User login
Widespread Hyperkeratotic Papules in a Transplant Recipient
The Diagnosis: Trichodysplasia Spinulosa
Trichodysplasia spinulosa has been described in case reports over the last several decades, with its causative virus trichodysplasia spinulosa-associated polyomavirus (TSPyV) identified in 2010 by van der Meijden et al.1 Trichodysplasia spinulosa-associated polyomavirus is a small, nonenveloped, double-stranded DNA virus in the Polyomaviridae family, among several other known cutaneous polyomaviruses including Merkel cell polyomavirus, human polyomavirus (HPyV) 6, HPyV7, HPyV10, and possibly HPyV13.2 The primary target of TSPyV is follicular keratinocytes, and it is believed to cause trichodysplasia spinulosa by primary infection rather than by reactivation. Trichodysplasia spinulosa presents in immunosuppressed patients as a folliculocentric eruption of papules with keratinous spines on the face, often with concurrent alopecia, eventually spreading to the trunk and extremities.3 The diagnosis often is clinical, but a biopsy may be performed for histopathologic confirmation. Alternatively, lesional spicules can be painlessly collected manually and submitted for viral polymerase chain reaction (PCR).4 The diagnosis of trichodysplasia spinulosa can be difficult due to similarities with other more common conditions such as keratosis pilaris, milia, filiform warts, or lichen spinulosus.
Similar to trichodysplasia spinulosa, keratosis pilaris also presents with folliculocentric and often erythematous papules.5 Keratosis pilaris most frequently affects the posterior upper arms and thighs but also may affect the cheeks, as seen in trichodysplasia spinulosa. Differentiation between the 2 diagnoses can be made on a clinical basis, as keratosis pilaris lacks the characteristic keratinous spines and often spares the central face and nose, locations that commonly are affected in trichodysplasia spinulosa.3
Milia typically appear as white to yellow papules, often on the cheeks, eyelids, nose, and chin.6 Given their predilection for the face, milia can appear similarly to trichodysplasia spinulosa. Differentiation can be made clinically, as milia typically are not as numerous as the spiculed papules seen in trichodysplasia spinulosa. Morphologically, milia will present as smooth, dome-shaped papules as opposed to the keratinous spicules seen in trichodysplasia spinulosa. The diagnosis of milia can be confirmed by incision and removal of the white chalky keratin core, a feature absent in trichodysplasia spinulosa.
Filiform warts are benign epidermal proliferations caused by human papillomavirus infection that manifest as flesh-colored, verrucous, hyperkeratotic papules.7 They can appear on virtually any skin surface, including the face, and thus may be mistaken for trichodysplasia spinulosa. Close inspection usually will reveal tiny black dots that represent thrombosed capillaries, a feature lacking in trichodysplasia spinulosa. In long-standing lesions or immunocompromised patients, confluent verrucous plaques may develop.8 Diagnosis of filiform warts can be confirmed with biopsy, which will demonstrate a compact stratum corneum, coarse hypergranulosis, and papillomatosis curving inward, while biopsy of a trichodysplasia spinulosa lesion would show polyomavirus infection of the hair follicle and characteristic eosinophilic inclusion bodies.9
Lichen spinulosus may appear as multiple folliculocentric scaly papules with hairlike horny spines.10 Lichen spinulosus differs from trichodysplasia spinulosa in that it commonly appears on the neck, abdomen, trochanteric region, arms, elbows, or knees. Lichen spinulosus also classically appears as a concrete cluster of papules, often localized to a certain region, in contrast to trichodysplasia spinulosa, which will be widespread, often spreading over time. Finally, clinical history may help differentiate the 2 entities. Lichen spinulosus most often appears in children and adolescents and often has an indolent course, typically resolving during puberty, while trichodysplasia spinulosa is seen in immunocompromised patients.
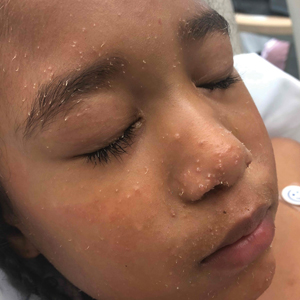
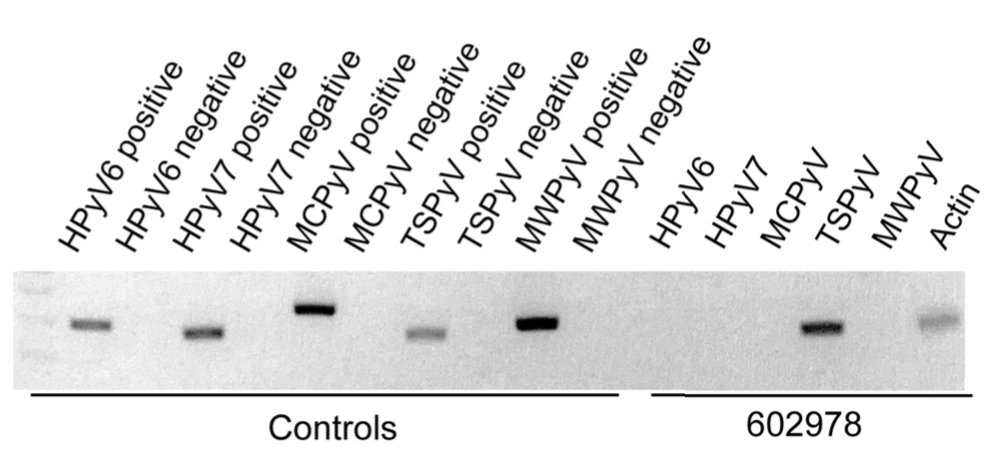
In our patient, the dermatology team made a diagnosis of trichodysplasia spinulosa based on the characteristic clinical presentation, which was confirmed after approximately 10 lesional spicules were removed by tissue forceps and submitted for PCR analysis showing TSPyV (Figure). Two other cases utilized spicule PCR analysis for confirmation of TSPyV.11,12 This technique may represent a viable option for diagnostic confirmation in pediatric cases.
Although some articles have examined the molecular and biologic features of trichodysplasia spinulosa, literature on clinical presentation and management is limited to isolated case reports with no comprehensive studies to establish a standardized treatment. Of these reports, oral valganciclovir 900 mg daily, topical retinoids, cidofovir cream 1% to 3%, and decreasing or altering the immunosuppressive regimen all have been noted to provide clinical improvement.13,14 Other therapies including leflunomide and routine manual extraction of spicules also have shown effectiveness in the treatment of trichodysplasia spinulosa.15
In our patient, treatment included decreasing immunosuppression, as she was getting recurrent sinus and upper respiratory infections. Mycophenolate mofetil was discontinued, and the patient was continued solely on tacrolimus therapy. She demonstrated notable improvement after 3 months, with approximately 50% clearance of the eruption. A mutual decision was made at that visit to initiate therapy with compounded cidofovir cream 1% daily to the lesions until the next follow-up visit. Unfortunately, the patient did not return for her scheduled dermatology visits and was lost to long-term follow-up.
Acknowledgment
We thank Richard C. Wang, MD, PhD (Dallas, Texas), for his dermatologic expertise and assistance in analysis of lesional samples for TSPyV.
- van der Meijden E, Janssens RWA, Lauber C, et al. Discovery of a new human polyomavirus associated with trichodysplasia spinulosa in an immunocompromised patient. PLoS Pathog. 2010;6:E1001024.
- Sheu JC, Tran J, Rady PL, et al. Polyomaviruses of the skin: integrating molecular and clinical advances in an emerging class of viruses. Br J Dermatol. 2019;180:1302-1311.
- Sperling LC, Tomaszewski MM, Thomas DA. Viral-associated trichodysplasia in patients who are immunocompromised. J Am Acad Dermatol. 2004;50:318-322.
- Wu JH, Nguyen HP, Rady PL, et al. Molecular insight into the viral biology and clinical features of trichodysplasia spinulosa. Br J Dermatol. 2016;174:490-498.
- Hwang S, Schwartz RA. Keratosis pilaris: a common follicular hyperkeratosis. Cutis. 2008;82:177-180.
- Berk DR, Bayliss SJ. Milia: a review and classification. J Am Acad Dermatol. 2008;59:1050-1063.
- Micali G, Dall'Oglio F, Nasca MR, et al. Management of cutaneous warts: an evidence-based approach. Am J Clin Dermatol. 2004;5:311-317.
- Bolognia J, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018.
- Elston DM, Ferringer T, Ko CJ. Dermatopathology. 3rd ed. Elsevier; 2018.
- Tilly JJ, Drolet BA, Esterly NB. Lichenoid eruptions in children. J Am Acad Dermatol. 2004;51:606-624.
- Chamseddin BH, Tran BAPD, Lee EE, et al. Trichodysplasia spinulosa in a child: identification of trichodysplasia spinulosa-associated polyomavirus in skin, serum, and urine. Pediatr Dermatol. 2019;36:723-724.
- Sonstegard A, Grossman M, Garg A. Trichodysplasia spinulosa in a kidney transplant recipient. JAMA Dermatol. 2021;157:105.
- Leitenberger JJ, Abdelmalek M, Wang RC, et al. Two cases of trichodysplasia spinulosa responsive to compounded topical cidofovir 3% cream. JAAD Case Rep. 2015;1:S33-S35.
- DeCrescenzo AJ, Philips RC, Wilkerson MG. Trichodysplasia spinulosa: a rare complication of immunosuppression. JAAD Case Rep. 2016;2:307-309.
- Nguyen KD, Chamseddin BH, Cockerell CJ, et al. The biology and clinical features of cutaneous polyomaviruses. J Invest Dermatol. 2019;139:285-292.
The Diagnosis: Trichodysplasia Spinulosa
Trichodysplasia spinulosa has been described in case reports over the last several decades, with its causative virus trichodysplasia spinulosa-associated polyomavirus (TSPyV) identified in 2010 by van der Meijden et al.1 Trichodysplasia spinulosa-associated polyomavirus is a small, nonenveloped, double-stranded DNA virus in the Polyomaviridae family, among several other known cutaneous polyomaviruses including Merkel cell polyomavirus, human polyomavirus (HPyV) 6, HPyV7, HPyV10, and possibly HPyV13.2 The primary target of TSPyV is follicular keratinocytes, and it is believed to cause trichodysplasia spinulosa by primary infection rather than by reactivation. Trichodysplasia spinulosa presents in immunosuppressed patients as a folliculocentric eruption of papules with keratinous spines on the face, often with concurrent alopecia, eventually spreading to the trunk and extremities.3 The diagnosis often is clinical, but a biopsy may be performed for histopathologic confirmation. Alternatively, lesional spicules can be painlessly collected manually and submitted for viral polymerase chain reaction (PCR).4 The diagnosis of trichodysplasia spinulosa can be difficult due to similarities with other more common conditions such as keratosis pilaris, milia, filiform warts, or lichen spinulosus.
Similar to trichodysplasia spinulosa, keratosis pilaris also presents with folliculocentric and often erythematous papules.5 Keratosis pilaris most frequently affects the posterior upper arms and thighs but also may affect the cheeks, as seen in trichodysplasia spinulosa. Differentiation between the 2 diagnoses can be made on a clinical basis, as keratosis pilaris lacks the characteristic keratinous spines and often spares the central face and nose, locations that commonly are affected in trichodysplasia spinulosa.3
Milia typically appear as white to yellow papules, often on the cheeks, eyelids, nose, and chin.6 Given their predilection for the face, milia can appear similarly to trichodysplasia spinulosa. Differentiation can be made clinically, as milia typically are not as numerous as the spiculed papules seen in trichodysplasia spinulosa. Morphologically, milia will present as smooth, dome-shaped papules as opposed to the keratinous spicules seen in trichodysplasia spinulosa. The diagnosis of milia can be confirmed by incision and removal of the white chalky keratin core, a feature absent in trichodysplasia spinulosa.
Filiform warts are benign epidermal proliferations caused by human papillomavirus infection that manifest as flesh-colored, verrucous, hyperkeratotic papules.7 They can appear on virtually any skin surface, including the face, and thus may be mistaken for trichodysplasia spinulosa. Close inspection usually will reveal tiny black dots that represent thrombosed capillaries, a feature lacking in trichodysplasia spinulosa. In long-standing lesions or immunocompromised patients, confluent verrucous plaques may develop.8 Diagnosis of filiform warts can be confirmed with biopsy, which will demonstrate a compact stratum corneum, coarse hypergranulosis, and papillomatosis curving inward, while biopsy of a trichodysplasia spinulosa lesion would show polyomavirus infection of the hair follicle and characteristic eosinophilic inclusion bodies.9
Lichen spinulosus may appear as multiple folliculocentric scaly papules with hairlike horny spines.10 Lichen spinulosus differs from trichodysplasia spinulosa in that it commonly appears on the neck, abdomen, trochanteric region, arms, elbows, or knees. Lichen spinulosus also classically appears as a concrete cluster of papules, often localized to a certain region, in contrast to trichodysplasia spinulosa, which will be widespread, often spreading over time. Finally, clinical history may help differentiate the 2 entities. Lichen spinulosus most often appears in children and adolescents and often has an indolent course, typically resolving during puberty, while trichodysplasia spinulosa is seen in immunocompromised patients.
In our patient, the dermatology team made a diagnosis of trichodysplasia spinulosa based on the characteristic clinical presentation, which was confirmed after approximately 10 lesional spicules were removed by tissue forceps and submitted for PCR analysis showing TSPyV (Figure). Two other cases utilized spicule PCR analysis for confirmation of TSPyV.11,12 This technique may represent a viable option for diagnostic confirmation in pediatric cases.

Although some articles have examined the molecular and biologic features of trichodysplasia spinulosa, literature on clinical presentation and management is limited to isolated case reports with no comprehensive studies to establish a standardized treatment. Of these reports, oral valganciclovir 900 mg daily, topical retinoids, cidofovir cream 1% to 3%, and decreasing or altering the immunosuppressive regimen all have been noted to provide clinical improvement.13,14 Other therapies including leflunomide and routine manual extraction of spicules also have shown effectiveness in the treatment of trichodysplasia spinulosa.15
In our patient, treatment included decreasing immunosuppression, as she was getting recurrent sinus and upper respiratory infections. Mycophenolate mofetil was discontinued, and the patient was continued solely on tacrolimus therapy. She demonstrated notable improvement after 3 months, with approximately 50% clearance of the eruption. A mutual decision was made at that visit to initiate therapy with compounded cidofovir cream 1% daily to the lesions until the next follow-up visit. Unfortunately, the patient did not return for her scheduled dermatology visits and was lost to long-term follow-up.
Acknowledgment
We thank Richard C. Wang, MD, PhD (Dallas, Texas), for his dermatologic expertise and assistance in analysis of lesional samples for TSPyV.
The Diagnosis: Trichodysplasia Spinulosa
Trichodysplasia spinulosa has been described in case reports over the last several decades, with its causative virus trichodysplasia spinulosa-associated polyomavirus (TSPyV) identified in 2010 by van der Meijden et al.1 Trichodysplasia spinulosa-associated polyomavirus is a small, nonenveloped, double-stranded DNA virus in the Polyomaviridae family, among several other known cutaneous polyomaviruses including Merkel cell polyomavirus, human polyomavirus (HPyV) 6, HPyV7, HPyV10, and possibly HPyV13.2 The primary target of TSPyV is follicular keratinocytes, and it is believed to cause trichodysplasia spinulosa by primary infection rather than by reactivation. Trichodysplasia spinulosa presents in immunosuppressed patients as a folliculocentric eruption of papules with keratinous spines on the face, often with concurrent alopecia, eventually spreading to the trunk and extremities.3 The diagnosis often is clinical, but a biopsy may be performed for histopathologic confirmation. Alternatively, lesional spicules can be painlessly collected manually and submitted for viral polymerase chain reaction (PCR).4 The diagnosis of trichodysplasia spinulosa can be difficult due to similarities with other more common conditions such as keratosis pilaris, milia, filiform warts, or lichen spinulosus.
Similar to trichodysplasia spinulosa, keratosis pilaris also presents with folliculocentric and often erythematous papules.5 Keratosis pilaris most frequently affects the posterior upper arms and thighs but also may affect the cheeks, as seen in trichodysplasia spinulosa. Differentiation between the 2 diagnoses can be made on a clinical basis, as keratosis pilaris lacks the characteristic keratinous spines and often spares the central face and nose, locations that commonly are affected in trichodysplasia spinulosa.3
Milia typically appear as white to yellow papules, often on the cheeks, eyelids, nose, and chin.6 Given their predilection for the face, milia can appear similarly to trichodysplasia spinulosa. Differentiation can be made clinically, as milia typically are not as numerous as the spiculed papules seen in trichodysplasia spinulosa. Morphologically, milia will present as smooth, dome-shaped papules as opposed to the keratinous spicules seen in trichodysplasia spinulosa. The diagnosis of milia can be confirmed by incision and removal of the white chalky keratin core, a feature absent in trichodysplasia spinulosa.
Filiform warts are benign epidermal proliferations caused by human papillomavirus infection that manifest as flesh-colored, verrucous, hyperkeratotic papules.7 They can appear on virtually any skin surface, including the face, and thus may be mistaken for trichodysplasia spinulosa. Close inspection usually will reveal tiny black dots that represent thrombosed capillaries, a feature lacking in trichodysplasia spinulosa. In long-standing lesions or immunocompromised patients, confluent verrucous plaques may develop.8 Diagnosis of filiform warts can be confirmed with biopsy, which will demonstrate a compact stratum corneum, coarse hypergranulosis, and papillomatosis curving inward, while biopsy of a trichodysplasia spinulosa lesion would show polyomavirus infection of the hair follicle and characteristic eosinophilic inclusion bodies.9
Lichen spinulosus may appear as multiple folliculocentric scaly papules with hairlike horny spines.10 Lichen spinulosus differs from trichodysplasia spinulosa in that it commonly appears on the neck, abdomen, trochanteric region, arms, elbows, or knees. Lichen spinulosus also classically appears as a concrete cluster of papules, often localized to a certain region, in contrast to trichodysplasia spinulosa, which will be widespread, often spreading over time. Finally, clinical history may help differentiate the 2 entities. Lichen spinulosus most often appears in children and adolescents and often has an indolent course, typically resolving during puberty, while trichodysplasia spinulosa is seen in immunocompromised patients.
In our patient, the dermatology team made a diagnosis of trichodysplasia spinulosa based on the characteristic clinical presentation, which was confirmed after approximately 10 lesional spicules were removed by tissue forceps and submitted for PCR analysis showing TSPyV (Figure). Two other cases utilized spicule PCR analysis for confirmation of TSPyV.11,12 This technique may represent a viable option for diagnostic confirmation in pediatric cases.

Although some articles have examined the molecular and biologic features of trichodysplasia spinulosa, literature on clinical presentation and management is limited to isolated case reports with no comprehensive studies to establish a standardized treatment. Of these reports, oral valganciclovir 900 mg daily, topical retinoids, cidofovir cream 1% to 3%, and decreasing or altering the immunosuppressive regimen all have been noted to provide clinical improvement.13,14 Other therapies including leflunomide and routine manual extraction of spicules also have shown effectiveness in the treatment of trichodysplasia spinulosa.15
In our patient, treatment included decreasing immunosuppression, as she was getting recurrent sinus and upper respiratory infections. Mycophenolate mofetil was discontinued, and the patient was continued solely on tacrolimus therapy. She demonstrated notable improvement after 3 months, with approximately 50% clearance of the eruption. A mutual decision was made at that visit to initiate therapy with compounded cidofovir cream 1% daily to the lesions until the next follow-up visit. Unfortunately, the patient did not return for her scheduled dermatology visits and was lost to long-term follow-up.
Acknowledgment
We thank Richard C. Wang, MD, PhD (Dallas, Texas), for his dermatologic expertise and assistance in analysis of lesional samples for TSPyV.
- van der Meijden E, Janssens RWA, Lauber C, et al. Discovery of a new human polyomavirus associated with trichodysplasia spinulosa in an immunocompromised patient. PLoS Pathog. 2010;6:E1001024.
- Sheu JC, Tran J, Rady PL, et al. Polyomaviruses of the skin: integrating molecular and clinical advances in an emerging class of viruses. Br J Dermatol. 2019;180:1302-1311.
- Sperling LC, Tomaszewski MM, Thomas DA. Viral-associated trichodysplasia in patients who are immunocompromised. J Am Acad Dermatol. 2004;50:318-322.
- Wu JH, Nguyen HP, Rady PL, et al. Molecular insight into the viral biology and clinical features of trichodysplasia spinulosa. Br J Dermatol. 2016;174:490-498.
- Hwang S, Schwartz RA. Keratosis pilaris: a common follicular hyperkeratosis. Cutis. 2008;82:177-180.
- Berk DR, Bayliss SJ. Milia: a review and classification. J Am Acad Dermatol. 2008;59:1050-1063.
- Micali G, Dall'Oglio F, Nasca MR, et al. Management of cutaneous warts: an evidence-based approach. Am J Clin Dermatol. 2004;5:311-317.
- Bolognia J, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018.
- Elston DM, Ferringer T, Ko CJ. Dermatopathology. 3rd ed. Elsevier; 2018.
- Tilly JJ, Drolet BA, Esterly NB. Lichenoid eruptions in children. J Am Acad Dermatol. 2004;51:606-624.
- Chamseddin BH, Tran BAPD, Lee EE, et al. Trichodysplasia spinulosa in a child: identification of trichodysplasia spinulosa-associated polyomavirus in skin, serum, and urine. Pediatr Dermatol. 2019;36:723-724.
- Sonstegard A, Grossman M, Garg A. Trichodysplasia spinulosa in a kidney transplant recipient. JAMA Dermatol. 2021;157:105.
- Leitenberger JJ, Abdelmalek M, Wang RC, et al. Two cases of trichodysplasia spinulosa responsive to compounded topical cidofovir 3% cream. JAAD Case Rep. 2015;1:S33-S35.
- DeCrescenzo AJ, Philips RC, Wilkerson MG. Trichodysplasia spinulosa: a rare complication of immunosuppression. JAAD Case Rep. 2016;2:307-309.
- Nguyen KD, Chamseddin BH, Cockerell CJ, et al. The biology and clinical features of cutaneous polyomaviruses. J Invest Dermatol. 2019;139:285-292.
- van der Meijden E, Janssens RWA, Lauber C, et al. Discovery of a new human polyomavirus associated with trichodysplasia spinulosa in an immunocompromised patient. PLoS Pathog. 2010;6:E1001024.
- Sheu JC, Tran J, Rady PL, et al. Polyomaviruses of the skin: integrating molecular and clinical advances in an emerging class of viruses. Br J Dermatol. 2019;180:1302-1311.
- Sperling LC, Tomaszewski MM, Thomas DA. Viral-associated trichodysplasia in patients who are immunocompromised. J Am Acad Dermatol. 2004;50:318-322.
- Wu JH, Nguyen HP, Rady PL, et al. Molecular insight into the viral biology and clinical features of trichodysplasia spinulosa. Br J Dermatol. 2016;174:490-498.
- Hwang S, Schwartz RA. Keratosis pilaris: a common follicular hyperkeratosis. Cutis. 2008;82:177-180.
- Berk DR, Bayliss SJ. Milia: a review and classification. J Am Acad Dermatol. 2008;59:1050-1063.
- Micali G, Dall'Oglio F, Nasca MR, et al. Management of cutaneous warts: an evidence-based approach. Am J Clin Dermatol. 2004;5:311-317.
- Bolognia J, Schaffer JV, Cerroni L. Dermatology. 4th ed. Elsevier; 2018.
- Elston DM, Ferringer T, Ko CJ. Dermatopathology. 3rd ed. Elsevier; 2018.
- Tilly JJ, Drolet BA, Esterly NB. Lichenoid eruptions in children. J Am Acad Dermatol. 2004;51:606-624.
- Chamseddin BH, Tran BAPD, Lee EE, et al. Trichodysplasia spinulosa in a child: identification of trichodysplasia spinulosa-associated polyomavirus in skin, serum, and urine. Pediatr Dermatol. 2019;36:723-724.
- Sonstegard A, Grossman M, Garg A. Trichodysplasia spinulosa in a kidney transplant recipient. JAMA Dermatol. 2021;157:105.
- Leitenberger JJ, Abdelmalek M, Wang RC, et al. Two cases of trichodysplasia spinulosa responsive to compounded topical cidofovir 3% cream. JAAD Case Rep. 2015;1:S33-S35.
- DeCrescenzo AJ, Philips RC, Wilkerson MG. Trichodysplasia spinulosa: a rare complication of immunosuppression. JAAD Case Rep. 2016;2:307-309.
- Nguyen KD, Chamseddin BH, Cockerell CJ, et al. The biology and clinical features of cutaneous polyomaviruses. J Invest Dermatol. 2019;139:285-292.
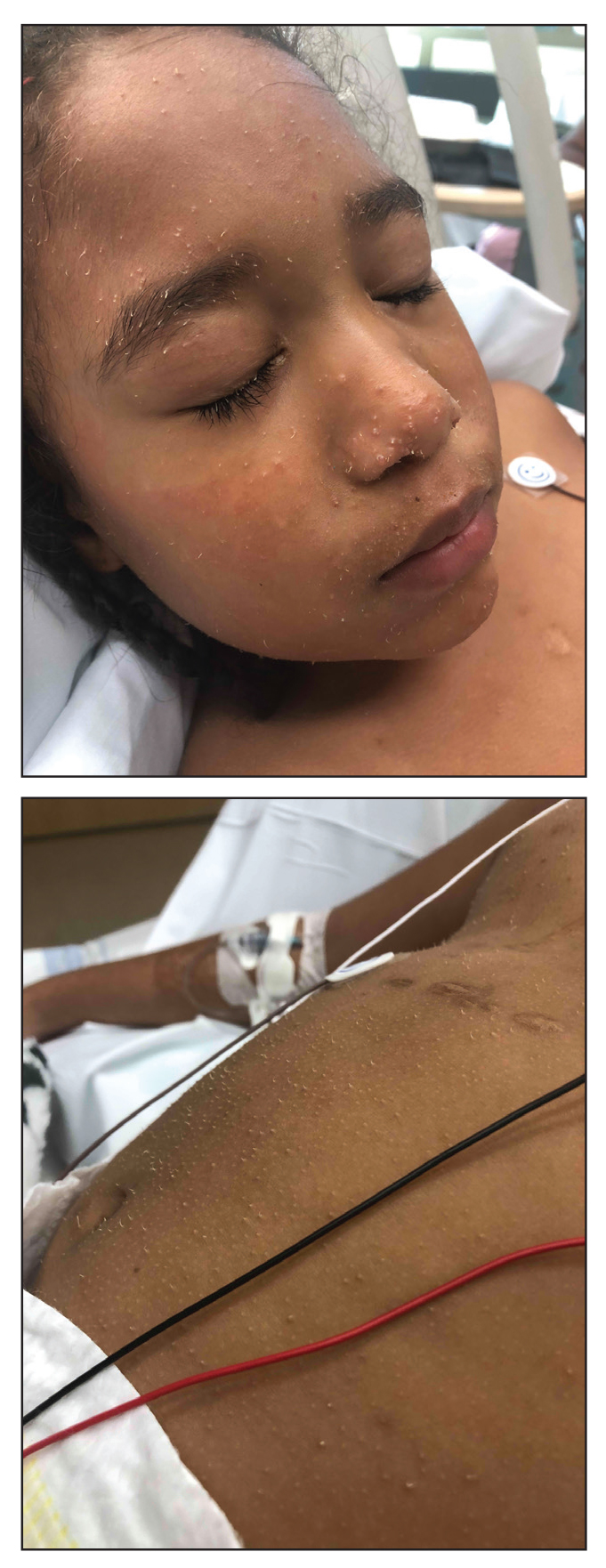
A 4-year-old girl with a history of cardiac transplantation 1 year prior for dilated cardiomyopathy presented to the dermatology consultation service with widespread hyperkeratotic papules of 2 months’ duration. The eruption initially had appeared on the face with subsequent involvement of the trunk and extremities. Her immunosuppressive medications included oral tacrolimus and mycophenolate mofetil. No over-the-counter or prescription treatments had been used for the eruption; the patient’s mother had been manually extracting the spicules from the nose, cheeks, and forehead with tweezers. The lesions were asymptomatic with only mild follicular erythema. Physical examination revealed multiple folliculocentric keratinous spicules on the nose, cheeks, forehead (top), trunk (bottom), arms, and legs.
How to improve our response to COVID’s mental tolls
We have no way of precisely knowing how many lives might have been saved, and how much grief and loneliness spared and economic ruin contained during COVID-19 if we had risen to its myriad challenges in a timely fashion. However, I feel we can safely say that the United States deserves to be graded with an “F” for its management of the pandemic.
To render this grade, we need only to read the countless verified reports of how critically needed public health measures were not taken soon enough, or sufficiently, to substantially mitigate human and societal suffering.
This began with the failure to protect doctors, nurses, and technicians, who did not have the personal protective equipment needed to prevent infection and spare risk to their loved ones. It soon extended to the country’s failure to adequately protect all its citizens and residents. COVID-19 then rained its grievous consequences disproportionately upon people of color, those living in poverty, and those with housing and food insecurity – those already greatly foreclosed from opportunities to exit from their circumstances.
We all have heard, “Fool me once, shame on you; fool me twice, shame on me.”
Bear witness, colleagues and friends: It will be our shared shame if we too continue to fail in our response to COVID-19. But failure need not happen because protecting ourselves and our country is a solvable problem; complex and demanding for sure, but solvable.
To battle trauma, we must first define it
The sine qua non of a disaster is its psychic and social trauma. I asked Maureen Sayres Van Niel, MD, chair of the American Psychiatric Association’s Minority and Underrepresented Caucus and a former steering committee member of the U.S. Preventive Services Task Force, to define trauma. She said, “It is [the product of] a catastrophic, unexpected event over which we have little control, with grave consequences to the lives and psychological functioning of those individuals and groups affected.”
The COVID-19 pandemic is a massively amplified traumatic event because of the virulence and contagious properties of the virus and its variants; the absence of end date on the horizon; its effect as a proverbial ax that disproportionately falls on the majority of the populace experiencing racial and social inequities; and the ironic yet necessary imperative to distance ourselves from those we care about and who care about us.
Four interdependent factors drive the magnitude of the traumatic impact of a disaster: the degree of exposure to the life-threatening event; the duration and threat of recurrence; an individual’s preexisting (natural and human-made) trauma and mental and addictive disorders; and the adequacy of family and fundamental resources such as housing, food, safety, and access to health care (the social dimensions of health and mental health). These factors underline the “who,” “what,” “where,” and “how” of what should have been (and continue to be) an effective public health response to the COVID-19 pandemic.
Yet existing categories that we have used to predict risk for trauma no longer hold. The gravity, prevalence, and persistence of COVID-19’s horrors erase any differences among victims, witnesses, and bystanders. Dr Sayres Van Niel asserts that we have a “collective, national trauma.” In April, the Kaiser Family Foundation’s Vaccine Monitor reported that 24% of U.S. adults had a close friend or family member who died of COVID-19. That’s 82 million Americans! Our country has eclipsed individual victimization and trauma because we are all in its maw.
Vital lessons from the past
In a previous column, I described my role as New York City’s mental health commissioner after 9/11 and the many lessons we learned during that multiyear process. Our work served as a template for other disasters to follow, such as Hurricane Sandy. Its value to COVID-19 is equally apparent.
We learned that those most at risk of developing symptomatic, functionally impairing mental illness had prior traumatic experiences (for example, from childhood abuse or neglect, violence, war, and forced displacement from their native land) and/or a preexisting mental or substance use disorder.
Once these individuals and communities were identified, we could prioritize their treatment and care. Doing so required mobilizing both inner and external (social) resources, which can be used before disaster strikes or in its wake.
For individuals, adaptive resources include developing any of a number of mind-body activities (for example, meditation, mindfulness, slow breathing, and yoga); sufficient but not necessarily excessive levels of exercise (as has been said, if exercise were a pill, it would be the most potent of medicines); nourishing diets; sleep, nature’s restorative state; and perhaps most important, attachment and human connection to people who care about you and whom you care about and trust.
This does not necessarily mean holding or following an institutional religion or belonging to house of worship (though, of course, that melds and augments faith with community). For a great many, myself included, there is spirituality, the belief in a greater power, which need not be a God yet instills a sense of the vastness, universality, and continuity of life.
For communities, adaptive resources include safe homes and neighborhoods; diminishing housing and food insecurity; education, including pre-K; employment, with a livable wage; ridding human interactions of the endless, so-called microaggressions (which are not micro at all, because they accrue) of race, ethnic, class, and age discrimination and injustice; and ready access to quality and affordable health care, now more than ever for the rising tide of mental and substance use disorders that COVID-19 has unleashed.
Every gain we make to ablate racism, social injustice, discrimination, and widely and deeply spread resource and opportunity inequities means more cohesion among the members of our collective tribe. Greater cohesion, a love for thy neighbor, and equity (in action, not polemics) will fuel the resilience we will need to withstand more of COVID-19’s ongoing trauma; that of other, inescapable disasters and losses; and the wear and tear of everyday life. The rewards of equity are priceless and include the dignity that derives from fairness and justice – given and received.
An unprecedented disaster requires a bold response
My, what a list. But to me, the encompassing nature of what’s needed means that we can make differences anywhere, everywhere, and in countless and continuous ways.
The measure of any society is in how it cares for those who are foreclosed, through no fault of their own, from what we all want: a life safe from violence, secure in housing and food, with loving relationships and the pride that comes of making contributions, each in our own, wonderfully unique way.
Where will we all be in a year, 2, or 3 from now? Prepared, or not? Emotionally inoculated, or not? Better equipped, or not? As divided, or more cohesive?
Well, I imagine that depends on each and every one of us.
Lloyd I. Sederer, MD, is a psychiatrist, public health doctor, and writer. He is an adjunct professor at the Columbia University School of Public Health, director of Columbia Psychiatry Media, chief medical officer of Bongo Media, and chair of the advisory board of Get Help. He has been chief medical officer of McLean Hospital, a Harvard teaching hospital; mental health commissioner of New York City (in the Bloomberg administration); and chief medical officer of the New York State Office of Mental Health, the nation’s largest state mental health agency.
A version of this article first appeared on Medscape.com.
We have no way of precisely knowing how many lives might have been saved, and how much grief and loneliness spared and economic ruin contained during COVID-19 if we had risen to its myriad challenges in a timely fashion. However, I feel we can safely say that the United States deserves to be graded with an “F” for its management of the pandemic.
To render this grade, we need only to read the countless verified reports of how critically needed public health measures were not taken soon enough, or sufficiently, to substantially mitigate human and societal suffering.
This began with the failure to protect doctors, nurses, and technicians, who did not have the personal protective equipment needed to prevent infection and spare risk to their loved ones. It soon extended to the country’s failure to adequately protect all its citizens and residents. COVID-19 then rained its grievous consequences disproportionately upon people of color, those living in poverty, and those with housing and food insecurity – those already greatly foreclosed from opportunities to exit from their circumstances.
We all have heard, “Fool me once, shame on you; fool me twice, shame on me.”
Bear witness, colleagues and friends: It will be our shared shame if we too continue to fail in our response to COVID-19. But failure need not happen because protecting ourselves and our country is a solvable problem; complex and demanding for sure, but solvable.
To battle trauma, we must first define it
The sine qua non of a disaster is its psychic and social trauma. I asked Maureen Sayres Van Niel, MD, chair of the American Psychiatric Association’s Minority and Underrepresented Caucus and a former steering committee member of the U.S. Preventive Services Task Force, to define trauma. She said, “It is [the product of] a catastrophic, unexpected event over which we have little control, with grave consequences to the lives and psychological functioning of those individuals and groups affected.”
The COVID-19 pandemic is a massively amplified traumatic event because of the virulence and contagious properties of the virus and its variants; the absence of end date on the horizon; its effect as a proverbial ax that disproportionately falls on the majority of the populace experiencing racial and social inequities; and the ironic yet necessary imperative to distance ourselves from those we care about and who care about us.
Four interdependent factors drive the magnitude of the traumatic impact of a disaster: the degree of exposure to the life-threatening event; the duration and threat of recurrence; an individual’s preexisting (natural and human-made) trauma and mental and addictive disorders; and the adequacy of family and fundamental resources such as housing, food, safety, and access to health care (the social dimensions of health and mental health). These factors underline the “who,” “what,” “where,” and “how” of what should have been (and continue to be) an effective public health response to the COVID-19 pandemic.
Yet existing categories that we have used to predict risk for trauma no longer hold. The gravity, prevalence, and persistence of COVID-19’s horrors erase any differences among victims, witnesses, and bystanders. Dr Sayres Van Niel asserts that we have a “collective, national trauma.” In April, the Kaiser Family Foundation’s Vaccine Monitor reported that 24% of U.S. adults had a close friend or family member who died of COVID-19. That’s 82 million Americans! Our country has eclipsed individual victimization and trauma because we are all in its maw.
Vital lessons from the past
In a previous column, I described my role as New York City’s mental health commissioner after 9/11 and the many lessons we learned during that multiyear process. Our work served as a template for other disasters to follow, such as Hurricane Sandy. Its value to COVID-19 is equally apparent.
We learned that those most at risk of developing symptomatic, functionally impairing mental illness had prior traumatic experiences (for example, from childhood abuse or neglect, violence, war, and forced displacement from their native land) and/or a preexisting mental or substance use disorder.
Once these individuals and communities were identified, we could prioritize their treatment and care. Doing so required mobilizing both inner and external (social) resources, which can be used before disaster strikes or in its wake.
For individuals, adaptive resources include developing any of a number of mind-body activities (for example, meditation, mindfulness, slow breathing, and yoga); sufficient but not necessarily excessive levels of exercise (as has been said, if exercise were a pill, it would be the most potent of medicines); nourishing diets; sleep, nature’s restorative state; and perhaps most important, attachment and human connection to people who care about you and whom you care about and trust.
This does not necessarily mean holding or following an institutional religion or belonging to house of worship (though, of course, that melds and augments faith with community). For a great many, myself included, there is spirituality, the belief in a greater power, which need not be a God yet instills a sense of the vastness, universality, and continuity of life.
For communities, adaptive resources include safe homes and neighborhoods; diminishing housing and food insecurity; education, including pre-K; employment, with a livable wage; ridding human interactions of the endless, so-called microaggressions (which are not micro at all, because they accrue) of race, ethnic, class, and age discrimination and injustice; and ready access to quality and affordable health care, now more than ever for the rising tide of mental and substance use disorders that COVID-19 has unleashed.
Every gain we make to ablate racism, social injustice, discrimination, and widely and deeply spread resource and opportunity inequities means more cohesion among the members of our collective tribe. Greater cohesion, a love for thy neighbor, and equity (in action, not polemics) will fuel the resilience we will need to withstand more of COVID-19’s ongoing trauma; that of other, inescapable disasters and losses; and the wear and tear of everyday life. The rewards of equity are priceless and include the dignity that derives from fairness and justice – given and received.
An unprecedented disaster requires a bold response
My, what a list. But to me, the encompassing nature of what’s needed means that we can make differences anywhere, everywhere, and in countless and continuous ways.
The measure of any society is in how it cares for those who are foreclosed, through no fault of their own, from what we all want: a life safe from violence, secure in housing and food, with loving relationships and the pride that comes of making contributions, each in our own, wonderfully unique way.
Where will we all be in a year, 2, or 3 from now? Prepared, or not? Emotionally inoculated, or not? Better equipped, or not? As divided, or more cohesive?
Well, I imagine that depends on each and every one of us.
Lloyd I. Sederer, MD, is a psychiatrist, public health doctor, and writer. He is an adjunct professor at the Columbia University School of Public Health, director of Columbia Psychiatry Media, chief medical officer of Bongo Media, and chair of the advisory board of Get Help. He has been chief medical officer of McLean Hospital, a Harvard teaching hospital; mental health commissioner of New York City (in the Bloomberg administration); and chief medical officer of the New York State Office of Mental Health, the nation’s largest state mental health agency.
A version of this article first appeared on Medscape.com.
We have no way of precisely knowing how many lives might have been saved, and how much grief and loneliness spared and economic ruin contained during COVID-19 if we had risen to its myriad challenges in a timely fashion. However, I feel we can safely say that the United States deserves to be graded with an “F” for its management of the pandemic.
To render this grade, we need only to read the countless verified reports of how critically needed public health measures were not taken soon enough, or sufficiently, to substantially mitigate human and societal suffering.
This began with the failure to protect doctors, nurses, and technicians, who did not have the personal protective equipment needed to prevent infection and spare risk to their loved ones. It soon extended to the country’s failure to adequately protect all its citizens and residents. COVID-19 then rained its grievous consequences disproportionately upon people of color, those living in poverty, and those with housing and food insecurity – those already greatly foreclosed from opportunities to exit from their circumstances.
We all have heard, “Fool me once, shame on you; fool me twice, shame on me.”
Bear witness, colleagues and friends: It will be our shared shame if we too continue to fail in our response to COVID-19. But failure need not happen because protecting ourselves and our country is a solvable problem; complex and demanding for sure, but solvable.
To battle trauma, we must first define it
The sine qua non of a disaster is its psychic and social trauma. I asked Maureen Sayres Van Niel, MD, chair of the American Psychiatric Association’s Minority and Underrepresented Caucus and a former steering committee member of the U.S. Preventive Services Task Force, to define trauma. She said, “It is [the product of] a catastrophic, unexpected event over which we have little control, with grave consequences to the lives and psychological functioning of those individuals and groups affected.”
The COVID-19 pandemic is a massively amplified traumatic event because of the virulence and contagious properties of the virus and its variants; the absence of end date on the horizon; its effect as a proverbial ax that disproportionately falls on the majority of the populace experiencing racial and social inequities; and the ironic yet necessary imperative to distance ourselves from those we care about and who care about us.
Four interdependent factors drive the magnitude of the traumatic impact of a disaster: the degree of exposure to the life-threatening event; the duration and threat of recurrence; an individual’s preexisting (natural and human-made) trauma and mental and addictive disorders; and the adequacy of family and fundamental resources such as housing, food, safety, and access to health care (the social dimensions of health and mental health). These factors underline the “who,” “what,” “where,” and “how” of what should have been (and continue to be) an effective public health response to the COVID-19 pandemic.
Yet existing categories that we have used to predict risk for trauma no longer hold. The gravity, prevalence, and persistence of COVID-19’s horrors erase any differences among victims, witnesses, and bystanders. Dr Sayres Van Niel asserts that we have a “collective, national trauma.” In April, the Kaiser Family Foundation’s Vaccine Monitor reported that 24% of U.S. adults had a close friend or family member who died of COVID-19. That’s 82 million Americans! Our country has eclipsed individual victimization and trauma because we are all in its maw.
Vital lessons from the past
In a previous column, I described my role as New York City’s mental health commissioner after 9/11 and the many lessons we learned during that multiyear process. Our work served as a template for other disasters to follow, such as Hurricane Sandy. Its value to COVID-19 is equally apparent.
We learned that those most at risk of developing symptomatic, functionally impairing mental illness had prior traumatic experiences (for example, from childhood abuse or neglect, violence, war, and forced displacement from their native land) and/or a preexisting mental or substance use disorder.
Once these individuals and communities were identified, we could prioritize their treatment and care. Doing so required mobilizing both inner and external (social) resources, which can be used before disaster strikes or in its wake.
For individuals, adaptive resources include developing any of a number of mind-body activities (for example, meditation, mindfulness, slow breathing, and yoga); sufficient but not necessarily excessive levels of exercise (as has been said, if exercise were a pill, it would be the most potent of medicines); nourishing diets; sleep, nature’s restorative state; and perhaps most important, attachment and human connection to people who care about you and whom you care about and trust.
This does not necessarily mean holding or following an institutional religion or belonging to house of worship (though, of course, that melds and augments faith with community). For a great many, myself included, there is spirituality, the belief in a greater power, which need not be a God yet instills a sense of the vastness, universality, and continuity of life.
For communities, adaptive resources include safe homes and neighborhoods; diminishing housing and food insecurity; education, including pre-K; employment, with a livable wage; ridding human interactions of the endless, so-called microaggressions (which are not micro at all, because they accrue) of race, ethnic, class, and age discrimination and injustice; and ready access to quality and affordable health care, now more than ever for the rising tide of mental and substance use disorders that COVID-19 has unleashed.
Every gain we make to ablate racism, social injustice, discrimination, and widely and deeply spread resource and opportunity inequities means more cohesion among the members of our collective tribe. Greater cohesion, a love for thy neighbor, and equity (in action, not polemics) will fuel the resilience we will need to withstand more of COVID-19’s ongoing trauma; that of other, inescapable disasters and losses; and the wear and tear of everyday life. The rewards of equity are priceless and include the dignity that derives from fairness and justice – given and received.
An unprecedented disaster requires a bold response
My, what a list. But to me, the encompassing nature of what’s needed means that we can make differences anywhere, everywhere, and in countless and continuous ways.
The measure of any society is in how it cares for those who are foreclosed, through no fault of their own, from what we all want: a life safe from violence, secure in housing and food, with loving relationships and the pride that comes of making contributions, each in our own, wonderfully unique way.
Where will we all be in a year, 2, or 3 from now? Prepared, or not? Emotionally inoculated, or not? Better equipped, or not? As divided, or more cohesive?
Well, I imagine that depends on each and every one of us.
Lloyd I. Sederer, MD, is a psychiatrist, public health doctor, and writer. He is an adjunct professor at the Columbia University School of Public Health, director of Columbia Psychiatry Media, chief medical officer of Bongo Media, and chair of the advisory board of Get Help. He has been chief medical officer of McLean Hospital, a Harvard teaching hospital; mental health commissioner of New York City (in the Bloomberg administration); and chief medical officer of the New York State Office of Mental Health, the nation’s largest state mental health agency.
A version of this article first appeared on Medscape.com.
Addressing an uncharted front in the war on COVID-19: Vaccination during pregnancy
In December 2020, the US Food and Drug Administration’s Emergency Use Authorization of the first COVID-19 vaccine presented us with a new tactic in the war against SARS-COV-2—and a new dilemma for obstetricians. What we had learned about COVID-19 infection in pregnancy by that point was alarming. While the vast majority (>90%) of pregnant women who contract COVID-19 recover without requiring hospitalization, pregnant women are at increased risk for severe illness and mechanical ventilation when compared with their nonpregnant counterparts.1 Vertical transmission to the fetus is a rare event, but the increased risk of preterm birth, miscarriage, and preeclampsia makes the fetus a second victim in many cases.2 Moreover, much is still unknown about the long-term impact of severe illness on maternal and fetal health.
Gaining vaccine approval
The COVID-19 vaccine, with its high efficacy rates in the nonpregnant adult population, presents an opportunity to reduce maternal morbidity related to this devastating illness. But unlike other vaccines, such as the flu shot and TDAP, results from prospective studies on COVID-19 vaccination of expectant women are pending. Under the best of circumstances, gaining acceptance of any vaccine during pregnancy faces barriers such as vaccine hesitancy and a general concern from pregnant women about the effect of medical interventions on the fetus. There is no reason to expect that either the mRNA vaccines or the replication-incompetent adenovirus recombinant vector vaccine could cause harm to the developing fetus, but the fact that currently available COVID-19 vaccines use newer technologies complicates the decision for many women.
Nevertheless, what we do know now is much more than we did in December, particularly when it comes to the mRNA vaccines. To date, observational studies of women who received the mRNA vaccine in pregnancy have shown no increased risk of adverse maternal, fetal, or obstetric outcomes.3 Emerging data also indicate that antibodies to the SARS-CoV-2 spike protein—the target of all 3 vaccines—is present in cord blood, potentially protecting the infant in the first months of life from contracting COVID-19 if the mother receives the vaccine during pregnancy.4,5
Our approach to counseling
How can we best help our patients navigate the risks and benefits of the COVID-19 vaccine? First, by acknowledging the obvious: We are in the midst of a pandemic with high rates of community spread, which makes COVID-19 different from any other vaccine-preventable disease at this time. Providing patients with a structure for making an educated decision is essential, taking into account (1) what we know about COVID-19 infection during pregnancy, (2) what we know about vaccine efficacy and safety to date, and (3) individual factors such as:
- The presence of comorbidities such as obesity, heart disease, respiratory disease, and diabetes.
- Potential exposures—“Do you have children in school or daycare? Do childcare providers or other workers come to your home? What is your occupation?”
- The ability to take precautions (social distancing, wearing a mask, etc)
All things considered, the decision to accept the COVID-19 vaccine or not ultimately belongs to the patient. Given disease prevalence and the latest information on vaccine safety in pregnancy, I have been advising my patients in the second trimester or beyond to receive the vaccine with the caveat that delaying the vaccine until the postpartum period is a completely valid alternative. The most important gift we can offer our patients is to arm them with the necessary information so that they can make the choice best for them and their family as we continue to fight this war on COVID-19.
- Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. BMJ. 2020;370:m3320. doi: 10.1136/bmj.m3320.
- Soheili M, Moradi G, Baradaran HR, et al. Clinical manifestation and maternal complications and neonatal outcomes in pregnant women with COVID-19: a comprehensive evidence synthesis and meta-analysis. J Matern Fetal Neonatal Med. February 18, 2021. doi: 10.1080/14767058.2021.1888923.
- Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA Covid-19 vaccine safety in pregnant persons. N Engl J Med. April 21, 2021. doi: 10.1056/NEJMoa2104983.
- Mithal LB, Otero S, Shanes ED, et al. Cord blood antibodies following maternal COVID-19 vaccination during pregnancy. Am J Obstet Gynecol. 2021;S0002-9378(21)00215-5. doi: 10.1016/j.ajog.2021.03.035.
- Rottenstreich A, Zarbiv G, Oiknine-Djian E, et al. Efficient maternofetal transplacental transfer of anti- SARS-CoV-2 spike antibodies after antenatal SARS-CoV-2 BNT162b2 mRNA vaccination. Clin Infect Dis. 2021;ciab266. doi: 10.1093/cid/ciab266.
In December 2020, the US Food and Drug Administration’s Emergency Use Authorization of the first COVID-19 vaccine presented us with a new tactic in the war against SARS-COV-2—and a new dilemma for obstetricians. What we had learned about COVID-19 infection in pregnancy by that point was alarming. While the vast majority (>90%) of pregnant women who contract COVID-19 recover without requiring hospitalization, pregnant women are at increased risk for severe illness and mechanical ventilation when compared with their nonpregnant counterparts.1 Vertical transmission to the fetus is a rare event, but the increased risk of preterm birth, miscarriage, and preeclampsia makes the fetus a second victim in many cases.2 Moreover, much is still unknown about the long-term impact of severe illness on maternal and fetal health.
Gaining vaccine approval
The COVID-19 vaccine, with its high efficacy rates in the nonpregnant adult population, presents an opportunity to reduce maternal morbidity related to this devastating illness. But unlike other vaccines, such as the flu shot and TDAP, results from prospective studies on COVID-19 vaccination of expectant women are pending. Under the best of circumstances, gaining acceptance of any vaccine during pregnancy faces barriers such as vaccine hesitancy and a general concern from pregnant women about the effect of medical interventions on the fetus. There is no reason to expect that either the mRNA vaccines or the replication-incompetent adenovirus recombinant vector vaccine could cause harm to the developing fetus, but the fact that currently available COVID-19 vaccines use newer technologies complicates the decision for many women.
Nevertheless, what we do know now is much more than we did in December, particularly when it comes to the mRNA vaccines. To date, observational studies of women who received the mRNA vaccine in pregnancy have shown no increased risk of adverse maternal, fetal, or obstetric outcomes.3 Emerging data also indicate that antibodies to the SARS-CoV-2 spike protein—the target of all 3 vaccines—is present in cord blood, potentially protecting the infant in the first months of life from contracting COVID-19 if the mother receives the vaccine during pregnancy.4,5
Our approach to counseling
How can we best help our patients navigate the risks and benefits of the COVID-19 vaccine? First, by acknowledging the obvious: We are in the midst of a pandemic with high rates of community spread, which makes COVID-19 different from any other vaccine-preventable disease at this time. Providing patients with a structure for making an educated decision is essential, taking into account (1) what we know about COVID-19 infection during pregnancy, (2) what we know about vaccine efficacy and safety to date, and (3) individual factors such as:
- The presence of comorbidities such as obesity, heart disease, respiratory disease, and diabetes.
- Potential exposures—“Do you have children in school or daycare? Do childcare providers or other workers come to your home? What is your occupation?”
- The ability to take precautions (social distancing, wearing a mask, etc)
All things considered, the decision to accept the COVID-19 vaccine or not ultimately belongs to the patient. Given disease prevalence and the latest information on vaccine safety in pregnancy, I have been advising my patients in the second trimester or beyond to receive the vaccine with the caveat that delaying the vaccine until the postpartum period is a completely valid alternative. The most important gift we can offer our patients is to arm them with the necessary information so that they can make the choice best for them and their family as we continue to fight this war on COVID-19.
In December 2020, the US Food and Drug Administration’s Emergency Use Authorization of the first COVID-19 vaccine presented us with a new tactic in the war against SARS-COV-2—and a new dilemma for obstetricians. What we had learned about COVID-19 infection in pregnancy by that point was alarming. While the vast majority (>90%) of pregnant women who contract COVID-19 recover without requiring hospitalization, pregnant women are at increased risk for severe illness and mechanical ventilation when compared with their nonpregnant counterparts.1 Vertical transmission to the fetus is a rare event, but the increased risk of preterm birth, miscarriage, and preeclampsia makes the fetus a second victim in many cases.2 Moreover, much is still unknown about the long-term impact of severe illness on maternal and fetal health.
Gaining vaccine approval
The COVID-19 vaccine, with its high efficacy rates in the nonpregnant adult population, presents an opportunity to reduce maternal morbidity related to this devastating illness. But unlike other vaccines, such as the flu shot and TDAP, results from prospective studies on COVID-19 vaccination of expectant women are pending. Under the best of circumstances, gaining acceptance of any vaccine during pregnancy faces barriers such as vaccine hesitancy and a general concern from pregnant women about the effect of medical interventions on the fetus. There is no reason to expect that either the mRNA vaccines or the replication-incompetent adenovirus recombinant vector vaccine could cause harm to the developing fetus, but the fact that currently available COVID-19 vaccines use newer technologies complicates the decision for many women.
Nevertheless, what we do know now is much more than we did in December, particularly when it comes to the mRNA vaccines. To date, observational studies of women who received the mRNA vaccine in pregnancy have shown no increased risk of adverse maternal, fetal, or obstetric outcomes.3 Emerging data also indicate that antibodies to the SARS-CoV-2 spike protein—the target of all 3 vaccines—is present in cord blood, potentially protecting the infant in the first months of life from contracting COVID-19 if the mother receives the vaccine during pregnancy.4,5
Our approach to counseling
How can we best help our patients navigate the risks and benefits of the COVID-19 vaccine? First, by acknowledging the obvious: We are in the midst of a pandemic with high rates of community spread, which makes COVID-19 different from any other vaccine-preventable disease at this time. Providing patients with a structure for making an educated decision is essential, taking into account (1) what we know about COVID-19 infection during pregnancy, (2) what we know about vaccine efficacy and safety to date, and (3) individual factors such as:
- The presence of comorbidities such as obesity, heart disease, respiratory disease, and diabetes.
- Potential exposures—“Do you have children in school or daycare? Do childcare providers or other workers come to your home? What is your occupation?”
- The ability to take precautions (social distancing, wearing a mask, etc)
All things considered, the decision to accept the COVID-19 vaccine or not ultimately belongs to the patient. Given disease prevalence and the latest information on vaccine safety in pregnancy, I have been advising my patients in the second trimester or beyond to receive the vaccine with the caveat that delaying the vaccine until the postpartum period is a completely valid alternative. The most important gift we can offer our patients is to arm them with the necessary information so that they can make the choice best for them and their family as we continue to fight this war on COVID-19.
- Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. BMJ. 2020;370:m3320. doi: 10.1136/bmj.m3320.
- Soheili M, Moradi G, Baradaran HR, et al. Clinical manifestation and maternal complications and neonatal outcomes in pregnant women with COVID-19: a comprehensive evidence synthesis and meta-analysis. J Matern Fetal Neonatal Med. February 18, 2021. doi: 10.1080/14767058.2021.1888923.
- Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA Covid-19 vaccine safety in pregnant persons. N Engl J Med. April 21, 2021. doi: 10.1056/NEJMoa2104983.
- Mithal LB, Otero S, Shanes ED, et al. Cord blood antibodies following maternal COVID-19 vaccination during pregnancy. Am J Obstet Gynecol. 2021;S0002-9378(21)00215-5. doi: 10.1016/j.ajog.2021.03.035.
- Rottenstreich A, Zarbiv G, Oiknine-Djian E, et al. Efficient maternofetal transplacental transfer of anti- SARS-CoV-2 spike antibodies after antenatal SARS-CoV-2 BNT162b2 mRNA vaccination. Clin Infect Dis. 2021;ciab266. doi: 10.1093/cid/ciab266.
- Allotey J, Stallings E, Bonet M, et al. Clinical manifestations, risk factors and maternal and perinatal outcomes of coronavirus disease 2019 in pregnancy: living systematic review and meta-analysis. BMJ. 2020;370:m3320. doi: 10.1136/bmj.m3320.
- Soheili M, Moradi G, Baradaran HR, et al. Clinical manifestation and maternal complications and neonatal outcomes in pregnant women with COVID-19: a comprehensive evidence synthesis and meta-analysis. J Matern Fetal Neonatal Med. February 18, 2021. doi: 10.1080/14767058.2021.1888923.
- Shimabukuro TT, Kim SY, Myers TR, et al. Preliminary findings of mRNA Covid-19 vaccine safety in pregnant persons. N Engl J Med. April 21, 2021. doi: 10.1056/NEJMoa2104983.
- Mithal LB, Otero S, Shanes ED, et al. Cord blood antibodies following maternal COVID-19 vaccination during pregnancy. Am J Obstet Gynecol. 2021;S0002-9378(21)00215-5. doi: 10.1016/j.ajog.2021.03.035.
- Rottenstreich A, Zarbiv G, Oiknine-Djian E, et al. Efficient maternofetal transplacental transfer of anti- SARS-CoV-2 spike antibodies after antenatal SARS-CoV-2 BNT162b2 mRNA vaccination. Clin Infect Dis. 2021;ciab266. doi: 10.1093/cid/ciab266.
Self-harm is a leading cause of death for new moms
Death by self-harm through suicide or overdose is a leading cause of death for women in the first year post partum, data indicate. Many of these deaths may be preventable, said Adrienne Griffen, MPP, executive director of the Maternal Mental Health Leadership Alliance.
Ms. Griffen discussed these findings and ways clinicians may be able to help at the 2021 virtual meeting of the American College of Obstetricians and Gynecologists.
Women “visit a health care provider an average of 25 times during a healthy pregnancy and first year of baby’s life,” she said. “Obstetric and primary care providers who serve pregnant and postpartum women are uniquely positioned to intervene effectively to screen and assess women for mental health disorders.”
To that end, clinicians should discuss mental health “early and often,” Ms. Griffen said.
“Asking about mental health issues and suicide will not cause women to think these thoughts,” she said. “We cannot wait for women to raise their hand and ask for help because by the time they do that, they needed help many weeks ago.”
For example, a doctor might tell a patient: “Your mental health is just as important as your physical health, and anxiety and depression are the most common complications of pregnancy and childbirth,” Ms. Griffen suggested. “Every time I see you, I’m going to ask you how you are doing, and we’ll do a formal screening assessment periodically over the course of the pregnancy. … Your job is to answer us honestly so that we can connect you with resources as soon as possible to minimize the impact on you and your baby.”
Although the obstetric provider should introduce this topic, a nurse, lactation consultant, or social worker may conduct screenings and help patients who are experiencing distress, she said.
During the past decade, several medical associations have issued new guidance around screening new mothers for anxiety and depression. One recent ACOG committee opinion recommends screening for depression at least once during pregnancy and once post partum, and encourages doctors to initiate medical therapy if possible and provide resources and referrals.
Another committee opinion suggests that doctors should have contact with a patient between 2 and 3 weeks post partum, primarily to assess for mental health.
Limited data
In discussing maternal suicide statistics, Ms. Griffen focused on data from Maternal Mortality Review Committees (MMRCs).
Two other sources of data about maternal mortality – the National Vital Statistics System and the Pregnancy Mortality Surveillance System – do not include information about suicide, which may be a reason this cause of death is not discussed more often, Ms. Griffen noted.
MMRCs, on the other hand, include information about suicide and self-harm. About half of the states in the United States have these multidisciplinary committees. Committee members review deaths of all women during pregnancy or within 1 year of pregnancy. Members consider a range of clinical and nonclinical data, including reports from social services and police, to try to understand the circumstances of each death.
A report that examined pregnancy-related deaths using data from 14 U.S. MMRCs between 2008 and 2017 showed that mental health conditions were the leading cause of death for non-Hispanic White women. In all, 34% of pregnancy-related suicide deaths had a documented prior suicide attempt, and the majority of suicides happened in the late postpartum time frame (43-365 days post partum).
Some physicians cite a lack of education, time, reimbursement, or referral resources as barriers to maternal mental health screening and treatment, but there may be useful options available, Ms. Griffen said. Postpartum Support International provides resources for physicians, as well as mothers. The National Curriculum in Reproductive Psychiatry and the Seleni Institute also have educational resources.
Some states have psychiatry access programs, where psychiatrists educate obstetricians, family physicians, and pediatricians about how to assess for and treat maternal mental health issues, Ms. Griffen noted.
Self care, social support, and talk therapy may help patients. “Sometimes medication is needed, but a combination of all of these things … can help women recover from maternal mental health conditions,” Ms. Griffen said.
Need to intervene
Although medical societies have emphasized the importance of maternal mental health screening and treatment in recent years, the risk of self-harm has been a concern for obstetricians and gynecologists long before then, said Marc Alan Landsberg, MD, a member of the meeting’s scientific committee who moderated the session.
“We have been talking about this at ACOG for a long time,” Dr. Landsberg said in an interview.
The presentation highlighted why obstetricians, gynecologists, and other doctors who deliver babies and care for women post partum “have got to screen these people,” he said. The finding that 34% of pregnancy-related suicide deaths had a prior suicide attempt indicates that clinicians may be able to identify these patients, Dr. Landsberg said. Suicide and overdose are leading causes of death in the first year post partum and “probably 100% of these are preventable,” he said.
As a first step, screening may be relatively simple. The Edinburgh Postnatal Depression Scale, highlighted during the talk, is an easy and quick tool to use, Dr. Landsberg said. It contains 10 items and assesses for anxiety and depression. It also specifically asks about suicide.
Ms. Griffen and Dr. Landsberg had no conflicts of interest.
Death by self-harm through suicide or overdose is a leading cause of death for women in the first year post partum, data indicate. Many of these deaths may be preventable, said Adrienne Griffen, MPP, executive director of the Maternal Mental Health Leadership Alliance.
Ms. Griffen discussed these findings and ways clinicians may be able to help at the 2021 virtual meeting of the American College of Obstetricians and Gynecologists.
Women “visit a health care provider an average of 25 times during a healthy pregnancy and first year of baby’s life,” she said. “Obstetric and primary care providers who serve pregnant and postpartum women are uniquely positioned to intervene effectively to screen and assess women for mental health disorders.”
To that end, clinicians should discuss mental health “early and often,” Ms. Griffen said.
“Asking about mental health issues and suicide will not cause women to think these thoughts,” she said. “We cannot wait for women to raise their hand and ask for help because by the time they do that, they needed help many weeks ago.”
For example, a doctor might tell a patient: “Your mental health is just as important as your physical health, and anxiety and depression are the most common complications of pregnancy and childbirth,” Ms. Griffen suggested. “Every time I see you, I’m going to ask you how you are doing, and we’ll do a formal screening assessment periodically over the course of the pregnancy. … Your job is to answer us honestly so that we can connect you with resources as soon as possible to minimize the impact on you and your baby.”
Although the obstetric provider should introduce this topic, a nurse, lactation consultant, or social worker may conduct screenings and help patients who are experiencing distress, she said.
During the past decade, several medical associations have issued new guidance around screening new mothers for anxiety and depression. One recent ACOG committee opinion recommends screening for depression at least once during pregnancy and once post partum, and encourages doctors to initiate medical therapy if possible and provide resources and referrals.
Another committee opinion suggests that doctors should have contact with a patient between 2 and 3 weeks post partum, primarily to assess for mental health.
Limited data
In discussing maternal suicide statistics, Ms. Griffen focused on data from Maternal Mortality Review Committees (MMRCs).
Two other sources of data about maternal mortality – the National Vital Statistics System and the Pregnancy Mortality Surveillance System – do not include information about suicide, which may be a reason this cause of death is not discussed more often, Ms. Griffen noted.
MMRCs, on the other hand, include information about suicide and self-harm. About half of the states in the United States have these multidisciplinary committees. Committee members review deaths of all women during pregnancy or within 1 year of pregnancy. Members consider a range of clinical and nonclinical data, including reports from social services and police, to try to understand the circumstances of each death.
A report that examined pregnancy-related deaths using data from 14 U.S. MMRCs between 2008 and 2017 showed that mental health conditions were the leading cause of death for non-Hispanic White women. In all, 34% of pregnancy-related suicide deaths had a documented prior suicide attempt, and the majority of suicides happened in the late postpartum time frame (43-365 days post partum).
Some physicians cite a lack of education, time, reimbursement, or referral resources as barriers to maternal mental health screening and treatment, but there may be useful options available, Ms. Griffen said. Postpartum Support International provides resources for physicians, as well as mothers. The National Curriculum in Reproductive Psychiatry and the Seleni Institute also have educational resources.
Some states have psychiatry access programs, where psychiatrists educate obstetricians, family physicians, and pediatricians about how to assess for and treat maternal mental health issues, Ms. Griffen noted.
Self care, social support, and talk therapy may help patients. “Sometimes medication is needed, but a combination of all of these things … can help women recover from maternal mental health conditions,” Ms. Griffen said.
Need to intervene
Although medical societies have emphasized the importance of maternal mental health screening and treatment in recent years, the risk of self-harm has been a concern for obstetricians and gynecologists long before then, said Marc Alan Landsberg, MD, a member of the meeting’s scientific committee who moderated the session.
“We have been talking about this at ACOG for a long time,” Dr. Landsberg said in an interview.
The presentation highlighted why obstetricians, gynecologists, and other doctors who deliver babies and care for women post partum “have got to screen these people,” he said. The finding that 34% of pregnancy-related suicide deaths had a prior suicide attempt indicates that clinicians may be able to identify these patients, Dr. Landsberg said. Suicide and overdose are leading causes of death in the first year post partum and “probably 100% of these are preventable,” he said.
As a first step, screening may be relatively simple. The Edinburgh Postnatal Depression Scale, highlighted during the talk, is an easy and quick tool to use, Dr. Landsberg said. It contains 10 items and assesses for anxiety and depression. It also specifically asks about suicide.
Ms. Griffen and Dr. Landsberg had no conflicts of interest.
Death by self-harm through suicide or overdose is a leading cause of death for women in the first year post partum, data indicate. Many of these deaths may be preventable, said Adrienne Griffen, MPP, executive director of the Maternal Mental Health Leadership Alliance.
Ms. Griffen discussed these findings and ways clinicians may be able to help at the 2021 virtual meeting of the American College of Obstetricians and Gynecologists.
Women “visit a health care provider an average of 25 times during a healthy pregnancy and first year of baby’s life,” she said. “Obstetric and primary care providers who serve pregnant and postpartum women are uniquely positioned to intervene effectively to screen and assess women for mental health disorders.”
To that end, clinicians should discuss mental health “early and often,” Ms. Griffen said.
“Asking about mental health issues and suicide will not cause women to think these thoughts,” she said. “We cannot wait for women to raise their hand and ask for help because by the time they do that, they needed help many weeks ago.”
For example, a doctor might tell a patient: “Your mental health is just as important as your physical health, and anxiety and depression are the most common complications of pregnancy and childbirth,” Ms. Griffen suggested. “Every time I see you, I’m going to ask you how you are doing, and we’ll do a formal screening assessment periodically over the course of the pregnancy. … Your job is to answer us honestly so that we can connect you with resources as soon as possible to minimize the impact on you and your baby.”
Although the obstetric provider should introduce this topic, a nurse, lactation consultant, or social worker may conduct screenings and help patients who are experiencing distress, she said.
During the past decade, several medical associations have issued new guidance around screening new mothers for anxiety and depression. One recent ACOG committee opinion recommends screening for depression at least once during pregnancy and once post partum, and encourages doctors to initiate medical therapy if possible and provide resources and referrals.
Another committee opinion suggests that doctors should have contact with a patient between 2 and 3 weeks post partum, primarily to assess for mental health.
Limited data
In discussing maternal suicide statistics, Ms. Griffen focused on data from Maternal Mortality Review Committees (MMRCs).
Two other sources of data about maternal mortality – the National Vital Statistics System and the Pregnancy Mortality Surveillance System – do not include information about suicide, which may be a reason this cause of death is not discussed more often, Ms. Griffen noted.
MMRCs, on the other hand, include information about suicide and self-harm. About half of the states in the United States have these multidisciplinary committees. Committee members review deaths of all women during pregnancy or within 1 year of pregnancy. Members consider a range of clinical and nonclinical data, including reports from social services and police, to try to understand the circumstances of each death.
A report that examined pregnancy-related deaths using data from 14 U.S. MMRCs between 2008 and 2017 showed that mental health conditions were the leading cause of death for non-Hispanic White women. In all, 34% of pregnancy-related suicide deaths had a documented prior suicide attempt, and the majority of suicides happened in the late postpartum time frame (43-365 days post partum).
Some physicians cite a lack of education, time, reimbursement, or referral resources as barriers to maternal mental health screening and treatment, but there may be useful options available, Ms. Griffen said. Postpartum Support International provides resources for physicians, as well as mothers. The National Curriculum in Reproductive Psychiatry and the Seleni Institute also have educational resources.
Some states have psychiatry access programs, where psychiatrists educate obstetricians, family physicians, and pediatricians about how to assess for and treat maternal mental health issues, Ms. Griffen noted.
Self care, social support, and talk therapy may help patients. “Sometimes medication is needed, but a combination of all of these things … can help women recover from maternal mental health conditions,” Ms. Griffen said.
Need to intervene
Although medical societies have emphasized the importance of maternal mental health screening and treatment in recent years, the risk of self-harm has been a concern for obstetricians and gynecologists long before then, said Marc Alan Landsberg, MD, a member of the meeting’s scientific committee who moderated the session.
“We have been talking about this at ACOG for a long time,” Dr. Landsberg said in an interview.
The presentation highlighted why obstetricians, gynecologists, and other doctors who deliver babies and care for women post partum “have got to screen these people,” he said. The finding that 34% of pregnancy-related suicide deaths had a prior suicide attempt indicates that clinicians may be able to identify these patients, Dr. Landsberg said. Suicide and overdose are leading causes of death in the first year post partum and “probably 100% of these are preventable,” he said.
As a first step, screening may be relatively simple. The Edinburgh Postnatal Depression Scale, highlighted during the talk, is an easy and quick tool to use, Dr. Landsberg said. It contains 10 items and assesses for anxiety and depression. It also specifically asks about suicide.
Ms. Griffen and Dr. Landsberg had no conflicts of interest.
FROM ACOG 2021
Blood biomarker a ‘promising’ predictor of psychosis relapse
Copeptin, a small peptide secreted with the hormone vasopressin, appears to be one of the first promising biomarkers for predicting psychosis relapse, results of an observational study suggest.
An analysis of plasma copeptin levels in patients with schizophrenia showed those with high plasma levels of the peptide were about three times more likely to experience psychotic relapse, compared with their counterparts with lower levels.
The results suggest, “copeptin could be a promising biomarker in predicting psychotic relapse in schizophrenia spectrum disorder,” said study investigator Jennifer Küster, MD, University Psychiatric Clinics Basel (Switzerland). Measuring copeptin levels upon hospital admission “could help to intensify” the care of at-risk patients, she added.
The findings were presented at the virtual Congress of the Schizophrenia International Research Society 2021.
Relapse prevention important
Two-thirds of patients with schizophrenia experience at least one relapse of a psychotic episode, which in turn increases the risk of the disorder having a chronic course, Dr. Küster noted.
In addition, a psychotic relapse is associated with deterioration of function and cognition and reduced treatment response, “so relapse prevention is important,” she said.
Previous research has explored various methods of predicting schizophrenia outcomes. These include measuring inflammatory markers, catecholamines, oxytocin, and cortisol in combination with imaging markers, “but so far no reliable biomarker has been found,” Dr. Küster said.
She noted that psychotic relapse is associated with increased psychological stress – and vasopressin, which is secreted by the pituitary gland, is a known marker of stress. It is involved in sodium homeostasis and higher brain function and is also elevated in acute psychosis.
However, vasopressin “is challenging to measure because assays are complicated and unreliable,” Dr. Küster said.
As a result, the researchers turned their attention to copeptin, a more stable, more reliable surrogate marker for vasopressin. Copeptin has been shown previously to be a predictor of outcomes in somatic diseases and is also increased during psychological distress.
To measure the utility of copeptin in predicting psychotic relapse,
Baseline characteristics were collected and fasting serum copeptin levels were measured. Disease severity was measured using a range of validated assessment scales.
Predictive factor
Among 69 patients available for analysis, 30 experienced psychotic relapse at 1-year follow-up. Relapse was defined as rehospitalization because of an acute psychotic episode.
There were no differences in baseline demographic characteristics between patients with, and without, psychotic relapse. There were also no differences in baseline psychopathology, including scores on the Positive and Negative Syndrome Scale, the Beck Depression Inventory, and the Global Assessment of Function.
Dr. Küster noted that there were no overall differences between patients with and without psychotic relapse in terms of their plasma copeptin or cortisol levels at baseline.
“The only difference we saw was in diagnosis,” she reported. Patients with psychotic relapse were significantly more likely to have comorbid drug abuse – 43% in patients who relapsed versus 15% of those who did not (P = .02).
However, when the investigators calculated the area under the receiver operating characteristics curve for copeptin levels, they found there was a significant difference in relapse rates in those with copeptin levels >6 pmol/L vs. those with lower levels (hazard ratio, 2.3; P = .039).
When the focus was on only patients with schizophrenia spectrum disorder, the results were even more pronounced. The HR for psychotic relapse in patients with higher vs. lower copeptin levels was 3.2 (P = .028).
“We also looked for other possible predicting factors,” Dr. Küster said. This included sex, age, duration of disease, reason for hospitalization, psychopathology, medication, comorbidities, and cortisol levels. “But none of these factors was associated with psychotic relapse,” she added.
The only factor positively associated with relapse was drug abuse, primarily via marijuana. However, the association with copeptin remained significant even after taking this factor into account.
In future studies, the researchers plan to examine whether copeptin levels could identify which patients at ultra-high risk will transition to first-episode psychosis, as well as to predict development of posttraumatic stress disorder, Dr. Küster said.
A proxy for ‘something simpler’?
Commenting on the findings for this news organization, Leah H. Rubin, PhD, associate professor of neurology, Johns Hopkins University, Baltimore, described the study as “interesting” – and noted that her own research has included measuring vasopressin in patients with untreated first-episode psychosis.
Dr. Rubin’s findings showed that levels of the hormone were associated with psychosis severity, and thus she is “not surprised that they found a marker” that may be promising in psychosis relapse prediction.
However, she took issue with the notion that vasopressin is an unreliable marker, pointing out that the work of her team demonstrates that it can be measured. Dr. Rubin added that she found it to be “pretty stable.”
In addition, because the current study had a small sample size, Dr. Rubin said she would be interested to see whether the findings can be replicated on a larger scale.
She also noted that more than two-thirds of the study population were men. “Vasopressin and oxytocin are sexually dimorphic neuropeptides,” she explained, “so I think it becomes important to ensure ... whether it’s the same for men and women.”
“Just from a psychosocial perspective, what’s going on in those folks’ lives?” Dr. Rubin asked. “Is it truly copeptin” or is it high stress levels that facilitate a relapse? Copeptin levels, she added, may be “a proxy for something simpler.”
The study authors and Dr. Rubin have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Copeptin, a small peptide secreted with the hormone vasopressin, appears to be one of the first promising biomarkers for predicting psychosis relapse, results of an observational study suggest.
An analysis of plasma copeptin levels in patients with schizophrenia showed those with high plasma levels of the peptide were about three times more likely to experience psychotic relapse, compared with their counterparts with lower levels.
The results suggest, “copeptin could be a promising biomarker in predicting psychotic relapse in schizophrenia spectrum disorder,” said study investigator Jennifer Küster, MD, University Psychiatric Clinics Basel (Switzerland). Measuring copeptin levels upon hospital admission “could help to intensify” the care of at-risk patients, she added.
The findings were presented at the virtual Congress of the Schizophrenia International Research Society 2021.
Relapse prevention important
Two-thirds of patients with schizophrenia experience at least one relapse of a psychotic episode, which in turn increases the risk of the disorder having a chronic course, Dr. Küster noted.
In addition, a psychotic relapse is associated with deterioration of function and cognition and reduced treatment response, “so relapse prevention is important,” she said.
Previous research has explored various methods of predicting schizophrenia outcomes. These include measuring inflammatory markers, catecholamines, oxytocin, and cortisol in combination with imaging markers, “but so far no reliable biomarker has been found,” Dr. Küster said.
She noted that psychotic relapse is associated with increased psychological stress – and vasopressin, which is secreted by the pituitary gland, is a known marker of stress. It is involved in sodium homeostasis and higher brain function and is also elevated in acute psychosis.
However, vasopressin “is challenging to measure because assays are complicated and unreliable,” Dr. Küster said.
As a result, the researchers turned their attention to copeptin, a more stable, more reliable surrogate marker for vasopressin. Copeptin has been shown previously to be a predictor of outcomes in somatic diseases and is also increased during psychological distress.
To measure the utility of copeptin in predicting psychotic relapse,
Baseline characteristics were collected and fasting serum copeptin levels were measured. Disease severity was measured using a range of validated assessment scales.
Predictive factor
Among 69 patients available for analysis, 30 experienced psychotic relapse at 1-year follow-up. Relapse was defined as rehospitalization because of an acute psychotic episode.
There were no differences in baseline demographic characteristics between patients with, and without, psychotic relapse. There were also no differences in baseline psychopathology, including scores on the Positive and Negative Syndrome Scale, the Beck Depression Inventory, and the Global Assessment of Function.
Dr. Küster noted that there were no overall differences between patients with and without psychotic relapse in terms of their plasma copeptin or cortisol levels at baseline.
“The only difference we saw was in diagnosis,” she reported. Patients with psychotic relapse were significantly more likely to have comorbid drug abuse – 43% in patients who relapsed versus 15% of those who did not (P = .02).
However, when the investigators calculated the area under the receiver operating characteristics curve for copeptin levels, they found there was a significant difference in relapse rates in those with copeptin levels >6 pmol/L vs. those with lower levels (hazard ratio, 2.3; P = .039).
When the focus was on only patients with schizophrenia spectrum disorder, the results were even more pronounced. The HR for psychotic relapse in patients with higher vs. lower copeptin levels was 3.2 (P = .028).
“We also looked for other possible predicting factors,” Dr. Küster said. This included sex, age, duration of disease, reason for hospitalization, psychopathology, medication, comorbidities, and cortisol levels. “But none of these factors was associated with psychotic relapse,” she added.
The only factor positively associated with relapse was drug abuse, primarily via marijuana. However, the association with copeptin remained significant even after taking this factor into account.
In future studies, the researchers plan to examine whether copeptin levels could identify which patients at ultra-high risk will transition to first-episode psychosis, as well as to predict development of posttraumatic stress disorder, Dr. Küster said.
A proxy for ‘something simpler’?
Commenting on the findings for this news organization, Leah H. Rubin, PhD, associate professor of neurology, Johns Hopkins University, Baltimore, described the study as “interesting” – and noted that her own research has included measuring vasopressin in patients with untreated first-episode psychosis.
Dr. Rubin’s findings showed that levels of the hormone were associated with psychosis severity, and thus she is “not surprised that they found a marker” that may be promising in psychosis relapse prediction.
However, she took issue with the notion that vasopressin is an unreliable marker, pointing out that the work of her team demonstrates that it can be measured. Dr. Rubin added that she found it to be “pretty stable.”
In addition, because the current study had a small sample size, Dr. Rubin said she would be interested to see whether the findings can be replicated on a larger scale.
She also noted that more than two-thirds of the study population were men. “Vasopressin and oxytocin are sexually dimorphic neuropeptides,” she explained, “so I think it becomes important to ensure ... whether it’s the same for men and women.”
“Just from a psychosocial perspective, what’s going on in those folks’ lives?” Dr. Rubin asked. “Is it truly copeptin” or is it high stress levels that facilitate a relapse? Copeptin levels, she added, may be “a proxy for something simpler.”
The study authors and Dr. Rubin have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Copeptin, a small peptide secreted with the hormone vasopressin, appears to be one of the first promising biomarkers for predicting psychosis relapse, results of an observational study suggest.
An analysis of plasma copeptin levels in patients with schizophrenia showed those with high plasma levels of the peptide were about three times more likely to experience psychotic relapse, compared with their counterparts with lower levels.
The results suggest, “copeptin could be a promising biomarker in predicting psychotic relapse in schizophrenia spectrum disorder,” said study investigator Jennifer Küster, MD, University Psychiatric Clinics Basel (Switzerland). Measuring copeptin levels upon hospital admission “could help to intensify” the care of at-risk patients, she added.
The findings were presented at the virtual Congress of the Schizophrenia International Research Society 2021.
Relapse prevention important
Two-thirds of patients with schizophrenia experience at least one relapse of a psychotic episode, which in turn increases the risk of the disorder having a chronic course, Dr. Küster noted.
In addition, a psychotic relapse is associated with deterioration of function and cognition and reduced treatment response, “so relapse prevention is important,” she said.
Previous research has explored various methods of predicting schizophrenia outcomes. These include measuring inflammatory markers, catecholamines, oxytocin, and cortisol in combination with imaging markers, “but so far no reliable biomarker has been found,” Dr. Küster said.
She noted that psychotic relapse is associated with increased psychological stress – and vasopressin, which is secreted by the pituitary gland, is a known marker of stress. It is involved in sodium homeostasis and higher brain function and is also elevated in acute psychosis.
However, vasopressin “is challenging to measure because assays are complicated and unreliable,” Dr. Küster said.
As a result, the researchers turned their attention to copeptin, a more stable, more reliable surrogate marker for vasopressin. Copeptin has been shown previously to be a predictor of outcomes in somatic diseases and is also increased during psychological distress.
To measure the utility of copeptin in predicting psychotic relapse,
Baseline characteristics were collected and fasting serum copeptin levels were measured. Disease severity was measured using a range of validated assessment scales.
Predictive factor
Among 69 patients available for analysis, 30 experienced psychotic relapse at 1-year follow-up. Relapse was defined as rehospitalization because of an acute psychotic episode.
There were no differences in baseline demographic characteristics between patients with, and without, psychotic relapse. There were also no differences in baseline psychopathology, including scores on the Positive and Negative Syndrome Scale, the Beck Depression Inventory, and the Global Assessment of Function.
Dr. Küster noted that there were no overall differences between patients with and without psychotic relapse in terms of their plasma copeptin or cortisol levels at baseline.
“The only difference we saw was in diagnosis,” she reported. Patients with psychotic relapse were significantly more likely to have comorbid drug abuse – 43% in patients who relapsed versus 15% of those who did not (P = .02).
However, when the investigators calculated the area under the receiver operating characteristics curve for copeptin levels, they found there was a significant difference in relapse rates in those with copeptin levels >6 pmol/L vs. those with lower levels (hazard ratio, 2.3; P = .039).
When the focus was on only patients with schizophrenia spectrum disorder, the results were even more pronounced. The HR for psychotic relapse in patients with higher vs. lower copeptin levels was 3.2 (P = .028).
“We also looked for other possible predicting factors,” Dr. Küster said. This included sex, age, duration of disease, reason for hospitalization, psychopathology, medication, comorbidities, and cortisol levels. “But none of these factors was associated with psychotic relapse,” she added.
The only factor positively associated with relapse was drug abuse, primarily via marijuana. However, the association with copeptin remained significant even after taking this factor into account.
In future studies, the researchers plan to examine whether copeptin levels could identify which patients at ultra-high risk will transition to first-episode psychosis, as well as to predict development of posttraumatic stress disorder, Dr. Küster said.
A proxy for ‘something simpler’?
Commenting on the findings for this news organization, Leah H. Rubin, PhD, associate professor of neurology, Johns Hopkins University, Baltimore, described the study as “interesting” – and noted that her own research has included measuring vasopressin in patients with untreated first-episode psychosis.
Dr. Rubin’s findings showed that levels of the hormone were associated with psychosis severity, and thus she is “not surprised that they found a marker” that may be promising in psychosis relapse prediction.
However, she took issue with the notion that vasopressin is an unreliable marker, pointing out that the work of her team demonstrates that it can be measured. Dr. Rubin added that she found it to be “pretty stable.”
In addition, because the current study had a small sample size, Dr. Rubin said she would be interested to see whether the findings can be replicated on a larger scale.
She also noted that more than two-thirds of the study population were men. “Vasopressin and oxytocin are sexually dimorphic neuropeptides,” she explained, “so I think it becomes important to ensure ... whether it’s the same for men and women.”
“Just from a psychosocial perspective, what’s going on in those folks’ lives?” Dr. Rubin asked. “Is it truly copeptin” or is it high stress levels that facilitate a relapse? Copeptin levels, she added, may be “a proxy for something simpler.”
The study authors and Dr. Rubin have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Cell phone, smart watch magnets can affect medical devices, FDA says
The Food and Drug Administration is recommending patients and caregivers keep cell phones and smart watches at least 6 inches away from implanted medical devices, such as pacemakers and defibrillators.
The warning, published on May 13, comes on the heels of recent research reporting that high–field strength magnets in newer smartphones may cause some implanted medical devices to switch to “magnet mode” and suspend normal lifesaving operations until the magnet is moved away.
This, for example, may cause a cardiac defibrillator to be unable to detect tachycardia events, the agency noted. The magnets may also change the operational mode such as turning on asynchronous mode in a pacemaker.
“The FDA is aware of published articles which describe the effect that sufficiently strong magnetic fields can turn on the magnetic safe mode when in close contact,” it said. “The FDA also conducted its own testing on some products that use the high–field strength magnet feature and have confirmed the magnetic field is both consistent with the publications and strong enough to turn on the magnetic safety mode of the medical devices in question.”
The FDA said it believes the risk to patients is low and is not aware of any adverse events associated with this issue at this time.
The American Heart Association has also cautioned that magnetic fields can inhibit the pulse generators for implantable cardioverter defibrillators and pacemakers.
The FDA offered the following simple precautions for individuals with implanted medical devices:
- Keep the consumer electronics, such as certain cell phones and smart watches, 6 inches away from implanted medical devices.
- Do not carry consumer electronics in a pocket over the medical device.
- Check your device using your home monitoring system, if you have one.
- Talk to your health care provider if you are experiencing any symptoms or have questions regarding magnets in consumer electronics and implanted medical devices.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration is recommending patients and caregivers keep cell phones and smart watches at least 6 inches away from implanted medical devices, such as pacemakers and defibrillators.
The warning, published on May 13, comes on the heels of recent research reporting that high–field strength magnets in newer smartphones may cause some implanted medical devices to switch to “magnet mode” and suspend normal lifesaving operations until the magnet is moved away.
This, for example, may cause a cardiac defibrillator to be unable to detect tachycardia events, the agency noted. The magnets may also change the operational mode such as turning on asynchronous mode in a pacemaker.
“The FDA is aware of published articles which describe the effect that sufficiently strong magnetic fields can turn on the magnetic safe mode when in close contact,” it said. “The FDA also conducted its own testing on some products that use the high–field strength magnet feature and have confirmed the magnetic field is both consistent with the publications and strong enough to turn on the magnetic safety mode of the medical devices in question.”
The FDA said it believes the risk to patients is low and is not aware of any adverse events associated with this issue at this time.
The American Heart Association has also cautioned that magnetic fields can inhibit the pulse generators for implantable cardioverter defibrillators and pacemakers.
The FDA offered the following simple precautions for individuals with implanted medical devices:
- Keep the consumer electronics, such as certain cell phones and smart watches, 6 inches away from implanted medical devices.
- Do not carry consumer electronics in a pocket over the medical device.
- Check your device using your home monitoring system, if you have one.
- Talk to your health care provider if you are experiencing any symptoms or have questions regarding magnets in consumer electronics and implanted medical devices.
A version of this article first appeared on Medscape.com.
The Food and Drug Administration is recommending patients and caregivers keep cell phones and smart watches at least 6 inches away from implanted medical devices, such as pacemakers and defibrillators.
The warning, published on May 13, comes on the heels of recent research reporting that high–field strength magnets in newer smartphones may cause some implanted medical devices to switch to “magnet mode” and suspend normal lifesaving operations until the magnet is moved away.
This, for example, may cause a cardiac defibrillator to be unable to detect tachycardia events, the agency noted. The magnets may also change the operational mode such as turning on asynchronous mode in a pacemaker.
“The FDA is aware of published articles which describe the effect that sufficiently strong magnetic fields can turn on the magnetic safe mode when in close contact,” it said. “The FDA also conducted its own testing on some products that use the high–field strength magnet feature and have confirmed the magnetic field is both consistent with the publications and strong enough to turn on the magnetic safety mode of the medical devices in question.”
The FDA said it believes the risk to patients is low and is not aware of any adverse events associated with this issue at this time.
The American Heart Association has also cautioned that magnetic fields can inhibit the pulse generators for implantable cardioverter defibrillators and pacemakers.
The FDA offered the following simple precautions for individuals with implanted medical devices:
- Keep the consumer electronics, such as certain cell phones and smart watches, 6 inches away from implanted medical devices.
- Do not carry consumer electronics in a pocket over the medical device.
- Check your device using your home monitoring system, if you have one.
- Talk to your health care provider if you are experiencing any symptoms or have questions regarding magnets in consumer electronics and implanted medical devices.
A version of this article first appeared on Medscape.com.
Study points to best treatments for depression in primary care
according to a network meta-analysis (NMA) comparing either and both approaches with control conditions in the primary care setting.
The findings are important, since the majority of depressed patients are treated by primary care physicians, yet relatively few randomized trials of treatment have focused on this setting, noted senior study author Pim Cuijpers, PhD, from Vrije Universiteit Amsterdam, and colleagues, in the paper, which was published in Annals of Family Medicine.
“The main message is that clinicians should certainly consider psychotherapy instead of pharmacotherapy, because this is preferred by most patients, and when possible, combined treatments should be the preferred choice because the outcomes are considerably better,” he said in an interview. Either way, he emphasized that “preference of patients is very important and all three treatments are better than usual care.”
The NMA included studies comparing psychotherapy, antidepressant medication, or a combination of both, with control conditions (defined as usual care, wait list, or pill placebo) in adult primary care patients with depression.
Patients could have major depression, persistent mood disorders (dysthymia), both, or high scores on self-rating depression scales. The primary outcome of the NMA was response, defined as a 50% improvement in the Hamilton Depression Rating scores (HAM-D).
A total of 58 studies met inclusion criteria, involving 9,301 patients.
Treatment options compared
Compared with usual care, both psychotherapy alone and pharmacotherapy alone had significantly better response rates, with no significant difference between them (relative risk, 1.60 and RR, 1.65, respectively). The combination of psychotherapy and pharmacotherapy was even better (RR, 2.15), whereas the wait list was less effective (RR, 0.68).
When comparing combined therapy with psychotherapy or pharmacotherapy, the superiority of combination therapy over psychotherapy was only slightly statistically significant (RR, 1.35; 95% confidence interval, 1.00-1.81), while pharmacotherapy was only slightly inferior (RR, 1.30; 95% CI, 0.98-1.73).
“The significance level is not very high, which is related to statistical power,” said Dr. Cuijpers. “But the mean benefit is quite substantial in my opinion, with a 35% higher chance of response in the combined treatment, compared to psychotherapy alone.”
Looking at the outcome of remission, (normally defined as a score of 7 or less on the HAM-D), the outcomes were “comparable to those for response, with the exception that combined treatment was not significantly different from psychotherapy,” they wrote.
One important caveat is that several studies included in the NMA included patients with moderate to severe depression, a population that is different from the usual primary care population of depressed patients who have mild to moderate symptoms. Antidepressant medications are also assumed to work better against more severe symptoms, added the authors. “The inclusion of these studies might therefore have resulted in an overestimation of the effects of pharmacotherapy in the present NMA.”
Among other limitations, the authors noted that studies included mixed populations of patients with dysthymia and major depression; they also made no distinction between different types of antidepressants.
Psychotherapies unknown, but meta-analysis is still useful
Commenting on these findings, Neil Skolnik, MD, professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, said this is “an important study, confirming and extending the conclusions” of a systematic review published in 2016 as a Clinical Practice Guideline from the American College of Physicians.
“Unfortunately, the authors did not specify what type of psychotherapy was studied in the meta-analysis, so we have to look elsewhere if we want to advise our patients on what type of psychotherapy to seek, since there are important differences between different types of therapy,” he said.
Still, he described the study as providing “helpful information for the practicing clinician, as it gives us solid information with which to engage and advise patients in a shared decision-making process for effective treatment of depression.”
“Some patients will choose psychotherapy, some will choose medications. They can make either choice with the confidence that both approaches are effective,” Dr. Skolnik elaborated. “In addition, if psychotherapy does not seem to be sufficiently helping we are on solid ground adding an antidepressant medication to psychotherapy, with this data showing that the combined treatment works better than psychotherapy alone.”
Dr. Cuijpers receives allowances for his memberships on the board of directors of Mind, Fonds Psychische Gezondheid, and Korrelatie, and for being chair of the PACO committee of the Raad voor Civiel-militaire Zorg en Onderzoek of the Dutch Ministry of Defense. He also serves as deputy editor of Depression and Anxiety and associate editor of Psychological Bulletin, and he receives royalties for books he has authored or coauthored. He received grants from the European Union, ZonMw, and PFGV. Another study author reported receiving personal fees from Mitsubishi-Tanabe, MSD, and Shionogi and a grant from Mitsubishi-Tanabe outside the submitted work. One author has received research and consultancy fees from INCiPiT (Italian Network for Paediatric Trials), CARIPLO Foundation, and Angelini Pharmam, while another reported receiving personal fees from Boehringer Ingelheim, Kyowa Kirin, ASKA Pharmaceutical, and Toyota Motor Corporation outside the submitted work. The other authors and Dr. Skolnik reported no conflicts.
according to a network meta-analysis (NMA) comparing either and both approaches with control conditions in the primary care setting.
The findings are important, since the majority of depressed patients are treated by primary care physicians, yet relatively few randomized trials of treatment have focused on this setting, noted senior study author Pim Cuijpers, PhD, from Vrije Universiteit Amsterdam, and colleagues, in the paper, which was published in Annals of Family Medicine.
“The main message is that clinicians should certainly consider psychotherapy instead of pharmacotherapy, because this is preferred by most patients, and when possible, combined treatments should be the preferred choice because the outcomes are considerably better,” he said in an interview. Either way, he emphasized that “preference of patients is very important and all three treatments are better than usual care.”
The NMA included studies comparing psychotherapy, antidepressant medication, or a combination of both, with control conditions (defined as usual care, wait list, or pill placebo) in adult primary care patients with depression.
Patients could have major depression, persistent mood disorders (dysthymia), both, or high scores on self-rating depression scales. The primary outcome of the NMA was response, defined as a 50% improvement in the Hamilton Depression Rating scores (HAM-D).
A total of 58 studies met inclusion criteria, involving 9,301 patients.
Treatment options compared
Compared with usual care, both psychotherapy alone and pharmacotherapy alone had significantly better response rates, with no significant difference between them (relative risk, 1.60 and RR, 1.65, respectively). The combination of psychotherapy and pharmacotherapy was even better (RR, 2.15), whereas the wait list was less effective (RR, 0.68).
When comparing combined therapy with psychotherapy or pharmacotherapy, the superiority of combination therapy over psychotherapy was only slightly statistically significant (RR, 1.35; 95% confidence interval, 1.00-1.81), while pharmacotherapy was only slightly inferior (RR, 1.30; 95% CI, 0.98-1.73).
“The significance level is not very high, which is related to statistical power,” said Dr. Cuijpers. “But the mean benefit is quite substantial in my opinion, with a 35% higher chance of response in the combined treatment, compared to psychotherapy alone.”
Looking at the outcome of remission, (normally defined as a score of 7 or less on the HAM-D), the outcomes were “comparable to those for response, with the exception that combined treatment was not significantly different from psychotherapy,” they wrote.
One important caveat is that several studies included in the NMA included patients with moderate to severe depression, a population that is different from the usual primary care population of depressed patients who have mild to moderate symptoms. Antidepressant medications are also assumed to work better against more severe symptoms, added the authors. “The inclusion of these studies might therefore have resulted in an overestimation of the effects of pharmacotherapy in the present NMA.”
Among other limitations, the authors noted that studies included mixed populations of patients with dysthymia and major depression; they also made no distinction between different types of antidepressants.
Psychotherapies unknown, but meta-analysis is still useful
Commenting on these findings, Neil Skolnik, MD, professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, said this is “an important study, confirming and extending the conclusions” of a systematic review published in 2016 as a Clinical Practice Guideline from the American College of Physicians.
“Unfortunately, the authors did not specify what type of psychotherapy was studied in the meta-analysis, so we have to look elsewhere if we want to advise our patients on what type of psychotherapy to seek, since there are important differences between different types of therapy,” he said.
Still, he described the study as providing “helpful information for the practicing clinician, as it gives us solid information with which to engage and advise patients in a shared decision-making process for effective treatment of depression.”
“Some patients will choose psychotherapy, some will choose medications. They can make either choice with the confidence that both approaches are effective,” Dr. Skolnik elaborated. “In addition, if psychotherapy does not seem to be sufficiently helping we are on solid ground adding an antidepressant medication to psychotherapy, with this data showing that the combined treatment works better than psychotherapy alone.”
Dr. Cuijpers receives allowances for his memberships on the board of directors of Mind, Fonds Psychische Gezondheid, and Korrelatie, and for being chair of the PACO committee of the Raad voor Civiel-militaire Zorg en Onderzoek of the Dutch Ministry of Defense. He also serves as deputy editor of Depression and Anxiety and associate editor of Psychological Bulletin, and he receives royalties for books he has authored or coauthored. He received grants from the European Union, ZonMw, and PFGV. Another study author reported receiving personal fees from Mitsubishi-Tanabe, MSD, and Shionogi and a grant from Mitsubishi-Tanabe outside the submitted work. One author has received research and consultancy fees from INCiPiT (Italian Network for Paediatric Trials), CARIPLO Foundation, and Angelini Pharmam, while another reported receiving personal fees from Boehringer Ingelheim, Kyowa Kirin, ASKA Pharmaceutical, and Toyota Motor Corporation outside the submitted work. The other authors and Dr. Skolnik reported no conflicts.
according to a network meta-analysis (NMA) comparing either and both approaches with control conditions in the primary care setting.
The findings are important, since the majority of depressed patients are treated by primary care physicians, yet relatively few randomized trials of treatment have focused on this setting, noted senior study author Pim Cuijpers, PhD, from Vrije Universiteit Amsterdam, and colleagues, in the paper, which was published in Annals of Family Medicine.
“The main message is that clinicians should certainly consider psychotherapy instead of pharmacotherapy, because this is preferred by most patients, and when possible, combined treatments should be the preferred choice because the outcomes are considerably better,” he said in an interview. Either way, he emphasized that “preference of patients is very important and all three treatments are better than usual care.”
The NMA included studies comparing psychotherapy, antidepressant medication, or a combination of both, with control conditions (defined as usual care, wait list, or pill placebo) in adult primary care patients with depression.
Patients could have major depression, persistent mood disorders (dysthymia), both, or high scores on self-rating depression scales. The primary outcome of the NMA was response, defined as a 50% improvement in the Hamilton Depression Rating scores (HAM-D).
A total of 58 studies met inclusion criteria, involving 9,301 patients.
Treatment options compared
Compared with usual care, both psychotherapy alone and pharmacotherapy alone had significantly better response rates, with no significant difference between them (relative risk, 1.60 and RR, 1.65, respectively). The combination of psychotherapy and pharmacotherapy was even better (RR, 2.15), whereas the wait list was less effective (RR, 0.68).
When comparing combined therapy with psychotherapy or pharmacotherapy, the superiority of combination therapy over psychotherapy was only slightly statistically significant (RR, 1.35; 95% confidence interval, 1.00-1.81), while pharmacotherapy was only slightly inferior (RR, 1.30; 95% CI, 0.98-1.73).
“The significance level is not very high, which is related to statistical power,” said Dr. Cuijpers. “But the mean benefit is quite substantial in my opinion, with a 35% higher chance of response in the combined treatment, compared to psychotherapy alone.”
Looking at the outcome of remission, (normally defined as a score of 7 or less on the HAM-D), the outcomes were “comparable to those for response, with the exception that combined treatment was not significantly different from psychotherapy,” they wrote.
One important caveat is that several studies included in the NMA included patients with moderate to severe depression, a population that is different from the usual primary care population of depressed patients who have mild to moderate symptoms. Antidepressant medications are also assumed to work better against more severe symptoms, added the authors. “The inclusion of these studies might therefore have resulted in an overestimation of the effects of pharmacotherapy in the present NMA.”
Among other limitations, the authors noted that studies included mixed populations of patients with dysthymia and major depression; they also made no distinction between different types of antidepressants.
Psychotherapies unknown, but meta-analysis is still useful
Commenting on these findings, Neil Skolnik, MD, professor of family and community medicine at Sidney Kimmel Medical College, Philadelphia, said this is “an important study, confirming and extending the conclusions” of a systematic review published in 2016 as a Clinical Practice Guideline from the American College of Physicians.
“Unfortunately, the authors did not specify what type of psychotherapy was studied in the meta-analysis, so we have to look elsewhere if we want to advise our patients on what type of psychotherapy to seek, since there are important differences between different types of therapy,” he said.
Still, he described the study as providing “helpful information for the practicing clinician, as it gives us solid information with which to engage and advise patients in a shared decision-making process for effective treatment of depression.”
“Some patients will choose psychotherapy, some will choose medications. They can make either choice with the confidence that both approaches are effective,” Dr. Skolnik elaborated. “In addition, if psychotherapy does not seem to be sufficiently helping we are on solid ground adding an antidepressant medication to psychotherapy, with this data showing that the combined treatment works better than psychotherapy alone.”
Dr. Cuijpers receives allowances for his memberships on the board of directors of Mind, Fonds Psychische Gezondheid, and Korrelatie, and for being chair of the PACO committee of the Raad voor Civiel-militaire Zorg en Onderzoek of the Dutch Ministry of Defense. He also serves as deputy editor of Depression and Anxiety and associate editor of Psychological Bulletin, and he receives royalties for books he has authored or coauthored. He received grants from the European Union, ZonMw, and PFGV. Another study author reported receiving personal fees from Mitsubishi-Tanabe, MSD, and Shionogi and a grant from Mitsubishi-Tanabe outside the submitted work. One author has received research and consultancy fees from INCiPiT (Italian Network for Paediatric Trials), CARIPLO Foundation, and Angelini Pharmam, while another reported receiving personal fees from Boehringer Ingelheim, Kyowa Kirin, ASKA Pharmaceutical, and Toyota Motor Corporation outside the submitted work. The other authors and Dr. Skolnik reported no conflicts.
FROM ANNALS OF FAMILY MEDICINE
IBD online: What do patients search for?
A new online survey of inflammatory bowel disease (IBD) patients found that individuals seeking information on social media are generally satisfied with the care that they get from their health care providers. However, the online activity suggested a desire for more information, especially with respect to supportive needs like diet and complementary/alternative medicine (CAM).
The study was led by Idan Goren, MD, and Henit Yanai, MD, of Rabin Medical Center, Petah Tikva, Israel.
The researchers suspected that social media users with IBD were looking for information they weren’t getting from their provider, so the researchers set out to identify those specific unmet needs. In a pilot exploratory phase of their investigation, they conducted an initial survey followed by an analysis of social media posts, then they conducted a second phase with a survey based on the findings in the pilot exploration.
The initial survey was conducted within a social media platform in Israel called Camoni, where patients can interact with each other and with health care providers who have experience treating IBD, including gastroenterologists, dietitians, and psychologists. The survey included 10 items about disease characteristics, information needs, information search habits, and other factors. The subsequent analysis step included individual posts on the network between January 2014 and January 2019; the investigators categorized posts by the topics of interest brought up in the initial survey and determined the frequency of posts related to each category.
Out of the 255 respondents to this initial survey, 72% reported satisfaction with the information they received in person. In addition, 67% said that search engines like Google were their most important source of disease-related information, 58% reported relying heavily on websites, and 53% reported relying on health care providers. The most common topics of interest were diet (65%), medications and their potential adverse effects (58%), disease management (48%), and CAM (43%).
After this pilot exploratory phase, the researchers developed a structured survey that they used in IBD-based forums on Facebook and other social networks. Data were collected from this survey during a 4-week period in November 2019.
About half of the 534 respondents to the more widely distributed follow-up survey were in Israel. Overall, 83% reported using IBD-related medications, 45% of which were biologics. Out of the 534 respondents, 70% primarily received treatment from IBD referral centers. Interestingly, 77% said that they would prefer to rely on social media that is guided by health care providers, but only 22% reported that they actually used such a network. Responding along a visual analog scale, they reported general satisfaction with their routine IBD care (mean score, 79 ± 27 out of 100), their providers’ effectiveness of communication (82 ± 24), and the providers’ ability to understand patient concerns (73 ± 28). Those who were active in social media rated accessibility of IBD service as 68 ± 30. Exploration of topical interest found the most common to be diet (46%), lifestyle (45%), CAM (43%), diagnostic test interpretation (34%), and specialist referrals and reviews (31%).
The general satisfaction with information from health care providers contrasted with some previous studies that had shown that patients seeking information online often felt the opposite: For example, a 2019 Canadian survey found that only 10%-36% of IBD patients believed they received adequate information on IBD issues during clinical visits. The authors of the current study speculated that the incongruence might be explained by the fact that the current survey included patients with greater disease burden, who might get more attention during clinic visits than might patients with milder illness.
“In conclusion, our results indicate that patients’ activity on [social media] appears to be independent of their satisfaction with formal IBD care and rather reflects the contemporary need for ongoing information, particularly focused on supportive needs, such as diet and CAM,” the investigators wrote.
“Try not to Google everything”
The findings weren’t surprising, but the researchers found that patients seeking information online often have a high level of disease burden, as evidenced by biologics use and a majority being seen by specialists. That’s worrisome, said Jason Reich, MD, a gastroenterologist in Fall River, Mass., who has also studied social media use among IBD patients but was not involved in this study. “The last person you want getting poor-quality information is someone with pretty active disease,” said Dr. Reich in an interview.
Dr. Reich agreed with the authors that IBD specialists should consider having a dietitian in their clinic, or at least refer patients to dietitians early on. He also advocated for gastroenterologists (and all physicians, really) to have an online presence, if possible. “At least make themselves and their office accessible. I always tell my patients, if you have questions, try not to Google everything online and just shoot me a message through the portal instead,” said Dr. Reich. He added that nurses can handle such duties, especially those trained in IBD. “Personally, I don’t mind sending my short messages back and forth. Especially if it’s just a question. That’s easy enough to do when it takes maybe a minute or 2.”
The authors disclosed no funding sources. Dr. Reich has no relevant financial disclosures.
A new online survey of inflammatory bowel disease (IBD) patients found that individuals seeking information on social media are generally satisfied with the care that they get from their health care providers. However, the online activity suggested a desire for more information, especially with respect to supportive needs like diet and complementary/alternative medicine (CAM).
The study was led by Idan Goren, MD, and Henit Yanai, MD, of Rabin Medical Center, Petah Tikva, Israel.
The researchers suspected that social media users with IBD were looking for information they weren’t getting from their provider, so the researchers set out to identify those specific unmet needs. In a pilot exploratory phase of their investigation, they conducted an initial survey followed by an analysis of social media posts, then they conducted a second phase with a survey based on the findings in the pilot exploration.
The initial survey was conducted within a social media platform in Israel called Camoni, where patients can interact with each other and with health care providers who have experience treating IBD, including gastroenterologists, dietitians, and psychologists. The survey included 10 items about disease characteristics, information needs, information search habits, and other factors. The subsequent analysis step included individual posts on the network between January 2014 and January 2019; the investigators categorized posts by the topics of interest brought up in the initial survey and determined the frequency of posts related to each category.
Out of the 255 respondents to this initial survey, 72% reported satisfaction with the information they received in person. In addition, 67% said that search engines like Google were their most important source of disease-related information, 58% reported relying heavily on websites, and 53% reported relying on health care providers. The most common topics of interest were diet (65%), medications and their potential adverse effects (58%), disease management (48%), and CAM (43%).
After this pilot exploratory phase, the researchers developed a structured survey that they used in IBD-based forums on Facebook and other social networks. Data were collected from this survey during a 4-week period in November 2019.
About half of the 534 respondents to the more widely distributed follow-up survey were in Israel. Overall, 83% reported using IBD-related medications, 45% of which were biologics. Out of the 534 respondents, 70% primarily received treatment from IBD referral centers. Interestingly, 77% said that they would prefer to rely on social media that is guided by health care providers, but only 22% reported that they actually used such a network. Responding along a visual analog scale, they reported general satisfaction with their routine IBD care (mean score, 79 ± 27 out of 100), their providers’ effectiveness of communication (82 ± 24), and the providers’ ability to understand patient concerns (73 ± 28). Those who were active in social media rated accessibility of IBD service as 68 ± 30. Exploration of topical interest found the most common to be diet (46%), lifestyle (45%), CAM (43%), diagnostic test interpretation (34%), and specialist referrals and reviews (31%).
The general satisfaction with information from health care providers contrasted with some previous studies that had shown that patients seeking information online often felt the opposite: For example, a 2019 Canadian survey found that only 10%-36% of IBD patients believed they received adequate information on IBD issues during clinical visits. The authors of the current study speculated that the incongruence might be explained by the fact that the current survey included patients with greater disease burden, who might get more attention during clinic visits than might patients with milder illness.
“In conclusion, our results indicate that patients’ activity on [social media] appears to be independent of their satisfaction with formal IBD care and rather reflects the contemporary need for ongoing information, particularly focused on supportive needs, such as diet and CAM,” the investigators wrote.
“Try not to Google everything”
The findings weren’t surprising, but the researchers found that patients seeking information online often have a high level of disease burden, as evidenced by biologics use and a majority being seen by specialists. That’s worrisome, said Jason Reich, MD, a gastroenterologist in Fall River, Mass., who has also studied social media use among IBD patients but was not involved in this study. “The last person you want getting poor-quality information is someone with pretty active disease,” said Dr. Reich in an interview.
Dr. Reich agreed with the authors that IBD specialists should consider having a dietitian in their clinic, or at least refer patients to dietitians early on. He also advocated for gastroenterologists (and all physicians, really) to have an online presence, if possible. “At least make themselves and their office accessible. I always tell my patients, if you have questions, try not to Google everything online and just shoot me a message through the portal instead,” said Dr. Reich. He added that nurses can handle such duties, especially those trained in IBD. “Personally, I don’t mind sending my short messages back and forth. Especially if it’s just a question. That’s easy enough to do when it takes maybe a minute or 2.”
The authors disclosed no funding sources. Dr. Reich has no relevant financial disclosures.
A new online survey of inflammatory bowel disease (IBD) patients found that individuals seeking information on social media are generally satisfied with the care that they get from their health care providers. However, the online activity suggested a desire for more information, especially with respect to supportive needs like diet and complementary/alternative medicine (CAM).
The study was led by Idan Goren, MD, and Henit Yanai, MD, of Rabin Medical Center, Petah Tikva, Israel.
The researchers suspected that social media users with IBD were looking for information they weren’t getting from their provider, so the researchers set out to identify those specific unmet needs. In a pilot exploratory phase of their investigation, they conducted an initial survey followed by an analysis of social media posts, then they conducted a second phase with a survey based on the findings in the pilot exploration.
The initial survey was conducted within a social media platform in Israel called Camoni, where patients can interact with each other and with health care providers who have experience treating IBD, including gastroenterologists, dietitians, and psychologists. The survey included 10 items about disease characteristics, information needs, information search habits, and other factors. The subsequent analysis step included individual posts on the network between January 2014 and January 2019; the investigators categorized posts by the topics of interest brought up in the initial survey and determined the frequency of posts related to each category.
Out of the 255 respondents to this initial survey, 72% reported satisfaction with the information they received in person. In addition, 67% said that search engines like Google were their most important source of disease-related information, 58% reported relying heavily on websites, and 53% reported relying on health care providers. The most common topics of interest were diet (65%), medications and their potential adverse effects (58%), disease management (48%), and CAM (43%).
After this pilot exploratory phase, the researchers developed a structured survey that they used in IBD-based forums on Facebook and other social networks. Data were collected from this survey during a 4-week period in November 2019.
About half of the 534 respondents to the more widely distributed follow-up survey were in Israel. Overall, 83% reported using IBD-related medications, 45% of which were biologics. Out of the 534 respondents, 70% primarily received treatment from IBD referral centers. Interestingly, 77% said that they would prefer to rely on social media that is guided by health care providers, but only 22% reported that they actually used such a network. Responding along a visual analog scale, they reported general satisfaction with their routine IBD care (mean score, 79 ± 27 out of 100), their providers’ effectiveness of communication (82 ± 24), and the providers’ ability to understand patient concerns (73 ± 28). Those who were active in social media rated accessibility of IBD service as 68 ± 30. Exploration of topical interest found the most common to be diet (46%), lifestyle (45%), CAM (43%), diagnostic test interpretation (34%), and specialist referrals and reviews (31%).
The general satisfaction with information from health care providers contrasted with some previous studies that had shown that patients seeking information online often felt the opposite: For example, a 2019 Canadian survey found that only 10%-36% of IBD patients believed they received adequate information on IBD issues during clinical visits. The authors of the current study speculated that the incongruence might be explained by the fact that the current survey included patients with greater disease burden, who might get more attention during clinic visits than might patients with milder illness.
“In conclusion, our results indicate that patients’ activity on [social media] appears to be independent of their satisfaction with formal IBD care and rather reflects the contemporary need for ongoing information, particularly focused on supportive needs, such as diet and CAM,” the investigators wrote.
“Try not to Google everything”
The findings weren’t surprising, but the researchers found that patients seeking information online often have a high level of disease burden, as evidenced by biologics use and a majority being seen by specialists. That’s worrisome, said Jason Reich, MD, a gastroenterologist in Fall River, Mass., who has also studied social media use among IBD patients but was not involved in this study. “The last person you want getting poor-quality information is someone with pretty active disease,” said Dr. Reich in an interview.
Dr. Reich agreed with the authors that IBD specialists should consider having a dietitian in their clinic, or at least refer patients to dietitians early on. He also advocated for gastroenterologists (and all physicians, really) to have an online presence, if possible. “At least make themselves and their office accessible. I always tell my patients, if you have questions, try not to Google everything online and just shoot me a message through the portal instead,” said Dr. Reich. He added that nurses can handle such duties, especially those trained in IBD. “Personally, I don’t mind sending my short messages back and forth. Especially if it’s just a question. That’s easy enough to do when it takes maybe a minute or 2.”
The authors disclosed no funding sources. Dr. Reich has no relevant financial disclosures.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Focus on prepregnancy care
Improving maternal morbidity and mortality begins prior to conception. Numerous modifiable and nonmodifiable factors—lifestyle behaviors, chronic medical conditions, medications, immunizations, prior pregnancy events—have been shown to improve pregnancy outcomes if they are reviewed, identified, and optimized before conception.
Laying a solid foundation for a healthy pregnancy requires a comprehensive approach to patient counseling. However, the national Pregnancy Risk Assessment Monitoring System (PRAMS; a surveillance program of the Centers for Disease Control and Prevention) data from 2014 show that only about 20% of women receive counseling on at least 5 out of 11 healthy lifestyle behaviors and prevention strategies before pregnancy. The ability to leverage technology-enabled smart device applications can provide clinicians with immediate access to information necessary to address with patients at a preconception visit. Apps built specifically for physicians offer a convenient, thorough, and peer-vetted reference that can increase the efficiency and quality of consultation in a busy practice.
Prepregnancy care app considerations
When applying the ACOG-recommended rubric to evaluate the quality of an app targeted to address preconception counseling, the accuracy and objectivity of the content, as well as the app’s ease of use, are vital characteristics to consider, and these criteria should score 4 out of 4 on the rubric.
Several apps offer suggestions on how to address important components of health, including counseling and intervention strategies and evidence-based recommendations. The most efficacious apps offer embedded references to more detailed resources for use when complexities inevitably arise during consultation. Truly comprehensive prepregnancy care requires clinicians to take a step beyond the review of patients’ medications and comorbidities. It is therefore helpful to implement point-of-care apps that prompt evaluation of the often-overlooked aspects of prepregnancy counseling, including risk of interpersonal violence and infectious diseases, occupational exposures, and immunization status.
Physician-focused prepregnancy apps that provide reminders, prompts, and strategies for addressing a comprehensive set of health components prior to conception can be valuable tools to incorporate into both educational environments and busy practices to address maternal morbidity and mortality. ●
Improving maternal morbidity and mortality begins prior to conception. Numerous modifiable and nonmodifiable factors—lifestyle behaviors, chronic medical conditions, medications, immunizations, prior pregnancy events—have been shown to improve pregnancy outcomes if they are reviewed, identified, and optimized before conception.
Laying a solid foundation for a healthy pregnancy requires a comprehensive approach to patient counseling. However, the national Pregnancy Risk Assessment Monitoring System (PRAMS; a surveillance program of the Centers for Disease Control and Prevention) data from 2014 show that only about 20% of women receive counseling on at least 5 out of 11 healthy lifestyle behaviors and prevention strategies before pregnancy. The ability to leverage technology-enabled smart device applications can provide clinicians with immediate access to information necessary to address with patients at a preconception visit. Apps built specifically for physicians offer a convenient, thorough, and peer-vetted reference that can increase the efficiency and quality of consultation in a busy practice.
Prepregnancy care app considerations
When applying the ACOG-recommended rubric to evaluate the quality of an app targeted to address preconception counseling, the accuracy and objectivity of the content, as well as the app’s ease of use, are vital characteristics to consider, and these criteria should score 4 out of 4 on the rubric.
Several apps offer suggestions on how to address important components of health, including counseling and intervention strategies and evidence-based recommendations. The most efficacious apps offer embedded references to more detailed resources for use when complexities inevitably arise during consultation. Truly comprehensive prepregnancy care requires clinicians to take a step beyond the review of patients’ medications and comorbidities. It is therefore helpful to implement point-of-care apps that prompt evaluation of the often-overlooked aspects of prepregnancy counseling, including risk of interpersonal violence and infectious diseases, occupational exposures, and immunization status.
Physician-focused prepregnancy apps that provide reminders, prompts, and strategies for addressing a comprehensive set of health components prior to conception can be valuable tools to incorporate into both educational environments and busy practices to address maternal morbidity and mortality. ●
Improving maternal morbidity and mortality begins prior to conception. Numerous modifiable and nonmodifiable factors—lifestyle behaviors, chronic medical conditions, medications, immunizations, prior pregnancy events—have been shown to improve pregnancy outcomes if they are reviewed, identified, and optimized before conception.
Laying a solid foundation for a healthy pregnancy requires a comprehensive approach to patient counseling. However, the national Pregnancy Risk Assessment Monitoring System (PRAMS; a surveillance program of the Centers for Disease Control and Prevention) data from 2014 show that only about 20% of women receive counseling on at least 5 out of 11 healthy lifestyle behaviors and prevention strategies before pregnancy. The ability to leverage technology-enabled smart device applications can provide clinicians with immediate access to information necessary to address with patients at a preconception visit. Apps built specifically for physicians offer a convenient, thorough, and peer-vetted reference that can increase the efficiency and quality of consultation in a busy practice.
Prepregnancy care app considerations
When applying the ACOG-recommended rubric to evaluate the quality of an app targeted to address preconception counseling, the accuracy and objectivity of the content, as well as the app’s ease of use, are vital characteristics to consider, and these criteria should score 4 out of 4 on the rubric.
Several apps offer suggestions on how to address important components of health, including counseling and intervention strategies and evidence-based recommendations. The most efficacious apps offer embedded references to more detailed resources for use when complexities inevitably arise during consultation. Truly comprehensive prepregnancy care requires clinicians to take a step beyond the review of patients’ medications and comorbidities. It is therefore helpful to implement point-of-care apps that prompt evaluation of the often-overlooked aspects of prepregnancy counseling, including risk of interpersonal violence and infectious diseases, occupational exposures, and immunization status.
Physician-focused prepregnancy apps that provide reminders, prompts, and strategies for addressing a comprehensive set of health components prior to conception can be valuable tools to incorporate into both educational environments and busy practices to address maternal morbidity and mortality. ●
Championing preventive care in ObGyn: A tool to evaluate for useful medical apps
Personalizing care is at the heart of the American College of Obstetricians and Gynecologists (ACOG) 2020–2021 President Dr. Eva Chalas’ initiative to “Revisit the Visit.” As obstetrician-gynecologists, we care for patients across the entirety of their life. This role gives us the opportunity to form long-term partnerships with women to address important preventive health care measures.
Dr. Chalas established a Presidential Task Force that identified 5 areas of preventive health that significantly influence the long-term morbidity of women: obesity, cardiovascular disease, preconception counseling, diabetes, and cancer risk. The annual visit can serve as a particularly impactful point of care to achieve specific preventive care objectives and offer mitigation strategies based on patient-specific risk factors. We are uniquely positioned to identify and initiate the conversation and subsequently manage, treat, and address these critical health areas.
Harnessing modern technology
To adopt these health topics into practice, we need improved, more effective tools both to increase productivity during the office visit and to provide more personalized care. Notably, the widespread adoption of and proliferation of mobile devices—and the medical apps accessible on them—is creating new and innovative ways to improve health and health care delivery. More than 90% of physicians use a smartphone at work, and 62% of smartphone users have used their device to gather health data.1
In addition, according to a US Food and Drug Administration (FDA) report, in 2017, 325,000 health care applications were available on smartphones; this equates to an expected 3.7 billion mobile health application downloads that year by 1.7 billion smartphone users worldwide.2 As of October 2020, 48,000-plus health apps were available on the iOS mobile operating system alone.3
For patients and clinicians, picking the most suitable apps can be challenging in the face of evolving clinical evidence, emerging privacy risks, functionality concerns, and the fact that apps constantly update and change. Many have relied on star rating systems and user reviews in app stores to guide their selection process despite mounting evidence that suggests that such evaluation methods are misleading, not always addressing such important parameters as usability, validity, security, and privacy.4,5
Approaches for evaluating medical apps
Many app evaluation frameworks have emerged, but none is universally accepted within the health care field.
The American Psychiatric Association’s (APA) App Evaluation Model represents a comprehensive resource to consider when evaluating medical apps. It stratifies numerous variables into 5 levels that form a pyramid. In this model, background information forms the base of this pyramid and includes factors such as business model, credibility, cost, and advertising of the app. The top of the pyramid is comprised of data integration that considers data ownership and therapeutic alliance.6 Although this model is beneficial in that it provides a framework, it is not practical for point-of-care purposes as it offers no objective way to rate or score an app for quick and easy comparison.
The privately owned and operated Health On the Net (HON) Foundation is well known for its HONcode, an ethical standard for quality medical information on the internet. It uses 8 principles to certify a health website. However, the HON website itself states that it cannot guarantee the accuracy or completeness of medical information presented by a site.7 Although HON certification by a website is a sign of good intention, it is not beneficial to the practicing clinician who is looking to use an app to directly assist in clinical care.
The Agency for Healthcare Research and Quality (AHRQ) is another well-respected body that has delineated essential details to consider when using a health website. The AHRQ identifies features (similar to those of the APA pyramid and HONcode) for users to consider, such as credibility, content, design, and disclosures.8 However, this model too lacks a concise user-friendly evaluating system.
Although the FDA plans to apply some regulatory authority to the evaluation of a certain subset of high-risk mobile medical apps, it is not planning to evaluate or regulate many of the medical apps that clinicians use in daily practice. This leaves us, and our patients, to be guided by the principle of caveat emptor, or “let the buyer beware.”
Thus, Dr. Chalas’ Presidential Task Force carefully considered various resources to provide a useful tool that would help obstetrician-gynecologists objectively vet a medical app in practice.
Continue to: The Task Force’s recommended rubric...
The Task Force’s recommended rubric
The rubric shown for evaluating mobile drug information apps was developed by the American Society of Health-System Pharmacists (ASHP). The ASHP rubric takes into account the criteria recognized by the APA pyramid, the HON Foundation, and the AHRQ and incorporates them into a user-friendly tool and scoring system that can be applied as an evaluation checklist.9 This tool is meant to aid clinicians in evaluating medical apps, but it ultimately is the user’s decision to determine if an app’s deficiencies should deter its use.
While all of the criteria are relevant and important, it is incumbent on us as medical experts to pay careful attention to the accuracy, authority, objectivity, timeliness, and security of any app we consider incorporating into clinical practice. A low score on these criteria would belie any perceived usefulness or value the app may have.
When applying the rubric to evaluate the quality of an app, we should be mindful of the primary user and which characteristics are more important than others to effect positive changes in health. For example, in addressing obesity, it is the patient who will be interacting with the app. Therefore, it’s important that the app should score, on a 1- to 4-point scale (1 point being major deficiencies, 4 points being no deficiencies), a 4 out of 4 on features like usefulness, functionality, and design. Coveted design features that enhance the user’s experience will appeal to patients and keep them engaged and motivated. However, when addressing a woman’s health with respect to cancer risk, the principal features on which the app should score 4 out of 4 would be authority, objectivity, timeliness, and accuracy.
In the upcoming articles in this series, a member of the Presidential Task Force will reference the ASHP rubric to guide clinicians in choosing apps to address one of the critical health areas with their patients. The author of the piece will highlight key features of an app to consider what would add the most value in incorporating its use in clinical practice.
It would be impossible to evaluate all health care apps even if we focused only on the medical apps relevant to obstetrics and gynecology. There is much value in having a framework for efficiently measuring an app’s benefit in clinical practice. The objective of this article series is to help clinicians Revisit the Visit by providing an effective tool to evaluate a medical app. ●
- Mobius MD website. 11 Surprising mobile health statistics. http://www.mobius.md/blog/2019/03/11-mobile-health -statistics/. Accessed January 19, 2021.
- US Food and Drug Administration website. Device software functions including medical applications. November 5, 2019. https://www.fda.gov/medical-devices/digital-health-center -excellence/device-software-functions-including-mobile -medical-applications. Accessed March 10, 2021.
- Statista website. Number of mHealth apps available in the Apple App Store from 1st quarter 2015 to 4th quarter 2020. https://www.statista.com/statistics/779910/health-apps -available-ios-worldwide/. Accessed January 19, 2021.
- Campbell L. Using star ratings to choose a medical app? There’s a better way. Healthline website. Updated August 3, 2018. http://healthline.com/health-news/using-ratings-to -choose-medical-app-theres-a-better-way. Accessed April 22, 2021.
- Levine DM, Co Z, Newmark LP, et al. Design and testing of a mobile health application rating tool. NPJ Digit Med. 2020;3:74.
- Torous JB, Chan SR, Gipson SY, et al. A hierarchical framework for evaluation and informed decision making regarding smartphone apps for clinical care. Psychiatr Serv. 2018;69:498-500.
- Health On the Net website. The commitment to reliable health and medical information on the internet. https:// www.hon.ch/HONcode/Patients/Visitor/visitor.html. Accessed January 19, 2021.
- Agency for Healthcare Research and Quality. Assessing the quality of internet health information. June 1999. http:// www.ahrq.gov/research/data/infoqual.html. Accessed April 22, 2021.
- Hanrahan C, Aungst TD, Cole S. Evaluating mobile medical applications. American Society of Health-System Pharmacists eReports. https://www.ashp.org/-/media/store-files /mobile-medical-apps.ashx. Accessed January 22, 2021.
Personalizing care is at the heart of the American College of Obstetricians and Gynecologists (ACOG) 2020–2021 President Dr. Eva Chalas’ initiative to “Revisit the Visit.” As obstetrician-gynecologists, we care for patients across the entirety of their life. This role gives us the opportunity to form long-term partnerships with women to address important preventive health care measures.
Dr. Chalas established a Presidential Task Force that identified 5 areas of preventive health that significantly influence the long-term morbidity of women: obesity, cardiovascular disease, preconception counseling, diabetes, and cancer risk. The annual visit can serve as a particularly impactful point of care to achieve specific preventive care objectives and offer mitigation strategies based on patient-specific risk factors. We are uniquely positioned to identify and initiate the conversation and subsequently manage, treat, and address these critical health areas.
Harnessing modern technology
To adopt these health topics into practice, we need improved, more effective tools both to increase productivity during the office visit and to provide more personalized care. Notably, the widespread adoption of and proliferation of mobile devices—and the medical apps accessible on them—is creating new and innovative ways to improve health and health care delivery. More than 90% of physicians use a smartphone at work, and 62% of smartphone users have used their device to gather health data.1
In addition, according to a US Food and Drug Administration (FDA) report, in 2017, 325,000 health care applications were available on smartphones; this equates to an expected 3.7 billion mobile health application downloads that year by 1.7 billion smartphone users worldwide.2 As of October 2020, 48,000-plus health apps were available on the iOS mobile operating system alone.3
For patients and clinicians, picking the most suitable apps can be challenging in the face of evolving clinical evidence, emerging privacy risks, functionality concerns, and the fact that apps constantly update and change. Many have relied on star rating systems and user reviews in app stores to guide their selection process despite mounting evidence that suggests that such evaluation methods are misleading, not always addressing such important parameters as usability, validity, security, and privacy.4,5
Approaches for evaluating medical apps
Many app evaluation frameworks have emerged, but none is universally accepted within the health care field.
The American Psychiatric Association’s (APA) App Evaluation Model represents a comprehensive resource to consider when evaluating medical apps. It stratifies numerous variables into 5 levels that form a pyramid. In this model, background information forms the base of this pyramid and includes factors such as business model, credibility, cost, and advertising of the app. The top of the pyramid is comprised of data integration that considers data ownership and therapeutic alliance.6 Although this model is beneficial in that it provides a framework, it is not practical for point-of-care purposes as it offers no objective way to rate or score an app for quick and easy comparison.
The privately owned and operated Health On the Net (HON) Foundation is well known for its HONcode, an ethical standard for quality medical information on the internet. It uses 8 principles to certify a health website. However, the HON website itself states that it cannot guarantee the accuracy or completeness of medical information presented by a site.7 Although HON certification by a website is a sign of good intention, it is not beneficial to the practicing clinician who is looking to use an app to directly assist in clinical care.
The Agency for Healthcare Research and Quality (AHRQ) is another well-respected body that has delineated essential details to consider when using a health website. The AHRQ identifies features (similar to those of the APA pyramid and HONcode) for users to consider, such as credibility, content, design, and disclosures.8 However, this model too lacks a concise user-friendly evaluating system.
Although the FDA plans to apply some regulatory authority to the evaluation of a certain subset of high-risk mobile medical apps, it is not planning to evaluate or regulate many of the medical apps that clinicians use in daily practice. This leaves us, and our patients, to be guided by the principle of caveat emptor, or “let the buyer beware.”
Thus, Dr. Chalas’ Presidential Task Force carefully considered various resources to provide a useful tool that would help obstetrician-gynecologists objectively vet a medical app in practice.
Continue to: The Task Force’s recommended rubric...
The Task Force’s recommended rubric
The rubric shown for evaluating mobile drug information apps was developed by the American Society of Health-System Pharmacists (ASHP). The ASHP rubric takes into account the criteria recognized by the APA pyramid, the HON Foundation, and the AHRQ and incorporates them into a user-friendly tool and scoring system that can be applied as an evaluation checklist.9 This tool is meant to aid clinicians in evaluating medical apps, but it ultimately is the user’s decision to determine if an app’s deficiencies should deter its use.
While all of the criteria are relevant and important, it is incumbent on us as medical experts to pay careful attention to the accuracy, authority, objectivity, timeliness, and security of any app we consider incorporating into clinical practice. A low score on these criteria would belie any perceived usefulness or value the app may have.
When applying the rubric to evaluate the quality of an app, we should be mindful of the primary user and which characteristics are more important than others to effect positive changes in health. For example, in addressing obesity, it is the patient who will be interacting with the app. Therefore, it’s important that the app should score, on a 1- to 4-point scale (1 point being major deficiencies, 4 points being no deficiencies), a 4 out of 4 on features like usefulness, functionality, and design. Coveted design features that enhance the user’s experience will appeal to patients and keep them engaged and motivated. However, when addressing a woman’s health with respect to cancer risk, the principal features on which the app should score 4 out of 4 would be authority, objectivity, timeliness, and accuracy.
In the upcoming articles in this series, a member of the Presidential Task Force will reference the ASHP rubric to guide clinicians in choosing apps to address one of the critical health areas with their patients. The author of the piece will highlight key features of an app to consider what would add the most value in incorporating its use in clinical practice.
It would be impossible to evaluate all health care apps even if we focused only on the medical apps relevant to obstetrics and gynecology. There is much value in having a framework for efficiently measuring an app’s benefit in clinical practice. The objective of this article series is to help clinicians Revisit the Visit by providing an effective tool to evaluate a medical app. ●
Personalizing care is at the heart of the American College of Obstetricians and Gynecologists (ACOG) 2020–2021 President Dr. Eva Chalas’ initiative to “Revisit the Visit.” As obstetrician-gynecologists, we care for patients across the entirety of their life. This role gives us the opportunity to form long-term partnerships with women to address important preventive health care measures.
Dr. Chalas established a Presidential Task Force that identified 5 areas of preventive health that significantly influence the long-term morbidity of women: obesity, cardiovascular disease, preconception counseling, diabetes, and cancer risk. The annual visit can serve as a particularly impactful point of care to achieve specific preventive care objectives and offer mitigation strategies based on patient-specific risk factors. We are uniquely positioned to identify and initiate the conversation and subsequently manage, treat, and address these critical health areas.
Harnessing modern technology
To adopt these health topics into practice, we need improved, more effective tools both to increase productivity during the office visit and to provide more personalized care. Notably, the widespread adoption of and proliferation of mobile devices—and the medical apps accessible on them—is creating new and innovative ways to improve health and health care delivery. More than 90% of physicians use a smartphone at work, and 62% of smartphone users have used their device to gather health data.1
In addition, according to a US Food and Drug Administration (FDA) report, in 2017, 325,000 health care applications were available on smartphones; this equates to an expected 3.7 billion mobile health application downloads that year by 1.7 billion smartphone users worldwide.2 As of October 2020, 48,000-plus health apps were available on the iOS mobile operating system alone.3
For patients and clinicians, picking the most suitable apps can be challenging in the face of evolving clinical evidence, emerging privacy risks, functionality concerns, and the fact that apps constantly update and change. Many have relied on star rating systems and user reviews in app stores to guide their selection process despite mounting evidence that suggests that such evaluation methods are misleading, not always addressing such important parameters as usability, validity, security, and privacy.4,5
Approaches for evaluating medical apps
Many app evaluation frameworks have emerged, but none is universally accepted within the health care field.
The American Psychiatric Association’s (APA) App Evaluation Model represents a comprehensive resource to consider when evaluating medical apps. It stratifies numerous variables into 5 levels that form a pyramid. In this model, background information forms the base of this pyramid and includes factors such as business model, credibility, cost, and advertising of the app. The top of the pyramid is comprised of data integration that considers data ownership and therapeutic alliance.6 Although this model is beneficial in that it provides a framework, it is not practical for point-of-care purposes as it offers no objective way to rate or score an app for quick and easy comparison.
The privately owned and operated Health On the Net (HON) Foundation is well known for its HONcode, an ethical standard for quality medical information on the internet. It uses 8 principles to certify a health website. However, the HON website itself states that it cannot guarantee the accuracy or completeness of medical information presented by a site.7 Although HON certification by a website is a sign of good intention, it is not beneficial to the practicing clinician who is looking to use an app to directly assist in clinical care.
The Agency for Healthcare Research and Quality (AHRQ) is another well-respected body that has delineated essential details to consider when using a health website. The AHRQ identifies features (similar to those of the APA pyramid and HONcode) for users to consider, such as credibility, content, design, and disclosures.8 However, this model too lacks a concise user-friendly evaluating system.
Although the FDA plans to apply some regulatory authority to the evaluation of a certain subset of high-risk mobile medical apps, it is not planning to evaluate or regulate many of the medical apps that clinicians use in daily practice. This leaves us, and our patients, to be guided by the principle of caveat emptor, or “let the buyer beware.”
Thus, Dr. Chalas’ Presidential Task Force carefully considered various resources to provide a useful tool that would help obstetrician-gynecologists objectively vet a medical app in practice.
Continue to: The Task Force’s recommended rubric...
The Task Force’s recommended rubric
The rubric shown for evaluating mobile drug information apps was developed by the American Society of Health-System Pharmacists (ASHP). The ASHP rubric takes into account the criteria recognized by the APA pyramid, the HON Foundation, and the AHRQ and incorporates them into a user-friendly tool and scoring system that can be applied as an evaluation checklist.9 This tool is meant to aid clinicians in evaluating medical apps, but it ultimately is the user’s decision to determine if an app’s deficiencies should deter its use.
While all of the criteria are relevant and important, it is incumbent on us as medical experts to pay careful attention to the accuracy, authority, objectivity, timeliness, and security of any app we consider incorporating into clinical practice. A low score on these criteria would belie any perceived usefulness or value the app may have.
When applying the rubric to evaluate the quality of an app, we should be mindful of the primary user and which characteristics are more important than others to effect positive changes in health. For example, in addressing obesity, it is the patient who will be interacting with the app. Therefore, it’s important that the app should score, on a 1- to 4-point scale (1 point being major deficiencies, 4 points being no deficiencies), a 4 out of 4 on features like usefulness, functionality, and design. Coveted design features that enhance the user’s experience will appeal to patients and keep them engaged and motivated. However, when addressing a woman’s health with respect to cancer risk, the principal features on which the app should score 4 out of 4 would be authority, objectivity, timeliness, and accuracy.
In the upcoming articles in this series, a member of the Presidential Task Force will reference the ASHP rubric to guide clinicians in choosing apps to address one of the critical health areas with their patients. The author of the piece will highlight key features of an app to consider what would add the most value in incorporating its use in clinical practice.
It would be impossible to evaluate all health care apps even if we focused only on the medical apps relevant to obstetrics and gynecology. There is much value in having a framework for efficiently measuring an app’s benefit in clinical practice. The objective of this article series is to help clinicians Revisit the Visit by providing an effective tool to evaluate a medical app. ●
- Mobius MD website. 11 Surprising mobile health statistics. http://www.mobius.md/blog/2019/03/11-mobile-health -statistics/. Accessed January 19, 2021.
- US Food and Drug Administration website. Device software functions including medical applications. November 5, 2019. https://www.fda.gov/medical-devices/digital-health-center -excellence/device-software-functions-including-mobile -medical-applications. Accessed March 10, 2021.
- Statista website. Number of mHealth apps available in the Apple App Store from 1st quarter 2015 to 4th quarter 2020. https://www.statista.com/statistics/779910/health-apps -available-ios-worldwide/. Accessed January 19, 2021.
- Campbell L. Using star ratings to choose a medical app? There’s a better way. Healthline website. Updated August 3, 2018. http://healthline.com/health-news/using-ratings-to -choose-medical-app-theres-a-better-way. Accessed April 22, 2021.
- Levine DM, Co Z, Newmark LP, et al. Design and testing of a mobile health application rating tool. NPJ Digit Med. 2020;3:74.
- Torous JB, Chan SR, Gipson SY, et al. A hierarchical framework for evaluation and informed decision making regarding smartphone apps for clinical care. Psychiatr Serv. 2018;69:498-500.
- Health On the Net website. The commitment to reliable health and medical information on the internet. https:// www.hon.ch/HONcode/Patients/Visitor/visitor.html. Accessed January 19, 2021.
- Agency for Healthcare Research and Quality. Assessing the quality of internet health information. June 1999. http:// www.ahrq.gov/research/data/infoqual.html. Accessed April 22, 2021.
- Hanrahan C, Aungst TD, Cole S. Evaluating mobile medical applications. American Society of Health-System Pharmacists eReports. https://www.ashp.org/-/media/store-files /mobile-medical-apps.ashx. Accessed January 22, 2021.
- Mobius MD website. 11 Surprising mobile health statistics. http://www.mobius.md/blog/2019/03/11-mobile-health -statistics/. Accessed January 19, 2021.
- US Food and Drug Administration website. Device software functions including medical applications. November 5, 2019. https://www.fda.gov/medical-devices/digital-health-center -excellence/device-software-functions-including-mobile -medical-applications. Accessed March 10, 2021.
- Statista website. Number of mHealth apps available in the Apple App Store from 1st quarter 2015 to 4th quarter 2020. https://www.statista.com/statistics/779910/health-apps -available-ios-worldwide/. Accessed January 19, 2021.
- Campbell L. Using star ratings to choose a medical app? There’s a better way. Healthline website. Updated August 3, 2018. http://healthline.com/health-news/using-ratings-to -choose-medical-app-theres-a-better-way. Accessed April 22, 2021.
- Levine DM, Co Z, Newmark LP, et al. Design and testing of a mobile health application rating tool. NPJ Digit Med. 2020;3:74.
- Torous JB, Chan SR, Gipson SY, et al. A hierarchical framework for evaluation and informed decision making regarding smartphone apps for clinical care. Psychiatr Serv. 2018;69:498-500.
- Health On the Net website. The commitment to reliable health and medical information on the internet. https:// www.hon.ch/HONcode/Patients/Visitor/visitor.html. Accessed January 19, 2021.
- Agency for Healthcare Research and Quality. Assessing the quality of internet health information. June 1999. http:// www.ahrq.gov/research/data/infoqual.html. Accessed April 22, 2021.
- Hanrahan C, Aungst TD, Cole S. Evaluating mobile medical applications. American Society of Health-System Pharmacists eReports. https://www.ashp.org/-/media/store-files /mobile-medical-apps.ashx. Accessed January 22, 2021.