User login
AGA publishes CPU for AI in colon polyp diagnosis and management
The American Gastroenterological Association has published a Clinical Practice Update (CPU) on artificial intelligence (AI) for diagnosing and managing colorectal polyps.
The CPU, authored by Jason Samarasena, MD, of UCI Health, Orange, Calif., and colleagues, draws on recent studies and clinical experience to discuss ways that AI is already reshaping colonoscopy, and what opportunities may lie ahead.
“As with any emerging technology, there are important questions and challenges that need to be addressed to ensure that AI tools are introduced safely and effectively into clinical endoscopic practice, ”they wrote in Gastroenterology.
With advances in processing speed and deep-learning technology, AI “computer vision” can now analyze live video of a colonoscopy in progress, enabling computer-aided detection (CADe) and computer-aided diagnosis (CADx), which the panelists described as the two most important developments in the area.
CADe
“In the last several years, numerous prospective, multicenter studies have found that real-time use of AI CADe tools during colonoscopy leads to improvements in adenoma detection and other related performance metrics,” Dr. Samarasena and colleagues wrote.
CADe has yielded mixed success in real-world practice, however, with some studies reporting worse detection metrics after implementing the new technology. Dr. Samarasena and colleagues offered a variety of possible explanations for these findings, including a “ceiling effect” among highly adept endoscopists, reduced operator vigilance caused by false confidence in the technology, and potential confounding inherent to unblinded trials.
CADe may also increase health care costs and burden, they suggested, as the technology tends to catch small benign polyps, prompting unnecessary resections and shortened colonoscopy surveillance intervals.
CADx
The above, unintended consequences of CADe may be counteracted by CADx, which uses computer vision to predict which lesions have benign histology, enabling “resect-and discard” or “diagnose-and-leave” strategies.
Such approaches could significantly reduce rates of polypectomy and/or histopathology, saving an estimated $33 million–150 million per year, according to the update.
Results of real-time CADx clinical trials have been “encouraging,” Dr. Samarasena and colleagues wrote, noting that emerging technology–compatible white-light endoscopy can achieve a negative predictive value of almost 98% for lesions less than 5 mm in diameter, potentially reducing polypectomy rate by almost half.
“Increasing endoscopist confidence in optical diagnosis may be an important step toward broader implementation of leave in situ and resect-and-discard strategies, but successful implementation will also require CADx tools that seamlessly integrate the endoscopic work flow, without the need for image enhancement or magnification,” the panelists wrote.
Reimbursement models may also need to be reworked, they suggested, as many GI practices depend on a steady stream of revenue from pathology services.
Computer-aided quality assessment systems
Beyond optical detection and diagnosis, AI tools are also being developed to improve colonoscopy technique.
Investigators are studying quality assessment systems that use AI offer feedback on a range of endoscopist skills, including colonic-fold evaluation, level of mucosal exposure, and withdrawal time, the latter of which is visualized by a “speedometer” that “paints” the mucosa with “a graphical representation of the colon.”
“In the future, these types of AI-based systems may support trainees and lower-performing endoscopists to reduce exposure errors and, more broadly, may empower physician practices and hospital systems with more nuanced and actionable data on an array of factors that contribute to colonoscopy quality,” the panelists wrote.
Looking ahead
Dr. Samarasena and colleagues concluded by suggesting that the AI tools in usage and development are just the beginning of a wave of technology that will revolutionize how colonoscopies are performed.
“Eventually, we predict an AI suite of tools for colonoscopy will seem indispensable, as a powerful adjunct to support safe and efficient clinical practice,” they wrote. “As technological innovation progresses, we can expect that the future for AI in endoscopy will be a hybrid model, where the unique capabilities of physicians and our AI tools will be seamlessly intertwined to optimize patient care.”
This CPU was commissioned and approved by the AGA Institute Clinical Practice Updates Committee and the AGA Governing Board. The investigators disclosed relationships with Olympus, Neptune Medical, Conmed, and others.
The American Gastroenterological Association has published a Clinical Practice Update (CPU) on artificial intelligence (AI) for diagnosing and managing colorectal polyps.
The CPU, authored by Jason Samarasena, MD, of UCI Health, Orange, Calif., and colleagues, draws on recent studies and clinical experience to discuss ways that AI is already reshaping colonoscopy, and what opportunities may lie ahead.
“As with any emerging technology, there are important questions and challenges that need to be addressed to ensure that AI tools are introduced safely and effectively into clinical endoscopic practice, ”they wrote in Gastroenterology.
With advances in processing speed and deep-learning technology, AI “computer vision” can now analyze live video of a colonoscopy in progress, enabling computer-aided detection (CADe) and computer-aided diagnosis (CADx), which the panelists described as the two most important developments in the area.
CADe
“In the last several years, numerous prospective, multicenter studies have found that real-time use of AI CADe tools during colonoscopy leads to improvements in adenoma detection and other related performance metrics,” Dr. Samarasena and colleagues wrote.
CADe has yielded mixed success in real-world practice, however, with some studies reporting worse detection metrics after implementing the new technology. Dr. Samarasena and colleagues offered a variety of possible explanations for these findings, including a “ceiling effect” among highly adept endoscopists, reduced operator vigilance caused by false confidence in the technology, and potential confounding inherent to unblinded trials.
CADe may also increase health care costs and burden, they suggested, as the technology tends to catch small benign polyps, prompting unnecessary resections and shortened colonoscopy surveillance intervals.
CADx
The above, unintended consequences of CADe may be counteracted by CADx, which uses computer vision to predict which lesions have benign histology, enabling “resect-and discard” or “diagnose-and-leave” strategies.
Such approaches could significantly reduce rates of polypectomy and/or histopathology, saving an estimated $33 million–150 million per year, according to the update.
Results of real-time CADx clinical trials have been “encouraging,” Dr. Samarasena and colleagues wrote, noting that emerging technology–compatible white-light endoscopy can achieve a negative predictive value of almost 98% for lesions less than 5 mm in diameter, potentially reducing polypectomy rate by almost half.
“Increasing endoscopist confidence in optical diagnosis may be an important step toward broader implementation of leave in situ and resect-and-discard strategies, but successful implementation will also require CADx tools that seamlessly integrate the endoscopic work flow, without the need for image enhancement or magnification,” the panelists wrote.
Reimbursement models may also need to be reworked, they suggested, as many GI practices depend on a steady stream of revenue from pathology services.
Computer-aided quality assessment systems
Beyond optical detection and diagnosis, AI tools are also being developed to improve colonoscopy technique.
Investigators are studying quality assessment systems that use AI offer feedback on a range of endoscopist skills, including colonic-fold evaluation, level of mucosal exposure, and withdrawal time, the latter of which is visualized by a “speedometer” that “paints” the mucosa with “a graphical representation of the colon.”
“In the future, these types of AI-based systems may support trainees and lower-performing endoscopists to reduce exposure errors and, more broadly, may empower physician practices and hospital systems with more nuanced and actionable data on an array of factors that contribute to colonoscopy quality,” the panelists wrote.
Looking ahead
Dr. Samarasena and colleagues concluded by suggesting that the AI tools in usage and development are just the beginning of a wave of technology that will revolutionize how colonoscopies are performed.
“Eventually, we predict an AI suite of tools for colonoscopy will seem indispensable, as a powerful adjunct to support safe and efficient clinical practice,” they wrote. “As technological innovation progresses, we can expect that the future for AI in endoscopy will be a hybrid model, where the unique capabilities of physicians and our AI tools will be seamlessly intertwined to optimize patient care.”
This CPU was commissioned and approved by the AGA Institute Clinical Practice Updates Committee and the AGA Governing Board. The investigators disclosed relationships with Olympus, Neptune Medical, Conmed, and others.
The American Gastroenterological Association has published a Clinical Practice Update (CPU) on artificial intelligence (AI) for diagnosing and managing colorectal polyps.
The CPU, authored by Jason Samarasena, MD, of UCI Health, Orange, Calif., and colleagues, draws on recent studies and clinical experience to discuss ways that AI is already reshaping colonoscopy, and what opportunities may lie ahead.
“As with any emerging technology, there are important questions and challenges that need to be addressed to ensure that AI tools are introduced safely and effectively into clinical endoscopic practice, ”they wrote in Gastroenterology.
With advances in processing speed and deep-learning technology, AI “computer vision” can now analyze live video of a colonoscopy in progress, enabling computer-aided detection (CADe) and computer-aided diagnosis (CADx), which the panelists described as the two most important developments in the area.
CADe
“In the last several years, numerous prospective, multicenter studies have found that real-time use of AI CADe tools during colonoscopy leads to improvements in adenoma detection and other related performance metrics,” Dr. Samarasena and colleagues wrote.
CADe has yielded mixed success in real-world practice, however, with some studies reporting worse detection metrics after implementing the new technology. Dr. Samarasena and colleagues offered a variety of possible explanations for these findings, including a “ceiling effect” among highly adept endoscopists, reduced operator vigilance caused by false confidence in the technology, and potential confounding inherent to unblinded trials.
CADe may also increase health care costs and burden, they suggested, as the technology tends to catch small benign polyps, prompting unnecessary resections and shortened colonoscopy surveillance intervals.
CADx
The above, unintended consequences of CADe may be counteracted by CADx, which uses computer vision to predict which lesions have benign histology, enabling “resect-and discard” or “diagnose-and-leave” strategies.
Such approaches could significantly reduce rates of polypectomy and/or histopathology, saving an estimated $33 million–150 million per year, according to the update.
Results of real-time CADx clinical trials have been “encouraging,” Dr. Samarasena and colleagues wrote, noting that emerging technology–compatible white-light endoscopy can achieve a negative predictive value of almost 98% for lesions less than 5 mm in diameter, potentially reducing polypectomy rate by almost half.
“Increasing endoscopist confidence in optical diagnosis may be an important step toward broader implementation of leave in situ and resect-and-discard strategies, but successful implementation will also require CADx tools that seamlessly integrate the endoscopic work flow, without the need for image enhancement or magnification,” the panelists wrote.
Reimbursement models may also need to be reworked, they suggested, as many GI practices depend on a steady stream of revenue from pathology services.
Computer-aided quality assessment systems
Beyond optical detection and diagnosis, AI tools are also being developed to improve colonoscopy technique.
Investigators are studying quality assessment systems that use AI offer feedback on a range of endoscopist skills, including colonic-fold evaluation, level of mucosal exposure, and withdrawal time, the latter of which is visualized by a “speedometer” that “paints” the mucosa with “a graphical representation of the colon.”
“In the future, these types of AI-based systems may support trainees and lower-performing endoscopists to reduce exposure errors and, more broadly, may empower physician practices and hospital systems with more nuanced and actionable data on an array of factors that contribute to colonoscopy quality,” the panelists wrote.
Looking ahead
Dr. Samarasena and colleagues concluded by suggesting that the AI tools in usage and development are just the beginning of a wave of technology that will revolutionize how colonoscopies are performed.
“Eventually, we predict an AI suite of tools for colonoscopy will seem indispensable, as a powerful adjunct to support safe and efficient clinical practice,” they wrote. “As technological innovation progresses, we can expect that the future for AI in endoscopy will be a hybrid model, where the unique capabilities of physicians and our AI tools will be seamlessly intertwined to optimize patient care.”
This CPU was commissioned and approved by the AGA Institute Clinical Practice Updates Committee and the AGA Governing Board. The investigators disclosed relationships with Olympus, Neptune Medical, Conmed, and others.
FROM GASTROENTEROLOGY
The steep costs of disrupting gut-barrier harmony
An interview with Elena Ivanina, DO, MPH
From Ayurveda to the teachings of Hippocrates, medicine’s earliest traditions advanced a belief that the gut was the foundation of all health and disease. It wasn’t until recently, however, that Western medicine has adopted the notion of gut-barrier dysfunction as a pathologic phenomenon critical to not only digestive health but also chronic allergic, inflammatory, and autoimmune disease.
To learn more, Medscape contributor Akash Goel, MD, interviewed Elena Ivanina, DO, MPH, an integrative gastroenterologist, on the role of the gut barrier. Dr. Ivanina is the founder of the Center for Integrative Gut Health and the former director of Neurogastroenterology and Motility at Lenox Hill Hospital in New York. She runs the educational platform for all things gut health, gutlove.com.
What is the role of the gut barrier in overall health and disease?
The gut contains the human body’s largest interface between a person and their external environment. The actual interface is at the gut barrier, where there needs to be an ideal homeostasis and selectivity mechanism to allow the absorption of healthy nutrients, but on the other hand prevent the penetration of harmful microbes, food antigens, and other proinflammatory factors and toxins.
The gut barrier is made up of the mucus layer, gut microbiome, epithelial cells, and immune cells in the lamina propria. When this apparatus is disrupted by factors such as infection, low-fiber diet, antibiotics, and alcohol, then it cannot function normally to selectively keep out the harmful intraluminal substances.
Gut-barrier disruption leads to translocation of dangerous intraluminal components, such as bacteria and their components, into the gut wall and, most importantly, exposes the immune system to them. This causes improper immune activation and dysregulation, which has been shown to lead to various diseases, including gastrointestinal inflammatory disorders such as inflammatory bowel disease (IBD) and celiac disease, systemic autoimmune diseases such as multiple sclerosis and rheumatoid arthritis, and metabolic diseases such as obesity and diabetes.
Is disruption of this barrier what is usually referred to as “leaky gut”?
Leaky gut is a colloquial term for increased intestinal permeability or intestinal hyperpermeability. In a 2019 review article, Dr. Michael Camilleri exposes leaky gut as a term that can be misleading and confusing to the general population. It calls upon clinicians to have an increased awareness of the potential of barrier dysfunction in diseases, and to consider the barrier as a target for treatment.
Is leaky gut more of a mechanism of underlying chronic disease or is it a disease of its own?
Intestinal permeability is a pathophysiologic process in the gut with certain risk factors that in some conditions has been shown to precede chronic disease. There has not been any convincing evidence that it can be diagnosed and treated as its own entity, but research is ongoing.
In IBD, the Crohn’s and Colitis Canada Genetic, Environmental, Microbial Project research consortium has been studying individuals at increased risk for Crohn’s disease because of a first-degree family member with Crohn’s disease. They found an increased abundance of Ruminococcus torques in the microbiomes of at-risk individuals who went on to develop the disease. R. torques are mucin degraders that induce an increase in other mucin-using bacteria, which can contribute to gut-barrier compromise.
In other studies, patients have been found to have asymptomatic intestinal hyperpermeability years before their diagnosis of Crohn’s disease. This supports understanding more about the potential of intestinal hyperpermeability as its own diagnosis that, if addressed, could possibly prevent disease development.
The many possible sources of gut-barrier disruption
What causes leaky gut, and when should physicians and patients be suspicious if they have it?
There are many risk factors that have been associated with leaky gut in both human studies and animal studies, including acrolein (food toxin), aging, alcohol, antacid drugs, antibiotics, burn injury, chemotherapy, circadian rhythm disruption, corticosteroids, emulsifiers (food additives), strenuous exercise (≥ 2 hours) at 60% VO2 max, starvation, fructose, fructans, gliadin (wheat protein), high-fat diet, high-salt diet, high-sugar diet, hyperglycemia, low-fiber diet, nonsteroidal anti-inflammatory drugs, pesticide, proinflammatory cytokines, psychological stress, radiation, sleep deprivation, smoking, and sweeteners.
Patients may be completely asymptomatic with leaky gut. Physicians should be suspicious if there is a genetic predisposition to chronic disease or if any risk factors are unveiled after assessing diet and lifestyle exposures.
What is the role of the Western diet and processed food consumption in driving disruptions of the gut barrier?
The Western diet reduces gut-barrier mucus thickness, leading to increased gut permeability. People who consume a Western diet typically eat less than 15 grams of fiber per day, which is significantly less than many other cultures, including the hunter-gatherers of Tanzania (Hadza), who get 100 or more grams of fiber a day in their food.
With a fiber-depleted diet, gut microbiota that normally feed on fiber gradually disappear and other commensals shift their metabolism to degrade the gut-barrier mucus layer.
A low-fiber diet also decreases short-chain fatty acid production, which reduces production of mucus and affects tight junction regulation.
Emerging evidence on causality
New evidence is demonstrating that previous functional conditions of the gastrointestinal tract, like functional dyspepsia, are associated with abnormalities to the intestinal barrier. What is the association between conditions like functional dyspepsia and irritable bowel syndrome (IBS) with gut-barrier disruption?
Conditions such as functional dyspepsia and IBS are similar in that their pathophysiology is incompletely understood and likely attributable to contributions from many different underlying mechanisms. This makes it difficult for clinicians to explain the condition to patients and often to treat without specific therapeutic targets.
Emerging evidence with new diagnostic tools, such as confocal laser endomicroscopy, has demonstrated altered mucosal barrier function in both conditions.
In patients with IBS who have a suspected food intolerance, studies looking at exposure to the food antigens found that the food caused immediate breaks, increased intervillous spaces, and increased inflammatory cells in the gut mucosa. These changes were associated with patient responses to exclusion diets.
In functional dyspepsia, another study, using confocal laser endomicroscopy, has shown that affected patients have significantly greater epithelial gap density in the duodenum, compared with healthy controls. There was also impaired duodenal-epithelial barrier integrity and evidence of increased cellular pyroptosis in the duodenal mucosa.
These findings suggest that while IBS and functional dyspepsia are still likely multifactorial, there may be a common preclinical state that can be further investigated as far as preventing its development and using it as a therapeutic target.
What diagnostic testing are you using to determine whether patients have disruptions to the gut barrier? Are they validated or more experimental?
There are various testing strategies that have been used in research to diagnose intestinal hyperpermeability. In a 2021 analysis, Dr. Michael Camilleri found that the optimal probes for measuring small intestinal and colonic permeability are the mass excreted of 13C-mannitol at 0-2 hours and lactulose during 2-8 hours or sucralose during 8-24 hours. Studies looking at postinfectious IBS have incorporated elevated urinary lactulose/mannitol ratios. Dr. Alessio Fasano and others have looked at using zonulin as a biomarker of impaired gut-barrier function. These tests are still considered experimental.
Is there an association between alterations in the gut microbiome and gut-barrier disruption?
There is an integral relationship between the gut microbiome and gut-barrier function, and dysbiosis can disrupt gut-barrier functionality.
The microbiota produce a variety of metabolites in close proximity to the gut epithelium, impacting gut-barrier function and immune response. For example, short-chain fatty acids produced by Bifidobacterium, Bacteroides, Enterobacter, Faecalibacterium, and Roseburia species impact host immune cell differentiation and metabolism as well as influence susceptibility to pathogens.
Studies have shown that sodium butyrate significantly improves epithelial-barrier function. Other experiments have used transplantation of the intestinal microbiota to show that introduction of certain microbial phenotypes can significantly increase gut permeability.
Practical advice for clinicians and patients
How do you advise patients to avoid gut-barrier disruption?
It is important to educate and counsel patients about the long list of risk factors, many of which are closely related to a Western diet and lifestyle, which can increase their risk for leaky gut.
Once one has it, can it be repaired? Can you share a bit about your protocols in general terms?
Many interventions have been shown to improve intestinal permeability. They include berberine, butyrate, caloric restriction and fasting, curcumin, dietary fiber (prebiotics), moderate exercise, fermented food, fish oil, glutamine, quercetin, probiotics, vagus nerve stimulation, vitamin D, and zinc.
Protocols have to be tailored to patients and their risk factors, diet, and lifestyle.
What are some tips from a nutrition and lifestyle standpoint that patients can follow to ensure a robust gut barrier?
It is important to emphasize a high-fiber diet with naturally fermented food, incorporating time-restricted eating, such as eating an early dinner and nothing else before bedtime, a moderate exercise routine, and gut-brain modulation with techniques such as acupuncture that can incorporate vagus nerve stimulation. Limited safe precision supplementation can be discussed on an individual basis based on the patient’s interest, additional testing, and other existing health conditions.
Dr. Akash Goel is a clinical assistant professor of medicine at Weill Cornell in gastroenterology and hepatology. He has disclosed no relevant financial relationships. His work has appeared on networks and publications such as CNN, The New York Times, Time Magazine, and Financial Times. He has a deep interest in nutrition, food as medicine, and the intersection between the gut microbiome and human health.
A version of this article appeared on Medscape.com.
An interview with Elena Ivanina, DO, MPH
An interview with Elena Ivanina, DO, MPH
From Ayurveda to the teachings of Hippocrates, medicine’s earliest traditions advanced a belief that the gut was the foundation of all health and disease. It wasn’t until recently, however, that Western medicine has adopted the notion of gut-barrier dysfunction as a pathologic phenomenon critical to not only digestive health but also chronic allergic, inflammatory, and autoimmune disease.
To learn more, Medscape contributor Akash Goel, MD, interviewed Elena Ivanina, DO, MPH, an integrative gastroenterologist, on the role of the gut barrier. Dr. Ivanina is the founder of the Center for Integrative Gut Health and the former director of Neurogastroenterology and Motility at Lenox Hill Hospital in New York. She runs the educational platform for all things gut health, gutlove.com.
What is the role of the gut barrier in overall health and disease?
The gut contains the human body’s largest interface between a person and their external environment. The actual interface is at the gut barrier, where there needs to be an ideal homeostasis and selectivity mechanism to allow the absorption of healthy nutrients, but on the other hand prevent the penetration of harmful microbes, food antigens, and other proinflammatory factors and toxins.
The gut barrier is made up of the mucus layer, gut microbiome, epithelial cells, and immune cells in the lamina propria. When this apparatus is disrupted by factors such as infection, low-fiber diet, antibiotics, and alcohol, then it cannot function normally to selectively keep out the harmful intraluminal substances.
Gut-barrier disruption leads to translocation of dangerous intraluminal components, such as bacteria and their components, into the gut wall and, most importantly, exposes the immune system to them. This causes improper immune activation and dysregulation, which has been shown to lead to various diseases, including gastrointestinal inflammatory disorders such as inflammatory bowel disease (IBD) and celiac disease, systemic autoimmune diseases such as multiple sclerosis and rheumatoid arthritis, and metabolic diseases such as obesity and diabetes.
Is disruption of this barrier what is usually referred to as “leaky gut”?
Leaky gut is a colloquial term for increased intestinal permeability or intestinal hyperpermeability. In a 2019 review article, Dr. Michael Camilleri exposes leaky gut as a term that can be misleading and confusing to the general population. It calls upon clinicians to have an increased awareness of the potential of barrier dysfunction in diseases, and to consider the barrier as a target for treatment.
Is leaky gut more of a mechanism of underlying chronic disease or is it a disease of its own?
Intestinal permeability is a pathophysiologic process in the gut with certain risk factors that in some conditions has been shown to precede chronic disease. There has not been any convincing evidence that it can be diagnosed and treated as its own entity, but research is ongoing.
In IBD, the Crohn’s and Colitis Canada Genetic, Environmental, Microbial Project research consortium has been studying individuals at increased risk for Crohn’s disease because of a first-degree family member with Crohn’s disease. They found an increased abundance of Ruminococcus torques in the microbiomes of at-risk individuals who went on to develop the disease. R. torques are mucin degraders that induce an increase in other mucin-using bacteria, which can contribute to gut-barrier compromise.
In other studies, patients have been found to have asymptomatic intestinal hyperpermeability years before their diagnosis of Crohn’s disease. This supports understanding more about the potential of intestinal hyperpermeability as its own diagnosis that, if addressed, could possibly prevent disease development.
The many possible sources of gut-barrier disruption
What causes leaky gut, and when should physicians and patients be suspicious if they have it?
There are many risk factors that have been associated with leaky gut in both human studies and animal studies, including acrolein (food toxin), aging, alcohol, antacid drugs, antibiotics, burn injury, chemotherapy, circadian rhythm disruption, corticosteroids, emulsifiers (food additives), strenuous exercise (≥ 2 hours) at 60% VO2 max, starvation, fructose, fructans, gliadin (wheat protein), high-fat diet, high-salt diet, high-sugar diet, hyperglycemia, low-fiber diet, nonsteroidal anti-inflammatory drugs, pesticide, proinflammatory cytokines, psychological stress, radiation, sleep deprivation, smoking, and sweeteners.
Patients may be completely asymptomatic with leaky gut. Physicians should be suspicious if there is a genetic predisposition to chronic disease or if any risk factors are unveiled after assessing diet and lifestyle exposures.
What is the role of the Western diet and processed food consumption in driving disruptions of the gut barrier?
The Western diet reduces gut-barrier mucus thickness, leading to increased gut permeability. People who consume a Western diet typically eat less than 15 grams of fiber per day, which is significantly less than many other cultures, including the hunter-gatherers of Tanzania (Hadza), who get 100 or more grams of fiber a day in their food.
With a fiber-depleted diet, gut microbiota that normally feed on fiber gradually disappear and other commensals shift their metabolism to degrade the gut-barrier mucus layer.
A low-fiber diet also decreases short-chain fatty acid production, which reduces production of mucus and affects tight junction regulation.
Emerging evidence on causality
New evidence is demonstrating that previous functional conditions of the gastrointestinal tract, like functional dyspepsia, are associated with abnormalities to the intestinal barrier. What is the association between conditions like functional dyspepsia and irritable bowel syndrome (IBS) with gut-barrier disruption?
Conditions such as functional dyspepsia and IBS are similar in that their pathophysiology is incompletely understood and likely attributable to contributions from many different underlying mechanisms. This makes it difficult for clinicians to explain the condition to patients and often to treat without specific therapeutic targets.
Emerging evidence with new diagnostic tools, such as confocal laser endomicroscopy, has demonstrated altered mucosal barrier function in both conditions.
In patients with IBS who have a suspected food intolerance, studies looking at exposure to the food antigens found that the food caused immediate breaks, increased intervillous spaces, and increased inflammatory cells in the gut mucosa. These changes were associated with patient responses to exclusion diets.
In functional dyspepsia, another study, using confocal laser endomicroscopy, has shown that affected patients have significantly greater epithelial gap density in the duodenum, compared with healthy controls. There was also impaired duodenal-epithelial barrier integrity and evidence of increased cellular pyroptosis in the duodenal mucosa.
These findings suggest that while IBS and functional dyspepsia are still likely multifactorial, there may be a common preclinical state that can be further investigated as far as preventing its development and using it as a therapeutic target.
What diagnostic testing are you using to determine whether patients have disruptions to the gut barrier? Are they validated or more experimental?
There are various testing strategies that have been used in research to diagnose intestinal hyperpermeability. In a 2021 analysis, Dr. Michael Camilleri found that the optimal probes for measuring small intestinal and colonic permeability are the mass excreted of 13C-mannitol at 0-2 hours and lactulose during 2-8 hours or sucralose during 8-24 hours. Studies looking at postinfectious IBS have incorporated elevated urinary lactulose/mannitol ratios. Dr. Alessio Fasano and others have looked at using zonulin as a biomarker of impaired gut-barrier function. These tests are still considered experimental.
Is there an association between alterations in the gut microbiome and gut-barrier disruption?
There is an integral relationship between the gut microbiome and gut-barrier function, and dysbiosis can disrupt gut-barrier functionality.
The microbiota produce a variety of metabolites in close proximity to the gut epithelium, impacting gut-barrier function and immune response. For example, short-chain fatty acids produced by Bifidobacterium, Bacteroides, Enterobacter, Faecalibacterium, and Roseburia species impact host immune cell differentiation and metabolism as well as influence susceptibility to pathogens.
Studies have shown that sodium butyrate significantly improves epithelial-barrier function. Other experiments have used transplantation of the intestinal microbiota to show that introduction of certain microbial phenotypes can significantly increase gut permeability.
Practical advice for clinicians and patients
How do you advise patients to avoid gut-barrier disruption?
It is important to educate and counsel patients about the long list of risk factors, many of which are closely related to a Western diet and lifestyle, which can increase their risk for leaky gut.
Once one has it, can it be repaired? Can you share a bit about your protocols in general terms?
Many interventions have been shown to improve intestinal permeability. They include berberine, butyrate, caloric restriction and fasting, curcumin, dietary fiber (prebiotics), moderate exercise, fermented food, fish oil, glutamine, quercetin, probiotics, vagus nerve stimulation, vitamin D, and zinc.
Protocols have to be tailored to patients and their risk factors, diet, and lifestyle.
What are some tips from a nutrition and lifestyle standpoint that patients can follow to ensure a robust gut barrier?
It is important to emphasize a high-fiber diet with naturally fermented food, incorporating time-restricted eating, such as eating an early dinner and nothing else before bedtime, a moderate exercise routine, and gut-brain modulation with techniques such as acupuncture that can incorporate vagus nerve stimulation. Limited safe precision supplementation can be discussed on an individual basis based on the patient’s interest, additional testing, and other existing health conditions.
Dr. Akash Goel is a clinical assistant professor of medicine at Weill Cornell in gastroenterology and hepatology. He has disclosed no relevant financial relationships. His work has appeared on networks and publications such as CNN, The New York Times, Time Magazine, and Financial Times. He has a deep interest in nutrition, food as medicine, and the intersection between the gut microbiome and human health.
A version of this article appeared on Medscape.com.
From Ayurveda to the teachings of Hippocrates, medicine’s earliest traditions advanced a belief that the gut was the foundation of all health and disease. It wasn’t until recently, however, that Western medicine has adopted the notion of gut-barrier dysfunction as a pathologic phenomenon critical to not only digestive health but also chronic allergic, inflammatory, and autoimmune disease.
To learn more, Medscape contributor Akash Goel, MD, interviewed Elena Ivanina, DO, MPH, an integrative gastroenterologist, on the role of the gut barrier. Dr. Ivanina is the founder of the Center for Integrative Gut Health and the former director of Neurogastroenterology and Motility at Lenox Hill Hospital in New York. She runs the educational platform for all things gut health, gutlove.com.
What is the role of the gut barrier in overall health and disease?
The gut contains the human body’s largest interface between a person and their external environment. The actual interface is at the gut barrier, where there needs to be an ideal homeostasis and selectivity mechanism to allow the absorption of healthy nutrients, but on the other hand prevent the penetration of harmful microbes, food antigens, and other proinflammatory factors and toxins.
The gut barrier is made up of the mucus layer, gut microbiome, epithelial cells, and immune cells in the lamina propria. When this apparatus is disrupted by factors such as infection, low-fiber diet, antibiotics, and alcohol, then it cannot function normally to selectively keep out the harmful intraluminal substances.
Gut-barrier disruption leads to translocation of dangerous intraluminal components, such as bacteria and their components, into the gut wall and, most importantly, exposes the immune system to them. This causes improper immune activation and dysregulation, which has been shown to lead to various diseases, including gastrointestinal inflammatory disorders such as inflammatory bowel disease (IBD) and celiac disease, systemic autoimmune diseases such as multiple sclerosis and rheumatoid arthritis, and metabolic diseases such as obesity and diabetes.
Is disruption of this barrier what is usually referred to as “leaky gut”?
Leaky gut is a colloquial term for increased intestinal permeability or intestinal hyperpermeability. In a 2019 review article, Dr. Michael Camilleri exposes leaky gut as a term that can be misleading and confusing to the general population. It calls upon clinicians to have an increased awareness of the potential of barrier dysfunction in diseases, and to consider the barrier as a target for treatment.
Is leaky gut more of a mechanism of underlying chronic disease or is it a disease of its own?
Intestinal permeability is a pathophysiologic process in the gut with certain risk factors that in some conditions has been shown to precede chronic disease. There has not been any convincing evidence that it can be diagnosed and treated as its own entity, but research is ongoing.
In IBD, the Crohn’s and Colitis Canada Genetic, Environmental, Microbial Project research consortium has been studying individuals at increased risk for Crohn’s disease because of a first-degree family member with Crohn’s disease. They found an increased abundance of Ruminococcus torques in the microbiomes of at-risk individuals who went on to develop the disease. R. torques are mucin degraders that induce an increase in other mucin-using bacteria, which can contribute to gut-barrier compromise.
In other studies, patients have been found to have asymptomatic intestinal hyperpermeability years before their diagnosis of Crohn’s disease. This supports understanding more about the potential of intestinal hyperpermeability as its own diagnosis that, if addressed, could possibly prevent disease development.
The many possible sources of gut-barrier disruption
What causes leaky gut, and when should physicians and patients be suspicious if they have it?
There are many risk factors that have been associated with leaky gut in both human studies and animal studies, including acrolein (food toxin), aging, alcohol, antacid drugs, antibiotics, burn injury, chemotherapy, circadian rhythm disruption, corticosteroids, emulsifiers (food additives), strenuous exercise (≥ 2 hours) at 60% VO2 max, starvation, fructose, fructans, gliadin (wheat protein), high-fat diet, high-salt diet, high-sugar diet, hyperglycemia, low-fiber diet, nonsteroidal anti-inflammatory drugs, pesticide, proinflammatory cytokines, psychological stress, radiation, sleep deprivation, smoking, and sweeteners.
Patients may be completely asymptomatic with leaky gut. Physicians should be suspicious if there is a genetic predisposition to chronic disease or if any risk factors are unveiled after assessing diet and lifestyle exposures.
What is the role of the Western diet and processed food consumption in driving disruptions of the gut barrier?
The Western diet reduces gut-barrier mucus thickness, leading to increased gut permeability. People who consume a Western diet typically eat less than 15 grams of fiber per day, which is significantly less than many other cultures, including the hunter-gatherers of Tanzania (Hadza), who get 100 or more grams of fiber a day in their food.
With a fiber-depleted diet, gut microbiota that normally feed on fiber gradually disappear and other commensals shift their metabolism to degrade the gut-barrier mucus layer.
A low-fiber diet also decreases short-chain fatty acid production, which reduces production of mucus and affects tight junction regulation.
Emerging evidence on causality
New evidence is demonstrating that previous functional conditions of the gastrointestinal tract, like functional dyspepsia, are associated with abnormalities to the intestinal barrier. What is the association between conditions like functional dyspepsia and irritable bowel syndrome (IBS) with gut-barrier disruption?
Conditions such as functional dyspepsia and IBS are similar in that their pathophysiology is incompletely understood and likely attributable to contributions from many different underlying mechanisms. This makes it difficult for clinicians to explain the condition to patients and often to treat without specific therapeutic targets.
Emerging evidence with new diagnostic tools, such as confocal laser endomicroscopy, has demonstrated altered mucosal barrier function in both conditions.
In patients with IBS who have a suspected food intolerance, studies looking at exposure to the food antigens found that the food caused immediate breaks, increased intervillous spaces, and increased inflammatory cells in the gut mucosa. These changes were associated with patient responses to exclusion diets.
In functional dyspepsia, another study, using confocal laser endomicroscopy, has shown that affected patients have significantly greater epithelial gap density in the duodenum, compared with healthy controls. There was also impaired duodenal-epithelial barrier integrity and evidence of increased cellular pyroptosis in the duodenal mucosa.
These findings suggest that while IBS and functional dyspepsia are still likely multifactorial, there may be a common preclinical state that can be further investigated as far as preventing its development and using it as a therapeutic target.
What diagnostic testing are you using to determine whether patients have disruptions to the gut barrier? Are they validated or more experimental?
There are various testing strategies that have been used in research to diagnose intestinal hyperpermeability. In a 2021 analysis, Dr. Michael Camilleri found that the optimal probes for measuring small intestinal and colonic permeability are the mass excreted of 13C-mannitol at 0-2 hours and lactulose during 2-8 hours or sucralose during 8-24 hours. Studies looking at postinfectious IBS have incorporated elevated urinary lactulose/mannitol ratios. Dr. Alessio Fasano and others have looked at using zonulin as a biomarker of impaired gut-barrier function. These tests are still considered experimental.
Is there an association between alterations in the gut microbiome and gut-barrier disruption?
There is an integral relationship between the gut microbiome and gut-barrier function, and dysbiosis can disrupt gut-barrier functionality.
The microbiota produce a variety of metabolites in close proximity to the gut epithelium, impacting gut-barrier function and immune response. For example, short-chain fatty acids produced by Bifidobacterium, Bacteroides, Enterobacter, Faecalibacterium, and Roseburia species impact host immune cell differentiation and metabolism as well as influence susceptibility to pathogens.
Studies have shown that sodium butyrate significantly improves epithelial-barrier function. Other experiments have used transplantation of the intestinal microbiota to show that introduction of certain microbial phenotypes can significantly increase gut permeability.
Practical advice for clinicians and patients
How do you advise patients to avoid gut-barrier disruption?
It is important to educate and counsel patients about the long list of risk factors, many of which are closely related to a Western diet and lifestyle, which can increase their risk for leaky gut.
Once one has it, can it be repaired? Can you share a bit about your protocols in general terms?
Many interventions have been shown to improve intestinal permeability. They include berberine, butyrate, caloric restriction and fasting, curcumin, dietary fiber (prebiotics), moderate exercise, fermented food, fish oil, glutamine, quercetin, probiotics, vagus nerve stimulation, vitamin D, and zinc.
Protocols have to be tailored to patients and their risk factors, diet, and lifestyle.
What are some tips from a nutrition and lifestyle standpoint that patients can follow to ensure a robust gut barrier?
It is important to emphasize a high-fiber diet with naturally fermented food, incorporating time-restricted eating, such as eating an early dinner and nothing else before bedtime, a moderate exercise routine, and gut-brain modulation with techniques such as acupuncture that can incorporate vagus nerve stimulation. Limited safe precision supplementation can be discussed on an individual basis based on the patient’s interest, additional testing, and other existing health conditions.
Dr. Akash Goel is a clinical assistant professor of medicine at Weill Cornell in gastroenterology and hepatology. He has disclosed no relevant financial relationships. His work has appeared on networks and publications such as CNN, The New York Times, Time Magazine, and Financial Times. He has a deep interest in nutrition, food as medicine, and the intersection between the gut microbiome and human health.
A version of this article appeared on Medscape.com.
Depression: Differential Diagnosis
Risk calculator for early-stage CKD may soon enter U.S. market
PHILADELPHIA – The analyses offer the possibility of focusing intensified medical management of early-stage CKD on those patients who could potentially receive the most benefit.
The Klinrisk model predicts the risk of an adult with early-stage CKD developing either a 40% or greater drop in estimated glomerular filtration rate or kidney failure. It calculates risk based on 20 lab-measured variables that include serum creatinine, urine albumin-to-creatinine ratio, and other values taken from routinely ordered tests such as complete blood cell counts, chemistry panels, comprehensive metabolic panels, and urinalysis.
In the most recent and largest external validation study using data from 4.6 million American adults enrolled in commercial and Medicare insurance plans, the results showed Klinrisk correctly predicted CKD progression in 80%-83% of individuals over 2 years and in 78%-83% of individuals over 5 years, depending on the insurance provider, Navdeep Tangri, MD, PhD, reported at the annual meeting of the American Society of Nephrology. When urinalysis data were available, the model correctly predicted CKD progression in 81%-87% of individuals over 2 years and in 80%-87% of individuals over 5 years. These results follow prior reports of several other successful validations of Klinrisk.
‘Ready to implement’
“The Klinrisk model is ready to implement by any payer, health system, or clinic where the needed lab data are available,” said Dr. Tangri, a nephrologist and professor at the University of Manitoba, Winnipeg, and founder of Klinrisk Inc., the company developing and commercializing the Klinrisk assessment tool.
For the time being, Dr. Tangri sees Klinrisk as a population health device that can allow insurers and health systems to track management quality and quality improvement and to target patients who stand to benefit most from relatively expensive resources. This includes prescriptions for finerenone (Kerendia, Bayer) for people who also have type 2 diabetes, and agents from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors such as dapagliflozin (Farxiga, AstraZeneca) and empagliflozin (Jardiance, Boehringer Ingelheim and Lilly).
He has also begun discussions with the Food and Drug Administration about the data the agency will need to consider Klinrisk for potential approval as a new medical device, perhaps in 2025. That’s how he envisions getting a Klinrisk assessment into the hands of caregivers that they could use with individual patients to create an appropriate treatment plan.
Results from his new analysis showed that “all the kidney disease action is in the 10%-20% of people with the highest risk on Klinrisk, while not much happens in those in the bottom half,” Dr. Tangri said during his presentation.
“We’re trying to find the patients who get the largest [absolute] benefit from intensified treatment,” he added in an interview. “Klinrisk finds people with high-risk kidney disease early on, when kidney function is still normal or near normal. High-risk patients are often completely unrecognized. Risk-based management” that identifies the early-stage CKD patients who would benefit most from treatment with an SGLT2 inhibitor, finerenone, and other foundational treatments to slow CKD progression “is better than the free-for-all that occurs today.”
Simplified data collection
“Klinrisk is very effective,” but requires follow-up by clinicians and health systems to implement its findings, commented Josef Coresh, MD, a professor of clinical epidemiology at Johns Hopkins Bloomberg, Baltimore. Dr. Coresh compared it with a free equation that estimates a person’s risk for a 40% drop in kidney function over the next 3 years developed by Dr. Tangri, Dr. Coresh, and many collaborators led by Morgan C. Grams, MD, PhD, of New York University that they published in 2022, and posted on a website of the CKD Prognosis Consortium.
The CKD Prognosis Consortium formula “takes a different approach” from Klinrisk. The commercial formula “is simpler, only using lab measures, and avoids inputs taken from physical examination such as systolic blood pressure and body mass index and health history data such as smoking, noted Dr. Coresh. He also speculated that “a commercial formula that must be paid for may counterintuitively result in better follow-up for making management changes if it uses some of the resources for education and system changes.”
Using data from multiple sources, like the CKD Prognosis Consortium equation, can create implementation challenges, said Dr. Tangri. “Lab results don’t vary much,” which makes Klinrisk “quite an improvement for implementation. It’s easier to implement.”
Other findings from the newest validation study that Dr. Tangri presented were that the people studied with Klinrisk scores in the top 10% had, over the next 2 years of follow-up and compared with people in the bottom half for Klinrisk staging, a 3- to 5-fold higher rate of all-cause medical costs, a 13-30-fold increase in CKD-related costs, and a 5- to 10-fold increase in hospitalizations and ED visits.
Early identification of CKD and early initiation of intensified treatment for high-risk patients can reduce the rate of progression to dialysis, reduce hospitalizations for heart failure, and lower the cost of care, Dr. Tangri said.
The validation study in 4.6 million Americans was sponsored by Boehringer Ingelheim. Dr. Tangri founded and has an ownership interest in Klinrisk. He has also received honoraria from, has ownership interests in, and has been a consultant to multiple pharmaceutical companies. Dr. Coresh had no disclosures.
PHILADELPHIA – The analyses offer the possibility of focusing intensified medical management of early-stage CKD on those patients who could potentially receive the most benefit.
The Klinrisk model predicts the risk of an adult with early-stage CKD developing either a 40% or greater drop in estimated glomerular filtration rate or kidney failure. It calculates risk based on 20 lab-measured variables that include serum creatinine, urine albumin-to-creatinine ratio, and other values taken from routinely ordered tests such as complete blood cell counts, chemistry panels, comprehensive metabolic panels, and urinalysis.
In the most recent and largest external validation study using data from 4.6 million American adults enrolled in commercial and Medicare insurance plans, the results showed Klinrisk correctly predicted CKD progression in 80%-83% of individuals over 2 years and in 78%-83% of individuals over 5 years, depending on the insurance provider, Navdeep Tangri, MD, PhD, reported at the annual meeting of the American Society of Nephrology. When urinalysis data were available, the model correctly predicted CKD progression in 81%-87% of individuals over 2 years and in 80%-87% of individuals over 5 years. These results follow prior reports of several other successful validations of Klinrisk.
‘Ready to implement’
“The Klinrisk model is ready to implement by any payer, health system, or clinic where the needed lab data are available,” said Dr. Tangri, a nephrologist and professor at the University of Manitoba, Winnipeg, and founder of Klinrisk Inc., the company developing and commercializing the Klinrisk assessment tool.
For the time being, Dr. Tangri sees Klinrisk as a population health device that can allow insurers and health systems to track management quality and quality improvement and to target patients who stand to benefit most from relatively expensive resources. This includes prescriptions for finerenone (Kerendia, Bayer) for people who also have type 2 diabetes, and agents from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors such as dapagliflozin (Farxiga, AstraZeneca) and empagliflozin (Jardiance, Boehringer Ingelheim and Lilly).
He has also begun discussions with the Food and Drug Administration about the data the agency will need to consider Klinrisk for potential approval as a new medical device, perhaps in 2025. That’s how he envisions getting a Klinrisk assessment into the hands of caregivers that they could use with individual patients to create an appropriate treatment plan.
Results from his new analysis showed that “all the kidney disease action is in the 10%-20% of people with the highest risk on Klinrisk, while not much happens in those in the bottom half,” Dr. Tangri said during his presentation.
“We’re trying to find the patients who get the largest [absolute] benefit from intensified treatment,” he added in an interview. “Klinrisk finds people with high-risk kidney disease early on, when kidney function is still normal or near normal. High-risk patients are often completely unrecognized. Risk-based management” that identifies the early-stage CKD patients who would benefit most from treatment with an SGLT2 inhibitor, finerenone, and other foundational treatments to slow CKD progression “is better than the free-for-all that occurs today.”
Simplified data collection
“Klinrisk is very effective,” but requires follow-up by clinicians and health systems to implement its findings, commented Josef Coresh, MD, a professor of clinical epidemiology at Johns Hopkins Bloomberg, Baltimore. Dr. Coresh compared it with a free equation that estimates a person’s risk for a 40% drop in kidney function over the next 3 years developed by Dr. Tangri, Dr. Coresh, and many collaborators led by Morgan C. Grams, MD, PhD, of New York University that they published in 2022, and posted on a website of the CKD Prognosis Consortium.
The CKD Prognosis Consortium formula “takes a different approach” from Klinrisk. The commercial formula “is simpler, only using lab measures, and avoids inputs taken from physical examination such as systolic blood pressure and body mass index and health history data such as smoking, noted Dr. Coresh. He also speculated that “a commercial formula that must be paid for may counterintuitively result in better follow-up for making management changes if it uses some of the resources for education and system changes.”
Using data from multiple sources, like the CKD Prognosis Consortium equation, can create implementation challenges, said Dr. Tangri. “Lab results don’t vary much,” which makes Klinrisk “quite an improvement for implementation. It’s easier to implement.”
Other findings from the newest validation study that Dr. Tangri presented were that the people studied with Klinrisk scores in the top 10% had, over the next 2 years of follow-up and compared with people in the bottom half for Klinrisk staging, a 3- to 5-fold higher rate of all-cause medical costs, a 13-30-fold increase in CKD-related costs, and a 5- to 10-fold increase in hospitalizations and ED visits.
Early identification of CKD and early initiation of intensified treatment for high-risk patients can reduce the rate of progression to dialysis, reduce hospitalizations for heart failure, and lower the cost of care, Dr. Tangri said.
The validation study in 4.6 million Americans was sponsored by Boehringer Ingelheim. Dr. Tangri founded and has an ownership interest in Klinrisk. He has also received honoraria from, has ownership interests in, and has been a consultant to multiple pharmaceutical companies. Dr. Coresh had no disclosures.
PHILADELPHIA – The analyses offer the possibility of focusing intensified medical management of early-stage CKD on those patients who could potentially receive the most benefit.
The Klinrisk model predicts the risk of an adult with early-stage CKD developing either a 40% or greater drop in estimated glomerular filtration rate or kidney failure. It calculates risk based on 20 lab-measured variables that include serum creatinine, urine albumin-to-creatinine ratio, and other values taken from routinely ordered tests such as complete blood cell counts, chemistry panels, comprehensive metabolic panels, and urinalysis.
In the most recent and largest external validation study using data from 4.6 million American adults enrolled in commercial and Medicare insurance plans, the results showed Klinrisk correctly predicted CKD progression in 80%-83% of individuals over 2 years and in 78%-83% of individuals over 5 years, depending on the insurance provider, Navdeep Tangri, MD, PhD, reported at the annual meeting of the American Society of Nephrology. When urinalysis data were available, the model correctly predicted CKD progression in 81%-87% of individuals over 2 years and in 80%-87% of individuals over 5 years. These results follow prior reports of several other successful validations of Klinrisk.
‘Ready to implement’
“The Klinrisk model is ready to implement by any payer, health system, or clinic where the needed lab data are available,” said Dr. Tangri, a nephrologist and professor at the University of Manitoba, Winnipeg, and founder of Klinrisk Inc., the company developing and commercializing the Klinrisk assessment tool.
For the time being, Dr. Tangri sees Klinrisk as a population health device that can allow insurers and health systems to track management quality and quality improvement and to target patients who stand to benefit most from relatively expensive resources. This includes prescriptions for finerenone (Kerendia, Bayer) for people who also have type 2 diabetes, and agents from the class of sodium-glucose cotransporter 2 (SGLT2) inhibitors such as dapagliflozin (Farxiga, AstraZeneca) and empagliflozin (Jardiance, Boehringer Ingelheim and Lilly).
He has also begun discussions with the Food and Drug Administration about the data the agency will need to consider Klinrisk for potential approval as a new medical device, perhaps in 2025. That’s how he envisions getting a Klinrisk assessment into the hands of caregivers that they could use with individual patients to create an appropriate treatment plan.
Results from his new analysis showed that “all the kidney disease action is in the 10%-20% of people with the highest risk on Klinrisk, while not much happens in those in the bottom half,” Dr. Tangri said during his presentation.
“We’re trying to find the patients who get the largest [absolute] benefit from intensified treatment,” he added in an interview. “Klinrisk finds people with high-risk kidney disease early on, when kidney function is still normal or near normal. High-risk patients are often completely unrecognized. Risk-based management” that identifies the early-stage CKD patients who would benefit most from treatment with an SGLT2 inhibitor, finerenone, and other foundational treatments to slow CKD progression “is better than the free-for-all that occurs today.”
Simplified data collection
“Klinrisk is very effective,” but requires follow-up by clinicians and health systems to implement its findings, commented Josef Coresh, MD, a professor of clinical epidemiology at Johns Hopkins Bloomberg, Baltimore. Dr. Coresh compared it with a free equation that estimates a person’s risk for a 40% drop in kidney function over the next 3 years developed by Dr. Tangri, Dr. Coresh, and many collaborators led by Morgan C. Grams, MD, PhD, of New York University that they published in 2022, and posted on a website of the CKD Prognosis Consortium.
The CKD Prognosis Consortium formula “takes a different approach” from Klinrisk. The commercial formula “is simpler, only using lab measures, and avoids inputs taken from physical examination such as systolic blood pressure and body mass index and health history data such as smoking, noted Dr. Coresh. He also speculated that “a commercial formula that must be paid for may counterintuitively result in better follow-up for making management changes if it uses some of the resources for education and system changes.”
Using data from multiple sources, like the CKD Prognosis Consortium equation, can create implementation challenges, said Dr. Tangri. “Lab results don’t vary much,” which makes Klinrisk “quite an improvement for implementation. It’s easier to implement.”
Other findings from the newest validation study that Dr. Tangri presented were that the people studied with Klinrisk scores in the top 10% had, over the next 2 years of follow-up and compared with people in the bottom half for Klinrisk staging, a 3- to 5-fold higher rate of all-cause medical costs, a 13-30-fold increase in CKD-related costs, and a 5- to 10-fold increase in hospitalizations and ED visits.
Early identification of CKD and early initiation of intensified treatment for high-risk patients can reduce the rate of progression to dialysis, reduce hospitalizations for heart failure, and lower the cost of care, Dr. Tangri said.
The validation study in 4.6 million Americans was sponsored by Boehringer Ingelheim. Dr. Tangri founded and has an ownership interest in Klinrisk. He has also received honoraria from, has ownership interests in, and has been a consultant to multiple pharmaceutical companies. Dr. Coresh had no disclosures.
AT KIDNEY WEEK 2023
MASLD, MASH projected to grow by 23% in the U.S. through 2050
BOSTON – The nomenclature may have changed, but the steady rise in the most common form of liver disease – metabolic dysfunction–associated steatotic liver disease (MASLD, formerly known as NAFLD) – is predicted to continue into the middle of this century.
That’s according to Phuc Le, PhD, MPH, and colleagues at the Cleveland Clinic. They created a mathematical model incorporating data on the growth of the U.S. population and the natural history of MASLD/NAFLD. The model projected a relative 23% increase in MASLD among U.S. adults from 2020 to 2050.
“Our model forecasts a substantial clinical burden of NAFLD over the next 3 decades. In the absence of effective treatments, health systems should plan for large increases in the number of liver cancer cases and the need for liver transplant,” Dr. Le said in a media briefing held on Nov. 7 prior to her presentation of the data at the annual meeting of the American Association for the Study of Liver Diseases.
The estimated worldwide prevalence of MASLD is 38%. In the United States, an estimated 27.8% of adults had MASLD as of 2020.
Dr. Le and colleagues wanted to get a clearer picture of the expected increase in the clinical burden of MASLD in the coming decades. The researchers used data from the medical literature to create an individual-level state transition model. They took into account projections of the growth of the U.S. population and the progression of MASLD and metabolic dysfunction–associated steatohepatitis (MASH, formerly NASH) through stages of fibrosis to decompensation, hepatocellular carcinoma (HCC), transplant, and liver-related death as a proportion of all-cause mortality.
Validated model
They validated the model by testing it against liver outcomes from 2000 through 2018 and published data on the U.S. population. The model closely matched trends in MASLD prevalence, MASH proportion, HCC and liver transplant incidences, and overall survival rates for patients with MASLD.
As noted, the model predicted a steady increase in MASLD prevalence, from 27.8% in 2020 to 34.3% by 2050, a relative increase of about 23%. The model also predicted a slight uptick in the proportion of MASH among patients with MASLD, from 20% to 21.8%.
The investigators said that the prevalence of MASLD/MASH would likely remain relatively stable among people aged 18-29 years but would increase significantly for all other age groups.
In addition, the model predicted an increase in the proportion of cirrhosis in patients with MASLD from 1.9% to 3.1%, as well as a rise in liver-related deaths from 0.4% of all deaths in 2020 to 1% by 2050.
The investigators also foresaw a rise in HCC cases, from 10,400 annually to 19,300 by 2050 and a more than twofold increase in liver transplants, from 1,700 in 2020 to 4,200 in 2050.
A “tsunami” of liver disease
In the question-and-answer portion of the briefing, Norah Terrault, MD, AASLD president and chief of gastroenterology and hepatology at the University of Southern California, Los Angeles, commented on the study findings and “the frightening trajectory in terms of disease burden.
“I’m thinking to myself there’s no way we’re going to be able to transplant our way out of this tsunami of disease that’s coming our way,” she said, and asked Dr. Le what policy or societal approaches might be implemented to help stem the tide.
“This is a really huge question,” Dr. Le acknowledged. The study only provides estimates of what the future burden of disease might be if there are no changes in clinical care for patients with MASLD or if the trajectory of contributing factors, such as obesity, diabetes, and other metabolic diseases, continued to increase, she cautioned.
Raising awareness of MASLD/MASH and working to improve collaboration among liver specialists and general practitioners could help to flatten the curve, she suggested.
The study was supported by a grant from the Agency for Healthcare Research and Quality. Dr. Le and Dr. Terrault have disclosed no relevant financial relations.
A version of this article first appeared on Medscape.com.
BOSTON – The nomenclature may have changed, but the steady rise in the most common form of liver disease – metabolic dysfunction–associated steatotic liver disease (MASLD, formerly known as NAFLD) – is predicted to continue into the middle of this century.
That’s according to Phuc Le, PhD, MPH, and colleagues at the Cleveland Clinic. They created a mathematical model incorporating data on the growth of the U.S. population and the natural history of MASLD/NAFLD. The model projected a relative 23% increase in MASLD among U.S. adults from 2020 to 2050.
“Our model forecasts a substantial clinical burden of NAFLD over the next 3 decades. In the absence of effective treatments, health systems should plan for large increases in the number of liver cancer cases and the need for liver transplant,” Dr. Le said in a media briefing held on Nov. 7 prior to her presentation of the data at the annual meeting of the American Association for the Study of Liver Diseases.
The estimated worldwide prevalence of MASLD is 38%. In the United States, an estimated 27.8% of adults had MASLD as of 2020.
Dr. Le and colleagues wanted to get a clearer picture of the expected increase in the clinical burden of MASLD in the coming decades. The researchers used data from the medical literature to create an individual-level state transition model. They took into account projections of the growth of the U.S. population and the progression of MASLD and metabolic dysfunction–associated steatohepatitis (MASH, formerly NASH) through stages of fibrosis to decompensation, hepatocellular carcinoma (HCC), transplant, and liver-related death as a proportion of all-cause mortality.
Validated model
They validated the model by testing it against liver outcomes from 2000 through 2018 and published data on the U.S. population. The model closely matched trends in MASLD prevalence, MASH proportion, HCC and liver transplant incidences, and overall survival rates for patients with MASLD.
As noted, the model predicted a steady increase in MASLD prevalence, from 27.8% in 2020 to 34.3% by 2050, a relative increase of about 23%. The model also predicted a slight uptick in the proportion of MASH among patients with MASLD, from 20% to 21.8%.
The investigators said that the prevalence of MASLD/MASH would likely remain relatively stable among people aged 18-29 years but would increase significantly for all other age groups.
In addition, the model predicted an increase in the proportion of cirrhosis in patients with MASLD from 1.9% to 3.1%, as well as a rise in liver-related deaths from 0.4% of all deaths in 2020 to 1% by 2050.
The investigators also foresaw a rise in HCC cases, from 10,400 annually to 19,300 by 2050 and a more than twofold increase in liver transplants, from 1,700 in 2020 to 4,200 in 2050.
A “tsunami” of liver disease
In the question-and-answer portion of the briefing, Norah Terrault, MD, AASLD president and chief of gastroenterology and hepatology at the University of Southern California, Los Angeles, commented on the study findings and “the frightening trajectory in terms of disease burden.
“I’m thinking to myself there’s no way we’re going to be able to transplant our way out of this tsunami of disease that’s coming our way,” she said, and asked Dr. Le what policy or societal approaches might be implemented to help stem the tide.
“This is a really huge question,” Dr. Le acknowledged. The study only provides estimates of what the future burden of disease might be if there are no changes in clinical care for patients with MASLD or if the trajectory of contributing factors, such as obesity, diabetes, and other metabolic diseases, continued to increase, she cautioned.
Raising awareness of MASLD/MASH and working to improve collaboration among liver specialists and general practitioners could help to flatten the curve, she suggested.
The study was supported by a grant from the Agency for Healthcare Research and Quality. Dr. Le and Dr. Terrault have disclosed no relevant financial relations.
A version of this article first appeared on Medscape.com.
BOSTON – The nomenclature may have changed, but the steady rise in the most common form of liver disease – metabolic dysfunction–associated steatotic liver disease (MASLD, formerly known as NAFLD) – is predicted to continue into the middle of this century.
That’s according to Phuc Le, PhD, MPH, and colleagues at the Cleveland Clinic. They created a mathematical model incorporating data on the growth of the U.S. population and the natural history of MASLD/NAFLD. The model projected a relative 23% increase in MASLD among U.S. adults from 2020 to 2050.
“Our model forecasts a substantial clinical burden of NAFLD over the next 3 decades. In the absence of effective treatments, health systems should plan for large increases in the number of liver cancer cases and the need for liver transplant,” Dr. Le said in a media briefing held on Nov. 7 prior to her presentation of the data at the annual meeting of the American Association for the Study of Liver Diseases.
The estimated worldwide prevalence of MASLD is 38%. In the United States, an estimated 27.8% of adults had MASLD as of 2020.
Dr. Le and colleagues wanted to get a clearer picture of the expected increase in the clinical burden of MASLD in the coming decades. The researchers used data from the medical literature to create an individual-level state transition model. They took into account projections of the growth of the U.S. population and the progression of MASLD and metabolic dysfunction–associated steatohepatitis (MASH, formerly NASH) through stages of fibrosis to decompensation, hepatocellular carcinoma (HCC), transplant, and liver-related death as a proportion of all-cause mortality.
Validated model
They validated the model by testing it against liver outcomes from 2000 through 2018 and published data on the U.S. population. The model closely matched trends in MASLD prevalence, MASH proportion, HCC and liver transplant incidences, and overall survival rates for patients with MASLD.
As noted, the model predicted a steady increase in MASLD prevalence, from 27.8% in 2020 to 34.3% by 2050, a relative increase of about 23%. The model also predicted a slight uptick in the proportion of MASH among patients with MASLD, from 20% to 21.8%.
The investigators said that the prevalence of MASLD/MASH would likely remain relatively stable among people aged 18-29 years but would increase significantly for all other age groups.
In addition, the model predicted an increase in the proportion of cirrhosis in patients with MASLD from 1.9% to 3.1%, as well as a rise in liver-related deaths from 0.4% of all deaths in 2020 to 1% by 2050.
The investigators also foresaw a rise in HCC cases, from 10,400 annually to 19,300 by 2050 and a more than twofold increase in liver transplants, from 1,700 in 2020 to 4,200 in 2050.
A “tsunami” of liver disease
In the question-and-answer portion of the briefing, Norah Terrault, MD, AASLD president and chief of gastroenterology and hepatology at the University of Southern California, Los Angeles, commented on the study findings and “the frightening trajectory in terms of disease burden.
“I’m thinking to myself there’s no way we’re going to be able to transplant our way out of this tsunami of disease that’s coming our way,” she said, and asked Dr. Le what policy or societal approaches might be implemented to help stem the tide.
“This is a really huge question,” Dr. Le acknowledged. The study only provides estimates of what the future burden of disease might be if there are no changes in clinical care for patients with MASLD or if the trajectory of contributing factors, such as obesity, diabetes, and other metabolic diseases, continued to increase, she cautioned.
Raising awareness of MASLD/MASH and working to improve collaboration among liver specialists and general practitioners could help to flatten the curve, she suggested.
The study was supported by a grant from the Agency for Healthcare Research and Quality. Dr. Le and Dr. Terrault have disclosed no relevant financial relations.
A version of this article first appeared on Medscape.com.
AT THE LIVER MEETING
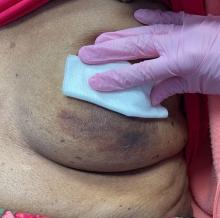
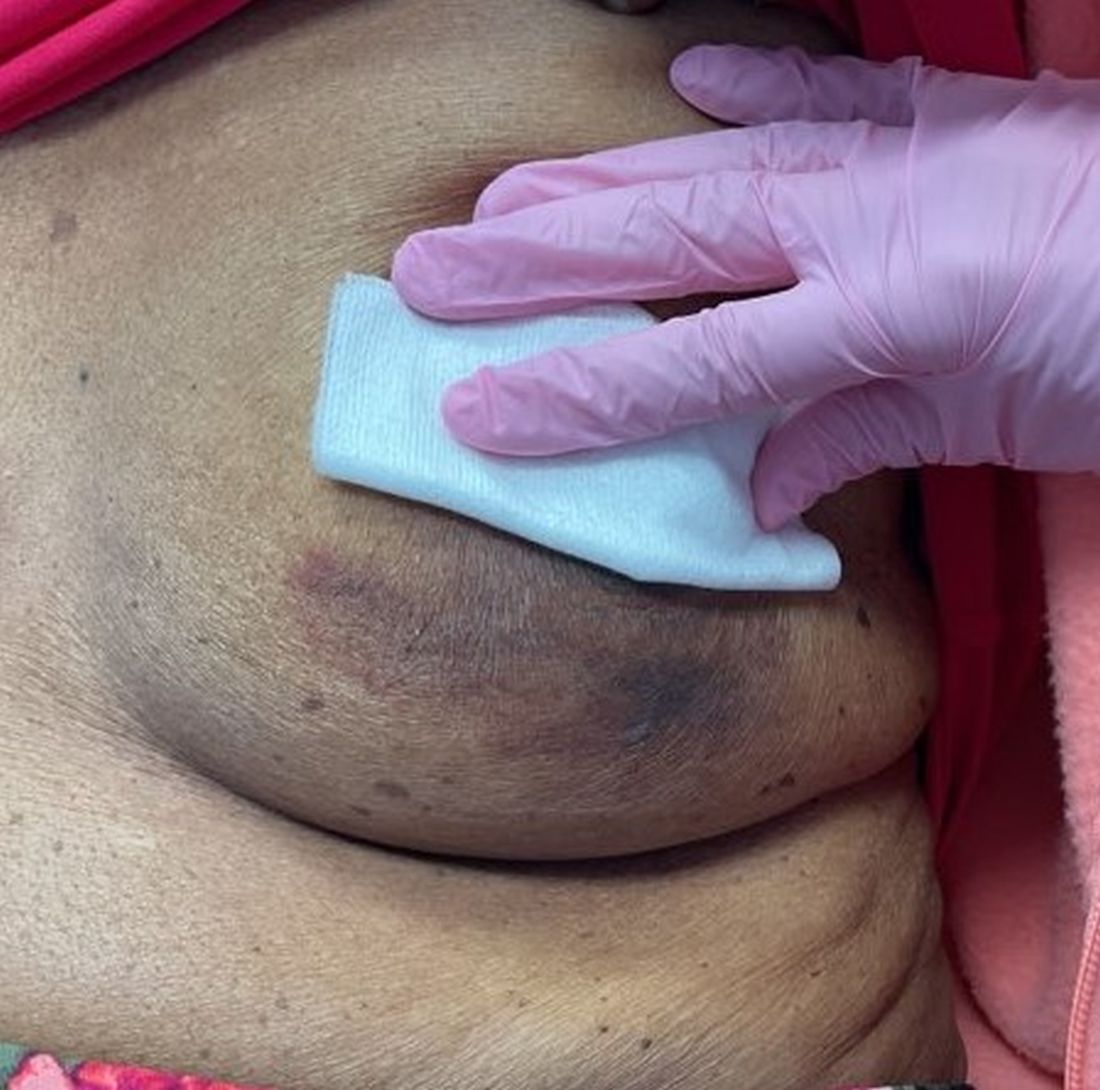
An 88-year-old Black woman presented with 3 months duration of asymptomatic, violaceous patches on the left breast
Angiosarcomas are uncommon, high-grade malignant tumors of endothelial cell origin that can arise via the lymphatics or vasculature. They typically occur spontaneously; however, there have been cases reported of benign vascular transformation. These tumors are more commonly found in elderly men on the head and neck in sun-damaged skin. . This is a late complication, typically occurring about 5-10 years after radiation. Stewart-Treves syndrome, chronic lymphedema occurring after breast cancer treatment with axillary node dissection, increases the risk of angiosarcoma. As a vascular tumor, angiosarcoma spreads hematogenously and carries a poor prognosis if not caught early. Differential diagnoses include other vascular tumors such as retiform hemangioendothelioma. In this specific patient, the differential diagnosis includes Paget’s disease, chronic radiation skin changes, and eczema.
Histopathologically, angiosarcomas exhibit abnormal, pleomorphic, malignant endothelial cells. As the tumor progresses, the cell architecture becomes more distorted and cells form layers with papillary projections into the vascular lumen. Malignant cells may stain positive for CD31, CD34, the oncogene ERG and the proto-oncogene FLI-1. Histology in this patient revealed radiation changes in the dermis, as well as few vascular channels lined by large endothelial cells with marked nuclear atypia, in the form of large nucleoli and variably coarse chromatin. The cells were positive for MYC.
Treatment of angiosarcoma involves a multidisciplinary approach. Resection with wide margins is generally the treatment of choice. However, recurrence is relatively common, which may be a result of microsatellite deposits of the tumor. Perioperative radiation is recommended, and adjuvant chemotherapy often is recommended for metastatic disease. Specifically, paclitaxel has been found to promote survival in some cases of cutaneous angiosarcoma. Metastatic disease may be treated with cytotoxic drugs such as anthracyclines and taxanes. Additionally, targeted therapy including anti-VEGF drugs and tyrosine kinase inhibitors have been tested.
The case and photo were submitted by Mr. Shapiro of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Dr. Bilu Martin. The column was edited by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cohen-Hallaleh RB et al. Clin Sarcoma Res. 2017 Aug 7:7:15.
Cozzi S et al. Rep Pract Oncol Radiother. 2021 Sep 30;26(5):827-32.
Spiker AM, Mangla A, Ramsey ML. Angiosarcoma. [Updated 2023 Jul 17]. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing; 2023 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK441983/
Angiosarcomas are uncommon, high-grade malignant tumors of endothelial cell origin that can arise via the lymphatics or vasculature. They typically occur spontaneously; however, there have been cases reported of benign vascular transformation. These tumors are more commonly found in elderly men on the head and neck in sun-damaged skin. . This is a late complication, typically occurring about 5-10 years after radiation. Stewart-Treves syndrome, chronic lymphedema occurring after breast cancer treatment with axillary node dissection, increases the risk of angiosarcoma. As a vascular tumor, angiosarcoma spreads hematogenously and carries a poor prognosis if not caught early. Differential diagnoses include other vascular tumors such as retiform hemangioendothelioma. In this specific patient, the differential diagnosis includes Paget’s disease, chronic radiation skin changes, and eczema.
Histopathologically, angiosarcomas exhibit abnormal, pleomorphic, malignant endothelial cells. As the tumor progresses, the cell architecture becomes more distorted and cells form layers with papillary projections into the vascular lumen. Malignant cells may stain positive for CD31, CD34, the oncogene ERG and the proto-oncogene FLI-1. Histology in this patient revealed radiation changes in the dermis, as well as few vascular channels lined by large endothelial cells with marked nuclear atypia, in the form of large nucleoli and variably coarse chromatin. The cells were positive for MYC.
Treatment of angiosarcoma involves a multidisciplinary approach. Resection with wide margins is generally the treatment of choice. However, recurrence is relatively common, which may be a result of microsatellite deposits of the tumor. Perioperative radiation is recommended, and adjuvant chemotherapy often is recommended for metastatic disease. Specifically, paclitaxel has been found to promote survival in some cases of cutaneous angiosarcoma. Metastatic disease may be treated with cytotoxic drugs such as anthracyclines and taxanes. Additionally, targeted therapy including anti-VEGF drugs and tyrosine kinase inhibitors have been tested.
The case and photo were submitted by Mr. Shapiro of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Dr. Bilu Martin. The column was edited by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cohen-Hallaleh RB et al. Clin Sarcoma Res. 2017 Aug 7:7:15.
Cozzi S et al. Rep Pract Oncol Radiother. 2021 Sep 30;26(5):827-32.
Spiker AM, Mangla A, Ramsey ML. Angiosarcoma. [Updated 2023 Jul 17]. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing; 2023 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK441983/
Angiosarcomas are uncommon, high-grade malignant tumors of endothelial cell origin that can arise via the lymphatics or vasculature. They typically occur spontaneously; however, there have been cases reported of benign vascular transformation. These tumors are more commonly found in elderly men on the head and neck in sun-damaged skin. . This is a late complication, typically occurring about 5-10 years after radiation. Stewart-Treves syndrome, chronic lymphedema occurring after breast cancer treatment with axillary node dissection, increases the risk of angiosarcoma. As a vascular tumor, angiosarcoma spreads hematogenously and carries a poor prognosis if not caught early. Differential diagnoses include other vascular tumors such as retiform hemangioendothelioma. In this specific patient, the differential diagnosis includes Paget’s disease, chronic radiation skin changes, and eczema.
Histopathologically, angiosarcomas exhibit abnormal, pleomorphic, malignant endothelial cells. As the tumor progresses, the cell architecture becomes more distorted and cells form layers with papillary projections into the vascular lumen. Malignant cells may stain positive for CD31, CD34, the oncogene ERG and the proto-oncogene FLI-1. Histology in this patient revealed radiation changes in the dermis, as well as few vascular channels lined by large endothelial cells with marked nuclear atypia, in the form of large nucleoli and variably coarse chromatin. The cells were positive for MYC.
Treatment of angiosarcoma involves a multidisciplinary approach. Resection with wide margins is generally the treatment of choice. However, recurrence is relatively common, which may be a result of microsatellite deposits of the tumor. Perioperative radiation is recommended, and adjuvant chemotherapy often is recommended for metastatic disease. Specifically, paclitaxel has been found to promote survival in some cases of cutaneous angiosarcoma. Metastatic disease may be treated with cytotoxic drugs such as anthracyclines and taxanes. Additionally, targeted therapy including anti-VEGF drugs and tyrosine kinase inhibitors have been tested.
The case and photo were submitted by Mr. Shapiro of Nova Southeastern University College of Osteopathic Medicine, Fort Lauderdale, Fla., and Dr. Bilu Martin. The column was edited by Dr. Bilu Martin.
Dr. Bilu Martin is a board-certified dermatologist in private practice at Premier Dermatology, MD, in Aventura, Fla. More diagnostic cases are available at mdedge.com/dermatology. To submit a case for possible publication, send an email to [email protected].
References
Cohen-Hallaleh RB et al. Clin Sarcoma Res. 2017 Aug 7:7:15.
Cozzi S et al. Rep Pract Oncol Radiother. 2021 Sep 30;26(5):827-32.
Spiker AM, Mangla A, Ramsey ML. Angiosarcoma. [Updated 2023 Jul 17]. In: StatPearls [Internet]. Treasure Island, Fla.: StatPearls Publishing; 2023 Jan-. Available from: www.ncbi.nlm.nih.gov/books/NBK441983/
FDA OKs Paradise Renal Denervation system for hypertension
Recor Medical and parent company Otsuka Medical Devices have announced.
Approval follows a positive review by the FDA’s Circulatory Systems Device panel in August that deemed the system both safe and effective in lowering blood pressure for adults with uncontrolled hypertension who may be inadequately responsive to, or who are intolerant of, antihypertensive medications.
Data supporting approval were provided by the RADIANCE program, the pivotal RADIANCE II trial, as well as RADIANCE-HTN SOLO and RADIANCE-HTN TRIO. RADIANCE II and RADIANCE-HTN SOLO studied patients with mild to moderate hypertension in an “off-meds” setting, and RADIANCE-HTN TRIO enrolled patients with resistant hypertension on standardized triple antihypertensive therapy.
Renal denervation is intended as an adjunctive treatment option when lifestyle changes and medication have not resulted in adequate blood pressure control, the statement notes. It works by denervating the sympathetic nerves surrounding the renal arteries, reducing the overactivity that can lead to hypertension.
The system delivers two to three doses of 360-degree ultrasound energy, lasting 7 seconds each, through each of the main renal arteries to the surrounding nerves. This particular system is water-cooled to protect the renal artery wall, the statement adds.
“Given the significant blood pressure reductions seen in the ultrasound renal denervation trials, the Paradise Ultrasound Renal Denervation system offers a much-needed advancement in our currently available options to control hypertension,” site principal investigator Naomi Fisher, MD, associate professor of medicine, Harvard Medical School, and director of hypertension service and hypertension innovation, division of endocrinology, diabetes, and hypertension at Brigham and Women’s Hospital, Boston, said in the statement.
Ultrasound renal denervation “has proven efficacy in patients with truly resistant hypertension, a population for whom medication therapy often fails. It is also effective in patients with mild to moderate hypertension who cannot tolerate enough medication to control their blood pressure,” Dr. Fisher added.
The Paradise ultrasound renal denervation system previously received CE mark and has been successfully introduced in Europe and is an investigational device in Japan, the companies note.
A second renal denervation system, the Symplicity Spyral Renal Denervation System (Medtronic) underwent FDA panel review the day after the Paradise system review in August, and although the panel voted unanimously that the Symplicity system is safe, they were split on whether or not it was efficacious. A final decision on approval by the FDA of that system is still pending.
A version of this article first appeared in Medscape.com.
Recor Medical and parent company Otsuka Medical Devices have announced.
Approval follows a positive review by the FDA’s Circulatory Systems Device panel in August that deemed the system both safe and effective in lowering blood pressure for adults with uncontrolled hypertension who may be inadequately responsive to, or who are intolerant of, antihypertensive medications.
Data supporting approval were provided by the RADIANCE program, the pivotal RADIANCE II trial, as well as RADIANCE-HTN SOLO and RADIANCE-HTN TRIO. RADIANCE II and RADIANCE-HTN SOLO studied patients with mild to moderate hypertension in an “off-meds” setting, and RADIANCE-HTN TRIO enrolled patients with resistant hypertension on standardized triple antihypertensive therapy.
Renal denervation is intended as an adjunctive treatment option when lifestyle changes and medication have not resulted in adequate blood pressure control, the statement notes. It works by denervating the sympathetic nerves surrounding the renal arteries, reducing the overactivity that can lead to hypertension.
The system delivers two to three doses of 360-degree ultrasound energy, lasting 7 seconds each, through each of the main renal arteries to the surrounding nerves. This particular system is water-cooled to protect the renal artery wall, the statement adds.
“Given the significant blood pressure reductions seen in the ultrasound renal denervation trials, the Paradise Ultrasound Renal Denervation system offers a much-needed advancement in our currently available options to control hypertension,” site principal investigator Naomi Fisher, MD, associate professor of medicine, Harvard Medical School, and director of hypertension service and hypertension innovation, division of endocrinology, diabetes, and hypertension at Brigham and Women’s Hospital, Boston, said in the statement.
Ultrasound renal denervation “has proven efficacy in patients with truly resistant hypertension, a population for whom medication therapy often fails. It is also effective in patients with mild to moderate hypertension who cannot tolerate enough medication to control their blood pressure,” Dr. Fisher added.
The Paradise ultrasound renal denervation system previously received CE mark and has been successfully introduced in Europe and is an investigational device in Japan, the companies note.
A second renal denervation system, the Symplicity Spyral Renal Denervation System (Medtronic) underwent FDA panel review the day after the Paradise system review in August, and although the panel voted unanimously that the Symplicity system is safe, they were split on whether or not it was efficacious. A final decision on approval by the FDA of that system is still pending.
A version of this article first appeared in Medscape.com.
Recor Medical and parent company Otsuka Medical Devices have announced.
Approval follows a positive review by the FDA’s Circulatory Systems Device panel in August that deemed the system both safe and effective in lowering blood pressure for adults with uncontrolled hypertension who may be inadequately responsive to, or who are intolerant of, antihypertensive medications.
Data supporting approval were provided by the RADIANCE program, the pivotal RADIANCE II trial, as well as RADIANCE-HTN SOLO and RADIANCE-HTN TRIO. RADIANCE II and RADIANCE-HTN SOLO studied patients with mild to moderate hypertension in an “off-meds” setting, and RADIANCE-HTN TRIO enrolled patients with resistant hypertension on standardized triple antihypertensive therapy.
Renal denervation is intended as an adjunctive treatment option when lifestyle changes and medication have not resulted in adequate blood pressure control, the statement notes. It works by denervating the sympathetic nerves surrounding the renal arteries, reducing the overactivity that can lead to hypertension.
The system delivers two to three doses of 360-degree ultrasound energy, lasting 7 seconds each, through each of the main renal arteries to the surrounding nerves. This particular system is water-cooled to protect the renal artery wall, the statement adds.
“Given the significant blood pressure reductions seen in the ultrasound renal denervation trials, the Paradise Ultrasound Renal Denervation system offers a much-needed advancement in our currently available options to control hypertension,” site principal investigator Naomi Fisher, MD, associate professor of medicine, Harvard Medical School, and director of hypertension service and hypertension innovation, division of endocrinology, diabetes, and hypertension at Brigham and Women’s Hospital, Boston, said in the statement.
Ultrasound renal denervation “has proven efficacy in patients with truly resistant hypertension, a population for whom medication therapy often fails. It is also effective in patients with mild to moderate hypertension who cannot tolerate enough medication to control their blood pressure,” Dr. Fisher added.
The Paradise ultrasound renal denervation system previously received CE mark and has been successfully introduced in Europe and is an investigational device in Japan, the companies note.
A second renal denervation system, the Symplicity Spyral Renal Denervation System (Medtronic) underwent FDA panel review the day after the Paradise system review in August, and although the panel voted unanimously that the Symplicity system is safe, they were split on whether or not it was efficacious. A final decision on approval by the FDA of that system is still pending.
A version of this article first appeared in Medscape.com.
Even a short course of opioids could jeopardize IBD patient health
These findings amplify the safety signal from previous inpatient studies by showing that even a short course of opioids in an outpatient setting may increase risks of corticosteroid use and emergency department utilization, prompting caution among prescribers, reported Laura Telfer, MS, of Penn State College of Medicine, Hershey, Pa., and colleagues.
“Opioids are frequently prescribed to treat pain associated with IBD,” the investigators wrote in Gastro Hep Advances. “Unfortunately, they are associated with many problems in IBD, including increased risk of emergency room visits, hospitalization, surgery, and mortality. Chronic opioid use may also exacerbate symptoms and induce IBD flares, prompting discontinuation, thus increasing the risk of opioid withdrawal syndrome. Ironically, there is no published evidence that opioids even help to improve abdominal pain in IBD, particularly in the long term. Notably, most studies investigating opioid use in IBD have been limited to hospitalized patients, and few have directly evaluated the impact of opioid prescription length.”
To address this knowledge gap, Ms. Telfer and colleagues conducted a retrospective, population-based cohort study involving patients with IBD who were classified as either long-term opioid users, short-term opioid users, or nonusers. Drawing data from more than 80,000 patients in the TriNetX Diamond Network, the investigators evaluated relative, intergroup risks for corticosteroid use, emergency department utilization, mortality, and IBD-related surgery.
Comparing short-term opioid users and nonusers revealed that short-term use more than doubled the risk of corticosteroid prescription (relative risk [RR], 2.517; P less than .001), and increased the risk of an emergency department visit by approximately 32% (RR, 1.315; P less than .001). Long-term use was associated with a similar doubling in risk of corticosteroid prescription (RR, 2.383; P less than .001), and an even greater risk of emergency department utilization (RR, 2.083; P less than .001). Risks of death or IBD-related surgery did not differ for either of these comparisons.
Next, the investigators compared long-term opioid use versus short-term opioid use. This suggested a duration-related effect, as long-term users were 57% more likely than were short-term users to utilize emergency department services (RR, 1.572; P less than .001). No significant differences for the other outcomes were detected in this comparison.
“Unlike previous studies, we did not find an association between opioid use and IBD-related surgery or death,” the investigators wrote. “Notably, these [previously reported] associations utilized opioid dosage (e.g., morphine equivalent or number of prescriptions), rather than length of opioid prescription (as we did). We also focused on IBD outpatients, while prior studies evaluated (in part or completely) inpatient populations, who typically present with more severe illness.”
Still, they added, the present findings should serve as a warning to prescribers considering even a short course of opioids for patients with IBD.
“This study demonstrates that prescribing opioids to IBD outpatients carries significant, specific risks, regardless of prescription length,” Ms. Telfer and colleagues wrote. “Healthcare professionals should exercise caution before prescribing these agents.”
The study was supported by the Peter and Marshia Carlino Early Career Professorship in Inflammatory Bowel Disease, the Margot E. Walrath Career Development Professorship in Gastroenterology, and the National Institutes of Health. The investigators disclosed no conflicts of interest.
Given that objective control of inflammation does not always correlate with improvement in abdominal pain scores, the use of opioids in patients with inflammatory bowel diseases (IBD) remains a difficult area of clinical practice and research. In this study, Telfer and colleagues performed a retrospective analysis using the TriNetX Diamond Network to assess the impact of opioid use on health-associated outcomes and evaluate for a differential impact on outcomes depending on the length of opioid prescription. When compared to non–opioid users, both short- and long-term opioid users were more likely to utilize corticosteroids and emergency department services. However, in contrast to prior studies, there was no increased risk for mortality demonstrated among those patients with short- or long-term opioid use.
Edward L. Barnes, MD, MPH, is assistant professor of medicine at the University of North Carolina at Chapel Hill. He disclosed having served as a consultant for Target RWE (not relevant to this commentary).
Given that objective control of inflammation does not always correlate with improvement in abdominal pain scores, the use of opioids in patients with inflammatory bowel diseases (IBD) remains a difficult area of clinical practice and research. In this study, Telfer and colleagues performed a retrospective analysis using the TriNetX Diamond Network to assess the impact of opioid use on health-associated outcomes and evaluate for a differential impact on outcomes depending on the length of opioid prescription. When compared to non–opioid users, both short- and long-term opioid users were more likely to utilize corticosteroids and emergency department services. However, in contrast to prior studies, there was no increased risk for mortality demonstrated among those patients with short- or long-term opioid use.
Edward L. Barnes, MD, MPH, is assistant professor of medicine at the University of North Carolina at Chapel Hill. He disclosed having served as a consultant for Target RWE (not relevant to this commentary).
Given that objective control of inflammation does not always correlate with improvement in abdominal pain scores, the use of opioids in patients with inflammatory bowel diseases (IBD) remains a difficult area of clinical practice and research. In this study, Telfer and colleagues performed a retrospective analysis using the TriNetX Diamond Network to assess the impact of opioid use on health-associated outcomes and evaluate for a differential impact on outcomes depending on the length of opioid prescription. When compared to non–opioid users, both short- and long-term opioid users were more likely to utilize corticosteroids and emergency department services. However, in contrast to prior studies, there was no increased risk for mortality demonstrated among those patients with short- or long-term opioid use.
Edward L. Barnes, MD, MPH, is assistant professor of medicine at the University of North Carolina at Chapel Hill. He disclosed having served as a consultant for Target RWE (not relevant to this commentary).
These findings amplify the safety signal from previous inpatient studies by showing that even a short course of opioids in an outpatient setting may increase risks of corticosteroid use and emergency department utilization, prompting caution among prescribers, reported Laura Telfer, MS, of Penn State College of Medicine, Hershey, Pa., and colleagues.
“Opioids are frequently prescribed to treat pain associated with IBD,” the investigators wrote in Gastro Hep Advances. “Unfortunately, they are associated with many problems in IBD, including increased risk of emergency room visits, hospitalization, surgery, and mortality. Chronic opioid use may also exacerbate symptoms and induce IBD flares, prompting discontinuation, thus increasing the risk of opioid withdrawal syndrome. Ironically, there is no published evidence that opioids even help to improve abdominal pain in IBD, particularly in the long term. Notably, most studies investigating opioid use in IBD have been limited to hospitalized patients, and few have directly evaluated the impact of opioid prescription length.”
To address this knowledge gap, Ms. Telfer and colleagues conducted a retrospective, population-based cohort study involving patients with IBD who were classified as either long-term opioid users, short-term opioid users, or nonusers. Drawing data from more than 80,000 patients in the TriNetX Diamond Network, the investigators evaluated relative, intergroup risks for corticosteroid use, emergency department utilization, mortality, and IBD-related surgery.
Comparing short-term opioid users and nonusers revealed that short-term use more than doubled the risk of corticosteroid prescription (relative risk [RR], 2.517; P less than .001), and increased the risk of an emergency department visit by approximately 32% (RR, 1.315; P less than .001). Long-term use was associated with a similar doubling in risk of corticosteroid prescription (RR, 2.383; P less than .001), and an even greater risk of emergency department utilization (RR, 2.083; P less than .001). Risks of death or IBD-related surgery did not differ for either of these comparisons.
Next, the investigators compared long-term opioid use versus short-term opioid use. This suggested a duration-related effect, as long-term users were 57% more likely than were short-term users to utilize emergency department services (RR, 1.572; P less than .001). No significant differences for the other outcomes were detected in this comparison.
“Unlike previous studies, we did not find an association between opioid use and IBD-related surgery or death,” the investigators wrote. “Notably, these [previously reported] associations utilized opioid dosage (e.g., morphine equivalent or number of prescriptions), rather than length of opioid prescription (as we did). We also focused on IBD outpatients, while prior studies evaluated (in part or completely) inpatient populations, who typically present with more severe illness.”
Still, they added, the present findings should serve as a warning to prescribers considering even a short course of opioids for patients with IBD.
“This study demonstrates that prescribing opioids to IBD outpatients carries significant, specific risks, regardless of prescription length,” Ms. Telfer and colleagues wrote. “Healthcare professionals should exercise caution before prescribing these agents.”
The study was supported by the Peter and Marshia Carlino Early Career Professorship in Inflammatory Bowel Disease, the Margot E. Walrath Career Development Professorship in Gastroenterology, and the National Institutes of Health. The investigators disclosed no conflicts of interest.
These findings amplify the safety signal from previous inpatient studies by showing that even a short course of opioids in an outpatient setting may increase risks of corticosteroid use and emergency department utilization, prompting caution among prescribers, reported Laura Telfer, MS, of Penn State College of Medicine, Hershey, Pa., and colleagues.
“Opioids are frequently prescribed to treat pain associated with IBD,” the investigators wrote in Gastro Hep Advances. “Unfortunately, they are associated with many problems in IBD, including increased risk of emergency room visits, hospitalization, surgery, and mortality. Chronic opioid use may also exacerbate symptoms and induce IBD flares, prompting discontinuation, thus increasing the risk of opioid withdrawal syndrome. Ironically, there is no published evidence that opioids even help to improve abdominal pain in IBD, particularly in the long term. Notably, most studies investigating opioid use in IBD have been limited to hospitalized patients, and few have directly evaluated the impact of opioid prescription length.”
To address this knowledge gap, Ms. Telfer and colleagues conducted a retrospective, population-based cohort study involving patients with IBD who were classified as either long-term opioid users, short-term opioid users, or nonusers. Drawing data from more than 80,000 patients in the TriNetX Diamond Network, the investigators evaluated relative, intergroup risks for corticosteroid use, emergency department utilization, mortality, and IBD-related surgery.
Comparing short-term opioid users and nonusers revealed that short-term use more than doubled the risk of corticosteroid prescription (relative risk [RR], 2.517; P less than .001), and increased the risk of an emergency department visit by approximately 32% (RR, 1.315; P less than .001). Long-term use was associated with a similar doubling in risk of corticosteroid prescription (RR, 2.383; P less than .001), and an even greater risk of emergency department utilization (RR, 2.083; P less than .001). Risks of death or IBD-related surgery did not differ for either of these comparisons.
Next, the investigators compared long-term opioid use versus short-term opioid use. This suggested a duration-related effect, as long-term users were 57% more likely than were short-term users to utilize emergency department services (RR, 1.572; P less than .001). No significant differences for the other outcomes were detected in this comparison.
“Unlike previous studies, we did not find an association between opioid use and IBD-related surgery or death,” the investigators wrote. “Notably, these [previously reported] associations utilized opioid dosage (e.g., morphine equivalent or number of prescriptions), rather than length of opioid prescription (as we did). We also focused on IBD outpatients, while prior studies evaluated (in part or completely) inpatient populations, who typically present with more severe illness.”
Still, they added, the present findings should serve as a warning to prescribers considering even a short course of opioids for patients with IBD.
“This study demonstrates that prescribing opioids to IBD outpatients carries significant, specific risks, regardless of prescription length,” Ms. Telfer and colleagues wrote. “Healthcare professionals should exercise caution before prescribing these agents.”
The study was supported by the Peter and Marshia Carlino Early Career Professorship in Inflammatory Bowel Disease, the Margot E. Walrath Career Development Professorship in Gastroenterology, and the National Institutes of Health. The investigators disclosed no conflicts of interest.
FROM GASTRO HEP ADVANCES
Particulate pollution increases the risk for breast cancer
MADRID – according to a new analysis of the XENAIR study presented at the European Society of Medical Oncology (ESMO) Congress 2023. Béatrice Fervers, MD, PhD, head of the environmental cancer prevention department at the Léon Bérard Center, Lyon, France, presented her findings.
“To our knowledge, this study is the first to examine the risk of breast cancer associated with long-term exposure of subjects to atmospheric pollution both at home and in the workplace, estimated using a very small spatial resolution [statistical] model,” said the researchers.
“Our data showed a statistically significant association between long-term exposure to fine particulate matter air pollution, at home and at work, and risk of breast cancer. This [finding] contrasts with previous research that looked only at fine particulate exposure where women were living and showed small or no effects on breast cancer risk,” said Dr. Fervers in a press release issued before the Congress.
The XENAIR study carried out on the prospective, longitudinal E3N cohort a year ago showed an increased risk for breast cancer after exposure to five atmospheric pollutants. Notably, it showed an increased risk in women exposed to BaP and PCB153, two pollutants classed as endocrine-disrupting chemicals, during perimenopause.
Increased linear risk
In this new analysis, exposure to PM2.5, PM10, and NO2 pollution at home and in the workplace of 2,419 women with breast cancer was compared with that of 2,984 women without breast cancer during the period from 1990 to 2011.
This was a case-control study in which participants were matched by department of residence in France, age (± 1 year), date (± 3 months), and menopausal status at the time of the blood draw.
Breast cancer risk increased by 28% when exposure to fine particulate (PM2.5) air pollution increased by 10 mcg/m3. The increment is approximately equivalent to the difference in PM2.5 particulate concentration typically seen in rural versus urban areas of Europe.
Smaller increases in breast cancer risk were also recorded in women exposed to high levels of larger particulate air pollution (PM10 and NO2).
No change in effect was seen according to menopausal status. Analyses that examined hormone receptor status showed a positive but not significant association for PM2.5 in cases of estrogen receptor positive breast cancer.
Dr. Fervers and colleagues plan to investigate the effects of pollution exposure during the commute to get a complete picture of effects on breast cancer risk.
Regulators respond
Charles Swanton, PhD, a clinician scientist at the Francis Crick Institute, London, emphasized the importance of these new results for breast cancer. At last year’s ESMO Congress, he explained how particulate matter air pollution caused tumor proliferation in patients with a certain type of genetic mutation.
“Fine particle pollutants can penetrate deep into the lungs, enter the bloodstream, and be absorbed into breast and other tissue. There is already evidence that air pollutants can change the architecture of the breast. It will be important to test if pollutants allow cells in breast tissue with pre-existing mutations to expand and drive tumor promotion, possibly through inflammatory processes, similar to our observations in nonsmokers with lung cancer,” said Dr. Swanton in the ESMO press release.
“It is very concerning that small pollutant particles in the air and indeed microplastic particles of similar size are getting into the environment when we don’t yet understand their potential to promote cancer. There is an urgent need to set up laboratory studies to investigate the effects of these small air pollutant particles on the latency, grade, aggression, and progression of breast tumors,” he added.
“There is now strong epidemiological and biological evidence for the link between PM2.5 particulate exposure and cancer, and there are good clinical and economic reasons for reducing pollution to prevent cancers,” said Jean-Yves Blay, MD, PhD, director of public policy for ESMO.
Following a proposal from the European Commission in October 2022 to reduce the limit for PM2.5 particulates in the air from the current 25 mcg/m3 to 10 mcg/m3 by 2030, ESMO urged a further reduction in the PM2.5 limit to 5 mcg/m3, in line with the World Health Organization’s air quality guidance, according to the press release.
“Reducing PM2.5 particles in the air to the WHO recommended level is critical because of their association with a variety of tumor types, including breast cancer,” Dr. Blay added.
In September 2023, the European Parliament adopted in a plenary session its report on the ongoing revision of the EU Ambient Air Quality Directives, which reflects ESMO’s recommendations to set the annual limit value for PM2.5 at 5 mcg/m³. This adoption opens interinstitutional negotiations between the legislators (the European Parliament, European Commission, and EU Council) to agree on the final text of the directive.
“By supporting our requests with solid scientific evidence, we are offering a new dimension to health public policy. The work is not over, and change will not happen overnight, but we are moving in the right direction,” concluded Dr. Blay.
The new analysis of the XENAIR study was funded by ARC Foundation for cancer research; the French Agency for Food, Environmental, and Occupational Health and Safety; French National League against Cancer; and Fondation de France, an independent private organization, recognized as being in the public interest. The authors report no relevant financial relationships.
This article was translated from the Medscape French edition and a version appeared on Medscape.com.
MADRID – according to a new analysis of the XENAIR study presented at the European Society of Medical Oncology (ESMO) Congress 2023. Béatrice Fervers, MD, PhD, head of the environmental cancer prevention department at the Léon Bérard Center, Lyon, France, presented her findings.
“To our knowledge, this study is the first to examine the risk of breast cancer associated with long-term exposure of subjects to atmospheric pollution both at home and in the workplace, estimated using a very small spatial resolution [statistical] model,” said the researchers.
“Our data showed a statistically significant association between long-term exposure to fine particulate matter air pollution, at home and at work, and risk of breast cancer. This [finding] contrasts with previous research that looked only at fine particulate exposure where women were living and showed small or no effects on breast cancer risk,” said Dr. Fervers in a press release issued before the Congress.
The XENAIR study carried out on the prospective, longitudinal E3N cohort a year ago showed an increased risk for breast cancer after exposure to five atmospheric pollutants. Notably, it showed an increased risk in women exposed to BaP and PCB153, two pollutants classed as endocrine-disrupting chemicals, during perimenopause.
Increased linear risk
In this new analysis, exposure to PM2.5, PM10, and NO2 pollution at home and in the workplace of 2,419 women with breast cancer was compared with that of 2,984 women without breast cancer during the period from 1990 to 2011.
This was a case-control study in which participants were matched by department of residence in France, age (± 1 year), date (± 3 months), and menopausal status at the time of the blood draw.
Breast cancer risk increased by 28% when exposure to fine particulate (PM2.5) air pollution increased by 10 mcg/m3. The increment is approximately equivalent to the difference in PM2.5 particulate concentration typically seen in rural versus urban areas of Europe.
Smaller increases in breast cancer risk were also recorded in women exposed to high levels of larger particulate air pollution (PM10 and NO2).
No change in effect was seen according to menopausal status. Analyses that examined hormone receptor status showed a positive but not significant association for PM2.5 in cases of estrogen receptor positive breast cancer.
Dr. Fervers and colleagues plan to investigate the effects of pollution exposure during the commute to get a complete picture of effects on breast cancer risk.
Regulators respond
Charles Swanton, PhD, a clinician scientist at the Francis Crick Institute, London, emphasized the importance of these new results for breast cancer. At last year’s ESMO Congress, he explained how particulate matter air pollution caused tumor proliferation in patients with a certain type of genetic mutation.
“Fine particle pollutants can penetrate deep into the lungs, enter the bloodstream, and be absorbed into breast and other tissue. There is already evidence that air pollutants can change the architecture of the breast. It will be important to test if pollutants allow cells in breast tissue with pre-existing mutations to expand and drive tumor promotion, possibly through inflammatory processes, similar to our observations in nonsmokers with lung cancer,” said Dr. Swanton in the ESMO press release.
“It is very concerning that small pollutant particles in the air and indeed microplastic particles of similar size are getting into the environment when we don’t yet understand their potential to promote cancer. There is an urgent need to set up laboratory studies to investigate the effects of these small air pollutant particles on the latency, grade, aggression, and progression of breast tumors,” he added.
“There is now strong epidemiological and biological evidence for the link between PM2.5 particulate exposure and cancer, and there are good clinical and economic reasons for reducing pollution to prevent cancers,” said Jean-Yves Blay, MD, PhD, director of public policy for ESMO.
Following a proposal from the European Commission in October 2022 to reduce the limit for PM2.5 particulates in the air from the current 25 mcg/m3 to 10 mcg/m3 by 2030, ESMO urged a further reduction in the PM2.5 limit to 5 mcg/m3, in line with the World Health Organization’s air quality guidance, according to the press release.
“Reducing PM2.5 particles in the air to the WHO recommended level is critical because of their association with a variety of tumor types, including breast cancer,” Dr. Blay added.
In September 2023, the European Parliament adopted in a plenary session its report on the ongoing revision of the EU Ambient Air Quality Directives, which reflects ESMO’s recommendations to set the annual limit value for PM2.5 at 5 mcg/m³. This adoption opens interinstitutional negotiations between the legislators (the European Parliament, European Commission, and EU Council) to agree on the final text of the directive.
“By supporting our requests with solid scientific evidence, we are offering a new dimension to health public policy. The work is not over, and change will not happen overnight, but we are moving in the right direction,” concluded Dr. Blay.
The new analysis of the XENAIR study was funded by ARC Foundation for cancer research; the French Agency for Food, Environmental, and Occupational Health and Safety; French National League against Cancer; and Fondation de France, an independent private organization, recognized as being in the public interest. The authors report no relevant financial relationships.
This article was translated from the Medscape French edition and a version appeared on Medscape.com.
MADRID – according to a new analysis of the XENAIR study presented at the European Society of Medical Oncology (ESMO) Congress 2023. Béatrice Fervers, MD, PhD, head of the environmental cancer prevention department at the Léon Bérard Center, Lyon, France, presented her findings.
“To our knowledge, this study is the first to examine the risk of breast cancer associated with long-term exposure of subjects to atmospheric pollution both at home and in the workplace, estimated using a very small spatial resolution [statistical] model,” said the researchers.
“Our data showed a statistically significant association between long-term exposure to fine particulate matter air pollution, at home and at work, and risk of breast cancer. This [finding] contrasts with previous research that looked only at fine particulate exposure where women were living and showed small or no effects on breast cancer risk,” said Dr. Fervers in a press release issued before the Congress.
The XENAIR study carried out on the prospective, longitudinal E3N cohort a year ago showed an increased risk for breast cancer after exposure to five atmospheric pollutants. Notably, it showed an increased risk in women exposed to BaP and PCB153, two pollutants classed as endocrine-disrupting chemicals, during perimenopause.
Increased linear risk
In this new analysis, exposure to PM2.5, PM10, and NO2 pollution at home and in the workplace of 2,419 women with breast cancer was compared with that of 2,984 women without breast cancer during the period from 1990 to 2011.
This was a case-control study in which participants were matched by department of residence in France, age (± 1 year), date (± 3 months), and menopausal status at the time of the blood draw.
Breast cancer risk increased by 28% when exposure to fine particulate (PM2.5) air pollution increased by 10 mcg/m3. The increment is approximately equivalent to the difference in PM2.5 particulate concentration typically seen in rural versus urban areas of Europe.
Smaller increases in breast cancer risk were also recorded in women exposed to high levels of larger particulate air pollution (PM10 and NO2).
No change in effect was seen according to menopausal status. Analyses that examined hormone receptor status showed a positive but not significant association for PM2.5 in cases of estrogen receptor positive breast cancer.
Dr. Fervers and colleagues plan to investigate the effects of pollution exposure during the commute to get a complete picture of effects on breast cancer risk.
Regulators respond
Charles Swanton, PhD, a clinician scientist at the Francis Crick Institute, London, emphasized the importance of these new results for breast cancer. At last year’s ESMO Congress, he explained how particulate matter air pollution caused tumor proliferation in patients with a certain type of genetic mutation.
“Fine particle pollutants can penetrate deep into the lungs, enter the bloodstream, and be absorbed into breast and other tissue. There is already evidence that air pollutants can change the architecture of the breast. It will be important to test if pollutants allow cells in breast tissue with pre-existing mutations to expand and drive tumor promotion, possibly through inflammatory processes, similar to our observations in nonsmokers with lung cancer,” said Dr. Swanton in the ESMO press release.
“It is very concerning that small pollutant particles in the air and indeed microplastic particles of similar size are getting into the environment when we don’t yet understand their potential to promote cancer. There is an urgent need to set up laboratory studies to investigate the effects of these small air pollutant particles on the latency, grade, aggression, and progression of breast tumors,” he added.
“There is now strong epidemiological and biological evidence for the link between PM2.5 particulate exposure and cancer, and there are good clinical and economic reasons for reducing pollution to prevent cancers,” said Jean-Yves Blay, MD, PhD, director of public policy for ESMO.
Following a proposal from the European Commission in October 2022 to reduce the limit for PM2.5 particulates in the air from the current 25 mcg/m3 to 10 mcg/m3 by 2030, ESMO urged a further reduction in the PM2.5 limit to 5 mcg/m3, in line with the World Health Organization’s air quality guidance, according to the press release.
“Reducing PM2.5 particles in the air to the WHO recommended level is critical because of their association with a variety of tumor types, including breast cancer,” Dr. Blay added.
In September 2023, the European Parliament adopted in a plenary session its report on the ongoing revision of the EU Ambient Air Quality Directives, which reflects ESMO’s recommendations to set the annual limit value for PM2.5 at 5 mcg/m³. This adoption opens interinstitutional negotiations between the legislators (the European Parliament, European Commission, and EU Council) to agree on the final text of the directive.
“By supporting our requests with solid scientific evidence, we are offering a new dimension to health public policy. The work is not over, and change will not happen overnight, but we are moving in the right direction,” concluded Dr. Blay.
The new analysis of the XENAIR study was funded by ARC Foundation for cancer research; the French Agency for Food, Environmental, and Occupational Health and Safety; French National League against Cancer; and Fondation de France, an independent private organization, recognized as being in the public interest. The authors report no relevant financial relationships.
This article was translated from the Medscape French edition and a version appeared on Medscape.com.
AT ESMO 2023
Standing BP measures improve hypertension diagnosis
TOPLINE:
results of a new study suggest.
METHODOLOGY:
- The study included 125 adults, mean age 49 years and 62% female, who were free of cardiovascular disease and had no previous history of hypertension.
- Researchers collected data on 24-hour ambulatory blood pressure monitoring (ABPM), and three BP measurements in the seated position, then three in the standing position.
- They assessed overall diagnostic accuracy of seated and standing BP using the area under the receiver operating characteristic (AUROC) curve and considered a Bayes factor (BF) of 3 or greater as significant.
- They defined the presence of hypertension (HTN) by the 2017 American College of Cardiology/American Heart Association and 2023 European Society of Hypertension HTN guidelines based on ABPM.
- Sensitivity and specificity of standing BP was determined using cutoffs derived from Youden index, while sensitivity and specificity of seated BP was determined using the cutoff of 130/80 mm Hg and by 140/90 mm Hg.
TAKEAWAY:
- The AUROC for standing office systolic blood pressure (SBP; 0.81; 0.71-0.92) was significantly higher than for seated office SBP (0.70; 0.49-0.91) in diagnosing HTN when defined as an average 24-hour SBP ≥ 125 mm Hg (BF = 11.8), and significantly higher for seated versus standing office diastolic blood pressure (DBP; 0.65; 0.49-0.82) in diagnosing HTN when defined as an average 24-hour DBP ≥ 75 mm Hg (BF = 4.9).
- The AUROCs for adding standing office BP to seated office BP improved the accuracy of detecting HTN, compared with seated office BP alone when HTN was defined as an average 24-hour SBP/DBP ≥ 125/75 mm Hg or daytime SBP/DBP ≥ 130/80 mm Hg, or when defined as an average 24-hour SBP/DBP ≥ 130/80 mm Hg or daytime SBP/DBP ≥ 135/85 mm Hg (all BFs > 3).
- Sensitivity of standing SBP was 71%, compared with 43% for seated SBP.
IN PRACTICE:
The “excellent diagnostic performance” for standing BP measures revealed by the study “highlights that standing office BP has acceptable discriminative capabilities in identifying the presence of hypertension in adults,” the authors write.
SOURCE:
The study was conducted by John M. Giacona, Hypertension Section, department of internal medicine, University of Texas Southwestern Medical Center, Dallas, and colleagues. It was published online in Scientific Reports.
LIMITATIONS:
As the study enrolled only adults free of comorbidities who were not taking antihypertensive medications, the results may not be applicable to other patients. The study design was retrospective, and the order of BP measurements was not randomized (standing BP measurements were obtained only after seated BP).
DISCLOSURES:
The study was supported by the National Institutes of Health. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a new study suggest.
METHODOLOGY:
- The study included 125 adults, mean age 49 years and 62% female, who were free of cardiovascular disease and had no previous history of hypertension.
- Researchers collected data on 24-hour ambulatory blood pressure monitoring (ABPM), and three BP measurements in the seated position, then three in the standing position.
- They assessed overall diagnostic accuracy of seated and standing BP using the area under the receiver operating characteristic (AUROC) curve and considered a Bayes factor (BF) of 3 or greater as significant.
- They defined the presence of hypertension (HTN) by the 2017 American College of Cardiology/American Heart Association and 2023 European Society of Hypertension HTN guidelines based on ABPM.
- Sensitivity and specificity of standing BP was determined using cutoffs derived from Youden index, while sensitivity and specificity of seated BP was determined using the cutoff of 130/80 mm Hg and by 140/90 mm Hg.
TAKEAWAY:
- The AUROC for standing office systolic blood pressure (SBP; 0.81; 0.71-0.92) was significantly higher than for seated office SBP (0.70; 0.49-0.91) in diagnosing HTN when defined as an average 24-hour SBP ≥ 125 mm Hg (BF = 11.8), and significantly higher for seated versus standing office diastolic blood pressure (DBP; 0.65; 0.49-0.82) in diagnosing HTN when defined as an average 24-hour DBP ≥ 75 mm Hg (BF = 4.9).
- The AUROCs for adding standing office BP to seated office BP improved the accuracy of detecting HTN, compared with seated office BP alone when HTN was defined as an average 24-hour SBP/DBP ≥ 125/75 mm Hg or daytime SBP/DBP ≥ 130/80 mm Hg, or when defined as an average 24-hour SBP/DBP ≥ 130/80 mm Hg or daytime SBP/DBP ≥ 135/85 mm Hg (all BFs > 3).
- Sensitivity of standing SBP was 71%, compared with 43% for seated SBP.
IN PRACTICE:
The “excellent diagnostic performance” for standing BP measures revealed by the study “highlights that standing office BP has acceptable discriminative capabilities in identifying the presence of hypertension in adults,” the authors write.
SOURCE:
The study was conducted by John M. Giacona, Hypertension Section, department of internal medicine, University of Texas Southwestern Medical Center, Dallas, and colleagues. It was published online in Scientific Reports.
LIMITATIONS:
As the study enrolled only adults free of comorbidities who were not taking antihypertensive medications, the results may not be applicable to other patients. The study design was retrospective, and the order of BP measurements was not randomized (standing BP measurements were obtained only after seated BP).
DISCLOSURES:
The study was supported by the National Institutes of Health. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
results of a new study suggest.
METHODOLOGY:
- The study included 125 adults, mean age 49 years and 62% female, who were free of cardiovascular disease and had no previous history of hypertension.
- Researchers collected data on 24-hour ambulatory blood pressure monitoring (ABPM), and three BP measurements in the seated position, then three in the standing position.
- They assessed overall diagnostic accuracy of seated and standing BP using the area under the receiver operating characteristic (AUROC) curve and considered a Bayes factor (BF) of 3 or greater as significant.
- They defined the presence of hypertension (HTN) by the 2017 American College of Cardiology/American Heart Association and 2023 European Society of Hypertension HTN guidelines based on ABPM.
- Sensitivity and specificity of standing BP was determined using cutoffs derived from Youden index, while sensitivity and specificity of seated BP was determined using the cutoff of 130/80 mm Hg and by 140/90 mm Hg.
TAKEAWAY:
- The AUROC for standing office systolic blood pressure (SBP; 0.81; 0.71-0.92) was significantly higher than for seated office SBP (0.70; 0.49-0.91) in diagnosing HTN when defined as an average 24-hour SBP ≥ 125 mm Hg (BF = 11.8), and significantly higher for seated versus standing office diastolic blood pressure (DBP; 0.65; 0.49-0.82) in diagnosing HTN when defined as an average 24-hour DBP ≥ 75 mm Hg (BF = 4.9).
- The AUROCs for adding standing office BP to seated office BP improved the accuracy of detecting HTN, compared with seated office BP alone when HTN was defined as an average 24-hour SBP/DBP ≥ 125/75 mm Hg or daytime SBP/DBP ≥ 130/80 mm Hg, or when defined as an average 24-hour SBP/DBP ≥ 130/80 mm Hg or daytime SBP/DBP ≥ 135/85 mm Hg (all BFs > 3).
- Sensitivity of standing SBP was 71%, compared with 43% for seated SBP.
IN PRACTICE:
The “excellent diagnostic performance” for standing BP measures revealed by the study “highlights that standing office BP has acceptable discriminative capabilities in identifying the presence of hypertension in adults,” the authors write.
SOURCE:
The study was conducted by John M. Giacona, Hypertension Section, department of internal medicine, University of Texas Southwestern Medical Center, Dallas, and colleagues. It was published online in Scientific Reports.
LIMITATIONS:
As the study enrolled only adults free of comorbidities who were not taking antihypertensive medications, the results may not be applicable to other patients. The study design was retrospective, and the order of BP measurements was not randomized (standing BP measurements were obtained only after seated BP).
DISCLOSURES:
The study was supported by the National Institutes of Health. The authors have no relevant conflicts of interest.
A version of this article appeared on Medscape.com.