User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Convalescent plasma actions spark trial recruitment concerns
The agency’s move took many investigators by surprise. The EUA was announced at the White House the day after President Donald J. Trump accused the FDA of delaying approval of therapeutics to hurt his re-election chances.
In a memo describing the decision, the FDA cited data from some controlled and uncontrolled studies and, primarily, data from an open-label expanded-access protocol overseen by the Mayo Clinic.
At the White House, FDA Commissioner Stephen Hahn, MD, said that plasma had been found to save the lives of 35 out of every 100 who were treated. That figure was later found to have been erroneous, and many experts pointed out that Hahn had conflated an absolute risk reduction with a relative reduction. After a firestorm of criticism, Hahn issued an apology.
“The criticism is entirely justified,” he tweeted. “What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
About 15 randomized controlled trials – out of 54 total studies involving convalescent plasma – are underway in the United States, according to ClinicalTrials.gov. The FDA’s Aug. 23 emergency authorization gave clinicians wide leeway to employ convalescent plasma in patients hospitalized with COVID-19.
The agency noted, however, that “adequate and well-controlled randomized trials remain necessary for a definitive demonstration of COVID-19 convalescent plasma efficacy and to determine the optimal product attributes and appropriate patient populations for its use.”
But it’s not clear that people with COVID-19, especially those who are severely ill and hospitalized, will choose to enlist in a clinical trial – where they could receive a placebo – when they instead could get plasma.
“I’ve been asked repeatedly whether the EUA will affect our ability to recruit people into our hospitalized patient trial,” said Liise-anne Pirofski, MD, FIDSA, chief of the department of medicine, infectious diseases division at Albert Einstein College of Medicine and Montefiore Medical Center in the Bronx, New York. “I do not know,” she said, on a call with reporters organized by the Infectious Diseases Society of America.
“But,” she said, “I do know that the trial will continue and that we will discuss the evidence that we have with our patients and give them all that we can to help them weigh the evidence and make up their minds.”
Pirofski said the study being conducted at Montefiore and four other sites has since late April enrolled 190 patients out of a hoped-for 300.
When the study – which compares convalescent plasma to saline in hospitalized patients – was first designed, “there was not any funding for our trial and honestly not a whole lot of interest,” Pirofski told reporters. Individual donors helped support the initial rollout in late April and the trial quickly enrolled 150 patients as the pandemic peaked in the New York City area.
The National Institutes of Health has since given funding, which allowed the study to expand to New York University, Yale University, the University of Miami, and the University of Texas at Houston.
Hopeful, but a long way to go
Shmuel Shoham, MD, FIDSA, associate director of the transplant and oncology infectious diseases center at Johns Hopkins University School of Medicine in Baltimore, said that he’s hopeful that people will continue to enroll in his trial, which is seeking to determine if plasma can prevent COVID-19 in those who’ve been recently exposed.
“Volunteers joining the study is the only way that we’re going to get to know whether this stuff works for prevention and treatment,” Shoham said on the call. He urged physicians and other healthcare workers to talk with patients about considering trial participation.
Shoham’s study is being conducted at 30 US sites and one at the Navajo Nation. It has enrolled 25 out of a hoped-for 500 participants. “We have a long way to go,” said Shoham.
Another Hopkins study to determine whether plasma is helpful in shortening illness in nonhospitalized patients, which is being conducted at the same 31 sites, has enrolled 50 out of 600.
Shoham said recruiting patients with COVID for any study had proven to be difficult. “The vast majority of people that have coronavirus do not come to centers that do clinical trials or interventional trials,” he said, adding that, in addition, most of those “who have coronavirus don’t want to be in a trial. They just want to have coronavirus and get it over with.”
But it’s important to understand how to conduct trials in a pandemic – in part to get answers quickly, he said. Researchers have been looking at convalescent plasma for months, said Shoham. “Why don’t we have the randomized clinical trial data that we want?”
Pirofski noted that trials have also been hobbled in part by “the shifting areas of the pandemic.” Fewer cases make for fewer potential plasma donors.
Both Shoham and Pirofski also said that more needed to be done to encourage plasma donors to participate.
The US Department of Health & Human Services clarified in August that hospitals, physicians, health plans, and other health care workers could contact individuals who had recovered from COVID-19 without violating the HIPAA privacy rule.
Pirofski said she believes that trial investigators know it is legal to reach out to patients. But, she said, “it probably could be better known.”
This article first appeared on Medscape.com.
The agency’s move took many investigators by surprise. The EUA was announced at the White House the day after President Donald J. Trump accused the FDA of delaying approval of therapeutics to hurt his re-election chances.
In a memo describing the decision, the FDA cited data from some controlled and uncontrolled studies and, primarily, data from an open-label expanded-access protocol overseen by the Mayo Clinic.
At the White House, FDA Commissioner Stephen Hahn, MD, said that plasma had been found to save the lives of 35 out of every 100 who were treated. That figure was later found to have been erroneous, and many experts pointed out that Hahn had conflated an absolute risk reduction with a relative reduction. After a firestorm of criticism, Hahn issued an apology.
“The criticism is entirely justified,” he tweeted. “What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
About 15 randomized controlled trials – out of 54 total studies involving convalescent plasma – are underway in the United States, according to ClinicalTrials.gov. The FDA’s Aug. 23 emergency authorization gave clinicians wide leeway to employ convalescent plasma in patients hospitalized with COVID-19.
The agency noted, however, that “adequate and well-controlled randomized trials remain necessary for a definitive demonstration of COVID-19 convalescent plasma efficacy and to determine the optimal product attributes and appropriate patient populations for its use.”
But it’s not clear that people with COVID-19, especially those who are severely ill and hospitalized, will choose to enlist in a clinical trial – where they could receive a placebo – when they instead could get plasma.
“I’ve been asked repeatedly whether the EUA will affect our ability to recruit people into our hospitalized patient trial,” said Liise-anne Pirofski, MD, FIDSA, chief of the department of medicine, infectious diseases division at Albert Einstein College of Medicine and Montefiore Medical Center in the Bronx, New York. “I do not know,” she said, on a call with reporters organized by the Infectious Diseases Society of America.
“But,” she said, “I do know that the trial will continue and that we will discuss the evidence that we have with our patients and give them all that we can to help them weigh the evidence and make up their minds.”
Pirofski said the study being conducted at Montefiore and four other sites has since late April enrolled 190 patients out of a hoped-for 300.
When the study – which compares convalescent plasma to saline in hospitalized patients – was first designed, “there was not any funding for our trial and honestly not a whole lot of interest,” Pirofski told reporters. Individual donors helped support the initial rollout in late April and the trial quickly enrolled 150 patients as the pandemic peaked in the New York City area.
The National Institutes of Health has since given funding, which allowed the study to expand to New York University, Yale University, the University of Miami, and the University of Texas at Houston.
Hopeful, but a long way to go
Shmuel Shoham, MD, FIDSA, associate director of the transplant and oncology infectious diseases center at Johns Hopkins University School of Medicine in Baltimore, said that he’s hopeful that people will continue to enroll in his trial, which is seeking to determine if plasma can prevent COVID-19 in those who’ve been recently exposed.
“Volunteers joining the study is the only way that we’re going to get to know whether this stuff works for prevention and treatment,” Shoham said on the call. He urged physicians and other healthcare workers to talk with patients about considering trial participation.
Shoham’s study is being conducted at 30 US sites and one at the Navajo Nation. It has enrolled 25 out of a hoped-for 500 participants. “We have a long way to go,” said Shoham.
Another Hopkins study to determine whether plasma is helpful in shortening illness in nonhospitalized patients, which is being conducted at the same 31 sites, has enrolled 50 out of 600.
Shoham said recruiting patients with COVID for any study had proven to be difficult. “The vast majority of people that have coronavirus do not come to centers that do clinical trials or interventional trials,” he said, adding that, in addition, most of those “who have coronavirus don’t want to be in a trial. They just want to have coronavirus and get it over with.”
But it’s important to understand how to conduct trials in a pandemic – in part to get answers quickly, he said. Researchers have been looking at convalescent plasma for months, said Shoham. “Why don’t we have the randomized clinical trial data that we want?”
Pirofski noted that trials have also been hobbled in part by “the shifting areas of the pandemic.” Fewer cases make for fewer potential plasma donors.
Both Shoham and Pirofski also said that more needed to be done to encourage plasma donors to participate.
The US Department of Health & Human Services clarified in August that hospitals, physicians, health plans, and other health care workers could contact individuals who had recovered from COVID-19 without violating the HIPAA privacy rule.
Pirofski said she believes that trial investigators know it is legal to reach out to patients. But, she said, “it probably could be better known.”
This article first appeared on Medscape.com.
The agency’s move took many investigators by surprise. The EUA was announced at the White House the day after President Donald J. Trump accused the FDA of delaying approval of therapeutics to hurt his re-election chances.
In a memo describing the decision, the FDA cited data from some controlled and uncontrolled studies and, primarily, data from an open-label expanded-access protocol overseen by the Mayo Clinic.
At the White House, FDA Commissioner Stephen Hahn, MD, said that plasma had been found to save the lives of 35 out of every 100 who were treated. That figure was later found to have been erroneous, and many experts pointed out that Hahn had conflated an absolute risk reduction with a relative reduction. After a firestorm of criticism, Hahn issued an apology.
“The criticism is entirely justified,” he tweeted. “What I should have said better is that the data show a relative risk reduction not an absolute risk reduction.”
About 15 randomized controlled trials – out of 54 total studies involving convalescent plasma – are underway in the United States, according to ClinicalTrials.gov. The FDA’s Aug. 23 emergency authorization gave clinicians wide leeway to employ convalescent plasma in patients hospitalized with COVID-19.
The agency noted, however, that “adequate and well-controlled randomized trials remain necessary for a definitive demonstration of COVID-19 convalescent plasma efficacy and to determine the optimal product attributes and appropriate patient populations for its use.”
But it’s not clear that people with COVID-19, especially those who are severely ill and hospitalized, will choose to enlist in a clinical trial – where they could receive a placebo – when they instead could get plasma.
“I’ve been asked repeatedly whether the EUA will affect our ability to recruit people into our hospitalized patient trial,” said Liise-anne Pirofski, MD, FIDSA, chief of the department of medicine, infectious diseases division at Albert Einstein College of Medicine and Montefiore Medical Center in the Bronx, New York. “I do not know,” she said, on a call with reporters organized by the Infectious Diseases Society of America.
“But,” she said, “I do know that the trial will continue and that we will discuss the evidence that we have with our patients and give them all that we can to help them weigh the evidence and make up their minds.”
Pirofski said the study being conducted at Montefiore and four other sites has since late April enrolled 190 patients out of a hoped-for 300.
When the study – which compares convalescent plasma to saline in hospitalized patients – was first designed, “there was not any funding for our trial and honestly not a whole lot of interest,” Pirofski told reporters. Individual donors helped support the initial rollout in late April and the trial quickly enrolled 150 patients as the pandemic peaked in the New York City area.
The National Institutes of Health has since given funding, which allowed the study to expand to New York University, Yale University, the University of Miami, and the University of Texas at Houston.
Hopeful, but a long way to go
Shmuel Shoham, MD, FIDSA, associate director of the transplant and oncology infectious diseases center at Johns Hopkins University School of Medicine in Baltimore, said that he’s hopeful that people will continue to enroll in his trial, which is seeking to determine if plasma can prevent COVID-19 in those who’ve been recently exposed.
“Volunteers joining the study is the only way that we’re going to get to know whether this stuff works for prevention and treatment,” Shoham said on the call. He urged physicians and other healthcare workers to talk with patients about considering trial participation.
Shoham’s study is being conducted at 30 US sites and one at the Navajo Nation. It has enrolled 25 out of a hoped-for 500 participants. “We have a long way to go,” said Shoham.
Another Hopkins study to determine whether plasma is helpful in shortening illness in nonhospitalized patients, which is being conducted at the same 31 sites, has enrolled 50 out of 600.
Shoham said recruiting patients with COVID for any study had proven to be difficult. “The vast majority of people that have coronavirus do not come to centers that do clinical trials or interventional trials,” he said, adding that, in addition, most of those “who have coronavirus don’t want to be in a trial. They just want to have coronavirus and get it over with.”
But it’s important to understand how to conduct trials in a pandemic – in part to get answers quickly, he said. Researchers have been looking at convalescent plasma for months, said Shoham. “Why don’t we have the randomized clinical trial data that we want?”
Pirofski noted that trials have also been hobbled in part by “the shifting areas of the pandemic.” Fewer cases make for fewer potential plasma donors.
Both Shoham and Pirofski also said that more needed to be done to encourage plasma donors to participate.
The US Department of Health & Human Services clarified in August that hospitals, physicians, health plans, and other health care workers could contact individuals who had recovered from COVID-19 without violating the HIPAA privacy rule.
Pirofski said she believes that trial investigators know it is legal to reach out to patients. But, she said, “it probably could be better known.”
This article first appeared on Medscape.com.
Prognosis for rural hospitals worsens with pandemic
Jerome Antone said he is one of the lucky ones.
After becoming ill with COVID-19, Mr. Antone was hospitalized only 65 miles away from his small Alabama town. He is the mayor of Georgiana – population 1,700.
“It hit our rural community so rabid,” Mr. Antone said. The town’s hospital closed last year. If hospitals in nearby communities don’t have beds available, “you may have to go 4 or 5 hours away.”
Eighteen rural hospitals closed last year and the first 3 months of 2020 were “really big months,” said Mark Holmes, PhD, director of the Cecil G. Sheps Center for Health Services Research at the University of North Carolina at Chapel Hill. Many of the losses are in Southern states like Florida and Texas. More than 170 rural hospitals have closed nationwide since 2005, according to data collected by the Sheps Center.
It’s a dangerous scenario. “We know that a closure leads to higher mortality pretty quickly” among the populations served, said Dr. Holmes, who is also a professor at UNC Gillings School of Global Public Health. “That’s pretty clear.”
One 2019 study found that death rates in the surrounding communities increase nearly 6% after a rural hospital closes – and that’s when there’s not a pandemic.
Add to that what is known about the coronavirus: People who are obese or live with diabetes, hypertension, asthma, and other underlying health issues are more susceptible to COVID-19. Rural areas tend to have higher rates of these conditions. And rural residents are more likely to be older, sicker and poorer than those in urban areas. All this leaves rural communities particularly vulnerable to the coronavirus.
Congress approved billions in federal relief funds for health care providers. Initially, federal officials based what a hospital would get on its Medicare payments, but by late April they heeded criticism and carved out funds for rural hospitals and COVID-19 hot spots. Rural hospitals leapt at the chance to shore up already-negative budgets and prepare for the pandemic.
The funds “helped rural hospitals with the immediate storm,” said Don Williamson, MD, president of the Alabama Hospital Association. Nearly 80% of Alabama’s rural hospitals began the year with negative balance sheets and about 8 days’ worth of cash on hand.
Before the pandemic hit this year, hundreds of rural hospitals “were just trying to keep their doors open,” said Maggie Elehwany, vice president of government affairs with the National Rural Health Association. Then an estimated 70% of their income stopped as patients avoided the emergency room, doctor’s appointments, and elective surgeries.
“It was devastating,” Ms. Elehwany said.
Paul Taylor, chief executive of a 25-bed critical-access hospital and outpatient clinics in northwestern Arkansas, accepted millions in grants and loan money Congress approved this spring, largely through the CARES (Coronavirus Aid, Relief, and Economic Security) Act.
“For us, this was survival money and we spent it already,” Mr. Taylor said. With those funds, Ozarks Community Hospital increased surge capacity, expanding from 25 beds to 50 beds, adding negative pressure rooms and buying six ventilators. Taylor also ramped up COVID-19 testing at his hospital and clinics, located near some meat-processing plants.
Throughout June and July, Ozarks tested 1,000 patients a day and reported a 20% positive rate. The rate dropped to 16.9% in late July. But patients continue to avoid routine care.
Mr. Taylor said revenue is still constrained and he does not know how he will pay back $8 million that he borrowed from Medicare. The program allowed hospitals to borrow against future payments from the federal government, but stipulated that repayment would begin within 120 days.
For Mr. Taylor, this seems impossible. Medicare makes up 40% of Ozarks’ income. And he has to pay the loan back before he gets any more payments from Medicare. He’s hoping to refinance the hospital’s mortgage.
“If I get no relief and they take the money ... we won’t still be open,” Mr. Taylor said. Ozarks provides 625 jobs and serves an area with a population of about 75,000.
There are 1,300 small critical-access hospitals like Ozarks in rural America, and of those, 859 took advantage of the Medicare loans, sending about $3.1 billion into the local communities. But many rural communities have not yet experienced a surge in coronavirus cases – national leaders fear it will come as part of a new phase.
“There are pockets of rural America who say, ‘We haven’t seen a single COVID patient yet and we do not believe it’s real,’ ” Mr. Taylor said. “They will get hit sooner or later.”
Across the country, the reduced patient numbers and increased spending required to fight and prepare for the coronavirus was “like a knife cutting into a hospital’s blood supply,” said Ge Bai, PhD, associate professor of health policy and management at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Dr. Bai said the way the federal government reimbursed small rural hospitals through federal programs like Medicare before the pandemic was faulty and inefficient. “They are too weak to survive,” she said.
In rural Texas, about 2 hours from Dallas, Titus Regional Medical Center chief executive officer Terry Scoggin cut staff and furloughed workers even as his rural hospital faced down the pandemic. Titus Regional lost about $4 million last fiscal year and broke even each of the three years before that.
Mr. Scoggin said he did not cut from his clinical staff, though. Titus is now facing its second surge of the virus in the community. “The last 7 days, we’ve been testing 30% positive,” he said, including the case of his father, who contracted it at a nursing home and survived.
“It’s personal and this is real,” Mr. Scoggin said. “You know the people who are infected. You know the people who are passing away.”
Of his roughly 700 employees, 48 have tested positive for the virus and 1 has died. They are short on testing kits, medication, and supplies.
“Right now the staff is strained,” Mr. Scoggin said. “I’ve been blown away by their selflessness and unbelievable spirit. We’re resilient, we’re nimble, and we will make it. We don’t have a choice.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Jerome Antone said he is one of the lucky ones.
After becoming ill with COVID-19, Mr. Antone was hospitalized only 65 miles away from his small Alabama town. He is the mayor of Georgiana – population 1,700.
“It hit our rural community so rabid,” Mr. Antone said. The town’s hospital closed last year. If hospitals in nearby communities don’t have beds available, “you may have to go 4 or 5 hours away.”
Eighteen rural hospitals closed last year and the first 3 months of 2020 were “really big months,” said Mark Holmes, PhD, director of the Cecil G. Sheps Center for Health Services Research at the University of North Carolina at Chapel Hill. Many of the losses are in Southern states like Florida and Texas. More than 170 rural hospitals have closed nationwide since 2005, according to data collected by the Sheps Center.
It’s a dangerous scenario. “We know that a closure leads to higher mortality pretty quickly” among the populations served, said Dr. Holmes, who is also a professor at UNC Gillings School of Global Public Health. “That’s pretty clear.”
One 2019 study found that death rates in the surrounding communities increase nearly 6% after a rural hospital closes – and that’s when there’s not a pandemic.
Add to that what is known about the coronavirus: People who are obese or live with diabetes, hypertension, asthma, and other underlying health issues are more susceptible to COVID-19. Rural areas tend to have higher rates of these conditions. And rural residents are more likely to be older, sicker and poorer than those in urban areas. All this leaves rural communities particularly vulnerable to the coronavirus.
Congress approved billions in federal relief funds for health care providers. Initially, federal officials based what a hospital would get on its Medicare payments, but by late April they heeded criticism and carved out funds for rural hospitals and COVID-19 hot spots. Rural hospitals leapt at the chance to shore up already-negative budgets and prepare for the pandemic.
The funds “helped rural hospitals with the immediate storm,” said Don Williamson, MD, president of the Alabama Hospital Association. Nearly 80% of Alabama’s rural hospitals began the year with negative balance sheets and about 8 days’ worth of cash on hand.
Before the pandemic hit this year, hundreds of rural hospitals “were just trying to keep their doors open,” said Maggie Elehwany, vice president of government affairs with the National Rural Health Association. Then an estimated 70% of their income stopped as patients avoided the emergency room, doctor’s appointments, and elective surgeries.
“It was devastating,” Ms. Elehwany said.
Paul Taylor, chief executive of a 25-bed critical-access hospital and outpatient clinics in northwestern Arkansas, accepted millions in grants and loan money Congress approved this spring, largely through the CARES (Coronavirus Aid, Relief, and Economic Security) Act.
“For us, this was survival money and we spent it already,” Mr. Taylor said. With those funds, Ozarks Community Hospital increased surge capacity, expanding from 25 beds to 50 beds, adding negative pressure rooms and buying six ventilators. Taylor also ramped up COVID-19 testing at his hospital and clinics, located near some meat-processing plants.
Throughout June and July, Ozarks tested 1,000 patients a day and reported a 20% positive rate. The rate dropped to 16.9% in late July. But patients continue to avoid routine care.
Mr. Taylor said revenue is still constrained and he does not know how he will pay back $8 million that he borrowed from Medicare. The program allowed hospitals to borrow against future payments from the federal government, but stipulated that repayment would begin within 120 days.
For Mr. Taylor, this seems impossible. Medicare makes up 40% of Ozarks’ income. And he has to pay the loan back before he gets any more payments from Medicare. He’s hoping to refinance the hospital’s mortgage.
“If I get no relief and they take the money ... we won’t still be open,” Mr. Taylor said. Ozarks provides 625 jobs and serves an area with a population of about 75,000.
There are 1,300 small critical-access hospitals like Ozarks in rural America, and of those, 859 took advantage of the Medicare loans, sending about $3.1 billion into the local communities. But many rural communities have not yet experienced a surge in coronavirus cases – national leaders fear it will come as part of a new phase.
“There are pockets of rural America who say, ‘We haven’t seen a single COVID patient yet and we do not believe it’s real,’ ” Mr. Taylor said. “They will get hit sooner or later.”
Across the country, the reduced patient numbers and increased spending required to fight and prepare for the coronavirus was “like a knife cutting into a hospital’s blood supply,” said Ge Bai, PhD, associate professor of health policy and management at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Dr. Bai said the way the federal government reimbursed small rural hospitals through federal programs like Medicare before the pandemic was faulty and inefficient. “They are too weak to survive,” she said.
In rural Texas, about 2 hours from Dallas, Titus Regional Medical Center chief executive officer Terry Scoggin cut staff and furloughed workers even as his rural hospital faced down the pandemic. Titus Regional lost about $4 million last fiscal year and broke even each of the three years before that.
Mr. Scoggin said he did not cut from his clinical staff, though. Titus is now facing its second surge of the virus in the community. “The last 7 days, we’ve been testing 30% positive,” he said, including the case of his father, who contracted it at a nursing home and survived.
“It’s personal and this is real,” Mr. Scoggin said. “You know the people who are infected. You know the people who are passing away.”
Of his roughly 700 employees, 48 have tested positive for the virus and 1 has died. They are short on testing kits, medication, and supplies.
“Right now the staff is strained,” Mr. Scoggin said. “I’ve been blown away by their selflessness and unbelievable spirit. We’re resilient, we’re nimble, and we will make it. We don’t have a choice.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Jerome Antone said he is one of the lucky ones.
After becoming ill with COVID-19, Mr. Antone was hospitalized only 65 miles away from his small Alabama town. He is the mayor of Georgiana – population 1,700.
“It hit our rural community so rabid,” Mr. Antone said. The town’s hospital closed last year. If hospitals in nearby communities don’t have beds available, “you may have to go 4 or 5 hours away.”
Eighteen rural hospitals closed last year and the first 3 months of 2020 were “really big months,” said Mark Holmes, PhD, director of the Cecil G. Sheps Center for Health Services Research at the University of North Carolina at Chapel Hill. Many of the losses are in Southern states like Florida and Texas. More than 170 rural hospitals have closed nationwide since 2005, according to data collected by the Sheps Center.
It’s a dangerous scenario. “We know that a closure leads to higher mortality pretty quickly” among the populations served, said Dr. Holmes, who is also a professor at UNC Gillings School of Global Public Health. “That’s pretty clear.”
One 2019 study found that death rates in the surrounding communities increase nearly 6% after a rural hospital closes – and that’s when there’s not a pandemic.
Add to that what is known about the coronavirus: People who are obese or live with diabetes, hypertension, asthma, and other underlying health issues are more susceptible to COVID-19. Rural areas tend to have higher rates of these conditions. And rural residents are more likely to be older, sicker and poorer than those in urban areas. All this leaves rural communities particularly vulnerable to the coronavirus.
Congress approved billions in federal relief funds for health care providers. Initially, federal officials based what a hospital would get on its Medicare payments, but by late April they heeded criticism and carved out funds for rural hospitals and COVID-19 hot spots. Rural hospitals leapt at the chance to shore up already-negative budgets and prepare for the pandemic.
The funds “helped rural hospitals with the immediate storm,” said Don Williamson, MD, president of the Alabama Hospital Association. Nearly 80% of Alabama’s rural hospitals began the year with negative balance sheets and about 8 days’ worth of cash on hand.
Before the pandemic hit this year, hundreds of rural hospitals “were just trying to keep their doors open,” said Maggie Elehwany, vice president of government affairs with the National Rural Health Association. Then an estimated 70% of their income stopped as patients avoided the emergency room, doctor’s appointments, and elective surgeries.
“It was devastating,” Ms. Elehwany said.
Paul Taylor, chief executive of a 25-bed critical-access hospital and outpatient clinics in northwestern Arkansas, accepted millions in grants and loan money Congress approved this spring, largely through the CARES (Coronavirus Aid, Relief, and Economic Security) Act.
“For us, this was survival money and we spent it already,” Mr. Taylor said. With those funds, Ozarks Community Hospital increased surge capacity, expanding from 25 beds to 50 beds, adding negative pressure rooms and buying six ventilators. Taylor also ramped up COVID-19 testing at his hospital and clinics, located near some meat-processing plants.
Throughout June and July, Ozarks tested 1,000 patients a day and reported a 20% positive rate. The rate dropped to 16.9% in late July. But patients continue to avoid routine care.
Mr. Taylor said revenue is still constrained and he does not know how he will pay back $8 million that he borrowed from Medicare. The program allowed hospitals to borrow against future payments from the federal government, but stipulated that repayment would begin within 120 days.
For Mr. Taylor, this seems impossible. Medicare makes up 40% of Ozarks’ income. And he has to pay the loan back before he gets any more payments from Medicare. He’s hoping to refinance the hospital’s mortgage.
“If I get no relief and they take the money ... we won’t still be open,” Mr. Taylor said. Ozarks provides 625 jobs and serves an area with a population of about 75,000.
There are 1,300 small critical-access hospitals like Ozarks in rural America, and of those, 859 took advantage of the Medicare loans, sending about $3.1 billion into the local communities. But many rural communities have not yet experienced a surge in coronavirus cases – national leaders fear it will come as part of a new phase.
“There are pockets of rural America who say, ‘We haven’t seen a single COVID patient yet and we do not believe it’s real,’ ” Mr. Taylor said. “They will get hit sooner or later.”
Across the country, the reduced patient numbers and increased spending required to fight and prepare for the coronavirus was “like a knife cutting into a hospital’s blood supply,” said Ge Bai, PhD, associate professor of health policy and management at the Johns Hopkins Bloomberg School of Public Health in Baltimore.
Dr. Bai said the way the federal government reimbursed small rural hospitals through federal programs like Medicare before the pandemic was faulty and inefficient. “They are too weak to survive,” she said.
In rural Texas, about 2 hours from Dallas, Titus Regional Medical Center chief executive officer Terry Scoggin cut staff and furloughed workers even as his rural hospital faced down the pandemic. Titus Regional lost about $4 million last fiscal year and broke even each of the three years before that.
Mr. Scoggin said he did not cut from his clinical staff, though. Titus is now facing its second surge of the virus in the community. “The last 7 days, we’ve been testing 30% positive,” he said, including the case of his father, who contracted it at a nursing home and survived.
“It’s personal and this is real,” Mr. Scoggin said. “You know the people who are infected. You know the people who are passing away.”
Of his roughly 700 employees, 48 have tested positive for the virus and 1 has died. They are short on testing kits, medication, and supplies.
“Right now the staff is strained,” Mr. Scoggin said. “I’ve been blown away by their selflessness and unbelievable spirit. We’re resilient, we’re nimble, and we will make it. We don’t have a choice.”
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of the Kaiser Family Foundation, which is not affiliated with Kaiser Permanente.
Large study finds no link between TCI use, skin cancer in patients with AD
The results also suggest dose, frequency, and exposure duration to the topical calcineurin inhibitors (TCIs) tacrolimus and pimecrolimus are not associated with an increased risk of keratinocyte carcinomas (KCs), basal cell carcinomas (BCCs), and squamous cell carcinomas (SCCs) in patients with atopic dermatitis (AD), according to Maryam M. Asgari, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues. In 2006, the Food and Drug Administration announced the addition of the boxed warning to the labeling of TCIs regarding a possible risk of cancer associated with use of pimecrolimus (Elidel) and with tacrolimus (Protopic), because of an increased risk of KCs associated with oral calcineurin inhibitors and reports of skin cancer in patients on TCIs.
“Controversy has surrounded the association between TCI exposure and KC risk since the black-box warning was issued by the FDA. A hypothesized mechanism of action for TCIs increasing KC risk includes a direct effect of calcineurin inhibition on DNA repair and apoptosis, which could influence keratinocyte carcinogenesis,” the authors of the study wrote in JAMA Dermatology. But, they added, there have been “conflicting results” in research exploring this association.
In the retrospective cohort study, Dr. Asgari and coauthors evaluated 93,746 adult patients with AD at Kaiser Permanente Northern California, diagnosed between January 2002 and December 2013, comparing skin cancer risk among 7,033 patients exposed to TCIs, 73,674 patients taking topical corticosteroids, and 46,141 patients who had not been exposed to TCIs or topical corticosteroids. Results were adjusted in a multivariate Cox regression analysis for age, gender, race/ethnicity, calendar year, number of dermatology visits per year, history of KCs, immunosuppression, prior systemic AD treatment, autoimmune disease, treatment with ultraviolet therapy, chemotherapy, and radiotherapy.
The researchers also examined how TCI dose, frequency and exposure duration impacted skin cancer risk. Patients were grouped by high-dose (0.1%) and low-dose (0.03%) formulations of tacrolimus; and the 1% formulation of pimecrolimus. Frequency of use was defined as low (once daily or less) or high (twice daily or more), and exposure duration was based on short- (less than 2 years), moderate- (2-4 years), and long-term (4 years or more) use. Patients were at least 40 years old (mean age, 58.5 years), 58.7% were women, 50.5% were White, 20.6% were Asian, 12.2% were Hispanic, and 7.9% were Black. They were followed for a mean of 7.70 years.
Compared with patients who were exposed to topical corticosteroids, there was no association between risk of KCs and exposure to TCIs in patients with AD (adjusted hazard ratio, 1.02; 95% confidence interval, 0.93-1.13). There were also no significant differences in risk of BCCs and TCI exposure (aHR, 1.01; 95% CI, 0.90-1.14) and risk of SCCs and TCI exposure (aHR, 0.94; 95% CI, 0.82-1.08), compared with patients exposed to topical corticosteroids.
Results were similar for risk of KCs (aHR, 1.03; 95% CI, 0.92-1.14), BCCs (aHR, 1.04; 95% CI, 0.91-1.19), and SCCs (aHR, 0.91; 95% CI, 0.78-1.06) when patients exposed to TCIs were compared with those with AD who were unexposed to any medication. In secondary analyses, Dr. Asgari and coauthors found no association with overall risk of KCs, or risk of BCCs or SCCs, and the dose, frequency, or exposure duration to TCIs.
“Our findings appear to support those of smaller postmarketing surveillance studies of TCI and KC risk and may provide some reassurance about the safety profile of this class of topical agents in the treatment of AD,” they concluded.
In an interview, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, said initial concerns surrounding TCIs were based on high doses potentially increasing the risk of malignancy, and off-label use of TCIs for inflammatory skin diseases other than AD.
“However, the FDA’s concerns may not have been justified,” he said. The manufacturers of pimecrolimus and tacrolimus have published results of 10-year observational registries that assess cancer risk, which “found no evidence of any associations between TCIs and malignancy,” noted Dr. Silverberg, who is also director of clinical research and contact dermatitis at George Washington University.
Elizabeth Hughes, MD, a dermatologist in private practice in San Antonio, said in an interview that initial enthusiasm was “huge” for use of TCIs like tacrolimus in patients with AD when they first became available, especially in the pediatric population, for whom clinicians are hesitant to use long-term strong topical steroids. However, parents of children taking the medication soon became concerned about potential side effects.
“The TCIs can be absorbed to a small extent through body surface area, so it was not a big leap to become concerned that infants and small children could absorb enough ... into the bloodstream to give a similar side effect profile as oral tacrolimus,” she said.
The addition of the boxed warning in 2006 was frustrating for dermatologists “because a medication we needed very much for a young population now was ‘labeled’ and parents were scared to use it,” Dr. Hughes explained.
Dr. Silverberg noted that, while the results of the new study are unlikely to change clinical practice, they are reassuring, and provide real-world data and “further confirmation of previous studies showing no associations between AD and malignancy.”
“Since AD and skin cancer are both commonly managed by dermatologists, there is potential for increased surveillance and detection of skin cancers in AD patients. So, the greatest chance of seeing a false-positive signal for malignancy would likely occur with skin cancers,” he pointed out. “Yet, even in the case of skin cancers, there were no demonstrable signals.”
Based on the results, “I think it is definitely reasonable to reconsider” the TCI boxed warning, but there isn’t much precedent for boxed warnings to be removed from labeling, Dr. Silverberg commented. “Unfortunately, the black-box warning may persist despite a lot of reassuring data.”
In a related editorial, Aaron M. Drucker, MD, ScM, and Mina Tadrous, PharmD, PhD, of the University of Toronto, said the boxed warning “had the intent of helping patients and clinicians understand possible risks,” but also carried the “potential for harm” if patients discontinued or did not adhere to treatment. “Safety warnings on topical medications could lead to undertreatment of atopic dermatitis, reduced quality of life and, potentially, increased use of more toxic systemic medications.”
Long-term studies of medications and cancer risk are challenging to perform, having to account for dose-response relationships, confounding by indication, and time bias, among other factors, and this study “recognizes and attempts to address many of these challenges,” Dr. Drucker and Dr. Tadrous wrote.
These results are similar to previous studies that have “consistently reported no or minimal association between TCI use and skin cancer,” they noted, adding that, “if an association exists, it is likely very small, meaning that skin cancer attributable to TCI use is rare. Clinicians can use this evidence to counsel and reassure patients for whom the benefits of ongoing treatment with TCIs may outweigh the harms.”
This study was funded by a grant from Valeant Pharmaceuticals. Dr. Asgari reported receiving grants from Valeant during the study, and from Pfizer not related to the study. The other authors reported no relevant conflicts of interest. Dr. Drucker reported relationships with the Canadian Agency for Drugs and Technology in Health, CME Outfitters, Eczema Society of Canada, Sanofi, Regeneron, and RTI Health Solutions in the form of paid fees, consultancies, honoraria, educational grants, and other compensation paid to him and/or his institution. Dr. Tadrous reported no relevant disclosures. Dr. Silverberg reported receiving honoraria for advisory board, speaker, and consultant services from numerous pharmaceutical manufacturers, and research grants for investigator services from GlaxoSmithKline and Galderma. Dr. Hughes Tichy reported no relevant financial disclosures. Dr. Silverberg is a member of the Dermatology News editorial advisory board.
SOURCE: Asgari MM et al. JAMA Dermatol. 2020 Aug 12. doi: 10.1001/jamadermatol.2020.2240.
The results also suggest dose, frequency, and exposure duration to the topical calcineurin inhibitors (TCIs) tacrolimus and pimecrolimus are not associated with an increased risk of keratinocyte carcinomas (KCs), basal cell carcinomas (BCCs), and squamous cell carcinomas (SCCs) in patients with atopic dermatitis (AD), according to Maryam M. Asgari, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues. In 2006, the Food and Drug Administration announced the addition of the boxed warning to the labeling of TCIs regarding a possible risk of cancer associated with use of pimecrolimus (Elidel) and with tacrolimus (Protopic), because of an increased risk of KCs associated with oral calcineurin inhibitors and reports of skin cancer in patients on TCIs.
“Controversy has surrounded the association between TCI exposure and KC risk since the black-box warning was issued by the FDA. A hypothesized mechanism of action for TCIs increasing KC risk includes a direct effect of calcineurin inhibition on DNA repair and apoptosis, which could influence keratinocyte carcinogenesis,” the authors of the study wrote in JAMA Dermatology. But, they added, there have been “conflicting results” in research exploring this association.
In the retrospective cohort study, Dr. Asgari and coauthors evaluated 93,746 adult patients with AD at Kaiser Permanente Northern California, diagnosed between January 2002 and December 2013, comparing skin cancer risk among 7,033 patients exposed to TCIs, 73,674 patients taking topical corticosteroids, and 46,141 patients who had not been exposed to TCIs or topical corticosteroids. Results were adjusted in a multivariate Cox regression analysis for age, gender, race/ethnicity, calendar year, number of dermatology visits per year, history of KCs, immunosuppression, prior systemic AD treatment, autoimmune disease, treatment with ultraviolet therapy, chemotherapy, and radiotherapy.
The researchers also examined how TCI dose, frequency and exposure duration impacted skin cancer risk. Patients were grouped by high-dose (0.1%) and low-dose (0.03%) formulations of tacrolimus; and the 1% formulation of pimecrolimus. Frequency of use was defined as low (once daily or less) or high (twice daily or more), and exposure duration was based on short- (less than 2 years), moderate- (2-4 years), and long-term (4 years or more) use. Patients were at least 40 years old (mean age, 58.5 years), 58.7% were women, 50.5% were White, 20.6% were Asian, 12.2% were Hispanic, and 7.9% were Black. They were followed for a mean of 7.70 years.
Compared with patients who were exposed to topical corticosteroids, there was no association between risk of KCs and exposure to TCIs in patients with AD (adjusted hazard ratio, 1.02; 95% confidence interval, 0.93-1.13). There were also no significant differences in risk of BCCs and TCI exposure (aHR, 1.01; 95% CI, 0.90-1.14) and risk of SCCs and TCI exposure (aHR, 0.94; 95% CI, 0.82-1.08), compared with patients exposed to topical corticosteroids.
Results were similar for risk of KCs (aHR, 1.03; 95% CI, 0.92-1.14), BCCs (aHR, 1.04; 95% CI, 0.91-1.19), and SCCs (aHR, 0.91; 95% CI, 0.78-1.06) when patients exposed to TCIs were compared with those with AD who were unexposed to any medication. In secondary analyses, Dr. Asgari and coauthors found no association with overall risk of KCs, or risk of BCCs or SCCs, and the dose, frequency, or exposure duration to TCIs.
“Our findings appear to support those of smaller postmarketing surveillance studies of TCI and KC risk and may provide some reassurance about the safety profile of this class of topical agents in the treatment of AD,” they concluded.
In an interview, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, said initial concerns surrounding TCIs were based on high doses potentially increasing the risk of malignancy, and off-label use of TCIs for inflammatory skin diseases other than AD.
“However, the FDA’s concerns may not have been justified,” he said. The manufacturers of pimecrolimus and tacrolimus have published results of 10-year observational registries that assess cancer risk, which “found no evidence of any associations between TCIs and malignancy,” noted Dr. Silverberg, who is also director of clinical research and contact dermatitis at George Washington University.
Elizabeth Hughes, MD, a dermatologist in private practice in San Antonio, said in an interview that initial enthusiasm was “huge” for use of TCIs like tacrolimus in patients with AD when they first became available, especially in the pediatric population, for whom clinicians are hesitant to use long-term strong topical steroids. However, parents of children taking the medication soon became concerned about potential side effects.
“The TCIs can be absorbed to a small extent through body surface area, so it was not a big leap to become concerned that infants and small children could absorb enough ... into the bloodstream to give a similar side effect profile as oral tacrolimus,” she said.
The addition of the boxed warning in 2006 was frustrating for dermatologists “because a medication we needed very much for a young population now was ‘labeled’ and parents were scared to use it,” Dr. Hughes explained.
Dr. Silverberg noted that, while the results of the new study are unlikely to change clinical practice, they are reassuring, and provide real-world data and “further confirmation of previous studies showing no associations between AD and malignancy.”
“Since AD and skin cancer are both commonly managed by dermatologists, there is potential for increased surveillance and detection of skin cancers in AD patients. So, the greatest chance of seeing a false-positive signal for malignancy would likely occur with skin cancers,” he pointed out. “Yet, even in the case of skin cancers, there were no demonstrable signals.”
Based on the results, “I think it is definitely reasonable to reconsider” the TCI boxed warning, but there isn’t much precedent for boxed warnings to be removed from labeling, Dr. Silverberg commented. “Unfortunately, the black-box warning may persist despite a lot of reassuring data.”
In a related editorial, Aaron M. Drucker, MD, ScM, and Mina Tadrous, PharmD, PhD, of the University of Toronto, said the boxed warning “had the intent of helping patients and clinicians understand possible risks,” but also carried the “potential for harm” if patients discontinued or did not adhere to treatment. “Safety warnings on topical medications could lead to undertreatment of atopic dermatitis, reduced quality of life and, potentially, increased use of more toxic systemic medications.”
Long-term studies of medications and cancer risk are challenging to perform, having to account for dose-response relationships, confounding by indication, and time bias, among other factors, and this study “recognizes and attempts to address many of these challenges,” Dr. Drucker and Dr. Tadrous wrote.
These results are similar to previous studies that have “consistently reported no or minimal association between TCI use and skin cancer,” they noted, adding that, “if an association exists, it is likely very small, meaning that skin cancer attributable to TCI use is rare. Clinicians can use this evidence to counsel and reassure patients for whom the benefits of ongoing treatment with TCIs may outweigh the harms.”
This study was funded by a grant from Valeant Pharmaceuticals. Dr. Asgari reported receiving grants from Valeant during the study, and from Pfizer not related to the study. The other authors reported no relevant conflicts of interest. Dr. Drucker reported relationships with the Canadian Agency for Drugs and Technology in Health, CME Outfitters, Eczema Society of Canada, Sanofi, Regeneron, and RTI Health Solutions in the form of paid fees, consultancies, honoraria, educational grants, and other compensation paid to him and/or his institution. Dr. Tadrous reported no relevant disclosures. Dr. Silverberg reported receiving honoraria for advisory board, speaker, and consultant services from numerous pharmaceutical manufacturers, and research grants for investigator services from GlaxoSmithKline and Galderma. Dr. Hughes Tichy reported no relevant financial disclosures. Dr. Silverberg is a member of the Dermatology News editorial advisory board.
SOURCE: Asgari MM et al. JAMA Dermatol. 2020 Aug 12. doi: 10.1001/jamadermatol.2020.2240.
The results also suggest dose, frequency, and exposure duration to the topical calcineurin inhibitors (TCIs) tacrolimus and pimecrolimus are not associated with an increased risk of keratinocyte carcinomas (KCs), basal cell carcinomas (BCCs), and squamous cell carcinomas (SCCs) in patients with atopic dermatitis (AD), according to Maryam M. Asgari, MD, MPH, professor of dermatology, Harvard Medical School, Boston, and colleagues. In 2006, the Food and Drug Administration announced the addition of the boxed warning to the labeling of TCIs regarding a possible risk of cancer associated with use of pimecrolimus (Elidel) and with tacrolimus (Protopic), because of an increased risk of KCs associated with oral calcineurin inhibitors and reports of skin cancer in patients on TCIs.
“Controversy has surrounded the association between TCI exposure and KC risk since the black-box warning was issued by the FDA. A hypothesized mechanism of action for TCIs increasing KC risk includes a direct effect of calcineurin inhibition on DNA repair and apoptosis, which could influence keratinocyte carcinogenesis,” the authors of the study wrote in JAMA Dermatology. But, they added, there have been “conflicting results” in research exploring this association.
In the retrospective cohort study, Dr. Asgari and coauthors evaluated 93,746 adult patients with AD at Kaiser Permanente Northern California, diagnosed between January 2002 and December 2013, comparing skin cancer risk among 7,033 patients exposed to TCIs, 73,674 patients taking topical corticosteroids, and 46,141 patients who had not been exposed to TCIs or topical corticosteroids. Results were adjusted in a multivariate Cox regression analysis for age, gender, race/ethnicity, calendar year, number of dermatology visits per year, history of KCs, immunosuppression, prior systemic AD treatment, autoimmune disease, treatment with ultraviolet therapy, chemotherapy, and radiotherapy.
The researchers also examined how TCI dose, frequency and exposure duration impacted skin cancer risk. Patients were grouped by high-dose (0.1%) and low-dose (0.03%) formulations of tacrolimus; and the 1% formulation of pimecrolimus. Frequency of use was defined as low (once daily or less) or high (twice daily or more), and exposure duration was based on short- (less than 2 years), moderate- (2-4 years), and long-term (4 years or more) use. Patients were at least 40 years old (mean age, 58.5 years), 58.7% were women, 50.5% were White, 20.6% were Asian, 12.2% were Hispanic, and 7.9% were Black. They were followed for a mean of 7.70 years.
Compared with patients who were exposed to topical corticosteroids, there was no association between risk of KCs and exposure to TCIs in patients with AD (adjusted hazard ratio, 1.02; 95% confidence interval, 0.93-1.13). There were also no significant differences in risk of BCCs and TCI exposure (aHR, 1.01; 95% CI, 0.90-1.14) and risk of SCCs and TCI exposure (aHR, 0.94; 95% CI, 0.82-1.08), compared with patients exposed to topical corticosteroids.
Results were similar for risk of KCs (aHR, 1.03; 95% CI, 0.92-1.14), BCCs (aHR, 1.04; 95% CI, 0.91-1.19), and SCCs (aHR, 0.91; 95% CI, 0.78-1.06) when patients exposed to TCIs were compared with those with AD who were unexposed to any medication. In secondary analyses, Dr. Asgari and coauthors found no association with overall risk of KCs, or risk of BCCs or SCCs, and the dose, frequency, or exposure duration to TCIs.
“Our findings appear to support those of smaller postmarketing surveillance studies of TCI and KC risk and may provide some reassurance about the safety profile of this class of topical agents in the treatment of AD,” they concluded.
In an interview, Jonathan Silverberg, MD, PhD, MPH, associate professor of dermatology, George Washington University, Washington, said initial concerns surrounding TCIs were based on high doses potentially increasing the risk of malignancy, and off-label use of TCIs for inflammatory skin diseases other than AD.
“However, the FDA’s concerns may not have been justified,” he said. The manufacturers of pimecrolimus and tacrolimus have published results of 10-year observational registries that assess cancer risk, which “found no evidence of any associations between TCIs and malignancy,” noted Dr. Silverberg, who is also director of clinical research and contact dermatitis at George Washington University.
Elizabeth Hughes, MD, a dermatologist in private practice in San Antonio, said in an interview that initial enthusiasm was “huge” for use of TCIs like tacrolimus in patients with AD when they first became available, especially in the pediatric population, for whom clinicians are hesitant to use long-term strong topical steroids. However, parents of children taking the medication soon became concerned about potential side effects.
“The TCIs can be absorbed to a small extent through body surface area, so it was not a big leap to become concerned that infants and small children could absorb enough ... into the bloodstream to give a similar side effect profile as oral tacrolimus,” she said.
The addition of the boxed warning in 2006 was frustrating for dermatologists “because a medication we needed very much for a young population now was ‘labeled’ and parents were scared to use it,” Dr. Hughes explained.
Dr. Silverberg noted that, while the results of the new study are unlikely to change clinical practice, they are reassuring, and provide real-world data and “further confirmation of previous studies showing no associations between AD and malignancy.”
“Since AD and skin cancer are both commonly managed by dermatologists, there is potential for increased surveillance and detection of skin cancers in AD patients. So, the greatest chance of seeing a false-positive signal for malignancy would likely occur with skin cancers,” he pointed out. “Yet, even in the case of skin cancers, there were no demonstrable signals.”
Based on the results, “I think it is definitely reasonable to reconsider” the TCI boxed warning, but there isn’t much precedent for boxed warnings to be removed from labeling, Dr. Silverberg commented. “Unfortunately, the black-box warning may persist despite a lot of reassuring data.”
In a related editorial, Aaron M. Drucker, MD, ScM, and Mina Tadrous, PharmD, PhD, of the University of Toronto, said the boxed warning “had the intent of helping patients and clinicians understand possible risks,” but also carried the “potential for harm” if patients discontinued or did not adhere to treatment. “Safety warnings on topical medications could lead to undertreatment of atopic dermatitis, reduced quality of life and, potentially, increased use of more toxic systemic medications.”
Long-term studies of medications and cancer risk are challenging to perform, having to account for dose-response relationships, confounding by indication, and time bias, among other factors, and this study “recognizes and attempts to address many of these challenges,” Dr. Drucker and Dr. Tadrous wrote.
These results are similar to previous studies that have “consistently reported no or minimal association between TCI use and skin cancer,” they noted, adding that, “if an association exists, it is likely very small, meaning that skin cancer attributable to TCI use is rare. Clinicians can use this evidence to counsel and reassure patients for whom the benefits of ongoing treatment with TCIs may outweigh the harms.”
This study was funded by a grant from Valeant Pharmaceuticals. Dr. Asgari reported receiving grants from Valeant during the study, and from Pfizer not related to the study. The other authors reported no relevant conflicts of interest. Dr. Drucker reported relationships with the Canadian Agency for Drugs and Technology in Health, CME Outfitters, Eczema Society of Canada, Sanofi, Regeneron, and RTI Health Solutions in the form of paid fees, consultancies, honoraria, educational grants, and other compensation paid to him and/or his institution. Dr. Tadrous reported no relevant disclosures. Dr. Silverberg reported receiving honoraria for advisory board, speaker, and consultant services from numerous pharmaceutical manufacturers, and research grants for investigator services from GlaxoSmithKline and Galderma. Dr. Hughes Tichy reported no relevant financial disclosures. Dr. Silverberg is a member of the Dermatology News editorial advisory board.
SOURCE: Asgari MM et al. JAMA Dermatol. 2020 Aug 12. doi: 10.1001/jamadermatol.2020.2240.
FROM JAMA DERMATOLOGY
Early psychosis: No need for antipsychotics to recover?
Two new studies highlight the importance of early intervention in first-episode psychosis (FEP).
In the first study, Australian investigators conclude that, for some FEP patients, early psychosocial interventions may fend off the need for immediate treatment with antipsychotic medications.
In the second study, UK researchers show that long duration of untreated psychosis (DUP) is linked to a significantly reduced treatment response.
For both studies, the findings highlight the importance of rapid access to a comprehensive range of treatments in the first weeks after FEP onset.
“In a select group of people with first-episode psychosis, we found there was no difference in symptoms and functioning between those who had antipsychotic medication and those who didn’t,” lead author Shona M. Francey, PhD, clinical psychologist at Orygen, the National Center of Excellence in Youth Mental Health, Parkville, Australia, told Medscape Medical News.
“These findings supported our idea that, in the early phases of psychosis, with close monitoring and good psychosocial intervention, antipsychotic medication can be delayed,” Francey said.
The Australian study was published in Schizophrenia Bulletin Open. The British study was published in Lancet Psychiatry.
Adverse effects
Francey and colleagues note that, in comparison with standard treatment, early interventions produce superior outcomes for patients with psychosis. Although there are a variety of treatment options, low-dose second-generation antipsychotics typically play a central role.
However, atypical antipsychotics have rapid metabolic side effects, including weight gain and altered glucose metabolism, that increase the risk for cardiovascular disease and premature mortality. Importantly, such adverse effects are amplified among patients with FEP, who tend to be younger and treatment naive.
On the other hand, a growing body of evidence shows the benefit of nonpharmacologic interventions for patients with FEP, the investigators note. In addition, clinical staging models appear to support the use of less aggressive treatment early in the disease course.
“We have been working in early intervention for psychosis for a number of years and have found it’s possible to intervene early with young people and either prevent the onset of psychosis or ameliorate its impact,” said Francey.
“Since we can see some improvement in people in the prepsychotic phase, we wanted to know if we can also see some benefit without medication after the onset of what we would call full-threshold psychosis,” she added.
Staged Treatment and Acceptability Guidelines in Early Psychosis (STAGES) was a 6-month, triple-blind, randomized controlled noninferiority study that included 90 participants between the ages of 15 and 25 years who had FEP.
To maximize safety, patients were required to have low levels of suicidality and aggression, a DUP of less than 6 months, and to be living in stable accommodation with social support.
Participants were randomly assigned to two groups – one in which patients underwent intensive psychosocial therapy and received low-dose antipsychotic medication (n = 44), and one in which patients underwent intensive psychosocial therapy and were given placebo (n = 46).
Depending on the timing of study enrollment, those in the medication group received risperidone 1 mg or paliperidone 3 mg.
that is strongly focused on therapeutic engagement.
CBCM delivers formulation-driven cognitive-behavioral therapy and psychoeducation within a therapeutic case management framework, Francey said.
The primary outcome was level of functioning at 6, 12, and 24 months, as measured by the Social and Occupational Functioning Scale (SOFAS). The primary prespecified endpoint was outcome at 6 months. A noninferiority margin of 10.5 on the SOFAS was used as the smallest value representing a clinically important effect.
Other assessment tools included the BPRS-4 to test for positive psychotic symptoms, the Scale for the Assessment of Negative Symptoms (SANS), the Hamilton Rating Scale for Depression, and the Hamilton Rating Scale for Anxiety.
At baseline, the two treatment groups were comparable with respect to all measures of functioning and psychopathology.
The study’s discontinuation rate was high. At 6 months, only 16 patients in the psychosocial group had completed therapy, compared with 11 in the antipsychotic group.
At this point, the two groups were comparable in terms of psychopathology and functioning ratings. Both groups had lower symptoms, higher functioning scores, and higher Quality of Life Scale (QLS) scores than at baseline.
SOFAS scores were not significantly different between the groups at this time point. The mean score was 61.7 ± 16.8 in the psychosocial group and 61.5 ± 13.4 in the medication group.
The researchers note that, because the upper limit of the confidence interval (CI) was less than the study’s a priori inferiority margin of 10.5, psychosocial therapy was not inferior to medication at the 6-month assessment point.
Antipsychotics: Use with caution
Although between-group differences in SOFAS scores were not significant at 12 and 24 months, noninferiority of psychosocial therapy alone could not be confirmed because the CIs included the inferiority margin at each time point.
The two groups were statistically comparable at 6 months with respect to all other measures of psychopathology and the QLS. Similar results were found at 12 and 24 months.
The lone exception was with SANS at 12 months, on which patients in the placebo group had significantly higher negative symptom scores than the patients in the medication group.
There were no significant differences between study groups with respect to the number of adverse events.
Francey noted that the findings are important because they suggest that some young people with early-stage FEP and short DUP may be able to achieve symptom remission and function better without antipsychotic medication, provided they receive psychological interventions and comprehensive case management.
This challenges conventional wisdom that antipsychotic medications should be used for all patients who experience psychosis, she added.
However, managing FEP with psychosocial interventions should only be considered when it is safe to do so, Francey noted. In addition, the benefits of psychosocial interventions in these patients are less clear at 12 and 24 months.
Given these caveats, she noted that antipsychotics still play an important role in the treatment of these patients.
“I think there is definitely a place for medications. But I think they should be used cautiously, and you need a good, strong relationship between your treating team and your [patient] to work out what is needed and when it’s needed,” said Francey.
In addition, “when we do use medications, we should use the smallest possible dose that we can and also incorporate psychological support. I think that’s a really important part of it as well,” she said.
Timing matters
In the Lancet Psychiatry study, the researchers note that prolonged DUP is associated with worse outcomes, including increased symptoms, diminished social functioning, and poorer quality of life. The mechanism by which delayed treatment causes more harm remains unclear.
It is possible that symptoms simply accumulate over time, thereby worsening presentation. Another possibility is that continued psychosis after an initial critical period may cause long-term harm, they write.
They hypothesize that untreated psychosis can cause general treatment resistance by exacerbating underlying disease processes and that such damage progresses faster in the early stages of illness and then slows over time.
In addition, socially disruptive symptoms that are evident prior to FEP presentation may have a confounding effect, thereby leading to earlier presentation.
The investigators used data from two longitudinal cohort studies – the National Evaluation of Development of Early Intervention Network (NEDEN) study and the Outlook study.
In the NEDEN trial, 290 of 901 FEP patients (32%) were assessed within 3 weeks of presentation. In Outlook, 69 of 332 patients (21%) were assessed within 3 weeks of presentation.
In both studies, patients were examined at baseline, 6 months, and 12 months using the Positive and Negative Syndrome Scale (PANSS), the Calgary Depression Scale for Schizophrenia, the Mania Rating Scale, the Insight Scale, and SOFAS. The latter two measures were used only at baseline and 12-month follow-up. Logistic regression analyses were used to determine the association between DUP and outcomes.
In the NEDEN study, 751 patients were assessed at 6 months, and 719 were assessed at 12 months. In the Outlook study, 238 and 220 were assessed at the same two time points, respectively.
Results showed a curvilinear relationship between DUP and symptom severity. Longer DUP was predictive of reduced treatment response. However, patient response worsened more slowly as DUP lengthened.
For example, increasing DUP by ten times was predictive of less improvement in PANSS total score by 7.34 (95% CI, 5.76 – 8.92; P < .0001) in NEDEN and by 3.85 (95% CI, 1.69 – 600; P =. 0005) in Outlook. Nevertheless, longer DUP was not associated with worse presentation for any symptoms except depression in NEDEN.
The findings seem to support that the potential harm incurred by delaying treatment among patients with FEP is greatest in the early weeks of psychosis and then levels off, the investigators note.
Given these insights, mental health professionals might consider focusing their efforts on the early detection and treatment of patients for whom DUP is short.
Similarly, because DUP was directly associated with all symptoms, early access to comprehensive treatment “might be preferable to early delivery of particular treatments with particular effects (eg, dopamine antagonists),” they write.
“A pragmatic call”
Commenting on the British study in an accompanying editorial, Lena K. Palaniyappan, MD, University of Western Ontario, London, Canada, and Rajeev Krishnadas, MD, University of Glasgow, Scotland, write that any illness left untreated can become more challenging to treat, including psychosis.
“This should make early intervention in psychosis a pragmatic call with no prima facie argument against it,” they write. A reduction in DUP “underpins the rationale behind early detection and intervention in psychosis.”
The editorialists note that the relationship between DUP and successful treatment in early psychosis “strengthens the argument for more proactive early assessment and intervention to shorten treatment delay.
“As we have learnt over the past two decades, even punctual treatment when symptoms first arise continues to be too late when it comes to psychosis,” they write.
Francey also recognizes the value of early intervention in FEP. However, she noted that comprehensive psychosocial therapy might well prove effective enough to stave off antipsychotic therapy in a certain subset of patients.
“For some people, antipsychotics may never need to be introduced,” she said. “Some people recover from their first episode of psychosis and don’t go on to have any more, while others have an episodic illness,” she said.
If another episode develops and the symptoms come back, further psychosocial interventions could then be tried “or you might want to move on” to psychotic medication “because trying to get people better and functioning as well as they can is our primary aim,” Francey said.
The STAGES study was supported by the Australian National Health and Medical Research Council. The British study was funded by the UK Department of Health, the National Institute of Health Research, and the Medical Research Council. Francey and Krishnadas have reported no relevant financial relationships. Palaniyappan has received grants and personal fees from Janssen Canada and Otsuka Canada, grants from Sunovion, and personal fees from SPMM Course UK and the Canadian Psychiatric Association.
This article first appeared on Medscape.com.
Two new studies highlight the importance of early intervention in first-episode psychosis (FEP).
In the first study, Australian investigators conclude that, for some FEP patients, early psychosocial interventions may fend off the need for immediate treatment with antipsychotic medications.
In the second study, UK researchers show that long duration of untreated psychosis (DUP) is linked to a significantly reduced treatment response.
For both studies, the findings highlight the importance of rapid access to a comprehensive range of treatments in the first weeks after FEP onset.
“In a select group of people with first-episode psychosis, we found there was no difference in symptoms and functioning between those who had antipsychotic medication and those who didn’t,” lead author Shona M. Francey, PhD, clinical psychologist at Orygen, the National Center of Excellence in Youth Mental Health, Parkville, Australia, told Medscape Medical News.
“These findings supported our idea that, in the early phases of psychosis, with close monitoring and good psychosocial intervention, antipsychotic medication can be delayed,” Francey said.
The Australian study was published in Schizophrenia Bulletin Open. The British study was published in Lancet Psychiatry.
Adverse effects
Francey and colleagues note that, in comparison with standard treatment, early interventions produce superior outcomes for patients with psychosis. Although there are a variety of treatment options, low-dose second-generation antipsychotics typically play a central role.
However, atypical antipsychotics have rapid metabolic side effects, including weight gain and altered glucose metabolism, that increase the risk for cardiovascular disease and premature mortality. Importantly, such adverse effects are amplified among patients with FEP, who tend to be younger and treatment naive.
On the other hand, a growing body of evidence shows the benefit of nonpharmacologic interventions for patients with FEP, the investigators note. In addition, clinical staging models appear to support the use of less aggressive treatment early in the disease course.
“We have been working in early intervention for psychosis for a number of years and have found it’s possible to intervene early with young people and either prevent the onset of psychosis or ameliorate its impact,” said Francey.
“Since we can see some improvement in people in the prepsychotic phase, we wanted to know if we can also see some benefit without medication after the onset of what we would call full-threshold psychosis,” she added.
Staged Treatment and Acceptability Guidelines in Early Psychosis (STAGES) was a 6-month, triple-blind, randomized controlled noninferiority study that included 90 participants between the ages of 15 and 25 years who had FEP.
To maximize safety, patients were required to have low levels of suicidality and aggression, a DUP of less than 6 months, and to be living in stable accommodation with social support.
Participants were randomly assigned to two groups – one in which patients underwent intensive psychosocial therapy and received low-dose antipsychotic medication (n = 44), and one in which patients underwent intensive psychosocial therapy and were given placebo (n = 46).
Depending on the timing of study enrollment, those in the medication group received risperidone 1 mg or paliperidone 3 mg.
that is strongly focused on therapeutic engagement.
CBCM delivers formulation-driven cognitive-behavioral therapy and psychoeducation within a therapeutic case management framework, Francey said.
The primary outcome was level of functioning at 6, 12, and 24 months, as measured by the Social and Occupational Functioning Scale (SOFAS). The primary prespecified endpoint was outcome at 6 months. A noninferiority margin of 10.5 on the SOFAS was used as the smallest value representing a clinically important effect.
Other assessment tools included the BPRS-4 to test for positive psychotic symptoms, the Scale for the Assessment of Negative Symptoms (SANS), the Hamilton Rating Scale for Depression, and the Hamilton Rating Scale for Anxiety.
At baseline, the two treatment groups were comparable with respect to all measures of functioning and psychopathology.
The study’s discontinuation rate was high. At 6 months, only 16 patients in the psychosocial group had completed therapy, compared with 11 in the antipsychotic group.
At this point, the two groups were comparable in terms of psychopathology and functioning ratings. Both groups had lower symptoms, higher functioning scores, and higher Quality of Life Scale (QLS) scores than at baseline.
SOFAS scores were not significantly different between the groups at this time point. The mean score was 61.7 ± 16.8 in the psychosocial group and 61.5 ± 13.4 in the medication group.
The researchers note that, because the upper limit of the confidence interval (CI) was less than the study’s a priori inferiority margin of 10.5, psychosocial therapy was not inferior to medication at the 6-month assessment point.
Antipsychotics: Use with caution
Although between-group differences in SOFAS scores were not significant at 12 and 24 months, noninferiority of psychosocial therapy alone could not be confirmed because the CIs included the inferiority margin at each time point.
The two groups were statistically comparable at 6 months with respect to all other measures of psychopathology and the QLS. Similar results were found at 12 and 24 months.
The lone exception was with SANS at 12 months, on which patients in the placebo group had significantly higher negative symptom scores than the patients in the medication group.
There were no significant differences between study groups with respect to the number of adverse events.
Francey noted that the findings are important because they suggest that some young people with early-stage FEP and short DUP may be able to achieve symptom remission and function better without antipsychotic medication, provided they receive psychological interventions and comprehensive case management.
This challenges conventional wisdom that antipsychotic medications should be used for all patients who experience psychosis, she added.
However, managing FEP with psychosocial interventions should only be considered when it is safe to do so, Francey noted. In addition, the benefits of psychosocial interventions in these patients are less clear at 12 and 24 months.
Given these caveats, she noted that antipsychotics still play an important role in the treatment of these patients.
“I think there is definitely a place for medications. But I think they should be used cautiously, and you need a good, strong relationship between your treating team and your [patient] to work out what is needed and when it’s needed,” said Francey.
In addition, “when we do use medications, we should use the smallest possible dose that we can and also incorporate psychological support. I think that’s a really important part of it as well,” she said.
Timing matters
In the Lancet Psychiatry study, the researchers note that prolonged DUP is associated with worse outcomes, including increased symptoms, diminished social functioning, and poorer quality of life. The mechanism by which delayed treatment causes more harm remains unclear.
It is possible that symptoms simply accumulate over time, thereby worsening presentation. Another possibility is that continued psychosis after an initial critical period may cause long-term harm, they write.
They hypothesize that untreated psychosis can cause general treatment resistance by exacerbating underlying disease processes and that such damage progresses faster in the early stages of illness and then slows over time.
In addition, socially disruptive symptoms that are evident prior to FEP presentation may have a confounding effect, thereby leading to earlier presentation.
The investigators used data from two longitudinal cohort studies – the National Evaluation of Development of Early Intervention Network (NEDEN) study and the Outlook study.
In the NEDEN trial, 290 of 901 FEP patients (32%) were assessed within 3 weeks of presentation. In Outlook, 69 of 332 patients (21%) were assessed within 3 weeks of presentation.
In both studies, patients were examined at baseline, 6 months, and 12 months using the Positive and Negative Syndrome Scale (PANSS), the Calgary Depression Scale for Schizophrenia, the Mania Rating Scale, the Insight Scale, and SOFAS. The latter two measures were used only at baseline and 12-month follow-up. Logistic regression analyses were used to determine the association between DUP and outcomes.
In the NEDEN study, 751 patients were assessed at 6 months, and 719 were assessed at 12 months. In the Outlook study, 238 and 220 were assessed at the same two time points, respectively.
Results showed a curvilinear relationship between DUP and symptom severity. Longer DUP was predictive of reduced treatment response. However, patient response worsened more slowly as DUP lengthened.
For example, increasing DUP by ten times was predictive of less improvement in PANSS total score by 7.34 (95% CI, 5.76 – 8.92; P < .0001) in NEDEN and by 3.85 (95% CI, 1.69 – 600; P =. 0005) in Outlook. Nevertheless, longer DUP was not associated with worse presentation for any symptoms except depression in NEDEN.
The findings seem to support that the potential harm incurred by delaying treatment among patients with FEP is greatest in the early weeks of psychosis and then levels off, the investigators note.
Given these insights, mental health professionals might consider focusing their efforts on the early detection and treatment of patients for whom DUP is short.
Similarly, because DUP was directly associated with all symptoms, early access to comprehensive treatment “might be preferable to early delivery of particular treatments with particular effects (eg, dopamine antagonists),” they write.
“A pragmatic call”
Commenting on the British study in an accompanying editorial, Lena K. Palaniyappan, MD, University of Western Ontario, London, Canada, and Rajeev Krishnadas, MD, University of Glasgow, Scotland, write that any illness left untreated can become more challenging to treat, including psychosis.
“This should make early intervention in psychosis a pragmatic call with no prima facie argument against it,” they write. A reduction in DUP “underpins the rationale behind early detection and intervention in psychosis.”
The editorialists note that the relationship between DUP and successful treatment in early psychosis “strengthens the argument for more proactive early assessment and intervention to shorten treatment delay.
“As we have learnt over the past two decades, even punctual treatment when symptoms first arise continues to be too late when it comes to psychosis,” they write.
Francey also recognizes the value of early intervention in FEP. However, she noted that comprehensive psychosocial therapy might well prove effective enough to stave off antipsychotic therapy in a certain subset of patients.
“For some people, antipsychotics may never need to be introduced,” she said. “Some people recover from their first episode of psychosis and don’t go on to have any more, while others have an episodic illness,” she said.
If another episode develops and the symptoms come back, further psychosocial interventions could then be tried “or you might want to move on” to psychotic medication “because trying to get people better and functioning as well as they can is our primary aim,” Francey said.
The STAGES study was supported by the Australian National Health and Medical Research Council. The British study was funded by the UK Department of Health, the National Institute of Health Research, and the Medical Research Council. Francey and Krishnadas have reported no relevant financial relationships. Palaniyappan has received grants and personal fees from Janssen Canada and Otsuka Canada, grants from Sunovion, and personal fees from SPMM Course UK and the Canadian Psychiatric Association.
This article first appeared on Medscape.com.
Two new studies highlight the importance of early intervention in first-episode psychosis (FEP).
In the first study, Australian investigators conclude that, for some FEP patients, early psychosocial interventions may fend off the need for immediate treatment with antipsychotic medications.
In the second study, UK researchers show that long duration of untreated psychosis (DUP) is linked to a significantly reduced treatment response.
For both studies, the findings highlight the importance of rapid access to a comprehensive range of treatments in the first weeks after FEP onset.
“In a select group of people with first-episode psychosis, we found there was no difference in symptoms and functioning between those who had antipsychotic medication and those who didn’t,” lead author Shona M. Francey, PhD, clinical psychologist at Orygen, the National Center of Excellence in Youth Mental Health, Parkville, Australia, told Medscape Medical News.
“These findings supported our idea that, in the early phases of psychosis, with close monitoring and good psychosocial intervention, antipsychotic medication can be delayed,” Francey said.
The Australian study was published in Schizophrenia Bulletin Open. The British study was published in Lancet Psychiatry.
Adverse effects
Francey and colleagues note that, in comparison with standard treatment, early interventions produce superior outcomes for patients with psychosis. Although there are a variety of treatment options, low-dose second-generation antipsychotics typically play a central role.
However, atypical antipsychotics have rapid metabolic side effects, including weight gain and altered glucose metabolism, that increase the risk for cardiovascular disease and premature mortality. Importantly, such adverse effects are amplified among patients with FEP, who tend to be younger and treatment naive.
On the other hand, a growing body of evidence shows the benefit of nonpharmacologic interventions for patients with FEP, the investigators note. In addition, clinical staging models appear to support the use of less aggressive treatment early in the disease course.
“We have been working in early intervention for psychosis for a number of years and have found it’s possible to intervene early with young people and either prevent the onset of psychosis or ameliorate its impact,” said Francey.
“Since we can see some improvement in people in the prepsychotic phase, we wanted to know if we can also see some benefit without medication after the onset of what we would call full-threshold psychosis,” she added.
Staged Treatment and Acceptability Guidelines in Early Psychosis (STAGES) was a 6-month, triple-blind, randomized controlled noninferiority study that included 90 participants between the ages of 15 and 25 years who had FEP.
To maximize safety, patients were required to have low levels of suicidality and aggression, a DUP of less than 6 months, and to be living in stable accommodation with social support.
Participants were randomly assigned to two groups – one in which patients underwent intensive psychosocial therapy and received low-dose antipsychotic medication (n = 44), and one in which patients underwent intensive psychosocial therapy and were given placebo (n = 46).
Depending on the timing of study enrollment, those in the medication group received risperidone 1 mg or paliperidone 3 mg.
that is strongly focused on therapeutic engagement.
CBCM delivers formulation-driven cognitive-behavioral therapy and psychoeducation within a therapeutic case management framework, Francey said.
The primary outcome was level of functioning at 6, 12, and 24 months, as measured by the Social and Occupational Functioning Scale (SOFAS). The primary prespecified endpoint was outcome at 6 months. A noninferiority margin of 10.5 on the SOFAS was used as the smallest value representing a clinically important effect.
Other assessment tools included the BPRS-4 to test for positive psychotic symptoms, the Scale for the Assessment of Negative Symptoms (SANS), the Hamilton Rating Scale for Depression, and the Hamilton Rating Scale for Anxiety.
At baseline, the two treatment groups were comparable with respect to all measures of functioning and psychopathology.
The study’s discontinuation rate was high. At 6 months, only 16 patients in the psychosocial group had completed therapy, compared with 11 in the antipsychotic group.
At this point, the two groups were comparable in terms of psychopathology and functioning ratings. Both groups had lower symptoms, higher functioning scores, and higher Quality of Life Scale (QLS) scores than at baseline.
SOFAS scores were not significantly different between the groups at this time point. The mean score was 61.7 ± 16.8 in the psychosocial group and 61.5 ± 13.4 in the medication group.
The researchers note that, because the upper limit of the confidence interval (CI) was less than the study’s a priori inferiority margin of 10.5, psychosocial therapy was not inferior to medication at the 6-month assessment point.
Antipsychotics: Use with caution
Although between-group differences in SOFAS scores were not significant at 12 and 24 months, noninferiority of psychosocial therapy alone could not be confirmed because the CIs included the inferiority margin at each time point.
The two groups were statistically comparable at 6 months with respect to all other measures of psychopathology and the QLS. Similar results were found at 12 and 24 months.
The lone exception was with SANS at 12 months, on which patients in the placebo group had significantly higher negative symptom scores than the patients in the medication group.
There were no significant differences between study groups with respect to the number of adverse events.
Francey noted that the findings are important because they suggest that some young people with early-stage FEP and short DUP may be able to achieve symptom remission and function better without antipsychotic medication, provided they receive psychological interventions and comprehensive case management.
This challenges conventional wisdom that antipsychotic medications should be used for all patients who experience psychosis, she added.
However, managing FEP with psychosocial interventions should only be considered when it is safe to do so, Francey noted. In addition, the benefits of psychosocial interventions in these patients are less clear at 12 and 24 months.
Given these caveats, she noted that antipsychotics still play an important role in the treatment of these patients.
“I think there is definitely a place for medications. But I think they should be used cautiously, and you need a good, strong relationship between your treating team and your [patient] to work out what is needed and when it’s needed,” said Francey.
In addition, “when we do use medications, we should use the smallest possible dose that we can and also incorporate psychological support. I think that’s a really important part of it as well,” she said.
Timing matters
In the Lancet Psychiatry study, the researchers note that prolonged DUP is associated with worse outcomes, including increased symptoms, diminished social functioning, and poorer quality of life. The mechanism by which delayed treatment causes more harm remains unclear.
It is possible that symptoms simply accumulate over time, thereby worsening presentation. Another possibility is that continued psychosis after an initial critical period may cause long-term harm, they write.
They hypothesize that untreated psychosis can cause general treatment resistance by exacerbating underlying disease processes and that such damage progresses faster in the early stages of illness and then slows over time.
In addition, socially disruptive symptoms that are evident prior to FEP presentation may have a confounding effect, thereby leading to earlier presentation.
The investigators used data from two longitudinal cohort studies – the National Evaluation of Development of Early Intervention Network (NEDEN) study and the Outlook study.
In the NEDEN trial, 290 of 901 FEP patients (32%) were assessed within 3 weeks of presentation. In Outlook, 69 of 332 patients (21%) were assessed within 3 weeks of presentation.
In both studies, patients were examined at baseline, 6 months, and 12 months using the Positive and Negative Syndrome Scale (PANSS), the Calgary Depression Scale for Schizophrenia, the Mania Rating Scale, the Insight Scale, and SOFAS. The latter two measures were used only at baseline and 12-month follow-up. Logistic regression analyses were used to determine the association between DUP and outcomes.
In the NEDEN study, 751 patients were assessed at 6 months, and 719 were assessed at 12 months. In the Outlook study, 238 and 220 were assessed at the same two time points, respectively.
Results showed a curvilinear relationship between DUP and symptom severity. Longer DUP was predictive of reduced treatment response. However, patient response worsened more slowly as DUP lengthened.
For example, increasing DUP by ten times was predictive of less improvement in PANSS total score by 7.34 (95% CI, 5.76 – 8.92; P < .0001) in NEDEN and by 3.85 (95% CI, 1.69 – 600; P =. 0005) in Outlook. Nevertheless, longer DUP was not associated with worse presentation for any symptoms except depression in NEDEN.
The findings seem to support that the potential harm incurred by delaying treatment among patients with FEP is greatest in the early weeks of psychosis and then levels off, the investigators note.
Given these insights, mental health professionals might consider focusing their efforts on the early detection and treatment of patients for whom DUP is short.
Similarly, because DUP was directly associated with all symptoms, early access to comprehensive treatment “might be preferable to early delivery of particular treatments with particular effects (eg, dopamine antagonists),” they write.
“A pragmatic call”
Commenting on the British study in an accompanying editorial, Lena K. Palaniyappan, MD, University of Western Ontario, London, Canada, and Rajeev Krishnadas, MD, University of Glasgow, Scotland, write that any illness left untreated can become more challenging to treat, including psychosis.
“This should make early intervention in psychosis a pragmatic call with no prima facie argument against it,” they write. A reduction in DUP “underpins the rationale behind early detection and intervention in psychosis.”
The editorialists note that the relationship between DUP and successful treatment in early psychosis “strengthens the argument for more proactive early assessment and intervention to shorten treatment delay.
“As we have learnt over the past two decades, even punctual treatment when symptoms first arise continues to be too late when it comes to psychosis,” they write.
Francey also recognizes the value of early intervention in FEP. However, she noted that comprehensive psychosocial therapy might well prove effective enough to stave off antipsychotic therapy in a certain subset of patients.
“For some people, antipsychotics may never need to be introduced,” she said. “Some people recover from their first episode of psychosis and don’t go on to have any more, while others have an episodic illness,” she said.
If another episode develops and the symptoms come back, further psychosocial interventions could then be tried “or you might want to move on” to psychotic medication “because trying to get people better and functioning as well as they can is our primary aim,” Francey said.
The STAGES study was supported by the Australian National Health and Medical Research Council. The British study was funded by the UK Department of Health, the National Institute of Health Research, and the Medical Research Council. Francey and Krishnadas have reported no relevant financial relationships. Palaniyappan has received grants and personal fees from Janssen Canada and Otsuka Canada, grants from Sunovion, and personal fees from SPMM Course UK and the Canadian Psychiatric Association.
This article first appeared on Medscape.com.
Why are many of my patients doing better during the pandemic?
The COVID-19 pandemic has, like it or not, made experimental labs rats out of us all.
Since the U.S. “shutdown” began in March, we have all had to adjust to a situation in which we are home more, stuck seeing less of our friends, exercising less, often eating and drinking more, or using recreational substances more – in part because of the severe stress. We have been ripped away from many of the social “anchors” of our weeks; that is, the spiritual, social and physical, and tactile supports that sustain and motivate us in our lives.
And yet, many of us, of all ages, stripes, and colors are thriving. Why is that so? Without necessarily being fully fledged, card carrying misanthropes, many of us are actually not bereft when forced to spend some alone time.
We may be self-starters and have hobbies and interests that we may have neglected but can fall back on with alacrity. Activities such as gardening, cooking, reading, working at our day jobs, listening to music, streaming TV, and so on are now more available to us.
The pandemic has produced unforeseen side effects, such as decreased pollution, less seismic “noise” on our planet, increasingly bold activity by wild life, and we can actually hear bird songs in our yards. Likewise, the social isolation has enabled us to focus more on “back burner” projects and to motivate us toward accessing and achieving other internally driven goals.
Also, to many, it has provided a surprising and unexpected privilege to meaningfully connect while in close quarters with spouses, children, and other loved ones, which has improved and cemented relationships under some level of duress, perhaps.
Similarly, and perhaps surprisingly, in addition to the above reasons, many of our patients with chronic mental illness may be functioning reasonably well, too, even better than their “walking wounded” loved ones and peers. They may be reaping the rewards of many years of consistent biopsychosocial support in strong mental health programs.
But another reason might be the lowered expectations. I’m just so much more relaxed; I’ve got this.” And certainly the Freudian “schadenfreude” defense has something to do with this as well. Seeing family members lose their jobs, become financially vulnerable, being unable to or stymied from demonstrating mastery in many different situations and skill sets elicit the empathy and galvanizes the support of well-managed patients with mental illness – already used to existential threats – for their generally higher functioning loved ones.
As one of my struggling patients said, “Welcome to my world!” Years of hardship, lack of intimate relationships because of social anxiety, and psychotic level obsessive-compulsive disorder have trained, indeed, inured her to the daily pain, constriction, and misery of social isolation. Her life, despite working full time, has remained static, while younger siblings have married, started a family, moved away. She is still living at home with her elderly parents. They now worry about catching COVID-19, while she is now their protector with roles reversed, doing their shopping, and providing moral support and encouragement for the whole family.
Many of us have lost jobs, been furloughed, seen our dreams disappear, and are unable to pay rent or mortgages. Those with chronic mental illness, especially those living in states with a strong social safety net, are continuing to receive their Social Security disability checks, and maintain their in-home health and family supports. They also have continued their adherence with the mental health system structure by continuing with telemedicine therapy and regular medications or monthly intramuscular shots. Their families are especially cognizant of the need for ongoing structure and stability, which is now easier to provide. And what of those patients who endured severe anxiety and panic disorders in their prepandemic states? It is true that many do require higher doses of their anxiolytics, especially benzodiazepines. They do know how to “roll with the punches” with their lifetime experience, as opposed to the “newbies” whose incipient anxiety is brought to the forefront and who might not even recognize these debilitating symptoms and are not keen, for reasons of stigma, to be seen by a mental health expert unless compelled to.
It is up to us as psychiatrists and other mental health clinicians to minimize dependence on those medications by using alternative non–dependence-forming anxiolytics and encouraging our patients to hone and develop the skills from cognitive-behavioral therapy. COVID-19 is just one more stressor, superimposed on many others, and unlikely to precipitate any “tipping point” in functioning, even if there are significant losses among loved ones to the virus.
How about our child and adolescent patients? As a rule of thumb, those with anxiety disorders, social anxiety, selective mutism – and those experiencing challenges and bullying in the rough and tumble world of schools – are doing significantly better. Those with ADHD and impulse control disorders, however, might be struggling with school, especially with Zoom calls and very high distractibility, boredom, and motivational challenges. They may need their doses of medications adjusted up, and their parents are struggling. The risk for unwitnessed and unmonitored abuse in home situations is higher.
Those with chronic mental illness often do have increased risk factors for COVID-19 that might be compounded by their psychopharmacologic treatment for conditions/behaviors such as diabetes, obesity, cardiovascular disease, and substance use. By proactively monitoring those comorbid disorders in a multimodal treatment program, we can help mitigate those baseline challenges.
This aspect of the COVID-19 pandemic is, alas, likely to prove to be an illusory positive “blip” on the radar screen for many with chronic mental illness. Nevertheless, the self-knowledge and awareness of hidden strengths rather than weakness, resilience rather than shrinking from challenges, is not insignificant. This “flight into normality” may be a change that can be internalized and nurtured once vaccines are available and life on planet Earth returns to a new normal.
Dr. Tofler is affiliated with Kaiser Permanente Psychiatry in Los Angeles. He also is a visiting faculty member in the department of psychiatry and biobehavioral sciences at the University of California, Los Angeles. Dr. Tofler has no conflicts of interest.
The COVID-19 pandemic has, like it or not, made experimental labs rats out of us all.
Since the U.S. “shutdown” began in March, we have all had to adjust to a situation in which we are home more, stuck seeing less of our friends, exercising less, often eating and drinking more, or using recreational substances more – in part because of the severe stress. We have been ripped away from many of the social “anchors” of our weeks; that is, the spiritual, social and physical, and tactile supports that sustain and motivate us in our lives.
And yet, many of us, of all ages, stripes, and colors are thriving. Why is that so? Without necessarily being fully fledged, card carrying misanthropes, many of us are actually not bereft when forced to spend some alone time.
We may be self-starters and have hobbies and interests that we may have neglected but can fall back on with alacrity. Activities such as gardening, cooking, reading, working at our day jobs, listening to music, streaming TV, and so on are now more available to us.
The pandemic has produced unforeseen side effects, such as decreased pollution, less seismic “noise” on our planet, increasingly bold activity by wild life, and we can actually hear bird songs in our yards. Likewise, the social isolation has enabled us to focus more on “back burner” projects and to motivate us toward accessing and achieving other internally driven goals.
Also, to many, it has provided a surprising and unexpected privilege to meaningfully connect while in close quarters with spouses, children, and other loved ones, which has improved and cemented relationships under some level of duress, perhaps.
Similarly, and perhaps surprisingly, in addition to the above reasons, many of our patients with chronic mental illness may be functioning reasonably well, too, even better than their “walking wounded” loved ones and peers. They may be reaping the rewards of many years of consistent biopsychosocial support in strong mental health programs.
But another reason might be the lowered expectations. I’m just so much more relaxed; I’ve got this.” And certainly the Freudian “schadenfreude” defense has something to do with this as well. Seeing family members lose their jobs, become financially vulnerable, being unable to or stymied from demonstrating mastery in many different situations and skill sets elicit the empathy and galvanizes the support of well-managed patients with mental illness – already used to existential threats – for their generally higher functioning loved ones.
As one of my struggling patients said, “Welcome to my world!” Years of hardship, lack of intimate relationships because of social anxiety, and psychotic level obsessive-compulsive disorder have trained, indeed, inured her to the daily pain, constriction, and misery of social isolation. Her life, despite working full time, has remained static, while younger siblings have married, started a family, moved away. She is still living at home with her elderly parents. They now worry about catching COVID-19, while she is now their protector with roles reversed, doing their shopping, and providing moral support and encouragement for the whole family.
Many of us have lost jobs, been furloughed, seen our dreams disappear, and are unable to pay rent or mortgages. Those with chronic mental illness, especially those living in states with a strong social safety net, are continuing to receive their Social Security disability checks, and maintain their in-home health and family supports. They also have continued their adherence with the mental health system structure by continuing with telemedicine therapy and regular medications or monthly intramuscular shots. Their families are especially cognizant of the need for ongoing structure and stability, which is now easier to provide. And what of those patients who endured severe anxiety and panic disorders in their prepandemic states? It is true that many do require higher doses of their anxiolytics, especially benzodiazepines. They do know how to “roll with the punches” with their lifetime experience, as opposed to the “newbies” whose incipient anxiety is brought to the forefront and who might not even recognize these debilitating symptoms and are not keen, for reasons of stigma, to be seen by a mental health expert unless compelled to.
It is up to us as psychiatrists and other mental health clinicians to minimize dependence on those medications by using alternative non–dependence-forming anxiolytics and encouraging our patients to hone and develop the skills from cognitive-behavioral therapy. COVID-19 is just one more stressor, superimposed on many others, and unlikely to precipitate any “tipping point” in functioning, even if there are significant losses among loved ones to the virus.
How about our child and adolescent patients? As a rule of thumb, those with anxiety disorders, social anxiety, selective mutism – and those experiencing challenges and bullying in the rough and tumble world of schools – are doing significantly better. Those with ADHD and impulse control disorders, however, might be struggling with school, especially with Zoom calls and very high distractibility, boredom, and motivational challenges. They may need their doses of medications adjusted up, and their parents are struggling. The risk for unwitnessed and unmonitored abuse in home situations is higher.
Those with chronic mental illness often do have increased risk factors for COVID-19 that might be compounded by their psychopharmacologic treatment for conditions/behaviors such as diabetes, obesity, cardiovascular disease, and substance use. By proactively monitoring those comorbid disorders in a multimodal treatment program, we can help mitigate those baseline challenges.
This aspect of the COVID-19 pandemic is, alas, likely to prove to be an illusory positive “blip” on the radar screen for many with chronic mental illness. Nevertheless, the self-knowledge and awareness of hidden strengths rather than weakness, resilience rather than shrinking from challenges, is not insignificant. This “flight into normality” may be a change that can be internalized and nurtured once vaccines are available and life on planet Earth returns to a new normal.
Dr. Tofler is affiliated with Kaiser Permanente Psychiatry in Los Angeles. He also is a visiting faculty member in the department of psychiatry and biobehavioral sciences at the University of California, Los Angeles. Dr. Tofler has no conflicts of interest.
The COVID-19 pandemic has, like it or not, made experimental labs rats out of us all.
Since the U.S. “shutdown” began in March, we have all had to adjust to a situation in which we are home more, stuck seeing less of our friends, exercising less, often eating and drinking more, or using recreational substances more – in part because of the severe stress. We have been ripped away from many of the social “anchors” of our weeks; that is, the spiritual, social and physical, and tactile supports that sustain and motivate us in our lives.
And yet, many of us, of all ages, stripes, and colors are thriving. Why is that so? Without necessarily being fully fledged, card carrying misanthropes, many of us are actually not bereft when forced to spend some alone time.
We may be self-starters and have hobbies and interests that we may have neglected but can fall back on with alacrity. Activities such as gardening, cooking, reading, working at our day jobs, listening to music, streaming TV, and so on are now more available to us.
The pandemic has produced unforeseen side effects, such as decreased pollution, less seismic “noise” on our planet, increasingly bold activity by wild life, and we can actually hear bird songs in our yards. Likewise, the social isolation has enabled us to focus more on “back burner” projects and to motivate us toward accessing and achieving other internally driven goals.
Also, to many, it has provided a surprising and unexpected privilege to meaningfully connect while in close quarters with spouses, children, and other loved ones, which has improved and cemented relationships under some level of duress, perhaps.
Similarly, and perhaps surprisingly, in addition to the above reasons, many of our patients with chronic mental illness may be functioning reasonably well, too, even better than their “walking wounded” loved ones and peers. They may be reaping the rewards of many years of consistent biopsychosocial support in strong mental health programs.
But another reason might be the lowered expectations. I’m just so much more relaxed; I’ve got this.” And certainly the Freudian “schadenfreude” defense has something to do with this as well. Seeing family members lose their jobs, become financially vulnerable, being unable to or stymied from demonstrating mastery in many different situations and skill sets elicit the empathy and galvanizes the support of well-managed patients with mental illness – already used to existential threats – for their generally higher functioning loved ones.
As one of my struggling patients said, “Welcome to my world!” Years of hardship, lack of intimate relationships because of social anxiety, and psychotic level obsessive-compulsive disorder have trained, indeed, inured her to the daily pain, constriction, and misery of social isolation. Her life, despite working full time, has remained static, while younger siblings have married, started a family, moved away. She is still living at home with her elderly parents. They now worry about catching COVID-19, while she is now their protector with roles reversed, doing their shopping, and providing moral support and encouragement for the whole family.
Many of us have lost jobs, been furloughed, seen our dreams disappear, and are unable to pay rent or mortgages. Those with chronic mental illness, especially those living in states with a strong social safety net, are continuing to receive their Social Security disability checks, and maintain their in-home health and family supports. They also have continued their adherence with the mental health system structure by continuing with telemedicine therapy and regular medications or monthly intramuscular shots. Their families are especially cognizant of the need for ongoing structure and stability, which is now easier to provide. And what of those patients who endured severe anxiety and panic disorders in their prepandemic states? It is true that many do require higher doses of their anxiolytics, especially benzodiazepines. They do know how to “roll with the punches” with their lifetime experience, as opposed to the “newbies” whose incipient anxiety is brought to the forefront and who might not even recognize these debilitating symptoms and are not keen, for reasons of stigma, to be seen by a mental health expert unless compelled to.
It is up to us as psychiatrists and other mental health clinicians to minimize dependence on those medications by using alternative non–dependence-forming anxiolytics and encouraging our patients to hone and develop the skills from cognitive-behavioral therapy. COVID-19 is just one more stressor, superimposed on many others, and unlikely to precipitate any “tipping point” in functioning, even if there are significant losses among loved ones to the virus.
How about our child and adolescent patients? As a rule of thumb, those with anxiety disorders, social anxiety, selective mutism – and those experiencing challenges and bullying in the rough and tumble world of schools – are doing significantly better. Those with ADHD and impulse control disorders, however, might be struggling with school, especially with Zoom calls and very high distractibility, boredom, and motivational challenges. They may need their doses of medications adjusted up, and their parents are struggling. The risk for unwitnessed and unmonitored abuse in home situations is higher.
Those with chronic mental illness often do have increased risk factors for COVID-19 that might be compounded by their psychopharmacologic treatment for conditions/behaviors such as diabetes, obesity, cardiovascular disease, and substance use. By proactively monitoring those comorbid disorders in a multimodal treatment program, we can help mitigate those baseline challenges.
This aspect of the COVID-19 pandemic is, alas, likely to prove to be an illusory positive “blip” on the radar screen for many with chronic mental illness. Nevertheless, the self-knowledge and awareness of hidden strengths rather than weakness, resilience rather than shrinking from challenges, is not insignificant. This “flight into normality” may be a change that can be internalized and nurtured once vaccines are available and life on planet Earth returns to a new normal.
Dr. Tofler is affiliated with Kaiser Permanente Psychiatry in Los Angeles. He also is a visiting faculty member in the department of psychiatry and biobehavioral sciences at the University of California, Los Angeles. Dr. Tofler has no conflicts of interest.
When viruses collide: Flu season during pandemic
The medical community is about to find out how prepared it is for the double whammy of influenza and COVID-19 that has been predicted for the fall of 2020. The complexities of diagnosis, management of vulnerable patients, and overflowing medical centers that have made the COVID-19 crisis so brutal may all be exacerbated by the arrival of seasonal influenza.
Lewis Jay Kaplan, MD, FCCP, a critical care surgeon at the University of Pennsylvania, Philadelphia, has seen his share of critically ill COVID-19 patients in the surgical ICU that he oversees. He’s approaching the upcoming flu season, poised to collide with the ongoing COVID-19 pandemic, ready to listen to each patient’s story to distinguish one from the other and determine treatment.
“The patients that have underlying comorbidities all have a story, and it’s up to you to figure out which chapter you’re in and how far along you happen to be,” he said. “It’s a very interesting approach to care, medical storytelling.”
With flu season closing in, pulmonologists are ruminating about how they’ll distinguish symptoms of COVID-19 and traditional influenza and how they’ll manage the most vulnerable patients, namely those with underlying respiratory disease and children. Influenza kills 12,000-61,000 people a year, according to the Centers for Disease Control, and results in 140,000-810,00 hospitalizations. Having a flu season in the midst of a pandemic of a disease with multiple overlapping symptoms threatens to overwhelm practitioners, hospitals, and the health system.
Dr. Kaplan said each patient’s story can point to the correct clinical approach. “Instead of just sharing data when you are on rounds, you’re really telling someone’s story.” It arises from a series of questions about how the disease has impacted them, specifics of their presentation, how their signs and symptoms differ from the usual, and how they responded to treatment. “It also helps you to then take what you’re doing, which can seem very, very complicated to individuals who are not medically sophisticated, and then help them to understand why you’re doing what you’re doing at this point.”
That can help get through to a patient with respiratory disease who insists he or she has or doesn’t have COVID-19 rather than the flu. “They form a different group that brings with them different fears and concerns, and you have to help them navigate that, too: all of this data and your decision-making around testing and admissions, and what you can omit doing and what you must do help them to navigate their own story,” Dr. Kaplan said.
Benjamin D. Singer, MD, a pulmonologist at Northwestern University, Chicago, authored an editorial in Science Advances that addressed four factors that will determine the scope of flu spread in the upcoming season: rate of transmission; vaccination rates; coinfection rates; and health disparities in minority populations, which are prone to higher rates of flu as well as COVID-19.
Flu vaccine ‘extra important’
The convergence of COVID-19 and influenza has the potential to overwhelm the health system, said Daniel A. Solomon, MD, of Brigham and Women’s in Boston. He coauthored a JAMA Insights clinical update on flu season during the COVID-19 pandemic that lists distinguishing and overlapping signs and symptoms of the two diseases.
The flu vaccine, he said, is “extra important this year,” especially in patients with existing respiratory disease, but COVID-19 has thrown up barriers to vaccination. Telemedicine has supplanted office visits. “People may miss that easy-touch opportunity to get the flu vaccine, so we have to be creative about making the flu vaccine highly accessible, maybe in nontraditional ways,” Dr. Solomon said. Some ideas he offered are pop-up vaccine fairs at schools and churches.
But just as COVID-19 may hinder flu vaccines, it may also be helping to mitigate flu transmission. “The interesting thing about transmission of the flu is that it’s transmitted the same way COVID is, so if we actually know how to decrease transmission of COVID, which we do – we’ve done it – we can actually decrease transmission of influenza as well,” Dr. Solomon said. Studies out of Hong Kong and Japan have reported a reduction in influenza cases during COVID-19 outbreaks in those places (Lancet Public Health. 2020;5:e279-88; JAMA. 2020;323:1969-71).
Risks of coinfection
About one in four COVID-19 patients have been diagnosed with an additional respiratory infection, including influenza (JAMA. 2020:323:2085-6). Pulmonologists must keep that in mind when managing COVID-19 suspects, said Dr. Singer.
“While it is true that most of the time COVID-19 travels alone, we have numerous examples in the literature and in our own experience that COVID-19 is accompanied by either another virus or another bacterial infection, including influenza,” Dr. Singer said. “The distinction is important. One is just for diagnostic reasons and public reporting reasons, but also because flu and COVID-19 have different requirements for how you care for patients in terms of the health system.”
Clinical suspicion for coinfection should remain high if the community spread of both COVID-19 and influenza is high, said Megan Conroy, MD, chief pulmonary and critical care fellow at Ohio State University, Columbus. “As the coronavirus first took hold in the United States in March 2020, we were at the tail end of influenza season, so it’s hard to predict what the upcoming influenza season will really look like with regards to coinfection.”
Distinguishing COVID-19 from flu
Multiple signs and symptoms between COVID-19 and the flu overlap. They include fever, chills, headache, myalgia, cough, and fatigue. Nasal congestion and sore throat are characteristic of the flu; shortness of breath and loss of the sense of smell have been widely reported in COVID-19. “While many upper respiratory infections can result in loss of smell, this may be more prevalent in COVID-19,” Dr. Conroy said. Other symptoms unique to COVID-19 are GI symptoms such as diarrhea and skin rashes such as acral ischemia.
Testing, however, is the cornerstone of the differential diagnosis. “You can’t confidently distinguish between them on symptoms alone,” Dr. Conroy added.
“I think the challenge we’ll face as clinicians, is caring for people with nonspecific symptoms of a respiratory viral illness, especially in the early phase of the illness,” said Dr. Solomon.
But even after that, symptoms can be difficult to distinguish.
“Later in the illness, COVID is more associated with a hypercoagulable state,” he said. “It is more associated with viral pneumonia on chest imaging, like the diffuse ground-glass infiltrates that we’ve all gotten used to seeing – but flu can do both of those things as well. So, without a test, it’s impossible to distinguish between the two infections in the clinic.”
But testing can have its shortcomings when flu season clashes with the COVID-19 pandemic. “Getting the test is not the same as getting the test results,” Dr. Solomon added. “Though a lot of people can get a test, if it takes 7 or 8 days to get the test result back, the result is useless.”
Widespread, rapid testing also depends on having adequate supplies of viral media transport and swabs. “I think that this is what we should be focusing on now: scaling up access to rapid turnaround testing,” he said. Distinguishing between the two is also important to preserve hospital resources. COVID-19 has more rigorous standards than flu for personal protective equipment and isolation of patients within the hospital.
Having chronic lung disease isn’t necessarily a risk factor for contracting COVID-19 or the flu, or both, Dr. Solomon said. “It’s a risk factor for having severe disease.” Again, he noted that flu vaccines are still necessary in these patients, as well as patients of advanced age and underlying medical conditions such as heart disease, diabetes, and obesity.
In managing children, it’s important to keep in mind that they communicate differently about their illnesses than adults, said Dr. Kaplan. “They may not have the words to tell you the same kind of thing that the adult tells you.” That’s where family members can help to flesh out the history. “They may present with an initially much milder form, if you will, where they’re not as critical up front, but then that small proportion of them comes back with the multi-inflammatory syndrome and then they are profoundly ill.”
Younger people make up a larger share of COVID-19 patients now, compared with the initial wave that hit the Northeast in the spring, Dr. Kaplan said. “We don’t know if that’s because the virus is a little different or the people that are getting sick are a little bit different.”
The COVID-19 strain now emerging may be less virulent than the strain that hit in early spring, he said. “That doesn’t mean that there aren’t still profoundly critical ill people with COVID of many different age ranges, that is true, but there are a lot of people that we now see will test positive, but aren’t really as profoundly ill as when it first landed here in the United States.”
That may be somewhat welcome as flu season arrives.
The physicians interviewed have no relevant disclosures.
The medical community is about to find out how prepared it is for the double whammy of influenza and COVID-19 that has been predicted for the fall of 2020. The complexities of diagnosis, management of vulnerable patients, and overflowing medical centers that have made the COVID-19 crisis so brutal may all be exacerbated by the arrival of seasonal influenza.
Lewis Jay Kaplan, MD, FCCP, a critical care surgeon at the University of Pennsylvania, Philadelphia, has seen his share of critically ill COVID-19 patients in the surgical ICU that he oversees. He’s approaching the upcoming flu season, poised to collide with the ongoing COVID-19 pandemic, ready to listen to each patient’s story to distinguish one from the other and determine treatment.
“The patients that have underlying comorbidities all have a story, and it’s up to you to figure out which chapter you’re in and how far along you happen to be,” he said. “It’s a very interesting approach to care, medical storytelling.”
With flu season closing in, pulmonologists are ruminating about how they’ll distinguish symptoms of COVID-19 and traditional influenza and how they’ll manage the most vulnerable patients, namely those with underlying respiratory disease and children. Influenza kills 12,000-61,000 people a year, according to the Centers for Disease Control, and results in 140,000-810,00 hospitalizations. Having a flu season in the midst of a pandemic of a disease with multiple overlapping symptoms threatens to overwhelm practitioners, hospitals, and the health system.
Dr. Kaplan said each patient’s story can point to the correct clinical approach. “Instead of just sharing data when you are on rounds, you’re really telling someone’s story.” It arises from a series of questions about how the disease has impacted them, specifics of their presentation, how their signs and symptoms differ from the usual, and how they responded to treatment. “It also helps you to then take what you’re doing, which can seem very, very complicated to individuals who are not medically sophisticated, and then help them to understand why you’re doing what you’re doing at this point.”
That can help get through to a patient with respiratory disease who insists he or she has or doesn’t have COVID-19 rather than the flu. “They form a different group that brings with them different fears and concerns, and you have to help them navigate that, too: all of this data and your decision-making around testing and admissions, and what you can omit doing and what you must do help them to navigate their own story,” Dr. Kaplan said.
Benjamin D. Singer, MD, a pulmonologist at Northwestern University, Chicago, authored an editorial in Science Advances that addressed four factors that will determine the scope of flu spread in the upcoming season: rate of transmission; vaccination rates; coinfection rates; and health disparities in minority populations, which are prone to higher rates of flu as well as COVID-19.
Flu vaccine ‘extra important’
The convergence of COVID-19 and influenza has the potential to overwhelm the health system, said Daniel A. Solomon, MD, of Brigham and Women’s in Boston. He coauthored a JAMA Insights clinical update on flu season during the COVID-19 pandemic that lists distinguishing and overlapping signs and symptoms of the two diseases.
The flu vaccine, he said, is “extra important this year,” especially in patients with existing respiratory disease, but COVID-19 has thrown up barriers to vaccination. Telemedicine has supplanted office visits. “People may miss that easy-touch opportunity to get the flu vaccine, so we have to be creative about making the flu vaccine highly accessible, maybe in nontraditional ways,” Dr. Solomon said. Some ideas he offered are pop-up vaccine fairs at schools and churches.
But just as COVID-19 may hinder flu vaccines, it may also be helping to mitigate flu transmission. “The interesting thing about transmission of the flu is that it’s transmitted the same way COVID is, so if we actually know how to decrease transmission of COVID, which we do – we’ve done it – we can actually decrease transmission of influenza as well,” Dr. Solomon said. Studies out of Hong Kong and Japan have reported a reduction in influenza cases during COVID-19 outbreaks in those places (Lancet Public Health. 2020;5:e279-88; JAMA. 2020;323:1969-71).
Risks of coinfection
About one in four COVID-19 patients have been diagnosed with an additional respiratory infection, including influenza (JAMA. 2020:323:2085-6). Pulmonologists must keep that in mind when managing COVID-19 suspects, said Dr. Singer.
“While it is true that most of the time COVID-19 travels alone, we have numerous examples in the literature and in our own experience that COVID-19 is accompanied by either another virus or another bacterial infection, including influenza,” Dr. Singer said. “The distinction is important. One is just for diagnostic reasons and public reporting reasons, but also because flu and COVID-19 have different requirements for how you care for patients in terms of the health system.”
Clinical suspicion for coinfection should remain high if the community spread of both COVID-19 and influenza is high, said Megan Conroy, MD, chief pulmonary and critical care fellow at Ohio State University, Columbus. “As the coronavirus first took hold in the United States in March 2020, we were at the tail end of influenza season, so it’s hard to predict what the upcoming influenza season will really look like with regards to coinfection.”
Distinguishing COVID-19 from flu
Multiple signs and symptoms between COVID-19 and the flu overlap. They include fever, chills, headache, myalgia, cough, and fatigue. Nasal congestion and sore throat are characteristic of the flu; shortness of breath and loss of the sense of smell have been widely reported in COVID-19. “While many upper respiratory infections can result in loss of smell, this may be more prevalent in COVID-19,” Dr. Conroy said. Other symptoms unique to COVID-19 are GI symptoms such as diarrhea and skin rashes such as acral ischemia.
Testing, however, is the cornerstone of the differential diagnosis. “You can’t confidently distinguish between them on symptoms alone,” Dr. Conroy added.
“I think the challenge we’ll face as clinicians, is caring for people with nonspecific symptoms of a respiratory viral illness, especially in the early phase of the illness,” said Dr. Solomon.
But even after that, symptoms can be difficult to distinguish.
“Later in the illness, COVID is more associated with a hypercoagulable state,” he said. “It is more associated with viral pneumonia on chest imaging, like the diffuse ground-glass infiltrates that we’ve all gotten used to seeing – but flu can do both of those things as well. So, without a test, it’s impossible to distinguish between the two infections in the clinic.”
But testing can have its shortcomings when flu season clashes with the COVID-19 pandemic. “Getting the test is not the same as getting the test results,” Dr. Solomon added. “Though a lot of people can get a test, if it takes 7 or 8 days to get the test result back, the result is useless.”
Widespread, rapid testing also depends on having adequate supplies of viral media transport and swabs. “I think that this is what we should be focusing on now: scaling up access to rapid turnaround testing,” he said. Distinguishing between the two is also important to preserve hospital resources. COVID-19 has more rigorous standards than flu for personal protective equipment and isolation of patients within the hospital.
Having chronic lung disease isn’t necessarily a risk factor for contracting COVID-19 or the flu, or both, Dr. Solomon said. “It’s a risk factor for having severe disease.” Again, he noted that flu vaccines are still necessary in these patients, as well as patients of advanced age and underlying medical conditions such as heart disease, diabetes, and obesity.
In managing children, it’s important to keep in mind that they communicate differently about their illnesses than adults, said Dr. Kaplan. “They may not have the words to tell you the same kind of thing that the adult tells you.” That’s where family members can help to flesh out the history. “They may present with an initially much milder form, if you will, where they’re not as critical up front, but then that small proportion of them comes back with the multi-inflammatory syndrome and then they are profoundly ill.”
Younger people make up a larger share of COVID-19 patients now, compared with the initial wave that hit the Northeast in the spring, Dr. Kaplan said. “We don’t know if that’s because the virus is a little different or the people that are getting sick are a little bit different.”
The COVID-19 strain now emerging may be less virulent than the strain that hit in early spring, he said. “That doesn’t mean that there aren’t still profoundly critical ill people with COVID of many different age ranges, that is true, but there are a lot of people that we now see will test positive, but aren’t really as profoundly ill as when it first landed here in the United States.”
That may be somewhat welcome as flu season arrives.
The physicians interviewed have no relevant disclosures.
The medical community is about to find out how prepared it is for the double whammy of influenza and COVID-19 that has been predicted for the fall of 2020. The complexities of diagnosis, management of vulnerable patients, and overflowing medical centers that have made the COVID-19 crisis so brutal may all be exacerbated by the arrival of seasonal influenza.
Lewis Jay Kaplan, MD, FCCP, a critical care surgeon at the University of Pennsylvania, Philadelphia, has seen his share of critically ill COVID-19 patients in the surgical ICU that he oversees. He’s approaching the upcoming flu season, poised to collide with the ongoing COVID-19 pandemic, ready to listen to each patient’s story to distinguish one from the other and determine treatment.
“The patients that have underlying comorbidities all have a story, and it’s up to you to figure out which chapter you’re in and how far along you happen to be,” he said. “It’s a very interesting approach to care, medical storytelling.”
With flu season closing in, pulmonologists are ruminating about how they’ll distinguish symptoms of COVID-19 and traditional influenza and how they’ll manage the most vulnerable patients, namely those with underlying respiratory disease and children. Influenza kills 12,000-61,000 people a year, according to the Centers for Disease Control, and results in 140,000-810,00 hospitalizations. Having a flu season in the midst of a pandemic of a disease with multiple overlapping symptoms threatens to overwhelm practitioners, hospitals, and the health system.
Dr. Kaplan said each patient’s story can point to the correct clinical approach. “Instead of just sharing data when you are on rounds, you’re really telling someone’s story.” It arises from a series of questions about how the disease has impacted them, specifics of their presentation, how their signs and symptoms differ from the usual, and how they responded to treatment. “It also helps you to then take what you’re doing, which can seem very, very complicated to individuals who are not medically sophisticated, and then help them to understand why you’re doing what you’re doing at this point.”
That can help get through to a patient with respiratory disease who insists he or she has or doesn’t have COVID-19 rather than the flu. “They form a different group that brings with them different fears and concerns, and you have to help them navigate that, too: all of this data and your decision-making around testing and admissions, and what you can omit doing and what you must do help them to navigate their own story,” Dr. Kaplan said.
Benjamin D. Singer, MD, a pulmonologist at Northwestern University, Chicago, authored an editorial in Science Advances that addressed four factors that will determine the scope of flu spread in the upcoming season: rate of transmission; vaccination rates; coinfection rates; and health disparities in minority populations, which are prone to higher rates of flu as well as COVID-19.
Flu vaccine ‘extra important’
The convergence of COVID-19 and influenza has the potential to overwhelm the health system, said Daniel A. Solomon, MD, of Brigham and Women’s in Boston. He coauthored a JAMA Insights clinical update on flu season during the COVID-19 pandemic that lists distinguishing and overlapping signs and symptoms of the two diseases.
The flu vaccine, he said, is “extra important this year,” especially in patients with existing respiratory disease, but COVID-19 has thrown up barriers to vaccination. Telemedicine has supplanted office visits. “People may miss that easy-touch opportunity to get the flu vaccine, so we have to be creative about making the flu vaccine highly accessible, maybe in nontraditional ways,” Dr. Solomon said. Some ideas he offered are pop-up vaccine fairs at schools and churches.
But just as COVID-19 may hinder flu vaccines, it may also be helping to mitigate flu transmission. “The interesting thing about transmission of the flu is that it’s transmitted the same way COVID is, so if we actually know how to decrease transmission of COVID, which we do – we’ve done it – we can actually decrease transmission of influenza as well,” Dr. Solomon said. Studies out of Hong Kong and Japan have reported a reduction in influenza cases during COVID-19 outbreaks in those places (Lancet Public Health. 2020;5:e279-88; JAMA. 2020;323:1969-71).
Risks of coinfection
About one in four COVID-19 patients have been diagnosed with an additional respiratory infection, including influenza (JAMA. 2020:323:2085-6). Pulmonologists must keep that in mind when managing COVID-19 suspects, said Dr. Singer.
“While it is true that most of the time COVID-19 travels alone, we have numerous examples in the literature and in our own experience that COVID-19 is accompanied by either another virus or another bacterial infection, including influenza,” Dr. Singer said. “The distinction is important. One is just for diagnostic reasons and public reporting reasons, but also because flu and COVID-19 have different requirements for how you care for patients in terms of the health system.”
Clinical suspicion for coinfection should remain high if the community spread of both COVID-19 and influenza is high, said Megan Conroy, MD, chief pulmonary and critical care fellow at Ohio State University, Columbus. “As the coronavirus first took hold in the United States in March 2020, we were at the tail end of influenza season, so it’s hard to predict what the upcoming influenza season will really look like with regards to coinfection.”
Distinguishing COVID-19 from flu
Multiple signs and symptoms between COVID-19 and the flu overlap. They include fever, chills, headache, myalgia, cough, and fatigue. Nasal congestion and sore throat are characteristic of the flu; shortness of breath and loss of the sense of smell have been widely reported in COVID-19. “While many upper respiratory infections can result in loss of smell, this may be more prevalent in COVID-19,” Dr. Conroy said. Other symptoms unique to COVID-19 are GI symptoms such as diarrhea and skin rashes such as acral ischemia.
Testing, however, is the cornerstone of the differential diagnosis. “You can’t confidently distinguish between them on symptoms alone,” Dr. Conroy added.
“I think the challenge we’ll face as clinicians, is caring for people with nonspecific symptoms of a respiratory viral illness, especially in the early phase of the illness,” said Dr. Solomon.
But even after that, symptoms can be difficult to distinguish.
“Later in the illness, COVID is more associated with a hypercoagulable state,” he said. “It is more associated with viral pneumonia on chest imaging, like the diffuse ground-glass infiltrates that we’ve all gotten used to seeing – but flu can do both of those things as well. So, without a test, it’s impossible to distinguish between the two infections in the clinic.”
But testing can have its shortcomings when flu season clashes with the COVID-19 pandemic. “Getting the test is not the same as getting the test results,” Dr. Solomon added. “Though a lot of people can get a test, if it takes 7 or 8 days to get the test result back, the result is useless.”
Widespread, rapid testing also depends on having adequate supplies of viral media transport and swabs. “I think that this is what we should be focusing on now: scaling up access to rapid turnaround testing,” he said. Distinguishing between the two is also important to preserve hospital resources. COVID-19 has more rigorous standards than flu for personal protective equipment and isolation of patients within the hospital.
Having chronic lung disease isn’t necessarily a risk factor for contracting COVID-19 or the flu, or both, Dr. Solomon said. “It’s a risk factor for having severe disease.” Again, he noted that flu vaccines are still necessary in these patients, as well as patients of advanced age and underlying medical conditions such as heart disease, diabetes, and obesity.
In managing children, it’s important to keep in mind that they communicate differently about their illnesses than adults, said Dr. Kaplan. “They may not have the words to tell you the same kind of thing that the adult tells you.” That’s where family members can help to flesh out the history. “They may present with an initially much milder form, if you will, where they’re not as critical up front, but then that small proportion of them comes back with the multi-inflammatory syndrome and then they are profoundly ill.”
Younger people make up a larger share of COVID-19 patients now, compared with the initial wave that hit the Northeast in the spring, Dr. Kaplan said. “We don’t know if that’s because the virus is a little different or the people that are getting sick are a little bit different.”
The COVID-19 strain now emerging may be less virulent than the strain that hit in early spring, he said. “That doesn’t mean that there aren’t still profoundly critical ill people with COVID of many different age ranges, that is true, but there are a lot of people that we now see will test positive, but aren’t really as profoundly ill as when it first landed here in the United States.”
That may be somewhat welcome as flu season arrives.
The physicians interviewed have no relevant disclosures.
As COVID-19 cases increase in children, deaths remain low
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.
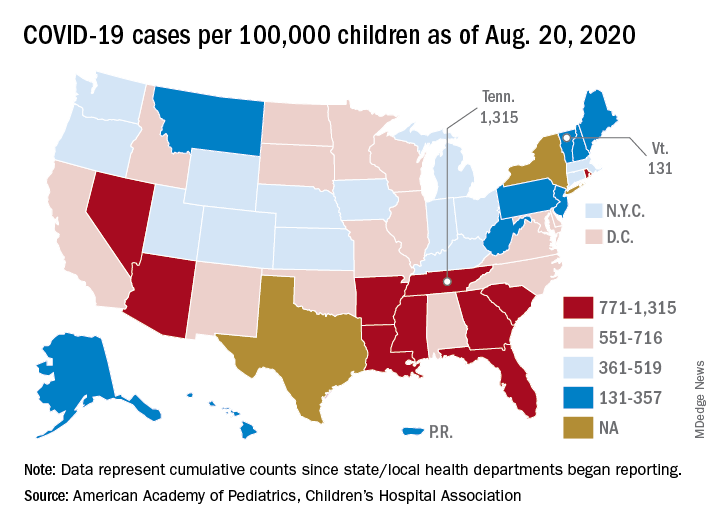
The cumulative number of pediatric cases reported up to that date was 442,785, or 9.3% of the total COVID-19 case load of more than 4.76 million among all ages. There have been only 92 pediatric deaths, however, which works out to just 0.06% of the 154,279 reported for all ages, the AAP and the CHA said Aug. 24 in their most recent update.
Child hospitalizations also were on the low side, representing 1.7% (4,062) of the cumulative total of 234,810 admissions among all ages as of Aug. 20, based on data from 21 states and New York City.
Nationally, the cumulative number of reported child cases is now up to 583 per 100,000 children, and that figure covers 49 states, Washington, D.C., Guam, New York City, and Puerto Rico.
There is some disagreement among the states, though, about the definition of “child.” Most states use an age range of 0-17, 0-18, or 0-19, but Florida and Utah go with a range of 0-14 years while South Carolina and Tennessee consider humans aged 0-20 years to be children. Other data limitations involve Texas, which has reported age distribution for only 8% of all cases, and New York, which is not reporting the age distribution of statewide cases, the AAP/CHA report noted.
The definition of child isn’t the only thing that varies between the states. The cumulative case rate for Tennessee, the highest in the country at 1,315 per 100,000 children, is 10 times that of Vermont, which is the lowest at 131 per 100,000, the AAP and CHA said. Vermont reports child COVID-19 cases using an age range of 0-19 years.
The other states with rates over 1,000 cases per 100,000 children are Arizona (1,300), which had the highest rate a week ago; South Carolina (1,214); Louisiana (1,127); Mississippi (1,120); and Nevada (1,068). Those with rates below 200 cases per 100,000 children are Maine (150), New Hampshire (175), and Hawaii (188), according to this week’s report.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The cumulative number of pediatric cases reported up to that date was 442,785, or 9.3% of the total COVID-19 case load of more than 4.76 million among all ages. There have been only 92 pediatric deaths, however, which works out to just 0.06% of the 154,279 reported for all ages, the AAP and the CHA said Aug. 24 in their most recent update.
Child hospitalizations also were on the low side, representing 1.7% (4,062) of the cumulative total of 234,810 admissions among all ages as of Aug. 20, based on data from 21 states and New York City.
Nationally, the cumulative number of reported child cases is now up to 583 per 100,000 children, and that figure covers 49 states, Washington, D.C., Guam, New York City, and Puerto Rico.
There is some disagreement among the states, though, about the definition of “child.” Most states use an age range of 0-17, 0-18, or 0-19, but Florida and Utah go with a range of 0-14 years while South Carolina and Tennessee consider humans aged 0-20 years to be children. Other data limitations involve Texas, which has reported age distribution for only 8% of all cases, and New York, which is not reporting the age distribution of statewide cases, the AAP/CHA report noted.
The definition of child isn’t the only thing that varies between the states. The cumulative case rate for Tennessee, the highest in the country at 1,315 per 100,000 children, is 10 times that of Vermont, which is the lowest at 131 per 100,000, the AAP and CHA said. Vermont reports child COVID-19 cases using an age range of 0-19 years.
The other states with rates over 1,000 cases per 100,000 children are Arizona (1,300), which had the highest rate a week ago; South Carolina (1,214); Louisiana (1,127); Mississippi (1,120); and Nevada (1,068). Those with rates below 200 cases per 100,000 children are Maine (150), New Hampshire (175), and Hawaii (188), according to this week’s report.
according to a report from the American Academy of Pediatrics and the Children’s Hospital Association.

The cumulative number of pediatric cases reported up to that date was 442,785, or 9.3% of the total COVID-19 case load of more than 4.76 million among all ages. There have been only 92 pediatric deaths, however, which works out to just 0.06% of the 154,279 reported for all ages, the AAP and the CHA said Aug. 24 in their most recent update.
Child hospitalizations also were on the low side, representing 1.7% (4,062) of the cumulative total of 234,810 admissions among all ages as of Aug. 20, based on data from 21 states and New York City.
Nationally, the cumulative number of reported child cases is now up to 583 per 100,000 children, and that figure covers 49 states, Washington, D.C., Guam, New York City, and Puerto Rico.
There is some disagreement among the states, though, about the definition of “child.” Most states use an age range of 0-17, 0-18, or 0-19, but Florida and Utah go with a range of 0-14 years while South Carolina and Tennessee consider humans aged 0-20 years to be children. Other data limitations involve Texas, which has reported age distribution for only 8% of all cases, and New York, which is not reporting the age distribution of statewide cases, the AAP/CHA report noted.
The definition of child isn’t the only thing that varies between the states. The cumulative case rate for Tennessee, the highest in the country at 1,315 per 100,000 children, is 10 times that of Vermont, which is the lowest at 131 per 100,000, the AAP and CHA said. Vermont reports child COVID-19 cases using an age range of 0-19 years.
The other states with rates over 1,000 cases per 100,000 children are Arizona (1,300), which had the highest rate a week ago; South Carolina (1,214); Louisiana (1,127); Mississippi (1,120); and Nevada (1,068). Those with rates below 200 cases per 100,000 children are Maine (150), New Hampshire (175), and Hawaii (188), according to this week’s report.
Famotidine associated with benefits in hospitalized COVID patients in another trial
It also demonstrated lower levels of serum markers for severe disease.
The findings come from an observational study of 83 hospitalized patients that was published in the American Journal of Gastroenterology.
“The mechanism of exactly how famotidine works has yet to be proven,” lead study author Jeffrey F. Mather, MS, said in an interview. “There’s thought that it works directly on the virus, and there is thought that it works through inactivating certain proteases that are required for the virus infection, but I think the most interesting [hypothesis] is by Malone et al. “They’re looking at the blocking of the histamine-2 receptor causing a decrease in the amount of histamine. It’s all speculative, but it will be interesting if that gets worked out.”
In a study that largely mimicked that of an earlier, larger published observational study on the topic (doi: 10.1053/j.gastro.2020.05.053), Mr. Mather and colleagues retrospectively evaluated 878 patients who tested positive for SARS-CoV-2 and who required admission to Hartford (Conn.) Hospital between Feb. 24, 2020, and May 14, 2020. Patients were classified as receiving famotidine if they were treated with either oral or intravenous drug within 1 week of COVID-19 screening and/or hospital admission. Primary outcomes of interest were in-hospital death as recorded in the discharge of the patients, requirement for mechanical ventilation, and the composite of death or requirement for ventilation. Secondary outcomes of interest were several serum markers of disease activity including white blood cell count, lymphocyte count, and eosinophil count.
Famotidine was administered orally in 83% of the patients and intravenously in the remaining 17%. Mr. Mather, director of data management in the division of research management at Hartford Hospital, and his colleagues reported that 83 of the 878 patients studied (9.5%) received famotidine. Compared with patients not treated with famotidine, those who received the drug were slightly younger (a mean of 64 vs. 68 years, respectively; P = .021); otherwise, there were no differences between the two groups in baseline demographics or in preexisting comorbidities.
The use of famotidine was associated with a decreased risk of in-hospital mortality (odds ratio, 0.37; P = .021) as well as combined death or intubation (OR, 0.47; P = .040). The outcomes were similar when the researchers performed propensity score matching to adjust for age differences between groups.
In addition, the use of famotidine was associated with lower levels of serum markers for severe disease including lower median peak C-reactive protein levels (9.4 vs. 12.7 mg/dL; P =. 002), lower median procalcitonin levels (0.16 vs. 0.30 ng/mL; P = .004), and a nonsignificant trend to lower median mean ferritin levels (797.5 vs. 964 ng/mL; P = .076).
Logistic regression analysis revealed that use of famotidine was an independent predictor of both lower mortality and combined death/intubation. In addition, predictors of both adverse outcomes included older age, a body mass index of greater than 30 kg/m2, chronic kidney disease, the national early warning score, and a higher neutrophil-lymphocyte ratio.
“This is an important stepping stone, but until we have a randomized, controlled trial, we really can’t speak about causation; we can only speak about association, and that’s okay,” Brennan Spiegel, MD, MSHS, director of health services research at Cedars-Sinai, Los Angeles, who was not affiliated with the study, said in an interview. “There’s nothing wrong with association because finding associations can raise important hypotheses that can then be tested in prospective randomized trials, for example.”
In July 2020, Dr. Spiegel and his colleagues published a separate paper looking at proton pump inhibitors and the risk of COVID-19. “In that study we did look at H2 blockers, and we did find that they were slightly associated with a reduction in COVID-19,” he said. “It was a small effect, but it was a benefit. When we see consistency among studies, it’s a signal in the noise we can try and follow and see if there is something more to it.”
Mr. Mather acknowledged certain limitations of the study, including the fact that patients who did and did not receive famotidine were propensity-matched for age. “The risk factors that others have shown for adverse events are equivalent in the groups, but anytime you do a retrospective study like this there is the potential for underlying factors that may play a role in the outcomes that you’re not considering,” Mr. Mather said. “That’s why the gold standard is the randomized trial, to wash those effects out. There’s only an association here, and it supports the need for a randomized trial.”
Famotidine is currently being tested in a double-blind randomized clinical trial in combination with either hydroxychloroquine or remdesivir (NCT 04370262).
“It’s fascinating because famotidine is a safe medicine,” added Dr. Spiegel, who is also co–editor in chief of the American Journal of Gastroenterology. “There are very few side effects; it’s something we’ve been using for decades.”
Mr. Mather and his colleagues reported having no financial disclosures. Dr. Spiegel disclosed that he has served on advisory boards for Allergan, Alnylam Pharmaceuticals, Arena Pharmaceuticals, Ironwood Pharmaceuticals, Salix Pharmaceuticals, Synergy Pharmaceuticals, and Takeda Pharmaceuticals.
SOURCE: Mather J et al. 2020 Aug 14. Am J Gastroenterol.
It also demonstrated lower levels of serum markers for severe disease.
The findings come from an observational study of 83 hospitalized patients that was published in the American Journal of Gastroenterology.
“The mechanism of exactly how famotidine works has yet to be proven,” lead study author Jeffrey F. Mather, MS, said in an interview. “There’s thought that it works directly on the virus, and there is thought that it works through inactivating certain proteases that are required for the virus infection, but I think the most interesting [hypothesis] is by Malone et al. “They’re looking at the blocking of the histamine-2 receptor causing a decrease in the amount of histamine. It’s all speculative, but it will be interesting if that gets worked out.”
In a study that largely mimicked that of an earlier, larger published observational study on the topic (doi: 10.1053/j.gastro.2020.05.053), Mr. Mather and colleagues retrospectively evaluated 878 patients who tested positive for SARS-CoV-2 and who required admission to Hartford (Conn.) Hospital between Feb. 24, 2020, and May 14, 2020. Patients were classified as receiving famotidine if they were treated with either oral or intravenous drug within 1 week of COVID-19 screening and/or hospital admission. Primary outcomes of interest were in-hospital death as recorded in the discharge of the patients, requirement for mechanical ventilation, and the composite of death or requirement for ventilation. Secondary outcomes of interest were several serum markers of disease activity including white blood cell count, lymphocyte count, and eosinophil count.
Famotidine was administered orally in 83% of the patients and intravenously in the remaining 17%. Mr. Mather, director of data management in the division of research management at Hartford Hospital, and his colleagues reported that 83 of the 878 patients studied (9.5%) received famotidine. Compared with patients not treated with famotidine, those who received the drug were slightly younger (a mean of 64 vs. 68 years, respectively; P = .021); otherwise, there were no differences between the two groups in baseline demographics or in preexisting comorbidities.
The use of famotidine was associated with a decreased risk of in-hospital mortality (odds ratio, 0.37; P = .021) as well as combined death or intubation (OR, 0.47; P = .040). The outcomes were similar when the researchers performed propensity score matching to adjust for age differences between groups.
In addition, the use of famotidine was associated with lower levels of serum markers for severe disease including lower median peak C-reactive protein levels (9.4 vs. 12.7 mg/dL; P =. 002), lower median procalcitonin levels (0.16 vs. 0.30 ng/mL; P = .004), and a nonsignificant trend to lower median mean ferritin levels (797.5 vs. 964 ng/mL; P = .076).
Logistic regression analysis revealed that use of famotidine was an independent predictor of both lower mortality and combined death/intubation. In addition, predictors of both adverse outcomes included older age, a body mass index of greater than 30 kg/m2, chronic kidney disease, the national early warning score, and a higher neutrophil-lymphocyte ratio.
“This is an important stepping stone, but until we have a randomized, controlled trial, we really can’t speak about causation; we can only speak about association, and that’s okay,” Brennan Spiegel, MD, MSHS, director of health services research at Cedars-Sinai, Los Angeles, who was not affiliated with the study, said in an interview. “There’s nothing wrong with association because finding associations can raise important hypotheses that can then be tested in prospective randomized trials, for example.”
In July 2020, Dr. Spiegel and his colleagues published a separate paper looking at proton pump inhibitors and the risk of COVID-19. “In that study we did look at H2 blockers, and we did find that they were slightly associated with a reduction in COVID-19,” he said. “It was a small effect, but it was a benefit. When we see consistency among studies, it’s a signal in the noise we can try and follow and see if there is something more to it.”
Mr. Mather acknowledged certain limitations of the study, including the fact that patients who did and did not receive famotidine were propensity-matched for age. “The risk factors that others have shown for adverse events are equivalent in the groups, but anytime you do a retrospective study like this there is the potential for underlying factors that may play a role in the outcomes that you’re not considering,” Mr. Mather said. “That’s why the gold standard is the randomized trial, to wash those effects out. There’s only an association here, and it supports the need for a randomized trial.”
Famotidine is currently being tested in a double-blind randomized clinical trial in combination with either hydroxychloroquine or remdesivir (NCT 04370262).
“It’s fascinating because famotidine is a safe medicine,” added Dr. Spiegel, who is also co–editor in chief of the American Journal of Gastroenterology. “There are very few side effects; it’s something we’ve been using for decades.”
Mr. Mather and his colleagues reported having no financial disclosures. Dr. Spiegel disclosed that he has served on advisory boards for Allergan, Alnylam Pharmaceuticals, Arena Pharmaceuticals, Ironwood Pharmaceuticals, Salix Pharmaceuticals, Synergy Pharmaceuticals, and Takeda Pharmaceuticals.
SOURCE: Mather J et al. 2020 Aug 14. Am J Gastroenterol.
It also demonstrated lower levels of serum markers for severe disease.
The findings come from an observational study of 83 hospitalized patients that was published in the American Journal of Gastroenterology.
“The mechanism of exactly how famotidine works has yet to be proven,” lead study author Jeffrey F. Mather, MS, said in an interview. “There’s thought that it works directly on the virus, and there is thought that it works through inactivating certain proteases that are required for the virus infection, but I think the most interesting [hypothesis] is by Malone et al. “They’re looking at the blocking of the histamine-2 receptor causing a decrease in the amount of histamine. It’s all speculative, but it will be interesting if that gets worked out.”
In a study that largely mimicked that of an earlier, larger published observational study on the topic (doi: 10.1053/j.gastro.2020.05.053), Mr. Mather and colleagues retrospectively evaluated 878 patients who tested positive for SARS-CoV-2 and who required admission to Hartford (Conn.) Hospital between Feb. 24, 2020, and May 14, 2020. Patients were classified as receiving famotidine if they were treated with either oral or intravenous drug within 1 week of COVID-19 screening and/or hospital admission. Primary outcomes of interest were in-hospital death as recorded in the discharge of the patients, requirement for mechanical ventilation, and the composite of death or requirement for ventilation. Secondary outcomes of interest were several serum markers of disease activity including white blood cell count, lymphocyte count, and eosinophil count.
Famotidine was administered orally in 83% of the patients and intravenously in the remaining 17%. Mr. Mather, director of data management in the division of research management at Hartford Hospital, and his colleagues reported that 83 of the 878 patients studied (9.5%) received famotidine. Compared with patients not treated with famotidine, those who received the drug were slightly younger (a mean of 64 vs. 68 years, respectively; P = .021); otherwise, there were no differences between the two groups in baseline demographics or in preexisting comorbidities.
The use of famotidine was associated with a decreased risk of in-hospital mortality (odds ratio, 0.37; P = .021) as well as combined death or intubation (OR, 0.47; P = .040). The outcomes were similar when the researchers performed propensity score matching to adjust for age differences between groups.
In addition, the use of famotidine was associated with lower levels of serum markers for severe disease including lower median peak C-reactive protein levels (9.4 vs. 12.7 mg/dL; P =. 002), lower median procalcitonin levels (0.16 vs. 0.30 ng/mL; P = .004), and a nonsignificant trend to lower median mean ferritin levels (797.5 vs. 964 ng/mL; P = .076).
Logistic regression analysis revealed that use of famotidine was an independent predictor of both lower mortality and combined death/intubation. In addition, predictors of both adverse outcomes included older age, a body mass index of greater than 30 kg/m2, chronic kidney disease, the national early warning score, and a higher neutrophil-lymphocyte ratio.
“This is an important stepping stone, but until we have a randomized, controlled trial, we really can’t speak about causation; we can only speak about association, and that’s okay,” Brennan Spiegel, MD, MSHS, director of health services research at Cedars-Sinai, Los Angeles, who was not affiliated with the study, said in an interview. “There’s nothing wrong with association because finding associations can raise important hypotheses that can then be tested in prospective randomized trials, for example.”
In July 2020, Dr. Spiegel and his colleagues published a separate paper looking at proton pump inhibitors and the risk of COVID-19. “In that study we did look at H2 blockers, and we did find that they were slightly associated with a reduction in COVID-19,” he said. “It was a small effect, but it was a benefit. When we see consistency among studies, it’s a signal in the noise we can try and follow and see if there is something more to it.”
Mr. Mather acknowledged certain limitations of the study, including the fact that patients who did and did not receive famotidine were propensity-matched for age. “The risk factors that others have shown for adverse events are equivalent in the groups, but anytime you do a retrospective study like this there is the potential for underlying factors that may play a role in the outcomes that you’re not considering,” Mr. Mather said. “That’s why the gold standard is the randomized trial, to wash those effects out. There’s only an association here, and it supports the need for a randomized trial.”
Famotidine is currently being tested in a double-blind randomized clinical trial in combination with either hydroxychloroquine or remdesivir (NCT 04370262).
“It’s fascinating because famotidine is a safe medicine,” added Dr. Spiegel, who is also co–editor in chief of the American Journal of Gastroenterology. “There are very few side effects; it’s something we’ve been using for decades.”
Mr. Mather and his colleagues reported having no financial disclosures. Dr. Spiegel disclosed that he has served on advisory boards for Allergan, Alnylam Pharmaceuticals, Arena Pharmaceuticals, Ironwood Pharmaceuticals, Salix Pharmaceuticals, Synergy Pharmaceuticals, and Takeda Pharmaceuticals.
SOURCE: Mather J et al. 2020 Aug 14. Am J Gastroenterol.
REPORTING FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Key clinical point: Among hospitalized COVID-19 patients, famotidine use was associated with a reduction in death and either death or intubation.
Major finding: The use of famotidine was associated with a decreased risk of in-hospital mortality (OR, 0.37; P = .021), as well as the combined endpoint of death or intubation (OR, 0.47; P = .040).
Study details: A single-center observational study of 83 patients hospitalized with COVID-19.
Disclosures: The researchers reported having no financial disclosures.
Source: Mather J et al. 2020 Aug 14. Am J Gastroenterol.
COVID-19, school reopenings, and safety: What should we tell parents?
Parents, teachers, children, and adolescents are facing stress and anxiety as K-12 school districts across the country debate whether to return to in-person instruction amid the COVID-19 pandemic. As we approach the opening of schools, the stress and anxiety seem to be heightening.
According to Education Week, which is tracking the reopening plans of public schools across the United States, 21 of the 25 largest school districts are opting to implement remote learning only as their model. I would like to see all of those districts adopt that model until we understand more about this illness, and can prevent and treat it.
Yes, it’s true – I am a psychiatrist – not an infectious disease specialist. And I realize that the American Academy of Pediatrics and the Centers for Disease Control and Prevention have taken nuanced positions on this issue. Their positions make it clear that it is within a child’s best interests – from an educational and social point of view – to attend school in person. Not only is the classroom experience important, but so is the socialization and the exercise. However, when I look at the science on children who have been exposed to the coronavirus, I worry.
For example, a study by Lael M. Yonker, MD, and associates on pediatric SARS-CoV-2 found that the children in days 0-2 of illness have far higher viral loads than adults who have been hospitalized for severe disease. “This study reveals that children may be a potential source of contagion in the SARS-CoV-2 pandemic in spite of milder disease or lack of symptoms, and immune dysregulation is implicated in severe post-infectious [multisystem inflammatory syndrome in children],” Dr. Yonker and associates wrote, referring to the illness associated with COVID-19 in children. Their study was published recently in the Journal of Pediatrics (2020 Aug 19. doi: 10.1016/j.jpeds.2020.08.037).
In my state, where positivity rates are fairly low, Gov. Andrew Cuomo admitted in an interview recently that sending children to school in New York City is a “tricky proposition.” At this point, New York City public schools are scheduled to open in mid-September using a hybrid mixture of in-person and remote learning.
And look at what happened several weeks ago in Israel, where schools reopened after the virus was beaten back. At one high school in Jerusalem, just days after the reopening, the virus spread so prolifically to students, teachers, and relatives that the schools had to be closed again. Other countries should not follow Israel’s example, Eli Weizmann, who chairs the team advising Israel’s National Security Council on the pandemic, reportedly told the New York Times. “It was a major failure.”
But I must be honest: I was worried about children returning to school before I heard about the study by Dr. Yonker and associates, Gov. Cuomo’s comments, and what happened in Israel. So far, here in the Northeast, particularly in New York, New Jersey, and Connecticut, we have managed to get COVID-19 under control. Perhaps, in this part of the country, opening classroom education might be feasible – with close monitoring and proper precautions.
But COVID-19 has taken the lives of hundreds of thousands of Americans – more than 176,000 as of this writing. A new model from the University of Washington’s Institute for Health Metrics and Evaluation projects that COVID-19 could lead to more than 300,000 U.S. deaths by Dec. 1. Thankfully, the number of COVID-19–positive children who have died has been low. But they could still pass on the virus to adults.
To get a better understanding of COVID-19, I spoke with Sheryl L. Wulkan, MD, an internist and expert in personal protective equipment (PPE) who has consulted for numerous health care agencies about these issues. Dr. Wulkan said that, in some areas with low infection rates, school openings might be appropriate. However, she said, without proper testing and contact tracing, we are at a loss of controlling the spread.
What we should tell patients, family, and friends
From a psychiatric point of view, how should we advise our patients, family, and friends about sending their children back to school? Is on-site learning better than remote learning? It is. Do our children need the socialization that a school brings? Yes, they do.
Socialization and relating to peers are, indeed, important, but today’s children socialize in many ways beyond attending school – and they have peer friendships and interactions with electronic devices at their disposal.
Can remote learning cause social isolation – an isolation so profound that school is necessary not only for learning but the psyche as well? A meta-analysis of 80 studies that looked at the impact of social isolation and loneliness on adolescents and children who were previously healthy found that the young people “are probably more likely to experience high rates of depression and probably anxiety during and after enforced isolation ends. This may increase as enforced isolation continues,” wrote Maria Elizabeth Loades, PhD, and associates (J Am Acad Child Adolesc Psychiatry. 2020 Jun 3. S0890-8567[20]30337-3).
I am concerned about young people who experience anxiety and depression, and agree with Dr. Loades that we mental health professionals need to be ready to intervene early and provide preventive support. To do this, we should encourage parents to keep us informed about how their children are doing.
So my advice is that, in the absence of a vaccine and an effective treatment like we have for influenza – such as Tamiflu – and effective testing, such the saliva-based test developed by Yale University researchers, if I had school-aged children, I would continue to keep them home from school. Ultimately, however, parents must look at the science and make their decisions based on that. My children are adults with their own children, and only they can make informed decisions about which options are best for their families.
Interestingly, Sanjay Gupta, MD, the neurosurgeon who works as chief medical correspondent of CNN, recently discussed the thought process he and his wife used to determine whether their daughters would return to the classroom. After weighing many factors, including the viral spread in Fulton County, Ga., where they live, the Guptas decided that, at this time, the risks of allowing the girls to return to the classroom outweigh the benefits. “This was not an easy decision, but one that we believe best respects the science, decreases the risk of further spread, and follows the task force criteria,” wrote Dr. Gupta, who is affiliated with Emory University in Atlanta. “After 2 weeks, we will reassess.”
I understand that parents worry about the social and psychological costs of remote learning. And I can only imagine the difficulty of those who must balance homeschooling with working. And frankly, remote learning is not an option for all students. For those less fortunate, substantial governmental aid is important to assist these people and to keep them safe and on their feet until this pandemic is done. Also, those who were under the care of a psychiatrist should continue to receive care during the pandemic. We must be prepared to step in with interventions that can address the suffering that is inevitable, such as the use of targeted cognitive-behavioral therapy.
Public TV as an educational tool
Families with Internet access and those without it could benefit from using public television as a tool.
I would advise educators and the entertainment industry to harness the wonder of TV to develop curricula that can be used to educate children. As we know, Sesame Street proved to be an effective early childhood intervention, particularly for boys (Am Econ J: Applied Economics. 2019;11[1]:318-50). I would like to see programming that goes beyond Sesame Street. Learning from watching this kind of programming would be no substitute for engaging with teachers in real, live classrooms, however.
Children and adolescents will be changed by learning remotely. They will miss their friends, teachers, and other staff members, but their lives will not be ruined. Mental health professionals should be prepared to intervene to address depression, anxiety, and other sequelae and problematic behaviors that could result from social isolation. Schools, businesses, and the economy will again flourish after we get the virus behind us but controlling and eliminating this pandemic need to come first. Let’s keep our children home – to the extent that we can – until we move beyond this pandemic.
Dr. London has been a practicing psychiatrist for 4 decades and a newspaper columnist for almost as long. He has a private practice in New York and is author of “Find Freedom Fast: Short-Term Therapy That Works” (New York: Kettlehole Publishing, 2019). Dr. London has no conflicts of interest.
Parents, teachers, children, and adolescents are facing stress and anxiety as K-12 school districts across the country debate whether to return to in-person instruction amid the COVID-19 pandemic. As we approach the opening of schools, the stress and anxiety seem to be heightening.
According to Education Week, which is tracking the reopening plans of public schools across the United States, 21 of the 25 largest school districts are opting to implement remote learning only as their model. I would like to see all of those districts adopt that model until we understand more about this illness, and can prevent and treat it.
Yes, it’s true – I am a psychiatrist – not an infectious disease specialist. And I realize that the American Academy of Pediatrics and the Centers for Disease Control and Prevention have taken nuanced positions on this issue. Their positions make it clear that it is within a child’s best interests – from an educational and social point of view – to attend school in person. Not only is the classroom experience important, but so is the socialization and the exercise. However, when I look at the science on children who have been exposed to the coronavirus, I worry.
For example, a study by Lael M. Yonker, MD, and associates on pediatric SARS-CoV-2 found that the children in days 0-2 of illness have far higher viral loads than adults who have been hospitalized for severe disease. “This study reveals that children may be a potential source of contagion in the SARS-CoV-2 pandemic in spite of milder disease or lack of symptoms, and immune dysregulation is implicated in severe post-infectious [multisystem inflammatory syndrome in children],” Dr. Yonker and associates wrote, referring to the illness associated with COVID-19 in children. Their study was published recently in the Journal of Pediatrics (2020 Aug 19. doi: 10.1016/j.jpeds.2020.08.037).
In my state, where positivity rates are fairly low, Gov. Andrew Cuomo admitted in an interview recently that sending children to school in New York City is a “tricky proposition.” At this point, New York City public schools are scheduled to open in mid-September using a hybrid mixture of in-person and remote learning.
And look at what happened several weeks ago in Israel, where schools reopened after the virus was beaten back. At one high school in Jerusalem, just days after the reopening, the virus spread so prolifically to students, teachers, and relatives that the schools had to be closed again. Other countries should not follow Israel’s example, Eli Weizmann, who chairs the team advising Israel’s National Security Council on the pandemic, reportedly told the New York Times. “It was a major failure.”
But I must be honest: I was worried about children returning to school before I heard about the study by Dr. Yonker and associates, Gov. Cuomo’s comments, and what happened in Israel. So far, here in the Northeast, particularly in New York, New Jersey, and Connecticut, we have managed to get COVID-19 under control. Perhaps, in this part of the country, opening classroom education might be feasible – with close monitoring and proper precautions.
But COVID-19 has taken the lives of hundreds of thousands of Americans – more than 176,000 as of this writing. A new model from the University of Washington’s Institute for Health Metrics and Evaluation projects that COVID-19 could lead to more than 300,000 U.S. deaths by Dec. 1. Thankfully, the number of COVID-19–positive children who have died has been low. But they could still pass on the virus to adults.
To get a better understanding of COVID-19, I spoke with Sheryl L. Wulkan, MD, an internist and expert in personal protective equipment (PPE) who has consulted for numerous health care agencies about these issues. Dr. Wulkan said that, in some areas with low infection rates, school openings might be appropriate. However, she said, without proper testing and contact tracing, we are at a loss of controlling the spread.
What we should tell patients, family, and friends
From a psychiatric point of view, how should we advise our patients, family, and friends about sending their children back to school? Is on-site learning better than remote learning? It is. Do our children need the socialization that a school brings? Yes, they do.
Socialization and relating to peers are, indeed, important, but today’s children socialize in many ways beyond attending school – and they have peer friendships and interactions with electronic devices at their disposal.
Can remote learning cause social isolation – an isolation so profound that school is necessary not only for learning but the psyche as well? A meta-analysis of 80 studies that looked at the impact of social isolation and loneliness on adolescents and children who were previously healthy found that the young people “are probably more likely to experience high rates of depression and probably anxiety during and after enforced isolation ends. This may increase as enforced isolation continues,” wrote Maria Elizabeth Loades, PhD, and associates (J Am Acad Child Adolesc Psychiatry. 2020 Jun 3. S0890-8567[20]30337-3).
I am concerned about young people who experience anxiety and depression, and agree with Dr. Loades that we mental health professionals need to be ready to intervene early and provide preventive support. To do this, we should encourage parents to keep us informed about how their children are doing.
So my advice is that, in the absence of a vaccine and an effective treatment like we have for influenza – such as Tamiflu – and effective testing, such the saliva-based test developed by Yale University researchers, if I had school-aged children, I would continue to keep them home from school. Ultimately, however, parents must look at the science and make their decisions based on that. My children are adults with their own children, and only they can make informed decisions about which options are best for their families.
Interestingly, Sanjay Gupta, MD, the neurosurgeon who works as chief medical correspondent of CNN, recently discussed the thought process he and his wife used to determine whether their daughters would return to the classroom. After weighing many factors, including the viral spread in Fulton County, Ga., where they live, the Guptas decided that, at this time, the risks of allowing the girls to return to the classroom outweigh the benefits. “This was not an easy decision, but one that we believe best respects the science, decreases the risk of further spread, and follows the task force criteria,” wrote Dr. Gupta, who is affiliated with Emory University in Atlanta. “After 2 weeks, we will reassess.”
I understand that parents worry about the social and psychological costs of remote learning. And I can only imagine the difficulty of those who must balance homeschooling with working. And frankly, remote learning is not an option for all students. For those less fortunate, substantial governmental aid is important to assist these people and to keep them safe and on their feet until this pandemic is done. Also, those who were under the care of a psychiatrist should continue to receive care during the pandemic. We must be prepared to step in with interventions that can address the suffering that is inevitable, such as the use of targeted cognitive-behavioral therapy.
Public TV as an educational tool
Families with Internet access and those without it could benefit from using public television as a tool.
I would advise educators and the entertainment industry to harness the wonder of TV to develop curricula that can be used to educate children. As we know, Sesame Street proved to be an effective early childhood intervention, particularly for boys (Am Econ J: Applied Economics. 2019;11[1]:318-50). I would like to see programming that goes beyond Sesame Street. Learning from watching this kind of programming would be no substitute for engaging with teachers in real, live classrooms, however.
Children and adolescents will be changed by learning remotely. They will miss their friends, teachers, and other staff members, but their lives will not be ruined. Mental health professionals should be prepared to intervene to address depression, anxiety, and other sequelae and problematic behaviors that could result from social isolation. Schools, businesses, and the economy will again flourish after we get the virus behind us but controlling and eliminating this pandemic need to come first. Let’s keep our children home – to the extent that we can – until we move beyond this pandemic.
Dr. London has been a practicing psychiatrist for 4 decades and a newspaper columnist for almost as long. He has a private practice in New York and is author of “Find Freedom Fast: Short-Term Therapy That Works” (New York: Kettlehole Publishing, 2019). Dr. London has no conflicts of interest.
Parents, teachers, children, and adolescents are facing stress and anxiety as K-12 school districts across the country debate whether to return to in-person instruction amid the COVID-19 pandemic. As we approach the opening of schools, the stress and anxiety seem to be heightening.
According to Education Week, which is tracking the reopening plans of public schools across the United States, 21 of the 25 largest school districts are opting to implement remote learning only as their model. I would like to see all of those districts adopt that model until we understand more about this illness, and can prevent and treat it.
Yes, it’s true – I am a psychiatrist – not an infectious disease specialist. And I realize that the American Academy of Pediatrics and the Centers for Disease Control and Prevention have taken nuanced positions on this issue. Their positions make it clear that it is within a child’s best interests – from an educational and social point of view – to attend school in person. Not only is the classroom experience important, but so is the socialization and the exercise. However, when I look at the science on children who have been exposed to the coronavirus, I worry.
For example, a study by Lael M. Yonker, MD, and associates on pediatric SARS-CoV-2 found that the children in days 0-2 of illness have far higher viral loads than adults who have been hospitalized for severe disease. “This study reveals that children may be a potential source of contagion in the SARS-CoV-2 pandemic in spite of milder disease or lack of symptoms, and immune dysregulation is implicated in severe post-infectious [multisystem inflammatory syndrome in children],” Dr. Yonker and associates wrote, referring to the illness associated with COVID-19 in children. Their study was published recently in the Journal of Pediatrics (2020 Aug 19. doi: 10.1016/j.jpeds.2020.08.037).
In my state, where positivity rates are fairly low, Gov. Andrew Cuomo admitted in an interview recently that sending children to school in New York City is a “tricky proposition.” At this point, New York City public schools are scheduled to open in mid-September using a hybrid mixture of in-person and remote learning.
And look at what happened several weeks ago in Israel, where schools reopened after the virus was beaten back. At one high school in Jerusalem, just days after the reopening, the virus spread so prolifically to students, teachers, and relatives that the schools had to be closed again. Other countries should not follow Israel’s example, Eli Weizmann, who chairs the team advising Israel’s National Security Council on the pandemic, reportedly told the New York Times. “It was a major failure.”
But I must be honest: I was worried about children returning to school before I heard about the study by Dr. Yonker and associates, Gov. Cuomo’s comments, and what happened in Israel. So far, here in the Northeast, particularly in New York, New Jersey, and Connecticut, we have managed to get COVID-19 under control. Perhaps, in this part of the country, opening classroom education might be feasible – with close monitoring and proper precautions.
But COVID-19 has taken the lives of hundreds of thousands of Americans – more than 176,000 as of this writing. A new model from the University of Washington’s Institute for Health Metrics and Evaluation projects that COVID-19 could lead to more than 300,000 U.S. deaths by Dec. 1. Thankfully, the number of COVID-19–positive children who have died has been low. But they could still pass on the virus to adults.
To get a better understanding of COVID-19, I spoke with Sheryl L. Wulkan, MD, an internist and expert in personal protective equipment (PPE) who has consulted for numerous health care agencies about these issues. Dr. Wulkan said that, in some areas with low infection rates, school openings might be appropriate. However, she said, without proper testing and contact tracing, we are at a loss of controlling the spread.
What we should tell patients, family, and friends
From a psychiatric point of view, how should we advise our patients, family, and friends about sending their children back to school? Is on-site learning better than remote learning? It is. Do our children need the socialization that a school brings? Yes, they do.
Socialization and relating to peers are, indeed, important, but today’s children socialize in many ways beyond attending school – and they have peer friendships and interactions with electronic devices at their disposal.
Can remote learning cause social isolation – an isolation so profound that school is necessary not only for learning but the psyche as well? A meta-analysis of 80 studies that looked at the impact of social isolation and loneliness on adolescents and children who were previously healthy found that the young people “are probably more likely to experience high rates of depression and probably anxiety during and after enforced isolation ends. This may increase as enforced isolation continues,” wrote Maria Elizabeth Loades, PhD, and associates (J Am Acad Child Adolesc Psychiatry. 2020 Jun 3. S0890-8567[20]30337-3).
I am concerned about young people who experience anxiety and depression, and agree with Dr. Loades that we mental health professionals need to be ready to intervene early and provide preventive support. To do this, we should encourage parents to keep us informed about how their children are doing.
So my advice is that, in the absence of a vaccine and an effective treatment like we have for influenza – such as Tamiflu – and effective testing, such the saliva-based test developed by Yale University researchers, if I had school-aged children, I would continue to keep them home from school. Ultimately, however, parents must look at the science and make their decisions based on that. My children are adults with their own children, and only they can make informed decisions about which options are best for their families.
Interestingly, Sanjay Gupta, MD, the neurosurgeon who works as chief medical correspondent of CNN, recently discussed the thought process he and his wife used to determine whether their daughters would return to the classroom. After weighing many factors, including the viral spread in Fulton County, Ga., where they live, the Guptas decided that, at this time, the risks of allowing the girls to return to the classroom outweigh the benefits. “This was not an easy decision, but one that we believe best respects the science, decreases the risk of further spread, and follows the task force criteria,” wrote Dr. Gupta, who is affiliated with Emory University in Atlanta. “After 2 weeks, we will reassess.”
I understand that parents worry about the social and psychological costs of remote learning. And I can only imagine the difficulty of those who must balance homeschooling with working. And frankly, remote learning is not an option for all students. For those less fortunate, substantial governmental aid is important to assist these people and to keep them safe and on their feet until this pandemic is done. Also, those who were under the care of a psychiatrist should continue to receive care during the pandemic. We must be prepared to step in with interventions that can address the suffering that is inevitable, such as the use of targeted cognitive-behavioral therapy.
Public TV as an educational tool
Families with Internet access and those without it could benefit from using public television as a tool.
I would advise educators and the entertainment industry to harness the wonder of TV to develop curricula that can be used to educate children. As we know, Sesame Street proved to be an effective early childhood intervention, particularly for boys (Am Econ J: Applied Economics. 2019;11[1]:318-50). I would like to see programming that goes beyond Sesame Street. Learning from watching this kind of programming would be no substitute for engaging with teachers in real, live classrooms, however.
Children and adolescents will be changed by learning remotely. They will miss their friends, teachers, and other staff members, but their lives will not be ruined. Mental health professionals should be prepared to intervene to address depression, anxiety, and other sequelae and problematic behaviors that could result from social isolation. Schools, businesses, and the economy will again flourish after we get the virus behind us but controlling and eliminating this pandemic need to come first. Let’s keep our children home – to the extent that we can – until we move beyond this pandemic.
Dr. London has been a practicing psychiatrist for 4 decades and a newspaper columnist for almost as long. He has a private practice in New York and is author of “Find Freedom Fast: Short-Term Therapy That Works” (New York: Kettlehole Publishing, 2019). Dr. London has no conflicts of interest.
Weighing children in school: No good can come of it
The United Kingdom’s National Obesity Forum has apparently decided that returning to school this fall in the middle of a pandemic isn’t stressful enough for kids, and is recommending that its National Child Measurement Programme be expanded to have 4- to 5-year-old and 10- to 11-year-old children weighed when they return to the classroom – and then weighed again in the spring – in a bid to tackle COVID-19–related gains.
It’s difficult to conceive a single plausible mechanism by which this recommendation could be helpful. Given that weight is, by a substantial margin, the No. 1 reported cause of schoolyard bullying, it’s certainly unlikely that children with obesity don’t already know that they have it. It’s also unlikely that they don’t know that obesity confers risks to health, given the near constant drumbeats of concern percussed by the media and public health authorities, and the fact that watching people with obesity be blamed, shamed, and berated for their condition has in the past 2 decades become a regularly repeated prime-time reality show spectacle.
It’s also unlikely, especially in younger grades, to be something within a child’s direct control.
What about the parents? Well, given that they dress their children and that changes in weight affect clothing sizes and fit, they’re already aware if their kids are gaining weight. And like their children, they have been exposed to constant public health alarms around obesity.
Many parents will have seen their time and resources, both real and mental, become significantly impaired during the time of COVID-19, which in turn understandably challenges change. Simply put, permanent intentional behavior change in the name of health requires tremendous privilege and is elusive for many people even during easier times. For non–evidence-based proof of this assertion, simply reflect on all of your own best-laid intentions and plans that might have been good for your health (fitness, relationships, CME, etc.) that you let slide despite probably having far more privilege than the average person.
Then, of course, there is the hugely inconvenient truth that we have yet to see the development of a parent- or child-based educational intervention or directive for weight gain that has shown itself to be beneficial on a population level.
Can something else be done instead?
At this point, we can only speculate about the potential risks associated with school room weigh-ins because randomized controlled trials, thankfully, have not been conducted to explore this area. But I can certainly tell you that I have met many adult patients in my office who traced their lifetime of yo-yo dieting – along with a history of teenage eating disorders, at times – to their well-intentioned physician, school nurse, gym teacher, or parent using a scale to measure their weights. And in doing so, they were teaching that scales measure health, happiness, success, self-worth, and effort.
If governments are concerned about weight gain in children, they need to look to initiatives that will help all children and parents. Weighing them will not somehow inspire parents or kids to discover an as-yet unknown effective childhood obesity treatment. Changes that would be helpful may include:
- Banning food advertisements to children.
- Reforming school cafeteria meals and then ensuring that school meals are made available to children during COVID-19–related school shutdowns.
- Bringing back home economics classes to teach children how to cook (and perhaps doing the same for parents during school off-hours or in community centers).
- Enacting sugar-sweetened beverage taxes and using revenues to fund aforementioned reforms and programs, along with others, which might include the subsidization of fresh produce.
- Reforming front-of-package health claims for foods with questionable nutritional quality.
Given that there is literally no age category in any country on the planet that hasn’t seen rising weights, this is clearly not a disease reflecting a pandemic loss of willpower. Rather, this is a disease of the world’s changing food environments and culture, and until we address both through systemic changes, schemes such as the one being proposed by the UK National Obesity Forum are far more likely to do harm than good.
Yoni Freedhoff is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of “The Diet Fix: Why Diets Fail and How to Make Yours Work.” A version of this article originally appeared on Medscape.com.
The United Kingdom’s National Obesity Forum has apparently decided that returning to school this fall in the middle of a pandemic isn’t stressful enough for kids, and is recommending that its National Child Measurement Programme be expanded to have 4- to 5-year-old and 10- to 11-year-old children weighed when they return to the classroom – and then weighed again in the spring – in a bid to tackle COVID-19–related gains.
It’s difficult to conceive a single plausible mechanism by which this recommendation could be helpful. Given that weight is, by a substantial margin, the No. 1 reported cause of schoolyard bullying, it’s certainly unlikely that children with obesity don’t already know that they have it. It’s also unlikely that they don’t know that obesity confers risks to health, given the near constant drumbeats of concern percussed by the media and public health authorities, and the fact that watching people with obesity be blamed, shamed, and berated for their condition has in the past 2 decades become a regularly repeated prime-time reality show spectacle.
It’s also unlikely, especially in younger grades, to be something within a child’s direct control.
What about the parents? Well, given that they dress their children and that changes in weight affect clothing sizes and fit, they’re already aware if their kids are gaining weight. And like their children, they have been exposed to constant public health alarms around obesity.
Many parents will have seen their time and resources, both real and mental, become significantly impaired during the time of COVID-19, which in turn understandably challenges change. Simply put, permanent intentional behavior change in the name of health requires tremendous privilege and is elusive for many people even during easier times. For non–evidence-based proof of this assertion, simply reflect on all of your own best-laid intentions and plans that might have been good for your health (fitness, relationships, CME, etc.) that you let slide despite probably having far more privilege than the average person.
Then, of course, there is the hugely inconvenient truth that we have yet to see the development of a parent- or child-based educational intervention or directive for weight gain that has shown itself to be beneficial on a population level.
Can something else be done instead?
At this point, we can only speculate about the potential risks associated with school room weigh-ins because randomized controlled trials, thankfully, have not been conducted to explore this area. But I can certainly tell you that I have met many adult patients in my office who traced their lifetime of yo-yo dieting – along with a history of teenage eating disorders, at times – to their well-intentioned physician, school nurse, gym teacher, or parent using a scale to measure their weights. And in doing so, they were teaching that scales measure health, happiness, success, self-worth, and effort.
If governments are concerned about weight gain in children, they need to look to initiatives that will help all children and parents. Weighing them will not somehow inspire parents or kids to discover an as-yet unknown effective childhood obesity treatment. Changes that would be helpful may include:
- Banning food advertisements to children.
- Reforming school cafeteria meals and then ensuring that school meals are made available to children during COVID-19–related school shutdowns.
- Bringing back home economics classes to teach children how to cook (and perhaps doing the same for parents during school off-hours or in community centers).
- Enacting sugar-sweetened beverage taxes and using revenues to fund aforementioned reforms and programs, along with others, which might include the subsidization of fresh produce.
- Reforming front-of-package health claims for foods with questionable nutritional quality.
Given that there is literally no age category in any country on the planet that hasn’t seen rising weights, this is clearly not a disease reflecting a pandemic loss of willpower. Rather, this is a disease of the world’s changing food environments and culture, and until we address both through systemic changes, schemes such as the one being proposed by the UK National Obesity Forum are far more likely to do harm than good.
Yoni Freedhoff is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of “The Diet Fix: Why Diets Fail and How to Make Yours Work.” A version of this article originally appeared on Medscape.com.
The United Kingdom’s National Obesity Forum has apparently decided that returning to school this fall in the middle of a pandemic isn’t stressful enough for kids, and is recommending that its National Child Measurement Programme be expanded to have 4- to 5-year-old and 10- to 11-year-old children weighed when they return to the classroom – and then weighed again in the spring – in a bid to tackle COVID-19–related gains.
It’s difficult to conceive a single plausible mechanism by which this recommendation could be helpful. Given that weight is, by a substantial margin, the No. 1 reported cause of schoolyard bullying, it’s certainly unlikely that children with obesity don’t already know that they have it. It’s also unlikely that they don’t know that obesity confers risks to health, given the near constant drumbeats of concern percussed by the media and public health authorities, and the fact that watching people with obesity be blamed, shamed, and berated for their condition has in the past 2 decades become a regularly repeated prime-time reality show spectacle.
It’s also unlikely, especially in younger grades, to be something within a child’s direct control.
What about the parents? Well, given that they dress their children and that changes in weight affect clothing sizes and fit, they’re already aware if their kids are gaining weight. And like their children, they have been exposed to constant public health alarms around obesity.
Many parents will have seen their time and resources, both real and mental, become significantly impaired during the time of COVID-19, which in turn understandably challenges change. Simply put, permanent intentional behavior change in the name of health requires tremendous privilege and is elusive for many people even during easier times. For non–evidence-based proof of this assertion, simply reflect on all of your own best-laid intentions and plans that might have been good for your health (fitness, relationships, CME, etc.) that you let slide despite probably having far more privilege than the average person.
Then, of course, there is the hugely inconvenient truth that we have yet to see the development of a parent- or child-based educational intervention or directive for weight gain that has shown itself to be beneficial on a population level.
Can something else be done instead?
At this point, we can only speculate about the potential risks associated with school room weigh-ins because randomized controlled trials, thankfully, have not been conducted to explore this area. But I can certainly tell you that I have met many adult patients in my office who traced their lifetime of yo-yo dieting – along with a history of teenage eating disorders, at times – to their well-intentioned physician, school nurse, gym teacher, or parent using a scale to measure their weights. And in doing so, they were teaching that scales measure health, happiness, success, self-worth, and effort.
If governments are concerned about weight gain in children, they need to look to initiatives that will help all children and parents. Weighing them will not somehow inspire parents or kids to discover an as-yet unknown effective childhood obesity treatment. Changes that would be helpful may include:
- Banning food advertisements to children.
- Reforming school cafeteria meals and then ensuring that school meals are made available to children during COVID-19–related school shutdowns.
- Bringing back home economics classes to teach children how to cook (and perhaps doing the same for parents during school off-hours or in community centers).
- Enacting sugar-sweetened beverage taxes and using revenues to fund aforementioned reforms and programs, along with others, which might include the subsidization of fresh produce.
- Reforming front-of-package health claims for foods with questionable nutritional quality.
Given that there is literally no age category in any country on the planet that hasn’t seen rising weights, this is clearly not a disease reflecting a pandemic loss of willpower. Rather, this is a disease of the world’s changing food environments and culture, and until we address both through systemic changes, schemes such as the one being proposed by the UK National Obesity Forum are far more likely to do harm than good.
Yoni Freedhoff is associate professor of family medicine at the University of Ottawa and medical director of the Bariatric Medical Institute, a nonsurgical weight management center. He is one of Canada’s most outspoken obesity experts and the author of “The Diet Fix: Why Diets Fail and How to Make Yours Work.” A version of this article originally appeared on Medscape.com.















