User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
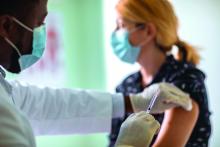
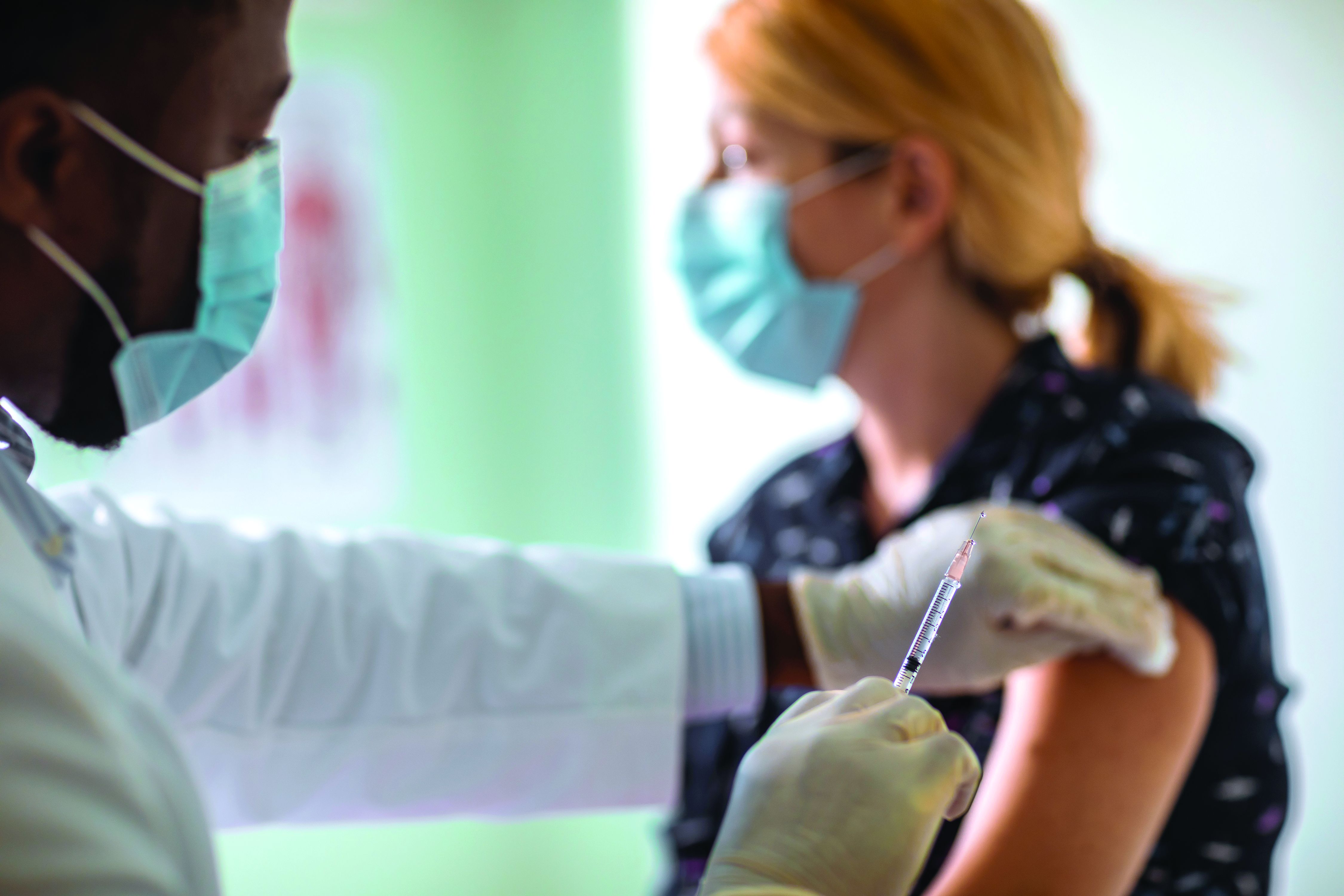
‘Empathy fatigue’ in clinicians rises with latest COVID-19 surge
Heidi Erickson, MD, is tired. As a pulmonary and critical care physician at Hennepin Healthcare in Minneapolis, she has been providing care for patients with COVID-19 since the start of the pandemic.
It was exhausting from the beginning, as she and her colleagues scrambled to understand how to deal with this new disease. But lately, she has noticed a different kind of exhaustion arising from the knowledge that with vaccines widely available, the latest surge was preventable.
Her intensive care unit is currently as full as it has ever been with COVID-19 patients, many of them young adults and most of them unvaccinated. After the recent death of one patient, an unvaccinated man with teenage children, she had to face his family’s questions about why ivermectin, an antiparasitic medication that was falsely promoted as a COVID-19 treatment, was not administered.
“I’m fatigued because I’m working more than ever, but more people don’t have to die,” Dr. Erickson said in an interview . “It’s been very hard physically, mentally, emotionally.”
Amid yet another surge in COVID-19 cases around the United States, clinicians are speaking out about their growing frustration with this preventable crisis.
Some are using the terms “empathy fatigue” and “compassion fatigue” – a sense that they are losing empathy for unvaccinated individuals who are fueling the pandemic.
Dr. Erickson says she is frustrated not by individual patients but by a system that has allowed disinformation to proliferate. Experts say these types of feelings fit into a widespread pattern of physician burnout that has taken a new turn at this stage of the pandemic.
Paradoxical choices
Empathy is a cornerstone of what clinicians do, and the ability to understand and share a patient’s feelings is an essential skill for providing effective care, says Kaz Nelson, MD, a psychiatrist at the University of Minnesota, Minneapolis.
Practitioners face paradoxical situations all the time, she notes. These include individuals who break bones and go skydiving again, people who have high cholesterol but continue to eat fried foods, and those with advanced lung cancer who continue to smoke.
To treat patients with compassion, practitioners learn to set aside judgment by acknowledging the complexity of human behavior. They may lament the addictive nature of nicotine and advertising that targets children, for example, while still listening and caring.
Empathy requires high-level brain function, but as stress levels rise, brain function that drives empathy tends to shut down. It’s a survival mechanism, Dr. Nelson says.
When health care workers feel overwhelmed, trapped, or threatened by patients demanding unproven treatments or by ICUs with more patients than ventilators, they may experience a fight-or-flight response that makes them defensive, frustrated, angry, or uncaring, notes Mona Masood, DO, a Philadelphia-area psychiatrist and founder of Physician Support Line, a free mental health hotline for doctors.
Some clinicians have taken to Twitter and other social media platforms to post about these types of experiences.
These feelings, which have been brewing for months, have been exacerbated by the complexity of the current situation. Clinicians see a disconnect between what is and what could be, Dr. Nelson notes.
“Prior to vaccines, there weren’t other options, and so we had toxic stress and we had fatigue, but we could still maintain little bits of empathy by saying, ‘You know, people didn’t choose to get infected, and we are in a pandemic.’ We could kind of hate the virus. Now with access to vaccines, that last connection to empathy is removed for many people,” she says.
Self-preservation vs. empathy
Compassion fatigue or empathy fatigue is just one reaction to feeling completely maxed out and overstressed, Dr. Nelson says. Anger at society, such as what Dr. Erickson experienced, is another response.
Practitioners may also feel as if they are just going through the motions of their job, or they might disassociate, ceasing to feel that their patients are human. Plenty of doctors and nurses have cried in their cars after shifts and have posted tearful videos on social media.
Early in the pandemic, Dr. Masood says, physicians who called the support hotline expressed sadness and grief. Now, she had her colleagues hear frustration and anger, along with guilt and shame for having feelings they believe they shouldn’t be having, especially toward patients. They may feel unprofessional or worse – unworthy of being physicians, she says.
One recent caller to the hotline was a long-time ICU physician who had been told so many times by patients that ivermectin was the only medicine that would cure them that he began to doubt himself, says Dr. Masood. This caller needed to be reassured by another physician that he was doing the right thing.
Another emergency department physician told Dr. Masood about a young child who had arrived at the hospital with COVID-19 symptoms. When asked whether the family had been exposed to anyone with COVID-19, the child’s parent lied so that they could be triaged faster.
The physician, who needed to step away from the situation, reached out to Dr. Masood to express her frustration so that she wouldn’t “let it out” on the patient.
“It’s hard to have empathy for people who, for all intents and purposes, are very self-centered,” Dr. Masood says. “We’re at a place where we’re having to choose between self-preservation and empathy.”
How to cope
To help practitioners cope, Dr. Masood offers words that describe what they’re experiencing. She often hears clinicians say things such as, “This is a type of burnout that I feel to my bones,” or “This makes me want to quit,” or “I feel like I’m at the end of my rope.”
She encourages them to consider the terms “empathy fatigue,” and “moral injury” in order to reconcile how their sense of responsibility to take care of people is compromised by factors outside of their control.
It is not shameful to acknowledge that they experience emotions, including difficult ones such as frustration, anger, sadness, and anxiety, Dr. Masood adds.
Being frustrated with a patient doesn’t make someone a bad doctor, and admitting those emotions is the first step toward dealing with them, she says.
before they cause a sense of callousness or other consequences that become harder to heal from as time goes on.
“We’re trained to just go, go, go and sometimes not pause and check in,” she says. Clinicians who open up are likely to find they are not the only ones feeling tired or frustrated right now, she adds.
“Connect with peers and colleagues, because chances are, they can relate,” Dr. Nelson says.
A version of this article first appeared on Medscape.com.
Heidi Erickson, MD, is tired. As a pulmonary and critical care physician at Hennepin Healthcare in Minneapolis, she has been providing care for patients with COVID-19 since the start of the pandemic.
It was exhausting from the beginning, as she and her colleagues scrambled to understand how to deal with this new disease. But lately, she has noticed a different kind of exhaustion arising from the knowledge that with vaccines widely available, the latest surge was preventable.
Her intensive care unit is currently as full as it has ever been with COVID-19 patients, many of them young adults and most of them unvaccinated. After the recent death of one patient, an unvaccinated man with teenage children, she had to face his family’s questions about why ivermectin, an antiparasitic medication that was falsely promoted as a COVID-19 treatment, was not administered.
“I’m fatigued because I’m working more than ever, but more people don’t have to die,” Dr. Erickson said in an interview . “It’s been very hard physically, mentally, emotionally.”
Amid yet another surge in COVID-19 cases around the United States, clinicians are speaking out about their growing frustration with this preventable crisis.
Some are using the terms “empathy fatigue” and “compassion fatigue” – a sense that they are losing empathy for unvaccinated individuals who are fueling the pandemic.
Dr. Erickson says she is frustrated not by individual patients but by a system that has allowed disinformation to proliferate. Experts say these types of feelings fit into a widespread pattern of physician burnout that has taken a new turn at this stage of the pandemic.
Paradoxical choices
Empathy is a cornerstone of what clinicians do, and the ability to understand and share a patient’s feelings is an essential skill for providing effective care, says Kaz Nelson, MD, a psychiatrist at the University of Minnesota, Minneapolis.
Practitioners face paradoxical situations all the time, she notes. These include individuals who break bones and go skydiving again, people who have high cholesterol but continue to eat fried foods, and those with advanced lung cancer who continue to smoke.
To treat patients with compassion, practitioners learn to set aside judgment by acknowledging the complexity of human behavior. They may lament the addictive nature of nicotine and advertising that targets children, for example, while still listening and caring.
Empathy requires high-level brain function, but as stress levels rise, brain function that drives empathy tends to shut down. It’s a survival mechanism, Dr. Nelson says.
When health care workers feel overwhelmed, trapped, or threatened by patients demanding unproven treatments or by ICUs with more patients than ventilators, they may experience a fight-or-flight response that makes them defensive, frustrated, angry, or uncaring, notes Mona Masood, DO, a Philadelphia-area psychiatrist and founder of Physician Support Line, a free mental health hotline for doctors.
Some clinicians have taken to Twitter and other social media platforms to post about these types of experiences.
These feelings, which have been brewing for months, have been exacerbated by the complexity of the current situation. Clinicians see a disconnect between what is and what could be, Dr. Nelson notes.
“Prior to vaccines, there weren’t other options, and so we had toxic stress and we had fatigue, but we could still maintain little bits of empathy by saying, ‘You know, people didn’t choose to get infected, and we are in a pandemic.’ We could kind of hate the virus. Now with access to vaccines, that last connection to empathy is removed for many people,” she says.
Self-preservation vs. empathy
Compassion fatigue or empathy fatigue is just one reaction to feeling completely maxed out and overstressed, Dr. Nelson says. Anger at society, such as what Dr. Erickson experienced, is another response.
Practitioners may also feel as if they are just going through the motions of their job, or they might disassociate, ceasing to feel that their patients are human. Plenty of doctors and nurses have cried in their cars after shifts and have posted tearful videos on social media.
Early in the pandemic, Dr. Masood says, physicians who called the support hotline expressed sadness and grief. Now, she had her colleagues hear frustration and anger, along with guilt and shame for having feelings they believe they shouldn’t be having, especially toward patients. They may feel unprofessional or worse – unworthy of being physicians, she says.
One recent caller to the hotline was a long-time ICU physician who had been told so many times by patients that ivermectin was the only medicine that would cure them that he began to doubt himself, says Dr. Masood. This caller needed to be reassured by another physician that he was doing the right thing.
Another emergency department physician told Dr. Masood about a young child who had arrived at the hospital with COVID-19 symptoms. When asked whether the family had been exposed to anyone with COVID-19, the child’s parent lied so that they could be triaged faster.
The physician, who needed to step away from the situation, reached out to Dr. Masood to express her frustration so that she wouldn’t “let it out” on the patient.
“It’s hard to have empathy for people who, for all intents and purposes, are very self-centered,” Dr. Masood says. “We’re at a place where we’re having to choose between self-preservation and empathy.”
How to cope
To help practitioners cope, Dr. Masood offers words that describe what they’re experiencing. She often hears clinicians say things such as, “This is a type of burnout that I feel to my bones,” or “This makes me want to quit,” or “I feel like I’m at the end of my rope.”
She encourages them to consider the terms “empathy fatigue,” and “moral injury” in order to reconcile how their sense of responsibility to take care of people is compromised by factors outside of their control.
It is not shameful to acknowledge that they experience emotions, including difficult ones such as frustration, anger, sadness, and anxiety, Dr. Masood adds.
Being frustrated with a patient doesn’t make someone a bad doctor, and admitting those emotions is the first step toward dealing with them, she says.
before they cause a sense of callousness or other consequences that become harder to heal from as time goes on.
“We’re trained to just go, go, go and sometimes not pause and check in,” she says. Clinicians who open up are likely to find they are not the only ones feeling tired or frustrated right now, she adds.
“Connect with peers and colleagues, because chances are, they can relate,” Dr. Nelson says.
A version of this article first appeared on Medscape.com.
Heidi Erickson, MD, is tired. As a pulmonary and critical care physician at Hennepin Healthcare in Minneapolis, she has been providing care for patients with COVID-19 since the start of the pandemic.
It was exhausting from the beginning, as she and her colleagues scrambled to understand how to deal with this new disease. But lately, she has noticed a different kind of exhaustion arising from the knowledge that with vaccines widely available, the latest surge was preventable.
Her intensive care unit is currently as full as it has ever been with COVID-19 patients, many of them young adults and most of them unvaccinated. After the recent death of one patient, an unvaccinated man with teenage children, she had to face his family’s questions about why ivermectin, an antiparasitic medication that was falsely promoted as a COVID-19 treatment, was not administered.
“I’m fatigued because I’m working more than ever, but more people don’t have to die,” Dr. Erickson said in an interview . “It’s been very hard physically, mentally, emotionally.”
Amid yet another surge in COVID-19 cases around the United States, clinicians are speaking out about their growing frustration with this preventable crisis.
Some are using the terms “empathy fatigue” and “compassion fatigue” – a sense that they are losing empathy for unvaccinated individuals who are fueling the pandemic.
Dr. Erickson says she is frustrated not by individual patients but by a system that has allowed disinformation to proliferate. Experts say these types of feelings fit into a widespread pattern of physician burnout that has taken a new turn at this stage of the pandemic.
Paradoxical choices
Empathy is a cornerstone of what clinicians do, and the ability to understand and share a patient’s feelings is an essential skill for providing effective care, says Kaz Nelson, MD, a psychiatrist at the University of Minnesota, Minneapolis.
Practitioners face paradoxical situations all the time, she notes. These include individuals who break bones and go skydiving again, people who have high cholesterol but continue to eat fried foods, and those with advanced lung cancer who continue to smoke.
To treat patients with compassion, practitioners learn to set aside judgment by acknowledging the complexity of human behavior. They may lament the addictive nature of nicotine and advertising that targets children, for example, while still listening and caring.
Empathy requires high-level brain function, but as stress levels rise, brain function that drives empathy tends to shut down. It’s a survival mechanism, Dr. Nelson says.
When health care workers feel overwhelmed, trapped, or threatened by patients demanding unproven treatments or by ICUs with more patients than ventilators, they may experience a fight-or-flight response that makes them defensive, frustrated, angry, or uncaring, notes Mona Masood, DO, a Philadelphia-area psychiatrist and founder of Physician Support Line, a free mental health hotline for doctors.
Some clinicians have taken to Twitter and other social media platforms to post about these types of experiences.
These feelings, which have been brewing for months, have been exacerbated by the complexity of the current situation. Clinicians see a disconnect between what is and what could be, Dr. Nelson notes.
“Prior to vaccines, there weren’t other options, and so we had toxic stress and we had fatigue, but we could still maintain little bits of empathy by saying, ‘You know, people didn’t choose to get infected, and we are in a pandemic.’ We could kind of hate the virus. Now with access to vaccines, that last connection to empathy is removed for many people,” she says.
Self-preservation vs. empathy
Compassion fatigue or empathy fatigue is just one reaction to feeling completely maxed out and overstressed, Dr. Nelson says. Anger at society, such as what Dr. Erickson experienced, is another response.
Practitioners may also feel as if they are just going through the motions of their job, or they might disassociate, ceasing to feel that their patients are human. Plenty of doctors and nurses have cried in their cars after shifts and have posted tearful videos on social media.
Early in the pandemic, Dr. Masood says, physicians who called the support hotline expressed sadness and grief. Now, she had her colleagues hear frustration and anger, along with guilt and shame for having feelings they believe they shouldn’t be having, especially toward patients. They may feel unprofessional or worse – unworthy of being physicians, she says.
One recent caller to the hotline was a long-time ICU physician who had been told so many times by patients that ivermectin was the only medicine that would cure them that he began to doubt himself, says Dr. Masood. This caller needed to be reassured by another physician that he was doing the right thing.
Another emergency department physician told Dr. Masood about a young child who had arrived at the hospital with COVID-19 symptoms. When asked whether the family had been exposed to anyone with COVID-19, the child’s parent lied so that they could be triaged faster.
The physician, who needed to step away from the situation, reached out to Dr. Masood to express her frustration so that she wouldn’t “let it out” on the patient.
“It’s hard to have empathy for people who, for all intents and purposes, are very self-centered,” Dr. Masood says. “We’re at a place where we’re having to choose between self-preservation and empathy.”
How to cope
To help practitioners cope, Dr. Masood offers words that describe what they’re experiencing. She often hears clinicians say things such as, “This is a type of burnout that I feel to my bones,” or “This makes me want to quit,” or “I feel like I’m at the end of my rope.”
She encourages them to consider the terms “empathy fatigue,” and “moral injury” in order to reconcile how their sense of responsibility to take care of people is compromised by factors outside of their control.
It is not shameful to acknowledge that they experience emotions, including difficult ones such as frustration, anger, sadness, and anxiety, Dr. Masood adds.
Being frustrated with a patient doesn’t make someone a bad doctor, and admitting those emotions is the first step toward dealing with them, she says.
before they cause a sense of callousness or other consequences that become harder to heal from as time goes on.
“We’re trained to just go, go, go and sometimes not pause and check in,” she says. Clinicians who open up are likely to find they are not the only ones feeling tired or frustrated right now, she adds.
“Connect with peers and colleagues, because chances are, they can relate,” Dr. Nelson says.
A version of this article first appeared on Medscape.com.
Finding employees during a pandemic
.
My own office is prime example: I have had job listings for both front- and back-office positions posted on all the major job boards and other employment portals for months, with a disappointing response. Of the few who do respond, many, incredibly, do not show up for their interviews!
It turns out that this is a widespread problem, and not just in medicine. A recent survey by the National Federation of Independent Business found that 42% of business owners, in all walks of life, had job openings that could not be filled, a record high. Over 90% of those hiring reported few or no qualified applicants and an increase in interview no-shows.
Clearly, this is a huge obstacle to growth – and even to conducting normal operations – for my practice and many others.
Reasons for the situation vary, but a big one has been the unfortunate fact that many open job positions actually pay less than the expanded unemployment benefits that many people have received under the March 2020 CARES Act. By one estimate, almost 70% of unemployed workers have been collecting more on unemployment than they earned while working. The CARES benefits expired in early September, but many potential workers continue to receive payments through a newer FEMA program, and some states have their own ongoing benefit programs.
Other reasons have been offered: Some candidates are unvaccinated (an immediate deal-breaker in my office), and some working parents continue to face a lack of childcare or in-person schooling for their children. Some applicants – regardless of vaccination status – have said they are hesitant to work in a medical office setting and risk getting COVID-19, despite all the precautions we have in place. Others have said they are waiting until the job market improves.
There are no easy solutions to this complicated problem, but here are a few suggestions culled from my research and conversations with HR professionals and others.
One obvious option is to offer higher wages, and perhaps even signing bonuses. “Whenever anyone says they can’t find the workers they need,” a consultant told me, “they are really saying they can’t find them at the wages they want to pay.” There are limits to the wages and benefits a private office with a very finite salary budget can offer, of course – but a few higher-paid employees may be preferable to no new workers at all.
For job candidates who fear COVID-19 exposure, assure them that their health and safety is a priority by spelling out the procedures your office is following (social distancing, reduced patient capacity, interaction barriers, face masks, avoidance of handshakes, enhanced cleaning procedures, symptom questionnaires, temperature checks, etc.) to minimize the risk of exposure.
You also may need to rework your interview process. In the Zoom era, most preliminary interviews can be conducted remotely. For on-site interviews, explain how you’re maintaining a safe interview environment by applying the same office safety policies to interactions with interviewees.
If a promising candidate doesn’t show up for an interview, the applicant could be making a token effort to obtain a job in order to perpetuate unemployment payments, but don’t jump to that conclusion. There may be extenuating circumstances, such as an emergency, illness, or traffic issues. Also, consider the possibility that it was your fault. If you waited too long to schedule the interview, another office could have lured them away. Or you may not have adequately explained your COVID-19 exposure safeguards. At the very least, a drawn-out process or a lack of transparency can make applicants apprehensive about accepting a job with you, particularly if other employers are pursuing them.
To counter the shortsighted appeal of collecting unemployment benefits, it may help to highlight the long-term growth opportunities available at your office. Consider outlining typical career tracks, or providing specific examples of how people have advanced their careers at your facility. I frequently cite the example of my current office manager, who began as an assistant receptionist almost 30 years ago.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
.
My own office is prime example: I have had job listings for both front- and back-office positions posted on all the major job boards and other employment portals for months, with a disappointing response. Of the few who do respond, many, incredibly, do not show up for their interviews!
It turns out that this is a widespread problem, and not just in medicine. A recent survey by the National Federation of Independent Business found that 42% of business owners, in all walks of life, had job openings that could not be filled, a record high. Over 90% of those hiring reported few or no qualified applicants and an increase in interview no-shows.
Clearly, this is a huge obstacle to growth – and even to conducting normal operations – for my practice and many others.
Reasons for the situation vary, but a big one has been the unfortunate fact that many open job positions actually pay less than the expanded unemployment benefits that many people have received under the March 2020 CARES Act. By one estimate, almost 70% of unemployed workers have been collecting more on unemployment than they earned while working. The CARES benefits expired in early September, but many potential workers continue to receive payments through a newer FEMA program, and some states have their own ongoing benefit programs.
Other reasons have been offered: Some candidates are unvaccinated (an immediate deal-breaker in my office), and some working parents continue to face a lack of childcare or in-person schooling for their children. Some applicants – regardless of vaccination status – have said they are hesitant to work in a medical office setting and risk getting COVID-19, despite all the precautions we have in place. Others have said they are waiting until the job market improves.
There are no easy solutions to this complicated problem, but here are a few suggestions culled from my research and conversations with HR professionals and others.
One obvious option is to offer higher wages, and perhaps even signing bonuses. “Whenever anyone says they can’t find the workers they need,” a consultant told me, “they are really saying they can’t find them at the wages they want to pay.” There are limits to the wages and benefits a private office with a very finite salary budget can offer, of course – but a few higher-paid employees may be preferable to no new workers at all.
For job candidates who fear COVID-19 exposure, assure them that their health and safety is a priority by spelling out the procedures your office is following (social distancing, reduced patient capacity, interaction barriers, face masks, avoidance of handshakes, enhanced cleaning procedures, symptom questionnaires, temperature checks, etc.) to minimize the risk of exposure.
You also may need to rework your interview process. In the Zoom era, most preliminary interviews can be conducted remotely. For on-site interviews, explain how you’re maintaining a safe interview environment by applying the same office safety policies to interactions with interviewees.
If a promising candidate doesn’t show up for an interview, the applicant could be making a token effort to obtain a job in order to perpetuate unemployment payments, but don’t jump to that conclusion. There may be extenuating circumstances, such as an emergency, illness, or traffic issues. Also, consider the possibility that it was your fault. If you waited too long to schedule the interview, another office could have lured them away. Or you may not have adequately explained your COVID-19 exposure safeguards. At the very least, a drawn-out process or a lack of transparency can make applicants apprehensive about accepting a job with you, particularly if other employers are pursuing them.
To counter the shortsighted appeal of collecting unemployment benefits, it may help to highlight the long-term growth opportunities available at your office. Consider outlining typical career tracks, or providing specific examples of how people have advanced their careers at your facility. I frequently cite the example of my current office manager, who began as an assistant receptionist almost 30 years ago.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
.
My own office is prime example: I have had job listings for both front- and back-office positions posted on all the major job boards and other employment portals for months, with a disappointing response. Of the few who do respond, many, incredibly, do not show up for their interviews!
It turns out that this is a widespread problem, and not just in medicine. A recent survey by the National Federation of Independent Business found that 42% of business owners, in all walks of life, had job openings that could not be filled, a record high. Over 90% of those hiring reported few or no qualified applicants and an increase in interview no-shows.
Clearly, this is a huge obstacle to growth – and even to conducting normal operations – for my practice and many others.
Reasons for the situation vary, but a big one has been the unfortunate fact that many open job positions actually pay less than the expanded unemployment benefits that many people have received under the March 2020 CARES Act. By one estimate, almost 70% of unemployed workers have been collecting more on unemployment than they earned while working. The CARES benefits expired in early September, but many potential workers continue to receive payments through a newer FEMA program, and some states have their own ongoing benefit programs.
Other reasons have been offered: Some candidates are unvaccinated (an immediate deal-breaker in my office), and some working parents continue to face a lack of childcare or in-person schooling for their children. Some applicants – regardless of vaccination status – have said they are hesitant to work in a medical office setting and risk getting COVID-19, despite all the precautions we have in place. Others have said they are waiting until the job market improves.
There are no easy solutions to this complicated problem, but here are a few suggestions culled from my research and conversations with HR professionals and others.
One obvious option is to offer higher wages, and perhaps even signing bonuses. “Whenever anyone says they can’t find the workers they need,” a consultant told me, “they are really saying they can’t find them at the wages they want to pay.” There are limits to the wages and benefits a private office with a very finite salary budget can offer, of course – but a few higher-paid employees may be preferable to no new workers at all.
For job candidates who fear COVID-19 exposure, assure them that their health and safety is a priority by spelling out the procedures your office is following (social distancing, reduced patient capacity, interaction barriers, face masks, avoidance of handshakes, enhanced cleaning procedures, symptom questionnaires, temperature checks, etc.) to minimize the risk of exposure.
You also may need to rework your interview process. In the Zoom era, most preliminary interviews can be conducted remotely. For on-site interviews, explain how you’re maintaining a safe interview environment by applying the same office safety policies to interactions with interviewees.
If a promising candidate doesn’t show up for an interview, the applicant could be making a token effort to obtain a job in order to perpetuate unemployment payments, but don’t jump to that conclusion. There may be extenuating circumstances, such as an emergency, illness, or traffic issues. Also, consider the possibility that it was your fault. If you waited too long to schedule the interview, another office could have lured them away. Or you may not have adequately explained your COVID-19 exposure safeguards. At the very least, a drawn-out process or a lack of transparency can make applicants apprehensive about accepting a job with you, particularly if other employers are pursuing them.
To counter the shortsighted appeal of collecting unemployment benefits, it may help to highlight the long-term growth opportunities available at your office. Consider outlining typical career tracks, or providing specific examples of how people have advanced their careers at your facility. I frequently cite the example of my current office manager, who began as an assistant receptionist almost 30 years ago.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
HPV infection during pregnancy ups risk of premature birth
Persistent human papillomavirus (HPV) 16 and HPV 18 during a pregnancy may be associated with an increased risk of premature birth.
Findings published online in JAMA Network Open found that 15.9% of individuals who had a persistent HPV 16 or 18 infection during the first and third trimesters of their pregnancy gave birth prematurely, compared with 5.6% of those who did not have an HPV infection at all.
The findings prompted the question of “the pathophysiology of HPV in pregnancy and how the virus is affecting the placenta,” said Lisette Davidson Tanner, MD, MPH, FACOG, who was not involved in the study.
Researchers said the findings are the first to show the association between preterm birth and HPV, which is an incurable virus that most sexually active individuals will get at some point in their lives, according to the Centers for Disease Control and Prevention.
“The results of this study are very important in helping us understand the burden caused by HPV in pregnancy,” study author Helen Trottier, MSc, PhD, researcher at the Centre Hospitalier Universitaire Sainte-Justine, said in an interview. “We may have just pinpointed an important cause of preterm birth that has so far been unexplained.”
Dr. Trottier and colleagues examined data from 1,052 pregnant women from three university-affiliated health care centers in Montreal between Nov. 8, 2010, and Oct. 16, 2016.
Only 5.6% of those who did not have an HPV infection had a premature birth, compared with 6.9% of those who tested positive for any HPV infection in the first trimester.
When looking at the first trimester, researchers found 12% of those diagnosed with HPV 16 and 18 had a preterm birth, compared to 4.9% of those who had a high-risk HPV infection other than HPV 16/18. When looking at the third trimester, researchers found that 15.9% of those with HPV 16/18 had an increased risk of giving birth prematurely, compared to those who had other high-risk HPV infections.
When researchers looked at the persistence of these HPV infections, they found that most HPV infections detected in the first trimester persist to the third trimester. The findings also revealed that persistent vaginal HPV 16/18 detection was significantly associated with all preterm births and spontaneous preterm births. This association was also found among those who had HPV infections detected in their placentas.
Meanwhile, 5.8% of those who had an HPV infection only during the first trimester experienced a preterm birth.
The researchers also found that HPV infections were frequent in pregnancy even among populations “considered to be at low risk based on sociodemographic and sexual history characteristics,” they wrote. Dr. Trottier said she hopes the findings will strengthen support for HPV vaccination.
Dr. Trottier’s study adds to a growing body of research regarding the adverse effects of HPV, according to Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta. “It is already well known that HPV is associated with a number of anogenital and oropharyngeal cancers,” Dr. Tanner said in an interview. “The potential association with preterm birth only adds weight to the recommendations to screen for and prevent HPV infection.”
HPV 16 and 18 are high-risk types that cause about 70% of cervical cancers and precancerous cervical lesions, according to the World Health Organization. However, there are three HPV vaccines – 9-valent HPV vaccine (Gardasil), quadrivalent HPV vaccine (Gardasil®, 4vHPV), and bivalent HPV vaccine (Cervarix) – that help protect against HPV 16/18.
The findings strengthen the benefits of HPV vaccination, Dr. Trottier explained. “There is no cure when the HPV infection is present,” Dr. Trottier said. “If the link [between preterm birth and HPV infections] is indeed causal, we can expect a greater risk of preterm delivery in these women. The effective tool we have is the HPV vaccination, but it should ideally be received before the start of sexual activity in order to prevent future infections that could occur in women.”
The American College of Obstetricians and Gynecologists recommends HPV vaccination for girls and women between the ages of 11 and 26 years old. However, Dr. Tanner said, women aged 27-45 who were previously unvaccinated may still receive benefit from the vaccine.
“Despite the known efficacy of the vaccine, only 50% of patients are up to date with their HPV vaccination,” Dr. Tanner explained. “This study further highlights the need to educate and encourage patients to be vaccinated.”
The researchers said future studies should investigate the association of HPV vaccination and vaccination programs with the risk of preterm birth.
The experts disclosed no conflicts of interest.
Persistent human papillomavirus (HPV) 16 and HPV 18 during a pregnancy may be associated with an increased risk of premature birth.
Findings published online in JAMA Network Open found that 15.9% of individuals who had a persistent HPV 16 or 18 infection during the first and third trimesters of their pregnancy gave birth prematurely, compared with 5.6% of those who did not have an HPV infection at all.
The findings prompted the question of “the pathophysiology of HPV in pregnancy and how the virus is affecting the placenta,” said Lisette Davidson Tanner, MD, MPH, FACOG, who was not involved in the study.
Researchers said the findings are the first to show the association between preterm birth and HPV, which is an incurable virus that most sexually active individuals will get at some point in their lives, according to the Centers for Disease Control and Prevention.
“The results of this study are very important in helping us understand the burden caused by HPV in pregnancy,” study author Helen Trottier, MSc, PhD, researcher at the Centre Hospitalier Universitaire Sainte-Justine, said in an interview. “We may have just pinpointed an important cause of preterm birth that has so far been unexplained.”
Dr. Trottier and colleagues examined data from 1,052 pregnant women from three university-affiliated health care centers in Montreal between Nov. 8, 2010, and Oct. 16, 2016.
Only 5.6% of those who did not have an HPV infection had a premature birth, compared with 6.9% of those who tested positive for any HPV infection in the first trimester.
When looking at the first trimester, researchers found 12% of those diagnosed with HPV 16 and 18 had a preterm birth, compared to 4.9% of those who had a high-risk HPV infection other than HPV 16/18. When looking at the third trimester, researchers found that 15.9% of those with HPV 16/18 had an increased risk of giving birth prematurely, compared to those who had other high-risk HPV infections.
When researchers looked at the persistence of these HPV infections, they found that most HPV infections detected in the first trimester persist to the third trimester. The findings also revealed that persistent vaginal HPV 16/18 detection was significantly associated with all preterm births and spontaneous preterm births. This association was also found among those who had HPV infections detected in their placentas.
Meanwhile, 5.8% of those who had an HPV infection only during the first trimester experienced a preterm birth.
The researchers also found that HPV infections were frequent in pregnancy even among populations “considered to be at low risk based on sociodemographic and sexual history characteristics,” they wrote. Dr. Trottier said she hopes the findings will strengthen support for HPV vaccination.
Dr. Trottier’s study adds to a growing body of research regarding the adverse effects of HPV, according to Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta. “It is already well known that HPV is associated with a number of anogenital and oropharyngeal cancers,” Dr. Tanner said in an interview. “The potential association with preterm birth only adds weight to the recommendations to screen for and prevent HPV infection.”
HPV 16 and 18 are high-risk types that cause about 70% of cervical cancers and precancerous cervical lesions, according to the World Health Organization. However, there are three HPV vaccines – 9-valent HPV vaccine (Gardasil), quadrivalent HPV vaccine (Gardasil®, 4vHPV), and bivalent HPV vaccine (Cervarix) – that help protect against HPV 16/18.
The findings strengthen the benefits of HPV vaccination, Dr. Trottier explained. “There is no cure when the HPV infection is present,” Dr. Trottier said. “If the link [between preterm birth and HPV infections] is indeed causal, we can expect a greater risk of preterm delivery in these women. The effective tool we have is the HPV vaccination, but it should ideally be received before the start of sexual activity in order to prevent future infections that could occur in women.”
The American College of Obstetricians and Gynecologists recommends HPV vaccination for girls and women between the ages of 11 and 26 years old. However, Dr. Tanner said, women aged 27-45 who were previously unvaccinated may still receive benefit from the vaccine.
“Despite the known efficacy of the vaccine, only 50% of patients are up to date with their HPV vaccination,” Dr. Tanner explained. “This study further highlights the need to educate and encourage patients to be vaccinated.”
The researchers said future studies should investigate the association of HPV vaccination and vaccination programs with the risk of preterm birth.
The experts disclosed no conflicts of interest.
Persistent human papillomavirus (HPV) 16 and HPV 18 during a pregnancy may be associated with an increased risk of premature birth.
Findings published online in JAMA Network Open found that 15.9% of individuals who had a persistent HPV 16 or 18 infection during the first and third trimesters of their pregnancy gave birth prematurely, compared with 5.6% of those who did not have an HPV infection at all.
The findings prompted the question of “the pathophysiology of HPV in pregnancy and how the virus is affecting the placenta,” said Lisette Davidson Tanner, MD, MPH, FACOG, who was not involved in the study.
Researchers said the findings are the first to show the association between preterm birth and HPV, which is an incurable virus that most sexually active individuals will get at some point in their lives, according to the Centers for Disease Control and Prevention.
“The results of this study are very important in helping us understand the burden caused by HPV in pregnancy,” study author Helen Trottier, MSc, PhD, researcher at the Centre Hospitalier Universitaire Sainte-Justine, said in an interview. “We may have just pinpointed an important cause of preterm birth that has so far been unexplained.”
Dr. Trottier and colleagues examined data from 1,052 pregnant women from three university-affiliated health care centers in Montreal between Nov. 8, 2010, and Oct. 16, 2016.
Only 5.6% of those who did not have an HPV infection had a premature birth, compared with 6.9% of those who tested positive for any HPV infection in the first trimester.
When looking at the first trimester, researchers found 12% of those diagnosed with HPV 16 and 18 had a preterm birth, compared to 4.9% of those who had a high-risk HPV infection other than HPV 16/18. When looking at the third trimester, researchers found that 15.9% of those with HPV 16/18 had an increased risk of giving birth prematurely, compared to those who had other high-risk HPV infections.
When researchers looked at the persistence of these HPV infections, they found that most HPV infections detected in the first trimester persist to the third trimester. The findings also revealed that persistent vaginal HPV 16/18 detection was significantly associated with all preterm births and spontaneous preterm births. This association was also found among those who had HPV infections detected in their placentas.
Meanwhile, 5.8% of those who had an HPV infection only during the first trimester experienced a preterm birth.
The researchers also found that HPV infections were frequent in pregnancy even among populations “considered to be at low risk based on sociodemographic and sexual history characteristics,” they wrote. Dr. Trottier said she hopes the findings will strengthen support for HPV vaccination.
Dr. Trottier’s study adds to a growing body of research regarding the adverse effects of HPV, according to Dr. Tanner, assistant professor of gynecology and obstetrics at Emory University, Atlanta. “It is already well known that HPV is associated with a number of anogenital and oropharyngeal cancers,” Dr. Tanner said in an interview. “The potential association with preterm birth only adds weight to the recommendations to screen for and prevent HPV infection.”
HPV 16 and 18 are high-risk types that cause about 70% of cervical cancers and precancerous cervical lesions, according to the World Health Organization. However, there are three HPV vaccines – 9-valent HPV vaccine (Gardasil), quadrivalent HPV vaccine (Gardasil®, 4vHPV), and bivalent HPV vaccine (Cervarix) – that help protect against HPV 16/18.
The findings strengthen the benefits of HPV vaccination, Dr. Trottier explained. “There is no cure when the HPV infection is present,” Dr. Trottier said. “If the link [between preterm birth and HPV infections] is indeed causal, we can expect a greater risk of preterm delivery in these women. The effective tool we have is the HPV vaccination, but it should ideally be received before the start of sexual activity in order to prevent future infections that could occur in women.”
The American College of Obstetricians and Gynecologists recommends HPV vaccination for girls and women between the ages of 11 and 26 years old. However, Dr. Tanner said, women aged 27-45 who were previously unvaccinated may still receive benefit from the vaccine.
“Despite the known efficacy of the vaccine, only 50% of patients are up to date with their HPV vaccination,” Dr. Tanner explained. “This study further highlights the need to educate and encourage patients to be vaccinated.”
The researchers said future studies should investigate the association of HPV vaccination and vaccination programs with the risk of preterm birth.
The experts disclosed no conflicts of interest.
FROM JAMA NETWORK OPEN
Breast density associated with increased invasive breast cancer risk after age 65
The findings, based on an analysis of Breast Cancer Surveillance Consortium data from Jan. 1, 1996, to Dec. 31, 2012, have potential implications for screening mammography decisions in older women – particularly those aged 75 years and older, for whom screening guidance is limited by a paucity of data, Dejana Braithwaite, PhD, of the University of Florida Health Cancer Center, Gainesville, and colleagues reported in JAMA Network Open.
The investigators analyzed 221,714 screening mammograms from 193,787 women aged 65 and older in the United States. About 65% of the mammograms were from women aged 65-74 years and about 35% were from women aged 75 years and older, who comprised 38% of the study population.
During a mean follow-up of 6.3 years, 5,069 invasive breast cancers were diagnosed, the authors noted.
The 5-year cumulative incidence of invasive breast cancer increased in tandem with increasing breast density among those aged 65-74 years and among those aged 75 and older: The cumulative incidence per 1,000 women aged 65-74 years was 11.3 for those with almost entirely fatty breasts, 17.2 for those with scattered fibroglandular densities, and 23.7 for those with extremely or heterogeneously dense breasts. The cumulative incidence rates for those aged 75 years and older were 13.5, 18.4, and 22.5 per 1,000 women, respectively, they found.
Extreme or heterogeneous breast density was associated with increased risk of breast cancer, compared with scattered fibroglandular breast density, in both age categories (hazard ratios, 1.39 and 1.23 for those aged 65-74 years and 75 years and older, respectively), whereas the risk of invasive breast cancer was about 30% lower among women with almost entirely fatty breasts, compared with women with scattered fibroglandular breast density (HRs, 0.66 and 0.73 for the 65-74 and 75-plus age groups, respectively).
The associations between breast density and breast cancer were statistically significant after adjustment for body mass index (BMI) and other risk factors.
However, no significant differences were seen between breast density and breast cancer risk based on BMI, noted the authors, who investigated this potential association as part of their effort to identify subpopulations of older women who might benefit from screening, “especially because the U.S. Preventive Service Task Force guidelines state that the current evidence is considered insufficient to recommend routine breast cancer screening for women aged 75 years or older,” they wrote.
Further, although breast density is important in risk assessment and could be evaluated in older women, some risk prediction models exclude women aged 75 or older in risk assessments, they noted, adding that this is concerning given “the aging of the population in the U.S. and worldwide.”
“The positive associations found in this study between breast density and breast cancer among women aged 75 years or older suggest that breast density and life expectancy should be considered together when discussing the potential benefits and harms of continued screening mammography in this population,” they concluded.
The new findings supplement those from prior studies and highlight “the intersection of ... two subjects that have garnered considerable lay public, healthy policy, and academic interest” in recent years: screening mammography in older women and the risk of breast cancer caused by breast density in older women, Catherine M. Tuite, MD, of ChristianaCare Helen F. Graham Cancer Center and Research Institute, Newark, Del., wrote in a commentary published with the study.
“Although there is a linear association between age and mammographic density, age is not a perfect surrogate for the latter, and there are meaningful numbers of older women with mammographically dense breast tissue,” she said, noting that a 75-year-old woman in the United States has a life expectancy of 12-14 additional years, and that “continuation of screening mammography in healthy women aged 75 years or older may offer a substantial opportunity to avoid morbidity and mortality from breast cancer in this age group.”
However, overdiagnosis also remains a concern, she said.
“Breast density and age are only a few of the many factors currently under investigation in the drive toward risk-based or personalized breast cancer screening,” she wrote. “We must remain cautious in the application of restrictive screening for women of any age with supposedly lower than average risk ... ultimately, the decision of when to stop screening is personal, and each woman deserves the agency to weigh her own wishes, values, and life experiences with an accurate and unbiased discussion of risks and benefits of screening mammography in making that decision.”
This study was supported by grants from the National Cancer Institute and the Breast Cancer Surveillance Consortium. Cancer and vital status data collection was supported in part by several state public health departments and cancer registries. Dr. Advani and Dr. Tuite each reported having no disclosures.
The findings, based on an analysis of Breast Cancer Surveillance Consortium data from Jan. 1, 1996, to Dec. 31, 2012, have potential implications for screening mammography decisions in older women – particularly those aged 75 years and older, for whom screening guidance is limited by a paucity of data, Dejana Braithwaite, PhD, of the University of Florida Health Cancer Center, Gainesville, and colleagues reported in JAMA Network Open.
The investigators analyzed 221,714 screening mammograms from 193,787 women aged 65 and older in the United States. About 65% of the mammograms were from women aged 65-74 years and about 35% were from women aged 75 years and older, who comprised 38% of the study population.
During a mean follow-up of 6.3 years, 5,069 invasive breast cancers were diagnosed, the authors noted.
The 5-year cumulative incidence of invasive breast cancer increased in tandem with increasing breast density among those aged 65-74 years and among those aged 75 and older: The cumulative incidence per 1,000 women aged 65-74 years was 11.3 for those with almost entirely fatty breasts, 17.2 for those with scattered fibroglandular densities, and 23.7 for those with extremely or heterogeneously dense breasts. The cumulative incidence rates for those aged 75 years and older were 13.5, 18.4, and 22.5 per 1,000 women, respectively, they found.
Extreme or heterogeneous breast density was associated with increased risk of breast cancer, compared with scattered fibroglandular breast density, in both age categories (hazard ratios, 1.39 and 1.23 for those aged 65-74 years and 75 years and older, respectively), whereas the risk of invasive breast cancer was about 30% lower among women with almost entirely fatty breasts, compared with women with scattered fibroglandular breast density (HRs, 0.66 and 0.73 for the 65-74 and 75-plus age groups, respectively).
The associations between breast density and breast cancer were statistically significant after adjustment for body mass index (BMI) and other risk factors.
However, no significant differences were seen between breast density and breast cancer risk based on BMI, noted the authors, who investigated this potential association as part of their effort to identify subpopulations of older women who might benefit from screening, “especially because the U.S. Preventive Service Task Force guidelines state that the current evidence is considered insufficient to recommend routine breast cancer screening for women aged 75 years or older,” they wrote.
Further, although breast density is important in risk assessment and could be evaluated in older women, some risk prediction models exclude women aged 75 or older in risk assessments, they noted, adding that this is concerning given “the aging of the population in the U.S. and worldwide.”
“The positive associations found in this study between breast density and breast cancer among women aged 75 years or older suggest that breast density and life expectancy should be considered together when discussing the potential benefits and harms of continued screening mammography in this population,” they concluded.
The new findings supplement those from prior studies and highlight “the intersection of ... two subjects that have garnered considerable lay public, healthy policy, and academic interest” in recent years: screening mammography in older women and the risk of breast cancer caused by breast density in older women, Catherine M. Tuite, MD, of ChristianaCare Helen F. Graham Cancer Center and Research Institute, Newark, Del., wrote in a commentary published with the study.
“Although there is a linear association between age and mammographic density, age is not a perfect surrogate for the latter, and there are meaningful numbers of older women with mammographically dense breast tissue,” she said, noting that a 75-year-old woman in the United States has a life expectancy of 12-14 additional years, and that “continuation of screening mammography in healthy women aged 75 years or older may offer a substantial opportunity to avoid morbidity and mortality from breast cancer in this age group.”
However, overdiagnosis also remains a concern, she said.
“Breast density and age are only a few of the many factors currently under investigation in the drive toward risk-based or personalized breast cancer screening,” she wrote. “We must remain cautious in the application of restrictive screening for women of any age with supposedly lower than average risk ... ultimately, the decision of when to stop screening is personal, and each woman deserves the agency to weigh her own wishes, values, and life experiences with an accurate and unbiased discussion of risks and benefits of screening mammography in making that decision.”
This study was supported by grants from the National Cancer Institute and the Breast Cancer Surveillance Consortium. Cancer and vital status data collection was supported in part by several state public health departments and cancer registries. Dr. Advani and Dr. Tuite each reported having no disclosures.
The findings, based on an analysis of Breast Cancer Surveillance Consortium data from Jan. 1, 1996, to Dec. 31, 2012, have potential implications for screening mammography decisions in older women – particularly those aged 75 years and older, for whom screening guidance is limited by a paucity of data, Dejana Braithwaite, PhD, of the University of Florida Health Cancer Center, Gainesville, and colleagues reported in JAMA Network Open.
The investigators analyzed 221,714 screening mammograms from 193,787 women aged 65 and older in the United States. About 65% of the mammograms were from women aged 65-74 years and about 35% were from women aged 75 years and older, who comprised 38% of the study population.
During a mean follow-up of 6.3 years, 5,069 invasive breast cancers were diagnosed, the authors noted.
The 5-year cumulative incidence of invasive breast cancer increased in tandem with increasing breast density among those aged 65-74 years and among those aged 75 and older: The cumulative incidence per 1,000 women aged 65-74 years was 11.3 for those with almost entirely fatty breasts, 17.2 for those with scattered fibroglandular densities, and 23.7 for those with extremely or heterogeneously dense breasts. The cumulative incidence rates for those aged 75 years and older were 13.5, 18.4, and 22.5 per 1,000 women, respectively, they found.
Extreme or heterogeneous breast density was associated with increased risk of breast cancer, compared with scattered fibroglandular breast density, in both age categories (hazard ratios, 1.39 and 1.23 for those aged 65-74 years and 75 years and older, respectively), whereas the risk of invasive breast cancer was about 30% lower among women with almost entirely fatty breasts, compared with women with scattered fibroglandular breast density (HRs, 0.66 and 0.73 for the 65-74 and 75-plus age groups, respectively).
The associations between breast density and breast cancer were statistically significant after adjustment for body mass index (BMI) and other risk factors.
However, no significant differences were seen between breast density and breast cancer risk based on BMI, noted the authors, who investigated this potential association as part of their effort to identify subpopulations of older women who might benefit from screening, “especially because the U.S. Preventive Service Task Force guidelines state that the current evidence is considered insufficient to recommend routine breast cancer screening for women aged 75 years or older,” they wrote.
Further, although breast density is important in risk assessment and could be evaluated in older women, some risk prediction models exclude women aged 75 or older in risk assessments, they noted, adding that this is concerning given “the aging of the population in the U.S. and worldwide.”
“The positive associations found in this study between breast density and breast cancer among women aged 75 years or older suggest that breast density and life expectancy should be considered together when discussing the potential benefits and harms of continued screening mammography in this population,” they concluded.
The new findings supplement those from prior studies and highlight “the intersection of ... two subjects that have garnered considerable lay public, healthy policy, and academic interest” in recent years: screening mammography in older women and the risk of breast cancer caused by breast density in older women, Catherine M. Tuite, MD, of ChristianaCare Helen F. Graham Cancer Center and Research Institute, Newark, Del., wrote in a commentary published with the study.
“Although there is a linear association between age and mammographic density, age is not a perfect surrogate for the latter, and there are meaningful numbers of older women with mammographically dense breast tissue,” she said, noting that a 75-year-old woman in the United States has a life expectancy of 12-14 additional years, and that “continuation of screening mammography in healthy women aged 75 years or older may offer a substantial opportunity to avoid morbidity and mortality from breast cancer in this age group.”
However, overdiagnosis also remains a concern, she said.
“Breast density and age are only a few of the many factors currently under investigation in the drive toward risk-based or personalized breast cancer screening,” she wrote. “We must remain cautious in the application of restrictive screening for women of any age with supposedly lower than average risk ... ultimately, the decision of when to stop screening is personal, and each woman deserves the agency to weigh her own wishes, values, and life experiences with an accurate and unbiased discussion of risks and benefits of screening mammography in making that decision.”
This study was supported by grants from the National Cancer Institute and the Breast Cancer Surveillance Consortium. Cancer and vital status data collection was supported in part by several state public health departments and cancer registries. Dr. Advani and Dr. Tuite each reported having no disclosures.
FROM JAMA NETWORK OPEN
Premature menopause a ‘warning sign’ for greater ASCVD risk
Premature menopause is well known to be linked to cardiovascular disease in women, but it may not carry as much weight as more traditional cardiovascular risk factors in determining a patient’s 10-year risk of having a heart attack or stroke in this population, a cohort study that evaluated the veracity of premature menopause found.
Premature menopause can serve as a “marker or warning sign” that cardiologists should pay closer attention to traditional atherosclerotic cardiovascular disease (ASCVD) risk factors, lead study author Sadiya S. Khan, MD, MS, said in an interview. “When we looked at the addition of premature menopause into the risk-prediction equation, we did not see that it meaningfully improved the ability of the risk predictions of pooled cohort equations [PCEs] to identify who developed cardiovascular disease,” said Dr. Khan, a cardiologist at Northwestern University, Chicago.
The cohort study included 5,466 Black women and 10,584 White women from seven U.S. population-based cohorts, including the Women’s Health Initiative, of whom 951 and 1,039, respectively, self-reported early menopause. The cohort study researchers noted that the 2019 American College of Cardiology/American Heart Association guideline for prevention of CVD acknowledged premature menopause as risk-enhancing factor in the CVD assessment in women younger than 40.
The cohort study found that Black women had almost twice the rate of premature menopause than White women, 17.4% and 9.8%, respectively. And it found that premature menopause was significantly linked with ASCVD in both populations independent of traditional risk factors – a 24% greater risk for Black women and 28% greater risk for White women.
‘Surprising’ finding
However, when premature menopause was added to the pooled cohort equations per the 2013 ACC/AHA guideline, the researchers found no incremental benefit, a finding Dr. Khan called “really surprising to us.”
She added, “If we look at the differences in the characteristics of women who have premature menopause, compared with those who didn’t, there were slight differences in terms of higher blood pressure, higher body mass index, and slightly higher glucose. So maybe what we’re seeing – and this is more speculative – is that risk factors are developing after early menopause, and the focus should be earlier in the patient’s life course to try to prevent hypertension, diabetes, and obesity.”
Dr. Khan emphasized that the findings don’t obviate the value of premature menopause in assessing ASCVD risk in women. “We still know that this is an important marker for women and their risk for heart disease, and it should be a warning sign to pay close attention to those other risk factors and what other preventive measures can be taken,” she said.
Christie Ballantyne, MD, said it’s important to note that the study did not dismiss the relevance of premature menopause in shared decision-making for postmenopausal women. “It certainly doesn’t mean that premature menopause is not a risk,” Dr. Ballantyne said in an interview. “Premature menopause may cause a worsening of traditional CVD risk factors, so that’s one possible explanation for it. The other possible explanation is that women with worse ASCVD risk factors – who are more overweight, have higher blood pressure, and have more diabetes and insulin resistance – are more likely to have earlier menopause.” Dr. Ballantyne is chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston.
“You should still look very carefully at the patient’s risk factors, calculate the pooled cohort equations, and make sure you get a recommendation,” he said. “If their risks are up, give recommendations on how to improve diet and exercise. Consider if you need to treat lipids or treat blood pressure with more than diet and exercise because there’s nothing magical about 7.5%”, the threshold for lipid-lowering therapy in the ASCVD risk calculator.
Dr. Khan and coauthors disclosed receiving grants from the National Institutes of Health and the American Heart Association. One coauthor reported a financial relationship with HGM Biopharmaceuticals. Dr. Ballantyne is a lead investigator of the Atherosclerosis Risk in Communities study, one of the population-based cohorts used in the cohort study. He has no other relevant relationships to disclose.
Premature menopause is well known to be linked to cardiovascular disease in women, but it may not carry as much weight as more traditional cardiovascular risk factors in determining a patient’s 10-year risk of having a heart attack or stroke in this population, a cohort study that evaluated the veracity of premature menopause found.
Premature menopause can serve as a “marker or warning sign” that cardiologists should pay closer attention to traditional atherosclerotic cardiovascular disease (ASCVD) risk factors, lead study author Sadiya S. Khan, MD, MS, said in an interview. “When we looked at the addition of premature menopause into the risk-prediction equation, we did not see that it meaningfully improved the ability of the risk predictions of pooled cohort equations [PCEs] to identify who developed cardiovascular disease,” said Dr. Khan, a cardiologist at Northwestern University, Chicago.
The cohort study included 5,466 Black women and 10,584 White women from seven U.S. population-based cohorts, including the Women’s Health Initiative, of whom 951 and 1,039, respectively, self-reported early menopause. The cohort study researchers noted that the 2019 American College of Cardiology/American Heart Association guideline for prevention of CVD acknowledged premature menopause as risk-enhancing factor in the CVD assessment in women younger than 40.
The cohort study found that Black women had almost twice the rate of premature menopause than White women, 17.4% and 9.8%, respectively. And it found that premature menopause was significantly linked with ASCVD in both populations independent of traditional risk factors – a 24% greater risk for Black women and 28% greater risk for White women.
‘Surprising’ finding
However, when premature menopause was added to the pooled cohort equations per the 2013 ACC/AHA guideline, the researchers found no incremental benefit, a finding Dr. Khan called “really surprising to us.”
She added, “If we look at the differences in the characteristics of women who have premature menopause, compared with those who didn’t, there were slight differences in terms of higher blood pressure, higher body mass index, and slightly higher glucose. So maybe what we’re seeing – and this is more speculative – is that risk factors are developing after early menopause, and the focus should be earlier in the patient’s life course to try to prevent hypertension, diabetes, and obesity.”
Dr. Khan emphasized that the findings don’t obviate the value of premature menopause in assessing ASCVD risk in women. “We still know that this is an important marker for women and their risk for heart disease, and it should be a warning sign to pay close attention to those other risk factors and what other preventive measures can be taken,” she said.
Christie Ballantyne, MD, said it’s important to note that the study did not dismiss the relevance of premature menopause in shared decision-making for postmenopausal women. “It certainly doesn’t mean that premature menopause is not a risk,” Dr. Ballantyne said in an interview. “Premature menopause may cause a worsening of traditional CVD risk factors, so that’s one possible explanation for it. The other possible explanation is that women with worse ASCVD risk factors – who are more overweight, have higher blood pressure, and have more diabetes and insulin resistance – are more likely to have earlier menopause.” Dr. Ballantyne is chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston.
“You should still look very carefully at the patient’s risk factors, calculate the pooled cohort equations, and make sure you get a recommendation,” he said. “If their risks are up, give recommendations on how to improve diet and exercise. Consider if you need to treat lipids or treat blood pressure with more than diet and exercise because there’s nothing magical about 7.5%”, the threshold for lipid-lowering therapy in the ASCVD risk calculator.
Dr. Khan and coauthors disclosed receiving grants from the National Institutes of Health and the American Heart Association. One coauthor reported a financial relationship with HGM Biopharmaceuticals. Dr. Ballantyne is a lead investigator of the Atherosclerosis Risk in Communities study, one of the population-based cohorts used in the cohort study. He has no other relevant relationships to disclose.
Premature menopause is well known to be linked to cardiovascular disease in women, but it may not carry as much weight as more traditional cardiovascular risk factors in determining a patient’s 10-year risk of having a heart attack or stroke in this population, a cohort study that evaluated the veracity of premature menopause found.
Premature menopause can serve as a “marker or warning sign” that cardiologists should pay closer attention to traditional atherosclerotic cardiovascular disease (ASCVD) risk factors, lead study author Sadiya S. Khan, MD, MS, said in an interview. “When we looked at the addition of premature menopause into the risk-prediction equation, we did not see that it meaningfully improved the ability of the risk predictions of pooled cohort equations [PCEs] to identify who developed cardiovascular disease,” said Dr. Khan, a cardiologist at Northwestern University, Chicago.
The cohort study included 5,466 Black women and 10,584 White women from seven U.S. population-based cohorts, including the Women’s Health Initiative, of whom 951 and 1,039, respectively, self-reported early menopause. The cohort study researchers noted that the 2019 American College of Cardiology/American Heart Association guideline for prevention of CVD acknowledged premature menopause as risk-enhancing factor in the CVD assessment in women younger than 40.
The cohort study found that Black women had almost twice the rate of premature menopause than White women, 17.4% and 9.8%, respectively. And it found that premature menopause was significantly linked with ASCVD in both populations independent of traditional risk factors – a 24% greater risk for Black women and 28% greater risk for White women.
‘Surprising’ finding
However, when premature menopause was added to the pooled cohort equations per the 2013 ACC/AHA guideline, the researchers found no incremental benefit, a finding Dr. Khan called “really surprising to us.”
She added, “If we look at the differences in the characteristics of women who have premature menopause, compared with those who didn’t, there were slight differences in terms of higher blood pressure, higher body mass index, and slightly higher glucose. So maybe what we’re seeing – and this is more speculative – is that risk factors are developing after early menopause, and the focus should be earlier in the patient’s life course to try to prevent hypertension, diabetes, and obesity.”
Dr. Khan emphasized that the findings don’t obviate the value of premature menopause in assessing ASCVD risk in women. “We still know that this is an important marker for women and their risk for heart disease, and it should be a warning sign to pay close attention to those other risk factors and what other preventive measures can be taken,” she said.
Christie Ballantyne, MD, said it’s important to note that the study did not dismiss the relevance of premature menopause in shared decision-making for postmenopausal women. “It certainly doesn’t mean that premature menopause is not a risk,” Dr. Ballantyne said in an interview. “Premature menopause may cause a worsening of traditional CVD risk factors, so that’s one possible explanation for it. The other possible explanation is that women with worse ASCVD risk factors – who are more overweight, have higher blood pressure, and have more diabetes and insulin resistance – are more likely to have earlier menopause.” Dr. Ballantyne is chief of cardiology at Baylor College of Medicine and director of cardiovascular disease prevention at Methodist DeBakey Heart Center, both in Houston.
“You should still look very carefully at the patient’s risk factors, calculate the pooled cohort equations, and make sure you get a recommendation,” he said. “If their risks are up, give recommendations on how to improve diet and exercise. Consider if you need to treat lipids or treat blood pressure with more than diet and exercise because there’s nothing magical about 7.5%”, the threshold for lipid-lowering therapy in the ASCVD risk calculator.
Dr. Khan and coauthors disclosed receiving grants from the National Institutes of Health and the American Heart Association. One coauthor reported a financial relationship with HGM Biopharmaceuticals. Dr. Ballantyne is a lead investigator of the Atherosclerosis Risk in Communities study, one of the population-based cohorts used in the cohort study. He has no other relevant relationships to disclose.
FROM JAMA CARDIOLOGY
Sacral nerve root endometriosis

Additional videos from SGS are available here, including these recent offerings:

Additional videos from SGS are available here, including these recent offerings:

Additional videos from SGS are available here, including these recent offerings:
Menopause society issues first osteoporosis advice in 10 years
In the first revision to its guidance on the management of osteoporosis in a decade, the North American Menopause Society has issued an updated position statement addressing evolving evidence on osteoporosis issues ranging from screening and risk assessment to appropriate use of preventive therapy in postmenopausal women.
“Since the 2010 statement, there have been important new developments in our field, including better delineation of risk factors for fracture, resulting in better strategies for assessing fracture risk,” Michael R. McClung, MD, who is a NAMS board member and colead of the editorial panel for the 2021 position statement, told this news organization. Dr. McClung is also director emeritus of the Oregon Osteoporosis Center in Portland.
“There is much more information about the long-term safety of therapies,” he added. Dr. McClung also noted “the availability of four new drugs for the prevention and treatment of osteoporosis and clinical experience informing us of the effects of using different treatments in various sequences.”
Osteoporosis is substantially underdiagnosed and undertreated
A basis for the update, recently published in Menopause: The Journal of the North American Menopause Society, is the need to tackle the troubling fact that approximately half of postmenopausal women will experience a fracture related to osteoporosis in their lifetime, yet the condition is “substantially underdiagnosed and undertreated,” NAMS underscores.
With that in mind, osteoporosis should be considered by practitioners treating menopausal and postmenopausal women at all levels of care.
“All physicians and advanced care providers caring for postmenopausal women should be comfortable assessing and managing their patients with, or at risk for, fractures,” Dr. McClung added.
Osteoporosis prevention in young menopausal women
The NAMS statement covers a broad range of issues, and while most recommendations generally follow those of other societies’ guidelines, a unique aspect is the emphasis on preventing osteoporosis in young menopausal women with estrogen or other drugs.
While underscoring that all menopausal women should be encouraged to adopt healthy lifestyles, with good diets and physical activity to reduce the risk of bone loss and fractures, pharmacologic interventions also have a role, NAMS says.
Though long an issue of debate, NAMS voices support for estrogen therapy as having an important role in osteoporosis prevention, as estrogen deficiency is the principal cause of bone loss in postmenopausal women.
“Hormone therapy is the most appropriate choice to prevent bone loss at the time of menopause for healthy women, particularly those who have menopause symptoms,” the group states. Drug interventions are specifically supported in women with premature menopause, at least until the average age of natural menopause, in addition to those with low bone mineral density (BMD) (T-score < –1.0) and those experiencing relatively rapid bone loss related to acute estrogen deficiency in the menopause transition or on discontinuing estrogen therapy.
“Although using drugs to prevent osteoporosis is not included in national osteoporosis guidelines, a strong clinical argument can be made for doing so, especially in women who come to menopause with low bone mass,” the report states.
And therapy is also recommended if patients have a low BMD and other risk factors for fracture, such as family history, but do not meet the criteria for osteoporosis treatment.
Ultimately, clinicians should work with patients when deciding the options, Dr. McClung said.
“After carefully weighing the small risks associated with hormone therapy or other therapies begun at the time of menopause, menopause practitioners and their patients can and should make informed decisions about the use of Food and Drug Administration–approved medications to prevent osteoporosis in women who are at risk for developing that condition,” he noted, adding that his view on the matter is his own and not necessarily that of NAMS.
New treatments endorsed for high-risk patients to avoid ‘bone attack’
While most patients are treated for osteoporosis with antiremodeling drugs such as bisphosphonates and denosumab, NAMS endorses “a new paradigm of beginning treatment with a bone-building agent followed by an antiremodeling agent” for women at very high risk of fracture.
“Consider osteoanabolic therapies for patients at very high risk of fracture, including older women with recent fractures, T-scores –3.0 and lower, or multiple other risk factors,” the statement suggests.
Among those at highest risk are women who have sustained a first fracture.
“A recent fracture in a postmenopausal woman is the strongest risk factor for another fracture,” Dr. McClung said.
In fact, “having a fracture should be thought of and assessed as a ‘bone attack,’ ” he asserted.
Therapy is recommended in such cases to rapidly increase bone density and reduce their subsequent fracture risk.
“For these patients, osteoanabolic or bone-building agents are more effective than bisphosphonates and are recommended as initial therapy,” Dr. McClung noted.
Treatment discontinuation?
On the issue of drug holidays and when or whether to stop therapy, as no therapies cure osteoporosis, medications should not be permanently stopped, even if bone density increases, NAMS recommends.
“By analogy, we do not stop diabetes therapy when A1c levels become normal,” Dr. McClung noted.
“Because the benefits of therapy on bone density and fracture protection wane, quickly for nonbisphosphonates and more slowly with bisphosphonates, short-term therapy, for instance 5 years, is not optimal treatment,” he said.
While the short-term interruption of bisphosphonate therapy may be considered in some patients, “the concept of ‘drug holidays’ does not pertain to nonbisphosphonate drugs,” Dr. McClung said.
NAMS adds that management of therapeutic choices should instead be ongoing.
“During therapy, reevaluate the treatment goals and the choice of medication on an ongoing basis through periodic medical examination and follow-up BMD testing,” NAMS recommends.
In terms of assessment, the measurement of bone mineral density while on treatment can gauge the current risk of fracture, and NAMS supports the use of the T-score at the hip as an appropriate clinical target in guiding choices of therapy.
Ultimately, “effective tools for diagnosing osteoporosis and assessing fracture risk are available, and well-studied strategies exist for managing bone health in women at both low and high risk of fracture,” NAMS concludes.
“By individualizing treatment approaches and monitoring and adjusting those approaches if the clinical picture changes, the consequences of osteoporosis on a menopausal woman’s activity and well-being can be minimized.”
Dr. McClung has reported receiving consulting fees from Amgen and Myovant, and honorarium for speaking from Amgen and Alexon. He serves on the boards of NAMS and the International Osteoporosis Foundation.
A version of this article first appeared on Medscape.com.
In the first revision to its guidance on the management of osteoporosis in a decade, the North American Menopause Society has issued an updated position statement addressing evolving evidence on osteoporosis issues ranging from screening and risk assessment to appropriate use of preventive therapy in postmenopausal women.
“Since the 2010 statement, there have been important new developments in our field, including better delineation of risk factors for fracture, resulting in better strategies for assessing fracture risk,” Michael R. McClung, MD, who is a NAMS board member and colead of the editorial panel for the 2021 position statement, told this news organization. Dr. McClung is also director emeritus of the Oregon Osteoporosis Center in Portland.
“There is much more information about the long-term safety of therapies,” he added. Dr. McClung also noted “the availability of four new drugs for the prevention and treatment of osteoporosis and clinical experience informing us of the effects of using different treatments in various sequences.”
Osteoporosis is substantially underdiagnosed and undertreated
A basis for the update, recently published in Menopause: The Journal of the North American Menopause Society, is the need to tackle the troubling fact that approximately half of postmenopausal women will experience a fracture related to osteoporosis in their lifetime, yet the condition is “substantially underdiagnosed and undertreated,” NAMS underscores.
With that in mind, osteoporosis should be considered by practitioners treating menopausal and postmenopausal women at all levels of care.
“All physicians and advanced care providers caring for postmenopausal women should be comfortable assessing and managing their patients with, or at risk for, fractures,” Dr. McClung added.
Osteoporosis prevention in young menopausal women
The NAMS statement covers a broad range of issues, and while most recommendations generally follow those of other societies’ guidelines, a unique aspect is the emphasis on preventing osteoporosis in young menopausal women with estrogen or other drugs.
While underscoring that all menopausal women should be encouraged to adopt healthy lifestyles, with good diets and physical activity to reduce the risk of bone loss and fractures, pharmacologic interventions also have a role, NAMS says.
Though long an issue of debate, NAMS voices support for estrogen therapy as having an important role in osteoporosis prevention, as estrogen deficiency is the principal cause of bone loss in postmenopausal women.
“Hormone therapy is the most appropriate choice to prevent bone loss at the time of menopause for healthy women, particularly those who have menopause symptoms,” the group states. Drug interventions are specifically supported in women with premature menopause, at least until the average age of natural menopause, in addition to those with low bone mineral density (BMD) (T-score < –1.0) and those experiencing relatively rapid bone loss related to acute estrogen deficiency in the menopause transition or on discontinuing estrogen therapy.
“Although using drugs to prevent osteoporosis is not included in national osteoporosis guidelines, a strong clinical argument can be made for doing so, especially in women who come to menopause with low bone mass,” the report states.
And therapy is also recommended if patients have a low BMD and other risk factors for fracture, such as family history, but do not meet the criteria for osteoporosis treatment.
Ultimately, clinicians should work with patients when deciding the options, Dr. McClung said.
“After carefully weighing the small risks associated with hormone therapy or other therapies begun at the time of menopause, menopause practitioners and their patients can and should make informed decisions about the use of Food and Drug Administration–approved medications to prevent osteoporosis in women who are at risk for developing that condition,” he noted, adding that his view on the matter is his own and not necessarily that of NAMS.
New treatments endorsed for high-risk patients to avoid ‘bone attack’
While most patients are treated for osteoporosis with antiremodeling drugs such as bisphosphonates and denosumab, NAMS endorses “a new paradigm of beginning treatment with a bone-building agent followed by an antiremodeling agent” for women at very high risk of fracture.
“Consider osteoanabolic therapies for patients at very high risk of fracture, including older women with recent fractures, T-scores –3.0 and lower, or multiple other risk factors,” the statement suggests.
Among those at highest risk are women who have sustained a first fracture.
“A recent fracture in a postmenopausal woman is the strongest risk factor for another fracture,” Dr. McClung said.
In fact, “having a fracture should be thought of and assessed as a ‘bone attack,’ ” he asserted.
Therapy is recommended in such cases to rapidly increase bone density and reduce their subsequent fracture risk.
“For these patients, osteoanabolic or bone-building agents are more effective than bisphosphonates and are recommended as initial therapy,” Dr. McClung noted.
Treatment discontinuation?
On the issue of drug holidays and when or whether to stop therapy, as no therapies cure osteoporosis, medications should not be permanently stopped, even if bone density increases, NAMS recommends.
“By analogy, we do not stop diabetes therapy when A1c levels become normal,” Dr. McClung noted.
“Because the benefits of therapy on bone density and fracture protection wane, quickly for nonbisphosphonates and more slowly with bisphosphonates, short-term therapy, for instance 5 years, is not optimal treatment,” he said.
While the short-term interruption of bisphosphonate therapy may be considered in some patients, “the concept of ‘drug holidays’ does not pertain to nonbisphosphonate drugs,” Dr. McClung said.
NAMS adds that management of therapeutic choices should instead be ongoing.
“During therapy, reevaluate the treatment goals and the choice of medication on an ongoing basis through periodic medical examination and follow-up BMD testing,” NAMS recommends.
In terms of assessment, the measurement of bone mineral density while on treatment can gauge the current risk of fracture, and NAMS supports the use of the T-score at the hip as an appropriate clinical target in guiding choices of therapy.
Ultimately, “effective tools for diagnosing osteoporosis and assessing fracture risk are available, and well-studied strategies exist for managing bone health in women at both low and high risk of fracture,” NAMS concludes.
“By individualizing treatment approaches and monitoring and adjusting those approaches if the clinical picture changes, the consequences of osteoporosis on a menopausal woman’s activity and well-being can be minimized.”
Dr. McClung has reported receiving consulting fees from Amgen and Myovant, and honorarium for speaking from Amgen and Alexon. He serves on the boards of NAMS and the International Osteoporosis Foundation.
A version of this article first appeared on Medscape.com.
In the first revision to its guidance on the management of osteoporosis in a decade, the North American Menopause Society has issued an updated position statement addressing evolving evidence on osteoporosis issues ranging from screening and risk assessment to appropriate use of preventive therapy in postmenopausal women.
“Since the 2010 statement, there have been important new developments in our field, including better delineation of risk factors for fracture, resulting in better strategies for assessing fracture risk,” Michael R. McClung, MD, who is a NAMS board member and colead of the editorial panel for the 2021 position statement, told this news organization. Dr. McClung is also director emeritus of the Oregon Osteoporosis Center in Portland.
“There is much more information about the long-term safety of therapies,” he added. Dr. McClung also noted “the availability of four new drugs for the prevention and treatment of osteoporosis and clinical experience informing us of the effects of using different treatments in various sequences.”
Osteoporosis is substantially underdiagnosed and undertreated
A basis for the update, recently published in Menopause: The Journal of the North American Menopause Society, is the need to tackle the troubling fact that approximately half of postmenopausal women will experience a fracture related to osteoporosis in their lifetime, yet the condition is “substantially underdiagnosed and undertreated,” NAMS underscores.
With that in mind, osteoporosis should be considered by practitioners treating menopausal and postmenopausal women at all levels of care.
“All physicians and advanced care providers caring for postmenopausal women should be comfortable assessing and managing their patients with, or at risk for, fractures,” Dr. McClung added.
Osteoporosis prevention in young menopausal women
The NAMS statement covers a broad range of issues, and while most recommendations generally follow those of other societies’ guidelines, a unique aspect is the emphasis on preventing osteoporosis in young menopausal women with estrogen or other drugs.
While underscoring that all menopausal women should be encouraged to adopt healthy lifestyles, with good diets and physical activity to reduce the risk of bone loss and fractures, pharmacologic interventions also have a role, NAMS says.
Though long an issue of debate, NAMS voices support for estrogen therapy as having an important role in osteoporosis prevention, as estrogen deficiency is the principal cause of bone loss in postmenopausal women.
“Hormone therapy is the most appropriate choice to prevent bone loss at the time of menopause for healthy women, particularly those who have menopause symptoms,” the group states. Drug interventions are specifically supported in women with premature menopause, at least until the average age of natural menopause, in addition to those with low bone mineral density (BMD) (T-score < –1.0) and those experiencing relatively rapid bone loss related to acute estrogen deficiency in the menopause transition or on discontinuing estrogen therapy.
“Although using drugs to prevent osteoporosis is not included in national osteoporosis guidelines, a strong clinical argument can be made for doing so, especially in women who come to menopause with low bone mass,” the report states.
And therapy is also recommended if patients have a low BMD and other risk factors for fracture, such as family history, but do not meet the criteria for osteoporosis treatment.
Ultimately, clinicians should work with patients when deciding the options, Dr. McClung said.
“After carefully weighing the small risks associated with hormone therapy or other therapies begun at the time of menopause, menopause practitioners and their patients can and should make informed decisions about the use of Food and Drug Administration–approved medications to prevent osteoporosis in women who are at risk for developing that condition,” he noted, adding that his view on the matter is his own and not necessarily that of NAMS.
New treatments endorsed for high-risk patients to avoid ‘bone attack’
While most patients are treated for osteoporosis with antiremodeling drugs such as bisphosphonates and denosumab, NAMS endorses “a new paradigm of beginning treatment with a bone-building agent followed by an antiremodeling agent” for women at very high risk of fracture.
“Consider osteoanabolic therapies for patients at very high risk of fracture, including older women with recent fractures, T-scores –3.0 and lower, or multiple other risk factors,” the statement suggests.
Among those at highest risk are women who have sustained a first fracture.
“A recent fracture in a postmenopausal woman is the strongest risk factor for another fracture,” Dr. McClung said.
In fact, “having a fracture should be thought of and assessed as a ‘bone attack,’ ” he asserted.
Therapy is recommended in such cases to rapidly increase bone density and reduce their subsequent fracture risk.
“For these patients, osteoanabolic or bone-building agents are more effective than bisphosphonates and are recommended as initial therapy,” Dr. McClung noted.
Treatment discontinuation?
On the issue of drug holidays and when or whether to stop therapy, as no therapies cure osteoporosis, medications should not be permanently stopped, even if bone density increases, NAMS recommends.
“By analogy, we do not stop diabetes therapy when A1c levels become normal,” Dr. McClung noted.
“Because the benefits of therapy on bone density and fracture protection wane, quickly for nonbisphosphonates and more slowly with bisphosphonates, short-term therapy, for instance 5 years, is not optimal treatment,” he said.
While the short-term interruption of bisphosphonate therapy may be considered in some patients, “the concept of ‘drug holidays’ does not pertain to nonbisphosphonate drugs,” Dr. McClung said.
NAMS adds that management of therapeutic choices should instead be ongoing.
“During therapy, reevaluate the treatment goals and the choice of medication on an ongoing basis through periodic medical examination and follow-up BMD testing,” NAMS recommends.
In terms of assessment, the measurement of bone mineral density while on treatment can gauge the current risk of fracture, and NAMS supports the use of the T-score at the hip as an appropriate clinical target in guiding choices of therapy.
Ultimately, “effective tools for diagnosing osteoporosis and assessing fracture risk are available, and well-studied strategies exist for managing bone health in women at both low and high risk of fracture,” NAMS concludes.
“By individualizing treatment approaches and monitoring and adjusting those approaches if the clinical picture changes, the consequences of osteoporosis on a menopausal woman’s activity and well-being can be minimized.”
Dr. McClung has reported receiving consulting fees from Amgen and Myovant, and honorarium for speaking from Amgen and Alexon. He serves on the boards of NAMS and the International Osteoporosis Foundation.
A version of this article first appeared on Medscape.com.
New Moderna vaccine data ‘support’ booster shot after 8 months
Moderna has released new data that it said support the argument for COVID-19 booster shots – specifically showing that people who received a first shot of their mRNA vaccine a median of 13 months ago are more likely to experience a breakthrough infection compared to individuals who received a first shot a median of 8 months ago.
The findings come from the ongoing phase 3 COVE clinical trial, the results of which the Food and Drug Administration considered in granting emergency use authorization for the vaccine. In the initial stage of the trial, people were randomly assigned to receive the company’s mRNA vaccine or placebo.
according to the analysis of the open-label extension of the study during which placebo participants could cross over and get immunized as well.
The updated COVE trial data show that 88 breakthrough cases of COVID-19 occurred among 11,431 participants vaccinated between December 2020 and March 2021 (49.0 cases per 1,000 person-years).
In contrast, there were 162 breakthrough cases among 14,746 people vaccinated between July and October 2020 (77.1 cases per 1,000 person-years).
The breakthrough infections include 19 severe cases. Although not statically different, there was a trend toward fewer severe cases among the more recently vaccinated, at a rate of 3.3 per 1,000 person-years, compared with 6.2 per 1,000 person-years in the group vaccinated in 2020
The findings were posted as a preprint to the medRxiv server and have not yet been peer reviewed.
“The increased risk of breakthrough infections in COVE study participants who were vaccinated last year compared to more recently illustrates the impact of waning immunity and supports the need for a booster to maintain high levels of protection,” Moderna CEO Stéphane Bancel said in a company statement.
An FDA advisory committee is meeting Sept. 17 to look at the available evidence on boosters to help the agency decide whether the additional shots are warranted.
There is still a lot of debate in the medical community about the need for boosters. U.S. physicians and nurses are divided about the need for them and about how the country should prioritize its vaccine supplies, according to a Medscape poll of more than 1,700 clinicians that collected responses from Aug. 25 to Sept. 6, 2020.
The research was funded by Moderna, and also supported by the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, and by the National Institute of Allergy and Infectious Diseases.
A version of this article first appeared on Medscape.com.
Moderna has released new data that it said support the argument for COVID-19 booster shots – specifically showing that people who received a first shot of their mRNA vaccine a median of 13 months ago are more likely to experience a breakthrough infection compared to individuals who received a first shot a median of 8 months ago.
The findings come from the ongoing phase 3 COVE clinical trial, the results of which the Food and Drug Administration considered in granting emergency use authorization for the vaccine. In the initial stage of the trial, people were randomly assigned to receive the company’s mRNA vaccine or placebo.
according to the analysis of the open-label extension of the study during which placebo participants could cross over and get immunized as well.
The updated COVE trial data show that 88 breakthrough cases of COVID-19 occurred among 11,431 participants vaccinated between December 2020 and March 2021 (49.0 cases per 1,000 person-years).
In contrast, there were 162 breakthrough cases among 14,746 people vaccinated between July and October 2020 (77.1 cases per 1,000 person-years).
The breakthrough infections include 19 severe cases. Although not statically different, there was a trend toward fewer severe cases among the more recently vaccinated, at a rate of 3.3 per 1,000 person-years, compared with 6.2 per 1,000 person-years in the group vaccinated in 2020
The findings were posted as a preprint to the medRxiv server and have not yet been peer reviewed.
“The increased risk of breakthrough infections in COVE study participants who were vaccinated last year compared to more recently illustrates the impact of waning immunity and supports the need for a booster to maintain high levels of protection,” Moderna CEO Stéphane Bancel said in a company statement.
An FDA advisory committee is meeting Sept. 17 to look at the available evidence on boosters to help the agency decide whether the additional shots are warranted.
There is still a lot of debate in the medical community about the need for boosters. U.S. physicians and nurses are divided about the need for them and about how the country should prioritize its vaccine supplies, according to a Medscape poll of more than 1,700 clinicians that collected responses from Aug. 25 to Sept. 6, 2020.
The research was funded by Moderna, and also supported by the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, and by the National Institute of Allergy and Infectious Diseases.
A version of this article first appeared on Medscape.com.
Moderna has released new data that it said support the argument for COVID-19 booster shots – specifically showing that people who received a first shot of their mRNA vaccine a median of 13 months ago are more likely to experience a breakthrough infection compared to individuals who received a first shot a median of 8 months ago.
The findings come from the ongoing phase 3 COVE clinical trial, the results of which the Food and Drug Administration considered in granting emergency use authorization for the vaccine. In the initial stage of the trial, people were randomly assigned to receive the company’s mRNA vaccine or placebo.
according to the analysis of the open-label extension of the study during which placebo participants could cross over and get immunized as well.
The updated COVE trial data show that 88 breakthrough cases of COVID-19 occurred among 11,431 participants vaccinated between December 2020 and March 2021 (49.0 cases per 1,000 person-years).
In contrast, there were 162 breakthrough cases among 14,746 people vaccinated between July and October 2020 (77.1 cases per 1,000 person-years).
The breakthrough infections include 19 severe cases. Although not statically different, there was a trend toward fewer severe cases among the more recently vaccinated, at a rate of 3.3 per 1,000 person-years, compared with 6.2 per 1,000 person-years in the group vaccinated in 2020
The findings were posted as a preprint to the medRxiv server and have not yet been peer reviewed.
“The increased risk of breakthrough infections in COVE study participants who were vaccinated last year compared to more recently illustrates the impact of waning immunity and supports the need for a booster to maintain high levels of protection,” Moderna CEO Stéphane Bancel said in a company statement.
An FDA advisory committee is meeting Sept. 17 to look at the available evidence on boosters to help the agency decide whether the additional shots are warranted.
There is still a lot of debate in the medical community about the need for boosters. U.S. physicians and nurses are divided about the need for them and about how the country should prioritize its vaccine supplies, according to a Medscape poll of more than 1,700 clinicians that collected responses from Aug. 25 to Sept. 6, 2020.
The research was funded by Moderna, and also supported by the Office of the Assistant Secretary for Preparedness and Response, Biomedical Advanced Research and Development Authority, and by the National Institute of Allergy and Infectious Diseases.
A version of this article first appeared on Medscape.com.
The new transdermal contraceptive patch expands available contraceptive options: Does it offer protection with less VTE risk?
The first transdermal contraceptive patch was approved by the US Food and Drug Administration (FDA) in 2001.1 A 2018 survey revealed that 5% of women in the United States between the ages of 15 and 49 years reported the use of a short-acting hormonal contraceptive method (ie, vaginal ring, transdermal patch, injectable) within the past month, with just 0.3% reporting the use of a transdermal patch.2 Transdermal contraceptive patches are an effective form of birth control that may be a convenient option for patients who do not want to take a daily oral contraceptive pill but want similar efficacy and tolerability. Typical failure rates of patches are similar to that of combined oral contraceptives (COCs).1,3
While transdermal hormone delivery results in less peaks and troughs of estrogen compared with COCs, the total estrogen exposure is higher than with COCs; therefore, the risk for venous thromboembolism (VTE) with previously available patches is about twice as high.1 Twirla (Agile), an ethinyl estradiol (EE)/levonorgestrel (LNG) patch, delivers a low and consistent daily dose of hormones over 3 patches replaced once weekly, with no patch on the fourth week.3 Twirla contains 120 μg/day LNG and 30 μg/day EE. OrthoEvra, FDA approved in 2001 as mentioned, contains 150 μg/day norelgestromin and 35 μg/day EE.1 A reduction of the EE dose in COCs has been associated with lower risk for VTE.4
The addition of Twirla to the market offers another contraceptive option for patients who opt for a weekly, self-administered method.
How much lower is the VTE risk?
OBG
Barbara Levy, MD: The reality is we can’t designate a reduction of risk, except, in general, when the dose of ethinyl estradiol is lower, we think that the VTE risk is lower. There has not been a head-to-head comparison in a large enough population to be able to say that the risk is reduced by a certain factor. We just look at the overall exposure to estrogen and say, “In general, for VTE risk, a lower dose is a better thing for women.”
That being said, look at birth control pills, like COCs. We don’t have actual numbers to say that a 30-μg pill is this much less risky than a 35-μg pill. We just put it into a hierarchy, and that’s what we can do with the patch. We can say that, in general, lower is better for VTE risk, but no one can provide absolute numbers.
Continue to: Efficacy...
Efficacy
OBG
Dr. Levy: You have to look at the pivotal trials and look at what the efficacy was in a trial setting. In the real-world setting, the effectiveness is never quite as good as it is in a clinical trial. I think the bottom line for all of us is that combined oral contraception, meaning estrogen with progestin, is equivalently effective across the different options that are available for women. Efficacy really isn’t the factor to use to distinguish which one I’m going to pick. It is about the patient’s convenience and many other factors. But in terms of its clinical effectiveness in preventing pregnancy, from a very practical standpoint, I think we consider them all the same.
Considering route of administration
OBG
Dr. Levy: I think there’s always a benefit in having lots of choices. And for some women, being able to put a patch on once a week is much more convenient, easier to remember, and delivers a very consistent dose of hormone absorbed through the skin, which is different than taking a pill in the morning when your levels go up quickly then diminish over the day. The hormones are higher at a certain time, and then they drop off, so there might be some advantages for people who are very sensitive to swings in hormonal levels. There’s also a convenience factor, where for some people they will choose that. Other people might really dislike having a relatively large patch on their skin somewhere, or they may have skin sensitivity to the adhesive. Overall, I always think that having more options is better and individual girls/women will choose what works best for them.
Counseling tips
OBG
Dr. Levy: Like other patches that are available on the market, these are a once-a-week patch. The patch should be placed on clean, dry skin. No lotions, perfumes, or anything on the skin because you really want them to stick for the whole week, and it’s not going to stick if there’s anything oily on the skin. The first patch is placed on day 1 of a menstrual cycle, the first day of bleeding, and then changed weekly for 3 weeks. Then there’s a 7-day patch-free time in which one would expect to have a period.
In general, breakthrough bleeding was not a significant problem with the patch, but some women will have some irregular spotting and bleeding with any sort of hormonal treatment; some women may have no periods at all. In other words, the estrogen dose and progestin may be of a balance that allows the patient not to have periods. But, in general, most of the women in the trial had regular light menstrual flow during the week when their patch was not on.5
Continue to: Pricing...
Pricing
OBG
Dr. Levy: That’s a tricky question. Insurance plans through Obamacare, the Affordable Care Act, are required to cover every form of contraception. That means they must cover a patch. It doesn’t mean that they have to cover this patch. And because there are generics available of the other patch formulation, it is likely that this would be a higher tier, meaning that there may be a higher copay for someone who wanted to use Twirla versus one of the generic patches.
I can’t say that that’s universally the case, but my experience with most of the health insurance plans is that they tend to put barriers in the way for any of us to prescribe, and for women to use, brand-name products. So Twirla is new on the market; it’s a brand-name product. It may work much better for some people; and in those cases, the health care provider might have to send a letter to the insurance company saying why this one is medically necessary for a patient. There probably will be some hoops to go through for coverage without a copay. I think coverage will be there, but there may be a substantial copay because of the tier level.
OBG
Dr. Levy: I don’t think that the payer is going to buy that argument unless the requirement is to use a patch. If the patient, for example, has some sort of gastrointestinal disease where they don’t absorb things well, so pills don’t work well, we might get to the place where they have to have a patch. If the patient has a lot of breast tenderness or has symptoms on the generic patch that delivers a higher level of estrogen, then we would have to document those symptoms to say, “She’s not tolerating this one and, therefore, we need to go to that one.” So, I think as prescribers we would have to justify not only the lower dose but also the form.
As a clinician, I would always like to put somebody on the lower dose. We do think lower is better, but we have to be sensitive to the costs of all of these things too. I’m very sensitive to my patients’ out-of-pocket costs because, in the end, if the costs are a lot of money or she can only afford one month at a time, then she may miss a window where she may not have the money to buy next month’s supply when it’s due, and get pregnant. We have to balance all of those things as we’re thinking through the best option for an individual.
We have more to learn
OBG
Dr. Levy: I think it’s always exciting when we have new products available, and there’s a lot more we’ll learn as Twirla comes into commercial use and millions instead of thousands of people are using it. Overall, I think it’s fantastic that there’s ongoing research and that there are new products out there. And kudos to the company for doing the research and for getting approval, and I’m looking forward to learning more about it.
- Galzote RA, Rafie S, Teal R, et al. Transdermal delivery of combined hormonal contraception: a review of the current literature. Int J Womens Health. 2017;9:315-321.
- Contraceptive use in the United States. Guttmacher website. Published May 2021. Accessed August 29, 2021. https://www.guttmacher.org/fact-sheet/contraceptive-method-use-united-states#.
- US National Library of Medicine. Estrogen and progestin (transdermal patch contraceptives). MedlinePlus website. Updated February 15, 2021. Accessed August 29, 2021. https://medlineplus.gov/druginfo/meds/a602006.html.
- American College of Obstetricians and Gynecologists Practice Bulletin No. 206: use of hormonal contraception in women with coexisting medical conditions [published correction appears in: ACOG Committee on Practice Bulletins—gynecology. Obstet Gynecol. 2019;133:1288.] Obstet Gynecol. 2019;133:E128-E150.
- Nelson AL, Kaunitz AM, Kroll R, et al. Efficacy, safety, and tolerability of a levonorgestrel/ethinyl estradiol transdermal delivery system: phase 3 clinical trial results. Contraception. 2021;103:137-143. doi: 10.1016/j.contraception.2020.
11.011.
The first transdermal contraceptive patch was approved by the US Food and Drug Administration (FDA) in 2001.1 A 2018 survey revealed that 5% of women in the United States between the ages of 15 and 49 years reported the use of a short-acting hormonal contraceptive method (ie, vaginal ring, transdermal patch, injectable) within the past month, with just 0.3% reporting the use of a transdermal patch.2 Transdermal contraceptive patches are an effective form of birth control that may be a convenient option for patients who do not want to take a daily oral contraceptive pill but want similar efficacy and tolerability. Typical failure rates of patches are similar to that of combined oral contraceptives (COCs).1,3
While transdermal hormone delivery results in less peaks and troughs of estrogen compared with COCs, the total estrogen exposure is higher than with COCs; therefore, the risk for venous thromboembolism (VTE) with previously available patches is about twice as high.1 Twirla (Agile), an ethinyl estradiol (EE)/levonorgestrel (LNG) patch, delivers a low and consistent daily dose of hormones over 3 patches replaced once weekly, with no patch on the fourth week.3 Twirla contains 120 μg/day LNG and 30 μg/day EE. OrthoEvra, FDA approved in 2001 as mentioned, contains 150 μg/day norelgestromin and 35 μg/day EE.1 A reduction of the EE dose in COCs has been associated with lower risk for VTE.4
The addition of Twirla to the market offers another contraceptive option for patients who opt for a weekly, self-administered method.
How much lower is the VTE risk?
OBG
Barbara Levy, MD: The reality is we can’t designate a reduction of risk, except, in general, when the dose of ethinyl estradiol is lower, we think that the VTE risk is lower. There has not been a head-to-head comparison in a large enough population to be able to say that the risk is reduced by a certain factor. We just look at the overall exposure to estrogen and say, “In general, for VTE risk, a lower dose is a better thing for women.”
That being said, look at birth control pills, like COCs. We don’t have actual numbers to say that a 30-μg pill is this much less risky than a 35-μg pill. We just put it into a hierarchy, and that’s what we can do with the patch. We can say that, in general, lower is better for VTE risk, but no one can provide absolute numbers.
Continue to: Efficacy...
Efficacy
OBG
Dr. Levy: You have to look at the pivotal trials and look at what the efficacy was in a trial setting. In the real-world setting, the effectiveness is never quite as good as it is in a clinical trial. I think the bottom line for all of us is that combined oral contraception, meaning estrogen with progestin, is equivalently effective across the different options that are available for women. Efficacy really isn’t the factor to use to distinguish which one I’m going to pick. It is about the patient’s convenience and many other factors. But in terms of its clinical effectiveness in preventing pregnancy, from a very practical standpoint, I think we consider them all the same.
Considering route of administration
OBG
Dr. Levy: I think there’s always a benefit in having lots of choices. And for some women, being able to put a patch on once a week is much more convenient, easier to remember, and delivers a very consistent dose of hormone absorbed through the skin, which is different than taking a pill in the morning when your levels go up quickly then diminish over the day. The hormones are higher at a certain time, and then they drop off, so there might be some advantages for people who are very sensitive to swings in hormonal levels. There’s also a convenience factor, where for some people they will choose that. Other people might really dislike having a relatively large patch on their skin somewhere, or they may have skin sensitivity to the adhesive. Overall, I always think that having more options is better and individual girls/women will choose what works best for them.
Counseling tips
OBG
Dr. Levy: Like other patches that are available on the market, these are a once-a-week patch. The patch should be placed on clean, dry skin. No lotions, perfumes, or anything on the skin because you really want them to stick for the whole week, and it’s not going to stick if there’s anything oily on the skin. The first patch is placed on day 1 of a menstrual cycle, the first day of bleeding, and then changed weekly for 3 weeks. Then there’s a 7-day patch-free time in which one would expect to have a period.
In general, breakthrough bleeding was not a significant problem with the patch, but some women will have some irregular spotting and bleeding with any sort of hormonal treatment; some women may have no periods at all. In other words, the estrogen dose and progestin may be of a balance that allows the patient not to have periods. But, in general, most of the women in the trial had regular light menstrual flow during the week when their patch was not on.5
Continue to: Pricing...
Pricing
OBG
Dr. Levy: That’s a tricky question. Insurance plans through Obamacare, the Affordable Care Act, are required to cover every form of contraception. That means they must cover a patch. It doesn’t mean that they have to cover this patch. And because there are generics available of the other patch formulation, it is likely that this would be a higher tier, meaning that there may be a higher copay for someone who wanted to use Twirla versus one of the generic patches.
I can’t say that that’s universally the case, but my experience with most of the health insurance plans is that they tend to put barriers in the way for any of us to prescribe, and for women to use, brand-name products. So Twirla is new on the market; it’s a brand-name product. It may work much better for some people; and in those cases, the health care provider might have to send a letter to the insurance company saying why this one is medically necessary for a patient. There probably will be some hoops to go through for coverage without a copay. I think coverage will be there, but there may be a substantial copay because of the tier level.
OBG
Dr. Levy: I don’t think that the payer is going to buy that argument unless the requirement is to use a patch. If the patient, for example, has some sort of gastrointestinal disease where they don’t absorb things well, so pills don’t work well, we might get to the place where they have to have a patch. If the patient has a lot of breast tenderness or has symptoms on the generic patch that delivers a higher level of estrogen, then we would have to document those symptoms to say, “She’s not tolerating this one and, therefore, we need to go to that one.” So, I think as prescribers we would have to justify not only the lower dose but also the form.
As a clinician, I would always like to put somebody on the lower dose. We do think lower is better, but we have to be sensitive to the costs of all of these things too. I’m very sensitive to my patients’ out-of-pocket costs because, in the end, if the costs are a lot of money or she can only afford one month at a time, then she may miss a window where she may not have the money to buy next month’s supply when it’s due, and get pregnant. We have to balance all of those things as we’re thinking through the best option for an individual.
We have more to learn
OBG
Dr. Levy: I think it’s always exciting when we have new products available, and there’s a lot more we’ll learn as Twirla comes into commercial use and millions instead of thousands of people are using it. Overall, I think it’s fantastic that there’s ongoing research and that there are new products out there. And kudos to the company for doing the research and for getting approval, and I’m looking forward to learning more about it.
The first transdermal contraceptive patch was approved by the US Food and Drug Administration (FDA) in 2001.1 A 2018 survey revealed that 5% of women in the United States between the ages of 15 and 49 years reported the use of a short-acting hormonal contraceptive method (ie, vaginal ring, transdermal patch, injectable) within the past month, with just 0.3% reporting the use of a transdermal patch.2 Transdermal contraceptive patches are an effective form of birth control that may be a convenient option for patients who do not want to take a daily oral contraceptive pill but want similar efficacy and tolerability. Typical failure rates of patches are similar to that of combined oral contraceptives (COCs).1,3
While transdermal hormone delivery results in less peaks and troughs of estrogen compared with COCs, the total estrogen exposure is higher than with COCs; therefore, the risk for venous thromboembolism (VTE) with previously available patches is about twice as high.1 Twirla (Agile), an ethinyl estradiol (EE)/levonorgestrel (LNG) patch, delivers a low and consistent daily dose of hormones over 3 patches replaced once weekly, with no patch on the fourth week.3 Twirla contains 120 μg/day LNG and 30 μg/day EE. OrthoEvra, FDA approved in 2001 as mentioned, contains 150 μg/day norelgestromin and 35 μg/day EE.1 A reduction of the EE dose in COCs has been associated with lower risk for VTE.4
The addition of Twirla to the market offers another contraceptive option for patients who opt for a weekly, self-administered method.
How much lower is the VTE risk?
OBG
Barbara Levy, MD: The reality is we can’t designate a reduction of risk, except, in general, when the dose of ethinyl estradiol is lower, we think that the VTE risk is lower. There has not been a head-to-head comparison in a large enough population to be able to say that the risk is reduced by a certain factor. We just look at the overall exposure to estrogen and say, “In general, for VTE risk, a lower dose is a better thing for women.”
That being said, look at birth control pills, like COCs. We don’t have actual numbers to say that a 30-μg pill is this much less risky than a 35-μg pill. We just put it into a hierarchy, and that’s what we can do with the patch. We can say that, in general, lower is better for VTE risk, but no one can provide absolute numbers.
Continue to: Efficacy...
Efficacy
OBG
Dr. Levy: You have to look at the pivotal trials and look at what the efficacy was in a trial setting. In the real-world setting, the effectiveness is never quite as good as it is in a clinical trial. I think the bottom line for all of us is that combined oral contraception, meaning estrogen with progestin, is equivalently effective across the different options that are available for women. Efficacy really isn’t the factor to use to distinguish which one I’m going to pick. It is about the patient’s convenience and many other factors. But in terms of its clinical effectiveness in preventing pregnancy, from a very practical standpoint, I think we consider them all the same.
Considering route of administration
OBG
Dr. Levy: I think there’s always a benefit in having lots of choices. And for some women, being able to put a patch on once a week is much more convenient, easier to remember, and delivers a very consistent dose of hormone absorbed through the skin, which is different than taking a pill in the morning when your levels go up quickly then diminish over the day. The hormones are higher at a certain time, and then they drop off, so there might be some advantages for people who are very sensitive to swings in hormonal levels. There’s also a convenience factor, where for some people they will choose that. Other people might really dislike having a relatively large patch on their skin somewhere, or they may have skin sensitivity to the adhesive. Overall, I always think that having more options is better and individual girls/women will choose what works best for them.
Counseling tips
OBG
Dr. Levy: Like other patches that are available on the market, these are a once-a-week patch. The patch should be placed on clean, dry skin. No lotions, perfumes, or anything on the skin because you really want them to stick for the whole week, and it’s not going to stick if there’s anything oily on the skin. The first patch is placed on day 1 of a menstrual cycle, the first day of bleeding, and then changed weekly for 3 weeks. Then there’s a 7-day patch-free time in which one would expect to have a period.
In general, breakthrough bleeding was not a significant problem with the patch, but some women will have some irregular spotting and bleeding with any sort of hormonal treatment; some women may have no periods at all. In other words, the estrogen dose and progestin may be of a balance that allows the patient not to have periods. But, in general, most of the women in the trial had regular light menstrual flow during the week when their patch was not on.5
Continue to: Pricing...
Pricing
OBG
Dr. Levy: That’s a tricky question. Insurance plans through Obamacare, the Affordable Care Act, are required to cover every form of contraception. That means they must cover a patch. It doesn’t mean that they have to cover this patch. And because there are generics available of the other patch formulation, it is likely that this would be a higher tier, meaning that there may be a higher copay for someone who wanted to use Twirla versus one of the generic patches.
I can’t say that that’s universally the case, but my experience with most of the health insurance plans is that they tend to put barriers in the way for any of us to prescribe, and for women to use, brand-name products. So Twirla is new on the market; it’s a brand-name product. It may work much better for some people; and in those cases, the health care provider might have to send a letter to the insurance company saying why this one is medically necessary for a patient. There probably will be some hoops to go through for coverage without a copay. I think coverage will be there, but there may be a substantial copay because of the tier level.
OBG
Dr. Levy: I don’t think that the payer is going to buy that argument unless the requirement is to use a patch. If the patient, for example, has some sort of gastrointestinal disease where they don’t absorb things well, so pills don’t work well, we might get to the place where they have to have a patch. If the patient has a lot of breast tenderness or has symptoms on the generic patch that delivers a higher level of estrogen, then we would have to document those symptoms to say, “She’s not tolerating this one and, therefore, we need to go to that one.” So, I think as prescribers we would have to justify not only the lower dose but also the form.
As a clinician, I would always like to put somebody on the lower dose. We do think lower is better, but we have to be sensitive to the costs of all of these things too. I’m very sensitive to my patients’ out-of-pocket costs because, in the end, if the costs are a lot of money or she can only afford one month at a time, then she may miss a window where she may not have the money to buy next month’s supply when it’s due, and get pregnant. We have to balance all of those things as we’re thinking through the best option for an individual.
We have more to learn
OBG
Dr. Levy: I think it’s always exciting when we have new products available, and there’s a lot more we’ll learn as Twirla comes into commercial use and millions instead of thousands of people are using it. Overall, I think it’s fantastic that there’s ongoing research and that there are new products out there. And kudos to the company for doing the research and for getting approval, and I’m looking forward to learning more about it.
- Galzote RA, Rafie S, Teal R, et al. Transdermal delivery of combined hormonal contraception: a review of the current literature. Int J Womens Health. 2017;9:315-321.
- Contraceptive use in the United States. Guttmacher website. Published May 2021. Accessed August 29, 2021. https://www.guttmacher.org/fact-sheet/contraceptive-method-use-united-states#.
- US National Library of Medicine. Estrogen and progestin (transdermal patch contraceptives). MedlinePlus website. Updated February 15, 2021. Accessed August 29, 2021. https://medlineplus.gov/druginfo/meds/a602006.html.
- American College of Obstetricians and Gynecologists Practice Bulletin No. 206: use of hormonal contraception in women with coexisting medical conditions [published correction appears in: ACOG Committee on Practice Bulletins—gynecology. Obstet Gynecol. 2019;133:1288.] Obstet Gynecol. 2019;133:E128-E150.
- Nelson AL, Kaunitz AM, Kroll R, et al. Efficacy, safety, and tolerability of a levonorgestrel/ethinyl estradiol transdermal delivery system: phase 3 clinical trial results. Contraception. 2021;103:137-143. doi: 10.1016/j.contraception.2020.
11.011.
- Galzote RA, Rafie S, Teal R, et al. Transdermal delivery of combined hormonal contraception: a review of the current literature. Int J Womens Health. 2017;9:315-321.
- Contraceptive use in the United States. Guttmacher website. Published May 2021. Accessed August 29, 2021. https://www.guttmacher.org/fact-sheet/contraceptive-method-use-united-states#.
- US National Library of Medicine. Estrogen and progestin (transdermal patch contraceptives). MedlinePlus website. Updated February 15, 2021. Accessed August 29, 2021. https://medlineplus.gov/druginfo/meds/a602006.html.
- American College of Obstetricians and Gynecologists Practice Bulletin No. 206: use of hormonal contraception in women with coexisting medical conditions [published correction appears in: ACOG Committee on Practice Bulletins—gynecology. Obstet Gynecol. 2019;133:1288.] Obstet Gynecol. 2019;133:E128-E150.
- Nelson AL, Kaunitz AM, Kroll R, et al. Efficacy, safety, and tolerability of a levonorgestrel/ethinyl estradiol transdermal delivery system: phase 3 clinical trial results. Contraception. 2021;103:137-143. doi: 10.1016/j.contraception.2020.
11.011.
Accelerated approval now full for pembro in bladder cancer
But one such accelerated approval has now been converted to a full approval, with a small label change, by the Food and Drug Administration.
The new full approval is for pembrolizumab (Keytruda) for first-line use for patients with locally advanced or metastatic urothelial carcinoma (mUC) who are not eligible for any platinum-containing chemotherapy. (The small label change is that mention of PD-L1 status and testing for this have been removed.)
The move is in accordance with recommendations by experts at a recent meeting of the Oncologic Drugs Advisory Committee. They voted 5-3 in favor of this accelerated approval staying. They also voted 10-1 in favor of another immunotherapy, atezolizumab (Tecentriq), for the same indication.
One of the arguments put forward to support these accelerated approvals staying in place is that there is an unmet need in this population of patients who are ineligible for platinum chemotherapy.
But this argument doesn’t hold water – the mere existence of one of these negates the “unmet need” argument for the other, wrote Bishal Gyawali, MD, PhD, from Queen’s University, Kingston, Ont., in a commentary on why the FDA’s accelerated approval pathway is broken.
“Even if there is a genuine ‘unmet need’ in a particular setting, these drugs did not meet the standard of improving survival. An unmet need doesn’t imply that the treatment void should be filled with a drug that provides nothing of value to patients,” Dr. Gyawali wrote.
“When we talk about an unmet need, we are speaking of drugs that provide a clinical benefit; any true unmet needs will continue to exist despite maintaining these approvals,” he argued.
After obtaining the accelerated approval for pembrolizumab for patients with bladder cancer who are not eligible for cisplatin-containing chemotherapy, the manufacturer (Merck) carried out a subsequent clinical trial but conducted it in patients who were eligible for platinum-containing chemotherapy (KEYNOTE-361). However, this trial did not meet its prespecified dual primary endpoints of overall survival or progression-free survival in comparison with standard of care chemotherapy, the company noted in a press release.
“We are working with urgency to advance studies to help more patients living with bladder and other types of cancer,” commented Scot Ebbinghaus, MD, vice president of clinical research, Merck Research Laboratories. The company said it has “an extensive clinical development in bladder cancer” and is exploring pembrolizumab use in many settings.
In addition to the new full approval for the first-line indication for patients who are ineligible for platinum chemotherapy, pembrolizumab has two other approved indications in this therapeutic area: the treatment of patients with locally advanced urothelial carcinoma or mUC who experience disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy; and the treatment of patients with bacillus Calmette-Guérin–unresponsive, high-risk, non–muscle-invasive bladder cancer with carcinoma in situ, with or without papillary tumors, who are ineligible for or have elected not to undergo cystectomy.
A version of this article first appeared on Medscape.com.
But one such accelerated approval has now been converted to a full approval, with a small label change, by the Food and Drug Administration.
The new full approval is for pembrolizumab (Keytruda) for first-line use for patients with locally advanced or metastatic urothelial carcinoma (mUC) who are not eligible for any platinum-containing chemotherapy. (The small label change is that mention of PD-L1 status and testing for this have been removed.)
The move is in accordance with recommendations by experts at a recent meeting of the Oncologic Drugs Advisory Committee. They voted 5-3 in favor of this accelerated approval staying. They also voted 10-1 in favor of another immunotherapy, atezolizumab (Tecentriq), for the same indication.
One of the arguments put forward to support these accelerated approvals staying in place is that there is an unmet need in this population of patients who are ineligible for platinum chemotherapy.
But this argument doesn’t hold water – the mere existence of one of these negates the “unmet need” argument for the other, wrote Bishal Gyawali, MD, PhD, from Queen’s University, Kingston, Ont., in a commentary on why the FDA’s accelerated approval pathway is broken.
“Even if there is a genuine ‘unmet need’ in a particular setting, these drugs did not meet the standard of improving survival. An unmet need doesn’t imply that the treatment void should be filled with a drug that provides nothing of value to patients,” Dr. Gyawali wrote.
“When we talk about an unmet need, we are speaking of drugs that provide a clinical benefit; any true unmet needs will continue to exist despite maintaining these approvals,” he argued.
After obtaining the accelerated approval for pembrolizumab for patients with bladder cancer who are not eligible for cisplatin-containing chemotherapy, the manufacturer (Merck) carried out a subsequent clinical trial but conducted it in patients who were eligible for platinum-containing chemotherapy (KEYNOTE-361). However, this trial did not meet its prespecified dual primary endpoints of overall survival or progression-free survival in comparison with standard of care chemotherapy, the company noted in a press release.
“We are working with urgency to advance studies to help more patients living with bladder and other types of cancer,” commented Scot Ebbinghaus, MD, vice president of clinical research, Merck Research Laboratories. The company said it has “an extensive clinical development in bladder cancer” and is exploring pembrolizumab use in many settings.
In addition to the new full approval for the first-line indication for patients who are ineligible for platinum chemotherapy, pembrolizumab has two other approved indications in this therapeutic area: the treatment of patients with locally advanced urothelial carcinoma or mUC who experience disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy; and the treatment of patients with bacillus Calmette-Guérin–unresponsive, high-risk, non–muscle-invasive bladder cancer with carcinoma in situ, with or without papillary tumors, who are ineligible for or have elected not to undergo cystectomy.
A version of this article first appeared on Medscape.com.
But one such accelerated approval has now been converted to a full approval, with a small label change, by the Food and Drug Administration.
The new full approval is for pembrolizumab (Keytruda) for first-line use for patients with locally advanced or metastatic urothelial carcinoma (mUC) who are not eligible for any platinum-containing chemotherapy. (The small label change is that mention of PD-L1 status and testing for this have been removed.)
The move is in accordance with recommendations by experts at a recent meeting of the Oncologic Drugs Advisory Committee. They voted 5-3 in favor of this accelerated approval staying. They also voted 10-1 in favor of another immunotherapy, atezolizumab (Tecentriq), for the same indication.
One of the arguments put forward to support these accelerated approvals staying in place is that there is an unmet need in this population of patients who are ineligible for platinum chemotherapy.
But this argument doesn’t hold water – the mere existence of one of these negates the “unmet need” argument for the other, wrote Bishal Gyawali, MD, PhD, from Queen’s University, Kingston, Ont., in a commentary on why the FDA’s accelerated approval pathway is broken.
“Even if there is a genuine ‘unmet need’ in a particular setting, these drugs did not meet the standard of improving survival. An unmet need doesn’t imply that the treatment void should be filled with a drug that provides nothing of value to patients,” Dr. Gyawali wrote.
“When we talk about an unmet need, we are speaking of drugs that provide a clinical benefit; any true unmet needs will continue to exist despite maintaining these approvals,” he argued.
After obtaining the accelerated approval for pembrolizumab for patients with bladder cancer who are not eligible for cisplatin-containing chemotherapy, the manufacturer (Merck) carried out a subsequent clinical trial but conducted it in patients who were eligible for platinum-containing chemotherapy (KEYNOTE-361). However, this trial did not meet its prespecified dual primary endpoints of overall survival or progression-free survival in comparison with standard of care chemotherapy, the company noted in a press release.
“We are working with urgency to advance studies to help more patients living with bladder and other types of cancer,” commented Scot Ebbinghaus, MD, vice president of clinical research, Merck Research Laboratories. The company said it has “an extensive clinical development in bladder cancer” and is exploring pembrolizumab use in many settings.
In addition to the new full approval for the first-line indication for patients who are ineligible for platinum chemotherapy, pembrolizumab has two other approved indications in this therapeutic area: the treatment of patients with locally advanced urothelial carcinoma or mUC who experience disease progression during or following platinum-containing chemotherapy or within 12 months of neoadjuvant or adjuvant treatment with platinum-containing chemotherapy; and the treatment of patients with bacillus Calmette-Guérin–unresponsive, high-risk, non–muscle-invasive bladder cancer with carcinoma in situ, with or without papillary tumors, who are ineligible for or have elected not to undergo cystectomy.
A version of this article first appeared on Medscape.com.