User login
Pharmacologic Treatments for Idiopathic Pulmonary Fibrosis
IN THIS ARTICLE
- Confirming the diagnosis
- Pirfenidone treatment
- Nintedanib treatment
A 64-year-old man has a one-year history of dyspnea on exertion and a nonproductive cough. His symptoms are gradually worsening and increasingly bothersome to him.
His medical history includes mild seasonal allergies and GERD, which is well-controlled by oral antihistamines and proton pump inhibitors. He has spent the past 30 years working a desk job as an accountant. He denies a history of smoking, exposure to secondhand smoke, and initiation of new medication.
He admits to increased fatigue, but denies fever, chills, lymphadenopathy, weight change, chest pain, wheezing, abdominal pain, diarrhea, vomiting, claudication, and swelling in the extremities. The rest of the review of systems is negative.
Lab results—complete blood count, comprehensive metabolic panel, TSH, antinuclear antibodies, erythrocyte sedimentation rate, and C-reactive protein—are within normal limits. Spirometry shows very mild restriction. A chest x-ray is abnormal but nonspecific, showing peripheral opacities. An ECG shows normal sinus rhythm.
The patient is given a trial of an inhaled steroid, which yields no improvement. Six months later, the patient is seen by a pulmonologist. Idiopathic pulmonary fibrosis (IPF) is diagnosed based on high-resolution CT (HRCT) and lung biopsy results.
IPF is a chronic, progressive, fibrosing interstitial disease that is limited to lung tissue. It most commonly manifests in older adults with vague symptoms of dyspnea on exertion and nonproductive cough, but symptoms can also include fatigue, muscle and joint aches, clubbing of the fingernails, and weight loss.1 The average life expectancy following diagnosis of IPF is two to five years, and the mortality rate is estimated at 64.3 per million men and 58.4 per million women per year.2,3
Continue to: DIAGNOSIS
DIAGNOSIS
IPF belongs in the general class of idiopathic interstitial pneumonias (IIPs), which are characterized by varying degrees of inflammation and fibrosis of lung interstitium.4 All subtypes of IIPs cause dyspnea and diffuse abnormalities on HRCT, and all vary from each other histologically. Table 1 outlines the key features of each.5-8
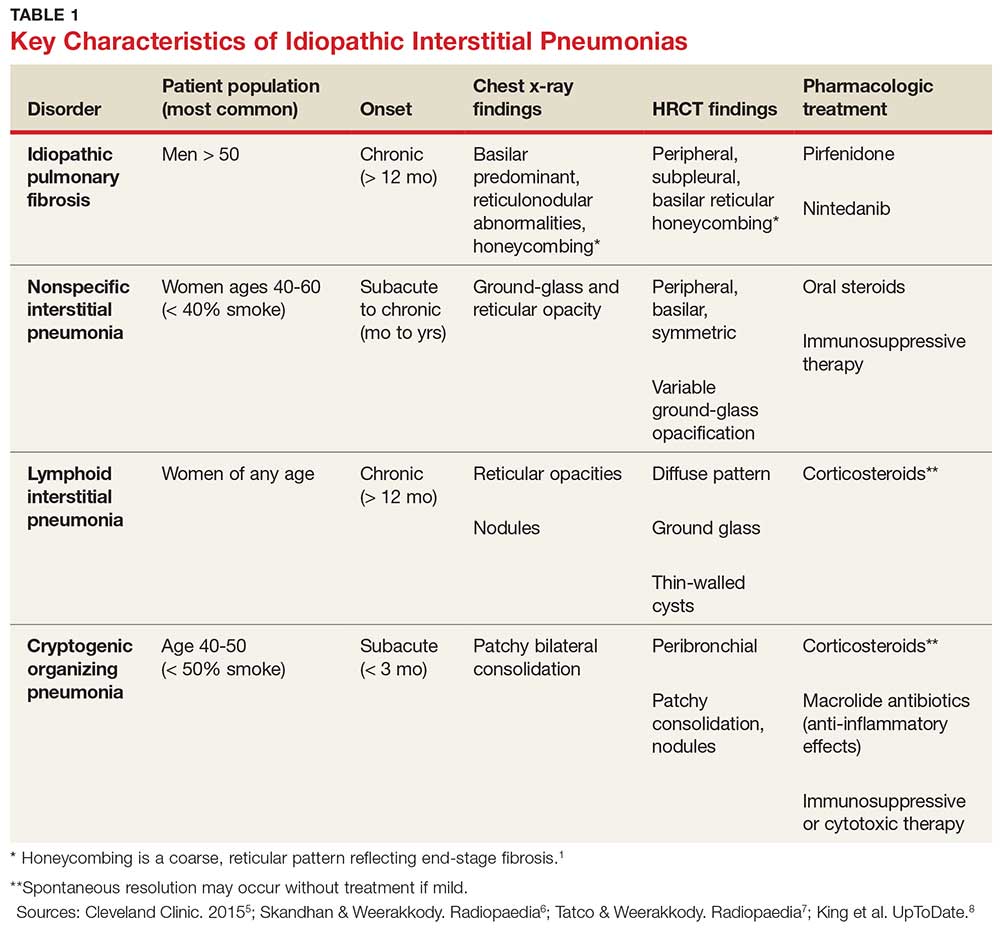
Because of its vague symptomology and the extensive workup needed to rule out other diseases, patients with IPF often have symptoms for one to two years before a diagnosis is made.1 Physical exam may reveal fine inspiratory rales in both lung bases and digital clubbing; eventual signs of pulmonary hypertension and right-sided heart failure may be appreciated.1,9
There are no specific diagnostic laboratory tests to confirm IPF; however, baseline labwork (as outlined in the case presentation) is typically ordered to rule out infection, thyroid disease, or connective tissue disease.10 Many patients are referred to a cardiologist before being seen by a pulmonologist; cardiac stress testing may be done, and an echocardiogram may be performed to rule out heart failure.
Diagnostic testing may include pulmonary function testing, HRCT of the chest, and lung biopsy.10 Tissue samples from patients with IPF reveal different stages of disease, including dense fibrosis with honeycombing, subpleural or paraseptal distribution, fibroblast foci, and normal tissue.11 Pulmonary function test results will show a restrictive pattern. Both forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) will be reduced, and the FEV1/FVC ratio preserved. Due to decreased functional lung volume, diffusing capacity of the lung for carbon monoxide (DLCO) will also be reduced.4,12
The differential is broad and includes allergic asthma, bronchitis, COPD, lung cancer, hypersensitivity pneumonitis, asbestosis, or pulmonary embolism.
Continue to: TREATMENT HISTORY
TREATMENT HISTORY
IPF has a long history of tried and failed treatment options. The American Thoracic Society (ATS), in concert with other professional organizations, has published comprehensive guidelines and recommendations pertaining to the use of pharmacologic medications to control disease progression. Warfarin and other anticoagulants have been studied, based on the observation that a procoagulant state promotes fibrotic changes in the lung tissue.13 However, anticoagulant use is not recommended in patients with IPF due to lack of efficacy and high potential for harm.13
Immunosuppressants have also been in the spotlight as possible treatment for IPF, but a clinical study investigating the efficacy of a three-drug regimen including prednisone, azathioprine, and N-acetylcysteine was stopped early due to increased risk for harm. Endothelin antagonists and potent tyrosine kinase inhibitors are also not recommended in the most recent edition of IPF guidelines, as they lack benefit.13
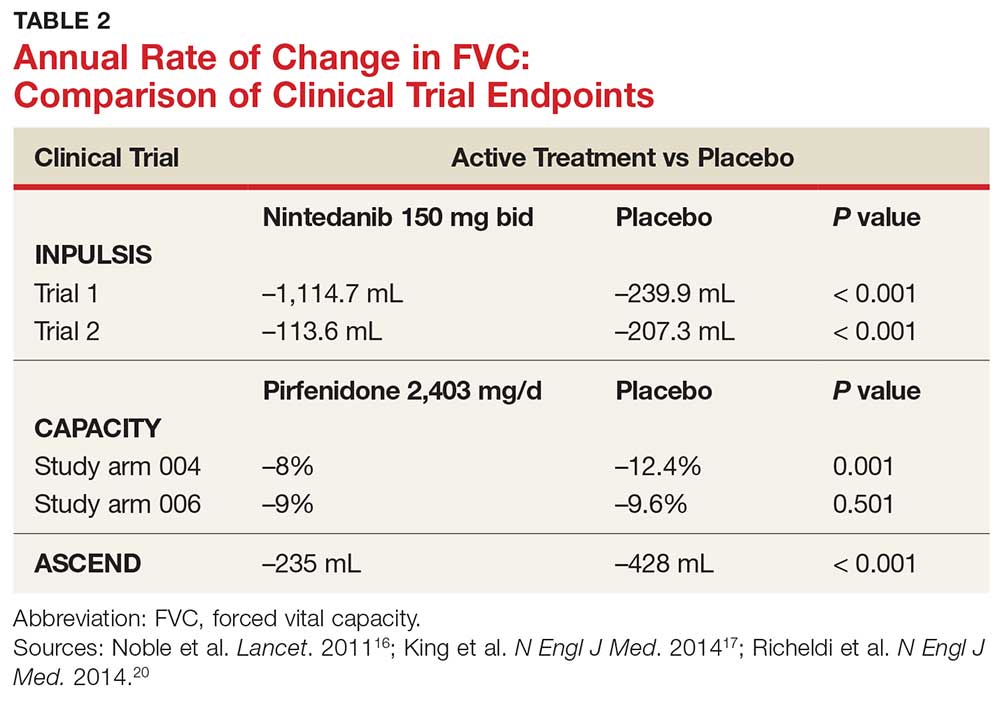
In fact, prior to the 2015 edition of the guidelines, no single medication was routinely recommended for patients with IPF. But this is now changing, following the 2014 FDA approval of two new drugs, nintedanib and pirfenidone, designed specifically to treat IPF.14 These drugs have shown promise in clinical trials (results of which are summarized in Table 2).
Continue to: NEW PHARMACOLOGIC OPTIONS
NEW PHARMACOLOGIC OPTIONS
Pirfenidone
In 2008, a study was conducted in Japan to determine the mechanism of action of pirfenidone.15 Through in vitro studies of healthy adult lung fibroblasts with added pro-fibrotic factor and transforming growth factor (TGF-ß 1), the researchers found that pirfenidone was effective at decreasing the production of a collagen-binding protein called HSP47. This protein is ubiquitous in fibrotic tissue. The study also showed that pirfenidone decreased the production of collagen type 1, which, when uninhibited, increases fibrosis.15
CAPACITY trials. In the CAPACITY trials, two phase 3 multinational studies conducted from 2006 to 2008, patients were given either pirfenidone or placebo.16 In the first study arm, patients were assigned to pirfenidone 2,403 mg/d (n = 174), pirfenidone 1,197 mg/d (n = 87), or placebo (n = 174). In the second study arm, 171 patients received pirfenidone 2,403 mg/d and 173 patients received placebo. Endpoints were measured at baseline and up to week 72.
The first study arm found that the mean rate of decline of FVC—the primary endpoint—was 4.4% less in the treatment group than in the placebo group (p = 0.001), and there was a 36% decrease in risk for death or disease progression in the treatment group (HR, 0.64; p, 0.023). (Endpoints were defined as: time to confirmed > 10% decline in percentage predicted FVC, > 15% decline in percentage predicted DLCO, or death.) The researchers found no clinically significant change in the six-minute walk test—a secondary endpoint of the study.16
The second study arm, however, found no statistically significant change in FVC between the treatment and placebo groups (with a 0.6% smaller decrease in FVC in the pirfenidone group), nor did they see a difference in progression-free survival. However, there was a significant change in the six-minute walk test between the treatment and placebo groups (p = 0.0009). Throughout the study, the most common adverse effects included nausea (36%), rash (32%), and dyspepsia (19%).16
ASCEND trial. The 2014 Assessment of Pirfenidone to Confirm Efficacy and Safety in Idiopathic Pulmonary Fibrosis (ASCEND) trial was a phase 3, multinational, randomized, double-blind, placebo-controlled study of the use of pirfenidone 2,403 mg/d.17 The study was conducted from 2012 to 2013. Of the total number of patients (N = 522), half received pirfenidone and half received placebo. After 52 weeks of treatment (the end of the study), the researchers found a smaller decline in FVC—the primary endpoint—in the treatment group compared to placebo (mean decline, 235 mL vs 428 mL, respectively [p < 0.001]). Regarding the six-minute walk test, the investigators found that 25.9% of the treatment group exhibited a decrease of ≥ 50 meters, compared to 35.7% of the placebo group (p = 0.04). (Progression-free survival was defined as a confirmed ≥ 10% decrease in predicted FVC, a confirmed decrease of 50 meters in the six-minute walk test, or death.)
The pirfenidone group in the ASCEND trial showed a 43% reduced risk for death or disease progression (HR, 0.57; p, < 0.001).16,17 All-cause mortality was lower in the pirfenidone group (4%) than in the placebo group (7.2%), but this was not statistically significant. Deaths from IPF in the pirfenidone group totaled three patients (1.1%) versus seven patients (2.5%) in the placebo group; this was also not statistically significant. The most common adverse effects seen during the study were nausea (36%), rash (28.1%), and headache (25.9%).17
Recommendations for use. Liver function testing should be performed at baseline, monthly for six months, and every three months afterward, as elevations in liver enzymes have been observed.18 Pirfenidone is a CYP1A2 substrate; moderate-to-strong CYP1A2 inhibitors should therefore be discontinued prior to initiation, as they are likely to decrease exposure and efficacy of pirfenidone. There are currently no black box warnings.18
Continue to: Nintedanib
Nintedanib
Hostettler et al studied lung samples from patients with IPF to determine the mechanism of action of nintedanib.19 Evaluation of fibroblasts derived from IPF samples revealed that they contained higher levels of platelet-derived growth factor (PDGF) than did nonfibrotic control cells. They also found that nintedanib, a tyrosine kinase inhibitor, significantly inhibited the phosphorylation of fibrotic-inducing growth factors—PDGF as well as vascular endothelial growth factor (VEGF).
INPULSIS trials. A phase 3 replicate of randomized, double-blind, multinational studies, the INPULSIS trials were performed between 2011 and 2012.20 Two study arms were used to evaluate a total of 638 patients who received nintedanib 150 mg bid for 52 weeks. The primary endpoint was annual rate of decline of FVC.
The researchers also evaluated efficacy through two other endpoints: patient-reported quality of life and symptoms via the St. George’s Respiratory Questionnaire (SGRQ) and evaluation of time to acute exacerbation. The latter was defined as worsening or new dyspnea, new diffuse pulmonary infiltrates visualized on chest radiography and/or HRCT, or the development of parenchymal abnormalities with no pneumothorax or pleural effusion since the preceding visit; and exclusion of any known causes of acute worsening, including infection, heart failure, pulmonary embolism, and any identifiable cause of acute lung injury.20
INPULSIS 1 (first arm) included 309 patients in the treatment group. Results showed an adjusted annual rate of decline in FVC of 114.7 mL/year, versus 239.9 mL/year in the placebo group (p < 0.001). In the treatment group, 52.8% exhibited ≤ 5% decline in FVC, compared to 38.2% in the placebo group (p = 0.001). No significant between-group differences were found in SGRQ score or time to acute exacerbation.20
INPULSIS 2 had 329 patients receiving nintedanib. An annual rate of decline in FVC of 113.6 mL/year from baseline was observed in the treatment group, compared to 207.3 mL/year in the placebo group (p < 0.001). In the treatment group, 53.2% showed ≤ 5% decline in FVC, versus 39.3% in the placebo group (p = 0.001). There was also a significantly smaller increase in total SGRQ score (meaning, less deterioration in quality of life) in the nintedanib group versus the placebo group (p = 0.02). A statistically significant increase in time to first acute exacerbation was observed in the nintedanib group (p = 0.005).20
There was no significant difference between groups in death from any cause, death from respiratory causation, or death that occurred between randomization and 28 days post treatment. The most common adverse effects seen throughout the two trials included diarrhea (trial 1, 61.5%; trial 2, 63.2%), nausea (trial 1, 22.7%; trial 2, 26.1%), and nasopharyngitis (trial 1, 12.6%; trial 2, 14.6%).20
Recommendations for use. Liver function testing should be performed at baseline, at regular intervals during the first three months, then periodically thereafter; patients in the treatment group of both INPULSIS trials had elevated liver enzymes, and cases of drug-induced liver injury have been observed with use of nintedanib.21 This medication may increase risk for bleeding due to its mechanism of action (VEGFR inhibition). Coadministration with CYP3A4 inhibitors may increase concentration of nintedanib; therefore, close monitoring is recommended. Avoid coadministration with CYP3A4 inducers, as this may decrease concentration of nintedanib by 50%. There are currently no black box warnings.21
Continue to: Patient monitoring
Patient monitoring
The ATS recommends measuring FVC and DLCO every three to six months, or sooner if clinically indicated.13 Pulse oximetry should be measured at rest and on exertion in all patients, regardless of symptoms, to assure proper saturation and identify the need for supplemental oxygen; this should also be done every three to six months.
The ATS recommends prompt detection and treatment of comorbidities such as pulmonary hypertension, emphysema, airflow obstruction, GERD, sleep apnea, and coronary artery disease.13 These recommendations are based on the organization’s 2015 guidelines.
OUTCOME FOR THE CASE PATIENT
The patient was started on pirfenidone (2,403 mg/d). He is continuing treatment and showing improvements in quality of life and slowed deterioration of lung function.
CONCLUSION
IPF causes progressive fibrosis of lung interstitium. The etiology is unknown, the symptoms and signs are vague, and mean life expectancy following diagnosis is two to five years. The most recent IPF guidelines recommend avoiding use of anticoagulants and immunosuppressants (eg, steroids, azathioprine, and N-acetylcysteine), due to their proven ineffectiveness and harm to patients with IPF.
Since the FDA’s approval of pirfenidone and nintedanib, the ATS has made recommendations for their use in patients with IPF. Despite mixed results in clinical trials, both drugs have demonstrated the ability to slow the decline in FVC over time, with relatively benign adverse effects. It is difficult to compare pirfenidone and nintedanib, or to recommend use of one drug over the other. However, it is promising that patients with this routinely fatal disease now have treatment options that can potentially modulate their disease progression.
1. Kim DS, Collard HR, King TE Jr. Classification and natural history of the idiopathic interstitial pneumonias. Proc Am Thorac Soc. 2006;3(4):285-292.
2. Frankel SK, Schwarz MI. Update in idiopathic pulmonary fibrosis. Curr Opin Pulm Med. 2009;15(5):463-469.
3. Olson AL, Swigris JJ, Lezotte DC, et al. Mortality from pulmonary fibrosis increased in the United States from 1992 to 2003. Am J Respir Crit Care Med. 2007;176(3):277-284.
4. Chapman JT. Interstitial lung disease. Cleveland Clinic. August 2010. www.clevelandclinicmeded.com/medical pubs/diseasemanagement/pulmonary/interstitial-lung-disease. Accessed March 12, 2018.
5. Cleveland Clinic. Nonspecific interstitial pneumonia. January 16, 2015. https://my.clevelandclinic.org/health/articles/nonspecific-interstitial-pneumonia. Accessed March 12, 2018.
6. Skandhan AKP, Weerakkody Y. Non-specific interstitial pneumonia. Radiopaedia. https://radiopaedia.org/articles/non-specific-interstitial-pneumonia-1. Accessed March 12, 2018.
7. Tatco V, Weerakkody Y. Lymphocytic interstitial pneumonitis. Radiopaedia. https://radiopaedia.org/articles/lymphocytic-interstitial-pneumonitis-1. Accessed March 12, 2018.
8. King TE Jr, Flaherty KR, Hollingsworth H. Cryptogenic organizing pneumonia. UpToDate. www.uptodate.com/contents/cryptogenic-organizing-pneumonia#H12. Accessed March 12, 2018.
9. Patel NM, Lederer DJ, Borczuk AC, Kawut SM. Pulmonary hypertension in idiopathic pulmonary fibrosis. Chest. 2007; 132(3):998-1006.
10. Lee J. Overview of idiopathic interstitial pneumonias. April 2016. www.merckmanuals.com/professional/pulmonary-disorders/interstitial-lung-diseases/overview-of-idiopathic-interstitial-pneumonias. Accessed March 12, 2018.
11. Lynch DA, Sverzellati N, Travis WD, et al. Diagnostic criteria for idiopathic pulmonary fibrosis: a Fleischner Society White Paper. Lancet Respir Med. 2018;6(2):138-153.
12. Martinez FJ, Flaherty K. Pulmonary function testing in idiopathic interstitial pneumonias. Proc Am Thorac Soc. 2006; 3(4):315-321.
13. Raghu G, Rochwerg B, Zhang Y, et al; American Thoracic Society; European Respiratory Society; Japanese Respiratory Society; Latin American Thoracic Association. An official ATS/ERS/JRS/ALAT clinical practice guideline: treatment of idiopathic pulmonary fibrosis. An update of the 2011 Clinical Practice Guideline. Am J Respir Crit Care Med. 2015; 192(2):e3-e19.
14. Chowdhury BA; FDA. Two FDA drug approvals for idiopathic pulmonary fibrosis (IPF). October 15, 2014. https://blogs.fda.gov/fdavoice/index.php/2014/10/two-fda-drug-approvals-for-idiopathic-pulmonary-fibrosis-ipf/. Accessed March 12, 2018.
15. Nakayama S, Mukae H, Sakamoto N, et al. Pirfenidone inhibits the expression of HSP47 in TGF-beta1-stimulated human lung fibroblasts. Life Sci. 2008; 82(3-4):210-217.
16. Noble PW, Albera C, Bradford WZ, et al; CAPACITY Study Group. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomized trials. Lancet. 2011;377: 1760-1769.
17. King TE Jr, Bradford WZ, Castro-Bernardini S, et al; ASCEND Study Group. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22): 2083-2092.
18. Esbriet [package insert]. South San Francisco, CA: Genentech, Inc; 2016.
19. Hostettler KE, Zhong J, Papakonstantinou E, et al. Anti-fibrotic effects of nintedanib in lung fibroblasts derived from patients with idiopathic pulmonary fibrosis. Respir Res. 2014;15(1):157.
20. Richeldi L, du Bois RM, Raghu G, et al; INPULSIS Trial Investigators. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2071-2082.
21. OFEV [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; 2018.
IN THIS ARTICLE
- Confirming the diagnosis
- Pirfenidone treatment
- Nintedanib treatment
A 64-year-old man has a one-year history of dyspnea on exertion and a nonproductive cough. His symptoms are gradually worsening and increasingly bothersome to him.
His medical history includes mild seasonal allergies and GERD, which is well-controlled by oral antihistamines and proton pump inhibitors. He has spent the past 30 years working a desk job as an accountant. He denies a history of smoking, exposure to secondhand smoke, and initiation of new medication.
He admits to increased fatigue, but denies fever, chills, lymphadenopathy, weight change, chest pain, wheezing, abdominal pain, diarrhea, vomiting, claudication, and swelling in the extremities. The rest of the review of systems is negative.
Lab results—complete blood count, comprehensive metabolic panel, TSH, antinuclear antibodies, erythrocyte sedimentation rate, and C-reactive protein—are within normal limits. Spirometry shows very mild restriction. A chest x-ray is abnormal but nonspecific, showing peripheral opacities. An ECG shows normal sinus rhythm.
The patient is given a trial of an inhaled steroid, which yields no improvement. Six months later, the patient is seen by a pulmonologist. Idiopathic pulmonary fibrosis (IPF) is diagnosed based on high-resolution CT (HRCT) and lung biopsy results.
IPF is a chronic, progressive, fibrosing interstitial disease that is limited to lung tissue. It most commonly manifests in older adults with vague symptoms of dyspnea on exertion and nonproductive cough, but symptoms can also include fatigue, muscle and joint aches, clubbing of the fingernails, and weight loss.1 The average life expectancy following diagnosis of IPF is two to five years, and the mortality rate is estimated at 64.3 per million men and 58.4 per million women per year.2,3
Continue to: DIAGNOSIS
DIAGNOSIS
IPF belongs in the general class of idiopathic interstitial pneumonias (IIPs), which are characterized by varying degrees of inflammation and fibrosis of lung interstitium.4 All subtypes of IIPs cause dyspnea and diffuse abnormalities on HRCT, and all vary from each other histologically. Table 1 outlines the key features of each.5-8

Because of its vague symptomology and the extensive workup needed to rule out other diseases, patients with IPF often have symptoms for one to two years before a diagnosis is made.1 Physical exam may reveal fine inspiratory rales in both lung bases and digital clubbing; eventual signs of pulmonary hypertension and right-sided heart failure may be appreciated.1,9
There are no specific diagnostic laboratory tests to confirm IPF; however, baseline labwork (as outlined in the case presentation) is typically ordered to rule out infection, thyroid disease, or connective tissue disease.10 Many patients are referred to a cardiologist before being seen by a pulmonologist; cardiac stress testing may be done, and an echocardiogram may be performed to rule out heart failure.
Diagnostic testing may include pulmonary function testing, HRCT of the chest, and lung biopsy.10 Tissue samples from patients with IPF reveal different stages of disease, including dense fibrosis with honeycombing, subpleural or paraseptal distribution, fibroblast foci, and normal tissue.11 Pulmonary function test results will show a restrictive pattern. Both forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) will be reduced, and the FEV1/FVC ratio preserved. Due to decreased functional lung volume, diffusing capacity of the lung for carbon monoxide (DLCO) will also be reduced.4,12

The differential is broad and includes allergic asthma, bronchitis, COPD, lung cancer, hypersensitivity pneumonitis, asbestosis, or pulmonary embolism.
Continue to: TREATMENT HISTORY
TREATMENT HISTORY
IPF has a long history of tried and failed treatment options. The American Thoracic Society (ATS), in concert with other professional organizations, has published comprehensive guidelines and recommendations pertaining to the use of pharmacologic medications to control disease progression. Warfarin and other anticoagulants have been studied, based on the observation that a procoagulant state promotes fibrotic changes in the lung tissue.13 However, anticoagulant use is not recommended in patients with IPF due to lack of efficacy and high potential for harm.13
Immunosuppressants have also been in the spotlight as possible treatment for IPF, but a clinical study investigating the efficacy of a three-drug regimen including prednisone, azathioprine, and N-acetylcysteine was stopped early due to increased risk for harm. Endothelin antagonists and potent tyrosine kinase inhibitors are also not recommended in the most recent edition of IPF guidelines, as they lack benefit.13
In fact, prior to the 2015 edition of the guidelines, no single medication was routinely recommended for patients with IPF. But this is now changing, following the 2014 FDA approval of two new drugs, nintedanib and pirfenidone, designed specifically to treat IPF.14 These drugs have shown promise in clinical trials (results of which are summarized in Table 2).

Continue to: NEW PHARMACOLOGIC OPTIONS
NEW PHARMACOLOGIC OPTIONS
Pirfenidone
In 2008, a study was conducted in Japan to determine the mechanism of action of pirfenidone.15 Through in vitro studies of healthy adult lung fibroblasts with added pro-fibrotic factor and transforming growth factor (TGF-ß 1), the researchers found that pirfenidone was effective at decreasing the production of a collagen-binding protein called HSP47. This protein is ubiquitous in fibrotic tissue. The study also showed that pirfenidone decreased the production of collagen type 1, which, when uninhibited, increases fibrosis.15
CAPACITY trials. In the CAPACITY trials, two phase 3 multinational studies conducted from 2006 to 2008, patients were given either pirfenidone or placebo.16 In the first study arm, patients were assigned to pirfenidone 2,403 mg/d (n = 174), pirfenidone 1,197 mg/d (n = 87), or placebo (n = 174). In the second study arm, 171 patients received pirfenidone 2,403 mg/d and 173 patients received placebo. Endpoints were measured at baseline and up to week 72.
The first study arm found that the mean rate of decline of FVC—the primary endpoint—was 4.4% less in the treatment group than in the placebo group (p = 0.001), and there was a 36% decrease in risk for death or disease progression in the treatment group (HR, 0.64; p, 0.023). (Endpoints were defined as: time to confirmed > 10% decline in percentage predicted FVC, > 15% decline in percentage predicted DLCO, or death.) The researchers found no clinically significant change in the six-minute walk test—a secondary endpoint of the study.16
The second study arm, however, found no statistically significant change in FVC between the treatment and placebo groups (with a 0.6% smaller decrease in FVC in the pirfenidone group), nor did they see a difference in progression-free survival. However, there was a significant change in the six-minute walk test between the treatment and placebo groups (p = 0.0009). Throughout the study, the most common adverse effects included nausea (36%), rash (32%), and dyspepsia (19%).16
ASCEND trial. The 2014 Assessment of Pirfenidone to Confirm Efficacy and Safety in Idiopathic Pulmonary Fibrosis (ASCEND) trial was a phase 3, multinational, randomized, double-blind, placebo-controlled study of the use of pirfenidone 2,403 mg/d.17 The study was conducted from 2012 to 2013. Of the total number of patients (N = 522), half received pirfenidone and half received placebo. After 52 weeks of treatment (the end of the study), the researchers found a smaller decline in FVC—the primary endpoint—in the treatment group compared to placebo (mean decline, 235 mL vs 428 mL, respectively [p < 0.001]). Regarding the six-minute walk test, the investigators found that 25.9% of the treatment group exhibited a decrease of ≥ 50 meters, compared to 35.7% of the placebo group (p = 0.04). (Progression-free survival was defined as a confirmed ≥ 10% decrease in predicted FVC, a confirmed decrease of 50 meters in the six-minute walk test, or death.)
The pirfenidone group in the ASCEND trial showed a 43% reduced risk for death or disease progression (HR, 0.57; p, < 0.001).16,17 All-cause mortality was lower in the pirfenidone group (4%) than in the placebo group (7.2%), but this was not statistically significant. Deaths from IPF in the pirfenidone group totaled three patients (1.1%) versus seven patients (2.5%) in the placebo group; this was also not statistically significant. The most common adverse effects seen during the study were nausea (36%), rash (28.1%), and headache (25.9%).17
Recommendations for use. Liver function testing should be performed at baseline, monthly for six months, and every three months afterward, as elevations in liver enzymes have been observed.18 Pirfenidone is a CYP1A2 substrate; moderate-to-strong CYP1A2 inhibitors should therefore be discontinued prior to initiation, as they are likely to decrease exposure and efficacy of pirfenidone. There are currently no black box warnings.18
Continue to: Nintedanib
Nintedanib
Hostettler et al studied lung samples from patients with IPF to determine the mechanism of action of nintedanib.19 Evaluation of fibroblasts derived from IPF samples revealed that they contained higher levels of platelet-derived growth factor (PDGF) than did nonfibrotic control cells. They also found that nintedanib, a tyrosine kinase inhibitor, significantly inhibited the phosphorylation of fibrotic-inducing growth factors—PDGF as well as vascular endothelial growth factor (VEGF).
INPULSIS trials. A phase 3 replicate of randomized, double-blind, multinational studies, the INPULSIS trials were performed between 2011 and 2012.20 Two study arms were used to evaluate a total of 638 patients who received nintedanib 150 mg bid for 52 weeks. The primary endpoint was annual rate of decline of FVC.
The researchers also evaluated efficacy through two other endpoints: patient-reported quality of life and symptoms via the St. George’s Respiratory Questionnaire (SGRQ) and evaluation of time to acute exacerbation. The latter was defined as worsening or new dyspnea, new diffuse pulmonary infiltrates visualized on chest radiography and/or HRCT, or the development of parenchymal abnormalities with no pneumothorax or pleural effusion since the preceding visit; and exclusion of any known causes of acute worsening, including infection, heart failure, pulmonary embolism, and any identifiable cause of acute lung injury.20
INPULSIS 1 (first arm) included 309 patients in the treatment group. Results showed an adjusted annual rate of decline in FVC of 114.7 mL/year, versus 239.9 mL/year in the placebo group (p < 0.001). In the treatment group, 52.8% exhibited ≤ 5% decline in FVC, compared to 38.2% in the placebo group (p = 0.001). No significant between-group differences were found in SGRQ score or time to acute exacerbation.20
INPULSIS 2 had 329 patients receiving nintedanib. An annual rate of decline in FVC of 113.6 mL/year from baseline was observed in the treatment group, compared to 207.3 mL/year in the placebo group (p < 0.001). In the treatment group, 53.2% showed ≤ 5% decline in FVC, versus 39.3% in the placebo group (p = 0.001). There was also a significantly smaller increase in total SGRQ score (meaning, less deterioration in quality of life) in the nintedanib group versus the placebo group (p = 0.02). A statistically significant increase in time to first acute exacerbation was observed in the nintedanib group (p = 0.005).20
There was no significant difference between groups in death from any cause, death from respiratory causation, or death that occurred between randomization and 28 days post treatment. The most common adverse effects seen throughout the two trials included diarrhea (trial 1, 61.5%; trial 2, 63.2%), nausea (trial 1, 22.7%; trial 2, 26.1%), and nasopharyngitis (trial 1, 12.6%; trial 2, 14.6%).20
Recommendations for use. Liver function testing should be performed at baseline, at regular intervals during the first three months, then periodically thereafter; patients in the treatment group of both INPULSIS trials had elevated liver enzymes, and cases of drug-induced liver injury have been observed with use of nintedanib.21 This medication may increase risk for bleeding due to its mechanism of action (VEGFR inhibition). Coadministration with CYP3A4 inhibitors may increase concentration of nintedanib; therefore, close monitoring is recommended. Avoid coadministration with CYP3A4 inducers, as this may decrease concentration of nintedanib by 50%. There are currently no black box warnings.21
Continue to: Patient monitoring
Patient monitoring
The ATS recommends measuring FVC and DLCO every three to six months, or sooner if clinically indicated.13 Pulse oximetry should be measured at rest and on exertion in all patients, regardless of symptoms, to assure proper saturation and identify the need for supplemental oxygen; this should also be done every three to six months.
The ATS recommends prompt detection and treatment of comorbidities such as pulmonary hypertension, emphysema, airflow obstruction, GERD, sleep apnea, and coronary artery disease.13 These recommendations are based on the organization’s 2015 guidelines.
OUTCOME FOR THE CASE PATIENT
The patient was started on pirfenidone (2,403 mg/d). He is continuing treatment and showing improvements in quality of life and slowed deterioration of lung function.
CONCLUSION
IPF causes progressive fibrosis of lung interstitium. The etiology is unknown, the symptoms and signs are vague, and mean life expectancy following diagnosis is two to five years. The most recent IPF guidelines recommend avoiding use of anticoagulants and immunosuppressants (eg, steroids, azathioprine, and N-acetylcysteine), due to their proven ineffectiveness and harm to patients with IPF.
Since the FDA’s approval of pirfenidone and nintedanib, the ATS has made recommendations for their use in patients with IPF. Despite mixed results in clinical trials, both drugs have demonstrated the ability to slow the decline in FVC over time, with relatively benign adverse effects. It is difficult to compare pirfenidone and nintedanib, or to recommend use of one drug over the other. However, it is promising that patients with this routinely fatal disease now have treatment options that can potentially modulate their disease progression.
IN THIS ARTICLE
- Confirming the diagnosis
- Pirfenidone treatment
- Nintedanib treatment
A 64-year-old man has a one-year history of dyspnea on exertion and a nonproductive cough. His symptoms are gradually worsening and increasingly bothersome to him.
His medical history includes mild seasonal allergies and GERD, which is well-controlled by oral antihistamines and proton pump inhibitors. He has spent the past 30 years working a desk job as an accountant. He denies a history of smoking, exposure to secondhand smoke, and initiation of new medication.
He admits to increased fatigue, but denies fever, chills, lymphadenopathy, weight change, chest pain, wheezing, abdominal pain, diarrhea, vomiting, claudication, and swelling in the extremities. The rest of the review of systems is negative.
Lab results—complete blood count, comprehensive metabolic panel, TSH, antinuclear antibodies, erythrocyte sedimentation rate, and C-reactive protein—are within normal limits. Spirometry shows very mild restriction. A chest x-ray is abnormal but nonspecific, showing peripheral opacities. An ECG shows normal sinus rhythm.
The patient is given a trial of an inhaled steroid, which yields no improvement. Six months later, the patient is seen by a pulmonologist. Idiopathic pulmonary fibrosis (IPF) is diagnosed based on high-resolution CT (HRCT) and lung biopsy results.
IPF is a chronic, progressive, fibrosing interstitial disease that is limited to lung tissue. It most commonly manifests in older adults with vague symptoms of dyspnea on exertion and nonproductive cough, but symptoms can also include fatigue, muscle and joint aches, clubbing of the fingernails, and weight loss.1 The average life expectancy following diagnosis of IPF is two to five years, and the mortality rate is estimated at 64.3 per million men and 58.4 per million women per year.2,3
Continue to: DIAGNOSIS
DIAGNOSIS
IPF belongs in the general class of idiopathic interstitial pneumonias (IIPs), which are characterized by varying degrees of inflammation and fibrosis of lung interstitium.4 All subtypes of IIPs cause dyspnea and diffuse abnormalities on HRCT, and all vary from each other histologically. Table 1 outlines the key features of each.5-8

Because of its vague symptomology and the extensive workup needed to rule out other diseases, patients with IPF often have symptoms for one to two years before a diagnosis is made.1 Physical exam may reveal fine inspiratory rales in both lung bases and digital clubbing; eventual signs of pulmonary hypertension and right-sided heart failure may be appreciated.1,9
There are no specific diagnostic laboratory tests to confirm IPF; however, baseline labwork (as outlined in the case presentation) is typically ordered to rule out infection, thyroid disease, or connective tissue disease.10 Many patients are referred to a cardiologist before being seen by a pulmonologist; cardiac stress testing may be done, and an echocardiogram may be performed to rule out heart failure.
Diagnostic testing may include pulmonary function testing, HRCT of the chest, and lung biopsy.10 Tissue samples from patients with IPF reveal different stages of disease, including dense fibrosis with honeycombing, subpleural or paraseptal distribution, fibroblast foci, and normal tissue.11 Pulmonary function test results will show a restrictive pattern. Both forced expiratory volume in one second (FEV1) and forced vital capacity (FVC) will be reduced, and the FEV1/FVC ratio preserved. Due to decreased functional lung volume, diffusing capacity of the lung for carbon monoxide (DLCO) will also be reduced.4,12

The differential is broad and includes allergic asthma, bronchitis, COPD, lung cancer, hypersensitivity pneumonitis, asbestosis, or pulmonary embolism.
Continue to: TREATMENT HISTORY
TREATMENT HISTORY
IPF has a long history of tried and failed treatment options. The American Thoracic Society (ATS), in concert with other professional organizations, has published comprehensive guidelines and recommendations pertaining to the use of pharmacologic medications to control disease progression. Warfarin and other anticoagulants have been studied, based on the observation that a procoagulant state promotes fibrotic changes in the lung tissue.13 However, anticoagulant use is not recommended in patients with IPF due to lack of efficacy and high potential for harm.13
Immunosuppressants have also been in the spotlight as possible treatment for IPF, but a clinical study investigating the efficacy of a three-drug regimen including prednisone, azathioprine, and N-acetylcysteine was stopped early due to increased risk for harm. Endothelin antagonists and potent tyrosine kinase inhibitors are also not recommended in the most recent edition of IPF guidelines, as they lack benefit.13
In fact, prior to the 2015 edition of the guidelines, no single medication was routinely recommended for patients with IPF. But this is now changing, following the 2014 FDA approval of two new drugs, nintedanib and pirfenidone, designed specifically to treat IPF.14 These drugs have shown promise in clinical trials (results of which are summarized in Table 2).

Continue to: NEW PHARMACOLOGIC OPTIONS
NEW PHARMACOLOGIC OPTIONS
Pirfenidone
In 2008, a study was conducted in Japan to determine the mechanism of action of pirfenidone.15 Through in vitro studies of healthy adult lung fibroblasts with added pro-fibrotic factor and transforming growth factor (TGF-ß 1), the researchers found that pirfenidone was effective at decreasing the production of a collagen-binding protein called HSP47. This protein is ubiquitous in fibrotic tissue. The study also showed that pirfenidone decreased the production of collagen type 1, which, when uninhibited, increases fibrosis.15
CAPACITY trials. In the CAPACITY trials, two phase 3 multinational studies conducted from 2006 to 2008, patients were given either pirfenidone or placebo.16 In the first study arm, patients were assigned to pirfenidone 2,403 mg/d (n = 174), pirfenidone 1,197 mg/d (n = 87), or placebo (n = 174). In the second study arm, 171 patients received pirfenidone 2,403 mg/d and 173 patients received placebo. Endpoints were measured at baseline and up to week 72.
The first study arm found that the mean rate of decline of FVC—the primary endpoint—was 4.4% less in the treatment group than in the placebo group (p = 0.001), and there was a 36% decrease in risk for death or disease progression in the treatment group (HR, 0.64; p, 0.023). (Endpoints were defined as: time to confirmed > 10% decline in percentage predicted FVC, > 15% decline in percentage predicted DLCO, or death.) The researchers found no clinically significant change in the six-minute walk test—a secondary endpoint of the study.16
The second study arm, however, found no statistically significant change in FVC between the treatment and placebo groups (with a 0.6% smaller decrease in FVC in the pirfenidone group), nor did they see a difference in progression-free survival. However, there was a significant change in the six-minute walk test between the treatment and placebo groups (p = 0.0009). Throughout the study, the most common adverse effects included nausea (36%), rash (32%), and dyspepsia (19%).16
ASCEND trial. The 2014 Assessment of Pirfenidone to Confirm Efficacy and Safety in Idiopathic Pulmonary Fibrosis (ASCEND) trial was a phase 3, multinational, randomized, double-blind, placebo-controlled study of the use of pirfenidone 2,403 mg/d.17 The study was conducted from 2012 to 2013. Of the total number of patients (N = 522), half received pirfenidone and half received placebo. After 52 weeks of treatment (the end of the study), the researchers found a smaller decline in FVC—the primary endpoint—in the treatment group compared to placebo (mean decline, 235 mL vs 428 mL, respectively [p < 0.001]). Regarding the six-minute walk test, the investigators found that 25.9% of the treatment group exhibited a decrease of ≥ 50 meters, compared to 35.7% of the placebo group (p = 0.04). (Progression-free survival was defined as a confirmed ≥ 10% decrease in predicted FVC, a confirmed decrease of 50 meters in the six-minute walk test, or death.)
The pirfenidone group in the ASCEND trial showed a 43% reduced risk for death or disease progression (HR, 0.57; p, < 0.001).16,17 All-cause mortality was lower in the pirfenidone group (4%) than in the placebo group (7.2%), but this was not statistically significant. Deaths from IPF in the pirfenidone group totaled three patients (1.1%) versus seven patients (2.5%) in the placebo group; this was also not statistically significant. The most common adverse effects seen during the study were nausea (36%), rash (28.1%), and headache (25.9%).17
Recommendations for use. Liver function testing should be performed at baseline, monthly for six months, and every three months afterward, as elevations in liver enzymes have been observed.18 Pirfenidone is a CYP1A2 substrate; moderate-to-strong CYP1A2 inhibitors should therefore be discontinued prior to initiation, as they are likely to decrease exposure and efficacy of pirfenidone. There are currently no black box warnings.18
Continue to: Nintedanib
Nintedanib
Hostettler et al studied lung samples from patients with IPF to determine the mechanism of action of nintedanib.19 Evaluation of fibroblasts derived from IPF samples revealed that they contained higher levels of platelet-derived growth factor (PDGF) than did nonfibrotic control cells. They also found that nintedanib, a tyrosine kinase inhibitor, significantly inhibited the phosphorylation of fibrotic-inducing growth factors—PDGF as well as vascular endothelial growth factor (VEGF).
INPULSIS trials. A phase 3 replicate of randomized, double-blind, multinational studies, the INPULSIS trials were performed between 2011 and 2012.20 Two study arms were used to evaluate a total of 638 patients who received nintedanib 150 mg bid for 52 weeks. The primary endpoint was annual rate of decline of FVC.
The researchers also evaluated efficacy through two other endpoints: patient-reported quality of life and symptoms via the St. George’s Respiratory Questionnaire (SGRQ) and evaluation of time to acute exacerbation. The latter was defined as worsening or new dyspnea, new diffuse pulmonary infiltrates visualized on chest radiography and/or HRCT, or the development of parenchymal abnormalities with no pneumothorax or pleural effusion since the preceding visit; and exclusion of any known causes of acute worsening, including infection, heart failure, pulmonary embolism, and any identifiable cause of acute lung injury.20
INPULSIS 1 (first arm) included 309 patients in the treatment group. Results showed an adjusted annual rate of decline in FVC of 114.7 mL/year, versus 239.9 mL/year in the placebo group (p < 0.001). In the treatment group, 52.8% exhibited ≤ 5% decline in FVC, compared to 38.2% in the placebo group (p = 0.001). No significant between-group differences were found in SGRQ score or time to acute exacerbation.20
INPULSIS 2 had 329 patients receiving nintedanib. An annual rate of decline in FVC of 113.6 mL/year from baseline was observed in the treatment group, compared to 207.3 mL/year in the placebo group (p < 0.001). In the treatment group, 53.2% showed ≤ 5% decline in FVC, versus 39.3% in the placebo group (p = 0.001). There was also a significantly smaller increase in total SGRQ score (meaning, less deterioration in quality of life) in the nintedanib group versus the placebo group (p = 0.02). A statistically significant increase in time to first acute exacerbation was observed in the nintedanib group (p = 0.005).20
There was no significant difference between groups in death from any cause, death from respiratory causation, or death that occurred between randomization and 28 days post treatment. The most common adverse effects seen throughout the two trials included diarrhea (trial 1, 61.5%; trial 2, 63.2%), nausea (trial 1, 22.7%; trial 2, 26.1%), and nasopharyngitis (trial 1, 12.6%; trial 2, 14.6%).20
Recommendations for use. Liver function testing should be performed at baseline, at regular intervals during the first three months, then periodically thereafter; patients in the treatment group of both INPULSIS trials had elevated liver enzymes, and cases of drug-induced liver injury have been observed with use of nintedanib.21 This medication may increase risk for bleeding due to its mechanism of action (VEGFR inhibition). Coadministration with CYP3A4 inhibitors may increase concentration of nintedanib; therefore, close monitoring is recommended. Avoid coadministration with CYP3A4 inducers, as this may decrease concentration of nintedanib by 50%. There are currently no black box warnings.21
Continue to: Patient monitoring
Patient monitoring
The ATS recommends measuring FVC and DLCO every three to six months, or sooner if clinically indicated.13 Pulse oximetry should be measured at rest and on exertion in all patients, regardless of symptoms, to assure proper saturation and identify the need for supplemental oxygen; this should also be done every three to six months.
The ATS recommends prompt detection and treatment of comorbidities such as pulmonary hypertension, emphysema, airflow obstruction, GERD, sleep apnea, and coronary artery disease.13 These recommendations are based on the organization’s 2015 guidelines.
OUTCOME FOR THE CASE PATIENT
The patient was started on pirfenidone (2,403 mg/d). He is continuing treatment and showing improvements in quality of life and slowed deterioration of lung function.
CONCLUSION
IPF causes progressive fibrosis of lung interstitium. The etiology is unknown, the symptoms and signs are vague, and mean life expectancy following diagnosis is two to five years. The most recent IPF guidelines recommend avoiding use of anticoagulants and immunosuppressants (eg, steroids, azathioprine, and N-acetylcysteine), due to their proven ineffectiveness and harm to patients with IPF.
Since the FDA’s approval of pirfenidone and nintedanib, the ATS has made recommendations for their use in patients with IPF. Despite mixed results in clinical trials, both drugs have demonstrated the ability to slow the decline in FVC over time, with relatively benign adverse effects. It is difficult to compare pirfenidone and nintedanib, or to recommend use of one drug over the other. However, it is promising that patients with this routinely fatal disease now have treatment options that can potentially modulate their disease progression.
1. Kim DS, Collard HR, King TE Jr. Classification and natural history of the idiopathic interstitial pneumonias. Proc Am Thorac Soc. 2006;3(4):285-292.
2. Frankel SK, Schwarz MI. Update in idiopathic pulmonary fibrosis. Curr Opin Pulm Med. 2009;15(5):463-469.
3. Olson AL, Swigris JJ, Lezotte DC, et al. Mortality from pulmonary fibrosis increased in the United States from 1992 to 2003. Am J Respir Crit Care Med. 2007;176(3):277-284.
4. Chapman JT. Interstitial lung disease. Cleveland Clinic. August 2010. www.clevelandclinicmeded.com/medical pubs/diseasemanagement/pulmonary/interstitial-lung-disease. Accessed March 12, 2018.
5. Cleveland Clinic. Nonspecific interstitial pneumonia. January 16, 2015. https://my.clevelandclinic.org/health/articles/nonspecific-interstitial-pneumonia. Accessed March 12, 2018.
6. Skandhan AKP, Weerakkody Y. Non-specific interstitial pneumonia. Radiopaedia. https://radiopaedia.org/articles/non-specific-interstitial-pneumonia-1. Accessed March 12, 2018.
7. Tatco V, Weerakkody Y. Lymphocytic interstitial pneumonitis. Radiopaedia. https://radiopaedia.org/articles/lymphocytic-interstitial-pneumonitis-1. Accessed March 12, 2018.
8. King TE Jr, Flaherty KR, Hollingsworth H. Cryptogenic organizing pneumonia. UpToDate. www.uptodate.com/contents/cryptogenic-organizing-pneumonia#H12. Accessed March 12, 2018.
9. Patel NM, Lederer DJ, Borczuk AC, Kawut SM. Pulmonary hypertension in idiopathic pulmonary fibrosis. Chest. 2007; 132(3):998-1006.
10. Lee J. Overview of idiopathic interstitial pneumonias. April 2016. www.merckmanuals.com/professional/pulmonary-disorders/interstitial-lung-diseases/overview-of-idiopathic-interstitial-pneumonias. Accessed March 12, 2018.
11. Lynch DA, Sverzellati N, Travis WD, et al. Diagnostic criteria for idiopathic pulmonary fibrosis: a Fleischner Society White Paper. Lancet Respir Med. 2018;6(2):138-153.
12. Martinez FJ, Flaherty K. Pulmonary function testing in idiopathic interstitial pneumonias. Proc Am Thorac Soc. 2006; 3(4):315-321.
13. Raghu G, Rochwerg B, Zhang Y, et al; American Thoracic Society; European Respiratory Society; Japanese Respiratory Society; Latin American Thoracic Association. An official ATS/ERS/JRS/ALAT clinical practice guideline: treatment of idiopathic pulmonary fibrosis. An update of the 2011 Clinical Practice Guideline. Am J Respir Crit Care Med. 2015; 192(2):e3-e19.
14. Chowdhury BA; FDA. Two FDA drug approvals for idiopathic pulmonary fibrosis (IPF). October 15, 2014. https://blogs.fda.gov/fdavoice/index.php/2014/10/two-fda-drug-approvals-for-idiopathic-pulmonary-fibrosis-ipf/. Accessed March 12, 2018.
15. Nakayama S, Mukae H, Sakamoto N, et al. Pirfenidone inhibits the expression of HSP47 in TGF-beta1-stimulated human lung fibroblasts. Life Sci. 2008; 82(3-4):210-217.
16. Noble PW, Albera C, Bradford WZ, et al; CAPACITY Study Group. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomized trials. Lancet. 2011;377: 1760-1769.
17. King TE Jr, Bradford WZ, Castro-Bernardini S, et al; ASCEND Study Group. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22): 2083-2092.
18. Esbriet [package insert]. South San Francisco, CA: Genentech, Inc; 2016.
19. Hostettler KE, Zhong J, Papakonstantinou E, et al. Anti-fibrotic effects of nintedanib in lung fibroblasts derived from patients with idiopathic pulmonary fibrosis. Respir Res. 2014;15(1):157.
20. Richeldi L, du Bois RM, Raghu G, et al; INPULSIS Trial Investigators. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2071-2082.
21. OFEV [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; 2018.
1. Kim DS, Collard HR, King TE Jr. Classification and natural history of the idiopathic interstitial pneumonias. Proc Am Thorac Soc. 2006;3(4):285-292.
2. Frankel SK, Schwarz MI. Update in idiopathic pulmonary fibrosis. Curr Opin Pulm Med. 2009;15(5):463-469.
3. Olson AL, Swigris JJ, Lezotte DC, et al. Mortality from pulmonary fibrosis increased in the United States from 1992 to 2003. Am J Respir Crit Care Med. 2007;176(3):277-284.
4. Chapman JT. Interstitial lung disease. Cleveland Clinic. August 2010. www.clevelandclinicmeded.com/medical pubs/diseasemanagement/pulmonary/interstitial-lung-disease. Accessed March 12, 2018.
5. Cleveland Clinic. Nonspecific interstitial pneumonia. January 16, 2015. https://my.clevelandclinic.org/health/articles/nonspecific-interstitial-pneumonia. Accessed March 12, 2018.
6. Skandhan AKP, Weerakkody Y. Non-specific interstitial pneumonia. Radiopaedia. https://radiopaedia.org/articles/non-specific-interstitial-pneumonia-1. Accessed March 12, 2018.
7. Tatco V, Weerakkody Y. Lymphocytic interstitial pneumonitis. Radiopaedia. https://radiopaedia.org/articles/lymphocytic-interstitial-pneumonitis-1. Accessed March 12, 2018.
8. King TE Jr, Flaherty KR, Hollingsworth H. Cryptogenic organizing pneumonia. UpToDate. www.uptodate.com/contents/cryptogenic-organizing-pneumonia#H12. Accessed March 12, 2018.
9. Patel NM, Lederer DJ, Borczuk AC, Kawut SM. Pulmonary hypertension in idiopathic pulmonary fibrosis. Chest. 2007; 132(3):998-1006.
10. Lee J. Overview of idiopathic interstitial pneumonias. April 2016. www.merckmanuals.com/professional/pulmonary-disorders/interstitial-lung-diseases/overview-of-idiopathic-interstitial-pneumonias. Accessed March 12, 2018.
11. Lynch DA, Sverzellati N, Travis WD, et al. Diagnostic criteria for idiopathic pulmonary fibrosis: a Fleischner Society White Paper. Lancet Respir Med. 2018;6(2):138-153.
12. Martinez FJ, Flaherty K. Pulmonary function testing in idiopathic interstitial pneumonias. Proc Am Thorac Soc. 2006; 3(4):315-321.
13. Raghu G, Rochwerg B, Zhang Y, et al; American Thoracic Society; European Respiratory Society; Japanese Respiratory Society; Latin American Thoracic Association. An official ATS/ERS/JRS/ALAT clinical practice guideline: treatment of idiopathic pulmonary fibrosis. An update of the 2011 Clinical Practice Guideline. Am J Respir Crit Care Med. 2015; 192(2):e3-e19.
14. Chowdhury BA; FDA. Two FDA drug approvals for idiopathic pulmonary fibrosis (IPF). October 15, 2014. https://blogs.fda.gov/fdavoice/index.php/2014/10/two-fda-drug-approvals-for-idiopathic-pulmonary-fibrosis-ipf/. Accessed March 12, 2018.
15. Nakayama S, Mukae H, Sakamoto N, et al. Pirfenidone inhibits the expression of HSP47 in TGF-beta1-stimulated human lung fibroblasts. Life Sci. 2008; 82(3-4):210-217.
16. Noble PW, Albera C, Bradford WZ, et al; CAPACITY Study Group. Pirfenidone in patients with idiopathic pulmonary fibrosis (CAPACITY): two randomized trials. Lancet. 2011;377: 1760-1769.
17. King TE Jr, Bradford WZ, Castro-Bernardini S, et al; ASCEND Study Group. A phase 3 trial of pirfenidone in patients with idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22): 2083-2092.
18. Esbriet [package insert]. South San Francisco, CA: Genentech, Inc; 2016.
19. Hostettler KE, Zhong J, Papakonstantinou E, et al. Anti-fibrotic effects of nintedanib in lung fibroblasts derived from patients with idiopathic pulmonary fibrosis. Respir Res. 2014;15(1):157.
20. Richeldi L, du Bois RM, Raghu G, et al; INPULSIS Trial Investigators. Efficacy and safety of nintedanib in idiopathic pulmonary fibrosis. N Engl J Med. 2014;370(22):2071-2082.
21. OFEV [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals, Inc; 2018.
Website improvements
Nowadays, almost half of all Americans (and nearly all millennials) seek out doctors online. But your practice website should be doing a lot more than simply describing your practice. How many visitors to your site actually schedule an appointment? With a few relatively simple but important modifications, you can convert casual website viewers to patients.
Start with a good title, one that not only describes your practice but also anticipates how prospective patients will search for you – usually by specialty plus geographic location. My site’s title, for example, is “Belleville Dermatology Center,” so when someone searches for a dermatologist near Belleville, N.J., my site will invariably rank near the top of their search results.
Follow with an understandable URL. Search engines use URLs to determine how well your site, or a portion of it, matches search criteria. URLs also need to make sense to searchers, especially when they link specific areas of expertise (more on that in a minute). For example, a URL like “bellevilledermatology[dot]com/?p=89021” is meaningless to anyone except programmers; but “bellevilledermatology[dot]com/psoriasistreatments” obviously leads to a page about psoriasis treatments. Search engines look for not only the most relevant, but also the most helpful and user-friendly, answers to a user’s query.
Incidentally, if the URL for your site is not your own name, you should register your name as a separate domain name – even if you never use it – to be sure that a trickster or troll, or someone with the same name but a bad reputation, doesn’t get it.
Continue with a good meta description. That’s the grayish text that follows the title and URL in search results. Searchers will read it to confirm that your site is what they seek, so make sure it describes exactly what you do, including any areas of special expertise. Aggressive marketers will sometimes pad their descriptions with a wide variety of other specialties, services, and locations, hoping to gain inclusion in a larger pool of search results. That tactic – “keyword stuffing” in IT parlance – is not only ineffective, but search engines tend to ignore sites that use it. An accurate, honest description works best.
Describe your principal services in detail. You never know which specific service a prospective patient is searching for, so describe everything you offer. Don’t try to summarize everything on a single page; relevance is determined by how deeply a topic is covered, so each principal service should have a detailed description on its own page. Not only will your skills become more visible to search engines, but you can also use the space to enumerate your qualifications and expertise in each area. Whenever possible, write your descriptions in question-and-answer form. Searchers tend to ask questions (“what is the best…?”), particularly in voice searches. Search engines increasingly value sites that ask and answer common questions.
How does your site look on small screens? More than half of all searches are now made on smartphones, so the more “mobile friendly” your site is, the higher it will be ranked. Besides, prospective patients who are forced to scroll forever, or zoom in to tap a link, are likely to become frustrated and move on. Mobile searchers prefer sites that provide the best experience for the least amount of effort, and rankings tend to reflect that preference.
Include photos. Especially yours; new patients are more comfortable when they know what you look like. Although some disagree, I feel family photos are also important; they help to present you as a person, as well as a doctor. Photos of your office – professional ones, not casual snapshots – will reassure prospective patients that they will be visiting a clean, modern, professional facility.
Provide online appointment scheduling. Once searchers make an appointment, they stop searching. If they have to wait until the next day to call your office, they may keep looking – and might find a competitor with online scheduling. You should also have a separate “contact” page, listing all of the ways people can reach you, along with a map. Finally, list which insurance plans you accept as a courtesy to patients and to decrease unnecessary calls for your staff.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Nowadays, almost half of all Americans (and nearly all millennials) seek out doctors online. But your practice website should be doing a lot more than simply describing your practice. How many visitors to your site actually schedule an appointment? With a few relatively simple but important modifications, you can convert casual website viewers to patients.
Start with a good title, one that not only describes your practice but also anticipates how prospective patients will search for you – usually by specialty plus geographic location. My site’s title, for example, is “Belleville Dermatology Center,” so when someone searches for a dermatologist near Belleville, N.J., my site will invariably rank near the top of their search results.
Follow with an understandable URL. Search engines use URLs to determine how well your site, or a portion of it, matches search criteria. URLs also need to make sense to searchers, especially when they link specific areas of expertise (more on that in a minute). For example, a URL like “bellevilledermatology[dot]com/?p=89021” is meaningless to anyone except programmers; but “bellevilledermatology[dot]com/psoriasistreatments” obviously leads to a page about psoriasis treatments. Search engines look for not only the most relevant, but also the most helpful and user-friendly, answers to a user’s query.
Incidentally, if the URL for your site is not your own name, you should register your name as a separate domain name – even if you never use it – to be sure that a trickster or troll, or someone with the same name but a bad reputation, doesn’t get it.
Continue with a good meta description. That’s the grayish text that follows the title and URL in search results. Searchers will read it to confirm that your site is what they seek, so make sure it describes exactly what you do, including any areas of special expertise. Aggressive marketers will sometimes pad their descriptions with a wide variety of other specialties, services, and locations, hoping to gain inclusion in a larger pool of search results. That tactic – “keyword stuffing” in IT parlance – is not only ineffective, but search engines tend to ignore sites that use it. An accurate, honest description works best.
Describe your principal services in detail. You never know which specific service a prospective patient is searching for, so describe everything you offer. Don’t try to summarize everything on a single page; relevance is determined by how deeply a topic is covered, so each principal service should have a detailed description on its own page. Not only will your skills become more visible to search engines, but you can also use the space to enumerate your qualifications and expertise in each area. Whenever possible, write your descriptions in question-and-answer form. Searchers tend to ask questions (“what is the best…?”), particularly in voice searches. Search engines increasingly value sites that ask and answer common questions.
How does your site look on small screens? More than half of all searches are now made on smartphones, so the more “mobile friendly” your site is, the higher it will be ranked. Besides, prospective patients who are forced to scroll forever, or zoom in to tap a link, are likely to become frustrated and move on. Mobile searchers prefer sites that provide the best experience for the least amount of effort, and rankings tend to reflect that preference.
Include photos. Especially yours; new patients are more comfortable when they know what you look like. Although some disagree, I feel family photos are also important; they help to present you as a person, as well as a doctor. Photos of your office – professional ones, not casual snapshots – will reassure prospective patients that they will be visiting a clean, modern, professional facility.
Provide online appointment scheduling. Once searchers make an appointment, they stop searching. If they have to wait until the next day to call your office, they may keep looking – and might find a competitor with online scheduling. You should also have a separate “contact” page, listing all of the ways people can reach you, along with a map. Finally, list which insurance plans you accept as a courtesy to patients and to decrease unnecessary calls for your staff.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
Nowadays, almost half of all Americans (and nearly all millennials) seek out doctors online. But your practice website should be doing a lot more than simply describing your practice. How many visitors to your site actually schedule an appointment? With a few relatively simple but important modifications, you can convert casual website viewers to patients.
Start with a good title, one that not only describes your practice but also anticipates how prospective patients will search for you – usually by specialty plus geographic location. My site’s title, for example, is “Belleville Dermatology Center,” so when someone searches for a dermatologist near Belleville, N.J., my site will invariably rank near the top of their search results.
Follow with an understandable URL. Search engines use URLs to determine how well your site, or a portion of it, matches search criteria. URLs also need to make sense to searchers, especially when they link specific areas of expertise (more on that in a minute). For example, a URL like “bellevilledermatology[dot]com/?p=89021” is meaningless to anyone except programmers; but “bellevilledermatology[dot]com/psoriasistreatments” obviously leads to a page about psoriasis treatments. Search engines look for not only the most relevant, but also the most helpful and user-friendly, answers to a user’s query.
Incidentally, if the URL for your site is not your own name, you should register your name as a separate domain name – even if you never use it – to be sure that a trickster or troll, or someone with the same name but a bad reputation, doesn’t get it.
Continue with a good meta description. That’s the grayish text that follows the title and URL in search results. Searchers will read it to confirm that your site is what they seek, so make sure it describes exactly what you do, including any areas of special expertise. Aggressive marketers will sometimes pad their descriptions with a wide variety of other specialties, services, and locations, hoping to gain inclusion in a larger pool of search results. That tactic – “keyword stuffing” in IT parlance – is not only ineffective, but search engines tend to ignore sites that use it. An accurate, honest description works best.
Describe your principal services in detail. You never know which specific service a prospective patient is searching for, so describe everything you offer. Don’t try to summarize everything on a single page; relevance is determined by how deeply a topic is covered, so each principal service should have a detailed description on its own page. Not only will your skills become more visible to search engines, but you can also use the space to enumerate your qualifications and expertise in each area. Whenever possible, write your descriptions in question-and-answer form. Searchers tend to ask questions (“what is the best…?”), particularly in voice searches. Search engines increasingly value sites that ask and answer common questions.
How does your site look on small screens? More than half of all searches are now made on smartphones, so the more “mobile friendly” your site is, the higher it will be ranked. Besides, prospective patients who are forced to scroll forever, or zoom in to tap a link, are likely to become frustrated and move on. Mobile searchers prefer sites that provide the best experience for the least amount of effort, and rankings tend to reflect that preference.
Include photos. Especially yours; new patients are more comfortable when they know what you look like. Although some disagree, I feel family photos are also important; they help to present you as a person, as well as a doctor. Photos of your office – professional ones, not casual snapshots – will reassure prospective patients that they will be visiting a clean, modern, professional facility.
Provide online appointment scheduling. Once searchers make an appointment, they stop searching. If they have to wait until the next day to call your office, they may keep looking – and might find a competitor with online scheduling. You should also have a separate “contact” page, listing all of the ways people can reach you, along with a map. Finally, list which insurance plans you accept as a courtesy to patients and to decrease unnecessary calls for your staff.
Dr. Eastern practices dermatology and dermatologic surgery in Belleville, N.J. He is the author of numerous articles and textbook chapters, and is a longtime monthly columnist for Dermatology News. Write to him at [email protected].
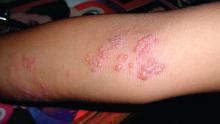
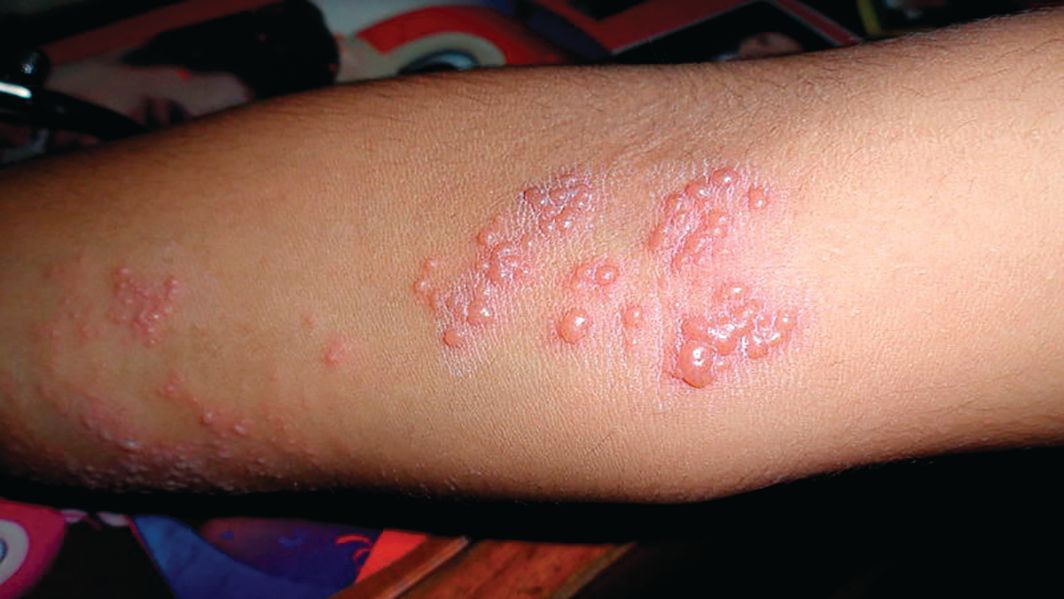
Herpes zoster boosts short-term stroke, TIA risk
ORLANDO – The risk of stroke and TIA – but not of acute MI – is significantly increased during the period surrounding diagnosis of herpes zoster, compared with the rate in matched zoster-free controls, according to a retrospective study of nearly 70,000 U.S. adults. A particularly striking study finding was the marked age disparity in the magnitude of vascular event risk associated with herpes zoster (HZ), with younger adults being at higher risk.
This increased vascular event risk associated with HZ was transitory. During the entire study follow-up period, which stretched from 1 month prior to HZ diagnosis to 12 months afterward, there was no overall increased vascular event risk associated with HZ, noted Dr. Rausch, an infectious diseases specialist who serves as director of clinical and medical affairs for the U.S. zoster program at GlaxoSmithKline in Philadelphia.
She presented a retrospective cohort study of U.S. Medicare and commercial health insurance claims data linked with EHRs for 2007-2014. The study included 23,339 adults diagnosed with HZ and 46,378 controls matched for sociodemographic and cardiovascular risk factors.
During the period from 1 month before to 1 month after HZ diagnosis, the rate of the composite vascular endpoint was 31.4 events per 1,000 person-years in the HZ group versus 24.5 per 1,000 person-years in controls. This difference was driven by a significantly higher rate of stroke/TIA in the HZ group. In contrast, the acute MI rates in the two groups were quite similar, at 6.9 per 1,000 person-years in the HZ group and 7.1 per 1,000 person-years in controls.
Further research is needed in order to shed light on the higher rate of vascular events observed in the younger patients with HZ. One possible explanation for the age-related difference is that HZ’s vascular effects is diluted in older patients, who have a higher burden of cardiovascular risk factors, Dr. Rausch noted.
The Centers for Disease Control and Prevention recommends the use of the two-dose recombinant Shingrix zoster vaccine in adults aged 50 years and older instead of the older, live-virus Zostavax vaccine, which is for adults aged 60 years and older.
Dr. Rausch’s study was sponsored by her employer, GlaxoSmithKline. The findings are supportive of an earlier study by other investigators, who found that the risk of hospitalization for stroke was up to twofold greater within the first 90 days after diagnosis of HZ in patients with rheumatoid arthritis and other autoimmune diseases than the stroke rate 366-730 days after HZ (Arthritis Rheumatol. 2017.69[2]:439-46).
SOURCE: Rausch DA. ACC 18
ORLANDO – The risk of stroke and TIA – but not of acute MI – is significantly increased during the period surrounding diagnosis of herpes zoster, compared with the rate in matched zoster-free controls, according to a retrospective study of nearly 70,000 U.S. adults. A particularly striking study finding was the marked age disparity in the magnitude of vascular event risk associated with herpes zoster (HZ), with younger adults being at higher risk.
This increased vascular event risk associated with HZ was transitory. During the entire study follow-up period, which stretched from 1 month prior to HZ diagnosis to 12 months afterward, there was no overall increased vascular event risk associated with HZ, noted Dr. Rausch, an infectious diseases specialist who serves as director of clinical and medical affairs for the U.S. zoster program at GlaxoSmithKline in Philadelphia.
She presented a retrospective cohort study of U.S. Medicare and commercial health insurance claims data linked with EHRs for 2007-2014. The study included 23,339 adults diagnosed with HZ and 46,378 controls matched for sociodemographic and cardiovascular risk factors.
During the period from 1 month before to 1 month after HZ diagnosis, the rate of the composite vascular endpoint was 31.4 events per 1,000 person-years in the HZ group versus 24.5 per 1,000 person-years in controls. This difference was driven by a significantly higher rate of stroke/TIA in the HZ group. In contrast, the acute MI rates in the two groups were quite similar, at 6.9 per 1,000 person-years in the HZ group and 7.1 per 1,000 person-years in controls.
Further research is needed in order to shed light on the higher rate of vascular events observed in the younger patients with HZ. One possible explanation for the age-related difference is that HZ’s vascular effects is diluted in older patients, who have a higher burden of cardiovascular risk factors, Dr. Rausch noted.
The Centers for Disease Control and Prevention recommends the use of the two-dose recombinant Shingrix zoster vaccine in adults aged 50 years and older instead of the older, live-virus Zostavax vaccine, which is for adults aged 60 years and older.
Dr. Rausch’s study was sponsored by her employer, GlaxoSmithKline. The findings are supportive of an earlier study by other investigators, who found that the risk of hospitalization for stroke was up to twofold greater within the first 90 days after diagnosis of HZ in patients with rheumatoid arthritis and other autoimmune diseases than the stroke rate 366-730 days after HZ (Arthritis Rheumatol. 2017.69[2]:439-46).
SOURCE: Rausch DA. ACC 18
ORLANDO – The risk of stroke and TIA – but not of acute MI – is significantly increased during the period surrounding diagnosis of herpes zoster, compared with the rate in matched zoster-free controls, according to a retrospective study of nearly 70,000 U.S. adults. A particularly striking study finding was the marked age disparity in the magnitude of vascular event risk associated with herpes zoster (HZ), with younger adults being at higher risk.
This increased vascular event risk associated with HZ was transitory. During the entire study follow-up period, which stretched from 1 month prior to HZ diagnosis to 12 months afterward, there was no overall increased vascular event risk associated with HZ, noted Dr. Rausch, an infectious diseases specialist who serves as director of clinical and medical affairs for the U.S. zoster program at GlaxoSmithKline in Philadelphia.
She presented a retrospective cohort study of U.S. Medicare and commercial health insurance claims data linked with EHRs for 2007-2014. The study included 23,339 adults diagnosed with HZ and 46,378 controls matched for sociodemographic and cardiovascular risk factors.
During the period from 1 month before to 1 month after HZ diagnosis, the rate of the composite vascular endpoint was 31.4 events per 1,000 person-years in the HZ group versus 24.5 per 1,000 person-years in controls. This difference was driven by a significantly higher rate of stroke/TIA in the HZ group. In contrast, the acute MI rates in the two groups were quite similar, at 6.9 per 1,000 person-years in the HZ group and 7.1 per 1,000 person-years in controls.
Further research is needed in order to shed light on the higher rate of vascular events observed in the younger patients with HZ. One possible explanation for the age-related difference is that HZ’s vascular effects is diluted in older patients, who have a higher burden of cardiovascular risk factors, Dr. Rausch noted.
The Centers for Disease Control and Prevention recommends the use of the two-dose recombinant Shingrix zoster vaccine in adults aged 50 years and older instead of the older, live-virus Zostavax vaccine, which is for adults aged 60 years and older.
Dr. Rausch’s study was sponsored by her employer, GlaxoSmithKline. The findings are supportive of an earlier study by other investigators, who found that the risk of hospitalization for stroke was up to twofold greater within the first 90 days after diagnosis of HZ in patients with rheumatoid arthritis and other autoimmune diseases than the stroke rate 366-730 days after HZ (Arthritis Rheumatol. 2017.69[2]:439-46).
SOURCE: Rausch DA. ACC 18
REPORTING FROM ACC 2018
Key clinical point: Herpes zoster is associated with significantly increased short-term risk of stroke/TIA.
Major finding: Adults aged 18-49 years were threefold more likely than controls to experience stroke or TIA within a month of herpes zoster diagnosis.
Study details: This retrospective study included 23,339 U.S. adults with herpes zoster and twice as many matched controls.
Disclosures: The study was sponsored by GlaxoSmithKline and presented by a GSK employee.
Source: Rausch DA. ACC 18
VIDEO: Indocyanine green finds more sentinel lymph nodes
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – Indocyanine green (ICG) worked better than isosulfan blue for mapping sentinel lymph nodes (SLNs) in a pivotal phase 3 trial with 176 patients who had stage I endometrial or cervical cancer.
Four injections of ICG resulted in detection of 96% of the identified SLNs in these patients, including bilateral SLNs in 78% of the patients. In contrast, four injections with isosulfan blue dye led to detection of 74% of all SLNs and identified bilateral SLNs in 31% of the patients, Michael M. Frumovitz, MD, said at the annual meeting of the Society for Gynecologic Oncology.
The FILM trial randomized 176 patients with stage I endometrial or cervical cancer at eight centers in the United States or Canada between December 2015 and May 2017. Patients first received one of the tagging agents and then the second, and then underwent mapping using white light to detect blue-tagged SLNs and near-infrared light to find green-tagged SLNs. The patients were aged 63 years on average, and 96% had endometrial cancer.
The researchers identified 279 sentinel lymph nodes that stained only green, nine SLNs that stained only blue, and 248 SLNs tagged with both dyes. They confirmed tumor cells within all nine of SLNs tagged with blue dye only, in 95% of those tagged with ICG only, and in 92% of the SLNs stained with both dyes. The isosulfan blue dye identified SLNs in two patients who did not have any SLNs detected by the ICG, whereas the ICG identified SLNs in 22 patients who did not have any SLNs detected using the blue dye. Sixteen patients had metastatic disease that had moved to 21 SLNs. The ICG system identified all 21 involved lymph nodes; the blue dye identified 13 of the 21 affected SLNs (62%).
Dr. Frumovitz and his associates designed FILM as primarily a test of noninferiority. The per-protocol analysis with 163 patients showed that ICG was noninferior to isosulfan blue (P less than .001). Once the results demonstrated noninferiority, the study protocol allowed the researchers to test for superiority in the full, intention-to-treat cohort of 176 patients. The results showed that ICG was significantly superior to isosulfan blue (P less than .001). In addition, ICG treatment produced no allergic or other adverse reactions, Dr. Frumovitz said.
Once ICG and the associated near-infrared detection camera receive FDA marketing approval, “I think this will become the standard within 5 years,” he predicted in an interview.
The results also showed that using both ICG and isosulfan blue was not better than using ICG alone. “If you’re using both dyes, you can drop the blue dye. At MD Anderson we’ve used only ICG for about the past year,” Dr. Frumovitz said.
FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
SOURCE: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
The results from the FILM trial are potentially practice changing. The findings presented by Michael M. Frumovitz, MD, and his associates showed that indocyanine green is superior to isosulfan blue dye for mapping sentinel lymph nodes in patients with stage I endometrial or cervical cancer. The results also showed that using both dyes was no better than using indocyanine green alone.
The report by Floor J. Backes, MD, addressed an important and still unresolved question in treating patients with stage I or II endometrial cancer: What is the significance of finding isolated tumor cells in sentinel lymph nodes in these patients? The retrospective findings she presented showed that the presence of isolated tumor cells had no apparent effect on recurrence-free survival, recurrence pattern, or patient response to various treatments. This suggested th at treatment decisions in these patients should depend on other high-risk uterine factors but not on whether some lymph nodes contained isolated tumor cells.
Brent Smith, MD , is a gynecologic oncologist at the Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The results from the FILM trial are potentially practice changing. The findings presented by Michael M. Frumovitz, MD, and his associates showed that indocyanine green is superior to isosulfan blue dye for mapping sentinel lymph nodes in patients with stage I endometrial or cervical cancer. The results also showed that using both dyes was no better than using indocyanine green alone.
The report by Floor J. Backes, MD, addressed an important and still unresolved question in treating patients with stage I or II endometrial cancer: What is the significance of finding isolated tumor cells in sentinel lymph nodes in these patients? The retrospective findings she presented showed that the presence of isolated tumor cells had no apparent effect on recurrence-free survival, recurrence pattern, or patient response to various treatments. This suggested th at treatment decisions in these patients should depend on other high-risk uterine factors but not on whether some lymph nodes contained isolated tumor cells.
Brent Smith, MD , is a gynecologic oncologist at the Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The results from the FILM trial are potentially practice changing. The findings presented by Michael M. Frumovitz, MD, and his associates showed that indocyanine green is superior to isosulfan blue dye for mapping sentinel lymph nodes in patients with stage I endometrial or cervical cancer. The results also showed that using both dyes was no better than using indocyanine green alone.
The report by Floor J. Backes, MD, addressed an important and still unresolved question in treating patients with stage I or II endometrial cancer: What is the significance of finding isolated tumor cells in sentinel lymph nodes in these patients? The retrospective findings she presented showed that the presence of isolated tumor cells had no apparent effect on recurrence-free survival, recurrence pattern, or patient response to various treatments. This suggested th at treatment decisions in these patients should depend on other high-risk uterine factors but not on whether some lymph nodes contained isolated tumor cells.
Brent Smith, MD , is a gynecologic oncologist at the Ohio State University, Columbus. He had no disclosures. Dr. Smith made these comments in a video interview.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – Indocyanine green (ICG) worked better than isosulfan blue for mapping sentinel lymph nodes (SLNs) in a pivotal phase 3 trial with 176 patients who had stage I endometrial or cervical cancer.
Four injections of ICG resulted in detection of 96% of the identified SLNs in these patients, including bilateral SLNs in 78% of the patients. In contrast, four injections with isosulfan blue dye led to detection of 74% of all SLNs and identified bilateral SLNs in 31% of the patients, Michael M. Frumovitz, MD, said at the annual meeting of the Society for Gynecologic Oncology.
The FILM trial randomized 176 patients with stage I endometrial or cervical cancer at eight centers in the United States or Canada between December 2015 and May 2017. Patients first received one of the tagging agents and then the second, and then underwent mapping using white light to detect blue-tagged SLNs and near-infrared light to find green-tagged SLNs. The patients were aged 63 years on average, and 96% had endometrial cancer.
The researchers identified 279 sentinel lymph nodes that stained only green, nine SLNs that stained only blue, and 248 SLNs tagged with both dyes. They confirmed tumor cells within all nine of SLNs tagged with blue dye only, in 95% of those tagged with ICG only, and in 92% of the SLNs stained with both dyes. The isosulfan blue dye identified SLNs in two patients who did not have any SLNs detected by the ICG, whereas the ICG identified SLNs in 22 patients who did not have any SLNs detected using the blue dye. Sixteen patients had metastatic disease that had moved to 21 SLNs. The ICG system identified all 21 involved lymph nodes; the blue dye identified 13 of the 21 affected SLNs (62%).
Dr. Frumovitz and his associates designed FILM as primarily a test of noninferiority. The per-protocol analysis with 163 patients showed that ICG was noninferior to isosulfan blue (P less than .001). Once the results demonstrated noninferiority, the study protocol allowed the researchers to test for superiority in the full, intention-to-treat cohort of 176 patients. The results showed that ICG was significantly superior to isosulfan blue (P less than .001). In addition, ICG treatment produced no allergic or other adverse reactions, Dr. Frumovitz said.
Once ICG and the associated near-infrared detection camera receive FDA marketing approval, “I think this will become the standard within 5 years,” he predicted in an interview.
The results also showed that using both ICG and isosulfan blue was not better than using ICG alone. “If you’re using both dyes, you can drop the blue dye. At MD Anderson we’ve used only ICG for about the past year,” Dr. Frumovitz said.
FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
SOURCE: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEW ORLEANS – Indocyanine green (ICG) worked better than isosulfan blue for mapping sentinel lymph nodes (SLNs) in a pivotal phase 3 trial with 176 patients who had stage I endometrial or cervical cancer.
Four injections of ICG resulted in detection of 96% of the identified SLNs in these patients, including bilateral SLNs in 78% of the patients. In contrast, four injections with isosulfan blue dye led to detection of 74% of all SLNs and identified bilateral SLNs in 31% of the patients, Michael M. Frumovitz, MD, said at the annual meeting of the Society for Gynecologic Oncology.
The FILM trial randomized 176 patients with stage I endometrial or cervical cancer at eight centers in the United States or Canada between December 2015 and May 2017. Patients first received one of the tagging agents and then the second, and then underwent mapping using white light to detect blue-tagged SLNs and near-infrared light to find green-tagged SLNs. The patients were aged 63 years on average, and 96% had endometrial cancer.
The researchers identified 279 sentinel lymph nodes that stained only green, nine SLNs that stained only blue, and 248 SLNs tagged with both dyes. They confirmed tumor cells within all nine of SLNs tagged with blue dye only, in 95% of those tagged with ICG only, and in 92% of the SLNs stained with both dyes. The isosulfan blue dye identified SLNs in two patients who did not have any SLNs detected by the ICG, whereas the ICG identified SLNs in 22 patients who did not have any SLNs detected using the blue dye. Sixteen patients had metastatic disease that had moved to 21 SLNs. The ICG system identified all 21 involved lymph nodes; the blue dye identified 13 of the 21 affected SLNs (62%).
Dr. Frumovitz and his associates designed FILM as primarily a test of noninferiority. The per-protocol analysis with 163 patients showed that ICG was noninferior to isosulfan blue (P less than .001). Once the results demonstrated noninferiority, the study protocol allowed the researchers to test for superiority in the full, intention-to-treat cohort of 176 patients. The results showed that ICG was significantly superior to isosulfan blue (P less than .001). In addition, ICG treatment produced no allergic or other adverse reactions, Dr. Frumovitz said.
Once ICG and the associated near-infrared detection camera receive FDA marketing approval, “I think this will become the standard within 5 years,” he predicted in an interview.
The results also showed that using both ICG and isosulfan blue was not better than using ICG alone. “If you’re using both dyes, you can drop the blue dye. At MD Anderson we’ve used only ICG for about the past year,” Dr. Frumovitz said.
FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
SOURCE: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
REPORTING FROM SGO 2018
Key clinical point: Indocyanine green surpassed isosulfan blue for sentinel lymph node mapping in a pivotal trial.
Major finding: Researchers mapped sentinel lymph nodes in 96% of patients with indocyanine green and in 74% with isosulfan blue.
Study details: FILM, a multicenter, randomized phase 3 trial with 176 patients.
Disclosures: FILM was sponsored by Novadaq/Stryker, the company developing the ICG PINPOINT imaging system. Dr. Frumovitz has been a consultant to Novadaq/Stryker and Genentech and has received research funding from Novadaq/Stryker and Navidea. Dr. Backes has been a consultant to Tesaro and has received research funding from Clovis, Eisai, and ImmunoGen. Dr. Buda had no disclosures.
Source: Frumovitz MM. SGO 2018, Abstract 12. Backes FJ. SGO 2018, Abstract 13.
Pot peaks in breast milk 1 hour after smoking
The levels of delta-9-tetrahydrocannabinol in breast milk peak around 1 hour after smoking, according to a pilot pharmacokinetic study.
Eight mothers who were either occasional or chronic cannabis smokers, and who exclusively breastfed their infants, were directed to smoke a preweighed, standardized amount of “Prezidential Kush” from a preselected Denver dispensary after initially discontinuing for 24 hours. Researchers collected breast milk samples from just before smoking, and at 20 minutes, 1 hour, 2 hours, and 4 hours after smoking, according to a paper published in the May issue of Obstetrics & Gynecology.
This translated to an estimated relative infant dose of 2.5% of the maternal dose, or 8 mcg per kilogram per day.
“It remains unclear what exposure to cannabis products during this critical neurobehavioral development period will mean for the infant,” wrote Teresa Baker, MD, of Texas Tech University Health Sciences Center, and her coauthors. “These questions will require an enormous effort to determine.”
Concentrations of delta-9-tetrahydrocannabinol metabolites 11-OH-delta-9-tetrahydrocannabinol and 11-Nor-9-carboxy-delta-9-tetrahydrocannabinol were too low to be detected.
The authors noted that these metabolites are known to be more water soluble and polar than delta-9-tetrahydrocannabinol itself, which may make it more difficult for them to enter the breast milk compartment.
Two of the participants had a low but measurable concentration of delta-9-tetrahydrocannabinol at zero time (the samples collected just before smoking), suggesting some residual accumulation of it from prior heavy use or use close to the start of breast milk collection.
“Although the transfer of delta-9-tetrahydrocannabinol into the plasma compartment is almost instantaneous, the transfer of delta-9-tetrahydrocannabinol into breast milk in our study appears to be slightly slower than the transfer into the plasma compartment,” the authors wrote.
The women in the study were all 2-5 months postpartum. The authors noted that in these women who were exclusively breastfeeding, the breast milk compartment, along with drug entry and exit, “remains fairly consistent.”
The researchers also noted that the lack of corresponding plasma samples was a major limitation of the study, but because of the nature of the study, anonymity was important.
No conflicts of interest were declared.
SOURCE: Baker T et al. Obstet Gynecol. 2018;131:783-8.
The levels of delta-9-tetrahydrocannabinol in breast milk peak around 1 hour after smoking, according to a pilot pharmacokinetic study.
Eight mothers who were either occasional or chronic cannabis smokers, and who exclusively breastfed their infants, were directed to smoke a preweighed, standardized amount of “Prezidential Kush” from a preselected Denver dispensary after initially discontinuing for 24 hours. Researchers collected breast milk samples from just before smoking, and at 20 minutes, 1 hour, 2 hours, and 4 hours after smoking, according to a paper published in the May issue of Obstetrics & Gynecology.
This translated to an estimated relative infant dose of 2.5% of the maternal dose, or 8 mcg per kilogram per day.
“It remains unclear what exposure to cannabis products during this critical neurobehavioral development period will mean for the infant,” wrote Teresa Baker, MD, of Texas Tech University Health Sciences Center, and her coauthors. “These questions will require an enormous effort to determine.”
Concentrations of delta-9-tetrahydrocannabinol metabolites 11-OH-delta-9-tetrahydrocannabinol and 11-Nor-9-carboxy-delta-9-tetrahydrocannabinol were too low to be detected.
The authors noted that these metabolites are known to be more water soluble and polar than delta-9-tetrahydrocannabinol itself, which may make it more difficult for them to enter the breast milk compartment.
Two of the participants had a low but measurable concentration of delta-9-tetrahydrocannabinol at zero time (the samples collected just before smoking), suggesting some residual accumulation of it from prior heavy use or use close to the start of breast milk collection.
“Although the transfer of delta-9-tetrahydrocannabinol into the plasma compartment is almost instantaneous, the transfer of delta-9-tetrahydrocannabinol into breast milk in our study appears to be slightly slower than the transfer into the plasma compartment,” the authors wrote.
The women in the study were all 2-5 months postpartum. The authors noted that in these women who were exclusively breastfeeding, the breast milk compartment, along with drug entry and exit, “remains fairly consistent.”
The researchers also noted that the lack of corresponding plasma samples was a major limitation of the study, but because of the nature of the study, anonymity was important.
No conflicts of interest were declared.
SOURCE: Baker T et al. Obstet Gynecol. 2018;131:783-8.
The levels of delta-9-tetrahydrocannabinol in breast milk peak around 1 hour after smoking, according to a pilot pharmacokinetic study.
Eight mothers who were either occasional or chronic cannabis smokers, and who exclusively breastfed their infants, were directed to smoke a preweighed, standardized amount of “Prezidential Kush” from a preselected Denver dispensary after initially discontinuing for 24 hours. Researchers collected breast milk samples from just before smoking, and at 20 minutes, 1 hour, 2 hours, and 4 hours after smoking, according to a paper published in the May issue of Obstetrics & Gynecology.
This translated to an estimated relative infant dose of 2.5% of the maternal dose, or 8 mcg per kilogram per day.
“It remains unclear what exposure to cannabis products during this critical neurobehavioral development period will mean for the infant,” wrote Teresa Baker, MD, of Texas Tech University Health Sciences Center, and her coauthors. “These questions will require an enormous effort to determine.”
Concentrations of delta-9-tetrahydrocannabinol metabolites 11-OH-delta-9-tetrahydrocannabinol and 11-Nor-9-carboxy-delta-9-tetrahydrocannabinol were too low to be detected.
The authors noted that these metabolites are known to be more water soluble and polar than delta-9-tetrahydrocannabinol itself, which may make it more difficult for them to enter the breast milk compartment.
Two of the participants had a low but measurable concentration of delta-9-tetrahydrocannabinol at zero time (the samples collected just before smoking), suggesting some residual accumulation of it from prior heavy use or use close to the start of breast milk collection.
“Although the transfer of delta-9-tetrahydrocannabinol into the plasma compartment is almost instantaneous, the transfer of delta-9-tetrahydrocannabinol into breast milk in our study appears to be slightly slower than the transfer into the plasma compartment,” the authors wrote.
The women in the study were all 2-5 months postpartum. The authors noted that in these women who were exclusively breastfeeding, the breast milk compartment, along with drug entry and exit, “remains fairly consistent.”
The researchers also noted that the lack of corresponding plasma samples was a major limitation of the study, but because of the nature of the study, anonymity was important.
No conflicts of interest were declared.
SOURCE: Baker T et al. Obstet Gynecol. 2018;131:783-8.
FROM OBSTETRICS & GYNECOLOGY
Key clinical point: Cannabinoid levels peak in breast milk 1 hour after smoking.
Major finding: Breastfeeding infants ingest around 2.5% of the maternal dose in breast milk.
Study details: A pilot pharmacokinetic study in eight women.
Disclosures: No conflicts of interest were declared.
Source: Baker T et al. Obstet Gynecol. 2018;131:783-8.
Disrupted sleep tied to alexithymia
a pair of studies shows.
Alexithymia is a condition characterized by difficulty identifying and expressing one’s emotions. “The mechanism by which alexithymia confers risk of disrupted sleep remains unclear, [but] suggestions include increased nocturnal arousal as a result of poor verbalization of emotions and increased light sleep,” wrote Jennifer Murphy, citing previous research.
The researchers found associations between total alexithymia scores and reduced sleep quality (P less than .001). They also found a significant association between the TAS-20 subscales and reduced sleep quality (all P less than .006).
In the second study, in which 73 men and women participated, Ms. Murphy and her associates sought to determine whether the association found in the first study was tied to depression or anxiety. Participants went online and completed three questionnaires: the TAS-20; the PSQI; and the Depression, Anxiety, & Stress Scale, or DASS-21, in a randomized order. Higher scores on the DASS-21 correlate with greater levels of depression, anxiety, and stress. None of the questionnaires asked about any aspects of sleep.
Ms. Murphy said in an interview that although it might be too early to make a clear clinical recommendation, the results suggest that “clinicians should be aware of the possibility of sleep problems characterized by heightened alexithymia and more generally in those with alexithymia.”
Whilst further research is needed to confirm the direction of causality between disrupted sleep and alexithymia and how these subjective sleep reports in alexithymia map onto objectively measured sleep problems, these data suggest a link that is independent of depression and anxiety, she said.
Meanwhile, other researchers report that alexithymia is becoming more clinically relevant. Rising rates of alexithymia are being reported in psychiatric conditions such as autism, eating disorders, schizophrenia, and alcohol and substance abuse. The condition is also seen in neurologic conditions such as multiple sclerosis and traumatic brain injury (Neuropsychologica. 2018;11:229-40).
Ms. Murphy and her associates cited several limitations of their research. One is that they did not control for factors that affect sleep quality and alexithymia such as body composition. They also cited reports of discrepancies between objective and subjective measures – such as those made by self-report – and the relatively small sample sizes.
The research was supported by the Economic and Social Research Council and the Baily Thomas Charitable Trust. No conflicts of interest were reported.
SOURCE: Murphy J et al. Pers Individ Dif. 2018 Mar 27;129:175-8.
a pair of studies shows.
Alexithymia is a condition characterized by difficulty identifying and expressing one’s emotions. “The mechanism by which alexithymia confers risk of disrupted sleep remains unclear, [but] suggestions include increased nocturnal arousal as a result of poor verbalization of emotions and increased light sleep,” wrote Jennifer Murphy, citing previous research.
The researchers found associations between total alexithymia scores and reduced sleep quality (P less than .001). They also found a significant association between the TAS-20 subscales and reduced sleep quality (all P less than .006).
In the second study, in which 73 men and women participated, Ms. Murphy and her associates sought to determine whether the association found in the first study was tied to depression or anxiety. Participants went online and completed three questionnaires: the TAS-20; the PSQI; and the Depression, Anxiety, & Stress Scale, or DASS-21, in a randomized order. Higher scores on the DASS-21 correlate with greater levels of depression, anxiety, and stress. None of the questionnaires asked about any aspects of sleep.
Ms. Murphy said in an interview that although it might be too early to make a clear clinical recommendation, the results suggest that “clinicians should be aware of the possibility of sleep problems characterized by heightened alexithymia and more generally in those with alexithymia.”
Whilst further research is needed to confirm the direction of causality between disrupted sleep and alexithymia and how these subjective sleep reports in alexithymia map onto objectively measured sleep problems, these data suggest a link that is independent of depression and anxiety, she said.
Meanwhile, other researchers report that alexithymia is becoming more clinically relevant. Rising rates of alexithymia are being reported in psychiatric conditions such as autism, eating disorders, schizophrenia, and alcohol and substance abuse. The condition is also seen in neurologic conditions such as multiple sclerosis and traumatic brain injury (Neuropsychologica. 2018;11:229-40).
Ms. Murphy and her associates cited several limitations of their research. One is that they did not control for factors that affect sleep quality and alexithymia such as body composition. They also cited reports of discrepancies between objective and subjective measures – such as those made by self-report – and the relatively small sample sizes.
The research was supported by the Economic and Social Research Council and the Baily Thomas Charitable Trust. No conflicts of interest were reported.
SOURCE: Murphy J et al. Pers Individ Dif. 2018 Mar 27;129:175-8.
a pair of studies shows.
Alexithymia is a condition characterized by difficulty identifying and expressing one’s emotions. “The mechanism by which alexithymia confers risk of disrupted sleep remains unclear, [but] suggestions include increased nocturnal arousal as a result of poor verbalization of emotions and increased light sleep,” wrote Jennifer Murphy, citing previous research.
The researchers found associations between total alexithymia scores and reduced sleep quality (P less than .001). They also found a significant association between the TAS-20 subscales and reduced sleep quality (all P less than .006).
In the second study, in which 73 men and women participated, Ms. Murphy and her associates sought to determine whether the association found in the first study was tied to depression or anxiety. Participants went online and completed three questionnaires: the TAS-20; the PSQI; and the Depression, Anxiety, & Stress Scale, or DASS-21, in a randomized order. Higher scores on the DASS-21 correlate with greater levels of depression, anxiety, and stress. None of the questionnaires asked about any aspects of sleep.
Ms. Murphy said in an interview that although it might be too early to make a clear clinical recommendation, the results suggest that “clinicians should be aware of the possibility of sleep problems characterized by heightened alexithymia and more generally in those with alexithymia.”
Whilst further research is needed to confirm the direction of causality between disrupted sleep and alexithymia and how these subjective sleep reports in alexithymia map onto objectively measured sleep problems, these data suggest a link that is independent of depression and anxiety, she said.
Meanwhile, other researchers report that alexithymia is becoming more clinically relevant. Rising rates of alexithymia are being reported in psychiatric conditions such as autism, eating disorders, schizophrenia, and alcohol and substance abuse. The condition is also seen in neurologic conditions such as multiple sclerosis and traumatic brain injury (Neuropsychologica. 2018;11:229-40).
Ms. Murphy and her associates cited several limitations of their research. One is that they did not control for factors that affect sleep quality and alexithymia such as body composition. They also cited reports of discrepancies between objective and subjective measures – such as those made by self-report – and the relatively small sample sizes.
The research was supported by the Economic and Social Research Council and the Baily Thomas Charitable Trust. No conflicts of interest were reported.
SOURCE: Murphy J et al. Pers Individ Dif. 2018 Mar 27;129:175-8.
FROM PERSONALITY AND INDIVIDUAL DIFFERENCES
Transcatheter valves underperform for native aortic regurgitation
WASHINGTON – Transcatheter heart valves (THV) developed for the treatment of symptomatic aortic stenosis have been used off label for the treatment of native aortic valve regurgitation (NAVR), but registry data suggest that outcomes have been disappointing, according to a presentation at CRT 2018 sponsored by the Cardiovascular Research Institute at Washington Hospital Center.
“Although significant improvement was seen with newer-generation THV devices, TAVR [transcatheter aortic valve replacement] for NAVR is a challenging approach associated with limited procedural efficacy,” reported Danny Dvir, MD, a prosthetic heart valve specialist and assistant professor of cardiology at the University of Washington, Seattle.
Results improved substantially with second-generation devices. For example, Dr. Dvir reported that the rate of device success climbed from 47% to 82% while correct positioning climbed from 67% to 91%. The proportion of patients without moderate or severe aortic regurgitation after placement of the THV climbed from 69% to 96%.
These improvements were reflected in clinical outcomes at 30 days. When second-generation devices were compared with first-generation devices, there was a reduction in all cause mortality (8% vs. 17%) and cardiac mortality (7% vs. 12%). There were also reductions from first- to second-generation devices in noncardiac mortality (1% vs. 5%), valve-related dysfunction (10% vs. 29%), and proportion of patients in New York Heart Association class III or IV (13% vs. 18%).
The improvement in outcomes from first- to second-generation devices is encouraging, but Dr. Dvir indicated that the main message is that TAVR for NAVR is producing success rates that “are suboptimal” and “not comparable to those being achieved when the indication is aortic stenosis.” The reasons cannot be derived from these data, but he suggested that optimal sizing of the device for NAVR might be different than it is for aortic stenosis.
“I wonder if we should have better devices designed specifically for aortic regurgitation,” Dr. Dvir said.
Despite the improved results with second-generation THV, receipt of a first-generation device was not a significant predictor of mortality at 1 year. Rather, in an analysis of predictors, mortality was significantly increased in those with moderate or worse aortic regurgitation, Society of Thoracic Surgeons risk score of 8% or greater, and acute kidney injury of grade 2 or higher. There was also a trend for increased mortality in those with pulmonary hypertension.
Most of the devices (76%) were placed with a transfemoral approach. No difference in mortality was observed when a transfemoral approach was compared with a nontransfemoral approach.
According to the registry data, a 10%-20% oversizing of the THV was associated with a reduced risk of malpositioning, relative to devices with less than 10% oversizing or greater than 20% oversizing, reinforcing Dr. Dvir’s hypothesis that sizing is a variable affecting outcome in NAVR.
Although Dr. Dvir contended that these data raise issues about the suitability of current THV designs for use in the treatment of NAVR, not all experts were convinced by these data. Jeffrey Popma, MD, director of the interventional cardiology clinical service at Beth Israel Deaconess Hospital, Boston, questioned whether more experience is placing these devices for NAVR might lead to greater success.
“There are two variables to consider,” Dr. Popma said. “One is the valve and one is how much we’ve evolved our procedure over the past couple of years.” He indicated that these data do not preclude advances that would improve results in NAVR even without developing new valves specific for this indication.
Dr. Dvir reported financial relationships with Edwards Lifesciences, Medtronic, Abbott, and Jena.
SOURCE: Dvir D. CRT 2018.
WASHINGTON – Transcatheter heart valves (THV) developed for the treatment of symptomatic aortic stenosis have been used off label for the treatment of native aortic valve regurgitation (NAVR), but registry data suggest that outcomes have been disappointing, according to a presentation at CRT 2018 sponsored by the Cardiovascular Research Institute at Washington Hospital Center.
“Although significant improvement was seen with newer-generation THV devices, TAVR [transcatheter aortic valve replacement] for NAVR is a challenging approach associated with limited procedural efficacy,” reported Danny Dvir, MD, a prosthetic heart valve specialist and assistant professor of cardiology at the University of Washington, Seattle.
Results improved substantially with second-generation devices. For example, Dr. Dvir reported that the rate of device success climbed from 47% to 82% while correct positioning climbed from 67% to 91%. The proportion of patients without moderate or severe aortic regurgitation after placement of the THV climbed from 69% to 96%.
These improvements were reflected in clinical outcomes at 30 days. When second-generation devices were compared with first-generation devices, there was a reduction in all cause mortality (8% vs. 17%) and cardiac mortality (7% vs. 12%). There were also reductions from first- to second-generation devices in noncardiac mortality (1% vs. 5%), valve-related dysfunction (10% vs. 29%), and proportion of patients in New York Heart Association class III or IV (13% vs. 18%).
The improvement in outcomes from first- to second-generation devices is encouraging, but Dr. Dvir indicated that the main message is that TAVR for NAVR is producing success rates that “are suboptimal” and “not comparable to those being achieved when the indication is aortic stenosis.” The reasons cannot be derived from these data, but he suggested that optimal sizing of the device for NAVR might be different than it is for aortic stenosis.
“I wonder if we should have better devices designed specifically for aortic regurgitation,” Dr. Dvir said.
Despite the improved results with second-generation THV, receipt of a first-generation device was not a significant predictor of mortality at 1 year. Rather, in an analysis of predictors, mortality was significantly increased in those with moderate or worse aortic regurgitation, Society of Thoracic Surgeons risk score of 8% or greater, and acute kidney injury of grade 2 or higher. There was also a trend for increased mortality in those with pulmonary hypertension.
Most of the devices (76%) were placed with a transfemoral approach. No difference in mortality was observed when a transfemoral approach was compared with a nontransfemoral approach.
According to the registry data, a 10%-20% oversizing of the THV was associated with a reduced risk of malpositioning, relative to devices with less than 10% oversizing or greater than 20% oversizing, reinforcing Dr. Dvir’s hypothesis that sizing is a variable affecting outcome in NAVR.
Although Dr. Dvir contended that these data raise issues about the suitability of current THV designs for use in the treatment of NAVR, not all experts were convinced by these data. Jeffrey Popma, MD, director of the interventional cardiology clinical service at Beth Israel Deaconess Hospital, Boston, questioned whether more experience is placing these devices for NAVR might lead to greater success.
“There are two variables to consider,” Dr. Popma said. “One is the valve and one is how much we’ve evolved our procedure over the past couple of years.” He indicated that these data do not preclude advances that would improve results in NAVR even without developing new valves specific for this indication.
Dr. Dvir reported financial relationships with Edwards Lifesciences, Medtronic, Abbott, and Jena.
SOURCE: Dvir D. CRT 2018.
WASHINGTON – Transcatheter heart valves (THV) developed for the treatment of symptomatic aortic stenosis have been used off label for the treatment of native aortic valve regurgitation (NAVR), but registry data suggest that outcomes have been disappointing, according to a presentation at CRT 2018 sponsored by the Cardiovascular Research Institute at Washington Hospital Center.
“Although significant improvement was seen with newer-generation THV devices, TAVR [transcatheter aortic valve replacement] for NAVR is a challenging approach associated with limited procedural efficacy,” reported Danny Dvir, MD, a prosthetic heart valve specialist and assistant professor of cardiology at the University of Washington, Seattle.
Results improved substantially with second-generation devices. For example, Dr. Dvir reported that the rate of device success climbed from 47% to 82% while correct positioning climbed from 67% to 91%. The proportion of patients without moderate or severe aortic regurgitation after placement of the THV climbed from 69% to 96%.
These improvements were reflected in clinical outcomes at 30 days. When second-generation devices were compared with first-generation devices, there was a reduction in all cause mortality (8% vs. 17%) and cardiac mortality (7% vs. 12%). There were also reductions from first- to second-generation devices in noncardiac mortality (1% vs. 5%), valve-related dysfunction (10% vs. 29%), and proportion of patients in New York Heart Association class III or IV (13% vs. 18%).
The improvement in outcomes from first- to second-generation devices is encouraging, but Dr. Dvir indicated that the main message is that TAVR for NAVR is producing success rates that “are suboptimal” and “not comparable to those being achieved when the indication is aortic stenosis.” The reasons cannot be derived from these data, but he suggested that optimal sizing of the device for NAVR might be different than it is for aortic stenosis.
“I wonder if we should have better devices designed specifically for aortic regurgitation,” Dr. Dvir said.
Despite the improved results with second-generation THV, receipt of a first-generation device was not a significant predictor of mortality at 1 year. Rather, in an analysis of predictors, mortality was significantly increased in those with moderate or worse aortic regurgitation, Society of Thoracic Surgeons risk score of 8% or greater, and acute kidney injury of grade 2 or higher. There was also a trend for increased mortality in those with pulmonary hypertension.
Most of the devices (76%) were placed with a transfemoral approach. No difference in mortality was observed when a transfemoral approach was compared with a nontransfemoral approach.
According to the registry data, a 10%-20% oversizing of the THV was associated with a reduced risk of malpositioning, relative to devices with less than 10% oversizing or greater than 20% oversizing, reinforcing Dr. Dvir’s hypothesis that sizing is a variable affecting outcome in NAVR.
Although Dr. Dvir contended that these data raise issues about the suitability of current THV designs for use in the treatment of NAVR, not all experts were convinced by these data. Jeffrey Popma, MD, director of the interventional cardiology clinical service at Beth Israel Deaconess Hospital, Boston, questioned whether more experience is placing these devices for NAVR might lead to greater success.
“There are two variables to consider,” Dr. Popma said. “One is the valve and one is how much we’ve evolved our procedure over the past couple of years.” He indicated that these data do not preclude advances that would improve results in NAVR even without developing new valves specific for this indication.
Dr. Dvir reported financial relationships with Edwards Lifesciences, Medtronic, Abbott, and Jena.
SOURCE: Dvir D. CRT 2018.
AT THE 2018 CRT MEETING
Key clinical point:
Major finding: With newer-generation THV, rates of incorrect positioning (9%), persistent regurgitation (4%), and 30-day mortality (8%) remain unacceptably high.
Data source: A registry data analysis.
Disclosures: Dr. Dvir reported financial relationships with Edwards Lifesciences, Medtronic, Abbott, and Jena.
Source: Dvir D. CRT 2018.
Hurricane relief and patient care
In October 2017, in support of the Federal Emergency Management Agency’s response to assist the Governor and people of Puerto Rico, three Department of Defense (DOD) military hospital platforms were deployed; one each, by the US Army, Navy, and Air Force. They arrived on the island at different times with predominantly wartime surgical capabilities and augmented the Federal Emergency Management Agency (FEMA), US Public Health Service, National Guard, and Puerto Rico Department of Health efforts. My perspective is that of patient care and transport between the Centro Medico hospital complex in San Juan, the larger regional hospitals, the Veterans Administration hospital, the DOD response, FEMA Disaster Medical Assistance Teams (DMAT), and FEMA Federal Medical Shelters about 4 to 6 weeks after Hurricanes Maria and Irma struck. Based upon this experience, I would like to offer the following.
Pre-Disaster: All clinicians have a few patients that teeter “on the edge.” When basic services go away, these patients fall over that edge and become inpatients. Establish a list of patients who require oxygen and devices such as vests, cough-assist, or ventilation. If evacuation before the disaster is possible, those patients need to leave. If they refuse, or are unable to leave, they need to be able to supply their own generated power for a prolonged period of time, as batteries will run out prior to power restoration. They must be able to use oxygen concentrators, as tank re-supply may not be readily available. By law, FEMA cannot give generators to individuals, so individuals must prepare for themselves. In a hurricane-prone area where seasonal risk can be established, planning medication refills at the beginning of the season or giving a larger than normal supply may prove useful. In an area prone to sudden disaster, such as earthquake or tornado, then counseling patients to request refills at least 2 weeks early may be adequate.
Post-Disaster: The most reliable form of communication will be text. You likely already have text contacts for your staff and family members; add other providers, responders, planners, pharmacists, and oxygen suppliers to your text contacts. While you may wish to share a text point of contact with patients, understand that your ability to actually help during the initial disaster will likely be limited. Identify possible language translation needs and possible translators among your staff and/or friends as telephone services will be limited or absent following the disaster. Finally, identify your local emergency response planners on Facebook, Twitter, or other social media feeds. This will allow you to direct others to these sites for accurate information after the disaster.
Responder Recommendations: A single social media post can DESTROY your plans and hamper your efforts. Advertise a single contact point and an information resource (eg, bulletin board, webpage) early and often. Publicly and accurately declare the means by which people will access health care and health-care services, such as medications, dialysis, and oxygen. There will be nongovernment organizations (NGOs), friends, and other well-meaning individuals who will try to assist people in need through unconventional channels. Yet, by requesting assistance through nonroutine channels, those efforts tend to delay assistance, cause confusion, and/or squander resources. Continue to direct those requests through the established response channels, ie, the local 911 equivalent.
Plan to use cellular texts to communicate. While satellite telephones are great in concept, in execution, they are difficult to utilize when transmitting complex medical information. If you have an expansive budget, there are now devices available that allow for Iridium satellite-based text communications that require batteries but not intact cellular towers.
Facilities with electricity, water, oxygen, medications, laboratory testing, and CT scanners need to be identified and advertised within the responder community. If FEMA is involved, these resources will be identified and updated on a routine basis. The information will be distributed to their DMAT teams. Those DMAT teams will be distributed throughout the response area. Additionally, if the resources and budgeting are approved, then FEMA will also help re-establish medical transport, as well as Federal Medical Shelters (FMS). The FMS can temporarily house patients who can perform basic activities of daily living but require power, oxygen, or medication administration. For those patients in need of medications without insurance, FEMA may activate medication assistance through the Emergency Prescription Assistance Program. This will allow up to 30 days of medication to be distributed at no cost to the individual through participating pharmacies.
External responders will obviously need to pair with local providers/professionals who can navigate the system and, if necessary, can translate medical terms and care plans. Additionally, external responders will be targets for individuals looking to obtain resources for secondary gain or profit. Establishing a plan or consistently redirecting people to the appropriate resources for those needs may limit the inevitable damage these individuals will cause. Additionally, understand that the efficiencies of the modern society will be gone, and tasks will take much longer than expected. Even if you can communicate by text, the transporting of patients, delivering supplies, meeting with groups, and assessing sites will take far longer than you are used to when none of the stoplights are functional or if gasoline is in limited supply.
Finally, there will be patients for whom no solution, short of an intact, well-resourced medical system, exists—those with severe congenital issues, patients with advanced dementia, patients with advanced cancer, and those with multiple-antibiotic-resistant osteomyelitis are a few of the patients that this response encountered. If transport out of the area is unavailable, NGOs and other charities may be the best, and at times, the only resource for these patients. During this response, I observed NGO and charities helping individual patients and their families with their power, shelter, and medical needs that could not be legally provided by federal government response.
While I hope you may never need to use them, preparations for evacuation, medication, power, and communications before a potential disaster occurs will prove helpful to your patients. After the disaster, consistent and simple communications to the public will be necessary to limit the damage from the social media rumor mill. Working within the organized response framework and leveraging local knowledge and targeted NGO involvement will maximize the effect of your efforts.
In October 2017, in support of the Federal Emergency Management Agency’s response to assist the Governor and people of Puerto Rico, three Department of Defense (DOD) military hospital platforms were deployed; one each, by the US Army, Navy, and Air Force. They arrived on the island at different times with predominantly wartime surgical capabilities and augmented the Federal Emergency Management Agency (FEMA), US Public Health Service, National Guard, and Puerto Rico Department of Health efforts. My perspective is that of patient care and transport between the Centro Medico hospital complex in San Juan, the larger regional hospitals, the Veterans Administration hospital, the DOD response, FEMA Disaster Medical Assistance Teams (DMAT), and FEMA Federal Medical Shelters about 4 to 6 weeks after Hurricanes Maria and Irma struck. Based upon this experience, I would like to offer the following.
Pre-Disaster: All clinicians have a few patients that teeter “on the edge.” When basic services go away, these patients fall over that edge and become inpatients. Establish a list of patients who require oxygen and devices such as vests, cough-assist, or ventilation. If evacuation before the disaster is possible, those patients need to leave. If they refuse, or are unable to leave, they need to be able to supply their own generated power for a prolonged period of time, as batteries will run out prior to power restoration. They must be able to use oxygen concentrators, as tank re-supply may not be readily available. By law, FEMA cannot give generators to individuals, so individuals must prepare for themselves. In a hurricane-prone area where seasonal risk can be established, planning medication refills at the beginning of the season or giving a larger than normal supply may prove useful. In an area prone to sudden disaster, such as earthquake or tornado, then counseling patients to request refills at least 2 weeks early may be adequate.
Post-Disaster: The most reliable form of communication will be text. You likely already have text contacts for your staff and family members; add other providers, responders, planners, pharmacists, and oxygen suppliers to your text contacts. While you may wish to share a text point of contact with patients, understand that your ability to actually help during the initial disaster will likely be limited. Identify possible language translation needs and possible translators among your staff and/or friends as telephone services will be limited or absent following the disaster. Finally, identify your local emergency response planners on Facebook, Twitter, or other social media feeds. This will allow you to direct others to these sites for accurate information after the disaster.
Responder Recommendations: A single social media post can DESTROY your plans and hamper your efforts. Advertise a single contact point and an information resource (eg, bulletin board, webpage) early and often. Publicly and accurately declare the means by which people will access health care and health-care services, such as medications, dialysis, and oxygen. There will be nongovernment organizations (NGOs), friends, and other well-meaning individuals who will try to assist people in need through unconventional channels. Yet, by requesting assistance through nonroutine channels, those efforts tend to delay assistance, cause confusion, and/or squander resources. Continue to direct those requests through the established response channels, ie, the local 911 equivalent.
Plan to use cellular texts to communicate. While satellite telephones are great in concept, in execution, they are difficult to utilize when transmitting complex medical information. If you have an expansive budget, there are now devices available that allow for Iridium satellite-based text communications that require batteries but not intact cellular towers.
Facilities with electricity, water, oxygen, medications, laboratory testing, and CT scanners need to be identified and advertised within the responder community. If FEMA is involved, these resources will be identified and updated on a routine basis. The information will be distributed to their DMAT teams. Those DMAT teams will be distributed throughout the response area. Additionally, if the resources and budgeting are approved, then FEMA will also help re-establish medical transport, as well as Federal Medical Shelters (FMS). The FMS can temporarily house patients who can perform basic activities of daily living but require power, oxygen, or medication administration. For those patients in need of medications without insurance, FEMA may activate medication assistance through the Emergency Prescription Assistance Program. This will allow up to 30 days of medication to be distributed at no cost to the individual through participating pharmacies.
External responders will obviously need to pair with local providers/professionals who can navigate the system and, if necessary, can translate medical terms and care plans. Additionally, external responders will be targets for individuals looking to obtain resources for secondary gain or profit. Establishing a plan or consistently redirecting people to the appropriate resources for those needs may limit the inevitable damage these individuals will cause. Additionally, understand that the efficiencies of the modern society will be gone, and tasks will take much longer than expected. Even if you can communicate by text, the transporting of patients, delivering supplies, meeting with groups, and assessing sites will take far longer than you are used to when none of the stoplights are functional or if gasoline is in limited supply.
Finally, there will be patients for whom no solution, short of an intact, well-resourced medical system, exists—those with severe congenital issues, patients with advanced dementia, patients with advanced cancer, and those with multiple-antibiotic-resistant osteomyelitis are a few of the patients that this response encountered. If transport out of the area is unavailable, NGOs and other charities may be the best, and at times, the only resource for these patients. During this response, I observed NGO and charities helping individual patients and their families with their power, shelter, and medical needs that could not be legally provided by federal government response.
While I hope you may never need to use them, preparations for evacuation, medication, power, and communications before a potential disaster occurs will prove helpful to your patients. After the disaster, consistent and simple communications to the public will be necessary to limit the damage from the social media rumor mill. Working within the organized response framework and leveraging local knowledge and targeted NGO involvement will maximize the effect of your efforts.
In October 2017, in support of the Federal Emergency Management Agency’s response to assist the Governor and people of Puerto Rico, three Department of Defense (DOD) military hospital platforms were deployed; one each, by the US Army, Navy, and Air Force. They arrived on the island at different times with predominantly wartime surgical capabilities and augmented the Federal Emergency Management Agency (FEMA), US Public Health Service, National Guard, and Puerto Rico Department of Health efforts. My perspective is that of patient care and transport between the Centro Medico hospital complex in San Juan, the larger regional hospitals, the Veterans Administration hospital, the DOD response, FEMA Disaster Medical Assistance Teams (DMAT), and FEMA Federal Medical Shelters about 4 to 6 weeks after Hurricanes Maria and Irma struck. Based upon this experience, I would like to offer the following.
Pre-Disaster: All clinicians have a few patients that teeter “on the edge.” When basic services go away, these patients fall over that edge and become inpatients. Establish a list of patients who require oxygen and devices such as vests, cough-assist, or ventilation. If evacuation before the disaster is possible, those patients need to leave. If they refuse, or are unable to leave, they need to be able to supply their own generated power for a prolonged period of time, as batteries will run out prior to power restoration. They must be able to use oxygen concentrators, as tank re-supply may not be readily available. By law, FEMA cannot give generators to individuals, so individuals must prepare for themselves. In a hurricane-prone area where seasonal risk can be established, planning medication refills at the beginning of the season or giving a larger than normal supply may prove useful. In an area prone to sudden disaster, such as earthquake or tornado, then counseling patients to request refills at least 2 weeks early may be adequate.
Post-Disaster: The most reliable form of communication will be text. You likely already have text contacts for your staff and family members; add other providers, responders, planners, pharmacists, and oxygen suppliers to your text contacts. While you may wish to share a text point of contact with patients, understand that your ability to actually help during the initial disaster will likely be limited. Identify possible language translation needs and possible translators among your staff and/or friends as telephone services will be limited or absent following the disaster. Finally, identify your local emergency response planners on Facebook, Twitter, or other social media feeds. This will allow you to direct others to these sites for accurate information after the disaster.
Responder Recommendations: A single social media post can DESTROY your plans and hamper your efforts. Advertise a single contact point and an information resource (eg, bulletin board, webpage) early and often. Publicly and accurately declare the means by which people will access health care and health-care services, such as medications, dialysis, and oxygen. There will be nongovernment organizations (NGOs), friends, and other well-meaning individuals who will try to assist people in need through unconventional channels. Yet, by requesting assistance through nonroutine channels, those efforts tend to delay assistance, cause confusion, and/or squander resources. Continue to direct those requests through the established response channels, ie, the local 911 equivalent.
Plan to use cellular texts to communicate. While satellite telephones are great in concept, in execution, they are difficult to utilize when transmitting complex medical information. If you have an expansive budget, there are now devices available that allow for Iridium satellite-based text communications that require batteries but not intact cellular towers.
Facilities with electricity, water, oxygen, medications, laboratory testing, and CT scanners need to be identified and advertised within the responder community. If FEMA is involved, these resources will be identified and updated on a routine basis. The information will be distributed to their DMAT teams. Those DMAT teams will be distributed throughout the response area. Additionally, if the resources and budgeting are approved, then FEMA will also help re-establish medical transport, as well as Federal Medical Shelters (FMS). The FMS can temporarily house patients who can perform basic activities of daily living but require power, oxygen, or medication administration. For those patients in need of medications without insurance, FEMA may activate medication assistance through the Emergency Prescription Assistance Program. This will allow up to 30 days of medication to be distributed at no cost to the individual through participating pharmacies.
External responders will obviously need to pair with local providers/professionals who can navigate the system and, if necessary, can translate medical terms and care plans. Additionally, external responders will be targets for individuals looking to obtain resources for secondary gain or profit. Establishing a plan or consistently redirecting people to the appropriate resources for those needs may limit the inevitable damage these individuals will cause. Additionally, understand that the efficiencies of the modern society will be gone, and tasks will take much longer than expected. Even if you can communicate by text, the transporting of patients, delivering supplies, meeting with groups, and assessing sites will take far longer than you are used to when none of the stoplights are functional or if gasoline is in limited supply.
Finally, there will be patients for whom no solution, short of an intact, well-resourced medical system, exists—those with severe congenital issues, patients with advanced dementia, patients with advanced cancer, and those with multiple-antibiotic-resistant osteomyelitis are a few of the patients that this response encountered. If transport out of the area is unavailable, NGOs and other charities may be the best, and at times, the only resource for these patients. During this response, I observed NGO and charities helping individual patients and their families with their power, shelter, and medical needs that could not be legally provided by federal government response.
While I hope you may never need to use them, preparations for evacuation, medication, power, and communications before a potential disaster occurs will prove helpful to your patients. After the disaster, consistent and simple communications to the public will be necessary to limit the damage from the social media rumor mill. Working within the organized response framework and leveraging local knowledge and targeted NGO involvement will maximize the effect of your efforts.
Five things to do around the convention center at CHEST 2018
Planning to attend CHEST 2018? We know you’re always on the go, so we’ve come up with a few quick things to do in San Antonio without having to go more than a few blocks outside of the convention center.
Whataburger
While some may be hardcore In-N-Out fans, there’s another well known burger joint in Texas with a location that happens to be next to the convention center on E Commerce St. Head on over to Whataburger and experience what the company calls a “bigger, better burger.”
San Antonio Riverwalk
Want to experience the San Antonio, Texas atmosphere but don’t have time for a long excursion? The Henry B. Gonzalez Convention Center is a few steps away from the Riverwalk, which winds throughout the city. Off of the northwest corner of the convention center, take a stroll and experience the picturesque beauty of the San Antonio river, the restaurants, and the bright colorful surroundings.
La Villita Historic Arts Village
Interested in art? Interested in architecture? La Villita, located on the west side of the convention center on S Alamo St, is on the US government’s National Register of Historic Places as a Historic District. Take a look at different architectural styles, like adobe, early Victorian, and Texas vernacular limestone buildings. You’ll find markers throughout La Villita with information about each building’s history. You’ll also find local artists, custom art, and unique dining options.
Tower of the Americas
Exit the south end of the convention center to go to the Tower of the Americas for a spectacular view of the city. This 750-foot tall tower has an observation deck, revolving restaurant with panoramic views, a stationary bar, and a 4D theater adventure ride great for the whole family. This is a great stop for lunch, dinner, or a nice afternoon activity.
The Alamo
Lastly, if you have an hour to spare, take a tour of the Alamo that commemorates the 1836 siege and battle. There are free and ticketed activities, including audio or guided tours (ticketed) or history talks, visiting the Alamo Church, exhibitions, and more! Don’t forget to stop at the gift shop for a souvenir or two to take home.
Planning to attend CHEST 2018? We know you’re always on the go, so we’ve come up with a few quick things to do in San Antonio without having to go more than a few blocks outside of the convention center.
Whataburger
While some may be hardcore In-N-Out fans, there’s another well known burger joint in Texas with a location that happens to be next to the convention center on E Commerce St. Head on over to Whataburger and experience what the company calls a “bigger, better burger.”
San Antonio Riverwalk
Want to experience the San Antonio, Texas atmosphere but don’t have time for a long excursion? The Henry B. Gonzalez Convention Center is a few steps away from the Riverwalk, which winds throughout the city. Off of the northwest corner of the convention center, take a stroll and experience the picturesque beauty of the San Antonio river, the restaurants, and the bright colorful surroundings.
La Villita Historic Arts Village
Interested in art? Interested in architecture? La Villita, located on the west side of the convention center on S Alamo St, is on the US government’s National Register of Historic Places as a Historic District. Take a look at different architectural styles, like adobe, early Victorian, and Texas vernacular limestone buildings. You’ll find markers throughout La Villita with information about each building’s history. You’ll also find local artists, custom art, and unique dining options.
Tower of the Americas
Exit the south end of the convention center to go to the Tower of the Americas for a spectacular view of the city. This 750-foot tall tower has an observation deck, revolving restaurant with panoramic views, a stationary bar, and a 4D theater adventure ride great for the whole family. This is a great stop for lunch, dinner, or a nice afternoon activity.
The Alamo
Lastly, if you have an hour to spare, take a tour of the Alamo that commemorates the 1836 siege and battle. There are free and ticketed activities, including audio or guided tours (ticketed) or history talks, visiting the Alamo Church, exhibitions, and more! Don’t forget to stop at the gift shop for a souvenir or two to take home.
Planning to attend CHEST 2018? We know you’re always on the go, so we’ve come up with a few quick things to do in San Antonio without having to go more than a few blocks outside of the convention center.
Whataburger
While some may be hardcore In-N-Out fans, there’s another well known burger joint in Texas with a location that happens to be next to the convention center on E Commerce St. Head on over to Whataburger and experience what the company calls a “bigger, better burger.”
San Antonio Riverwalk
Want to experience the San Antonio, Texas atmosphere but don’t have time for a long excursion? The Henry B. Gonzalez Convention Center is a few steps away from the Riverwalk, which winds throughout the city. Off of the northwest corner of the convention center, take a stroll and experience the picturesque beauty of the San Antonio river, the restaurants, and the bright colorful surroundings.
La Villita Historic Arts Village
Interested in art? Interested in architecture? La Villita, located on the west side of the convention center on S Alamo St, is on the US government’s National Register of Historic Places as a Historic District. Take a look at different architectural styles, like adobe, early Victorian, and Texas vernacular limestone buildings. You’ll find markers throughout La Villita with information about each building’s history. You’ll also find local artists, custom art, and unique dining options.
Tower of the Americas
Exit the south end of the convention center to go to the Tower of the Americas for a spectacular view of the city. This 750-foot tall tower has an observation deck, revolving restaurant with panoramic views, a stationary bar, and a 4D theater adventure ride great for the whole family. This is a great stop for lunch, dinner, or a nice afternoon activity.
The Alamo
Lastly, if you have an hour to spare, take a tour of the Alamo that commemorates the 1836 siege and battle. There are free and ticketed activities, including audio or guided tours (ticketed) or history talks, visiting the Alamo Church, exhibitions, and more! Don’t forget to stop at the gift shop for a souvenir or two to take home.
Medical marijuana’s ‘Catch-22’: Federal limits on research hinder patients’ relief
By the time Ann Marie Owen turned to marijuana to treat her pain, she was struggling to walk and talk. She also hallucinated.
For 4 years, her doctor prescribed the 61-year-old a wide range of opioids for her transverse myelitis, a debilitating disease that caused pain, muscle weakness, and paralysis.
The drugs not only failed to ease her symptoms, they also hooked her.
When her home state of New York legalized marijuana for the treatment of select medical ailments, Ms. Owens decided it was time to swap pills for pot. But her doctors refused to help.
“Even though medical marijuana is legal, none of my doctors were willing to talk to me about it,” she said. “They just kept telling me to take opioids.”
While 29 states have legalized marijuana to treat pain and other ailments, the growing number of Americans like Ms. Owen who use marijuana and the doctors who treat them are caught in the middle of a conflict in federal and state laws – a predicament that is only worsened by thin scientific data.
Because the federal government classifies marijuana a Schedule 1 drug – by definition a substance with no currently accepted medical use and a high potential for abuse – research on marijuana or its active ingredients is highly restricted and even discouraged in some cases.
Underscoring the federal government’s position, Health & Human Services Secretary Alex Azar recently pronounced that there was “no such thing as medical marijuana.”
Scientists say that stance prevents them from conducting the high-quality research required for Food and Drug Administration approval, even as some early research indicates marijuana might be a promising alternative to opioids or other medicines.
Patients and physicians, meanwhile, lack guidance when making decisions about medical treatment for an array of serious conditions.
“We have the federal government and the state governments driving a hundred miles an hour in the opposite direction when they should be coming together to obtain more scientific data,” said Orrin Devinsky, MD, who is researching the effects of cannabidiol, an active ingredient of marijuana, on epilepsy. “It’s like saying in 1960, ‘We’re not going to the moon because no one agrees how to get there.’ ”
The problem stems partly from the fact that the federal government’s restrictive marijuana research policies have not been overhauled in more than 40 years, researchers say.
Only one federal government contractor grows marijuana for federally funded research. Researchers complain the pot grown by the contractor at the University of Mississippi is inadequate for high-quality studies.
The marijuana, which comes in a micronized powder form, is less potent than the pot offered at dispensaries, researchers say. It also differs from other products offered at dispensaries, such as so-called edibles, which are eaten like snacks. The difference makes it difficult to compare the real-life effects of the marijuana compounds.
Researchers also face time-consuming and costly hurdles in completing the complicated federal application process for using marijuana in long-term clinical trials.
“It’s public policy before science,” said Chinazo Cunningham, MD, a primary care doctor who is the lead investigator on one of the few federally funded studies exploring marijuana as a treatment for pain. “The federal government’s policies really make it much more difficult.”
Dr. Cunningham, who received a 5-year, $3.8 million federal grant, will not be administering marijuana directly to participants. Instead, she will follow 250 HIV-positive and HIV-negative adults with chronic pain who use opioids and have been certified to get medical marijuana from a dispensary.
“It’s a Catch-22,” said Dr. Cunningham, who is with the Albert Einstein College of Medicine, New York. “We’re going to be looking at all of these issues – age, disease, level of pain – but when we’re done, there’s the danger that people are going to say ‘Oh, it’s anecdotal’ or that it’s inherently flawed because it’s not a randomized trial.’’
Without clear answers, hospitals, doctors, and patients are left to their own devices, which can result in poor treatment and needless suffering.
Hospitals and other medical facilities have to decide what to do with newly hospitalized patients who normally take medical marijuana at home.
Some have a “don’t ask, don’t tell” approach, said Dr. Devinsky, who sometimes advises his patients to use it. Others ban its use and substitute opioids or other prescriptions.
Young adults, for instance, have had to stop taking cannabidiol compounds for their epilepsy because they’re in federally funded group homes, said Dr. Devinsky, the director of New York University’s Langone Comprehensive Epilepsy Center.
“These kids end up getting seizures again,” he said. “This whole situation has created a hodgepodge of insanity.”
The Trump administration, however, has resisted policy changes.
Last year, the Drug Enforcement Administration had been gearing up to allow facilities other than the University of Mississippi to grow pot for research. But after the DEA received 26 applications from other growers, Attorney General Jeff Sessions halted the initiative.
The Department of Veterans Affairs also recently announced it would not fund studies of using marijuana compounds to treat ailments such as pain.
The DEA and HHS have cited concerns about medical supervision, addiction, and a lack of “well-controlled studies proving efficacy.”
Patients, meanwhile, forge ahead.
While experts say they don’t know exactly how many older Americans rely on marijuana for medicinal purposes, the number of Americans aged 65 years and older who say they are using the drug skyrocketed 250% from 2006 to 2013.
Some patients turn to friends, patient advocacy groups, or online support groups for information.
Ms. Owen, for one, kept searching for a doctor and eventually found a neurologist willing to certify her to use marijuana and advise her on what to take.
“It’s saved my life,” said the retired university administrative assistant who credited marijuana for weaning her off opioids. “It not only helps my pain, but I can think, walk, and talk again.”
Mary Jo, a Minnesotan, was afraid of being identified as a medical marijuana user, even though she now helps friends navigate the process and it’s legal in her home state.
“There’s still a stigma,” said Mary Jo, who found it effective for treating her pain from a nerve condition. “Nobody helps you figure it out, so you kind of play around with it on your own.”
Still, doctors and scientists worry about the implications of such experimentation.
In a sweeping report last year, the National Academies of Sciences, Engineering and Medicine called on the federal government to support better research, decrying the “lack of definitive evidence on using medical marijuana.”
The national academies’ committee reviewed more than 10,000 scientific abstracts related to the topic. It made 100 conclusions based on its review, including finding evidence that marijuana relieves pain and chemotherapy-induced nausea. But it found “inadequate information” to support or refute effects on Parkinson’s disease.
Yet those who find that medical marijuana helps them can become fierce advocates no matter what their doctors say.
Caryl Barrett, a 54-year-old who lives in Georgia, said she decided to travel out of state to Colorado to treat her pain from her transverse myelitis and the autoimmune disease neurosarcoidosis.
“I realized it worked and I decided to bring it back with me,” she said. “I broke federal law.”
Georgia, meanwhile, permitted limited medicinal use of marijuana but did not set up dispensaries. As a result, patients resort to ordering it online or driving to another state to get it.
The conflict in the law makes her uneasy. But Ms. Barrett, who had been on opioids for a decade, said she feels so strongly about it working that “if someone wants to arrest me, bring it on.”
Others experience mixed results.
Melodie Beckham, who had metastatic lung cancer, tried medical marijuana for 13 days in a clinical trial at Connecticut Hospice before deciding to quit.
“She was hopeful that it would help her relax and just kind of enjoy those days,” said her daughter, Laura Beckham.
Instead, it seemed to make her mother, who died in July at age 69, “a little more agitated or more paranoid.”
The marijuana “didn’t seem effective,” nor did it keep her mother from hitting her pain pump to get extra doses of an opioid, her daughter said.
The researchers running the trial at Connecticut Hospice spent 2 years getting necessary approvals from the FDA, the National Institute on Drug Abuse (NIDA), and the DEA.
Since May, the trial has enrolled only 7 of the 66 patients it plans to sign up because many patients were too sick, too close to death, or simply couldn’t swallow the pills. So far, the trial has shown “mixed results,” said James Prota, director of pharmacy for the hospice.
Researchers point out they are still exploring the basics when it comes to marijuana’s effects on older adults or the terminally ill.
“We just have no data on how many older adults are using medical marijuana, what they are using it for, and most importantly, what are the outcomes,” said Brian Kaskie, an associate professor at the University of Iowa’s College of Public Health in Iowa City. “It’s all anecdotal.”
Mr. Kaskie, who specializes in public policy and the aging, received grants from the state of Colorado and the Chicago-based Retirement Research Foundation to survey the use of medical marijuana by older Americans.
In many quarters, there’s a growing appetite for solid information, he said.
“When I first started this, my colleagues joked we were going to find all the aging hippies who listen to the Grateful Dead,” said Mr. Kaskie, who has been studying medicinal marijuana for years. “Now, they’re starting to realize this is a legitimate area of research.”
Twenty researchers received marijuana from the federal program last year, which was more than any previous year since 2010, according to NIDA statistics.
In a recent funding announcement, the National Institutes of Health requested grant applications to study the effects of marijuana and other drugs on older adults and pain. NIH, however, continues to funnel much of its funding into studying the adverse effects of marijuana, researchers said.
Although NIH acknowledged in one of the announcements that some research supports “possible benefits” of marijuana, it emphasized “there have not been adequate large controlled trials to support these claims.”
Kaiser Health News (KHN) is a national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
By the time Ann Marie Owen turned to marijuana to treat her pain, she was struggling to walk and talk. She also hallucinated.
For 4 years, her doctor prescribed the 61-year-old a wide range of opioids for her transverse myelitis, a debilitating disease that caused pain, muscle weakness, and paralysis.
The drugs not only failed to ease her symptoms, they also hooked her.
When her home state of New York legalized marijuana for the treatment of select medical ailments, Ms. Owens decided it was time to swap pills for pot. But her doctors refused to help.
“Even though medical marijuana is legal, none of my doctors were willing to talk to me about it,” she said. “They just kept telling me to take opioids.”
While 29 states have legalized marijuana to treat pain and other ailments, the growing number of Americans like Ms. Owen who use marijuana and the doctors who treat them are caught in the middle of a conflict in federal and state laws – a predicament that is only worsened by thin scientific data.
Because the federal government classifies marijuana a Schedule 1 drug – by definition a substance with no currently accepted medical use and a high potential for abuse – research on marijuana or its active ingredients is highly restricted and even discouraged in some cases.
Underscoring the federal government’s position, Health & Human Services Secretary Alex Azar recently pronounced that there was “no such thing as medical marijuana.”
Scientists say that stance prevents them from conducting the high-quality research required for Food and Drug Administration approval, even as some early research indicates marijuana might be a promising alternative to opioids or other medicines.
Patients and physicians, meanwhile, lack guidance when making decisions about medical treatment for an array of serious conditions.
“We have the federal government and the state governments driving a hundred miles an hour in the opposite direction when they should be coming together to obtain more scientific data,” said Orrin Devinsky, MD, who is researching the effects of cannabidiol, an active ingredient of marijuana, on epilepsy. “It’s like saying in 1960, ‘We’re not going to the moon because no one agrees how to get there.’ ”
The problem stems partly from the fact that the federal government’s restrictive marijuana research policies have not been overhauled in more than 40 years, researchers say.
Only one federal government contractor grows marijuana for federally funded research. Researchers complain the pot grown by the contractor at the University of Mississippi is inadequate for high-quality studies.
The marijuana, which comes in a micronized powder form, is less potent than the pot offered at dispensaries, researchers say. It also differs from other products offered at dispensaries, such as so-called edibles, which are eaten like snacks. The difference makes it difficult to compare the real-life effects of the marijuana compounds.
Researchers also face time-consuming and costly hurdles in completing the complicated federal application process for using marijuana in long-term clinical trials.
“It’s public policy before science,” said Chinazo Cunningham, MD, a primary care doctor who is the lead investigator on one of the few federally funded studies exploring marijuana as a treatment for pain. “The federal government’s policies really make it much more difficult.”
Dr. Cunningham, who received a 5-year, $3.8 million federal grant, will not be administering marijuana directly to participants. Instead, she will follow 250 HIV-positive and HIV-negative adults with chronic pain who use opioids and have been certified to get medical marijuana from a dispensary.
“It’s a Catch-22,” said Dr. Cunningham, who is with the Albert Einstein College of Medicine, New York. “We’re going to be looking at all of these issues – age, disease, level of pain – but when we’re done, there’s the danger that people are going to say ‘Oh, it’s anecdotal’ or that it’s inherently flawed because it’s not a randomized trial.’’
Without clear answers, hospitals, doctors, and patients are left to their own devices, which can result in poor treatment and needless suffering.
Hospitals and other medical facilities have to decide what to do with newly hospitalized patients who normally take medical marijuana at home.
Some have a “don’t ask, don’t tell” approach, said Dr. Devinsky, who sometimes advises his patients to use it. Others ban its use and substitute opioids or other prescriptions.
Young adults, for instance, have had to stop taking cannabidiol compounds for their epilepsy because they’re in federally funded group homes, said Dr. Devinsky, the director of New York University’s Langone Comprehensive Epilepsy Center.
“These kids end up getting seizures again,” he said. “This whole situation has created a hodgepodge of insanity.”
The Trump administration, however, has resisted policy changes.
Last year, the Drug Enforcement Administration had been gearing up to allow facilities other than the University of Mississippi to grow pot for research. But after the DEA received 26 applications from other growers, Attorney General Jeff Sessions halted the initiative.
The Department of Veterans Affairs also recently announced it would not fund studies of using marijuana compounds to treat ailments such as pain.
The DEA and HHS have cited concerns about medical supervision, addiction, and a lack of “well-controlled studies proving efficacy.”
Patients, meanwhile, forge ahead.
While experts say they don’t know exactly how many older Americans rely on marijuana for medicinal purposes, the number of Americans aged 65 years and older who say they are using the drug skyrocketed 250% from 2006 to 2013.
Some patients turn to friends, patient advocacy groups, or online support groups for information.
Ms. Owen, for one, kept searching for a doctor and eventually found a neurologist willing to certify her to use marijuana and advise her on what to take.
“It’s saved my life,” said the retired university administrative assistant who credited marijuana for weaning her off opioids. “It not only helps my pain, but I can think, walk, and talk again.”
Mary Jo, a Minnesotan, was afraid of being identified as a medical marijuana user, even though she now helps friends navigate the process and it’s legal in her home state.
“There’s still a stigma,” said Mary Jo, who found it effective for treating her pain from a nerve condition. “Nobody helps you figure it out, so you kind of play around with it on your own.”
Still, doctors and scientists worry about the implications of such experimentation.
In a sweeping report last year, the National Academies of Sciences, Engineering and Medicine called on the federal government to support better research, decrying the “lack of definitive evidence on using medical marijuana.”
The national academies’ committee reviewed more than 10,000 scientific abstracts related to the topic. It made 100 conclusions based on its review, including finding evidence that marijuana relieves pain and chemotherapy-induced nausea. But it found “inadequate information” to support or refute effects on Parkinson’s disease.
Yet those who find that medical marijuana helps them can become fierce advocates no matter what their doctors say.
Caryl Barrett, a 54-year-old who lives in Georgia, said she decided to travel out of state to Colorado to treat her pain from her transverse myelitis and the autoimmune disease neurosarcoidosis.
“I realized it worked and I decided to bring it back with me,” she said. “I broke federal law.”
Georgia, meanwhile, permitted limited medicinal use of marijuana but did not set up dispensaries. As a result, patients resort to ordering it online or driving to another state to get it.
The conflict in the law makes her uneasy. But Ms. Barrett, who had been on opioids for a decade, said she feels so strongly about it working that “if someone wants to arrest me, bring it on.”
Others experience mixed results.
Melodie Beckham, who had metastatic lung cancer, tried medical marijuana for 13 days in a clinical trial at Connecticut Hospice before deciding to quit.
“She was hopeful that it would help her relax and just kind of enjoy those days,” said her daughter, Laura Beckham.
Instead, it seemed to make her mother, who died in July at age 69, “a little more agitated or more paranoid.”
The marijuana “didn’t seem effective,” nor did it keep her mother from hitting her pain pump to get extra doses of an opioid, her daughter said.
The researchers running the trial at Connecticut Hospice spent 2 years getting necessary approvals from the FDA, the National Institute on Drug Abuse (NIDA), and the DEA.
Since May, the trial has enrolled only 7 of the 66 patients it plans to sign up because many patients were too sick, too close to death, or simply couldn’t swallow the pills. So far, the trial has shown “mixed results,” said James Prota, director of pharmacy for the hospice.
Researchers point out they are still exploring the basics when it comes to marijuana’s effects on older adults or the terminally ill.
“We just have no data on how many older adults are using medical marijuana, what they are using it for, and most importantly, what are the outcomes,” said Brian Kaskie, an associate professor at the University of Iowa’s College of Public Health in Iowa City. “It’s all anecdotal.”
Mr. Kaskie, who specializes in public policy and the aging, received grants from the state of Colorado and the Chicago-based Retirement Research Foundation to survey the use of medical marijuana by older Americans.
In many quarters, there’s a growing appetite for solid information, he said.
“When I first started this, my colleagues joked we were going to find all the aging hippies who listen to the Grateful Dead,” said Mr. Kaskie, who has been studying medicinal marijuana for years. “Now, they’re starting to realize this is a legitimate area of research.”
Twenty researchers received marijuana from the federal program last year, which was more than any previous year since 2010, according to NIDA statistics.
In a recent funding announcement, the National Institutes of Health requested grant applications to study the effects of marijuana and other drugs on older adults and pain. NIH, however, continues to funnel much of its funding into studying the adverse effects of marijuana, researchers said.
Although NIH acknowledged in one of the announcements that some research supports “possible benefits” of marijuana, it emphasized “there have not been adequate large controlled trials to support these claims.”
Kaiser Health News (KHN) is a national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.
By the time Ann Marie Owen turned to marijuana to treat her pain, she was struggling to walk and talk. She also hallucinated.
For 4 years, her doctor prescribed the 61-year-old a wide range of opioids for her transverse myelitis, a debilitating disease that caused pain, muscle weakness, and paralysis.
The drugs not only failed to ease her symptoms, they also hooked her.
When her home state of New York legalized marijuana for the treatment of select medical ailments, Ms. Owens decided it was time to swap pills for pot. But her doctors refused to help.
“Even though medical marijuana is legal, none of my doctors were willing to talk to me about it,” she said. “They just kept telling me to take opioids.”
While 29 states have legalized marijuana to treat pain and other ailments, the growing number of Americans like Ms. Owen who use marijuana and the doctors who treat them are caught in the middle of a conflict in federal and state laws – a predicament that is only worsened by thin scientific data.
Because the federal government classifies marijuana a Schedule 1 drug – by definition a substance with no currently accepted medical use and a high potential for abuse – research on marijuana or its active ingredients is highly restricted and even discouraged in some cases.
Underscoring the federal government’s position, Health & Human Services Secretary Alex Azar recently pronounced that there was “no such thing as medical marijuana.”
Scientists say that stance prevents them from conducting the high-quality research required for Food and Drug Administration approval, even as some early research indicates marijuana might be a promising alternative to opioids or other medicines.
Patients and physicians, meanwhile, lack guidance when making decisions about medical treatment for an array of serious conditions.
“We have the federal government and the state governments driving a hundred miles an hour in the opposite direction when they should be coming together to obtain more scientific data,” said Orrin Devinsky, MD, who is researching the effects of cannabidiol, an active ingredient of marijuana, on epilepsy. “It’s like saying in 1960, ‘We’re not going to the moon because no one agrees how to get there.’ ”
The problem stems partly from the fact that the federal government’s restrictive marijuana research policies have not been overhauled in more than 40 years, researchers say.
Only one federal government contractor grows marijuana for federally funded research. Researchers complain the pot grown by the contractor at the University of Mississippi is inadequate for high-quality studies.
The marijuana, which comes in a micronized powder form, is less potent than the pot offered at dispensaries, researchers say. It also differs from other products offered at dispensaries, such as so-called edibles, which are eaten like snacks. The difference makes it difficult to compare the real-life effects of the marijuana compounds.
Researchers also face time-consuming and costly hurdles in completing the complicated federal application process for using marijuana in long-term clinical trials.
“It’s public policy before science,” said Chinazo Cunningham, MD, a primary care doctor who is the lead investigator on one of the few federally funded studies exploring marijuana as a treatment for pain. “The federal government’s policies really make it much more difficult.”
Dr. Cunningham, who received a 5-year, $3.8 million federal grant, will not be administering marijuana directly to participants. Instead, she will follow 250 HIV-positive and HIV-negative adults with chronic pain who use opioids and have been certified to get medical marijuana from a dispensary.
“It’s a Catch-22,” said Dr. Cunningham, who is with the Albert Einstein College of Medicine, New York. “We’re going to be looking at all of these issues – age, disease, level of pain – but when we’re done, there’s the danger that people are going to say ‘Oh, it’s anecdotal’ or that it’s inherently flawed because it’s not a randomized trial.’’
Without clear answers, hospitals, doctors, and patients are left to their own devices, which can result in poor treatment and needless suffering.
Hospitals and other medical facilities have to decide what to do with newly hospitalized patients who normally take medical marijuana at home.
Some have a “don’t ask, don’t tell” approach, said Dr. Devinsky, who sometimes advises his patients to use it. Others ban its use and substitute opioids or other prescriptions.
Young adults, for instance, have had to stop taking cannabidiol compounds for their epilepsy because they’re in federally funded group homes, said Dr. Devinsky, the director of New York University’s Langone Comprehensive Epilepsy Center.
“These kids end up getting seizures again,” he said. “This whole situation has created a hodgepodge of insanity.”
The Trump administration, however, has resisted policy changes.
Last year, the Drug Enforcement Administration had been gearing up to allow facilities other than the University of Mississippi to grow pot for research. But after the DEA received 26 applications from other growers, Attorney General Jeff Sessions halted the initiative.
The Department of Veterans Affairs also recently announced it would not fund studies of using marijuana compounds to treat ailments such as pain.
The DEA and HHS have cited concerns about medical supervision, addiction, and a lack of “well-controlled studies proving efficacy.”
Patients, meanwhile, forge ahead.
While experts say they don’t know exactly how many older Americans rely on marijuana for medicinal purposes, the number of Americans aged 65 years and older who say they are using the drug skyrocketed 250% from 2006 to 2013.
Some patients turn to friends, patient advocacy groups, or online support groups for information.
Ms. Owen, for one, kept searching for a doctor and eventually found a neurologist willing to certify her to use marijuana and advise her on what to take.
“It’s saved my life,” said the retired university administrative assistant who credited marijuana for weaning her off opioids. “It not only helps my pain, but I can think, walk, and talk again.”
Mary Jo, a Minnesotan, was afraid of being identified as a medical marijuana user, even though she now helps friends navigate the process and it’s legal in her home state.
“There’s still a stigma,” said Mary Jo, who found it effective for treating her pain from a nerve condition. “Nobody helps you figure it out, so you kind of play around with it on your own.”
Still, doctors and scientists worry about the implications of such experimentation.
In a sweeping report last year, the National Academies of Sciences, Engineering and Medicine called on the federal government to support better research, decrying the “lack of definitive evidence on using medical marijuana.”
The national academies’ committee reviewed more than 10,000 scientific abstracts related to the topic. It made 100 conclusions based on its review, including finding evidence that marijuana relieves pain and chemotherapy-induced nausea. But it found “inadequate information” to support or refute effects on Parkinson’s disease.
Yet those who find that medical marijuana helps them can become fierce advocates no matter what their doctors say.
Caryl Barrett, a 54-year-old who lives in Georgia, said she decided to travel out of state to Colorado to treat her pain from her transverse myelitis and the autoimmune disease neurosarcoidosis.
“I realized it worked and I decided to bring it back with me,” she said. “I broke federal law.”
Georgia, meanwhile, permitted limited medicinal use of marijuana but did not set up dispensaries. As a result, patients resort to ordering it online or driving to another state to get it.
The conflict in the law makes her uneasy. But Ms. Barrett, who had been on opioids for a decade, said she feels so strongly about it working that “if someone wants to arrest me, bring it on.”
Others experience mixed results.
Melodie Beckham, who had metastatic lung cancer, tried medical marijuana for 13 days in a clinical trial at Connecticut Hospice before deciding to quit.
“She was hopeful that it would help her relax and just kind of enjoy those days,” said her daughter, Laura Beckham.
Instead, it seemed to make her mother, who died in July at age 69, “a little more agitated or more paranoid.”
The marijuana “didn’t seem effective,” nor did it keep her mother from hitting her pain pump to get extra doses of an opioid, her daughter said.
The researchers running the trial at Connecticut Hospice spent 2 years getting necessary approvals from the FDA, the National Institute on Drug Abuse (NIDA), and the DEA.
Since May, the trial has enrolled only 7 of the 66 patients it plans to sign up because many patients were too sick, too close to death, or simply couldn’t swallow the pills. So far, the trial has shown “mixed results,” said James Prota, director of pharmacy for the hospice.
Researchers point out they are still exploring the basics when it comes to marijuana’s effects on older adults or the terminally ill.
“We just have no data on how many older adults are using medical marijuana, what they are using it for, and most importantly, what are the outcomes,” said Brian Kaskie, an associate professor at the University of Iowa’s College of Public Health in Iowa City. “It’s all anecdotal.”
Mr. Kaskie, who specializes in public policy and the aging, received grants from the state of Colorado and the Chicago-based Retirement Research Foundation to survey the use of medical marijuana by older Americans.
In many quarters, there’s a growing appetite for solid information, he said.
“When I first started this, my colleagues joked we were going to find all the aging hippies who listen to the Grateful Dead,” said Mr. Kaskie, who has been studying medicinal marijuana for years. “Now, they’re starting to realize this is a legitimate area of research.”
Twenty researchers received marijuana from the federal program last year, which was more than any previous year since 2010, according to NIDA statistics.
In a recent funding announcement, the National Institutes of Health requested grant applications to study the effects of marijuana and other drugs on older adults and pain. NIH, however, continues to funnel much of its funding into studying the adverse effects of marijuana, researchers said.
Although NIH acknowledged in one of the announcements that some research supports “possible benefits” of marijuana, it emphasized “there have not been adequate large controlled trials to support these claims.”
Kaiser Health News (KHN) is a national health policy news service. It is an editorially independent program of the Henry J. Kaiser Family Foundation that is not affiliated with Kaiser Permanente.