User login
Data on potential risks of COVID-19 in psoriasis patients limited, but reassuring
The available according to a summary of published studies and expert opinions summarized at the annual Coastal Dermatology Symposium, held virtually.
For patients with psoriasis concerned about their outcome if infected with COVID-19, “there is no evidence to support stopping biologics or systemic agents, so I am asking my patients to continue,” Kristina C. Duffin, MD, professor and chair of dermatology at the University of Utah, Salt Lake City, said at the meeting.
The National Psoriasis Foundation, which created a COVID-19 task force and maintains a COVID-19 Resource Center on its website, has provided similar advice. Many statements are phrased cautiously and clinicians are encouraged to practice shared decision-making, but the NPF guidance supports continuing effective therapy – or, in newly diagnosed patients, starting effective therapy – among those who are not infected with SARS-CoV2.
Patients with a new diagnosis of psoriasis “should be aware that untreated psoriatic disease is associated with serious impact on physical and emotional health, and in the case of psoriatic arthritis, can lead to permanent joint damage and disability,” according to the NPF guidance.
Overall, the “existing data generally suggest” that most treatments for psoriasis and psoriatic arthritis “do not meaningfully alter the risks of contracting SARS-CoV2 or having a worse course of COVID-19 illness,” the current guidance states. Yet, because of limited data this “is not known with certainty.”
Chronic systemic steroids are an exception. In a review of recently published studies evaluating whether psoriasis or its therapies increase risk of adverse outcomes in patients with COVID-19 infection, Dr. Duffin pointed to several that associated systemic steroids with hospitalization or other markers of severe disease.
The NPF guidance also recommends avoiding chronic systemic steroids in patients with psoriasis during the current COVID-19 era “if possible.” In patients with psoriatic arthritis who require systemic steroids, the guidance recommends “the lowest dose necessary to achieve the desired therapeutic effect.”
This is not necessarily true in patients with psoriasis and COVID-19 infection. Based on the potential for systemic steroids to improve outcomes in hospitalized COVID-19 patients requiring oxygen, steroids “should not be withheld” even when the justification is concern about the potential risk of flares with withdrawal, according to the NPF guidance statement.
The NPF guidance specifically cautions against use of hydroxychloroquine or chloroquine for prevention or treatment of COVID-19. In addition to an uncertain benefit, these antimalarial drugs have been associated previously with flares of psoriasis.
Dr. Duffin agreed and went on to warn that COVID-19 infection itself is a potential trigger for flares. She cited two published case reports of flares associated with psoriasis. Although one patient had also been exposed to hydroxychloroquine, she said the risk of psoriasis-induced flare “makes sense” based on previous associations made between flares and other viral infections and stress.
In patients with psoriasis who contract COVID-19 infection, Dr. Duffin concurred with the NPF guidance that management decisions should be made on a “case-by-case basis.” Although the NPF guidance states that “most patients can restart psoriasis and/or psoriatic arthritis treatments after complete resolution of COVID-19 symptoms,” no specific advice was offered on the decision to stop treatments.
For protecting psoriasis patients from infection and managing COVID-19 in those who become infected, much of the NPF advice is consistent with that offered to patients without psoriasis. This involves practicing infection control that reduces risk of transmission. Both the NPF guidance and Dr. Duffin suggested telemedicine is appropriate for limiting in-patient visits under pandemic conditions.
Although patients with psoriasis are more likely than the general population to have the comorbidities associated with bad COVID-19 infection outcomes, according to the NPF guidance, Dr. Duffin called the overall data evaluating susceptibility among psoriasis patients “reassuring.” She cautioned that the data are still limited, but the evidence so far suggests that neither psoriasis nor biologics are independent risk factors for acquiring COVID-19 or having a worse outcome if infected.
Yet, more definitive data are needed, and Dr. Duffin advised clinicians and patients to consult the NPF website for updates. “More up-to-date information will certainly be added as we go forward,” she said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
This NPF task force on COVID-19 is meeting every 2 weeks, according to Joel M. Gelfand, MD, professor of dermatology, University of Pennsylvania, Philadelphia, and cochair of the task force. Dr. Gelfand reported that updates are based on a discussion of the available data.
“We will be releasing additional recommendations as necessary based on the developments,” he said in an interview. Updates are not necessarily required at this frequency but can be if appropriate. The goal is to keep recommendations current and evidence-based.
Dr. Duffin reported financial relationships with Amgen, AbbVie, Bristol-Myers Squibb, Boehringer-Ingelheim, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, Siena, and UCB. Dr. Gelfand reported financial relationships with AbbVie, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, Pfizer, Roche, and UCB.
This publication and Global Academy for Medical Education are owned by the same parent company.
The available according to a summary of published studies and expert opinions summarized at the annual Coastal Dermatology Symposium, held virtually.
For patients with psoriasis concerned about their outcome if infected with COVID-19, “there is no evidence to support stopping biologics or systemic agents, so I am asking my patients to continue,” Kristina C. Duffin, MD, professor and chair of dermatology at the University of Utah, Salt Lake City, said at the meeting.
The National Psoriasis Foundation, which created a COVID-19 task force and maintains a COVID-19 Resource Center on its website, has provided similar advice. Many statements are phrased cautiously and clinicians are encouraged to practice shared decision-making, but the NPF guidance supports continuing effective therapy – or, in newly diagnosed patients, starting effective therapy – among those who are not infected with SARS-CoV2.
Patients with a new diagnosis of psoriasis “should be aware that untreated psoriatic disease is associated with serious impact on physical and emotional health, and in the case of psoriatic arthritis, can lead to permanent joint damage and disability,” according to the NPF guidance.
Overall, the “existing data generally suggest” that most treatments for psoriasis and psoriatic arthritis “do not meaningfully alter the risks of contracting SARS-CoV2 or having a worse course of COVID-19 illness,” the current guidance states. Yet, because of limited data this “is not known with certainty.”
Chronic systemic steroids are an exception. In a review of recently published studies evaluating whether psoriasis or its therapies increase risk of adverse outcomes in patients with COVID-19 infection, Dr. Duffin pointed to several that associated systemic steroids with hospitalization or other markers of severe disease.
The NPF guidance also recommends avoiding chronic systemic steroids in patients with psoriasis during the current COVID-19 era “if possible.” In patients with psoriatic arthritis who require systemic steroids, the guidance recommends “the lowest dose necessary to achieve the desired therapeutic effect.”
This is not necessarily true in patients with psoriasis and COVID-19 infection. Based on the potential for systemic steroids to improve outcomes in hospitalized COVID-19 patients requiring oxygen, steroids “should not be withheld” even when the justification is concern about the potential risk of flares with withdrawal, according to the NPF guidance statement.
The NPF guidance specifically cautions against use of hydroxychloroquine or chloroquine for prevention or treatment of COVID-19. In addition to an uncertain benefit, these antimalarial drugs have been associated previously with flares of psoriasis.
Dr. Duffin agreed and went on to warn that COVID-19 infection itself is a potential trigger for flares. She cited two published case reports of flares associated with psoriasis. Although one patient had also been exposed to hydroxychloroquine, she said the risk of psoriasis-induced flare “makes sense” based on previous associations made between flares and other viral infections and stress.
In patients with psoriasis who contract COVID-19 infection, Dr. Duffin concurred with the NPF guidance that management decisions should be made on a “case-by-case basis.” Although the NPF guidance states that “most patients can restart psoriasis and/or psoriatic arthritis treatments after complete resolution of COVID-19 symptoms,” no specific advice was offered on the decision to stop treatments.
For protecting psoriasis patients from infection and managing COVID-19 in those who become infected, much of the NPF advice is consistent with that offered to patients without psoriasis. This involves practicing infection control that reduces risk of transmission. Both the NPF guidance and Dr. Duffin suggested telemedicine is appropriate for limiting in-patient visits under pandemic conditions.
Although patients with psoriasis are more likely than the general population to have the comorbidities associated with bad COVID-19 infection outcomes, according to the NPF guidance, Dr. Duffin called the overall data evaluating susceptibility among psoriasis patients “reassuring.” She cautioned that the data are still limited, but the evidence so far suggests that neither psoriasis nor biologics are independent risk factors for acquiring COVID-19 or having a worse outcome if infected.
Yet, more definitive data are needed, and Dr. Duffin advised clinicians and patients to consult the NPF website for updates. “More up-to-date information will certainly be added as we go forward,” she said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
This NPF task force on COVID-19 is meeting every 2 weeks, according to Joel M. Gelfand, MD, professor of dermatology, University of Pennsylvania, Philadelphia, and cochair of the task force. Dr. Gelfand reported that updates are based on a discussion of the available data.
“We will be releasing additional recommendations as necessary based on the developments,” he said in an interview. Updates are not necessarily required at this frequency but can be if appropriate. The goal is to keep recommendations current and evidence-based.
Dr. Duffin reported financial relationships with Amgen, AbbVie, Bristol-Myers Squibb, Boehringer-Ingelheim, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, Siena, and UCB. Dr. Gelfand reported financial relationships with AbbVie, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, Pfizer, Roche, and UCB.
This publication and Global Academy for Medical Education are owned by the same parent company.
The available according to a summary of published studies and expert opinions summarized at the annual Coastal Dermatology Symposium, held virtually.
For patients with psoriasis concerned about their outcome if infected with COVID-19, “there is no evidence to support stopping biologics or systemic agents, so I am asking my patients to continue,” Kristina C. Duffin, MD, professor and chair of dermatology at the University of Utah, Salt Lake City, said at the meeting.
The National Psoriasis Foundation, which created a COVID-19 task force and maintains a COVID-19 Resource Center on its website, has provided similar advice. Many statements are phrased cautiously and clinicians are encouraged to practice shared decision-making, but the NPF guidance supports continuing effective therapy – or, in newly diagnosed patients, starting effective therapy – among those who are not infected with SARS-CoV2.
Patients with a new diagnosis of psoriasis “should be aware that untreated psoriatic disease is associated with serious impact on physical and emotional health, and in the case of psoriatic arthritis, can lead to permanent joint damage and disability,” according to the NPF guidance.
Overall, the “existing data generally suggest” that most treatments for psoriasis and psoriatic arthritis “do not meaningfully alter the risks of contracting SARS-CoV2 or having a worse course of COVID-19 illness,” the current guidance states. Yet, because of limited data this “is not known with certainty.”
Chronic systemic steroids are an exception. In a review of recently published studies evaluating whether psoriasis or its therapies increase risk of adverse outcomes in patients with COVID-19 infection, Dr. Duffin pointed to several that associated systemic steroids with hospitalization or other markers of severe disease.
The NPF guidance also recommends avoiding chronic systemic steroids in patients with psoriasis during the current COVID-19 era “if possible.” In patients with psoriatic arthritis who require systemic steroids, the guidance recommends “the lowest dose necessary to achieve the desired therapeutic effect.”
This is not necessarily true in patients with psoriasis and COVID-19 infection. Based on the potential for systemic steroids to improve outcomes in hospitalized COVID-19 patients requiring oxygen, steroids “should not be withheld” even when the justification is concern about the potential risk of flares with withdrawal, according to the NPF guidance statement.
The NPF guidance specifically cautions against use of hydroxychloroquine or chloroquine for prevention or treatment of COVID-19. In addition to an uncertain benefit, these antimalarial drugs have been associated previously with flares of psoriasis.
Dr. Duffin agreed and went on to warn that COVID-19 infection itself is a potential trigger for flares. She cited two published case reports of flares associated with psoriasis. Although one patient had also been exposed to hydroxychloroquine, she said the risk of psoriasis-induced flare “makes sense” based on previous associations made between flares and other viral infections and stress.
In patients with psoriasis who contract COVID-19 infection, Dr. Duffin concurred with the NPF guidance that management decisions should be made on a “case-by-case basis.” Although the NPF guidance states that “most patients can restart psoriasis and/or psoriatic arthritis treatments after complete resolution of COVID-19 symptoms,” no specific advice was offered on the decision to stop treatments.
For protecting psoriasis patients from infection and managing COVID-19 in those who become infected, much of the NPF advice is consistent with that offered to patients without psoriasis. This involves practicing infection control that reduces risk of transmission. Both the NPF guidance and Dr. Duffin suggested telemedicine is appropriate for limiting in-patient visits under pandemic conditions.
Although patients with psoriasis are more likely than the general population to have the comorbidities associated with bad COVID-19 infection outcomes, according to the NPF guidance, Dr. Duffin called the overall data evaluating susceptibility among psoriasis patients “reassuring.” She cautioned that the data are still limited, but the evidence so far suggests that neither psoriasis nor biologics are independent risk factors for acquiring COVID-19 or having a worse outcome if infected.
Yet, more definitive data are needed, and Dr. Duffin advised clinicians and patients to consult the NPF website for updates. “More up-to-date information will certainly be added as we go forward,” she said at the meeting, jointly presented by the University of Louisville and Global Academy for Medical Education.
This NPF task force on COVID-19 is meeting every 2 weeks, according to Joel M. Gelfand, MD, professor of dermatology, University of Pennsylvania, Philadelphia, and cochair of the task force. Dr. Gelfand reported that updates are based on a discussion of the available data.
“We will be releasing additional recommendations as necessary based on the developments,” he said in an interview. Updates are not necessarily required at this frequency but can be if appropriate. The goal is to keep recommendations current and evidence-based.
Dr. Duffin reported financial relationships with Amgen, AbbVie, Bristol-Myers Squibb, Boehringer-Ingelheim, Celgene, Eli Lilly, Janssen, Novartis, Pfizer, Siena, and UCB. Dr. Gelfand reported financial relationships with AbbVie, Bristol-Myers Squibb, GlaxoSmithKline, Lilly, Pfizer, Roche, and UCB.
This publication and Global Academy for Medical Education are owned by the same parent company.
FROM COASTAL DERM
Dr. Anique K. Forrester joins editorial advisory board of Clinical Psychiatry News
Clinical Psychiatry News is pleased to announce that Anique K. Forrester, MD, has joined its editorial advisory board.
Dr. Forrester, who is board certified in the subspecialty of psychosomatic medicine (consultation-liaison psychiatry), holds numerous leadership positions at the University of Maryland, Baltimore. She is director of the consultation-liaison psychiatry fellowship at the university and serves as director of education for the C-L psychiatry PGY-2 rotation. Dr. Forrester, an assistant professor, also serves as chair of the department of psychiatry’s diversity committee and is the coordinator of the cultural psychiatry resident course.
Dr. Forrester completed her psychiatry residency training as well as psychosomatic medicine fellowship training at Sidney Kimmel Medical College, Philadelphia. She is a diplomate of the American Board of Psychiatry and Neurology. In addition, Dr. Forrester is a member of the Academy of Consultation-Liaison Psychiatry. She is a graduate of Howard University, Washington.
Her research interests include perinatal psychiatry, psycho-oncology, and cultural competence in medicine.
Welcome aboard, Dr. Forrester!
Clinical Psychiatry News is pleased to announce that Anique K. Forrester, MD, has joined its editorial advisory board.
Dr. Forrester, who is board certified in the subspecialty of psychosomatic medicine (consultation-liaison psychiatry), holds numerous leadership positions at the University of Maryland, Baltimore. She is director of the consultation-liaison psychiatry fellowship at the university and serves as director of education for the C-L psychiatry PGY-2 rotation. Dr. Forrester, an assistant professor, also serves as chair of the department of psychiatry’s diversity committee and is the coordinator of the cultural psychiatry resident course.
Dr. Forrester completed her psychiatry residency training as well as psychosomatic medicine fellowship training at Sidney Kimmel Medical College, Philadelphia. She is a diplomate of the American Board of Psychiatry and Neurology. In addition, Dr. Forrester is a member of the Academy of Consultation-Liaison Psychiatry. She is a graduate of Howard University, Washington.
Her research interests include perinatal psychiatry, psycho-oncology, and cultural competence in medicine.
Welcome aboard, Dr. Forrester!
Clinical Psychiatry News is pleased to announce that Anique K. Forrester, MD, has joined its editorial advisory board.
Dr. Forrester, who is board certified in the subspecialty of psychosomatic medicine (consultation-liaison psychiatry), holds numerous leadership positions at the University of Maryland, Baltimore. She is director of the consultation-liaison psychiatry fellowship at the university and serves as director of education for the C-L psychiatry PGY-2 rotation. Dr. Forrester, an assistant professor, also serves as chair of the department of psychiatry’s diversity committee and is the coordinator of the cultural psychiatry resident course.
Dr. Forrester completed her psychiatry residency training as well as psychosomatic medicine fellowship training at Sidney Kimmel Medical College, Philadelphia. She is a diplomate of the American Board of Psychiatry and Neurology. In addition, Dr. Forrester is a member of the Academy of Consultation-Liaison Psychiatry. She is a graduate of Howard University, Washington.
Her research interests include perinatal psychiatry, psycho-oncology, and cultural competence in medicine.
Welcome aboard, Dr. Forrester!
Direct-acting agents cure hepatitis C in children
Between 23,000 and 46,000 U.S. children live with chronic hepatitis C virus with a prevalence of 0.17% anti–hepatitis C virus (HCV) antibody positivity in those aged 6-11 years and 0.39% among children aged 12-19 years. In the United States, genotype 1 is most frequent, followed by genotypes 2 and 3. About 99% of cases result from vertical transmission; transfusion-related cases have not been observed in recent decades.Only viremic mothers are at risk of transmission as those who have spontaneously cleared HCV viremia or have been treated successfully do not risk transmission. Maternal HCV viral load appears to be a risk factor for HCV transmission, however transmission is reported at all levels of viremia.
In conjunction with the opioid epidemics, the prevalence of HCV infection has increased over the last decade. The Centers for Disease Control and Prevention reported that, between 2009 and 2014, the prevalence of HCV infection increased from 1.8 to 3.4 per 1,000 live births. They identified substantial state-to-state variation with the highest rate in West Virginia (22.6 per 1,000 live births), and the lowest in Hawaii (0.7 per 1,000 live births). The implications are clear that increasing numbers of newborns are exposed to HCV and, if transmission rates are between 1% and 5%, 80-400 U.S. infants each year acquire HCV infection.
HCV in children
HCV in children is almost always associated with persistent transaminitis. Chronic infection is defined as the persistence of HCV RNA for at least 6 months, and clearance of HCV infection is determined by the persistent disappearance of HCV RNA. Regardless of infection status, an infant may have detectable maternal anti-HCV antibody in serum until 18 months of age, resulting from passive transfer. In addition, prolonged infection can lead to cirrhosis, hepatocellular carcinoma, or decompensated liver disease. Potential extrahepatic manifestations including reduced physical and psychosocial health also are linked to chronic HCV. Autoimmune disease also has been reported in children with HCV. As well, the stigma of HCV elicits fear in school and child care settings that is a result of public misunderstanding regarding routes of hepatitis C transmission. No restriction of regular childhood activities is required in the daily life of HCV-infected children.
Taken together, increasing rates of HCV infection in pregnant women, increasing numbers of exposed and infected infants annually, potential for both short- and long-term morbidity, and curative nontoxic treatment,
Screening for HCV
There is considerable discussion about which strategy for screening of at-risk infants is more appropriate. Some groups advocate for HCV-RNA testing within the first year of life. Proponents argue the use of a highly sensitive RNA assay early in life has potential to increase detection of infected infants while a negative result allows the conclusion the infant is not infected. Advocates hypothesize that early identification has potential to improve continued follow-up.
Opponents argue that early testing does not change the need for repeat testing after 18 months to confirm diagnosis. They also argue that HCV RNA is more expensive than an antibody-based testing; and treatment will not begin prior to age 3 as there is still opportunity for viremia to spontaneously clear.
Direct acting agents licensed
Ledipasvir/sofosbuvir (Harvoni) was initially demonstrated as curative for genotype 1, 4, 5, or 6 infection in a phase 2, multicenter, open-label study of 100 adolescents with genotype 1 treated for 12 weeks. Sustained virologic response (SVR) was documented in 98% of participants.The regimen was safe and well tolerated in this population, and the adult dosage formulation resulted in pharmacokinetic characteristics similar to those observed in adults. Two clinical trials supported the efficacy of ledipasvir/sofosbuvir in the pediatric population aged 3-11 years. This regimen also is recommended for interferon-experienced (± ribavirin, with or without an HCV protease inhibitor) children and adolescents aged 3 years or older with genotype 1 or 4. A 12-week course is recommended for patients without cirrhosis; 24 weeks is recommended for those with compensated cirrhosis. The combination of ledipasvir/sofosbuvir is the only treatment option for children aged 3-6 years with genotype 1, 4, 5, or 6 infection.
The efficacy of sofosbuvir/velpatasvir (Epclusa) once daily for 12 weeks was first evaluated in an open-label trial among children aged 6 years and older with genotype 1, 2, 3, 4, or 6 infection, without cirrhosis or with compensated cirrhosis. Subsequently, the “cocktail” was evaluated in children aged 6-12 years, with 76% genotype 1, 3% genotype 2, 15% genotype 3, and 6% genotype 4. SVR12 rates were 93% (50/54) in children with genotype 1, 91% (10/11) in those with genotype 3, and 100% in participants with genotype 2 (2/2) or genotype 4 (4/4). Sofosbuvir/velpatasvir was approved in March 2020 by the Food and Drug Administration for pediatric patients aged 6 years and older. Given its pangenotypic activity, safety, and efficacy, sofosbuvir/velpatasvir is currently recommended as a first choice for HCV treatment in children and adolescents aged at least 6 years.
The daily fixed-dose combination of glecaprevir/pibrentasvir (Mavyret) was approved in April 2019 for adolescents aged 12-17 years, and weighing at least 45 kg.Treatment is for 8 weeks, and includes treatment-naive patients without cirrhosis or those with compensated cirrhosis. SVR12 rates for Mavyret have ranged from 91% to 100 % across clinic trials. FDA approval and HCV guideline treatment recommendations for direct-acting antiviral (DAA)–experienced adolescents are based on clinical trial data from adults. Given its pangenotypic activity, safety, and efficacy record in adult patients, glecaprevir/pibrentasvir is recommended as a first choice for adolescent HCV treatment. Glecaprevir/pibrentasvir once approved for children less than 3 years of age will be safe and efficacious as a pangenotypic treatment option in children with chronic HCV infection.
Current recommendations
Tools for identifying HCV infected infants as early as a few months of age are available, yet studies demonstrate that a minority of at-risk children are tested for HCV using either an HCV polymerase chain reaction strategy early in life or an anti-HCV antibody strategy after 18 months of age.
Therapy with direct-acting agents is now licensed to those aged 3 years and offers the potential for cure, eliminating concern for possible progression after prolonged infection. Such therapy offers the potential to eliminate the stigma faced by many children as well as the hepatic and extrahepatic manifestations observed in children. Medication formulation and the child’s abilities to take the medication needs to be considered when prescribing DAAs. It is important to assess if the child can successfully swallow pills. Currently, Harvoni is the only medication that comes in both pellet and pill formulations. The dose is based on weight. The pellets need to be given in a small amount of nonacidic food; they cannot be chewed.
All children with chronic HCV infection are candidates for treatment. When significant fibrosis and/or cirrhosis is present treatment should not be delayed once the child is age 3 years; when only transaminitis is present, treatment can be delayed. In our experience, parents are eager to complete treatment before starting kindergarten.
Liver biopsy for obtaining liver tissue for histopathologic examination is not routinely indicated in children with chronic HCV infection but should be evaluated case by case. Noninvasive tests of hepatic fibrosis have been used in children, these include serologic markers (i.e., FibroSure) and radiologic tests such as ultrasound-based transient elastography (i.e., Fibroscan). Validation for pediatric patients is variable for the different serologic tests. Studies have shown that Fibroscan using the M probe is feasible for a wide range of ages, but poor patient cooperation may make measurement difficult.
Further details regarding dosing and choice of formulation is available at https://www.hcvguidelines.org/unique-populations/children.
Dr. Sabharwal is assistant professor of pediatrics at Boston University and attending physician in pediatric infectious diseases at Boston Medical Center. Ms. Moloney is an instructor in pediatrics at Boston University and a pediatric nurse practitioner in pediatric infectious diseases at Boston Medicine Center. Dr. Pelton is professor of pediatrics and epidemiology at Boston University and public health and senior attending physician at Boston Medical Center. Boston Medical Center received funding from AbbVie for study of Harvoni in Children 3 years of age and older. Email them at [email protected].
References
MMWR Morb Mortal Wkly Rep. 2017 May 12;66(18):470-3. Hepatol Commun. 2017 March 23. doi: 10.1002/hep4.1028. Hepatology. 2020 Feb;71(2):422-30. Lancet Gastroenterol Hepatol. 2019 Apr 11. doi: 10.1016/S2468-1253(19)30046-9. Arch Dis Child. 2006 Sep;91(9):781-5. J Pediatr Gastroenterol Nutr. 2010 Feb;50(2):123-31.
Between 23,000 and 46,000 U.S. children live with chronic hepatitis C virus with a prevalence of 0.17% anti–hepatitis C virus (HCV) antibody positivity in those aged 6-11 years and 0.39% among children aged 12-19 years. In the United States, genotype 1 is most frequent, followed by genotypes 2 and 3. About 99% of cases result from vertical transmission; transfusion-related cases have not been observed in recent decades.Only viremic mothers are at risk of transmission as those who have spontaneously cleared HCV viremia or have been treated successfully do not risk transmission. Maternal HCV viral load appears to be a risk factor for HCV transmission, however transmission is reported at all levels of viremia.
In conjunction with the opioid epidemics, the prevalence of HCV infection has increased over the last decade. The Centers for Disease Control and Prevention reported that, between 2009 and 2014, the prevalence of HCV infection increased from 1.8 to 3.4 per 1,000 live births. They identified substantial state-to-state variation with the highest rate in West Virginia (22.6 per 1,000 live births), and the lowest in Hawaii (0.7 per 1,000 live births). The implications are clear that increasing numbers of newborns are exposed to HCV and, if transmission rates are between 1% and 5%, 80-400 U.S. infants each year acquire HCV infection.
HCV in children
HCV in children is almost always associated with persistent transaminitis. Chronic infection is defined as the persistence of HCV RNA for at least 6 months, and clearance of HCV infection is determined by the persistent disappearance of HCV RNA. Regardless of infection status, an infant may have detectable maternal anti-HCV antibody in serum until 18 months of age, resulting from passive transfer. In addition, prolonged infection can lead to cirrhosis, hepatocellular carcinoma, or decompensated liver disease. Potential extrahepatic manifestations including reduced physical and psychosocial health also are linked to chronic HCV. Autoimmune disease also has been reported in children with HCV. As well, the stigma of HCV elicits fear in school and child care settings that is a result of public misunderstanding regarding routes of hepatitis C transmission. No restriction of regular childhood activities is required in the daily life of HCV-infected children.
Taken together, increasing rates of HCV infection in pregnant women, increasing numbers of exposed and infected infants annually, potential for both short- and long-term morbidity, and curative nontoxic treatment,
Screening for HCV
There is considerable discussion about which strategy for screening of at-risk infants is more appropriate. Some groups advocate for HCV-RNA testing within the first year of life. Proponents argue the use of a highly sensitive RNA assay early in life has potential to increase detection of infected infants while a negative result allows the conclusion the infant is not infected. Advocates hypothesize that early identification has potential to improve continued follow-up.
Opponents argue that early testing does not change the need for repeat testing after 18 months to confirm diagnosis. They also argue that HCV RNA is more expensive than an antibody-based testing; and treatment will not begin prior to age 3 as there is still opportunity for viremia to spontaneously clear.
Direct acting agents licensed
Ledipasvir/sofosbuvir (Harvoni) was initially demonstrated as curative for genotype 1, 4, 5, or 6 infection in a phase 2, multicenter, open-label study of 100 adolescents with genotype 1 treated for 12 weeks. Sustained virologic response (SVR) was documented in 98% of participants.The regimen was safe and well tolerated in this population, and the adult dosage formulation resulted in pharmacokinetic characteristics similar to those observed in adults. Two clinical trials supported the efficacy of ledipasvir/sofosbuvir in the pediatric population aged 3-11 years. This regimen also is recommended for interferon-experienced (± ribavirin, with or without an HCV protease inhibitor) children and adolescents aged 3 years or older with genotype 1 or 4. A 12-week course is recommended for patients without cirrhosis; 24 weeks is recommended for those with compensated cirrhosis. The combination of ledipasvir/sofosbuvir is the only treatment option for children aged 3-6 years with genotype 1, 4, 5, or 6 infection.
The efficacy of sofosbuvir/velpatasvir (Epclusa) once daily for 12 weeks was first evaluated in an open-label trial among children aged 6 years and older with genotype 1, 2, 3, 4, or 6 infection, without cirrhosis or with compensated cirrhosis. Subsequently, the “cocktail” was evaluated in children aged 6-12 years, with 76% genotype 1, 3% genotype 2, 15% genotype 3, and 6% genotype 4. SVR12 rates were 93% (50/54) in children with genotype 1, 91% (10/11) in those with genotype 3, and 100% in participants with genotype 2 (2/2) or genotype 4 (4/4). Sofosbuvir/velpatasvir was approved in March 2020 by the Food and Drug Administration for pediatric patients aged 6 years and older. Given its pangenotypic activity, safety, and efficacy, sofosbuvir/velpatasvir is currently recommended as a first choice for HCV treatment in children and adolescents aged at least 6 years.
The daily fixed-dose combination of glecaprevir/pibrentasvir (Mavyret) was approved in April 2019 for adolescents aged 12-17 years, and weighing at least 45 kg.Treatment is for 8 weeks, and includes treatment-naive patients without cirrhosis or those with compensated cirrhosis. SVR12 rates for Mavyret have ranged from 91% to 100 % across clinic trials. FDA approval and HCV guideline treatment recommendations for direct-acting antiviral (DAA)–experienced adolescents are based on clinical trial data from adults. Given its pangenotypic activity, safety, and efficacy record in adult patients, glecaprevir/pibrentasvir is recommended as a first choice for adolescent HCV treatment. Glecaprevir/pibrentasvir once approved for children less than 3 years of age will be safe and efficacious as a pangenotypic treatment option in children with chronic HCV infection.
Current recommendations
Tools for identifying HCV infected infants as early as a few months of age are available, yet studies demonstrate that a minority of at-risk children are tested for HCV using either an HCV polymerase chain reaction strategy early in life or an anti-HCV antibody strategy after 18 months of age.
Therapy with direct-acting agents is now licensed to those aged 3 years and offers the potential for cure, eliminating concern for possible progression after prolonged infection. Such therapy offers the potential to eliminate the stigma faced by many children as well as the hepatic and extrahepatic manifestations observed in children. Medication formulation and the child’s abilities to take the medication needs to be considered when prescribing DAAs. It is important to assess if the child can successfully swallow pills. Currently, Harvoni is the only medication that comes in both pellet and pill formulations. The dose is based on weight. The pellets need to be given in a small amount of nonacidic food; they cannot be chewed.
All children with chronic HCV infection are candidates for treatment. When significant fibrosis and/or cirrhosis is present treatment should not be delayed once the child is age 3 years; when only transaminitis is present, treatment can be delayed. In our experience, parents are eager to complete treatment before starting kindergarten.
Liver biopsy for obtaining liver tissue for histopathologic examination is not routinely indicated in children with chronic HCV infection but should be evaluated case by case. Noninvasive tests of hepatic fibrosis have been used in children, these include serologic markers (i.e., FibroSure) and radiologic tests such as ultrasound-based transient elastography (i.e., Fibroscan). Validation for pediatric patients is variable for the different serologic tests. Studies have shown that Fibroscan using the M probe is feasible for a wide range of ages, but poor patient cooperation may make measurement difficult.
Further details regarding dosing and choice of formulation is available at https://www.hcvguidelines.org/unique-populations/children.
Dr. Sabharwal is assistant professor of pediatrics at Boston University and attending physician in pediatric infectious diseases at Boston Medical Center. Ms. Moloney is an instructor in pediatrics at Boston University and a pediatric nurse practitioner in pediatric infectious diseases at Boston Medicine Center. Dr. Pelton is professor of pediatrics and epidemiology at Boston University and public health and senior attending physician at Boston Medical Center. Boston Medical Center received funding from AbbVie for study of Harvoni in Children 3 years of age and older. Email them at [email protected].
References
MMWR Morb Mortal Wkly Rep. 2017 May 12;66(18):470-3. Hepatol Commun. 2017 March 23. doi: 10.1002/hep4.1028. Hepatology. 2020 Feb;71(2):422-30. Lancet Gastroenterol Hepatol. 2019 Apr 11. doi: 10.1016/S2468-1253(19)30046-9. Arch Dis Child. 2006 Sep;91(9):781-5. J Pediatr Gastroenterol Nutr. 2010 Feb;50(2):123-31.
Between 23,000 and 46,000 U.S. children live with chronic hepatitis C virus with a prevalence of 0.17% anti–hepatitis C virus (HCV) antibody positivity in those aged 6-11 years and 0.39% among children aged 12-19 years. In the United States, genotype 1 is most frequent, followed by genotypes 2 and 3. About 99% of cases result from vertical transmission; transfusion-related cases have not been observed in recent decades.Only viremic mothers are at risk of transmission as those who have spontaneously cleared HCV viremia or have been treated successfully do not risk transmission. Maternal HCV viral load appears to be a risk factor for HCV transmission, however transmission is reported at all levels of viremia.
In conjunction with the opioid epidemics, the prevalence of HCV infection has increased over the last decade. The Centers for Disease Control and Prevention reported that, between 2009 and 2014, the prevalence of HCV infection increased from 1.8 to 3.4 per 1,000 live births. They identified substantial state-to-state variation with the highest rate in West Virginia (22.6 per 1,000 live births), and the lowest in Hawaii (0.7 per 1,000 live births). The implications are clear that increasing numbers of newborns are exposed to HCV and, if transmission rates are between 1% and 5%, 80-400 U.S. infants each year acquire HCV infection.
HCV in children
HCV in children is almost always associated with persistent transaminitis. Chronic infection is defined as the persistence of HCV RNA for at least 6 months, and clearance of HCV infection is determined by the persistent disappearance of HCV RNA. Regardless of infection status, an infant may have detectable maternal anti-HCV antibody in serum until 18 months of age, resulting from passive transfer. In addition, prolonged infection can lead to cirrhosis, hepatocellular carcinoma, or decompensated liver disease. Potential extrahepatic manifestations including reduced physical and psychosocial health also are linked to chronic HCV. Autoimmune disease also has been reported in children with HCV. As well, the stigma of HCV elicits fear in school and child care settings that is a result of public misunderstanding regarding routes of hepatitis C transmission. No restriction of regular childhood activities is required in the daily life of HCV-infected children.
Taken together, increasing rates of HCV infection in pregnant women, increasing numbers of exposed and infected infants annually, potential for both short- and long-term morbidity, and curative nontoxic treatment,
Screening for HCV
There is considerable discussion about which strategy for screening of at-risk infants is more appropriate. Some groups advocate for HCV-RNA testing within the first year of life. Proponents argue the use of a highly sensitive RNA assay early in life has potential to increase detection of infected infants while a negative result allows the conclusion the infant is not infected. Advocates hypothesize that early identification has potential to improve continued follow-up.
Opponents argue that early testing does not change the need for repeat testing after 18 months to confirm diagnosis. They also argue that HCV RNA is more expensive than an antibody-based testing; and treatment will not begin prior to age 3 as there is still opportunity for viremia to spontaneously clear.
Direct acting agents licensed
Ledipasvir/sofosbuvir (Harvoni) was initially demonstrated as curative for genotype 1, 4, 5, or 6 infection in a phase 2, multicenter, open-label study of 100 adolescents with genotype 1 treated for 12 weeks. Sustained virologic response (SVR) was documented in 98% of participants.The regimen was safe and well tolerated in this population, and the adult dosage formulation resulted in pharmacokinetic characteristics similar to those observed in adults. Two clinical trials supported the efficacy of ledipasvir/sofosbuvir in the pediatric population aged 3-11 years. This regimen also is recommended for interferon-experienced (± ribavirin, with or without an HCV protease inhibitor) children and adolescents aged 3 years or older with genotype 1 or 4. A 12-week course is recommended for patients without cirrhosis; 24 weeks is recommended for those with compensated cirrhosis. The combination of ledipasvir/sofosbuvir is the only treatment option for children aged 3-6 years with genotype 1, 4, 5, or 6 infection.
The efficacy of sofosbuvir/velpatasvir (Epclusa) once daily for 12 weeks was first evaluated in an open-label trial among children aged 6 years and older with genotype 1, 2, 3, 4, or 6 infection, without cirrhosis or with compensated cirrhosis. Subsequently, the “cocktail” was evaluated in children aged 6-12 years, with 76% genotype 1, 3% genotype 2, 15% genotype 3, and 6% genotype 4. SVR12 rates were 93% (50/54) in children with genotype 1, 91% (10/11) in those with genotype 3, and 100% in participants with genotype 2 (2/2) or genotype 4 (4/4). Sofosbuvir/velpatasvir was approved in March 2020 by the Food and Drug Administration for pediatric patients aged 6 years and older. Given its pangenotypic activity, safety, and efficacy, sofosbuvir/velpatasvir is currently recommended as a first choice for HCV treatment in children and adolescents aged at least 6 years.
The daily fixed-dose combination of glecaprevir/pibrentasvir (Mavyret) was approved in April 2019 for adolescents aged 12-17 years, and weighing at least 45 kg.Treatment is for 8 weeks, and includes treatment-naive patients without cirrhosis or those with compensated cirrhosis. SVR12 rates for Mavyret have ranged from 91% to 100 % across clinic trials. FDA approval and HCV guideline treatment recommendations for direct-acting antiviral (DAA)–experienced adolescents are based on clinical trial data from adults. Given its pangenotypic activity, safety, and efficacy record in adult patients, glecaprevir/pibrentasvir is recommended as a first choice for adolescent HCV treatment. Glecaprevir/pibrentasvir once approved for children less than 3 years of age will be safe and efficacious as a pangenotypic treatment option in children with chronic HCV infection.
Current recommendations
Tools for identifying HCV infected infants as early as a few months of age are available, yet studies demonstrate that a minority of at-risk children are tested for HCV using either an HCV polymerase chain reaction strategy early in life or an anti-HCV antibody strategy after 18 months of age.
Therapy with direct-acting agents is now licensed to those aged 3 years and offers the potential for cure, eliminating concern for possible progression after prolonged infection. Such therapy offers the potential to eliminate the stigma faced by many children as well as the hepatic and extrahepatic manifestations observed in children. Medication formulation and the child’s abilities to take the medication needs to be considered when prescribing DAAs. It is important to assess if the child can successfully swallow pills. Currently, Harvoni is the only medication that comes in both pellet and pill formulations. The dose is based on weight. The pellets need to be given in a small amount of nonacidic food; they cannot be chewed.
All children with chronic HCV infection are candidates for treatment. When significant fibrosis and/or cirrhosis is present treatment should not be delayed once the child is age 3 years; when only transaminitis is present, treatment can be delayed. In our experience, parents are eager to complete treatment before starting kindergarten.
Liver biopsy for obtaining liver tissue for histopathologic examination is not routinely indicated in children with chronic HCV infection but should be evaluated case by case. Noninvasive tests of hepatic fibrosis have been used in children, these include serologic markers (i.e., FibroSure) and radiologic tests such as ultrasound-based transient elastography (i.e., Fibroscan). Validation for pediatric patients is variable for the different serologic tests. Studies have shown that Fibroscan using the M probe is feasible for a wide range of ages, but poor patient cooperation may make measurement difficult.
Further details regarding dosing and choice of formulation is available at https://www.hcvguidelines.org/unique-populations/children.
Dr. Sabharwal is assistant professor of pediatrics at Boston University and attending physician in pediatric infectious diseases at Boston Medical Center. Ms. Moloney is an instructor in pediatrics at Boston University and a pediatric nurse practitioner in pediatric infectious diseases at Boston Medicine Center. Dr. Pelton is professor of pediatrics and epidemiology at Boston University and public health and senior attending physician at Boston Medical Center. Boston Medical Center received funding from AbbVie for study of Harvoni in Children 3 years of age and older. Email them at [email protected].
References
MMWR Morb Mortal Wkly Rep. 2017 May 12;66(18):470-3. Hepatol Commun. 2017 March 23. doi: 10.1002/hep4.1028. Hepatology. 2020 Feb;71(2):422-30. Lancet Gastroenterol Hepatol. 2019 Apr 11. doi: 10.1016/S2468-1253(19)30046-9. Arch Dis Child. 2006 Sep;91(9):781-5. J Pediatr Gastroenterol Nutr. 2010 Feb;50(2):123-31.
Trends in COVID-19 Risk-Adjusted Mortality Rates
Early reports showed high mortality from coronavirus disease 2019 (COVID-19), while current United States data mortality rates are lower, raising hope that new treatments and management strategies have improved outcomes. For instance, Centers for Disease Control and Prevention data show that 6.7% of cases resulted in death in April, compared with 1.9% in September.1 However, the demographics of those infected have also changed, and more available testing may mean more comprehensive identification and earlier treatment. Nationally, for instance, the median age of confirmed cases was 38 years at the end of August, down from 46 years at the start of May.2 Therefore, whether decreasing COVID-19 mortality rates simply reflect changing demographics or represent actual improvements in clinical care is unknown. The objective of this analysis was to assess outcomes over time in a single health system, accounting for changes in demographics, clinical factors, and severity of disease at presentation.
METHODS
We analyzed monthly mortality rates for admissions between March 1 and August 31, 2020, in a single health system in New York City. Outcomes were obtained as of October 8, 2020. We included all hospitalizations of people 18 years and older with laboratory-confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection identified during the hospitalization or in the prior 2 weeks, excluding those admitted to hospice care. Patients with multiple hospitalizations (N=208 patients, 229 hospitalizations, 4.4%) were included repeatedly if they continued to have laboratory-confirmed disease. Patients without admission vital signs (N=28) were excluded. Mortality was defined as in-hospital death or discharge to hospice care. In-house laboratory testing began March 16 and all inpatients were tested for SARS-CoV-2 by April 1; elective surgeries resumed May 4-11 and were only conducted on confirmed SARS-CoV-2–negative patients.
All data were obtained from the electronic health record (Epic Systems, Verona, Wisconsin). Diagnosis codes were obtained from the problem list, past medical history, and billing codes. In addition, we used objective data such as hemoglobin A1c, ejection fraction, outpatient creatinine, and outpatient blood pressure to augment problem list diagnoses where relevant.
Based on prior literature, we constructed multivariable logistic regression models for mortality adjusting for age; sex; self-reported race and ethnicity; body mass index; smoking history; presence of hypertension, heart failure, hyperlipidemia, coronary artery disease, diabetes, cancer, chronic kidney disease, dementia, or pulmonary disease individually as dummy variables; and admission oxygen saturation, D-dimer, ferritin, and C-reactive protein.3-6 In the first model (C statistic 0.82), we did not include month of admission as a covariate and calculated the ratio of the sum of observed and expected deaths (obtained from the model) in each month to obtain the standardized mortality ratio (SMR) for each month. We then multiplied each period’s SMR by the overall average crude mortality to generate monthly adjusted mortality rates. We calculated Poisson control limits and indicated points outside the control limits as significantly different.
In a second model (C statistic 0.84), we included month as a covariate and calculated average marginal effects (AME) for each time period by using the margins library in R,7 which uses a discrete first-difference in predicted outcomes to obtain the AME. The average marginal effect represents the percentage point difference between the reference period (March) and a subsequent time period in probability of death or discharge to hospice, for equivalent patients. We obtained lower and upper confidence intervals for the AME using a bootstrapping approach described in Green.8 Finally, we conducted two sensitivity analyses: one, restricting the analysis to only those patients with principal diagnosis of COVID-19, sepsis, or respiratory disease (see Appendix A for complete list of codes) and one restricting the analysis to only those with length of stay of at least 3 days.
All statistical analyses were conducted with R, version 4.0.2. All analyses used 2-sided statistical tests, and we considered a P value < .05 to be statistically significant without adjustment for multiple testing. The NYU institutional review board approved the study and granted a waiver of consent and a waiver of the Health Information Portability and Accountability Act.
RESULTS
We included 5,121 hospitalizations, of which 5,118 (99.94%) had known outcomes (death or hospital discharge). Peak hospitalizations occurred in late March to mid-April, which accounted for 53% of the hospitalizations. Median length of stay for patients who died or were discharged to hospice was 8 days (interquartile range, 4-15; max 140 days). The median age and the proportion male or with any comorbidity decreased over time (Table). For instance, the proportion with any chronic condition decreased from 81% in March to 72% in August.
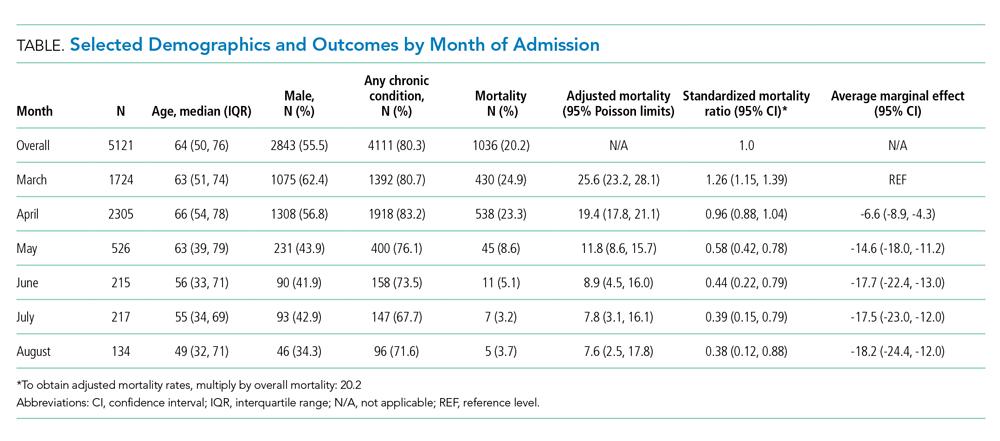
Adjusted mortality dropped each month, from 25.6% in March to 7.6% in August (Table and Figure). The SMR declined progressively over time, from 1.26 (95% CI, 1.15-1.39) in March to 0.38 (95% CI, 0.12-0.88) in August (Table). The adjusted average marginal effect was also significantly lower than in March in every subsequent month, reaching a maximum of an average 18.2 (95% CI, 12.0-24.4) percentage point decrease in probability of death in August, accounting for changes in demographics and clinical severity (Table and Appendix B). The decrease in unadjusted mortality over time was observed across age groups (Appendix C).
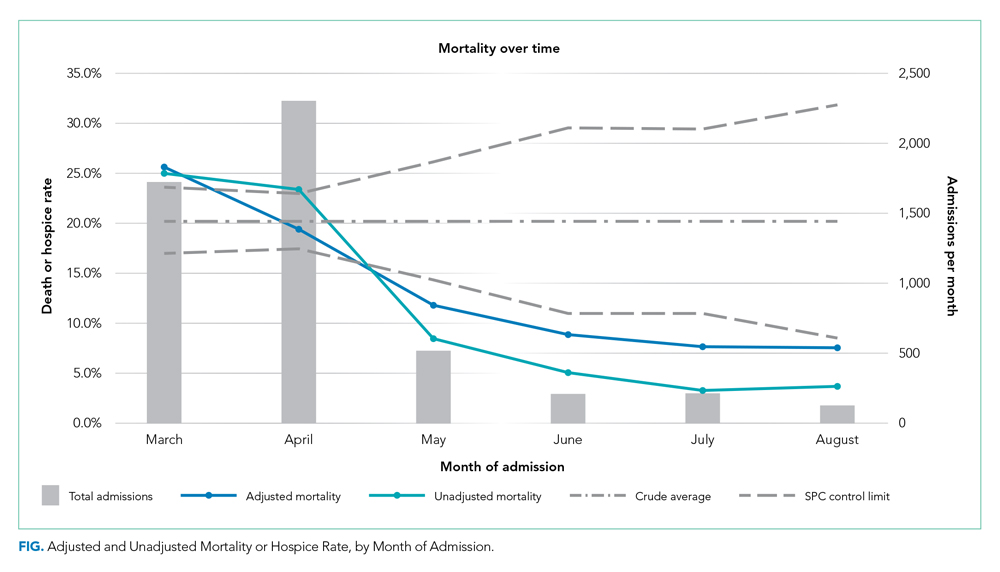
Results of the two sensitivity analyses were similar (Appendices D and E), though attenuated in the case of the sepsis/respiratory cohort, with adjusted mortality falling from 31.4% to 14.4%, SMR decreasing from 1.28 (95% CI, 1.16-1.41) to 0.59 (95% CI, 0.16-1.50), and AME in August 17.0 percentage points (95% CI, 6.0-28.1).
DISCUSSION
In this study of COVID-19 mortality over 6 months at a single health system, we found that changes in demographics and severity of illness at presentation did not fully explain decreases in mortality seen over time. Even after risk adjustment for a variety of clinical and demographic factors, including severity of illness at presentation, mortality was significantly and progressively lower over the course of the study period.
Similar risk-adjusted results have been preliminarily reported among intensive care unit patients in a preprint from the United Kingdom.9 Incremental improvements in outcomes are likely a combination of increasing clinical experience, decreasing hospital volume, growing use of new pharmacologic treatments (such as systemic corticosteroids,10 remdesivir,11 and anticytokine treatments), nonpharmacologic treatments (such as placing the patient in the prone position, or proning, rather than on their back), earlier intervention, community awareness, and, potentially, lower viral load exposure from increased mask wearing and social distancing.12
Strengths of this study include highly detailed electronic health record data on hospitalizations at three different hospitals, a diverse patient population,6 near-complete study outcomes, and a lengthy period of investigation of 6 months. However, this study does have limitations. All patients were from a single geographic region and treated within a single health system, though restricting data to one system reduces institution-level variability and allows us to assess how care may have evolved with growing experience. Aggregating data from numerous health systems that might be at different stages of local outbreaks, provide different quality of care, and contribute different numbers of patients in each period introduces its own biases. We were also unable to disentangle different potential explanatory factors given the observational nature of the study. Residual confounding, such as a higher proportion of particularly frail patients admitted in earlier periods, is also a possibility, though the fact that we observed declines across all age groups mitigates this concern. Thresholds for hospital admission may also have changed over time with less severely ill patients being admitted in the later time periods. While changing admission thresholds could have contributed to higher survival rates in the latter portions of the study, our inclusion of several highly predictive clinical and laboratory results likely captured many aspects of disease severity.
CONCLUSION
In summary, data from one health system suggest that COVID-19 remains a serious disease for high-risk patients, but that mortality rates are improving over time.
1. CDC COVID Data Tracker. 2020. Centers for Disease Control and Prevention. Accessed October 14, 2020. https://covid.cdc.gov/covid-data-tracker/#trends_dailytrendscases
2. Boehmer TK, DeVies J, Caruso E, et al. Changing age distribution of the COVID-19 pandemic - United States, May-August 2020. MMWR Morb Mortal Wkly Rep. 2020;69(39):1404-1409 http://dx.doi.org/0.15585/mmwr.mm6939e1
3. Lu L, Zhong W, Bian Z, et al. A comparison of mortality-related risk factors of COVID-19, SARS, and MERS: A systematic review and meta-analysis. J Infect. 2020;81(4):318-e25. https://doi.org/10.1016/j.jinf.2020.07.002
4. Parohan M, Yaghoubi S, Seraji A, Javanbakht MH, Sarraf P, Djalali M. Risk factors for mortality in patients with coronavirus disease 2019 (COVID-19) infection: a systematic review and meta-analysis of observational studies. Aging Male. 2020;Jun8:1-9. https://doi.org/10.1080/13685538.2020.1774748
5. Zheng Z, Peng F, Xu B, et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect. 2020;81(2):e16-e25. https://doi.org/10.1016/j.jinf.2020.04.021
6. Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. https://doi.org/10.1136/bmj.m1966
7. margins: Marginal Effects for Model Objects [computer program]. Version R package version 0.3.232018. Accessed October 1, 2020. https://rdrr.io/cran/margins/
8. Greene WH. Econometric Analysis. 7th ed. Pearson; 2012.
9. Doidge JC, Mouncey PR, Thomas K, et al. Trends in intensive care for patients with COVID-19 in England, Wales and Northern Ireland. Preprints 2020. Preprint posted online August 11, 2020. https://doi.org/10.20944/preprints202008.0267.v1
10. Recovery Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N Engl J Med. 2020. Online first July 17, 2020. https://doi.org/10.1056/NEJMoa2021436
11. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19 – final report. N Enl J Med. 2020. Online first October 8, 2020. https://doi.org/10.1056/NEJMoa2007764
12. Gandhi M, Rutherford GW. Facial masking for Covid-19 - potential for “variolation” as we await a vaccine. N Engl J Med. 2020. Online first September 8, 2020. https://doi.org/10.1056/NEJMp2026913
Early reports showed high mortality from coronavirus disease 2019 (COVID-19), while current United States data mortality rates are lower, raising hope that new treatments and management strategies have improved outcomes. For instance, Centers for Disease Control and Prevention data show that 6.7% of cases resulted in death in April, compared with 1.9% in September.1 However, the demographics of those infected have also changed, and more available testing may mean more comprehensive identification and earlier treatment. Nationally, for instance, the median age of confirmed cases was 38 years at the end of August, down from 46 years at the start of May.2 Therefore, whether decreasing COVID-19 mortality rates simply reflect changing demographics or represent actual improvements in clinical care is unknown. The objective of this analysis was to assess outcomes over time in a single health system, accounting for changes in demographics, clinical factors, and severity of disease at presentation.
METHODS
We analyzed monthly mortality rates for admissions between March 1 and August 31, 2020, in a single health system in New York City. Outcomes were obtained as of October 8, 2020. We included all hospitalizations of people 18 years and older with laboratory-confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection identified during the hospitalization or in the prior 2 weeks, excluding those admitted to hospice care. Patients with multiple hospitalizations (N=208 patients, 229 hospitalizations, 4.4%) were included repeatedly if they continued to have laboratory-confirmed disease. Patients without admission vital signs (N=28) were excluded. Mortality was defined as in-hospital death or discharge to hospice care. In-house laboratory testing began March 16 and all inpatients were tested for SARS-CoV-2 by April 1; elective surgeries resumed May 4-11 and were only conducted on confirmed SARS-CoV-2–negative patients.
All data were obtained from the electronic health record (Epic Systems, Verona, Wisconsin). Diagnosis codes were obtained from the problem list, past medical history, and billing codes. In addition, we used objective data such as hemoglobin A1c, ejection fraction, outpatient creatinine, and outpatient blood pressure to augment problem list diagnoses where relevant.
Based on prior literature, we constructed multivariable logistic regression models for mortality adjusting for age; sex; self-reported race and ethnicity; body mass index; smoking history; presence of hypertension, heart failure, hyperlipidemia, coronary artery disease, diabetes, cancer, chronic kidney disease, dementia, or pulmonary disease individually as dummy variables; and admission oxygen saturation, D-dimer, ferritin, and C-reactive protein.3-6 In the first model (C statistic 0.82), we did not include month of admission as a covariate and calculated the ratio of the sum of observed and expected deaths (obtained from the model) in each month to obtain the standardized mortality ratio (SMR) for each month. We then multiplied each period’s SMR by the overall average crude mortality to generate monthly adjusted mortality rates. We calculated Poisson control limits and indicated points outside the control limits as significantly different.
In a second model (C statistic 0.84), we included month as a covariate and calculated average marginal effects (AME) for each time period by using the margins library in R,7 which uses a discrete first-difference in predicted outcomes to obtain the AME. The average marginal effect represents the percentage point difference between the reference period (March) and a subsequent time period in probability of death or discharge to hospice, for equivalent patients. We obtained lower and upper confidence intervals for the AME using a bootstrapping approach described in Green.8 Finally, we conducted two sensitivity analyses: one, restricting the analysis to only those patients with principal diagnosis of COVID-19, sepsis, or respiratory disease (see Appendix A for complete list of codes) and one restricting the analysis to only those with length of stay of at least 3 days.
All statistical analyses were conducted with R, version 4.0.2. All analyses used 2-sided statistical tests, and we considered a P value < .05 to be statistically significant without adjustment for multiple testing. The NYU institutional review board approved the study and granted a waiver of consent and a waiver of the Health Information Portability and Accountability Act.
RESULTS
We included 5,121 hospitalizations, of which 5,118 (99.94%) had known outcomes (death or hospital discharge). Peak hospitalizations occurred in late March to mid-April, which accounted for 53% of the hospitalizations. Median length of stay for patients who died or were discharged to hospice was 8 days (interquartile range, 4-15; max 140 days). The median age and the proportion male or with any comorbidity decreased over time (Table). For instance, the proportion with any chronic condition decreased from 81% in March to 72% in August.

Adjusted mortality dropped each month, from 25.6% in March to 7.6% in August (Table and Figure). The SMR declined progressively over time, from 1.26 (95% CI, 1.15-1.39) in March to 0.38 (95% CI, 0.12-0.88) in August (Table). The adjusted average marginal effect was also significantly lower than in March in every subsequent month, reaching a maximum of an average 18.2 (95% CI, 12.0-24.4) percentage point decrease in probability of death in August, accounting for changes in demographics and clinical severity (Table and Appendix B). The decrease in unadjusted mortality over time was observed across age groups (Appendix C).

Results of the two sensitivity analyses were similar (Appendices D and E), though attenuated in the case of the sepsis/respiratory cohort, with adjusted mortality falling from 31.4% to 14.4%, SMR decreasing from 1.28 (95% CI, 1.16-1.41) to 0.59 (95% CI, 0.16-1.50), and AME in August 17.0 percentage points (95% CI, 6.0-28.1).
DISCUSSION
In this study of COVID-19 mortality over 6 months at a single health system, we found that changes in demographics and severity of illness at presentation did not fully explain decreases in mortality seen over time. Even after risk adjustment for a variety of clinical and demographic factors, including severity of illness at presentation, mortality was significantly and progressively lower over the course of the study period.
Similar risk-adjusted results have been preliminarily reported among intensive care unit patients in a preprint from the United Kingdom.9 Incremental improvements in outcomes are likely a combination of increasing clinical experience, decreasing hospital volume, growing use of new pharmacologic treatments (such as systemic corticosteroids,10 remdesivir,11 and anticytokine treatments), nonpharmacologic treatments (such as placing the patient in the prone position, or proning, rather than on their back), earlier intervention, community awareness, and, potentially, lower viral load exposure from increased mask wearing and social distancing.12
Strengths of this study include highly detailed electronic health record data on hospitalizations at three different hospitals, a diverse patient population,6 near-complete study outcomes, and a lengthy period of investigation of 6 months. However, this study does have limitations. All patients were from a single geographic region and treated within a single health system, though restricting data to one system reduces institution-level variability and allows us to assess how care may have evolved with growing experience. Aggregating data from numerous health systems that might be at different stages of local outbreaks, provide different quality of care, and contribute different numbers of patients in each period introduces its own biases. We were also unable to disentangle different potential explanatory factors given the observational nature of the study. Residual confounding, such as a higher proportion of particularly frail patients admitted in earlier periods, is also a possibility, though the fact that we observed declines across all age groups mitigates this concern. Thresholds for hospital admission may also have changed over time with less severely ill patients being admitted in the later time periods. While changing admission thresholds could have contributed to higher survival rates in the latter portions of the study, our inclusion of several highly predictive clinical and laboratory results likely captured many aspects of disease severity.
CONCLUSION
In summary, data from one health system suggest that COVID-19 remains a serious disease for high-risk patients, but that mortality rates are improving over time.
Early reports showed high mortality from coronavirus disease 2019 (COVID-19), while current United States data mortality rates are lower, raising hope that new treatments and management strategies have improved outcomes. For instance, Centers for Disease Control and Prevention data show that 6.7% of cases resulted in death in April, compared with 1.9% in September.1 However, the demographics of those infected have also changed, and more available testing may mean more comprehensive identification and earlier treatment. Nationally, for instance, the median age of confirmed cases was 38 years at the end of August, down from 46 years at the start of May.2 Therefore, whether decreasing COVID-19 mortality rates simply reflect changing demographics or represent actual improvements in clinical care is unknown. The objective of this analysis was to assess outcomes over time in a single health system, accounting for changes in demographics, clinical factors, and severity of disease at presentation.
METHODS
We analyzed monthly mortality rates for admissions between March 1 and August 31, 2020, in a single health system in New York City. Outcomes were obtained as of October 8, 2020. We included all hospitalizations of people 18 years and older with laboratory-confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection identified during the hospitalization or in the prior 2 weeks, excluding those admitted to hospice care. Patients with multiple hospitalizations (N=208 patients, 229 hospitalizations, 4.4%) were included repeatedly if they continued to have laboratory-confirmed disease. Patients without admission vital signs (N=28) were excluded. Mortality was defined as in-hospital death or discharge to hospice care. In-house laboratory testing began March 16 and all inpatients were tested for SARS-CoV-2 by April 1; elective surgeries resumed May 4-11 and were only conducted on confirmed SARS-CoV-2–negative patients.
All data were obtained from the electronic health record (Epic Systems, Verona, Wisconsin). Diagnosis codes were obtained from the problem list, past medical history, and billing codes. In addition, we used objective data such as hemoglobin A1c, ejection fraction, outpatient creatinine, and outpatient blood pressure to augment problem list diagnoses where relevant.
Based on prior literature, we constructed multivariable logistic regression models for mortality adjusting for age; sex; self-reported race and ethnicity; body mass index; smoking history; presence of hypertension, heart failure, hyperlipidemia, coronary artery disease, diabetes, cancer, chronic kidney disease, dementia, or pulmonary disease individually as dummy variables; and admission oxygen saturation, D-dimer, ferritin, and C-reactive protein.3-6 In the first model (C statistic 0.82), we did not include month of admission as a covariate and calculated the ratio of the sum of observed and expected deaths (obtained from the model) in each month to obtain the standardized mortality ratio (SMR) for each month. We then multiplied each period’s SMR by the overall average crude mortality to generate monthly adjusted mortality rates. We calculated Poisson control limits and indicated points outside the control limits as significantly different.
In a second model (C statistic 0.84), we included month as a covariate and calculated average marginal effects (AME) for each time period by using the margins library in R,7 which uses a discrete first-difference in predicted outcomes to obtain the AME. The average marginal effect represents the percentage point difference between the reference period (March) and a subsequent time period in probability of death or discharge to hospice, for equivalent patients. We obtained lower and upper confidence intervals for the AME using a bootstrapping approach described in Green.8 Finally, we conducted two sensitivity analyses: one, restricting the analysis to only those patients with principal diagnosis of COVID-19, sepsis, or respiratory disease (see Appendix A for complete list of codes) and one restricting the analysis to only those with length of stay of at least 3 days.
All statistical analyses were conducted with R, version 4.0.2. All analyses used 2-sided statistical tests, and we considered a P value < .05 to be statistically significant without adjustment for multiple testing. The NYU institutional review board approved the study and granted a waiver of consent and a waiver of the Health Information Portability and Accountability Act.
RESULTS
We included 5,121 hospitalizations, of which 5,118 (99.94%) had known outcomes (death or hospital discharge). Peak hospitalizations occurred in late March to mid-April, which accounted for 53% of the hospitalizations. Median length of stay for patients who died or were discharged to hospice was 8 days (interquartile range, 4-15; max 140 days). The median age and the proportion male or with any comorbidity decreased over time (Table). For instance, the proportion with any chronic condition decreased from 81% in March to 72% in August.

Adjusted mortality dropped each month, from 25.6% in March to 7.6% in August (Table and Figure). The SMR declined progressively over time, from 1.26 (95% CI, 1.15-1.39) in March to 0.38 (95% CI, 0.12-0.88) in August (Table). The adjusted average marginal effect was also significantly lower than in March in every subsequent month, reaching a maximum of an average 18.2 (95% CI, 12.0-24.4) percentage point decrease in probability of death in August, accounting for changes in demographics and clinical severity (Table and Appendix B). The decrease in unadjusted mortality over time was observed across age groups (Appendix C).

Results of the two sensitivity analyses were similar (Appendices D and E), though attenuated in the case of the sepsis/respiratory cohort, with adjusted mortality falling from 31.4% to 14.4%, SMR decreasing from 1.28 (95% CI, 1.16-1.41) to 0.59 (95% CI, 0.16-1.50), and AME in August 17.0 percentage points (95% CI, 6.0-28.1).
DISCUSSION
In this study of COVID-19 mortality over 6 months at a single health system, we found that changes in demographics and severity of illness at presentation did not fully explain decreases in mortality seen over time. Even after risk adjustment for a variety of clinical and demographic factors, including severity of illness at presentation, mortality was significantly and progressively lower over the course of the study period.
Similar risk-adjusted results have been preliminarily reported among intensive care unit patients in a preprint from the United Kingdom.9 Incremental improvements in outcomes are likely a combination of increasing clinical experience, decreasing hospital volume, growing use of new pharmacologic treatments (such as systemic corticosteroids,10 remdesivir,11 and anticytokine treatments), nonpharmacologic treatments (such as placing the patient in the prone position, or proning, rather than on their back), earlier intervention, community awareness, and, potentially, lower viral load exposure from increased mask wearing and social distancing.12
Strengths of this study include highly detailed electronic health record data on hospitalizations at three different hospitals, a diverse patient population,6 near-complete study outcomes, and a lengthy period of investigation of 6 months. However, this study does have limitations. All patients were from a single geographic region and treated within a single health system, though restricting data to one system reduces institution-level variability and allows us to assess how care may have evolved with growing experience. Aggregating data from numerous health systems that might be at different stages of local outbreaks, provide different quality of care, and contribute different numbers of patients in each period introduces its own biases. We were also unable to disentangle different potential explanatory factors given the observational nature of the study. Residual confounding, such as a higher proportion of particularly frail patients admitted in earlier periods, is also a possibility, though the fact that we observed declines across all age groups mitigates this concern. Thresholds for hospital admission may also have changed over time with less severely ill patients being admitted in the later time periods. While changing admission thresholds could have contributed to higher survival rates in the latter portions of the study, our inclusion of several highly predictive clinical and laboratory results likely captured many aspects of disease severity.
CONCLUSION
In summary, data from one health system suggest that COVID-19 remains a serious disease for high-risk patients, but that mortality rates are improving over time.
1. CDC COVID Data Tracker. 2020. Centers for Disease Control and Prevention. Accessed October 14, 2020. https://covid.cdc.gov/covid-data-tracker/#trends_dailytrendscases
2. Boehmer TK, DeVies J, Caruso E, et al. Changing age distribution of the COVID-19 pandemic - United States, May-August 2020. MMWR Morb Mortal Wkly Rep. 2020;69(39):1404-1409 http://dx.doi.org/0.15585/mmwr.mm6939e1
3. Lu L, Zhong W, Bian Z, et al. A comparison of mortality-related risk factors of COVID-19, SARS, and MERS: A systematic review and meta-analysis. J Infect. 2020;81(4):318-e25. https://doi.org/10.1016/j.jinf.2020.07.002
4. Parohan M, Yaghoubi S, Seraji A, Javanbakht MH, Sarraf P, Djalali M. Risk factors for mortality in patients with coronavirus disease 2019 (COVID-19) infection: a systematic review and meta-analysis of observational studies. Aging Male. 2020;Jun8:1-9. https://doi.org/10.1080/13685538.2020.1774748
5. Zheng Z, Peng F, Xu B, et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect. 2020;81(2):e16-e25. https://doi.org/10.1016/j.jinf.2020.04.021
6. Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. https://doi.org/10.1136/bmj.m1966
7. margins: Marginal Effects for Model Objects [computer program]. Version R package version 0.3.232018. Accessed October 1, 2020. https://rdrr.io/cran/margins/
8. Greene WH. Econometric Analysis. 7th ed. Pearson; 2012.
9. Doidge JC, Mouncey PR, Thomas K, et al. Trends in intensive care for patients with COVID-19 in England, Wales and Northern Ireland. Preprints 2020. Preprint posted online August 11, 2020. https://doi.org/10.20944/preprints202008.0267.v1
10. Recovery Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N Engl J Med. 2020. Online first July 17, 2020. https://doi.org/10.1056/NEJMoa2021436
11. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19 – final report. N Enl J Med. 2020. Online first October 8, 2020. https://doi.org/10.1056/NEJMoa2007764
12. Gandhi M, Rutherford GW. Facial masking for Covid-19 - potential for “variolation” as we await a vaccine. N Engl J Med. 2020. Online first September 8, 2020. https://doi.org/10.1056/NEJMp2026913
1. CDC COVID Data Tracker. 2020. Centers for Disease Control and Prevention. Accessed October 14, 2020. https://covid.cdc.gov/covid-data-tracker/#trends_dailytrendscases
2. Boehmer TK, DeVies J, Caruso E, et al. Changing age distribution of the COVID-19 pandemic - United States, May-August 2020. MMWR Morb Mortal Wkly Rep. 2020;69(39):1404-1409 http://dx.doi.org/0.15585/mmwr.mm6939e1
3. Lu L, Zhong W, Bian Z, et al. A comparison of mortality-related risk factors of COVID-19, SARS, and MERS: A systematic review and meta-analysis. J Infect. 2020;81(4):318-e25. https://doi.org/10.1016/j.jinf.2020.07.002
4. Parohan M, Yaghoubi S, Seraji A, Javanbakht MH, Sarraf P, Djalali M. Risk factors for mortality in patients with coronavirus disease 2019 (COVID-19) infection: a systematic review and meta-analysis of observational studies. Aging Male. 2020;Jun8:1-9. https://doi.org/10.1080/13685538.2020.1774748
5. Zheng Z, Peng F, Xu B, et al. Risk factors of critical & mortal COVID-19 cases: a systematic literature review and meta-analysis. J Infect. 2020;81(2):e16-e25. https://doi.org/10.1016/j.jinf.2020.04.021
6. Petrilli CM, Jones SA, Yang J, et al. Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York City: prospective cohort study. BMJ. 2020;369:m1966. https://doi.org/10.1136/bmj.m1966
7. margins: Marginal Effects for Model Objects [computer program]. Version R package version 0.3.232018. Accessed October 1, 2020. https://rdrr.io/cran/margins/
8. Greene WH. Econometric Analysis. 7th ed. Pearson; 2012.
9. Doidge JC, Mouncey PR, Thomas K, et al. Trends in intensive care for patients with COVID-19 in England, Wales and Northern Ireland. Preprints 2020. Preprint posted online August 11, 2020. https://doi.org/10.20944/preprints202008.0267.v1
10. Recovery Collaborative Group, Horby P, Lim WS, et al. Dexamethasone in hospitalized patients with Covid-19 - preliminary report. N Engl J Med. 2020. Online first July 17, 2020. https://doi.org/10.1056/NEJMoa2021436
11. Beigel JH, Tomashek KM, Dodd LE, et al. Remdesivir for the treatment of Covid-19 – final report. N Enl J Med. 2020. Online first October 8, 2020. https://doi.org/10.1056/NEJMoa2007764
12. Gandhi M, Rutherford GW. Facial masking for Covid-19 - potential for “variolation” as we await a vaccine. N Engl J Med. 2020. Online first September 8, 2020. https://doi.org/10.1056/NEJMp2026913
© 2020 Society of Hospital Medicine
Final ASCEND study data: Acalabrutinib beat standard of care for r/r CLL
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
Acalabrutinib, a next-generation Bruton tyrosine kinase inhibitor, provides prolonged progression-free survival and better tolerability, compared with standard-of-care regimens for relapsed or refractory chronic lymphocytic leukemia (CLL), according to final results from the phase 3 ASCEND study.
The estimated 18-month progression-free survival (PFS) at a median of 22 months was 82% in 155 patients treated with acalabrutinib, compared with 48% in 155 treated with investigator’s choice of either idelalisib-rituximab (IdR) or bendamustine-rituximab (BR), which were given in 119 and 36 patients, respectively, Paolo Ghia, MD, PhD, reported at the Society of Hematologic Oncology virtual meeting.
The benefits of acalabrutinib were apparent regardless of high-risk genetic characteristics: Those with and without both del(17p) and TP53 mutations had similarly good PFS outcomes with acalabrutinib versus IdR/BR (HRs, 0.11 and 0.29, respectively), as did those with versus without unmutated IgVH (HRs, 0.28 and 0.30, respectively), said Dr. Ghia, professor of medical oncology at the Università Vita-Salute San Raffaele and IRCCS Ospedale San Raffaele, Milan.
The median overall survival was not reached in either arm, but estimated 18-month OS was 88% in both groups, likely because of the crossover being allowed for nonresponders in the IdR/BR groups, he noted.
Overall responses
The investigator-assessed overall response rates, including partial response or better, were also similar in the groups at 80% and 84%, respectively, and ORR, including partial response with lymphocytosis, was 92% versus 88%.
The duration of response was not reached in the acalabrutinib arm versus 18 months with IdR/BR, and estimated duration of response was 85% versus 49%.
The median drug exposure with acalabrutinib was approximately double that with IdR and about four times that of BR, Dr. Ghia said, noting that the difference between acalabrutinib and BR is explained by the short 6-month duration of treatment with BR, but the difference between acalabrutinib and IdR is because of adverse events (AEs).
Adverse events
AEs were the most common reason for treatment discontinuation in all three groups, but they led to discontinuation in only 16% with acalabrutinib versus 56% with IdR, he added.
The rates of AEs and AEs of clinical interest were generally similar to those reported at the interim analysis as presented in 2019 at the European Hematology Association annual meeting and published in the Journal of Clinical Oncology, despite the additional 6 months of follow up, he said.
Additionally, the incidence of grade 3 or higher AEs, serious AEs, and treatment-related AEs were all greater with IdR than with acalabrutinib or BR. The most common AEs with acalabrutinib were headache, neutropenia, diarrhea, and upper-respiratory infection, which were mostly grade 1 or 2. The most common grade 3 or higher AEs were neutropenia, anemia, and pneumonia, which were reported in 12%, 17%, and 7% of patients.
Confirmatory results
“The final results from the ASCEND study confirm the findings at the interim analysis and support the favorable efficacy and safety of acalabrutinib versus standard-of-care regimens ... in patients with relapsed/refractory CLL,” Dr. Ghia said.
“Overall, these final results from ASCENT support the use of acalabrutinib in patients with relapsed/refractory CLL, including those with high-risk genetic features.”
This study was sponsored by Acerta Pharma. Dr. Ghia reported consulting or advisory roles, grant or research funding, and/or honoraria from Abbvie, BeiGene, Janssen, Gilead Sciences, Sunesis Pharmaceuticals, Juno Therapeutics, ArQule, Adaptive Biotechnologies, Dynamo Therapeutics, MEI Pharma, and Novartis.
SOURCE: Ghia P et al. SOHO 2020, Abstract CLL-091.
FROM SOHO 2020
How to assess erythema in children with skin of color
When assessing inflammatory dermatoses in children with skin of color, it may be necessary to train the eye to recognize subtle changes and colors other than red, a doctor suggested at the virtual American Academy of Pediatrics annual meeting.
First, doctors should see whether they can detect any erythema, said Latanya T. Benjamin, MD, associate professor of pediatric dermatology at Florida Atlantic University, Boca Raton. “If the answer is no because of the background competing chromophore, then shift your focus off of the erythema and perhaps onto other colors that the skin can demonstrate,” such as red-brown, violaceous, or grayish hues.
Comparing involved areas with normal skin also may help. “Sometimes you can pick up subtleties in colors that way,” Dr. Benjamin said.
Finally, look for other changes that could relate to the patient’s condition. For example, when diagnosing acne, Dr. Benjamin looks for pigmentary sequelae like hyperpigmentation. “If a patient has atopic dermatitis, is there hypopigmentation on other areas of the face?”
Consider cutaneous T-cell lymphoma in the differential diagnosis of generalized hypopigmented patches and plaques in patients with darker skin types, Dr. Benjamin noted. Other diagnoses that may result in hypopigmentation include pityriasis alba, vitiligo, tinea versicolor, ash-leaf macules, Hansen’s disease, postinflammatory hypopigmentation secondary to atopic dermatitis, and tinea corporis.
Be sensitive to the fact that changes in skin color can be “very annoying or devastating to the family,” even with medically benign conditions such as pityriasis alba, Dr. Benjamin added.
Detecting redness in brown skin tones can take practice, Candrice R. Heath, MD, a member of the board of directors for the Skin of Color Society, commented in an interview.
Furthermore, presentations vary. For instance, depictions of atopic dermatitis in educational materials may focus on red patches and plaques but “miss that there are several presentations in those with darker skin tones, including follicular prominence, hyperpigmented plaques, and coin-shaped lesions,” said Dr. Heath, assistant professor of dermatology at Temple University, Philadelphia.
“The skin of color population is growing,” noted Dr. Heath. “By 2023, there will be more children with skin of color than without in the United States.”
While Dr. Heath has lectured about skin of color as it relates to pediatric patients for years, “now with the nation’s renewed interest in disparities in health care, it is the perfect time to highlight conditions that present more commonly in skin of color and present differently in those with skin of color.”
Dr. Benjamin had no conflicts of interest. Dr. Heath serves as associate editor of Cutis, which is owned by the same company as this publication.
When assessing inflammatory dermatoses in children with skin of color, it may be necessary to train the eye to recognize subtle changes and colors other than red, a doctor suggested at the virtual American Academy of Pediatrics annual meeting.
First, doctors should see whether they can detect any erythema, said Latanya T. Benjamin, MD, associate professor of pediatric dermatology at Florida Atlantic University, Boca Raton. “If the answer is no because of the background competing chromophore, then shift your focus off of the erythema and perhaps onto other colors that the skin can demonstrate,” such as red-brown, violaceous, or grayish hues.
Comparing involved areas with normal skin also may help. “Sometimes you can pick up subtleties in colors that way,” Dr. Benjamin said.
Finally, look for other changes that could relate to the patient’s condition. For example, when diagnosing acne, Dr. Benjamin looks for pigmentary sequelae like hyperpigmentation. “If a patient has atopic dermatitis, is there hypopigmentation on other areas of the face?”
Consider cutaneous T-cell lymphoma in the differential diagnosis of generalized hypopigmented patches and plaques in patients with darker skin types, Dr. Benjamin noted. Other diagnoses that may result in hypopigmentation include pityriasis alba, vitiligo, tinea versicolor, ash-leaf macules, Hansen’s disease, postinflammatory hypopigmentation secondary to atopic dermatitis, and tinea corporis.
Be sensitive to the fact that changes in skin color can be “very annoying or devastating to the family,” even with medically benign conditions such as pityriasis alba, Dr. Benjamin added.
Detecting redness in brown skin tones can take practice, Candrice R. Heath, MD, a member of the board of directors for the Skin of Color Society, commented in an interview.
Furthermore, presentations vary. For instance, depictions of atopic dermatitis in educational materials may focus on red patches and plaques but “miss that there are several presentations in those with darker skin tones, including follicular prominence, hyperpigmented plaques, and coin-shaped lesions,” said Dr. Heath, assistant professor of dermatology at Temple University, Philadelphia.
“The skin of color population is growing,” noted Dr. Heath. “By 2023, there will be more children with skin of color than without in the United States.”
While Dr. Heath has lectured about skin of color as it relates to pediatric patients for years, “now with the nation’s renewed interest in disparities in health care, it is the perfect time to highlight conditions that present more commonly in skin of color and present differently in those with skin of color.”
Dr. Benjamin had no conflicts of interest. Dr. Heath serves as associate editor of Cutis, which is owned by the same company as this publication.
When assessing inflammatory dermatoses in children with skin of color, it may be necessary to train the eye to recognize subtle changes and colors other than red, a doctor suggested at the virtual American Academy of Pediatrics annual meeting.
First, doctors should see whether they can detect any erythema, said Latanya T. Benjamin, MD, associate professor of pediatric dermatology at Florida Atlantic University, Boca Raton. “If the answer is no because of the background competing chromophore, then shift your focus off of the erythema and perhaps onto other colors that the skin can demonstrate,” such as red-brown, violaceous, or grayish hues.
Comparing involved areas with normal skin also may help. “Sometimes you can pick up subtleties in colors that way,” Dr. Benjamin said.
Finally, look for other changes that could relate to the patient’s condition. For example, when diagnosing acne, Dr. Benjamin looks for pigmentary sequelae like hyperpigmentation. “If a patient has atopic dermatitis, is there hypopigmentation on other areas of the face?”
Consider cutaneous T-cell lymphoma in the differential diagnosis of generalized hypopigmented patches and plaques in patients with darker skin types, Dr. Benjamin noted. Other diagnoses that may result in hypopigmentation include pityriasis alba, vitiligo, tinea versicolor, ash-leaf macules, Hansen’s disease, postinflammatory hypopigmentation secondary to atopic dermatitis, and tinea corporis.
Be sensitive to the fact that changes in skin color can be “very annoying or devastating to the family,” even with medically benign conditions such as pityriasis alba, Dr. Benjamin added.
Detecting redness in brown skin tones can take practice, Candrice R. Heath, MD, a member of the board of directors for the Skin of Color Society, commented in an interview.
Furthermore, presentations vary. For instance, depictions of atopic dermatitis in educational materials may focus on red patches and plaques but “miss that there are several presentations in those with darker skin tones, including follicular prominence, hyperpigmented plaques, and coin-shaped lesions,” said Dr. Heath, assistant professor of dermatology at Temple University, Philadelphia.
“The skin of color population is growing,” noted Dr. Heath. “By 2023, there will be more children with skin of color than without in the United States.”
While Dr. Heath has lectured about skin of color as it relates to pediatric patients for years, “now with the nation’s renewed interest in disparities in health care, it is the perfect time to highlight conditions that present more commonly in skin of color and present differently in those with skin of color.”
Dr. Benjamin had no conflicts of interest. Dr. Heath serves as associate editor of Cutis, which is owned by the same company as this publication.
FROM AAP 2020
Red hair in women linked to elevated CRP levels in Nurses’ Health Study
Red-haired women were significantly more likely than were women with nonred hair to have elevated levels of C-reactive protein that may increase risk for cardiovascular conditions, according to data from nearly 9,000 women participating in the Nurses’ Health Study.
“Positive associations between red hair and cardiovascular disease and cancer in women, but not men, have been reported,” wrote Rebecca I. Hartman, MD, of Brigham and Women’s Hospital, Harvard Medical School, Boston, and colleagues.
In a study published in the Journal of Investigative Dermatology, they reviewed data from the Nurses’ Health Study, a 1976 cohort study of 121,700 women registered nurses in the United States. They analyzed blood specimens from 8,994 women that were collected between 1989 and 1990. Participants’ natural hair color was determined by asking them their natural hair color at age 21 years, with choices of red, blonde, light brown, dark brown, or black. Overall, dark brown/black hair was the most common color (45%) and 390 of the women (4.3%) had red hair.
The average CRP levels were significantly higher for women with red hair (3.7 mg/L), compared with those with blonde (3.3 mg/L), light brown (3.0 mg/mL), or dark brown/black (3.2 mg/L).
Using the CRP levels for red-haired women as a reference, women with blond, light brown, and dark brown/black hair averaged significantly lower CRP levels than those of red-haired women in an age-adjusted model (–15.2%, –18/1%, and –14.2%, respectively) and in a multivariate analysis (–12.7%, –14.1%, and –10.9%, respectively).
Non-red-haired women had significantly lower odds of high CRP levels compared with red-haired women, with odds ratios of 0.62, 0.60, and 0.67 for women with blonde, light brown, and dark brown/black hair, respectively, in multivariate analysis, the researchers found.
The study was limited by several factors including the use of self-reports for hair color and the relative homogeneity of the Nurses’ Health Study, which has a population of mostly white, female health professionals, the researchers noted.
However, the findings of significantly increased CRP levels “could potentially explain a prior report of increased risks of cardiovascular disease and cancer in red-haired women,” they said. “Although, we observed similar associations in the NHS between red hair and cardiovascular disease and cancer, they were not statistically significant,” they added.
Additional studies are needed to validate and examine the clinical significance of the results, they concluded.
“Elevated CRP levels, a marker of inflammation, have been associated with increased risk for several diseases, including colon cancer and heart disease,” lead author Dr. Hartman said in an interview. “Another study suggested red-haired women have elevated risks of cardiovascular disease and cancer. We wanted to see if different levels of inflammation in red-haired women could possibly explain these findings.”
She said she was not surprised by the findings, “as they were in line with our hypothesis.” In addition, “animal studies suggest that the gene most responsible for red hair, MC1R, may be linked to inflammation,” she said.
While red-haired women were found to have higher CRP levels in the study, “the underlying mechanism and clinical significance remain unknown,” and more research is needed, Dr. Hartman emphasized. “First, our findings need to be validated in women and also examined in men. If our findings are validated, future studies should examine the mechanism of CRP elevation in red-haired women, and whether these women have elevated risks of colon cancer and heart disease,” she said.
“If red-haired women do have increased levels of inflammation, and as a result have elevated risks of colon cancer and heart disease, then future interventions can focus on enhanced screening and possibly chemoprevention in this population,” she added.
The study was supported by the National Institutes of Health. Lead author Dr. Hartman was supported by an American Skin Association Research Grant.
SOURCE: Hartman RI et al. J Invest Dermatol. 2020 Oct 12. doi: 10.1016/j.jid.2020.09.015.
Red-haired women were significantly more likely than were women with nonred hair to have elevated levels of C-reactive protein that may increase risk for cardiovascular conditions, according to data from nearly 9,000 women participating in the Nurses’ Health Study.
“Positive associations between red hair and cardiovascular disease and cancer in women, but not men, have been reported,” wrote Rebecca I. Hartman, MD, of Brigham and Women’s Hospital, Harvard Medical School, Boston, and colleagues.
In a study published in the Journal of Investigative Dermatology, they reviewed data from the Nurses’ Health Study, a 1976 cohort study of 121,700 women registered nurses in the United States. They analyzed blood specimens from 8,994 women that were collected between 1989 and 1990. Participants’ natural hair color was determined by asking them their natural hair color at age 21 years, with choices of red, blonde, light brown, dark brown, or black. Overall, dark brown/black hair was the most common color (45%) and 390 of the women (4.3%) had red hair.
The average CRP levels were significantly higher for women with red hair (3.7 mg/L), compared with those with blonde (3.3 mg/L), light brown (3.0 mg/mL), or dark brown/black (3.2 mg/L).
Using the CRP levels for red-haired women as a reference, women with blond, light brown, and dark brown/black hair averaged significantly lower CRP levels than those of red-haired women in an age-adjusted model (–15.2%, –18/1%, and –14.2%, respectively) and in a multivariate analysis (–12.7%, –14.1%, and –10.9%, respectively).
Non-red-haired women had significantly lower odds of high CRP levels compared with red-haired women, with odds ratios of 0.62, 0.60, and 0.67 for women with blonde, light brown, and dark brown/black hair, respectively, in multivariate analysis, the researchers found.
The study was limited by several factors including the use of self-reports for hair color and the relative homogeneity of the Nurses’ Health Study, which has a population of mostly white, female health professionals, the researchers noted.
However, the findings of significantly increased CRP levels “could potentially explain a prior report of increased risks of cardiovascular disease and cancer in red-haired women,” they said. “Although, we observed similar associations in the NHS between red hair and cardiovascular disease and cancer, they were not statistically significant,” they added.
Additional studies are needed to validate and examine the clinical significance of the results, they concluded.
“Elevated CRP levels, a marker of inflammation, have been associated with increased risk for several diseases, including colon cancer and heart disease,” lead author Dr. Hartman said in an interview. “Another study suggested red-haired women have elevated risks of cardiovascular disease and cancer. We wanted to see if different levels of inflammation in red-haired women could possibly explain these findings.”
She said she was not surprised by the findings, “as they were in line with our hypothesis.” In addition, “animal studies suggest that the gene most responsible for red hair, MC1R, may be linked to inflammation,” she said.
While red-haired women were found to have higher CRP levels in the study, “the underlying mechanism and clinical significance remain unknown,” and more research is needed, Dr. Hartman emphasized. “First, our findings need to be validated in women and also examined in men. If our findings are validated, future studies should examine the mechanism of CRP elevation in red-haired women, and whether these women have elevated risks of colon cancer and heart disease,” she said.
“If red-haired women do have increased levels of inflammation, and as a result have elevated risks of colon cancer and heart disease, then future interventions can focus on enhanced screening and possibly chemoprevention in this population,” she added.
The study was supported by the National Institutes of Health. Lead author Dr. Hartman was supported by an American Skin Association Research Grant.
SOURCE: Hartman RI et al. J Invest Dermatol. 2020 Oct 12. doi: 10.1016/j.jid.2020.09.015.
Red-haired women were significantly more likely than were women with nonred hair to have elevated levels of C-reactive protein that may increase risk for cardiovascular conditions, according to data from nearly 9,000 women participating in the Nurses’ Health Study.
“Positive associations between red hair and cardiovascular disease and cancer in women, but not men, have been reported,” wrote Rebecca I. Hartman, MD, of Brigham and Women’s Hospital, Harvard Medical School, Boston, and colleagues.
In a study published in the Journal of Investigative Dermatology, they reviewed data from the Nurses’ Health Study, a 1976 cohort study of 121,700 women registered nurses in the United States. They analyzed blood specimens from 8,994 women that were collected between 1989 and 1990. Participants’ natural hair color was determined by asking them their natural hair color at age 21 years, with choices of red, blonde, light brown, dark brown, or black. Overall, dark brown/black hair was the most common color (45%) and 390 of the women (4.3%) had red hair.
The average CRP levels were significantly higher for women with red hair (3.7 mg/L), compared with those with blonde (3.3 mg/L), light brown (3.0 mg/mL), or dark brown/black (3.2 mg/L).
Using the CRP levels for red-haired women as a reference, women with blond, light brown, and dark brown/black hair averaged significantly lower CRP levels than those of red-haired women in an age-adjusted model (–15.2%, –18/1%, and –14.2%, respectively) and in a multivariate analysis (–12.7%, –14.1%, and –10.9%, respectively).
Non-red-haired women had significantly lower odds of high CRP levels compared with red-haired women, with odds ratios of 0.62, 0.60, and 0.67 for women with blonde, light brown, and dark brown/black hair, respectively, in multivariate analysis, the researchers found.
The study was limited by several factors including the use of self-reports for hair color and the relative homogeneity of the Nurses’ Health Study, which has a population of mostly white, female health professionals, the researchers noted.
However, the findings of significantly increased CRP levels “could potentially explain a prior report of increased risks of cardiovascular disease and cancer in red-haired women,” they said. “Although, we observed similar associations in the NHS between red hair and cardiovascular disease and cancer, they were not statistically significant,” they added.
Additional studies are needed to validate and examine the clinical significance of the results, they concluded.
“Elevated CRP levels, a marker of inflammation, have been associated with increased risk for several diseases, including colon cancer and heart disease,” lead author Dr. Hartman said in an interview. “Another study suggested red-haired women have elevated risks of cardiovascular disease and cancer. We wanted to see if different levels of inflammation in red-haired women could possibly explain these findings.”
She said she was not surprised by the findings, “as they were in line with our hypothesis.” In addition, “animal studies suggest that the gene most responsible for red hair, MC1R, may be linked to inflammation,” she said.
While red-haired women were found to have higher CRP levels in the study, “the underlying mechanism and clinical significance remain unknown,” and more research is needed, Dr. Hartman emphasized. “First, our findings need to be validated in women and also examined in men. If our findings are validated, future studies should examine the mechanism of CRP elevation in red-haired women, and whether these women have elevated risks of colon cancer and heart disease,” she said.
“If red-haired women do have increased levels of inflammation, and as a result have elevated risks of colon cancer and heart disease, then future interventions can focus on enhanced screening and possibly chemoprevention in this population,” she added.
The study was supported by the National Institutes of Health. Lead author Dr. Hartman was supported by an American Skin Association Research Grant.
SOURCE: Hartman RI et al. J Invest Dermatol. 2020 Oct 12. doi: 10.1016/j.jid.2020.09.015.
FROM THE JOURNAL OF INVESTIGATIVE DERMATOLOGY
CDC expands definition of COVID-19 exposure from ‘close contact’
New data suggest each close encounter – coming within 6 feet of an infected person – can increase the risk for transmission, CDC director Robert Redfield, MD, said during a media briefing.
“As we get more data and understand the science of COVID, we’re going to continue to incorporate that in our recommendations,” Dr. Redfield said in response to a reporter’s question about a recent study.
Previously, the CDC cautioned against spending 15 minutes or longer in close proximity to an infected person, particularly in enclosed indoor spaces.
In a new report published online Oct. 21 in Morbidity and Mortality Weekly Report, however, investigators “determined that an individual who had a series of shorter contacts that over time added up to more than 15 minutes became infected.”
Beware of brief encounters?
On July 28, a 20-year-old male correctional officer in Vermont had multiple brief encounters with six transferred incarcerated or detained people while their SARS-CoV-2 test results were pending. The six were asymptomatic at the time and were housed in a quarantine unit, reported CDC researcher Julia Pringle, PhD, and colleagues.
The following day, all six inmates tested polymerase chain reaction (PCR) positive for COVID-19. The correctional officer did not spend 15 minutes or more within 6 feet of any of the inmates, according to video surveillance footage, and he continued to work.
On Aug. 4, however, he developed symptoms that included loss of smell and taste, myalgia, runny nose, cough, shortness of breath, headache, loss of appetite, and gastrointestinal symptoms. He stayed home starting the next day and tested PCR positive for COVID-19 on Aug. 11.
Further review of the surveillance video showed that the officer had numerous brief encounters of approximately 1 minute each that cumulatively exceeded 15 minutes over a 24-hour period, the researchers reported.
During all the interactions with inmates, the correctional officer wore a cloth mask, gown, and eye protection. The inmates wore masks while in their cells but did not have them on during brief cell doorway interactions or in the recreation room, according to the report.
No interaction is 100% safe
“We know that every activity that involves interacting with others has some degree of risk right now,” said Jay Butler, MD, CDC deputy director for infectious diseases.
“Unfortunately, we’re seeing a distressing trend here in the United States with COVID-19 cases increasing in nearly 75% of the country,” he said. “We’ve confirmed 8.1 million cases and, sadly, over 220,000 deaths since January.
“I know these are numbers, but these are also people,” Dr. Butler added.
“The pandemic is not over,” Dr. Redfield said. “Earlier this week, COVID virus cases reached over 40 million globally. Here in the United States we are approaching a critical phase.”
Four factors associated with higher risk for transmission are the proximity of each encounter, its duration, whether an interaction takes place indoors or outdoors, and the number of people encountered, Dr. Butler said.
Dr. Butler acknowledged widespread fatigue with adherence to personal protection measures, but added that social distancing, mask-wearing, and other measures are more important now than ever. He noted that more Americans will be spending time indoors with the onset of cooler weather and the upcoming holidays.
A note of optimism
Dr. Redfield remains optimistic about the limited availability of a vaccine or vaccines by year’s end but added that “it’s important for all of us to remain diligent in our efforts to defeat this virus.”
“There is hope on the way, in the form of safe and effective vaccines in a matter of weeks or months. To bridge to that next phase, we have to take steps to keep ourselves, our families, and our communities safe,” said Alex Azar, secretary of the Department of Health & Human Services.
“I know it’s been a difficult year for Americans, but we are going to come through this on the other side,” Dr. Redfield said.
New data suggest each close encounter – coming within 6 feet of an infected person – can increase the risk for transmission, CDC director Robert Redfield, MD, said during a media briefing.
“As we get more data and understand the science of COVID, we’re going to continue to incorporate that in our recommendations,” Dr. Redfield said in response to a reporter’s question about a recent study.
Previously, the CDC cautioned against spending 15 minutes or longer in close proximity to an infected person, particularly in enclosed indoor spaces.
In a new report published online Oct. 21 in Morbidity and Mortality Weekly Report, however, investigators “determined that an individual who had a series of shorter contacts that over time added up to more than 15 minutes became infected.”
Beware of brief encounters?
On July 28, a 20-year-old male correctional officer in Vermont had multiple brief encounters with six transferred incarcerated or detained people while their SARS-CoV-2 test results were pending. The six were asymptomatic at the time and were housed in a quarantine unit, reported CDC researcher Julia Pringle, PhD, and colleagues.
The following day, all six inmates tested polymerase chain reaction (PCR) positive for COVID-19. The correctional officer did not spend 15 minutes or more within 6 feet of any of the inmates, according to video surveillance footage, and he continued to work.
On Aug. 4, however, he developed symptoms that included loss of smell and taste, myalgia, runny nose, cough, shortness of breath, headache, loss of appetite, and gastrointestinal symptoms. He stayed home starting the next day and tested PCR positive for COVID-19 on Aug. 11.
Further review of the surveillance video showed that the officer had numerous brief encounters of approximately 1 minute each that cumulatively exceeded 15 minutes over a 24-hour period, the researchers reported.
During all the interactions with inmates, the correctional officer wore a cloth mask, gown, and eye protection. The inmates wore masks while in their cells but did not have them on during brief cell doorway interactions or in the recreation room, according to the report.
No interaction is 100% safe
“We know that every activity that involves interacting with others has some degree of risk right now,” said Jay Butler, MD, CDC deputy director for infectious diseases.
“Unfortunately, we’re seeing a distressing trend here in the United States with COVID-19 cases increasing in nearly 75% of the country,” he said. “We’ve confirmed 8.1 million cases and, sadly, over 220,000 deaths since January.
“I know these are numbers, but these are also people,” Dr. Butler added.
“The pandemic is not over,” Dr. Redfield said. “Earlier this week, COVID virus cases reached over 40 million globally. Here in the United States we are approaching a critical phase.”
Four factors associated with higher risk for transmission are the proximity of each encounter, its duration, whether an interaction takes place indoors or outdoors, and the number of people encountered, Dr. Butler said.
Dr. Butler acknowledged widespread fatigue with adherence to personal protection measures, but added that social distancing, mask-wearing, and other measures are more important now than ever. He noted that more Americans will be spending time indoors with the onset of cooler weather and the upcoming holidays.
A note of optimism
Dr. Redfield remains optimistic about the limited availability of a vaccine or vaccines by year’s end but added that “it’s important for all of us to remain diligent in our efforts to defeat this virus.”
“There is hope on the way, in the form of safe and effective vaccines in a matter of weeks or months. To bridge to that next phase, we have to take steps to keep ourselves, our families, and our communities safe,” said Alex Azar, secretary of the Department of Health & Human Services.
“I know it’s been a difficult year for Americans, but we are going to come through this on the other side,” Dr. Redfield said.
New data suggest each close encounter – coming within 6 feet of an infected person – can increase the risk for transmission, CDC director Robert Redfield, MD, said during a media briefing.
“As we get more data and understand the science of COVID, we’re going to continue to incorporate that in our recommendations,” Dr. Redfield said in response to a reporter’s question about a recent study.
Previously, the CDC cautioned against spending 15 minutes or longer in close proximity to an infected person, particularly in enclosed indoor spaces.
In a new report published online Oct. 21 in Morbidity and Mortality Weekly Report, however, investigators “determined that an individual who had a series of shorter contacts that over time added up to more than 15 minutes became infected.”
Beware of brief encounters?
On July 28, a 20-year-old male correctional officer in Vermont had multiple brief encounters with six transferred incarcerated or detained people while their SARS-CoV-2 test results were pending. The six were asymptomatic at the time and were housed in a quarantine unit, reported CDC researcher Julia Pringle, PhD, and colleagues.
The following day, all six inmates tested polymerase chain reaction (PCR) positive for COVID-19. The correctional officer did not spend 15 minutes or more within 6 feet of any of the inmates, according to video surveillance footage, and he continued to work.
On Aug. 4, however, he developed symptoms that included loss of smell and taste, myalgia, runny nose, cough, shortness of breath, headache, loss of appetite, and gastrointestinal symptoms. He stayed home starting the next day and tested PCR positive for COVID-19 on Aug. 11.
Further review of the surveillance video showed that the officer had numerous brief encounters of approximately 1 minute each that cumulatively exceeded 15 minutes over a 24-hour period, the researchers reported.
During all the interactions with inmates, the correctional officer wore a cloth mask, gown, and eye protection. The inmates wore masks while in their cells but did not have them on during brief cell doorway interactions or in the recreation room, according to the report.
No interaction is 100% safe
“We know that every activity that involves interacting with others has some degree of risk right now,” said Jay Butler, MD, CDC deputy director for infectious diseases.
“Unfortunately, we’re seeing a distressing trend here in the United States with COVID-19 cases increasing in nearly 75% of the country,” he said. “We’ve confirmed 8.1 million cases and, sadly, over 220,000 deaths since January.
“I know these are numbers, but these are also people,” Dr. Butler added.
“The pandemic is not over,” Dr. Redfield said. “Earlier this week, COVID virus cases reached over 40 million globally. Here in the United States we are approaching a critical phase.”
Four factors associated with higher risk for transmission are the proximity of each encounter, its duration, whether an interaction takes place indoors or outdoors, and the number of people encountered, Dr. Butler said.
Dr. Butler acknowledged widespread fatigue with adherence to personal protection measures, but added that social distancing, mask-wearing, and other measures are more important now than ever. He noted that more Americans will be spending time indoors with the onset of cooler weather and the upcoming holidays.
A note of optimism
Dr. Redfield remains optimistic about the limited availability of a vaccine or vaccines by year’s end but added that “it’s important for all of us to remain diligent in our efforts to defeat this virus.”
“There is hope on the way, in the form of safe and effective vaccines in a matter of weeks or months. To bridge to that next phase, we have to take steps to keep ourselves, our families, and our communities safe,” said Alex Azar, secretary of the Department of Health & Human Services.
“I know it’s been a difficult year for Americans, but we are going to come through this on the other side,” Dr. Redfield said.
COVID-19: Convalescent plasma falls short in phase 2 trial
Convalescent plasma may not prevent progression to severe disease or reduce mortality risk in hospitalized patients with moderate COVID-19, based on a phase 2 trial involving more than 400 patients in India.
The PLACID trial offers real-world data with “high generalizability,” according to lead author Anup Agarwal, MD, of the Indian Council of Medical Research, New Delhi, and colleagues.
“Evidence suggests that convalescent plasma collected from survivors of COVID-19 contains receptor binding domain specific antibodies with potent antiviral activity,” the investigators wrote in the BMJ. “However, effective titers of antiviral neutralizing antibodies, optimal timing for convalescent plasma treatment, optimal timing for plasma donation, and the severity class of patients who are likely to benefit from convalescent plasma remain unclear.”
According to Dr. Agarwal and colleagues, case series and observational studies have suggested that convalescent plasma may reduce viral load, hospital stay, and mortality, but randomized controlled trials to date have ended prematurely because of issues with enrollment and design, making PLACID the first randomized controlled trial of its kind to reach completion.
The open-label, multicenter study involved 464 hospitalized adults who tested positive for SARS-CoV-2 via reverse transcription polymerase chain reaction (RT-PCR). Enrollment also required a respiratory rate of more than 24 breaths/min with an oxygen saturation (SpO2) of 93% or less on room air, or a partial pressure of oxygen in arterial blood/fraction of inspired oxygen (PaO2 /FiO2 ) ratio between 200 and 300 mm Hg.
Patients were randomly assigned in a 1:1 ratio to receive either best standard of care (control), or best standard of care plus convalescent plasma, which was given in two doses of 200 mL, 24 hours apart. Patients were assessed via clinical examination, chest imaging, and serial laboratory testing, the latter of which included neutralizing antibody titers on days 0, 3, and 7.
The primary outcome was a 28-day composite of progression to severe disease (PaO2/FiO2 ratio < 100 mm Hg) and all-cause mortality. An array of secondary outcomes were also reported, including symptom resolution, total duration of respiratory support, change in oxygen requirement, and others.
In the convalescent plasma group, 19% of patients progressed to severe disease or died within 28 days, compared with 18% of those in the control group (risk ratio, 1.04; 95% confidence interval, 0.71-1.54), suggesting no statistically significant benefit from the intervention. This lack of benefit was also found in a subgroup analysis of patients with detectable titers of antibodies to SARS-CoV-2, and when progression to severe disease and all-cause mortality were analyzed independently across all patients.
Still, at day 7, patients treated with convalescent plasma were significantly more likely to have resolution of fatigue (RR, 1.21; 95% CI, 1.02-1.42) and shortness of breath (RR, 1.16; 95% CI, 1.02-1.32). And at the same time point, patients treated with convalescent plasma were 20% more likely to test negative for SARS-CoV-2 RNA (RR, 1.2; 95% CI, 1.04-1.5).
In an accompanying editorial, Elizabeth B. Pathak, PhD, of the Women’s Institute for Independent Social Enquiry, Olney, Md., suggested that the reported symptom improvements need to be viewed with skepticism.
“These results should be interpreted with caution, because the trial was not blinded, so knowledge of treatment status could have influenced the reporting of subjective symptoms by patients who survived to day 7,” Dr. Pathak wrote.
Dr. Pathak noted that convalescent plasma did appear to have an antiviral effect, based on the higher rate of negative RNA test results at day 7. She hypothesized that the lack of major corresponding clinical benefit could be explained by detrimental thrombotic processes.
“The net effect of plasma is prothrombotic,” Dr. Pathak wrote, which should raise safety concerns, since “COVID-19 is a life-threatening thrombotic disorder.”
According to Dr. Pathak, large-scale datasets may be giving a false sense of security. She cited a recent safety analysis of 20,000 U.S. patients who received convalescent plasma, in which the investigators excluded 88.2% of cardiac events and 66.3% of thrombotic events, as these were deemed unrelated to transfusion; but this decision was made by the treating physician, without independent review or a defined protocol.
Michael J. Joyner, MD, of the Mayo Clinic in Rochester, Minn., was the lead author of the above safety study, and is leading the Food and Drug Administration expanded access program for convalescent plasma in patients with COVID-19. He suggested that the study by Dr. Agarwal and colleagues was admirable, but flaws in the treatment protocol cast doubt upon the efficacy findings.
“It is very impressive that these investigators performed a large trial of convalescent plasma in the midst of a pandemic,” Dr. Joyner said. “Unfortunately it is unclear how generalizable the findings are because many of the units of plasma had either very low or no antibody titers and because the plasma was given late in the course of the disease. It has been known since at least the 1930s that antibody therapy works best when enough product is given either prophylactically or early in the course of disease.”
Dr. Joyner had a more positive interpretation of the reported symptom improvements.
“It is also interesting to note that while there was no mortality benefit, that – even with the limitations of the study – there was some evidence of improved patient physiology at 7 days,” he said. “So, at one level, [this is] a negative study, but at least [there are] some hints of efficacy given the suboptimal use case in the patients studied.”
The study was funded by the Indian Council of Medical Research, which employs several of the authors and PLACID Trial Collaborators. Dr. Pathak and Dr. Joyner reported no conflicts of interest.
SOURCE: Agarwal A et al. BMJ. 2020 Oct 23. doi: 10.1136/bmj.m3939 .
Convalescent plasma may not prevent progression to severe disease or reduce mortality risk in hospitalized patients with moderate COVID-19, based on a phase 2 trial involving more than 400 patients in India.
The PLACID trial offers real-world data with “high generalizability,” according to lead author Anup Agarwal, MD, of the Indian Council of Medical Research, New Delhi, and colleagues.
“Evidence suggests that convalescent plasma collected from survivors of COVID-19 contains receptor binding domain specific antibodies with potent antiviral activity,” the investigators wrote in the BMJ. “However, effective titers of antiviral neutralizing antibodies, optimal timing for convalescent plasma treatment, optimal timing for plasma donation, and the severity class of patients who are likely to benefit from convalescent plasma remain unclear.”
According to Dr. Agarwal and colleagues, case series and observational studies have suggested that convalescent plasma may reduce viral load, hospital stay, and mortality, but randomized controlled trials to date have ended prematurely because of issues with enrollment and design, making PLACID the first randomized controlled trial of its kind to reach completion.
The open-label, multicenter study involved 464 hospitalized adults who tested positive for SARS-CoV-2 via reverse transcription polymerase chain reaction (RT-PCR). Enrollment also required a respiratory rate of more than 24 breaths/min with an oxygen saturation (SpO2) of 93% or less on room air, or a partial pressure of oxygen in arterial blood/fraction of inspired oxygen (PaO2 /FiO2 ) ratio between 200 and 300 mm Hg.
Patients were randomly assigned in a 1:1 ratio to receive either best standard of care (control), or best standard of care plus convalescent plasma, which was given in two doses of 200 mL, 24 hours apart. Patients were assessed via clinical examination, chest imaging, and serial laboratory testing, the latter of which included neutralizing antibody titers on days 0, 3, and 7.
The primary outcome was a 28-day composite of progression to severe disease (PaO2/FiO2 ratio < 100 mm Hg) and all-cause mortality. An array of secondary outcomes were also reported, including symptom resolution, total duration of respiratory support, change in oxygen requirement, and others.
In the convalescent plasma group, 19% of patients progressed to severe disease or died within 28 days, compared with 18% of those in the control group (risk ratio, 1.04; 95% confidence interval, 0.71-1.54), suggesting no statistically significant benefit from the intervention. This lack of benefit was also found in a subgroup analysis of patients with detectable titers of antibodies to SARS-CoV-2, and when progression to severe disease and all-cause mortality were analyzed independently across all patients.
Still, at day 7, patients treated with convalescent plasma were significantly more likely to have resolution of fatigue (RR, 1.21; 95% CI, 1.02-1.42) and shortness of breath (RR, 1.16; 95% CI, 1.02-1.32). And at the same time point, patients treated with convalescent plasma were 20% more likely to test negative for SARS-CoV-2 RNA (RR, 1.2; 95% CI, 1.04-1.5).
In an accompanying editorial, Elizabeth B. Pathak, PhD, of the Women’s Institute for Independent Social Enquiry, Olney, Md., suggested that the reported symptom improvements need to be viewed with skepticism.
“These results should be interpreted with caution, because the trial was not blinded, so knowledge of treatment status could have influenced the reporting of subjective symptoms by patients who survived to day 7,” Dr. Pathak wrote.
Dr. Pathak noted that convalescent plasma did appear to have an antiviral effect, based on the higher rate of negative RNA test results at day 7. She hypothesized that the lack of major corresponding clinical benefit could be explained by detrimental thrombotic processes.
“The net effect of plasma is prothrombotic,” Dr. Pathak wrote, which should raise safety concerns, since “COVID-19 is a life-threatening thrombotic disorder.”
According to Dr. Pathak, large-scale datasets may be giving a false sense of security. She cited a recent safety analysis of 20,000 U.S. patients who received convalescent plasma, in which the investigators excluded 88.2% of cardiac events and 66.3% of thrombotic events, as these were deemed unrelated to transfusion; but this decision was made by the treating physician, without independent review or a defined protocol.
Michael J. Joyner, MD, of the Mayo Clinic in Rochester, Minn., was the lead author of the above safety study, and is leading the Food and Drug Administration expanded access program for convalescent plasma in patients with COVID-19. He suggested that the study by Dr. Agarwal and colleagues was admirable, but flaws in the treatment protocol cast doubt upon the efficacy findings.
“It is very impressive that these investigators performed a large trial of convalescent plasma in the midst of a pandemic,” Dr. Joyner said. “Unfortunately it is unclear how generalizable the findings are because many of the units of plasma had either very low or no antibody titers and because the plasma was given late in the course of the disease. It has been known since at least the 1930s that antibody therapy works best when enough product is given either prophylactically or early in the course of disease.”
Dr. Joyner had a more positive interpretation of the reported symptom improvements.
“It is also interesting to note that while there was no mortality benefit, that – even with the limitations of the study – there was some evidence of improved patient physiology at 7 days,” he said. “So, at one level, [this is] a negative study, but at least [there are] some hints of efficacy given the suboptimal use case in the patients studied.”
The study was funded by the Indian Council of Medical Research, which employs several of the authors and PLACID Trial Collaborators. Dr. Pathak and Dr. Joyner reported no conflicts of interest.
SOURCE: Agarwal A et al. BMJ. 2020 Oct 23. doi: 10.1136/bmj.m3939 .
Convalescent plasma may not prevent progression to severe disease or reduce mortality risk in hospitalized patients with moderate COVID-19, based on a phase 2 trial involving more than 400 patients in India.
The PLACID trial offers real-world data with “high generalizability,” according to lead author Anup Agarwal, MD, of the Indian Council of Medical Research, New Delhi, and colleagues.
“Evidence suggests that convalescent plasma collected from survivors of COVID-19 contains receptor binding domain specific antibodies with potent antiviral activity,” the investigators wrote in the BMJ. “However, effective titers of antiviral neutralizing antibodies, optimal timing for convalescent plasma treatment, optimal timing for plasma donation, and the severity class of patients who are likely to benefit from convalescent plasma remain unclear.”
According to Dr. Agarwal and colleagues, case series and observational studies have suggested that convalescent plasma may reduce viral load, hospital stay, and mortality, but randomized controlled trials to date have ended prematurely because of issues with enrollment and design, making PLACID the first randomized controlled trial of its kind to reach completion.
The open-label, multicenter study involved 464 hospitalized adults who tested positive for SARS-CoV-2 via reverse transcription polymerase chain reaction (RT-PCR). Enrollment also required a respiratory rate of more than 24 breaths/min with an oxygen saturation (SpO2) of 93% or less on room air, or a partial pressure of oxygen in arterial blood/fraction of inspired oxygen (PaO2 /FiO2 ) ratio between 200 and 300 mm Hg.
Patients were randomly assigned in a 1:1 ratio to receive either best standard of care (control), or best standard of care plus convalescent plasma, which was given in two doses of 200 mL, 24 hours apart. Patients were assessed via clinical examination, chest imaging, and serial laboratory testing, the latter of which included neutralizing antibody titers on days 0, 3, and 7.
The primary outcome was a 28-day composite of progression to severe disease (PaO2/FiO2 ratio < 100 mm Hg) and all-cause mortality. An array of secondary outcomes were also reported, including symptom resolution, total duration of respiratory support, change in oxygen requirement, and others.
In the convalescent plasma group, 19% of patients progressed to severe disease or died within 28 days, compared with 18% of those in the control group (risk ratio, 1.04; 95% confidence interval, 0.71-1.54), suggesting no statistically significant benefit from the intervention. This lack of benefit was also found in a subgroup analysis of patients with detectable titers of antibodies to SARS-CoV-2, and when progression to severe disease and all-cause mortality were analyzed independently across all patients.
Still, at day 7, patients treated with convalescent plasma were significantly more likely to have resolution of fatigue (RR, 1.21; 95% CI, 1.02-1.42) and shortness of breath (RR, 1.16; 95% CI, 1.02-1.32). And at the same time point, patients treated with convalescent plasma were 20% more likely to test negative for SARS-CoV-2 RNA (RR, 1.2; 95% CI, 1.04-1.5).
In an accompanying editorial, Elizabeth B. Pathak, PhD, of the Women’s Institute for Independent Social Enquiry, Olney, Md., suggested that the reported symptom improvements need to be viewed with skepticism.
“These results should be interpreted with caution, because the trial was not blinded, so knowledge of treatment status could have influenced the reporting of subjective symptoms by patients who survived to day 7,” Dr. Pathak wrote.
Dr. Pathak noted that convalescent plasma did appear to have an antiviral effect, based on the higher rate of negative RNA test results at day 7. She hypothesized that the lack of major corresponding clinical benefit could be explained by detrimental thrombotic processes.
“The net effect of plasma is prothrombotic,” Dr. Pathak wrote, which should raise safety concerns, since “COVID-19 is a life-threatening thrombotic disorder.”
According to Dr. Pathak, large-scale datasets may be giving a false sense of security. She cited a recent safety analysis of 20,000 U.S. patients who received convalescent plasma, in which the investigators excluded 88.2% of cardiac events and 66.3% of thrombotic events, as these were deemed unrelated to transfusion; but this decision was made by the treating physician, without independent review or a defined protocol.
Michael J. Joyner, MD, of the Mayo Clinic in Rochester, Minn., was the lead author of the above safety study, and is leading the Food and Drug Administration expanded access program for convalescent plasma in patients with COVID-19. He suggested that the study by Dr. Agarwal and colleagues was admirable, but flaws in the treatment protocol cast doubt upon the efficacy findings.
“It is very impressive that these investigators performed a large trial of convalescent plasma in the midst of a pandemic,” Dr. Joyner said. “Unfortunately it is unclear how generalizable the findings are because many of the units of plasma had either very low or no antibody titers and because the plasma was given late in the course of the disease. It has been known since at least the 1930s that antibody therapy works best when enough product is given either prophylactically or early in the course of disease.”
Dr. Joyner had a more positive interpretation of the reported symptom improvements.
“It is also interesting to note that while there was no mortality benefit, that – even with the limitations of the study – there was some evidence of improved patient physiology at 7 days,” he said. “So, at one level, [this is] a negative study, but at least [there are] some hints of efficacy given the suboptimal use case in the patients studied.”
The study was funded by the Indian Council of Medical Research, which employs several of the authors and PLACID Trial Collaborators. Dr. Pathak and Dr. Joyner reported no conflicts of interest.
SOURCE: Agarwal A et al. BMJ. 2020 Oct 23. doi: 10.1136/bmj.m3939 .
FROM BMJ
FDA approves remdesivir, first treatment for COVID-19
making it the first and only approved treatment for COVID-19, according to a release from drug manufacturer Gilead Sciences.
The FDA’s initial Emergency Use Authorization (EUA) of the antiviral, issued in May, allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who needed oxygen therapy or mechanical ventilation.
An August EUA expanded treatment to include all adult and pediatric hospitalized COVID-19 patients, regardless of the severity of their disease. The FDA also issued a new EUA for remdesivir Oct. 22 allowing treatment of hospitalized pediatric patients younger than 12 weighing at least 3.5 kg.
Today’s approval is based on three randomized controlled trials, according to Gilead.
Final trial results from one of them, the National Institute of Allergy and Infectious Disease–funded ACTT-1 trial, published earlier in October, showed that hospitalized patients with COVID-19 who received remdesivir had a shorter median recovery time than those who received a placebo – 10 days versus 15 days.
This difference and some related secondary endpoints were statistically significant in the randomized trial, but there was not a statistically significant difference in mortality between the treatment and placebo groups.
The other two trials used for the approval, the SIMPLE trials, were open-label phase 3 trials conducted in countries with a high prevalence of COVID-19 infections, according to Gilead.
The SIMPLE-Severe trial was a randomized, multicenter study that evaluated the efficacy and safety of 5-day and 10-day dosing plus standard of care in 397 hospitalized adult patients with severe COVID-19. The primary endpoint was clinical status on day 14 assessed on a 7-point ordinal scale, according to Gilead.
The trial found that a 5-day or a 10-day treatment course of Veklury achieved similar clinical outcomes to the ACTT-1 trial (odds ratio, 0.75; 95% confidence interval, 0.51-1.12).
The SIMPLE-Moderate trial was a randomized, controlled, multicenter study that evaluated the efficacy and safety of 5-day and 10-day dosing durations of Veklury plus standard of care, compared with standard of care alone in 600 hospitalized adult patients with moderate COVID-19, Gilead stated in its release.
The primary endpoint was clinical status on day 11 assessed on a 7-point ordinal scale.
The results showed statistically improved clinical outcomes with a 5-day treatment course of Veklury, compared with standard of care (OR, 1.65; 95% CI, 1.0-2.48; P = .017), according to Gilead.
This article first appeared on Medscape.com.
making it the first and only approved treatment for COVID-19, according to a release from drug manufacturer Gilead Sciences.
The FDA’s initial Emergency Use Authorization (EUA) of the antiviral, issued in May, allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who needed oxygen therapy or mechanical ventilation.
An August EUA expanded treatment to include all adult and pediatric hospitalized COVID-19 patients, regardless of the severity of their disease. The FDA also issued a new EUA for remdesivir Oct. 22 allowing treatment of hospitalized pediatric patients younger than 12 weighing at least 3.5 kg.
Today’s approval is based on three randomized controlled trials, according to Gilead.
Final trial results from one of them, the National Institute of Allergy and Infectious Disease–funded ACTT-1 trial, published earlier in October, showed that hospitalized patients with COVID-19 who received remdesivir had a shorter median recovery time than those who received a placebo – 10 days versus 15 days.
This difference and some related secondary endpoints were statistically significant in the randomized trial, but there was not a statistically significant difference in mortality between the treatment and placebo groups.
The other two trials used for the approval, the SIMPLE trials, were open-label phase 3 trials conducted in countries with a high prevalence of COVID-19 infections, according to Gilead.
The SIMPLE-Severe trial was a randomized, multicenter study that evaluated the efficacy and safety of 5-day and 10-day dosing plus standard of care in 397 hospitalized adult patients with severe COVID-19. The primary endpoint was clinical status on day 14 assessed on a 7-point ordinal scale, according to Gilead.
The trial found that a 5-day or a 10-day treatment course of Veklury achieved similar clinical outcomes to the ACTT-1 trial (odds ratio, 0.75; 95% confidence interval, 0.51-1.12).
The SIMPLE-Moderate trial was a randomized, controlled, multicenter study that evaluated the efficacy and safety of 5-day and 10-day dosing durations of Veklury plus standard of care, compared with standard of care alone in 600 hospitalized adult patients with moderate COVID-19, Gilead stated in its release.
The primary endpoint was clinical status on day 11 assessed on a 7-point ordinal scale.
The results showed statistically improved clinical outcomes with a 5-day treatment course of Veklury, compared with standard of care (OR, 1.65; 95% CI, 1.0-2.48; P = .017), according to Gilead.
This article first appeared on Medscape.com.
making it the first and only approved treatment for COVID-19, according to a release from drug manufacturer Gilead Sciences.
The FDA’s initial Emergency Use Authorization (EUA) of the antiviral, issued in May, allowed the drug to be used only for patients with severe COVID-19, specifically, COVID-19 patients with low blood oxygen levels or who needed oxygen therapy or mechanical ventilation.
An August EUA expanded treatment to include all adult and pediatric hospitalized COVID-19 patients, regardless of the severity of their disease. The FDA also issued a new EUA for remdesivir Oct. 22 allowing treatment of hospitalized pediatric patients younger than 12 weighing at least 3.5 kg.
Today’s approval is based on three randomized controlled trials, according to Gilead.
Final trial results from one of them, the National Institute of Allergy and Infectious Disease–funded ACTT-1 trial, published earlier in October, showed that hospitalized patients with COVID-19 who received remdesivir had a shorter median recovery time than those who received a placebo – 10 days versus 15 days.
This difference and some related secondary endpoints were statistically significant in the randomized trial, but there was not a statistically significant difference in mortality between the treatment and placebo groups.
The other two trials used for the approval, the SIMPLE trials, were open-label phase 3 trials conducted in countries with a high prevalence of COVID-19 infections, according to Gilead.
The SIMPLE-Severe trial was a randomized, multicenter study that evaluated the efficacy and safety of 5-day and 10-day dosing plus standard of care in 397 hospitalized adult patients with severe COVID-19. The primary endpoint was clinical status on day 14 assessed on a 7-point ordinal scale, according to Gilead.
The trial found that a 5-day or a 10-day treatment course of Veklury achieved similar clinical outcomes to the ACTT-1 trial (odds ratio, 0.75; 95% confidence interval, 0.51-1.12).
The SIMPLE-Moderate trial was a randomized, controlled, multicenter study that evaluated the efficacy and safety of 5-day and 10-day dosing durations of Veklury plus standard of care, compared with standard of care alone in 600 hospitalized adult patients with moderate COVID-19, Gilead stated in its release.
The primary endpoint was clinical status on day 11 assessed on a 7-point ordinal scale.
The results showed statistically improved clinical outcomes with a 5-day treatment course of Veklury, compared with standard of care (OR, 1.65; 95% CI, 1.0-2.48; P = .017), according to Gilead.
This article first appeared on Medscape.com.