User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Experts call to revise the Uniform Determination of Death Act
, according to an editorial published online Dec. 24, 2019, in Annals of Internal Medicine. Proposed revisions would identify the standards for determining death by neurologic criteria and address the question of whether consent is required to make this determination. If accepted, the revisions would enhance public trust in the determination of death by neurologic criteria, the authors said.
“There is a disconnect between the medical and legal standards for brain death,” said Ariane K. Lewis, MD, associate professor of neurology and neurosurgery at New York University and lead author of the editorial. The discrepancy must be remedied because it has led to lawsuits and has proved to be problematic from a societal standpoint, she added.
“We defend changing the law to match medical practice, rather than changing medical practice to match the law,” said Thaddeus Mason Pope, JD, PhD, director of the Health Law Institute at Mitchell Hamline School of Law in Saint Paul, Minnesota, and an author of the editorial.
Accepted medical standards are unclear
The UDDA was drafted in 1981 to establish a uniform legal standard for death by neurologic criteria. A person with “irreversible cessation of all functions of the entire brain, including the brainstem,” is dead, according to the statute. A determination of death, it adds, “must be made in accordance with accepted medical standards.”
But the medical standards used to determine death by neurologic cause have not been uniform. In 2015, the Supreme Court of Nevada ruled that it was not clear that the standard published by the American Academy of Neurology (AAN), which had been used in the case at issue, was the “accepted medical standard.” An AAN summit later affirmed that the accepted medical standards for determination of death by neurologic cause are the 2010 AAN standard for determination of brain death in adults and the 2011 Society of Critical Care Medicine (SCCM), American Academy of Pediatrics (AAP), and Child Neurology Society (CNS) standard for determination of brain death in children. The Nevada legislature amended the state UDDA to identify these standards as the accepted standards. A revised UDDA also should identify these standards and grant an administrative agency (i.e., the board of medicine) the power to review and update the accepted medical standards as needed, according to the editorial.
To the extent that hospitals are not following the AAN or SCCM/AAP/CNS standards for determining death by neurologic cause, “enshrining” these standards in a revised UDDA “should increase uniformity and consistency” in hospitals’ policies on brain death, Dr. Pope said.
The question of hormonal function
Lawsuits in California and Nevada raised the question of whether the pituitary gland and hypothalamus are parts of the brain. If so, then the accepted medical standards for death by neurologic cause are not consistent with the statutory requirements for the determination of death, since the former do not test for cessation of hormonal function.
The current edition of the adult standards for determining death by neurologic cause were published in 2010. “Whenever we measure brain death, we’re not measuring the cessation of all functions of the entire brain,” Dr. Pope said. “That’s not a new thing; that’s been the case for a long time.”
To address the discrepancy between medical practice and the legal statute, Dr. Lewis and colleagues proposed that the UDDA’s reference to “irreversible cessation of functions of the entire brain” be followed by the following clause: “including the brainstem, leading to unresponsive coma with loss of capacity for consciousness, brainstem areflexia, and the inability to breathe spontaneously.” An alternative revision would be to add the briefer phrase “... with the exception of hormonal function.”
Authors say consent is not required for testing
Other complications have arisen from the UDDA’s failure to specify whether consent is required for a determination of death by neurologic cause. Court rulings on this question have not been consistent. Dr. Lewis and colleagues propose adding the following text to the UDDA: “Reasonable efforts should be made to notify a patient’s legally authorized decision-maker before performing a determination of death by neurologic criteria, but consent is not required to initiate such an evaluation.”
The proposed revisions to the UDDA “might give [clinicians] more confidence to proceed with brain death testing, because it would clarify that they don’t need the parents’ [or the patient’s legally authorized decision-maker] consent to do the tests,” said Dr. Pope. “If anything, they might even have a duty to do the tests.”
The final problem with the UDDA that Dr. Lewis and colleagues cited is that it does not provide clear guidance about how to respond to religious objections to discontinuation of organ support after a determination of death by neurologic cause. “Because the issue is rather complicated, we have not advocated for a singular position related to this [question] in our revised UDDA,” Dr. Lewis said. “Rather, we recommended the need for a multidisciplinary group to come together to determine what is the best approach. In an ideal world, this [approach] would be universal throughout the country.”
Although a revised UDDA would provide greater clarity to physicians and promote uniformity of practice, it would not resolve ongoing theological and philosophical debates about whether brain death is biological death, Dr. Pope said. “The key thing is that it would give clinicians a green light or certainty and clarity that they may proceed to do the test in the first place. If the tests are positive and the patient really is dead, then they could proceed to organ procurement or to move to the morgue.”
Dr. Lewis is a member of various AAN committees and working groups but receives no compensation for her role. A coauthor received personal fees from the AAN that were unrelated to the editorial.
SOURCE: Lewis A et al. Ann Intern Med. 2019 Dec 24. doi: 10.7326/M19-2731.
, according to an editorial published online Dec. 24, 2019, in Annals of Internal Medicine. Proposed revisions would identify the standards for determining death by neurologic criteria and address the question of whether consent is required to make this determination. If accepted, the revisions would enhance public trust in the determination of death by neurologic criteria, the authors said.
“There is a disconnect between the medical and legal standards for brain death,” said Ariane K. Lewis, MD, associate professor of neurology and neurosurgery at New York University and lead author of the editorial. The discrepancy must be remedied because it has led to lawsuits and has proved to be problematic from a societal standpoint, she added.
“We defend changing the law to match medical practice, rather than changing medical practice to match the law,” said Thaddeus Mason Pope, JD, PhD, director of the Health Law Institute at Mitchell Hamline School of Law in Saint Paul, Minnesota, and an author of the editorial.
Accepted medical standards are unclear
The UDDA was drafted in 1981 to establish a uniform legal standard for death by neurologic criteria. A person with “irreversible cessation of all functions of the entire brain, including the brainstem,” is dead, according to the statute. A determination of death, it adds, “must be made in accordance with accepted medical standards.”
But the medical standards used to determine death by neurologic cause have not been uniform. In 2015, the Supreme Court of Nevada ruled that it was not clear that the standard published by the American Academy of Neurology (AAN), which had been used in the case at issue, was the “accepted medical standard.” An AAN summit later affirmed that the accepted medical standards for determination of death by neurologic cause are the 2010 AAN standard for determination of brain death in adults and the 2011 Society of Critical Care Medicine (SCCM), American Academy of Pediatrics (AAP), and Child Neurology Society (CNS) standard for determination of brain death in children. The Nevada legislature amended the state UDDA to identify these standards as the accepted standards. A revised UDDA also should identify these standards and grant an administrative agency (i.e., the board of medicine) the power to review and update the accepted medical standards as needed, according to the editorial.
To the extent that hospitals are not following the AAN or SCCM/AAP/CNS standards for determining death by neurologic cause, “enshrining” these standards in a revised UDDA “should increase uniformity and consistency” in hospitals’ policies on brain death, Dr. Pope said.
The question of hormonal function
Lawsuits in California and Nevada raised the question of whether the pituitary gland and hypothalamus are parts of the brain. If so, then the accepted medical standards for death by neurologic cause are not consistent with the statutory requirements for the determination of death, since the former do not test for cessation of hormonal function.
The current edition of the adult standards for determining death by neurologic cause were published in 2010. “Whenever we measure brain death, we’re not measuring the cessation of all functions of the entire brain,” Dr. Pope said. “That’s not a new thing; that’s been the case for a long time.”
To address the discrepancy between medical practice and the legal statute, Dr. Lewis and colleagues proposed that the UDDA’s reference to “irreversible cessation of functions of the entire brain” be followed by the following clause: “including the brainstem, leading to unresponsive coma with loss of capacity for consciousness, brainstem areflexia, and the inability to breathe spontaneously.” An alternative revision would be to add the briefer phrase “... with the exception of hormonal function.”
Authors say consent is not required for testing
Other complications have arisen from the UDDA’s failure to specify whether consent is required for a determination of death by neurologic cause. Court rulings on this question have not been consistent. Dr. Lewis and colleagues propose adding the following text to the UDDA: “Reasonable efforts should be made to notify a patient’s legally authorized decision-maker before performing a determination of death by neurologic criteria, but consent is not required to initiate such an evaluation.”
The proposed revisions to the UDDA “might give [clinicians] more confidence to proceed with brain death testing, because it would clarify that they don’t need the parents’ [or the patient’s legally authorized decision-maker] consent to do the tests,” said Dr. Pope. “If anything, they might even have a duty to do the tests.”
The final problem with the UDDA that Dr. Lewis and colleagues cited is that it does not provide clear guidance about how to respond to religious objections to discontinuation of organ support after a determination of death by neurologic cause. “Because the issue is rather complicated, we have not advocated for a singular position related to this [question] in our revised UDDA,” Dr. Lewis said. “Rather, we recommended the need for a multidisciplinary group to come together to determine what is the best approach. In an ideal world, this [approach] would be universal throughout the country.”
Although a revised UDDA would provide greater clarity to physicians and promote uniformity of practice, it would not resolve ongoing theological and philosophical debates about whether brain death is biological death, Dr. Pope said. “The key thing is that it would give clinicians a green light or certainty and clarity that they may proceed to do the test in the first place. If the tests are positive and the patient really is dead, then they could proceed to organ procurement or to move to the morgue.”
Dr. Lewis is a member of various AAN committees and working groups but receives no compensation for her role. A coauthor received personal fees from the AAN that were unrelated to the editorial.
SOURCE: Lewis A et al. Ann Intern Med. 2019 Dec 24. doi: 10.7326/M19-2731.
, according to an editorial published online Dec. 24, 2019, in Annals of Internal Medicine. Proposed revisions would identify the standards for determining death by neurologic criteria and address the question of whether consent is required to make this determination. If accepted, the revisions would enhance public trust in the determination of death by neurologic criteria, the authors said.
“There is a disconnect between the medical and legal standards for brain death,” said Ariane K. Lewis, MD, associate professor of neurology and neurosurgery at New York University and lead author of the editorial. The discrepancy must be remedied because it has led to lawsuits and has proved to be problematic from a societal standpoint, she added.
“We defend changing the law to match medical practice, rather than changing medical practice to match the law,” said Thaddeus Mason Pope, JD, PhD, director of the Health Law Institute at Mitchell Hamline School of Law in Saint Paul, Minnesota, and an author of the editorial.
Accepted medical standards are unclear
The UDDA was drafted in 1981 to establish a uniform legal standard for death by neurologic criteria. A person with “irreversible cessation of all functions of the entire brain, including the brainstem,” is dead, according to the statute. A determination of death, it adds, “must be made in accordance with accepted medical standards.”
But the medical standards used to determine death by neurologic cause have not been uniform. In 2015, the Supreme Court of Nevada ruled that it was not clear that the standard published by the American Academy of Neurology (AAN), which had been used in the case at issue, was the “accepted medical standard.” An AAN summit later affirmed that the accepted medical standards for determination of death by neurologic cause are the 2010 AAN standard for determination of brain death in adults and the 2011 Society of Critical Care Medicine (SCCM), American Academy of Pediatrics (AAP), and Child Neurology Society (CNS) standard for determination of brain death in children. The Nevada legislature amended the state UDDA to identify these standards as the accepted standards. A revised UDDA also should identify these standards and grant an administrative agency (i.e., the board of medicine) the power to review and update the accepted medical standards as needed, according to the editorial.
To the extent that hospitals are not following the AAN or SCCM/AAP/CNS standards for determining death by neurologic cause, “enshrining” these standards in a revised UDDA “should increase uniformity and consistency” in hospitals’ policies on brain death, Dr. Pope said.
The question of hormonal function
Lawsuits in California and Nevada raised the question of whether the pituitary gland and hypothalamus are parts of the brain. If so, then the accepted medical standards for death by neurologic cause are not consistent with the statutory requirements for the determination of death, since the former do not test for cessation of hormonal function.
The current edition of the adult standards for determining death by neurologic cause were published in 2010. “Whenever we measure brain death, we’re not measuring the cessation of all functions of the entire brain,” Dr. Pope said. “That’s not a new thing; that’s been the case for a long time.”
To address the discrepancy between medical practice and the legal statute, Dr. Lewis and colleagues proposed that the UDDA’s reference to “irreversible cessation of functions of the entire brain” be followed by the following clause: “including the brainstem, leading to unresponsive coma with loss of capacity for consciousness, brainstem areflexia, and the inability to breathe spontaneously.” An alternative revision would be to add the briefer phrase “... with the exception of hormonal function.”
Authors say consent is not required for testing
Other complications have arisen from the UDDA’s failure to specify whether consent is required for a determination of death by neurologic cause. Court rulings on this question have not been consistent. Dr. Lewis and colleagues propose adding the following text to the UDDA: “Reasonable efforts should be made to notify a patient’s legally authorized decision-maker before performing a determination of death by neurologic criteria, but consent is not required to initiate such an evaluation.”
The proposed revisions to the UDDA “might give [clinicians] more confidence to proceed with brain death testing, because it would clarify that they don’t need the parents’ [or the patient’s legally authorized decision-maker] consent to do the tests,” said Dr. Pope. “If anything, they might even have a duty to do the tests.”
The final problem with the UDDA that Dr. Lewis and colleagues cited is that it does not provide clear guidance about how to respond to religious objections to discontinuation of organ support after a determination of death by neurologic cause. “Because the issue is rather complicated, we have not advocated for a singular position related to this [question] in our revised UDDA,” Dr. Lewis said. “Rather, we recommended the need for a multidisciplinary group to come together to determine what is the best approach. In an ideal world, this [approach] would be universal throughout the country.”
Although a revised UDDA would provide greater clarity to physicians and promote uniformity of practice, it would not resolve ongoing theological and philosophical debates about whether brain death is biological death, Dr. Pope said. “The key thing is that it would give clinicians a green light or certainty and clarity that they may proceed to do the test in the first place. If the tests are positive and the patient really is dead, then they could proceed to organ procurement or to move to the morgue.”
Dr. Lewis is a member of various AAN committees and working groups but receives no compensation for her role. A coauthor received personal fees from the AAN that were unrelated to the editorial.
SOURCE: Lewis A et al. Ann Intern Med. 2019 Dec 24. doi: 10.7326/M19-2731.
FROM ANNALS OF INTERNAL MEDICINE
FDA targets flavored cartridge-based e-cigarettes, but says it is not a ‘ban’
but states it is not a “ban.”
On Jan. 2, the agency issued enforcement guidance alerting companies that manufacture, distribute, and sell unauthorized flavored cartridge-based e-cigarettes within the next 30 days will risk FDA enforcement action.
FDA has had the authority to require premarket authorization of all e-cigarettes and other electronic nicotine delivery systems (ENDS) since August 2016, but thus far has exercised enforcement discretion regarding the need for premarket authorization for these types of products.
“By prioritizing enforcement against the products that are most widely used by children, our action today seeks to strike the right public health balance by maintaining e-cigarettes as a potential off-ramp for adults using combustible tobacco while ensuring these products don’t provide an on-ramp to nicotine addiction for our youth,” Department of Health & Human Services Secretary Alex Azar said in a statement.
The action comes in the wake of more than 2,500 vaping-related injuries being reported, including more than 50 deaths associated with vaping reported by the Centers for Disease Control and Prevention (although many are related to the use of tetrahydrocannabinol [THC] within vaping products) and a continued rise in youth use of e-cigarettes noted in government surveys.
The agency noted in a Jan. 2 statement announcing the enforcement action that, to date, no ENDS products have received a premarket authorization, “meaning that all ENDS products currently on the market are considered illegally marketed and are subject to enforcement, at any time, in the FDA’s discretion.”
FDA said it is prioritizing enforcement in 30 days against:
- Any flavored, cartridge-based ENDS product, other than those with a tobacco or menthol flavoring.
- All other ENDS products for which manufacturers are failing to take adequate measures to prevent access by minors.
- Any ENDS product that is targeted to minors or is likely to promote use by minors.
In the last category, this might include labeling or advertising resembling “kid-friendly food and drinks such as juice boxes or kid-friendly cereal; products marketed directly to minors by promoting ease of concealing the product or disguising it as another product; and products marketed with characters designed to appeal to youth,” according to the FDA statement.
As of May 12, FDA also will prioritize enforcement against any ENDS product for which the manufacturer has not submitted a premarket application. The agency will continue to exercise enforcement discretion for up to 1 year on these products if an application has been submitted, pending the review of that application.
“By not prioritizing enforcement against other flavored ENDS products in the same way as flavored cartridge-based ENDS products, the FDA has attempted to balance the public health concerns related to youth use of ENDS products with consideration regarding addicted adult cigarette smokers who may try to use ENDS products to transition away from combustible tobacco products,” the agency stated, adding that cartridge-based ENDS products are most commonly used among youth.
The FDA statement noted that the enforcement priorities outlined in the guidance document were not a “ban” on flavored or cartridge-based ENDS, noting the agency “has already accepted and begun review of several premarket applications for flavored ENDS products through the pathway that Congress established in the Tobacco Control Act. ... If a company can demonstrate to the FDA that a specific product meets the applicable standard set forth by Congress, including considering how the marketing of the product may affect youth initiation and use, then the FDA could authorize that product for sale.”
“Coupled with the recently signed legislation increasing the minimum age of sale of tobacco to 21, we believe this policy balances the urgency with which we must address the public health threat of youth use of e-cigarette products with the potential role that e-cigarettes may play in helping adult smokers transition completely away from combustible tobacco to a potentially less risky form of nicotine delivery,” FDA Commissioner Stephen Hahn, MD, said in a statement. “While we expect that responsible members of industry will comply with premarket requirements, we’re ready to take action against any unauthorized e-cigarette products as outlined in our priorities. We’ll also closely monitor the use rates of all e-cigarette products and take additional steps to address youth use as necessary.”
The American Medical Association criticized the action as not going far enough, even though it was a step in the right direction.
“The AMA is disappointed that menthol flavors, one of the most popular, will still be allowed, and that flavored e-liquids will remain on the market, leaving young people with easy access to alternative flavored e-cigarette products,” AMA President Patrice A. Harris, MD, said in a statement. “If we are serious about tackling this epidemic and keeping these harmful products out of the hands of young people, a total ban on all flavored e-cigarettes, in all forms and at all locations, is prudent and urgently needed. We are pleased the administration committed today to closely monitoring the situation and trends in e-cigarette use among young people, and to taking further action if needed.”
but states it is not a “ban.”
On Jan. 2, the agency issued enforcement guidance alerting companies that manufacture, distribute, and sell unauthorized flavored cartridge-based e-cigarettes within the next 30 days will risk FDA enforcement action.
FDA has had the authority to require premarket authorization of all e-cigarettes and other electronic nicotine delivery systems (ENDS) since August 2016, but thus far has exercised enforcement discretion regarding the need for premarket authorization for these types of products.
“By prioritizing enforcement against the products that are most widely used by children, our action today seeks to strike the right public health balance by maintaining e-cigarettes as a potential off-ramp for adults using combustible tobacco while ensuring these products don’t provide an on-ramp to nicotine addiction for our youth,” Department of Health & Human Services Secretary Alex Azar said in a statement.
The action comes in the wake of more than 2,500 vaping-related injuries being reported, including more than 50 deaths associated with vaping reported by the Centers for Disease Control and Prevention (although many are related to the use of tetrahydrocannabinol [THC] within vaping products) and a continued rise in youth use of e-cigarettes noted in government surveys.
The agency noted in a Jan. 2 statement announcing the enforcement action that, to date, no ENDS products have received a premarket authorization, “meaning that all ENDS products currently on the market are considered illegally marketed and are subject to enforcement, at any time, in the FDA’s discretion.”
FDA said it is prioritizing enforcement in 30 days against:
- Any flavored, cartridge-based ENDS product, other than those with a tobacco or menthol flavoring.
- All other ENDS products for which manufacturers are failing to take adequate measures to prevent access by minors.
- Any ENDS product that is targeted to minors or is likely to promote use by minors.
In the last category, this might include labeling or advertising resembling “kid-friendly food and drinks such as juice boxes or kid-friendly cereal; products marketed directly to minors by promoting ease of concealing the product or disguising it as another product; and products marketed with characters designed to appeal to youth,” according to the FDA statement.
As of May 12, FDA also will prioritize enforcement against any ENDS product for which the manufacturer has not submitted a premarket application. The agency will continue to exercise enforcement discretion for up to 1 year on these products if an application has been submitted, pending the review of that application.
“By not prioritizing enforcement against other flavored ENDS products in the same way as flavored cartridge-based ENDS products, the FDA has attempted to balance the public health concerns related to youth use of ENDS products with consideration regarding addicted adult cigarette smokers who may try to use ENDS products to transition away from combustible tobacco products,” the agency stated, adding that cartridge-based ENDS products are most commonly used among youth.
The FDA statement noted that the enforcement priorities outlined in the guidance document were not a “ban” on flavored or cartridge-based ENDS, noting the agency “has already accepted and begun review of several premarket applications for flavored ENDS products through the pathway that Congress established in the Tobacco Control Act. ... If a company can demonstrate to the FDA that a specific product meets the applicable standard set forth by Congress, including considering how the marketing of the product may affect youth initiation and use, then the FDA could authorize that product for sale.”
“Coupled with the recently signed legislation increasing the minimum age of sale of tobacco to 21, we believe this policy balances the urgency with which we must address the public health threat of youth use of e-cigarette products with the potential role that e-cigarettes may play in helping adult smokers transition completely away from combustible tobacco to a potentially less risky form of nicotine delivery,” FDA Commissioner Stephen Hahn, MD, said in a statement. “While we expect that responsible members of industry will comply with premarket requirements, we’re ready to take action against any unauthorized e-cigarette products as outlined in our priorities. We’ll also closely monitor the use rates of all e-cigarette products and take additional steps to address youth use as necessary.”
The American Medical Association criticized the action as not going far enough, even though it was a step in the right direction.
“The AMA is disappointed that menthol flavors, one of the most popular, will still be allowed, and that flavored e-liquids will remain on the market, leaving young people with easy access to alternative flavored e-cigarette products,” AMA President Patrice A. Harris, MD, said in a statement. “If we are serious about tackling this epidemic and keeping these harmful products out of the hands of young people, a total ban on all flavored e-cigarettes, in all forms and at all locations, is prudent and urgently needed. We are pleased the administration committed today to closely monitoring the situation and trends in e-cigarette use among young people, and to taking further action if needed.”
but states it is not a “ban.”
On Jan. 2, the agency issued enforcement guidance alerting companies that manufacture, distribute, and sell unauthorized flavored cartridge-based e-cigarettes within the next 30 days will risk FDA enforcement action.
FDA has had the authority to require premarket authorization of all e-cigarettes and other electronic nicotine delivery systems (ENDS) since August 2016, but thus far has exercised enforcement discretion regarding the need for premarket authorization for these types of products.
“By prioritizing enforcement against the products that are most widely used by children, our action today seeks to strike the right public health balance by maintaining e-cigarettes as a potential off-ramp for adults using combustible tobacco while ensuring these products don’t provide an on-ramp to nicotine addiction for our youth,” Department of Health & Human Services Secretary Alex Azar said in a statement.
The action comes in the wake of more than 2,500 vaping-related injuries being reported, including more than 50 deaths associated with vaping reported by the Centers for Disease Control and Prevention (although many are related to the use of tetrahydrocannabinol [THC] within vaping products) and a continued rise in youth use of e-cigarettes noted in government surveys.
The agency noted in a Jan. 2 statement announcing the enforcement action that, to date, no ENDS products have received a premarket authorization, “meaning that all ENDS products currently on the market are considered illegally marketed and are subject to enforcement, at any time, in the FDA’s discretion.”
FDA said it is prioritizing enforcement in 30 days against:
- Any flavored, cartridge-based ENDS product, other than those with a tobacco or menthol flavoring.
- All other ENDS products for which manufacturers are failing to take adequate measures to prevent access by minors.
- Any ENDS product that is targeted to minors or is likely to promote use by minors.
In the last category, this might include labeling or advertising resembling “kid-friendly food and drinks such as juice boxes or kid-friendly cereal; products marketed directly to minors by promoting ease of concealing the product or disguising it as another product; and products marketed with characters designed to appeal to youth,” according to the FDA statement.
As of May 12, FDA also will prioritize enforcement against any ENDS product for which the manufacturer has not submitted a premarket application. The agency will continue to exercise enforcement discretion for up to 1 year on these products if an application has been submitted, pending the review of that application.
“By not prioritizing enforcement against other flavored ENDS products in the same way as flavored cartridge-based ENDS products, the FDA has attempted to balance the public health concerns related to youth use of ENDS products with consideration regarding addicted adult cigarette smokers who may try to use ENDS products to transition away from combustible tobacco products,” the agency stated, adding that cartridge-based ENDS products are most commonly used among youth.
The FDA statement noted that the enforcement priorities outlined in the guidance document were not a “ban” on flavored or cartridge-based ENDS, noting the agency “has already accepted and begun review of several premarket applications for flavored ENDS products through the pathway that Congress established in the Tobacco Control Act. ... If a company can demonstrate to the FDA that a specific product meets the applicable standard set forth by Congress, including considering how the marketing of the product may affect youth initiation and use, then the FDA could authorize that product for sale.”
“Coupled with the recently signed legislation increasing the minimum age of sale of tobacco to 21, we believe this policy balances the urgency with which we must address the public health threat of youth use of e-cigarette products with the potential role that e-cigarettes may play in helping adult smokers transition completely away from combustible tobacco to a potentially less risky form of nicotine delivery,” FDA Commissioner Stephen Hahn, MD, said in a statement. “While we expect that responsible members of industry will comply with premarket requirements, we’re ready to take action against any unauthorized e-cigarette products as outlined in our priorities. We’ll also closely monitor the use rates of all e-cigarette products and take additional steps to address youth use as necessary.”
The American Medical Association criticized the action as not going far enough, even though it was a step in the right direction.
“The AMA is disappointed that menthol flavors, one of the most popular, will still be allowed, and that flavored e-liquids will remain on the market, leaving young people with easy access to alternative flavored e-cigarette products,” AMA President Patrice A. Harris, MD, said in a statement. “If we are serious about tackling this epidemic and keeping these harmful products out of the hands of young people, a total ban on all flavored e-cigarettes, in all forms and at all locations, is prudent and urgently needed. We are pleased the administration committed today to closely monitoring the situation and trends in e-cigarette use among young people, and to taking further action if needed.”
QoL good for MGMT-methylated glioblastoma patients with lomustine-temozolide
In addition to offering an overall survival benefit for patients with MGMT-methylated glioblastoma, the combination of lomustine and temozolomide did not impair health-related quality of life (HRQOL) compared with temozolomide alone, investigators report.
Among 129 patients with newly-diagnosed glioblastoma with methylation of the MGMT promoter, there were no significant differences in any items on the European Organisation for Research and Treatment of Cancer (EORTC) quality of life questionnaire core-30 and the EORTC brain cancer module (BN20) between patients who received oral combined lomustine and temozolomide or temozolomide alone, reported Johannes Weller, MD, of University Hospital Bonn, Germany, and colleagues.
Although the combination was associated with slightly lower scores on the Mini-Mental State Exam (MMSE), the differences were not clinically significant, the investigators asserted.
“The absence of systematic and clinically relevant changes in HRQOL and neurocognitive function combined with the survival benefit of lomustine-temozolomide versus temozolomide alone suggests that a long-term net clinical benefit exists for patients with newly diagnosed glioblastoma with methylation of the MGMT promoter and supports the use of lomustine-temozolomide as a treatment option for these patients,” they wrote. The report is in The Lancet Oncology.
The investigators previously reported that median overall survival was improved from 31.4 months with temozolomide to 48.1 months with lomustine-temozolomide, translating into a hazard ratio (HR) for death with the combination of 0.60 (P = .0492).
In the current report, Dr. Weller and associates looked at the secondary endpoints of HRQOL as measured by the EORTC scales, and at neurocognitive function as assessed by the MMSE and a neurocognitive test battery (NOA-07) that include Trail Making Test A and B (TMT-A and B), working memory tests, and tests for word and semantic verbal fluency.
The modified intention-to-treat analysis included all patients who received at least one dose of study chemotherapy. The analysis included data on 63 patients randomly assigned to receive standard oral temozolomide, consisting of 75 mg/m² daily during radiotherapy plus six 4-week courses of temozolomide at doses ranging from 150 to 200 mg/m² on days 1-5, every 4 weeks; and 66 patients assigned to receive oral combined lomustine consisting of a 100 mg/m² dose on day 1, plus temozolomide 100 to 200 mg/m² on days 2-6 for six cycles of 6 weeks each.
After a median follow-up of 19.4 months for the HRQOL endpoint, there were no significant differences between the groups in decline from baselines in Karnofsky Performance Score, global health, physical functioning, cognitive functioning, social functioning, or communication deficit.
As noted before, however, there were small but significant differences between the groups favoring temozolomide on the MMSE, after a median follow-up for this measure of 15.3 months. The authors noted that the differences “were not significant when adjusted for multiple testing and were also not clinically relevant, because even over the time course of 4 years the differences between the groups would only add up to 1.76/30 points and clinically significant results would require a difference of more than 3/30 points.”
There were also no significant differences between the groups in any item of the neurocognitive test, they added.
The investigators acknowledged that the trial was limited by its relatively small size, and that after 3.5 years of follow-up about half of all the expected HRQOL forms were missing, which might lead to reporting bias.
“Overall, we conclude that the addition of lomustine to temozolomide in patients with newly diagnosed MGMT-methylated glioblastoma is associated with a clear long-term net clinical benefit and our data provide a good rationale for the trial regimen as a treatment option for these patients. Nevertheless, changes in HRQOL during the first year after beginning treatment needs further exploration in future studies,” Dr. Weller and colleagues wrote.
The German Federal Ministry of Education and Research funded the study. Dr. Weller reported having no conflict of interest. Several coauthors reported relationships with industry outside the submitted work.
SOURCE: Weller J et al. Lancet Oncol. Sept 2. doi: 10.1016/S1470-2045(19)30502-9.
In addition to offering an overall survival benefit for patients with MGMT-methylated glioblastoma, the combination of lomustine and temozolomide did not impair health-related quality of life (HRQOL) compared with temozolomide alone, investigators report.
Among 129 patients with newly-diagnosed glioblastoma with methylation of the MGMT promoter, there were no significant differences in any items on the European Organisation for Research and Treatment of Cancer (EORTC) quality of life questionnaire core-30 and the EORTC brain cancer module (BN20) between patients who received oral combined lomustine and temozolomide or temozolomide alone, reported Johannes Weller, MD, of University Hospital Bonn, Germany, and colleagues.
Although the combination was associated with slightly lower scores on the Mini-Mental State Exam (MMSE), the differences were not clinically significant, the investigators asserted.
“The absence of systematic and clinically relevant changes in HRQOL and neurocognitive function combined with the survival benefit of lomustine-temozolomide versus temozolomide alone suggests that a long-term net clinical benefit exists for patients with newly diagnosed glioblastoma with methylation of the MGMT promoter and supports the use of lomustine-temozolomide as a treatment option for these patients,” they wrote. The report is in The Lancet Oncology.
The investigators previously reported that median overall survival was improved from 31.4 months with temozolomide to 48.1 months with lomustine-temozolomide, translating into a hazard ratio (HR) for death with the combination of 0.60 (P = .0492).
In the current report, Dr. Weller and associates looked at the secondary endpoints of HRQOL as measured by the EORTC scales, and at neurocognitive function as assessed by the MMSE and a neurocognitive test battery (NOA-07) that include Trail Making Test A and B (TMT-A and B), working memory tests, and tests for word and semantic verbal fluency.
The modified intention-to-treat analysis included all patients who received at least one dose of study chemotherapy. The analysis included data on 63 patients randomly assigned to receive standard oral temozolomide, consisting of 75 mg/m² daily during radiotherapy plus six 4-week courses of temozolomide at doses ranging from 150 to 200 mg/m² on days 1-5, every 4 weeks; and 66 patients assigned to receive oral combined lomustine consisting of a 100 mg/m² dose on day 1, plus temozolomide 100 to 200 mg/m² on days 2-6 for six cycles of 6 weeks each.
After a median follow-up of 19.4 months for the HRQOL endpoint, there were no significant differences between the groups in decline from baselines in Karnofsky Performance Score, global health, physical functioning, cognitive functioning, social functioning, or communication deficit.
As noted before, however, there were small but significant differences between the groups favoring temozolomide on the MMSE, after a median follow-up for this measure of 15.3 months. The authors noted that the differences “were not significant when adjusted for multiple testing and were also not clinically relevant, because even over the time course of 4 years the differences between the groups would only add up to 1.76/30 points and clinically significant results would require a difference of more than 3/30 points.”
There were also no significant differences between the groups in any item of the neurocognitive test, they added.
The investigators acknowledged that the trial was limited by its relatively small size, and that after 3.5 years of follow-up about half of all the expected HRQOL forms were missing, which might lead to reporting bias.
“Overall, we conclude that the addition of lomustine to temozolomide in patients with newly diagnosed MGMT-methylated glioblastoma is associated with a clear long-term net clinical benefit and our data provide a good rationale for the trial regimen as a treatment option for these patients. Nevertheless, changes in HRQOL during the first year after beginning treatment needs further exploration in future studies,” Dr. Weller and colleagues wrote.
The German Federal Ministry of Education and Research funded the study. Dr. Weller reported having no conflict of interest. Several coauthors reported relationships with industry outside the submitted work.
SOURCE: Weller J et al. Lancet Oncol. Sept 2. doi: 10.1016/S1470-2045(19)30502-9.
In addition to offering an overall survival benefit for patients with MGMT-methylated glioblastoma, the combination of lomustine and temozolomide did not impair health-related quality of life (HRQOL) compared with temozolomide alone, investigators report.
Among 129 patients with newly-diagnosed glioblastoma with methylation of the MGMT promoter, there were no significant differences in any items on the European Organisation for Research and Treatment of Cancer (EORTC) quality of life questionnaire core-30 and the EORTC brain cancer module (BN20) between patients who received oral combined lomustine and temozolomide or temozolomide alone, reported Johannes Weller, MD, of University Hospital Bonn, Germany, and colleagues.
Although the combination was associated with slightly lower scores on the Mini-Mental State Exam (MMSE), the differences were not clinically significant, the investigators asserted.
“The absence of systematic and clinically relevant changes in HRQOL and neurocognitive function combined with the survival benefit of lomustine-temozolomide versus temozolomide alone suggests that a long-term net clinical benefit exists for patients with newly diagnosed glioblastoma with methylation of the MGMT promoter and supports the use of lomustine-temozolomide as a treatment option for these patients,” they wrote. The report is in The Lancet Oncology.
The investigators previously reported that median overall survival was improved from 31.4 months with temozolomide to 48.1 months with lomustine-temozolomide, translating into a hazard ratio (HR) for death with the combination of 0.60 (P = .0492).
In the current report, Dr. Weller and associates looked at the secondary endpoints of HRQOL as measured by the EORTC scales, and at neurocognitive function as assessed by the MMSE and a neurocognitive test battery (NOA-07) that include Trail Making Test A and B (TMT-A and B), working memory tests, and tests for word and semantic verbal fluency.
The modified intention-to-treat analysis included all patients who received at least one dose of study chemotherapy. The analysis included data on 63 patients randomly assigned to receive standard oral temozolomide, consisting of 75 mg/m² daily during radiotherapy plus six 4-week courses of temozolomide at doses ranging from 150 to 200 mg/m² on days 1-5, every 4 weeks; and 66 patients assigned to receive oral combined lomustine consisting of a 100 mg/m² dose on day 1, plus temozolomide 100 to 200 mg/m² on days 2-6 for six cycles of 6 weeks each.
After a median follow-up of 19.4 months for the HRQOL endpoint, there were no significant differences between the groups in decline from baselines in Karnofsky Performance Score, global health, physical functioning, cognitive functioning, social functioning, or communication deficit.
As noted before, however, there were small but significant differences between the groups favoring temozolomide on the MMSE, after a median follow-up for this measure of 15.3 months. The authors noted that the differences “were not significant when adjusted for multiple testing and were also not clinically relevant, because even over the time course of 4 years the differences between the groups would only add up to 1.76/30 points and clinically significant results would require a difference of more than 3/30 points.”
There were also no significant differences between the groups in any item of the neurocognitive test, they added.
The investigators acknowledged that the trial was limited by its relatively small size, and that after 3.5 years of follow-up about half of all the expected HRQOL forms were missing, which might lead to reporting bias.
“Overall, we conclude that the addition of lomustine to temozolomide in patients with newly diagnosed MGMT-methylated glioblastoma is associated with a clear long-term net clinical benefit and our data provide a good rationale for the trial regimen as a treatment option for these patients. Nevertheless, changes in HRQOL during the first year after beginning treatment needs further exploration in future studies,” Dr. Weller and colleagues wrote.
The German Federal Ministry of Education and Research funded the study. Dr. Weller reported having no conflict of interest. Several coauthors reported relationships with industry outside the submitted work.
SOURCE: Weller J et al. Lancet Oncol. Sept 2. doi: 10.1016/S1470-2045(19)30502-9.
FROM THE LANCET ONCOLOGY
Top research findings of 2018-2019 for clinical practice
Medical knowledge is growing faster than ever, as is the challenge of keeping up with this ever-growing body of information. Clinicians need a system or method to help them sort and evaluate the quality of new information before they can apply it to clinical care. Without such a system, when facing an overload of information, most of us tend to take the first or the most easily accessed information, without considering the quality of such information. As a result, the use of poor-quality information affects the quality and outcome of care we provide, and costs billions of dollars annually in problems associated with underuse, overuse, and misuse of treatments.
In an effort to sort and evaluate recently published research that is ready for clinical use, the first author (SAS) used the following 3-step methodology:
1. Searched literature for research findings suggesting readiness for clinical utilization published between July 1, 2018 and June 30, 2019.
2. Surveyed members of the American Association of Chairs of Departments of Psychiatry, the American Association of Community Psychiatrists, the American Association of Psychiatric Administrators, the North Carolina Psychiatric Association, the Group for the Advancement of Psychiatry, and many other colleagues by asking them: “Among the articles published from July 1, 2018 to June 30, 2019, which ones in your opinion have (or are likely to have or should have) affected/changed the clinical practice of psychiatry?”
3. Looked for appraisals in post-publication reviews such as NEJM Journal Watch, F1000 Prime, Evidence-Based Mental Health, commentaries in peer-reviewed journals, and other sources (see Related Resources).
We chose 12 articles based on their clinical relevance/applicability. Here in Part 1 we present brief descriptions of the 6 of top 12 papers chosen by this methodology; these studies are summarized in the Table.1-6 The order in which they appear in this article is arbitrary. The remaining 6 studies will be reviewed in Part 2 in the February 2020 issue of
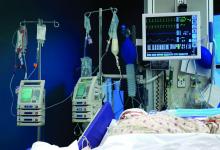
1. Ray WA, Stein CM, Murray KT, et al. Association of antipsychotic treatment with risk of unexpected death among children and youths. JAMA Psychiatry. 2019;76(2):162-171.
Children and young adults are increasingly being prescribed antipsychotic medications. Studies have suggested that when these medications are used in adults and older patients, they are associated with an increased risk of death.7-9 Whether or not these medications are associated with an increased risk of death in children and youth has been unknown. Ray et al1 compared the risk of unexpected death among children and youths who were beginning treatment with an antipsychotic or control medications.
Study design
- This retrospective cohort study evaluated children and young adults age 5 to 24 who were enrolled in Medicaid in Tennessee between 1999 and 2014.
- New antipsychotic use at both a higher dose (>50 mg chlorpromazine equivalents) and a lower dose (≤50 mg chlorpromazine equivalents) was compared with new use of a control medication, including attention-deficit/hyperactivity disorder medications, antidepressants, and mood stabilizers.
- There were 189,361 participants in the control group, 28,377 participants in the lower-dose antipsychotic group, and 30,120 participants in the higher-dose antipsychotic group.
Outcomes
- The primary outcome was death due to injury or suicide or unexpected death occurring during study follow-up.
- The incidence of death in the higher-dose antipsychotic group (146.2 per 100,000 person-years) was significantly higher (P < .001) than the incidence of death in the control medications group (54.5 per 100,000 person years).
- There was no similar significant difference between the lower-dose antipsychotic group and the control medications group.
Continue to: Conclusion
Conclusion
- Higher-dose antipsychotic use is associated with increased rates of unexpected deaths in children and young adults.
- As with all association studies, no direct line connected cause and effect. However, these results reinforce recommendations for careful prescribing and monitoring of antipsychotic regimens for children and youths, and the need for larger antipsychotic safety studies in this population.
- Examining risks associated with specific antipsychotics will require larger datasets, but will be critical for our understanding of the risks and benefits.
2. Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893-903.
Controlled studies have shown esketamine has efficacy for treatment-resistant depression (TRD), but these studies have been only short-term, and the long-term effects of esketamine for TRD have not been established. To fill that gap, Daly et al2 assessed the efficacy of esketamine nasal spray plus an oral antidepressant vs a placebo nasal spray plus an oral antidepressant in delaying relapse of depressive symptoms in patients with TRD. All patients were in stable remission after an optimization course of esketamine nasal spray plus an oral antidepressant.
Study design
- Between October 2015 and February 2018, researchers conducted a phase III, multicenter, double-blind, randomized withdrawal study to evaluate the effect of continuation of esketamine on rates of relapse in patients with TRD who had responded to initial treatment with esketamine.
- Initially, 705 adults were enrolled. Of these participants, 455 proceeded to the optimization phase, in which they were treated with esketamine nasal spray plus an oral antidepressant.
- After 16 weeks of optimization treatment, 297 participants achieved remission or stable response and were randomized to a treatment group, which received continued esketamine nasal spray plus an oral antidepressant, or to a control group, which received a placebo nasal spray plus an oral antidepressant.
Outcomes
- Treatment with esketamine nasal spray and an oral antidepressant was associated with decreased rates of relapse compared with treatment with placebo nasal spray and an oral antidepressant. This was the case among patients who had achieved remission as well as those who had achieved stable response.
- Continued treatment with esketamine decreased the risk of relapse by 51%, with 40 participants in the treatment group experiencing relapse compared with 73 participants in the placebo group.
Continue to: Conclusion
Conclusion
- In patients with TRD who responded to initial treatment with esketamine, continuing esketamine plus an oral antidepressant resulted in clinically meaningful superiority in preventing relapse compared with a placebo nasal spray plus an oral antidepressant.
3. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
Many studies have documented the efficacy of ketamine as a rapid-onset antidepressant. Studies investigating the mechanism of this effect have focused on antagonism of N-methyl-
Study design
- This double-blind crossover study evaluated if opioid receptor activation is necessary for ketamine to have an antidepressant effect in patients with TRD.
- Twelve participants completed both sides of the study in a randomized order. Participants received placebo or naltrexone prior to an IV infusion of ketamine.
- Researchers measured patients’ scores on the Hamilton Depression Rating Scale (HAM-D) at baseline and 1 day after infusion. Response was defined as a ≥50% reduction in HAM-D score.
Outcomes
- Reductions in HAM-D scores among participants in the ketamine plus naltrexone group were significantly lower than those of participants in the ketamine plus placebo group.
- Dissociation related to ketamine use did not differ significantly between the naltrexone group and the placebo group.
Continue to: Conclusion
Conclusion
- This small study found a significant decrease in the antidepressant effect of ketamine infusion in patients with TRD when opioid receptors are blocked with naltrexone prior to infusion, which suggests opioid receptor activation is necessary for ketamine to be effective as an antidepressant.
- This appears to be consistent with observations of buprenorphine’s antidepressant effects. Caution is indicated until additional studies can further elucidate the mechanism of action of ketamine’s antidepressant effects (see "Ketamine/esketamine: Putative mechanism of action," page 32).
4. Nidich S, Mills PJ, Rainforth M, et al. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: a randomised controlled trial. Lancet Psychiatry. 2018;5(12):975-986.
Posttraumatic stress disorder (PTSD) is a common and important public health problem. Evidence-based treatments for PTSD include trauma-focused therapies such as prolonged exposure therapy (PE). However, some patients may not respond to PE, drop out, or elect not to pursue it. Researchers continue to explore treatments that are non-trauma-focused, such as mindfulness meditation and interpersonal psychotherapy. In a 3-group comparative effectiveness trial, Nidich et al4 examined the efficacy of a non-trauma-focused intervention, transcendental meditation (TM), in reducing PTSD symptom severity and depression in veterans.
Study design
- Researchers recruited 203 veterans with PTSD from the Department of Veterans Affairs (VA) San Diego Healthcare System between June 2013 and October 2016.
- Participants were randomly assigned to 1 of 3 groups: 68 to TM, 68 to PE, and 67 to PTSD health education (HE).
- Each group received 12 sessions over 12 weeks. In addition to group and individual sessions, all participants received daily practice or assignments.
- The Clinician-Administered PTSD Scale (CAPS) was used to assess symptoms before and after treatment.
Outcomes
- The primary outcome assessed was change in PTSD symptom severity at the end of the study compared with baseline as measured by change in CAPS score.
- Transcendental meditation was found to be significantly non-inferior to PE, with a mean change in CAPS score of −16.1 in the TM group and −11.2 in the PE group.
- Both the TM and PE groups also had significant reductions in CAPS scores compared with the HE group, which had a mean change in CAPS score of −2.5.
Continue to: Conclusion
Conclusion
- Transcendental meditation is significantly not inferior to PE in the treatment of veterans with PTSD.
- The findings from this first comparative effectiveness trial comparing TM with an established psychotherapy for PTSD suggests the feasibility and efficacy of TM as an alternative therapy for veterans with PTSD.
- Because TM is self-administered after an initial expert training, it may offer an easy-to-implement approach that may be more accessible to veterans than other treatments.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
Several smaller randomized trials of prazosin involving a total of 283 active-duty service members, veterans, and civilian participants have shown efficacy of prazosin for PTSD-related nightmares, sleep disturbance, and overall clinical functioning. However, in a recent trial, Raskind et al5 failed to demonstrate such efficacy.
Study design
- Veterans with chronic PTSD nightmares were recruited from 13 VA medical centers to participate in a 26-week, double-blind, randomized controlled trial.
- A total of 304 participants were randomized to a prazosin treatment group (n = 152) or a placebo control group (n = 152).
- During the first 10 weeks, prazosin or placebo were administered in an escalating fashion up to a maximum dose.
- The CAPS, Pittsburgh Sleep Quality Index (PSQI), and Clinical Global Impressions of Change (CGIC) scores were measured at baseline, after 10 weeks, and after 26 weeks.
Outcomes
- Three primary outcomes measures were assessed: change in score from baseline to 10 weeks on CAPS item B2, the PSQI, and the CGIC.
- A secondary measure was change in score from baseline of the same measures at 26 weeks.
- There was no significant difference between the prazosin group and the placebo group in any of the primary or secondary measures.
Continue to: Conclusion
Conclusion
- Compared with placebo, prazosin was not associated with improvement in nightmares or sleep quality for veterans with chronic PTSD nightmares.
- Because psychosocial instability was an exclusion criterion, it is possible that a selection bias resulting from recruitment of patients who were mainly in clinically stable condition accounted for these negative results, since symptoms in such patients were less likely to be ameliorated with antiadrenergic treatment.
Treatment-resistant depression in veterans is a major clinical challenge because of these patients’ increased risk of suicide. Repetitive transcranial magnetic stimulation (rTMS) has shown promising results for TRD. In a randomized trial, Yesavage et al6 compared rTMS vs sham rTMS in veterans with TRD.
Study design
- Veterans with TRD were recruited from 9 VA medical centers throughout the United States between September 2012 and May 2016.
- Researchers randomized 164 participants into 1 of 2 groups in a double-blind fashion. The treatment group (n = 81) received left prefrontal rTMS, and the control group (n = 83) received sham rTMS.
Outcomes
- In an intention-to-treat analysis, remission rate (defined as a HAM-D score of ≤10) was assessed as the primary outcome measure.
- Remission was seen in both groups, with 40.7% of the treatment group achieving remission and 37.4% of the control group achieving remission. However, the difference between the 2 groups was not significant (P = .67), with an odds ratio of 1.16.
Continue to: Conclusion
Conclusion
- In this study, treatment with rTMS did not show a statistically significant difference in rates of remission from TRD in veterans compared with sham rTMS. This differs from previous rTMS trials in non-veteran patients.
- The findings of this study also differed from those of other rTMS research in terms of the high remission rates that were seen in both the active and sham groups.
Bottom Line
The risk of death might be increased in children and young adults who receive highdose antipsychotics. Continued treatment with intranasal esketamine may help prevent relapse in patients with treatment-resistant depression (TRD) who initially respond to esketamine. The antidepressant effects of ketamine might be associated with opioid receptor activation. Transcendental meditation may be helpful for patients with posttraumatic stress disorder (PTSD), while prazosin might not improve nightmares or sleep quality in patients with PTSD. Repetitive transcranial magnetic stimulation (rTMS) might not be any more effective than sham rTMS for veterans with TRD.
Related Resources
- NEJM Journal Watch. www.jwatch.org.
- F1000 Prime. https://f1000.com/prime/home.
- BMJ Journals Evidence-Based Mental Health. https://ebmh.bmj.com.
Drug Brand Names
Buprenorphine • Subutex
Chlorpromazine • Thorazine
Esketamine nasal spray • Spravato
Ketamine • Ketalar
Naltrexone • Narcan
Prazosin • Minipress
1. Ray WA, Stein CM, Murray KT, et al. Association of antipsychotic treatment with risk of unexpected death among children and youths. JAMA Psychiatry. 2019;76(2):162-171.
2. Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893-903.
3. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
4. Nidich S, Mills PJ, Rainforth M, et al. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: a randomized controlled trial. Lancet Psychiatry. 2018;5(12):975-986.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
6. Yesavage JA, Fairchild JK, Mi Z, et al. Effect of repetitive transcranial magnetic stimulation on treatment-resistant major depression in US veterans: a randomized clinical trial. JAMA Psychiatry. 2018;75(9):884-893.
7. Ray WA, Meredith S, Thapa PB, et al. Antipsychotics and the risk of sudden cardiac death. Arch Gen Psychiatry. 2001;58(12):1161-1167.
8. Ray WA, Chung CP, Murray KT, Hall K, Stein CM. Atypical antipsychotic drugs and the risk of sudden cardiac death. N Engl J Med. 2009;360(3):225-235.
9. Jeste DV, Blazer D, Casey D, et al. ACNP White Paper: update on use of antipsychotic drugs in elderly persons with dementia. Neuropsychopharmacology. 2008;33(5):957-970.
Medical knowledge is growing faster than ever, as is the challenge of keeping up with this ever-growing body of information. Clinicians need a system or method to help them sort and evaluate the quality of new information before they can apply it to clinical care. Without such a system, when facing an overload of information, most of us tend to take the first or the most easily accessed information, without considering the quality of such information. As a result, the use of poor-quality information affects the quality and outcome of care we provide, and costs billions of dollars annually in problems associated with underuse, overuse, and misuse of treatments.
In an effort to sort and evaluate recently published research that is ready for clinical use, the first author (SAS) used the following 3-step methodology:
1. Searched literature for research findings suggesting readiness for clinical utilization published between July 1, 2018 and June 30, 2019.
2. Surveyed members of the American Association of Chairs of Departments of Psychiatry, the American Association of Community Psychiatrists, the American Association of Psychiatric Administrators, the North Carolina Psychiatric Association, the Group for the Advancement of Psychiatry, and many other colleagues by asking them: “Among the articles published from July 1, 2018 to June 30, 2019, which ones in your opinion have (or are likely to have or should have) affected/changed the clinical practice of psychiatry?”
3. Looked for appraisals in post-publication reviews such as NEJM Journal Watch, F1000 Prime, Evidence-Based Mental Health, commentaries in peer-reviewed journals, and other sources (see Related Resources).
We chose 12 articles based on their clinical relevance/applicability. Here in Part 1 we present brief descriptions of the 6 of top 12 papers chosen by this methodology; these studies are summarized in the Table.1-6 The order in which they appear in this article is arbitrary. The remaining 6 studies will be reviewed in Part 2 in the February 2020 issue of

1. Ray WA, Stein CM, Murray KT, et al. Association of antipsychotic treatment with risk of unexpected death among children and youths. JAMA Psychiatry. 2019;76(2):162-171.
Children and young adults are increasingly being prescribed antipsychotic medications. Studies have suggested that when these medications are used in adults and older patients, they are associated with an increased risk of death.7-9 Whether or not these medications are associated with an increased risk of death in children and youth has been unknown. Ray et al1 compared the risk of unexpected death among children and youths who were beginning treatment with an antipsychotic or control medications.
Study design
- This retrospective cohort study evaluated children and young adults age 5 to 24 who were enrolled in Medicaid in Tennessee between 1999 and 2014.
- New antipsychotic use at both a higher dose (>50 mg chlorpromazine equivalents) and a lower dose (≤50 mg chlorpromazine equivalents) was compared with new use of a control medication, including attention-deficit/hyperactivity disorder medications, antidepressants, and mood stabilizers.
- There were 189,361 participants in the control group, 28,377 participants in the lower-dose antipsychotic group, and 30,120 participants in the higher-dose antipsychotic group.
Outcomes
- The primary outcome was death due to injury or suicide or unexpected death occurring during study follow-up.
- The incidence of death in the higher-dose antipsychotic group (146.2 per 100,000 person-years) was significantly higher (P < .001) than the incidence of death in the control medications group (54.5 per 100,000 person years).
- There was no similar significant difference between the lower-dose antipsychotic group and the control medications group.
Continue to: Conclusion
Conclusion
- Higher-dose antipsychotic use is associated with increased rates of unexpected deaths in children and young adults.
- As with all association studies, no direct line connected cause and effect. However, these results reinforce recommendations for careful prescribing and monitoring of antipsychotic regimens for children and youths, and the need for larger antipsychotic safety studies in this population.
- Examining risks associated with specific antipsychotics will require larger datasets, but will be critical for our understanding of the risks and benefits.
2. Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893-903.
Controlled studies have shown esketamine has efficacy for treatment-resistant depression (TRD), but these studies have been only short-term, and the long-term effects of esketamine for TRD have not been established. To fill that gap, Daly et al2 assessed the efficacy of esketamine nasal spray plus an oral antidepressant vs a placebo nasal spray plus an oral antidepressant in delaying relapse of depressive symptoms in patients with TRD. All patients were in stable remission after an optimization course of esketamine nasal spray plus an oral antidepressant.
Study design
- Between October 2015 and February 2018, researchers conducted a phase III, multicenter, double-blind, randomized withdrawal study to evaluate the effect of continuation of esketamine on rates of relapse in patients with TRD who had responded to initial treatment with esketamine.
- Initially, 705 adults were enrolled. Of these participants, 455 proceeded to the optimization phase, in which they were treated with esketamine nasal spray plus an oral antidepressant.
- After 16 weeks of optimization treatment, 297 participants achieved remission or stable response and were randomized to a treatment group, which received continued esketamine nasal spray plus an oral antidepressant, or to a control group, which received a placebo nasal spray plus an oral antidepressant.
Outcomes
- Treatment with esketamine nasal spray and an oral antidepressant was associated with decreased rates of relapse compared with treatment with placebo nasal spray and an oral antidepressant. This was the case among patients who had achieved remission as well as those who had achieved stable response.
- Continued treatment with esketamine decreased the risk of relapse by 51%, with 40 participants in the treatment group experiencing relapse compared with 73 participants in the placebo group.
Continue to: Conclusion
Conclusion
- In patients with TRD who responded to initial treatment with esketamine, continuing esketamine plus an oral antidepressant resulted in clinically meaningful superiority in preventing relapse compared with a placebo nasal spray plus an oral antidepressant.
3. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
Many studies have documented the efficacy of ketamine as a rapid-onset antidepressant. Studies investigating the mechanism of this effect have focused on antagonism of N-methyl-
Study design
- This double-blind crossover study evaluated if opioid receptor activation is necessary for ketamine to have an antidepressant effect in patients with TRD.
- Twelve participants completed both sides of the study in a randomized order. Participants received placebo or naltrexone prior to an IV infusion of ketamine.
- Researchers measured patients’ scores on the Hamilton Depression Rating Scale (HAM-D) at baseline and 1 day after infusion. Response was defined as a ≥50% reduction in HAM-D score.
Outcomes
- Reductions in HAM-D scores among participants in the ketamine plus naltrexone group were significantly lower than those of participants in the ketamine plus placebo group.
- Dissociation related to ketamine use did not differ significantly between the naltrexone group and the placebo group.
Continue to: Conclusion
Conclusion
- This small study found a significant decrease in the antidepressant effect of ketamine infusion in patients with TRD when opioid receptors are blocked with naltrexone prior to infusion, which suggests opioid receptor activation is necessary for ketamine to be effective as an antidepressant.
- This appears to be consistent with observations of buprenorphine’s antidepressant effects. Caution is indicated until additional studies can further elucidate the mechanism of action of ketamine’s antidepressant effects (see "Ketamine/esketamine: Putative mechanism of action," page 32).
4. Nidich S, Mills PJ, Rainforth M, et al. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: a randomised controlled trial. Lancet Psychiatry. 2018;5(12):975-986.
Posttraumatic stress disorder (PTSD) is a common and important public health problem. Evidence-based treatments for PTSD include trauma-focused therapies such as prolonged exposure therapy (PE). However, some patients may not respond to PE, drop out, or elect not to pursue it. Researchers continue to explore treatments that are non-trauma-focused, such as mindfulness meditation and interpersonal psychotherapy. In a 3-group comparative effectiveness trial, Nidich et al4 examined the efficacy of a non-trauma-focused intervention, transcendental meditation (TM), in reducing PTSD symptom severity and depression in veterans.
Study design
- Researchers recruited 203 veterans with PTSD from the Department of Veterans Affairs (VA) San Diego Healthcare System between June 2013 and October 2016.
- Participants were randomly assigned to 1 of 3 groups: 68 to TM, 68 to PE, and 67 to PTSD health education (HE).
- Each group received 12 sessions over 12 weeks. In addition to group and individual sessions, all participants received daily practice or assignments.
- The Clinician-Administered PTSD Scale (CAPS) was used to assess symptoms before and after treatment.
Outcomes
- The primary outcome assessed was change in PTSD symptom severity at the end of the study compared with baseline as measured by change in CAPS score.
- Transcendental meditation was found to be significantly non-inferior to PE, with a mean change in CAPS score of −16.1 in the TM group and −11.2 in the PE group.
- Both the TM and PE groups also had significant reductions in CAPS scores compared with the HE group, which had a mean change in CAPS score of −2.5.
Continue to: Conclusion
Conclusion
- Transcendental meditation is significantly not inferior to PE in the treatment of veterans with PTSD.
- The findings from this first comparative effectiveness trial comparing TM with an established psychotherapy for PTSD suggests the feasibility and efficacy of TM as an alternative therapy for veterans with PTSD.
- Because TM is self-administered after an initial expert training, it may offer an easy-to-implement approach that may be more accessible to veterans than other treatments.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
Several smaller randomized trials of prazosin involving a total of 283 active-duty service members, veterans, and civilian participants have shown efficacy of prazosin for PTSD-related nightmares, sleep disturbance, and overall clinical functioning. However, in a recent trial, Raskind et al5 failed to demonstrate such efficacy.
Study design
- Veterans with chronic PTSD nightmares were recruited from 13 VA medical centers to participate in a 26-week, double-blind, randomized controlled trial.
- A total of 304 participants were randomized to a prazosin treatment group (n = 152) or a placebo control group (n = 152).
- During the first 10 weeks, prazosin or placebo were administered in an escalating fashion up to a maximum dose.
- The CAPS, Pittsburgh Sleep Quality Index (PSQI), and Clinical Global Impressions of Change (CGIC) scores were measured at baseline, after 10 weeks, and after 26 weeks.
Outcomes
- Three primary outcomes measures were assessed: change in score from baseline to 10 weeks on CAPS item B2, the PSQI, and the CGIC.
- A secondary measure was change in score from baseline of the same measures at 26 weeks.
- There was no significant difference between the prazosin group and the placebo group in any of the primary or secondary measures.
Continue to: Conclusion
Conclusion
- Compared with placebo, prazosin was not associated with improvement in nightmares or sleep quality for veterans with chronic PTSD nightmares.
- Because psychosocial instability was an exclusion criterion, it is possible that a selection bias resulting from recruitment of patients who were mainly in clinically stable condition accounted for these negative results, since symptoms in such patients were less likely to be ameliorated with antiadrenergic treatment.
Treatment-resistant depression in veterans is a major clinical challenge because of these patients’ increased risk of suicide. Repetitive transcranial magnetic stimulation (rTMS) has shown promising results for TRD. In a randomized trial, Yesavage et al6 compared rTMS vs sham rTMS in veterans with TRD.
Study design
- Veterans with TRD were recruited from 9 VA medical centers throughout the United States between September 2012 and May 2016.
- Researchers randomized 164 participants into 1 of 2 groups in a double-blind fashion. The treatment group (n = 81) received left prefrontal rTMS, and the control group (n = 83) received sham rTMS.
Outcomes
- In an intention-to-treat analysis, remission rate (defined as a HAM-D score of ≤10) was assessed as the primary outcome measure.
- Remission was seen in both groups, with 40.7% of the treatment group achieving remission and 37.4% of the control group achieving remission. However, the difference between the 2 groups was not significant (P = .67), with an odds ratio of 1.16.
Continue to: Conclusion
Conclusion
- In this study, treatment with rTMS did not show a statistically significant difference in rates of remission from TRD in veterans compared with sham rTMS. This differs from previous rTMS trials in non-veteran patients.
- The findings of this study also differed from those of other rTMS research in terms of the high remission rates that were seen in both the active and sham groups.
Bottom Line
The risk of death might be increased in children and young adults who receive highdose antipsychotics. Continued treatment with intranasal esketamine may help prevent relapse in patients with treatment-resistant depression (TRD) who initially respond to esketamine. The antidepressant effects of ketamine might be associated with opioid receptor activation. Transcendental meditation may be helpful for patients with posttraumatic stress disorder (PTSD), while prazosin might not improve nightmares or sleep quality in patients with PTSD. Repetitive transcranial magnetic stimulation (rTMS) might not be any more effective than sham rTMS for veterans with TRD.
Related Resources
- NEJM Journal Watch. www.jwatch.org.
- F1000 Prime. https://f1000.com/prime/home.
- BMJ Journals Evidence-Based Mental Health. https://ebmh.bmj.com.
Drug Brand Names
Buprenorphine • Subutex
Chlorpromazine • Thorazine
Esketamine nasal spray • Spravato
Ketamine • Ketalar
Naltrexone • Narcan
Prazosin • Minipress
Medical knowledge is growing faster than ever, as is the challenge of keeping up with this ever-growing body of information. Clinicians need a system or method to help them sort and evaluate the quality of new information before they can apply it to clinical care. Without such a system, when facing an overload of information, most of us tend to take the first or the most easily accessed information, without considering the quality of such information. As a result, the use of poor-quality information affects the quality and outcome of care we provide, and costs billions of dollars annually in problems associated with underuse, overuse, and misuse of treatments.
In an effort to sort and evaluate recently published research that is ready for clinical use, the first author (SAS) used the following 3-step methodology:
1. Searched literature for research findings suggesting readiness for clinical utilization published between July 1, 2018 and June 30, 2019.
2. Surveyed members of the American Association of Chairs of Departments of Psychiatry, the American Association of Community Psychiatrists, the American Association of Psychiatric Administrators, the North Carolina Psychiatric Association, the Group for the Advancement of Psychiatry, and many other colleagues by asking them: “Among the articles published from July 1, 2018 to June 30, 2019, which ones in your opinion have (or are likely to have or should have) affected/changed the clinical practice of psychiatry?”
3. Looked for appraisals in post-publication reviews such as NEJM Journal Watch, F1000 Prime, Evidence-Based Mental Health, commentaries in peer-reviewed journals, and other sources (see Related Resources).
We chose 12 articles based on their clinical relevance/applicability. Here in Part 1 we present brief descriptions of the 6 of top 12 papers chosen by this methodology; these studies are summarized in the Table.1-6 The order in which they appear in this article is arbitrary. The remaining 6 studies will be reviewed in Part 2 in the February 2020 issue of

1. Ray WA, Stein CM, Murray KT, et al. Association of antipsychotic treatment with risk of unexpected death among children and youths. JAMA Psychiatry. 2019;76(2):162-171.
Children and young adults are increasingly being prescribed antipsychotic medications. Studies have suggested that when these medications are used in adults and older patients, they are associated with an increased risk of death.7-9 Whether or not these medications are associated with an increased risk of death in children and youth has been unknown. Ray et al1 compared the risk of unexpected death among children and youths who were beginning treatment with an antipsychotic or control medications.
Study design
- This retrospective cohort study evaluated children and young adults age 5 to 24 who were enrolled in Medicaid in Tennessee between 1999 and 2014.
- New antipsychotic use at both a higher dose (>50 mg chlorpromazine equivalents) and a lower dose (≤50 mg chlorpromazine equivalents) was compared with new use of a control medication, including attention-deficit/hyperactivity disorder medications, antidepressants, and mood stabilizers.
- There were 189,361 participants in the control group, 28,377 participants in the lower-dose antipsychotic group, and 30,120 participants in the higher-dose antipsychotic group.
Outcomes
- The primary outcome was death due to injury or suicide or unexpected death occurring during study follow-up.
- The incidence of death in the higher-dose antipsychotic group (146.2 per 100,000 person-years) was significantly higher (P < .001) than the incidence of death in the control medications group (54.5 per 100,000 person years).
- There was no similar significant difference between the lower-dose antipsychotic group and the control medications group.
Continue to: Conclusion
Conclusion
- Higher-dose antipsychotic use is associated with increased rates of unexpected deaths in children and young adults.
- As with all association studies, no direct line connected cause and effect. However, these results reinforce recommendations for careful prescribing and monitoring of antipsychotic regimens for children and youths, and the need for larger antipsychotic safety studies in this population.
- Examining risks associated with specific antipsychotics will require larger datasets, but will be critical for our understanding of the risks and benefits.
2. Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893-903.
Controlled studies have shown esketamine has efficacy for treatment-resistant depression (TRD), but these studies have been only short-term, and the long-term effects of esketamine for TRD have not been established. To fill that gap, Daly et al2 assessed the efficacy of esketamine nasal spray plus an oral antidepressant vs a placebo nasal spray plus an oral antidepressant in delaying relapse of depressive symptoms in patients with TRD. All patients were in stable remission after an optimization course of esketamine nasal spray plus an oral antidepressant.
Study design
- Between October 2015 and February 2018, researchers conducted a phase III, multicenter, double-blind, randomized withdrawal study to evaluate the effect of continuation of esketamine on rates of relapse in patients with TRD who had responded to initial treatment with esketamine.
- Initially, 705 adults were enrolled. Of these participants, 455 proceeded to the optimization phase, in which they were treated with esketamine nasal spray plus an oral antidepressant.
- After 16 weeks of optimization treatment, 297 participants achieved remission or stable response and were randomized to a treatment group, which received continued esketamine nasal spray plus an oral antidepressant, or to a control group, which received a placebo nasal spray plus an oral antidepressant.
Outcomes
- Treatment with esketamine nasal spray and an oral antidepressant was associated with decreased rates of relapse compared with treatment with placebo nasal spray and an oral antidepressant. This was the case among patients who had achieved remission as well as those who had achieved stable response.
- Continued treatment with esketamine decreased the risk of relapse by 51%, with 40 participants in the treatment group experiencing relapse compared with 73 participants in the placebo group.
Continue to: Conclusion
Conclusion
- In patients with TRD who responded to initial treatment with esketamine, continuing esketamine plus an oral antidepressant resulted in clinically meaningful superiority in preventing relapse compared with a placebo nasal spray plus an oral antidepressant.
3. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
Many studies have documented the efficacy of ketamine as a rapid-onset antidepressant. Studies investigating the mechanism of this effect have focused on antagonism of N-methyl-
Study design
- This double-blind crossover study evaluated if opioid receptor activation is necessary for ketamine to have an antidepressant effect in patients with TRD.
- Twelve participants completed both sides of the study in a randomized order. Participants received placebo or naltrexone prior to an IV infusion of ketamine.
- Researchers measured patients’ scores on the Hamilton Depression Rating Scale (HAM-D) at baseline and 1 day after infusion. Response was defined as a ≥50% reduction in HAM-D score.
Outcomes
- Reductions in HAM-D scores among participants in the ketamine plus naltrexone group were significantly lower than those of participants in the ketamine plus placebo group.
- Dissociation related to ketamine use did not differ significantly between the naltrexone group and the placebo group.
Continue to: Conclusion
Conclusion
- This small study found a significant decrease in the antidepressant effect of ketamine infusion in patients with TRD when opioid receptors are blocked with naltrexone prior to infusion, which suggests opioid receptor activation is necessary for ketamine to be effective as an antidepressant.
- This appears to be consistent with observations of buprenorphine’s antidepressant effects. Caution is indicated until additional studies can further elucidate the mechanism of action of ketamine’s antidepressant effects (see "Ketamine/esketamine: Putative mechanism of action," page 32).
4. Nidich S, Mills PJ, Rainforth M, et al. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: a randomised controlled trial. Lancet Psychiatry. 2018;5(12):975-986.
Posttraumatic stress disorder (PTSD) is a common and important public health problem. Evidence-based treatments for PTSD include trauma-focused therapies such as prolonged exposure therapy (PE). However, some patients may not respond to PE, drop out, or elect not to pursue it. Researchers continue to explore treatments that are non-trauma-focused, such as mindfulness meditation and interpersonal psychotherapy. In a 3-group comparative effectiveness trial, Nidich et al4 examined the efficacy of a non-trauma-focused intervention, transcendental meditation (TM), in reducing PTSD symptom severity and depression in veterans.
Study design
- Researchers recruited 203 veterans with PTSD from the Department of Veterans Affairs (VA) San Diego Healthcare System between June 2013 and October 2016.
- Participants were randomly assigned to 1 of 3 groups: 68 to TM, 68 to PE, and 67 to PTSD health education (HE).
- Each group received 12 sessions over 12 weeks. In addition to group and individual sessions, all participants received daily practice or assignments.
- The Clinician-Administered PTSD Scale (CAPS) was used to assess symptoms before and after treatment.
Outcomes
- The primary outcome assessed was change in PTSD symptom severity at the end of the study compared with baseline as measured by change in CAPS score.
- Transcendental meditation was found to be significantly non-inferior to PE, with a mean change in CAPS score of −16.1 in the TM group and −11.2 in the PE group.
- Both the TM and PE groups also had significant reductions in CAPS scores compared with the HE group, which had a mean change in CAPS score of −2.5.
Continue to: Conclusion
Conclusion
- Transcendental meditation is significantly not inferior to PE in the treatment of veterans with PTSD.
- The findings from this first comparative effectiveness trial comparing TM with an established psychotherapy for PTSD suggests the feasibility and efficacy of TM as an alternative therapy for veterans with PTSD.
- Because TM is self-administered after an initial expert training, it may offer an easy-to-implement approach that may be more accessible to veterans than other treatments.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
Several smaller randomized trials of prazosin involving a total of 283 active-duty service members, veterans, and civilian participants have shown efficacy of prazosin for PTSD-related nightmares, sleep disturbance, and overall clinical functioning. However, in a recent trial, Raskind et al5 failed to demonstrate such efficacy.
Study design
- Veterans with chronic PTSD nightmares were recruited from 13 VA medical centers to participate in a 26-week, double-blind, randomized controlled trial.
- A total of 304 participants were randomized to a prazosin treatment group (n = 152) or a placebo control group (n = 152).
- During the first 10 weeks, prazosin or placebo were administered in an escalating fashion up to a maximum dose.
- The CAPS, Pittsburgh Sleep Quality Index (PSQI), and Clinical Global Impressions of Change (CGIC) scores were measured at baseline, after 10 weeks, and after 26 weeks.
Outcomes
- Three primary outcomes measures were assessed: change in score from baseline to 10 weeks on CAPS item B2, the PSQI, and the CGIC.
- A secondary measure was change in score from baseline of the same measures at 26 weeks.
- There was no significant difference between the prazosin group and the placebo group in any of the primary or secondary measures.
Continue to: Conclusion
Conclusion
- Compared with placebo, prazosin was not associated with improvement in nightmares or sleep quality for veterans with chronic PTSD nightmares.
- Because psychosocial instability was an exclusion criterion, it is possible that a selection bias resulting from recruitment of patients who were mainly in clinically stable condition accounted for these negative results, since symptoms in such patients were less likely to be ameliorated with antiadrenergic treatment.
Treatment-resistant depression in veterans is a major clinical challenge because of these patients’ increased risk of suicide. Repetitive transcranial magnetic stimulation (rTMS) has shown promising results for TRD. In a randomized trial, Yesavage et al6 compared rTMS vs sham rTMS in veterans with TRD.
Study design
- Veterans with TRD were recruited from 9 VA medical centers throughout the United States between September 2012 and May 2016.
- Researchers randomized 164 participants into 1 of 2 groups in a double-blind fashion. The treatment group (n = 81) received left prefrontal rTMS, and the control group (n = 83) received sham rTMS.
Outcomes
- In an intention-to-treat analysis, remission rate (defined as a HAM-D score of ≤10) was assessed as the primary outcome measure.
- Remission was seen in both groups, with 40.7% of the treatment group achieving remission and 37.4% of the control group achieving remission. However, the difference between the 2 groups was not significant (P = .67), with an odds ratio of 1.16.
Continue to: Conclusion
Conclusion
- In this study, treatment with rTMS did not show a statistically significant difference in rates of remission from TRD in veterans compared with sham rTMS. This differs from previous rTMS trials in non-veteran patients.
- The findings of this study also differed from those of other rTMS research in terms of the high remission rates that were seen in both the active and sham groups.
Bottom Line
The risk of death might be increased in children and young adults who receive highdose antipsychotics. Continued treatment with intranasal esketamine may help prevent relapse in patients with treatment-resistant depression (TRD) who initially respond to esketamine. The antidepressant effects of ketamine might be associated with opioid receptor activation. Transcendental meditation may be helpful for patients with posttraumatic stress disorder (PTSD), while prazosin might not improve nightmares or sleep quality in patients with PTSD. Repetitive transcranial magnetic stimulation (rTMS) might not be any more effective than sham rTMS for veterans with TRD.
Related Resources
- NEJM Journal Watch. www.jwatch.org.
- F1000 Prime. https://f1000.com/prime/home.
- BMJ Journals Evidence-Based Mental Health. https://ebmh.bmj.com.
Drug Brand Names
Buprenorphine • Subutex
Chlorpromazine • Thorazine
Esketamine nasal spray • Spravato
Ketamine • Ketalar
Naltrexone • Narcan
Prazosin • Minipress
1. Ray WA, Stein CM, Murray KT, et al. Association of antipsychotic treatment with risk of unexpected death among children and youths. JAMA Psychiatry. 2019;76(2):162-171.
2. Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893-903.
3. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
4. Nidich S, Mills PJ, Rainforth M, et al. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: a randomized controlled trial. Lancet Psychiatry. 2018;5(12):975-986.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
6. Yesavage JA, Fairchild JK, Mi Z, et al. Effect of repetitive transcranial magnetic stimulation on treatment-resistant major depression in US veterans: a randomized clinical trial. JAMA Psychiatry. 2018;75(9):884-893.
7. Ray WA, Meredith S, Thapa PB, et al. Antipsychotics and the risk of sudden cardiac death. Arch Gen Psychiatry. 2001;58(12):1161-1167.
8. Ray WA, Chung CP, Murray KT, Hall K, Stein CM. Atypical antipsychotic drugs and the risk of sudden cardiac death. N Engl J Med. 2009;360(3):225-235.
9. Jeste DV, Blazer D, Casey D, et al. ACNP White Paper: update on use of antipsychotic drugs in elderly persons with dementia. Neuropsychopharmacology. 2008;33(5):957-970.
1. Ray WA, Stein CM, Murray KT, et al. Association of antipsychotic treatment with risk of unexpected death among children and youths. JAMA Psychiatry. 2019;76(2):162-171.
2. Daly EJ, Trivedi MH, Janik A, et al. Efficacy of esketamine nasal spray plus oral antidepressant treatment for relapse prevention in patients with treatment-resistant depression: a randomized clinical trial. JAMA Psychiatry. 2019;76(9):893-903.
3. Williams NR, Heifets BD, Blasey C, et al. Attenuation of antidepressant effects of ketamine by opioid receptor antagonism. Am J Psychiatry. 2018;175(12):1205-1215.
4. Nidich S, Mills PJ, Rainforth M, et al. Non-trauma-focused meditation versus exposure therapy in veterans with post-traumatic stress disorder: a randomized controlled trial. Lancet Psychiatry. 2018;5(12):975-986.
5. Raskind MA, Peskind ER, Chow B, et al. Trial of prazosin for post-traumatic stress disorder in military veterans. N Engl J Med. 2018;378(6):507-517.
6. Yesavage JA, Fairchild JK, Mi Z, et al. Effect of repetitive transcranial magnetic stimulation on treatment-resistant major depression in US veterans: a randomized clinical trial. JAMA Psychiatry. 2018;75(9):884-893.
7. Ray WA, Meredith S, Thapa PB, et al. Antipsychotics and the risk of sudden cardiac death. Arch Gen Psychiatry. 2001;58(12):1161-1167.
8. Ray WA, Chung CP, Murray KT, Hall K, Stein CM. Atypical antipsychotic drugs and the risk of sudden cardiac death. N Engl J Med. 2009;360(3):225-235.
9. Jeste DV, Blazer D, Casey D, et al. ACNP White Paper: update on use of antipsychotic drugs in elderly persons with dementia. Neuropsychopharmacology. 2008;33(5):957-970.
SABCS research changes practice
In this edition of “How I Will Treat My Next Patient,” I highlight two presentations from the recent San Antonio Breast Cancer Symposium (SABCS) that will likely change practice for some breast cancer patients – even before the ball drops in Times Square on New Year’s Eve.
Residual cancer burden
Researchers reported a multi-institutional analysis of individual patient-level data on 5,160 patients who had received neoadjuvant chemotherapy (NAC) for localized breast cancer at 11 different centers. They found that residual cancer burden (RCB) was significantly associated with event-free (EFS) and distant recurrence-free survival. The value of calculating RCB was seen across all breast cancer tumor phenotypes (SABCS 2019, Abstract GS5-01).
RCB is calculated by analyzing the residual disease after NAC for the primary tumor bed, the number of positive axillary nodes, and the size of largest node metastasis. It is graded from RCB-0 (pCR) to RCB-III (extensive residual disease).
Both EFS and distant recurrence-free survival were strongly associated with RCB for the overall population and for each breast cancer subtype. For hormone receptor–positive/HER2-negative disease there was a slight hiccup in that RCB-0 was associated with a 10-year EFS of 81%, but EFS was 86% for RCB-I. Fortunately, the prognostic reliability of RCB was clear for RCB-II (69%) and RCB-III (52%). The presenter, W. Fraser Symmans, MD, commented that RCB is most prognostic when higher levels of residual disease are present. RCB remained prognostic in multivariate models adjusting for age, grade, and clinical T and N stage at diagnosis.
How these results influence practice
After neoadjuvant chemotherapy in patients with localized breast cancer, we struggle when patients ask: “So, doctor, how am I likely to do?” We piece together a complicated – and inconsistent – answer, based on breast cancer subtype, original stage of disease, whether pCR was attained, and other factors.
For patients with triple-negative breast cancer, we have the option of adding capecitabine and/or participation in ongoing clinical trials, for patients with residual disease after NAC. Among the HER2-positive patients, we have data from the KATHERINE, ExtaNET, and APHINITY trials that provide options for additional treatment in those patients, as well. For patients with potentially hormonally responsive, HER2-negative disease, we can emphasize the importance of postoperative adjuvant endocrine therapy, the mandate for continued adherence to oral therapy, and the lower likelihood of (and prognostic value of) pCR in that breast cancer subtype.
Where we previously had a complicated, nuanced discussion, we now have data from a multi-institutional, meticulous analysis to guide further treatment, candidacy for clinical trials, and our expectations of what would be meaningful results from additional therapy. This meets my definition of “practice-changing” research.
APHINITY follow-up
APHINITY was a randomized, multicenter, double-blind, placebo-controlled trial that previously demonstrated that pertuzumab added to standard chemotherapy plus 1 year of trastuzumab in operable HER2-positive breast cancer was associated with modest, but statistically significant, improvement in invasive disease–free survival (IDFS), compared with placebo and chemotherapy plus trastuzumab (hazard ratio, 0.81; P = .04). When the initial results – with a median follow-up of 45.4 months – were published, the authors promised an update at 6 years. Martine Piccart, MD, PhD, provided that update at the 2019 SABCS (Abstract GS1-04).
In the updated analysis, overall survival was 94.8% among patients on the pertuzumab arm and 93.9% among patients taking placebo (HR, 0.85). IDFS rates were 90.6% versus 87.8% in the intent-to-treat population. The investigator commented that this difference was caused mainly by a reduction in distant and loco-regional recurrences.
Central nervous system metastases, contralateral invasive breast cancers, and death without a prior event were no different between the two treatment groups.
Improvement in IDFS (the primary endpoint) from pertuzumab was most impressive in the node-positive cohort, 87.9% versus 83.4% for placebo (HR, 0.72). There was no benefit in the node-negative population (95.0% vs. 94.9%; HR, 1.02).
In contrast to the 3-year analysis, the benefits in IDFS were seen regardless of hormone receptor status. Additionally, no new safety concerns emerged. The rate of severe cardiac events was below 1% in both groups.
How these results influence practice
“Patience is a virtue” appeared for the first time in the English language around 1360, in William Langland’s poem “Piers Plowman.” They are true words, indeed.
When the initial results of APHINITY were published in 2017, they led to the approval of pertuzumab as an addition to chemotherapy plus trastuzumab for high-risk, early, HER2-positive breast cancer patients. Still, the difference in IDFS curves was visually unimpressive (absolute difference 1.7% in the intent-to-treat and 3.2% in the node-positive patients) and pertuzumab was associated with more grade 3 toxicities (primarily diarrhea) and treatment discontinuations.
An optimist would have observed that the IDFS curves in 2017 did not start to diverge until 24 months and would have noted that the divergence increased with time. That virtuously patient person would have expected that the second interim analysis would show larger absolute benefits, and that the hormone receptor–positive patients (64% of the total) would not yet have relapsed by the first interim analysis and that pertuzumab’s benefit in that group would emerge late.
That appears to be what is happening. It provides hope that an overall survival benefit will become more evident with time. If the target P value for an overall survival difference (.0012) is met by the time of the third interim analysis, our patience (and the FDA’s decision to grant approval for pertuzumab for this indication) will have been amply rewarded. For selected HER2-posiitve patients with high RCB, especially those who tolerated neoadjuvant trastuzumab plus pertuzumab regimens, postoperative adjuvant pertuzumab may be an appealing option.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight two presentations from the recent San Antonio Breast Cancer Symposium (SABCS) that will likely change practice for some breast cancer patients – even before the ball drops in Times Square on New Year’s Eve.
Residual cancer burden
Researchers reported a multi-institutional analysis of individual patient-level data on 5,160 patients who had received neoadjuvant chemotherapy (NAC) for localized breast cancer at 11 different centers. They found that residual cancer burden (RCB) was significantly associated with event-free (EFS) and distant recurrence-free survival. The value of calculating RCB was seen across all breast cancer tumor phenotypes (SABCS 2019, Abstract GS5-01).
RCB is calculated by analyzing the residual disease after NAC for the primary tumor bed, the number of positive axillary nodes, and the size of largest node metastasis. It is graded from RCB-0 (pCR) to RCB-III (extensive residual disease).
Both EFS and distant recurrence-free survival were strongly associated with RCB for the overall population and for each breast cancer subtype. For hormone receptor–positive/HER2-negative disease there was a slight hiccup in that RCB-0 was associated with a 10-year EFS of 81%, but EFS was 86% for RCB-I. Fortunately, the prognostic reliability of RCB was clear for RCB-II (69%) and RCB-III (52%). The presenter, W. Fraser Symmans, MD, commented that RCB is most prognostic when higher levels of residual disease are present. RCB remained prognostic in multivariate models adjusting for age, grade, and clinical T and N stage at diagnosis.
How these results influence practice
After neoadjuvant chemotherapy in patients with localized breast cancer, we struggle when patients ask: “So, doctor, how am I likely to do?” We piece together a complicated – and inconsistent – answer, based on breast cancer subtype, original stage of disease, whether pCR was attained, and other factors.
For patients with triple-negative breast cancer, we have the option of adding capecitabine and/or participation in ongoing clinical trials, for patients with residual disease after NAC. Among the HER2-positive patients, we have data from the KATHERINE, ExtaNET, and APHINITY trials that provide options for additional treatment in those patients, as well. For patients with potentially hormonally responsive, HER2-negative disease, we can emphasize the importance of postoperative adjuvant endocrine therapy, the mandate for continued adherence to oral therapy, and the lower likelihood of (and prognostic value of) pCR in that breast cancer subtype.
Where we previously had a complicated, nuanced discussion, we now have data from a multi-institutional, meticulous analysis to guide further treatment, candidacy for clinical trials, and our expectations of what would be meaningful results from additional therapy. This meets my definition of “practice-changing” research.
APHINITY follow-up
APHINITY was a randomized, multicenter, double-blind, placebo-controlled trial that previously demonstrated that pertuzumab added to standard chemotherapy plus 1 year of trastuzumab in operable HER2-positive breast cancer was associated with modest, but statistically significant, improvement in invasive disease–free survival (IDFS), compared with placebo and chemotherapy plus trastuzumab (hazard ratio, 0.81; P = .04). When the initial results – with a median follow-up of 45.4 months – were published, the authors promised an update at 6 years. Martine Piccart, MD, PhD, provided that update at the 2019 SABCS (Abstract GS1-04).
In the updated analysis, overall survival was 94.8% among patients on the pertuzumab arm and 93.9% among patients taking placebo (HR, 0.85). IDFS rates were 90.6% versus 87.8% in the intent-to-treat population. The investigator commented that this difference was caused mainly by a reduction in distant and loco-regional recurrences.
Central nervous system metastases, contralateral invasive breast cancers, and death without a prior event were no different between the two treatment groups.
Improvement in IDFS (the primary endpoint) from pertuzumab was most impressive in the node-positive cohort, 87.9% versus 83.4% for placebo (HR, 0.72). There was no benefit in the node-negative population (95.0% vs. 94.9%; HR, 1.02).
In contrast to the 3-year analysis, the benefits in IDFS were seen regardless of hormone receptor status. Additionally, no new safety concerns emerged. The rate of severe cardiac events was below 1% in both groups.
How these results influence practice
“Patience is a virtue” appeared for the first time in the English language around 1360, in William Langland’s poem “Piers Plowman.” They are true words, indeed.
When the initial results of APHINITY were published in 2017, they led to the approval of pertuzumab as an addition to chemotherapy plus trastuzumab for high-risk, early, HER2-positive breast cancer patients. Still, the difference in IDFS curves was visually unimpressive (absolute difference 1.7% in the intent-to-treat and 3.2% in the node-positive patients) and pertuzumab was associated with more grade 3 toxicities (primarily diarrhea) and treatment discontinuations.
An optimist would have observed that the IDFS curves in 2017 did not start to diverge until 24 months and would have noted that the divergence increased with time. That virtuously patient person would have expected that the second interim analysis would show larger absolute benefits, and that the hormone receptor–positive patients (64% of the total) would not yet have relapsed by the first interim analysis and that pertuzumab’s benefit in that group would emerge late.
That appears to be what is happening. It provides hope that an overall survival benefit will become more evident with time. If the target P value for an overall survival difference (.0012) is met by the time of the third interim analysis, our patience (and the FDA’s decision to grant approval for pertuzumab for this indication) will have been amply rewarded. For selected HER2-posiitve patients with high RCB, especially those who tolerated neoadjuvant trastuzumab plus pertuzumab regimens, postoperative adjuvant pertuzumab may be an appealing option.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I Will Treat My Next Patient,” I highlight two presentations from the recent San Antonio Breast Cancer Symposium (SABCS) that will likely change practice for some breast cancer patients – even before the ball drops in Times Square on New Year’s Eve.
Residual cancer burden
Researchers reported a multi-institutional analysis of individual patient-level data on 5,160 patients who had received neoadjuvant chemotherapy (NAC) for localized breast cancer at 11 different centers. They found that residual cancer burden (RCB) was significantly associated with event-free (EFS) and distant recurrence-free survival. The value of calculating RCB was seen across all breast cancer tumor phenotypes (SABCS 2019, Abstract GS5-01).
RCB is calculated by analyzing the residual disease after NAC for the primary tumor bed, the number of positive axillary nodes, and the size of largest node metastasis. It is graded from RCB-0 (pCR) to RCB-III (extensive residual disease).
Both EFS and distant recurrence-free survival were strongly associated with RCB for the overall population and for each breast cancer subtype. For hormone receptor–positive/HER2-negative disease there was a slight hiccup in that RCB-0 was associated with a 10-year EFS of 81%, but EFS was 86% for RCB-I. Fortunately, the prognostic reliability of RCB was clear for RCB-II (69%) and RCB-III (52%). The presenter, W. Fraser Symmans, MD, commented that RCB is most prognostic when higher levels of residual disease are present. RCB remained prognostic in multivariate models adjusting for age, grade, and clinical T and N stage at diagnosis.
How these results influence practice
After neoadjuvant chemotherapy in patients with localized breast cancer, we struggle when patients ask: “So, doctor, how am I likely to do?” We piece together a complicated – and inconsistent – answer, based on breast cancer subtype, original stage of disease, whether pCR was attained, and other factors.
For patients with triple-negative breast cancer, we have the option of adding capecitabine and/or participation in ongoing clinical trials, for patients with residual disease after NAC. Among the HER2-positive patients, we have data from the KATHERINE, ExtaNET, and APHINITY trials that provide options for additional treatment in those patients, as well. For patients with potentially hormonally responsive, HER2-negative disease, we can emphasize the importance of postoperative adjuvant endocrine therapy, the mandate for continued adherence to oral therapy, and the lower likelihood of (and prognostic value of) pCR in that breast cancer subtype.
Where we previously had a complicated, nuanced discussion, we now have data from a multi-institutional, meticulous analysis to guide further treatment, candidacy for clinical trials, and our expectations of what would be meaningful results from additional therapy. This meets my definition of “practice-changing” research.
APHINITY follow-up
APHINITY was a randomized, multicenter, double-blind, placebo-controlled trial that previously demonstrated that pertuzumab added to standard chemotherapy plus 1 year of trastuzumab in operable HER2-positive breast cancer was associated with modest, but statistically significant, improvement in invasive disease–free survival (IDFS), compared with placebo and chemotherapy plus trastuzumab (hazard ratio, 0.81; P = .04). When the initial results – with a median follow-up of 45.4 months – were published, the authors promised an update at 6 years. Martine Piccart, MD, PhD, provided that update at the 2019 SABCS (Abstract GS1-04).
In the updated analysis, overall survival was 94.8% among patients on the pertuzumab arm and 93.9% among patients taking placebo (HR, 0.85). IDFS rates were 90.6% versus 87.8% in the intent-to-treat population. The investigator commented that this difference was caused mainly by a reduction in distant and loco-regional recurrences.
Central nervous system metastases, contralateral invasive breast cancers, and death without a prior event were no different between the two treatment groups.
Improvement in IDFS (the primary endpoint) from pertuzumab was most impressive in the node-positive cohort, 87.9% versus 83.4% for placebo (HR, 0.72). There was no benefit in the node-negative population (95.0% vs. 94.9%; HR, 1.02).
In contrast to the 3-year analysis, the benefits in IDFS were seen regardless of hormone receptor status. Additionally, no new safety concerns emerged. The rate of severe cardiac events was below 1% in both groups.
How these results influence practice
“Patience is a virtue” appeared for the first time in the English language around 1360, in William Langland’s poem “Piers Plowman.” They are true words, indeed.
When the initial results of APHINITY were published in 2017, they led to the approval of pertuzumab as an addition to chemotherapy plus trastuzumab for high-risk, early, HER2-positive breast cancer patients. Still, the difference in IDFS curves was visually unimpressive (absolute difference 1.7% in the intent-to-treat and 3.2% in the node-positive patients) and pertuzumab was associated with more grade 3 toxicities (primarily diarrhea) and treatment discontinuations.
An optimist would have observed that the IDFS curves in 2017 did not start to diverge until 24 months and would have noted that the divergence increased with time. That virtuously patient person would have expected that the second interim analysis would show larger absolute benefits, and that the hormone receptor–positive patients (64% of the total) would not yet have relapsed by the first interim analysis and that pertuzumab’s benefit in that group would emerge late.
That appears to be what is happening. It provides hope that an overall survival benefit will become more evident with time. If the target P value for an overall survival difference (.0012) is met by the time of the third interim analysis, our patience (and the FDA’s decision to grant approval for pertuzumab for this indication) will have been amply rewarded. For selected HER2-posiitve patients with high RCB, especially those who tolerated neoadjuvant trastuzumab plus pertuzumab regimens, postoperative adjuvant pertuzumab may be an appealing option.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
SGLT2 inhibitors for diabetes safe, effective in older adults
BUSAN, SOUTH KOREA – new research suggests.
Findings from a real-world observational study of 50 older adults with type 2 diabetes were recently presented at the International Diabetes Federation congress by Carlos Trescoli-Serrano, MD, of Hospital Universitario de la Ribera, Valencia, Spain.
“The results are quite similar to those of younger people. … In a selected population they are safe. It doesn’t matter if patients are older or younger. You have to review the patients for complications,” Dr. Trescoli-Serrano said in an interview.
Asked to comment, session moderator Samuel Dagogo-Jack, MD, said: “We need more and more studies in older people for reassurance. The elderly have impaired kidneys, and these drugs depend on kidney filtration. It’s good to have data on elderly patients in the real world.”
Most had comorbidities
The 50 adults were a mean age of 67 years, and the oldest was 81 years. They had a mean diabetes duration of 12.5 years, and 40% were women. They had all been taking SGLT2 inhibitors for more than 3 years (mean 43.9 months) during 2015-2019.
At baseline, most (75%) were also taking metformin, 45% also took sulfonylureas, 37% dipeptidyl peptidase-4 (DPP-4) inhibitors, and 36% insulin.
Most (81%) also had hypertension, half (51%) had hypercholesterolemia, a third (33%) had obesity, and 32% had a previous cardiovascular event.
With SGLT2-inhibitor treatment, the average hemoglobin A1c level dropped from 8.5% to 7.3% (P less than .01), body weight was reduced from 91.0 kg to 84.7 kg (P less than .01), systolic blood pressure from 134.5 mm Hg to 130.4 mm Hg (P = .02), and diastolic blood pressure from 76.2 mm Hg to 73.3 mm Hg (P = .04).
This effect on blood pressure “should be considered” because it means that “antihypertensive treatment might need to be reviewed,” Dr. Trescoli-Serrano commented.
There were no significant changes in estimated glomerular filtration rate (eGFR), microalbuminuria, lipid profile, hematocrit, or heart rate, although there were positive trends in both renal function and lipid profiles.
No patient had a fall, volume depletion symptoms, or diabetic ketoacidosis. However, 38% of patients were treated for urinary-genital infections, and 8% had to stop taking the SGLT2 inhibitor because of such infections.
That percentage seemed unusually high and was “an outlier, not the general experience,” commented Dr. Dagogo-Jack, chief of the Division of Endocrinology, Diabetes and Metabolism at the University of Tennessee Health Science Center, Memphis. He noted that infections with SGLT2 inhibitors are more often genital fungal than urinary tract infections, but the latter are more common in people with diabetes overall, and the study wasn’t randomized, so this might explain the finding.
New nonfatal cardiovascular events occurred in 22% of the patients, mainly cerebrovascular disease during the treatment period. Of those, 66% had had a previous cardiovascular event prior to starting on SGLT2 inhibitors.
Severe hypoglycemia was recorded in one patient, who was also taking insulin. Overall 10% died, mainly because of neoplastic causes.
“Long-term treatment with SGLT2 inhibitors are safe and effective when added to not-well-controlled elderly type 2 diabetes patients in a real-world experience,” Dr. Trescoli-Serrano concluded.
Dr. Dagogo-Jack is a consultant for Merck, Janssen, and Sanofi and owns stock in Dance Pharma and Jana Care.
A version of this story originally appeared on Medscape.com.
BUSAN, SOUTH KOREA – new research suggests.
Findings from a real-world observational study of 50 older adults with type 2 diabetes were recently presented at the International Diabetes Federation congress by Carlos Trescoli-Serrano, MD, of Hospital Universitario de la Ribera, Valencia, Spain.
“The results are quite similar to those of younger people. … In a selected population they are safe. It doesn’t matter if patients are older or younger. You have to review the patients for complications,” Dr. Trescoli-Serrano said in an interview.
Asked to comment, session moderator Samuel Dagogo-Jack, MD, said: “We need more and more studies in older people for reassurance. The elderly have impaired kidneys, and these drugs depend on kidney filtration. It’s good to have data on elderly patients in the real world.”
Most had comorbidities
The 50 adults were a mean age of 67 years, and the oldest was 81 years. They had a mean diabetes duration of 12.5 years, and 40% were women. They had all been taking SGLT2 inhibitors for more than 3 years (mean 43.9 months) during 2015-2019.
At baseline, most (75%) were also taking metformin, 45% also took sulfonylureas, 37% dipeptidyl peptidase-4 (DPP-4) inhibitors, and 36% insulin.
Most (81%) also had hypertension, half (51%) had hypercholesterolemia, a third (33%) had obesity, and 32% had a previous cardiovascular event.
With SGLT2-inhibitor treatment, the average hemoglobin A1c level dropped from 8.5% to 7.3% (P less than .01), body weight was reduced from 91.0 kg to 84.7 kg (P less than .01), systolic blood pressure from 134.5 mm Hg to 130.4 mm Hg (P = .02), and diastolic blood pressure from 76.2 mm Hg to 73.3 mm Hg (P = .04).
This effect on blood pressure “should be considered” because it means that “antihypertensive treatment might need to be reviewed,” Dr. Trescoli-Serrano commented.
There were no significant changes in estimated glomerular filtration rate (eGFR), microalbuminuria, lipid profile, hematocrit, or heart rate, although there were positive trends in both renal function and lipid profiles.
No patient had a fall, volume depletion symptoms, or diabetic ketoacidosis. However, 38% of patients were treated for urinary-genital infections, and 8% had to stop taking the SGLT2 inhibitor because of such infections.
That percentage seemed unusually high and was “an outlier, not the general experience,” commented Dr. Dagogo-Jack, chief of the Division of Endocrinology, Diabetes and Metabolism at the University of Tennessee Health Science Center, Memphis. He noted that infections with SGLT2 inhibitors are more often genital fungal than urinary tract infections, but the latter are more common in people with diabetes overall, and the study wasn’t randomized, so this might explain the finding.
New nonfatal cardiovascular events occurred in 22% of the patients, mainly cerebrovascular disease during the treatment period. Of those, 66% had had a previous cardiovascular event prior to starting on SGLT2 inhibitors.
Severe hypoglycemia was recorded in one patient, who was also taking insulin. Overall 10% died, mainly because of neoplastic causes.
“Long-term treatment with SGLT2 inhibitors are safe and effective when added to not-well-controlled elderly type 2 diabetes patients in a real-world experience,” Dr. Trescoli-Serrano concluded.
Dr. Dagogo-Jack is a consultant for Merck, Janssen, and Sanofi and owns stock in Dance Pharma and Jana Care.
A version of this story originally appeared on Medscape.com.
BUSAN, SOUTH KOREA – new research suggests.
Findings from a real-world observational study of 50 older adults with type 2 diabetes were recently presented at the International Diabetes Federation congress by Carlos Trescoli-Serrano, MD, of Hospital Universitario de la Ribera, Valencia, Spain.
“The results are quite similar to those of younger people. … In a selected population they are safe. It doesn’t matter if patients are older or younger. You have to review the patients for complications,” Dr. Trescoli-Serrano said in an interview.
Asked to comment, session moderator Samuel Dagogo-Jack, MD, said: “We need more and more studies in older people for reassurance. The elderly have impaired kidneys, and these drugs depend on kidney filtration. It’s good to have data on elderly patients in the real world.”
Most had comorbidities
The 50 adults were a mean age of 67 years, and the oldest was 81 years. They had a mean diabetes duration of 12.5 years, and 40% were women. They had all been taking SGLT2 inhibitors for more than 3 years (mean 43.9 months) during 2015-2019.
At baseline, most (75%) were also taking metformin, 45% also took sulfonylureas, 37% dipeptidyl peptidase-4 (DPP-4) inhibitors, and 36% insulin.
Most (81%) also had hypertension, half (51%) had hypercholesterolemia, a third (33%) had obesity, and 32% had a previous cardiovascular event.
With SGLT2-inhibitor treatment, the average hemoglobin A1c level dropped from 8.5% to 7.3% (P less than .01), body weight was reduced from 91.0 kg to 84.7 kg (P less than .01), systolic blood pressure from 134.5 mm Hg to 130.4 mm Hg (P = .02), and diastolic blood pressure from 76.2 mm Hg to 73.3 mm Hg (P = .04).
This effect on blood pressure “should be considered” because it means that “antihypertensive treatment might need to be reviewed,” Dr. Trescoli-Serrano commented.
There were no significant changes in estimated glomerular filtration rate (eGFR), microalbuminuria, lipid profile, hematocrit, or heart rate, although there were positive trends in both renal function and lipid profiles.
No patient had a fall, volume depletion symptoms, or diabetic ketoacidosis. However, 38% of patients were treated for urinary-genital infections, and 8% had to stop taking the SGLT2 inhibitor because of such infections.
That percentage seemed unusually high and was “an outlier, not the general experience,” commented Dr. Dagogo-Jack, chief of the Division of Endocrinology, Diabetes and Metabolism at the University of Tennessee Health Science Center, Memphis. He noted that infections with SGLT2 inhibitors are more often genital fungal than urinary tract infections, but the latter are more common in people with diabetes overall, and the study wasn’t randomized, so this might explain the finding.
New nonfatal cardiovascular events occurred in 22% of the patients, mainly cerebrovascular disease during the treatment period. Of those, 66% had had a previous cardiovascular event prior to starting on SGLT2 inhibitors.
Severe hypoglycemia was recorded in one patient, who was also taking insulin. Overall 10% died, mainly because of neoplastic causes.
“Long-term treatment with SGLT2 inhibitors are safe and effective when added to not-well-controlled elderly type 2 diabetes patients in a real-world experience,” Dr. Trescoli-Serrano concluded.
Dr. Dagogo-Jack is a consultant for Merck, Janssen, and Sanofi and owns stock in Dance Pharma and Jana Care.
A version of this story originally appeared on Medscape.com.
Vermont tops America’s Health Rankings for 2019
The award for healthiest state goes to Vermont in 2019, marking the fifth time the Green Mountain State has taken the top spot in the 30-year span of America’s Health Rankings.

The New England states took 3 of the top 5 spots and 4 of the top 10, while last year’s winner, Hawaii, dropped to third and missed out on the title for only the second time in the last 8 years, according to the America’s Heath Rankings annual report.
Another rankings tradition lived on, however, as Mississippi and Louisiana continued their battle to be the state with the “greatest opportunity for improvement.” In 2019, Mississippi managed to take that dishonor away from Louisiana, which had finished 50th in 2018. The two states have occupied the 49th and 50th spots in the rankings for the last 5 years, with Mississippi ahead 3-2 on 50th-place finishes, based on data from the AHR website.
Alaska (2019 rank, 27th), Virginia (15th), and Wyoming (19th) made the largest improvements, each moving up five spots since 2018, while Maine dropped from 16th to 21st for the largest decline among the states. A look back to the original rankings from 1990 puts New York on top of the list of improvers with a +29 over 30 years and shows Kansas to be the largest decliner with a change of –21, the report said.
At the national level, the report noted some key long-term health improvements and challenges:
- Smoking among adults is down 45% since 1990.
- Infant mortality declined by 43% and decreased in all 50 states.
- Diabetes prevalence has risen by 166% in adults since 1996.
- Obesity has increased by 166% since 1990.
The model used by AHR ranks states using 35 measures of public health in five broad categories: behaviors (Utah, 1st; La., 50th), community and environment (N.H., 1st; La. 50th), policy (Mass., 1st; Tex., 50th), clinical care (Mass., 1st; Miss. 50th), and health outcomes (Hawaii, 1st; Ala., 50th). Health measures include rates of excessive drinking, occupational fatalities, uninsured, preventable hospitalizations, and infant mortality.
America’s Health Rankings are produced by the American Public Health Association and the private, not-for-profit United Health Foundation, which was founded by UnitedHealth Group, operator of UnitedHealthcare.
The award for healthiest state goes to Vermont in 2019, marking the fifth time the Green Mountain State has taken the top spot in the 30-year span of America’s Health Rankings.

The New England states took 3 of the top 5 spots and 4 of the top 10, while last year’s winner, Hawaii, dropped to third and missed out on the title for only the second time in the last 8 years, according to the America’s Heath Rankings annual report.
Another rankings tradition lived on, however, as Mississippi and Louisiana continued their battle to be the state with the “greatest opportunity for improvement.” In 2019, Mississippi managed to take that dishonor away from Louisiana, which had finished 50th in 2018. The two states have occupied the 49th and 50th spots in the rankings for the last 5 years, with Mississippi ahead 3-2 on 50th-place finishes, based on data from the AHR website.
Alaska (2019 rank, 27th), Virginia (15th), and Wyoming (19th) made the largest improvements, each moving up five spots since 2018, while Maine dropped from 16th to 21st for the largest decline among the states. A look back to the original rankings from 1990 puts New York on top of the list of improvers with a +29 over 30 years and shows Kansas to be the largest decliner with a change of –21, the report said.
At the national level, the report noted some key long-term health improvements and challenges:
- Smoking among adults is down 45% since 1990.
- Infant mortality declined by 43% and decreased in all 50 states.
- Diabetes prevalence has risen by 166% in adults since 1996.
- Obesity has increased by 166% since 1990.
The model used by AHR ranks states using 35 measures of public health in five broad categories: behaviors (Utah, 1st; La., 50th), community and environment (N.H., 1st; La. 50th), policy (Mass., 1st; Tex., 50th), clinical care (Mass., 1st; Miss. 50th), and health outcomes (Hawaii, 1st; Ala., 50th). Health measures include rates of excessive drinking, occupational fatalities, uninsured, preventable hospitalizations, and infant mortality.
America’s Health Rankings are produced by the American Public Health Association and the private, not-for-profit United Health Foundation, which was founded by UnitedHealth Group, operator of UnitedHealthcare.
The award for healthiest state goes to Vermont in 2019, marking the fifth time the Green Mountain State has taken the top spot in the 30-year span of America’s Health Rankings.

The New England states took 3 of the top 5 spots and 4 of the top 10, while last year’s winner, Hawaii, dropped to third and missed out on the title for only the second time in the last 8 years, according to the America’s Heath Rankings annual report.
Another rankings tradition lived on, however, as Mississippi and Louisiana continued their battle to be the state with the “greatest opportunity for improvement.” In 2019, Mississippi managed to take that dishonor away from Louisiana, which had finished 50th in 2018. The two states have occupied the 49th and 50th spots in the rankings for the last 5 years, with Mississippi ahead 3-2 on 50th-place finishes, based on data from the AHR website.
Alaska (2019 rank, 27th), Virginia (15th), and Wyoming (19th) made the largest improvements, each moving up five spots since 2018, while Maine dropped from 16th to 21st for the largest decline among the states. A look back to the original rankings from 1990 puts New York on top of the list of improvers with a +29 over 30 years and shows Kansas to be the largest decliner with a change of –21, the report said.
At the national level, the report noted some key long-term health improvements and challenges:
- Smoking among adults is down 45% since 1990.
- Infant mortality declined by 43% and decreased in all 50 states.
- Diabetes prevalence has risen by 166% in adults since 1996.
- Obesity has increased by 166% since 1990.
The model used by AHR ranks states using 35 measures of public health in five broad categories: behaviors (Utah, 1st; La., 50th), community and environment (N.H., 1st; La. 50th), policy (Mass., 1st; Tex., 50th), clinical care (Mass., 1st; Miss. 50th), and health outcomes (Hawaii, 1st; Ala., 50th). Health measures include rates of excessive drinking, occupational fatalities, uninsured, preventable hospitalizations, and infant mortality.
America’s Health Rankings are produced by the American Public Health Association and the private, not-for-profit United Health Foundation, which was founded by UnitedHealth Group, operator of UnitedHealthcare.
Administrative data can help drive quality improvement in breast cancer care
Quality indicators of breast cancer care were successfully computed for more than 15,000 incident cases of breast cancer using electronic administrative databases in a project led by five regional oncology networks in Italy.
The project has shown that, despite some limitations in the use of administrative data to measure health care performance, “evaluating the quality of breast cancer care at a population level is possible,” investigators reported in the Journal of Oncology Practice.
The data obtained “from multiple administrative databases gathered in a real-world setting across five Italian regions” highlighted regional variations in breast cancer care and ways in which clinical guidelines were being overlooked, they wrote.
In doing so, the project confirmed that administrative data is “suitable” for measuring performance in health care and potentially useful for guiding quality improvement interventions. For instance, the project identified extensive use of blood tumor markers in breast cancer follow-up, wrote Valentina Guarneri, PhD, MD, of the University of Padova (Italy) and the Istituto Oncologico Veneto, also in Padova, and coauthors.
Oncologists and epidemiologists from the Italian regional oncology networks identified 46 clinically relevant indicators (9 structure indicators, 29 dealing with process, and 8 outcome indicators) by comparing pathways of care established by each network and identifying commonalities.
Of the 46 indicators, 22 were considered by the project leaders to be “potentially computable” from information retrieved by regional administrative databases. And of these 22 designed to be extractable, 9 (2 indicators of structure and 7 of process) were found to be actually evaluable for 15,342 cases of newly diagnosed invasive and/or in situ breast cancer diagnosed during 2016.
Blood tumor markers were tested in 44.2%-64.5% of patients in the first year after surgery – higher than the benchmark of 20% or less that was established to account for stage IV patients and other specific conditions in which markers might be indicated. National, international, and regional guidelines “discourage the use of blood tumor markers” in breast cancer follow-up, the investigators wrote.
The extensive use of these markers – observed across all five regions – is “a starting point to understanding how to improve clinical practice,” they added.
Other quality indicators that were evaluable included radiotherapy within 12 weeks after surgery if adjuvant chemotherapy is not administered (42%-83.8% in the project, compared with the benchmark of 90% or greater) and mammography 6-18 months after surgery (administered in 55.1%-72.6%, compared with the benchmark of 90% or greater), as well as the proportion of patients starting adjuvant systemic treatment (chemotherapy or endocrine therapy) within 60 days of surgery (for patients receiving systemic treatment).
To calculate the indicators, each regional cancer network used computerized sources of information including hospital discharge forms, outpatient records of diagnostic and therapeutic procedures, prescriptions of drugs reimbursed by the National Health Service in the hospital and outpatient settings, regional health registries, and the regional mortality registries.
All data used in the project came from regional repositories, which collect data from all National Health Service providers in the region, and not from single institutional repositories, the investigators noted.
More than half of the indicators expected to be assessable – but not found to be – were not computable as a result of data being unavailable (for example, pathology data) or incomplete, and as a result of data not being reliable for various reasons. The fact that examinations paid for directly by patients are not reported by the management systems of the National Health System was another complicating factor, they reported.
The authors disclosed funding and relationships with various pharmaceutical companies. The research was supported by the Periplo Association.
SOURCE: Guarneri V et al. J Oncol Pract. 2019 Dec 19. doi: 10.1200/JOP.19.00466.
Quality indicators of breast cancer care were successfully computed for more than 15,000 incident cases of breast cancer using electronic administrative databases in a project led by five regional oncology networks in Italy.
The project has shown that, despite some limitations in the use of administrative data to measure health care performance, “evaluating the quality of breast cancer care at a population level is possible,” investigators reported in the Journal of Oncology Practice.
The data obtained “from multiple administrative databases gathered in a real-world setting across five Italian regions” highlighted regional variations in breast cancer care and ways in which clinical guidelines were being overlooked, they wrote.
In doing so, the project confirmed that administrative data is “suitable” for measuring performance in health care and potentially useful for guiding quality improvement interventions. For instance, the project identified extensive use of blood tumor markers in breast cancer follow-up, wrote Valentina Guarneri, PhD, MD, of the University of Padova (Italy) and the Istituto Oncologico Veneto, also in Padova, and coauthors.
Oncologists and epidemiologists from the Italian regional oncology networks identified 46 clinically relevant indicators (9 structure indicators, 29 dealing with process, and 8 outcome indicators) by comparing pathways of care established by each network and identifying commonalities.
Of the 46 indicators, 22 were considered by the project leaders to be “potentially computable” from information retrieved by regional administrative databases. And of these 22 designed to be extractable, 9 (2 indicators of structure and 7 of process) were found to be actually evaluable for 15,342 cases of newly diagnosed invasive and/or in situ breast cancer diagnosed during 2016.
Blood tumor markers were tested in 44.2%-64.5% of patients in the first year after surgery – higher than the benchmark of 20% or less that was established to account for stage IV patients and other specific conditions in which markers might be indicated. National, international, and regional guidelines “discourage the use of blood tumor markers” in breast cancer follow-up, the investigators wrote.
The extensive use of these markers – observed across all five regions – is “a starting point to understanding how to improve clinical practice,” they added.
Other quality indicators that were evaluable included radiotherapy within 12 weeks after surgery if adjuvant chemotherapy is not administered (42%-83.8% in the project, compared with the benchmark of 90% or greater) and mammography 6-18 months after surgery (administered in 55.1%-72.6%, compared with the benchmark of 90% or greater), as well as the proportion of patients starting adjuvant systemic treatment (chemotherapy or endocrine therapy) within 60 days of surgery (for patients receiving systemic treatment).
To calculate the indicators, each regional cancer network used computerized sources of information including hospital discharge forms, outpatient records of diagnostic and therapeutic procedures, prescriptions of drugs reimbursed by the National Health Service in the hospital and outpatient settings, regional health registries, and the regional mortality registries.
All data used in the project came from regional repositories, which collect data from all National Health Service providers in the region, and not from single institutional repositories, the investigators noted.
More than half of the indicators expected to be assessable – but not found to be – were not computable as a result of data being unavailable (for example, pathology data) or incomplete, and as a result of data not being reliable for various reasons. The fact that examinations paid for directly by patients are not reported by the management systems of the National Health System was another complicating factor, they reported.
The authors disclosed funding and relationships with various pharmaceutical companies. The research was supported by the Periplo Association.
SOURCE: Guarneri V et al. J Oncol Pract. 2019 Dec 19. doi: 10.1200/JOP.19.00466.
Quality indicators of breast cancer care were successfully computed for more than 15,000 incident cases of breast cancer using electronic administrative databases in a project led by five regional oncology networks in Italy.
The project has shown that, despite some limitations in the use of administrative data to measure health care performance, “evaluating the quality of breast cancer care at a population level is possible,” investigators reported in the Journal of Oncology Practice.
The data obtained “from multiple administrative databases gathered in a real-world setting across five Italian regions” highlighted regional variations in breast cancer care and ways in which clinical guidelines were being overlooked, they wrote.
In doing so, the project confirmed that administrative data is “suitable” for measuring performance in health care and potentially useful for guiding quality improvement interventions. For instance, the project identified extensive use of blood tumor markers in breast cancer follow-up, wrote Valentina Guarneri, PhD, MD, of the University of Padova (Italy) and the Istituto Oncologico Veneto, also in Padova, and coauthors.
Oncologists and epidemiologists from the Italian regional oncology networks identified 46 clinically relevant indicators (9 structure indicators, 29 dealing with process, and 8 outcome indicators) by comparing pathways of care established by each network and identifying commonalities.
Of the 46 indicators, 22 were considered by the project leaders to be “potentially computable” from information retrieved by regional administrative databases. And of these 22 designed to be extractable, 9 (2 indicators of structure and 7 of process) were found to be actually evaluable for 15,342 cases of newly diagnosed invasive and/or in situ breast cancer diagnosed during 2016.
Blood tumor markers were tested in 44.2%-64.5% of patients in the first year after surgery – higher than the benchmark of 20% or less that was established to account for stage IV patients and other specific conditions in which markers might be indicated. National, international, and regional guidelines “discourage the use of blood tumor markers” in breast cancer follow-up, the investigators wrote.
The extensive use of these markers – observed across all five regions – is “a starting point to understanding how to improve clinical practice,” they added.
Other quality indicators that were evaluable included radiotherapy within 12 weeks after surgery if adjuvant chemotherapy is not administered (42%-83.8% in the project, compared with the benchmark of 90% or greater) and mammography 6-18 months after surgery (administered in 55.1%-72.6%, compared with the benchmark of 90% or greater), as well as the proportion of patients starting adjuvant systemic treatment (chemotherapy or endocrine therapy) within 60 days of surgery (for patients receiving systemic treatment).
To calculate the indicators, each regional cancer network used computerized sources of information including hospital discharge forms, outpatient records of diagnostic and therapeutic procedures, prescriptions of drugs reimbursed by the National Health Service in the hospital and outpatient settings, regional health registries, and the regional mortality registries.
All data used in the project came from regional repositories, which collect data from all National Health Service providers in the region, and not from single institutional repositories, the investigators noted.
More than half of the indicators expected to be assessable – but not found to be – were not computable as a result of data being unavailable (for example, pathology data) or incomplete, and as a result of data not being reliable for various reasons. The fact that examinations paid for directly by patients are not reported by the management systems of the National Health System was another complicating factor, they reported.
The authors disclosed funding and relationships with various pharmaceutical companies. The research was supported by the Periplo Association.
SOURCE: Guarneri V et al. J Oncol Pract. 2019 Dec 19. doi: 10.1200/JOP.19.00466.
FROM THE JOURNAL OF ONCOLOGY PRACTICE
Dual e-cigarette and combustible tobacco use compound respiratory disease risk
according to recent longitudinal analysis published in the American Journal of Preventive Medicine.
E-cigarettes have been promoted as a safer alternative to combustible tobacco, and until recently, there has been little and conflicting evidence by which to test this hypothesis. This study conducted by Dharma N. Bhatta, PhD, and Stanton A. Glantz, PhD, of the Center for Tobacco Control Research and Education at the University of California, San Francisco, is one of the first longitudinal examinations of e-cigarette use and controlling for combustible tobacco use.
Dr. Bhatta and Dr. Glantz performed a multivariable, logistic regression analysis of adults enrolled in the nationally representative, population-based, longitudinal Population Assessment of Tobacco and Health study. The researchers analyzed the tobacco use of adults in the study in three waves, following them through wave 1 (September 2013 to December 2014), wave 2 (October 2014 to October 2015), and wave 3 (October 2015 to October 2016), analyzing the data between 2018 and 2019. Overall, wave 1 began with 32,320 participants, and 15.1% of adults reported respiratory disease at baseline.
Lung or respiratory disease was assessed by asking participants whether they had been told by a health professional that they had chronic obstructive pulmonary disease, chronic bronchitis, emphysema, or asthma. The researchers defined e-cigarette and combustible tobacco use as participants who never, currently, or formerly used e-cigarettes or smoked combustible tobacco. Participants who indicated they used e-cigarettes or combustible tobacco frequently or infrequently were placed in the current-user group, while past users were those participants who said they used to, but no longer use e-cigarettes or combustible tobacco.
The results showed former e-cigarette use (adjusted odds ratio, 1.34; 95% confidence interval, 1.23-1.46) and current e-cigarette use (aOR, 1.32; 95% CI, 1.17-1.49) were associated with an increased risk of having incident respiratory disease.
The data showed a not unexpected statistically significant association between former combustible tobacco use (aOR, 1.29; 95% CI, 1.14-1.47) as well as current combustible tobacco use (aOR, 1.61; 95% CI, 1.42-1.82) and incident respiratory disease risk.
There was a statistically significant association between respiratory disease and former or current e-cigarette use for adults who did not have respiratory disease at baseline, after adjusting for factors such as current combustible tobacco use, clinical variables, and demographic differences. Participants in wave 1 who reported former (aOR, 1.31; 95% CI, 1.07-1.60) or current e-cigarette use (aOR, 1.29; 95% CI, 1.03-1.61) had a significantly higher risk of developing incident respiratory disease in subsequent waves. There was also a statistically significant association between use of combustible tobacco and subsequent respiratory disease in later waves of the study (aOR, 2.56; 95% CI, 1.92-3.41), which the researchers noted was independent of the usual risks associated with combustible tobacco.
The investigators also looked at the link between dual use of e-cigarettes and combustible tobacco and respiratory disease risk. “The much more common pattern is dual use, in which an e-cigarette user continues to smoke combusted tobacco products at the same time (93.7% of e-cigarette users at wave 2 and 91.2% at wave 3 also used combustible tobacco; 73.3% of e-cigarette users at wave 2 and 64.9% at wave 3 also smoked cigarettes),” they wrote.
The odds of developing respiratory disease for participants who used both e-cigarettes and combustible tobacco were 3.30, compared with a participant who never used e-cigarettes, with similar results seen when comparing e-cigarettes and cigarettes.
“Although switching from combustible tobacco, including cigarettes, to e-cigarettes theoretically could reduce the risk of developing respiratory disease, current evidence indicates a high prevalence of dual use, which is associated with in-creased risk beyond combustible tobacco use,” the investigators wrote.
Harold J. Farber, MD, FCCP, professor of pediatrics in the pulmonary section at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, said in an interview that the increased respiratory risk among dual users, who are likely using e-cigarettes and combustible tobacco together as a way to quit smoking, is particularly concerning.
“There is substantial reason to be concerned about efficacy of electronic cigarette products. Real-world observational studies have shown that, on average, tobacco smokers who use electronic cigarettes are less likely to stop smoking than those who do not use electronic cigarettes,” he said. “People who have stopped tobacco smoking but use electronic cigarettes are more likely to relapse to tobacco smoking than those who do not use electronic cigarettes.”
Dr. Farber noted that there are other Food and Drug Administration–approved medications for treating tobacco addiction. In addition, the World Health Organization, American Medical Association, Centers for Disease Control and Prevention, and FDA have all advised that e-cigarettes should not be used as smoking cessation aids, he said, especially in light of current outbreak of life-threatening e-cigarette and vaping lung injuries currently being investigated by the CDC and FDA.
“These study results suggest that the CDC reports of e-cigarette, or vaping, product use–associated lung injury are likely to be just the tip of the iceberg,” he said. “Although the CDC has identified vitamin E acetate–containing products as an important culprit, it is unlikely to be the only one. There are many substances in the emissions of e-cigarettes that have known irritant and/or toxic effects on the airways.”
Dr. Bhatta and Dr. Glantz acknowledged several limitations in their analysis, including the possibility of recall bias, not distinguishing between nondaily and daily e-cigarette or combustible tobacco use, and combining respiratory conditions together to achieve adequate power. The study shows an association, but the mechanism by which e-cigarettes may contribute to the development of lung disease remains under investigation.
This study was supported by grants from the National Institute on Drug Abuse; the National Cancer Institute; the FDA Center for Tobacco Products; the National Heart, Lung, and Blood Institute; and the University of California, San Francisco Helen Diller Family Comprehensive Cancer Center Global Cancer Program. Dr. Bhatta and Dr. Glantz reported no relevant conflicts of interest.
SOURCE: Bhatta DN, Glantz SA. Am J Prev Med. 2019 Dec 16. doi: 10.1016/j.amepre.2019.07.028.
according to recent longitudinal analysis published in the American Journal of Preventive Medicine.
E-cigarettes have been promoted as a safer alternative to combustible tobacco, and until recently, there has been little and conflicting evidence by which to test this hypothesis. This study conducted by Dharma N. Bhatta, PhD, and Stanton A. Glantz, PhD, of the Center for Tobacco Control Research and Education at the University of California, San Francisco, is one of the first longitudinal examinations of e-cigarette use and controlling for combustible tobacco use.
Dr. Bhatta and Dr. Glantz performed a multivariable, logistic regression analysis of adults enrolled in the nationally representative, population-based, longitudinal Population Assessment of Tobacco and Health study. The researchers analyzed the tobacco use of adults in the study in three waves, following them through wave 1 (September 2013 to December 2014), wave 2 (October 2014 to October 2015), and wave 3 (October 2015 to October 2016), analyzing the data between 2018 and 2019. Overall, wave 1 began with 32,320 participants, and 15.1% of adults reported respiratory disease at baseline.
Lung or respiratory disease was assessed by asking participants whether they had been told by a health professional that they had chronic obstructive pulmonary disease, chronic bronchitis, emphysema, or asthma. The researchers defined e-cigarette and combustible tobacco use as participants who never, currently, or formerly used e-cigarettes or smoked combustible tobacco. Participants who indicated they used e-cigarettes or combustible tobacco frequently or infrequently were placed in the current-user group, while past users were those participants who said they used to, but no longer use e-cigarettes or combustible tobacco.
The results showed former e-cigarette use (adjusted odds ratio, 1.34; 95% confidence interval, 1.23-1.46) and current e-cigarette use (aOR, 1.32; 95% CI, 1.17-1.49) were associated with an increased risk of having incident respiratory disease.
The data showed a not unexpected statistically significant association between former combustible tobacco use (aOR, 1.29; 95% CI, 1.14-1.47) as well as current combustible tobacco use (aOR, 1.61; 95% CI, 1.42-1.82) and incident respiratory disease risk.
There was a statistically significant association between respiratory disease and former or current e-cigarette use for adults who did not have respiratory disease at baseline, after adjusting for factors such as current combustible tobacco use, clinical variables, and demographic differences. Participants in wave 1 who reported former (aOR, 1.31; 95% CI, 1.07-1.60) or current e-cigarette use (aOR, 1.29; 95% CI, 1.03-1.61) had a significantly higher risk of developing incident respiratory disease in subsequent waves. There was also a statistically significant association between use of combustible tobacco and subsequent respiratory disease in later waves of the study (aOR, 2.56; 95% CI, 1.92-3.41), which the researchers noted was independent of the usual risks associated with combustible tobacco.
The investigators also looked at the link between dual use of e-cigarettes and combustible tobacco and respiratory disease risk. “The much more common pattern is dual use, in which an e-cigarette user continues to smoke combusted tobacco products at the same time (93.7% of e-cigarette users at wave 2 and 91.2% at wave 3 also used combustible tobacco; 73.3% of e-cigarette users at wave 2 and 64.9% at wave 3 also smoked cigarettes),” they wrote.
The odds of developing respiratory disease for participants who used both e-cigarettes and combustible tobacco were 3.30, compared with a participant who never used e-cigarettes, with similar results seen when comparing e-cigarettes and cigarettes.
“Although switching from combustible tobacco, including cigarettes, to e-cigarettes theoretically could reduce the risk of developing respiratory disease, current evidence indicates a high prevalence of dual use, which is associated with in-creased risk beyond combustible tobacco use,” the investigators wrote.
Harold J. Farber, MD, FCCP, professor of pediatrics in the pulmonary section at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, said in an interview that the increased respiratory risk among dual users, who are likely using e-cigarettes and combustible tobacco together as a way to quit smoking, is particularly concerning.
“There is substantial reason to be concerned about efficacy of electronic cigarette products. Real-world observational studies have shown that, on average, tobacco smokers who use electronic cigarettes are less likely to stop smoking than those who do not use electronic cigarettes,” he said. “People who have stopped tobacco smoking but use electronic cigarettes are more likely to relapse to tobacco smoking than those who do not use electronic cigarettes.”
Dr. Farber noted that there are other Food and Drug Administration–approved medications for treating tobacco addiction. In addition, the World Health Organization, American Medical Association, Centers for Disease Control and Prevention, and FDA have all advised that e-cigarettes should not be used as smoking cessation aids, he said, especially in light of current outbreak of life-threatening e-cigarette and vaping lung injuries currently being investigated by the CDC and FDA.
“These study results suggest that the CDC reports of e-cigarette, or vaping, product use–associated lung injury are likely to be just the tip of the iceberg,” he said. “Although the CDC has identified vitamin E acetate–containing products as an important culprit, it is unlikely to be the only one. There are many substances in the emissions of e-cigarettes that have known irritant and/or toxic effects on the airways.”
Dr. Bhatta and Dr. Glantz acknowledged several limitations in their analysis, including the possibility of recall bias, not distinguishing between nondaily and daily e-cigarette or combustible tobacco use, and combining respiratory conditions together to achieve adequate power. The study shows an association, but the mechanism by which e-cigarettes may contribute to the development of lung disease remains under investigation.
This study was supported by grants from the National Institute on Drug Abuse; the National Cancer Institute; the FDA Center for Tobacco Products; the National Heart, Lung, and Blood Institute; and the University of California, San Francisco Helen Diller Family Comprehensive Cancer Center Global Cancer Program. Dr. Bhatta and Dr. Glantz reported no relevant conflicts of interest.
SOURCE: Bhatta DN, Glantz SA. Am J Prev Med. 2019 Dec 16. doi: 10.1016/j.amepre.2019.07.028.
according to recent longitudinal analysis published in the American Journal of Preventive Medicine.
E-cigarettes have been promoted as a safer alternative to combustible tobacco, and until recently, there has been little and conflicting evidence by which to test this hypothesis. This study conducted by Dharma N. Bhatta, PhD, and Stanton A. Glantz, PhD, of the Center for Tobacco Control Research and Education at the University of California, San Francisco, is one of the first longitudinal examinations of e-cigarette use and controlling for combustible tobacco use.
Dr. Bhatta and Dr. Glantz performed a multivariable, logistic regression analysis of adults enrolled in the nationally representative, population-based, longitudinal Population Assessment of Tobacco and Health study. The researchers analyzed the tobacco use of adults in the study in three waves, following them through wave 1 (September 2013 to December 2014), wave 2 (October 2014 to October 2015), and wave 3 (October 2015 to October 2016), analyzing the data between 2018 and 2019. Overall, wave 1 began with 32,320 participants, and 15.1% of adults reported respiratory disease at baseline.
Lung or respiratory disease was assessed by asking participants whether they had been told by a health professional that they had chronic obstructive pulmonary disease, chronic bronchitis, emphysema, or asthma. The researchers defined e-cigarette and combustible tobacco use as participants who never, currently, or formerly used e-cigarettes or smoked combustible tobacco. Participants who indicated they used e-cigarettes or combustible tobacco frequently or infrequently were placed in the current-user group, while past users were those participants who said they used to, but no longer use e-cigarettes or combustible tobacco.
The results showed former e-cigarette use (adjusted odds ratio, 1.34; 95% confidence interval, 1.23-1.46) and current e-cigarette use (aOR, 1.32; 95% CI, 1.17-1.49) were associated with an increased risk of having incident respiratory disease.
The data showed a not unexpected statistically significant association between former combustible tobacco use (aOR, 1.29; 95% CI, 1.14-1.47) as well as current combustible tobacco use (aOR, 1.61; 95% CI, 1.42-1.82) and incident respiratory disease risk.
There was a statistically significant association between respiratory disease and former or current e-cigarette use for adults who did not have respiratory disease at baseline, after adjusting for factors such as current combustible tobacco use, clinical variables, and demographic differences. Participants in wave 1 who reported former (aOR, 1.31; 95% CI, 1.07-1.60) or current e-cigarette use (aOR, 1.29; 95% CI, 1.03-1.61) had a significantly higher risk of developing incident respiratory disease in subsequent waves. There was also a statistically significant association between use of combustible tobacco and subsequent respiratory disease in later waves of the study (aOR, 2.56; 95% CI, 1.92-3.41), which the researchers noted was independent of the usual risks associated with combustible tobacco.
The investigators also looked at the link between dual use of e-cigarettes and combustible tobacco and respiratory disease risk. “The much more common pattern is dual use, in which an e-cigarette user continues to smoke combusted tobacco products at the same time (93.7% of e-cigarette users at wave 2 and 91.2% at wave 3 also used combustible tobacco; 73.3% of e-cigarette users at wave 2 and 64.9% at wave 3 also smoked cigarettes),” they wrote.
The odds of developing respiratory disease for participants who used both e-cigarettes and combustible tobacco were 3.30, compared with a participant who never used e-cigarettes, with similar results seen when comparing e-cigarettes and cigarettes.
“Although switching from combustible tobacco, including cigarettes, to e-cigarettes theoretically could reduce the risk of developing respiratory disease, current evidence indicates a high prevalence of dual use, which is associated with in-creased risk beyond combustible tobacco use,” the investigators wrote.
Harold J. Farber, MD, FCCP, professor of pediatrics in the pulmonary section at Baylor College of Medicine and Texas Children’s Hospital, both in Houston, said in an interview that the increased respiratory risk among dual users, who are likely using e-cigarettes and combustible tobacco together as a way to quit smoking, is particularly concerning.
“There is substantial reason to be concerned about efficacy of electronic cigarette products. Real-world observational studies have shown that, on average, tobacco smokers who use electronic cigarettes are less likely to stop smoking than those who do not use electronic cigarettes,” he said. “People who have stopped tobacco smoking but use electronic cigarettes are more likely to relapse to tobacco smoking than those who do not use electronic cigarettes.”
Dr. Farber noted that there are other Food and Drug Administration–approved medications for treating tobacco addiction. In addition, the World Health Organization, American Medical Association, Centers for Disease Control and Prevention, and FDA have all advised that e-cigarettes should not be used as smoking cessation aids, he said, especially in light of current outbreak of life-threatening e-cigarette and vaping lung injuries currently being investigated by the CDC and FDA.
“These study results suggest that the CDC reports of e-cigarette, or vaping, product use–associated lung injury are likely to be just the tip of the iceberg,” he said. “Although the CDC has identified vitamin E acetate–containing products as an important culprit, it is unlikely to be the only one. There are many substances in the emissions of e-cigarettes that have known irritant and/or toxic effects on the airways.”
Dr. Bhatta and Dr. Glantz acknowledged several limitations in their analysis, including the possibility of recall bias, not distinguishing between nondaily and daily e-cigarette or combustible tobacco use, and combining respiratory conditions together to achieve adequate power. The study shows an association, but the mechanism by which e-cigarettes may contribute to the development of lung disease remains under investigation.
This study was supported by grants from the National Institute on Drug Abuse; the National Cancer Institute; the FDA Center for Tobacco Products; the National Heart, Lung, and Blood Institute; and the University of California, San Francisco Helen Diller Family Comprehensive Cancer Center Global Cancer Program. Dr. Bhatta and Dr. Glantz reported no relevant conflicts of interest.
SOURCE: Bhatta DN, Glantz SA. Am J Prev Med. 2019 Dec 16. doi: 10.1016/j.amepre.2019.07.028.
FROM THE AMERICAN JOURNAL OF PREVENTIVE MEDICINE
Early increase in flu activity shows no signs of slowing
An important measure of U.S. flu activity for the 2019-2020 season has already surpassed last season’s high, and more than half the states are experiencing high levels of activity, according to the Centers for Disease Control and Prevention.
reported Dec. 27.
The last time the outpatient visit rate was higher than that was in February of the 2017-2018 season, when it peaked at 7.5%. The peak month of flu activity occurs most often – about once every 3 years – in February, and the odds of a December peak are about one in five, the CDC has said.
Outpatient illness activity also increased at the state level during the week ending Dec. 21. There were 20 jurisdictions – 18 states, the District of Columbia, and Puerto Rico – at level 10 on the CDC’s 1-10 scale of activity, compared with 13 the previous week, and the number of jurisdictions in the “high” range (levels 8-10) jumped from 21 to 28, the CDC data show.
The influenza division estimated that there have been 4.6 million flu illnesses so far this season, nearly a million more than the total after last week, along with 39,000 hospitalizations. The overall hospitalization rate for the season is up to 6.6 per 100,000 population, which is about average at this point. The proportion of deaths attributed to pneumonia and influenza increased to 5.7%, which is below the epidemic threshold, the CDC said.
Three pediatric deaths related to influenza-like illness were reported during the week ending Dec. 21, two of which occurred in an earlier week. For the 2019-2020 season so far, a total of 22 pediatric deaths have been reported to the CDC.
An important measure of U.S. flu activity for the 2019-2020 season has already surpassed last season’s high, and more than half the states are experiencing high levels of activity, according to the Centers for Disease Control and Prevention.

reported Dec. 27.
The last time the outpatient visit rate was higher than that was in February of the 2017-2018 season, when it peaked at 7.5%. The peak month of flu activity occurs most often – about once every 3 years – in February, and the odds of a December peak are about one in five, the CDC has said.
Outpatient illness activity also increased at the state level during the week ending Dec. 21. There were 20 jurisdictions – 18 states, the District of Columbia, and Puerto Rico – at level 10 on the CDC’s 1-10 scale of activity, compared with 13 the previous week, and the number of jurisdictions in the “high” range (levels 8-10) jumped from 21 to 28, the CDC data show.
The influenza division estimated that there have been 4.6 million flu illnesses so far this season, nearly a million more than the total after last week, along with 39,000 hospitalizations. The overall hospitalization rate for the season is up to 6.6 per 100,000 population, which is about average at this point. The proportion of deaths attributed to pneumonia and influenza increased to 5.7%, which is below the epidemic threshold, the CDC said.
Three pediatric deaths related to influenza-like illness were reported during the week ending Dec. 21, two of which occurred in an earlier week. For the 2019-2020 season so far, a total of 22 pediatric deaths have been reported to the CDC.
An important measure of U.S. flu activity for the 2019-2020 season has already surpassed last season’s high, and more than half the states are experiencing high levels of activity, according to the Centers for Disease Control and Prevention.

reported Dec. 27.
The last time the outpatient visit rate was higher than that was in February of the 2017-2018 season, when it peaked at 7.5%. The peak month of flu activity occurs most often – about once every 3 years – in February, and the odds of a December peak are about one in five, the CDC has said.
Outpatient illness activity also increased at the state level during the week ending Dec. 21. There were 20 jurisdictions – 18 states, the District of Columbia, and Puerto Rico – at level 10 on the CDC’s 1-10 scale of activity, compared with 13 the previous week, and the number of jurisdictions in the “high” range (levels 8-10) jumped from 21 to 28, the CDC data show.
The influenza division estimated that there have been 4.6 million flu illnesses so far this season, nearly a million more than the total after last week, along with 39,000 hospitalizations. The overall hospitalization rate for the season is up to 6.6 per 100,000 population, which is about average at this point. The proportion of deaths attributed to pneumonia and influenza increased to 5.7%, which is below the epidemic threshold, the CDC said.
Three pediatric deaths related to influenza-like illness were reported during the week ending Dec. 21, two of which occurred in an earlier week. For the 2019-2020 season so far, a total of 22 pediatric deaths have been reported to the CDC.