User login
Isometric exercise found optimal for lowering blood pressure?
The findings support the development of new exercise guidelines for blood pressure control, the authors said.
Previous research, based on older data that excluded high-intensity interval training (HIIT) and isometric exercise training (IET), led to aerobic exercise training (AET) being recommended for managing blood pressure, according to the authors.
Although AET, HIIT, dynamic resistance training (RT), and combined training (CT) are also effective in reducing both systolic and diastolic blood pressure, the new analysis suggests that IET does it best.
The analysis showed reductions in blood pressure of 8.24/4 mm Hg after IET, compared with 4.49/2.53 mm Hg after AET; 4.55/3.04 mm Hg after RT; 6.04/2.54 mm Hg after CT; and 4.08/2.50 mm Hg after HIIT.
“These findings mirror our smaller-scale trials, and therefore we anticipated that isometrics would be largely effective,” Jamie O’Driscoll, PhD, of Canterbury (England) Christ Church University, said in an interview. However, “the magnitude of difference between isometrics and some other modes was surprising.”
The study was published online in the British Journal of Sports Medicine.
All modes effective
The investigators analyzed data from 270 randomized controlled trials including 15,827 people published between 1990 and February 2023. For consistency, the protocol/intensity of each included paper was screened against the Exercise Prescription in Everyday Practice and Rehabilitative Training tool to be defined and categorized.
All protocols were then stratified as AET, RT, CT, HIIT or IET.
As appropriate, protocols were then further stratified into subgroups: AET included walking, running, and cycling; HIIT included sprint interval training and aerobic interval training; and IET included isometric leg extension and isometric wall squat.
Healthy resting blood pressure was defined as a reading below 130/85 mm Hg, prehypertension as 130-139/85-89 mm Hg, and hypertension as 140/90 mm Hg or higher.
All exercise modes led to statistically significant reductions in systolic BP in normal blood pressure cohorts; however, all reductions were substantially larger in individuals with hypertension.
Pairwise analyses showed significant reductions in resting systolic BP and diastolic BP following AET (−4.49/–2.53 mm Hg); RT (–4.55/–3.04 mm Hg), CT (–6.04/–2.54 mm Hg), HIT (–4.08/–2.50 mm Hg); and IET (–8.24/–4.00 mm Hg).
In the network meta-analysis, the rank order of effectiveness for systolic BP based on surface under the cumulative ranking curve values were IET (SUCRA: 98.3%), CT (75.7%), RT (46.1%), AET (40.5%), and HIIT (39.4%).
Secondary network meta-analyses showed that isometric wall squat was the most effective submode for reducing systolic BP (90.4%), followed by isometric leg extension, isometric hand grip, cycling, running, CT, sprint interval training, other aerobic, RT, aerobic interval training, and walking.
Running was the most effective submode for lowering diastolic BP (91.3%), followed by isometric wall squat, isometric handgrip, isometric leg extension, cycling, sprint interval training, RT, AIT, other aerobic, CT, and walking.
The authors acknowledged limitations, including variability in exercise interventions, missing data, variable quality of exercise monitoring and analyses, lack of blinding to group allocation, varying participant populations, and publication bias.
Nevertheless, they concluded, “the results of this analysis should inform future exercise guideline recommendations for the prevention and treatment of arterial hypertension.”
Guideline changing?
“There are numerous organizations involved in providing and communicating population exercise guidelines,” including World Health Organization, American and European exercise guidelines, and the National Institute for Health and Care Excellence, Dr. O’Driscoll said. “We are currently developing an international collaborative project with other world leaders in the area to develop this line of enquiry.”
In addition, the team is exploring the prescription of IET within England’s National Health Service and extending the study to wider clinical populations.
In a comment, John A. Osborne, MD, PhD, founder and director of State of the Heart Cardiology in Southlake, Tex., said: “This study further lends credence that other forms of exercise, beyond the usually recommended aerobic exercise promulgated in prior guidelines, have significant value for blood pressure lowering, and, potentially, may offer even greater benefits for ... controlling hypertension.”
“This study should inform contemporary nonpharmacological approaches to blood pressure management and allows providers more flexibility in different strategies of exercise to combat high blood pressure,” said Dr. Osborne, a volunteer spokesperson for the American Heart Association.
That said, “while this study by itself is extremely provocative, thoughtful, and rigorously performed, it should be used as hypothesis generating and hopefully [will be followed by] head-to-head studies of aerobic exercise versus resistance training to confirm the findings.”
The study received no funding. Dr. O’Driscoll and Dr. Osborne reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings support the development of new exercise guidelines for blood pressure control, the authors said.
Previous research, based on older data that excluded high-intensity interval training (HIIT) and isometric exercise training (IET), led to aerobic exercise training (AET) being recommended for managing blood pressure, according to the authors.
Although AET, HIIT, dynamic resistance training (RT), and combined training (CT) are also effective in reducing both systolic and diastolic blood pressure, the new analysis suggests that IET does it best.
The analysis showed reductions in blood pressure of 8.24/4 mm Hg after IET, compared with 4.49/2.53 mm Hg after AET; 4.55/3.04 mm Hg after RT; 6.04/2.54 mm Hg after CT; and 4.08/2.50 mm Hg after HIIT.
“These findings mirror our smaller-scale trials, and therefore we anticipated that isometrics would be largely effective,” Jamie O’Driscoll, PhD, of Canterbury (England) Christ Church University, said in an interview. However, “the magnitude of difference between isometrics and some other modes was surprising.”
The study was published online in the British Journal of Sports Medicine.
All modes effective
The investigators analyzed data from 270 randomized controlled trials including 15,827 people published between 1990 and February 2023. For consistency, the protocol/intensity of each included paper was screened against the Exercise Prescription in Everyday Practice and Rehabilitative Training tool to be defined and categorized.
All protocols were then stratified as AET, RT, CT, HIIT or IET.
As appropriate, protocols were then further stratified into subgroups: AET included walking, running, and cycling; HIIT included sprint interval training and aerobic interval training; and IET included isometric leg extension and isometric wall squat.
Healthy resting blood pressure was defined as a reading below 130/85 mm Hg, prehypertension as 130-139/85-89 mm Hg, and hypertension as 140/90 mm Hg or higher.
All exercise modes led to statistically significant reductions in systolic BP in normal blood pressure cohorts; however, all reductions were substantially larger in individuals with hypertension.
Pairwise analyses showed significant reductions in resting systolic BP and diastolic BP following AET (−4.49/–2.53 mm Hg); RT (–4.55/–3.04 mm Hg), CT (–6.04/–2.54 mm Hg), HIT (–4.08/–2.50 mm Hg); and IET (–8.24/–4.00 mm Hg).
In the network meta-analysis, the rank order of effectiveness for systolic BP based on surface under the cumulative ranking curve values were IET (SUCRA: 98.3%), CT (75.7%), RT (46.1%), AET (40.5%), and HIIT (39.4%).
Secondary network meta-analyses showed that isometric wall squat was the most effective submode for reducing systolic BP (90.4%), followed by isometric leg extension, isometric hand grip, cycling, running, CT, sprint interval training, other aerobic, RT, aerobic interval training, and walking.
Running was the most effective submode for lowering diastolic BP (91.3%), followed by isometric wall squat, isometric handgrip, isometric leg extension, cycling, sprint interval training, RT, AIT, other aerobic, CT, and walking.
The authors acknowledged limitations, including variability in exercise interventions, missing data, variable quality of exercise monitoring and analyses, lack of blinding to group allocation, varying participant populations, and publication bias.
Nevertheless, they concluded, “the results of this analysis should inform future exercise guideline recommendations for the prevention and treatment of arterial hypertension.”
Guideline changing?
“There are numerous organizations involved in providing and communicating population exercise guidelines,” including World Health Organization, American and European exercise guidelines, and the National Institute for Health and Care Excellence, Dr. O’Driscoll said. “We are currently developing an international collaborative project with other world leaders in the area to develop this line of enquiry.”
In addition, the team is exploring the prescription of IET within England’s National Health Service and extending the study to wider clinical populations.
In a comment, John A. Osborne, MD, PhD, founder and director of State of the Heart Cardiology in Southlake, Tex., said: “This study further lends credence that other forms of exercise, beyond the usually recommended aerobic exercise promulgated in prior guidelines, have significant value for blood pressure lowering, and, potentially, may offer even greater benefits for ... controlling hypertension.”
“This study should inform contemporary nonpharmacological approaches to blood pressure management and allows providers more flexibility in different strategies of exercise to combat high blood pressure,” said Dr. Osborne, a volunteer spokesperson for the American Heart Association.
That said, “while this study by itself is extremely provocative, thoughtful, and rigorously performed, it should be used as hypothesis generating and hopefully [will be followed by] head-to-head studies of aerobic exercise versus resistance training to confirm the findings.”
The study received no funding. Dr. O’Driscoll and Dr. Osborne reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The findings support the development of new exercise guidelines for blood pressure control, the authors said.
Previous research, based on older data that excluded high-intensity interval training (HIIT) and isometric exercise training (IET), led to aerobic exercise training (AET) being recommended for managing blood pressure, according to the authors.
Although AET, HIIT, dynamic resistance training (RT), and combined training (CT) are also effective in reducing both systolic and diastolic blood pressure, the new analysis suggests that IET does it best.
The analysis showed reductions in blood pressure of 8.24/4 mm Hg after IET, compared with 4.49/2.53 mm Hg after AET; 4.55/3.04 mm Hg after RT; 6.04/2.54 mm Hg after CT; and 4.08/2.50 mm Hg after HIIT.
“These findings mirror our smaller-scale trials, and therefore we anticipated that isometrics would be largely effective,” Jamie O’Driscoll, PhD, of Canterbury (England) Christ Church University, said in an interview. However, “the magnitude of difference between isometrics and some other modes was surprising.”
The study was published online in the British Journal of Sports Medicine.
All modes effective
The investigators analyzed data from 270 randomized controlled trials including 15,827 people published between 1990 and February 2023. For consistency, the protocol/intensity of each included paper was screened against the Exercise Prescription in Everyday Practice and Rehabilitative Training tool to be defined and categorized.
All protocols were then stratified as AET, RT, CT, HIIT or IET.
As appropriate, protocols were then further stratified into subgroups: AET included walking, running, and cycling; HIIT included sprint interval training and aerobic interval training; and IET included isometric leg extension and isometric wall squat.
Healthy resting blood pressure was defined as a reading below 130/85 mm Hg, prehypertension as 130-139/85-89 mm Hg, and hypertension as 140/90 mm Hg or higher.
All exercise modes led to statistically significant reductions in systolic BP in normal blood pressure cohorts; however, all reductions were substantially larger in individuals with hypertension.
Pairwise analyses showed significant reductions in resting systolic BP and diastolic BP following AET (−4.49/–2.53 mm Hg); RT (–4.55/–3.04 mm Hg), CT (–6.04/–2.54 mm Hg), HIT (–4.08/–2.50 mm Hg); and IET (–8.24/–4.00 mm Hg).
In the network meta-analysis, the rank order of effectiveness for systolic BP based on surface under the cumulative ranking curve values were IET (SUCRA: 98.3%), CT (75.7%), RT (46.1%), AET (40.5%), and HIIT (39.4%).
Secondary network meta-analyses showed that isometric wall squat was the most effective submode for reducing systolic BP (90.4%), followed by isometric leg extension, isometric hand grip, cycling, running, CT, sprint interval training, other aerobic, RT, aerobic interval training, and walking.
Running was the most effective submode for lowering diastolic BP (91.3%), followed by isometric wall squat, isometric handgrip, isometric leg extension, cycling, sprint interval training, RT, AIT, other aerobic, CT, and walking.
The authors acknowledged limitations, including variability in exercise interventions, missing data, variable quality of exercise monitoring and analyses, lack of blinding to group allocation, varying participant populations, and publication bias.
Nevertheless, they concluded, “the results of this analysis should inform future exercise guideline recommendations for the prevention and treatment of arterial hypertension.”
Guideline changing?
“There are numerous organizations involved in providing and communicating population exercise guidelines,” including World Health Organization, American and European exercise guidelines, and the National Institute for Health and Care Excellence, Dr. O’Driscoll said. “We are currently developing an international collaborative project with other world leaders in the area to develop this line of enquiry.”
In addition, the team is exploring the prescription of IET within England’s National Health Service and extending the study to wider clinical populations.
In a comment, John A. Osborne, MD, PhD, founder and director of State of the Heart Cardiology in Southlake, Tex., said: “This study further lends credence that other forms of exercise, beyond the usually recommended aerobic exercise promulgated in prior guidelines, have significant value for blood pressure lowering, and, potentially, may offer even greater benefits for ... controlling hypertension.”
“This study should inform contemporary nonpharmacological approaches to blood pressure management and allows providers more flexibility in different strategies of exercise to combat high blood pressure,” said Dr. Osborne, a volunteer spokesperson for the American Heart Association.
That said, “while this study by itself is extremely provocative, thoughtful, and rigorously performed, it should be used as hypothesis generating and hopefully [will be followed by] head-to-head studies of aerobic exercise versus resistance training to confirm the findings.”
The study received no funding. Dr. O’Driscoll and Dr. Osborne reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE BRITISH JOURNAL OF SPORTS MEDICINE
‘Triple G’ agonist hits new weight loss heights
A novel triple agonist to receptors for three nutrient-stimulated hormones led to weight loss as high as 24% among people with overweight or obesity but who did not have type 2 diabetes when used at the highest tested dose for 48 weeks. The results are from a phase 2 study of retatrutide that was published in The New England Journal of Medicine (2023 Aug 10. doi: 10.1056/NEJMoa2301972).
This level of weight loss is “unprecedented” for a medication administered for 48 weeks, Mary-Elizabeth Patti, MD, said in an editorial that accompanied the report.
The findings “offer further optimism ... that effective pharmacologic management of obesity and related disorders is possible,” wrote Dr. Patti, a principal investigator at the Joslin Diabetes Center in Boston.
The study randomly assigned 338 adults with obesity or overweight – a body mass index (BMI) of ≥ 27 kg/m2 – and at least one weight-related complication to receive either weekly subcutaneous injections of retatrutide in any of six dose regimens or placebo over 48 weeks. The primary outcome was weight change from baseline after 24 weeks.
The highest dose of retatrutide safely produced an average 17.5% drop from baseline weight, compared with an average 1.6% reduction in the placebo group, after 24 weeks, a significant difference.
After 48 weeks, the highest retatrutide dose safely cut baseline weight by an average of 24.2%, compared with an average 2.1% drop among placebo control patients, Ania M. Jastreboff, MD, PhD, and her coauthors wrote in their report. Weight loss levels after 24 and 48 weeks of retatrutide treatment followed a clear dose-response pattern.
Weight losses never before seen
“I have never seen weight loss at this level” after nearly 1 year of treatment, Dr. Jastreboff said when she discussed these findings in a press conference at the annual scientific sessions of the American Diabetes Association in San Diego in late June.
A separate presentation at the ADA meeting documented unprecedented weight loss levels in a study of 281 people with obesity or overweight and type 2 diabetes.
“No other medication has shown an average 17% reduction from baseline bodyweight after 36 weeks in people with type 2 diabetes,” said Julio Rosenstock, MD, director of the Dallas Diabetes Research Center at Medical City, Texas, who formally presented the results from the study of retatrutide in people with type 2 diabetes at the ADA meeting.
The mechanism behind retatrutide’s potent weight-loss effect seems likely tied to its action on three human receptors that naturally respond to three nutrient-stimulated hormones that control appetite, metabolism, fat mobilization, and related functions.
The three hormones that the retatrutide molecule simultaneously mimics are glucagon-like peptide-1 (GLP-1), such as agents in the class of GLP-1 agonists that includes liraglutide (Victoza/Saxenda) and semaglutide (Ozempic/Wegovy); the glucose-dependent insulinotropic polypeptide (GIP), the receptor that is also activated by tirzepatide (Mounjaro), a dual-incretin receptor agonist that mimics both GLP-1 and GIP; and glucagon. Survodutide is a dual GLP-1 and glucagon receptor agonist in phase 2 development.
Retatrutide is currently unique among agents with reported clinical results by having agonist effects on the receptors for all three of these hormones, a property that led Dr. Patti to call retatrutide a “triple G” hormone-receptor agonist in her editorial.
Triple agonist has added effect on liver fat clearance
The glucagon-receptor agonism appears to give retatrutide added effects beyond those of the GLP-1 agonists or GLP-1/GIP dual agonists that are increasingly used in U.S. practice.
A prespecified subgroup analysis of the no diabetes/Jastreboff study (but that was not included in the NEJM report) showed that at both 8-mg and 12-mg weekly doses, 24 weeks of retatrutide produced complete resolution of excess liver fat (hepatic steatosis) in about 80% of the people eligible for the analysis (those whose liver volume was at least 10% fat at study entry).
That percentage increased to about 90% of people receiving these doses after 48 weeks, Lee M. Kaplan, MD, reported during a separate presentation at the ADA meeting.
“When you add glucagon activity, liver-fat clearance goes up tremendously,” observed Dr. Kaplan, director of the Obesity, Metabolism and Nutrition Institute at Massachusetts General Hospital in Boston.
The average age of the participants in the new study of the use of retatrutide for those with obesity/overweight but not diabetes was 48 years. By design, 52% were men. (The study sought to enroll roughly equal numbers of men and women.) Average BMI at study entry was 37 kg/m2.
Treatment with retatrutide was also significantly associated with improvements in several cardiometabolic measures in exploratory analyses, including systolic and diastolic blood pressure, A1c, fasting glucose, insulin, and some (but not all) lipids, Dr. Jastreboff, director of the Yale Obesity Research Center of Yale University in New Haven, Conn., and her coauthors reported in the NEJM article.
The safety profile of retatrutide was consistent with reported phase 1 findings for the agent among people with type 2 diabetes and resembled the safety profiles of other agents based on GLP-1 or GIP–GLP-1 mimicry for the treatment of type 2 diabetes or obesity.
The most frequently reported adverse events from retatrutide were transient, mostly mild to moderate gastrointestinal events. They occurred primarily during dose escalation. Discontinuation of retatrutide or placebo because of adverse events occurred in 6% to 16% of the participants who received retatrutide and in none of the participants who received placebo.
Lilly, the company developing retatrutide, previously announced the launch of four phase 3 trials to gather further data on retatrutide for use in a marketing-approval application to the Food and Drug Administration.
The four trials – TRIUMPH-1, TRIUMPH-2, TRIUMPH-3, and TRIUMPH-4 – are evaluating the safety and efficacy of retatrutide for chronic weight management for those with obesity or overweight, including those who also have obstructive sleep apnea, knee osteoarthritis, type 2 diabetes, or cardiovascular disease.
The study was sponsored by Lilly, the company developing retatrutide. Dr. Patti has been a consultant to AstraZeneca, Dexcom, Hanmi, and MBX. She has received funding from Dexcom and has been a monitor for a trial funded by Fractyl. Dr. Jastreboff, Dr. Kaplan, and Dr. Rosenstock have reported financial relationships with Lilly as well as other companies.
A version of this article first appeared on Medscape.com.
A novel triple agonist to receptors for three nutrient-stimulated hormones led to weight loss as high as 24% among people with overweight or obesity but who did not have type 2 diabetes when used at the highest tested dose for 48 weeks. The results are from a phase 2 study of retatrutide that was published in The New England Journal of Medicine (2023 Aug 10. doi: 10.1056/NEJMoa2301972).
This level of weight loss is “unprecedented” for a medication administered for 48 weeks, Mary-Elizabeth Patti, MD, said in an editorial that accompanied the report.
The findings “offer further optimism ... that effective pharmacologic management of obesity and related disorders is possible,” wrote Dr. Patti, a principal investigator at the Joslin Diabetes Center in Boston.
The study randomly assigned 338 adults with obesity or overweight – a body mass index (BMI) of ≥ 27 kg/m2 – and at least one weight-related complication to receive either weekly subcutaneous injections of retatrutide in any of six dose regimens or placebo over 48 weeks. The primary outcome was weight change from baseline after 24 weeks.
The highest dose of retatrutide safely produced an average 17.5% drop from baseline weight, compared with an average 1.6% reduction in the placebo group, after 24 weeks, a significant difference.
After 48 weeks, the highest retatrutide dose safely cut baseline weight by an average of 24.2%, compared with an average 2.1% drop among placebo control patients, Ania M. Jastreboff, MD, PhD, and her coauthors wrote in their report. Weight loss levels after 24 and 48 weeks of retatrutide treatment followed a clear dose-response pattern.
Weight losses never before seen
“I have never seen weight loss at this level” after nearly 1 year of treatment, Dr. Jastreboff said when she discussed these findings in a press conference at the annual scientific sessions of the American Diabetes Association in San Diego in late June.
A separate presentation at the ADA meeting documented unprecedented weight loss levels in a study of 281 people with obesity or overweight and type 2 diabetes.
“No other medication has shown an average 17% reduction from baseline bodyweight after 36 weeks in people with type 2 diabetes,” said Julio Rosenstock, MD, director of the Dallas Diabetes Research Center at Medical City, Texas, who formally presented the results from the study of retatrutide in people with type 2 diabetes at the ADA meeting.
The mechanism behind retatrutide’s potent weight-loss effect seems likely tied to its action on three human receptors that naturally respond to three nutrient-stimulated hormones that control appetite, metabolism, fat mobilization, and related functions.
The three hormones that the retatrutide molecule simultaneously mimics are glucagon-like peptide-1 (GLP-1), such as agents in the class of GLP-1 agonists that includes liraglutide (Victoza/Saxenda) and semaglutide (Ozempic/Wegovy); the glucose-dependent insulinotropic polypeptide (GIP), the receptor that is also activated by tirzepatide (Mounjaro), a dual-incretin receptor agonist that mimics both GLP-1 and GIP; and glucagon. Survodutide is a dual GLP-1 and glucagon receptor agonist in phase 2 development.
Retatrutide is currently unique among agents with reported clinical results by having agonist effects on the receptors for all three of these hormones, a property that led Dr. Patti to call retatrutide a “triple G” hormone-receptor agonist in her editorial.
Triple agonist has added effect on liver fat clearance
The glucagon-receptor agonism appears to give retatrutide added effects beyond those of the GLP-1 agonists or GLP-1/GIP dual agonists that are increasingly used in U.S. practice.
A prespecified subgroup analysis of the no diabetes/Jastreboff study (but that was not included in the NEJM report) showed that at both 8-mg and 12-mg weekly doses, 24 weeks of retatrutide produced complete resolution of excess liver fat (hepatic steatosis) in about 80% of the people eligible for the analysis (those whose liver volume was at least 10% fat at study entry).
That percentage increased to about 90% of people receiving these doses after 48 weeks, Lee M. Kaplan, MD, reported during a separate presentation at the ADA meeting.
“When you add glucagon activity, liver-fat clearance goes up tremendously,” observed Dr. Kaplan, director of the Obesity, Metabolism and Nutrition Institute at Massachusetts General Hospital in Boston.
The average age of the participants in the new study of the use of retatrutide for those with obesity/overweight but not diabetes was 48 years. By design, 52% were men. (The study sought to enroll roughly equal numbers of men and women.) Average BMI at study entry was 37 kg/m2.
Treatment with retatrutide was also significantly associated with improvements in several cardiometabolic measures in exploratory analyses, including systolic and diastolic blood pressure, A1c, fasting glucose, insulin, and some (but not all) lipids, Dr. Jastreboff, director of the Yale Obesity Research Center of Yale University in New Haven, Conn., and her coauthors reported in the NEJM article.
The safety profile of retatrutide was consistent with reported phase 1 findings for the agent among people with type 2 diabetes and resembled the safety profiles of other agents based on GLP-1 or GIP–GLP-1 mimicry for the treatment of type 2 diabetes or obesity.
The most frequently reported adverse events from retatrutide were transient, mostly mild to moderate gastrointestinal events. They occurred primarily during dose escalation. Discontinuation of retatrutide or placebo because of adverse events occurred in 6% to 16% of the participants who received retatrutide and in none of the participants who received placebo.
Lilly, the company developing retatrutide, previously announced the launch of four phase 3 trials to gather further data on retatrutide for use in a marketing-approval application to the Food and Drug Administration.
The four trials – TRIUMPH-1, TRIUMPH-2, TRIUMPH-3, and TRIUMPH-4 – are evaluating the safety and efficacy of retatrutide for chronic weight management for those with obesity or overweight, including those who also have obstructive sleep apnea, knee osteoarthritis, type 2 diabetes, or cardiovascular disease.
The study was sponsored by Lilly, the company developing retatrutide. Dr. Patti has been a consultant to AstraZeneca, Dexcom, Hanmi, and MBX. She has received funding from Dexcom and has been a monitor for a trial funded by Fractyl. Dr. Jastreboff, Dr. Kaplan, and Dr. Rosenstock have reported financial relationships with Lilly as well as other companies.
A version of this article first appeared on Medscape.com.
A novel triple agonist to receptors for three nutrient-stimulated hormones led to weight loss as high as 24% among people with overweight or obesity but who did not have type 2 diabetes when used at the highest tested dose for 48 weeks. The results are from a phase 2 study of retatrutide that was published in The New England Journal of Medicine (2023 Aug 10. doi: 10.1056/NEJMoa2301972).
This level of weight loss is “unprecedented” for a medication administered for 48 weeks, Mary-Elizabeth Patti, MD, said in an editorial that accompanied the report.
The findings “offer further optimism ... that effective pharmacologic management of obesity and related disorders is possible,” wrote Dr. Patti, a principal investigator at the Joslin Diabetes Center in Boston.
The study randomly assigned 338 adults with obesity or overweight – a body mass index (BMI) of ≥ 27 kg/m2 – and at least one weight-related complication to receive either weekly subcutaneous injections of retatrutide in any of six dose regimens or placebo over 48 weeks. The primary outcome was weight change from baseline after 24 weeks.
The highest dose of retatrutide safely produced an average 17.5% drop from baseline weight, compared with an average 1.6% reduction in the placebo group, after 24 weeks, a significant difference.
After 48 weeks, the highest retatrutide dose safely cut baseline weight by an average of 24.2%, compared with an average 2.1% drop among placebo control patients, Ania M. Jastreboff, MD, PhD, and her coauthors wrote in their report. Weight loss levels after 24 and 48 weeks of retatrutide treatment followed a clear dose-response pattern.
Weight losses never before seen
“I have never seen weight loss at this level” after nearly 1 year of treatment, Dr. Jastreboff said when she discussed these findings in a press conference at the annual scientific sessions of the American Diabetes Association in San Diego in late June.
A separate presentation at the ADA meeting documented unprecedented weight loss levels in a study of 281 people with obesity or overweight and type 2 diabetes.
“No other medication has shown an average 17% reduction from baseline bodyweight after 36 weeks in people with type 2 diabetes,” said Julio Rosenstock, MD, director of the Dallas Diabetes Research Center at Medical City, Texas, who formally presented the results from the study of retatrutide in people with type 2 diabetes at the ADA meeting.
The mechanism behind retatrutide’s potent weight-loss effect seems likely tied to its action on three human receptors that naturally respond to three nutrient-stimulated hormones that control appetite, metabolism, fat mobilization, and related functions.
The three hormones that the retatrutide molecule simultaneously mimics are glucagon-like peptide-1 (GLP-1), such as agents in the class of GLP-1 agonists that includes liraglutide (Victoza/Saxenda) and semaglutide (Ozempic/Wegovy); the glucose-dependent insulinotropic polypeptide (GIP), the receptor that is also activated by tirzepatide (Mounjaro), a dual-incretin receptor agonist that mimics both GLP-1 and GIP; and glucagon. Survodutide is a dual GLP-1 and glucagon receptor agonist in phase 2 development.
Retatrutide is currently unique among agents with reported clinical results by having agonist effects on the receptors for all three of these hormones, a property that led Dr. Patti to call retatrutide a “triple G” hormone-receptor agonist in her editorial.
Triple agonist has added effect on liver fat clearance
The glucagon-receptor agonism appears to give retatrutide added effects beyond those of the GLP-1 agonists or GLP-1/GIP dual agonists that are increasingly used in U.S. practice.
A prespecified subgroup analysis of the no diabetes/Jastreboff study (but that was not included in the NEJM report) showed that at both 8-mg and 12-mg weekly doses, 24 weeks of retatrutide produced complete resolution of excess liver fat (hepatic steatosis) in about 80% of the people eligible for the analysis (those whose liver volume was at least 10% fat at study entry).
That percentage increased to about 90% of people receiving these doses after 48 weeks, Lee M. Kaplan, MD, reported during a separate presentation at the ADA meeting.
“When you add glucagon activity, liver-fat clearance goes up tremendously,” observed Dr. Kaplan, director of the Obesity, Metabolism and Nutrition Institute at Massachusetts General Hospital in Boston.
The average age of the participants in the new study of the use of retatrutide for those with obesity/overweight but not diabetes was 48 years. By design, 52% were men. (The study sought to enroll roughly equal numbers of men and women.) Average BMI at study entry was 37 kg/m2.
Treatment with retatrutide was also significantly associated with improvements in several cardiometabolic measures in exploratory analyses, including systolic and diastolic blood pressure, A1c, fasting glucose, insulin, and some (but not all) lipids, Dr. Jastreboff, director of the Yale Obesity Research Center of Yale University in New Haven, Conn., and her coauthors reported in the NEJM article.
The safety profile of retatrutide was consistent with reported phase 1 findings for the agent among people with type 2 diabetes and resembled the safety profiles of other agents based on GLP-1 or GIP–GLP-1 mimicry for the treatment of type 2 diabetes or obesity.
The most frequently reported adverse events from retatrutide were transient, mostly mild to moderate gastrointestinal events. They occurred primarily during dose escalation. Discontinuation of retatrutide or placebo because of adverse events occurred in 6% to 16% of the participants who received retatrutide and in none of the participants who received placebo.
Lilly, the company developing retatrutide, previously announced the launch of four phase 3 trials to gather further data on retatrutide for use in a marketing-approval application to the Food and Drug Administration.
The four trials – TRIUMPH-1, TRIUMPH-2, TRIUMPH-3, and TRIUMPH-4 – are evaluating the safety and efficacy of retatrutide for chronic weight management for those with obesity or overweight, including those who also have obstructive sleep apnea, knee osteoarthritis, type 2 diabetes, or cardiovascular disease.
The study was sponsored by Lilly, the company developing retatrutide. Dr. Patti has been a consultant to AstraZeneca, Dexcom, Hanmi, and MBX. She has received funding from Dexcom and has been a monitor for a trial funded by Fractyl. Dr. Jastreboff, Dr. Kaplan, and Dr. Rosenstock have reported financial relationships with Lilly as well as other companies.
A version of this article first appeared on Medscape.com.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
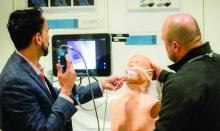
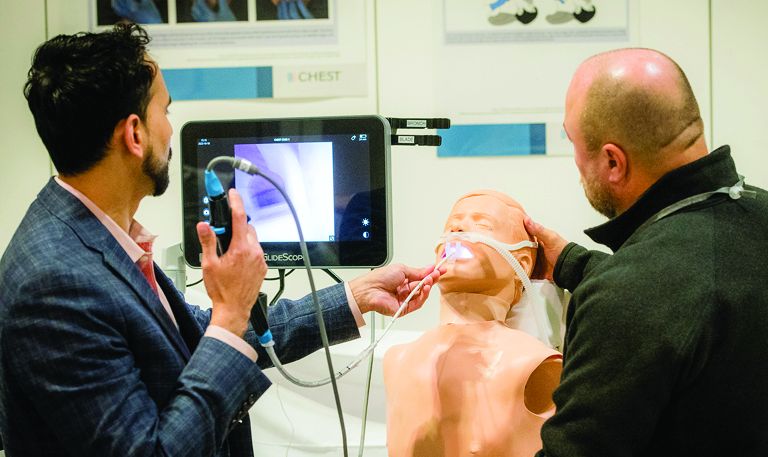
Upper airway ultrasound: Easy to learn, facile to use!
Thoracic Oncology & Chest Procedures Network
Ultrasound & Chest Imaging Section
Point-of-care ultrasound (POCUS) is integral to the delivery of high-quality patient care. The benefits of POCUS for timely diagnosis and procedural assistance are well documented. With continued innovation, its novel benefits can extend to the upper airway evaluation in both inpatient and outpatient settings.
Adi et al notes that POCUS can serve as an adjunct to traditional airway checklists and help intensivists/anesthesiologists identify potentially difficult laryngoscopies, choose the correct endotracheal tube size to reduce the risk of subglottic stenosis, and help confirm appropriate endotracheal tube placement (Adi, et al. J Emerg Crit Care Med. 2019;3:31).
The prediction of a difficult airway is a potentially lifesaving use for this technology. The authors note that smaller studies demonstrate promising results in four techniques: the inability to visualize the hyoid bone using the sublingual approach, a shorter hyomental distance in morbidly obese patients, anterior neck thickness at different anatomical levels (vocal cords, hyoid bone, and thyroid membrane), and a tongue thickness of more than 6.1 cm from the submental approach were all capable of predicting difficult tracheal intubation with varying degrees of sensitivity and specificity.
In the outpatient setting, an understanding of the upper airway anatomy can help with sleep apnea screenings. Korotun, et al. demonstrated in a small sample that ultrasound evaluation of hyoid bone excursion during hypoglossal nerve stimulation may be a useful tool to predict response to therapy and guide hypoglossal nerve stimulator settings (Korotun, et al. Sleep. 2020;43[Suppl_1]:A247-A248).Upper airway ultrasound is easy to learn. The anatomical landmarks are similar in most patients. This convenient tool can be added to your patient care repertoire in a variety of clinical settings.
Sameer Khanijo, MD, FCCP
Section Member-at-Large
Navitha Ramesh, MD, FCCP
Section Vice-Chair
Thoracic Oncology & Chest Procedures Network
Ultrasound & Chest Imaging Section
Point-of-care ultrasound (POCUS) is integral to the delivery of high-quality patient care. The benefits of POCUS for timely diagnosis and procedural assistance are well documented. With continued innovation, its novel benefits can extend to the upper airway evaluation in both inpatient and outpatient settings.
Adi et al notes that POCUS can serve as an adjunct to traditional airway checklists and help intensivists/anesthesiologists identify potentially difficult laryngoscopies, choose the correct endotracheal tube size to reduce the risk of subglottic stenosis, and help confirm appropriate endotracheal tube placement (Adi, et al. J Emerg Crit Care Med. 2019;3:31).
The prediction of a difficult airway is a potentially lifesaving use for this technology. The authors note that smaller studies demonstrate promising results in four techniques: the inability to visualize the hyoid bone using the sublingual approach, a shorter hyomental distance in morbidly obese patients, anterior neck thickness at different anatomical levels (vocal cords, hyoid bone, and thyroid membrane), and a tongue thickness of more than 6.1 cm from the submental approach were all capable of predicting difficult tracheal intubation with varying degrees of sensitivity and specificity.
In the outpatient setting, an understanding of the upper airway anatomy can help with sleep apnea screenings. Korotun, et al. demonstrated in a small sample that ultrasound evaluation of hyoid bone excursion during hypoglossal nerve stimulation may be a useful tool to predict response to therapy and guide hypoglossal nerve stimulator settings (Korotun, et al. Sleep. 2020;43[Suppl_1]:A247-A248).Upper airway ultrasound is easy to learn. The anatomical landmarks are similar in most patients. This convenient tool can be added to your patient care repertoire in a variety of clinical settings.
Sameer Khanijo, MD, FCCP
Section Member-at-Large
Navitha Ramesh, MD, FCCP
Section Vice-Chair
Thoracic Oncology & Chest Procedures Network
Ultrasound & Chest Imaging Section
Point-of-care ultrasound (POCUS) is integral to the delivery of high-quality patient care. The benefits of POCUS for timely diagnosis and procedural assistance are well documented. With continued innovation, its novel benefits can extend to the upper airway evaluation in both inpatient and outpatient settings.
Adi et al notes that POCUS can serve as an adjunct to traditional airway checklists and help intensivists/anesthesiologists identify potentially difficult laryngoscopies, choose the correct endotracheal tube size to reduce the risk of subglottic stenosis, and help confirm appropriate endotracheal tube placement (Adi, et al. J Emerg Crit Care Med. 2019;3:31).
The prediction of a difficult airway is a potentially lifesaving use for this technology. The authors note that smaller studies demonstrate promising results in four techniques: the inability to visualize the hyoid bone using the sublingual approach, a shorter hyomental distance in morbidly obese patients, anterior neck thickness at different anatomical levels (vocal cords, hyoid bone, and thyroid membrane), and a tongue thickness of more than 6.1 cm from the submental approach were all capable of predicting difficult tracheal intubation with varying degrees of sensitivity and specificity.
In the outpatient setting, an understanding of the upper airway anatomy can help with sleep apnea screenings. Korotun, et al. demonstrated in a small sample that ultrasound evaluation of hyoid bone excursion during hypoglossal nerve stimulation may be a useful tool to predict response to therapy and guide hypoglossal nerve stimulator settings (Korotun, et al. Sleep. 2020;43[Suppl_1]:A247-A248).Upper airway ultrasound is easy to learn. The anatomical landmarks are similar in most patients. This convenient tool can be added to your patient care repertoire in a variety of clinical settings.
Sameer Khanijo, MD, FCCP
Section Member-at-Large
Navitha Ramesh, MD, FCCP
Section Vice-Chair
Addressing disparities in goals-of-care conversations
Critical Care Network
Nonrespiratory Critical Care Section
Goals-of-care discussions are essential to management of the intensive care unit (ICU) patient. Racial inequities in end-of-life decision making have been documented for many years, with literature demonstrating that marginalized populations are less likely to have EHR-documented goals-of-care discussions and more likely to have concerns regarding clinician communication.
A recently published randomized control trial in JAMA highlights an intervention that offers promise in addressing disparities in goals-of-care conversations. Curtis, et al. studied whether priming physicians with a communication guide advising on discussion prompts and language for goals-of-care could improve the rate of documented goals-of-care discussions among hospitalized older adults with serious illness. The study found that for patients in the intervention arm, there was a significant increase in proportion of goals-of-care discussions within 30 days. Notably, the difference in documented goals-of-care discussions between arms was greater in the subgroup of patients from underserved groups (Curtis JR, et al. JAMA. 2023;329[23]:2028-37).
Nevertheless, while interventions may help increase the rate of goals-of-care discussions, it is also important to address the content of discussions themselves. You and colleagues recently published a mixed-methods study assessing the impact of race on shared decision-making behaviors during family/caregiver meetings. The authors found that while ICU physicians approached shared decision making with White and Black families similarly, Black families felt their physicians provided less validation of the family role in decision making than White families did (You H, et al. Ann Am Thorac Soc. 2023 May;20[5]:759-62). These findings highlight the importance of ongoing work that focuses not only on quantity but also on quality of communication regarding goals-of-care for patients from diverse backgrounds.
Divya Shankar MD
Section Fellow-in-Training
Muhammad Hayat-Syed MD
Section Vice Chair
Critical Care Network
Nonrespiratory Critical Care Section
Goals-of-care discussions are essential to management of the intensive care unit (ICU) patient. Racial inequities in end-of-life decision making have been documented for many years, with literature demonstrating that marginalized populations are less likely to have EHR-documented goals-of-care discussions and more likely to have concerns regarding clinician communication.
A recently published randomized control trial in JAMA highlights an intervention that offers promise in addressing disparities in goals-of-care conversations. Curtis, et al. studied whether priming physicians with a communication guide advising on discussion prompts and language for goals-of-care could improve the rate of documented goals-of-care discussions among hospitalized older adults with serious illness. The study found that for patients in the intervention arm, there was a significant increase in proportion of goals-of-care discussions within 30 days. Notably, the difference in documented goals-of-care discussions between arms was greater in the subgroup of patients from underserved groups (Curtis JR, et al. JAMA. 2023;329[23]:2028-37).
Nevertheless, while interventions may help increase the rate of goals-of-care discussions, it is also important to address the content of discussions themselves. You and colleagues recently published a mixed-methods study assessing the impact of race on shared decision-making behaviors during family/caregiver meetings. The authors found that while ICU physicians approached shared decision making with White and Black families similarly, Black families felt their physicians provided less validation of the family role in decision making than White families did (You H, et al. Ann Am Thorac Soc. 2023 May;20[5]:759-62). These findings highlight the importance of ongoing work that focuses not only on quantity but also on quality of communication regarding goals-of-care for patients from diverse backgrounds.
Divya Shankar MD
Section Fellow-in-Training
Muhammad Hayat-Syed MD
Section Vice Chair
Critical Care Network
Nonrespiratory Critical Care Section
Goals-of-care discussions are essential to management of the intensive care unit (ICU) patient. Racial inequities in end-of-life decision making have been documented for many years, with literature demonstrating that marginalized populations are less likely to have EHR-documented goals-of-care discussions and more likely to have concerns regarding clinician communication.
A recently published randomized control trial in JAMA highlights an intervention that offers promise in addressing disparities in goals-of-care conversations. Curtis, et al. studied whether priming physicians with a communication guide advising on discussion prompts and language for goals-of-care could improve the rate of documented goals-of-care discussions among hospitalized older adults with serious illness. The study found that for patients in the intervention arm, there was a significant increase in proportion of goals-of-care discussions within 30 days. Notably, the difference in documented goals-of-care discussions between arms was greater in the subgroup of patients from underserved groups (Curtis JR, et al. JAMA. 2023;329[23]:2028-37).
Nevertheless, while interventions may help increase the rate of goals-of-care discussions, it is also important to address the content of discussions themselves. You and colleagues recently published a mixed-methods study assessing the impact of race on shared decision-making behaviors during family/caregiver meetings. The authors found that while ICU physicians approached shared decision making with White and Black families similarly, Black families felt their physicians provided less validation of the family role in decision making than White families did (You H, et al. Ann Am Thorac Soc. 2023 May;20[5]:759-62). These findings highlight the importance of ongoing work that focuses not only on quantity but also on quality of communication regarding goals-of-care for patients from diverse backgrounds.
Divya Shankar MD
Section Fellow-in-Training
Muhammad Hayat-Syed MD
Section Vice Chair
Use of frailty assessment in lung transplant evaluation
Diffuse Lung Disease & Transplant Network
Lung Transplant Section
Frailty, a concept that originated in the geriatric population, is a state of vulnerability resulting from a decline in reserve and function across physiological systems. While it is more commonly observed in older adults, some aging-associated syndromes, such as sarcopenia, impaired cognition, inflammation, and malnutrition, may be present in younger patients with end-stage organ disease. These syndromes can be associated with biological age, as opposed to chronological age, which explains why younger patients with end-stage organ disease can develop frailty (Schaenman JM, et al. Am J Transplant. 2021 Jun;21[6]:2018-24). Frailty in the lung transplant population is associated with increased morbidity and mortality while on the waitlist and post-transplant (Montgomery E, et al. J Transplant. 2020 Aug 7:3239495). In 2021, the International Society of Heart and Lung Transplantation recommended including a frailty assessment to complete a patient’s transplant evaluation. The committee cautioned using current assessment tools, as they are not yet accepted as the standard of care (Leard, et al. J Heart Lung Transplant. 2021 Nov;40[11]:1349-79). Existing tools being used evolved from studies of community-dwelling older adults with no predilection for distinct organ disease, which include the Fried Physical Frailty Phenotype (FPFP) and the Short Physical Performance Battery (SPPB). Along with physical limitations, frail patients tend to have abnormal biomarkers including higher inflammatory cytokines, such as plasma IL-6 and tumor necrosis factor receptor 1, and lower insulin-like growth factor I and leptin (Singer JP, et al. Am J Respir Crit Care Med. 2015;192[11]1325-34). The concept of a lung-focused approach to frailty, which considers biomarkers and body composition, is currently being researched (Singer JP, et al. J Heart Lung Transplant. 2023;S1053-S2498[23]00049-9). This disease-specific frailty scale would identify lung transplant candidates who may benefit from targeted interventions, and such frailty would also be expected to improve after transplant.
Erin Meier, MD
Section Fellow-in-Training
Anupam Kumar, MD, FCCP
Section Member-at-Large
Diffuse Lung Disease & Transplant Network
Lung Transplant Section
Frailty, a concept that originated in the geriatric population, is a state of vulnerability resulting from a decline in reserve and function across physiological systems. While it is more commonly observed in older adults, some aging-associated syndromes, such as sarcopenia, impaired cognition, inflammation, and malnutrition, may be present in younger patients with end-stage organ disease. These syndromes can be associated with biological age, as opposed to chronological age, which explains why younger patients with end-stage organ disease can develop frailty (Schaenman JM, et al. Am J Transplant. 2021 Jun;21[6]:2018-24). Frailty in the lung transplant population is associated with increased morbidity and mortality while on the waitlist and post-transplant (Montgomery E, et al. J Transplant. 2020 Aug 7:3239495). In 2021, the International Society of Heart and Lung Transplantation recommended including a frailty assessment to complete a patient’s transplant evaluation. The committee cautioned using current assessment tools, as they are not yet accepted as the standard of care (Leard, et al. J Heart Lung Transplant. 2021 Nov;40[11]:1349-79). Existing tools being used evolved from studies of community-dwelling older adults with no predilection for distinct organ disease, which include the Fried Physical Frailty Phenotype (FPFP) and the Short Physical Performance Battery (SPPB). Along with physical limitations, frail patients tend to have abnormal biomarkers including higher inflammatory cytokines, such as plasma IL-6 and tumor necrosis factor receptor 1, and lower insulin-like growth factor I and leptin (Singer JP, et al. Am J Respir Crit Care Med. 2015;192[11]1325-34). The concept of a lung-focused approach to frailty, which considers biomarkers and body composition, is currently being researched (Singer JP, et al. J Heart Lung Transplant. 2023;S1053-S2498[23]00049-9). This disease-specific frailty scale would identify lung transplant candidates who may benefit from targeted interventions, and such frailty would also be expected to improve after transplant.
Erin Meier, MD
Section Fellow-in-Training
Anupam Kumar, MD, FCCP
Section Member-at-Large
Diffuse Lung Disease & Transplant Network
Lung Transplant Section
Frailty, a concept that originated in the geriatric population, is a state of vulnerability resulting from a decline in reserve and function across physiological systems. While it is more commonly observed in older adults, some aging-associated syndromes, such as sarcopenia, impaired cognition, inflammation, and malnutrition, may be present in younger patients with end-stage organ disease. These syndromes can be associated with biological age, as opposed to chronological age, which explains why younger patients with end-stage organ disease can develop frailty (Schaenman JM, et al. Am J Transplant. 2021 Jun;21[6]:2018-24). Frailty in the lung transplant population is associated with increased morbidity and mortality while on the waitlist and post-transplant (Montgomery E, et al. J Transplant. 2020 Aug 7:3239495). In 2021, the International Society of Heart and Lung Transplantation recommended including a frailty assessment to complete a patient’s transplant evaluation. The committee cautioned using current assessment tools, as they are not yet accepted as the standard of care (Leard, et al. J Heart Lung Transplant. 2021 Nov;40[11]:1349-79). Existing tools being used evolved from studies of community-dwelling older adults with no predilection for distinct organ disease, which include the Fried Physical Frailty Phenotype (FPFP) and the Short Physical Performance Battery (SPPB). Along with physical limitations, frail patients tend to have abnormal biomarkers including higher inflammatory cytokines, such as plasma IL-6 and tumor necrosis factor receptor 1, and lower insulin-like growth factor I and leptin (Singer JP, et al. Am J Respir Crit Care Med. 2015;192[11]1325-34). The concept of a lung-focused approach to frailty, which considers biomarkers and body composition, is currently being researched (Singer JP, et al. J Heart Lung Transplant. 2023;S1053-S2498[23]00049-9). This disease-specific frailty scale would identify lung transplant candidates who may benefit from targeted interventions, and such frailty would also be expected to improve after transplant.
Erin Meier, MD
Section Fellow-in-Training
Anupam Kumar, MD, FCCP
Section Member-at-Large
DPP1 a promising target for bronchiectasis
Airway Disorders Network
Bronchiectasis Section
and persistent inflammation. In bronchiectasis, excessive neutrophil accumulation in the airways leads to release of neutrophil serine proteases (NSPs), which contributes to tissue damage and perpetuates the inflammatory process in the lungs. The three main NSPs include neutrophil elastase (NE), proteinase3, and cathepsin G. Elevations in NE activity in sputum in NCFBE are associated with increased exacerbations and declines in lung function. Dipeptidyl peptidase 1 (DPP1), an enzyme primarily found in neutrophils, is responsible for activating NSPs during neutrophil maturation. In bronchiectasis, increased DPP1 activity results in an augmented production of active NSPs, exacerbating lung damage and inflammation.
Brensocatib, an oral, reversible inhibitor of DPP1 is currently being developed as a novel approach to managing bronchiectasis. Brensocatib was evaluated in a phase 2 clinical trial (WILLOW), a randomized, double-blind, placebo-controlled trial involving adults with non–cystic fibrosis bronchiectasis (NCFBE). Treatment with brensocatib for 24 weeks significantly prolonged the time to the first exacerbation at both the 10 mg and 25 mg doses and lowered the risk of exacerbation by 40% relative to placebo. The treatment was well tolerated, with no significant safety concerns. Results of a recent post hoc analysis from the WILLOW study show that brensocatib effectively reduces exacerbations and slows lung function decline across different severities of bronchiectasis. These findings suggest that brensocatib holds potential as the 1st new therapeutic option for patients with NCFBE, with currently no FDA-approved drugs. Results of a larger-scale phase 3 trial are awaited later this year, which will hopefully confirm these results and ascertain the long-term safety.
Shyamsunder Subramanian, MD, MBBS, FCCP
Section Chair
Airway Disorders Network
Bronchiectasis Section
and persistent inflammation. In bronchiectasis, excessive neutrophil accumulation in the airways leads to release of neutrophil serine proteases (NSPs), which contributes to tissue damage and perpetuates the inflammatory process in the lungs. The three main NSPs include neutrophil elastase (NE), proteinase3, and cathepsin G. Elevations in NE activity in sputum in NCFBE are associated with increased exacerbations and declines in lung function. Dipeptidyl peptidase 1 (DPP1), an enzyme primarily found in neutrophils, is responsible for activating NSPs during neutrophil maturation. In bronchiectasis, increased DPP1 activity results in an augmented production of active NSPs, exacerbating lung damage and inflammation.
Brensocatib, an oral, reversible inhibitor of DPP1 is currently being developed as a novel approach to managing bronchiectasis. Brensocatib was evaluated in a phase 2 clinical trial (WILLOW), a randomized, double-blind, placebo-controlled trial involving adults with non–cystic fibrosis bronchiectasis (NCFBE). Treatment with brensocatib for 24 weeks significantly prolonged the time to the first exacerbation at both the 10 mg and 25 mg doses and lowered the risk of exacerbation by 40% relative to placebo. The treatment was well tolerated, with no significant safety concerns. Results of a recent post hoc analysis from the WILLOW study show that brensocatib effectively reduces exacerbations and slows lung function decline across different severities of bronchiectasis. These findings suggest that brensocatib holds potential as the 1st new therapeutic option for patients with NCFBE, with currently no FDA-approved drugs. Results of a larger-scale phase 3 trial are awaited later this year, which will hopefully confirm these results and ascertain the long-term safety.
Shyamsunder Subramanian, MD, MBBS, FCCP
Section Chair
Airway Disorders Network
Bronchiectasis Section
and persistent inflammation. In bronchiectasis, excessive neutrophil accumulation in the airways leads to release of neutrophil serine proteases (NSPs), which contributes to tissue damage and perpetuates the inflammatory process in the lungs. The three main NSPs include neutrophil elastase (NE), proteinase3, and cathepsin G. Elevations in NE activity in sputum in NCFBE are associated with increased exacerbations and declines in lung function. Dipeptidyl peptidase 1 (DPP1), an enzyme primarily found in neutrophils, is responsible for activating NSPs during neutrophil maturation. In bronchiectasis, increased DPP1 activity results in an augmented production of active NSPs, exacerbating lung damage and inflammation.
Brensocatib, an oral, reversible inhibitor of DPP1 is currently being developed as a novel approach to managing bronchiectasis. Brensocatib was evaluated in a phase 2 clinical trial (WILLOW), a randomized, double-blind, placebo-controlled trial involving adults with non–cystic fibrosis bronchiectasis (NCFBE). Treatment with brensocatib for 24 weeks significantly prolonged the time to the first exacerbation at both the 10 mg and 25 mg doses and lowered the risk of exacerbation by 40% relative to placebo. The treatment was well tolerated, with no significant safety concerns. Results of a recent post hoc analysis from the WILLOW study show that brensocatib effectively reduces exacerbations and slows lung function decline across different severities of bronchiectasis. These findings suggest that brensocatib holds potential as the 1st new therapeutic option for patients with NCFBE, with currently no FDA-approved drugs. Results of a larger-scale phase 3 trial are awaited later this year, which will hopefully confirm these results and ascertain the long-term safety.
Shyamsunder Subramanian, MD, MBBS, FCCP
Section Chair
Celebrating the inaugural issues of CHEST’s new open access journals
After much anticipation, the inaugural issues of both CHEST® Critical Care and CHEST® Pulmonary officially launched in late June. – promoting transparency, inclusiveness, and collaboration in research – and offer authors more avenues to share their practice-changing research.
The first issue of CHEST Critical Care featured research into ICU mortality across prepandemic and pandemic cohorts in resource-limited settings in South Africa, an exploration into symptom trajectory in recipients of hematopoietic stem-cell transplantation, a narrative review of post-intensive care syndrome, and an investigation into early echocardiographic and ultrasonographic findings in critically ill patients with COVID-19.
In addition, an editorial from Hayley Gershengorn, MD, Editor in Chief of CHEST Critical Care, offers readers more insights into the need for a publication focused on the breadth of clinical topics in critical care and her goals for the new publication.
“I’m ecstatic for this launch. We are grateful to our authors for the trust they put in us and are excited to share their work with our critical care colleagues around the world,” Dr. Gershengorn said. “The editorial team and the American College of Chest Physicians staff have worked tirelessly on this journal, and it’s incredibly gratifying to see the first issue publish.”
Read the full issue and new research from the journal at www.chestcc.org.
In his own editorial featured in the inaugural issue of CHEST Pulmonary, Editor in Chief Matthew Miles, MD, MEd, FCCP, shares how the flagship journal’s proud heritage of sharing impactful clinical research – and the need to target areas of pulmonary and sleep medicine research not covered by other journals – inspired the creation of this new publication.
The issue also includes research into mobile health opportunities for asthma management, an exploration into telemedicine for patients with interstitial lung diseases, an in-depth review into the rare and often underdiagnosed disorder primary ciliary dyskinesia, research on the impact of the social vulnerability index on pulmonary embolism mortality, and an investigation into pneumothorax complications after percutaneous lung biopsy.
“I am deeply grateful to our authors, reviewers, editorial board, and staff who have contributed to the launch of our first issue,” Dr. Miles said. “The journal CHEST is known for excellence in clinically relevant research and patient management guidance. CHEST Pulmonary expands the CHEST portfolio with additional opportunity for researchers to share their work in an exclusively open access format to reach the broadest possible audience. I know our readers will enjoy learning from the research and reviews in issue one.”
Review the full issue and new articles from CHEST Pulmonary at www.chestpulmonary.org.
After much anticipation, the inaugural issues of both CHEST® Critical Care and CHEST® Pulmonary officially launched in late June. – promoting transparency, inclusiveness, and collaboration in research – and offer authors more avenues to share their practice-changing research.
The first issue of CHEST Critical Care featured research into ICU mortality across prepandemic and pandemic cohorts in resource-limited settings in South Africa, an exploration into symptom trajectory in recipients of hematopoietic stem-cell transplantation, a narrative review of post-intensive care syndrome, and an investigation into early echocardiographic and ultrasonographic findings in critically ill patients with COVID-19.
In addition, an editorial from Hayley Gershengorn, MD, Editor in Chief of CHEST Critical Care, offers readers more insights into the need for a publication focused on the breadth of clinical topics in critical care and her goals for the new publication.
“I’m ecstatic for this launch. We are grateful to our authors for the trust they put in us and are excited to share their work with our critical care colleagues around the world,” Dr. Gershengorn said. “The editorial team and the American College of Chest Physicians staff have worked tirelessly on this journal, and it’s incredibly gratifying to see the first issue publish.”
Read the full issue and new research from the journal at www.chestcc.org.
In his own editorial featured in the inaugural issue of CHEST Pulmonary, Editor in Chief Matthew Miles, MD, MEd, FCCP, shares how the flagship journal’s proud heritage of sharing impactful clinical research – and the need to target areas of pulmonary and sleep medicine research not covered by other journals – inspired the creation of this new publication.
The issue also includes research into mobile health opportunities for asthma management, an exploration into telemedicine for patients with interstitial lung diseases, an in-depth review into the rare and often underdiagnosed disorder primary ciliary dyskinesia, research on the impact of the social vulnerability index on pulmonary embolism mortality, and an investigation into pneumothorax complications after percutaneous lung biopsy.
“I am deeply grateful to our authors, reviewers, editorial board, and staff who have contributed to the launch of our first issue,” Dr. Miles said. “The journal CHEST is known for excellence in clinically relevant research and patient management guidance. CHEST Pulmonary expands the CHEST portfolio with additional opportunity for researchers to share their work in an exclusively open access format to reach the broadest possible audience. I know our readers will enjoy learning from the research and reviews in issue one.”
Review the full issue and new articles from CHEST Pulmonary at www.chestpulmonary.org.
After much anticipation, the inaugural issues of both CHEST® Critical Care and CHEST® Pulmonary officially launched in late June. – promoting transparency, inclusiveness, and collaboration in research – and offer authors more avenues to share their practice-changing research.
The first issue of CHEST Critical Care featured research into ICU mortality across prepandemic and pandemic cohorts in resource-limited settings in South Africa, an exploration into symptom trajectory in recipients of hematopoietic stem-cell transplantation, a narrative review of post-intensive care syndrome, and an investigation into early echocardiographic and ultrasonographic findings in critically ill patients with COVID-19.
In addition, an editorial from Hayley Gershengorn, MD, Editor in Chief of CHEST Critical Care, offers readers more insights into the need for a publication focused on the breadth of clinical topics in critical care and her goals for the new publication.
“I’m ecstatic for this launch. We are grateful to our authors for the trust they put in us and are excited to share their work with our critical care colleagues around the world,” Dr. Gershengorn said. “The editorial team and the American College of Chest Physicians staff have worked tirelessly on this journal, and it’s incredibly gratifying to see the first issue publish.”
Read the full issue and new research from the journal at www.chestcc.org.
In his own editorial featured in the inaugural issue of CHEST Pulmonary, Editor in Chief Matthew Miles, MD, MEd, FCCP, shares how the flagship journal’s proud heritage of sharing impactful clinical research – and the need to target areas of pulmonary and sleep medicine research not covered by other journals – inspired the creation of this new publication.
The issue also includes research into mobile health opportunities for asthma management, an exploration into telemedicine for patients with interstitial lung diseases, an in-depth review into the rare and often underdiagnosed disorder primary ciliary dyskinesia, research on the impact of the social vulnerability index on pulmonary embolism mortality, and an investigation into pneumothorax complications after percutaneous lung biopsy.
“I am deeply grateful to our authors, reviewers, editorial board, and staff who have contributed to the launch of our first issue,” Dr. Miles said. “The journal CHEST is known for excellence in clinically relevant research and patient management guidance. CHEST Pulmonary expands the CHEST portfolio with additional opportunity for researchers to share their work in an exclusively open access format to reach the broadest possible audience. I know our readers will enjoy learning from the research and reviews in issue one.”
Review the full issue and new articles from CHEST Pulmonary at www.chestpulmonary.org.
Which biologic therapy should I use in patients who have moderate to severe asthma with associated comorbidities?
Dr. Hossri and Dr. Ivashchuk are with UTHealth Houston –Texas Medical Center, Department of Internal Medicine; Division of Pulmonary, Critical Care, and Sleep Medicine.
As new treatments for specific moderate to severe asthma phenotypes have been developed, management decisions have grown more complicated. The treatment indications for asthma are clear; however, there is overlap with certain therapeutics that target the same pathway with similar end results. because it is not a one-size-fits-all approach that follows a rigid algorithm. Instead, it is a customized treatment plan that accounts for patient-specific risk factors and comorbidities.
Comorbidities commonly associated with asthma include atopic dermatitis, chronic rhinosinusitis with nasal polyposis, eosinophilic granulomatosis with polyangiitis, eosinophilic esophagitis, bronchiectasis and allergic bronchopulmonary aspergillosis. While we lack consensus or a universally accepted treatment algorithm for treating asthma when these comorbidities are present, recent evidence helps guide us to which therapies work best.
Atopic dermatitis
There is a higher prevalence of asthma in patients with atopic dermatitis. A concept called the “atopic march” refers to the progression of childhood atopic dermatitis to manifestations such as asthma, food allergies, and hay fever. The more severe the atopic dermatitis is in childhood, the higher the risk for asthma later on in life. The data on the biologic pathogenesis of atopic dermatitis point to the involvement of interleukins – interleukin (IL)-4 and IL 13 (Silverberg JI. Ann Allergy Asthma Immunol. 2019;123[2]:144-51).
These same interleukins are active in what is called “Th2-high” asthma. The activation of Th2 cells in the inflammatory pathway occurs in atopic dermatitis and asthma irrespective of immunoglobulin E levels. Preliminary data show therapies that target IL-13 alone are effective for treating asthma with comorbid atopic dermatitis but those blocking both IL-4 and IL-13, like dupilumab, are superior. Both interleukins are considered pivotal in the Th-2 pathway. This suggests that dual inhibition is an integral component in the treatment of moderate to severe atopic dermatitis with asthma. Analysis of other Th2 mediators, such as mepolizumab (IL-5 antagonist) and omalizumab (anti-IgE) have shown minimal efficacy, further supporting the use of dupilumab (Guttman-Yassky E, et al. J Allergy Clin. Immunol. 2019 Jan;143[1]:155-72).
Chronic rhinosinusitis with nasal polyposis
The “unified airway” concept holds that because the upper airways (nasal mucosa, pharynx, and larynx) are in direct communication with the lower airways (bronchi and bronchioles). This would explain the correlation between chronic rhinosinusitis with nasal polyposis (CRSwNP) and asthma. Many studies also show the severity of one disease increases the severity of the other.
Patients with both CRSwNP and asthma typically experience a more treatment-resistant course characterized by higher rates of corticosteroid dependence and nasal polyposis recurrences when compared with asthma alone (Laidlaw TM, et al. J Allergy Clin Immunol. 2021 Mar;9[3]:1133-41). They typically have Th2-high asthma and are usually eosinophilic. The optimal treatment approach is mindful of the unified airway concept. Large-scale studies demonstrate significant benefit when targeting IL-5, especially in those with bilateral nasal polyps, need for systemic steroids in the past 2 years, significant impairment in quality of life, loss of smell, and a concomitant diagnosis of asthma (Fokkens WJ, et al. Allergy. 2019 Dec;74[12]:2312). Although data are inconsistent, there is enough evidence to suggest dupilumab be considered for those with eosinophilic asthma and CRSwNP along with atopy, atopic dermatitis, and/or high FeNO levels. In those without atopic symptoms, an anti-IL5/anti-IL5R (mainly mepolizumab and benralizumab) is preferred. Having said this, direct comparative analyses between biologics are lacking, and the above approach relies on an indirect assessment of existing data coupled with clinical experience. The approach may change as new data become available.
Eosinophilic granulomatosis with polyangiitis
Eosinophilic granulomatosis with polyangiitis (EGPA) is a vasculitis characterized by disseminated necrotizing eosinophilic granulomas. EGPA is driven by a response similar to that seen in Th2-high asthma. Adult-onset asthma with sinusitis and allergic rhinitis is the most common EGPA presentation. Of all the biologics, mepolizumab has been best studied as treatment for those with EGPA and asthma symptoms. One small study demonstrated disease remission in 8 of 10 cases (Moosig F, et al. Ann Intern Med. 2011 Sep 6;155[5]:341-3). However, many of these patients relapsed after discontinuing therapy.
Eosinophilic esophagitis
Recent reports demonstrated a large portion of adults with a
diagnosis of eosinophilic
esophagitis (EoE) also have a history of asthma. Currently, standard treatment is proton pump
inhibitors and diet modifications. The prevalence of EoE has increased with growing awareness of the disease. Unrecognized and untreated EoE can lead to devastating complications such as esophageal fibrosis, strictures, and food impaction. Similar to some of the above-mentioned syndromes,
EoE is also driven by a Th2 response and eosinophilic inflammation. A recent study in 2022 showed that 31% to 38% of
people with EoE had concomitant asthma (Dellon ES, et al. N Engl J Med. 2022 Dec 22;387 [25]:2317-30). In this population, a weekly dose of dupilumab, 300 mg, led
to a significant improvement in dysphagia symptoms and
histology when compared with placebo.
Allergic bronchopulmonary aspergillosis
Despite its low prevalence worldwide, allergic bronchopulmonary aspergillosis (ABPA) is frequently encountered when managing severe asthma. Current treatment is long-term, relatively high dose systemic corticosteroids. In light of their unfavorable side effect profile, steroid-sparing approaches are being sought. Dupilumab, omalizumab, mepolizumab, and benralizumab have all been tested for their effects on ABPA. Thus far, mepolizumab has the most convincing evidence to support its use for asthma with concomitant ABPA, mainly because it has the most rapid onset of action. Up to 90% of patients with ABPA were able to stop systemic steroids between 2 and 14 months after starting mepolizumab (Schleich F, et al. J Allergy Clin Immunol. 2020 Jul-Aug;8[7]:2412-3.e2).
Bronchiectasis
Asthma and bronchiectasis can coexist in up to 77% of patients. Typically, the pathophysiology behind bronchiectasis is focused around neutrophilic inflammation. New evidence suggests some patients with bronchiectasis, usually in the setting of comorbid adult-onset asthma, demonstrate an eosinophilic Th-2 response. The association is seen more commonly in female patients, the elderly, and nonsmokers. A small prospective study with four patients with severe asthma and bronchiectasis showed significant improvement with less exacerbations, increased pre-bronchodilator FEV1, and a reduction of serum and sputum eosinophils after starting mepolizumab treatment (Carpagnano GE, et al. J Asthma Allergy. 2019 Mar 5;12:83-90). Clinical trials designed to clarify the role for biologics for asthma with co-morbid bronchiectasis are currently underway.
Dr. Hossri and Dr. Ivashchuk are with UTHealth Houston –Texas Medical Center, Department of Internal Medicine; Division of Pulmonary, Critical Care, and Sleep Medicine.
As new treatments for specific moderate to severe asthma phenotypes have been developed, management decisions have grown more complicated. The treatment indications for asthma are clear; however, there is overlap with certain therapeutics that target the same pathway with similar end results. because it is not a one-size-fits-all approach that follows a rigid algorithm. Instead, it is a customized treatment plan that accounts for patient-specific risk factors and comorbidities.
Comorbidities commonly associated with asthma include atopic dermatitis, chronic rhinosinusitis with nasal polyposis, eosinophilic granulomatosis with polyangiitis, eosinophilic esophagitis, bronchiectasis and allergic bronchopulmonary aspergillosis. While we lack consensus or a universally accepted treatment algorithm for treating asthma when these comorbidities are present, recent evidence helps guide us to which therapies work best.
Atopic dermatitis
There is a higher prevalence of asthma in patients with atopic dermatitis. A concept called the “atopic march” refers to the progression of childhood atopic dermatitis to manifestations such as asthma, food allergies, and hay fever. The more severe the atopic dermatitis is in childhood, the higher the risk for asthma later on in life. The data on the biologic pathogenesis of atopic dermatitis point to the involvement of interleukins – interleukin (IL)-4 and IL 13 (Silverberg JI. Ann Allergy Asthma Immunol. 2019;123[2]:144-51).
These same interleukins are active in what is called “Th2-high” asthma. The activation of Th2 cells in the inflammatory pathway occurs in atopic dermatitis and asthma irrespective of immunoglobulin E levels. Preliminary data show therapies that target IL-13 alone are effective for treating asthma with comorbid atopic dermatitis but those blocking both IL-4 and IL-13, like dupilumab, are superior. Both interleukins are considered pivotal in the Th-2 pathway. This suggests that dual inhibition is an integral component in the treatment of moderate to severe atopic dermatitis with asthma. Analysis of other Th2 mediators, such as mepolizumab (IL-5 antagonist) and omalizumab (anti-IgE) have shown minimal efficacy, further supporting the use of dupilumab (Guttman-Yassky E, et al. J Allergy Clin. Immunol. 2019 Jan;143[1]:155-72).
Chronic rhinosinusitis with nasal polyposis
The “unified airway” concept holds that because the upper airways (nasal mucosa, pharynx, and larynx) are in direct communication with the lower airways (bronchi and bronchioles). This would explain the correlation between chronic rhinosinusitis with nasal polyposis (CRSwNP) and asthma. Many studies also show the severity of one disease increases the severity of the other.
Patients with both CRSwNP and asthma typically experience a more treatment-resistant course characterized by higher rates of corticosteroid dependence and nasal polyposis recurrences when compared with asthma alone (Laidlaw TM, et al. J Allergy Clin Immunol. 2021 Mar;9[3]:1133-41). They typically have Th2-high asthma and are usually eosinophilic. The optimal treatment approach is mindful of the unified airway concept. Large-scale studies demonstrate significant benefit when targeting IL-5, especially in those with bilateral nasal polyps, need for systemic steroids in the past 2 years, significant impairment in quality of life, loss of smell, and a concomitant diagnosis of asthma (Fokkens WJ, et al. Allergy. 2019 Dec;74[12]:2312). Although data are inconsistent, there is enough evidence to suggest dupilumab be considered for those with eosinophilic asthma and CRSwNP along with atopy, atopic dermatitis, and/or high FeNO levels. In those without atopic symptoms, an anti-IL5/anti-IL5R (mainly mepolizumab and benralizumab) is preferred. Having said this, direct comparative analyses between biologics are lacking, and the above approach relies on an indirect assessment of existing data coupled with clinical experience. The approach may change as new data become available.
Eosinophilic granulomatosis with polyangiitis
Eosinophilic granulomatosis with polyangiitis (EGPA) is a vasculitis characterized by disseminated necrotizing eosinophilic granulomas. EGPA is driven by a response similar to that seen in Th2-high asthma. Adult-onset asthma with sinusitis and allergic rhinitis is the most common EGPA presentation. Of all the biologics, mepolizumab has been best studied as treatment for those with EGPA and asthma symptoms. One small study demonstrated disease remission in 8 of 10 cases (Moosig F, et al. Ann Intern Med. 2011 Sep 6;155[5]:341-3). However, many of these patients relapsed after discontinuing therapy.
Eosinophilic esophagitis
Recent reports demonstrated a large portion of adults with a
diagnosis of eosinophilic
esophagitis (EoE) also have a history of asthma. Currently, standard treatment is proton pump
inhibitors and diet modifications. The prevalence of EoE has increased with growing awareness of the disease. Unrecognized and untreated EoE can lead to devastating complications such as esophageal fibrosis, strictures, and food impaction. Similar to some of the above-mentioned syndromes,
EoE is also driven by a Th2 response and eosinophilic inflammation. A recent study in 2022 showed that 31% to 38% of
people with EoE had concomitant asthma (Dellon ES, et al. N Engl J Med. 2022 Dec 22;387 [25]:2317-30). In this population, a weekly dose of dupilumab, 300 mg, led
to a significant improvement in dysphagia symptoms and
histology when compared with placebo.
Allergic bronchopulmonary aspergillosis
Despite its low prevalence worldwide, allergic bronchopulmonary aspergillosis (ABPA) is frequently encountered when managing severe asthma. Current treatment is long-term, relatively high dose systemic corticosteroids. In light of their unfavorable side effect profile, steroid-sparing approaches are being sought. Dupilumab, omalizumab, mepolizumab, and benralizumab have all been tested for their effects on ABPA. Thus far, mepolizumab has the most convincing evidence to support its use for asthma with concomitant ABPA, mainly because it has the most rapid onset of action. Up to 90% of patients with ABPA were able to stop systemic steroids between 2 and 14 months after starting mepolizumab (Schleich F, et al. J Allergy Clin Immunol. 2020 Jul-Aug;8[7]:2412-3.e2).
Bronchiectasis
Asthma and bronchiectasis can coexist in up to 77% of patients. Typically, the pathophysiology behind bronchiectasis is focused around neutrophilic inflammation. New evidence suggests some patients with bronchiectasis, usually in the setting of comorbid adult-onset asthma, demonstrate an eosinophilic Th-2 response. The association is seen more commonly in female patients, the elderly, and nonsmokers. A small prospective study with four patients with severe asthma and bronchiectasis showed significant improvement with less exacerbations, increased pre-bronchodilator FEV1, and a reduction of serum and sputum eosinophils after starting mepolizumab treatment (Carpagnano GE, et al. J Asthma Allergy. 2019 Mar 5;12:83-90). Clinical trials designed to clarify the role for biologics for asthma with co-morbid bronchiectasis are currently underway.
Dr. Hossri and Dr. Ivashchuk are with UTHealth Houston –Texas Medical Center, Department of Internal Medicine; Division of Pulmonary, Critical Care, and Sleep Medicine.
As new treatments for specific moderate to severe asthma phenotypes have been developed, management decisions have grown more complicated. The treatment indications for asthma are clear; however, there is overlap with certain therapeutics that target the same pathway with similar end results. because it is not a one-size-fits-all approach that follows a rigid algorithm. Instead, it is a customized treatment plan that accounts for patient-specific risk factors and comorbidities.
Comorbidities commonly associated with asthma include atopic dermatitis, chronic rhinosinusitis with nasal polyposis, eosinophilic granulomatosis with polyangiitis, eosinophilic esophagitis, bronchiectasis and allergic bronchopulmonary aspergillosis. While we lack consensus or a universally accepted treatment algorithm for treating asthma when these comorbidities are present, recent evidence helps guide us to which therapies work best.
Atopic dermatitis
There is a higher prevalence of asthma in patients with atopic dermatitis. A concept called the “atopic march” refers to the progression of childhood atopic dermatitis to manifestations such as asthma, food allergies, and hay fever. The more severe the atopic dermatitis is in childhood, the higher the risk for asthma later on in life. The data on the biologic pathogenesis of atopic dermatitis point to the involvement of interleukins – interleukin (IL)-4 and IL 13 (Silverberg JI. Ann Allergy Asthma Immunol. 2019;123[2]:144-51).
These same interleukins are active in what is called “Th2-high” asthma. The activation of Th2 cells in the inflammatory pathway occurs in atopic dermatitis and asthma irrespective of immunoglobulin E levels. Preliminary data show therapies that target IL-13 alone are effective for treating asthma with comorbid atopic dermatitis but those blocking both IL-4 and IL-13, like dupilumab, are superior. Both interleukins are considered pivotal in the Th-2 pathway. This suggests that dual inhibition is an integral component in the treatment of moderate to severe atopic dermatitis with asthma. Analysis of other Th2 mediators, such as mepolizumab (IL-5 antagonist) and omalizumab (anti-IgE) have shown minimal efficacy, further supporting the use of dupilumab (Guttman-Yassky E, et al. J Allergy Clin. Immunol. 2019 Jan;143[1]:155-72).
Chronic rhinosinusitis with nasal polyposis
The “unified airway” concept holds that because the upper airways (nasal mucosa, pharynx, and larynx) are in direct communication with the lower airways (bronchi and bronchioles). This would explain the correlation between chronic rhinosinusitis with nasal polyposis (CRSwNP) and asthma. Many studies also show the severity of one disease increases the severity of the other.
Patients with both CRSwNP and asthma typically experience a more treatment-resistant course characterized by higher rates of corticosteroid dependence and nasal polyposis recurrences when compared with asthma alone (Laidlaw TM, et al. J Allergy Clin Immunol. 2021 Mar;9[3]:1133-41). They typically have Th2-high asthma and are usually eosinophilic. The optimal treatment approach is mindful of the unified airway concept. Large-scale studies demonstrate significant benefit when targeting IL-5, especially in those with bilateral nasal polyps, need for systemic steroids in the past 2 years, significant impairment in quality of life, loss of smell, and a concomitant diagnosis of asthma (Fokkens WJ, et al. Allergy. 2019 Dec;74[12]:2312). Although data are inconsistent, there is enough evidence to suggest dupilumab be considered for those with eosinophilic asthma and CRSwNP along with atopy, atopic dermatitis, and/or high FeNO levels. In those without atopic symptoms, an anti-IL5/anti-IL5R (mainly mepolizumab and benralizumab) is preferred. Having said this, direct comparative analyses between biologics are lacking, and the above approach relies on an indirect assessment of existing data coupled with clinical experience. The approach may change as new data become available.
Eosinophilic granulomatosis with polyangiitis
Eosinophilic granulomatosis with polyangiitis (EGPA) is a vasculitis characterized by disseminated necrotizing eosinophilic granulomas. EGPA is driven by a response similar to that seen in Th2-high asthma. Adult-onset asthma with sinusitis and allergic rhinitis is the most common EGPA presentation. Of all the biologics, mepolizumab has been best studied as treatment for those with EGPA and asthma symptoms. One small study demonstrated disease remission in 8 of 10 cases (Moosig F, et al. Ann Intern Med. 2011 Sep 6;155[5]:341-3). However, many of these patients relapsed after discontinuing therapy.
Eosinophilic esophagitis
Recent reports demonstrated a large portion of adults with a
diagnosis of eosinophilic
esophagitis (EoE) also have a history of asthma. Currently, standard treatment is proton pump
inhibitors and diet modifications. The prevalence of EoE has increased with growing awareness of the disease. Unrecognized and untreated EoE can lead to devastating complications such as esophageal fibrosis, strictures, and food impaction. Similar to some of the above-mentioned syndromes,
EoE is also driven by a Th2 response and eosinophilic inflammation. A recent study in 2022 showed that 31% to 38% of
people with EoE had concomitant asthma (Dellon ES, et al. N Engl J Med. 2022 Dec 22;387 [25]:2317-30). In this population, a weekly dose of dupilumab, 300 mg, led
to a significant improvement in dysphagia symptoms and
histology when compared with placebo.
Allergic bronchopulmonary aspergillosis
Despite its low prevalence worldwide, allergic bronchopulmonary aspergillosis (ABPA) is frequently encountered when managing severe asthma. Current treatment is long-term, relatively high dose systemic corticosteroids. In light of their unfavorable side effect profile, steroid-sparing approaches are being sought. Dupilumab, omalizumab, mepolizumab, and benralizumab have all been tested for their effects on ABPA. Thus far, mepolizumab has the most convincing evidence to support its use for asthma with concomitant ABPA, mainly because it has the most rapid onset of action. Up to 90% of patients with ABPA were able to stop systemic steroids between 2 and 14 months after starting mepolizumab (Schleich F, et al. J Allergy Clin Immunol. 2020 Jul-Aug;8[7]:2412-3.e2).
Bronchiectasis
Asthma and bronchiectasis can coexist in up to 77% of patients. Typically, the pathophysiology behind bronchiectasis is focused around neutrophilic inflammation. New evidence suggests some patients with bronchiectasis, usually in the setting of comorbid adult-onset asthma, demonstrate an eosinophilic Th-2 response. The association is seen more commonly in female patients, the elderly, and nonsmokers. A small prospective study with four patients with severe asthma and bronchiectasis showed significant improvement with less exacerbations, increased pre-bronchodilator FEV1, and a reduction of serum and sputum eosinophils after starting mepolizumab treatment (Carpagnano GE, et al. J Asthma Allergy. 2019 Mar 5;12:83-90). Clinical trials designed to clarify the role for biologics for asthma with co-morbid bronchiectasis are currently underway.
Use the SCAI stages to identify and treat cardiogenic shock
Cardiogenic shock (CS) is being recognized more often in critically ill patients. This increased prevalence is likely due to a better understanding of CS and the benefit of improving cardiac output (CO) to ensure adequate oxygen delivery (DO2).
CS is often, but not always, caused by a cardiac dysfunction. The heart is not able to provide adequate DO2 to the tissues. Hypoperfusion ensues. The body attempts to compensate for the poor perfusion by increasing heart rate, vasoconstriction, and shunting blood flow to vital organs. These compensatory mechanisms worsen perfusion by increasing myocardial ischemia which further worsens cardiac dysfunction. This is known as the downward spiral of CS (Ann Intern Med. 1999 Jul 6;131[1]).
There is a number of different etiologies for CS. Historically, acute myocardial infarctions (AMI) was the most common cause. In the last 20 years, AMI-induced CS has become less prevalent due to more aggressive reperfusion strategies. CS due to etiologies such as cardiomyopathy, myocarditis, right ventricle failure, and valvular pathologies have become more common. While the overarching goal is to restore DO2 to the tissue, the optimal treatment may differ based on the etiology of the CS. The Society for Cardiovascular Angiography and Intervention (SCAI) published CS classification stages in 2019 and then updated the stages 2022 (J Am Coll Cardiol. 2022 Mar 8;79[9]:933-46). In addition to the stages, there is now a three-axis model to address risk stratification. These classifications are a practically means of identifying and treating patients presenting with or concern for acute CS.
Stage A (At Risk) patients are not experiencing CS, but they are the at risk population. The patient’s hemodynamics, physical exam, and markers of hypoperfusion are normal. Stage A includes patients who have had a recent AMI or have heart failure.
Stage B (Beginning) patients have evidence of hemodynamic instability but are able to maintain tissue perfusion. These patients will have true or relative hypotension or tachycardia (in an attempt to maintain CO). Distal perfusion is adequate, but signs of ensuing decompensation (eg, elevated jugular venous pressure [JVP]) are present. Lactate is <2.0 mmol/L. Clinicians must be vigilant and treat these patients aggressively, so they do not decompensate further. It can be difficult to identify these patients because their blood pressure may be “normal,” but upon investigation, the blood pressure is actually a drop from the patient’s baseline.
Chronic heart failure patients with a history of depressed cardiac function will often have periods of cardiac decompensation between stages A and B. These patients are able to maintain perfusion for longer periods of time before further decompensation with hypoperfusion. If and when they do decompensate, they will often have a steep downward trajectory, so it is advantageous to the patient to be aggressive early.
Stage C (Classic) patients have evidence of tissue hypoperfusion. While these patients will often have true or relative hypotension, it is not a definition of stage C. These patients have evidence of volume overload with elevated JVP and rales throughout their lung fields. They will have poor distal perfusion and cool extremities that may become mottled. Lactate is ≥ 2 mmol/L. B-type natriuretic peptide (BNP) and liver function test (LFTs) results are elevated, and urine output is diminished. If a pulmonary arterial catheter is placed (highly recommended), the cardiac index (CI) is < 2.2 L/min/m2 and the pulmonary capillary wedge pressure (PCWP) is > 15 mm Hg. These patients look like what many clinicians think of when they think of CS.
These patients need better tissue perfusion. Inotropic support is needed to augment CO and DO2. Pharmacologic support is often the initial step. These patients also benefit from volume removal. This is usually accomplished with aggressive diuresis with a loop diuretic.
Stage D (Deteriorating) patients have failed initial treatment with single inotropic support. Hypoperfusion is not getting better and is often worsening. Lactate is staying > 2 mmol/L or rising. BNP and LFTs are also rising. These patients require additional inotropes and usually need vasopressors. Mechanical cardiac support (MCS) is often needed in addition to pharmacologic inotropic support.
Stage E (Extremis) patients have actual or impending circulatory collapse. These patients are peri-arrest with profound hypotension, lactic acidosis (often > 8 mmol/L), and unconsciousness. These patients are worsening despite multiple strategies to augment CO and DO2. These patients will likely die without emergent veno-arterial (VA) extracorporeal membrane oxygenation (ECMO). The goal of treatment is to stabilize the patient as quickly as possible to prevent cardiac arrest.
In addition to the stage of CS, SCAI developed the three-axis model of risk stratification as a conceptual model to be used for evaluation and prognostication. Etiology and phenotype, shock severity, and risk modifiers are factors related to patient outcomes from CS. This model is a way to individualize treatment to a specific patient.
Shock severity: What is the patient’s shock stage? What are the hemodynamics and metabolic abnormalities? What are the doses of the inotropes or vasopressors? Risk goes up with higher shock stages and vasoactive agent doses and worsening metabolic disturbances or hemodynamics.
Phenotype and etiology: what is the clinical etiology of the patient’s CS? Is this acute or acute on chronic? Which ventricle is involved? Is this cardiac driven or are other organs the driving factor? Single ventricle involvement is better than bi-ventricular failure. Cardiogenic collapse due to an overdose may have a better outcome than a massive AMI.
Risk modifiers: how old is the patient? What are the comorbidities? Did the patient have a cardiac arrest? What is the patient’s mental status? Some factors are modifiable, but others are not. The concept of chronologic vs. physiologic age may come into play. A frail 40 year old with stage 4 cancer and end stage renal failure may be assessed differently than a 70 year old with mild hypertension and an AMI.
The SCAI stages of CS are a pragmatic way to assess patients with an acute presentation of CS. These stages have defined criteria and treatment recommendations for all patients. The three-axis model allows the clinician to individualize patient care based on shock severity, etiology/phenotype, and risk modification. The goal of these stages is to identify and aggressively treat patients with CS, as well as identify when treatment is failing and additional therapies may be needed.
Dr. Gaillard is Associate Professor in the Departments of Anesthesiology, Section on Critical Care; Internal Medicine, Section on Pulmonology, Critical Care, Allergy, and Immunologic Diseases; and Emergency Medicine; Wake Forest School of Medicine, Winston-Salem, N.C.
Cardiogenic shock (CS) is being recognized more often in critically ill patients. This increased prevalence is likely due to a better understanding of CS and the benefit of improving cardiac output (CO) to ensure adequate oxygen delivery (DO2).
CS is often, but not always, caused by a cardiac dysfunction. The heart is not able to provide adequate DO2 to the tissues. Hypoperfusion ensues. The body attempts to compensate for the poor perfusion by increasing heart rate, vasoconstriction, and shunting blood flow to vital organs. These compensatory mechanisms worsen perfusion by increasing myocardial ischemia which further worsens cardiac dysfunction. This is known as the downward spiral of CS (Ann Intern Med. 1999 Jul 6;131[1]).
There is a number of different etiologies for CS. Historically, acute myocardial infarctions (AMI) was the most common cause. In the last 20 years, AMI-induced CS has become less prevalent due to more aggressive reperfusion strategies. CS due to etiologies such as cardiomyopathy, myocarditis, right ventricle failure, and valvular pathologies have become more common. While the overarching goal is to restore DO2 to the tissue, the optimal treatment may differ based on the etiology of the CS. The Society for Cardiovascular Angiography and Intervention (SCAI) published CS classification stages in 2019 and then updated the stages 2022 (J Am Coll Cardiol. 2022 Mar 8;79[9]:933-46). In addition to the stages, there is now a three-axis model to address risk stratification. These classifications are a practically means of identifying and treating patients presenting with or concern for acute CS.
Stage A (At Risk) patients are not experiencing CS, but they are the at risk population. The patient’s hemodynamics, physical exam, and markers of hypoperfusion are normal. Stage A includes patients who have had a recent AMI or have heart failure.
Stage B (Beginning) patients have evidence of hemodynamic instability but are able to maintain tissue perfusion. These patients will have true or relative hypotension or tachycardia (in an attempt to maintain CO). Distal perfusion is adequate, but signs of ensuing decompensation (eg, elevated jugular venous pressure [JVP]) are present. Lactate is <2.0 mmol/L. Clinicians must be vigilant and treat these patients aggressively, so they do not decompensate further. It can be difficult to identify these patients because their blood pressure may be “normal,” but upon investigation, the blood pressure is actually a drop from the patient’s baseline.
Chronic heart failure patients with a history of depressed cardiac function will often have periods of cardiac decompensation between stages A and B. These patients are able to maintain perfusion for longer periods of time before further decompensation with hypoperfusion. If and when they do decompensate, they will often have a steep downward trajectory, so it is advantageous to the patient to be aggressive early.
Stage C (Classic) patients have evidence of tissue hypoperfusion. While these patients will often have true or relative hypotension, it is not a definition of stage C. These patients have evidence of volume overload with elevated JVP and rales throughout their lung fields. They will have poor distal perfusion and cool extremities that may become mottled. Lactate is ≥ 2 mmol/L. B-type natriuretic peptide (BNP) and liver function test (LFTs) results are elevated, and urine output is diminished. If a pulmonary arterial catheter is placed (highly recommended), the cardiac index (CI) is < 2.2 L/min/m2 and the pulmonary capillary wedge pressure (PCWP) is > 15 mm Hg. These patients look like what many clinicians think of when they think of CS.
These patients need better tissue perfusion. Inotropic support is needed to augment CO and DO2. Pharmacologic support is often the initial step. These patients also benefit from volume removal. This is usually accomplished with aggressive diuresis with a loop diuretic.
Stage D (Deteriorating) patients have failed initial treatment with single inotropic support. Hypoperfusion is not getting better and is often worsening. Lactate is staying > 2 mmol/L or rising. BNP and LFTs are also rising. These patients require additional inotropes and usually need vasopressors. Mechanical cardiac support (MCS) is often needed in addition to pharmacologic inotropic support.
Stage E (Extremis) patients have actual or impending circulatory collapse. These patients are peri-arrest with profound hypotension, lactic acidosis (often > 8 mmol/L), and unconsciousness. These patients are worsening despite multiple strategies to augment CO and DO2. These patients will likely die without emergent veno-arterial (VA) extracorporeal membrane oxygenation (ECMO). The goal of treatment is to stabilize the patient as quickly as possible to prevent cardiac arrest.
In addition to the stage of CS, SCAI developed the three-axis model of risk stratification as a conceptual model to be used for evaluation and prognostication. Etiology and phenotype, shock severity, and risk modifiers are factors related to patient outcomes from CS. This model is a way to individualize treatment to a specific patient.
Shock severity: What is the patient’s shock stage? What are the hemodynamics and metabolic abnormalities? What are the doses of the inotropes or vasopressors? Risk goes up with higher shock stages and vasoactive agent doses and worsening metabolic disturbances or hemodynamics.
Phenotype and etiology: what is the clinical etiology of the patient’s CS? Is this acute or acute on chronic? Which ventricle is involved? Is this cardiac driven or are other organs the driving factor? Single ventricle involvement is better than bi-ventricular failure. Cardiogenic collapse due to an overdose may have a better outcome than a massive AMI.
Risk modifiers: how old is the patient? What are the comorbidities? Did the patient have a cardiac arrest? What is the patient’s mental status? Some factors are modifiable, but others are not. The concept of chronologic vs. physiologic age may come into play. A frail 40 year old with stage 4 cancer and end stage renal failure may be assessed differently than a 70 year old with mild hypertension and an AMI.
The SCAI stages of CS are a pragmatic way to assess patients with an acute presentation of CS. These stages have defined criteria and treatment recommendations for all patients. The three-axis model allows the clinician to individualize patient care based on shock severity, etiology/phenotype, and risk modification. The goal of these stages is to identify and aggressively treat patients with CS, as well as identify when treatment is failing and additional therapies may be needed.
Dr. Gaillard is Associate Professor in the Departments of Anesthesiology, Section on Critical Care; Internal Medicine, Section on Pulmonology, Critical Care, Allergy, and Immunologic Diseases; and Emergency Medicine; Wake Forest School of Medicine, Winston-Salem, N.C.
Cardiogenic shock (CS) is being recognized more often in critically ill patients. This increased prevalence is likely due to a better understanding of CS and the benefit of improving cardiac output (CO) to ensure adequate oxygen delivery (DO2).
CS is often, but not always, caused by a cardiac dysfunction. The heart is not able to provide adequate DO2 to the tissues. Hypoperfusion ensues. The body attempts to compensate for the poor perfusion by increasing heart rate, vasoconstriction, and shunting blood flow to vital organs. These compensatory mechanisms worsen perfusion by increasing myocardial ischemia which further worsens cardiac dysfunction. This is known as the downward spiral of CS (Ann Intern Med. 1999 Jul 6;131[1]).
There is a number of different etiologies for CS. Historically, acute myocardial infarctions (AMI) was the most common cause. In the last 20 years, AMI-induced CS has become less prevalent due to more aggressive reperfusion strategies. CS due to etiologies such as cardiomyopathy, myocarditis, right ventricle failure, and valvular pathologies have become more common. While the overarching goal is to restore DO2 to the tissue, the optimal treatment may differ based on the etiology of the CS. The Society for Cardiovascular Angiography and Intervention (SCAI) published CS classification stages in 2019 and then updated the stages 2022 (J Am Coll Cardiol. 2022 Mar 8;79[9]:933-46). In addition to the stages, there is now a three-axis model to address risk stratification. These classifications are a practically means of identifying and treating patients presenting with or concern for acute CS.
Stage A (At Risk) patients are not experiencing CS, but they are the at risk population. The patient’s hemodynamics, physical exam, and markers of hypoperfusion are normal. Stage A includes patients who have had a recent AMI or have heart failure.
Stage B (Beginning) patients have evidence of hemodynamic instability but are able to maintain tissue perfusion. These patients will have true or relative hypotension or tachycardia (in an attempt to maintain CO). Distal perfusion is adequate, but signs of ensuing decompensation (eg, elevated jugular venous pressure [JVP]) are present. Lactate is <2.0 mmol/L. Clinicians must be vigilant and treat these patients aggressively, so they do not decompensate further. It can be difficult to identify these patients because their blood pressure may be “normal,” but upon investigation, the blood pressure is actually a drop from the patient’s baseline.
Chronic heart failure patients with a history of depressed cardiac function will often have periods of cardiac decompensation between stages A and B. These patients are able to maintain perfusion for longer periods of time before further decompensation with hypoperfusion. If and when they do decompensate, they will often have a steep downward trajectory, so it is advantageous to the patient to be aggressive early.
Stage C (Classic) patients have evidence of tissue hypoperfusion. While these patients will often have true or relative hypotension, it is not a definition of stage C. These patients have evidence of volume overload with elevated JVP and rales throughout their lung fields. They will have poor distal perfusion and cool extremities that may become mottled. Lactate is ≥ 2 mmol/L. B-type natriuretic peptide (BNP) and liver function test (LFTs) results are elevated, and urine output is diminished. If a pulmonary arterial catheter is placed (highly recommended), the cardiac index (CI) is < 2.2 L/min/m2 and the pulmonary capillary wedge pressure (PCWP) is > 15 mm Hg. These patients look like what many clinicians think of when they think of CS.
These patients need better tissue perfusion. Inotropic support is needed to augment CO and DO2. Pharmacologic support is often the initial step. These patients also benefit from volume removal. This is usually accomplished with aggressive diuresis with a loop diuretic.
Stage D (Deteriorating) patients have failed initial treatment with single inotropic support. Hypoperfusion is not getting better and is often worsening. Lactate is staying > 2 mmol/L or rising. BNP and LFTs are also rising. These patients require additional inotropes and usually need vasopressors. Mechanical cardiac support (MCS) is often needed in addition to pharmacologic inotropic support.
Stage E (Extremis) patients have actual or impending circulatory collapse. These patients are peri-arrest with profound hypotension, lactic acidosis (often > 8 mmol/L), and unconsciousness. These patients are worsening despite multiple strategies to augment CO and DO2. These patients will likely die without emergent veno-arterial (VA) extracorporeal membrane oxygenation (ECMO). The goal of treatment is to stabilize the patient as quickly as possible to prevent cardiac arrest.
In addition to the stage of CS, SCAI developed the three-axis model of risk stratification as a conceptual model to be used for evaluation and prognostication. Etiology and phenotype, shock severity, and risk modifiers are factors related to patient outcomes from CS. This model is a way to individualize treatment to a specific patient.
Shock severity: What is the patient’s shock stage? What are the hemodynamics and metabolic abnormalities? What are the doses of the inotropes or vasopressors? Risk goes up with higher shock stages and vasoactive agent doses and worsening metabolic disturbances or hemodynamics.
Phenotype and etiology: what is the clinical etiology of the patient’s CS? Is this acute or acute on chronic? Which ventricle is involved? Is this cardiac driven or are other organs the driving factor? Single ventricle involvement is better than bi-ventricular failure. Cardiogenic collapse due to an overdose may have a better outcome than a massive AMI.
Risk modifiers: how old is the patient? What are the comorbidities? Did the patient have a cardiac arrest? What is the patient’s mental status? Some factors are modifiable, but others are not. The concept of chronologic vs. physiologic age may come into play. A frail 40 year old with stage 4 cancer and end stage renal failure may be assessed differently than a 70 year old with mild hypertension and an AMI.
The SCAI stages of CS are a pragmatic way to assess patients with an acute presentation of CS. These stages have defined criteria and treatment recommendations for all patients. The three-axis model allows the clinician to individualize patient care based on shock severity, etiology/phenotype, and risk modification. The goal of these stages is to identify and aggressively treat patients with CS, as well as identify when treatment is failing and additional therapies may be needed.
Dr. Gaillard is Associate Professor in the Departments of Anesthesiology, Section on Critical Care; Internal Medicine, Section on Pulmonology, Critical Care, Allergy, and Immunologic Diseases; and Emergency Medicine; Wake Forest School of Medicine, Winston-Salem, N.C.
Add hands-on and interactive learning opportunities to your CHEST 2023 schedule
Explore the many ticketed sessions, and sign up early in case they sell out.
Simulation sessions
If you’re looking to gain hands-on exposure to equipment and tools that may not be available at your home institution, look no further than these simulation sessions. Choose from 25 different sessions offering firsthand experience with procedures relevant to your clinical practice.
“It’s a great opportunity to teach higher stakes procedures in a very low stakes environment where everybody’s comfortable and everybody’s learning from each other,” said Live Learning Subcommittee Chair, Nicholas Pastis, MD, FCCP.
CHEST 2023 simulation sessions will address clinical topics, including endobronchial ultrasound, cardiopulmonary exercise testing (CPET), intubation and cricothyrotomy, bronchoscopy management, and more. These sessions are taught by experts who use these real-world strategies in their daily practice.
CHEST 2022 attendee, Weston Bowker, MD, found value in the simulation courses he was able to attend in Nashville.
“It’s fantastic just to work with some of the leading experts in the field, especially from an interventional pulmonology standpoint. And, you truly get a different experience than maybe what your home institution offers,” he said.
Problem-based learning sessions
Exercise your critical thinking skills by working to resolve real-world clinical problems during these small group sessions. Refine your expertise on topics like lung cancer screening and staging, biologics in asthma, pneumonia, and more.
“Problem-based learning courses take a clinical problem or case study that is somewhat controversial to create a learning environment where the problem itself drives the learning with participants,” said CHEST 2023 Scientific Program Committee Chair, Aneesa Das, MD, FCCP. “These are small group sessions where learners can actively participate and collaborate to discuss various perspectives on the issue and work toward potential solutions.”
This year’s problem-based learning courses were chosen based on common controversies in chest medicine and current hot topics in medicine.
Dr. Das is excited for the Using CPET to Solve Your Difficult Cases course. “Cardiopulmonary exercise tests can sometimes be difficult even for seasoned physicians. This is always an amazing problem-based learning topic,” she added.
Meet the Professor sessions
Connect with leading chest medicine experts during these limited-capacity discussions capped at 24 registrants per session. Meet the Professor attendees will have the opportunity to engage in stimulating conversations on bronchiectasis, central airway obstructions, obesity hypoventilation, and sublobar resection.
“Meet the Professor sessions are a unique opportunity to interact and learn from a leader in the field in a very small group setting on a high-yield topic,” said Dr. Das. “These sessions allow for a learning environment that is personalized and intimate.”
Explore the many ticketed sessions, and sign up early in case they sell out.
Simulation sessions
If you’re looking to gain hands-on exposure to equipment and tools that may not be available at your home institution, look no further than these simulation sessions. Choose from 25 different sessions offering firsthand experience with procedures relevant to your clinical practice.
“It’s a great opportunity to teach higher stakes procedures in a very low stakes environment where everybody’s comfortable and everybody’s learning from each other,” said Live Learning Subcommittee Chair, Nicholas Pastis, MD, FCCP.
CHEST 2023 simulation sessions will address clinical topics, including endobronchial ultrasound, cardiopulmonary exercise testing (CPET), intubation and cricothyrotomy, bronchoscopy management, and more. These sessions are taught by experts who use these real-world strategies in their daily practice.
CHEST 2022 attendee, Weston Bowker, MD, found value in the simulation courses he was able to attend in Nashville.
“It’s fantastic just to work with some of the leading experts in the field, especially from an interventional pulmonology standpoint. And, you truly get a different experience than maybe what your home institution offers,” he said.
Problem-based learning sessions
Exercise your critical thinking skills by working to resolve real-world clinical problems during these small group sessions. Refine your expertise on topics like lung cancer screening and staging, biologics in asthma, pneumonia, and more.
“Problem-based learning courses take a clinical problem or case study that is somewhat controversial to create a learning environment where the problem itself drives the learning with participants,” said CHEST 2023 Scientific Program Committee Chair, Aneesa Das, MD, FCCP. “These are small group sessions where learners can actively participate and collaborate to discuss various perspectives on the issue and work toward potential solutions.”
This year’s problem-based learning courses were chosen based on common controversies in chest medicine and current hot topics in medicine.
Dr. Das is excited for the Using CPET to Solve Your Difficult Cases course. “Cardiopulmonary exercise tests can sometimes be difficult even for seasoned physicians. This is always an amazing problem-based learning topic,” she added.
Meet the Professor sessions
Connect with leading chest medicine experts during these limited-capacity discussions capped at 24 registrants per session. Meet the Professor attendees will have the opportunity to engage in stimulating conversations on bronchiectasis, central airway obstructions, obesity hypoventilation, and sublobar resection.
“Meet the Professor sessions are a unique opportunity to interact and learn from a leader in the field in a very small group setting on a high-yield topic,” said Dr. Das. “These sessions allow for a learning environment that is personalized and intimate.”
Explore the many ticketed sessions, and sign up early in case they sell out.
Simulation sessions
If you’re looking to gain hands-on exposure to equipment and tools that may not be available at your home institution, look no further than these simulation sessions. Choose from 25 different sessions offering firsthand experience with procedures relevant to your clinical practice.
“It’s a great opportunity to teach higher stakes procedures in a very low stakes environment where everybody’s comfortable and everybody’s learning from each other,” said Live Learning Subcommittee Chair, Nicholas Pastis, MD, FCCP.
CHEST 2023 simulation sessions will address clinical topics, including endobronchial ultrasound, cardiopulmonary exercise testing (CPET), intubation and cricothyrotomy, bronchoscopy management, and more. These sessions are taught by experts who use these real-world strategies in their daily practice.
CHEST 2022 attendee, Weston Bowker, MD, found value in the simulation courses he was able to attend in Nashville.
“It’s fantastic just to work with some of the leading experts in the field, especially from an interventional pulmonology standpoint. And, you truly get a different experience than maybe what your home institution offers,” he said.
Problem-based learning sessions
Exercise your critical thinking skills by working to resolve real-world clinical problems during these small group sessions. Refine your expertise on topics like lung cancer screening and staging, biologics in asthma, pneumonia, and more.
“Problem-based learning courses take a clinical problem or case study that is somewhat controversial to create a learning environment where the problem itself drives the learning with participants,” said CHEST 2023 Scientific Program Committee Chair, Aneesa Das, MD, FCCP. “These are small group sessions where learners can actively participate and collaborate to discuss various perspectives on the issue and work toward potential solutions.”
This year’s problem-based learning courses were chosen based on common controversies in chest medicine and current hot topics in medicine.
Dr. Das is excited for the Using CPET to Solve Your Difficult Cases course. “Cardiopulmonary exercise tests can sometimes be difficult even for seasoned physicians. This is always an amazing problem-based learning topic,” she added.
Meet the Professor sessions
Connect with leading chest medicine experts during these limited-capacity discussions capped at 24 registrants per session. Meet the Professor attendees will have the opportunity to engage in stimulating conversations on bronchiectasis, central airway obstructions, obesity hypoventilation, and sublobar resection.
“Meet the Professor sessions are a unique opportunity to interact and learn from a leader in the field in a very small group setting on a high-yield topic,” said Dr. Das. “These sessions allow for a learning environment that is personalized and intimate.”