User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Should antenatal testing be performed in patients with a pre-pregnancy BMI ≥ 35?
Possibly. Elevated body mass index (BMI) is associated with an increased risk for stillbirth (strength of recommendation (SOR), B; Cohort studies and meta-analysis of cohort studies). Three studies found an association between elevated BMI and stillbirth and one did not. However, no studies demonstrate that antenatal testing in pregnant people with higher BMIs decreases stillbirth rates, or that no harm is caused by unnecessary testing or resultant interventions.
Still, in 2021, the American College of Obstetricians and Gynecologists (ACOG) suggested weekly antenatal testing may be considered from 34 weeks' 0 days' gestation for pregnant people with a BMI ≥ 40.0 kg/m2 and from 37 weeks' 0 days' gestation for pregnant people with a BMI between 35.0 and 39.9 kg/m2 (SOR, C; consensus guideline). Thus, doing the antenatal testing recommended in the ACOG guideline in an attempt to prevent stillbirth is reasonable, given evidence that elevated BMI is associated with stillbirth.
Evidence summary
Association between higher maternal BMI and increased risk for stillbirth
The purpose of antenatal testing is to decrease the risk for stillbirth between visits. Because of the resources involved and the risk for false-positives when testing low-risk patients, antenatal testing is reserved for pregnant people with higher risk for stillbirth.
In a retrospective cohort study of more than 2.8 million singleton births including 9,030 stillbirths, pregnant people with an elevated BMI had an increased risk for stillbirth compared with those with a normal BMI. The adjusted hazard ratio was 1.71 (95% confidence interval (CI), 1.62-1.83) for those with a BMI of 30.0 to 34.9 kg/m2; 2.04 (95% CI, 1.8-2.21) for those with a BMI of 35.0 to 39.9 kg/m2; and 2.50 (95% CI, 2.28-2.74) for those with a BMI ≥ 40 kg/m2.1
A meta-analysis of 38 studies, which included data on 16,274 stillbirths, found that a 5-unit increase in BMI was associated with an increased risk for stillbirth (relative risk, 1.24; 95% CI, 1.18-1.30).2
Another meta-analysis included 6 cohort studies involving more than 1 million pregnancies and 3 case-control studies involving 2,530 stillbirths and 2,837 controls from 1980-2005. There was an association between increasing BMI and stillbirth: the odds ratio (OR) was 1.47 (95% CI, 1.08-1.94) for those with a BMI of 25.0 to 29.9 kg/m2 and 2.07 (95% CI, 1.59-2.74) for those with a BMI ≥ 30.0, compared with those with a normal BMI.3
However, a retrospective cohort study of 182,362 singleton births including 442 stillbirths found no association between stillbirth and increasing BMI. The OR was 1.10 (95% CI, 0.90-1.36) for those with a BMI of 25.0 to 29.9 and 1.09 (95% CI, 0.87-1.37) for those with a BMI ≥ 30.0 kg/m2, compared with those with a normal BMI.4 However, this cohort study may have been underpowered to detect an association between stillbirth and BMI.
Recommendations from others
In 2021, ACOG suggested that weekly antenatal testing may be considered from 34 weeks' and 0 days' gestation for pregnant people with a BMI ≥ 40.0 kg/m2 and from 37 weeks' and 0 days' gestation for pregnant people with a BMI between 35.0 and 39.9 kg/m2.5 The 2021 ACOG Practice Bulletin on obesity in pregnancy rates this recommendation as Level C—based primarily on consensus and expert opinion.6
A 2018 Royal College of Obstetricians and Gynecologists Green-top Guideline recognizes “definitive recommendations for fetal surveillance are hampered by the lack of randomized controlled trials demonstrating that antepartum fetal surveillance decreases perinatal morbidity or mortality in late-term and post-term gestations…. There are no definitive studies determining the optimal type or frequency of such testing and no evidence specific for women with obesity.”7
A 2019 Society of Obstetricians and Gynecologists of Canada practice guideline states “stillbirth is more common with maternal obesity” and recommends “increased fetal surveillance … in the third trimester if reduced fetal movements are reported.” The guideline notes “the role for non-stress tests … in surveillance of well-being in this population is uncertain.” Also, for pregnant people with a BMI > 30 kg/m2, “assessment of fetal well-being is … recommended weekly from 37 weeks until delivery.” Finally, increased fetal surveillance is recommended in the setting of increased BMI and an abnormal pulsatility index of the umbilical artery and/or maternal uterine artery.8
Editor’s takeaway
Evidence demonstrates that increased maternal BMI is associated with increased stillbirths. However, evidence has not shown that third-trimester antenatal testing decreases this morbidity and mortality. Expert opinion varies, with ACOG recommending weekly antenatal testing from 34 and 37 weeks’ gestation, respectively, for pregnant people with BMIs of ≥ 40 kg/m2 and of 35 to 39.9 kg/m2. ●
- Yao R, Ananth C, Park B, et al; Perinatal Research Consortium. Obesity and the risk of stillbirth: a population-based cohort study. Am J Obstet Gynecol. 2014;210:e1-e9. doi: 10.1016/j. ajog. 2014.01.044
- Aune D, Saugstad O, Henriksen T, et al. Maternal body mass index and the risk of fetal death, stillbirth, and infant death: a systematic review and meta-analysis. JAMA. 2014;311:15361546. doi: 10.1001/jama.2014.2269
- Chu S, Kim S, Lau J, et al. Maternal obesity and risk of stillbirth: a meta-analysis. Am J Obstet Gynecol. 2007;197:223-228. doi: 10.1016/j.ajog.2007.03.027
- Mahomed K, Chan G, Norton M. Obesity and the risk of stillbirth—a reappraisal—a retrospective cohort study. Eur J Obstet Gynecol Reprod Biol. 2020;255:25-28. doi: 10.1016/j. ejogrb. 2020.09.044
- American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, Society for MaternalFetal Medicine. Indications for outpatient antenatal fetal surveillance: ACOG committee opinion, number 828. Obstet Gynecol. 2021;137:e177-e197. doi: 10.1097/ AOG.0000000000004407
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Obesity in pregnancy: ACOG practice bulletin, number 230. Obstet Gynecol. 2021;137:e128-e144. doi: 10.1097/ AOG.0000000000004395
- Denison F, Aedla N, Keag O, et al; Royal College of Obstetricians and Gynaecologists. Care of women with obesity in pregnancy: Green-top Guideline No. 72. BJOG. 2019;126:e62-e106. doi: 10.1111/1471-0528.15386
- Maxwell C, Gaudet L, Cassir G, et al. Guideline No. 391Pregnancy and maternal obesity part 1: pre-conception and prenatal care. J Obstet Gynaecol Can. 2019;41:1623-1640. doi: 10.1016/j.jogc. 2019.03.026
Possibly. Elevated body mass index (BMI) is associated with an increased risk for stillbirth (strength of recommendation (SOR), B; Cohort studies and meta-analysis of cohort studies). Three studies found an association between elevated BMI and stillbirth and one did not. However, no studies demonstrate that antenatal testing in pregnant people with higher BMIs decreases stillbirth rates, or that no harm is caused by unnecessary testing or resultant interventions.
Still, in 2021, the American College of Obstetricians and Gynecologists (ACOG) suggested weekly antenatal testing may be considered from 34 weeks' 0 days' gestation for pregnant people with a BMI ≥ 40.0 kg/m2 and from 37 weeks' 0 days' gestation for pregnant people with a BMI between 35.0 and 39.9 kg/m2 (SOR, C; consensus guideline). Thus, doing the antenatal testing recommended in the ACOG guideline in an attempt to prevent stillbirth is reasonable, given evidence that elevated BMI is associated with stillbirth.
Evidence summary
Association between higher maternal BMI and increased risk for stillbirth
The purpose of antenatal testing is to decrease the risk for stillbirth between visits. Because of the resources involved and the risk for false-positives when testing low-risk patients, antenatal testing is reserved for pregnant people with higher risk for stillbirth.
In a retrospective cohort study of more than 2.8 million singleton births including 9,030 stillbirths, pregnant people with an elevated BMI had an increased risk for stillbirth compared with those with a normal BMI. The adjusted hazard ratio was 1.71 (95% confidence interval (CI), 1.62-1.83) for those with a BMI of 30.0 to 34.9 kg/m2; 2.04 (95% CI, 1.8-2.21) for those with a BMI of 35.0 to 39.9 kg/m2; and 2.50 (95% CI, 2.28-2.74) for those with a BMI ≥ 40 kg/m2.1
A meta-analysis of 38 studies, which included data on 16,274 stillbirths, found that a 5-unit increase in BMI was associated with an increased risk for stillbirth (relative risk, 1.24; 95% CI, 1.18-1.30).2
Another meta-analysis included 6 cohort studies involving more than 1 million pregnancies and 3 case-control studies involving 2,530 stillbirths and 2,837 controls from 1980-2005. There was an association between increasing BMI and stillbirth: the odds ratio (OR) was 1.47 (95% CI, 1.08-1.94) for those with a BMI of 25.0 to 29.9 kg/m2 and 2.07 (95% CI, 1.59-2.74) for those with a BMI ≥ 30.0, compared with those with a normal BMI.3
However, a retrospective cohort study of 182,362 singleton births including 442 stillbirths found no association between stillbirth and increasing BMI. The OR was 1.10 (95% CI, 0.90-1.36) for those with a BMI of 25.0 to 29.9 and 1.09 (95% CI, 0.87-1.37) for those with a BMI ≥ 30.0 kg/m2, compared with those with a normal BMI.4 However, this cohort study may have been underpowered to detect an association between stillbirth and BMI.
Recommendations from others
In 2021, ACOG suggested that weekly antenatal testing may be considered from 34 weeks' and 0 days' gestation for pregnant people with a BMI ≥ 40.0 kg/m2 and from 37 weeks' and 0 days' gestation for pregnant people with a BMI between 35.0 and 39.9 kg/m2.5 The 2021 ACOG Practice Bulletin on obesity in pregnancy rates this recommendation as Level C—based primarily on consensus and expert opinion.6
A 2018 Royal College of Obstetricians and Gynecologists Green-top Guideline recognizes “definitive recommendations for fetal surveillance are hampered by the lack of randomized controlled trials demonstrating that antepartum fetal surveillance decreases perinatal morbidity or mortality in late-term and post-term gestations…. There are no definitive studies determining the optimal type or frequency of such testing and no evidence specific for women with obesity.”7
A 2019 Society of Obstetricians and Gynecologists of Canada practice guideline states “stillbirth is more common with maternal obesity” and recommends “increased fetal surveillance … in the third trimester if reduced fetal movements are reported.” The guideline notes “the role for non-stress tests … in surveillance of well-being in this population is uncertain.” Also, for pregnant people with a BMI > 30 kg/m2, “assessment of fetal well-being is … recommended weekly from 37 weeks until delivery.” Finally, increased fetal surveillance is recommended in the setting of increased BMI and an abnormal pulsatility index of the umbilical artery and/or maternal uterine artery.8
Editor’s takeaway
Evidence demonstrates that increased maternal BMI is associated with increased stillbirths. However, evidence has not shown that third-trimester antenatal testing decreases this morbidity and mortality. Expert opinion varies, with ACOG recommending weekly antenatal testing from 34 and 37 weeks’ gestation, respectively, for pregnant people with BMIs of ≥ 40 kg/m2 and of 35 to 39.9 kg/m2. ●
Possibly. Elevated body mass index (BMI) is associated with an increased risk for stillbirth (strength of recommendation (SOR), B; Cohort studies and meta-analysis of cohort studies). Three studies found an association between elevated BMI and stillbirth and one did not. However, no studies demonstrate that antenatal testing in pregnant people with higher BMIs decreases stillbirth rates, or that no harm is caused by unnecessary testing or resultant interventions.
Still, in 2021, the American College of Obstetricians and Gynecologists (ACOG) suggested weekly antenatal testing may be considered from 34 weeks' 0 days' gestation for pregnant people with a BMI ≥ 40.0 kg/m2 and from 37 weeks' 0 days' gestation for pregnant people with a BMI between 35.0 and 39.9 kg/m2 (SOR, C; consensus guideline). Thus, doing the antenatal testing recommended in the ACOG guideline in an attempt to prevent stillbirth is reasonable, given evidence that elevated BMI is associated with stillbirth.
Evidence summary
Association between higher maternal BMI and increased risk for stillbirth
The purpose of antenatal testing is to decrease the risk for stillbirth between visits. Because of the resources involved and the risk for false-positives when testing low-risk patients, antenatal testing is reserved for pregnant people with higher risk for stillbirth.
In a retrospective cohort study of more than 2.8 million singleton births including 9,030 stillbirths, pregnant people with an elevated BMI had an increased risk for stillbirth compared with those with a normal BMI. The adjusted hazard ratio was 1.71 (95% confidence interval (CI), 1.62-1.83) for those with a BMI of 30.0 to 34.9 kg/m2; 2.04 (95% CI, 1.8-2.21) for those with a BMI of 35.0 to 39.9 kg/m2; and 2.50 (95% CI, 2.28-2.74) for those with a BMI ≥ 40 kg/m2.1
A meta-analysis of 38 studies, which included data on 16,274 stillbirths, found that a 5-unit increase in BMI was associated with an increased risk for stillbirth (relative risk, 1.24; 95% CI, 1.18-1.30).2
Another meta-analysis included 6 cohort studies involving more than 1 million pregnancies and 3 case-control studies involving 2,530 stillbirths and 2,837 controls from 1980-2005. There was an association between increasing BMI and stillbirth: the odds ratio (OR) was 1.47 (95% CI, 1.08-1.94) for those with a BMI of 25.0 to 29.9 kg/m2 and 2.07 (95% CI, 1.59-2.74) for those with a BMI ≥ 30.0, compared with those with a normal BMI.3
However, a retrospective cohort study of 182,362 singleton births including 442 stillbirths found no association between stillbirth and increasing BMI. The OR was 1.10 (95% CI, 0.90-1.36) for those with a BMI of 25.0 to 29.9 and 1.09 (95% CI, 0.87-1.37) for those with a BMI ≥ 30.0 kg/m2, compared with those with a normal BMI.4 However, this cohort study may have been underpowered to detect an association between stillbirth and BMI.
Recommendations from others
In 2021, ACOG suggested that weekly antenatal testing may be considered from 34 weeks' and 0 days' gestation for pregnant people with a BMI ≥ 40.0 kg/m2 and from 37 weeks' and 0 days' gestation for pregnant people with a BMI between 35.0 and 39.9 kg/m2.5 The 2021 ACOG Practice Bulletin on obesity in pregnancy rates this recommendation as Level C—based primarily on consensus and expert opinion.6
A 2018 Royal College of Obstetricians and Gynecologists Green-top Guideline recognizes “definitive recommendations for fetal surveillance are hampered by the lack of randomized controlled trials demonstrating that antepartum fetal surveillance decreases perinatal morbidity or mortality in late-term and post-term gestations…. There are no definitive studies determining the optimal type or frequency of such testing and no evidence specific for women with obesity.”7
A 2019 Society of Obstetricians and Gynecologists of Canada practice guideline states “stillbirth is more common with maternal obesity” and recommends “increased fetal surveillance … in the third trimester if reduced fetal movements are reported.” The guideline notes “the role for non-stress tests … in surveillance of well-being in this population is uncertain.” Also, for pregnant people with a BMI > 30 kg/m2, “assessment of fetal well-being is … recommended weekly from 37 weeks until delivery.” Finally, increased fetal surveillance is recommended in the setting of increased BMI and an abnormal pulsatility index of the umbilical artery and/or maternal uterine artery.8
Editor’s takeaway
Evidence demonstrates that increased maternal BMI is associated with increased stillbirths. However, evidence has not shown that third-trimester antenatal testing decreases this morbidity and mortality. Expert opinion varies, with ACOG recommending weekly antenatal testing from 34 and 37 weeks’ gestation, respectively, for pregnant people with BMIs of ≥ 40 kg/m2 and of 35 to 39.9 kg/m2. ●
- Yao R, Ananth C, Park B, et al; Perinatal Research Consortium. Obesity and the risk of stillbirth: a population-based cohort study. Am J Obstet Gynecol. 2014;210:e1-e9. doi: 10.1016/j. ajog. 2014.01.044
- Aune D, Saugstad O, Henriksen T, et al. Maternal body mass index and the risk of fetal death, stillbirth, and infant death: a systematic review and meta-analysis. JAMA. 2014;311:15361546. doi: 10.1001/jama.2014.2269
- Chu S, Kim S, Lau J, et al. Maternal obesity and risk of stillbirth: a meta-analysis. Am J Obstet Gynecol. 2007;197:223-228. doi: 10.1016/j.ajog.2007.03.027
- Mahomed K, Chan G, Norton M. Obesity and the risk of stillbirth—a reappraisal—a retrospective cohort study. Eur J Obstet Gynecol Reprod Biol. 2020;255:25-28. doi: 10.1016/j. ejogrb. 2020.09.044
- American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, Society for MaternalFetal Medicine. Indications for outpatient antenatal fetal surveillance: ACOG committee opinion, number 828. Obstet Gynecol. 2021;137:e177-e197. doi: 10.1097/ AOG.0000000000004407
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Obesity in pregnancy: ACOG practice bulletin, number 230. Obstet Gynecol. 2021;137:e128-e144. doi: 10.1097/ AOG.0000000000004395
- Denison F, Aedla N, Keag O, et al; Royal College of Obstetricians and Gynaecologists. Care of women with obesity in pregnancy: Green-top Guideline No. 72. BJOG. 2019;126:e62-e106. doi: 10.1111/1471-0528.15386
- Maxwell C, Gaudet L, Cassir G, et al. Guideline No. 391Pregnancy and maternal obesity part 1: pre-conception and prenatal care. J Obstet Gynaecol Can. 2019;41:1623-1640. doi: 10.1016/j.jogc. 2019.03.026
- Yao R, Ananth C, Park B, et al; Perinatal Research Consortium. Obesity and the risk of stillbirth: a population-based cohort study. Am J Obstet Gynecol. 2014;210:e1-e9. doi: 10.1016/j. ajog. 2014.01.044
- Aune D, Saugstad O, Henriksen T, et al. Maternal body mass index and the risk of fetal death, stillbirth, and infant death: a systematic review and meta-analysis. JAMA. 2014;311:15361546. doi: 10.1001/jama.2014.2269
- Chu S, Kim S, Lau J, et al. Maternal obesity and risk of stillbirth: a meta-analysis. Am J Obstet Gynecol. 2007;197:223-228. doi: 10.1016/j.ajog.2007.03.027
- Mahomed K, Chan G, Norton M. Obesity and the risk of stillbirth—a reappraisal—a retrospective cohort study. Eur J Obstet Gynecol Reprod Biol. 2020;255:25-28. doi: 10.1016/j. ejogrb. 2020.09.044
- American College of Obstetricians and Gynecologists’ Committee on Obstetric Practice, Society for MaternalFetal Medicine. Indications for outpatient antenatal fetal surveillance: ACOG committee opinion, number 828. Obstet Gynecol. 2021;137:e177-e197. doi: 10.1097/ AOG.0000000000004407
- American College of Obstetricians and Gynecologists’ Committee on Practice Bulletins–Obstetrics. Obesity in pregnancy: ACOG practice bulletin, number 230. Obstet Gynecol. 2021;137:e128-e144. doi: 10.1097/ AOG.0000000000004395
- Denison F, Aedla N, Keag O, et al; Royal College of Obstetricians and Gynaecologists. Care of women with obesity in pregnancy: Green-top Guideline No. 72. BJOG. 2019;126:e62-e106. doi: 10.1111/1471-0528.15386
- Maxwell C, Gaudet L, Cassir G, et al. Guideline No. 391Pregnancy and maternal obesity part 1: pre-conception and prenatal care. J Obstet Gynaecol Can. 2019;41:1623-1640. doi: 10.1016/j.jogc. 2019.03.026
Cycle timing may reduce hormonal dosage for contraception
Progesterone and estrogen are often used for contraception by preventing ovulation, but the adverse effects associated with large doses of these hormones remain a concern, wrote Brenda Lyn A. Gavina, a PhD candidate at the University of the Philippines Diliman, Quezon City, and colleagues.
In a study published in PLoS Computational Biology, the researchers examined how the timing of hormone administration during a cycle might impact the amount of hormones needed for contraception. Previous research shown that combining hormones can reduce the dosage needed, but the impact of timing on further dose reduction has not been well studied, they said.
The researchers applied optimal control theory in a mathematical model to show the contraceptive effect of estrogen and/or progesterone at different times in the menstrual cycle. The model was based on a normal menstrual cycle with pituitary and ovarian phases. The model assumed that synthesis of luteinizing hormone and follicle-stimulating hormone occurs in the pituitary, that LH and FSH are held in reserve before release into the bloodstream, and that the follicular/luteal mass goes through nine ovarian stages of development. The model also included the activity of ovarian hormones estradiol (E2), progesterone (P4), and inhibin (Inh), in a normal cycle. In the model, LH, FSH, and E2 peaked in the late follicular phase, and P4 and Inh peaked in the luteal phase.
The pituitary model predicted the synthesis, release, and clearance of LH and FSH, and the response of the pituitary to E2, P4, and Inh. The ovarian model predicted the response of E2, P4, and Inh as functions of LH and FSH.
The researchers simulated a constant dose of exogenous progesterone monotherapy and combined exogenous estrogen/progesterone. They determined that a P4 peak of 4.99 ng/mL was taken as the optimum constant dosage for progesterone monotherapy, and for combination estrogen/progesterone.
The researchers then assessed the impact of time on dosage. They found that estrogen administration starting on the first day of a normal cycle preventing FHS from reaching maximum value, and that the low level of FHS in the follicular phase and additional P4 inhibition slowed follicular growth, and use of combination estrogen/progesterone caused similar inhibition at a later follicular stage.
“The combination therapy suggests that time-varying doses of estrogen and progesterone given simultaneously from the start to the end of the 28-day period, only requires a surge in estrogen dose around the 12th day of the cycle (a delayed administration compared to the estrogen monotherapy),” they noted.
With attention to timing, the maximum progesterone levels throughout a menstrual cycle were 4.43 ng/mL, 4.66 ng/mL, and 4.31 ng/mL for estrogen monotherapy, progesterone monotherapy, and combination therapy, respectively. Total doses of the optimal exogenous hormone were 77.76 pg/mL and 48.84 ng/mL for estrogen and progesterone monotherapy, respectively, and 35.58 pg/mL and 21.67 ng/mL for estrogen and progesterone in combination.
The findings were limited by the use of a standard model that does not account for variations in cycle length, the researchers noted. However, the results reflect other studies of hormonal activity, and the model can be used in future studies of the effect of hormones on cycle length, they said.
Overall, the researchers determined that timing dosage with estrogen monotherapy based on their model could provide effective contraception with about 92% of the minimum total constant dosage, while progesterone monotherapy would be effective with approximately 43% of the total constant dose.
Although more work is needed, the current study results may guide clinicians in experimenting with the optimal treatment regimen for anovulation, the researchers said.
“The results presented here give insights on construction of timed devices that give contraception at certain parts of the menstrual cycle,” they concluded.
Model aims to improve women’s control of contraception
“Aside from wanting to contribute to controlling population growth, we aim to empower women more by giving them more control on when to conceive and start motherhood,” and be in control of contraception in a safer way, said lead author Ms. Gavina, in an interview. In addition, studies are showing the noncontraceptive benefits of suppressing ovulation for managing premenstrual syndromes such as breast tenderness and irritability, and for managing diseases such as endometriosis, she said. “Anovulation also lowers the risk of ACL injuries in female athletes,” she added.
Ms. Gavina said that she was surprised primarily by the optimal control result for estrogen monotherapy. “It was surprising that, theoretically, our mathematical model, with the simplifying assumptions, showed that as low as 10% of the total dose in constant administration could achieve contraception as long as the administration of this dosage is perfectly timed, and the timing was also shown in our optimization result,” she said.
“Our model does not capture all factors in contraception, since the reproductive function in women is a very complex multiscale dynamical system highly dependent on both endogenous and exogenous hormones,” Ms. Gavina told this news organization. However, “with the emergence of more data, it can be refined to address other contraception issues. Further, although the results of this study are not directly translatable to the clinical setting, we hope that these results may aid clinicians in identifying the minimum dose and treatment schedule for contraception,” she said.
Future research directions include examining within and between women’s variabilities and adding a pharmacokinetics model to account for the effects of specific drugs, she said. “We also hope to expand or modify the current model to investigate reproductive health concerns in women, such as [polycystic ovary syndrome] and ovarian cysts,” she added.
Ms. Gavina disclosed support from the University of the Philippines Office of International Linkages, a Continuous Operational and Outcomes-based Partnership for Excellence in Research and Academic Training Enhancement grant, and a Commission on Higher Education Faculty Development Program-II scholarship.
Progesterone and estrogen are often used for contraception by preventing ovulation, but the adverse effects associated with large doses of these hormones remain a concern, wrote Brenda Lyn A. Gavina, a PhD candidate at the University of the Philippines Diliman, Quezon City, and colleagues.
In a study published in PLoS Computational Biology, the researchers examined how the timing of hormone administration during a cycle might impact the amount of hormones needed for contraception. Previous research shown that combining hormones can reduce the dosage needed, but the impact of timing on further dose reduction has not been well studied, they said.
The researchers applied optimal control theory in a mathematical model to show the contraceptive effect of estrogen and/or progesterone at different times in the menstrual cycle. The model was based on a normal menstrual cycle with pituitary and ovarian phases. The model assumed that synthesis of luteinizing hormone and follicle-stimulating hormone occurs in the pituitary, that LH and FSH are held in reserve before release into the bloodstream, and that the follicular/luteal mass goes through nine ovarian stages of development. The model also included the activity of ovarian hormones estradiol (E2), progesterone (P4), and inhibin (Inh), in a normal cycle. In the model, LH, FSH, and E2 peaked in the late follicular phase, and P4 and Inh peaked in the luteal phase.
The pituitary model predicted the synthesis, release, and clearance of LH and FSH, and the response of the pituitary to E2, P4, and Inh. The ovarian model predicted the response of E2, P4, and Inh as functions of LH and FSH.
The researchers simulated a constant dose of exogenous progesterone monotherapy and combined exogenous estrogen/progesterone. They determined that a P4 peak of 4.99 ng/mL was taken as the optimum constant dosage for progesterone monotherapy, and for combination estrogen/progesterone.
The researchers then assessed the impact of time on dosage. They found that estrogen administration starting on the first day of a normal cycle preventing FHS from reaching maximum value, and that the low level of FHS in the follicular phase and additional P4 inhibition slowed follicular growth, and use of combination estrogen/progesterone caused similar inhibition at a later follicular stage.
“The combination therapy suggests that time-varying doses of estrogen and progesterone given simultaneously from the start to the end of the 28-day period, only requires a surge in estrogen dose around the 12th day of the cycle (a delayed administration compared to the estrogen monotherapy),” they noted.
With attention to timing, the maximum progesterone levels throughout a menstrual cycle were 4.43 ng/mL, 4.66 ng/mL, and 4.31 ng/mL for estrogen monotherapy, progesterone monotherapy, and combination therapy, respectively. Total doses of the optimal exogenous hormone were 77.76 pg/mL and 48.84 ng/mL for estrogen and progesterone monotherapy, respectively, and 35.58 pg/mL and 21.67 ng/mL for estrogen and progesterone in combination.
The findings were limited by the use of a standard model that does not account for variations in cycle length, the researchers noted. However, the results reflect other studies of hormonal activity, and the model can be used in future studies of the effect of hormones on cycle length, they said.
Overall, the researchers determined that timing dosage with estrogen monotherapy based on their model could provide effective contraception with about 92% of the minimum total constant dosage, while progesterone monotherapy would be effective with approximately 43% of the total constant dose.
Although more work is needed, the current study results may guide clinicians in experimenting with the optimal treatment regimen for anovulation, the researchers said.
“The results presented here give insights on construction of timed devices that give contraception at certain parts of the menstrual cycle,” they concluded.
Model aims to improve women’s control of contraception
“Aside from wanting to contribute to controlling population growth, we aim to empower women more by giving them more control on when to conceive and start motherhood,” and be in control of contraception in a safer way, said lead author Ms. Gavina, in an interview. In addition, studies are showing the noncontraceptive benefits of suppressing ovulation for managing premenstrual syndromes such as breast tenderness and irritability, and for managing diseases such as endometriosis, she said. “Anovulation also lowers the risk of ACL injuries in female athletes,” she added.
Ms. Gavina said that she was surprised primarily by the optimal control result for estrogen monotherapy. “It was surprising that, theoretically, our mathematical model, with the simplifying assumptions, showed that as low as 10% of the total dose in constant administration could achieve contraception as long as the administration of this dosage is perfectly timed, and the timing was also shown in our optimization result,” she said.
“Our model does not capture all factors in contraception, since the reproductive function in women is a very complex multiscale dynamical system highly dependent on both endogenous and exogenous hormones,” Ms. Gavina told this news organization. However, “with the emergence of more data, it can be refined to address other contraception issues. Further, although the results of this study are not directly translatable to the clinical setting, we hope that these results may aid clinicians in identifying the minimum dose and treatment schedule for contraception,” she said.
Future research directions include examining within and between women’s variabilities and adding a pharmacokinetics model to account for the effects of specific drugs, she said. “We also hope to expand or modify the current model to investigate reproductive health concerns in women, such as [polycystic ovary syndrome] and ovarian cysts,” she added.
Ms. Gavina disclosed support from the University of the Philippines Office of International Linkages, a Continuous Operational and Outcomes-based Partnership for Excellence in Research and Academic Training Enhancement grant, and a Commission on Higher Education Faculty Development Program-II scholarship.
Progesterone and estrogen are often used for contraception by preventing ovulation, but the adverse effects associated with large doses of these hormones remain a concern, wrote Brenda Lyn A. Gavina, a PhD candidate at the University of the Philippines Diliman, Quezon City, and colleagues.
In a study published in PLoS Computational Biology, the researchers examined how the timing of hormone administration during a cycle might impact the amount of hormones needed for contraception. Previous research shown that combining hormones can reduce the dosage needed, but the impact of timing on further dose reduction has not been well studied, they said.
The researchers applied optimal control theory in a mathematical model to show the contraceptive effect of estrogen and/or progesterone at different times in the menstrual cycle. The model was based on a normal menstrual cycle with pituitary and ovarian phases. The model assumed that synthesis of luteinizing hormone and follicle-stimulating hormone occurs in the pituitary, that LH and FSH are held in reserve before release into the bloodstream, and that the follicular/luteal mass goes through nine ovarian stages of development. The model also included the activity of ovarian hormones estradiol (E2), progesterone (P4), and inhibin (Inh), in a normal cycle. In the model, LH, FSH, and E2 peaked in the late follicular phase, and P4 and Inh peaked in the luteal phase.
The pituitary model predicted the synthesis, release, and clearance of LH and FSH, and the response of the pituitary to E2, P4, and Inh. The ovarian model predicted the response of E2, P4, and Inh as functions of LH and FSH.
The researchers simulated a constant dose of exogenous progesterone monotherapy and combined exogenous estrogen/progesterone. They determined that a P4 peak of 4.99 ng/mL was taken as the optimum constant dosage for progesterone monotherapy, and for combination estrogen/progesterone.
The researchers then assessed the impact of time on dosage. They found that estrogen administration starting on the first day of a normal cycle preventing FHS from reaching maximum value, and that the low level of FHS in the follicular phase and additional P4 inhibition slowed follicular growth, and use of combination estrogen/progesterone caused similar inhibition at a later follicular stage.
“The combination therapy suggests that time-varying doses of estrogen and progesterone given simultaneously from the start to the end of the 28-day period, only requires a surge in estrogen dose around the 12th day of the cycle (a delayed administration compared to the estrogen monotherapy),” they noted.
With attention to timing, the maximum progesterone levels throughout a menstrual cycle were 4.43 ng/mL, 4.66 ng/mL, and 4.31 ng/mL for estrogen monotherapy, progesterone monotherapy, and combination therapy, respectively. Total doses of the optimal exogenous hormone were 77.76 pg/mL and 48.84 ng/mL for estrogen and progesterone monotherapy, respectively, and 35.58 pg/mL and 21.67 ng/mL for estrogen and progesterone in combination.
The findings were limited by the use of a standard model that does not account for variations in cycle length, the researchers noted. However, the results reflect other studies of hormonal activity, and the model can be used in future studies of the effect of hormones on cycle length, they said.
Overall, the researchers determined that timing dosage with estrogen monotherapy based on their model could provide effective contraception with about 92% of the minimum total constant dosage, while progesterone monotherapy would be effective with approximately 43% of the total constant dose.
Although more work is needed, the current study results may guide clinicians in experimenting with the optimal treatment regimen for anovulation, the researchers said.
“The results presented here give insights on construction of timed devices that give contraception at certain parts of the menstrual cycle,” they concluded.
Model aims to improve women’s control of contraception
“Aside from wanting to contribute to controlling population growth, we aim to empower women more by giving them more control on when to conceive and start motherhood,” and be in control of contraception in a safer way, said lead author Ms. Gavina, in an interview. In addition, studies are showing the noncontraceptive benefits of suppressing ovulation for managing premenstrual syndromes such as breast tenderness and irritability, and for managing diseases such as endometriosis, she said. “Anovulation also lowers the risk of ACL injuries in female athletes,” she added.
Ms. Gavina said that she was surprised primarily by the optimal control result for estrogen monotherapy. “It was surprising that, theoretically, our mathematical model, with the simplifying assumptions, showed that as low as 10% of the total dose in constant administration could achieve contraception as long as the administration of this dosage is perfectly timed, and the timing was also shown in our optimization result,” she said.
“Our model does not capture all factors in contraception, since the reproductive function in women is a very complex multiscale dynamical system highly dependent on both endogenous and exogenous hormones,” Ms. Gavina told this news organization. However, “with the emergence of more data, it can be refined to address other contraception issues. Further, although the results of this study are not directly translatable to the clinical setting, we hope that these results may aid clinicians in identifying the minimum dose and treatment schedule for contraception,” she said.
Future research directions include examining within and between women’s variabilities and adding a pharmacokinetics model to account for the effects of specific drugs, she said. “We also hope to expand or modify the current model to investigate reproductive health concerns in women, such as [polycystic ovary syndrome] and ovarian cysts,” she added.
Ms. Gavina disclosed support from the University of the Philippines Office of International Linkages, a Continuous Operational and Outcomes-based Partnership for Excellence in Research and Academic Training Enhancement grant, and a Commission on Higher Education Faculty Development Program-II scholarship.
FROM PLOS COMPUTATIONAL BIOLOGY
Guidelines for assessing cancer risk may need updating
The authors of the clinical trial suggest that these guidelines may need to be revised.
Individuals with hereditary breast and ovarian cancer (HBOC) have an 80% lifetime risk of breast cancer and are at greater risk of ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Those with Lynch syndrome (LS) have an 80% lifetime risk of colorectal cancer, a 60% lifetime risk of endometrial cancer, and heightened risk of upper gastrointestinal, urinary tract, skin, and other tumors, said study coauthor N. Jewel Samadder, MD in a statement.
The National Cancer Control Network has guidelines for determining family risk for colorectal cancer and breast, ovarian, and pancreatic cancer to identify individuals who should be screened for LS and HBOC, but these rely on personal and family health histories.
“These criteria were created at a time when genetic testing was cost prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing. However, [LS and HBOC] are poorly identified in current practice, and many patients are not aware of their cancer risk,” said Dr. Samadder, professor of medicine and coleader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center, Phoenix, in the statement.
Whole-exome sequencing covers only protein-coding regions of the genome, which is less than 2% of the total genome but includes more than 85% of known disease-related genetic variants, according to Emily Gay, who presented the trial results (Abstract 5768) on April 18 at the annual meeting of the American Association for Cancer Research.
“In recent years, the cost of whole-exome sequencing has been rapidly decreasing, allowing us to complete this test on saliva samples from thousands, if not tens of thousands of patients covering large populations and large health systems,” said Ms. Gay, a genetic counseling graduate student at the University of Arizona, during her presentation.
She described results from the TAPESTRY clinical trial, with 44,306 participants from Mayo Clinic centers in Arizona, Florida, and Minnesota, who were identified as definitely or likely to be harboring pathogenic mutations and consented to whole-exome sequencing from saliva samples. They used electronic health records to determine whether patients would satisfy the testing criteria from NCCN guidelines.
The researchers identified 1.24% of participants to be carriers of HBOC or LS. Of the HBOC carriers, 62.8% were female, and of the LS carriers, 62.6% were female. The percentages of HBOC and LS carriers who were White were 88.6 and 94.5, respectively. The median age of both groups was 57 years. Of HBOC carriers, 47.3% had personal histories of cancers; for LS carries, the percentage was 44.2.
Of HBOC carriers, 49.1% had been previously unaware of their genetic condition, while an even higher percentage of patients with LS – 59.3% – fell into that category. Thirty-two percent of those with HBOC and 56.2% of those with LS would not have qualified for screening using the relevant NCCN guidelines.
“Most strikingly,” 63.8% of individuals with mutations in the MSH6 gene and 83.7% of those mutations in the PMS2 gene would not have met NCCN criteria, Ms. Gay said.
Having a cancer type not known to be related to a genetic syndrome was a reason for 58.6% of individuals failing to meet NCCN guidelines, while 60.5% did not meet the guidelines because of an insufficient number of relatives known to have a history of cancer, and 63.3% did not because they had no personal history of cancer. Among individuals with a pathogenic mutation who met NCCN criteria, 34% were not aware of their condition.
“This suggests that the NCCN guidelines are underutilized in clinical practice, potentially due to the busy schedule of clinicians or because the complexity of using these criteria,” said Ms. Gay.
The numbers were even more striking among minorities: “There is additional data analysis and research needed in this area, but based on our preliminary findings, we saw that nearly 50% of the individuals who are [part of an underrepresented minority group] did not meet criteria, compared with 32% of the white cohort,” said Ms. Gay.
Asked what new NCCN guidelines should be, Ms. Gay replied: “I think maybe limiting the number of relatives that you have to have with a certain type of cancer, especially as we see families get smaller and smaller, especially in the United States – that family data isn’t necessarily available or as useful. And then also, I think, incorporating in the size of a family into the calculation, so more of maybe a point-based system like we see with other genetic conditions rather than a ‘yes you meet or no, you don’t.’ More of a range to say ‘you fall on the low-risk, medium-risk, or high-risk stage,’” said Ms. Gay.
During the Q&A period, session cochair Andrew Godwin, PhD, who is a professor of molecular oncology and pathology at University of Kansas Medical Center, Kansas City, said he wondered if whole-exome sequencing was capable of picking up cancer risk mutations that standard targeted tests don’t look for.
Dr. Samadder, who was in the audience, answered the question, saying that targeted tests are actually better at picking up some types of mutations like intronic mutations, single-nucleotide polymorphisms, and deletions.
“There are some limitations to whole-exome sequencing. Our estimate here of 1.2% [of participants carrying HBOC or LS mutations] is probably an underestimate. There are additional variants that exome sequencing probably doesn’t pick up easily or as well. That’s why we qualify that exome sequencing is a screening test, not a diagnostic,” he continued.
Ms. Gay and Dr. Samadder have no relevant financial disclosures. Dr. Godwin has financial relationships with Clara Biotech, VITRAC Therapeutics, and Sinochips Diagnostics.
The authors of the clinical trial suggest that these guidelines may need to be revised.
Individuals with hereditary breast and ovarian cancer (HBOC) have an 80% lifetime risk of breast cancer and are at greater risk of ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Those with Lynch syndrome (LS) have an 80% lifetime risk of colorectal cancer, a 60% lifetime risk of endometrial cancer, and heightened risk of upper gastrointestinal, urinary tract, skin, and other tumors, said study coauthor N. Jewel Samadder, MD in a statement.
The National Cancer Control Network has guidelines for determining family risk for colorectal cancer and breast, ovarian, and pancreatic cancer to identify individuals who should be screened for LS and HBOC, but these rely on personal and family health histories.
“These criteria were created at a time when genetic testing was cost prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing. However, [LS and HBOC] are poorly identified in current practice, and many patients are not aware of their cancer risk,” said Dr. Samadder, professor of medicine and coleader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center, Phoenix, in the statement.
Whole-exome sequencing covers only protein-coding regions of the genome, which is less than 2% of the total genome but includes more than 85% of known disease-related genetic variants, according to Emily Gay, who presented the trial results (Abstract 5768) on April 18 at the annual meeting of the American Association for Cancer Research.
“In recent years, the cost of whole-exome sequencing has been rapidly decreasing, allowing us to complete this test on saliva samples from thousands, if not tens of thousands of patients covering large populations and large health systems,” said Ms. Gay, a genetic counseling graduate student at the University of Arizona, during her presentation.
She described results from the TAPESTRY clinical trial, with 44,306 participants from Mayo Clinic centers in Arizona, Florida, and Minnesota, who were identified as definitely or likely to be harboring pathogenic mutations and consented to whole-exome sequencing from saliva samples. They used electronic health records to determine whether patients would satisfy the testing criteria from NCCN guidelines.
The researchers identified 1.24% of participants to be carriers of HBOC or LS. Of the HBOC carriers, 62.8% were female, and of the LS carriers, 62.6% were female. The percentages of HBOC and LS carriers who were White were 88.6 and 94.5, respectively. The median age of both groups was 57 years. Of HBOC carriers, 47.3% had personal histories of cancers; for LS carries, the percentage was 44.2.
Of HBOC carriers, 49.1% had been previously unaware of their genetic condition, while an even higher percentage of patients with LS – 59.3% – fell into that category. Thirty-two percent of those with HBOC and 56.2% of those with LS would not have qualified for screening using the relevant NCCN guidelines.
“Most strikingly,” 63.8% of individuals with mutations in the MSH6 gene and 83.7% of those mutations in the PMS2 gene would not have met NCCN criteria, Ms. Gay said.
Having a cancer type not known to be related to a genetic syndrome was a reason for 58.6% of individuals failing to meet NCCN guidelines, while 60.5% did not meet the guidelines because of an insufficient number of relatives known to have a history of cancer, and 63.3% did not because they had no personal history of cancer. Among individuals with a pathogenic mutation who met NCCN criteria, 34% were not aware of their condition.
“This suggests that the NCCN guidelines are underutilized in clinical practice, potentially due to the busy schedule of clinicians or because the complexity of using these criteria,” said Ms. Gay.
The numbers were even more striking among minorities: “There is additional data analysis and research needed in this area, but based on our preliminary findings, we saw that nearly 50% of the individuals who are [part of an underrepresented minority group] did not meet criteria, compared with 32% of the white cohort,” said Ms. Gay.
Asked what new NCCN guidelines should be, Ms. Gay replied: “I think maybe limiting the number of relatives that you have to have with a certain type of cancer, especially as we see families get smaller and smaller, especially in the United States – that family data isn’t necessarily available or as useful. And then also, I think, incorporating in the size of a family into the calculation, so more of maybe a point-based system like we see with other genetic conditions rather than a ‘yes you meet or no, you don’t.’ More of a range to say ‘you fall on the low-risk, medium-risk, or high-risk stage,’” said Ms. Gay.
During the Q&A period, session cochair Andrew Godwin, PhD, who is a professor of molecular oncology and pathology at University of Kansas Medical Center, Kansas City, said he wondered if whole-exome sequencing was capable of picking up cancer risk mutations that standard targeted tests don’t look for.
Dr. Samadder, who was in the audience, answered the question, saying that targeted tests are actually better at picking up some types of mutations like intronic mutations, single-nucleotide polymorphisms, and deletions.
“There are some limitations to whole-exome sequencing. Our estimate here of 1.2% [of participants carrying HBOC or LS mutations] is probably an underestimate. There are additional variants that exome sequencing probably doesn’t pick up easily or as well. That’s why we qualify that exome sequencing is a screening test, not a diagnostic,” he continued.
Ms. Gay and Dr. Samadder have no relevant financial disclosures. Dr. Godwin has financial relationships with Clara Biotech, VITRAC Therapeutics, and Sinochips Diagnostics.
The authors of the clinical trial suggest that these guidelines may need to be revised.
Individuals with hereditary breast and ovarian cancer (HBOC) have an 80% lifetime risk of breast cancer and are at greater risk of ovarian cancer, pancreatic cancer, prostate cancer, and melanoma. Those with Lynch syndrome (LS) have an 80% lifetime risk of colorectal cancer, a 60% lifetime risk of endometrial cancer, and heightened risk of upper gastrointestinal, urinary tract, skin, and other tumors, said study coauthor N. Jewel Samadder, MD in a statement.
The National Cancer Control Network has guidelines for determining family risk for colorectal cancer and breast, ovarian, and pancreatic cancer to identify individuals who should be screened for LS and HBOC, but these rely on personal and family health histories.
“These criteria were created at a time when genetic testing was cost prohibitive and thus aimed to identify those at the greatest chance of being a mutation carrier in the absence of population-wide whole-exome sequencing. However, [LS and HBOC] are poorly identified in current practice, and many patients are not aware of their cancer risk,” said Dr. Samadder, professor of medicine and coleader of the precision oncology program at the Mayo Clinic Comprehensive Cancer Center, Phoenix, in the statement.
Whole-exome sequencing covers only protein-coding regions of the genome, which is less than 2% of the total genome but includes more than 85% of known disease-related genetic variants, according to Emily Gay, who presented the trial results (Abstract 5768) on April 18 at the annual meeting of the American Association for Cancer Research.
“In recent years, the cost of whole-exome sequencing has been rapidly decreasing, allowing us to complete this test on saliva samples from thousands, if not tens of thousands of patients covering large populations and large health systems,” said Ms. Gay, a genetic counseling graduate student at the University of Arizona, during her presentation.
She described results from the TAPESTRY clinical trial, with 44,306 participants from Mayo Clinic centers in Arizona, Florida, and Minnesota, who were identified as definitely or likely to be harboring pathogenic mutations and consented to whole-exome sequencing from saliva samples. They used electronic health records to determine whether patients would satisfy the testing criteria from NCCN guidelines.
The researchers identified 1.24% of participants to be carriers of HBOC or LS. Of the HBOC carriers, 62.8% were female, and of the LS carriers, 62.6% were female. The percentages of HBOC and LS carriers who were White were 88.6 and 94.5, respectively. The median age of both groups was 57 years. Of HBOC carriers, 47.3% had personal histories of cancers; for LS carries, the percentage was 44.2.
Of HBOC carriers, 49.1% had been previously unaware of their genetic condition, while an even higher percentage of patients with LS – 59.3% – fell into that category. Thirty-two percent of those with HBOC and 56.2% of those with LS would not have qualified for screening using the relevant NCCN guidelines.
“Most strikingly,” 63.8% of individuals with mutations in the MSH6 gene and 83.7% of those mutations in the PMS2 gene would not have met NCCN criteria, Ms. Gay said.
Having a cancer type not known to be related to a genetic syndrome was a reason for 58.6% of individuals failing to meet NCCN guidelines, while 60.5% did not meet the guidelines because of an insufficient number of relatives known to have a history of cancer, and 63.3% did not because they had no personal history of cancer. Among individuals with a pathogenic mutation who met NCCN criteria, 34% were not aware of their condition.
“This suggests that the NCCN guidelines are underutilized in clinical practice, potentially due to the busy schedule of clinicians or because the complexity of using these criteria,” said Ms. Gay.
The numbers were even more striking among minorities: “There is additional data analysis and research needed in this area, but based on our preliminary findings, we saw that nearly 50% of the individuals who are [part of an underrepresented minority group] did not meet criteria, compared with 32% of the white cohort,” said Ms. Gay.
Asked what new NCCN guidelines should be, Ms. Gay replied: “I think maybe limiting the number of relatives that you have to have with a certain type of cancer, especially as we see families get smaller and smaller, especially in the United States – that family data isn’t necessarily available or as useful. And then also, I think, incorporating in the size of a family into the calculation, so more of maybe a point-based system like we see with other genetic conditions rather than a ‘yes you meet or no, you don’t.’ More of a range to say ‘you fall on the low-risk, medium-risk, or high-risk stage,’” said Ms. Gay.
During the Q&A period, session cochair Andrew Godwin, PhD, who is a professor of molecular oncology and pathology at University of Kansas Medical Center, Kansas City, said he wondered if whole-exome sequencing was capable of picking up cancer risk mutations that standard targeted tests don’t look for.
Dr. Samadder, who was in the audience, answered the question, saying that targeted tests are actually better at picking up some types of mutations like intronic mutations, single-nucleotide polymorphisms, and deletions.
“There are some limitations to whole-exome sequencing. Our estimate here of 1.2% [of participants carrying HBOC or LS mutations] is probably an underestimate. There are additional variants that exome sequencing probably doesn’t pick up easily or as well. That’s why we qualify that exome sequencing is a screening test, not a diagnostic,” he continued.
Ms. Gay and Dr. Samadder have no relevant financial disclosures. Dr. Godwin has financial relationships with Clara Biotech, VITRAC Therapeutics, and Sinochips Diagnostics.
FROM AACR 2023
Proposed Medicare bill would raise docs’ pay with inflation
Introduced by four physician U.S. House representatives, HR 2474 would link Medicare fee schedule updates to the Medicare Economic Index, a measure of inflation related to physicians’ practice costs and wages.
That’s a long-sought goal of the American Medical Association, which is leading 120 state medical societies and medical specialty groups in championing the bill.
The legislation is essential to enabling physician practices to better absorb payment distributions triggered by budget neutrality rules, performance adjustments, and periods of high inflation, the groups wrote in a joint letter sent to the bill’s sponsors. The sponsors say they hope the legislation will improve access to care, as low reimbursements cause some physicians to limit their number of Medicare patients.
Physicians groups for years have urged federal lawmakers to scrap short-term fixes staving off Medicare pay cuts in favor of permanent reforms. Unlike nearly all other Medicare clinicians including hospitals, physicians’ Medicare payment updates aren’t currently tied to inflation.
Adjusted for inflation, Medicare payments to physicians have declined 26% between 2001 and 2023, including a 2% payment reduction in 2023, according to the AMA. Small and rural physician practices have been disproportionately affected by these reductions, as have doctors treating low-income or uninsured patients, the AMA said.
Last month, an influential federal advisory panel recommended permanently tying Medicare physician pay increases to inflation. Clinicians’ cost of providing services, measured by the Medicare Economic Index, rose by 2.6% in 2021 and are estimated to have risen 4.7% in 2022, significantly more than in recent years, the Medicare Payment Advisory Commission said.
A version of this article originally appeared on Medscape.com.
Introduced by four physician U.S. House representatives, HR 2474 would link Medicare fee schedule updates to the Medicare Economic Index, a measure of inflation related to physicians’ practice costs and wages.
That’s a long-sought goal of the American Medical Association, which is leading 120 state medical societies and medical specialty groups in championing the bill.
The legislation is essential to enabling physician practices to better absorb payment distributions triggered by budget neutrality rules, performance adjustments, and periods of high inflation, the groups wrote in a joint letter sent to the bill’s sponsors. The sponsors say they hope the legislation will improve access to care, as low reimbursements cause some physicians to limit their number of Medicare patients.
Physicians groups for years have urged federal lawmakers to scrap short-term fixes staving off Medicare pay cuts in favor of permanent reforms. Unlike nearly all other Medicare clinicians including hospitals, physicians’ Medicare payment updates aren’t currently tied to inflation.
Adjusted for inflation, Medicare payments to physicians have declined 26% between 2001 and 2023, including a 2% payment reduction in 2023, according to the AMA. Small and rural physician practices have been disproportionately affected by these reductions, as have doctors treating low-income or uninsured patients, the AMA said.
Last month, an influential federal advisory panel recommended permanently tying Medicare physician pay increases to inflation. Clinicians’ cost of providing services, measured by the Medicare Economic Index, rose by 2.6% in 2021 and are estimated to have risen 4.7% in 2022, significantly more than in recent years, the Medicare Payment Advisory Commission said.
A version of this article originally appeared on Medscape.com.
Introduced by four physician U.S. House representatives, HR 2474 would link Medicare fee schedule updates to the Medicare Economic Index, a measure of inflation related to physicians’ practice costs and wages.
That’s a long-sought goal of the American Medical Association, which is leading 120 state medical societies and medical specialty groups in championing the bill.
The legislation is essential to enabling physician practices to better absorb payment distributions triggered by budget neutrality rules, performance adjustments, and periods of high inflation, the groups wrote in a joint letter sent to the bill’s sponsors. The sponsors say they hope the legislation will improve access to care, as low reimbursements cause some physicians to limit their number of Medicare patients.
Physicians groups for years have urged federal lawmakers to scrap short-term fixes staving off Medicare pay cuts in favor of permanent reforms. Unlike nearly all other Medicare clinicians including hospitals, physicians’ Medicare payment updates aren’t currently tied to inflation.
Adjusted for inflation, Medicare payments to physicians have declined 26% between 2001 and 2023, including a 2% payment reduction in 2023, according to the AMA. Small and rural physician practices have been disproportionately affected by these reductions, as have doctors treating low-income or uninsured patients, the AMA said.
Last month, an influential federal advisory panel recommended permanently tying Medicare physician pay increases to inflation. Clinicians’ cost of providing services, measured by the Medicare Economic Index, rose by 2.6% in 2021 and are estimated to have risen 4.7% in 2022, significantly more than in recent years, the Medicare Payment Advisory Commission said.
A version of this article originally appeared on Medscape.com.
Surgeons, intensivists earn more than do colleagues from private insurance
General and orthopedic surgeons and intensivists earn the highest net reimbursements from private U.S. insurers, a new report estimates.
On average in 2021, they were paid $5.8 million, $4.9 million, and $3.3 million, respectively, according to figures compiled by AMN Healthcare, a Dallas-based health staffing company.
None of 15 other physician specialties topped $3 million in net reimbursement on average, and three – dermatology, pediatrics, and family medicine – didn’t reach $1 million.
The report doesn’t include data about reimbursement from Medicare and Medicaid, and its numbers assume that 50% of insurance claims are denied. Denial rates differ from practice to practice.
Still, the findings offer a “benchmark tool” to help clinicians understand how they rank against their peers, Linda Murphy, president of AMN Healthcare’s Revenue Cycle Solutions division, said in an interview.
This is the first year that the company has calculated physician reimbursement levels by using claim and clearinghouse data, Ms. Murphy said. Previously, a division of the firm compiled data by surveying chief financial officers from hospitals.
The report’s estimate that insurers deny 50% of claims is “conservative,” Ms. Murphy said. Miscoding is a significant factor behind that number.
The estimated 2021 net private insurance reimbursements by specialty for direct services, assuming a 50% denial rate:
- Anesthesiology: $1,665,510
- Cardiology: $1,703,013
- Critical Care (intensivist): $3,338,656
- Dermatology: $729,107
- Family medicine: $697,094
- Gastroenterology: $2,765,110
- Internal medicine: $1,297,200
- Neurology: $1,390,181
- Obstetrician/gynecology: $1,880,888
- Otolaryngology: $2,095,277
- Pediatrics: $661,552
- Psychiatry: $1,348,730
- Pulmonology: $1,561,617
- Radiology: $1,015,750
- Rheumatology: $1,705,140
- General surgery: $5,834,508
- Orthopedic surgery: $4,904,757
- Urology: $2,943,381
Among 18 physician specialties overall, the report estimated that the average net reimbursement in 2021 was $1.9 million.
The report also estimated that the net reimbursement amounts at $875,140 for certified registered nurse anesthetists and $388,696 for nurse practitioners.
Surprisingly, Ms. Murphy said, there’s “a really large swing” among reimbursement levels for individual specialties. The quartile of cardiologists with the lowest level of reimbursement, for example, submitted $2.1 million in claims in 2021, netting about $1 million at a 50% denial rate versus the $7.3 million made by those in the highest quartile, netting about $3.6 million.
The gap seems to be due to regional variations, she said, adding that a rural cardiologist will have different billing practices than does one practicing in New York City.
The quartile of general surgeons with the highest reimbursement levels billed for $21.1 million on average in 2021, making about $10.5 million at a 50% denial rate. The lowest quartile billed for $5.5 million, making about $2.7 million at a 50% denial rate.
The report noted that primary care physicians – that is, family medicine, internal medicine, and pediatrics specialists – have much lower levels of reimbursement, compared with most other specialties. But the work of primary care physicians “may lead to considerable ‘downstream revenue’ through the hospital admissions, tests and treatment they order.”
A previous analysis by a division of AMN Healthcare found that primary care physicians, on average, generate $2,113,273 a year in net annual revenue for their affiliated hospitals, nearing the $2,446,429 in net annual hospital revenue generated by specialists.
AMN Healthcare is preparing another report that will examine Medicare reimbursements, Ms. Murphy said. According to the new report, payments by nonprivate insurers amount to about one-third of the total amount of reimbursement by commercial insurers.
A version of this article originally appeared on Medscape.com.
General and orthopedic surgeons and intensivists earn the highest net reimbursements from private U.S. insurers, a new report estimates.
On average in 2021, they were paid $5.8 million, $4.9 million, and $3.3 million, respectively, according to figures compiled by AMN Healthcare, a Dallas-based health staffing company.
None of 15 other physician specialties topped $3 million in net reimbursement on average, and three – dermatology, pediatrics, and family medicine – didn’t reach $1 million.
The report doesn’t include data about reimbursement from Medicare and Medicaid, and its numbers assume that 50% of insurance claims are denied. Denial rates differ from practice to practice.
Still, the findings offer a “benchmark tool” to help clinicians understand how they rank against their peers, Linda Murphy, president of AMN Healthcare’s Revenue Cycle Solutions division, said in an interview.
This is the first year that the company has calculated physician reimbursement levels by using claim and clearinghouse data, Ms. Murphy said. Previously, a division of the firm compiled data by surveying chief financial officers from hospitals.
The report’s estimate that insurers deny 50% of claims is “conservative,” Ms. Murphy said. Miscoding is a significant factor behind that number.
The estimated 2021 net private insurance reimbursements by specialty for direct services, assuming a 50% denial rate:
- Anesthesiology: $1,665,510
- Cardiology: $1,703,013
- Critical Care (intensivist): $3,338,656
- Dermatology: $729,107
- Family medicine: $697,094
- Gastroenterology: $2,765,110
- Internal medicine: $1,297,200
- Neurology: $1,390,181
- Obstetrician/gynecology: $1,880,888
- Otolaryngology: $2,095,277
- Pediatrics: $661,552
- Psychiatry: $1,348,730
- Pulmonology: $1,561,617
- Radiology: $1,015,750
- Rheumatology: $1,705,140
- General surgery: $5,834,508
- Orthopedic surgery: $4,904,757
- Urology: $2,943,381
Among 18 physician specialties overall, the report estimated that the average net reimbursement in 2021 was $1.9 million.
The report also estimated that the net reimbursement amounts at $875,140 for certified registered nurse anesthetists and $388,696 for nurse practitioners.
Surprisingly, Ms. Murphy said, there’s “a really large swing” among reimbursement levels for individual specialties. The quartile of cardiologists with the lowest level of reimbursement, for example, submitted $2.1 million in claims in 2021, netting about $1 million at a 50% denial rate versus the $7.3 million made by those in the highest quartile, netting about $3.6 million.
The gap seems to be due to regional variations, she said, adding that a rural cardiologist will have different billing practices than does one practicing in New York City.
The quartile of general surgeons with the highest reimbursement levels billed for $21.1 million on average in 2021, making about $10.5 million at a 50% denial rate. The lowest quartile billed for $5.5 million, making about $2.7 million at a 50% denial rate.
The report noted that primary care physicians – that is, family medicine, internal medicine, and pediatrics specialists – have much lower levels of reimbursement, compared with most other specialties. But the work of primary care physicians “may lead to considerable ‘downstream revenue’ through the hospital admissions, tests and treatment they order.”
A previous analysis by a division of AMN Healthcare found that primary care physicians, on average, generate $2,113,273 a year in net annual revenue for their affiliated hospitals, nearing the $2,446,429 in net annual hospital revenue generated by specialists.
AMN Healthcare is preparing another report that will examine Medicare reimbursements, Ms. Murphy said. According to the new report, payments by nonprivate insurers amount to about one-third of the total amount of reimbursement by commercial insurers.
A version of this article originally appeared on Medscape.com.
General and orthopedic surgeons and intensivists earn the highest net reimbursements from private U.S. insurers, a new report estimates.
On average in 2021, they were paid $5.8 million, $4.9 million, and $3.3 million, respectively, according to figures compiled by AMN Healthcare, a Dallas-based health staffing company.
None of 15 other physician specialties topped $3 million in net reimbursement on average, and three – dermatology, pediatrics, and family medicine – didn’t reach $1 million.
The report doesn’t include data about reimbursement from Medicare and Medicaid, and its numbers assume that 50% of insurance claims are denied. Denial rates differ from practice to practice.
Still, the findings offer a “benchmark tool” to help clinicians understand how they rank against their peers, Linda Murphy, president of AMN Healthcare’s Revenue Cycle Solutions division, said in an interview.
This is the first year that the company has calculated physician reimbursement levels by using claim and clearinghouse data, Ms. Murphy said. Previously, a division of the firm compiled data by surveying chief financial officers from hospitals.
The report’s estimate that insurers deny 50% of claims is “conservative,” Ms. Murphy said. Miscoding is a significant factor behind that number.
The estimated 2021 net private insurance reimbursements by specialty for direct services, assuming a 50% denial rate:
- Anesthesiology: $1,665,510
- Cardiology: $1,703,013
- Critical Care (intensivist): $3,338,656
- Dermatology: $729,107
- Family medicine: $697,094
- Gastroenterology: $2,765,110
- Internal medicine: $1,297,200
- Neurology: $1,390,181
- Obstetrician/gynecology: $1,880,888
- Otolaryngology: $2,095,277
- Pediatrics: $661,552
- Psychiatry: $1,348,730
- Pulmonology: $1,561,617
- Radiology: $1,015,750
- Rheumatology: $1,705,140
- General surgery: $5,834,508
- Orthopedic surgery: $4,904,757
- Urology: $2,943,381
Among 18 physician specialties overall, the report estimated that the average net reimbursement in 2021 was $1.9 million.
The report also estimated that the net reimbursement amounts at $875,140 for certified registered nurse anesthetists and $388,696 for nurse practitioners.
Surprisingly, Ms. Murphy said, there’s “a really large swing” among reimbursement levels for individual specialties. The quartile of cardiologists with the lowest level of reimbursement, for example, submitted $2.1 million in claims in 2021, netting about $1 million at a 50% denial rate versus the $7.3 million made by those in the highest quartile, netting about $3.6 million.
The gap seems to be due to regional variations, she said, adding that a rural cardiologist will have different billing practices than does one practicing in New York City.
The quartile of general surgeons with the highest reimbursement levels billed for $21.1 million on average in 2021, making about $10.5 million at a 50% denial rate. The lowest quartile billed for $5.5 million, making about $2.7 million at a 50% denial rate.
The report noted that primary care physicians – that is, family medicine, internal medicine, and pediatrics specialists – have much lower levels of reimbursement, compared with most other specialties. But the work of primary care physicians “may lead to considerable ‘downstream revenue’ through the hospital admissions, tests and treatment they order.”
A previous analysis by a division of AMN Healthcare found that primary care physicians, on average, generate $2,113,273 a year in net annual revenue for their affiliated hospitals, nearing the $2,446,429 in net annual hospital revenue generated by specialists.
AMN Healthcare is preparing another report that will examine Medicare reimbursements, Ms. Murphy said. According to the new report, payments by nonprivate insurers amount to about one-third of the total amount of reimbursement by commercial insurers.
A version of this article originally appeared on Medscape.com.
Long COVID mobile monitoring study hunts for answers
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
A new federal research project aims to answer lingering questions about long COVID using mobile monitoring devices to help track the condition.
The federally funded RECOVER Initiative expects to give out 10,000 sensors to people with long COVID to collect data in real time.
The hope is that researchers will be able to provide doctors and patients with a wealth of information to address gaps in knowledge about long COVID.
The project takes advantage of the approach other researchers have used to track patients’ health data on heart rate, exercise, and more using mobile monitoring devices such as Fitbits, smartwatches, and other remote sensors.
Researchers believe the initiative could be particularly useful for people with long COVID – whose symptoms come and go. They can use a wristband sensor to passively collect data in real time.
For a condition defined by its symptoms, that kind of data promises to be useful, experts said.
But not everyone has room in their budget for a smartwatch or a fitness tracker. Until recently, most clinical trials were BYOD: Bring your own device. At a time when researchers are trying to make sure that clinical trials reflect the diversity of the population, that leaves a lot of people out.
So, researchers are starting to supply subjects with their own monitors. The RECOVER Initiative expects to give out 10,000 sensors to people who are eligible based on race/ethnicity, income, and other demographic factors (rural residents for example). After 2 months, all people in the RECOVER study over the age of 13 will be eligible for the sensors.
The federal program builds on earlier research at places like The Scripps Institute, a center of research into remote monitoring. The institute supplied 7,000 monitors to people in an arm of the All of Us study, a 5-year-old multisite cohort that aims to collect medical information from 1 million people.
The devices went to people who have been historically underrepresented in biomedical research, said Scripps researchers, who plan to give out more this year.
In March of 2023, Scripps researchers published a study on the tracking data that found a significant post-COVID-19 drop in physical activity. But the data are incomplete because many people can’t always afford these devices. Most of the people in the study were “White, young, and active,” they wrote.
Researchers at an All of Us site at Vanderbilt University, which also used a BYOD approach, realized that they produced biased results. They reported their findings at the Pacific Symposium on Biocomputing in January.
“[The] majority of participants who provided Fitbit data reported being White and employed for wages,” they said. “However, these data represent participants who had their own Fitbit devices and consented to share EHR [electronic health record] data.”
Their solution: The program has begun providing Fitbit devices to all study participants who do not own one or cannot afford one.
Now, the web page for the All of Us study asks visitors to “Learn about the All of Us WEAR study. You could get a Fitbit at no cost! … As a part of the WEAR Study, you could receive a new Fitbit to wear at no cost to you. All of Us will be able to get the data the Fitbit collects. This data may help us understand how behavior impacts health.”
Jennifer Radin, PhD, an epidemiologist at Scripps Research Translational Institute, is heading up the DETECT study, which is a remote monitoring research project that has enrolled over 40,000 people who have their own sensors – be it a smartwatch or Fitbit. She was looking at remote monitoring for disease before COVID emerged.
Dr. Radin said she began researching remote sensing after working in public health and dealing with outdated data collection systems.
“They typically rely on case reports that are recorded by pen and paper and faxed or mailed in,” she said. “Then, they have to be entered into a database. “
In addition to offering objective data on a subject’s physical response to the infection, she said, the data collection can be long-term and continuous.
DETECT collects data on resting heart rate, which is unique to every person, and activity levels. Both measures are meaningful for those with long COVID. Her research found differences in sleep, heart rate, and activity between those with COVID and those without.
Joseph Kvedar, MD, is a Harvard Medical School researcher and the editor of NPJ Digital Medicine. He’s been studying digital health systems and called clinical research a “beachhead” for the use of data from monitors. But he also said problems remain that need to be worked out. The quality of the devices and their Bluetooth connections are better. But different devices measure different things, and a counted step can vary from person to person, he said. And the problems of the early days of electronic health records have not been fully resolved.
“We haven’t gotten to this universal language to connect all these things and make them relevant,” he said.
The All of Us researchers are working with the RECOVER project to address some of those issues. Usually not focused on a single condition, the All of Us researchers are testing a machine-learning approach for identifying long COVID.
A version of this article originally appeared on WebMD.com.
Evaluating patients with breast concerns: Lump, pain, and mastitis
The vast majority of symptomatic breast conditions are benign, with the most common symptoms being palpable mass and breast pain. Clinicians, including primary care clinicians and gynecologists, play a crucial role by performing the initial assessment and subsequent therapies and referrals and serve as the mediator between the specialists and by being the patient’s spokesperson. It is therefore important for clinicians to be aware of the various possible causes of these breast symptoms, to know which imaging tests to order, and also to understand the indications for biopsies and surgical referral.
Common types of breast lumps: Imaging workup and management
Accounting for 8% of women who present with breast symptoms, breast lump is the second most common symptom after breast pain.1 The positive likelihood ratio of finding breast cancer is highest among women with breast lumps compared with any other breast symptoms. Therefore, anxiety is related to this symptom, and a thorough evaluation is recommended.1 Cysts, fibroadenoma, and fat necrosis are 3 common benign causes of breast lumps.2
In this section, we review clinical presentation, imaging workup, and management strategies for common types of breast lumps.
CASE 1 Woman with tender breast lump
A 45-year-old woman presents with a breast lump of 6 months’ duration that is associated with a change in size with the menstrual cycle and pain. Clinical examination reveals a 4 x 4.5–cm mass in the right breast in the retroareolar region, which is smooth with some tenderness on palpation.
Breast cyst
According to the American College of Radiology appropriateness criteria for an adult woman 40 years of age or older who presents with a palpable breast mass, the initial imaging study is diagnostic mammography with or without digital tomosynthesis, usually followed by a directed ultrasound. If the mammogram is suspicious or highly suggestive of malignancy, or in cases where the mammogram does not show an abnormality, the next recommended step is breast ultrasonography. Any suspicious findings on ultrasound or mammogram should be followed by an image guided biopsy. Ultrasonography also may be appropriate if the mammogram findings are benign or probably benign.
For an adult woman younger than age 30 who presents with a palpable breast mass, breast ultrasonography is the appropriate initial imaging study. If the ultrasound is suspicious or highly suggestive of malignancy, then performing diagnostic mammography with or without digital tomosynthesis or ultrasound-guided core needle biopsy of the mass are both considered appropriate. However, no further imaging is recommended if the ultrasound is benign, probably benign, or negative. Breast ultrasonography or mammography is appropriate as the initial imaging test for adult women aged 30 to 39 years who present with a palpable breast mass.3,4
Approximately 50% of women after age 30 may develop fibrocystic breast disease, and 20% of them can present with pain or lump due to a macrocysts. Simple cysts must be distinguished from complex cysts with the help of ultrasound as the latter are associated with 23% to 31% increased risk of malignancy.
In this 45-year-old patient, the initial mammogram demonstrated a circumscribed mass underneath the area of palpable concern (FIGURE 1a, 1b). Targeted breast ultrasonography was performed for further assessment, which depicted the mass as a benign simple cyst (FIGURE 1c).
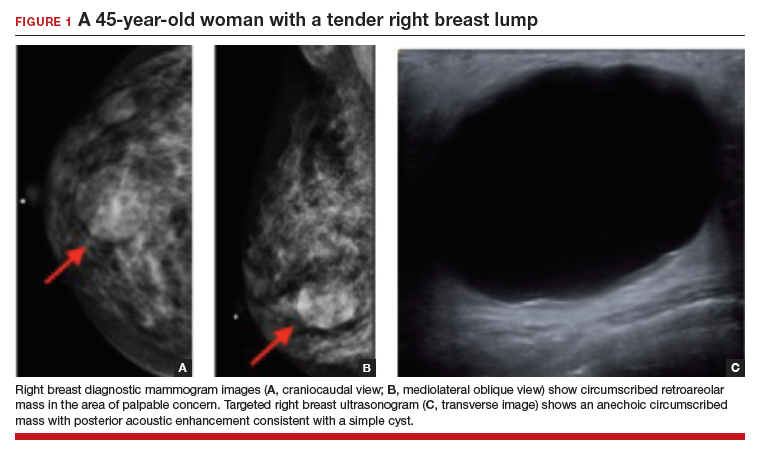
On ultrasound, a simple cyst is an anechoic, well-circumscribed mass with a thin capsule and with increased through transmission. Patients with small and asymptomatic simple cysts do not need imaging follow-up and can return for routine screening mammograms.
A breast surgeon, radiologist, or gynecologist can perform percutaneous aspiration if a cyst is large and symptomatic. A cyst with low-level internal echoes, fluid-fluid, or fluid-debris levels is considered a complicated cyst. Differential diagnosis also includes hematoma, fat necrosis, abscess, and galactocele, depending on the clinical presentation. Fine-needle aspiration or short-interval follow-up5,6 is appropriate for complicated cysts, while incision and drainage is indicated in patients with infected cysts and abscesses. A cyst with a solid component is considered a cystic, solid mass, and core needle biopsy is recommended. The differential diagnosis for cysts with solid components includes intracystic papilloma, papillary carcinoma, ductal carcinoma in situ, and necrotic cancers.5,6
Continue to: CASE 2 Painless breast mass in a young woman...
CASE 2 Painless breast mass in a young woman
A 22-year-old woman presents with a 2-month history of breast lump, which is not associated with pain or nipple discharge. On examination, there is a 2 x 2–cm mass in the right breast at 12 o’clock, 2 cm from the nipple, which is mobile, smooth, and nontender on palpation.
Fibroadenoma
In this 22-year-old, the initial imaging of choice is breast ultrasonography. Breast ultrasonography can differentiate a cystic mass from a solid mass, and it does not involve radiation. Right breast targeted ultrasound showed a circumscribed oval homogeneous hypoechoic mass that is wider than tall (FIGURE 2). The patient desired surgical removal, and a pre-lumpectomy core needle biopsy revealed a fibroadenoma.
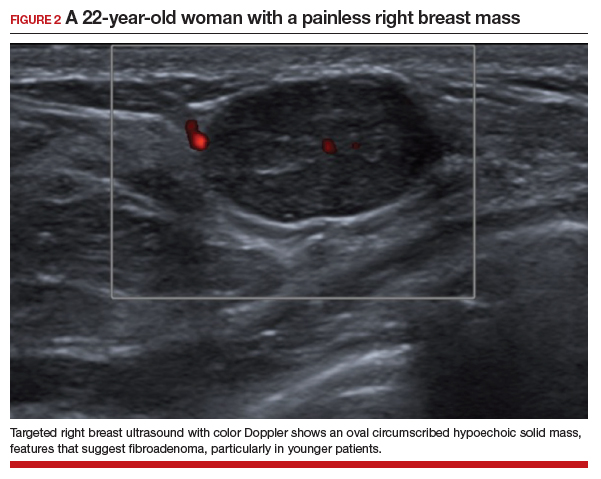
Fibroadenoma is the most common benign tumor of the breast. It is most often encountered in premenopausal women. Patients present with a painless breast lump, which is smooth and mobile on palpation. Fibroadenoma can be followed expectantly with repeat ultrasound (to assess over time for growth) if it is small and asymptomatic. No further action is needed if it remains stable. If a patient desires surgical excision, a core needle biopsy is usually performed before lumpectomy.
Excisional biopsy or removal of the mass is recommended if the mass is greater than 3 or 4 cm, is symptomatic, or if there is an increase in size that raises clinical concern for phyllodes tumor. Imaging features that are concerning for phyllodes tumors are size greater than 3 cm, indistinct or microlobulated margins, and heterogeneous echo pattern.7,8 In cases in which the imaging features are concerning for phyllodes tumor and a core needle biopsy is not definitive, wide surgical excision is recommended for definitive diagnosis.8
CASE 3 Patient develops breast mass post-surgery
A 45-year-old woman presents with a tender left breast mass that she noticed 2 months after breast reduction surgery. It has been increasing in size since. On clinical examination, a 4 x 4–cm mass is found at the surgical scar site, which is indurated on palpation and tender.
Fat necrosis
In this 45-year-old, the initial test of choice is diagnostic mammography, which showed a somewhat circumscribed area with fat under the palpable marker (FIGURE 3a). Breast ultrasonography was performed for further evaluation, which was inconclusive as the ultrasound showed ill-defined areas of mixed echogenicity (FIGURE 3b). Breast magnetic resonance imaging (MRI) clearly demonstrated fat necrosis in the area of the palpable lump (FIGURE 3c).

Fat necrosis of the breast is an inflammatory process that is seen after breast trauma or surgery. It can present as an incidental mammogram finding or a palpable mass. The patient may give a history of trauma, breast reduction surgery, or breast cancer surgery followed by radiation treatment. On clinical examination, fat necrosis occasionally can present as a firm mass with skin retraction or swelling concerning for cancer. Imaging features are variable depending on the stage of fat necrosis and inflammation.9-11
A mammogram may demonstrate a circumscribed fat-containing mass, an ill-defined mass, asymmetry or calcified oil cyst, and dystrophic calcifications. On ultrasound, fat necrosis can appear as anechoic or hypoechoic or as a complicated cyst or a mixed cystic, solid mass. MRI demonstrates a circumscribed or irregular fat-containing mass, with or without enhancement, and architectural distortion.
When the imaging features are clearly benign—for example, a circumscribed fat-containing mass on mammogram or on ultrasound or, on MRI, marked hypointensity of fat in the center of a circumscribed mass when compared with surrounding fat (keyhole sign)—no further follow-up is needed. When the imaging features are indeterminate, however, a short-interval follow-up can be considered. In cases with irregular fat-containing mass with enhancement, core needle biopsy is indicated to exclude cancer. If the workup remains inconclusive and the level of clinical suspicion is high, surgical excision can be performed for a definitive diagnosis.12
Continue to: Investigating breast pain: Imaging workup and management...
Investigating breast pain: Imaging workup and management
Breast pain, or mastalgia, is the most common concern of women presenting to a breast clinic and accounts for approximately half of such encounters.13 Causes of breast pain include hormonal changes, fibrocystic changes, musculoskeletal causes (such as costochondritis), lack of support, infection, and injury. While mastalgia often causes patient concern, the risk of malignancy in a woman presenting with breast pain alone is low. Still, it is essential to rule out other findings suspicious for cancer (mass, skin changes, or nipple discharge) with a thorough history and breast examination.
In this section, we review clinical presentation, imaging workup, and management for breast pain.
CASE 4 Woman with noncyclic breast pain
A 26-year-old woman presents to the clinic with mastalgia. The pain is noncyclic and primarily located in the upper outer quadrant of her left breast. There is no history of breast cancer in her family. She has no suspicious findings on the breast examination.
Mastalgia
The test of choice for this 26-year-old with focal left breast pain is targeted breast ultrasound. The patient’s ultrasound image showed no suspicious findings or solid or cystic mass (FIGURE 4).
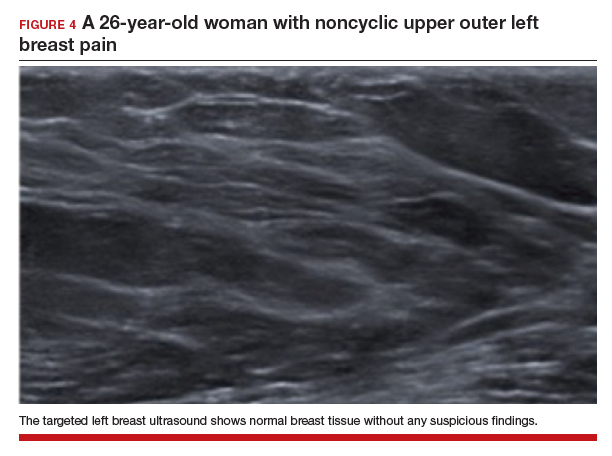
Two important characteristics of breast pain are whether it is noncyclical and whether it is focal. According to the American College of Radiology, no breast imaging is recommended for clinically insignificant cyclical, nonfocal (greater than 1 quadrant)/diffuse pain, as this type of mastalgia is not associated with malignancy.14
For patients age 40 or older, if they are not up to date with their annual screening mammogram, then a mammogram should be performed. An imaging workup is warranted for clinically significant mastalgia that is noncyclical and focal. Even then, no malignancy is identified in most patients with clinically significant mastalgia; in patients with breast pain as their only symptom, the prevalence of breast cancer is 0% to 3.0%.15-19
The initial imaging modality differs by patient age: younger than 30 years, ultrasonography; between 30 and 40 years, mammography or ultrasonography; and older than 40 years, mammography first followed by ultrasonography.14
Treatment of breast pain is primarily symptomatic, and evidence for specific treatments is generally lacking. Cyclical breast pain resolves spontaneously in 20% to 30% of women, while noncyclical pain responds poorly to treatment but resolves spontaneously in half of women.20 Reassurance is important and wearing a supportive bra often can alleviate breast pain. In addition, reducing caffeine intake can be helpful.
As a first-line treatment, both topical (diclofenac) and oral nonsteroidal anti-inflammatory drugs effectively can relieve breast pain. Supplements and herbal remedies (for example, evening primrose oil, vitamin E, flaxseed) have varying effectiveness and are of questionable benefit as few have trials to support their effectiveness.4 Danazol and tamoxifen have been shown to have some benefits but they also have adverse effects.20 Surgery does not play a role in the treatment of mastalgia.
CASE 5 Breastfeeding woman with breast pain
A 27-year-old postpartum woman presents with concerns for redness and pain in the upper inner left breast. She has been breastfeeding for the past few months. Breast examination demonstrates a 5-cm area of erythema and warmth but no fluctuance or masses.
Lactational mastitis
Targeted ultrasonography is the test of choice for this 27-year-old patient with focal breast pain, and the imaging revealed edema of subcutaneous tissues and ill-defined hypoechoic areas, likely inflamed fat lobules (FIGURE 5). These findings suggest uncomplicated lactational mastitis, which can be treated with antibiotics. Generally, the mastitis will improve within days of starting the antibiotics; if it does not improve, repeat examination and ultrasound should be performed to look for formation of an abscess that may require aspiration.
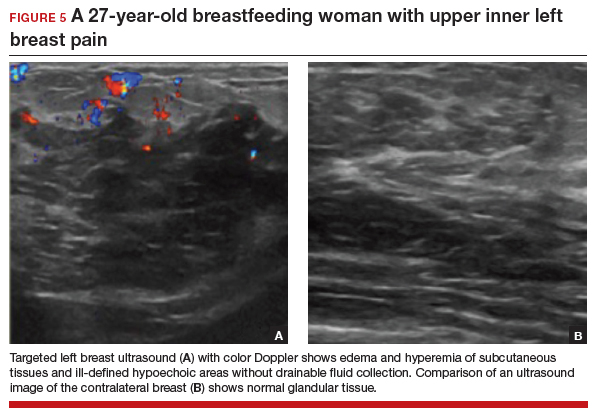
Continue to: CASE 6 Woman with painful periareolar mass...
CASE 6 Woman with painful periareolar mass
A 42-year-old perimenopausal woman describes having pain near the nipple of her right breast. She is a smoker and has no history of breast cancer in her family. Examination demonstrates a palpable, erythematous, painful, 3-cm periareolar fluctuant mass.
Nonpuerperal periareolar abscess
Appropriate initial imaging for this 42-year-old patient with focal pain is a diagnostic mammogram, which showed skin thickening and a retroareolar mass (FIGURE 6a). Further evaluation with targeted ultrasound showed a thick-walled anechoic collection with echoes compatible with an abscess (FIGURE 6b).
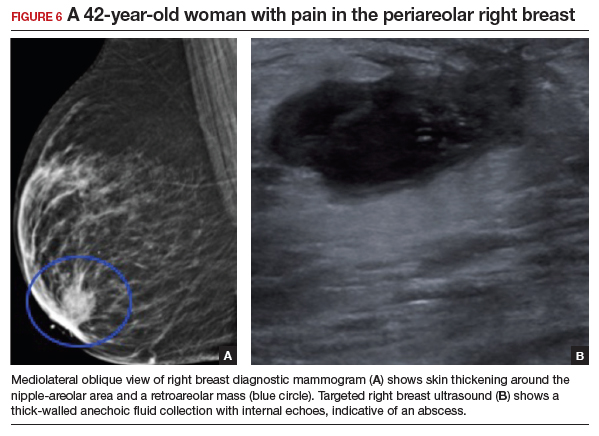
Mammographic findings in a patient with mastitis may be normal or demonstrate skin and trabecular thickening. Ultrasound imaging may show dilated ducts and heterogeneous tissue secondary to inflammation and edema without a discrete fluid collection. In cases with breast abscess, in addition to the mammographic findings described above, a mass, or an asymmetry, may be seen, most commonly in a subareolar location. On ultrasound, a hypoechoic collection with mobile debris, no internal flow on Doppler, and thick hypervascular walls can be seen with abscess, occasionally giving the appearance of a complicated cyst or a mixed cystic, solid mass.
The most important differential for mastitis is inflammatory breast cancer. Most cancers appear solid but can have central necrosis, mimicking a complicated cystic mass on ultrasound. The location for mastitis or abscess is most frequently subareolar. The presence of microcalcifications in a mass indicates the possibility of cancer.
Contrast-enhanced MRI can be helpful to differentiate between infection and cancer, with cancers showing initial early enhancement and washout kinetics compared with infected collections that show no enhancement or peripheral enhancement with a plateau or persistent enhancement curves. When clinical and imaging findings are unchanged after treatment of mastitis and abscesses, a core needle biopsy should be performed.21,22
There are 2 categories of mastitis and breast abscess: lactational and nonpuerperal (all mastitis that occurs outside the lactational period). The World Health Organization definition of puerperal mastitis includes pain, local redness, warmth and swelling of the breast (usually unilateral), fever, and malaise.4 Concerning etiology, epithelial lesions in the nipple area caused by breastfeeding can allow pathogens to enter and cause infection. The most common microorganism is Staphylococcus aureus.4 Continued emptying of the breast is important, combined with early antibiotic therapy (dicloxacillin is often the first line; if the patient is penicillin allergic, use a macrolide such as clindamycin). If no improvement is seen in 48 to 72 hours, imaging should be performed.
In most cases, continuation of breastfeeding is possible. If mastitis has evolved into an abscess in a lactating woman, it can be aspirated under ultrasound guidance. Incision and drainage should be avoided unless the abscess persists after multiple aspiration attempts, it is large, or if the overlying skin is thin or otherwise appears nonviable.
Nonpuerperal mastitis includes peripheral, periductal, and idiopathic granulomatous mastitis (IGM). Peripheral mastitis behaves like infections/abscesses in other soft tissues, responds well to treatment (antibiotics and percutaneous drainage), and is less likely to recur than periductal mastitis and IGM.21,23
Periductal mastitis and abscess, also known as Zuska disease, has a pathogenesis distinct from other forms of mastitis. Squamous metaplasia of the usual cuboidal epithelium of the breast ducts leads to keratin plugging that can cause infection.23 Risk factors include obesity, smoking, and macromastia. The typical presentation of Zuska disease is a woman with a history of chronic smoking and/or a congenital cleft in the central nipple.23 Periareolar signs of inflammation (redness, swelling, warmth) may be accompanied by an abscess. These can recur and lead to chronic fistula formation, especially if there is a history of intervention (such as aspiration, incision, and drainage).
Treatment of Zuska disease includes symptom relief and antibiotics. If S aureus is present, infection with methicillin-resistant S aureus is likely, and treatment with clindamycin or amoxicillin/clavulanic acid is preferred. If abscess is present, aspiration (preferred, often under ultrasound guidance) or incision and drainage (if the skin is compromised) may be required. If disease is recurrent or associated with a chronically draining fistula, surgical intervention may be warranted, in which resolution requires removing the keratin-plugged ducts in and immediately below the central core of the nipple. Given the association between Zuska disease and smoking, cessation should be encouraged, although there is no guarantee that this will resolve the issue.23
Continue to: CASE 7 Patient with breast pain and swelling...
CASE 7 Patient with breast pain and swelling
A 39-year-old woman presents with left breast swelling and pain of 1 month’s duration. On examination, there is a 6-cm area of edema, induration, and erythema.
Granulomatous mastitis
A diagnostic mammogram and ultrasound demonstrated an ill-defined hypoechoic mass (FIGURE 7a). Ultrasound-guided biopsy was performed, which showed granulomatous mastitis, negative for fungus and acid-fast bacilli. The patient was treated with prednisone and gradually improved (FIGURE 7b).
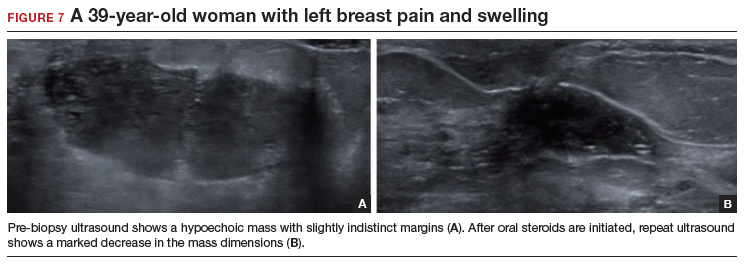
Granulomatous mastitis (GM) is a rare benign inflammatory process, with etiologies that include fungal infections, tuberculosis, Wegener granulomatosis, sarcoidosis, and idiopathic causes. Imaging can be nonspecific and show variable features. Mammograms can appear normal or show asymmetry or mass and skin thickening. Ultrasound can show heterogeneous parenchyma, ill-defined hypoechoic collection, or a mass with margins that can be circumscribed or indistinct or with tubular extensions, with or without overlying skin thickening, fistulas, and reactive lymph nodes.24
In this clinical setting, the differential diagnosis includes infectious mastitis, inflammatory breast cancer, foreign body injection granulomas, and diabetic mastopathy. Treatment involves drainage and fluid culture if there is a collection on imaging. A core biopsy is performed if imaging demonstrates a solid mass or fluid culture is negative and symptoms persist or recur. Oral steroids represent the mainstay of treatment if a core biopsy shows GM. However, immunosuppressants, including methotrexate, and surgery are options if initial treatment is not helpful.25,26
Conclusion
Breast symptoms are common reasons for patient visits to obstetricians and gynecologists. With a good understanding of the various symptomatic breast diseases and conditions, and by having a close collaboration with radiologists and breast surgeons, clinicians can provide excellent care to these patients and thereby improve patient outcomes and satisfaction. ●
- Eberl MM, Phillips RL Jr, Lamberts H, et al. Characterizing breast symptoms in family practice. Ann Fam Med. 2008;6:528-533.
- Malherbe F, Nel D, Molabe H, et al. Palpable breast lumps: an age-based approach to evaluation and diagnosis. S Afr Fam Pract (2022). 2022;64:e1-e5.
- Expert Panel on Breast Imaging; Klein KA, Kocher M, Lourenco AP, et al. American College of Radiology ACR appropriateness criteria: palpable breast masses. Accessed February 15, 2023. https://acsearch.acr.org/docs/69495/Narrative/
- Stachs A, Stubert J, Reimer T, et al. Benign breast disease in women. Dtsch Arztebl Int. 2019;116:565574.
- Hines N, Slanetz PJ, Eisenberg RL. Cystic masses of the breast. AJR Am J Roentgenol. 2010;194:W122133.
- Berg WA. Reducing unnecessary biopsy and follow-up of benign cystic breast lesions. Radiology. 2020;295:52-53.
- Duman L, Gezer NS, Balcı P, et al. Differentiation between phyllodes tumors and fibroadenomas based on mammographic sonographic and MRI features. Breast Care. 2016;11:123-127.
- Lerwill MF, Lee AHS, Tan PH. Fibroepithelial tumours of the breast—a review. Virchows Arch. 2022;480:45-63.
- Vasei N, Shishegar A, Ghalkhani F, et al. Fat necrosis in the breast: a systematic review of clinical. Lipids Health Dis. 2019;18:139.
- Kerridge WD, Kryvenko ON, Thompson A, et al. Fat necrosis of the breast: a pictorial review of the mammographic, ultrasound, CT, and MRI findings with histopathologic correlation. Radiol Res Pract. 2015;2015:613139.
- Taboada JL, Stephens TW, Krishnamurthy S, et al. The many faces of fat necrosis in the breast. AJR Am J Roentgenol. 2009;192:815-825.
- Tan PH, Lai LM, Carrington EV, et al. Fat necrosis of the breast—a review. Breast. 2006;15:313-318.
- Holbrook AI. Breast pain, a common grievance: guidance to radiologists. AJR Am J Roentgenol. 2020;214:259-264.
- Expert Panel on Breast Imaging; Moy L, Heller SL, Bailey L, et al. ACR appropriateness criteria: palpable breast masses. J Am Coll Radiol. 2017;14:S203-S224.
- Chetlen AL, Kapoor MM, Watts MR. Mastalgia: imaging workup appropriateness. Acad Radiol. 2017;24:345-349.
- Arslan M, Kucukerdem HS, Can H, et al. Retrospective analysis of women with only mastalgia. J Breast Health. 2016;12:151-154.
- Fariselli G, Lepera P, Viganotti G, et al. Localized mastalgia as presenting symptom in breast cancer. Eur J Surg Oncol. 1988;14:213-215.
- Leddy R, Irshad A, Zerwas E, et al. Role of breast ultrasound and mammography in evaluating patients presenting with focal breast pain in the absence of a palpable lump. Breast J. 2013;19:582-589.
- Leung JW, Kornguth PJ, Gotway MB. Utility of targeted sonography in the evaluation of focal breast pain. J Ultrasound Med. 2002;21:521-526.
- Goyal A. Breast pain. BMJ Clin Evid. 2011; 2011:0812.
- Kasales CJ, Han B, Smith Jr JS, et al. Nonpuerperal mastitis and subareolar abscess of the breast. AJR Am J Roentgenol. 2014;202:W133-W139.
- Mahoney MC, Ingram AD. Breast emergencies: types, imaging features, and management. AJR Am J Roentgenol. 2014;202:W390-W399.
- Snider HC. Management of mastitis, abscess, and fistula. Surg Clin North Am. 2022;102:1103-1116.
- Oztekin PS, Durhan G, Kosar PN, et al. Imaging findings in patients with granulomatous mastitis. Iran J Radiol. 2016;13:e33900.
- Pluguez-Turull CW, Nanyes JE, Quintero CJ, et al. Idiopathic granulomatous mastitis: manifestations at multimodality imaging and pitfalls. Radiographics. 2018;38:330-356.
- Hovanessian-Larsen LJ, Peyvandi B, Klipfel N, et al. Granulomatous lobular mastitis: imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193:574-581.
The vast majority of symptomatic breast conditions are benign, with the most common symptoms being palpable mass and breast pain. Clinicians, including primary care clinicians and gynecologists, play a crucial role by performing the initial assessment and subsequent therapies and referrals and serve as the mediator between the specialists and by being the patient’s spokesperson. It is therefore important for clinicians to be aware of the various possible causes of these breast symptoms, to know which imaging tests to order, and also to understand the indications for biopsies and surgical referral.
Common types of breast lumps: Imaging workup and management
Accounting for 8% of women who present with breast symptoms, breast lump is the second most common symptom after breast pain.1 The positive likelihood ratio of finding breast cancer is highest among women with breast lumps compared with any other breast symptoms. Therefore, anxiety is related to this symptom, and a thorough evaluation is recommended.1 Cysts, fibroadenoma, and fat necrosis are 3 common benign causes of breast lumps.2
In this section, we review clinical presentation, imaging workup, and management strategies for common types of breast lumps.
CASE 1 Woman with tender breast lump
A 45-year-old woman presents with a breast lump of 6 months’ duration that is associated with a change in size with the menstrual cycle and pain. Clinical examination reveals a 4 x 4.5–cm mass in the right breast in the retroareolar region, which is smooth with some tenderness on palpation.
Breast cyst
According to the American College of Radiology appropriateness criteria for an adult woman 40 years of age or older who presents with a palpable breast mass, the initial imaging study is diagnostic mammography with or without digital tomosynthesis, usually followed by a directed ultrasound. If the mammogram is suspicious or highly suggestive of malignancy, or in cases where the mammogram does not show an abnormality, the next recommended step is breast ultrasonography. Any suspicious findings on ultrasound or mammogram should be followed by an image guided biopsy. Ultrasonography also may be appropriate if the mammogram findings are benign or probably benign.
For an adult woman younger than age 30 who presents with a palpable breast mass, breast ultrasonography is the appropriate initial imaging study. If the ultrasound is suspicious or highly suggestive of malignancy, then performing diagnostic mammography with or without digital tomosynthesis or ultrasound-guided core needle biopsy of the mass are both considered appropriate. However, no further imaging is recommended if the ultrasound is benign, probably benign, or negative. Breast ultrasonography or mammography is appropriate as the initial imaging test for adult women aged 30 to 39 years who present with a palpable breast mass.3,4
Approximately 50% of women after age 30 may develop fibrocystic breast disease, and 20% of them can present with pain or lump due to a macrocysts. Simple cysts must be distinguished from complex cysts with the help of ultrasound as the latter are associated with 23% to 31% increased risk of malignancy.
In this 45-year-old patient, the initial mammogram demonstrated a circumscribed mass underneath the area of palpable concern (FIGURE 1a, 1b). Targeted breast ultrasonography was performed for further assessment, which depicted the mass as a benign simple cyst (FIGURE 1c).

On ultrasound, a simple cyst is an anechoic, well-circumscribed mass with a thin capsule and with increased through transmission. Patients with small and asymptomatic simple cysts do not need imaging follow-up and can return for routine screening mammograms.
A breast surgeon, radiologist, or gynecologist can perform percutaneous aspiration if a cyst is large and symptomatic. A cyst with low-level internal echoes, fluid-fluid, or fluid-debris levels is considered a complicated cyst. Differential diagnosis also includes hematoma, fat necrosis, abscess, and galactocele, depending on the clinical presentation. Fine-needle aspiration or short-interval follow-up5,6 is appropriate for complicated cysts, while incision and drainage is indicated in patients with infected cysts and abscesses. A cyst with a solid component is considered a cystic, solid mass, and core needle biopsy is recommended. The differential diagnosis for cysts with solid components includes intracystic papilloma, papillary carcinoma, ductal carcinoma in situ, and necrotic cancers.5,6
Continue to: CASE 2 Painless breast mass in a young woman...
CASE 2 Painless breast mass in a young woman
A 22-year-old woman presents with a 2-month history of breast lump, which is not associated with pain or nipple discharge. On examination, there is a 2 x 2–cm mass in the right breast at 12 o’clock, 2 cm from the nipple, which is mobile, smooth, and nontender on palpation.
Fibroadenoma
In this 22-year-old, the initial imaging of choice is breast ultrasonography. Breast ultrasonography can differentiate a cystic mass from a solid mass, and it does not involve radiation. Right breast targeted ultrasound showed a circumscribed oval homogeneous hypoechoic mass that is wider than tall (FIGURE 2). The patient desired surgical removal, and a pre-lumpectomy core needle biopsy revealed a fibroadenoma.

Fibroadenoma is the most common benign tumor of the breast. It is most often encountered in premenopausal women. Patients present with a painless breast lump, which is smooth and mobile on palpation. Fibroadenoma can be followed expectantly with repeat ultrasound (to assess over time for growth) if it is small and asymptomatic. No further action is needed if it remains stable. If a patient desires surgical excision, a core needle biopsy is usually performed before lumpectomy.
Excisional biopsy or removal of the mass is recommended if the mass is greater than 3 or 4 cm, is symptomatic, or if there is an increase in size that raises clinical concern for phyllodes tumor. Imaging features that are concerning for phyllodes tumors are size greater than 3 cm, indistinct or microlobulated margins, and heterogeneous echo pattern.7,8 In cases in which the imaging features are concerning for phyllodes tumor and a core needle biopsy is not definitive, wide surgical excision is recommended for definitive diagnosis.8
CASE 3 Patient develops breast mass post-surgery
A 45-year-old woman presents with a tender left breast mass that she noticed 2 months after breast reduction surgery. It has been increasing in size since. On clinical examination, a 4 x 4–cm mass is found at the surgical scar site, which is indurated on palpation and tender.
Fat necrosis
In this 45-year-old, the initial test of choice is diagnostic mammography, which showed a somewhat circumscribed area with fat under the palpable marker (FIGURE 3a). Breast ultrasonography was performed for further evaluation, which was inconclusive as the ultrasound showed ill-defined areas of mixed echogenicity (FIGURE 3b). Breast magnetic resonance imaging (MRI) clearly demonstrated fat necrosis in the area of the palpable lump (FIGURE 3c).

Fat necrosis of the breast is an inflammatory process that is seen after breast trauma or surgery. It can present as an incidental mammogram finding or a palpable mass. The patient may give a history of trauma, breast reduction surgery, or breast cancer surgery followed by radiation treatment. On clinical examination, fat necrosis occasionally can present as a firm mass with skin retraction or swelling concerning for cancer. Imaging features are variable depending on the stage of fat necrosis and inflammation.9-11
A mammogram may demonstrate a circumscribed fat-containing mass, an ill-defined mass, asymmetry or calcified oil cyst, and dystrophic calcifications. On ultrasound, fat necrosis can appear as anechoic or hypoechoic or as a complicated cyst or a mixed cystic, solid mass. MRI demonstrates a circumscribed or irregular fat-containing mass, with or without enhancement, and architectural distortion.
When the imaging features are clearly benign—for example, a circumscribed fat-containing mass on mammogram or on ultrasound or, on MRI, marked hypointensity of fat in the center of a circumscribed mass when compared with surrounding fat (keyhole sign)—no further follow-up is needed. When the imaging features are indeterminate, however, a short-interval follow-up can be considered. In cases with irregular fat-containing mass with enhancement, core needle biopsy is indicated to exclude cancer. If the workup remains inconclusive and the level of clinical suspicion is high, surgical excision can be performed for a definitive diagnosis.12
Continue to: Investigating breast pain: Imaging workup and management...
Investigating breast pain: Imaging workup and management
Breast pain, or mastalgia, is the most common concern of women presenting to a breast clinic and accounts for approximately half of such encounters.13 Causes of breast pain include hormonal changes, fibrocystic changes, musculoskeletal causes (such as costochondritis), lack of support, infection, and injury. While mastalgia often causes patient concern, the risk of malignancy in a woman presenting with breast pain alone is low. Still, it is essential to rule out other findings suspicious for cancer (mass, skin changes, or nipple discharge) with a thorough history and breast examination.
In this section, we review clinical presentation, imaging workup, and management for breast pain.
CASE 4 Woman with noncyclic breast pain
A 26-year-old woman presents to the clinic with mastalgia. The pain is noncyclic and primarily located in the upper outer quadrant of her left breast. There is no history of breast cancer in her family. She has no suspicious findings on the breast examination.
Mastalgia
The test of choice for this 26-year-old with focal left breast pain is targeted breast ultrasound. The patient’s ultrasound image showed no suspicious findings or solid or cystic mass (FIGURE 4).

Two important characteristics of breast pain are whether it is noncyclical and whether it is focal. According to the American College of Radiology, no breast imaging is recommended for clinically insignificant cyclical, nonfocal (greater than 1 quadrant)/diffuse pain, as this type of mastalgia is not associated with malignancy.14
For patients age 40 or older, if they are not up to date with their annual screening mammogram, then a mammogram should be performed. An imaging workup is warranted for clinically significant mastalgia that is noncyclical and focal. Even then, no malignancy is identified in most patients with clinically significant mastalgia; in patients with breast pain as their only symptom, the prevalence of breast cancer is 0% to 3.0%.15-19
The initial imaging modality differs by patient age: younger than 30 years, ultrasonography; between 30 and 40 years, mammography or ultrasonography; and older than 40 years, mammography first followed by ultrasonography.14
Treatment of breast pain is primarily symptomatic, and evidence for specific treatments is generally lacking. Cyclical breast pain resolves spontaneously in 20% to 30% of women, while noncyclical pain responds poorly to treatment but resolves spontaneously in half of women.20 Reassurance is important and wearing a supportive bra often can alleviate breast pain. In addition, reducing caffeine intake can be helpful.
As a first-line treatment, both topical (diclofenac) and oral nonsteroidal anti-inflammatory drugs effectively can relieve breast pain. Supplements and herbal remedies (for example, evening primrose oil, vitamin E, flaxseed) have varying effectiveness and are of questionable benefit as few have trials to support their effectiveness.4 Danazol and tamoxifen have been shown to have some benefits but they also have adverse effects.20 Surgery does not play a role in the treatment of mastalgia.
CASE 5 Breastfeeding woman with breast pain
A 27-year-old postpartum woman presents with concerns for redness and pain in the upper inner left breast. She has been breastfeeding for the past few months. Breast examination demonstrates a 5-cm area of erythema and warmth but no fluctuance or masses.
Lactational mastitis
Targeted ultrasonography is the test of choice for this 27-year-old patient with focal breast pain, and the imaging revealed edema of subcutaneous tissues and ill-defined hypoechoic areas, likely inflamed fat lobules (FIGURE 5). These findings suggest uncomplicated lactational mastitis, which can be treated with antibiotics. Generally, the mastitis will improve within days of starting the antibiotics; if it does not improve, repeat examination and ultrasound should be performed to look for formation of an abscess that may require aspiration.

Continue to: CASE 6 Woman with painful periareolar mass...
CASE 6 Woman with painful periareolar mass
A 42-year-old perimenopausal woman describes having pain near the nipple of her right breast. She is a smoker and has no history of breast cancer in her family. Examination demonstrates a palpable, erythematous, painful, 3-cm periareolar fluctuant mass.
Nonpuerperal periareolar abscess
Appropriate initial imaging for this 42-year-old patient with focal pain is a diagnostic mammogram, which showed skin thickening and a retroareolar mass (FIGURE 6a). Further evaluation with targeted ultrasound showed a thick-walled anechoic collection with echoes compatible with an abscess (FIGURE 6b).

Mammographic findings in a patient with mastitis may be normal or demonstrate skin and trabecular thickening. Ultrasound imaging may show dilated ducts and heterogeneous tissue secondary to inflammation and edema without a discrete fluid collection. In cases with breast abscess, in addition to the mammographic findings described above, a mass, or an asymmetry, may be seen, most commonly in a subareolar location. On ultrasound, a hypoechoic collection with mobile debris, no internal flow on Doppler, and thick hypervascular walls can be seen with abscess, occasionally giving the appearance of a complicated cyst or a mixed cystic, solid mass.
The most important differential for mastitis is inflammatory breast cancer. Most cancers appear solid but can have central necrosis, mimicking a complicated cystic mass on ultrasound. The location for mastitis or abscess is most frequently subareolar. The presence of microcalcifications in a mass indicates the possibility of cancer.
Contrast-enhanced MRI can be helpful to differentiate between infection and cancer, with cancers showing initial early enhancement and washout kinetics compared with infected collections that show no enhancement or peripheral enhancement with a plateau or persistent enhancement curves. When clinical and imaging findings are unchanged after treatment of mastitis and abscesses, a core needle biopsy should be performed.21,22
There are 2 categories of mastitis and breast abscess: lactational and nonpuerperal (all mastitis that occurs outside the lactational period). The World Health Organization definition of puerperal mastitis includes pain, local redness, warmth and swelling of the breast (usually unilateral), fever, and malaise.4 Concerning etiology, epithelial lesions in the nipple area caused by breastfeeding can allow pathogens to enter and cause infection. The most common microorganism is Staphylococcus aureus.4 Continued emptying of the breast is important, combined with early antibiotic therapy (dicloxacillin is often the first line; if the patient is penicillin allergic, use a macrolide such as clindamycin). If no improvement is seen in 48 to 72 hours, imaging should be performed.
In most cases, continuation of breastfeeding is possible. If mastitis has evolved into an abscess in a lactating woman, it can be aspirated under ultrasound guidance. Incision and drainage should be avoided unless the abscess persists after multiple aspiration attempts, it is large, or if the overlying skin is thin or otherwise appears nonviable.
Nonpuerperal mastitis includes peripheral, periductal, and idiopathic granulomatous mastitis (IGM). Peripheral mastitis behaves like infections/abscesses in other soft tissues, responds well to treatment (antibiotics and percutaneous drainage), and is less likely to recur than periductal mastitis and IGM.21,23
Periductal mastitis and abscess, also known as Zuska disease, has a pathogenesis distinct from other forms of mastitis. Squamous metaplasia of the usual cuboidal epithelium of the breast ducts leads to keratin plugging that can cause infection.23 Risk factors include obesity, smoking, and macromastia. The typical presentation of Zuska disease is a woman with a history of chronic smoking and/or a congenital cleft in the central nipple.23 Periareolar signs of inflammation (redness, swelling, warmth) may be accompanied by an abscess. These can recur and lead to chronic fistula formation, especially if there is a history of intervention (such as aspiration, incision, and drainage).
Treatment of Zuska disease includes symptom relief and antibiotics. If S aureus is present, infection with methicillin-resistant S aureus is likely, and treatment with clindamycin or amoxicillin/clavulanic acid is preferred. If abscess is present, aspiration (preferred, often under ultrasound guidance) or incision and drainage (if the skin is compromised) may be required. If disease is recurrent or associated with a chronically draining fistula, surgical intervention may be warranted, in which resolution requires removing the keratin-plugged ducts in and immediately below the central core of the nipple. Given the association between Zuska disease and smoking, cessation should be encouraged, although there is no guarantee that this will resolve the issue.23
Continue to: CASE 7 Patient with breast pain and swelling...
CASE 7 Patient with breast pain and swelling
A 39-year-old woman presents with left breast swelling and pain of 1 month’s duration. On examination, there is a 6-cm area of edema, induration, and erythema.
Granulomatous mastitis
A diagnostic mammogram and ultrasound demonstrated an ill-defined hypoechoic mass (FIGURE 7a). Ultrasound-guided biopsy was performed, which showed granulomatous mastitis, negative for fungus and acid-fast bacilli. The patient was treated with prednisone and gradually improved (FIGURE 7b).

Granulomatous mastitis (GM) is a rare benign inflammatory process, with etiologies that include fungal infections, tuberculosis, Wegener granulomatosis, sarcoidosis, and idiopathic causes. Imaging can be nonspecific and show variable features. Mammograms can appear normal or show asymmetry or mass and skin thickening. Ultrasound can show heterogeneous parenchyma, ill-defined hypoechoic collection, or a mass with margins that can be circumscribed or indistinct or with tubular extensions, with or without overlying skin thickening, fistulas, and reactive lymph nodes.24
In this clinical setting, the differential diagnosis includes infectious mastitis, inflammatory breast cancer, foreign body injection granulomas, and diabetic mastopathy. Treatment involves drainage and fluid culture if there is a collection on imaging. A core biopsy is performed if imaging demonstrates a solid mass or fluid culture is negative and symptoms persist or recur. Oral steroids represent the mainstay of treatment if a core biopsy shows GM. However, immunosuppressants, including methotrexate, and surgery are options if initial treatment is not helpful.25,26
Conclusion
Breast symptoms are common reasons for patient visits to obstetricians and gynecologists. With a good understanding of the various symptomatic breast diseases and conditions, and by having a close collaboration with radiologists and breast surgeons, clinicians can provide excellent care to these patients and thereby improve patient outcomes and satisfaction. ●
The vast majority of symptomatic breast conditions are benign, with the most common symptoms being palpable mass and breast pain. Clinicians, including primary care clinicians and gynecologists, play a crucial role by performing the initial assessment and subsequent therapies and referrals and serve as the mediator between the specialists and by being the patient’s spokesperson. It is therefore important for clinicians to be aware of the various possible causes of these breast symptoms, to know which imaging tests to order, and also to understand the indications for biopsies and surgical referral.
Common types of breast lumps: Imaging workup and management
Accounting for 8% of women who present with breast symptoms, breast lump is the second most common symptom after breast pain.1 The positive likelihood ratio of finding breast cancer is highest among women with breast lumps compared with any other breast symptoms. Therefore, anxiety is related to this symptom, and a thorough evaluation is recommended.1 Cysts, fibroadenoma, and fat necrosis are 3 common benign causes of breast lumps.2
In this section, we review clinical presentation, imaging workup, and management strategies for common types of breast lumps.
CASE 1 Woman with tender breast lump
A 45-year-old woman presents with a breast lump of 6 months’ duration that is associated with a change in size with the menstrual cycle and pain. Clinical examination reveals a 4 x 4.5–cm mass in the right breast in the retroareolar region, which is smooth with some tenderness on palpation.
Breast cyst
According to the American College of Radiology appropriateness criteria for an adult woman 40 years of age or older who presents with a palpable breast mass, the initial imaging study is diagnostic mammography with or without digital tomosynthesis, usually followed by a directed ultrasound. If the mammogram is suspicious or highly suggestive of malignancy, or in cases where the mammogram does not show an abnormality, the next recommended step is breast ultrasonography. Any suspicious findings on ultrasound or mammogram should be followed by an image guided biopsy. Ultrasonography also may be appropriate if the mammogram findings are benign or probably benign.
For an adult woman younger than age 30 who presents with a palpable breast mass, breast ultrasonography is the appropriate initial imaging study. If the ultrasound is suspicious or highly suggestive of malignancy, then performing diagnostic mammography with or without digital tomosynthesis or ultrasound-guided core needle biopsy of the mass are both considered appropriate. However, no further imaging is recommended if the ultrasound is benign, probably benign, or negative. Breast ultrasonography or mammography is appropriate as the initial imaging test for adult women aged 30 to 39 years who present with a palpable breast mass.3,4
Approximately 50% of women after age 30 may develop fibrocystic breast disease, and 20% of them can present with pain or lump due to a macrocysts. Simple cysts must be distinguished from complex cysts with the help of ultrasound as the latter are associated with 23% to 31% increased risk of malignancy.
In this 45-year-old patient, the initial mammogram demonstrated a circumscribed mass underneath the area of palpable concern (FIGURE 1a, 1b). Targeted breast ultrasonography was performed for further assessment, which depicted the mass as a benign simple cyst (FIGURE 1c).

On ultrasound, a simple cyst is an anechoic, well-circumscribed mass with a thin capsule and with increased through transmission. Patients with small and asymptomatic simple cysts do not need imaging follow-up and can return for routine screening mammograms.
A breast surgeon, radiologist, or gynecologist can perform percutaneous aspiration if a cyst is large and symptomatic. A cyst with low-level internal echoes, fluid-fluid, or fluid-debris levels is considered a complicated cyst. Differential diagnosis also includes hematoma, fat necrosis, abscess, and galactocele, depending on the clinical presentation. Fine-needle aspiration or short-interval follow-up5,6 is appropriate for complicated cysts, while incision and drainage is indicated in patients with infected cysts and abscesses. A cyst with a solid component is considered a cystic, solid mass, and core needle biopsy is recommended. The differential diagnosis for cysts with solid components includes intracystic papilloma, papillary carcinoma, ductal carcinoma in situ, and necrotic cancers.5,6
Continue to: CASE 2 Painless breast mass in a young woman...
CASE 2 Painless breast mass in a young woman
A 22-year-old woman presents with a 2-month history of breast lump, which is not associated with pain or nipple discharge. On examination, there is a 2 x 2–cm mass in the right breast at 12 o’clock, 2 cm from the nipple, which is mobile, smooth, and nontender on palpation.
Fibroadenoma
In this 22-year-old, the initial imaging of choice is breast ultrasonography. Breast ultrasonography can differentiate a cystic mass from a solid mass, and it does not involve radiation. Right breast targeted ultrasound showed a circumscribed oval homogeneous hypoechoic mass that is wider than tall (FIGURE 2). The patient desired surgical removal, and a pre-lumpectomy core needle biopsy revealed a fibroadenoma.

Fibroadenoma is the most common benign tumor of the breast. It is most often encountered in premenopausal women. Patients present with a painless breast lump, which is smooth and mobile on palpation. Fibroadenoma can be followed expectantly with repeat ultrasound (to assess over time for growth) if it is small and asymptomatic. No further action is needed if it remains stable. If a patient desires surgical excision, a core needle biopsy is usually performed before lumpectomy.
Excisional biopsy or removal of the mass is recommended if the mass is greater than 3 or 4 cm, is symptomatic, or if there is an increase in size that raises clinical concern for phyllodes tumor. Imaging features that are concerning for phyllodes tumors are size greater than 3 cm, indistinct or microlobulated margins, and heterogeneous echo pattern.7,8 In cases in which the imaging features are concerning for phyllodes tumor and a core needle biopsy is not definitive, wide surgical excision is recommended for definitive diagnosis.8
CASE 3 Patient develops breast mass post-surgery
A 45-year-old woman presents with a tender left breast mass that she noticed 2 months after breast reduction surgery. It has been increasing in size since. On clinical examination, a 4 x 4–cm mass is found at the surgical scar site, which is indurated on palpation and tender.
Fat necrosis
In this 45-year-old, the initial test of choice is diagnostic mammography, which showed a somewhat circumscribed area with fat under the palpable marker (FIGURE 3a). Breast ultrasonography was performed for further evaluation, which was inconclusive as the ultrasound showed ill-defined areas of mixed echogenicity (FIGURE 3b). Breast magnetic resonance imaging (MRI) clearly demonstrated fat necrosis in the area of the palpable lump (FIGURE 3c).

Fat necrosis of the breast is an inflammatory process that is seen after breast trauma or surgery. It can present as an incidental mammogram finding or a palpable mass. The patient may give a history of trauma, breast reduction surgery, or breast cancer surgery followed by radiation treatment. On clinical examination, fat necrosis occasionally can present as a firm mass with skin retraction or swelling concerning for cancer. Imaging features are variable depending on the stage of fat necrosis and inflammation.9-11
A mammogram may demonstrate a circumscribed fat-containing mass, an ill-defined mass, asymmetry or calcified oil cyst, and dystrophic calcifications. On ultrasound, fat necrosis can appear as anechoic or hypoechoic or as a complicated cyst or a mixed cystic, solid mass. MRI demonstrates a circumscribed or irregular fat-containing mass, with or without enhancement, and architectural distortion.
When the imaging features are clearly benign—for example, a circumscribed fat-containing mass on mammogram or on ultrasound or, on MRI, marked hypointensity of fat in the center of a circumscribed mass when compared with surrounding fat (keyhole sign)—no further follow-up is needed. When the imaging features are indeterminate, however, a short-interval follow-up can be considered. In cases with irregular fat-containing mass with enhancement, core needle biopsy is indicated to exclude cancer. If the workup remains inconclusive and the level of clinical suspicion is high, surgical excision can be performed for a definitive diagnosis.12
Continue to: Investigating breast pain: Imaging workup and management...
Investigating breast pain: Imaging workup and management
Breast pain, or mastalgia, is the most common concern of women presenting to a breast clinic and accounts for approximately half of such encounters.13 Causes of breast pain include hormonal changes, fibrocystic changes, musculoskeletal causes (such as costochondritis), lack of support, infection, and injury. While mastalgia often causes patient concern, the risk of malignancy in a woman presenting with breast pain alone is low. Still, it is essential to rule out other findings suspicious for cancer (mass, skin changes, or nipple discharge) with a thorough history and breast examination.
In this section, we review clinical presentation, imaging workup, and management for breast pain.
CASE 4 Woman with noncyclic breast pain
A 26-year-old woman presents to the clinic with mastalgia. The pain is noncyclic and primarily located in the upper outer quadrant of her left breast. There is no history of breast cancer in her family. She has no suspicious findings on the breast examination.
Mastalgia
The test of choice for this 26-year-old with focal left breast pain is targeted breast ultrasound. The patient’s ultrasound image showed no suspicious findings or solid or cystic mass (FIGURE 4).

Two important characteristics of breast pain are whether it is noncyclical and whether it is focal. According to the American College of Radiology, no breast imaging is recommended for clinically insignificant cyclical, nonfocal (greater than 1 quadrant)/diffuse pain, as this type of mastalgia is not associated with malignancy.14
For patients age 40 or older, if they are not up to date with their annual screening mammogram, then a mammogram should be performed. An imaging workup is warranted for clinically significant mastalgia that is noncyclical and focal. Even then, no malignancy is identified in most patients with clinically significant mastalgia; in patients with breast pain as their only symptom, the prevalence of breast cancer is 0% to 3.0%.15-19
The initial imaging modality differs by patient age: younger than 30 years, ultrasonography; between 30 and 40 years, mammography or ultrasonography; and older than 40 years, mammography first followed by ultrasonography.14
Treatment of breast pain is primarily symptomatic, and evidence for specific treatments is generally lacking. Cyclical breast pain resolves spontaneously in 20% to 30% of women, while noncyclical pain responds poorly to treatment but resolves spontaneously in half of women.20 Reassurance is important and wearing a supportive bra often can alleviate breast pain. In addition, reducing caffeine intake can be helpful.
As a first-line treatment, both topical (diclofenac) and oral nonsteroidal anti-inflammatory drugs effectively can relieve breast pain. Supplements and herbal remedies (for example, evening primrose oil, vitamin E, flaxseed) have varying effectiveness and are of questionable benefit as few have trials to support their effectiveness.4 Danazol and tamoxifen have been shown to have some benefits but they also have adverse effects.20 Surgery does not play a role in the treatment of mastalgia.
CASE 5 Breastfeeding woman with breast pain
A 27-year-old postpartum woman presents with concerns for redness and pain in the upper inner left breast. She has been breastfeeding for the past few months. Breast examination demonstrates a 5-cm area of erythema and warmth but no fluctuance or masses.
Lactational mastitis
Targeted ultrasonography is the test of choice for this 27-year-old patient with focal breast pain, and the imaging revealed edema of subcutaneous tissues and ill-defined hypoechoic areas, likely inflamed fat lobules (FIGURE 5). These findings suggest uncomplicated lactational mastitis, which can be treated with antibiotics. Generally, the mastitis will improve within days of starting the antibiotics; if it does not improve, repeat examination and ultrasound should be performed to look for formation of an abscess that may require aspiration.

Continue to: CASE 6 Woman with painful periareolar mass...
CASE 6 Woman with painful periareolar mass
A 42-year-old perimenopausal woman describes having pain near the nipple of her right breast. She is a smoker and has no history of breast cancer in her family. Examination demonstrates a palpable, erythematous, painful, 3-cm periareolar fluctuant mass.
Nonpuerperal periareolar abscess
Appropriate initial imaging for this 42-year-old patient with focal pain is a diagnostic mammogram, which showed skin thickening and a retroareolar mass (FIGURE 6a). Further evaluation with targeted ultrasound showed a thick-walled anechoic collection with echoes compatible with an abscess (FIGURE 6b).

Mammographic findings in a patient with mastitis may be normal or demonstrate skin and trabecular thickening. Ultrasound imaging may show dilated ducts and heterogeneous tissue secondary to inflammation and edema without a discrete fluid collection. In cases with breast abscess, in addition to the mammographic findings described above, a mass, or an asymmetry, may be seen, most commonly in a subareolar location. On ultrasound, a hypoechoic collection with mobile debris, no internal flow on Doppler, and thick hypervascular walls can be seen with abscess, occasionally giving the appearance of a complicated cyst or a mixed cystic, solid mass.
The most important differential for mastitis is inflammatory breast cancer. Most cancers appear solid but can have central necrosis, mimicking a complicated cystic mass on ultrasound. The location for mastitis or abscess is most frequently subareolar. The presence of microcalcifications in a mass indicates the possibility of cancer.
Contrast-enhanced MRI can be helpful to differentiate between infection and cancer, with cancers showing initial early enhancement and washout kinetics compared with infected collections that show no enhancement or peripheral enhancement with a plateau or persistent enhancement curves. When clinical and imaging findings are unchanged after treatment of mastitis and abscesses, a core needle biopsy should be performed.21,22
There are 2 categories of mastitis and breast abscess: lactational and nonpuerperal (all mastitis that occurs outside the lactational period). The World Health Organization definition of puerperal mastitis includes pain, local redness, warmth and swelling of the breast (usually unilateral), fever, and malaise.4 Concerning etiology, epithelial lesions in the nipple area caused by breastfeeding can allow pathogens to enter and cause infection. The most common microorganism is Staphylococcus aureus.4 Continued emptying of the breast is important, combined with early antibiotic therapy (dicloxacillin is often the first line; if the patient is penicillin allergic, use a macrolide such as clindamycin). If no improvement is seen in 48 to 72 hours, imaging should be performed.
In most cases, continuation of breastfeeding is possible. If mastitis has evolved into an abscess in a lactating woman, it can be aspirated under ultrasound guidance. Incision and drainage should be avoided unless the abscess persists after multiple aspiration attempts, it is large, or if the overlying skin is thin or otherwise appears nonviable.
Nonpuerperal mastitis includes peripheral, periductal, and idiopathic granulomatous mastitis (IGM). Peripheral mastitis behaves like infections/abscesses in other soft tissues, responds well to treatment (antibiotics and percutaneous drainage), and is less likely to recur than periductal mastitis and IGM.21,23
Periductal mastitis and abscess, also known as Zuska disease, has a pathogenesis distinct from other forms of mastitis. Squamous metaplasia of the usual cuboidal epithelium of the breast ducts leads to keratin plugging that can cause infection.23 Risk factors include obesity, smoking, and macromastia. The typical presentation of Zuska disease is a woman with a history of chronic smoking and/or a congenital cleft in the central nipple.23 Periareolar signs of inflammation (redness, swelling, warmth) may be accompanied by an abscess. These can recur and lead to chronic fistula formation, especially if there is a history of intervention (such as aspiration, incision, and drainage).
Treatment of Zuska disease includes symptom relief and antibiotics. If S aureus is present, infection with methicillin-resistant S aureus is likely, and treatment with clindamycin or amoxicillin/clavulanic acid is preferred. If abscess is present, aspiration (preferred, often under ultrasound guidance) or incision and drainage (if the skin is compromised) may be required. If disease is recurrent or associated with a chronically draining fistula, surgical intervention may be warranted, in which resolution requires removing the keratin-plugged ducts in and immediately below the central core of the nipple. Given the association between Zuska disease and smoking, cessation should be encouraged, although there is no guarantee that this will resolve the issue.23
Continue to: CASE 7 Patient with breast pain and swelling...
CASE 7 Patient with breast pain and swelling
A 39-year-old woman presents with left breast swelling and pain of 1 month’s duration. On examination, there is a 6-cm area of edema, induration, and erythema.
Granulomatous mastitis
A diagnostic mammogram and ultrasound demonstrated an ill-defined hypoechoic mass (FIGURE 7a). Ultrasound-guided biopsy was performed, which showed granulomatous mastitis, negative for fungus and acid-fast bacilli. The patient was treated with prednisone and gradually improved (FIGURE 7b).

Granulomatous mastitis (GM) is a rare benign inflammatory process, with etiologies that include fungal infections, tuberculosis, Wegener granulomatosis, sarcoidosis, and idiopathic causes. Imaging can be nonspecific and show variable features. Mammograms can appear normal or show asymmetry or mass and skin thickening. Ultrasound can show heterogeneous parenchyma, ill-defined hypoechoic collection, or a mass with margins that can be circumscribed or indistinct or with tubular extensions, with or without overlying skin thickening, fistulas, and reactive lymph nodes.24
In this clinical setting, the differential diagnosis includes infectious mastitis, inflammatory breast cancer, foreign body injection granulomas, and diabetic mastopathy. Treatment involves drainage and fluid culture if there is a collection on imaging. A core biopsy is performed if imaging demonstrates a solid mass or fluid culture is negative and symptoms persist or recur. Oral steroids represent the mainstay of treatment if a core biopsy shows GM. However, immunosuppressants, including methotrexate, and surgery are options if initial treatment is not helpful.25,26
Conclusion
Breast symptoms are common reasons for patient visits to obstetricians and gynecologists. With a good understanding of the various symptomatic breast diseases and conditions, and by having a close collaboration with radiologists and breast surgeons, clinicians can provide excellent care to these patients and thereby improve patient outcomes and satisfaction. ●
- Eberl MM, Phillips RL Jr, Lamberts H, et al. Characterizing breast symptoms in family practice. Ann Fam Med. 2008;6:528-533.
- Malherbe F, Nel D, Molabe H, et al. Palpable breast lumps: an age-based approach to evaluation and diagnosis. S Afr Fam Pract (2022). 2022;64:e1-e5.
- Expert Panel on Breast Imaging; Klein KA, Kocher M, Lourenco AP, et al. American College of Radiology ACR appropriateness criteria: palpable breast masses. Accessed February 15, 2023. https://acsearch.acr.org/docs/69495/Narrative/
- Stachs A, Stubert J, Reimer T, et al. Benign breast disease in women. Dtsch Arztebl Int. 2019;116:565574.
- Hines N, Slanetz PJ, Eisenberg RL. Cystic masses of the breast. AJR Am J Roentgenol. 2010;194:W122133.
- Berg WA. Reducing unnecessary biopsy and follow-up of benign cystic breast lesions. Radiology. 2020;295:52-53.
- Duman L, Gezer NS, Balcı P, et al. Differentiation between phyllodes tumors and fibroadenomas based on mammographic sonographic and MRI features. Breast Care. 2016;11:123-127.
- Lerwill MF, Lee AHS, Tan PH. Fibroepithelial tumours of the breast—a review. Virchows Arch. 2022;480:45-63.
- Vasei N, Shishegar A, Ghalkhani F, et al. Fat necrosis in the breast: a systematic review of clinical. Lipids Health Dis. 2019;18:139.
- Kerridge WD, Kryvenko ON, Thompson A, et al. Fat necrosis of the breast: a pictorial review of the mammographic, ultrasound, CT, and MRI findings with histopathologic correlation. Radiol Res Pract. 2015;2015:613139.
- Taboada JL, Stephens TW, Krishnamurthy S, et al. The many faces of fat necrosis in the breast. AJR Am J Roentgenol. 2009;192:815-825.
- Tan PH, Lai LM, Carrington EV, et al. Fat necrosis of the breast—a review. Breast. 2006;15:313-318.
- Holbrook AI. Breast pain, a common grievance: guidance to radiologists. AJR Am J Roentgenol. 2020;214:259-264.
- Expert Panel on Breast Imaging; Moy L, Heller SL, Bailey L, et al. ACR appropriateness criteria: palpable breast masses. J Am Coll Radiol. 2017;14:S203-S224.
- Chetlen AL, Kapoor MM, Watts MR. Mastalgia: imaging workup appropriateness. Acad Radiol. 2017;24:345-349.
- Arslan M, Kucukerdem HS, Can H, et al. Retrospective analysis of women with only mastalgia. J Breast Health. 2016;12:151-154.
- Fariselli G, Lepera P, Viganotti G, et al. Localized mastalgia as presenting symptom in breast cancer. Eur J Surg Oncol. 1988;14:213-215.
- Leddy R, Irshad A, Zerwas E, et al. Role of breast ultrasound and mammography in evaluating patients presenting with focal breast pain in the absence of a palpable lump. Breast J. 2013;19:582-589.
- Leung JW, Kornguth PJ, Gotway MB. Utility of targeted sonography in the evaluation of focal breast pain. J Ultrasound Med. 2002;21:521-526.
- Goyal A. Breast pain. BMJ Clin Evid. 2011; 2011:0812.
- Kasales CJ, Han B, Smith Jr JS, et al. Nonpuerperal mastitis and subareolar abscess of the breast. AJR Am J Roentgenol. 2014;202:W133-W139.
- Mahoney MC, Ingram AD. Breast emergencies: types, imaging features, and management. AJR Am J Roentgenol. 2014;202:W390-W399.
- Snider HC. Management of mastitis, abscess, and fistula. Surg Clin North Am. 2022;102:1103-1116.
- Oztekin PS, Durhan G, Kosar PN, et al. Imaging findings in patients with granulomatous mastitis. Iran J Radiol. 2016;13:e33900.
- Pluguez-Turull CW, Nanyes JE, Quintero CJ, et al. Idiopathic granulomatous mastitis: manifestations at multimodality imaging and pitfalls. Radiographics. 2018;38:330-356.
- Hovanessian-Larsen LJ, Peyvandi B, Klipfel N, et al. Granulomatous lobular mastitis: imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193:574-581.
- Eberl MM, Phillips RL Jr, Lamberts H, et al. Characterizing breast symptoms in family practice. Ann Fam Med. 2008;6:528-533.
- Malherbe F, Nel D, Molabe H, et al. Palpable breast lumps: an age-based approach to evaluation and diagnosis. S Afr Fam Pract (2022). 2022;64:e1-e5.
- Expert Panel on Breast Imaging; Klein KA, Kocher M, Lourenco AP, et al. American College of Radiology ACR appropriateness criteria: palpable breast masses. Accessed February 15, 2023. https://acsearch.acr.org/docs/69495/Narrative/
- Stachs A, Stubert J, Reimer T, et al. Benign breast disease in women. Dtsch Arztebl Int. 2019;116:565574.
- Hines N, Slanetz PJ, Eisenberg RL. Cystic masses of the breast. AJR Am J Roentgenol. 2010;194:W122133.
- Berg WA. Reducing unnecessary biopsy and follow-up of benign cystic breast lesions. Radiology. 2020;295:52-53.
- Duman L, Gezer NS, Balcı P, et al. Differentiation between phyllodes tumors and fibroadenomas based on mammographic sonographic and MRI features. Breast Care. 2016;11:123-127.
- Lerwill MF, Lee AHS, Tan PH. Fibroepithelial tumours of the breast—a review. Virchows Arch. 2022;480:45-63.
- Vasei N, Shishegar A, Ghalkhani F, et al. Fat necrosis in the breast: a systematic review of clinical. Lipids Health Dis. 2019;18:139.
- Kerridge WD, Kryvenko ON, Thompson A, et al. Fat necrosis of the breast: a pictorial review of the mammographic, ultrasound, CT, and MRI findings with histopathologic correlation. Radiol Res Pract. 2015;2015:613139.
- Taboada JL, Stephens TW, Krishnamurthy S, et al. The many faces of fat necrosis in the breast. AJR Am J Roentgenol. 2009;192:815-825.
- Tan PH, Lai LM, Carrington EV, et al. Fat necrosis of the breast—a review. Breast. 2006;15:313-318.
- Holbrook AI. Breast pain, a common grievance: guidance to radiologists. AJR Am J Roentgenol. 2020;214:259-264.
- Expert Panel on Breast Imaging; Moy L, Heller SL, Bailey L, et al. ACR appropriateness criteria: palpable breast masses. J Am Coll Radiol. 2017;14:S203-S224.
- Chetlen AL, Kapoor MM, Watts MR. Mastalgia: imaging workup appropriateness. Acad Radiol. 2017;24:345-349.
- Arslan M, Kucukerdem HS, Can H, et al. Retrospective analysis of women with only mastalgia. J Breast Health. 2016;12:151-154.
- Fariselli G, Lepera P, Viganotti G, et al. Localized mastalgia as presenting symptom in breast cancer. Eur J Surg Oncol. 1988;14:213-215.
- Leddy R, Irshad A, Zerwas E, et al. Role of breast ultrasound and mammography in evaluating patients presenting with focal breast pain in the absence of a palpable lump. Breast J. 2013;19:582-589.
- Leung JW, Kornguth PJ, Gotway MB. Utility of targeted sonography in the evaluation of focal breast pain. J Ultrasound Med. 2002;21:521-526.
- Goyal A. Breast pain. BMJ Clin Evid. 2011; 2011:0812.
- Kasales CJ, Han B, Smith Jr JS, et al. Nonpuerperal mastitis and subareolar abscess of the breast. AJR Am J Roentgenol. 2014;202:W133-W139.
- Mahoney MC, Ingram AD. Breast emergencies: types, imaging features, and management. AJR Am J Roentgenol. 2014;202:W390-W399.
- Snider HC. Management of mastitis, abscess, and fistula. Surg Clin North Am. 2022;102:1103-1116.
- Oztekin PS, Durhan G, Kosar PN, et al. Imaging findings in patients with granulomatous mastitis. Iran J Radiol. 2016;13:e33900.
- Pluguez-Turull CW, Nanyes JE, Quintero CJ, et al. Idiopathic granulomatous mastitis: manifestations at multimodality imaging and pitfalls. Radiographics. 2018;38:330-356.
- Hovanessian-Larsen LJ, Peyvandi B, Klipfel N, et al. Granulomatous lobular mastitis: imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193:574-581.
Telemedicine: Medicolegal aspects in ObGyn

Telemedicine (or telehealth) originated in the early 1900s, when radios were used to communicate medical advice to clinics aboard ships.1 According to the American Telemedicine Association, telemedicine is namely “the use of medical information exchanged from one site to another via electronic communications to improve a patient’s clinical health status.”2 These communications use 2-way video, email, smartphones, wireless tools, and other forms of telecommunications technology.
During the COVID-19 pandemic, many ObGyns—encouraged and advised by professional organizations—began providing telemedicine services.3 The first reported case of COVID-19 was in late 2019; the use of telemedicine was 38 times higher in February 2021 than in February 2020,4 illustrating how many physicians quickly moved to telemedicine practices.
CASE Dr. TM’s telemedicine dream
Before COVID-19, Dr. TM (an ObGyn practi-tioner) practiced in-person medicine in his home state. With the onset of the pandemic, Dr. TM struggled to switch to primarily seeing patients online (generally using Zoom or Facebook Live), with 1 day per week in the office for essential in-person visits.
After several months, however, Dr. TM’s routine became very efficient. He could see many more patients in a shorter time than with the former, in-person system. Therefore, as staff left his practice, Dr. TM did not replace them and also laid off others. Ultimately, the practice had 1 full-time records/insurance secretary who worked from home and 1 part-time nurse who helped with the in-person day and answered some patient inquiries by email. In part as an effort to add new patients, Dr. TM built an engaging website through which his current patients could receive medical information and new patients could sign up.
In late 2022, Dr. TM offered a $100 credit to any current patient who referred a friend or family member who then became a patient. This promotion was surprisingly effective and resulted in an influx of new patients. For example, Patient Z (a long-time patient) received 3 credits for referring her 3 sisters who lived out of state and became telepatients: Patient D, who lived 200 hundred miles away; Patient E, who lived 50 miles away in the adjoining state; and Patient F, who lived 150 miles away. Patient D contacted Dr. TM because she thought she was pregnant and wanted prenatal care, Patient E thought she might have a sexually transmitted infection (STI) and wanted treatment, and Patient F wanted general care and was inquiring about a medical abortion. Dr. TM agreed to treat Patient D but required 1 in-person visit. After 1 brief telemedicine session each with Patients E and F, Dr. TM wrote prescriptions for them.
By 2023, Dr. TM was enthusiastic about telemedicine as a professional practice. However, problems would ensue.
Dos and don’ts of telemedicine2
- Do take the initiative and inform patients of the availability of telemedicine/telehealth services
- Do use the services of medical malpractice insurance companies with regard to telemedicine
- Do integrate telemedicine into practice protocols and account for their limitations
- Don’t assume there are blanket exemptions or waivers in the states where your patients are located
Medical considerations
Telemedicine is endorsed by the American College of Obstetricians and Gynecologists (ACOG) as a vehicle for delivering prenatal and postpartum care.5 This represents an effort to reduce maternal and neonatal morbidity and mortality,5 as well as expand access to care and address the deficit in primary care providers and services, especially in rural and underserved populations.5,6 For obstetrics, prenatal care is designed to optimize pregnancy, childbirth, and postpartum care, with a focus on nutrition and genetic consultation and patient education on pregnancy, childbearing, breastfeeding, and newborn care.7
Benefits of telemedicine include its convenience for patients and providers, its efficiency and lower costs for providers (and hopefully patients, as well), and the potential improved access to care for patients.8 It is estimated that if a woman inititates obstetric care at 6 weeks, over the course of the 40-week gestation period, 15 prenatal visits will occur.9 Ultimately, the number of visits is determined based on the specifics of the pregnancy. With telemedicine, clinicians can provide those consultations, and information related to: ultrasonography, fetal echocardiography, and postpartum care services remotely.10 Using telemedicine may reduce missed visits, and remote monitoring may improve the quality of care.11
Barriers to telemedicine care include technical limitations, time constraints, and patient concerns of telehealth (visits). Technical limitations include the lack of a high speed internet connection and/or a smart device and the initial technical set-up–related problems,12 which affect providers as well as patients. Time constraints primarly refer to the ObGyn practice’s lack of time to establish telehealth services.13 Other challenges include integrating translation services, billing-related problems,10 and reimbursement and licensing barriers.14
Before the COVID-19 pandemic, obstetrics led the way in telemedicine with the development of the OB Nest model. Designed to replace in-person obstetrics care visits with telehealth,15 it includes home management tools such as blood pressure cuffs, cardiotocography, scales for weight checks, and Doppler ultrasounds.10 Patients can be instructed to measure fundal height and receive medications by mail. Anesthesia consultation can occur via this venue by having the patient complete a questionnaire prior to arriving at the labor and delivery unit.16
Legal considerations
With the COVID-19 pandemic, temporary changes were made to encourage the rapid adoption of telemedicine, including changes to licensing laws, certain prescription requirements, Health Insurance Portability and Accountability Act (HIPAA) privacy-security regulations, and reimbursement rules that required in-person visits. Thus, many ObGyns started using telemedicine during this rarified period, in which the rules appeared to be few and far between, with limited enforcement of the law and professional obligations.17 However, now that many of the legal rules that were suspended or ignored have been (or are being) reimposed and enforced, it is important for providers to become familiar with the legal issues involved in practicing telemedicine.
First, where is the patient? When discussing the legal issues of telemedicine, it is important to remember that many legal rules for medical care (ie, liability, informed consent, and licensing) vary from state to state. If the patient resides in a different state (“foreign” state) from the physician’s practice location (the physician’s “home” state), the care is considered delivered in the state where the patient is located. Thus, the patient’s location generally establishes the law covering the telemedicine transaction. In the following discussion, the rules refer to the law and professional obligations, with commentary on some key legal issues that are relevant to ObGyn telemedicine.
Continue to: Reinforcing the rules...
Reinforcing the rules
Licensing
During the height of the COVID-19 pandemic, the federal government and almost all states temporarily modified the licensing requirement to allow telemedicine based on an existing medical license in any state—disregarding the “where is the patient” rule. As those rules begin to lapse or change with the official end of the pandemic declared by President Biden as May 2023,17 the rules under which a physician began telemedicine interstate practice in 2020 also may be changing.
Simply put, “The same standards for licensure apply to health care providers regardless of whether care is delivered in-person or virtually through telehealth services.”18 When a physician is engaged in telemedicine treatment of a patient in the physician’s home state, there is generally no licensing issue. Telemedicine generally does not require a separate specific license.19 However, when the patient is in another state (a “foreign” state), there can be a substantial licensing issue.20 Ordinarily, to provide that treatment, the physician must, in some manner, be approved to practice in the patient’s state. That may occur, for example, in the following ways: (1) the physician may hold an additional regular license in the patient’s state, which allows practice there, or (2) the physician may have received permission for “temporary practice” in another state.
Many states (often adjoining states) have formal agreements with other states that allow telemedicine practice by providers in each other’s states. There also are “compacts”, or agreements that enable providers in any of the participating states to practice in the other associated states without a separate license.18 Although several websites provide information about compact licensing and the like, clinicians should not rely on simple lists or maps. Individual states may have special provisions about applying their laws to out-of-state “compact” physicians. In addition, under the Interstate Medical Licensure Compact, “physicians have to pay licensing fees and satisfy the requirements of each medical board in the states where they wish to practice.”21
Consequences. Practicing telemedicine with a patient in a state where the physician does not have a license is generally a crime. Furthermore, it may be the basis for license discipline in the physician’s home state and result in a report to the National Practi-tioner Databank.22 In addition, reimbursement often depends on the practitioner being licensed, and the absence of a license may be a basis for denying payment for services.23 Finally, malpractice insurance generally is limited to licensed practice. Thus, the insurer may decline to defend the unlicensed clinician against a malpractice claim or pay any damages.
Prescribing privileges
Prescribing privileges usually are connected to licensing, so as the rules for licensing change postpandemic, so do the rules for prescribing. In most cases, the physician must have a license in the state where care is given to prescribe medication—which in telemedicine, as noted, typically means the state where the patient is located. Exceptions vary by state, but in general, if a physician does not have a license to provide care, the physician is unlikely to be authorized to prescribe medication.24 Failure to abide by the applicable state rules may result in civil and even criminal liability for illegal prescribing activity.
In addition, the US Food and Drug Administration (FDA) and Drug Enforcement Administration (DEA, which enforces laws concerning controlled substances) also regulate the prescription and sale of pharmaceuticals.25 There are state and federal limits on the ability of clinicians to order controlled substances without an in-person visit. The Ryan Haight Online Pharmacy Consumer Protection Act, for example, sets limits on controlled substance prescriptions without an in-person examination.26 Federal law was modified due to COVID-19 to permit prescribing of many controlled substances by telemedicine if there is synchronous audio and visual examination of the patient. Physicians who write such prescriptions also are required to have a DEA registration in the patient’s state. This is an essential consideration for physicians considering interstate telemedicine practice.27
HIPAA and privacy
Governments waived some of the legal requirements related to health information during the pandemic, but those waivers either have expired or will do so soon. Federal and state laws regarding privacy and security—notably including HIPAA—apply to telemedicine and are of particular concern given the considerable amount of communication of protected health information with telemedicine.
HIPAA security rules essentially require making sure health information cannot be hacked or intercepted. Audio-only telemedicine by landline (not cell) is acceptable under the security rules, but almost all other remote communication requires secure communications.28
Clinicians also need to adhere to the more usual HIPAA privacy rules when practicingtelehealth. State laws protecting patient privacy vary and may be more stringent than HIPAA, so clinicians also must know the requirements in any state where they practice—whether in office or telemedicine.29
Making sure telemedicine practices are consistent with these security and privacy rules often requires particular technical expertise that is outside the realm of most practicing clinicians. However, without modification, the pre-telemedicine technology of many medical offices likely is insufficient for the full range of telemedicine services.30
Reimbursement and fraud
Before COVID-19, Medicare and Medicaid reimbursement for telemedicine was limited. Government decisions to substantially broaden those reimbursement rules (at least temporarily) provided a substantial boost to telemedicine early in the pandemic.23 Federal regulations and statutes also expanded telemedicine reimbursement for various services. Some will end shortly after the health emergency, and others will be permanent. Parts of that will not be sorted out for several years, so it will likely be a changing landscape for reimbursement.
Continue to: Rules that are evolving...
Rules that are evolving
Informed consent
The ethical and legal obligations to obtain informed consent are present in telemedicineas well as in-person care, with the same basic requirements regarding risks, benefits, alternative care, etc.32 However, with telemedicine, information related to remote care should be included and is outlined in TABLE 1.

Certain states may have somewhat unique informed consent requirements—especially for reproductive care, including abortion.34 Therefore, it is important for clinicians to ensure their consent process and forms comply with any legal jurisdiction in which a patient is located.
Medical malpractice
The basics of medical malpractice (or negligence) are the same in telemedicine as in in-person care: duty, breach of duty, and injury caused by the breach. That is, there may be liability when a medical professional breaches the duty of care, causing the patient’s injury. The physician’s duty is defined by the quality of care that the profession (specialty) accepts as reasonably good. This is defined by the opinions of physicians within the specialty and formal statements from professional organizations, including ACOG.3
Maintaining the standard of care and quality. The use of telemedicine is not an excuse to lower the quality of health care. There are some circumstances for which it is medically better to have an in-person visit. In these instances, the provider should recommend the appropriate care, even if telemedicine would be more convenient for the provider and staff.35
If the patient insists and telemedicine might result in less than optimal care, the reasons for using a remote visit should be clearly documented contemporaneously with the decision. Furthermore, when the limitations of being unable to physically examine the patient result in less information than is needed for the patient’s care, the provider must find alternatives to make up for the information gap.11,36 It also may be necessary to inform patients about how to maximize telemedicine care.37 At the beginning of telemedicine care the provider should include information about the nature and limits of telehealth, and the patient’s responsibilities. (See TABLE 1) Throughout treatment of the patient, that information should be updated by the provider. That, of course, is particularly important for patients who have not previously used telemedice services.
Malpractice rules vary by state. Many states have special rules regarding malpractice cases. These differences in malpractice standards and regulations “can be problematic for physicians who use telemedicine services to provide care outside the state in which they practice.”38 Caps on noneconomic damages are an example. Those state rules would apply to telemedicine in the patient’s state.
Malpractice insurance
Malpractice insurance now commonly includes telemedicine legally practiced within the physician’s home state. Practitioners who treat patients in foreign states should carefully examine their malpractice insurance policies to confirm that the coverage extends to practice in those states.39 Malpractice carriers may require notification by a covered physician who routinely provides services to patients in another state.3
Keep in mind, malpractice insurance generally does not cover the practice of medicine that is illegal. Practicing telemedicine in a foreign state, where the physician or other provider does not have a license and where that state does not otherwise permit the practice, is illegal. Most likely, the physician’s malpractice insurance will not cover claims that arise from this illegal practice in a foreign state or provide defense for malpractice claims, including frivolous lawsuits. Thus, the physician will pay out of pocket for the costs of a defense attorney.
Telemedicine treatment of minors
Children and adolescents present special legal issues for ObGyn care, which may become more complicated with telemedicine. Historically, parents are responsible for minors (those aged <18 years): they consent to medical treatment, are responsible for paying for it, and have the right to receive information about treatment.
Over the years, though, many states have made exceptions to these principles, especially with regard to contraception and treatment of sexually transmitted diseases.40 For abortion, in particular, there is considerable variation among the states in parental consent and notification.41 The Supreme Court’s decision in Dobbs v Jackson Women’s Health42 may (depending on the state) be followed with more stringent limitations on adolescent consent to abortions, including medical abortions.43
Use of telehealth does not change any obligations regarding adolescent consent or parental notification. Because those differ considerably among states, it is important for all practitioners to know their states’ requirements and keep reasonably complete records demonstrating their compliance with state law.
Abortion
The most heated current controversy about telemedicine involves abortion—specifically medical abortion, which is the combination of mifepristone and misoprostol.44,45 The FDA approved the combination in 2000. Almost immediately, many states required in-person visits with a certified clinician to receive a prescription for mifepristone and misoprostol, and eventually, the FDA adopted similar requirements.46 However, during the pandemic from 2021 to 2022, the FDA permitted telemedicine prescriptions. Several states still require in-person physician visits, although the constitutionality of those requirements has not been established.47
With the Supreme Court’s decision in Dobbs v Jackson Women’s Health in 2022,42 disagreements have ensued about the degree to which states may regulate the prescription of FDA-approved medical abortion drugs. Thorny constitutional issues exist in the plans of both abortion opponents and proponents in the battle over medical abortion in antiabortion states. It may be that federal drug law preempts state laws limiting access to FDA-approved drugs. On the other hand, it may be that states can make it a crime within the state to possess or provide abortion-inducing drugs. Courts will probably take years to resolve the many tangled legal questions.48
Thus, while the pandemic telemedicine rules may have advanced access to abortion,34 there may be some pending downsides.49 States that prohibit abortion will likely include prohibitions on medical abortions. In addition, they may prohibit anyone in the state (including pharmacies) from selling, possessing, or obtaining any drug used for causing or inducing an abortion.50 If, for constitutional reasons, they cannot press criminal charges or undertake licensing discipline for prescribing abortion, some states will likely withdraw from telehealth licensing compacts to avoid out-of-state prescriptions. This area of telemedicine has considerable uncertainty.
Continue to: CASE Conclusion...
CASE Conclusion
Patient concerns come to the fore
By 2023, Dr. TM started receiving bad news. Patient D called complaining that after following the advice on the website, she suffered a severe reaction and had to be rushed to an emergency department. Patient E (who had only 1 in-office visit early in her pregnancy) notified the office that she developed very high blood pressure that resulted in severe placental abruption, requiring emergency care and resulting in the loss of the fetus. Patient F complained that someone hacked the TikTok direct message communication with Dr. TM and tried to “blackmail” or harass her.
Discussion. Patients D, E, and F represent potential problems of telemedicine practice. Patient D was injured because she relied on her doctor’s website (to which Dr. TM directed patients). It contained an error that caused an injury. A doctor-patient relationship existed, and bad medical advice likely caused the injury. Physicians providing advice online must ensure the advice is correct and kept current.
Patient E demonstrates the importance of monitoring patients remotely (blood pressure transmitted to the office) or with periodic in-office visits. It is not clear whether she was a no-show for office visits (and whether the office followed up on any missed appointments) or if such visits were never scheduled. Liability for failure to monitor adequately is a possibility.
Patient F’s seemingly minor complaint could be a potential problem. Dr. TM used an insecure mode of communication. Although some HIPAA security regulations were modified or suspended during the pandemic, using such an unsecure platform is problematic, especially if temporary HIPAA rules expired. The outcome of the complaint is in doubt.

(See TABLE 2 for additional comments on patients D, E, and F.)
Out-of-state practice
Dr. TM treated 3 out-of-state residents (D, E, and F) via telemedicine. Recently Dr. TM received a complaint from the State Medical Licensure Board for practicing medicine without a license (Patient D), followed by similar charges from Patient E’s and Patient F’s state licensing boards. He has received a licensing inquiry from his home state board about those claims of illegal practice in other states and incompetent treatment.
Patient D’s pregnancy did not go well. The 1 in-person visit did not occur and she has filed a malpractice suit against Dr. TM. Patient E is threatening a malpractice case because the STI was not appropriately diagnosed and had advanced before another physician treated it.
In addition, a private citizen in Patient F’s state has filed suit against Dr. TM for abetting an illegal abortion (for Patient F).
Discussion. Patients D, E, and F illustrate the risk of even incidental out-of-state practice. The medical board inquiries arose from anonymous tips to all 4 states reporting Dr. TM was “practicing medicine without a license.” Patient E’s home state did have a licensing compact with the adjoining state (ie, Dr. TM’s home state). However, it required physicians to register and file an annual report, which Dr. TM had not done. The other 2 states did not have compacts with Dr. TM’s home state. Thus, he was illegally practicing medicine and would be subject to penalties. His home state also might impose license discipline based on his illegal practice in other states.
Continue to: What’s the verdict?...
What’s the verdict?
Dr. TM’s malpractice carrier is refusing to defend the claims of medical malpractice threatened by Patients D, E, and F. The company first notes that the terms of the malpractice policy specifically exclude the illegal practice of medicine. Furthermore, when a physician legally practices in another state, the policy requires a written notice to the insurance carrier of such practice. Dr. TM will likely have to engage and pay for a malpractice attorney for these cases. Because the claims are filed in 3 different states, more than a home-state attorney will likely be involved in the defense of these cases. Dr. TM will need to pay the attorneys and any damages from a settlement or trial.
Malpractice claims. Patient D claims that the doctor essentially abandoned her by never reaching out to her or arranging an in-person visit. Dr. TM claims the patient was responsible for scheduling the in-person visit. Patient E claims it was malpractice not to determine the specific nature of the STI and to do follow-up testing to determine that it was cured. All patients claim there was no genuine informed consent to the telemedicine. An attorney has warned Dr. TM that it is “not going to look good to the jury” that he was practicing without a license in the state and suggests he settle the cases quickly by paying damages.
Abortion-related claims. Patient F presents a different set of problems. Dr. TM’s home state is “proabortion.” Patient F’s home state is strongly “antiabortion,” making it a felony to participate in, assist, or facilitate an abortion (including medical abortion). Criminal charges have been filed against Dr. TM for the illegal practice of medicine, for aiding and facilitating an abortion, and for failure to notify a parent that a minor is seeking an abortion. For now, Dr. TM’s state is refusing to extradite on the abortion charge. Still, the patient’s state insists that it do so on the illegal practice of medicine charges and new charges of insurance fraud and failure to report suspected sexual abuse of a child. (Under the patient’s state law, anyone having sex with Patient F would have engaged in sexual abuse or “statutory rape,” so the state insists that the fact she was pregnant proves someone had sex with her.)
Patient F’s state also has a statute that allows private citizens to file civil claims against anyone procuring or assisting with an abortion (a successful private citizen can receive a minimum of $10,000 from the defendant). Several citizens from the patient’s state have already filed claims against Dr. TM in his state courts. Only one of them, probably the first to file, could succeed. Courts in the state have issued subpoenas and ordered Dr. TM to appear and reply to the civil suits. If he does not respond, there will be a default judgment.
Dr. TM’s attorney tells him that these lawsuits will not settle and will take a long time to defend and resolve. That will be expensive.
Billing and fraud. Dr. TM’s office recently received a series of notices from private health insurers stating they are investigating previously made payments as being fraudulent (unlicensed). They will not pay any new claims pending the investigation. On behalf of Medicare-Medicaid and other federal programs, the US Attorney’s office has notified Dr. TM that it has opened an investigation into fraudulent federal payments. F’s home state also is filing a (criminal) insurance fraud case, although the basis for it is unclear. (Dr. TM’s attorney believes it might be to increase pressure on the physician’s state to extradite Dr. TM for Patient F’s case.)
In addition, a disgruntled former employee of Dr. TM has filed a federal FCA case against him for filing inflated claims with various federally funded programs. The employee also made whistleblower calls to insurance companies and some state-funded medical programs. A forensic accounting investigation by Dr. TM’s accountant confirmed a pattern of very sloppy records and recurring billing for televisits that did not occur. Dr. TM believes that this was the act of one of the temporary assistants he hired in a pinch, who did not understand the system and just guessed when filing some insurance claims.
During the investigation, the federal and state attorneys are looking into a possible violation of state and federal Anti-Kickback Statutes. This is based on the original offer of a $100 credit for referrals to Dr. TM’s telemedicine practice.
The attorneys are concerned that other legal problems may present themselves. They are thoroughly reviewing Dr. TM’s practice and making several critical but somewhat modest changes to his practice. They also have insisted that Dr. TM have appropriate staff to handle the details of the practice and billing.
Conclusions
Telemedicine presents notable legal challenges to medical practice. As the pandemic status ends, ObGyn physicians practicing telemedicine need to be aware of the rules and how they are changing. For those physicians who want to continue or start a telemedicine practice, securing legal and technical support to ensure your operations are inline with the legal requirements can minimize any risk of legal troubles in the future. ●
A physician in State A, where abortion is legal, has a telemedicine patient in State B, where it is illegal to assist, provide, or procure an abortion. If the physician prescribes a medical abortion, he would violate the law of State B by using telemedicine to help the patient (located in State B) obtain an abortion. This could result in criminal charges against the prescribing physician.
- Board on Health Care Services; Institute of Medicine. The Role of Telehealth in an Evolving Health Care Environment: Workshop Summary. National Academies Press: 2012. https://www.ncbi.nlm.nih.gov/books/NBK207145/. Accessed March 30, 2023.
- Bruhn HK. Telemedicine: dos and don’ts to mitigate liability risk. J APPOS. 2020;24:195-196. doi:10.1016/j.jaapos. 2020.07.002
- Implementing telehealth in practice: ACOG Committee Opinion Summary, number 798. Obstet Gynecol. 2020; 2135:493-494. doi:10.1097/AOG.0000000000003672
- Bestsennyy O, Gilbert G, Harris A, et al. Telehealth: a quarter-trillion-dollar post-COVID-19 reality? McKinsey & Company. July 9, 2021. Accessed March 2, 2023. https://www.mckinsey.com/industries/healthcare/our-insights /telehealth-a-quarter-trillion-dollar-post-covid-19-reality
- Stanley AY, Wallace JB. Telehealth to improve perinatal care access. MCN Am J Matern Child Nurs. 2022;47:281-287. doi: 10.1097/NMC.0000000000000841
- Warshaw R. Health disparities affect millions in rural US communities. Association of American Medical Colleges. Published October 31, 2017. Accessed March 31, 2023. https://www.aamc.org/news-insights/health-disparities -affect-millions-rural-us-communities
- Almuslin H, AlDossary S. Models of incorporating telehealth into obstetric care during the COVID-19 pandemic, its benefits and barriers: a scoping review. Telemed J E Health. 2022;28:24-38. doi:10.1089/tmj.2020.0553
- Gold AE, Gilbert A, McMichael BJ. Socially distant health care. Tul L Rev. 2021;96:423-468. https://scholarship .law.ua.edu/cgi/viewcontent.cgi?article=1713&context =fac_articles. Accessed March 4, 2023.
- Zolotor AJ, Carlough MC. Update on prenatal care. Am Fam Physician. 2014;89:199-208.
- Odibo IN, Wendel PJ, Magann EF. Telemedicine in obstetrics. Clin Obstet Gynecol. 2013;56:422-433. doi:10.1097/ GRF.0b013e318290fef0
- Shmerling A, Hoss M, Malam N, et al. Prenatal care via telehealth. Prim Care. 2022;49:609-619. doi:10.1016/j. pop.2022.05.002
- Madden N, Emeruwa UN, Friedman AM, et al. Telehealth uptake into prenatal care and provider attitudes during COVID-19 pandemic in New York City: a quantitative and qualitative analysis. Am J Perinatol. 2020;37:1005-1014. doi:10.1055/s-0040-1712939
- Dosaj A, Thiyagarajan D, Ter Haar C, et al. Rapid implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2020;27:116-120. doi:10.1089/ tmj.2020.0219
- Lurie N, Carr B. The role of telehealth in the medical response to disasters. JAMA Intern Med. 2018;187:745-746. doi: 10.1001/jamainternmed.2018.1314
- Tobah YSB, LeBlanc A, Branda E, et al. Randomized comparison of a reduced-visit prenatal care model enhanced with remote monitoring. Am J Obstet Gynecol. 2019;221:638-e1-638.e8. doi:10.1016/j.ajog.2019.06.034
- Vivanti AJ, Deruelle P, Piccone O, et al. Follow-up for pregnant women during the COVID-19 pandemic: French national authority for health recommendations. J Gynecol Obstet Hum Reprod. 2020;49:101804. doi:10.1016/j. jogoh.2020.101804
- Ellimoottil C. Takeaways from 2 key studies on interstate telehealth use among Medicare fee-for-service beneficiaries. JAMA Health Forum. 2022;3:e223020-E223020. doi:10.1001/ jamahealthforum.2022.3020
- Harris J, Hartnett T, Hoagland GW, et al. What eliminating barriers to interstate telehealth taught us during the pandemic. Bipartisan Policy Center. Published November 2021. Accessed March 9, 2023. https://bipartisanpolicy .org/download/?file=/wp-content/uploads/2021/11/BPC -Health-Licensure-Brief_WEB.pdf.
- Center for Connected Health Policy. Cross-state licensing. Accessed February 21, 2023. https://www.cchpca.org/topic /cross-state-licensing-professional-requirements.
- US Department of Health & Human Services. Telehealth. Getting started with licensure. Published February 3, 2023. Accessed February 27, 2023. https://telehealth.hhs.gov /licensure/getting-started-licensure/
- US Department of Health & Human Services. Telehealth. Licensure. Accessed February 27, 2023. https://telehealth .hhs.gov/licensure
- US Department of Health & Human Services. National Practitioner Data Bank (NPDB) code lists. Published December 2022. Accessed March 9, 2023. https://www.npdb .hrsa.gov/software/CodeLists.pdf
- American College of Obstetricians and Gynecologists. COVID-19 FAQs for obstetrician-gynecologists, telehealth. 2020. Accessed March 5, 2023. https://www.acog.org /clinical-information/physician-faqs/covid-19-faqs-for -ob-gyns-telehealth
- Gorman RK. Prescribing medication through the practice of telemedicine: a comparative analysis of federal and state online prescribing policies, and policy considerations for the future. S Cal Interdisc Law J. 2020;30:739-769. https://gould .usc.edu/why/students/orgs/ilj/assets/docs/30-3-Gorman. pdf. Accessed March 10, 2023.
- Farringer DR. A telehealth explosion: using lessons from the pandemic to shape the future of telehealth regulation. Tex A&M Law Rev. 2021;9:1-47. https://scholarship.law.tamu. edu/cgi/viewcontent.cgi?article=1232&context=lawreview. Accessed February 28, 2023.
- Sterba KR, Johnson EE, Douglas E, et al. Implementation of a women’s reproductive behavioral health telemedicine program: a qualitative study of barriers and facilitators in obstetric and pediatric clinics. BMC Pregnancy Childbirth. 2023;23:167, 1-10. doi:10.1186/s12884-023-05463-2.
- US Department of Justice. COVID-19 FAQ (telemedicine). https://www.deadiversion.usdoj.gov/faq/coronavirus_faq .htm#TELE_FAQ2. Accessed March 13, 2023.
- US Department of Health & Human Services. Guidance on how the HIPAA rules permit covered health care providers and health plans to use remote communication technologies for audio-only telehealth. Published June 13, 2022. Accessed February 22, 2023. https://www.hhs.gov/hipaa/for-professionals/privacy /guidance/hipaa-audio-telehealth/index.html.
- Gray JME. HIPAA, telehealth, and the treatment of mental illness in a post-COVID world. Okla City Uni Law Rev. 2021;46:1-26. https://law.okcu.edu/wp-content /uploads/2022/04/J-Michael-E-Gray-HIPAA-Telehealth -and-Treament.pdf. Accessed March 9, 2023.
- Kurzweil C. Telemental health care and data privacy: current HIPAA privacy pitfalls and a proposed solution. Ann Health L Adv Dir. 2022;31:165.
- US Department of Health & Human Services and US Department of Justice. Health care fraud and abuse control program FY 2020: annual report. July 2021. Accessed March 9, 2023. https://oig.hhs.gov/publications/docs/hcfac /FY2020-hcfac.pdf
- Copeland KB. Telemedicine scams. Iowa Law Rev. 2022: 108:69-126. https://ilr.law.uiowa.edu/sites/ilr.law.uiowa.edu /files/2023-01/A2_Copeland.pdf. Accessed March 10, 2023.
- Solimini R, Busardò FP, Gibelli F, et al. Ethical and legal challenges of telemedicine in the era of the COVID-19 pandemic. Medicina (Kaunas). 2021;57:13141324. doi:10.3390/medicina57121314
- Reed A. COVID: a silver linings playbook. mobilizing pandemic era success stories to advance reproductive justice. Berkeley J Gender Law Justice. 2022;37:221-266. https://lawcat.berkeley.edu/record/1237158/files/16%20 Reed_final.pdf. Accessed March 11, 2023.
- Women’s Preventive Services Initiative and The American College of Obstetricians and Gynecologists. FAQ for telehealth services. Accessed March 2, 2023. https://www .womenspreventivehealth.org/wp-content/uploads/WPSI -Telehealth-FAQ.pdf
- Warren L, Chen KT. Telehealth apps in ObGyn practice. OBG Manag. 2022;34:46-47. doi:10.12788/obgm.0178
- American College of Obstetricians and Gynecologists. 10 telehealth tips for an Ob-Gyn visit. 2020. Accessed March 2, 2023. https://www.acog.org/womens-health /infographics/10-telehealth-tips-for-an-ob-gyn-visit
- Wolf TD. Telemedicine and malpractice: creating uniformity at the national level. Wm Mary Law Rev. 2019;61:15051536. https://scholarship.law.wm.edu/cgi/viewcontent.cgi ?article=3862&context=wmlr. Accessed March 11, 2023.
- Cahan E. Lawsuits, reimbursement, and liability insurance— facing the realities of a post-Roe era. JAMA. 2022;328:515517. doi:10.1001/jama.2022.9193
- Heinrich L, Hernandez AK, Laurie AR. Telehealth considerations for the adolescent patient. Prim Care. 2022;49:597-607. doi:10.1016/j.pop.2022.04.006
- Guttmacher Institute. An overview of consent to reproductive health services by young people. Published March 1, 2023. Accessed April 1, 2023. https://www.guttmacher.org /state-policy/explore/overview-minors-consent-law.
- Dobbs v. Jackson Women’s Health. No. 19–1392. June 24, 2022. Accessed April 1, 2023. https://www.supremecourt .gov/opinions/21pdf/19-1392_6j37.pdf
- Lindgren Y. Dobbs v. Jackson Women’s Health and the post-Roe landscape. J Am Acad Matrimonial Law. 2022;35:235283. https://www.aaml.org/wp-content/uploads/MAT110-1 .pdf. Accessed March 11, 2023.
- Mohiuddin H. The use of telemedicine during a pandemic to provide access to medication abortion. Hous J Health Law Policy. 2021;21:483-525. https://houstonhealthlaw. scholasticahq.com/article/34611.pdf. Accessed March 10, 2023.
- Rebouché R. The public health turn in reproductive rights. Wash & Lee Law Rev. 2021;78:1355-1432. https:// scholarlycommons.law.wlu.edu/cgi/viewcontent .cgi?article=4743&context=wlulr. Accessed March 10, 2023.
- Fliegel R. Access to medication abortion: now more important than ever. Am J Law Med. 2022;48:286-304. doi:10.1017/amj.2022.24
- Guttmacher Institute. Medication abortion. March 1, 2023. Accessed April 1, 2023 https://www.guttmacher.org /state-policy/explore/medication-abortion#:~:text=In%20 January%202023%2C%20the%20FDA,order%20to%20 dispense%20the%20pills
- Cohen DS, Donley G, Rebouché R. The new abortion battleground. Columbia Law Rev. 2023;123:1-100. https:// columbialawreview.org/content/the-new-abortion -battleground/. Accessed March 1, 2023.
- Hunt SA. Call me, beep me, if you want to reach me: utilizing telemedicine to expand abortion access. Vanderbilt Law Rev. 2023;76:323-359. Accessed March 10, 2023. https:// vanderbiltlawreview.org/lawreview/wp-content/uploads /sites/278/2023/01/Call-Me-Beep-Me-If-You-Want-toReach-Me-Utilizing-Telemedicine-to-Expand-AbortionAccess.pdf
- Gleckel JA, Wulkan SL. Abortion and telemedicine: looking beyond COVID-19 and the shadow docket. UC Davis Law Rev Online. 2020;54:105-121. https://lawreview.law.ucdavis. edu/online/54/files/54-online-Gleckel_Wulkan.pdf. Accessed April 1, 2023.

Telemedicine (or telehealth) originated in the early 1900s, when radios were used to communicate medical advice to clinics aboard ships.1 According to the American Telemedicine Association, telemedicine is namely “the use of medical information exchanged from one site to another via electronic communications to improve a patient’s clinical health status.”2 These communications use 2-way video, email, smartphones, wireless tools, and other forms of telecommunications technology.
During the COVID-19 pandemic, many ObGyns—encouraged and advised by professional organizations—began providing telemedicine services.3 The first reported case of COVID-19 was in late 2019; the use of telemedicine was 38 times higher in February 2021 than in February 2020,4 illustrating how many physicians quickly moved to telemedicine practices.
CASE Dr. TM’s telemedicine dream
Before COVID-19, Dr. TM (an ObGyn practi-tioner) practiced in-person medicine in his home state. With the onset of the pandemic, Dr. TM struggled to switch to primarily seeing patients online (generally using Zoom or Facebook Live), with 1 day per week in the office for essential in-person visits.
After several months, however, Dr. TM’s routine became very efficient. He could see many more patients in a shorter time than with the former, in-person system. Therefore, as staff left his practice, Dr. TM did not replace them and also laid off others. Ultimately, the practice had 1 full-time records/insurance secretary who worked from home and 1 part-time nurse who helped with the in-person day and answered some patient inquiries by email. In part as an effort to add new patients, Dr. TM built an engaging website through which his current patients could receive medical information and new patients could sign up.
In late 2022, Dr. TM offered a $100 credit to any current patient who referred a friend or family member who then became a patient. This promotion was surprisingly effective and resulted in an influx of new patients. For example, Patient Z (a long-time patient) received 3 credits for referring her 3 sisters who lived out of state and became telepatients: Patient D, who lived 200 hundred miles away; Patient E, who lived 50 miles away in the adjoining state; and Patient F, who lived 150 miles away. Patient D contacted Dr. TM because she thought she was pregnant and wanted prenatal care, Patient E thought she might have a sexually transmitted infection (STI) and wanted treatment, and Patient F wanted general care and was inquiring about a medical abortion. Dr. TM agreed to treat Patient D but required 1 in-person visit. After 1 brief telemedicine session each with Patients E and F, Dr. TM wrote prescriptions for them.
By 2023, Dr. TM was enthusiastic about telemedicine as a professional practice. However, problems would ensue.
Dos and don’ts of telemedicine2
- Do take the initiative and inform patients of the availability of telemedicine/telehealth services
- Do use the services of medical malpractice insurance companies with regard to telemedicine
- Do integrate telemedicine into practice protocols and account for their limitations
- Don’t assume there are blanket exemptions or waivers in the states where your patients are located
Medical considerations
Telemedicine is endorsed by the American College of Obstetricians and Gynecologists (ACOG) as a vehicle for delivering prenatal and postpartum care.5 This represents an effort to reduce maternal and neonatal morbidity and mortality,5 as well as expand access to care and address the deficit in primary care providers and services, especially in rural and underserved populations.5,6 For obstetrics, prenatal care is designed to optimize pregnancy, childbirth, and postpartum care, with a focus on nutrition and genetic consultation and patient education on pregnancy, childbearing, breastfeeding, and newborn care.7
Benefits of telemedicine include its convenience for patients and providers, its efficiency and lower costs for providers (and hopefully patients, as well), and the potential improved access to care for patients.8 It is estimated that if a woman inititates obstetric care at 6 weeks, over the course of the 40-week gestation period, 15 prenatal visits will occur.9 Ultimately, the number of visits is determined based on the specifics of the pregnancy. With telemedicine, clinicians can provide those consultations, and information related to: ultrasonography, fetal echocardiography, and postpartum care services remotely.10 Using telemedicine may reduce missed visits, and remote monitoring may improve the quality of care.11
Barriers to telemedicine care include technical limitations, time constraints, and patient concerns of telehealth (visits). Technical limitations include the lack of a high speed internet connection and/or a smart device and the initial technical set-up–related problems,12 which affect providers as well as patients. Time constraints primarly refer to the ObGyn practice’s lack of time to establish telehealth services.13 Other challenges include integrating translation services, billing-related problems,10 and reimbursement and licensing barriers.14
Before the COVID-19 pandemic, obstetrics led the way in telemedicine with the development of the OB Nest model. Designed to replace in-person obstetrics care visits with telehealth,15 it includes home management tools such as blood pressure cuffs, cardiotocography, scales for weight checks, and Doppler ultrasounds.10 Patients can be instructed to measure fundal height and receive medications by mail. Anesthesia consultation can occur via this venue by having the patient complete a questionnaire prior to arriving at the labor and delivery unit.16
Legal considerations
With the COVID-19 pandemic, temporary changes were made to encourage the rapid adoption of telemedicine, including changes to licensing laws, certain prescription requirements, Health Insurance Portability and Accountability Act (HIPAA) privacy-security regulations, and reimbursement rules that required in-person visits. Thus, many ObGyns started using telemedicine during this rarified period, in which the rules appeared to be few and far between, with limited enforcement of the law and professional obligations.17 However, now that many of the legal rules that were suspended or ignored have been (or are being) reimposed and enforced, it is important for providers to become familiar with the legal issues involved in practicing telemedicine.
First, where is the patient? When discussing the legal issues of telemedicine, it is important to remember that many legal rules for medical care (ie, liability, informed consent, and licensing) vary from state to state. If the patient resides in a different state (“foreign” state) from the physician’s practice location (the physician’s “home” state), the care is considered delivered in the state where the patient is located. Thus, the patient’s location generally establishes the law covering the telemedicine transaction. In the following discussion, the rules refer to the law and professional obligations, with commentary on some key legal issues that are relevant to ObGyn telemedicine.
Continue to: Reinforcing the rules...
Reinforcing the rules
Licensing
During the height of the COVID-19 pandemic, the federal government and almost all states temporarily modified the licensing requirement to allow telemedicine based on an existing medical license in any state—disregarding the “where is the patient” rule. As those rules begin to lapse or change with the official end of the pandemic declared by President Biden as May 2023,17 the rules under which a physician began telemedicine interstate practice in 2020 also may be changing.
Simply put, “The same standards for licensure apply to health care providers regardless of whether care is delivered in-person or virtually through telehealth services.”18 When a physician is engaged in telemedicine treatment of a patient in the physician’s home state, there is generally no licensing issue. Telemedicine generally does not require a separate specific license.19 However, when the patient is in another state (a “foreign” state), there can be a substantial licensing issue.20 Ordinarily, to provide that treatment, the physician must, in some manner, be approved to practice in the patient’s state. That may occur, for example, in the following ways: (1) the physician may hold an additional regular license in the patient’s state, which allows practice there, or (2) the physician may have received permission for “temporary practice” in another state.
Many states (often adjoining states) have formal agreements with other states that allow telemedicine practice by providers in each other’s states. There also are “compacts”, or agreements that enable providers in any of the participating states to practice in the other associated states without a separate license.18 Although several websites provide information about compact licensing and the like, clinicians should not rely on simple lists or maps. Individual states may have special provisions about applying their laws to out-of-state “compact” physicians. In addition, under the Interstate Medical Licensure Compact, “physicians have to pay licensing fees and satisfy the requirements of each medical board in the states where they wish to practice.”21
Consequences. Practicing telemedicine with a patient in a state where the physician does not have a license is generally a crime. Furthermore, it may be the basis for license discipline in the physician’s home state and result in a report to the National Practi-tioner Databank.22 In addition, reimbursement often depends on the practitioner being licensed, and the absence of a license may be a basis for denying payment for services.23 Finally, malpractice insurance generally is limited to licensed practice. Thus, the insurer may decline to defend the unlicensed clinician against a malpractice claim or pay any damages.
Prescribing privileges
Prescribing privileges usually are connected to licensing, so as the rules for licensing change postpandemic, so do the rules for prescribing. In most cases, the physician must have a license in the state where care is given to prescribe medication—which in telemedicine, as noted, typically means the state where the patient is located. Exceptions vary by state, but in general, if a physician does not have a license to provide care, the physician is unlikely to be authorized to prescribe medication.24 Failure to abide by the applicable state rules may result in civil and even criminal liability for illegal prescribing activity.
In addition, the US Food and Drug Administration (FDA) and Drug Enforcement Administration (DEA, which enforces laws concerning controlled substances) also regulate the prescription and sale of pharmaceuticals.25 There are state and federal limits on the ability of clinicians to order controlled substances without an in-person visit. The Ryan Haight Online Pharmacy Consumer Protection Act, for example, sets limits on controlled substance prescriptions without an in-person examination.26 Federal law was modified due to COVID-19 to permit prescribing of many controlled substances by telemedicine if there is synchronous audio and visual examination of the patient. Physicians who write such prescriptions also are required to have a DEA registration in the patient’s state. This is an essential consideration for physicians considering interstate telemedicine practice.27
HIPAA and privacy
Governments waived some of the legal requirements related to health information during the pandemic, but those waivers either have expired or will do so soon. Federal and state laws regarding privacy and security—notably including HIPAA—apply to telemedicine and are of particular concern given the considerable amount of communication of protected health information with telemedicine.
HIPAA security rules essentially require making sure health information cannot be hacked or intercepted. Audio-only telemedicine by landline (not cell) is acceptable under the security rules, but almost all other remote communication requires secure communications.28
Clinicians also need to adhere to the more usual HIPAA privacy rules when practicingtelehealth. State laws protecting patient privacy vary and may be more stringent than HIPAA, so clinicians also must know the requirements in any state where they practice—whether in office or telemedicine.29
Making sure telemedicine practices are consistent with these security and privacy rules often requires particular technical expertise that is outside the realm of most practicing clinicians. However, without modification, the pre-telemedicine technology of many medical offices likely is insufficient for the full range of telemedicine services.30
Reimbursement and fraud
Before COVID-19, Medicare and Medicaid reimbursement for telemedicine was limited. Government decisions to substantially broaden those reimbursement rules (at least temporarily) provided a substantial boost to telemedicine early in the pandemic.23 Federal regulations and statutes also expanded telemedicine reimbursement for various services. Some will end shortly after the health emergency, and others will be permanent. Parts of that will not be sorted out for several years, so it will likely be a changing landscape for reimbursement.
Continue to: Rules that are evolving...
Rules that are evolving
Informed consent
The ethical and legal obligations to obtain informed consent are present in telemedicineas well as in-person care, with the same basic requirements regarding risks, benefits, alternative care, etc.32 However, with telemedicine, information related to remote care should be included and is outlined in TABLE 1.

Certain states may have somewhat unique informed consent requirements—especially for reproductive care, including abortion.34 Therefore, it is important for clinicians to ensure their consent process and forms comply with any legal jurisdiction in which a patient is located.
Medical malpractice
The basics of medical malpractice (or negligence) are the same in telemedicine as in in-person care: duty, breach of duty, and injury caused by the breach. That is, there may be liability when a medical professional breaches the duty of care, causing the patient’s injury. The physician’s duty is defined by the quality of care that the profession (specialty) accepts as reasonably good. This is defined by the opinions of physicians within the specialty and formal statements from professional organizations, including ACOG.3
Maintaining the standard of care and quality. The use of telemedicine is not an excuse to lower the quality of health care. There are some circumstances for which it is medically better to have an in-person visit. In these instances, the provider should recommend the appropriate care, even if telemedicine would be more convenient for the provider and staff.35
If the patient insists and telemedicine might result in less than optimal care, the reasons for using a remote visit should be clearly documented contemporaneously with the decision. Furthermore, when the limitations of being unable to physically examine the patient result in less information than is needed for the patient’s care, the provider must find alternatives to make up for the information gap.11,36 It also may be necessary to inform patients about how to maximize telemedicine care.37 At the beginning of telemedicine care the provider should include information about the nature and limits of telehealth, and the patient’s responsibilities. (See TABLE 1) Throughout treatment of the patient, that information should be updated by the provider. That, of course, is particularly important for patients who have not previously used telemedice services.
Malpractice rules vary by state. Many states have special rules regarding malpractice cases. These differences in malpractice standards and regulations “can be problematic for physicians who use telemedicine services to provide care outside the state in which they practice.”38 Caps on noneconomic damages are an example. Those state rules would apply to telemedicine in the patient’s state.
Malpractice insurance
Malpractice insurance now commonly includes telemedicine legally practiced within the physician’s home state. Practitioners who treat patients in foreign states should carefully examine their malpractice insurance policies to confirm that the coverage extends to practice in those states.39 Malpractice carriers may require notification by a covered physician who routinely provides services to patients in another state.3
Keep in mind, malpractice insurance generally does not cover the practice of medicine that is illegal. Practicing telemedicine in a foreign state, where the physician or other provider does not have a license and where that state does not otherwise permit the practice, is illegal. Most likely, the physician’s malpractice insurance will not cover claims that arise from this illegal practice in a foreign state or provide defense for malpractice claims, including frivolous lawsuits. Thus, the physician will pay out of pocket for the costs of a defense attorney.
Telemedicine treatment of minors
Children and adolescents present special legal issues for ObGyn care, which may become more complicated with telemedicine. Historically, parents are responsible for minors (those aged <18 years): they consent to medical treatment, are responsible for paying for it, and have the right to receive information about treatment.
Over the years, though, many states have made exceptions to these principles, especially with regard to contraception and treatment of sexually transmitted diseases.40 For abortion, in particular, there is considerable variation among the states in parental consent and notification.41 The Supreme Court’s decision in Dobbs v Jackson Women’s Health42 may (depending on the state) be followed with more stringent limitations on adolescent consent to abortions, including medical abortions.43
Use of telehealth does not change any obligations regarding adolescent consent or parental notification. Because those differ considerably among states, it is important for all practitioners to know their states’ requirements and keep reasonably complete records demonstrating their compliance with state law.
Abortion
The most heated current controversy about telemedicine involves abortion—specifically medical abortion, which is the combination of mifepristone and misoprostol.44,45 The FDA approved the combination in 2000. Almost immediately, many states required in-person visits with a certified clinician to receive a prescription for mifepristone and misoprostol, and eventually, the FDA adopted similar requirements.46 However, during the pandemic from 2021 to 2022, the FDA permitted telemedicine prescriptions. Several states still require in-person physician visits, although the constitutionality of those requirements has not been established.47
With the Supreme Court’s decision in Dobbs v Jackson Women’s Health in 2022,42 disagreements have ensued about the degree to which states may regulate the prescription of FDA-approved medical abortion drugs. Thorny constitutional issues exist in the plans of both abortion opponents and proponents in the battle over medical abortion in antiabortion states. It may be that federal drug law preempts state laws limiting access to FDA-approved drugs. On the other hand, it may be that states can make it a crime within the state to possess or provide abortion-inducing drugs. Courts will probably take years to resolve the many tangled legal questions.48
Thus, while the pandemic telemedicine rules may have advanced access to abortion,34 there may be some pending downsides.49 States that prohibit abortion will likely include prohibitions on medical abortions. In addition, they may prohibit anyone in the state (including pharmacies) from selling, possessing, or obtaining any drug used for causing or inducing an abortion.50 If, for constitutional reasons, they cannot press criminal charges or undertake licensing discipline for prescribing abortion, some states will likely withdraw from telehealth licensing compacts to avoid out-of-state prescriptions. This area of telemedicine has considerable uncertainty.
Continue to: CASE Conclusion...
CASE Conclusion
Patient concerns come to the fore
By 2023, Dr. TM started receiving bad news. Patient D called complaining that after following the advice on the website, she suffered a severe reaction and had to be rushed to an emergency department. Patient E (who had only 1 in-office visit early in her pregnancy) notified the office that she developed very high blood pressure that resulted in severe placental abruption, requiring emergency care and resulting in the loss of the fetus. Patient F complained that someone hacked the TikTok direct message communication with Dr. TM and tried to “blackmail” or harass her.
Discussion. Patients D, E, and F represent potential problems of telemedicine practice. Patient D was injured because she relied on her doctor’s website (to which Dr. TM directed patients). It contained an error that caused an injury. A doctor-patient relationship existed, and bad medical advice likely caused the injury. Physicians providing advice online must ensure the advice is correct and kept current.
Patient E demonstrates the importance of monitoring patients remotely (blood pressure transmitted to the office) or with periodic in-office visits. It is not clear whether she was a no-show for office visits (and whether the office followed up on any missed appointments) or if such visits were never scheduled. Liability for failure to monitor adequately is a possibility.
Patient F’s seemingly minor complaint could be a potential problem. Dr. TM used an insecure mode of communication. Although some HIPAA security regulations were modified or suspended during the pandemic, using such an unsecure platform is problematic, especially if temporary HIPAA rules expired. The outcome of the complaint is in doubt.

(See TABLE 2 for additional comments on patients D, E, and F.)
Out-of-state practice
Dr. TM treated 3 out-of-state residents (D, E, and F) via telemedicine. Recently Dr. TM received a complaint from the State Medical Licensure Board for practicing medicine without a license (Patient D), followed by similar charges from Patient E’s and Patient F’s state licensing boards. He has received a licensing inquiry from his home state board about those claims of illegal practice in other states and incompetent treatment.
Patient D’s pregnancy did not go well. The 1 in-person visit did not occur and she has filed a malpractice suit against Dr. TM. Patient E is threatening a malpractice case because the STI was not appropriately diagnosed and had advanced before another physician treated it.
In addition, a private citizen in Patient F’s state has filed suit against Dr. TM for abetting an illegal abortion (for Patient F).
Discussion. Patients D, E, and F illustrate the risk of even incidental out-of-state practice. The medical board inquiries arose from anonymous tips to all 4 states reporting Dr. TM was “practicing medicine without a license.” Patient E’s home state did have a licensing compact with the adjoining state (ie, Dr. TM’s home state). However, it required physicians to register and file an annual report, which Dr. TM had not done. The other 2 states did not have compacts with Dr. TM’s home state. Thus, he was illegally practicing medicine and would be subject to penalties. His home state also might impose license discipline based on his illegal practice in other states.
Continue to: What’s the verdict?...
What’s the verdict?
Dr. TM’s malpractice carrier is refusing to defend the claims of medical malpractice threatened by Patients D, E, and F. The company first notes that the terms of the malpractice policy specifically exclude the illegal practice of medicine. Furthermore, when a physician legally practices in another state, the policy requires a written notice to the insurance carrier of such practice. Dr. TM will likely have to engage and pay for a malpractice attorney for these cases. Because the claims are filed in 3 different states, more than a home-state attorney will likely be involved in the defense of these cases. Dr. TM will need to pay the attorneys and any damages from a settlement or trial.
Malpractice claims. Patient D claims that the doctor essentially abandoned her by never reaching out to her or arranging an in-person visit. Dr. TM claims the patient was responsible for scheduling the in-person visit. Patient E claims it was malpractice not to determine the specific nature of the STI and to do follow-up testing to determine that it was cured. All patients claim there was no genuine informed consent to the telemedicine. An attorney has warned Dr. TM that it is “not going to look good to the jury” that he was practicing without a license in the state and suggests he settle the cases quickly by paying damages.
Abortion-related claims. Patient F presents a different set of problems. Dr. TM’s home state is “proabortion.” Patient F’s home state is strongly “antiabortion,” making it a felony to participate in, assist, or facilitate an abortion (including medical abortion). Criminal charges have been filed against Dr. TM for the illegal practice of medicine, for aiding and facilitating an abortion, and for failure to notify a parent that a minor is seeking an abortion. For now, Dr. TM’s state is refusing to extradite on the abortion charge. Still, the patient’s state insists that it do so on the illegal practice of medicine charges and new charges of insurance fraud and failure to report suspected sexual abuse of a child. (Under the patient’s state law, anyone having sex with Patient F would have engaged in sexual abuse or “statutory rape,” so the state insists that the fact she was pregnant proves someone had sex with her.)
Patient F’s state also has a statute that allows private citizens to file civil claims against anyone procuring or assisting with an abortion (a successful private citizen can receive a minimum of $10,000 from the defendant). Several citizens from the patient’s state have already filed claims against Dr. TM in his state courts. Only one of them, probably the first to file, could succeed. Courts in the state have issued subpoenas and ordered Dr. TM to appear and reply to the civil suits. If he does not respond, there will be a default judgment.
Dr. TM’s attorney tells him that these lawsuits will not settle and will take a long time to defend and resolve. That will be expensive.
Billing and fraud. Dr. TM’s office recently received a series of notices from private health insurers stating they are investigating previously made payments as being fraudulent (unlicensed). They will not pay any new claims pending the investigation. On behalf of Medicare-Medicaid and other federal programs, the US Attorney’s office has notified Dr. TM that it has opened an investigation into fraudulent federal payments. F’s home state also is filing a (criminal) insurance fraud case, although the basis for it is unclear. (Dr. TM’s attorney believes it might be to increase pressure on the physician’s state to extradite Dr. TM for Patient F’s case.)
In addition, a disgruntled former employee of Dr. TM has filed a federal FCA case against him for filing inflated claims with various federally funded programs. The employee also made whistleblower calls to insurance companies and some state-funded medical programs. A forensic accounting investigation by Dr. TM’s accountant confirmed a pattern of very sloppy records and recurring billing for televisits that did not occur. Dr. TM believes that this was the act of one of the temporary assistants he hired in a pinch, who did not understand the system and just guessed when filing some insurance claims.
During the investigation, the federal and state attorneys are looking into a possible violation of state and federal Anti-Kickback Statutes. This is based on the original offer of a $100 credit for referrals to Dr. TM’s telemedicine practice.
The attorneys are concerned that other legal problems may present themselves. They are thoroughly reviewing Dr. TM’s practice and making several critical but somewhat modest changes to his practice. They also have insisted that Dr. TM have appropriate staff to handle the details of the practice and billing.
Conclusions
Telemedicine presents notable legal challenges to medical practice. As the pandemic status ends, ObGyn physicians practicing telemedicine need to be aware of the rules and how they are changing. For those physicians who want to continue or start a telemedicine practice, securing legal and technical support to ensure your operations are inline with the legal requirements can minimize any risk of legal troubles in the future. ●
A physician in State A, where abortion is legal, has a telemedicine patient in State B, where it is illegal to assist, provide, or procure an abortion. If the physician prescribes a medical abortion, he would violate the law of State B by using telemedicine to help the patient (located in State B) obtain an abortion. This could result in criminal charges against the prescribing physician.

Telemedicine (or telehealth) originated in the early 1900s, when radios were used to communicate medical advice to clinics aboard ships.1 According to the American Telemedicine Association, telemedicine is namely “the use of medical information exchanged from one site to another via electronic communications to improve a patient’s clinical health status.”2 These communications use 2-way video, email, smartphones, wireless tools, and other forms of telecommunications technology.
During the COVID-19 pandemic, many ObGyns—encouraged and advised by professional organizations—began providing telemedicine services.3 The first reported case of COVID-19 was in late 2019; the use of telemedicine was 38 times higher in February 2021 than in February 2020,4 illustrating how many physicians quickly moved to telemedicine practices.
CASE Dr. TM’s telemedicine dream
Before COVID-19, Dr. TM (an ObGyn practi-tioner) practiced in-person medicine in his home state. With the onset of the pandemic, Dr. TM struggled to switch to primarily seeing patients online (generally using Zoom or Facebook Live), with 1 day per week in the office for essential in-person visits.
After several months, however, Dr. TM’s routine became very efficient. He could see many more patients in a shorter time than with the former, in-person system. Therefore, as staff left his practice, Dr. TM did not replace them and also laid off others. Ultimately, the practice had 1 full-time records/insurance secretary who worked from home and 1 part-time nurse who helped with the in-person day and answered some patient inquiries by email. In part as an effort to add new patients, Dr. TM built an engaging website through which his current patients could receive medical information and new patients could sign up.
In late 2022, Dr. TM offered a $100 credit to any current patient who referred a friend or family member who then became a patient. This promotion was surprisingly effective and resulted in an influx of new patients. For example, Patient Z (a long-time patient) received 3 credits for referring her 3 sisters who lived out of state and became telepatients: Patient D, who lived 200 hundred miles away; Patient E, who lived 50 miles away in the adjoining state; and Patient F, who lived 150 miles away. Patient D contacted Dr. TM because she thought she was pregnant and wanted prenatal care, Patient E thought she might have a sexually transmitted infection (STI) and wanted treatment, and Patient F wanted general care and was inquiring about a medical abortion. Dr. TM agreed to treat Patient D but required 1 in-person visit. After 1 brief telemedicine session each with Patients E and F, Dr. TM wrote prescriptions for them.
By 2023, Dr. TM was enthusiastic about telemedicine as a professional practice. However, problems would ensue.
Dos and don’ts of telemedicine2
- Do take the initiative and inform patients of the availability of telemedicine/telehealth services
- Do use the services of medical malpractice insurance companies with regard to telemedicine
- Do integrate telemedicine into practice protocols and account for their limitations
- Don’t assume there are blanket exemptions or waivers in the states where your patients are located
Medical considerations
Telemedicine is endorsed by the American College of Obstetricians and Gynecologists (ACOG) as a vehicle for delivering prenatal and postpartum care.5 This represents an effort to reduce maternal and neonatal morbidity and mortality,5 as well as expand access to care and address the deficit in primary care providers and services, especially in rural and underserved populations.5,6 For obstetrics, prenatal care is designed to optimize pregnancy, childbirth, and postpartum care, with a focus on nutrition and genetic consultation and patient education on pregnancy, childbearing, breastfeeding, and newborn care.7
Benefits of telemedicine include its convenience for patients and providers, its efficiency and lower costs for providers (and hopefully patients, as well), and the potential improved access to care for patients.8 It is estimated that if a woman inititates obstetric care at 6 weeks, over the course of the 40-week gestation period, 15 prenatal visits will occur.9 Ultimately, the number of visits is determined based on the specifics of the pregnancy. With telemedicine, clinicians can provide those consultations, and information related to: ultrasonography, fetal echocardiography, and postpartum care services remotely.10 Using telemedicine may reduce missed visits, and remote monitoring may improve the quality of care.11
Barriers to telemedicine care include technical limitations, time constraints, and patient concerns of telehealth (visits). Technical limitations include the lack of a high speed internet connection and/or a smart device and the initial technical set-up–related problems,12 which affect providers as well as patients. Time constraints primarly refer to the ObGyn practice’s lack of time to establish telehealth services.13 Other challenges include integrating translation services, billing-related problems,10 and reimbursement and licensing barriers.14
Before the COVID-19 pandemic, obstetrics led the way in telemedicine with the development of the OB Nest model. Designed to replace in-person obstetrics care visits with telehealth,15 it includes home management tools such as blood pressure cuffs, cardiotocography, scales for weight checks, and Doppler ultrasounds.10 Patients can be instructed to measure fundal height and receive medications by mail. Anesthesia consultation can occur via this venue by having the patient complete a questionnaire prior to arriving at the labor and delivery unit.16
Legal considerations
With the COVID-19 pandemic, temporary changes were made to encourage the rapid adoption of telemedicine, including changes to licensing laws, certain prescription requirements, Health Insurance Portability and Accountability Act (HIPAA) privacy-security regulations, and reimbursement rules that required in-person visits. Thus, many ObGyns started using telemedicine during this rarified period, in which the rules appeared to be few and far between, with limited enforcement of the law and professional obligations.17 However, now that many of the legal rules that were suspended or ignored have been (or are being) reimposed and enforced, it is important for providers to become familiar with the legal issues involved in practicing telemedicine.
First, where is the patient? When discussing the legal issues of telemedicine, it is important to remember that many legal rules for medical care (ie, liability, informed consent, and licensing) vary from state to state. If the patient resides in a different state (“foreign” state) from the physician’s practice location (the physician’s “home” state), the care is considered delivered in the state where the patient is located. Thus, the patient’s location generally establishes the law covering the telemedicine transaction. In the following discussion, the rules refer to the law and professional obligations, with commentary on some key legal issues that are relevant to ObGyn telemedicine.
Continue to: Reinforcing the rules...
Reinforcing the rules
Licensing
During the height of the COVID-19 pandemic, the federal government and almost all states temporarily modified the licensing requirement to allow telemedicine based on an existing medical license in any state—disregarding the “where is the patient” rule. As those rules begin to lapse or change with the official end of the pandemic declared by President Biden as May 2023,17 the rules under which a physician began telemedicine interstate practice in 2020 also may be changing.
Simply put, “The same standards for licensure apply to health care providers regardless of whether care is delivered in-person or virtually through telehealth services.”18 When a physician is engaged in telemedicine treatment of a patient in the physician’s home state, there is generally no licensing issue. Telemedicine generally does not require a separate specific license.19 However, when the patient is in another state (a “foreign” state), there can be a substantial licensing issue.20 Ordinarily, to provide that treatment, the physician must, in some manner, be approved to practice in the patient’s state. That may occur, for example, in the following ways: (1) the physician may hold an additional regular license in the patient’s state, which allows practice there, or (2) the physician may have received permission for “temporary practice” in another state.
Many states (often adjoining states) have formal agreements with other states that allow telemedicine practice by providers in each other’s states. There also are “compacts”, or agreements that enable providers in any of the participating states to practice in the other associated states without a separate license.18 Although several websites provide information about compact licensing and the like, clinicians should not rely on simple lists or maps. Individual states may have special provisions about applying their laws to out-of-state “compact” physicians. In addition, under the Interstate Medical Licensure Compact, “physicians have to pay licensing fees and satisfy the requirements of each medical board in the states where they wish to practice.”21
Consequences. Practicing telemedicine with a patient in a state where the physician does not have a license is generally a crime. Furthermore, it may be the basis for license discipline in the physician’s home state and result in a report to the National Practi-tioner Databank.22 In addition, reimbursement often depends on the practitioner being licensed, and the absence of a license may be a basis for denying payment for services.23 Finally, malpractice insurance generally is limited to licensed practice. Thus, the insurer may decline to defend the unlicensed clinician against a malpractice claim or pay any damages.
Prescribing privileges
Prescribing privileges usually are connected to licensing, so as the rules for licensing change postpandemic, so do the rules for prescribing. In most cases, the physician must have a license in the state where care is given to prescribe medication—which in telemedicine, as noted, typically means the state where the patient is located. Exceptions vary by state, but in general, if a physician does not have a license to provide care, the physician is unlikely to be authorized to prescribe medication.24 Failure to abide by the applicable state rules may result in civil and even criminal liability for illegal prescribing activity.
In addition, the US Food and Drug Administration (FDA) and Drug Enforcement Administration (DEA, which enforces laws concerning controlled substances) also regulate the prescription and sale of pharmaceuticals.25 There are state and federal limits on the ability of clinicians to order controlled substances without an in-person visit. The Ryan Haight Online Pharmacy Consumer Protection Act, for example, sets limits on controlled substance prescriptions without an in-person examination.26 Federal law was modified due to COVID-19 to permit prescribing of many controlled substances by telemedicine if there is synchronous audio and visual examination of the patient. Physicians who write such prescriptions also are required to have a DEA registration in the patient’s state. This is an essential consideration for physicians considering interstate telemedicine practice.27
HIPAA and privacy
Governments waived some of the legal requirements related to health information during the pandemic, but those waivers either have expired or will do so soon. Federal and state laws regarding privacy and security—notably including HIPAA—apply to telemedicine and are of particular concern given the considerable amount of communication of protected health information with telemedicine.
HIPAA security rules essentially require making sure health information cannot be hacked or intercepted. Audio-only telemedicine by landline (not cell) is acceptable under the security rules, but almost all other remote communication requires secure communications.28
Clinicians also need to adhere to the more usual HIPAA privacy rules when practicingtelehealth. State laws protecting patient privacy vary and may be more stringent than HIPAA, so clinicians also must know the requirements in any state where they practice—whether in office or telemedicine.29
Making sure telemedicine practices are consistent with these security and privacy rules often requires particular technical expertise that is outside the realm of most practicing clinicians. However, without modification, the pre-telemedicine technology of many medical offices likely is insufficient for the full range of telemedicine services.30
Reimbursement and fraud
Before COVID-19, Medicare and Medicaid reimbursement for telemedicine was limited. Government decisions to substantially broaden those reimbursement rules (at least temporarily) provided a substantial boost to telemedicine early in the pandemic.23 Federal regulations and statutes also expanded telemedicine reimbursement for various services. Some will end shortly after the health emergency, and others will be permanent. Parts of that will not be sorted out for several years, so it will likely be a changing landscape for reimbursement.
Continue to: Rules that are evolving...
Rules that are evolving
Informed consent
The ethical and legal obligations to obtain informed consent are present in telemedicineas well as in-person care, with the same basic requirements regarding risks, benefits, alternative care, etc.32 However, with telemedicine, information related to remote care should be included and is outlined in TABLE 1.

Certain states may have somewhat unique informed consent requirements—especially for reproductive care, including abortion.34 Therefore, it is important for clinicians to ensure their consent process and forms comply with any legal jurisdiction in which a patient is located.
Medical malpractice
The basics of medical malpractice (or negligence) are the same in telemedicine as in in-person care: duty, breach of duty, and injury caused by the breach. That is, there may be liability when a medical professional breaches the duty of care, causing the patient’s injury. The physician’s duty is defined by the quality of care that the profession (specialty) accepts as reasonably good. This is defined by the opinions of physicians within the specialty and formal statements from professional organizations, including ACOG.3
Maintaining the standard of care and quality. The use of telemedicine is not an excuse to lower the quality of health care. There are some circumstances for which it is medically better to have an in-person visit. In these instances, the provider should recommend the appropriate care, even if telemedicine would be more convenient for the provider and staff.35
If the patient insists and telemedicine might result in less than optimal care, the reasons for using a remote visit should be clearly documented contemporaneously with the decision. Furthermore, when the limitations of being unable to physically examine the patient result in less information than is needed for the patient’s care, the provider must find alternatives to make up for the information gap.11,36 It also may be necessary to inform patients about how to maximize telemedicine care.37 At the beginning of telemedicine care the provider should include information about the nature and limits of telehealth, and the patient’s responsibilities. (See TABLE 1) Throughout treatment of the patient, that information should be updated by the provider. That, of course, is particularly important for patients who have not previously used telemedice services.
Malpractice rules vary by state. Many states have special rules regarding malpractice cases. These differences in malpractice standards and regulations “can be problematic for physicians who use telemedicine services to provide care outside the state in which they practice.”38 Caps on noneconomic damages are an example. Those state rules would apply to telemedicine in the patient’s state.
Malpractice insurance
Malpractice insurance now commonly includes telemedicine legally practiced within the physician’s home state. Practitioners who treat patients in foreign states should carefully examine their malpractice insurance policies to confirm that the coverage extends to practice in those states.39 Malpractice carriers may require notification by a covered physician who routinely provides services to patients in another state.3
Keep in mind, malpractice insurance generally does not cover the practice of medicine that is illegal. Practicing telemedicine in a foreign state, where the physician or other provider does not have a license and where that state does not otherwise permit the practice, is illegal. Most likely, the physician’s malpractice insurance will not cover claims that arise from this illegal practice in a foreign state or provide defense for malpractice claims, including frivolous lawsuits. Thus, the physician will pay out of pocket for the costs of a defense attorney.
Telemedicine treatment of minors
Children and adolescents present special legal issues for ObGyn care, which may become more complicated with telemedicine. Historically, parents are responsible for minors (those aged <18 years): they consent to medical treatment, are responsible for paying for it, and have the right to receive information about treatment.
Over the years, though, many states have made exceptions to these principles, especially with regard to contraception and treatment of sexually transmitted diseases.40 For abortion, in particular, there is considerable variation among the states in parental consent and notification.41 The Supreme Court’s decision in Dobbs v Jackson Women’s Health42 may (depending on the state) be followed with more stringent limitations on adolescent consent to abortions, including medical abortions.43
Use of telehealth does not change any obligations regarding adolescent consent or parental notification. Because those differ considerably among states, it is important for all practitioners to know their states’ requirements and keep reasonably complete records demonstrating their compliance with state law.
Abortion
The most heated current controversy about telemedicine involves abortion—specifically medical abortion, which is the combination of mifepristone and misoprostol.44,45 The FDA approved the combination in 2000. Almost immediately, many states required in-person visits with a certified clinician to receive a prescription for mifepristone and misoprostol, and eventually, the FDA adopted similar requirements.46 However, during the pandemic from 2021 to 2022, the FDA permitted telemedicine prescriptions. Several states still require in-person physician visits, although the constitutionality of those requirements has not been established.47
With the Supreme Court’s decision in Dobbs v Jackson Women’s Health in 2022,42 disagreements have ensued about the degree to which states may regulate the prescription of FDA-approved medical abortion drugs. Thorny constitutional issues exist in the plans of both abortion opponents and proponents in the battle over medical abortion in antiabortion states. It may be that federal drug law preempts state laws limiting access to FDA-approved drugs. On the other hand, it may be that states can make it a crime within the state to possess or provide abortion-inducing drugs. Courts will probably take years to resolve the many tangled legal questions.48
Thus, while the pandemic telemedicine rules may have advanced access to abortion,34 there may be some pending downsides.49 States that prohibit abortion will likely include prohibitions on medical abortions. In addition, they may prohibit anyone in the state (including pharmacies) from selling, possessing, or obtaining any drug used for causing or inducing an abortion.50 If, for constitutional reasons, they cannot press criminal charges or undertake licensing discipline for prescribing abortion, some states will likely withdraw from telehealth licensing compacts to avoid out-of-state prescriptions. This area of telemedicine has considerable uncertainty.
Continue to: CASE Conclusion...
CASE Conclusion
Patient concerns come to the fore
By 2023, Dr. TM started receiving bad news. Patient D called complaining that after following the advice on the website, she suffered a severe reaction and had to be rushed to an emergency department. Patient E (who had only 1 in-office visit early in her pregnancy) notified the office that she developed very high blood pressure that resulted in severe placental abruption, requiring emergency care and resulting in the loss of the fetus. Patient F complained that someone hacked the TikTok direct message communication with Dr. TM and tried to “blackmail” or harass her.
Discussion. Patients D, E, and F represent potential problems of telemedicine practice. Patient D was injured because she relied on her doctor’s website (to which Dr. TM directed patients). It contained an error that caused an injury. A doctor-patient relationship existed, and bad medical advice likely caused the injury. Physicians providing advice online must ensure the advice is correct and kept current.
Patient E demonstrates the importance of monitoring patients remotely (blood pressure transmitted to the office) or with periodic in-office visits. It is not clear whether she was a no-show for office visits (and whether the office followed up on any missed appointments) or if such visits were never scheduled. Liability for failure to monitor adequately is a possibility.
Patient F’s seemingly minor complaint could be a potential problem. Dr. TM used an insecure mode of communication. Although some HIPAA security regulations were modified or suspended during the pandemic, using such an unsecure platform is problematic, especially if temporary HIPAA rules expired. The outcome of the complaint is in doubt.

(See TABLE 2 for additional comments on patients D, E, and F.)
Out-of-state practice
Dr. TM treated 3 out-of-state residents (D, E, and F) via telemedicine. Recently Dr. TM received a complaint from the State Medical Licensure Board for practicing medicine without a license (Patient D), followed by similar charges from Patient E’s and Patient F’s state licensing boards. He has received a licensing inquiry from his home state board about those claims of illegal practice in other states and incompetent treatment.
Patient D’s pregnancy did not go well. The 1 in-person visit did not occur and she has filed a malpractice suit against Dr. TM. Patient E is threatening a malpractice case because the STI was not appropriately diagnosed and had advanced before another physician treated it.
In addition, a private citizen in Patient F’s state has filed suit against Dr. TM for abetting an illegal abortion (for Patient F).
Discussion. Patients D, E, and F illustrate the risk of even incidental out-of-state practice. The medical board inquiries arose from anonymous tips to all 4 states reporting Dr. TM was “practicing medicine without a license.” Patient E’s home state did have a licensing compact with the adjoining state (ie, Dr. TM’s home state). However, it required physicians to register and file an annual report, which Dr. TM had not done. The other 2 states did not have compacts with Dr. TM’s home state. Thus, he was illegally practicing medicine and would be subject to penalties. His home state also might impose license discipline based on his illegal practice in other states.
Continue to: What’s the verdict?...
What’s the verdict?
Dr. TM’s malpractice carrier is refusing to defend the claims of medical malpractice threatened by Patients D, E, and F. The company first notes that the terms of the malpractice policy specifically exclude the illegal practice of medicine. Furthermore, when a physician legally practices in another state, the policy requires a written notice to the insurance carrier of such practice. Dr. TM will likely have to engage and pay for a malpractice attorney for these cases. Because the claims are filed in 3 different states, more than a home-state attorney will likely be involved in the defense of these cases. Dr. TM will need to pay the attorneys and any damages from a settlement or trial.
Malpractice claims. Patient D claims that the doctor essentially abandoned her by never reaching out to her or arranging an in-person visit. Dr. TM claims the patient was responsible for scheduling the in-person visit. Patient E claims it was malpractice not to determine the specific nature of the STI and to do follow-up testing to determine that it was cured. All patients claim there was no genuine informed consent to the telemedicine. An attorney has warned Dr. TM that it is “not going to look good to the jury” that he was practicing without a license in the state and suggests he settle the cases quickly by paying damages.
Abortion-related claims. Patient F presents a different set of problems. Dr. TM’s home state is “proabortion.” Patient F’s home state is strongly “antiabortion,” making it a felony to participate in, assist, or facilitate an abortion (including medical abortion). Criminal charges have been filed against Dr. TM for the illegal practice of medicine, for aiding and facilitating an abortion, and for failure to notify a parent that a minor is seeking an abortion. For now, Dr. TM’s state is refusing to extradite on the abortion charge. Still, the patient’s state insists that it do so on the illegal practice of medicine charges and new charges of insurance fraud and failure to report suspected sexual abuse of a child. (Under the patient’s state law, anyone having sex with Patient F would have engaged in sexual abuse or “statutory rape,” so the state insists that the fact she was pregnant proves someone had sex with her.)
Patient F’s state also has a statute that allows private citizens to file civil claims against anyone procuring or assisting with an abortion (a successful private citizen can receive a minimum of $10,000 from the defendant). Several citizens from the patient’s state have already filed claims against Dr. TM in his state courts. Only one of them, probably the first to file, could succeed. Courts in the state have issued subpoenas and ordered Dr. TM to appear and reply to the civil suits. If he does not respond, there will be a default judgment.
Dr. TM’s attorney tells him that these lawsuits will not settle and will take a long time to defend and resolve. That will be expensive.
Billing and fraud. Dr. TM’s office recently received a series of notices from private health insurers stating they are investigating previously made payments as being fraudulent (unlicensed). They will not pay any new claims pending the investigation. On behalf of Medicare-Medicaid and other federal programs, the US Attorney’s office has notified Dr. TM that it has opened an investigation into fraudulent federal payments. F’s home state also is filing a (criminal) insurance fraud case, although the basis for it is unclear. (Dr. TM’s attorney believes it might be to increase pressure on the physician’s state to extradite Dr. TM for Patient F’s case.)
In addition, a disgruntled former employee of Dr. TM has filed a federal FCA case against him for filing inflated claims with various federally funded programs. The employee also made whistleblower calls to insurance companies and some state-funded medical programs. A forensic accounting investigation by Dr. TM’s accountant confirmed a pattern of very sloppy records and recurring billing for televisits that did not occur. Dr. TM believes that this was the act of one of the temporary assistants he hired in a pinch, who did not understand the system and just guessed when filing some insurance claims.
During the investigation, the federal and state attorneys are looking into a possible violation of state and federal Anti-Kickback Statutes. This is based on the original offer of a $100 credit for referrals to Dr. TM’s telemedicine practice.
The attorneys are concerned that other legal problems may present themselves. They are thoroughly reviewing Dr. TM’s practice and making several critical but somewhat modest changes to his practice. They also have insisted that Dr. TM have appropriate staff to handle the details of the practice and billing.
Conclusions
Telemedicine presents notable legal challenges to medical practice. As the pandemic status ends, ObGyn physicians practicing telemedicine need to be aware of the rules and how they are changing. For those physicians who want to continue or start a telemedicine practice, securing legal and technical support to ensure your operations are inline with the legal requirements can minimize any risk of legal troubles in the future. ●
A physician in State A, where abortion is legal, has a telemedicine patient in State B, where it is illegal to assist, provide, or procure an abortion. If the physician prescribes a medical abortion, he would violate the law of State B by using telemedicine to help the patient (located in State B) obtain an abortion. This could result in criminal charges against the prescribing physician.
- Board on Health Care Services; Institute of Medicine. The Role of Telehealth in an Evolving Health Care Environment: Workshop Summary. National Academies Press: 2012. https://www.ncbi.nlm.nih.gov/books/NBK207145/. Accessed March 30, 2023.
- Bruhn HK. Telemedicine: dos and don’ts to mitigate liability risk. J APPOS. 2020;24:195-196. doi:10.1016/j.jaapos. 2020.07.002
- Implementing telehealth in practice: ACOG Committee Opinion Summary, number 798. Obstet Gynecol. 2020; 2135:493-494. doi:10.1097/AOG.0000000000003672
- Bestsennyy O, Gilbert G, Harris A, et al. Telehealth: a quarter-trillion-dollar post-COVID-19 reality? McKinsey & Company. July 9, 2021. Accessed March 2, 2023. https://www.mckinsey.com/industries/healthcare/our-insights /telehealth-a-quarter-trillion-dollar-post-covid-19-reality
- Stanley AY, Wallace JB. Telehealth to improve perinatal care access. MCN Am J Matern Child Nurs. 2022;47:281-287. doi: 10.1097/NMC.0000000000000841
- Warshaw R. Health disparities affect millions in rural US communities. Association of American Medical Colleges. Published October 31, 2017. Accessed March 31, 2023. https://www.aamc.org/news-insights/health-disparities -affect-millions-rural-us-communities
- Almuslin H, AlDossary S. Models of incorporating telehealth into obstetric care during the COVID-19 pandemic, its benefits and barriers: a scoping review. Telemed J E Health. 2022;28:24-38. doi:10.1089/tmj.2020.0553
- Gold AE, Gilbert A, McMichael BJ. Socially distant health care. Tul L Rev. 2021;96:423-468. https://scholarship .law.ua.edu/cgi/viewcontent.cgi?article=1713&context =fac_articles. Accessed March 4, 2023.
- Zolotor AJ, Carlough MC. Update on prenatal care. Am Fam Physician. 2014;89:199-208.
- Odibo IN, Wendel PJ, Magann EF. Telemedicine in obstetrics. Clin Obstet Gynecol. 2013;56:422-433. doi:10.1097/ GRF.0b013e318290fef0
- Shmerling A, Hoss M, Malam N, et al. Prenatal care via telehealth. Prim Care. 2022;49:609-619. doi:10.1016/j. pop.2022.05.002
- Madden N, Emeruwa UN, Friedman AM, et al. Telehealth uptake into prenatal care and provider attitudes during COVID-19 pandemic in New York City: a quantitative and qualitative analysis. Am J Perinatol. 2020;37:1005-1014. doi:10.1055/s-0040-1712939
- Dosaj A, Thiyagarajan D, Ter Haar C, et al. Rapid implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2020;27:116-120. doi:10.1089/ tmj.2020.0219
- Lurie N, Carr B. The role of telehealth in the medical response to disasters. JAMA Intern Med. 2018;187:745-746. doi: 10.1001/jamainternmed.2018.1314
- Tobah YSB, LeBlanc A, Branda E, et al. Randomized comparison of a reduced-visit prenatal care model enhanced with remote monitoring. Am J Obstet Gynecol. 2019;221:638-e1-638.e8. doi:10.1016/j.ajog.2019.06.034
- Vivanti AJ, Deruelle P, Piccone O, et al. Follow-up for pregnant women during the COVID-19 pandemic: French national authority for health recommendations. J Gynecol Obstet Hum Reprod. 2020;49:101804. doi:10.1016/j. jogoh.2020.101804
- Ellimoottil C. Takeaways from 2 key studies on interstate telehealth use among Medicare fee-for-service beneficiaries. JAMA Health Forum. 2022;3:e223020-E223020. doi:10.1001/ jamahealthforum.2022.3020
- Harris J, Hartnett T, Hoagland GW, et al. What eliminating barriers to interstate telehealth taught us during the pandemic. Bipartisan Policy Center. Published November 2021. Accessed March 9, 2023. https://bipartisanpolicy .org/download/?file=/wp-content/uploads/2021/11/BPC -Health-Licensure-Brief_WEB.pdf.
- Center for Connected Health Policy. Cross-state licensing. Accessed February 21, 2023. https://www.cchpca.org/topic /cross-state-licensing-professional-requirements.
- US Department of Health & Human Services. Telehealth. Getting started with licensure. Published February 3, 2023. Accessed February 27, 2023. https://telehealth.hhs.gov /licensure/getting-started-licensure/
- US Department of Health & Human Services. Telehealth. Licensure. Accessed February 27, 2023. https://telehealth .hhs.gov/licensure
- US Department of Health & Human Services. National Practitioner Data Bank (NPDB) code lists. Published December 2022. Accessed March 9, 2023. https://www.npdb .hrsa.gov/software/CodeLists.pdf
- American College of Obstetricians and Gynecologists. COVID-19 FAQs for obstetrician-gynecologists, telehealth. 2020. Accessed March 5, 2023. https://www.acog.org /clinical-information/physician-faqs/covid-19-faqs-for -ob-gyns-telehealth
- Gorman RK. Prescribing medication through the practice of telemedicine: a comparative analysis of federal and state online prescribing policies, and policy considerations for the future. S Cal Interdisc Law J. 2020;30:739-769. https://gould .usc.edu/why/students/orgs/ilj/assets/docs/30-3-Gorman. pdf. Accessed March 10, 2023.
- Farringer DR. A telehealth explosion: using lessons from the pandemic to shape the future of telehealth regulation. Tex A&M Law Rev. 2021;9:1-47. https://scholarship.law.tamu. edu/cgi/viewcontent.cgi?article=1232&context=lawreview. Accessed February 28, 2023.
- Sterba KR, Johnson EE, Douglas E, et al. Implementation of a women’s reproductive behavioral health telemedicine program: a qualitative study of barriers and facilitators in obstetric and pediatric clinics. BMC Pregnancy Childbirth. 2023;23:167, 1-10. doi:10.1186/s12884-023-05463-2.
- US Department of Justice. COVID-19 FAQ (telemedicine). https://www.deadiversion.usdoj.gov/faq/coronavirus_faq .htm#TELE_FAQ2. Accessed March 13, 2023.
- US Department of Health & Human Services. Guidance on how the HIPAA rules permit covered health care providers and health plans to use remote communication technologies for audio-only telehealth. Published June 13, 2022. Accessed February 22, 2023. https://www.hhs.gov/hipaa/for-professionals/privacy /guidance/hipaa-audio-telehealth/index.html.
- Gray JME. HIPAA, telehealth, and the treatment of mental illness in a post-COVID world. Okla City Uni Law Rev. 2021;46:1-26. https://law.okcu.edu/wp-content /uploads/2022/04/J-Michael-E-Gray-HIPAA-Telehealth -and-Treament.pdf. Accessed March 9, 2023.
- Kurzweil C. Telemental health care and data privacy: current HIPAA privacy pitfalls and a proposed solution. Ann Health L Adv Dir. 2022;31:165.
- US Department of Health & Human Services and US Department of Justice. Health care fraud and abuse control program FY 2020: annual report. July 2021. Accessed March 9, 2023. https://oig.hhs.gov/publications/docs/hcfac /FY2020-hcfac.pdf
- Copeland KB. Telemedicine scams. Iowa Law Rev. 2022: 108:69-126. https://ilr.law.uiowa.edu/sites/ilr.law.uiowa.edu /files/2023-01/A2_Copeland.pdf. Accessed March 10, 2023.
- Solimini R, Busardò FP, Gibelli F, et al. Ethical and legal challenges of telemedicine in the era of the COVID-19 pandemic. Medicina (Kaunas). 2021;57:13141324. doi:10.3390/medicina57121314
- Reed A. COVID: a silver linings playbook. mobilizing pandemic era success stories to advance reproductive justice. Berkeley J Gender Law Justice. 2022;37:221-266. https://lawcat.berkeley.edu/record/1237158/files/16%20 Reed_final.pdf. Accessed March 11, 2023.
- Women’s Preventive Services Initiative and The American College of Obstetricians and Gynecologists. FAQ for telehealth services. Accessed March 2, 2023. https://www .womenspreventivehealth.org/wp-content/uploads/WPSI -Telehealth-FAQ.pdf
- Warren L, Chen KT. Telehealth apps in ObGyn practice. OBG Manag. 2022;34:46-47. doi:10.12788/obgm.0178
- American College of Obstetricians and Gynecologists. 10 telehealth tips for an Ob-Gyn visit. 2020. Accessed March 2, 2023. https://www.acog.org/womens-health /infographics/10-telehealth-tips-for-an-ob-gyn-visit
- Wolf TD. Telemedicine and malpractice: creating uniformity at the national level. Wm Mary Law Rev. 2019;61:15051536. https://scholarship.law.wm.edu/cgi/viewcontent.cgi ?article=3862&context=wmlr. Accessed March 11, 2023.
- Cahan E. Lawsuits, reimbursement, and liability insurance— facing the realities of a post-Roe era. JAMA. 2022;328:515517. doi:10.1001/jama.2022.9193
- Heinrich L, Hernandez AK, Laurie AR. Telehealth considerations for the adolescent patient. Prim Care. 2022;49:597-607. doi:10.1016/j.pop.2022.04.006
- Guttmacher Institute. An overview of consent to reproductive health services by young people. Published March 1, 2023. Accessed April 1, 2023. https://www.guttmacher.org /state-policy/explore/overview-minors-consent-law.
- Dobbs v. Jackson Women’s Health. No. 19–1392. June 24, 2022. Accessed April 1, 2023. https://www.supremecourt .gov/opinions/21pdf/19-1392_6j37.pdf
- Lindgren Y. Dobbs v. Jackson Women’s Health and the post-Roe landscape. J Am Acad Matrimonial Law. 2022;35:235283. https://www.aaml.org/wp-content/uploads/MAT110-1 .pdf. Accessed March 11, 2023.
- Mohiuddin H. The use of telemedicine during a pandemic to provide access to medication abortion. Hous J Health Law Policy. 2021;21:483-525. https://houstonhealthlaw. scholasticahq.com/article/34611.pdf. Accessed March 10, 2023.
- Rebouché R. The public health turn in reproductive rights. Wash & Lee Law Rev. 2021;78:1355-1432. https:// scholarlycommons.law.wlu.edu/cgi/viewcontent .cgi?article=4743&context=wlulr. Accessed March 10, 2023.
- Fliegel R. Access to medication abortion: now more important than ever. Am J Law Med. 2022;48:286-304. doi:10.1017/amj.2022.24
- Guttmacher Institute. Medication abortion. March 1, 2023. Accessed April 1, 2023 https://www.guttmacher.org /state-policy/explore/medication-abortion#:~:text=In%20 January%202023%2C%20the%20FDA,order%20to%20 dispense%20the%20pills
- Cohen DS, Donley G, Rebouché R. The new abortion battleground. Columbia Law Rev. 2023;123:1-100. https:// columbialawreview.org/content/the-new-abortion -battleground/. Accessed March 1, 2023.
- Hunt SA. Call me, beep me, if you want to reach me: utilizing telemedicine to expand abortion access. Vanderbilt Law Rev. 2023;76:323-359. Accessed March 10, 2023. https:// vanderbiltlawreview.org/lawreview/wp-content/uploads /sites/278/2023/01/Call-Me-Beep-Me-If-You-Want-toReach-Me-Utilizing-Telemedicine-to-Expand-AbortionAccess.pdf
- Gleckel JA, Wulkan SL. Abortion and telemedicine: looking beyond COVID-19 and the shadow docket. UC Davis Law Rev Online. 2020;54:105-121. https://lawreview.law.ucdavis. edu/online/54/files/54-online-Gleckel_Wulkan.pdf. Accessed April 1, 2023.
- Board on Health Care Services; Institute of Medicine. The Role of Telehealth in an Evolving Health Care Environment: Workshop Summary. National Academies Press: 2012. https://www.ncbi.nlm.nih.gov/books/NBK207145/. Accessed March 30, 2023.
- Bruhn HK. Telemedicine: dos and don’ts to mitigate liability risk. J APPOS. 2020;24:195-196. doi:10.1016/j.jaapos. 2020.07.002
- Implementing telehealth in practice: ACOG Committee Opinion Summary, number 798. Obstet Gynecol. 2020; 2135:493-494. doi:10.1097/AOG.0000000000003672
- Bestsennyy O, Gilbert G, Harris A, et al. Telehealth: a quarter-trillion-dollar post-COVID-19 reality? McKinsey & Company. July 9, 2021. Accessed March 2, 2023. https://www.mckinsey.com/industries/healthcare/our-insights /telehealth-a-quarter-trillion-dollar-post-covid-19-reality
- Stanley AY, Wallace JB. Telehealth to improve perinatal care access. MCN Am J Matern Child Nurs. 2022;47:281-287. doi: 10.1097/NMC.0000000000000841
- Warshaw R. Health disparities affect millions in rural US communities. Association of American Medical Colleges. Published October 31, 2017. Accessed March 31, 2023. https://www.aamc.org/news-insights/health-disparities -affect-millions-rural-us-communities
- Almuslin H, AlDossary S. Models of incorporating telehealth into obstetric care during the COVID-19 pandemic, its benefits and barriers: a scoping review. Telemed J E Health. 2022;28:24-38. doi:10.1089/tmj.2020.0553
- Gold AE, Gilbert A, McMichael BJ. Socially distant health care. Tul L Rev. 2021;96:423-468. https://scholarship .law.ua.edu/cgi/viewcontent.cgi?article=1713&context =fac_articles. Accessed March 4, 2023.
- Zolotor AJ, Carlough MC. Update on prenatal care. Am Fam Physician. 2014;89:199-208.
- Odibo IN, Wendel PJ, Magann EF. Telemedicine in obstetrics. Clin Obstet Gynecol. 2013;56:422-433. doi:10.1097/ GRF.0b013e318290fef0
- Shmerling A, Hoss M, Malam N, et al. Prenatal care via telehealth. Prim Care. 2022;49:609-619. doi:10.1016/j. pop.2022.05.002
- Madden N, Emeruwa UN, Friedman AM, et al. Telehealth uptake into prenatal care and provider attitudes during COVID-19 pandemic in New York City: a quantitative and qualitative analysis. Am J Perinatol. 2020;37:1005-1014. doi:10.1055/s-0040-1712939
- Dosaj A, Thiyagarajan D, Ter Haar C, et al. Rapid implementation of telehealth services during the COVID-19 pandemic. Telemed J E Health. 2020;27:116-120. doi:10.1089/ tmj.2020.0219
- Lurie N, Carr B. The role of telehealth in the medical response to disasters. JAMA Intern Med. 2018;187:745-746. doi: 10.1001/jamainternmed.2018.1314
- Tobah YSB, LeBlanc A, Branda E, et al. Randomized comparison of a reduced-visit prenatal care model enhanced with remote monitoring. Am J Obstet Gynecol. 2019;221:638-e1-638.e8. doi:10.1016/j.ajog.2019.06.034
- Vivanti AJ, Deruelle P, Piccone O, et al. Follow-up for pregnant women during the COVID-19 pandemic: French national authority for health recommendations. J Gynecol Obstet Hum Reprod. 2020;49:101804. doi:10.1016/j. jogoh.2020.101804
- Ellimoottil C. Takeaways from 2 key studies on interstate telehealth use among Medicare fee-for-service beneficiaries. JAMA Health Forum. 2022;3:e223020-E223020. doi:10.1001/ jamahealthforum.2022.3020
- Harris J, Hartnett T, Hoagland GW, et al. What eliminating barriers to interstate telehealth taught us during the pandemic. Bipartisan Policy Center. Published November 2021. Accessed March 9, 2023. https://bipartisanpolicy .org/download/?file=/wp-content/uploads/2021/11/BPC -Health-Licensure-Brief_WEB.pdf.
- Center for Connected Health Policy. Cross-state licensing. Accessed February 21, 2023. https://www.cchpca.org/topic /cross-state-licensing-professional-requirements.
- US Department of Health & Human Services. Telehealth. Getting started with licensure. Published February 3, 2023. Accessed February 27, 2023. https://telehealth.hhs.gov /licensure/getting-started-licensure/
- US Department of Health & Human Services. Telehealth. Licensure. Accessed February 27, 2023. https://telehealth .hhs.gov/licensure
- US Department of Health & Human Services. National Practitioner Data Bank (NPDB) code lists. Published December 2022. Accessed March 9, 2023. https://www.npdb .hrsa.gov/software/CodeLists.pdf
- American College of Obstetricians and Gynecologists. COVID-19 FAQs for obstetrician-gynecologists, telehealth. 2020. Accessed March 5, 2023. https://www.acog.org /clinical-information/physician-faqs/covid-19-faqs-for -ob-gyns-telehealth
- Gorman RK. Prescribing medication through the practice of telemedicine: a comparative analysis of federal and state online prescribing policies, and policy considerations for the future. S Cal Interdisc Law J. 2020;30:739-769. https://gould .usc.edu/why/students/orgs/ilj/assets/docs/30-3-Gorman. pdf. Accessed March 10, 2023.
- Farringer DR. A telehealth explosion: using lessons from the pandemic to shape the future of telehealth regulation. Tex A&M Law Rev. 2021;9:1-47. https://scholarship.law.tamu. edu/cgi/viewcontent.cgi?article=1232&context=lawreview. Accessed February 28, 2023.
- Sterba KR, Johnson EE, Douglas E, et al. Implementation of a women’s reproductive behavioral health telemedicine program: a qualitative study of barriers and facilitators in obstetric and pediatric clinics. BMC Pregnancy Childbirth. 2023;23:167, 1-10. doi:10.1186/s12884-023-05463-2.
- US Department of Justice. COVID-19 FAQ (telemedicine). https://www.deadiversion.usdoj.gov/faq/coronavirus_faq .htm#TELE_FAQ2. Accessed March 13, 2023.
- US Department of Health & Human Services. Guidance on how the HIPAA rules permit covered health care providers and health plans to use remote communication technologies for audio-only telehealth. Published June 13, 2022. Accessed February 22, 2023. https://www.hhs.gov/hipaa/for-professionals/privacy /guidance/hipaa-audio-telehealth/index.html.
- Gray JME. HIPAA, telehealth, and the treatment of mental illness in a post-COVID world. Okla City Uni Law Rev. 2021;46:1-26. https://law.okcu.edu/wp-content /uploads/2022/04/J-Michael-E-Gray-HIPAA-Telehealth -and-Treament.pdf. Accessed March 9, 2023.
- Kurzweil C. Telemental health care and data privacy: current HIPAA privacy pitfalls and a proposed solution. Ann Health L Adv Dir. 2022;31:165.
- US Department of Health & Human Services and US Department of Justice. Health care fraud and abuse control program FY 2020: annual report. July 2021. Accessed March 9, 2023. https://oig.hhs.gov/publications/docs/hcfac /FY2020-hcfac.pdf
- Copeland KB. Telemedicine scams. Iowa Law Rev. 2022: 108:69-126. https://ilr.law.uiowa.edu/sites/ilr.law.uiowa.edu /files/2023-01/A2_Copeland.pdf. Accessed March 10, 2023.
- Solimini R, Busardò FP, Gibelli F, et al. Ethical and legal challenges of telemedicine in the era of the COVID-19 pandemic. Medicina (Kaunas). 2021;57:13141324. doi:10.3390/medicina57121314
- Reed A. COVID: a silver linings playbook. mobilizing pandemic era success stories to advance reproductive justice. Berkeley J Gender Law Justice. 2022;37:221-266. https://lawcat.berkeley.edu/record/1237158/files/16%20 Reed_final.pdf. Accessed March 11, 2023.
- Women’s Preventive Services Initiative and The American College of Obstetricians and Gynecologists. FAQ for telehealth services. Accessed March 2, 2023. https://www .womenspreventivehealth.org/wp-content/uploads/WPSI -Telehealth-FAQ.pdf
- Warren L, Chen KT. Telehealth apps in ObGyn practice. OBG Manag. 2022;34:46-47. doi:10.12788/obgm.0178
- American College of Obstetricians and Gynecologists. 10 telehealth tips for an Ob-Gyn visit. 2020. Accessed March 2, 2023. https://www.acog.org/womens-health /infographics/10-telehealth-tips-for-an-ob-gyn-visit
- Wolf TD. Telemedicine and malpractice: creating uniformity at the national level. Wm Mary Law Rev. 2019;61:15051536. https://scholarship.law.wm.edu/cgi/viewcontent.cgi ?article=3862&context=wmlr. Accessed March 11, 2023.
- Cahan E. Lawsuits, reimbursement, and liability insurance— facing the realities of a post-Roe era. JAMA. 2022;328:515517. doi:10.1001/jama.2022.9193
- Heinrich L, Hernandez AK, Laurie AR. Telehealth considerations for the adolescent patient. Prim Care. 2022;49:597-607. doi:10.1016/j.pop.2022.04.006
- Guttmacher Institute. An overview of consent to reproductive health services by young people. Published March 1, 2023. Accessed April 1, 2023. https://www.guttmacher.org /state-policy/explore/overview-minors-consent-law.
- Dobbs v. Jackson Women’s Health. No. 19–1392. June 24, 2022. Accessed April 1, 2023. https://www.supremecourt .gov/opinions/21pdf/19-1392_6j37.pdf
- Lindgren Y. Dobbs v. Jackson Women’s Health and the post-Roe landscape. J Am Acad Matrimonial Law. 2022;35:235283. https://www.aaml.org/wp-content/uploads/MAT110-1 .pdf. Accessed March 11, 2023.
- Mohiuddin H. The use of telemedicine during a pandemic to provide access to medication abortion. Hous J Health Law Policy. 2021;21:483-525. https://houstonhealthlaw. scholasticahq.com/article/34611.pdf. Accessed March 10, 2023.
- Rebouché R. The public health turn in reproductive rights. Wash & Lee Law Rev. 2021;78:1355-1432. https:// scholarlycommons.law.wlu.edu/cgi/viewcontent .cgi?article=4743&context=wlulr. Accessed March 10, 2023.
- Fliegel R. Access to medication abortion: now more important than ever. Am J Law Med. 2022;48:286-304. doi:10.1017/amj.2022.24
- Guttmacher Institute. Medication abortion. March 1, 2023. Accessed April 1, 2023 https://www.guttmacher.org /state-policy/explore/medication-abortion#:~:text=In%20 January%202023%2C%20the%20FDA,order%20to%20 dispense%20the%20pills
- Cohen DS, Donley G, Rebouché R. The new abortion battleground. Columbia Law Rev. 2023;123:1-100. https:// columbialawreview.org/content/the-new-abortion -battleground/. Accessed March 1, 2023.
- Hunt SA. Call me, beep me, if you want to reach me: utilizing telemedicine to expand abortion access. Vanderbilt Law Rev. 2023;76:323-359. Accessed March 10, 2023. https:// vanderbiltlawreview.org/lawreview/wp-content/uploads /sites/278/2023/01/Call-Me-Beep-Me-If-You-Want-toReach-Me-Utilizing-Telemedicine-to-Expand-AbortionAccess.pdf
- Gleckel JA, Wulkan SL. Abortion and telemedicine: looking beyond COVID-19 and the shadow docket. UC Davis Law Rev Online. 2020;54:105-121. https://lawreview.law.ucdavis. edu/online/54/files/54-online-Gleckel_Wulkan.pdf. Accessed April 1, 2023.
Breast cancer screening advice ‘dangerous’ for black women
The U.S. Preventive Services Task Force currently recommends that breast cancer screening start at age 50 years, regardless of race or ethnicity.
But
The current “one-size-fits-all” policy to screen the entire female population from a certain age may be “neither fair and equitable nor optimal,” noted the authors, led by Tianhui Chen, PhD, with Zhejiang Cancer Hospital, Hangzhou, China.
The study was published online in JAMA Network Open.
Laurie R. Margolies, MD, chief of breast imaging at the Dubin Breast Center of the Mount Sinai Tisch Cancer Center in New York City, agreed.
Black women get breast cancer at a much younger age, are less likely to be diagnosed with early breast cancer, and are more likely to die of breast cancer, explained Dr. Margolies, who was not involved in the study.
“That’s why the guidelines that say begin at age 50 are flawed and so dangerous,” she said in an interview with this news organization. “This study is really important to highlight that we’re missing an opportunity to detect and treat breast cancer early in the Black population.”
The current study explored the optimal race- and ethnicity-specific ages to initiate breast cancer screening to address racial disparities in breast cancer mortality.
Using a nationwide population-based cross-sectional study design, the team analyzed data on 415,277 women who died of breast cancer in the United States from 2011 to 2020.
The cohort was 75% White, 15% Black, 7% Hispanic, 3% Asian or Pacific Islander, and < 1% Native American or Alaska Native. A total of 115,214 women (28%) died before age 60. The team calculated the 10-year cumulative risk of breast cancer–specific death by age and by race and ethnicity.
For those aged 40-49, breast cancer mortality was highest among Black women (27 deaths per 100,000 person-years), followed by White women (15 deaths per 100,000 person-years) and American Indian/Alaska Native, Hispanic, and Asian/Pacific Islander women (11 deaths per 100,000 person-years).
If breast screening started at age 50 for the entire population, the mean 10-year cumulative risk of dying from breast cancer would be 0.329%. Black women reached this risk threshold level at age 42, whereas non-Hispanic White women reached the threshold at age 51, American Indian/Alaska Native and Hispanic women at age 57, and Asian/Pacific Islander women at age 61.
If screening started at age 45 for all women, the mean 10-year cumulative risk of breast cancer death would be 0.235%. Black women reached this risk threshold level at age 38, non-Hispanic White women at age 46, Hispanic women at age 49, Asian/Pacific Islander women at age 50, and American Indian/Alaska Native women at age 51.
If screening started at age 40 for all women, with a mean 10-year cumulative risk of 0.154%, Black women would reach this risk threshold at age 34, White women at age 41, Hispanic women at age 43, and American Indian/Alaska Native and Asian/Pacific Islander women at age 43.
Dr. Chen and colleagues concluded that failure to consider race and ethnicity in breast cancer screening guidelines “may pose a significant risk for greater harm to a group already at increased risk.
“Changing guidelines based on readily available risk factors, such as race and ethnicity, is possible and may be the first, yet important step toward a personalized and fair screening program,” the team explained.
Dr. Margolies said she believes individualized screening recommendations will likely come, but first, all women should start screening at age 40 instead of age 50.
“Most American women are starting in their 40s, or starting at 40, because we know what the current guidelines are,” she said. “The question that this study doesn’t answer is, is age 40 young enough for the Black population? Maybe it should be 35.”
The study was supported by grants from the National Key Research-Development Program of China and from the Ten-Thousand Talents Plan of Zhejiang Province and by Start-Up Funds for Recruited Talents in Zhejiang Cancer Hospital. Dr. Chen and Dr. Margolies have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The U.S. Preventive Services Task Force currently recommends that breast cancer screening start at age 50 years, regardless of race or ethnicity.
But
The current “one-size-fits-all” policy to screen the entire female population from a certain age may be “neither fair and equitable nor optimal,” noted the authors, led by Tianhui Chen, PhD, with Zhejiang Cancer Hospital, Hangzhou, China.
The study was published online in JAMA Network Open.
Laurie R. Margolies, MD, chief of breast imaging at the Dubin Breast Center of the Mount Sinai Tisch Cancer Center in New York City, agreed.
Black women get breast cancer at a much younger age, are less likely to be diagnosed with early breast cancer, and are more likely to die of breast cancer, explained Dr. Margolies, who was not involved in the study.
“That’s why the guidelines that say begin at age 50 are flawed and so dangerous,” she said in an interview with this news organization. “This study is really important to highlight that we’re missing an opportunity to detect and treat breast cancer early in the Black population.”
The current study explored the optimal race- and ethnicity-specific ages to initiate breast cancer screening to address racial disparities in breast cancer mortality.
Using a nationwide population-based cross-sectional study design, the team analyzed data on 415,277 women who died of breast cancer in the United States from 2011 to 2020.
The cohort was 75% White, 15% Black, 7% Hispanic, 3% Asian or Pacific Islander, and < 1% Native American or Alaska Native. A total of 115,214 women (28%) died before age 60. The team calculated the 10-year cumulative risk of breast cancer–specific death by age and by race and ethnicity.
For those aged 40-49, breast cancer mortality was highest among Black women (27 deaths per 100,000 person-years), followed by White women (15 deaths per 100,000 person-years) and American Indian/Alaska Native, Hispanic, and Asian/Pacific Islander women (11 deaths per 100,000 person-years).
If breast screening started at age 50 for the entire population, the mean 10-year cumulative risk of dying from breast cancer would be 0.329%. Black women reached this risk threshold level at age 42, whereas non-Hispanic White women reached the threshold at age 51, American Indian/Alaska Native and Hispanic women at age 57, and Asian/Pacific Islander women at age 61.
If screening started at age 45 for all women, the mean 10-year cumulative risk of breast cancer death would be 0.235%. Black women reached this risk threshold level at age 38, non-Hispanic White women at age 46, Hispanic women at age 49, Asian/Pacific Islander women at age 50, and American Indian/Alaska Native women at age 51.
If screening started at age 40 for all women, with a mean 10-year cumulative risk of 0.154%, Black women would reach this risk threshold at age 34, White women at age 41, Hispanic women at age 43, and American Indian/Alaska Native and Asian/Pacific Islander women at age 43.
Dr. Chen and colleagues concluded that failure to consider race and ethnicity in breast cancer screening guidelines “may pose a significant risk for greater harm to a group already at increased risk.
“Changing guidelines based on readily available risk factors, such as race and ethnicity, is possible and may be the first, yet important step toward a personalized and fair screening program,” the team explained.
Dr. Margolies said she believes individualized screening recommendations will likely come, but first, all women should start screening at age 40 instead of age 50.
“Most American women are starting in their 40s, or starting at 40, because we know what the current guidelines are,” she said. “The question that this study doesn’t answer is, is age 40 young enough for the Black population? Maybe it should be 35.”
The study was supported by grants from the National Key Research-Development Program of China and from the Ten-Thousand Talents Plan of Zhejiang Province and by Start-Up Funds for Recruited Talents in Zhejiang Cancer Hospital. Dr. Chen and Dr. Margolies have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
The U.S. Preventive Services Task Force currently recommends that breast cancer screening start at age 50 years, regardless of race or ethnicity.
But
The current “one-size-fits-all” policy to screen the entire female population from a certain age may be “neither fair and equitable nor optimal,” noted the authors, led by Tianhui Chen, PhD, with Zhejiang Cancer Hospital, Hangzhou, China.
The study was published online in JAMA Network Open.
Laurie R. Margolies, MD, chief of breast imaging at the Dubin Breast Center of the Mount Sinai Tisch Cancer Center in New York City, agreed.
Black women get breast cancer at a much younger age, are less likely to be diagnosed with early breast cancer, and are more likely to die of breast cancer, explained Dr. Margolies, who was not involved in the study.
“That’s why the guidelines that say begin at age 50 are flawed and so dangerous,” she said in an interview with this news organization. “This study is really important to highlight that we’re missing an opportunity to detect and treat breast cancer early in the Black population.”
The current study explored the optimal race- and ethnicity-specific ages to initiate breast cancer screening to address racial disparities in breast cancer mortality.
Using a nationwide population-based cross-sectional study design, the team analyzed data on 415,277 women who died of breast cancer in the United States from 2011 to 2020.
The cohort was 75% White, 15% Black, 7% Hispanic, 3% Asian or Pacific Islander, and < 1% Native American or Alaska Native. A total of 115,214 women (28%) died before age 60. The team calculated the 10-year cumulative risk of breast cancer–specific death by age and by race and ethnicity.
For those aged 40-49, breast cancer mortality was highest among Black women (27 deaths per 100,000 person-years), followed by White women (15 deaths per 100,000 person-years) and American Indian/Alaska Native, Hispanic, and Asian/Pacific Islander women (11 deaths per 100,000 person-years).
If breast screening started at age 50 for the entire population, the mean 10-year cumulative risk of dying from breast cancer would be 0.329%. Black women reached this risk threshold level at age 42, whereas non-Hispanic White women reached the threshold at age 51, American Indian/Alaska Native and Hispanic women at age 57, and Asian/Pacific Islander women at age 61.
If screening started at age 45 for all women, the mean 10-year cumulative risk of breast cancer death would be 0.235%. Black women reached this risk threshold level at age 38, non-Hispanic White women at age 46, Hispanic women at age 49, Asian/Pacific Islander women at age 50, and American Indian/Alaska Native women at age 51.
If screening started at age 40 for all women, with a mean 10-year cumulative risk of 0.154%, Black women would reach this risk threshold at age 34, White women at age 41, Hispanic women at age 43, and American Indian/Alaska Native and Asian/Pacific Islander women at age 43.
Dr. Chen and colleagues concluded that failure to consider race and ethnicity in breast cancer screening guidelines “may pose a significant risk for greater harm to a group already at increased risk.
“Changing guidelines based on readily available risk factors, such as race and ethnicity, is possible and may be the first, yet important step toward a personalized and fair screening program,” the team explained.
Dr. Margolies said she believes individualized screening recommendations will likely come, but first, all women should start screening at age 40 instead of age 50.
“Most American women are starting in their 40s, or starting at 40, because we know what the current guidelines are,” she said. “The question that this study doesn’t answer is, is age 40 young enough for the Black population? Maybe it should be 35.”
The study was supported by grants from the National Key Research-Development Program of China and from the Ten-Thousand Talents Plan of Zhejiang Province and by Start-Up Funds for Recruited Talents in Zhejiang Cancer Hospital. Dr. Chen and Dr. Margolies have disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Early menopause, delayed HT tied to Alzheimer’s pathology
Investigators found elevated levels of tau protein in the brains of women who initiated HT more than 5 years after menopause onset, while those who started the therapy earlier had normal levels.
Tau levels were also higher in women who started menopause before age 45, either naturally or following surgery, but only in those who already had high levels of beta-amyloid.
The findings were published online in JAMA Neurology.
Hotly debated
Previous research has suggested the timing of menopause and HT initiation may be associated with AD. However, the current research is the first to suggest tau deposition may explain that link.
“There have been a lot of conflicting findings around whether HT induces risk for Alzheimer’s disease dementia or not, and – at least in our hands – our observational evidence suggests that any risk is fairly limited to those rarer cases when women might delay their initiation of HT considerably,” senior investigator Rachel Buckley, PhD, assistant investigator in neurology at Massachusetts General Hospital and assistant professor of neurology at Harvard Medical School, Boston, told this news organization.
The link between HT, dementia, and cognitive decline has been hotly debated since the initial release of findings from the Women’s Health Initiative Memory Study, reported 20 years ago.
Since then, dozens of studies have yielded conflicting evidence about HT and AD risk, with some showing a protective effect and others showing the treatment may increase AD risk.
For this study, researchers analyzed data from 292 cognitively unimpaired participants (66.1% female) in the Wisconsin Registry for Alzheimer Prevention. About half of the women had received HT.
Women had higher levels of tau measured on PET imaging than age-matched males, even after adjustment for APOE status and other potential confounders.
Higher tau levels were found in those with an earlier age at menopause (P < .001) and HT use (P = .008) compared with male sex; later menopause onset; or HT nonuse – but only in patients who also had a higher beta-amyloid burden.
Late initiation of HT (> 5 years following age at menopause) was associated with higher tau compared with early initiation (P = .001), regardless of amyloid levels.
Surprising finding
Although researchers expected to find that surgical history (specifically oophorectomy) might have a greater impact on risk, that wasn’t the case.
“Given that bilateral oophorectomy involves the removal of both ovaries, and the immediate ceasing of estrogen production, I had expected this to be the primary driver of higher tau levels,” Dr. Buckley said. “But early age at menopause – regardless of whether the genesis was natural or surgical – seemed to have similar impacts.”
These findings are the latest from Dr. Buckley’s group that indicate that women tend to have higher levels of tau than men, regardless of preexisting amyloid burden in the brain.
“We see this in healthy older women, women with dementia, and even in postmortem cases,” Dr. Buckley said. “It really remains to be seen whether women tend to accumulate tau faster in the brain than men, or whether this is simply a one-shot phenomenon that we see in observational studies at the baseline.”
“One could really flip this finding on its head and suggest that women are truly resilient to the disease,” she continued. “That is, they can hold much more tau in their brain and remain well enough to be studied, unlike men.”
Among the study’s limitations is that the data were collected at a single time point and did not include information on subsequent Alzheimer’s diagnosis or cognitive decline.
“It is important to remember that the participants in this study were not as representative of the general population in the United States, so we cannot extrapolate our findings to women from a range of socioeconomic, racial and ethnic backgrounds or education levels,” she said.
The study’s observational design left researchers unable to demonstrate causation. What’s more, the findings don’t support the assertion that hormone therapy may protect against AD, Dr. Buckley added.
“I would more confidently say that evidence from our work, and that of many others, seems to suggest that HT initiated around the time of menopause may be benign – not providing benefit or risk, at least in the context of Alzheimer’s disease risk,” she said.
Another important takeaway from the study, Dr. Buckley said, is that not all women are at high risk for AD.
“Often the headlines might make you think that most women are destined to progress to dementia, but this simply is not the case,” Dr. Buckley said. “We are now starting to really drill down on what might elevate risk for AD in women and use this information to better inform clinical trials and doctors on how best to think about treating these higher-risk groups.”
New mechanism?
Commenting on the findings, Pauline Maki, PhD, professor of psychiatry, psychology and obstetrics & gynecology at the University of Illinois at Chicago, called the study “interesting.”
“It identifies a new mechanism in humans that could underlie a possible link between sex hormones and dementia,” Dr. Maki said.
However, Dr. Maki noted that the study wasn’t randomized and information about menopause onset was self-reported.
“We must remember that many of the hypotheses about hormone therapy and brain health that came from observational studies were not validated in randomized trials, including the hypothesis that hormone therapy prevents dementia,” she said.
The findings don’t resolve the debate over hormone therapy and AD risk and point to the need for randomized, prospective studies on the topic, Dr. Maki added. Still, she said, they underscore the gender disparity in AD risk.
“It’s a good reminder to clinicians that women have a higher lifetime risk of Alzheimer’s disease and should be advised on factors that might lower their risk,” she said.
The study was funded by the National Institutes of Health. Dr. Buckley reports no relevant financial conflicts. Dr. Maki serves on the advisory boards for Astellas, Bayer, Johnson & Johnson, consults for Pfizer and Mithra, and has equity in Estrigenix, Midi-Health, and Alloy.
A version of this article originally appeared on Medscape.com.
Investigators found elevated levels of tau protein in the brains of women who initiated HT more than 5 years after menopause onset, while those who started the therapy earlier had normal levels.
Tau levels were also higher in women who started menopause before age 45, either naturally or following surgery, but only in those who already had high levels of beta-amyloid.
The findings were published online in JAMA Neurology.
Hotly debated
Previous research has suggested the timing of menopause and HT initiation may be associated with AD. However, the current research is the first to suggest tau deposition may explain that link.
“There have been a lot of conflicting findings around whether HT induces risk for Alzheimer’s disease dementia or not, and – at least in our hands – our observational evidence suggests that any risk is fairly limited to those rarer cases when women might delay their initiation of HT considerably,” senior investigator Rachel Buckley, PhD, assistant investigator in neurology at Massachusetts General Hospital and assistant professor of neurology at Harvard Medical School, Boston, told this news organization.
The link between HT, dementia, and cognitive decline has been hotly debated since the initial release of findings from the Women’s Health Initiative Memory Study, reported 20 years ago.
Since then, dozens of studies have yielded conflicting evidence about HT and AD risk, with some showing a protective effect and others showing the treatment may increase AD risk.
For this study, researchers analyzed data from 292 cognitively unimpaired participants (66.1% female) in the Wisconsin Registry for Alzheimer Prevention. About half of the women had received HT.
Women had higher levels of tau measured on PET imaging than age-matched males, even after adjustment for APOE status and other potential confounders.
Higher tau levels were found in those with an earlier age at menopause (P < .001) and HT use (P = .008) compared with male sex; later menopause onset; or HT nonuse – but only in patients who also had a higher beta-amyloid burden.
Late initiation of HT (> 5 years following age at menopause) was associated with higher tau compared with early initiation (P = .001), regardless of amyloid levels.
Surprising finding
Although researchers expected to find that surgical history (specifically oophorectomy) might have a greater impact on risk, that wasn’t the case.
“Given that bilateral oophorectomy involves the removal of both ovaries, and the immediate ceasing of estrogen production, I had expected this to be the primary driver of higher tau levels,” Dr. Buckley said. “But early age at menopause – regardless of whether the genesis was natural or surgical – seemed to have similar impacts.”
These findings are the latest from Dr. Buckley’s group that indicate that women tend to have higher levels of tau than men, regardless of preexisting amyloid burden in the brain.
“We see this in healthy older women, women with dementia, and even in postmortem cases,” Dr. Buckley said. “It really remains to be seen whether women tend to accumulate tau faster in the brain than men, or whether this is simply a one-shot phenomenon that we see in observational studies at the baseline.”
“One could really flip this finding on its head and suggest that women are truly resilient to the disease,” she continued. “That is, they can hold much more tau in their brain and remain well enough to be studied, unlike men.”
Among the study’s limitations is that the data were collected at a single time point and did not include information on subsequent Alzheimer’s diagnosis or cognitive decline.
“It is important to remember that the participants in this study were not as representative of the general population in the United States, so we cannot extrapolate our findings to women from a range of socioeconomic, racial and ethnic backgrounds or education levels,” she said.
The study’s observational design left researchers unable to demonstrate causation. What’s more, the findings don’t support the assertion that hormone therapy may protect against AD, Dr. Buckley added.
“I would more confidently say that evidence from our work, and that of many others, seems to suggest that HT initiated around the time of menopause may be benign – not providing benefit or risk, at least in the context of Alzheimer’s disease risk,” she said.
Another important takeaway from the study, Dr. Buckley said, is that not all women are at high risk for AD.
“Often the headlines might make you think that most women are destined to progress to dementia, but this simply is not the case,” Dr. Buckley said. “We are now starting to really drill down on what might elevate risk for AD in women and use this information to better inform clinical trials and doctors on how best to think about treating these higher-risk groups.”
New mechanism?
Commenting on the findings, Pauline Maki, PhD, professor of psychiatry, psychology and obstetrics & gynecology at the University of Illinois at Chicago, called the study “interesting.”
“It identifies a new mechanism in humans that could underlie a possible link between sex hormones and dementia,” Dr. Maki said.
However, Dr. Maki noted that the study wasn’t randomized and information about menopause onset was self-reported.
“We must remember that many of the hypotheses about hormone therapy and brain health that came from observational studies were not validated in randomized trials, including the hypothesis that hormone therapy prevents dementia,” she said.
The findings don’t resolve the debate over hormone therapy and AD risk and point to the need for randomized, prospective studies on the topic, Dr. Maki added. Still, she said, they underscore the gender disparity in AD risk.
“It’s a good reminder to clinicians that women have a higher lifetime risk of Alzheimer’s disease and should be advised on factors that might lower their risk,” she said.
The study was funded by the National Institutes of Health. Dr. Buckley reports no relevant financial conflicts. Dr. Maki serves on the advisory boards for Astellas, Bayer, Johnson & Johnson, consults for Pfizer and Mithra, and has equity in Estrigenix, Midi-Health, and Alloy.
A version of this article originally appeared on Medscape.com.
Investigators found elevated levels of tau protein in the brains of women who initiated HT more than 5 years after menopause onset, while those who started the therapy earlier had normal levels.
Tau levels were also higher in women who started menopause before age 45, either naturally or following surgery, but only in those who already had high levels of beta-amyloid.
The findings were published online in JAMA Neurology.
Hotly debated
Previous research has suggested the timing of menopause and HT initiation may be associated with AD. However, the current research is the first to suggest tau deposition may explain that link.
“There have been a lot of conflicting findings around whether HT induces risk for Alzheimer’s disease dementia or not, and – at least in our hands – our observational evidence suggests that any risk is fairly limited to those rarer cases when women might delay their initiation of HT considerably,” senior investigator Rachel Buckley, PhD, assistant investigator in neurology at Massachusetts General Hospital and assistant professor of neurology at Harvard Medical School, Boston, told this news organization.
The link between HT, dementia, and cognitive decline has been hotly debated since the initial release of findings from the Women’s Health Initiative Memory Study, reported 20 years ago.
Since then, dozens of studies have yielded conflicting evidence about HT and AD risk, with some showing a protective effect and others showing the treatment may increase AD risk.
For this study, researchers analyzed data from 292 cognitively unimpaired participants (66.1% female) in the Wisconsin Registry for Alzheimer Prevention. About half of the women had received HT.
Women had higher levels of tau measured on PET imaging than age-matched males, even after adjustment for APOE status and other potential confounders.
Higher tau levels were found in those with an earlier age at menopause (P < .001) and HT use (P = .008) compared with male sex; later menopause onset; or HT nonuse – but only in patients who also had a higher beta-amyloid burden.
Late initiation of HT (> 5 years following age at menopause) was associated with higher tau compared with early initiation (P = .001), regardless of amyloid levels.
Surprising finding
Although researchers expected to find that surgical history (specifically oophorectomy) might have a greater impact on risk, that wasn’t the case.
“Given that bilateral oophorectomy involves the removal of both ovaries, and the immediate ceasing of estrogen production, I had expected this to be the primary driver of higher tau levels,” Dr. Buckley said. “But early age at menopause – regardless of whether the genesis was natural or surgical – seemed to have similar impacts.”
These findings are the latest from Dr. Buckley’s group that indicate that women tend to have higher levels of tau than men, regardless of preexisting amyloid burden in the brain.
“We see this in healthy older women, women with dementia, and even in postmortem cases,” Dr. Buckley said. “It really remains to be seen whether women tend to accumulate tau faster in the brain than men, or whether this is simply a one-shot phenomenon that we see in observational studies at the baseline.”
“One could really flip this finding on its head and suggest that women are truly resilient to the disease,” she continued. “That is, they can hold much more tau in their brain and remain well enough to be studied, unlike men.”
Among the study’s limitations is that the data were collected at a single time point and did not include information on subsequent Alzheimer’s diagnosis or cognitive decline.
“It is important to remember that the participants in this study were not as representative of the general population in the United States, so we cannot extrapolate our findings to women from a range of socioeconomic, racial and ethnic backgrounds or education levels,” she said.
The study’s observational design left researchers unable to demonstrate causation. What’s more, the findings don’t support the assertion that hormone therapy may protect against AD, Dr. Buckley added.
“I would more confidently say that evidence from our work, and that of many others, seems to suggest that HT initiated around the time of menopause may be benign – not providing benefit or risk, at least in the context of Alzheimer’s disease risk,” she said.
Another important takeaway from the study, Dr. Buckley said, is that not all women are at high risk for AD.
“Often the headlines might make you think that most women are destined to progress to dementia, but this simply is not the case,” Dr. Buckley said. “We are now starting to really drill down on what might elevate risk for AD in women and use this information to better inform clinical trials and doctors on how best to think about treating these higher-risk groups.”
New mechanism?
Commenting on the findings, Pauline Maki, PhD, professor of psychiatry, psychology and obstetrics & gynecology at the University of Illinois at Chicago, called the study “interesting.”
“It identifies a new mechanism in humans that could underlie a possible link between sex hormones and dementia,” Dr. Maki said.
However, Dr. Maki noted that the study wasn’t randomized and information about menopause onset was self-reported.
“We must remember that many of the hypotheses about hormone therapy and brain health that came from observational studies were not validated in randomized trials, including the hypothesis that hormone therapy prevents dementia,” she said.
The findings don’t resolve the debate over hormone therapy and AD risk and point to the need for randomized, prospective studies on the topic, Dr. Maki added. Still, she said, they underscore the gender disparity in AD risk.
“It’s a good reminder to clinicians that women have a higher lifetime risk of Alzheimer’s disease and should be advised on factors that might lower their risk,” she said.
The study was funded by the National Institutes of Health. Dr. Buckley reports no relevant financial conflicts. Dr. Maki serves on the advisory boards for Astellas, Bayer, Johnson & Johnson, consults for Pfizer and Mithra, and has equity in Estrigenix, Midi-Health, and Alloy.
A version of this article originally appeared on Medscape.com.
FROM JAMA NEUROLOGY