User login
Celecoxib oral solution treats migraine effectively in randomized trial
, according to trial results published in the January issue of Headache.
Two hours after treatment, a significantly greater proportion of patients who received the liquid solution, known as DFN-15, had freedom from pain and freedom from their most bothersome accompanying symptom – nausea, photophobia, or phonophobia – compared with patients who received placebo. The pain freedom rates were 35.6% with celecoxib oral solution and 21.7% with placebo. The rates of freedom from the most bothersome symptom were 57.8% with celecoxib oral solution and 44.8% with placebo.
About 9% of patients who received celecoxib oral solution had treatment-emergent adverse events related to the study drug, the most common of which were dysgeusia (4.2%) and nausea (3.2%). In comparison, about 6% of patients who received placebo had treatment-emergent adverse events. There were no serious treatment-emergent adverse events.
“DFN‐15 has the potential to become a reliable and convenient acute therapeutic option for patients with migraine,” said lead author Richard B. Lipton, MD, and colleagues. Dr. Lipton is affiliated with the Albert Einstein College of Medicine in New York.
Assessing celecoxib in migraineurs
Evidence-based guidelines recommend nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, diclofenac, ibuprofen, and naproxen, as effective acute migraine treatments, but these medications may increase the risk of adverse gastrointestinal events, the authors said. Celecoxib, a selective cyclooxygenase (COX)-2 inhibitor, is indicated for the treatment of acute pain in patients with ankylosing spondylitis, osteoarthritis, primary dysmenorrhea, and rheumatoid arthritis. Although it produces analgesia similar to other NSAIDs, among patients with osteoarthritis and rheumatoid arthritis, celecoxib is associated with significantly lower risk of gastrointestinal events, compared with naproxen and ibuprofen, and significantly lower risk of renal events, compared with ibuprofen.
Researchers have studied an oral capsule form of celecoxib (Celebrex, Pfizer) as an acute treatment for migraine in an open-label study that compared celecoxib with naproxen sodium. “While preliminary results suggest comparable efficacy but better tolerability than widely used and guideline-recommended NSAIDs, celecoxib is not currently approved for migraine,” the authors said.
Compared with the oral capsule formulation, the oral liquid solution DFN-15 has a faster median time to peak concentration under fasting conditions (within 1 hour vs. 2.5 hours), which “could translate into more rapid onset of pain relief,” the authors said. In addition, DFN-15 may have greater bioavailability, which could lower dose requirements and improve safety and tolerability. To compare the efficacy, tolerability, and safety of 120-mg DFN-15 with placebo for the acute treatment of migraine, researchers conducted a randomized, double-blind, placebo-controlled study.
Participants used single-dose bottles
Researchers randomized 622 patients 1:1 to DFN-15 or placebo, and 567 treated a migraine during the trial. Patients had a mean age of 40 years, and 87% were female. Patients had episodic migraine with or without aura, no signs of medication overuse, and two-eight migraine attacks per month. For the trial, patients treated a single migraine attack of moderate to severe intensity within 1 hour of onset. “Each subject was given a single‐dose bottle of DFN‐15 120 mg or matching placebo containing 4.8 mL liquid,” Dr. Lipton and colleagues said. “They were instructed to drink the entire contents of the bottle to ensure complete consumption of study medication.”
Freedom from pain and freedom from the most bothersome symptom at 2 hours were the coprimary endpoints. “DFN‐15 was also significantly superior to placebo on multiple secondary 2‐hour endpoints, including freedom from photophobia, pain relief, change in functional disability from baseline, overall and 24‐hour satisfaction with treatment, and use of rescue medication,” they reported.
“A new COX‐2 inhibitor that is effective and rapidly absorbed could provide an important new option for a wide range of patients,” the authors said. “Though cross‐study comparisons are problematic, the current results for DFN‐15 indicate that its efficacy is similar to that of NSAIDs and small‐molecule calcitonin gene‐related peptide receptor antagonists (gepants), based on placebo‐subtracted rates pain freedom in acute treatment trials (14%‐21%). DFN‐15 may also be useful among triptan users, who are at elevated risk of medication‐overuse headache and for whom TEAEs within 24 hours postdose are common. ... The form and delivery system of DFN‐15 – a ready‐to‐use solution in a 4.8‐mL single‐use bottle – may support patient adherence.”
The trial had robust placebo response rates, which may have been influenced by “the novelty of a ready‐made oral solution, which has not been previously tested for the acute treatment of migraine,” the authors noted. In addition, the trial does not address the treatment of mild pain or treatment across multiple attacks.
The trial was supported by Dr. Reddy’s Laboratories, manufacturer of DFN-15. Two authors are employed by and own stock in Dr. Reddy’s. Dr. Lipton and a coauthor disclosed research support from and consulting for Dr. Reddy’s.
SOURCE: Lipton RB et al. Headache. 2020;60(1):58-70. doi: 10.1111/head.13663.
, according to trial results published in the January issue of Headache.
Two hours after treatment, a significantly greater proportion of patients who received the liquid solution, known as DFN-15, had freedom from pain and freedom from their most bothersome accompanying symptom – nausea, photophobia, or phonophobia – compared with patients who received placebo. The pain freedom rates were 35.6% with celecoxib oral solution and 21.7% with placebo. The rates of freedom from the most bothersome symptom were 57.8% with celecoxib oral solution and 44.8% with placebo.
About 9% of patients who received celecoxib oral solution had treatment-emergent adverse events related to the study drug, the most common of which were dysgeusia (4.2%) and nausea (3.2%). In comparison, about 6% of patients who received placebo had treatment-emergent adverse events. There were no serious treatment-emergent adverse events.
“DFN‐15 has the potential to become a reliable and convenient acute therapeutic option for patients with migraine,” said lead author Richard B. Lipton, MD, and colleagues. Dr. Lipton is affiliated with the Albert Einstein College of Medicine in New York.
Assessing celecoxib in migraineurs
Evidence-based guidelines recommend nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, diclofenac, ibuprofen, and naproxen, as effective acute migraine treatments, but these medications may increase the risk of adverse gastrointestinal events, the authors said. Celecoxib, a selective cyclooxygenase (COX)-2 inhibitor, is indicated for the treatment of acute pain in patients with ankylosing spondylitis, osteoarthritis, primary dysmenorrhea, and rheumatoid arthritis. Although it produces analgesia similar to other NSAIDs, among patients with osteoarthritis and rheumatoid arthritis, celecoxib is associated with significantly lower risk of gastrointestinal events, compared with naproxen and ibuprofen, and significantly lower risk of renal events, compared with ibuprofen.
Researchers have studied an oral capsule form of celecoxib (Celebrex, Pfizer) as an acute treatment for migraine in an open-label study that compared celecoxib with naproxen sodium. “While preliminary results suggest comparable efficacy but better tolerability than widely used and guideline-recommended NSAIDs, celecoxib is not currently approved for migraine,” the authors said.
Compared with the oral capsule formulation, the oral liquid solution DFN-15 has a faster median time to peak concentration under fasting conditions (within 1 hour vs. 2.5 hours), which “could translate into more rapid onset of pain relief,” the authors said. In addition, DFN-15 may have greater bioavailability, which could lower dose requirements and improve safety and tolerability. To compare the efficacy, tolerability, and safety of 120-mg DFN-15 with placebo for the acute treatment of migraine, researchers conducted a randomized, double-blind, placebo-controlled study.
Participants used single-dose bottles
Researchers randomized 622 patients 1:1 to DFN-15 or placebo, and 567 treated a migraine during the trial. Patients had a mean age of 40 years, and 87% were female. Patients had episodic migraine with or without aura, no signs of medication overuse, and two-eight migraine attacks per month. For the trial, patients treated a single migraine attack of moderate to severe intensity within 1 hour of onset. “Each subject was given a single‐dose bottle of DFN‐15 120 mg or matching placebo containing 4.8 mL liquid,” Dr. Lipton and colleagues said. “They were instructed to drink the entire contents of the bottle to ensure complete consumption of study medication.”
Freedom from pain and freedom from the most bothersome symptom at 2 hours were the coprimary endpoints. “DFN‐15 was also significantly superior to placebo on multiple secondary 2‐hour endpoints, including freedom from photophobia, pain relief, change in functional disability from baseline, overall and 24‐hour satisfaction with treatment, and use of rescue medication,” they reported.
“A new COX‐2 inhibitor that is effective and rapidly absorbed could provide an important new option for a wide range of patients,” the authors said. “Though cross‐study comparisons are problematic, the current results for DFN‐15 indicate that its efficacy is similar to that of NSAIDs and small‐molecule calcitonin gene‐related peptide receptor antagonists (gepants), based on placebo‐subtracted rates pain freedom in acute treatment trials (14%‐21%). DFN‐15 may also be useful among triptan users, who are at elevated risk of medication‐overuse headache and for whom TEAEs within 24 hours postdose are common. ... The form and delivery system of DFN‐15 – a ready‐to‐use solution in a 4.8‐mL single‐use bottle – may support patient adherence.”
The trial had robust placebo response rates, which may have been influenced by “the novelty of a ready‐made oral solution, which has not been previously tested for the acute treatment of migraine,” the authors noted. In addition, the trial does not address the treatment of mild pain or treatment across multiple attacks.
The trial was supported by Dr. Reddy’s Laboratories, manufacturer of DFN-15. Two authors are employed by and own stock in Dr. Reddy’s. Dr. Lipton and a coauthor disclosed research support from and consulting for Dr. Reddy’s.
SOURCE: Lipton RB et al. Headache. 2020;60(1):58-70. doi: 10.1111/head.13663.
, according to trial results published in the January issue of Headache.
Two hours after treatment, a significantly greater proportion of patients who received the liquid solution, known as DFN-15, had freedom from pain and freedom from their most bothersome accompanying symptom – nausea, photophobia, or phonophobia – compared with patients who received placebo. The pain freedom rates were 35.6% with celecoxib oral solution and 21.7% with placebo. The rates of freedom from the most bothersome symptom were 57.8% with celecoxib oral solution and 44.8% with placebo.
About 9% of patients who received celecoxib oral solution had treatment-emergent adverse events related to the study drug, the most common of which were dysgeusia (4.2%) and nausea (3.2%). In comparison, about 6% of patients who received placebo had treatment-emergent adverse events. There were no serious treatment-emergent adverse events.
“DFN‐15 has the potential to become a reliable and convenient acute therapeutic option for patients with migraine,” said lead author Richard B. Lipton, MD, and colleagues. Dr. Lipton is affiliated with the Albert Einstein College of Medicine in New York.
Assessing celecoxib in migraineurs
Evidence-based guidelines recommend nonsteroidal anti-inflammatory drugs (NSAIDs), including aspirin, diclofenac, ibuprofen, and naproxen, as effective acute migraine treatments, but these medications may increase the risk of adverse gastrointestinal events, the authors said. Celecoxib, a selective cyclooxygenase (COX)-2 inhibitor, is indicated for the treatment of acute pain in patients with ankylosing spondylitis, osteoarthritis, primary dysmenorrhea, and rheumatoid arthritis. Although it produces analgesia similar to other NSAIDs, among patients with osteoarthritis and rheumatoid arthritis, celecoxib is associated with significantly lower risk of gastrointestinal events, compared with naproxen and ibuprofen, and significantly lower risk of renal events, compared with ibuprofen.
Researchers have studied an oral capsule form of celecoxib (Celebrex, Pfizer) as an acute treatment for migraine in an open-label study that compared celecoxib with naproxen sodium. “While preliminary results suggest comparable efficacy but better tolerability than widely used and guideline-recommended NSAIDs, celecoxib is not currently approved for migraine,” the authors said.
Compared with the oral capsule formulation, the oral liquid solution DFN-15 has a faster median time to peak concentration under fasting conditions (within 1 hour vs. 2.5 hours), which “could translate into more rapid onset of pain relief,” the authors said. In addition, DFN-15 may have greater bioavailability, which could lower dose requirements and improve safety and tolerability. To compare the efficacy, tolerability, and safety of 120-mg DFN-15 with placebo for the acute treatment of migraine, researchers conducted a randomized, double-blind, placebo-controlled study.
Participants used single-dose bottles
Researchers randomized 622 patients 1:1 to DFN-15 or placebo, and 567 treated a migraine during the trial. Patients had a mean age of 40 years, and 87% were female. Patients had episodic migraine with or without aura, no signs of medication overuse, and two-eight migraine attacks per month. For the trial, patients treated a single migraine attack of moderate to severe intensity within 1 hour of onset. “Each subject was given a single‐dose bottle of DFN‐15 120 mg or matching placebo containing 4.8 mL liquid,” Dr. Lipton and colleagues said. “They were instructed to drink the entire contents of the bottle to ensure complete consumption of study medication.”
Freedom from pain and freedom from the most bothersome symptom at 2 hours were the coprimary endpoints. “DFN‐15 was also significantly superior to placebo on multiple secondary 2‐hour endpoints, including freedom from photophobia, pain relief, change in functional disability from baseline, overall and 24‐hour satisfaction with treatment, and use of rescue medication,” they reported.
“A new COX‐2 inhibitor that is effective and rapidly absorbed could provide an important new option for a wide range of patients,” the authors said. “Though cross‐study comparisons are problematic, the current results for DFN‐15 indicate that its efficacy is similar to that of NSAIDs and small‐molecule calcitonin gene‐related peptide receptor antagonists (gepants), based on placebo‐subtracted rates pain freedom in acute treatment trials (14%‐21%). DFN‐15 may also be useful among triptan users, who are at elevated risk of medication‐overuse headache and for whom TEAEs within 24 hours postdose are common. ... The form and delivery system of DFN‐15 – a ready‐to‐use solution in a 4.8‐mL single‐use bottle – may support patient adherence.”
The trial had robust placebo response rates, which may have been influenced by “the novelty of a ready‐made oral solution, which has not been previously tested for the acute treatment of migraine,” the authors noted. In addition, the trial does not address the treatment of mild pain or treatment across multiple attacks.
The trial was supported by Dr. Reddy’s Laboratories, manufacturer of DFN-15. Two authors are employed by and own stock in Dr. Reddy’s. Dr. Lipton and a coauthor disclosed research support from and consulting for Dr. Reddy’s.
SOURCE: Lipton RB et al. Headache. 2020;60(1):58-70. doi: 10.1111/head.13663.
FROM HEADACHE
Esophageal length ratio predicts hiatal hernia recurrence
A new ratio based on manometric esophageal length in relation to patient height could offer an objective means of preoperatively identifying shortened esophagus, which could improve surgical planning and outcomes with hiatal hernia repair, according to investigators.
In a retrospective analysis, patients with a lower manometric esophageal length-to-height (MELH) ratio had a higher rate of hiatal hernia recurrence, reported lead author Pooja Lal, MD, of the Cleveland Clinic, and colleagues.
A short esophagus increases tension at the gastroesophageal junction, which may necessitate a lengthening procedure in addition to hiatal hernia repair, the investigators wrote in the Journal of Clinical Gastroenterology. As lengthening may require additional expertise, preoperative knowledge of a short esophagus is beneficial; however, until this point, short esophagus could only be identified intraoperatively. Since previous attempts to define short esophagus were confounded by patient height, the investigators devised the MELH ratio to account for this variable.
The investigators evaluated data from 245 patients who underwent hiatal hernia repair by Nissen fundoplication, of whom 157 also underwent esophageal lengthening with a Collis gastroplasty. The decision to perform a Collis gastroplasty was made intraoperatively if a patient did not have at least 2-3 cm of intra-abdominal esophageal length with minimal tension.
For all patients, the MELH ratio was determined by dividing manometric esophageal length by patient height (both in centimeters).
On average, patients who needed a Collis gastroplasty had a shorter esophagus (20.2 vs. 22.4 cm; P less than .001) and a lower MELH ratio (0.12 vs. 0.13; P less than .001).
Multivariable hazard regression showed that regardless of surgical approach, for every 0.01 U-increment increase in MELH ratio, risk of hernia recurrence decreased by 33% (hazard ratio, 0.67; P less than .001). In contrast, regardless of MELH ratio, repair without Collis was associated with a 500% increased risk of recurrence (HR, 6.1; P less than .001). Over 5 years, the benefit of Collis gastroplasty translated to a significantly lower rate of both hernia recurrence (18% vs. 55%; P less than .001) and reoperations for recurrence (0% vs. 10%; P less than .001).
“We suggest that surgeons and gastroenterologists calculate the MELH ratio before repair of a hiatal hernia, and be cognizant of patients with a shortened esophagus,” the investigators concluded. “An esophageal lengthening procedure such as a Collis gastroplasty may reduce the risk of hernia recurrence and reoperation for recurrence, especially for patients with a MELH ratio less than 0.12.”The investigators reported no conflicts of interest.
SOURCE: Lal P et al. J Clin Gastroenterol. 2020 Jan 20. doi: 10.1097/MCG.0000000000001316.
A new ratio based on manometric esophageal length in relation to patient height could offer an objective means of preoperatively identifying shortened esophagus, which could improve surgical planning and outcomes with hiatal hernia repair, according to investigators.
In a retrospective analysis, patients with a lower manometric esophageal length-to-height (MELH) ratio had a higher rate of hiatal hernia recurrence, reported lead author Pooja Lal, MD, of the Cleveland Clinic, and colleagues.
A short esophagus increases tension at the gastroesophageal junction, which may necessitate a lengthening procedure in addition to hiatal hernia repair, the investigators wrote in the Journal of Clinical Gastroenterology. As lengthening may require additional expertise, preoperative knowledge of a short esophagus is beneficial; however, until this point, short esophagus could only be identified intraoperatively. Since previous attempts to define short esophagus were confounded by patient height, the investigators devised the MELH ratio to account for this variable.
The investigators evaluated data from 245 patients who underwent hiatal hernia repair by Nissen fundoplication, of whom 157 also underwent esophageal lengthening with a Collis gastroplasty. The decision to perform a Collis gastroplasty was made intraoperatively if a patient did not have at least 2-3 cm of intra-abdominal esophageal length with minimal tension.
For all patients, the MELH ratio was determined by dividing manometric esophageal length by patient height (both in centimeters).
On average, patients who needed a Collis gastroplasty had a shorter esophagus (20.2 vs. 22.4 cm; P less than .001) and a lower MELH ratio (0.12 vs. 0.13; P less than .001).
Multivariable hazard regression showed that regardless of surgical approach, for every 0.01 U-increment increase in MELH ratio, risk of hernia recurrence decreased by 33% (hazard ratio, 0.67; P less than .001). In contrast, regardless of MELH ratio, repair without Collis was associated with a 500% increased risk of recurrence (HR, 6.1; P less than .001). Over 5 years, the benefit of Collis gastroplasty translated to a significantly lower rate of both hernia recurrence (18% vs. 55%; P less than .001) and reoperations for recurrence (0% vs. 10%; P less than .001).
“We suggest that surgeons and gastroenterologists calculate the MELH ratio before repair of a hiatal hernia, and be cognizant of patients with a shortened esophagus,” the investigators concluded. “An esophageal lengthening procedure such as a Collis gastroplasty may reduce the risk of hernia recurrence and reoperation for recurrence, especially for patients with a MELH ratio less than 0.12.”The investigators reported no conflicts of interest.
SOURCE: Lal P et al. J Clin Gastroenterol. 2020 Jan 20. doi: 10.1097/MCG.0000000000001316.
A new ratio based on manometric esophageal length in relation to patient height could offer an objective means of preoperatively identifying shortened esophagus, which could improve surgical planning and outcomes with hiatal hernia repair, according to investigators.
In a retrospective analysis, patients with a lower manometric esophageal length-to-height (MELH) ratio had a higher rate of hiatal hernia recurrence, reported lead author Pooja Lal, MD, of the Cleveland Clinic, and colleagues.
A short esophagus increases tension at the gastroesophageal junction, which may necessitate a lengthening procedure in addition to hiatal hernia repair, the investigators wrote in the Journal of Clinical Gastroenterology. As lengthening may require additional expertise, preoperative knowledge of a short esophagus is beneficial; however, until this point, short esophagus could only be identified intraoperatively. Since previous attempts to define short esophagus were confounded by patient height, the investigators devised the MELH ratio to account for this variable.
The investigators evaluated data from 245 patients who underwent hiatal hernia repair by Nissen fundoplication, of whom 157 also underwent esophageal lengthening with a Collis gastroplasty. The decision to perform a Collis gastroplasty was made intraoperatively if a patient did not have at least 2-3 cm of intra-abdominal esophageal length with minimal tension.
For all patients, the MELH ratio was determined by dividing manometric esophageal length by patient height (both in centimeters).
On average, patients who needed a Collis gastroplasty had a shorter esophagus (20.2 vs. 22.4 cm; P less than .001) and a lower MELH ratio (0.12 vs. 0.13; P less than .001).
Multivariable hazard regression showed that regardless of surgical approach, for every 0.01 U-increment increase in MELH ratio, risk of hernia recurrence decreased by 33% (hazard ratio, 0.67; P less than .001). In contrast, regardless of MELH ratio, repair without Collis was associated with a 500% increased risk of recurrence (HR, 6.1; P less than .001). Over 5 years, the benefit of Collis gastroplasty translated to a significantly lower rate of both hernia recurrence (18% vs. 55%; P less than .001) and reoperations for recurrence (0% vs. 10%; P less than .001).
“We suggest that surgeons and gastroenterologists calculate the MELH ratio before repair of a hiatal hernia, and be cognizant of patients with a shortened esophagus,” the investigators concluded. “An esophageal lengthening procedure such as a Collis gastroplasty may reduce the risk of hernia recurrence and reoperation for recurrence, especially for patients with a MELH ratio less than 0.12.”The investigators reported no conflicts of interest.
SOURCE: Lal P et al. J Clin Gastroenterol. 2020 Jan 20. doi: 10.1097/MCG.0000000000001316.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Genetic factor linked to impaired memory after heading many soccer balls
according to authors of a recent longitudinal study. Worse verbal memory was linked to high levels of ball heading among those players who were APOE e4–positive, compared with those who were APOE e4–negative, according to the authors, led by Liane E. Hunter, PhD, of the Gruss Magnetic Resonance Imaging Center at Albert Einstein College of Medicine, New York.
These findings, while preliminary, do raise the possibility that “safe levels for soccer heading” could be proposed to protect players from harm or that APOE e4-positive players might be advised to limit their exposure to head impacts, Dr. Hunter and coauthors wrote in a report in JAMA Neurology.
However, the findings should “in no way” be used to justify APOE testing to make clinical decisions regarding the safety of playing soccer, said Sarah J. Banks, PhD, of the University of California, San Diego, and Jesse Mez, MD, of Boston University in a related editorial (doi: 10.1001/jamaneurol.2019.4451). “Like most good science, the study provides an important, but incremental, step to understanding gene-environment interactions in sports,” Dr. Banks and Dr. Mez wrote in their editorial.
While there are some studies tying APOE e4 to poorer neuropsychiatric performance in boxers and U.S. football players, there are no such studies looking at the role of APOE e4 in soccer players exposed to repetitive “subconcussive” ball heading, according to Dr. Hunter and coresearchers. Accordingly, they sought to analyze APOE e4 and neuropsychological performance in relation to ball heading in 352 adult amateur soccer players enrolled in the Einstein Soccer Study between November 2013 and January 2018. About three-quarters of the players were male, and the median age at enrollment was 23 years.
The players completed a computer-based questionnaire designed to estimate their exposure to soccer heading at enrollment and at follow-up visits every 3-6 months. To test verbal memory at each visit, players were asked to memorize a 12-item grocery list, and then asked to recall the items 20 minutes later.
High levels of heading were linked to poorer performance on the verbal memory task, similar to one previously reported study, investigators said.
There was no association overall of APOE e4 and heading with performance on the shopping list task, according to investigators. By contrast, there was a 4.1-fold increased deficit in verbal memory for APOE e4–positive players with high heading exposure, compared with those with low exposure, investigators reported. Likewise, there was an 8.5-fold increased deficit in verbal memory for APOE e4–positive players with high versus moderate heading exposure.
That said, the absolute difference in performance was “subtle” and difficult to interpret in the context of a cross-sectional study, Dr. Banks and Dr. Mez said in their editorial.
In absolute terms, the mean decrease in scores on the 13-point shopping list task between the high and low heading exposure was 1.13 points greater for the APOE e4–positive group, compared with the APOE e4–negative group, and the decrease between the high and moderate heading exposure groups was 0.98 points greater, according to the report.
“The effect size of our interaction is relatively small,” Dr. Hunter and colleagues acknowledged in their report. “However, similar to the widely cited model of disease evolution in Alzheimer disease, our findings may be evidence of early subclinical effects, which could accumulate in APOE e4–positive players over a protracted time frame and ultimately be associated with overt clinical dysfunction.”
Several study authors said they had received grants from the National Institutes of Health and affiliated institutes, the Migraine Research Foundation, and the National Headache Foundation. They reported disclosures related to Amgen, Avanir, Biohaven Holdings, Biovision, Boston Scientific, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, and Pfizer, among others.
SOURCE: Hunter LE et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4828.
according to authors of a recent longitudinal study. Worse verbal memory was linked to high levels of ball heading among those players who were APOE e4–positive, compared with those who were APOE e4–negative, according to the authors, led by Liane E. Hunter, PhD, of the Gruss Magnetic Resonance Imaging Center at Albert Einstein College of Medicine, New York.
These findings, while preliminary, do raise the possibility that “safe levels for soccer heading” could be proposed to protect players from harm or that APOE e4-positive players might be advised to limit their exposure to head impacts, Dr. Hunter and coauthors wrote in a report in JAMA Neurology.
However, the findings should “in no way” be used to justify APOE testing to make clinical decisions regarding the safety of playing soccer, said Sarah J. Banks, PhD, of the University of California, San Diego, and Jesse Mez, MD, of Boston University in a related editorial (doi: 10.1001/jamaneurol.2019.4451). “Like most good science, the study provides an important, but incremental, step to understanding gene-environment interactions in sports,” Dr. Banks and Dr. Mez wrote in their editorial.
While there are some studies tying APOE e4 to poorer neuropsychiatric performance in boxers and U.S. football players, there are no such studies looking at the role of APOE e4 in soccer players exposed to repetitive “subconcussive” ball heading, according to Dr. Hunter and coresearchers. Accordingly, they sought to analyze APOE e4 and neuropsychological performance in relation to ball heading in 352 adult amateur soccer players enrolled in the Einstein Soccer Study between November 2013 and January 2018. About three-quarters of the players were male, and the median age at enrollment was 23 years.
The players completed a computer-based questionnaire designed to estimate their exposure to soccer heading at enrollment and at follow-up visits every 3-6 months. To test verbal memory at each visit, players were asked to memorize a 12-item grocery list, and then asked to recall the items 20 minutes later.
High levels of heading were linked to poorer performance on the verbal memory task, similar to one previously reported study, investigators said.
There was no association overall of APOE e4 and heading with performance on the shopping list task, according to investigators. By contrast, there was a 4.1-fold increased deficit in verbal memory for APOE e4–positive players with high heading exposure, compared with those with low exposure, investigators reported. Likewise, there was an 8.5-fold increased deficit in verbal memory for APOE e4–positive players with high versus moderate heading exposure.
That said, the absolute difference in performance was “subtle” and difficult to interpret in the context of a cross-sectional study, Dr. Banks and Dr. Mez said in their editorial.
In absolute terms, the mean decrease in scores on the 13-point shopping list task between the high and low heading exposure was 1.13 points greater for the APOE e4–positive group, compared with the APOE e4–negative group, and the decrease between the high and moderate heading exposure groups was 0.98 points greater, according to the report.
“The effect size of our interaction is relatively small,” Dr. Hunter and colleagues acknowledged in their report. “However, similar to the widely cited model of disease evolution in Alzheimer disease, our findings may be evidence of early subclinical effects, which could accumulate in APOE e4–positive players over a protracted time frame and ultimately be associated with overt clinical dysfunction.”
Several study authors said they had received grants from the National Institutes of Health and affiliated institutes, the Migraine Research Foundation, and the National Headache Foundation. They reported disclosures related to Amgen, Avanir, Biohaven Holdings, Biovision, Boston Scientific, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, and Pfizer, among others.
SOURCE: Hunter LE et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4828.
according to authors of a recent longitudinal study. Worse verbal memory was linked to high levels of ball heading among those players who were APOE e4–positive, compared with those who were APOE e4–negative, according to the authors, led by Liane E. Hunter, PhD, of the Gruss Magnetic Resonance Imaging Center at Albert Einstein College of Medicine, New York.
These findings, while preliminary, do raise the possibility that “safe levels for soccer heading” could be proposed to protect players from harm or that APOE e4-positive players might be advised to limit their exposure to head impacts, Dr. Hunter and coauthors wrote in a report in JAMA Neurology.
However, the findings should “in no way” be used to justify APOE testing to make clinical decisions regarding the safety of playing soccer, said Sarah J. Banks, PhD, of the University of California, San Diego, and Jesse Mez, MD, of Boston University in a related editorial (doi: 10.1001/jamaneurol.2019.4451). “Like most good science, the study provides an important, but incremental, step to understanding gene-environment interactions in sports,” Dr. Banks and Dr. Mez wrote in their editorial.
While there are some studies tying APOE e4 to poorer neuropsychiatric performance in boxers and U.S. football players, there are no such studies looking at the role of APOE e4 in soccer players exposed to repetitive “subconcussive” ball heading, according to Dr. Hunter and coresearchers. Accordingly, they sought to analyze APOE e4 and neuropsychological performance in relation to ball heading in 352 adult amateur soccer players enrolled in the Einstein Soccer Study between November 2013 and January 2018. About three-quarters of the players were male, and the median age at enrollment was 23 years.
The players completed a computer-based questionnaire designed to estimate their exposure to soccer heading at enrollment and at follow-up visits every 3-6 months. To test verbal memory at each visit, players were asked to memorize a 12-item grocery list, and then asked to recall the items 20 minutes later.
High levels of heading were linked to poorer performance on the verbal memory task, similar to one previously reported study, investigators said.
There was no association overall of APOE e4 and heading with performance on the shopping list task, according to investigators. By contrast, there was a 4.1-fold increased deficit in verbal memory for APOE e4–positive players with high heading exposure, compared with those with low exposure, investigators reported. Likewise, there was an 8.5-fold increased deficit in verbal memory for APOE e4–positive players with high versus moderate heading exposure.
That said, the absolute difference in performance was “subtle” and difficult to interpret in the context of a cross-sectional study, Dr. Banks and Dr. Mez said in their editorial.
In absolute terms, the mean decrease in scores on the 13-point shopping list task between the high and low heading exposure was 1.13 points greater for the APOE e4–positive group, compared with the APOE e4–negative group, and the decrease between the high and moderate heading exposure groups was 0.98 points greater, according to the report.
“The effect size of our interaction is relatively small,” Dr. Hunter and colleagues acknowledged in their report. “However, similar to the widely cited model of disease evolution in Alzheimer disease, our findings may be evidence of early subclinical effects, which could accumulate in APOE e4–positive players over a protracted time frame and ultimately be associated with overt clinical dysfunction.”
Several study authors said they had received grants from the National Institutes of Health and affiliated institutes, the Migraine Research Foundation, and the National Headache Foundation. They reported disclosures related to Amgen, Avanir, Biohaven Holdings, Biovision, Boston Scientific, Eli Lilly, eNeura Therapeutics, GlaxoSmithKline, Merck, and Pfizer, among others.
SOURCE: Hunter LE et al. JAMA Neurol. 2020 Jan 27. doi: 10.1001/jamaneurol.2019.4828.
FROM JAMA Neurology
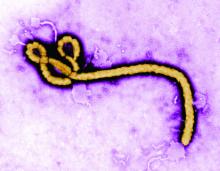
ID Blog: Wuhan coronavirus – just a stop on the zoonotic highway
Emerging viruses that spread to humans from an animal host are commonplace and represent some of the deadliest diseases known. Given the details of the Wuhan coronavirus (2019-nCoV) outbreak, including the genetic profile of the disease agent, the hypothesis of a snake origin was the first raised in the peer-reviewed literature.
It is a highly controversial origin story, however, given that mammals have been the sources of all other such zoonotic coronaviruses, as well as a host of other zoonotic diseases.
An animal source for emerging infections such as the 2019-nCoV is the default hypothesis, because “around 60% of all infectious diseases in humans are zoonotic, as are 75% of all emerging infectious diseases,” according to a United Nations report. The report goes on to say that, “on average, one new infectious disease emerges in humans every 4 months.”
To appreciate the emergence and nature of 2019-nCoV, it is important to examine the history of zoonotic outbreaks of other such diseases, especially with regard to the “mixing-vessel” phenomenon, which has been noted in closely related coronaviruses, including SARS and MERS, as well as the widely disparate HIV, Ebola, and influenza viruses.
Mutants in the mixing vessel
The mixing-vessel phenomenon is conceptually easy but molecularly complex. A single animal is coinfected with two related viruses; the virus genomes recombine together (virus “sex”) in that animal to form a new variant of virus. Such new mutant viruses can be more or less infective, more or less deadly, and more or less able to jump the species or even genus barrier. An emerging viral zoonosis can occur when a human being is exposed to one of these new viruses (either from the origin species or another species intermediate) that is capable of also infecting a human cell. Such exposure can occur from close proximity to animal waste or body fluids, as in the farm environment, or from wildlife pets or the capturing and slaughtering of wildlife for food, as is proposed in the case of the Wuhan seafood market scenario. In fact, the scientists who postulated a snake intermediary as the potential mixing vessel also stated that 2019‐nCoV appears to be a recombinant virus between a bat coronavirus and an origin‐unknown coronavirus.
Coronaviruses in particular have a history of moving from animal to human hosts (and even back again), and their detailed genetic pattern and taxonomy can reveal the animal origin of these diseases.
Going batty
Bats, in particular, have been shown to be a reservoir species for both alphacoronaviruses and betacoronaviruses. Given their ecology and behavior, they have been found to play a key role in transmitting coronaviruses between species. A highly pertinent example of this is the SARS coronavirus, which was shown to have likely originated in Chinese horseshoe bats. The SARS virus, which is genetically closely related to the new Wuhan coronavirus, first infected humans in the Guangdong province of southern China in 2002.
Scientists speculate that the virus was then either transmitted directly to humans from bats, or passed through an intermediate host species, with SARS-like viruses isolated from Himalayan palm civets found in a live-animal market in Guangdong. The virus infection was also detected in other animals (including a raccoon dog, Nyctereutes procyonoides) and in humans working at the market.
The MERS coronavirus is a betacoronavirus that was first reported in Saudi Arabia in 2012. It turned out to be far more deadly than either SARS or the Wuhan virus (at least as far as current estimates of the new coronavirus’s behavior). The MERS genotype was found to be closely related to MERS-like viruses in bats in Saudi Arabia, Africa, Europe, and Asia. Studies done on the cell receptor for MERS showed an apparently conserved viral receptor in both bats and humans. And an identical strain of MERS was found in bats in a nearby cave and near the workplace of the first known human patient.
However, in many of the other locations of the outbreak in the Middle East, there appeared to be limited contact between bats and humans, so scientists looked for another vector species, perhaps one that was acting as an intermediate. A high seroprevalence of MERS-CoV or a closely related virus was found in camels across the Arabian Peninsula and parts of eastern and northern Africa, while tests for MERS antibodies were negative in the most-likely other species of livestock or pet animals, including chickens, cows, goats, horses, and sheep.
In addition, the MERS-related CoV carried by camels was genetically highly similar to that detected in humans, as demonstrated in one particular outbreak on a farm in Qatar where the genetic sequences of MERS-CoV in the nasal swabs from 3 of 14 seropositive camels were similar to those of 2 human cases on the same farm. Similar genomic results were found in MERS-CoV from nasal swabs from camels in Saudi Arabia.
Other mixing-vessel zoonoses
HIV, the viral cause of AIDS, provides an almost-textbook origin story of the rise of a zoonotic supervillain. The virus was genetically traced to have a chimpanzee-to-human origin, but it was found to be more complicated than that. The virus first emerged in the 1920s in Africa in what is now the Democratic Republic of the Congo, well before its rise to a global pandemic in the 1980s.
Researchers believe the chimpanzee virus is a hybrid of the simian immunodeficiency viruses (SIVs) naturally infecting two different monkey species: the red-capped mangabey (Cercocebus torquatus) and the greater spot-nosed monkey (Cercopithecus nictitans). Chimpanzees kill and eat monkeys, which is likely how they acquired the monkey viruses. The viruses hybridized in a chimpanzee; the hybrid virus then spread through the chimpanzee population and was later transmitted to humans who captured and slaughtered chimps for meat (becoming exposed to their blood). This was the most likely origin of HIV-1.
HIV-1 also shows one of the major risks of zoonotic infections. They can continue to mutate in its human host, increasing the risk of greater virulence, but also interfering with the production of a universally effective vaccine. Since its transmission to humans, for example, many subtypes of the HIV-1 strain have developed, with genetic differences even in the same subtypes found to be up to 20%.
Ebolavirus, first detected in 1976, is another case of bats being the potential culprit. Genetic analysis has shown that African fruit bats are likely involved in the spread of the virus and may be its reservoir host. Further evidence of this was found in the most recent human-infecting Bombali variant of the virus, which was identified in samples from bats collected from Sierra Leone.
It was also found that pigs can also become infected with Zaire ebolavirus, leading to the fear that pigs could serve as a mixing vessel for it and other filoviruses. Pigs have their own forms of Ebola-like disease viruses, which are not currently transmissible to humans, but could provide a potential mixing-vessel reservoir.
Emergent influenzas
The Western world has been most affected by these highly mutable, multispecies zoonotic viruses. The 1957 and 1968 flu pandemics contained a mixture of gene segments from human and avian influenza viruses. “What is clear from genetic analysis of the viruses that caused these past pandemics is that reassortment (gene swapping) occurred to produce novel influenza viruses that caused the pandemics. In both of these cases, the new viruses that emerged showed major differences from the parent viruses,” according to the Centers for Disease Control and Prevention.
Influenza is, however, a good example that all zoonoses are not the result of a mixing-vessel phenomenon, with evidence showing that the origin of the catastrophic 1918 virus pandemic likely resulted from a bird influenza virus directly infecting humans and pigs at about the same time without reassortment, according to the CDC.
Building a protective infrastructure
The first 2 decades of the 21st century saw a huge increase in efforts to develop an infrastructure to monitor and potentially prevent the spread of new zoonoses. As part of a global effort led by the United Nations, the U.S. Agency for International AID developed the PREDICT program in 2009 “to strengthen global capacity for detection and discovery of zoonotic viruses with pandemic potential. Those include coronaviruses, the family to which SARS and MERS belong; paramyxoviruses, like Nipah virus; influenza viruses; and filoviruses, like the ebolavirus.”
PREDICT funding to the EcoHealth Alliance led to discovery of the likely bat origins of the Zaire ebolavirus during the 2013-2016 outbreak. And throughout the existence of PREDICT, more than 145,000 animals and people were surveyed in areas of likely zoonotic outbreaks, leading to the detection of more than “1,100 unique viruses, including zoonotic diseases of public health concern such as Bombali ebolavirus, Zaire ebolavirus, Marburg virus, and MERS- and SARS-like coronaviruses,” according to PREDICT partner, the University of California, Davis.
PREDICT-2 was launched in 2014 with the continuing goals of “identifying and better characterizing pathogens of known epidemic and unknown pandemic potential; recognizing animal reservoirs and amplification hosts of human-infectious viruses; and efficiently targeting intervention action at human behaviors which amplify disease transmission at critical animal-animal and animal-human interfaces in hotspots of viral evolution, spillover, amplification, and spread.”
However, in October 2019, the Trump administration cut all funding to the PREDICT program, leading to its shutdown. In a New York Times interview, Peter Daszak, president of the EcoHealth Alliance, stated: “PREDICT was an approach to heading off pandemics, instead of sitting there waiting for them to emerge and then mobilizing.”
Ultimately, in addition to its human cost, the current Wuhan coronavirus outbreak can be looked at an object lesson – a test of the pandemic surveillance and control systems currently in place, and a practice run for the next and potentially deadlier zoonotic outbreaks to come. Perhaps it is also a reminder that cutting resources to detect zoonoses at their source in their animal hosts – before they enter the human chain– is perhaps not the most prudent of ideas.
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & celluar biology at Georgetown University, Washington.
Emerging viruses that spread to humans from an animal host are commonplace and represent some of the deadliest diseases known. Given the details of the Wuhan coronavirus (2019-nCoV) outbreak, including the genetic profile of the disease agent, the hypothesis of a snake origin was the first raised in the peer-reviewed literature.
It is a highly controversial origin story, however, given that mammals have been the sources of all other such zoonotic coronaviruses, as well as a host of other zoonotic diseases.
An animal source for emerging infections such as the 2019-nCoV is the default hypothesis, because “around 60% of all infectious diseases in humans are zoonotic, as are 75% of all emerging infectious diseases,” according to a United Nations report. The report goes on to say that, “on average, one new infectious disease emerges in humans every 4 months.”
To appreciate the emergence and nature of 2019-nCoV, it is important to examine the history of zoonotic outbreaks of other such diseases, especially with regard to the “mixing-vessel” phenomenon, which has been noted in closely related coronaviruses, including SARS and MERS, as well as the widely disparate HIV, Ebola, and influenza viruses.
Mutants in the mixing vessel
The mixing-vessel phenomenon is conceptually easy but molecularly complex. A single animal is coinfected with two related viruses; the virus genomes recombine together (virus “sex”) in that animal to form a new variant of virus. Such new mutant viruses can be more or less infective, more or less deadly, and more or less able to jump the species or even genus barrier. An emerging viral zoonosis can occur when a human being is exposed to one of these new viruses (either from the origin species or another species intermediate) that is capable of also infecting a human cell. Such exposure can occur from close proximity to animal waste or body fluids, as in the farm environment, or from wildlife pets or the capturing and slaughtering of wildlife for food, as is proposed in the case of the Wuhan seafood market scenario. In fact, the scientists who postulated a snake intermediary as the potential mixing vessel also stated that 2019‐nCoV appears to be a recombinant virus between a bat coronavirus and an origin‐unknown coronavirus.
Coronaviruses in particular have a history of moving from animal to human hosts (and even back again), and their detailed genetic pattern and taxonomy can reveal the animal origin of these diseases.
Going batty
Bats, in particular, have been shown to be a reservoir species for both alphacoronaviruses and betacoronaviruses. Given their ecology and behavior, they have been found to play a key role in transmitting coronaviruses between species. A highly pertinent example of this is the SARS coronavirus, which was shown to have likely originated in Chinese horseshoe bats. The SARS virus, which is genetically closely related to the new Wuhan coronavirus, first infected humans in the Guangdong province of southern China in 2002.
Scientists speculate that the virus was then either transmitted directly to humans from bats, or passed through an intermediate host species, with SARS-like viruses isolated from Himalayan palm civets found in a live-animal market in Guangdong. The virus infection was also detected in other animals (including a raccoon dog, Nyctereutes procyonoides) and in humans working at the market.
The MERS coronavirus is a betacoronavirus that was first reported in Saudi Arabia in 2012. It turned out to be far more deadly than either SARS or the Wuhan virus (at least as far as current estimates of the new coronavirus’s behavior). The MERS genotype was found to be closely related to MERS-like viruses in bats in Saudi Arabia, Africa, Europe, and Asia. Studies done on the cell receptor for MERS showed an apparently conserved viral receptor in both bats and humans. And an identical strain of MERS was found in bats in a nearby cave and near the workplace of the first known human patient.
However, in many of the other locations of the outbreak in the Middle East, there appeared to be limited contact between bats and humans, so scientists looked for another vector species, perhaps one that was acting as an intermediate. A high seroprevalence of MERS-CoV or a closely related virus was found in camels across the Arabian Peninsula and parts of eastern and northern Africa, while tests for MERS antibodies were negative in the most-likely other species of livestock or pet animals, including chickens, cows, goats, horses, and sheep.
In addition, the MERS-related CoV carried by camels was genetically highly similar to that detected in humans, as demonstrated in one particular outbreak on a farm in Qatar where the genetic sequences of MERS-CoV in the nasal swabs from 3 of 14 seropositive camels were similar to those of 2 human cases on the same farm. Similar genomic results were found in MERS-CoV from nasal swabs from camels in Saudi Arabia.
Other mixing-vessel zoonoses
HIV, the viral cause of AIDS, provides an almost-textbook origin story of the rise of a zoonotic supervillain. The virus was genetically traced to have a chimpanzee-to-human origin, but it was found to be more complicated than that. The virus first emerged in the 1920s in Africa in what is now the Democratic Republic of the Congo, well before its rise to a global pandemic in the 1980s.
Researchers believe the chimpanzee virus is a hybrid of the simian immunodeficiency viruses (SIVs) naturally infecting two different monkey species: the red-capped mangabey (Cercocebus torquatus) and the greater spot-nosed monkey (Cercopithecus nictitans). Chimpanzees kill and eat monkeys, which is likely how they acquired the monkey viruses. The viruses hybridized in a chimpanzee; the hybrid virus then spread through the chimpanzee population and was later transmitted to humans who captured and slaughtered chimps for meat (becoming exposed to their blood). This was the most likely origin of HIV-1.
HIV-1 also shows one of the major risks of zoonotic infections. They can continue to mutate in its human host, increasing the risk of greater virulence, but also interfering with the production of a universally effective vaccine. Since its transmission to humans, for example, many subtypes of the HIV-1 strain have developed, with genetic differences even in the same subtypes found to be up to 20%.
Ebolavirus, first detected in 1976, is another case of bats being the potential culprit. Genetic analysis has shown that African fruit bats are likely involved in the spread of the virus and may be its reservoir host. Further evidence of this was found in the most recent human-infecting Bombali variant of the virus, which was identified in samples from bats collected from Sierra Leone.
It was also found that pigs can also become infected with Zaire ebolavirus, leading to the fear that pigs could serve as a mixing vessel for it and other filoviruses. Pigs have their own forms of Ebola-like disease viruses, which are not currently transmissible to humans, but could provide a potential mixing-vessel reservoir.
Emergent influenzas
The Western world has been most affected by these highly mutable, multispecies zoonotic viruses. The 1957 and 1968 flu pandemics contained a mixture of gene segments from human and avian influenza viruses. “What is clear from genetic analysis of the viruses that caused these past pandemics is that reassortment (gene swapping) occurred to produce novel influenza viruses that caused the pandemics. In both of these cases, the new viruses that emerged showed major differences from the parent viruses,” according to the Centers for Disease Control and Prevention.
Influenza is, however, a good example that all zoonoses are not the result of a mixing-vessel phenomenon, with evidence showing that the origin of the catastrophic 1918 virus pandemic likely resulted from a bird influenza virus directly infecting humans and pigs at about the same time without reassortment, according to the CDC.
Building a protective infrastructure
The first 2 decades of the 21st century saw a huge increase in efforts to develop an infrastructure to monitor and potentially prevent the spread of new zoonoses. As part of a global effort led by the United Nations, the U.S. Agency for International AID developed the PREDICT program in 2009 “to strengthen global capacity for detection and discovery of zoonotic viruses with pandemic potential. Those include coronaviruses, the family to which SARS and MERS belong; paramyxoviruses, like Nipah virus; influenza viruses; and filoviruses, like the ebolavirus.”
PREDICT funding to the EcoHealth Alliance led to discovery of the likely bat origins of the Zaire ebolavirus during the 2013-2016 outbreak. And throughout the existence of PREDICT, more than 145,000 animals and people were surveyed in areas of likely zoonotic outbreaks, leading to the detection of more than “1,100 unique viruses, including zoonotic diseases of public health concern such as Bombali ebolavirus, Zaire ebolavirus, Marburg virus, and MERS- and SARS-like coronaviruses,” according to PREDICT partner, the University of California, Davis.
PREDICT-2 was launched in 2014 with the continuing goals of “identifying and better characterizing pathogens of known epidemic and unknown pandemic potential; recognizing animal reservoirs and amplification hosts of human-infectious viruses; and efficiently targeting intervention action at human behaviors which amplify disease transmission at critical animal-animal and animal-human interfaces in hotspots of viral evolution, spillover, amplification, and spread.”
However, in October 2019, the Trump administration cut all funding to the PREDICT program, leading to its shutdown. In a New York Times interview, Peter Daszak, president of the EcoHealth Alliance, stated: “PREDICT was an approach to heading off pandemics, instead of sitting there waiting for them to emerge and then mobilizing.”
Ultimately, in addition to its human cost, the current Wuhan coronavirus outbreak can be looked at an object lesson – a test of the pandemic surveillance and control systems currently in place, and a practice run for the next and potentially deadlier zoonotic outbreaks to come. Perhaps it is also a reminder that cutting resources to detect zoonoses at their source in their animal hosts – before they enter the human chain– is perhaps not the most prudent of ideas.
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & celluar biology at Georgetown University, Washington.
Emerging viruses that spread to humans from an animal host are commonplace and represent some of the deadliest diseases known. Given the details of the Wuhan coronavirus (2019-nCoV) outbreak, including the genetic profile of the disease agent, the hypothesis of a snake origin was the first raised in the peer-reviewed literature.
It is a highly controversial origin story, however, given that mammals have been the sources of all other such zoonotic coronaviruses, as well as a host of other zoonotic diseases.
An animal source for emerging infections such as the 2019-nCoV is the default hypothesis, because “around 60% of all infectious diseases in humans are zoonotic, as are 75% of all emerging infectious diseases,” according to a United Nations report. The report goes on to say that, “on average, one new infectious disease emerges in humans every 4 months.”
To appreciate the emergence and nature of 2019-nCoV, it is important to examine the history of zoonotic outbreaks of other such diseases, especially with regard to the “mixing-vessel” phenomenon, which has been noted in closely related coronaviruses, including SARS and MERS, as well as the widely disparate HIV, Ebola, and influenza viruses.
Mutants in the mixing vessel
The mixing-vessel phenomenon is conceptually easy but molecularly complex. A single animal is coinfected with two related viruses; the virus genomes recombine together (virus “sex”) in that animal to form a new variant of virus. Such new mutant viruses can be more or less infective, more or less deadly, and more or less able to jump the species or even genus barrier. An emerging viral zoonosis can occur when a human being is exposed to one of these new viruses (either from the origin species or another species intermediate) that is capable of also infecting a human cell. Such exposure can occur from close proximity to animal waste or body fluids, as in the farm environment, or from wildlife pets or the capturing and slaughtering of wildlife for food, as is proposed in the case of the Wuhan seafood market scenario. In fact, the scientists who postulated a snake intermediary as the potential mixing vessel also stated that 2019‐nCoV appears to be a recombinant virus between a bat coronavirus and an origin‐unknown coronavirus.
Coronaviruses in particular have a history of moving from animal to human hosts (and even back again), and their detailed genetic pattern and taxonomy can reveal the animal origin of these diseases.
Going batty
Bats, in particular, have been shown to be a reservoir species for both alphacoronaviruses and betacoronaviruses. Given their ecology and behavior, they have been found to play a key role in transmitting coronaviruses between species. A highly pertinent example of this is the SARS coronavirus, which was shown to have likely originated in Chinese horseshoe bats. The SARS virus, which is genetically closely related to the new Wuhan coronavirus, first infected humans in the Guangdong province of southern China in 2002.
Scientists speculate that the virus was then either transmitted directly to humans from bats, or passed through an intermediate host species, with SARS-like viruses isolated from Himalayan palm civets found in a live-animal market in Guangdong. The virus infection was also detected in other animals (including a raccoon dog, Nyctereutes procyonoides) and in humans working at the market.
The MERS coronavirus is a betacoronavirus that was first reported in Saudi Arabia in 2012. It turned out to be far more deadly than either SARS or the Wuhan virus (at least as far as current estimates of the new coronavirus’s behavior). The MERS genotype was found to be closely related to MERS-like viruses in bats in Saudi Arabia, Africa, Europe, and Asia. Studies done on the cell receptor for MERS showed an apparently conserved viral receptor in both bats and humans. And an identical strain of MERS was found in bats in a nearby cave and near the workplace of the first known human patient.
However, in many of the other locations of the outbreak in the Middle East, there appeared to be limited contact between bats and humans, so scientists looked for another vector species, perhaps one that was acting as an intermediate. A high seroprevalence of MERS-CoV or a closely related virus was found in camels across the Arabian Peninsula and parts of eastern and northern Africa, while tests for MERS antibodies were negative in the most-likely other species of livestock or pet animals, including chickens, cows, goats, horses, and sheep.
In addition, the MERS-related CoV carried by camels was genetically highly similar to that detected in humans, as demonstrated in one particular outbreak on a farm in Qatar where the genetic sequences of MERS-CoV in the nasal swabs from 3 of 14 seropositive camels were similar to those of 2 human cases on the same farm. Similar genomic results were found in MERS-CoV from nasal swabs from camels in Saudi Arabia.
Other mixing-vessel zoonoses
HIV, the viral cause of AIDS, provides an almost-textbook origin story of the rise of a zoonotic supervillain. The virus was genetically traced to have a chimpanzee-to-human origin, but it was found to be more complicated than that. The virus first emerged in the 1920s in Africa in what is now the Democratic Republic of the Congo, well before its rise to a global pandemic in the 1980s.
Researchers believe the chimpanzee virus is a hybrid of the simian immunodeficiency viruses (SIVs) naturally infecting two different monkey species: the red-capped mangabey (Cercocebus torquatus) and the greater spot-nosed monkey (Cercopithecus nictitans). Chimpanzees kill and eat monkeys, which is likely how they acquired the monkey viruses. The viruses hybridized in a chimpanzee; the hybrid virus then spread through the chimpanzee population and was later transmitted to humans who captured and slaughtered chimps for meat (becoming exposed to their blood). This was the most likely origin of HIV-1.
HIV-1 also shows one of the major risks of zoonotic infections. They can continue to mutate in its human host, increasing the risk of greater virulence, but also interfering with the production of a universally effective vaccine. Since its transmission to humans, for example, many subtypes of the HIV-1 strain have developed, with genetic differences even in the same subtypes found to be up to 20%.
Ebolavirus, first detected in 1976, is another case of bats being the potential culprit. Genetic analysis has shown that African fruit bats are likely involved in the spread of the virus and may be its reservoir host. Further evidence of this was found in the most recent human-infecting Bombali variant of the virus, which was identified in samples from bats collected from Sierra Leone.
It was also found that pigs can also become infected with Zaire ebolavirus, leading to the fear that pigs could serve as a mixing vessel for it and other filoviruses. Pigs have their own forms of Ebola-like disease viruses, which are not currently transmissible to humans, but could provide a potential mixing-vessel reservoir.
Emergent influenzas
The Western world has been most affected by these highly mutable, multispecies zoonotic viruses. The 1957 and 1968 flu pandemics contained a mixture of gene segments from human and avian influenza viruses. “What is clear from genetic analysis of the viruses that caused these past pandemics is that reassortment (gene swapping) occurred to produce novel influenza viruses that caused the pandemics. In both of these cases, the new viruses that emerged showed major differences from the parent viruses,” according to the Centers for Disease Control and Prevention.
Influenza is, however, a good example that all zoonoses are not the result of a mixing-vessel phenomenon, with evidence showing that the origin of the catastrophic 1918 virus pandemic likely resulted from a bird influenza virus directly infecting humans and pigs at about the same time without reassortment, according to the CDC.
Building a protective infrastructure
The first 2 decades of the 21st century saw a huge increase in efforts to develop an infrastructure to monitor and potentially prevent the spread of new zoonoses. As part of a global effort led by the United Nations, the U.S. Agency for International AID developed the PREDICT program in 2009 “to strengthen global capacity for detection and discovery of zoonotic viruses with pandemic potential. Those include coronaviruses, the family to which SARS and MERS belong; paramyxoviruses, like Nipah virus; influenza viruses; and filoviruses, like the ebolavirus.”
PREDICT funding to the EcoHealth Alliance led to discovery of the likely bat origins of the Zaire ebolavirus during the 2013-2016 outbreak. And throughout the existence of PREDICT, more than 145,000 animals and people were surveyed in areas of likely zoonotic outbreaks, leading to the detection of more than “1,100 unique viruses, including zoonotic diseases of public health concern such as Bombali ebolavirus, Zaire ebolavirus, Marburg virus, and MERS- and SARS-like coronaviruses,” according to PREDICT partner, the University of California, Davis.
PREDICT-2 was launched in 2014 with the continuing goals of “identifying and better characterizing pathogens of known epidemic and unknown pandemic potential; recognizing animal reservoirs and amplification hosts of human-infectious viruses; and efficiently targeting intervention action at human behaviors which amplify disease transmission at critical animal-animal and animal-human interfaces in hotspots of viral evolution, spillover, amplification, and spread.”
However, in October 2019, the Trump administration cut all funding to the PREDICT program, leading to its shutdown. In a New York Times interview, Peter Daszak, president of the EcoHealth Alliance, stated: “PREDICT was an approach to heading off pandemics, instead of sitting there waiting for them to emerge and then mobilizing.”
Ultimately, in addition to its human cost, the current Wuhan coronavirus outbreak can be looked at an object lesson – a test of the pandemic surveillance and control systems currently in place, and a practice run for the next and potentially deadlier zoonotic outbreaks to come. Perhaps it is also a reminder that cutting resources to detect zoonoses at their source in their animal hosts – before they enter the human chain– is perhaps not the most prudent of ideas.
Mark Lesney is the managing editor of MDedge.com/IDPractioner. He has a PhD in plant virology and a PhD in the history of science, with a focus on the history of biotechnology and medicine. He has served as an adjunct assistant professor of the department of biochemistry and molecular & celluar biology at Georgetown University, Washington.
Serum keratin 18 promising as AAH biomarker
Outperforms MELD, ABIC
.
Standard biomarker scores – Model for End-stage Liver Disease (MELD), age, serum bilirubin, International Normalized Ratio, and serum creatinine (ABIC), as well as others – predict prognosis and severity of alcoholic liver disease, but they don’t reflect “the magnitude of cell death nor the form of cell death (apoptosis/necrosis), which may be important in distinguishing various forms of liver injury” and guiding therapy, explained investigators led by Vatsalya Vatsalya, MD, of the division of gastroenterology, hepatology, and nutrition at the University of Louisville (Ky.).
It’s important, for instance, to identify people with alcoholic cirrhosis but not active hepatitis, as they “would likely not benefit from anti-inflammatory agents such as steroids or [interleukin]-1 receptor antagonists, but would incur their side effects.” For those and other reasons, “new biomarkers are needed for diagnosing AAH, assessing the degree of hepatocyte death, and predicting mortality,” they said (Clin Gastroenterol Hepatol. 2019 Dec 4. doi: 10.1016/j.cgh.2019.11.050).
Keratin 18 – both the cleaved form (K18M30) and the uncleaved protein (K18M65) – have been suggested before as a marker for AAH, so the investigators took a closer look.
They analyzed serum from 57 people with severe AAH (MELD score above 20), 27 people with moderate AAH (MELD score 12-19), 34 with nonalcoholic steatohepatitis, 17 healthy controls, and 38 people with alcohol use disorder and either mild or no liver injury.
Overall, 51.9% of moderate AAH cases and 38.9% of severe cases had K18M65 levels between 641 and 2,000 IU/L; 25.9% of moderate and 61.1% of severe cases had K18M65 levels greater than 2,000 IU/L. All severe cases had levels above 641 IU/L. Serum levels of K18 also identified patients who died within 90 days with greater accuracy than did MELD, ABIC, and other scores, the investigators said.
The K18M65:ALT [alanine aminotransferase] ratio distinguished AAH from nonalcoholic steatohepatitis with a sensitivity of 0.971 and specificity of 0.829. Findings were similar for the K18M30:ALT ratio.
Levels of K18M65 and K18M30 increased significantly as liver disease worsened, as did the degree of necrosis as indicated by the K18M65:K18M30 ratio. Meanwhile, although k18 levels correlated with MELD scores, levels of ALT, aspartate aminotransferase (AST), and the ratio of AST:ALT did not.
“There is a stronger association between serum level of keratin 18 and amount of hepatocyte death and liver disease severity than for other biomarkers,” the team concluded.
Patients were in their mid 40s, on average, and there were more men than women.
The National Institutes of Health supported the work, and the investigators had no disclosures.
Outperforms MELD, ABIC
Outperforms MELD, ABIC
.
Standard biomarker scores – Model for End-stage Liver Disease (MELD), age, serum bilirubin, International Normalized Ratio, and serum creatinine (ABIC), as well as others – predict prognosis and severity of alcoholic liver disease, but they don’t reflect “the magnitude of cell death nor the form of cell death (apoptosis/necrosis), which may be important in distinguishing various forms of liver injury” and guiding therapy, explained investigators led by Vatsalya Vatsalya, MD, of the division of gastroenterology, hepatology, and nutrition at the University of Louisville (Ky.).
It’s important, for instance, to identify people with alcoholic cirrhosis but not active hepatitis, as they “would likely not benefit from anti-inflammatory agents such as steroids or [interleukin]-1 receptor antagonists, but would incur their side effects.” For those and other reasons, “new biomarkers are needed for diagnosing AAH, assessing the degree of hepatocyte death, and predicting mortality,” they said (Clin Gastroenterol Hepatol. 2019 Dec 4. doi: 10.1016/j.cgh.2019.11.050).
Keratin 18 – both the cleaved form (K18M30) and the uncleaved protein (K18M65) – have been suggested before as a marker for AAH, so the investigators took a closer look.
They analyzed serum from 57 people with severe AAH (MELD score above 20), 27 people with moderate AAH (MELD score 12-19), 34 with nonalcoholic steatohepatitis, 17 healthy controls, and 38 people with alcohol use disorder and either mild or no liver injury.
Overall, 51.9% of moderate AAH cases and 38.9% of severe cases had K18M65 levels between 641 and 2,000 IU/L; 25.9% of moderate and 61.1% of severe cases had K18M65 levels greater than 2,000 IU/L. All severe cases had levels above 641 IU/L. Serum levels of K18 also identified patients who died within 90 days with greater accuracy than did MELD, ABIC, and other scores, the investigators said.
The K18M65:ALT [alanine aminotransferase] ratio distinguished AAH from nonalcoholic steatohepatitis with a sensitivity of 0.971 and specificity of 0.829. Findings were similar for the K18M30:ALT ratio.
Levels of K18M65 and K18M30 increased significantly as liver disease worsened, as did the degree of necrosis as indicated by the K18M65:K18M30 ratio. Meanwhile, although k18 levels correlated with MELD scores, levels of ALT, aspartate aminotransferase (AST), and the ratio of AST:ALT did not.
“There is a stronger association between serum level of keratin 18 and amount of hepatocyte death and liver disease severity than for other biomarkers,” the team concluded.
Patients were in their mid 40s, on average, and there were more men than women.
The National Institutes of Health supported the work, and the investigators had no disclosures.
.
Standard biomarker scores – Model for End-stage Liver Disease (MELD), age, serum bilirubin, International Normalized Ratio, and serum creatinine (ABIC), as well as others – predict prognosis and severity of alcoholic liver disease, but they don’t reflect “the magnitude of cell death nor the form of cell death (apoptosis/necrosis), which may be important in distinguishing various forms of liver injury” and guiding therapy, explained investigators led by Vatsalya Vatsalya, MD, of the division of gastroenterology, hepatology, and nutrition at the University of Louisville (Ky.).
It’s important, for instance, to identify people with alcoholic cirrhosis but not active hepatitis, as they “would likely not benefit from anti-inflammatory agents such as steroids or [interleukin]-1 receptor antagonists, but would incur their side effects.” For those and other reasons, “new biomarkers are needed for diagnosing AAH, assessing the degree of hepatocyte death, and predicting mortality,” they said (Clin Gastroenterol Hepatol. 2019 Dec 4. doi: 10.1016/j.cgh.2019.11.050).
Keratin 18 – both the cleaved form (K18M30) and the uncleaved protein (K18M65) – have been suggested before as a marker for AAH, so the investigators took a closer look.
They analyzed serum from 57 people with severe AAH (MELD score above 20), 27 people with moderate AAH (MELD score 12-19), 34 with nonalcoholic steatohepatitis, 17 healthy controls, and 38 people with alcohol use disorder and either mild or no liver injury.
Overall, 51.9% of moderate AAH cases and 38.9% of severe cases had K18M65 levels between 641 and 2,000 IU/L; 25.9% of moderate and 61.1% of severe cases had K18M65 levels greater than 2,000 IU/L. All severe cases had levels above 641 IU/L. Serum levels of K18 also identified patients who died within 90 days with greater accuracy than did MELD, ABIC, and other scores, the investigators said.
The K18M65:ALT [alanine aminotransferase] ratio distinguished AAH from nonalcoholic steatohepatitis with a sensitivity of 0.971 and specificity of 0.829. Findings were similar for the K18M30:ALT ratio.
Levels of K18M65 and K18M30 increased significantly as liver disease worsened, as did the degree of necrosis as indicated by the K18M65:K18M30 ratio. Meanwhile, although k18 levels correlated with MELD scores, levels of ALT, aspartate aminotransferase (AST), and the ratio of AST:ALT did not.
“There is a stronger association between serum level of keratin 18 and amount of hepatocyte death and liver disease severity than for other biomarkers,” the team concluded.
Patients were in their mid 40s, on average, and there were more men than women.
The National Institutes of Health supported the work, and the investigators had no disclosures.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Performing gender-reaffirming surgery: Guidelines for the general ob.gyn.
According to the DSM-V, gender dysphoria in adolescents and adults “involves a difference between one’s experienced/expressed gender and assigned gender, and significant distress or problems functioning. It lasts at least 6 months,” and several other criteria must be met.1 Many patients with gender dysphoria also identify as transgender. A “transition” or “transitioning” is a process by which individuals come to inhabit their gender identity.2 A gender transition may take many forms, and only some people will choose to include medical assistance in their transition process. Although the scope of this article will not address these concerns, it should be noted that many people in the transgender and gender nonconforming community would object to the concepts of gender dysphoria and gender transition because they rely on a binary model of gender that may exclude individuals that see themselves as something other than “man or woman.”
There are both medical and surgical options for medical assistance in a gender transition. This article will focus on the surgical care of patients assigned female at birth who are seeking masculinizing surgical therapy. Many writers will discuss “gender-affirming” surgery, but we will use the term “gender-reaffirming” surgery because transgender patients have already affirmed their own genders and do not require surgery to inhabit this affirmation. Surgical options might include bilateral mastectomy, hysterectomy, bilateral salpingo-oophorectomy (BSO), metoidioplasty (surgical formation of a neophallus with existing genital tissue), or phalloplasty. There currently is no single surgical subspecialty that encompasses training in all forms of gender-reaffirming surgical therapies. In some areas of the country, centers of excellence have given rise to multidisciplinary teams that combine the skill sets of surgical subspecialists to provide a streamlined approach to gender-reaffirming surgery. Because of the scarcity of these integrated centers, most patients seeking gender-reaffirming surgeries will need to find individual subspecialists whose surgical training focuses on one area of the body. For example, patients seeking all possible surgical options may need a breast surgeon to perform their mastectomy, an ob.gyn. to perform their hysterectomy and BSO, a urologist to perform their metoidioplasty, and a plastic surgeon to perform their phalloplasty. In these scenarios,
There are many reasons why transgender men might desire hysterectomy/BSO as part of their transition. Removal of the uterus and cervix eliminates concerns surrounding menstruation, pregnancy, and cervical cancer screening, all of which may add to their experience of gender dysphoria. Furthermore, removal of the ovaries may simplify long-term hormonal therapy with testosterone by eliminating the need for estrogen suppression. Lastly, a hysterectomy/BSO is a lower-risk and more cost-effective masculinizing surgery, compared with metoidioplasty or phalloplasty.
While the technical aspect of performing a hysterectomy/BSO certainly is within the scope of training for a general ob.gyn., there are several nuances of which providers should be aware when planning gender-reaffirming surgery for a transgender man. During the preoperative planning phase, it is of utmost importance to provide an environment of safety so that the focus of the preop visit is not clouded by communication mishaps between office staff and the patient. These barriers can be avoided by implementing office intake forms that give patients the opportunity to inform the health care team of their chosen name and personal pronouns upon registration for the visit.
A pelvic exam is commonly performed by ob.gyns. to determine surgical approach for a hysterectomy/BSO. When approaching transgender male patients for preoperative pelvic exams, it is important to be mindful of the fact that this type of exam may trigger gender dysphoria. While pelvic exams should be handled in sensitive fashion regardless of a patient’s gender identity, a patient who is a transgender man may benefit from some added steps in discussing the pelvic exam. One approach is to acknowledge that these exams/discussions may be especially triggering of gender dysphoria, and ask if the patient would prefer certain words to be used or not used in reference to their anatomy. As with any patient, the provider should explain the purpose of the examination and offer opportunities for the patient to have some control in the exam such as by assisting with insertion of the speculum or designating a “safe word” that would signal the provider to stop or pause the exam. In some cases, patients may not be able to tolerate the pelvic exam while awake because of the degree of gender dysphoria that the exam would induce. Providers might consider noninvasive imaging studies to help with surgical planning if they find they need more information before scheduling the operation, or they may offer a staged procedure with exam under anesthesia prior to the definitive surgery.
In conclusion, performing a gender-reaffirming hysterectomy/BSO requires thoughtful preparation to ensure a safe surgical environment for this vulnerable population. Care should be taken to plan the operation with a culturally sensitive approach.
Dr. Joyner is an assistant professor at Emory University, and is the director of gynecologic services in the Gender Center at Grady Memorial Hospital, both in Atlanta. Dr. Joyner identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Joey Bahng is a PGY-1 resident physician in Emory University’s gynecology & obstetrics residency program. Dr. Bahng identifies as nonbinary and uses they/them/their as their personal pronouns. Dr. Joyner and Dr. Bahng reported no relevant financial disclosures.
References
1. American Psychiatric Association. What is Gender Dysphoria? https://www.psychiatry.org/patients-families/gender-dysphoria/what-is-gender-dysphoria
2. UCSF Transgender Care. Transition Roadmap. https://transcare.ucsf.edu/transition-roadmap
According to the DSM-V, gender dysphoria in adolescents and adults “involves a difference between one’s experienced/expressed gender and assigned gender, and significant distress or problems functioning. It lasts at least 6 months,” and several other criteria must be met.1 Many patients with gender dysphoria also identify as transgender. A “transition” or “transitioning” is a process by which individuals come to inhabit their gender identity.2 A gender transition may take many forms, and only some people will choose to include medical assistance in their transition process. Although the scope of this article will not address these concerns, it should be noted that many people in the transgender and gender nonconforming community would object to the concepts of gender dysphoria and gender transition because they rely on a binary model of gender that may exclude individuals that see themselves as something other than “man or woman.”
There are both medical and surgical options for medical assistance in a gender transition. This article will focus on the surgical care of patients assigned female at birth who are seeking masculinizing surgical therapy. Many writers will discuss “gender-affirming” surgery, but we will use the term “gender-reaffirming” surgery because transgender patients have already affirmed their own genders and do not require surgery to inhabit this affirmation. Surgical options might include bilateral mastectomy, hysterectomy, bilateral salpingo-oophorectomy (BSO), metoidioplasty (surgical formation of a neophallus with existing genital tissue), or phalloplasty. There currently is no single surgical subspecialty that encompasses training in all forms of gender-reaffirming surgical therapies. In some areas of the country, centers of excellence have given rise to multidisciplinary teams that combine the skill sets of surgical subspecialists to provide a streamlined approach to gender-reaffirming surgery. Because of the scarcity of these integrated centers, most patients seeking gender-reaffirming surgeries will need to find individual subspecialists whose surgical training focuses on one area of the body. For example, patients seeking all possible surgical options may need a breast surgeon to perform their mastectomy, an ob.gyn. to perform their hysterectomy and BSO, a urologist to perform their metoidioplasty, and a plastic surgeon to perform their phalloplasty. In these scenarios,
There are many reasons why transgender men might desire hysterectomy/BSO as part of their transition. Removal of the uterus and cervix eliminates concerns surrounding menstruation, pregnancy, and cervical cancer screening, all of which may add to their experience of gender dysphoria. Furthermore, removal of the ovaries may simplify long-term hormonal therapy with testosterone by eliminating the need for estrogen suppression. Lastly, a hysterectomy/BSO is a lower-risk and more cost-effective masculinizing surgery, compared with metoidioplasty or phalloplasty.
While the technical aspect of performing a hysterectomy/BSO certainly is within the scope of training for a general ob.gyn., there are several nuances of which providers should be aware when planning gender-reaffirming surgery for a transgender man. During the preoperative planning phase, it is of utmost importance to provide an environment of safety so that the focus of the preop visit is not clouded by communication mishaps between office staff and the patient. These barriers can be avoided by implementing office intake forms that give patients the opportunity to inform the health care team of their chosen name and personal pronouns upon registration for the visit.
A pelvic exam is commonly performed by ob.gyns. to determine surgical approach for a hysterectomy/BSO. When approaching transgender male patients for preoperative pelvic exams, it is important to be mindful of the fact that this type of exam may trigger gender dysphoria. While pelvic exams should be handled in sensitive fashion regardless of a patient’s gender identity, a patient who is a transgender man may benefit from some added steps in discussing the pelvic exam. One approach is to acknowledge that these exams/discussions may be especially triggering of gender dysphoria, and ask if the patient would prefer certain words to be used or not used in reference to their anatomy. As with any patient, the provider should explain the purpose of the examination and offer opportunities for the patient to have some control in the exam such as by assisting with insertion of the speculum or designating a “safe word” that would signal the provider to stop or pause the exam. In some cases, patients may not be able to tolerate the pelvic exam while awake because of the degree of gender dysphoria that the exam would induce. Providers might consider noninvasive imaging studies to help with surgical planning if they find they need more information before scheduling the operation, or they may offer a staged procedure with exam under anesthesia prior to the definitive surgery.
In conclusion, performing a gender-reaffirming hysterectomy/BSO requires thoughtful preparation to ensure a safe surgical environment for this vulnerable population. Care should be taken to plan the operation with a culturally sensitive approach.
Dr. Joyner is an assistant professor at Emory University, and is the director of gynecologic services in the Gender Center at Grady Memorial Hospital, both in Atlanta. Dr. Joyner identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Joey Bahng is a PGY-1 resident physician in Emory University’s gynecology & obstetrics residency program. Dr. Bahng identifies as nonbinary and uses they/them/their as their personal pronouns. Dr. Joyner and Dr. Bahng reported no relevant financial disclosures.
References
1. American Psychiatric Association. What is Gender Dysphoria? https://www.psychiatry.org/patients-families/gender-dysphoria/what-is-gender-dysphoria
2. UCSF Transgender Care. Transition Roadmap. https://transcare.ucsf.edu/transition-roadmap
According to the DSM-V, gender dysphoria in adolescents and adults “involves a difference between one’s experienced/expressed gender and assigned gender, and significant distress or problems functioning. It lasts at least 6 months,” and several other criteria must be met.1 Many patients with gender dysphoria also identify as transgender. A “transition” or “transitioning” is a process by which individuals come to inhabit their gender identity.2 A gender transition may take many forms, and only some people will choose to include medical assistance in their transition process. Although the scope of this article will not address these concerns, it should be noted that many people in the transgender and gender nonconforming community would object to the concepts of gender dysphoria and gender transition because they rely on a binary model of gender that may exclude individuals that see themselves as something other than “man or woman.”
There are both medical and surgical options for medical assistance in a gender transition. This article will focus on the surgical care of patients assigned female at birth who are seeking masculinizing surgical therapy. Many writers will discuss “gender-affirming” surgery, but we will use the term “gender-reaffirming” surgery because transgender patients have already affirmed their own genders and do not require surgery to inhabit this affirmation. Surgical options might include bilateral mastectomy, hysterectomy, bilateral salpingo-oophorectomy (BSO), metoidioplasty (surgical formation of a neophallus with existing genital tissue), or phalloplasty. There currently is no single surgical subspecialty that encompasses training in all forms of gender-reaffirming surgical therapies. In some areas of the country, centers of excellence have given rise to multidisciplinary teams that combine the skill sets of surgical subspecialists to provide a streamlined approach to gender-reaffirming surgery. Because of the scarcity of these integrated centers, most patients seeking gender-reaffirming surgeries will need to find individual subspecialists whose surgical training focuses on one area of the body. For example, patients seeking all possible surgical options may need a breast surgeon to perform their mastectomy, an ob.gyn. to perform their hysterectomy and BSO, a urologist to perform their metoidioplasty, and a plastic surgeon to perform their phalloplasty. In these scenarios,
There are many reasons why transgender men might desire hysterectomy/BSO as part of their transition. Removal of the uterus and cervix eliminates concerns surrounding menstruation, pregnancy, and cervical cancer screening, all of which may add to their experience of gender dysphoria. Furthermore, removal of the ovaries may simplify long-term hormonal therapy with testosterone by eliminating the need for estrogen suppression. Lastly, a hysterectomy/BSO is a lower-risk and more cost-effective masculinizing surgery, compared with metoidioplasty or phalloplasty.
While the technical aspect of performing a hysterectomy/BSO certainly is within the scope of training for a general ob.gyn., there are several nuances of which providers should be aware when planning gender-reaffirming surgery for a transgender man. During the preoperative planning phase, it is of utmost importance to provide an environment of safety so that the focus of the preop visit is not clouded by communication mishaps between office staff and the patient. These barriers can be avoided by implementing office intake forms that give patients the opportunity to inform the health care team of their chosen name and personal pronouns upon registration for the visit.
A pelvic exam is commonly performed by ob.gyns. to determine surgical approach for a hysterectomy/BSO. When approaching transgender male patients for preoperative pelvic exams, it is important to be mindful of the fact that this type of exam may trigger gender dysphoria. While pelvic exams should be handled in sensitive fashion regardless of a patient’s gender identity, a patient who is a transgender man may benefit from some added steps in discussing the pelvic exam. One approach is to acknowledge that these exams/discussions may be especially triggering of gender dysphoria, and ask if the patient would prefer certain words to be used or not used in reference to their anatomy. As with any patient, the provider should explain the purpose of the examination and offer opportunities for the patient to have some control in the exam such as by assisting with insertion of the speculum or designating a “safe word” that would signal the provider to stop or pause the exam. In some cases, patients may not be able to tolerate the pelvic exam while awake because of the degree of gender dysphoria that the exam would induce. Providers might consider noninvasive imaging studies to help with surgical planning if they find they need more information before scheduling the operation, or they may offer a staged procedure with exam under anesthesia prior to the definitive surgery.
In conclusion, performing a gender-reaffirming hysterectomy/BSO requires thoughtful preparation to ensure a safe surgical environment for this vulnerable population. Care should be taken to plan the operation with a culturally sensitive approach.
Dr. Joyner is an assistant professor at Emory University, and is the director of gynecologic services in the Gender Center at Grady Memorial Hospital, both in Atlanta. Dr. Joyner identifies as a cisgender female and uses she/hers/her as her personal pronouns. Dr. Joey Bahng is a PGY-1 resident physician in Emory University’s gynecology & obstetrics residency program. Dr. Bahng identifies as nonbinary and uses they/them/their as their personal pronouns. Dr. Joyner and Dr. Bahng reported no relevant financial disclosures.
References
1. American Psychiatric Association. What is Gender Dysphoria? https://www.psychiatry.org/patients-families/gender-dysphoria/what-is-gender-dysphoria
2. UCSF Transgender Care. Transition Roadmap. https://transcare.ucsf.edu/transition-roadmap
Molar pregnancy: The next steps after diagnosis
Molar pregnancy is an uncommon but serious condition that affects young women of reproductive age. The diagnosis and management of molar pregnancy is familiar to most gynecologists. However, in the days and weeks following evacuation of molar pregnancy, clinicians face a critical time period in which they must be vigilant for the development of postmolar gestational trophoblastic neoplasia (GTN). If recognized early and treated appropriately, it almost always can be cured; however, errors or delays in the management of this condition can have catastrophic consequences for patients, including decreasing the likelihood of cure. Here we will review some of the steps and actions that can be taken immediately following the diagnosis of a molar pregnancy to expeditiously identify postmolar GTN and ensure patients are appropriately prepared for further consultation and intervention.
Postmolar GTN includes the diagnoses of invasive mole and choriocarcinoma that contain highly atypical trophoblasts with the capacity for local invasion and metastasis. Typically, the diagnosis is made clinically and not distinguished with histology. While molar pregnancies are a benign condition, invasive moles and choriocarcinoma are malignant conditions in which the molar tissue infiltrates the uterine myometrium, vasculature, and frequently is associated with hematogenous spread with distant metastases. It is a highly chemosensitive disease, and cure with chemotherapy typically is achieved with the ability to preserve fertility if desired even in advanced stage disease.1
After evacuation of a molar pregnancy, gynecologists should be on alert for the development of postmolar GTN if the following known risk factors are present: a history of a prior GTN diagnosis, complete mole on pathology (as opposed to partial mole), serum human chorionic gonadotropin (hCG) levels greater than 100,000 mIU/mL, age greater than 40 years, an enlarged uterus or large ovarian theca lutein cysts, and slow to normalize (more than 2 months) hCG. Symptoms for the development of postmolar GTN include persistent vaginal bleeding after evacuation, a persistently enlarged or enlarging uterine size, and adnexal masses. Ultimately, the diagnosis is made through plateaued or rising serum hCG assessments.2 (See graphic.)
Following the evacuation of a molar pregnancy, hCG levels should be drawn at the same laboratory every 1-2 weeks until normalization and then three consecutive normal values. Once this has been achieved, hCG levels should be tested once at 3 months and again at 6 months. During this 6 month period, patients should use reliable contraception, ideally, and through oral contraceptive pills that suppress the secretion of pituitary hCG if not contraindicated. Should a woman become pregnant during this 6-month surveillance, it becomes impossible to rule out occult postmolar GTN.
Typically after evacuation of a molar pregnancy, there is rapid fall in hCG levels, but this does not occur when the molar pregnancy has become invasive or is associated with choriocarcinoma. In these cases, after an initial drop in hCG levels, there is an observed rise or plateau in levels (as defined in the accompanying table), and this establishes the diagnosis of postmolar GTN. It is common for hCG to fall in fits and starts, rather than have a smooth, consistent diminution, and this can be worrying for gynecologists; however, provided there is a consistent reduction in values in accordance with the stated definitions, observation can continue.
Another source of confusion and concern is an HCG level that fails to completely normalize during observation, yet reaches a very low level. If this is observed, clinicians should consider the diagnosis of quiescent hCG, pituitary hCG, or phantom hCG.3 These can be difficult to distinguish from postmolar GTN, and consultation with a gynecologic oncologist with experience in the diagnosis and management of these rare tumors is helpful to determine if the persistent low levels in hCG require intervention.
Once a clinician has observed a plateau or rise in hCG levels, a gynecologic examination should be performed because the lower genital tract is a common site for metastatic postmolar GTN. If during this evaluation, a suspicious lesion is identified (typically a blue-black, slightly raised, hemorrhagic-appearing lesion), it should not be biopsied, but rather assumed to be a metastatic site. The vasculature of metastatic sites is extremely fragile, and biopsy or disruption can result in catastrophic hemorrhage, even from very small lesions.
In addition to physical examination, several diagnostic studies should be performed which may expedite the triage and management of the case. A pelvic ultrasound should evaluate the endometrial cavity for a new viable pregnancy, and residual molar tissue; sometimes, myometrial invasion consistent with an invasive mole can be appreciated. Chest x-ray or CT scan should be ordered to evaluate for pulmonary metastatic lesions. Additionally, CT scans of the abdomen and pelvis should be ordered, and if lung metastases are present, brain imaging with either MRI or CT scan also should be obtained. These imaging studies will provide the necessary information to stage the GTN (as metastatic or not).
Treatment for postmolar GTN is determined based on further prognostic categorization (“high risk” or “low risk”) in accordance with the WHO classification, which is derived using several prognostic clinical variables including age, antecedent pregnancy, interval from index pregnancy, pretreatment hCG, largest tumor size, sites and number of metastases, and response to previous chemotherapy.4 These assignments are necessary to determine whether single-agent or multiagent chemotherapy should be prescribed.
Laboratory studies are helpful to obtain at this time and include metabolic panels (which can ensure that renal and hepatic function are within normal limits in anticipation of future chemotherapy), and complete blood count ,which can establish viable bone marrow function prior to chemotherapy.
Once postmolar GTN has been diagnosed, it is most appropriate to refer the patient to a gynecologic oncologist with experience in the treatment of these relatively rare malignancies. At that point, the patient will be formally staged, and offered treatment based on these staging results.
Among women with low-risk, nonmetastatic GTN who desire future fertility it is appropriate to offer a repeat dilation and curettage (D&C) procedure rather than immediately proceeding with chemotherapy. Approximately two-thirds of women with low risk disease can avoid chemotherapy with repeat curettage.5 Risk factors for needing chemotherapy after repeat D&C include the presence of trophoblastic disease in the pathology specimen and urinary hCG levels greater than 1,500 mIU/mL at the time of curettage. In my experience, many women appreciate this option to potentially avoid toxic chemotherapy.
For women with low-risk, nonmetastatic postmolar GTN who do not desire future fertility, and hope to avoid chemotherapy, hysterectomy also is a reasonable first option. This can be performed via either minimally invasive, laparotomy, or vaginal route. If performing a minimally invasive procedure in the setting of GTN, there should be caution or avoidance of use of a uterine manipulator because the uterine wall typically is soft and prone to perforation, and bleeding can be significant secondary to disruption of the tumor.
If repeat D&C or hysterectomy are adopted instead of chemotherapy, it is important that patients are very closely monitored post operatively to ensure normalization of their hCG levels (as described above). If it fails to normalize, restaging scans and examinations should be performed, and referral for the appropriate chemotherapy regimen should be initiated without delay.
Postmolar GTN is a serious condition that usually can be cured with chemotherapy or, if appropriate, surgery. and refer to a gynecologic oncologist when criteria are met to ensure that overtreatment is avoided and essential therapy is ensured.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Lancet Oncol. 2007 Aug;8(8):715-24.
2. J Natl Compr Canc Netw. 2019 Nov 1;17(11):1374-91.
3. Gynecol Oncol. 2009 Mar;112(3):663-72.
4. World Health Organ Tech Rep Ser. 1983;692:7-81.
5. Obstet Gynecol. 2016;128(3):535-42.
Molar pregnancy is an uncommon but serious condition that affects young women of reproductive age. The diagnosis and management of molar pregnancy is familiar to most gynecologists. However, in the days and weeks following evacuation of molar pregnancy, clinicians face a critical time period in which they must be vigilant for the development of postmolar gestational trophoblastic neoplasia (GTN). If recognized early and treated appropriately, it almost always can be cured; however, errors or delays in the management of this condition can have catastrophic consequences for patients, including decreasing the likelihood of cure. Here we will review some of the steps and actions that can be taken immediately following the diagnosis of a molar pregnancy to expeditiously identify postmolar GTN and ensure patients are appropriately prepared for further consultation and intervention.
Postmolar GTN includes the diagnoses of invasive mole and choriocarcinoma that contain highly atypical trophoblasts with the capacity for local invasion and metastasis. Typically, the diagnosis is made clinically and not distinguished with histology. While molar pregnancies are a benign condition, invasive moles and choriocarcinoma are malignant conditions in which the molar tissue infiltrates the uterine myometrium, vasculature, and frequently is associated with hematogenous spread with distant metastases. It is a highly chemosensitive disease, and cure with chemotherapy typically is achieved with the ability to preserve fertility if desired even in advanced stage disease.1
After evacuation of a molar pregnancy, gynecologists should be on alert for the development of postmolar GTN if the following known risk factors are present: a history of a prior GTN diagnosis, complete mole on pathology (as opposed to partial mole), serum human chorionic gonadotropin (hCG) levels greater than 100,000 mIU/mL, age greater than 40 years, an enlarged uterus or large ovarian theca lutein cysts, and slow to normalize (more than 2 months) hCG. Symptoms for the development of postmolar GTN include persistent vaginal bleeding after evacuation, a persistently enlarged or enlarging uterine size, and adnexal masses. Ultimately, the diagnosis is made through plateaued or rising serum hCG assessments.2 (See graphic.)
Following the evacuation of a molar pregnancy, hCG levels should be drawn at the same laboratory every 1-2 weeks until normalization and then three consecutive normal values. Once this has been achieved, hCG levels should be tested once at 3 months and again at 6 months. During this 6 month period, patients should use reliable contraception, ideally, and through oral contraceptive pills that suppress the secretion of pituitary hCG if not contraindicated. Should a woman become pregnant during this 6-month surveillance, it becomes impossible to rule out occult postmolar GTN.
Typically after evacuation of a molar pregnancy, there is rapid fall in hCG levels, but this does not occur when the molar pregnancy has become invasive or is associated with choriocarcinoma. In these cases, after an initial drop in hCG levels, there is an observed rise or plateau in levels (as defined in the accompanying table), and this establishes the diagnosis of postmolar GTN. It is common for hCG to fall in fits and starts, rather than have a smooth, consistent diminution, and this can be worrying for gynecologists; however, provided there is a consistent reduction in values in accordance with the stated definitions, observation can continue.
Another source of confusion and concern is an HCG level that fails to completely normalize during observation, yet reaches a very low level. If this is observed, clinicians should consider the diagnosis of quiescent hCG, pituitary hCG, or phantom hCG.3 These can be difficult to distinguish from postmolar GTN, and consultation with a gynecologic oncologist with experience in the diagnosis and management of these rare tumors is helpful to determine if the persistent low levels in hCG require intervention.
Once a clinician has observed a plateau or rise in hCG levels, a gynecologic examination should be performed because the lower genital tract is a common site for metastatic postmolar GTN. If during this evaluation, a suspicious lesion is identified (typically a blue-black, slightly raised, hemorrhagic-appearing lesion), it should not be biopsied, but rather assumed to be a metastatic site. The vasculature of metastatic sites is extremely fragile, and biopsy or disruption can result in catastrophic hemorrhage, even from very small lesions.
In addition to physical examination, several diagnostic studies should be performed which may expedite the triage and management of the case. A pelvic ultrasound should evaluate the endometrial cavity for a new viable pregnancy, and residual molar tissue; sometimes, myometrial invasion consistent with an invasive mole can be appreciated. Chest x-ray or CT scan should be ordered to evaluate for pulmonary metastatic lesions. Additionally, CT scans of the abdomen and pelvis should be ordered, and if lung metastases are present, brain imaging with either MRI or CT scan also should be obtained. These imaging studies will provide the necessary information to stage the GTN (as metastatic or not).
Treatment for postmolar GTN is determined based on further prognostic categorization (“high risk” or “low risk”) in accordance with the WHO classification, which is derived using several prognostic clinical variables including age, antecedent pregnancy, interval from index pregnancy, pretreatment hCG, largest tumor size, sites and number of metastases, and response to previous chemotherapy.4 These assignments are necessary to determine whether single-agent or multiagent chemotherapy should be prescribed.
Laboratory studies are helpful to obtain at this time and include metabolic panels (which can ensure that renal and hepatic function are within normal limits in anticipation of future chemotherapy), and complete blood count ,which can establish viable bone marrow function prior to chemotherapy.
Once postmolar GTN has been diagnosed, it is most appropriate to refer the patient to a gynecologic oncologist with experience in the treatment of these relatively rare malignancies. At that point, the patient will be formally staged, and offered treatment based on these staging results.
Among women with low-risk, nonmetastatic GTN who desire future fertility it is appropriate to offer a repeat dilation and curettage (D&C) procedure rather than immediately proceeding with chemotherapy. Approximately two-thirds of women with low risk disease can avoid chemotherapy with repeat curettage.5 Risk factors for needing chemotherapy after repeat D&C include the presence of trophoblastic disease in the pathology specimen and urinary hCG levels greater than 1,500 mIU/mL at the time of curettage. In my experience, many women appreciate this option to potentially avoid toxic chemotherapy.
For women with low-risk, nonmetastatic postmolar GTN who do not desire future fertility, and hope to avoid chemotherapy, hysterectomy also is a reasonable first option. This can be performed via either minimally invasive, laparotomy, or vaginal route. If performing a minimally invasive procedure in the setting of GTN, there should be caution or avoidance of use of a uterine manipulator because the uterine wall typically is soft and prone to perforation, and bleeding can be significant secondary to disruption of the tumor.
If repeat D&C or hysterectomy are adopted instead of chemotherapy, it is important that patients are very closely monitored post operatively to ensure normalization of their hCG levels (as described above). If it fails to normalize, restaging scans and examinations should be performed, and referral for the appropriate chemotherapy regimen should be initiated without delay.
Postmolar GTN is a serious condition that usually can be cured with chemotherapy or, if appropriate, surgery. and refer to a gynecologic oncologist when criteria are met to ensure that overtreatment is avoided and essential therapy is ensured.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Lancet Oncol. 2007 Aug;8(8):715-24.
2. J Natl Compr Canc Netw. 2019 Nov 1;17(11):1374-91.
3. Gynecol Oncol. 2009 Mar;112(3):663-72.
4. World Health Organ Tech Rep Ser. 1983;692:7-81.
5. Obstet Gynecol. 2016;128(3):535-42.
Molar pregnancy is an uncommon but serious condition that affects young women of reproductive age. The diagnosis and management of molar pregnancy is familiar to most gynecologists. However, in the days and weeks following evacuation of molar pregnancy, clinicians face a critical time period in which they must be vigilant for the development of postmolar gestational trophoblastic neoplasia (GTN). If recognized early and treated appropriately, it almost always can be cured; however, errors or delays in the management of this condition can have catastrophic consequences for patients, including decreasing the likelihood of cure. Here we will review some of the steps and actions that can be taken immediately following the diagnosis of a molar pregnancy to expeditiously identify postmolar GTN and ensure patients are appropriately prepared for further consultation and intervention.
Postmolar GTN includes the diagnoses of invasive mole and choriocarcinoma that contain highly atypical trophoblasts with the capacity for local invasion and metastasis. Typically, the diagnosis is made clinically and not distinguished with histology. While molar pregnancies are a benign condition, invasive moles and choriocarcinoma are malignant conditions in which the molar tissue infiltrates the uterine myometrium, vasculature, and frequently is associated with hematogenous spread with distant metastases. It is a highly chemosensitive disease, and cure with chemotherapy typically is achieved with the ability to preserve fertility if desired even in advanced stage disease.1
After evacuation of a molar pregnancy, gynecologists should be on alert for the development of postmolar GTN if the following known risk factors are present: a history of a prior GTN diagnosis, complete mole on pathology (as opposed to partial mole), serum human chorionic gonadotropin (hCG) levels greater than 100,000 mIU/mL, age greater than 40 years, an enlarged uterus or large ovarian theca lutein cysts, and slow to normalize (more than 2 months) hCG. Symptoms for the development of postmolar GTN include persistent vaginal bleeding after evacuation, a persistently enlarged or enlarging uterine size, and adnexal masses. Ultimately, the diagnosis is made through plateaued or rising serum hCG assessments.2 (See graphic.)
Following the evacuation of a molar pregnancy, hCG levels should be drawn at the same laboratory every 1-2 weeks until normalization and then three consecutive normal values. Once this has been achieved, hCG levels should be tested once at 3 months and again at 6 months. During this 6 month period, patients should use reliable contraception, ideally, and through oral contraceptive pills that suppress the secretion of pituitary hCG if not contraindicated. Should a woman become pregnant during this 6-month surveillance, it becomes impossible to rule out occult postmolar GTN.
Typically after evacuation of a molar pregnancy, there is rapid fall in hCG levels, but this does not occur when the molar pregnancy has become invasive or is associated with choriocarcinoma. In these cases, after an initial drop in hCG levels, there is an observed rise or plateau in levels (as defined in the accompanying table), and this establishes the diagnosis of postmolar GTN. It is common for hCG to fall in fits and starts, rather than have a smooth, consistent diminution, and this can be worrying for gynecologists; however, provided there is a consistent reduction in values in accordance with the stated definitions, observation can continue.
Another source of confusion and concern is an HCG level that fails to completely normalize during observation, yet reaches a very low level. If this is observed, clinicians should consider the diagnosis of quiescent hCG, pituitary hCG, or phantom hCG.3 These can be difficult to distinguish from postmolar GTN, and consultation with a gynecologic oncologist with experience in the diagnosis and management of these rare tumors is helpful to determine if the persistent low levels in hCG require intervention.
Once a clinician has observed a plateau or rise in hCG levels, a gynecologic examination should be performed because the lower genital tract is a common site for metastatic postmolar GTN. If during this evaluation, a suspicious lesion is identified (typically a blue-black, slightly raised, hemorrhagic-appearing lesion), it should not be biopsied, but rather assumed to be a metastatic site. The vasculature of metastatic sites is extremely fragile, and biopsy or disruption can result in catastrophic hemorrhage, even from very small lesions.
In addition to physical examination, several diagnostic studies should be performed which may expedite the triage and management of the case. A pelvic ultrasound should evaluate the endometrial cavity for a new viable pregnancy, and residual molar tissue; sometimes, myometrial invasion consistent with an invasive mole can be appreciated. Chest x-ray or CT scan should be ordered to evaluate for pulmonary metastatic lesions. Additionally, CT scans of the abdomen and pelvis should be ordered, and if lung metastases are present, brain imaging with either MRI or CT scan also should be obtained. These imaging studies will provide the necessary information to stage the GTN (as metastatic or not).
Treatment for postmolar GTN is determined based on further prognostic categorization (“high risk” or “low risk”) in accordance with the WHO classification, which is derived using several prognostic clinical variables including age, antecedent pregnancy, interval from index pregnancy, pretreatment hCG, largest tumor size, sites and number of metastases, and response to previous chemotherapy.4 These assignments are necessary to determine whether single-agent or multiagent chemotherapy should be prescribed.
Laboratory studies are helpful to obtain at this time and include metabolic panels (which can ensure that renal and hepatic function are within normal limits in anticipation of future chemotherapy), and complete blood count ,which can establish viable bone marrow function prior to chemotherapy.
Once postmolar GTN has been diagnosed, it is most appropriate to refer the patient to a gynecologic oncologist with experience in the treatment of these relatively rare malignancies. At that point, the patient will be formally staged, and offered treatment based on these staging results.
Among women with low-risk, nonmetastatic GTN who desire future fertility it is appropriate to offer a repeat dilation and curettage (D&C) procedure rather than immediately proceeding with chemotherapy. Approximately two-thirds of women with low risk disease can avoid chemotherapy with repeat curettage.5 Risk factors for needing chemotherapy after repeat D&C include the presence of trophoblastic disease in the pathology specimen and urinary hCG levels greater than 1,500 mIU/mL at the time of curettage. In my experience, many women appreciate this option to potentially avoid toxic chemotherapy.
For women with low-risk, nonmetastatic postmolar GTN who do not desire future fertility, and hope to avoid chemotherapy, hysterectomy also is a reasonable first option. This can be performed via either minimally invasive, laparotomy, or vaginal route. If performing a minimally invasive procedure in the setting of GTN, there should be caution or avoidance of use of a uterine manipulator because the uterine wall typically is soft and prone to perforation, and bleeding can be significant secondary to disruption of the tumor.
If repeat D&C or hysterectomy are adopted instead of chemotherapy, it is important that patients are very closely monitored post operatively to ensure normalization of their hCG levels (as described above). If it fails to normalize, restaging scans and examinations should be performed, and referral for the appropriate chemotherapy regimen should be initiated without delay.
Postmolar GTN is a serious condition that usually can be cured with chemotherapy or, if appropriate, surgery. and refer to a gynecologic oncologist when criteria are met to ensure that overtreatment is avoided and essential therapy is ensured.
Dr. Rossi is assistant professor in the division of gynecologic oncology at the University of North Carolina at Chapel Hill. She said she had no relevant financial disclosures. Email her at [email protected].
References
1. Lancet Oncol. 2007 Aug;8(8):715-24.
2. J Natl Compr Canc Netw. 2019 Nov 1;17(11):1374-91.
3. Gynecol Oncol. 2009 Mar;112(3):663-72.
4. World Health Organ Tech Rep Ser. 1983;692:7-81.
5. Obstet Gynecol. 2016;128(3):535-42.
Quick Byte: EHRs and clinician wellness
Concerns abound regarding the workload created by electronic health records (EHRs), but little attention has been paid to the relationship between physicians’ well-being and the volume and sources of in-basket messages they receive.
According to a recent paper in Health Affairs, “in a survey, 36 percent of the physicians reported burnout symptoms, and 29 percent intended to reduce their clinical work time in the upcoming year. Receiving more than the average number of system-generated in-basket messages was associated with 40 percent higher probability of burnout and 38 percent higher probability of intending to reduce clinical work time.”
Reference
1. Tai-Seale M, Dillon EC, Yang Y, et al. Physicians’ Well-Being Linked To In-Basket Messages Generated By Algorithms In Electronic Health Records. Health Aff. 2019 Jul. doi: 10.1377/hlthaff.2018.05509.
Concerns abound regarding the workload created by electronic health records (EHRs), but little attention has been paid to the relationship between physicians’ well-being and the volume and sources of in-basket messages they receive.
According to a recent paper in Health Affairs, “in a survey, 36 percent of the physicians reported burnout symptoms, and 29 percent intended to reduce their clinical work time in the upcoming year. Receiving more than the average number of system-generated in-basket messages was associated with 40 percent higher probability of burnout and 38 percent higher probability of intending to reduce clinical work time.”
Reference
1. Tai-Seale M, Dillon EC, Yang Y, et al. Physicians’ Well-Being Linked To In-Basket Messages Generated By Algorithms In Electronic Health Records. Health Aff. 2019 Jul. doi: 10.1377/hlthaff.2018.05509.
Concerns abound regarding the workload created by electronic health records (EHRs), but little attention has been paid to the relationship between physicians’ well-being and the volume and sources of in-basket messages they receive.
According to a recent paper in Health Affairs, “in a survey, 36 percent of the physicians reported burnout symptoms, and 29 percent intended to reduce their clinical work time in the upcoming year. Receiving more than the average number of system-generated in-basket messages was associated with 40 percent higher probability of burnout and 38 percent higher probability of intending to reduce clinical work time.”
Reference
1. Tai-Seale M, Dillon EC, Yang Y, et al. Physicians’ Well-Being Linked To In-Basket Messages Generated By Algorithms In Electronic Health Records. Health Aff. 2019 Jul. doi: 10.1377/hlthaff.2018.05509.
Journal editors seek more complete disclosure from authors
A group of leading medical journal editors is seeking to improve the completeness and transparency of financial disclosure reporting with a proposed new disclosure form that puts more onus on readers to decide whether relationships and activities should influence how they view published papers.
The proposed changes are described in an editorial published simultaneously today in the Annals of Internal Medicine, British Medical Journal, Journal of the American Medical Association, The Lancet, New England Journal of Medicine, and several other journals whose editors are members of the International Committee of Medical Journal Editors (ICMJE).
“While no approach to disclosure will be perfect or foolproof, we hope the changes we propose will help promote transparency and trust,” the editorial stated (Ann Intern Med. 2020 Jan 27. doi: 10.7326/M19-3933).
The ICMJE adopted its currently used electronic form – the “ICMJE Form for the Disclosure of Potential Conflicts of Interest” – 10 years ago in an effort to create some uniformity amidst a patchwork of differing disclosure requirements for authors.
It’s not known how many journals outside of the ICMJE’s member journals routinely use the disclosure form, but the organization’s website houses an extensive list of journals whose editors or publishers have requested to be listed as following the ICMJE’s recommendations for editing, reporting, and publishing, including those concerning disclosures. The ICMJE does not “certify” journals. The full set of recommendations was updated in December 2019.
Most authors are committed to transparent reporting, but “opinions differ over which relationships or activities to report,” the editorial stated.
An author might choose to omit an item that others deem important because of a difference in opinion regarding “relevance,” confusion over definitions, or a simple oversight. Some authors may be “concerned that readers will interpret the listing of any item as a ‘potential conflict of interest’ as indicative of problematic influence and wrongdoing,” the editorial stated.
The revised form, like the current one, asks authors to disclose relationships and activities that are directly related to the reported work, as well as those that are topically related (within the broadly defined field addressed in the work). But unlike the current form, the new version provides a checklist of relationships and activities and asks authors to check ‘yes’ or ‘no’ for each one (and to name them when the answer is ‘yes’).
Items in the checklist include grants, payments/honoraria for lectures, patents issued or planned, stock/stock options, and leadership or fiduciary roles in committees, boards, or societies.
The proposed new form makes no mention of “potential conflicts of interest” or “relevancy,” per say. Authors aren’t asked to determine what might be interpreted as a potential conflict of interest, but instead are asked for a “complete listing” of what readers may find “pertinent” to their work.
“We’re trying to move away from calling everything a [potential] ‘conflict,’ ” Darren B. Taichman, MD, PhD, secretary of ICMJE and executive editor of the Annals of Internal Medicine, said in an interview. “We want to remove for authors the concern or stigma, if you will, that anything listed on a form implies that there is something wrong, because that’s just not true. … We want readers to decide what relationships are important as they interpret the work.”
Dr. Taichman said in the interview that the ICMJE’s updating of the form was more a function of “good housekeeping” and continuous appreciation of disclosure as an important issue, rather than any one specific issue, such as concern over a “relevancy” approach to disclosures.
The ICMJE is seeking feedback about its proposed form, which is available with a link for providing comments, at www.icmje.org.
Broader national efforts
Editors and others have been increasingly moving, however, toward asking for more complete disclosures where authors aren’t asked to judge “relevancy” and where readers can make decisions on their own. The American Society of Clinical Oncology, which produces the Journal of Clinical Oncology (JCO) as well as practice guidelines and continuing medical education programs, moved about 5 years ago to a system of general disclosure that asks physicians and others to disclose all financial interests and industry relationships, with no qualifiers.
Earlier in January 2020, the Accreditation Council for Continuing Medical Education issued proposed revisions to its Standards for Integrity and Independence in Accredited Continuing Education. These revisions, which are open for comment, require CME providers to collect disclosure information about all financial relationships of speakers and presenters. It’s up to the CME provider to then determine which relationships are relevant, according to the proposed document.
More change is on the way, as disclosure issues are being deliberated nationally in the wake of a highly publicized disclosure failure at Memorial Sloan Kettering Cancer Center in 2018. Chief medical officer José Baselga, MD, PhD, failed to report millions of dollars of industry payments and ownership interests in journal articles he wrote or cowrote over several years.
In February 2019, leaders from journals, academia, medical societies, and other institutions gathered in Washington for a closed-door meeting to hash out various disclosure related issues.
Hosted by the Association of American Medical Colleges and cosponsored by Memorial Sloan Kettering Cancer Center, ASCO, JAMA, and the Council of Medical Specialty Societies, the meeting led to a series of working groups that are creating additional recommendations “due out soon in 2020,” Heather Pierce, senior director of science policy and regulatory counsel for the AAMC, said in an interview.
Among the questions being discussed: What disclosures should be verified and who should do so? How can disclosures be made more complete and easier for researchers? And, “most importantly,” said Ms. Pierce, how can policy requirements across each of these sectors be aligned so that there’s more coordination and oversight – and with it, public trust?
Some critics of current disclosure policies have called for more reporting of compensation amounts, and Ms. Pierce said that this has been part of cross-sector discussions.
The ICMJE’s proposed form invites, but does not require, authors to indicate what payments were made to them or their institutions. “Part of this is due to the fact that it’s hard to define, let alone agree on, what’s an important amount,” Dr. Taichman said.
A push for registries
The ICMJE is also aiming to make the disclosure process more efficient for authors – and to eliminate inconsistent and incomplete disclosures – by accepting disclosures from web-based repositories, according to the editorial. Repositories allow authors to maintain an inventory of their relationships and activities and then create electronic disclosures that are tailored to the requirements of the ICMJE, medical societies, and other entities.
The AAMC-run repository, called Convey, is consistent with ICMJE reporting requirements and other criteria (e.g., there are no fees for individuals to enter, store, or export their data), but the development of other repositories may be helpful “for meeting regional, linguistic, and regulatory needs” of authors across the world, the editorial stated.
The Annals of Internal Medicine and the New England Journal of Medicine are both currently collecting disclosures through Convey. The platform was born from discussions that followed a 2009 Institute of Medicine report on conflicts of interest.
Signers of the ICMJE editorial include representatives of the National Library of Medicine and the World Association of Medical Editors, in addition to editors in chief and other leaders of the ICMJE member journals.
A group of leading medical journal editors is seeking to improve the completeness and transparency of financial disclosure reporting with a proposed new disclosure form that puts more onus on readers to decide whether relationships and activities should influence how they view published papers.
The proposed changes are described in an editorial published simultaneously today in the Annals of Internal Medicine, British Medical Journal, Journal of the American Medical Association, The Lancet, New England Journal of Medicine, and several other journals whose editors are members of the International Committee of Medical Journal Editors (ICMJE).
“While no approach to disclosure will be perfect or foolproof, we hope the changes we propose will help promote transparency and trust,” the editorial stated (Ann Intern Med. 2020 Jan 27. doi: 10.7326/M19-3933).
The ICMJE adopted its currently used electronic form – the “ICMJE Form for the Disclosure of Potential Conflicts of Interest” – 10 years ago in an effort to create some uniformity amidst a patchwork of differing disclosure requirements for authors.
It’s not known how many journals outside of the ICMJE’s member journals routinely use the disclosure form, but the organization’s website houses an extensive list of journals whose editors or publishers have requested to be listed as following the ICMJE’s recommendations for editing, reporting, and publishing, including those concerning disclosures. The ICMJE does not “certify” journals. The full set of recommendations was updated in December 2019.
Most authors are committed to transparent reporting, but “opinions differ over which relationships or activities to report,” the editorial stated.
An author might choose to omit an item that others deem important because of a difference in opinion regarding “relevance,” confusion over definitions, or a simple oversight. Some authors may be “concerned that readers will interpret the listing of any item as a ‘potential conflict of interest’ as indicative of problematic influence and wrongdoing,” the editorial stated.
The revised form, like the current one, asks authors to disclose relationships and activities that are directly related to the reported work, as well as those that are topically related (within the broadly defined field addressed in the work). But unlike the current form, the new version provides a checklist of relationships and activities and asks authors to check ‘yes’ or ‘no’ for each one (and to name them when the answer is ‘yes’).
Items in the checklist include grants, payments/honoraria for lectures, patents issued or planned, stock/stock options, and leadership or fiduciary roles in committees, boards, or societies.
The proposed new form makes no mention of “potential conflicts of interest” or “relevancy,” per say. Authors aren’t asked to determine what might be interpreted as a potential conflict of interest, but instead are asked for a “complete listing” of what readers may find “pertinent” to their work.
“We’re trying to move away from calling everything a [potential] ‘conflict,’ ” Darren B. Taichman, MD, PhD, secretary of ICMJE and executive editor of the Annals of Internal Medicine, said in an interview. “We want to remove for authors the concern or stigma, if you will, that anything listed on a form implies that there is something wrong, because that’s just not true. … We want readers to decide what relationships are important as they interpret the work.”
Dr. Taichman said in the interview that the ICMJE’s updating of the form was more a function of “good housekeeping” and continuous appreciation of disclosure as an important issue, rather than any one specific issue, such as concern over a “relevancy” approach to disclosures.
The ICMJE is seeking feedback about its proposed form, which is available with a link for providing comments, at www.icmje.org.
Broader national efforts
Editors and others have been increasingly moving, however, toward asking for more complete disclosures where authors aren’t asked to judge “relevancy” and where readers can make decisions on their own. The American Society of Clinical Oncology, which produces the Journal of Clinical Oncology (JCO) as well as practice guidelines and continuing medical education programs, moved about 5 years ago to a system of general disclosure that asks physicians and others to disclose all financial interests and industry relationships, with no qualifiers.
Earlier in January 2020, the Accreditation Council for Continuing Medical Education issued proposed revisions to its Standards for Integrity and Independence in Accredited Continuing Education. These revisions, which are open for comment, require CME providers to collect disclosure information about all financial relationships of speakers and presenters. It’s up to the CME provider to then determine which relationships are relevant, according to the proposed document.
More change is on the way, as disclosure issues are being deliberated nationally in the wake of a highly publicized disclosure failure at Memorial Sloan Kettering Cancer Center in 2018. Chief medical officer José Baselga, MD, PhD, failed to report millions of dollars of industry payments and ownership interests in journal articles he wrote or cowrote over several years.
In February 2019, leaders from journals, academia, medical societies, and other institutions gathered in Washington for a closed-door meeting to hash out various disclosure related issues.
Hosted by the Association of American Medical Colleges and cosponsored by Memorial Sloan Kettering Cancer Center, ASCO, JAMA, and the Council of Medical Specialty Societies, the meeting led to a series of working groups that are creating additional recommendations “due out soon in 2020,” Heather Pierce, senior director of science policy and regulatory counsel for the AAMC, said in an interview.
Among the questions being discussed: What disclosures should be verified and who should do so? How can disclosures be made more complete and easier for researchers? And, “most importantly,” said Ms. Pierce, how can policy requirements across each of these sectors be aligned so that there’s more coordination and oversight – and with it, public trust?
Some critics of current disclosure policies have called for more reporting of compensation amounts, and Ms. Pierce said that this has been part of cross-sector discussions.
The ICMJE’s proposed form invites, but does not require, authors to indicate what payments were made to them or their institutions. “Part of this is due to the fact that it’s hard to define, let alone agree on, what’s an important amount,” Dr. Taichman said.
A push for registries
The ICMJE is also aiming to make the disclosure process more efficient for authors – and to eliminate inconsistent and incomplete disclosures – by accepting disclosures from web-based repositories, according to the editorial. Repositories allow authors to maintain an inventory of their relationships and activities and then create electronic disclosures that are tailored to the requirements of the ICMJE, medical societies, and other entities.
The AAMC-run repository, called Convey, is consistent with ICMJE reporting requirements and other criteria (e.g., there are no fees for individuals to enter, store, or export their data), but the development of other repositories may be helpful “for meeting regional, linguistic, and regulatory needs” of authors across the world, the editorial stated.
The Annals of Internal Medicine and the New England Journal of Medicine are both currently collecting disclosures through Convey. The platform was born from discussions that followed a 2009 Institute of Medicine report on conflicts of interest.
Signers of the ICMJE editorial include representatives of the National Library of Medicine and the World Association of Medical Editors, in addition to editors in chief and other leaders of the ICMJE member journals.
A group of leading medical journal editors is seeking to improve the completeness and transparency of financial disclosure reporting with a proposed new disclosure form that puts more onus on readers to decide whether relationships and activities should influence how they view published papers.
The proposed changes are described in an editorial published simultaneously today in the Annals of Internal Medicine, British Medical Journal, Journal of the American Medical Association, The Lancet, New England Journal of Medicine, and several other journals whose editors are members of the International Committee of Medical Journal Editors (ICMJE).
“While no approach to disclosure will be perfect or foolproof, we hope the changes we propose will help promote transparency and trust,” the editorial stated (Ann Intern Med. 2020 Jan 27. doi: 10.7326/M19-3933).
The ICMJE adopted its currently used electronic form – the “ICMJE Form for the Disclosure of Potential Conflicts of Interest” – 10 years ago in an effort to create some uniformity amidst a patchwork of differing disclosure requirements for authors.
It’s not known how many journals outside of the ICMJE’s member journals routinely use the disclosure form, but the organization’s website houses an extensive list of journals whose editors or publishers have requested to be listed as following the ICMJE’s recommendations for editing, reporting, and publishing, including those concerning disclosures. The ICMJE does not “certify” journals. The full set of recommendations was updated in December 2019.
Most authors are committed to transparent reporting, but “opinions differ over which relationships or activities to report,” the editorial stated.
An author might choose to omit an item that others deem important because of a difference in opinion regarding “relevance,” confusion over definitions, or a simple oversight. Some authors may be “concerned that readers will interpret the listing of any item as a ‘potential conflict of interest’ as indicative of problematic influence and wrongdoing,” the editorial stated.
The revised form, like the current one, asks authors to disclose relationships and activities that are directly related to the reported work, as well as those that are topically related (within the broadly defined field addressed in the work). But unlike the current form, the new version provides a checklist of relationships and activities and asks authors to check ‘yes’ or ‘no’ for each one (and to name them when the answer is ‘yes’).
Items in the checklist include grants, payments/honoraria for lectures, patents issued or planned, stock/stock options, and leadership or fiduciary roles in committees, boards, or societies.
The proposed new form makes no mention of “potential conflicts of interest” or “relevancy,” per say. Authors aren’t asked to determine what might be interpreted as a potential conflict of interest, but instead are asked for a “complete listing” of what readers may find “pertinent” to their work.
“We’re trying to move away from calling everything a [potential] ‘conflict,’ ” Darren B. Taichman, MD, PhD, secretary of ICMJE and executive editor of the Annals of Internal Medicine, said in an interview. “We want to remove for authors the concern or stigma, if you will, that anything listed on a form implies that there is something wrong, because that’s just not true. … We want readers to decide what relationships are important as they interpret the work.”
Dr. Taichman said in the interview that the ICMJE’s updating of the form was more a function of “good housekeeping” and continuous appreciation of disclosure as an important issue, rather than any one specific issue, such as concern over a “relevancy” approach to disclosures.
The ICMJE is seeking feedback about its proposed form, which is available with a link for providing comments, at www.icmje.org.
Broader national efforts
Editors and others have been increasingly moving, however, toward asking for more complete disclosures where authors aren’t asked to judge “relevancy” and where readers can make decisions on their own. The American Society of Clinical Oncology, which produces the Journal of Clinical Oncology (JCO) as well as practice guidelines and continuing medical education programs, moved about 5 years ago to a system of general disclosure that asks physicians and others to disclose all financial interests and industry relationships, with no qualifiers.
Earlier in January 2020, the Accreditation Council for Continuing Medical Education issued proposed revisions to its Standards for Integrity and Independence in Accredited Continuing Education. These revisions, which are open for comment, require CME providers to collect disclosure information about all financial relationships of speakers and presenters. It’s up to the CME provider to then determine which relationships are relevant, according to the proposed document.
More change is on the way, as disclosure issues are being deliberated nationally in the wake of a highly publicized disclosure failure at Memorial Sloan Kettering Cancer Center in 2018. Chief medical officer José Baselga, MD, PhD, failed to report millions of dollars of industry payments and ownership interests in journal articles he wrote or cowrote over several years.
In February 2019, leaders from journals, academia, medical societies, and other institutions gathered in Washington for a closed-door meeting to hash out various disclosure related issues.
Hosted by the Association of American Medical Colleges and cosponsored by Memorial Sloan Kettering Cancer Center, ASCO, JAMA, and the Council of Medical Specialty Societies, the meeting led to a series of working groups that are creating additional recommendations “due out soon in 2020,” Heather Pierce, senior director of science policy and regulatory counsel for the AAMC, said in an interview.
Among the questions being discussed: What disclosures should be verified and who should do so? How can disclosures be made more complete and easier for researchers? And, “most importantly,” said Ms. Pierce, how can policy requirements across each of these sectors be aligned so that there’s more coordination and oversight – and with it, public trust?
Some critics of current disclosure policies have called for more reporting of compensation amounts, and Ms. Pierce said that this has been part of cross-sector discussions.
The ICMJE’s proposed form invites, but does not require, authors to indicate what payments were made to them or their institutions. “Part of this is due to the fact that it’s hard to define, let alone agree on, what’s an important amount,” Dr. Taichman said.
A push for registries
The ICMJE is also aiming to make the disclosure process more efficient for authors – and to eliminate inconsistent and incomplete disclosures – by accepting disclosures from web-based repositories, according to the editorial. Repositories allow authors to maintain an inventory of their relationships and activities and then create electronic disclosures that are tailored to the requirements of the ICMJE, medical societies, and other entities.
The AAMC-run repository, called Convey, is consistent with ICMJE reporting requirements and other criteria (e.g., there are no fees for individuals to enter, store, or export their data), but the development of other repositories may be helpful “for meeting regional, linguistic, and regulatory needs” of authors across the world, the editorial stated.
The Annals of Internal Medicine and the New England Journal of Medicine are both currently collecting disclosures through Convey. The platform was born from discussions that followed a 2009 Institute of Medicine report on conflicts of interest.
Signers of the ICMJE editorial include representatives of the National Library of Medicine and the World Association of Medical Editors, in addition to editors in chief and other leaders of the ICMJE member journals.
FROM ANNALS OF INTERNAL MEDICINE
Wuhan coronavirus cluster suggests human-to-human spread
A Chinese man became ill from a novel coronavirus (2019-nCoV) 4 days after arriving in Vietnam to visit his 27-year-old son. Three days later the healthy young man was also stricken, according to a report published online Jan. 28 in the New England Journal of Medicine.
“This family cluster of 2019-nCoV infection that occurred outside China arouses concern regarding human-to-human transmission,” the authors wrote.
The father, age 65 years and with multiple comorbidities including hypertension, type 2 diabetes, coronary heart disease with stent placement, and lung cancer, flew to Hanoi with his wife on January 13; they traveled from the Wuchang district in Wuhan, China, where outbreaks of 2019-nCoV have been occurring.
On Jan. 17, the older man and his wife met their adult son in Ho Chi Minh City, Vietnam, and shared a hotel room with him for 3 days. The father developed a fever that same day and the son developed a dry cough, fever, diarrhea, and vomiting on Jan. 20. Both men went to a hospital ED on Jan. 22.
The authors say the timing of the son’s symptoms suggests the incubation period may have been 3 days or fewer.
Upon admission to the hospital, the father reported that he had not visited a “wet market” where live and dead animals are sold while he was in Wuhan. Throat swabs were positive for 2019-nCoV on real-time reverse-transcription–polymerase-chain-reaction assays.
The man was placed in isolation and “treated empirically with antiviral agents, broad-spectrum antibiotics, and supportive therapies,” wrote Lan T. Phan, PhD, from the Pasteur Institute Ho Chi Minh City and coauthors.
On admission, chest radiographs revealed an infiltrate in the upper lobe of his left lung; he developed worsening dyspnea with hypoxemia on Jan. 25 and required supplemental oxygen at 5 L/min by nasal cannula. Chest radiographs showed a progressive infiltrate and consolidation. His fever resolved on that day and he has progressively improved.
The man’s son had a fever of 39° C (102.2° F) when the two men arrived at the hospital on Jan. 22; hospital staff isolated the son, and chest radiographs and other laboratory tests were normal with the exception of an increased C-reactive protein level.
The son’s throat swab was positive for 2019-nCoV and he is believed to have been exposed from his father; however, the strains have not been ascertained.
“This family had traveled to four cities across Vietnam using various forms of transportation, including planes, trains, and taxis,” the authors wrote. A total of 28 close contacts were identified, none of whom have developed respiratory symptoms. The older man’s wife has been healthy as well.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A Chinese man became ill from a novel coronavirus (2019-nCoV) 4 days after arriving in Vietnam to visit his 27-year-old son. Three days later the healthy young man was also stricken, according to a report published online Jan. 28 in the New England Journal of Medicine.
“This family cluster of 2019-nCoV infection that occurred outside China arouses concern regarding human-to-human transmission,” the authors wrote.
The father, age 65 years and with multiple comorbidities including hypertension, type 2 diabetes, coronary heart disease with stent placement, and lung cancer, flew to Hanoi with his wife on January 13; they traveled from the Wuchang district in Wuhan, China, where outbreaks of 2019-nCoV have been occurring.
On Jan. 17, the older man and his wife met their adult son in Ho Chi Minh City, Vietnam, and shared a hotel room with him for 3 days. The father developed a fever that same day and the son developed a dry cough, fever, diarrhea, and vomiting on Jan. 20. Both men went to a hospital ED on Jan. 22.
The authors say the timing of the son’s symptoms suggests the incubation period may have been 3 days or fewer.
Upon admission to the hospital, the father reported that he had not visited a “wet market” where live and dead animals are sold while he was in Wuhan. Throat swabs were positive for 2019-nCoV on real-time reverse-transcription–polymerase-chain-reaction assays.
The man was placed in isolation and “treated empirically with antiviral agents, broad-spectrum antibiotics, and supportive therapies,” wrote Lan T. Phan, PhD, from the Pasteur Institute Ho Chi Minh City and coauthors.
On admission, chest radiographs revealed an infiltrate in the upper lobe of his left lung; he developed worsening dyspnea with hypoxemia on Jan. 25 and required supplemental oxygen at 5 L/min by nasal cannula. Chest radiographs showed a progressive infiltrate and consolidation. His fever resolved on that day and he has progressively improved.
The man’s son had a fever of 39° C (102.2° F) when the two men arrived at the hospital on Jan. 22; hospital staff isolated the son, and chest radiographs and other laboratory tests were normal with the exception of an increased C-reactive protein level.
The son’s throat swab was positive for 2019-nCoV and he is believed to have been exposed from his father; however, the strains have not been ascertained.
“This family had traveled to four cities across Vietnam using various forms of transportation, including planes, trains, and taxis,” the authors wrote. A total of 28 close contacts were identified, none of whom have developed respiratory symptoms. The older man’s wife has been healthy as well.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
A Chinese man became ill from a novel coronavirus (2019-nCoV) 4 days after arriving in Vietnam to visit his 27-year-old son. Three days later the healthy young man was also stricken, according to a report published online Jan. 28 in the New England Journal of Medicine.
“This family cluster of 2019-nCoV infection that occurred outside China arouses concern regarding human-to-human transmission,” the authors wrote.
The father, age 65 years and with multiple comorbidities including hypertension, type 2 diabetes, coronary heart disease with stent placement, and lung cancer, flew to Hanoi with his wife on January 13; they traveled from the Wuchang district in Wuhan, China, where outbreaks of 2019-nCoV have been occurring.
On Jan. 17, the older man and his wife met their adult son in Ho Chi Minh City, Vietnam, and shared a hotel room with him for 3 days. The father developed a fever that same day and the son developed a dry cough, fever, diarrhea, and vomiting on Jan. 20. Both men went to a hospital ED on Jan. 22.
The authors say the timing of the son’s symptoms suggests the incubation period may have been 3 days or fewer.
Upon admission to the hospital, the father reported that he had not visited a “wet market” where live and dead animals are sold while he was in Wuhan. Throat swabs were positive for 2019-nCoV on real-time reverse-transcription–polymerase-chain-reaction assays.
The man was placed in isolation and “treated empirically with antiviral agents, broad-spectrum antibiotics, and supportive therapies,” wrote Lan T. Phan, PhD, from the Pasteur Institute Ho Chi Minh City and coauthors.
On admission, chest radiographs revealed an infiltrate in the upper lobe of his left lung; he developed worsening dyspnea with hypoxemia on Jan. 25 and required supplemental oxygen at 5 L/min by nasal cannula. Chest radiographs showed a progressive infiltrate and consolidation. His fever resolved on that day and he has progressively improved.
The man’s son had a fever of 39° C (102.2° F) when the two men arrived at the hospital on Jan. 22; hospital staff isolated the son, and chest radiographs and other laboratory tests were normal with the exception of an increased C-reactive protein level.
The son’s throat swab was positive for 2019-nCoV and he is believed to have been exposed from his father; however, the strains have not been ascertained.
“This family had traveled to four cities across Vietnam using various forms of transportation, including planes, trains, and taxis,” the authors wrote. A total of 28 close contacts were identified, none of whom have developed respiratory symptoms. The older man’s wife has been healthy as well.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.