User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Trump could clean house at health agencies
Others may soon depart voluntarily. Politico reported in late October that more than two dozen political appointees had already left the U.S. Department Health and Human Services (HHS) since the start of the COVID-19 pandemic in February and that potentially dozens of the more than 100 in the department would leave if Trump was not reelected.
Trump hasn’t conceded, he is challenging the election results, and he has already fired his Defense Secretary, Mark Esper.
Among those possibly in Trump’s sights: HHS Secretary Alex Azar, US Food and Drug Administration (FDA) Commissioner Stephen Hahn, MD, Centers for Disease Control and Prevention (CDC) Director Robert Redfield, MD, and White House Coronavirus Task Force member Anthony Fauci, MD, who is also the director of the National Institutes of Allergy and Infectious Diseases.
Seema Verma, the administrator of the Centers for Medicare & Medicaid Services (CMS), is likely safe. According to Politico, Verma is expected to leave on her own terms.
Azar has had a long run as a Trump appointee. He took office in January 2018 and has been a staunch loyalist. But he’s frequently been the butt of grousing by Trump for not doing enough to help lower drug prices and for his handling of the coronavirus pandemic. Azar was initially in charge of the Trump virus effort but was quickly replaced by Vice President Mike Pence.
It was widely reported in late April that Trump was considering firing Azar, but the president called that “fake news” in a tweet.
Azar has complained about Hahn, who was confirmed in December 2019. According to Politico, Azar was looking into how to remove Hahn as commissioner because of the FDA’s battle with the White House over standards for emergency use authorization of a coronavirus vaccine.
In addition, Trump was infuriated by the agency’s insistence that it stick to the highest bar for an emergency approval. “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics. Obviously, they are hoping to delay the answer until after November 3rd,” Trump tweeted at Hahn.
Fauci on the firing line?
Most of the president’s ire has been directed at Fauci. As far back as April, Trump retweeted a call for Fauci’s firing. Twitter removed the original tweet but kept Trump’s comments on the original tweet.
The president has frequently questioned Fauci’s advice, sidelined him from task force meetings, and infrequently met with him. Trump called Fauci a “disaster” during a call with supporters in October, and then, at a campaign rally in November, intimated that he would fire the scientist after the election, according to The Washington Post.
But such a firing cannot be easily done. Some have speculated that Trump could pressure Fauci’s boss, Francis Collins, MD, PhD — the director of the National Institutes of Health (NIH), who is a political appointee — to get rid of him. But Collins would have to come up with a reason to fire Fauci. Because he is not a political appointee, Fauci is afforded a raft of protections given to civil service employees of the federal government.
To demote or fire Fauci, Collins would have to give him 30 days’ notice unless there’s a belief that he committed a crime. Fauci would have at least a week to offer evidence and affidavits in support of his service.
He’d also be entitled to legal representation, a written decision, and the specific reasons for the action being taken quickly. He could also request a hearing, and he’d be able to appeal any action to the Merit Systems Protection Board. The process could take months, if not years.
In late October, Trump issued an executive order that would reclassify certain federal employees so that they wouldn’t have such protections. But agencies have until mid-January to come up with lists of such workers, according to Government Executive.
Collins has been with NIH since 1993, when he headed the Human Genome Project and the National Human Genome Research Institute. Politico has speculated that Collins, 70, might retire if Trump was reelected. It’s unclear what he’ll do now.
Redfield, who has taken heat for his leadership from many in public health — and was asked in October to stand up to Trump by former CDC Director William H. Foege, MD — has been openly contradicted by the president on more than one occasion, according to The New York Times.
In September, The Hill reported that Trump told reporters that he’d chastised Redfield by phone soon after Redfield had told a Senate committee that a coronavirus vaccine would not be available until mid-2021.
This article first appeared on Medscape.com.
Others may soon depart voluntarily. Politico reported in late October that more than two dozen political appointees had already left the U.S. Department Health and Human Services (HHS) since the start of the COVID-19 pandemic in February and that potentially dozens of the more than 100 in the department would leave if Trump was not reelected.
Trump hasn’t conceded, he is challenging the election results, and he has already fired his Defense Secretary, Mark Esper.
Among those possibly in Trump’s sights: HHS Secretary Alex Azar, US Food and Drug Administration (FDA) Commissioner Stephen Hahn, MD, Centers for Disease Control and Prevention (CDC) Director Robert Redfield, MD, and White House Coronavirus Task Force member Anthony Fauci, MD, who is also the director of the National Institutes of Allergy and Infectious Diseases.
Seema Verma, the administrator of the Centers for Medicare & Medicaid Services (CMS), is likely safe. According to Politico, Verma is expected to leave on her own terms.
Azar has had a long run as a Trump appointee. He took office in January 2018 and has been a staunch loyalist. But he’s frequently been the butt of grousing by Trump for not doing enough to help lower drug prices and for his handling of the coronavirus pandemic. Azar was initially in charge of the Trump virus effort but was quickly replaced by Vice President Mike Pence.
It was widely reported in late April that Trump was considering firing Azar, but the president called that “fake news” in a tweet.
Azar has complained about Hahn, who was confirmed in December 2019. According to Politico, Azar was looking into how to remove Hahn as commissioner because of the FDA’s battle with the White House over standards for emergency use authorization of a coronavirus vaccine.
In addition, Trump was infuriated by the agency’s insistence that it stick to the highest bar for an emergency approval. “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics. Obviously, they are hoping to delay the answer until after November 3rd,” Trump tweeted at Hahn.
Fauci on the firing line?
Most of the president’s ire has been directed at Fauci. As far back as April, Trump retweeted a call for Fauci’s firing. Twitter removed the original tweet but kept Trump’s comments on the original tweet.
The president has frequently questioned Fauci’s advice, sidelined him from task force meetings, and infrequently met with him. Trump called Fauci a “disaster” during a call with supporters in October, and then, at a campaign rally in November, intimated that he would fire the scientist after the election, according to The Washington Post.
But such a firing cannot be easily done. Some have speculated that Trump could pressure Fauci’s boss, Francis Collins, MD, PhD — the director of the National Institutes of Health (NIH), who is a political appointee — to get rid of him. But Collins would have to come up with a reason to fire Fauci. Because he is not a political appointee, Fauci is afforded a raft of protections given to civil service employees of the federal government.
To demote or fire Fauci, Collins would have to give him 30 days’ notice unless there’s a belief that he committed a crime. Fauci would have at least a week to offer evidence and affidavits in support of his service.
He’d also be entitled to legal representation, a written decision, and the specific reasons for the action being taken quickly. He could also request a hearing, and he’d be able to appeal any action to the Merit Systems Protection Board. The process could take months, if not years.
In late October, Trump issued an executive order that would reclassify certain federal employees so that they wouldn’t have such protections. But agencies have until mid-January to come up with lists of such workers, according to Government Executive.
Collins has been with NIH since 1993, when he headed the Human Genome Project and the National Human Genome Research Institute. Politico has speculated that Collins, 70, might retire if Trump was reelected. It’s unclear what he’ll do now.
Redfield, who has taken heat for his leadership from many in public health — and was asked in October to stand up to Trump by former CDC Director William H. Foege, MD — has been openly contradicted by the president on more than one occasion, according to The New York Times.
In September, The Hill reported that Trump told reporters that he’d chastised Redfield by phone soon after Redfield had told a Senate committee that a coronavirus vaccine would not be available until mid-2021.
This article first appeared on Medscape.com.
Others may soon depart voluntarily. Politico reported in late October that more than two dozen political appointees had already left the U.S. Department Health and Human Services (HHS) since the start of the COVID-19 pandemic in February and that potentially dozens of the more than 100 in the department would leave if Trump was not reelected.
Trump hasn’t conceded, he is challenging the election results, and he has already fired his Defense Secretary, Mark Esper.
Among those possibly in Trump’s sights: HHS Secretary Alex Azar, US Food and Drug Administration (FDA) Commissioner Stephen Hahn, MD, Centers for Disease Control and Prevention (CDC) Director Robert Redfield, MD, and White House Coronavirus Task Force member Anthony Fauci, MD, who is also the director of the National Institutes of Allergy and Infectious Diseases.
Seema Verma, the administrator of the Centers for Medicare & Medicaid Services (CMS), is likely safe. According to Politico, Verma is expected to leave on her own terms.
Azar has had a long run as a Trump appointee. He took office in January 2018 and has been a staunch loyalist. But he’s frequently been the butt of grousing by Trump for not doing enough to help lower drug prices and for his handling of the coronavirus pandemic. Azar was initially in charge of the Trump virus effort but was quickly replaced by Vice President Mike Pence.
It was widely reported in late April that Trump was considering firing Azar, but the president called that “fake news” in a tweet.
Azar has complained about Hahn, who was confirmed in December 2019. According to Politico, Azar was looking into how to remove Hahn as commissioner because of the FDA’s battle with the White House over standards for emergency use authorization of a coronavirus vaccine.
In addition, Trump was infuriated by the agency’s insistence that it stick to the highest bar for an emergency approval. “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics. Obviously, they are hoping to delay the answer until after November 3rd,” Trump tweeted at Hahn.
Fauci on the firing line?
Most of the president’s ire has been directed at Fauci. As far back as April, Trump retweeted a call for Fauci’s firing. Twitter removed the original tweet but kept Trump’s comments on the original tweet.
The president has frequently questioned Fauci’s advice, sidelined him from task force meetings, and infrequently met with him. Trump called Fauci a “disaster” during a call with supporters in October, and then, at a campaign rally in November, intimated that he would fire the scientist after the election, according to The Washington Post.
But such a firing cannot be easily done. Some have speculated that Trump could pressure Fauci’s boss, Francis Collins, MD, PhD — the director of the National Institutes of Health (NIH), who is a political appointee — to get rid of him. But Collins would have to come up with a reason to fire Fauci. Because he is not a political appointee, Fauci is afforded a raft of protections given to civil service employees of the federal government.
To demote or fire Fauci, Collins would have to give him 30 days’ notice unless there’s a belief that he committed a crime. Fauci would have at least a week to offer evidence and affidavits in support of his service.
He’d also be entitled to legal representation, a written decision, and the specific reasons for the action being taken quickly. He could also request a hearing, and he’d be able to appeal any action to the Merit Systems Protection Board. The process could take months, if not years.
In late October, Trump issued an executive order that would reclassify certain federal employees so that they wouldn’t have such protections. But agencies have until mid-January to come up with lists of such workers, according to Government Executive.
Collins has been with NIH since 1993, when he headed the Human Genome Project and the National Human Genome Research Institute. Politico has speculated that Collins, 70, might retire if Trump was reelected. It’s unclear what he’ll do now.
Redfield, who has taken heat for his leadership from many in public health — and was asked in October to stand up to Trump by former CDC Director William H. Foege, MD — has been openly contradicted by the president on more than one occasion, according to The New York Times.
In September, The Hill reported that Trump told reporters that he’d chastised Redfield by phone soon after Redfield had told a Senate committee that a coronavirus vaccine would not be available until mid-2021.
This article first appeared on Medscape.com.
Supreme Court Justices seem skeptical of case to overturn ACA
The Justices conducted arguments by telephone in the case, California v Texas (previously California v US), which was brought by 18 Republican state officials and two individual plaintiffs. The Trump administration joined the plaintiffs in June, arguing that the entire law should be overturned. The ACA is being defended by Democratic state officials from 16 states and Washington, D.C.
The Republican plaintiffs have essentially argued that the ACA cannot stand without the individual mandate requirement – that it is not possible to “sever” it from the rest of the Act. In 2017, Congress set the tax penalty to $0 if an individual did not buy insurance. The mandate to buy insurance was left in place, but there were no longer any consequences. The plaintiffs said that congressional act was equivalent to severing the mandate.
But many Justices appeared to take a dim view of that argument.
“It’s a very straightforward case for severability under our precedents,” said Justice Brett Kavanaugh. “Meaning that we would excise the mandate and leave the rest of the Act in play. Congress knows how to write an inseverability clause and that is not the language that they chose here,” he said.
Justice Elena Kagan also questioned how it would jibe with legal precedent to allow the severing of one part of a law when there was no clear instruction from Congress on the issue. She also raised the concern that it would open the door to all sorts of challenges.
“It would seem a big deal to say that, if you can point to injury with respect to one provision and you can concoct some kind of inseverability argument, that allows you to challenge anything else in the statute,” she said.
“Isn’t that something that really cuts against all of our doctrine?” asked Kagan.
“I think it’s hard for you to argue that Congress intended the entire Act to fall if the mandate was struck down when the same Congress that lowered the penalty to zero did not even try to repeal the rest of the act,” said Chief Justice John Roberts.
“I think, frankly, that they wanted the Court to do that but that’s not our job,” he added.
Proof of harm?
To have the standing to sue, the plaintiffs have to prove they have been harmed by the ACA. Texas Solicitor General Kyle Hawkins said that individuals feel compelled to buy insurance – even without a penalty hanging over their heads.
Justice Stephen Breyer argued that many laws include what he called “precatory” language – that is, they seek to compel citizens to do something. But most don’t penalize those who fail to act – just like the ACA currently.
If, as the Texas plaintiffs argued, it’s still unconstitutional to make such a request, “I think there will be an awful lot of language in an awful lot of statutes that will suddenly be the subject of court constitutional challenge,” he said.
Hawkins disagreed. He said the ACA’s mandate “is not some suggestion, not some hortatory statement. It is the law of the United States of America today that you have to purchase health insurance and not just any health insurance, but health insurance that the federal government has decided would be best for you.”
Hawkins said that, if just one additional person signed up for Medicaid, the state of Texas and the other plaintiff states would be harmed. He said people were continuing to enroll in the program because they believed the law required them to get health insurance.
Justice Sonia Sotomayor said that defied common sense. “The problem is that your theory assumes people that people are going to pay a tax and break the law by not buying insurance, but they wouldn’t do it when the tax is zero.”
What’s at stake
It’s unlikely the justices will issue a decision immediately. They have until the end of the term in June to rule.
Katie Keith, JD, MPH, a principal at Keith Policy Solutions, LLC, outlined the potential outcomes in Health Affairs .
“The most likely scenario is that the Court maintains the status quo,” she wrote. They could get there by deciding Texas et al. did not have standing to bring the case. Or they could decide that either the mandate is constitutional or that it is unconstitutional but can be severed from the rest of the ACA.
The Court could alternatively find that some or all of the law’s insurance provisions – such as protections for people with pre-existing conditions – can’t be severed from the mandate. Or the justices could strike down all of the insurance consumer protections, the health insurance marketplaces, premium tax credits, and other provisions, which would force states to come up with the money to help people buy insurance. And states are unlikely to be able to do so, especially with the pandemic stretching their budgets.
Finally, the Court could find that the mandate can’t be separated, which would essentially overturn the law.
If that happens, some 15 million people could lose Medicaid coverage, 11 million who buy on health insurance exchanges could lose coverage, and 2.3 million young adults would no longer be able to stay on parents’ policies, according to the Kaiser Family Foundation. Kaiser also estimates that 54 million people under age 65 who have pre-existing conditions would no longer be guaranteed coverage.
The Urban Institute estimates that 21 million people could lose insurance – 15 million through Medicaid and the Children’s Health Insurance Program (CHIP) and 7.6 million through private nongroup coverage.
Medical societies weigh in
Multiple physicians’ groups, patient advocates, and hospital organizations have filed briefs with the Court in favor of keeping the law intact.
Twenty patient groups representing millions with pre-existing conditions – including the American Cancer Society, American Diabetes Association, American Heart Association, National Alliance on Mental Illness, National Organization for Rare Disorders, and the Kennedy Forum – filed a court brief in May arguing that the law has expanded access to insurance and improved patient outcomes.
“The coronavirus pandemic has only served to underscore the necessity of meaningful coverage – especially for those who are at high risk of being severely affected by the virus – including countless Americans who have pre-existing, acute or chronic conditions like heart disease, cancer, diabetes, lung diseases and multiple sclerosis,” they said in a statement.
Jacqueline W. Fincher, MD, MACP, president of the American College of Physicians, which joined a court brief in support of the law with 19 other medical organizations, said the law has worked.
“The coverage, protections and benefits provided by the ACA are critical to the well-being of millions of Americans,” she said in a statement.
“If the ACA were to be thrown out at the same time that we face the pandemic, it would cause chaos for physicians and our patients, and for the entire health care system,” said Fincher, adding that millions of Americans who have been infected could lose insurance if protections for pre-existing conditions disappeared.
“The ACA has revolutionized access to care for tens of millions of women by helping them obtain meaningful health coverage, ensuring that essential care is covered by insurers, and protecting patients from unfair insurance practices,” said Maureen G. Phipps, MD, MPH, CEO of the American College of Obstetricians and Gynecologists (ACOG), in a statement.
Overturning the ACA “would be one of the most singularly disruptive acts to be committed during this public health crisis,” she said.
American Psychiatric Association President Jeffrey Geller, MD, MPH, also warned of disruptions to care, especially for those with mental health and substance use disorders. “We urge the Supreme Court to preserve the entire Act, including the individual mandate,” he said, in a statement.
“In the midst of COVID is no time to let down the millions who we serve as our patients,” said Chip Kahn, Federation of American Health Systems president and CEO, in a statement.
“As caregivers, the goal of hospitals for our patients is to see increased access to affordable coverage for all Americans – not new obstacles,” he said, adding that the ACA “can accomplish this goal. We hope the Supreme Court will see its way clear to allow it to go forward.”
For the defense
Many legal analysts on social media who listened in to today’s hearing agreed that the tenor of the proceedings seemed to lean toward survival of the ACA.
“At this point I would say it is *extremely* likely that the ACA will be upheld, but the mandate struck down and severed out,” tweeted Raffi Melkonian, an appellate lawyer in Houston, Texas. “A decision on standing (throwing out the case entirely) is also possible. The chance that the ACA is struck down v. low.”
“Both Kavanaugh and Roberts have suggested this morning that they may view the individual mandate as severable from the rest of the law. If those two justices join the court’s three liberals in finding that the mandate is severable, that would be five votes to save the ACA,” tweeted the analysts at SCOTUS Blog.
Sean Marotta, a lawyer with Hogan Lovells’ Supreme Court group, agreed. “Oral argument is always an imperfect measure, but the Act’s defenders should feel good today,” he tweeted.
This article first appeared on Medscape.com.
The Justices conducted arguments by telephone in the case, California v Texas (previously California v US), which was brought by 18 Republican state officials and two individual plaintiffs. The Trump administration joined the plaintiffs in June, arguing that the entire law should be overturned. The ACA is being defended by Democratic state officials from 16 states and Washington, D.C.
The Republican plaintiffs have essentially argued that the ACA cannot stand without the individual mandate requirement – that it is not possible to “sever” it from the rest of the Act. In 2017, Congress set the tax penalty to $0 if an individual did not buy insurance. The mandate to buy insurance was left in place, but there were no longer any consequences. The plaintiffs said that congressional act was equivalent to severing the mandate.
But many Justices appeared to take a dim view of that argument.
“It’s a very straightforward case for severability under our precedents,” said Justice Brett Kavanaugh. “Meaning that we would excise the mandate and leave the rest of the Act in play. Congress knows how to write an inseverability clause and that is not the language that they chose here,” he said.
Justice Elena Kagan also questioned how it would jibe with legal precedent to allow the severing of one part of a law when there was no clear instruction from Congress on the issue. She also raised the concern that it would open the door to all sorts of challenges.
“It would seem a big deal to say that, if you can point to injury with respect to one provision and you can concoct some kind of inseverability argument, that allows you to challenge anything else in the statute,” she said.
“Isn’t that something that really cuts against all of our doctrine?” asked Kagan.
“I think it’s hard for you to argue that Congress intended the entire Act to fall if the mandate was struck down when the same Congress that lowered the penalty to zero did not even try to repeal the rest of the act,” said Chief Justice John Roberts.
“I think, frankly, that they wanted the Court to do that but that’s not our job,” he added.
Proof of harm?
To have the standing to sue, the plaintiffs have to prove they have been harmed by the ACA. Texas Solicitor General Kyle Hawkins said that individuals feel compelled to buy insurance – even without a penalty hanging over their heads.
Justice Stephen Breyer argued that many laws include what he called “precatory” language – that is, they seek to compel citizens to do something. But most don’t penalize those who fail to act – just like the ACA currently.
If, as the Texas plaintiffs argued, it’s still unconstitutional to make such a request, “I think there will be an awful lot of language in an awful lot of statutes that will suddenly be the subject of court constitutional challenge,” he said.
Hawkins disagreed. He said the ACA’s mandate “is not some suggestion, not some hortatory statement. It is the law of the United States of America today that you have to purchase health insurance and not just any health insurance, but health insurance that the federal government has decided would be best for you.”
Hawkins said that, if just one additional person signed up for Medicaid, the state of Texas and the other plaintiff states would be harmed. He said people were continuing to enroll in the program because they believed the law required them to get health insurance.
Justice Sonia Sotomayor said that defied common sense. “The problem is that your theory assumes people that people are going to pay a tax and break the law by not buying insurance, but they wouldn’t do it when the tax is zero.”
What’s at stake
It’s unlikely the justices will issue a decision immediately. They have until the end of the term in June to rule.
Katie Keith, JD, MPH, a principal at Keith Policy Solutions, LLC, outlined the potential outcomes in Health Affairs .
“The most likely scenario is that the Court maintains the status quo,” she wrote. They could get there by deciding Texas et al. did not have standing to bring the case. Or they could decide that either the mandate is constitutional or that it is unconstitutional but can be severed from the rest of the ACA.
The Court could alternatively find that some or all of the law’s insurance provisions – such as protections for people with pre-existing conditions – can’t be severed from the mandate. Or the justices could strike down all of the insurance consumer protections, the health insurance marketplaces, premium tax credits, and other provisions, which would force states to come up with the money to help people buy insurance. And states are unlikely to be able to do so, especially with the pandemic stretching their budgets.
Finally, the Court could find that the mandate can’t be separated, which would essentially overturn the law.
If that happens, some 15 million people could lose Medicaid coverage, 11 million who buy on health insurance exchanges could lose coverage, and 2.3 million young adults would no longer be able to stay on parents’ policies, according to the Kaiser Family Foundation. Kaiser also estimates that 54 million people under age 65 who have pre-existing conditions would no longer be guaranteed coverage.
The Urban Institute estimates that 21 million people could lose insurance – 15 million through Medicaid and the Children’s Health Insurance Program (CHIP) and 7.6 million through private nongroup coverage.
Medical societies weigh in
Multiple physicians’ groups, patient advocates, and hospital organizations have filed briefs with the Court in favor of keeping the law intact.
Twenty patient groups representing millions with pre-existing conditions – including the American Cancer Society, American Diabetes Association, American Heart Association, National Alliance on Mental Illness, National Organization for Rare Disorders, and the Kennedy Forum – filed a court brief in May arguing that the law has expanded access to insurance and improved patient outcomes.
“The coronavirus pandemic has only served to underscore the necessity of meaningful coverage – especially for those who are at high risk of being severely affected by the virus – including countless Americans who have pre-existing, acute or chronic conditions like heart disease, cancer, diabetes, lung diseases and multiple sclerosis,” they said in a statement.
Jacqueline W. Fincher, MD, MACP, president of the American College of Physicians, which joined a court brief in support of the law with 19 other medical organizations, said the law has worked.
“The coverage, protections and benefits provided by the ACA are critical to the well-being of millions of Americans,” she said in a statement.
“If the ACA were to be thrown out at the same time that we face the pandemic, it would cause chaos for physicians and our patients, and for the entire health care system,” said Fincher, adding that millions of Americans who have been infected could lose insurance if protections for pre-existing conditions disappeared.
“The ACA has revolutionized access to care for tens of millions of women by helping them obtain meaningful health coverage, ensuring that essential care is covered by insurers, and protecting patients from unfair insurance practices,” said Maureen G. Phipps, MD, MPH, CEO of the American College of Obstetricians and Gynecologists (ACOG), in a statement.
Overturning the ACA “would be one of the most singularly disruptive acts to be committed during this public health crisis,” she said.
American Psychiatric Association President Jeffrey Geller, MD, MPH, also warned of disruptions to care, especially for those with mental health and substance use disorders. “We urge the Supreme Court to preserve the entire Act, including the individual mandate,” he said, in a statement.
“In the midst of COVID is no time to let down the millions who we serve as our patients,” said Chip Kahn, Federation of American Health Systems president and CEO, in a statement.
“As caregivers, the goal of hospitals for our patients is to see increased access to affordable coverage for all Americans – not new obstacles,” he said, adding that the ACA “can accomplish this goal. We hope the Supreme Court will see its way clear to allow it to go forward.”
For the defense
Many legal analysts on social media who listened in to today’s hearing agreed that the tenor of the proceedings seemed to lean toward survival of the ACA.
“At this point I would say it is *extremely* likely that the ACA will be upheld, but the mandate struck down and severed out,” tweeted Raffi Melkonian, an appellate lawyer in Houston, Texas. “A decision on standing (throwing out the case entirely) is also possible. The chance that the ACA is struck down v. low.”
“Both Kavanaugh and Roberts have suggested this morning that they may view the individual mandate as severable from the rest of the law. If those two justices join the court’s three liberals in finding that the mandate is severable, that would be five votes to save the ACA,” tweeted the analysts at SCOTUS Blog.
Sean Marotta, a lawyer with Hogan Lovells’ Supreme Court group, agreed. “Oral argument is always an imperfect measure, but the Act’s defenders should feel good today,” he tweeted.
This article first appeared on Medscape.com.
The Justices conducted arguments by telephone in the case, California v Texas (previously California v US), which was brought by 18 Republican state officials and two individual plaintiffs. The Trump administration joined the plaintiffs in June, arguing that the entire law should be overturned. The ACA is being defended by Democratic state officials from 16 states and Washington, D.C.
The Republican plaintiffs have essentially argued that the ACA cannot stand without the individual mandate requirement – that it is not possible to “sever” it from the rest of the Act. In 2017, Congress set the tax penalty to $0 if an individual did not buy insurance. The mandate to buy insurance was left in place, but there were no longer any consequences. The plaintiffs said that congressional act was equivalent to severing the mandate.
But many Justices appeared to take a dim view of that argument.
“It’s a very straightforward case for severability under our precedents,” said Justice Brett Kavanaugh. “Meaning that we would excise the mandate and leave the rest of the Act in play. Congress knows how to write an inseverability clause and that is not the language that they chose here,” he said.
Justice Elena Kagan also questioned how it would jibe with legal precedent to allow the severing of one part of a law when there was no clear instruction from Congress on the issue. She also raised the concern that it would open the door to all sorts of challenges.
“It would seem a big deal to say that, if you can point to injury with respect to one provision and you can concoct some kind of inseverability argument, that allows you to challenge anything else in the statute,” she said.
“Isn’t that something that really cuts against all of our doctrine?” asked Kagan.
“I think it’s hard for you to argue that Congress intended the entire Act to fall if the mandate was struck down when the same Congress that lowered the penalty to zero did not even try to repeal the rest of the act,” said Chief Justice John Roberts.
“I think, frankly, that they wanted the Court to do that but that’s not our job,” he added.
Proof of harm?
To have the standing to sue, the plaintiffs have to prove they have been harmed by the ACA. Texas Solicitor General Kyle Hawkins said that individuals feel compelled to buy insurance – even without a penalty hanging over their heads.
Justice Stephen Breyer argued that many laws include what he called “precatory” language – that is, they seek to compel citizens to do something. But most don’t penalize those who fail to act – just like the ACA currently.
If, as the Texas plaintiffs argued, it’s still unconstitutional to make such a request, “I think there will be an awful lot of language in an awful lot of statutes that will suddenly be the subject of court constitutional challenge,” he said.
Hawkins disagreed. He said the ACA’s mandate “is not some suggestion, not some hortatory statement. It is the law of the United States of America today that you have to purchase health insurance and not just any health insurance, but health insurance that the federal government has decided would be best for you.”
Hawkins said that, if just one additional person signed up for Medicaid, the state of Texas and the other plaintiff states would be harmed. He said people were continuing to enroll in the program because they believed the law required them to get health insurance.
Justice Sonia Sotomayor said that defied common sense. “The problem is that your theory assumes people that people are going to pay a tax and break the law by not buying insurance, but they wouldn’t do it when the tax is zero.”
What’s at stake
It’s unlikely the justices will issue a decision immediately. They have until the end of the term in June to rule.
Katie Keith, JD, MPH, a principal at Keith Policy Solutions, LLC, outlined the potential outcomes in Health Affairs .
“The most likely scenario is that the Court maintains the status quo,” she wrote. They could get there by deciding Texas et al. did not have standing to bring the case. Or they could decide that either the mandate is constitutional or that it is unconstitutional but can be severed from the rest of the ACA.
The Court could alternatively find that some or all of the law’s insurance provisions – such as protections for people with pre-existing conditions – can’t be severed from the mandate. Or the justices could strike down all of the insurance consumer protections, the health insurance marketplaces, premium tax credits, and other provisions, which would force states to come up with the money to help people buy insurance. And states are unlikely to be able to do so, especially with the pandemic stretching their budgets.
Finally, the Court could find that the mandate can’t be separated, which would essentially overturn the law.
If that happens, some 15 million people could lose Medicaid coverage, 11 million who buy on health insurance exchanges could lose coverage, and 2.3 million young adults would no longer be able to stay on parents’ policies, according to the Kaiser Family Foundation. Kaiser also estimates that 54 million people under age 65 who have pre-existing conditions would no longer be guaranteed coverage.
The Urban Institute estimates that 21 million people could lose insurance – 15 million through Medicaid and the Children’s Health Insurance Program (CHIP) and 7.6 million through private nongroup coverage.
Medical societies weigh in
Multiple physicians’ groups, patient advocates, and hospital organizations have filed briefs with the Court in favor of keeping the law intact.
Twenty patient groups representing millions with pre-existing conditions – including the American Cancer Society, American Diabetes Association, American Heart Association, National Alliance on Mental Illness, National Organization for Rare Disorders, and the Kennedy Forum – filed a court brief in May arguing that the law has expanded access to insurance and improved patient outcomes.
“The coronavirus pandemic has only served to underscore the necessity of meaningful coverage – especially for those who are at high risk of being severely affected by the virus – including countless Americans who have pre-existing, acute or chronic conditions like heart disease, cancer, diabetes, lung diseases and multiple sclerosis,” they said in a statement.
Jacqueline W. Fincher, MD, MACP, president of the American College of Physicians, which joined a court brief in support of the law with 19 other medical organizations, said the law has worked.
“The coverage, protections and benefits provided by the ACA are critical to the well-being of millions of Americans,” she said in a statement.
“If the ACA were to be thrown out at the same time that we face the pandemic, it would cause chaos for physicians and our patients, and for the entire health care system,” said Fincher, adding that millions of Americans who have been infected could lose insurance if protections for pre-existing conditions disappeared.
“The ACA has revolutionized access to care for tens of millions of women by helping them obtain meaningful health coverage, ensuring that essential care is covered by insurers, and protecting patients from unfair insurance practices,” said Maureen G. Phipps, MD, MPH, CEO of the American College of Obstetricians and Gynecologists (ACOG), in a statement.
Overturning the ACA “would be one of the most singularly disruptive acts to be committed during this public health crisis,” she said.
American Psychiatric Association President Jeffrey Geller, MD, MPH, also warned of disruptions to care, especially for those with mental health and substance use disorders. “We urge the Supreme Court to preserve the entire Act, including the individual mandate,” he said, in a statement.
“In the midst of COVID is no time to let down the millions who we serve as our patients,” said Chip Kahn, Federation of American Health Systems president and CEO, in a statement.
“As caregivers, the goal of hospitals for our patients is to see increased access to affordable coverage for all Americans – not new obstacles,” he said, adding that the ACA “can accomplish this goal. We hope the Supreme Court will see its way clear to allow it to go forward.”
For the defense
Many legal analysts on social media who listened in to today’s hearing agreed that the tenor of the proceedings seemed to lean toward survival of the ACA.
“At this point I would say it is *extremely* likely that the ACA will be upheld, but the mandate struck down and severed out,” tweeted Raffi Melkonian, an appellate lawyer in Houston, Texas. “A decision on standing (throwing out the case entirely) is also possible. The chance that the ACA is struck down v. low.”
“Both Kavanaugh and Roberts have suggested this morning that they may view the individual mandate as severable from the rest of the law. If those two justices join the court’s three liberals in finding that the mandate is severable, that would be five votes to save the ACA,” tweeted the analysts at SCOTUS Blog.
Sean Marotta, a lawyer with Hogan Lovells’ Supreme Court group, agreed. “Oral argument is always an imperfect measure, but the Act’s defenders should feel good today,” he tweeted.
This article first appeared on Medscape.com.
FDA grants emergency use authorization to Lilly’s antibody COVID-19 therapy
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
The monoclonal antibody therapy has emergency authorization for treating patients who have tested positive for SARS-CoV-2 infection and who are considered to be at high risk for progression to severe COVID-19 or hospitalization. To be eligible for treatment with bamlanivimab, patients must be at least 12 years of age and weigh at least 40 kg (approximately 88 lb). The agency notes that this includes patients aged 65 years and older or people with certain chronic conditions.
Bamlanivimab is not authorized for use in patients who are hospitalized or who require oxygen therapy because of COVID-19. The FDA’s action comes less than 2 weeks after Eli Lilly halted the ACTIV-3 study of the therapy for severe, hospitalized COVID-19 patients after evidence showed that adding the antibody therapy to standard care did not improve outcomes over standard care alone for patients with advanced COVID-19.
The government contract with Eli Lilly involves the purchase of 300,000 doses through December, with the option to procure another 650,000 doses through June 2021.
Because of Operation Warp Speed, “we have supplies to distribute now. Product distribution will begin this week,” US Health & Human Services (HHS) Secretary Alex Azar said at a news conference today.
“We talked about building the bridge to safe and effective vaccines” for COVID-19, Azar added. “With this therapeutic, the bridge is taking shape.”
Bamlanivimab 700 mg will be administered as a 1-hour infusion followed by a 1-hour observation period for detecting any infusion-related side effects. The authorized dose is 700 mg, which was on the lower end of the dose range evaluated in studies.
During the press conference, a reporter asked whether the lower dose was chosen in order that more doses of the antibody could be made available. “The lower dose is a rational choice in this situation because we don’t want to give more of a drug than you need,” said Janet Woodcock, MD, the therapeutics lead for Operation Warp Speed. “I think we could probably go lower.”
Bamlanivimab works by attaching to the virus and blocking its entry into the cells and possibly by helping the patients’ immune system clear the virus, said Woodcock, who is also director of the FDA’s Center for Drug Evaluation and Research.
“The goal is to treat high-risk people as soon as possible after they show symptoms and are diagnosed,” she added.
Infusions an initial challenge?
There could be some logistic challenges at first because the antibody is administered via infusion. “We expect there will initially be a challenge in administering ... these infusions and setting up infusion centers,” Woodcock said.
Outpatient intravenous infusions are normally performed at infusion centers for patients with cancer and immune disorders, she noted. “You really don’t want them mixing with people who have COVID-19 disease, so we will need to set up separate sites.”
Bamlanivimab will be provided free of cost to patients, Azar said. Patients should be aware that coinsurance may be required for the infusion.
“Fair and equitable” distribution planned
During phase 1 of distribution, the agent will first be allocated to hospitals and hospital-affiliated locations only, John Redd, MD, MPH, chief medical officer, Office of the Assistant Secretary for Preparedness and Response at HHS, said at the press conference.
During phase 2, “there will be expanded distribution to outpatient sites,” he said. In an effort to keep the process transparent, a new website features the latest updates on the distribution of bamlanivimab.
Allocation will be based on two factors: the number of new cases reported in a state or territory in the prior 7 days, and rates of COVID-19 hospitalization during the same period.
Asked why the government would determine distribution of the antibody on the basis of the number of hospitalized patients when the indication includes prevention of admission, Woodcock replied that hospitalization is a surrogate measure that can reflect risk factors in a particular state population, such as obesity, diabetes, or the proportion of older people.
Furthermore, the confirmed cases are a “leading indicator,” she said, that can help identify a steep rise in COVID-19 cases that could indicate more hospitalizations are likely soon. “We don’t want to miss that.”
Data underlying the EUA decision
A decrease in hospitalizations or emergency department visits within 28 days of treatment in preclinical studies was “the most important evidence that bamlanivimab may be effective,” the agency noted in the press release announcing the EUA. Among patients at high risk for progression, 3% required such interventions, compared with 10% of placebo-treated patients.
Potential side effects of bamlanivimab include anaphylaxis, infusion-related reactions, nausea, diarrhea, dizziness, headache, itching, and vomiting.
“As illustrated by today’s action, the FDA remains committed to expediting the development and availability of potential COVID-19 treatments and providing sick patients timely access to new therapies where appropriate,” FDA Commissioner Stephen M. Hahn, MD, said in the news release.
Healthcare providers can download a detailed FDA fact sheet on the EUA for bamlanivimab, which includes dosing instructions.
This article first appeared on Medscape.com.
United States adds nearly 74,000 more children with COVID-19
The new weekly high for COVID-19 cases in children announced last week has been surpassed already, as the United States experienced almost 74,000 new pediatric cases for the week ending Nov. 5, according to the American Academy of Pediatrics and the Children’s Hospital Association.
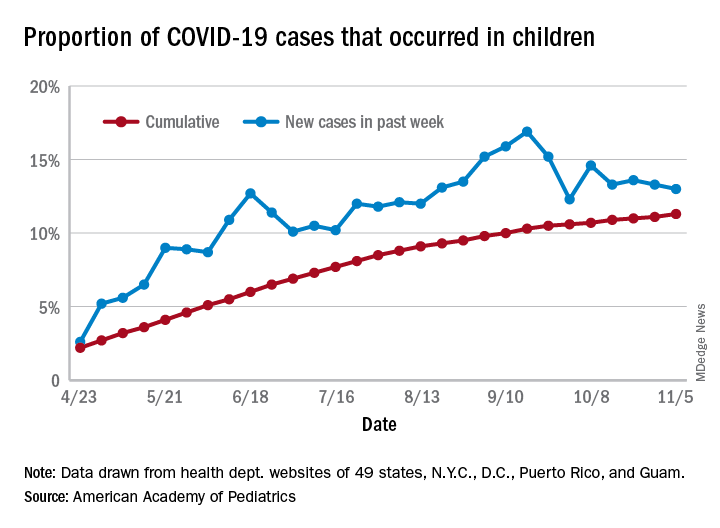
The total number of COVID-19 cases in children is now 927,518 in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA said in their weekly report.
Cumulatively, children represent 11.3% of all COVID-19 cases in those jurisdictions, up from 11.1% a week ago. For just the past week, those 73,883 children represent 13.0% of the 567,672 new cases reported among all ages. That proportion peaked at 16.9% in mid-September, the AAP/CHA data show.
Dropping down to the state level, cumulative proportions as of Nov. 5 range from 5.2% in New Jersey to 23.3% in Wyoming, with 11 other states over 15%. California has had more cases, 100,856, than any other state, and Vermont the fewest at 329, the AAP and CHA said.
The national rate per 100,000 children is now 1,232, up from 1,134 the previous week and more than doubled since mid-August (582.2 per 100,000 on Aug. 20). North Dakota’s rate of 3,990 per 100,000 children is the highest of any state (South Dakota is next at 2,779), while Vermont is again the lowest at 245 per 100,000, based on data collected from state health department websites.
Two COVID-19–related deaths in children were reported during the week ending Nov. 5, bringing the total to 123 but leaving the overall proportion of deaths in children unchanged at 0.06% of all deaths. Texas has reported the most COVID-19 deaths in children with 29, while 15 states have recorded no deaths so far (mortality data in children reported by 42 states and New York City), the AAP and CHA said.
The new weekly high for COVID-19 cases in children announced last week has been surpassed already, as the United States experienced almost 74,000 new pediatric cases for the week ending Nov. 5, according to the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of COVID-19 cases in children is now 927,518 in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA said in their weekly report.
Cumulatively, children represent 11.3% of all COVID-19 cases in those jurisdictions, up from 11.1% a week ago. For just the past week, those 73,883 children represent 13.0% of the 567,672 new cases reported among all ages. That proportion peaked at 16.9% in mid-September, the AAP/CHA data show.
Dropping down to the state level, cumulative proportions as of Nov. 5 range from 5.2% in New Jersey to 23.3% in Wyoming, with 11 other states over 15%. California has had more cases, 100,856, than any other state, and Vermont the fewest at 329, the AAP and CHA said.
The national rate per 100,000 children is now 1,232, up from 1,134 the previous week and more than doubled since mid-August (582.2 per 100,000 on Aug. 20). North Dakota’s rate of 3,990 per 100,000 children is the highest of any state (South Dakota is next at 2,779), while Vermont is again the lowest at 245 per 100,000, based on data collected from state health department websites.
Two COVID-19–related deaths in children were reported during the week ending Nov. 5, bringing the total to 123 but leaving the overall proportion of deaths in children unchanged at 0.06% of all deaths. Texas has reported the most COVID-19 deaths in children with 29, while 15 states have recorded no deaths so far (mortality data in children reported by 42 states and New York City), the AAP and CHA said.
The new weekly high for COVID-19 cases in children announced last week has been surpassed already, as the United States experienced almost 74,000 new pediatric cases for the week ending Nov. 5, according to the American Academy of Pediatrics and the Children’s Hospital Association.

The total number of COVID-19 cases in children is now 927,518 in 49 states, the District of Columbia, New York City, Puerto Rico, and Guam, the AAP and CHA said in their weekly report.
Cumulatively, children represent 11.3% of all COVID-19 cases in those jurisdictions, up from 11.1% a week ago. For just the past week, those 73,883 children represent 13.0% of the 567,672 new cases reported among all ages. That proportion peaked at 16.9% in mid-September, the AAP/CHA data show.
Dropping down to the state level, cumulative proportions as of Nov. 5 range from 5.2% in New Jersey to 23.3% in Wyoming, with 11 other states over 15%. California has had more cases, 100,856, than any other state, and Vermont the fewest at 329, the AAP and CHA said.
The national rate per 100,000 children is now 1,232, up from 1,134 the previous week and more than doubled since mid-August (582.2 per 100,000 on Aug. 20). North Dakota’s rate of 3,990 per 100,000 children is the highest of any state (South Dakota is next at 2,779), while Vermont is again the lowest at 245 per 100,000, based on data collected from state health department websites.
Two COVID-19–related deaths in children were reported during the week ending Nov. 5, bringing the total to 123 but leaving the overall proportion of deaths in children unchanged at 0.06% of all deaths. Texas has reported the most COVID-19 deaths in children with 29, while 15 states have recorded no deaths so far (mortality data in children reported by 42 states and New York City), the AAP and CHA said.
Food insecurity called urgent issue you must address
and advocate on behalf of those experiencing or at risk of food insecurity, according to Kofi Essel, MD, MPH, a pediatrician at Children’s National Hospital in Washington.
More than one in four adults are dealing with food access hardships during the pandemic, Dr. Essel said at the virtual annual meeting of the American Academy of Pediatrics. Food insecurity is often interchangeable with hunger and refers to limited or uncertain availability of foods that are nutritious and safe.
“Food insecurity is as much about the threat of deprivation as it is about deprivation itself: A food-insecure life means a life lived in fear of hunger, and the psychological toll that takes,” according to a 2020 New York Times photo feature on food insecurity by Brenda Ann Kenneally that Dr. Essel quoted.
The lived experience of food insecure households includes food anxiety, a preoccupation with being able to get enough food that takes up cognitive bandwidth and prevents people from being able to focus on other important things. Another feature of food-insecure homes is a monotony of diet, which often involves an increase in caloric density and decrease in nutritional quality. As food insecurity grows more dire, adults’ food intake decreases, and then children’s intake decreases as adults seek out any way to get food, including “socially unacceptable” ways, which can include food pantries and bartering for food.
Food insecurity is associated with a wide range of negative outcomes even after accounting for other confounders, including decreased overall health, mental health, and educational outcomes. It’s also associated with an increase in developmental delays, hospitalizations, iron deficiency, asthma, and birth defects, among other problems. Somewhat paradoxically, it’s associated with both an increase and a decrease in obesity in the research.
Megan J. Gray, MD, MPH, assistant professor of pediatrics and population health at Dell Medical School at the The University of Texas at Austin, attended Dr. Essel’s session because food insecurity during COVID-19 now affects about half her patients, according to screening research she’s conducted.
“I wanted to learn more about the nuances of screening and using language and talking points that are helpful with families and with staff in building a culture of discussing food insecurity in our clinics,” Dr. Gray said in an interview. “What I’ve learned in my clinic is that if we don’t ask about it, families aren’t telling us – food insecurity is hiding in plain sight.”
She particularly appreciated Dr. Essel’s slides on the progression of food insecurity and how they acknowledged the mental health burden of food insecurity among parents.
“Right now during COVID-19, I see more patients I would call ‘socially complex’ rather than ‘medically complex,’ ” she said. “We all need to get a crash course in social work and Dr. Essel’s presentation is a great starting place.”
Screening for food insecurity
Beginning in 2015, an AAP policy statement charged pediatricians to “screen and intervene” with regard to food insecurity and their patients, Dr. Essel said. The statement also called for pediatricians to advocate for programs and policies that end childhood food insecurity.
The policy statement recommended a validated two-question screening tool called the Hunger Vital Sign:
1. “Within the past 12 months, we worried whether our food would run out before we got money to buy more.”
2. “Within the past 12 months, the food that we bought just didn’t last and we didn’t have money to get more.”
But in screening, you need to be conscious of how dignity intersects with food insecurity concerns, Dr. Essel said.
“We need to create dignity for our families,” he said. “We need to create a safe environment for our families and use appropriate tools when necessary to be able to identify families that are struggling with food insecurity.”
That need is seen in research on food screening. The Hunger Vital Signs questions can be asked with a dichotomous variable, as a yes/no question, or on a Likert scale, though the latter is a more complex way to ask.
A 2017 study found, however, that asking with “yes/no” answers missed more than a quarter of at-risk families. In the AAP survey using “yes/no” answers, 31% of families screened positive for being at risk of food insecurity, compared with 46% when the same question was asked on a Likert scale. It seems the ability to answer with “sometimes” feels “safer” than answering “yes,” Dr. Essel said.
Another factor that potentially affects answers is how doctors ask. In a March 2020 study at a single primary care practice, 16% of families screened positive with yes/no responses to a food insecurity screen when the questions were written, compared with 10% of positive screens with verbal responses (P < .001).
Epidemiology of food insecurity
The most updated United States Department of Agriculture report on food insecurity released in September shows the United States finally reached prerecession levels in 2019, with 11% of families designated as “food insecure.” But 2019 data cannot show what has occurred since the pandemic.
Further, the numbers are higher in households with children: Fourteen percent, or one in seven households with children, are experiencing food insecurity. Racial and ethnic disparities in food insecurity have remained consistent over the past 2 decades, with about twice as many Black and Hispanic homes experiencing food insecurity as White homes.
More recent research using Census Household Pulse Surveys has found a tremendous increase in food insecurity for children in 2020. One in three Black children and one in four Hispanic children are food insecure, according to these surveys. The rates are one in six for Asian households and one in ten for White households.
“The disparity is consistent,” Dr. Essel said. “We see what COVID has done. We once may have described it as a great equalizer – everyone is touched in the same way – but the reality is, this is actually a great magnifier. It’s revealing to us and magnifying disparities that have existed for far too long and has really allowed us to see it in a new way.”
A big part of disparities in food insecurity is disparities in wealth, “the safety net or cushion for families when things go wrong,” Dr. Essel said. The median wealth of White Americans in 2016 was $171,000, compared to $20,700 among Latinx Americans and $17,600 among Black Americans, according to the Federal Reserve Board Survey of Consumer Finances.
Food insecurity interventions
Federal nutrition programs – such as Supplemental Nutrition Assistance Program (SNAP), the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), and school meal programs – are key to addressing food insecurity, Dr. Essel said.
“They have a long track record of rescuing families out of poverty, of rescuing families from food security and improving overall health of families,” he said.
But emergency food relief programs are important as well. Four in 10 families currently coming into food pantries are new recipients, and these resources have seen a 60% increase in clients, he said.
“This is utterly unreasonable for them to be able to manage,” he said. “Food pantries are essential but inadequate to compensate for large numbers of families,” even while they also may be the only option for families unable or unwilling to access federal programs. For example, for every one meal that food banks can provide, SNAP can provide nine meals, Dr. Essel said. Further, during times of economic downtown, every SNAP $1 spent generates $1.50 to $2 in economic activity.
Currently, the Pandemic Electronic Benefit Transfer (P-EBT) program provides benefits to families for school breakfast and lunch and has been extended through December 2021. Another federal pandemic response was to increase SNAP to the maximum household benefit for families, about $646 for a family of four, although 40% of households were already receiving the maximum benefit.
Food insecurity advocacy
You can advocate for any one of multiple pillars when it comes to food insecurity, Dr. Essel said. “Food cannot solve food insecurity by itself,” he said. “We have to think about root causes – systemic causes – and think about unemployment, livable wage, systemic racism, oppression, an inequitable food system. All of these things are pillars that any of you can advocate for when recognizing a family that is struggling with food insecurity.”
He offered several suggestions for advocacy:
- Join your local AAP chapter and prioritize food insecurity.
- Join a local antihunger task force.
- Make your clinical environment as safe as possible for families to respond to questions about food insecurity.
- Know what’s happening in your community immigrant populations.
- Provide up-to-date information to families about eligibility for federal programs.
- Share stories through op-eds and letters to the editor, and by contacting congressional representatives and providing expert testimony to school boards and city councils.
- Educate others about food insecurity through the above channels and on social media.
Jessica Lazerov, MD, a general pediatrician at Children’s National Anacostia and assistant professor of pediatrics at George Washington University, Washington, said the session was fantastic.
“Dr. Essel went beyond the basics of food insecurity, delving into the root causes, potential solutions, and important considerations when screening for food insecurity in practice,” Dr. Lazerov said in an interview. “I enjoyed his focus on advocacy, as well as the fact that he spent a bit of time reviewing how the COVID pandemic has affected food insecurity. I truly felt empowered to take my advocacy efforts a step further as Dr. Essel laid out concrete, actionable next steps, as well as a review of the most relevant and current information about food insecurity.”
Dr. Essel, Dr. Lazerov, and Dr. Gray have no relevant financial disclosures.
and advocate on behalf of those experiencing or at risk of food insecurity, according to Kofi Essel, MD, MPH, a pediatrician at Children’s National Hospital in Washington.
More than one in four adults are dealing with food access hardships during the pandemic, Dr. Essel said at the virtual annual meeting of the American Academy of Pediatrics. Food insecurity is often interchangeable with hunger and refers to limited or uncertain availability of foods that are nutritious and safe.
“Food insecurity is as much about the threat of deprivation as it is about deprivation itself: A food-insecure life means a life lived in fear of hunger, and the psychological toll that takes,” according to a 2020 New York Times photo feature on food insecurity by Brenda Ann Kenneally that Dr. Essel quoted.
The lived experience of food insecure households includes food anxiety, a preoccupation with being able to get enough food that takes up cognitive bandwidth and prevents people from being able to focus on other important things. Another feature of food-insecure homes is a monotony of diet, which often involves an increase in caloric density and decrease in nutritional quality. As food insecurity grows more dire, adults’ food intake decreases, and then children’s intake decreases as adults seek out any way to get food, including “socially unacceptable” ways, which can include food pantries and bartering for food.
Food insecurity is associated with a wide range of negative outcomes even after accounting for other confounders, including decreased overall health, mental health, and educational outcomes. It’s also associated with an increase in developmental delays, hospitalizations, iron deficiency, asthma, and birth defects, among other problems. Somewhat paradoxically, it’s associated with both an increase and a decrease in obesity in the research.
Megan J. Gray, MD, MPH, assistant professor of pediatrics and population health at Dell Medical School at the The University of Texas at Austin, attended Dr. Essel’s session because food insecurity during COVID-19 now affects about half her patients, according to screening research she’s conducted.
“I wanted to learn more about the nuances of screening and using language and talking points that are helpful with families and with staff in building a culture of discussing food insecurity in our clinics,” Dr. Gray said in an interview. “What I’ve learned in my clinic is that if we don’t ask about it, families aren’t telling us – food insecurity is hiding in plain sight.”
She particularly appreciated Dr. Essel’s slides on the progression of food insecurity and how they acknowledged the mental health burden of food insecurity among parents.
“Right now during COVID-19, I see more patients I would call ‘socially complex’ rather than ‘medically complex,’ ” she said. “We all need to get a crash course in social work and Dr. Essel’s presentation is a great starting place.”
Screening for food insecurity
Beginning in 2015, an AAP policy statement charged pediatricians to “screen and intervene” with regard to food insecurity and their patients, Dr. Essel said. The statement also called for pediatricians to advocate for programs and policies that end childhood food insecurity.
The policy statement recommended a validated two-question screening tool called the Hunger Vital Sign:
1. “Within the past 12 months, we worried whether our food would run out before we got money to buy more.”
2. “Within the past 12 months, the food that we bought just didn’t last and we didn’t have money to get more.”
But in screening, you need to be conscious of how dignity intersects with food insecurity concerns, Dr. Essel said.
“We need to create dignity for our families,” he said. “We need to create a safe environment for our families and use appropriate tools when necessary to be able to identify families that are struggling with food insecurity.”
That need is seen in research on food screening. The Hunger Vital Signs questions can be asked with a dichotomous variable, as a yes/no question, or on a Likert scale, though the latter is a more complex way to ask.
A 2017 study found, however, that asking with “yes/no” answers missed more than a quarter of at-risk families. In the AAP survey using “yes/no” answers, 31% of families screened positive for being at risk of food insecurity, compared with 46% when the same question was asked on a Likert scale. It seems the ability to answer with “sometimes” feels “safer” than answering “yes,” Dr. Essel said.
Another factor that potentially affects answers is how doctors ask. In a March 2020 study at a single primary care practice, 16% of families screened positive with yes/no responses to a food insecurity screen when the questions were written, compared with 10% of positive screens with verbal responses (P < .001).
Epidemiology of food insecurity
The most updated United States Department of Agriculture report on food insecurity released in September shows the United States finally reached prerecession levels in 2019, with 11% of families designated as “food insecure.” But 2019 data cannot show what has occurred since the pandemic.
Further, the numbers are higher in households with children: Fourteen percent, or one in seven households with children, are experiencing food insecurity. Racial and ethnic disparities in food insecurity have remained consistent over the past 2 decades, with about twice as many Black and Hispanic homes experiencing food insecurity as White homes.
More recent research using Census Household Pulse Surveys has found a tremendous increase in food insecurity for children in 2020. One in three Black children and one in four Hispanic children are food insecure, according to these surveys. The rates are one in six for Asian households and one in ten for White households.
“The disparity is consistent,” Dr. Essel said. “We see what COVID has done. We once may have described it as a great equalizer – everyone is touched in the same way – but the reality is, this is actually a great magnifier. It’s revealing to us and magnifying disparities that have existed for far too long and has really allowed us to see it in a new way.”
A big part of disparities in food insecurity is disparities in wealth, “the safety net or cushion for families when things go wrong,” Dr. Essel said. The median wealth of White Americans in 2016 was $171,000, compared to $20,700 among Latinx Americans and $17,600 among Black Americans, according to the Federal Reserve Board Survey of Consumer Finances.
Food insecurity interventions
Federal nutrition programs – such as Supplemental Nutrition Assistance Program (SNAP), the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), and school meal programs – are key to addressing food insecurity, Dr. Essel said.
“They have a long track record of rescuing families out of poverty, of rescuing families from food security and improving overall health of families,” he said.
But emergency food relief programs are important as well. Four in 10 families currently coming into food pantries are new recipients, and these resources have seen a 60% increase in clients, he said.
“This is utterly unreasonable for them to be able to manage,” he said. “Food pantries are essential but inadequate to compensate for large numbers of families,” even while they also may be the only option for families unable or unwilling to access federal programs. For example, for every one meal that food banks can provide, SNAP can provide nine meals, Dr. Essel said. Further, during times of economic downtown, every SNAP $1 spent generates $1.50 to $2 in economic activity.
Currently, the Pandemic Electronic Benefit Transfer (P-EBT) program provides benefits to families for school breakfast and lunch and has been extended through December 2021. Another federal pandemic response was to increase SNAP to the maximum household benefit for families, about $646 for a family of four, although 40% of households were already receiving the maximum benefit.
Food insecurity advocacy
You can advocate for any one of multiple pillars when it comes to food insecurity, Dr. Essel said. “Food cannot solve food insecurity by itself,” he said. “We have to think about root causes – systemic causes – and think about unemployment, livable wage, systemic racism, oppression, an inequitable food system. All of these things are pillars that any of you can advocate for when recognizing a family that is struggling with food insecurity.”
He offered several suggestions for advocacy:
- Join your local AAP chapter and prioritize food insecurity.
- Join a local antihunger task force.
- Make your clinical environment as safe as possible for families to respond to questions about food insecurity.
- Know what’s happening in your community immigrant populations.
- Provide up-to-date information to families about eligibility for federal programs.
- Share stories through op-eds and letters to the editor, and by contacting congressional representatives and providing expert testimony to school boards and city councils.
- Educate others about food insecurity through the above channels and on social media.
Jessica Lazerov, MD, a general pediatrician at Children’s National Anacostia and assistant professor of pediatrics at George Washington University, Washington, said the session was fantastic.
“Dr. Essel went beyond the basics of food insecurity, delving into the root causes, potential solutions, and important considerations when screening for food insecurity in practice,” Dr. Lazerov said in an interview. “I enjoyed his focus on advocacy, as well as the fact that he spent a bit of time reviewing how the COVID pandemic has affected food insecurity. I truly felt empowered to take my advocacy efforts a step further as Dr. Essel laid out concrete, actionable next steps, as well as a review of the most relevant and current information about food insecurity.”
Dr. Essel, Dr. Lazerov, and Dr. Gray have no relevant financial disclosures.
and advocate on behalf of those experiencing or at risk of food insecurity, according to Kofi Essel, MD, MPH, a pediatrician at Children’s National Hospital in Washington.
More than one in four adults are dealing with food access hardships during the pandemic, Dr. Essel said at the virtual annual meeting of the American Academy of Pediatrics. Food insecurity is often interchangeable with hunger and refers to limited or uncertain availability of foods that are nutritious and safe.
“Food insecurity is as much about the threat of deprivation as it is about deprivation itself: A food-insecure life means a life lived in fear of hunger, and the psychological toll that takes,” according to a 2020 New York Times photo feature on food insecurity by Brenda Ann Kenneally that Dr. Essel quoted.
The lived experience of food insecure households includes food anxiety, a preoccupation with being able to get enough food that takes up cognitive bandwidth and prevents people from being able to focus on other important things. Another feature of food-insecure homes is a monotony of diet, which often involves an increase in caloric density and decrease in nutritional quality. As food insecurity grows more dire, adults’ food intake decreases, and then children’s intake decreases as adults seek out any way to get food, including “socially unacceptable” ways, which can include food pantries and bartering for food.
Food insecurity is associated with a wide range of negative outcomes even after accounting for other confounders, including decreased overall health, mental health, and educational outcomes. It’s also associated with an increase in developmental delays, hospitalizations, iron deficiency, asthma, and birth defects, among other problems. Somewhat paradoxically, it’s associated with both an increase and a decrease in obesity in the research.
Megan J. Gray, MD, MPH, assistant professor of pediatrics and population health at Dell Medical School at the The University of Texas at Austin, attended Dr. Essel’s session because food insecurity during COVID-19 now affects about half her patients, according to screening research she’s conducted.
“I wanted to learn more about the nuances of screening and using language and talking points that are helpful with families and with staff in building a culture of discussing food insecurity in our clinics,” Dr. Gray said in an interview. “What I’ve learned in my clinic is that if we don’t ask about it, families aren’t telling us – food insecurity is hiding in plain sight.”
She particularly appreciated Dr. Essel’s slides on the progression of food insecurity and how they acknowledged the mental health burden of food insecurity among parents.
“Right now during COVID-19, I see more patients I would call ‘socially complex’ rather than ‘medically complex,’ ” she said. “We all need to get a crash course in social work and Dr. Essel’s presentation is a great starting place.”
Screening for food insecurity
Beginning in 2015, an AAP policy statement charged pediatricians to “screen and intervene” with regard to food insecurity and their patients, Dr. Essel said. The statement also called for pediatricians to advocate for programs and policies that end childhood food insecurity.
The policy statement recommended a validated two-question screening tool called the Hunger Vital Sign:
1. “Within the past 12 months, we worried whether our food would run out before we got money to buy more.”
2. “Within the past 12 months, the food that we bought just didn’t last and we didn’t have money to get more.”
But in screening, you need to be conscious of how dignity intersects with food insecurity concerns, Dr. Essel said.
“We need to create dignity for our families,” he said. “We need to create a safe environment for our families and use appropriate tools when necessary to be able to identify families that are struggling with food insecurity.”
That need is seen in research on food screening. The Hunger Vital Signs questions can be asked with a dichotomous variable, as a yes/no question, or on a Likert scale, though the latter is a more complex way to ask.
A 2017 study found, however, that asking with “yes/no” answers missed more than a quarter of at-risk families. In the AAP survey using “yes/no” answers, 31% of families screened positive for being at risk of food insecurity, compared with 46% when the same question was asked on a Likert scale. It seems the ability to answer with “sometimes” feels “safer” than answering “yes,” Dr. Essel said.
Another factor that potentially affects answers is how doctors ask. In a March 2020 study at a single primary care practice, 16% of families screened positive with yes/no responses to a food insecurity screen when the questions were written, compared with 10% of positive screens with verbal responses (P < .001).
Epidemiology of food insecurity
The most updated United States Department of Agriculture report on food insecurity released in September shows the United States finally reached prerecession levels in 2019, with 11% of families designated as “food insecure.” But 2019 data cannot show what has occurred since the pandemic.
Further, the numbers are higher in households with children: Fourteen percent, or one in seven households with children, are experiencing food insecurity. Racial and ethnic disparities in food insecurity have remained consistent over the past 2 decades, with about twice as many Black and Hispanic homes experiencing food insecurity as White homes.
More recent research using Census Household Pulse Surveys has found a tremendous increase in food insecurity for children in 2020. One in three Black children and one in four Hispanic children are food insecure, according to these surveys. The rates are one in six for Asian households and one in ten for White households.
“The disparity is consistent,” Dr. Essel said. “We see what COVID has done. We once may have described it as a great equalizer – everyone is touched in the same way – but the reality is, this is actually a great magnifier. It’s revealing to us and magnifying disparities that have existed for far too long and has really allowed us to see it in a new way.”
A big part of disparities in food insecurity is disparities in wealth, “the safety net or cushion for families when things go wrong,” Dr. Essel said. The median wealth of White Americans in 2016 was $171,000, compared to $20,700 among Latinx Americans and $17,600 among Black Americans, according to the Federal Reserve Board Survey of Consumer Finances.
Food insecurity interventions
Federal nutrition programs – such as Supplemental Nutrition Assistance Program (SNAP), the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC), and school meal programs – are key to addressing food insecurity, Dr. Essel said.
“They have a long track record of rescuing families out of poverty, of rescuing families from food security and improving overall health of families,” he said.
But emergency food relief programs are important as well. Four in 10 families currently coming into food pantries are new recipients, and these resources have seen a 60% increase in clients, he said.
“This is utterly unreasonable for them to be able to manage,” he said. “Food pantries are essential but inadequate to compensate for large numbers of families,” even while they also may be the only option for families unable or unwilling to access federal programs. For example, for every one meal that food banks can provide, SNAP can provide nine meals, Dr. Essel said. Further, during times of economic downtown, every SNAP $1 spent generates $1.50 to $2 in economic activity.
Currently, the Pandemic Electronic Benefit Transfer (P-EBT) program provides benefits to families for school breakfast and lunch and has been extended through December 2021. Another federal pandemic response was to increase SNAP to the maximum household benefit for families, about $646 for a family of four, although 40% of households were already receiving the maximum benefit.
Food insecurity advocacy
You can advocate for any one of multiple pillars when it comes to food insecurity, Dr. Essel said. “Food cannot solve food insecurity by itself,” he said. “We have to think about root causes – systemic causes – and think about unemployment, livable wage, systemic racism, oppression, an inequitable food system. All of these things are pillars that any of you can advocate for when recognizing a family that is struggling with food insecurity.”
He offered several suggestions for advocacy:
- Join your local AAP chapter and prioritize food insecurity.
- Join a local antihunger task force.
- Make your clinical environment as safe as possible for families to respond to questions about food insecurity.
- Know what’s happening in your community immigrant populations.
- Provide up-to-date information to families about eligibility for federal programs.
- Share stories through op-eds and letters to the editor, and by contacting congressional representatives and providing expert testimony to school boards and city councils.
- Educate others about food insecurity through the above channels and on social media.
Jessica Lazerov, MD, a general pediatrician at Children’s National Anacostia and assistant professor of pediatrics at George Washington University, Washington, said the session was fantastic.
“Dr. Essel went beyond the basics of food insecurity, delving into the root causes, potential solutions, and important considerations when screening for food insecurity in practice,” Dr. Lazerov said in an interview. “I enjoyed his focus on advocacy, as well as the fact that he spent a bit of time reviewing how the COVID pandemic has affected food insecurity. I truly felt empowered to take my advocacy efforts a step further as Dr. Essel laid out concrete, actionable next steps, as well as a review of the most relevant and current information about food insecurity.”
Dr. Essel, Dr. Lazerov, and Dr. Gray have no relevant financial disclosures.
EXPERT ANALYSIS FROM AAP 2020
Pfizer vaccine data show 90% efficacy in early results
A vaccine candidate against SARS-CoV-2 has been found to be 90% effective in preventing COVID-19 in trial volunteers who were without evidence of prior infection of the virus, results from an interim analysis of a phase 3 study demonstrated.
BTN162b2, a messenger RNA–based vaccine candidate that requires two doses, is being developed by Pfizer and BioNTech SE independently of the Trump administration’s Operation Warp Speed. A global phase 3 clinical trial of BTN162b2 began on July 27 and has enrolled 43,538 participants to date; 42% of enrollees have racially and ethnically diverse backgrounds.
According to a press release issued by the two companies, 38,955 trial volunteers had received a second dose of either vaccine or placebo as of Nov. 8. An interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine efficacy rate was above 90% 7 days after the second dose. This means that protection was achieved 28 days after the first vaccine dose.
“It’s promising in that it validates the genetic strategy – whether it’s mRNA vaccines or DNA vaccines,” Paul A. Offit, MD, told Medscape Medical News. Offit is a member of the US Food and Drug Administraiton’s COVID-19 Vaccine Advisory Committee. “All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it’s effective. That’s good. We knew that it seemed to work in experimental animals, but you never know until you put it into people.”
According to Pfizer and BioNTech SE, a final data analysis is planned once 164 confirmed COVID-19 cases have accrued. So far, the DMC has not reported any serious safety concerns. It recommends that the study continue to collect safety and efficacy data as planned. The companies plan to apply to the FDA for emergency use authorization soon after the required safety milestone is achieved.
Pfizer CEO Albert Bourla, DVM, PhD, added in a separate press release, “It’s important to note that we cannot apply for FDA Emergency Use Authorization based on these efficacy results alone. More data on safety is also needed, and we are continuing to accumulate that safety data as part of our ongoing clinical study.
“We estimate that a median of two months of safety data following the second and final dose of the vaccine candidate – required by FDA’s guidance for potential Emergency Use Authorization – will be available by the third week of November.”
Offit, professor of pediatrics in the Division of Infectious Diseases at the Children’s Hospital of Philadelphia, said that, if BTN162b2 is approved, administering it will be tricky. “This particular vaccine has to be shipped and stored at –70° C or –80° C, which we’ve never done before in this country,” he said. “That means maintaining the product on dry ice. That’s going to be a challenge for distribution, I think.”
Good news, but…
In the press release, BioNTech SE’s cofounder and CEO, Ugur Sahin, MD, characterized the findings as “a victory for innovation, science and a global collaborative effort. When we embarked on this journey 10 months ago this is what we aspired to achieve. Especially today, while we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality.”
President-elect Joe Biden also weighed in, calling the results “excellent news” in a news release.
“At the same time, it is also important to understand that the end of the battle against COVID-19 is still months away,” he said. “This news follows a previously announced timeline by industry officials that forecast vaccine approval by late November. Even if that is achieved, and some Americans are vaccinated later this year, it will be many more months before there is widespread vaccination in this country.
“Today’s news does not change this urgent reality. Americans will have to rely on masking, distancing, contact tracing, hand washing, and other measures to keep themselves safe well into next year,” Biden added.
This article first appeared on Medscape.com.
A vaccine candidate against SARS-CoV-2 has been found to be 90% effective in preventing COVID-19 in trial volunteers who were without evidence of prior infection of the virus, results from an interim analysis of a phase 3 study demonstrated.
BTN162b2, a messenger RNA–based vaccine candidate that requires two doses, is being developed by Pfizer and BioNTech SE independently of the Trump administration’s Operation Warp Speed. A global phase 3 clinical trial of BTN162b2 began on July 27 and has enrolled 43,538 participants to date; 42% of enrollees have racially and ethnically diverse backgrounds.
According to a press release issued by the two companies, 38,955 trial volunteers had received a second dose of either vaccine or placebo as of Nov. 8. An interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine efficacy rate was above 90% 7 days after the second dose. This means that protection was achieved 28 days after the first vaccine dose.
“It’s promising in that it validates the genetic strategy – whether it’s mRNA vaccines or DNA vaccines,” Paul A. Offit, MD, told Medscape Medical News. Offit is a member of the US Food and Drug Administraiton’s COVID-19 Vaccine Advisory Committee. “All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it’s effective. That’s good. We knew that it seemed to work in experimental animals, but you never know until you put it into people.”
According to Pfizer and BioNTech SE, a final data analysis is planned once 164 confirmed COVID-19 cases have accrued. So far, the DMC has not reported any serious safety concerns. It recommends that the study continue to collect safety and efficacy data as planned. The companies plan to apply to the FDA for emergency use authorization soon after the required safety milestone is achieved.
Pfizer CEO Albert Bourla, DVM, PhD, added in a separate press release, “It’s important to note that we cannot apply for FDA Emergency Use Authorization based on these efficacy results alone. More data on safety is also needed, and we are continuing to accumulate that safety data as part of our ongoing clinical study.
“We estimate that a median of two months of safety data following the second and final dose of the vaccine candidate – required by FDA’s guidance for potential Emergency Use Authorization – will be available by the third week of November.”
Offit, professor of pediatrics in the Division of Infectious Diseases at the Children’s Hospital of Philadelphia, said that, if BTN162b2 is approved, administering it will be tricky. “This particular vaccine has to be shipped and stored at –70° C or –80° C, which we’ve never done before in this country,” he said. “That means maintaining the product on dry ice. That’s going to be a challenge for distribution, I think.”
Good news, but…
In the press release, BioNTech SE’s cofounder and CEO, Ugur Sahin, MD, characterized the findings as “a victory for innovation, science and a global collaborative effort. When we embarked on this journey 10 months ago this is what we aspired to achieve. Especially today, while we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality.”
President-elect Joe Biden also weighed in, calling the results “excellent news” in a news release.
“At the same time, it is also important to understand that the end of the battle against COVID-19 is still months away,” he said. “This news follows a previously announced timeline by industry officials that forecast vaccine approval by late November. Even if that is achieved, and some Americans are vaccinated later this year, it will be many more months before there is widespread vaccination in this country.
“Today’s news does not change this urgent reality. Americans will have to rely on masking, distancing, contact tracing, hand washing, and other measures to keep themselves safe well into next year,” Biden added.
This article first appeared on Medscape.com.
A vaccine candidate against SARS-CoV-2 has been found to be 90% effective in preventing COVID-19 in trial volunteers who were without evidence of prior infection of the virus, results from an interim analysis of a phase 3 study demonstrated.
BTN162b2, a messenger RNA–based vaccine candidate that requires two doses, is being developed by Pfizer and BioNTech SE independently of the Trump administration’s Operation Warp Speed. A global phase 3 clinical trial of BTN162b2 began on July 27 and has enrolled 43,538 participants to date; 42% of enrollees have racially and ethnically diverse backgrounds.
According to a press release issued by the two companies, 38,955 trial volunteers had received a second dose of either vaccine or placebo as of Nov. 8. An interim analysis of 94 individuals conducted by an independent data monitoring committee (DMC) found that the vaccine efficacy rate was above 90% 7 days after the second dose. This means that protection was achieved 28 days after the first vaccine dose.
“It’s promising in that it validates the genetic strategy – whether it’s mRNA vaccines or DNA vaccines,” Paul A. Offit, MD, told Medscape Medical News. Offit is a member of the US Food and Drug Administraiton’s COVID-19 Vaccine Advisory Committee. “All of them have the same approach, which is that they introduce the gene that codes for the coronavirus spike protein into the cell. Your cell makes the spike protein, and your immune system makes antibodies to the spike protein. At least in these preliminary data, which involved 94 people getting sick, it looks like it’s effective. That’s good. We knew that it seemed to work in experimental animals, but you never know until you put it into people.”
According to Pfizer and BioNTech SE, a final data analysis is planned once 164 confirmed COVID-19 cases have accrued. So far, the DMC has not reported any serious safety concerns. It recommends that the study continue to collect safety and efficacy data as planned. The companies plan to apply to the FDA for emergency use authorization soon after the required safety milestone is achieved.
Pfizer CEO Albert Bourla, DVM, PhD, added in a separate press release, “It’s important to note that we cannot apply for FDA Emergency Use Authorization based on these efficacy results alone. More data on safety is also needed, and we are continuing to accumulate that safety data as part of our ongoing clinical study.
“We estimate that a median of two months of safety data following the second and final dose of the vaccine candidate – required by FDA’s guidance for potential Emergency Use Authorization – will be available by the third week of November.”
Offit, professor of pediatrics in the Division of Infectious Diseases at the Children’s Hospital of Philadelphia, said that, if BTN162b2 is approved, administering it will be tricky. “This particular vaccine has to be shipped and stored at –70° C or –80° C, which we’ve never done before in this country,” he said. “That means maintaining the product on dry ice. That’s going to be a challenge for distribution, I think.”
Good news, but…
In the press release, BioNTech SE’s cofounder and CEO, Ugur Sahin, MD, characterized the findings as “a victory for innovation, science and a global collaborative effort. When we embarked on this journey 10 months ago this is what we aspired to achieve. Especially today, while we are all in the midst of a second wave and many of us in lockdown, we appreciate even more how important this milestone is on our path towards ending this pandemic and for all of us to regain a sense of normality.”
President-elect Joe Biden also weighed in, calling the results “excellent news” in a news release.
“At the same time, it is also important to understand that the end of the battle against COVID-19 is still months away,” he said. “This news follows a previously announced timeline by industry officials that forecast vaccine approval by late November. Even if that is achieved, and some Americans are vaccinated later this year, it will be many more months before there is widespread vaccination in this country.
“Today’s news does not change this urgent reality. Americans will have to rely on masking, distancing, contact tracing, hand washing, and other measures to keep themselves safe well into next year,” Biden added.
This article first appeared on Medscape.com.
What to know as ACA heads to Supreme Court – again
The case, California v. Texas, is the result of a change to the health law made by Congress in 2017. As part of a major tax bill, Congress reduced to zero the penalty for not having health insurance. But it was that penalty – a tax – that the high court ruled made the law constitutional in a 2012 decision, argues a group of Republican state attorneys general. Without the tax, they say in their suit, the rest of the law must fall, too.
After originally contending that the entire law should not be struck down when the suit was filed in 2018, the Trump administration changed course in 2019 and joined the GOP officials who brought the case.
Here are some key questions and answers about the case.
What are the possibilities for how the court could rule?
There is a long list of ways this could play out.
The justices could declare the entire law unconstitutional – which is what a federal district judge in Texas ruled in December 2018. But legal experts say that’s not the most likely outcome of this case.
First, the court may avoid deciding the case on its merits entirely by ruling that the plaintiffs do not have “standing” to sue. The central issue in the case is whether the requirement in the law to have insurance – which remains even though Congress eliminated the penalty or tax – is constitutional. But states are not subject to the so-called individual mandate, so some analysts suggest the Republican officials have no standing. In addition, questions have been raised about the individual plaintiffs in the case, two consultants from Texas who argue that they felt compelled to buy insurance even without a possible penalty.
The court could also rule that, by eliminating the penalty but not the rest of the mandate (which Congress could not do in that 2017 tax bill for procedural reasons), lawmakers “didn’t mean to coerce anyone to do anything, and so there’s no constitutional problem,” University of Michigan law professor Nicholas Bagley said in a recent webinar for the NIHCM Foundation, the Commonwealth Fund, and the University of Southern California’s Center for Health Journalism.
Or, said Bagley, the court could rule that, without the tax, the requirement to have health insurance is unconstitutional, but the rest of the law is not. In that case, the justices might strike the mandate only, which would have basically no impact.
It gets more complicated if the court decides that, as the plaintiffs argue, the individual mandate language without the penalty is unconstitutional and so closely tied to other parts of the law that some of them must fall as well.
Even there the court has choices. One option would be, as the Trump administration originally argued, to strike down the mandate and just the pieces of the law most closely related to it – which happen to include the insurance protections for people with preexisting conditions, an extremely popular provision of the law. The two parts are connected because the original purpose of the mandate was to make sure enough healthy people sign up for insurance to offset the added costs to insurers of sicker people.
Another option, of course, would be for the court to follow the lead of the Texas judge and strike down the entire law.
While that’s not the most likely outcome, said Bagley, if it happens it could be “a hot mess” for the nation’s entire health care system. As just one example, he said, “every hospital is getting paid pursuant to changes made by the ACA. How do you even go about making payments if the thing that you are looking to guide what those payments ought to be is itself invalid?”
What impact will new Justice Amy Coney Barrett have?
Perhaps a lot. Before the death of Justice Ruth Bader Ginsburg, most court observers thought the case was highly unlikely to result in the entire law being struck down. That’s because Chief Justice John Roberts voted to uphold the law in 2012, and again when it was challenged in a less sweeping way in 2015.
But with Barrett replacing Ginsburg, even if Roberts joined the court’s remaining three liberals they could still be outvoted by the other five conservatives. Barrett was coy about her views on the Affordable Care Act during her confirmation hearings in October, but she has written that she thinks Roberts was wrong to uphold the law in 2012.
Could a new president and Congress make the case go away?
Many have suggested that, if Joe Biden assumes the presidency, his Justice Department could simply drop the case. But the administration did not bring the case; the GOP state officials did. And while normally the Justice Department’s job is to defend existing laws in court, in this case the ACA is being defended by a group of Democratic state attorneys general. A new administration could change that position, but that’s not the same as dropping the case.
Congress, on the other hand, could easily make the case moot. It could add back even a nominal financial penalty for not having insurance. It could eliminate the mandate altogether, although that would require 60 votes in the Senate under current rules. Congress could also pass a “severability” provision saying that, if any portion of the law is struck down, the rest should remain.
“The problem is not technical,” said Bagley. “It’s political.”
What is the timeline for a decision? Could the court delay implementation of its ruling?
The court usually hears oral arguments in a case months before it issues a decision. Unless the decision is unanimous or turns out to be very simple, Bagley said, he would expect to see an opinion “sometime in the spring.”
As to whether the court could find some or all of the law unconstitutional but delay when its decision takes effect, Bagley said that happened from time to time as recently as the 1970s. “That practice has been more or less abandoned,” he said, but in the case of a law so large, “you could imagine the Supreme Court using its discretion to say the decision wouldn’t take effect immediately.”
If the court does invalidate the entire ACA, Congress could act to fix things, but it’s unclear if it will be able to, especially if Republicans still control the Senate. If the justices strike the law, Bagley said, “I honestly think the likeliest outcome is that Congress runs around like a chicken with its head cut off, doesn’t come to a deal, and we’re back to where we were before 2010” when the ACA passed.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
The case, California v. Texas, is the result of a change to the health law made by Congress in 2017. As part of a major tax bill, Congress reduced to zero the penalty for not having health insurance. But it was that penalty – a tax – that the high court ruled made the law constitutional in a 2012 decision, argues a group of Republican state attorneys general. Without the tax, they say in their suit, the rest of the law must fall, too.
After originally contending that the entire law should not be struck down when the suit was filed in 2018, the Trump administration changed course in 2019 and joined the GOP officials who brought the case.
Here are some key questions and answers about the case.
What are the possibilities for how the court could rule?
There is a long list of ways this could play out.
The justices could declare the entire law unconstitutional – which is what a federal district judge in Texas ruled in December 2018. But legal experts say that’s not the most likely outcome of this case.
First, the court may avoid deciding the case on its merits entirely by ruling that the plaintiffs do not have “standing” to sue. The central issue in the case is whether the requirement in the law to have insurance – which remains even though Congress eliminated the penalty or tax – is constitutional. But states are not subject to the so-called individual mandate, so some analysts suggest the Republican officials have no standing. In addition, questions have been raised about the individual plaintiffs in the case, two consultants from Texas who argue that they felt compelled to buy insurance even without a possible penalty.
The court could also rule that, by eliminating the penalty but not the rest of the mandate (which Congress could not do in that 2017 tax bill for procedural reasons), lawmakers “didn’t mean to coerce anyone to do anything, and so there’s no constitutional problem,” University of Michigan law professor Nicholas Bagley said in a recent webinar for the NIHCM Foundation, the Commonwealth Fund, and the University of Southern California’s Center for Health Journalism.
Or, said Bagley, the court could rule that, without the tax, the requirement to have health insurance is unconstitutional, but the rest of the law is not. In that case, the justices might strike the mandate only, which would have basically no impact.
It gets more complicated if the court decides that, as the plaintiffs argue, the individual mandate language without the penalty is unconstitutional and so closely tied to other parts of the law that some of them must fall as well.
Even there the court has choices. One option would be, as the Trump administration originally argued, to strike down the mandate and just the pieces of the law most closely related to it – which happen to include the insurance protections for people with preexisting conditions, an extremely popular provision of the law. The two parts are connected because the original purpose of the mandate was to make sure enough healthy people sign up for insurance to offset the added costs to insurers of sicker people.
Another option, of course, would be for the court to follow the lead of the Texas judge and strike down the entire law.
While that’s not the most likely outcome, said Bagley, if it happens it could be “a hot mess” for the nation’s entire health care system. As just one example, he said, “every hospital is getting paid pursuant to changes made by the ACA. How do you even go about making payments if the thing that you are looking to guide what those payments ought to be is itself invalid?”
What impact will new Justice Amy Coney Barrett have?
Perhaps a lot. Before the death of Justice Ruth Bader Ginsburg, most court observers thought the case was highly unlikely to result in the entire law being struck down. That’s because Chief Justice John Roberts voted to uphold the law in 2012, and again when it was challenged in a less sweeping way in 2015.
But with Barrett replacing Ginsburg, even if Roberts joined the court’s remaining three liberals they could still be outvoted by the other five conservatives. Barrett was coy about her views on the Affordable Care Act during her confirmation hearings in October, but she has written that she thinks Roberts was wrong to uphold the law in 2012.
Could a new president and Congress make the case go away?
Many have suggested that, if Joe Biden assumes the presidency, his Justice Department could simply drop the case. But the administration did not bring the case; the GOP state officials did. And while normally the Justice Department’s job is to defend existing laws in court, in this case the ACA is being defended by a group of Democratic state attorneys general. A new administration could change that position, but that’s not the same as dropping the case.
Congress, on the other hand, could easily make the case moot. It could add back even a nominal financial penalty for not having insurance. It could eliminate the mandate altogether, although that would require 60 votes in the Senate under current rules. Congress could also pass a “severability” provision saying that, if any portion of the law is struck down, the rest should remain.
“The problem is not technical,” said Bagley. “It’s political.”
What is the timeline for a decision? Could the court delay implementation of its ruling?
The court usually hears oral arguments in a case months before it issues a decision. Unless the decision is unanimous or turns out to be very simple, Bagley said, he would expect to see an opinion “sometime in the spring.”
As to whether the court could find some or all of the law unconstitutional but delay when its decision takes effect, Bagley said that happened from time to time as recently as the 1970s. “That practice has been more or less abandoned,” he said, but in the case of a law so large, “you could imagine the Supreme Court using its discretion to say the decision wouldn’t take effect immediately.”
If the court does invalidate the entire ACA, Congress could act to fix things, but it’s unclear if it will be able to, especially if Republicans still control the Senate. If the justices strike the law, Bagley said, “I honestly think the likeliest outcome is that Congress runs around like a chicken with its head cut off, doesn’t come to a deal, and we’re back to where we were before 2010” when the ACA passed.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
The case, California v. Texas, is the result of a change to the health law made by Congress in 2017. As part of a major tax bill, Congress reduced to zero the penalty for not having health insurance. But it was that penalty – a tax – that the high court ruled made the law constitutional in a 2012 decision, argues a group of Republican state attorneys general. Without the tax, they say in their suit, the rest of the law must fall, too.
After originally contending that the entire law should not be struck down when the suit was filed in 2018, the Trump administration changed course in 2019 and joined the GOP officials who brought the case.
Here are some key questions and answers about the case.
What are the possibilities for how the court could rule?
There is a long list of ways this could play out.
The justices could declare the entire law unconstitutional – which is what a federal district judge in Texas ruled in December 2018. But legal experts say that’s not the most likely outcome of this case.
First, the court may avoid deciding the case on its merits entirely by ruling that the plaintiffs do not have “standing” to sue. The central issue in the case is whether the requirement in the law to have insurance – which remains even though Congress eliminated the penalty or tax – is constitutional. But states are not subject to the so-called individual mandate, so some analysts suggest the Republican officials have no standing. In addition, questions have been raised about the individual plaintiffs in the case, two consultants from Texas who argue that they felt compelled to buy insurance even without a possible penalty.
The court could also rule that, by eliminating the penalty but not the rest of the mandate (which Congress could not do in that 2017 tax bill for procedural reasons), lawmakers “didn’t mean to coerce anyone to do anything, and so there’s no constitutional problem,” University of Michigan law professor Nicholas Bagley said in a recent webinar for the NIHCM Foundation, the Commonwealth Fund, and the University of Southern California’s Center for Health Journalism.
Or, said Bagley, the court could rule that, without the tax, the requirement to have health insurance is unconstitutional, but the rest of the law is not. In that case, the justices might strike the mandate only, which would have basically no impact.
It gets more complicated if the court decides that, as the plaintiffs argue, the individual mandate language without the penalty is unconstitutional and so closely tied to other parts of the law that some of them must fall as well.
Even there the court has choices. One option would be, as the Trump administration originally argued, to strike down the mandate and just the pieces of the law most closely related to it – which happen to include the insurance protections for people with preexisting conditions, an extremely popular provision of the law. The two parts are connected because the original purpose of the mandate was to make sure enough healthy people sign up for insurance to offset the added costs to insurers of sicker people.
Another option, of course, would be for the court to follow the lead of the Texas judge and strike down the entire law.
While that’s not the most likely outcome, said Bagley, if it happens it could be “a hot mess” for the nation’s entire health care system. As just one example, he said, “every hospital is getting paid pursuant to changes made by the ACA. How do you even go about making payments if the thing that you are looking to guide what those payments ought to be is itself invalid?”
What impact will new Justice Amy Coney Barrett have?
Perhaps a lot. Before the death of Justice Ruth Bader Ginsburg, most court observers thought the case was highly unlikely to result in the entire law being struck down. That’s because Chief Justice John Roberts voted to uphold the law in 2012, and again when it was challenged in a less sweeping way in 2015.
But with Barrett replacing Ginsburg, even if Roberts joined the court’s remaining three liberals they could still be outvoted by the other five conservatives. Barrett was coy about her views on the Affordable Care Act during her confirmation hearings in October, but she has written that she thinks Roberts was wrong to uphold the law in 2012.
Could a new president and Congress make the case go away?
Many have suggested that, if Joe Biden assumes the presidency, his Justice Department could simply drop the case. But the administration did not bring the case; the GOP state officials did. And while normally the Justice Department’s job is to defend existing laws in court, in this case the ACA is being defended by a group of Democratic state attorneys general. A new administration could change that position, but that’s not the same as dropping the case.
Congress, on the other hand, could easily make the case moot. It could add back even a nominal financial penalty for not having insurance. It could eliminate the mandate altogether, although that would require 60 votes in the Senate under current rules. Congress could also pass a “severability” provision saying that, if any portion of the law is struck down, the rest should remain.
“The problem is not technical,” said Bagley. “It’s political.”
What is the timeline for a decision? Could the court delay implementation of its ruling?
The court usually hears oral arguments in a case months before it issues a decision. Unless the decision is unanimous or turns out to be very simple, Bagley said, he would expect to see an opinion “sometime in the spring.”
As to whether the court could find some or all of the law unconstitutional but delay when its decision takes effect, Bagley said that happened from time to time as recently as the 1970s. “That practice has been more or less abandoned,” he said, but in the case of a law so large, “you could imagine the Supreme Court using its discretion to say the decision wouldn’t take effect immediately.”
If the court does invalidate the entire ACA, Congress could act to fix things, but it’s unclear if it will be able to, especially if Republicans still control the Senate. If the justices strike the law, Bagley said, “I honestly think the likeliest outcome is that Congress runs around like a chicken with its head cut off, doesn’t come to a deal, and we’re back to where we were before 2010” when the ACA passed.
Kaiser Health News is a nonprofit news service covering health issues. It is an editorially independent program of KFF (Kaiser Family Foundation), which is not affiliated with Kaiser Permanente.
‘Disordered eating’ drops after teens undergo bariatric surgery
Kristina M. Decker, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital Medical Center, presented these findings during the virtual ObesityWeek 2020.
Dr. Decker and associates examined rates of disordered eating in more than 200 adolescents (aged 13-18 years) who were severely obese, of whom 141 underwent bariatric surgery and the remainder did not.
At baseline (presurgery), the teens in both groups had rates of disordered eating ranging from 11% to 50%, with higher rates in those who went on to have bariatric surgery.
Six years later, rates of disordered eating were much lower in those who had bariatric surgery.
The data nevertheless “underscore that young adults with persistent severe obesity are at high risk for poor health and well-being,” Dr. Decker said in an interview.
“This means disordered eating behaviors should be closely monitored” in all such patients, both those who undergo surgery and those who don’t, she stressed.
Robust findings because of long follow-up and controls
The findings are not unexpected, based on adult bariatric literature, but are “novel because of the age of the patients,” senior author Margaret H. Zeller, PhD, Cincinnati Children’s Hospital Medical Center and professor at the University of Cincinnati, added.
In a comment comment, psychologist Kajsa Järvholm, PhD, of the Childhood Obesity Unit at Skåne University Hospital, Malmö̈, Sweden, who has published related work, said that this is “a needed study.”
Notably, it had “long-term follow-up and a control group,” and it “confirms that adolescents are in better control of their eating after surgery.”
However, an important additional takeaway for clinicians is that “disordered eating is associated with other mental health problems and self-worth. Clinicians treating obesity must address problems related to eating disorders to improve outcomes and well-being,” she stressed.
How does bariatric surgery impact overeating, binge eating, in teens?
“For teens with severe obesity, metabolic and bariatric surgery is the most effective treatment for improved cardiometabolic functioning, weight loss, and improved quality of life,” Dr. Decker stressed.
However, pre- and postsurgical disordered eating behaviors have been associated with a lower percentage change in body mass index (BMI), although this has not been well studied.
To investigate how disordered eating is affected by bariatric surgery in adolescents with severe obesity, researchers used data from Teen-LABS, which enrolled 242 participants aged 19 years and under who mainly underwent Roux-en-Y gastric bypass (67%) or sleeve gastrectomy (28%) from 2007 to 2012 at five adolescent bariatric surgery centers.
The current analysis examined data from 141 participants in Teen-LABS who underwent bariatric surgery at a mean age of 16.8 years. Mean BMI was 51.5, most were girls (80%), and they had diverse race/ethnicity (66% were White).
Researchers also identified a control group of 83 adolescents of a similar age and gender who had diverse race/ethnicity (54% White) and a mean BMI of 46.9.
At year 6, data were available for 123 young adults in the surgery group (who by then had a mean BMI of 39.7) and 63 young adults in the nonsurgery group (who had a mean BMI of 52.6).
At baseline and year 6, participants replied to questionnaires that identified three eating disorders: continuous eating (eating in an unplanned and repetitious way between meals and snacks), objective overeating (eating a “large” amount of food without loss of control), and objective binge eating (eating a “large” amount of food with loss of control).
At baseline, rates of continuous eating, overeating, and binge eating were higher in the surgical group (50%, 40%, and 30%, respectively) than the nonsurgical group (40%, 22%, and 11%, respectively).
Six years later, when participants were aged 19-24 years, rates of continuous eating, overeating, and binge eating had declined in the surgical group (to 17%, 5%, and 1%, respectively). In the nonsurgical group, only continuous eating and overeating declined (to 24% and 7%, respectively), and binge eating increased slightly (to 13%).
Disordered eating associated with low self-worth, anxiety, and depression
In young adulthood in both groups, disordered eating was associated with lower self-worth. In the surgical group, it was also associated with lower weight-related quality of life, and in the nonsurgical group, it was also associated with anxiety and/or depression.
“The current findings cannot tell us whether disordered eating is a direct result or caused by anxiety, depression, low self-worth, or poor quality of life,” Dr. Decker said.
“These findings do give us insight about what other areas of clinical concern might present together [in] young adults (e.g., disordered eating, low self-esteem).”
Bariatric surgery affects the amount of food people can eat at one time, she noted in reply to a question from the audience. If people eat too much at a time they can experience vomiting, dumping syndrome (where certain food is “dumped” into the small intestine without being digested, causing nausea and vomiting), and plugging (a sense of food becoming stuck).
The home environment and transition to adulthood might impact disordered eating in young adults, she said in reply to another question, but these issues were not examined in this study.
A version of this article originally appeared on Medscape.com.
Kristina M. Decker, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital Medical Center, presented these findings during the virtual ObesityWeek 2020.
Dr. Decker and associates examined rates of disordered eating in more than 200 adolescents (aged 13-18 years) who were severely obese, of whom 141 underwent bariatric surgery and the remainder did not.
At baseline (presurgery), the teens in both groups had rates of disordered eating ranging from 11% to 50%, with higher rates in those who went on to have bariatric surgery.
Six years later, rates of disordered eating were much lower in those who had bariatric surgery.
The data nevertheless “underscore that young adults with persistent severe obesity are at high risk for poor health and well-being,” Dr. Decker said in an interview.
“This means disordered eating behaviors should be closely monitored” in all such patients, both those who undergo surgery and those who don’t, she stressed.
Robust findings because of long follow-up and controls
The findings are not unexpected, based on adult bariatric literature, but are “novel because of the age of the patients,” senior author Margaret H. Zeller, PhD, Cincinnati Children’s Hospital Medical Center and professor at the University of Cincinnati, added.
In a comment comment, psychologist Kajsa Järvholm, PhD, of the Childhood Obesity Unit at Skåne University Hospital, Malmö̈, Sweden, who has published related work, said that this is “a needed study.”
Notably, it had “long-term follow-up and a control group,” and it “confirms that adolescents are in better control of their eating after surgery.”
However, an important additional takeaway for clinicians is that “disordered eating is associated with other mental health problems and self-worth. Clinicians treating obesity must address problems related to eating disorders to improve outcomes and well-being,” she stressed.
How does bariatric surgery impact overeating, binge eating, in teens?
“For teens with severe obesity, metabolic and bariatric surgery is the most effective treatment for improved cardiometabolic functioning, weight loss, and improved quality of life,” Dr. Decker stressed.
However, pre- and postsurgical disordered eating behaviors have been associated with a lower percentage change in body mass index (BMI), although this has not been well studied.
To investigate how disordered eating is affected by bariatric surgery in adolescents with severe obesity, researchers used data from Teen-LABS, which enrolled 242 participants aged 19 years and under who mainly underwent Roux-en-Y gastric bypass (67%) or sleeve gastrectomy (28%) from 2007 to 2012 at five adolescent bariatric surgery centers.
The current analysis examined data from 141 participants in Teen-LABS who underwent bariatric surgery at a mean age of 16.8 years. Mean BMI was 51.5, most were girls (80%), and they had diverse race/ethnicity (66% were White).
Researchers also identified a control group of 83 adolescents of a similar age and gender who had diverse race/ethnicity (54% White) and a mean BMI of 46.9.
At year 6, data were available for 123 young adults in the surgery group (who by then had a mean BMI of 39.7) and 63 young adults in the nonsurgery group (who had a mean BMI of 52.6).
At baseline and year 6, participants replied to questionnaires that identified three eating disorders: continuous eating (eating in an unplanned and repetitious way between meals and snacks), objective overeating (eating a “large” amount of food without loss of control), and objective binge eating (eating a “large” amount of food with loss of control).
At baseline, rates of continuous eating, overeating, and binge eating were higher in the surgical group (50%, 40%, and 30%, respectively) than the nonsurgical group (40%, 22%, and 11%, respectively).
Six years later, when participants were aged 19-24 years, rates of continuous eating, overeating, and binge eating had declined in the surgical group (to 17%, 5%, and 1%, respectively). In the nonsurgical group, only continuous eating and overeating declined (to 24% and 7%, respectively), and binge eating increased slightly (to 13%).
Disordered eating associated with low self-worth, anxiety, and depression
In young adulthood in both groups, disordered eating was associated with lower self-worth. In the surgical group, it was also associated with lower weight-related quality of life, and in the nonsurgical group, it was also associated with anxiety and/or depression.
“The current findings cannot tell us whether disordered eating is a direct result or caused by anxiety, depression, low self-worth, or poor quality of life,” Dr. Decker said.
“These findings do give us insight about what other areas of clinical concern might present together [in] young adults (e.g., disordered eating, low self-esteem).”
Bariatric surgery affects the amount of food people can eat at one time, she noted in reply to a question from the audience. If people eat too much at a time they can experience vomiting, dumping syndrome (where certain food is “dumped” into the small intestine without being digested, causing nausea and vomiting), and plugging (a sense of food becoming stuck).
The home environment and transition to adulthood might impact disordered eating in young adults, she said in reply to another question, but these issues were not examined in this study.
A version of this article originally appeared on Medscape.com.
Kristina M. Decker, PhD, a postdoctoral fellow at Cincinnati Children’s Hospital Medical Center, presented these findings during the virtual ObesityWeek 2020.
Dr. Decker and associates examined rates of disordered eating in more than 200 adolescents (aged 13-18 years) who were severely obese, of whom 141 underwent bariatric surgery and the remainder did not.
At baseline (presurgery), the teens in both groups had rates of disordered eating ranging from 11% to 50%, with higher rates in those who went on to have bariatric surgery.
Six years later, rates of disordered eating were much lower in those who had bariatric surgery.
The data nevertheless “underscore that young adults with persistent severe obesity are at high risk for poor health and well-being,” Dr. Decker said in an interview.
“This means disordered eating behaviors should be closely monitored” in all such patients, both those who undergo surgery and those who don’t, she stressed.
Robust findings because of long follow-up and controls
The findings are not unexpected, based on adult bariatric literature, but are “novel because of the age of the patients,” senior author Margaret H. Zeller, PhD, Cincinnati Children’s Hospital Medical Center and professor at the University of Cincinnati, added.
In a comment comment, psychologist Kajsa Järvholm, PhD, of the Childhood Obesity Unit at Skåne University Hospital, Malmö̈, Sweden, who has published related work, said that this is “a needed study.”
Notably, it had “long-term follow-up and a control group,” and it “confirms that adolescents are in better control of their eating after surgery.”
However, an important additional takeaway for clinicians is that “disordered eating is associated with other mental health problems and self-worth. Clinicians treating obesity must address problems related to eating disorders to improve outcomes and well-being,” she stressed.
How does bariatric surgery impact overeating, binge eating, in teens?
“For teens with severe obesity, metabolic and bariatric surgery is the most effective treatment for improved cardiometabolic functioning, weight loss, and improved quality of life,” Dr. Decker stressed.
However, pre- and postsurgical disordered eating behaviors have been associated with a lower percentage change in body mass index (BMI), although this has not been well studied.
To investigate how disordered eating is affected by bariatric surgery in adolescents with severe obesity, researchers used data from Teen-LABS, which enrolled 242 participants aged 19 years and under who mainly underwent Roux-en-Y gastric bypass (67%) or sleeve gastrectomy (28%) from 2007 to 2012 at five adolescent bariatric surgery centers.
The current analysis examined data from 141 participants in Teen-LABS who underwent bariatric surgery at a mean age of 16.8 years. Mean BMI was 51.5, most were girls (80%), and they had diverse race/ethnicity (66% were White).
Researchers also identified a control group of 83 adolescents of a similar age and gender who had diverse race/ethnicity (54% White) and a mean BMI of 46.9.
At year 6, data were available for 123 young adults in the surgery group (who by then had a mean BMI of 39.7) and 63 young adults in the nonsurgery group (who had a mean BMI of 52.6).
At baseline and year 6, participants replied to questionnaires that identified three eating disorders: continuous eating (eating in an unplanned and repetitious way between meals and snacks), objective overeating (eating a “large” amount of food without loss of control), and objective binge eating (eating a “large” amount of food with loss of control).
At baseline, rates of continuous eating, overeating, and binge eating were higher in the surgical group (50%, 40%, and 30%, respectively) than the nonsurgical group (40%, 22%, and 11%, respectively).
Six years later, when participants were aged 19-24 years, rates of continuous eating, overeating, and binge eating had declined in the surgical group (to 17%, 5%, and 1%, respectively). In the nonsurgical group, only continuous eating and overeating declined (to 24% and 7%, respectively), and binge eating increased slightly (to 13%).
Disordered eating associated with low self-worth, anxiety, and depression
In young adulthood in both groups, disordered eating was associated with lower self-worth. In the surgical group, it was also associated with lower weight-related quality of life, and in the nonsurgical group, it was also associated with anxiety and/or depression.
“The current findings cannot tell us whether disordered eating is a direct result or caused by anxiety, depression, low self-worth, or poor quality of life,” Dr. Decker said.
“These findings do give us insight about what other areas of clinical concern might present together [in] young adults (e.g., disordered eating, low self-esteem).”
Bariatric surgery affects the amount of food people can eat at one time, she noted in reply to a question from the audience. If people eat too much at a time they can experience vomiting, dumping syndrome (where certain food is “dumped” into the small intestine without being digested, causing nausea and vomiting), and plugging (a sense of food becoming stuck).
The home environment and transition to adulthood might impact disordered eating in young adults, she said in reply to another question, but these issues were not examined in this study.
A version of this article originally appeared on Medscape.com.
COVID-19 risks in rheumatic disease remain unclear
ACR 2020 studies offer conflicting findings.
Among people with COVID-19, those with systemic autoimmune rheumatic diseases had an elevated 30-day risk of hospitalization, ICU admission, need for mechanical ventilation, and acute kidney injury, compared to a group without rheumatic diseases at 4 months in a match-controlled study.
When investigators expanded the study to 6 months, the difference in need for mechanical ventilation disappeared. However, relative risk for venous thromboembolism (VTE) emerged as 74% higher among people with COVID-19 and with rheumatic disease, said Kristin D’Silva, MD, who presented the findings during a plenary session at the virtual annual meeting of the American College of Rheumatology. She noted that rheumatic disease itself could contribute to VTE risk.
Comorbidities including hypertension, diabetes, and asthma were more common among people with systemic autoimmune rheumatic diseases (SARDs). After adjustment for comorbidities, “the risks of hospitalization and ICU admission were attenuated, suggesting comorbidities are likely key mediators of the increased risk of severe COVID-19 outcomes observed in SARDs patients versus comparators,” Dr. D’Silva, a rheumatology fellow at Massachusetts General Hospital in Boston, said in an interview.
“The risk of venous thromboembolism persisted even after adjusting for comorbidities,” Dr. D’Silva said. Patients with SARDs should be closely monitored for VTE during COVID-19 infection, she added. “Patients with significant cardiovascular, pulmonary, and metabolic comorbidities should be closely monitored for severe COVID-19.”
At the same time, a systematic review of 15 published studies revealed a low incidence of COVID-19 infection among people with rheumatic disease. Furthermore, most experienced a mild clinical course and low mortality, Akhil Sood, MD, said when presenting results of his poster at the meeting.
Underlying immunosuppression, chronic inflammation, comorbidities, and disparities based on racial, ethnic, and socioeconomic status could predispose people with rheumatic disease to poorer COVID-19 outcomes. However, the risks and outcomes of COVID-19 infection among this population “are not well understood,” said Dr. Sood, a second-year resident in internal medicine at the University of Texas Medical Branch in Galveston.
Elevated risks in match-controlled study
Dr. D’Silva and colleagues examined a COVID-19 population and compared 716 people with SARDs and another 716 people from the general public at 4 months, as well as 2,379 people each in similar groups at 6 months. They used real-time electronic medical record data from the TriNetX research network to identify ICD-10 codes for inflammatory arthritis, connective tissue diseases, and systemic vasculitis. They also used ICD-10 codes and positive PCR tests to identify people with COVID-19.
Mean age was 57 years and women accounted for 79% of both groups evaluated at 4 months. Those with SARDs were 23% more likely to be hospitalized (relative risk, 1.23; 95% confidence interval, 1.01-1.50). This group was 75% more likely to be admitted to the ICU (RR, 1.75; 95% CI, 1.11-2.75), 77% more likely to require mechanical ventilation (RR, 1.77; 95% CI, 1.06-2.96), and 83% more likely to experience acute kidney injury (RR, 1.83; 95% CI, 1.11-3.00).
Risk of death was not significantly higher in the SARDs group (RR, 1.16; 95% CI, 0.73-1.86).
When Dr. D’Silva expanded the study to more people at 6 months, they added additional 30-day outcomes of interest: renal replacement therapy, VTE, and ischemic stroke. Risk of need for renal replacement therapy, for example, was 81% higher in the SARDs group (RR, 1.81; 95% CI, 1.07-3.07). Risk of stroke was not significantly different between groups.The improvement in mechanical ventilation risk between 4 and 6 months was not completely unexpected, Dr. D’Silva said. The relative risk dropped from 1.77 to 1.05. “This is not particularly surprising given national trends in the general population reporting decreased severe outcomes of COVID-19 including mortality as the pandemic progresses. This is likely multifactorial including changes in COVID-19 management (such as increasing use of nonintubated prone positioning rather than early intubation and treatments such as dexamethasone and remdesivir), decreased strain on hospitals and staffing compared to the early crisis phase of the pandemic, and higher testing capacity leading to detection of milder cases.”
When the 6-month analysis was further adjusted for comorbidities and a history of prior hospitalization within 1 year, only risk for acute kidney injury and VTE remained significant with relative risks of 1.33 and 1.60, respectively, likely because comorbidities are causal intermediates of COVID-19 30-day outcomes rather than confounders.
When asked to comment on the results, session comoderator Victoria K. Shanmugam, MD, said in an interview that the study “is of great interest both to rheumatologists and to patients with rheumatic disease.”
The higher risk of hospitalization, ICU admission, mechanical ventilation, acute kidney injury, and heart failure “is an important finding with implications for how our patients navigate risk during this pandemic,” said Dr. Shanmugam, director of the division of rheumatology at George Washington University in Washington.
Lower risks emerge in systematic review
The 15 observational studies in the systematic review included 11,815 participants. A total of 179, or 1.5%, tested positive for COVID-19.
“The incidence of COVID-19 infection among patients with rheumatic disease was low,” Dr. Sood said.
Within the COVID-19-positive group, almost 50% required hospitalization, 10% required ICU admission, and 8% died. The pooled event rate for hospitalization was 0.440 (95% CI, 0.296-0.596), while for ICU admission it was 0.132 (95% CI, 0.087-0.194) and for death it was 0.125 (95% CI, 0.082-0.182).
Different calculations of risk
The two studies seem to offer contradictory findings, but the disparities could be explained by study design differences. For example, Dr. D’Silva’s study evaluated a population with COVID-19 and compared those with SARDs versus a matched group from the general public. Dr. Sood and colleagues assessed study populations with rheumatic disease and assessed incidence of SARS-CoV-2 infection and difference in outcomes.
“We are asking very different questions,” Dr. D’Silva said.
“The study by D’Silva et al. was able to account for different factors to reduce confounding,” Dr. Sood said, adding that Dr. D’Silva and colleagues included a high proportion of minorities, compared with a less diverse population in the systematic review, which featured a large number of studies from Italy.
The authors of the two studies had no relevant financial disclosures to report.
SOURCES: D’Silva K et al. Arthritis Rheumatol. 2020;72(suppl 10): Abstract 0430, and Sood A et al. Arthritis Rheumatol. 2020;72(suppl 10): Abstract 0008.
ACR 2020 studies offer conflicting findings.
ACR 2020 studies offer conflicting findings.
Among people with COVID-19, those with systemic autoimmune rheumatic diseases had an elevated 30-day risk of hospitalization, ICU admission, need for mechanical ventilation, and acute kidney injury, compared to a group without rheumatic diseases at 4 months in a match-controlled study.
When investigators expanded the study to 6 months, the difference in need for mechanical ventilation disappeared. However, relative risk for venous thromboembolism (VTE) emerged as 74% higher among people with COVID-19 and with rheumatic disease, said Kristin D’Silva, MD, who presented the findings during a plenary session at the virtual annual meeting of the American College of Rheumatology. She noted that rheumatic disease itself could contribute to VTE risk.
Comorbidities including hypertension, diabetes, and asthma were more common among people with systemic autoimmune rheumatic diseases (SARDs). After adjustment for comorbidities, “the risks of hospitalization and ICU admission were attenuated, suggesting comorbidities are likely key mediators of the increased risk of severe COVID-19 outcomes observed in SARDs patients versus comparators,” Dr. D’Silva, a rheumatology fellow at Massachusetts General Hospital in Boston, said in an interview.
“The risk of venous thromboembolism persisted even after adjusting for comorbidities,” Dr. D’Silva said. Patients with SARDs should be closely monitored for VTE during COVID-19 infection, she added. “Patients with significant cardiovascular, pulmonary, and metabolic comorbidities should be closely monitored for severe COVID-19.”
At the same time, a systematic review of 15 published studies revealed a low incidence of COVID-19 infection among people with rheumatic disease. Furthermore, most experienced a mild clinical course and low mortality, Akhil Sood, MD, said when presenting results of his poster at the meeting.
Underlying immunosuppression, chronic inflammation, comorbidities, and disparities based on racial, ethnic, and socioeconomic status could predispose people with rheumatic disease to poorer COVID-19 outcomes. However, the risks and outcomes of COVID-19 infection among this population “are not well understood,” said Dr. Sood, a second-year resident in internal medicine at the University of Texas Medical Branch in Galveston.
Elevated risks in match-controlled study
Dr. D’Silva and colleagues examined a COVID-19 population and compared 716 people with SARDs and another 716 people from the general public at 4 months, as well as 2,379 people each in similar groups at 6 months. They used real-time electronic medical record data from the TriNetX research network to identify ICD-10 codes for inflammatory arthritis, connective tissue diseases, and systemic vasculitis. They also used ICD-10 codes and positive PCR tests to identify people with COVID-19.
Mean age was 57 years and women accounted for 79% of both groups evaluated at 4 months. Those with SARDs were 23% more likely to be hospitalized (relative risk, 1.23; 95% confidence interval, 1.01-1.50). This group was 75% more likely to be admitted to the ICU (RR, 1.75; 95% CI, 1.11-2.75), 77% more likely to require mechanical ventilation (RR, 1.77; 95% CI, 1.06-2.96), and 83% more likely to experience acute kidney injury (RR, 1.83; 95% CI, 1.11-3.00).
Risk of death was not significantly higher in the SARDs group (RR, 1.16; 95% CI, 0.73-1.86).
When Dr. D’Silva expanded the study to more people at 6 months, they added additional 30-day outcomes of interest: renal replacement therapy, VTE, and ischemic stroke. Risk of need for renal replacement therapy, for example, was 81% higher in the SARDs group (RR, 1.81; 95% CI, 1.07-3.07). Risk of stroke was not significantly different between groups.The improvement in mechanical ventilation risk between 4 and 6 months was not completely unexpected, Dr. D’Silva said. The relative risk dropped from 1.77 to 1.05. “This is not particularly surprising given national trends in the general population reporting decreased severe outcomes of COVID-19 including mortality as the pandemic progresses. This is likely multifactorial including changes in COVID-19 management (such as increasing use of nonintubated prone positioning rather than early intubation and treatments such as dexamethasone and remdesivir), decreased strain on hospitals and staffing compared to the early crisis phase of the pandemic, and higher testing capacity leading to detection of milder cases.”
When the 6-month analysis was further adjusted for comorbidities and a history of prior hospitalization within 1 year, only risk for acute kidney injury and VTE remained significant with relative risks of 1.33 and 1.60, respectively, likely because comorbidities are causal intermediates of COVID-19 30-day outcomes rather than confounders.
When asked to comment on the results, session comoderator Victoria K. Shanmugam, MD, said in an interview that the study “is of great interest both to rheumatologists and to patients with rheumatic disease.”
The higher risk of hospitalization, ICU admission, mechanical ventilation, acute kidney injury, and heart failure “is an important finding with implications for how our patients navigate risk during this pandemic,” said Dr. Shanmugam, director of the division of rheumatology at George Washington University in Washington.
Lower risks emerge in systematic review
The 15 observational studies in the systematic review included 11,815 participants. A total of 179, or 1.5%, tested positive for COVID-19.
“The incidence of COVID-19 infection among patients with rheumatic disease was low,” Dr. Sood said.
Within the COVID-19-positive group, almost 50% required hospitalization, 10% required ICU admission, and 8% died. The pooled event rate for hospitalization was 0.440 (95% CI, 0.296-0.596), while for ICU admission it was 0.132 (95% CI, 0.087-0.194) and for death it was 0.125 (95% CI, 0.082-0.182).
Different calculations of risk
The two studies seem to offer contradictory findings, but the disparities could be explained by study design differences. For example, Dr. D’Silva’s study evaluated a population with COVID-19 and compared those with SARDs versus a matched group from the general public. Dr. Sood and colleagues assessed study populations with rheumatic disease and assessed incidence of SARS-CoV-2 infection and difference in outcomes.
“We are asking very different questions,” Dr. D’Silva said.
“The study by D’Silva et al. was able to account for different factors to reduce confounding,” Dr. Sood said, adding that Dr. D’Silva and colleagues included a high proportion of minorities, compared with a less diverse population in the systematic review, which featured a large number of studies from Italy.
The authors of the two studies had no relevant financial disclosures to report.
SOURCES: D’Silva K et al. Arthritis Rheumatol. 2020;72(suppl 10): Abstract 0430, and Sood A et al. Arthritis Rheumatol. 2020;72(suppl 10): Abstract 0008.
Among people with COVID-19, those with systemic autoimmune rheumatic diseases had an elevated 30-day risk of hospitalization, ICU admission, need for mechanical ventilation, and acute kidney injury, compared to a group without rheumatic diseases at 4 months in a match-controlled study.
When investigators expanded the study to 6 months, the difference in need for mechanical ventilation disappeared. However, relative risk for venous thromboembolism (VTE) emerged as 74% higher among people with COVID-19 and with rheumatic disease, said Kristin D’Silva, MD, who presented the findings during a plenary session at the virtual annual meeting of the American College of Rheumatology. She noted that rheumatic disease itself could contribute to VTE risk.
Comorbidities including hypertension, diabetes, and asthma were more common among people with systemic autoimmune rheumatic diseases (SARDs). After adjustment for comorbidities, “the risks of hospitalization and ICU admission were attenuated, suggesting comorbidities are likely key mediators of the increased risk of severe COVID-19 outcomes observed in SARDs patients versus comparators,” Dr. D’Silva, a rheumatology fellow at Massachusetts General Hospital in Boston, said in an interview.
“The risk of venous thromboembolism persisted even after adjusting for comorbidities,” Dr. D’Silva said. Patients with SARDs should be closely monitored for VTE during COVID-19 infection, she added. “Patients with significant cardiovascular, pulmonary, and metabolic comorbidities should be closely monitored for severe COVID-19.”
At the same time, a systematic review of 15 published studies revealed a low incidence of COVID-19 infection among people with rheumatic disease. Furthermore, most experienced a mild clinical course and low mortality, Akhil Sood, MD, said when presenting results of his poster at the meeting.
Underlying immunosuppression, chronic inflammation, comorbidities, and disparities based on racial, ethnic, and socioeconomic status could predispose people with rheumatic disease to poorer COVID-19 outcomes. However, the risks and outcomes of COVID-19 infection among this population “are not well understood,” said Dr. Sood, a second-year resident in internal medicine at the University of Texas Medical Branch in Galveston.
Elevated risks in match-controlled study
Dr. D’Silva and colleagues examined a COVID-19 population and compared 716 people with SARDs and another 716 people from the general public at 4 months, as well as 2,379 people each in similar groups at 6 months. They used real-time electronic medical record data from the TriNetX research network to identify ICD-10 codes for inflammatory arthritis, connective tissue diseases, and systemic vasculitis. They also used ICD-10 codes and positive PCR tests to identify people with COVID-19.
Mean age was 57 years and women accounted for 79% of both groups evaluated at 4 months. Those with SARDs were 23% more likely to be hospitalized (relative risk, 1.23; 95% confidence interval, 1.01-1.50). This group was 75% more likely to be admitted to the ICU (RR, 1.75; 95% CI, 1.11-2.75), 77% more likely to require mechanical ventilation (RR, 1.77; 95% CI, 1.06-2.96), and 83% more likely to experience acute kidney injury (RR, 1.83; 95% CI, 1.11-3.00).
Risk of death was not significantly higher in the SARDs group (RR, 1.16; 95% CI, 0.73-1.86).
When Dr. D’Silva expanded the study to more people at 6 months, they added additional 30-day outcomes of interest: renal replacement therapy, VTE, and ischemic stroke. Risk of need for renal replacement therapy, for example, was 81% higher in the SARDs group (RR, 1.81; 95% CI, 1.07-3.07). Risk of stroke was not significantly different between groups.The improvement in mechanical ventilation risk between 4 and 6 months was not completely unexpected, Dr. D’Silva said. The relative risk dropped from 1.77 to 1.05. “This is not particularly surprising given national trends in the general population reporting decreased severe outcomes of COVID-19 including mortality as the pandemic progresses. This is likely multifactorial including changes in COVID-19 management (such as increasing use of nonintubated prone positioning rather than early intubation and treatments such as dexamethasone and remdesivir), decreased strain on hospitals and staffing compared to the early crisis phase of the pandemic, and higher testing capacity leading to detection of milder cases.”
When the 6-month analysis was further adjusted for comorbidities and a history of prior hospitalization within 1 year, only risk for acute kidney injury and VTE remained significant with relative risks of 1.33 and 1.60, respectively, likely because comorbidities are causal intermediates of COVID-19 30-day outcomes rather than confounders.
When asked to comment on the results, session comoderator Victoria K. Shanmugam, MD, said in an interview that the study “is of great interest both to rheumatologists and to patients with rheumatic disease.”
The higher risk of hospitalization, ICU admission, mechanical ventilation, acute kidney injury, and heart failure “is an important finding with implications for how our patients navigate risk during this pandemic,” said Dr. Shanmugam, director of the division of rheumatology at George Washington University in Washington.
Lower risks emerge in systematic review
The 15 observational studies in the systematic review included 11,815 participants. A total of 179, or 1.5%, tested positive for COVID-19.
“The incidence of COVID-19 infection among patients with rheumatic disease was low,” Dr. Sood said.
Within the COVID-19-positive group, almost 50% required hospitalization, 10% required ICU admission, and 8% died. The pooled event rate for hospitalization was 0.440 (95% CI, 0.296-0.596), while for ICU admission it was 0.132 (95% CI, 0.087-0.194) and for death it was 0.125 (95% CI, 0.082-0.182).
Different calculations of risk
The two studies seem to offer contradictory findings, but the disparities could be explained by study design differences. For example, Dr. D’Silva’s study evaluated a population with COVID-19 and compared those with SARDs versus a matched group from the general public. Dr. Sood and colleagues assessed study populations with rheumatic disease and assessed incidence of SARS-CoV-2 infection and difference in outcomes.
“We are asking very different questions,” Dr. D’Silva said.
“The study by D’Silva et al. was able to account for different factors to reduce confounding,” Dr. Sood said, adding that Dr. D’Silva and colleagues included a high proportion of minorities, compared with a less diverse population in the systematic review, which featured a large number of studies from Italy.
The authors of the two studies had no relevant financial disclosures to report.
SOURCES: D’Silva K et al. Arthritis Rheumatol. 2020;72(suppl 10): Abstract 0430, and Sood A et al. Arthritis Rheumatol. 2020;72(suppl 10): Abstract 0008.
FROM ACR 2020
Biden victory: What it means for COVID, health care
The former vice president has sketched out a big health agenda: ramping up the federal response to COVID-19, boosting the Affordable Care Act, creating a new “public option” to cover uninsured Americans, and expanding Medicare and Medicaid.
But the president-elect’s long to-do list on health is likely to face significant roadblocks in Congress and the courts, experts say.
For instance, Biden’s ambitious proposals on COVID-19 -- including his recent call for a national mask mandate -- could be waylaid by legal challenges and run into political hurdles on Capitol Hill, where he may face a divided Congress.
Joseph Antos, PhD, a health policy expert with the conservative American Enterprise Institute, predicts Biden will encounter the same type of congressional “gridlock situation” that President Barack Obama ran into during his second term.
“We have a situation that has been like this for a very, very long time -- lack of cooperation, lack of recognition that either party is capable of rising above their own electoral views to deal with problems that the country actually has.”
Antos also suggests that Biden may also face enormous political pressure to address the economic fallout from the coronavirus, including record unemployment and business closures, before anything else.
“I think it’s really going to be efforts that are intended to promote economic development and promote the economy,” he says.
In addition, Biden’s plans to expand Obamacare might face a new challenge from the Supreme Court in the year ahead. This month, the high court will take up a new case seeking to overturn the law.
Even so, experts say Biden’s plans on COVID-19 and expanding health care are likely to define his tenure in the White House as a central focus of his presidency.
“Health care will be at the very top of the list of the president’s priorities,” says Sabrina Corlette, JD, co-director of the Center on Health Insurance Reforms at Georgetown University’s McCourt School of Public Policy. “I do think, however, that the administration is going to be very preoccupied with the response to COVID-19 and the economic fallout … particularly in the first year.”
Here’s a closer look at what we can expect from a Biden presidency.
COVID-19: Federalizing response efforts
Biden will move to federalize the response to COVID-19. He has said he will take back major responsibilities from the states -- such as setting national policies on mask wearing, social distancing, and the reopening of schools and businesses, based on CDC guidance. In the days leading up to the election, Biden called for a national mask mandate, after waffling on the issue throughout the summer.
He has said he will let public health science drive political policy. Biden is also planning to create his own task force to advise officials during the transition on managing the new surge in COVID-19 cases, vaccine safety and protecting at-risk populations, Politico reported this week. He received a virtual briefing on the pandemic from a panel of experts as he awaited the election’s outcome.
“I think we will no longer have this confused and contradictory public messaging,” Corlette says, “but I also think there will be humility and the recognition that the evidence is evolving -- that we don’t have all the answers, but we’re learning as we go.”
But national mandates on masks and social distancing will be challenging to enforce, experts say. They are also likely to face pushback from business interests, opposition from public officials in GOP-led states, and even legal challenges.
Biden’s ability to work with Congress -- or not -- may determine whether he is able to implement some of the key components of his coronavirus action plan, which includes:
- Providing free COVID-19 testing for all Americans
- Hiring 100,000 contact tracers
- Eliminating out-of-pocket expenses for coronavirus treatment
- Delivering “sufficient” PPE for essential workers
- Supporting science-backed vaccines and medical treatments being developed
- Requiring the reopening of businesses, workplaces, and schools only after “sufficient” reductions in community transmission -- under evidence-based protocols put forward by the CDC
- Giving emergency paid leave for workers dislocated by the pandemic and more financial aid for workers, families, and small businesses
- Shoring up safeguards to protect at-risk Americans, including older people
- Boosting pay for health care workers on the front lines
Biden has not detailed how he would pay for many of these, beyond promising to force wealthy Americans to “pay their fair share” of taxes to help. He has proposed a tax increase on Americans making more than $400,000 a year, which would require congressional approval.
Antos says he expects Biden’s proposed COVID-19 action plan to be virtually the same as Trump’s in two areas: efforts to develop a vaccine and antiviral treatments.
The administration has spent some $225 million on COVID-19 testing efforts, with a particular focus on rural areas.
Trump launched Operation Warp Speed to fast-track a vaccine. As part of that, the federal government has contracted with six drug companies, spending nearly $11 billion. The operation aims to provide at least 300 million doses of a coronavirus vaccine by January 2021.
Antos would like to see “a more sophisticated approach to social distancing” from the president-elect that takes into account the different challenges facing Americans depending on their income, work situation, and other factors during the pandemic.
“There are a lot of people in this country where working from home is fine and their jobs are secure,” he notes. “It’s the person who used to work at a restaurant that closed, it’s the line worker at a factory that has severely cut back its hours. It’s basically lower-middle-class people, low-income people, middle-class people, and it’s not the elite.
“And the policies have not given enough consideration to the fact that their circumstances and their tradeoffs would differ from the tradeoffs of somebody who doesn’t have anything to worry about economically.
“So, what we need is a more supple policy [that] will give people the information they need and give them the financial support that they also need … so they can make good decisions for themselves and their families. And we basically haven’t done that.”
Obamacare on the blocks?
The Supreme Court’s decision to take up another case seeking to overturn the Affordable Care Act could hand Biden’s health agenda a major setback -- and put the medical care for millions of Americans in jeopardy.
On Nov. 10, the high court will hear oral arguments on a lawsuit that would strike down all of Obamacare. A decision is not expected until next year.
The court has previously upheld the 2010 law, which Biden helped usher through Congress as vice president. But the addition of right-leaning Supreme Court Justice Amy Coney Barrett to the bench last month gives the court a clear conservative majority that could mean the end of Obamacare, legal experts say.
Republicans have opposed the law since its passage, but they have been unable to muster the votes to repeal it, or to pass an alternative
Antos, from the American Enterprise Institute, notes conservatives believe the law has increased costs for health care and insurance over the past decade, in part because of its protections for Americans with preexisting conditions and requiring insurers to provide comprehensive “gold-plated” policies.
“It’s driven up costs, offers plans that are not very strong, put high-risk folks into the same [insurance pool], which has increased costs for everyone, the employer mandate … these are all the reasons,” he says.
The Supreme Court isn’t expected to deliver a decision on the Affordable Care Act before the middle of next year. But the uncertainty will likely push back Biden’s proposals to expand on the law.
Overturning Obamacare would have huge impacts on millions of Americans:
- As many as 133 million Americans -- roughly half the U.S. population -- with preexisting conditions could find it harder, if not impossible, to find affordable health insurance. That figure does not include Americans infected with COVID-19.
- About 165 million who require expensive treatments -- for cancer and other conditions -- would no longer be protected from huge costs for care by federal caps on out-of-pocket expenditures the Affordable Care Act requires.
- An estimated 21 million who now buy insurance through the Obamacare Marketplaces could lose their coverage.
- Another 12 million on Medicaid could find themselves without insurance.
- At least 2 million young adults ages 26 and under, now on their parents’ health policies, could be kicked off.
- Millions of people who use Medicare could face higher costs.
- Federal subsidies for lower-income Americans to buy policies would disappear.
Throughout the campaign, Biden repeatedly stressed the need to preserve the law’s provision barring insurance companies from refusing coverage for Americans with preexisting conditions, such as diabetes, cancer, and heart disease. It also outlaws charging higher premiums on the basis of health status, age, or gender.
Biden has also pledged to bolster the law as president.
He has proposed a variety of add-ons to the Affordable Care Act he says will “insure more than an estimated 97% of Americans,” according to the Biden campaign site.
Biden’s proposals include offering larger federal subsidies to help low- and middle-income Americans pay for policies purchased through Obamacare insurance Marketplaces.
The boldest of Biden’s proposals is the creation of a “public option” for insurance -- a Medicare-like program that small businesses and individuals could choose if they do not have coverage, cannot afford it, or don’t like their employer-based coverage.
It would also automatically enroll millions of uninsured Americans living in the 14 states that have not expanded Medicaid, which covers low-income people.
But such a plan would require congressional approval -- including a “super majority” of 60 Senate votes to block a likely GOP filibuster. That will be a significant challenge Biden will have to overcome, with Congress so evenly divided.
The White House would also have to defeat heavy lobbying from some of the most influential industry interest groups in Washington, Corlette says.
“I’m not even confident they would get all the Democrat votes,” she says.
“So, it’s a going to be an uphill battle to get a public option passed.”
Taken together, Biden’s plans for expanding Obamacare are projected to cost $750 billion over 10 years. He has said much of that financing would come from increasing taxes on the wealthy.
That means it would likely require congressional approval, which Antos suggests is unlikely given the polarization on Capitol Hill.
Medicare, Medicaid, and drug costs
Biden has called for a host of reforms targeting Medicare, Medicaid, and rising drug costs.
On Medicare, which primarily covers seniors 65 and older, Biden has proposed lowering the eligibility age from 65 to 60. That could extend Medicare to up to 20 million more Americans.
On Medicaid, the health care safety net for low-income and disabled Americans, the president-elect supports increased federal funding to states during the current economic crisis, and potentially beyond.
Medicare is likely to become a key focus of the new administration, in light of the pressures the pandemic is placing on Medicare funding.
In April, Medicare’s trustees said that the Part A trust fund for the program, which pays for hospital and inpatient care, could start to run dry in 2026.
But those projections did not include the impact of COVID-19. Some economists have since projected that Medicare Part A could become insolvent as early as 2022.
Medicare Part B, which pays for doctor and outpatient costs, is funded by general tax funding and beneficiary insurance premiums, so it is not in danger of drying up.
Adding to those pressures is an executive order Trump signed in August temporarily deferring payroll taxes, a primary funding vehicle for Medicare and Social Security.
Under these taxes, employees pay 6.2% of their earnings (on annual income up to $137,700) toward Social Security and 1.45% for Medicare taxes each pay period. Employers pay the same rate per paycheck, adding up to a combined 12.4% Social Security tax and 2.9% Medicare tax.
Biden has said he would reverse the tax cut when he takes office.
But to get a handle on Medicare and Medicaid funding issues, he is likely to need congressional support. Corlette and other experts say that could be a challenge while the nation remains in the grip of the coronavirus pandemic.
In addition to his Medicare and Medicaid reforms, Biden has proposed several plans to lower drug prices, a subset of rising health care and insurance costs.
U.S. spending on prescription drugs has increased nearly 42% over the past decade -- from $253.1 billion in 2010 to $358.7 billion in 2020 (projected) -- according to the Centers for Medicare & Medicaid Services.
In 2020, retail prices for 460 commonly prescribed drugs have spiked an average of 5.2%, according to new analysis by 3 Axis Advisors, a health research firm.
That’s more than double the projected rate of inflation.
To control drug costs, Biden supports legislation approved by the Democratic-led House of Representatives last year that would empower Medicare to negotiate drug prices with drug companies, as private insurers do.
Federal law now bars Medicare from negotiating prices on behalf of the 67.7 million Americans who use it. Drug companies and many GOP leaders argue that the current law is necessary to allow them to spend more on research and development of new medications.
In addition, Biden supports the idea of lifting bans on importing drugs from foreign countries with lower costs.
He also backs creating an independent review board to set price caps for new medications with no competitors; making high-quality generics more available; ending tax breaks for drug company advertising; and limiting their leeway in raising prices.
All of these proposals would likely require congressional approval and could face legal challenges in the courts.
This article first appeared on WebMD.com.
The former vice president has sketched out a big health agenda: ramping up the federal response to COVID-19, boosting the Affordable Care Act, creating a new “public option” to cover uninsured Americans, and expanding Medicare and Medicaid.
But the president-elect’s long to-do list on health is likely to face significant roadblocks in Congress and the courts, experts say.
For instance, Biden’s ambitious proposals on COVID-19 -- including his recent call for a national mask mandate -- could be waylaid by legal challenges and run into political hurdles on Capitol Hill, where he may face a divided Congress.
Joseph Antos, PhD, a health policy expert with the conservative American Enterprise Institute, predicts Biden will encounter the same type of congressional “gridlock situation” that President Barack Obama ran into during his second term.
“We have a situation that has been like this for a very, very long time -- lack of cooperation, lack of recognition that either party is capable of rising above their own electoral views to deal with problems that the country actually has.”
Antos also suggests that Biden may also face enormous political pressure to address the economic fallout from the coronavirus, including record unemployment and business closures, before anything else.
“I think it’s really going to be efforts that are intended to promote economic development and promote the economy,” he says.
In addition, Biden’s plans to expand Obamacare might face a new challenge from the Supreme Court in the year ahead. This month, the high court will take up a new case seeking to overturn the law.
Even so, experts say Biden’s plans on COVID-19 and expanding health care are likely to define his tenure in the White House as a central focus of his presidency.
“Health care will be at the very top of the list of the president’s priorities,” says Sabrina Corlette, JD, co-director of the Center on Health Insurance Reforms at Georgetown University’s McCourt School of Public Policy. “I do think, however, that the administration is going to be very preoccupied with the response to COVID-19 and the economic fallout … particularly in the first year.”
Here’s a closer look at what we can expect from a Biden presidency.
COVID-19: Federalizing response efforts
Biden will move to federalize the response to COVID-19. He has said he will take back major responsibilities from the states -- such as setting national policies on mask wearing, social distancing, and the reopening of schools and businesses, based on CDC guidance. In the days leading up to the election, Biden called for a national mask mandate, after waffling on the issue throughout the summer.
He has said he will let public health science drive political policy. Biden is also planning to create his own task force to advise officials during the transition on managing the new surge in COVID-19 cases, vaccine safety and protecting at-risk populations, Politico reported this week. He received a virtual briefing on the pandemic from a panel of experts as he awaited the election’s outcome.
“I think we will no longer have this confused and contradictory public messaging,” Corlette says, “but I also think there will be humility and the recognition that the evidence is evolving -- that we don’t have all the answers, but we’re learning as we go.”
But national mandates on masks and social distancing will be challenging to enforce, experts say. They are also likely to face pushback from business interests, opposition from public officials in GOP-led states, and even legal challenges.
Biden’s ability to work with Congress -- or not -- may determine whether he is able to implement some of the key components of his coronavirus action plan, which includes:
- Providing free COVID-19 testing for all Americans
- Hiring 100,000 contact tracers
- Eliminating out-of-pocket expenses for coronavirus treatment
- Delivering “sufficient” PPE for essential workers
- Supporting science-backed vaccines and medical treatments being developed
- Requiring the reopening of businesses, workplaces, and schools only after “sufficient” reductions in community transmission -- under evidence-based protocols put forward by the CDC
- Giving emergency paid leave for workers dislocated by the pandemic and more financial aid for workers, families, and small businesses
- Shoring up safeguards to protect at-risk Americans, including older people
- Boosting pay for health care workers on the front lines
Biden has not detailed how he would pay for many of these, beyond promising to force wealthy Americans to “pay their fair share” of taxes to help. He has proposed a tax increase on Americans making more than $400,000 a year, which would require congressional approval.
Antos says he expects Biden’s proposed COVID-19 action plan to be virtually the same as Trump’s in two areas: efforts to develop a vaccine and antiviral treatments.
The administration has spent some $225 million on COVID-19 testing efforts, with a particular focus on rural areas.
Trump launched Operation Warp Speed to fast-track a vaccine. As part of that, the federal government has contracted with six drug companies, spending nearly $11 billion. The operation aims to provide at least 300 million doses of a coronavirus vaccine by January 2021.
Antos would like to see “a more sophisticated approach to social distancing” from the president-elect that takes into account the different challenges facing Americans depending on their income, work situation, and other factors during the pandemic.
“There are a lot of people in this country where working from home is fine and their jobs are secure,” he notes. “It’s the person who used to work at a restaurant that closed, it’s the line worker at a factory that has severely cut back its hours. It’s basically lower-middle-class people, low-income people, middle-class people, and it’s not the elite.
“And the policies have not given enough consideration to the fact that their circumstances and their tradeoffs would differ from the tradeoffs of somebody who doesn’t have anything to worry about economically.
“So, what we need is a more supple policy [that] will give people the information they need and give them the financial support that they also need … so they can make good decisions for themselves and their families. And we basically haven’t done that.”
Obamacare on the blocks?
The Supreme Court’s decision to take up another case seeking to overturn the Affordable Care Act could hand Biden’s health agenda a major setback -- and put the medical care for millions of Americans in jeopardy.
On Nov. 10, the high court will hear oral arguments on a lawsuit that would strike down all of Obamacare. A decision is not expected until next year.
The court has previously upheld the 2010 law, which Biden helped usher through Congress as vice president. But the addition of right-leaning Supreme Court Justice Amy Coney Barrett to the bench last month gives the court a clear conservative majority that could mean the end of Obamacare, legal experts say.
Republicans have opposed the law since its passage, but they have been unable to muster the votes to repeal it, or to pass an alternative
Antos, from the American Enterprise Institute, notes conservatives believe the law has increased costs for health care and insurance over the past decade, in part because of its protections for Americans with preexisting conditions and requiring insurers to provide comprehensive “gold-plated” policies.
“It’s driven up costs, offers plans that are not very strong, put high-risk folks into the same [insurance pool], which has increased costs for everyone, the employer mandate … these are all the reasons,” he says.
The Supreme Court isn’t expected to deliver a decision on the Affordable Care Act before the middle of next year. But the uncertainty will likely push back Biden’s proposals to expand on the law.
Overturning Obamacare would have huge impacts on millions of Americans:
- As many as 133 million Americans -- roughly half the U.S. population -- with preexisting conditions could find it harder, if not impossible, to find affordable health insurance. That figure does not include Americans infected with COVID-19.
- About 165 million who require expensive treatments -- for cancer and other conditions -- would no longer be protected from huge costs for care by federal caps on out-of-pocket expenditures the Affordable Care Act requires.
- An estimated 21 million who now buy insurance through the Obamacare Marketplaces could lose their coverage.
- Another 12 million on Medicaid could find themselves without insurance.
- At least 2 million young adults ages 26 and under, now on their parents’ health policies, could be kicked off.
- Millions of people who use Medicare could face higher costs.
- Federal subsidies for lower-income Americans to buy policies would disappear.
Throughout the campaign, Biden repeatedly stressed the need to preserve the law’s provision barring insurance companies from refusing coverage for Americans with preexisting conditions, such as diabetes, cancer, and heart disease. It also outlaws charging higher premiums on the basis of health status, age, or gender.
Biden has also pledged to bolster the law as president.
He has proposed a variety of add-ons to the Affordable Care Act he says will “insure more than an estimated 97% of Americans,” according to the Biden campaign site.
Biden’s proposals include offering larger federal subsidies to help low- and middle-income Americans pay for policies purchased through Obamacare insurance Marketplaces.
The boldest of Biden’s proposals is the creation of a “public option” for insurance -- a Medicare-like program that small businesses and individuals could choose if they do not have coverage, cannot afford it, or don’t like their employer-based coverage.
It would also automatically enroll millions of uninsured Americans living in the 14 states that have not expanded Medicaid, which covers low-income people.
But such a plan would require congressional approval -- including a “super majority” of 60 Senate votes to block a likely GOP filibuster. That will be a significant challenge Biden will have to overcome, with Congress so evenly divided.
The White House would also have to defeat heavy lobbying from some of the most influential industry interest groups in Washington, Corlette says.
“I’m not even confident they would get all the Democrat votes,” she says.
“So, it’s a going to be an uphill battle to get a public option passed.”
Taken together, Biden’s plans for expanding Obamacare are projected to cost $750 billion over 10 years. He has said much of that financing would come from increasing taxes on the wealthy.
That means it would likely require congressional approval, which Antos suggests is unlikely given the polarization on Capitol Hill.
Medicare, Medicaid, and drug costs
Biden has called for a host of reforms targeting Medicare, Medicaid, and rising drug costs.
On Medicare, which primarily covers seniors 65 and older, Biden has proposed lowering the eligibility age from 65 to 60. That could extend Medicare to up to 20 million more Americans.
On Medicaid, the health care safety net for low-income and disabled Americans, the president-elect supports increased federal funding to states during the current economic crisis, and potentially beyond.
Medicare is likely to become a key focus of the new administration, in light of the pressures the pandemic is placing on Medicare funding.
In April, Medicare’s trustees said that the Part A trust fund for the program, which pays for hospital and inpatient care, could start to run dry in 2026.
But those projections did not include the impact of COVID-19. Some economists have since projected that Medicare Part A could become insolvent as early as 2022.
Medicare Part B, which pays for doctor and outpatient costs, is funded by general tax funding and beneficiary insurance premiums, so it is not in danger of drying up.
Adding to those pressures is an executive order Trump signed in August temporarily deferring payroll taxes, a primary funding vehicle for Medicare and Social Security.
Under these taxes, employees pay 6.2% of their earnings (on annual income up to $137,700) toward Social Security and 1.45% for Medicare taxes each pay period. Employers pay the same rate per paycheck, adding up to a combined 12.4% Social Security tax and 2.9% Medicare tax.
Biden has said he would reverse the tax cut when he takes office.
But to get a handle on Medicare and Medicaid funding issues, he is likely to need congressional support. Corlette and other experts say that could be a challenge while the nation remains in the grip of the coronavirus pandemic.
In addition to his Medicare and Medicaid reforms, Biden has proposed several plans to lower drug prices, a subset of rising health care and insurance costs.
U.S. spending on prescription drugs has increased nearly 42% over the past decade -- from $253.1 billion in 2010 to $358.7 billion in 2020 (projected) -- according to the Centers for Medicare & Medicaid Services.
In 2020, retail prices for 460 commonly prescribed drugs have spiked an average of 5.2%, according to new analysis by 3 Axis Advisors, a health research firm.
That’s more than double the projected rate of inflation.
To control drug costs, Biden supports legislation approved by the Democratic-led House of Representatives last year that would empower Medicare to negotiate drug prices with drug companies, as private insurers do.
Federal law now bars Medicare from negotiating prices on behalf of the 67.7 million Americans who use it. Drug companies and many GOP leaders argue that the current law is necessary to allow them to spend more on research and development of new medications.
In addition, Biden supports the idea of lifting bans on importing drugs from foreign countries with lower costs.
He also backs creating an independent review board to set price caps for new medications with no competitors; making high-quality generics more available; ending tax breaks for drug company advertising; and limiting their leeway in raising prices.
All of these proposals would likely require congressional approval and could face legal challenges in the courts.
This article first appeared on WebMD.com.
The former vice president has sketched out a big health agenda: ramping up the federal response to COVID-19, boosting the Affordable Care Act, creating a new “public option” to cover uninsured Americans, and expanding Medicare and Medicaid.
But the president-elect’s long to-do list on health is likely to face significant roadblocks in Congress and the courts, experts say.
For instance, Biden’s ambitious proposals on COVID-19 -- including his recent call for a national mask mandate -- could be waylaid by legal challenges and run into political hurdles on Capitol Hill, where he may face a divided Congress.
Joseph Antos, PhD, a health policy expert with the conservative American Enterprise Institute, predicts Biden will encounter the same type of congressional “gridlock situation” that President Barack Obama ran into during his second term.
“We have a situation that has been like this for a very, very long time -- lack of cooperation, lack of recognition that either party is capable of rising above their own electoral views to deal with problems that the country actually has.”
Antos also suggests that Biden may also face enormous political pressure to address the economic fallout from the coronavirus, including record unemployment and business closures, before anything else.
“I think it’s really going to be efforts that are intended to promote economic development and promote the economy,” he says.
In addition, Biden’s plans to expand Obamacare might face a new challenge from the Supreme Court in the year ahead. This month, the high court will take up a new case seeking to overturn the law.
Even so, experts say Biden’s plans on COVID-19 and expanding health care are likely to define his tenure in the White House as a central focus of his presidency.
“Health care will be at the very top of the list of the president’s priorities,” says Sabrina Corlette, JD, co-director of the Center on Health Insurance Reforms at Georgetown University’s McCourt School of Public Policy. “I do think, however, that the administration is going to be very preoccupied with the response to COVID-19 and the economic fallout … particularly in the first year.”
Here’s a closer look at what we can expect from a Biden presidency.
COVID-19: Federalizing response efforts
Biden will move to federalize the response to COVID-19. He has said he will take back major responsibilities from the states -- such as setting national policies on mask wearing, social distancing, and the reopening of schools and businesses, based on CDC guidance. In the days leading up to the election, Biden called for a national mask mandate, after waffling on the issue throughout the summer.
He has said he will let public health science drive political policy. Biden is also planning to create his own task force to advise officials during the transition on managing the new surge in COVID-19 cases, vaccine safety and protecting at-risk populations, Politico reported this week. He received a virtual briefing on the pandemic from a panel of experts as he awaited the election’s outcome.
“I think we will no longer have this confused and contradictory public messaging,” Corlette says, “but I also think there will be humility and the recognition that the evidence is evolving -- that we don’t have all the answers, but we’re learning as we go.”
But national mandates on masks and social distancing will be challenging to enforce, experts say. They are also likely to face pushback from business interests, opposition from public officials in GOP-led states, and even legal challenges.
Biden’s ability to work with Congress -- or not -- may determine whether he is able to implement some of the key components of his coronavirus action plan, which includes:
- Providing free COVID-19 testing for all Americans
- Hiring 100,000 contact tracers
- Eliminating out-of-pocket expenses for coronavirus treatment
- Delivering “sufficient” PPE for essential workers
- Supporting science-backed vaccines and medical treatments being developed
- Requiring the reopening of businesses, workplaces, and schools only after “sufficient” reductions in community transmission -- under evidence-based protocols put forward by the CDC
- Giving emergency paid leave for workers dislocated by the pandemic and more financial aid for workers, families, and small businesses
- Shoring up safeguards to protect at-risk Americans, including older people
- Boosting pay for health care workers on the front lines
Biden has not detailed how he would pay for many of these, beyond promising to force wealthy Americans to “pay their fair share” of taxes to help. He has proposed a tax increase on Americans making more than $400,000 a year, which would require congressional approval.
Antos says he expects Biden’s proposed COVID-19 action plan to be virtually the same as Trump’s in two areas: efforts to develop a vaccine and antiviral treatments.
The administration has spent some $225 million on COVID-19 testing efforts, with a particular focus on rural areas.
Trump launched Operation Warp Speed to fast-track a vaccine. As part of that, the federal government has contracted with six drug companies, spending nearly $11 billion. The operation aims to provide at least 300 million doses of a coronavirus vaccine by January 2021.
Antos would like to see “a more sophisticated approach to social distancing” from the president-elect that takes into account the different challenges facing Americans depending on their income, work situation, and other factors during the pandemic.
“There are a lot of people in this country where working from home is fine and their jobs are secure,” he notes. “It’s the person who used to work at a restaurant that closed, it’s the line worker at a factory that has severely cut back its hours. It’s basically lower-middle-class people, low-income people, middle-class people, and it’s not the elite.
“And the policies have not given enough consideration to the fact that their circumstances and their tradeoffs would differ from the tradeoffs of somebody who doesn’t have anything to worry about economically.
“So, what we need is a more supple policy [that] will give people the information they need and give them the financial support that they also need … so they can make good decisions for themselves and their families. And we basically haven’t done that.”
Obamacare on the blocks?
The Supreme Court’s decision to take up another case seeking to overturn the Affordable Care Act could hand Biden’s health agenda a major setback -- and put the medical care for millions of Americans in jeopardy.
On Nov. 10, the high court will hear oral arguments on a lawsuit that would strike down all of Obamacare. A decision is not expected until next year.
The court has previously upheld the 2010 law, which Biden helped usher through Congress as vice president. But the addition of right-leaning Supreme Court Justice Amy Coney Barrett to the bench last month gives the court a clear conservative majority that could mean the end of Obamacare, legal experts say.
Republicans have opposed the law since its passage, but they have been unable to muster the votes to repeal it, or to pass an alternative
Antos, from the American Enterprise Institute, notes conservatives believe the law has increased costs for health care and insurance over the past decade, in part because of its protections for Americans with preexisting conditions and requiring insurers to provide comprehensive “gold-plated” policies.
“It’s driven up costs, offers plans that are not very strong, put high-risk folks into the same [insurance pool], which has increased costs for everyone, the employer mandate … these are all the reasons,” he says.
The Supreme Court isn’t expected to deliver a decision on the Affordable Care Act before the middle of next year. But the uncertainty will likely push back Biden’s proposals to expand on the law.
Overturning Obamacare would have huge impacts on millions of Americans:
- As many as 133 million Americans -- roughly half the U.S. population -- with preexisting conditions could find it harder, if not impossible, to find affordable health insurance. That figure does not include Americans infected with COVID-19.
- About 165 million who require expensive treatments -- for cancer and other conditions -- would no longer be protected from huge costs for care by federal caps on out-of-pocket expenditures the Affordable Care Act requires.
- An estimated 21 million who now buy insurance through the Obamacare Marketplaces could lose their coverage.
- Another 12 million on Medicaid could find themselves without insurance.
- At least 2 million young adults ages 26 and under, now on their parents’ health policies, could be kicked off.
- Millions of people who use Medicare could face higher costs.
- Federal subsidies for lower-income Americans to buy policies would disappear.
Throughout the campaign, Biden repeatedly stressed the need to preserve the law’s provision barring insurance companies from refusing coverage for Americans with preexisting conditions, such as diabetes, cancer, and heart disease. It also outlaws charging higher premiums on the basis of health status, age, or gender.
Biden has also pledged to bolster the law as president.
He has proposed a variety of add-ons to the Affordable Care Act he says will “insure more than an estimated 97% of Americans,” according to the Biden campaign site.
Biden’s proposals include offering larger federal subsidies to help low- and middle-income Americans pay for policies purchased through Obamacare insurance Marketplaces.
The boldest of Biden’s proposals is the creation of a “public option” for insurance -- a Medicare-like program that small businesses and individuals could choose if they do not have coverage, cannot afford it, or don’t like their employer-based coverage.
It would also automatically enroll millions of uninsured Americans living in the 14 states that have not expanded Medicaid, which covers low-income people.
But such a plan would require congressional approval -- including a “super majority” of 60 Senate votes to block a likely GOP filibuster. That will be a significant challenge Biden will have to overcome, with Congress so evenly divided.
The White House would also have to defeat heavy lobbying from some of the most influential industry interest groups in Washington, Corlette says.
“I’m not even confident they would get all the Democrat votes,” she says.
“So, it’s a going to be an uphill battle to get a public option passed.”
Taken together, Biden’s plans for expanding Obamacare are projected to cost $750 billion over 10 years. He has said much of that financing would come from increasing taxes on the wealthy.
That means it would likely require congressional approval, which Antos suggests is unlikely given the polarization on Capitol Hill.
Medicare, Medicaid, and drug costs
Biden has called for a host of reforms targeting Medicare, Medicaid, and rising drug costs.
On Medicare, which primarily covers seniors 65 and older, Biden has proposed lowering the eligibility age from 65 to 60. That could extend Medicare to up to 20 million more Americans.
On Medicaid, the health care safety net for low-income and disabled Americans, the president-elect supports increased federal funding to states during the current economic crisis, and potentially beyond.
Medicare is likely to become a key focus of the new administration, in light of the pressures the pandemic is placing on Medicare funding.
In April, Medicare’s trustees said that the Part A trust fund for the program, which pays for hospital and inpatient care, could start to run dry in 2026.
But those projections did not include the impact of COVID-19. Some economists have since projected that Medicare Part A could become insolvent as early as 2022.
Medicare Part B, which pays for doctor and outpatient costs, is funded by general tax funding and beneficiary insurance premiums, so it is not in danger of drying up.
Adding to those pressures is an executive order Trump signed in August temporarily deferring payroll taxes, a primary funding vehicle for Medicare and Social Security.
Under these taxes, employees pay 6.2% of their earnings (on annual income up to $137,700) toward Social Security and 1.45% for Medicare taxes each pay period. Employers pay the same rate per paycheck, adding up to a combined 12.4% Social Security tax and 2.9% Medicare tax.
Biden has said he would reverse the tax cut when he takes office.
But to get a handle on Medicare and Medicaid funding issues, he is likely to need congressional support. Corlette and other experts say that could be a challenge while the nation remains in the grip of the coronavirus pandemic.
In addition to his Medicare and Medicaid reforms, Biden has proposed several plans to lower drug prices, a subset of rising health care and insurance costs.
U.S. spending on prescription drugs has increased nearly 42% over the past decade -- from $253.1 billion in 2010 to $358.7 billion in 2020 (projected) -- according to the Centers for Medicare & Medicaid Services.
In 2020, retail prices for 460 commonly prescribed drugs have spiked an average of 5.2%, according to new analysis by 3 Axis Advisors, a health research firm.
That’s more than double the projected rate of inflation.
To control drug costs, Biden supports legislation approved by the Democratic-led House of Representatives last year that would empower Medicare to negotiate drug prices with drug companies, as private insurers do.
Federal law now bars Medicare from negotiating prices on behalf of the 67.7 million Americans who use it. Drug companies and many GOP leaders argue that the current law is necessary to allow them to spend more on research and development of new medications.
In addition, Biden supports the idea of lifting bans on importing drugs from foreign countries with lower costs.
He also backs creating an independent review board to set price caps for new medications with no competitors; making high-quality generics more available; ending tax breaks for drug company advertising; and limiting their leeway in raising prices.
All of these proposals would likely require congressional approval and could face legal challenges in the courts.
This article first appeared on WebMD.com.






