User login
Inadequate differential proves fatal ... Death by fentanyl patch and methadone ... more
Culture results go undiscussed, man suffers stroke
TWO WEEKS AFTER PROSTATE SURGERY, a 76-year-old man went to the ED because he was having trouble urinating. The ED physician catheterized the patient, ordered a urine culture, and discharged him.
The culture results, showing methicillin-resistant Staphylococcus aureus, were sent to a printer in the ED twice, as was the usual practice, but evidently no one saw them.
The patient returned to the ED 2 weeks after his initial visit with the same complaint of difficult urination and was seen by the same physician. The physician again discharged him with a catheter but without mentioning the culture results. Two days later, the patient suffered a stroke, which paralyzed his left side.
PLAINTIFF’S CLAIM The bacteria had spread from the patient’s urine to his bloodstream, sparking a cascade of events that led to the stroke.
THE DEFENSE No information about the defense is available.
VERDICT $2.25 million New Jersey settlement.
COMMENT The repeated missed opportunities to diagnose and treat this patient’s infection were regrettable—and costly.
Inadequate differential proves fatal
SHORTNESS OF BREATH led a 52-year-old woman to visit her medical group, where she was a long-time patient. The family practitioner who saw her noted tachycardia and ordered an electrocardiogram, which was abnormal. The physician also ordered a chest x-ray and, because the woman had a history of anemia, a complete blood count and a number of other blood tests. He subsequently called the patient at home to tell her that the blood tests were normal and she didn’t have anemia.
Three days later, the patient went to an urgent care center complaining of shortness of breath and tightness in her chest. A pulmonary embolism was diagnosed, and she was transferred to a hospital ED. Later that evening, a code blue was called and the patient was resuscitated. She died the following day.
PLAINTIFF’S CLAIM The doctor assumed that the patient had anemia and failed to develop a differential diagnosis. The patient had risk factors for pulmonary embolism—obesity and the use of an ethinyl estradiol-etonogestrel vaginal contraceptive ring—which should have prompted the doctor to consider that possibility. If he had done so, the pulmonary embolism would have been diagnosed and the patient’s death prevented.
THE DEFENSE The patient’s presentation wasn’t typical for pulmonary embolism, and there wasn’t any way to know whether an earlier diagnosis would have resulted in survival.
VERDICT $1.9 million California verdict.
COMMENT Although pulmonary embolism can be a challenging diagnosis to make, it needs to be considered carefully in all patients with shortness of breath, chest pain, or poorly defined pulmonary or cardiac symptoms.
The correct diagnosis comes too late
FLU-LIKE SYMPTOMS AND AN IRREGULAR HEART RATE prompted a man to go to the ED, where the physician diagnosed a viral infection, prescribed pain medication, and discharged him. The following day, a laboratory report indicating a staph infection was sent to an ED secretary, but the patient wasn’t told the results.
The patient returned to the hospital 2 days later in a confused state. Tests revealed a staph infection and meningitis, for which the patient received antibiotics. A week later, the patient suffered a stroke, resulting in diminished cognitive ability, impaired vision, and right-sided motor deficits.
PLAINTIFF’S CLAIM The white blood cell count and C-reactive protein level measured at the patient’s first visit to the ED would have led to a diagnosis of bacterial infection. The patient should have been admitted to the hospital and given antibiotics at that time.
THE DEFENSE The original diagnosis was reasonable.
VERDICT Confidential settlement with the hospital. $900,000 net verdict against the physician in New Jersey.
COMMENT Lab reports gone awry and the lack of a fail-safe for abnormal tests result in a $900,000 judgment. Do you have adequate systems in place to avoid a communication failure like this one?
Slow response turns a bad situation into a disaster
A 66-YEAR-OLD MAN on warfarin therapy for chronic atrial fibrillation and a transient ischemic attack underwent lithotripsy for kidney stones. Three days after the lithotripsy, he went to the ED complaining of severe flank pain. A computed tomography (CT) scan of the abdomen showed a large retroperitoneal hematoma and prominent perinephric and pararenal hemorrhages.
The patient remained on a gurney in the hallway of the ED in deteriorating condition until he was admitted to the intensive care unit, by which time his condition was critical. He died the next day.
PLAINTIFF’S CLAIM The ED physician and admitting urologists failed to monitor and treat the patient’s active hemorrhage for 9 hours. They didn’t order coagulation studies or respond to signs of escalating hemorrhagic shock. They failed to seek timely consults from surgery and interventional radiology.
THE DEFENSE No information about the defense is available.
VERDICT $825,000 Virginia settlement.
COMMENT Preventing complications of anticoagulation is hard enough; the lack of a timely response in this case made a bad outcome disastrous.
Were steps taken quickly enough?
SEVERE LOWER ABDOMINAL PAIN prompted a 52-year-old woman to go to the ED. She said she hadn’t had a bowel movement in almost a week. The ED physician, in consultation with the attending physician, admitted her to the hospital and ordered intravenous fluids and a soap suds enema, which didn’t relieve the constipation. The patient’s vital signs deteriorated, and she was crying and restless.
When the attending physician saw the patient almost 3 hours after admission, she had a fever of 101.4°F. He ordered additional tests, a computed tomography (CT) scan, and antibiotics, but didn’t order them STAT.
About 1½ hours later, a house physician examined the patient, and, after speaking with the attending physician, transferred her to a step-down telemetry unit. About 1½ hours after the transfer, a nurse called the house physician to report that the patient’s condition was worsening. The house physician ordered pain relievers and a second enema but didn’t come to the hospital.
Because the patient wasn’t in the intensive care unit, no one checked on her again for 3½ hours. When the nurse did check, she found the patient pale, cold, and turning blue. The nurse called the house physician, who came to the hospital. The patient had a fever of 102.4°F and her blood pressure couldn’t be measured.
After speaking with the attending physician, the house physician had the patient admitted to the ICU and also ordered a STAT surgical consultation and CT scan. In the meantime, the patient went into cardiac arrest and couldn’t be revived. Death was caused by peritonitis with sepsis resulting from a large intestinal obstruction.
PLAINTIFF’S CLAIM The patient showed early signs of sepsis. She should have undergone testing sooner and been transferred to the ICU earlier.
THE DEFENSE The doctors claimed that all their actions were appropriate and that the actions suggested by the plaintiff wouldn’t have resulted in the patient’s survival.
VERDICT $3.8 million Pennsylvania verdict.
COMMENT Prompt evaluation and monitoring of this patient might have prevented death and a substantial verdict.
2 analgesic calamities: Death by fentanyl patch …
AFTER A WEEK OF INCREASING BACK PAIN, which had begun to shoot down his right leg, a 37-year-old man went to the ED. He was examined and given prescriptions for pain killers, including acetaminophen and hydrocodone, and muscle relaxants and discharged with instructions to return in 3 days for magnetic resonance imaging (MRI).
While he was at the hospital for the MRI, the patient returned to the ED because he was still in pain and his acetaminophen-hydrocodone prescription was running out. The ED physician prescribed a 0.75-mg fentanyl transdermal patch and instructed the patient to put it on his chest.
Three days later, the patient filled the prescription and applied the patch. The following day, his girlfriend found him dead in bed. Postmortem toxicology results showed a blood fentanyl level of 9.85 ng/mL, markedly higher than the therapeutic level. Respiratory failure caused by fentanyl toxicity was cited as the cause of death.
PLAINTIFF’S CLAIM The ED physician prescribed an excessive dose of fentanyl.
THE DEFENSE A defective patch or misuse of the patch caused the patient’s death.
VERDICT $1.2 million Indiana verdict.
… and methadone
A 36-YEAR-OLD MAN started treatment with a pain specialist for pain arising from a back problem, for which he had taken pain medication previously. The pain specialist prescribed methadone, 360 10-mg tablets. The prescription limited the patient to 2 tablets every 4 hours for a maximum dosage of 12 tablets (120 mg) per day.
Three days after the patient filled the prescription, he was found dead. An autopsy determined the cause of death to be drug toxicity from methadone. At the time the patient died, the bottle of methadone tablets contained 342 tablets, indicating that he had taken only 18 tablets, well within the maximum dosage authorized by the prescription.
PLAINTIFF’S CLAIM The prescribed methadone dosage was excessive for a patient just beginning to use the drug. A proper initial dosage is between 2.5 and 10 mg every 8 to 12 hours for a maximum of 30 mg per day.
THE DEFENSE No information about the defense is available.
VERDICT Confidential Utah settlement.
COMMENT These 2 cases have a common thread. The effects of opioids are often idiosyncratic. A plan for careful monitoring and follow-up should be prepared at initiation of treatment and when escalating the dosage.
Culture results go undiscussed, man suffers stroke
TWO WEEKS AFTER PROSTATE SURGERY, a 76-year-old man went to the ED because he was having trouble urinating. The ED physician catheterized the patient, ordered a urine culture, and discharged him.
The culture results, showing methicillin-resistant Staphylococcus aureus, were sent to a printer in the ED twice, as was the usual practice, but evidently no one saw them.
The patient returned to the ED 2 weeks after his initial visit with the same complaint of difficult urination and was seen by the same physician. The physician again discharged him with a catheter but without mentioning the culture results. Two days later, the patient suffered a stroke, which paralyzed his left side.
PLAINTIFF’S CLAIM The bacteria had spread from the patient’s urine to his bloodstream, sparking a cascade of events that led to the stroke.
THE DEFENSE No information about the defense is available.
VERDICT $2.25 million New Jersey settlement.
COMMENT The repeated missed opportunities to diagnose and treat this patient’s infection were regrettable—and costly.
Inadequate differential proves fatal
SHORTNESS OF BREATH led a 52-year-old woman to visit her medical group, where she was a long-time patient. The family practitioner who saw her noted tachycardia and ordered an electrocardiogram, which was abnormal. The physician also ordered a chest x-ray and, because the woman had a history of anemia, a complete blood count and a number of other blood tests. He subsequently called the patient at home to tell her that the blood tests were normal and she didn’t have anemia.
Three days later, the patient went to an urgent care center complaining of shortness of breath and tightness in her chest. A pulmonary embolism was diagnosed, and she was transferred to a hospital ED. Later that evening, a code blue was called and the patient was resuscitated. She died the following day.
PLAINTIFF’S CLAIM The doctor assumed that the patient had anemia and failed to develop a differential diagnosis. The patient had risk factors for pulmonary embolism—obesity and the use of an ethinyl estradiol-etonogestrel vaginal contraceptive ring—which should have prompted the doctor to consider that possibility. If he had done so, the pulmonary embolism would have been diagnosed and the patient’s death prevented.
THE DEFENSE The patient’s presentation wasn’t typical for pulmonary embolism, and there wasn’t any way to know whether an earlier diagnosis would have resulted in survival.
VERDICT $1.9 million California verdict.
COMMENT Although pulmonary embolism can be a challenging diagnosis to make, it needs to be considered carefully in all patients with shortness of breath, chest pain, or poorly defined pulmonary or cardiac symptoms.
The correct diagnosis comes too late
FLU-LIKE SYMPTOMS AND AN IRREGULAR HEART RATE prompted a man to go to the ED, where the physician diagnosed a viral infection, prescribed pain medication, and discharged him. The following day, a laboratory report indicating a staph infection was sent to an ED secretary, but the patient wasn’t told the results.
The patient returned to the hospital 2 days later in a confused state. Tests revealed a staph infection and meningitis, for which the patient received antibiotics. A week later, the patient suffered a stroke, resulting in diminished cognitive ability, impaired vision, and right-sided motor deficits.
PLAINTIFF’S CLAIM The white blood cell count and C-reactive protein level measured at the patient’s first visit to the ED would have led to a diagnosis of bacterial infection. The patient should have been admitted to the hospital and given antibiotics at that time.
THE DEFENSE The original diagnosis was reasonable.
VERDICT Confidential settlement with the hospital. $900,000 net verdict against the physician in New Jersey.
COMMENT Lab reports gone awry and the lack of a fail-safe for abnormal tests result in a $900,000 judgment. Do you have adequate systems in place to avoid a communication failure like this one?
Slow response turns a bad situation into a disaster
A 66-YEAR-OLD MAN on warfarin therapy for chronic atrial fibrillation and a transient ischemic attack underwent lithotripsy for kidney stones. Three days after the lithotripsy, he went to the ED complaining of severe flank pain. A computed tomography (CT) scan of the abdomen showed a large retroperitoneal hematoma and prominent perinephric and pararenal hemorrhages.
The patient remained on a gurney in the hallway of the ED in deteriorating condition until he was admitted to the intensive care unit, by which time his condition was critical. He died the next day.
PLAINTIFF’S CLAIM The ED physician and admitting urologists failed to monitor and treat the patient’s active hemorrhage for 9 hours. They didn’t order coagulation studies or respond to signs of escalating hemorrhagic shock. They failed to seek timely consults from surgery and interventional radiology.
THE DEFENSE No information about the defense is available.
VERDICT $825,000 Virginia settlement.
COMMENT Preventing complications of anticoagulation is hard enough; the lack of a timely response in this case made a bad outcome disastrous.
Were steps taken quickly enough?
SEVERE LOWER ABDOMINAL PAIN prompted a 52-year-old woman to go to the ED. She said she hadn’t had a bowel movement in almost a week. The ED physician, in consultation with the attending physician, admitted her to the hospital and ordered intravenous fluids and a soap suds enema, which didn’t relieve the constipation. The patient’s vital signs deteriorated, and she was crying and restless.
When the attending physician saw the patient almost 3 hours after admission, she had a fever of 101.4°F. He ordered additional tests, a computed tomography (CT) scan, and antibiotics, but didn’t order them STAT.
About 1½ hours later, a house physician examined the patient, and, after speaking with the attending physician, transferred her to a step-down telemetry unit. About 1½ hours after the transfer, a nurse called the house physician to report that the patient’s condition was worsening. The house physician ordered pain relievers and a second enema but didn’t come to the hospital.
Because the patient wasn’t in the intensive care unit, no one checked on her again for 3½ hours. When the nurse did check, she found the patient pale, cold, and turning blue. The nurse called the house physician, who came to the hospital. The patient had a fever of 102.4°F and her blood pressure couldn’t be measured.
After speaking with the attending physician, the house physician had the patient admitted to the ICU and also ordered a STAT surgical consultation and CT scan. In the meantime, the patient went into cardiac arrest and couldn’t be revived. Death was caused by peritonitis with sepsis resulting from a large intestinal obstruction.
PLAINTIFF’S CLAIM The patient showed early signs of sepsis. She should have undergone testing sooner and been transferred to the ICU earlier.
THE DEFENSE The doctors claimed that all their actions were appropriate and that the actions suggested by the plaintiff wouldn’t have resulted in the patient’s survival.
VERDICT $3.8 million Pennsylvania verdict.
COMMENT Prompt evaluation and monitoring of this patient might have prevented death and a substantial verdict.
2 analgesic calamities: Death by fentanyl patch …
AFTER A WEEK OF INCREASING BACK PAIN, which had begun to shoot down his right leg, a 37-year-old man went to the ED. He was examined and given prescriptions for pain killers, including acetaminophen and hydrocodone, and muscle relaxants and discharged with instructions to return in 3 days for magnetic resonance imaging (MRI).
While he was at the hospital for the MRI, the patient returned to the ED because he was still in pain and his acetaminophen-hydrocodone prescription was running out. The ED physician prescribed a 0.75-mg fentanyl transdermal patch and instructed the patient to put it on his chest.
Three days later, the patient filled the prescription and applied the patch. The following day, his girlfriend found him dead in bed. Postmortem toxicology results showed a blood fentanyl level of 9.85 ng/mL, markedly higher than the therapeutic level. Respiratory failure caused by fentanyl toxicity was cited as the cause of death.
PLAINTIFF’S CLAIM The ED physician prescribed an excessive dose of fentanyl.
THE DEFENSE A defective patch or misuse of the patch caused the patient’s death.
VERDICT $1.2 million Indiana verdict.
… and methadone
A 36-YEAR-OLD MAN started treatment with a pain specialist for pain arising from a back problem, for which he had taken pain medication previously. The pain specialist prescribed methadone, 360 10-mg tablets. The prescription limited the patient to 2 tablets every 4 hours for a maximum dosage of 12 tablets (120 mg) per day.
Three days after the patient filled the prescription, he was found dead. An autopsy determined the cause of death to be drug toxicity from methadone. At the time the patient died, the bottle of methadone tablets contained 342 tablets, indicating that he had taken only 18 tablets, well within the maximum dosage authorized by the prescription.
PLAINTIFF’S CLAIM The prescribed methadone dosage was excessive for a patient just beginning to use the drug. A proper initial dosage is between 2.5 and 10 mg every 8 to 12 hours for a maximum of 30 mg per day.
THE DEFENSE No information about the defense is available.
VERDICT Confidential Utah settlement.
COMMENT These 2 cases have a common thread. The effects of opioids are often idiosyncratic. A plan for careful monitoring and follow-up should be prepared at initiation of treatment and when escalating the dosage.
Culture results go undiscussed, man suffers stroke
TWO WEEKS AFTER PROSTATE SURGERY, a 76-year-old man went to the ED because he was having trouble urinating. The ED physician catheterized the patient, ordered a urine culture, and discharged him.
The culture results, showing methicillin-resistant Staphylococcus aureus, were sent to a printer in the ED twice, as was the usual practice, but evidently no one saw them.
The patient returned to the ED 2 weeks after his initial visit with the same complaint of difficult urination and was seen by the same physician. The physician again discharged him with a catheter but without mentioning the culture results. Two days later, the patient suffered a stroke, which paralyzed his left side.
PLAINTIFF’S CLAIM The bacteria had spread from the patient’s urine to his bloodstream, sparking a cascade of events that led to the stroke.
THE DEFENSE No information about the defense is available.
VERDICT $2.25 million New Jersey settlement.
COMMENT The repeated missed opportunities to diagnose and treat this patient’s infection were regrettable—and costly.
Inadequate differential proves fatal
SHORTNESS OF BREATH led a 52-year-old woman to visit her medical group, where she was a long-time patient. The family practitioner who saw her noted tachycardia and ordered an electrocardiogram, which was abnormal. The physician also ordered a chest x-ray and, because the woman had a history of anemia, a complete blood count and a number of other blood tests. He subsequently called the patient at home to tell her that the blood tests were normal and she didn’t have anemia.
Three days later, the patient went to an urgent care center complaining of shortness of breath and tightness in her chest. A pulmonary embolism was diagnosed, and she was transferred to a hospital ED. Later that evening, a code blue was called and the patient was resuscitated. She died the following day.
PLAINTIFF’S CLAIM The doctor assumed that the patient had anemia and failed to develop a differential diagnosis. The patient had risk factors for pulmonary embolism—obesity and the use of an ethinyl estradiol-etonogestrel vaginal contraceptive ring—which should have prompted the doctor to consider that possibility. If he had done so, the pulmonary embolism would have been diagnosed and the patient’s death prevented.
THE DEFENSE The patient’s presentation wasn’t typical for pulmonary embolism, and there wasn’t any way to know whether an earlier diagnosis would have resulted in survival.
VERDICT $1.9 million California verdict.
COMMENT Although pulmonary embolism can be a challenging diagnosis to make, it needs to be considered carefully in all patients with shortness of breath, chest pain, or poorly defined pulmonary or cardiac symptoms.
The correct diagnosis comes too late
FLU-LIKE SYMPTOMS AND AN IRREGULAR HEART RATE prompted a man to go to the ED, where the physician diagnosed a viral infection, prescribed pain medication, and discharged him. The following day, a laboratory report indicating a staph infection was sent to an ED secretary, but the patient wasn’t told the results.
The patient returned to the hospital 2 days later in a confused state. Tests revealed a staph infection and meningitis, for which the patient received antibiotics. A week later, the patient suffered a stroke, resulting in diminished cognitive ability, impaired vision, and right-sided motor deficits.
PLAINTIFF’S CLAIM The white blood cell count and C-reactive protein level measured at the patient’s first visit to the ED would have led to a diagnosis of bacterial infection. The patient should have been admitted to the hospital and given antibiotics at that time.
THE DEFENSE The original diagnosis was reasonable.
VERDICT Confidential settlement with the hospital. $900,000 net verdict against the physician in New Jersey.
COMMENT Lab reports gone awry and the lack of a fail-safe for abnormal tests result in a $900,000 judgment. Do you have adequate systems in place to avoid a communication failure like this one?
Slow response turns a bad situation into a disaster
A 66-YEAR-OLD MAN on warfarin therapy for chronic atrial fibrillation and a transient ischemic attack underwent lithotripsy for kidney stones. Three days after the lithotripsy, he went to the ED complaining of severe flank pain. A computed tomography (CT) scan of the abdomen showed a large retroperitoneal hematoma and prominent perinephric and pararenal hemorrhages.
The patient remained on a gurney in the hallway of the ED in deteriorating condition until he was admitted to the intensive care unit, by which time his condition was critical. He died the next day.
PLAINTIFF’S CLAIM The ED physician and admitting urologists failed to monitor and treat the patient’s active hemorrhage for 9 hours. They didn’t order coagulation studies or respond to signs of escalating hemorrhagic shock. They failed to seek timely consults from surgery and interventional radiology.
THE DEFENSE No information about the defense is available.
VERDICT $825,000 Virginia settlement.
COMMENT Preventing complications of anticoagulation is hard enough; the lack of a timely response in this case made a bad outcome disastrous.
Were steps taken quickly enough?
SEVERE LOWER ABDOMINAL PAIN prompted a 52-year-old woman to go to the ED. She said she hadn’t had a bowel movement in almost a week. The ED physician, in consultation with the attending physician, admitted her to the hospital and ordered intravenous fluids and a soap suds enema, which didn’t relieve the constipation. The patient’s vital signs deteriorated, and she was crying and restless.
When the attending physician saw the patient almost 3 hours after admission, she had a fever of 101.4°F. He ordered additional tests, a computed tomography (CT) scan, and antibiotics, but didn’t order them STAT.
About 1½ hours later, a house physician examined the patient, and, after speaking with the attending physician, transferred her to a step-down telemetry unit. About 1½ hours after the transfer, a nurse called the house physician to report that the patient’s condition was worsening. The house physician ordered pain relievers and a second enema but didn’t come to the hospital.
Because the patient wasn’t in the intensive care unit, no one checked on her again for 3½ hours. When the nurse did check, she found the patient pale, cold, and turning blue. The nurse called the house physician, who came to the hospital. The patient had a fever of 102.4°F and her blood pressure couldn’t be measured.
After speaking with the attending physician, the house physician had the patient admitted to the ICU and also ordered a STAT surgical consultation and CT scan. In the meantime, the patient went into cardiac arrest and couldn’t be revived. Death was caused by peritonitis with sepsis resulting from a large intestinal obstruction.
PLAINTIFF’S CLAIM The patient showed early signs of sepsis. She should have undergone testing sooner and been transferred to the ICU earlier.
THE DEFENSE The doctors claimed that all their actions were appropriate and that the actions suggested by the plaintiff wouldn’t have resulted in the patient’s survival.
VERDICT $3.8 million Pennsylvania verdict.
COMMENT Prompt evaluation and monitoring of this patient might have prevented death and a substantial verdict.
2 analgesic calamities: Death by fentanyl patch …
AFTER A WEEK OF INCREASING BACK PAIN, which had begun to shoot down his right leg, a 37-year-old man went to the ED. He was examined and given prescriptions for pain killers, including acetaminophen and hydrocodone, and muscle relaxants and discharged with instructions to return in 3 days for magnetic resonance imaging (MRI).
While he was at the hospital for the MRI, the patient returned to the ED because he was still in pain and his acetaminophen-hydrocodone prescription was running out. The ED physician prescribed a 0.75-mg fentanyl transdermal patch and instructed the patient to put it on his chest.
Three days later, the patient filled the prescription and applied the patch. The following day, his girlfriend found him dead in bed. Postmortem toxicology results showed a blood fentanyl level of 9.85 ng/mL, markedly higher than the therapeutic level. Respiratory failure caused by fentanyl toxicity was cited as the cause of death.
PLAINTIFF’S CLAIM The ED physician prescribed an excessive dose of fentanyl.
THE DEFENSE A defective patch or misuse of the patch caused the patient’s death.
VERDICT $1.2 million Indiana verdict.
… and methadone
A 36-YEAR-OLD MAN started treatment with a pain specialist for pain arising from a back problem, for which he had taken pain medication previously. The pain specialist prescribed methadone, 360 10-mg tablets. The prescription limited the patient to 2 tablets every 4 hours for a maximum dosage of 12 tablets (120 mg) per day.
Three days after the patient filled the prescription, he was found dead. An autopsy determined the cause of death to be drug toxicity from methadone. At the time the patient died, the bottle of methadone tablets contained 342 tablets, indicating that he had taken only 18 tablets, well within the maximum dosage authorized by the prescription.
PLAINTIFF’S CLAIM The prescribed methadone dosage was excessive for a patient just beginning to use the drug. A proper initial dosage is between 2.5 and 10 mg every 8 to 12 hours for a maximum of 30 mg per day.
THE DEFENSE No information about the defense is available.
VERDICT Confidential Utah settlement.
COMMENT These 2 cases have a common thread. The effects of opioids are often idiosyncratic. A plan for careful monitoring and follow-up should be prepared at initiation of treatment and when escalating the dosage.
Postprostatectomy incontinence? Here’s help
Recommend behavioral therapy—incorporating pelvic floor muscle training and bladder control strategies—to men experiencing incontinence after prostatectomy.1
STRENGTH OF RECOMMENDATION
B: Based on a single unblinded randomized controlled trial (RCT).
Goode PS, Burgio KL, Johnson TM, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA. 2011;305:151-159.1
ILLUSTRATIVE CASE
Mr. H, age 75, underwent radical prostatectomy for prostate cancer a year ago. Since then, he leaks urine when he coughs and occasionally has complete loss of bladder control. His lack of control has forced him to curtail many of his normal activities and he wants to know what help you can provide.
Routine prostate-specific antigen (PSA) screening has led to an increase in the diagnosis of localized prostate cancer, and prostatectomy is a common treatment. Approximately 90,000 US men undergo radical prostatectomy each year,2 and most are left with some degree of incontinence.
Years later, bladder control problems remain
Surgical advances have attempted to minimize nerve and bladder neck damage, but some degree of incontinence is still common after radical prostatectomy. According to the 2000 Prostate Cancer Outcomes Study, 80% of men experienced some incontinence 6 months after radical prostatectomy. After 2 years, 68% of men still had some degree of incontinence,3 and 8% reported frequent or total incontinence. Five years after prostatectomy, only 35% of patients reported complete bladder control vs 87% presurgery.4
A 2004 systematic review showed that behavioral therapy (mostly biofeedback) reduced symptoms in patients with all types of urinary incontinence.5 Many studies, including a 2010 Cochrane review,6 have shown that women with incontinence benefit from pelvic floor muscle training. No randomized trials have assessed the benefit of behavioral therapy for men with incontinence related to postprostatectomy—until now.
STUDY SUMMARY: Behavioral therapy has long-term payoff
The study by Goode et al was an RCT of behavioral therapy for men who had urinary incontinence after radical prostatectomy and whose symptoms persisted more than a year later. It included patients with ≥3 episodes of incontinence per week. Men were excluded if they were undergoing other treatment for prostate cancer, had a high postvoid residual volume or a history of certain bladder surgeries, or were unable to reliably report symptoms. Participants were permitted to continue taking medication for incontinence, with the exception of anticholinergics.
Participants (n=208 from 3 sites) were randomized to one of 3 arms in a blinded fashion with concealed allocation: behavioral therapy alone, behavioral therapy and biofeedback with electrical stimulation, or a control group of men who could elect to try these therapies at a later date.
Behavioral therapy consisted of 4 visits with a physician or nurse practitioner over an 8-week period. At the first visit, patients received instruction in pelvic floor muscle training. Patients then practiced contraction and relaxation exercises and urinary flow control at home. At the second visit, patients learned techniques to avoid episodes of incontinence, such as performing pelvic muscle contractions with stress symptom triggers. During the final 2 visits, patients received advice about control of persistent problems identified in symptom diaries they were required to keep. In addition, continued daily pelvic floor exercises were recommended at the last session.
Men in the group that received biofeedback with electrical stimulation had the same visit schedule, but received additional pelvic floor training—with electrode-mediated feedback and electrical stimulation of pelvic floor muscles during each visit and daily at home. Patients in the control group had the same visit frequency but received no treatment. After 8 weeks, however, the controls were given the opportunity to try behavioral therapy.
Baseline characteristics and attrition rates were similar in all 3 groups. Outcomes were based on an intention-to-treat analysis. At 8 weeks, men receiving behavioral therapy, with or without electrical stimulation and biofeedback, experienced a 55% decrease in incontinence (from 28 episodes per week at baseline to 13 per week); patients in the control group had a 24% decrease (from 25 episodes to 20 per week) (P=.001). More patients in the behavioral groups were completely continent at 8 weeks (16% vs 6% for the controls); the number needed to treat to achieve complete continence was 10. Electrical stimulation and biofeedback provided no added benefit compared with behavioral therapy alone.
Patients in the 2 treatment groups also had clinically significant benefits in some quality-of-life measures (impact of urinary symptoms on travel, emotion, and voiding) and in symptom-specific quality-of-life scores. Patient satisfaction at 8 weeks was higher in the treatment groups: 26 of the 58 men who received behavioral therapy were “delighted, pleased, or mostly satisfied,” vs 9 of 60 in the control group (P=.006 for overall group difference).
Adherence to the behavioral therapy protocol was 100% at 8 weeks and remained high (91%) one year later. Improvement in symptoms continued at one year, with patients in both treatment groups reporting a clinically significant (50%) reduction in incontinence episodes compared with baseline.
WHAT’S NEW: We have evidence-based help for postprostatectomy incontinence
We now have evidence that an 8-week program of pelvic floor training and bladder control strategies reduces the frequency of incontinence in men who have undergone radical prostatectomy.
CAVEATS: The effects of time weren’t factored in
Patients were obviously aware of group assignment, so there is the possibility of treatment bias contributing to the positive self-reported outcomes. While the treatment groups showed both a greater initial improvement and persistent improvement in their symptoms at one year, symptoms of patients in the control group were not measured after a year, so the sustained improvement could reflect resolution of incontinence with time.
CHALLENGES TO IMPLEMENTATION: Locating clinicians who can train patients
The type of behavioral therapy featured in this study may not be easily accessible to all patients. The researchers suggest consulting the National Association for Continence (http://www.nafc.org), a private nonprofit organization whose members include physical therapists, nurses, and physicians. They also cite the Wound Ostomy and Continence Nurses Society (http://www.wocn.org) as a resource in locating nurses who provide these services.
Acknowledgement
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Click here to view PURL METHODOLOGY
1. Goode PS, Burgio KL, Johnson TM, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA. 2011;305:151-159.
2. Barbash GI, Glied SA. New technology and health care costs—the case of robot-assisted surgery. N Engl J Med. 2010;363:701-704.
3. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA. 2000;283:354-360.
4. Penson DF, McLerran D, Feng Z, et al. 5-year urinary and sexual outcomes after radical prostatectomy: results from the prostate cancer outcomes study. J Urol. 2005;173:1701-1705.
5. Teunisse TA, de Jonge A, van Weel C, et al. Treating urinary incontinence in the elderly—conservative therapies that work: a systematic review. J Fam Pract. 2004;53:25-30, 32.
6. Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2010;(1):CD005654.-
Recommend behavioral therapy—incorporating pelvic floor muscle training and bladder control strategies—to men experiencing incontinence after prostatectomy.1
STRENGTH OF RECOMMENDATION
B: Based on a single unblinded randomized controlled trial (RCT).
Goode PS, Burgio KL, Johnson TM, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA. 2011;305:151-159.1
ILLUSTRATIVE CASE
Mr. H, age 75, underwent radical prostatectomy for prostate cancer a year ago. Since then, he leaks urine when he coughs and occasionally has complete loss of bladder control. His lack of control has forced him to curtail many of his normal activities and he wants to know what help you can provide.
Routine prostate-specific antigen (PSA) screening has led to an increase in the diagnosis of localized prostate cancer, and prostatectomy is a common treatment. Approximately 90,000 US men undergo radical prostatectomy each year,2 and most are left with some degree of incontinence.
Years later, bladder control problems remain
Surgical advances have attempted to minimize nerve and bladder neck damage, but some degree of incontinence is still common after radical prostatectomy. According to the 2000 Prostate Cancer Outcomes Study, 80% of men experienced some incontinence 6 months after radical prostatectomy. After 2 years, 68% of men still had some degree of incontinence,3 and 8% reported frequent or total incontinence. Five years after prostatectomy, only 35% of patients reported complete bladder control vs 87% presurgery.4
A 2004 systematic review showed that behavioral therapy (mostly biofeedback) reduced symptoms in patients with all types of urinary incontinence.5 Many studies, including a 2010 Cochrane review,6 have shown that women with incontinence benefit from pelvic floor muscle training. No randomized trials have assessed the benefit of behavioral therapy for men with incontinence related to postprostatectomy—until now.
STUDY SUMMARY: Behavioral therapy has long-term payoff
The study by Goode et al was an RCT of behavioral therapy for men who had urinary incontinence after radical prostatectomy and whose symptoms persisted more than a year later. It included patients with ≥3 episodes of incontinence per week. Men were excluded if they were undergoing other treatment for prostate cancer, had a high postvoid residual volume or a history of certain bladder surgeries, or were unable to reliably report symptoms. Participants were permitted to continue taking medication for incontinence, with the exception of anticholinergics.
Participants (n=208 from 3 sites) were randomized to one of 3 arms in a blinded fashion with concealed allocation: behavioral therapy alone, behavioral therapy and biofeedback with electrical stimulation, or a control group of men who could elect to try these therapies at a later date.
Behavioral therapy consisted of 4 visits with a physician or nurse practitioner over an 8-week period. At the first visit, patients received instruction in pelvic floor muscle training. Patients then practiced contraction and relaxation exercises and urinary flow control at home. At the second visit, patients learned techniques to avoid episodes of incontinence, such as performing pelvic muscle contractions with stress symptom triggers. During the final 2 visits, patients received advice about control of persistent problems identified in symptom diaries they were required to keep. In addition, continued daily pelvic floor exercises were recommended at the last session.
Men in the group that received biofeedback with electrical stimulation had the same visit schedule, but received additional pelvic floor training—with electrode-mediated feedback and electrical stimulation of pelvic floor muscles during each visit and daily at home. Patients in the control group had the same visit frequency but received no treatment. After 8 weeks, however, the controls were given the opportunity to try behavioral therapy.
Baseline characteristics and attrition rates were similar in all 3 groups. Outcomes were based on an intention-to-treat analysis. At 8 weeks, men receiving behavioral therapy, with or without electrical stimulation and biofeedback, experienced a 55% decrease in incontinence (from 28 episodes per week at baseline to 13 per week); patients in the control group had a 24% decrease (from 25 episodes to 20 per week) (P=.001). More patients in the behavioral groups were completely continent at 8 weeks (16% vs 6% for the controls); the number needed to treat to achieve complete continence was 10. Electrical stimulation and biofeedback provided no added benefit compared with behavioral therapy alone.
Patients in the 2 treatment groups also had clinically significant benefits in some quality-of-life measures (impact of urinary symptoms on travel, emotion, and voiding) and in symptom-specific quality-of-life scores. Patient satisfaction at 8 weeks was higher in the treatment groups: 26 of the 58 men who received behavioral therapy were “delighted, pleased, or mostly satisfied,” vs 9 of 60 in the control group (P=.006 for overall group difference).
Adherence to the behavioral therapy protocol was 100% at 8 weeks and remained high (91%) one year later. Improvement in symptoms continued at one year, with patients in both treatment groups reporting a clinically significant (50%) reduction in incontinence episodes compared with baseline.
WHAT’S NEW: We have evidence-based help for postprostatectomy incontinence
We now have evidence that an 8-week program of pelvic floor training and bladder control strategies reduces the frequency of incontinence in men who have undergone radical prostatectomy.
CAVEATS: The effects of time weren’t factored in
Patients were obviously aware of group assignment, so there is the possibility of treatment bias contributing to the positive self-reported outcomes. While the treatment groups showed both a greater initial improvement and persistent improvement in their symptoms at one year, symptoms of patients in the control group were not measured after a year, so the sustained improvement could reflect resolution of incontinence with time.
CHALLENGES TO IMPLEMENTATION: Locating clinicians who can train patients
The type of behavioral therapy featured in this study may not be easily accessible to all patients. The researchers suggest consulting the National Association for Continence (http://www.nafc.org), a private nonprofit organization whose members include physical therapists, nurses, and physicians. They also cite the Wound Ostomy and Continence Nurses Society (http://www.wocn.org) as a resource in locating nurses who provide these services.
Acknowledgement
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Click here to view PURL METHODOLOGY
Recommend behavioral therapy—incorporating pelvic floor muscle training and bladder control strategies—to men experiencing incontinence after prostatectomy.1
STRENGTH OF RECOMMENDATION
B: Based on a single unblinded randomized controlled trial (RCT).
Goode PS, Burgio KL, Johnson TM, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA. 2011;305:151-159.1
ILLUSTRATIVE CASE
Mr. H, age 75, underwent radical prostatectomy for prostate cancer a year ago. Since then, he leaks urine when he coughs and occasionally has complete loss of bladder control. His lack of control has forced him to curtail many of his normal activities and he wants to know what help you can provide.
Routine prostate-specific antigen (PSA) screening has led to an increase in the diagnosis of localized prostate cancer, and prostatectomy is a common treatment. Approximately 90,000 US men undergo radical prostatectomy each year,2 and most are left with some degree of incontinence.
Years later, bladder control problems remain
Surgical advances have attempted to minimize nerve and bladder neck damage, but some degree of incontinence is still common after radical prostatectomy. According to the 2000 Prostate Cancer Outcomes Study, 80% of men experienced some incontinence 6 months after radical prostatectomy. After 2 years, 68% of men still had some degree of incontinence,3 and 8% reported frequent or total incontinence. Five years after prostatectomy, only 35% of patients reported complete bladder control vs 87% presurgery.4
A 2004 systematic review showed that behavioral therapy (mostly biofeedback) reduced symptoms in patients with all types of urinary incontinence.5 Many studies, including a 2010 Cochrane review,6 have shown that women with incontinence benefit from pelvic floor muscle training. No randomized trials have assessed the benefit of behavioral therapy for men with incontinence related to postprostatectomy—until now.
STUDY SUMMARY: Behavioral therapy has long-term payoff
The study by Goode et al was an RCT of behavioral therapy for men who had urinary incontinence after radical prostatectomy and whose symptoms persisted more than a year later. It included patients with ≥3 episodes of incontinence per week. Men were excluded if they were undergoing other treatment for prostate cancer, had a high postvoid residual volume or a history of certain bladder surgeries, or were unable to reliably report symptoms. Participants were permitted to continue taking medication for incontinence, with the exception of anticholinergics.
Participants (n=208 from 3 sites) were randomized to one of 3 arms in a blinded fashion with concealed allocation: behavioral therapy alone, behavioral therapy and biofeedback with electrical stimulation, or a control group of men who could elect to try these therapies at a later date.
Behavioral therapy consisted of 4 visits with a physician or nurse practitioner over an 8-week period. At the first visit, patients received instruction in pelvic floor muscle training. Patients then practiced contraction and relaxation exercises and urinary flow control at home. At the second visit, patients learned techniques to avoid episodes of incontinence, such as performing pelvic muscle contractions with stress symptom triggers. During the final 2 visits, patients received advice about control of persistent problems identified in symptom diaries they were required to keep. In addition, continued daily pelvic floor exercises were recommended at the last session.
Men in the group that received biofeedback with electrical stimulation had the same visit schedule, but received additional pelvic floor training—with electrode-mediated feedback and electrical stimulation of pelvic floor muscles during each visit and daily at home. Patients in the control group had the same visit frequency but received no treatment. After 8 weeks, however, the controls were given the opportunity to try behavioral therapy.
Baseline characteristics and attrition rates were similar in all 3 groups. Outcomes were based on an intention-to-treat analysis. At 8 weeks, men receiving behavioral therapy, with or without electrical stimulation and biofeedback, experienced a 55% decrease in incontinence (from 28 episodes per week at baseline to 13 per week); patients in the control group had a 24% decrease (from 25 episodes to 20 per week) (P=.001). More patients in the behavioral groups were completely continent at 8 weeks (16% vs 6% for the controls); the number needed to treat to achieve complete continence was 10. Electrical stimulation and biofeedback provided no added benefit compared with behavioral therapy alone.
Patients in the 2 treatment groups also had clinically significant benefits in some quality-of-life measures (impact of urinary symptoms on travel, emotion, and voiding) and in symptom-specific quality-of-life scores. Patient satisfaction at 8 weeks was higher in the treatment groups: 26 of the 58 men who received behavioral therapy were “delighted, pleased, or mostly satisfied,” vs 9 of 60 in the control group (P=.006 for overall group difference).
Adherence to the behavioral therapy protocol was 100% at 8 weeks and remained high (91%) one year later. Improvement in symptoms continued at one year, with patients in both treatment groups reporting a clinically significant (50%) reduction in incontinence episodes compared with baseline.
WHAT’S NEW: We have evidence-based help for postprostatectomy incontinence
We now have evidence that an 8-week program of pelvic floor training and bladder control strategies reduces the frequency of incontinence in men who have undergone radical prostatectomy.
CAVEATS: The effects of time weren’t factored in
Patients were obviously aware of group assignment, so there is the possibility of treatment bias contributing to the positive self-reported outcomes. While the treatment groups showed both a greater initial improvement and persistent improvement in their symptoms at one year, symptoms of patients in the control group were not measured after a year, so the sustained improvement could reflect resolution of incontinence with time.
CHALLENGES TO IMPLEMENTATION: Locating clinicians who can train patients
The type of behavioral therapy featured in this study may not be easily accessible to all patients. The researchers suggest consulting the National Association for Continence (http://www.nafc.org), a private nonprofit organization whose members include physical therapists, nurses, and physicians. They also cite the Wound Ostomy and Continence Nurses Society (http://www.wocn.org) as a resource in locating nurses who provide these services.
Acknowledgement
The PURLs Surveillance System is supported in part by Grant Number UL1RR024999 from the National Center for Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Click here to view PURL METHODOLOGY
1. Goode PS, Burgio KL, Johnson TM, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA. 2011;305:151-159.
2. Barbash GI, Glied SA. New technology and health care costs—the case of robot-assisted surgery. N Engl J Med. 2010;363:701-704.
3. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA. 2000;283:354-360.
4. Penson DF, McLerran D, Feng Z, et al. 5-year urinary and sexual outcomes after radical prostatectomy: results from the prostate cancer outcomes study. J Urol. 2005;173:1701-1705.
5. Teunisse TA, de Jonge A, van Weel C, et al. Treating urinary incontinence in the elderly—conservative therapies that work: a systematic review. J Fam Pract. 2004;53:25-30, 32.
6. Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2010;(1):CD005654.-
1. Goode PS, Burgio KL, Johnson TM, et al. Behavioral therapy with or without biofeedback and pelvic floor electrical stimulation for persistent postprostatectomy incontinence: a randomized controlled trial. JAMA. 2011;305:151-159.
2. Barbash GI, Glied SA. New technology and health care costs—the case of robot-assisted surgery. N Engl J Med. 2010;363:701-704.
3. Stanford JL, Feng Z, Hamilton AS, et al. Urinary and sexual function after radical prostatectomy for clinically localized prostate cancer: the Prostate Cancer Outcomes Study. JAMA. 2000;283:354-360.
4. Penson DF, McLerran D, Feng Z, et al. 5-year urinary and sexual outcomes after radical prostatectomy: results from the prostate cancer outcomes study. J Urol. 2005;173:1701-1705.
5. Teunisse TA, de Jonge A, van Weel C, et al. Treating urinary incontinence in the elderly—conservative therapies that work: a systematic review. J Fam Pract. 2004;53:25-30, 32.
6. Dumoulin C, Hay-Smith J. Pelvic floor muscle training versus no treatment, or inactive control treatments, for urinary incontinence in women. Cochrane Database Syst Rev. 2010;(1):CD005654.-
Copyright © 2011 The Family Physicians Inquiries Network.
All rights reserved.
Acute abdominal pain in an elderly patient
Nausea, Vomiting, and Weakness for 4 days prompted a 76-year-old woman to seek care at our hospital. She was admitted for possible large bowel obstruction and severe dehydration. Her medical history was significant for a metastatic lung cancer to the mediastinal lymph nodes and to the left hip (for which she underwent a hip replacement 4 months earlier), anemia, and diverticulosis.
On Day 1 of her hospital stay, the patient became hypotensive and developed labored breathing. She also had mottled skin and cool fingertips with poor capillary refill. Her abdomen was distended, firm, and diffusely tympanic with diffuse pain to deep palpation and absent bowel sounds.
Her laboratory values revealed leukocytosis (with a significant left shift), metabolic acidosis, and an elevated lactic acid level. Her upright chest x-ray (FIGURE) is shown. The patient was transferred to the intensive care unit for further management.
FIGURE
Upright chest x-ray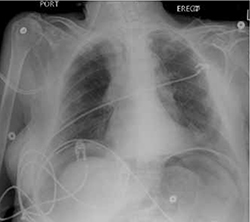
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pneumoperitoneum
This patient had free air under her diaphragm (due to a viscus perforation) and concomitant septic shock. Free air in the peritoneal cavity—pneumoperitoneum— indicates visceral perforation in 85% to 95% of cases.1,2 A ruptured intra-abdominal viscus is considered a surgical emergency. Pneumoperitoneum is often linked to peptic ulcer disease and is seen in 50% of cases of bowel perforations.3 This condition has a higher prevalence in the elderly and carries a higher mortality rate (up to 30% compared with 19% in a younger population).4
A picture that shifts according to the patient’s age
Physical findings suggestive of visceral perforation include sharp abdominal pain with a rigid abdominal wall. Patients will usually lie still because of the peritoneal irritation. Tachycardia and tachypnea are seen early in the disease process, while hypotension and fever usually develop within 4 to 6 hours.5
Elderly patients, however, can present with milder or nonspecific symptoms. Rather than pain, they may complain of the urge to defecate. Physical exam findings such as tachycardia or fever can also be absent due to autonomic dysregulation or medication. Furthermore, laboratory analysis is commonly within normal limits, making the diagnosis even more challenging in this population.5,6
Imaging confirms the Dx
The standard imaging test used to confirm pneumoperitoneum is a standing chest x-ray that will detect free air in almost 80% of cases.7 The sensitivity is influenced by the location of the perforation: Free air will be seen in 69% of gastroduodenal perforations, 30% to 41% of distal small bowel perforations, and 37% to 46% of large bowel perforations.1 Abdominal computed tomography scans have been reported to be more sensitive (up to 100%), especially in identifying small pneumoperitoneum.8,9
Surgery is the next step
Management of pneumoperitoneum includes a prompt surgical consult for a possible emergent laparotomy, nasogastric suctioning, supportive measures for blood pressure, and broad-spectrum antibiotics such as a fourth-generation penicillin or a third-generation cephalosporin plus metronidazole.10
The end of the fight
Given the high mortality rate and the atypical presentation of perforated viscus in the elderly, it is important to maintain a high index of suspicion in this population and to intervene rapidly to improve the outcome.
In the case of our patient, the family followed her wishes and declined surgery. She was aggressively managed with broad-spectrum antibiotics, IV fluids, and vasopressors—but unfortunately died 2 days later.
CORRESPONDENCE Balaji Yegneswaran, MD, University of Pittsburgh Medical Center, 651, Scaife Hall, Pittsburgh, PA 15261; [email protected]
1. Winek TG, Mosely HS, Grout G. Pneumoperitoneum and its association with ruptured abdominal viscus. Arch Surg. 1988;123:709-712.
2. Roh JJ, Thompson S, Harned RK, et al. Value of pneumoperitoneum in the diagnosis of visceral perforation. Am J Surg. 1983;146:830-833.
3. Borum ML. Peptic-ulcer disease in the elderly. Clin Geriatr Med. 1999;15:457-471.
4. Blomgren LG. Perforated peptic ulcer: long-term results after simple closure in the elderly. World J Surg. 1997;21:412-415.
5. Hendrickson M, Naparst TR. Abdominal surgical emergencies in the elderly. Emerg Med Clin N Am. 2003;21:937-969.
6. Kane E, Fried G, McSherry CK. Perforated peptic ulcer in the elderly. J Am Geriatr Soc. 1981;29:224-227.
7. Chen CH, Yang CC, Yen YH. Role of upright chest radiography and ultrasonography in demonstrating free air of perforated peptic ulcers. Hepatogastroenterology. 2001;48:1082-1084.
8. Stapakis JC, Thickman D. Diagnosis of pneumoperitoneum: abdominal CT vs upright chest film. J Comput Assist Tomogr. 1992;16:713-716.
9. Chen CH, Huang HS, Yang CC. The features of perforated peptic ulcers in conventional computed tomography. Hepatogastroenterology. 2001;48:1393-1396.
10. Gorbach SL. Intraabdominal infections. Clin Infect Dis. 1993;17:961-965.
Nausea, Vomiting, and Weakness for 4 days prompted a 76-year-old woman to seek care at our hospital. She was admitted for possible large bowel obstruction and severe dehydration. Her medical history was significant for a metastatic lung cancer to the mediastinal lymph nodes and to the left hip (for which she underwent a hip replacement 4 months earlier), anemia, and diverticulosis.
On Day 1 of her hospital stay, the patient became hypotensive and developed labored breathing. She also had mottled skin and cool fingertips with poor capillary refill. Her abdomen was distended, firm, and diffusely tympanic with diffuse pain to deep palpation and absent bowel sounds.
Her laboratory values revealed leukocytosis (with a significant left shift), metabolic acidosis, and an elevated lactic acid level. Her upright chest x-ray (FIGURE) is shown. The patient was transferred to the intensive care unit for further management.
FIGURE
Upright chest x-ray
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pneumoperitoneum
This patient had free air under her diaphragm (due to a viscus perforation) and concomitant septic shock. Free air in the peritoneal cavity—pneumoperitoneum— indicates visceral perforation in 85% to 95% of cases.1,2 A ruptured intra-abdominal viscus is considered a surgical emergency. Pneumoperitoneum is often linked to peptic ulcer disease and is seen in 50% of cases of bowel perforations.3 This condition has a higher prevalence in the elderly and carries a higher mortality rate (up to 30% compared with 19% in a younger population).4
A picture that shifts according to the patient’s age
Physical findings suggestive of visceral perforation include sharp abdominal pain with a rigid abdominal wall. Patients will usually lie still because of the peritoneal irritation. Tachycardia and tachypnea are seen early in the disease process, while hypotension and fever usually develop within 4 to 6 hours.5
Elderly patients, however, can present with milder or nonspecific symptoms. Rather than pain, they may complain of the urge to defecate. Physical exam findings such as tachycardia or fever can also be absent due to autonomic dysregulation or medication. Furthermore, laboratory analysis is commonly within normal limits, making the diagnosis even more challenging in this population.5,6
Imaging confirms the Dx
The standard imaging test used to confirm pneumoperitoneum is a standing chest x-ray that will detect free air in almost 80% of cases.7 The sensitivity is influenced by the location of the perforation: Free air will be seen in 69% of gastroduodenal perforations, 30% to 41% of distal small bowel perforations, and 37% to 46% of large bowel perforations.1 Abdominal computed tomography scans have been reported to be more sensitive (up to 100%), especially in identifying small pneumoperitoneum.8,9
Surgery is the next step
Management of pneumoperitoneum includes a prompt surgical consult for a possible emergent laparotomy, nasogastric suctioning, supportive measures for blood pressure, and broad-spectrum antibiotics such as a fourth-generation penicillin or a third-generation cephalosporin plus metronidazole.10
The end of the fight
Given the high mortality rate and the atypical presentation of perforated viscus in the elderly, it is important to maintain a high index of suspicion in this population and to intervene rapidly to improve the outcome.
In the case of our patient, the family followed her wishes and declined surgery. She was aggressively managed with broad-spectrum antibiotics, IV fluids, and vasopressors—but unfortunately died 2 days later.
CORRESPONDENCE Balaji Yegneswaran, MD, University of Pittsburgh Medical Center, 651, Scaife Hall, Pittsburgh, PA 15261; [email protected]
Nausea, Vomiting, and Weakness for 4 days prompted a 76-year-old woman to seek care at our hospital. She was admitted for possible large bowel obstruction and severe dehydration. Her medical history was significant for a metastatic lung cancer to the mediastinal lymph nodes and to the left hip (for which she underwent a hip replacement 4 months earlier), anemia, and diverticulosis.
On Day 1 of her hospital stay, the patient became hypotensive and developed labored breathing. She also had mottled skin and cool fingertips with poor capillary refill. Her abdomen was distended, firm, and diffusely tympanic with diffuse pain to deep palpation and absent bowel sounds.
Her laboratory values revealed leukocytosis (with a significant left shift), metabolic acidosis, and an elevated lactic acid level. Her upright chest x-ray (FIGURE) is shown. The patient was transferred to the intensive care unit for further management.
FIGURE
Upright chest x-ray
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Pneumoperitoneum
This patient had free air under her diaphragm (due to a viscus perforation) and concomitant septic shock. Free air in the peritoneal cavity—pneumoperitoneum— indicates visceral perforation in 85% to 95% of cases.1,2 A ruptured intra-abdominal viscus is considered a surgical emergency. Pneumoperitoneum is often linked to peptic ulcer disease and is seen in 50% of cases of bowel perforations.3 This condition has a higher prevalence in the elderly and carries a higher mortality rate (up to 30% compared with 19% in a younger population).4
A picture that shifts according to the patient’s age
Physical findings suggestive of visceral perforation include sharp abdominal pain with a rigid abdominal wall. Patients will usually lie still because of the peritoneal irritation. Tachycardia and tachypnea are seen early in the disease process, while hypotension and fever usually develop within 4 to 6 hours.5
Elderly patients, however, can present with milder or nonspecific symptoms. Rather than pain, they may complain of the urge to defecate. Physical exam findings such as tachycardia or fever can also be absent due to autonomic dysregulation or medication. Furthermore, laboratory analysis is commonly within normal limits, making the diagnosis even more challenging in this population.5,6
Imaging confirms the Dx
The standard imaging test used to confirm pneumoperitoneum is a standing chest x-ray that will detect free air in almost 80% of cases.7 The sensitivity is influenced by the location of the perforation: Free air will be seen in 69% of gastroduodenal perforations, 30% to 41% of distal small bowel perforations, and 37% to 46% of large bowel perforations.1 Abdominal computed tomography scans have been reported to be more sensitive (up to 100%), especially in identifying small pneumoperitoneum.8,9
Surgery is the next step
Management of pneumoperitoneum includes a prompt surgical consult for a possible emergent laparotomy, nasogastric suctioning, supportive measures for blood pressure, and broad-spectrum antibiotics such as a fourth-generation penicillin or a third-generation cephalosporin plus metronidazole.10
The end of the fight
Given the high mortality rate and the atypical presentation of perforated viscus in the elderly, it is important to maintain a high index of suspicion in this population and to intervene rapidly to improve the outcome.
In the case of our patient, the family followed her wishes and declined surgery. She was aggressively managed with broad-spectrum antibiotics, IV fluids, and vasopressors—but unfortunately died 2 days later.
CORRESPONDENCE Balaji Yegneswaran, MD, University of Pittsburgh Medical Center, 651, Scaife Hall, Pittsburgh, PA 15261; [email protected]
1. Winek TG, Mosely HS, Grout G. Pneumoperitoneum and its association with ruptured abdominal viscus. Arch Surg. 1988;123:709-712.
2. Roh JJ, Thompson S, Harned RK, et al. Value of pneumoperitoneum in the diagnosis of visceral perforation. Am J Surg. 1983;146:830-833.
3. Borum ML. Peptic-ulcer disease in the elderly. Clin Geriatr Med. 1999;15:457-471.
4. Blomgren LG. Perforated peptic ulcer: long-term results after simple closure in the elderly. World J Surg. 1997;21:412-415.
5. Hendrickson M, Naparst TR. Abdominal surgical emergencies in the elderly. Emerg Med Clin N Am. 2003;21:937-969.
6. Kane E, Fried G, McSherry CK. Perforated peptic ulcer in the elderly. J Am Geriatr Soc. 1981;29:224-227.
7. Chen CH, Yang CC, Yen YH. Role of upright chest radiography and ultrasonography in demonstrating free air of perforated peptic ulcers. Hepatogastroenterology. 2001;48:1082-1084.
8. Stapakis JC, Thickman D. Diagnosis of pneumoperitoneum: abdominal CT vs upright chest film. J Comput Assist Tomogr. 1992;16:713-716.
9. Chen CH, Huang HS, Yang CC. The features of perforated peptic ulcers in conventional computed tomography. Hepatogastroenterology. 2001;48:1393-1396.
10. Gorbach SL. Intraabdominal infections. Clin Infect Dis. 1993;17:961-965.
1. Winek TG, Mosely HS, Grout G. Pneumoperitoneum and its association with ruptured abdominal viscus. Arch Surg. 1988;123:709-712.
2. Roh JJ, Thompson S, Harned RK, et al. Value of pneumoperitoneum in the diagnosis of visceral perforation. Am J Surg. 1983;146:830-833.
3. Borum ML. Peptic-ulcer disease in the elderly. Clin Geriatr Med. 1999;15:457-471.
4. Blomgren LG. Perforated peptic ulcer: long-term results after simple closure in the elderly. World J Surg. 1997;21:412-415.
5. Hendrickson M, Naparst TR. Abdominal surgical emergencies in the elderly. Emerg Med Clin N Am. 2003;21:937-969.
6. Kane E, Fried G, McSherry CK. Perforated peptic ulcer in the elderly. J Am Geriatr Soc. 1981;29:224-227.
7. Chen CH, Yang CC, Yen YH. Role of upright chest radiography and ultrasonography in demonstrating free air of perforated peptic ulcers. Hepatogastroenterology. 2001;48:1082-1084.
8. Stapakis JC, Thickman D. Diagnosis of pneumoperitoneum: abdominal CT vs upright chest film. J Comput Assist Tomogr. 1992;16:713-716.
9. Chen CH, Huang HS, Yang CC. The features of perforated peptic ulcers in conventional computed tomography. Hepatogastroenterology. 2001;48:1393-1396.
10. Gorbach SL. Intraabdominal infections. Clin Infect Dis. 1993;17:961-965.
Colon cleansing perils: Where’s the evidence?
Various media outlets have sensationalized your article on colon cleansing, “The dangers of colon cleansing” (J Fam Pract. 2011;60:454-457). The article has been perceived by many as a generic criticism of many forms of colon cleansing, with some inappropriate conclusions about widespread harm.
The article included 2 case reports that were incomplete and unclear; no specific diagnosis was made in either case. Authors Mishori et al tried to ascribe negative outcomes to colon cleansing, but the co-mingling of different treatments is apparent in their writing. In other words, colonic hydrotherapy and laxative agents are 2 quite distinct treatments used to facilitate the passage of stool, with different mechanisms of action and potential outcomes, beneficial or otherwise. The authors, in effect, compared apples and oranges, then reached conclusions about colon cleansing that were potentially misleading, generalized, or even naïve.
The first case report described an alleged negative outcome of colon hydrotherapy (colonic irrigation), which may or may not have been directly attributable to the procedure. Furthermore, the procedure was undertaken in an individual with Crohn’s disease, a clear contraindication. To conclude from this case report that colon hydrotherapy is harmful overall has no scientific basis.
The second case report involved the consumption of some form of herbal laxative formula that is not disclosed by the authors. The gastroenterologist who performed a colonoscopy and biopsy on this patient reached a “diagnosis” of “herbal intoxication,” in the presence of some histological evidence of both acute and chronic inflammation.
In both case reports, the actual underlying diagnosis is not clear. One could construct a differential diagnosis that could explain the complaints of these patients as a consequence of events unrelated to the act of colon cleansing.
While I agree that a clear evidence base to support the widespread practice of colonic irrigation is not available in current scientific literature, the procedure should not be summarily condemned. Many individuals report beneficial outcomes of colon hydrotherapy, even if such data are anecdotal and not archived with consistency.
My principal criticism of this article is that it did not present a complete or balanced perspective on the alleged dangers of the procedures in question. The authors failed to acknowledge that the frequency of reported complications of colon hydrotherapy may be significantly less than those reported with various diagnostic tests, such as barium enema examination, sigmoidoscopy, or colonoscopy.
I encourage your readers not to summarily reject “colon cleansing.” I submit that Mishori et al failed to fulfill the criteria for concluding that the act of colon cleansing is overly dangerous or ineffective when applied in an appropriate or medically indicated manner.
Stephen Holt, MD, DSc
Little Falls, NJ
Various media outlets have sensationalized your article on colon cleansing, “The dangers of colon cleansing” (J Fam Pract. 2011;60:454-457). The article has been perceived by many as a generic criticism of many forms of colon cleansing, with some inappropriate conclusions about widespread harm.
The article included 2 case reports that were incomplete and unclear; no specific diagnosis was made in either case. Authors Mishori et al tried to ascribe negative outcomes to colon cleansing, but the co-mingling of different treatments is apparent in their writing. In other words, colonic hydrotherapy and laxative agents are 2 quite distinct treatments used to facilitate the passage of stool, with different mechanisms of action and potential outcomes, beneficial or otherwise. The authors, in effect, compared apples and oranges, then reached conclusions about colon cleansing that were potentially misleading, generalized, or even naïve.
The first case report described an alleged negative outcome of colon hydrotherapy (colonic irrigation), which may or may not have been directly attributable to the procedure. Furthermore, the procedure was undertaken in an individual with Crohn’s disease, a clear contraindication. To conclude from this case report that colon hydrotherapy is harmful overall has no scientific basis.
The second case report involved the consumption of some form of herbal laxative formula that is not disclosed by the authors. The gastroenterologist who performed a colonoscopy and biopsy on this patient reached a “diagnosis” of “herbal intoxication,” in the presence of some histological evidence of both acute and chronic inflammation.
In both case reports, the actual underlying diagnosis is not clear. One could construct a differential diagnosis that could explain the complaints of these patients as a consequence of events unrelated to the act of colon cleansing.
While I agree that a clear evidence base to support the widespread practice of colonic irrigation is not available in current scientific literature, the procedure should not be summarily condemned. Many individuals report beneficial outcomes of colon hydrotherapy, even if such data are anecdotal and not archived with consistency.
My principal criticism of this article is that it did not present a complete or balanced perspective on the alleged dangers of the procedures in question. The authors failed to acknowledge that the frequency of reported complications of colon hydrotherapy may be significantly less than those reported with various diagnostic tests, such as barium enema examination, sigmoidoscopy, or colonoscopy.
I encourage your readers not to summarily reject “colon cleansing.” I submit that Mishori et al failed to fulfill the criteria for concluding that the act of colon cleansing is overly dangerous or ineffective when applied in an appropriate or medically indicated manner.
Stephen Holt, MD, DSc
Little Falls, NJ
Various media outlets have sensationalized your article on colon cleansing, “The dangers of colon cleansing” (J Fam Pract. 2011;60:454-457). The article has been perceived by many as a generic criticism of many forms of colon cleansing, with some inappropriate conclusions about widespread harm.
The article included 2 case reports that were incomplete and unclear; no specific diagnosis was made in either case. Authors Mishori et al tried to ascribe negative outcomes to colon cleansing, but the co-mingling of different treatments is apparent in their writing. In other words, colonic hydrotherapy and laxative agents are 2 quite distinct treatments used to facilitate the passage of stool, with different mechanisms of action and potential outcomes, beneficial or otherwise. The authors, in effect, compared apples and oranges, then reached conclusions about colon cleansing that were potentially misleading, generalized, or even naïve.
The first case report described an alleged negative outcome of colon hydrotherapy (colonic irrigation), which may or may not have been directly attributable to the procedure. Furthermore, the procedure was undertaken in an individual with Crohn’s disease, a clear contraindication. To conclude from this case report that colon hydrotherapy is harmful overall has no scientific basis.
The second case report involved the consumption of some form of herbal laxative formula that is not disclosed by the authors. The gastroenterologist who performed a colonoscopy and biopsy on this patient reached a “diagnosis” of “herbal intoxication,” in the presence of some histological evidence of both acute and chronic inflammation.
In both case reports, the actual underlying diagnosis is not clear. One could construct a differential diagnosis that could explain the complaints of these patients as a consequence of events unrelated to the act of colon cleansing.
While I agree that a clear evidence base to support the widespread practice of colonic irrigation is not available in current scientific literature, the procedure should not be summarily condemned. Many individuals report beneficial outcomes of colon hydrotherapy, even if such data are anecdotal and not archived with consistency.
My principal criticism of this article is that it did not present a complete or balanced perspective on the alleged dangers of the procedures in question. The authors failed to acknowledge that the frequency of reported complications of colon hydrotherapy may be significantly less than those reported with various diagnostic tests, such as barium enema examination, sigmoidoscopy, or colonoscopy.
I encourage your readers not to summarily reject “colon cleansing.” I submit that Mishori et al failed to fulfill the criteria for concluding that the act of colon cleansing is overly dangerous or ineffective when applied in an appropriate or medically indicated manner.
Stephen Holt, MD, DSc
Little Falls, NJ
How can we minimize recurrent ankle sprains?
USING EXTERNAL ANKLE SUPPORTS during physical activity significantly reduces the likelihood of primary and secondary sprains (strength of recommendation [SOR]: A, systematic review).
Proprioception rehabilitation substantially decreases further injury after an ankle sprain (SOR: A, 3 randomized control trials [RCTs] and 1 prospective cohort study).
Evidence summary
A Cochrane review of 14 randomized and quasi-randomized trials concluded that patients who used external ankle supports, such as semi-rigid orthotics or air cast braces, suffered significantly fewer ankle sprains than controls (relative risk [RR]=0.53; 95% confidence interval [CI], 0.40-0.69; number needed to treat [NNT]=22).1 Participants in the trials ranged in age from adolescence to middle age and were either at risk of injury or had suffered a previous ligament injury.
The benefits of ankle supports were most apparent in patients with previous injuries but still evident in patients who hadn’t been injured. External ankle support is recommended for sports with a high risk of ankle injury, such as soccer and basketball, but the decision to use it should be based on perceived risk of injury as opposed to perceived loss of performance.1
Research is insufficient to support wearing high-top shoes to prevent primary and secondary ankle sprains.
Also helpful: Balance and proprioceptive training
A systematic review of 2 RCTs with 703 and 1057 patients concluded that completing a minimum of 6 weeks of balance and coordination training after an acute injury substantially reduced the risk of recurrent ankle sprains for as long as a year (NNT=22; absolute risk reduction=4.5%).2
Proprioceptive training appears to effectively prevent primary and secondary ankle injuries but is more beneficial for patients with a previous ankle injury. A recent RCT that enrolled 522 active sports participants with recent ankle injuries found that those who completed an 8-week, self-guided, proprioceptive training program suffered significantly fewer recurrent sprains at 1 year than the control group (22% vs 33%; relative risk reduction=35%; NNT=9).3
Recommendations
The American Orthopaedic Society for Sports Medicine continues to endorse rest, ice, compression, and elevation for optimal initial care of ankle sprains.4 The American College of Sports Medicine suggests that rehabilitation after an ankle injury should include guided stretching and strengthening of the ankle joint as well as balance training to prevent future injuries.5 Both groups also recommend external ankle supports instead of taping to prevent ankle reinjury.4,5
1. Handoll HH, Rowe BH, Quinn KM, et al. Interventions for preventing ankle ligament injuries. Cochrane Database Syst Rev. 2009;(3):CD000018.-
2. Patrick OM, Hertel J. Systematic review of postural control and lateral ankle instability, part II: is balance training clinically effective? J Athletic Trng. 2008;43:305-315.
3. Hupperets MW, Verhagen EA, VanMechelen W. Effect of unsupervised home based proprioceptive training on recurrences of ankle sprain: randomised control trial. Available at: www.bmj.com/cgi/content/full/339/jul09_1/b2684?maxtoshow=&hits=10&RESULTFORMAT=&fulltext=hupperets&searchid=1&FIRSTINDEX=0&sortspec=date&resourcetype=HWCIT. Accessed July 29, 2010.
4. American Orthopaedic Society for Sports Medicine. Ankle sprains: how to speed your recovery. Available at: http://www.evanekman.com/pdfs/3ST%20Ankle%20Sprains%2008.pdf. Accessed October 10, 2011.
5. American College of Sports Medicine. Current comment: ankle sprains and the athlete. Available at: http://www.acsm.org/docs/current-comments/anklesprainstemp.pdf. Accessed on October 10, 2011.
USING EXTERNAL ANKLE SUPPORTS during physical activity significantly reduces the likelihood of primary and secondary sprains (strength of recommendation [SOR]: A, systematic review).
Proprioception rehabilitation substantially decreases further injury after an ankle sprain (SOR: A, 3 randomized control trials [RCTs] and 1 prospective cohort study).
Evidence summary
A Cochrane review of 14 randomized and quasi-randomized trials concluded that patients who used external ankle supports, such as semi-rigid orthotics or air cast braces, suffered significantly fewer ankle sprains than controls (relative risk [RR]=0.53; 95% confidence interval [CI], 0.40-0.69; number needed to treat [NNT]=22).1 Participants in the trials ranged in age from adolescence to middle age and were either at risk of injury or had suffered a previous ligament injury.
The benefits of ankle supports were most apparent in patients with previous injuries but still evident in patients who hadn’t been injured. External ankle support is recommended for sports with a high risk of ankle injury, such as soccer and basketball, but the decision to use it should be based on perceived risk of injury as opposed to perceived loss of performance.1
Research is insufficient to support wearing high-top shoes to prevent primary and secondary ankle sprains.
Also helpful: Balance and proprioceptive training
A systematic review of 2 RCTs with 703 and 1057 patients concluded that completing a minimum of 6 weeks of balance and coordination training after an acute injury substantially reduced the risk of recurrent ankle sprains for as long as a year (NNT=22; absolute risk reduction=4.5%).2
Proprioceptive training appears to effectively prevent primary and secondary ankle injuries but is more beneficial for patients with a previous ankle injury. A recent RCT that enrolled 522 active sports participants with recent ankle injuries found that those who completed an 8-week, self-guided, proprioceptive training program suffered significantly fewer recurrent sprains at 1 year than the control group (22% vs 33%; relative risk reduction=35%; NNT=9).3
Recommendations
The American Orthopaedic Society for Sports Medicine continues to endorse rest, ice, compression, and elevation for optimal initial care of ankle sprains.4 The American College of Sports Medicine suggests that rehabilitation after an ankle injury should include guided stretching and strengthening of the ankle joint as well as balance training to prevent future injuries.5 Both groups also recommend external ankle supports instead of taping to prevent ankle reinjury.4,5
USING EXTERNAL ANKLE SUPPORTS during physical activity significantly reduces the likelihood of primary and secondary sprains (strength of recommendation [SOR]: A, systematic review).
Proprioception rehabilitation substantially decreases further injury after an ankle sprain (SOR: A, 3 randomized control trials [RCTs] and 1 prospective cohort study).
Evidence summary
A Cochrane review of 14 randomized and quasi-randomized trials concluded that patients who used external ankle supports, such as semi-rigid orthotics or air cast braces, suffered significantly fewer ankle sprains than controls (relative risk [RR]=0.53; 95% confidence interval [CI], 0.40-0.69; number needed to treat [NNT]=22).1 Participants in the trials ranged in age from adolescence to middle age and were either at risk of injury or had suffered a previous ligament injury.
The benefits of ankle supports were most apparent in patients with previous injuries but still evident in patients who hadn’t been injured. External ankle support is recommended for sports with a high risk of ankle injury, such as soccer and basketball, but the decision to use it should be based on perceived risk of injury as opposed to perceived loss of performance.1
Research is insufficient to support wearing high-top shoes to prevent primary and secondary ankle sprains.
Also helpful: Balance and proprioceptive training
A systematic review of 2 RCTs with 703 and 1057 patients concluded that completing a minimum of 6 weeks of balance and coordination training after an acute injury substantially reduced the risk of recurrent ankle sprains for as long as a year (NNT=22; absolute risk reduction=4.5%).2
Proprioceptive training appears to effectively prevent primary and secondary ankle injuries but is more beneficial for patients with a previous ankle injury. A recent RCT that enrolled 522 active sports participants with recent ankle injuries found that those who completed an 8-week, self-guided, proprioceptive training program suffered significantly fewer recurrent sprains at 1 year than the control group (22% vs 33%; relative risk reduction=35%; NNT=9).3
Recommendations
The American Orthopaedic Society for Sports Medicine continues to endorse rest, ice, compression, and elevation for optimal initial care of ankle sprains.4 The American College of Sports Medicine suggests that rehabilitation after an ankle injury should include guided stretching and strengthening of the ankle joint as well as balance training to prevent future injuries.5 Both groups also recommend external ankle supports instead of taping to prevent ankle reinjury.4,5
1. Handoll HH, Rowe BH, Quinn KM, et al. Interventions for preventing ankle ligament injuries. Cochrane Database Syst Rev. 2009;(3):CD000018.-
2. Patrick OM, Hertel J. Systematic review of postural control and lateral ankle instability, part II: is balance training clinically effective? J Athletic Trng. 2008;43:305-315.
3. Hupperets MW, Verhagen EA, VanMechelen W. Effect of unsupervised home based proprioceptive training on recurrences of ankle sprain: randomised control trial. Available at: www.bmj.com/cgi/content/full/339/jul09_1/b2684?maxtoshow=&hits=10&RESULTFORMAT=&fulltext=hupperets&searchid=1&FIRSTINDEX=0&sortspec=date&resourcetype=HWCIT. Accessed July 29, 2010.
4. American Orthopaedic Society for Sports Medicine. Ankle sprains: how to speed your recovery. Available at: http://www.evanekman.com/pdfs/3ST%20Ankle%20Sprains%2008.pdf. Accessed October 10, 2011.
5. American College of Sports Medicine. Current comment: ankle sprains and the athlete. Available at: http://www.acsm.org/docs/current-comments/anklesprainstemp.pdf. Accessed on October 10, 2011.
1. Handoll HH, Rowe BH, Quinn KM, et al. Interventions for preventing ankle ligament injuries. Cochrane Database Syst Rev. 2009;(3):CD000018.-
2. Patrick OM, Hertel J. Systematic review of postural control and lateral ankle instability, part II: is balance training clinically effective? J Athletic Trng. 2008;43:305-315.
3. Hupperets MW, Verhagen EA, VanMechelen W. Effect of unsupervised home based proprioceptive training on recurrences of ankle sprain: randomised control trial. Available at: www.bmj.com/cgi/content/full/339/jul09_1/b2684?maxtoshow=&hits=10&RESULTFORMAT=&fulltext=hupperets&searchid=1&FIRSTINDEX=0&sortspec=date&resourcetype=HWCIT. Accessed July 29, 2010.
4. American Orthopaedic Society for Sports Medicine. Ankle sprains: how to speed your recovery. Available at: http://www.evanekman.com/pdfs/3ST%20Ankle%20Sprains%2008.pdf. Accessed October 10, 2011.
5. American College of Sports Medicine. Current comment: ankle sprains and the athlete. Available at: http://www.acsm.org/docs/current-comments/anklesprainstemp.pdf. Accessed on October 10, 2011.
Evidence-based answers from the Family Physicians Inquiries Network
Diabetes and alcohol use: Detecting at-risk drinking
• Ask a question such as “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” to ascertain a patient’s quantity of alcohol use. A
• Apply elements of the FRAMES approach to help patients curtail at-risk drinking—eg, use elevated HbA1c levels as evidence of a need to change behavior. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
There are enough challenges in controlling diabetes mellitus without the hindrance of undetected problematic alcohol use. The good news is that asking a single nonthreatening question can help you detect at-risk drinking—defined by the National Institute on Alcohol Abuse and Alcoholism (NIAAA) as 5 or more drinks on one occasion or more than 14 drinks per week for men; and 4 or more drinks on one occasion or more than 7 drinks per week for women.1,2 And, for patients who may be compromising their diabetes care and overall health through problem drinking, brief intervention techniques used in the office can enable them to reduce alcohol consumption significantly.
When alcohol becomes a problem in diabetes care
Several studies have explored the long-term benefits of moderate alcohol use on glycemic control—with mixed results. A 2007 study found that diabetes patients who drink 1 glass of wine per day exhibited a lower fasting glucose level than abstainers after 3 months.3 There was no difference, however, on postprandial glucose levels. A 2008 study found that individuals who drank one to 2 glasses of wine per day for a month had lower fasting serum insulin levels relative to when they have abstained for a month,4 although levels of fasting plasma cholesterol, HDL cholesterol, glucose, and hemoglobin A1c (HbA1c) remained unchanged relative to periods of abstinence.4
Furthermore, rates of coronary heart disease and CHD mortality in a meta-analysis were significantly lower in 3 categories of alcohol consumption (<6 g/d, 6 to <18 g/d, and ≥18 g/d) compared with abstinence.5 Nondrinkers also had a greater risk of total mortality compared with the lightest drinking group. Notably, however, the lower limit of the highest drinking category was only 1.5 drinks per day.
How big is the problem? In a study of insulin-treated patients seen for severe hypoglycemia, 17% had been drinking before the episode.6 In a primary care sample, 28% of randomly selected patients with type 2 diabetes met Diagnostic and Statistical Manual of Mental Disorders-IV criteria for a lifetime incidence of alcohol abuse and 13% met either current or lifetime criteria for alcohol dependence.7 Another study of primary care patients with diabetes8 found that 13.4% met NIAAA criteria for at-risk drinking; 11.1% of these at-risk drinkers met criteria for current alcohol dependence. (According to the NIAAA, the rate of at-risk/heavy drinking among US adults is 30%, and about one in 4 heavy drinkers meets the criteria for alcohol abuse or dependence.1)
Detrimental effects with immoderate drinking. Individuals who engage in at-risk drinking, as defined by the NIAAA, are at increased risk for alcohol dependence9 and associated complications such as diabetic neuropathy and retinopathy,10 atherosclerosis,11 and total and CHD mortality.3,12 Heavy drinking also interferes with neuroendocrine, gastrointestinal, and sexual function,13 and its interaction with diabetes increases the risk for hepatocellular carcinoma after controlling for hepatitis B and C serology.14
Interference with diabetes control. Research examining the short-term effect of alcohol use has produced contradictory results, partly due to differences among studies, such as whether alcohol is administered with a meal and whether a fasting glucose level is measured.15 However, alcohol affects glycemic control and, when used excessively, can impair glucose production.16,17 Alcohol may induce hypoglycemia,10,18 and even small amounts may jeopardize diabetes control.13 In a study of patients with insulin-treated diabetes, alcohol use in the presence of mild hypoglycemia increased diastolic blood pressure or exacerbated hypoglycemia-related cognitive deficits.19 Another concern—in both the short and long term—is that alcohol interacts negatively with certain diabetes medications. It is more likely to induce hypoglycemia in the presence of sulfonylurea medications.10 Chlorpropamide decreases the rate of ethanol elimination from the blood.20 And, in those taking metformin, excessive alcohol use elevates risk for lactic acidosis. 21
Diminished self-care. Alcohol use can interfere with self-care,22,23 which is a crucial component of diabetes treatment.24 It may lead to reduced eating16 or to decreased willingness to adhere to prescribed dietary regimens.13 It also impairs other self-care behaviors13,15,25 such as self-monitoring blood glucose and showing up for medical appointments.26 In a large, diverse sample of patients with diabetes,24 heavy drinkers had the highest rates of morbidity. Importantly, alcohol and diabetes self-care behavior were significantly negatively associated. Studies of ethnic minority samples have yielded comparable results.27
Assessing alcohol use: Obstacles and solutions
Although alcohol use can be readily evaluated during routine primary care appointments, it is often neglected, perhaps due to a lack of awareness about its impact on diabetes.15 Those who are most often assessed tend to have a psychiatric diagnosis or other condition raising a red flag for physicians.28 When internists, general practitioners, and psychiatrists were questioned in a study regarding patients’ alcohol and drug use, all 3 groups were misinformed about which substance-use treatments were empirically supported29 and did not believe that treatment for alcohol abuse held much promise. Another study showed that physicians can be reluctant to screen for alcohol use because of the difficulty in recognizing a problem, the perceived unimportance of alcohol use as a health risk, a supposed lack of adequate intervention tools, and a fear of stigmatizing patients.30 Physicians are more likely to discuss alcohol use under certain extreme conditions such as when a patient smells of alcohol.
Multiple opportunities to ask in the VA system. In the Veterans Health Administration, primary care VA providers have reported that prompts for alcohol screening embedded in computerized progress notes, clinical reminder lists, and annual health evaluation forms encourage them to assess alcohol use. Other useful materials include manual checklists and reference cards.31 These providers also report that education, feedback on rates of alcohol screening, and increased supervision facilitate assessment. Finally, providers indicate that asking nurses or clerical staff to administer the screen improves completion rates.
Ask a simple question
“How often have you had a drink containing alcohol in the last year?” or “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” are questions that can help you compare a patient’s alcohol use to the at-risk drinking cutoffs established by the NIAAA.1,2
Recent research has also validated the use of a single question in identifying NIAAA-defined at-risk drinking.32 Simply ask patients, “How many times in the past year have you had X or more drinks in a day?” (X=5 for men or 4 for women). The screen is positive when a patient acknowledges having done so at least once in the past year. This question was 81.8% sensitive and 79.3% specific for unhealthy alcohol use, and 87.9% sensitive and 66.8% specific for current alcohol abuse or dependence.32 Advantages of this method are its brevity, ease of scoring, validity in the primary care setting,32 and ease of recollection for treatment providers (TABLE 1).1,2
TABLE 1
Ask these simple questions to assess alcohol use1,2
| To assess… | Ask… |
|---|---|
| Frequency of alcohol use | “How often do you drink alcohol (including beer or wine)?” or “How often have you had a drink containing alcohol in the last year?” |
| Quantity of alcohol use | “When you do drink alcohol, how many standard* drinks do you have?” or “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” |
| Binge drinking | For men: “How often do you have 5 or more standard* drinks on one occasion?” For women: “How often do you have 4 or more standard* drinks on one occasion?” |
| Alcohol use with the NIAAA single-question screen | “How many times in the past year have you had X or more drinks in a day?” (X = 5 for men and X = 4 for women; positive response ≥1) |
| *A standard drink is equal to 12 oz. of regular beer, 8 to 9 oz. of malt liquor, 5 oz. of wine, or 1.5 oz. of 80-proof spirits.1 NIAAA, National Institute on Alcohol Abuse and Alcoholism. | |
Brief intervention works in primary care
Brief interventions for drinking have strong empirical support. In a review of treatments for alcohol abuse and dependence,33 brief intervention was one of only 2 “efficacious” treatments.
Although some individual studies of brief alcohol interventions in primary care have not shown favorable results, several systematic reviews have demonstrated the efficacy of such interventions in this setting. General practitioner–delivered brief interventions led to significantly better patient outcomes compared with standard care, and “very brief advice” resulted in reductions in alcohol consumption overall and in the percentage of “excessive drinkers.”34 In a review of health behavior interventions,35 brief interventions reduced risky or harmful drinking. In one of 2 meta-analyses that support this finding, brief interventions with primary care patients not seeking treatment for alcohol abuse yielded small-to-medium effect sizes relative to control conditions.36 In the other study, brief interventions significantly reduced longer term alcohol use in primary care patients.37
The US Preventive Services Task Force conducted a systematic review of behavioral counseling interventions and recommends screening and brief interventions for unhealthy drinking in primary care.38,39 Its findings indicate that alcohol use declines significantly after brief interventions containing at least 2 of the following elements: feedback regarding drinking, advice to reduce drinking, or goal setting.
Brief advice is a form of intervention that shows considerable promise in primary care.40-42 Two 10- to 15-minute sessions have led to significant reductions in the mean number of drinks and frequency of excessive drinking in the 7 days before a follow-up interview, as well as a reduction in binge drinking episodes in the previous 30 days.41,42
One study produced positive results with just a 5- to 10-minute counseling session involving advice for drinking goals delivered by primary care providers as part of a routine medical visit.43 This intervention led to significant decreases in alcohol use at a 6-month follow-up for high-risk drinkers compared with controls.43 Brief interventions additionally work within the time constraints of a busy primary care practice and are cost effective.
Some primary care providers think a specialist should conduct interventions and suggest that having a specialist immediately available would enable intervention.31 In fact, some research has supported the idea of special training. In a European study, primary care providers reported that more practical training, information about brief intervention studies, personal training, and lectures would facilitate interventions.44
Applying brief alcohol interventions to diabetes patients
Newer research has tested the efficacy of alcohol interventions with diabetes patients in the primary care setting. In one study,45 brief advice was given in 2 15-minute sessions and 2 5-minute follow-up telephone calls. Compared with controls, significantly more participants who received the intervention reduced heavy drinking from baseline to follow-up. One caveat is that patients with hypertension were included in the sample, making it difficult to determine the impact of the intervention on diabetes patients specifically.
In a small study of patients with diabetes exhibiting at-risk drinking,8 a single-session intervention based on motivational interviewing (MI) principles46 gave participants personalized feedback in relation to sex-based norms of drinking rates and HbA1c and triglyceride laboratory results. Patients were given information on the physiologic effects of alcohol on diabetes, the potential interactions between alcohol and diabetes medications, and the effect of alcohol on diabetes self-care behavior. They were asked to identify pros and cons of their drinking and to develop personal change goals. One of 2 PhD-level clinical psychologists trained in MI administered the single 50-minute intervention. By 1 month and continuing through to the 6-month follow-up, participants had reduced the proportion of drinking days, mean number of daily drinks, and proportion of heavy drinking days.
Ramsey and colleagues47 extended this work by comparatively examining a group of patients exhibiting at-risk drinking who received no intervention. The results favored the intervention group, with a medium-to-large effect size for the proportion of drinking days, a medium effect size for the reduction of mean number of daily drinks, and a small-to-medium effect size in the reduction of heavy drinking days. Furthermore, in the intervention group there was a trend toward better diabetes adherence behavior.
Implementing brief intervention in practice. Despite differences among interventions, the elements of brief interventions tend to be similar.48 Incorporating these elements in the primary care setting provides a useful framework that will likely prove beneficial. Specifically, brief interventions typically contain elements of the FRAMES (TABLE 2)46 acronym:
- Feedback about one’s drinking relative to others
- Responsibility for deciding to change
- Advice to change drinking
- Menu of options for implementing a change strategy
- Empathic listening
- Self-efficacy enhancement.
Decision-making models indicate that expectations about the effects of behavior change play a significant role in determining whether a decision to change is made.49 The perceived costs and benefits of changing drinking50,51 and positive52 and negative alcohol expectancies53,54 predict future alcohol use. For patients with diabetes who are at-risk drinkers, primary care appointments may provide “teachable moments” in which brief advice can have a significant impact—particularly when patients are told laboratory test results; advised about the sugar and carbohydrate content of alcohol; or given information regarding the effect of alcohol on diabetes, medications, and self-care behavior. Finally, primary care providers will also likely have knowledge of a patient’s comorbid conditions (eg, depression) that may relate to diabetes or alcohol use.
TABLE 2
How to implement the FRAMES approach in brief interventions46
| FRAMES elements | Sample statements |
|---|---|
| Feedback about one’s drinking relative to others | “Based on what you told me, you are drinking an average of 21 drinks per week, which puts you above the cutoff for at-risk drinking” or “According to what you told me, it looks like you are drinking more than 88% of men (or women) in the United States.” |
| Responsibility for deciding to change | “You’re in the best position to decide how you would like to make a change” or “Although reducing your drinking would be good for your health, it’s up to you to decide when you’re ready to make a change.” |
| Advice to change drinking | “Your test results indicate your HbA1c, an important measure of blood sugar, is elevated; making a change in your drinking will likely improve your blood sugar” or “I am concerned about the effect on your health of drinking alcohol while taking your diabetes medications. Making a change in your drinking is likely to protect you from complications.” |
| Menu of options for implementing a change strategy | “If it’s okay with you, I can share what has worked for others whose drinking is similar to yours. Some people alternate a drink containing alcohol with water or diet soda. Others will bring only a certain amount of money with them when they go to a bar.” |
| Empathic listening | “It sounds like this has been a concern" or "I know that change can be difficult.” |
| Self-efficacy enhancement | “I wonder if you could use some of the same strategies you used to lose 10 pounds last year?” or “It sounds like you have some ideas for how to make this happen.” |
CORRESPONDENCE Patricia A. Engler, PhD, DGIM, 111 Plain Street Building, Providence, RI 02903; [email protected]
1. National Institute on Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician’s guide. 2005. Available at: http://pubs.niaaa.nih.gov/publications/Practitioner/CliniciansGuide2005/guide.pdf. Accessed November 8, 2011.
2. Bradley KA, Kivlahan DR, Williams E. Brief approaches to alcohol screening: practical alternatives for primary care. J Gen Intern Med 2009;24:881-883.
3. Shai I, Fraser D, Wainstein J, et al. Glycemic effects of moderate alcohol intake among patients with type 2 diabetes. Diabetes Care 2007;30:3011-3016.
4. Bantle AE, Tomas W, Bantle JP. Metabolic effects of alcohol in the form of wine in persons with type 2 diabetes mellitus. Metabolism 2008;57:241-245.
5. Koppes LLJ, Dekker JM, Hendriks HFJ, et al. Meta-analysis of the relationship between alcohol consumption and coronary heart disease and mortality in type 2 diabetic patients. Diabetologia 2006;49:648-652.
6. Pedersen-Bjergaard U, Reubsaet JLE, Nielsen SL, et al. Psychoactive drugs, alcohol, and severe hypoglycemia in insulin-treated diabetes: analysis of 141 cases. Am J Med 2005;118:307-310.
7. Fleming M, Mundt M. Carbohydrate-deficient transferrin: validity of a new alcohol biomarker in a sample of patients with diabetes and hypertension. J Am Board Fam Pract 2004;17:247-255.
8. Engler PA, Ramsey SE, Stein MD. Brief alcohol intervention among diabetic patients: a pilot study. Presented at: Annual Meeting of the Society for Behavioral Medicine; March 26–29, 2008; San Diego, CA.
9. Saha TD, Stinson FS, Grant BF. The role of alcohol consumption in future classifications of alcohol use disorders. Drug Alcohol Depend 2007;89:82-92.
10. Shai I, Rimm EB, Schulze MB, et al. Moderate alcohol intake and markers of inflammation and endothelial dysfunction among diabetic men. Diabetologia 2004;14:1760-1767.
11. Wakabayashi I, Kobaba-Wakabayashi R, Masuda H. Relation of drinking alcohol to atherosclerotic risk in type 2 diabetes. Diabetes Care 2002;25:1223-1228.
12. Diem M, Deplazes M, Fajfr R, et al. Effects of alcohol consumption on mortality in patients with type 2 diabetes mellitus. Diabetologia 2003;46:1581-1585.
13. Cox WM, Blount JP, Crowe PA, et al. Diabetic patients’ alcohol use and quality of life: relationships with prescribed treatment compliance among older males. Alcohol Clin Exp Res 1996;20:327-331.
14. Yuan JM, Govindarajan S, Arakawa K, et al. Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer 2004;101:1009-1017.
15. Howard AA, Arnsten JH, Gourevitch MN. Effect of alcohol consumption on diabetes mellitus. Ann Intern Med 2004;140:211-219.
16. Glasgow AM, Tynan D, Schwartz R, et al. Alcohol and drug use in teenagers with diabetes mellitus. J Adolesc Health 1991;12:11-14.
17. Turner BC, Jenkins E, Kerr D, et al. The effect of evening alcohol consumption on next-morning glucose control in type 1 diabetes. Diabetes Care 2001;24:1888-1893.
18. Richardson T, Weiss M, Thomas P, et al. Day after the night before. Influence of evening alcohol on risk of hypoglycemia in patients with type 1 diabetes. Diabetes Care. 2005;28:1801-1802.
19. Cheyne EH, Sherwin RS, Lunt MJ, et al. Influence of alcohol on cognitive performance during mild hypoglycaemia: implications for type 1 diabetes. Diabet Med 2004;21:230-237.
20. Lao B, Czyzyk A, Szutowski M, et al. Alcohol tolerance in patients with non-insulin-dependent (type 2) diabetes treated with sulphonylurea derivatives. Arzneimittelforschung 1994;44:727-734.
21. PDR Staff Physicians’ Desk Reference 2003. 57th ed. Montvale, NJ: Medical Economics Company; 2003.
22. Ramchandani N, Cantey-Kiser JM, Alter CA, et al. Self-reported factors that affect glycemic control in college students with type 1 diabetes. Diabetes Educ 2000;26:656-666.
23. Kyngas H. Compliance of adolescents with chronic disease. J Clin Nurs 2000;9:549-556.
24. Ahmed AT, Karter AJ, Liu J. Alcohol consumption is inversely associated with adherence to diabetes self-care behaviours. Diabet Med 2006;23:795-802.
25. Karter AJ, Ferrara A, Darbinian JA, et al. Self-monitoring of blood glucose. Diabetes Care 2004;23:477-483.
26. Chew LD, Nelson KM, Young BA, et al. Association between alcohol consumption and diabetes preventative practices. Fam Med 2005;37:589-594.
27. Johnson KH, Bazargan M, Bing E. Alcohol consumption and compliance among inner-city minority patients with type 2 diabetes mellitus. Arch Fam Med 2000;9:964-970.
28. D’Amico EJ, Paddock SM, Burnam A, et al. Identification of and guidance for problem drinking by general medical providers. Med Care 2005;43:229-236.
29. Roche AM, Parle MD, Stubbs JM, et al. Management and treatment efficacy of drug and alcohol problems: what do doctors believe? Addiction. 1995;90:1357-1366.
30. Aira M, Kauhanen J, Larivaara P, et al. Differences in brief interventions on excessive drinking and smoking by primary care physicians: qualitative study. Prev Med 2004;38:473-478.
31. Barry KL, Blow FC, Willenbring M, et al. Use of alcohol screening and brief interventions in primary care settings: implementation and barriers. Subst Abus 2004;25:27-36.
32. Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single-question alcohol screening test. J Gen Intern Med 2009;24:783-788.
33. McCrady BS. Alcohol use disorders and the Division 12 Task Force of the American Psychological Association. Psychol Addict Behav 2000;14:267-276.
34. Richmond RL, Anderson P. Research in general practice for smokers and excessive drinkers in Australia and the UK. I. Interpretation of results. Addiction 1994;89:35-40.
35. Goldstein MG, Whitlock EP, DePue J. Multiple behavioral risk factor interventions in primary care. Am J Prev Med 2004;27:61-79.
36. Moyer A, Finney JW, Swearingen CE, et al. Brief interventions for alcohol problems: a meta-analytic review of controlled investigations in treatment-seeking and non-treatment-seeking populations. Addiction 2002;97:279-292.
37. Bertholet N, Daeppen JB, Wietlisbach V, et al. Reduction of alcohol consumption by brief alcohol intervention in primary care: a systematic review and meta-analysis. Arch Intern Med 2005;165:986-995.
38. Whitlock EP, Polen MR, Green CA, et al. Behavioral counseling interventions in primary care to reduce risky/harmful alcohol use by adults: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2004;140:557-568.
39. U. S. Preventive Services Task Force. Screening for problem drinking. In: DiGuiseppi C, Atkins D, Woolf SH, Kamerow DB, eds. Guide to Clinical Preventive Services. 2nd ed. Alexandria, VA: International Medical Services; 1996;567-582.
40. Anderson P, Scott E. The effect of general practitioners’ advice to heavy drinking men. Br J Addict 1992;87:891-900.
41. Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res 2002;26:36-43.
42. Fleming MF, Barry KL, Manwell LB, et al. Brief physician advice for problem alcohol drinkers. JAMA 1997;277:1039-1045.
43. Ockene JK, Adams A, Hurley TG, et al. Brief physician- and nurse practitioner-delivered counseling for high-risk drinkers. Arch Intern Med 1999;159:2198-2205.
44. Aalto M, Pekuri P, Seppa K. Primary health care nurses’ and physicians’ attitudes, knowledge and beliefs regarding brief intervention for heavy drinkers. Addiction 2001;96:305-311.
45. Fleming M, Brown R, Brown D. The efficacy of a brief alcohol intervention combined with %CDT feedback in patients being treated for type 2 diabetes and/or hypertension. J Stud Alcohol 2004;65:631-637.
46. Miller WR, Rollnick S. Motivational Interviewing: Preparing People for Change. New York: Guilford Press; 2002.
47. Ramsey SE, Engler PA, Harrington M, et al. A brief alcohol intervention with at-risk drinking diabetics. Subst Abus 2010;4:1-8.
48. Bien TH, Miller WR, Tonigan JS. Brief interventions for alcohol problems: a review. Addiction 1993;88:315-335.
49. Sutton S. Social-psychological approaches to understanding addictive behaviours: attitude-behaviour and decision-making models. Br J Addict 1987;82:355-370.
50. Cunningham JA, Sobell LC, Gavin DR, et al. Assessing motivation for change: preliminary development and evaluation of a scale measuring the costs and benefits of changing alcohol or drug use. Psychol Addict Behav 1997;11:107-114.
51. Rollnick S, Morgan M, Heather N. The development of a brief scale to measure outcome expectations of reduced consumption among excessive drinkers. Addict Behav 1996;21:377-387.
52. Brown SA. Reinforcement expectancies and alcoholism treatment outcome after a one-year follow-up. J Stud Alcohol 1985;46:304-308.
53. Jones BT, McMahon J. Negative alcohol expectancy predicts post-treatment abstinence survivorship: the whether, when and why of relapse to a first drink. Addiction 1994;89:1653-1665.
54. Jones BT, McMahon J. Negative and positive alcohol expectancies as predictors of abstinence after discharge from a residential treatment program: a one-month and three-month follow-up study in men. J Stud Alcohol 1994;55:543-548.
• Ask a question such as “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” to ascertain a patient’s quantity of alcohol use. A
• Apply elements of the FRAMES approach to help patients curtail at-risk drinking—eg, use elevated HbA1c levels as evidence of a need to change behavior. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
There are enough challenges in controlling diabetes mellitus without the hindrance of undetected problematic alcohol use. The good news is that asking a single nonthreatening question can help you detect at-risk drinking—defined by the National Institute on Alcohol Abuse and Alcoholism (NIAAA) as 5 or more drinks on one occasion or more than 14 drinks per week for men; and 4 or more drinks on one occasion or more than 7 drinks per week for women.1,2 And, for patients who may be compromising their diabetes care and overall health through problem drinking, brief intervention techniques used in the office can enable them to reduce alcohol consumption significantly.
When alcohol becomes a problem in diabetes care
Several studies have explored the long-term benefits of moderate alcohol use on glycemic control—with mixed results. A 2007 study found that diabetes patients who drink 1 glass of wine per day exhibited a lower fasting glucose level than abstainers after 3 months.3 There was no difference, however, on postprandial glucose levels. A 2008 study found that individuals who drank one to 2 glasses of wine per day for a month had lower fasting serum insulin levels relative to when they have abstained for a month,4 although levels of fasting plasma cholesterol, HDL cholesterol, glucose, and hemoglobin A1c (HbA1c) remained unchanged relative to periods of abstinence.4
Furthermore, rates of coronary heart disease and CHD mortality in a meta-analysis were significantly lower in 3 categories of alcohol consumption (<6 g/d, 6 to <18 g/d, and ≥18 g/d) compared with abstinence.5 Nondrinkers also had a greater risk of total mortality compared with the lightest drinking group. Notably, however, the lower limit of the highest drinking category was only 1.5 drinks per day.
How big is the problem? In a study of insulin-treated patients seen for severe hypoglycemia, 17% had been drinking before the episode.6 In a primary care sample, 28% of randomly selected patients with type 2 diabetes met Diagnostic and Statistical Manual of Mental Disorders-IV criteria for a lifetime incidence of alcohol abuse and 13% met either current or lifetime criteria for alcohol dependence.7 Another study of primary care patients with diabetes8 found that 13.4% met NIAAA criteria for at-risk drinking; 11.1% of these at-risk drinkers met criteria for current alcohol dependence. (According to the NIAAA, the rate of at-risk/heavy drinking among US adults is 30%, and about one in 4 heavy drinkers meets the criteria for alcohol abuse or dependence.1)
Detrimental effects with immoderate drinking. Individuals who engage in at-risk drinking, as defined by the NIAAA, are at increased risk for alcohol dependence9 and associated complications such as diabetic neuropathy and retinopathy,10 atherosclerosis,11 and total and CHD mortality.3,12 Heavy drinking also interferes with neuroendocrine, gastrointestinal, and sexual function,13 and its interaction with diabetes increases the risk for hepatocellular carcinoma after controlling for hepatitis B and C serology.14
Interference with diabetes control. Research examining the short-term effect of alcohol use has produced contradictory results, partly due to differences among studies, such as whether alcohol is administered with a meal and whether a fasting glucose level is measured.15 However, alcohol affects glycemic control and, when used excessively, can impair glucose production.16,17 Alcohol may induce hypoglycemia,10,18 and even small amounts may jeopardize diabetes control.13 In a study of patients with insulin-treated diabetes, alcohol use in the presence of mild hypoglycemia increased diastolic blood pressure or exacerbated hypoglycemia-related cognitive deficits.19 Another concern—in both the short and long term—is that alcohol interacts negatively with certain diabetes medications. It is more likely to induce hypoglycemia in the presence of sulfonylurea medications.10 Chlorpropamide decreases the rate of ethanol elimination from the blood.20 And, in those taking metformin, excessive alcohol use elevates risk for lactic acidosis. 21
Diminished self-care. Alcohol use can interfere with self-care,22,23 which is a crucial component of diabetes treatment.24 It may lead to reduced eating16 or to decreased willingness to adhere to prescribed dietary regimens.13 It also impairs other self-care behaviors13,15,25 such as self-monitoring blood glucose and showing up for medical appointments.26 In a large, diverse sample of patients with diabetes,24 heavy drinkers had the highest rates of morbidity. Importantly, alcohol and diabetes self-care behavior were significantly negatively associated. Studies of ethnic minority samples have yielded comparable results.27
Assessing alcohol use: Obstacles and solutions
Although alcohol use can be readily evaluated during routine primary care appointments, it is often neglected, perhaps due to a lack of awareness about its impact on diabetes.15 Those who are most often assessed tend to have a psychiatric diagnosis or other condition raising a red flag for physicians.28 When internists, general practitioners, and psychiatrists were questioned in a study regarding patients’ alcohol and drug use, all 3 groups were misinformed about which substance-use treatments were empirically supported29 and did not believe that treatment for alcohol abuse held much promise. Another study showed that physicians can be reluctant to screen for alcohol use because of the difficulty in recognizing a problem, the perceived unimportance of alcohol use as a health risk, a supposed lack of adequate intervention tools, and a fear of stigmatizing patients.30 Physicians are more likely to discuss alcohol use under certain extreme conditions such as when a patient smells of alcohol.
Multiple opportunities to ask in the VA system. In the Veterans Health Administration, primary care VA providers have reported that prompts for alcohol screening embedded in computerized progress notes, clinical reminder lists, and annual health evaluation forms encourage them to assess alcohol use. Other useful materials include manual checklists and reference cards.31 These providers also report that education, feedback on rates of alcohol screening, and increased supervision facilitate assessment. Finally, providers indicate that asking nurses or clerical staff to administer the screen improves completion rates.
Ask a simple question
“How often have you had a drink containing alcohol in the last year?” or “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” are questions that can help you compare a patient’s alcohol use to the at-risk drinking cutoffs established by the NIAAA.1,2
Recent research has also validated the use of a single question in identifying NIAAA-defined at-risk drinking.32 Simply ask patients, “How many times in the past year have you had X or more drinks in a day?” (X=5 for men or 4 for women). The screen is positive when a patient acknowledges having done so at least once in the past year. This question was 81.8% sensitive and 79.3% specific for unhealthy alcohol use, and 87.9% sensitive and 66.8% specific for current alcohol abuse or dependence.32 Advantages of this method are its brevity, ease of scoring, validity in the primary care setting,32 and ease of recollection for treatment providers (TABLE 1).1,2
TABLE 1
Ask these simple questions to assess alcohol use1,2
| To assess… | Ask… |
|---|---|
| Frequency of alcohol use | “How often do you drink alcohol (including beer or wine)?” or “How often have you had a drink containing alcohol in the last year?” |
| Quantity of alcohol use | “When you do drink alcohol, how many standard* drinks do you have?” or “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” |
| Binge drinking | For men: “How often do you have 5 or more standard* drinks on one occasion?” For women: “How often do you have 4 or more standard* drinks on one occasion?” |
| Alcohol use with the NIAAA single-question screen | “How many times in the past year have you had X or more drinks in a day?” (X = 5 for men and X = 4 for women; positive response ≥1) |
| *A standard drink is equal to 12 oz. of regular beer, 8 to 9 oz. of malt liquor, 5 oz. of wine, or 1.5 oz. of 80-proof spirits.1 NIAAA, National Institute on Alcohol Abuse and Alcoholism. | |
Brief intervention works in primary care
Brief interventions for drinking have strong empirical support. In a review of treatments for alcohol abuse and dependence,33 brief intervention was one of only 2 “efficacious” treatments.
Although some individual studies of brief alcohol interventions in primary care have not shown favorable results, several systematic reviews have demonstrated the efficacy of such interventions in this setting. General practitioner–delivered brief interventions led to significantly better patient outcomes compared with standard care, and “very brief advice” resulted in reductions in alcohol consumption overall and in the percentage of “excessive drinkers.”34 In a review of health behavior interventions,35 brief interventions reduced risky or harmful drinking. In one of 2 meta-analyses that support this finding, brief interventions with primary care patients not seeking treatment for alcohol abuse yielded small-to-medium effect sizes relative to control conditions.36 In the other study, brief interventions significantly reduced longer term alcohol use in primary care patients.37
The US Preventive Services Task Force conducted a systematic review of behavioral counseling interventions and recommends screening and brief interventions for unhealthy drinking in primary care.38,39 Its findings indicate that alcohol use declines significantly after brief interventions containing at least 2 of the following elements: feedback regarding drinking, advice to reduce drinking, or goal setting.
Brief advice is a form of intervention that shows considerable promise in primary care.40-42 Two 10- to 15-minute sessions have led to significant reductions in the mean number of drinks and frequency of excessive drinking in the 7 days before a follow-up interview, as well as a reduction in binge drinking episodes in the previous 30 days.41,42
One study produced positive results with just a 5- to 10-minute counseling session involving advice for drinking goals delivered by primary care providers as part of a routine medical visit.43 This intervention led to significant decreases in alcohol use at a 6-month follow-up for high-risk drinkers compared with controls.43 Brief interventions additionally work within the time constraints of a busy primary care practice and are cost effective.
Some primary care providers think a specialist should conduct interventions and suggest that having a specialist immediately available would enable intervention.31 In fact, some research has supported the idea of special training. In a European study, primary care providers reported that more practical training, information about brief intervention studies, personal training, and lectures would facilitate interventions.44
Applying brief alcohol interventions to diabetes patients
Newer research has tested the efficacy of alcohol interventions with diabetes patients in the primary care setting. In one study,45 brief advice was given in 2 15-minute sessions and 2 5-minute follow-up telephone calls. Compared with controls, significantly more participants who received the intervention reduced heavy drinking from baseline to follow-up. One caveat is that patients with hypertension were included in the sample, making it difficult to determine the impact of the intervention on diabetes patients specifically.
In a small study of patients with diabetes exhibiting at-risk drinking,8 a single-session intervention based on motivational interviewing (MI) principles46 gave participants personalized feedback in relation to sex-based norms of drinking rates and HbA1c and triglyceride laboratory results. Patients were given information on the physiologic effects of alcohol on diabetes, the potential interactions between alcohol and diabetes medications, and the effect of alcohol on diabetes self-care behavior. They were asked to identify pros and cons of their drinking and to develop personal change goals. One of 2 PhD-level clinical psychologists trained in MI administered the single 50-minute intervention. By 1 month and continuing through to the 6-month follow-up, participants had reduced the proportion of drinking days, mean number of daily drinks, and proportion of heavy drinking days.
Ramsey and colleagues47 extended this work by comparatively examining a group of patients exhibiting at-risk drinking who received no intervention. The results favored the intervention group, with a medium-to-large effect size for the proportion of drinking days, a medium effect size for the reduction of mean number of daily drinks, and a small-to-medium effect size in the reduction of heavy drinking days. Furthermore, in the intervention group there was a trend toward better diabetes adherence behavior.
Implementing brief intervention in practice. Despite differences among interventions, the elements of brief interventions tend to be similar.48 Incorporating these elements in the primary care setting provides a useful framework that will likely prove beneficial. Specifically, brief interventions typically contain elements of the FRAMES (TABLE 2)46 acronym:
- Feedback about one’s drinking relative to others
- Responsibility for deciding to change
- Advice to change drinking
- Menu of options for implementing a change strategy
- Empathic listening
- Self-efficacy enhancement.
Decision-making models indicate that expectations about the effects of behavior change play a significant role in determining whether a decision to change is made.49 The perceived costs and benefits of changing drinking50,51 and positive52 and negative alcohol expectancies53,54 predict future alcohol use. For patients with diabetes who are at-risk drinkers, primary care appointments may provide “teachable moments” in which brief advice can have a significant impact—particularly when patients are told laboratory test results; advised about the sugar and carbohydrate content of alcohol; or given information regarding the effect of alcohol on diabetes, medications, and self-care behavior. Finally, primary care providers will also likely have knowledge of a patient’s comorbid conditions (eg, depression) that may relate to diabetes or alcohol use.
TABLE 2
How to implement the FRAMES approach in brief interventions46
| FRAMES elements | Sample statements |
|---|---|
| Feedback about one’s drinking relative to others | “Based on what you told me, you are drinking an average of 21 drinks per week, which puts you above the cutoff for at-risk drinking” or “According to what you told me, it looks like you are drinking more than 88% of men (or women) in the United States.” |
| Responsibility for deciding to change | “You’re in the best position to decide how you would like to make a change” or “Although reducing your drinking would be good for your health, it’s up to you to decide when you’re ready to make a change.” |
| Advice to change drinking | “Your test results indicate your HbA1c, an important measure of blood sugar, is elevated; making a change in your drinking will likely improve your blood sugar” or “I am concerned about the effect on your health of drinking alcohol while taking your diabetes medications. Making a change in your drinking is likely to protect you from complications.” |
| Menu of options for implementing a change strategy | “If it’s okay with you, I can share what has worked for others whose drinking is similar to yours. Some people alternate a drink containing alcohol with water or diet soda. Others will bring only a certain amount of money with them when they go to a bar.” |
| Empathic listening | “It sounds like this has been a concern" or "I know that change can be difficult.” |
| Self-efficacy enhancement | “I wonder if you could use some of the same strategies you used to lose 10 pounds last year?” or “It sounds like you have some ideas for how to make this happen.” |
CORRESPONDENCE Patricia A. Engler, PhD, DGIM, 111 Plain Street Building, Providence, RI 02903; [email protected]
• Ask a question such as “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” to ascertain a patient’s quantity of alcohol use. A
• Apply elements of the FRAMES approach to help patients curtail at-risk drinking—eg, use elevated HbA1c levels as evidence of a need to change behavior. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
There are enough challenges in controlling diabetes mellitus without the hindrance of undetected problematic alcohol use. The good news is that asking a single nonthreatening question can help you detect at-risk drinking—defined by the National Institute on Alcohol Abuse and Alcoholism (NIAAA) as 5 or more drinks on one occasion or more than 14 drinks per week for men; and 4 or more drinks on one occasion or more than 7 drinks per week for women.1,2 And, for patients who may be compromising their diabetes care and overall health through problem drinking, brief intervention techniques used in the office can enable them to reduce alcohol consumption significantly.
When alcohol becomes a problem in diabetes care
Several studies have explored the long-term benefits of moderate alcohol use on glycemic control—with mixed results. A 2007 study found that diabetes patients who drink 1 glass of wine per day exhibited a lower fasting glucose level than abstainers after 3 months.3 There was no difference, however, on postprandial glucose levels. A 2008 study found that individuals who drank one to 2 glasses of wine per day for a month had lower fasting serum insulin levels relative to when they have abstained for a month,4 although levels of fasting plasma cholesterol, HDL cholesterol, glucose, and hemoglobin A1c (HbA1c) remained unchanged relative to periods of abstinence.4
Furthermore, rates of coronary heart disease and CHD mortality in a meta-analysis were significantly lower in 3 categories of alcohol consumption (<6 g/d, 6 to <18 g/d, and ≥18 g/d) compared with abstinence.5 Nondrinkers also had a greater risk of total mortality compared with the lightest drinking group. Notably, however, the lower limit of the highest drinking category was only 1.5 drinks per day.
How big is the problem? In a study of insulin-treated patients seen for severe hypoglycemia, 17% had been drinking before the episode.6 In a primary care sample, 28% of randomly selected patients with type 2 diabetes met Diagnostic and Statistical Manual of Mental Disorders-IV criteria for a lifetime incidence of alcohol abuse and 13% met either current or lifetime criteria for alcohol dependence.7 Another study of primary care patients with diabetes8 found that 13.4% met NIAAA criteria for at-risk drinking; 11.1% of these at-risk drinkers met criteria for current alcohol dependence. (According to the NIAAA, the rate of at-risk/heavy drinking among US adults is 30%, and about one in 4 heavy drinkers meets the criteria for alcohol abuse or dependence.1)
Detrimental effects with immoderate drinking. Individuals who engage in at-risk drinking, as defined by the NIAAA, are at increased risk for alcohol dependence9 and associated complications such as diabetic neuropathy and retinopathy,10 atherosclerosis,11 and total and CHD mortality.3,12 Heavy drinking also interferes with neuroendocrine, gastrointestinal, and sexual function,13 and its interaction with diabetes increases the risk for hepatocellular carcinoma after controlling for hepatitis B and C serology.14
Interference with diabetes control. Research examining the short-term effect of alcohol use has produced contradictory results, partly due to differences among studies, such as whether alcohol is administered with a meal and whether a fasting glucose level is measured.15 However, alcohol affects glycemic control and, when used excessively, can impair glucose production.16,17 Alcohol may induce hypoglycemia,10,18 and even small amounts may jeopardize diabetes control.13 In a study of patients with insulin-treated diabetes, alcohol use in the presence of mild hypoglycemia increased diastolic blood pressure or exacerbated hypoglycemia-related cognitive deficits.19 Another concern—in both the short and long term—is that alcohol interacts negatively with certain diabetes medications. It is more likely to induce hypoglycemia in the presence of sulfonylurea medications.10 Chlorpropamide decreases the rate of ethanol elimination from the blood.20 And, in those taking metformin, excessive alcohol use elevates risk for lactic acidosis. 21
Diminished self-care. Alcohol use can interfere with self-care,22,23 which is a crucial component of diabetes treatment.24 It may lead to reduced eating16 or to decreased willingness to adhere to prescribed dietary regimens.13 It also impairs other self-care behaviors13,15,25 such as self-monitoring blood glucose and showing up for medical appointments.26 In a large, diverse sample of patients with diabetes,24 heavy drinkers had the highest rates of morbidity. Importantly, alcohol and diabetes self-care behavior were significantly negatively associated. Studies of ethnic minority samples have yielded comparable results.27
Assessing alcohol use: Obstacles and solutions
Although alcohol use can be readily evaluated during routine primary care appointments, it is often neglected, perhaps due to a lack of awareness about its impact on diabetes.15 Those who are most often assessed tend to have a psychiatric diagnosis or other condition raising a red flag for physicians.28 When internists, general practitioners, and psychiatrists were questioned in a study regarding patients’ alcohol and drug use, all 3 groups were misinformed about which substance-use treatments were empirically supported29 and did not believe that treatment for alcohol abuse held much promise. Another study showed that physicians can be reluctant to screen for alcohol use because of the difficulty in recognizing a problem, the perceived unimportance of alcohol use as a health risk, a supposed lack of adequate intervention tools, and a fear of stigmatizing patients.30 Physicians are more likely to discuss alcohol use under certain extreme conditions such as when a patient smells of alcohol.
Multiple opportunities to ask in the VA system. In the Veterans Health Administration, primary care VA providers have reported that prompts for alcohol screening embedded in computerized progress notes, clinical reminder lists, and annual health evaluation forms encourage them to assess alcohol use. Other useful materials include manual checklists and reference cards.31 These providers also report that education, feedback on rates of alcohol screening, and increased supervision facilitate assessment. Finally, providers indicate that asking nurses or clerical staff to administer the screen improves completion rates.
Ask a simple question
“How often have you had a drink containing alcohol in the last year?” or “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” are questions that can help you compare a patient’s alcohol use to the at-risk drinking cutoffs established by the NIAAA.1,2
Recent research has also validated the use of a single question in identifying NIAAA-defined at-risk drinking.32 Simply ask patients, “How many times in the past year have you had X or more drinks in a day?” (X=5 for men or 4 for women). The screen is positive when a patient acknowledges having done so at least once in the past year. This question was 81.8% sensitive and 79.3% specific for unhealthy alcohol use, and 87.9% sensitive and 66.8% specific for current alcohol abuse or dependence.32 Advantages of this method are its brevity, ease of scoring, validity in the primary care setting,32 and ease of recollection for treatment providers (TABLE 1).1,2
TABLE 1
Ask these simple questions to assess alcohol use1,2
| To assess… | Ask… |
|---|---|
| Frequency of alcohol use | “How often do you drink alcohol (including beer or wine)?” or “How often have you had a drink containing alcohol in the last year?” |
| Quantity of alcohol use | “When you do drink alcohol, how many standard* drinks do you have?” or “How many drinks containing alcohol did you have on a typical day when you were drinking in the last year?” |
| Binge drinking | For men: “How often do you have 5 or more standard* drinks on one occasion?” For women: “How often do you have 4 or more standard* drinks on one occasion?” |
| Alcohol use with the NIAAA single-question screen | “How many times in the past year have you had X or more drinks in a day?” (X = 5 for men and X = 4 for women; positive response ≥1) |
| *A standard drink is equal to 12 oz. of regular beer, 8 to 9 oz. of malt liquor, 5 oz. of wine, or 1.5 oz. of 80-proof spirits.1 NIAAA, National Institute on Alcohol Abuse and Alcoholism. | |
Brief intervention works in primary care
Brief interventions for drinking have strong empirical support. In a review of treatments for alcohol abuse and dependence,33 brief intervention was one of only 2 “efficacious” treatments.
Although some individual studies of brief alcohol interventions in primary care have not shown favorable results, several systematic reviews have demonstrated the efficacy of such interventions in this setting. General practitioner–delivered brief interventions led to significantly better patient outcomes compared with standard care, and “very brief advice” resulted in reductions in alcohol consumption overall and in the percentage of “excessive drinkers.”34 In a review of health behavior interventions,35 brief interventions reduced risky or harmful drinking. In one of 2 meta-analyses that support this finding, brief interventions with primary care patients not seeking treatment for alcohol abuse yielded small-to-medium effect sizes relative to control conditions.36 In the other study, brief interventions significantly reduced longer term alcohol use in primary care patients.37
The US Preventive Services Task Force conducted a systematic review of behavioral counseling interventions and recommends screening and brief interventions for unhealthy drinking in primary care.38,39 Its findings indicate that alcohol use declines significantly after brief interventions containing at least 2 of the following elements: feedback regarding drinking, advice to reduce drinking, or goal setting.
Brief advice is a form of intervention that shows considerable promise in primary care.40-42 Two 10- to 15-minute sessions have led to significant reductions in the mean number of drinks and frequency of excessive drinking in the 7 days before a follow-up interview, as well as a reduction in binge drinking episodes in the previous 30 days.41,42
One study produced positive results with just a 5- to 10-minute counseling session involving advice for drinking goals delivered by primary care providers as part of a routine medical visit.43 This intervention led to significant decreases in alcohol use at a 6-month follow-up for high-risk drinkers compared with controls.43 Brief interventions additionally work within the time constraints of a busy primary care practice and are cost effective.
Some primary care providers think a specialist should conduct interventions and suggest that having a specialist immediately available would enable intervention.31 In fact, some research has supported the idea of special training. In a European study, primary care providers reported that more practical training, information about brief intervention studies, personal training, and lectures would facilitate interventions.44
Applying brief alcohol interventions to diabetes patients
Newer research has tested the efficacy of alcohol interventions with diabetes patients in the primary care setting. In one study,45 brief advice was given in 2 15-minute sessions and 2 5-minute follow-up telephone calls. Compared with controls, significantly more participants who received the intervention reduced heavy drinking from baseline to follow-up. One caveat is that patients with hypertension were included in the sample, making it difficult to determine the impact of the intervention on diabetes patients specifically.
In a small study of patients with diabetes exhibiting at-risk drinking,8 a single-session intervention based on motivational interviewing (MI) principles46 gave participants personalized feedback in relation to sex-based norms of drinking rates and HbA1c and triglyceride laboratory results. Patients were given information on the physiologic effects of alcohol on diabetes, the potential interactions between alcohol and diabetes medications, and the effect of alcohol on diabetes self-care behavior. They were asked to identify pros and cons of their drinking and to develop personal change goals. One of 2 PhD-level clinical psychologists trained in MI administered the single 50-minute intervention. By 1 month and continuing through to the 6-month follow-up, participants had reduced the proportion of drinking days, mean number of daily drinks, and proportion of heavy drinking days.
Ramsey and colleagues47 extended this work by comparatively examining a group of patients exhibiting at-risk drinking who received no intervention. The results favored the intervention group, with a medium-to-large effect size for the proportion of drinking days, a medium effect size for the reduction of mean number of daily drinks, and a small-to-medium effect size in the reduction of heavy drinking days. Furthermore, in the intervention group there was a trend toward better diabetes adherence behavior.
Implementing brief intervention in practice. Despite differences among interventions, the elements of brief interventions tend to be similar.48 Incorporating these elements in the primary care setting provides a useful framework that will likely prove beneficial. Specifically, brief interventions typically contain elements of the FRAMES (TABLE 2)46 acronym:
- Feedback about one’s drinking relative to others
- Responsibility for deciding to change
- Advice to change drinking
- Menu of options for implementing a change strategy
- Empathic listening
- Self-efficacy enhancement.
Decision-making models indicate that expectations about the effects of behavior change play a significant role in determining whether a decision to change is made.49 The perceived costs and benefits of changing drinking50,51 and positive52 and negative alcohol expectancies53,54 predict future alcohol use. For patients with diabetes who are at-risk drinkers, primary care appointments may provide “teachable moments” in which brief advice can have a significant impact—particularly when patients are told laboratory test results; advised about the sugar and carbohydrate content of alcohol; or given information regarding the effect of alcohol on diabetes, medications, and self-care behavior. Finally, primary care providers will also likely have knowledge of a patient’s comorbid conditions (eg, depression) that may relate to diabetes or alcohol use.
TABLE 2
How to implement the FRAMES approach in brief interventions46
| FRAMES elements | Sample statements |
|---|---|
| Feedback about one’s drinking relative to others | “Based on what you told me, you are drinking an average of 21 drinks per week, which puts you above the cutoff for at-risk drinking” or “According to what you told me, it looks like you are drinking more than 88% of men (or women) in the United States.” |
| Responsibility for deciding to change | “You’re in the best position to decide how you would like to make a change” or “Although reducing your drinking would be good for your health, it’s up to you to decide when you’re ready to make a change.” |
| Advice to change drinking | “Your test results indicate your HbA1c, an important measure of blood sugar, is elevated; making a change in your drinking will likely improve your blood sugar” or “I am concerned about the effect on your health of drinking alcohol while taking your diabetes medications. Making a change in your drinking is likely to protect you from complications.” |
| Menu of options for implementing a change strategy | “If it’s okay with you, I can share what has worked for others whose drinking is similar to yours. Some people alternate a drink containing alcohol with water or diet soda. Others will bring only a certain amount of money with them when they go to a bar.” |
| Empathic listening | “It sounds like this has been a concern" or "I know that change can be difficult.” |
| Self-efficacy enhancement | “I wonder if you could use some of the same strategies you used to lose 10 pounds last year?” or “It sounds like you have some ideas for how to make this happen.” |
CORRESPONDENCE Patricia A. Engler, PhD, DGIM, 111 Plain Street Building, Providence, RI 02903; [email protected]
1. National Institute on Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician’s guide. 2005. Available at: http://pubs.niaaa.nih.gov/publications/Practitioner/CliniciansGuide2005/guide.pdf. Accessed November 8, 2011.
2. Bradley KA, Kivlahan DR, Williams E. Brief approaches to alcohol screening: practical alternatives for primary care. J Gen Intern Med 2009;24:881-883.
3. Shai I, Fraser D, Wainstein J, et al. Glycemic effects of moderate alcohol intake among patients with type 2 diabetes. Diabetes Care 2007;30:3011-3016.
4. Bantle AE, Tomas W, Bantle JP. Metabolic effects of alcohol in the form of wine in persons with type 2 diabetes mellitus. Metabolism 2008;57:241-245.
5. Koppes LLJ, Dekker JM, Hendriks HFJ, et al. Meta-analysis of the relationship between alcohol consumption and coronary heart disease and mortality in type 2 diabetic patients. Diabetologia 2006;49:648-652.
6. Pedersen-Bjergaard U, Reubsaet JLE, Nielsen SL, et al. Psychoactive drugs, alcohol, and severe hypoglycemia in insulin-treated diabetes: analysis of 141 cases. Am J Med 2005;118:307-310.
7. Fleming M, Mundt M. Carbohydrate-deficient transferrin: validity of a new alcohol biomarker in a sample of patients with diabetes and hypertension. J Am Board Fam Pract 2004;17:247-255.
8. Engler PA, Ramsey SE, Stein MD. Brief alcohol intervention among diabetic patients: a pilot study. Presented at: Annual Meeting of the Society for Behavioral Medicine; March 26–29, 2008; San Diego, CA.
9. Saha TD, Stinson FS, Grant BF. The role of alcohol consumption in future classifications of alcohol use disorders. Drug Alcohol Depend 2007;89:82-92.
10. Shai I, Rimm EB, Schulze MB, et al. Moderate alcohol intake and markers of inflammation and endothelial dysfunction among diabetic men. Diabetologia 2004;14:1760-1767.
11. Wakabayashi I, Kobaba-Wakabayashi R, Masuda H. Relation of drinking alcohol to atherosclerotic risk in type 2 diabetes. Diabetes Care 2002;25:1223-1228.
12. Diem M, Deplazes M, Fajfr R, et al. Effects of alcohol consumption on mortality in patients with type 2 diabetes mellitus. Diabetologia 2003;46:1581-1585.
13. Cox WM, Blount JP, Crowe PA, et al. Diabetic patients’ alcohol use and quality of life: relationships with prescribed treatment compliance among older males. Alcohol Clin Exp Res 1996;20:327-331.
14. Yuan JM, Govindarajan S, Arakawa K, et al. Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer 2004;101:1009-1017.
15. Howard AA, Arnsten JH, Gourevitch MN. Effect of alcohol consumption on diabetes mellitus. Ann Intern Med 2004;140:211-219.
16. Glasgow AM, Tynan D, Schwartz R, et al. Alcohol and drug use in teenagers with diabetes mellitus. J Adolesc Health 1991;12:11-14.
17. Turner BC, Jenkins E, Kerr D, et al. The effect of evening alcohol consumption on next-morning glucose control in type 1 diabetes. Diabetes Care 2001;24:1888-1893.
18. Richardson T, Weiss M, Thomas P, et al. Day after the night before. Influence of evening alcohol on risk of hypoglycemia in patients with type 1 diabetes. Diabetes Care. 2005;28:1801-1802.
19. Cheyne EH, Sherwin RS, Lunt MJ, et al. Influence of alcohol on cognitive performance during mild hypoglycaemia: implications for type 1 diabetes. Diabet Med 2004;21:230-237.
20. Lao B, Czyzyk A, Szutowski M, et al. Alcohol tolerance in patients with non-insulin-dependent (type 2) diabetes treated with sulphonylurea derivatives. Arzneimittelforschung 1994;44:727-734.
21. PDR Staff Physicians’ Desk Reference 2003. 57th ed. Montvale, NJ: Medical Economics Company; 2003.
22. Ramchandani N, Cantey-Kiser JM, Alter CA, et al. Self-reported factors that affect glycemic control in college students with type 1 diabetes. Diabetes Educ 2000;26:656-666.
23. Kyngas H. Compliance of adolescents with chronic disease. J Clin Nurs 2000;9:549-556.
24. Ahmed AT, Karter AJ, Liu J. Alcohol consumption is inversely associated with adherence to diabetes self-care behaviours. Diabet Med 2006;23:795-802.
25. Karter AJ, Ferrara A, Darbinian JA, et al. Self-monitoring of blood glucose. Diabetes Care 2004;23:477-483.
26. Chew LD, Nelson KM, Young BA, et al. Association between alcohol consumption and diabetes preventative practices. Fam Med 2005;37:589-594.
27. Johnson KH, Bazargan M, Bing E. Alcohol consumption and compliance among inner-city minority patients with type 2 diabetes mellitus. Arch Fam Med 2000;9:964-970.
28. D’Amico EJ, Paddock SM, Burnam A, et al. Identification of and guidance for problem drinking by general medical providers. Med Care 2005;43:229-236.
29. Roche AM, Parle MD, Stubbs JM, et al. Management and treatment efficacy of drug and alcohol problems: what do doctors believe? Addiction. 1995;90:1357-1366.
30. Aira M, Kauhanen J, Larivaara P, et al. Differences in brief interventions on excessive drinking and smoking by primary care physicians: qualitative study. Prev Med 2004;38:473-478.
31. Barry KL, Blow FC, Willenbring M, et al. Use of alcohol screening and brief interventions in primary care settings: implementation and barriers. Subst Abus 2004;25:27-36.
32. Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single-question alcohol screening test. J Gen Intern Med 2009;24:783-788.
33. McCrady BS. Alcohol use disorders and the Division 12 Task Force of the American Psychological Association. Psychol Addict Behav 2000;14:267-276.
34. Richmond RL, Anderson P. Research in general practice for smokers and excessive drinkers in Australia and the UK. I. Interpretation of results. Addiction 1994;89:35-40.
35. Goldstein MG, Whitlock EP, DePue J. Multiple behavioral risk factor interventions in primary care. Am J Prev Med 2004;27:61-79.
36. Moyer A, Finney JW, Swearingen CE, et al. Brief interventions for alcohol problems: a meta-analytic review of controlled investigations in treatment-seeking and non-treatment-seeking populations. Addiction 2002;97:279-292.
37. Bertholet N, Daeppen JB, Wietlisbach V, et al. Reduction of alcohol consumption by brief alcohol intervention in primary care: a systematic review and meta-analysis. Arch Intern Med 2005;165:986-995.
38. Whitlock EP, Polen MR, Green CA, et al. Behavioral counseling interventions in primary care to reduce risky/harmful alcohol use by adults: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2004;140:557-568.
39. U. S. Preventive Services Task Force. Screening for problem drinking. In: DiGuiseppi C, Atkins D, Woolf SH, Kamerow DB, eds. Guide to Clinical Preventive Services. 2nd ed. Alexandria, VA: International Medical Services; 1996;567-582.
40. Anderson P, Scott E. The effect of general practitioners’ advice to heavy drinking men. Br J Addict 1992;87:891-900.
41. Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res 2002;26:36-43.
42. Fleming MF, Barry KL, Manwell LB, et al. Brief physician advice for problem alcohol drinkers. JAMA 1997;277:1039-1045.
43. Ockene JK, Adams A, Hurley TG, et al. Brief physician- and nurse practitioner-delivered counseling for high-risk drinkers. Arch Intern Med 1999;159:2198-2205.
44. Aalto M, Pekuri P, Seppa K. Primary health care nurses’ and physicians’ attitudes, knowledge and beliefs regarding brief intervention for heavy drinkers. Addiction 2001;96:305-311.
45. Fleming M, Brown R, Brown D. The efficacy of a brief alcohol intervention combined with %CDT feedback in patients being treated for type 2 diabetes and/or hypertension. J Stud Alcohol 2004;65:631-637.
46. Miller WR, Rollnick S. Motivational Interviewing: Preparing People for Change. New York: Guilford Press; 2002.
47. Ramsey SE, Engler PA, Harrington M, et al. A brief alcohol intervention with at-risk drinking diabetics. Subst Abus 2010;4:1-8.
48. Bien TH, Miller WR, Tonigan JS. Brief interventions for alcohol problems: a review. Addiction 1993;88:315-335.
49. Sutton S. Social-psychological approaches to understanding addictive behaviours: attitude-behaviour and decision-making models. Br J Addict 1987;82:355-370.
50. Cunningham JA, Sobell LC, Gavin DR, et al. Assessing motivation for change: preliminary development and evaluation of a scale measuring the costs and benefits of changing alcohol or drug use. Psychol Addict Behav 1997;11:107-114.
51. Rollnick S, Morgan M, Heather N. The development of a brief scale to measure outcome expectations of reduced consumption among excessive drinkers. Addict Behav 1996;21:377-387.
52. Brown SA. Reinforcement expectancies and alcoholism treatment outcome after a one-year follow-up. J Stud Alcohol 1985;46:304-308.
53. Jones BT, McMahon J. Negative alcohol expectancy predicts post-treatment abstinence survivorship: the whether, when and why of relapse to a first drink. Addiction 1994;89:1653-1665.
54. Jones BT, McMahon J. Negative and positive alcohol expectancies as predictors of abstinence after discharge from a residential treatment program: a one-month and three-month follow-up study in men. J Stud Alcohol 1994;55:543-548.
1. National Institute on Alcohol Abuse and Alcoholism. Helping patients who drink too much: a clinician’s guide. 2005. Available at: http://pubs.niaaa.nih.gov/publications/Practitioner/CliniciansGuide2005/guide.pdf. Accessed November 8, 2011.
2. Bradley KA, Kivlahan DR, Williams E. Brief approaches to alcohol screening: practical alternatives for primary care. J Gen Intern Med 2009;24:881-883.
3. Shai I, Fraser D, Wainstein J, et al. Glycemic effects of moderate alcohol intake among patients with type 2 diabetes. Diabetes Care 2007;30:3011-3016.
4. Bantle AE, Tomas W, Bantle JP. Metabolic effects of alcohol in the form of wine in persons with type 2 diabetes mellitus. Metabolism 2008;57:241-245.
5. Koppes LLJ, Dekker JM, Hendriks HFJ, et al. Meta-analysis of the relationship between alcohol consumption and coronary heart disease and mortality in type 2 diabetic patients. Diabetologia 2006;49:648-652.
6. Pedersen-Bjergaard U, Reubsaet JLE, Nielsen SL, et al. Psychoactive drugs, alcohol, and severe hypoglycemia in insulin-treated diabetes: analysis of 141 cases. Am J Med 2005;118:307-310.
7. Fleming M, Mundt M. Carbohydrate-deficient transferrin: validity of a new alcohol biomarker in a sample of patients with diabetes and hypertension. J Am Board Fam Pract 2004;17:247-255.
8. Engler PA, Ramsey SE, Stein MD. Brief alcohol intervention among diabetic patients: a pilot study. Presented at: Annual Meeting of the Society for Behavioral Medicine; March 26–29, 2008; San Diego, CA.
9. Saha TD, Stinson FS, Grant BF. The role of alcohol consumption in future classifications of alcohol use disorders. Drug Alcohol Depend 2007;89:82-92.
10. Shai I, Rimm EB, Schulze MB, et al. Moderate alcohol intake and markers of inflammation and endothelial dysfunction among diabetic men. Diabetologia 2004;14:1760-1767.
11. Wakabayashi I, Kobaba-Wakabayashi R, Masuda H. Relation of drinking alcohol to atherosclerotic risk in type 2 diabetes. Diabetes Care 2002;25:1223-1228.
12. Diem M, Deplazes M, Fajfr R, et al. Effects of alcohol consumption on mortality in patients with type 2 diabetes mellitus. Diabetologia 2003;46:1581-1585.
13. Cox WM, Blount JP, Crowe PA, et al. Diabetic patients’ alcohol use and quality of life: relationships with prescribed treatment compliance among older males. Alcohol Clin Exp Res 1996;20:327-331.
14. Yuan JM, Govindarajan S, Arakawa K, et al. Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer 2004;101:1009-1017.
15. Howard AA, Arnsten JH, Gourevitch MN. Effect of alcohol consumption on diabetes mellitus. Ann Intern Med 2004;140:211-219.
16. Glasgow AM, Tynan D, Schwartz R, et al. Alcohol and drug use in teenagers with diabetes mellitus. J Adolesc Health 1991;12:11-14.
17. Turner BC, Jenkins E, Kerr D, et al. The effect of evening alcohol consumption on next-morning glucose control in type 1 diabetes. Diabetes Care 2001;24:1888-1893.
18. Richardson T, Weiss M, Thomas P, et al. Day after the night before. Influence of evening alcohol on risk of hypoglycemia in patients with type 1 diabetes. Diabetes Care. 2005;28:1801-1802.
19. Cheyne EH, Sherwin RS, Lunt MJ, et al. Influence of alcohol on cognitive performance during mild hypoglycaemia: implications for type 1 diabetes. Diabet Med 2004;21:230-237.
20. Lao B, Czyzyk A, Szutowski M, et al. Alcohol tolerance in patients with non-insulin-dependent (type 2) diabetes treated with sulphonylurea derivatives. Arzneimittelforschung 1994;44:727-734.
21. PDR Staff Physicians’ Desk Reference 2003. 57th ed. Montvale, NJ: Medical Economics Company; 2003.
22. Ramchandani N, Cantey-Kiser JM, Alter CA, et al. Self-reported factors that affect glycemic control in college students with type 1 diabetes. Diabetes Educ 2000;26:656-666.
23. Kyngas H. Compliance of adolescents with chronic disease. J Clin Nurs 2000;9:549-556.
24. Ahmed AT, Karter AJ, Liu J. Alcohol consumption is inversely associated with adherence to diabetes self-care behaviours. Diabet Med 2006;23:795-802.
25. Karter AJ, Ferrara A, Darbinian JA, et al. Self-monitoring of blood glucose. Diabetes Care 2004;23:477-483.
26. Chew LD, Nelson KM, Young BA, et al. Association between alcohol consumption and diabetes preventative practices. Fam Med 2005;37:589-594.
27. Johnson KH, Bazargan M, Bing E. Alcohol consumption and compliance among inner-city minority patients with type 2 diabetes mellitus. Arch Fam Med 2000;9:964-970.
28. D’Amico EJ, Paddock SM, Burnam A, et al. Identification of and guidance for problem drinking by general medical providers. Med Care 2005;43:229-236.
29. Roche AM, Parle MD, Stubbs JM, et al. Management and treatment efficacy of drug and alcohol problems: what do doctors believe? Addiction. 1995;90:1357-1366.
30. Aira M, Kauhanen J, Larivaara P, et al. Differences in brief interventions on excessive drinking and smoking by primary care physicians: qualitative study. Prev Med 2004;38:473-478.
31. Barry KL, Blow FC, Willenbring M, et al. Use of alcohol screening and brief interventions in primary care settings: implementation and barriers. Subst Abus 2004;25:27-36.
32. Smith PC, Schmidt SM, Allensworth-Davies D, et al. Primary care validation of a single-question alcohol screening test. J Gen Intern Med 2009;24:783-788.
33. McCrady BS. Alcohol use disorders and the Division 12 Task Force of the American Psychological Association. Psychol Addict Behav 2000;14:267-276.
34. Richmond RL, Anderson P. Research in general practice for smokers and excessive drinkers in Australia and the UK. I. Interpretation of results. Addiction 1994;89:35-40.
35. Goldstein MG, Whitlock EP, DePue J. Multiple behavioral risk factor interventions in primary care. Am J Prev Med 2004;27:61-79.
36. Moyer A, Finney JW, Swearingen CE, et al. Brief interventions for alcohol problems: a meta-analytic review of controlled investigations in treatment-seeking and non-treatment-seeking populations. Addiction 2002;97:279-292.
37. Bertholet N, Daeppen JB, Wietlisbach V, et al. Reduction of alcohol consumption by brief alcohol intervention in primary care: a systematic review and meta-analysis. Arch Intern Med 2005;165:986-995.
38. Whitlock EP, Polen MR, Green CA, et al. Behavioral counseling interventions in primary care to reduce risky/harmful alcohol use by adults: a summary of the evidence for the U.S. Preventive Services Task Force. Ann Intern Med 2004;140:557-568.
39. U. S. Preventive Services Task Force. Screening for problem drinking. In: DiGuiseppi C, Atkins D, Woolf SH, Kamerow DB, eds. Guide to Clinical Preventive Services. 2nd ed. Alexandria, VA: International Medical Services; 1996;567-582.
40. Anderson P, Scott E. The effect of general practitioners’ advice to heavy drinking men. Br J Addict 1992;87:891-900.
41. Fleming MF, Mundt MP, French MT, et al. Brief physician advice for problem drinkers: long-term efficacy and benefit-cost analysis. Alcohol Clin Exp Res 2002;26:36-43.
42. Fleming MF, Barry KL, Manwell LB, et al. Brief physician advice for problem alcohol drinkers. JAMA 1997;277:1039-1045.
43. Ockene JK, Adams A, Hurley TG, et al. Brief physician- and nurse practitioner-delivered counseling for high-risk drinkers. Arch Intern Med 1999;159:2198-2205.
44. Aalto M, Pekuri P, Seppa K. Primary health care nurses’ and physicians’ attitudes, knowledge and beliefs regarding brief intervention for heavy drinkers. Addiction 2001;96:305-311.
45. Fleming M, Brown R, Brown D. The efficacy of a brief alcohol intervention combined with %CDT feedback in patients being treated for type 2 diabetes and/or hypertension. J Stud Alcohol 2004;65:631-637.
46. Miller WR, Rollnick S. Motivational Interviewing: Preparing People for Change. New York: Guilford Press; 2002.
47. Ramsey SE, Engler PA, Harrington M, et al. A brief alcohol intervention with at-risk drinking diabetics. Subst Abus 2010;4:1-8.
48. Bien TH, Miller WR, Tonigan JS. Brief interventions for alcohol problems: a review. Addiction 1993;88:315-335.
49. Sutton S. Social-psychological approaches to understanding addictive behaviours: attitude-behaviour and decision-making models. Br J Addict 1987;82:355-370.
50. Cunningham JA, Sobell LC, Gavin DR, et al. Assessing motivation for change: preliminary development and evaluation of a scale measuring the costs and benefits of changing alcohol or drug use. Psychol Addict Behav 1997;11:107-114.
51. Rollnick S, Morgan M, Heather N. The development of a brief scale to measure outcome expectations of reduced consumption among excessive drinkers. Addict Behav 1996;21:377-387.
52. Brown SA. Reinforcement expectancies and alcoholism treatment outcome after a one-year follow-up. J Stud Alcohol 1985;46:304-308.
53. Jones BT, McMahon J. Negative alcohol expectancy predicts post-treatment abstinence survivorship: the whether, when and why of relapse to a first drink. Addiction 1994;89:1653-1665.
54. Jones BT, McMahon J. Negative and positive alcohol expectancies as predictors of abstinence after discharge from a residential treatment program: a one-month and three-month follow-up study in men. J Stud Alcohol 1994;55:543-548.
CDC update on gonorrhea: Expand treatment to limit resistance
Public health efforts have decreased the incidence of gonorrhea over the past several decades, but this progress is threatened by emergent bacteria resistance to the few remaining antibiotics available to treat it.
Gonococcal resistance to penicillin and tetracycline began in the 1970s and was widespread by the 1980s. Resistance to fluoroquinolones developed during the last decade and led the Centers for Disease Control and Prevention (CDC) in 2007 to stop recommending this class of antibiotics for treatment of gonorrhea.1 (See “The decline of gonorrhea: A success story now threatened by antibiotic resistance”.)
Given the speed with which gonococci developed resistance to fluoroquinolones, the CDC sees as inevitable the eventual development of resistance to cephalosporins—the currently favored agents for gonorrhea.2 This is the main reason behind the new recommendations for treating all cases of gonorrhea with both a cephalosporin and azithromycin, whether or not co-infection with Chlamydia trachomatis is documented or suspected.3
The reported rate of gonorrhea rose steadily from the early 1960s until the mid-1970s, when it was close to 500 cases per 100,000. With implementation of the national gonorrhea control program, the annual rate began to fall, and by the mid-1990s it had declined by 74%. Between 1996 and 2006, the rate remained at about 115 cases per 100,000. Between 2006 and 2009, it decreased to the lowest rate since national reporting began, but increased 2.8% between 2009 and 2010 (FIGURE 1).
The highest rates of gonorrhea are in the South (FIGURE 2) and in women ages 15 to 24 and men ages 20 to 24 (FIGURE 3). Rates are highest among blacks (432.5 cases per 100,000), followed by American Indians/Alaska natives (105.7) and Hispanics (49.9). Between 2009 and 2010, gonorrhea rates increased among American Indians/Alaska natives (21.5%), Asians/Pacific Islanders (13.1%), Hispanics (11.9%), whites (9.0%), and blacks (0.3%).
Recent trends in Gonococcus susceptibility to cephalosporins have the CDC concerned. While cephalosporin resistance remains rare, the proportion of Gonococcus isolates that have shown elevated minimum inhibitory concentrations to cephalosporins has increased.
Gonorrhea control depends in part on appropriate screening of individuals at risk (TABLE). Risk factors for gonorrhea include a history of previous gonorrhea infection, other sexually transmitted infections, new or multiple sexual partners, inconsistent condom use, sex work, and drug use. Risk factors for pregnant women are the same as for nonpregnant women. Prevalence of gonorrhea infection varies widely among communities and patient populations.
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 1
A decline in gonorrhea that began in the mid-1970s*
*The initiation of a national gonorrhea control program reaped immediate benefits that have continued through the present.
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates, United States, 1941-2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 2
Gonorrhea prevalence, 2010
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates by state, United States and outlying areas, 2010. Available at: http://www.cdc.gov/std/10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 3
Gonorrhea prevalence by age and sex, 2010
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates by age and sex, United States, 2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
Augment therapy, follow up thoroughly
Family physicians can assist with public health efforts to control gonorrhea and delay the development of cephalosporin resistance by screening for and detecting the infection, diagnosing those with symptoms, and treating according to newer recommendations. It’s also essential to report cases to local public health departments, assist with finding and treating sexual contacts of infected individuals, and immediately report suspected treatment failures.
A 2-drug regimen is imperative. The latest recommendation for treating gonorrhea is ceftriaxone 250 mg IM in a single dose and azithromycin 1 g orally in a single dose. Until 2010, the dose of ceftriaxone had been 125 mg. This dual drug regimen is recommended for several reasons: As with using multiple drugs to treat tuberculosis, it is hoped dual drug therapy will slow development of resistance to both cephalosporins and azithromycin; co-infection with C trachomatis remains a significant problem; and the combination may be more effective against pharyngeal gonorrhea, which is hard to detect.
Alternative regimens. Cefixime 400 mg orally as a single dose is an option in lieu of ceftriaxone, but is not preferred because of the higher number of reported failures of treatment with oral cephalosporins and less efficacy against pharyngeal disease.3 Other injectable cephalosporins are also an option, but less is known about their effectiveness in treating pharyngeal infection. Injectable options include ceftizoxime 500 mg IM, cefoxitin 2 g IM with probenecid 1 g orally, and cefotaxime 500 mg IM.
Regardless of the cephalosporin chosen, always administer azithromycin. If necessary, an alternative to azithromycin is doxycycline 100 mg orally twice a day. But doxycycline is not preferred because it has a multiple daily dosing requirement and higher levels of gonococcal resistance than is seen with azithromycin.
Necessary follow-up. Although routine testing for cure is not advocated for those treated with a recommended antibiotic regimen, a gonococcal culture and testing for antibiotic susceptibility should be done for any patient whose symptoms persist after treatment. Rapid tests using nucleic acid amplification are unsuitable for testing antibiotic susceptibility. The CDC does recommend retesting patients 3 months after treatment is completed because of a high prevalence of reinfection.3 If cephalosporin resistance becomes prevalent, routine tests of cure might become a recommended standard.
Report all patients with gonorrhea to the local public health department so that sexual contacts within the past 60 days can be notified, tested, and treated presumptively with the dual drug regimen. Recommend simultaneous treatment for all current sex partners, and discourage sexual intercourse until symptoms have resolved. Promptly report any patient with suspected treatment failure to the local health department, and consult the local or state health department for recommendations on subsequent treatment regimens.
The US Preventive Services Task Force (USPSTF) recommends routine screening for asymptomatic infection in women at risk, as per the details in the TABLE.4 While the USPSTF found insufficient evidence to recommend screening of high-risk men, physicians might still consider screening men who have sex with multiple male partners.
TABLE
USPSTF recommendations on screening for gonorrhea4
|
|
|
|
| USPSTF, US Preventive Services Task Force. *For more on the USPSTF’s grade definitions, see: http://www.uspreventiveservicestaskforce.org/uspstf/gradespre.htm#brec. |
Doing our best in the face of uncertainty
Although evidence is lacking that dual drug therapy will delay the progression of resistance, the strategy makes empirical sense. If gonorrhea develops resistance to cephalosporins, it will seriously challenge public health efforts to control this infection. Family physicians have an important role in controlling this sexually transmitted infection and helping to prevent drug resistance.
1. Centers for Disease Control and Prevention. Update to CDC’s sexually transmitted disease treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR Morb Mortal Wkly Rep. 2007;56:332-336.
2. Centers for Disease Control and Prevention. Cephalosporin susceptibility among Neisseria gonorrhoeae isolates—United States 2000-2010. MMWR Morb Mortal Wkly Rep. 2011;60:873-877.
3. Workowski KA, Berman S. Centers for Disease Control and Prevention (CDC). Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep. 2010;59(RR-12):1-110.
4. US Preventive Services Task Force. Screening for gonorrhea. Available at: http://www.uspreventiveservicestaskforce.org/uspstf/uspsgono.htm. Accessed September 26, 2011.
Public health efforts have decreased the incidence of gonorrhea over the past several decades, but this progress is threatened by emergent bacteria resistance to the few remaining antibiotics available to treat it.
Gonococcal resistance to penicillin and tetracycline began in the 1970s and was widespread by the 1980s. Resistance to fluoroquinolones developed during the last decade and led the Centers for Disease Control and Prevention (CDC) in 2007 to stop recommending this class of antibiotics for treatment of gonorrhea.1 (See “The decline of gonorrhea: A success story now threatened by antibiotic resistance”.)
Given the speed with which gonococci developed resistance to fluoroquinolones, the CDC sees as inevitable the eventual development of resistance to cephalosporins—the currently favored agents for gonorrhea.2 This is the main reason behind the new recommendations for treating all cases of gonorrhea with both a cephalosporin and azithromycin, whether or not co-infection with Chlamydia trachomatis is documented or suspected.3
The reported rate of gonorrhea rose steadily from the early 1960s until the mid-1970s, when it was close to 500 cases per 100,000. With implementation of the national gonorrhea control program, the annual rate began to fall, and by the mid-1990s it had declined by 74%. Between 1996 and 2006, the rate remained at about 115 cases per 100,000. Between 2006 and 2009, it decreased to the lowest rate since national reporting began, but increased 2.8% between 2009 and 2010 (FIGURE 1).
The highest rates of gonorrhea are in the South (FIGURE 2) and in women ages 15 to 24 and men ages 20 to 24 (FIGURE 3). Rates are highest among blacks (432.5 cases per 100,000), followed by American Indians/Alaska natives (105.7) and Hispanics (49.9). Between 2009 and 2010, gonorrhea rates increased among American Indians/Alaska natives (21.5%), Asians/Pacific Islanders (13.1%), Hispanics (11.9%), whites (9.0%), and blacks (0.3%).
Recent trends in Gonococcus susceptibility to cephalosporins have the CDC concerned. While cephalosporin resistance remains rare, the proportion of Gonococcus isolates that have shown elevated minimum inhibitory concentrations to cephalosporins has increased.
Gonorrhea control depends in part on appropriate screening of individuals at risk (TABLE). Risk factors for gonorrhea include a history of previous gonorrhea infection, other sexually transmitted infections, new or multiple sexual partners, inconsistent condom use, sex work, and drug use. Risk factors for pregnant women are the same as for nonpregnant women. Prevalence of gonorrhea infection varies widely among communities and patient populations.
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 1
A decline in gonorrhea that began in the mid-1970s*
*The initiation of a national gonorrhea control program reaped immediate benefits that have continued through the present.
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates, United States, 1941-2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 2
Gonorrhea prevalence, 2010
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates by state, United States and outlying areas, 2010. Available at: http://www.cdc.gov/std/10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 3
Gonorrhea prevalence by age and sex, 2010
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates by age and sex, United States, 2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
Augment therapy, follow up thoroughly
Family physicians can assist with public health efforts to control gonorrhea and delay the development of cephalosporin resistance by screening for and detecting the infection, diagnosing those with symptoms, and treating according to newer recommendations. It’s also essential to report cases to local public health departments, assist with finding and treating sexual contacts of infected individuals, and immediately report suspected treatment failures.
A 2-drug regimen is imperative. The latest recommendation for treating gonorrhea is ceftriaxone 250 mg IM in a single dose and azithromycin 1 g orally in a single dose. Until 2010, the dose of ceftriaxone had been 125 mg. This dual drug regimen is recommended for several reasons: As with using multiple drugs to treat tuberculosis, it is hoped dual drug therapy will slow development of resistance to both cephalosporins and azithromycin; co-infection with C trachomatis remains a significant problem; and the combination may be more effective against pharyngeal gonorrhea, which is hard to detect.
Alternative regimens. Cefixime 400 mg orally as a single dose is an option in lieu of ceftriaxone, but is not preferred because of the higher number of reported failures of treatment with oral cephalosporins and less efficacy against pharyngeal disease.3 Other injectable cephalosporins are also an option, but less is known about their effectiveness in treating pharyngeal infection. Injectable options include ceftizoxime 500 mg IM, cefoxitin 2 g IM with probenecid 1 g orally, and cefotaxime 500 mg IM.
Regardless of the cephalosporin chosen, always administer azithromycin. If necessary, an alternative to azithromycin is doxycycline 100 mg orally twice a day. But doxycycline is not preferred because it has a multiple daily dosing requirement and higher levels of gonococcal resistance than is seen with azithromycin.
Necessary follow-up. Although routine testing for cure is not advocated for those treated with a recommended antibiotic regimen, a gonococcal culture and testing for antibiotic susceptibility should be done for any patient whose symptoms persist after treatment. Rapid tests using nucleic acid amplification are unsuitable for testing antibiotic susceptibility. The CDC does recommend retesting patients 3 months after treatment is completed because of a high prevalence of reinfection.3 If cephalosporin resistance becomes prevalent, routine tests of cure might become a recommended standard.
Report all patients with gonorrhea to the local public health department so that sexual contacts within the past 60 days can be notified, tested, and treated presumptively with the dual drug regimen. Recommend simultaneous treatment for all current sex partners, and discourage sexual intercourse until symptoms have resolved. Promptly report any patient with suspected treatment failure to the local health department, and consult the local or state health department for recommendations on subsequent treatment regimens.
The US Preventive Services Task Force (USPSTF) recommends routine screening for asymptomatic infection in women at risk, as per the details in the TABLE.4 While the USPSTF found insufficient evidence to recommend screening of high-risk men, physicians might still consider screening men who have sex with multiple male partners.
TABLE
USPSTF recommendations on screening for gonorrhea4
|
|
|
|
| USPSTF, US Preventive Services Task Force. *For more on the USPSTF’s grade definitions, see: http://www.uspreventiveservicestaskforce.org/uspstf/gradespre.htm#brec. |
Doing our best in the face of uncertainty
Although evidence is lacking that dual drug therapy will delay the progression of resistance, the strategy makes empirical sense. If gonorrhea develops resistance to cephalosporins, it will seriously challenge public health efforts to control this infection. Family physicians have an important role in controlling this sexually transmitted infection and helping to prevent drug resistance.
Public health efforts have decreased the incidence of gonorrhea over the past several decades, but this progress is threatened by emergent bacteria resistance to the few remaining antibiotics available to treat it.
Gonococcal resistance to penicillin and tetracycline began in the 1970s and was widespread by the 1980s. Resistance to fluoroquinolones developed during the last decade and led the Centers for Disease Control and Prevention (CDC) in 2007 to stop recommending this class of antibiotics for treatment of gonorrhea.1 (See “The decline of gonorrhea: A success story now threatened by antibiotic resistance”.)
Given the speed with which gonococci developed resistance to fluoroquinolones, the CDC sees as inevitable the eventual development of resistance to cephalosporins—the currently favored agents for gonorrhea.2 This is the main reason behind the new recommendations for treating all cases of gonorrhea with both a cephalosporin and azithromycin, whether or not co-infection with Chlamydia trachomatis is documented or suspected.3
The reported rate of gonorrhea rose steadily from the early 1960s until the mid-1970s, when it was close to 500 cases per 100,000. With implementation of the national gonorrhea control program, the annual rate began to fall, and by the mid-1990s it had declined by 74%. Between 1996 and 2006, the rate remained at about 115 cases per 100,000. Between 2006 and 2009, it decreased to the lowest rate since national reporting began, but increased 2.8% between 2009 and 2010 (FIGURE 1).
The highest rates of gonorrhea are in the South (FIGURE 2) and in women ages 15 to 24 and men ages 20 to 24 (FIGURE 3). Rates are highest among blacks (432.5 cases per 100,000), followed by American Indians/Alaska natives (105.7) and Hispanics (49.9). Between 2009 and 2010, gonorrhea rates increased among American Indians/Alaska natives (21.5%), Asians/Pacific Islanders (13.1%), Hispanics (11.9%), whites (9.0%), and blacks (0.3%).
Recent trends in Gonococcus susceptibility to cephalosporins have the CDC concerned. While cephalosporin resistance remains rare, the proportion of Gonococcus isolates that have shown elevated minimum inhibitory concentrations to cephalosporins has increased.
Gonorrhea control depends in part on appropriate screening of individuals at risk (TABLE). Risk factors for gonorrhea include a history of previous gonorrhea infection, other sexually transmitted infections, new or multiple sexual partners, inconsistent condom use, sex work, and drug use. Risk factors for pregnant women are the same as for nonpregnant women. Prevalence of gonorrhea infection varies widely among communities and patient populations.
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 1
A decline in gonorrhea that began in the mid-1970s*
*The initiation of a national gonorrhea control program reaped immediate benefits that have continued through the present.
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates, United States, 1941-2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 2
Gonorrhea prevalence, 2010
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates by state, United States and outlying areas, 2010. Available at: http://www.cdc.gov/std/10/surv2010.pdf. Accessed November 17, 2011.
FIGURE 3
Gonorrhea prevalence by age and sex, 2010
Source: Centers for Disease Control and Prevention. Sexually transmitted disease surveillance 2010. Gonorrhea—rates by age and sex, United States, 2010. Available at: http://www.cdc.gov/std/stats10/surv2010.pdf. Accessed November 17, 2011.
Augment therapy, follow up thoroughly
Family physicians can assist with public health efforts to control gonorrhea and delay the development of cephalosporin resistance by screening for and detecting the infection, diagnosing those with symptoms, and treating according to newer recommendations. It’s also essential to report cases to local public health departments, assist with finding and treating sexual contacts of infected individuals, and immediately report suspected treatment failures.
A 2-drug regimen is imperative. The latest recommendation for treating gonorrhea is ceftriaxone 250 mg IM in a single dose and azithromycin 1 g orally in a single dose. Until 2010, the dose of ceftriaxone had been 125 mg. This dual drug regimen is recommended for several reasons: As with using multiple drugs to treat tuberculosis, it is hoped dual drug therapy will slow development of resistance to both cephalosporins and azithromycin; co-infection with C trachomatis remains a significant problem; and the combination may be more effective against pharyngeal gonorrhea, which is hard to detect.
Alternative regimens. Cefixime 400 mg orally as a single dose is an option in lieu of ceftriaxone, but is not preferred because of the higher number of reported failures of treatment with oral cephalosporins and less efficacy against pharyngeal disease.3 Other injectable cephalosporins are also an option, but less is known about their effectiveness in treating pharyngeal infection. Injectable options include ceftizoxime 500 mg IM, cefoxitin 2 g IM with probenecid 1 g orally, and cefotaxime 500 mg IM.
Regardless of the cephalosporin chosen, always administer azithromycin. If necessary, an alternative to azithromycin is doxycycline 100 mg orally twice a day. But doxycycline is not preferred because it has a multiple daily dosing requirement and higher levels of gonococcal resistance than is seen with azithromycin.
Necessary follow-up. Although routine testing for cure is not advocated for those treated with a recommended antibiotic regimen, a gonococcal culture and testing for antibiotic susceptibility should be done for any patient whose symptoms persist after treatment. Rapid tests using nucleic acid amplification are unsuitable for testing antibiotic susceptibility. The CDC does recommend retesting patients 3 months after treatment is completed because of a high prevalence of reinfection.3 If cephalosporin resistance becomes prevalent, routine tests of cure might become a recommended standard.
Report all patients with gonorrhea to the local public health department so that sexual contacts within the past 60 days can be notified, tested, and treated presumptively with the dual drug regimen. Recommend simultaneous treatment for all current sex partners, and discourage sexual intercourse until symptoms have resolved. Promptly report any patient with suspected treatment failure to the local health department, and consult the local or state health department for recommendations on subsequent treatment regimens.
The US Preventive Services Task Force (USPSTF) recommends routine screening for asymptomatic infection in women at risk, as per the details in the TABLE.4 While the USPSTF found insufficient evidence to recommend screening of high-risk men, physicians might still consider screening men who have sex with multiple male partners.
TABLE
USPSTF recommendations on screening for gonorrhea4
|
|
|
|
| USPSTF, US Preventive Services Task Force. *For more on the USPSTF’s grade definitions, see: http://www.uspreventiveservicestaskforce.org/uspstf/gradespre.htm#brec. |
Doing our best in the face of uncertainty
Although evidence is lacking that dual drug therapy will delay the progression of resistance, the strategy makes empirical sense. If gonorrhea develops resistance to cephalosporins, it will seriously challenge public health efforts to control this infection. Family physicians have an important role in controlling this sexually transmitted infection and helping to prevent drug resistance.
1. Centers for Disease Control and Prevention. Update to CDC’s sexually transmitted disease treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR Morb Mortal Wkly Rep. 2007;56:332-336.
2. Centers for Disease Control and Prevention. Cephalosporin susceptibility among Neisseria gonorrhoeae isolates—United States 2000-2010. MMWR Morb Mortal Wkly Rep. 2011;60:873-877.
3. Workowski KA, Berman S. Centers for Disease Control and Prevention (CDC). Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep. 2010;59(RR-12):1-110.
4. US Preventive Services Task Force. Screening for gonorrhea. Available at: http://www.uspreventiveservicestaskforce.org/uspstf/uspsgono.htm. Accessed September 26, 2011.
1. Centers for Disease Control and Prevention. Update to CDC’s sexually transmitted disease treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR Morb Mortal Wkly Rep. 2007;56:332-336.
2. Centers for Disease Control and Prevention. Cephalosporin susceptibility among Neisseria gonorrhoeae isolates—United States 2000-2010. MMWR Morb Mortal Wkly Rep. 2011;60:873-877.
3. Workowski KA, Berman S. Centers for Disease Control and Prevention (CDC). Sexually transmitted diseases treatment guidelines, 2010. MMWR Recomm Rep. 2010;59(RR-12):1-110.
4. US Preventive Services Task Force. Screening for gonorrhea. Available at: http://www.uspreventiveservicestaskforce.org/uspstf/uspsgono.htm. Accessed September 26, 2011.
Detecting and treating delirium—key interventions you may be missing
• Nonpharmacologic interventions are the mainstay of treatment for delirium. B
• When medication is needed, atypical antipsychotics are as effective as typical antipsychotics for treating delirium in elderly patients, and have fewer side effects. B
• Benzodiazepines should be avoided in elderly patients with delirium that is not associated with alcohol withdrawal. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Mr. D, a 75-year-old patient with a history of hypertension and congestive heart failure, sustained a femoral neck fracture and was admitted to the hospital for surgery. He underwent open reduction and internal fixation and was doing well postoperatively, until Day 2—when his primary care physician made morning rounds and noted that Mr. D was somnolent. The nurse on duty assured the physician that Mr. D was fine and “was awake and alert earlier,” and attributed his somnolence to the oxycodone (10 mg) the patient was taking for pain. The physician ordered a reduction in dosage.
If Mr. D had been your patient, would you have considered other possible causes of his somnolence? Or do you think the physician’s action was sufficient?
Derived from Latin, the word delirium literally means “off the [ploughed] track.”1 Dozens of terms have been used to describe delirium, with acute confusion state, organic brain syndrome, acute brain syndrome, and toxic psychosis among them.
Delirium has been reported to occur in 15% to 30% of patients on general medical units,2 about 40% of postoperative patients, and up to 70% of terminally ill patients.3 The true prevalence is hard to determine, as up to 66% of cases may be missed.4
Delirium is being diagnosed more frequently, however—a likely result of a growing geriatric population, increased longevity, and greater awareness of the condition. Each year, an estimated 2.3 million US residents are affected, leading to prolonged hospitalization; poor functional outcomes; the development or worsening of dementia; increased nursing home placement; and a significant burden for families and the US health care system.5
Delirium is also associated with an increase in mortality.6,7 The mortality rate among hospitalized patients who develop delirium is reported to be 18%, rising to an estimated 47% within the first 3 months after discharge.6 Greater awareness of risk factors, rapid recognition of signs and symptoms of delirium, and early intervention—detailed in the text and tables that follow—will lead to better outcomes.
Assessing risk, evaluating mental status
In addition to advanced age, risk factors for delirium (TABLE 1)8-14 include alcohol use, brain dysfunction, comorbidities, hypertension, malignancy, anticholinergic medications, anemia, metabolic abnormalities, and male sex. In patients who, like Mr. D, have numerous risk factors, early—and frequent—evaluation of mental status is needed. One way to do this is to treat mental status as a vital sign, to be included in the assessment of every elderly patient.15
The Confusion Assessment Method, a quick and easy-to-use delirium screening tool (TABLE 2), has a sensitivity of 94% to 100% and a specificity of 90% to 95%.16,17 There are a number of other screening tools, including the widely used Mini-Mental State Exam (MMSE), as well as the Delirium Rating Scale, Delirium Symptom Interview, and Delirium Severity Scale.
TABLE 1
Risk factors for delirium8-14
Advanced age Alcohol use Brain dysfunction (dementia, epilepsy) Hypertension Male sex Malignancy Medications (mainly anticholinergic) Metabolic abnormalities:
Old age Preoperative anemia Preoperative metabolic abnormalities |
| BUN, blood urea nitrogen; Cr, creatinine; Na, sodium. |
TABLE 2
Screening for delirium: The Confusion Assessment Method*16,17
| Criteria | Evidence Yes to questions 1, 2, and 3 plus 4 or 5 (or both) suggests a delirium diagnosis |
|---|---|
| 1. Acute onset | Is there evidence of an acute change in mental status from the patient’s baseline? |
| 2. Fluctuating course | Did the abnormal behavior fluctuate during the day—ie, tend to come and go or increase and decrease in severity? |
| 3. Inattention | Did the patient have difficulty focusing attention, eg, being easily distractible or having difficulty keeping track of what was being said? |
| PLUS | |
| 4. Disorganized thinking | Was the patient’s thinking disorganized or incoherent, such as rambling or irrelevant conversation, unclear or illogical flow of ideas, or unpredictable switching from subject to subject? |
| 5. Altered level of consciousness | Would you rate the patient’s level of consciousness as (any of the following): – Vigilant (hyperalert) – Lethargic (drowsy, easily aroused) – Stupor (difficult to arouse) – Coma (unarousable) |
| *CAM shortened version worksheet. Adapted from: Inouye SK et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113:941-948; Inouye SK. Confusion Assessment Method (CAM): Training Manual and Coding Guide. Copyright 2003, Hospital Elder Life Program, LLC. | |
Arriving at a delirium diagnosis
The clinical presentation of delirium is characterized by acute—and reversible—impairment of cognition, attention, orientation, and memory, and disruption of the normal sleep/wake cycle. The Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) criteria for a delirium diagnosis include all of the following:
- disturbance of consciousness, with a reduced ability to focus, sustain, or shift attention
- change in cognition, or a perceptual disturbance, that is not accounted for by a preexisting or developing dementia
- rapid onset of cognitive impairment, with fluctuation likely during the course of the day
- evidence from the history, physical exam, or laboratory findings that the disturbed consciousness is a direct physiological consequence of a general medical condition.17
There are 3 basic types of delirium, each associated with a different psychomotor disturbance.
- Hyperactive delirium—the least common—is characterized by restlessness and agitation, and is therefore the easiest to diagnose.
- Hypoactive delirium is characterized by psychomotor retardation and hypoalertness. It is often misdiagnosed as depression, and has the poorest prognosis.
- Mixed delirium—the most common—is characterized by symptoms that fluctuate between hyper- and hypoactivity.18
CASE By lunchtime, Mr. D had awakened; however, he needed help with his meal. After eating, he slept for the rest of the day. At night, a nurse paged the resident to report that the patient’s blood pressure was 82/60 mm Hg and his heart rate was 115. The physician ordered an intravenous fluid bolus, which corrected the patient’s hypotension, but only temporarily.
The fluctuating nature of delirium—most notably, in patients’ level of alertness—is helpful in establishing a diagnosis. The history and physical exam are the gold standard tools, both for diagnosing delirium and identifying the underlying cause (TABLE 3).19,20 A review of the patient’s medications should be a key component of the medical history, as drugs—particularly those with anticholinergic properties—are often associated with delirium. Environmental shifts, including hospitalization and a disruption of the normal sleep/wake cycle, endocrine disorders, infection, and nutritional deficiencies are also potential causes of delirium, among others.
If history and physical exam fail to identify the underlying cause, laboratory testing, including complete blood count, complete metabolic profile, and urinalysis, should be done. Brain imaging is usually not needed for individuals with symptoms of delirium, but computed tomography (CT) may be indicated if a patient’s condition continues to deteriorate while the underlying cause remains unidentified.21 Electroencephalography (EEG) may be used to confirm a delirium diagnosis that’s uncertain, in a patient with underlying dementia, for instance. (In more than 16% of cases of delirium, the cause is unknown.22)
The most common structural abnormalities found in patients with delirium are brain atrophy and increased white matter lesions, as well as basal ganglia lesions.23 Single-photon emission CT (SPECT) shows a reduction of regional cerebral perfusion by 50%,24 while EEG shows slowing of the posterior dominant rhythm and increased generalized slow-wave activity.25
TABLE 3
A DELIRIUM mnemonic to get to the heart of the problem19,20
| Cause | Comment |
|---|---|
| Drugs | Drug classes: Anesthesia, anticholinergics, anticonvulsants, antiemetics, antihistamines, antihypertensives, antimicrobials, antipsychotics, benzodiazepines, corticosteroids, hypnotics, H2 blockers, muscle relaxants, NSAIDs, opioids, SSRIs, tricyclic antidepressants Drugs: digoxin, levodopa, lithium, theophylline OTCs: henbane, Jimson weed, mandrake, Atropa belladonna extract |
| Environmental | Change of environment, sensory deprivation, sleep deprivation |
| Endocrine | Hyperparathyroidism, hyper-/hypothyroidism |
| Low perfusion | MI, pulmonary embolism, CVA |
| Infection | Pneumonia, sepsis, systemic infection, UTI |
| Retention | Fecal impaction, urinary retention |
| Intoxication | Alcohol, illegal drugs/drug overdose |
| Undernutrition | Malnutrition, thiamin deficiency, vitamin B12 deficiency |
| Metabolic | Acid-base disturbances, fluid and electrolyte abnormalities, hepatic or uremic encephalopathy, hypercarbia, hyper-/hypoglycemia, hyperosmolality, hypoxia |
| Subdural | History of falls |
| CVA, cerebrovascular accident; MI, myocardial infarction; NSAIDs, nonsteroidal anti-inflammatory drugs; OTCs, over-the-counter agents; SSRIs, selective serotonin reuptake inhibitors; UTI, urinary tract infection. | |
Treating (or preventing) delirium: Start with these steps
Nonpharmacologic interventions are the mainstay of treatment for patients with delirium, and may also help to prevent the development of delirium in patients at risk. One key measure is to correct, or avoid, disruptions in the patient’s normal sleep/wake cycle—eg, restoring circadian rhythm by avoiding,
to the extent possible, awakening the patient at night for medication or vital signs. Preventing sensory deprivation, by ensuring that the patient’s eyeglasses and hearing aid are nearby and that there is a clock and calendar nearby and adequate light, is also helpful. Other key interventions (TABLE 4)26-28 include:
- limiting medications associated with delirium (and eliminating any nonessential medication)
- improving nutrition and ambulation
- correcting electrolyte and fluid disturbances
- treating infection
- involving family members in patient care
- ensuring that patients receive adequate pain management
- avoiding transfers (if the patient is hospitalized) and trying to secure a single room.
Several studies have evaluated the effectiveness of nonpharmacologic interventions in preventing or lowering the incidence of delirium. A large multicomponent delirium prevention study of patients >70 years on general medical units focused on managing risk factors. The interventions studied included (1) avoidance of sensory deprivation, (2) early mobilization, (3) treating dehydration, (4) implementing noise reduction strategies and sleep enhancement programs, and (5) avoiding the use of sleep medications. These interventions proved to be effective not only in lowering the incidence of delirium, but in shortening the duration of delirium in affected patients (NNT=20).27
One study found that proactively using a geriatric consultation model (ie, implementing standardized protocols for the management of 6 risk factors) for elderly hospitalized patients led to a reduction in the incidence of delirium by more than a third.26 Admission to a specialized geriatric unit is associated with a lower incidence of delirium compared with being hospitalized on a general medical unit.29
Reducing the incidence of postoperative delirium. Bright light therapy (a light intensity of 5000 lux with a distance from the light source of 100 cm), implemented postoperatively, may play a role in reducing the incidence of delirium, research suggests.30 Music may be helpful, as well. An RCT involving patients (>65 years) undergoing elective knee or hip surgery found that those who listened to classical music postoperatively had a lower incidence of delirium.31 Similarly, playing music in nursing homes has been shown to decrease aggressive behavior and agitation.32
TABLE 4
Helpful interventions in the hospital or at home26-28
|
When medication is needed, proceed with caution
None of the medications currently used to treat delirium are approved by the US Food and Drug Administration for this indication, and many of them have substantial side effects. Nonetheless, palliative or symptomatic treatment requires some form of sedation for agitated patients with delirium. Thus, it is necessary to strike a balance in order to manage the symptoms of delirium and avoid potential side effects (primarily, sedation). Overly sedating patients can confuse the clinical picture of delirium and make it difficult to differentiate between ongoing delirium and medication side effects. Medication should be started at a low, but frequent, dose to achieve an effective therapeutic level, after which a lower maintenance dose can be used until the cause of delirium is resolved.
Antipsychotics are the cornerstone of drug treatment
Haloperidol has traditionally been used to treat delirium33 and has proven effectiveness. However, it is associated with increased risk of extrapyramidal manifestations compared with atypical antipsychotics.
Atypical antipsychotics (olanzapine, risperidone, quetiapine) are increasingly being used to treat delirium because they have fewer extrapyramidal side effects.34 With the exception of olanzapine (available in intramuscular and oral disintegrating form), atypical antipsychotics are available only in oral form, which may limit their usefulness as a treatment for agitated, delirious patients.
Risperidone (at a dose ranging from 0.25 to 1 mg/d) and olanzapine (1.25 to 2.5 mg/d) have shown similar efficacy to haloperidol (0.75 to 1.5 mg/d) in both the prevention and treatment of delirium, but with fewer extrapyramidal side effects.35-39 Quetiapine, a second-generation antipsychotic, is widely used to treat inpatient delirium, although there are no large RCTs comparing it with placebo. One pilot study and another open-label trial found the drug to be beneficial for patients with delirium, with fewer extrapyramidal side effects than haloperidol.40,41
Do a risk-benefit analysis. The use of antipsychotics in elderly patients with delirium has been associated with increased morbidity and mortality. The incidence of stroke and death were higher for community-dwelling patients (NNH=100) and patients in long-term care (N=67) who received typical or atypical antipsychotics for 6 months compared with that of patients who did not receive any antipsychotics.42,43 Thus, a risk-benefit analysis should be done before prescribing antipsychotics for elderly patients. Both typical and atypical antipsychotics carry black box warnings of increased mortality rates in the elderly.
Other drugs for delirium? More research is needed
Cholinesterase inhibitors. Procholinergic agents would be expected to be helpful in treating delirium, as cholinergic deficiency has been implicated as a predisposing factor for delirium and medications with anticholinergic effects have been shown to induce delirium. However, several studies of cholinesterase inhibitors have not found this to be the case.44-47
Benzodiazepines. There is no evidence to support the use of benzodiazepines in the treatment of delirium, except when the delirium is related to alcohol withdrawal.48 When indicated, the use of a short-acting benzodiazepine such as lorazepam is preferred for elderly patients (vs long-acting agents like diazepam) because of its shorter half-life and better side effect profile.2 Drowsiness, ataxia, and disinhibition are common side effects of benzodiazepines.
Gabapentin. A pilot study conducted to assess the efficacy of gabapentin (900 mg/d) for the prevention of postoperative delirium found a significantly lower incidence of delirium among patients who received gabapentin compared with placebo. This may be associated with gabapentin’s opioid-sparing effect.49 Larger studies are needed to recommend for or against the use of gabapentin in patients receiving opiates.
Further study of the pathophysiology of delirium is needed, as well, to increase our ability to prevent and treat it.
CASE After receiving the IV fluid bolus, Mr. D became increasingly short of breath and required more oxygen to keep his oxygen saturation in the 90s. Labs were ordered during morning rounds, and the patient was found to have urosepsis. He was admitted to the ICU in septic shock, and was intubated and died several days later.
In retrospect, it was determined that Mr. D had developed hypoactive delirium brought on by the infection—and that his somnolence on the second postoperative day was not a sign of overmedication. Had this been recognized early on through the use of an appropriate screening tool, the outcome would likely have been more favorable.
CORRESPONDENCE Abdulraouf Ghandour, MD, Green Meadows Clinic University Physicians, 3217 Providence Road, Columbia, MO 65203; [email protected]
1. Casselman WG. Dictionary of Medical Derivations. The Real Meaning of Medical Terms. New York, NY: Informa Healthcare; 1998.
2. Kiely DK, Bergmann MA, Murphy KM, et al. Delirium among newly admitted postacute facility patients, prevalence, symptoms, and severity. J Gerontol Biol Sci Med Sci. 2003;58:M441-M445.
3. Inouye SK, Charpentier PA. Precipitating factors for delirium in hospitalized elderly persons. Predictive model and interrelationship with baseline vulnerability. JAMA. 1996;275:852-857.
4. Inouye SK. The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med. 1994;97:278-288.
5. Pompei P, Foreman M, Rudberg M, et al. Delirium in hospitalized older persons: outcomes and predictors. J Am Geriatr Soc. 1994;42:809-815.
6. Kolbeinsson H, Jonsson A. Delirium and dementia in acute medical admissions of elderly patients in Iceland. Acta Psychiatr Scand. 1993;87:123-127.
7. Cole MG, Primeau FJ. Prognosis of delirium in elderly hospital patients. CMAJ. 1993;149:41-46.
8. Rahkonen T, Eloniemi-Sulkava U, Halonen P, et al. Delirium in the non-demented oldest old in the general population: risk factors and prognosis. Int J Geriatr Psychiatry. 2001;16:415-421.
9. Edlund A, Lundstrom M, Brannstrom B, et al. Delirium before and after operation for femoral neck fracture. J Am Geriatr Soc. 2001;49:1335-1340.
10. Andersson EM, Gustafson L, Hallberg IR. Acute confusional state in elderly orthopaedic patients: factors of importance for detection in nursing care. Int J Geriatr Psychiatry. 2001;16:7-17.
11. Inouye SK, Viscoli CM, Horwitz RI, et al. A predictive model for delirium in hospitalized elderly medical patients based on admission characteristics. Ann Intern Med. 1993;119:474-481.
12. Marcantonio ER, Juarez G, Goldman L, et al. The relationship of postoperative delirium with psychoactive medications. JAMA. 1994;272:1518-1522.
13. Marcantonio ER, Goldman L, Orav EJ, et al. The association of intraoperative factors with the development of postoperative delirium. Am J Med. 1998;105:380-384.
14. Tune L, Carr S, Hoag E, et al. Anticholinergic effects of drugs commonly prescribed for the elderly: potential means for assessing risk of delirium. Am J Psychiatry. 1992;149:1393-1394.
15. Flaherty JH, Shay K, Weir C, et al. The development of a mental status vital sign for use across the spectrum of care . J Am Med Dir Assoc. 2009;10:379-380.
16. Inouye SK, Van Dyck CH, Alessi CA, et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113:941-948.
17. Inouye SK. Confusion Assessment Method (CAM): Training Manual and Coding Guide. New Haven, Conn: Yale University School of Medicine; 2003.
18. Halter J, Ouslander J, Tinetti M, et al. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York, NY: McGraw-Hill; 2009;648-658.
19. Eriksson S. Social and environmental contributants to delirium in the elderly. Dement Geriatr Cogn Disord. 1999;10:350-352.
20. Francis J, Martin D, Kapoor WN. A prospective study of delirium in hospitalized elderly. JAMA. 1990;263:1097-1101.
21. Francis J, Hilko EM, Kapoor WN. Acute mental change: when are head scans needed? Clin Res. 1991;39:103.-
22. Rudberg MA, Pompei P, Foreman MD, et al. The natural history of delirium in older hospitalized patients: a syndrome of heterogeneity. Age Ageing. 1997;26:169-174.
23. Soiza RL, Sharma V, Ferguson K, et al. Neuroimaging studies of delirium: a systematic review. J Psychosom Res. 2008;65:239-248.
24. Fong TG, Bogardus ST Jr, Daftary A, et al. Cerebral perfusion changes in older delirious patients using 99mTc HMPAO SPECT. J Gerontol A Biol Sci Med Sci. 2006;61:1294-1299.
25. Jacobson SA, Leuchter AF, Walter DO. Conventional and quantitative EEG in the diagnosis of delirium among the elderly. J Neurol Neurosurg Psychiatry. 1993;56:153-158.
26. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2001;49:516-522.
27. Inouye SK, Bogardus ST Jr, Charpentier PA, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. N Engl J Med. 1999;340:669-676.
28. Weber JB, Coverdale JH, Kunik ME. Delirium: current trends in prevention and treatment. Intern Med J. 2004;34:115-121.
29. Bo M, Martini B, Ruatta C, et al. Geriatric ward hospitalization reduced incidence delirium among older medical inpatients. Am J Geriatr Psychiatry. 2009;17:760-768.
30. Taguchi T, Yano M, Kido Y. Influence of bright light therapy on postoperative patients: a pilot study. Intensive Crit Care Nurs. 2007;23:289-297.
31. McCaffrey R, Locsin R. The effect of music listening on acute confusion and delirium in elders undergoing elective hip and knee surgery. J Clin Nurs. 2004;13:91-96.
32. Remington R. Calming music and hand massage with agitated elderly. Nurs Res. 2004;51:317-323.
33. Seitz DP, Gill SS, van Zyl LT. Antipsychotics in the treatment of delirium: a systematic review. J Clin Psychiatry. 2007;68:11-21.
34. Schwartz T, Masand PS. The role of atypical antipsychotics in the treatment of delirium. Psychosomatics. 2002;43:171-174.
35. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594.-
36. Hu H, Deng W, Yang H. A prospective random control study comparison of olanzapine and haloperidol in senile delirium. Chongqing Med J. 2004;8:1234-1237.
37. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45:297-301.
38. Kim SW, Yoo JA, Lee SY, et al. Risperidone versus olanzapine for the treatment of delirium. Hum Psychopharmacol. 2010;25:298-302.
39. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35:714-719.
40. Maneeton B, Maneeton N, Srisurapanont M. An open-label study of quetiapine for delirium. J Med Assoc Thai. 2007;90:2158-2163.
41. Devlin JW, Roberts RJ, Fong JJ, et al. Efficacy and safety of quetiapine in critically ill patients with delirium: a prospective, multicenter, randomized, double-blind, placebo-controlled pilot study. Crit Care Med. 2010;38:419-427.
42. Gill SS, Bronskill SE, Normand SL, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146:775-786.
43. Wang PS, Schneeweiss S, Avorn J, et al. Death in elderly users of conventional vs. atypical antipsychotic medications. N Engl J Med. 2005;353:2335-2341.
44. Liptzin B, Laki A, Garb JL, et al. Donepezil in the prevention and treatment of post-surgical delirium. Am J Geriatr Psychiatry. 2005;13:1100-1106.
45. Sampson EL, Raven PR, Ndhlovu PN, et al. A randomized, double-blind, placebo-controlled trial of donepezil hydrochloride (Aricept) for reducing the incidence of postoperative delirium after elective total hip replacement. Int J Geriatr Psychiatry. 2007;22:343-349.
46. Gamberini M, Bolliger D, Lurati Buse GA, et al. Rivastigmine for the prevention of postoperative delirium in elderly patients undergoing elective cardiac surgery—a randomized controlled trial. Crit Care Med. 2009;37:1762-1768.
47. Overshott R, Vernon M, Morris J, et al. Rivastigmine in the treatment of delirium in older people: a pilot study. Int Psychogeriatr. 2010;22:812-818.
48. Lonergan E, Luxenberg J, Areosa Sastre A. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;(4):CD006379.-
49. Leung JM, Sands LP, Rico M, et al. Pilot clinical trial of gabapentin to decrease postoperative delirium in older patients. Neurology. 2006;67:1251-1253.
• Nonpharmacologic interventions are the mainstay of treatment for delirium. B
• When medication is needed, atypical antipsychotics are as effective as typical antipsychotics for treating delirium in elderly patients, and have fewer side effects. B
• Benzodiazepines should be avoided in elderly patients with delirium that is not associated with alcohol withdrawal. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Mr. D, a 75-year-old patient with a history of hypertension and congestive heart failure, sustained a femoral neck fracture and was admitted to the hospital for surgery. He underwent open reduction and internal fixation and was doing well postoperatively, until Day 2—when his primary care physician made morning rounds and noted that Mr. D was somnolent. The nurse on duty assured the physician that Mr. D was fine and “was awake and alert earlier,” and attributed his somnolence to the oxycodone (10 mg) the patient was taking for pain. The physician ordered a reduction in dosage.
If Mr. D had been your patient, would you have considered other possible causes of his somnolence? Or do you think the physician’s action was sufficient?
Derived from Latin, the word delirium literally means “off the [ploughed] track.”1 Dozens of terms have been used to describe delirium, with acute confusion state, organic brain syndrome, acute brain syndrome, and toxic psychosis among them.
Delirium has been reported to occur in 15% to 30% of patients on general medical units,2 about 40% of postoperative patients, and up to 70% of terminally ill patients.3 The true prevalence is hard to determine, as up to 66% of cases may be missed.4
Delirium is being diagnosed more frequently, however—a likely result of a growing geriatric population, increased longevity, and greater awareness of the condition. Each year, an estimated 2.3 million US residents are affected, leading to prolonged hospitalization; poor functional outcomes; the development or worsening of dementia; increased nursing home placement; and a significant burden for families and the US health care system.5
Delirium is also associated with an increase in mortality.6,7 The mortality rate among hospitalized patients who develop delirium is reported to be 18%, rising to an estimated 47% within the first 3 months after discharge.6 Greater awareness of risk factors, rapid recognition of signs and symptoms of delirium, and early intervention—detailed in the text and tables that follow—will lead to better outcomes.
Assessing risk, evaluating mental status
In addition to advanced age, risk factors for delirium (TABLE 1)8-14 include alcohol use, brain dysfunction, comorbidities, hypertension, malignancy, anticholinergic medications, anemia, metabolic abnormalities, and male sex. In patients who, like Mr. D, have numerous risk factors, early—and frequent—evaluation of mental status is needed. One way to do this is to treat mental status as a vital sign, to be included in the assessment of every elderly patient.15
The Confusion Assessment Method, a quick and easy-to-use delirium screening tool (TABLE 2), has a sensitivity of 94% to 100% and a specificity of 90% to 95%.16,17 There are a number of other screening tools, including the widely used Mini-Mental State Exam (MMSE), as well as the Delirium Rating Scale, Delirium Symptom Interview, and Delirium Severity Scale.
TABLE 1
Risk factors for delirium8-14
Advanced age Alcohol use Brain dysfunction (dementia, epilepsy) Hypertension Male sex Malignancy Medications (mainly anticholinergic) Metabolic abnormalities:
Old age Preoperative anemia Preoperative metabolic abnormalities |
| BUN, blood urea nitrogen; Cr, creatinine; Na, sodium. |
TABLE 2
Screening for delirium: The Confusion Assessment Method*16,17
| Criteria | Evidence Yes to questions 1, 2, and 3 plus 4 or 5 (or both) suggests a delirium diagnosis |
|---|---|
| 1. Acute onset | Is there evidence of an acute change in mental status from the patient’s baseline? |
| 2. Fluctuating course | Did the abnormal behavior fluctuate during the day—ie, tend to come and go or increase and decrease in severity? |
| 3. Inattention | Did the patient have difficulty focusing attention, eg, being easily distractible or having difficulty keeping track of what was being said? |
| PLUS | |
| 4. Disorganized thinking | Was the patient’s thinking disorganized or incoherent, such as rambling or irrelevant conversation, unclear or illogical flow of ideas, or unpredictable switching from subject to subject? |
| 5. Altered level of consciousness | Would you rate the patient’s level of consciousness as (any of the following): – Vigilant (hyperalert) – Lethargic (drowsy, easily aroused) – Stupor (difficult to arouse) – Coma (unarousable) |
| *CAM shortened version worksheet. Adapted from: Inouye SK et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113:941-948; Inouye SK. Confusion Assessment Method (CAM): Training Manual and Coding Guide. Copyright 2003, Hospital Elder Life Program, LLC. | |
Arriving at a delirium diagnosis
The clinical presentation of delirium is characterized by acute—and reversible—impairment of cognition, attention, orientation, and memory, and disruption of the normal sleep/wake cycle. The Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) criteria for a delirium diagnosis include all of the following:
- disturbance of consciousness, with a reduced ability to focus, sustain, or shift attention
- change in cognition, or a perceptual disturbance, that is not accounted for by a preexisting or developing dementia
- rapid onset of cognitive impairment, with fluctuation likely during the course of the day
- evidence from the history, physical exam, or laboratory findings that the disturbed consciousness is a direct physiological consequence of a general medical condition.17
There are 3 basic types of delirium, each associated with a different psychomotor disturbance.
- Hyperactive delirium—the least common—is characterized by restlessness and agitation, and is therefore the easiest to diagnose.
- Hypoactive delirium is characterized by psychomotor retardation and hypoalertness. It is often misdiagnosed as depression, and has the poorest prognosis.
- Mixed delirium—the most common—is characterized by symptoms that fluctuate between hyper- and hypoactivity.18
CASE By lunchtime, Mr. D had awakened; however, he needed help with his meal. After eating, he slept for the rest of the day. At night, a nurse paged the resident to report that the patient’s blood pressure was 82/60 mm Hg and his heart rate was 115. The physician ordered an intravenous fluid bolus, which corrected the patient’s hypotension, but only temporarily.
The fluctuating nature of delirium—most notably, in patients’ level of alertness—is helpful in establishing a diagnosis. The history and physical exam are the gold standard tools, both for diagnosing delirium and identifying the underlying cause (TABLE 3).19,20 A review of the patient’s medications should be a key component of the medical history, as drugs—particularly those with anticholinergic properties—are often associated with delirium. Environmental shifts, including hospitalization and a disruption of the normal sleep/wake cycle, endocrine disorders, infection, and nutritional deficiencies are also potential causes of delirium, among others.
If history and physical exam fail to identify the underlying cause, laboratory testing, including complete blood count, complete metabolic profile, and urinalysis, should be done. Brain imaging is usually not needed for individuals with symptoms of delirium, but computed tomography (CT) may be indicated if a patient’s condition continues to deteriorate while the underlying cause remains unidentified.21 Electroencephalography (EEG) may be used to confirm a delirium diagnosis that’s uncertain, in a patient with underlying dementia, for instance. (In more than 16% of cases of delirium, the cause is unknown.22)
The most common structural abnormalities found in patients with delirium are brain atrophy and increased white matter lesions, as well as basal ganglia lesions.23 Single-photon emission CT (SPECT) shows a reduction of regional cerebral perfusion by 50%,24 while EEG shows slowing of the posterior dominant rhythm and increased generalized slow-wave activity.25
TABLE 3
A DELIRIUM mnemonic to get to the heart of the problem19,20
| Cause | Comment |
|---|---|
| Drugs | Drug classes: Anesthesia, anticholinergics, anticonvulsants, antiemetics, antihistamines, antihypertensives, antimicrobials, antipsychotics, benzodiazepines, corticosteroids, hypnotics, H2 blockers, muscle relaxants, NSAIDs, opioids, SSRIs, tricyclic antidepressants Drugs: digoxin, levodopa, lithium, theophylline OTCs: henbane, Jimson weed, mandrake, Atropa belladonna extract |
| Environmental | Change of environment, sensory deprivation, sleep deprivation |
| Endocrine | Hyperparathyroidism, hyper-/hypothyroidism |
| Low perfusion | MI, pulmonary embolism, CVA |
| Infection | Pneumonia, sepsis, systemic infection, UTI |
| Retention | Fecal impaction, urinary retention |
| Intoxication | Alcohol, illegal drugs/drug overdose |
| Undernutrition | Malnutrition, thiamin deficiency, vitamin B12 deficiency |
| Metabolic | Acid-base disturbances, fluid and electrolyte abnormalities, hepatic or uremic encephalopathy, hypercarbia, hyper-/hypoglycemia, hyperosmolality, hypoxia |
| Subdural | History of falls |
| CVA, cerebrovascular accident; MI, myocardial infarction; NSAIDs, nonsteroidal anti-inflammatory drugs; OTCs, over-the-counter agents; SSRIs, selective serotonin reuptake inhibitors; UTI, urinary tract infection. | |
Treating (or preventing) delirium: Start with these steps
Nonpharmacologic interventions are the mainstay of treatment for patients with delirium, and may also help to prevent the development of delirium in patients at risk. One key measure is to correct, or avoid, disruptions in the patient’s normal sleep/wake cycle—eg, restoring circadian rhythm by avoiding,
to the extent possible, awakening the patient at night for medication or vital signs. Preventing sensory deprivation, by ensuring that the patient’s eyeglasses and hearing aid are nearby and that there is a clock and calendar nearby and adequate light, is also helpful. Other key interventions (TABLE 4)26-28 include:
- limiting medications associated with delirium (and eliminating any nonessential medication)
- improving nutrition and ambulation
- correcting electrolyte and fluid disturbances
- treating infection
- involving family members in patient care
- ensuring that patients receive adequate pain management
- avoiding transfers (if the patient is hospitalized) and trying to secure a single room.
Several studies have evaluated the effectiveness of nonpharmacologic interventions in preventing or lowering the incidence of delirium. A large multicomponent delirium prevention study of patients >70 years on general medical units focused on managing risk factors. The interventions studied included (1) avoidance of sensory deprivation, (2) early mobilization, (3) treating dehydration, (4) implementing noise reduction strategies and sleep enhancement programs, and (5) avoiding the use of sleep medications. These interventions proved to be effective not only in lowering the incidence of delirium, but in shortening the duration of delirium in affected patients (NNT=20).27
One study found that proactively using a geriatric consultation model (ie, implementing standardized protocols for the management of 6 risk factors) for elderly hospitalized patients led to a reduction in the incidence of delirium by more than a third.26 Admission to a specialized geriatric unit is associated with a lower incidence of delirium compared with being hospitalized on a general medical unit.29
Reducing the incidence of postoperative delirium. Bright light therapy (a light intensity of 5000 lux with a distance from the light source of 100 cm), implemented postoperatively, may play a role in reducing the incidence of delirium, research suggests.30 Music may be helpful, as well. An RCT involving patients (>65 years) undergoing elective knee or hip surgery found that those who listened to classical music postoperatively had a lower incidence of delirium.31 Similarly, playing music in nursing homes has been shown to decrease aggressive behavior and agitation.32
TABLE 4
Helpful interventions in the hospital or at home26-28
|
When medication is needed, proceed with caution
None of the medications currently used to treat delirium are approved by the US Food and Drug Administration for this indication, and many of them have substantial side effects. Nonetheless, palliative or symptomatic treatment requires some form of sedation for agitated patients with delirium. Thus, it is necessary to strike a balance in order to manage the symptoms of delirium and avoid potential side effects (primarily, sedation). Overly sedating patients can confuse the clinical picture of delirium and make it difficult to differentiate between ongoing delirium and medication side effects. Medication should be started at a low, but frequent, dose to achieve an effective therapeutic level, after which a lower maintenance dose can be used until the cause of delirium is resolved.
Antipsychotics are the cornerstone of drug treatment
Haloperidol has traditionally been used to treat delirium33 and has proven effectiveness. However, it is associated with increased risk of extrapyramidal manifestations compared with atypical antipsychotics.
Atypical antipsychotics (olanzapine, risperidone, quetiapine) are increasingly being used to treat delirium because they have fewer extrapyramidal side effects.34 With the exception of olanzapine (available in intramuscular and oral disintegrating form), atypical antipsychotics are available only in oral form, which may limit their usefulness as a treatment for agitated, delirious patients.
Risperidone (at a dose ranging from 0.25 to 1 mg/d) and olanzapine (1.25 to 2.5 mg/d) have shown similar efficacy to haloperidol (0.75 to 1.5 mg/d) in both the prevention and treatment of delirium, but with fewer extrapyramidal side effects.35-39 Quetiapine, a second-generation antipsychotic, is widely used to treat inpatient delirium, although there are no large RCTs comparing it with placebo. One pilot study and another open-label trial found the drug to be beneficial for patients with delirium, with fewer extrapyramidal side effects than haloperidol.40,41
Do a risk-benefit analysis. The use of antipsychotics in elderly patients with delirium has been associated with increased morbidity and mortality. The incidence of stroke and death were higher for community-dwelling patients (NNH=100) and patients in long-term care (N=67) who received typical or atypical antipsychotics for 6 months compared with that of patients who did not receive any antipsychotics.42,43 Thus, a risk-benefit analysis should be done before prescribing antipsychotics for elderly patients. Both typical and atypical antipsychotics carry black box warnings of increased mortality rates in the elderly.
Other drugs for delirium? More research is needed
Cholinesterase inhibitors. Procholinergic agents would be expected to be helpful in treating delirium, as cholinergic deficiency has been implicated as a predisposing factor for delirium and medications with anticholinergic effects have been shown to induce delirium. However, several studies of cholinesterase inhibitors have not found this to be the case.44-47
Benzodiazepines. There is no evidence to support the use of benzodiazepines in the treatment of delirium, except when the delirium is related to alcohol withdrawal.48 When indicated, the use of a short-acting benzodiazepine such as lorazepam is preferred for elderly patients (vs long-acting agents like diazepam) because of its shorter half-life and better side effect profile.2 Drowsiness, ataxia, and disinhibition are common side effects of benzodiazepines.
Gabapentin. A pilot study conducted to assess the efficacy of gabapentin (900 mg/d) for the prevention of postoperative delirium found a significantly lower incidence of delirium among patients who received gabapentin compared with placebo. This may be associated with gabapentin’s opioid-sparing effect.49 Larger studies are needed to recommend for or against the use of gabapentin in patients receiving opiates.
Further study of the pathophysiology of delirium is needed, as well, to increase our ability to prevent and treat it.
CASE After receiving the IV fluid bolus, Mr. D became increasingly short of breath and required more oxygen to keep his oxygen saturation in the 90s. Labs were ordered during morning rounds, and the patient was found to have urosepsis. He was admitted to the ICU in septic shock, and was intubated and died several days later.
In retrospect, it was determined that Mr. D had developed hypoactive delirium brought on by the infection—and that his somnolence on the second postoperative day was not a sign of overmedication. Had this been recognized early on through the use of an appropriate screening tool, the outcome would likely have been more favorable.
CORRESPONDENCE Abdulraouf Ghandour, MD, Green Meadows Clinic University Physicians, 3217 Providence Road, Columbia, MO 65203; [email protected]
• Nonpharmacologic interventions are the mainstay of treatment for delirium. B
• When medication is needed, atypical antipsychotics are as effective as typical antipsychotics for treating delirium in elderly patients, and have fewer side effects. B
• Benzodiazepines should be avoided in elderly patients with delirium that is not associated with alcohol withdrawal. A
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
CASE Mr. D, a 75-year-old patient with a history of hypertension and congestive heart failure, sustained a femoral neck fracture and was admitted to the hospital for surgery. He underwent open reduction and internal fixation and was doing well postoperatively, until Day 2—when his primary care physician made morning rounds and noted that Mr. D was somnolent. The nurse on duty assured the physician that Mr. D was fine and “was awake and alert earlier,” and attributed his somnolence to the oxycodone (10 mg) the patient was taking for pain. The physician ordered a reduction in dosage.
If Mr. D had been your patient, would you have considered other possible causes of his somnolence? Or do you think the physician’s action was sufficient?
Derived from Latin, the word delirium literally means “off the [ploughed] track.”1 Dozens of terms have been used to describe delirium, with acute confusion state, organic brain syndrome, acute brain syndrome, and toxic psychosis among them.
Delirium has been reported to occur in 15% to 30% of patients on general medical units,2 about 40% of postoperative patients, and up to 70% of terminally ill patients.3 The true prevalence is hard to determine, as up to 66% of cases may be missed.4
Delirium is being diagnosed more frequently, however—a likely result of a growing geriatric population, increased longevity, and greater awareness of the condition. Each year, an estimated 2.3 million US residents are affected, leading to prolonged hospitalization; poor functional outcomes; the development or worsening of dementia; increased nursing home placement; and a significant burden for families and the US health care system.5
Delirium is also associated with an increase in mortality.6,7 The mortality rate among hospitalized patients who develop delirium is reported to be 18%, rising to an estimated 47% within the first 3 months after discharge.6 Greater awareness of risk factors, rapid recognition of signs and symptoms of delirium, and early intervention—detailed in the text and tables that follow—will lead to better outcomes.
Assessing risk, evaluating mental status
In addition to advanced age, risk factors for delirium (TABLE 1)8-14 include alcohol use, brain dysfunction, comorbidities, hypertension, malignancy, anticholinergic medications, anemia, metabolic abnormalities, and male sex. In patients who, like Mr. D, have numerous risk factors, early—and frequent—evaluation of mental status is needed. One way to do this is to treat mental status as a vital sign, to be included in the assessment of every elderly patient.15
The Confusion Assessment Method, a quick and easy-to-use delirium screening tool (TABLE 2), has a sensitivity of 94% to 100% and a specificity of 90% to 95%.16,17 There are a number of other screening tools, including the widely used Mini-Mental State Exam (MMSE), as well as the Delirium Rating Scale, Delirium Symptom Interview, and Delirium Severity Scale.
TABLE 1
Risk factors for delirium8-14
Advanced age Alcohol use Brain dysfunction (dementia, epilepsy) Hypertension Male sex Malignancy Medications (mainly anticholinergic) Metabolic abnormalities:
Old age Preoperative anemia Preoperative metabolic abnormalities |
| BUN, blood urea nitrogen; Cr, creatinine; Na, sodium. |
TABLE 2
Screening for delirium: The Confusion Assessment Method*16,17
| Criteria | Evidence Yes to questions 1, 2, and 3 plus 4 or 5 (or both) suggests a delirium diagnosis |
|---|---|
| 1. Acute onset | Is there evidence of an acute change in mental status from the patient’s baseline? |
| 2. Fluctuating course | Did the abnormal behavior fluctuate during the day—ie, tend to come and go or increase and decrease in severity? |
| 3. Inattention | Did the patient have difficulty focusing attention, eg, being easily distractible or having difficulty keeping track of what was being said? |
| PLUS | |
| 4. Disorganized thinking | Was the patient’s thinking disorganized or incoherent, such as rambling or irrelevant conversation, unclear or illogical flow of ideas, or unpredictable switching from subject to subject? |
| 5. Altered level of consciousness | Would you rate the patient’s level of consciousness as (any of the following): – Vigilant (hyperalert) – Lethargic (drowsy, easily aroused) – Stupor (difficult to arouse) – Coma (unarousable) |
| *CAM shortened version worksheet. Adapted from: Inouye SK et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113:941-948; Inouye SK. Confusion Assessment Method (CAM): Training Manual and Coding Guide. Copyright 2003, Hospital Elder Life Program, LLC. | |
Arriving at a delirium diagnosis
The clinical presentation of delirium is characterized by acute—and reversible—impairment of cognition, attention, orientation, and memory, and disruption of the normal sleep/wake cycle. The Diagnostic and Statistical Manual of Mental Disorders (DSM-IV-TR) criteria for a delirium diagnosis include all of the following:
- disturbance of consciousness, with a reduced ability to focus, sustain, or shift attention
- change in cognition, or a perceptual disturbance, that is not accounted for by a preexisting or developing dementia
- rapid onset of cognitive impairment, with fluctuation likely during the course of the day
- evidence from the history, physical exam, or laboratory findings that the disturbed consciousness is a direct physiological consequence of a general medical condition.17
There are 3 basic types of delirium, each associated with a different psychomotor disturbance.
- Hyperactive delirium—the least common—is characterized by restlessness and agitation, and is therefore the easiest to diagnose.
- Hypoactive delirium is characterized by psychomotor retardation and hypoalertness. It is often misdiagnosed as depression, and has the poorest prognosis.
- Mixed delirium—the most common—is characterized by symptoms that fluctuate between hyper- and hypoactivity.18
CASE By lunchtime, Mr. D had awakened; however, he needed help with his meal. After eating, he slept for the rest of the day. At night, a nurse paged the resident to report that the patient’s blood pressure was 82/60 mm Hg and his heart rate was 115. The physician ordered an intravenous fluid bolus, which corrected the patient’s hypotension, but only temporarily.
The fluctuating nature of delirium—most notably, in patients’ level of alertness—is helpful in establishing a diagnosis. The history and physical exam are the gold standard tools, both for diagnosing delirium and identifying the underlying cause (TABLE 3).19,20 A review of the patient’s medications should be a key component of the medical history, as drugs—particularly those with anticholinergic properties—are often associated with delirium. Environmental shifts, including hospitalization and a disruption of the normal sleep/wake cycle, endocrine disorders, infection, and nutritional deficiencies are also potential causes of delirium, among others.
If history and physical exam fail to identify the underlying cause, laboratory testing, including complete blood count, complete metabolic profile, and urinalysis, should be done. Brain imaging is usually not needed for individuals with symptoms of delirium, but computed tomography (CT) may be indicated if a patient’s condition continues to deteriorate while the underlying cause remains unidentified.21 Electroencephalography (EEG) may be used to confirm a delirium diagnosis that’s uncertain, in a patient with underlying dementia, for instance. (In more than 16% of cases of delirium, the cause is unknown.22)
The most common structural abnormalities found in patients with delirium are brain atrophy and increased white matter lesions, as well as basal ganglia lesions.23 Single-photon emission CT (SPECT) shows a reduction of regional cerebral perfusion by 50%,24 while EEG shows slowing of the posterior dominant rhythm and increased generalized slow-wave activity.25
TABLE 3
A DELIRIUM mnemonic to get to the heart of the problem19,20
| Cause | Comment |
|---|---|
| Drugs | Drug classes: Anesthesia, anticholinergics, anticonvulsants, antiemetics, antihistamines, antihypertensives, antimicrobials, antipsychotics, benzodiazepines, corticosteroids, hypnotics, H2 blockers, muscle relaxants, NSAIDs, opioids, SSRIs, tricyclic antidepressants Drugs: digoxin, levodopa, lithium, theophylline OTCs: henbane, Jimson weed, mandrake, Atropa belladonna extract |
| Environmental | Change of environment, sensory deprivation, sleep deprivation |
| Endocrine | Hyperparathyroidism, hyper-/hypothyroidism |
| Low perfusion | MI, pulmonary embolism, CVA |
| Infection | Pneumonia, sepsis, systemic infection, UTI |
| Retention | Fecal impaction, urinary retention |
| Intoxication | Alcohol, illegal drugs/drug overdose |
| Undernutrition | Malnutrition, thiamin deficiency, vitamin B12 deficiency |
| Metabolic | Acid-base disturbances, fluid and electrolyte abnormalities, hepatic or uremic encephalopathy, hypercarbia, hyper-/hypoglycemia, hyperosmolality, hypoxia |
| Subdural | History of falls |
| CVA, cerebrovascular accident; MI, myocardial infarction; NSAIDs, nonsteroidal anti-inflammatory drugs; OTCs, over-the-counter agents; SSRIs, selective serotonin reuptake inhibitors; UTI, urinary tract infection. | |
Treating (or preventing) delirium: Start with these steps
Nonpharmacologic interventions are the mainstay of treatment for patients with delirium, and may also help to prevent the development of delirium in patients at risk. One key measure is to correct, or avoid, disruptions in the patient’s normal sleep/wake cycle—eg, restoring circadian rhythm by avoiding,
to the extent possible, awakening the patient at night for medication or vital signs. Preventing sensory deprivation, by ensuring that the patient’s eyeglasses and hearing aid are nearby and that there is a clock and calendar nearby and adequate light, is also helpful. Other key interventions (TABLE 4)26-28 include:
- limiting medications associated with delirium (and eliminating any nonessential medication)
- improving nutrition and ambulation
- correcting electrolyte and fluid disturbances
- treating infection
- involving family members in patient care
- ensuring that patients receive adequate pain management
- avoiding transfers (if the patient is hospitalized) and trying to secure a single room.
Several studies have evaluated the effectiveness of nonpharmacologic interventions in preventing or lowering the incidence of delirium. A large multicomponent delirium prevention study of patients >70 years on general medical units focused on managing risk factors. The interventions studied included (1) avoidance of sensory deprivation, (2) early mobilization, (3) treating dehydration, (4) implementing noise reduction strategies and sleep enhancement programs, and (5) avoiding the use of sleep medications. These interventions proved to be effective not only in lowering the incidence of delirium, but in shortening the duration of delirium in affected patients (NNT=20).27
One study found that proactively using a geriatric consultation model (ie, implementing standardized protocols for the management of 6 risk factors) for elderly hospitalized patients led to a reduction in the incidence of delirium by more than a third.26 Admission to a specialized geriatric unit is associated with a lower incidence of delirium compared with being hospitalized on a general medical unit.29
Reducing the incidence of postoperative delirium. Bright light therapy (a light intensity of 5000 lux with a distance from the light source of 100 cm), implemented postoperatively, may play a role in reducing the incidence of delirium, research suggests.30 Music may be helpful, as well. An RCT involving patients (>65 years) undergoing elective knee or hip surgery found that those who listened to classical music postoperatively had a lower incidence of delirium.31 Similarly, playing music in nursing homes has been shown to decrease aggressive behavior and agitation.32
TABLE 4
Helpful interventions in the hospital or at home26-28
|
When medication is needed, proceed with caution
None of the medications currently used to treat delirium are approved by the US Food and Drug Administration for this indication, and many of them have substantial side effects. Nonetheless, palliative or symptomatic treatment requires some form of sedation for agitated patients with delirium. Thus, it is necessary to strike a balance in order to manage the symptoms of delirium and avoid potential side effects (primarily, sedation). Overly sedating patients can confuse the clinical picture of delirium and make it difficult to differentiate between ongoing delirium and medication side effects. Medication should be started at a low, but frequent, dose to achieve an effective therapeutic level, after which a lower maintenance dose can be used until the cause of delirium is resolved.
Antipsychotics are the cornerstone of drug treatment
Haloperidol has traditionally been used to treat delirium33 and has proven effectiveness. However, it is associated with increased risk of extrapyramidal manifestations compared with atypical antipsychotics.
Atypical antipsychotics (olanzapine, risperidone, quetiapine) are increasingly being used to treat delirium because they have fewer extrapyramidal side effects.34 With the exception of olanzapine (available in intramuscular and oral disintegrating form), atypical antipsychotics are available only in oral form, which may limit their usefulness as a treatment for agitated, delirious patients.
Risperidone (at a dose ranging from 0.25 to 1 mg/d) and olanzapine (1.25 to 2.5 mg/d) have shown similar efficacy to haloperidol (0.75 to 1.5 mg/d) in both the prevention and treatment of delirium, but with fewer extrapyramidal side effects.35-39 Quetiapine, a second-generation antipsychotic, is widely used to treat inpatient delirium, although there are no large RCTs comparing it with placebo. One pilot study and another open-label trial found the drug to be beneficial for patients with delirium, with fewer extrapyramidal side effects than haloperidol.40,41
Do a risk-benefit analysis. The use of antipsychotics in elderly patients with delirium has been associated with increased morbidity and mortality. The incidence of stroke and death were higher for community-dwelling patients (NNH=100) and patients in long-term care (N=67) who received typical or atypical antipsychotics for 6 months compared with that of patients who did not receive any antipsychotics.42,43 Thus, a risk-benefit analysis should be done before prescribing antipsychotics for elderly patients. Both typical and atypical antipsychotics carry black box warnings of increased mortality rates in the elderly.
Other drugs for delirium? More research is needed
Cholinesterase inhibitors. Procholinergic agents would be expected to be helpful in treating delirium, as cholinergic deficiency has been implicated as a predisposing factor for delirium and medications with anticholinergic effects have been shown to induce delirium. However, several studies of cholinesterase inhibitors have not found this to be the case.44-47
Benzodiazepines. There is no evidence to support the use of benzodiazepines in the treatment of delirium, except when the delirium is related to alcohol withdrawal.48 When indicated, the use of a short-acting benzodiazepine such as lorazepam is preferred for elderly patients (vs long-acting agents like diazepam) because of its shorter half-life and better side effect profile.2 Drowsiness, ataxia, and disinhibition are common side effects of benzodiazepines.
Gabapentin. A pilot study conducted to assess the efficacy of gabapentin (900 mg/d) for the prevention of postoperative delirium found a significantly lower incidence of delirium among patients who received gabapentin compared with placebo. This may be associated with gabapentin’s opioid-sparing effect.49 Larger studies are needed to recommend for or against the use of gabapentin in patients receiving opiates.
Further study of the pathophysiology of delirium is needed, as well, to increase our ability to prevent and treat it.
CASE After receiving the IV fluid bolus, Mr. D became increasingly short of breath and required more oxygen to keep his oxygen saturation in the 90s. Labs were ordered during morning rounds, and the patient was found to have urosepsis. He was admitted to the ICU in septic shock, and was intubated and died several days later.
In retrospect, it was determined that Mr. D had developed hypoactive delirium brought on by the infection—and that his somnolence on the second postoperative day was not a sign of overmedication. Had this been recognized early on through the use of an appropriate screening tool, the outcome would likely have been more favorable.
CORRESPONDENCE Abdulraouf Ghandour, MD, Green Meadows Clinic University Physicians, 3217 Providence Road, Columbia, MO 65203; [email protected]
1. Casselman WG. Dictionary of Medical Derivations. The Real Meaning of Medical Terms. New York, NY: Informa Healthcare; 1998.
2. Kiely DK, Bergmann MA, Murphy KM, et al. Delirium among newly admitted postacute facility patients, prevalence, symptoms, and severity. J Gerontol Biol Sci Med Sci. 2003;58:M441-M445.
3. Inouye SK, Charpentier PA. Precipitating factors for delirium in hospitalized elderly persons. Predictive model and interrelationship with baseline vulnerability. JAMA. 1996;275:852-857.
4. Inouye SK. The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med. 1994;97:278-288.
5. Pompei P, Foreman M, Rudberg M, et al. Delirium in hospitalized older persons: outcomes and predictors. J Am Geriatr Soc. 1994;42:809-815.
6. Kolbeinsson H, Jonsson A. Delirium and dementia in acute medical admissions of elderly patients in Iceland. Acta Psychiatr Scand. 1993;87:123-127.
7. Cole MG, Primeau FJ. Prognosis of delirium in elderly hospital patients. CMAJ. 1993;149:41-46.
8. Rahkonen T, Eloniemi-Sulkava U, Halonen P, et al. Delirium in the non-demented oldest old in the general population: risk factors and prognosis. Int J Geriatr Psychiatry. 2001;16:415-421.
9. Edlund A, Lundstrom M, Brannstrom B, et al. Delirium before and after operation for femoral neck fracture. J Am Geriatr Soc. 2001;49:1335-1340.
10. Andersson EM, Gustafson L, Hallberg IR. Acute confusional state in elderly orthopaedic patients: factors of importance for detection in nursing care. Int J Geriatr Psychiatry. 2001;16:7-17.
11. Inouye SK, Viscoli CM, Horwitz RI, et al. A predictive model for delirium in hospitalized elderly medical patients based on admission characteristics. Ann Intern Med. 1993;119:474-481.
12. Marcantonio ER, Juarez G, Goldman L, et al. The relationship of postoperative delirium with psychoactive medications. JAMA. 1994;272:1518-1522.
13. Marcantonio ER, Goldman L, Orav EJ, et al. The association of intraoperative factors with the development of postoperative delirium. Am J Med. 1998;105:380-384.
14. Tune L, Carr S, Hoag E, et al. Anticholinergic effects of drugs commonly prescribed for the elderly: potential means for assessing risk of delirium. Am J Psychiatry. 1992;149:1393-1394.
15. Flaherty JH, Shay K, Weir C, et al. The development of a mental status vital sign for use across the spectrum of care . J Am Med Dir Assoc. 2009;10:379-380.
16. Inouye SK, Van Dyck CH, Alessi CA, et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113:941-948.
17. Inouye SK. Confusion Assessment Method (CAM): Training Manual and Coding Guide. New Haven, Conn: Yale University School of Medicine; 2003.
18. Halter J, Ouslander J, Tinetti M, et al. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York, NY: McGraw-Hill; 2009;648-658.
19. Eriksson S. Social and environmental contributants to delirium in the elderly. Dement Geriatr Cogn Disord. 1999;10:350-352.
20. Francis J, Martin D, Kapoor WN. A prospective study of delirium in hospitalized elderly. JAMA. 1990;263:1097-1101.
21. Francis J, Hilko EM, Kapoor WN. Acute mental change: when are head scans needed? Clin Res. 1991;39:103.-
22. Rudberg MA, Pompei P, Foreman MD, et al. The natural history of delirium in older hospitalized patients: a syndrome of heterogeneity. Age Ageing. 1997;26:169-174.
23. Soiza RL, Sharma V, Ferguson K, et al. Neuroimaging studies of delirium: a systematic review. J Psychosom Res. 2008;65:239-248.
24. Fong TG, Bogardus ST Jr, Daftary A, et al. Cerebral perfusion changes in older delirious patients using 99mTc HMPAO SPECT. J Gerontol A Biol Sci Med Sci. 2006;61:1294-1299.
25. Jacobson SA, Leuchter AF, Walter DO. Conventional and quantitative EEG in the diagnosis of delirium among the elderly. J Neurol Neurosurg Psychiatry. 1993;56:153-158.
26. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2001;49:516-522.
27. Inouye SK, Bogardus ST Jr, Charpentier PA, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. N Engl J Med. 1999;340:669-676.
28. Weber JB, Coverdale JH, Kunik ME. Delirium: current trends in prevention and treatment. Intern Med J. 2004;34:115-121.
29. Bo M, Martini B, Ruatta C, et al. Geriatric ward hospitalization reduced incidence delirium among older medical inpatients. Am J Geriatr Psychiatry. 2009;17:760-768.
30. Taguchi T, Yano M, Kido Y. Influence of bright light therapy on postoperative patients: a pilot study. Intensive Crit Care Nurs. 2007;23:289-297.
31. McCaffrey R, Locsin R. The effect of music listening on acute confusion and delirium in elders undergoing elective hip and knee surgery. J Clin Nurs. 2004;13:91-96.
32. Remington R. Calming music and hand massage with agitated elderly. Nurs Res. 2004;51:317-323.
33. Seitz DP, Gill SS, van Zyl LT. Antipsychotics in the treatment of delirium: a systematic review. J Clin Psychiatry. 2007;68:11-21.
34. Schwartz T, Masand PS. The role of atypical antipsychotics in the treatment of delirium. Psychosomatics. 2002;43:171-174.
35. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594.-
36. Hu H, Deng W, Yang H. A prospective random control study comparison of olanzapine and haloperidol in senile delirium. Chongqing Med J. 2004;8:1234-1237.
37. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45:297-301.
38. Kim SW, Yoo JA, Lee SY, et al. Risperidone versus olanzapine for the treatment of delirium. Hum Psychopharmacol. 2010;25:298-302.
39. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35:714-719.
40. Maneeton B, Maneeton N, Srisurapanont M. An open-label study of quetiapine for delirium. J Med Assoc Thai. 2007;90:2158-2163.
41. Devlin JW, Roberts RJ, Fong JJ, et al. Efficacy and safety of quetiapine in critically ill patients with delirium: a prospective, multicenter, randomized, double-blind, placebo-controlled pilot study. Crit Care Med. 2010;38:419-427.
42. Gill SS, Bronskill SE, Normand SL, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146:775-786.
43. Wang PS, Schneeweiss S, Avorn J, et al. Death in elderly users of conventional vs. atypical antipsychotic medications. N Engl J Med. 2005;353:2335-2341.
44. Liptzin B, Laki A, Garb JL, et al. Donepezil in the prevention and treatment of post-surgical delirium. Am J Geriatr Psychiatry. 2005;13:1100-1106.
45. Sampson EL, Raven PR, Ndhlovu PN, et al. A randomized, double-blind, placebo-controlled trial of donepezil hydrochloride (Aricept) for reducing the incidence of postoperative delirium after elective total hip replacement. Int J Geriatr Psychiatry. 2007;22:343-349.
46. Gamberini M, Bolliger D, Lurati Buse GA, et al. Rivastigmine for the prevention of postoperative delirium in elderly patients undergoing elective cardiac surgery—a randomized controlled trial. Crit Care Med. 2009;37:1762-1768.
47. Overshott R, Vernon M, Morris J, et al. Rivastigmine in the treatment of delirium in older people: a pilot study. Int Psychogeriatr. 2010;22:812-818.
48. Lonergan E, Luxenberg J, Areosa Sastre A. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;(4):CD006379.-
49. Leung JM, Sands LP, Rico M, et al. Pilot clinical trial of gabapentin to decrease postoperative delirium in older patients. Neurology. 2006;67:1251-1253.
1. Casselman WG. Dictionary of Medical Derivations. The Real Meaning of Medical Terms. New York, NY: Informa Healthcare; 1998.
2. Kiely DK, Bergmann MA, Murphy KM, et al. Delirium among newly admitted postacute facility patients, prevalence, symptoms, and severity. J Gerontol Biol Sci Med Sci. 2003;58:M441-M445.
3. Inouye SK, Charpentier PA. Precipitating factors for delirium in hospitalized elderly persons. Predictive model and interrelationship with baseline vulnerability. JAMA. 1996;275:852-857.
4. Inouye SK. The dilemma of delirium: clinical and research controversies regarding diagnosis and evaluation of delirium in hospitalized elderly medical patients. Am J Med. 1994;97:278-288.
5. Pompei P, Foreman M, Rudberg M, et al. Delirium in hospitalized older persons: outcomes and predictors. J Am Geriatr Soc. 1994;42:809-815.
6. Kolbeinsson H, Jonsson A. Delirium and dementia in acute medical admissions of elderly patients in Iceland. Acta Psychiatr Scand. 1993;87:123-127.
7. Cole MG, Primeau FJ. Prognosis of delirium in elderly hospital patients. CMAJ. 1993;149:41-46.
8. Rahkonen T, Eloniemi-Sulkava U, Halonen P, et al. Delirium in the non-demented oldest old in the general population: risk factors and prognosis. Int J Geriatr Psychiatry. 2001;16:415-421.
9. Edlund A, Lundstrom M, Brannstrom B, et al. Delirium before and after operation for femoral neck fracture. J Am Geriatr Soc. 2001;49:1335-1340.
10. Andersson EM, Gustafson L, Hallberg IR. Acute confusional state in elderly orthopaedic patients: factors of importance for detection in nursing care. Int J Geriatr Psychiatry. 2001;16:7-17.
11. Inouye SK, Viscoli CM, Horwitz RI, et al. A predictive model for delirium in hospitalized elderly medical patients based on admission characteristics. Ann Intern Med. 1993;119:474-481.
12. Marcantonio ER, Juarez G, Goldman L, et al. The relationship of postoperative delirium with psychoactive medications. JAMA. 1994;272:1518-1522.
13. Marcantonio ER, Goldman L, Orav EJ, et al. The association of intraoperative factors with the development of postoperative delirium. Am J Med. 1998;105:380-384.
14. Tune L, Carr S, Hoag E, et al. Anticholinergic effects of drugs commonly prescribed for the elderly: potential means for assessing risk of delirium. Am J Psychiatry. 1992;149:1393-1394.
15. Flaherty JH, Shay K, Weir C, et al. The development of a mental status vital sign for use across the spectrum of care . J Am Med Dir Assoc. 2009;10:379-380.
16. Inouye SK, Van Dyck CH, Alessi CA, et al. Clarifying confusion: the Confusion Assessment Method. A new method for detection of delirium. Ann Intern Med. 1990;113:941-948.
17. Inouye SK. Confusion Assessment Method (CAM): Training Manual and Coding Guide. New Haven, Conn: Yale University School of Medicine; 2003.
18. Halter J, Ouslander J, Tinetti M, et al. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York, NY: McGraw-Hill; 2009;648-658.
19. Eriksson S. Social and environmental contributants to delirium in the elderly. Dement Geriatr Cogn Disord. 1999;10:350-352.
20. Francis J, Martin D, Kapoor WN. A prospective study of delirium in hospitalized elderly. JAMA. 1990;263:1097-1101.
21. Francis J, Hilko EM, Kapoor WN. Acute mental change: when are head scans needed? Clin Res. 1991;39:103.-
22. Rudberg MA, Pompei P, Foreman MD, et al. The natural history of delirium in older hospitalized patients: a syndrome of heterogeneity. Age Ageing. 1997;26:169-174.
23. Soiza RL, Sharma V, Ferguson K, et al. Neuroimaging studies of delirium: a systematic review. J Psychosom Res. 2008;65:239-248.
24. Fong TG, Bogardus ST Jr, Daftary A, et al. Cerebral perfusion changes in older delirious patients using 99mTc HMPAO SPECT. J Gerontol A Biol Sci Med Sci. 2006;61:1294-1299.
25. Jacobson SA, Leuchter AF, Walter DO. Conventional and quantitative EEG in the diagnosis of delirium among the elderly. J Neurol Neurosurg Psychiatry. 1993;56:153-158.
26. Marcantonio ER, Flacker JM, Wright RJ, et al. Reducing delirium after hip fracture: a randomized trial. J Am Geriatr Soc. 2001;49:516-522.
27. Inouye SK, Bogardus ST Jr, Charpentier PA, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. N Engl J Med. 1999;340:669-676.
28. Weber JB, Coverdale JH, Kunik ME. Delirium: current trends in prevention and treatment. Intern Med J. 2004;34:115-121.
29. Bo M, Martini B, Ruatta C, et al. Geriatric ward hospitalization reduced incidence delirium among older medical inpatients. Am J Geriatr Psychiatry. 2009;17:760-768.
30. Taguchi T, Yano M, Kido Y. Influence of bright light therapy on postoperative patients: a pilot study. Intensive Crit Care Nurs. 2007;23:289-297.
31. McCaffrey R, Locsin R. The effect of music listening on acute confusion and delirium in elders undergoing elective hip and knee surgery. J Clin Nurs. 2004;13:91-96.
32. Remington R. Calming music and hand massage with agitated elderly. Nurs Res. 2004;51:317-323.
33. Seitz DP, Gill SS, van Zyl LT. Antipsychotics in the treatment of delirium: a systematic review. J Clin Psychiatry. 2007;68:11-21.
34. Schwartz T, Masand PS. The role of atypical antipsychotics in the treatment of delirium. Psychosomatics. 2002;43:171-174.
35. Lonergan E, Britton AM, Luxenberg J, et al. Antipsychotics for delirium. Cochrane Database Syst Rev. 2007;(2):CD005594.-
36. Hu H, Deng W, Yang H. A prospective random control study comparison of olanzapine and haloperidol in senile delirium. Chongqing Med J. 2004;8:1234-1237.
37. Han CS, Kim YK. A double-blind trial of risperidone and haloperidol for the treatment of delirium. Psychosomatics. 2004;45:297-301.
38. Kim SW, Yoo JA, Lee SY, et al. Risperidone versus olanzapine for the treatment of delirium. Hum Psychopharmacol. 2010;25:298-302.
39. Prakanrattana U, Prapaitrakool S. Efficacy of risperidone for prevention of postoperative delirium in cardiac surgery. Anaesth Intensive Care. 2007;35:714-719.
40. Maneeton B, Maneeton N, Srisurapanont M. An open-label study of quetiapine for delirium. J Med Assoc Thai. 2007;90:2158-2163.
41. Devlin JW, Roberts RJ, Fong JJ, et al. Efficacy and safety of quetiapine in critically ill patients with delirium: a prospective, multicenter, randomized, double-blind, placebo-controlled pilot study. Crit Care Med. 2010;38:419-427.
42. Gill SS, Bronskill SE, Normand SL, et al. Antipsychotic drug use and mortality in older adults with dementia. Ann Intern Med. 2007;146:775-786.
43. Wang PS, Schneeweiss S, Avorn J, et al. Death in elderly users of conventional vs. atypical antipsychotic medications. N Engl J Med. 2005;353:2335-2341.
44. Liptzin B, Laki A, Garb JL, et al. Donepezil in the prevention and treatment of post-surgical delirium. Am J Geriatr Psychiatry. 2005;13:1100-1106.
45. Sampson EL, Raven PR, Ndhlovu PN, et al. A randomized, double-blind, placebo-controlled trial of donepezil hydrochloride (Aricept) for reducing the incidence of postoperative delirium after elective total hip replacement. Int J Geriatr Psychiatry. 2007;22:343-349.
46. Gamberini M, Bolliger D, Lurati Buse GA, et al. Rivastigmine for the prevention of postoperative delirium in elderly patients undergoing elective cardiac surgery—a randomized controlled trial. Crit Care Med. 2009;37:1762-1768.
47. Overshott R, Vernon M, Morris J, et al. Rivastigmine in the treatment of delirium in older people: a pilot study. Int Psychogeriatr. 2010;22:812-818.
48. Lonergan E, Luxenberg J, Areosa Sastre A. Benzodiazepines for delirium. Cochrane Database Syst Rev. 2009;(4):CD006379.-
49. Leung JM, Sands LP, Rico M, et al. Pilot clinical trial of gabapentin to decrease postoperative delirium in older patients. Neurology. 2006;67:1251-1253.
How best to address these common movement disorders
• Initiate neuroprotective therapy with a monoamine oxidase B inhibitor to slow the progression of Parkinson’s disease. With onset of functional impairment, give levodopa at the lowest effective dose. A
• Give propranolol for essential tremor causing a patient distress, starting at 20 to 40 mg twice daily and increasing the dose (to a maximum of 320 mg/d) until relief is achieved. B
• Consider giving a dopamine receptor blocker for Tourette syndrome or other tic disorder; alternative agents are clonidine or a newer agent, tetrabenazine. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Movement disorders often require consultation with a neurologist, and a working knowledge of established and novel treatments can set the stage for optimal long-term cooperative management.1 In this article, we review therapeutic options for common movement disorders, including hypokinetic, hyperkinetic, and dyskinetic disturbances.
Parkinson’s disease treatment: MAO-B inhibitor, levodopa are mainstays
Parkinson’s disease, the most common hypokinetic movement disorder, is a chronic, progressive, neurodegenerative disease. It affects 1% of individuals older than 65 years and 4% to 5% of individuals older than 85 years. Its cardinal symptoms are resting tremor, bradykinesia, rigidity, a flexed posture, and loss of postural reflexes. Resting tremor, referred to as “pill rolling” tremor, is 4 to 6 Hz and usually begins unilaterally.2,3 Associated symptoms can include dystonia, dementia, psychiatric disorders, sleep disorders, and autonomic symptoms.
Neuroprotective therapy is used to slow the progression of the disease, particularly in its early stage. The monoamine oxidase B (MAO-B) inhibitor selegiline has proven effective in this regard2 (strength of recommendation [SOR]: A). In randomized controlled studies, selegiline has delayed the need for levodopa for 9 to 12 months4 (SOR: A). Another MAO-B inhibitor, rasagiline, has demonstrated neuroprotective effects as well5 (SOR: B). These medications may also be used with levodopa for symptom control and as adjuvant therapy in patients with motor fluctuations.2 A conventional dose of selegiline is 10 mg/d (5 mg at breakfast; 5 mg at lunch). Rasagiline is given at 1 mg/d. Concomitant use of ciprofloxacin or other CYP1A2 inhibitors limits its effectiveness.6,7
Symptomatic therapy is indicated at the onset of functional impairment. The dopamine precursor levodopa is the most widely used and effective drug for Parkinson’s disease symptoms, especially bradykinesia and rigidity. Use the lowest possible dose to control symptoms (eg, 100 mg twice daily) and protect against motor complications of the drug7-9 (SOR: A). To prevent conversion of levodopa to dopamine outside the blood-brain barrier, combine it with the decarboxylase inhibitor carbidopa. Dietary restriction of proteins may be needed, because amino acids can interfere with the absorption of levodopa.
Especially with prolonged use, levodopa can cause disturbing adverse effects, such as nausea, vomiting, psychosis, cardiac arrhythmia, and orthostatic hypotension. Dyskinesias and motor fluctuations are complications of long-term treatment and are irreversible. Adding a cathecol-O-methyltransferase (COMT) inhibitor, such as entacapone, to increase levodopa’s effectiveness has been shown to reduce motor fluctuations2,3,10 (SOR: B). Dopamine agonists such as bromocriptine, ropinirole, and pramipexole used in early Parkinson’s disease can also reduce dyskinesias and motor fluctuations. Dopamine agonists may be preferred to levodopa in early Parkinson’s disease because they are better tolerated and cause fewer adverse effects. Or they may be used as adjuncts for patients whose response to levodopa is deteriorating or fluctuating3,7,8 (SOR: B). In advanced disease, motor complications can also be managed by augmenting levodopa therapy with a dopamine agonist, MAO-B inhibitor, or COMT inhibitor7,8 (SOR: A).
Anticholinergics, mainly benztropine and trihexyphenidyl, may be used as symptomatic treatment, especially in young people with early Parkinson’s disease and severe tremor. However, they are not the first drugs of choice due to limited efficacy and the potential for neuropsychiatric side effects8 (SOR: C). Amantadine can reduce dyskinesia in people with advanced Parkinson’s disease8 (SOR: C). For patients who have Parkinson’s disease with severe motor complications, intermittent apomorphine injections can help reduce “off time” periods in the daily treatment cycle when the efficacy of drugs wanes9 (SOR: B).
Deep brain stimulation of the subthalamic nucleus has only SOR C support for reducing dyskinesias and off time.9
Treating nonmotor symptoms of Parkinson’s disease can be challenging. For dementia in these patients, consider cholinesterase inhibitors6,8 (SOR: C). For depression, selective serotonin reuptake inhibitors are effective6,8,9 (SOR: C). For psychosis, preferred agents are low-dose clozapine or quetiapine6,8-10 (SOR: C). Plan for supportive and symptomatic management of constipation, dysphagia, sialorrhea, orthostatic hypotension, sleep disturbances, and urinary urgency.2,3
Tremor
Tremor is a common form of hyperkinesia, presenting either as a primary disorder or as a symptom of another condition.11 By definition, it is a rhythmical, involuntary, oscillatory movement of 1 or more body parts. Tremors are classified as rest or action tremors, with the latter being further categorized as postural (occurring while the patient maintains a position against gravity) or kinetic (occurring during voluntary movement).2,10
Physiologic tremor: Pharmacologic Tx is usually not needed
Physiologic tremor is benign, high frequency (8-12 Hz), low amplitude, and postural. An exaggerated form of this tremor may result from anxiety, hyperthyroidism, pheochromocytoma, hypoglycemia, excessive caffeine consumption, fever, withdrawal from opioids and sedatives, and some medications. No drug treatment is necessary unless symptoms become bothersome. Correct the underlying cause or have the patient avoid the triggering factor, and offer reassurance that the condition is not pathological or progressive.2,12 For anxiety, consider cognitive-behavioral/relaxation therapy or benzodiazepines (if tremor did not result from withdrawal of benzodiazepines) or beta-adrenergic antagonists (eg, propranolol).12,13
Essential tremor: Try propranolol or primidone first
Essential tremor (ET) is the most common movement disorder. It often results in functional disability and leads to many physical and emotional difficulties. ET is bilateral, usually symmetric (although mild asymmetry is possible), and postural or kinetic, typically affecting hands and forearms. The frequency of ET is 4 to 12 Hz. Cranial musculature may be involved in 30% of cases, affecting the head and voice.3 Prevalence ranges from 4 to 40 cases per 1000 people. The age-adjusted incidence is 17.5/100,000 per year; it peaks during the teen years and the fifth decade.2,3
Autosomal dominant type of inheritance is common, and a family history of ET is often present, particularly with younger patients. The differential diagnosis includes Parkinson’s disease tremor; dystonic, cerebellar, rubral, and psychogenic tremors; and asterixis.3 Unlike ET, many of these disorders have associated neurologic, psychiatric, or systemic signs.
Treatment with propranolol or primidone is indicated if ET causes functional impairment or social or emotional problems for the patient.2,3,10,13 Both propranolol and primidone reduce limb tremor2,10,13 (SOR: B), but only propranolol is approved by the US Food and Drug Administration (FDA) for treatment of ET. Propranolol is more effective for hand and forearm tremor than for head and voice tremor. Start propranolol at 20 to 40 mg twice a day and increase the dose as needed to achieve symptom relief.14
A maintenance dose of 240 to 320 mg/d may be needed. Major adverse effects are fatigue, sedation, depression, and erectile dysfunction. Contraindications to propranolol include asthma, second-degree atrioventricular block, and insulin-dependent diabetes.
If starting with primidone alone, prescribe at a dose <25 mg at bedtime and increase the dose slowly over several weeks to prevent onset of nausea, vomiting, sedation, confusion, or ataxia. The maximum allowable dose is 750 mg/d in 3 divided doses.10 Primidone and propranolol may be used in combination to treat limb tremor when monotherapy is insufficient (SOR: B).13
Thirty percent of patients with ET will not respond to propranolol or primidone. An alternative choice is the anticonvulsant gabapentin10,12-14 (SOR: C). However, clinical experience with it is limited. Lethargy, fatigue, decreased libido, dizziness, nervousness, and shortness of breath are adverse effects of gabapentin; they are usually mild and tolerable.13 Topiramate is another option that seems to be as effective as gabapentin10,13 (SOR: C), but studies of long-term outcomes are lacking. Topiramate’s side effects include weight loss and paresthesias. Additionally, alprazolam, clonazepam, clozapine, olanzapine, atenolol, sotalol, nadolol, and nimodipine may reduce limb tremor2 (SOR: C). Alcohol reduces tremor amplitude in 50% to 90% of patients, but tremor may worsen after the effect of alcohol has worn off.15
For patients with essential hand tremor that fails to respond to oral agents, consider botulinum toxin A16 (SOR: B). However, it is also associated with dose-dependent hand weakness16 (SOR: C). Botulinum toxin may reduce head and voice tremor16 (SOR: C), but hoarseness and swallowing difficulties may occur after use for voice tremor.16
Invasive therapies may benefit patients with refractory tremor. Deep brain stimulation and thalamotomy are highly effective in reducing limb tremor13 (SOR: C). Each carries a small risk of major complications. Some deep brain stimulation adverse events may resolve with time. Other adverse events may resolve with adjustment of stimulator settings. No evidence exists for surgical treatment for voice and head tremor or for gamma-knife thalamotomy.13
Drug-induced tremor
Drugs with the potential to cause postural tremor, intention tremor, or rest tremor include the following: 15
- alcohol (chronic)
- amiodarone
- amphetamines
- beta-adrenergic agonists
- caffeine
- calcitonin
- carbamazepine
- cocaine
- cyclosporine
- dopamine
- lithium
- metoclopramide
- neuroleptics
- procainamide
- steroids
- theophylline
- thyroid hormones
- tricyclic antidepressants
- trifluoperazine
- valproic acid
With drug-induced tremor, carefully evaluate a patient’s need for the drug. Discontinue the offending agent if possible, or try lowering the dose.
Psychogenic tremor: A history of somatization is a clue
Psychogenic tremor can occur at rest or during postural or kinetic movement. Clinical features include an abrupt onset, a static course, spontaneous remission, and unclassifiable tremors.17 Psychogenic tremor increases under direct observation and decreases with distraction. Patients with psychogenic tremor often have a history of somatization.18 Electrophysiologic testing can help confirm the diagnosis. If remission does not occur spontaneously, patients may find relief with psychotherapy or placebo.19
Tic disorders: Opt for dopamine receptor blockers
Tics are involuntary or semivoluntary movements or sounds that are sudden, brief, intermittent, repetitive, nonrhythmic, unpredictable, and purposeless. Tics can occur in any part of the body.20
The most common tic disorder is Tourette syndrome—a combination of motor and phonic tics with onset before age 21. It affects approximately 5 to 10 children out of 10,000. Boys are more commonly affected than girls. Attention deficit hyperactivity disorder frequently accompanies this syndrome.2
The goal of treatment with any tic disorder is to improve social functioning, self-esteem, and quality of life. Education and support of patients is important. Tic disorders, including Tourette, rarely require drugs. But if tics become too distressing, first-line treatment would be a dopamine modulator, tetrabenazine, or clonidine. Randomized controlled trials with various neuroleptics have revealed dramatic reductions in tic severity. However, many patients do not tolerate the acute adverse effects (most commonly sedation, weight gain, depression, lethargy, and akathisia), and prolonged treatment confers a small risk of tardive dyskinesia. Behavioral therapy is an important part of management.20
Dopamine-receptor blocking drugs such as haloperidol, pimozide, and fluphenazine are the most effective treatment for tics20 (SOR: B). Tetrabenazine is a promising new dopamine-depleting drug; controlled trials are ongoing2,20 (SOR: B). Clonidine, an alpha 2-adrenergic agonist, is useful in treating patients with Tourette syndrome, helping to improve sleep and attention2,21 (SOR: C). Medically refractory motor and disabling phonic tics such as coprolalia can be ameliorated by botulinum toxin injections21 (SOR: B). Deep brain stimulation is being used at an increasing rate for medically refractory tics in Tourette syndrome21 (SOR: B).
Restless legs syndrome: Dopamine agonists are preferred
Restless legs syndrome (RLS) is a disorder characterized by sensory symptoms and motor disturbances of the legs, mainly during rest. Treatment may not be necessary for patients with mild or sporadic symptoms. For moderate to severe RLS with significant impairment, dopamine agonists are the preferred agents22 (SOR: A). RLS can also occur secondary to such conditions as iron deficiency and uremia, and correction of the underlying disorder is the goal. Prescribe iron replacement for patients with a ferritin level <50 ng/mL22 (SOR: C). Medications known to cause or exacerbate the symptoms of RLS are anti-dopaminergic agents (such as neuroleptics), diphenhydramine, tricyclic antidepressants, alcohol, caffeine, lithium, and beta-blockers. If a patient is taking medications that exacerbate symptoms of RLS, discontinue them and use appropriate substitutes22 (SOR: C).
Myoclonus: Clonazepam for essential disorder
Myoclonus is a brief, sudden, shock-like movement caused by involuntary muscle contractions or lapse of muscle contraction (asterixis). Given the complex origins of myoclonus, multiple drugs may be needed. Essential myoclonus is disabling and can be treated with clonazepam. Start with 0.25 mg orally twice daily, and increase the dosage over 3 days to 1 mg/d23 (SOR: C). Most cases of myoclonus are secondary due to drugs such as lithium, toxins, advanced liver disease, infections including human immunodeficiency virus, dementia, and brain lesions. Treatment should also address the underlying disorder.2,23
Chorea
Chorea is an abnormal involuntary movement disorder described as “a state of excessive, spontaneous movements, irregularly timed, nonrepetitive, randomly distributed, and abrupt in character.”24
Treatment of chorea is symptomatic, aiming to reduce morbidity and prevent complications. Haloperidol and fluphenazine are effective but can impair voluntary movements2,10,25 (SOR: C). The dopamine-depleting drugs reserpine and tetrabenazine are also effective2,10,25 (SOR: C). GABAergic drugs, such as clonazepam, gabapentin, and valproate, can be used adjunctively.10,25
Dystonia
Dystonia is a syndrome involving sustained contractions of opposing muscles that cause twisting, repetitive movements and abnormal postures. Primary dystonia can be treated successfully with high doses of trihexyphenidyl alone, starting with 1 mg orally per day and increasing gradually to 6 to 80 mg/d until symptoms are controlled; or in combination with baclofen, starting with 10 mg orally once daily and increasing to a maximum dose of 30 to 120 mg/d1,2 (SOR: C).
Consider botulinum neurotoxin injection for focal upper extremity dystonia and adductor spasmodic dysphonia16 (SOR: B).
Ataxia
Ataxia is an unsteady gait associated with cerebellar dysfunction, proprioceptive defects, or both. Ataxia may be primary (Friedreich ataxia and spinocerebellar ataxia) or secondary to stroke, trauma, alcoholic degeneration, multiple sclerosis, vitamin B12 deficiency, and hydrocephalus. Treatment, when possible, should target the underlying cause.1,2
CORRESPONDENCE Hakan Yaman, MD, Akdeniz University, Department of Family Medicine, Antalya, Turkey 07058; [email protected]
1. Deuschl G, Bain P, Brin M. Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee. Mov Disord. 1998;13(suppl 3):2-23.
2. Yaman A, Yaman H, Rao G. Tremors and other movement disorders. In: Mengel MB, et al, eds. Family Medicine Ambulatory Care and Prevention. 5th ed. New York: McGraw-Hill; 2009:400–407.
3. Harris MK, Shneyder N, Borazanci A, et al. Movement disorders. Med Clin North Am. 2009;93:371-388.
4. Palhagen S, Heinonen E, Hagglund J, et al. Selegiline slows the progression of the symptoms of Parkinson disease. Neurology. 2006;66:1200-1206.
5. Olanow CW, Rascol O, Hauser R, et al. A double-blind, delayed-start trial of rasagiline in Parkinson’s disease. N Engl J Med. 2009;361:1268-1278.
6. Zesiewicz TA, Sullivan KL, Arnulf I, et al. Treatment of nonmotor symptoms of Parkinson disease. Neurology. 2010;74:924-931.
7. Suchowersky O, Reich S, Perlmutter J, et al. Practice parameter: diagnosis and prognosis of new onset Parkinson disease (an evidence-based review). Neurology. 2006;66:968-975.
8. Rao SS, Hofmann LA, Shakil A. Parkinson’s disease: diagnosis and treatment. Am Fam Physician. 2006;74:2046-2054.
9. The National Collaborating Centre for Chronic Conditions Parkinson’s Disease. National Clinical Guideline for Diagnosis and Management in Primary and Secondary Care. 2006. Available at: http://www.nice.org.uk/nicemedia/live/10984/30087/30087.pdf. Accessed May 12, 2010.
10. Jankovic J. Treatment of hyperkinetic movement disorders. Lancet Neurol. 2009;8:844-856.
11. Kerlsberg G, Rubenstein C, St Anna L, et al. Differential diagnosis of tremor. Am Fam Physician. 2008;77:1305-1306.
12. Burke DA, Hauser RA, McClain T. Essential tremor. Available at: http://emedicine.medscape.com/article/1150290-overview. Accessed May 12, 2010.
13. Zesiewicz TA, Elble R, Louis ED, et al. Practice parameter: therapies for essential tremor. Neurology. 2005;64:2008-2020.
14. Elble RJ. Tremor: clinical features, pathophysiology, and treatment. Neurol Clin. 2009;27:679-695.
15. Smaga S. Tremor. Am Fam Physician. 2003;68:1545-1552.
16. Use of botulinum neurotoxin for the treatment of movement disorders. AAN summary of evidence-based guidelines for clinicians. 2008. Available at: http://www.aan.com/practice/guideline/uploads/280.pdf. Accessed May 25, 2010.
17. Redondo L, Morgado Y, Durán E. Psychogenic tremor: a positive diagnosis [ in Spanish]. Neurología. 2010;25:51-57.
18. Schwingenschuh P, Katschnig P, Seiler S, et al. Moving toward ‘‘laboratory-supported’’ criteria for psychogenic tremor. Mov Disord. 2011;Sep 28. [Epub ahead of print].
19. McKeon A, Ahlskog JE, Bower JH, et al. Psychogenic tremor: long-term prognosis in patients with electrophysiologically confirmed disease. Mov Disord. 2009;24:72-76.
20. Shprecher D, Kurlan R. The management of tics. Mov Disord. 2009;24:15-24.
21. Kenney C, Kuo SH, Jimenez-Shahed J. Tourette’s syndrome. Am Fam Physician. 2008;77:651-658.
22. Bayard M, Avonda T, Wadzinsky J. Restless legs syndrome. Am Fam Physician. 2008;78:235-240.
23. Caviness JN. Pathophysiology and treatment of myoclonus. Neurol Clin. 2009;27:757-777.
24. Barbeau A, Duvoisin RC, Gerstenbrand F, et al. Classification of extrapyramidal disorders. J Neurol Sci. 1981;51:311-327.
25. Vertrees SM, Berman SA. Chorea in adults: treatment & management. Available at: http://emedicine.medscape.com/article/1149854-treatment. Accessed February 12, 2010.
• Initiate neuroprotective therapy with a monoamine oxidase B inhibitor to slow the progression of Parkinson’s disease. With onset of functional impairment, give levodopa at the lowest effective dose. A
• Give propranolol for essential tremor causing a patient distress, starting at 20 to 40 mg twice daily and increasing the dose (to a maximum of 320 mg/d) until relief is achieved. B
• Consider giving a dopamine receptor blocker for Tourette syndrome or other tic disorder; alternative agents are clonidine or a newer agent, tetrabenazine. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Movement disorders often require consultation with a neurologist, and a working knowledge of established and novel treatments can set the stage for optimal long-term cooperative management.1 In this article, we review therapeutic options for common movement disorders, including hypokinetic, hyperkinetic, and dyskinetic disturbances.
Parkinson’s disease treatment: MAO-B inhibitor, levodopa are mainstays
Parkinson’s disease, the most common hypokinetic movement disorder, is a chronic, progressive, neurodegenerative disease. It affects 1% of individuals older than 65 years and 4% to 5% of individuals older than 85 years. Its cardinal symptoms are resting tremor, bradykinesia, rigidity, a flexed posture, and loss of postural reflexes. Resting tremor, referred to as “pill rolling” tremor, is 4 to 6 Hz and usually begins unilaterally.2,3 Associated symptoms can include dystonia, dementia, psychiatric disorders, sleep disorders, and autonomic symptoms.
Neuroprotective therapy is used to slow the progression of the disease, particularly in its early stage. The monoamine oxidase B (MAO-B) inhibitor selegiline has proven effective in this regard2 (strength of recommendation [SOR]: A). In randomized controlled studies, selegiline has delayed the need for levodopa for 9 to 12 months4 (SOR: A). Another MAO-B inhibitor, rasagiline, has demonstrated neuroprotective effects as well5 (SOR: B). These medications may also be used with levodopa for symptom control and as adjuvant therapy in patients with motor fluctuations.2 A conventional dose of selegiline is 10 mg/d (5 mg at breakfast; 5 mg at lunch). Rasagiline is given at 1 mg/d. Concomitant use of ciprofloxacin or other CYP1A2 inhibitors limits its effectiveness.6,7
Symptomatic therapy is indicated at the onset of functional impairment. The dopamine precursor levodopa is the most widely used and effective drug for Parkinson’s disease symptoms, especially bradykinesia and rigidity. Use the lowest possible dose to control symptoms (eg, 100 mg twice daily) and protect against motor complications of the drug7-9 (SOR: A). To prevent conversion of levodopa to dopamine outside the blood-brain barrier, combine it with the decarboxylase inhibitor carbidopa. Dietary restriction of proteins may be needed, because amino acids can interfere with the absorption of levodopa.
Especially with prolonged use, levodopa can cause disturbing adverse effects, such as nausea, vomiting, psychosis, cardiac arrhythmia, and orthostatic hypotension. Dyskinesias and motor fluctuations are complications of long-term treatment and are irreversible. Adding a cathecol-O-methyltransferase (COMT) inhibitor, such as entacapone, to increase levodopa’s effectiveness has been shown to reduce motor fluctuations2,3,10 (SOR: B). Dopamine agonists such as bromocriptine, ropinirole, and pramipexole used in early Parkinson’s disease can also reduce dyskinesias and motor fluctuations. Dopamine agonists may be preferred to levodopa in early Parkinson’s disease because they are better tolerated and cause fewer adverse effects. Or they may be used as adjuncts for patients whose response to levodopa is deteriorating or fluctuating3,7,8 (SOR: B). In advanced disease, motor complications can also be managed by augmenting levodopa therapy with a dopamine agonist, MAO-B inhibitor, or COMT inhibitor7,8 (SOR: A).
Anticholinergics, mainly benztropine and trihexyphenidyl, may be used as symptomatic treatment, especially in young people with early Parkinson’s disease and severe tremor. However, they are not the first drugs of choice due to limited efficacy and the potential for neuropsychiatric side effects8 (SOR: C). Amantadine can reduce dyskinesia in people with advanced Parkinson’s disease8 (SOR: C). For patients who have Parkinson’s disease with severe motor complications, intermittent apomorphine injections can help reduce “off time” periods in the daily treatment cycle when the efficacy of drugs wanes9 (SOR: B).
Deep brain stimulation of the subthalamic nucleus has only SOR C support for reducing dyskinesias and off time.9
Treating nonmotor symptoms of Parkinson’s disease can be challenging. For dementia in these patients, consider cholinesterase inhibitors6,8 (SOR: C). For depression, selective serotonin reuptake inhibitors are effective6,8,9 (SOR: C). For psychosis, preferred agents are low-dose clozapine or quetiapine6,8-10 (SOR: C). Plan for supportive and symptomatic management of constipation, dysphagia, sialorrhea, orthostatic hypotension, sleep disturbances, and urinary urgency.2,3
Tremor
Tremor is a common form of hyperkinesia, presenting either as a primary disorder or as a symptom of another condition.11 By definition, it is a rhythmical, involuntary, oscillatory movement of 1 or more body parts. Tremors are classified as rest or action tremors, with the latter being further categorized as postural (occurring while the patient maintains a position against gravity) or kinetic (occurring during voluntary movement).2,10
Physiologic tremor: Pharmacologic Tx is usually not needed
Physiologic tremor is benign, high frequency (8-12 Hz), low amplitude, and postural. An exaggerated form of this tremor may result from anxiety, hyperthyroidism, pheochromocytoma, hypoglycemia, excessive caffeine consumption, fever, withdrawal from opioids and sedatives, and some medications. No drug treatment is necessary unless symptoms become bothersome. Correct the underlying cause or have the patient avoid the triggering factor, and offer reassurance that the condition is not pathological or progressive.2,12 For anxiety, consider cognitive-behavioral/relaxation therapy or benzodiazepines (if tremor did not result from withdrawal of benzodiazepines) or beta-adrenergic antagonists (eg, propranolol).12,13
Essential tremor: Try propranolol or primidone first
Essential tremor (ET) is the most common movement disorder. It often results in functional disability and leads to many physical and emotional difficulties. ET is bilateral, usually symmetric (although mild asymmetry is possible), and postural or kinetic, typically affecting hands and forearms. The frequency of ET is 4 to 12 Hz. Cranial musculature may be involved in 30% of cases, affecting the head and voice.3 Prevalence ranges from 4 to 40 cases per 1000 people. The age-adjusted incidence is 17.5/100,000 per year; it peaks during the teen years and the fifth decade.2,3
Autosomal dominant type of inheritance is common, and a family history of ET is often present, particularly with younger patients. The differential diagnosis includes Parkinson’s disease tremor; dystonic, cerebellar, rubral, and psychogenic tremors; and asterixis.3 Unlike ET, many of these disorders have associated neurologic, psychiatric, or systemic signs.
Treatment with propranolol or primidone is indicated if ET causes functional impairment or social or emotional problems for the patient.2,3,10,13 Both propranolol and primidone reduce limb tremor2,10,13 (SOR: B), but only propranolol is approved by the US Food and Drug Administration (FDA) for treatment of ET. Propranolol is more effective for hand and forearm tremor than for head and voice tremor. Start propranolol at 20 to 40 mg twice a day and increase the dose as needed to achieve symptom relief.14
A maintenance dose of 240 to 320 mg/d may be needed. Major adverse effects are fatigue, sedation, depression, and erectile dysfunction. Contraindications to propranolol include asthma, second-degree atrioventricular block, and insulin-dependent diabetes.
If starting with primidone alone, prescribe at a dose <25 mg at bedtime and increase the dose slowly over several weeks to prevent onset of nausea, vomiting, sedation, confusion, or ataxia. The maximum allowable dose is 750 mg/d in 3 divided doses.10 Primidone and propranolol may be used in combination to treat limb tremor when monotherapy is insufficient (SOR: B).13
Thirty percent of patients with ET will not respond to propranolol or primidone. An alternative choice is the anticonvulsant gabapentin10,12-14 (SOR: C). However, clinical experience with it is limited. Lethargy, fatigue, decreased libido, dizziness, nervousness, and shortness of breath are adverse effects of gabapentin; they are usually mild and tolerable.13 Topiramate is another option that seems to be as effective as gabapentin10,13 (SOR: C), but studies of long-term outcomes are lacking. Topiramate’s side effects include weight loss and paresthesias. Additionally, alprazolam, clonazepam, clozapine, olanzapine, atenolol, sotalol, nadolol, and nimodipine may reduce limb tremor2 (SOR: C). Alcohol reduces tremor amplitude in 50% to 90% of patients, but tremor may worsen after the effect of alcohol has worn off.15
For patients with essential hand tremor that fails to respond to oral agents, consider botulinum toxin A16 (SOR: B). However, it is also associated with dose-dependent hand weakness16 (SOR: C). Botulinum toxin may reduce head and voice tremor16 (SOR: C), but hoarseness and swallowing difficulties may occur after use for voice tremor.16
Invasive therapies may benefit patients with refractory tremor. Deep brain stimulation and thalamotomy are highly effective in reducing limb tremor13 (SOR: C). Each carries a small risk of major complications. Some deep brain stimulation adverse events may resolve with time. Other adverse events may resolve with adjustment of stimulator settings. No evidence exists for surgical treatment for voice and head tremor or for gamma-knife thalamotomy.13
Drug-induced tremor
Drugs with the potential to cause postural tremor, intention tremor, or rest tremor include the following: 15
- alcohol (chronic)
- amiodarone
- amphetamines
- beta-adrenergic agonists
- caffeine
- calcitonin
- carbamazepine
- cocaine
- cyclosporine
- dopamine
- lithium
- metoclopramide
- neuroleptics
- procainamide
- steroids
- theophylline
- thyroid hormones
- tricyclic antidepressants
- trifluoperazine
- valproic acid
With drug-induced tremor, carefully evaluate a patient’s need for the drug. Discontinue the offending agent if possible, or try lowering the dose.
Psychogenic tremor: A history of somatization is a clue
Psychogenic tremor can occur at rest or during postural or kinetic movement. Clinical features include an abrupt onset, a static course, spontaneous remission, and unclassifiable tremors.17 Psychogenic tremor increases under direct observation and decreases with distraction. Patients with psychogenic tremor often have a history of somatization.18 Electrophysiologic testing can help confirm the diagnosis. If remission does not occur spontaneously, patients may find relief with psychotherapy or placebo.19
Tic disorders: Opt for dopamine receptor blockers
Tics are involuntary or semivoluntary movements or sounds that are sudden, brief, intermittent, repetitive, nonrhythmic, unpredictable, and purposeless. Tics can occur in any part of the body.20
The most common tic disorder is Tourette syndrome—a combination of motor and phonic tics with onset before age 21. It affects approximately 5 to 10 children out of 10,000. Boys are more commonly affected than girls. Attention deficit hyperactivity disorder frequently accompanies this syndrome.2
The goal of treatment with any tic disorder is to improve social functioning, self-esteem, and quality of life. Education and support of patients is important. Tic disorders, including Tourette, rarely require drugs. But if tics become too distressing, first-line treatment would be a dopamine modulator, tetrabenazine, or clonidine. Randomized controlled trials with various neuroleptics have revealed dramatic reductions in tic severity. However, many patients do not tolerate the acute adverse effects (most commonly sedation, weight gain, depression, lethargy, and akathisia), and prolonged treatment confers a small risk of tardive dyskinesia. Behavioral therapy is an important part of management.20
Dopamine-receptor blocking drugs such as haloperidol, pimozide, and fluphenazine are the most effective treatment for tics20 (SOR: B). Tetrabenazine is a promising new dopamine-depleting drug; controlled trials are ongoing2,20 (SOR: B). Clonidine, an alpha 2-adrenergic agonist, is useful in treating patients with Tourette syndrome, helping to improve sleep and attention2,21 (SOR: C). Medically refractory motor and disabling phonic tics such as coprolalia can be ameliorated by botulinum toxin injections21 (SOR: B). Deep brain stimulation is being used at an increasing rate for medically refractory tics in Tourette syndrome21 (SOR: B).
Restless legs syndrome: Dopamine agonists are preferred
Restless legs syndrome (RLS) is a disorder characterized by sensory symptoms and motor disturbances of the legs, mainly during rest. Treatment may not be necessary for patients with mild or sporadic symptoms. For moderate to severe RLS with significant impairment, dopamine agonists are the preferred agents22 (SOR: A). RLS can also occur secondary to such conditions as iron deficiency and uremia, and correction of the underlying disorder is the goal. Prescribe iron replacement for patients with a ferritin level <50 ng/mL22 (SOR: C). Medications known to cause or exacerbate the symptoms of RLS are anti-dopaminergic agents (such as neuroleptics), diphenhydramine, tricyclic antidepressants, alcohol, caffeine, lithium, and beta-blockers. If a patient is taking medications that exacerbate symptoms of RLS, discontinue them and use appropriate substitutes22 (SOR: C).
Myoclonus: Clonazepam for essential disorder
Myoclonus is a brief, sudden, shock-like movement caused by involuntary muscle contractions or lapse of muscle contraction (asterixis). Given the complex origins of myoclonus, multiple drugs may be needed. Essential myoclonus is disabling and can be treated with clonazepam. Start with 0.25 mg orally twice daily, and increase the dosage over 3 days to 1 mg/d23 (SOR: C). Most cases of myoclonus are secondary due to drugs such as lithium, toxins, advanced liver disease, infections including human immunodeficiency virus, dementia, and brain lesions. Treatment should also address the underlying disorder.2,23
Chorea
Chorea is an abnormal involuntary movement disorder described as “a state of excessive, spontaneous movements, irregularly timed, nonrepetitive, randomly distributed, and abrupt in character.”24
Treatment of chorea is symptomatic, aiming to reduce morbidity and prevent complications. Haloperidol and fluphenazine are effective but can impair voluntary movements2,10,25 (SOR: C). The dopamine-depleting drugs reserpine and tetrabenazine are also effective2,10,25 (SOR: C). GABAergic drugs, such as clonazepam, gabapentin, and valproate, can be used adjunctively.10,25
Dystonia
Dystonia is a syndrome involving sustained contractions of opposing muscles that cause twisting, repetitive movements and abnormal postures. Primary dystonia can be treated successfully with high doses of trihexyphenidyl alone, starting with 1 mg orally per day and increasing gradually to 6 to 80 mg/d until symptoms are controlled; or in combination with baclofen, starting with 10 mg orally once daily and increasing to a maximum dose of 30 to 120 mg/d1,2 (SOR: C).
Consider botulinum neurotoxin injection for focal upper extremity dystonia and adductor spasmodic dysphonia16 (SOR: B).
Ataxia
Ataxia is an unsteady gait associated with cerebellar dysfunction, proprioceptive defects, or both. Ataxia may be primary (Friedreich ataxia and spinocerebellar ataxia) or secondary to stroke, trauma, alcoholic degeneration, multiple sclerosis, vitamin B12 deficiency, and hydrocephalus. Treatment, when possible, should target the underlying cause.1,2
CORRESPONDENCE Hakan Yaman, MD, Akdeniz University, Department of Family Medicine, Antalya, Turkey 07058; [email protected]
• Initiate neuroprotective therapy with a monoamine oxidase B inhibitor to slow the progression of Parkinson’s disease. With onset of functional impairment, give levodopa at the lowest effective dose. A
• Give propranolol for essential tremor causing a patient distress, starting at 20 to 40 mg twice daily and increasing the dose (to a maximum of 320 mg/d) until relief is achieved. B
• Consider giving a dopamine receptor blocker for Tourette syndrome or other tic disorder; alternative agents are clonidine or a newer agent, tetrabenazine. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Movement disorders often require consultation with a neurologist, and a working knowledge of established and novel treatments can set the stage for optimal long-term cooperative management.1 In this article, we review therapeutic options for common movement disorders, including hypokinetic, hyperkinetic, and dyskinetic disturbances.
Parkinson’s disease treatment: MAO-B inhibitor, levodopa are mainstays
Parkinson’s disease, the most common hypokinetic movement disorder, is a chronic, progressive, neurodegenerative disease. It affects 1% of individuals older than 65 years and 4% to 5% of individuals older than 85 years. Its cardinal symptoms are resting tremor, bradykinesia, rigidity, a flexed posture, and loss of postural reflexes. Resting tremor, referred to as “pill rolling” tremor, is 4 to 6 Hz and usually begins unilaterally.2,3 Associated symptoms can include dystonia, dementia, psychiatric disorders, sleep disorders, and autonomic symptoms.
Neuroprotective therapy is used to slow the progression of the disease, particularly in its early stage. The monoamine oxidase B (MAO-B) inhibitor selegiline has proven effective in this regard2 (strength of recommendation [SOR]: A). In randomized controlled studies, selegiline has delayed the need for levodopa for 9 to 12 months4 (SOR: A). Another MAO-B inhibitor, rasagiline, has demonstrated neuroprotective effects as well5 (SOR: B). These medications may also be used with levodopa for symptom control and as adjuvant therapy in patients with motor fluctuations.2 A conventional dose of selegiline is 10 mg/d (5 mg at breakfast; 5 mg at lunch). Rasagiline is given at 1 mg/d. Concomitant use of ciprofloxacin or other CYP1A2 inhibitors limits its effectiveness.6,7
Symptomatic therapy is indicated at the onset of functional impairment. The dopamine precursor levodopa is the most widely used and effective drug for Parkinson’s disease symptoms, especially bradykinesia and rigidity. Use the lowest possible dose to control symptoms (eg, 100 mg twice daily) and protect against motor complications of the drug7-9 (SOR: A). To prevent conversion of levodopa to dopamine outside the blood-brain barrier, combine it with the decarboxylase inhibitor carbidopa. Dietary restriction of proteins may be needed, because amino acids can interfere with the absorption of levodopa.
Especially with prolonged use, levodopa can cause disturbing adverse effects, such as nausea, vomiting, psychosis, cardiac arrhythmia, and orthostatic hypotension. Dyskinesias and motor fluctuations are complications of long-term treatment and are irreversible. Adding a cathecol-O-methyltransferase (COMT) inhibitor, such as entacapone, to increase levodopa’s effectiveness has been shown to reduce motor fluctuations2,3,10 (SOR: B). Dopamine agonists such as bromocriptine, ropinirole, and pramipexole used in early Parkinson’s disease can also reduce dyskinesias and motor fluctuations. Dopamine agonists may be preferred to levodopa in early Parkinson’s disease because they are better tolerated and cause fewer adverse effects. Or they may be used as adjuncts for patients whose response to levodopa is deteriorating or fluctuating3,7,8 (SOR: B). In advanced disease, motor complications can also be managed by augmenting levodopa therapy with a dopamine agonist, MAO-B inhibitor, or COMT inhibitor7,8 (SOR: A).
Anticholinergics, mainly benztropine and trihexyphenidyl, may be used as symptomatic treatment, especially in young people with early Parkinson’s disease and severe tremor. However, they are not the first drugs of choice due to limited efficacy and the potential for neuropsychiatric side effects8 (SOR: C). Amantadine can reduce dyskinesia in people with advanced Parkinson’s disease8 (SOR: C). For patients who have Parkinson’s disease with severe motor complications, intermittent apomorphine injections can help reduce “off time” periods in the daily treatment cycle when the efficacy of drugs wanes9 (SOR: B).
Deep brain stimulation of the subthalamic nucleus has only SOR C support for reducing dyskinesias and off time.9
Treating nonmotor symptoms of Parkinson’s disease can be challenging. For dementia in these patients, consider cholinesterase inhibitors6,8 (SOR: C). For depression, selective serotonin reuptake inhibitors are effective6,8,9 (SOR: C). For psychosis, preferred agents are low-dose clozapine or quetiapine6,8-10 (SOR: C). Plan for supportive and symptomatic management of constipation, dysphagia, sialorrhea, orthostatic hypotension, sleep disturbances, and urinary urgency.2,3
Tremor
Tremor is a common form of hyperkinesia, presenting either as a primary disorder or as a symptom of another condition.11 By definition, it is a rhythmical, involuntary, oscillatory movement of 1 or more body parts. Tremors are classified as rest or action tremors, with the latter being further categorized as postural (occurring while the patient maintains a position against gravity) or kinetic (occurring during voluntary movement).2,10
Physiologic tremor: Pharmacologic Tx is usually not needed
Physiologic tremor is benign, high frequency (8-12 Hz), low amplitude, and postural. An exaggerated form of this tremor may result from anxiety, hyperthyroidism, pheochromocytoma, hypoglycemia, excessive caffeine consumption, fever, withdrawal from opioids and sedatives, and some medications. No drug treatment is necessary unless symptoms become bothersome. Correct the underlying cause or have the patient avoid the triggering factor, and offer reassurance that the condition is not pathological or progressive.2,12 For anxiety, consider cognitive-behavioral/relaxation therapy or benzodiazepines (if tremor did not result from withdrawal of benzodiazepines) or beta-adrenergic antagonists (eg, propranolol).12,13
Essential tremor: Try propranolol or primidone first
Essential tremor (ET) is the most common movement disorder. It often results in functional disability and leads to many physical and emotional difficulties. ET is bilateral, usually symmetric (although mild asymmetry is possible), and postural or kinetic, typically affecting hands and forearms. The frequency of ET is 4 to 12 Hz. Cranial musculature may be involved in 30% of cases, affecting the head and voice.3 Prevalence ranges from 4 to 40 cases per 1000 people. The age-adjusted incidence is 17.5/100,000 per year; it peaks during the teen years and the fifth decade.2,3
Autosomal dominant type of inheritance is common, and a family history of ET is often present, particularly with younger patients. The differential diagnosis includes Parkinson’s disease tremor; dystonic, cerebellar, rubral, and psychogenic tremors; and asterixis.3 Unlike ET, many of these disorders have associated neurologic, psychiatric, or systemic signs.
Treatment with propranolol or primidone is indicated if ET causes functional impairment or social or emotional problems for the patient.2,3,10,13 Both propranolol and primidone reduce limb tremor2,10,13 (SOR: B), but only propranolol is approved by the US Food and Drug Administration (FDA) for treatment of ET. Propranolol is more effective for hand and forearm tremor than for head and voice tremor. Start propranolol at 20 to 40 mg twice a day and increase the dose as needed to achieve symptom relief.14
A maintenance dose of 240 to 320 mg/d may be needed. Major adverse effects are fatigue, sedation, depression, and erectile dysfunction. Contraindications to propranolol include asthma, second-degree atrioventricular block, and insulin-dependent diabetes.
If starting with primidone alone, prescribe at a dose <25 mg at bedtime and increase the dose slowly over several weeks to prevent onset of nausea, vomiting, sedation, confusion, or ataxia. The maximum allowable dose is 750 mg/d in 3 divided doses.10 Primidone and propranolol may be used in combination to treat limb tremor when monotherapy is insufficient (SOR: B).13
Thirty percent of patients with ET will not respond to propranolol or primidone. An alternative choice is the anticonvulsant gabapentin10,12-14 (SOR: C). However, clinical experience with it is limited. Lethargy, fatigue, decreased libido, dizziness, nervousness, and shortness of breath are adverse effects of gabapentin; they are usually mild and tolerable.13 Topiramate is another option that seems to be as effective as gabapentin10,13 (SOR: C), but studies of long-term outcomes are lacking. Topiramate’s side effects include weight loss and paresthesias. Additionally, alprazolam, clonazepam, clozapine, olanzapine, atenolol, sotalol, nadolol, and nimodipine may reduce limb tremor2 (SOR: C). Alcohol reduces tremor amplitude in 50% to 90% of patients, but tremor may worsen after the effect of alcohol has worn off.15
For patients with essential hand tremor that fails to respond to oral agents, consider botulinum toxin A16 (SOR: B). However, it is also associated with dose-dependent hand weakness16 (SOR: C). Botulinum toxin may reduce head and voice tremor16 (SOR: C), but hoarseness and swallowing difficulties may occur after use for voice tremor.16
Invasive therapies may benefit patients with refractory tremor. Deep brain stimulation and thalamotomy are highly effective in reducing limb tremor13 (SOR: C). Each carries a small risk of major complications. Some deep brain stimulation adverse events may resolve with time. Other adverse events may resolve with adjustment of stimulator settings. No evidence exists for surgical treatment for voice and head tremor or for gamma-knife thalamotomy.13
Drug-induced tremor
Drugs with the potential to cause postural tremor, intention tremor, or rest tremor include the following: 15
- alcohol (chronic)
- amiodarone
- amphetamines
- beta-adrenergic agonists
- caffeine
- calcitonin
- carbamazepine
- cocaine
- cyclosporine
- dopamine
- lithium
- metoclopramide
- neuroleptics
- procainamide
- steroids
- theophylline
- thyroid hormones
- tricyclic antidepressants
- trifluoperazine
- valproic acid
With drug-induced tremor, carefully evaluate a patient’s need for the drug. Discontinue the offending agent if possible, or try lowering the dose.
Psychogenic tremor: A history of somatization is a clue
Psychogenic tremor can occur at rest or during postural or kinetic movement. Clinical features include an abrupt onset, a static course, spontaneous remission, and unclassifiable tremors.17 Psychogenic tremor increases under direct observation and decreases with distraction. Patients with psychogenic tremor often have a history of somatization.18 Electrophysiologic testing can help confirm the diagnosis. If remission does not occur spontaneously, patients may find relief with psychotherapy or placebo.19
Tic disorders: Opt for dopamine receptor blockers
Tics are involuntary or semivoluntary movements or sounds that are sudden, brief, intermittent, repetitive, nonrhythmic, unpredictable, and purposeless. Tics can occur in any part of the body.20
The most common tic disorder is Tourette syndrome—a combination of motor and phonic tics with onset before age 21. It affects approximately 5 to 10 children out of 10,000. Boys are more commonly affected than girls. Attention deficit hyperactivity disorder frequently accompanies this syndrome.2
The goal of treatment with any tic disorder is to improve social functioning, self-esteem, and quality of life. Education and support of patients is important. Tic disorders, including Tourette, rarely require drugs. But if tics become too distressing, first-line treatment would be a dopamine modulator, tetrabenazine, or clonidine. Randomized controlled trials with various neuroleptics have revealed dramatic reductions in tic severity. However, many patients do not tolerate the acute adverse effects (most commonly sedation, weight gain, depression, lethargy, and akathisia), and prolonged treatment confers a small risk of tardive dyskinesia. Behavioral therapy is an important part of management.20
Dopamine-receptor blocking drugs such as haloperidol, pimozide, and fluphenazine are the most effective treatment for tics20 (SOR: B). Tetrabenazine is a promising new dopamine-depleting drug; controlled trials are ongoing2,20 (SOR: B). Clonidine, an alpha 2-adrenergic agonist, is useful in treating patients with Tourette syndrome, helping to improve sleep and attention2,21 (SOR: C). Medically refractory motor and disabling phonic tics such as coprolalia can be ameliorated by botulinum toxin injections21 (SOR: B). Deep brain stimulation is being used at an increasing rate for medically refractory tics in Tourette syndrome21 (SOR: B).
Restless legs syndrome: Dopamine agonists are preferred
Restless legs syndrome (RLS) is a disorder characterized by sensory symptoms and motor disturbances of the legs, mainly during rest. Treatment may not be necessary for patients with mild or sporadic symptoms. For moderate to severe RLS with significant impairment, dopamine agonists are the preferred agents22 (SOR: A). RLS can also occur secondary to such conditions as iron deficiency and uremia, and correction of the underlying disorder is the goal. Prescribe iron replacement for patients with a ferritin level <50 ng/mL22 (SOR: C). Medications known to cause or exacerbate the symptoms of RLS are anti-dopaminergic agents (such as neuroleptics), diphenhydramine, tricyclic antidepressants, alcohol, caffeine, lithium, and beta-blockers. If a patient is taking medications that exacerbate symptoms of RLS, discontinue them and use appropriate substitutes22 (SOR: C).
Myoclonus: Clonazepam for essential disorder
Myoclonus is a brief, sudden, shock-like movement caused by involuntary muscle contractions or lapse of muscle contraction (asterixis). Given the complex origins of myoclonus, multiple drugs may be needed. Essential myoclonus is disabling and can be treated with clonazepam. Start with 0.25 mg orally twice daily, and increase the dosage over 3 days to 1 mg/d23 (SOR: C). Most cases of myoclonus are secondary due to drugs such as lithium, toxins, advanced liver disease, infections including human immunodeficiency virus, dementia, and brain lesions. Treatment should also address the underlying disorder.2,23
Chorea
Chorea is an abnormal involuntary movement disorder described as “a state of excessive, spontaneous movements, irregularly timed, nonrepetitive, randomly distributed, and abrupt in character.”24
Treatment of chorea is symptomatic, aiming to reduce morbidity and prevent complications. Haloperidol and fluphenazine are effective but can impair voluntary movements2,10,25 (SOR: C). The dopamine-depleting drugs reserpine and tetrabenazine are also effective2,10,25 (SOR: C). GABAergic drugs, such as clonazepam, gabapentin, and valproate, can be used adjunctively.10,25
Dystonia
Dystonia is a syndrome involving sustained contractions of opposing muscles that cause twisting, repetitive movements and abnormal postures. Primary dystonia can be treated successfully with high doses of trihexyphenidyl alone, starting with 1 mg orally per day and increasing gradually to 6 to 80 mg/d until symptoms are controlled; or in combination with baclofen, starting with 10 mg orally once daily and increasing to a maximum dose of 30 to 120 mg/d1,2 (SOR: C).
Consider botulinum neurotoxin injection for focal upper extremity dystonia and adductor spasmodic dysphonia16 (SOR: B).
Ataxia
Ataxia is an unsteady gait associated with cerebellar dysfunction, proprioceptive defects, or both. Ataxia may be primary (Friedreich ataxia and spinocerebellar ataxia) or secondary to stroke, trauma, alcoholic degeneration, multiple sclerosis, vitamin B12 deficiency, and hydrocephalus. Treatment, when possible, should target the underlying cause.1,2
CORRESPONDENCE Hakan Yaman, MD, Akdeniz University, Department of Family Medicine, Antalya, Turkey 07058; [email protected]
1. Deuschl G, Bain P, Brin M. Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee. Mov Disord. 1998;13(suppl 3):2-23.
2. Yaman A, Yaman H, Rao G. Tremors and other movement disorders. In: Mengel MB, et al, eds. Family Medicine Ambulatory Care and Prevention. 5th ed. New York: McGraw-Hill; 2009:400–407.
3. Harris MK, Shneyder N, Borazanci A, et al. Movement disorders. Med Clin North Am. 2009;93:371-388.
4. Palhagen S, Heinonen E, Hagglund J, et al. Selegiline slows the progression of the symptoms of Parkinson disease. Neurology. 2006;66:1200-1206.
5. Olanow CW, Rascol O, Hauser R, et al. A double-blind, delayed-start trial of rasagiline in Parkinson’s disease. N Engl J Med. 2009;361:1268-1278.
6. Zesiewicz TA, Sullivan KL, Arnulf I, et al. Treatment of nonmotor symptoms of Parkinson disease. Neurology. 2010;74:924-931.
7. Suchowersky O, Reich S, Perlmutter J, et al. Practice parameter: diagnosis and prognosis of new onset Parkinson disease (an evidence-based review). Neurology. 2006;66:968-975.
8. Rao SS, Hofmann LA, Shakil A. Parkinson’s disease: diagnosis and treatment. Am Fam Physician. 2006;74:2046-2054.
9. The National Collaborating Centre for Chronic Conditions Parkinson’s Disease. National Clinical Guideline for Diagnosis and Management in Primary and Secondary Care. 2006. Available at: http://www.nice.org.uk/nicemedia/live/10984/30087/30087.pdf. Accessed May 12, 2010.
10. Jankovic J. Treatment of hyperkinetic movement disorders. Lancet Neurol. 2009;8:844-856.
11. Kerlsberg G, Rubenstein C, St Anna L, et al. Differential diagnosis of tremor. Am Fam Physician. 2008;77:1305-1306.
12. Burke DA, Hauser RA, McClain T. Essential tremor. Available at: http://emedicine.medscape.com/article/1150290-overview. Accessed May 12, 2010.
13. Zesiewicz TA, Elble R, Louis ED, et al. Practice parameter: therapies for essential tremor. Neurology. 2005;64:2008-2020.
14. Elble RJ. Tremor: clinical features, pathophysiology, and treatment. Neurol Clin. 2009;27:679-695.
15. Smaga S. Tremor. Am Fam Physician. 2003;68:1545-1552.
16. Use of botulinum neurotoxin for the treatment of movement disorders. AAN summary of evidence-based guidelines for clinicians. 2008. Available at: http://www.aan.com/practice/guideline/uploads/280.pdf. Accessed May 25, 2010.
17. Redondo L, Morgado Y, Durán E. Psychogenic tremor: a positive diagnosis [ in Spanish]. Neurología. 2010;25:51-57.
18. Schwingenschuh P, Katschnig P, Seiler S, et al. Moving toward ‘‘laboratory-supported’’ criteria for psychogenic tremor. Mov Disord. 2011;Sep 28. [Epub ahead of print].
19. McKeon A, Ahlskog JE, Bower JH, et al. Psychogenic tremor: long-term prognosis in patients with electrophysiologically confirmed disease. Mov Disord. 2009;24:72-76.
20. Shprecher D, Kurlan R. The management of tics. Mov Disord. 2009;24:15-24.
21. Kenney C, Kuo SH, Jimenez-Shahed J. Tourette’s syndrome. Am Fam Physician. 2008;77:651-658.
22. Bayard M, Avonda T, Wadzinsky J. Restless legs syndrome. Am Fam Physician. 2008;78:235-240.
23. Caviness JN. Pathophysiology and treatment of myoclonus. Neurol Clin. 2009;27:757-777.
24. Barbeau A, Duvoisin RC, Gerstenbrand F, et al. Classification of extrapyramidal disorders. J Neurol Sci. 1981;51:311-327.
25. Vertrees SM, Berman SA. Chorea in adults: treatment & management. Available at: http://emedicine.medscape.com/article/1149854-treatment. Accessed February 12, 2010.
1. Deuschl G, Bain P, Brin M. Consensus statement of the Movement Disorder Society on Tremor. Ad Hoc Scientific Committee. Mov Disord. 1998;13(suppl 3):2-23.
2. Yaman A, Yaman H, Rao G. Tremors and other movement disorders. In: Mengel MB, et al, eds. Family Medicine Ambulatory Care and Prevention. 5th ed. New York: McGraw-Hill; 2009:400–407.
3. Harris MK, Shneyder N, Borazanci A, et al. Movement disorders. Med Clin North Am. 2009;93:371-388.
4. Palhagen S, Heinonen E, Hagglund J, et al. Selegiline slows the progression of the symptoms of Parkinson disease. Neurology. 2006;66:1200-1206.
5. Olanow CW, Rascol O, Hauser R, et al. A double-blind, delayed-start trial of rasagiline in Parkinson’s disease. N Engl J Med. 2009;361:1268-1278.
6. Zesiewicz TA, Sullivan KL, Arnulf I, et al. Treatment of nonmotor symptoms of Parkinson disease. Neurology. 2010;74:924-931.
7. Suchowersky O, Reich S, Perlmutter J, et al. Practice parameter: diagnosis and prognosis of new onset Parkinson disease (an evidence-based review). Neurology. 2006;66:968-975.
8. Rao SS, Hofmann LA, Shakil A. Parkinson’s disease: diagnosis and treatment. Am Fam Physician. 2006;74:2046-2054.
9. The National Collaborating Centre for Chronic Conditions Parkinson’s Disease. National Clinical Guideline for Diagnosis and Management in Primary and Secondary Care. 2006. Available at: http://www.nice.org.uk/nicemedia/live/10984/30087/30087.pdf. Accessed May 12, 2010.
10. Jankovic J. Treatment of hyperkinetic movement disorders. Lancet Neurol. 2009;8:844-856.
11. Kerlsberg G, Rubenstein C, St Anna L, et al. Differential diagnosis of tremor. Am Fam Physician. 2008;77:1305-1306.
12. Burke DA, Hauser RA, McClain T. Essential tremor. Available at: http://emedicine.medscape.com/article/1150290-overview. Accessed May 12, 2010.
13. Zesiewicz TA, Elble R, Louis ED, et al. Practice parameter: therapies for essential tremor. Neurology. 2005;64:2008-2020.
14. Elble RJ. Tremor: clinical features, pathophysiology, and treatment. Neurol Clin. 2009;27:679-695.
15. Smaga S. Tremor. Am Fam Physician. 2003;68:1545-1552.
16. Use of botulinum neurotoxin for the treatment of movement disorders. AAN summary of evidence-based guidelines for clinicians. 2008. Available at: http://www.aan.com/practice/guideline/uploads/280.pdf. Accessed May 25, 2010.
17. Redondo L, Morgado Y, Durán E. Psychogenic tremor: a positive diagnosis [ in Spanish]. Neurología. 2010;25:51-57.
18. Schwingenschuh P, Katschnig P, Seiler S, et al. Moving toward ‘‘laboratory-supported’’ criteria for psychogenic tremor. Mov Disord. 2011;Sep 28. [Epub ahead of print].
19. McKeon A, Ahlskog JE, Bower JH, et al. Psychogenic tremor: long-term prognosis in patients with electrophysiologically confirmed disease. Mov Disord. 2009;24:72-76.
20. Shprecher D, Kurlan R. The management of tics. Mov Disord. 2009;24:15-24.
21. Kenney C, Kuo SH, Jimenez-Shahed J. Tourette’s syndrome. Am Fam Physician. 2008;77:651-658.
22. Bayard M, Avonda T, Wadzinsky J. Restless legs syndrome. Am Fam Physician. 2008;78:235-240.
23. Caviness JN. Pathophysiology and treatment of myoclonus. Neurol Clin. 2009;27:757-777.
24. Barbeau A, Duvoisin RC, Gerstenbrand F, et al. Classification of extrapyramidal disorders. J Neurol Sci. 1981;51:311-327.
25. Vertrees SM, Berman SA. Chorea in adults: treatment & management. Available at: http://emedicine.medscape.com/article/1149854-treatment. Accessed February 12, 2010.
Inhalation therapy: Help patients avoid these mistakes
• Stress the importance of exhaling gently for a few seconds before inhaling (deeply and slowly for a metered dose inhaler, and deeply and rapidly for most dry powder inhalers). C
• Observe the inhaler technique of every patient receiving inhalation therapy on more than one occasion. C
• Don’t rely on self-reports regarding inhaler technique; despite claims of proficiency, most patients make at least one mistake. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
For patients with asthma or chronic obstructive pulmonary disease (COPD), inhalation therapy is the foundation of treatment. Yet all too often, patients don’t get the full value of their inhaled medications because they use their inhaler incorrectly. When technique is markedly flawed, suboptimal outcomes typically result.
Given the number of Americans with asthma (at least 22 million)1 and COPD (more than 13 million adults),2 faulty inhaler technique is a major public health problem. In fact, the number of people suffering from COPD may be even larger: Close to 24 million US adults are believed to have impaired lung function.3,4 For patients with asthma or COPD—many of whom are treated by family physicians—comprehensive education with a focus on correct use of an inhaler is essential.
In this review, we present evidence of frequent inhaler errors (from clinical studies) and highlight some of the more common mistakes (based on our clinical experience [TABLE]5). Finally, we offer ‘‘time-efficient’’ solutions to inhaler problems—steps that physicians in busy primary care practices can take to ensure that patients with asthma or COPD get the maximum benefit from inhalation therapy.
TABLE
Caution patients about these device-specific mistakes*
| Metered dose inhaler |
|---|
|
| Metered dose inhaler plus spacer/VHC |
|
| Dry powder inhaler |
|
| *These are examples based on the experience of the authors; other errors are possible. †Timing is not as crucial as it is for an MDI without a spacer, but the drug is still lost if inhalation is delayed. ‡Correct use varies by type of product (see product literature for specifics). DPI, dry powder inhaler; MDI, metered dose inhaler; VHC, valved holding chamber. Source: Adapted with permission from Self TH, et al. Consultant. 2003.5 |
Inhaler error is well documented
Since 1965, when it was first reported that many patients used metered dose inhalers (MDIs) incorrectly,6 evidence has accumulated supporting the magnitude of the problem.7-12 (Studies conducted in family practice settings are described in “Researchers look at inhaler problems in primary care” and in TABLE W1.13-20)
A number of studies of various sizes (from 41 to 3955 patients) have assessed inhaler technique in patients being treated by clinicians in primary care. The researchers used a variety of scoring methods, as well. Among them were a simple 4-step (0-4) rating system, a 9-step system, a standardized inhaler-specific checklist, and a system that tracked the number of omissions patients made.13-20 All found significant problems with inhaler technique. (You’ll find a detailed look at the studies in TABLE W1 at jfponline.com.)
In one study of 422 patients,13 including young children, adolescents, and adults, participants received one point for correctly performing each of the following steps:
- Adequate preparation (shaking well for those using a metered dose inhaler [MDI]; loading correctly for patients using a dry powder inhaler [DPI])
- Adequate expiration, correct head position
- Adequate inspiratory technique
- Holding breath afterwards.
The researchers found that 25% of the patients had inadequate technique (≤2 on a 0-4 point scale). In this study, as in others that included patients using various types of devices, use of an MDI was associated with a higher rate of incorrect technique.
Another much-smaller study14 used the same 4-step system to assess the technique of 50 patients, all of whom had the same type of DPI and had received extensive training in the correct use of the device. Despite the training, 27% of the patients received scores of ≤2 (inadequate technique). Sixty-eight percent received a score of 3 (adequate); only 5% received a score of 4 (good).
The 2 largest studies—one including 3955 patients using MDIs20 and the other looking at 3811 patients using various kinds of devices18—found high levels of errors, as well. In the latter study, 76% of patients with MDIs made at least one error vs 49% to 55% of patients using DPIs.18 The results convinced a large majority of the physicians caring for these patients of the need to check inhaler technique more frequently. In the study of MDI users alone, 71% of the patients made at least one mistake.20 inhaler misuse was associated with higher asthma instability scores, this study showed.
More recently, a researcher assessed the effects of an integrated primary care model on the management of asthma and/or COPD in middle-aged and elderly patients, in a study of 260 patients in 44 family practices.19 The study included an evaluation of inhaler technique.
Participants were divided into an intervention group—137 patients who received education regarding inhaler use from a nurse—and a usual care group (123 patients). After 2 years, correct inhaler technique among those in the intervention group went from 41% at baseline to 54%. At the same time, the proportion of those in the usual care group with correct technique fell from 47% to 29%.19
Error rates vary widely from one clinical trial to another, depending on study criteria, type of device, and extent of patient education, among other factors. Nonetheless, several studies (spanning 3 decades) found the error rate to be close to, or greater than, 90%.7,10,21
The most recent of these, published in 2009,21 was based on observation of the inhaler technique used by patients with asthma or COPD directly following appointments in an outpatient clinic. The authors found that, although >98% of the study participants claimed to know how to use their inhalers, 94% committed at least one error. In this study and a number of others, user error was more likely in patients using MDIs.13,18,21,22
Adding a spacer (eg, a valved holding chamber such as the AeroChamber) can be helpful, as the spacer affords the patient more time to inhale the medication. But patients who use an MDI with a spacer often make mistakes, too, and patient education is essential.23-26
Breath-activated dry powder inhalers (DPIs)—such as the Flexhaler, HandiHaler, Aerolizer, and Diskus—also reduce the likelihood of error. DPIs eliminate a step that MDI users often struggle with: the need to simultaneously press down on the canister and begin a slow, deep inhalation.
What’s more, DPIs do not have to be shaken before use. Nonetheless, using a DPI still involves a series of actions. For the HandiHaler and Aerolizer, patients must load the dose, and some patients fail to read the directions and swallow the capsule instead of loading it into the device. Patients must remember to exhale away from the device (ie, not into the dry powder) before inhaling, then hold their breath for approximately 10 seconds. There is potential for error at each step.
Stress the need to exhale before using the inhaler
Forgetting to exhale before inhaling is a common, and significant, mistake regardless of the type of device. It is paramount to stress the need to exhale gently for a few seconds before inhaling (slowly and deeply for patients using an MDI, rapidly and deeply with most DPIs). For MDI users, poor timing, described earlier, is another common and serious mistake. Patients using an MDI with a valved holding chamber sometimes inhale for too long before pressing down on the inhaler, then are unable to continue inhaling although the aerosol is still in the chamber. A common error made by patients using multidose DPIs is simply to forget to load the dose.
Physicians need to brush up on their skills, too
It’s not just patients who lack proficiency in inhaler technique. Numerous studies have demonstrated poor skill among physicians and other health care professionals.27-34 Evidence also shows that targeted education results in substantial improvement.32,35
In one study undertaken to evaluate family medicine residents’ proficiency in using asthma inhalers, participants (an intervention group at one clinic and a control group at another) all were given a pretest. The intervention group then received educational materials and a tutorial, as well as the opportunity for hands-on practice, after which both groups were given a post-test. The residents who received the training had a 170% jump, on average, in proficiency score, vs a 55% increase for the control group (P<.001).35
Inhaled Medication Instructional Videos
Courtesy of: National Jewish Health
Go to http://www.nationaljewish.org/healthinfo/medications/lung-diseases/devices/instructional-videos
Another study—this one involving first-year interns—looked at level of improvement based on the type of education provided. Initially, only 5% of the interns could use an MDI without error. After a lecture and demonstration, 13% had an error-free technique. But when each intern participated in an intensive one-on-one session, the error-free rate reached 73%. The researchers’ conclusion: Lectures are relatively ineffective in teaching interns inhaler technique compared with a one-on-one approach.32
The Chicago Breathe Project,36 a new program aimed at improving education in the use of asthma inhalers for physicians and minority patients, provides further evidence of the value of clinician education. After a series of workshops for residents at 5 academic institutions, the physicians’ knowledge of proper use of inhalers rose dramatically—from just 5% preprogram to 91% postprogram (P<.001). Six months after the educational activity, the residents (n=161) were more likely (44% vs 11% preprogram) to assess patients’ inhaler technique.36
Teaching patients when time is tight
National and international guidelines stress the need to teach patients correct use of asthma and COPD inhalers.1,37,38 Providing the requisite education includes observation of each patient’s inhaler technique with proper use demonstrated, as needed.
The problem, of course, is how to provide that level of patient education within the time constraints of a busy family practice. We recommend these time-efficient solutions:
Enlist the help of other clinicians. While it is important that someone in your office be well trained and able to instruct patients in the proper use of inhalers, that individual need not be you. The National Institutes of Health recommends that the “principal clinician” introduce key educational messages, which can be reinforced and expanded on by other members of the health care team.1
After you advise patients that it is crucial for them to be trained in and adhere to proper inhaler technique, another health care professional—often a clinic nurse or pharmacist who has had special training—can provide the hands-on education. Studies have shown that when pharmacists who are competent in asthma management, including inhaler technique, work with physicians to optimize the education and overall management of patients with asthma, better outcomes often result, including a reduction in both emergency department visits and hospitalizations.1,39,40
Use videos to demonstrate correct technique. Videos are an effective teaching tool,9 and many of them are device-specific. National Jewish Health, which is world renowned for its asthma care, has a set of instructional videos posted on You-Tube and accessible from its Web site (http://www.nationaljewish.org/healthinfo/medications/lung-diseases/devices/instructional-videos). In addition to videos that demonstrate the use of an MDI alone and an MDI plus a valved holding chamber, the site has links to 6 DPI videos, each covering a different device.
Use intermittent observation. After the patient views the appropriate video, you or a member of your staff will still need to observe the patient’s inhaler technique to ensure correct use. Ideally, this should occur at every visit.1,37 When that’s not possible, use intermittent observation, starting with the first 2 or 3 visits after the introduction of inhalation therapy and then switching to periodic observation to ensure that the patient is maintaining good technique.
In determining how often observation is necessary, keep in mind that simply asking patients whether they are having inhaler problems is not sufficient.1 Patients tend to say they have little or no trouble when, in fact, most struggle, at times, with the devices. What’s more, good technique tends to decrease over time, and repetitive education is important.
To motivate patients, try this communication technique
Motivational interviewing, a technique that has been used to help patients battle obesity, quit smoking, and control hypertension,41-43 among other health problems, can help you identify inhaler problems that need to be addressed. It involves the use of open-ended questions (eg, “What worries you most about your asthma?”), affirmations (“You’ve done a great job testing your peak flow level every morning”), reflective listening (“You’re tired of taking medicine every day”), and summary statements (“You know you should take your medicine every day but you’re having trouble remembering it. Is that right?”).
A pilot study44 showed that when this technique was incorporated into an asthma education session, patient motivation increased. The ratio of perceived advantages vs disadvantages of taking asthma medication correctly improved, as well. Another study45 found that when motivational interviewing was used during home visits to inner-city African American adolescents for asthma care, the patients’ motivation, readiness to adhere to treatment, and asthma-related quality of life improved, although self-reported adherence to asthma medication did not. Further studies involving patients with asthma are under way (www.clinicaltrials.gov/ct2/results?term=asthma).
It is important to note that the use of motivational interviewing does not require a lengthy visit. One study found that on average, visits in which primary care physicians used this communication technique lasted less than 10 minutes.46
CORRESPONDENCE Timothy H. Self, PharmD, University of Tennessee Health Science Center, 881 Madison Avenue, Room 235, Memphis, TN 38163; [email protected]
1. National Heart, Lung, and Blood Institute; National Asthma Education and Prevention Program Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma. Bethesda, MD: National Institutes of Health; 2007.
2. Centers for Disease Control and Prevention. National Center for Health Statistics: National health interview survey raw data, 2008. Analysis performed by American Lung Association Research and Program Services.
3. American Lung Association. COPD—Helping the missing millions. February 24, 2010. Available at: http://www.lungusa.org/about-us/our-impact/top-stories/copd-helping-the-missing.html. Accessed November 9, 2011.
4. Centers for Disease Control and Prevention. Chronic obstructive pulmonary disease surveillance—United States, 1971-2000. MMWR Surveill Summ. 2002;51(6):1-16.
5. Self TH, Kilgore KE, Shelton V. MDIs, spacers, and dry powder inhalers: what patients are likely to do wrong. Consultant. 2003;49:702-705.
6. Saunders KB. Misuse of inhaled bronchodilator agents. Br Med J. 1965;1:1037-1038.
7. Epstein SW, Manning CPR, Ashley MJ, et al. Survey of the clinical uses of pressurized aerosol inhalers. Can Med Assoc J. 1979;120:813-816.
8. Shim C, Williams MH. The adequacy of inhalation of aerosol from canister nebulizers. Am J Med. 1980;69:891-894.
9. Self TH, Brooks JB, Lieberman P, et al. The value of demonstration and role of the pharmacist in teaching the correct use of pressurized bronchodilators. Can Med Assoc J. 1983;128:129-131.
10. Hartert TV, Windom HH, Peeples RS, et al. Inadequate outpatient medical therapy for patients with asthma admitted to two urban hospitals. Am J Med. 1996;100:386-394.
11. Goodman DE, Israel E, Rosenberg M, et al. The influence of age, diagnosis, and gender on proper use of metered-dose inhalers. Am J Respir Crit Care Med. 1994;150:1256-1261.
12. Newman SP, Pavia D, Clarke SW. How should a pressurized beta-adrenergic bronchodilator be inhaled? Eur J Respir Dis. 1981;62:3-21.
13. Hilton S. An audit of inhaler technique among asthma patients of 34 general practitioners. Br J Gen Pract. 1990;40:505-506.
14. Dompeling E, Van Grunsven PM, Van Schayck GP, et al. Treatment with inhaled steroids in asthma and chronic bronchitis: long-term compliance and inhaler technique. Fam Pract. 1992;9:161-166.
15. Verver S, Poelman M, Bogels A, et al. Effects of instruction by practice assistants on inhaler technique and respiratory symptoms of patients. A controlled randomized videotaped intervention study. Fam Pract. 1996;13:35-40.
16. Dickinson J, Hutton S, Atkin A, et al. Reducing asthma morbidity in the community: the effect of a targeted nurse-run asthma clinic in an English general practice. Respir Med. 1997;91:634-640.
17. Hesselink AE, Penninx BW, Wijnhoven HA, et al. Determinants of an incorrect inhalation technique in patients with asthma or COPD. Scand J Prim Health Care. 2001;19:255-260.
18. Molimard M, Raherison C, Lignot S, et al. Assessment of handling of inhaler devices in real life: An observational study in 3811 patients in primary care. J Aerosol Med. 2003;16:249-254.
19. Meulepas MA, Jacobs JE, Smeenk FW, et al. Effect of an integrated primary care model on the management of middle-aged and old patients with obstructive lung diseases. Scand J Prim Health Care. 2007;25:186-192.
20. Giraud V, Roche N. Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability. Eur Respir J. 2002;19:246-251.
21. Souza ML, Meneghini AC, Ferraz E, et al. Knowledge of and technique for using inhalation devices among asthma patients and COPD patients. J Bras Pneumol. 2009;35:824-831.
22. Rootmensen GN, van Keimpema AR, Jansen HM, et al. Predictors of incorrect inhalation technique in patients with asthma or COPD: a study using a validated videotaped scoring method. J Aerosol Med Pulm Drug Deliv. 2010;23:323-328.
23. Rachelefsky GS, Rohr AS, Wo J, et al. Use of a tube spacer to improve the efficacy of a metered dose inhaler in asthmatic children. Am J Dis Child. 1986;140:1191-1193.
24. Demirkan K, Tolley E, Mastin T, et al. Salmeterol administration by metered-dose inhaler alone vs metered-dose inhaler plus valved holding chamber. Chest. 2000;117:1314-1318.
25. Pedersen S, Ostergaard PA. Nasal inhalation as a cause of inefficient pulmonal aerosol inhalation technique in children. Allergy. 1983;38:191-194.
26. Dolovich MD, Ahrens RS, Hess DR, et al. Device selection an outcomes of aerosol therapy: evidence-based guidelines: American College of Chest Physicians/American College of Asthma, Allergy, and Immunology. Chest. 2005;127:335-371.
27. Interiano B, Guntupalli KK. Metered-dose inhalers: do health care providers know what to teach? Arch Intern Med. 1993;153:81-85.
28. Hanania NA, Wittman R, Kesten S, et al. Medical personnel’s knowledge of and ability to use inhaling devices. Metered-dose inhalers, spacing chambers, and breath-actuated dry powder inhalers. Chest. 1994;105:111-116.
29. Amirav I, Goren A, Pawlowski NA. What do pediatricians in training know about the correct use of inhalers and spacer devices? J Allergy Clin Immunol. 1994;94:669-675.
30. Chopra N, Oprescu N, Fask A, et al. Does introduction of new “easy to use” inhalational devices improve medical personnel’s knowledge of their proper use? Ann Allergy Asthma Immunol. 2002;88:395-400.
31. Self TH, Arnold LB, Czosnowski LM, et al. Inadequate skill of healthcare professionals in using asthma inhalation devices. J Asthma. 2007;44:593-598.
32. Lee-Wong M, Mayo PH. Results of a programme to improve house staff use of metered dose inhalers and spacers. Postgrad Med J. 2003;79:221-225.
33. Muchao FP, Pern SL, Rodriques JC, et al. Evaluation of the knowledge of health professionals at a pediatric hospital regarding the use of metered dose inhalers. J Bras Pneumol. 2008;34:4-12.
34. Kim SH, Kwak HJ, Kim TB, et al. Inappropriate techniques used by internal medicine residents with three kinds of inhalers (a metered dose inhaler, Diskus, and Turbuhaler): changes after a single teaching session. J Asthma. 2009;46:944-950.
35. Kelcher S, Brownoff R. Teaching residents to use asthma devices. Assessing family residents’ skill and a brief intervention. Can Fam Physician. 1994;40:2090-2095.
36. Press VG, Pincayage AT, Pappalardo AA, et al. The Chicago Breathe Project: a regional approach to improving education on asthma inhalers for resident physicians and minority patients. J Natl Med Assoc. 2010;102:548-555.
37. Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention. 2010. Available at: www.ginasthma.org. Accessed November 9, 2011.
38. Executive Summary: global strategy on the diagnosis and management and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Workshop Report, 2009. Available at: www.goldcopd.com. Accessed November 9, 2011.
39. Self TH, Chrisman CR, Mason DL, et al. Reducing emergency department visits and hospitalizations in African American and Hispanic patients: a 15-year review. J Asthma. 2005;42:807-812.
40. Armour C, Bosnic-Anticevich S, Brillant M, et al. Pharmacy asthma care program (PACP) improves outcomes for patients in the community. Thorax. 2007;62:496-502.
41. DiLillo V, Nicole J, West DS. Incorporating motivational interviewing into behavioral obesity treatment. Cogn Behav Pract. 2003;10:120-130.
42. Borrelli B, Novak S, Hecht J, et al. Home health care nurses as a new channel for smoking cessation treatment: outcomes from project CARES (Community-nurse Assisted Research and Education on Smoking). Prev Med. 2005;41:815-821.
43. Woollard L, Beilin L, Lord T, et al. A controlled trial of nurse counselling on lifestyle change for hypertensives treated in general practice: preliminary results. Clin Exp Pharmacol Physiol. 1995;22:466-468.
44. Schmaling K, Blume A, Afari N. A randomized controlled pilot study of motivational interviewing to change attitudes about adherence to medications for asthma. J Clin Psych Med Settings. 2001;8:167-172.
45. Riekert KA, Borrelli B, Bilderback A, et al. The development of a motivational interviewing intervention to promote medication adherence among inner-city, African-American adolescents with asthma. Patient Educ Couns. 2011;82:117-122.
46. Butler C, Rollnick S, Cohen D, et al. Motivational consulting versus brief advice for smokers in general practice: a randomized trial. Br J Gen Pract. 1999;49:611-616.
• Stress the importance of exhaling gently for a few seconds before inhaling (deeply and slowly for a metered dose inhaler, and deeply and rapidly for most dry powder inhalers). C
• Observe the inhaler technique of every patient receiving inhalation therapy on more than one occasion. C
• Don’t rely on self-reports regarding inhaler technique; despite claims of proficiency, most patients make at least one mistake. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
For patients with asthma or chronic obstructive pulmonary disease (COPD), inhalation therapy is the foundation of treatment. Yet all too often, patients don’t get the full value of their inhaled medications because they use their inhaler incorrectly. When technique is markedly flawed, suboptimal outcomes typically result.
Given the number of Americans with asthma (at least 22 million)1 and COPD (more than 13 million adults),2 faulty inhaler technique is a major public health problem. In fact, the number of people suffering from COPD may be even larger: Close to 24 million US adults are believed to have impaired lung function.3,4 For patients with asthma or COPD—many of whom are treated by family physicians—comprehensive education with a focus on correct use of an inhaler is essential.
In this review, we present evidence of frequent inhaler errors (from clinical studies) and highlight some of the more common mistakes (based on our clinical experience [TABLE]5). Finally, we offer ‘‘time-efficient’’ solutions to inhaler problems—steps that physicians in busy primary care practices can take to ensure that patients with asthma or COPD get the maximum benefit from inhalation therapy.
TABLE
Caution patients about these device-specific mistakes*
| Metered dose inhaler |
|---|
|
| Metered dose inhaler plus spacer/VHC |
|
| Dry powder inhaler |
|
| *These are examples based on the experience of the authors; other errors are possible. †Timing is not as crucial as it is for an MDI without a spacer, but the drug is still lost if inhalation is delayed. ‡Correct use varies by type of product (see product literature for specifics). DPI, dry powder inhaler; MDI, metered dose inhaler; VHC, valved holding chamber. Source: Adapted with permission from Self TH, et al. Consultant. 2003.5 |
Inhaler error is well documented
Since 1965, when it was first reported that many patients used metered dose inhalers (MDIs) incorrectly,6 evidence has accumulated supporting the magnitude of the problem.7-12 (Studies conducted in family practice settings are described in “Researchers look at inhaler problems in primary care” and in TABLE W1.13-20)
A number of studies of various sizes (from 41 to 3955 patients) have assessed inhaler technique in patients being treated by clinicians in primary care. The researchers used a variety of scoring methods, as well. Among them were a simple 4-step (0-4) rating system, a 9-step system, a standardized inhaler-specific checklist, and a system that tracked the number of omissions patients made.13-20 All found significant problems with inhaler technique. (You’ll find a detailed look at the studies in TABLE W1 at jfponline.com.)
In one study of 422 patients,13 including young children, adolescents, and adults, participants received one point for correctly performing each of the following steps:
- Adequate preparation (shaking well for those using a metered dose inhaler [MDI]; loading correctly for patients using a dry powder inhaler [DPI])
- Adequate expiration, correct head position
- Adequate inspiratory technique
- Holding breath afterwards.
The researchers found that 25% of the patients had inadequate technique (≤2 on a 0-4 point scale). In this study, as in others that included patients using various types of devices, use of an MDI was associated with a higher rate of incorrect technique.
Another much-smaller study14 used the same 4-step system to assess the technique of 50 patients, all of whom had the same type of DPI and had received extensive training in the correct use of the device. Despite the training, 27% of the patients received scores of ≤2 (inadequate technique). Sixty-eight percent received a score of 3 (adequate); only 5% received a score of 4 (good).
The 2 largest studies—one including 3955 patients using MDIs20 and the other looking at 3811 patients using various kinds of devices18—found high levels of errors, as well. In the latter study, 76% of patients with MDIs made at least one error vs 49% to 55% of patients using DPIs.18 The results convinced a large majority of the physicians caring for these patients of the need to check inhaler technique more frequently. In the study of MDI users alone, 71% of the patients made at least one mistake.20 inhaler misuse was associated with higher asthma instability scores, this study showed.
More recently, a researcher assessed the effects of an integrated primary care model on the management of asthma and/or COPD in middle-aged and elderly patients, in a study of 260 patients in 44 family practices.19 The study included an evaluation of inhaler technique.
Participants were divided into an intervention group—137 patients who received education regarding inhaler use from a nurse—and a usual care group (123 patients). After 2 years, correct inhaler technique among those in the intervention group went from 41% at baseline to 54%. At the same time, the proportion of those in the usual care group with correct technique fell from 47% to 29%.19
Error rates vary widely from one clinical trial to another, depending on study criteria, type of device, and extent of patient education, among other factors. Nonetheless, several studies (spanning 3 decades) found the error rate to be close to, or greater than, 90%.7,10,21
The most recent of these, published in 2009,21 was based on observation of the inhaler technique used by patients with asthma or COPD directly following appointments in an outpatient clinic. The authors found that, although >98% of the study participants claimed to know how to use their inhalers, 94% committed at least one error. In this study and a number of others, user error was more likely in patients using MDIs.13,18,21,22
Adding a spacer (eg, a valved holding chamber such as the AeroChamber) can be helpful, as the spacer affords the patient more time to inhale the medication. But patients who use an MDI with a spacer often make mistakes, too, and patient education is essential.23-26
Breath-activated dry powder inhalers (DPIs)—such as the Flexhaler, HandiHaler, Aerolizer, and Diskus—also reduce the likelihood of error. DPIs eliminate a step that MDI users often struggle with: the need to simultaneously press down on the canister and begin a slow, deep inhalation.
What’s more, DPIs do not have to be shaken before use. Nonetheless, using a DPI still involves a series of actions. For the HandiHaler and Aerolizer, patients must load the dose, and some patients fail to read the directions and swallow the capsule instead of loading it into the device. Patients must remember to exhale away from the device (ie, not into the dry powder) before inhaling, then hold their breath for approximately 10 seconds. There is potential for error at each step.
Stress the need to exhale before using the inhaler
Forgetting to exhale before inhaling is a common, and significant, mistake regardless of the type of device. It is paramount to stress the need to exhale gently for a few seconds before inhaling (slowly and deeply for patients using an MDI, rapidly and deeply with most DPIs). For MDI users, poor timing, described earlier, is another common and serious mistake. Patients using an MDI with a valved holding chamber sometimes inhale for too long before pressing down on the inhaler, then are unable to continue inhaling although the aerosol is still in the chamber. A common error made by patients using multidose DPIs is simply to forget to load the dose.
Physicians need to brush up on their skills, too
It’s not just patients who lack proficiency in inhaler technique. Numerous studies have demonstrated poor skill among physicians and other health care professionals.27-34 Evidence also shows that targeted education results in substantial improvement.32,35
In one study undertaken to evaluate family medicine residents’ proficiency in using asthma inhalers, participants (an intervention group at one clinic and a control group at another) all were given a pretest. The intervention group then received educational materials and a tutorial, as well as the opportunity for hands-on practice, after which both groups were given a post-test. The residents who received the training had a 170% jump, on average, in proficiency score, vs a 55% increase for the control group (P<.001).35
Inhaled Medication Instructional Videos
Courtesy of: National Jewish Health
Go to http://www.nationaljewish.org/healthinfo/medications/lung-diseases/devices/instructional-videos
Another study—this one involving first-year interns—looked at level of improvement based on the type of education provided. Initially, only 5% of the interns could use an MDI without error. After a lecture and demonstration, 13% had an error-free technique. But when each intern participated in an intensive one-on-one session, the error-free rate reached 73%. The researchers’ conclusion: Lectures are relatively ineffective in teaching interns inhaler technique compared with a one-on-one approach.32
The Chicago Breathe Project,36 a new program aimed at improving education in the use of asthma inhalers for physicians and minority patients, provides further evidence of the value of clinician education. After a series of workshops for residents at 5 academic institutions, the physicians’ knowledge of proper use of inhalers rose dramatically—from just 5% preprogram to 91% postprogram (P<.001). Six months after the educational activity, the residents (n=161) were more likely (44% vs 11% preprogram) to assess patients’ inhaler technique.36
Teaching patients when time is tight
National and international guidelines stress the need to teach patients correct use of asthma and COPD inhalers.1,37,38 Providing the requisite education includes observation of each patient’s inhaler technique with proper use demonstrated, as needed.
The problem, of course, is how to provide that level of patient education within the time constraints of a busy family practice. We recommend these time-efficient solutions:
Enlist the help of other clinicians. While it is important that someone in your office be well trained and able to instruct patients in the proper use of inhalers, that individual need not be you. The National Institutes of Health recommends that the “principal clinician” introduce key educational messages, which can be reinforced and expanded on by other members of the health care team.1
After you advise patients that it is crucial for them to be trained in and adhere to proper inhaler technique, another health care professional—often a clinic nurse or pharmacist who has had special training—can provide the hands-on education. Studies have shown that when pharmacists who are competent in asthma management, including inhaler technique, work with physicians to optimize the education and overall management of patients with asthma, better outcomes often result, including a reduction in both emergency department visits and hospitalizations.1,39,40
Use videos to demonstrate correct technique. Videos are an effective teaching tool,9 and many of them are device-specific. National Jewish Health, which is world renowned for its asthma care, has a set of instructional videos posted on You-Tube and accessible from its Web site (http://www.nationaljewish.org/healthinfo/medications/lung-diseases/devices/instructional-videos). In addition to videos that demonstrate the use of an MDI alone and an MDI plus a valved holding chamber, the site has links to 6 DPI videos, each covering a different device.
Use intermittent observation. After the patient views the appropriate video, you or a member of your staff will still need to observe the patient’s inhaler technique to ensure correct use. Ideally, this should occur at every visit.1,37 When that’s not possible, use intermittent observation, starting with the first 2 or 3 visits after the introduction of inhalation therapy and then switching to periodic observation to ensure that the patient is maintaining good technique.
In determining how often observation is necessary, keep in mind that simply asking patients whether they are having inhaler problems is not sufficient.1 Patients tend to say they have little or no trouble when, in fact, most struggle, at times, with the devices. What’s more, good technique tends to decrease over time, and repetitive education is important.
To motivate patients, try this communication technique
Motivational interviewing, a technique that has been used to help patients battle obesity, quit smoking, and control hypertension,41-43 among other health problems, can help you identify inhaler problems that need to be addressed. It involves the use of open-ended questions (eg, “What worries you most about your asthma?”), affirmations (“You’ve done a great job testing your peak flow level every morning”), reflective listening (“You’re tired of taking medicine every day”), and summary statements (“You know you should take your medicine every day but you’re having trouble remembering it. Is that right?”).
A pilot study44 showed that when this technique was incorporated into an asthma education session, patient motivation increased. The ratio of perceived advantages vs disadvantages of taking asthma medication correctly improved, as well. Another study45 found that when motivational interviewing was used during home visits to inner-city African American adolescents for asthma care, the patients’ motivation, readiness to adhere to treatment, and asthma-related quality of life improved, although self-reported adherence to asthma medication did not. Further studies involving patients with asthma are under way (www.clinicaltrials.gov/ct2/results?term=asthma).
It is important to note that the use of motivational interviewing does not require a lengthy visit. One study found that on average, visits in which primary care physicians used this communication technique lasted less than 10 minutes.46
CORRESPONDENCE Timothy H. Self, PharmD, University of Tennessee Health Science Center, 881 Madison Avenue, Room 235, Memphis, TN 38163; [email protected]
• Stress the importance of exhaling gently for a few seconds before inhaling (deeply and slowly for a metered dose inhaler, and deeply and rapidly for most dry powder inhalers). C
• Observe the inhaler technique of every patient receiving inhalation therapy on more than one occasion. C
• Don’t rely on self-reports regarding inhaler technique; despite claims of proficiency, most patients make at least one mistake. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
For patients with asthma or chronic obstructive pulmonary disease (COPD), inhalation therapy is the foundation of treatment. Yet all too often, patients don’t get the full value of their inhaled medications because they use their inhaler incorrectly. When technique is markedly flawed, suboptimal outcomes typically result.
Given the number of Americans with asthma (at least 22 million)1 and COPD (more than 13 million adults),2 faulty inhaler technique is a major public health problem. In fact, the number of people suffering from COPD may be even larger: Close to 24 million US adults are believed to have impaired lung function.3,4 For patients with asthma or COPD—many of whom are treated by family physicians—comprehensive education with a focus on correct use of an inhaler is essential.
In this review, we present evidence of frequent inhaler errors (from clinical studies) and highlight some of the more common mistakes (based on our clinical experience [TABLE]5). Finally, we offer ‘‘time-efficient’’ solutions to inhaler problems—steps that physicians in busy primary care practices can take to ensure that patients with asthma or COPD get the maximum benefit from inhalation therapy.
TABLE
Caution patients about these device-specific mistakes*
| Metered dose inhaler |
|---|
|
| Metered dose inhaler plus spacer/VHC |
|
| Dry powder inhaler |
|
| *These are examples based on the experience of the authors; other errors are possible. †Timing is not as crucial as it is for an MDI without a spacer, but the drug is still lost if inhalation is delayed. ‡Correct use varies by type of product (see product literature for specifics). DPI, dry powder inhaler; MDI, metered dose inhaler; VHC, valved holding chamber. Source: Adapted with permission from Self TH, et al. Consultant. 2003.5 |
Inhaler error is well documented
Since 1965, when it was first reported that many patients used metered dose inhalers (MDIs) incorrectly,6 evidence has accumulated supporting the magnitude of the problem.7-12 (Studies conducted in family practice settings are described in “Researchers look at inhaler problems in primary care” and in TABLE W1.13-20)
A number of studies of various sizes (from 41 to 3955 patients) have assessed inhaler technique in patients being treated by clinicians in primary care. The researchers used a variety of scoring methods, as well. Among them were a simple 4-step (0-4) rating system, a 9-step system, a standardized inhaler-specific checklist, and a system that tracked the number of omissions patients made.13-20 All found significant problems with inhaler technique. (You’ll find a detailed look at the studies in TABLE W1 at jfponline.com.)
In one study of 422 patients,13 including young children, adolescents, and adults, participants received one point for correctly performing each of the following steps:
- Adequate preparation (shaking well for those using a metered dose inhaler [MDI]; loading correctly for patients using a dry powder inhaler [DPI])
- Adequate expiration, correct head position
- Adequate inspiratory technique
- Holding breath afterwards.
The researchers found that 25% of the patients had inadequate technique (≤2 on a 0-4 point scale). In this study, as in others that included patients using various types of devices, use of an MDI was associated with a higher rate of incorrect technique.
Another much-smaller study14 used the same 4-step system to assess the technique of 50 patients, all of whom had the same type of DPI and had received extensive training in the correct use of the device. Despite the training, 27% of the patients received scores of ≤2 (inadequate technique). Sixty-eight percent received a score of 3 (adequate); only 5% received a score of 4 (good).
The 2 largest studies—one including 3955 patients using MDIs20 and the other looking at 3811 patients using various kinds of devices18—found high levels of errors, as well. In the latter study, 76% of patients with MDIs made at least one error vs 49% to 55% of patients using DPIs.18 The results convinced a large majority of the physicians caring for these patients of the need to check inhaler technique more frequently. In the study of MDI users alone, 71% of the patients made at least one mistake.20 inhaler misuse was associated with higher asthma instability scores, this study showed.
More recently, a researcher assessed the effects of an integrated primary care model on the management of asthma and/or COPD in middle-aged and elderly patients, in a study of 260 patients in 44 family practices.19 The study included an evaluation of inhaler technique.
Participants were divided into an intervention group—137 patients who received education regarding inhaler use from a nurse—and a usual care group (123 patients). After 2 years, correct inhaler technique among those in the intervention group went from 41% at baseline to 54%. At the same time, the proportion of those in the usual care group with correct technique fell from 47% to 29%.19
Error rates vary widely from one clinical trial to another, depending on study criteria, type of device, and extent of patient education, among other factors. Nonetheless, several studies (spanning 3 decades) found the error rate to be close to, or greater than, 90%.7,10,21
The most recent of these, published in 2009,21 was based on observation of the inhaler technique used by patients with asthma or COPD directly following appointments in an outpatient clinic. The authors found that, although >98% of the study participants claimed to know how to use their inhalers, 94% committed at least one error. In this study and a number of others, user error was more likely in patients using MDIs.13,18,21,22
Adding a spacer (eg, a valved holding chamber such as the AeroChamber) can be helpful, as the spacer affords the patient more time to inhale the medication. But patients who use an MDI with a spacer often make mistakes, too, and patient education is essential.23-26
Breath-activated dry powder inhalers (DPIs)—such as the Flexhaler, HandiHaler, Aerolizer, and Diskus—also reduce the likelihood of error. DPIs eliminate a step that MDI users often struggle with: the need to simultaneously press down on the canister and begin a slow, deep inhalation.
What’s more, DPIs do not have to be shaken before use. Nonetheless, using a DPI still involves a series of actions. For the HandiHaler and Aerolizer, patients must load the dose, and some patients fail to read the directions and swallow the capsule instead of loading it into the device. Patients must remember to exhale away from the device (ie, not into the dry powder) before inhaling, then hold their breath for approximately 10 seconds. There is potential for error at each step.
Stress the need to exhale before using the inhaler
Forgetting to exhale before inhaling is a common, and significant, mistake regardless of the type of device. It is paramount to stress the need to exhale gently for a few seconds before inhaling (slowly and deeply for patients using an MDI, rapidly and deeply with most DPIs). For MDI users, poor timing, described earlier, is another common and serious mistake. Patients using an MDI with a valved holding chamber sometimes inhale for too long before pressing down on the inhaler, then are unable to continue inhaling although the aerosol is still in the chamber. A common error made by patients using multidose DPIs is simply to forget to load the dose.
Physicians need to brush up on their skills, too
It’s not just patients who lack proficiency in inhaler technique. Numerous studies have demonstrated poor skill among physicians and other health care professionals.27-34 Evidence also shows that targeted education results in substantial improvement.32,35
In one study undertaken to evaluate family medicine residents’ proficiency in using asthma inhalers, participants (an intervention group at one clinic and a control group at another) all were given a pretest. The intervention group then received educational materials and a tutorial, as well as the opportunity for hands-on practice, after which both groups were given a post-test. The residents who received the training had a 170% jump, on average, in proficiency score, vs a 55% increase for the control group (P<.001).35
Inhaled Medication Instructional Videos
Courtesy of: National Jewish Health
Go to http://www.nationaljewish.org/healthinfo/medications/lung-diseases/devices/instructional-videos
Another study—this one involving first-year interns—looked at level of improvement based on the type of education provided. Initially, only 5% of the interns could use an MDI without error. After a lecture and demonstration, 13% had an error-free technique. But when each intern participated in an intensive one-on-one session, the error-free rate reached 73%. The researchers’ conclusion: Lectures are relatively ineffective in teaching interns inhaler technique compared with a one-on-one approach.32
The Chicago Breathe Project,36 a new program aimed at improving education in the use of asthma inhalers for physicians and minority patients, provides further evidence of the value of clinician education. After a series of workshops for residents at 5 academic institutions, the physicians’ knowledge of proper use of inhalers rose dramatically—from just 5% preprogram to 91% postprogram (P<.001). Six months after the educational activity, the residents (n=161) were more likely (44% vs 11% preprogram) to assess patients’ inhaler technique.36
Teaching patients when time is tight
National and international guidelines stress the need to teach patients correct use of asthma and COPD inhalers.1,37,38 Providing the requisite education includes observation of each patient’s inhaler technique with proper use demonstrated, as needed.
The problem, of course, is how to provide that level of patient education within the time constraints of a busy family practice. We recommend these time-efficient solutions:
Enlist the help of other clinicians. While it is important that someone in your office be well trained and able to instruct patients in the proper use of inhalers, that individual need not be you. The National Institutes of Health recommends that the “principal clinician” introduce key educational messages, which can be reinforced and expanded on by other members of the health care team.1
After you advise patients that it is crucial for them to be trained in and adhere to proper inhaler technique, another health care professional—often a clinic nurse or pharmacist who has had special training—can provide the hands-on education. Studies have shown that when pharmacists who are competent in asthma management, including inhaler technique, work with physicians to optimize the education and overall management of patients with asthma, better outcomes often result, including a reduction in both emergency department visits and hospitalizations.1,39,40
Use videos to demonstrate correct technique. Videos are an effective teaching tool,9 and many of them are device-specific. National Jewish Health, which is world renowned for its asthma care, has a set of instructional videos posted on You-Tube and accessible from its Web site (http://www.nationaljewish.org/healthinfo/medications/lung-diseases/devices/instructional-videos). In addition to videos that demonstrate the use of an MDI alone and an MDI plus a valved holding chamber, the site has links to 6 DPI videos, each covering a different device.
Use intermittent observation. After the patient views the appropriate video, you or a member of your staff will still need to observe the patient’s inhaler technique to ensure correct use. Ideally, this should occur at every visit.1,37 When that’s not possible, use intermittent observation, starting with the first 2 or 3 visits after the introduction of inhalation therapy and then switching to periodic observation to ensure that the patient is maintaining good technique.
In determining how often observation is necessary, keep in mind that simply asking patients whether they are having inhaler problems is not sufficient.1 Patients tend to say they have little or no trouble when, in fact, most struggle, at times, with the devices. What’s more, good technique tends to decrease over time, and repetitive education is important.
To motivate patients, try this communication technique
Motivational interviewing, a technique that has been used to help patients battle obesity, quit smoking, and control hypertension,41-43 among other health problems, can help you identify inhaler problems that need to be addressed. It involves the use of open-ended questions (eg, “What worries you most about your asthma?”), affirmations (“You’ve done a great job testing your peak flow level every morning”), reflective listening (“You’re tired of taking medicine every day”), and summary statements (“You know you should take your medicine every day but you’re having trouble remembering it. Is that right?”).
A pilot study44 showed that when this technique was incorporated into an asthma education session, patient motivation increased. The ratio of perceived advantages vs disadvantages of taking asthma medication correctly improved, as well. Another study45 found that when motivational interviewing was used during home visits to inner-city African American adolescents for asthma care, the patients’ motivation, readiness to adhere to treatment, and asthma-related quality of life improved, although self-reported adherence to asthma medication did not. Further studies involving patients with asthma are under way (www.clinicaltrials.gov/ct2/results?term=asthma).
It is important to note that the use of motivational interviewing does not require a lengthy visit. One study found that on average, visits in which primary care physicians used this communication technique lasted less than 10 minutes.46
CORRESPONDENCE Timothy H. Self, PharmD, University of Tennessee Health Science Center, 881 Madison Avenue, Room 235, Memphis, TN 38163; [email protected]
1. National Heart, Lung, and Blood Institute; National Asthma Education and Prevention Program Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma. Bethesda, MD: National Institutes of Health; 2007.
2. Centers for Disease Control and Prevention. National Center for Health Statistics: National health interview survey raw data, 2008. Analysis performed by American Lung Association Research and Program Services.
3. American Lung Association. COPD—Helping the missing millions. February 24, 2010. Available at: http://www.lungusa.org/about-us/our-impact/top-stories/copd-helping-the-missing.html. Accessed November 9, 2011.
4. Centers for Disease Control and Prevention. Chronic obstructive pulmonary disease surveillance—United States, 1971-2000. MMWR Surveill Summ. 2002;51(6):1-16.
5. Self TH, Kilgore KE, Shelton V. MDIs, spacers, and dry powder inhalers: what patients are likely to do wrong. Consultant. 2003;49:702-705.
6. Saunders KB. Misuse of inhaled bronchodilator agents. Br Med J. 1965;1:1037-1038.
7. Epstein SW, Manning CPR, Ashley MJ, et al. Survey of the clinical uses of pressurized aerosol inhalers. Can Med Assoc J. 1979;120:813-816.
8. Shim C, Williams MH. The adequacy of inhalation of aerosol from canister nebulizers. Am J Med. 1980;69:891-894.
9. Self TH, Brooks JB, Lieberman P, et al. The value of demonstration and role of the pharmacist in teaching the correct use of pressurized bronchodilators. Can Med Assoc J. 1983;128:129-131.
10. Hartert TV, Windom HH, Peeples RS, et al. Inadequate outpatient medical therapy for patients with asthma admitted to two urban hospitals. Am J Med. 1996;100:386-394.
11. Goodman DE, Israel E, Rosenberg M, et al. The influence of age, diagnosis, and gender on proper use of metered-dose inhalers. Am J Respir Crit Care Med. 1994;150:1256-1261.
12. Newman SP, Pavia D, Clarke SW. How should a pressurized beta-adrenergic bronchodilator be inhaled? Eur J Respir Dis. 1981;62:3-21.
13. Hilton S. An audit of inhaler technique among asthma patients of 34 general practitioners. Br J Gen Pract. 1990;40:505-506.
14. Dompeling E, Van Grunsven PM, Van Schayck GP, et al. Treatment with inhaled steroids in asthma and chronic bronchitis: long-term compliance and inhaler technique. Fam Pract. 1992;9:161-166.
15. Verver S, Poelman M, Bogels A, et al. Effects of instruction by practice assistants on inhaler technique and respiratory symptoms of patients. A controlled randomized videotaped intervention study. Fam Pract. 1996;13:35-40.
16. Dickinson J, Hutton S, Atkin A, et al. Reducing asthma morbidity in the community: the effect of a targeted nurse-run asthma clinic in an English general practice. Respir Med. 1997;91:634-640.
17. Hesselink AE, Penninx BW, Wijnhoven HA, et al. Determinants of an incorrect inhalation technique in patients with asthma or COPD. Scand J Prim Health Care. 2001;19:255-260.
18. Molimard M, Raherison C, Lignot S, et al. Assessment of handling of inhaler devices in real life: An observational study in 3811 patients in primary care. J Aerosol Med. 2003;16:249-254.
19. Meulepas MA, Jacobs JE, Smeenk FW, et al. Effect of an integrated primary care model on the management of middle-aged and old patients with obstructive lung diseases. Scand J Prim Health Care. 2007;25:186-192.
20. Giraud V, Roche N. Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability. Eur Respir J. 2002;19:246-251.
21. Souza ML, Meneghini AC, Ferraz E, et al. Knowledge of and technique for using inhalation devices among asthma patients and COPD patients. J Bras Pneumol. 2009;35:824-831.
22. Rootmensen GN, van Keimpema AR, Jansen HM, et al. Predictors of incorrect inhalation technique in patients with asthma or COPD: a study using a validated videotaped scoring method. J Aerosol Med Pulm Drug Deliv. 2010;23:323-328.
23. Rachelefsky GS, Rohr AS, Wo J, et al. Use of a tube spacer to improve the efficacy of a metered dose inhaler in asthmatic children. Am J Dis Child. 1986;140:1191-1193.
24. Demirkan K, Tolley E, Mastin T, et al. Salmeterol administration by metered-dose inhaler alone vs metered-dose inhaler plus valved holding chamber. Chest. 2000;117:1314-1318.
25. Pedersen S, Ostergaard PA. Nasal inhalation as a cause of inefficient pulmonal aerosol inhalation technique in children. Allergy. 1983;38:191-194.
26. Dolovich MD, Ahrens RS, Hess DR, et al. Device selection an outcomes of aerosol therapy: evidence-based guidelines: American College of Chest Physicians/American College of Asthma, Allergy, and Immunology. Chest. 2005;127:335-371.
27. Interiano B, Guntupalli KK. Metered-dose inhalers: do health care providers know what to teach? Arch Intern Med. 1993;153:81-85.
28. Hanania NA, Wittman R, Kesten S, et al. Medical personnel’s knowledge of and ability to use inhaling devices. Metered-dose inhalers, spacing chambers, and breath-actuated dry powder inhalers. Chest. 1994;105:111-116.
29. Amirav I, Goren A, Pawlowski NA. What do pediatricians in training know about the correct use of inhalers and spacer devices? J Allergy Clin Immunol. 1994;94:669-675.
30. Chopra N, Oprescu N, Fask A, et al. Does introduction of new “easy to use” inhalational devices improve medical personnel’s knowledge of their proper use? Ann Allergy Asthma Immunol. 2002;88:395-400.
31. Self TH, Arnold LB, Czosnowski LM, et al. Inadequate skill of healthcare professionals in using asthma inhalation devices. J Asthma. 2007;44:593-598.
32. Lee-Wong M, Mayo PH. Results of a programme to improve house staff use of metered dose inhalers and spacers. Postgrad Med J. 2003;79:221-225.
33. Muchao FP, Pern SL, Rodriques JC, et al. Evaluation of the knowledge of health professionals at a pediatric hospital regarding the use of metered dose inhalers. J Bras Pneumol. 2008;34:4-12.
34. Kim SH, Kwak HJ, Kim TB, et al. Inappropriate techniques used by internal medicine residents with three kinds of inhalers (a metered dose inhaler, Diskus, and Turbuhaler): changes after a single teaching session. J Asthma. 2009;46:944-950.
35. Kelcher S, Brownoff R. Teaching residents to use asthma devices. Assessing family residents’ skill and a brief intervention. Can Fam Physician. 1994;40:2090-2095.
36. Press VG, Pincayage AT, Pappalardo AA, et al. The Chicago Breathe Project: a regional approach to improving education on asthma inhalers for resident physicians and minority patients. J Natl Med Assoc. 2010;102:548-555.
37. Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention. 2010. Available at: www.ginasthma.org. Accessed November 9, 2011.
38. Executive Summary: global strategy on the diagnosis and management and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Workshop Report, 2009. Available at: www.goldcopd.com. Accessed November 9, 2011.
39. Self TH, Chrisman CR, Mason DL, et al. Reducing emergency department visits and hospitalizations in African American and Hispanic patients: a 15-year review. J Asthma. 2005;42:807-812.
40. Armour C, Bosnic-Anticevich S, Brillant M, et al. Pharmacy asthma care program (PACP) improves outcomes for patients in the community. Thorax. 2007;62:496-502.
41. DiLillo V, Nicole J, West DS. Incorporating motivational interviewing into behavioral obesity treatment. Cogn Behav Pract. 2003;10:120-130.
42. Borrelli B, Novak S, Hecht J, et al. Home health care nurses as a new channel for smoking cessation treatment: outcomes from project CARES (Community-nurse Assisted Research and Education on Smoking). Prev Med. 2005;41:815-821.
43. Woollard L, Beilin L, Lord T, et al. A controlled trial of nurse counselling on lifestyle change for hypertensives treated in general practice: preliminary results. Clin Exp Pharmacol Physiol. 1995;22:466-468.
44. Schmaling K, Blume A, Afari N. A randomized controlled pilot study of motivational interviewing to change attitudes about adherence to medications for asthma. J Clin Psych Med Settings. 2001;8:167-172.
45. Riekert KA, Borrelli B, Bilderback A, et al. The development of a motivational interviewing intervention to promote medication adherence among inner-city, African-American adolescents with asthma. Patient Educ Couns. 2011;82:117-122.
46. Butler C, Rollnick S, Cohen D, et al. Motivational consulting versus brief advice for smokers in general practice: a randomized trial. Br J Gen Pract. 1999;49:611-616.
1. National Heart, Lung, and Blood Institute; National Asthma Education and Prevention Program Expert Panel Report 3: Guidelines for the Diagnosis and Management of Asthma. Bethesda, MD: National Institutes of Health; 2007.
2. Centers for Disease Control and Prevention. National Center for Health Statistics: National health interview survey raw data, 2008. Analysis performed by American Lung Association Research and Program Services.
3. American Lung Association. COPD—Helping the missing millions. February 24, 2010. Available at: http://www.lungusa.org/about-us/our-impact/top-stories/copd-helping-the-missing.html. Accessed November 9, 2011.
4. Centers for Disease Control and Prevention. Chronic obstructive pulmonary disease surveillance—United States, 1971-2000. MMWR Surveill Summ. 2002;51(6):1-16.
5. Self TH, Kilgore KE, Shelton V. MDIs, spacers, and dry powder inhalers: what patients are likely to do wrong. Consultant. 2003;49:702-705.
6. Saunders KB. Misuse of inhaled bronchodilator agents. Br Med J. 1965;1:1037-1038.
7. Epstein SW, Manning CPR, Ashley MJ, et al. Survey of the clinical uses of pressurized aerosol inhalers. Can Med Assoc J. 1979;120:813-816.
8. Shim C, Williams MH. The adequacy of inhalation of aerosol from canister nebulizers. Am J Med. 1980;69:891-894.
9. Self TH, Brooks JB, Lieberman P, et al. The value of demonstration and role of the pharmacist in teaching the correct use of pressurized bronchodilators. Can Med Assoc J. 1983;128:129-131.
10. Hartert TV, Windom HH, Peeples RS, et al. Inadequate outpatient medical therapy for patients with asthma admitted to two urban hospitals. Am J Med. 1996;100:386-394.
11. Goodman DE, Israel E, Rosenberg M, et al. The influence of age, diagnosis, and gender on proper use of metered-dose inhalers. Am J Respir Crit Care Med. 1994;150:1256-1261.
12. Newman SP, Pavia D, Clarke SW. How should a pressurized beta-adrenergic bronchodilator be inhaled? Eur J Respir Dis. 1981;62:3-21.
13. Hilton S. An audit of inhaler technique among asthma patients of 34 general practitioners. Br J Gen Pract. 1990;40:505-506.
14. Dompeling E, Van Grunsven PM, Van Schayck GP, et al. Treatment with inhaled steroids in asthma and chronic bronchitis: long-term compliance and inhaler technique. Fam Pract. 1992;9:161-166.
15. Verver S, Poelman M, Bogels A, et al. Effects of instruction by practice assistants on inhaler technique and respiratory symptoms of patients. A controlled randomized videotaped intervention study. Fam Pract. 1996;13:35-40.
16. Dickinson J, Hutton S, Atkin A, et al. Reducing asthma morbidity in the community: the effect of a targeted nurse-run asthma clinic in an English general practice. Respir Med. 1997;91:634-640.
17. Hesselink AE, Penninx BW, Wijnhoven HA, et al. Determinants of an incorrect inhalation technique in patients with asthma or COPD. Scand J Prim Health Care. 2001;19:255-260.
18. Molimard M, Raherison C, Lignot S, et al. Assessment of handling of inhaler devices in real life: An observational study in 3811 patients in primary care. J Aerosol Med. 2003;16:249-254.
19. Meulepas MA, Jacobs JE, Smeenk FW, et al. Effect of an integrated primary care model on the management of middle-aged and old patients with obstructive lung diseases. Scand J Prim Health Care. 2007;25:186-192.
20. Giraud V, Roche N. Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability. Eur Respir J. 2002;19:246-251.
21. Souza ML, Meneghini AC, Ferraz E, et al. Knowledge of and technique for using inhalation devices among asthma patients and COPD patients. J Bras Pneumol. 2009;35:824-831.
22. Rootmensen GN, van Keimpema AR, Jansen HM, et al. Predictors of incorrect inhalation technique in patients with asthma or COPD: a study using a validated videotaped scoring method. J Aerosol Med Pulm Drug Deliv. 2010;23:323-328.
23. Rachelefsky GS, Rohr AS, Wo J, et al. Use of a tube spacer to improve the efficacy of a metered dose inhaler in asthmatic children. Am J Dis Child. 1986;140:1191-1193.
24. Demirkan K, Tolley E, Mastin T, et al. Salmeterol administration by metered-dose inhaler alone vs metered-dose inhaler plus valved holding chamber. Chest. 2000;117:1314-1318.
25. Pedersen S, Ostergaard PA. Nasal inhalation as a cause of inefficient pulmonal aerosol inhalation technique in children. Allergy. 1983;38:191-194.
26. Dolovich MD, Ahrens RS, Hess DR, et al. Device selection an outcomes of aerosol therapy: evidence-based guidelines: American College of Chest Physicians/American College of Asthma, Allergy, and Immunology. Chest. 2005;127:335-371.
27. Interiano B, Guntupalli KK. Metered-dose inhalers: do health care providers know what to teach? Arch Intern Med. 1993;153:81-85.
28. Hanania NA, Wittman R, Kesten S, et al. Medical personnel’s knowledge of and ability to use inhaling devices. Metered-dose inhalers, spacing chambers, and breath-actuated dry powder inhalers. Chest. 1994;105:111-116.
29. Amirav I, Goren A, Pawlowski NA. What do pediatricians in training know about the correct use of inhalers and spacer devices? J Allergy Clin Immunol. 1994;94:669-675.
30. Chopra N, Oprescu N, Fask A, et al. Does introduction of new “easy to use” inhalational devices improve medical personnel’s knowledge of their proper use? Ann Allergy Asthma Immunol. 2002;88:395-400.
31. Self TH, Arnold LB, Czosnowski LM, et al. Inadequate skill of healthcare professionals in using asthma inhalation devices. J Asthma. 2007;44:593-598.
32. Lee-Wong M, Mayo PH. Results of a programme to improve house staff use of metered dose inhalers and spacers. Postgrad Med J. 2003;79:221-225.
33. Muchao FP, Pern SL, Rodriques JC, et al. Evaluation of the knowledge of health professionals at a pediatric hospital regarding the use of metered dose inhalers. J Bras Pneumol. 2008;34:4-12.
34. Kim SH, Kwak HJ, Kim TB, et al. Inappropriate techniques used by internal medicine residents with three kinds of inhalers (a metered dose inhaler, Diskus, and Turbuhaler): changes after a single teaching session. J Asthma. 2009;46:944-950.
35. Kelcher S, Brownoff R. Teaching residents to use asthma devices. Assessing family residents’ skill and a brief intervention. Can Fam Physician. 1994;40:2090-2095.
36. Press VG, Pincayage AT, Pappalardo AA, et al. The Chicago Breathe Project: a regional approach to improving education on asthma inhalers for resident physicians and minority patients. J Natl Med Assoc. 2010;102:548-555.
37. Global Initiative for Asthma (GINA). Global strategy for asthma management and prevention. 2010. Available at: www.ginasthma.org. Accessed November 9, 2011.
38. Executive Summary: global strategy on the diagnosis and management and prevention of chronic obstructive pulmonary disease. NHLBI/WHO Workshop Report, 2009. Available at: www.goldcopd.com. Accessed November 9, 2011.
39. Self TH, Chrisman CR, Mason DL, et al. Reducing emergency department visits and hospitalizations in African American and Hispanic patients: a 15-year review. J Asthma. 2005;42:807-812.
40. Armour C, Bosnic-Anticevich S, Brillant M, et al. Pharmacy asthma care program (PACP) improves outcomes for patients in the community. Thorax. 2007;62:496-502.
41. DiLillo V, Nicole J, West DS. Incorporating motivational interviewing into behavioral obesity treatment. Cogn Behav Pract. 2003;10:120-130.
42. Borrelli B, Novak S, Hecht J, et al. Home health care nurses as a new channel for smoking cessation treatment: outcomes from project CARES (Community-nurse Assisted Research and Education on Smoking). Prev Med. 2005;41:815-821.
43. Woollard L, Beilin L, Lord T, et al. A controlled trial of nurse counselling on lifestyle change for hypertensives treated in general practice: preliminary results. Clin Exp Pharmacol Physiol. 1995;22:466-468.
44. Schmaling K, Blume A, Afari N. A randomized controlled pilot study of motivational interviewing to change attitudes about adherence to medications for asthma. J Clin Psych Med Settings. 2001;8:167-172.
45. Riekert KA, Borrelli B, Bilderback A, et al. The development of a motivational interviewing intervention to promote medication adherence among inner-city, African-American adolescents with asthma. Patient Educ Couns. 2011;82:117-122.
46. Butler C, Rollnick S, Cohen D, et al. Motivational consulting versus brief advice for smokers in general practice: a randomized trial. Br J Gen Pract. 1999;49:611-616.