These are both very interesting. Another company is in the pharmacy benefit management (PBM) space. Anything you could say about how that works?
Plumlee: I can give an overview of how I look at this as an investor in the PBM space. Three companies control about 75% of a multibillion dollar market. Several initiatives have been pursued politically to provide transparent pricing between these PBMs and pharmaceutical companies, and a lot of people are pointing fingers, but ultimately, drug prices just keep going up. Everybody knows it.
A couple of start-up founders are really set on bringing a competitive marketplace back to the pharmacy benefit manager. As an investor, when you see three people controlling a market, and you have small or medium PBMs that depend on aggregators to get competitive pricing with those big three, you get interested. It’s an interesting industry. My feeling is that somebody is going to disrupt it and bring competition back to that space. Ultimately, drug prices will come down because it’s not sustainable. The insurance companies just accommodate whatever the drug pricing is. If the drug prices go up, your premiums go up. I think these new companies will be level-setting.
Henry: In my world of oncology, we’re just a little more than halfway through 2020 and we’ve had five, six, seven new drugs approved. They all will be very expensive. One of the nicer things that’s happening and may help to tamp this down involves biosimilars. When you go to CVS or Rite Aid, you go down the aspirin aisle and see the generics, and they’re identical to the brand name aspirin. Well, these very complex molecules we used to treat cancer are antibodies or proteins, and they’re made in nature’s factories called cells. They’re not identical to the brand name drugs, but they’re called biosimilars. They work exactly the same as the branded drugs with exactly the same safety–our U.S. FDA has done a nice job of vetting that, to be sure. X, Y, Z Company has copied the brand drug after the patent expires. They were hoping for about a 30% discount in price but we’re seeing more like 15%. Nothing’s ever easy. So you make a very good point. This is not sustainable and the competition will be wonderful to tamp down these prices.
Plumlee: My hope is that those biosimilars and generics get placement in these formularies because the formularies are what’s valuable to the drug manufacturers. But they have to accommodate what the Big Three want in the PBM space. To me, making things affordable and accessible is what a lot of these startups are trying to do. And hopefully they will win.
Henry: What have you been going through, in terms of COVID? Have you recovered fully? Have your taste and smell returned, and you’re back to normal?
Plumlee: I’m all good. It caught me off guard but the symptoms weren’t too intense. For me, it was less than a flu, but more than a cold. And I’m all good today.
Henry: We’re so glad and wish you the best of luck.
Dr. Henry is a clinical professor of medicine at the University of Pennsylvania and vice chairman of the department of medicine at Pennsylvania Hospital in Philadelphia and the host of the Blood & Cancer podcast. He has no relevant financial conflicts.
Mr. Plumlee is a board advisor to both Formsense and the Prysm Institute and a board observer with Voiceitt.


