User login
The Journal of Clinical Outcomes Management® is an independent, peer-reviewed journal offering evidence-based, practical information for improving the quality, safety, and value of health care.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
First report of MM patient successfully treated for COVID-19 with tocilizumab
Recent research has shown that severe cases of COVID-19 show an excessive immune response and a strong cytokine storm, which may include high levels of granulocyte-macrophage colony-stimulating factor (GSF) and interleukin-6 (IL-6). Following up on that research, investigators from China reported the first case of COVID-19 in a patient with multiple myeloma (MM) who was successfully treated with the humanized anti–IL-6 receptor antibody tocilizumab (an off-label use in the United States). The exceptional case report was published online in Blood Advances, an American Society of Hematology journal.
A 60-year-old man working in Wuhan, China, developed chest tightness without fever and cough on Feb. 1, 2020, and was admitted immediately after computed tomography (CT) imaging of his chest showed multiple ground-glass opacities and pneumatocele located in both subpleural spaces. He received 400 mg of moxifloxacin IV daily for 3 days while swab specimens were collected and tested by real-time reverse transcriptase–polymerase chain reaction. A positive result for SARS-CoV-2 infection was received 3 days later. The patient was subsequently given 200-mg umifenovir (Arbidol) tablets orally, three times daily, for antiviral treatment.
The patient had a history of symptomatic MM, which was diagnosed in 2015. The patient received two cycles of induction chemotherapy consisting of bortezomib, thalidomide, and dexamethasone, and his symptoms completely disappeared. After that, he received thalidomide for maintenance.
Chest CT imaging on hospital day 8 showed that the bilateral, multiple ground-glass opacities from the first scan remained, and laboratory investigations revealed a high level of serum IL-6. On hospital day 9, the patient was given a single, one-time dose of 8 mg/kg tocilizumab, administered by IV. On hospital day 12, his chest tightness disappeared. “After tocilizumab administration, the IL-6 level decreased gradually over the following 10 days (from 121.59 to 20.81 pg/mL), then increased rapidly to the peak (317.38 pg/mL), and then decreased to a low level (117.10 pg/mL). The transient rebounding of the IL-6 level to the peak does not mean COVID-19 relapse: Instead, this might be attributed to the recovery of the normal T cells,” the authors wrote.
On hospital day 19, the patient’s chest CT scan showed that the range of ground-glass opacities had obviously decreased, and he was declared cured and discharged from the hospital. The patient had no symptoms of MM, and related laboratory findings were all in normal ranges, according to the researchers.
“This case is the first to prove that tocilizumab is effective in the treatment of COVID-19 in MM with obvious clinical recovery; however, randomized controlled trials are needed to determine the safety and efficacy of tocilizumab,” the researchers concluded.
The authors declared that they had no conflicts of interest.
SOURCE: Zhang X et al. Blood Adv. 2020;4(7):1307-10.
Recent research has shown that severe cases of COVID-19 show an excessive immune response and a strong cytokine storm, which may include high levels of granulocyte-macrophage colony-stimulating factor (GSF) and interleukin-6 (IL-6). Following up on that research, investigators from China reported the first case of COVID-19 in a patient with multiple myeloma (MM) who was successfully treated with the humanized anti–IL-6 receptor antibody tocilizumab (an off-label use in the United States). The exceptional case report was published online in Blood Advances, an American Society of Hematology journal.
A 60-year-old man working in Wuhan, China, developed chest tightness without fever and cough on Feb. 1, 2020, and was admitted immediately after computed tomography (CT) imaging of his chest showed multiple ground-glass opacities and pneumatocele located in both subpleural spaces. He received 400 mg of moxifloxacin IV daily for 3 days while swab specimens were collected and tested by real-time reverse transcriptase–polymerase chain reaction. A positive result for SARS-CoV-2 infection was received 3 days later. The patient was subsequently given 200-mg umifenovir (Arbidol) tablets orally, three times daily, for antiviral treatment.
The patient had a history of symptomatic MM, which was diagnosed in 2015. The patient received two cycles of induction chemotherapy consisting of bortezomib, thalidomide, and dexamethasone, and his symptoms completely disappeared. After that, he received thalidomide for maintenance.
Chest CT imaging on hospital day 8 showed that the bilateral, multiple ground-glass opacities from the first scan remained, and laboratory investigations revealed a high level of serum IL-6. On hospital day 9, the patient was given a single, one-time dose of 8 mg/kg tocilizumab, administered by IV. On hospital day 12, his chest tightness disappeared. “After tocilizumab administration, the IL-6 level decreased gradually over the following 10 days (from 121.59 to 20.81 pg/mL), then increased rapidly to the peak (317.38 pg/mL), and then decreased to a low level (117.10 pg/mL). The transient rebounding of the IL-6 level to the peak does not mean COVID-19 relapse: Instead, this might be attributed to the recovery of the normal T cells,” the authors wrote.
On hospital day 19, the patient’s chest CT scan showed that the range of ground-glass opacities had obviously decreased, and he was declared cured and discharged from the hospital. The patient had no symptoms of MM, and related laboratory findings were all in normal ranges, according to the researchers.
“This case is the first to prove that tocilizumab is effective in the treatment of COVID-19 in MM with obvious clinical recovery; however, randomized controlled trials are needed to determine the safety and efficacy of tocilizumab,” the researchers concluded.
The authors declared that they had no conflicts of interest.
SOURCE: Zhang X et al. Blood Adv. 2020;4(7):1307-10.
Recent research has shown that severe cases of COVID-19 show an excessive immune response and a strong cytokine storm, which may include high levels of granulocyte-macrophage colony-stimulating factor (GSF) and interleukin-6 (IL-6). Following up on that research, investigators from China reported the first case of COVID-19 in a patient with multiple myeloma (MM) who was successfully treated with the humanized anti–IL-6 receptor antibody tocilizumab (an off-label use in the United States). The exceptional case report was published online in Blood Advances, an American Society of Hematology journal.
A 60-year-old man working in Wuhan, China, developed chest tightness without fever and cough on Feb. 1, 2020, and was admitted immediately after computed tomography (CT) imaging of his chest showed multiple ground-glass opacities and pneumatocele located in both subpleural spaces. He received 400 mg of moxifloxacin IV daily for 3 days while swab specimens were collected and tested by real-time reverse transcriptase–polymerase chain reaction. A positive result for SARS-CoV-2 infection was received 3 days later. The patient was subsequently given 200-mg umifenovir (Arbidol) tablets orally, three times daily, for antiviral treatment.
The patient had a history of symptomatic MM, which was diagnosed in 2015. The patient received two cycles of induction chemotherapy consisting of bortezomib, thalidomide, and dexamethasone, and his symptoms completely disappeared. After that, he received thalidomide for maintenance.
Chest CT imaging on hospital day 8 showed that the bilateral, multiple ground-glass opacities from the first scan remained, and laboratory investigations revealed a high level of serum IL-6. On hospital day 9, the patient was given a single, one-time dose of 8 mg/kg tocilizumab, administered by IV. On hospital day 12, his chest tightness disappeared. “After tocilizumab administration, the IL-6 level decreased gradually over the following 10 days (from 121.59 to 20.81 pg/mL), then increased rapidly to the peak (317.38 pg/mL), and then decreased to a low level (117.10 pg/mL). The transient rebounding of the IL-6 level to the peak does not mean COVID-19 relapse: Instead, this might be attributed to the recovery of the normal T cells,” the authors wrote.
On hospital day 19, the patient’s chest CT scan showed that the range of ground-glass opacities had obviously decreased, and he was declared cured and discharged from the hospital. The patient had no symptoms of MM, and related laboratory findings were all in normal ranges, according to the researchers.
“This case is the first to prove that tocilizumab is effective in the treatment of COVID-19 in MM with obvious clinical recovery; however, randomized controlled trials are needed to determine the safety and efficacy of tocilizumab,” the researchers concluded.
The authors declared that they had no conflicts of interest.
SOURCE: Zhang X et al. Blood Adv. 2020;4(7):1307-10.
FROM BLOOD ADVANCES
Rethink urologic cancer treatment in the era of COVID-19
according to an editorial set to be published in European Urology.
“Regimens with a clear survival advantage should be prioritized, with curative treatments remaining mandatory,” wrote Silke Gillessen Sommer, MD, of Istituto Oncologico della Svizzera Italiana in Bellizona, Switzerland, and Thomas Powles, MD, of Barts Cancer Institute in London.
However, it may be appropriate to stop or delay therapies with modest or unproven survival benefits. “Delaying the start of therapy ... is an appropriate measure for many of the therapies in urology cancer,” they wrote.
Timely recommendations for oncologists
The COVID-19 pandemic is limiting resources for cancer, noted Zachery Reichert, MD, PhD, a urological oncologist and assistant professor at the University of Michigan, Ann Arbor, who was asked for his thoughts about the editorial.
Oncologists and oncology nurses are being shifted to care for COVID-19 patients, space once devoted to cancer care is being repurposed for the pandemic, and personal protective equipment needed to prepare chemotherapies is in short supply.
Meanwhile, cancer patients are at increased risk of dying from the virus (Lancet Oncol. 2020;21:335-7), so there’s a need to minimize their contact with the health care system to protect them from nosocomial infection, and a need to keep their immune system as strong as possible to fight it off.
To help cancer patients fight off infection and keep them out of the hospital, the editorialists recommended growth factors and prophylactic antibiotics after chemotherapy, palliative therapies at doses that avoid febrile neutropenia, discontinuing steroids or at least reducing their doses, and avoiding bisphosphonates if they involve potential COVID-19 exposure in medical facilities.
The advice in the editorial mirrors many of the discussions going on right now at the University of Michigan, Dr. Reichert said, and perhaps other oncology services across the United States.
It will come down to how severe the pandemic becomes locally, but he said it seems likely “a lot of us are going to be wearing a different hat for a while.”
Patients who have symptoms from a growing tumor will likely take precedence at the university, but treatment might be postponed until after COVID-19 peaks if tumors don’t affect quality of life. Also, bladder cancer surgery will probably remain urgent “because the longer you wait, the worse the outcomes,” but perhaps not prostate and kidney cancer surgery, where delay is safer, Dr. Reichert said.
Prostate/renal cancers and germ cell tumors
The editorialists noted that oral androgen receptor therapy should be preferred over chemotherapy for prostate cancer. Dr. Reichert explained that’s because androgen blockade is effective, requires less contact with health care providers, and doesn’t suppress the immune system or tie up hospital resources as much as chemotherapy. “In the world we are in right now, oral pills are a better choice,” he said.
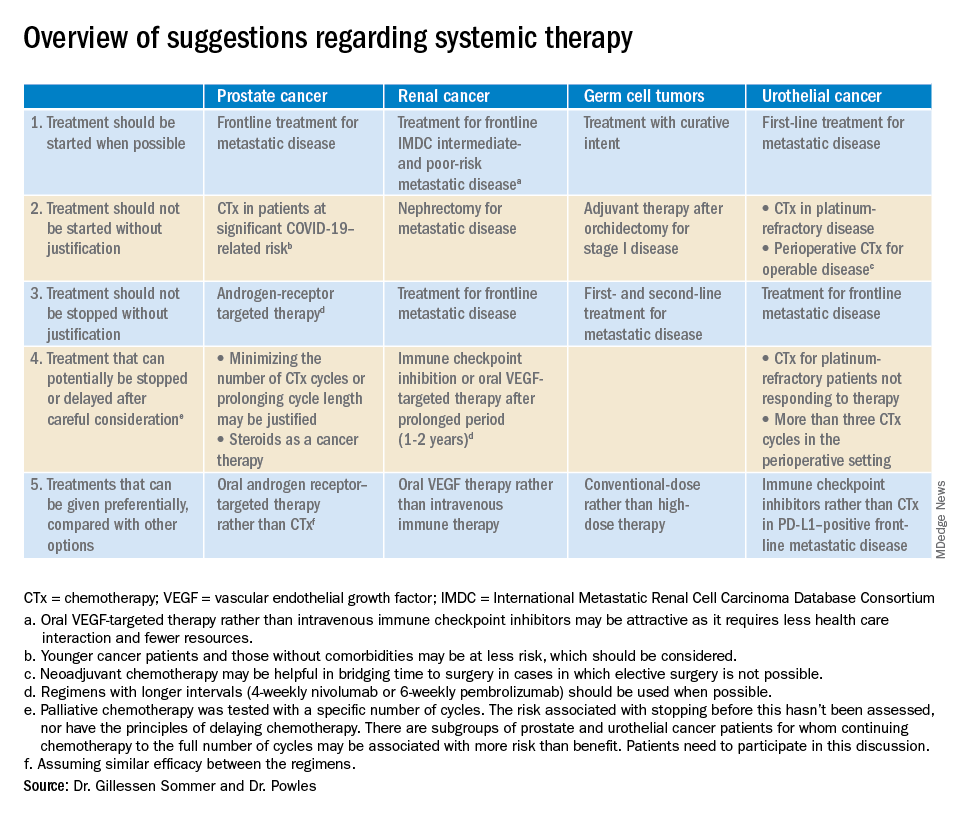
The editorialists recommended against both nephrectomy for metastatic renal cancer and adjuvant therapy after orchidectomy for stage 1 germ cell tumors for similar reasons, and also because there’s minimal evidence of benefit.
Dr. Powles and Dr. Gillessen Sommer suggested considering a break from immune checkpoint inhibitors (ICIs) and oral vascular endothelial growth factors (VEGFs) for renal cancer patients who have been on them a year or two. It’s something that would be considered even under normal circumstances, Dr. Reichert explained, but it’s more urgent now to keep people out of the hospital. VEGFs should also be prioritized over ICIs; they have similar efficacy in renal cancer, but VEGFs are a pill.
They also called for oncologists to favor conventional-dose treatments for germ cell tumors over high-dose treatments, meaning bone marrow transplants or high-intensity chemotherapy. Amid a pandemic, the preference is for options “that don’t require a hospital bed,” Dr. Reichert said.
Urothelial cancer
Dr. Powles and Dr. Gillessen Sommer suggested not starting or continuing second-line chemotherapies in urothelial cancer patients refractory to first-line platinum-based therapies. The chance they will respond to second-line options is low, perhaps around 10%. That might have been enough before the pandemic, but it’s less justified amid resource shortages and the risk of COVID-19 in the infusion suite, Dr. Reichert explained.
Along the same lines, they also suggested reconsidering perioperative chemotherapy for urothelial cancer, and, if it’s still a go, recommended against going past three cycles, as the benefits in both scenarios are likely marginal. However, if COVID-19 cancels surgeries, neoadjuvant therapy might be the right – and only – call, according to the editorialists.
They recommended prioritizing ICIs over chemotherapy in patients with metastatic urothelial cancer who are positive for programmed death-ligand 1 (PD-L1). PD-L1–positive patients have a good chance of responding, and ICIs don’t suppress the immune system.
“Chemotherapy still has a slightly higher percent response, but right now, this is a better choice for” PD-L1-positive patients, Dr. Reichert said.
Dr. Gillessen Sommer and Dr. Powles disclosed ties to Bristol-Myers Squibb, Roche, and numerous other companies. Dr. Reichert has no relevant disclosures.
SOURCE: Gillessen Sommer S, Powles T. “Advice regarding systemic therapy in patients with urological cancers during the COVID-19 pandemic.” Eur Urol. https://els-jbs-prod-cdn.jbs.elsevierhealth.com/pb/assets/raw/Health%20Advance/journals/eururo/EURUROL-D-20-00382-1585928967060.pdf.
according to an editorial set to be published in European Urology.
“Regimens with a clear survival advantage should be prioritized, with curative treatments remaining mandatory,” wrote Silke Gillessen Sommer, MD, of Istituto Oncologico della Svizzera Italiana in Bellizona, Switzerland, and Thomas Powles, MD, of Barts Cancer Institute in London.
However, it may be appropriate to stop or delay therapies with modest or unproven survival benefits. “Delaying the start of therapy ... is an appropriate measure for many of the therapies in urology cancer,” they wrote.
Timely recommendations for oncologists
The COVID-19 pandemic is limiting resources for cancer, noted Zachery Reichert, MD, PhD, a urological oncologist and assistant professor at the University of Michigan, Ann Arbor, who was asked for his thoughts about the editorial.
Oncologists and oncology nurses are being shifted to care for COVID-19 patients, space once devoted to cancer care is being repurposed for the pandemic, and personal protective equipment needed to prepare chemotherapies is in short supply.
Meanwhile, cancer patients are at increased risk of dying from the virus (Lancet Oncol. 2020;21:335-7), so there’s a need to minimize their contact with the health care system to protect them from nosocomial infection, and a need to keep their immune system as strong as possible to fight it off.
To help cancer patients fight off infection and keep them out of the hospital, the editorialists recommended growth factors and prophylactic antibiotics after chemotherapy, palliative therapies at doses that avoid febrile neutropenia, discontinuing steroids or at least reducing their doses, and avoiding bisphosphonates if they involve potential COVID-19 exposure in medical facilities.
The advice in the editorial mirrors many of the discussions going on right now at the University of Michigan, Dr. Reichert said, and perhaps other oncology services across the United States.
It will come down to how severe the pandemic becomes locally, but he said it seems likely “a lot of us are going to be wearing a different hat for a while.”
Patients who have symptoms from a growing tumor will likely take precedence at the university, but treatment might be postponed until after COVID-19 peaks if tumors don’t affect quality of life. Also, bladder cancer surgery will probably remain urgent “because the longer you wait, the worse the outcomes,” but perhaps not prostate and kidney cancer surgery, where delay is safer, Dr. Reichert said.
Prostate/renal cancers and germ cell tumors
The editorialists noted that oral androgen receptor therapy should be preferred over chemotherapy for prostate cancer. Dr. Reichert explained that’s because androgen blockade is effective, requires less contact with health care providers, and doesn’t suppress the immune system or tie up hospital resources as much as chemotherapy. “In the world we are in right now, oral pills are a better choice,” he said.

The editorialists recommended against both nephrectomy for metastatic renal cancer and adjuvant therapy after orchidectomy for stage 1 germ cell tumors for similar reasons, and also because there’s minimal evidence of benefit.
Dr. Powles and Dr. Gillessen Sommer suggested considering a break from immune checkpoint inhibitors (ICIs) and oral vascular endothelial growth factors (VEGFs) for renal cancer patients who have been on them a year or two. It’s something that would be considered even under normal circumstances, Dr. Reichert explained, but it’s more urgent now to keep people out of the hospital. VEGFs should also be prioritized over ICIs; they have similar efficacy in renal cancer, but VEGFs are a pill.
They also called for oncologists to favor conventional-dose treatments for germ cell tumors over high-dose treatments, meaning bone marrow transplants or high-intensity chemotherapy. Amid a pandemic, the preference is for options “that don’t require a hospital bed,” Dr. Reichert said.
Urothelial cancer
Dr. Powles and Dr. Gillessen Sommer suggested not starting or continuing second-line chemotherapies in urothelial cancer patients refractory to first-line platinum-based therapies. The chance they will respond to second-line options is low, perhaps around 10%. That might have been enough before the pandemic, but it’s less justified amid resource shortages and the risk of COVID-19 in the infusion suite, Dr. Reichert explained.
Along the same lines, they also suggested reconsidering perioperative chemotherapy for urothelial cancer, and, if it’s still a go, recommended against going past three cycles, as the benefits in both scenarios are likely marginal. However, if COVID-19 cancels surgeries, neoadjuvant therapy might be the right – and only – call, according to the editorialists.
They recommended prioritizing ICIs over chemotherapy in patients with metastatic urothelial cancer who are positive for programmed death-ligand 1 (PD-L1). PD-L1–positive patients have a good chance of responding, and ICIs don’t suppress the immune system.
“Chemotherapy still has a slightly higher percent response, but right now, this is a better choice for” PD-L1-positive patients, Dr. Reichert said.
Dr. Gillessen Sommer and Dr. Powles disclosed ties to Bristol-Myers Squibb, Roche, and numerous other companies. Dr. Reichert has no relevant disclosures.
SOURCE: Gillessen Sommer S, Powles T. “Advice regarding systemic therapy in patients with urological cancers during the COVID-19 pandemic.” Eur Urol. https://els-jbs-prod-cdn.jbs.elsevierhealth.com/pb/assets/raw/Health%20Advance/journals/eururo/EURUROL-D-20-00382-1585928967060.pdf.
according to an editorial set to be published in European Urology.
“Regimens with a clear survival advantage should be prioritized, with curative treatments remaining mandatory,” wrote Silke Gillessen Sommer, MD, of Istituto Oncologico della Svizzera Italiana in Bellizona, Switzerland, and Thomas Powles, MD, of Barts Cancer Institute in London.
However, it may be appropriate to stop or delay therapies with modest or unproven survival benefits. “Delaying the start of therapy ... is an appropriate measure for many of the therapies in urology cancer,” they wrote.
Timely recommendations for oncologists
The COVID-19 pandemic is limiting resources for cancer, noted Zachery Reichert, MD, PhD, a urological oncologist and assistant professor at the University of Michigan, Ann Arbor, who was asked for his thoughts about the editorial.
Oncologists and oncology nurses are being shifted to care for COVID-19 patients, space once devoted to cancer care is being repurposed for the pandemic, and personal protective equipment needed to prepare chemotherapies is in short supply.
Meanwhile, cancer patients are at increased risk of dying from the virus (Lancet Oncol. 2020;21:335-7), so there’s a need to minimize their contact with the health care system to protect them from nosocomial infection, and a need to keep their immune system as strong as possible to fight it off.
To help cancer patients fight off infection and keep them out of the hospital, the editorialists recommended growth factors and prophylactic antibiotics after chemotherapy, palliative therapies at doses that avoid febrile neutropenia, discontinuing steroids or at least reducing their doses, and avoiding bisphosphonates if they involve potential COVID-19 exposure in medical facilities.
The advice in the editorial mirrors many of the discussions going on right now at the University of Michigan, Dr. Reichert said, and perhaps other oncology services across the United States.
It will come down to how severe the pandemic becomes locally, but he said it seems likely “a lot of us are going to be wearing a different hat for a while.”
Patients who have symptoms from a growing tumor will likely take precedence at the university, but treatment might be postponed until after COVID-19 peaks if tumors don’t affect quality of life. Also, bladder cancer surgery will probably remain urgent “because the longer you wait, the worse the outcomes,” but perhaps not prostate and kidney cancer surgery, where delay is safer, Dr. Reichert said.
Prostate/renal cancers and germ cell tumors
The editorialists noted that oral androgen receptor therapy should be preferred over chemotherapy for prostate cancer. Dr. Reichert explained that’s because androgen blockade is effective, requires less contact with health care providers, and doesn’t suppress the immune system or tie up hospital resources as much as chemotherapy. “In the world we are in right now, oral pills are a better choice,” he said.

The editorialists recommended against both nephrectomy for metastatic renal cancer and adjuvant therapy after orchidectomy for stage 1 germ cell tumors for similar reasons, and also because there’s minimal evidence of benefit.
Dr. Powles and Dr. Gillessen Sommer suggested considering a break from immune checkpoint inhibitors (ICIs) and oral vascular endothelial growth factors (VEGFs) for renal cancer patients who have been on them a year or two. It’s something that would be considered even under normal circumstances, Dr. Reichert explained, but it’s more urgent now to keep people out of the hospital. VEGFs should also be prioritized over ICIs; they have similar efficacy in renal cancer, but VEGFs are a pill.
They also called for oncologists to favor conventional-dose treatments for germ cell tumors over high-dose treatments, meaning bone marrow transplants or high-intensity chemotherapy. Amid a pandemic, the preference is for options “that don’t require a hospital bed,” Dr. Reichert said.
Urothelial cancer
Dr. Powles and Dr. Gillessen Sommer suggested not starting or continuing second-line chemotherapies in urothelial cancer patients refractory to first-line platinum-based therapies. The chance they will respond to second-line options is low, perhaps around 10%. That might have been enough before the pandemic, but it’s less justified amid resource shortages and the risk of COVID-19 in the infusion suite, Dr. Reichert explained.
Along the same lines, they also suggested reconsidering perioperative chemotherapy for urothelial cancer, and, if it’s still a go, recommended against going past three cycles, as the benefits in both scenarios are likely marginal. However, if COVID-19 cancels surgeries, neoadjuvant therapy might be the right – and only – call, according to the editorialists.
They recommended prioritizing ICIs over chemotherapy in patients with metastatic urothelial cancer who are positive for programmed death-ligand 1 (PD-L1). PD-L1–positive patients have a good chance of responding, and ICIs don’t suppress the immune system.
“Chemotherapy still has a slightly higher percent response, but right now, this is a better choice for” PD-L1-positive patients, Dr. Reichert said.
Dr. Gillessen Sommer and Dr. Powles disclosed ties to Bristol-Myers Squibb, Roche, and numerous other companies. Dr. Reichert has no relevant disclosures.
SOURCE: Gillessen Sommer S, Powles T. “Advice regarding systemic therapy in patients with urological cancers during the COVID-19 pandemic.” Eur Urol. https://els-jbs-prod-cdn.jbs.elsevierhealth.com/pb/assets/raw/Health%20Advance/journals/eururo/EURUROL-D-20-00382-1585928967060.pdf.
FROM EUROPEAN UROLOGY
High and low trauma yield similar future osteoporotic fracture risk
Average measures of bone mineral density were similar for individuals with high-trauma and low-trauma fractures, and both were significantly distinct from those with no fracture history, based on data from a cohort study of adults aged 40 years and older.
In the past, low-trauma fractures have typically been associated with osteoporosis, wrote William D. Leslie, MD, of the University of Manitoba, Canada, and his colleagues. However, features distinguishing between low- and high-trauma fractures are often arbitrary and “empirical data have questioned whether distinguishing low-trauma from high-trauma fractures is clinically useful for purposes of risk assessment and treatment,” they wrote.
In a study published in Osteoporosis International, the researchers reviewed data from 64,626 individuals with no prior fracture, 858 with high-trauma fractures, and 14,758 with low-trauma fractures. Overall, the average BMD Z-scores for individuals with no previous fracture were slightly positive, while those with either a high-trauma or low-trauma fracture were negative. The scores for individuals with high-trauma fractures or major osteoporotic fractures were similar to those with low-trauma fractures, and significantly lower (P less than .001) than among individuals with no prior fractures.
The study population included adults aged 40 years and older with baseline DXA scans between Jan. 1, 1996, and Mar. 31, 2016. Those with high-trauma fractures were younger than those with low-trauma fractures (65 years vs. 67 years), and fewer individuals with high-trauma fractures were women (77% vs. 87%).
Both high-trauma and low-trauma fractures were similarly and significantly associated with increased risk for incident major osteoporotic fractures (adjusted hazard ratios 1.31 and 1.55, respectively).
The study findings were limited by several factors including incomplete data on external injury codes, the retrospective study design, and the lack of analysis of the time since prior fractures, the researchers noted. However, the results were strengthened by the large sample size, long-term follow-up, and large numbers of incident fractures, they wrote.
The results support data from previous studies and support “the inclusion of high-trauma clinical fractures in clinical assessment for underlying osteoporosis and in the evaluation for intervention to reduce future fracture risk,” they wrote.
In an accompanying editorial, Steven R. Cummings, MD, of California Pacific Medical Center Research Institute, San Francisco, and Richard Eastell, MD, of the University of Sheffield, England, wrote that the practice of rating fractures according to degree of trauma should be eliminated.
“The study adds evidence to the case that it is time to abandon the mistaken beliefs that fractures rated as high trauma are not associated with decreased BMD, indicate no higher risk of subsequent fracture, or are less likely to be prevented by treatments for osteoporosis,” they wrote.
Describing some fractures as due to trauma reinforces the mistaken belief that the fractures are simply due to the trauma, not decreased bone strength, they noted.
“Indeed, we recommend that people stop attempting to rate or record degree of trauma because such ratings are at best inaccurate and would promote the continued neglect of those patients who are misclassified as having fractures that do not warrant evaluation and treatment,” they concluded.
The study received no outside funding. Dr. Leslie, the study’s first author, reported having no financial conflicts to disclose.
Dr. Cummings disclosed consultancy and grant funding from Amgen and Radius. Dr. Eastell disclosed consultancy funding from IDS, Roche Diagnostics, GSK Nutrition, FNIH, Mereo, Lilly, Sandoz, Nittobo, Abbvie, Samsung, and Haoma Medica and grant funding from Nittobo, IDS, Roche, Amgen, and Alexion.
SOURCE: Leslie WD et al. Osteroporos Int. 2020 Mar 16. doi: 10.1007/s00198-019-05274-2.
Average measures of bone mineral density were similar for individuals with high-trauma and low-trauma fractures, and both were significantly distinct from those with no fracture history, based on data from a cohort study of adults aged 40 years and older.
In the past, low-trauma fractures have typically been associated with osteoporosis, wrote William D. Leslie, MD, of the University of Manitoba, Canada, and his colleagues. However, features distinguishing between low- and high-trauma fractures are often arbitrary and “empirical data have questioned whether distinguishing low-trauma from high-trauma fractures is clinically useful for purposes of risk assessment and treatment,” they wrote.
In a study published in Osteoporosis International, the researchers reviewed data from 64,626 individuals with no prior fracture, 858 with high-trauma fractures, and 14,758 with low-trauma fractures. Overall, the average BMD Z-scores for individuals with no previous fracture were slightly positive, while those with either a high-trauma or low-trauma fracture were negative. The scores for individuals with high-trauma fractures or major osteoporotic fractures were similar to those with low-trauma fractures, and significantly lower (P less than .001) than among individuals with no prior fractures.
The study population included adults aged 40 years and older with baseline DXA scans between Jan. 1, 1996, and Mar. 31, 2016. Those with high-trauma fractures were younger than those with low-trauma fractures (65 years vs. 67 years), and fewer individuals with high-trauma fractures were women (77% vs. 87%).
Both high-trauma and low-trauma fractures were similarly and significantly associated with increased risk for incident major osteoporotic fractures (adjusted hazard ratios 1.31 and 1.55, respectively).
The study findings were limited by several factors including incomplete data on external injury codes, the retrospective study design, and the lack of analysis of the time since prior fractures, the researchers noted. However, the results were strengthened by the large sample size, long-term follow-up, and large numbers of incident fractures, they wrote.
The results support data from previous studies and support “the inclusion of high-trauma clinical fractures in clinical assessment for underlying osteoporosis and in the evaluation for intervention to reduce future fracture risk,” they wrote.
In an accompanying editorial, Steven R. Cummings, MD, of California Pacific Medical Center Research Institute, San Francisco, and Richard Eastell, MD, of the University of Sheffield, England, wrote that the practice of rating fractures according to degree of trauma should be eliminated.
“The study adds evidence to the case that it is time to abandon the mistaken beliefs that fractures rated as high trauma are not associated with decreased BMD, indicate no higher risk of subsequent fracture, or are less likely to be prevented by treatments for osteoporosis,” they wrote.
Describing some fractures as due to trauma reinforces the mistaken belief that the fractures are simply due to the trauma, not decreased bone strength, they noted.
“Indeed, we recommend that people stop attempting to rate or record degree of trauma because such ratings are at best inaccurate and would promote the continued neglect of those patients who are misclassified as having fractures that do not warrant evaluation and treatment,” they concluded.
The study received no outside funding. Dr. Leslie, the study’s first author, reported having no financial conflicts to disclose.
Dr. Cummings disclosed consultancy and grant funding from Amgen and Radius. Dr. Eastell disclosed consultancy funding from IDS, Roche Diagnostics, GSK Nutrition, FNIH, Mereo, Lilly, Sandoz, Nittobo, Abbvie, Samsung, and Haoma Medica and grant funding from Nittobo, IDS, Roche, Amgen, and Alexion.
SOURCE: Leslie WD et al. Osteroporos Int. 2020 Mar 16. doi: 10.1007/s00198-019-05274-2.
Average measures of bone mineral density were similar for individuals with high-trauma and low-trauma fractures, and both were significantly distinct from those with no fracture history, based on data from a cohort study of adults aged 40 years and older.
In the past, low-trauma fractures have typically been associated with osteoporosis, wrote William D. Leslie, MD, of the University of Manitoba, Canada, and his colleagues. However, features distinguishing between low- and high-trauma fractures are often arbitrary and “empirical data have questioned whether distinguishing low-trauma from high-trauma fractures is clinically useful for purposes of risk assessment and treatment,” they wrote.
In a study published in Osteoporosis International, the researchers reviewed data from 64,626 individuals with no prior fracture, 858 with high-trauma fractures, and 14,758 with low-trauma fractures. Overall, the average BMD Z-scores for individuals with no previous fracture were slightly positive, while those with either a high-trauma or low-trauma fracture were negative. The scores for individuals with high-trauma fractures or major osteoporotic fractures were similar to those with low-trauma fractures, and significantly lower (P less than .001) than among individuals with no prior fractures.
The study population included adults aged 40 years and older with baseline DXA scans between Jan. 1, 1996, and Mar. 31, 2016. Those with high-trauma fractures were younger than those with low-trauma fractures (65 years vs. 67 years), and fewer individuals with high-trauma fractures were women (77% vs. 87%).
Both high-trauma and low-trauma fractures were similarly and significantly associated with increased risk for incident major osteoporotic fractures (adjusted hazard ratios 1.31 and 1.55, respectively).
The study findings were limited by several factors including incomplete data on external injury codes, the retrospective study design, and the lack of analysis of the time since prior fractures, the researchers noted. However, the results were strengthened by the large sample size, long-term follow-up, and large numbers of incident fractures, they wrote.
The results support data from previous studies and support “the inclusion of high-trauma clinical fractures in clinical assessment for underlying osteoporosis and in the evaluation for intervention to reduce future fracture risk,” they wrote.
In an accompanying editorial, Steven R. Cummings, MD, of California Pacific Medical Center Research Institute, San Francisco, and Richard Eastell, MD, of the University of Sheffield, England, wrote that the practice of rating fractures according to degree of trauma should be eliminated.
“The study adds evidence to the case that it is time to abandon the mistaken beliefs that fractures rated as high trauma are not associated with decreased BMD, indicate no higher risk of subsequent fracture, or are less likely to be prevented by treatments for osteoporosis,” they wrote.
Describing some fractures as due to trauma reinforces the mistaken belief that the fractures are simply due to the trauma, not decreased bone strength, they noted.
“Indeed, we recommend that people stop attempting to rate or record degree of trauma because such ratings are at best inaccurate and would promote the continued neglect of those patients who are misclassified as having fractures that do not warrant evaluation and treatment,” they concluded.
The study received no outside funding. Dr. Leslie, the study’s first author, reported having no financial conflicts to disclose.
Dr. Cummings disclosed consultancy and grant funding from Amgen and Radius. Dr. Eastell disclosed consultancy funding from IDS, Roche Diagnostics, GSK Nutrition, FNIH, Mereo, Lilly, Sandoz, Nittobo, Abbvie, Samsung, and Haoma Medica and grant funding from Nittobo, IDS, Roche, Amgen, and Alexion.
SOURCE: Leslie WD et al. Osteroporos Int. 2020 Mar 16. doi: 10.1007/s00198-019-05274-2.
FROM OSTEOPOROSIS INTERNATIONAL
FDA grants emergency authorization for first rapid antibody test for COVID-19
The U.S. Food and Drug Administration has granted Cellex an emergency use authorization to market a rapid antibody test for COVID-19, the first antibody test released amidst the pandemic.
“It is reasonable to believe that your product may be effective in diagnosing COVID-19,” and “there is no adequate, approved, and available alternative,” the agency said in a letter to Cellex.
A drop of serum, plasma, or whole blood is placed into a well on a small cartridge, and the results are read 15-20 minutes later; lines indicate the presence of IgM, IgG, or both antibodies against the SARS-CoV-2 virus.
Of 128 samples confirmed positive by reverse transcription polymerase chain reaction in premarket testing, 120 tested positive by IgG, IgM, or both. Of 250 confirmed negative, 239 were negative by the rapid test.
The numbers translated to a positive percent agreement with RT-PCR of 93.8% (95% CI: 88.06-97.26%) and a negative percent agreement of 96.4% (95% CI: 92.26-97.78%), according to labeling.
“Results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection,” the labeling states.
Negative results do not rule out infection; antibodies might not have had enough time to form or the virus could have had a minor amino acid mutation in the epitope recognized by the antibodies screened for in the test. False positives can occur due to cross-reactivity with antibodies from previous infections, such as from other coronaviruses.
Labeling suggests that people who test negative should be checked again in a few days, and positive results should be confirmed by other methods. Also, the intensity of the test lines do not necessarily correlate with SARS-CoV-2 antibody titers.
As part of its authorization, the FDA waived good manufacturing practice requirements, but stipulated that advertising must state that the test has not been formally approved by the agency.
Testing is limited to Clinical Laboratory Improvement Amendments-certified labs. Positive results are required to be reported to public health authorities. The test can be ordered through Cellex distributors or directly from the company.
IgM antibodies are generally detectable several days after the initial infection, while IgG antibodies take longer. It’s not known how long COVID-19 antibodies persist after the infection has cleared, the agency said.
The U.S. Food and Drug Administration has granted Cellex an emergency use authorization to market a rapid antibody test for COVID-19, the first antibody test released amidst the pandemic.
“It is reasonable to believe that your product may be effective in diagnosing COVID-19,” and “there is no adequate, approved, and available alternative,” the agency said in a letter to Cellex.
A drop of serum, plasma, or whole blood is placed into a well on a small cartridge, and the results are read 15-20 minutes later; lines indicate the presence of IgM, IgG, or both antibodies against the SARS-CoV-2 virus.
Of 128 samples confirmed positive by reverse transcription polymerase chain reaction in premarket testing, 120 tested positive by IgG, IgM, or both. Of 250 confirmed negative, 239 were negative by the rapid test.
The numbers translated to a positive percent agreement with RT-PCR of 93.8% (95% CI: 88.06-97.26%) and a negative percent agreement of 96.4% (95% CI: 92.26-97.78%), according to labeling.
“Results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection,” the labeling states.
Negative results do not rule out infection; antibodies might not have had enough time to form or the virus could have had a minor amino acid mutation in the epitope recognized by the antibodies screened for in the test. False positives can occur due to cross-reactivity with antibodies from previous infections, such as from other coronaviruses.
Labeling suggests that people who test negative should be checked again in a few days, and positive results should be confirmed by other methods. Also, the intensity of the test lines do not necessarily correlate with SARS-CoV-2 antibody titers.
As part of its authorization, the FDA waived good manufacturing practice requirements, but stipulated that advertising must state that the test has not been formally approved by the agency.
Testing is limited to Clinical Laboratory Improvement Amendments-certified labs. Positive results are required to be reported to public health authorities. The test can be ordered through Cellex distributors or directly from the company.
IgM antibodies are generally detectable several days after the initial infection, while IgG antibodies take longer. It’s not known how long COVID-19 antibodies persist after the infection has cleared, the agency said.
The U.S. Food and Drug Administration has granted Cellex an emergency use authorization to market a rapid antibody test for COVID-19, the first antibody test released amidst the pandemic.
“It is reasonable to believe that your product may be effective in diagnosing COVID-19,” and “there is no adequate, approved, and available alternative,” the agency said in a letter to Cellex.
A drop of serum, plasma, or whole blood is placed into a well on a small cartridge, and the results are read 15-20 minutes later; lines indicate the presence of IgM, IgG, or both antibodies against the SARS-CoV-2 virus.
Of 128 samples confirmed positive by reverse transcription polymerase chain reaction in premarket testing, 120 tested positive by IgG, IgM, or both. Of 250 confirmed negative, 239 were negative by the rapid test.
The numbers translated to a positive percent agreement with RT-PCR of 93.8% (95% CI: 88.06-97.26%) and a negative percent agreement of 96.4% (95% CI: 92.26-97.78%), according to labeling.
“Results from antibody testing should not be used as the sole basis to diagnose or exclude SARS-CoV-2 infection,” the labeling states.
Negative results do not rule out infection; antibodies might not have had enough time to form or the virus could have had a minor amino acid mutation in the epitope recognized by the antibodies screened for in the test. False positives can occur due to cross-reactivity with antibodies from previous infections, such as from other coronaviruses.
Labeling suggests that people who test negative should be checked again in a few days, and positive results should be confirmed by other methods. Also, the intensity of the test lines do not necessarily correlate with SARS-CoV-2 antibody titers.
As part of its authorization, the FDA waived good manufacturing practice requirements, but stipulated that advertising must state that the test has not been formally approved by the agency.
Testing is limited to Clinical Laboratory Improvement Amendments-certified labs. Positive results are required to be reported to public health authorities. The test can be ordered through Cellex distributors or directly from the company.
IgM antibodies are generally detectable several days after the initial infection, while IgG antibodies take longer. It’s not known how long COVID-19 antibodies persist after the infection has cleared, the agency said.
Survey: COVID-19 is getting in our heads
As the COVID-19 pandemic sweeps across the United States, it is increasingly affecting those who are not infected. Social bonds are being broken, businesses are closing, jobs are being lost, and the stress is mounting.

In a poll conducted March 25-30, 45% of Americans said that stress resulting from the pandemic is having a negative impact on their mental health, compared with 32% expressing that view just 2 weeks earlier, the Kaiser Family Foundation reported April 2.
In the later survey, the effect looked like this: 19% of all respondents said that the pandemic has had a major negative impact and 26% said it has been minor so far. Women were more likely than men (24% vs. 15%) to report a major impact, as were blacks and Hispanic adults (both at 24%) compared with whites (17%), the KFF investigators said.
More Hispanic (44%) and black (42%) respondents also said that they had already lost their job, lost income, or had their hours reduced without pay as a result of the pandemic, compared with whites (36%). Among all respondents, 26% had lost income from a job or business and 28% had lost their job, been laid off, or had their hours reduced without pay, according to KFF.
A majority of respondents (57%) reported “being worried they will put themselves at risk of exposure to coronavirus because they can’t afford to stay home and miss work,” the researchers said. That figure is up from 35% in the earlier survey.
Anxiety about work-related exposure was even higher among hourly workers or those who get paid by the job (61%) and among employed adults who earn less than $40,000 annually (72%), they reported.
Overall, 72% of respondents said that their lives have been disrupted “a lot” or “some” by the coronavirus outbreak, and that is a jump of 32 percentage points over the previous poll, the investigators noted.
The disruption is expected to continue, it seems, as 74% believe that the worst is yet to come “in spite of the health, social and economic upheaval that Americans are already experiencing,” they wrote.
As the COVID-19 pandemic sweeps across the United States, it is increasingly affecting those who are not infected. Social bonds are being broken, businesses are closing, jobs are being lost, and the stress is mounting.

In a poll conducted March 25-30, 45% of Americans said that stress resulting from the pandemic is having a negative impact on their mental health, compared with 32% expressing that view just 2 weeks earlier, the Kaiser Family Foundation reported April 2.
In the later survey, the effect looked like this: 19% of all respondents said that the pandemic has had a major negative impact and 26% said it has been minor so far. Women were more likely than men (24% vs. 15%) to report a major impact, as were blacks and Hispanic adults (both at 24%) compared with whites (17%), the KFF investigators said.
More Hispanic (44%) and black (42%) respondents also said that they had already lost their job, lost income, or had their hours reduced without pay as a result of the pandemic, compared with whites (36%). Among all respondents, 26% had lost income from a job or business and 28% had lost their job, been laid off, or had their hours reduced without pay, according to KFF.
A majority of respondents (57%) reported “being worried they will put themselves at risk of exposure to coronavirus because they can’t afford to stay home and miss work,” the researchers said. That figure is up from 35% in the earlier survey.
Anxiety about work-related exposure was even higher among hourly workers or those who get paid by the job (61%) and among employed adults who earn less than $40,000 annually (72%), they reported.
Overall, 72% of respondents said that their lives have been disrupted “a lot” or “some” by the coronavirus outbreak, and that is a jump of 32 percentage points over the previous poll, the investigators noted.
The disruption is expected to continue, it seems, as 74% believe that the worst is yet to come “in spite of the health, social and economic upheaval that Americans are already experiencing,” they wrote.
As the COVID-19 pandemic sweeps across the United States, it is increasingly affecting those who are not infected. Social bonds are being broken, businesses are closing, jobs are being lost, and the stress is mounting.

In a poll conducted March 25-30, 45% of Americans said that stress resulting from the pandemic is having a negative impact on their mental health, compared with 32% expressing that view just 2 weeks earlier, the Kaiser Family Foundation reported April 2.
In the later survey, the effect looked like this: 19% of all respondents said that the pandemic has had a major negative impact and 26% said it has been minor so far. Women were more likely than men (24% vs. 15%) to report a major impact, as were blacks and Hispanic adults (both at 24%) compared with whites (17%), the KFF investigators said.
More Hispanic (44%) and black (42%) respondents also said that they had already lost their job, lost income, or had their hours reduced without pay as a result of the pandemic, compared with whites (36%). Among all respondents, 26% had lost income from a job or business and 28% had lost their job, been laid off, or had their hours reduced without pay, according to KFF.
A majority of respondents (57%) reported “being worried they will put themselves at risk of exposure to coronavirus because they can’t afford to stay home and miss work,” the researchers said. That figure is up from 35% in the earlier survey.
Anxiety about work-related exposure was even higher among hourly workers or those who get paid by the job (61%) and among employed adults who earn less than $40,000 annually (72%), they reported.
Overall, 72% of respondents said that their lives have been disrupted “a lot” or “some” by the coronavirus outbreak, and that is a jump of 32 percentage points over the previous poll, the investigators noted.
The disruption is expected to continue, it seems, as 74% believe that the worst is yet to come “in spite of the health, social and economic upheaval that Americans are already experiencing,” they wrote.
First presumptive case of encephalitis linked to COVID-19 reported
“As the number of patients with COVID-19 increases worldwide, clinicians and radiologists should be watching for this presentation among patients presenting with COVID-19 and altered mental status,” the clinicians advise in a report published online March 31 in Radiology.
“This is significant for all providers to be aware of and looking out for in [COVID-19] patients who present with an altered level of consciousness. This complication is as devastating as severe lung disease,” Elissa Fory, MD, a neurologist with Henry Ford who was part of the team of medical experts that made the diagnosis, said in a statement.
“We need to be thinking of how we’re going to incorporate patients with severe neurological disease into our treatment paradigm,” Fory added.
Brent Griffith, MD, radiologist with Henry Ford and senior author of the case report, said the case shows “the important role that imaging can play in COVID-19 cases.”
Diagnosed via neuroimaging
The 58-year-old woman presented with a 3-day history of fever, cough, and muscle aches ― symptoms consistent with COVID-19. She was transported by ambulance to the emergency department and showed signs of confusion, lethargy, and disorientation.
The woman tested negative for influenza, but a rapid COVID-19 test confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. She was later diagnosed with acute hemorrhagic necrotizing encephalopathy.
“The team had suspected encephalitis at the outset, but then back-to-back CT and MRI scans made the diagnosis,” Fory said in the statement.
Noncontrast head CT revealed “symmetric hypoattenuation within the bilateral medial thalami with a normal CT angiogram and CT venogram,” the team reports in their article. Brain MRI showed “hemorrhagic rim enhancing lesions within the bilateral thalami, medial temporal lobes, and subinsular regions.”
The patient was started on intravenous immunoglobulin but not high-dose steroids, because of concern for respiratory compromise. As of April 1, the patient was hospitalized in serious condition. Henry Ford Hospital has not provided an update.
Acute necrotizing encephalopathy (ANE) is a rare complication of viral infections, but until now, it has not been known to have occurred as a result of COVID-19 infection. ANE has been associated with intracranial “cytokine storms,” and a recent report in the Lancet suggested that a subgroup of patients with severe COVID-19 might develop a cytokine storm syndrome.
Commenting for Medscape Medical News, Cyrus A. Raji, MD, PhD, assistant professor of radiology and neurology, Washington University in St. Louis, Missouri, said, “Since this is just one report of one patient, the findings are the most preliminary we can conceive, and more research is needed to determine the extent to which COVID-19 may affect the central nervous system.”
Fory, Griffith, and Raji have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
“As the number of patients with COVID-19 increases worldwide, clinicians and radiologists should be watching for this presentation among patients presenting with COVID-19 and altered mental status,” the clinicians advise in a report published online March 31 in Radiology.
“This is significant for all providers to be aware of and looking out for in [COVID-19] patients who present with an altered level of consciousness. This complication is as devastating as severe lung disease,” Elissa Fory, MD, a neurologist with Henry Ford who was part of the team of medical experts that made the diagnosis, said in a statement.
“We need to be thinking of how we’re going to incorporate patients with severe neurological disease into our treatment paradigm,” Fory added.
Brent Griffith, MD, radiologist with Henry Ford and senior author of the case report, said the case shows “the important role that imaging can play in COVID-19 cases.”
Diagnosed via neuroimaging
The 58-year-old woman presented with a 3-day history of fever, cough, and muscle aches ― symptoms consistent with COVID-19. She was transported by ambulance to the emergency department and showed signs of confusion, lethargy, and disorientation.
The woman tested negative for influenza, but a rapid COVID-19 test confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. She was later diagnosed with acute hemorrhagic necrotizing encephalopathy.
“The team had suspected encephalitis at the outset, but then back-to-back CT and MRI scans made the diagnosis,” Fory said in the statement.
Noncontrast head CT revealed “symmetric hypoattenuation within the bilateral medial thalami with a normal CT angiogram and CT venogram,” the team reports in their article. Brain MRI showed “hemorrhagic rim enhancing lesions within the bilateral thalami, medial temporal lobes, and subinsular regions.”
The patient was started on intravenous immunoglobulin but not high-dose steroids, because of concern for respiratory compromise. As of April 1, the patient was hospitalized in serious condition. Henry Ford Hospital has not provided an update.
Acute necrotizing encephalopathy (ANE) is a rare complication of viral infections, but until now, it has not been known to have occurred as a result of COVID-19 infection. ANE has been associated with intracranial “cytokine storms,” and a recent report in the Lancet suggested that a subgroup of patients with severe COVID-19 might develop a cytokine storm syndrome.
Commenting for Medscape Medical News, Cyrus A. Raji, MD, PhD, assistant professor of radiology and neurology, Washington University in St. Louis, Missouri, said, “Since this is just one report of one patient, the findings are the most preliminary we can conceive, and more research is needed to determine the extent to which COVID-19 may affect the central nervous system.”
Fory, Griffith, and Raji have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
“As the number of patients with COVID-19 increases worldwide, clinicians and radiologists should be watching for this presentation among patients presenting with COVID-19 and altered mental status,” the clinicians advise in a report published online March 31 in Radiology.
“This is significant for all providers to be aware of and looking out for in [COVID-19] patients who present with an altered level of consciousness. This complication is as devastating as severe lung disease,” Elissa Fory, MD, a neurologist with Henry Ford who was part of the team of medical experts that made the diagnosis, said in a statement.
“We need to be thinking of how we’re going to incorporate patients with severe neurological disease into our treatment paradigm,” Fory added.
Brent Griffith, MD, radiologist with Henry Ford and senior author of the case report, said the case shows “the important role that imaging can play in COVID-19 cases.”
Diagnosed via neuroimaging
The 58-year-old woman presented with a 3-day history of fever, cough, and muscle aches ― symptoms consistent with COVID-19. She was transported by ambulance to the emergency department and showed signs of confusion, lethargy, and disorientation.
The woman tested negative for influenza, but a rapid COVID-19 test confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection. She was later diagnosed with acute hemorrhagic necrotizing encephalopathy.
“The team had suspected encephalitis at the outset, but then back-to-back CT and MRI scans made the diagnosis,” Fory said in the statement.
Noncontrast head CT revealed “symmetric hypoattenuation within the bilateral medial thalami with a normal CT angiogram and CT venogram,” the team reports in their article. Brain MRI showed “hemorrhagic rim enhancing lesions within the bilateral thalami, medial temporal lobes, and subinsular regions.”
The patient was started on intravenous immunoglobulin but not high-dose steroids, because of concern for respiratory compromise. As of April 1, the patient was hospitalized in serious condition. Henry Ford Hospital has not provided an update.
Acute necrotizing encephalopathy (ANE) is a rare complication of viral infections, but until now, it has not been known to have occurred as a result of COVID-19 infection. ANE has been associated with intracranial “cytokine storms,” and a recent report in the Lancet suggested that a subgroup of patients with severe COVID-19 might develop a cytokine storm syndrome.
Commenting for Medscape Medical News, Cyrus A. Raji, MD, PhD, assistant professor of radiology and neurology, Washington University in St. Louis, Missouri, said, “Since this is just one report of one patient, the findings are the most preliminary we can conceive, and more research is needed to determine the extent to which COVID-19 may affect the central nervous system.”
Fory, Griffith, and Raji have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Survey shows just how dire PPE shortages are at many hospitals
As the COVID-19 pandemic spreads over the country, nearly half (48%) of US healthcare facilities — of various types and sizes — are already or almost out of respirators for treating patients, according to the results of a national online survey of infection prevention professionals.
Conducted during March 23-25 by the Association for Professionals in Infection Control and Epidemiology (APIC), the survey asked APIC’s 11,922 US-based infection preventionist members to rank their facilities’ supply of personal protective equipment (PPE) and key items, such as hand sanitizer and cleaning products, on a 5-point scale from having “plenty” to “none.”
Overall, 1,140 (9.6%) infection preventionists responded. Almost 70% of respondents represented a healthcare system rather than a single facility, and facilities ranged from hospitals (42.7%) to ambulatory care (17.4%) and dialysis (2.7%). The centers, from all 50 states and Washington, D.C., ranged in size from those with 1 to 50 beds to those with more than 300 beds.
and 317 (27.8%) said they were almost out of the devices, which are needed to protect healthcare workers managing patients with COVID-19 and different infectious diseases.
The survey was posted Friday on the APIC website.
Other findings from the survey include:
- Nearly half of respondents (49.2%) said their centers lack sufficient enough face shields, with 36.5% reporting being almost out and 12.6% reporting being completely out.
- Approximately one third (31.7%) of respondents reported being completely or nearly out of face masks.
- Even simple hand sanitizer is in short supply at more than 1 in 4 facilities surveyed; 25.6% of respondents said they are almost out and 2.6% are completely out.
- Nearly 30% of respondents reported accessing supplemental PPE through state or local resources, while 24.6% said they accepted private donations of supplies.
- Fewer than one-third (31.5%) said they had sufficient gowns.
- About 28% said they were almost out of protective respirators, while 20.5% said they have none.
- Only 12.3% said they have received supplies from federal resources, including the Strategic National Stockpile, which is controlled by the Department of Health and Human Services.
- 17.2% of respondents reported resorting to DIY measures such as sewing their own masks.
In terms of staffing resources, 67% of respondents said their center has only one (or fewer) full-time–equivalent infection preventionist on staff to develop protocols for managing COVID-19. That is not surprising given the general underresourcing of infection control programs, the survey compilers said.
“Hospitals and health facilities with fewer than one full-time person on staff to direct infection prevention activities may have been disadvantaged even before the COVID-19 pandemic,” said APIC president Connie Steed, MSN, RN, in a related news release.
On a more positive note, about two thirds of facilities said they have sufficient supplies of gloves (63.4%) and hand washing soap (67.1%).
“I am concerned that many facilities will not be able to protect healthcare workers and patients from not only COVID-19, but also MRSA, C diff., and other antibiotic-resistant infections,” Steed said.
At some centers, however, the situation is not so grim — yet. The large Harris Health System in Houston has enough PPE on hand to support all infection prevention protocols in place, according to Bryan McLeod, director of corporate communications. “The PPE inventory varies from a few weeks to well over a month depending on the specific item,” McLeod told Medscape Medical News. “But everything is dependent on the utilization rate, which can vary with patient volume. Our concern is long-term resupply while demand is peaking around the world, and we continue to pursue all avenues to secure resupply.”
Above all, Steed emphasizes healthcare workers’ need for clarity. “They need to know when exactly they can expect desperately needed supplies to arrive so they don’t have to turn to unproven crisis methods for PPE,” she said. “There have been grim reports from health officials about the supply shortage for weeks and we’re not getting any answers. This is unacceptable.”
APIC is urging the federal government for immediate activation of the Cold War–era Defense Production Act and any other available means to quickly manufacture vital supplies to protect healthcare workers treating the escalating numbers of COVID-19 patients.
In the meantime, frontline healthcare workers are scouring the Internet for suppliers and begging online for donations of masks.
APIC notes that the COVID-19 pandemic is compounded by this year’s particularly severe influenza season, which had already led overcrowded healthcare facilities.
This article first appeared on Medscape.com.
As the COVID-19 pandemic spreads over the country, nearly half (48%) of US healthcare facilities — of various types and sizes — are already or almost out of respirators for treating patients, according to the results of a national online survey of infection prevention professionals.
Conducted during March 23-25 by the Association for Professionals in Infection Control and Epidemiology (APIC), the survey asked APIC’s 11,922 US-based infection preventionist members to rank their facilities’ supply of personal protective equipment (PPE) and key items, such as hand sanitizer and cleaning products, on a 5-point scale from having “plenty” to “none.”
Overall, 1,140 (9.6%) infection preventionists responded. Almost 70% of respondents represented a healthcare system rather than a single facility, and facilities ranged from hospitals (42.7%) to ambulatory care (17.4%) and dialysis (2.7%). The centers, from all 50 states and Washington, D.C., ranged in size from those with 1 to 50 beds to those with more than 300 beds.
and 317 (27.8%) said they were almost out of the devices, which are needed to protect healthcare workers managing patients with COVID-19 and different infectious diseases.
The survey was posted Friday on the APIC website.
Other findings from the survey include:
- Nearly half of respondents (49.2%) said their centers lack sufficient enough face shields, with 36.5% reporting being almost out and 12.6% reporting being completely out.
- Approximately one third (31.7%) of respondents reported being completely or nearly out of face masks.
- Even simple hand sanitizer is in short supply at more than 1 in 4 facilities surveyed; 25.6% of respondents said they are almost out and 2.6% are completely out.
- Nearly 30% of respondents reported accessing supplemental PPE through state or local resources, while 24.6% said they accepted private donations of supplies.
- Fewer than one-third (31.5%) said they had sufficient gowns.
- About 28% said they were almost out of protective respirators, while 20.5% said they have none.
- Only 12.3% said they have received supplies from federal resources, including the Strategic National Stockpile, which is controlled by the Department of Health and Human Services.
- 17.2% of respondents reported resorting to DIY measures such as sewing their own masks.
In terms of staffing resources, 67% of respondents said their center has only one (or fewer) full-time–equivalent infection preventionist on staff to develop protocols for managing COVID-19. That is not surprising given the general underresourcing of infection control programs, the survey compilers said.
“Hospitals and health facilities with fewer than one full-time person on staff to direct infection prevention activities may have been disadvantaged even before the COVID-19 pandemic,” said APIC president Connie Steed, MSN, RN, in a related news release.
On a more positive note, about two thirds of facilities said they have sufficient supplies of gloves (63.4%) and hand washing soap (67.1%).
“I am concerned that many facilities will not be able to protect healthcare workers and patients from not only COVID-19, but also MRSA, C diff., and other antibiotic-resistant infections,” Steed said.
At some centers, however, the situation is not so grim — yet. The large Harris Health System in Houston has enough PPE on hand to support all infection prevention protocols in place, according to Bryan McLeod, director of corporate communications. “The PPE inventory varies from a few weeks to well over a month depending on the specific item,” McLeod told Medscape Medical News. “But everything is dependent on the utilization rate, which can vary with patient volume. Our concern is long-term resupply while demand is peaking around the world, and we continue to pursue all avenues to secure resupply.”
Above all, Steed emphasizes healthcare workers’ need for clarity. “They need to know when exactly they can expect desperately needed supplies to arrive so they don’t have to turn to unproven crisis methods for PPE,” she said. “There have been grim reports from health officials about the supply shortage for weeks and we’re not getting any answers. This is unacceptable.”
APIC is urging the federal government for immediate activation of the Cold War–era Defense Production Act and any other available means to quickly manufacture vital supplies to protect healthcare workers treating the escalating numbers of COVID-19 patients.
In the meantime, frontline healthcare workers are scouring the Internet for suppliers and begging online for donations of masks.
APIC notes that the COVID-19 pandemic is compounded by this year’s particularly severe influenza season, which had already led overcrowded healthcare facilities.
This article first appeared on Medscape.com.
As the COVID-19 pandemic spreads over the country, nearly half (48%) of US healthcare facilities — of various types and sizes — are already or almost out of respirators for treating patients, according to the results of a national online survey of infection prevention professionals.
Conducted during March 23-25 by the Association for Professionals in Infection Control and Epidemiology (APIC), the survey asked APIC’s 11,922 US-based infection preventionist members to rank their facilities’ supply of personal protective equipment (PPE) and key items, such as hand sanitizer and cleaning products, on a 5-point scale from having “plenty” to “none.”
Overall, 1,140 (9.6%) infection preventionists responded. Almost 70% of respondents represented a healthcare system rather than a single facility, and facilities ranged from hospitals (42.7%) to ambulatory care (17.4%) and dialysis (2.7%). The centers, from all 50 states and Washington, D.C., ranged in size from those with 1 to 50 beds to those with more than 300 beds.
and 317 (27.8%) said they were almost out of the devices, which are needed to protect healthcare workers managing patients with COVID-19 and different infectious diseases.
The survey was posted Friday on the APIC website.
Other findings from the survey include:
- Nearly half of respondents (49.2%) said their centers lack sufficient enough face shields, with 36.5% reporting being almost out and 12.6% reporting being completely out.
- Approximately one third (31.7%) of respondents reported being completely or nearly out of face masks.
- Even simple hand sanitizer is in short supply at more than 1 in 4 facilities surveyed; 25.6% of respondents said they are almost out and 2.6% are completely out.
- Nearly 30% of respondents reported accessing supplemental PPE through state or local resources, while 24.6% said they accepted private donations of supplies.
- Fewer than one-third (31.5%) said they had sufficient gowns.
- About 28% said they were almost out of protective respirators, while 20.5% said they have none.
- Only 12.3% said they have received supplies from federal resources, including the Strategic National Stockpile, which is controlled by the Department of Health and Human Services.
- 17.2% of respondents reported resorting to DIY measures such as sewing their own masks.
In terms of staffing resources, 67% of respondents said their center has only one (or fewer) full-time–equivalent infection preventionist on staff to develop protocols for managing COVID-19. That is not surprising given the general underresourcing of infection control programs, the survey compilers said.
“Hospitals and health facilities with fewer than one full-time person on staff to direct infection prevention activities may have been disadvantaged even before the COVID-19 pandemic,” said APIC president Connie Steed, MSN, RN, in a related news release.
On a more positive note, about two thirds of facilities said they have sufficient supplies of gloves (63.4%) and hand washing soap (67.1%).
“I am concerned that many facilities will not be able to protect healthcare workers and patients from not only COVID-19, but also MRSA, C diff., and other antibiotic-resistant infections,” Steed said.
At some centers, however, the situation is not so grim — yet. The large Harris Health System in Houston has enough PPE on hand to support all infection prevention protocols in place, according to Bryan McLeod, director of corporate communications. “The PPE inventory varies from a few weeks to well over a month depending on the specific item,” McLeod told Medscape Medical News. “But everything is dependent on the utilization rate, which can vary with patient volume. Our concern is long-term resupply while demand is peaking around the world, and we continue to pursue all avenues to secure resupply.”
Above all, Steed emphasizes healthcare workers’ need for clarity. “They need to know when exactly they can expect desperately needed supplies to arrive so they don’t have to turn to unproven crisis methods for PPE,” she said. “There have been grim reports from health officials about the supply shortage for weeks and we’re not getting any answers. This is unacceptable.”
APIC is urging the federal government for immediate activation of the Cold War–era Defense Production Act and any other available means to quickly manufacture vital supplies to protect healthcare workers treating the escalating numbers of COVID-19 patients.
In the meantime, frontline healthcare workers are scouring the Internet for suppliers and begging online for donations of masks.
APIC notes that the COVID-19 pandemic is compounded by this year’s particularly severe influenza season, which had already led overcrowded healthcare facilities.
This article first appeared on Medscape.com.
CBT by phone reduces depression in Parkinson’s disease
, according to trial results published in Neurology. The treatment’s effect on depression is “moderated by the reduction of negative thoughts,” the target of the intervention, the researchers said.
Telephone-based CBT may be a convenient option for patients, said lead study author Roseanne D. Dobkin, PhD, of the department of psychiatry at Rutgers Robert Wood Johnson Medical School in Piscataway, N.J., and the VA New Jersey Health Care System in Lyons. “A notable proportion of people with Parkinson’s [disease] do not receive the much needed mental health treatment to facilitate proactive coping with the daily challenges superimposed by their medical condition,” Dr. Dobkin said in a news release. “This study suggests that the effects of the [CBT] last long beyond when the treatment stopped and can be used alongside standard neurological care.”
An undertreated problem
Although depression affects about half of patients with Parkinson’s disease and is associated with physical and cognitive decline, it often goes overlooked and undertreated, the study authors said. Data about the efficacy and tolerability of antidepressants are mixed. CBT holds promise for reducing depression in Parkinson’s disease, prior research suggests, but patients may have limited access to in-person sessions because of physical and geographic barriers.
To assess the efficacy of telephone-based CBT for depression in Parkinson’s disease, compared with community-based treatment as usual, Dr. Dobkin and colleagues conducted a randomized controlled trial. Their study included 72 patients with Parkinson’s disease at an academic medical center. Participants had a depressive disorder, were between aged 35 and 85 years, had stable Parkinson’s disease and mental health treatment for at least 6 weeks, and had a family member or friend willing to participate in the study. The investigators excluded patients with possible dementia or marked cognitive impairment and active suicidal plans or intent.
Participants were randomly assigned to receive usual care plus telephone-based CBT or usual care only. Patients taking antidepressants were evenly divided between the groups.
Telephone-based CBT consisted of weekly 1-hour sessions for 10 weeks. During 6 months of follow-up, patients could receive one session per month if desired. The CBT “targeted negative thoughts (e.g., ‘I have no control’; ‘I am helpless’) and behaviors (e.g., avoidance, excessive worry, lack of exercise),” the investigators said. In addition, therapists trained patients’ care partners by telephone to help patients between sessions. Treatment as usual was defined by patients’ health care teams. For most participants in both groups, treatment as usual included taking antidepressant medication or receiving psychotherapy in the community.
Change in Hamilton Depression Rating Scale (HAM-D) score was the primary outcome. Secondary outcomes included whether patients considered their depression much improved and improvements in depression severity (as measured by the Beck Depression Inventory [BDI]), anxiety (as measured by the Hamilton Anxiety Rating Scale [HAM-A]), and quality of life. The researchers also assessed negative thinking using the Inference Questionnaire. Blinded raters assessed outcomes.
Sustained improvements
Thirty-seven patients were randomized to receive telephone-based CBT, and 35 were randomized to treatment as usual. Overall, 70% were taking antidepressants, and 14% continued receiving psychotherapy from community providers of their choice during the trial. Participants’ average age was 65 years, and 51% were female.
Post treatment, mean improvement in HAM-D score from baseline was 6.53 points in the telephone-based CBT group, compared with −0.27 points in the control group. “Effects at the end of treatment were maintained at 6-month follow-up,” the researchers reported.
About 40% of patients in the CBT group reported that their depression was much improved or very much improved, compared with none of the patients in the control group. Responders had mild to minimal symptomatology on the HAM-D, which indicates that the changes were clinically significant, the authors said.
Secondary outcomes also favored telephone-based CBT. “The intervention was feasible and highly acceptable, yielding an 88% retention rate over the 9-month trial,” Dr. Dobkin and colleagues said.
Compared with other control conditions, treatment-as-usual controls may enhance the effect size of an intervention, the authors noted. In addition, factors such as therapeutic relationship, time, and attention likely contribute to psychotherapy outcomes.
Success may hinge on cognitive ability
“The success of this trial highlights the need for further efficacy studies targeting neuropsychiatric manifestations of [Parkinson’s disease] and adds urgency to the discussion over policies regarding access to tele–mental health, especially for vulnerable populations with limited access to in-person mental health services,” Gregory M. Pontone, MD, and Kelly A. Mills, MD, wrote in an accompanying editorial. Dr. Pontone and Dr. Mills are affiliated with Johns Hopkins University in Baltimore.
“Only rudimentary evidence” exists to guide the treatment of depression in patients with Parkinson’s disease, the editorialists said. “Patient preference and tolerability suggest that nonpharmacologic therapies, such as CBT, are preferred as first-line treatment. Yet access to qualified CBT practitioners, especially those with a clinical knowledge of [Parkinson’s disease], is limited.”
Despite its advantages and the encouraging results, CBT may have important limitations as well, they said. Patients require a certain degree of cognitive ability to benefit from CBT, and the prevalence of dementia among patients with Parkinson’s disease is about 30%.
Nevertheless, the trial provided evidence of target engagement. “Though caveats include the single-blind design and potential confounding by time spent with patient and caregiver, the authors demonstrated that improvement was mediated by the mechanism of CBT – a reduction in negative thinking.”
The trial was funded by the Michael J. Fox Foundation for Parkinson’s Research and the Parkinson’s Alliance (Parkinson’s Unity Walk). Dr. Mills disclosed a patent pending for a system for phase-dependent cortical brain stimulation, National Institutes of Health funding, pending funding from the Michael J. Fox Foundation, and commercial research support from Global Kinetics Corporation. Dr. Pontone is a consultant for Acadia Pharmaceuticals.
SOURCE: Dobkin RD et al. Neurology. 2020 Apr 1. doi: 10.1212/WNL.0000000000009292.
, according to trial results published in Neurology. The treatment’s effect on depression is “moderated by the reduction of negative thoughts,” the target of the intervention, the researchers said.
Telephone-based CBT may be a convenient option for patients, said lead study author Roseanne D. Dobkin, PhD, of the department of psychiatry at Rutgers Robert Wood Johnson Medical School in Piscataway, N.J., and the VA New Jersey Health Care System in Lyons. “A notable proportion of people with Parkinson’s [disease] do not receive the much needed mental health treatment to facilitate proactive coping with the daily challenges superimposed by their medical condition,” Dr. Dobkin said in a news release. “This study suggests that the effects of the [CBT] last long beyond when the treatment stopped and can be used alongside standard neurological care.”
An undertreated problem
Although depression affects about half of patients with Parkinson’s disease and is associated with physical and cognitive decline, it often goes overlooked and undertreated, the study authors said. Data about the efficacy and tolerability of antidepressants are mixed. CBT holds promise for reducing depression in Parkinson’s disease, prior research suggests, but patients may have limited access to in-person sessions because of physical and geographic barriers.
To assess the efficacy of telephone-based CBT for depression in Parkinson’s disease, compared with community-based treatment as usual, Dr. Dobkin and colleagues conducted a randomized controlled trial. Their study included 72 patients with Parkinson’s disease at an academic medical center. Participants had a depressive disorder, were between aged 35 and 85 years, had stable Parkinson’s disease and mental health treatment for at least 6 weeks, and had a family member or friend willing to participate in the study. The investigators excluded patients with possible dementia or marked cognitive impairment and active suicidal plans or intent.
Participants were randomly assigned to receive usual care plus telephone-based CBT or usual care only. Patients taking antidepressants were evenly divided between the groups.
Telephone-based CBT consisted of weekly 1-hour sessions for 10 weeks. During 6 months of follow-up, patients could receive one session per month if desired. The CBT “targeted negative thoughts (e.g., ‘I have no control’; ‘I am helpless’) and behaviors (e.g., avoidance, excessive worry, lack of exercise),” the investigators said. In addition, therapists trained patients’ care partners by telephone to help patients between sessions. Treatment as usual was defined by patients’ health care teams. For most participants in both groups, treatment as usual included taking antidepressant medication or receiving psychotherapy in the community.
Change in Hamilton Depression Rating Scale (HAM-D) score was the primary outcome. Secondary outcomes included whether patients considered their depression much improved and improvements in depression severity (as measured by the Beck Depression Inventory [BDI]), anxiety (as measured by the Hamilton Anxiety Rating Scale [HAM-A]), and quality of life. The researchers also assessed negative thinking using the Inference Questionnaire. Blinded raters assessed outcomes.
Sustained improvements
Thirty-seven patients were randomized to receive telephone-based CBT, and 35 were randomized to treatment as usual. Overall, 70% were taking antidepressants, and 14% continued receiving psychotherapy from community providers of their choice during the trial. Participants’ average age was 65 years, and 51% were female.
Post treatment, mean improvement in HAM-D score from baseline was 6.53 points in the telephone-based CBT group, compared with −0.27 points in the control group. “Effects at the end of treatment were maintained at 6-month follow-up,” the researchers reported.
About 40% of patients in the CBT group reported that their depression was much improved or very much improved, compared with none of the patients in the control group. Responders had mild to minimal symptomatology on the HAM-D, which indicates that the changes were clinically significant, the authors said.
Secondary outcomes also favored telephone-based CBT. “The intervention was feasible and highly acceptable, yielding an 88% retention rate over the 9-month trial,” Dr. Dobkin and colleagues said.
Compared with other control conditions, treatment-as-usual controls may enhance the effect size of an intervention, the authors noted. In addition, factors such as therapeutic relationship, time, and attention likely contribute to psychotherapy outcomes.
Success may hinge on cognitive ability
“The success of this trial highlights the need for further efficacy studies targeting neuropsychiatric manifestations of [Parkinson’s disease] and adds urgency to the discussion over policies regarding access to tele–mental health, especially for vulnerable populations with limited access to in-person mental health services,” Gregory M. Pontone, MD, and Kelly A. Mills, MD, wrote in an accompanying editorial. Dr. Pontone and Dr. Mills are affiliated with Johns Hopkins University in Baltimore.
“Only rudimentary evidence” exists to guide the treatment of depression in patients with Parkinson’s disease, the editorialists said. “Patient preference and tolerability suggest that nonpharmacologic therapies, such as CBT, are preferred as first-line treatment. Yet access to qualified CBT practitioners, especially those with a clinical knowledge of [Parkinson’s disease], is limited.”
Despite its advantages and the encouraging results, CBT may have important limitations as well, they said. Patients require a certain degree of cognitive ability to benefit from CBT, and the prevalence of dementia among patients with Parkinson’s disease is about 30%.
Nevertheless, the trial provided evidence of target engagement. “Though caveats include the single-blind design and potential confounding by time spent with patient and caregiver, the authors demonstrated that improvement was mediated by the mechanism of CBT – a reduction in negative thinking.”
The trial was funded by the Michael J. Fox Foundation for Parkinson’s Research and the Parkinson’s Alliance (Parkinson’s Unity Walk). Dr. Mills disclosed a patent pending for a system for phase-dependent cortical brain stimulation, National Institutes of Health funding, pending funding from the Michael J. Fox Foundation, and commercial research support from Global Kinetics Corporation. Dr. Pontone is a consultant for Acadia Pharmaceuticals.
SOURCE: Dobkin RD et al. Neurology. 2020 Apr 1. doi: 10.1212/WNL.0000000000009292.
, according to trial results published in Neurology. The treatment’s effect on depression is “moderated by the reduction of negative thoughts,” the target of the intervention, the researchers said.
Telephone-based CBT may be a convenient option for patients, said lead study author Roseanne D. Dobkin, PhD, of the department of psychiatry at Rutgers Robert Wood Johnson Medical School in Piscataway, N.J., and the VA New Jersey Health Care System in Lyons. “A notable proportion of people with Parkinson’s [disease] do not receive the much needed mental health treatment to facilitate proactive coping with the daily challenges superimposed by their medical condition,” Dr. Dobkin said in a news release. “This study suggests that the effects of the [CBT] last long beyond when the treatment stopped and can be used alongside standard neurological care.”
An undertreated problem
Although depression affects about half of patients with Parkinson’s disease and is associated with physical and cognitive decline, it often goes overlooked and undertreated, the study authors said. Data about the efficacy and tolerability of antidepressants are mixed. CBT holds promise for reducing depression in Parkinson’s disease, prior research suggests, but patients may have limited access to in-person sessions because of physical and geographic barriers.
To assess the efficacy of telephone-based CBT for depression in Parkinson’s disease, compared with community-based treatment as usual, Dr. Dobkin and colleagues conducted a randomized controlled trial. Their study included 72 patients with Parkinson’s disease at an academic medical center. Participants had a depressive disorder, were between aged 35 and 85 years, had stable Parkinson’s disease and mental health treatment for at least 6 weeks, and had a family member or friend willing to participate in the study. The investigators excluded patients with possible dementia or marked cognitive impairment and active suicidal plans or intent.
Participants were randomly assigned to receive usual care plus telephone-based CBT or usual care only. Patients taking antidepressants were evenly divided between the groups.
Telephone-based CBT consisted of weekly 1-hour sessions for 10 weeks. During 6 months of follow-up, patients could receive one session per month if desired. The CBT “targeted negative thoughts (e.g., ‘I have no control’; ‘I am helpless’) and behaviors (e.g., avoidance, excessive worry, lack of exercise),” the investigators said. In addition, therapists trained patients’ care partners by telephone to help patients between sessions. Treatment as usual was defined by patients’ health care teams. For most participants in both groups, treatment as usual included taking antidepressant medication or receiving psychotherapy in the community.
Change in Hamilton Depression Rating Scale (HAM-D) score was the primary outcome. Secondary outcomes included whether patients considered their depression much improved and improvements in depression severity (as measured by the Beck Depression Inventory [BDI]), anxiety (as measured by the Hamilton Anxiety Rating Scale [HAM-A]), and quality of life. The researchers also assessed negative thinking using the Inference Questionnaire. Blinded raters assessed outcomes.
Sustained improvements
Thirty-seven patients were randomized to receive telephone-based CBT, and 35 were randomized to treatment as usual. Overall, 70% were taking antidepressants, and 14% continued receiving psychotherapy from community providers of their choice during the trial. Participants’ average age was 65 years, and 51% were female.
Post treatment, mean improvement in HAM-D score from baseline was 6.53 points in the telephone-based CBT group, compared with −0.27 points in the control group. “Effects at the end of treatment were maintained at 6-month follow-up,” the researchers reported.
About 40% of patients in the CBT group reported that their depression was much improved or very much improved, compared with none of the patients in the control group. Responders had mild to minimal symptomatology on the HAM-D, which indicates that the changes were clinically significant, the authors said.
Secondary outcomes also favored telephone-based CBT. “The intervention was feasible and highly acceptable, yielding an 88% retention rate over the 9-month trial,” Dr. Dobkin and colleagues said.
Compared with other control conditions, treatment-as-usual controls may enhance the effect size of an intervention, the authors noted. In addition, factors such as therapeutic relationship, time, and attention likely contribute to psychotherapy outcomes.
Success may hinge on cognitive ability
“The success of this trial highlights the need for further efficacy studies targeting neuropsychiatric manifestations of [Parkinson’s disease] and adds urgency to the discussion over policies regarding access to tele–mental health, especially for vulnerable populations with limited access to in-person mental health services,” Gregory M. Pontone, MD, and Kelly A. Mills, MD, wrote in an accompanying editorial. Dr. Pontone and Dr. Mills are affiliated with Johns Hopkins University in Baltimore.
“Only rudimentary evidence” exists to guide the treatment of depression in patients with Parkinson’s disease, the editorialists said. “Patient preference and tolerability suggest that nonpharmacologic therapies, such as CBT, are preferred as first-line treatment. Yet access to qualified CBT practitioners, especially those with a clinical knowledge of [Parkinson’s disease], is limited.”
Despite its advantages and the encouraging results, CBT may have important limitations as well, they said. Patients require a certain degree of cognitive ability to benefit from CBT, and the prevalence of dementia among patients with Parkinson’s disease is about 30%.
Nevertheless, the trial provided evidence of target engagement. “Though caveats include the single-blind design and potential confounding by time spent with patient and caregiver, the authors demonstrated that improvement was mediated by the mechanism of CBT – a reduction in negative thinking.”
The trial was funded by the Michael J. Fox Foundation for Parkinson’s Research and the Parkinson’s Alliance (Parkinson’s Unity Walk). Dr. Mills disclosed a patent pending for a system for phase-dependent cortical brain stimulation, National Institutes of Health funding, pending funding from the Michael J. Fox Foundation, and commercial research support from Global Kinetics Corporation. Dr. Pontone is a consultant for Acadia Pharmaceuticals.
SOURCE: Dobkin RD et al. Neurology. 2020 Apr 1. doi: 10.1212/WNL.0000000000009292.
FROM NEUROLOGY
Water-only fasting may reduce chemo modifications, hospital admissions
Patients with gynecologic malignancies who consumed only water for 24 hours before and 24 hours after each chemotherapy cycle had fewer dose delays and reductions compared with patients who didn’t fast, results of a small study showed.
The study included 23 women with ovarian, uterine, or cervical cancer, most of whom received platinum-based chemotherapy and taxanes. Fewer treatment modifications were required among the 11 patients randomized to a 24-hour water-only fast before and after each chemotherapy cycle than among the 12 patients randomized to standard care. Furthermore, there were no hospital admissions in the fasting group and two admissions in the control group, according to study author Courtney J. Riedinger, MD, of the University of Tennessee Medical Center in Knoxville.
She and her colleagues detailed the rationale and results of this study in an abstract that had been slated for presentation at the Society of Gynecologic Oncology’s Annual Meeting on Women’s Cancer. The meeting was canceled because of the COVID-19 pandemic. Data have been updated from the abstract.
Rationale
“There’s a lot of new research and interest about nonpharmacologic interventions and lifestyle modifications to help patients cope with chemotherapy and even help with treatment, potentially,” Dr. Riedinger said in an interview.
“We decided to test water-only fasting because there’s not much data about the cell-fitness effects of fasting” on chemotherapy outcomes, she said.
Pre-chemotherapy fasting is based on the concept of differential stress resistance intended to protect normal cells but not cancer cells from the effects of chemotherapy. Fasting decreases levels of insulin-like growth factor 1, which leads healthy cells to enter a protective state by decreasing cell growth and proliferation. Cancer cells, in contrast, cannot enter the protective state, and are therefore more vulnerable than healthy, quiescent cells when exposed to drugs that target the cell cycle, Dr. Riedinger and colleagues noted.
The team cited two studies suggesting a benefit from fasting prior to chemotherapy. In the first study, mice that underwent 48-60 hours of short-term fasting were significantly less likely to die after exposure to a high dose of etoposide, compared with mice that did not fast before exposure (PNAS; 105[24]: 8215-822).
The second study showed that breast and ovarian cancer patients had improved quality of life scores and decreased fatigue when they fasted for 36 hours before and 24 hours after a chemotherapy cycle (BMC Cancer;18: article 476).
Study details
Dr. Riedinger and colleagues conducted a nonblinded, randomized trial of fasting in women, aged 34-73 years, who had gynecologic malignancies treated with a planned six cycles of chemotherapy. The patients were instructed to maintain a water-only fast for 24 hours before and 24 hours after each cycle. Controls did not fast.
Patient functional status and quality of life were investigated with the National Comprehensive Cancer Network–Functional Assessment of Cancer Therapy Ovarian Symptom Index (NCCN-FACT FOSI-18). Questionnaires were completed at each chemotherapy visit, and the records were reviewed to evaluate compliance, changes in treatment plan, and hospitalizations.
In all, 92% of chemotherapy cycles were completed with fasting as directed.
There were no significant differences in any of the study measures between patients who fasted and those who did not. However, this study was not powered to detect a difference, according to Dr. Riedinger.
Still, there were trends suggesting a benefit to fasting. Fasting patients had a higher mean change in NCCN-FACT FOSI-18 score compared with controls – increases of 5.11 and .22, respectively.
Five patients in the fasting group required changes to their treatment regimen, compared with eight patients in the control group. In addition, there were no hospital admissions in the fasting group and two admissions in the control group.
Patients tolerated the fast well without significant weight loss, and there were no grade 3 or 4 toxicities among patients who fasted.
The investigators are planning a larger study to further evaluate the effect of fasting on quality of life scores and treatment, and to evaluate the effects of fasting on hematologic toxicities. Future studies will focus on the optimal duration of fasting and the use of fasting-mimicking diets to allow for longer fasting periods, Dr. Riedinger said.
The study was internally funded. The authors reported no conflicts of interest.
SOURCE: Riedinger CJ et al. SGO 2020. Abstract 22.
Patients with gynecologic malignancies who consumed only water for 24 hours before and 24 hours after each chemotherapy cycle had fewer dose delays and reductions compared with patients who didn’t fast, results of a small study showed.
The study included 23 women with ovarian, uterine, or cervical cancer, most of whom received platinum-based chemotherapy and taxanes. Fewer treatment modifications were required among the 11 patients randomized to a 24-hour water-only fast before and after each chemotherapy cycle than among the 12 patients randomized to standard care. Furthermore, there were no hospital admissions in the fasting group and two admissions in the control group, according to study author Courtney J. Riedinger, MD, of the University of Tennessee Medical Center in Knoxville.
She and her colleagues detailed the rationale and results of this study in an abstract that had been slated for presentation at the Society of Gynecologic Oncology’s Annual Meeting on Women’s Cancer. The meeting was canceled because of the COVID-19 pandemic. Data have been updated from the abstract.
Rationale
“There’s a lot of new research and interest about nonpharmacologic interventions and lifestyle modifications to help patients cope with chemotherapy and even help with treatment, potentially,” Dr. Riedinger said in an interview.
“We decided to test water-only fasting because there’s not much data about the cell-fitness effects of fasting” on chemotherapy outcomes, she said.
Pre-chemotherapy fasting is based on the concept of differential stress resistance intended to protect normal cells but not cancer cells from the effects of chemotherapy. Fasting decreases levels of insulin-like growth factor 1, which leads healthy cells to enter a protective state by decreasing cell growth and proliferation. Cancer cells, in contrast, cannot enter the protective state, and are therefore more vulnerable than healthy, quiescent cells when exposed to drugs that target the cell cycle, Dr. Riedinger and colleagues noted.
The team cited two studies suggesting a benefit from fasting prior to chemotherapy. In the first study, mice that underwent 48-60 hours of short-term fasting were significantly less likely to die after exposure to a high dose of etoposide, compared with mice that did not fast before exposure (PNAS; 105[24]: 8215-822).
The second study showed that breast and ovarian cancer patients had improved quality of life scores and decreased fatigue when they fasted for 36 hours before and 24 hours after a chemotherapy cycle (BMC Cancer;18: article 476).
Study details
Dr. Riedinger and colleagues conducted a nonblinded, randomized trial of fasting in women, aged 34-73 years, who had gynecologic malignancies treated with a planned six cycles of chemotherapy. The patients were instructed to maintain a water-only fast for 24 hours before and 24 hours after each cycle. Controls did not fast.
Patient functional status and quality of life were investigated with the National Comprehensive Cancer Network–Functional Assessment of Cancer Therapy Ovarian Symptom Index (NCCN-FACT FOSI-18). Questionnaires were completed at each chemotherapy visit, and the records were reviewed to evaluate compliance, changes in treatment plan, and hospitalizations.
In all, 92% of chemotherapy cycles were completed with fasting as directed.
There were no significant differences in any of the study measures between patients who fasted and those who did not. However, this study was not powered to detect a difference, according to Dr. Riedinger.
Still, there were trends suggesting a benefit to fasting. Fasting patients had a higher mean change in NCCN-FACT FOSI-18 score compared with controls – increases of 5.11 and .22, respectively.
Five patients in the fasting group required changes to their treatment regimen, compared with eight patients in the control group. In addition, there were no hospital admissions in the fasting group and two admissions in the control group.
Patients tolerated the fast well without significant weight loss, and there were no grade 3 or 4 toxicities among patients who fasted.
The investigators are planning a larger study to further evaluate the effect of fasting on quality of life scores and treatment, and to evaluate the effects of fasting on hematologic toxicities. Future studies will focus on the optimal duration of fasting and the use of fasting-mimicking diets to allow for longer fasting periods, Dr. Riedinger said.
The study was internally funded. The authors reported no conflicts of interest.
SOURCE: Riedinger CJ et al. SGO 2020. Abstract 22.
Patients with gynecologic malignancies who consumed only water for 24 hours before and 24 hours after each chemotherapy cycle had fewer dose delays and reductions compared with patients who didn’t fast, results of a small study showed.
The study included 23 women with ovarian, uterine, or cervical cancer, most of whom received platinum-based chemotherapy and taxanes. Fewer treatment modifications were required among the 11 patients randomized to a 24-hour water-only fast before and after each chemotherapy cycle than among the 12 patients randomized to standard care. Furthermore, there were no hospital admissions in the fasting group and two admissions in the control group, according to study author Courtney J. Riedinger, MD, of the University of Tennessee Medical Center in Knoxville.
She and her colleagues detailed the rationale and results of this study in an abstract that had been slated for presentation at the Society of Gynecologic Oncology’s Annual Meeting on Women’s Cancer. The meeting was canceled because of the COVID-19 pandemic. Data have been updated from the abstract.
Rationale
“There’s a lot of new research and interest about nonpharmacologic interventions and lifestyle modifications to help patients cope with chemotherapy and even help with treatment, potentially,” Dr. Riedinger said in an interview.
“We decided to test water-only fasting because there’s not much data about the cell-fitness effects of fasting” on chemotherapy outcomes, she said.
Pre-chemotherapy fasting is based on the concept of differential stress resistance intended to protect normal cells but not cancer cells from the effects of chemotherapy. Fasting decreases levels of insulin-like growth factor 1, which leads healthy cells to enter a protective state by decreasing cell growth and proliferation. Cancer cells, in contrast, cannot enter the protective state, and are therefore more vulnerable than healthy, quiescent cells when exposed to drugs that target the cell cycle, Dr. Riedinger and colleagues noted.
The team cited two studies suggesting a benefit from fasting prior to chemotherapy. In the first study, mice that underwent 48-60 hours of short-term fasting were significantly less likely to die after exposure to a high dose of etoposide, compared with mice that did not fast before exposure (PNAS; 105[24]: 8215-822).
The second study showed that breast and ovarian cancer patients had improved quality of life scores and decreased fatigue when they fasted for 36 hours before and 24 hours after a chemotherapy cycle (BMC Cancer;18: article 476).
Study details
Dr. Riedinger and colleagues conducted a nonblinded, randomized trial of fasting in women, aged 34-73 years, who had gynecologic malignancies treated with a planned six cycles of chemotherapy. The patients were instructed to maintain a water-only fast for 24 hours before and 24 hours after each cycle. Controls did not fast.
Patient functional status and quality of life were investigated with the National Comprehensive Cancer Network–Functional Assessment of Cancer Therapy Ovarian Symptom Index (NCCN-FACT FOSI-18). Questionnaires were completed at each chemotherapy visit, and the records were reviewed to evaluate compliance, changes in treatment plan, and hospitalizations.
In all, 92% of chemotherapy cycles were completed with fasting as directed.
There were no significant differences in any of the study measures between patients who fasted and those who did not. However, this study was not powered to detect a difference, according to Dr. Riedinger.
Still, there were trends suggesting a benefit to fasting. Fasting patients had a higher mean change in NCCN-FACT FOSI-18 score compared with controls – increases of 5.11 and .22, respectively.
Five patients in the fasting group required changes to their treatment regimen, compared with eight patients in the control group. In addition, there were no hospital admissions in the fasting group and two admissions in the control group.
Patients tolerated the fast well without significant weight loss, and there were no grade 3 or 4 toxicities among patients who fasted.
The investigators are planning a larger study to further evaluate the effect of fasting on quality of life scores and treatment, and to evaluate the effects of fasting on hematologic toxicities. Future studies will focus on the optimal duration of fasting and the use of fasting-mimicking diets to allow for longer fasting periods, Dr. Riedinger said.
The study was internally funded. The authors reported no conflicts of interest.
SOURCE: Riedinger CJ et al. SGO 2020. Abstract 22.
FROM SGO 2020
iPLEDGE allows at-home pregnancy tests during pandemic
tests to comply with the requirements of the iPLEDGE program during the COVID-19 pandemic, according to an update program posted on the iPLEDGE website.
The program’s other requirements – the prescription window and two forms of birth control – remain unchanged.
The change follows recent guidance from the Department of Health & Human Services and the Food and Drug Administration regarding accommodations for medical care and drugs subject to Risk Evaluation and Mitigation Strategies (REMS) in the midst of a public health emergency that requires most people to remain in their homes except for essential services.
Allowing females to take at-home pregnancy tests and communicate the results to physician according to their preference is “a game changer for the middle of a pandemic, obviously,” Neil Goldberg, MD, a dermatologist in Westchester County, New York, said in an interview. “These are patients who don’t need to spend time outside just to get pregnancy tests done. It makes it a lot easier.”
Dr. Goldberg is frustrated, however, that the accommodations have not been more widely publicized; he discovered the change incidentally when speaking to an iPLEDGE program representative to request a waiver for a patient who had taken her pregnancy test too early. The program had denied a similar request for a 15-year-old patient of his the previous week, despite the patient being abstinent and having been in shelter-in-place for several weeks.
“The size of your notice [on the website] should be proportionate to how important it is,” Dr. Goldberg said, and the small red box on the site is easy to miss. By contrast, asking anyone to leave their homes to go to a lab for a pregnancy test in the midst of a global pandemic so they can continue their medication would be putting patients at risk, he added.
The iPLEDGE program is designed in part to ensure unplanned pregnancies do not occur in females while taking the teratogenic acne drug. But the rules are onerous and difficult even during normal times, pointed out Hilary Baldwin, MD, medical director of the Acne Treatment and Research Center in New York City and past president of the American Acne and Rosacea Society.
Male patients taking isotretinoin must visit their physician every month to get a new no-refills prescription, but females must get a pregnancy test at a Clinical Laboratory Improvement Amendments–certified lab, which must then provide physical results to the prescribing physician. The doctor enters the negative pregnancy test and the two forms of birth control the patient is taking in the iPLEDGE program site.
Then the patient must take an online test at home to acknowledge they understand what it means to not get pregnant and enter the two forms of birth control they are using – which must match what the doctor enters – before the pharmacy can dispense the drug. The entire process must occur within 7 days or else the patient has to wait 19 days before starting the process over.
“We run a very tight schedule for girls. And every month, we would worry that something would interfere, a snow storm or something else, and that they wouldn’t be able to complete their objectives within the 7-day period,” Dr Baldwin said in an interview. “It was always difficult, and now with us not being able to see the patient and the patient not wanting to go to the lab, this became completely impossible.”
Until this change, some patients may not have been able to get their prescription for severe nodulocystic acne, which can cause physical and psychological scarring, and “postponing treatment increases the likelihood of scarring,” Dr. Baldwin pointed out.

Dr. Goldberg’s patients now take a pregnancy test at home and send him a photo of the negative test that he then inserts into their EMR.
According to a March 17 statement from HHS, potential penalties for HIPAA violations are waived for good-faith use of “everyday communication technologies,” such as Skype or FaceTime, for telehealth treatment or diagnostics. The change was intended to allow telehealth services to continue healthcare for practices that had not previously had secure telehealth technology established.
Despite the changes for at-home pregnancy tests for females and in-person visits for all patients, the program has not altered the 7-day prescription window or the requirement to have two forms of birth control.
With reports of a global condom shortage, Dr Baldwin said she has more concerns about her adult patients being able to find a required barrier method of birth control than about her adolescent patients.
“This is a unique opportunity for us to trust our teenage patients because they can’t leave the house,” Dr. Baldwin said. “I’m actually more worried about my adult women on the drug who are bored and cooped up in a house with their significant other.”
Dr. Baldwin and Dr. Goldberg had no relevant disclosures. Dr. Goldberg is a Dermatology News board member.
tests to comply with the requirements of the iPLEDGE program during the COVID-19 pandemic, according to an update program posted on the iPLEDGE website.
The program’s other requirements – the prescription window and two forms of birth control – remain unchanged.
The change follows recent guidance from the Department of Health & Human Services and the Food and Drug Administration regarding accommodations for medical care and drugs subject to Risk Evaluation and Mitigation Strategies (REMS) in the midst of a public health emergency that requires most people to remain in their homes except for essential services.
Allowing females to take at-home pregnancy tests and communicate the results to physician according to their preference is “a game changer for the middle of a pandemic, obviously,” Neil Goldberg, MD, a dermatologist in Westchester County, New York, said in an interview. “These are patients who don’t need to spend time outside just to get pregnancy tests done. It makes it a lot easier.”
Dr. Goldberg is frustrated, however, that the accommodations have not been more widely publicized; he discovered the change incidentally when speaking to an iPLEDGE program representative to request a waiver for a patient who had taken her pregnancy test too early. The program had denied a similar request for a 15-year-old patient of his the previous week, despite the patient being abstinent and having been in shelter-in-place for several weeks.
“The size of your notice [on the website] should be proportionate to how important it is,” Dr. Goldberg said, and the small red box on the site is easy to miss. By contrast, asking anyone to leave their homes to go to a lab for a pregnancy test in the midst of a global pandemic so they can continue their medication would be putting patients at risk, he added.
The iPLEDGE program is designed in part to ensure unplanned pregnancies do not occur in females while taking the teratogenic acne drug. But the rules are onerous and difficult even during normal times, pointed out Hilary Baldwin, MD, medical director of the Acne Treatment and Research Center in New York City and past president of the American Acne and Rosacea Society.
Male patients taking isotretinoin must visit their physician every month to get a new no-refills prescription, but females must get a pregnancy test at a Clinical Laboratory Improvement Amendments–certified lab, which must then provide physical results to the prescribing physician. The doctor enters the negative pregnancy test and the two forms of birth control the patient is taking in the iPLEDGE program site.
Then the patient must take an online test at home to acknowledge they understand what it means to not get pregnant and enter the two forms of birth control they are using – which must match what the doctor enters – before the pharmacy can dispense the drug. The entire process must occur within 7 days or else the patient has to wait 19 days before starting the process over.
“We run a very tight schedule for girls. And every month, we would worry that something would interfere, a snow storm or something else, and that they wouldn’t be able to complete their objectives within the 7-day period,” Dr Baldwin said in an interview. “It was always difficult, and now with us not being able to see the patient and the patient not wanting to go to the lab, this became completely impossible.”
Until this change, some patients may not have been able to get their prescription for severe nodulocystic acne, which can cause physical and psychological scarring, and “postponing treatment increases the likelihood of scarring,” Dr. Baldwin pointed out.

Dr. Goldberg’s patients now take a pregnancy test at home and send him a photo of the negative test that he then inserts into their EMR.
According to a March 17 statement from HHS, potential penalties for HIPAA violations are waived for good-faith use of “everyday communication technologies,” such as Skype or FaceTime, for telehealth treatment or diagnostics. The change was intended to allow telehealth services to continue healthcare for practices that had not previously had secure telehealth technology established.
Despite the changes for at-home pregnancy tests for females and in-person visits for all patients, the program has not altered the 7-day prescription window or the requirement to have two forms of birth control.
With reports of a global condom shortage, Dr Baldwin said she has more concerns about her adult patients being able to find a required barrier method of birth control than about her adolescent patients.
“This is a unique opportunity for us to trust our teenage patients because they can’t leave the house,” Dr. Baldwin said. “I’m actually more worried about my adult women on the drug who are bored and cooped up in a house with their significant other.”
Dr. Baldwin and Dr. Goldberg had no relevant disclosures. Dr. Goldberg is a Dermatology News board member.
tests to comply with the requirements of the iPLEDGE program during the COVID-19 pandemic, according to an update program posted on the iPLEDGE website.
The program’s other requirements – the prescription window and two forms of birth control – remain unchanged.
The change follows recent guidance from the Department of Health & Human Services and the Food and Drug Administration regarding accommodations for medical care and drugs subject to Risk Evaluation and Mitigation Strategies (REMS) in the midst of a public health emergency that requires most people to remain in their homes except for essential services.
Allowing females to take at-home pregnancy tests and communicate the results to physician according to their preference is “a game changer for the middle of a pandemic, obviously,” Neil Goldberg, MD, a dermatologist in Westchester County, New York, said in an interview. “These are patients who don’t need to spend time outside just to get pregnancy tests done. It makes it a lot easier.”
Dr. Goldberg is frustrated, however, that the accommodations have not been more widely publicized; he discovered the change incidentally when speaking to an iPLEDGE program representative to request a waiver for a patient who had taken her pregnancy test too early. The program had denied a similar request for a 15-year-old patient of his the previous week, despite the patient being abstinent and having been in shelter-in-place for several weeks.
“The size of your notice [on the website] should be proportionate to how important it is,” Dr. Goldberg said, and the small red box on the site is easy to miss. By contrast, asking anyone to leave their homes to go to a lab for a pregnancy test in the midst of a global pandemic so they can continue their medication would be putting patients at risk, he added.
The iPLEDGE program is designed in part to ensure unplanned pregnancies do not occur in females while taking the teratogenic acne drug. But the rules are onerous and difficult even during normal times, pointed out Hilary Baldwin, MD, medical director of the Acne Treatment and Research Center in New York City and past president of the American Acne and Rosacea Society.
Male patients taking isotretinoin must visit their physician every month to get a new no-refills prescription, but females must get a pregnancy test at a Clinical Laboratory Improvement Amendments–certified lab, which must then provide physical results to the prescribing physician. The doctor enters the negative pregnancy test and the two forms of birth control the patient is taking in the iPLEDGE program site.
Then the patient must take an online test at home to acknowledge they understand what it means to not get pregnant and enter the two forms of birth control they are using – which must match what the doctor enters – before the pharmacy can dispense the drug. The entire process must occur within 7 days or else the patient has to wait 19 days before starting the process over.
“We run a very tight schedule for girls. And every month, we would worry that something would interfere, a snow storm or something else, and that they wouldn’t be able to complete their objectives within the 7-day period,” Dr Baldwin said in an interview. “It was always difficult, and now with us not being able to see the patient and the patient not wanting to go to the lab, this became completely impossible.”
Until this change, some patients may not have been able to get their prescription for severe nodulocystic acne, which can cause physical and psychological scarring, and “postponing treatment increases the likelihood of scarring,” Dr. Baldwin pointed out.

Dr. Goldberg’s patients now take a pregnancy test at home and send him a photo of the negative test that he then inserts into their EMR.
According to a March 17 statement from HHS, potential penalties for HIPAA violations are waived for good-faith use of “everyday communication technologies,” such as Skype or FaceTime, for telehealth treatment or diagnostics. The change was intended to allow telehealth services to continue healthcare for practices that had not previously had secure telehealth technology established.
Despite the changes for at-home pregnancy tests for females and in-person visits for all patients, the program has not altered the 7-day prescription window or the requirement to have two forms of birth control.
With reports of a global condom shortage, Dr Baldwin said she has more concerns about her adult patients being able to find a required barrier method of birth control than about her adolescent patients.
“This is a unique opportunity for us to trust our teenage patients because they can’t leave the house,” Dr. Baldwin said. “I’m actually more worried about my adult women on the drug who are bored and cooped up in a house with their significant other.”
Dr. Baldwin and Dr. Goldberg had no relevant disclosures. Dr. Goldberg is a Dermatology News board member.