User login
New CLTI Global Guidelines Available
On May 31, new global guidelines on the best ways to treat Chronic Limb-Threatening Ischemia were co-published in the Journal of Vascular Surgery and the European Journal of Vascular and Endovascular Surgery. This comes after four years of collaboration between vascular experts from around the world. According to the SVS’ own Dr. Conte, a co-editor, the group created a unique practice guideline that reflects the spectrum of the diseases and the approaches seen worldwide. Read the guidelines in the JVS here.
On May 31, new global guidelines on the best ways to treat Chronic Limb-Threatening Ischemia were co-published in the Journal of Vascular Surgery and the European Journal of Vascular and Endovascular Surgery. This comes after four years of collaboration between vascular experts from around the world. According to the SVS’ own Dr. Conte, a co-editor, the group created a unique practice guideline that reflects the spectrum of the diseases and the approaches seen worldwide. Read the guidelines in the JVS here.
On May 31, new global guidelines on the best ways to treat Chronic Limb-Threatening Ischemia were co-published in the Journal of Vascular Surgery and the European Journal of Vascular and Endovascular Surgery. This comes after four years of collaboration between vascular experts from around the world. According to the SVS’ own Dr. Conte, a co-editor, the group created a unique practice guideline that reflects the spectrum of the diseases and the approaches seen worldwide. Read the guidelines in the JVS here.
Tailored intervention improves asthma self-management for older patients
A needs- and barriers-based intervention that addressed psychosocial, physical, cognitive, and environmental barriers to self-management of asthma for older adults was successful in improving asthma outcomes and management, a recent trial has shown.
“This study demonstrates the value of patient centeredness and care coaching in supporting older adults with asthma and for ongoing efforts to engage patients in care delivery design and personalization,” Alex D. Federman, MD, of the division of general internal medicine at Icahn School of Medicine at Mount Sinai, New York, and colleagues wrote in their study, which was published in JAMA Internal Medicine. “It also highlights the challenges of engaging vulnerable populations in self-management support, including modest retention rates and reduced impact over time despite repeated encounters designed to sustain its effects.”
The researchers said older adults often have difficulty with self-management tasks like inhaler technique and use of inhaled corticosteroids, which can be caused by various psychosocial, physical, cognitive, or environmental barriers. However, an attempt at creating self-management tools around specific problems, rather than generalized training, has not been traditionally attempted, they noted.
For the SAMBA trial, Dr. Federman and colleagues enrolled 391 patients who were randomized to receive a home-based intervention, clinic-based intervention, or usual care, where an asthma care coach would identify the barriers to asthma control, train the patient in areas of improvement, and provide reinforcement when necessary. Patients were at least age 60 years (15.1% men) with uncontrolled asthma in New York City and were enrolled between February 2014 and December 2017. Researchers used the Mini Asthma Quality of Life Questionnaire, Asthma Control Test, metered dose inhaler technique, Medication Adherence Rating Scale, and visits to the emergency room to assess outcomes between interventions and usual care, and between home and clinic care. The data was analyzed using the ‘difference in differences’ statistical technique to compare the change differential between the groups.
They found significantly better asthma control scores between the intervention group and the control groups at 3 months (difference-in-differences, 1.2; 95% confidence interval, 0.2-2.2; P = .02), 6 months (D-in-Ds, 1.0; 95% CI, 0.0-2.1; P = .049), and 12 months (D-inDs, 0.6; 95% CI, −0.5 to 1.8; P = .28). Quality of life was significantly improved in the intervention group, compared with control patients (overall effect, chi-squared = 10.5; with 4 degrees of freedom; P = .01), as was adherence to medication (overall effect, chi-squared = 9.5, with 4 degrees of freedom; P = .049), and inhaler technique as measured by correctly completed steps at 12 months (75% vs. 58%). Visits to the emergency room were also lower in the intervention group, compared with the control group (6.2% vs. 12.7%; adjusted odds ratio, 0.8; 95% CI, 0.6-0.99; both P = .03). The researchers noted there were no significant differences between home care and clinic care.
Potential limitations in the study included a lower-than-planned statistical power, 70% retention in the intervention arms, low generalizability of the findings, and lack of blinding on the part of research assistants as well as some improvement in asthma control and outcomes in the control group.
This study was funded in part by the Patient-Centered Outcomes Research Institute. Coauthors Nandini Shroff reported grants from the Patient-Centered Outcomes Research Institute; Michael S. Wolf reported grants from Eli Lilly; and Juan P. Wisnivesky reported personal fees from Sanofi, Quintiles, and Banook, and grants from Sanofi and Quorum. The other authors reported no relevant conflicts of interest.
SOURCE: Federman AD et al. JAMA Intern Med. 2019; doi: 10.1001/jamainternmed.2019.1201.
A needs- and barriers-based intervention that addressed psychosocial, physical, cognitive, and environmental barriers to self-management of asthma for older adults was successful in improving asthma outcomes and management, a recent trial has shown.
“This study demonstrates the value of patient centeredness and care coaching in supporting older adults with asthma and for ongoing efforts to engage patients in care delivery design and personalization,” Alex D. Federman, MD, of the division of general internal medicine at Icahn School of Medicine at Mount Sinai, New York, and colleagues wrote in their study, which was published in JAMA Internal Medicine. “It also highlights the challenges of engaging vulnerable populations in self-management support, including modest retention rates and reduced impact over time despite repeated encounters designed to sustain its effects.”
The researchers said older adults often have difficulty with self-management tasks like inhaler technique and use of inhaled corticosteroids, which can be caused by various psychosocial, physical, cognitive, or environmental barriers. However, an attempt at creating self-management tools around specific problems, rather than generalized training, has not been traditionally attempted, they noted.
For the SAMBA trial, Dr. Federman and colleagues enrolled 391 patients who were randomized to receive a home-based intervention, clinic-based intervention, or usual care, where an asthma care coach would identify the barriers to asthma control, train the patient in areas of improvement, and provide reinforcement when necessary. Patients were at least age 60 years (15.1% men) with uncontrolled asthma in New York City and were enrolled between February 2014 and December 2017. Researchers used the Mini Asthma Quality of Life Questionnaire, Asthma Control Test, metered dose inhaler technique, Medication Adherence Rating Scale, and visits to the emergency room to assess outcomes between interventions and usual care, and between home and clinic care. The data was analyzed using the ‘difference in differences’ statistical technique to compare the change differential between the groups.
They found significantly better asthma control scores between the intervention group and the control groups at 3 months (difference-in-differences, 1.2; 95% confidence interval, 0.2-2.2; P = .02), 6 months (D-in-Ds, 1.0; 95% CI, 0.0-2.1; P = .049), and 12 months (D-inDs, 0.6; 95% CI, −0.5 to 1.8; P = .28). Quality of life was significantly improved in the intervention group, compared with control patients (overall effect, chi-squared = 10.5; with 4 degrees of freedom; P = .01), as was adherence to medication (overall effect, chi-squared = 9.5, with 4 degrees of freedom; P = .049), and inhaler technique as measured by correctly completed steps at 12 months (75% vs. 58%). Visits to the emergency room were also lower in the intervention group, compared with the control group (6.2% vs. 12.7%; adjusted odds ratio, 0.8; 95% CI, 0.6-0.99; both P = .03). The researchers noted there were no significant differences between home care and clinic care.
Potential limitations in the study included a lower-than-planned statistical power, 70% retention in the intervention arms, low generalizability of the findings, and lack of blinding on the part of research assistants as well as some improvement in asthma control and outcomes in the control group.
This study was funded in part by the Patient-Centered Outcomes Research Institute. Coauthors Nandini Shroff reported grants from the Patient-Centered Outcomes Research Institute; Michael S. Wolf reported grants from Eli Lilly; and Juan P. Wisnivesky reported personal fees from Sanofi, Quintiles, and Banook, and grants from Sanofi and Quorum. The other authors reported no relevant conflicts of interest.
SOURCE: Federman AD et al. JAMA Intern Med. 2019; doi: 10.1001/jamainternmed.2019.1201.
A needs- and barriers-based intervention that addressed psychosocial, physical, cognitive, and environmental barriers to self-management of asthma for older adults was successful in improving asthma outcomes and management, a recent trial has shown.
“This study demonstrates the value of patient centeredness and care coaching in supporting older adults with asthma and for ongoing efforts to engage patients in care delivery design and personalization,” Alex D. Federman, MD, of the division of general internal medicine at Icahn School of Medicine at Mount Sinai, New York, and colleagues wrote in their study, which was published in JAMA Internal Medicine. “It also highlights the challenges of engaging vulnerable populations in self-management support, including modest retention rates and reduced impact over time despite repeated encounters designed to sustain its effects.”
The researchers said older adults often have difficulty with self-management tasks like inhaler technique and use of inhaled corticosteroids, which can be caused by various psychosocial, physical, cognitive, or environmental barriers. However, an attempt at creating self-management tools around specific problems, rather than generalized training, has not been traditionally attempted, they noted.
For the SAMBA trial, Dr. Federman and colleagues enrolled 391 patients who were randomized to receive a home-based intervention, clinic-based intervention, or usual care, where an asthma care coach would identify the barriers to asthma control, train the patient in areas of improvement, and provide reinforcement when necessary. Patients were at least age 60 years (15.1% men) with uncontrolled asthma in New York City and were enrolled between February 2014 and December 2017. Researchers used the Mini Asthma Quality of Life Questionnaire, Asthma Control Test, metered dose inhaler technique, Medication Adherence Rating Scale, and visits to the emergency room to assess outcomes between interventions and usual care, and between home and clinic care. The data was analyzed using the ‘difference in differences’ statistical technique to compare the change differential between the groups.
They found significantly better asthma control scores between the intervention group and the control groups at 3 months (difference-in-differences, 1.2; 95% confidence interval, 0.2-2.2; P = .02), 6 months (D-in-Ds, 1.0; 95% CI, 0.0-2.1; P = .049), and 12 months (D-inDs, 0.6; 95% CI, −0.5 to 1.8; P = .28). Quality of life was significantly improved in the intervention group, compared with control patients (overall effect, chi-squared = 10.5; with 4 degrees of freedom; P = .01), as was adherence to medication (overall effect, chi-squared = 9.5, with 4 degrees of freedom; P = .049), and inhaler technique as measured by correctly completed steps at 12 months (75% vs. 58%). Visits to the emergency room were also lower in the intervention group, compared with the control group (6.2% vs. 12.7%; adjusted odds ratio, 0.8; 95% CI, 0.6-0.99; both P = .03). The researchers noted there were no significant differences between home care and clinic care.
Potential limitations in the study included a lower-than-planned statistical power, 70% retention in the intervention arms, low generalizability of the findings, and lack of blinding on the part of research assistants as well as some improvement in asthma control and outcomes in the control group.
This study was funded in part by the Patient-Centered Outcomes Research Institute. Coauthors Nandini Shroff reported grants from the Patient-Centered Outcomes Research Institute; Michael S. Wolf reported grants from Eli Lilly; and Juan P. Wisnivesky reported personal fees from Sanofi, Quintiles, and Banook, and grants from Sanofi and Quorum. The other authors reported no relevant conflicts of interest.
SOURCE: Federman AD et al. JAMA Intern Med. 2019; doi: 10.1001/jamainternmed.2019.1201.
FROM JAMA INTERNAL MEDICINE
Induced seizures as effective as spontaneous in identifying epileptic generator
according to a study of patients with focal drug-resistant epilepsy.
“This finding might lead to a more time-efficient intracranial presurgical investigation of focal epilepsy by reducing the need to record spontaneous seizures,” wrote Carolina Cuello Oderiz, MD, formerly of McGill University, and her coauthors. The study was published in JAMA Neurology.
To determine if cortical stimulation-induced seizures and subsequent removal of the informed seizure-onset zone (SOZ) could lead to good surgical outcomes, the researchers selected 103 patients with focal drug-resistant epilepsy who underwent stereoelectroencephalography (SEEG). All participants had to have undergone cortical stimulation during SEEG, followed by open epilepsy surgical procedure with a minimum 1-year follow-up. In addition, complete brain imaging for exact localization of individual electrode contacts and resection cavity was also required.
Of the 103 patients, 59 (57.3%) had cortical stimulation-induced seizures. The percentage of these patients in the good outcome group was higher than in the poor outcome group (70.5% versus 47.5%). The median percentage of resected cortical stimulation-informed SOZ contacts was also higher in the good than in the poor outcome group (63.2% versus 33.3%). The results were similar for spontaneous seizures, where the median percentage of resected contacts of the spontaneous SOZ was 57.1% in the good outcome group and 32.7% in the poor outcome group.
The coauthors noted their study’s limitations, including the exclusion of many patients due to the need for hi-resolution neuroimaging and sufficient postsurgical imaging and follow-up. They added that the strict criteria were “key to the main outcome of this study,” however, and noted that generalizability of the data was supported by similar rates in excluded patients.
The study was supported by grants from the Canadian Institute of Health Research and the Savoy Epilepsy Foundation. Numerous authors reported receiving grants, personal fees, and other funding from organizations like the Montreal Neurological Institute and various pharmaceutical companies.
Dr. Cuello Oderiz is now at SUNY Upstate Medical University, Syracuse, N.Y.
SOURCE: Cuello Oderiz C et al. JAMA Neurol. 2019 Jun 10. doi: 10.1001/jamaneurol.2019.1464.
according to a study of patients with focal drug-resistant epilepsy.
“This finding might lead to a more time-efficient intracranial presurgical investigation of focal epilepsy by reducing the need to record spontaneous seizures,” wrote Carolina Cuello Oderiz, MD, formerly of McGill University, and her coauthors. The study was published in JAMA Neurology.
To determine if cortical stimulation-induced seizures and subsequent removal of the informed seizure-onset zone (SOZ) could lead to good surgical outcomes, the researchers selected 103 patients with focal drug-resistant epilepsy who underwent stereoelectroencephalography (SEEG). All participants had to have undergone cortical stimulation during SEEG, followed by open epilepsy surgical procedure with a minimum 1-year follow-up. In addition, complete brain imaging for exact localization of individual electrode contacts and resection cavity was also required.
Of the 103 patients, 59 (57.3%) had cortical stimulation-induced seizures. The percentage of these patients in the good outcome group was higher than in the poor outcome group (70.5% versus 47.5%). The median percentage of resected cortical stimulation-informed SOZ contacts was also higher in the good than in the poor outcome group (63.2% versus 33.3%). The results were similar for spontaneous seizures, where the median percentage of resected contacts of the spontaneous SOZ was 57.1% in the good outcome group and 32.7% in the poor outcome group.
The coauthors noted their study’s limitations, including the exclusion of many patients due to the need for hi-resolution neuroimaging and sufficient postsurgical imaging and follow-up. They added that the strict criteria were “key to the main outcome of this study,” however, and noted that generalizability of the data was supported by similar rates in excluded patients.
The study was supported by grants from the Canadian Institute of Health Research and the Savoy Epilepsy Foundation. Numerous authors reported receiving grants, personal fees, and other funding from organizations like the Montreal Neurological Institute and various pharmaceutical companies.
Dr. Cuello Oderiz is now at SUNY Upstate Medical University, Syracuse, N.Y.
SOURCE: Cuello Oderiz C et al. JAMA Neurol. 2019 Jun 10. doi: 10.1001/jamaneurol.2019.1464.
according to a study of patients with focal drug-resistant epilepsy.
“This finding might lead to a more time-efficient intracranial presurgical investigation of focal epilepsy by reducing the need to record spontaneous seizures,” wrote Carolina Cuello Oderiz, MD, formerly of McGill University, and her coauthors. The study was published in JAMA Neurology.
To determine if cortical stimulation-induced seizures and subsequent removal of the informed seizure-onset zone (SOZ) could lead to good surgical outcomes, the researchers selected 103 patients with focal drug-resistant epilepsy who underwent stereoelectroencephalography (SEEG). All participants had to have undergone cortical stimulation during SEEG, followed by open epilepsy surgical procedure with a minimum 1-year follow-up. In addition, complete brain imaging for exact localization of individual electrode contacts and resection cavity was also required.
Of the 103 patients, 59 (57.3%) had cortical stimulation-induced seizures. The percentage of these patients in the good outcome group was higher than in the poor outcome group (70.5% versus 47.5%). The median percentage of resected cortical stimulation-informed SOZ contacts was also higher in the good than in the poor outcome group (63.2% versus 33.3%). The results were similar for spontaneous seizures, where the median percentage of resected contacts of the spontaneous SOZ was 57.1% in the good outcome group and 32.7% in the poor outcome group.
The coauthors noted their study’s limitations, including the exclusion of many patients due to the need for hi-resolution neuroimaging and sufficient postsurgical imaging and follow-up. They added that the strict criteria were “key to the main outcome of this study,” however, and noted that generalizability of the data was supported by similar rates in excluded patients.
The study was supported by grants from the Canadian Institute of Health Research and the Savoy Epilepsy Foundation. Numerous authors reported receiving grants, personal fees, and other funding from organizations like the Montreal Neurological Institute and various pharmaceutical companies.
Dr. Cuello Oderiz is now at SUNY Upstate Medical University, Syracuse, N.Y.
SOURCE: Cuello Oderiz C et al. JAMA Neurol. 2019 Jun 10. doi: 10.1001/jamaneurol.2019.1464.
FROM JAMA NEUROLOGY
Key clinical point: Seizures induced by cortical stimulation and spontaneous seizures both led to a similar percentage of good surgical outcomes in patients with epilepsy.
Major finding: The percentage of patients who received cortical stimulation-induced seizures in the good outcome group was higher than in the poor outcome group (70.5% versus 47.5%).
Study details: A cohort study of 103 patients with focal drug-resistant epilepsy who underwent stereoelectroencephalography.
Disclosures: The study was supported by grants from the Canadian Institute of Health Research and the Savoy Epilepsy Foundation. Numerous authors reported receiving grants, personal fees, and other funding from organizations like the Montreal Neurological Institute and various pharmaceutical companies.
Source: Cuello Oderiz C et al. JAMA Neurol. 2019 Jun 10. doi: 10.1001/jamaneurol.2019.1464.
How effective is spironolactone for treating resistant hypertension?
EVIDENCE SUMMARY
A 2017 meta-analysis of 4 RCTs (869 patients) evaluated the effectiveness of prescribing spironolactone for patients with resistant hypertension, defined as above-goal blood pressure (BP) despite treatment with at least 3 BP-lowering drugs (at least 1 of which was a diuretic).1 All 4 trials compared spironolactone 25 to 50 mg/d with placebo. Follow-up periods ranged from 8 to 16 weeks. The primary outcomes were systolic and diastolic BPs, which were evaluated in the office, at home, or with an ambulatory monitor.
Spironolactone markedly lowers systolic and diastolic BP
A statistically significant reduction in SBP occurred in the spironolactone group compared with the placebo group (weighted mean difference [WMD] = −16.7 mm Hg; 95% confidence interval [CI], −27.5 to −5.8 mm Hg). DBP also decreased (WMD = −6.11 mm Hg; 95% CI, −9.34 to −2.88 mm Hg).
Because significant heterogeneity was found in the initial pooled results (I2 = 96% for SBP; I2 = 85% for DBP), investigators performed an analysis that excluded a single study with a small sample size. The re-analysis continued to show significant reductions in SBP and DBP for spironolactone compared with placebo (SBP: WMD = −10.8 mm Hg; 95% CI, −13.16 to −8.43 mm Hg; DBP: WMD = −4.62 mm Hg; 95% CI, −6.05 to −3.2 mm Hg; I2 = 35%), confirming that the excluded trial was the source of heterogeneity in the initial analysis and that spironolactone continued to significantly lower BP for the treatment group compared with controls.
Add-on treatment with spironolactone also reduces BP
A 2016 meta-analysis of 5 RCTs with a total of 553 patients examined the effectiveness of add-on treatment with spironolactone (25-50 mg/d) for patients with resistant hypertension, defined as failure to achieve BP < 140/90 mm Hg despite treatment with 3 or more BP-lowering drugs, including one diuretic.2 Spironolactone was compared with placebo in 4 trials and with ramipril in the remaining study. The follow-up periods were 8 to 16 weeks. Researchers separated BP outcomes into 24-hour ambulatory systolic/diastolic BPs and office systolic/diastolic BPs.
The 24-hour ambulatory BPs were significantly lower in the spironolactone group compared with the control group (24-hour SBP: WMD = −10.5 mm Hg; 95% CI, −12.3 to −8.71 mm Hg; 24-hour DBP: WMD = −4.09 mm Hg; 95% CI, −5.28 to −2.91 mm Hg). No significant heterogeneity was noted in these analyses.
Office-based BPs also were markedly reduced in spironolactone groups compared with controls (office SBP: WMD = −17 mm Hg; 95% CI, −25 to −8.95 mm Hg); office DBP: WMD = −6.18 mm Hg; 95% CI, −9.3 to −3.05 mm Hg). Because the office-based BP data showed significant heterogeneity (I2 = 94% for SBP and 84.2% for DBP), 2 studies determined to be of lower quality caused by lack of detailed methodology were excluded from analysis, yielding continued statistically significant reductions in SBP (WMD = −11.7 mm Hg; 95% CI, −14.4 to −8.95 mm Hg) and DBP (WMD = −4.07 mm Hg; 95% CI, −5.6 to −2.54 mm Hg) compared with controls. Heterogeneity also decreased when the 2 studies were excluded (I2 = 21% for SBP and I2 = 59% for DBP).
How spironolactone compares with alternative drugs
A 2017 meta-analysis of 5 RCTs with 662 patients evaluated the effectiveness of spironolactone (25-50 mg/d) on resistant hypertension in patients taking 3 medications compared with a control group—placebo in 3 trials, placebo or bisoprolol (5-10 mg) in 1 trial, and an alternative treatment (candesartan 8 mg, atenolol 100 mg, or alpha methyldopa 750 mg) in 1 trial.3 Follow-up periods ranged from 4 to 16 weeks. Researchers evaluated changes in office and 24-hour ambulatory or home BP and completed separate analyses of pooled data for spironolactone compared with placebo groups, and spironolactone compared with alternative treatment groups.
Continue to: Investigators found a statistically significant...
Investigators found a statistically significant reduction in office SBP and DBP among patients taking spironolactone compared with control groups (SBP: WMD = −15.7 mm Hg; 95% CI, −20.5 to −11 mm Hg; DBP: WMD = −6.21 mm Hg; 95% CI, −8.33 to −4.1 mm Hg). A significant decrease also occurred in 24-hour ambulatory home SBP and DBP (SBP: MD = −8.7 mm Hg; 95% CI, −8.79 to −8.62 mm Hg; DBP: WMD = −4.12 mm Hg; 95% CI, −4.48 to −3.75 mm Hg).
Patients treated with spironolactone showed a marked decrease in home SBP compared with alternative drug groups (WMD = −4.5 mm Hg; 95% CI, −4.63 to −4.37 mm Hg), but alternative drugs reduced home DBP significantly more than spironolactone (WMD = 0.6 mm Hg; 95% CI, 0.55-0.65 mm Hg). Marked heterogeneity was found in these analyses, and the authors also noted that reductions in SBP are more clinically relevant than decreases in DBP.
RECOMMENDATIONS
The 2017 American Heart Association/American College of Cardiology evidence-based guideline recommends considering adding a mineralocorticoid receptor agonist to treatment regimens for resistant hypertension when: office BP remains ≥ 130/80 mm Hg; the patient is prescribed at least 3 antihypertensive agents at optimal doses including a diuretic; pseudoresistance (nonadherence, inaccurate measurements) is excluded; reversible lifestyle factors have been addressed; substances that interfere with BP treatment (such as nonsteroidal anti-inflammatory drugs and oral contraceptive pills) are excluded; and screening for secondary causes of hypertension is complete.4
The United Kingdom’s National Institute for Health and Care Excellence (NICE) evidence-based guideline recommends considering spironolactone 25 mg/d to treat resistant hypertension if the patient’s potassium level is 4.5 mmol/L or lower and BP is higher than 140/90 mm Hg despite treatment with an optimal or best-tolerated dose of an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker plus a calcium-channel blocker and diuretic.5
Editor’s takeaway
The evidence from multiple RCTs convincingly shows the effectiveness of spironolactone. Despite the SOR of C because of a disease-oriented outcome, we do treat to blood pressure goals, and therefore, spironolactone is a good option.
1. Zhao D, Liu H, Dong P, et al. A meta-analysis of add-on use of spironolactone in patients with resistant hypertension. Int J Cardiol. 2017;233:113-117.
2. Wang C, Xiong B, Huang J. Efficacy and safety of spironolactone in patients with resistant hypertension: a meta-analysis of randomised controlled trials. Heart Lung Circ. 2016;25:1021-1030.
3. Liu L, Xu B, Ju Y. Addition of spironolactone in patients with resistant hypertension: a meta-analysis of randomized controlled trials. Clin Exp Hypertens. 2017;39:257-263.
4. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2017. https://doi.org/10.1161/HYP.0000000000000065. Accessed June 6, 2019.
5. National Institute for Health and Care Excellence. Hypertension in adults: diagnosis and management. Clinical guideline [CG127]. August 2011. https://www.nice.org.uk/guidance/cg127/chapter/1-guidance#initiating-and-monitoring-antihypertensive-drug-treatment-including-blood-pressure-targets-2. Accessed June 6, 2019.
EVIDENCE SUMMARY
A 2017 meta-analysis of 4 RCTs (869 patients) evaluated the effectiveness of prescribing spironolactone for patients with resistant hypertension, defined as above-goal blood pressure (BP) despite treatment with at least 3 BP-lowering drugs (at least 1 of which was a diuretic).1 All 4 trials compared spironolactone 25 to 50 mg/d with placebo. Follow-up periods ranged from 8 to 16 weeks. The primary outcomes were systolic and diastolic BPs, which were evaluated in the office, at home, or with an ambulatory monitor.
Spironolactone markedly lowers systolic and diastolic BP
A statistically significant reduction in SBP occurred in the spironolactone group compared with the placebo group (weighted mean difference [WMD] = −16.7 mm Hg; 95% confidence interval [CI], −27.5 to −5.8 mm Hg). DBP also decreased (WMD = −6.11 mm Hg; 95% CI, −9.34 to −2.88 mm Hg).
Because significant heterogeneity was found in the initial pooled results (I2 = 96% for SBP; I2 = 85% for DBP), investigators performed an analysis that excluded a single study with a small sample size. The re-analysis continued to show significant reductions in SBP and DBP for spironolactone compared with placebo (SBP: WMD = −10.8 mm Hg; 95% CI, −13.16 to −8.43 mm Hg; DBP: WMD = −4.62 mm Hg; 95% CI, −6.05 to −3.2 mm Hg; I2 = 35%), confirming that the excluded trial was the source of heterogeneity in the initial analysis and that spironolactone continued to significantly lower BP for the treatment group compared with controls.
Add-on treatment with spironolactone also reduces BP
A 2016 meta-analysis of 5 RCTs with a total of 553 patients examined the effectiveness of add-on treatment with spironolactone (25-50 mg/d) for patients with resistant hypertension, defined as failure to achieve BP < 140/90 mm Hg despite treatment with 3 or more BP-lowering drugs, including one diuretic.2 Spironolactone was compared with placebo in 4 trials and with ramipril in the remaining study. The follow-up periods were 8 to 16 weeks. Researchers separated BP outcomes into 24-hour ambulatory systolic/diastolic BPs and office systolic/diastolic BPs.
The 24-hour ambulatory BPs were significantly lower in the spironolactone group compared with the control group (24-hour SBP: WMD = −10.5 mm Hg; 95% CI, −12.3 to −8.71 mm Hg; 24-hour DBP: WMD = −4.09 mm Hg; 95% CI, −5.28 to −2.91 mm Hg). No significant heterogeneity was noted in these analyses.
Office-based BPs also were markedly reduced in spironolactone groups compared with controls (office SBP: WMD = −17 mm Hg; 95% CI, −25 to −8.95 mm Hg); office DBP: WMD = −6.18 mm Hg; 95% CI, −9.3 to −3.05 mm Hg). Because the office-based BP data showed significant heterogeneity (I2 = 94% for SBP and 84.2% for DBP), 2 studies determined to be of lower quality caused by lack of detailed methodology were excluded from analysis, yielding continued statistically significant reductions in SBP (WMD = −11.7 mm Hg; 95% CI, −14.4 to −8.95 mm Hg) and DBP (WMD = −4.07 mm Hg; 95% CI, −5.6 to −2.54 mm Hg) compared with controls. Heterogeneity also decreased when the 2 studies were excluded (I2 = 21% for SBP and I2 = 59% for DBP).
How spironolactone compares with alternative drugs
A 2017 meta-analysis of 5 RCTs with 662 patients evaluated the effectiveness of spironolactone (25-50 mg/d) on resistant hypertension in patients taking 3 medications compared with a control group—placebo in 3 trials, placebo or bisoprolol (5-10 mg) in 1 trial, and an alternative treatment (candesartan 8 mg, atenolol 100 mg, or alpha methyldopa 750 mg) in 1 trial.3 Follow-up periods ranged from 4 to 16 weeks. Researchers evaluated changes in office and 24-hour ambulatory or home BP and completed separate analyses of pooled data for spironolactone compared with placebo groups, and spironolactone compared with alternative treatment groups.
Continue to: Investigators found a statistically significant...
Investigators found a statistically significant reduction in office SBP and DBP among patients taking spironolactone compared with control groups (SBP: WMD = −15.7 mm Hg; 95% CI, −20.5 to −11 mm Hg; DBP: WMD = −6.21 mm Hg; 95% CI, −8.33 to −4.1 mm Hg). A significant decrease also occurred in 24-hour ambulatory home SBP and DBP (SBP: MD = −8.7 mm Hg; 95% CI, −8.79 to −8.62 mm Hg; DBP: WMD = −4.12 mm Hg; 95% CI, −4.48 to −3.75 mm Hg).
Patients treated with spironolactone showed a marked decrease in home SBP compared with alternative drug groups (WMD = −4.5 mm Hg; 95% CI, −4.63 to −4.37 mm Hg), but alternative drugs reduced home DBP significantly more than spironolactone (WMD = 0.6 mm Hg; 95% CI, 0.55-0.65 mm Hg). Marked heterogeneity was found in these analyses, and the authors also noted that reductions in SBP are more clinically relevant than decreases in DBP.
RECOMMENDATIONS
The 2017 American Heart Association/American College of Cardiology evidence-based guideline recommends considering adding a mineralocorticoid receptor agonist to treatment regimens for resistant hypertension when: office BP remains ≥ 130/80 mm Hg; the patient is prescribed at least 3 antihypertensive agents at optimal doses including a diuretic; pseudoresistance (nonadherence, inaccurate measurements) is excluded; reversible lifestyle factors have been addressed; substances that interfere with BP treatment (such as nonsteroidal anti-inflammatory drugs and oral contraceptive pills) are excluded; and screening for secondary causes of hypertension is complete.4
The United Kingdom’s National Institute for Health and Care Excellence (NICE) evidence-based guideline recommends considering spironolactone 25 mg/d to treat resistant hypertension if the patient’s potassium level is 4.5 mmol/L or lower and BP is higher than 140/90 mm Hg despite treatment with an optimal or best-tolerated dose of an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker plus a calcium-channel blocker and diuretic.5
Editor’s takeaway
The evidence from multiple RCTs convincingly shows the effectiveness of spironolactone. Despite the SOR of C because of a disease-oriented outcome, we do treat to blood pressure goals, and therefore, spironolactone is a good option.
EVIDENCE SUMMARY
A 2017 meta-analysis of 4 RCTs (869 patients) evaluated the effectiveness of prescribing spironolactone for patients with resistant hypertension, defined as above-goal blood pressure (BP) despite treatment with at least 3 BP-lowering drugs (at least 1 of which was a diuretic).1 All 4 trials compared spironolactone 25 to 50 mg/d with placebo. Follow-up periods ranged from 8 to 16 weeks. The primary outcomes were systolic and diastolic BPs, which were evaluated in the office, at home, or with an ambulatory monitor.
Spironolactone markedly lowers systolic and diastolic BP
A statistically significant reduction in SBP occurred in the spironolactone group compared with the placebo group (weighted mean difference [WMD] = −16.7 mm Hg; 95% confidence interval [CI], −27.5 to −5.8 mm Hg). DBP also decreased (WMD = −6.11 mm Hg; 95% CI, −9.34 to −2.88 mm Hg).
Because significant heterogeneity was found in the initial pooled results (I2 = 96% for SBP; I2 = 85% for DBP), investigators performed an analysis that excluded a single study with a small sample size. The re-analysis continued to show significant reductions in SBP and DBP for spironolactone compared with placebo (SBP: WMD = −10.8 mm Hg; 95% CI, −13.16 to −8.43 mm Hg; DBP: WMD = −4.62 mm Hg; 95% CI, −6.05 to −3.2 mm Hg; I2 = 35%), confirming that the excluded trial was the source of heterogeneity in the initial analysis and that spironolactone continued to significantly lower BP for the treatment group compared with controls.
Add-on treatment with spironolactone also reduces BP
A 2016 meta-analysis of 5 RCTs with a total of 553 patients examined the effectiveness of add-on treatment with spironolactone (25-50 mg/d) for patients with resistant hypertension, defined as failure to achieve BP < 140/90 mm Hg despite treatment with 3 or more BP-lowering drugs, including one diuretic.2 Spironolactone was compared with placebo in 4 trials and with ramipril in the remaining study. The follow-up periods were 8 to 16 weeks. Researchers separated BP outcomes into 24-hour ambulatory systolic/diastolic BPs and office systolic/diastolic BPs.
The 24-hour ambulatory BPs were significantly lower in the spironolactone group compared with the control group (24-hour SBP: WMD = −10.5 mm Hg; 95% CI, −12.3 to −8.71 mm Hg; 24-hour DBP: WMD = −4.09 mm Hg; 95% CI, −5.28 to −2.91 mm Hg). No significant heterogeneity was noted in these analyses.
Office-based BPs also were markedly reduced in spironolactone groups compared with controls (office SBP: WMD = −17 mm Hg; 95% CI, −25 to −8.95 mm Hg); office DBP: WMD = −6.18 mm Hg; 95% CI, −9.3 to −3.05 mm Hg). Because the office-based BP data showed significant heterogeneity (I2 = 94% for SBP and 84.2% for DBP), 2 studies determined to be of lower quality caused by lack of detailed methodology were excluded from analysis, yielding continued statistically significant reductions in SBP (WMD = −11.7 mm Hg; 95% CI, −14.4 to −8.95 mm Hg) and DBP (WMD = −4.07 mm Hg; 95% CI, −5.6 to −2.54 mm Hg) compared with controls. Heterogeneity also decreased when the 2 studies were excluded (I2 = 21% for SBP and I2 = 59% for DBP).
How spironolactone compares with alternative drugs
A 2017 meta-analysis of 5 RCTs with 662 patients evaluated the effectiveness of spironolactone (25-50 mg/d) on resistant hypertension in patients taking 3 medications compared with a control group—placebo in 3 trials, placebo or bisoprolol (5-10 mg) in 1 trial, and an alternative treatment (candesartan 8 mg, atenolol 100 mg, or alpha methyldopa 750 mg) in 1 trial.3 Follow-up periods ranged from 4 to 16 weeks. Researchers evaluated changes in office and 24-hour ambulatory or home BP and completed separate analyses of pooled data for spironolactone compared with placebo groups, and spironolactone compared with alternative treatment groups.
Continue to: Investigators found a statistically significant...
Investigators found a statistically significant reduction in office SBP and DBP among patients taking spironolactone compared with control groups (SBP: WMD = −15.7 mm Hg; 95% CI, −20.5 to −11 mm Hg; DBP: WMD = −6.21 mm Hg; 95% CI, −8.33 to −4.1 mm Hg). A significant decrease also occurred in 24-hour ambulatory home SBP and DBP (SBP: MD = −8.7 mm Hg; 95% CI, −8.79 to −8.62 mm Hg; DBP: WMD = −4.12 mm Hg; 95% CI, −4.48 to −3.75 mm Hg).
Patients treated with spironolactone showed a marked decrease in home SBP compared with alternative drug groups (WMD = −4.5 mm Hg; 95% CI, −4.63 to −4.37 mm Hg), but alternative drugs reduced home DBP significantly more than spironolactone (WMD = 0.6 mm Hg; 95% CI, 0.55-0.65 mm Hg). Marked heterogeneity was found in these analyses, and the authors also noted that reductions in SBP are more clinically relevant than decreases in DBP.
RECOMMENDATIONS
The 2017 American Heart Association/American College of Cardiology evidence-based guideline recommends considering adding a mineralocorticoid receptor agonist to treatment regimens for resistant hypertension when: office BP remains ≥ 130/80 mm Hg; the patient is prescribed at least 3 antihypertensive agents at optimal doses including a diuretic; pseudoresistance (nonadherence, inaccurate measurements) is excluded; reversible lifestyle factors have been addressed; substances that interfere with BP treatment (such as nonsteroidal anti-inflammatory drugs and oral contraceptive pills) are excluded; and screening for secondary causes of hypertension is complete.4
The United Kingdom’s National Institute for Health and Care Excellence (NICE) evidence-based guideline recommends considering spironolactone 25 mg/d to treat resistant hypertension if the patient’s potassium level is 4.5 mmol/L or lower and BP is higher than 140/90 mm Hg despite treatment with an optimal or best-tolerated dose of an angiotensin-converting enzyme inhibitor or angiotensin II receptor blocker plus a calcium-channel blocker and diuretic.5
Editor’s takeaway
The evidence from multiple RCTs convincingly shows the effectiveness of spironolactone. Despite the SOR of C because of a disease-oriented outcome, we do treat to blood pressure goals, and therefore, spironolactone is a good option.
1. Zhao D, Liu H, Dong P, et al. A meta-analysis of add-on use of spironolactone in patients with resistant hypertension. Int J Cardiol. 2017;233:113-117.
2. Wang C, Xiong B, Huang J. Efficacy and safety of spironolactone in patients with resistant hypertension: a meta-analysis of randomised controlled trials. Heart Lung Circ. 2016;25:1021-1030.
3. Liu L, Xu B, Ju Y. Addition of spironolactone in patients with resistant hypertension: a meta-analysis of randomized controlled trials. Clin Exp Hypertens. 2017;39:257-263.
4. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2017. https://doi.org/10.1161/HYP.0000000000000065. Accessed June 6, 2019.
5. National Institute for Health and Care Excellence. Hypertension in adults: diagnosis and management. Clinical guideline [CG127]. August 2011. https://www.nice.org.uk/guidance/cg127/chapter/1-guidance#initiating-and-monitoring-antihypertensive-drug-treatment-including-blood-pressure-targets-2. Accessed June 6, 2019.
1. Zhao D, Liu H, Dong P, et al. A meta-analysis of add-on use of spironolactone in patients with resistant hypertension. Int J Cardiol. 2017;233:113-117.
2. Wang C, Xiong B, Huang J. Efficacy and safety of spironolactone in patients with resistant hypertension: a meta-analysis of randomised controlled trials. Heart Lung Circ. 2016;25:1021-1030.
3. Liu L, Xu B, Ju Y. Addition of spironolactone in patients with resistant hypertension: a meta-analysis of randomized controlled trials. Clin Exp Hypertens. 2017;39:257-263.
4. Whelton PK, Carey RM, Aronow WS, et al. 2017 ACC/AHA/AAPA/ABC/ACPM/AGS/APhA/ASH/ASPC/NMA/PCNA Guideline for the prevention, detection, evaluation, and management of high blood pressure in adults: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. Hypertension. 2017. https://doi.org/10.1161/HYP.0000000000000065. Accessed June 6, 2019.
5. National Institute for Health and Care Excellence. Hypertension in adults: diagnosis and management. Clinical guideline [CG127]. August 2011. https://www.nice.org.uk/guidance/cg127/chapter/1-guidance#initiating-and-monitoring-antihypertensive-drug-treatment-including-blood-pressure-targets-2. Accessed June 6, 2019.
EVIDENCE-BASED ANSWER:
Very effective. Spironolactone reduces systolic blood pressure (SBP) by 11 to 17 mm Hg and diastolic blood pressure (DBP) by up to 6 mm Hg in patients with resistant hypertension taking 3 or more medications (strength of recommendation [SOR]: C, meta-analysis of randomized controlled trials [RCTs] of disease-oriented evidence).
4-year-old girl • genital discomfort and dysuria • clitoral hood swelling • Blood blister on the labia minora • Dx?
THE CASE
A 4-year-old girl presented to her pediatrician with genital discomfort and dysuria of 6 months’ duration. The patient’s mother said that 3 days earlier, she noticed a tear near the child’s clitoris and a scab on the labia minora that the mother attributed to minor trauma from scratching. The pediatrician was concerned about genital trauma from sexual abuse and referred the patient to the emergency department, where a report with child protective services (CPS) was filed. The mother reported that the patient and her 8-year-old sibling spent 3 to 4 hours a day with a babysitter, who was always supervised, and the parents had no concerns about possible sexual abuse.
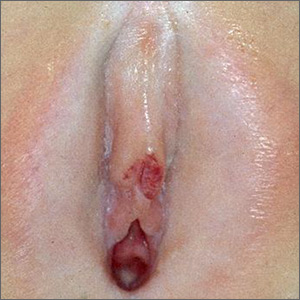
Physical examination by our institution’s Child Protection Team revealed clitoral hood swelling with subepithelial hemorrhages, a blood blister on the right labia minora, a fissure and subepithelial hemorrhages on the posterior fourchette, and a thin depigmented figure-of-eight lesion around the vulva and anus.
THE DIAGNOSIS
Since the clinical findings were consistent with prepubertal lichen sclerosus (LS), the CPS case was closed and the patient was referred to Pediatric Gynecology. Treatment with high-potency topical steroids was initiated with clobetasol ointment 0.05% twice daily for 2 weeks, then once daily for 2 weeks. She was then switched to triamcinolone ointment 0.01% twice daily for 2 weeks, then once daily for 2 weeks. These treatments were enough to stop the LS flare and decrease the anogenital itching.
DISCUSSION
Lichen sclerosus is a chronic inflammatory skin disease that primarily presents in the anogenital region; however, extragenital lesions on the upper extremities, thighs, and breasts have been reported in 15% to 20% of patients.1 Lichen sclerosus most commonly affects females as a result of low estrogen and may occur during puberty or following menopause, but it also is seen in males.1,2 The estimated prevalence of LS in prepubertal girls is 1 in 900.3 The effects of increased estrogen exposure on LS during puberty are not entirely clear. Lichen sclerosus previously was thought to improve with puberty, since it is not as common in women of reproductive age; however, studies have shown persistent symptoms after menarche in some patients.4-6
The pathogenesis of LS is multifactorial, likely with an autoimmune component, as it often is associated with other autoimmune findings such as thyroiditis, alopecia, pernicious anemia, and vitiligo.2 Diagnosis of prepubertal LS usually is made based on a review of the patient’s history and clinical examination. Presenting symptoms may include pruritus, skin irritation, vulvar pain, dysuria, bleeding from excoriations, fissures, and constipation.1,3,7
On physical examination, LS can present on the anogenital skin as smooth white spots or wrinkled, blotchy, atrophic patches. The skin around the vaginal opening and anus is thin and often is described as resembling parchment or cigarette paper in a figure-of-eight pattern (FIGURE 1A). Vulvar and anal fissures and subepithelial hemorrhages with the appearance of blood blisters also can be found (FIGURE 1B).8 Affected areas are fragile and susceptible to minor trauma, which may result in bruising or bleeding (FIGURE 1C).
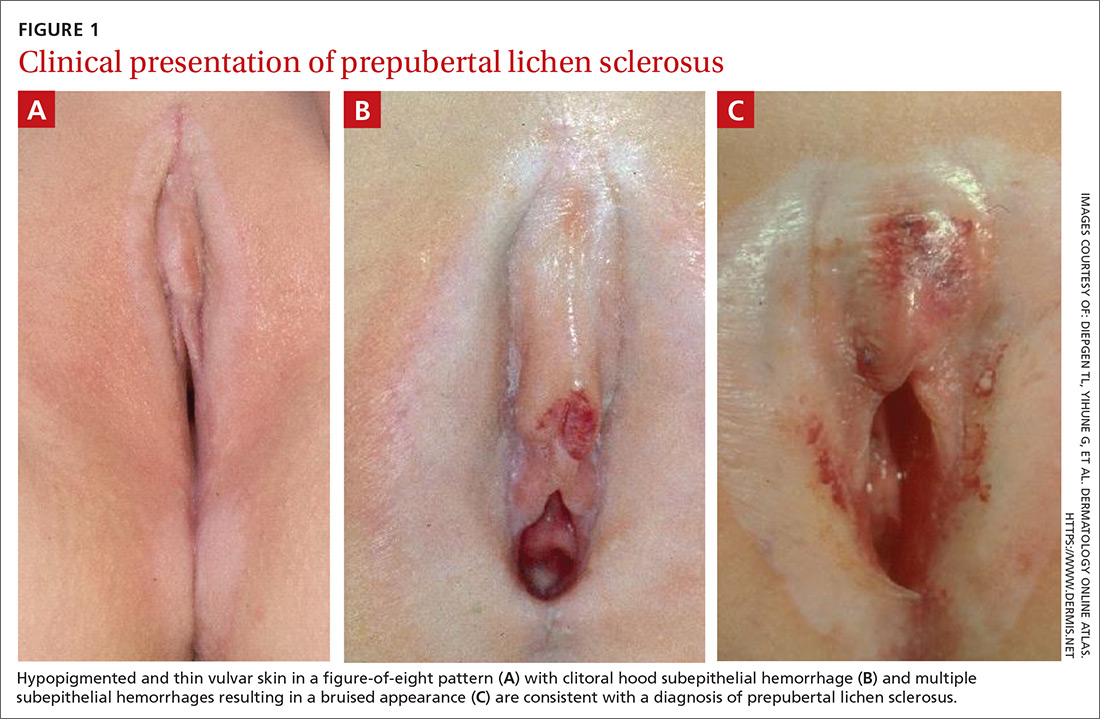
Over time, scarring can occur and may result in disruption of the anogenital architecture—specifically loss of the labia minora, narrowing of the introitus, and burying of the clitoris.1,2 These changes can be similar to the scarring seen in postmenopausal women with LS.
Continue to: The differential diagnosis...
The differential diagnosis for prepubertal LS includes vitiligo, lichen planus, lichen simplex chronicus, psoriasis, eczema, vulvovaginitis, contact dermatitis, and trauma.2,7 On average, it takes 1 to 2 years after onset of symptoms before a correct diagnosis of prepubertal LS is made, and trauma and/or sexual abuse often are first suspected.7,9 For clinicians who are unfamiliar with prepubertal LS, the clinical findings of anogenital bruising and bleeding understandably may be suggestive of abuse. It is important to note that diagnosis of LS does not preclude the possibility of sexual abuse; in some cases, LS can be triggered or exacerbated by anogenital trauma, known as the Koebner phenomenon.2
Treatment. After the diagnosis of prepubertal LS is established, the goals of treatment are to provide symptom relief and prevent scarring of the external genitalia. To our knowledge, there have been no randomized controlled trials for treatment of LS in prepubertal girls. In general, acute symptoms are treated with high-potency topical steroids, such as clobetasol propionate or betamethasone valerate, and treatment regimens are variable.7
LS has an unpredictable clinical course and there often are recurrences that require repeat courses of topical steroids.9 Since concurrent bacterial infection is common,10 genital cultures should be obtained prior to initiation of topical steroids if an infection is suspected.
Topical calcineurin inhibitors have been used successfully, but proof of their effectiveness is limited to case reports in the literature.7 Surgical treatment of LS typically is reserved for complications associated with symptomatic adhesions that are refractory to medical management.7,11 Vulvar hygiene is paramount to symptom control, and topical emollients can be used to manage minor irritation.7,8 In our patient, clobetasol and triamcinolone ointments were enough to stop the LS flare and decrease the anogenital itching.
THE TAKEAWAY
Although LS has very characteristic skin findings, the diagnosis continues to be challenging for physicians who are unfamiliar with this condition. Failure to recognize prepubertal LS not only delays diagnosis and treatment but also may lead to repeated genital examinations and investigation by CPS for suspected sexual abuse. As with any genital complaint in a prepubertal girl, diagnosis of LS should not preclude appropriate screening for sexual abuse. Although providers should be vigilant about potential sexual abuse, familiarity with skin conditions that mimic genital trauma is essential.
CORRESPONDENCE
Monica Rosen, MD, L4000 Women’s Hospital, 1500 E Medical Center Drive, SPC 5276 Ann Arbor, MI 48109; [email protected]
1. Powell JJ, Wojnarowska F. Lichen sclerosus. Lancet. 1999;353:1777-1783.
2. Murphy R. Lichen sclerosus. Dermatol Clin. 2010;28:707-715.
3. Powell J, Wojnarowska F. Childhood vulvar lichen sclerosus: an increasingly common problem. J Am Acad Dermatol. 2001;44:803-806.
4. Powell J, Wojnarowska F. Childhood vulvar lichen sclerosus. The course after puberty. J Reprod Med. 2002;47:706-709.
5. Smith SD, Fischer G. Childhood onset vulvar lichen sclerosus does not resolve at puberty: a prospective case series. Pediatr Dermatol. 2009;26:725-729.
6. Focseneanu MA, Gupta M, Squires KC, et al. The course of lichen sclerosus diagnosed prior to puberty. J Pediatr Adolesc Gynecol. 2013;26:153-155.
7. Bercaw-Pratt JL, Boardman LA, Simms-Cendan JS. Clinical recommendation: pediatric lichen sclerosus. J Pediatr Adolesc Gynecol. 2014;27:111-116.
8. Jenny C, Kirby P, Fuquay D. Genital lichen sclerosus mistaken for child sexual abuse. Pediatrics. 1989;83:597-599.
9. Dendrinos ML, Quint EH. Lichen sclerosus in children and adolescents. Curr Opin Obstet Gynecol. 2013;25:370-374.
10. Lagerstedt M, Karvinen K, Joki-Erkkila M, et al. Childhood lichen sclerosus—a challenge for clinicians. Pediatr Dermatol. 2013;30:444-450.
11. Gurumurthy M, Morah N, Gioffre G, et al. The surgical management of complications of vulval lichen sclerosus. Eur J Obstet Gynecol Reprod Biol. 2012;162:79-82.
THE CASE
A 4-year-old girl presented to her pediatrician with genital discomfort and dysuria of 6 months’ duration. The patient’s mother said that 3 days earlier, she noticed a tear near the child’s clitoris and a scab on the labia minora that the mother attributed to minor trauma from scratching. The pediatrician was concerned about genital trauma from sexual abuse and referred the patient to the emergency department, where a report with child protective services (CPS) was filed. The mother reported that the patient and her 8-year-old sibling spent 3 to 4 hours a day with a babysitter, who was always supervised, and the parents had no concerns about possible sexual abuse.
Physical examination by our institution’s Child Protection Team revealed clitoral hood swelling with subepithelial hemorrhages, a blood blister on the right labia minora, a fissure and subepithelial hemorrhages on the posterior fourchette, and a thin depigmented figure-of-eight lesion around the vulva and anus.
THE DIAGNOSIS
Since the clinical findings were consistent with prepubertal lichen sclerosus (LS), the CPS case was closed and the patient was referred to Pediatric Gynecology. Treatment with high-potency topical steroids was initiated with clobetasol ointment 0.05% twice daily for 2 weeks, then once daily for 2 weeks. She was then switched to triamcinolone ointment 0.01% twice daily for 2 weeks, then once daily for 2 weeks. These treatments were enough to stop the LS flare and decrease the anogenital itching.
DISCUSSION
Lichen sclerosus is a chronic inflammatory skin disease that primarily presents in the anogenital region; however, extragenital lesions on the upper extremities, thighs, and breasts have been reported in 15% to 20% of patients.1 Lichen sclerosus most commonly affects females as a result of low estrogen and may occur during puberty or following menopause, but it also is seen in males.1,2 The estimated prevalence of LS in prepubertal girls is 1 in 900.3 The effects of increased estrogen exposure on LS during puberty are not entirely clear. Lichen sclerosus previously was thought to improve with puberty, since it is not as common in women of reproductive age; however, studies have shown persistent symptoms after menarche in some patients.4-6
The pathogenesis of LS is multifactorial, likely with an autoimmune component, as it often is associated with other autoimmune findings such as thyroiditis, alopecia, pernicious anemia, and vitiligo.2 Diagnosis of prepubertal LS usually is made based on a review of the patient’s history and clinical examination. Presenting symptoms may include pruritus, skin irritation, vulvar pain, dysuria, bleeding from excoriations, fissures, and constipation.1,3,7
On physical examination, LS can present on the anogenital skin as smooth white spots or wrinkled, blotchy, atrophic patches. The skin around the vaginal opening and anus is thin and often is described as resembling parchment or cigarette paper in a figure-of-eight pattern (FIGURE 1A). Vulvar and anal fissures and subepithelial hemorrhages with the appearance of blood blisters also can be found (FIGURE 1B).8 Affected areas are fragile and susceptible to minor trauma, which may result in bruising or bleeding (FIGURE 1C).

Over time, scarring can occur and may result in disruption of the anogenital architecture—specifically loss of the labia minora, narrowing of the introitus, and burying of the clitoris.1,2 These changes can be similar to the scarring seen in postmenopausal women with LS.
Continue to: The differential diagnosis...
The differential diagnosis for prepubertal LS includes vitiligo, lichen planus, lichen simplex chronicus, psoriasis, eczema, vulvovaginitis, contact dermatitis, and trauma.2,7 On average, it takes 1 to 2 years after onset of symptoms before a correct diagnosis of prepubertal LS is made, and trauma and/or sexual abuse often are first suspected.7,9 For clinicians who are unfamiliar with prepubertal LS, the clinical findings of anogenital bruising and bleeding understandably may be suggestive of abuse. It is important to note that diagnosis of LS does not preclude the possibility of sexual abuse; in some cases, LS can be triggered or exacerbated by anogenital trauma, known as the Koebner phenomenon.2
Treatment. After the diagnosis of prepubertal LS is established, the goals of treatment are to provide symptom relief and prevent scarring of the external genitalia. To our knowledge, there have been no randomized controlled trials for treatment of LS in prepubertal girls. In general, acute symptoms are treated with high-potency topical steroids, such as clobetasol propionate or betamethasone valerate, and treatment regimens are variable.7
LS has an unpredictable clinical course and there often are recurrences that require repeat courses of topical steroids.9 Since concurrent bacterial infection is common,10 genital cultures should be obtained prior to initiation of topical steroids if an infection is suspected.
Topical calcineurin inhibitors have been used successfully, but proof of their effectiveness is limited to case reports in the literature.7 Surgical treatment of LS typically is reserved for complications associated with symptomatic adhesions that are refractory to medical management.7,11 Vulvar hygiene is paramount to symptom control, and topical emollients can be used to manage minor irritation.7,8 In our patient, clobetasol and triamcinolone ointments were enough to stop the LS flare and decrease the anogenital itching.
THE TAKEAWAY
Although LS has very characteristic skin findings, the diagnosis continues to be challenging for physicians who are unfamiliar with this condition. Failure to recognize prepubertal LS not only delays diagnosis and treatment but also may lead to repeated genital examinations and investigation by CPS for suspected sexual abuse. As with any genital complaint in a prepubertal girl, diagnosis of LS should not preclude appropriate screening for sexual abuse. Although providers should be vigilant about potential sexual abuse, familiarity with skin conditions that mimic genital trauma is essential.
CORRESPONDENCE
Monica Rosen, MD, L4000 Women’s Hospital, 1500 E Medical Center Drive, SPC 5276 Ann Arbor, MI 48109; [email protected]
THE CASE
A 4-year-old girl presented to her pediatrician with genital discomfort and dysuria of 6 months’ duration. The patient’s mother said that 3 days earlier, she noticed a tear near the child’s clitoris and a scab on the labia minora that the mother attributed to minor trauma from scratching. The pediatrician was concerned about genital trauma from sexual abuse and referred the patient to the emergency department, where a report with child protective services (CPS) was filed. The mother reported that the patient and her 8-year-old sibling spent 3 to 4 hours a day with a babysitter, who was always supervised, and the parents had no concerns about possible sexual abuse.
Physical examination by our institution’s Child Protection Team revealed clitoral hood swelling with subepithelial hemorrhages, a blood blister on the right labia minora, a fissure and subepithelial hemorrhages on the posterior fourchette, and a thin depigmented figure-of-eight lesion around the vulva and anus.
THE DIAGNOSIS
Since the clinical findings were consistent with prepubertal lichen sclerosus (LS), the CPS case was closed and the patient was referred to Pediatric Gynecology. Treatment with high-potency topical steroids was initiated with clobetasol ointment 0.05% twice daily for 2 weeks, then once daily for 2 weeks. She was then switched to triamcinolone ointment 0.01% twice daily for 2 weeks, then once daily for 2 weeks. These treatments were enough to stop the LS flare and decrease the anogenital itching.
DISCUSSION
Lichen sclerosus is a chronic inflammatory skin disease that primarily presents in the anogenital region; however, extragenital lesions on the upper extremities, thighs, and breasts have been reported in 15% to 20% of patients.1 Lichen sclerosus most commonly affects females as a result of low estrogen and may occur during puberty or following menopause, but it also is seen in males.1,2 The estimated prevalence of LS in prepubertal girls is 1 in 900.3 The effects of increased estrogen exposure on LS during puberty are not entirely clear. Lichen sclerosus previously was thought to improve with puberty, since it is not as common in women of reproductive age; however, studies have shown persistent symptoms after menarche in some patients.4-6
The pathogenesis of LS is multifactorial, likely with an autoimmune component, as it often is associated with other autoimmune findings such as thyroiditis, alopecia, pernicious anemia, and vitiligo.2 Diagnosis of prepubertal LS usually is made based on a review of the patient’s history and clinical examination. Presenting symptoms may include pruritus, skin irritation, vulvar pain, dysuria, bleeding from excoriations, fissures, and constipation.1,3,7
On physical examination, LS can present on the anogenital skin as smooth white spots or wrinkled, blotchy, atrophic patches. The skin around the vaginal opening and anus is thin and often is described as resembling parchment or cigarette paper in a figure-of-eight pattern (FIGURE 1A). Vulvar and anal fissures and subepithelial hemorrhages with the appearance of blood blisters also can be found (FIGURE 1B).8 Affected areas are fragile and susceptible to minor trauma, which may result in bruising or bleeding (FIGURE 1C).

Over time, scarring can occur and may result in disruption of the anogenital architecture—specifically loss of the labia minora, narrowing of the introitus, and burying of the clitoris.1,2 These changes can be similar to the scarring seen in postmenopausal women with LS.
Continue to: The differential diagnosis...
The differential diagnosis for prepubertal LS includes vitiligo, lichen planus, lichen simplex chronicus, psoriasis, eczema, vulvovaginitis, contact dermatitis, and trauma.2,7 On average, it takes 1 to 2 years after onset of symptoms before a correct diagnosis of prepubertal LS is made, and trauma and/or sexual abuse often are first suspected.7,9 For clinicians who are unfamiliar with prepubertal LS, the clinical findings of anogenital bruising and bleeding understandably may be suggestive of abuse. It is important to note that diagnosis of LS does not preclude the possibility of sexual abuse; in some cases, LS can be triggered or exacerbated by anogenital trauma, known as the Koebner phenomenon.2
Treatment. After the diagnosis of prepubertal LS is established, the goals of treatment are to provide symptom relief and prevent scarring of the external genitalia. To our knowledge, there have been no randomized controlled trials for treatment of LS in prepubertal girls. In general, acute symptoms are treated with high-potency topical steroids, such as clobetasol propionate or betamethasone valerate, and treatment regimens are variable.7
LS has an unpredictable clinical course and there often are recurrences that require repeat courses of topical steroids.9 Since concurrent bacterial infection is common,10 genital cultures should be obtained prior to initiation of topical steroids if an infection is suspected.
Topical calcineurin inhibitors have been used successfully, but proof of their effectiveness is limited to case reports in the literature.7 Surgical treatment of LS typically is reserved for complications associated with symptomatic adhesions that are refractory to medical management.7,11 Vulvar hygiene is paramount to symptom control, and topical emollients can be used to manage minor irritation.7,8 In our patient, clobetasol and triamcinolone ointments were enough to stop the LS flare and decrease the anogenital itching.
THE TAKEAWAY
Although LS has very characteristic skin findings, the diagnosis continues to be challenging for physicians who are unfamiliar with this condition. Failure to recognize prepubertal LS not only delays diagnosis and treatment but also may lead to repeated genital examinations and investigation by CPS for suspected sexual abuse. As with any genital complaint in a prepubertal girl, diagnosis of LS should not preclude appropriate screening for sexual abuse. Although providers should be vigilant about potential sexual abuse, familiarity with skin conditions that mimic genital trauma is essential.
CORRESPONDENCE
Monica Rosen, MD, L4000 Women’s Hospital, 1500 E Medical Center Drive, SPC 5276 Ann Arbor, MI 48109; [email protected]
1. Powell JJ, Wojnarowska F. Lichen sclerosus. Lancet. 1999;353:1777-1783.
2. Murphy R. Lichen sclerosus. Dermatol Clin. 2010;28:707-715.
3. Powell J, Wojnarowska F. Childhood vulvar lichen sclerosus: an increasingly common problem. J Am Acad Dermatol. 2001;44:803-806.
4. Powell J, Wojnarowska F. Childhood vulvar lichen sclerosus. The course after puberty. J Reprod Med. 2002;47:706-709.
5. Smith SD, Fischer G. Childhood onset vulvar lichen sclerosus does not resolve at puberty: a prospective case series. Pediatr Dermatol. 2009;26:725-729.
6. Focseneanu MA, Gupta M, Squires KC, et al. The course of lichen sclerosus diagnosed prior to puberty. J Pediatr Adolesc Gynecol. 2013;26:153-155.
7. Bercaw-Pratt JL, Boardman LA, Simms-Cendan JS. Clinical recommendation: pediatric lichen sclerosus. J Pediatr Adolesc Gynecol. 2014;27:111-116.
8. Jenny C, Kirby P, Fuquay D. Genital lichen sclerosus mistaken for child sexual abuse. Pediatrics. 1989;83:597-599.
9. Dendrinos ML, Quint EH. Lichen sclerosus in children and adolescents. Curr Opin Obstet Gynecol. 2013;25:370-374.
10. Lagerstedt M, Karvinen K, Joki-Erkkila M, et al. Childhood lichen sclerosus—a challenge for clinicians. Pediatr Dermatol. 2013;30:444-450.
11. Gurumurthy M, Morah N, Gioffre G, et al. The surgical management of complications of vulval lichen sclerosus. Eur J Obstet Gynecol Reprod Biol. 2012;162:79-82.
1. Powell JJ, Wojnarowska F. Lichen sclerosus. Lancet. 1999;353:1777-1783.
2. Murphy R. Lichen sclerosus. Dermatol Clin. 2010;28:707-715.
3. Powell J, Wojnarowska F. Childhood vulvar lichen sclerosus: an increasingly common problem. J Am Acad Dermatol. 2001;44:803-806.
4. Powell J, Wojnarowska F. Childhood vulvar lichen sclerosus. The course after puberty. J Reprod Med. 2002;47:706-709.
5. Smith SD, Fischer G. Childhood onset vulvar lichen sclerosus does not resolve at puberty: a prospective case series. Pediatr Dermatol. 2009;26:725-729.
6. Focseneanu MA, Gupta M, Squires KC, et al. The course of lichen sclerosus diagnosed prior to puberty. J Pediatr Adolesc Gynecol. 2013;26:153-155.
7. Bercaw-Pratt JL, Boardman LA, Simms-Cendan JS. Clinical recommendation: pediatric lichen sclerosus. J Pediatr Adolesc Gynecol. 2014;27:111-116.
8. Jenny C, Kirby P, Fuquay D. Genital lichen sclerosus mistaken for child sexual abuse. Pediatrics. 1989;83:597-599.
9. Dendrinos ML, Quint EH. Lichen sclerosus in children and adolescents. Curr Opin Obstet Gynecol. 2013;25:370-374.
10. Lagerstedt M, Karvinen K, Joki-Erkkila M, et al. Childhood lichen sclerosus—a challenge for clinicians. Pediatr Dermatol. 2013;30:444-450.
11. Gurumurthy M, Morah N, Gioffre G, et al. The surgical management of complications of vulval lichen sclerosus. Eur J Obstet Gynecol Reprod Biol. 2012;162:79-82.
We all benefit from this powerful pairing
In this issue of JFP, Rajesh and colleagues present a scholarly review that details why it makes sense to integrate behavioral health into primary care. There is strong evidence that the presence of behavioral health care managers in primary care practices improves outcomes for patients with anxiety and depression. The mental health–trained care manager serves as a link between the primary care physician and mental health professional and can provide psychotherapy, as well.
A more integrated model, however, includes a full range of behavioral health services on site. Although not as well studied, co-location is a powerful pairing of physical and mental health treatment. Primary care physicians benefit because referral and feedback are immediate and seamless through warm hand-offs and easy access to medical and mental health notes in a common medical record. Patients benefit because they are more likely to engage with treatment when the physician introduces them to the mental health professional and expresses confidence in his or her abilities.
We know that trust improves treatment outcomes. What better way to encourage trust than with a warm smile and handshake when a patient is most vulnerable? In addition, integrating behavioral health into primary care helps patients avoid the stigma of going to a “mental health clinic.”
Integrating medical and mental health professionals into one practice is hardly a new idea and is spreading quickly in some parts of the country. It is perhaps no coincidence that Rajesh refers to a study of behavioral health integration in Colorado family practice offices. Recently, I (JH) presented a CME program to family physicians in Colorado, and, after reviewing recent studies of anxiety and depression, I asked how many participants had mental health professionals working in their practices. A full third raised their hands.
I (JH) had an excellent psychologist in my practice as far back as 1980, and he was an integral member of the care team. It seems that behavioral health integration has been a long time coming, and as a health care community we would be wise to spread this model of whole-person care to all primary care practices.
In this issue of JFP, Rajesh and colleagues present a scholarly review that details why it makes sense to integrate behavioral health into primary care. There is strong evidence that the presence of behavioral health care managers in primary care practices improves outcomes for patients with anxiety and depression. The mental health–trained care manager serves as a link between the primary care physician and mental health professional and can provide psychotherapy, as well.
A more integrated model, however, includes a full range of behavioral health services on site. Although not as well studied, co-location is a powerful pairing of physical and mental health treatment. Primary care physicians benefit because referral and feedback are immediate and seamless through warm hand-offs and easy access to medical and mental health notes in a common medical record. Patients benefit because they are more likely to engage with treatment when the physician introduces them to the mental health professional and expresses confidence in his or her abilities.
We know that trust improves treatment outcomes. What better way to encourage trust than with a warm smile and handshake when a patient is most vulnerable? In addition, integrating behavioral health into primary care helps patients avoid the stigma of going to a “mental health clinic.”
Integrating medical and mental health professionals into one practice is hardly a new idea and is spreading quickly in some parts of the country. It is perhaps no coincidence that Rajesh refers to a study of behavioral health integration in Colorado family practice offices. Recently, I (JH) presented a CME program to family physicians in Colorado, and, after reviewing recent studies of anxiety and depression, I asked how many participants had mental health professionals working in their practices. A full third raised their hands.
I (JH) had an excellent psychologist in my practice as far back as 1980, and he was an integral member of the care team. It seems that behavioral health integration has been a long time coming, and as a health care community we would be wise to spread this model of whole-person care to all primary care practices.
In this issue of JFP, Rajesh and colleagues present a scholarly review that details why it makes sense to integrate behavioral health into primary care. There is strong evidence that the presence of behavioral health care managers in primary care practices improves outcomes for patients with anxiety and depression. The mental health–trained care manager serves as a link between the primary care physician and mental health professional and can provide psychotherapy, as well.
A more integrated model, however, includes a full range of behavioral health services on site. Although not as well studied, co-location is a powerful pairing of physical and mental health treatment. Primary care physicians benefit because referral and feedback are immediate and seamless through warm hand-offs and easy access to medical and mental health notes in a common medical record. Patients benefit because they are more likely to engage with treatment when the physician introduces them to the mental health professional and expresses confidence in his or her abilities.
We know that trust improves treatment outcomes. What better way to encourage trust than with a warm smile and handshake when a patient is most vulnerable? In addition, integrating behavioral health into primary care helps patients avoid the stigma of going to a “mental health clinic.”
Integrating medical and mental health professionals into one practice is hardly a new idea and is spreading quickly in some parts of the country. It is perhaps no coincidence that Rajesh refers to a study of behavioral health integration in Colorado family practice offices. Recently, I (JH) presented a CME program to family physicians in Colorado, and, after reviewing recent studies of anxiety and depression, I asked how many participants had mental health professionals working in their practices. A full third raised their hands.
I (JH) had an excellent psychologist in my practice as far back as 1980, and he was an integral member of the care team. It seems that behavioral health integration has been a long time coming, and as a health care community we would be wise to spread this model of whole-person care to all primary care practices.
Rapidly growing lesions on the forehead
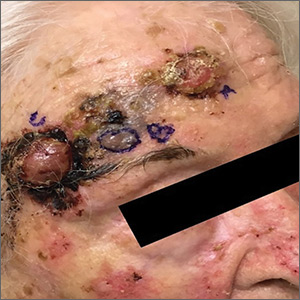
A 97-year-old woman with a history of atrial fibrillation and nonmelanoma skin cancer presented to our clinic from an assisted living facility with a several-month history of rapidly growing forehead lesions. She denied symptoms, other than some bleeding and crusting, but was concerned about their appearance. She reported a notable history of sun exposure.
The patient had 3 confluent, but distinct, lesions on her forehead: an erythematous crateriform nodule with overlying hyperkeratotic scale (FIGURE, Lesion A); a nodular hyperpigmented plaque with irregular color and borders (Lesion B); and a pearly well-vascularized erythematous nodule with surrounding hemorrhagic crust (Lesion C).
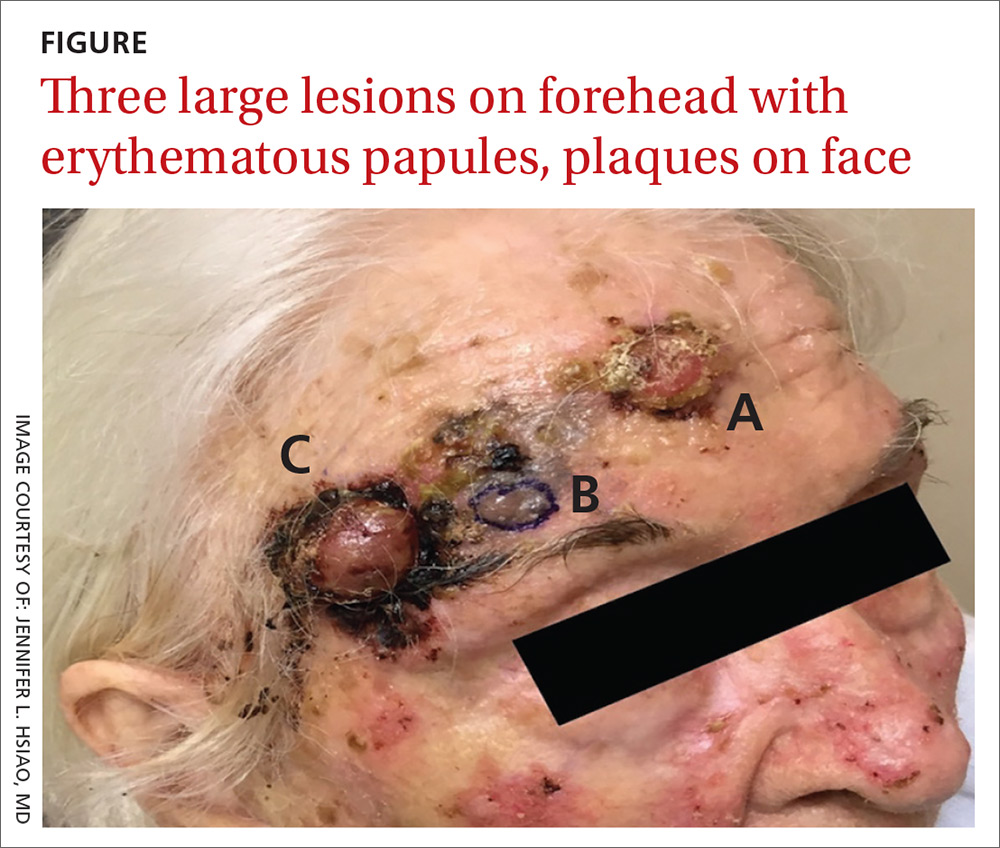
She also had scattered, thin, gritty pink papules and plaques on the face that were thought to be actinic keratosis and nonmelanoma skin cancers based on clinical morphology; however, the patient deferred workup and treatment of these lesions to focus on the forehead lesions. The decision was made to biopsy all 3 clinical morphologies seen. The risks and benefits of biopsy were reviewed with the patient and her daughter, and they opted to proceed. The areas were anesthetized with an injection of 1% lidocaine and epinephrine 1:100,000; 3 shave biopsies were performed. Hemostasis was obtained with electrodesiccation.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Skin cancer
A histopathology report revealed that Lesion A was squamous cell carcinoma (SCC), Lesion B was a melanoma with a Breslow depth of at least 1.2 mm, and Lesion C was basal cell carcinoma (BCC). It is unusual to have a patient present with BCC, SCC, and melanoma concurrently in the same anatomic region.
Two of the lesions were nonmelanoma skin cancers (NMSC). BCC is the most common NMSC in the United States, affecting more than 3.3 million people per year.1 Although there are several subtypes of BCC with varying clinical presentations, the most classic appearance is a pearly papule with or without surface telangiectasias.2
SCC has an incidence of 200,000 to 400,000 cases per year in the United States and the lifetime risk is 9% to 14% in men and 4% to 9% in women.3 SCC most commonly presents as a hyperkeratotic papule or plaque.2 Lesions suspicious for SCC and BCC should be biopsied and the diagnosis confirmed by histopathologic analysis. These NMSCs are locally destructive, but rarely metastatic with a generally good prognosis. The standard treatment for both is surgical excision with consideration for other treatment modalities, such as topical therapies, chemotherapy, and radiation, depending on tumor characteristics as well as whether the patient is a good surgical candidate.1,3
Melanoma is rising in incidence each year, with nearly 100,000 new cases expected in the United States this year.4 It is the leading cause of skin cancer related mortality.5 The most common suspicious lesions are variably pigmented macules with irregular borders. Biopsy and subsequent histopathologic analysis will confirm the diagnosis.
When a lesion is clinically suspicious for melanoma, it is particularly important to consider an excisional biopsy to allow for proper staging.5 Examples of appropriate excisional biopsies include elliptical excisions, punch biopsies, and deep shave biopsies.5 Definitive treatment involves a wider and deeper excision with histologically confirmed clear margins.5
Continue to: This case required a multidisciplinary team
This case required a multidisciplinary team
The patient was cleared for surgery; however, after the patient held her warfarin in preparation for the resection, she suffered a left frontal operculum infarction. At this point, she was re-evaluated by her head and neck physician, cardiologist, and anesthesiologist. Consensus was reached that the patient was at high perioperative risk for morbidity and mortality, and surgical intervention was no longer considered a viable option.
The patient then opted for palliative radiation therapy to all 3 lesions, with the understanding that the local control offered by radiotherapy would be inferior to what resection would provide for the melanoma lesion. Although not curative, radiotherapy was expected to provide local symptom relief for the melanoma, consistent with the patient’s palliative goals of care. In the past, melanoma was thought to be resistant to radiation, but recent evidence suggests that it may be at least partially susceptible to hypofractionated courses of radiation.6
Radiation oncology recommended a 6 to 15 fraction regimen and she had a good clinical response with > 50% decrease in the size of all 3 lesions along with cessation of bleeding.
The take-home lesson. The findings in this case serve as an important reminder to biopsy lesions with varying morphologies—even when they are in close proximity to one another. Foregoing any of the biopsies in this case would have led to a missed diagnosis, which has implications for optimal management and treatment.
CORRESPONDENCE
Jennifer L. Hsiao, MD, 2020 Santa Monica Boulevard, Suite 510, Santa Monica, CA 90404; [email protected]
1. Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559.
2. Firnhaber JM. Diagnosis and treatment of basal cell and squamous cell carcinoma. Am Fam Physician. 2012;86:161-168.
3. Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
4. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
5. Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
6. Vuong W, Lin J, Wei RL. Palliative radiotherapy for skin malignancies. Ann Palliat Med. 2017;6:165-172.
A 97-year-old woman with a history of atrial fibrillation and nonmelanoma skin cancer presented to our clinic from an assisted living facility with a several-month history of rapidly growing forehead lesions. She denied symptoms, other than some bleeding and crusting, but was concerned about their appearance. She reported a notable history of sun exposure.
The patient had 3 confluent, but distinct, lesions on her forehead: an erythematous crateriform nodule with overlying hyperkeratotic scale (FIGURE, Lesion A); a nodular hyperpigmented plaque with irregular color and borders (Lesion B); and a pearly well-vascularized erythematous nodule with surrounding hemorrhagic crust (Lesion C).

She also had scattered, thin, gritty pink papules and plaques on the face that were thought to be actinic keratosis and nonmelanoma skin cancers based on clinical morphology; however, the patient deferred workup and treatment of these lesions to focus on the forehead lesions. The decision was made to biopsy all 3 clinical morphologies seen. The risks and benefits of biopsy were reviewed with the patient and her daughter, and they opted to proceed. The areas were anesthetized with an injection of 1% lidocaine and epinephrine 1:100,000; 3 shave biopsies were performed. Hemostasis was obtained with electrodesiccation.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Skin cancer
A histopathology report revealed that Lesion A was squamous cell carcinoma (SCC), Lesion B was a melanoma with a Breslow depth of at least 1.2 mm, and Lesion C was basal cell carcinoma (BCC). It is unusual to have a patient present with BCC, SCC, and melanoma concurrently in the same anatomic region.
Two of the lesions were nonmelanoma skin cancers (NMSC). BCC is the most common NMSC in the United States, affecting more than 3.3 million people per year.1 Although there are several subtypes of BCC with varying clinical presentations, the most classic appearance is a pearly papule with or without surface telangiectasias.2
SCC has an incidence of 200,000 to 400,000 cases per year in the United States and the lifetime risk is 9% to 14% in men and 4% to 9% in women.3 SCC most commonly presents as a hyperkeratotic papule or plaque.2 Lesions suspicious for SCC and BCC should be biopsied and the diagnosis confirmed by histopathologic analysis. These NMSCs are locally destructive, but rarely metastatic with a generally good prognosis. The standard treatment for both is surgical excision with consideration for other treatment modalities, such as topical therapies, chemotherapy, and radiation, depending on tumor characteristics as well as whether the patient is a good surgical candidate.1,3
Melanoma is rising in incidence each year, with nearly 100,000 new cases expected in the United States this year.4 It is the leading cause of skin cancer related mortality.5 The most common suspicious lesions are variably pigmented macules with irregular borders. Biopsy and subsequent histopathologic analysis will confirm the diagnosis.
When a lesion is clinically suspicious for melanoma, it is particularly important to consider an excisional biopsy to allow for proper staging.5 Examples of appropriate excisional biopsies include elliptical excisions, punch biopsies, and deep shave biopsies.5 Definitive treatment involves a wider and deeper excision with histologically confirmed clear margins.5
Continue to: This case required a multidisciplinary team
This case required a multidisciplinary team
The patient was cleared for surgery; however, after the patient held her warfarin in preparation for the resection, she suffered a left frontal operculum infarction. At this point, she was re-evaluated by her head and neck physician, cardiologist, and anesthesiologist. Consensus was reached that the patient was at high perioperative risk for morbidity and mortality, and surgical intervention was no longer considered a viable option.
The patient then opted for palliative radiation therapy to all 3 lesions, with the understanding that the local control offered by radiotherapy would be inferior to what resection would provide for the melanoma lesion. Although not curative, radiotherapy was expected to provide local symptom relief for the melanoma, consistent with the patient’s palliative goals of care. In the past, melanoma was thought to be resistant to radiation, but recent evidence suggests that it may be at least partially susceptible to hypofractionated courses of radiation.6
Radiation oncology recommended a 6 to 15 fraction regimen and she had a good clinical response with > 50% decrease in the size of all 3 lesions along with cessation of bleeding.
The take-home lesson. The findings in this case serve as an important reminder to biopsy lesions with varying morphologies—even when they are in close proximity to one another. Foregoing any of the biopsies in this case would have led to a missed diagnosis, which has implications for optimal management and treatment.
CORRESPONDENCE
Jennifer L. Hsiao, MD, 2020 Santa Monica Boulevard, Suite 510, Santa Monica, CA 90404; [email protected]
A 97-year-old woman with a history of atrial fibrillation and nonmelanoma skin cancer presented to our clinic from an assisted living facility with a several-month history of rapidly growing forehead lesions. She denied symptoms, other than some bleeding and crusting, but was concerned about their appearance. She reported a notable history of sun exposure.
The patient had 3 confluent, but distinct, lesions on her forehead: an erythematous crateriform nodule with overlying hyperkeratotic scale (FIGURE, Lesion A); a nodular hyperpigmented plaque with irregular color and borders (Lesion B); and a pearly well-vascularized erythematous nodule with surrounding hemorrhagic crust (Lesion C).

She also had scattered, thin, gritty pink papules and plaques on the face that were thought to be actinic keratosis and nonmelanoma skin cancers based on clinical morphology; however, the patient deferred workup and treatment of these lesions to focus on the forehead lesions. The decision was made to biopsy all 3 clinical morphologies seen. The risks and benefits of biopsy were reviewed with the patient and her daughter, and they opted to proceed. The areas were anesthetized with an injection of 1% lidocaine and epinephrine 1:100,000; 3 shave biopsies were performed. Hemostasis was obtained with electrodesiccation.
WHAT IS YOUR DIAGNOSIS?
HOW WOULD YOU TREAT THIS PATIENT?
Diagnosis: Skin cancer
A histopathology report revealed that Lesion A was squamous cell carcinoma (SCC), Lesion B was a melanoma with a Breslow depth of at least 1.2 mm, and Lesion C was basal cell carcinoma (BCC). It is unusual to have a patient present with BCC, SCC, and melanoma concurrently in the same anatomic region.
Two of the lesions were nonmelanoma skin cancers (NMSC). BCC is the most common NMSC in the United States, affecting more than 3.3 million people per year.1 Although there are several subtypes of BCC with varying clinical presentations, the most classic appearance is a pearly papule with or without surface telangiectasias.2
SCC has an incidence of 200,000 to 400,000 cases per year in the United States and the lifetime risk is 9% to 14% in men and 4% to 9% in women.3 SCC most commonly presents as a hyperkeratotic papule or plaque.2 Lesions suspicious for SCC and BCC should be biopsied and the diagnosis confirmed by histopathologic analysis. These NMSCs are locally destructive, but rarely metastatic with a generally good prognosis. The standard treatment for both is surgical excision with consideration for other treatment modalities, such as topical therapies, chemotherapy, and radiation, depending on tumor characteristics as well as whether the patient is a good surgical candidate.1,3
Melanoma is rising in incidence each year, with nearly 100,000 new cases expected in the United States this year.4 It is the leading cause of skin cancer related mortality.5 The most common suspicious lesions are variably pigmented macules with irregular borders. Biopsy and subsequent histopathologic analysis will confirm the diagnosis.
When a lesion is clinically suspicious for melanoma, it is particularly important to consider an excisional biopsy to allow for proper staging.5 Examples of appropriate excisional biopsies include elliptical excisions, punch biopsies, and deep shave biopsies.5 Definitive treatment involves a wider and deeper excision with histologically confirmed clear margins.5
Continue to: This case required a multidisciplinary team
This case required a multidisciplinary team
The patient was cleared for surgery; however, after the patient held her warfarin in preparation for the resection, she suffered a left frontal operculum infarction. At this point, she was re-evaluated by her head and neck physician, cardiologist, and anesthesiologist. Consensus was reached that the patient was at high perioperative risk for morbidity and mortality, and surgical intervention was no longer considered a viable option.
The patient then opted for palliative radiation therapy to all 3 lesions, with the understanding that the local control offered by radiotherapy would be inferior to what resection would provide for the melanoma lesion. Although not curative, radiotherapy was expected to provide local symptom relief for the melanoma, consistent with the patient’s palliative goals of care. In the past, melanoma was thought to be resistant to radiation, but recent evidence suggests that it may be at least partially susceptible to hypofractionated courses of radiation.6
Radiation oncology recommended a 6 to 15 fraction regimen and she had a good clinical response with > 50% decrease in the size of all 3 lesions along with cessation of bleeding.
The take-home lesson. The findings in this case serve as an important reminder to biopsy lesions with varying morphologies—even when they are in close proximity to one another. Foregoing any of the biopsies in this case would have led to a missed diagnosis, which has implications for optimal management and treatment.
CORRESPONDENCE
Jennifer L. Hsiao, MD, 2020 Santa Monica Boulevard, Suite 510, Santa Monica, CA 90404; [email protected]
1. Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559.
2. Firnhaber JM. Diagnosis and treatment of basal cell and squamous cell carcinoma. Am Fam Physician. 2012;86:161-168.
3. Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
4. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
5. Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
6. Vuong W, Lin J, Wei RL. Palliative radiotherapy for skin malignancies. Ann Palliat Med. 2017;6:165-172.
1. Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of basal cell carcinoma. J Am Acad Dermatol. 2018;78:540-559.
2. Firnhaber JM. Diagnosis and treatment of basal cell and squamous cell carcinoma. Am Fam Physician. 2012;86:161-168.
3. Kim JYS, Kozlow JH, Mittal B, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
4. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69:7-34.
5. Swetter SM, Tsao H, Bichakjian CK, et al. Guidelines of care for the management of primary cutaneous melanoma. J Am Acad Dermatol. 2019;80:208-250.
6. Vuong W, Lin J, Wei RL. Palliative radiotherapy for skin malignancies. Ann Palliat Med. 2017;6:165-172.
Would you be able to recognize the signs and symptoms of this particular drug overdose?
CASE 1
Two days after reviving her boyfriend with naloxone, a woman and her 30-year-old boyfriend presented to our family medicine clinic. They explained that he had injected heroin and shortly thereafter he stopped breathing and his lips turned blue. The patient’s girlfriend did not call emergency medical services (EMS) at the time because she was afraid of getting arrested due to past incarceration for possession of illegal drugs. Instead, she revived him with naloxone that she found in his bag.
Both the patient and his girlfriend were scared and surprised by his “terrible reaction,” as he had previously purchased heroin from the same dealer and used the same dose without similar effects. However, the patient did note that the drug he purchased this time had a bright white tinge, when normally the drug was light yellow.
On physical examination, the patient’s heart rate and blood pressure were normal. There were needle track marks on both forearms, elbows, and upper arms. A laboratory workup obtained during this visit revealed anemia and a normal basic metabolic panel. A hepatitis C virus antibody test was positive, and a hepatic function panel revealed elevated transaminase levels. Urine toxicology was positive for opioids and negative for other substances.
CASE 2
A 58-year-old man with a history of chronic hepatitis C, polysubstance abuse, and schizophrenia was transported to the emergency department by EMS after his family found him unresponsive in his bedroom. The patient had agonal breathing when EMS arrived, so they administered naloxone (4 mg intranasal and 4 mg intravenous). His breathing improved, but his mental status did not. He was still obtunded upon arrival in the emergency department and vomited 4 tan-colored patches. The patient was tachycardic (heart rate, 108 beats/min), hypertensive (blood pressure, 189/95 mm Hg), and had rapid shallow breathing (respiratory rate, 38 breaths/min). He was intubated for airway protection, at which time 2 more tan-colored patches were removed from his pharynx.
Laboratory evaluation revealed an acute kidney injury with a high anion metabolic acidosis. A hepatic function panel showed elevated transaminase levels. Plasma acetaminophen and salicylate levels were normal. A computed tomography head scan was normal. Urine toxicology was negative for opioids but was positive for cocaine and benzodiazepines.
THE DIAGNOSIS
Opioid overdose caused the acute respiratory depression in both cases. In Case 1, the patient unknowingly overdosed on heroin laced with fentanyl, known as China White, which likely caused the drug’s bright white tinge. In Case 2, the patient’s overdose was the result of oral ingestion of fentanyl patches. (Limited urine toxicology was negative for opiates because fentanyl is a fully synthetic opioid that shows up only with a specific or extended assay. More on this in a bit.)
DISCUSSION
The fatal drug overdose epidemic in the United States is growing. From 2000 to 2014, the mortality rate from drug overdose increased by 137%, including a 200% increase in the rate of overdose deaths related to opioids (ie, pain medications, heroin).1 Between 2013 and 2014, the age-adjusted mortality rate related to methadone, a synthetic opioid, remained unchanged; however, age-adjusted mortality rates related to natural and semisynthetic opioid pain medications, heroin, and synthetic opioids other than methadone (eg, fentanyl) increased by 9%, 26%, and 80%, respectively. In 2014, a sharp increase in overdose deaths related to synthetic opioids other than methadone coincided with law enforcement reports of increased availability of illegal fentanyl; however, the toxicology panel used by coroners and medical examiners at that time could not distinguish between illegal and prescription fentanyl.1
Continue to: Among 70,237 drug overdose deaths...
Among 70,237 drug overdose deaths in the United States in 2017, 47,600 (67.8%) involved an opioid. From 2013 to 2017, drug overdose death rates increased in 35 of 50 states and the District of Columbia, and significant increases in death rates involving synthetic opioids occurred in 15 out of 20 states, likely driven by illicitly manufactured fentanyl.2
Fentanyl-laced heroin: More common, but not new
In October 1991, 3-methylfentanyl was identified in 16 fatal drug overdoses in Allegheny County, Pennsylvania, contributing to a 4-fold increase in overdose deaths compared to the previous year. Fentanyl mixed with heroin and other drugs is commonly found in the Midwest, Northeast, and Southern regions of the United States; in 2014, more than 80% of fentanyl confiscations occurred in 10 states within these regions, with the highest incidence occurring in Ohio.3
When combined with fentanyl, heroin becomes 50 to 100 times more potent, resulting in a subjective high with exaggerated central nervous system depression manifesting as lethargy, miosis, and respiratory depression.4 Most drug users are unaware and unable to identify when heroin is laced with fentanyl, which may contribute to the rise in deaths from unintentional drug overdose.1,5,6
Oral abuse of fentanyl patches can be fatal
Outcomes from oral abuse of fentanyl patches have ranged from transient overdose symptoms, such as lethargy and respiratory depression, to death.7-9 When administered in a medical setting, transbuccal fentanyl has a bioavailability of 50% to 65% across the buccal membrane. Nearly 20% of the drug escapes hepatic first pass metabolism when fentanyl patches are ingested orally and enters the systemic circulation, resulting in severe overdose and potentially death. Prolonged chewing and sucking on fentanyl patches increases the contact time with the buccal membrane, resulting in increased systemic absorption compared to oral ingestion without chewing/sucking.7-9
Urine toxicology screening detects compounds based on a chemical assay for drugs—generally codeine, morphine, and their metabolites. Because fentanyl is a fully synthetic opioid, its structure is not
Continue to: Survival of fentanyl overdose depends on naloxone availability
Survival of fentanyl overdose depends on naloxone availability
Naloxone is a safe and effective antidote to an opioid overdose. It comes in 3 preparations, including intramuscular and subcutaneous injections and an intranasal spray.12 Concerns that naloxone will harm patients with opioid dependence are unfounded. Naloxone can induce symptoms of opioid withdrawal, such as yawning, lacrimation, piloerection, diaphoresis, myalgia, vomiting, and diarrhea. While these withdrawal symptoms are unpleasant, they are not life threatening.12 Due to its high potency, large doses of naloxone (ie, 4–16 mg) are required to reverse the effects of a fentanyl overdose.13 Intranasal naloxone hydrochloride 4 mg delivered in a single spray is preferred due to the ease of administration. Repeat doses may be necessary if respiratory depression continues or recurs prior to the arrival of emergency medical services. Increasing the availability of naloxone to first responders has the potential to save many lives.6
THE TAKEAWAY
Fentanyl is a major contributor to the growing drug overdose crisis in the United States. When laced with heroin or consumed orally in the form of transdermal patches, fentanyl becomes more potent and is increasingly fatal. It’s crucial that primary care physicians be able to identify and educate at-risk patients about the fatal consequences of fentanyl overdose and coordinate care to help get them into an appropriate rehabilitation program.
In order to quickly recognize the signs of fentanyl-related overdose, it’s important to be alert for this possibility. At the bedside, the most easily recognized abnormality associated with fentanyl or other opioid overdose is a decline in respiratory rate culminating in apnea.10 A respiratory rate of 12 breaths/min or less in a patient who is not in physiologic sleep strongly suggests acute opioid intoxication, particularly when accompanied by miosis or stupor. Other signs include bradycardia, hypotension, and seizures from anoxia.10
Apart from the severity of symptoms, it is hard to clinically distinguish fentanyl overdose from other opiate overdose incidents. Given the degree to which illegal opiates are contaminated with fentanyl in the United States,3 it is appropriate to screen for fentanyl with extended panel urine toxicology testing in patients with suspected opioid overdose.
CORRESPONDENCE
Jaividhya Dasarathy, MD, 2500 MetroHealth Medical Center, Cleveland, OH 44109; [email protected]
1. Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths—United States, 2000–2014. MMWR Morb Mortal Wkly Rep. 2016;64:1378-1382.
2. Scholl L, Seth P, Kariisa M, et al. Drug and opioid-involved overdose deaths—United States, 2013–2017. MMWR Morb Mortal Wkly Rep. 2019;67:1419-1427.
3. Hibbs J, Perper J, Winek CL. An outbreak of designer drug-related deaths in Pennsylvania. JAMA. 1991;265:1011-1013.
4. Increases in fentanyl drug confiscations and fentanyl-related overdose fatalities. Centers for Disease Control and Prevention Web site. https://emergency.cdc.gov/han/han00384.asp. Published October 26, 2015. Accessed May 3, 2019.
5. Fentanyl. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/drugoverdose/opioids/fentanyl.html. Updated December 19, 2018. Accessed May 3, 2019.
6. Peterson AB, Gladden RM, Delcher C, et al. Increases in fentanyl-related overdose deaths—Florida and Ohio, 2013–2015. MMWR Morb Mortal Wkly Rep. 2016;65:844-849.
7. Streisand JB, Varvel JR, Stanski DR, et al. Absorption and bioavailability of oral transmucosal fentanyl citrate. Anesthesiology. 1991;75:223-229.
8. Kharasch ED, Whittington D, Hoffer C. Influence of hepatic and intestinal cytochrome P4503A activity on the acute disposition and effects of oral transmucosal fentanyl citrate. Anesthesiology. 2004;101:729-737.
9. Woodall KL, Martin TL, McLellan BA. Oral abuse of fentanyl patches (Duragesic): seven case reports. J Forensic Sci. 2008;53:222-225.
10. Moeller KE, Lee KC, Kissack JC. Urine drug screening: practical guide for clinicians. Mayo Clin Proc. 2008;83:66-76.
11. Appropriate Use of Drug Testing in Clinical Addiction Medicine. American Society of Addiction Medicine Web site. https://www.asam.org/docs/default-source/quality-science/appropriate_use_of_drug_testing_in_clinical-1-(7).pdf?sfvrsn=2. Published April 5, 2017. Accessed May 30, 2019.
12. Boyer EW. Management of opioid analgesic overdose. N Engl J Med. 2012;367:146-155.
13. Drugs@FDA: FDA approved drug products. US Food and Drug Administration Web site. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=208411. Accessed May 22, 2019.
CASE 1
Two days after reviving her boyfriend with naloxone, a woman and her 30-year-old boyfriend presented to our family medicine clinic. They explained that he had injected heroin and shortly thereafter he stopped breathing and his lips turned blue. The patient’s girlfriend did not call emergency medical services (EMS) at the time because she was afraid of getting arrested due to past incarceration for possession of illegal drugs. Instead, she revived him with naloxone that she found in his bag.
Both the patient and his girlfriend were scared and surprised by his “terrible reaction,” as he had previously purchased heroin from the same dealer and used the same dose without similar effects. However, the patient did note that the drug he purchased this time had a bright white tinge, when normally the drug was light yellow.
On physical examination, the patient’s heart rate and blood pressure were normal. There were needle track marks on both forearms, elbows, and upper arms. A laboratory workup obtained during this visit revealed anemia and a normal basic metabolic panel. A hepatitis C virus antibody test was positive, and a hepatic function panel revealed elevated transaminase levels. Urine toxicology was positive for opioids and negative for other substances.
CASE 2
A 58-year-old man with a history of chronic hepatitis C, polysubstance abuse, and schizophrenia was transported to the emergency department by EMS after his family found him unresponsive in his bedroom. The patient had agonal breathing when EMS arrived, so they administered naloxone (4 mg intranasal and 4 mg intravenous). His breathing improved, but his mental status did not. He was still obtunded upon arrival in the emergency department and vomited 4 tan-colored patches. The patient was tachycardic (heart rate, 108 beats/min), hypertensive (blood pressure, 189/95 mm Hg), and had rapid shallow breathing (respiratory rate, 38 breaths/min). He was intubated for airway protection, at which time 2 more tan-colored patches were removed from his pharynx.
Laboratory evaluation revealed an acute kidney injury with a high anion metabolic acidosis. A hepatic function panel showed elevated transaminase levels. Plasma acetaminophen and salicylate levels were normal. A computed tomography head scan was normal. Urine toxicology was negative for opioids but was positive for cocaine and benzodiazepines.
THE DIAGNOSIS
Opioid overdose caused the acute respiratory depression in both cases. In Case 1, the patient unknowingly overdosed on heroin laced with fentanyl, known as China White, which likely caused the drug’s bright white tinge. In Case 2, the patient’s overdose was the result of oral ingestion of fentanyl patches. (Limited urine toxicology was negative for opiates because fentanyl is a fully synthetic opioid that shows up only with a specific or extended assay. More on this in a bit.)
DISCUSSION
The fatal drug overdose epidemic in the United States is growing. From 2000 to 2014, the mortality rate from drug overdose increased by 137%, including a 200% increase in the rate of overdose deaths related to opioids (ie, pain medications, heroin).1 Between 2013 and 2014, the age-adjusted mortality rate related to methadone, a synthetic opioid, remained unchanged; however, age-adjusted mortality rates related to natural and semisynthetic opioid pain medications, heroin, and synthetic opioids other than methadone (eg, fentanyl) increased by 9%, 26%, and 80%, respectively. In 2014, a sharp increase in overdose deaths related to synthetic opioids other than methadone coincided with law enforcement reports of increased availability of illegal fentanyl; however, the toxicology panel used by coroners and medical examiners at that time could not distinguish between illegal and prescription fentanyl.1
Continue to: Among 70,237 drug overdose deaths...
Among 70,237 drug overdose deaths in the United States in 2017, 47,600 (67.8%) involved an opioid. From 2013 to 2017, drug overdose death rates increased in 35 of 50 states and the District of Columbia, and significant increases in death rates involving synthetic opioids occurred in 15 out of 20 states, likely driven by illicitly manufactured fentanyl.2
Fentanyl-laced heroin: More common, but not new
In October 1991, 3-methylfentanyl was identified in 16 fatal drug overdoses in Allegheny County, Pennsylvania, contributing to a 4-fold increase in overdose deaths compared to the previous year. Fentanyl mixed with heroin and other drugs is commonly found in the Midwest, Northeast, and Southern regions of the United States; in 2014, more than 80% of fentanyl confiscations occurred in 10 states within these regions, with the highest incidence occurring in Ohio.3
When combined with fentanyl, heroin becomes 50 to 100 times more potent, resulting in a subjective high with exaggerated central nervous system depression manifesting as lethargy, miosis, and respiratory depression.4 Most drug users are unaware and unable to identify when heroin is laced with fentanyl, which may contribute to the rise in deaths from unintentional drug overdose.1,5,6
Oral abuse of fentanyl patches can be fatal
Outcomes from oral abuse of fentanyl patches have ranged from transient overdose symptoms, such as lethargy and respiratory depression, to death.7-9 When administered in a medical setting, transbuccal fentanyl has a bioavailability of 50% to 65% across the buccal membrane. Nearly 20% of the drug escapes hepatic first pass metabolism when fentanyl patches are ingested orally and enters the systemic circulation, resulting in severe overdose and potentially death. Prolonged chewing and sucking on fentanyl patches increases the contact time with the buccal membrane, resulting in increased systemic absorption compared to oral ingestion without chewing/sucking.7-9
Urine toxicology screening detects compounds based on a chemical assay for drugs—generally codeine, morphine, and their metabolites. Because fentanyl is a fully synthetic opioid, its structure is not
Continue to: Survival of fentanyl overdose depends on naloxone availability
Survival of fentanyl overdose depends on naloxone availability
Naloxone is a safe and effective antidote to an opioid overdose. It comes in 3 preparations, including intramuscular and subcutaneous injections and an intranasal spray.12 Concerns that naloxone will harm patients with opioid dependence are unfounded. Naloxone can induce symptoms of opioid withdrawal, such as yawning, lacrimation, piloerection, diaphoresis, myalgia, vomiting, and diarrhea. While these withdrawal symptoms are unpleasant, they are not life threatening.12 Due to its high potency, large doses of naloxone (ie, 4–16 mg) are required to reverse the effects of a fentanyl overdose.13 Intranasal naloxone hydrochloride 4 mg delivered in a single spray is preferred due to the ease of administration. Repeat doses may be necessary if respiratory depression continues or recurs prior to the arrival of emergency medical services. Increasing the availability of naloxone to first responders has the potential to save many lives.6
THE TAKEAWAY
Fentanyl is a major contributor to the growing drug overdose crisis in the United States. When laced with heroin or consumed orally in the form of transdermal patches, fentanyl becomes more potent and is increasingly fatal. It’s crucial that primary care physicians be able to identify and educate at-risk patients about the fatal consequences of fentanyl overdose and coordinate care to help get them into an appropriate rehabilitation program.
In order to quickly recognize the signs of fentanyl-related overdose, it’s important to be alert for this possibility. At the bedside, the most easily recognized abnormality associated with fentanyl or other opioid overdose is a decline in respiratory rate culminating in apnea.10 A respiratory rate of 12 breaths/min or less in a patient who is not in physiologic sleep strongly suggests acute opioid intoxication, particularly when accompanied by miosis or stupor. Other signs include bradycardia, hypotension, and seizures from anoxia.10
Apart from the severity of symptoms, it is hard to clinically distinguish fentanyl overdose from other opiate overdose incidents. Given the degree to which illegal opiates are contaminated with fentanyl in the United States,3 it is appropriate to screen for fentanyl with extended panel urine toxicology testing in patients with suspected opioid overdose.
CORRESPONDENCE
Jaividhya Dasarathy, MD, 2500 MetroHealth Medical Center, Cleveland, OH 44109; [email protected]
CASE 1
Two days after reviving her boyfriend with naloxone, a woman and her 30-year-old boyfriend presented to our family medicine clinic. They explained that he had injected heroin and shortly thereafter he stopped breathing and his lips turned blue. The patient’s girlfriend did not call emergency medical services (EMS) at the time because she was afraid of getting arrested due to past incarceration for possession of illegal drugs. Instead, she revived him with naloxone that she found in his bag.
Both the patient and his girlfriend were scared and surprised by his “terrible reaction,” as he had previously purchased heroin from the same dealer and used the same dose without similar effects. However, the patient did note that the drug he purchased this time had a bright white tinge, when normally the drug was light yellow.
On physical examination, the patient’s heart rate and blood pressure were normal. There were needle track marks on both forearms, elbows, and upper arms. A laboratory workup obtained during this visit revealed anemia and a normal basic metabolic panel. A hepatitis C virus antibody test was positive, and a hepatic function panel revealed elevated transaminase levels. Urine toxicology was positive for opioids and negative for other substances.
CASE 2
A 58-year-old man with a history of chronic hepatitis C, polysubstance abuse, and schizophrenia was transported to the emergency department by EMS after his family found him unresponsive in his bedroom. The patient had agonal breathing when EMS arrived, so they administered naloxone (4 mg intranasal and 4 mg intravenous). His breathing improved, but his mental status did not. He was still obtunded upon arrival in the emergency department and vomited 4 tan-colored patches. The patient was tachycardic (heart rate, 108 beats/min), hypertensive (blood pressure, 189/95 mm Hg), and had rapid shallow breathing (respiratory rate, 38 breaths/min). He was intubated for airway protection, at which time 2 more tan-colored patches were removed from his pharynx.
Laboratory evaluation revealed an acute kidney injury with a high anion metabolic acidosis. A hepatic function panel showed elevated transaminase levels. Plasma acetaminophen and salicylate levels were normal. A computed tomography head scan was normal. Urine toxicology was negative for opioids but was positive for cocaine and benzodiazepines.
THE DIAGNOSIS
Opioid overdose caused the acute respiratory depression in both cases. In Case 1, the patient unknowingly overdosed on heroin laced with fentanyl, known as China White, which likely caused the drug’s bright white tinge. In Case 2, the patient’s overdose was the result of oral ingestion of fentanyl patches. (Limited urine toxicology was negative for opiates because fentanyl is a fully synthetic opioid that shows up only with a specific or extended assay. More on this in a bit.)
DISCUSSION
The fatal drug overdose epidemic in the United States is growing. From 2000 to 2014, the mortality rate from drug overdose increased by 137%, including a 200% increase in the rate of overdose deaths related to opioids (ie, pain medications, heroin).1 Between 2013 and 2014, the age-adjusted mortality rate related to methadone, a synthetic opioid, remained unchanged; however, age-adjusted mortality rates related to natural and semisynthetic opioid pain medications, heroin, and synthetic opioids other than methadone (eg, fentanyl) increased by 9%, 26%, and 80%, respectively. In 2014, a sharp increase in overdose deaths related to synthetic opioids other than methadone coincided with law enforcement reports of increased availability of illegal fentanyl; however, the toxicology panel used by coroners and medical examiners at that time could not distinguish between illegal and prescription fentanyl.1
Continue to: Among 70,237 drug overdose deaths...
Among 70,237 drug overdose deaths in the United States in 2017, 47,600 (67.8%) involved an opioid. From 2013 to 2017, drug overdose death rates increased in 35 of 50 states and the District of Columbia, and significant increases in death rates involving synthetic opioids occurred in 15 out of 20 states, likely driven by illicitly manufactured fentanyl.2
Fentanyl-laced heroin: More common, but not new
In October 1991, 3-methylfentanyl was identified in 16 fatal drug overdoses in Allegheny County, Pennsylvania, contributing to a 4-fold increase in overdose deaths compared to the previous year. Fentanyl mixed with heroin and other drugs is commonly found in the Midwest, Northeast, and Southern regions of the United States; in 2014, more than 80% of fentanyl confiscations occurred in 10 states within these regions, with the highest incidence occurring in Ohio.3
When combined with fentanyl, heroin becomes 50 to 100 times more potent, resulting in a subjective high with exaggerated central nervous system depression manifesting as lethargy, miosis, and respiratory depression.4 Most drug users are unaware and unable to identify when heroin is laced with fentanyl, which may contribute to the rise in deaths from unintentional drug overdose.1,5,6
Oral abuse of fentanyl patches can be fatal
Outcomes from oral abuse of fentanyl patches have ranged from transient overdose symptoms, such as lethargy and respiratory depression, to death.7-9 When administered in a medical setting, transbuccal fentanyl has a bioavailability of 50% to 65% across the buccal membrane. Nearly 20% of the drug escapes hepatic first pass metabolism when fentanyl patches are ingested orally and enters the systemic circulation, resulting in severe overdose and potentially death. Prolonged chewing and sucking on fentanyl patches increases the contact time with the buccal membrane, resulting in increased systemic absorption compared to oral ingestion without chewing/sucking.7-9
Urine toxicology screening detects compounds based on a chemical assay for drugs—generally codeine, morphine, and their metabolites. Because fentanyl is a fully synthetic opioid, its structure is not
Continue to: Survival of fentanyl overdose depends on naloxone availability
Survival of fentanyl overdose depends on naloxone availability
Naloxone is a safe and effective antidote to an opioid overdose. It comes in 3 preparations, including intramuscular and subcutaneous injections and an intranasal spray.12 Concerns that naloxone will harm patients with opioid dependence are unfounded. Naloxone can induce symptoms of opioid withdrawal, such as yawning, lacrimation, piloerection, diaphoresis, myalgia, vomiting, and diarrhea. While these withdrawal symptoms are unpleasant, they are not life threatening.12 Due to its high potency, large doses of naloxone (ie, 4–16 mg) are required to reverse the effects of a fentanyl overdose.13 Intranasal naloxone hydrochloride 4 mg delivered in a single spray is preferred due to the ease of administration. Repeat doses may be necessary if respiratory depression continues or recurs prior to the arrival of emergency medical services. Increasing the availability of naloxone to first responders has the potential to save many lives.6
THE TAKEAWAY
Fentanyl is a major contributor to the growing drug overdose crisis in the United States. When laced with heroin or consumed orally in the form of transdermal patches, fentanyl becomes more potent and is increasingly fatal. It’s crucial that primary care physicians be able to identify and educate at-risk patients about the fatal consequences of fentanyl overdose and coordinate care to help get them into an appropriate rehabilitation program.
In order to quickly recognize the signs of fentanyl-related overdose, it’s important to be alert for this possibility. At the bedside, the most easily recognized abnormality associated with fentanyl or other opioid overdose is a decline in respiratory rate culminating in apnea.10 A respiratory rate of 12 breaths/min or less in a patient who is not in physiologic sleep strongly suggests acute opioid intoxication, particularly when accompanied by miosis or stupor. Other signs include bradycardia, hypotension, and seizures from anoxia.10
Apart from the severity of symptoms, it is hard to clinically distinguish fentanyl overdose from other opiate overdose incidents. Given the degree to which illegal opiates are contaminated with fentanyl in the United States,3 it is appropriate to screen for fentanyl with extended panel urine toxicology testing in patients with suspected opioid overdose.
CORRESPONDENCE
Jaividhya Dasarathy, MD, 2500 MetroHealth Medical Center, Cleveland, OH 44109; [email protected]
1. Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths—United States, 2000–2014. MMWR Morb Mortal Wkly Rep. 2016;64:1378-1382.
2. Scholl L, Seth P, Kariisa M, et al. Drug and opioid-involved overdose deaths—United States, 2013–2017. MMWR Morb Mortal Wkly Rep. 2019;67:1419-1427.
3. Hibbs J, Perper J, Winek CL. An outbreak of designer drug-related deaths in Pennsylvania. JAMA. 1991;265:1011-1013.
4. Increases in fentanyl drug confiscations and fentanyl-related overdose fatalities. Centers for Disease Control and Prevention Web site. https://emergency.cdc.gov/han/han00384.asp. Published October 26, 2015. Accessed May 3, 2019.
5. Fentanyl. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/drugoverdose/opioids/fentanyl.html. Updated December 19, 2018. Accessed May 3, 2019.
6. Peterson AB, Gladden RM, Delcher C, et al. Increases in fentanyl-related overdose deaths—Florida and Ohio, 2013–2015. MMWR Morb Mortal Wkly Rep. 2016;65:844-849.
7. Streisand JB, Varvel JR, Stanski DR, et al. Absorption and bioavailability of oral transmucosal fentanyl citrate. Anesthesiology. 1991;75:223-229.
8. Kharasch ED, Whittington D, Hoffer C. Influence of hepatic and intestinal cytochrome P4503A activity on the acute disposition and effects of oral transmucosal fentanyl citrate. Anesthesiology. 2004;101:729-737.
9. Woodall KL, Martin TL, McLellan BA. Oral abuse of fentanyl patches (Duragesic): seven case reports. J Forensic Sci. 2008;53:222-225.
10. Moeller KE, Lee KC, Kissack JC. Urine drug screening: practical guide for clinicians. Mayo Clin Proc. 2008;83:66-76.
11. Appropriate Use of Drug Testing in Clinical Addiction Medicine. American Society of Addiction Medicine Web site. https://www.asam.org/docs/default-source/quality-science/appropriate_use_of_drug_testing_in_clinical-1-(7).pdf?sfvrsn=2. Published April 5, 2017. Accessed May 30, 2019.
12. Boyer EW. Management of opioid analgesic overdose. N Engl J Med. 2012;367:146-155.
13. Drugs@FDA: FDA approved drug products. US Food and Drug Administration Web site. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=208411. Accessed May 22, 2019.
1. Rudd RA, Aleshire N, Zibbell JE, et al. Increases in drug and opioid overdose deaths—United States, 2000–2014. MMWR Morb Mortal Wkly Rep. 2016;64:1378-1382.
2. Scholl L, Seth P, Kariisa M, et al. Drug and opioid-involved overdose deaths—United States, 2013–2017. MMWR Morb Mortal Wkly Rep. 2019;67:1419-1427.
3. Hibbs J, Perper J, Winek CL. An outbreak of designer drug-related deaths in Pennsylvania. JAMA. 1991;265:1011-1013.
4. Increases in fentanyl drug confiscations and fentanyl-related overdose fatalities. Centers for Disease Control and Prevention Web site. https://emergency.cdc.gov/han/han00384.asp. Published October 26, 2015. Accessed May 3, 2019.
5. Fentanyl. Centers for Disease Control and Prevention Web site. https://www.cdc.gov/drugoverdose/opioids/fentanyl.html. Updated December 19, 2018. Accessed May 3, 2019.
6. Peterson AB, Gladden RM, Delcher C, et al. Increases in fentanyl-related overdose deaths—Florida and Ohio, 2013–2015. MMWR Morb Mortal Wkly Rep. 2016;65:844-849.
7. Streisand JB, Varvel JR, Stanski DR, et al. Absorption and bioavailability of oral transmucosal fentanyl citrate. Anesthesiology. 1991;75:223-229.
8. Kharasch ED, Whittington D, Hoffer C. Influence of hepatic and intestinal cytochrome P4503A activity on the acute disposition and effects of oral transmucosal fentanyl citrate. Anesthesiology. 2004;101:729-737.
9. Woodall KL, Martin TL, McLellan BA. Oral abuse of fentanyl patches (Duragesic): seven case reports. J Forensic Sci. 2008;53:222-225.
10. Moeller KE, Lee KC, Kissack JC. Urine drug screening: practical guide for clinicians. Mayo Clin Proc. 2008;83:66-76.
11. Appropriate Use of Drug Testing in Clinical Addiction Medicine. American Society of Addiction Medicine Web site. https://www.asam.org/docs/default-source/quality-science/appropriate_use_of_drug_testing_in_clinical-1-(7).pdf?sfvrsn=2. Published April 5, 2017. Accessed May 30, 2019.
12. Boyer EW. Management of opioid analgesic overdose. N Engl J Med. 2012;367:146-155.
13. Drugs@FDA: FDA approved drug products. US Food and Drug Administration Web site. https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm?event=overview.process&ApplNo=208411. Accessed May 22, 2019.
A better approach to the diagnosis of PE
ILLUSTRATIVE CASE
Penny E is a 48-year-old woman with a history of asthma who presents with wheezing and respiratory distress. There are no clinical signs of deep vein thrombosis or hemoptysis. Pulmonary embolism (PE) is not your most likely diagnosis, but it is included in the differential, so you order a D-dimer concentration and it returns at 700 ng/mL. Should you order computed tomography pulmonary angiography (CTPA) to evaluate for PE?
PE is the third most common type of cardiovascular disease after coronary artery disease and stroke, with an estimated incidence in the United States of 1-2 people/1000 population and a 30-day mortality rate between 10% and 30%.2 Improved adherence to a clinical decision support system has been shown to significantly decrease the number of diagnostic tests performed and the number of diagnostic failures.3
The use of a diagnostic algorithm that includes the Wells’ criteria and a
Further, it is common for a
Three items of the original Wells’ criteria—clinical signs of deep vein thrombosis, hemoptysis, and whether PE is the most likely diagnosis—are the most predictive for PE.8 The development of a more efficient algorithm based on these 3 items that uses differential D
STUDY SUMMARY
Simplified algorithm diagnoses PE with fewer CTPAs
The YEARS study was a prospective cohort study conducted in 12 hospitals in the Netherlands that included 3616 patients with clinically suspected PE.1 After excluding 151 patients who met exclusion criteria (life expectancy < 3 months, ongoing anticoagulation treatment, pregnancy, and contraindication to CTPA), investigators managed 3465 study patients according to the YEARS algorithm. This algorithm called for obtaining a
Of the 1743 patients who had none of the 3 YEARS items, 1320 had a
Continue to: Eighteen of the 2964 patients...
Eighteen of the 2964 patients who had PE ruled out by the YEARS algorithm at baseline were found to have symptomatic VTE during the follow-up period (0.61%; 95% CI, 0.36-0.96), with 6 patients (0.20%; 95% CI, 0.07-0.44) sustaining a fatal PE. The 3-month incidence of VTE in patients who did not have CTPA was 0.43% (95% CI, 0.17-0.88), which is similar to the 0.34% (0.036-0.96) reported in a previous meta-analysis of the Wells’ rule algorithm.13 Overall, fatal PE occurred in 0.3% (95% CI, 0.12-0.78) of patients in the YEARS cohort vs 0.6% (0.4-1.1) in a meta-analysis of studies using standard algorithms.14
Using an intention-to-diagnose analysis, 1611 (46%) patients did not have a CTPA indicated by the YEARS algorithm compared with 1174 (34%) using the Wells’ algorithm, for an absolute difference of 13% (95% CI, 10-15) and estimated cost savings of $283,176 in this sample. The per-protocol analysis also had a decrease of CTPA examinations in favor of the YEARS algorithm, ruling out 1651 (48%) patients—a decrease of 14% (95% CI, 12-16) and an estimated savings of $309,096.
WHAT’S NEW
High-level evidence says 14% fewer CTPAs
The YEARS study provides a high level of evidence that a new, simple diagnostic algorithm can reliably and efficiently exclude PE and decrease the need for CTPA by 14% (absolute difference; 95% CI, 12-16) when compared with using the Wells’ rule and fixed
CAVEATS
No adjusting D -dimer for age
The YEARS criteria does not consider an age-adjusted
CHALLENGES TO IMPLEMENTATION
None to speak of
We see no challenges to the implementation of this recommendation.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. van der Hulle T, Cheung WY, Kooij S, et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017;390:289-297.
2. Beckman MG, Hooper WC, Critchley SE, et al. Venous thromboembolism: a public health concern. Am J Prev Med. 2010;38:S495-S501.
3. Douma RA, Mos ICM, Erkens PMG, et al. Performance of 4 clinical decision rules in the diagnostic management of acute pulmonary embolism. Ann Intern Med. 2011;154:709-718.
4. van Es N, van der Hulle T, van Es J, et al. Wells Rule and D-dimer testing to rule out pulmonary embolism. Ann Intern Med. 2016;165:253-261.
5. Roy P-M, Meyer G, Vielle B, et al. Appropriateness of diagnostic management and outcomes of suspected pulmonary embolism. Ann Intern Med. 2006;144:157-164.
6. Newnham M, Stone H, Summerfield R, et al. Performance of algorithms and pre-test probability scores is often overlooked in the diagnosis of pulmonary embolism. BMJ. 2013;346:f1557.
7. Righini M, Van Es J, Den Exter PL, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism. JAMA. 2014;311:1117-1124.
8. van Es J, Beenen LFM, Douma RA, et al. A simple decision rule including D-dimer to reduce the need for computed tomography scanning in patients with suspected pulmonary embolism. J Thromb Haemost. 2015;13:1428-1435.
9. Kooiman J, Klok FA, Mos ICM, et al. Incidence and predictors of contrast-induced nephropathy following CT-angiography for clinically suspected acute pulmonary embolism. J Thromb Haemost. 2010;8:409-411.
10. Sarma A, Heilbrun ME, Conner KE, et al. Radiation and chest CT scan examinations: what do we know? Chest. 2012;142:750-760.
11. Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169:2071-2077.
12. Verma K, Legnani C, Palareti G. Cost-minimization analysis of venous thromboembolism diagnosis: comparison of standalone imaging with a strategy incorporating D-dimer for exclusion of venous thromboembolism. Res Pract Thromb Haemost. 2017;1:57-61.
13. Pasha SM, Klok FA, Snoep JD, et al. Safety of excluding acute pulmonary embolism based on an unlikely clinical probability by the Wells rule and normal D-dimer concentration: a meta-analysis. Thromb Res. 2010;125:e123-e127.
14. Mos ICM, Klok FA, Kroft LJM, et al. Safety of ruling out acute pulmonary embolism by normal computed tomography pulmonary angiography in patients with an indication for computed tomography: systematic review and meta-analysis. J Thromb Haemost. 2009;7:1491-1498.
ILLUSTRATIVE CASE
Penny E is a 48-year-old woman with a history of asthma who presents with wheezing and respiratory distress. There are no clinical signs of deep vein thrombosis or hemoptysis. Pulmonary embolism (PE) is not your most likely diagnosis, but it is included in the differential, so you order a D-dimer concentration and it returns at 700 ng/mL. Should you order computed tomography pulmonary angiography (CTPA) to evaluate for PE?
PE is the third most common type of cardiovascular disease after coronary artery disease and stroke, with an estimated incidence in the United States of 1-2 people/1000 population and a 30-day mortality rate between 10% and 30%.2 Improved adherence to a clinical decision support system has been shown to significantly decrease the number of diagnostic tests performed and the number of diagnostic failures.3
The use of a diagnostic algorithm that includes the Wells’ criteria and a
Further, it is common for a
Three items of the original Wells’ criteria—clinical signs of deep vein thrombosis, hemoptysis, and whether PE is the most likely diagnosis—are the most predictive for PE.8 The development of a more efficient algorithm based on these 3 items that uses differential D
STUDY SUMMARY
Simplified algorithm diagnoses PE with fewer CTPAs
The YEARS study was a prospective cohort study conducted in 12 hospitals in the Netherlands that included 3616 patients with clinically suspected PE.1 After excluding 151 patients who met exclusion criteria (life expectancy < 3 months, ongoing anticoagulation treatment, pregnancy, and contraindication to CTPA), investigators managed 3465 study patients according to the YEARS algorithm. This algorithm called for obtaining a
Of the 1743 patients who had none of the 3 YEARS items, 1320 had a
Continue to: Eighteen of the 2964 patients...
Eighteen of the 2964 patients who had PE ruled out by the YEARS algorithm at baseline were found to have symptomatic VTE during the follow-up period (0.61%; 95% CI, 0.36-0.96), with 6 patients (0.20%; 95% CI, 0.07-0.44) sustaining a fatal PE. The 3-month incidence of VTE in patients who did not have CTPA was 0.43% (95% CI, 0.17-0.88), which is similar to the 0.34% (0.036-0.96) reported in a previous meta-analysis of the Wells’ rule algorithm.13 Overall, fatal PE occurred in 0.3% (95% CI, 0.12-0.78) of patients in the YEARS cohort vs 0.6% (0.4-1.1) in a meta-analysis of studies using standard algorithms.14
Using an intention-to-diagnose analysis, 1611 (46%) patients did not have a CTPA indicated by the YEARS algorithm compared with 1174 (34%) using the Wells’ algorithm, for an absolute difference of 13% (95% CI, 10-15) and estimated cost savings of $283,176 in this sample. The per-protocol analysis also had a decrease of CTPA examinations in favor of the YEARS algorithm, ruling out 1651 (48%) patients—a decrease of 14% (95% CI, 12-16) and an estimated savings of $309,096.
WHAT’S NEW
High-level evidence says 14% fewer CTPAs
The YEARS study provides a high level of evidence that a new, simple diagnostic algorithm can reliably and efficiently exclude PE and decrease the need for CTPA by 14% (absolute difference; 95% CI, 12-16) when compared with using the Wells’ rule and fixed
CAVEATS
No adjusting D -dimer for age
The YEARS criteria does not consider an age-adjusted
CHALLENGES TO IMPLEMENTATION
None to speak of
We see no challenges to the implementation of this recommendation.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
ILLUSTRATIVE CASE
Penny E is a 48-year-old woman with a history of asthma who presents with wheezing and respiratory distress. There are no clinical signs of deep vein thrombosis or hemoptysis. Pulmonary embolism (PE) is not your most likely diagnosis, but it is included in the differential, so you order a D-dimer concentration and it returns at 700 ng/mL. Should you order computed tomography pulmonary angiography (CTPA) to evaluate for PE?
PE is the third most common type of cardiovascular disease after coronary artery disease and stroke, with an estimated incidence in the United States of 1-2 people/1000 population and a 30-day mortality rate between 10% and 30%.2 Improved adherence to a clinical decision support system has been shown to significantly decrease the number of diagnostic tests performed and the number of diagnostic failures.3
The use of a diagnostic algorithm that includes the Wells’ criteria and a
Further, it is common for a
Three items of the original Wells’ criteria—clinical signs of deep vein thrombosis, hemoptysis, and whether PE is the most likely diagnosis—are the most predictive for PE.8 The development of a more efficient algorithm based on these 3 items that uses differential D
STUDY SUMMARY
Simplified algorithm diagnoses PE with fewer CTPAs
The YEARS study was a prospective cohort study conducted in 12 hospitals in the Netherlands that included 3616 patients with clinically suspected PE.1 After excluding 151 patients who met exclusion criteria (life expectancy < 3 months, ongoing anticoagulation treatment, pregnancy, and contraindication to CTPA), investigators managed 3465 study patients according to the YEARS algorithm. This algorithm called for obtaining a
Of the 1743 patients who had none of the 3 YEARS items, 1320 had a
Continue to: Eighteen of the 2964 patients...
Eighteen of the 2964 patients who had PE ruled out by the YEARS algorithm at baseline were found to have symptomatic VTE during the follow-up period (0.61%; 95% CI, 0.36-0.96), with 6 patients (0.20%; 95% CI, 0.07-0.44) sustaining a fatal PE. The 3-month incidence of VTE in patients who did not have CTPA was 0.43% (95% CI, 0.17-0.88), which is similar to the 0.34% (0.036-0.96) reported in a previous meta-analysis of the Wells’ rule algorithm.13 Overall, fatal PE occurred in 0.3% (95% CI, 0.12-0.78) of patients in the YEARS cohort vs 0.6% (0.4-1.1) in a meta-analysis of studies using standard algorithms.14
Using an intention-to-diagnose analysis, 1611 (46%) patients did not have a CTPA indicated by the YEARS algorithm compared with 1174 (34%) using the Wells’ algorithm, for an absolute difference of 13% (95% CI, 10-15) and estimated cost savings of $283,176 in this sample. The per-protocol analysis also had a decrease of CTPA examinations in favor of the YEARS algorithm, ruling out 1651 (48%) patients—a decrease of 14% (95% CI, 12-16) and an estimated savings of $309,096.
WHAT’S NEW
High-level evidence says 14% fewer CTPAs
The YEARS study provides a high level of evidence that a new, simple diagnostic algorithm can reliably and efficiently exclude PE and decrease the need for CTPA by 14% (absolute difference; 95% CI, 12-16) when compared with using the Wells’ rule and fixed
CAVEATS
No adjusting D -dimer for age
The YEARS criteria does not consider an age-adjusted
CHALLENGES TO IMPLEMENTATION
None to speak of
We see no challenges to the implementation of this recommendation.
ACKNOWLEDGEMENT
The PURLs Surveillance System was supported in part by Grant Number UL1RR024999 from the National Center For Research Resources, a Clinical Translational Science Award to the University of Chicago. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center For Research Resources or the National Institutes of Health.
1. van der Hulle T, Cheung WY, Kooij S, et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017;390:289-297.
2. Beckman MG, Hooper WC, Critchley SE, et al. Venous thromboembolism: a public health concern. Am J Prev Med. 2010;38:S495-S501.
3. Douma RA, Mos ICM, Erkens PMG, et al. Performance of 4 clinical decision rules in the diagnostic management of acute pulmonary embolism. Ann Intern Med. 2011;154:709-718.
4. van Es N, van der Hulle T, van Es J, et al. Wells Rule and D-dimer testing to rule out pulmonary embolism. Ann Intern Med. 2016;165:253-261.
5. Roy P-M, Meyer G, Vielle B, et al. Appropriateness of diagnostic management and outcomes of suspected pulmonary embolism. Ann Intern Med. 2006;144:157-164.
6. Newnham M, Stone H, Summerfield R, et al. Performance of algorithms and pre-test probability scores is often overlooked in the diagnosis of pulmonary embolism. BMJ. 2013;346:f1557.
7. Righini M, Van Es J, Den Exter PL, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism. JAMA. 2014;311:1117-1124.
8. van Es J, Beenen LFM, Douma RA, et al. A simple decision rule including D-dimer to reduce the need for computed tomography scanning in patients with suspected pulmonary embolism. J Thromb Haemost. 2015;13:1428-1435.
9. Kooiman J, Klok FA, Mos ICM, et al. Incidence and predictors of contrast-induced nephropathy following CT-angiography for clinically suspected acute pulmonary embolism. J Thromb Haemost. 2010;8:409-411.
10. Sarma A, Heilbrun ME, Conner KE, et al. Radiation and chest CT scan examinations: what do we know? Chest. 2012;142:750-760.
11. Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169:2071-2077.
12. Verma K, Legnani C, Palareti G. Cost-minimization analysis of venous thromboembolism diagnosis: comparison of standalone imaging with a strategy incorporating D-dimer for exclusion of venous thromboembolism. Res Pract Thromb Haemost. 2017;1:57-61.
13. Pasha SM, Klok FA, Snoep JD, et al. Safety of excluding acute pulmonary embolism based on an unlikely clinical probability by the Wells rule and normal D-dimer concentration: a meta-analysis. Thromb Res. 2010;125:e123-e127.
14. Mos ICM, Klok FA, Kroft LJM, et al. Safety of ruling out acute pulmonary embolism by normal computed tomography pulmonary angiography in patients with an indication for computed tomography: systematic review and meta-analysis. J Thromb Haemost. 2009;7:1491-1498.
1. van der Hulle T, Cheung WY, Kooij S, et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017;390:289-297.
2. Beckman MG, Hooper WC, Critchley SE, et al. Venous thromboembolism: a public health concern. Am J Prev Med. 2010;38:S495-S501.
3. Douma RA, Mos ICM, Erkens PMG, et al. Performance of 4 clinical decision rules in the diagnostic management of acute pulmonary embolism. Ann Intern Med. 2011;154:709-718.
4. van Es N, van der Hulle T, van Es J, et al. Wells Rule and D-dimer testing to rule out pulmonary embolism. Ann Intern Med. 2016;165:253-261.
5. Roy P-M, Meyer G, Vielle B, et al. Appropriateness of diagnostic management and outcomes of suspected pulmonary embolism. Ann Intern Med. 2006;144:157-164.
6. Newnham M, Stone H, Summerfield R, et al. Performance of algorithms and pre-test probability scores is often overlooked in the diagnosis of pulmonary embolism. BMJ. 2013;346:f1557.
7. Righini M, Van Es J, Den Exter PL, et al. Age-adjusted D-dimer cutoff levels to rule out pulmonary embolism. JAMA. 2014;311:1117-1124.
8. van Es J, Beenen LFM, Douma RA, et al. A simple decision rule including D-dimer to reduce the need for computed tomography scanning in patients with suspected pulmonary embolism. J Thromb Haemost. 2015;13:1428-1435.
9. Kooiman J, Klok FA, Mos ICM, et al. Incidence and predictors of contrast-induced nephropathy following CT-angiography for clinically suspected acute pulmonary embolism. J Thromb Haemost. 2010;8:409-411.
10. Sarma A, Heilbrun ME, Conner KE, et al. Radiation and chest CT scan examinations: what do we know? Chest. 2012;142:750-760.
11. Berrington de González A, Mahesh M, Kim KP, et al. Projected cancer risks from computed tomographic scans performed in the United States in 2007. Arch Intern Med. 2009;169:2071-2077.
12. Verma K, Legnani C, Palareti G. Cost-minimization analysis of venous thromboembolism diagnosis: comparison of standalone imaging with a strategy incorporating D-dimer for exclusion of venous thromboembolism. Res Pract Thromb Haemost. 2017;1:57-61.
13. Pasha SM, Klok FA, Snoep JD, et al. Safety of excluding acute pulmonary embolism based on an unlikely clinical probability by the Wells rule and normal D-dimer concentration: a meta-analysis. Thromb Res. 2010;125:e123-e127.
14. Mos ICM, Klok FA, Kroft LJM, et al. Safety of ruling out acute pulmonary embolism by normal computed tomography pulmonary angiography in patients with an indication for computed tomography: systematic review and meta-analysis. J Thromb Haemost. 2009;7:1491-1498.
PRACTICE CHANGER
Do not order computed tomography pulmonary angiography when evaluating patients for suspected pulmonary embolism unless: (1) the patient has a
STRENGTH OF RECOMMENDATION
A: Based on a prospective, multicenter, cohort study of 3616 patients with clinically suspected pulmonary embolism.1
van der Hulle T, Cheung WY, Kooij S, et al. Simplified diagnostic management of suspected pulmonary embolism (the YEARS study): a prospective, multicentre, cohort study. Lancet. 2017;390:289-297.
The case for behavioral health integration into primary care
In a typical primary care practice, detecting and managing mental health problems competes with other priorities such as treating acute physical illness, monitoring chronic disease, providing preventive health services, and assessing compliance with standards of care.1 These competing demands for a primary care provider’s time, paired with limited mental health resources in the community, may result in suboptimal behavioral health care.1-3 Even when referrals are made to
Approximately 30% of adults with physical disorders also have one or more behavioral health conditions, such as anxiety, panic, mood, or substance use disorders.6 Although physical and behavioral health conditions are inextricably linked, their assessment and treatment get separated into different silos.7 Given that fewer than 20% of depressed patients are seen by a psychiatrist or psychologist,8 the responsibility of providing mental health care often falls on the primary care physician.8,9
Efforts to improve the treatment of common mental disorders in primary care have traditionally focused on screening for these disorders, educating primary care providers, developing treatment guidelines, and referring patients to mental health specialty care.10 However, behavioral health integration offers another way forward.
WHAT IS BEHAVIORAL HEALTH INTEGRATION?
Behavioral health integration (BHI) in primary care refers to primary care physicians and behavioral health clinicians working in concert with patients to address their primary care and behavioral health needs.11
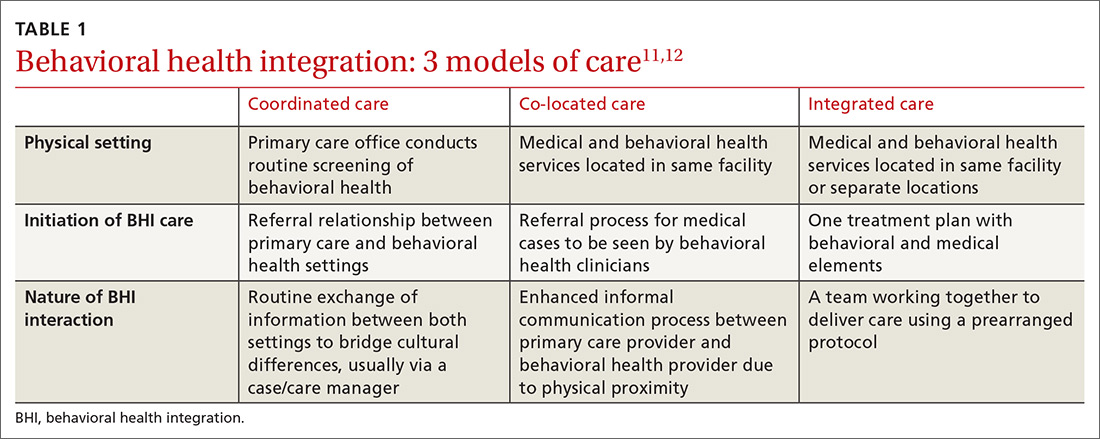
Numerous overlapping terms have been used to describe BHI, and this has caused some confusion. In 2013, the Agency for Healthcare Research and Quality (AHRQ) issued a lexicon standardizing the terminology used in BHI.11 The commonly used terms are

COORDINATED CARE AND THE COLLABORATIVE CARE MODEL
BHI at the level of coordinated care has almost exclusively been studied and practiced along the lines of the collaborative care model (CCM).14-16 This model represents an advanced level of coordinated care in the BHI continuum. The most substantial evidence for CCM lies in the management of depression and anxiety.14-16
Usual care involves the primary care physician and the patient. CCM adds 2 vital roles—a behavioral health care manager and a psychiatric consultant. A behavioral health care manager is typically a counselor, clinical social worker, psychologist, or psychiatric nurse who performs all care-management tasks including offering psychotherapy when that is part of the treatment plan.
Continue to: The care manager's functions include...
The care manager’s functions include systematic follow-up with structured monitoring of symptoms and treatment adherence, coordination and communication among care providers, patient education, and self-management support, including the use of motivational interviewing. The behavioral health care manager performs this systematic follow up by maintaining a patient “registry”—case-management software used in conjunction with, or embedded in, the practice electronic health record to track patients’ data and clinical outcomes, as well as to facilitate decision-making.
The care manager communicates with the psychiatrist, who offers suggestions for drug therapy, which is prescribed by the primary care physician. The care manager also regularly evaluates the patient’s status using a standardized scale, communicates these scores to the psychiatrist, and transmits any recommendations to the primary care physician (FIGURE 2).17
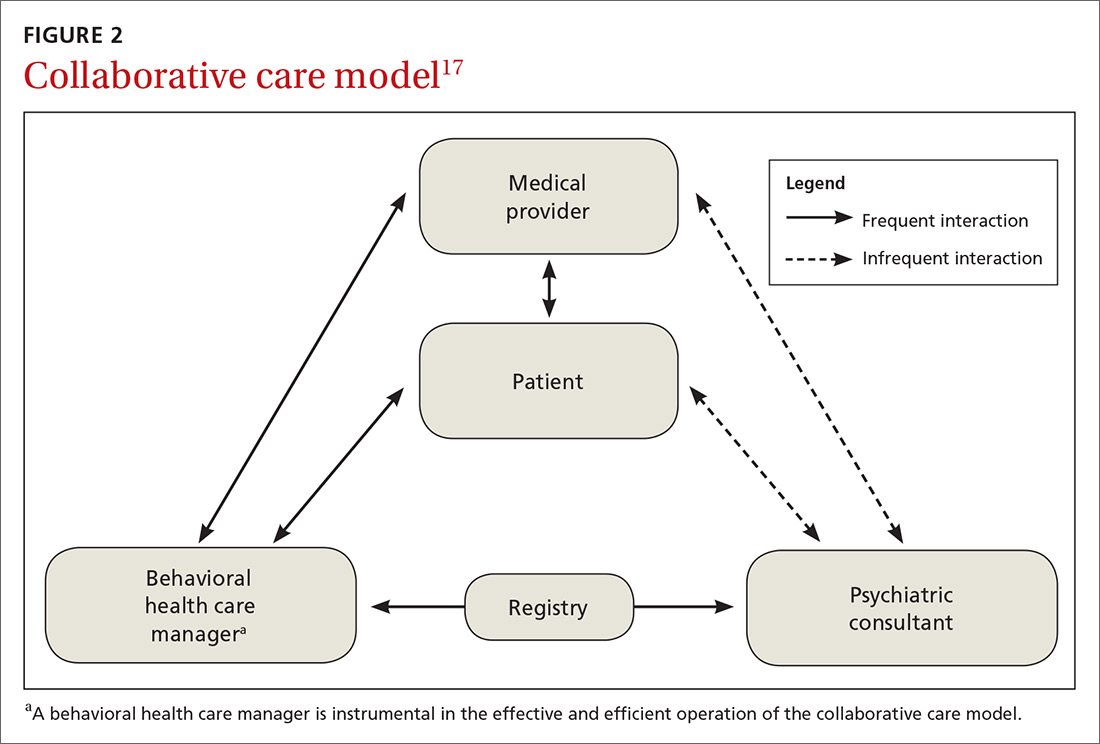
EVIDENCE FOR CCM
Collaborative and routine care were compared in a 2012 Cochrane review that included 79 randomized controlled trials (RCTs) involving 24,308 patients worldwide.16 Seventy-two of the 79 RCTs focused on patients with depression or depression with anxiety, while 6 studies included participants with only anxiety disorders.16 One additional study focused on mental health quality of life. (To learn about CCM and severe mental illness and substance use disorder, see “Less well studied: CCM and severe mental illness, alcohol dependence.”18-20)
SIDEBAR
Less well studied: CCM and severe mental illness, alcohol dependence
Evidence for collaborative care in severe mental illness (SMI) is very limited. SMI is defined as schizophrenia or other schizophrenia-like psychoses (eg, schizophreniform and schizoaffective disorders), bipolar affective disorder, or other psychosis.
A 2013 Cochrane review identified only 1 RCT involving 306 veterans with bipolar disease.18 The review concluded that there was low-quality evidence that collaborative care led to a relative risk reduction of 25% for psychiatric admissions at Year 2 compared with standard care (RR = 0.75; 95% CI, 0.57-0.99).18
One 2017 RCT involving 245 veterans that looked at a collaborative care model for patients with severe mental illness found a modest benefit for physical health-related quality of life, but did not find any benefit in mental health outcomes.19
Alcohol dependence. There is very limited, but high-quality, evidence for the utility of CCM in alcohol dependence. In one RCT, 163 veterans were assigned to either CCM or referral to standard treatment in a specialty outpatient addiction treatment program. The CCM group had a significantly higher proportion of participants engaged in treatment over the study’s 26 weeks (odds ratio [OR] = 5.36; 95% CI, 2.99-9.59). The percentage of heavy drinking days was significantly lower in the CCM group (OR = 2.16; 95% CI, 1.27-3.66), while overall abstinence did not differ between groups.20
For adults with depression treated with the CCM, significantly greater improvement in depression outcome measures was seen in the short-term (standardized mean difference [SMD] = -0.34; 95% confidence interval [CI], -0.41 to -0.27; risk ratio [RR] = 1.32; 95% CI, 1.22-1.43), in the medium term (SMD = -0.28; 95% CI, -0.41 to -0.15; RR = 1.31; 95% CI, 1.17–1.48), and in the long term (SMD = -0.35; 95% CI, -0.46 to -0.24; RR = 1.29; 95% CI, 1.18–1.41).16
Comparisons of mental health quality of life over the short term (0-6 months), medium term (7-12 months), and long term (13-24 months) did not show any significant difference between CCM and routine care.16 Comparisons of physical health quality of life over the short term and medium term did not show any significant difference between CCM and routine care.16
Continue to: Significantly greater improvement...
Significantly greater improvement in anxiety outcomes was seen for adults treated with CCM in the short term (SMD = -0.30; 95% CI, -0.44 to -0.17; RR = 1.50; 95% CI, 1.21–1.87), in the medium term (SMD = -0.33; 95% CI, -0.47 to -0.19; RR = 1.41; 95% CI, 1.18-1.69), and in the long term (SMD = -0.20; 95% CI, -0.34 to -0.06; RR = 1.26; 95% CI, 1.11–1.42).16
A 2016 systematic review of 94 RCTs involving more than 25,000 patients also provided high-quality evidence that collaborative care yields small-to-moderate improvements in symptoms from mood disorders and mental health-related quality of life.15 A 2006 meta-analysis of 37 RCTs comprising 12,355 patients showed that collaborative care involving a case manager is more effective than standard care in improving depression outcomes at 6 months (SMD = 0.25; 95% CI, 0.18-0.32) and up to 5 years (SMD = 0.15; 95% CI, 0.001-0.31).21
Better care of mental health disorders also improves medical outcomes
Several trials have focused on jointly managing depression and a chronic physical condition such as chronic pain, diabetes, and coronary heart disease,22 demonstrating improved outcomes for both depression and the comanaged conditions.
- Chronic pain. When compared with usual care, collaborative care resulted in moderate reductions in both pain severity and associated disability (41.5% vs 17.3%; RR = 2.4; 95% CI, 1.6-3.2).23
- Diabetes. Patients managed collaboratively were more likely to have a decrease of ≥ 1% in the glycated hemoglobin level from baseline (36% vs 19%; P = .006).24
- Cardiovascular disease. Significant real-world risk reduction was achieved by improving blood pressure control (58% achieved blood pressure control compared with a projected target of 20%).22
IS THERE A COMMON THREAD AMONG SUCCESSFUL CCMs?
Attempts to identify commonalities between the many iterations of successful CCMs have produced varying results due to differing selections of relevant RCTs.25-29 However, a few common features have been identified:
- care managers assess symptoms at baseline and at follow-up using a standardized measure such as the Patient Health Questionnaire (PHQ-9);
- care managers monitor treatment adherence;
- follow-up is active for at least 16 weeks;
- primary care and mental health providers actively engage in patient management; and
- mental health specialists regularly supervise care managers.
The one feature that is consistent with improved outcomes is the presence of the care manager.25-29
Continue to: The improvement associated...
The improvement associated with collaborative care is clinically meaningful to patients and physicians. In one RCT, collaborative care doubled response rates of depression treatment compared with usual care.3 Quality improvement data from real-world implementation of collaborative care programs suggests that similar outcomes can be achieved in a variety of settings.30
COST BENEFITS OF CCM
Collaborative care for depression is associated with lower health care costs.29,31
A meta-analysis of 57 RCTs in 2012 showed that CCM improves depression outcomes across populations, settings, and outcome domains, and that these results are achieved at little to no increase in treatment costs compared with usual care (Cohen’s d = 0.05; 95% CI, –0.02–0.12).26
When collaborative care was compared with routine care in an RCT involving 1801 primary care patients ≥ 60 years who were suffering from depression, a cost saving of $3363 per patient over 4 years was demonstrated in the intervention arm.31
A technical analysis of 94 RCTs in 2015 concluded that CCM is cost effective compared with usual care, with a range of $15,000 to $80,000 per quality-adjusted life year gained. These studies also indicated that organizations’ costs to implement CCM increase in the short term. Based on this analysis, organizations would need to invest between $3 to $22 per patient per month to implement and sustain CCMs, depending on the prevalence of depression in the population.29
Continue to: OTHER MODELS OF BHI
OTHER MODELS OF BHI
Higher levels of BHI such as co-location and integration do not have the same quality of evidence as CCM.
A 2009 Cochrane review of 42 studies involving 3880 patients found that mental health workers delivering psychological therapy and psychosocial interventions in primary care settings brought about significant reductions in primary care physician consultations (SMD = ‐0.17; 95% CI, ‐0.30 to ‐0.05); a relative risk reduction of 23% in psychotropic prescribing (RR = 0.67; 95% CI, 0.56–0.79); a decrease in prescribing costs (SMD = ‐0.22; 95% CI, ‐0.38 to ‐0.07); and a relative risk reduction in mental health referral of 87% (RR = 0.13; 95% CI, 0.09–0.20) for the patients they were seeing.32 The authors concluded the changes were modest in magnitude and inconsistent across different studies.32
Embedding medical providers in behavior health centers—ie, the reverse co-location model—also has very limited evidence. An RCT involving 120 veterans found that patients enrolled in a reverse co-location clinic did significantly better than controls seen in a general care clinic in terms of continuity of care and preventive care such as screening for hypertension (84.7% vs 65.6%; X 2 = 5.9, P = .01), diabetes (71.2% vs 45.9%; X 2 = 7.9, P < .005), hepatitis (39% vs 14.8%; X 2 = 9, P = .003), and cholesterol (79.7% vs 57.4%; X 2 = 6.9, P = .009).33
HOW TO IMPLEMENT A SUCCESSFUL BHI PROGRAM
A demonstration and evaluation project involving 11 diverse practices in Colorado explored ways to integrate behavioral health in primary care. Five main themes emerged34,35:
- Frame integrated care as a necessary paradigm shift to patient-centered, whole-person health care.
- Define relationships and protocols up front, understanding that they will evolve.
- Build inclusive, empowered teams to provide the foundation for integration.
- Develop a change management strategy of continuous evaluation and course correction.
- Use targeted data collection pertinent to integrated care to drive improvement and impart accountability.
The Institute for Clinical and Economic Review has organized an extensive list of resources36 for implementing BHI models, a sampling of which is shown in TABLE 2.

Continue to: TAKE-AWAY POINTS
TAKE-AWAY POINTS
There is high quality evidence that collaborative care works for the management of depression and anxiety disorder in primary care, and this is associated with significant cost savings.
CORRESPONDENCE
Rajesh (FNU) Rajesh, MD, Main Campus Family Medicine Clinic, MetroHealth, 2500 MetroHealth Drive, Cleveland, OH 44109; [email protected]
1. Rost K, Nutting P, Smith J, et al. The role of competing demands in the treatment provided primary care patients with major depression. Arch Fam Med. 2000;9:150-154.
2. Rush A, Trivedi M, Carmody T, et al. One-year clinical outcomes of depressed public sector outpatients: a benchmark for subsequent studies. Biol Psychiatry. 2004;56:46-53.
3. Unützer J, Katon W, Callahan CM, et al. Collaborative care management of late-life depression in the primary care setting. JAMA. 2002;288:2836-2845.
4. Department of Veterans Affairs. Bradford DW, Slubicki MN, McDuffie J, et al. Effects of care models to improve general medical outcomes for individuals with serious mental illness. 2011. https://www.hsrd.research.va.gov/publications/esp/smi-REPORT.pdf. Accessed August 22, 2018.
5. Druss BG, von Esenwein S. Improving general medical care for persons with mental and addictive disorders: systematic review. Gen Hosp Psychiatry. 2006;28:145-153.
6. Druss BG, Walker ER. Mental Disorders and Medical Comorbidity. Research Synthesis Report No. 21. Princeton, NJ: The Robert Wood Johnson Foundation; February 2011.
7. Reed SJ, Shore KK, Tice JA. Effectiveness and value of integrating behavioral health into primary care. JAMA Intern Med. 2016;176:691-692.
8. Young AS, Klap R, Sherbourne CD, et al. The quality of care for depressive and anxiety disorders in the United States. Arch Gen Psychiatry. 2001;58:55-61.
9. Butler M, Kane RL, McAlpine D, et al. Integration of mental health/substance abuse and primary care. Rockville, MD: Agency for Healthcare Research and Quality; 2008. http://www.ncbi.nlm.nih.gov/books/NBK38632/. Accessed March 2, 2019.
10. Unützer J, Schoenbaum M, Druss B, et al. Transforming mental health care at the interface with general medicine: report for the presidents commission. Psychiatr Serv. 2006;57:37-47. doi: 10.1176/appi.ps.57.1.37.
11. Peek CJ; the National Integration Academy Council. Lexicon for behavioral health and primary care integration: concepts and definitions developed by expert consensus. AHRQ. https://integrationacademy.ahrq.gov/sites/default/files/Lexicon.pdf. Published April 2013. Accessed May 29, 2019.
12. Heath B, Wise Romero P, Reynolds K. A standard framework for levels of integrated healthcare and update throughout the document. SAMHSA-HRSA. https://www.integration.samhsa.gov/integrated-care-models/A_Standard_Framework_for_Levels_of_Integrated_Healthcare.pdf. Published March 2013. Accessed May 29, 2019.
13. Integrating physical and behavioral health care: promising Medicaid models. The Henry J. Kaiser Family Foundation. https://www.kff.org/wp-content/uploads/2014/02/8553-integrating-physical-and-behavioral-health-care-promising-medicaid-models.pdf. Published February 2014. Accessed May 29, 2019.
14. Vanderlip ER, Rundell J, Avery M, et al. Dissemination of integrated care within adult primary care settings: the collaborative care model. SAMHSA-HRSA. https://www.integration.samhsa.gov/integrated-care-models/APA-APM-Dissemination-Integrated-Care-Report.pdf. Published 2016. Accessed May 29, 2019.
15. Gerrity M. Evolving models of behavioral health integration: evidence update 2010-2015. Milbank Memorial Fund. https://www.milbank.org/wp-content/uploads/2016/05/Evolving-Models-of-BHI.pdf. Published May 2016. Accessed May 29, 2019.
16. Archer J, Bower P, Gilbody S, et al. Collaborative care for depression and anxiety problems. Cochrane Database Syst Rev. 2012;10.1002/14651858.cd006525.pub2.
17. Team Structure. University of Washington AIMs Center. https://aims.uw.edu/collaborative-care/team-structure. Published 2017.Accessed May 29, 2019.
18. Reilly S, Planner C, Gask L, et al. Collaborative care approaches for people with severe mental illness. Cochrane Database Syst Rev. 2013;(11):CD009531.
19. Kilbourne AM, Barbaresso MM, Lai Z, et al. Improving physical health in patients with chronic mental disorders. J Clin Psychiatry. 2017;78:129-137.
20. Oslin DW, Lynch KG, Maisto HSA, et al. A randomized clinical trial of alcohol care management delivered in Department of Veterans Affairs primary care clinics versus specialty addiction treatment. J Gen Intern Med. 2013;29:162-168.
21. Gilbody S, Bower P, Fletcher J, et al. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes. Arch Intern Med. 2006;166:2314-2321.
22. Rossom RC, Solberg LI, Magnan S, et al. Impact of a national collaborative care initiative for patients with depression and diabetes or cardiovascular disease. Gen Hosp Psychiatry. 2017;15:77-85.
23. Kroenke K, Bair MJ, Damush TM, et al. Optimized antidepressant therapy and pain self-management in primary care patients with depression and musculoskeletal pain. JAMA. 2009;301:2009-2110.
24. Katon WJ, Lin EH, Von Korff M, et al. Collaborative care for patients with depression and chronic illnesses. N Engl J Med. 2010;363:2611-2620.
25. Miller CJ, Grogan-Kaylor A, Perron BE, et al. Collaborative chronic care models for mental health conditions. Med Care. 2013;51:922-930.
26. Woltmann E, Grogan-Kaylor A, Perron B, et al. Comparative effectiveness of collaborative chronic care models for mental health conditions across primary, specialty, and behavioral health care settings: systematic review and meta-analysis. Am J Psychiatry. 2012;11:790-804.
27. U.S. Department of Veterans Affairs. Rubenstein LV, Williams JW Jr, Danz M, et al. Determining key features of effective depression interventions. 2009. http://www.hsrd.research.va.gov/publications/esp/depinter.cfm. Accessed August 22, 2018.
28. Coventry PA, Hudson JL, Kontopantelis E, et al. Characteristics of effective collaborative care for treatment of depression: a systematic review and meta-regression of 74 randomised controlled trials. PLoS One. 2014;9:e108114.
29. Institute for Clinical and Economic Review. Tice JA, Ollendorf DA, Reed SJ, et al. Integrating behavioral health into primary care. 2015. https://icer-review.org/wp-content/uploads/2016/01/BHI_Final_Report_0602151.pdf. Accessed August 27, 2018.
30. Rubenstein LV, Chaney EF, Ober S, et al. Using evidence-based quality improvement methods for translating depression collaborative care research into practice. Fam Syst Health. 2010;28:91-113.
31. Unützer J, Katon WJ, Fan MY, et al. Long-term cost effects of collaborative care for late-life depression. Am J Manag Care. 2008;14:95-100.
32. Harkness EF, Bower PJ. On-site mental health workers delivering psychological therapy and psychosocial interventions to patients in primary care: effects on the professional practice of primary care providers. Cochrane Database Syst Rev. 2009;(1):CD000532.
33. Druss BG, Rohrbaugh RM, Levinson CM, et al. Integrated medical care for patients with serious psychiatric illness. Arch Gen Psychiatry. 2001;58:861-868.
34. Davis M, Balasubramanian BA, Waller E, et al. Integrating behavioral and physical health care in the real world: early lessons from advancing care together. J Am Board Fam Med. 2013;26:588-602.
35. Gold SB, Green LA, Peek CJ. From our practices to yours: key messages for the journey to integrated behavioral health. J Am Board Fam Med. 2017;30:25-34.
36. Institute for Clinical and Economic Review. Integrating behavioral health into primary care. 2015. https://icer-review.org/wp-content/uploads/2016/02/CTAF_BHI_Action_Guide_060215.pdf. Accessed April 25, 2019.
In a typical primary care practice, detecting and managing mental health problems competes with other priorities such as treating acute physical illness, monitoring chronic disease, providing preventive health services, and assessing compliance with standards of care.1 These competing demands for a primary care provider’s time, paired with limited mental health resources in the community, may result in suboptimal behavioral health care.1-3 Even when referrals are made to
Approximately 30% of adults with physical disorders also have one or more behavioral health conditions, such as anxiety, panic, mood, or substance use disorders.6 Although physical and behavioral health conditions are inextricably linked, their assessment and treatment get separated into different silos.7 Given that fewer than 20% of depressed patients are seen by a psychiatrist or psychologist,8 the responsibility of providing mental health care often falls on the primary care physician.8,9
Efforts to improve the treatment of common mental disorders in primary care have traditionally focused on screening for these disorders, educating primary care providers, developing treatment guidelines, and referring patients to mental health specialty care.10 However, behavioral health integration offers another way forward.
WHAT IS BEHAVIORAL HEALTH INTEGRATION?
Behavioral health integration (BHI) in primary care refers to primary care physicians and behavioral health clinicians working in concert with patients to address their primary care and behavioral health needs.11

Numerous overlapping terms have been used to describe BHI, and this has caused some confusion. In 2013, the Agency for Healthcare Research and Quality (AHRQ) issued a lexicon standardizing the terminology used in BHI.11 The commonly used terms are

COORDINATED CARE AND THE COLLABORATIVE CARE MODEL
BHI at the level of coordinated care has almost exclusively been studied and practiced along the lines of the collaborative care model (CCM).14-16 This model represents an advanced level of coordinated care in the BHI continuum. The most substantial evidence for CCM lies in the management of depression and anxiety.14-16
Usual care involves the primary care physician and the patient. CCM adds 2 vital roles—a behavioral health care manager and a psychiatric consultant. A behavioral health care manager is typically a counselor, clinical social worker, psychologist, or psychiatric nurse who performs all care-management tasks including offering psychotherapy when that is part of the treatment plan.
Continue to: The care manager's functions include...
The care manager’s functions include systematic follow-up with structured monitoring of symptoms and treatment adherence, coordination and communication among care providers, patient education, and self-management support, including the use of motivational interviewing. The behavioral health care manager performs this systematic follow up by maintaining a patient “registry”—case-management software used in conjunction with, or embedded in, the practice electronic health record to track patients’ data and clinical outcomes, as well as to facilitate decision-making.
The care manager communicates with the psychiatrist, who offers suggestions for drug therapy, which is prescribed by the primary care physician. The care manager also regularly evaluates the patient’s status using a standardized scale, communicates these scores to the psychiatrist, and transmits any recommendations to the primary care physician (FIGURE 2).17

EVIDENCE FOR CCM
Collaborative and routine care were compared in a 2012 Cochrane review that included 79 randomized controlled trials (RCTs) involving 24,308 patients worldwide.16 Seventy-two of the 79 RCTs focused on patients with depression or depression with anxiety, while 6 studies included participants with only anxiety disorders.16 One additional study focused on mental health quality of life. (To learn about CCM and severe mental illness and substance use disorder, see “Less well studied: CCM and severe mental illness, alcohol dependence.”18-20)
SIDEBAR
Less well studied: CCM and severe mental illness, alcohol dependence
Evidence for collaborative care in severe mental illness (SMI) is very limited. SMI is defined as schizophrenia or other schizophrenia-like psychoses (eg, schizophreniform and schizoaffective disorders), bipolar affective disorder, or other psychosis.
A 2013 Cochrane review identified only 1 RCT involving 306 veterans with bipolar disease.18 The review concluded that there was low-quality evidence that collaborative care led to a relative risk reduction of 25% for psychiatric admissions at Year 2 compared with standard care (RR = 0.75; 95% CI, 0.57-0.99).18
One 2017 RCT involving 245 veterans that looked at a collaborative care model for patients with severe mental illness found a modest benefit for physical health-related quality of life, but did not find any benefit in mental health outcomes.19
Alcohol dependence. There is very limited, but high-quality, evidence for the utility of CCM in alcohol dependence. In one RCT, 163 veterans were assigned to either CCM or referral to standard treatment in a specialty outpatient addiction treatment program. The CCM group had a significantly higher proportion of participants engaged in treatment over the study’s 26 weeks (odds ratio [OR] = 5.36; 95% CI, 2.99-9.59). The percentage of heavy drinking days was significantly lower in the CCM group (OR = 2.16; 95% CI, 1.27-3.66), while overall abstinence did not differ between groups.20
For adults with depression treated with the CCM, significantly greater improvement in depression outcome measures was seen in the short-term (standardized mean difference [SMD] = -0.34; 95% confidence interval [CI], -0.41 to -0.27; risk ratio [RR] = 1.32; 95% CI, 1.22-1.43), in the medium term (SMD = -0.28; 95% CI, -0.41 to -0.15; RR = 1.31; 95% CI, 1.17–1.48), and in the long term (SMD = -0.35; 95% CI, -0.46 to -0.24; RR = 1.29; 95% CI, 1.18–1.41).16
Comparisons of mental health quality of life over the short term (0-6 months), medium term (7-12 months), and long term (13-24 months) did not show any significant difference between CCM and routine care.16 Comparisons of physical health quality of life over the short term and medium term did not show any significant difference between CCM and routine care.16
Continue to: Significantly greater improvement...
Significantly greater improvement in anxiety outcomes was seen for adults treated with CCM in the short term (SMD = -0.30; 95% CI, -0.44 to -0.17; RR = 1.50; 95% CI, 1.21–1.87), in the medium term (SMD = -0.33; 95% CI, -0.47 to -0.19; RR = 1.41; 95% CI, 1.18-1.69), and in the long term (SMD = -0.20; 95% CI, -0.34 to -0.06; RR = 1.26; 95% CI, 1.11–1.42).16
A 2016 systematic review of 94 RCTs involving more than 25,000 patients also provided high-quality evidence that collaborative care yields small-to-moderate improvements in symptoms from mood disorders and mental health-related quality of life.15 A 2006 meta-analysis of 37 RCTs comprising 12,355 patients showed that collaborative care involving a case manager is more effective than standard care in improving depression outcomes at 6 months (SMD = 0.25; 95% CI, 0.18-0.32) and up to 5 years (SMD = 0.15; 95% CI, 0.001-0.31).21
Better care of mental health disorders also improves medical outcomes
Several trials have focused on jointly managing depression and a chronic physical condition such as chronic pain, diabetes, and coronary heart disease,22 demonstrating improved outcomes for both depression and the comanaged conditions.
- Chronic pain. When compared with usual care, collaborative care resulted in moderate reductions in both pain severity and associated disability (41.5% vs 17.3%; RR = 2.4; 95% CI, 1.6-3.2).23
- Diabetes. Patients managed collaboratively were more likely to have a decrease of ≥ 1% in the glycated hemoglobin level from baseline (36% vs 19%; P = .006).24
- Cardiovascular disease. Significant real-world risk reduction was achieved by improving blood pressure control (58% achieved blood pressure control compared with a projected target of 20%).22
IS THERE A COMMON THREAD AMONG SUCCESSFUL CCMs?
Attempts to identify commonalities between the many iterations of successful CCMs have produced varying results due to differing selections of relevant RCTs.25-29 However, a few common features have been identified:
- care managers assess symptoms at baseline and at follow-up using a standardized measure such as the Patient Health Questionnaire (PHQ-9);
- care managers monitor treatment adherence;
- follow-up is active for at least 16 weeks;
- primary care and mental health providers actively engage in patient management; and
- mental health specialists regularly supervise care managers.
The one feature that is consistent with improved outcomes is the presence of the care manager.25-29
Continue to: The improvement associated...
The improvement associated with collaborative care is clinically meaningful to patients and physicians. In one RCT, collaborative care doubled response rates of depression treatment compared with usual care.3 Quality improvement data from real-world implementation of collaborative care programs suggests that similar outcomes can be achieved in a variety of settings.30
COST BENEFITS OF CCM
Collaborative care for depression is associated with lower health care costs.29,31
A meta-analysis of 57 RCTs in 2012 showed that CCM improves depression outcomes across populations, settings, and outcome domains, and that these results are achieved at little to no increase in treatment costs compared with usual care (Cohen’s d = 0.05; 95% CI, –0.02–0.12).26
When collaborative care was compared with routine care in an RCT involving 1801 primary care patients ≥ 60 years who were suffering from depression, a cost saving of $3363 per patient over 4 years was demonstrated in the intervention arm.31
A technical analysis of 94 RCTs in 2015 concluded that CCM is cost effective compared with usual care, with a range of $15,000 to $80,000 per quality-adjusted life year gained. These studies also indicated that organizations’ costs to implement CCM increase in the short term. Based on this analysis, organizations would need to invest between $3 to $22 per patient per month to implement and sustain CCMs, depending on the prevalence of depression in the population.29
Continue to: OTHER MODELS OF BHI
OTHER MODELS OF BHI
Higher levels of BHI such as co-location and integration do not have the same quality of evidence as CCM.
A 2009 Cochrane review of 42 studies involving 3880 patients found that mental health workers delivering psychological therapy and psychosocial interventions in primary care settings brought about significant reductions in primary care physician consultations (SMD = ‐0.17; 95% CI, ‐0.30 to ‐0.05); a relative risk reduction of 23% in psychotropic prescribing (RR = 0.67; 95% CI, 0.56–0.79); a decrease in prescribing costs (SMD = ‐0.22; 95% CI, ‐0.38 to ‐0.07); and a relative risk reduction in mental health referral of 87% (RR = 0.13; 95% CI, 0.09–0.20) for the patients they were seeing.32 The authors concluded the changes were modest in magnitude and inconsistent across different studies.32
Embedding medical providers in behavior health centers—ie, the reverse co-location model—also has very limited evidence. An RCT involving 120 veterans found that patients enrolled in a reverse co-location clinic did significantly better than controls seen in a general care clinic in terms of continuity of care and preventive care such as screening for hypertension (84.7% vs 65.6%; X 2 = 5.9, P = .01), diabetes (71.2% vs 45.9%; X 2 = 7.9, P < .005), hepatitis (39% vs 14.8%; X 2 = 9, P = .003), and cholesterol (79.7% vs 57.4%; X 2 = 6.9, P = .009).33
HOW TO IMPLEMENT A SUCCESSFUL BHI PROGRAM
A demonstration and evaluation project involving 11 diverse practices in Colorado explored ways to integrate behavioral health in primary care. Five main themes emerged34,35:
- Frame integrated care as a necessary paradigm shift to patient-centered, whole-person health care.
- Define relationships and protocols up front, understanding that they will evolve.
- Build inclusive, empowered teams to provide the foundation for integration.
- Develop a change management strategy of continuous evaluation and course correction.
- Use targeted data collection pertinent to integrated care to drive improvement and impart accountability.
The Institute for Clinical and Economic Review has organized an extensive list of resources36 for implementing BHI models, a sampling of which is shown in TABLE 2.

Continue to: TAKE-AWAY POINTS
TAKE-AWAY POINTS
There is high quality evidence that collaborative care works for the management of depression and anxiety disorder in primary care, and this is associated with significant cost savings.
CORRESPONDENCE
Rajesh (FNU) Rajesh, MD, Main Campus Family Medicine Clinic, MetroHealth, 2500 MetroHealth Drive, Cleveland, OH 44109; [email protected]
In a typical primary care practice, detecting and managing mental health problems competes with other priorities such as treating acute physical illness, monitoring chronic disease, providing preventive health services, and assessing compliance with standards of care.1 These competing demands for a primary care provider’s time, paired with limited mental health resources in the community, may result in suboptimal behavioral health care.1-3 Even when referrals are made to
Approximately 30% of adults with physical disorders also have one or more behavioral health conditions, such as anxiety, panic, mood, or substance use disorders.6 Although physical and behavioral health conditions are inextricably linked, their assessment and treatment get separated into different silos.7 Given that fewer than 20% of depressed patients are seen by a psychiatrist or psychologist,8 the responsibility of providing mental health care often falls on the primary care physician.8,9
Efforts to improve the treatment of common mental disorders in primary care have traditionally focused on screening for these disorders, educating primary care providers, developing treatment guidelines, and referring patients to mental health specialty care.10 However, behavioral health integration offers another way forward.
WHAT IS BEHAVIORAL HEALTH INTEGRATION?
Behavioral health integration (BHI) in primary care refers to primary care physicians and behavioral health clinicians working in concert with patients to address their primary care and behavioral health needs.11

Numerous overlapping terms have been used to describe BHI, and this has caused some confusion. In 2013, the Agency for Healthcare Research and Quality (AHRQ) issued a lexicon standardizing the terminology used in BHI.11 The commonly used terms are

COORDINATED CARE AND THE COLLABORATIVE CARE MODEL
BHI at the level of coordinated care has almost exclusively been studied and practiced along the lines of the collaborative care model (CCM).14-16 This model represents an advanced level of coordinated care in the BHI continuum. The most substantial evidence for CCM lies in the management of depression and anxiety.14-16
Usual care involves the primary care physician and the patient. CCM adds 2 vital roles—a behavioral health care manager and a psychiatric consultant. A behavioral health care manager is typically a counselor, clinical social worker, psychologist, or psychiatric nurse who performs all care-management tasks including offering psychotherapy when that is part of the treatment plan.
Continue to: The care manager's functions include...
The care manager’s functions include systematic follow-up with structured monitoring of symptoms and treatment adherence, coordination and communication among care providers, patient education, and self-management support, including the use of motivational interviewing. The behavioral health care manager performs this systematic follow up by maintaining a patient “registry”—case-management software used in conjunction with, or embedded in, the practice electronic health record to track patients’ data and clinical outcomes, as well as to facilitate decision-making.
The care manager communicates with the psychiatrist, who offers suggestions for drug therapy, which is prescribed by the primary care physician. The care manager also regularly evaluates the patient’s status using a standardized scale, communicates these scores to the psychiatrist, and transmits any recommendations to the primary care physician (FIGURE 2).17

EVIDENCE FOR CCM
Collaborative and routine care were compared in a 2012 Cochrane review that included 79 randomized controlled trials (RCTs) involving 24,308 patients worldwide.16 Seventy-two of the 79 RCTs focused on patients with depression or depression with anxiety, while 6 studies included participants with only anxiety disorders.16 One additional study focused on mental health quality of life. (To learn about CCM and severe mental illness and substance use disorder, see “Less well studied: CCM and severe mental illness, alcohol dependence.”18-20)
SIDEBAR
Less well studied: CCM and severe mental illness, alcohol dependence
Evidence for collaborative care in severe mental illness (SMI) is very limited. SMI is defined as schizophrenia or other schizophrenia-like psychoses (eg, schizophreniform and schizoaffective disorders), bipolar affective disorder, or other psychosis.
A 2013 Cochrane review identified only 1 RCT involving 306 veterans with bipolar disease.18 The review concluded that there was low-quality evidence that collaborative care led to a relative risk reduction of 25% for psychiatric admissions at Year 2 compared with standard care (RR = 0.75; 95% CI, 0.57-0.99).18
One 2017 RCT involving 245 veterans that looked at a collaborative care model for patients with severe mental illness found a modest benefit for physical health-related quality of life, but did not find any benefit in mental health outcomes.19
Alcohol dependence. There is very limited, but high-quality, evidence for the utility of CCM in alcohol dependence. In one RCT, 163 veterans were assigned to either CCM or referral to standard treatment in a specialty outpatient addiction treatment program. The CCM group had a significantly higher proportion of participants engaged in treatment over the study’s 26 weeks (odds ratio [OR] = 5.36; 95% CI, 2.99-9.59). The percentage of heavy drinking days was significantly lower in the CCM group (OR = 2.16; 95% CI, 1.27-3.66), while overall abstinence did not differ between groups.20
For adults with depression treated with the CCM, significantly greater improvement in depression outcome measures was seen in the short-term (standardized mean difference [SMD] = -0.34; 95% confidence interval [CI], -0.41 to -0.27; risk ratio [RR] = 1.32; 95% CI, 1.22-1.43), in the medium term (SMD = -0.28; 95% CI, -0.41 to -0.15; RR = 1.31; 95% CI, 1.17–1.48), and in the long term (SMD = -0.35; 95% CI, -0.46 to -0.24; RR = 1.29; 95% CI, 1.18–1.41).16
Comparisons of mental health quality of life over the short term (0-6 months), medium term (7-12 months), and long term (13-24 months) did not show any significant difference between CCM and routine care.16 Comparisons of physical health quality of life over the short term and medium term did not show any significant difference between CCM and routine care.16
Continue to: Significantly greater improvement...
Significantly greater improvement in anxiety outcomes was seen for adults treated with CCM in the short term (SMD = -0.30; 95% CI, -0.44 to -0.17; RR = 1.50; 95% CI, 1.21–1.87), in the medium term (SMD = -0.33; 95% CI, -0.47 to -0.19; RR = 1.41; 95% CI, 1.18-1.69), and in the long term (SMD = -0.20; 95% CI, -0.34 to -0.06; RR = 1.26; 95% CI, 1.11–1.42).16
A 2016 systematic review of 94 RCTs involving more than 25,000 patients also provided high-quality evidence that collaborative care yields small-to-moderate improvements in symptoms from mood disorders and mental health-related quality of life.15 A 2006 meta-analysis of 37 RCTs comprising 12,355 patients showed that collaborative care involving a case manager is more effective than standard care in improving depression outcomes at 6 months (SMD = 0.25; 95% CI, 0.18-0.32) and up to 5 years (SMD = 0.15; 95% CI, 0.001-0.31).21
Better care of mental health disorders also improves medical outcomes
Several trials have focused on jointly managing depression and a chronic physical condition such as chronic pain, diabetes, and coronary heart disease,22 demonstrating improved outcomes for both depression and the comanaged conditions.
- Chronic pain. When compared with usual care, collaborative care resulted in moderate reductions in both pain severity and associated disability (41.5% vs 17.3%; RR = 2.4; 95% CI, 1.6-3.2).23
- Diabetes. Patients managed collaboratively were more likely to have a decrease of ≥ 1% in the glycated hemoglobin level from baseline (36% vs 19%; P = .006).24
- Cardiovascular disease. Significant real-world risk reduction was achieved by improving blood pressure control (58% achieved blood pressure control compared with a projected target of 20%).22
IS THERE A COMMON THREAD AMONG SUCCESSFUL CCMs?
Attempts to identify commonalities between the many iterations of successful CCMs have produced varying results due to differing selections of relevant RCTs.25-29 However, a few common features have been identified:
- care managers assess symptoms at baseline and at follow-up using a standardized measure such as the Patient Health Questionnaire (PHQ-9);
- care managers monitor treatment adherence;
- follow-up is active for at least 16 weeks;
- primary care and mental health providers actively engage in patient management; and
- mental health specialists regularly supervise care managers.
The one feature that is consistent with improved outcomes is the presence of the care manager.25-29
Continue to: The improvement associated...
The improvement associated with collaborative care is clinically meaningful to patients and physicians. In one RCT, collaborative care doubled response rates of depression treatment compared with usual care.3 Quality improvement data from real-world implementation of collaborative care programs suggests that similar outcomes can be achieved in a variety of settings.30
COST BENEFITS OF CCM
Collaborative care for depression is associated with lower health care costs.29,31
A meta-analysis of 57 RCTs in 2012 showed that CCM improves depression outcomes across populations, settings, and outcome domains, and that these results are achieved at little to no increase in treatment costs compared with usual care (Cohen’s d = 0.05; 95% CI, –0.02–0.12).26
When collaborative care was compared with routine care in an RCT involving 1801 primary care patients ≥ 60 years who were suffering from depression, a cost saving of $3363 per patient over 4 years was demonstrated in the intervention arm.31
A technical analysis of 94 RCTs in 2015 concluded that CCM is cost effective compared with usual care, with a range of $15,000 to $80,000 per quality-adjusted life year gained. These studies also indicated that organizations’ costs to implement CCM increase in the short term. Based on this analysis, organizations would need to invest between $3 to $22 per patient per month to implement and sustain CCMs, depending on the prevalence of depression in the population.29
Continue to: OTHER MODELS OF BHI
OTHER MODELS OF BHI
Higher levels of BHI such as co-location and integration do not have the same quality of evidence as CCM.
A 2009 Cochrane review of 42 studies involving 3880 patients found that mental health workers delivering psychological therapy and psychosocial interventions in primary care settings brought about significant reductions in primary care physician consultations (SMD = ‐0.17; 95% CI, ‐0.30 to ‐0.05); a relative risk reduction of 23% in psychotropic prescribing (RR = 0.67; 95% CI, 0.56–0.79); a decrease in prescribing costs (SMD = ‐0.22; 95% CI, ‐0.38 to ‐0.07); and a relative risk reduction in mental health referral of 87% (RR = 0.13; 95% CI, 0.09–0.20) for the patients they were seeing.32 The authors concluded the changes were modest in magnitude and inconsistent across different studies.32
Embedding medical providers in behavior health centers—ie, the reverse co-location model—also has very limited evidence. An RCT involving 120 veterans found that patients enrolled in a reverse co-location clinic did significantly better than controls seen in a general care clinic in terms of continuity of care and preventive care such as screening for hypertension (84.7% vs 65.6%; X 2 = 5.9, P = .01), diabetes (71.2% vs 45.9%; X 2 = 7.9, P < .005), hepatitis (39% vs 14.8%; X 2 = 9, P = .003), and cholesterol (79.7% vs 57.4%; X 2 = 6.9, P = .009).33
HOW TO IMPLEMENT A SUCCESSFUL BHI PROGRAM
A demonstration and evaluation project involving 11 diverse practices in Colorado explored ways to integrate behavioral health in primary care. Five main themes emerged34,35:
- Frame integrated care as a necessary paradigm shift to patient-centered, whole-person health care.
- Define relationships and protocols up front, understanding that they will evolve.
- Build inclusive, empowered teams to provide the foundation for integration.
- Develop a change management strategy of continuous evaluation and course correction.
- Use targeted data collection pertinent to integrated care to drive improvement and impart accountability.
The Institute for Clinical and Economic Review has organized an extensive list of resources36 for implementing BHI models, a sampling of which is shown in TABLE 2.

Continue to: TAKE-AWAY POINTS
TAKE-AWAY POINTS
There is high quality evidence that collaborative care works for the management of depression and anxiety disorder in primary care, and this is associated with significant cost savings.
CORRESPONDENCE
Rajesh (FNU) Rajesh, MD, Main Campus Family Medicine Clinic, MetroHealth, 2500 MetroHealth Drive, Cleveland, OH 44109; [email protected]
1. Rost K, Nutting P, Smith J, et al. The role of competing demands in the treatment provided primary care patients with major depression. Arch Fam Med. 2000;9:150-154.
2. Rush A, Trivedi M, Carmody T, et al. One-year clinical outcomes of depressed public sector outpatients: a benchmark for subsequent studies. Biol Psychiatry. 2004;56:46-53.
3. Unützer J, Katon W, Callahan CM, et al. Collaborative care management of late-life depression in the primary care setting. JAMA. 2002;288:2836-2845.
4. Department of Veterans Affairs. Bradford DW, Slubicki MN, McDuffie J, et al. Effects of care models to improve general medical outcomes for individuals with serious mental illness. 2011. https://www.hsrd.research.va.gov/publications/esp/smi-REPORT.pdf. Accessed August 22, 2018.
5. Druss BG, von Esenwein S. Improving general medical care for persons with mental and addictive disorders: systematic review. Gen Hosp Psychiatry. 2006;28:145-153.
6. Druss BG, Walker ER. Mental Disorders and Medical Comorbidity. Research Synthesis Report No. 21. Princeton, NJ: The Robert Wood Johnson Foundation; February 2011.
7. Reed SJ, Shore KK, Tice JA. Effectiveness and value of integrating behavioral health into primary care. JAMA Intern Med. 2016;176:691-692.
8. Young AS, Klap R, Sherbourne CD, et al. The quality of care for depressive and anxiety disorders in the United States. Arch Gen Psychiatry. 2001;58:55-61.
9. Butler M, Kane RL, McAlpine D, et al. Integration of mental health/substance abuse and primary care. Rockville, MD: Agency for Healthcare Research and Quality; 2008. http://www.ncbi.nlm.nih.gov/books/NBK38632/. Accessed March 2, 2019.
10. Unützer J, Schoenbaum M, Druss B, et al. Transforming mental health care at the interface with general medicine: report for the presidents commission. Psychiatr Serv. 2006;57:37-47. doi: 10.1176/appi.ps.57.1.37.
11. Peek CJ; the National Integration Academy Council. Lexicon for behavioral health and primary care integration: concepts and definitions developed by expert consensus. AHRQ. https://integrationacademy.ahrq.gov/sites/default/files/Lexicon.pdf. Published April 2013. Accessed May 29, 2019.
12. Heath B, Wise Romero P, Reynolds K. A standard framework for levels of integrated healthcare and update throughout the document. SAMHSA-HRSA. https://www.integration.samhsa.gov/integrated-care-models/A_Standard_Framework_for_Levels_of_Integrated_Healthcare.pdf. Published March 2013. Accessed May 29, 2019.
13. Integrating physical and behavioral health care: promising Medicaid models. The Henry J. Kaiser Family Foundation. https://www.kff.org/wp-content/uploads/2014/02/8553-integrating-physical-and-behavioral-health-care-promising-medicaid-models.pdf. Published February 2014. Accessed May 29, 2019.
14. Vanderlip ER, Rundell J, Avery M, et al. Dissemination of integrated care within adult primary care settings: the collaborative care model. SAMHSA-HRSA. https://www.integration.samhsa.gov/integrated-care-models/APA-APM-Dissemination-Integrated-Care-Report.pdf. Published 2016. Accessed May 29, 2019.
15. Gerrity M. Evolving models of behavioral health integration: evidence update 2010-2015. Milbank Memorial Fund. https://www.milbank.org/wp-content/uploads/2016/05/Evolving-Models-of-BHI.pdf. Published May 2016. Accessed May 29, 2019.
16. Archer J, Bower P, Gilbody S, et al. Collaborative care for depression and anxiety problems. Cochrane Database Syst Rev. 2012;10.1002/14651858.cd006525.pub2.
17. Team Structure. University of Washington AIMs Center. https://aims.uw.edu/collaborative-care/team-structure. Published 2017.Accessed May 29, 2019.
18. Reilly S, Planner C, Gask L, et al. Collaborative care approaches for people with severe mental illness. Cochrane Database Syst Rev. 2013;(11):CD009531.
19. Kilbourne AM, Barbaresso MM, Lai Z, et al. Improving physical health in patients with chronic mental disorders. J Clin Psychiatry. 2017;78:129-137.
20. Oslin DW, Lynch KG, Maisto HSA, et al. A randomized clinical trial of alcohol care management delivered in Department of Veterans Affairs primary care clinics versus specialty addiction treatment. J Gen Intern Med. 2013;29:162-168.
21. Gilbody S, Bower P, Fletcher J, et al. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes. Arch Intern Med. 2006;166:2314-2321.
22. Rossom RC, Solberg LI, Magnan S, et al. Impact of a national collaborative care initiative for patients with depression and diabetes or cardiovascular disease. Gen Hosp Psychiatry. 2017;15:77-85.
23. Kroenke K, Bair MJ, Damush TM, et al. Optimized antidepressant therapy and pain self-management in primary care patients with depression and musculoskeletal pain. JAMA. 2009;301:2009-2110.
24. Katon WJ, Lin EH, Von Korff M, et al. Collaborative care for patients with depression and chronic illnesses. N Engl J Med. 2010;363:2611-2620.
25. Miller CJ, Grogan-Kaylor A, Perron BE, et al. Collaborative chronic care models for mental health conditions. Med Care. 2013;51:922-930.
26. Woltmann E, Grogan-Kaylor A, Perron B, et al. Comparative effectiveness of collaborative chronic care models for mental health conditions across primary, specialty, and behavioral health care settings: systematic review and meta-analysis. Am J Psychiatry. 2012;11:790-804.
27. U.S. Department of Veterans Affairs. Rubenstein LV, Williams JW Jr, Danz M, et al. Determining key features of effective depression interventions. 2009. http://www.hsrd.research.va.gov/publications/esp/depinter.cfm. Accessed August 22, 2018.
28. Coventry PA, Hudson JL, Kontopantelis E, et al. Characteristics of effective collaborative care for treatment of depression: a systematic review and meta-regression of 74 randomised controlled trials. PLoS One. 2014;9:e108114.
29. Institute for Clinical and Economic Review. Tice JA, Ollendorf DA, Reed SJ, et al. Integrating behavioral health into primary care. 2015. https://icer-review.org/wp-content/uploads/2016/01/BHI_Final_Report_0602151.pdf. Accessed August 27, 2018.
30. Rubenstein LV, Chaney EF, Ober S, et al. Using evidence-based quality improvement methods for translating depression collaborative care research into practice. Fam Syst Health. 2010;28:91-113.
31. Unützer J, Katon WJ, Fan MY, et al. Long-term cost effects of collaborative care for late-life depression. Am J Manag Care. 2008;14:95-100.
32. Harkness EF, Bower PJ. On-site mental health workers delivering psychological therapy and psychosocial interventions to patients in primary care: effects on the professional practice of primary care providers. Cochrane Database Syst Rev. 2009;(1):CD000532.
33. Druss BG, Rohrbaugh RM, Levinson CM, et al. Integrated medical care for patients with serious psychiatric illness. Arch Gen Psychiatry. 2001;58:861-868.
34. Davis M, Balasubramanian BA, Waller E, et al. Integrating behavioral and physical health care in the real world: early lessons from advancing care together. J Am Board Fam Med. 2013;26:588-602.
35. Gold SB, Green LA, Peek CJ. From our practices to yours: key messages for the journey to integrated behavioral health. J Am Board Fam Med. 2017;30:25-34.
36. Institute for Clinical and Economic Review. Integrating behavioral health into primary care. 2015. https://icer-review.org/wp-content/uploads/2016/02/CTAF_BHI_Action_Guide_060215.pdf. Accessed April 25, 2019.
1. Rost K, Nutting P, Smith J, et al. The role of competing demands in the treatment provided primary care patients with major depression. Arch Fam Med. 2000;9:150-154.
2. Rush A, Trivedi M, Carmody T, et al. One-year clinical outcomes of depressed public sector outpatients: a benchmark for subsequent studies. Biol Psychiatry. 2004;56:46-53.
3. Unützer J, Katon W, Callahan CM, et al. Collaborative care management of late-life depression in the primary care setting. JAMA. 2002;288:2836-2845.
4. Department of Veterans Affairs. Bradford DW, Slubicki MN, McDuffie J, et al. Effects of care models to improve general medical outcomes for individuals with serious mental illness. 2011. https://www.hsrd.research.va.gov/publications/esp/smi-REPORT.pdf. Accessed August 22, 2018.
5. Druss BG, von Esenwein S. Improving general medical care for persons with mental and addictive disorders: systematic review. Gen Hosp Psychiatry. 2006;28:145-153.
6. Druss BG, Walker ER. Mental Disorders and Medical Comorbidity. Research Synthesis Report No. 21. Princeton, NJ: The Robert Wood Johnson Foundation; February 2011.
7. Reed SJ, Shore KK, Tice JA. Effectiveness and value of integrating behavioral health into primary care. JAMA Intern Med. 2016;176:691-692.
8. Young AS, Klap R, Sherbourne CD, et al. The quality of care for depressive and anxiety disorders in the United States. Arch Gen Psychiatry. 2001;58:55-61.
9. Butler M, Kane RL, McAlpine D, et al. Integration of mental health/substance abuse and primary care. Rockville, MD: Agency for Healthcare Research and Quality; 2008. http://www.ncbi.nlm.nih.gov/books/NBK38632/. Accessed March 2, 2019.
10. Unützer J, Schoenbaum M, Druss B, et al. Transforming mental health care at the interface with general medicine: report for the presidents commission. Psychiatr Serv. 2006;57:37-47. doi: 10.1176/appi.ps.57.1.37.
11. Peek CJ; the National Integration Academy Council. Lexicon for behavioral health and primary care integration: concepts and definitions developed by expert consensus. AHRQ. https://integrationacademy.ahrq.gov/sites/default/files/Lexicon.pdf. Published April 2013. Accessed May 29, 2019.
12. Heath B, Wise Romero P, Reynolds K. A standard framework for levels of integrated healthcare and update throughout the document. SAMHSA-HRSA. https://www.integration.samhsa.gov/integrated-care-models/A_Standard_Framework_for_Levels_of_Integrated_Healthcare.pdf. Published March 2013. Accessed May 29, 2019.
13. Integrating physical and behavioral health care: promising Medicaid models. The Henry J. Kaiser Family Foundation. https://www.kff.org/wp-content/uploads/2014/02/8553-integrating-physical-and-behavioral-health-care-promising-medicaid-models.pdf. Published February 2014. Accessed May 29, 2019.
14. Vanderlip ER, Rundell J, Avery M, et al. Dissemination of integrated care within adult primary care settings: the collaborative care model. SAMHSA-HRSA. https://www.integration.samhsa.gov/integrated-care-models/APA-APM-Dissemination-Integrated-Care-Report.pdf. Published 2016. Accessed May 29, 2019.
15. Gerrity M. Evolving models of behavioral health integration: evidence update 2010-2015. Milbank Memorial Fund. https://www.milbank.org/wp-content/uploads/2016/05/Evolving-Models-of-BHI.pdf. Published May 2016. Accessed May 29, 2019.
16. Archer J, Bower P, Gilbody S, et al. Collaborative care for depression and anxiety problems. Cochrane Database Syst Rev. 2012;10.1002/14651858.cd006525.pub2.
17. Team Structure. University of Washington AIMs Center. https://aims.uw.edu/collaborative-care/team-structure. Published 2017.Accessed May 29, 2019.
18. Reilly S, Planner C, Gask L, et al. Collaborative care approaches for people with severe mental illness. Cochrane Database Syst Rev. 2013;(11):CD009531.
19. Kilbourne AM, Barbaresso MM, Lai Z, et al. Improving physical health in patients with chronic mental disorders. J Clin Psychiatry. 2017;78:129-137.
20. Oslin DW, Lynch KG, Maisto HSA, et al. A randomized clinical trial of alcohol care management delivered in Department of Veterans Affairs primary care clinics versus specialty addiction treatment. J Gen Intern Med. 2013;29:162-168.
21. Gilbody S, Bower P, Fletcher J, et al. Collaborative care for depression: a cumulative meta-analysis and review of longer-term outcomes. Arch Intern Med. 2006;166:2314-2321.
22. Rossom RC, Solberg LI, Magnan S, et al. Impact of a national collaborative care initiative for patients with depression and diabetes or cardiovascular disease. Gen Hosp Psychiatry. 2017;15:77-85.
23. Kroenke K, Bair MJ, Damush TM, et al. Optimized antidepressant therapy and pain self-management in primary care patients with depression and musculoskeletal pain. JAMA. 2009;301:2009-2110.
24. Katon WJ, Lin EH, Von Korff M, et al. Collaborative care for patients with depression and chronic illnesses. N Engl J Med. 2010;363:2611-2620.
25. Miller CJ, Grogan-Kaylor A, Perron BE, et al. Collaborative chronic care models for mental health conditions. Med Care. 2013;51:922-930.
26. Woltmann E, Grogan-Kaylor A, Perron B, et al. Comparative effectiveness of collaborative chronic care models for mental health conditions across primary, specialty, and behavioral health care settings: systematic review and meta-analysis. Am J Psychiatry. 2012;11:790-804.
27. U.S. Department of Veterans Affairs. Rubenstein LV, Williams JW Jr, Danz M, et al. Determining key features of effective depression interventions. 2009. http://www.hsrd.research.va.gov/publications/esp/depinter.cfm. Accessed August 22, 2018.
28. Coventry PA, Hudson JL, Kontopantelis E, et al. Characteristics of effective collaborative care for treatment of depression: a systematic review and meta-regression of 74 randomised controlled trials. PLoS One. 2014;9:e108114.
29. Institute for Clinical and Economic Review. Tice JA, Ollendorf DA, Reed SJ, et al. Integrating behavioral health into primary care. 2015. https://icer-review.org/wp-content/uploads/2016/01/BHI_Final_Report_0602151.pdf. Accessed August 27, 2018.
30. Rubenstein LV, Chaney EF, Ober S, et al. Using evidence-based quality improvement methods for translating depression collaborative care research into practice. Fam Syst Health. 2010;28:91-113.
31. Unützer J, Katon WJ, Fan MY, et al. Long-term cost effects of collaborative care for late-life depression. Am J Manag Care. 2008;14:95-100.
32. Harkness EF, Bower PJ. On-site mental health workers delivering psychological therapy and psychosocial interventions to patients in primary care: effects on the professional practice of primary care providers. Cochrane Database Syst Rev. 2009;(1):CD000532.
33. Druss BG, Rohrbaugh RM, Levinson CM, et al. Integrated medical care for patients with serious psychiatric illness. Arch Gen Psychiatry. 2001;58:861-868.
34. Davis M, Balasubramanian BA, Waller E, et al. Integrating behavioral and physical health care in the real world: early lessons from advancing care together. J Am Board Fam Med. 2013;26:588-602.
35. Gold SB, Green LA, Peek CJ. From our practices to yours: key messages for the journey to integrated behavioral health. J Am Board Fam Med. 2017;30:25-34.
36. Institute for Clinical and Economic Review. Integrating behavioral health into primary care. 2015. https://icer-review.org/wp-content/uploads/2016/02/CTAF_BHI_Action_Guide_060215.pdf. Accessed April 25, 2019.