User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
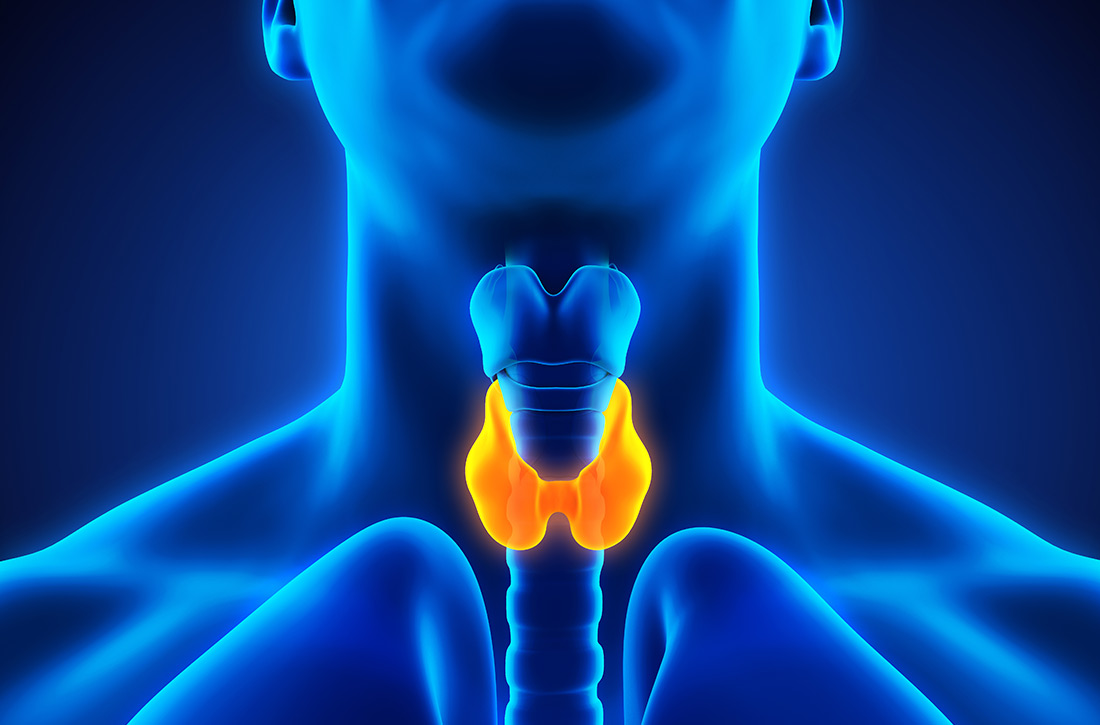
Refining your approach to hypothyroidism treatment
CASE
A 38-year-old woman presents for a routine physical. Other than urgent care visits for 1 episode of influenza and 2 upper respiratory illnesses, she has not seen a physician for a physical in 5 years. She denies any significant medical history. She takes naproxen occasionally for chronic right knee pain. She does not use tobacco or alcohol. Recently, she has started using a meal replacement shake at lunchtime for weight management. She performs aerobic exercise 30 to 40 minutes per day, 5 days per week. Her family history is significant for type 2 diabetes mellitus, arthritis, heart disease, and hyperlipidemia on her mother’s side. She is single, is not currently sexually active, works as a pharmacy technician, and has no children. A high-risk human papillomavirus test was normal 4 years ago.
A review of systems is notable for a 20-pound weight gain over the past year, worsening heartburn over the past 2 weeks, and chronic knee pain, which is greater in the right knee than the left. She denies weakness, fatigue, nausea, diarrhea, constipation, or abdominal pain. Vital signs reveal a blood pressure of 146/88 mm Hg, a heart rate of 63 bpm, a temperature of 98°F (36.7°C), a respiratory rate of 16, a height of 5’7’’ (1.7 m), a weight of 217 lbs (98.4 kg), and a peripheral capillary oxygen saturation (SpO2) of 99% on room air. The physical exam reveals a body mass index (BMI) of 34, warm dry skin, and coarse brittle hair.
Lab results reveal a thyroid-stimulating hormone (TSH) level of 11.17 mIU/L (reference range, 0.45-4.5 mIU/L) and a free thyroxine (T4) of 0.58 ng/dL (reference range, 0.8-2.8 ng/dL). A basic metabolic panel and hemoglobin A1C level are normal.
What would you recommend?
In the United States, the prevalence of overt hypothyroidism (defined as a TSH level > 4.5 mIU/L and a low free T4) among people ≥ 12 years of age was estimated at 0.3% based on National Health and Nutrition Examination Survey (NHANES) data from 1999-2002.1 Subclinical hypothyroidism (TSH level > 4.5 mIU/L but < 10 mIU/L and a normal T4 level) is even more common, with an estimated prevalence of 3.4%.1 Hypothyroidism is more common in females and occurs more frequently in Caucasian Americans and Mexican Americans than in African Americans.1
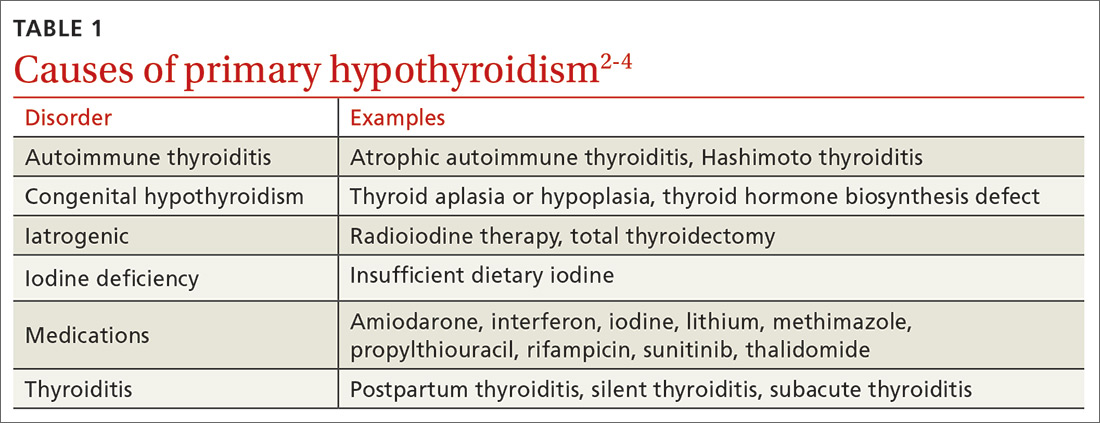
The most common etiologies of hypothyroidism include autoimmune thyroiditis (eg, Hashimoto thyroiditis, atrophic autoimmune thyroiditis) and iatrogenic causes (eg, after radioactive iodine ablation or thyroidectomy) (TABLE 1).2-4
Initiating thyroid hormone replacement
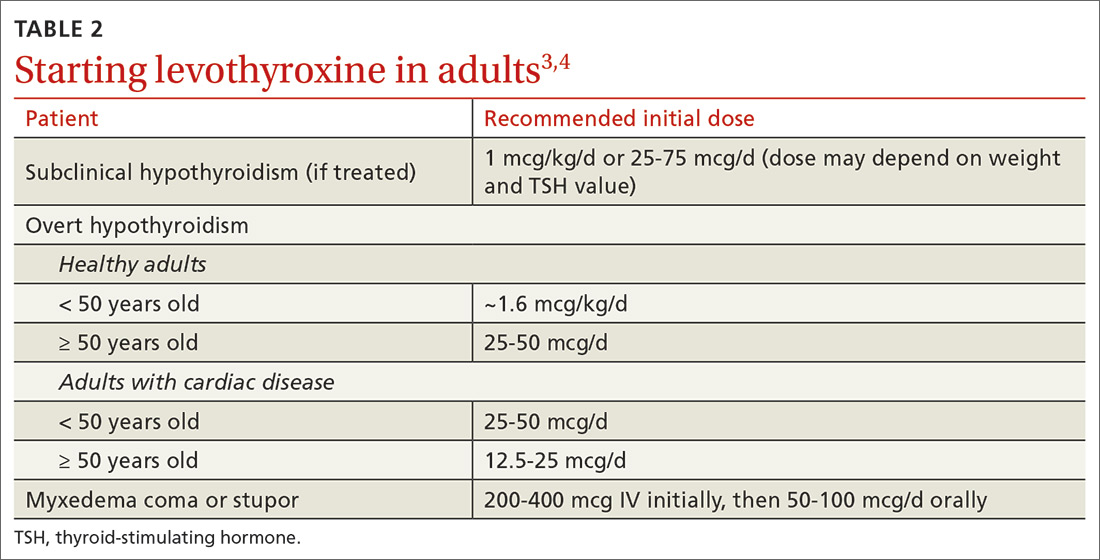
Factors to consider when starting a patient on thyroid hormone replacement include age, weight, symptom severity, TSH level, goal TSH value, adverse effects from thyroid supplements, history of cardiac disease, and, for women of child-bearing age, the desire for pregnancy vs the use of contraceptives. Most adult patients < 50 years with overt hypothyroidism can begin a weight-based dose of levothyroxine: ~1.6 mcg/kg/d (based on ideal body weight).3
Continue to: For adults with cardiac disease...
For adults with cardiac disease, the risk of over-replacement limits initial dosing to 25 to 50 mcg/d for patients < 50 years (12.5-25 mcg/d; ≥ 50 years).3 For adults with subclinical hypothyroidism, it is reasonable to begin therapy at a lower daily dose (eg, 25-75 mcg/d) depending on baseline TSH level, symptoms (the patient may be asymptomatic), and the presence of cardiac disease (TABLE 23,4). Consider treatment in patients with subclinical hypothyroidism particularly when patients have a goiter or dyslipidemia and in women contemplating pregnancy in the near future. Elderly patients may require a dose 20% to 25% lower than younger adults because of decreased body mass.3
Levothyroxine is considered first-line therapy for hypothyroidism because of its low cost, dose consistency, low risk of allergic reactions, and potential to cause fewer cardiac adverse effects than triiodothyronine (T3) products such as desiccated thyroid extract.5 Although data have not shown an absolute increase in cardiovascular adverse effects, T3 products have a higher T3 vs T4 ratio, giving them a theoretically increased risk.5,6 Desiccated thyroid extract also has been associated with allergic reactions.5
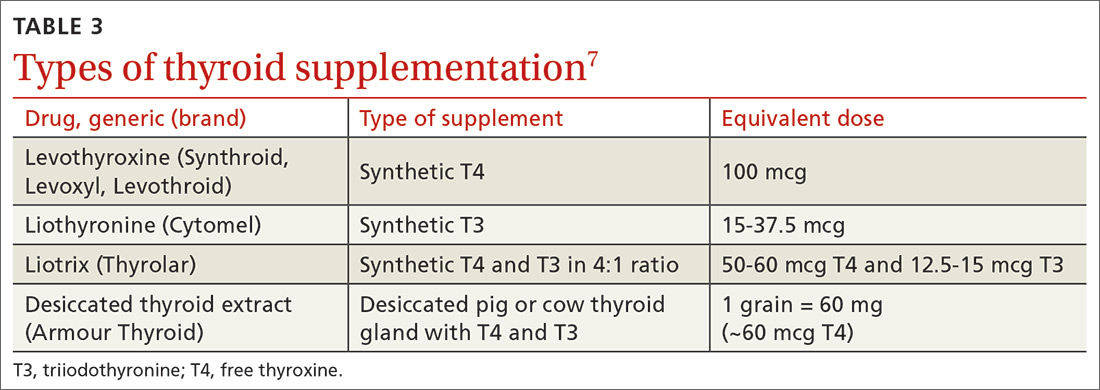
Use of liothyronine alone or in combination with levothyroxine lacks evidence and guideline support.4 Furthermore, it is dosed twice daily, which makes it less convenient, and concerns still exist that there may be an increase in cardiovascular adverse effects.4,6 See TABLE 37 for a summary of available products and their equivalent doses.
Maintaining patients on therapy
The maintenance phase begins once hypothyroidism is diagnosed and treatment is initiated. This phase includes regular monitoring with laboratory studies, office visits, and as-needed adjustments in hormone replacement dosing. The frequency at which all of these occur is variable and based on a number of factors including the patient’s other medical conditions, use of other medications including over-the-counter agents, the patient’s age, weight changes, and pregnancy status.3,4,8 In general, dosage adjustments of 12.5 to 25 mcg can be made at 6- to 8-week intervals based on repeat TSH measurements, patient symptoms, and comorbidities.3
Once a patient is symptomatically stable and laboratory values have normalized, the recommended frequency of laboratory evaluation and office visits is every 12 months, barring significant changes in any of the factors mentioned above. At each visit, physicians should perform medication (including supplements) reconciliation and discuss any health condition updates. Changes to the therapy plan, including frequency or timing of laboratory tests, may be necessary if patients begin taking medications that alter the absorption or function of levothyroxine (eg, steroids).
Continue to: To maximize absorption...
To maximize absorption, providers should review with patients the optimal way to take thyroid hormones. Levothyroxine is approximately 70% to 80% absorbed under ideal conditions, which means taking it in the morning at least 30 to 60 minutes before eating or 3 to 4 hours after the last meal of the day.3,9-13 Of note, TSH levels may increase slightly in patients taking proton pump inhibitors, but this does not usually require a dose increase of thyroid hormone.11 Given that some supplements, particularly iron and calcium, can interfere with absorption, it is recommended to maintain a 3- to 4-hour gap between taking those supplements and taking levothyroxine.12-14 For those patients unable or unwilling to adhere to these recommendations, an increase in levothyroxine dose may be required in order to compensate for the decreased absorption.
Don’t adjust hormone therapy based on clinical presentation alone. While clinical symptoms are important, it is not recommended to adjust hormone therapy based solely on clinical presentation. Common hypothyroid symptoms of dry skin, edema, weight gain, and fatigue may be caused by other medical conditions. While indices including Achilles reflex time and basal metabolic rate have shown some correlation to thyroid dysfunction, there has been limited evidence to show that longitudinal index changes reflect subtle changes in thyroid hormone levels.3
The most recent guidelines from the American Thyroid Association recommend that, “Symptoms should be followed, but considered in the context of serum thyrotropin values, relevant comorbidities, and other potential causes.”3
Special populations/circumstances to keep in mind
Malabsorption conditions. When a higher than expected weight-based dose of levothyroxine is required, physicians should review administration timing, adherence, and comorbid medical conditions that can affect absorption.
Several studies, for example, have demonstrated the impact of Helicobacter pylori gastritis on levothyroxine absorption and subsequent TSH levels.15-17 In one nonrandomized prospective study, patients with H pylori and hypothyroidism who were previously thought to be unresponsive to levothyroxine therapy had a decrease in average TSH level from 30.5 mIU/L to 4.2 mIU/L after H pylori was eradicated.15 Autoimmune atrophic gastritis and celiac disease, both of which are more common in those with other autoimmune diseases, are also associated with the need for higher than expected levothyroxine doses.17,18
Continue to: A history of gastric bypass surgery...
A history of gastric bypass surgery alone is not considered a risk factor for poor absorption of thyroid hormone, given that the majority of levothyroxine absorption occurs in the ileum.19,20 However, advancing age (> 70 years) and extreme obesity (BMI > 40) are independent risk factors for decreased levothyroxine absorption.20,21
Women of reproductive age and pregnant women. Overt untreated or undertreated hypothyroidism can be associated with increased risk of maternal and fetal complications including decreased fertility, miscarriage, preterm delivery, lower birth rates, and infant cognitive deficits.3,22 Therefore, the main focus should be optimization of thyroid hormone levels prior to and during pregnancy.3,4,8,22 Thyroid hormone replacement needs to be increased during pregnancy in approximately 50% to 85% of women using thyroid replacement prior to pregnancy, but the dose requirements vary based on the underlying etiology of thyroid dysfunction.
One initial option for patients on a stable dose before pregnancy is to increase their daily dose by a half tablet (1.5 × daily dose) immediately after home confirmation of pregnancy, until finer dose adjustments (usually increases of 25%-60% ) can be made by a physician. Experts recommend that a TSH level be obtained every 4 weeks until mid-gestation and then at least once around 30 weeks’ gestation to ensure specific targets are being met with dose adjustments.22 Optimal thyrotropin reference ranges during conception and pregnancy can be found in the literature.23
Patients who have positive antibodies and normal thyroid function tests. Patients who are screened for thyroid disorders may demonstrate normal thyroid function (ie, euthyroid) with TSH, free T4, and, if checked, free T3, all within normal ranges. Despite these normal lab results, patients may have additional test results that demonstrate positive thyroid autoantibodies including thyroglobulin antibodies and/or thyroid peroxidase antibodies. Thyroid autoimmunity itself has been associated with a range of other autoimmune conditions as well as an increased risk of thyroid cancer in those with Hashimoto thyroiditis.24 Two studies showed that prophylactic treatment of euthyroid patients with levothyroxine led to a reduction in antibody levels and a lower TSH level.25,26 However, no studies have focused on patient-oriented outcomes such as hospitalizations, quality of life, or symptoms. If the patient remains asymptomatic, we recommend no treatment, but that the patient’s TSH levels be monitored every 12 months.27
Elderly patients. Population data have shown that TSH increases normally with age, with a TSH level of 7.5 mIU/L being the upper limit of normal for a population of healthy adults > 80 years of age.28,29 Overall, studies have failed to show any benefit in treating elderly patients with subclinical hypothyroidism unless their TSH level exceeds 10 mIU/L.6,21 The one exception is elderly patients with heart failure in whom untreated subclinical hypothyroidism has been shown to be associated with higher mortality.30
Continue to: Elderly patients are at higher risk...
Elderly patients are at higher risk for adverse effects of thyroid over-replacement, including atrial fibrillation and osteoporosis. While there have been no randomized trials examining target TSH levels in this population, a reasonable recommendation is a goal TSH level of 4 to 6 mIU/L for elderly patients ≥ 70 years.4
CASE
As a result of the patient’s elevated TSH level and symptoms of hypothyroidism, you start levothyroxine 150 mcg/d by mouth, counsel her on potential adverse effects, and schedule a follow-up visit with another TSH check in 6 weeks.
Follow-up laboratory studies 6 weeks later reveal a TSH level of 5.86 mIU/L (reference range, 0.45-4.5 mIU/L) and a free T4 level of 0.74 ng/dL (reference range, 0.8-2.8 ng/dL). Based on those results, you increase the dose of levothyroxine to 175 mcg/d.
At her follow-up visit 12 weeks after initial presentation, her TSH level is 3.85 mIU/L. She reports feeling better overall with less fatigue, and she has lost 5 pounds since her last visit. You recommend she continue levothyroxine 175 mcg/d after reviewing medication compliance with the patient and ensuring she is indeed taking it in the morning, at least 30 minutes prior to eating. With improved but not resolved symptoms, she agrees to follow-up with repeat TSH laboratory studies in 6 weeks to determine whether further dose adjustments are necessary. Given that she is of reproductive age and her TSH level is suboptimal for pregnancy, you caution her about heightened pregnancy/fetal risks with a suboptimal TSH and recommend that she use reliable contraception.
CORRESPONDENCE
Christopher Bunt, MD, FAAFP, 5 Charleston Center Drive, Suite 263, MSC 192,Charleston, SC 29425; [email protected]
1. Aoki Y, Belin RM, Clickner R, et al. Serum TSH and total T4 in the United States population and their association with participant characteristics: National Health and Nutrition Examination Survey (NHANES 1999-2002). Thyroid. 2007;17:1211-1223.
2. Vaidya B, Pearce SH. Management of hypothyroidism in adults. BMJ. 2008;337:a801.
3. Garber JR, Cobin RH, Gharib H, et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18:988-1028.
4. Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid. 2014;24:1670-1751.
5. Toft AD. Thyroxine therapy. N Engl J Med. 1994;331:174-180.
6. Floriani C, Gencer B, Collet TH, et al. Subclinical thyroid dysfunction and cardiovascular diseases: 2016 update. Eur Heart J. 2018;39:503-507.
7. Lexi-Comp, Inc. (Lexi-Drugs®). https://online.lexi.com/lco/action/login. Accessed July 7, 2017.
8. Okosieme O, Gilbert J, Abraham P, et al. Management of primary hypothyroidism: statement by the British Thyroid Association Executive Committee. Clin Endocrinol (Oxf). 2016;84:799-808.
9. Fish LH, Schwartz HL, Cavanaugh J, et al. Replacement dose, metabolism, and bioavailability of levothyroxine in the treatment of hypothyroidism. Role of triiodothyronine in pituitary feedback in humans. N Engl J Med. 1987;316:764-770.
10. John-Kalarickal J, Pearlman G, Carlson HE. New medications which decrease levothyroxine absorption. Thyroid. 2007;17:763-765.
11. Sachmechi I, Reich DM, Aninyei M, et al. Effect of proton pump inhibitors on serum thyroid-stimulating hormone level in euthyroid patients treated with levothyroxine for hypothyroidism. Endocr Pract. 2007;13:345-349.
12. Sperber AD, Liel Y. Evidence for interference with the intestinal absorption of levothyroxine sodium by aluminum hydroxide. Arch Intern Med. 1992;152:183-184.
13. Zamfirescu I, Carlson HE. Absorption of levothyroxine when coadministered with various calcium formulations. Thyroid. 2011;21:483-486.
14. Campbell NR, Hasinoff BB, Stalts H, et al. Ferrous sulfate reduces thyroxine efficacy in patients with hypothyroidism. Ann Intern Med. 1992;117:1010-1013.
15. Bugdaci MS, Zuhur SS, Sokmen M, et al. The role of Helicobacter pylori in patients with hypothyroidism in whom could not be achieved normal thyrotropin levels despite treatment with high doses of thyroxine. Helicobacter. 2011;16:124-130.
16. Centanni M, Gargano L, Canettieri G, et al. Thyroxine in goiter, Helicobacter pylori infection, and chronic gastritis. N Engl J Med. 2006;354:1787-1795.
17. Centanni M, Marignani M, Gargano L, et al. Atrophic body gastritis in patients with autoimmune thyroid disease: an underdiagnosed association. Arch Intern Med. 1999;159:1726-1730.
18. Collins D, Wilcox R, Nathan M, et al. Celiac disease and hypothyroidism. Am J Med. 2012;125:278-282.
19. Azizi F, Belur R, Albano J. Malabsorption of thyroid hormones after jejunoileal bypass for obesity. Ann Intern Med. 1979;90:941-942.
20. Gkotsina M, Michalaki M, Mamali I, et al. Improved levothyroxine pharmacokinetics after bariatric surgery. Thyroid. 2013;23:414-419.
21. Hennessey JV, Espaillat R. Diagnosis and management of subclinical hypothyroidism in elderly adults: a review of the literature. J Am Geriatr Soc. 2015;63:1663-1673.
22. Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid. 2017;27:315-389.
23. Carney LA, Quinlan JD, West JM. Thyroid disease in pregnancy. Am Fam Physician. 2014;89:273-278.
24. Fröhlich E, Wahl R. Thyroid autoimmunity: role of anti-thyroid antibodies in thyroid and extra-thyroidal diseases. Front Immunol. 2017;8:521.
25. Aksoy DY, Kerimoglu U, Okur H, et al. Effects of prophylactic thyroid hormone replacement in euthyroid Hashimoto’s thyroiditis. Endocr J. 2005;52:337-343.
26. Padberg S, Heller K, Usadel KH, et al. One-year prophylactic treatment of euthyroid Hashimoto’s thyroiditis patients with levothyroxine: is there a benefit? Thyroid. 2001;11:249-255.
27. Rugge B, Balshem H, Sehgal R, et al. Screening and Treatment of Subclinical Hypothyroidism or Hyperthyroidism [Internet]. Comparative Effectiveness Reviews, No. 24. Rockville, MD: Agency for Healthcare Research and Quality; October 2011. www.ncbi.nlm.nih.gov/books/NBK83492/. Accessed February 21, 2020.
28. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87:489-499.
29. Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92:4575-4582.
30. Pasqualetti G, Tognini S, Polini A, et al. Is subclinical hypothyroidism a cardiovascular risk factor in the elderly? J Clin Endocrinol Metab. 2013;98:2256-2266.
CASE
A 38-year-old woman presents for a routine physical. Other than urgent care visits for 1 episode of influenza and 2 upper respiratory illnesses, she has not seen a physician for a physical in 5 years. She denies any significant medical history. She takes naproxen occasionally for chronic right knee pain. She does not use tobacco or alcohol. Recently, she has started using a meal replacement shake at lunchtime for weight management. She performs aerobic exercise 30 to 40 minutes per day, 5 days per week. Her family history is significant for type 2 diabetes mellitus, arthritis, heart disease, and hyperlipidemia on her mother’s side. She is single, is not currently sexually active, works as a pharmacy technician, and has no children. A high-risk human papillomavirus test was normal 4 years ago.
A review of systems is notable for a 20-pound weight gain over the past year, worsening heartburn over the past 2 weeks, and chronic knee pain, which is greater in the right knee than the left. She denies weakness, fatigue, nausea, diarrhea, constipation, or abdominal pain. Vital signs reveal a blood pressure of 146/88 mm Hg, a heart rate of 63 bpm, a temperature of 98°F (36.7°C), a respiratory rate of 16, a height of 5’7’’ (1.7 m), a weight of 217 lbs (98.4 kg), and a peripheral capillary oxygen saturation (SpO2) of 99% on room air. The physical exam reveals a body mass index (BMI) of 34, warm dry skin, and coarse brittle hair.
Lab results reveal a thyroid-stimulating hormone (TSH) level of 11.17 mIU/L (reference range, 0.45-4.5 mIU/L) and a free thyroxine (T4) of 0.58 ng/dL (reference range, 0.8-2.8 ng/dL). A basic metabolic panel and hemoglobin A1C level are normal.
What would you recommend?
In the United States, the prevalence of overt hypothyroidism (defined as a TSH level > 4.5 mIU/L and a low free T4) among people ≥ 12 years of age was estimated at 0.3% based on National Health and Nutrition Examination Survey (NHANES) data from 1999-2002.1 Subclinical hypothyroidism (TSH level > 4.5 mIU/L but < 10 mIU/L and a normal T4 level) is even more common, with an estimated prevalence of 3.4%.1 Hypothyroidism is more common in females and occurs more frequently in Caucasian Americans and Mexican Americans than in African Americans.1
The most common etiologies of hypothyroidism include autoimmune thyroiditis (eg, Hashimoto thyroiditis, atrophic autoimmune thyroiditis) and iatrogenic causes (eg, after radioactive iodine ablation or thyroidectomy) (TABLE 1).2-4

Initiating thyroid hormone replacement
Factors to consider when starting a patient on thyroid hormone replacement include age, weight, symptom severity, TSH level, goal TSH value, adverse effects from thyroid supplements, history of cardiac disease, and, for women of child-bearing age, the desire for pregnancy vs the use of contraceptives. Most adult patients < 50 years with overt hypothyroidism can begin a weight-based dose of levothyroxine: ~1.6 mcg/kg/d (based on ideal body weight).3
Continue to: For adults with cardiac disease...
For adults with cardiac disease, the risk of over-replacement limits initial dosing to 25 to 50 mcg/d for patients < 50 years (12.5-25 mcg/d; ≥ 50 years).3 For adults with subclinical hypothyroidism, it is reasonable to begin therapy at a lower daily dose (eg, 25-75 mcg/d) depending on baseline TSH level, symptoms (the patient may be asymptomatic), and the presence of cardiac disease (TABLE 23,4). Consider treatment in patients with subclinical hypothyroidism particularly when patients have a goiter or dyslipidemia and in women contemplating pregnancy in the near future. Elderly patients may require a dose 20% to 25% lower than younger adults because of decreased body mass.3

Levothyroxine is considered first-line therapy for hypothyroidism because of its low cost, dose consistency, low risk of allergic reactions, and potential to cause fewer cardiac adverse effects than triiodothyronine (T3) products such as desiccated thyroid extract.5 Although data have not shown an absolute increase in cardiovascular adverse effects, T3 products have a higher T3 vs T4 ratio, giving them a theoretically increased risk.5,6 Desiccated thyroid extract also has been associated with allergic reactions.5
Use of liothyronine alone or in combination with levothyroxine lacks evidence and guideline support.4 Furthermore, it is dosed twice daily, which makes it less convenient, and concerns still exist that there may be an increase in cardiovascular adverse effects.4,6 See TABLE 37 for a summary of available products and their equivalent doses.

Maintaining patients on therapy
The maintenance phase begins once hypothyroidism is diagnosed and treatment is initiated. This phase includes regular monitoring with laboratory studies, office visits, and as-needed adjustments in hormone replacement dosing. The frequency at which all of these occur is variable and based on a number of factors including the patient’s other medical conditions, use of other medications including over-the-counter agents, the patient’s age, weight changes, and pregnancy status.3,4,8 In general, dosage adjustments of 12.5 to 25 mcg can be made at 6- to 8-week intervals based on repeat TSH measurements, patient symptoms, and comorbidities.3
Once a patient is symptomatically stable and laboratory values have normalized, the recommended frequency of laboratory evaluation and office visits is every 12 months, barring significant changes in any of the factors mentioned above. At each visit, physicians should perform medication (including supplements) reconciliation and discuss any health condition updates. Changes to the therapy plan, including frequency or timing of laboratory tests, may be necessary if patients begin taking medications that alter the absorption or function of levothyroxine (eg, steroids).
Continue to: To maximize absorption...
To maximize absorption, providers should review with patients the optimal way to take thyroid hormones. Levothyroxine is approximately 70% to 80% absorbed under ideal conditions, which means taking it in the morning at least 30 to 60 minutes before eating or 3 to 4 hours after the last meal of the day.3,9-13 Of note, TSH levels may increase slightly in patients taking proton pump inhibitors, but this does not usually require a dose increase of thyroid hormone.11 Given that some supplements, particularly iron and calcium, can interfere with absorption, it is recommended to maintain a 3- to 4-hour gap between taking those supplements and taking levothyroxine.12-14 For those patients unable or unwilling to adhere to these recommendations, an increase in levothyroxine dose may be required in order to compensate for the decreased absorption.
Don’t adjust hormone therapy based on clinical presentation alone. While clinical symptoms are important, it is not recommended to adjust hormone therapy based solely on clinical presentation. Common hypothyroid symptoms of dry skin, edema, weight gain, and fatigue may be caused by other medical conditions. While indices including Achilles reflex time and basal metabolic rate have shown some correlation to thyroid dysfunction, there has been limited evidence to show that longitudinal index changes reflect subtle changes in thyroid hormone levels.3
The most recent guidelines from the American Thyroid Association recommend that, “Symptoms should be followed, but considered in the context of serum thyrotropin values, relevant comorbidities, and other potential causes.”3
Special populations/circumstances to keep in mind
Malabsorption conditions. When a higher than expected weight-based dose of levothyroxine is required, physicians should review administration timing, adherence, and comorbid medical conditions that can affect absorption.
Several studies, for example, have demonstrated the impact of Helicobacter pylori gastritis on levothyroxine absorption and subsequent TSH levels.15-17 In one nonrandomized prospective study, patients with H pylori and hypothyroidism who were previously thought to be unresponsive to levothyroxine therapy had a decrease in average TSH level from 30.5 mIU/L to 4.2 mIU/L after H pylori was eradicated.15 Autoimmune atrophic gastritis and celiac disease, both of which are more common in those with other autoimmune diseases, are also associated with the need for higher than expected levothyroxine doses.17,18
Continue to: A history of gastric bypass surgery...
A history of gastric bypass surgery alone is not considered a risk factor for poor absorption of thyroid hormone, given that the majority of levothyroxine absorption occurs in the ileum.19,20 However, advancing age (> 70 years) and extreme obesity (BMI > 40) are independent risk factors for decreased levothyroxine absorption.20,21
Women of reproductive age and pregnant women. Overt untreated or undertreated hypothyroidism can be associated with increased risk of maternal and fetal complications including decreased fertility, miscarriage, preterm delivery, lower birth rates, and infant cognitive deficits.3,22 Therefore, the main focus should be optimization of thyroid hormone levels prior to and during pregnancy.3,4,8,22 Thyroid hormone replacement needs to be increased during pregnancy in approximately 50% to 85% of women using thyroid replacement prior to pregnancy, but the dose requirements vary based on the underlying etiology of thyroid dysfunction.
One initial option for patients on a stable dose before pregnancy is to increase their daily dose by a half tablet (1.5 × daily dose) immediately after home confirmation of pregnancy, until finer dose adjustments (usually increases of 25%-60% ) can be made by a physician. Experts recommend that a TSH level be obtained every 4 weeks until mid-gestation and then at least once around 30 weeks’ gestation to ensure specific targets are being met with dose adjustments.22 Optimal thyrotropin reference ranges during conception and pregnancy can be found in the literature.23
Patients who have positive antibodies and normal thyroid function tests. Patients who are screened for thyroid disorders may demonstrate normal thyroid function (ie, euthyroid) with TSH, free T4, and, if checked, free T3, all within normal ranges. Despite these normal lab results, patients may have additional test results that demonstrate positive thyroid autoantibodies including thyroglobulin antibodies and/or thyroid peroxidase antibodies. Thyroid autoimmunity itself has been associated with a range of other autoimmune conditions as well as an increased risk of thyroid cancer in those with Hashimoto thyroiditis.24 Two studies showed that prophylactic treatment of euthyroid patients with levothyroxine led to a reduction in antibody levels and a lower TSH level.25,26 However, no studies have focused on patient-oriented outcomes such as hospitalizations, quality of life, or symptoms. If the patient remains asymptomatic, we recommend no treatment, but that the patient’s TSH levels be monitored every 12 months.27
Elderly patients. Population data have shown that TSH increases normally with age, with a TSH level of 7.5 mIU/L being the upper limit of normal for a population of healthy adults > 80 years of age.28,29 Overall, studies have failed to show any benefit in treating elderly patients with subclinical hypothyroidism unless their TSH level exceeds 10 mIU/L.6,21 The one exception is elderly patients with heart failure in whom untreated subclinical hypothyroidism has been shown to be associated with higher mortality.30
Continue to: Elderly patients are at higher risk...
Elderly patients are at higher risk for adverse effects of thyroid over-replacement, including atrial fibrillation and osteoporosis. While there have been no randomized trials examining target TSH levels in this population, a reasonable recommendation is a goal TSH level of 4 to 6 mIU/L for elderly patients ≥ 70 years.4
CASE
As a result of the patient’s elevated TSH level and symptoms of hypothyroidism, you start levothyroxine 150 mcg/d by mouth, counsel her on potential adverse effects, and schedule a follow-up visit with another TSH check in 6 weeks.
Follow-up laboratory studies 6 weeks later reveal a TSH level of 5.86 mIU/L (reference range, 0.45-4.5 mIU/L) and a free T4 level of 0.74 ng/dL (reference range, 0.8-2.8 ng/dL). Based on those results, you increase the dose of levothyroxine to 175 mcg/d.
At her follow-up visit 12 weeks after initial presentation, her TSH level is 3.85 mIU/L. She reports feeling better overall with less fatigue, and she has lost 5 pounds since her last visit. You recommend she continue levothyroxine 175 mcg/d after reviewing medication compliance with the patient and ensuring she is indeed taking it in the morning, at least 30 minutes prior to eating. With improved but not resolved symptoms, she agrees to follow-up with repeat TSH laboratory studies in 6 weeks to determine whether further dose adjustments are necessary. Given that she is of reproductive age and her TSH level is suboptimal for pregnancy, you caution her about heightened pregnancy/fetal risks with a suboptimal TSH and recommend that she use reliable contraception.
CORRESPONDENCE
Christopher Bunt, MD, FAAFP, 5 Charleston Center Drive, Suite 263, MSC 192,Charleston, SC 29425; [email protected]
CASE
A 38-year-old woman presents for a routine physical. Other than urgent care visits for 1 episode of influenza and 2 upper respiratory illnesses, she has not seen a physician for a physical in 5 years. She denies any significant medical history. She takes naproxen occasionally for chronic right knee pain. She does not use tobacco or alcohol. Recently, she has started using a meal replacement shake at lunchtime for weight management. She performs aerobic exercise 30 to 40 minutes per day, 5 days per week. Her family history is significant for type 2 diabetes mellitus, arthritis, heart disease, and hyperlipidemia on her mother’s side. She is single, is not currently sexually active, works as a pharmacy technician, and has no children. A high-risk human papillomavirus test was normal 4 years ago.
A review of systems is notable for a 20-pound weight gain over the past year, worsening heartburn over the past 2 weeks, and chronic knee pain, which is greater in the right knee than the left. She denies weakness, fatigue, nausea, diarrhea, constipation, or abdominal pain. Vital signs reveal a blood pressure of 146/88 mm Hg, a heart rate of 63 bpm, a temperature of 98°F (36.7°C), a respiratory rate of 16, a height of 5’7’’ (1.7 m), a weight of 217 lbs (98.4 kg), and a peripheral capillary oxygen saturation (SpO2) of 99% on room air. The physical exam reveals a body mass index (BMI) of 34, warm dry skin, and coarse brittle hair.
Lab results reveal a thyroid-stimulating hormone (TSH) level of 11.17 mIU/L (reference range, 0.45-4.5 mIU/L) and a free thyroxine (T4) of 0.58 ng/dL (reference range, 0.8-2.8 ng/dL). A basic metabolic panel and hemoglobin A1C level are normal.
What would you recommend?
In the United States, the prevalence of overt hypothyroidism (defined as a TSH level > 4.5 mIU/L and a low free T4) among people ≥ 12 years of age was estimated at 0.3% based on National Health and Nutrition Examination Survey (NHANES) data from 1999-2002.1 Subclinical hypothyroidism (TSH level > 4.5 mIU/L but < 10 mIU/L and a normal T4 level) is even more common, with an estimated prevalence of 3.4%.1 Hypothyroidism is more common in females and occurs more frequently in Caucasian Americans and Mexican Americans than in African Americans.1
The most common etiologies of hypothyroidism include autoimmune thyroiditis (eg, Hashimoto thyroiditis, atrophic autoimmune thyroiditis) and iatrogenic causes (eg, after radioactive iodine ablation or thyroidectomy) (TABLE 1).2-4

Initiating thyroid hormone replacement
Factors to consider when starting a patient on thyroid hormone replacement include age, weight, symptom severity, TSH level, goal TSH value, adverse effects from thyroid supplements, history of cardiac disease, and, for women of child-bearing age, the desire for pregnancy vs the use of contraceptives. Most adult patients < 50 years with overt hypothyroidism can begin a weight-based dose of levothyroxine: ~1.6 mcg/kg/d (based on ideal body weight).3
Continue to: For adults with cardiac disease...
For adults with cardiac disease, the risk of over-replacement limits initial dosing to 25 to 50 mcg/d for patients < 50 years (12.5-25 mcg/d; ≥ 50 years).3 For adults with subclinical hypothyroidism, it is reasonable to begin therapy at a lower daily dose (eg, 25-75 mcg/d) depending on baseline TSH level, symptoms (the patient may be asymptomatic), and the presence of cardiac disease (TABLE 23,4). Consider treatment in patients with subclinical hypothyroidism particularly when patients have a goiter or dyslipidemia and in women contemplating pregnancy in the near future. Elderly patients may require a dose 20% to 25% lower than younger adults because of decreased body mass.3

Levothyroxine is considered first-line therapy for hypothyroidism because of its low cost, dose consistency, low risk of allergic reactions, and potential to cause fewer cardiac adverse effects than triiodothyronine (T3) products such as desiccated thyroid extract.5 Although data have not shown an absolute increase in cardiovascular adverse effects, T3 products have a higher T3 vs T4 ratio, giving them a theoretically increased risk.5,6 Desiccated thyroid extract also has been associated with allergic reactions.5
Use of liothyronine alone or in combination with levothyroxine lacks evidence and guideline support.4 Furthermore, it is dosed twice daily, which makes it less convenient, and concerns still exist that there may be an increase in cardiovascular adverse effects.4,6 See TABLE 37 for a summary of available products and their equivalent doses.

Maintaining patients on therapy
The maintenance phase begins once hypothyroidism is diagnosed and treatment is initiated. This phase includes regular monitoring with laboratory studies, office visits, and as-needed adjustments in hormone replacement dosing. The frequency at which all of these occur is variable and based on a number of factors including the patient’s other medical conditions, use of other medications including over-the-counter agents, the patient’s age, weight changes, and pregnancy status.3,4,8 In general, dosage adjustments of 12.5 to 25 mcg can be made at 6- to 8-week intervals based on repeat TSH measurements, patient symptoms, and comorbidities.3
Once a patient is symptomatically stable and laboratory values have normalized, the recommended frequency of laboratory evaluation and office visits is every 12 months, barring significant changes in any of the factors mentioned above. At each visit, physicians should perform medication (including supplements) reconciliation and discuss any health condition updates. Changes to the therapy plan, including frequency or timing of laboratory tests, may be necessary if patients begin taking medications that alter the absorption or function of levothyroxine (eg, steroids).
Continue to: To maximize absorption...
To maximize absorption, providers should review with patients the optimal way to take thyroid hormones. Levothyroxine is approximately 70% to 80% absorbed under ideal conditions, which means taking it in the morning at least 30 to 60 minutes before eating or 3 to 4 hours after the last meal of the day.3,9-13 Of note, TSH levels may increase slightly in patients taking proton pump inhibitors, but this does not usually require a dose increase of thyroid hormone.11 Given that some supplements, particularly iron and calcium, can interfere with absorption, it is recommended to maintain a 3- to 4-hour gap between taking those supplements and taking levothyroxine.12-14 For those patients unable or unwilling to adhere to these recommendations, an increase in levothyroxine dose may be required in order to compensate for the decreased absorption.
Don’t adjust hormone therapy based on clinical presentation alone. While clinical symptoms are important, it is not recommended to adjust hormone therapy based solely on clinical presentation. Common hypothyroid symptoms of dry skin, edema, weight gain, and fatigue may be caused by other medical conditions. While indices including Achilles reflex time and basal metabolic rate have shown some correlation to thyroid dysfunction, there has been limited evidence to show that longitudinal index changes reflect subtle changes in thyroid hormone levels.3
The most recent guidelines from the American Thyroid Association recommend that, “Symptoms should be followed, but considered in the context of serum thyrotropin values, relevant comorbidities, and other potential causes.”3
Special populations/circumstances to keep in mind
Malabsorption conditions. When a higher than expected weight-based dose of levothyroxine is required, physicians should review administration timing, adherence, and comorbid medical conditions that can affect absorption.
Several studies, for example, have demonstrated the impact of Helicobacter pylori gastritis on levothyroxine absorption and subsequent TSH levels.15-17 In one nonrandomized prospective study, patients with H pylori and hypothyroidism who were previously thought to be unresponsive to levothyroxine therapy had a decrease in average TSH level from 30.5 mIU/L to 4.2 mIU/L after H pylori was eradicated.15 Autoimmune atrophic gastritis and celiac disease, both of which are more common in those with other autoimmune diseases, are also associated with the need for higher than expected levothyroxine doses.17,18
Continue to: A history of gastric bypass surgery...
A history of gastric bypass surgery alone is not considered a risk factor for poor absorption of thyroid hormone, given that the majority of levothyroxine absorption occurs in the ileum.19,20 However, advancing age (> 70 years) and extreme obesity (BMI > 40) are independent risk factors for decreased levothyroxine absorption.20,21
Women of reproductive age and pregnant women. Overt untreated or undertreated hypothyroidism can be associated with increased risk of maternal and fetal complications including decreased fertility, miscarriage, preterm delivery, lower birth rates, and infant cognitive deficits.3,22 Therefore, the main focus should be optimization of thyroid hormone levels prior to and during pregnancy.3,4,8,22 Thyroid hormone replacement needs to be increased during pregnancy in approximately 50% to 85% of women using thyroid replacement prior to pregnancy, but the dose requirements vary based on the underlying etiology of thyroid dysfunction.
One initial option for patients on a stable dose before pregnancy is to increase their daily dose by a half tablet (1.5 × daily dose) immediately after home confirmation of pregnancy, until finer dose adjustments (usually increases of 25%-60% ) can be made by a physician. Experts recommend that a TSH level be obtained every 4 weeks until mid-gestation and then at least once around 30 weeks’ gestation to ensure specific targets are being met with dose adjustments.22 Optimal thyrotropin reference ranges during conception and pregnancy can be found in the literature.23
Patients who have positive antibodies and normal thyroid function tests. Patients who are screened for thyroid disorders may demonstrate normal thyroid function (ie, euthyroid) with TSH, free T4, and, if checked, free T3, all within normal ranges. Despite these normal lab results, patients may have additional test results that demonstrate positive thyroid autoantibodies including thyroglobulin antibodies and/or thyroid peroxidase antibodies. Thyroid autoimmunity itself has been associated with a range of other autoimmune conditions as well as an increased risk of thyroid cancer in those with Hashimoto thyroiditis.24 Two studies showed that prophylactic treatment of euthyroid patients with levothyroxine led to a reduction in antibody levels and a lower TSH level.25,26 However, no studies have focused on patient-oriented outcomes such as hospitalizations, quality of life, or symptoms. If the patient remains asymptomatic, we recommend no treatment, but that the patient’s TSH levels be monitored every 12 months.27
Elderly patients. Population data have shown that TSH increases normally with age, with a TSH level of 7.5 mIU/L being the upper limit of normal for a population of healthy adults > 80 years of age.28,29 Overall, studies have failed to show any benefit in treating elderly patients with subclinical hypothyroidism unless their TSH level exceeds 10 mIU/L.6,21 The one exception is elderly patients with heart failure in whom untreated subclinical hypothyroidism has been shown to be associated with higher mortality.30
Continue to: Elderly patients are at higher risk...
Elderly patients are at higher risk for adverse effects of thyroid over-replacement, including atrial fibrillation and osteoporosis. While there have been no randomized trials examining target TSH levels in this population, a reasonable recommendation is a goal TSH level of 4 to 6 mIU/L for elderly patients ≥ 70 years.4
CASE
As a result of the patient’s elevated TSH level and symptoms of hypothyroidism, you start levothyroxine 150 mcg/d by mouth, counsel her on potential adverse effects, and schedule a follow-up visit with another TSH check in 6 weeks.
Follow-up laboratory studies 6 weeks later reveal a TSH level of 5.86 mIU/L (reference range, 0.45-4.5 mIU/L) and a free T4 level of 0.74 ng/dL (reference range, 0.8-2.8 ng/dL). Based on those results, you increase the dose of levothyroxine to 175 mcg/d.
At her follow-up visit 12 weeks after initial presentation, her TSH level is 3.85 mIU/L. She reports feeling better overall with less fatigue, and she has lost 5 pounds since her last visit. You recommend she continue levothyroxine 175 mcg/d after reviewing medication compliance with the patient and ensuring she is indeed taking it in the morning, at least 30 minutes prior to eating. With improved but not resolved symptoms, she agrees to follow-up with repeat TSH laboratory studies in 6 weeks to determine whether further dose adjustments are necessary. Given that she is of reproductive age and her TSH level is suboptimal for pregnancy, you caution her about heightened pregnancy/fetal risks with a suboptimal TSH and recommend that she use reliable contraception.
CORRESPONDENCE
Christopher Bunt, MD, FAAFP, 5 Charleston Center Drive, Suite 263, MSC 192,Charleston, SC 29425; [email protected]
1. Aoki Y, Belin RM, Clickner R, et al. Serum TSH and total T4 in the United States population and their association with participant characteristics: National Health and Nutrition Examination Survey (NHANES 1999-2002). Thyroid. 2007;17:1211-1223.
2. Vaidya B, Pearce SH. Management of hypothyroidism in adults. BMJ. 2008;337:a801.
3. Garber JR, Cobin RH, Gharib H, et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18:988-1028.
4. Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid. 2014;24:1670-1751.
5. Toft AD. Thyroxine therapy. N Engl J Med. 1994;331:174-180.
6. Floriani C, Gencer B, Collet TH, et al. Subclinical thyroid dysfunction and cardiovascular diseases: 2016 update. Eur Heart J. 2018;39:503-507.
7. Lexi-Comp, Inc. (Lexi-Drugs®). https://online.lexi.com/lco/action/login. Accessed July 7, 2017.
8. Okosieme O, Gilbert J, Abraham P, et al. Management of primary hypothyroidism: statement by the British Thyroid Association Executive Committee. Clin Endocrinol (Oxf). 2016;84:799-808.
9. Fish LH, Schwartz HL, Cavanaugh J, et al. Replacement dose, metabolism, and bioavailability of levothyroxine in the treatment of hypothyroidism. Role of triiodothyronine in pituitary feedback in humans. N Engl J Med. 1987;316:764-770.
10. John-Kalarickal J, Pearlman G, Carlson HE. New medications which decrease levothyroxine absorption. Thyroid. 2007;17:763-765.
11. Sachmechi I, Reich DM, Aninyei M, et al. Effect of proton pump inhibitors on serum thyroid-stimulating hormone level in euthyroid patients treated with levothyroxine for hypothyroidism. Endocr Pract. 2007;13:345-349.
12. Sperber AD, Liel Y. Evidence for interference with the intestinal absorption of levothyroxine sodium by aluminum hydroxide. Arch Intern Med. 1992;152:183-184.
13. Zamfirescu I, Carlson HE. Absorption of levothyroxine when coadministered with various calcium formulations. Thyroid. 2011;21:483-486.
14. Campbell NR, Hasinoff BB, Stalts H, et al. Ferrous sulfate reduces thyroxine efficacy in patients with hypothyroidism. Ann Intern Med. 1992;117:1010-1013.
15. Bugdaci MS, Zuhur SS, Sokmen M, et al. The role of Helicobacter pylori in patients with hypothyroidism in whom could not be achieved normal thyrotropin levels despite treatment with high doses of thyroxine. Helicobacter. 2011;16:124-130.
16. Centanni M, Gargano L, Canettieri G, et al. Thyroxine in goiter, Helicobacter pylori infection, and chronic gastritis. N Engl J Med. 2006;354:1787-1795.
17. Centanni M, Marignani M, Gargano L, et al. Atrophic body gastritis in patients with autoimmune thyroid disease: an underdiagnosed association. Arch Intern Med. 1999;159:1726-1730.
18. Collins D, Wilcox R, Nathan M, et al. Celiac disease and hypothyroidism. Am J Med. 2012;125:278-282.
19. Azizi F, Belur R, Albano J. Malabsorption of thyroid hormones after jejunoileal bypass for obesity. Ann Intern Med. 1979;90:941-942.
20. Gkotsina M, Michalaki M, Mamali I, et al. Improved levothyroxine pharmacokinetics after bariatric surgery. Thyroid. 2013;23:414-419.
21. Hennessey JV, Espaillat R. Diagnosis and management of subclinical hypothyroidism in elderly adults: a review of the literature. J Am Geriatr Soc. 2015;63:1663-1673.
22. Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid. 2017;27:315-389.
23. Carney LA, Quinlan JD, West JM. Thyroid disease in pregnancy. Am Fam Physician. 2014;89:273-278.
24. Fröhlich E, Wahl R. Thyroid autoimmunity: role of anti-thyroid antibodies in thyroid and extra-thyroidal diseases. Front Immunol. 2017;8:521.
25. Aksoy DY, Kerimoglu U, Okur H, et al. Effects of prophylactic thyroid hormone replacement in euthyroid Hashimoto’s thyroiditis. Endocr J. 2005;52:337-343.
26. Padberg S, Heller K, Usadel KH, et al. One-year prophylactic treatment of euthyroid Hashimoto’s thyroiditis patients with levothyroxine: is there a benefit? Thyroid. 2001;11:249-255.
27. Rugge B, Balshem H, Sehgal R, et al. Screening and Treatment of Subclinical Hypothyroidism or Hyperthyroidism [Internet]. Comparative Effectiveness Reviews, No. 24. Rockville, MD: Agency for Healthcare Research and Quality; October 2011. www.ncbi.nlm.nih.gov/books/NBK83492/. Accessed February 21, 2020.
28. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87:489-499.
29. Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92:4575-4582.
30. Pasqualetti G, Tognini S, Polini A, et al. Is subclinical hypothyroidism a cardiovascular risk factor in the elderly? J Clin Endocrinol Metab. 2013;98:2256-2266.
1. Aoki Y, Belin RM, Clickner R, et al. Serum TSH and total T4 in the United States population and their association with participant characteristics: National Health and Nutrition Examination Survey (NHANES 1999-2002). Thyroid. 2007;17:1211-1223.
2. Vaidya B, Pearce SH. Management of hypothyroidism in adults. BMJ. 2008;337:a801.
3. Garber JR, Cobin RH, Gharib H, et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Endocr Pract. 2012;18:988-1028.
4. Jonklaas J, Bianco AC, Bauer AJ, et al. Guidelines for the treatment of hypothyroidism: prepared by the American Thyroid Association task force on thyroid hormone replacement. Thyroid. 2014;24:1670-1751.
5. Toft AD. Thyroxine therapy. N Engl J Med. 1994;331:174-180.
6. Floriani C, Gencer B, Collet TH, et al. Subclinical thyroid dysfunction and cardiovascular diseases: 2016 update. Eur Heart J. 2018;39:503-507.
7. Lexi-Comp, Inc. (Lexi-Drugs®). https://online.lexi.com/lco/action/login. Accessed July 7, 2017.
8. Okosieme O, Gilbert J, Abraham P, et al. Management of primary hypothyroidism: statement by the British Thyroid Association Executive Committee. Clin Endocrinol (Oxf). 2016;84:799-808.
9. Fish LH, Schwartz HL, Cavanaugh J, et al. Replacement dose, metabolism, and bioavailability of levothyroxine in the treatment of hypothyroidism. Role of triiodothyronine in pituitary feedback in humans. N Engl J Med. 1987;316:764-770.
10. John-Kalarickal J, Pearlman G, Carlson HE. New medications which decrease levothyroxine absorption. Thyroid. 2007;17:763-765.
11. Sachmechi I, Reich DM, Aninyei M, et al. Effect of proton pump inhibitors on serum thyroid-stimulating hormone level in euthyroid patients treated with levothyroxine for hypothyroidism. Endocr Pract. 2007;13:345-349.
12. Sperber AD, Liel Y. Evidence for interference with the intestinal absorption of levothyroxine sodium by aluminum hydroxide. Arch Intern Med. 1992;152:183-184.
13. Zamfirescu I, Carlson HE. Absorption of levothyroxine when coadministered with various calcium formulations. Thyroid. 2011;21:483-486.
14. Campbell NR, Hasinoff BB, Stalts H, et al. Ferrous sulfate reduces thyroxine efficacy in patients with hypothyroidism. Ann Intern Med. 1992;117:1010-1013.
15. Bugdaci MS, Zuhur SS, Sokmen M, et al. The role of Helicobacter pylori in patients with hypothyroidism in whom could not be achieved normal thyrotropin levels despite treatment with high doses of thyroxine. Helicobacter. 2011;16:124-130.
16. Centanni M, Gargano L, Canettieri G, et al. Thyroxine in goiter, Helicobacter pylori infection, and chronic gastritis. N Engl J Med. 2006;354:1787-1795.
17. Centanni M, Marignani M, Gargano L, et al. Atrophic body gastritis in patients with autoimmune thyroid disease: an underdiagnosed association. Arch Intern Med. 1999;159:1726-1730.
18. Collins D, Wilcox R, Nathan M, et al. Celiac disease and hypothyroidism. Am J Med. 2012;125:278-282.
19. Azizi F, Belur R, Albano J. Malabsorption of thyroid hormones after jejunoileal bypass for obesity. Ann Intern Med. 1979;90:941-942.
20. Gkotsina M, Michalaki M, Mamali I, et al. Improved levothyroxine pharmacokinetics after bariatric surgery. Thyroid. 2013;23:414-419.
21. Hennessey JV, Espaillat R. Diagnosis and management of subclinical hypothyroidism in elderly adults: a review of the literature. J Am Geriatr Soc. 2015;63:1663-1673.
22. Alexander EK, Pearce EN, Brent GA, et al. 2017 Guidelines of the American Thyroid Association for the diagnosis and management of thyroid disease during pregnancy and the postpartum. Thyroid. 2017;27:315-389.
23. Carney LA, Quinlan JD, West JM. Thyroid disease in pregnancy. Am Fam Physician. 2014;89:273-278.
24. Fröhlich E, Wahl R. Thyroid autoimmunity: role of anti-thyroid antibodies in thyroid and extra-thyroidal diseases. Front Immunol. 2017;8:521.
25. Aksoy DY, Kerimoglu U, Okur H, et al. Effects of prophylactic thyroid hormone replacement in euthyroid Hashimoto’s thyroiditis. Endocr J. 2005;52:337-343.
26. Padberg S, Heller K, Usadel KH, et al. One-year prophylactic treatment of euthyroid Hashimoto’s thyroiditis patients with levothyroxine: is there a benefit? Thyroid. 2001;11:249-255.
27. Rugge B, Balshem H, Sehgal R, et al. Screening and Treatment of Subclinical Hypothyroidism or Hyperthyroidism [Internet]. Comparative Effectiveness Reviews, No. 24. Rockville, MD: Agency for Healthcare Research and Quality; October 2011. www.ncbi.nlm.nih.gov/books/NBK83492/. Accessed February 21, 2020.
28. Hollowell JG, Staehling NW, Flanders WD, et al. Serum TSH, T(4), and thyroid antibodies in the United States population (1988 to 1994): National Health and Nutrition Examination Survey (NHANES III). J Clin Endocrinol Metab. 2002;87:489-499.
29. Surks MI, Hollowell JG. Age-specific distribution of serum thyrotropin and antithyroid antibodies in the US population: implications for the prevalence of subclinical hypothyroidism. J Clin Endocrinol Metab. 2007;92:4575-4582.
30. Pasqualetti G, Tognini S, Polini A, et al. Is subclinical hypothyroidism a cardiovascular risk factor in the elderly? J Clin Endocrinol Metab. 2013;98:2256-2266.
PRACTICE RECOMMENDATIONS
› Prescribe levothyroxine 1.6 mcg/kg/d for healthy adult patients < 50 years of age with overt hypothyroidism. B
› Consider lower initial doses of levothyroxine in patients with cardiac disease (12.5-50 mcg/d) or subclinical hypothyroidism (25-75 mcg/d). B
› Titrate levothyroxine by 12.5 to 25 mcg/d at 6- to 8-week intervals based on thyroid-stimulating hormone measurements, comorbidities, and symptoms. C
› Closely monitor and provide thyroid supplementation to female patients who are pregnant or of reproductive age with concomitant hypothyroidism. C
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Intervention improves antibiotics use in UTIs
A multifaceted intervention significantly changed clinicians’ use of antibiotics to treat urinary tract infections (UTIs) in children, according to data from more than 2,000 cases observed between January 2014 and September 2018.
“Changing clinicians’ antibiotic prescribing practices can be challenging; barriers to change include lack of awareness of new evidence, competing clinical demands, and concern about treatment failure,” wrote Matthew F. Daley, MD, of Kaiser Permanente Colorado, Aurora, and colleagues in Pediatrics.
To promote judicious antibiotic use, the researchers designed an intervention including the development of new local UTI guidelines; a live, case-based educational session; emailed knowledge assessments before and after the session; and a specific UTI order set in the EHR.
The researchers divided the study period into a preintervention period (January 1, 2014, to April 25, 2017) and a postintervention period (April 26, 2017, to September 30, 2018). They collected data on 2,142 incident outpatient UTIs; 1,636 from the preintervention period and 506 from the postintervention period. The patients were younger than 18 years and older than 60 days, and children with complicated urologic or neurologic conditions were excluded.
(P less than .0001). In particular, the use of first-line, narrow spectrum cephalexin increased significantly from 29% during the preintervention period to 53% during the postintervention period (P less than .0001). In addition, use of broad spectrum cefixime decreased from 17% during the preintervention period to 3% during the postintervention period (P less than .0001). These changes in prescribing patterns continued through the end of the study period, the researchers said.
The study was limited by several factors, notably that “the interrupted time-series design prevents us from inferring that the intervention caused the observed change in practice,” the researchers wrote. However, other factors including the immediate change in prescribing patterns after the intervention, multiple time points, large sample size, and consistent UTI case mix support the impact of the intervention, they suggested. Although the results might not translate completely to other settings, “developing a UTI-specific EHR order set is relatively straightforward” and might be applied elsewhere, they noted.
“Despite the limitations inherent in a nonexperimental study design, the methods and interventions developed in the current study may be informative to other learning health systems and other content areas when conducting organization-wide quality improvement initiatives,” they concluded.
The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
SOURCE: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
A multifaceted intervention significantly changed clinicians’ use of antibiotics to treat urinary tract infections (UTIs) in children, according to data from more than 2,000 cases observed between January 2014 and September 2018.
“Changing clinicians’ antibiotic prescribing practices can be challenging; barriers to change include lack of awareness of new evidence, competing clinical demands, and concern about treatment failure,” wrote Matthew F. Daley, MD, of Kaiser Permanente Colorado, Aurora, and colleagues in Pediatrics.
To promote judicious antibiotic use, the researchers designed an intervention including the development of new local UTI guidelines; a live, case-based educational session; emailed knowledge assessments before and after the session; and a specific UTI order set in the EHR.
The researchers divided the study period into a preintervention period (January 1, 2014, to April 25, 2017) and a postintervention period (April 26, 2017, to September 30, 2018). They collected data on 2,142 incident outpatient UTIs; 1,636 from the preintervention period and 506 from the postintervention period. The patients were younger than 18 years and older than 60 days, and children with complicated urologic or neurologic conditions were excluded.
(P less than .0001). In particular, the use of first-line, narrow spectrum cephalexin increased significantly from 29% during the preintervention period to 53% during the postintervention period (P less than .0001). In addition, use of broad spectrum cefixime decreased from 17% during the preintervention period to 3% during the postintervention period (P less than .0001). These changes in prescribing patterns continued through the end of the study period, the researchers said.
The study was limited by several factors, notably that “the interrupted time-series design prevents us from inferring that the intervention caused the observed change in practice,” the researchers wrote. However, other factors including the immediate change in prescribing patterns after the intervention, multiple time points, large sample size, and consistent UTI case mix support the impact of the intervention, they suggested. Although the results might not translate completely to other settings, “developing a UTI-specific EHR order set is relatively straightforward” and might be applied elsewhere, they noted.
“Despite the limitations inherent in a nonexperimental study design, the methods and interventions developed in the current study may be informative to other learning health systems and other content areas when conducting organization-wide quality improvement initiatives,” they concluded.
The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
SOURCE: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
A multifaceted intervention significantly changed clinicians’ use of antibiotics to treat urinary tract infections (UTIs) in children, according to data from more than 2,000 cases observed between January 2014 and September 2018.
“Changing clinicians’ antibiotic prescribing practices can be challenging; barriers to change include lack of awareness of new evidence, competing clinical demands, and concern about treatment failure,” wrote Matthew F. Daley, MD, of Kaiser Permanente Colorado, Aurora, and colleagues in Pediatrics.
To promote judicious antibiotic use, the researchers designed an intervention including the development of new local UTI guidelines; a live, case-based educational session; emailed knowledge assessments before and after the session; and a specific UTI order set in the EHR.
The researchers divided the study period into a preintervention period (January 1, 2014, to April 25, 2017) and a postintervention period (April 26, 2017, to September 30, 2018). They collected data on 2,142 incident outpatient UTIs; 1,636 from the preintervention period and 506 from the postintervention period. The patients were younger than 18 years and older than 60 days, and children with complicated urologic or neurologic conditions were excluded.
(P less than .0001). In particular, the use of first-line, narrow spectrum cephalexin increased significantly from 29% during the preintervention period to 53% during the postintervention period (P less than .0001). In addition, use of broad spectrum cefixime decreased from 17% during the preintervention period to 3% during the postintervention period (P less than .0001). These changes in prescribing patterns continued through the end of the study period, the researchers said.
The study was limited by several factors, notably that “the interrupted time-series design prevents us from inferring that the intervention caused the observed change in practice,” the researchers wrote. However, other factors including the immediate change in prescribing patterns after the intervention, multiple time points, large sample size, and consistent UTI case mix support the impact of the intervention, they suggested. Although the results might not translate completely to other settings, “developing a UTI-specific EHR order set is relatively straightforward” and might be applied elsewhere, they noted.
“Despite the limitations inherent in a nonexperimental study design, the methods and interventions developed in the current study may be informative to other learning health systems and other content areas when conducting organization-wide quality improvement initiatives,” they concluded.
The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
SOURCE: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
FROM PEDIATRICS
Key clinical point: After an educational intervention, approximately 62% of clinicians prescribed first-line antibiotics, up from 43% before the intervention.
Major finding: Cephalexin use increased from 29% before the intervention to 53% after the intervention.
Study details: The data come from a review of 2,142 incident outpatient cases of urinary tract infection in patients aged older than 60 days up to 18 years.
Disclosures: The study was supported by unrestricted internal resources from the Colorado Permanente Medical Group. The researchers had no financial conflicts to disclose.
Source: Daley MF et al. Pediatrics. 2020 Mar 3. doi: 10.1542/peds.2019-2503.
Washington State grapples with coronavirus outbreak
As the first COVID-19 outbreak in the United States emerges in Washington State, the city of Seattle, King County, and Washington State health officials provided the beginnings of a roadmap for how the region will address the rapidly evolving health crisis.
Health officials announced that four new cases were reported over the weekend in King County, Wash. There have now been 10 hospitalizations and 6 COVID-19 deaths at Evergreen Health, Kirkland, Wash. Of the deaths, five were King County residents and one was a resident of Snohomish County. Three patients died on March 1; all were in their 70s or 80s with comorbidities. Two had been residents of the Life Care senior residential facility that is at the center of the Kirkland outbreak. The number of cases in Washington now totals 18, with four cases in Snohomish County and the balance in neighboring King County.
Approximately 29 cases are under investigation with test results pending; a Centers for Disease Control and Prevention (CDC) team is on-site.
Speaking at a news conference March 2, officials sought to strike a balance between giving the community a realistic appraisal of the likely scope of the COVID-19 outbreak and avoiding sparking a panic.
“This is a complex and unprecedented challenge nationally, globally, and locally. The vast majority of the infected have mild or moderate disease and do not need hospitalization,” said Jeffrey Duchin, MD, health officer and chief, Communicable Disease EPI/Immunization Section, Public Health, Seattle and King County, and a professor of infectious diseases at the University of Washington, Seattle. “On the other hand, it’s obvious that this infection can cause very serious disease in people who are older and have underlying health conditions. We expect cases to continue to increase. We are taking the situation extremely seriously; the risk for all of us becoming infected is increasing. ...There is the potential for many to become ill at the same time.”
Among the measures being taken immediately are the purchase by King County of a hotel to house individuals who require isolation and those who are convalescing from the virus. Officials are also placing a number of prefabricated stand-alone housing units on public grounds in Seattle, with the recognition that the area has a large transient and homeless community. The stand-alone units will house homeless individuals who need isolation, treatment, or recuperation but who aren’t ill enough to be hospitalized.
Dr. Duchin said that testing capacity is ramping up rapidly in Washington State: The state lab can now accommodate up to about 200 tests daily, and expects to be able to do up to 1,000 daily soon. The University of Washington’s testing capacity will come online March 2 or 3 as a testing facility with similar initial and future peak testing capacities.
The testing strategy will continue to include very ill individuals with pneumonia or other respiratory illness of unknown etiology, but will also expand to include less ill people. This shift is being made in accordance with a shift in CDC guidelines, because of increased testing capacity, and to provide a better picture of the severity, scope, geography, and timing of the current COVID-19 outbreak in the greater Seattle area.
No school closures or cancellation of gatherings are currently recommended by public health authorities. There are currently no COVID-19 cases in Washington schools. The expectation is that any recommendations regarding closures will be re-evaluated as the outbreak progresses.
Repeatedly, officials asked the general public to employ basic measures such as handwashing and avoidance of touching the face, and to spare masks for the ill and for those who care for them. “The vast majority of people will not have serious illness. In turn we need to do everything we can to help those health care workers. I’m asking the public to do things like save the masks for our health care workers. …We need assets for our front-line health care workers and also for those who may be needing them,” said King County Health Department director Patty Hayes, RN, MN.
Now is also the time for households to initiate basic emergency preparedness measures, such as having adequate food and medication, and to make arrangements for childcare in the event of school closures, said several officials.
“We can decrease the impact on our health care system by reducing our individual risk. We are making individual- and community-level recommendations to limit the spread of disease. These are very similar to what we recommend for influenza,” said Dr. Duchin.
Ettore Palazzo, MD, chief medical and quality officer at EvergreenHealth, gave a sense of how the hospital is coping with being Ground Zero for COVID-19 in the United States. “We have made adjustments for airborne precautions,” he said, including transforming the entire critical care unit to a negative pressure unit. “We have these capabilities in other parts of the hospital as well.” Staff are working hard, but thus far staffing has kept pace with demand, he said, but all are feeling the strain already.
Dr. Duchin made the point that Washington is relatively well equipped to handle the increasingly likely scenario of a large spike in coronavirus cases, since it’s part of the Northwest Healthcare Response Network. The network is planning for sharing resources such as staff, respirators, and intensive care unit beds as circumstances warrant.
“What you just heard illustrates the challenge of this disease,” said Dr. Duchin, summing up. “The public health service and clinical health care delivery systems don’t have the capacity to track down every case in the community. I’m guessing we will see more cases of coronavirus than we see of influenza. At some point we will be shifting from counting every case” to focusing on outbreaks and the critically ill in hospitals, he said.
“We are still trying to contain the outbreak, but we are at the same time pivoting to a more community-based approach,” similar to the approach with influenza, said Dr. Duchin.
A summary of deaths and ongoing cases, drawn from the press release, is below:
The four new cases are:
• A male in his 50s, hospitalized at Highline Hospital. He has no known exposures. He is in stable but critical condition. He had no underlying health conditions.
• A male in his 70s, a resident of Life Care, hospitalized at EvergreenHealth in Kirkland. The man had underlying health conditions, and died March 1.
• A female in her 70s, a resident of Life Care, hospitalized at EvergreenHealth in Kirkland. The woman had underlying health conditions, and died March 1.
• A female in her 80s, a resident of Life Care, was hospitalized at EvergreenHealth. She is in critical condition.
In addition, a woman in her 80s, who was already reported as in critical condition at Evergreen, has died. She died on March 1.
Ten other cases, already reported earlier by Public Health, include:
• A female in her 80s, hospitalized at EvergreenHealth in Kirkland. This person has now died, and is reported as such above.
• A female in her 90s, hospitalized at EvergreenHealth in Kirkland. The woman has underlying health conditions, and is in critical condition.
• A male in his 70s, hospitalized at EvergreenHealth in Kirkland. The man has underlying health conditions, and is in critical condition.
• A male in his 70s was hospitalized at EvergreenHealth. He had underlying health conditions and died on Feb. 29.
• A man in his 60s, hospitalized at Valley Medical Center in Renton.
• A man in 60s, hospitalized at Virginia Mason Medical Center.
• A woman in her 50s, who had traveled to South Korea; recovering at home.
• A woman in her 70s, who was a resident of Life Care in Kirkland, hospitalized at EvergreenHealth.
• A woman in her 40s, employed by Life Care, who is hospitalized at Overlake Medical Center.
• A man in his 50s, who was hospitalized and died at EvergreenHealth.
As the first COVID-19 outbreak in the United States emerges in Washington State, the city of Seattle, King County, and Washington State health officials provided the beginnings of a roadmap for how the region will address the rapidly evolving health crisis.
Health officials announced that four new cases were reported over the weekend in King County, Wash. There have now been 10 hospitalizations and 6 COVID-19 deaths at Evergreen Health, Kirkland, Wash. Of the deaths, five were King County residents and one was a resident of Snohomish County. Three patients died on March 1; all were in their 70s or 80s with comorbidities. Two had been residents of the Life Care senior residential facility that is at the center of the Kirkland outbreak. The number of cases in Washington now totals 18, with four cases in Snohomish County and the balance in neighboring King County.
Approximately 29 cases are under investigation with test results pending; a Centers for Disease Control and Prevention (CDC) team is on-site.
Speaking at a news conference March 2, officials sought to strike a balance between giving the community a realistic appraisal of the likely scope of the COVID-19 outbreak and avoiding sparking a panic.
“This is a complex and unprecedented challenge nationally, globally, and locally. The vast majority of the infected have mild or moderate disease and do not need hospitalization,” said Jeffrey Duchin, MD, health officer and chief, Communicable Disease EPI/Immunization Section, Public Health, Seattle and King County, and a professor of infectious diseases at the University of Washington, Seattle. “On the other hand, it’s obvious that this infection can cause very serious disease in people who are older and have underlying health conditions. We expect cases to continue to increase. We are taking the situation extremely seriously; the risk for all of us becoming infected is increasing. ...There is the potential for many to become ill at the same time.”
Among the measures being taken immediately are the purchase by King County of a hotel to house individuals who require isolation and those who are convalescing from the virus. Officials are also placing a number of prefabricated stand-alone housing units on public grounds in Seattle, with the recognition that the area has a large transient and homeless community. The stand-alone units will house homeless individuals who need isolation, treatment, or recuperation but who aren’t ill enough to be hospitalized.
Dr. Duchin said that testing capacity is ramping up rapidly in Washington State: The state lab can now accommodate up to about 200 tests daily, and expects to be able to do up to 1,000 daily soon. The University of Washington’s testing capacity will come online March 2 or 3 as a testing facility with similar initial and future peak testing capacities.
The testing strategy will continue to include very ill individuals with pneumonia or other respiratory illness of unknown etiology, but will also expand to include less ill people. This shift is being made in accordance with a shift in CDC guidelines, because of increased testing capacity, and to provide a better picture of the severity, scope, geography, and timing of the current COVID-19 outbreak in the greater Seattle area.
No school closures or cancellation of gatherings are currently recommended by public health authorities. There are currently no COVID-19 cases in Washington schools. The expectation is that any recommendations regarding closures will be re-evaluated as the outbreak progresses.
Repeatedly, officials asked the general public to employ basic measures such as handwashing and avoidance of touching the face, and to spare masks for the ill and for those who care for them. “The vast majority of people will not have serious illness. In turn we need to do everything we can to help those health care workers. I’m asking the public to do things like save the masks for our health care workers. …We need assets for our front-line health care workers and also for those who may be needing them,” said King County Health Department director Patty Hayes, RN, MN.
Now is also the time for households to initiate basic emergency preparedness measures, such as having adequate food and medication, and to make arrangements for childcare in the event of school closures, said several officials.
“We can decrease the impact on our health care system by reducing our individual risk. We are making individual- and community-level recommendations to limit the spread of disease. These are very similar to what we recommend for influenza,” said Dr. Duchin.
Ettore Palazzo, MD, chief medical and quality officer at EvergreenHealth, gave a sense of how the hospital is coping with being Ground Zero for COVID-19 in the United States. “We have made adjustments for airborne precautions,” he said, including transforming the entire critical care unit to a negative pressure unit. “We have these capabilities in other parts of the hospital as well.” Staff are working hard, but thus far staffing has kept pace with demand, he said, but all are feeling the strain already.
Dr. Duchin made the point that Washington is relatively well equipped to handle the increasingly likely scenario of a large spike in coronavirus cases, since it’s part of the Northwest Healthcare Response Network. The network is planning for sharing resources such as staff, respirators, and intensive care unit beds as circumstances warrant.
“What you just heard illustrates the challenge of this disease,” said Dr. Duchin, summing up. “The public health service and clinical health care delivery systems don’t have the capacity to track down every case in the community. I’m guessing we will see more cases of coronavirus than we see of influenza. At some point we will be shifting from counting every case” to focusing on outbreaks and the critically ill in hospitals, he said.
“We are still trying to contain the outbreak, but we are at the same time pivoting to a more community-based approach,” similar to the approach with influenza, said Dr. Duchin.
A summary of deaths and ongoing cases, drawn from the press release, is below:
The four new cases are:
• A male in his 50s, hospitalized at Highline Hospital. He has no known exposures. He is in stable but critical condition. He had no underlying health conditions.
• A male in his 70s, a resident of Life Care, hospitalized at EvergreenHealth in Kirkland. The man had underlying health conditions, and died March 1.
• A female in her 70s, a resident of Life Care, hospitalized at EvergreenHealth in Kirkland. The woman had underlying health conditions, and died March 1.
• A female in her 80s, a resident of Life Care, was hospitalized at EvergreenHealth. She is in critical condition.
In addition, a woman in her 80s, who was already reported as in critical condition at Evergreen, has died. She died on March 1.
Ten other cases, already reported earlier by Public Health, include:
• A female in her 80s, hospitalized at EvergreenHealth in Kirkland. This person has now died, and is reported as such above.
• A female in her 90s, hospitalized at EvergreenHealth in Kirkland. The woman has underlying health conditions, and is in critical condition.
• A male in his 70s, hospitalized at EvergreenHealth in Kirkland. The man has underlying health conditions, and is in critical condition.
• A male in his 70s was hospitalized at EvergreenHealth. He had underlying health conditions and died on Feb. 29.
• A man in his 60s, hospitalized at Valley Medical Center in Renton.
• A man in 60s, hospitalized at Virginia Mason Medical Center.
• A woman in her 50s, who had traveled to South Korea; recovering at home.
• A woman in her 70s, who was a resident of Life Care in Kirkland, hospitalized at EvergreenHealth.
• A woman in her 40s, employed by Life Care, who is hospitalized at Overlake Medical Center.
• A man in his 50s, who was hospitalized and died at EvergreenHealth.
As the first COVID-19 outbreak in the United States emerges in Washington State, the city of Seattle, King County, and Washington State health officials provided the beginnings of a roadmap for how the region will address the rapidly evolving health crisis.
Health officials announced that four new cases were reported over the weekend in King County, Wash. There have now been 10 hospitalizations and 6 COVID-19 deaths at Evergreen Health, Kirkland, Wash. Of the deaths, five were King County residents and one was a resident of Snohomish County. Three patients died on March 1; all were in their 70s or 80s with comorbidities. Two had been residents of the Life Care senior residential facility that is at the center of the Kirkland outbreak. The number of cases in Washington now totals 18, with four cases in Snohomish County and the balance in neighboring King County.
Approximately 29 cases are under investigation with test results pending; a Centers for Disease Control and Prevention (CDC) team is on-site.
Speaking at a news conference March 2, officials sought to strike a balance between giving the community a realistic appraisal of the likely scope of the COVID-19 outbreak and avoiding sparking a panic.
“This is a complex and unprecedented challenge nationally, globally, and locally. The vast majority of the infected have mild or moderate disease and do not need hospitalization,” said Jeffrey Duchin, MD, health officer and chief, Communicable Disease EPI/Immunization Section, Public Health, Seattle and King County, and a professor of infectious diseases at the University of Washington, Seattle. “On the other hand, it’s obvious that this infection can cause very serious disease in people who are older and have underlying health conditions. We expect cases to continue to increase. We are taking the situation extremely seriously; the risk for all of us becoming infected is increasing. ...There is the potential for many to become ill at the same time.”
Among the measures being taken immediately are the purchase by King County of a hotel to house individuals who require isolation and those who are convalescing from the virus. Officials are also placing a number of prefabricated stand-alone housing units on public grounds in Seattle, with the recognition that the area has a large transient and homeless community. The stand-alone units will house homeless individuals who need isolation, treatment, or recuperation but who aren’t ill enough to be hospitalized.
Dr. Duchin said that testing capacity is ramping up rapidly in Washington State: The state lab can now accommodate up to about 200 tests daily, and expects to be able to do up to 1,000 daily soon. The University of Washington’s testing capacity will come online March 2 or 3 as a testing facility with similar initial and future peak testing capacities.
The testing strategy will continue to include very ill individuals with pneumonia or other respiratory illness of unknown etiology, but will also expand to include less ill people. This shift is being made in accordance with a shift in CDC guidelines, because of increased testing capacity, and to provide a better picture of the severity, scope, geography, and timing of the current COVID-19 outbreak in the greater Seattle area.
No school closures or cancellation of gatherings are currently recommended by public health authorities. There are currently no COVID-19 cases in Washington schools. The expectation is that any recommendations regarding closures will be re-evaluated as the outbreak progresses.
Repeatedly, officials asked the general public to employ basic measures such as handwashing and avoidance of touching the face, and to spare masks for the ill and for those who care for them. “The vast majority of people will not have serious illness. In turn we need to do everything we can to help those health care workers. I’m asking the public to do things like save the masks for our health care workers. …We need assets for our front-line health care workers and also for those who may be needing them,” said King County Health Department director Patty Hayes, RN, MN.
Now is also the time for households to initiate basic emergency preparedness measures, such as having adequate food and medication, and to make arrangements for childcare in the event of school closures, said several officials.
“We can decrease the impact on our health care system by reducing our individual risk. We are making individual- and community-level recommendations to limit the spread of disease. These are very similar to what we recommend for influenza,” said Dr. Duchin.
Ettore Palazzo, MD, chief medical and quality officer at EvergreenHealth, gave a sense of how the hospital is coping with being Ground Zero for COVID-19 in the United States. “We have made adjustments for airborne precautions,” he said, including transforming the entire critical care unit to a negative pressure unit. “We have these capabilities in other parts of the hospital as well.” Staff are working hard, but thus far staffing has kept pace with demand, he said, but all are feeling the strain already.
Dr. Duchin made the point that Washington is relatively well equipped to handle the increasingly likely scenario of a large spike in coronavirus cases, since it’s part of the Northwest Healthcare Response Network. The network is planning for sharing resources such as staff, respirators, and intensive care unit beds as circumstances warrant.
“What you just heard illustrates the challenge of this disease,” said Dr. Duchin, summing up. “The public health service and clinical health care delivery systems don’t have the capacity to track down every case in the community. I’m guessing we will see more cases of coronavirus than we see of influenza. At some point we will be shifting from counting every case” to focusing on outbreaks and the critically ill in hospitals, he said.
“We are still trying to contain the outbreak, but we are at the same time pivoting to a more community-based approach,” similar to the approach with influenza, said Dr. Duchin.
A summary of deaths and ongoing cases, drawn from the press release, is below:
The four new cases are:
• A male in his 50s, hospitalized at Highline Hospital. He has no known exposures. He is in stable but critical condition. He had no underlying health conditions.
• A male in his 70s, a resident of Life Care, hospitalized at EvergreenHealth in Kirkland. The man had underlying health conditions, and died March 1.
• A female in her 70s, a resident of Life Care, hospitalized at EvergreenHealth in Kirkland. The woman had underlying health conditions, and died March 1.
• A female in her 80s, a resident of Life Care, was hospitalized at EvergreenHealth. She is in critical condition.
In addition, a woman in her 80s, who was already reported as in critical condition at Evergreen, has died. She died on March 1.
Ten other cases, already reported earlier by Public Health, include:
• A female in her 80s, hospitalized at EvergreenHealth in Kirkland. This person has now died, and is reported as such above.
• A female in her 90s, hospitalized at EvergreenHealth in Kirkland. The woman has underlying health conditions, and is in critical condition.
• A male in his 70s, hospitalized at EvergreenHealth in Kirkland. The man has underlying health conditions, and is in critical condition.
• A male in his 70s was hospitalized at EvergreenHealth. He had underlying health conditions and died on Feb. 29.
• A man in his 60s, hospitalized at Valley Medical Center in Renton.
• A man in 60s, hospitalized at Virginia Mason Medical Center.
• A woman in her 50s, who had traveled to South Korea; recovering at home.
• A woman in her 70s, who was a resident of Life Care in Kirkland, hospitalized at EvergreenHealth.
• A woman in her 40s, employed by Life Care, who is hospitalized at Overlake Medical Center.
• A man in his 50s, who was hospitalized and died at EvergreenHealth.
FROM A KING COUNTY, WASH. NEWS BRIEFING
New strategies cut esophageal damage from AFib catheter ablation
NATIONAL HARBOR, MD. – Thermal injury of a patient’s esophagus during radiofrequency catheter ablation of atrial fibrillation is notorious as a relatively common and problematic complication of the procedure, but two new approaches showed promise for substantially cutting the risk of esophageal thermal injury and the potential for the most severe damage: perforation.
One of these innovations is intensive esophageal cooling with a commercially marketed, fluid-chilled catheter placed in a patient’s esophagus during radiofrequency catheter ablation that keeps the inner surface of the esophagus at 4°C. This approach cut the incidence of periprocedural episodes of endoscopically detected esophageal thermal injury from 20% among controls to 3% in patients who had esophageal cooling in a randomized study with 120 patients, Mark M. Gallagher, MD, said at the annual International AF Symposium. The same device can also maintain a temperature on the inner surface of the esophagus of 42 ° C in patients undergoing cryoablation of atrial fibrillation, noted Dr. Gallagher, a cardiac electrophysiologist at St. George’s University Hospitals in London.
A second approach to cutting esophageal damage focuses on modifying the energy delivery with a radiofrequency ablation method known as high-power short-duration (HPSD). As the name says, this strategy uses a relatively high level of radiofrequency energy, 50 watts in the reported experience, for the brief interval of about 7 seconds, ideally delivering an overall Ablation Index of at least 350 but below 360, said Thomas Deneke, MD, an electrophysiologist, professor, and cochief of cardiology at the Heart Center in Bad Neustadt, Germany.
Dr. Deneke and his associates in Bad Neustadt began using this HPSD approach in mid-2019, and by early 2020 they had data from 179 patients who underwent first-time catheter ablation of atrial fibrillation (AFib), all of whom had undergone routine esophageal endoscopy 1-3 days after their treatment. Eight patients (4%) showed evidence of endoscopically detected esophageal lesions (EDEL), including three patients (2%) with an actual esophageal ulcer, and one (0.6%) who developed a perforation that healed after 52 days, Dr. Deneke reported. An additional 55 patients underwent a redo catheter ablation procedure using the HPSD method during this period, and in that group follow-up endoscopy in all patients showed EDEL in two patients (4%). In contrast, during Jan. 2012–May 2019, the same German center treated 2,102 patients who had a first radiofrequency catheter ablation using convention energy levels and treatment times, which resulted in 291 patients having an EDEL (14%), including 94 (4%) with an ulcer, and six patients (0.3%) with an esophageal perforation, he said.
His center’s recent safety experience with HPSD radiofrequncy ablation, compared with the historical controls, suggests that this technique can produce a substantial reduction in esophageal thermal injury, but HPSD has not completely eliminated the risk and hence there is need for continued alertness for this potential complication Dr. Deneke concluded. The HPSD method is also limited by having “a very narrow window” between efficacy at an Ablation Index of 350 and safety when the index remains below 360, he added.
The randomized study that Dr. Gallagher ran at St. George’s followed an analysis he and his associates recently published that suggested efficacy using esophageal cooling in prior reports when the data combined in a meta-analysis (J Interv Card Electrophysiol. 2019 Nov 22. doi: 10.1007/s10840-019-00661-5). They also concluded that the clinical setting required a temperature control device with an enhanced capacity for rapid cooling, which prior studies had lacked. So they turned to a Food and Drug Administration–approved catheter designed for placement in the esophagus for the purpose of either whole-body cooling or warming.
The study randomized a total of 187 patients, but collected follow-up endoscopy at 5-7 days after the ablation procedure on 120 patients, of whom 60 received esophageal cooling and 60 did not. The types of ablations performed on patients in the two study arms were similar, and use of esophageal cooling had no impact on treatment duration or efficacy, either acute and longer term, Dr. Gallagher reported.
Cooling had a marked and statistically significant impact on endoscopically detected thermal injury. Although two patients in the group that underwent cooling had injuries, in one of these cases the injury involved a protocol violation: Radiofrequency ablation mistakenly occurred after the cooling device shut off, and it was during this period when the injury happened. In the second case of thermal injury, blinded scoring judged the injury as grade 2 in severity – an erosion of less than 5 mm – on a nine-item scale that ranged from zero to grade 6, the most severe level denoting a fistula. By contrast, among the 12 patients with thermal injury in the nonprotected subgroup, one patient had a grade 5a lesion denoting a deep ulcer, one had a 4b denoting a superficial ulcer with a clot, and four had a 4a lesion defined as a clean superficial ulcer.
“This is really effective. It’s the first study to show reduced damage without affecting ablation efficacy,” Dr. Gallagher said. He plans to now use this method of esophageal protection routinely for his AFib ablation patients who pay privately, and for patients insured under the national U.K. system once this coverage is approved. Dr. Deneke expressed his interest in also using this approach to esophageal protection, but noted that currently he did not have access to the cooling catheter that Dr. Gallagher used because of regulatory constraints.
The esophageal cooling study was sponsored by Attune Medical, which markets the cooling device. Dr. Gallagher has received research funding from Attune Medical, and has received honoraria as a speaker on behalf of Biosense Webster and Medtronic. Dr. Deneke has been a speaker on behalf of Abbott, Biosense Webster, Biotronik, and Boston Scientific, and his institution has received research funding from Biosense Webster and Securus/Boston Scientific.
NATIONAL HARBOR, MD. – Thermal injury of a patient’s esophagus during radiofrequency catheter ablation of atrial fibrillation is notorious as a relatively common and problematic complication of the procedure, but two new approaches showed promise for substantially cutting the risk of esophageal thermal injury and the potential for the most severe damage: perforation.
One of these innovations is intensive esophageal cooling with a commercially marketed, fluid-chilled catheter placed in a patient’s esophagus during radiofrequency catheter ablation that keeps the inner surface of the esophagus at 4°C. This approach cut the incidence of periprocedural episodes of endoscopically detected esophageal thermal injury from 20% among controls to 3% in patients who had esophageal cooling in a randomized study with 120 patients, Mark M. Gallagher, MD, said at the annual International AF Symposium. The same device can also maintain a temperature on the inner surface of the esophagus of 42 ° C in patients undergoing cryoablation of atrial fibrillation, noted Dr. Gallagher, a cardiac electrophysiologist at St. George’s University Hospitals in London.
A second approach to cutting esophageal damage focuses on modifying the energy delivery with a radiofrequency ablation method known as high-power short-duration (HPSD). As the name says, this strategy uses a relatively high level of radiofrequency energy, 50 watts in the reported experience, for the brief interval of about 7 seconds, ideally delivering an overall Ablation Index of at least 350 but below 360, said Thomas Deneke, MD, an electrophysiologist, professor, and cochief of cardiology at the Heart Center in Bad Neustadt, Germany.
Dr. Deneke and his associates in Bad Neustadt began using this HPSD approach in mid-2019, and by early 2020 they had data from 179 patients who underwent first-time catheter ablation of atrial fibrillation (AFib), all of whom had undergone routine esophageal endoscopy 1-3 days after their treatment. Eight patients (4%) showed evidence of endoscopically detected esophageal lesions (EDEL), including three patients (2%) with an actual esophageal ulcer, and one (0.6%) who developed a perforation that healed after 52 days, Dr. Deneke reported. An additional 55 patients underwent a redo catheter ablation procedure using the HPSD method during this period, and in that group follow-up endoscopy in all patients showed EDEL in two patients (4%). In contrast, during Jan. 2012–May 2019, the same German center treated 2,102 patients who had a first radiofrequency catheter ablation using convention energy levels and treatment times, which resulted in 291 patients having an EDEL (14%), including 94 (4%) with an ulcer, and six patients (0.3%) with an esophageal perforation, he said.
His center’s recent safety experience with HPSD radiofrequncy ablation, compared with the historical controls, suggests that this technique can produce a substantial reduction in esophageal thermal injury, but HPSD has not completely eliminated the risk and hence there is need for continued alertness for this potential complication Dr. Deneke concluded. The HPSD method is also limited by having “a very narrow window” between efficacy at an Ablation Index of 350 and safety when the index remains below 360, he added.
The randomized study that Dr. Gallagher ran at St. George’s followed an analysis he and his associates recently published that suggested efficacy using esophageal cooling in prior reports when the data combined in a meta-analysis (J Interv Card Electrophysiol. 2019 Nov 22. doi: 10.1007/s10840-019-00661-5). They also concluded that the clinical setting required a temperature control device with an enhanced capacity for rapid cooling, which prior studies had lacked. So they turned to a Food and Drug Administration–approved catheter designed for placement in the esophagus for the purpose of either whole-body cooling or warming.
The study randomized a total of 187 patients, but collected follow-up endoscopy at 5-7 days after the ablation procedure on 120 patients, of whom 60 received esophageal cooling and 60 did not. The types of ablations performed on patients in the two study arms were similar, and use of esophageal cooling had no impact on treatment duration or efficacy, either acute and longer term, Dr. Gallagher reported.
Cooling had a marked and statistically significant impact on endoscopically detected thermal injury. Although two patients in the group that underwent cooling had injuries, in one of these cases the injury involved a protocol violation: Radiofrequency ablation mistakenly occurred after the cooling device shut off, and it was during this period when the injury happened. In the second case of thermal injury, blinded scoring judged the injury as grade 2 in severity – an erosion of less than 5 mm – on a nine-item scale that ranged from zero to grade 6, the most severe level denoting a fistula. By contrast, among the 12 patients with thermal injury in the nonprotected subgroup, one patient had a grade 5a lesion denoting a deep ulcer, one had a 4b denoting a superficial ulcer with a clot, and four had a 4a lesion defined as a clean superficial ulcer.
“This is really effective. It’s the first study to show reduced damage without affecting ablation efficacy,” Dr. Gallagher said. He plans to now use this method of esophageal protection routinely for his AFib ablation patients who pay privately, and for patients insured under the national U.K. system once this coverage is approved. Dr. Deneke expressed his interest in also using this approach to esophageal protection, but noted that currently he did not have access to the cooling catheter that Dr. Gallagher used because of regulatory constraints.
The esophageal cooling study was sponsored by Attune Medical, which markets the cooling device. Dr. Gallagher has received research funding from Attune Medical, and has received honoraria as a speaker on behalf of Biosense Webster and Medtronic. Dr. Deneke has been a speaker on behalf of Abbott, Biosense Webster, Biotronik, and Boston Scientific, and his institution has received research funding from Biosense Webster and Securus/Boston Scientific.
NATIONAL HARBOR, MD. – Thermal injury of a patient’s esophagus during radiofrequency catheter ablation of atrial fibrillation is notorious as a relatively common and problematic complication of the procedure, but two new approaches showed promise for substantially cutting the risk of esophageal thermal injury and the potential for the most severe damage: perforation.
One of these innovations is intensive esophageal cooling with a commercially marketed, fluid-chilled catheter placed in a patient’s esophagus during radiofrequency catheter ablation that keeps the inner surface of the esophagus at 4°C. This approach cut the incidence of periprocedural episodes of endoscopically detected esophageal thermal injury from 20% among controls to 3% in patients who had esophageal cooling in a randomized study with 120 patients, Mark M. Gallagher, MD, said at the annual International AF Symposium. The same device can also maintain a temperature on the inner surface of the esophagus of 42 ° C in patients undergoing cryoablation of atrial fibrillation, noted Dr. Gallagher, a cardiac electrophysiologist at St. George’s University Hospitals in London.
A second approach to cutting esophageal damage focuses on modifying the energy delivery with a radiofrequency ablation method known as high-power short-duration (HPSD). As the name says, this strategy uses a relatively high level of radiofrequency energy, 50 watts in the reported experience, for the brief interval of about 7 seconds, ideally delivering an overall Ablation Index of at least 350 but below 360, said Thomas Deneke, MD, an electrophysiologist, professor, and cochief of cardiology at the Heart Center in Bad Neustadt, Germany.
Dr. Deneke and his associates in Bad Neustadt began using this HPSD approach in mid-2019, and by early 2020 they had data from 179 patients who underwent first-time catheter ablation of atrial fibrillation (AFib), all of whom had undergone routine esophageal endoscopy 1-3 days after their treatment. Eight patients (4%) showed evidence of endoscopically detected esophageal lesions (EDEL), including three patients (2%) with an actual esophageal ulcer, and one (0.6%) who developed a perforation that healed after 52 days, Dr. Deneke reported. An additional 55 patients underwent a redo catheter ablation procedure using the HPSD method during this period, and in that group follow-up endoscopy in all patients showed EDEL in two patients (4%). In contrast, during Jan. 2012–May 2019, the same German center treated 2,102 patients who had a first radiofrequency catheter ablation using convention energy levels and treatment times, which resulted in 291 patients having an EDEL (14%), including 94 (4%) with an ulcer, and six patients (0.3%) with an esophageal perforation, he said.
His center’s recent safety experience with HPSD radiofrequncy ablation, compared with the historical controls, suggests that this technique can produce a substantial reduction in esophageal thermal injury, but HPSD has not completely eliminated the risk and hence there is need for continued alertness for this potential complication Dr. Deneke concluded. The HPSD method is also limited by having “a very narrow window” between efficacy at an Ablation Index of 350 and safety when the index remains below 360, he added.
The randomized study that Dr. Gallagher ran at St. George’s followed an analysis he and his associates recently published that suggested efficacy using esophageal cooling in prior reports when the data combined in a meta-analysis (J Interv Card Electrophysiol. 2019 Nov 22. doi: 10.1007/s10840-019-00661-5). They also concluded that the clinical setting required a temperature control device with an enhanced capacity for rapid cooling, which prior studies had lacked. So they turned to a Food and Drug Administration–approved catheter designed for placement in the esophagus for the purpose of either whole-body cooling or warming.
The study randomized a total of 187 patients, but collected follow-up endoscopy at 5-7 days after the ablation procedure on 120 patients, of whom 60 received esophageal cooling and 60 did not. The types of ablations performed on patients in the two study arms were similar, and use of esophageal cooling had no impact on treatment duration or efficacy, either acute and longer term, Dr. Gallagher reported.
Cooling had a marked and statistically significant impact on endoscopically detected thermal injury. Although two patients in the group that underwent cooling had injuries, in one of these cases the injury involved a protocol violation: Radiofrequency ablation mistakenly occurred after the cooling device shut off, and it was during this period when the injury happened. In the second case of thermal injury, blinded scoring judged the injury as grade 2 in severity – an erosion of less than 5 mm – on a nine-item scale that ranged from zero to grade 6, the most severe level denoting a fistula. By contrast, among the 12 patients with thermal injury in the nonprotected subgroup, one patient had a grade 5a lesion denoting a deep ulcer, one had a 4b denoting a superficial ulcer with a clot, and four had a 4a lesion defined as a clean superficial ulcer.
“This is really effective. It’s the first study to show reduced damage without affecting ablation efficacy,” Dr. Gallagher said. He plans to now use this method of esophageal protection routinely for his AFib ablation patients who pay privately, and for patients insured under the national U.K. system once this coverage is approved. Dr. Deneke expressed his interest in also using this approach to esophageal protection, but noted that currently he did not have access to the cooling catheter that Dr. Gallagher used because of regulatory constraints.
The esophageal cooling study was sponsored by Attune Medical, which markets the cooling device. Dr. Gallagher has received research funding from Attune Medical, and has received honoraria as a speaker on behalf of Biosense Webster and Medtronic. Dr. Deneke has been a speaker on behalf of Abbott, Biosense Webster, Biotronik, and Boston Scientific, and his institution has received research funding from Biosense Webster and Securus/Boston Scientific.
THE AF SYMPOSIUM 2020
FDA approves new drug for relapsed/refractory multiple myeloma
The U.S. Food and Drug Administration today approved isatuximab (Sarclisa, Sanofi) in combination with pomalidomide (Revlimid, Celgene) and dexamethasone for the treatment of adult patients with multiple myeloma who have received two or more prior therapies including lenalidomide and a proteasome inhibitor.
Isatuximab is an anti-CD38 monoclonal antibody administered by intravenous infusion that works by helping the immune system attack multiple myeloma cancer cells.
“While there is no cure for multiple myeloma, Sarclisa is now another CD38-directed treatment option added to the list of FDA-approved treatments of patients with multiple myeloma who have progressive disease after previous therapies,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the FDA’s Center for Drug Evaluation and Research.
“In the clinical trial, there was a 40% reduction in the risk of disease progression or death with this therapy,” he added.
The new approval is based on results from ICARIA-MM, an open-label, randomized phase 3 clinical trial of isatuximab among 307 patients in this setting.
In the trial, at a median follow-up of 11.6 months, median progression-free survival was 11.5 months in the isatuximab-pomalidomide-dexamethasone group versus 6.5 months in the pomalidomide-dexamethasone group (hazard ratio, 0.60; P = .001), as reported last year. Overall response rates were 60.4% for the triplet-treated group versus 35.3% for the doublet-treated group.
The most common side effects for isatuximab included neutropenia, infusion-related reactions, pneumonia, upper respiratory tract infection, diarrhea, anemia, lymphopenia, and thrombocytopenia.
Deaths because of treatment-related adverse events were reported for one patient (less than 1%) in the isatuximab-pomalidomide-dexamethasone group (sepsis) and two patients (1%) in the pomalidomide-dexamethasone group (pneumonia and urinary tract infection).
The drug can also cause serious side effects, including IV infusion-related reactions. In the case of a grade 3 or higher reaction, the drug should be permanently discontinued and health care professionals should institute appropriate medical management.
The FDA notes there have been higher incidences of second primary malignancies observed in a controlled clinical trial of patients with multiple myeloma receiving the drug.
The FDA also highlighted that laboratory test interference may be caused by isatuximab and that blood banks should be informed that patients are receiving the drug. Isatuximab may interfere with, for example, antibody screening for patients who need a blood transfusion. Isatuximab may also interfere with the assays used to monitor M-protein, which may impact the determination of complete response.
This article originally appeared on Medscape.com.
The U.S. Food and Drug Administration today approved isatuximab (Sarclisa, Sanofi) in combination with pomalidomide (Revlimid, Celgene) and dexamethasone for the treatment of adult patients with multiple myeloma who have received two or more prior therapies including lenalidomide and a proteasome inhibitor.
Isatuximab is an anti-CD38 monoclonal antibody administered by intravenous infusion that works by helping the immune system attack multiple myeloma cancer cells.
“While there is no cure for multiple myeloma, Sarclisa is now another CD38-directed treatment option added to the list of FDA-approved treatments of patients with multiple myeloma who have progressive disease after previous therapies,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the FDA’s Center for Drug Evaluation and Research.
“In the clinical trial, there was a 40% reduction in the risk of disease progression or death with this therapy,” he added.
The new approval is based on results from ICARIA-MM, an open-label, randomized phase 3 clinical trial of isatuximab among 307 patients in this setting.
In the trial, at a median follow-up of 11.6 months, median progression-free survival was 11.5 months in the isatuximab-pomalidomide-dexamethasone group versus 6.5 months in the pomalidomide-dexamethasone group (hazard ratio, 0.60; P = .001), as reported last year. Overall response rates were 60.4% for the triplet-treated group versus 35.3% for the doublet-treated group.
The most common side effects for isatuximab included neutropenia, infusion-related reactions, pneumonia, upper respiratory tract infection, diarrhea, anemia, lymphopenia, and thrombocytopenia.
Deaths because of treatment-related adverse events were reported for one patient (less than 1%) in the isatuximab-pomalidomide-dexamethasone group (sepsis) and two patients (1%) in the pomalidomide-dexamethasone group (pneumonia and urinary tract infection).
The drug can also cause serious side effects, including IV infusion-related reactions. In the case of a grade 3 or higher reaction, the drug should be permanently discontinued and health care professionals should institute appropriate medical management.
The FDA notes there have been higher incidences of second primary malignancies observed in a controlled clinical trial of patients with multiple myeloma receiving the drug.
The FDA also highlighted that laboratory test interference may be caused by isatuximab and that blood banks should be informed that patients are receiving the drug. Isatuximab may interfere with, for example, antibody screening for patients who need a blood transfusion. Isatuximab may also interfere with the assays used to monitor M-protein, which may impact the determination of complete response.
This article originally appeared on Medscape.com.
The U.S. Food and Drug Administration today approved isatuximab (Sarclisa, Sanofi) in combination with pomalidomide (Revlimid, Celgene) and dexamethasone for the treatment of adult patients with multiple myeloma who have received two or more prior therapies including lenalidomide and a proteasome inhibitor.
Isatuximab is an anti-CD38 monoclonal antibody administered by intravenous infusion that works by helping the immune system attack multiple myeloma cancer cells.
“While there is no cure for multiple myeloma, Sarclisa is now another CD38-directed treatment option added to the list of FDA-approved treatments of patients with multiple myeloma who have progressive disease after previous therapies,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Oncologic Diseases in the FDA’s Center for Drug Evaluation and Research.
“In the clinical trial, there was a 40% reduction in the risk of disease progression or death with this therapy,” he added.
The new approval is based on results from ICARIA-MM, an open-label, randomized phase 3 clinical trial of isatuximab among 307 patients in this setting.
In the trial, at a median follow-up of 11.6 months, median progression-free survival was 11.5 months in the isatuximab-pomalidomide-dexamethasone group versus 6.5 months in the pomalidomide-dexamethasone group (hazard ratio, 0.60; P = .001), as reported last year. Overall response rates were 60.4% for the triplet-treated group versus 35.3% for the doublet-treated group.
The most common side effects for isatuximab included neutropenia, infusion-related reactions, pneumonia, upper respiratory tract infection, diarrhea, anemia, lymphopenia, and thrombocytopenia.
Deaths because of treatment-related adverse events were reported for one patient (less than 1%) in the isatuximab-pomalidomide-dexamethasone group (sepsis) and two patients (1%) in the pomalidomide-dexamethasone group (pneumonia and urinary tract infection).
The drug can also cause serious side effects, including IV infusion-related reactions. In the case of a grade 3 or higher reaction, the drug should be permanently discontinued and health care professionals should institute appropriate medical management.
The FDA notes there have been higher incidences of second primary malignancies observed in a controlled clinical trial of patients with multiple myeloma receiving the drug.
The FDA also highlighted that laboratory test interference may be caused by isatuximab and that blood banks should be informed that patients are receiving the drug. Isatuximab may interfere with, for example, antibody screening for patients who need a blood transfusion. Isatuximab may also interfere with the assays used to monitor M-protein, which may impact the determination of complete response.
This article originally appeared on Medscape.com.
The fate of the ACA now rests with the U.S. Supreme Court
The U.S. Supreme Court has agreed to hear Texas v. California, a closely watched case that could upend the Affordable Care Act.
The justices will hear oral arguments in the case in fall 2020, with a ruling likely in 2021.
The Texas case, consolidated with a similar challenge, stems from a lawsuit by 20 Republican state attorneys general and governors that was filed after Congress zeroed out the ACA’s individual mandate penalty in 2017. The plaintiffs contend the now-valueless mandate is no longer constitutional and thus, the entire ACA should be struck down. Since the Trump administration declined to defend the ACA, a coalition of Democratic attorneys general and governors intervened in the case as defendants.
In 2018, a Texas district court ruled in favor of the plaintiffs and declared the entire health care law invalid. The 5th U.S. Circuit Court of Appeals partially affirmed the district court’s decision, ruling that the mandate was unconstitutional, but sending the case back to the lower court for more analysis on severability. On March 2, the U.S. Supreme Court granted two petitions by the defendants requesting that the high court review the appeals court decision.
The review follows a previous look at the ACA’s mandate by the Supreme Court in 2012. In National Federation of Independent Business v. Sebelius, justices upheld the ACA’s insurance mandate as constitutional, ruling the requirement was authorized by Congress’ power to levy taxes. The vote was 5-4, with Chief Justice John G. Roberts Jr. in agreement with the court’s four more liberal members.
The U.S. Supreme Court has agreed to hear Texas v. California, a closely watched case that could upend the Affordable Care Act.
The justices will hear oral arguments in the case in fall 2020, with a ruling likely in 2021.
The Texas case, consolidated with a similar challenge, stems from a lawsuit by 20 Republican state attorneys general and governors that was filed after Congress zeroed out the ACA’s individual mandate penalty in 2017. The plaintiffs contend the now-valueless mandate is no longer constitutional and thus, the entire ACA should be struck down. Since the Trump administration declined to defend the ACA, a coalition of Democratic attorneys general and governors intervened in the case as defendants.
In 2018, a Texas district court ruled in favor of the plaintiffs and declared the entire health care law invalid. The 5th U.S. Circuit Court of Appeals partially affirmed the district court’s decision, ruling that the mandate was unconstitutional, but sending the case back to the lower court for more analysis on severability. On March 2, the U.S. Supreme Court granted two petitions by the defendants requesting that the high court review the appeals court decision.
The review follows a previous look at the ACA’s mandate by the Supreme Court in 2012. In National Federation of Independent Business v. Sebelius, justices upheld the ACA’s insurance mandate as constitutional, ruling the requirement was authorized by Congress’ power to levy taxes. The vote was 5-4, with Chief Justice John G. Roberts Jr. in agreement with the court’s four more liberal members.
The U.S. Supreme Court has agreed to hear Texas v. California, a closely watched case that could upend the Affordable Care Act.
The justices will hear oral arguments in the case in fall 2020, with a ruling likely in 2021.
The Texas case, consolidated with a similar challenge, stems from a lawsuit by 20 Republican state attorneys general and governors that was filed after Congress zeroed out the ACA’s individual mandate penalty in 2017. The plaintiffs contend the now-valueless mandate is no longer constitutional and thus, the entire ACA should be struck down. Since the Trump administration declined to defend the ACA, a coalition of Democratic attorneys general and governors intervened in the case as defendants.
In 2018, a Texas district court ruled in favor of the plaintiffs and declared the entire health care law invalid. The 5th U.S. Circuit Court of Appeals partially affirmed the district court’s decision, ruling that the mandate was unconstitutional, but sending the case back to the lower court for more analysis on severability. On March 2, the U.S. Supreme Court granted two petitions by the defendants requesting that the high court review the appeals court decision.
The review follows a previous look at the ACA’s mandate by the Supreme Court in 2012. In National Federation of Independent Business v. Sebelius, justices upheld the ACA’s insurance mandate as constitutional, ruling the requirement was authorized by Congress’ power to levy taxes. The vote was 5-4, with Chief Justice John G. Roberts Jr. in agreement with the court’s four more liberal members.
Mortality sevenfold higher post TAVR with severe kidney injury
NATIONAL HARBOR, MD. – Acute kidney injury (AKI), a potentially modifiable risk factor in some cases, predicts increased mortality within the first year after transcatheter aortic valve transplantation (TAVR), according to an analysis of a U.S. registry presented at CRT 2020 sponsored by MedStar Heart & Vascular Institute.
“After adjustment, there are higher rates of all-cause mortality regardless of the severity of AKI,” reported Howard M. Julien, MD, of the University of Pennsylvania, Philadelphia.
Relative to the absence of AKI (stage 0), the hazard ratio for death at 1 year was more than threefold greater (HR, 3.26), even for those with stage 1 AKI. When unadjusted for covariates, it remained more than twice as high (HR, 2.67; P less than .001), Dr. Julien reported.
For stage 3 AKI, the unadjusted risk was more than nine times higher and remained roughly seven times greater after adjustment (HR, 7.04; P less than .001). Stage 2 AKI was linked with an adjusted risk of about the same magnitude.
Drawn from the National Cardiovascular TAVR Registry, which is maintained jointly by the Society of Thoracic Surgeons and the American College of Cardiology, data were analyzed on more than 100,000 TAVRs performed during 2012-2018. A subset of TAVRs performed between January 2016 and June 2018 served as a source of trends in what Dr. Julien described as the “modern era” of this procedure.
The incidence of AKI overall was about 10%, but rates were higher at the earliest time point in the analysis and fell modestly over the study period for all three stages. In a logistic regression analysis, the factors associated with the greatest odds ratio of developing AKI in patients following TAVR were conversion to open heart surgery (OR, 10.84, P less than .001), nonfemoral access (OR, 2.33; P less than .001), anemia (OR, 1.90; P less than .001), general versus moderate sedation (OR, 1.62; P less than .001), diabetes (OR, 1.61; P less than .001), and cardiogenic shock within 24 hours (OR, 1.60; P less than .023).
Other factors with a significant but lower relative risk association with AKI included a high contrast volume (OR, 1.004; P less than .001), use of a self-expanding valve (HR, 1.22; P = .009), severe lung disease (OR, 1.21; P = .043) and prior peripheral artery disease (HR, 1.20; P = .043).
“The message from these data is that there appears to be a cluster of patients who are unstable at the time of their procedure and are more likely to develop the most severe forms of AKI,” Dr. Julien reported.
The higher rate of AKI in patients who have diabetes is “not surprising,” but several of the factors associated with AKI are potentially modifiable. This includes choices in regard to sedation and arterial access. The value of modifying the amount of contrast is less clear, because the volume of contrast was no longer significant after an adjustment with multivariate analysis.
In fact, all of these factors require validation. Dr. Julien warned that neither the cause of AKI nor its temporal relationship to TAVR could be consistently determined from the registry data. In addition, retrospective analyses always include the potential for unrecognized residual confounders.
Still, these data are useful for drawing attention to the fact that AKI is a common complication of TAVR and one that is associated with adverse outcomes, including reduced survival at 1 year.
“The factors taken from these data might be useful to help identify patients who are at risk of the most severe forms of AKI and, hopefully, lead to prevention strategies that take these characteristics into consideration,” Dr. Julien said.
Dr. Julien reported no potential financial conflicts of interest.
NATIONAL HARBOR, MD. – Acute kidney injury (AKI), a potentially modifiable risk factor in some cases, predicts increased mortality within the first year after transcatheter aortic valve transplantation (TAVR), according to an analysis of a U.S. registry presented at CRT 2020 sponsored by MedStar Heart & Vascular Institute.
“After adjustment, there are higher rates of all-cause mortality regardless of the severity of AKI,” reported Howard M. Julien, MD, of the University of Pennsylvania, Philadelphia.
Relative to the absence of AKI (stage 0), the hazard ratio for death at 1 year was more than threefold greater (HR, 3.26), even for those with stage 1 AKI. When unadjusted for covariates, it remained more than twice as high (HR, 2.67; P less than .001), Dr. Julien reported.
For stage 3 AKI, the unadjusted risk was more than nine times higher and remained roughly seven times greater after adjustment (HR, 7.04; P less than .001). Stage 2 AKI was linked with an adjusted risk of about the same magnitude.
Drawn from the National Cardiovascular TAVR Registry, which is maintained jointly by the Society of Thoracic Surgeons and the American College of Cardiology, data were analyzed on more than 100,000 TAVRs performed during 2012-2018. A subset of TAVRs performed between January 2016 and June 2018 served as a source of trends in what Dr. Julien described as the “modern era” of this procedure.
The incidence of AKI overall was about 10%, but rates were higher at the earliest time point in the analysis and fell modestly over the study period for all three stages. In a logistic regression analysis, the factors associated with the greatest odds ratio of developing AKI in patients following TAVR were conversion to open heart surgery (OR, 10.84, P less than .001), nonfemoral access (OR, 2.33; P less than .001), anemia (OR, 1.90; P less than .001), general versus moderate sedation (OR, 1.62; P less than .001), diabetes (OR, 1.61; P less than .001), and cardiogenic shock within 24 hours (OR, 1.60; P less than .023).
Other factors with a significant but lower relative risk association with AKI included a high contrast volume (OR, 1.004; P less than .001), use of a self-expanding valve (HR, 1.22; P = .009), severe lung disease (OR, 1.21; P = .043) and prior peripheral artery disease (HR, 1.20; P = .043).
“The message from these data is that there appears to be a cluster of patients who are unstable at the time of their procedure and are more likely to develop the most severe forms of AKI,” Dr. Julien reported.
The higher rate of AKI in patients who have diabetes is “not surprising,” but several of the factors associated with AKI are potentially modifiable. This includes choices in regard to sedation and arterial access. The value of modifying the amount of contrast is less clear, because the volume of contrast was no longer significant after an adjustment with multivariate analysis.
In fact, all of these factors require validation. Dr. Julien warned that neither the cause of AKI nor its temporal relationship to TAVR could be consistently determined from the registry data. In addition, retrospective analyses always include the potential for unrecognized residual confounders.
Still, these data are useful for drawing attention to the fact that AKI is a common complication of TAVR and one that is associated with adverse outcomes, including reduced survival at 1 year.
“The factors taken from these data might be useful to help identify patients who are at risk of the most severe forms of AKI and, hopefully, lead to prevention strategies that take these characteristics into consideration,” Dr. Julien said.
Dr. Julien reported no potential financial conflicts of interest.
NATIONAL HARBOR, MD. – Acute kidney injury (AKI), a potentially modifiable risk factor in some cases, predicts increased mortality within the first year after transcatheter aortic valve transplantation (TAVR), according to an analysis of a U.S. registry presented at CRT 2020 sponsored by MedStar Heart & Vascular Institute.
“After adjustment, there are higher rates of all-cause mortality regardless of the severity of AKI,” reported Howard M. Julien, MD, of the University of Pennsylvania, Philadelphia.
Relative to the absence of AKI (stage 0), the hazard ratio for death at 1 year was more than threefold greater (HR, 3.26), even for those with stage 1 AKI. When unadjusted for covariates, it remained more than twice as high (HR, 2.67; P less than .001), Dr. Julien reported.
For stage 3 AKI, the unadjusted risk was more than nine times higher and remained roughly seven times greater after adjustment (HR, 7.04; P less than .001). Stage 2 AKI was linked with an adjusted risk of about the same magnitude.
Drawn from the National Cardiovascular TAVR Registry, which is maintained jointly by the Society of Thoracic Surgeons and the American College of Cardiology, data were analyzed on more than 100,000 TAVRs performed during 2012-2018. A subset of TAVRs performed between January 2016 and June 2018 served as a source of trends in what Dr. Julien described as the “modern era” of this procedure.
The incidence of AKI overall was about 10%, but rates were higher at the earliest time point in the analysis and fell modestly over the study period for all three stages. In a logistic regression analysis, the factors associated with the greatest odds ratio of developing AKI in patients following TAVR were conversion to open heart surgery (OR, 10.84, P less than .001), nonfemoral access (OR, 2.33; P less than .001), anemia (OR, 1.90; P less than .001), general versus moderate sedation (OR, 1.62; P less than .001), diabetes (OR, 1.61; P less than .001), and cardiogenic shock within 24 hours (OR, 1.60; P less than .023).
Other factors with a significant but lower relative risk association with AKI included a high contrast volume (OR, 1.004; P less than .001), use of a self-expanding valve (HR, 1.22; P = .009), severe lung disease (OR, 1.21; P = .043) and prior peripheral artery disease (HR, 1.20; P = .043).
“The message from these data is that there appears to be a cluster of patients who are unstable at the time of their procedure and are more likely to develop the most severe forms of AKI,” Dr. Julien reported.
The higher rate of AKI in patients who have diabetes is “not surprising,” but several of the factors associated with AKI are potentially modifiable. This includes choices in regard to sedation and arterial access. The value of modifying the amount of contrast is less clear, because the volume of contrast was no longer significant after an adjustment with multivariate analysis.
In fact, all of these factors require validation. Dr. Julien warned that neither the cause of AKI nor its temporal relationship to TAVR could be consistently determined from the registry data. In addition, retrospective analyses always include the potential for unrecognized residual confounders.
Still, these data are useful for drawing attention to the fact that AKI is a common complication of TAVR and one that is associated with adverse outcomes, including reduced survival at 1 year.
“The factors taken from these data might be useful to help identify patients who are at risk of the most severe forms of AKI and, hopefully, lead to prevention strategies that take these characteristics into consideration,” Dr. Julien said.
Dr. Julien reported no potential financial conflicts of interest.
REPORTING FROM CRT 2020
No sedation fails to improve mortality in mechanically ventilated patients
ORLANDO – For critically ill, according to results of a multicenter, randomized trial.
The lack of sedation did significantly improve certain secondary endpoints, including a reduced number of thromboembolic events and preservation of physical function, according to Palle Toft, PhD, DMSc, of Odense (Denmark) University Hospital.
However, the 90-day mortality rate was 42.4% in the no-sedation group versus 37.0% in the sedation group in the NONSEDA study, which was intended to test the hypothesis that mortality would be lower in the no-sedation group.
That 5.4 percentage point difference between arms in NONSEDA was not statistically significant (P = .65) in results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine and concurrently published in the New England Journal of Medicine.
Yet that mortality trend is in the “opposite direction” of an earlier, single-center trial by Dr. Toft and colleagues, noted Claude Guérin, MD, PhD, in a related editorial that also appeared in the journal. In that earlier study, the reported hospital mortality rates were 36% for no sedation and 47% for sedation with daily interruption.
“The results from this trial [NONSEDA] are important because they arouse concern about omitting sedation in mechanically ventilated patients and reinforce the need to monitor sedation clinically, with the aim of discontinuing it as early as possible or at least interrupting it daily,” Dr. Guérin wrote in his editorial.
That said, the earlier, single-center trial was not statistically powered to show between-group differences in mortality, Dr. Toft and coauthors wrote in their journal article.
In his presentation, Dr. Toft emphasized that light sedation with a wake-up trial was “comparable” with no sedation with regard to mortality.
“I think my main message is that we have to individualize patient treatment,” Dr. Toft told attendees at a late-breaking literature session. “Many patients would benefit from nonsedation, and some would benefit by light sedation with a daily wake-up trial. We have to respect patient autonomy, and try to establish a two-way communication with patients in 2020.”
Sandra L. Kane-Gill, PharmD, treasurer of SCCM and assistant professor of pharmacy and therapeutics at the University of Pittsburgh, said that current SCCM guidelines recommend using light sedation in critically ill, mechanically ventilated adults.
“I think we should stay consistent with what the guidelines are saying,” Dr. Kane-Gill said in an interview. “How you do that may vary, but targeting light sedation is consistent with what the evidence is suggesting in those guidelines.”
The depth of sedation between the no-sedation group in the light sedation group in the present study was not as great as the investigators had anticipated, which may explain the lack of statistically significant difference in mortality, according to Dr. Kane-Gill.
According to the report, 38.4% of patients in the no-sedation group received medication for sedation during their ICU stay, while Richmond Agitation and Sedation Scores increased in both groups, indicating a more alert state in both groups.
The multicenter NONSEDA trial included 700 mechanically ventilated ICU patients randomized either to no sedation or to light sedation, such that the patient was arousable, with daily interruption.
Previous studies have shown that daily interruption of sedation reduced mechanical ventilation duration, ICU stay length, and mortality in comparison with no interruption, the investigators noted.
While mortality at 90 days did not differ significantly between the no-sedation and light-sedation approaches, no sedation reduced thromboembolic events, Dr. Toft said at the meeting. The number of thrombolic events within 90 days was 10 (5%) in the sedation group and 1 (0.5%) in the no-sedation group (P less than .05), according to the reported data.
Likewise, several measures of physical function significantly improved in an a prior defined subgroup of 200 patients, he said. Those measures included hand grip at extubation and ICU discharge, as well as scores on the Barthel Index for Activities of Daily Living.
Nonsedation might improve kidney function, based on other reported outcomes of the study, Dr. Toft said. The number of coma- and delirium-free days was 3.0 in the no-sedation group versus 1.0 in the sedation group (P less than .01), he added.
The benefits of no sedation may extend beyond objective changes in health outcomes, according to Dr. Toft. “The patients are able to communicate with the staff, they might be able to enjoy food, in the evening they can look at the television instead of being sedated – and they can be mobilized and they can write their opinion about the treatments to the doctor, and in this way, you have two-way communication,” he explained in his presentation.
Dr. Toft reported that he had no financial relationships to disclose.
SOURCE: Toft P et al. N Engl J Med. 2019 Feb 16. doi: 10.1056/NEJMoa1906759.
ORLANDO – For critically ill, according to results of a multicenter, randomized trial.
The lack of sedation did significantly improve certain secondary endpoints, including a reduced number of thromboembolic events and preservation of physical function, according to Palle Toft, PhD, DMSc, of Odense (Denmark) University Hospital.
However, the 90-day mortality rate was 42.4% in the no-sedation group versus 37.0% in the sedation group in the NONSEDA study, which was intended to test the hypothesis that mortality would be lower in the no-sedation group.
That 5.4 percentage point difference between arms in NONSEDA was not statistically significant (P = .65) in results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine and concurrently published in the New England Journal of Medicine.
Yet that mortality trend is in the “opposite direction” of an earlier, single-center trial by Dr. Toft and colleagues, noted Claude Guérin, MD, PhD, in a related editorial that also appeared in the journal. In that earlier study, the reported hospital mortality rates were 36% for no sedation and 47% for sedation with daily interruption.
“The results from this trial [NONSEDA] are important because they arouse concern about omitting sedation in mechanically ventilated patients and reinforce the need to monitor sedation clinically, with the aim of discontinuing it as early as possible or at least interrupting it daily,” Dr. Guérin wrote in his editorial.
That said, the earlier, single-center trial was not statistically powered to show between-group differences in mortality, Dr. Toft and coauthors wrote in their journal article.
In his presentation, Dr. Toft emphasized that light sedation with a wake-up trial was “comparable” with no sedation with regard to mortality.
“I think my main message is that we have to individualize patient treatment,” Dr. Toft told attendees at a late-breaking literature session. “Many patients would benefit from nonsedation, and some would benefit by light sedation with a daily wake-up trial. We have to respect patient autonomy, and try to establish a two-way communication with patients in 2020.”
Sandra L. Kane-Gill, PharmD, treasurer of SCCM and assistant professor of pharmacy and therapeutics at the University of Pittsburgh, said that current SCCM guidelines recommend using light sedation in critically ill, mechanically ventilated adults.
“I think we should stay consistent with what the guidelines are saying,” Dr. Kane-Gill said in an interview. “How you do that may vary, but targeting light sedation is consistent with what the evidence is suggesting in those guidelines.”
The depth of sedation between the no-sedation group in the light sedation group in the present study was not as great as the investigators had anticipated, which may explain the lack of statistically significant difference in mortality, according to Dr. Kane-Gill.
According to the report, 38.4% of patients in the no-sedation group received medication for sedation during their ICU stay, while Richmond Agitation and Sedation Scores increased in both groups, indicating a more alert state in both groups.
The multicenter NONSEDA trial included 700 mechanically ventilated ICU patients randomized either to no sedation or to light sedation, such that the patient was arousable, with daily interruption.
Previous studies have shown that daily interruption of sedation reduced mechanical ventilation duration, ICU stay length, and mortality in comparison with no interruption, the investigators noted.
While mortality at 90 days did not differ significantly between the no-sedation and light-sedation approaches, no sedation reduced thromboembolic events, Dr. Toft said at the meeting. The number of thrombolic events within 90 days was 10 (5%) in the sedation group and 1 (0.5%) in the no-sedation group (P less than .05), according to the reported data.
Likewise, several measures of physical function significantly improved in an a prior defined subgroup of 200 patients, he said. Those measures included hand grip at extubation and ICU discharge, as well as scores on the Barthel Index for Activities of Daily Living.
Nonsedation might improve kidney function, based on other reported outcomes of the study, Dr. Toft said. The number of coma- and delirium-free days was 3.0 in the no-sedation group versus 1.0 in the sedation group (P less than .01), he added.
The benefits of no sedation may extend beyond objective changes in health outcomes, according to Dr. Toft. “The patients are able to communicate with the staff, they might be able to enjoy food, in the evening they can look at the television instead of being sedated – and they can be mobilized and they can write their opinion about the treatments to the doctor, and in this way, you have two-way communication,” he explained in his presentation.
Dr. Toft reported that he had no financial relationships to disclose.
SOURCE: Toft P et al. N Engl J Med. 2019 Feb 16. doi: 10.1056/NEJMoa1906759.
ORLANDO – For critically ill, according to results of a multicenter, randomized trial.
The lack of sedation did significantly improve certain secondary endpoints, including a reduced number of thromboembolic events and preservation of physical function, according to Palle Toft, PhD, DMSc, of Odense (Denmark) University Hospital.
However, the 90-day mortality rate was 42.4% in the no-sedation group versus 37.0% in the sedation group in the NONSEDA study, which was intended to test the hypothesis that mortality would be lower in the no-sedation group.
That 5.4 percentage point difference between arms in NONSEDA was not statistically significant (P = .65) in results of the study, presented at the Critical Care Congress sponsored by the Society of Critical Care Medicine and concurrently published in the New England Journal of Medicine.
Yet that mortality trend is in the “opposite direction” of an earlier, single-center trial by Dr. Toft and colleagues, noted Claude Guérin, MD, PhD, in a related editorial that also appeared in the journal. In that earlier study, the reported hospital mortality rates were 36% for no sedation and 47% for sedation with daily interruption.
“The results from this trial [NONSEDA] are important because they arouse concern about omitting sedation in mechanically ventilated patients and reinforce the need to monitor sedation clinically, with the aim of discontinuing it as early as possible or at least interrupting it daily,” Dr. Guérin wrote in his editorial.
That said, the earlier, single-center trial was not statistically powered to show between-group differences in mortality, Dr. Toft and coauthors wrote in their journal article.
In his presentation, Dr. Toft emphasized that light sedation with a wake-up trial was “comparable” with no sedation with regard to mortality.
“I think my main message is that we have to individualize patient treatment,” Dr. Toft told attendees at a late-breaking literature session. “Many patients would benefit from nonsedation, and some would benefit by light sedation with a daily wake-up trial. We have to respect patient autonomy, and try to establish a two-way communication with patients in 2020.”
Sandra L. Kane-Gill, PharmD, treasurer of SCCM and assistant professor of pharmacy and therapeutics at the University of Pittsburgh, said that current SCCM guidelines recommend using light sedation in critically ill, mechanically ventilated adults.
“I think we should stay consistent with what the guidelines are saying,” Dr. Kane-Gill said in an interview. “How you do that may vary, but targeting light sedation is consistent with what the evidence is suggesting in those guidelines.”
The depth of sedation between the no-sedation group in the light sedation group in the present study was not as great as the investigators had anticipated, which may explain the lack of statistically significant difference in mortality, according to Dr. Kane-Gill.
According to the report, 38.4% of patients in the no-sedation group received medication for sedation during their ICU stay, while Richmond Agitation and Sedation Scores increased in both groups, indicating a more alert state in both groups.
The multicenter NONSEDA trial included 700 mechanically ventilated ICU patients randomized either to no sedation or to light sedation, such that the patient was arousable, with daily interruption.
Previous studies have shown that daily interruption of sedation reduced mechanical ventilation duration, ICU stay length, and mortality in comparison with no interruption, the investigators noted.
While mortality at 90 days did not differ significantly between the no-sedation and light-sedation approaches, no sedation reduced thromboembolic events, Dr. Toft said at the meeting. The number of thrombolic events within 90 days was 10 (5%) in the sedation group and 1 (0.5%) in the no-sedation group (P less than .05), according to the reported data.
Likewise, several measures of physical function significantly improved in an a prior defined subgroup of 200 patients, he said. Those measures included hand grip at extubation and ICU discharge, as well as scores on the Barthel Index for Activities of Daily Living.
Nonsedation might improve kidney function, based on other reported outcomes of the study, Dr. Toft said. The number of coma- and delirium-free days was 3.0 in the no-sedation group versus 1.0 in the sedation group (P less than .01), he added.
The benefits of no sedation may extend beyond objective changes in health outcomes, according to Dr. Toft. “The patients are able to communicate with the staff, they might be able to enjoy food, in the evening they can look at the television instead of being sedated – and they can be mobilized and they can write their opinion about the treatments to the doctor, and in this way, you have two-way communication,” he explained in his presentation.
Dr. Toft reported that he had no financial relationships to disclose.
SOURCE: Toft P et al. N Engl J Med. 2019 Feb 16. doi: 10.1056/NEJMoa1906759.
REPORTING FROM CCC49
Conservative care or surgery for rotator cuff tears?
Rotator cuff disease accounts for as many as 65% of shoulder-related visits to physicians’ offices,1 yet the natural course of rotator cuff tears is still not well understood.2 Treatment options are controversial because both conservative and surgical management have been successful. Physical therapy is a durable and reliable treatment option, but there are concerns about long-term progression of the tear.3 Surgical arthroscopic techniques, which result in less morbidity than open surgery, have improved overall surgical care; as such, the rate of rotator cuff procedures has increased significantly.4
Our goal in this article is to provide clinical guidance to the primary care provider. We review management options for rotator cuff injury; summarize considerations for proceeding with conservative or surgical management; and discuss surgical risks and complications.
Conservative management: Who is most likely to benefit?
The choice of treatment for rotator cuff injury depends on a host of variables, including shoulder dominance, duration of symptoms, type of tear (partial or full), age, demands (activity level, occupation, sport), and comorbidities (diabetes, tobacco use). Treatment goals include resolution of pain, normalized range of motion and strength, and restored arm and shoulder function.5
Initial nonoperative management is indicated in patients who
- have a partial-thickness tear (a notable exception is young patients with traumatic injury),6
- have lower functional demands and moderate symptoms, or
- refuse surgery.7
Patients who respond to nonoperative management will, typically, do so within 6 to 12 weeks.5,8
Few randomized, controlled trials have compared conservative and surgical management of rotator cuff tears; furthermore, the findings of these studies have been mixed. Nonoperative management has been shown to be the favored initial treatment for isolated, symptomatic, nontraumatic, supraspinatus tears in older patients.9 In a recent study,10 5-year outcomes were examined in a prospective cohort enrolled in a rotator cuff treatment program: Approximately 75% of patients remained successfully treated with nonoperative management, and clinical outcomes of the operative and nonoperative groups were not significantly different at 5-year follow-up. Investigators concluded that nonoperative treatment is effective for many patients who have a chronic, full-thickness rotator cuff tear.
In a study investigating the treatment of degenerative rotator cuff tear, patients were randomly treated using an operative or nonoperative protocol. No differences in functional outcomes were observed at 1 year after treatment; however, surgical treatment significantly improved subjective parameters of pain and disability.11 A similar study suggested statistically significant improvement in outcomes for patients managed operatively, compared with those treated nonoperatively, but differences in shoulder outcome and the visual analog pain score were small and failed to meet thresholds considered clinically significant. Larger studies, with longer follow-up, are required to determine whether clinical differences between these types of treatment become more evident over time.12
Continue to: A look at nonoperative options and outcomes
A look at nonoperative options and outcomes
Surveillance. Rotator cuff disease of the supraspinatus tendon often results from a degenerative process that progresses to partial and, eventually, full-thickness tearing.8 Once a tear develops, progression is difficult to predict. Many rotator cuff tears grow larger over time; this progression is commonly associated with new or increased pain and weakness, or both. Although asymptomatic progression of a tear is uncommon, many patients—and physicians—are apprehensive about proceeding with nonoperative treatment for a full-thickness tear.8
To diminish such fears, surveillance can include regular assessment of shoulder motion and strength, with consideration of repeat imaging until surgery is performed or the patient is no longer a surgical candidate or interested in surgical treatment.7 Patients and providers need to remain vigilant because tears that are initially graded as repairable can become irreparable if the tendon retracts or there is fatty infiltration of the muscle belly. Results of secondary surgical repair following failed prolonged nonoperative treatment tend to be inferior to results seen in patients who undergo primary tendon repair.7
Analgesics. Simple analgesics, such as acetaminophen, are a low-risk first-line option for pain relief; however, there are limited data on the efficacy of acetaminophen in rotator cuff disease. A topical or oral nonsteroidal anti-inflammatory drug (NSAID), or both, can be considered, but potential contraindications, such as gastrointestinal, renal, and cardiovascular risks, should be monitored.13 Avoid opioids, given the potential for abuse, except during the immediate postoperative period.5
Glucocorticoid injection. Injection of a glucocorticoid drug into the subacromial space should be considered in patients whose pain interferes with sleep, limits activities of daily living, or hinders the ability to participate in physical therapy.5 A recent systematic review demonstrated that NSAIDs and glucocorticoids brought similar pain relief and active abduction at 4 to 6 weeks, but that glucocorticoids were significantly better at achieving remission of symptoms.14 There are no data comparing glucocorticoid preparations (ie, different glucocorticoids or anesthetics, dosages, volumes), and ultrasound guidance does not appear to be necessary for short-term pain relief.15 Note: Repeated injection has been shown to decrease the durability of surgically repaired tendons16; if a patient is a candidate for surgery, repeat injections should be carefully considered—and avoided if possible.
Physical therapy. The goals of physical therapy are activity modification, stretching the shoulder capsule, and strengthening the surrounding musculature (periscapular, rotator cuff, and deltoid). Patients advance through 3 phases of recovery: shoulder mobility, strengthening, and function (ie, joint reactivation to improve shoulder proprioception and coordination).
Continue to: A recent meta-analysis...
A recent meta-analysis17 found comparative evidence on treating rotator cuff tears with physical therapy to be inconclusive. At 1-year follow-up, there was no clinically significant difference between surgery and active physical therapy in either improving the Constant Shoulder Score (an assessment of function) or reducing pain caused by a rotator cuff tear. Therefore, the authors proposed, given the low risk of harm, a conservative approach should be the initial treatment modality for a tear.
A Cochrane review18 examined 60 eligible trials, in which the mean age of patients was 51 years and the mean duration of symptoms, 11 months. Overall, the review concluded that the effects of manual therapy and exercise might be similar to those of glucocorticoid injection and arthroscopic subacromial decompression. The authors noted that this conclusion is based on low-quality evidence, with only 1 study in the review that compared the combination of manual therapy and exercise to placebo.
Other conservative options. Ultrasound, topical nitroglycerin, topical lidocaine, glucocorticoid iontophoresis, transcutaneous electrical nerve stimulation, massage, acupuncture, extracorporeal shockwave therapy, hyaluronic acid, and platelet-rich plasma have been used to treat rotator cuff disease. These modalities require further study, however, to determine their effectiveness for this indication.7,19
Who is a candidate for surgical management?
Although nonoperative treatment is preferred for rotator cuff tendinitis or tendinosis and partial-thickness tears, appropriate management of full-thickness tears is debatable.20 Some surgeons advocate early operative intervention of repairable full-thickness tears to prevent further progression and reduce the risk of long-term dysfunction.
The decision to pursue operative repair depends on
- patient characteristics (age, activity level, comorbidities),
- patient function (amount of disability caused by the tear),
- characteristics of the tear (length, depth, retraction), and
- chronicity of the tear (acuity).
Continue to: TABLE 1...
TABLE 121,22 highlights variables that influence the decision to proceed, or not to proceed, with operative intervention. Because enlargement of a tear usually exacerbates symptoms,23 patients with a tear who are successfully managed nonoperatively should be counseled on the potential of the tear to progress.

What are the surgical options?
Little clinical evidence favors one exposure technique over another. This equivalency has been demonstrated by a systematic review of randomized controlled trials comparing arthroscopic and mini-open rotator cuff repair, which showed no difference in function, pain, or range of motion.24 That conclusion notwithstanding, arthroscopic repair is increasingly popular because it results in less pain, initially, and faster return to work.20
There is controversy among surgeons regarding the choice of fixation technique: Tendons can be secured using 1 or 2 rows of anchors (FIGURE). Advocates of single-row repair cite shorter surgical time, decreased cost, and equivalent outcomes; surgeons who favor double-row, or so-called transosseous-equivalent, repair claim that it provides better restoration of normal anatomy and biomechanical superiority.25,26
Regardless of technique, most patients are immobilized for 4 to 6 weeks postoperatively.27 Physical therapy usually commences within the first week or 2 postop, limited to passive motion for 6 to 12 weeks. Active motion and strengthening of rotator-cuff muscles often is initiated by 3 months postop, although this phase is sometimes delayed because of concern over slow tendon healing. Typically, patients make a full return to sports and manual work at 6 months postop. Patients experience most symptomatic improvement during the first 6 months following surgery, although functional gains can be realized for as long as 2 years after surgery.28
Most torn rotator cuffs can be fixed back to the greater tuberosity, but some chronic, massive, retracted tears lack the mobility to be repaired, or re-tear shortly after repair. Over time, the humeral head in a rotator cuff–deficient shoulder can migrate superiorly to abut the undersurface of the acromion, which can lead to significant glenohumeral osteoarthritis. To prevent or remedy elevation of the humeral head, salvage procedures—debridement, partial repair, spanning graft, tendon transfer, superior capsule reconstruction, balloon arthroplasty, reverse total shoulder replacement—can be used to alleviate pain and restore function. These procedures have significant limitations, however, and usually provide less favorable outcomes than standard repair.29-35
Continue to: Surgical outcomes
Surgical outcomes
Pain, function, and patient satisfaction outcomes following rotator cuff repair are generally favorable: 90% of patients are “happy” 6 months postop.28 Younger populations often have traumatic rotator cuff tears; they generally are interested in returning to sporting activities following their injury. Nearly 85% of younger patients who undergo rotator cuff repair return to sports, and 65.9% return to an equivalent level of play.36
Variables associated with an unfavorable outcome include increasing age, smoking, increased size of the tear, poor tendon quality, hyperlipidemia, workers’ compensation status, fatty infiltration of muscle, obesity, diabetes, and additional procedures to the biceps tendon and acromioclavicular joint performed at the time of rotator cuff repair.37-39 Interestingly, a study concluded that, if a patient expects a good surgical outcome, they are more likely to go on to report a favorable outcome—suggesting that a patient’s expectations might influence their actual outcome.40
Risks and complications
Although rotator cuff surgery has much lower morbidity than other orthopedic surgeries, it is not without risk of complications. If re-tears are excluded, postop complications have been reported in approximately 10% of patients.41 Common complications and their anticipated rate of occurrence are listed in TABLE 2.42-49
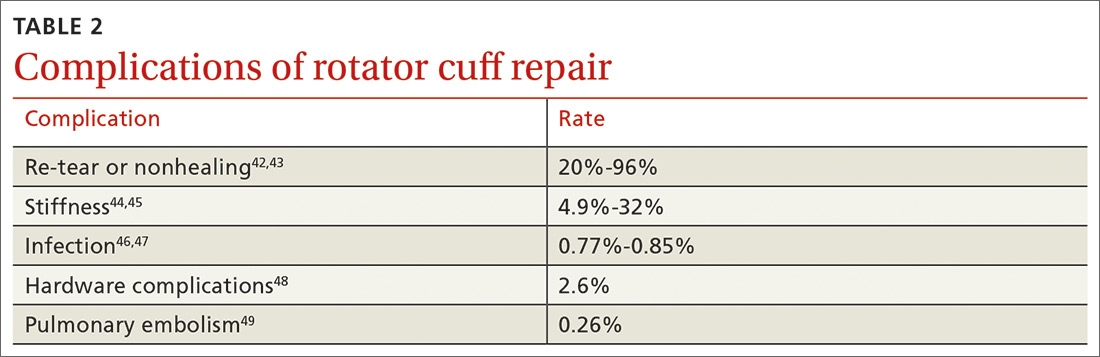
Re-tear of the surgically repaired tendon is the most common postop complication. Published re-tear rates range from 20% to 96%42,43 and generally correlate with initial tear size: A small tear is twice as likely to heal as a massive tear.50 That large range—a span of 76%—results from using a variety of methods to measure re-tear and might not have clinical meaning. A meta-analysis that examined more than 8000 shoulder surgeries reported an overall re-tear rate of 26.6%; however, both patients whose tendons healed and those who re-tore demonstrated clinical improvement.51 In a separate study, patients reported improvement in pain, function, range of motion, and satisfaction regardless of the integrity of the tendon; however, significant improvement in strength was seen only in those whose repair had healed.52
Postop stiffness is more common with arthroscopic repair than with open surgery, and with smaller rather than larger tears.53 Patient variables associated with an increased risk of postop adhesive capsulitis include workers’ compensation insurance, age < 50 years, and preoperative calcific tendonitis or adhesive capsulitis.53 Stiffness generally responds to physical therapy and rarely requires surgical lysis of adhesions or capsular release.
Continue to: Significant injury...
Significant injury to the deltoid muscle has become increasingly uncommon with the advancement of arthroscopic surgery. In traditional open surgery, detachment of the deltoid (and subsequent repair) is required to improve visualization; however, doing so can lead to atrophy and muscle rupture and dehiscence. Deltoid damage occurs in ≤ 60% of open surgeries but is negligible in arthroscopic and mini-open repairs, which involve splitting deltoid fibers to gain exposure of the underlying rotator cuff.54
SIDEBAR
Key takeaways in the management of rotator cuff injury
- Chronic, nontraumatic, and partial-thickness tears respond well to conservative management as first-line treatment. Poor surgical candidates should also be offered a trial of conservative therapy.
- Consider referral for surgical consultation if the patient does not respond to conservative therapy in 6 to 12 weeks; also, patients who have a full-thickness tear and young patients with traumatic injury should be referred for surgical consultation.
- Arthroscopy has become the preferred approach to rotator cuff repair because it is associated with less pain, fewer complications, and faster recovery.
- Patients should be counseled that recovery from surgical repair of a torn rotator cuff takes, on average, 6 months. Some massive or retracted rotator cuff injuries require more extensive procedures that increase healing time.
- Overall, patients are “happy” with rotator cuff repair at 6 months; clinical complications are uncommon, making surgery a suitable option in appropriately selected patients.
CORRESPONDENCE
Cayce Onks, DO, MS, ATC, Penn State Health Milton S. Hershey Medical Center, Penn State College of Medicine, Family and Community Medicine H154, 500 University Drive, PO Box 850, Hershey, PA 17033-0850; [email protected].
1. Vecchio P, Kavanagh R, Hazleman BL, et al. Shoulder pain in a community-based rheumatology clinic. Br J Rheumatol. 1995;34:440-442.
2. Eljabu W, Klinger HM, von Knoch M. The natural history of rotator cuff tears: a systematic review. Arch Orthop Trauma Surg. 2015;135:1055-1061.
3. Dunn WR, Kuhn JE, Sanders R, et al; MOON Shoulder Group. 2013 Neer Award: predictors of failure of nonoperative treatment of chronic, symptomatic, full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2016;25:1303-1311.
4. Colvin AC, Egorova N, Harrison AK, et al. National trends in rotator cuff repair. J Bone Joint Surg Am. 2012;94:227-233.
5. Whittle S, Buchbinder R. In the clinic. Rotator cuff disease. Ann Intern Med. 2015;162:ITC1-ITC15.
6. Lazarides AL, Alentorn-Geli E, Choi JHJ, et al. Rotator cuff tears in young patients: a different disease than rotator cuff tears in elderly patients. J Shoulder Elbow Surg. 2015;24:1834-1843.
7. Petri M, Ettinger M, Brand S, et al. Non-operative management of rotator cuff tears. Open Orthop J. 2016;10:349-356.
8. Schmidt CC, Jarrett CD, Brown BT. Management of rotator cuff tears. J Hand Surg Am. 2015;40:399-408.
9. Kukkonen J, Joukainen A, Lehtinen J, et al. Treatment of nontraumatic rotator cuff tears: a randomized controlled trial with two years of clinical and imaging follow-up. J Bone Joint Surg Am. 2015;97:1729-1737.
10. Boorman RS, More KD, Hollinshead RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27:444-448.
11. Lambers Heerspink FO, van Raay JJ, Koorevaar RCT, et al. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: a randomized controlled trial. J Shoulder Elbow Surg. 2015;24:1274-1281.
12. Piper CC, Hughes AJ, Ma Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27:572-576.
13. Boudreault J, Desmeules F, Roy J-S, et al. The efficacy of oral non-steroidal anti-inflammatory drugs for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. 2014;46:294-306.
14. Zheng X-Q, Li K, Wei Y-D, et al. Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95:1824-1831.
15. Bloom JE, Rischin A, Johnston RV, et al. Image-guided versus blind glucocorticoid injection for shoulder pain. Cochrane Database Syst Rev. 2012;(8):CD009147.
16. Wiggins ME, Fadale PD, Ehrlich MG, et al. Effects of local injection of corticosteroids on the healing of ligaments. A follow-up report. J Bone Joint Surg Am. 1995;77:1682-1691.
17. Ryösä A, Laimi K, Äärimaa V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39:1357-1363.
18. Page MJ, Green S, McBain B, et al. Manual therapy and exercise for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012224.
19. Page MJ, Green S, Mrocki MA, et al. Electrotherapy modalities for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012225.
20. Acevedo DC, Paxton ES, Williams GR, et al. A survey of expert opinion regarding rotator cuff repair. J Bone Joint Surg Am. 2014;96:e123.
21. Pedowitz RA, Yamaguchi K, Ahmad CS, et al. American Academy of Orthopaedic Surgeons Clinical Practice Guideline on: optimizing the management of rotator cuff problems. J Bone Joint Surg Am. 2012;94:163-167.
22. Thorpe A, Hurworth M, O’Sullivan P, et al. Rotator cuff disease: opinion regarding surgical criteria and likely outcome. ANZ J Surg. 2017;87:291-295.
23. Mall NA, Kim HM, Keener JD, et al. Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am. 2010;92:2623-2633.
24. Ji X, Bi C, Wang F, et al. Arthroscopic versus mini-open rotator cuff repair: an up-to-date meta-analysis of randomized controlled trials. Arthroscopy. 2015;31:118-124.
25. Duquin TR, Buyea C, Bisson LJ. Which method of rotator cuff repair leads to the highest rate of structural healing? A systematic review. Am J Sports Med. 2010;38:835-841.
26. Choi S, Kim MK, Kim GM, et al. Factors associated with clinical and structural outcomes after arthroscopic rotator cuff repair with a suture bridge technique in medium, large, and massive tears. J Shoulder Elbow Surg. 2014;23:1675-1681.
27. Shen C, Tang Z-H, Hu J-Z, et al. Does immobilization after arthroscopic rotator cuff repair increase tendon healing? A systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1279-1285.
28. Gulotta LV, Nho SJ, Dodson CC, et al; . Prospective evaluation of arthroscopic rotator cuff repairs at 5 years: part I. Functional outcomes and radiographic healing rates. J Shoulder Elbow Surg. 2011;20:934-940.
29. Liem D, Lengers N, Dedy N, et al. Arthroscopic debridement of massive irreparable rotator cuff tears. Arthroscopy. 2008;24:743-748.
30. Weber SC. Partial rotator cuff repair in massive rotator cuff tears: long-term follow-up. J Shoulder Elbow Surg. 2017;26:e171.
31. Lewington MR, Ferguson DP, Smith TD, et al. Graft utilization in the bridging reconstruction of irreparable rotator cuff tears: a systematic review. Am J Sports Med. 2017;45:3149-3157.
32. Longo UG, Franceschetti E, Petrillo S, et al. Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc Rev. 2011;19:428-437.
33. Noyes MP, Denard PJ. Arthroscopic superior capsular reconstruction: indications and outcomes. Oper Tech Sports Med. 2018;26:29-34.
34. Piekaar RSM, Bouman ICE, van Kampen PM, et al. Early promising outcome following arthroscopic implantation of the subacromial balloon spacer for treating massive rotator cuff tear. Musculoskeletal Surg. 2018;102:247-255.
35. Ek ETH, Neukom L, Catanzaro S, et al. Reverse total shoulder arthroplasty for massive irreparable rotator cuff tears in patients younger than 65 years old: results after five to fifteen years. J Shoulder Elbow Surg. 2013;22:1199-1208.
36. Klouche S, Lefevre N, Herman S, et al. Return to sport after rotator cuff tear repair: a systematic review and meta-analysis. Am J Sports Med. 2016;44:1877-1887.
37. Garcia GH, Liu JN, Wong A, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2017;26:2086-2090.
38. Khair MM, Lehman J, Tsouris N, et al. A systematic review of preoperative fatty infiltration and rotator cuff outcomes. HSS J. 2016;12:170-176.
39. Lambers Heerspink FO, Dorrestijn O, van Raay JJAM, et al. Specific patient-related prognostic factors for rotator cuff repair: a systematic review. J Shoulder Elbow Surg. 2014;23:1073-1080.
40. Henn RF 3rd, Kang L, Tashjian RZ, et al. Patients’ preoperative expectations predict the outcome of rotator cuff repair. J Bone Joint Surg Am. 2007;89:1913-1919.
41. Mansat P, Cofield RH, Kersten TE, et al. Complications of rotator cuff repair. Orthop Clin North Am. 1997;28:205-213.
42. Boileau P, Brassart N, Watkinson DJ, et al. Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am. 2005;87:1229-1240.
43. Galatz LM, Ball CM, Teefey SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219-224.
44. Aydin N, Kocaoglu B, Guven O. Single-row versus double-row arthroscopic rotator cuff repair in small- to medium-sized tears. J Shoulder Elbow Surg. 2010;19:722-725.
45. Peltz CD, Dourte LM, Kuntz AF, et al. The effect of postoperative passive motion on rotator cuff healing in a rat model. J Bone Joint Surg Am. 2009;91:2421-2429.
46. Vopat BG, Lee BJ, DeStefano S, et al. Risk factors for infection after rotator cuff repair. Arthroscopy. 2016;32:428-434.
47. Pauzenberger L, Grieb A, Hexel M, et al. Infections following arthroscopic rotator cuff repair: incidence, risk factors, and prophylaxis. Knee Surg Sports Traumatol Arthrosc. 2017;25:595-601.
48. Randelli P, Spennacchio P, Ragone V, et al. Complications associated with arthroscopic rotator cuff repair: a literature review. Musculoskelet Surg. 2012;96:9-16.
49. Hoxie SC, Sperling JW, Cofield RH. Pulmonary embolism following rotator cuff repair. Int J Shoulder Surg. 2008;2:49-51.
50. Wu XL, Briggs L, Murrell GAC. Intraoperative determinants of rotator cuff repair integrity: an analysis of 500 consecutive repairs. Am J Sports Med. 2012;40:2771-2776.
51. McElvany MD, McGoldrick E, Gee AO, et al. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43:491-500.
52. Yoo JH, Cho NS, Rhee YG. Effect of postoperative repair integrity on health-related quality of life after rotator cuff repair: healed versus retear group. Am J Sports Med. 2013;41;2637-2644.
53. Huberty DP, Schoolfield JD, Brady PC, et al. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy. 2009;25:880-890.
54. Cho NS, Cha SW, Rhee YG. Alterations of the deltoid muscle after open versus arthroscopic rotator cuff repair. Am J Sports Med. 2015;43:2927-2934.
Rotator cuff disease accounts for as many as 65% of shoulder-related visits to physicians’ offices,1 yet the natural course of rotator cuff tears is still not well understood.2 Treatment options are controversial because both conservative and surgical management have been successful. Physical therapy is a durable and reliable treatment option, but there are concerns about long-term progression of the tear.3 Surgical arthroscopic techniques, which result in less morbidity than open surgery, have improved overall surgical care; as such, the rate of rotator cuff procedures has increased significantly.4
Our goal in this article is to provide clinical guidance to the primary care provider. We review management options for rotator cuff injury; summarize considerations for proceeding with conservative or surgical management; and discuss surgical risks and complications.
Conservative management: Who is most likely to benefit?
The choice of treatment for rotator cuff injury depends on a host of variables, including shoulder dominance, duration of symptoms, type of tear (partial or full), age, demands (activity level, occupation, sport), and comorbidities (diabetes, tobacco use). Treatment goals include resolution of pain, normalized range of motion and strength, and restored arm and shoulder function.5
Initial nonoperative management is indicated in patients who
- have a partial-thickness tear (a notable exception is young patients with traumatic injury),6
- have lower functional demands and moderate symptoms, or
- refuse surgery.7
Patients who respond to nonoperative management will, typically, do so within 6 to 12 weeks.5,8
Few randomized, controlled trials have compared conservative and surgical management of rotator cuff tears; furthermore, the findings of these studies have been mixed. Nonoperative management has been shown to be the favored initial treatment for isolated, symptomatic, nontraumatic, supraspinatus tears in older patients.9 In a recent study,10 5-year outcomes were examined in a prospective cohort enrolled in a rotator cuff treatment program: Approximately 75% of patients remained successfully treated with nonoperative management, and clinical outcomes of the operative and nonoperative groups were not significantly different at 5-year follow-up. Investigators concluded that nonoperative treatment is effective for many patients who have a chronic, full-thickness rotator cuff tear.
In a study investigating the treatment of degenerative rotator cuff tear, patients were randomly treated using an operative or nonoperative protocol. No differences in functional outcomes were observed at 1 year after treatment; however, surgical treatment significantly improved subjective parameters of pain and disability.11 A similar study suggested statistically significant improvement in outcomes for patients managed operatively, compared with those treated nonoperatively, but differences in shoulder outcome and the visual analog pain score were small and failed to meet thresholds considered clinically significant. Larger studies, with longer follow-up, are required to determine whether clinical differences between these types of treatment become more evident over time.12
Continue to: A look at nonoperative options and outcomes
A look at nonoperative options and outcomes
Surveillance. Rotator cuff disease of the supraspinatus tendon often results from a degenerative process that progresses to partial and, eventually, full-thickness tearing.8 Once a tear develops, progression is difficult to predict. Many rotator cuff tears grow larger over time; this progression is commonly associated with new or increased pain and weakness, or both. Although asymptomatic progression of a tear is uncommon, many patients—and physicians—are apprehensive about proceeding with nonoperative treatment for a full-thickness tear.8
To diminish such fears, surveillance can include regular assessment of shoulder motion and strength, with consideration of repeat imaging until surgery is performed or the patient is no longer a surgical candidate or interested in surgical treatment.7 Patients and providers need to remain vigilant because tears that are initially graded as repairable can become irreparable if the tendon retracts or there is fatty infiltration of the muscle belly. Results of secondary surgical repair following failed prolonged nonoperative treatment tend to be inferior to results seen in patients who undergo primary tendon repair.7
Analgesics. Simple analgesics, such as acetaminophen, are a low-risk first-line option for pain relief; however, there are limited data on the efficacy of acetaminophen in rotator cuff disease. A topical or oral nonsteroidal anti-inflammatory drug (NSAID), or both, can be considered, but potential contraindications, such as gastrointestinal, renal, and cardiovascular risks, should be monitored.13 Avoid opioids, given the potential for abuse, except during the immediate postoperative period.5
Glucocorticoid injection. Injection of a glucocorticoid drug into the subacromial space should be considered in patients whose pain interferes with sleep, limits activities of daily living, or hinders the ability to participate in physical therapy.5 A recent systematic review demonstrated that NSAIDs and glucocorticoids brought similar pain relief and active abduction at 4 to 6 weeks, but that glucocorticoids were significantly better at achieving remission of symptoms.14 There are no data comparing glucocorticoid preparations (ie, different glucocorticoids or anesthetics, dosages, volumes), and ultrasound guidance does not appear to be necessary for short-term pain relief.15 Note: Repeated injection has been shown to decrease the durability of surgically repaired tendons16; if a patient is a candidate for surgery, repeat injections should be carefully considered—and avoided if possible.
Physical therapy. The goals of physical therapy are activity modification, stretching the shoulder capsule, and strengthening the surrounding musculature (periscapular, rotator cuff, and deltoid). Patients advance through 3 phases of recovery: shoulder mobility, strengthening, and function (ie, joint reactivation to improve shoulder proprioception and coordination).
Continue to: A recent meta-analysis...
A recent meta-analysis17 found comparative evidence on treating rotator cuff tears with physical therapy to be inconclusive. At 1-year follow-up, there was no clinically significant difference between surgery and active physical therapy in either improving the Constant Shoulder Score (an assessment of function) or reducing pain caused by a rotator cuff tear. Therefore, the authors proposed, given the low risk of harm, a conservative approach should be the initial treatment modality for a tear.
A Cochrane review18 examined 60 eligible trials, in which the mean age of patients was 51 years and the mean duration of symptoms, 11 months. Overall, the review concluded that the effects of manual therapy and exercise might be similar to those of glucocorticoid injection and arthroscopic subacromial decompression. The authors noted that this conclusion is based on low-quality evidence, with only 1 study in the review that compared the combination of manual therapy and exercise to placebo.
Other conservative options. Ultrasound, topical nitroglycerin, topical lidocaine, glucocorticoid iontophoresis, transcutaneous electrical nerve stimulation, massage, acupuncture, extracorporeal shockwave therapy, hyaluronic acid, and platelet-rich plasma have been used to treat rotator cuff disease. These modalities require further study, however, to determine their effectiveness for this indication.7,19
Who is a candidate for surgical management?
Although nonoperative treatment is preferred for rotator cuff tendinitis or tendinosis and partial-thickness tears, appropriate management of full-thickness tears is debatable.20 Some surgeons advocate early operative intervention of repairable full-thickness tears to prevent further progression and reduce the risk of long-term dysfunction.
The decision to pursue operative repair depends on
- patient characteristics (age, activity level, comorbidities),
- patient function (amount of disability caused by the tear),
- characteristics of the tear (length, depth, retraction), and
- chronicity of the tear (acuity).
Continue to: TABLE 1...
TABLE 121,22 highlights variables that influence the decision to proceed, or not to proceed, with operative intervention. Because enlargement of a tear usually exacerbates symptoms,23 patients with a tear who are successfully managed nonoperatively should be counseled on the potential of the tear to progress.

What are the surgical options?
Little clinical evidence favors one exposure technique over another. This equivalency has been demonstrated by a systematic review of randomized controlled trials comparing arthroscopic and mini-open rotator cuff repair, which showed no difference in function, pain, or range of motion.24 That conclusion notwithstanding, arthroscopic repair is increasingly popular because it results in less pain, initially, and faster return to work.20
There is controversy among surgeons regarding the choice of fixation technique: Tendons can be secured using 1 or 2 rows of anchors (FIGURE). Advocates of single-row repair cite shorter surgical time, decreased cost, and equivalent outcomes; surgeons who favor double-row, or so-called transosseous-equivalent, repair claim that it provides better restoration of normal anatomy and biomechanical superiority.25,26

Regardless of technique, most patients are immobilized for 4 to 6 weeks postoperatively.27 Physical therapy usually commences within the first week or 2 postop, limited to passive motion for 6 to 12 weeks. Active motion and strengthening of rotator-cuff muscles often is initiated by 3 months postop, although this phase is sometimes delayed because of concern over slow tendon healing. Typically, patients make a full return to sports and manual work at 6 months postop. Patients experience most symptomatic improvement during the first 6 months following surgery, although functional gains can be realized for as long as 2 years after surgery.28
Most torn rotator cuffs can be fixed back to the greater tuberosity, but some chronic, massive, retracted tears lack the mobility to be repaired, or re-tear shortly after repair. Over time, the humeral head in a rotator cuff–deficient shoulder can migrate superiorly to abut the undersurface of the acromion, which can lead to significant glenohumeral osteoarthritis. To prevent or remedy elevation of the humeral head, salvage procedures—debridement, partial repair, spanning graft, tendon transfer, superior capsule reconstruction, balloon arthroplasty, reverse total shoulder replacement—can be used to alleviate pain and restore function. These procedures have significant limitations, however, and usually provide less favorable outcomes than standard repair.29-35
Continue to: Surgical outcomes
Surgical outcomes
Pain, function, and patient satisfaction outcomes following rotator cuff repair are generally favorable: 90% of patients are “happy” 6 months postop.28 Younger populations often have traumatic rotator cuff tears; they generally are interested in returning to sporting activities following their injury. Nearly 85% of younger patients who undergo rotator cuff repair return to sports, and 65.9% return to an equivalent level of play.36
Variables associated with an unfavorable outcome include increasing age, smoking, increased size of the tear, poor tendon quality, hyperlipidemia, workers’ compensation status, fatty infiltration of muscle, obesity, diabetes, and additional procedures to the biceps tendon and acromioclavicular joint performed at the time of rotator cuff repair.37-39 Interestingly, a study concluded that, if a patient expects a good surgical outcome, they are more likely to go on to report a favorable outcome—suggesting that a patient’s expectations might influence their actual outcome.40
Risks and complications
Although rotator cuff surgery has much lower morbidity than other orthopedic surgeries, it is not without risk of complications. If re-tears are excluded, postop complications have been reported in approximately 10% of patients.41 Common complications and their anticipated rate of occurrence are listed in TABLE 2.42-49

Re-tear of the surgically repaired tendon is the most common postop complication. Published re-tear rates range from 20% to 96%42,43 and generally correlate with initial tear size: A small tear is twice as likely to heal as a massive tear.50 That large range—a span of 76%—results from using a variety of methods to measure re-tear and might not have clinical meaning. A meta-analysis that examined more than 8000 shoulder surgeries reported an overall re-tear rate of 26.6%; however, both patients whose tendons healed and those who re-tore demonstrated clinical improvement.51 In a separate study, patients reported improvement in pain, function, range of motion, and satisfaction regardless of the integrity of the tendon; however, significant improvement in strength was seen only in those whose repair had healed.52
Postop stiffness is more common with arthroscopic repair than with open surgery, and with smaller rather than larger tears.53 Patient variables associated with an increased risk of postop adhesive capsulitis include workers’ compensation insurance, age < 50 years, and preoperative calcific tendonitis or adhesive capsulitis.53 Stiffness generally responds to physical therapy and rarely requires surgical lysis of adhesions or capsular release.
Continue to: Significant injury...
Significant injury to the deltoid muscle has become increasingly uncommon with the advancement of arthroscopic surgery. In traditional open surgery, detachment of the deltoid (and subsequent repair) is required to improve visualization; however, doing so can lead to atrophy and muscle rupture and dehiscence. Deltoid damage occurs in ≤ 60% of open surgeries but is negligible in arthroscopic and mini-open repairs, which involve splitting deltoid fibers to gain exposure of the underlying rotator cuff.54
SIDEBAR
Key takeaways in the management of rotator cuff injury
- Chronic, nontraumatic, and partial-thickness tears respond well to conservative management as first-line treatment. Poor surgical candidates should also be offered a trial of conservative therapy.
- Consider referral for surgical consultation if the patient does not respond to conservative therapy in 6 to 12 weeks; also, patients who have a full-thickness tear and young patients with traumatic injury should be referred for surgical consultation.
- Arthroscopy has become the preferred approach to rotator cuff repair because it is associated with less pain, fewer complications, and faster recovery.
- Patients should be counseled that recovery from surgical repair of a torn rotator cuff takes, on average, 6 months. Some massive or retracted rotator cuff injuries require more extensive procedures that increase healing time.
- Overall, patients are “happy” with rotator cuff repair at 6 months; clinical complications are uncommon, making surgery a suitable option in appropriately selected patients.
CORRESPONDENCE
Cayce Onks, DO, MS, ATC, Penn State Health Milton S. Hershey Medical Center, Penn State College of Medicine, Family and Community Medicine H154, 500 University Drive, PO Box 850, Hershey, PA 17033-0850; [email protected].
Rotator cuff disease accounts for as many as 65% of shoulder-related visits to physicians’ offices,1 yet the natural course of rotator cuff tears is still not well understood.2 Treatment options are controversial because both conservative and surgical management have been successful. Physical therapy is a durable and reliable treatment option, but there are concerns about long-term progression of the tear.3 Surgical arthroscopic techniques, which result in less morbidity than open surgery, have improved overall surgical care; as such, the rate of rotator cuff procedures has increased significantly.4
Our goal in this article is to provide clinical guidance to the primary care provider. We review management options for rotator cuff injury; summarize considerations for proceeding with conservative or surgical management; and discuss surgical risks and complications.
Conservative management: Who is most likely to benefit?
The choice of treatment for rotator cuff injury depends on a host of variables, including shoulder dominance, duration of symptoms, type of tear (partial or full), age, demands (activity level, occupation, sport), and comorbidities (diabetes, tobacco use). Treatment goals include resolution of pain, normalized range of motion and strength, and restored arm and shoulder function.5
Initial nonoperative management is indicated in patients who
- have a partial-thickness tear (a notable exception is young patients with traumatic injury),6
- have lower functional demands and moderate symptoms, or
- refuse surgery.7
Patients who respond to nonoperative management will, typically, do so within 6 to 12 weeks.5,8
Few randomized, controlled trials have compared conservative and surgical management of rotator cuff tears; furthermore, the findings of these studies have been mixed. Nonoperative management has been shown to be the favored initial treatment for isolated, symptomatic, nontraumatic, supraspinatus tears in older patients.9 In a recent study,10 5-year outcomes were examined in a prospective cohort enrolled in a rotator cuff treatment program: Approximately 75% of patients remained successfully treated with nonoperative management, and clinical outcomes of the operative and nonoperative groups were not significantly different at 5-year follow-up. Investigators concluded that nonoperative treatment is effective for many patients who have a chronic, full-thickness rotator cuff tear.
In a study investigating the treatment of degenerative rotator cuff tear, patients were randomly treated using an operative or nonoperative protocol. No differences in functional outcomes were observed at 1 year after treatment; however, surgical treatment significantly improved subjective parameters of pain and disability.11 A similar study suggested statistically significant improvement in outcomes for patients managed operatively, compared with those treated nonoperatively, but differences in shoulder outcome and the visual analog pain score were small and failed to meet thresholds considered clinically significant. Larger studies, with longer follow-up, are required to determine whether clinical differences between these types of treatment become more evident over time.12
Continue to: A look at nonoperative options and outcomes
A look at nonoperative options and outcomes
Surveillance. Rotator cuff disease of the supraspinatus tendon often results from a degenerative process that progresses to partial and, eventually, full-thickness tearing.8 Once a tear develops, progression is difficult to predict. Many rotator cuff tears grow larger over time; this progression is commonly associated with new or increased pain and weakness, or both. Although asymptomatic progression of a tear is uncommon, many patients—and physicians—are apprehensive about proceeding with nonoperative treatment for a full-thickness tear.8
To diminish such fears, surveillance can include regular assessment of shoulder motion and strength, with consideration of repeat imaging until surgery is performed or the patient is no longer a surgical candidate or interested in surgical treatment.7 Patients and providers need to remain vigilant because tears that are initially graded as repairable can become irreparable if the tendon retracts or there is fatty infiltration of the muscle belly. Results of secondary surgical repair following failed prolonged nonoperative treatment tend to be inferior to results seen in patients who undergo primary tendon repair.7
Analgesics. Simple analgesics, such as acetaminophen, are a low-risk first-line option for pain relief; however, there are limited data on the efficacy of acetaminophen in rotator cuff disease. A topical or oral nonsteroidal anti-inflammatory drug (NSAID), or both, can be considered, but potential contraindications, such as gastrointestinal, renal, and cardiovascular risks, should be monitored.13 Avoid opioids, given the potential for abuse, except during the immediate postoperative period.5
Glucocorticoid injection. Injection of a glucocorticoid drug into the subacromial space should be considered in patients whose pain interferes with sleep, limits activities of daily living, or hinders the ability to participate in physical therapy.5 A recent systematic review demonstrated that NSAIDs and glucocorticoids brought similar pain relief and active abduction at 4 to 6 weeks, but that glucocorticoids were significantly better at achieving remission of symptoms.14 There are no data comparing glucocorticoid preparations (ie, different glucocorticoids or anesthetics, dosages, volumes), and ultrasound guidance does not appear to be necessary for short-term pain relief.15 Note: Repeated injection has been shown to decrease the durability of surgically repaired tendons16; if a patient is a candidate for surgery, repeat injections should be carefully considered—and avoided if possible.
Physical therapy. The goals of physical therapy are activity modification, stretching the shoulder capsule, and strengthening the surrounding musculature (periscapular, rotator cuff, and deltoid). Patients advance through 3 phases of recovery: shoulder mobility, strengthening, and function (ie, joint reactivation to improve shoulder proprioception and coordination).
Continue to: A recent meta-analysis...
A recent meta-analysis17 found comparative evidence on treating rotator cuff tears with physical therapy to be inconclusive. At 1-year follow-up, there was no clinically significant difference between surgery and active physical therapy in either improving the Constant Shoulder Score (an assessment of function) or reducing pain caused by a rotator cuff tear. Therefore, the authors proposed, given the low risk of harm, a conservative approach should be the initial treatment modality for a tear.
A Cochrane review18 examined 60 eligible trials, in which the mean age of patients was 51 years and the mean duration of symptoms, 11 months. Overall, the review concluded that the effects of manual therapy and exercise might be similar to those of glucocorticoid injection and arthroscopic subacromial decompression. The authors noted that this conclusion is based on low-quality evidence, with only 1 study in the review that compared the combination of manual therapy and exercise to placebo.
Other conservative options. Ultrasound, topical nitroglycerin, topical lidocaine, glucocorticoid iontophoresis, transcutaneous electrical nerve stimulation, massage, acupuncture, extracorporeal shockwave therapy, hyaluronic acid, and platelet-rich plasma have been used to treat rotator cuff disease. These modalities require further study, however, to determine their effectiveness for this indication.7,19
Who is a candidate for surgical management?
Although nonoperative treatment is preferred for rotator cuff tendinitis or tendinosis and partial-thickness tears, appropriate management of full-thickness tears is debatable.20 Some surgeons advocate early operative intervention of repairable full-thickness tears to prevent further progression and reduce the risk of long-term dysfunction.
The decision to pursue operative repair depends on
- patient characteristics (age, activity level, comorbidities),
- patient function (amount of disability caused by the tear),
- characteristics of the tear (length, depth, retraction), and
- chronicity of the tear (acuity).
Continue to: TABLE 1...
TABLE 121,22 highlights variables that influence the decision to proceed, or not to proceed, with operative intervention. Because enlargement of a tear usually exacerbates symptoms,23 patients with a tear who are successfully managed nonoperatively should be counseled on the potential of the tear to progress.

What are the surgical options?
Little clinical evidence favors one exposure technique over another. This equivalency has been demonstrated by a systematic review of randomized controlled trials comparing arthroscopic and mini-open rotator cuff repair, which showed no difference in function, pain, or range of motion.24 That conclusion notwithstanding, arthroscopic repair is increasingly popular because it results in less pain, initially, and faster return to work.20
There is controversy among surgeons regarding the choice of fixation technique: Tendons can be secured using 1 or 2 rows of anchors (FIGURE). Advocates of single-row repair cite shorter surgical time, decreased cost, and equivalent outcomes; surgeons who favor double-row, or so-called transosseous-equivalent, repair claim that it provides better restoration of normal anatomy and biomechanical superiority.25,26

Regardless of technique, most patients are immobilized for 4 to 6 weeks postoperatively.27 Physical therapy usually commences within the first week or 2 postop, limited to passive motion for 6 to 12 weeks. Active motion and strengthening of rotator-cuff muscles often is initiated by 3 months postop, although this phase is sometimes delayed because of concern over slow tendon healing. Typically, patients make a full return to sports and manual work at 6 months postop. Patients experience most symptomatic improvement during the first 6 months following surgery, although functional gains can be realized for as long as 2 years after surgery.28
Most torn rotator cuffs can be fixed back to the greater tuberosity, but some chronic, massive, retracted tears lack the mobility to be repaired, or re-tear shortly after repair. Over time, the humeral head in a rotator cuff–deficient shoulder can migrate superiorly to abut the undersurface of the acromion, which can lead to significant glenohumeral osteoarthritis. To prevent or remedy elevation of the humeral head, salvage procedures—debridement, partial repair, spanning graft, tendon transfer, superior capsule reconstruction, balloon arthroplasty, reverse total shoulder replacement—can be used to alleviate pain and restore function. These procedures have significant limitations, however, and usually provide less favorable outcomes than standard repair.29-35
Continue to: Surgical outcomes
Surgical outcomes
Pain, function, and patient satisfaction outcomes following rotator cuff repair are generally favorable: 90% of patients are “happy” 6 months postop.28 Younger populations often have traumatic rotator cuff tears; they generally are interested in returning to sporting activities following their injury. Nearly 85% of younger patients who undergo rotator cuff repair return to sports, and 65.9% return to an equivalent level of play.36
Variables associated with an unfavorable outcome include increasing age, smoking, increased size of the tear, poor tendon quality, hyperlipidemia, workers’ compensation status, fatty infiltration of muscle, obesity, diabetes, and additional procedures to the biceps tendon and acromioclavicular joint performed at the time of rotator cuff repair.37-39 Interestingly, a study concluded that, if a patient expects a good surgical outcome, they are more likely to go on to report a favorable outcome—suggesting that a patient’s expectations might influence their actual outcome.40
Risks and complications
Although rotator cuff surgery has much lower morbidity than other orthopedic surgeries, it is not without risk of complications. If re-tears are excluded, postop complications have been reported in approximately 10% of patients.41 Common complications and their anticipated rate of occurrence are listed in TABLE 2.42-49

Re-tear of the surgically repaired tendon is the most common postop complication. Published re-tear rates range from 20% to 96%42,43 and generally correlate with initial tear size: A small tear is twice as likely to heal as a massive tear.50 That large range—a span of 76%—results from using a variety of methods to measure re-tear and might not have clinical meaning. A meta-analysis that examined more than 8000 shoulder surgeries reported an overall re-tear rate of 26.6%; however, both patients whose tendons healed and those who re-tore demonstrated clinical improvement.51 In a separate study, patients reported improvement in pain, function, range of motion, and satisfaction regardless of the integrity of the tendon; however, significant improvement in strength was seen only in those whose repair had healed.52
Postop stiffness is more common with arthroscopic repair than with open surgery, and with smaller rather than larger tears.53 Patient variables associated with an increased risk of postop adhesive capsulitis include workers’ compensation insurance, age < 50 years, and preoperative calcific tendonitis or adhesive capsulitis.53 Stiffness generally responds to physical therapy and rarely requires surgical lysis of adhesions or capsular release.
Continue to: Significant injury...
Significant injury to the deltoid muscle has become increasingly uncommon with the advancement of arthroscopic surgery. In traditional open surgery, detachment of the deltoid (and subsequent repair) is required to improve visualization; however, doing so can lead to atrophy and muscle rupture and dehiscence. Deltoid damage occurs in ≤ 60% of open surgeries but is negligible in arthroscopic and mini-open repairs, which involve splitting deltoid fibers to gain exposure of the underlying rotator cuff.54
SIDEBAR
Key takeaways in the management of rotator cuff injury
- Chronic, nontraumatic, and partial-thickness tears respond well to conservative management as first-line treatment. Poor surgical candidates should also be offered a trial of conservative therapy.
- Consider referral for surgical consultation if the patient does not respond to conservative therapy in 6 to 12 weeks; also, patients who have a full-thickness tear and young patients with traumatic injury should be referred for surgical consultation.
- Arthroscopy has become the preferred approach to rotator cuff repair because it is associated with less pain, fewer complications, and faster recovery.
- Patients should be counseled that recovery from surgical repair of a torn rotator cuff takes, on average, 6 months. Some massive or retracted rotator cuff injuries require more extensive procedures that increase healing time.
- Overall, patients are “happy” with rotator cuff repair at 6 months; clinical complications are uncommon, making surgery a suitable option in appropriately selected patients.
CORRESPONDENCE
Cayce Onks, DO, MS, ATC, Penn State Health Milton S. Hershey Medical Center, Penn State College of Medicine, Family and Community Medicine H154, 500 University Drive, PO Box 850, Hershey, PA 17033-0850; [email protected].
1. Vecchio P, Kavanagh R, Hazleman BL, et al. Shoulder pain in a community-based rheumatology clinic. Br J Rheumatol. 1995;34:440-442.
2. Eljabu W, Klinger HM, von Knoch M. The natural history of rotator cuff tears: a systematic review. Arch Orthop Trauma Surg. 2015;135:1055-1061.
3. Dunn WR, Kuhn JE, Sanders R, et al; MOON Shoulder Group. 2013 Neer Award: predictors of failure of nonoperative treatment of chronic, symptomatic, full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2016;25:1303-1311.
4. Colvin AC, Egorova N, Harrison AK, et al. National trends in rotator cuff repair. J Bone Joint Surg Am. 2012;94:227-233.
5. Whittle S, Buchbinder R. In the clinic. Rotator cuff disease. Ann Intern Med. 2015;162:ITC1-ITC15.
6. Lazarides AL, Alentorn-Geli E, Choi JHJ, et al. Rotator cuff tears in young patients: a different disease than rotator cuff tears in elderly patients. J Shoulder Elbow Surg. 2015;24:1834-1843.
7. Petri M, Ettinger M, Brand S, et al. Non-operative management of rotator cuff tears. Open Orthop J. 2016;10:349-356.
8. Schmidt CC, Jarrett CD, Brown BT. Management of rotator cuff tears. J Hand Surg Am. 2015;40:399-408.
9. Kukkonen J, Joukainen A, Lehtinen J, et al. Treatment of nontraumatic rotator cuff tears: a randomized controlled trial with two years of clinical and imaging follow-up. J Bone Joint Surg Am. 2015;97:1729-1737.
10. Boorman RS, More KD, Hollinshead RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27:444-448.
11. Lambers Heerspink FO, van Raay JJ, Koorevaar RCT, et al. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: a randomized controlled trial. J Shoulder Elbow Surg. 2015;24:1274-1281.
12. Piper CC, Hughes AJ, Ma Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27:572-576.
13. Boudreault J, Desmeules F, Roy J-S, et al. The efficacy of oral non-steroidal anti-inflammatory drugs for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. 2014;46:294-306.
14. Zheng X-Q, Li K, Wei Y-D, et al. Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95:1824-1831.
15. Bloom JE, Rischin A, Johnston RV, et al. Image-guided versus blind glucocorticoid injection for shoulder pain. Cochrane Database Syst Rev. 2012;(8):CD009147.
16. Wiggins ME, Fadale PD, Ehrlich MG, et al. Effects of local injection of corticosteroids on the healing of ligaments. A follow-up report. J Bone Joint Surg Am. 1995;77:1682-1691.
17. Ryösä A, Laimi K, Äärimaa V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39:1357-1363.
18. Page MJ, Green S, McBain B, et al. Manual therapy and exercise for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012224.
19. Page MJ, Green S, Mrocki MA, et al. Electrotherapy modalities for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012225.
20. Acevedo DC, Paxton ES, Williams GR, et al. A survey of expert opinion regarding rotator cuff repair. J Bone Joint Surg Am. 2014;96:e123.
21. Pedowitz RA, Yamaguchi K, Ahmad CS, et al. American Academy of Orthopaedic Surgeons Clinical Practice Guideline on: optimizing the management of rotator cuff problems. J Bone Joint Surg Am. 2012;94:163-167.
22. Thorpe A, Hurworth M, O’Sullivan P, et al. Rotator cuff disease: opinion regarding surgical criteria and likely outcome. ANZ J Surg. 2017;87:291-295.
23. Mall NA, Kim HM, Keener JD, et al. Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am. 2010;92:2623-2633.
24. Ji X, Bi C, Wang F, et al. Arthroscopic versus mini-open rotator cuff repair: an up-to-date meta-analysis of randomized controlled trials. Arthroscopy. 2015;31:118-124.
25. Duquin TR, Buyea C, Bisson LJ. Which method of rotator cuff repair leads to the highest rate of structural healing? A systematic review. Am J Sports Med. 2010;38:835-841.
26. Choi S, Kim MK, Kim GM, et al. Factors associated with clinical and structural outcomes after arthroscopic rotator cuff repair with a suture bridge technique in medium, large, and massive tears. J Shoulder Elbow Surg. 2014;23:1675-1681.
27. Shen C, Tang Z-H, Hu J-Z, et al. Does immobilization after arthroscopic rotator cuff repair increase tendon healing? A systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1279-1285.
28. Gulotta LV, Nho SJ, Dodson CC, et al; . Prospective evaluation of arthroscopic rotator cuff repairs at 5 years: part I. Functional outcomes and radiographic healing rates. J Shoulder Elbow Surg. 2011;20:934-940.
29. Liem D, Lengers N, Dedy N, et al. Arthroscopic debridement of massive irreparable rotator cuff tears. Arthroscopy. 2008;24:743-748.
30. Weber SC. Partial rotator cuff repair in massive rotator cuff tears: long-term follow-up. J Shoulder Elbow Surg. 2017;26:e171.
31. Lewington MR, Ferguson DP, Smith TD, et al. Graft utilization in the bridging reconstruction of irreparable rotator cuff tears: a systematic review. Am J Sports Med. 2017;45:3149-3157.
32. Longo UG, Franceschetti E, Petrillo S, et al. Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc Rev. 2011;19:428-437.
33. Noyes MP, Denard PJ. Arthroscopic superior capsular reconstruction: indications and outcomes. Oper Tech Sports Med. 2018;26:29-34.
34. Piekaar RSM, Bouman ICE, van Kampen PM, et al. Early promising outcome following arthroscopic implantation of the subacromial balloon spacer for treating massive rotator cuff tear. Musculoskeletal Surg. 2018;102:247-255.
35. Ek ETH, Neukom L, Catanzaro S, et al. Reverse total shoulder arthroplasty for massive irreparable rotator cuff tears in patients younger than 65 years old: results after five to fifteen years. J Shoulder Elbow Surg. 2013;22:1199-1208.
36. Klouche S, Lefevre N, Herman S, et al. Return to sport after rotator cuff tear repair: a systematic review and meta-analysis. Am J Sports Med. 2016;44:1877-1887.
37. Garcia GH, Liu JN, Wong A, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2017;26:2086-2090.
38. Khair MM, Lehman J, Tsouris N, et al. A systematic review of preoperative fatty infiltration and rotator cuff outcomes. HSS J. 2016;12:170-176.
39. Lambers Heerspink FO, Dorrestijn O, van Raay JJAM, et al. Specific patient-related prognostic factors for rotator cuff repair: a systematic review. J Shoulder Elbow Surg. 2014;23:1073-1080.
40. Henn RF 3rd, Kang L, Tashjian RZ, et al. Patients’ preoperative expectations predict the outcome of rotator cuff repair. J Bone Joint Surg Am. 2007;89:1913-1919.
41. Mansat P, Cofield RH, Kersten TE, et al. Complications of rotator cuff repair. Orthop Clin North Am. 1997;28:205-213.
42. Boileau P, Brassart N, Watkinson DJ, et al. Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am. 2005;87:1229-1240.
43. Galatz LM, Ball CM, Teefey SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219-224.
44. Aydin N, Kocaoglu B, Guven O. Single-row versus double-row arthroscopic rotator cuff repair in small- to medium-sized tears. J Shoulder Elbow Surg. 2010;19:722-725.
45. Peltz CD, Dourte LM, Kuntz AF, et al. The effect of postoperative passive motion on rotator cuff healing in a rat model. J Bone Joint Surg Am. 2009;91:2421-2429.
46. Vopat BG, Lee BJ, DeStefano S, et al. Risk factors for infection after rotator cuff repair. Arthroscopy. 2016;32:428-434.
47. Pauzenberger L, Grieb A, Hexel M, et al. Infections following arthroscopic rotator cuff repair: incidence, risk factors, and prophylaxis. Knee Surg Sports Traumatol Arthrosc. 2017;25:595-601.
48. Randelli P, Spennacchio P, Ragone V, et al. Complications associated with arthroscopic rotator cuff repair: a literature review. Musculoskelet Surg. 2012;96:9-16.
49. Hoxie SC, Sperling JW, Cofield RH. Pulmonary embolism following rotator cuff repair. Int J Shoulder Surg. 2008;2:49-51.
50. Wu XL, Briggs L, Murrell GAC. Intraoperative determinants of rotator cuff repair integrity: an analysis of 500 consecutive repairs. Am J Sports Med. 2012;40:2771-2776.
51. McElvany MD, McGoldrick E, Gee AO, et al. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43:491-500.
52. Yoo JH, Cho NS, Rhee YG. Effect of postoperative repair integrity on health-related quality of life after rotator cuff repair: healed versus retear group. Am J Sports Med. 2013;41;2637-2644.
53. Huberty DP, Schoolfield JD, Brady PC, et al. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy. 2009;25:880-890.
54. Cho NS, Cha SW, Rhee YG. Alterations of the deltoid muscle after open versus arthroscopic rotator cuff repair. Am J Sports Med. 2015;43:2927-2934.
1. Vecchio P, Kavanagh R, Hazleman BL, et al. Shoulder pain in a community-based rheumatology clinic. Br J Rheumatol. 1995;34:440-442.
2. Eljabu W, Klinger HM, von Knoch M. The natural history of rotator cuff tears: a systematic review. Arch Orthop Trauma Surg. 2015;135:1055-1061.
3. Dunn WR, Kuhn JE, Sanders R, et al; MOON Shoulder Group. 2013 Neer Award: predictors of failure of nonoperative treatment of chronic, symptomatic, full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2016;25:1303-1311.
4. Colvin AC, Egorova N, Harrison AK, et al. National trends in rotator cuff repair. J Bone Joint Surg Am. 2012;94:227-233.
5. Whittle S, Buchbinder R. In the clinic. Rotator cuff disease. Ann Intern Med. 2015;162:ITC1-ITC15.
6. Lazarides AL, Alentorn-Geli E, Choi JHJ, et al. Rotator cuff tears in young patients: a different disease than rotator cuff tears in elderly patients. J Shoulder Elbow Surg. 2015;24:1834-1843.
7. Petri M, Ettinger M, Brand S, et al. Non-operative management of rotator cuff tears. Open Orthop J. 2016;10:349-356.
8. Schmidt CC, Jarrett CD, Brown BT. Management of rotator cuff tears. J Hand Surg Am. 2015;40:399-408.
9. Kukkonen J, Joukainen A, Lehtinen J, et al. Treatment of nontraumatic rotator cuff tears: a randomized controlled trial with two years of clinical and imaging follow-up. J Bone Joint Surg Am. 2015;97:1729-1737.
10. Boorman RS, More KD, Hollinshead RM, et al. What happens to patients when we do not repair their cuff tears? Five-year rotator cuff quality-of-life index outcomes following nonoperative treatment of patients with full-thickness rotator cuff tears. J Shoulder Elbow Surg. 2018;27:444-448.
11. Lambers Heerspink FO, van Raay JJ, Koorevaar RCT, et al. Comparing surgical repair with conservative treatment for degenerative rotator cuff tears: a randomized controlled trial. J Shoulder Elbow Surg. 2015;24:1274-1281.
12. Piper CC, Hughes AJ, Ma Y, et al. Operative versus nonoperative treatment for the management of full-thickness rotator cuff tears: a systematic review and meta-analysis. J Shoulder Elbow Surg. 2018;27:572-576.
13. Boudreault J, Desmeules F, Roy J-S, et al. The efficacy of oral non-steroidal anti-inflammatory drugs for rotator cuff tendinopathy: a systematic review and meta-analysis. J Rehabil Med. 2014;46:294-306.
14. Zheng X-Q, Li K, Wei Y-D, et al. Nonsteroidal anti-inflammatory drugs versus corticosteroid for treatment of shoulder pain: a systematic review and meta-analysis. Arch Phys Med Rehabil. 2014;95:1824-1831.
15. Bloom JE, Rischin A, Johnston RV, et al. Image-guided versus blind glucocorticoid injection for shoulder pain. Cochrane Database Syst Rev. 2012;(8):CD009147.
16. Wiggins ME, Fadale PD, Ehrlich MG, et al. Effects of local injection of corticosteroids on the healing of ligaments. A follow-up report. J Bone Joint Surg Am. 1995;77:1682-1691.
17. Ryösä A, Laimi K, Äärimaa V, et al. Surgery or conservative treatment for rotator cuff tear: a meta-analysis. Disabil Rehabil. 2017;39:1357-1363.
18. Page MJ, Green S, McBain B, et al. Manual therapy and exercise for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012224.
19. Page MJ, Green S, Mrocki MA, et al. Electrotherapy modalities for rotator cuff disease. Cochrane Database Syst Rev. 2016;(6):CD012225.
20. Acevedo DC, Paxton ES, Williams GR, et al. A survey of expert opinion regarding rotator cuff repair. J Bone Joint Surg Am. 2014;96:e123.
21. Pedowitz RA, Yamaguchi K, Ahmad CS, et al. American Academy of Orthopaedic Surgeons Clinical Practice Guideline on: optimizing the management of rotator cuff problems. J Bone Joint Surg Am. 2012;94:163-167.
22. Thorpe A, Hurworth M, O’Sullivan P, et al. Rotator cuff disease: opinion regarding surgical criteria and likely outcome. ANZ J Surg. 2017;87:291-295.
23. Mall NA, Kim HM, Keener JD, et al. Symptomatic progression of asymptomatic rotator cuff tears: a prospective study of clinical and sonographic variables. J Bone Joint Surg Am. 2010;92:2623-2633.
24. Ji X, Bi C, Wang F, et al. Arthroscopic versus mini-open rotator cuff repair: an up-to-date meta-analysis of randomized controlled trials. Arthroscopy. 2015;31:118-124.
25. Duquin TR, Buyea C, Bisson LJ. Which method of rotator cuff repair leads to the highest rate of structural healing? A systematic review. Am J Sports Med. 2010;38:835-841.
26. Choi S, Kim MK, Kim GM, et al. Factors associated with clinical and structural outcomes after arthroscopic rotator cuff repair with a suture bridge technique in medium, large, and massive tears. J Shoulder Elbow Surg. 2014;23:1675-1681.
27. Shen C, Tang Z-H, Hu J-Z, et al. Does immobilization after arthroscopic rotator cuff repair increase tendon healing? A systematic review and meta-analysis. Arch Orthop Trauma Surg. 2014;134:1279-1285.
28. Gulotta LV, Nho SJ, Dodson CC, et al; . Prospective evaluation of arthroscopic rotator cuff repairs at 5 years: part I. Functional outcomes and radiographic healing rates. J Shoulder Elbow Surg. 2011;20:934-940.
29. Liem D, Lengers N, Dedy N, et al. Arthroscopic debridement of massive irreparable rotator cuff tears. Arthroscopy. 2008;24:743-748.
30. Weber SC. Partial rotator cuff repair in massive rotator cuff tears: long-term follow-up. J Shoulder Elbow Surg. 2017;26:e171.
31. Lewington MR, Ferguson DP, Smith TD, et al. Graft utilization in the bridging reconstruction of irreparable rotator cuff tears: a systematic review. Am J Sports Med. 2017;45:3149-3157.
32. Longo UG, Franceschetti E, Petrillo S, et al. Latissimus dorsi tendon transfer for massive irreparable rotator cuff tears: a systematic review. Sports Med Arthrosc Rev. 2011;19:428-437.
33. Noyes MP, Denard PJ. Arthroscopic superior capsular reconstruction: indications and outcomes. Oper Tech Sports Med. 2018;26:29-34.
34. Piekaar RSM, Bouman ICE, van Kampen PM, et al. Early promising outcome following arthroscopic implantation of the subacromial balloon spacer for treating massive rotator cuff tear. Musculoskeletal Surg. 2018;102:247-255.
35. Ek ETH, Neukom L, Catanzaro S, et al. Reverse total shoulder arthroplasty for massive irreparable rotator cuff tears in patients younger than 65 years old: results after five to fifteen years. J Shoulder Elbow Surg. 2013;22:1199-1208.
36. Klouche S, Lefevre N, Herman S, et al. Return to sport after rotator cuff tear repair: a systematic review and meta-analysis. Am J Sports Med. 2016;44:1877-1887.
37. Garcia GH, Liu JN, Wong A, et al. Hyperlipidemia increases the risk of retear after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2017;26:2086-2090.
38. Khair MM, Lehman J, Tsouris N, et al. A systematic review of preoperative fatty infiltration and rotator cuff outcomes. HSS J. 2016;12:170-176.
39. Lambers Heerspink FO, Dorrestijn O, van Raay JJAM, et al. Specific patient-related prognostic factors for rotator cuff repair: a systematic review. J Shoulder Elbow Surg. 2014;23:1073-1080.
40. Henn RF 3rd, Kang L, Tashjian RZ, et al. Patients’ preoperative expectations predict the outcome of rotator cuff repair. J Bone Joint Surg Am. 2007;89:1913-1919.
41. Mansat P, Cofield RH, Kersten TE, et al. Complications of rotator cuff repair. Orthop Clin North Am. 1997;28:205-213.
42. Boileau P, Brassart N, Watkinson DJ, et al. Arthroscopic repair of full-thickness tears of the supraspinatus: does the tendon really heal? J Bone Joint Surg Am. 2005;87:1229-1240.
43. Galatz LM, Ball CM, Teefey SA, et al. The outcome and repair integrity of completely arthroscopically repaired large and massive rotator cuff tears. J Bone Joint Surg Am. 2004;86:219-224.
44. Aydin N, Kocaoglu B, Guven O. Single-row versus double-row arthroscopic rotator cuff repair in small- to medium-sized tears. J Shoulder Elbow Surg. 2010;19:722-725.
45. Peltz CD, Dourte LM, Kuntz AF, et al. The effect of postoperative passive motion on rotator cuff healing in a rat model. J Bone Joint Surg Am. 2009;91:2421-2429.
46. Vopat BG, Lee BJ, DeStefano S, et al. Risk factors for infection after rotator cuff repair. Arthroscopy. 2016;32:428-434.
47. Pauzenberger L, Grieb A, Hexel M, et al. Infections following arthroscopic rotator cuff repair: incidence, risk factors, and prophylaxis. Knee Surg Sports Traumatol Arthrosc. 2017;25:595-601.
48. Randelli P, Spennacchio P, Ragone V, et al. Complications associated with arthroscopic rotator cuff repair: a literature review. Musculoskelet Surg. 2012;96:9-16.
49. Hoxie SC, Sperling JW, Cofield RH. Pulmonary embolism following rotator cuff repair. Int J Shoulder Surg. 2008;2:49-51.
50. Wu XL, Briggs L, Murrell GAC. Intraoperative determinants of rotator cuff repair integrity: an analysis of 500 consecutive repairs. Am J Sports Med. 2012;40:2771-2776.
51. McElvany MD, McGoldrick E, Gee AO, et al. Rotator cuff repair: published evidence on factors associated with repair integrity and clinical outcome. Am J Sports Med. 2015;43:491-500.
52. Yoo JH, Cho NS, Rhee YG. Effect of postoperative repair integrity on health-related quality of life after rotator cuff repair: healed versus retear group. Am J Sports Med. 2013;41;2637-2644.
53. Huberty DP, Schoolfield JD, Brady PC, et al. Incidence and treatment of postoperative stiffness following arthroscopic rotator cuff repair. Arthroscopy. 2009;25:880-890.
54. Cho NS, Cha SW, Rhee YG. Alterations of the deltoid muscle after open versus arthroscopic rotator cuff repair. Am J Sports Med. 2015;43:2927-2934.
PRACTICE RECOMMENDATIONS
› Offer a trial of conservative management to patients with chronic, nontraumatic, or partial-thickness rotator cuff injury and to those who are poor surgical candidates. B
› Counsel patients that the rate of surgical complications is low and outcomes are favorable in properly selected patients for operative repair of rotator cuff tear. B
Strength of recommendation (SOR)
A Good-quality patient-oriented evidence
B Inconsistent or limited-quality patient-oriented evidence
C Consensus, usual practice, opinion, disease-oriented evidence, case series
Screen all adults for hepatitis C, says USPSTF
Adults aged 18-79 years should be screened for hepatitis C virus infection, according to an updated grade B recommendation from the U.S. Preventive Services Task Force.
Cases of acute hepatitis C virus (HCV) infection have spiked in the last decade, in part because of increased use of injection drugs and in part because of better surveillance, Douglas K. Owens, MD, of Stanford (Calif.) University, and colleagues wrote in the recommendation statement published in JAMA.
The recommendation applies to all asymptomatic adults aged 18-79 years without known liver disease, and expands on the 2013 recommendation to screen adults born between 1945 and 1965. The grade B designation means that the task force concluded with moderate certainty that HCV screening for adults aged 18-79 years had “substantial net benefit.”
The recommendations are based on an evidence report including 8 randomized, controlled trials, 48 other treatment studies, and 33 cohort studies published through February 2019 for a total of 179,230 individuals.
The screening is a one-time procedure for most adults, according to the task force, but clinicians should periodically screen individuals at increased risk, such as those with a past or current history of injection drug use. In addition, clinicians should consider screening individuals at increased risk who are above or below the recommended age range.
Although the task force identified no direct evidence on the benefit of screening for HCV infection in asymptomatic adults, a notable finding was that the newer direct-acting antiviral (DAA) regimens are sufficiently effective to support the expanded screening recommendation, they said. However, clinicians should inform patients that screening is voluntary and conducted only with the patient’s knowledge. Clinicians should educate patients about hepatitis C and give them an opportunity to ask questions and to make a decision about screening, according to the task force.
In the evidence report, a total of 49 studies including 10,181 individuals showed DAA treatment associated with pooled sustained virologic response rates greater than 95% across all virus genotypes, and a short-term serious adverse event rate of 1.9%. In addition, sustained virologic response following an antiviral therapy was associated with a reduction in risk of all-cause mortality (pooled hazard ratio 0.40) and of hepatocellular carcinoma (pooled HR 0.29) compared with cases of no sustained virologic response.
The evidence report findings were limited by several factors, including the relatively small number of randomized trials involving current DAA treatments, limited data on baseline symptoms, limited data on adolescents, and limited evidence on potential long-term harms of DAA therapy, noted Richard Chou, MD, of Oregon Health & Science University, Portland, and colleagues. However, new pooled evidence “indicates that SVR rates with currently recommended all-oral DAA regimens are substantially higher (more than 95%) than with interferon-based therapies evaluated in the prior review (68%-78%),” they said.
Several editorials were published concurrently with the recommendation.
In an editorial published in JAMA, Camilla S. Graham, MD, of Harvard Medical School, Boston, and Stacey Trooskin, MD, of the University of Pennsylvania, Philadelphia, wrote that the new recommendation reflects changes in hepatitis C virus management.
“With the approvals of sofosbuvir and simeprevir in 2013, patients with hepatitis C, a chronic viral illness associated with the deaths of more U.S. patients than the next 60 reportable infectious diseases combined, including HIV and tuberculosis, could expect a greater than 90% rate of achieving sustained virologic response (SVR, defined as undetectable HCV levels 12 weeks or longer after treatment completion, which is consistent with virologic cure of HCV infection) following 12 weeks of treatment,” they said.
These medications are effective but expensive; however, the combination of the availability of generic medications and the ongoing opioid epidemic in the United States are important contributors to the expanded recommendations, which “are welcome,” and may help meeting WHO 2030 targets for reducing new HCV infections, they said.
Dr. Graham disclosed personal fees from UpToDate. Dr. Trooskin disclosed grants from Gilead Sciences and personal fees from Merck, AbbVie, and Gilead Sciences.
In an editorial published in JAMA Internal Medicine, Jennifer C. Price, MD, and Danielle Brandman, MD, both of the University of California, San Francisco, wrote that “the advancements in HCV diagnosis and treatment have been extraordinary,” but that the new recommendation does not go far enough. “Implementation of HCV screening and linkage to treatment requires large-scale coordinated efforts, innovation, and resources. For example, point-of-care HCV RNA testing would enable scale-up of HCV screening and confirmatory testing among individuals at greatest risk of HCV infection,” they said. “Additionally, barriers remain between diagnosis and treatment, such as access to a health care provider who can treat HCV and authorization to receive affordable DAAs,” they noted. “Although the USPSTF HCV screening recommendation is a step forward for controlling HCV infection in the U.S., it will take a coordinated and funded effort to ensure that the anticipated benefits are realized,” they concluded.
Dr. Price disclosed research funding from Gilead Sciences and Merck. Dr. Brandman disclosed research funding from Gilead Sciences, Pfizer, Conatus, Allergan, and Grifols, as well as personal fees from Alnylam.
In an editorial published in JAMA Network Open, Eli S. Rosenberg, PhD, of the University at Albany (N.Y.) School of Public Health, and Joshua A. Barocas, MD, of Boston University, emphasized the need to change the stigma surrounding HCV infection in the United States.
“Given the changing epidemiology of HCV infection, new public health priorities, advancements in treatment, and unmet diagnostic needs, it is wise to periodically reevaluate screening recommendations to ensure that they are maximally addressing these areas and patients’ individual needs,” they said. “The Affordable Care Act requires private insurers and Medicaid to cover preventive services recommended by the USPSTF with a grade of A or B with no cost sharing (i.e., no deductible or copayment),” they noted. Although the new recommendation for one-time screening will likely identify more cases, improve outcomes, and reduce deaths, the editorialists cautioned that “one-time screening should not be interpreted like catch-up vaccinations, whereby we immunize someone at any age for hepatitis B virus, for example, and they are then immunized for the remainder of their life,” and that reassessments are needed, especially for younger adults.
In addition, they emphasized the need to reduce the stigma surrounding HCV and allow for recommendations based on risk, rather than age. “We have forced the USPSTF to adopt age-based screening recommendations because we, as a society, have created a culture in which we have stigmatized these behaviors and we, as practitioners, have proven to be inadequate at eliciting HCV risk behaviors,” they said. “Our responsibility as a society and practice community is to address structural and individual factors that limit our ability to most precisely address the needs of our patients and truly move toward HCV elimination,” they concluded.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The task force researchers had no financial conflicts to disclose.
SOURCES: Owens DK et al. JAMA. 2020 Mar 2. doi: 10.1001/jama.2020.1123; Chou R et al. JAMA. 2020 Mar 2. doi: 10.1001/jama.2019.20788; Graham CS, Trooskin S. JAMA. 2020 Mar 2. doi: 10.1001/jama.2019.22313; Price JC and Brandman D. JAMA Intern Med. 2020 Mar 2. doi: 10.1001/jamainternmed.2019.7334; Rosenberg ES, Barocas JA. JAMA Network Open. 2020 Mar 2. doi: 10.1001/jamanetworkopen.2020.0538.
Adults aged 18-79 years should be screened for hepatitis C virus infection, according to an updated grade B recommendation from the U.S. Preventive Services Task Force.
Cases of acute hepatitis C virus (HCV) infection have spiked in the last decade, in part because of increased use of injection drugs and in part because of better surveillance, Douglas K. Owens, MD, of Stanford (Calif.) University, and colleagues wrote in the recommendation statement published in JAMA.
The recommendation applies to all asymptomatic adults aged 18-79 years without known liver disease, and expands on the 2013 recommendation to screen adults born between 1945 and 1965. The grade B designation means that the task force concluded with moderate certainty that HCV screening for adults aged 18-79 years had “substantial net benefit.”
The recommendations are based on an evidence report including 8 randomized, controlled trials, 48 other treatment studies, and 33 cohort studies published through February 2019 for a total of 179,230 individuals.
The screening is a one-time procedure for most adults, according to the task force, but clinicians should periodically screen individuals at increased risk, such as those with a past or current history of injection drug use. In addition, clinicians should consider screening individuals at increased risk who are above or below the recommended age range.
Although the task force identified no direct evidence on the benefit of screening for HCV infection in asymptomatic adults, a notable finding was that the newer direct-acting antiviral (DAA) regimens are sufficiently effective to support the expanded screening recommendation, they said. However, clinicians should inform patients that screening is voluntary and conducted only with the patient’s knowledge. Clinicians should educate patients about hepatitis C and give them an opportunity to ask questions and to make a decision about screening, according to the task force.
In the evidence report, a total of 49 studies including 10,181 individuals showed DAA treatment associated with pooled sustained virologic response rates greater than 95% across all virus genotypes, and a short-term serious adverse event rate of 1.9%. In addition, sustained virologic response following an antiviral therapy was associated with a reduction in risk of all-cause mortality (pooled hazard ratio 0.40) and of hepatocellular carcinoma (pooled HR 0.29) compared with cases of no sustained virologic response.
The evidence report findings were limited by several factors, including the relatively small number of randomized trials involving current DAA treatments, limited data on baseline symptoms, limited data on adolescents, and limited evidence on potential long-term harms of DAA therapy, noted Richard Chou, MD, of Oregon Health & Science University, Portland, and colleagues. However, new pooled evidence “indicates that SVR rates with currently recommended all-oral DAA regimens are substantially higher (more than 95%) than with interferon-based therapies evaluated in the prior review (68%-78%),” they said.
Several editorials were published concurrently with the recommendation.
In an editorial published in JAMA, Camilla S. Graham, MD, of Harvard Medical School, Boston, and Stacey Trooskin, MD, of the University of Pennsylvania, Philadelphia, wrote that the new recommendation reflects changes in hepatitis C virus management.
“With the approvals of sofosbuvir and simeprevir in 2013, patients with hepatitis C, a chronic viral illness associated with the deaths of more U.S. patients than the next 60 reportable infectious diseases combined, including HIV and tuberculosis, could expect a greater than 90% rate of achieving sustained virologic response (SVR, defined as undetectable HCV levels 12 weeks or longer after treatment completion, which is consistent with virologic cure of HCV infection) following 12 weeks of treatment,” they said.
These medications are effective but expensive; however, the combination of the availability of generic medications and the ongoing opioid epidemic in the United States are important contributors to the expanded recommendations, which “are welcome,” and may help meeting WHO 2030 targets for reducing new HCV infections, they said.
Dr. Graham disclosed personal fees from UpToDate. Dr. Trooskin disclosed grants from Gilead Sciences and personal fees from Merck, AbbVie, and Gilead Sciences.
In an editorial published in JAMA Internal Medicine, Jennifer C. Price, MD, and Danielle Brandman, MD, both of the University of California, San Francisco, wrote that “the advancements in HCV diagnosis and treatment have been extraordinary,” but that the new recommendation does not go far enough. “Implementation of HCV screening and linkage to treatment requires large-scale coordinated efforts, innovation, and resources. For example, point-of-care HCV RNA testing would enable scale-up of HCV screening and confirmatory testing among individuals at greatest risk of HCV infection,” they said. “Additionally, barriers remain between diagnosis and treatment, such as access to a health care provider who can treat HCV and authorization to receive affordable DAAs,” they noted. “Although the USPSTF HCV screening recommendation is a step forward for controlling HCV infection in the U.S., it will take a coordinated and funded effort to ensure that the anticipated benefits are realized,” they concluded.
Dr. Price disclosed research funding from Gilead Sciences and Merck. Dr. Brandman disclosed research funding from Gilead Sciences, Pfizer, Conatus, Allergan, and Grifols, as well as personal fees from Alnylam.
In an editorial published in JAMA Network Open, Eli S. Rosenberg, PhD, of the University at Albany (N.Y.) School of Public Health, and Joshua A. Barocas, MD, of Boston University, emphasized the need to change the stigma surrounding HCV infection in the United States.
“Given the changing epidemiology of HCV infection, new public health priorities, advancements in treatment, and unmet diagnostic needs, it is wise to periodically reevaluate screening recommendations to ensure that they are maximally addressing these areas and patients’ individual needs,” they said. “The Affordable Care Act requires private insurers and Medicaid to cover preventive services recommended by the USPSTF with a grade of A or B with no cost sharing (i.e., no deductible or copayment),” they noted. Although the new recommendation for one-time screening will likely identify more cases, improve outcomes, and reduce deaths, the editorialists cautioned that “one-time screening should not be interpreted like catch-up vaccinations, whereby we immunize someone at any age for hepatitis B virus, for example, and they are then immunized for the remainder of their life,” and that reassessments are needed, especially for younger adults.
In addition, they emphasized the need to reduce the stigma surrounding HCV and allow for recommendations based on risk, rather than age. “We have forced the USPSTF to adopt age-based screening recommendations because we, as a society, have created a culture in which we have stigmatized these behaviors and we, as practitioners, have proven to be inadequate at eliciting HCV risk behaviors,” they said. “Our responsibility as a society and practice community is to address structural and individual factors that limit our ability to most precisely address the needs of our patients and truly move toward HCV elimination,” they concluded.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The task force researchers had no financial conflicts to disclose.
SOURCES: Owens DK et al. JAMA. 2020 Mar 2. doi: 10.1001/jama.2020.1123; Chou R et al. JAMA. 2020 Mar 2. doi: 10.1001/jama.2019.20788; Graham CS, Trooskin S. JAMA. 2020 Mar 2. doi: 10.1001/jama.2019.22313; Price JC and Brandman D. JAMA Intern Med. 2020 Mar 2. doi: 10.1001/jamainternmed.2019.7334; Rosenberg ES, Barocas JA. JAMA Network Open. 2020 Mar 2. doi: 10.1001/jamanetworkopen.2020.0538.
Adults aged 18-79 years should be screened for hepatitis C virus infection, according to an updated grade B recommendation from the U.S. Preventive Services Task Force.
Cases of acute hepatitis C virus (HCV) infection have spiked in the last decade, in part because of increased use of injection drugs and in part because of better surveillance, Douglas K. Owens, MD, of Stanford (Calif.) University, and colleagues wrote in the recommendation statement published in JAMA.
The recommendation applies to all asymptomatic adults aged 18-79 years without known liver disease, and expands on the 2013 recommendation to screen adults born between 1945 and 1965. The grade B designation means that the task force concluded with moderate certainty that HCV screening for adults aged 18-79 years had “substantial net benefit.”
The recommendations are based on an evidence report including 8 randomized, controlled trials, 48 other treatment studies, and 33 cohort studies published through February 2019 for a total of 179,230 individuals.
The screening is a one-time procedure for most adults, according to the task force, but clinicians should periodically screen individuals at increased risk, such as those with a past or current history of injection drug use. In addition, clinicians should consider screening individuals at increased risk who are above or below the recommended age range.
Although the task force identified no direct evidence on the benefit of screening for HCV infection in asymptomatic adults, a notable finding was that the newer direct-acting antiviral (DAA) regimens are sufficiently effective to support the expanded screening recommendation, they said. However, clinicians should inform patients that screening is voluntary and conducted only with the patient’s knowledge. Clinicians should educate patients about hepatitis C and give them an opportunity to ask questions and to make a decision about screening, according to the task force.
In the evidence report, a total of 49 studies including 10,181 individuals showed DAA treatment associated with pooled sustained virologic response rates greater than 95% across all virus genotypes, and a short-term serious adverse event rate of 1.9%. In addition, sustained virologic response following an antiviral therapy was associated with a reduction in risk of all-cause mortality (pooled hazard ratio 0.40) and of hepatocellular carcinoma (pooled HR 0.29) compared with cases of no sustained virologic response.
The evidence report findings were limited by several factors, including the relatively small number of randomized trials involving current DAA treatments, limited data on baseline symptoms, limited data on adolescents, and limited evidence on potential long-term harms of DAA therapy, noted Richard Chou, MD, of Oregon Health & Science University, Portland, and colleagues. However, new pooled evidence “indicates that SVR rates with currently recommended all-oral DAA regimens are substantially higher (more than 95%) than with interferon-based therapies evaluated in the prior review (68%-78%),” they said.
Several editorials were published concurrently with the recommendation.
In an editorial published in JAMA, Camilla S. Graham, MD, of Harvard Medical School, Boston, and Stacey Trooskin, MD, of the University of Pennsylvania, Philadelphia, wrote that the new recommendation reflects changes in hepatitis C virus management.
“With the approvals of sofosbuvir and simeprevir in 2013, patients with hepatitis C, a chronic viral illness associated with the deaths of more U.S. patients than the next 60 reportable infectious diseases combined, including HIV and tuberculosis, could expect a greater than 90% rate of achieving sustained virologic response (SVR, defined as undetectable HCV levels 12 weeks or longer after treatment completion, which is consistent with virologic cure of HCV infection) following 12 weeks of treatment,” they said.
These medications are effective but expensive; however, the combination of the availability of generic medications and the ongoing opioid epidemic in the United States are important contributors to the expanded recommendations, which “are welcome,” and may help meeting WHO 2030 targets for reducing new HCV infections, they said.
Dr. Graham disclosed personal fees from UpToDate. Dr. Trooskin disclosed grants from Gilead Sciences and personal fees from Merck, AbbVie, and Gilead Sciences.
In an editorial published in JAMA Internal Medicine, Jennifer C. Price, MD, and Danielle Brandman, MD, both of the University of California, San Francisco, wrote that “the advancements in HCV diagnosis and treatment have been extraordinary,” but that the new recommendation does not go far enough. “Implementation of HCV screening and linkage to treatment requires large-scale coordinated efforts, innovation, and resources. For example, point-of-care HCV RNA testing would enable scale-up of HCV screening and confirmatory testing among individuals at greatest risk of HCV infection,” they said. “Additionally, barriers remain between diagnosis and treatment, such as access to a health care provider who can treat HCV and authorization to receive affordable DAAs,” they noted. “Although the USPSTF HCV screening recommendation is a step forward for controlling HCV infection in the U.S., it will take a coordinated and funded effort to ensure that the anticipated benefits are realized,” they concluded.
Dr. Price disclosed research funding from Gilead Sciences and Merck. Dr. Brandman disclosed research funding from Gilead Sciences, Pfizer, Conatus, Allergan, and Grifols, as well as personal fees from Alnylam.
In an editorial published in JAMA Network Open, Eli S. Rosenberg, PhD, of the University at Albany (N.Y.) School of Public Health, and Joshua A. Barocas, MD, of Boston University, emphasized the need to change the stigma surrounding HCV infection in the United States.
“Given the changing epidemiology of HCV infection, new public health priorities, advancements in treatment, and unmet diagnostic needs, it is wise to periodically reevaluate screening recommendations to ensure that they are maximally addressing these areas and patients’ individual needs,” they said. “The Affordable Care Act requires private insurers and Medicaid to cover preventive services recommended by the USPSTF with a grade of A or B with no cost sharing (i.e., no deductible or copayment),” they noted. Although the new recommendation for one-time screening will likely identify more cases, improve outcomes, and reduce deaths, the editorialists cautioned that “one-time screening should not be interpreted like catch-up vaccinations, whereby we immunize someone at any age for hepatitis B virus, for example, and they are then immunized for the remainder of their life,” and that reassessments are needed, especially for younger adults.
In addition, they emphasized the need to reduce the stigma surrounding HCV and allow for recommendations based on risk, rather than age. “We have forced the USPSTF to adopt age-based screening recommendations because we, as a society, have created a culture in which we have stigmatized these behaviors and we, as practitioners, have proven to be inadequate at eliciting HCV risk behaviors,” they said. “Our responsibility as a society and practice community is to address structural and individual factors that limit our ability to most precisely address the needs of our patients and truly move toward HCV elimination,” they concluded.
The USPSTF is supported by the Agency for Healthcare Research and Quality. The task force researchers had no financial conflicts to disclose.
SOURCES: Owens DK et al. JAMA. 2020 Mar 2. doi: 10.1001/jama.2020.1123; Chou R et al. JAMA. 2020 Mar 2. doi: 10.1001/jama.2019.20788; Graham CS, Trooskin S. JAMA. 2020 Mar 2. doi: 10.1001/jama.2019.22313; Price JC and Brandman D. JAMA Intern Med. 2020 Mar 2. doi: 10.1001/jamainternmed.2019.7334; Rosenberg ES, Barocas JA. JAMA Network Open. 2020 Mar 2. doi: 10.1001/jamanetworkopen.2020.0538.
FROM JAMA