User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Why exercise doesn’t help people with long COVID
When Joel Fram woke up on the morning of March 12, 2020, he had a pretty good idea why he felt so lousy.
He lives in New York, where the first wave of the coronavirus was tearing through the city. “I instantly knew,” said the 55-year-old Broadway music director. It was COVID-19.
What started with a general sense of having been hit by a truck soon included a sore throat and such severe fatigue that he once fell asleep in the middle of sending a text to his sister. The final symptoms were chest tightness and trouble breathing.
And then he started to feel better. “By mid-April, my body was feeling essentially back to normal,” he said.
So he did what would have been smart after almost any other illness: He began working out. That didn’t last long. “It felt like someone pulled the carpet out from under me,” he remembered. “I couldn’t walk three blocks without getting breathless and fatigued.”
That was the first indication Mr. Fram had long COVID.
According to the National Center for Health Statistics, at least 7.5% of American adults – close to 20 million people – have symptoms of long COVID.
COVID-19 patients who had the most severe illness will struggle the most with exercise later, according to a review published in June from researchers at the University of California, San Francisco. But even people with mild symptoms can struggle to regain their previous levels of fitness.
“We have participants in our study who had relatively mild acute symptoms and went on to have really profound decreases in their ability to exercise,” said Matthew S. Durstenfeld, MD, a cardiologist at UCSF and principal author of the review.
Most people with long COVID will have lower-than-expected scores on tests of aerobic fitness, as shown by Yale researchers in a study published in August 2021.
“Some amount of that is due to deconditioning,” Dr. Durstenfeld said. “You’re not feeling well, so you’re not exercising to the same degree you might have been before you got infected.”
In a study published in April, people with long COVID told researchers at Britain’s University of Leeds they spent 93% less time in physical activity than they did before their infection.
But multiple studies have found deconditioning is not entirely – or even mostly – to blame.
A 2021 study found that 89% of participants with long COVID had postexertional malaise (PEM), which happens when a patient’s symptoms get worse after they do even minor physical or mental activities. According to the CDC, postexertional malaise can hit as long as 12-48 hours after the activity, and it can take people up to 2 weeks to fully recover.
Unfortunately, the advice patients get from their doctors sometimes makes the problem worse.
How long COVID defies simple solutions
Long COVID is a “dynamic disability” that requires health professionals to go off script when a patient’s symptoms don’t respond in a predictable way to treatment, said David Putrino, PhD, a neuroscientist, physical therapist, and director of rehabilitation innovation for the Mount Sinai Health System in New York.
“We’re not so good at dealing with somebody who, for all intents and purposes, can appear healthy and nondisabled on one day and be completely debilitated the next day,” he said.
Dr. Putrino said more than half of his clinic’s long-COVID patients told his team they had at least one of these persistent problems:
- Fatigue (82%).
- Brain fog (67%).
- Headache (60%).
- Sleep problems (59%).
- Dizziness (54%).
And 86% said exercise worsened their symptoms.
The symptoms are similar to what doctors see with illnesses such as lupus, Lyme disease, and chronic fatigue syndrome – something many experts compare long COVID to. Researchers and medical professionals still don’t know exactly how COVID-19 causes those symptoms. But there are some theories.
Potential causes of long-COVID symptoms
Dr. Putrino said it is possible the virus enters a patient’s cells and hijacks the mitochondria – a part of the cell that provides energy. It can linger there for weeks or months – something known as viral persistence.
“All of a sudden, the body’s getting less energy for itself, even though it’s producing the same amount, or even a little more,” he said. And there is a consequence to this extra stress on the cells. “Creating energy isn’t free. You’re producing more waste products, which puts your body in a state of oxidative stress,” Dr. Putrino said. Oxidative stress damages cells as molecules interact with oxygen in harmful ways.
“The other big mechanism is autonomic dysfunction,” Dr. Putrino said. It’s marked by breathing problems, heart palpitations, and other glitches in areas most healthy people never have to think about. About 70% of long-COVID patients at Mount Sinai’s clinic have some degree of autonomic dysfunction, he said.
For a person with autonomic dysfunction, something as basic as changing posture can trigger a storm of cytokines, a chemical messenger that tells the immune system where and how to respond to challenges like an injury or infection.
“Suddenly, you have this on-off switch,” Dr. Putrino said. “You go straight to ‘fight or flight,’ ” with a surge of adrenaline and a spiking heart rate, “then plunge back to ‘rest or digest.’ You go from fired up to so sleepy, you can’t keep your eyes open.”
A patient with viral persistence and one with autonomic dysfunction may have the same negative reaction to exercise, even though the triggers are completely different.
So how can doctors help long-COVID patients?
The first step, Dr. Putrino said, is to understand the difference between long COVID and a long recovery from COVID-19 infection.
Many of the patients in the latter group still have symptoms 4 weeks after their first infection. “At 4 weeks, yeah, they’re still feeling symptoms, but that’s not long COVID,” he said. “That’s just taking a while to get over a viral infection.”
Fitness advice is simple for those people: Take it easy at first, and gradually increase the amount and intensity of aerobic exercise and strength training.
But that advice would be disastrous for someone who meets Dr. Putrino’s stricter definition of long COVID: “Three to 4 months out from initial infection, they’re experiencing severe fatigue, exertional symptoms, cognitive symptoms, heart palpitations, shortness of breath,” he said.
“Our clinic is extraordinarily cautious with exercise” for those patients, he said.
In Dr. Putrino’s experience, about 20%-30% of patients will make significant progress after 12 weeks. “They’re feeling more or less like they felt pre-COVID,” he said.
The unluckiest 10%-20% won’t make any progress at all. Any type of therapy, even if it’s as simple as moving their legs from a flat position, worsens their symptoms.
The majority – 50%-60% – will have some improvement in their symptoms. But then progress will stop, for reasons researchers are still trying to figure out.
“My sense is that gradually increasing your exercise is still good advice for the vast majority of people,” UCSF’s Dr. Durstenfeld said.
Ideally, that exercise will be supervised by someone trained in cardiac, pulmonary, and/or autonomic rehabilitation – a specialized type of therapy aimed at resyncing the autonomic nervous system that governs breathing and other unconscious functions, he said. But those therapies are rarely covered by insurance, which means most long-COVID patients are on their own.
Dr. Durstenfeld said it’s important that patients keep trying and not give up. “With slow and steady progress, a lot of people can get profoundly better,” he said.
Mr. Fram, who’s worked with careful supervision, says he’s getting closer to something like his pre-COVID-19 life.
But he’s not there yet. Long COVID, he said, “affects my life every single day.”
A version of this article first appeared on WebMD.com.
When Joel Fram woke up on the morning of March 12, 2020, he had a pretty good idea why he felt so lousy.
He lives in New York, where the first wave of the coronavirus was tearing through the city. “I instantly knew,” said the 55-year-old Broadway music director. It was COVID-19.
What started with a general sense of having been hit by a truck soon included a sore throat and such severe fatigue that he once fell asleep in the middle of sending a text to his sister. The final symptoms were chest tightness and trouble breathing.
And then he started to feel better. “By mid-April, my body was feeling essentially back to normal,” he said.
So he did what would have been smart after almost any other illness: He began working out. That didn’t last long. “It felt like someone pulled the carpet out from under me,” he remembered. “I couldn’t walk three blocks without getting breathless and fatigued.”
That was the first indication Mr. Fram had long COVID.
According to the National Center for Health Statistics, at least 7.5% of American adults – close to 20 million people – have symptoms of long COVID.
COVID-19 patients who had the most severe illness will struggle the most with exercise later, according to a review published in June from researchers at the University of California, San Francisco. But even people with mild symptoms can struggle to regain their previous levels of fitness.
“We have participants in our study who had relatively mild acute symptoms and went on to have really profound decreases in their ability to exercise,” said Matthew S. Durstenfeld, MD, a cardiologist at UCSF and principal author of the review.
Most people with long COVID will have lower-than-expected scores on tests of aerobic fitness, as shown by Yale researchers in a study published in August 2021.
“Some amount of that is due to deconditioning,” Dr. Durstenfeld said. “You’re not feeling well, so you’re not exercising to the same degree you might have been before you got infected.”
In a study published in April, people with long COVID told researchers at Britain’s University of Leeds they spent 93% less time in physical activity than they did before their infection.
But multiple studies have found deconditioning is not entirely – or even mostly – to blame.
A 2021 study found that 89% of participants with long COVID had postexertional malaise (PEM), which happens when a patient’s symptoms get worse after they do even minor physical or mental activities. According to the CDC, postexertional malaise can hit as long as 12-48 hours after the activity, and it can take people up to 2 weeks to fully recover.
Unfortunately, the advice patients get from their doctors sometimes makes the problem worse.
How long COVID defies simple solutions
Long COVID is a “dynamic disability” that requires health professionals to go off script when a patient’s symptoms don’t respond in a predictable way to treatment, said David Putrino, PhD, a neuroscientist, physical therapist, and director of rehabilitation innovation for the Mount Sinai Health System in New York.
“We’re not so good at dealing with somebody who, for all intents and purposes, can appear healthy and nondisabled on one day and be completely debilitated the next day,” he said.
Dr. Putrino said more than half of his clinic’s long-COVID patients told his team they had at least one of these persistent problems:
- Fatigue (82%).
- Brain fog (67%).
- Headache (60%).
- Sleep problems (59%).
- Dizziness (54%).
And 86% said exercise worsened their symptoms.
The symptoms are similar to what doctors see with illnesses such as lupus, Lyme disease, and chronic fatigue syndrome – something many experts compare long COVID to. Researchers and medical professionals still don’t know exactly how COVID-19 causes those symptoms. But there are some theories.
Potential causes of long-COVID symptoms
Dr. Putrino said it is possible the virus enters a patient’s cells and hijacks the mitochondria – a part of the cell that provides energy. It can linger there for weeks or months – something known as viral persistence.
“All of a sudden, the body’s getting less energy for itself, even though it’s producing the same amount, or even a little more,” he said. And there is a consequence to this extra stress on the cells. “Creating energy isn’t free. You’re producing more waste products, which puts your body in a state of oxidative stress,” Dr. Putrino said. Oxidative stress damages cells as molecules interact with oxygen in harmful ways.
“The other big mechanism is autonomic dysfunction,” Dr. Putrino said. It’s marked by breathing problems, heart palpitations, and other glitches in areas most healthy people never have to think about. About 70% of long-COVID patients at Mount Sinai’s clinic have some degree of autonomic dysfunction, he said.
For a person with autonomic dysfunction, something as basic as changing posture can trigger a storm of cytokines, a chemical messenger that tells the immune system where and how to respond to challenges like an injury or infection.
“Suddenly, you have this on-off switch,” Dr. Putrino said. “You go straight to ‘fight or flight,’ ” with a surge of adrenaline and a spiking heart rate, “then plunge back to ‘rest or digest.’ You go from fired up to so sleepy, you can’t keep your eyes open.”
A patient with viral persistence and one with autonomic dysfunction may have the same negative reaction to exercise, even though the triggers are completely different.
So how can doctors help long-COVID patients?
The first step, Dr. Putrino said, is to understand the difference between long COVID and a long recovery from COVID-19 infection.
Many of the patients in the latter group still have symptoms 4 weeks after their first infection. “At 4 weeks, yeah, they’re still feeling symptoms, but that’s not long COVID,” he said. “That’s just taking a while to get over a viral infection.”
Fitness advice is simple for those people: Take it easy at first, and gradually increase the amount and intensity of aerobic exercise and strength training.
But that advice would be disastrous for someone who meets Dr. Putrino’s stricter definition of long COVID: “Three to 4 months out from initial infection, they’re experiencing severe fatigue, exertional symptoms, cognitive symptoms, heart palpitations, shortness of breath,” he said.
“Our clinic is extraordinarily cautious with exercise” for those patients, he said.
In Dr. Putrino’s experience, about 20%-30% of patients will make significant progress after 12 weeks. “They’re feeling more or less like they felt pre-COVID,” he said.
The unluckiest 10%-20% won’t make any progress at all. Any type of therapy, even if it’s as simple as moving their legs from a flat position, worsens their symptoms.
The majority – 50%-60% – will have some improvement in their symptoms. But then progress will stop, for reasons researchers are still trying to figure out.
“My sense is that gradually increasing your exercise is still good advice for the vast majority of people,” UCSF’s Dr. Durstenfeld said.
Ideally, that exercise will be supervised by someone trained in cardiac, pulmonary, and/or autonomic rehabilitation – a specialized type of therapy aimed at resyncing the autonomic nervous system that governs breathing and other unconscious functions, he said. But those therapies are rarely covered by insurance, which means most long-COVID patients are on their own.
Dr. Durstenfeld said it’s important that patients keep trying and not give up. “With slow and steady progress, a lot of people can get profoundly better,” he said.
Mr. Fram, who’s worked with careful supervision, says he’s getting closer to something like his pre-COVID-19 life.
But he’s not there yet. Long COVID, he said, “affects my life every single day.”
A version of this article first appeared on WebMD.com.
When Joel Fram woke up on the morning of March 12, 2020, he had a pretty good idea why he felt so lousy.
He lives in New York, where the first wave of the coronavirus was tearing through the city. “I instantly knew,” said the 55-year-old Broadway music director. It was COVID-19.
What started with a general sense of having been hit by a truck soon included a sore throat and such severe fatigue that he once fell asleep in the middle of sending a text to his sister. The final symptoms were chest tightness and trouble breathing.
And then he started to feel better. “By mid-April, my body was feeling essentially back to normal,” he said.
So he did what would have been smart after almost any other illness: He began working out. That didn’t last long. “It felt like someone pulled the carpet out from under me,” he remembered. “I couldn’t walk three blocks without getting breathless and fatigued.”
That was the first indication Mr. Fram had long COVID.
According to the National Center for Health Statistics, at least 7.5% of American adults – close to 20 million people – have symptoms of long COVID.
COVID-19 patients who had the most severe illness will struggle the most with exercise later, according to a review published in June from researchers at the University of California, San Francisco. But even people with mild symptoms can struggle to regain their previous levels of fitness.
“We have participants in our study who had relatively mild acute symptoms and went on to have really profound decreases in their ability to exercise,” said Matthew S. Durstenfeld, MD, a cardiologist at UCSF and principal author of the review.
Most people with long COVID will have lower-than-expected scores on tests of aerobic fitness, as shown by Yale researchers in a study published in August 2021.
“Some amount of that is due to deconditioning,” Dr. Durstenfeld said. “You’re not feeling well, so you’re not exercising to the same degree you might have been before you got infected.”
In a study published in April, people with long COVID told researchers at Britain’s University of Leeds they spent 93% less time in physical activity than they did before their infection.
But multiple studies have found deconditioning is not entirely – or even mostly – to blame.
A 2021 study found that 89% of participants with long COVID had postexertional malaise (PEM), which happens when a patient’s symptoms get worse after they do even minor physical or mental activities. According to the CDC, postexertional malaise can hit as long as 12-48 hours after the activity, and it can take people up to 2 weeks to fully recover.
Unfortunately, the advice patients get from their doctors sometimes makes the problem worse.
How long COVID defies simple solutions
Long COVID is a “dynamic disability” that requires health professionals to go off script when a patient’s symptoms don’t respond in a predictable way to treatment, said David Putrino, PhD, a neuroscientist, physical therapist, and director of rehabilitation innovation for the Mount Sinai Health System in New York.
“We’re not so good at dealing with somebody who, for all intents and purposes, can appear healthy and nondisabled on one day and be completely debilitated the next day,” he said.
Dr. Putrino said more than half of his clinic’s long-COVID patients told his team they had at least one of these persistent problems:
- Fatigue (82%).
- Brain fog (67%).
- Headache (60%).
- Sleep problems (59%).
- Dizziness (54%).
And 86% said exercise worsened their symptoms.
The symptoms are similar to what doctors see with illnesses such as lupus, Lyme disease, and chronic fatigue syndrome – something many experts compare long COVID to. Researchers and medical professionals still don’t know exactly how COVID-19 causes those symptoms. But there are some theories.
Potential causes of long-COVID symptoms
Dr. Putrino said it is possible the virus enters a patient’s cells and hijacks the mitochondria – a part of the cell that provides energy. It can linger there for weeks or months – something known as viral persistence.
“All of a sudden, the body’s getting less energy for itself, even though it’s producing the same amount, or even a little more,” he said. And there is a consequence to this extra stress on the cells. “Creating energy isn’t free. You’re producing more waste products, which puts your body in a state of oxidative stress,” Dr. Putrino said. Oxidative stress damages cells as molecules interact with oxygen in harmful ways.
“The other big mechanism is autonomic dysfunction,” Dr. Putrino said. It’s marked by breathing problems, heart palpitations, and other glitches in areas most healthy people never have to think about. About 70% of long-COVID patients at Mount Sinai’s clinic have some degree of autonomic dysfunction, he said.
For a person with autonomic dysfunction, something as basic as changing posture can trigger a storm of cytokines, a chemical messenger that tells the immune system where and how to respond to challenges like an injury or infection.
“Suddenly, you have this on-off switch,” Dr. Putrino said. “You go straight to ‘fight or flight,’ ” with a surge of adrenaline and a spiking heart rate, “then plunge back to ‘rest or digest.’ You go from fired up to so sleepy, you can’t keep your eyes open.”
A patient with viral persistence and one with autonomic dysfunction may have the same negative reaction to exercise, even though the triggers are completely different.
So how can doctors help long-COVID patients?
The first step, Dr. Putrino said, is to understand the difference between long COVID and a long recovery from COVID-19 infection.
Many of the patients in the latter group still have symptoms 4 weeks after their first infection. “At 4 weeks, yeah, they’re still feeling symptoms, but that’s not long COVID,” he said. “That’s just taking a while to get over a viral infection.”
Fitness advice is simple for those people: Take it easy at first, and gradually increase the amount and intensity of aerobic exercise and strength training.
But that advice would be disastrous for someone who meets Dr. Putrino’s stricter definition of long COVID: “Three to 4 months out from initial infection, they’re experiencing severe fatigue, exertional symptoms, cognitive symptoms, heart palpitations, shortness of breath,” he said.
“Our clinic is extraordinarily cautious with exercise” for those patients, he said.
In Dr. Putrino’s experience, about 20%-30% of patients will make significant progress after 12 weeks. “They’re feeling more or less like they felt pre-COVID,” he said.
The unluckiest 10%-20% won’t make any progress at all. Any type of therapy, even if it’s as simple as moving their legs from a flat position, worsens their symptoms.
The majority – 50%-60% – will have some improvement in their symptoms. But then progress will stop, for reasons researchers are still trying to figure out.
“My sense is that gradually increasing your exercise is still good advice for the vast majority of people,” UCSF’s Dr. Durstenfeld said.
Ideally, that exercise will be supervised by someone trained in cardiac, pulmonary, and/or autonomic rehabilitation – a specialized type of therapy aimed at resyncing the autonomic nervous system that governs breathing and other unconscious functions, he said. But those therapies are rarely covered by insurance, which means most long-COVID patients are on their own.
Dr. Durstenfeld said it’s important that patients keep trying and not give up. “With slow and steady progress, a lot of people can get profoundly better,” he said.
Mr. Fram, who’s worked with careful supervision, says he’s getting closer to something like his pre-COVID-19 life.
But he’s not there yet. Long COVID, he said, “affects my life every single day.”
A version of this article first appeared on WebMD.com.
The gut microbes have spoken: All fiber is good fiber
Finding a fiber of good moral fiber
If you’ve ever wandered into the supplement aisle at your local grocery store, you’ve probably noticed an overabundance of fiber supplements that claim to do this for you and benefit that. Since there’s no Food and Drug Administration regulation on fiber supplements, manufacturers are free to (and do) make whatever wild claims they like. And much like choosing which of 500 shows to watch on Netflix, when you’re spoiled for choice, it can be difficult to pick.
Enter a team of molecular geneticists and microbiologists from Duke University. They can’t tell you what show to watch next, but they can tell you which fiber to choose, thanks to their new study. And the answer? Yes.
Well that’s not very helpful, but let us explain. For their study, a group of 28 received three of the main fiber supplements (inulin, dextrin, and galactooligosaccharides) for a week each, followed by a week off of fibers for their gut to return to baseline until they’d received all three. Those who consumed the least fiber at baseline saw the greatest benefit from fiber supplementation, with no appreciable difference between the three types. It was the same story for study participants who already consumed enough fiber; because their guts already hosted a more-optimal microbiome, the type of supplement didn’t matter. The benefits were the same across the board.
In an additional study, the Duke researchers found that gut microbiomes reacted to new fiber within a day, being primed to consume fiber on the first dose and digesting it more quickly on the second fiber dose.
The results, the researchers pointed out, make sense, since the average American only consumes 20%-40% of their daily recommended supply of fiber. Our digestive systems aren’t picky; they just want more, so go out there and choose whatever fiber you’d like. Do that, and then feel free to eat as many double bacon cheeseburgers as you’d like. That is the pinnacle of diet right there. Dietitians literally could not complain about it.
Jarlsberg vs. Camembert: This time it’s skeletal
Fiber is fabulous, of course, but the road to dietary health and wellness fulfillment takes us to many other, equally wondrous places. Hey, look! This next exit is covered with cheese.
All the cheeses are here, from Abbaye de Belloc to Zwitser, and there, right between the jalapeno cheddar and the Jermi tortes you’ll find Jarlsberg, a mild, semisoft, nutty-flavored cheese that comes from Jarlsberg in eastern Norway. A recent study also suggests that Jarlsberg may help to prevent osteopenia and osteoporosis.
A group of Norwegian investigators gathered together 66 healthy women and gave them a daily portion of either Jarlsberg or Camembert for 6 weeks, at which point the Camembert group was switched to Jarlsberg for another 6 weeks.
The research team choose Camembert because of its similarity to Jarlsberg in fat and protein content. Jarlsberg, however, also is rich in vitamin K2, which is important for bone health, and a substance known as DHNA, which “might combat bone thinning and increase bone tissue formation,” they said in a Eurekalert release.
After the first 6 weeks, blood levels of osteocalcin; vitamin K2; and PINP, a peptide involved in bone turnover, were significantly higher in the Jarlsberg group only. All those measures rose significantly after the switch from Camembert to Jarlsberg, while levels of total and LDL cholesterol “fell significantly in the Camembert group after they switched to Jarlsberg,” the team added.
But wait! There’s more! HbA1c fell significantly among those initially eating the Jarlsberg but rose sharply in those eating Camembert. Do you see where this is going? After the Camembert group made the switch to Jarlsberg, their HbA1c levels fell significantly as well.
So it’s not just a cheese thing: The effects are specific to Jarlsberg. Can you guess what we’re having for lunch? Double bacon and fiber Jarlsbergers. Mmm, Jarlsburgers.
Luck be a lady: The mother of twins
It’s widely believed that women who have twins must be more fertile, giving birth to more than one child at a time. Some studies have supported the idea, but more recent work is refuting that claim. In actuality, it might just be more statistics and luck than fertility after all.
Those earlier studies supporting fertility didn’t specify whether the chances of twin births were based on the ability to produce more than one egg at a time or on the number of births that women had overall. Looking at 100,000 preindustrial European births, before contraception was available, researchers from Norway, Germany, France, and the United Kingdom found that the number of total births, twins included, makes all the difference.
“When a woman gives birth several times, the chances increase that at least one of these births will be a twin birth,” investigator Gine Roll Skjærvø of the Norwegian University of Science and Technology said in a written statement.
Since twins occur in 1%-3% of all births, the more births that a woman has, the better her chances of giving birth to twins. The researchers compared it to playing the lottery. You buy enough tickets, eventually your numbers are going to come up. Despite that, however, they found that women who give birth to twins give birth less often than those who don’t have twins. Which raises the idea of sheer luck.
The researchers said that there’s still a lot to uncover in twin births, noting that “uncritically comparing groups of women with and without twins can trick us into believing the opposite of what is really true. These groupings may either hide the effects of twinning and fertility genes where they exist, or vice versa, create the illusion of these if they do not exist.”
For now, this new research claims that it’s basically a lottery. And women who give birth to twins hit the jackpot.
Those with low wages may be earning future memory loss
Not only are low wages detrimental to our souls, hopes, and dreams, but a new study shows that low wages also are linked to quicker memory decline later in life. Sustained low wages not only cause stress and food insecurity in the lives of many, but they also can cause diseases such as depression, obesity, and high blood pressure, which are risk factors for cognitive aging.
The study was conducted using records from the Health and Retirement Study for the years 1992-2016 and focused on 2,879 adults born between 1936 and 1941. The participants were divided into three groups: those who never earned low wages, those who sometimes did, and those who always did.
The investigators found that workers who earned sustained low wages – defined as an hourly wage lower than two-thirds of the federal median wage for the corresponding year – “experienced significantly faster memory decline in older age” than did those who never earned low wages.
There are signs of inflation everywhere we look these days, but many people are not earning higher wages to compensate for the extra expenses. “Increasing the federal minimum wage, for example to $15 per hour, remains a gridlock issue in Congress,” lead author Katrina Kezios of the Columbia University Mailman School of Public Health, said in a statement released by the university.
If only salaries would rise instead of prices for once.
Finding a fiber of good moral fiber
If you’ve ever wandered into the supplement aisle at your local grocery store, you’ve probably noticed an overabundance of fiber supplements that claim to do this for you and benefit that. Since there’s no Food and Drug Administration regulation on fiber supplements, manufacturers are free to (and do) make whatever wild claims they like. And much like choosing which of 500 shows to watch on Netflix, when you’re spoiled for choice, it can be difficult to pick.
Enter a team of molecular geneticists and microbiologists from Duke University. They can’t tell you what show to watch next, but they can tell you which fiber to choose, thanks to their new study. And the answer? Yes.
Well that’s not very helpful, but let us explain. For their study, a group of 28 received three of the main fiber supplements (inulin, dextrin, and galactooligosaccharides) for a week each, followed by a week off of fibers for their gut to return to baseline until they’d received all three. Those who consumed the least fiber at baseline saw the greatest benefit from fiber supplementation, with no appreciable difference between the three types. It was the same story for study participants who already consumed enough fiber; because their guts already hosted a more-optimal microbiome, the type of supplement didn’t matter. The benefits were the same across the board.
In an additional study, the Duke researchers found that gut microbiomes reacted to new fiber within a day, being primed to consume fiber on the first dose and digesting it more quickly on the second fiber dose.
The results, the researchers pointed out, make sense, since the average American only consumes 20%-40% of their daily recommended supply of fiber. Our digestive systems aren’t picky; they just want more, so go out there and choose whatever fiber you’d like. Do that, and then feel free to eat as many double bacon cheeseburgers as you’d like. That is the pinnacle of diet right there. Dietitians literally could not complain about it.
Jarlsberg vs. Camembert: This time it’s skeletal
Fiber is fabulous, of course, but the road to dietary health and wellness fulfillment takes us to many other, equally wondrous places. Hey, look! This next exit is covered with cheese.
All the cheeses are here, from Abbaye de Belloc to Zwitser, and there, right between the jalapeno cheddar and the Jermi tortes you’ll find Jarlsberg, a mild, semisoft, nutty-flavored cheese that comes from Jarlsberg in eastern Norway. A recent study also suggests that Jarlsberg may help to prevent osteopenia and osteoporosis.
A group of Norwegian investigators gathered together 66 healthy women and gave them a daily portion of either Jarlsberg or Camembert for 6 weeks, at which point the Camembert group was switched to Jarlsberg for another 6 weeks.
The research team choose Camembert because of its similarity to Jarlsberg in fat and protein content. Jarlsberg, however, also is rich in vitamin K2, which is important for bone health, and a substance known as DHNA, which “might combat bone thinning and increase bone tissue formation,” they said in a Eurekalert release.
After the first 6 weeks, blood levels of osteocalcin; vitamin K2; and PINP, a peptide involved in bone turnover, were significantly higher in the Jarlsberg group only. All those measures rose significantly after the switch from Camembert to Jarlsberg, while levels of total and LDL cholesterol “fell significantly in the Camembert group after they switched to Jarlsberg,” the team added.
But wait! There’s more! HbA1c fell significantly among those initially eating the Jarlsberg but rose sharply in those eating Camembert. Do you see where this is going? After the Camembert group made the switch to Jarlsberg, their HbA1c levels fell significantly as well.
So it’s not just a cheese thing: The effects are specific to Jarlsberg. Can you guess what we’re having for lunch? Double bacon and fiber Jarlsbergers. Mmm, Jarlsburgers.
Luck be a lady: The mother of twins
It’s widely believed that women who have twins must be more fertile, giving birth to more than one child at a time. Some studies have supported the idea, but more recent work is refuting that claim. In actuality, it might just be more statistics and luck than fertility after all.
Those earlier studies supporting fertility didn’t specify whether the chances of twin births were based on the ability to produce more than one egg at a time or on the number of births that women had overall. Looking at 100,000 preindustrial European births, before contraception was available, researchers from Norway, Germany, France, and the United Kingdom found that the number of total births, twins included, makes all the difference.
“When a woman gives birth several times, the chances increase that at least one of these births will be a twin birth,” investigator Gine Roll Skjærvø of the Norwegian University of Science and Technology said in a written statement.
Since twins occur in 1%-3% of all births, the more births that a woman has, the better her chances of giving birth to twins. The researchers compared it to playing the lottery. You buy enough tickets, eventually your numbers are going to come up. Despite that, however, they found that women who give birth to twins give birth less often than those who don’t have twins. Which raises the idea of sheer luck.
The researchers said that there’s still a lot to uncover in twin births, noting that “uncritically comparing groups of women with and without twins can trick us into believing the opposite of what is really true. These groupings may either hide the effects of twinning and fertility genes where they exist, or vice versa, create the illusion of these if they do not exist.”
For now, this new research claims that it’s basically a lottery. And women who give birth to twins hit the jackpot.
Those with low wages may be earning future memory loss
Not only are low wages detrimental to our souls, hopes, and dreams, but a new study shows that low wages also are linked to quicker memory decline later in life. Sustained low wages not only cause stress and food insecurity in the lives of many, but they also can cause diseases such as depression, obesity, and high blood pressure, which are risk factors for cognitive aging.
The study was conducted using records from the Health and Retirement Study for the years 1992-2016 and focused on 2,879 adults born between 1936 and 1941. The participants were divided into three groups: those who never earned low wages, those who sometimes did, and those who always did.
The investigators found that workers who earned sustained low wages – defined as an hourly wage lower than two-thirds of the federal median wage for the corresponding year – “experienced significantly faster memory decline in older age” than did those who never earned low wages.
There are signs of inflation everywhere we look these days, but many people are not earning higher wages to compensate for the extra expenses. “Increasing the federal minimum wage, for example to $15 per hour, remains a gridlock issue in Congress,” lead author Katrina Kezios of the Columbia University Mailman School of Public Health, said in a statement released by the university.
If only salaries would rise instead of prices for once.
Finding a fiber of good moral fiber
If you’ve ever wandered into the supplement aisle at your local grocery store, you’ve probably noticed an overabundance of fiber supplements that claim to do this for you and benefit that. Since there’s no Food and Drug Administration regulation on fiber supplements, manufacturers are free to (and do) make whatever wild claims they like. And much like choosing which of 500 shows to watch on Netflix, when you’re spoiled for choice, it can be difficult to pick.
Enter a team of molecular geneticists and microbiologists from Duke University. They can’t tell you what show to watch next, but they can tell you which fiber to choose, thanks to their new study. And the answer? Yes.
Well that’s not very helpful, but let us explain. For their study, a group of 28 received three of the main fiber supplements (inulin, dextrin, and galactooligosaccharides) for a week each, followed by a week off of fibers for their gut to return to baseline until they’d received all three. Those who consumed the least fiber at baseline saw the greatest benefit from fiber supplementation, with no appreciable difference between the three types. It was the same story for study participants who already consumed enough fiber; because their guts already hosted a more-optimal microbiome, the type of supplement didn’t matter. The benefits were the same across the board.
In an additional study, the Duke researchers found that gut microbiomes reacted to new fiber within a day, being primed to consume fiber on the first dose and digesting it more quickly on the second fiber dose.
The results, the researchers pointed out, make sense, since the average American only consumes 20%-40% of their daily recommended supply of fiber. Our digestive systems aren’t picky; they just want more, so go out there and choose whatever fiber you’d like. Do that, and then feel free to eat as many double bacon cheeseburgers as you’d like. That is the pinnacle of diet right there. Dietitians literally could not complain about it.
Jarlsberg vs. Camembert: This time it’s skeletal
Fiber is fabulous, of course, but the road to dietary health and wellness fulfillment takes us to many other, equally wondrous places. Hey, look! This next exit is covered with cheese.
All the cheeses are here, from Abbaye de Belloc to Zwitser, and there, right between the jalapeno cheddar and the Jermi tortes you’ll find Jarlsberg, a mild, semisoft, nutty-flavored cheese that comes from Jarlsberg in eastern Norway. A recent study also suggests that Jarlsberg may help to prevent osteopenia and osteoporosis.
A group of Norwegian investigators gathered together 66 healthy women and gave them a daily portion of either Jarlsberg or Camembert for 6 weeks, at which point the Camembert group was switched to Jarlsberg for another 6 weeks.
The research team choose Camembert because of its similarity to Jarlsberg in fat and protein content. Jarlsberg, however, also is rich in vitamin K2, which is important for bone health, and a substance known as DHNA, which “might combat bone thinning and increase bone tissue formation,” they said in a Eurekalert release.
After the first 6 weeks, blood levels of osteocalcin; vitamin K2; and PINP, a peptide involved in bone turnover, were significantly higher in the Jarlsberg group only. All those measures rose significantly after the switch from Camembert to Jarlsberg, while levels of total and LDL cholesterol “fell significantly in the Camembert group after they switched to Jarlsberg,” the team added.
But wait! There’s more! HbA1c fell significantly among those initially eating the Jarlsberg but rose sharply in those eating Camembert. Do you see where this is going? After the Camembert group made the switch to Jarlsberg, their HbA1c levels fell significantly as well.
So it’s not just a cheese thing: The effects are specific to Jarlsberg. Can you guess what we’re having for lunch? Double bacon and fiber Jarlsbergers. Mmm, Jarlsburgers.
Luck be a lady: The mother of twins
It’s widely believed that women who have twins must be more fertile, giving birth to more than one child at a time. Some studies have supported the idea, but more recent work is refuting that claim. In actuality, it might just be more statistics and luck than fertility after all.
Those earlier studies supporting fertility didn’t specify whether the chances of twin births were based on the ability to produce more than one egg at a time or on the number of births that women had overall. Looking at 100,000 preindustrial European births, before contraception was available, researchers from Norway, Germany, France, and the United Kingdom found that the number of total births, twins included, makes all the difference.
“When a woman gives birth several times, the chances increase that at least one of these births will be a twin birth,” investigator Gine Roll Skjærvø of the Norwegian University of Science and Technology said in a written statement.
Since twins occur in 1%-3% of all births, the more births that a woman has, the better her chances of giving birth to twins. The researchers compared it to playing the lottery. You buy enough tickets, eventually your numbers are going to come up. Despite that, however, they found that women who give birth to twins give birth less often than those who don’t have twins. Which raises the idea of sheer luck.
The researchers said that there’s still a lot to uncover in twin births, noting that “uncritically comparing groups of women with and without twins can trick us into believing the opposite of what is really true. These groupings may either hide the effects of twinning and fertility genes where they exist, or vice versa, create the illusion of these if they do not exist.”
For now, this new research claims that it’s basically a lottery. And women who give birth to twins hit the jackpot.
Those with low wages may be earning future memory loss
Not only are low wages detrimental to our souls, hopes, and dreams, but a new study shows that low wages also are linked to quicker memory decline later in life. Sustained low wages not only cause stress and food insecurity in the lives of many, but they also can cause diseases such as depression, obesity, and high blood pressure, which are risk factors for cognitive aging.
The study was conducted using records from the Health and Retirement Study for the years 1992-2016 and focused on 2,879 adults born between 1936 and 1941. The participants were divided into three groups: those who never earned low wages, those who sometimes did, and those who always did.
The investigators found that workers who earned sustained low wages – defined as an hourly wage lower than two-thirds of the federal median wage for the corresponding year – “experienced significantly faster memory decline in older age” than did those who never earned low wages.
There are signs of inflation everywhere we look these days, but many people are not earning higher wages to compensate for the extra expenses. “Increasing the federal minimum wage, for example to $15 per hour, remains a gridlock issue in Congress,” lead author Katrina Kezios of the Columbia University Mailman School of Public Health, said in a statement released by the university.
If only salaries would rise instead of prices for once.
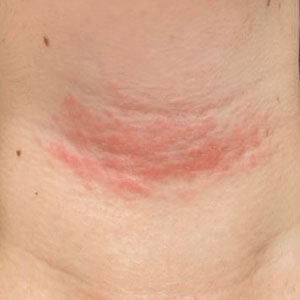
Long COVID comes in three forms: Study
, according to a new preprint study published on MedRxiv that hasn’t yet been peer-reviewed.
Long COVID has been hard to define due to its large number of symptoms, but researchers at King’s College London have identified three distinct profiles – with long-term symptoms focused on neurological, respiratory, or physical conditions. So far, they also found patterns among people infected with the original coronavirus strain, the Alpha variant, and the Delta variant.
“These data show clearly that post-COVID syndrome is not just one condition but appears to have several subtypes,” Claire Steves, PhD, one of the study authors and a senior clinical lecturer in King’s College London’s School of Life Course & Population Sciences, said in a statement.
“Understanding the root causes of these subtypes may help in finding treatment strategies,” she said. “Moreover, these data emphasize the need for long-COVID services to incorporate a personalized approach sensitive to the issues of each individual.”
The research team analyzed ZOE COVID app data for 1,459 people who have had symptoms for more than 84 days, or 12 weeks, according to their definition of long COVID or post-COVID syndrome.
They found that the largest group had a cluster of symptoms in the nervous system, such as fatigue, brain fog, and headaches. It was the most common subtype among the Alpha variant, which was dominant in winter 2020-2021, and the Delta variant, which was dominant in 2021.
The second group had respiratory symptoms, such as chest pain and severe shortness of breath, which could suggest lung damage, the researchers wrote. It was the largest cluster for the original coronavirus strain in spring 2020, when people were unvaccinated.
The third group included people who reported a diverse range of physical symptoms, including heart palpitations, muscle aches and pain, and changes to their skin and hair. This group had some of the “most severe and debilitating multi-organ symptoms,” the researchers wrote.
The researchers found that the subtypes were similar in vaccinated and unvaccinated people based on the variants investigated so far. But the data showed that the risk of long COVID was reduced by vaccination.
In addition, although the three subtypes were present in all the variants, other symptom clusters had subtle differences among the variants, such as symptoms in the stomach and intestines. The differences could be due to other things that changed during the pandemic, such as the time of year, social behaviors, and treatments, the researchers said.
“Machine learning approaches, such as clustering analysis, have made it possible to start exploring and identifying different profiles of post-COVID syndrome,” Marc Modat, PhD, who led the analysis and is a senior lecturer at King’s College London’s School of Biomedical Engineering & Imaging Sciences, said in the statement.
“This opens new avenues of research to better understand COVID-19 and to motivate clinical research that might mitigate the long-term effects of the disease,” he said.
A version of this article first appeared on WebMD.com.
, according to a new preprint study published on MedRxiv that hasn’t yet been peer-reviewed.
Long COVID has been hard to define due to its large number of symptoms, but researchers at King’s College London have identified three distinct profiles – with long-term symptoms focused on neurological, respiratory, or physical conditions. So far, they also found patterns among people infected with the original coronavirus strain, the Alpha variant, and the Delta variant.
“These data show clearly that post-COVID syndrome is not just one condition but appears to have several subtypes,” Claire Steves, PhD, one of the study authors and a senior clinical lecturer in King’s College London’s School of Life Course & Population Sciences, said in a statement.
“Understanding the root causes of these subtypes may help in finding treatment strategies,” she said. “Moreover, these data emphasize the need for long-COVID services to incorporate a personalized approach sensitive to the issues of each individual.”
The research team analyzed ZOE COVID app data for 1,459 people who have had symptoms for more than 84 days, or 12 weeks, according to their definition of long COVID or post-COVID syndrome.
They found that the largest group had a cluster of symptoms in the nervous system, such as fatigue, brain fog, and headaches. It was the most common subtype among the Alpha variant, which was dominant in winter 2020-2021, and the Delta variant, which was dominant in 2021.
The second group had respiratory symptoms, such as chest pain and severe shortness of breath, which could suggest lung damage, the researchers wrote. It was the largest cluster for the original coronavirus strain in spring 2020, when people were unvaccinated.
The third group included people who reported a diverse range of physical symptoms, including heart palpitations, muscle aches and pain, and changes to their skin and hair. This group had some of the “most severe and debilitating multi-organ symptoms,” the researchers wrote.
The researchers found that the subtypes were similar in vaccinated and unvaccinated people based on the variants investigated so far. But the data showed that the risk of long COVID was reduced by vaccination.
In addition, although the three subtypes were present in all the variants, other symptom clusters had subtle differences among the variants, such as symptoms in the stomach and intestines. The differences could be due to other things that changed during the pandemic, such as the time of year, social behaviors, and treatments, the researchers said.
“Machine learning approaches, such as clustering analysis, have made it possible to start exploring and identifying different profiles of post-COVID syndrome,” Marc Modat, PhD, who led the analysis and is a senior lecturer at King’s College London’s School of Biomedical Engineering & Imaging Sciences, said in the statement.
“This opens new avenues of research to better understand COVID-19 and to motivate clinical research that might mitigate the long-term effects of the disease,” he said.
A version of this article first appeared on WebMD.com.
, according to a new preprint study published on MedRxiv that hasn’t yet been peer-reviewed.
Long COVID has been hard to define due to its large number of symptoms, but researchers at King’s College London have identified three distinct profiles – with long-term symptoms focused on neurological, respiratory, or physical conditions. So far, they also found patterns among people infected with the original coronavirus strain, the Alpha variant, and the Delta variant.
“These data show clearly that post-COVID syndrome is not just one condition but appears to have several subtypes,” Claire Steves, PhD, one of the study authors and a senior clinical lecturer in King’s College London’s School of Life Course & Population Sciences, said in a statement.
“Understanding the root causes of these subtypes may help in finding treatment strategies,” she said. “Moreover, these data emphasize the need for long-COVID services to incorporate a personalized approach sensitive to the issues of each individual.”
The research team analyzed ZOE COVID app data for 1,459 people who have had symptoms for more than 84 days, or 12 weeks, according to their definition of long COVID or post-COVID syndrome.
They found that the largest group had a cluster of symptoms in the nervous system, such as fatigue, brain fog, and headaches. It was the most common subtype among the Alpha variant, which was dominant in winter 2020-2021, and the Delta variant, which was dominant in 2021.
The second group had respiratory symptoms, such as chest pain and severe shortness of breath, which could suggest lung damage, the researchers wrote. It was the largest cluster for the original coronavirus strain in spring 2020, when people were unvaccinated.
The third group included people who reported a diverse range of physical symptoms, including heart palpitations, muscle aches and pain, and changes to their skin and hair. This group had some of the “most severe and debilitating multi-organ symptoms,” the researchers wrote.
The researchers found that the subtypes were similar in vaccinated and unvaccinated people based on the variants investigated so far. But the data showed that the risk of long COVID was reduced by vaccination.
In addition, although the three subtypes were present in all the variants, other symptom clusters had subtle differences among the variants, such as symptoms in the stomach and intestines. The differences could be due to other things that changed during the pandemic, such as the time of year, social behaviors, and treatments, the researchers said.
“Machine learning approaches, such as clustering analysis, have made it possible to start exploring and identifying different profiles of post-COVID syndrome,” Marc Modat, PhD, who led the analysis and is a senior lecturer at King’s College London’s School of Biomedical Engineering & Imaging Sciences, said in the statement.
“This opens new avenues of research to better understand COVID-19 and to motivate clinical research that might mitigate the long-term effects of the disease,” he said.
A version of this article first appeared on WebMD.com.
Death risk doubles for Black infants with bronchopulmonary dysplasia
Infants with bronchopulmonary dysplasia (BPD) who were born to Black mothers were significantly more likely to die or to have a longer hospital stay than infants of other ethnicities, based on data from more than 800 infants.
The overall incidence of BPD is rising, in part because of improved survival for extremely preterm infants, wrote Tamorah R. Lewis, MD, of the University of Missouri, Kansas City, and colleagues.
Previous studies suggest that racial disparities may affect outcomes for preterm infants with a range of neonatal morbidities during neonatal ICU (NICU) hospitalization, including respiratory distress syndrome, intraventricular hemorrhage, and necrotizing enterocolitis. However, the association of racial disparities with outcomes for preterm infants with BPD remains unclear, they said.
In a study published in JAMA Pediatrics, the researchers, on behalf of the Bronchopulmonary Dysplasia Collaborative, reviewed data from 834 preterm infants enrolled in the BPD Collaborative registry from Jan. 1, 2015, to July 19, 2021, at eight centers in the United States.
The study infants were born at less than 32 weeks’ gestation and were diagnosed with severe BPD according to the 2001 National Institutes of Health Consensus Criteria. The study population included 276 Black infants and 558 white infants. The median gestational age was 24 weeks, and 41% of the infants were female.
The primary outcomes were infant death and length of hospital stay.
Although death was infrequent (4% overall), Black maternal race was significantly associated with an increased risk of death from BPD (adjusted odds ratio, 2.1). Black maternal race also was significantly associated with a longer hospital stay for the infants, with an adjusted between-group difference of 10 days.
Infants of Black mothers also were more likely than those with White mothers to receive invasive respiratory support at the time of delivery. Black infants were more likely than White infants to have lower gestational age, lower birth weight and length, and smaller head circumference.
However, the proportions of cesarean deliveries, gender distribution, and infants small for gestational age were similar between Black and White infant groups. Medication exposure at 36 weeks postmenstrual age (PMA) also was similar for Black and White infants, and 50% of patients overall were treated with nasal continuous positive airway pressure at 36 weeks’ PMA. Awareness of the increased risk of death and longer hospital stay for Black infants is critical, “given the highly variable outcomes for patients with BPD and the uncertainty regarding demographic factors that contribute to late respiratory morbidity in severe BPD,” the researchers wrote.
The study findings were limited by several factors including variations among study centers in the identification and recording of maternal race, lack of data on paternal race, and the focus specifically on Black maternal race and not other ethnicities. Given the documented health disparities for Black individuals in the United States, “we restricted our cohort to only those patients born to Black or White mothers to estimate the association of Black maternal race and adverse in-hospital outcomes in infants with severe BPD,” the researchers wrote
Other limitations include the lack of data surrounding infant death and inability to adjust for all potential modifiers of BPD pathogenesis and progression, such as BPD comorbidities.
Prospective studies are needed to identify the sociodemographic mechanisms that may contribute to health outcome disparities for Black infants with severe BPD, the researchers emphasized.
In the meantime, the results highlight the need for more attention to variations in care for infants with BPD of different races, and approaches to family-centered care should consider “the precise needs of high-risk, structurally disadvantaged families while informing the design of prospective trials that improve outcomes for high-risk subgroups of children with severe BPD,” they concluded.
Data raise questions about the origin of disparities
The current study findings contribute to the knowledge and awareness of disparities in the high-risk NICU population, Nicolas A. Bamat, MD, and colleagues wrote in an accompanying editorial. “Further, their findings oppose the central tendency in the literature: that infants of Black mothers have less severe lung disease of prematurity during the birth hospitalization.”
The editorial authors noted that the study’s inclusion of racial characteristics as confounding variables to assess the effect of race on health “can imply questionable assumptions about where in a causal pathway racism begins to exert an effect,” whether after a diagnosis of BPD, during pregnancy in response to inequitable obstetric care, or “centuries ago, propagating forward through the shared experience of communities oppressed by the legacy of racism and its ongoing contemporary manifestations.”
The editorial authors added that, “in lung disease of prematurity, few variables are reliable antecedents to race as an exposure. Complex adjustment is necessary to reduce bias in targeted research questions.” However, the current study findings highlight the need to move toward more equitable neonatal care, and to prioritize interventions to reduce racial health disparities at the level of the NICU as well as at the hospital and government policy levels.
Consider range of contributing factors and confounders
The current study is important because “it is imperative to measure racial outcomes in health care in order to highlight and address disparities and biases,” Tim Joos, MD, said in an interview. However, “it can be difficult to determine how much race is a factor in itself versus a proxy for other important characteristics, such as socioeconomic status and level of education, that can confound the results.”
In the current study, the twofold-increased death rate in the premature infants of Black mothers is concerning and deserves further attention, Dr. Joos said. “The 10-day longer length of stay for infants of Black mothers seems quite shocking at first glance, but because of the long hospital stays for these extremely premature infants in general, it is about 7% longer than the infants born to White mothers.”
The take-home message is that this difference is still significant, and can reflect many factors including disease severity and complications, need for feeding assistance, teaching, and setting up home supports, said Dr. Joos.
As for additional research, “it would be useful for hospitals to break down why the differences exist, although I worry a provider or institution will feel they need to discharge Black families sooner to avoid being biased. Family preference and comfort level should be given high priority,” he emphasized.
The study received no outside funding, but lead author Dr. Lewis was supported by the National Institute on Child Health and Development and the Robert Wood Johnson Foundation. Several coauthors were supported by other grants from the National Institutes of Health. Dr. Barnat and one coauthor were supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Joos had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
Infants with bronchopulmonary dysplasia (BPD) who were born to Black mothers were significantly more likely to die or to have a longer hospital stay than infants of other ethnicities, based on data from more than 800 infants.
The overall incidence of BPD is rising, in part because of improved survival for extremely preterm infants, wrote Tamorah R. Lewis, MD, of the University of Missouri, Kansas City, and colleagues.
Previous studies suggest that racial disparities may affect outcomes for preterm infants with a range of neonatal morbidities during neonatal ICU (NICU) hospitalization, including respiratory distress syndrome, intraventricular hemorrhage, and necrotizing enterocolitis. However, the association of racial disparities with outcomes for preterm infants with BPD remains unclear, they said.
In a study published in JAMA Pediatrics, the researchers, on behalf of the Bronchopulmonary Dysplasia Collaborative, reviewed data from 834 preterm infants enrolled in the BPD Collaborative registry from Jan. 1, 2015, to July 19, 2021, at eight centers in the United States.
The study infants were born at less than 32 weeks’ gestation and were diagnosed with severe BPD according to the 2001 National Institutes of Health Consensus Criteria. The study population included 276 Black infants and 558 white infants. The median gestational age was 24 weeks, and 41% of the infants were female.
The primary outcomes were infant death and length of hospital stay.
Although death was infrequent (4% overall), Black maternal race was significantly associated with an increased risk of death from BPD (adjusted odds ratio, 2.1). Black maternal race also was significantly associated with a longer hospital stay for the infants, with an adjusted between-group difference of 10 days.
Infants of Black mothers also were more likely than those with White mothers to receive invasive respiratory support at the time of delivery. Black infants were more likely than White infants to have lower gestational age, lower birth weight and length, and smaller head circumference.
However, the proportions of cesarean deliveries, gender distribution, and infants small for gestational age were similar between Black and White infant groups. Medication exposure at 36 weeks postmenstrual age (PMA) also was similar for Black and White infants, and 50% of patients overall were treated with nasal continuous positive airway pressure at 36 weeks’ PMA. Awareness of the increased risk of death and longer hospital stay for Black infants is critical, “given the highly variable outcomes for patients with BPD and the uncertainty regarding demographic factors that contribute to late respiratory morbidity in severe BPD,” the researchers wrote.
The study findings were limited by several factors including variations among study centers in the identification and recording of maternal race, lack of data on paternal race, and the focus specifically on Black maternal race and not other ethnicities. Given the documented health disparities for Black individuals in the United States, “we restricted our cohort to only those patients born to Black or White mothers to estimate the association of Black maternal race and adverse in-hospital outcomes in infants with severe BPD,” the researchers wrote
Other limitations include the lack of data surrounding infant death and inability to adjust for all potential modifiers of BPD pathogenesis and progression, such as BPD comorbidities.
Prospective studies are needed to identify the sociodemographic mechanisms that may contribute to health outcome disparities for Black infants with severe BPD, the researchers emphasized.
In the meantime, the results highlight the need for more attention to variations in care for infants with BPD of different races, and approaches to family-centered care should consider “the precise needs of high-risk, structurally disadvantaged families while informing the design of prospective trials that improve outcomes for high-risk subgroups of children with severe BPD,” they concluded.
Data raise questions about the origin of disparities
The current study findings contribute to the knowledge and awareness of disparities in the high-risk NICU population, Nicolas A. Bamat, MD, and colleagues wrote in an accompanying editorial. “Further, their findings oppose the central tendency in the literature: that infants of Black mothers have less severe lung disease of prematurity during the birth hospitalization.”
The editorial authors noted that the study’s inclusion of racial characteristics as confounding variables to assess the effect of race on health “can imply questionable assumptions about where in a causal pathway racism begins to exert an effect,” whether after a diagnosis of BPD, during pregnancy in response to inequitable obstetric care, or “centuries ago, propagating forward through the shared experience of communities oppressed by the legacy of racism and its ongoing contemporary manifestations.”
The editorial authors added that, “in lung disease of prematurity, few variables are reliable antecedents to race as an exposure. Complex adjustment is necessary to reduce bias in targeted research questions.” However, the current study findings highlight the need to move toward more equitable neonatal care, and to prioritize interventions to reduce racial health disparities at the level of the NICU as well as at the hospital and government policy levels.
Consider range of contributing factors and confounders
The current study is important because “it is imperative to measure racial outcomes in health care in order to highlight and address disparities and biases,” Tim Joos, MD, said in an interview. However, “it can be difficult to determine how much race is a factor in itself versus a proxy for other important characteristics, such as socioeconomic status and level of education, that can confound the results.”
In the current study, the twofold-increased death rate in the premature infants of Black mothers is concerning and deserves further attention, Dr. Joos said. “The 10-day longer length of stay for infants of Black mothers seems quite shocking at first glance, but because of the long hospital stays for these extremely premature infants in general, it is about 7% longer than the infants born to White mothers.”
The take-home message is that this difference is still significant, and can reflect many factors including disease severity and complications, need for feeding assistance, teaching, and setting up home supports, said Dr. Joos.
As for additional research, “it would be useful for hospitals to break down why the differences exist, although I worry a provider or institution will feel they need to discharge Black families sooner to avoid being biased. Family preference and comfort level should be given high priority,” he emphasized.
The study received no outside funding, but lead author Dr. Lewis was supported by the National Institute on Child Health and Development and the Robert Wood Johnson Foundation. Several coauthors were supported by other grants from the National Institutes of Health. Dr. Barnat and one coauthor were supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Joos had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
Infants with bronchopulmonary dysplasia (BPD) who were born to Black mothers were significantly more likely to die or to have a longer hospital stay than infants of other ethnicities, based on data from more than 800 infants.
The overall incidence of BPD is rising, in part because of improved survival for extremely preterm infants, wrote Tamorah R. Lewis, MD, of the University of Missouri, Kansas City, and colleagues.
Previous studies suggest that racial disparities may affect outcomes for preterm infants with a range of neonatal morbidities during neonatal ICU (NICU) hospitalization, including respiratory distress syndrome, intraventricular hemorrhage, and necrotizing enterocolitis. However, the association of racial disparities with outcomes for preterm infants with BPD remains unclear, they said.
In a study published in JAMA Pediatrics, the researchers, on behalf of the Bronchopulmonary Dysplasia Collaborative, reviewed data from 834 preterm infants enrolled in the BPD Collaborative registry from Jan. 1, 2015, to July 19, 2021, at eight centers in the United States.
The study infants were born at less than 32 weeks’ gestation and were diagnosed with severe BPD according to the 2001 National Institutes of Health Consensus Criteria. The study population included 276 Black infants and 558 white infants. The median gestational age was 24 weeks, and 41% of the infants were female.
The primary outcomes were infant death and length of hospital stay.
Although death was infrequent (4% overall), Black maternal race was significantly associated with an increased risk of death from BPD (adjusted odds ratio, 2.1). Black maternal race also was significantly associated with a longer hospital stay for the infants, with an adjusted between-group difference of 10 days.
Infants of Black mothers also were more likely than those with White mothers to receive invasive respiratory support at the time of delivery. Black infants were more likely than White infants to have lower gestational age, lower birth weight and length, and smaller head circumference.
However, the proportions of cesarean deliveries, gender distribution, and infants small for gestational age were similar between Black and White infant groups. Medication exposure at 36 weeks postmenstrual age (PMA) also was similar for Black and White infants, and 50% of patients overall were treated with nasal continuous positive airway pressure at 36 weeks’ PMA. Awareness of the increased risk of death and longer hospital stay for Black infants is critical, “given the highly variable outcomes for patients with BPD and the uncertainty regarding demographic factors that contribute to late respiratory morbidity in severe BPD,” the researchers wrote.
The study findings were limited by several factors including variations among study centers in the identification and recording of maternal race, lack of data on paternal race, and the focus specifically on Black maternal race and not other ethnicities. Given the documented health disparities for Black individuals in the United States, “we restricted our cohort to only those patients born to Black or White mothers to estimate the association of Black maternal race and adverse in-hospital outcomes in infants with severe BPD,” the researchers wrote
Other limitations include the lack of data surrounding infant death and inability to adjust for all potential modifiers of BPD pathogenesis and progression, such as BPD comorbidities.
Prospective studies are needed to identify the sociodemographic mechanisms that may contribute to health outcome disparities for Black infants with severe BPD, the researchers emphasized.
In the meantime, the results highlight the need for more attention to variations in care for infants with BPD of different races, and approaches to family-centered care should consider “the precise needs of high-risk, structurally disadvantaged families while informing the design of prospective trials that improve outcomes for high-risk subgroups of children with severe BPD,” they concluded.
Data raise questions about the origin of disparities
The current study findings contribute to the knowledge and awareness of disparities in the high-risk NICU population, Nicolas A. Bamat, MD, and colleagues wrote in an accompanying editorial. “Further, their findings oppose the central tendency in the literature: that infants of Black mothers have less severe lung disease of prematurity during the birth hospitalization.”
The editorial authors noted that the study’s inclusion of racial characteristics as confounding variables to assess the effect of race on health “can imply questionable assumptions about where in a causal pathway racism begins to exert an effect,” whether after a diagnosis of BPD, during pregnancy in response to inequitable obstetric care, or “centuries ago, propagating forward through the shared experience of communities oppressed by the legacy of racism and its ongoing contemporary manifestations.”
The editorial authors added that, “in lung disease of prematurity, few variables are reliable antecedents to race as an exposure. Complex adjustment is necessary to reduce bias in targeted research questions.” However, the current study findings highlight the need to move toward more equitable neonatal care, and to prioritize interventions to reduce racial health disparities at the level of the NICU as well as at the hospital and government policy levels.
Consider range of contributing factors and confounders
The current study is important because “it is imperative to measure racial outcomes in health care in order to highlight and address disparities and biases,” Tim Joos, MD, said in an interview. However, “it can be difficult to determine how much race is a factor in itself versus a proxy for other important characteristics, such as socioeconomic status and level of education, that can confound the results.”
In the current study, the twofold-increased death rate in the premature infants of Black mothers is concerning and deserves further attention, Dr. Joos said. “The 10-day longer length of stay for infants of Black mothers seems quite shocking at first glance, but because of the long hospital stays for these extremely premature infants in general, it is about 7% longer than the infants born to White mothers.”
The take-home message is that this difference is still significant, and can reflect many factors including disease severity and complications, need for feeding assistance, teaching, and setting up home supports, said Dr. Joos.
As for additional research, “it would be useful for hospitals to break down why the differences exist, although I worry a provider or institution will feel they need to discharge Black families sooner to avoid being biased. Family preference and comfort level should be given high priority,” he emphasized.
The study received no outside funding, but lead author Dr. Lewis was supported by the National Institute on Child Health and Development and the Robert Wood Johnson Foundation. Several coauthors were supported by other grants from the National Institutes of Health. Dr. Barnat and one coauthor were supported by the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Joos had no financial conflicts to disclose and serves on the editorial advisory board of Pediatric News.
FROM JAMA PEDIATRICS
Burnout and stress of today: How do we cope?
Interestingly, the group that seems to be least impacted by this was health care administrators (with 12% of them planning on leaving their jobs).
I couldn’t stop thinking about these percentages.
I am reminded every day of the commitment and excellence of my colleagues in the health care field, and I do not want to lose them. I am hoping the following information and my thoughts on this topic will be helpful for those thinking about leaving health care.
Surgeon general’s burnout report
The surgeon general recently released a report on addressing health care worker burnout.2 It includes several very interesting and appropriate observations. I will summarize the most important ones here:
1. Our health depends on the well-being of our health workforce.
2. Direct harm to health care workers can lead to anxiety, depression, insomnia, and interpersonal and relationship struggles.
3. Health care workers experience exhaustion from providing overwhelming care and empathy.
4. Health care workers spend less time with patients and too much time with EHRs.
5. There are health workforce shortages.
The report is comprehensive, and everything in it is correct. The real issue is how does it go from being a report to true actionable items that we as health care professionals benefit from? I think in regards to exhaustion from overwhelming care responsibilities, and empathy fatigue, we need better boundaries.
Those who go into medicine, and especially those who go into primary care, always put the patients’ needs first. When operating in a broken system, it stays broken when individuals cover for the deficiencies in the system. Adding four extra patients every day because there is no one to refer them to with availability is injurious to the health care provider, and those providers who accept these additional patients will eventually be part of the 23% who want to leave their jobs. It feels awful to say no, but until the system stops accommodating there will not be substantial change.
The empathy drain
One of the unreported stresses of open access for patients through EHR communications is the empathy drain on physicians. When I see a patient in clinic with chronic symptoms or issues, I spend important time making sure we have a plan and an agreed upon time frame.
With the EHR, patients frequently send multiple messages for the same symptoms between visits. It is okay to redirect the patient and share that these issues will be discussed at length at appointments. My reasoning on this is that I think it is better for me to better care for myself and stay as the doctor for my patients, than always say yes to limitless needs and soon be looking for the off ramp.
The following statistic in the surgeon general’s report really hit home. For every hour of direct patient care, physicians currently spend 2 hours on the EHR system. Most practices allow 10%-20% of time for catch up, where with statistics like this it should be 50%. This concept is fully lost on administrators, or ignored.
It is only when we refuse to continue to accept and follow a broken system that it will change. A minority of internal medicine and family doctors (4.5% in 2018) practice in direct primary care models, where these issues are addressed. Unfortunately, this model as it is currently available is not an option for lower income patients.
A major theme in the surgeon general’s report was that administrative burdens need to be reduced by 75% by 2025. When I look at the report, I see the suggestions, I just don’t see how it will be achieved. Despite almost all clinics moving to the EHR, paperwork in the form of faxes and forms has increased.
A sweeping reform would be needed to eliminate daily faxes from PT offices, visiting nurse services, prior authorization, patients reminders from insurance companies, and disability forms from patients. I am glad that there is acknowledgment of the problem, but this change will take more than 3 years.
Takeaways
So what do we do?
Be good to yourself, and your colleagues. The pandemic has isolated us, which accelerates burnout.
Reach out to people you care about.
We are all feeling this. Set boundaries that allow you to care for yourself, and accept that you are doing your best, even if you can’t meet the needs of all your patients all the time.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Sinsky CA et al. Covid-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021 Dec;5(6):1165-73.
2. Addressing health worker burnout. The U.S. Surgeon General’s advisory on building a thriving health workforce.
Interestingly, the group that seems to be least impacted by this was health care administrators (with 12% of them planning on leaving their jobs).
I couldn’t stop thinking about these percentages.
I am reminded every day of the commitment and excellence of my colleagues in the health care field, and I do not want to lose them. I am hoping the following information and my thoughts on this topic will be helpful for those thinking about leaving health care.
Surgeon general’s burnout report
The surgeon general recently released a report on addressing health care worker burnout.2 It includes several very interesting and appropriate observations. I will summarize the most important ones here:
1. Our health depends on the well-being of our health workforce.
2. Direct harm to health care workers can lead to anxiety, depression, insomnia, and interpersonal and relationship struggles.
3. Health care workers experience exhaustion from providing overwhelming care and empathy.
4. Health care workers spend less time with patients and too much time with EHRs.
5. There are health workforce shortages.
The report is comprehensive, and everything in it is correct. The real issue is how does it go from being a report to true actionable items that we as health care professionals benefit from? I think in regards to exhaustion from overwhelming care responsibilities, and empathy fatigue, we need better boundaries.
Those who go into medicine, and especially those who go into primary care, always put the patients’ needs first. When operating in a broken system, it stays broken when individuals cover for the deficiencies in the system. Adding four extra patients every day because there is no one to refer them to with availability is injurious to the health care provider, and those providers who accept these additional patients will eventually be part of the 23% who want to leave their jobs. It feels awful to say no, but until the system stops accommodating there will not be substantial change.
The empathy drain
One of the unreported stresses of open access for patients through EHR communications is the empathy drain on physicians. When I see a patient in clinic with chronic symptoms or issues, I spend important time making sure we have a plan and an agreed upon time frame.
With the EHR, patients frequently send multiple messages for the same symptoms between visits. It is okay to redirect the patient and share that these issues will be discussed at length at appointments. My reasoning on this is that I think it is better for me to better care for myself and stay as the doctor for my patients, than always say yes to limitless needs and soon be looking for the off ramp.
The following statistic in the surgeon general’s report really hit home. For every hour of direct patient care, physicians currently spend 2 hours on the EHR system. Most practices allow 10%-20% of time for catch up, where with statistics like this it should be 50%. This concept is fully lost on administrators, or ignored.
It is only when we refuse to continue to accept and follow a broken system that it will change. A minority of internal medicine and family doctors (4.5% in 2018) practice in direct primary care models, where these issues are addressed. Unfortunately, this model as it is currently available is not an option for lower income patients.
A major theme in the surgeon general’s report was that administrative burdens need to be reduced by 75% by 2025. When I look at the report, I see the suggestions, I just don’t see how it will be achieved. Despite almost all clinics moving to the EHR, paperwork in the form of faxes and forms has increased.
A sweeping reform would be needed to eliminate daily faxes from PT offices, visiting nurse services, prior authorization, patients reminders from insurance companies, and disability forms from patients. I am glad that there is acknowledgment of the problem, but this change will take more than 3 years.
Takeaways
So what do we do?
Be good to yourself, and your colleagues. The pandemic has isolated us, which accelerates burnout.
Reach out to people you care about.
We are all feeling this. Set boundaries that allow you to care for yourself, and accept that you are doing your best, even if you can’t meet the needs of all your patients all the time.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Sinsky CA et al. Covid-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021 Dec;5(6):1165-73.
2. Addressing health worker burnout. The U.S. Surgeon General’s advisory on building a thriving health workforce.
Interestingly, the group that seems to be least impacted by this was health care administrators (with 12% of them planning on leaving their jobs).
I couldn’t stop thinking about these percentages.
I am reminded every day of the commitment and excellence of my colleagues in the health care field, and I do not want to lose them. I am hoping the following information and my thoughts on this topic will be helpful for those thinking about leaving health care.
Surgeon general’s burnout report
The surgeon general recently released a report on addressing health care worker burnout.2 It includes several very interesting and appropriate observations. I will summarize the most important ones here:
1. Our health depends on the well-being of our health workforce.
2. Direct harm to health care workers can lead to anxiety, depression, insomnia, and interpersonal and relationship struggles.
3. Health care workers experience exhaustion from providing overwhelming care and empathy.
4. Health care workers spend less time with patients and too much time with EHRs.
5. There are health workforce shortages.
The report is comprehensive, and everything in it is correct. The real issue is how does it go from being a report to true actionable items that we as health care professionals benefit from? I think in regards to exhaustion from overwhelming care responsibilities, and empathy fatigue, we need better boundaries.
Those who go into medicine, and especially those who go into primary care, always put the patients’ needs first. When operating in a broken system, it stays broken when individuals cover for the deficiencies in the system. Adding four extra patients every day because there is no one to refer them to with availability is injurious to the health care provider, and those providers who accept these additional patients will eventually be part of the 23% who want to leave their jobs. It feels awful to say no, but until the system stops accommodating there will not be substantial change.
The empathy drain
One of the unreported stresses of open access for patients through EHR communications is the empathy drain on physicians. When I see a patient in clinic with chronic symptoms or issues, I spend important time making sure we have a plan and an agreed upon time frame.
With the EHR, patients frequently send multiple messages for the same symptoms between visits. It is okay to redirect the patient and share that these issues will be discussed at length at appointments. My reasoning on this is that I think it is better for me to better care for myself and stay as the doctor for my patients, than always say yes to limitless needs and soon be looking for the off ramp.
The following statistic in the surgeon general’s report really hit home. For every hour of direct patient care, physicians currently spend 2 hours on the EHR system. Most practices allow 10%-20% of time for catch up, where with statistics like this it should be 50%. This concept is fully lost on administrators, or ignored.
It is only when we refuse to continue to accept and follow a broken system that it will change. A minority of internal medicine and family doctors (4.5% in 2018) practice in direct primary care models, where these issues are addressed. Unfortunately, this model as it is currently available is not an option for lower income patients.
A major theme in the surgeon general’s report was that administrative burdens need to be reduced by 75% by 2025. When I look at the report, I see the suggestions, I just don’t see how it will be achieved. Despite almost all clinics moving to the EHR, paperwork in the form of faxes and forms has increased.
A sweeping reform would be needed to eliminate daily faxes from PT offices, visiting nurse services, prior authorization, patients reminders from insurance companies, and disability forms from patients. I am glad that there is acknowledgment of the problem, but this change will take more than 3 years.
Takeaways
So what do we do?
Be good to yourself, and your colleagues. The pandemic has isolated us, which accelerates burnout.
Reach out to people you care about.
We are all feeling this. Set boundaries that allow you to care for yourself, and accept that you are doing your best, even if you can’t meet the needs of all your patients all the time.
Dr. Paauw is professor of medicine in the division of general internal medicine at the University of Washington, Seattle, and he serves as third-year medical student clerkship director at the University of Washington. He is a member of the editorial advisory board of Internal Medicine News. Dr. Paauw has no conflicts to disclose. Contact him at [email protected].
References
1. Sinsky CA et al. Covid-related stress and work intentions in a sample of US health care workers. Mayo Clin Proc Innov Qual Outcomes. 2021 Dec;5(6):1165-73.
2. Addressing health worker burnout. The U.S. Surgeon General’s advisory on building a thriving health workforce.
Cultural humility required to optimize treatment of eczema patients with skin of color
INDIANAPOLIS – Treating atopic dermatitis (AD) in children and adolescents with skin of color requires an acumen that extends well beyond the skin, said Candrice R. Heath, MD, at the annual meeting of the Society for Pediatric Dermatology.
This involves the practice of cultural humility, which Dr. Heath defined as a commitment to learn about all aspects of patients to truly understand them, including their race, access to health care, and socioeconomic status.
“We can continue to prioritize learning about all different types of skin tones and hair types, but we really have to commit to advocating for what our patients deserve in every way,” Dr. Heath, director of pediatric dermatology at Temple University, Philadelphia, said during her presentation at the meeting.
“That means advocating for kids to have access to better housing and for increasing health literacy programs in our hospitals, so that all our patients can understand what’s happening and how to navigate the health system,” she said. “It also means increasing diversity in our clinical trials by taking a few extra moments with the patient and family of color who might be eligible to participate in a clinical trial. We have work to do.”
To illustrate her points, she discussed the case of a 6-year-old Black patient, whose parents bring him into the clinic complaining about dark marks on the skin. The areas are itchy and the doctor figures, “this is a slam dunk; this is AD,” Dr. Heath said. “You talk about the diagnosis, and you give your treatment plan.
“But the issue is, in the parking lot when the patient’s family leaves, they feel like you didn’t help them at all,” she continued. “You didn’t understand what they came in for. They didn’t receive a treatment for what they came in for, because the initial complaint was dark marks on the skin, which is postinflammatory hyperpigmentation. We know that patients are distressed by this.”
As evidence, she cited a cross-sectional study that assessed the impact of hyperpigmentation and hyperchromia on quality of life in adults, published in the Journal of the American Academy of Dermatology. People who reported the highest levels of distress were women, those with postinflammatory hyperpigmentation, those with fewer formal years of education, and those who had higher out-of-pocket spending on skin-enhancing products.
“So, when you see hyperpigmentation in your AD patients of color, acknowledge it; say, ‘I see this pigmentation change,’ ” Dr. Heath advised. “Talk about how controlling the AD with a topical steroid or other treatment option can have a positive impact on that.”
However, she added that sometimes patients have steroid phobia, possibly because they believe the topical steroids are causing the pigmentation changes, “especially in cases of hypopigmentation, so I take the time to reassure patients so that they will not be fearful about using the medication.”
Parents of patients with skin of color who have AD may harbor other “invisible” concerns during office visits, she continued, including prior experiences with dermatologists that may not have been positive, difficulty accessing pediatric dermatologists, or a general mistrust of the health care system.
“All of that is going on in the room with your patients, particularly those with skin of color and those who feel marginalized,” said Dr. Heath, who is also a faculty scholar at Temple University medical school’s office of health equity, diversity and inclusion. “Of course, we can’t fix everything. But we can commit to approaching our visits with cultural humility.”
For patients with skin of color, she pointed out, other upstream effects impact AD care and outcomes, including well-documented socioeconomic factors.
“One of the equalizing factors is that we as pediatric dermatologists can think about increasing our education regarding skin of color,” Dr. Heath said.
For example, an analysis of data from the 2002 to 2012 National Inpatient Sample found that the main risk factors for inpatient hospitalization for AD were being non-White, having lowest-quartile household income, and having Medicaid or no insurance, researchers reported in 2018.
A separate multicenter study of 1,437 mother-child pairs with known AD found that non-Hispanic Black children and Hispanic children had greater odds of persistent AD than non-Hispanic White children, according to a 2019 study. Another large prospective cohort study published in 2019 found that AD prevalence and persistence is highest in U.S. urban children who are female or Black, and urban children with AD are more likely to have poor quality of life and asthma.
A few months after that study was published, researchers reported results from an analysis of data from the 2007-2008 National Survey of Children’s Health, which found that children who perceive the neighborhood they lived in as unsafe, unsupportive, or underdeveloped had a higher prevalence of AD and a higher severity of AD. The same year, a study of the social and economic risk factors for moderate to severe AD found that Black children were more likely to come from homes with a lower household income, lower parental education attainment, lack of home ownership, and live between two residences, and have exposure to smoke.
“Disease recognition is one thing, but we also want everyone to be aware of these other factors,” she said, “because some patients do need a little bit more care and help to be able to access the medications that they need and gain access to us.”
Follicular, nummular eczema
In her clinical experience, the most common clinical variants of AD in patients with skin of color is follicular eczema. “Examine the patient, apply your hand to the affected area, and you can feel the papules beneath your fingertips,” she advised.
“That’s what I teach my residents and medical students,” she said. “If you are looking for erythema to seal your diagnosis of AD, it may not happen. You may see more of a violaceous hue and sometimes you may not find it at all, depending on the patient’s skin tone. If I find an area of normal appearing skin and then look back at the area of active skin disease, I go back and forth until I’m able to train my eye to be able to see those violaceous and erythematous hues more easily.”
Nummular eczema can also be a challenge in AD patients with skin of color.
“I like to listen to buzz words,” Dr. Heath said. “If a parent says, ‘my child has been diagnosed with ringworm multiple times,’ I zoom in on that. We know that kids can get tinea corporis, but usually not multiple times. I ask about all the things that can be associated with AD, and often we do see these nummular plaques on the skin and do some education about that. I also talk to their pediatrician or send information to that person so that they can be aware that nummular eczema is a form of AD.”
She noted that AD of the scalp may be confused with tinea capitis, especially in young Black children with moderate to severe AD. In her experience, triamcinolone 0.1% ointment works well for AD of the scalp.
She concluded her presentation by noting that there is no easy solution to treating AD in young patients with skin of color. “It’s way more than just eczema. We can help people see AD in a different way. My goal is to see the value in challenging ourselves to understand the impact of what happens outside of the exam room on these patients.”
Dr. Heath disclosed that she has served as a consultant for several pharmaceutical companies, including Regeneron, Janssen, Arcutis, Johnson and Johnson, Cassiopea, and Lilly.
INDIANAPOLIS – Treating atopic dermatitis (AD) in children and adolescents with skin of color requires an acumen that extends well beyond the skin, said Candrice R. Heath, MD, at the annual meeting of the Society for Pediatric Dermatology.
This involves the practice of cultural humility, which Dr. Heath defined as a commitment to learn about all aspects of patients to truly understand them, including their race, access to health care, and socioeconomic status.
“We can continue to prioritize learning about all different types of skin tones and hair types, but we really have to commit to advocating for what our patients deserve in every way,” Dr. Heath, director of pediatric dermatology at Temple University, Philadelphia, said during her presentation at the meeting.
“That means advocating for kids to have access to better housing and for increasing health literacy programs in our hospitals, so that all our patients can understand what’s happening and how to navigate the health system,” she said. “It also means increasing diversity in our clinical trials by taking a few extra moments with the patient and family of color who might be eligible to participate in a clinical trial. We have work to do.”
To illustrate her points, she discussed the case of a 6-year-old Black patient, whose parents bring him into the clinic complaining about dark marks on the skin. The areas are itchy and the doctor figures, “this is a slam dunk; this is AD,” Dr. Heath said. “You talk about the diagnosis, and you give your treatment plan.
“But the issue is, in the parking lot when the patient’s family leaves, they feel like you didn’t help them at all,” she continued. “You didn’t understand what they came in for. They didn’t receive a treatment for what they came in for, because the initial complaint was dark marks on the skin, which is postinflammatory hyperpigmentation. We know that patients are distressed by this.”
As evidence, she cited a cross-sectional study that assessed the impact of hyperpigmentation and hyperchromia on quality of life in adults, published in the Journal of the American Academy of Dermatology. People who reported the highest levels of distress were women, those with postinflammatory hyperpigmentation, those with fewer formal years of education, and those who had higher out-of-pocket spending on skin-enhancing products.
“So, when you see hyperpigmentation in your AD patients of color, acknowledge it; say, ‘I see this pigmentation change,’ ” Dr. Heath advised. “Talk about how controlling the AD with a topical steroid or other treatment option can have a positive impact on that.”
However, she added that sometimes patients have steroid phobia, possibly because they believe the topical steroids are causing the pigmentation changes, “especially in cases of hypopigmentation, so I take the time to reassure patients so that they will not be fearful about using the medication.”
Parents of patients with skin of color who have AD may harbor other “invisible” concerns during office visits, she continued, including prior experiences with dermatologists that may not have been positive, difficulty accessing pediatric dermatologists, or a general mistrust of the health care system.
“All of that is going on in the room with your patients, particularly those with skin of color and those who feel marginalized,” said Dr. Heath, who is also a faculty scholar at Temple University medical school’s office of health equity, diversity and inclusion. “Of course, we can’t fix everything. But we can commit to approaching our visits with cultural humility.”
For patients with skin of color, she pointed out, other upstream effects impact AD care and outcomes, including well-documented socioeconomic factors.
“One of the equalizing factors is that we as pediatric dermatologists can think about increasing our education regarding skin of color,” Dr. Heath said.
For example, an analysis of data from the 2002 to 2012 National Inpatient Sample found that the main risk factors for inpatient hospitalization for AD were being non-White, having lowest-quartile household income, and having Medicaid or no insurance, researchers reported in 2018.
A separate multicenter study of 1,437 mother-child pairs with known AD found that non-Hispanic Black children and Hispanic children had greater odds of persistent AD than non-Hispanic White children, according to a 2019 study. Another large prospective cohort study published in 2019 found that AD prevalence and persistence is highest in U.S. urban children who are female or Black, and urban children with AD are more likely to have poor quality of life and asthma.
A few months after that study was published, researchers reported results from an analysis of data from the 2007-2008 National Survey of Children’s Health, which found that children who perceive the neighborhood they lived in as unsafe, unsupportive, or underdeveloped had a higher prevalence of AD and a higher severity of AD. The same year, a study of the social and economic risk factors for moderate to severe AD found that Black children were more likely to come from homes with a lower household income, lower parental education attainment, lack of home ownership, and live between two residences, and have exposure to smoke.
“Disease recognition is one thing, but we also want everyone to be aware of these other factors,” she said, “because some patients do need a little bit more care and help to be able to access the medications that they need and gain access to us.”
Follicular, nummular eczema
In her clinical experience, the most common clinical variants of AD in patients with skin of color is follicular eczema. “Examine the patient, apply your hand to the affected area, and you can feel the papules beneath your fingertips,” she advised.
“That’s what I teach my residents and medical students,” she said. “If you are looking for erythema to seal your diagnosis of AD, it may not happen. You may see more of a violaceous hue and sometimes you may not find it at all, depending on the patient’s skin tone. If I find an area of normal appearing skin and then look back at the area of active skin disease, I go back and forth until I’m able to train my eye to be able to see those violaceous and erythematous hues more easily.”
Nummular eczema can also be a challenge in AD patients with skin of color.
“I like to listen to buzz words,” Dr. Heath said. “If a parent says, ‘my child has been diagnosed with ringworm multiple times,’ I zoom in on that. We know that kids can get tinea corporis, but usually not multiple times. I ask about all the things that can be associated with AD, and often we do see these nummular plaques on the skin and do some education about that. I also talk to their pediatrician or send information to that person so that they can be aware that nummular eczema is a form of AD.”
She noted that AD of the scalp may be confused with tinea capitis, especially in young Black children with moderate to severe AD. In her experience, triamcinolone 0.1% ointment works well for AD of the scalp.
She concluded her presentation by noting that there is no easy solution to treating AD in young patients with skin of color. “It’s way more than just eczema. We can help people see AD in a different way. My goal is to see the value in challenging ourselves to understand the impact of what happens outside of the exam room on these patients.”
Dr. Heath disclosed that she has served as a consultant for several pharmaceutical companies, including Regeneron, Janssen, Arcutis, Johnson and Johnson, Cassiopea, and Lilly.
INDIANAPOLIS – Treating atopic dermatitis (AD) in children and adolescents with skin of color requires an acumen that extends well beyond the skin, said Candrice R. Heath, MD, at the annual meeting of the Society for Pediatric Dermatology.
This involves the practice of cultural humility, which Dr. Heath defined as a commitment to learn about all aspects of patients to truly understand them, including their race, access to health care, and socioeconomic status.
“We can continue to prioritize learning about all different types of skin tones and hair types, but we really have to commit to advocating for what our patients deserve in every way,” Dr. Heath, director of pediatric dermatology at Temple University, Philadelphia, said during her presentation at the meeting.
“That means advocating for kids to have access to better housing and for increasing health literacy programs in our hospitals, so that all our patients can understand what’s happening and how to navigate the health system,” she said. “It also means increasing diversity in our clinical trials by taking a few extra moments with the patient and family of color who might be eligible to participate in a clinical trial. We have work to do.”
To illustrate her points, she discussed the case of a 6-year-old Black patient, whose parents bring him into the clinic complaining about dark marks on the skin. The areas are itchy and the doctor figures, “this is a slam dunk; this is AD,” Dr. Heath said. “You talk about the diagnosis, and you give your treatment plan.
“But the issue is, in the parking lot when the patient’s family leaves, they feel like you didn’t help them at all,” she continued. “You didn’t understand what they came in for. They didn’t receive a treatment for what they came in for, because the initial complaint was dark marks on the skin, which is postinflammatory hyperpigmentation. We know that patients are distressed by this.”
As evidence, she cited a cross-sectional study that assessed the impact of hyperpigmentation and hyperchromia on quality of life in adults, published in the Journal of the American Academy of Dermatology. People who reported the highest levels of distress were women, those with postinflammatory hyperpigmentation, those with fewer formal years of education, and those who had higher out-of-pocket spending on skin-enhancing products.
“So, when you see hyperpigmentation in your AD patients of color, acknowledge it; say, ‘I see this pigmentation change,’ ” Dr. Heath advised. “Talk about how controlling the AD with a topical steroid or other treatment option can have a positive impact on that.”
However, she added that sometimes patients have steroid phobia, possibly because they believe the topical steroids are causing the pigmentation changes, “especially in cases of hypopigmentation, so I take the time to reassure patients so that they will not be fearful about using the medication.”
Parents of patients with skin of color who have AD may harbor other “invisible” concerns during office visits, she continued, including prior experiences with dermatologists that may not have been positive, difficulty accessing pediatric dermatologists, or a general mistrust of the health care system.
“All of that is going on in the room with your patients, particularly those with skin of color and those who feel marginalized,” said Dr. Heath, who is also a faculty scholar at Temple University medical school’s office of health equity, diversity and inclusion. “Of course, we can’t fix everything. But we can commit to approaching our visits with cultural humility.”
For patients with skin of color, she pointed out, other upstream effects impact AD care and outcomes, including well-documented socioeconomic factors.
“One of the equalizing factors is that we as pediatric dermatologists can think about increasing our education regarding skin of color,” Dr. Heath said.
For example, an analysis of data from the 2002 to 2012 National Inpatient Sample found that the main risk factors for inpatient hospitalization for AD were being non-White, having lowest-quartile household income, and having Medicaid or no insurance, researchers reported in 2018.
A separate multicenter study of 1,437 mother-child pairs with known AD found that non-Hispanic Black children and Hispanic children had greater odds of persistent AD than non-Hispanic White children, according to a 2019 study. Another large prospective cohort study published in 2019 found that AD prevalence and persistence is highest in U.S. urban children who are female or Black, and urban children with AD are more likely to have poor quality of life and asthma.
A few months after that study was published, researchers reported results from an analysis of data from the 2007-2008 National Survey of Children’s Health, which found that children who perceive the neighborhood they lived in as unsafe, unsupportive, or underdeveloped had a higher prevalence of AD and a higher severity of AD. The same year, a study of the social and economic risk factors for moderate to severe AD found that Black children were more likely to come from homes with a lower household income, lower parental education attainment, lack of home ownership, and live between two residences, and have exposure to smoke.
“Disease recognition is one thing, but we also want everyone to be aware of these other factors,” she said, “because some patients do need a little bit more care and help to be able to access the medications that they need and gain access to us.”
Follicular, nummular eczema
In her clinical experience, the most common clinical variants of AD in patients with skin of color is follicular eczema. “Examine the patient, apply your hand to the affected area, and you can feel the papules beneath your fingertips,” she advised.
“That’s what I teach my residents and medical students,” she said. “If you are looking for erythema to seal your diagnosis of AD, it may not happen. You may see more of a violaceous hue and sometimes you may not find it at all, depending on the patient’s skin tone. If I find an area of normal appearing skin and then look back at the area of active skin disease, I go back and forth until I’m able to train my eye to be able to see those violaceous and erythematous hues more easily.”
Nummular eczema can also be a challenge in AD patients with skin of color.
“I like to listen to buzz words,” Dr. Heath said. “If a parent says, ‘my child has been diagnosed with ringworm multiple times,’ I zoom in on that. We know that kids can get tinea corporis, but usually not multiple times. I ask about all the things that can be associated with AD, and often we do see these nummular plaques on the skin and do some education about that. I also talk to their pediatrician or send information to that person so that they can be aware that nummular eczema is a form of AD.”
She noted that AD of the scalp may be confused with tinea capitis, especially in young Black children with moderate to severe AD. In her experience, triamcinolone 0.1% ointment works well for AD of the scalp.
She concluded her presentation by noting that there is no easy solution to treating AD in young patients with skin of color. “It’s way more than just eczema. We can help people see AD in a different way. My goal is to see the value in challenging ourselves to understand the impact of what happens outside of the exam room on these patients.”
Dr. Heath disclosed that she has served as a consultant for several pharmaceutical companies, including Regeneron, Janssen, Arcutis, Johnson and Johnson, Cassiopea, and Lilly.
AT SPD 2022
Summer flu, RSV in July, ‘super colds?’
Richard Martinello, MD, a professor of medicine and pediatric infectious diseases at Yale University, New haven, Conn., doesn’t expect to see a child hospitalized with respiratory syncytial virus (RSV) in the middle of summer. The illness, which can strike infants and older adults especially hard, is known as a “winter virus.”
But not this year. Over the last several weeks, he says, admissions for children with RSV have increased at the Yale New Haven Children’s Hospital. While the numbers aren’t large, they are out of the ordinary, he says, “because usually, at this time of year, we see zero. For lack of a better term, it’s weird.”
Likewise, William Schaffner, MD, a professor of infectious diseases at Vanderbilt University in Nashville, says RSV is on the rise there. Tennessee is one of 10 states taking part in a Centers for Disease Control and Prevention surveillance system that tracks influenza, RSV, and COVID-19.
He says RSV cases have risen by at least a third during the past week, including all age ranges. At this time of year, he says, “We aren’t supposed to have any RSV.”
RSV isn’t the only virus thriving out of season or otherwise acting strangely. Since the pandemic began, flu seasons have been out of whack – sometimes nearly nonexistent and other times extending well beyond “normal” seasons. Some experts say one influenza “B” strain may now be extinct, while others say it will be back.
Severe colds – what some call “super colds” – also seem to be on the rise in recent warm-weather months, although that evidence is mostly based on personal experience, not science.
Trying to explain these out-of-season variations has sparked much discussion among epidemiologists and virologists, Dr. Schaffner says, with debates ongoing about whether human behavior and habits or the seasons play a bigger role in the transmission of viral illness.
On top of that, scientists are also looking at the interactions between the SARS-CoV-2 virus that causes COVID-19 and other viruses. When people get hit with COVID-19 and other viruses at the same time, does that make COVID-19 more severe, or less?
Research is conflicting.
Summer of 2022: A repeat of 2021?
RSV. Most children contract the virus by age 2, and while it’s generally mild, about 58,000 children under age 5 years are hospitalized each year. During the pandemic, RSV cases decreased from January to April 2020, the CDC reported, and then remained at “historically low levels”: less than 1% positive RSV results a week, for the next year.
But cases began rising in April 2021.
“Last year, we did have an unusual summer,” Dr. Schaffner says. After lockdown ended, to everyone’s surprise, RSV infections rose.
That increase triggered a CDC health advisory in June 2021, telling doctors and caregivers about the increase in “interseasonal” RSV cases across parts of the Southern United States, recommending broader testing for RSV in patients who had a respiratory illness but tested negative for COVID.
Because of the reduced circulation of RSV during the winter of 2020 to 2021, the CDC warned, older infants and toddlers might have a higher risk of RSV since they weren’t exposed to typical levels of RSV for the previous 15 months.
What about 2022? “At the moment,” Dr. Schaffner says, “it looks like we are having a repeat [of 2021].”
On Twitter, other pediatricians, including those from Maine and Texas, have reported an increase in RSV cases this summer.
Influenza. From October 2020 until May 2021, flu activity was lower than during any previous flu season since at least 1997, according to the CDC.
In late 2021, researchers suggested that one line of influenza known as B/Yamagata may have become extinct.
The 2021-2022 flu season has been mild, the CDC says, but it has come in two waves, with the second wave lingering longer than previous ones. While flu activity is decreasing, last week the CDC said doctors should be alert to flu infections throughout the summer.
Colds. In reports on colds that aren’t based on science, several doctors say they are seeing more colds than usual in the summer, and they’re more severe than usual. According to the CDC, common coronaviruses and respiratory adenoviruses have been increasing since early 2021, and rhinoviruses since June 2020.
Behavior vs. seasons
In explaining the spread of viral respiratory diseases, infectious disease doctors consider two things. “One is that temperature and humidity in the winter favors longer survival of some viruses, leading to longer periods of possible transmission,” says Dean Blumberg, MD, a professor of pediatrics and chief of pediatric infectious disease at University of California Davis Health.
“The other is differences in human behavior, with people spending more time outside in the summer, which results in more distancing and [less] virus concentration due to very large air volume,” he says, and vice versa in winter.
What about the “super colds?” COVID-19 lockdowns and social distancing greatly reduced people’s exposure to common viruses like those that cause colds, says Neil A. Mabbott, PhD, a professor of immunopathology at the University of Edinburgh (Scotland).
“Immunity to these common cold viruses gained through natural infection is considered to last around 8 or 9 months or so,” he says. “Each winter, when we are exposed to the new circulating variants of these viruses, our immunity receives a natural boost.”
That explains why most people get a cold that’s relatively mild. But with all the pandemic lockdowns and the use of hand sanitizers, most people had limited exposure to other viruses, including the common cold. When people emerged from lockdown, the common cold viruses were beginning to circulate again.
“Our immune systems were less able to clear the infection than previously,” Dr. Mabbott says. “As a consequence, some may have experienced increased symptoms, giving the impression of being infected with a ‘super cold.’ ”
“The colds themselves are probably not different to those we got prepandemic,” says Ian Mackay, PhD, a virologist at the University of Queensland, Brisbane, Australia. “But there might be more of them. So I doubt they are ‘super colds’ as much as they are ‘super-perfect circumstances.’ ”
The colds themselves are probably not different to those we got prepandemic. But there might be more of them.
Those super-perfect circumstances, he says, include people gathering after lockdown; a lack of immunity in new babies; viruses that have remained, even if at low levels, but continue to mutate; and our waning immunity to the range of viruses we’d normally encounter.
While lack of exposure may partly explain why some viruses become rampant out of season, it’s likely not the only reason. For example, the reduced circulation of RSV in the population as a whole also may have reduced the transfer of immunity from mothers to infants, some researchers say, making those infants more vulnerable than usual.
Interactions of viruses
Another thing that may be driving the different behavior of viruses is that the SARS-CoV-2 virus could somehow be interacting with other respiratory viruses, Dr. Schaffner says. “And if so, what sort of interactions?”
Many researchers are looking into that, and how coinfections with other respiratory diseases, including the common cold and flu, may affect the course of COVID-19. Some studies have found that the T cells – a source of deeper, cellular immunity in people – generated after a common cold “may also provide cross-protection in some people against COVID-19.”
But another study found immunity against common cold–causing coronaviruses might make COVID-19 more severe.
When researchers in the United Kingdom studied nearly 7,000 patients infected with COVID-19, including 583 also infected with RSV, flu, or adenoviruses (causing flulike or coldlike illness), those with flu or adenovirus, compared with the others, were at higher risk of death.
To be continued …
Exactly how COVID-19 will be changing what we know of other viruses is yet to be determined, too.
Even before the pandemic, Dr. Martinello says, there were already some shifts in RSV. Florida, for instance, has an RSV season longer than the rest of the country, mimicking the pattern in the tropics.
Will the atypical patterns continue? “My guess is that this will settle out,” he says, with some sort of pattern developing. At this point, there are many unknowns. “We still can’t answer whether there will be some seasonality to COVID.”
A version of this article first appeared on WebMD.com.
Richard Martinello, MD, a professor of medicine and pediatric infectious diseases at Yale University, New haven, Conn., doesn’t expect to see a child hospitalized with respiratory syncytial virus (RSV) in the middle of summer. The illness, which can strike infants and older adults especially hard, is known as a “winter virus.”
But not this year. Over the last several weeks, he says, admissions for children with RSV have increased at the Yale New Haven Children’s Hospital. While the numbers aren’t large, they are out of the ordinary, he says, “because usually, at this time of year, we see zero. For lack of a better term, it’s weird.”
Likewise, William Schaffner, MD, a professor of infectious diseases at Vanderbilt University in Nashville, says RSV is on the rise there. Tennessee is one of 10 states taking part in a Centers for Disease Control and Prevention surveillance system that tracks influenza, RSV, and COVID-19.
He says RSV cases have risen by at least a third during the past week, including all age ranges. At this time of year, he says, “We aren’t supposed to have any RSV.”
RSV isn’t the only virus thriving out of season or otherwise acting strangely. Since the pandemic began, flu seasons have been out of whack – sometimes nearly nonexistent and other times extending well beyond “normal” seasons. Some experts say one influenza “B” strain may now be extinct, while others say it will be back.
Severe colds – what some call “super colds” – also seem to be on the rise in recent warm-weather months, although that evidence is mostly based on personal experience, not science.
Trying to explain these out-of-season variations has sparked much discussion among epidemiologists and virologists, Dr. Schaffner says, with debates ongoing about whether human behavior and habits or the seasons play a bigger role in the transmission of viral illness.
On top of that, scientists are also looking at the interactions between the SARS-CoV-2 virus that causes COVID-19 and other viruses. When people get hit with COVID-19 and other viruses at the same time, does that make COVID-19 more severe, or less?
Research is conflicting.
Summer of 2022: A repeat of 2021?
RSV. Most children contract the virus by age 2, and while it’s generally mild, about 58,000 children under age 5 years are hospitalized each year. During the pandemic, RSV cases decreased from January to April 2020, the CDC reported, and then remained at “historically low levels”: less than 1% positive RSV results a week, for the next year.
But cases began rising in April 2021.
“Last year, we did have an unusual summer,” Dr. Schaffner says. After lockdown ended, to everyone’s surprise, RSV infections rose.
That increase triggered a CDC health advisory in June 2021, telling doctors and caregivers about the increase in “interseasonal” RSV cases across parts of the Southern United States, recommending broader testing for RSV in patients who had a respiratory illness but tested negative for COVID.
Because of the reduced circulation of RSV during the winter of 2020 to 2021, the CDC warned, older infants and toddlers might have a higher risk of RSV since they weren’t exposed to typical levels of RSV for the previous 15 months.
What about 2022? “At the moment,” Dr. Schaffner says, “it looks like we are having a repeat [of 2021].”
On Twitter, other pediatricians, including those from Maine and Texas, have reported an increase in RSV cases this summer.
Influenza. From October 2020 until May 2021, flu activity was lower than during any previous flu season since at least 1997, according to the CDC.
In late 2021, researchers suggested that one line of influenza known as B/Yamagata may have become extinct.
The 2021-2022 flu season has been mild, the CDC says, but it has come in two waves, with the second wave lingering longer than previous ones. While flu activity is decreasing, last week the CDC said doctors should be alert to flu infections throughout the summer.
Colds. In reports on colds that aren’t based on science, several doctors say they are seeing more colds than usual in the summer, and they’re more severe than usual. According to the CDC, common coronaviruses and respiratory adenoviruses have been increasing since early 2021, and rhinoviruses since June 2020.
Behavior vs. seasons
In explaining the spread of viral respiratory diseases, infectious disease doctors consider two things. “One is that temperature and humidity in the winter favors longer survival of some viruses, leading to longer periods of possible transmission,” says Dean Blumberg, MD, a professor of pediatrics and chief of pediatric infectious disease at University of California Davis Health.
“The other is differences in human behavior, with people spending more time outside in the summer, which results in more distancing and [less] virus concentration due to very large air volume,” he says, and vice versa in winter.
What about the “super colds?” COVID-19 lockdowns and social distancing greatly reduced people’s exposure to common viruses like those that cause colds, says Neil A. Mabbott, PhD, a professor of immunopathology at the University of Edinburgh (Scotland).
“Immunity to these common cold viruses gained through natural infection is considered to last around 8 or 9 months or so,” he says. “Each winter, when we are exposed to the new circulating variants of these viruses, our immunity receives a natural boost.”
That explains why most people get a cold that’s relatively mild. But with all the pandemic lockdowns and the use of hand sanitizers, most people had limited exposure to other viruses, including the common cold. When people emerged from lockdown, the common cold viruses were beginning to circulate again.
“Our immune systems were less able to clear the infection than previously,” Dr. Mabbott says. “As a consequence, some may have experienced increased symptoms, giving the impression of being infected with a ‘super cold.’ ”
“The colds themselves are probably not different to those we got prepandemic,” says Ian Mackay, PhD, a virologist at the University of Queensland, Brisbane, Australia. “But there might be more of them. So I doubt they are ‘super colds’ as much as they are ‘super-perfect circumstances.’ ”
The colds themselves are probably not different to those we got prepandemic. But there might be more of them.
Those super-perfect circumstances, he says, include people gathering after lockdown; a lack of immunity in new babies; viruses that have remained, even if at low levels, but continue to mutate; and our waning immunity to the range of viruses we’d normally encounter.
While lack of exposure may partly explain why some viruses become rampant out of season, it’s likely not the only reason. For example, the reduced circulation of RSV in the population as a whole also may have reduced the transfer of immunity from mothers to infants, some researchers say, making those infants more vulnerable than usual.
Interactions of viruses
Another thing that may be driving the different behavior of viruses is that the SARS-CoV-2 virus could somehow be interacting with other respiratory viruses, Dr. Schaffner says. “And if so, what sort of interactions?”
Many researchers are looking into that, and how coinfections with other respiratory diseases, including the common cold and flu, may affect the course of COVID-19. Some studies have found that the T cells – a source of deeper, cellular immunity in people – generated after a common cold “may also provide cross-protection in some people against COVID-19.”
But another study found immunity against common cold–causing coronaviruses might make COVID-19 more severe.
When researchers in the United Kingdom studied nearly 7,000 patients infected with COVID-19, including 583 also infected with RSV, flu, or adenoviruses (causing flulike or coldlike illness), those with flu or adenovirus, compared with the others, were at higher risk of death.
To be continued …
Exactly how COVID-19 will be changing what we know of other viruses is yet to be determined, too.
Even before the pandemic, Dr. Martinello says, there were already some shifts in RSV. Florida, for instance, has an RSV season longer than the rest of the country, mimicking the pattern in the tropics.
Will the atypical patterns continue? “My guess is that this will settle out,” he says, with some sort of pattern developing. At this point, there are many unknowns. “We still can’t answer whether there will be some seasonality to COVID.”
A version of this article first appeared on WebMD.com.
Richard Martinello, MD, a professor of medicine and pediatric infectious diseases at Yale University, New haven, Conn., doesn’t expect to see a child hospitalized with respiratory syncytial virus (RSV) in the middle of summer. The illness, which can strike infants and older adults especially hard, is known as a “winter virus.”
But not this year. Over the last several weeks, he says, admissions for children with RSV have increased at the Yale New Haven Children’s Hospital. While the numbers aren’t large, they are out of the ordinary, he says, “because usually, at this time of year, we see zero. For lack of a better term, it’s weird.”
Likewise, William Schaffner, MD, a professor of infectious diseases at Vanderbilt University in Nashville, says RSV is on the rise there. Tennessee is one of 10 states taking part in a Centers for Disease Control and Prevention surveillance system that tracks influenza, RSV, and COVID-19.
He says RSV cases have risen by at least a third during the past week, including all age ranges. At this time of year, he says, “We aren’t supposed to have any RSV.”
RSV isn’t the only virus thriving out of season or otherwise acting strangely. Since the pandemic began, flu seasons have been out of whack – sometimes nearly nonexistent and other times extending well beyond “normal” seasons. Some experts say one influenza “B” strain may now be extinct, while others say it will be back.
Severe colds – what some call “super colds” – also seem to be on the rise in recent warm-weather months, although that evidence is mostly based on personal experience, not science.
Trying to explain these out-of-season variations has sparked much discussion among epidemiologists and virologists, Dr. Schaffner says, with debates ongoing about whether human behavior and habits or the seasons play a bigger role in the transmission of viral illness.
On top of that, scientists are also looking at the interactions between the SARS-CoV-2 virus that causes COVID-19 and other viruses. When people get hit with COVID-19 and other viruses at the same time, does that make COVID-19 more severe, or less?
Research is conflicting.
Summer of 2022: A repeat of 2021?
RSV. Most children contract the virus by age 2, and while it’s generally mild, about 58,000 children under age 5 years are hospitalized each year. During the pandemic, RSV cases decreased from January to April 2020, the CDC reported, and then remained at “historically low levels”: less than 1% positive RSV results a week, for the next year.
But cases began rising in April 2021.
“Last year, we did have an unusual summer,” Dr. Schaffner says. After lockdown ended, to everyone’s surprise, RSV infections rose.
That increase triggered a CDC health advisory in June 2021, telling doctors and caregivers about the increase in “interseasonal” RSV cases across parts of the Southern United States, recommending broader testing for RSV in patients who had a respiratory illness but tested negative for COVID.
Because of the reduced circulation of RSV during the winter of 2020 to 2021, the CDC warned, older infants and toddlers might have a higher risk of RSV since they weren’t exposed to typical levels of RSV for the previous 15 months.
What about 2022? “At the moment,” Dr. Schaffner says, “it looks like we are having a repeat [of 2021].”
On Twitter, other pediatricians, including those from Maine and Texas, have reported an increase in RSV cases this summer.
Influenza. From October 2020 until May 2021, flu activity was lower than during any previous flu season since at least 1997, according to the CDC.
In late 2021, researchers suggested that one line of influenza known as B/Yamagata may have become extinct.
The 2021-2022 flu season has been mild, the CDC says, but it has come in two waves, with the second wave lingering longer than previous ones. While flu activity is decreasing, last week the CDC said doctors should be alert to flu infections throughout the summer.
Colds. In reports on colds that aren’t based on science, several doctors say they are seeing more colds than usual in the summer, and they’re more severe than usual. According to the CDC, common coronaviruses and respiratory adenoviruses have been increasing since early 2021, and rhinoviruses since June 2020.
Behavior vs. seasons
In explaining the spread of viral respiratory diseases, infectious disease doctors consider two things. “One is that temperature and humidity in the winter favors longer survival of some viruses, leading to longer periods of possible transmission,” says Dean Blumberg, MD, a professor of pediatrics and chief of pediatric infectious disease at University of California Davis Health.
“The other is differences in human behavior, with people spending more time outside in the summer, which results in more distancing and [less] virus concentration due to very large air volume,” he says, and vice versa in winter.
What about the “super colds?” COVID-19 lockdowns and social distancing greatly reduced people’s exposure to common viruses like those that cause colds, says Neil A. Mabbott, PhD, a professor of immunopathology at the University of Edinburgh (Scotland).
“Immunity to these common cold viruses gained through natural infection is considered to last around 8 or 9 months or so,” he says. “Each winter, when we are exposed to the new circulating variants of these viruses, our immunity receives a natural boost.”
That explains why most people get a cold that’s relatively mild. But with all the pandemic lockdowns and the use of hand sanitizers, most people had limited exposure to other viruses, including the common cold. When people emerged from lockdown, the common cold viruses were beginning to circulate again.
“Our immune systems were less able to clear the infection than previously,” Dr. Mabbott says. “As a consequence, some may have experienced increased symptoms, giving the impression of being infected with a ‘super cold.’ ”
“The colds themselves are probably not different to those we got prepandemic,” says Ian Mackay, PhD, a virologist at the University of Queensland, Brisbane, Australia. “But there might be more of them. So I doubt they are ‘super colds’ as much as they are ‘super-perfect circumstances.’ ”
The colds themselves are probably not different to those we got prepandemic. But there might be more of them.
Those super-perfect circumstances, he says, include people gathering after lockdown; a lack of immunity in new babies; viruses that have remained, even if at low levels, but continue to mutate; and our waning immunity to the range of viruses we’d normally encounter.
While lack of exposure may partly explain why some viruses become rampant out of season, it’s likely not the only reason. For example, the reduced circulation of RSV in the population as a whole also may have reduced the transfer of immunity from mothers to infants, some researchers say, making those infants more vulnerable than usual.
Interactions of viruses
Another thing that may be driving the different behavior of viruses is that the SARS-CoV-2 virus could somehow be interacting with other respiratory viruses, Dr. Schaffner says. “And if so, what sort of interactions?”
Many researchers are looking into that, and how coinfections with other respiratory diseases, including the common cold and flu, may affect the course of COVID-19. Some studies have found that the T cells – a source of deeper, cellular immunity in people – generated after a common cold “may also provide cross-protection in some people against COVID-19.”
But another study found immunity against common cold–causing coronaviruses might make COVID-19 more severe.
When researchers in the United Kingdom studied nearly 7,000 patients infected with COVID-19, including 583 also infected with RSV, flu, or adenoviruses (causing flulike or coldlike illness), those with flu or adenovirus, compared with the others, were at higher risk of death.
To be continued …
Exactly how COVID-19 will be changing what we know of other viruses is yet to be determined, too.
Even before the pandemic, Dr. Martinello says, there were already some shifts in RSV. Florida, for instance, has an RSV season longer than the rest of the country, mimicking the pattern in the tropics.
Will the atypical patterns continue? “My guess is that this will settle out,” he says, with some sort of pattern developing. At this point, there are many unknowns. “We still can’t answer whether there will be some seasonality to COVID.”
A version of this article first appeared on WebMD.com.
Solitary Pink Plaque on the Neck
The Diagnosis: Plaque-type Syringoma
A biopsy demonstrated multiple basaloid islands of tumor cells in the reticular dermis with ductal differentiation, some with a commalike tail. The ducts were lined by 2 to 3 layers of small uniform cuboidal cells without atypia and contained inspissated secretions within the lumina of scattered ducts. There was an associated fibrotic collagenous stroma. There was no evidence of perineural invasion and no deep dermal or subcutaneous extension (Figure 1). Additional cytokeratin immunohistochemical staining highlighted the adnexal proliferation (Figure 2). A diagnosis of plaque-type syringoma (PTS) was made.
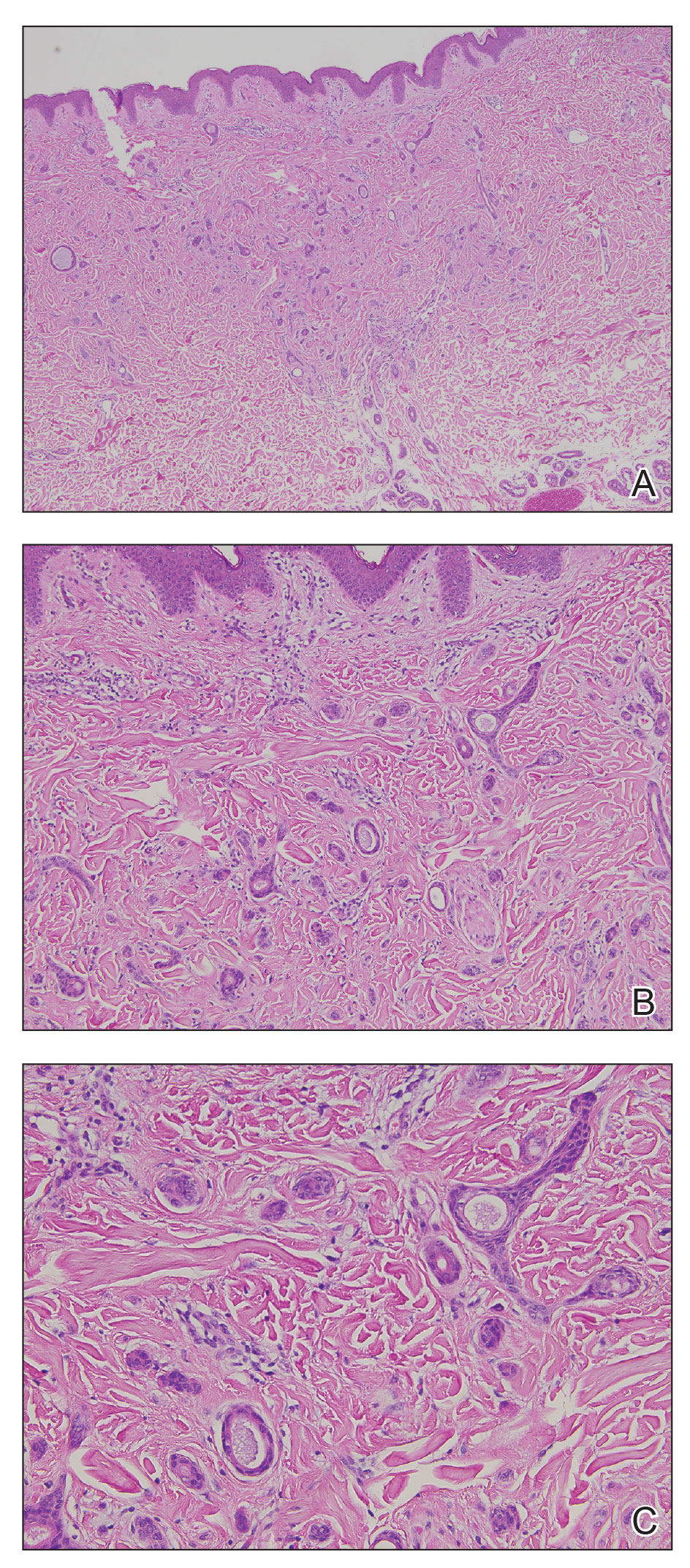
Syringomas are benign dermal sweat gland tumors that typically present as flesh-colored papules on the cheeks or periorbital area of young females. Plaque-type tumors as well as papulonodular, eruptive, disseminated, urticaria pigmentosa–like, lichen planus–like, or milialike syringomas also have been reported. Syringomas may be associated with certain medical conditions such as Down syndrome, Nicolau-Balus syndrome, and both scarring and nonscarring alopecias.1 The clear cell variant of syringoma often is associated with diabetes mellitus.2 Kikuchi et al3 first described PTS in 1979. Plaque-type syringomas rarely are reported in the literature, and sites of involvement include the head and neck region, upper lip, chest, upper extremities, vulva, penis, and scrotum.4-6
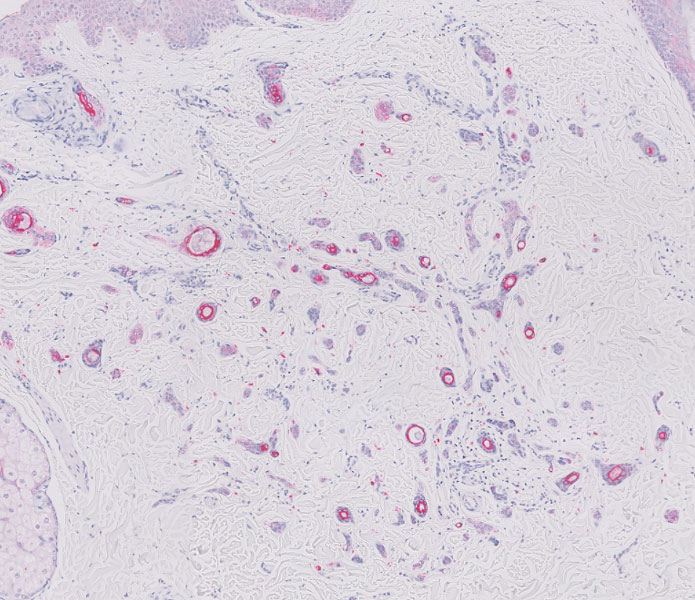
Histologically, syringomatous lesions are composed of multiple small ducts lined by 2 to 3 layers of cuboidal epithelium. The ducts may be arranged in nests or strands of basaloid cells surrounded by a dense fibrotic stroma. Occasionally, the ducts will form a comma- or teardropshaped tail; however, this also may be observed in desmoplastic trichoepithelioma (DTE).7 Perineural invasion is absent in syringomas. Syringomas exhibit a lateral growth pattern that typically is limited to the upper half of the reticular dermis and spares the underlying subcutis, muscle, and bone. The growth pattern may be discontinuous with proliferations juxtaposed by normal-appearing skin.8 Syringomas usually express progesterone receptors and are known to proliferate at puberty, suggesting that these neoplasms are under hormonal control.9 Although syringomas are benign, various treatment options that may be pursued for cosmetic purposes include radiofrequency, staged excision, laser ablation, and oral isotretinoin.8,10 If only a superficial biopsy is obtained, syringomas may display features of other adnexal neoplasms, including microcystic adnexal carcinoma (MAC), DTE, morpheaform basal cell carcinoma (BCC), and inflammatory linear verrucous epidermal nevus (ILVEN).
Microcystic adnexal carcinoma is a locally aggressive neoplasm first described by Goldstein et al11 in 1982 an indurated, ill-defined plaque or nodule on the face with a predilection for the upper and lower lip. Prior radiation therapy and immunosuppression are risk factors for the development of MAC.12 Histologically, the superficial portion displays small cornifying cysts interspersed with islands of basaloid cells and may mimic a syringoma. However, the deeper portions demonstrate ducts lined by a single layer of cells with a background of hyalinized and sclerotic stroma. The tumor cells may occupy the deep dermis and underlying subcutis, muscle, or bone and demonstrate an infiltrative growth pattern and perineural invasion. Treatment includes Mohs micrographic surgery.
Desmoplastic trichoepitheliomas most commonly present as solitary white to yellowish annular papules or plaques with a central dell located on sun-exposed areas of the face, cheeks, or chin. This benign neoplasm has a bimodal age distribution, primarily affecting females either in childhood or adulthood.13 Histologically, strands and nests of basaloid epithelial cells proliferate in a dense eosinophilic desmoplastic stroma. The basaloid islands are narrow and cordlike with growth parallel to the surface epidermis and do not dive deeply into the deep dermis or subcutis. Ductal differentiation with associated secretions typically is not seen in DTE.1 Calcifications and foreign body granulomatous infiltrates may be present. Merkel cells also are present in this tumor and may be highlighted by immunohistochemistry with cytokeratin 20.14 Rarely, desmoplastic trichoepitheliomas may transform into trichoblastic carcinomas. Treatment may consist of surgical excision or Mohs micrographic surgery.
Morpheaform BCC also is included in the clinical and histopathologic differential diagnosis of infiltrative basaloid neoplasms. It is one of the more aggressive variants of BCC. The use of immunohistochemical staining may aid in differentiating between these sclerosing adnexal neoplasms.15 For example, pleckstrin homologylike domain family A member 1 (PHLDA1) is a stem cell marker that is heavily expressed in DTE as a specific follicular bulge marker but is not present in a morpheaform BCC. This highlights the follicular nature of DTEs at the molecular level. BerEP4 is a monoclonal antibody that serves as an epithelial marker for 2 glycopolypeptides: 34 and 39 kDa. This antibody may demonstrate positivity in morpheaform BCC but does not stain cells of interest in MAC.
Inflammatory linear verrucous epidermal nevus clinically presents with erythematous and warty papules in a linear distribution following the Blaschko lines. The papules often are reported to be intensely pruritic and usually are localized to one extremity.16 Although adultonset forms of ILVEN have been described,17 it most commonly is diagnosed in young children. Histologically, ILVEN consists of psoriasiform epidermal hyperplasia with alternating areas of parakeratosis and orthokeratosis with underlying agranulosis and hypergranulosis, respectively.18 The upper dermis contains a perivascular lymphocytic infiltrate. Treatment with laser therapy and surgical excision has led to both symptomatic and clinical improvement of ILVEN.16
Plaque-type syringomas are a rare variant of syringomas that clinically may mimic other common inflammatory and neoplastic conditions. An adequate biopsy is imperative to differentiate between adnexal neoplasms, as a small superficial biopsy of a syringoma may demonstrate features observed in other malignant or locally aggressive neoplasms. In our patient, the small ducts lined by cuboidal epithelium with no cellular atypia and no deep dermal growth or perineural invasion allowed for the diagnosis of PTS. Therapeutic options were reviewed with our patient, including oral isotretinoin, laser therapy, and staged excision. Ultimately, our patient elected not to pursue treatment, and she is being monitored clinically for any changes in appearance or symptoms.
- Suwattee P, McClelland MC, Huiras EE, et al. Plaque-type syringoma: two cases misdiagnosed as microcystic adnexal carcinoma [published online November 12, 2007]. J Cutan Pathol. 2008;35:570-574.
- Furue M, Hori Y, Nakabayashi Y. Clear-cell syringoma. association with diabetes mellitus. Am J Dermatopathol. 1984;6:131-138.
- Kikuchi I, Idemori M, Okazaki M. Plaque type syringoma. J Dermatol. 1979;6:329-331.
- Kavala M, Can B, Zindanci I, et al. Vulvar pruritus caused by syringoma of the vulva. Int J Dermatol. 2008;47:831-832.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Okuda H, Tei N, Shimizu K, et al. Chondroid syringoma of the scrotum. Int J Urol. 2008;15:944-945.
- Wallace JS, Bond JS, Seidel GD, et al. An important mimicker: plaquetype syringoma mistakenly diagnosed as microcystic adnexal carcinoma. Dermatol Surg. 2014;40:810-812.
- Clark M, Duprey C, Sutton A, et al. Plaque-type syringoma masquerading as microcystic adnexal carcinoma: review of the literature and description of a novel technique that emphasizes lesion architecture to help make the diagnosis. Am J Dermatopathol. 2019;41:E98-E101.
- Wallace ML, Smoller BR. Progesterone receptor positivity supports hormonal control of syringomas. J Cutan Pathol. 1995;22:442-445.
- Mainitz M, Schmidt JB, Gebhart W. Response of multiple syringomas to isotretinoin. Acta Derm Venereol. 1986;66:51-55.
- Goldstein DJ, Barr RJ, Santa Cruz DJ. Microcystic adnexal carcinoma: a distinct clinicopathologic entity. Cancer. 1982;50:566-572.
- Pujol RM, LeBoit PE, Su WP. Microcystic adnexal carcinoma with extensive sebaceous differentiation. Am J Dermatopathol. 1997;19:358-362.
- Rahman J, Tahir M, Arekemase H, et al. Desmoplastic trichoepithelioma: histopathologic and immunohistochemical criteria for differentiation of a rare benign hair follicle tumor from other cutaneous adnexal tumors. Cureus. 2020;12:E9703.
- Abesamis-Cubillan E, El-Shabrawi-Caelen L, LeBoit PE. Merkel cells and sclerosing epithelial neoplasms. Am J Dermatopathol. 2000;22:311-315.
- Sellheyer K, Nelson P, Kutzner H, et al. The immunohistochemical differential diagnosis of microcystic adnexal carcinoma, desmoplastic trichoepithelioma and morpheaform basal cell carcinoma using BerEP4 and stem cell markers. J Cutan Pathol. 2013;40:363-370.
- Gianfaldoni S, Tchernev G, Gianfaldoni R, et al. A case of “inflammatory linear verrucous epidermal nevus” (ILVEN) treated with CO2 laser ablation. Open Access Maced J Med Sci. 2017;5:454-457.
- Kawaguchi H, Takeuchi M, Ono H, et al. Adult onset of inflammatory linear verrucous epidermal nevus [published online October 27, 1999]. J Dermatol. 1999;26:599-602.
- Patterson JW, Hosler GA, Prenshaw KL, et al. The psoriasiform reaction pattern. In: Patterson JW. Weedon’s Skin Pathology. 5th ed. Elsevier; 2021:99-120.
The Diagnosis: Plaque-type Syringoma
A biopsy demonstrated multiple basaloid islands of tumor cells in the reticular dermis with ductal differentiation, some with a commalike tail. The ducts were lined by 2 to 3 layers of small uniform cuboidal cells without atypia and contained inspissated secretions within the lumina of scattered ducts. There was an associated fibrotic collagenous stroma. There was no evidence of perineural invasion and no deep dermal or subcutaneous extension (Figure 1). Additional cytokeratin immunohistochemical staining highlighted the adnexal proliferation (Figure 2). A diagnosis of plaque-type syringoma (PTS) was made.

Syringomas are benign dermal sweat gland tumors that typically present as flesh-colored papules on the cheeks or periorbital area of young females. Plaque-type tumors as well as papulonodular, eruptive, disseminated, urticaria pigmentosa–like, lichen planus–like, or milialike syringomas also have been reported. Syringomas may be associated with certain medical conditions such as Down syndrome, Nicolau-Balus syndrome, and both scarring and nonscarring alopecias.1 The clear cell variant of syringoma often is associated with diabetes mellitus.2 Kikuchi et al3 first described PTS in 1979. Plaque-type syringomas rarely are reported in the literature, and sites of involvement include the head and neck region, upper lip, chest, upper extremities, vulva, penis, and scrotum.4-6

Histologically, syringomatous lesions are composed of multiple small ducts lined by 2 to 3 layers of cuboidal epithelium. The ducts may be arranged in nests or strands of basaloid cells surrounded by a dense fibrotic stroma. Occasionally, the ducts will form a comma- or teardropshaped tail; however, this also may be observed in desmoplastic trichoepithelioma (DTE).7 Perineural invasion is absent in syringomas. Syringomas exhibit a lateral growth pattern that typically is limited to the upper half of the reticular dermis and spares the underlying subcutis, muscle, and bone. The growth pattern may be discontinuous with proliferations juxtaposed by normal-appearing skin.8 Syringomas usually express progesterone receptors and are known to proliferate at puberty, suggesting that these neoplasms are under hormonal control.9 Although syringomas are benign, various treatment options that may be pursued for cosmetic purposes include radiofrequency, staged excision, laser ablation, and oral isotretinoin.8,10 If only a superficial biopsy is obtained, syringomas may display features of other adnexal neoplasms, including microcystic adnexal carcinoma (MAC), DTE, morpheaform basal cell carcinoma (BCC), and inflammatory linear verrucous epidermal nevus (ILVEN).
Microcystic adnexal carcinoma is a locally aggressive neoplasm first described by Goldstein et al11 in 1982 an indurated, ill-defined plaque or nodule on the face with a predilection for the upper and lower lip. Prior radiation therapy and immunosuppression are risk factors for the development of MAC.12 Histologically, the superficial portion displays small cornifying cysts interspersed with islands of basaloid cells and may mimic a syringoma. However, the deeper portions demonstrate ducts lined by a single layer of cells with a background of hyalinized and sclerotic stroma. The tumor cells may occupy the deep dermis and underlying subcutis, muscle, or bone and demonstrate an infiltrative growth pattern and perineural invasion. Treatment includes Mohs micrographic surgery.
Desmoplastic trichoepitheliomas most commonly present as solitary white to yellowish annular papules or plaques with a central dell located on sun-exposed areas of the face, cheeks, or chin. This benign neoplasm has a bimodal age distribution, primarily affecting females either in childhood or adulthood.13 Histologically, strands and nests of basaloid epithelial cells proliferate in a dense eosinophilic desmoplastic stroma. The basaloid islands are narrow and cordlike with growth parallel to the surface epidermis and do not dive deeply into the deep dermis or subcutis. Ductal differentiation with associated secretions typically is not seen in DTE.1 Calcifications and foreign body granulomatous infiltrates may be present. Merkel cells also are present in this tumor and may be highlighted by immunohistochemistry with cytokeratin 20.14 Rarely, desmoplastic trichoepitheliomas may transform into trichoblastic carcinomas. Treatment may consist of surgical excision or Mohs micrographic surgery.
Morpheaform BCC also is included in the clinical and histopathologic differential diagnosis of infiltrative basaloid neoplasms. It is one of the more aggressive variants of BCC. The use of immunohistochemical staining may aid in differentiating between these sclerosing adnexal neoplasms.15 For example, pleckstrin homologylike domain family A member 1 (PHLDA1) is a stem cell marker that is heavily expressed in DTE as a specific follicular bulge marker but is not present in a morpheaform BCC. This highlights the follicular nature of DTEs at the molecular level. BerEP4 is a monoclonal antibody that serves as an epithelial marker for 2 glycopolypeptides: 34 and 39 kDa. This antibody may demonstrate positivity in morpheaform BCC but does not stain cells of interest in MAC.
Inflammatory linear verrucous epidermal nevus clinically presents with erythematous and warty papules in a linear distribution following the Blaschko lines. The papules often are reported to be intensely pruritic and usually are localized to one extremity.16 Although adultonset forms of ILVEN have been described,17 it most commonly is diagnosed in young children. Histologically, ILVEN consists of psoriasiform epidermal hyperplasia with alternating areas of parakeratosis and orthokeratosis with underlying agranulosis and hypergranulosis, respectively.18 The upper dermis contains a perivascular lymphocytic infiltrate. Treatment with laser therapy and surgical excision has led to both symptomatic and clinical improvement of ILVEN.16
Plaque-type syringomas are a rare variant of syringomas that clinically may mimic other common inflammatory and neoplastic conditions. An adequate biopsy is imperative to differentiate between adnexal neoplasms, as a small superficial biopsy of a syringoma may demonstrate features observed in other malignant or locally aggressive neoplasms. In our patient, the small ducts lined by cuboidal epithelium with no cellular atypia and no deep dermal growth or perineural invasion allowed for the diagnosis of PTS. Therapeutic options were reviewed with our patient, including oral isotretinoin, laser therapy, and staged excision. Ultimately, our patient elected not to pursue treatment, and she is being monitored clinically for any changes in appearance or symptoms.
The Diagnosis: Plaque-type Syringoma
A biopsy demonstrated multiple basaloid islands of tumor cells in the reticular dermis with ductal differentiation, some with a commalike tail. The ducts were lined by 2 to 3 layers of small uniform cuboidal cells without atypia and contained inspissated secretions within the lumina of scattered ducts. There was an associated fibrotic collagenous stroma. There was no evidence of perineural invasion and no deep dermal or subcutaneous extension (Figure 1). Additional cytokeratin immunohistochemical staining highlighted the adnexal proliferation (Figure 2). A diagnosis of plaque-type syringoma (PTS) was made.

Syringomas are benign dermal sweat gland tumors that typically present as flesh-colored papules on the cheeks or periorbital area of young females. Plaque-type tumors as well as papulonodular, eruptive, disseminated, urticaria pigmentosa–like, lichen planus–like, or milialike syringomas also have been reported. Syringomas may be associated with certain medical conditions such as Down syndrome, Nicolau-Balus syndrome, and both scarring and nonscarring alopecias.1 The clear cell variant of syringoma often is associated with diabetes mellitus.2 Kikuchi et al3 first described PTS in 1979. Plaque-type syringomas rarely are reported in the literature, and sites of involvement include the head and neck region, upper lip, chest, upper extremities, vulva, penis, and scrotum.4-6

Histologically, syringomatous lesions are composed of multiple small ducts lined by 2 to 3 layers of cuboidal epithelium. The ducts may be arranged in nests or strands of basaloid cells surrounded by a dense fibrotic stroma. Occasionally, the ducts will form a comma- or teardropshaped tail; however, this also may be observed in desmoplastic trichoepithelioma (DTE).7 Perineural invasion is absent in syringomas. Syringomas exhibit a lateral growth pattern that typically is limited to the upper half of the reticular dermis and spares the underlying subcutis, muscle, and bone. The growth pattern may be discontinuous with proliferations juxtaposed by normal-appearing skin.8 Syringomas usually express progesterone receptors and are known to proliferate at puberty, suggesting that these neoplasms are under hormonal control.9 Although syringomas are benign, various treatment options that may be pursued for cosmetic purposes include radiofrequency, staged excision, laser ablation, and oral isotretinoin.8,10 If only a superficial biopsy is obtained, syringomas may display features of other adnexal neoplasms, including microcystic adnexal carcinoma (MAC), DTE, morpheaform basal cell carcinoma (BCC), and inflammatory linear verrucous epidermal nevus (ILVEN).
Microcystic adnexal carcinoma is a locally aggressive neoplasm first described by Goldstein et al11 in 1982 an indurated, ill-defined plaque or nodule on the face with a predilection for the upper and lower lip. Prior radiation therapy and immunosuppression are risk factors for the development of MAC.12 Histologically, the superficial portion displays small cornifying cysts interspersed with islands of basaloid cells and may mimic a syringoma. However, the deeper portions demonstrate ducts lined by a single layer of cells with a background of hyalinized and sclerotic stroma. The tumor cells may occupy the deep dermis and underlying subcutis, muscle, or bone and demonstrate an infiltrative growth pattern and perineural invasion. Treatment includes Mohs micrographic surgery.
Desmoplastic trichoepitheliomas most commonly present as solitary white to yellowish annular papules or plaques with a central dell located on sun-exposed areas of the face, cheeks, or chin. This benign neoplasm has a bimodal age distribution, primarily affecting females either in childhood or adulthood.13 Histologically, strands and nests of basaloid epithelial cells proliferate in a dense eosinophilic desmoplastic stroma. The basaloid islands are narrow and cordlike with growth parallel to the surface epidermis and do not dive deeply into the deep dermis or subcutis. Ductal differentiation with associated secretions typically is not seen in DTE.1 Calcifications and foreign body granulomatous infiltrates may be present. Merkel cells also are present in this tumor and may be highlighted by immunohistochemistry with cytokeratin 20.14 Rarely, desmoplastic trichoepitheliomas may transform into trichoblastic carcinomas. Treatment may consist of surgical excision or Mohs micrographic surgery.
Morpheaform BCC also is included in the clinical and histopathologic differential diagnosis of infiltrative basaloid neoplasms. It is one of the more aggressive variants of BCC. The use of immunohistochemical staining may aid in differentiating between these sclerosing adnexal neoplasms.15 For example, pleckstrin homologylike domain family A member 1 (PHLDA1) is a stem cell marker that is heavily expressed in DTE as a specific follicular bulge marker but is not present in a morpheaform BCC. This highlights the follicular nature of DTEs at the molecular level. BerEP4 is a monoclonal antibody that serves as an epithelial marker for 2 glycopolypeptides: 34 and 39 kDa. This antibody may demonstrate positivity in morpheaform BCC but does not stain cells of interest in MAC.
Inflammatory linear verrucous epidermal nevus clinically presents with erythematous and warty papules in a linear distribution following the Blaschko lines. The papules often are reported to be intensely pruritic and usually are localized to one extremity.16 Although adultonset forms of ILVEN have been described,17 it most commonly is diagnosed in young children. Histologically, ILVEN consists of psoriasiform epidermal hyperplasia with alternating areas of parakeratosis and orthokeratosis with underlying agranulosis and hypergranulosis, respectively.18 The upper dermis contains a perivascular lymphocytic infiltrate. Treatment with laser therapy and surgical excision has led to both symptomatic and clinical improvement of ILVEN.16
Plaque-type syringomas are a rare variant of syringomas that clinically may mimic other common inflammatory and neoplastic conditions. An adequate biopsy is imperative to differentiate between adnexal neoplasms, as a small superficial biopsy of a syringoma may demonstrate features observed in other malignant or locally aggressive neoplasms. In our patient, the small ducts lined by cuboidal epithelium with no cellular atypia and no deep dermal growth or perineural invasion allowed for the diagnosis of PTS. Therapeutic options were reviewed with our patient, including oral isotretinoin, laser therapy, and staged excision. Ultimately, our patient elected not to pursue treatment, and she is being monitored clinically for any changes in appearance or symptoms.
- Suwattee P, McClelland MC, Huiras EE, et al. Plaque-type syringoma: two cases misdiagnosed as microcystic adnexal carcinoma [published online November 12, 2007]. J Cutan Pathol. 2008;35:570-574.
- Furue M, Hori Y, Nakabayashi Y. Clear-cell syringoma. association with diabetes mellitus. Am J Dermatopathol. 1984;6:131-138.
- Kikuchi I, Idemori M, Okazaki M. Plaque type syringoma. J Dermatol. 1979;6:329-331.
- Kavala M, Can B, Zindanci I, et al. Vulvar pruritus caused by syringoma of the vulva. Int J Dermatol. 2008;47:831-832.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Okuda H, Tei N, Shimizu K, et al. Chondroid syringoma of the scrotum. Int J Urol. 2008;15:944-945.
- Wallace JS, Bond JS, Seidel GD, et al. An important mimicker: plaquetype syringoma mistakenly diagnosed as microcystic adnexal carcinoma. Dermatol Surg. 2014;40:810-812.
- Clark M, Duprey C, Sutton A, et al. Plaque-type syringoma masquerading as microcystic adnexal carcinoma: review of the literature and description of a novel technique that emphasizes lesion architecture to help make the diagnosis. Am J Dermatopathol. 2019;41:E98-E101.
- Wallace ML, Smoller BR. Progesterone receptor positivity supports hormonal control of syringomas. J Cutan Pathol. 1995;22:442-445.
- Mainitz M, Schmidt JB, Gebhart W. Response of multiple syringomas to isotretinoin. Acta Derm Venereol. 1986;66:51-55.
- Goldstein DJ, Barr RJ, Santa Cruz DJ. Microcystic adnexal carcinoma: a distinct clinicopathologic entity. Cancer. 1982;50:566-572.
- Pujol RM, LeBoit PE, Su WP. Microcystic adnexal carcinoma with extensive sebaceous differentiation. Am J Dermatopathol. 1997;19:358-362.
- Rahman J, Tahir M, Arekemase H, et al. Desmoplastic trichoepithelioma: histopathologic and immunohistochemical criteria for differentiation of a rare benign hair follicle tumor from other cutaneous adnexal tumors. Cureus. 2020;12:E9703.
- Abesamis-Cubillan E, El-Shabrawi-Caelen L, LeBoit PE. Merkel cells and sclerosing epithelial neoplasms. Am J Dermatopathol. 2000;22:311-315.
- Sellheyer K, Nelson P, Kutzner H, et al. The immunohistochemical differential diagnosis of microcystic adnexal carcinoma, desmoplastic trichoepithelioma and morpheaform basal cell carcinoma using BerEP4 and stem cell markers. J Cutan Pathol. 2013;40:363-370.
- Gianfaldoni S, Tchernev G, Gianfaldoni R, et al. A case of “inflammatory linear verrucous epidermal nevus” (ILVEN) treated with CO2 laser ablation. Open Access Maced J Med Sci. 2017;5:454-457.
- Kawaguchi H, Takeuchi M, Ono H, et al. Adult onset of inflammatory linear verrucous epidermal nevus [published online October 27, 1999]. J Dermatol. 1999;26:599-602.
- Patterson JW, Hosler GA, Prenshaw KL, et al. The psoriasiform reaction pattern. In: Patterson JW. Weedon’s Skin Pathology. 5th ed. Elsevier; 2021:99-120.
- Suwattee P, McClelland MC, Huiras EE, et al. Plaque-type syringoma: two cases misdiagnosed as microcystic adnexal carcinoma [published online November 12, 2007]. J Cutan Pathol. 2008;35:570-574.
- Furue M, Hori Y, Nakabayashi Y. Clear-cell syringoma. association with diabetes mellitus. Am J Dermatopathol. 1984;6:131-138.
- Kikuchi I, Idemori M, Okazaki M. Plaque type syringoma. J Dermatol. 1979;6:329-331.
- Kavala M, Can B, Zindanci I, et al. Vulvar pruritus caused by syringoma of the vulva. Int J Dermatol. 2008;47:831-832.
- Cohen PR, Tschen JA, Rapini RP. Penile syringoma: reports and review of patients with syringoma located on the penis. J Clin Aesthet Dermatol. 2013;6:38-42.
- Okuda H, Tei N, Shimizu K, et al. Chondroid syringoma of the scrotum. Int J Urol. 2008;15:944-945.
- Wallace JS, Bond JS, Seidel GD, et al. An important mimicker: plaquetype syringoma mistakenly diagnosed as microcystic adnexal carcinoma. Dermatol Surg. 2014;40:810-812.
- Clark M, Duprey C, Sutton A, et al. Plaque-type syringoma masquerading as microcystic adnexal carcinoma: review of the literature and description of a novel technique that emphasizes lesion architecture to help make the diagnosis. Am J Dermatopathol. 2019;41:E98-E101.
- Wallace ML, Smoller BR. Progesterone receptor positivity supports hormonal control of syringomas. J Cutan Pathol. 1995;22:442-445.
- Mainitz M, Schmidt JB, Gebhart W. Response of multiple syringomas to isotretinoin. Acta Derm Venereol. 1986;66:51-55.
- Goldstein DJ, Barr RJ, Santa Cruz DJ. Microcystic adnexal carcinoma: a distinct clinicopathologic entity. Cancer. 1982;50:566-572.
- Pujol RM, LeBoit PE, Su WP. Microcystic adnexal carcinoma with extensive sebaceous differentiation. Am J Dermatopathol. 1997;19:358-362.
- Rahman J, Tahir M, Arekemase H, et al. Desmoplastic trichoepithelioma: histopathologic and immunohistochemical criteria for differentiation of a rare benign hair follicle tumor from other cutaneous adnexal tumors. Cureus. 2020;12:E9703.
- Abesamis-Cubillan E, El-Shabrawi-Caelen L, LeBoit PE. Merkel cells and sclerosing epithelial neoplasms. Am J Dermatopathol. 2000;22:311-315.
- Sellheyer K, Nelson P, Kutzner H, et al. The immunohistochemical differential diagnosis of microcystic adnexal carcinoma, desmoplastic trichoepithelioma and morpheaform basal cell carcinoma using BerEP4 and stem cell markers. J Cutan Pathol. 2013;40:363-370.
- Gianfaldoni S, Tchernev G, Gianfaldoni R, et al. A case of “inflammatory linear verrucous epidermal nevus” (ILVEN) treated with CO2 laser ablation. Open Access Maced J Med Sci. 2017;5:454-457.
- Kawaguchi H, Takeuchi M, Ono H, et al. Adult onset of inflammatory linear verrucous epidermal nevus [published online October 27, 1999]. J Dermatol. 1999;26:599-602.
- Patterson JW, Hosler GA, Prenshaw KL, et al. The psoriasiform reaction pattern. In: Patterson JW. Weedon’s Skin Pathology. 5th ed. Elsevier; 2021:99-120.
A 17-year-old adolescent girl presented with a solitary, 8-cm, pink plaque on the anterior aspect of the neck of 5 years’ duration. No similar skin findings were present elsewhere on the body. The rash was not painful or pruritic, and she denied prior trauma to the site. The patient previously had tried a salicylic acid bodywash as well as mupirocin cream 2% and mometasone ointment with no improvement. Her medical history was unremarkable, and she had no known allergies. There was no family history of a similar rash. Physical examination revealed no palpable subcutaneous lumps or masses and no lymphadenopathy of the head or neck. An incisional biopsy was performed.
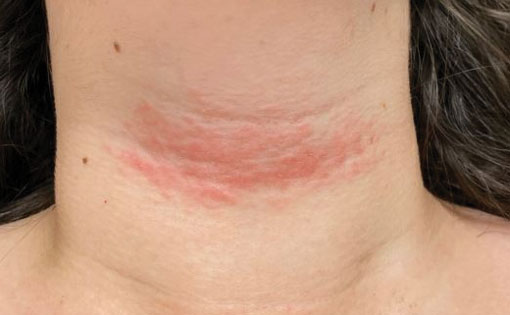
Ustekinumab becomes second biologic approved for PsA in kids
The Food and Drug Administration has approved the dual interleukin-12 and IL-23 inhibitor ustekinumab (Stelara) for the treatment of juvenile psoriatic arthritis (jPsA) in patients aged 6 years and older, according to an Aug. 1 announcement from its manufacturer, Janssen.
The approval makes jPsA the sixth approved indication for ustekinumab, which include active psoriatic arthritis in adults, moderate to severe plaque psoriasis in both adults and children aged 6 years or older who are candidates for phototherapy or systemic therapy, moderately to severely active Crohn’s disease in adults, and moderately to severely active ulcerative colitis in adults.
In addition, ustekinumab is now the second biologic to be approved for jPsA, following the agency’s December 2021 approval of secukinumab (Cosentyx) to treat jPsA in children and adolescents aged 2 years and older as well as enthesitis-related arthritis in children and adolescents aged 4 years and older.
In pediatric patients, ustekinumab is administered as a subcutaneous injection dosed four times per year after two starter doses.
Ustekinumab’s approval is based on “an extrapolation of the established data and existing safety profile” of ustekinumab in multiple phase 3 studies in adult and pediatric patients with moderate to severe plaque psoriasis and adult patients with active PsA, according to Janssen.
“With the limited availability of pediatric patients for clinical trial inclusion, researchers can extrapolate data from trials with adults to determine the potential efficacy and tolerability of a treatment for a pediatric population,” according to the October 2021 announcement from the company that the Biologics License Application had been submitted to the FDA.
Juvenile arthritis occurs in an estimated 20-45 children per 100,000 in the United States, with about 5% of those children having jPsA, according to the National Psoriasis Foundation.
The prescribing information for ustekinumab includes specific warnings and areas of concern. The drug should not be administered to individuals with known hypersensitivity to ustekinumab. The drug may lower the ability of the immune system to fight infections and may increase risk of infections, sometimes serious, and a test for tuberculosis infection should be given before administration.
Patients taking ustekinumab should not be given a live vaccine, and their doctors should be informed if anyone in their household needs a live vaccine. They also should not receive the BCG vaccine during the 1 year before receiving the drug or 1 year after they stop taking it, according to Johnson & Johnson.
The most common adverse effects include nasal congestion, sore throat, runny nose, upper respiratory infections, fever, headache, tiredness, itching, nausea and vomiting, redness at the injection site, vaginal yeast infections, urinary tract infections, sinus infection, bronchitis, diarrhea, stomach pain, and joint pain.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved the dual interleukin-12 and IL-23 inhibitor ustekinumab (Stelara) for the treatment of juvenile psoriatic arthritis (jPsA) in patients aged 6 years and older, according to an Aug. 1 announcement from its manufacturer, Janssen.
The approval makes jPsA the sixth approved indication for ustekinumab, which include active psoriatic arthritis in adults, moderate to severe plaque psoriasis in both adults and children aged 6 years or older who are candidates for phototherapy or systemic therapy, moderately to severely active Crohn’s disease in adults, and moderately to severely active ulcerative colitis in adults.
In addition, ustekinumab is now the second biologic to be approved for jPsA, following the agency’s December 2021 approval of secukinumab (Cosentyx) to treat jPsA in children and adolescents aged 2 years and older as well as enthesitis-related arthritis in children and adolescents aged 4 years and older.
In pediatric patients, ustekinumab is administered as a subcutaneous injection dosed four times per year after two starter doses.
Ustekinumab’s approval is based on “an extrapolation of the established data and existing safety profile” of ustekinumab in multiple phase 3 studies in adult and pediatric patients with moderate to severe plaque psoriasis and adult patients with active PsA, according to Janssen.
“With the limited availability of pediatric patients for clinical trial inclusion, researchers can extrapolate data from trials with adults to determine the potential efficacy and tolerability of a treatment for a pediatric population,” according to the October 2021 announcement from the company that the Biologics License Application had been submitted to the FDA.
Juvenile arthritis occurs in an estimated 20-45 children per 100,000 in the United States, with about 5% of those children having jPsA, according to the National Psoriasis Foundation.
The prescribing information for ustekinumab includes specific warnings and areas of concern. The drug should not be administered to individuals with known hypersensitivity to ustekinumab. The drug may lower the ability of the immune system to fight infections and may increase risk of infections, sometimes serious, and a test for tuberculosis infection should be given before administration.
Patients taking ustekinumab should not be given a live vaccine, and their doctors should be informed if anyone in their household needs a live vaccine. They also should not receive the BCG vaccine during the 1 year before receiving the drug or 1 year after they stop taking it, according to Johnson & Johnson.
The most common adverse effects include nasal congestion, sore throat, runny nose, upper respiratory infections, fever, headache, tiredness, itching, nausea and vomiting, redness at the injection site, vaginal yeast infections, urinary tract infections, sinus infection, bronchitis, diarrhea, stomach pain, and joint pain.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved the dual interleukin-12 and IL-23 inhibitor ustekinumab (Stelara) for the treatment of juvenile psoriatic arthritis (jPsA) in patients aged 6 years and older, according to an Aug. 1 announcement from its manufacturer, Janssen.
The approval makes jPsA the sixth approved indication for ustekinumab, which include active psoriatic arthritis in adults, moderate to severe plaque psoriasis in both adults and children aged 6 years or older who are candidates for phototherapy or systemic therapy, moderately to severely active Crohn’s disease in adults, and moderately to severely active ulcerative colitis in adults.
In addition, ustekinumab is now the second biologic to be approved for jPsA, following the agency’s December 2021 approval of secukinumab (Cosentyx) to treat jPsA in children and adolescents aged 2 years and older as well as enthesitis-related arthritis in children and adolescents aged 4 years and older.
In pediatric patients, ustekinumab is administered as a subcutaneous injection dosed four times per year after two starter doses.
Ustekinumab’s approval is based on “an extrapolation of the established data and existing safety profile” of ustekinumab in multiple phase 3 studies in adult and pediatric patients with moderate to severe plaque psoriasis and adult patients with active PsA, according to Janssen.
“With the limited availability of pediatric patients for clinical trial inclusion, researchers can extrapolate data from trials with adults to determine the potential efficacy and tolerability of a treatment for a pediatric population,” according to the October 2021 announcement from the company that the Biologics License Application had been submitted to the FDA.
Juvenile arthritis occurs in an estimated 20-45 children per 100,000 in the United States, with about 5% of those children having jPsA, according to the National Psoriasis Foundation.
The prescribing information for ustekinumab includes specific warnings and areas of concern. The drug should not be administered to individuals with known hypersensitivity to ustekinumab. The drug may lower the ability of the immune system to fight infections and may increase risk of infections, sometimes serious, and a test for tuberculosis infection should be given before administration.
Patients taking ustekinumab should not be given a live vaccine, and their doctors should be informed if anyone in their household needs a live vaccine. They also should not receive the BCG vaccine during the 1 year before receiving the drug or 1 year after they stop taking it, according to Johnson & Johnson.
The most common adverse effects include nasal congestion, sore throat, runny nose, upper respiratory infections, fever, headache, tiredness, itching, nausea and vomiting, redness at the injection site, vaginal yeast infections, urinary tract infections, sinus infection, bronchitis, diarrhea, stomach pain, and joint pain.
A version of this article first appeared on Medscape.com.
Children and COVID: Weekly cases top 95,000, admissions continue to rise
New pediatric COVID-19 cases increased for the third straight week as a substantial number of children under age 5 years started to receive their second doses of the vaccine.
Despite the 3-week trend, however, there are some positive signs. The new-case count for the latest reporting week (July 22-28) was over 95,000, but the 3.9% increase over the previous week’s 92,000 cases is much smaller than that week’s (July 15-21) corresponding jump of almost 22% over the July 8-14 total (75,000), according to the American Academy of Pediatrics and the Children’s Hospital Association.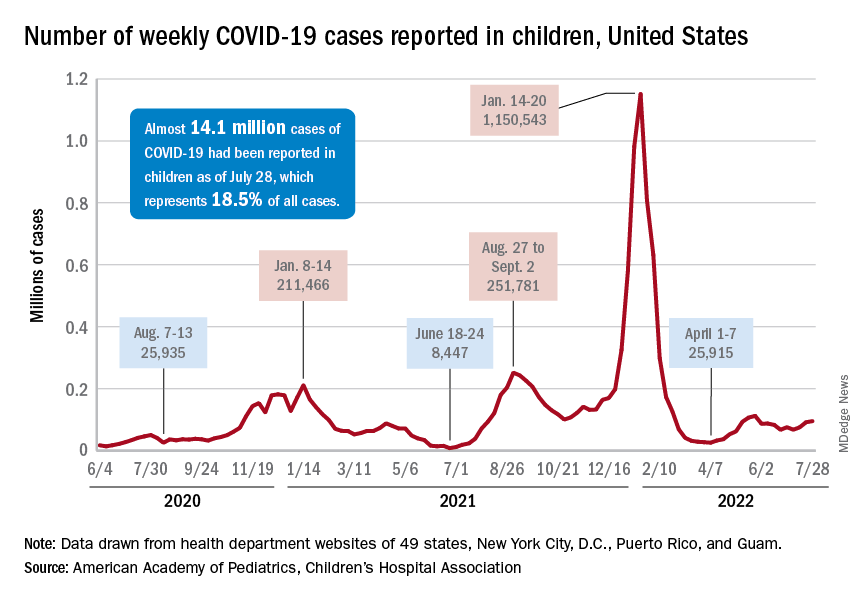
On the not-so-positive side is the trend in admissions among children aged 0-17 years, which continue to climb steadily and have nearly equaled the highest rate seen during the Delta surge in 2021. The rate on July 29 was 0.46 admissions per 100,000 population, and the highest rate over the course of the Delta surge was 0.47 per 100,000, but the all-time high from the Omicron surge – 1.25 per 100,000 in mid-January – is still a long way off, based on data from the Centers for Disease Control and Prevention.
A similar situation is occurring with emergency department visits, but there is differentiation by age group. Among those aged 0-11 years, visits with diagnosed COVID made up 6.5% of all their ED visits on July 25, which was well above the high (4.0%) during the Delta surge, the CDC said.
That is not the case, however, for the older children, for whom rates are rising more slowly. Those aged 12-15 have reached 3.4% so far this summer, as have the 16- to 17-years-olds, versus Delta highs last year of around 7%, the CDC said on its COVID Data Tracker. As with admissions, though, current rates are well below the all-time Omicron high points, the CDC data show.
Joining the ranks of the fully vaccinated
Over the last 2 weeks, the first children to receive the COVID vaccine after its approval for those under age 5 years have been coming back for their second doses. Almost 50,000, about 0.3% of all those in that age group, had done so by July 27. Just over 662,000, about 3.4% of the total under-5 population, have received at least one dose, the CDC said.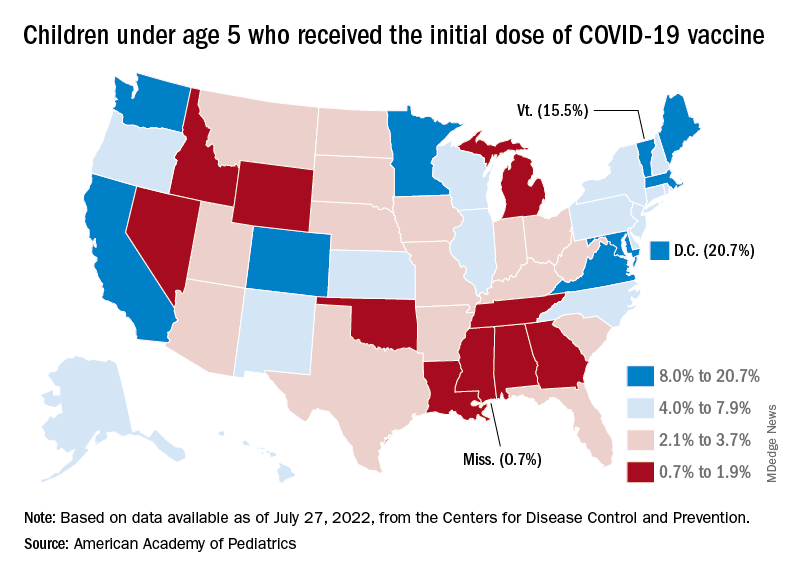
Meanwhile, analysis of “data from the first several weeks following availability of the vaccine in this age group indicate high variability across states,” the AAP said in its weekly vaccination report. In the District of Columbia, 20.7% of all children under age 5 have received an initial dose as of July 27, as have 15.5% of those in Vermont and 12.5% in Massachusetts. No other state was above 10%, but Mississippi, at 0.7%, was the only one below 1%.
The older children, obviously, have a head start, so their numbers are much higher. At the state level, Vermont has the highest initial dose rate, 69%, for those aged 5-11 years, while Alabama, Mississippi, and Wyoming, at 17%, are looking up at everyone else in the country. Among children aged 12-17 years, D.C. is the highest with 100% vaccination – Massachusetts and Rhode Island are at 98% – and Wyoming is the lowest with 40%, the AAP said.
New pediatric COVID-19 cases increased for the third straight week as a substantial number of children under age 5 years started to receive their second doses of the vaccine.
Despite the 3-week trend, however, there are some positive signs. The new-case count for the latest reporting week (July 22-28) was over 95,000, but the 3.9% increase over the previous week’s 92,000 cases is much smaller than that week’s (July 15-21) corresponding jump of almost 22% over the July 8-14 total (75,000), according to the American Academy of Pediatrics and the Children’s Hospital Association.
On the not-so-positive side is the trend in admissions among children aged 0-17 years, which continue to climb steadily and have nearly equaled the highest rate seen during the Delta surge in 2021. The rate on July 29 was 0.46 admissions per 100,000 population, and the highest rate over the course of the Delta surge was 0.47 per 100,000, but the all-time high from the Omicron surge – 1.25 per 100,000 in mid-January – is still a long way off, based on data from the Centers for Disease Control and Prevention.
A similar situation is occurring with emergency department visits, but there is differentiation by age group. Among those aged 0-11 years, visits with diagnosed COVID made up 6.5% of all their ED visits on July 25, which was well above the high (4.0%) during the Delta surge, the CDC said.
That is not the case, however, for the older children, for whom rates are rising more slowly. Those aged 12-15 have reached 3.4% so far this summer, as have the 16- to 17-years-olds, versus Delta highs last year of around 7%, the CDC said on its COVID Data Tracker. As with admissions, though, current rates are well below the all-time Omicron high points, the CDC data show.
Joining the ranks of the fully vaccinated
Over the last 2 weeks, the first children to receive the COVID vaccine after its approval for those under age 5 years have been coming back for their second doses. Almost 50,000, about 0.3% of all those in that age group, had done so by July 27. Just over 662,000, about 3.4% of the total under-5 population, have received at least one dose, the CDC said.
Meanwhile, analysis of “data from the first several weeks following availability of the vaccine in this age group indicate high variability across states,” the AAP said in its weekly vaccination report. In the District of Columbia, 20.7% of all children under age 5 have received an initial dose as of July 27, as have 15.5% of those in Vermont and 12.5% in Massachusetts. No other state was above 10%, but Mississippi, at 0.7%, was the only one below 1%.
The older children, obviously, have a head start, so their numbers are much higher. At the state level, Vermont has the highest initial dose rate, 69%, for those aged 5-11 years, while Alabama, Mississippi, and Wyoming, at 17%, are looking up at everyone else in the country. Among children aged 12-17 years, D.C. is the highest with 100% vaccination – Massachusetts and Rhode Island are at 98% – and Wyoming is the lowest with 40%, the AAP said.
New pediatric COVID-19 cases increased for the third straight week as a substantial number of children under age 5 years started to receive their second doses of the vaccine.
Despite the 3-week trend, however, there are some positive signs. The new-case count for the latest reporting week (July 22-28) was over 95,000, but the 3.9% increase over the previous week’s 92,000 cases is much smaller than that week’s (July 15-21) corresponding jump of almost 22% over the July 8-14 total (75,000), according to the American Academy of Pediatrics and the Children’s Hospital Association.
On the not-so-positive side is the trend in admissions among children aged 0-17 years, which continue to climb steadily and have nearly equaled the highest rate seen during the Delta surge in 2021. The rate on July 29 was 0.46 admissions per 100,000 population, and the highest rate over the course of the Delta surge was 0.47 per 100,000, but the all-time high from the Omicron surge – 1.25 per 100,000 in mid-January – is still a long way off, based on data from the Centers for Disease Control and Prevention.
A similar situation is occurring with emergency department visits, but there is differentiation by age group. Among those aged 0-11 years, visits with diagnosed COVID made up 6.5% of all their ED visits on July 25, which was well above the high (4.0%) during the Delta surge, the CDC said.
That is not the case, however, for the older children, for whom rates are rising more slowly. Those aged 12-15 have reached 3.4% so far this summer, as have the 16- to 17-years-olds, versus Delta highs last year of around 7%, the CDC said on its COVID Data Tracker. As with admissions, though, current rates are well below the all-time Omicron high points, the CDC data show.
Joining the ranks of the fully vaccinated
Over the last 2 weeks, the first children to receive the COVID vaccine after its approval for those under age 5 years have been coming back for their second doses. Almost 50,000, about 0.3% of all those in that age group, had done so by July 27. Just over 662,000, about 3.4% of the total under-5 population, have received at least one dose, the CDC said.
Meanwhile, analysis of “data from the first several weeks following availability of the vaccine in this age group indicate high variability across states,” the AAP said in its weekly vaccination report. In the District of Columbia, 20.7% of all children under age 5 have received an initial dose as of July 27, as have 15.5% of those in Vermont and 12.5% in Massachusetts. No other state was above 10%, but Mississippi, at 0.7%, was the only one below 1%.
The older children, obviously, have a head start, so their numbers are much higher. At the state level, Vermont has the highest initial dose rate, 69%, for those aged 5-11 years, while Alabama, Mississippi, and Wyoming, at 17%, are looking up at everyone else in the country. Among children aged 12-17 years, D.C. is the highest with 100% vaccination – Massachusetts and Rhode Island are at 98% – and Wyoming is the lowest with 40%, the AAP said.