User login
News and Views that Matter to Pediatricians
The leading independent newspaper covering news and commentary in pediatrics.
Diabetes surge expected in young people
according to a new study published in Diabetes Care.
It is expected that as many as 526,000 people younger than 20 years in the United States will have diabetes by 2060, researchers from the Centers for Disease Control and Prevention report. Their projections found that the number of young people with diabetes will increase 12%, from 213,000 in 2017 to 239,000 in 2060.
The estimates include a 673% rise in the number of youth with type 2 diabetes and a 65% increase in cases of type 1 diabetes over the next 4 decades.
Most of the new cases are projected to occur among non-Hispanic Blacks, exacerbating the already significant racial disparities in type 2 diabetes in particular, the study found.
“This study’s startling projections of type 2 diabetes increases show why it is crucial to advance health equity and reduce the widespread disparities that already take a toll on people’s health,” Christopher Holliday, PhD, MPH, FACHE, director of CDC’s Division of Diabetes Translation, said in a press release about the new estimates.
Even if trends remain the same in coming decades, researchers said diagnoses of type 2 diabetes will rise almost 70% and that diagnoses of type 1 diabetes will increase by 3%.
The researchers attribute the increase in diabetes cases among youth to a variety of factors, including the growing prevalence of childhood obesity and the presence of diabetes in women of childbearing age, which is linked to obesity in their offspring.
Debra Houry, MD, MPH, acting principal director of the CDC, said the focus should be on prevention.
“This new research should serve as a wake-up call for all of us. It’s vital that we focus our efforts to ensure all Americans, especially our young people, are the healthiest they can be,” she said in a press release.
The findings come from the SEARCH for Diabetes in Youth study, funded by the CDC and the National Institutes of Health. Dr. Houry and Dr. Holliday report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a new study published in Diabetes Care.
It is expected that as many as 526,000 people younger than 20 years in the United States will have diabetes by 2060, researchers from the Centers for Disease Control and Prevention report. Their projections found that the number of young people with diabetes will increase 12%, from 213,000 in 2017 to 239,000 in 2060.
The estimates include a 673% rise in the number of youth with type 2 diabetes and a 65% increase in cases of type 1 diabetes over the next 4 decades.
Most of the new cases are projected to occur among non-Hispanic Blacks, exacerbating the already significant racial disparities in type 2 diabetes in particular, the study found.
“This study’s startling projections of type 2 diabetes increases show why it is crucial to advance health equity and reduce the widespread disparities that already take a toll on people’s health,” Christopher Holliday, PhD, MPH, FACHE, director of CDC’s Division of Diabetes Translation, said in a press release about the new estimates.
Even if trends remain the same in coming decades, researchers said diagnoses of type 2 diabetes will rise almost 70% and that diagnoses of type 1 diabetes will increase by 3%.
The researchers attribute the increase in diabetes cases among youth to a variety of factors, including the growing prevalence of childhood obesity and the presence of diabetes in women of childbearing age, which is linked to obesity in their offspring.
Debra Houry, MD, MPH, acting principal director of the CDC, said the focus should be on prevention.
“This new research should serve as a wake-up call for all of us. It’s vital that we focus our efforts to ensure all Americans, especially our young people, are the healthiest they can be,” she said in a press release.
The findings come from the SEARCH for Diabetes in Youth study, funded by the CDC and the National Institutes of Health. Dr. Houry and Dr. Holliday report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a new study published in Diabetes Care.
It is expected that as many as 526,000 people younger than 20 years in the United States will have diabetes by 2060, researchers from the Centers for Disease Control and Prevention report. Their projections found that the number of young people with diabetes will increase 12%, from 213,000 in 2017 to 239,000 in 2060.
The estimates include a 673% rise in the number of youth with type 2 diabetes and a 65% increase in cases of type 1 diabetes over the next 4 decades.
Most of the new cases are projected to occur among non-Hispanic Blacks, exacerbating the already significant racial disparities in type 2 diabetes in particular, the study found.
“This study’s startling projections of type 2 diabetes increases show why it is crucial to advance health equity and reduce the widespread disparities that already take a toll on people’s health,” Christopher Holliday, PhD, MPH, FACHE, director of CDC’s Division of Diabetes Translation, said in a press release about the new estimates.
Even if trends remain the same in coming decades, researchers said diagnoses of type 2 diabetes will rise almost 70% and that diagnoses of type 1 diabetes will increase by 3%.
The researchers attribute the increase in diabetes cases among youth to a variety of factors, including the growing prevalence of childhood obesity and the presence of diabetes in women of childbearing age, which is linked to obesity in their offspring.
Debra Houry, MD, MPH, acting principal director of the CDC, said the focus should be on prevention.
“This new research should serve as a wake-up call for all of us. It’s vital that we focus our efforts to ensure all Americans, especially our young people, are the healthiest they can be,” she said in a press release.
The findings come from the SEARCH for Diabetes in Youth study, funded by the CDC and the National Institutes of Health. Dr. Houry and Dr. Holliday report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DIABETES CARE
Nearly 1,400% rise in young children ingesting cannabis edibles
according to a new analysis of data from poison control centers.
In 2017, centers received 207 reports of children aged 5 years and younger who ingested edible cannabis. In 2021, 3,054 such cases were reported, according to the study, which was published online in Pediatrics.
Many of the children experienced clinical effects, such as depression of the central nervous system, impaired coordination, confusion, agitation, an increase in heart rate, or dilated pupils. No deaths were reported.
“These exposures can cause significant toxicity and are responsible for an increasing number of hospitalizations,” study coauthor Marit S. Tweet, MD, of Southern Illinois University, Springfield, and colleagues wrote.
About 97% of the exposures occurred in residences – 90% at the child’s own home – and about half of the cases involved 2- and 3-year-olds, they noted.
Examining national trends
Twenty-one states have approved recreational cannabis for people aged 21 years and older.
Prior research has shown that calls to poison centers and visits to emergency departments for pediatric cannabis consumption increased in certain states after the drug became legal in those jurisdictions.
To assess national trends, Dr. Tweet’s group analyzed cases in the National Poison Data System, which tracks potentially toxic exposures reported to poison control centers in the United States.
During the 5-year period, they identified 7,043 exposures to edible cannabis by children younger than age 6. In 2.2% of the cases, the drug had a major effect, defined as being either life-threatening or causing residual disability. In 21.9% of cases, the effect was considered to be moderate, with symptoms that were more pronounced, prolonged, or systemic than minor effects.
About 8% of the children were admitted to critical care units; 14.6% were admitted to non–critical care units.
Of 4,827 cases for which there was information about the clinical effects of the exposure and therapies used, 70% involved CNS depression, including 1.9% with “more severe CNS effects, including major CNS depression or coma,” according to the report.
Patients also experienced ataxia (7.4%), agitation (7.1%), confusion (6.1%), tremor (2%), and seizures (1.6%). Other common symptoms included tachycardia (11.4%), vomiting (9.5%), mydriasis (5.9%), and respiratory depression (3.1%).
Treatments for the exposures included intravenous fluids (20.7%), food or snacks (10.3%), and oxygen therapy (4%). Some patients also received naloxone (1.4%) or charcoal (2.1%).
“The total number of children requiring intubation during the study period was 35, or approximately 1 in 140,” the researchers reported. “Although this was a relatively rare occurrence, it is important for clinicians to be aware that life-threatening sequelae can develop and may necessitate invasive supportive care measures.”
Tempting and toxic
For toddlers, edible cannabis may be especially tempting and toxic. Edibles can “resemble common treats such as candies, chocolates, cookies, or other baked goods,” the researchers wrote. Children would not recognize, for example, that one chocolate bar might contain multiple 10-mg servings of tetrahydrocannabinol intended for adults.
Poison centers have been fielding more calls about edible cannabis use by older children, as well.
Adrienne Hughes, MD, assistant professor of emergency medicine at Oregon Health and Science University, Portland, recently found that many cases of intentional misuse and abuse by adolescents involve edible forms of cannabis.
“While marijuana carries a low risk for severe toxicity, it can be inebriating to the point of poor judgment, risk of falls or other injury, and occasionally a panic reaction in the novice user and unsuspecting children who accidentally ingest these products,” Dr. Hughes said in an interview.
Measures to keep edibles away from children could include changing how the products are packaged, limiting the maximum dose of drug per package, and educating the public about the risks to children, Dr. Tweet’s group wrote. They highlighted a 2019 position statement from the American College of Medical Toxicology that includes recommendations for responsible storage habits.
Dr. Hughes echoed one suggestion that is mentioned in the position statement: Parents should consider keeping their cannabis products locked up.
The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a new analysis of data from poison control centers.
In 2017, centers received 207 reports of children aged 5 years and younger who ingested edible cannabis. In 2021, 3,054 such cases were reported, according to the study, which was published online in Pediatrics.
Many of the children experienced clinical effects, such as depression of the central nervous system, impaired coordination, confusion, agitation, an increase in heart rate, or dilated pupils. No deaths were reported.
“These exposures can cause significant toxicity and are responsible for an increasing number of hospitalizations,” study coauthor Marit S. Tweet, MD, of Southern Illinois University, Springfield, and colleagues wrote.
About 97% of the exposures occurred in residences – 90% at the child’s own home – and about half of the cases involved 2- and 3-year-olds, they noted.
Examining national trends
Twenty-one states have approved recreational cannabis for people aged 21 years and older.
Prior research has shown that calls to poison centers and visits to emergency departments for pediatric cannabis consumption increased in certain states after the drug became legal in those jurisdictions.
To assess national trends, Dr. Tweet’s group analyzed cases in the National Poison Data System, which tracks potentially toxic exposures reported to poison control centers in the United States.
During the 5-year period, they identified 7,043 exposures to edible cannabis by children younger than age 6. In 2.2% of the cases, the drug had a major effect, defined as being either life-threatening or causing residual disability. In 21.9% of cases, the effect was considered to be moderate, with symptoms that were more pronounced, prolonged, or systemic than minor effects.
About 8% of the children were admitted to critical care units; 14.6% were admitted to non–critical care units.
Of 4,827 cases for which there was information about the clinical effects of the exposure and therapies used, 70% involved CNS depression, including 1.9% with “more severe CNS effects, including major CNS depression or coma,” according to the report.
Patients also experienced ataxia (7.4%), agitation (7.1%), confusion (6.1%), tremor (2%), and seizures (1.6%). Other common symptoms included tachycardia (11.4%), vomiting (9.5%), mydriasis (5.9%), and respiratory depression (3.1%).
Treatments for the exposures included intravenous fluids (20.7%), food or snacks (10.3%), and oxygen therapy (4%). Some patients also received naloxone (1.4%) or charcoal (2.1%).
“The total number of children requiring intubation during the study period was 35, or approximately 1 in 140,” the researchers reported. “Although this was a relatively rare occurrence, it is important for clinicians to be aware that life-threatening sequelae can develop and may necessitate invasive supportive care measures.”
Tempting and toxic
For toddlers, edible cannabis may be especially tempting and toxic. Edibles can “resemble common treats such as candies, chocolates, cookies, or other baked goods,” the researchers wrote. Children would not recognize, for example, that one chocolate bar might contain multiple 10-mg servings of tetrahydrocannabinol intended for adults.
Poison centers have been fielding more calls about edible cannabis use by older children, as well.
Adrienne Hughes, MD, assistant professor of emergency medicine at Oregon Health and Science University, Portland, recently found that many cases of intentional misuse and abuse by adolescents involve edible forms of cannabis.
“While marijuana carries a low risk for severe toxicity, it can be inebriating to the point of poor judgment, risk of falls or other injury, and occasionally a panic reaction in the novice user and unsuspecting children who accidentally ingest these products,” Dr. Hughes said in an interview.
Measures to keep edibles away from children could include changing how the products are packaged, limiting the maximum dose of drug per package, and educating the public about the risks to children, Dr. Tweet’s group wrote. They highlighted a 2019 position statement from the American College of Medical Toxicology that includes recommendations for responsible storage habits.
Dr. Hughes echoed one suggestion that is mentioned in the position statement: Parents should consider keeping their cannabis products locked up.
The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
according to a new analysis of data from poison control centers.
In 2017, centers received 207 reports of children aged 5 years and younger who ingested edible cannabis. In 2021, 3,054 such cases were reported, according to the study, which was published online in Pediatrics.
Many of the children experienced clinical effects, such as depression of the central nervous system, impaired coordination, confusion, agitation, an increase in heart rate, or dilated pupils. No deaths were reported.
“These exposures can cause significant toxicity and are responsible for an increasing number of hospitalizations,” study coauthor Marit S. Tweet, MD, of Southern Illinois University, Springfield, and colleagues wrote.
About 97% of the exposures occurred in residences – 90% at the child’s own home – and about half of the cases involved 2- and 3-year-olds, they noted.
Examining national trends
Twenty-one states have approved recreational cannabis for people aged 21 years and older.
Prior research has shown that calls to poison centers and visits to emergency departments for pediatric cannabis consumption increased in certain states after the drug became legal in those jurisdictions.
To assess national trends, Dr. Tweet’s group analyzed cases in the National Poison Data System, which tracks potentially toxic exposures reported to poison control centers in the United States.
During the 5-year period, they identified 7,043 exposures to edible cannabis by children younger than age 6. In 2.2% of the cases, the drug had a major effect, defined as being either life-threatening or causing residual disability. In 21.9% of cases, the effect was considered to be moderate, with symptoms that were more pronounced, prolonged, or systemic than minor effects.
About 8% of the children were admitted to critical care units; 14.6% were admitted to non–critical care units.
Of 4,827 cases for which there was information about the clinical effects of the exposure and therapies used, 70% involved CNS depression, including 1.9% with “more severe CNS effects, including major CNS depression or coma,” according to the report.
Patients also experienced ataxia (7.4%), agitation (7.1%), confusion (6.1%), tremor (2%), and seizures (1.6%). Other common symptoms included tachycardia (11.4%), vomiting (9.5%), mydriasis (5.9%), and respiratory depression (3.1%).
Treatments for the exposures included intravenous fluids (20.7%), food or snacks (10.3%), and oxygen therapy (4%). Some patients also received naloxone (1.4%) or charcoal (2.1%).
“The total number of children requiring intubation during the study period was 35, or approximately 1 in 140,” the researchers reported. “Although this was a relatively rare occurrence, it is important for clinicians to be aware that life-threatening sequelae can develop and may necessitate invasive supportive care measures.”
Tempting and toxic
For toddlers, edible cannabis may be especially tempting and toxic. Edibles can “resemble common treats such as candies, chocolates, cookies, or other baked goods,” the researchers wrote. Children would not recognize, for example, that one chocolate bar might contain multiple 10-mg servings of tetrahydrocannabinol intended for adults.
Poison centers have been fielding more calls about edible cannabis use by older children, as well.
Adrienne Hughes, MD, assistant professor of emergency medicine at Oregon Health and Science University, Portland, recently found that many cases of intentional misuse and abuse by adolescents involve edible forms of cannabis.
“While marijuana carries a low risk for severe toxicity, it can be inebriating to the point of poor judgment, risk of falls or other injury, and occasionally a panic reaction in the novice user and unsuspecting children who accidentally ingest these products,” Dr. Hughes said in an interview.
Measures to keep edibles away from children could include changing how the products are packaged, limiting the maximum dose of drug per package, and educating the public about the risks to children, Dr. Tweet’s group wrote. They highlighted a 2019 position statement from the American College of Medical Toxicology that includes recommendations for responsible storage habits.
Dr. Hughes echoed one suggestion that is mentioned in the position statement: Parents should consider keeping their cannabis products locked up.
The researchers disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM PEDIATRICS
FDA approves Wegovy (semaglutide) for obesity in teens 12 and up
The Food and Drug Administration has approved semaglutide 2.4 mg (Wegovy), a once-weekly subcutaneous injection, for the additional indication of treating obesity in adolescents aged 12 years and older.
This is defined as those with an initial body mass index at or above the 95th percentile for age and sex (based on CDC growth charts). Semaglutide must be administered along with lifestyle intervention of a reduced calorie meal plan and increased physical activity.
When Wegovy was approved for use in adults with obesity in June 2021, it was labeled a “game changer.”
The new approval is based on the results of the STEP TEENS phase 3 trial of once-weekly 2.4 mg of semaglutide in adolescents 12- to <18 years old with obesity, the drug’s manufacturer, Novo Nordisk, announced in a press release.
In STEP TEENS, reported at Obesity Week 2022 in November, and simultaneously published in the New England Journal of Medicine, adolescents with obesity treated with semaglutide for 68 weeks had a 16.1% reduction in BMI compared with a 0.6% increase in BMI in those receiving placebo. Both groups also received lifestyle intervention. Mean weight loss was 15.3 kg (33.7 pounds) among teens on semaglutide, while those on placebo gained 2.4 kg (5.3 pounds).
At the time, Claudia K. Fox, MD, MPH, codirector of the Center for Pediatric Obesity Medicine at the University of Minnesota – who was not involved with the research – told this news organization the results were “mind-blowing ... we are getting close to bariatric surgery results” in these adolescent patients with obesity.
Semaglutide is a GLP-1 agonist, as is a related agent, also from Novo Nordisk, liraglutide (Saxenda), a daily subcutaneous injection, which was approved for use in adolescents aged 12 and older in December 2020. Wegovy is the first weekly subcutaneous injection approved for use in adolescents.
Other agents approved for obesity in those older than 12 in the United States include the combination phentermine and topiramate extended-release capsules (Qsymia) in June 2022, and orlistat (Alli). Phentermine is approved for those aged 16 and older.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved semaglutide 2.4 mg (Wegovy), a once-weekly subcutaneous injection, for the additional indication of treating obesity in adolescents aged 12 years and older.
This is defined as those with an initial body mass index at or above the 95th percentile for age and sex (based on CDC growth charts). Semaglutide must be administered along with lifestyle intervention of a reduced calorie meal plan and increased physical activity.
When Wegovy was approved for use in adults with obesity in June 2021, it was labeled a “game changer.”
The new approval is based on the results of the STEP TEENS phase 3 trial of once-weekly 2.4 mg of semaglutide in adolescents 12- to <18 years old with obesity, the drug’s manufacturer, Novo Nordisk, announced in a press release.
In STEP TEENS, reported at Obesity Week 2022 in November, and simultaneously published in the New England Journal of Medicine, adolescents with obesity treated with semaglutide for 68 weeks had a 16.1% reduction in BMI compared with a 0.6% increase in BMI in those receiving placebo. Both groups also received lifestyle intervention. Mean weight loss was 15.3 kg (33.7 pounds) among teens on semaglutide, while those on placebo gained 2.4 kg (5.3 pounds).
At the time, Claudia K. Fox, MD, MPH, codirector of the Center for Pediatric Obesity Medicine at the University of Minnesota – who was not involved with the research – told this news organization the results were “mind-blowing ... we are getting close to bariatric surgery results” in these adolescent patients with obesity.
Semaglutide is a GLP-1 agonist, as is a related agent, also from Novo Nordisk, liraglutide (Saxenda), a daily subcutaneous injection, which was approved for use in adolescents aged 12 and older in December 2020. Wegovy is the first weekly subcutaneous injection approved for use in adolescents.
Other agents approved for obesity in those older than 12 in the United States include the combination phentermine and topiramate extended-release capsules (Qsymia) in June 2022, and orlistat (Alli). Phentermine is approved for those aged 16 and older.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration has approved semaglutide 2.4 mg (Wegovy), a once-weekly subcutaneous injection, for the additional indication of treating obesity in adolescents aged 12 years and older.
This is defined as those with an initial body mass index at or above the 95th percentile for age and sex (based on CDC growth charts). Semaglutide must be administered along with lifestyle intervention of a reduced calorie meal plan and increased physical activity.
When Wegovy was approved for use in adults with obesity in June 2021, it was labeled a “game changer.”
The new approval is based on the results of the STEP TEENS phase 3 trial of once-weekly 2.4 mg of semaglutide in adolescents 12- to <18 years old with obesity, the drug’s manufacturer, Novo Nordisk, announced in a press release.
In STEP TEENS, reported at Obesity Week 2022 in November, and simultaneously published in the New England Journal of Medicine, adolescents with obesity treated with semaglutide for 68 weeks had a 16.1% reduction in BMI compared with a 0.6% increase in BMI in those receiving placebo. Both groups also received lifestyle intervention. Mean weight loss was 15.3 kg (33.7 pounds) among teens on semaglutide, while those on placebo gained 2.4 kg (5.3 pounds).
At the time, Claudia K. Fox, MD, MPH, codirector of the Center for Pediatric Obesity Medicine at the University of Minnesota – who was not involved with the research – told this news organization the results were “mind-blowing ... we are getting close to bariatric surgery results” in these adolescent patients with obesity.
Semaglutide is a GLP-1 agonist, as is a related agent, also from Novo Nordisk, liraglutide (Saxenda), a daily subcutaneous injection, which was approved for use in adolescents aged 12 and older in December 2020. Wegovy is the first weekly subcutaneous injection approved for use in adolescents.
Other agents approved for obesity in those older than 12 in the United States include the combination phentermine and topiramate extended-release capsules (Qsymia) in June 2022, and orlistat (Alli). Phentermine is approved for those aged 16 and older.
A version of this article first appeared on Medscape.com.
Study of beliefs about what causes cancer sparks debate
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
The study, entitled, “Everything Causes Cancer? Beliefs and Attitudes Towards Cancer Prevention Among Anti-Vaxxers, Flat Earthers, and Reptilian Conspiracists: Online Cross Sectional Survey,” was published in the Christmas 2022 issue of The British Medical Journal (BMJ).
The authors explain that they set out to evaluate “the patterns of beliefs about cancer among people who believed in conspiracies, rejected the COVID-19 vaccine, or preferred alternative medicine.”
They sought such people on social media and online chat platforms and asked them questions about real and mythical causes of cancer.
Almost half of survey participants agreed with the statement, “It seems like everything causes cancer.”
Overall, among all participants, awareness of the actual causes of cancer was greater than awareness of the mythical causes of cancer, the authors report. However, awareness of the actual causes of cancer was lower among the unvaccinated and members of conspiracy groups than among their counterparts.
The authors are concerned that their findings suggest “a direct connection between digital misinformation and consequent potential erroneous health decisions, which may represent a further preventable fraction of cancer.”
Backlash and criticism
The study “highlights the difficulty society encounters in distinguishing the actual causes of cancer from mythical causes,” The BMJ commented on Twitter.
However, both the study and the journal received some backlash.
This is a “horrible article seeking to smear people with concerns about COVID vaccines,” commented Clare Craig, a British consultant pathologist who specializes in cancer diagnostics.
The study and its methodology were also harshly criticized on Twitter by Normal Fenton, professor of risk information management at the Queen Mary University of London.
The senior author of the study, Laura Costas, a medical epidemiologist with the Catalan Institute of Oncology, Barcelona, told this news organization that the naysayers on social media, many of whom focused their comments on the COVID-19 vaccine, prove the purpose of the study – that misinformation spreads widely on the internet.
“Most comments focused on spreading COVID-19 myths, which were not the direct subject of the study, and questioned the motivations of BMJ authors and the scientific community, assuming they had a common malevolent hidden agenda,” Ms. Costas said.
“They stated the need of having critical thinking, a trait in common with the scientific method, but dogmatically dismissed any information that comes from official sources,” she added.
Ms. Costas commented that “society encounters difficulty in differentiating actual from mythical causes of cancer owing to mass information. We therefore planned this study with a certain satire, which is in line with the essence of The BMJ Christmas issue.”
The BMJ has a long history of publishing a lighthearted Christmas edition full of original, satirical, and nontraditional studies. Previous years have seen studies that explored potential harms from holly and ivy, survival time of chocolates on hospital wards, and the question, “Were James Bond’s drinks shaken because of alcohol induced tremor?”
Study details
Ms. Costas and colleagues sought participants for their survey from online forums that included 4chan and Reddit, which are known for their controversial content posted by anonymous users. Data were also collected from ForoCoches and HispaChan, well-known Spanish online forums. These online sites were intentionally chosen because researchers thought “conspiracy beliefs would be more prevalent,” according to Ms. Costas.
Across the multiple forums, there were 1,494 participants. Of these, 209 participants were unvaccinated against COVID-19, 112 preferred alternatives rather than conventional medicine, and 62 reported that they believed the earth was flat or believed that humanoids take reptilian forms to manipulate human societies.
The team then sought to assess beliefs about actual and mythical (nonestablished) causes of cancer by presenting the participants with the closed risk factor questions on two validated scales – the Cancer Awareness Measure (CAM) and CAM–Mythical Causes Scale (CAM-MYCS).
Responses to both were recorded on a five-point scale; answers ranged from “strongly disagree” to “strongly agree.”
The CAM assesses cancer risk perceptions of 11 established risk factors for cancer: smoking actively or passively, consuming alcohol, low levels of physical activity, consuming red or processed meat, getting sunburnt as a child, family history of cancer, human papillomavirus infection, being overweight, age greater than or equal to 70 years, and low vegetable and fruit consumption.
The CAM-MYCS measure includes 12 questions on risk perceptions of mythical causes of cancer – nonestablished causes that are commonly believed to cause cancer but for which there is no supporting scientific evidence, the authors explain. These items include drinking from plastic bottles; eating food containing artificial sweeteners or additives and genetically modified food; using microwave ovens, aerosol containers, mobile phones, and cleaning products; living near power lines; feeling stressed; experiencing physical trauma; and being exposed to electromagnetic frequencies/non-ionizing radiation, such as wi-fi networks, radio, and television.
The most endorsed mythical causes of cancer were eating food containing additives (63.9%) or sweeteners (50.7%), feeling stressed (59.7%), and eating genetically modified foods (38.4%).
A version of this article first appeared on Medscape.com.
Time for a rest
“More than Jews have kept Shabbat, Shabbat has kept the Jews.” – Ahad Ha’am
You should all be well rested by now. After all, we’ve just come through the festive shutdown of the holiday season where all of your pumpkin/peppermint/marshmallow flavored coffees were sipped while walking around in your jimjams at 10 a.m. It was the time of year for you to take time off to get a proper rest and be energized to get back to work. Yet, I’m not feeling it from you.
So let’s talk about burnout – just kidding, that would only make it worse. “Burned-out’’ is a hackneyed and defective phrase to describe what many of us are feeling. We are not “destroyed, gutted by fire or by overheating.” No, we are, as one of our docs put it to me: “Just tired.” Ah, a much better Old English word! “Tired” captures it. It means to feel “in need of rest.” We are not ruined, we are just depleted. We don’t need discarding. We need some rest.
I asked some docs when they thought this feeling of exhaustion first began. We agreed that the pandemic, doubledemic, tripledemic, backlog have taken a toll. But The consumerization of medicine? All factors, but not the beginning. No, the beginning was before paper charts. Well, actually it was before paper. We have to go back to the 5th or 6th century BCE. That is when scholars believe the book of Genesis originated from the Yahwist source. In it, it is written that the 7th day be set aside as a day of rest from labor. It is not written that burnout would ensue if sabbath wasn’t observed; however, if you failed to keep it, then you might have been killed. They took rest seriously back then.
This innovation of setting aside a day each week to rest, reflect, and worship was such a good idea that it was codified as one of the 10 commandments. It spread widely. Early Christians kept the Jewish tradition of observing Shabbat from Friday sundown to Saturday until the ever practical Romans decided that Sunday would be a better day. Sunday was already the day to worship the sun god. The newly-converted Christian Emperor Constantine issued an edict on March 7th, 321 CE that all “city people and craftsmen shall rest from labor upon the venerable day of the sun.” And so Sunday it was.
Protestant Seventh-day denomination churches later shifted sabbath back to Saturday believing that Sunday must have been the Pope’s idea. The best deal seems to have been around 1273 when the Ethiopian Orthodox leader Ewostatewos decreed that both Saturday AND Sunday would be days of rest. (But when would one go to Costco?!) In Islam, there is Jumu’ah on Friday. Buddhists have Uposatha, a day of rest and observance every 7 or 8 days. Bah’ai keep Friday as a day of rest and worship. So vital are days of respite to the health of our communities that the state has made working on certain days a violation of the law, “blue laws” they are called. We’ve had blue laws on the books since the time of the Jamestown Colony in 1619 where the first Virginia Assembly required taking Sunday off for worship. Most of these laws have been repealed, although a few states, such as Rhode Island, still have blue laws prohibiting retail and grocery stores from opening on Thanksgiving or Christmas. So there – enjoy your two days off this year!
Ironically, this column, like most of mine, comes to you after my having written it on a Saturday and Sunday. I also just logged on to my EMR and checked results, renewed a few prescriptions, and answered a couple messages. If I didn’t, my Monday’s work would be crushingly heavy.
Maybe I need to be more efficient and finish my work during the week. Or maybe I need to realize that work has not let up since about 600 BCE and taking one day off each week to rest is an obligation to myself, my family and my community.
I wonder if I can choose Mondays.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
“More than Jews have kept Shabbat, Shabbat has kept the Jews.” – Ahad Ha’am
You should all be well rested by now. After all, we’ve just come through the festive shutdown of the holiday season where all of your pumpkin/peppermint/marshmallow flavored coffees were sipped while walking around in your jimjams at 10 a.m. It was the time of year for you to take time off to get a proper rest and be energized to get back to work. Yet, I’m not feeling it from you.
So let’s talk about burnout – just kidding, that would only make it worse. “Burned-out’’ is a hackneyed and defective phrase to describe what many of us are feeling. We are not “destroyed, gutted by fire or by overheating.” No, we are, as one of our docs put it to me: “Just tired.” Ah, a much better Old English word! “Tired” captures it. It means to feel “in need of rest.” We are not ruined, we are just depleted. We don’t need discarding. We need some rest.
I asked some docs when they thought this feeling of exhaustion first began. We agreed that the pandemic, doubledemic, tripledemic, backlog have taken a toll. But The consumerization of medicine? All factors, but not the beginning. No, the beginning was before paper charts. Well, actually it was before paper. We have to go back to the 5th or 6th century BCE. That is when scholars believe the book of Genesis originated from the Yahwist source. In it, it is written that the 7th day be set aside as a day of rest from labor. It is not written that burnout would ensue if sabbath wasn’t observed; however, if you failed to keep it, then you might have been killed. They took rest seriously back then.
This innovation of setting aside a day each week to rest, reflect, and worship was such a good idea that it was codified as one of the 10 commandments. It spread widely. Early Christians kept the Jewish tradition of observing Shabbat from Friday sundown to Saturday until the ever practical Romans decided that Sunday would be a better day. Sunday was already the day to worship the sun god. The newly-converted Christian Emperor Constantine issued an edict on March 7th, 321 CE that all “city people and craftsmen shall rest from labor upon the venerable day of the sun.” And so Sunday it was.
Protestant Seventh-day denomination churches later shifted sabbath back to Saturday believing that Sunday must have been the Pope’s idea. The best deal seems to have been around 1273 when the Ethiopian Orthodox leader Ewostatewos decreed that both Saturday AND Sunday would be days of rest. (But when would one go to Costco?!) In Islam, there is Jumu’ah on Friday. Buddhists have Uposatha, a day of rest and observance every 7 or 8 days. Bah’ai keep Friday as a day of rest and worship. So vital are days of respite to the health of our communities that the state has made working on certain days a violation of the law, “blue laws” they are called. We’ve had blue laws on the books since the time of the Jamestown Colony in 1619 where the first Virginia Assembly required taking Sunday off for worship. Most of these laws have been repealed, although a few states, such as Rhode Island, still have blue laws prohibiting retail and grocery stores from opening on Thanksgiving or Christmas. So there – enjoy your two days off this year!
Ironically, this column, like most of mine, comes to you after my having written it on a Saturday and Sunday. I also just logged on to my EMR and checked results, renewed a few prescriptions, and answered a couple messages. If I didn’t, my Monday’s work would be crushingly heavy.
Maybe I need to be more efficient and finish my work during the week. Or maybe I need to realize that work has not let up since about 600 BCE and taking one day off each week to rest is an obligation to myself, my family and my community.
I wonder if I can choose Mondays.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
“More than Jews have kept Shabbat, Shabbat has kept the Jews.” – Ahad Ha’am
You should all be well rested by now. After all, we’ve just come through the festive shutdown of the holiday season where all of your pumpkin/peppermint/marshmallow flavored coffees were sipped while walking around in your jimjams at 10 a.m. It was the time of year for you to take time off to get a proper rest and be energized to get back to work. Yet, I’m not feeling it from you.
So let’s talk about burnout – just kidding, that would only make it worse. “Burned-out’’ is a hackneyed and defective phrase to describe what many of us are feeling. We are not “destroyed, gutted by fire or by overheating.” No, we are, as one of our docs put it to me: “Just tired.” Ah, a much better Old English word! “Tired” captures it. It means to feel “in need of rest.” We are not ruined, we are just depleted. We don’t need discarding. We need some rest.
I asked some docs when they thought this feeling of exhaustion first began. We agreed that the pandemic, doubledemic, tripledemic, backlog have taken a toll. But The consumerization of medicine? All factors, but not the beginning. No, the beginning was before paper charts. Well, actually it was before paper. We have to go back to the 5th or 6th century BCE. That is when scholars believe the book of Genesis originated from the Yahwist source. In it, it is written that the 7th day be set aside as a day of rest from labor. It is not written that burnout would ensue if sabbath wasn’t observed; however, if you failed to keep it, then you might have been killed. They took rest seriously back then.
This innovation of setting aside a day each week to rest, reflect, and worship was such a good idea that it was codified as one of the 10 commandments. It spread widely. Early Christians kept the Jewish tradition of observing Shabbat from Friday sundown to Saturday until the ever practical Romans decided that Sunday would be a better day. Sunday was already the day to worship the sun god. The newly-converted Christian Emperor Constantine issued an edict on March 7th, 321 CE that all “city people and craftsmen shall rest from labor upon the venerable day of the sun.” And so Sunday it was.
Protestant Seventh-day denomination churches later shifted sabbath back to Saturday believing that Sunday must have been the Pope’s idea. The best deal seems to have been around 1273 when the Ethiopian Orthodox leader Ewostatewos decreed that both Saturday AND Sunday would be days of rest. (But when would one go to Costco?!) In Islam, there is Jumu’ah on Friday. Buddhists have Uposatha, a day of rest and observance every 7 or 8 days. Bah’ai keep Friday as a day of rest and worship. So vital are days of respite to the health of our communities that the state has made working on certain days a violation of the law, “blue laws” they are called. We’ve had blue laws on the books since the time of the Jamestown Colony in 1619 where the first Virginia Assembly required taking Sunday off for worship. Most of these laws have been repealed, although a few states, such as Rhode Island, still have blue laws prohibiting retail and grocery stores from opening on Thanksgiving or Christmas. So there – enjoy your two days off this year!
Ironically, this column, like most of mine, comes to you after my having written it on a Saturday and Sunday. I also just logged on to my EMR and checked results, renewed a few prescriptions, and answered a couple messages. If I didn’t, my Monday’s work would be crushingly heavy.
Maybe I need to be more efficient and finish my work during the week. Or maybe I need to realize that work has not let up since about 600 BCE and taking one day off each week to rest is an obligation to myself, my family and my community.
I wonder if I can choose Mondays.
Dr. Benabio is director of Healthcare Transformation and chief of dermatology at Kaiser Permanente San Diego. The opinions expressed in this column are his own and do not represent those of Kaiser Permanente. Dr. Benabio is @Dermdoc on Twitter. Write to him at [email protected].
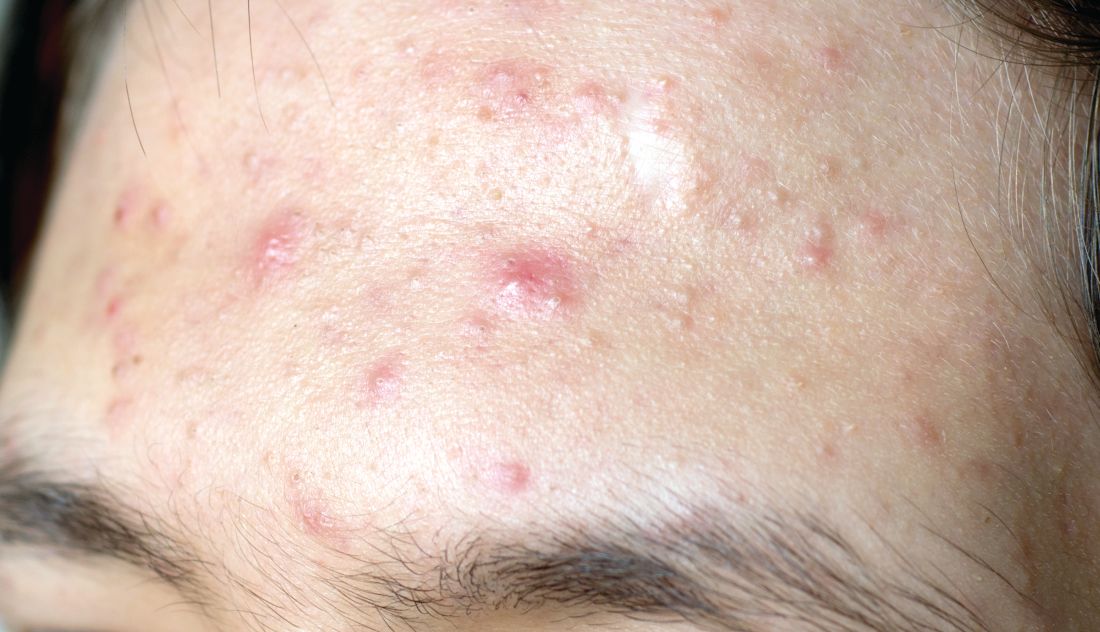
Topical treatment options for acne continue to expand
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
SAN DIEGO – , according to Lawrence F. Eichenfield, MD.
The product, known as IDP-126 and being developed by Ortho Dermatologics, is a fixed dose triple combination of clindamycin 1.2% plus benzoyl peroxide 3.1% and adapalene 0.15% being evaluated in patients nine years of age and older. According to a 2021 press release from the company, results from a second 12-week pivotal phase 3 trial showed a treatment success of 50.5% and 20.5% for IDP-126 and its vehicle, respectively, along with significant changes from baseline in inflammatory lesion count and non-inflammatory lesion count.
More recently, researchers led by Linda Stein Gold, MD, conducted a 12-week multicenter, randomized, double-blind study of IDP-126 in 741 children, adolescents, and adults with moderate to severe acne. They reported 52.5% of patients treated with IDP-126 gel achieved treatment success by week 12, with over 70% reduction in inflammatory and noninflammatory lesions.
“This will be interesting to follow as it moves along,” Dr. Eichenfield, chief of pediatric and adolescent dermatology at Rady Children’s Hospital, San Diego, said at the annual Masters of Aesthetics Symposium in a presentation on the newest topical acne treatments.
“If approved, we probably will be able to decrease our need for systemic therapies in some individuals,” he said. “It’s something that may become important in practices that mix and match between medical and procedural or surgical approaches to acne.”
Dr. Eichenfield highlighted other products for the topical treatment of acne:
- Trifarotene cream 0.005% (Aklief). In 2019, Food and Drug Administration approval made trifarotene cream the first new retinoid indicated for acne in several decades. It is indicated for the topical treatment of acne vulgaris in patients age 9 and older and has been studied in acne of the face, chest, and back.
- Tazarotene lotion 0.045% (Arazlo). The 0.1% formulation of tazarotene is commonly used for acne, but it can cause skin irritation, dryness, and erythema. The new 0.045% formulation was developed in a three-dimensional mesh matrix, with ingredients from an oil-in-water emulsion. “Many of the new acne products come with a background of vehicle delivery systems that minimize the concentration gradient, so it decreases irritation,” said Dr. Eichenfield, one of the authors of a 2021 review article on the management of acne vulgaris in JAMA. “This has very good efficacy without the traditional irritation of other tazarotene products,” Dr. Eichenfield said.
- Minocycline 4% topical foam (Amzeeq). The 2019 U.S. approval marked the first and so far only topical minocycline prescription treatment for acne. “Its hydrophobic composition allows for stable and efficient delivery of inherently unstable pharmaceutical ingredients,” he said. “It’s generally well tolerated.”
- Clascoterone cream 1% (Winlevi). This first-in-class topical androgen receptor inhibitor is approved for the treatment of acne in patients 12 years and older. It competes with dihydrotestosterone and selectively targets androgen receptors in sebocytes and hair papilla cells. “It is safe for use in men, has been studied on the face and trunk, and has been shown to inhibit sebum production, reduce secretion of inflammatory cytokines, and inhibit inflammatory pathways,” Dr. Eichenfield said.
- Micro-encapsulated benzoyl peroxide 3% and tretinoin 0.1% cream (Twyneo). This is a once-daily fixed-dose combination of tretinoin and benzoyl peroxide indicated for the treatment of acne vulgaris in patients age 9 and older. According to a press release from Sol-Gel, the manufacturer, silica (silicon dioxide) core shell structures separate micro-encapsulate tretinoin crystals and benzoyl peroxide crystals, enabling inclusion of the two active ingredients in the cream.
Dr. Eichenfield disclosed that he has been an investigator and/or consultant for Almirall, Cassiopea, Dermata, Galderma, and Ortho Dermatologics.
AT MOAS 2022
Genetic test identifies parental source of cancer variant
A new hereditary test can determine whether a cancer-disposing gene was inherited from a patient’s father or mother without the need for parental DNA, potentially improving disease screening and management.
“The presence of parental imprints in regions of the genome has been known for a long time,” study author Peter Lansdorp, MD, PhD, of the BC Cancer Research Centre in Vancouver, said in an interview. In addition, the ability of a specific sequencing technology (Strand-seq) to generate a set of DNA variants that tend to be inherited together from a single parent has been documented in several studies.
“That these two pieces can be put together to assign alleles in a patient to one of the parents without studying the DNA of the parents is a major advance,” said Dr. Lansdorp.
Principal author Steven J.M. Jones, PhD, associate director of bioinformatics at BC Cancer Research Centre, explained, “for directing cascade genetic testing, the test could be used almost immediately, even as a research test. It just guides which side of the family to focus familial genetic testing efforts on and is internally validated by the patient’s variant and later confirmed by clinical testing in the family.”
Dr. Jones added, especially when parents are deceased or unavailable.
The study was published online in Cell Genomics.
Low error rate
Determining a parent of origin for hereditary variants “is essential to evaluate disease risk when a pathogenic variant has PofO effects, that is, when a patient’s risk of disease depends on from which parent it is inherited,” the authors wrote. An example is hereditary paraganglioma-pheochromocytoma syndrome as a result of pathogenic variants in SDHD or SDHAF2 genes. Individuals with the variants are at high risk of developing certain cancers, but only if a defective gene is inherited from their father. If inherited from their mother, there is no increased risk.
The new method relies on a technique called “phased DNA methylation” at maternally and paternally imprinted gene loci, as well as chromosome length phasing of DNA sequences.
The team used five human genome “trios” – two parents and the proband (the first person in a family to receive genetic testing or counseling for a suspected hereditary risk) – to pilot the approach. They showed that the method can correctly identify the PofO with an average mismatch error rate of 0.31% for single nucleotide variants and 1.89% for insertions or deletions (indels).
“We will need to validate this technology for different genes in real-world samples from individuals of diverse backgrounds,” said Dr. Jones. The first step is to validate the technology in scenarios with immediate clinical utility, like with SDHD, where lifelong medical management is affected by knowledge of whether the variant was inherited from the mother or father.
“We would also like to quickly validate this for common hereditary cancer genes, like BRCA1, BRCA2, and Lynch syndrome–associated genes, where prediction of PofO may improve low rates of genetic testing in family members by providing more accurate estimates of their risk to carry the familial variant.”
Challenges to moving the test to the clinic, Dr. Jones said, include scaling up the technology, demonstrating clinical and economic utility, compared with existing testing approaches, “and familiarizing clinicians with a new type of test that will routinely give this added dimension of information.”
‘Tremendously promising technology’
Pathologist Stephen Yip, MD, PhD, of the Vancouver Coastal Health Research Institute, who was not involved in the study but disclosed that he collaborates with the authors on other grant-funded projects, said in a comment that “this is a tremendously promising technology that has immediate practical implications in the investigation of PofO of a pathogenic locus, particularly when genetic material is available only from the proband.”
However, “rigorous validation against the current gold standard of short-reading, next-generation sequencing of trios is needed prior to clinical deployment,” he said. “This will take time and effort. However, the promise of this technology is worth the effort.
“Also, there is the possibility of uncovering novel genetics during testing, which could present an ethical dilemma,” he noted. “A robust consenting and ethical framework and early involvement of an ethicist would be helpful.”
Research in Dr. Lansdorp’s laboratory is funded by the Terry Fox Research Institute, the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, and the government of British Columbia. Dr. Lansdorp, Dr. Jones, and Dr. Yip reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new hereditary test can determine whether a cancer-disposing gene was inherited from a patient’s father or mother without the need for parental DNA, potentially improving disease screening and management.
“The presence of parental imprints in regions of the genome has been known for a long time,” study author Peter Lansdorp, MD, PhD, of the BC Cancer Research Centre in Vancouver, said in an interview. In addition, the ability of a specific sequencing technology (Strand-seq) to generate a set of DNA variants that tend to be inherited together from a single parent has been documented in several studies.
“That these two pieces can be put together to assign alleles in a patient to one of the parents without studying the DNA of the parents is a major advance,” said Dr. Lansdorp.
Principal author Steven J.M. Jones, PhD, associate director of bioinformatics at BC Cancer Research Centre, explained, “for directing cascade genetic testing, the test could be used almost immediately, even as a research test. It just guides which side of the family to focus familial genetic testing efforts on and is internally validated by the patient’s variant and later confirmed by clinical testing in the family.”
Dr. Jones added, especially when parents are deceased or unavailable.
The study was published online in Cell Genomics.
Low error rate
Determining a parent of origin for hereditary variants “is essential to evaluate disease risk when a pathogenic variant has PofO effects, that is, when a patient’s risk of disease depends on from which parent it is inherited,” the authors wrote. An example is hereditary paraganglioma-pheochromocytoma syndrome as a result of pathogenic variants in SDHD or SDHAF2 genes. Individuals with the variants are at high risk of developing certain cancers, but only if a defective gene is inherited from their father. If inherited from their mother, there is no increased risk.
The new method relies on a technique called “phased DNA methylation” at maternally and paternally imprinted gene loci, as well as chromosome length phasing of DNA sequences.
The team used five human genome “trios” – two parents and the proband (the first person in a family to receive genetic testing or counseling for a suspected hereditary risk) – to pilot the approach. They showed that the method can correctly identify the PofO with an average mismatch error rate of 0.31% for single nucleotide variants and 1.89% for insertions or deletions (indels).
“We will need to validate this technology for different genes in real-world samples from individuals of diverse backgrounds,” said Dr. Jones. The first step is to validate the technology in scenarios with immediate clinical utility, like with SDHD, where lifelong medical management is affected by knowledge of whether the variant was inherited from the mother or father.
“We would also like to quickly validate this for common hereditary cancer genes, like BRCA1, BRCA2, and Lynch syndrome–associated genes, where prediction of PofO may improve low rates of genetic testing in family members by providing more accurate estimates of their risk to carry the familial variant.”
Challenges to moving the test to the clinic, Dr. Jones said, include scaling up the technology, demonstrating clinical and economic utility, compared with existing testing approaches, “and familiarizing clinicians with a new type of test that will routinely give this added dimension of information.”
‘Tremendously promising technology’
Pathologist Stephen Yip, MD, PhD, of the Vancouver Coastal Health Research Institute, who was not involved in the study but disclosed that he collaborates with the authors on other grant-funded projects, said in a comment that “this is a tremendously promising technology that has immediate practical implications in the investigation of PofO of a pathogenic locus, particularly when genetic material is available only from the proband.”
However, “rigorous validation against the current gold standard of short-reading, next-generation sequencing of trios is needed prior to clinical deployment,” he said. “This will take time and effort. However, the promise of this technology is worth the effort.
“Also, there is the possibility of uncovering novel genetics during testing, which could present an ethical dilemma,” he noted. “A robust consenting and ethical framework and early involvement of an ethicist would be helpful.”
Research in Dr. Lansdorp’s laboratory is funded by the Terry Fox Research Institute, the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, and the government of British Columbia. Dr. Lansdorp, Dr. Jones, and Dr. Yip reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new hereditary test can determine whether a cancer-disposing gene was inherited from a patient’s father or mother without the need for parental DNA, potentially improving disease screening and management.
“The presence of parental imprints in regions of the genome has been known for a long time,” study author Peter Lansdorp, MD, PhD, of the BC Cancer Research Centre in Vancouver, said in an interview. In addition, the ability of a specific sequencing technology (Strand-seq) to generate a set of DNA variants that tend to be inherited together from a single parent has been documented in several studies.
“That these two pieces can be put together to assign alleles in a patient to one of the parents without studying the DNA of the parents is a major advance,” said Dr. Lansdorp.
Principal author Steven J.M. Jones, PhD, associate director of bioinformatics at BC Cancer Research Centre, explained, “for directing cascade genetic testing, the test could be used almost immediately, even as a research test. It just guides which side of the family to focus familial genetic testing efforts on and is internally validated by the patient’s variant and later confirmed by clinical testing in the family.”
Dr. Jones added, especially when parents are deceased or unavailable.
The study was published online in Cell Genomics.
Low error rate
Determining a parent of origin for hereditary variants “is essential to evaluate disease risk when a pathogenic variant has PofO effects, that is, when a patient’s risk of disease depends on from which parent it is inherited,” the authors wrote. An example is hereditary paraganglioma-pheochromocytoma syndrome as a result of pathogenic variants in SDHD or SDHAF2 genes. Individuals with the variants are at high risk of developing certain cancers, but only if a defective gene is inherited from their father. If inherited from their mother, there is no increased risk.
The new method relies on a technique called “phased DNA methylation” at maternally and paternally imprinted gene loci, as well as chromosome length phasing of DNA sequences.
The team used five human genome “trios” – two parents and the proband (the first person in a family to receive genetic testing or counseling for a suspected hereditary risk) – to pilot the approach. They showed that the method can correctly identify the PofO with an average mismatch error rate of 0.31% for single nucleotide variants and 1.89% for insertions or deletions (indels).
“We will need to validate this technology for different genes in real-world samples from individuals of diverse backgrounds,” said Dr. Jones. The first step is to validate the technology in scenarios with immediate clinical utility, like with SDHD, where lifelong medical management is affected by knowledge of whether the variant was inherited from the mother or father.
“We would also like to quickly validate this for common hereditary cancer genes, like BRCA1, BRCA2, and Lynch syndrome–associated genes, where prediction of PofO may improve low rates of genetic testing in family members by providing more accurate estimates of their risk to carry the familial variant.”
Challenges to moving the test to the clinic, Dr. Jones said, include scaling up the technology, demonstrating clinical and economic utility, compared with existing testing approaches, “and familiarizing clinicians with a new type of test that will routinely give this added dimension of information.”
‘Tremendously promising technology’
Pathologist Stephen Yip, MD, PhD, of the Vancouver Coastal Health Research Institute, who was not involved in the study but disclosed that he collaborates with the authors on other grant-funded projects, said in a comment that “this is a tremendously promising technology that has immediate practical implications in the investigation of PofO of a pathogenic locus, particularly when genetic material is available only from the proband.”
However, “rigorous validation against the current gold standard of short-reading, next-generation sequencing of trios is needed prior to clinical deployment,” he said. “This will take time and effort. However, the promise of this technology is worth the effort.
“Also, there is the possibility of uncovering novel genetics during testing, which could present an ethical dilemma,” he noted. “A robust consenting and ethical framework and early involvement of an ethicist would be helpful.”
Research in Dr. Lansdorp’s laboratory is funded by the Terry Fox Research Institute, the Canadian Institutes of Health Research, the Canadian Foundation for Innovation, and the government of British Columbia. Dr. Lansdorp, Dr. Jones, and Dr. Yip reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM CELL GENOMICS
Double CF combination safe, effective in 1- to 2-year-olds
In children with cystic fibrosis (CF) and homozygous for the F508 mutation, the combination of lumacaftor and ivacaftor appears generally safe, and biomarker data suggest its efficacy, according to results from a new open-label phase 3 trial.
In September, the Food and Drug Administration approved lumacaftor/ivacaftor for patients aged 1 years and above. Waiting in the wings is the triple combination of elexacaftor, tezacaftor, and ivacaftor (ETI), which is available for patients with at least one copy of the F508 mutation aged 6 and over. ETI is also being tested in younger patients.
One driving factor for early treatment is countering the malnutrition that can occur among CF patients because of poor pancreatic insufficiency and chronic inflammation. “We’ve known for many years that (being) at or above average body weight and height predict better lung function. And we’ve known for quite a while that the height-for-age percentile, in preschool years, actually predicts your lung function later, and how long you’re going to live, so nutrition is incredibly important,” said study author Susanna McColley, MD, in an interview. It’s also difficult to use lung function tests in young children, since even adults can find them challenging, she said.
“FEV1 [forced expiratory volume in 1 second] is the strongest predictor of survival, and then nutrition is the highest predictor of FEV1, so that’s kind of the construct. They had similar improvement in the functional measures of their pancreas and in the measures of inflammation in the gut. I think the story here is that starting a modulator early has a likelihood to have positive health effects that go forward. We can’t say that from the data in the paper. It’s a 24-week study, but looking at the pancreatic and intestinal functioning and also the fact that there was a decrease in sweat chloride is important,” said Dr. McColley, who is a professor of pediatrics in pulmonary and sleep medicine at Northwestern University, Chicago.
The study adds more evidence that earlier treatment in CF may lead to better outcomes, but the digestive improvements are an overlooked factor, according to Dr. McColley. “Even with early treatment, and even with pancreatic enzymes and supplements taken to digest food, it’s a huge burden. When they come in and say that digestion seems better, there is less bloating, things like that, these are the things that aren’t captured so much in the clinical trial data, but they’re meaningful to families,” she said.
Single-agent ivacaftor is available for children as young as 4 months, but is limited to patients with the G551D gating mutation. Most young children with CF can only be treated for symptoms.
The lack of new safety signals in the new study is reassuring, and the research presents some hope to young children who are not yet eligible to receive ETI, according to Carlos Milla, MD, who is a pediatric pulmonary physician at Stanford (Calif.) University. “We already know that the next version of this drug is much more efficacious, the triple-combination therapy. It’s a little bit like we’re falling behind when it comes to treating these young kids because we are offering right now what we know is a less effective drug as opposed to the ones that are available now down to age 6, and hopefully sometime soon down to age 2. It’s better [than] to have no treatment at all, so it’s a good start,” said Dr. Milla.
“I think this is a great bridge for babies while they’re waiting to grow up to be old enough to get [triple combination therapy] and will prevent some of the complications until they can get the even more highly effective therapy in the future,” said Jennifer Taylor-Cousar, MD, who was asked to comment on the study. Dr. Taylor-Cousar is codirector of the Adult CF Program at National Jewish Health, Denver.
She also noted that the therapy could rapidly become more important. Since the approval of elexacaftor/tezacaftor/ivacaftor in 2019, pregnancies in women with CF have increased markedly. There were 310 such pregnancies in 2019, and 675 in 2020 after the combination became generally available in November of 2019. Many of the resulting babies had false-negative CF diagnoses because the mother was taking the triple combination and the medication crossed the placenta and prevented disease progression. The drugs are present in breast milk, but when breastfeeding isn’t possible, newborns are left without a therapeutic option. “There was no approval for babies who had two copies of F508. This helps tremendously with that albeit small population, although I suspect it may grow larger over the upcoming years as we continue to see so many pregnancies in women with CF because they are so much healthier,” said Dr. Taylor-Cousar.
The study was a phase 3, open-label trial with a cohort aged 18-24 months (cohort 1, n = 14) and another aged 12-18 months (cohort 2, n = 46). Participants received a 15-day treatment with a dose based on weight at screening. Participants then underwent a 24-week treatment period with a dose determined by pharmacokinetic data collected during the initial treatment, the authors wrote in the American Journal of Respiratory and Critical Care Medicine.
A total of 95.7% of children experienced adverse events during the 24-week treatment period; 52.2% of events were mild, and 39.1% were moderate. The most frequent adverse events were cough (34.8%), infective exacerbation of CF (21.7%), pyrexia (21.7%), and vomiting (17.4%); 10.9% had elevations of alanine aminotransferase and/or aspartate aminotransferase higher than three times the upper limit of normal, and one (2.2%) had concentrations of both high enough that the study drug was discontinued.
There were significant reductions in sweat chloride concentration at week 24, suggesting strong efficacy (–29.1 mmol/L; 95% confidence interval, –34.8 to –23.4 mmol/L). Body mass, weight, and length remained normal during the 24-week treatment period, and there were trends towards improvement in biomarkers of pancreatic function and intestinal inflammation, including fecal elastase-1 (+73.1mcg/g; 95% CI, 29.40-116.80 mcg/g), serum immunoreactive trypsinogen (–295.50 mcg/g; 95% CI, –416.60 to –174.50 mcg/g), and fecal calprotectin (–106.63 mg/kg; 95% CI, –180.60 to –32.66 mg/kg)
Dr. McColley, Dr. Taylor-Cousar, and Dr. Milla have no relevant financial disclosures. The study was funded by Merck.
In children with cystic fibrosis (CF) and homozygous for the F508 mutation, the combination of lumacaftor and ivacaftor appears generally safe, and biomarker data suggest its efficacy, according to results from a new open-label phase 3 trial.
In September, the Food and Drug Administration approved lumacaftor/ivacaftor for patients aged 1 years and above. Waiting in the wings is the triple combination of elexacaftor, tezacaftor, and ivacaftor (ETI), which is available for patients with at least one copy of the F508 mutation aged 6 and over. ETI is also being tested in younger patients.
One driving factor for early treatment is countering the malnutrition that can occur among CF patients because of poor pancreatic insufficiency and chronic inflammation. “We’ve known for many years that (being) at or above average body weight and height predict better lung function. And we’ve known for quite a while that the height-for-age percentile, in preschool years, actually predicts your lung function later, and how long you’re going to live, so nutrition is incredibly important,” said study author Susanna McColley, MD, in an interview. It’s also difficult to use lung function tests in young children, since even adults can find them challenging, she said.
“FEV1 [forced expiratory volume in 1 second] is the strongest predictor of survival, and then nutrition is the highest predictor of FEV1, so that’s kind of the construct. They had similar improvement in the functional measures of their pancreas and in the measures of inflammation in the gut. I think the story here is that starting a modulator early has a likelihood to have positive health effects that go forward. We can’t say that from the data in the paper. It’s a 24-week study, but looking at the pancreatic and intestinal functioning and also the fact that there was a decrease in sweat chloride is important,” said Dr. McColley, who is a professor of pediatrics in pulmonary and sleep medicine at Northwestern University, Chicago.
The study adds more evidence that earlier treatment in CF may lead to better outcomes, but the digestive improvements are an overlooked factor, according to Dr. McColley. “Even with early treatment, and even with pancreatic enzymes and supplements taken to digest food, it’s a huge burden. When they come in and say that digestion seems better, there is less bloating, things like that, these are the things that aren’t captured so much in the clinical trial data, but they’re meaningful to families,” she said.
Single-agent ivacaftor is available for children as young as 4 months, but is limited to patients with the G551D gating mutation. Most young children with CF can only be treated for symptoms.
The lack of new safety signals in the new study is reassuring, and the research presents some hope to young children who are not yet eligible to receive ETI, according to Carlos Milla, MD, who is a pediatric pulmonary physician at Stanford (Calif.) University. “We already know that the next version of this drug is much more efficacious, the triple-combination therapy. It’s a little bit like we’re falling behind when it comes to treating these young kids because we are offering right now what we know is a less effective drug as opposed to the ones that are available now down to age 6, and hopefully sometime soon down to age 2. It’s better [than] to have no treatment at all, so it’s a good start,” said Dr. Milla.
“I think this is a great bridge for babies while they’re waiting to grow up to be old enough to get [triple combination therapy] and will prevent some of the complications until they can get the even more highly effective therapy in the future,” said Jennifer Taylor-Cousar, MD, who was asked to comment on the study. Dr. Taylor-Cousar is codirector of the Adult CF Program at National Jewish Health, Denver.
She also noted that the therapy could rapidly become more important. Since the approval of elexacaftor/tezacaftor/ivacaftor in 2019, pregnancies in women with CF have increased markedly. There were 310 such pregnancies in 2019, and 675 in 2020 after the combination became generally available in November of 2019. Many of the resulting babies had false-negative CF diagnoses because the mother was taking the triple combination and the medication crossed the placenta and prevented disease progression. The drugs are present in breast milk, but when breastfeeding isn’t possible, newborns are left without a therapeutic option. “There was no approval for babies who had two copies of F508. This helps tremendously with that albeit small population, although I suspect it may grow larger over the upcoming years as we continue to see so many pregnancies in women with CF because they are so much healthier,” said Dr. Taylor-Cousar.
The study was a phase 3, open-label trial with a cohort aged 18-24 months (cohort 1, n = 14) and another aged 12-18 months (cohort 2, n = 46). Participants received a 15-day treatment with a dose based on weight at screening. Participants then underwent a 24-week treatment period with a dose determined by pharmacokinetic data collected during the initial treatment, the authors wrote in the American Journal of Respiratory and Critical Care Medicine.
A total of 95.7% of children experienced adverse events during the 24-week treatment period; 52.2% of events were mild, and 39.1% were moderate. The most frequent adverse events were cough (34.8%), infective exacerbation of CF (21.7%), pyrexia (21.7%), and vomiting (17.4%); 10.9% had elevations of alanine aminotransferase and/or aspartate aminotransferase higher than three times the upper limit of normal, and one (2.2%) had concentrations of both high enough that the study drug was discontinued.
There were significant reductions in sweat chloride concentration at week 24, suggesting strong efficacy (–29.1 mmol/L; 95% confidence interval, –34.8 to –23.4 mmol/L). Body mass, weight, and length remained normal during the 24-week treatment period, and there were trends towards improvement in biomarkers of pancreatic function and intestinal inflammation, including fecal elastase-1 (+73.1mcg/g; 95% CI, 29.40-116.80 mcg/g), serum immunoreactive trypsinogen (–295.50 mcg/g; 95% CI, –416.60 to –174.50 mcg/g), and fecal calprotectin (–106.63 mg/kg; 95% CI, –180.60 to –32.66 mg/kg)
Dr. McColley, Dr. Taylor-Cousar, and Dr. Milla have no relevant financial disclosures. The study was funded by Merck.
In children with cystic fibrosis (CF) and homozygous for the F508 mutation, the combination of lumacaftor and ivacaftor appears generally safe, and biomarker data suggest its efficacy, according to results from a new open-label phase 3 trial.
In September, the Food and Drug Administration approved lumacaftor/ivacaftor for patients aged 1 years and above. Waiting in the wings is the triple combination of elexacaftor, tezacaftor, and ivacaftor (ETI), which is available for patients with at least one copy of the F508 mutation aged 6 and over. ETI is also being tested in younger patients.
One driving factor for early treatment is countering the malnutrition that can occur among CF patients because of poor pancreatic insufficiency and chronic inflammation. “We’ve known for many years that (being) at or above average body weight and height predict better lung function. And we’ve known for quite a while that the height-for-age percentile, in preschool years, actually predicts your lung function later, and how long you’re going to live, so nutrition is incredibly important,” said study author Susanna McColley, MD, in an interview. It’s also difficult to use lung function tests in young children, since even adults can find them challenging, she said.
“FEV1 [forced expiratory volume in 1 second] is the strongest predictor of survival, and then nutrition is the highest predictor of FEV1, so that’s kind of the construct. They had similar improvement in the functional measures of their pancreas and in the measures of inflammation in the gut. I think the story here is that starting a modulator early has a likelihood to have positive health effects that go forward. We can’t say that from the data in the paper. It’s a 24-week study, but looking at the pancreatic and intestinal functioning and also the fact that there was a decrease in sweat chloride is important,” said Dr. McColley, who is a professor of pediatrics in pulmonary and sleep medicine at Northwestern University, Chicago.
The study adds more evidence that earlier treatment in CF may lead to better outcomes, but the digestive improvements are an overlooked factor, according to Dr. McColley. “Even with early treatment, and even with pancreatic enzymes and supplements taken to digest food, it’s a huge burden. When they come in and say that digestion seems better, there is less bloating, things like that, these are the things that aren’t captured so much in the clinical trial data, but they’re meaningful to families,” she said.
Single-agent ivacaftor is available for children as young as 4 months, but is limited to patients with the G551D gating mutation. Most young children with CF can only be treated for symptoms.
The lack of new safety signals in the new study is reassuring, and the research presents some hope to young children who are not yet eligible to receive ETI, according to Carlos Milla, MD, who is a pediatric pulmonary physician at Stanford (Calif.) University. “We already know that the next version of this drug is much more efficacious, the triple-combination therapy. It’s a little bit like we’re falling behind when it comes to treating these young kids because we are offering right now what we know is a less effective drug as opposed to the ones that are available now down to age 6, and hopefully sometime soon down to age 2. It’s better [than] to have no treatment at all, so it’s a good start,” said Dr. Milla.
“I think this is a great bridge for babies while they’re waiting to grow up to be old enough to get [triple combination therapy] and will prevent some of the complications until they can get the even more highly effective therapy in the future,” said Jennifer Taylor-Cousar, MD, who was asked to comment on the study. Dr. Taylor-Cousar is codirector of the Adult CF Program at National Jewish Health, Denver.
She also noted that the therapy could rapidly become more important. Since the approval of elexacaftor/tezacaftor/ivacaftor in 2019, pregnancies in women with CF have increased markedly. There were 310 such pregnancies in 2019, and 675 in 2020 after the combination became generally available in November of 2019. Many of the resulting babies had false-negative CF diagnoses because the mother was taking the triple combination and the medication crossed the placenta and prevented disease progression. The drugs are present in breast milk, but when breastfeeding isn’t possible, newborns are left without a therapeutic option. “There was no approval for babies who had two copies of F508. This helps tremendously with that albeit small population, although I suspect it may grow larger over the upcoming years as we continue to see so many pregnancies in women with CF because they are so much healthier,” said Dr. Taylor-Cousar.
The study was a phase 3, open-label trial with a cohort aged 18-24 months (cohort 1, n = 14) and another aged 12-18 months (cohort 2, n = 46). Participants received a 15-day treatment with a dose based on weight at screening. Participants then underwent a 24-week treatment period with a dose determined by pharmacokinetic data collected during the initial treatment, the authors wrote in the American Journal of Respiratory and Critical Care Medicine.
A total of 95.7% of children experienced adverse events during the 24-week treatment period; 52.2% of events were mild, and 39.1% were moderate. The most frequent adverse events were cough (34.8%), infective exacerbation of CF (21.7%), pyrexia (21.7%), and vomiting (17.4%); 10.9% had elevations of alanine aminotransferase and/or aspartate aminotransferase higher than three times the upper limit of normal, and one (2.2%) had concentrations of both high enough that the study drug was discontinued.
There were significant reductions in sweat chloride concentration at week 24, suggesting strong efficacy (–29.1 mmol/L; 95% confidence interval, –34.8 to –23.4 mmol/L). Body mass, weight, and length remained normal during the 24-week treatment period, and there were trends towards improvement in biomarkers of pancreatic function and intestinal inflammation, including fecal elastase-1 (+73.1mcg/g; 95% CI, 29.40-116.80 mcg/g), serum immunoreactive trypsinogen (–295.50 mcg/g; 95% CI, –416.60 to –174.50 mcg/g), and fecal calprotectin (–106.63 mg/kg; 95% CI, –180.60 to –32.66 mg/kg)
Dr. McColley, Dr. Taylor-Cousar, and Dr. Milla have no relevant financial disclosures. The study was funded by Merck.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Study evaluates features of alopecia areata in Hispanic/Latinx patients
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
.
Those are among key findings from a retrospective analysis of Hispanic/Latinx patients at the University of California, Irvine (UCI) by Natasha Mesinkovska, MD, PhD, of UCI’s department of dermatology, and her coauthors. The findings were published online in the Journal of the American Academy of Dermatology.
A recent study examined the epidemiology of alopecia areata (AA) in Black patients, wrote Dr. Mesinkovska and coauthors Celine Phong, a UCI medical student, and Amy J. McMichael, MD, professor of dermatology at Wake Forest University, Winston-Salem, N.C. “A similar unmet need exists to describe the characteristics of AA in Hispanic/Latinx (H/L) patients, the prevalent majority in California,” they added.
Drawing from chart reviews, ICD codes, and documented physical exams, they retrospectively identified 197 Hispanic/Latinx patients diagnosed with AA at UCI between 2015 and 2022, including alopecia totalis and alopecia universalis.
Nearly two-thirds of patients with alopecia were female (63%), and their mean age at diagnosis was 33 years. Most patients (79%) presented with patchy pattern AA, 13% had diffuse pattern AA, and only 12% had eyebrow, eyelash, or beard involvement. The most common comorbidity in patients overall was atopy (24%), including allergic rhinitis (12%), asthma (10%), and/or atopic dermatitis (7%).
The authors found that 18% of patients had one or more coexisting autoimmune conditions, most commonly rheumatoid arthritis (9%) and thyroid disease (6%). No patients had celiac disease, myasthenia gravis, or inflammatory bowel disease, but 43% had another dermatologic condition.
In other findings, 22% of patients had vitamin D deficiency, 20% had hyperlipidemia, 18% had obesity, 16% had gastroesophageal reflux disease, and 12% had anemia. At the same time, depression, anxiety, or sleep disorders were identified in 14% of patients.
“Interestingly, the most common autoimmune comorbidity in H/L was rheumatoid arthritis, compared to thyroid disease in Black patients and overall AA patients,” the authors wrote. “This finding may be a reflection of a larger trend, as rheumatoid arthritis in the H/L population has been on the rise.”
The authors acknowledged certain limitations of the study including its small sample size and lack of a control group, and reported having no financial disclosures.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Bad breath? Mouthwash is out. Yogurt is in.
Leave the mouthwash. Take the yogurt
Most of us have experienced some sort of bad breath. It’s common in the morning right after waking up, but it also may be a sign for underlying medical issues like dental problems or acid reflux. Wherever it comes from, we always want to get rid of it. A recent meta-analysis in BMJ Open may have found the answer in some common foods.
For those with halitosis, the basic problem is that the bacteria in their mouths are not happy about where they are. The researchers looked at 130 studies and found seven that suggested fermented food has some effect in combating bad breath.
Now when we say fermented food, we’re not talking about that science project waiting to happen in the back of the refrigerator. Think yogurt, sourdough bread, or miso soup. Anything that contains probiotic bacteria.
Matthew J. Messina, DDS, assistant professor of dentistry at Ohio State University, who was not involved with the study, told Healthline that “the whole idea behind probiotics is [bacteria replacement]. Supplant the ‘bad guys’ with the ‘good guys,’ then we’ll end up with a better result.” Essentially balancing the scales in your mouth.
It may not be a long-term solution, Dr. Messina said, but the short-term data are positive. So if you experience bad breath from time to time, try a little bowl of yogurt instead of chewing gum. If nothing else, the bacteria in your mouth will thank you.
You can talk the silly talk, but can you walk the silly walk?
The Ministry of Silly Walks sketch from Monty Python is an enduring comedy classic, and one of surprising relevance for doctors. After all, this isn’t the first time a study has analyzed the unusual strides of Mr. Putey and Mr. Teabag.
The BMJ Christmas edition truly is the gift that keeps on giving. For this plunge into the Flying Circus, the study authors recruited a small group of fairly average adults and had them walk normally around a track for 5 minutes, monitoring their oxygen intake and energy expenditure. After that, the study participants imitated Mr. Putey’s walk and then Mr. Teabag’s.
In the sketch, Mr. Teabag notes that Mr. Putey’s walk is “not particularly silly,” which is borne out in the research. When imitating Mr. Putey’s walk, oxygen intake and energy expenditure were barely higher than a normal walk, not enough to achieve a meaningful difference. Hopefully he’ll get that government grant to further develop his silly walk, because right now Mr. Putey’s walk simply doesn’t cut it.
Mr. Teabag’s walk is a different story and the very image of inefficiency. Oxygen intake was 2.5 times higher than during the normal walk, and energy expenditure was noticeably higher (8 kcal in men and 5.2 kcal in women). In fact, the walk was so inefficient and its effect so drastic it actually reached the level of vigorous exercise. Thanks to this, the study authors noted that just 11 minutes a day of walking like Mr. Teabag would be enough to reach the general goal of 75 minutes of vigorous exercise per week. Boosting that to 12-19 minutes would increase daily energy expenditure by 100 kcal.
The study authors wrote, “Had an initiative to promote inefficient movement been adopted in the early 1970s, we might now be living among a healthier society. Efforts to promote higher energy – and perhaps more joyful – walking should ensure inclusivity and inefficiency for all.” We think they just advocated for a real-life Ministry of Silly Walks. Well, there have been worse ideas. Just look at Twitter.
When efficient gut microbes go bad
With the latest news from the Ministry of Silly Walks, is it time for humans to embrace all things inefficient? Maybe.
Turns out that individuals with more efficient digestive systems – those that extract more energy from the fuel supplied to them by the busy mouths above – tend to gain more weight than those with less efficient guts, even when they eat the same food, according to a recent study published in Microbiome.
The researchers took a look at the composition of gut microbes in a group of 85 volunteers and found that about 40% had microbiomes dominated by Bacteroides bacteria, which are more effective at extracting nutrients from food. That group also weighed 10% more on average, amounting to an extra 9 kg.
In a rather blatant demonstration of efficiency, the investigators also measured the speed of the participants’ digestion, as they had hypothesized that those with the longest digestive travel times would be the ones who harvested the most nutrition from their food. That was not the case.
The study subjects with the most efficient gut bacteria “also have the fastest passage through the gastrointestinal system, which has given us something to think about,” senior author Henrik Roager of the University of Copenhagen said in a written statement.
You know what gives us something to think about? Stool energy density and intestinal transit time and faecal bacterial cell counts, that’s what. Ick. Sometimes science is gross.
Here’s another thought, though: Seeing faecal instead of fecal is kind of funny to our American eyes, but adding that extra letter is also inefficient, which could mean that it’s good. So, in the spirit of embracing the inefficient as a new year begins, we’re resolving to wrap our editorial arms around faecal and the faeces it represents. Well, not literally, of course. More like we’re embracing the spirit of faeces.
Leave the mouthwash. Take the yogurt
Most of us have experienced some sort of bad breath. It’s common in the morning right after waking up, but it also may be a sign for underlying medical issues like dental problems or acid reflux. Wherever it comes from, we always want to get rid of it. A recent meta-analysis in BMJ Open may have found the answer in some common foods.
For those with halitosis, the basic problem is that the bacteria in their mouths are not happy about where they are. The researchers looked at 130 studies and found seven that suggested fermented food has some effect in combating bad breath.
Now when we say fermented food, we’re not talking about that science project waiting to happen in the back of the refrigerator. Think yogurt, sourdough bread, or miso soup. Anything that contains probiotic bacteria.
Matthew J. Messina, DDS, assistant professor of dentistry at Ohio State University, who was not involved with the study, told Healthline that “the whole idea behind probiotics is [bacteria replacement]. Supplant the ‘bad guys’ with the ‘good guys,’ then we’ll end up with a better result.” Essentially balancing the scales in your mouth.
It may not be a long-term solution, Dr. Messina said, but the short-term data are positive. So if you experience bad breath from time to time, try a little bowl of yogurt instead of chewing gum. If nothing else, the bacteria in your mouth will thank you.
You can talk the silly talk, but can you walk the silly walk?
The Ministry of Silly Walks sketch from Monty Python is an enduring comedy classic, and one of surprising relevance for doctors. After all, this isn’t the first time a study has analyzed the unusual strides of Mr. Putey and Mr. Teabag.
The BMJ Christmas edition truly is the gift that keeps on giving. For this plunge into the Flying Circus, the study authors recruited a small group of fairly average adults and had them walk normally around a track for 5 minutes, monitoring their oxygen intake and energy expenditure. After that, the study participants imitated Mr. Putey’s walk and then Mr. Teabag’s.
In the sketch, Mr. Teabag notes that Mr. Putey’s walk is “not particularly silly,” which is borne out in the research. When imitating Mr. Putey’s walk, oxygen intake and energy expenditure were barely higher than a normal walk, not enough to achieve a meaningful difference. Hopefully he’ll get that government grant to further develop his silly walk, because right now Mr. Putey’s walk simply doesn’t cut it.
Mr. Teabag’s walk is a different story and the very image of inefficiency. Oxygen intake was 2.5 times higher than during the normal walk, and energy expenditure was noticeably higher (8 kcal in men and 5.2 kcal in women). In fact, the walk was so inefficient and its effect so drastic it actually reached the level of vigorous exercise. Thanks to this, the study authors noted that just 11 minutes a day of walking like Mr. Teabag would be enough to reach the general goal of 75 minutes of vigorous exercise per week. Boosting that to 12-19 minutes would increase daily energy expenditure by 100 kcal.
The study authors wrote, “Had an initiative to promote inefficient movement been adopted in the early 1970s, we might now be living among a healthier society. Efforts to promote higher energy – and perhaps more joyful – walking should ensure inclusivity and inefficiency for all.” We think they just advocated for a real-life Ministry of Silly Walks. Well, there have been worse ideas. Just look at Twitter.
When efficient gut microbes go bad
With the latest news from the Ministry of Silly Walks, is it time for humans to embrace all things inefficient? Maybe.
Turns out that individuals with more efficient digestive systems – those that extract more energy from the fuel supplied to them by the busy mouths above – tend to gain more weight than those with less efficient guts, even when they eat the same food, according to a recent study published in Microbiome.
The researchers took a look at the composition of gut microbes in a group of 85 volunteers and found that about 40% had microbiomes dominated by Bacteroides bacteria, which are more effective at extracting nutrients from food. That group also weighed 10% more on average, amounting to an extra 9 kg.
In a rather blatant demonstration of efficiency, the investigators also measured the speed of the participants’ digestion, as they had hypothesized that those with the longest digestive travel times would be the ones who harvested the most nutrition from their food. That was not the case.
The study subjects with the most efficient gut bacteria “also have the fastest passage through the gastrointestinal system, which has given us something to think about,” senior author Henrik Roager of the University of Copenhagen said in a written statement.
You know what gives us something to think about? Stool energy density and intestinal transit time and faecal bacterial cell counts, that’s what. Ick. Sometimes science is gross.
Here’s another thought, though: Seeing faecal instead of fecal is kind of funny to our American eyes, but adding that extra letter is also inefficient, which could mean that it’s good. So, in the spirit of embracing the inefficient as a new year begins, we’re resolving to wrap our editorial arms around faecal and the faeces it represents. Well, not literally, of course. More like we’re embracing the spirit of faeces.
Leave the mouthwash. Take the yogurt
Most of us have experienced some sort of bad breath. It’s common in the morning right after waking up, but it also may be a sign for underlying medical issues like dental problems or acid reflux. Wherever it comes from, we always want to get rid of it. A recent meta-analysis in BMJ Open may have found the answer in some common foods.
For those with halitosis, the basic problem is that the bacteria in their mouths are not happy about where they are. The researchers looked at 130 studies and found seven that suggested fermented food has some effect in combating bad breath.
Now when we say fermented food, we’re not talking about that science project waiting to happen in the back of the refrigerator. Think yogurt, sourdough bread, or miso soup. Anything that contains probiotic bacteria.
Matthew J. Messina, DDS, assistant professor of dentistry at Ohio State University, who was not involved with the study, told Healthline that “the whole idea behind probiotics is [bacteria replacement]. Supplant the ‘bad guys’ with the ‘good guys,’ then we’ll end up with a better result.” Essentially balancing the scales in your mouth.
It may not be a long-term solution, Dr. Messina said, but the short-term data are positive. So if you experience bad breath from time to time, try a little bowl of yogurt instead of chewing gum. If nothing else, the bacteria in your mouth will thank you.
You can talk the silly talk, but can you walk the silly walk?
The Ministry of Silly Walks sketch from Monty Python is an enduring comedy classic, and one of surprising relevance for doctors. After all, this isn’t the first time a study has analyzed the unusual strides of Mr. Putey and Mr. Teabag.
The BMJ Christmas edition truly is the gift that keeps on giving. For this plunge into the Flying Circus, the study authors recruited a small group of fairly average adults and had them walk normally around a track for 5 minutes, monitoring their oxygen intake and energy expenditure. After that, the study participants imitated Mr. Putey’s walk and then Mr. Teabag’s.
In the sketch, Mr. Teabag notes that Mr. Putey’s walk is “not particularly silly,” which is borne out in the research. When imitating Mr. Putey’s walk, oxygen intake and energy expenditure were barely higher than a normal walk, not enough to achieve a meaningful difference. Hopefully he’ll get that government grant to further develop his silly walk, because right now Mr. Putey’s walk simply doesn’t cut it.
Mr. Teabag’s walk is a different story and the very image of inefficiency. Oxygen intake was 2.5 times higher than during the normal walk, and energy expenditure was noticeably higher (8 kcal in men and 5.2 kcal in women). In fact, the walk was so inefficient and its effect so drastic it actually reached the level of vigorous exercise. Thanks to this, the study authors noted that just 11 minutes a day of walking like Mr. Teabag would be enough to reach the general goal of 75 minutes of vigorous exercise per week. Boosting that to 12-19 minutes would increase daily energy expenditure by 100 kcal.
The study authors wrote, “Had an initiative to promote inefficient movement been adopted in the early 1970s, we might now be living among a healthier society. Efforts to promote higher energy – and perhaps more joyful – walking should ensure inclusivity and inefficiency for all.” We think they just advocated for a real-life Ministry of Silly Walks. Well, there have been worse ideas. Just look at Twitter.
When efficient gut microbes go bad
With the latest news from the Ministry of Silly Walks, is it time for humans to embrace all things inefficient? Maybe.
Turns out that individuals with more efficient digestive systems – those that extract more energy from the fuel supplied to them by the busy mouths above – tend to gain more weight than those with less efficient guts, even when they eat the same food, according to a recent study published in Microbiome.
The researchers took a look at the composition of gut microbes in a group of 85 volunteers and found that about 40% had microbiomes dominated by Bacteroides bacteria, which are more effective at extracting nutrients from food. That group also weighed 10% more on average, amounting to an extra 9 kg.
In a rather blatant demonstration of efficiency, the investigators also measured the speed of the participants’ digestion, as they had hypothesized that those with the longest digestive travel times would be the ones who harvested the most nutrition from their food. That was not the case.
The study subjects with the most efficient gut bacteria “also have the fastest passage through the gastrointestinal system, which has given us something to think about,” senior author Henrik Roager of the University of Copenhagen said in a written statement.
You know what gives us something to think about? Stool energy density and intestinal transit time and faecal bacterial cell counts, that’s what. Ick. Sometimes science is gross.
Here’s another thought, though: Seeing faecal instead of fecal is kind of funny to our American eyes, but adding that extra letter is also inefficient, which could mean that it’s good. So, in the spirit of embracing the inefficient as a new year begins, we’re resolving to wrap our editorial arms around faecal and the faeces it represents. Well, not literally, of course. More like we’re embracing the spirit of faeces.