User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Med students dismayed that residency match process won’t change
– mostly medical students, residents, and fellows – who supported the change.
The program’s decision comes after nearly 3 months of feedback from the public, medical students, and education community. Although about 60% of public respondents believed the change could reduce stress and allow students more time for momentous career decisions, the program’s board of directors decided the disadvantages were “of greater consequence,” according to a Oct. 28 statement.
Those disadvantages included introducing application or interview behaviors that could increase students’ stress; potentially identifying partially matched or unmatched applicants, which could lead to bias; and extending the match process time for those applicants.
In addition, members of 12 medical education and student organizations raised other concerns, such as the proposed change not addressing high application numbers, according to the statement. NRMP has reported record numbers of applicants over the past few years, typically with more applicants than available program slots.
“While the testimony gave nod to the positive aspects of the proposal ... there was substantially more concern voiced about the potential negative consequences identified in the public comments,” NRMP President and CEO Donna L. Lamb, DHSc, MBA, BSN, told this news organization. Some of those issues could not be addressed without further study, so the board decided not to proceed with the proposal, she explained.
The proposal would have separated the Main Residency Match into two phases and replaced the Supplemental Offer and Acceptance Program (SOAP), in which unmatched or partially matched applicants apply for unfilled residency positions. Under the proposed change, each phase would have operated the same way, from rank order lists and using a matching algorithm to a pair of Match Days instead of a single day.
The two-phase process would have given students who didn’t match more time to carefully weigh residency programs – they can apply to up to 45 placements as part of SOAP – that will guide their career path for the next few years, PGY-1 intern Asim A., who asked not to be identified further, told this news organization. The alternative is a hasty decision once students learn which residency spots are available, he added. “Applicants would have breathing room to make a more informed decision.”
Asim, who is Canadian, said he is participating in a transitional year in internal medicine in the hopes of being matched into internal medicine or psychiatry. He said Canada’s two-phase match is a “lot less stressful” than the U.S. system.
Meanwhile, students on Reddit’s medical school community also questioned NRMP’s decision.
“A significant majority of those surveyed thought it would be beneficial. But NRMP decides to not go through with it,” one Reddit user wrote. Another posted, “The one thing that could have improved the match and they chose not to do it.”
Others supported the decision to retain a 1-day match.
“I think this was the right call,” Bryan Carmody, MD, an outspoken medical education blogger, tweeted after learning of NRMP’s decision. Dr. Carmody, a pediatric nephrologist, previously expressed to this news organization misgivings about whether the two-phase match would make it difficult for programs to thoroughly review candidates and vice versa. He was concerned that it would compress the interview season and pressure programs to rapidly review applicants and conduct interviews.
More than 8,000 people responded to the public survey that began in August and ran for a month. Nearly two-thirds of the respondents (60%) were students, residents, or fellows. About 25% included faculty, program directors, and staff. Among the survey findings, respondents were equally divided between whether the two-phase match would be modestly advantageous (30%) or significantly advantageous (30%) compared to 20% who viewed it as modestly or significantly disadvantageous.
The NRMP said it would continue engaging with the community through focus groups and other means to improve the match experience and transition to residency.
“It is important to remember that a proposal is just that,” Dr. Lamb told this news orgnization, “an opportunity to discuss the pros and cons of an idea or framework ... and to mitigate unwanted consequences determined to be detrimental to learners and programs.”
The NRMP will involve the community in future discussions “to continue to give learners a voice,” she said.
A version of this article first appeared on Medscape.com.
– mostly medical students, residents, and fellows – who supported the change.
The program’s decision comes after nearly 3 months of feedback from the public, medical students, and education community. Although about 60% of public respondents believed the change could reduce stress and allow students more time for momentous career decisions, the program’s board of directors decided the disadvantages were “of greater consequence,” according to a Oct. 28 statement.
Those disadvantages included introducing application or interview behaviors that could increase students’ stress; potentially identifying partially matched or unmatched applicants, which could lead to bias; and extending the match process time for those applicants.
In addition, members of 12 medical education and student organizations raised other concerns, such as the proposed change not addressing high application numbers, according to the statement. NRMP has reported record numbers of applicants over the past few years, typically with more applicants than available program slots.
“While the testimony gave nod to the positive aspects of the proposal ... there was substantially more concern voiced about the potential negative consequences identified in the public comments,” NRMP President and CEO Donna L. Lamb, DHSc, MBA, BSN, told this news organization. Some of those issues could not be addressed without further study, so the board decided not to proceed with the proposal, she explained.
The proposal would have separated the Main Residency Match into two phases and replaced the Supplemental Offer and Acceptance Program (SOAP), in which unmatched or partially matched applicants apply for unfilled residency positions. Under the proposed change, each phase would have operated the same way, from rank order lists and using a matching algorithm to a pair of Match Days instead of a single day.
The two-phase process would have given students who didn’t match more time to carefully weigh residency programs – they can apply to up to 45 placements as part of SOAP – that will guide their career path for the next few years, PGY-1 intern Asim A., who asked not to be identified further, told this news organization. The alternative is a hasty decision once students learn which residency spots are available, he added. “Applicants would have breathing room to make a more informed decision.”
Asim, who is Canadian, said he is participating in a transitional year in internal medicine in the hopes of being matched into internal medicine or psychiatry. He said Canada’s two-phase match is a “lot less stressful” than the U.S. system.
Meanwhile, students on Reddit’s medical school community also questioned NRMP’s decision.
“A significant majority of those surveyed thought it would be beneficial. But NRMP decides to not go through with it,” one Reddit user wrote. Another posted, “The one thing that could have improved the match and they chose not to do it.”
Others supported the decision to retain a 1-day match.
“I think this was the right call,” Bryan Carmody, MD, an outspoken medical education blogger, tweeted after learning of NRMP’s decision. Dr. Carmody, a pediatric nephrologist, previously expressed to this news organization misgivings about whether the two-phase match would make it difficult for programs to thoroughly review candidates and vice versa. He was concerned that it would compress the interview season and pressure programs to rapidly review applicants and conduct interviews.
More than 8,000 people responded to the public survey that began in August and ran for a month. Nearly two-thirds of the respondents (60%) were students, residents, or fellows. About 25% included faculty, program directors, and staff. Among the survey findings, respondents were equally divided between whether the two-phase match would be modestly advantageous (30%) or significantly advantageous (30%) compared to 20% who viewed it as modestly or significantly disadvantageous.
The NRMP said it would continue engaging with the community through focus groups and other means to improve the match experience and transition to residency.
“It is important to remember that a proposal is just that,” Dr. Lamb told this news orgnization, “an opportunity to discuss the pros and cons of an idea or framework ... and to mitigate unwanted consequences determined to be detrimental to learners and programs.”
The NRMP will involve the community in future discussions “to continue to give learners a voice,” she said.
A version of this article first appeared on Medscape.com.
– mostly medical students, residents, and fellows – who supported the change.
The program’s decision comes after nearly 3 months of feedback from the public, medical students, and education community. Although about 60% of public respondents believed the change could reduce stress and allow students more time for momentous career decisions, the program’s board of directors decided the disadvantages were “of greater consequence,” according to a Oct. 28 statement.
Those disadvantages included introducing application or interview behaviors that could increase students’ stress; potentially identifying partially matched or unmatched applicants, which could lead to bias; and extending the match process time for those applicants.
In addition, members of 12 medical education and student organizations raised other concerns, such as the proposed change not addressing high application numbers, according to the statement. NRMP has reported record numbers of applicants over the past few years, typically with more applicants than available program slots.
“While the testimony gave nod to the positive aspects of the proposal ... there was substantially more concern voiced about the potential negative consequences identified in the public comments,” NRMP President and CEO Donna L. Lamb, DHSc, MBA, BSN, told this news organization. Some of those issues could not be addressed without further study, so the board decided not to proceed with the proposal, she explained.
The proposal would have separated the Main Residency Match into two phases and replaced the Supplemental Offer and Acceptance Program (SOAP), in which unmatched or partially matched applicants apply for unfilled residency positions. Under the proposed change, each phase would have operated the same way, from rank order lists and using a matching algorithm to a pair of Match Days instead of a single day.
The two-phase process would have given students who didn’t match more time to carefully weigh residency programs – they can apply to up to 45 placements as part of SOAP – that will guide their career path for the next few years, PGY-1 intern Asim A., who asked not to be identified further, told this news organization. The alternative is a hasty decision once students learn which residency spots are available, he added. “Applicants would have breathing room to make a more informed decision.”
Asim, who is Canadian, said he is participating in a transitional year in internal medicine in the hopes of being matched into internal medicine or psychiatry. He said Canada’s two-phase match is a “lot less stressful” than the U.S. system.
Meanwhile, students on Reddit’s medical school community also questioned NRMP’s decision.
“A significant majority of those surveyed thought it would be beneficial. But NRMP decides to not go through with it,” one Reddit user wrote. Another posted, “The one thing that could have improved the match and they chose not to do it.”
Others supported the decision to retain a 1-day match.
“I think this was the right call,” Bryan Carmody, MD, an outspoken medical education blogger, tweeted after learning of NRMP’s decision. Dr. Carmody, a pediatric nephrologist, previously expressed to this news organization misgivings about whether the two-phase match would make it difficult for programs to thoroughly review candidates and vice versa. He was concerned that it would compress the interview season and pressure programs to rapidly review applicants and conduct interviews.
More than 8,000 people responded to the public survey that began in August and ran for a month. Nearly two-thirds of the respondents (60%) were students, residents, or fellows. About 25% included faculty, program directors, and staff. Among the survey findings, respondents were equally divided between whether the two-phase match would be modestly advantageous (30%) or significantly advantageous (30%) compared to 20% who viewed it as modestly or significantly disadvantageous.
The NRMP said it would continue engaging with the community through focus groups and other means to improve the match experience and transition to residency.
“It is important to remember that a proposal is just that,” Dr. Lamb told this news orgnization, “an opportunity to discuss the pros and cons of an idea or framework ... and to mitigate unwanted consequences determined to be detrimental to learners and programs.”
The NRMP will involve the community in future discussions “to continue to give learners a voice,” she said.
A version of this article first appeared on Medscape.com.
Lego introduces first character with vitiligo
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The
The character appears with the customizable array of players to assemble for a table football team.
It’s the latest representation of the disease as toymakers diversify their lines.
In May 2022, Mattel released a Ken doll with vitiligo after a Barbie with vitiligo was released in 2020. Rainbow High and other toy makers also have character versions.
The Lego addition follows a big summer medically for vitiligo as the first treatment was approved for repigmentation. In July, a cream formulation of ruxolitinib (Opzelura), a Janus kinase inhibitor, became the first repigmentation treatment approved by the Food and Drug Administration for nonsegmental vitiligo, the most common form of the disease.
Vitiligo is estimated to affect 1.9 million–2.8 million adults in the United States and more than 100 million people worldwide. It cuts across races and genders and can be psychologically painful for many who live with it.
John E. Harris, MD, director of the Vitiligo Clinic and Research Center at the University of Massachusetts, Worcester, wrote about the Lego character in his blog “Speaking of Vitiligo ...” saying: “I could not be more excited. This new minifigure also serves as a way to educate both children and adults who are not familiar with vitiligo about the disease.”
He noted that until recently vitiligo representation in kids’ toys has been limited. “By adding diversity such as representations of vitiligo in toys, it can help remove stigmas associated with vitiligo and give children more options that they can relate to.”
Erika Page of Richmond, Va., who founded and edits the vitiligo blog “Living Dappled,” told this news organization she was thrilled to see the new Lego character.
“Growing up I didn’t know anyone who looked like me, let alone a toy or a character,” she said. The message the representations send is important not just for the kids but for the parents of kids with vitiligo who want to help their kids in any way they can.
Ms. Page was diagnosed with vitiligo at age 7 and struggled emotionally in her high school and college years when she often looked in the mirror, saw “giraffe-like” spots, and cried. Over time she lost 100% of her pigment to the condition and today at age 33, lives with universal vitiligo or overall very pale skin.
She founded the Living Dappled blog 6 years ago to help people with the disease feel less alone. The Lego character will also help with that, she said.
“Growing up with vitiligo was so isolating and you felt so different,” Ms. Page said. “Today we see billboards and models and dolls and now Legos that look like us. I hope this is a first of many to come for Lego.”
Dr. Harris and Ms. Page declared no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Pediatric celiac disease incidence varies across U.S., Europe
, according to a new report.
The overall high incidence among pediatric patients warrants a low threshold for screening and additional research on region-specific celiac disease triggers, the authors write.
“Determining the true incidence of celiac disease (CD) is not possible without nonbiased screening for the disease. This is because many cases occur with neither a family history nor with classic symptoms,” write Edwin Liu, MD, a pediatric gastroenterologist at the Children’s Hospital Colorado Anschutz Medical Campus and director of the Colorado Center for Celiac Disease, and colleagues.
“Individuals may have celiac disease autoimmunity without having CD if they have transient or fluctuating antibody levels, low antibody levels without biopsy evaluation, dietary modification influencing further evaluation, or potential celiac disease,” they write.
The study was published online in The American Journal of Gastroenterology.
Celiac disease incidence
The Environmental Determinants of Diabetes in the Young (TEDDY) study prospectively follows children born between 2004 and 2010 who are at genetic risk for both type 1 diabetes and CD at six clinical sites in four countries: the United States, Finland, Germany, and Sweden. In the United States, patients are enrolled in Colorado, Georgia, and Washington.
As part of TEDDY, children are longitudinally monitored for celiac disease autoimmunity (CDA) by assessment of autoantibodies to tissue transglutaminase (tTGA). The protocol is designed to analyze the development of persistent tTGA positivity, CDA, and subsequent CD. The study population contains various DQ2.5 and DQ8.1 combinations, which represent the highest-risk human leukocyte antigen (HLA) DQ haplogentotypes for CD.
From September 2004 through February 2010, more than 424,000 newborns were screened for specific HLA haplogenotypes, and 8,676 children were enrolled in TEDDY at the six clinical sites. The eligible haplogenotypes included DQ2.5/DQ2.5, DQ2.5/DQ8.1, DQ8.1/DQ8.1, and DQ8.1/DQ4.2.
Blood samples were obtained and stored every 3 months until age 48 months and at least every 6 months after that. At age 2, participants were screened annually for tTGA. With the first tTGA-positive result, all prior collected samples from the patient were tested for tTGA to determine the earliest time point of autoimmunity.
CDA, a primary study outcome, was defined as positivity in two consecutive tTGA tests at least 3 months apart.
In seropositive children, CD was defined on the basis of a duodenal biopsy with a Marsh score of 2 or higher. The decision to perform a biopsy was determined by the clinical gastroenterologist and was outside of the study protocol. When a biopsy wasn’t performed, participants with an average tTGA of 100 units or greater from two positive tests were considered to have CD for the study purposes.
As of July 2020, among the children who had undergone one or more tTGA tests, 6,628 HLA-typed eligible children were found to carry the DQ2.5, the D8.1, or both haplogenotypes and were included in the analysis. The median follow-up period was 11.5 years.
Overall, 580 children (9%) had a first-degree relative with type 1 diabetes, and 317 children (5%) reported a first-degree relative with CD.
Among the 6,628 children, 1,299 (20%) met the CDA outcome, and 529 (8%) met the study diagnostic criteria for CD on the basis of biopsy or persistently high tTGA levels. The median age at CDA across all sites was 41 months. Most children with CDA were asymptomatic.
Overall, the 10-year cumulative incidence was highest in Sweden, at 8.4% for CDA and 3% for CD. Within the United States, Colorado had the highest cumulative incidence for both endpoints, at 6.5% for CDA and 2.4% for CD. Washington had the lowest incidence across all sites, at 4.6% for CDA and 0.9% for CD.
“CDA and CD risk varied substantially by haplogenotype and by clinical center, but the relative risk by region was preserved regardless of the haplogenotype,” the authors write. “For example, the disease burden for each region remained highest in Sweden and lowest in Washington state for all haplogenotypes.”
Site-specific risks
In the HLA, sex, and family-adjusted model, Colorado children had a 2.5-fold higher risk of CD, compared with Washington children. Likewise, Swedish children had a 1.8-fold higher risk of CD than children in Germany, a 1.7-fold higher than children in the United States, and a 1.4-fold higher risk than children in Finland.
Among DQ2.5 participants, Sweden demonstrated the highest risk, with 63.1% of patients developing CDA by age 10 and 28.3% developing CD by age 10. Finland consistently had a higher incidence of CDA than Colorado, at 60.4% versus 50.9%, for DQ2.5 participants but a lower incidence of CD than Colorado, at 20.3% versus 22.6%.
The research team performed a post hoc sensitivity analysis using a lower tTGA cutoff to reduce bias in site differences for biopsy referral and to increase sensitivity of the CD definition for incidence estimation. When the tTGA cutoff was lowered to an average two-visit tTGA of 67.4 or higher, more children met the serologic criteria for CD.
“Even with this lower cutoff, the differences in the risk of CD between clinical sites and countries were still observed with statistical significance,” the authors write. “This indicates that the regional differences in CD incidence could not be solely attributed to detection biases posed by differential biopsy rates.”
Multiple environmental factors likely account for the differences in autoimmunity among regions, the authors write. These variables include diet, chemical exposures, vaccination patterns, early-life gastrointestinal infections, and interactions among these factors. For instance, the Swedish site has the lowest rotavirus vaccination rates and the highest median gluten intake among the TEDDY sites.
Future prospective studies should capture environmental, genetic, and epigenetic exposures to assess causal pathways and plan for preventive strategies, the authors write. The TEDDY study is pursuing this research.
“From a policy standpoint, this informs future screening practices and supports efforts toward mass screening, at least in some areas,” the authors write. “In the clinical setting, this points to the importance for clinicians to have a low threshold for CD screening in the appropriate clinical setting.”
The TEDDY study is funded by several grants from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute of Environmental Health Sciences, the Centers for Disease Control and Prevention, and the Juvenile Diabetes Research Foundation. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to a new report.
The overall high incidence among pediatric patients warrants a low threshold for screening and additional research on region-specific celiac disease triggers, the authors write.
“Determining the true incidence of celiac disease (CD) is not possible without nonbiased screening for the disease. This is because many cases occur with neither a family history nor with classic symptoms,” write Edwin Liu, MD, a pediatric gastroenterologist at the Children’s Hospital Colorado Anschutz Medical Campus and director of the Colorado Center for Celiac Disease, and colleagues.
“Individuals may have celiac disease autoimmunity without having CD if they have transient or fluctuating antibody levels, low antibody levels without biopsy evaluation, dietary modification influencing further evaluation, or potential celiac disease,” they write.
The study was published online in The American Journal of Gastroenterology.
Celiac disease incidence
The Environmental Determinants of Diabetes in the Young (TEDDY) study prospectively follows children born between 2004 and 2010 who are at genetic risk for both type 1 diabetes and CD at six clinical sites in four countries: the United States, Finland, Germany, and Sweden. In the United States, patients are enrolled in Colorado, Georgia, and Washington.
As part of TEDDY, children are longitudinally monitored for celiac disease autoimmunity (CDA) by assessment of autoantibodies to tissue transglutaminase (tTGA). The protocol is designed to analyze the development of persistent tTGA positivity, CDA, and subsequent CD. The study population contains various DQ2.5 and DQ8.1 combinations, which represent the highest-risk human leukocyte antigen (HLA) DQ haplogentotypes for CD.
From September 2004 through February 2010, more than 424,000 newborns were screened for specific HLA haplogenotypes, and 8,676 children were enrolled in TEDDY at the six clinical sites. The eligible haplogenotypes included DQ2.5/DQ2.5, DQ2.5/DQ8.1, DQ8.1/DQ8.1, and DQ8.1/DQ4.2.
Blood samples were obtained and stored every 3 months until age 48 months and at least every 6 months after that. At age 2, participants were screened annually for tTGA. With the first tTGA-positive result, all prior collected samples from the patient were tested for tTGA to determine the earliest time point of autoimmunity.
CDA, a primary study outcome, was defined as positivity in two consecutive tTGA tests at least 3 months apart.
In seropositive children, CD was defined on the basis of a duodenal biopsy with a Marsh score of 2 or higher. The decision to perform a biopsy was determined by the clinical gastroenterologist and was outside of the study protocol. When a biopsy wasn’t performed, participants with an average tTGA of 100 units or greater from two positive tests were considered to have CD for the study purposes.
As of July 2020, among the children who had undergone one or more tTGA tests, 6,628 HLA-typed eligible children were found to carry the DQ2.5, the D8.1, or both haplogenotypes and were included in the analysis. The median follow-up period was 11.5 years.
Overall, 580 children (9%) had a first-degree relative with type 1 diabetes, and 317 children (5%) reported a first-degree relative with CD.
Among the 6,628 children, 1,299 (20%) met the CDA outcome, and 529 (8%) met the study diagnostic criteria for CD on the basis of biopsy or persistently high tTGA levels. The median age at CDA across all sites was 41 months. Most children with CDA were asymptomatic.
Overall, the 10-year cumulative incidence was highest in Sweden, at 8.4% for CDA and 3% for CD. Within the United States, Colorado had the highest cumulative incidence for both endpoints, at 6.5% for CDA and 2.4% for CD. Washington had the lowest incidence across all sites, at 4.6% for CDA and 0.9% for CD.
“CDA and CD risk varied substantially by haplogenotype and by clinical center, but the relative risk by region was preserved regardless of the haplogenotype,” the authors write. “For example, the disease burden for each region remained highest in Sweden and lowest in Washington state for all haplogenotypes.”
Site-specific risks
In the HLA, sex, and family-adjusted model, Colorado children had a 2.5-fold higher risk of CD, compared with Washington children. Likewise, Swedish children had a 1.8-fold higher risk of CD than children in Germany, a 1.7-fold higher than children in the United States, and a 1.4-fold higher risk than children in Finland.
Among DQ2.5 participants, Sweden demonstrated the highest risk, with 63.1% of patients developing CDA by age 10 and 28.3% developing CD by age 10. Finland consistently had a higher incidence of CDA than Colorado, at 60.4% versus 50.9%, for DQ2.5 participants but a lower incidence of CD than Colorado, at 20.3% versus 22.6%.
The research team performed a post hoc sensitivity analysis using a lower tTGA cutoff to reduce bias in site differences for biopsy referral and to increase sensitivity of the CD definition for incidence estimation. When the tTGA cutoff was lowered to an average two-visit tTGA of 67.4 or higher, more children met the serologic criteria for CD.
“Even with this lower cutoff, the differences in the risk of CD between clinical sites and countries were still observed with statistical significance,” the authors write. “This indicates that the regional differences in CD incidence could not be solely attributed to detection biases posed by differential biopsy rates.”
Multiple environmental factors likely account for the differences in autoimmunity among regions, the authors write. These variables include diet, chemical exposures, vaccination patterns, early-life gastrointestinal infections, and interactions among these factors. For instance, the Swedish site has the lowest rotavirus vaccination rates and the highest median gluten intake among the TEDDY sites.
Future prospective studies should capture environmental, genetic, and epigenetic exposures to assess causal pathways and plan for preventive strategies, the authors write. The TEDDY study is pursuing this research.
“From a policy standpoint, this informs future screening practices and supports efforts toward mass screening, at least in some areas,” the authors write. “In the clinical setting, this points to the importance for clinicians to have a low threshold for CD screening in the appropriate clinical setting.”
The TEDDY study is funded by several grants from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute of Environmental Health Sciences, the Centers for Disease Control and Prevention, and the Juvenile Diabetes Research Foundation. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
, according to a new report.
The overall high incidence among pediatric patients warrants a low threshold for screening and additional research on region-specific celiac disease triggers, the authors write.
“Determining the true incidence of celiac disease (CD) is not possible without nonbiased screening for the disease. This is because many cases occur with neither a family history nor with classic symptoms,” write Edwin Liu, MD, a pediatric gastroenterologist at the Children’s Hospital Colorado Anschutz Medical Campus and director of the Colorado Center for Celiac Disease, and colleagues.
“Individuals may have celiac disease autoimmunity without having CD if they have transient or fluctuating antibody levels, low antibody levels without biopsy evaluation, dietary modification influencing further evaluation, or potential celiac disease,” they write.
The study was published online in The American Journal of Gastroenterology.
Celiac disease incidence
The Environmental Determinants of Diabetes in the Young (TEDDY) study prospectively follows children born between 2004 and 2010 who are at genetic risk for both type 1 diabetes and CD at six clinical sites in four countries: the United States, Finland, Germany, and Sweden. In the United States, patients are enrolled in Colorado, Georgia, and Washington.
As part of TEDDY, children are longitudinally monitored for celiac disease autoimmunity (CDA) by assessment of autoantibodies to tissue transglutaminase (tTGA). The protocol is designed to analyze the development of persistent tTGA positivity, CDA, and subsequent CD. The study population contains various DQ2.5 and DQ8.1 combinations, which represent the highest-risk human leukocyte antigen (HLA) DQ haplogentotypes for CD.
From September 2004 through February 2010, more than 424,000 newborns were screened for specific HLA haplogenotypes, and 8,676 children were enrolled in TEDDY at the six clinical sites. The eligible haplogenotypes included DQ2.5/DQ2.5, DQ2.5/DQ8.1, DQ8.1/DQ8.1, and DQ8.1/DQ4.2.
Blood samples were obtained and stored every 3 months until age 48 months and at least every 6 months after that. At age 2, participants were screened annually for tTGA. With the first tTGA-positive result, all prior collected samples from the patient were tested for tTGA to determine the earliest time point of autoimmunity.
CDA, a primary study outcome, was defined as positivity in two consecutive tTGA tests at least 3 months apart.
In seropositive children, CD was defined on the basis of a duodenal biopsy with a Marsh score of 2 or higher. The decision to perform a biopsy was determined by the clinical gastroenterologist and was outside of the study protocol. When a biopsy wasn’t performed, participants with an average tTGA of 100 units or greater from two positive tests were considered to have CD for the study purposes.
As of July 2020, among the children who had undergone one or more tTGA tests, 6,628 HLA-typed eligible children were found to carry the DQ2.5, the D8.1, or both haplogenotypes and were included in the analysis. The median follow-up period was 11.5 years.
Overall, 580 children (9%) had a first-degree relative with type 1 diabetes, and 317 children (5%) reported a first-degree relative with CD.
Among the 6,628 children, 1,299 (20%) met the CDA outcome, and 529 (8%) met the study diagnostic criteria for CD on the basis of biopsy or persistently high tTGA levels. The median age at CDA across all sites was 41 months. Most children with CDA were asymptomatic.
Overall, the 10-year cumulative incidence was highest in Sweden, at 8.4% for CDA and 3% for CD. Within the United States, Colorado had the highest cumulative incidence for both endpoints, at 6.5% for CDA and 2.4% for CD. Washington had the lowest incidence across all sites, at 4.6% for CDA and 0.9% for CD.
“CDA and CD risk varied substantially by haplogenotype and by clinical center, but the relative risk by region was preserved regardless of the haplogenotype,” the authors write. “For example, the disease burden for each region remained highest in Sweden and lowest in Washington state for all haplogenotypes.”
Site-specific risks
In the HLA, sex, and family-adjusted model, Colorado children had a 2.5-fold higher risk of CD, compared with Washington children. Likewise, Swedish children had a 1.8-fold higher risk of CD than children in Germany, a 1.7-fold higher than children in the United States, and a 1.4-fold higher risk than children in Finland.
Among DQ2.5 participants, Sweden demonstrated the highest risk, with 63.1% of patients developing CDA by age 10 and 28.3% developing CD by age 10. Finland consistently had a higher incidence of CDA than Colorado, at 60.4% versus 50.9%, for DQ2.5 participants but a lower incidence of CD than Colorado, at 20.3% versus 22.6%.
The research team performed a post hoc sensitivity analysis using a lower tTGA cutoff to reduce bias in site differences for biopsy referral and to increase sensitivity of the CD definition for incidence estimation. When the tTGA cutoff was lowered to an average two-visit tTGA of 67.4 or higher, more children met the serologic criteria for CD.
“Even with this lower cutoff, the differences in the risk of CD between clinical sites and countries were still observed with statistical significance,” the authors write. “This indicates that the regional differences in CD incidence could not be solely attributed to detection biases posed by differential biopsy rates.”
Multiple environmental factors likely account for the differences in autoimmunity among regions, the authors write. These variables include diet, chemical exposures, vaccination patterns, early-life gastrointestinal infections, and interactions among these factors. For instance, the Swedish site has the lowest rotavirus vaccination rates and the highest median gluten intake among the TEDDY sites.
Future prospective studies should capture environmental, genetic, and epigenetic exposures to assess causal pathways and plan for preventive strategies, the authors write. The TEDDY study is pursuing this research.
“From a policy standpoint, this informs future screening practices and supports efforts toward mass screening, at least in some areas,” the authors write. “In the clinical setting, this points to the importance for clinicians to have a low threshold for CD screening in the appropriate clinical setting.”
The TEDDY study is funded by several grants from the National Institute of Diabetes and Digestive and Kidney Diseases, the National Institute of Allergy and Infectious Diseases, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Institute of Environmental Health Sciences, the Centers for Disease Control and Prevention, and the Juvenile Diabetes Research Foundation. The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM AMERICAN JOURNAL OF GASTROENTEROLOGY
Children and COVID: New cases increase for second straight week
New COVID-19 cases rose among U.S. children for the second consecutive week, while hospitals saw signs of renewed activity on the part of SARS-CoV-2.
, when the count fell to its lowest level in more than a year, the American Academy of Pediatrics and the Children’s Hospital Association said in their joint report.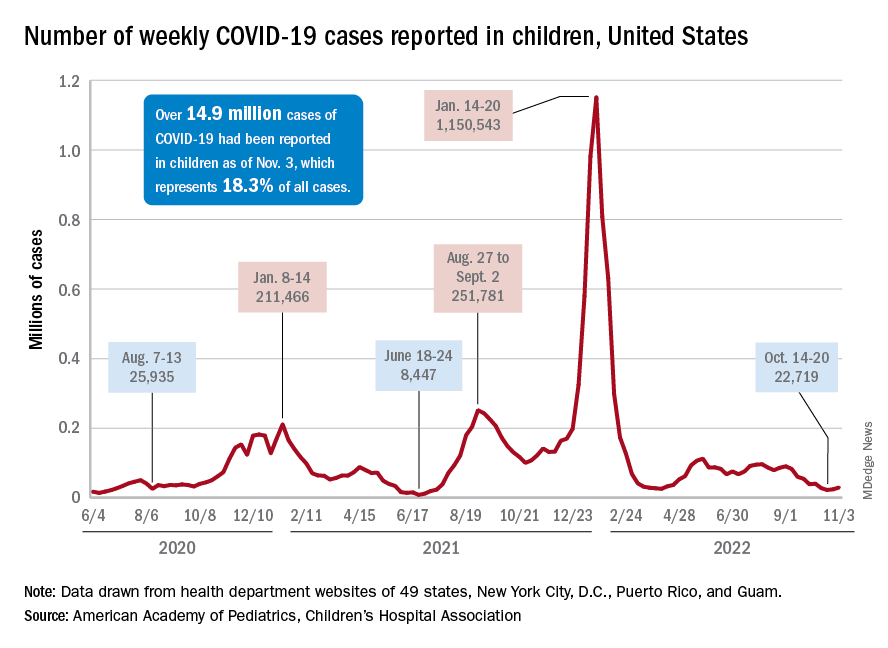
The 7-day average for ED visits with diagnosed COVID was down to just 0.6% of all ED visits for 12- to 15-year-olds as late as Oct. 23 but has moved up to 0.7% since then. Among those aged 16-17 years, the 7-day average was also down to 0.6% for just one day, Oct. 19, but was up to 0.8% as of Nov. 4. So far, though, a similar increase has not yet occurred for ED visits among children aged 0-11 years, the CDC said on its COVID Data Tracker.
The trend is discernible, however, when looking at hospitalizations of children with confirmed COVID. The rate of new admissions of children aged 0-17 years was 0.16 per 100,000 population as late as Oct. 23 but ticked up a notch after that and has been 0.17 per 100,000 since, according to the CDC. As with the ED rate, hospitalizations had been steadily declining since late August.
Vaccine initiation continues to slow
During the week of Oct. 27 to Nov. 2, about 30,000 children under 5 years of age received their initial COVID vaccination. A month earlier (Sept. 29 to Oct. 5), that number was about 40,000. A month before that, about 53,000 children aged 0-5 years received their initial dose, the AAP said in a separate vaccination report based on CDC data.
All of that reduced interest adds up to 7.4% of the age group having received at least one dose and just 3.2% being fully vaccinated as of Nov. 2. Among children aged 5-11 years, the corresponding vaccination rates are 38.9% and 31.8%, while those aged 12-17 years are at 71.3% and 61.1%, the CDC said.
Looking at just the first 20 weeks of the vaccination experience for each age group shows that 1.6 million children under 5 years of age had received at least an initial dose, compared with 8.1 million children aged 5-11 years and 8.1 million children aged 12-15, the AAP said.
New COVID-19 cases rose among U.S. children for the second consecutive week, while hospitals saw signs of renewed activity on the part of SARS-CoV-2.
, when the count fell to its lowest level in more than a year, the American Academy of Pediatrics and the Children’s Hospital Association said in their joint report.
The 7-day average for ED visits with diagnosed COVID was down to just 0.6% of all ED visits for 12- to 15-year-olds as late as Oct. 23 but has moved up to 0.7% since then. Among those aged 16-17 years, the 7-day average was also down to 0.6% for just one day, Oct. 19, but was up to 0.8% as of Nov. 4. So far, though, a similar increase has not yet occurred for ED visits among children aged 0-11 years, the CDC said on its COVID Data Tracker.
The trend is discernible, however, when looking at hospitalizations of children with confirmed COVID. The rate of new admissions of children aged 0-17 years was 0.16 per 100,000 population as late as Oct. 23 but ticked up a notch after that and has been 0.17 per 100,000 since, according to the CDC. As with the ED rate, hospitalizations had been steadily declining since late August.
Vaccine initiation continues to slow
During the week of Oct. 27 to Nov. 2, about 30,000 children under 5 years of age received their initial COVID vaccination. A month earlier (Sept. 29 to Oct. 5), that number was about 40,000. A month before that, about 53,000 children aged 0-5 years received their initial dose, the AAP said in a separate vaccination report based on CDC data.
All of that reduced interest adds up to 7.4% of the age group having received at least one dose and just 3.2% being fully vaccinated as of Nov. 2. Among children aged 5-11 years, the corresponding vaccination rates are 38.9% and 31.8%, while those aged 12-17 years are at 71.3% and 61.1%, the CDC said.
Looking at just the first 20 weeks of the vaccination experience for each age group shows that 1.6 million children under 5 years of age had received at least an initial dose, compared with 8.1 million children aged 5-11 years and 8.1 million children aged 12-15, the AAP said.
New COVID-19 cases rose among U.S. children for the second consecutive week, while hospitals saw signs of renewed activity on the part of SARS-CoV-2.
, when the count fell to its lowest level in more than a year, the American Academy of Pediatrics and the Children’s Hospital Association said in their joint report.
The 7-day average for ED visits with diagnosed COVID was down to just 0.6% of all ED visits for 12- to 15-year-olds as late as Oct. 23 but has moved up to 0.7% since then. Among those aged 16-17 years, the 7-day average was also down to 0.6% for just one day, Oct. 19, but was up to 0.8% as of Nov. 4. So far, though, a similar increase has not yet occurred for ED visits among children aged 0-11 years, the CDC said on its COVID Data Tracker.
The trend is discernible, however, when looking at hospitalizations of children with confirmed COVID. The rate of new admissions of children aged 0-17 years was 0.16 per 100,000 population as late as Oct. 23 but ticked up a notch after that and has been 0.17 per 100,000 since, according to the CDC. As with the ED rate, hospitalizations had been steadily declining since late August.
Vaccine initiation continues to slow
During the week of Oct. 27 to Nov. 2, about 30,000 children under 5 years of age received their initial COVID vaccination. A month earlier (Sept. 29 to Oct. 5), that number was about 40,000. A month before that, about 53,000 children aged 0-5 years received their initial dose, the AAP said in a separate vaccination report based on CDC data.
All of that reduced interest adds up to 7.4% of the age group having received at least one dose and just 3.2% being fully vaccinated as of Nov. 2. Among children aged 5-11 years, the corresponding vaccination rates are 38.9% and 31.8%, while those aged 12-17 years are at 71.3% and 61.1%, the CDC said.
Looking at just the first 20 weeks of the vaccination experience for each age group shows that 1.6 million children under 5 years of age had received at least an initial dose, compared with 8.1 million children aged 5-11 years and 8.1 million children aged 12-15, the AAP said.
Many moms don’t remember well-child nutrition advice
Recent findings from a study examining mothers’ recall of doctors’ advice on early-child nutrition suggest that key feeding messages may not be heard, remembered, or even delivered.
During a typical child wellness visit, pediatricians provide parents with anticipatory guidance on all aspects of child development and safety, up to the age of 5 years.
The analysis of data from a subset of 1,302 mothers participating in the 2017-2019 National Survey of Family Growth showed that those older than 31 years of age and those who identified as non-Hispanic White were more likely to recall discussion of certain child nutrition topics compared with younger mothers or those who identified as Hispanic.
Of the six child-feeding topics referenced from the American Academy of Pediatrics’ “Bright Futures Guidelines,” less than half of the mothers, all of whom had a child between the ages of 6 months and 5 years, recalled guidance on limiting meals in front of the television or other electronic devices. Similarly, fewer than 50% remembered being told not to force their child to finish a bottle or food, the analysis showed.
When it came to the best time to introduce solid foods, 37% didn’t recall being told to wait at least 4 months and preferably, 6 months. In fact, these mothers reported being advised to introduce solid foods before 6 months, said Andrea McGowan, MPH, of the National Center for Chronic Disease Prevention and Health Promotion, U.S. Centers for Disease Control and Prevention, Atlanta, and colleagues.
The study was published in the Journal of Nutrition Education and Behavior.
“All in all, this research draws attention to certain nutrition guidance topics or subpopulations that might be prioritized to improve receipt and recall of guidance,” said Ms. McGowan, now a first-year medical student at the University of Michigan, Ann Arbor, in a podcast. “This research ... implores us to consider ways to revamp the existing standard practice for pediatric well-child care to improve recall of messages.”
The analysis also included data on mothers’ recall of advice on offering foods with different tastes and textures; offering a variety of fruits and vegetables; and limiting added sugar. More than half of mothers remembered discussing four or five child nutrition topics, but 31% recalled talking about only one or two. Offering a variety of fruits and vegetables had the highest percentage of recall.
The study wasn’t powered to determine whether the nutrition guidance provided at a well-child visit was not remembered or not provided, Ms. McGowan said, adding: “So exploring this is definitely the goal of future research.”
However, pediatricians report spending an average of 18 minutes with children and their parents, she noted. “This is definitely not enough time to cover every single topic a pediatrician or a parent might want to discuss.” Other barriers, such as a lack of insurance or transportation, may limit parents’ access to this kind of anticipatory guidance, the researchers said.
Priority should be given to certain topics and to certain mothers, they suggested. “Innovative strategies tailored to families’ needs might alleviate the HCP [health care provider] burden and could enhance parental recall, especially when messaging is culturally relevant and personalized,” Ms. McGowan said.
Two independent experts agreed in interviews. Pediatricians must do their best to tailor advice to each particular family so that parents can engage in the conversation, said Lauren Fiechtner, MD, director of the center for pediatric nutrition at Mass General for Children, Boston. “As the authors suggest, we should seek to understand the cultural relevance of our recommendations and to understand the barriers our patient families might face in implementing our advice,” said Dr. Fiechtner, who is also an assistant professor at Harvard Medical School, also in Boston.
“Much of the instructions we as pediatricians give to parents must be repeated and reinforced,” said Rebecca S. Fisk, MD, a pediatrician at Lenox Hill Hospital, Northwell Health, in New York. Often, the doctor’s advice runs counter to what family and friends recommend, she pointed out. Some parents may believe that “the baby who starts solid food earlier will sleep through the night earlier or that eating in front of the TV relaxes the child or allows them to eat more,” Dr. Fisk explained. In her practice, a nurse goes over her instructions, answers questions, and provides specific examples and written information.
Sometimes, even that’s not enough, Dr. Fisk admitted. “I, myself, have fielded many repeated questions about feeding, when to start, how much to give, and so on, despite printed guidance given to parents at well-child visits.”
This study was funded by the U.S. Centers for Disease Control and Prevention. Ms. McGowan and study coauthors reported having no potential conflicts of interest. Dr. Fiechtner and Dr. Fisk disclosed having no potential conflicts of interest.
Recent findings from a study examining mothers’ recall of doctors’ advice on early-child nutrition suggest that key feeding messages may not be heard, remembered, or even delivered.
During a typical child wellness visit, pediatricians provide parents with anticipatory guidance on all aspects of child development and safety, up to the age of 5 years.
The analysis of data from a subset of 1,302 mothers participating in the 2017-2019 National Survey of Family Growth showed that those older than 31 years of age and those who identified as non-Hispanic White were more likely to recall discussion of certain child nutrition topics compared with younger mothers or those who identified as Hispanic.
Of the six child-feeding topics referenced from the American Academy of Pediatrics’ “Bright Futures Guidelines,” less than half of the mothers, all of whom had a child between the ages of 6 months and 5 years, recalled guidance on limiting meals in front of the television or other electronic devices. Similarly, fewer than 50% remembered being told not to force their child to finish a bottle or food, the analysis showed.
When it came to the best time to introduce solid foods, 37% didn’t recall being told to wait at least 4 months and preferably, 6 months. In fact, these mothers reported being advised to introduce solid foods before 6 months, said Andrea McGowan, MPH, of the National Center for Chronic Disease Prevention and Health Promotion, U.S. Centers for Disease Control and Prevention, Atlanta, and colleagues.
The study was published in the Journal of Nutrition Education and Behavior.
“All in all, this research draws attention to certain nutrition guidance topics or subpopulations that might be prioritized to improve receipt and recall of guidance,” said Ms. McGowan, now a first-year medical student at the University of Michigan, Ann Arbor, in a podcast. “This research ... implores us to consider ways to revamp the existing standard practice for pediatric well-child care to improve recall of messages.”
The analysis also included data on mothers’ recall of advice on offering foods with different tastes and textures; offering a variety of fruits and vegetables; and limiting added sugar. More than half of mothers remembered discussing four or five child nutrition topics, but 31% recalled talking about only one or two. Offering a variety of fruits and vegetables had the highest percentage of recall.
The study wasn’t powered to determine whether the nutrition guidance provided at a well-child visit was not remembered or not provided, Ms. McGowan said, adding: “So exploring this is definitely the goal of future research.”
However, pediatricians report spending an average of 18 minutes with children and their parents, she noted. “This is definitely not enough time to cover every single topic a pediatrician or a parent might want to discuss.” Other barriers, such as a lack of insurance or transportation, may limit parents’ access to this kind of anticipatory guidance, the researchers said.
Priority should be given to certain topics and to certain mothers, they suggested. “Innovative strategies tailored to families’ needs might alleviate the HCP [health care provider] burden and could enhance parental recall, especially when messaging is culturally relevant and personalized,” Ms. McGowan said.
Two independent experts agreed in interviews. Pediatricians must do their best to tailor advice to each particular family so that parents can engage in the conversation, said Lauren Fiechtner, MD, director of the center for pediatric nutrition at Mass General for Children, Boston. “As the authors suggest, we should seek to understand the cultural relevance of our recommendations and to understand the barriers our patient families might face in implementing our advice,” said Dr. Fiechtner, who is also an assistant professor at Harvard Medical School, also in Boston.
“Much of the instructions we as pediatricians give to parents must be repeated and reinforced,” said Rebecca S. Fisk, MD, a pediatrician at Lenox Hill Hospital, Northwell Health, in New York. Often, the doctor’s advice runs counter to what family and friends recommend, she pointed out. Some parents may believe that “the baby who starts solid food earlier will sleep through the night earlier or that eating in front of the TV relaxes the child or allows them to eat more,” Dr. Fisk explained. In her practice, a nurse goes over her instructions, answers questions, and provides specific examples and written information.
Sometimes, even that’s not enough, Dr. Fisk admitted. “I, myself, have fielded many repeated questions about feeding, when to start, how much to give, and so on, despite printed guidance given to parents at well-child visits.”
This study was funded by the U.S. Centers for Disease Control and Prevention. Ms. McGowan and study coauthors reported having no potential conflicts of interest. Dr. Fiechtner and Dr. Fisk disclosed having no potential conflicts of interest.
Recent findings from a study examining mothers’ recall of doctors’ advice on early-child nutrition suggest that key feeding messages may not be heard, remembered, or even delivered.
During a typical child wellness visit, pediatricians provide parents with anticipatory guidance on all aspects of child development and safety, up to the age of 5 years.
The analysis of data from a subset of 1,302 mothers participating in the 2017-2019 National Survey of Family Growth showed that those older than 31 years of age and those who identified as non-Hispanic White were more likely to recall discussion of certain child nutrition topics compared with younger mothers or those who identified as Hispanic.
Of the six child-feeding topics referenced from the American Academy of Pediatrics’ “Bright Futures Guidelines,” less than half of the mothers, all of whom had a child between the ages of 6 months and 5 years, recalled guidance on limiting meals in front of the television or other electronic devices. Similarly, fewer than 50% remembered being told not to force their child to finish a bottle or food, the analysis showed.
When it came to the best time to introduce solid foods, 37% didn’t recall being told to wait at least 4 months and preferably, 6 months. In fact, these mothers reported being advised to introduce solid foods before 6 months, said Andrea McGowan, MPH, of the National Center for Chronic Disease Prevention and Health Promotion, U.S. Centers for Disease Control and Prevention, Atlanta, and colleagues.
The study was published in the Journal of Nutrition Education and Behavior.
“All in all, this research draws attention to certain nutrition guidance topics or subpopulations that might be prioritized to improve receipt and recall of guidance,” said Ms. McGowan, now a first-year medical student at the University of Michigan, Ann Arbor, in a podcast. “This research ... implores us to consider ways to revamp the existing standard practice for pediatric well-child care to improve recall of messages.”
The analysis also included data on mothers’ recall of advice on offering foods with different tastes and textures; offering a variety of fruits and vegetables; and limiting added sugar. More than half of mothers remembered discussing four or five child nutrition topics, but 31% recalled talking about only one or two. Offering a variety of fruits and vegetables had the highest percentage of recall.
The study wasn’t powered to determine whether the nutrition guidance provided at a well-child visit was not remembered or not provided, Ms. McGowan said, adding: “So exploring this is definitely the goal of future research.”
However, pediatricians report spending an average of 18 minutes with children and their parents, she noted. “This is definitely not enough time to cover every single topic a pediatrician or a parent might want to discuss.” Other barriers, such as a lack of insurance or transportation, may limit parents’ access to this kind of anticipatory guidance, the researchers said.
Priority should be given to certain topics and to certain mothers, they suggested. “Innovative strategies tailored to families’ needs might alleviate the HCP [health care provider] burden and could enhance parental recall, especially when messaging is culturally relevant and personalized,” Ms. McGowan said.
Two independent experts agreed in interviews. Pediatricians must do their best to tailor advice to each particular family so that parents can engage in the conversation, said Lauren Fiechtner, MD, director of the center for pediatric nutrition at Mass General for Children, Boston. “As the authors suggest, we should seek to understand the cultural relevance of our recommendations and to understand the barriers our patient families might face in implementing our advice,” said Dr. Fiechtner, who is also an assistant professor at Harvard Medical School, also in Boston.
“Much of the instructions we as pediatricians give to parents must be repeated and reinforced,” said Rebecca S. Fisk, MD, a pediatrician at Lenox Hill Hospital, Northwell Health, in New York. Often, the doctor’s advice runs counter to what family and friends recommend, she pointed out. Some parents may believe that “the baby who starts solid food earlier will sleep through the night earlier or that eating in front of the TV relaxes the child or allows them to eat more,” Dr. Fisk explained. In her practice, a nurse goes over her instructions, answers questions, and provides specific examples and written information.
Sometimes, even that’s not enough, Dr. Fisk admitted. “I, myself, have fielded many repeated questions about feeding, when to start, how much to give, and so on, despite printed guidance given to parents at well-child visits.”
This study was funded by the U.S. Centers for Disease Control and Prevention. Ms. McGowan and study coauthors reported having no potential conflicts of interest. Dr. Fiechtner and Dr. Fisk disclosed having no potential conflicts of interest.
With a little help from your friends
Case: You are talking with one of your teenage patients, who has a history of significant suicidal ideation and an aborted attempt, and you ask her if there is someone she can talk with if she is feeling suicidal. “I call a friend,” she says. “That’s the only thing that works when I’m feeling bad.”
During difficult times, it is important to have a repertoire of coping skills to address stress, tension, frustration, anxiety, anger, sadness, and to help avoid dangerous behaviors. It is also important to have someone to talk to. For many youth, talking with friends is their preferred coping skill and contact when struggling with intense feelings.
This is hardly surprising. Peer relations are central to adolescent development. The ongoing individuation-separation process means that adolescents are peeling away from the family and into a community of their peers, where they figure out who they are through social interactions in subtle and complex ways. Adolescents are often profoundly immersed in the world of their peers; they often spend more time with their peers in educational and social settings than with their parents or other adults; and their connections with peers are often pleasurable, engaging, supportive, and intense. It is natural that they would want to communicate with their peers during stressful times.
At the same time, they may also want to avoid talking with adults. They may identify adult figures with authority, expectations, and control. So much adolescent psychic suffering and so many mental health crises involve shame, guilt, and fear, and are associated with romance, love, disappointment, and trauma – all of which may be difficult to share with parents and adult figures.
Adults also struggle with these kinds of conversations. Even benign attempts at comforting the youth (“Don’t worry, it’ll get better,” “Everyone feels this way sometimes”) can be seen as invalidating. And in stressful times, a difficult conversation can be ignited by the fuel of adult anxieties about the independence and autonomy of the child that is coming, which can make charged conversations all the more inflammatory.
Reaching out to peers during stressful times is therefore developmentally appropriate and often feels far more comfortable, validating, and sympathetic.
One of the most important things we can do is to help kids understand when, how, and why they can support each other – and when they cannot. Whether we like it or not, for many youth, peers are peer mental health counselors. They have shared vocabularies and can share experiences in the mental health care system. In addition to relying on their peers, a great many youth we work with also see themselves as supports to their peers, so it’s not just a one-way street.
So we talk with them frankly about when, how, and why talking with their friends can be an effective way of getting through a hard time and when, how, and why they need to reach out to an adult.
Recognizing how positive peer support can be, we ask them to identify problems with it. Kids often recognize the drawbacks of relying on their peers for support. They can see how it can be a burden to their friends. They often acknowledge that their friends may be experts in some aspects of their lives but not in others. For example, they can have shared stressors in school, can have similar understandings of the drama in their lives, and can relate to each other’s worlds, but will also not necessarily know what to do if a situation becomes dangerous.
The youth also tend to understand that the stakes in these conversations are high. We have seen peer groups suffer terribly when the youth have felt responsible – and even been the last preceding contact – in bad or even fatal outcomes.
We need to open up conversations about different forms of communication: when teens need understanding, compassion, patience; when they need a good understanding of local, cultural contexts, and a sense of support without anxieties and stressors; and when they need support and adult capacities and connections to solve problems. We can help them understand how to access people – both peers and adults – but also discuss responsibility: who you are responsible for, how you cannot be responsible alone for your friends’ mental health, how they cannot be responsible for yours, and who can be responsible for you.
To this end, we validate the importance of peers and ask more specifically when the adolescent thinks it is helpful to contact peers and when they think it would not be helpful. Having teens explain the difference may help them identify the right times to connect with peers or adults.
We can then talk about how to understand that there are different kinds of crisis: the kind where comfort, understanding, and support from friends can alleviate the crisis, and times when it is imperative to involve adults.
We can then identify which adults in their lives they can contact and how they would do so, both in terms of method of communication (texting an older sister, speaking in person with a parent, calling a therapist) and what they could say.
Then comes a more difficult step. We help them think about how to identify adults whom they do not know: how to contact a hotline or go to an emergency room or call 911. It is important not just to provide the numbers or address, but to help them run through a brief script so they know what to say and would be comfortable saying in their own words (but effectively saying, “I really need to speak with someone right now, I’m not safe.”)
Helping youth understand the advantages and disadvantages of reaching out to peers, and when and how to reach out to adults, can be a constructive conversation. It is a chance not only to speak with and hear about a youth’s life and relationships but also a chance to give them a stronger and safer support network.
Dr. Henderson is a psychiatrist who treats children and adolescents at NYU Langone Health, New York.
Case: You are talking with one of your teenage patients, who has a history of significant suicidal ideation and an aborted attempt, and you ask her if there is someone she can talk with if she is feeling suicidal. “I call a friend,” she says. “That’s the only thing that works when I’m feeling bad.”
During difficult times, it is important to have a repertoire of coping skills to address stress, tension, frustration, anxiety, anger, sadness, and to help avoid dangerous behaviors. It is also important to have someone to talk to. For many youth, talking with friends is their preferred coping skill and contact when struggling with intense feelings.
This is hardly surprising. Peer relations are central to adolescent development. The ongoing individuation-separation process means that adolescents are peeling away from the family and into a community of their peers, where they figure out who they are through social interactions in subtle and complex ways. Adolescents are often profoundly immersed in the world of their peers; they often spend more time with their peers in educational and social settings than with their parents or other adults; and their connections with peers are often pleasurable, engaging, supportive, and intense. It is natural that they would want to communicate with their peers during stressful times.
At the same time, they may also want to avoid talking with adults. They may identify adult figures with authority, expectations, and control. So much adolescent psychic suffering and so many mental health crises involve shame, guilt, and fear, and are associated with romance, love, disappointment, and trauma – all of which may be difficult to share with parents and adult figures.
Adults also struggle with these kinds of conversations. Even benign attempts at comforting the youth (“Don’t worry, it’ll get better,” “Everyone feels this way sometimes”) can be seen as invalidating. And in stressful times, a difficult conversation can be ignited by the fuel of adult anxieties about the independence and autonomy of the child that is coming, which can make charged conversations all the more inflammatory.
Reaching out to peers during stressful times is therefore developmentally appropriate and often feels far more comfortable, validating, and sympathetic.
One of the most important things we can do is to help kids understand when, how, and why they can support each other – and when they cannot. Whether we like it or not, for many youth, peers are peer mental health counselors. They have shared vocabularies and can share experiences in the mental health care system. In addition to relying on their peers, a great many youth we work with also see themselves as supports to their peers, so it’s not just a one-way street.
So we talk with them frankly about when, how, and why talking with their friends can be an effective way of getting through a hard time and when, how, and why they need to reach out to an adult.
Recognizing how positive peer support can be, we ask them to identify problems with it. Kids often recognize the drawbacks of relying on their peers for support. They can see how it can be a burden to their friends. They often acknowledge that their friends may be experts in some aspects of their lives but not in others. For example, they can have shared stressors in school, can have similar understandings of the drama in their lives, and can relate to each other’s worlds, but will also not necessarily know what to do if a situation becomes dangerous.
The youth also tend to understand that the stakes in these conversations are high. We have seen peer groups suffer terribly when the youth have felt responsible – and even been the last preceding contact – in bad or even fatal outcomes.
We need to open up conversations about different forms of communication: when teens need understanding, compassion, patience; when they need a good understanding of local, cultural contexts, and a sense of support without anxieties and stressors; and when they need support and adult capacities and connections to solve problems. We can help them understand how to access people – both peers and adults – but also discuss responsibility: who you are responsible for, how you cannot be responsible alone for your friends’ mental health, how they cannot be responsible for yours, and who can be responsible for you.
To this end, we validate the importance of peers and ask more specifically when the adolescent thinks it is helpful to contact peers and when they think it would not be helpful. Having teens explain the difference may help them identify the right times to connect with peers or adults.
We can then talk about how to understand that there are different kinds of crisis: the kind where comfort, understanding, and support from friends can alleviate the crisis, and times when it is imperative to involve adults.
We can then identify which adults in their lives they can contact and how they would do so, both in terms of method of communication (texting an older sister, speaking in person with a parent, calling a therapist) and what they could say.
Then comes a more difficult step. We help them think about how to identify adults whom they do not know: how to contact a hotline or go to an emergency room or call 911. It is important not just to provide the numbers or address, but to help them run through a brief script so they know what to say and would be comfortable saying in their own words (but effectively saying, “I really need to speak with someone right now, I’m not safe.”)
Helping youth understand the advantages and disadvantages of reaching out to peers, and when and how to reach out to adults, can be a constructive conversation. It is a chance not only to speak with and hear about a youth’s life and relationships but also a chance to give them a stronger and safer support network.
Dr. Henderson is a psychiatrist who treats children and adolescents at NYU Langone Health, New York.
Case: You are talking with one of your teenage patients, who has a history of significant suicidal ideation and an aborted attempt, and you ask her if there is someone she can talk with if she is feeling suicidal. “I call a friend,” she says. “That’s the only thing that works when I’m feeling bad.”
During difficult times, it is important to have a repertoire of coping skills to address stress, tension, frustration, anxiety, anger, sadness, and to help avoid dangerous behaviors. It is also important to have someone to talk to. For many youth, talking with friends is their preferred coping skill and contact when struggling with intense feelings.
This is hardly surprising. Peer relations are central to adolescent development. The ongoing individuation-separation process means that adolescents are peeling away from the family and into a community of their peers, where they figure out who they are through social interactions in subtle and complex ways. Adolescents are often profoundly immersed in the world of their peers; they often spend more time with their peers in educational and social settings than with their parents or other adults; and their connections with peers are often pleasurable, engaging, supportive, and intense. It is natural that they would want to communicate with their peers during stressful times.
At the same time, they may also want to avoid talking with adults. They may identify adult figures with authority, expectations, and control. So much adolescent psychic suffering and so many mental health crises involve shame, guilt, and fear, and are associated with romance, love, disappointment, and trauma – all of which may be difficult to share with parents and adult figures.
Adults also struggle with these kinds of conversations. Even benign attempts at comforting the youth (“Don’t worry, it’ll get better,” “Everyone feels this way sometimes”) can be seen as invalidating. And in stressful times, a difficult conversation can be ignited by the fuel of adult anxieties about the independence and autonomy of the child that is coming, which can make charged conversations all the more inflammatory.
Reaching out to peers during stressful times is therefore developmentally appropriate and often feels far more comfortable, validating, and sympathetic.
One of the most important things we can do is to help kids understand when, how, and why they can support each other – and when they cannot. Whether we like it or not, for many youth, peers are peer mental health counselors. They have shared vocabularies and can share experiences in the mental health care system. In addition to relying on their peers, a great many youth we work with also see themselves as supports to their peers, so it’s not just a one-way street.
So we talk with them frankly about when, how, and why talking with their friends can be an effective way of getting through a hard time and when, how, and why they need to reach out to an adult.
Recognizing how positive peer support can be, we ask them to identify problems with it. Kids often recognize the drawbacks of relying on their peers for support. They can see how it can be a burden to their friends. They often acknowledge that their friends may be experts in some aspects of their lives but not in others. For example, they can have shared stressors in school, can have similar understandings of the drama in their lives, and can relate to each other’s worlds, but will also not necessarily know what to do if a situation becomes dangerous.
The youth also tend to understand that the stakes in these conversations are high. We have seen peer groups suffer terribly when the youth have felt responsible – and even been the last preceding contact – in bad or even fatal outcomes.
We need to open up conversations about different forms of communication: when teens need understanding, compassion, patience; when they need a good understanding of local, cultural contexts, and a sense of support without anxieties and stressors; and when they need support and adult capacities and connections to solve problems. We can help them understand how to access people – both peers and adults – but also discuss responsibility: who you are responsible for, how you cannot be responsible alone for your friends’ mental health, how they cannot be responsible for yours, and who can be responsible for you.
To this end, we validate the importance of peers and ask more specifically when the adolescent thinks it is helpful to contact peers and when they think it would not be helpful. Having teens explain the difference may help them identify the right times to connect with peers or adults.
We can then talk about how to understand that there are different kinds of crisis: the kind where comfort, understanding, and support from friends can alleviate the crisis, and times when it is imperative to involve adults.
We can then identify which adults in their lives they can contact and how they would do so, both in terms of method of communication (texting an older sister, speaking in person with a parent, calling a therapist) and what they could say.
Then comes a more difficult step. We help them think about how to identify adults whom they do not know: how to contact a hotline or go to an emergency room or call 911. It is important not just to provide the numbers or address, but to help them run through a brief script so they know what to say and would be comfortable saying in their own words (but effectively saying, “I really need to speak with someone right now, I’m not safe.”)
Helping youth understand the advantages and disadvantages of reaching out to peers, and when and how to reach out to adults, can be a constructive conversation. It is a chance not only to speak with and hear about a youth’s life and relationships but also a chance to give them a stronger and safer support network.
Dr. Henderson is a psychiatrist who treats children and adolescents at NYU Langone Health, New York.
HPV vaccine effectiveness dependent on age at receipt
The effectiveness of the human papillomavirus (HPV) vaccine against HPV types 16 and 18 is highly dependent on the age at which it is given. Prevalence rates have been shown to be significantly lower among girls who are vaccinated at the recommended ages of 9-12 years, compared with those who are vaccinated after their sexual debut, data from the National Health and Nutrition Examination Survey (NHANES) indicate.
“HPV vaccination does not have any therapeutic effect on HPV infections already acquired, which is more likely to explain the difference in prevalence between predebut versus postdebut recipients than a lower immune response [among older recipients],” lead study author Didem Egemen, PhD, National Cancer Institute, Rockville, Md., told this news organization in an email.
“Still, among older females, the immune response of the vaccine is likely to still be quite strong, and we would encourage vaccination [of female patients] if unvaccinated, as our paper showed that vaccination post debut will still reduce HPV 16/18 prevalence by half,” she added.
The research letter was published online in JAMA Network Open.
National sample evaluated
Using data from NHANES, a biennial, cross-sectional sample (cycles 2011 through 2018), the researchers identified female persons who were aged 26 years or younger in 2006, when HPV vaccination was introduced, and who were eligible for routine vaccination or “catch-up” vaccination (given between the ages of 13 and 26 years), as per recommendations from the Advisory Committee on Immunization Practices. The investigators then compared the prevalence of HPV types 16 and 18 among unvaccinated female patients, female patients who had been vaccinated prior to their sexual debut (predebut group), and those who had been vaccinated after their sexual debut (postdebut group).
They also estimated vaccine uptake among those who were eligible for routine vaccination, as well as the proportion of vaccinated female patients with respect to racial and ethnic subgroups.
In the overall cohort, the prevalence of HPV types 16 and 18 decreased by 6% (95% confidence interval, 4%-7%) in the unvaccinated group to 3% (95% CI, 1%-6%) in the postdebut group and to less than 1% (95% CI, <1%-1%) in the predebut group, Dr. Egemen and colleagues report.
In real percentages, the prevalence of HPV 16 and 18 was 89% lower in the predebut group (P < .001) but only 41% lower in the postdebut group (P = .29) compared with unvaccinated female patients. And compared with female patients who were vaccinated after their sexual debut, the prevalence of HPV 16 and 18 was reduced by 82% among those who had received the vaccine at the recommended ages of 9-12 years (P = .08).
In the current study, Dr. Egeman acknowledged that only 38% of ever-eligible female patients received the vaccine, although the prevalence increased to 56% when only female patients who were eligible for routine vaccination were taken into account. On the other hand, only 21% (95% CI, 14%-28%) of female patients eligible for routine vaccination received their first dose by age 12 years.
Indeed, the mean age on receipt of the first vaccination dose was 14.5 years (95% CI, 14.1-14.8 years), the authors note, and only 59% of girls received their first dose prior to their sexual debut. Additionally, among routine vaccination–eligible girls aged 12 years or younger in 2006, 33% were vaccinated before and 23% after their sexual debut, and the rest were not vaccinated.
Interestingly, differences in the age at which the HPV vaccine was received by race and ethnicity were negligible, the investigators point out.
Vaccination rates increasing
Asked to comment on the findings, Rebecca Perkins, MD, professor of obstetrics and gynecology at Boston University, Boston Medical Center, pointed out that the investigators evaluated data from 2011 to 2018. “We know that HPV vaccination rates have increased over that period and continue to increase,” she emphasized in an email to this news organization.
Physicians also know that more persons are being vaccinated between the ages of 9 and 12 than was the case at the beginning of this study. “This is good news,” she said, “as it means that more adolescents now in 2022 are benefiting fully from vaccination than they were in 2011,” she added.
At the same time, Dr. Perkins acknowledged that many persons are still missing out on the chance to receive the vaccine on time – which means they are missing out on the chance to prevent cancer.
“Making sure that all adolescents receive vaccination between the ages of 9 to 12 has the potential to prevent up to 40,000 cancers every year in the U.S., [including] the most common HPV-related cancers, such as cervical cancer in women and tongue and tonsillar cancer in men,” Dr. Perkins noted.
“Thus, it’s critical that doctors and parents get the message that you can’t vaccinate too early, only too late,” she emphasized.
Dr. Edgman and Dr. Perkins report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The effectiveness of the human papillomavirus (HPV) vaccine against HPV types 16 and 18 is highly dependent on the age at which it is given. Prevalence rates have been shown to be significantly lower among girls who are vaccinated at the recommended ages of 9-12 years, compared with those who are vaccinated after their sexual debut, data from the National Health and Nutrition Examination Survey (NHANES) indicate.
“HPV vaccination does not have any therapeutic effect on HPV infections already acquired, which is more likely to explain the difference in prevalence between predebut versus postdebut recipients than a lower immune response [among older recipients],” lead study author Didem Egemen, PhD, National Cancer Institute, Rockville, Md., told this news organization in an email.
“Still, among older females, the immune response of the vaccine is likely to still be quite strong, and we would encourage vaccination [of female patients] if unvaccinated, as our paper showed that vaccination post debut will still reduce HPV 16/18 prevalence by half,” she added.
The research letter was published online in JAMA Network Open.
National sample evaluated
Using data from NHANES, a biennial, cross-sectional sample (cycles 2011 through 2018), the researchers identified female persons who were aged 26 years or younger in 2006, when HPV vaccination was introduced, and who were eligible for routine vaccination or “catch-up” vaccination (given between the ages of 13 and 26 years), as per recommendations from the Advisory Committee on Immunization Practices. The investigators then compared the prevalence of HPV types 16 and 18 among unvaccinated female patients, female patients who had been vaccinated prior to their sexual debut (predebut group), and those who had been vaccinated after their sexual debut (postdebut group).
They also estimated vaccine uptake among those who were eligible for routine vaccination, as well as the proportion of vaccinated female patients with respect to racial and ethnic subgroups.
In the overall cohort, the prevalence of HPV types 16 and 18 decreased by 6% (95% confidence interval, 4%-7%) in the unvaccinated group to 3% (95% CI, 1%-6%) in the postdebut group and to less than 1% (95% CI, <1%-1%) in the predebut group, Dr. Egemen and colleagues report.
In real percentages, the prevalence of HPV 16 and 18 was 89% lower in the predebut group (P < .001) but only 41% lower in the postdebut group (P = .29) compared with unvaccinated female patients. And compared with female patients who were vaccinated after their sexual debut, the prevalence of HPV 16 and 18 was reduced by 82% among those who had received the vaccine at the recommended ages of 9-12 years (P = .08).
In the current study, Dr. Egeman acknowledged that only 38% of ever-eligible female patients received the vaccine, although the prevalence increased to 56% when only female patients who were eligible for routine vaccination were taken into account. On the other hand, only 21% (95% CI, 14%-28%) of female patients eligible for routine vaccination received their first dose by age 12 years.
Indeed, the mean age on receipt of the first vaccination dose was 14.5 years (95% CI, 14.1-14.8 years), the authors note, and only 59% of girls received their first dose prior to their sexual debut. Additionally, among routine vaccination–eligible girls aged 12 years or younger in 2006, 33% were vaccinated before and 23% after their sexual debut, and the rest were not vaccinated.
Interestingly, differences in the age at which the HPV vaccine was received by race and ethnicity were negligible, the investigators point out.
Vaccination rates increasing
Asked to comment on the findings, Rebecca Perkins, MD, professor of obstetrics and gynecology at Boston University, Boston Medical Center, pointed out that the investigators evaluated data from 2011 to 2018. “We know that HPV vaccination rates have increased over that period and continue to increase,” she emphasized in an email to this news organization.
Physicians also know that more persons are being vaccinated between the ages of 9 and 12 than was the case at the beginning of this study. “This is good news,” she said, “as it means that more adolescents now in 2022 are benefiting fully from vaccination than they were in 2011,” she added.
At the same time, Dr. Perkins acknowledged that many persons are still missing out on the chance to receive the vaccine on time – which means they are missing out on the chance to prevent cancer.
“Making sure that all adolescents receive vaccination between the ages of 9 to 12 has the potential to prevent up to 40,000 cancers every year in the U.S., [including] the most common HPV-related cancers, such as cervical cancer in women and tongue and tonsillar cancer in men,” Dr. Perkins noted.
“Thus, it’s critical that doctors and parents get the message that you can’t vaccinate too early, only too late,” she emphasized.
Dr. Edgman and Dr. Perkins report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The effectiveness of the human papillomavirus (HPV) vaccine against HPV types 16 and 18 is highly dependent on the age at which it is given. Prevalence rates have been shown to be significantly lower among girls who are vaccinated at the recommended ages of 9-12 years, compared with those who are vaccinated after their sexual debut, data from the National Health and Nutrition Examination Survey (NHANES) indicate.
“HPV vaccination does not have any therapeutic effect on HPV infections already acquired, which is more likely to explain the difference in prevalence between predebut versus postdebut recipients than a lower immune response [among older recipients],” lead study author Didem Egemen, PhD, National Cancer Institute, Rockville, Md., told this news organization in an email.
“Still, among older females, the immune response of the vaccine is likely to still be quite strong, and we would encourage vaccination [of female patients] if unvaccinated, as our paper showed that vaccination post debut will still reduce HPV 16/18 prevalence by half,” she added.
The research letter was published online in JAMA Network Open.
National sample evaluated
Using data from NHANES, a biennial, cross-sectional sample (cycles 2011 through 2018), the researchers identified female persons who were aged 26 years or younger in 2006, when HPV vaccination was introduced, and who were eligible for routine vaccination or “catch-up” vaccination (given between the ages of 13 and 26 years), as per recommendations from the Advisory Committee on Immunization Practices. The investigators then compared the prevalence of HPV types 16 and 18 among unvaccinated female patients, female patients who had been vaccinated prior to their sexual debut (predebut group), and those who had been vaccinated after their sexual debut (postdebut group).
They also estimated vaccine uptake among those who were eligible for routine vaccination, as well as the proportion of vaccinated female patients with respect to racial and ethnic subgroups.
In the overall cohort, the prevalence of HPV types 16 and 18 decreased by 6% (95% confidence interval, 4%-7%) in the unvaccinated group to 3% (95% CI, 1%-6%) in the postdebut group and to less than 1% (95% CI, <1%-1%) in the predebut group, Dr. Egemen and colleagues report.
In real percentages, the prevalence of HPV 16 and 18 was 89% lower in the predebut group (P < .001) but only 41% lower in the postdebut group (P = .29) compared with unvaccinated female patients. And compared with female patients who were vaccinated after their sexual debut, the prevalence of HPV 16 and 18 was reduced by 82% among those who had received the vaccine at the recommended ages of 9-12 years (P = .08).
In the current study, Dr. Egeman acknowledged that only 38% of ever-eligible female patients received the vaccine, although the prevalence increased to 56% when only female patients who were eligible for routine vaccination were taken into account. On the other hand, only 21% (95% CI, 14%-28%) of female patients eligible for routine vaccination received their first dose by age 12 years.
Indeed, the mean age on receipt of the first vaccination dose was 14.5 years (95% CI, 14.1-14.8 years), the authors note, and only 59% of girls received their first dose prior to their sexual debut. Additionally, among routine vaccination–eligible girls aged 12 years or younger in 2006, 33% were vaccinated before and 23% after their sexual debut, and the rest were not vaccinated.
Interestingly, differences in the age at which the HPV vaccine was received by race and ethnicity were negligible, the investigators point out.
Vaccination rates increasing
Asked to comment on the findings, Rebecca Perkins, MD, professor of obstetrics and gynecology at Boston University, Boston Medical Center, pointed out that the investigators evaluated data from 2011 to 2018. “We know that HPV vaccination rates have increased over that period and continue to increase,” she emphasized in an email to this news organization.
Physicians also know that more persons are being vaccinated between the ages of 9 and 12 than was the case at the beginning of this study. “This is good news,” she said, “as it means that more adolescents now in 2022 are benefiting fully from vaccination than they were in 2011,” she added.
At the same time, Dr. Perkins acknowledged that many persons are still missing out on the chance to receive the vaccine on time – which means they are missing out on the chance to prevent cancer.
“Making sure that all adolescents receive vaccination between the ages of 9 to 12 has the potential to prevent up to 40,000 cancers every year in the U.S., [including] the most common HPV-related cancers, such as cervical cancer in women and tongue and tonsillar cancer in men,” Dr. Perkins noted.
“Thus, it’s critical that doctors and parents get the message that you can’t vaccinate too early, only too late,” she emphasized.
Dr. Edgman and Dr. Perkins report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
CDC warns of early uptick in respiratory disease
The Centers for Disease Control and Prevention is warning of an early surge in respiratory disease caused by multiple viruses. As influenza viruses, respiratory syncytial virus (RSV), SARS-CoV-2, and rhinovirus/enterovirus simultaneously circulate, the agency cautioned that this confluence of viral activity could strain the health care system, according to a CDC Health Network Alert advisory issued Nov. 4.
“This early increase in disease incidence highlights the importance of optimizing respiratory virus prevention and treatment measures, including prompt vaccination and antiviral treatment,” the alert stated.
The CDC reports that RSV activity is increasing nationally, but in some areas – such as the South and Mountain West – cases appear to be trending downward.
Influenza cases continue to climb, with the virus activity being the highest in the South, Mid-Atlantic, and the south-central West Coast, according to CDC data. “In fact, we’re seeing the highest influenza hospitalization rates going back a decade,” said José Romero, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing. The agency estimates that there have been 1.6 million illnesses, 13,000 hospitalizations, and 730 deaths from the flu so far this season. As of Nov. 4, there have been two pediatric deaths.
COVID-19 cases appear to have plateaued in the past three weeks, Dr. Romero said; however, the CDC expects that there will be “high-level circulation of SARS-CoV-2 this fall and winter,” the health alert stated.
The CDC advised that all eligible individuals aged 6-months or older should be vaccinated against COVID-19 and influenza. To protect against RSV-hospitalization, high-risk children should receive the monoclonal antibody drug palivizumab (Synagis). High-risk children include infants born before 29 weeks, children younger than age 2 with chronic lung disease or hemodynamically significant congenital heart disease, and children with suppressed immune systems or neuromuscular disorders.
Any patient with confirmed or suspected flu who is hospitalized, at higher risk for influenza complications, or who has a severe or progressive illness should be treated as early as possible with antivirals, such as oral oseltamivir (Tamiflu).
Patients with confirmed SARS-CoV-2 infection with increased risk of complications should also be treated with antivirals, such as nirmatrelvir and ritonavir (Paxlovid) or remdesivir (Veklury).
Patients should also be reminded to wash their hands frequently, cover coughs and sneezes, stay home when sick, and avoid close contact with people who are sick, the CDC advised.
“There’s no doubt that we will face some challenges this winter,” said Dawn O’Connell, HHS Assistant Secretary for Preparedness and Response, “but it’s important to remember that RSV and flu are not new, and we have safe and effective vaccines for COVID-19 and the flu.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is warning of an early surge in respiratory disease caused by multiple viruses. As influenza viruses, respiratory syncytial virus (RSV), SARS-CoV-2, and rhinovirus/enterovirus simultaneously circulate, the agency cautioned that this confluence of viral activity could strain the health care system, according to a CDC Health Network Alert advisory issued Nov. 4.
“This early increase in disease incidence highlights the importance of optimizing respiratory virus prevention and treatment measures, including prompt vaccination and antiviral treatment,” the alert stated.
The CDC reports that RSV activity is increasing nationally, but in some areas – such as the South and Mountain West – cases appear to be trending downward.
Influenza cases continue to climb, with the virus activity being the highest in the South, Mid-Atlantic, and the south-central West Coast, according to CDC data. “In fact, we’re seeing the highest influenza hospitalization rates going back a decade,” said José Romero, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing. The agency estimates that there have been 1.6 million illnesses, 13,000 hospitalizations, and 730 deaths from the flu so far this season. As of Nov. 4, there have been two pediatric deaths.
COVID-19 cases appear to have plateaued in the past three weeks, Dr. Romero said; however, the CDC expects that there will be “high-level circulation of SARS-CoV-2 this fall and winter,” the health alert stated.
The CDC advised that all eligible individuals aged 6-months or older should be vaccinated against COVID-19 and influenza. To protect against RSV-hospitalization, high-risk children should receive the monoclonal antibody drug palivizumab (Synagis). High-risk children include infants born before 29 weeks, children younger than age 2 with chronic lung disease or hemodynamically significant congenital heart disease, and children with suppressed immune systems or neuromuscular disorders.
Any patient with confirmed or suspected flu who is hospitalized, at higher risk for influenza complications, or who has a severe or progressive illness should be treated as early as possible with antivirals, such as oral oseltamivir (Tamiflu).
Patients with confirmed SARS-CoV-2 infection with increased risk of complications should also be treated with antivirals, such as nirmatrelvir and ritonavir (Paxlovid) or remdesivir (Veklury).
Patients should also be reminded to wash their hands frequently, cover coughs and sneezes, stay home when sick, and avoid close contact with people who are sick, the CDC advised.
“There’s no doubt that we will face some challenges this winter,” said Dawn O’Connell, HHS Assistant Secretary for Preparedness and Response, “but it’s important to remember that RSV and flu are not new, and we have safe and effective vaccines for COVID-19 and the flu.”
A version of this article first appeared on Medscape.com.
The Centers for Disease Control and Prevention is warning of an early surge in respiratory disease caused by multiple viruses. As influenza viruses, respiratory syncytial virus (RSV), SARS-CoV-2, and rhinovirus/enterovirus simultaneously circulate, the agency cautioned that this confluence of viral activity could strain the health care system, according to a CDC Health Network Alert advisory issued Nov. 4.
“This early increase in disease incidence highlights the importance of optimizing respiratory virus prevention and treatment measures, including prompt vaccination and antiviral treatment,” the alert stated.
The CDC reports that RSV activity is increasing nationally, but in some areas – such as the South and Mountain West – cases appear to be trending downward.
Influenza cases continue to climb, with the virus activity being the highest in the South, Mid-Atlantic, and the south-central West Coast, according to CDC data. “In fact, we’re seeing the highest influenza hospitalization rates going back a decade,” said José Romero, MD, director of the CDC’s National Center for Immunization and Respiratory Diseases, during a press briefing. The agency estimates that there have been 1.6 million illnesses, 13,000 hospitalizations, and 730 deaths from the flu so far this season. As of Nov. 4, there have been two pediatric deaths.
COVID-19 cases appear to have plateaued in the past three weeks, Dr. Romero said; however, the CDC expects that there will be “high-level circulation of SARS-CoV-2 this fall and winter,” the health alert stated.
The CDC advised that all eligible individuals aged 6-months or older should be vaccinated against COVID-19 and influenza. To protect against RSV-hospitalization, high-risk children should receive the monoclonal antibody drug palivizumab (Synagis). High-risk children include infants born before 29 weeks, children younger than age 2 with chronic lung disease or hemodynamically significant congenital heart disease, and children with suppressed immune systems or neuromuscular disorders.
Any patient with confirmed or suspected flu who is hospitalized, at higher risk for influenza complications, or who has a severe or progressive illness should be treated as early as possible with antivirals, such as oral oseltamivir (Tamiflu).
Patients with confirmed SARS-CoV-2 infection with increased risk of complications should also be treated with antivirals, such as nirmatrelvir and ritonavir (Paxlovid) or remdesivir (Veklury).
Patients should also be reminded to wash their hands frequently, cover coughs and sneezes, stay home when sick, and avoid close contact with people who are sick, the CDC advised.
“There’s no doubt that we will face some challenges this winter,” said Dawn O’Connell, HHS Assistant Secretary for Preparedness and Response, “but it’s important to remember that RSV and flu are not new, and we have safe and effective vaccines for COVID-19 and the flu.”
A version of this article first appeared on Medscape.com.
COVID bivalent booster better vs. recent Omicron subvariants: Pfizer
the company reported on Nov. 4, supporting calls by public health officials for eligible people to get this booster before a potential COVID-19 surge this winter.
The company’s ongoing phase 2/3 study of their Omicron BA.4 and BA.5 bivalent – which targets both the virus’ original strain and the two subvariants – shows that the vaccine offered the strongest protection in people older than 55 years.
One month after receiving a 30-mcg booster with the bivalent vaccine, those older than 55 had four times more neutralizing antibodies against these Omicron subvariants, compared with people who received the original monovalent vaccine as a booster in the study.
Researchers compared the geometric mean titer (GMT) levels of these antibodies in three groups before and 1 month after boosting. The 36 people older than 55 years in the released study findings had an GMT level of 896 with the bivalent booster, a level 13 times higher than before this immunization.
For the 38 adults ages 18-55 in the study, the GMT level increased to 606 at 1 month after the bivalent booster, an increase of almost 10-fold, compared with baseline. In a comparator group of 40 people receiving the original vaccine as a fourth dose, the GMT level was 236, or threefold higher than before their booster shot.
The newly released data is “very encouraging and consistent now with three studies all showing a substantial 3-4 fold increased level of neutralizing antibodies versus BA.5 as compared with the original booster,” said Eric Topol, MD, director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News.
Pfizer and BioNTech announced the updated findings in a Nov. 4 press release.
A booster dose of the BA.4/BA.5-adapted bivalent vaccine is authorized for emergency use by the Food and Drug Administration for ages 5 years and older. The safety and tolerability profile of the Pfizer/BioNTech bivalent booster remains favorable and similar to the original COVID-19 vaccine, the company reported.
Until recently, the BA.5 Omicron variant was the dominant strain in the United States, but is now getting elbowed out by the subvariants BQ.1.1, BQ.1, and BA.4.6, which together make up almost 45% of the circulating virus.
Some skepticism
“It is important to note that these data are press-release level, which does not allow a view of the data totality,” Hana El Sahly, MD, professor of molecular virology and microbiology, Baylor College of Medicine, Houston, said in an interview.
“For example, there may be significant differences between the groups, and the release mentions at least one difference that is of importance: the interval since the last vaccination which often affects the response to subsequent boosting,” she said.
Dr. El Sahly added that the findings are not surprising. “In the short term, a variant-specific vaccine produces a higher level of antibody against the variant in the vaccine than the vaccines based on the ancestral strains.”
More researcher results are warranted. “These data do not indicate that these differences between the two vaccines translate into a meaningful clinical benefit at a population level,” Dr. El Sahly said.
An uncertain winter ahead
“As we head into the holiday season, we hope these updated data will encourage people to seek out a COVID-19 bivalent booster as soon as they are eligible in order to maintain high levels of protection against the widely circulating Omicron BA.4 and BA.5 sublineages,” Albert Bourla, Pfizer chairman and CEO, stated in the release.
The updated data from the Pfizer/BioNTech study are “all the more reason to get a booster, with added protection also versus BQ.1.1, which will soon become dominant in the U.S.,” Dr. Topol predicted.
It is unclear when the next surge will happen, as COVID-19 does not always follow a seasonal pattern, at least not yet, Dr. El Sahly said. “Regardless, it is reasonable to recommend additional vaccine doses to immunocompromised and frail or older persons. More importantly, influenza vaccination and being up to date on pneumococcal vaccines are highly recommended as soon as feasible, given the early and intense flu season.”
A version of this article first appeared on Medscape.com.
the company reported on Nov. 4, supporting calls by public health officials for eligible people to get this booster before a potential COVID-19 surge this winter.
The company’s ongoing phase 2/3 study of their Omicron BA.4 and BA.5 bivalent – which targets both the virus’ original strain and the two subvariants – shows that the vaccine offered the strongest protection in people older than 55 years.
One month after receiving a 30-mcg booster with the bivalent vaccine, those older than 55 had four times more neutralizing antibodies against these Omicron subvariants, compared with people who received the original monovalent vaccine as a booster in the study.
Researchers compared the geometric mean titer (GMT) levels of these antibodies in three groups before and 1 month after boosting. The 36 people older than 55 years in the released study findings had an GMT level of 896 with the bivalent booster, a level 13 times higher than before this immunization.
For the 38 adults ages 18-55 in the study, the GMT level increased to 606 at 1 month after the bivalent booster, an increase of almost 10-fold, compared with baseline. In a comparator group of 40 people receiving the original vaccine as a fourth dose, the GMT level was 236, or threefold higher than before their booster shot.
The newly released data is “very encouraging and consistent now with three studies all showing a substantial 3-4 fold increased level of neutralizing antibodies versus BA.5 as compared with the original booster,” said Eric Topol, MD, director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News.
Pfizer and BioNTech announced the updated findings in a Nov. 4 press release.
A booster dose of the BA.4/BA.5-adapted bivalent vaccine is authorized for emergency use by the Food and Drug Administration for ages 5 years and older. The safety and tolerability profile of the Pfizer/BioNTech bivalent booster remains favorable and similar to the original COVID-19 vaccine, the company reported.
Until recently, the BA.5 Omicron variant was the dominant strain in the United States, but is now getting elbowed out by the subvariants BQ.1.1, BQ.1, and BA.4.6, which together make up almost 45% of the circulating virus.
Some skepticism
“It is important to note that these data are press-release level, which does not allow a view of the data totality,” Hana El Sahly, MD, professor of molecular virology and microbiology, Baylor College of Medicine, Houston, said in an interview.
“For example, there may be significant differences between the groups, and the release mentions at least one difference that is of importance: the interval since the last vaccination which often affects the response to subsequent boosting,” she said.
Dr. El Sahly added that the findings are not surprising. “In the short term, a variant-specific vaccine produces a higher level of antibody against the variant in the vaccine than the vaccines based on the ancestral strains.”
More researcher results are warranted. “These data do not indicate that these differences between the two vaccines translate into a meaningful clinical benefit at a population level,” Dr. El Sahly said.
An uncertain winter ahead
“As we head into the holiday season, we hope these updated data will encourage people to seek out a COVID-19 bivalent booster as soon as they are eligible in order to maintain high levels of protection against the widely circulating Omicron BA.4 and BA.5 sublineages,” Albert Bourla, Pfizer chairman and CEO, stated in the release.
The updated data from the Pfizer/BioNTech study are “all the more reason to get a booster, with added protection also versus BQ.1.1, which will soon become dominant in the U.S.,” Dr. Topol predicted.
It is unclear when the next surge will happen, as COVID-19 does not always follow a seasonal pattern, at least not yet, Dr. El Sahly said. “Regardless, it is reasonable to recommend additional vaccine doses to immunocompromised and frail or older persons. More importantly, influenza vaccination and being up to date on pneumococcal vaccines are highly recommended as soon as feasible, given the early and intense flu season.”
A version of this article first appeared on Medscape.com.
the company reported on Nov. 4, supporting calls by public health officials for eligible people to get this booster before a potential COVID-19 surge this winter.
The company’s ongoing phase 2/3 study of their Omicron BA.4 and BA.5 bivalent – which targets both the virus’ original strain and the two subvariants – shows that the vaccine offered the strongest protection in people older than 55 years.
One month after receiving a 30-mcg booster with the bivalent vaccine, those older than 55 had four times more neutralizing antibodies against these Omicron subvariants, compared with people who received the original monovalent vaccine as a booster in the study.
Researchers compared the geometric mean titer (GMT) levels of these antibodies in three groups before and 1 month after boosting. The 36 people older than 55 years in the released study findings had an GMT level of 896 with the bivalent booster, a level 13 times higher than before this immunization.
For the 38 adults ages 18-55 in the study, the GMT level increased to 606 at 1 month after the bivalent booster, an increase of almost 10-fold, compared with baseline. In a comparator group of 40 people receiving the original vaccine as a fourth dose, the GMT level was 236, or threefold higher than before their booster shot.
The newly released data is “very encouraging and consistent now with three studies all showing a substantial 3-4 fold increased level of neutralizing antibodies versus BA.5 as compared with the original booster,” said Eric Topol, MD, director of the Scripps Research Translational Institute in La Jolla, Calif., and editor-in-chief of Medscape Medical News.
Pfizer and BioNTech announced the updated findings in a Nov. 4 press release.
A booster dose of the BA.4/BA.5-adapted bivalent vaccine is authorized for emergency use by the Food and Drug Administration for ages 5 years and older. The safety and tolerability profile of the Pfizer/BioNTech bivalent booster remains favorable and similar to the original COVID-19 vaccine, the company reported.
Until recently, the BA.5 Omicron variant was the dominant strain in the United States, but is now getting elbowed out by the subvariants BQ.1.1, BQ.1, and BA.4.6, which together make up almost 45% of the circulating virus.
Some skepticism
“It is important to note that these data are press-release level, which does not allow a view of the data totality,” Hana El Sahly, MD, professor of molecular virology and microbiology, Baylor College of Medicine, Houston, said in an interview.
“For example, there may be significant differences between the groups, and the release mentions at least one difference that is of importance: the interval since the last vaccination which often affects the response to subsequent boosting,” she said.
Dr. El Sahly added that the findings are not surprising. “In the short term, a variant-specific vaccine produces a higher level of antibody against the variant in the vaccine than the vaccines based on the ancestral strains.”
More researcher results are warranted. “These data do not indicate that these differences between the two vaccines translate into a meaningful clinical benefit at a population level,” Dr. El Sahly said.
An uncertain winter ahead
“As we head into the holiday season, we hope these updated data will encourage people to seek out a COVID-19 bivalent booster as soon as they are eligible in order to maintain high levels of protection against the widely circulating Omicron BA.4 and BA.5 sublineages,” Albert Bourla, Pfizer chairman and CEO, stated in the release.
The updated data from the Pfizer/BioNTech study are “all the more reason to get a booster, with added protection also versus BQ.1.1, which will soon become dominant in the U.S.,” Dr. Topol predicted.
It is unclear when the next surge will happen, as COVID-19 does not always follow a seasonal pattern, at least not yet, Dr. El Sahly said. “Regardless, it is reasonable to recommend additional vaccine doses to immunocompromised and frail or older persons. More importantly, influenza vaccination and being up to date on pneumococcal vaccines are highly recommended as soon as feasible, given the early and intense flu season.”
A version of this article first appeared on Medscape.com.
FDA expands tenofovir alafenamide (Vemlidy) use to adolescents with chronic HBV
the drug’s manufacturer has announced.
The approval in the pediatric patient population was supported by 24-week data from a phase 2 clinical trial comparing treatment with tenofovir alafenamide (25 mg once daily) with placebo in 70 treatment-naive and treatment-experienced patients aged 12-18 years weighing at least 35 kg.
The study met its primary endpoint of percentage of patients with HBV DNA levels less than 20 IU/mL at 24 weeks of therapy, Gilead Sciences said in a press release.
Overall, 10 of 47 (21%) patients treated with tenofovir alafenamide achieved HBV DNA less than 20 IU/mL at 24 weeks, compared with 0 of 23 (0%) treated with placebo.
The rates of serum ALT normalization were higher with tenofovir alafenamide than with placebo (44% vs 0%).
The mean percent changes in bone mineral density (BMD) from baseline to 24 weeks were numerically similar for tenofovir alafenamide– and placebo-treated patients (2.4% and 1.9% for lumbar spine, and 1.5% and 1.9% for whole body, respectively).
The mean changes from baseline BMD z scores were –0.03 and –0.09 for lumbar spine, and –0.05 and –0.01 for whole body, for tenofovir alafenamide and placebo groups, respectively.
The FDA initially approved the nucleoside analog reverse transcriptase inhibitor in 2016 for adults with chronic HBV.
The drug was approved in Europe in 2017 for chronic HBV infection in adults and adolescents aged 12 years and older weighing at least 35 kg.
Tenofovir alafenamide carries a boxed warning citing risks for lactic acidosis/severe hepatomegaly with steatosis and posttreatment severe acute exacerbation of HBV.
A version of this article first appeared on Medscape.com.
the drug’s manufacturer has announced.
The approval in the pediatric patient population was supported by 24-week data from a phase 2 clinical trial comparing treatment with tenofovir alafenamide (25 mg once daily) with placebo in 70 treatment-naive and treatment-experienced patients aged 12-18 years weighing at least 35 kg.
The study met its primary endpoint of percentage of patients with HBV DNA levels less than 20 IU/mL at 24 weeks of therapy, Gilead Sciences said in a press release.
Overall, 10 of 47 (21%) patients treated with tenofovir alafenamide achieved HBV DNA less than 20 IU/mL at 24 weeks, compared with 0 of 23 (0%) treated with placebo.
The rates of serum ALT normalization were higher with tenofovir alafenamide than with placebo (44% vs 0%).
The mean percent changes in bone mineral density (BMD) from baseline to 24 weeks were numerically similar for tenofovir alafenamide– and placebo-treated patients (2.4% and 1.9% for lumbar spine, and 1.5% and 1.9% for whole body, respectively).
The mean changes from baseline BMD z scores were –0.03 and –0.09 for lumbar spine, and –0.05 and –0.01 for whole body, for tenofovir alafenamide and placebo groups, respectively.
The FDA initially approved the nucleoside analog reverse transcriptase inhibitor in 2016 for adults with chronic HBV.
The drug was approved in Europe in 2017 for chronic HBV infection in adults and adolescents aged 12 years and older weighing at least 35 kg.
Tenofovir alafenamide carries a boxed warning citing risks for lactic acidosis/severe hepatomegaly with steatosis and posttreatment severe acute exacerbation of HBV.
A version of this article first appeared on Medscape.com.
the drug’s manufacturer has announced.
The approval in the pediatric patient population was supported by 24-week data from a phase 2 clinical trial comparing treatment with tenofovir alafenamide (25 mg once daily) with placebo in 70 treatment-naive and treatment-experienced patients aged 12-18 years weighing at least 35 kg.
The study met its primary endpoint of percentage of patients with HBV DNA levels less than 20 IU/mL at 24 weeks of therapy, Gilead Sciences said in a press release.
Overall, 10 of 47 (21%) patients treated with tenofovir alafenamide achieved HBV DNA less than 20 IU/mL at 24 weeks, compared with 0 of 23 (0%) treated with placebo.
The rates of serum ALT normalization were higher with tenofovir alafenamide than with placebo (44% vs 0%).
The mean percent changes in bone mineral density (BMD) from baseline to 24 weeks were numerically similar for tenofovir alafenamide– and placebo-treated patients (2.4% and 1.9% for lumbar spine, and 1.5% and 1.9% for whole body, respectively).
The mean changes from baseline BMD z scores were –0.03 and –0.09 for lumbar spine, and –0.05 and –0.01 for whole body, for tenofovir alafenamide and placebo groups, respectively.
The FDA initially approved the nucleoside analog reverse transcriptase inhibitor in 2016 for adults with chronic HBV.
The drug was approved in Europe in 2017 for chronic HBV infection in adults and adolescents aged 12 years and older weighing at least 35 kg.
Tenofovir alafenamide carries a boxed warning citing risks for lactic acidosis/severe hepatomegaly with steatosis and posttreatment severe acute exacerbation of HBV.
A version of this article first appeared on Medscape.com.

