User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
Fungi that cause lung infections now found in most states: Study
Soil-dwelling fungi that can cause lung infections are more widespread than most doctors thought, sometimes leading to missed diagnoses, according to a new study.
Researchers studying fungi-linked lung infections realized that many infections were occurring in places the fungi weren’t thought to exist. They found that maps doctors use to know if the fungi are a threat in their area hadn’t been updated in half a century.
University of California, Davis infectious disease professor George Thompson, MD, said in a commentary published along with the study.
Published in the journal Clinical Infectious Diseases, the study sought to identify illnesses linked to three types of soil fungi in the United States that are known to cause lung infections. They are called histoplasma, blastomyces, and coccidioides, the latter of which causes an illness known as Valley fever, which has been on the rise in California.
Researchers used data for more than 45 million people who use Medicare and found that at least 1 of these 3 fungi are present in 48 of 50 U.S. states and Washington, D.C.
Symptoms after breathing in the fungi spores include fever and cough and can be similar to symptoms of other illnesses, according to the Centers for Disease Control.
The researchers said health care providers need to increase their suspicion for these fungi, which “would likely result in fewer missed diagnoses, fewer diagnostic delays, and improved patient outcomes.”
A version of this article first appeared on WebMD.com.
Soil-dwelling fungi that can cause lung infections are more widespread than most doctors thought, sometimes leading to missed diagnoses, according to a new study.
Researchers studying fungi-linked lung infections realized that many infections were occurring in places the fungi weren’t thought to exist. They found that maps doctors use to know if the fungi are a threat in their area hadn’t been updated in half a century.
University of California, Davis infectious disease professor George Thompson, MD, said in a commentary published along with the study.
Published in the journal Clinical Infectious Diseases, the study sought to identify illnesses linked to three types of soil fungi in the United States that are known to cause lung infections. They are called histoplasma, blastomyces, and coccidioides, the latter of which causes an illness known as Valley fever, which has been on the rise in California.
Researchers used data for more than 45 million people who use Medicare and found that at least 1 of these 3 fungi are present in 48 of 50 U.S. states and Washington, D.C.
Symptoms after breathing in the fungi spores include fever and cough and can be similar to symptoms of other illnesses, according to the Centers for Disease Control.
The researchers said health care providers need to increase their suspicion for these fungi, which “would likely result in fewer missed diagnoses, fewer diagnostic delays, and improved patient outcomes.”
A version of this article first appeared on WebMD.com.
Soil-dwelling fungi that can cause lung infections are more widespread than most doctors thought, sometimes leading to missed diagnoses, according to a new study.
Researchers studying fungi-linked lung infections realized that many infections were occurring in places the fungi weren’t thought to exist. They found that maps doctors use to know if the fungi are a threat in their area hadn’t been updated in half a century.
University of California, Davis infectious disease professor George Thompson, MD, said in a commentary published along with the study.
Published in the journal Clinical Infectious Diseases, the study sought to identify illnesses linked to three types of soil fungi in the United States that are known to cause lung infections. They are called histoplasma, blastomyces, and coccidioides, the latter of which causes an illness known as Valley fever, which has been on the rise in California.
Researchers used data for more than 45 million people who use Medicare and found that at least 1 of these 3 fungi are present in 48 of 50 U.S. states and Washington, D.C.
Symptoms after breathing in the fungi spores include fever and cough and can be similar to symptoms of other illnesses, according to the Centers for Disease Control.
The researchers said health care providers need to increase their suspicion for these fungi, which “would likely result in fewer missed diagnoses, fewer diagnostic delays, and improved patient outcomes.”
A version of this article first appeared on WebMD.com.
FROM CLINICAL INFECTIOUS DISEASE
Ohio measles outbreak sickens nearly 60 children
None of the children had been fully vaccinated against measles, and 23 of them have been hospitalized, local officials report.
“Measles can be very serious, especially for children under age 5,” Columbus Public Health spokesperson Kelli Newman told CNN.
Nearly all of the infected children are under age 5, with 12 of them being under 1 year old.
“Many children are hospitalized for dehydration,” Ms. Newman told CNN in an email. “Other serious complications also can include pneumonia and neurological conditions such as encephalitis. There’s no way of knowing which children will become so sick they have to be hospitalized. The safest way to protect children from measles is to make sure they are vaccinated with MMR.”
Of the 59 infected children, 56 were unvaccinated and three had been partially vaccinated. The MMR (measles, mumps, and rubella) vaccine is recommended for children beginning at 12 months old, according to the Centers for Disease Control and American Academy of Pediatrics. Two doses are needed to be considered fully vaccinated, and the second dose is usually given between 4 and 6 years old.
Measles “is one of the most infectious agents known to man,” the academy says.
It is so contagious that if one person has it, up to 9 out of 10 people around that person will also become infected if they are not protected, the CDC explains. Measles infection causes a rash and a fever that can spike beyond 104° F. Sometimes, the illness can lead to brain swelling, brain damage, or death.
Last month, the World Health Organization and CDC warned that 40 million children worldwide missed their measles vaccinations in 2021, partly due to pandemic disruptions. The American Academy of Pediatrics also notes that many parents choose not to vaccinate their children due to misinformation.
Infants are at heightened risk because they are too young to be vaccinated.
The academy offered several tips for protecting unvaccinated infants during a measles outbreak:
- Limit your baby’s exposure to crowds, other children, and people with cold symptoms.
- Disinfect objects and surfaces at home regularly, because the measles virus can live on surfaces or suspended in the air for 2 hours.
- If possible, feed your baby breast milk, because it has antibodies to prevent and fight infections.
A version of this article first appeared on WebMD.com.
None of the children had been fully vaccinated against measles, and 23 of them have been hospitalized, local officials report.
“Measles can be very serious, especially for children under age 5,” Columbus Public Health spokesperson Kelli Newman told CNN.
Nearly all of the infected children are under age 5, with 12 of them being under 1 year old.
“Many children are hospitalized for dehydration,” Ms. Newman told CNN in an email. “Other serious complications also can include pneumonia and neurological conditions such as encephalitis. There’s no way of knowing which children will become so sick they have to be hospitalized. The safest way to protect children from measles is to make sure they are vaccinated with MMR.”
Of the 59 infected children, 56 were unvaccinated and three had been partially vaccinated. The MMR (measles, mumps, and rubella) vaccine is recommended for children beginning at 12 months old, according to the Centers for Disease Control and American Academy of Pediatrics. Two doses are needed to be considered fully vaccinated, and the second dose is usually given between 4 and 6 years old.
Measles “is one of the most infectious agents known to man,” the academy says.
It is so contagious that if one person has it, up to 9 out of 10 people around that person will also become infected if they are not protected, the CDC explains. Measles infection causes a rash and a fever that can spike beyond 104° F. Sometimes, the illness can lead to brain swelling, brain damage, or death.
Last month, the World Health Organization and CDC warned that 40 million children worldwide missed their measles vaccinations in 2021, partly due to pandemic disruptions. The American Academy of Pediatrics also notes that many parents choose not to vaccinate their children due to misinformation.
Infants are at heightened risk because they are too young to be vaccinated.
The academy offered several tips for protecting unvaccinated infants during a measles outbreak:
- Limit your baby’s exposure to crowds, other children, and people with cold symptoms.
- Disinfect objects and surfaces at home regularly, because the measles virus can live on surfaces or suspended in the air for 2 hours.
- If possible, feed your baby breast milk, because it has antibodies to prevent and fight infections.
A version of this article first appeared on WebMD.com.
None of the children had been fully vaccinated against measles, and 23 of them have been hospitalized, local officials report.
“Measles can be very serious, especially for children under age 5,” Columbus Public Health spokesperson Kelli Newman told CNN.
Nearly all of the infected children are under age 5, with 12 of them being under 1 year old.
“Many children are hospitalized for dehydration,” Ms. Newman told CNN in an email. “Other serious complications also can include pneumonia and neurological conditions such as encephalitis. There’s no way of knowing which children will become so sick they have to be hospitalized. The safest way to protect children from measles is to make sure they are vaccinated with MMR.”
Of the 59 infected children, 56 were unvaccinated and three had been partially vaccinated. The MMR (measles, mumps, and rubella) vaccine is recommended for children beginning at 12 months old, according to the Centers for Disease Control and American Academy of Pediatrics. Two doses are needed to be considered fully vaccinated, and the second dose is usually given between 4 and 6 years old.
Measles “is one of the most infectious agents known to man,” the academy says.
It is so contagious that if one person has it, up to 9 out of 10 people around that person will also become infected if they are not protected, the CDC explains. Measles infection causes a rash and a fever that can spike beyond 104° F. Sometimes, the illness can lead to brain swelling, brain damage, or death.
Last month, the World Health Organization and CDC warned that 40 million children worldwide missed their measles vaccinations in 2021, partly due to pandemic disruptions. The American Academy of Pediatrics also notes that many parents choose not to vaccinate their children due to misinformation.
Infants are at heightened risk because they are too young to be vaccinated.
The academy offered several tips for protecting unvaccinated infants during a measles outbreak:
- Limit your baby’s exposure to crowds, other children, and people with cold symptoms.
- Disinfect objects and surfaces at home regularly, because the measles virus can live on surfaces or suspended in the air for 2 hours.
- If possible, feed your baby breast milk, because it has antibodies to prevent and fight infections.
A version of this article first appeared on WebMD.com.
Vaccination cuts long COVID risk for rheumatic disease patients
Patients with rheumatic disease are at least half as likely to develop long COVID after a SARS-CoV-2 infection if they have been fully vaccinated against COVID-19, according to research published in Annals of the Rheumatic Diseases (2022 Nov 28. doi: 10.1136/ard-2022-223439).
“Moreover, those who were vaccinated prior to getting COVID-19 had less pain and fatigue after their infection,” Zachary S. Wallace, MD, MSc, an assistant professor of medicine at Harvard Medical School, Boston, and a study author, said in an interview. “These findings reinforce the importance of vaccination in this population.”
Messaging around the value of COVID vaccination has been confusing for some with rheumatic disease “because our concern regarding a blunted response to vaccination has led many patients to think that they do not provide much benefit if they are on immunosuppression,” Dr. Wallace said. “In our cohort, which included many patients on immunosuppression of varying degrees, being vaccinated was quite beneficial.”
Leonard H. Calabrese, DO, director of the R.J. Fasenmyer Center for Clinical Immunology and a professor of medicine at the Cleveland Clinic, said in an interview that the study is an “extremely important contribution to our understanding of COVID-19 and its pattern of recovery in patients with immune-mediated inflammatory diseases [IMIDs].” Remaining unanswered questions are “whether patients with IMIDs develop more frequent PASC [post–acute sequelae of COVID-19] from COVID-19 and, if so, is it milder or more severe, and does it differ in its clinical phenotype?”
Long COVID risk assessed at 4 weeks and 3 months after infection
The researchers prospectively tracked 280 adult patients in the Mass General Brigham health care system in the greater Boston area who had systemic autoimmune rheumatic diseases and had an acute COVID-19 infection between March 2020 and July 2022. Patients were an average 53 years old, and most were White (82%) and female (80%). More than half (59%) had inflammatory arthritis, a quarter (24%) had connective tissue disease, and most others had a vasculitis condition or multiple conditions.
A total of 11% of patients were unvaccinated, 28% were partially vaccinated with one mRNA COVID-19 vaccine dose, and 41% were fully vaccinated with two mRNA vaccine doses or one Johnson & Johnson dose. The 116 fully vaccinated patients were considered to have a breakthrough infection while the other 164 were considered to have a nonbreakthrough infection. The breakthrough and nonbreakthrough groups were similar in terms of age, sex, race, ethnicity, smoking status, and type of rheumatic disease. Comorbidities were also similar, except obesity, which was more common in the non–breakthrough infection group (25%) than the breakthrough infection group (10%).
The researchers queried patients on their COVID-19 symptoms, how long symptoms lasted, treatments they received, and hospitalization details. COVID-19 symptoms assessed included fever, sore throat, new cough, nasal congestion/rhinorrhea, dyspnea, chest pain, rash, myalgia, fatigue/malaise, headache, nausea/vomiting, diarrhea, anosmia, dysgeusia, and joint pain.
Patients completed surveys about symptoms at 4 weeks and 3 months after infection. Long COVID, or PASC, was defined as any persistent symptom at the times assessed.
Vaccinated patients fared better across outcomes
At 4 weeks after infection, 41% of fully vaccinated patients had at least one persistent symptom, compared with 54% of unvaccinated or partially vaccinated patients (P = .04). At 3 months after infection, 21% of fully vaccinated patients had at least one persistent symptom, compared with 41% of unvaccinated or partially vaccinated patients (P < .0001).
Vaccinated patients were half as likely to have long COVID at 4 weeks after infection (adjusted odds ratio, 0.49) and 90% less likely to have long COVID 3 months after infection (aOR, 0.1), after adjustment for age, sex, race, comorbidities, and use of any of four immune-suppressing medications (anti-CD20 monoclonal antibodies, methotrexate, mycophenolate, or glucocorticoids).
Fully vaccinated patients with breakthrough infections had an average 21 additional days without symptoms during follow-up, compared with unvaccinated and partially vaccinated patients (P = .04).
Reduced risk of long COVID did not change for vaccinated patients after sensitivity analyses for those who did not receive nirmatrelvir/ritonavir (Paxlovid) or monoclonal antibodies, those who didn’t receive any COVID-19-related treatment, those who completed their questionnaires within 6 months after infection, and those who were not hospitalized.
“One important message is that among those who did get PASC, the severity appears similar among those with and without a breakthrough infection,” Dr. Wallace said. “This highlights the need for ongoing research to improve recognition, diagnosis, and treatment of PASC.”
Many more breakthrough infections (72%) than nonbreakthrough infections (2%) occurred during Omicron. The authors acknowledged that different variants might play a role in different long COVID risks but said such potential confounding is unlikely to fully explain the results.
“Even with data suggesting that the Omicron variants may be intrinsically less severe, vaccination still has an impact on severity of infection, rates of hospitalization, and other outcomes and thus may play a role in the risk of PASC,” lead author Naomi Patel, MD, an instructor at Harvard Medical School and a rheumatologist at Massachusetts General Hospital, said in an interview. “A study evaluating the proportions with PASC by vaccination status during the time in which a single variant is predominant, such as the early Omicron era, could help to better assess the more isolated impact of vaccination on PASC.”
Dr. Calabrese said he is convinced that Omicron infections are less likely to result in more severe forms of acute COVID than pre-Omicron infections, and he suspects Omicron infections are also less likely to result in long COVID, although less evidence currently supports this hypothesis.
Hospitalization was more common in unvaccinated/partly vaccinated patients than in vaccinated patients (27% vs. 5%; P = .001). Although pain and fatigue were lower in those with breakthrough infections, functional scores and health-related quality of life were similar in both groups.
Some symptoms significantly differed between vaccinated and unvaccinated/partly vaccinated groups, possibly caused partly by different variants. Nasal congestion was more common (73%) in those with breakthrough infections than in those with nonbreakthrough infections (46%; P < .0001). Those who were unvaccinated/partly vaccinated were significantly more likely to have loss of smell (46% vs. 22%) or taste (45% vs. 28%) or to have joint pain (11% vs. 4%).
Treatment with nirmatrelvir/ritonavir was also more common in vaccinated patients (12%) than in unvaccinated/partly vaccinated patients (1%; P < .0001), as was treatment with monoclonal antibodies (34% vs. 8%; P < .0001).
The study was limited by its low diversity and being at a single health care system, the authors said. Study coauthor Jeffrey A. Sparks, MD, MMSc, an assistant professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, said in an interview that the group is planning additional studies as their cohort grows, including “investigating the relationships between COVID-19 and specific rheumatic diseases and immunomodulating medications, expansion of autoimmunity and systemic inflammation, and lung damage among specific patient populations.”
Dr. Calabrese said it will be important for follow-up study of the symptomatic patients to “determine how many of these patients will fit the clinical picture of long COVID or long-haul phenotypes over the months and years ahead, including documenting exertional malaise and quality of life.
This study only assessed patients who received zero, one, or two doses of a vaccine, but many patients with rheumatic disease today will likely have received booster doses. However, Dr. Calabrese said it would be difficult to quantify whether a third, fourth, or fifth dose offers additional protection from long-term COVID complications after full vaccination or hybrid vaccination.
The research was funded by the Rheumatology Research Foundation, the National Institutes of Health, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Wallace has received research support from Bristol-Myers Squibb and Principia/Sanofi and consulting fees from Zenas BioPharma, Horizon, Sanofi, Shionogi, Viela Bio, and Medpace. Dr. Sparks has received research support from Bristol-Myers Squibb and consulting fees from AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer. Dr. Patel has received consulting fees from FVC Health. Calabrese has consulted for Genentech, Sanofi-Regeneron, AstraZeneca, and GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
Patients with rheumatic disease are at least half as likely to develop long COVID after a SARS-CoV-2 infection if they have been fully vaccinated against COVID-19, according to research published in Annals of the Rheumatic Diseases (2022 Nov 28. doi: 10.1136/ard-2022-223439).
“Moreover, those who were vaccinated prior to getting COVID-19 had less pain and fatigue after their infection,” Zachary S. Wallace, MD, MSc, an assistant professor of medicine at Harvard Medical School, Boston, and a study author, said in an interview. “These findings reinforce the importance of vaccination in this population.”
Messaging around the value of COVID vaccination has been confusing for some with rheumatic disease “because our concern regarding a blunted response to vaccination has led many patients to think that they do not provide much benefit if they are on immunosuppression,” Dr. Wallace said. “In our cohort, which included many patients on immunosuppression of varying degrees, being vaccinated was quite beneficial.”
Leonard H. Calabrese, DO, director of the R.J. Fasenmyer Center for Clinical Immunology and a professor of medicine at the Cleveland Clinic, said in an interview that the study is an “extremely important contribution to our understanding of COVID-19 and its pattern of recovery in patients with immune-mediated inflammatory diseases [IMIDs].” Remaining unanswered questions are “whether patients with IMIDs develop more frequent PASC [post–acute sequelae of COVID-19] from COVID-19 and, if so, is it milder or more severe, and does it differ in its clinical phenotype?”
Long COVID risk assessed at 4 weeks and 3 months after infection
The researchers prospectively tracked 280 adult patients in the Mass General Brigham health care system in the greater Boston area who had systemic autoimmune rheumatic diseases and had an acute COVID-19 infection between March 2020 and July 2022. Patients were an average 53 years old, and most were White (82%) and female (80%). More than half (59%) had inflammatory arthritis, a quarter (24%) had connective tissue disease, and most others had a vasculitis condition or multiple conditions.
A total of 11% of patients were unvaccinated, 28% were partially vaccinated with one mRNA COVID-19 vaccine dose, and 41% were fully vaccinated with two mRNA vaccine doses or one Johnson & Johnson dose. The 116 fully vaccinated patients were considered to have a breakthrough infection while the other 164 were considered to have a nonbreakthrough infection. The breakthrough and nonbreakthrough groups were similar in terms of age, sex, race, ethnicity, smoking status, and type of rheumatic disease. Comorbidities were also similar, except obesity, which was more common in the non–breakthrough infection group (25%) than the breakthrough infection group (10%).
The researchers queried patients on their COVID-19 symptoms, how long symptoms lasted, treatments they received, and hospitalization details. COVID-19 symptoms assessed included fever, sore throat, new cough, nasal congestion/rhinorrhea, dyspnea, chest pain, rash, myalgia, fatigue/malaise, headache, nausea/vomiting, diarrhea, anosmia, dysgeusia, and joint pain.
Patients completed surveys about symptoms at 4 weeks and 3 months after infection. Long COVID, or PASC, was defined as any persistent symptom at the times assessed.
Vaccinated patients fared better across outcomes
At 4 weeks after infection, 41% of fully vaccinated patients had at least one persistent symptom, compared with 54% of unvaccinated or partially vaccinated patients (P = .04). At 3 months after infection, 21% of fully vaccinated patients had at least one persistent symptom, compared with 41% of unvaccinated or partially vaccinated patients (P < .0001).
Vaccinated patients were half as likely to have long COVID at 4 weeks after infection (adjusted odds ratio, 0.49) and 90% less likely to have long COVID 3 months after infection (aOR, 0.1), after adjustment for age, sex, race, comorbidities, and use of any of four immune-suppressing medications (anti-CD20 monoclonal antibodies, methotrexate, mycophenolate, or glucocorticoids).
Fully vaccinated patients with breakthrough infections had an average 21 additional days without symptoms during follow-up, compared with unvaccinated and partially vaccinated patients (P = .04).
Reduced risk of long COVID did not change for vaccinated patients after sensitivity analyses for those who did not receive nirmatrelvir/ritonavir (Paxlovid) or monoclonal antibodies, those who didn’t receive any COVID-19-related treatment, those who completed their questionnaires within 6 months after infection, and those who were not hospitalized.
“One important message is that among those who did get PASC, the severity appears similar among those with and without a breakthrough infection,” Dr. Wallace said. “This highlights the need for ongoing research to improve recognition, diagnosis, and treatment of PASC.”
Many more breakthrough infections (72%) than nonbreakthrough infections (2%) occurred during Omicron. The authors acknowledged that different variants might play a role in different long COVID risks but said such potential confounding is unlikely to fully explain the results.
“Even with data suggesting that the Omicron variants may be intrinsically less severe, vaccination still has an impact on severity of infection, rates of hospitalization, and other outcomes and thus may play a role in the risk of PASC,” lead author Naomi Patel, MD, an instructor at Harvard Medical School and a rheumatologist at Massachusetts General Hospital, said in an interview. “A study evaluating the proportions with PASC by vaccination status during the time in which a single variant is predominant, such as the early Omicron era, could help to better assess the more isolated impact of vaccination on PASC.”
Dr. Calabrese said he is convinced that Omicron infections are less likely to result in more severe forms of acute COVID than pre-Omicron infections, and he suspects Omicron infections are also less likely to result in long COVID, although less evidence currently supports this hypothesis.
Hospitalization was more common in unvaccinated/partly vaccinated patients than in vaccinated patients (27% vs. 5%; P = .001). Although pain and fatigue were lower in those with breakthrough infections, functional scores and health-related quality of life were similar in both groups.
Some symptoms significantly differed between vaccinated and unvaccinated/partly vaccinated groups, possibly caused partly by different variants. Nasal congestion was more common (73%) in those with breakthrough infections than in those with nonbreakthrough infections (46%; P < .0001). Those who were unvaccinated/partly vaccinated were significantly more likely to have loss of smell (46% vs. 22%) or taste (45% vs. 28%) or to have joint pain (11% vs. 4%).
Treatment with nirmatrelvir/ritonavir was also more common in vaccinated patients (12%) than in unvaccinated/partly vaccinated patients (1%; P < .0001), as was treatment with monoclonal antibodies (34% vs. 8%; P < .0001).
The study was limited by its low diversity and being at a single health care system, the authors said. Study coauthor Jeffrey A. Sparks, MD, MMSc, an assistant professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, said in an interview that the group is planning additional studies as their cohort grows, including “investigating the relationships between COVID-19 and specific rheumatic diseases and immunomodulating medications, expansion of autoimmunity and systemic inflammation, and lung damage among specific patient populations.”
Dr. Calabrese said it will be important for follow-up study of the symptomatic patients to “determine how many of these patients will fit the clinical picture of long COVID or long-haul phenotypes over the months and years ahead, including documenting exertional malaise and quality of life.
This study only assessed patients who received zero, one, or two doses of a vaccine, but many patients with rheumatic disease today will likely have received booster doses. However, Dr. Calabrese said it would be difficult to quantify whether a third, fourth, or fifth dose offers additional protection from long-term COVID complications after full vaccination or hybrid vaccination.
The research was funded by the Rheumatology Research Foundation, the National Institutes of Health, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Wallace has received research support from Bristol-Myers Squibb and Principia/Sanofi and consulting fees from Zenas BioPharma, Horizon, Sanofi, Shionogi, Viela Bio, and Medpace. Dr. Sparks has received research support from Bristol-Myers Squibb and consulting fees from AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer. Dr. Patel has received consulting fees from FVC Health. Calabrese has consulted for Genentech, Sanofi-Regeneron, AstraZeneca, and GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
Patients with rheumatic disease are at least half as likely to develop long COVID after a SARS-CoV-2 infection if they have been fully vaccinated against COVID-19, according to research published in Annals of the Rheumatic Diseases (2022 Nov 28. doi: 10.1136/ard-2022-223439).
“Moreover, those who were vaccinated prior to getting COVID-19 had less pain and fatigue after their infection,” Zachary S. Wallace, MD, MSc, an assistant professor of medicine at Harvard Medical School, Boston, and a study author, said in an interview. “These findings reinforce the importance of vaccination in this population.”
Messaging around the value of COVID vaccination has been confusing for some with rheumatic disease “because our concern regarding a blunted response to vaccination has led many patients to think that they do not provide much benefit if they are on immunosuppression,” Dr. Wallace said. “In our cohort, which included many patients on immunosuppression of varying degrees, being vaccinated was quite beneficial.”
Leonard H. Calabrese, DO, director of the R.J. Fasenmyer Center for Clinical Immunology and a professor of medicine at the Cleveland Clinic, said in an interview that the study is an “extremely important contribution to our understanding of COVID-19 and its pattern of recovery in patients with immune-mediated inflammatory diseases [IMIDs].” Remaining unanswered questions are “whether patients with IMIDs develop more frequent PASC [post–acute sequelae of COVID-19] from COVID-19 and, if so, is it milder or more severe, and does it differ in its clinical phenotype?”
Long COVID risk assessed at 4 weeks and 3 months after infection
The researchers prospectively tracked 280 adult patients in the Mass General Brigham health care system in the greater Boston area who had systemic autoimmune rheumatic diseases and had an acute COVID-19 infection between March 2020 and July 2022. Patients were an average 53 years old, and most were White (82%) and female (80%). More than half (59%) had inflammatory arthritis, a quarter (24%) had connective tissue disease, and most others had a vasculitis condition or multiple conditions.
A total of 11% of patients were unvaccinated, 28% were partially vaccinated with one mRNA COVID-19 vaccine dose, and 41% were fully vaccinated with two mRNA vaccine doses or one Johnson & Johnson dose. The 116 fully vaccinated patients were considered to have a breakthrough infection while the other 164 were considered to have a nonbreakthrough infection. The breakthrough and nonbreakthrough groups were similar in terms of age, sex, race, ethnicity, smoking status, and type of rheumatic disease. Comorbidities were also similar, except obesity, which was more common in the non–breakthrough infection group (25%) than the breakthrough infection group (10%).
The researchers queried patients on their COVID-19 symptoms, how long symptoms lasted, treatments they received, and hospitalization details. COVID-19 symptoms assessed included fever, sore throat, new cough, nasal congestion/rhinorrhea, dyspnea, chest pain, rash, myalgia, fatigue/malaise, headache, nausea/vomiting, diarrhea, anosmia, dysgeusia, and joint pain.
Patients completed surveys about symptoms at 4 weeks and 3 months after infection. Long COVID, or PASC, was defined as any persistent symptom at the times assessed.
Vaccinated patients fared better across outcomes
At 4 weeks after infection, 41% of fully vaccinated patients had at least one persistent symptom, compared with 54% of unvaccinated or partially vaccinated patients (P = .04). At 3 months after infection, 21% of fully vaccinated patients had at least one persistent symptom, compared with 41% of unvaccinated or partially vaccinated patients (P < .0001).
Vaccinated patients were half as likely to have long COVID at 4 weeks after infection (adjusted odds ratio, 0.49) and 90% less likely to have long COVID 3 months after infection (aOR, 0.1), after adjustment for age, sex, race, comorbidities, and use of any of four immune-suppressing medications (anti-CD20 monoclonal antibodies, methotrexate, mycophenolate, or glucocorticoids).
Fully vaccinated patients with breakthrough infections had an average 21 additional days without symptoms during follow-up, compared with unvaccinated and partially vaccinated patients (P = .04).
Reduced risk of long COVID did not change for vaccinated patients after sensitivity analyses for those who did not receive nirmatrelvir/ritonavir (Paxlovid) or monoclonal antibodies, those who didn’t receive any COVID-19-related treatment, those who completed their questionnaires within 6 months after infection, and those who were not hospitalized.
“One important message is that among those who did get PASC, the severity appears similar among those with and without a breakthrough infection,” Dr. Wallace said. “This highlights the need for ongoing research to improve recognition, diagnosis, and treatment of PASC.”
Many more breakthrough infections (72%) than nonbreakthrough infections (2%) occurred during Omicron. The authors acknowledged that different variants might play a role in different long COVID risks but said such potential confounding is unlikely to fully explain the results.
“Even with data suggesting that the Omicron variants may be intrinsically less severe, vaccination still has an impact on severity of infection, rates of hospitalization, and other outcomes and thus may play a role in the risk of PASC,” lead author Naomi Patel, MD, an instructor at Harvard Medical School and a rheumatologist at Massachusetts General Hospital, said in an interview. “A study evaluating the proportions with PASC by vaccination status during the time in which a single variant is predominant, such as the early Omicron era, could help to better assess the more isolated impact of vaccination on PASC.”
Dr. Calabrese said he is convinced that Omicron infections are less likely to result in more severe forms of acute COVID than pre-Omicron infections, and he suspects Omicron infections are also less likely to result in long COVID, although less evidence currently supports this hypothesis.
Hospitalization was more common in unvaccinated/partly vaccinated patients than in vaccinated patients (27% vs. 5%; P = .001). Although pain and fatigue were lower in those with breakthrough infections, functional scores and health-related quality of life were similar in both groups.
Some symptoms significantly differed between vaccinated and unvaccinated/partly vaccinated groups, possibly caused partly by different variants. Nasal congestion was more common (73%) in those with breakthrough infections than in those with nonbreakthrough infections (46%; P < .0001). Those who were unvaccinated/partly vaccinated were significantly more likely to have loss of smell (46% vs. 22%) or taste (45% vs. 28%) or to have joint pain (11% vs. 4%).
Treatment with nirmatrelvir/ritonavir was also more common in vaccinated patients (12%) than in unvaccinated/partly vaccinated patients (1%; P < .0001), as was treatment with monoclonal antibodies (34% vs. 8%; P < .0001).
The study was limited by its low diversity and being at a single health care system, the authors said. Study coauthor Jeffrey A. Sparks, MD, MMSc, an assistant professor of medicine at Brigham and Women’s Hospital and Harvard Medical School, said in an interview that the group is planning additional studies as their cohort grows, including “investigating the relationships between COVID-19 and specific rheumatic diseases and immunomodulating medications, expansion of autoimmunity and systemic inflammation, and lung damage among specific patient populations.”
Dr. Calabrese said it will be important for follow-up study of the symptomatic patients to “determine how many of these patients will fit the clinical picture of long COVID or long-haul phenotypes over the months and years ahead, including documenting exertional malaise and quality of life.
This study only assessed patients who received zero, one, or two doses of a vaccine, but many patients with rheumatic disease today will likely have received booster doses. However, Dr. Calabrese said it would be difficult to quantify whether a third, fourth, or fifth dose offers additional protection from long-term COVID complications after full vaccination or hybrid vaccination.
The research was funded by the Rheumatology Research Foundation, the National Institutes of Health, the R. Bruce and Joan M. Mickey Research Scholar Fund, and the Llura Gund Award for Rheumatoid Arthritis Research and Care. Dr. Wallace has received research support from Bristol-Myers Squibb and Principia/Sanofi and consulting fees from Zenas BioPharma, Horizon, Sanofi, Shionogi, Viela Bio, and Medpace. Dr. Sparks has received research support from Bristol-Myers Squibb and consulting fees from AbbVie, Amgen, Boehringer Ingelheim, Bristol-Myers Squibb, Gilead, Inova Diagnostics, Janssen, Optum, and Pfizer. Dr. Patel has received consulting fees from FVC Health. Calabrese has consulted for Genentech, Sanofi-Regeneron, AstraZeneca, and GlaxoSmithKline.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF THE RHEUMATIC DISEASES
Children and COVID: Hospitalizations provide a tale of two sources
New cases of COVID-19 in children largely held steady over the Thanksgiving holiday, but hospital admissions are telling a somewhat different story.
New pediatric COVID cases for the week ending on Thanksgiving (11/18-11/24) were up by 5.3% over the previous week, but in the most recent week (11/25-12/1) new cases dropped by 2.6%, according to state data collected by the American Academy of Pediatrics and the Children’s Hospital Association.
In both weeks, though, the total case count stayed below 30,000 – a streak that has now lasted 8 weeks – so the actual number of weekly cases remained fairly low, the AAP/CHA weekly report indicates.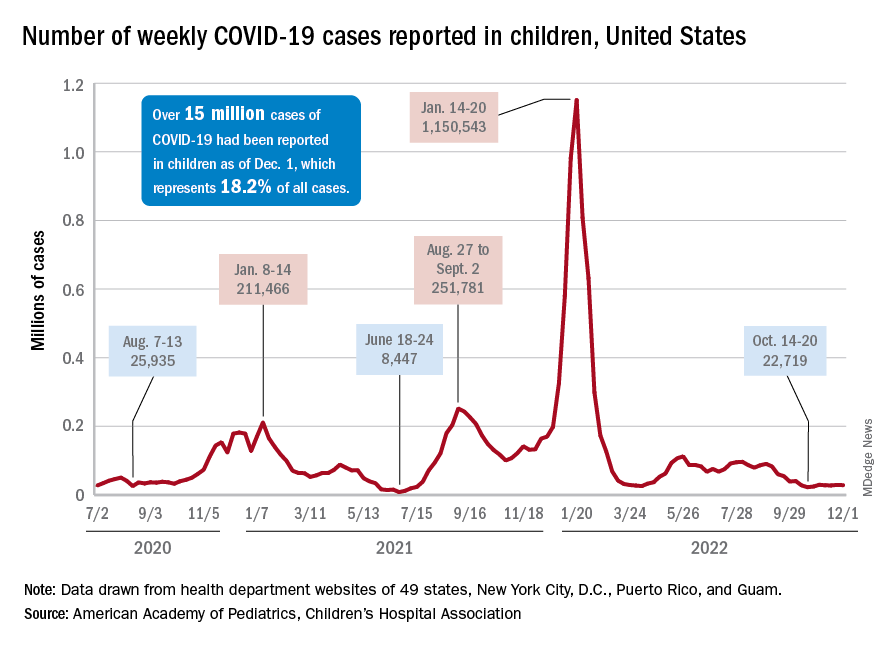
The nation’s emergency departments also experienced a small Thanksgiving bump, as the proportion of visits with diagnosed COVID went from 1.0% of all ED visits for children aged 0-11 years on Nov. 14 to 2.0% on Nov. 27, just 3 days after the official holiday, based on data from the Centers for Disease Control and Prevention. The rate was down to 1.5% on Dec. 1, and similar patterns can be seen for children aged 12-15 and 16-17 years.
New hospital admissions, on the other hand, seem to be following a different path, at least according to the CDC. The hospitalization rate for children aged 0-17 years bottomed out at 0.16 new admissions per 100,000 population back on Oct. 21 and has climbed fairly steadily since then. It was up to 0.20 per 100,000 by Nov. 14, had reached 0.22 per 100,000 on Thanksgiving day (11/24), and then continued to 0.26 per 100,000 by Dec. 2, the latest date for which CDC data are available.
The hospitalization story, however, offers yet another twist. The New York Times, using data from the U.S. Department of Health & Human Services, reports that new COVID-related admissions have held steady at 1.0 per 100,000 since Nov. 18. The rate is much higher than has been reported by the CDC, but no increase can be seen in recent weeks among children, which is not the case for Americans overall, Medscape recently reported.
New cases of COVID-19 in children largely held steady over the Thanksgiving holiday, but hospital admissions are telling a somewhat different story.
New pediatric COVID cases for the week ending on Thanksgiving (11/18-11/24) were up by 5.3% over the previous week, but in the most recent week (11/25-12/1) new cases dropped by 2.6%, according to state data collected by the American Academy of Pediatrics and the Children’s Hospital Association.
In both weeks, though, the total case count stayed below 30,000 – a streak that has now lasted 8 weeks – so the actual number of weekly cases remained fairly low, the AAP/CHA weekly report indicates.
The nation’s emergency departments also experienced a small Thanksgiving bump, as the proportion of visits with diagnosed COVID went from 1.0% of all ED visits for children aged 0-11 years on Nov. 14 to 2.0% on Nov. 27, just 3 days after the official holiday, based on data from the Centers for Disease Control and Prevention. The rate was down to 1.5% on Dec. 1, and similar patterns can be seen for children aged 12-15 and 16-17 years.
New hospital admissions, on the other hand, seem to be following a different path, at least according to the CDC. The hospitalization rate for children aged 0-17 years bottomed out at 0.16 new admissions per 100,000 population back on Oct. 21 and has climbed fairly steadily since then. It was up to 0.20 per 100,000 by Nov. 14, had reached 0.22 per 100,000 on Thanksgiving day (11/24), and then continued to 0.26 per 100,000 by Dec. 2, the latest date for which CDC data are available.
The hospitalization story, however, offers yet another twist. The New York Times, using data from the U.S. Department of Health & Human Services, reports that new COVID-related admissions have held steady at 1.0 per 100,000 since Nov. 18. The rate is much higher than has been reported by the CDC, but no increase can be seen in recent weeks among children, which is not the case for Americans overall, Medscape recently reported.
New cases of COVID-19 in children largely held steady over the Thanksgiving holiday, but hospital admissions are telling a somewhat different story.
New pediatric COVID cases for the week ending on Thanksgiving (11/18-11/24) were up by 5.3% over the previous week, but in the most recent week (11/25-12/1) new cases dropped by 2.6%, according to state data collected by the American Academy of Pediatrics and the Children’s Hospital Association.
In both weeks, though, the total case count stayed below 30,000 – a streak that has now lasted 8 weeks – so the actual number of weekly cases remained fairly low, the AAP/CHA weekly report indicates.
The nation’s emergency departments also experienced a small Thanksgiving bump, as the proportion of visits with diagnosed COVID went from 1.0% of all ED visits for children aged 0-11 years on Nov. 14 to 2.0% on Nov. 27, just 3 days after the official holiday, based on data from the Centers for Disease Control and Prevention. The rate was down to 1.5% on Dec. 1, and similar patterns can be seen for children aged 12-15 and 16-17 years.
New hospital admissions, on the other hand, seem to be following a different path, at least according to the CDC. The hospitalization rate for children aged 0-17 years bottomed out at 0.16 new admissions per 100,000 population back on Oct. 21 and has climbed fairly steadily since then. It was up to 0.20 per 100,000 by Nov. 14, had reached 0.22 per 100,000 on Thanksgiving day (11/24), and then continued to 0.26 per 100,000 by Dec. 2, the latest date for which CDC data are available.
The hospitalization story, however, offers yet another twist. The New York Times, using data from the U.S. Department of Health & Human Services, reports that new COVID-related admissions have held steady at 1.0 per 100,000 since Nov. 18. The rate is much higher than has been reported by the CDC, but no increase can be seen in recent weeks among children, which is not the case for Americans overall, Medscape recently reported.
Study comparing surgical and N95 masks sparks concern
The study’s senior author is John Conly, MD, an infectious disease specialist and professor at the University of Calgary (Alta.), and Alberta Health Services. The findings are not consistent with those of many other studies on this topic.
Commenting about Dr. Conly’s study, Eric Topol, MD, editor-in-chief of Medscape, wrote: “It’s woefully underpowered but ruled out a doubling of hazard for use of medical masks.”
The study, which was partially funded by the World Health Organization, was published online in Annals of Internal Medicine.
This is not the first time that Dr. Conly, who also advises the WHO, has been the subject of controversy. He previously denied that COVID-19 is airborne – a position that is contradicted by strong evidence. In 2021, Dr. Conly made headlines with his controversial claim that N95 respirators can cause harms, including oxygen depletion and carbon dioxide retention.
A detailed examination by the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota, Minneapolis, pointed out numerous scientific flaws in the study, including inconsistent use of both types of masks. The study also examined health care workers in four very different countries (Canada, Israel, Egypt, and Pakistan) during different periods of the pandemic, which may have affected the results. Furthermore, the study did not account for vaccination status and lacked a control group. CIDRAP receives funding from 3M, which makes N95 respirators.
In a commentary published alongside the study, Roger Chou, MD, professor of medicine at Oregon Health & Science University, Portland, said that the results were “not definitive,” with “a generous noninferiority threshold” that is actually “consistent with up to a relative 70% increased risk ... which may be unacceptable to many health workers.”
Lead study author Mark Loeb, MD, professor of infectious diseases at McMaster University, Hamilton, Ont., defended the findings. “The confidence intervals around this, that is, what the possible results could be if the trial was repeated many times, range from −2.5% to 4.9%,” he told this news organization. “This means that the risk of a COVID-19 infection in those using the medical masks could have ranged from anywhere from 2.5% reduction in risk to a 4.9% increase in risk. Readers and policy makers can decide for themselves about this.”
“There is no point continuing to run underpowered, poorly designed studies that are designed to confirm existing biases,” Raina MacIntyre, PhD, professor of global biosecurity and head of the Biosecurity Program at the Kirby Institute, Sydney, said in an interview. “The new study in Annals of Internal Medicine is entirely consistent with our finding that to prevent infection, you need an N95, and it needs to be worn throughout the whole shift. A surgical mask and intermittent use of N95 are equally ineffective. This should not surprise anyone, given a surgical mask is not designed as respiratory protection but is designed to prevent splash or spray of liquid on the face. Only a respirator is designed as respiratory protection through both the seal around the face and the filter of the face piece to prevent inhalation of virus laden aerosols, but you need to wear it continually in a high-risk environment like a hospital.”
“It makes zero sense to do a randomized trial on something you can measure directly,” said Kimberly Prather, PhD, an atmospheric chemist, professor, and director of the NSF Center for Aerosol Impacts on Chemistry of the Environment at the University of California, San Diego. “In fact, many studies have shown aerosols leaking out of surgical masks. Surgical masks are designed to block large spray droplets. Aerosols (0.5-3 mcm), which have been shown to contain infectious SARS-CoV-2 virus, travel with the air flow, and escape.”
“This study ... will be used to justify policies of supplying health care workers, and perhaps patients and visitors, too, with inadequate protection,” Trish Greenhalgh, MD, professor of primary care health sciences at the University of Oxford (England), told this news organization.
“These authors have been pushing back against treating COVID as airborne for 3 years,” David Fisman, MD, an epidemiologist and infectious disease specialist at the University of Toronto, said in an interview. “So, you’ll see these folks brandishing this very flawed trial to justify continuing the infection control practices that have been so disastrous throughout the pandemic.”
The study was funded by the World Health Organization, the Canadian Institutes of Health Research, and the Juravinski Research Institute. Dr. Conly reported receiving grants from the Canadian Institutes for Health Research, Pfizer, and the WHO. Dr. Chou disclosed being a methodologist for WHO guidelines on infection prevention and control measures for COVID-19. Dr. Loeb disclosed payment for expert testimony on personal protective equipment from the government of Manitoba and the Peel District School Board. Dr. MacIntyre has led a large body of research on masks and respirators in health workers, including four randomized clinical trials. She is the author of a book, “Dark Winter: An insider’s guide to pandemics and biosecurity” (Syndey: NewSouth Publishing, 2022), which covers the history and politics of the controversies around N95 and masks. Dr. Prather reported no disclosures. Dr. Greenhalgh is a member of Independent SAGE and an unpaid adviser to the philanthropic fund Balvi. Dr. Fisman has served as a paid legal expert for the Ontario Nurses’ Association in their challenge to Directive 5, which restricted access to N95 masks in health care. He also served as a paid legal expert for the Elementary Teachers’ Federation of Ontario in its efforts to make schools safer in Ontario.
A version of this article first appeared on Medscape.com.
The study’s senior author is John Conly, MD, an infectious disease specialist and professor at the University of Calgary (Alta.), and Alberta Health Services. The findings are not consistent with those of many other studies on this topic.
Commenting about Dr. Conly’s study, Eric Topol, MD, editor-in-chief of Medscape, wrote: “It’s woefully underpowered but ruled out a doubling of hazard for use of medical masks.”
The study, which was partially funded by the World Health Organization, was published online in Annals of Internal Medicine.
This is not the first time that Dr. Conly, who also advises the WHO, has been the subject of controversy. He previously denied that COVID-19 is airborne – a position that is contradicted by strong evidence. In 2021, Dr. Conly made headlines with his controversial claim that N95 respirators can cause harms, including oxygen depletion and carbon dioxide retention.
A detailed examination by the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota, Minneapolis, pointed out numerous scientific flaws in the study, including inconsistent use of both types of masks. The study also examined health care workers in four very different countries (Canada, Israel, Egypt, and Pakistan) during different periods of the pandemic, which may have affected the results. Furthermore, the study did not account for vaccination status and lacked a control group. CIDRAP receives funding from 3M, which makes N95 respirators.
In a commentary published alongside the study, Roger Chou, MD, professor of medicine at Oregon Health & Science University, Portland, said that the results were “not definitive,” with “a generous noninferiority threshold” that is actually “consistent with up to a relative 70% increased risk ... which may be unacceptable to many health workers.”
Lead study author Mark Loeb, MD, professor of infectious diseases at McMaster University, Hamilton, Ont., defended the findings. “The confidence intervals around this, that is, what the possible results could be if the trial was repeated many times, range from −2.5% to 4.9%,” he told this news organization. “This means that the risk of a COVID-19 infection in those using the medical masks could have ranged from anywhere from 2.5% reduction in risk to a 4.9% increase in risk. Readers and policy makers can decide for themselves about this.”
“There is no point continuing to run underpowered, poorly designed studies that are designed to confirm existing biases,” Raina MacIntyre, PhD, professor of global biosecurity and head of the Biosecurity Program at the Kirby Institute, Sydney, said in an interview. “The new study in Annals of Internal Medicine is entirely consistent with our finding that to prevent infection, you need an N95, and it needs to be worn throughout the whole shift. A surgical mask and intermittent use of N95 are equally ineffective. This should not surprise anyone, given a surgical mask is not designed as respiratory protection but is designed to prevent splash or spray of liquid on the face. Only a respirator is designed as respiratory protection through both the seal around the face and the filter of the face piece to prevent inhalation of virus laden aerosols, but you need to wear it continually in a high-risk environment like a hospital.”
“It makes zero sense to do a randomized trial on something you can measure directly,” said Kimberly Prather, PhD, an atmospheric chemist, professor, and director of the NSF Center for Aerosol Impacts on Chemistry of the Environment at the University of California, San Diego. “In fact, many studies have shown aerosols leaking out of surgical masks. Surgical masks are designed to block large spray droplets. Aerosols (0.5-3 mcm), which have been shown to contain infectious SARS-CoV-2 virus, travel with the air flow, and escape.”
“This study ... will be used to justify policies of supplying health care workers, and perhaps patients and visitors, too, with inadequate protection,” Trish Greenhalgh, MD, professor of primary care health sciences at the University of Oxford (England), told this news organization.
“These authors have been pushing back against treating COVID as airborne for 3 years,” David Fisman, MD, an epidemiologist and infectious disease specialist at the University of Toronto, said in an interview. “So, you’ll see these folks brandishing this very flawed trial to justify continuing the infection control practices that have been so disastrous throughout the pandemic.”
The study was funded by the World Health Organization, the Canadian Institutes of Health Research, and the Juravinski Research Institute. Dr. Conly reported receiving grants from the Canadian Institutes for Health Research, Pfizer, and the WHO. Dr. Chou disclosed being a methodologist for WHO guidelines on infection prevention and control measures for COVID-19. Dr. Loeb disclosed payment for expert testimony on personal protective equipment from the government of Manitoba and the Peel District School Board. Dr. MacIntyre has led a large body of research on masks and respirators in health workers, including four randomized clinical trials. She is the author of a book, “Dark Winter: An insider’s guide to pandemics and biosecurity” (Syndey: NewSouth Publishing, 2022), which covers the history and politics of the controversies around N95 and masks. Dr. Prather reported no disclosures. Dr. Greenhalgh is a member of Independent SAGE and an unpaid adviser to the philanthropic fund Balvi. Dr. Fisman has served as a paid legal expert for the Ontario Nurses’ Association in their challenge to Directive 5, which restricted access to N95 masks in health care. He also served as a paid legal expert for the Elementary Teachers’ Federation of Ontario in its efforts to make schools safer in Ontario.
A version of this article first appeared on Medscape.com.
The study’s senior author is John Conly, MD, an infectious disease specialist and professor at the University of Calgary (Alta.), and Alberta Health Services. The findings are not consistent with those of many other studies on this topic.
Commenting about Dr. Conly’s study, Eric Topol, MD, editor-in-chief of Medscape, wrote: “It’s woefully underpowered but ruled out a doubling of hazard for use of medical masks.”
The study, which was partially funded by the World Health Organization, was published online in Annals of Internal Medicine.
This is not the first time that Dr. Conly, who also advises the WHO, has been the subject of controversy. He previously denied that COVID-19 is airborne – a position that is contradicted by strong evidence. In 2021, Dr. Conly made headlines with his controversial claim that N95 respirators can cause harms, including oxygen depletion and carbon dioxide retention.
A detailed examination by the Center for Infectious Disease Research and Policy (CIDRAP) at the University of Minnesota, Minneapolis, pointed out numerous scientific flaws in the study, including inconsistent use of both types of masks. The study also examined health care workers in four very different countries (Canada, Israel, Egypt, and Pakistan) during different periods of the pandemic, which may have affected the results. Furthermore, the study did not account for vaccination status and lacked a control group. CIDRAP receives funding from 3M, which makes N95 respirators.
In a commentary published alongside the study, Roger Chou, MD, professor of medicine at Oregon Health & Science University, Portland, said that the results were “not definitive,” with “a generous noninferiority threshold” that is actually “consistent with up to a relative 70% increased risk ... which may be unacceptable to many health workers.”
Lead study author Mark Loeb, MD, professor of infectious diseases at McMaster University, Hamilton, Ont., defended the findings. “The confidence intervals around this, that is, what the possible results could be if the trial was repeated many times, range from −2.5% to 4.9%,” he told this news organization. “This means that the risk of a COVID-19 infection in those using the medical masks could have ranged from anywhere from 2.5% reduction in risk to a 4.9% increase in risk. Readers and policy makers can decide for themselves about this.”
“There is no point continuing to run underpowered, poorly designed studies that are designed to confirm existing biases,” Raina MacIntyre, PhD, professor of global biosecurity and head of the Biosecurity Program at the Kirby Institute, Sydney, said in an interview. “The new study in Annals of Internal Medicine is entirely consistent with our finding that to prevent infection, you need an N95, and it needs to be worn throughout the whole shift. A surgical mask and intermittent use of N95 are equally ineffective. This should not surprise anyone, given a surgical mask is not designed as respiratory protection but is designed to prevent splash or spray of liquid on the face. Only a respirator is designed as respiratory protection through both the seal around the face and the filter of the face piece to prevent inhalation of virus laden aerosols, but you need to wear it continually in a high-risk environment like a hospital.”
“It makes zero sense to do a randomized trial on something you can measure directly,” said Kimberly Prather, PhD, an atmospheric chemist, professor, and director of the NSF Center for Aerosol Impacts on Chemistry of the Environment at the University of California, San Diego. “In fact, many studies have shown aerosols leaking out of surgical masks. Surgical masks are designed to block large spray droplets. Aerosols (0.5-3 mcm), which have been shown to contain infectious SARS-CoV-2 virus, travel with the air flow, and escape.”
“This study ... will be used to justify policies of supplying health care workers, and perhaps patients and visitors, too, with inadequate protection,” Trish Greenhalgh, MD, professor of primary care health sciences at the University of Oxford (England), told this news organization.
“These authors have been pushing back against treating COVID as airborne for 3 years,” David Fisman, MD, an epidemiologist and infectious disease specialist at the University of Toronto, said in an interview. “So, you’ll see these folks brandishing this very flawed trial to justify continuing the infection control practices that have been so disastrous throughout the pandemic.”
The study was funded by the World Health Organization, the Canadian Institutes of Health Research, and the Juravinski Research Institute. Dr. Conly reported receiving grants from the Canadian Institutes for Health Research, Pfizer, and the WHO. Dr. Chou disclosed being a methodologist for WHO guidelines on infection prevention and control measures for COVID-19. Dr. Loeb disclosed payment for expert testimony on personal protective equipment from the government of Manitoba and the Peel District School Board. Dr. MacIntyre has led a large body of research on masks and respirators in health workers, including four randomized clinical trials. She is the author of a book, “Dark Winter: An insider’s guide to pandemics and biosecurity” (Syndey: NewSouth Publishing, 2022), which covers the history and politics of the controversies around N95 and masks. Dr. Prather reported no disclosures. Dr. Greenhalgh is a member of Independent SAGE and an unpaid adviser to the philanthropic fund Balvi. Dr. Fisman has served as a paid legal expert for the Ontario Nurses’ Association in their challenge to Directive 5, which restricted access to N95 masks in health care. He also served as a paid legal expert for the Elementary Teachers’ Federation of Ontario in its efforts to make schools safer in Ontario.
A version of this article first appeared on Medscape.com.
FROM ANNALS OF INTERNAL MEDICINE
Have long COVID? Newest booster vaccines may help you
Yet at 58, the Arizona writer is in no hurry to get the latest vaccine booster. “I just don’t want to risk getting any sicker,” she said.
Ms. Dishner has had two doses of vaccine plus two boosters. Each time, she had what regulators consider to be mild reactions, including a sore arm, slight fever, nausea, and body aches. Still, there’s some evidence that the newest booster, which protects against some of the later variants, could help people like Ms. Dishner in several ways, said Ziyad Al-Aly, MD, a clinical epidemiologist and prolific long COVID researcher at Washington University in St. Louis.
“A bivalent booster might actually [help with] your long COVID,” he said.
There may be other benefits. “What vaccines or current vaccine boosters do is reduce your risk of progression to severe COVID-19 illness,” Dr. Al-Aly said. “You are avoiding hospital stays or even worse; you’re avoiding potentially fatal outcomes after infection. And that’s really worth it. Who wants to be in the hospital this Christmas holiday?”
Each time people are infected with SARS-CoV-2, the virus that causes COVID-19, they have a fresh risk of not only getting severely ill or dying, but of developing long COVID, Dr. Al-Aly and colleagues found in a study published in Nature Medicine. “If you dodged the bullet the first time and did not get long COVID after the first infection, if you get reinfected, you’re trying your luck again,” Dr. Al-Aly said. “I would advise people not to get reinfected, which is another reason to get the booster.”
In a recent review in The Lancet eClinicalMedicine, an international team of researchers looked at 11 studies that sought to find out if vaccines affected long COVID symptoms. Seven of those studies found that people’s symptoms improved after they were vaccinated, and four found that symptoms mostly remained the same. One found symptoms got worse in some patients.
A study of 28,000 people published in the British Medical Journal found more evidence that vaccination may help ease symptoms. “Vaccination may contribute to a reduction in the population health burden of long COVID,” the team at the United Kingdom’s Office for National Statistics concluded. Most studies found vaccination reduced the risk of getting long COVID in the first place.
Vaccines prompt the body to produce antibodies, which stop a microbe from infecting cells. They also prompt the production of immune cells called T cells, which continue to hunt down and attack a pathogen even after infection.
A booster dose could help rev up that immune response in a patient with long COVID, said Stephen J. Thomas, MD, an infectious disease specialist at Upstate Medical Center in Syracuse, N.Y., and the center’s lead principal investigator for Pfizer/BioNTech’s COVID-19 2020 vaccine trial.
Some scientists believe long COVID might be caused when the virus persists in parts of the body where the immune system isn’t particularly active. Although they don’t fully understand the workings of the many and varied long COVID symptoms, they have a good idea about why people with long COVID often do better after receiving a vaccine or booster.
“The theory is that by boosting, the immune system may be able to ‘mop up’ those virus stragglers that have remained behind after your first cleanup attempt,” Dr. Thomas said.
“The vaccine is almost lending a hand or helping your immune response to clear that virus,” Dr. Al-Aly said.
It could be difficult for long COVID patients to make an informed decision about boosters, given the lack of studies that focus exclusively on the relationship between long COVID and boosters, according to Scott Roberts, MD, associate medical director for infection prevention at Yale New Haven (Conn.) Hospital.
Dr. Roberts recommended that patients speak with their health care providers and read about the bivalent booster on trusted sites such as those sponsored by the Food and Drug Administration and the Centers for Disease Control and Prevention. Long COVID patients should get the latest boosters, especially as there’s no evidence they are unsafe for them. “The antibody response is appropriately boosted, and there is a decent chance this will help reduce the impact of long COVID as well,” he said. “Waiting will only increase the risk of getting infected and increase the chances of long COVID.”
Only 12% of Americans 5 years and older have received the updated booster, according to the CDC, although it’s recommended for everyone. Just over 80% of Americans have gotten at least one vaccine dose. Dr. Thomas understands why the uptake has been so low: Along with people like Ms. Dishner, who fear more side effects or worse symptoms, there are those who believe that hybrid immunity – vaccination immunity plus natural infection – is superior to vaccination alone and that they don’t need a booster.
Studies show that the bivalent boosters, which protect against older and newer variants, can target even the new, predominant COVID-19 strains. Whether that is enough to convince people in the no-booster camp who lost faith when their vaccinated peers started getting COVID-19 is unclear, although, as Dr. Al-Aly has pointed out, vaccinations help keep people from getting so sick that they wind up in the hospital. And, with most of the population having received at least one dose of vaccine, most of those getting infected will naturally come from among the vaccinated.
Thomas describes the expectation that vaccines would prevent everyone from getting sick as “one of the major fails” of the pandemic.
Counting on a vaccine to confer 100% immunity is “a very high bar,” he said. “I think that’s what people expected, and when they weren’t seeing it, they kind of said: ‘Well, what’s the point? You know, things are getting better. I’d rather take my chances than keep going and getting boosted.’ ”
One point – and it’s a critical one – is that vaccination immunity wanes. Plus new variants arise that can evade at least some of the immunity provided by vaccination. That’s why boosters are built into the COVID vaccination program.
While it’s not clear why some long COVID patients see improvements in their symptoms after being vaccinated or boosted and others do not, Dr. Al-Aly said there’s little evidence vaccines can make long COVID worse. “There are some reports out there that some people with long COVID, when they got a vaccine or booster, their symptoms got worse. You’ll read anecdotes on this side,” he said, adding that efforts to see if this is really happening have been inconclusive.
“The general consensus is that vaccines really save lives,” Dr. Al-Aly said. “Getting vaccinated, even if you are a long COVID patient, is better than not getting vaccinated.”
A version of this article first appeared on WebMD.com.
Yet at 58, the Arizona writer is in no hurry to get the latest vaccine booster. “I just don’t want to risk getting any sicker,” she said.
Ms. Dishner has had two doses of vaccine plus two boosters. Each time, she had what regulators consider to be mild reactions, including a sore arm, slight fever, nausea, and body aches. Still, there’s some evidence that the newest booster, which protects against some of the later variants, could help people like Ms. Dishner in several ways, said Ziyad Al-Aly, MD, a clinical epidemiologist and prolific long COVID researcher at Washington University in St. Louis.
“A bivalent booster might actually [help with] your long COVID,” he said.
There may be other benefits. “What vaccines or current vaccine boosters do is reduce your risk of progression to severe COVID-19 illness,” Dr. Al-Aly said. “You are avoiding hospital stays or even worse; you’re avoiding potentially fatal outcomes after infection. And that’s really worth it. Who wants to be in the hospital this Christmas holiday?”
Each time people are infected with SARS-CoV-2, the virus that causes COVID-19, they have a fresh risk of not only getting severely ill or dying, but of developing long COVID, Dr. Al-Aly and colleagues found in a study published in Nature Medicine. “If you dodged the bullet the first time and did not get long COVID after the first infection, if you get reinfected, you’re trying your luck again,” Dr. Al-Aly said. “I would advise people not to get reinfected, which is another reason to get the booster.”
In a recent review in The Lancet eClinicalMedicine, an international team of researchers looked at 11 studies that sought to find out if vaccines affected long COVID symptoms. Seven of those studies found that people’s symptoms improved after they were vaccinated, and four found that symptoms mostly remained the same. One found symptoms got worse in some patients.
A study of 28,000 people published in the British Medical Journal found more evidence that vaccination may help ease symptoms. “Vaccination may contribute to a reduction in the population health burden of long COVID,” the team at the United Kingdom’s Office for National Statistics concluded. Most studies found vaccination reduced the risk of getting long COVID in the first place.
Vaccines prompt the body to produce antibodies, which stop a microbe from infecting cells. They also prompt the production of immune cells called T cells, which continue to hunt down and attack a pathogen even after infection.
A booster dose could help rev up that immune response in a patient with long COVID, said Stephen J. Thomas, MD, an infectious disease specialist at Upstate Medical Center in Syracuse, N.Y., and the center’s lead principal investigator for Pfizer/BioNTech’s COVID-19 2020 vaccine trial.
Some scientists believe long COVID might be caused when the virus persists in parts of the body where the immune system isn’t particularly active. Although they don’t fully understand the workings of the many and varied long COVID symptoms, they have a good idea about why people with long COVID often do better after receiving a vaccine or booster.
“The theory is that by boosting, the immune system may be able to ‘mop up’ those virus stragglers that have remained behind after your first cleanup attempt,” Dr. Thomas said.
“The vaccine is almost lending a hand or helping your immune response to clear that virus,” Dr. Al-Aly said.
It could be difficult for long COVID patients to make an informed decision about boosters, given the lack of studies that focus exclusively on the relationship between long COVID and boosters, according to Scott Roberts, MD, associate medical director for infection prevention at Yale New Haven (Conn.) Hospital.
Dr. Roberts recommended that patients speak with their health care providers and read about the bivalent booster on trusted sites such as those sponsored by the Food and Drug Administration and the Centers for Disease Control and Prevention. Long COVID patients should get the latest boosters, especially as there’s no evidence they are unsafe for them. “The antibody response is appropriately boosted, and there is a decent chance this will help reduce the impact of long COVID as well,” he said. “Waiting will only increase the risk of getting infected and increase the chances of long COVID.”
Only 12% of Americans 5 years and older have received the updated booster, according to the CDC, although it’s recommended for everyone. Just over 80% of Americans have gotten at least one vaccine dose. Dr. Thomas understands why the uptake has been so low: Along with people like Ms. Dishner, who fear more side effects or worse symptoms, there are those who believe that hybrid immunity – vaccination immunity plus natural infection – is superior to vaccination alone and that they don’t need a booster.
Studies show that the bivalent boosters, which protect against older and newer variants, can target even the new, predominant COVID-19 strains. Whether that is enough to convince people in the no-booster camp who lost faith when their vaccinated peers started getting COVID-19 is unclear, although, as Dr. Al-Aly has pointed out, vaccinations help keep people from getting so sick that they wind up in the hospital. And, with most of the population having received at least one dose of vaccine, most of those getting infected will naturally come from among the vaccinated.
Thomas describes the expectation that vaccines would prevent everyone from getting sick as “one of the major fails” of the pandemic.
Counting on a vaccine to confer 100% immunity is “a very high bar,” he said. “I think that’s what people expected, and when they weren’t seeing it, they kind of said: ‘Well, what’s the point? You know, things are getting better. I’d rather take my chances than keep going and getting boosted.’ ”
One point – and it’s a critical one – is that vaccination immunity wanes. Plus new variants arise that can evade at least some of the immunity provided by vaccination. That’s why boosters are built into the COVID vaccination program.
While it’s not clear why some long COVID patients see improvements in their symptoms after being vaccinated or boosted and others do not, Dr. Al-Aly said there’s little evidence vaccines can make long COVID worse. “There are some reports out there that some people with long COVID, when they got a vaccine or booster, their symptoms got worse. You’ll read anecdotes on this side,” he said, adding that efforts to see if this is really happening have been inconclusive.
“The general consensus is that vaccines really save lives,” Dr. Al-Aly said. “Getting vaccinated, even if you are a long COVID patient, is better than not getting vaccinated.”
A version of this article first appeared on WebMD.com.
Yet at 58, the Arizona writer is in no hurry to get the latest vaccine booster. “I just don’t want to risk getting any sicker,” she said.
Ms. Dishner has had two doses of vaccine plus two boosters. Each time, she had what regulators consider to be mild reactions, including a sore arm, slight fever, nausea, and body aches. Still, there’s some evidence that the newest booster, which protects against some of the later variants, could help people like Ms. Dishner in several ways, said Ziyad Al-Aly, MD, a clinical epidemiologist and prolific long COVID researcher at Washington University in St. Louis.
“A bivalent booster might actually [help with] your long COVID,” he said.
There may be other benefits. “What vaccines or current vaccine boosters do is reduce your risk of progression to severe COVID-19 illness,” Dr. Al-Aly said. “You are avoiding hospital stays or even worse; you’re avoiding potentially fatal outcomes after infection. And that’s really worth it. Who wants to be in the hospital this Christmas holiday?”
Each time people are infected with SARS-CoV-2, the virus that causes COVID-19, they have a fresh risk of not only getting severely ill or dying, but of developing long COVID, Dr. Al-Aly and colleagues found in a study published in Nature Medicine. “If you dodged the bullet the first time and did not get long COVID after the first infection, if you get reinfected, you’re trying your luck again,” Dr. Al-Aly said. “I would advise people not to get reinfected, which is another reason to get the booster.”
In a recent review in The Lancet eClinicalMedicine, an international team of researchers looked at 11 studies that sought to find out if vaccines affected long COVID symptoms. Seven of those studies found that people’s symptoms improved after they were vaccinated, and four found that symptoms mostly remained the same. One found symptoms got worse in some patients.
A study of 28,000 people published in the British Medical Journal found more evidence that vaccination may help ease symptoms. “Vaccination may contribute to a reduction in the population health burden of long COVID,” the team at the United Kingdom’s Office for National Statistics concluded. Most studies found vaccination reduced the risk of getting long COVID in the first place.
Vaccines prompt the body to produce antibodies, which stop a microbe from infecting cells. They also prompt the production of immune cells called T cells, which continue to hunt down and attack a pathogen even after infection.
A booster dose could help rev up that immune response in a patient with long COVID, said Stephen J. Thomas, MD, an infectious disease specialist at Upstate Medical Center in Syracuse, N.Y., and the center’s lead principal investigator for Pfizer/BioNTech’s COVID-19 2020 vaccine trial.
Some scientists believe long COVID might be caused when the virus persists in parts of the body where the immune system isn’t particularly active. Although they don’t fully understand the workings of the many and varied long COVID symptoms, they have a good idea about why people with long COVID often do better after receiving a vaccine or booster.
“The theory is that by boosting, the immune system may be able to ‘mop up’ those virus stragglers that have remained behind after your first cleanup attempt,” Dr. Thomas said.
“The vaccine is almost lending a hand or helping your immune response to clear that virus,” Dr. Al-Aly said.
It could be difficult for long COVID patients to make an informed decision about boosters, given the lack of studies that focus exclusively on the relationship between long COVID and boosters, according to Scott Roberts, MD, associate medical director for infection prevention at Yale New Haven (Conn.) Hospital.
Dr. Roberts recommended that patients speak with their health care providers and read about the bivalent booster on trusted sites such as those sponsored by the Food and Drug Administration and the Centers for Disease Control and Prevention. Long COVID patients should get the latest boosters, especially as there’s no evidence they are unsafe for them. “The antibody response is appropriately boosted, and there is a decent chance this will help reduce the impact of long COVID as well,” he said. “Waiting will only increase the risk of getting infected and increase the chances of long COVID.”
Only 12% of Americans 5 years and older have received the updated booster, according to the CDC, although it’s recommended for everyone. Just over 80% of Americans have gotten at least one vaccine dose. Dr. Thomas understands why the uptake has been so low: Along with people like Ms. Dishner, who fear more side effects or worse symptoms, there are those who believe that hybrid immunity – vaccination immunity plus natural infection – is superior to vaccination alone and that they don’t need a booster.
Studies show that the bivalent boosters, which protect against older and newer variants, can target even the new, predominant COVID-19 strains. Whether that is enough to convince people in the no-booster camp who lost faith when their vaccinated peers started getting COVID-19 is unclear, although, as Dr. Al-Aly has pointed out, vaccinations help keep people from getting so sick that they wind up in the hospital. And, with most of the population having received at least one dose of vaccine, most of those getting infected will naturally come from among the vaccinated.
Thomas describes the expectation that vaccines would prevent everyone from getting sick as “one of the major fails” of the pandemic.
Counting on a vaccine to confer 100% immunity is “a very high bar,” he said. “I think that’s what people expected, and when they weren’t seeing it, they kind of said: ‘Well, what’s the point? You know, things are getting better. I’d rather take my chances than keep going and getting boosted.’ ”
One point – and it’s a critical one – is that vaccination immunity wanes. Plus new variants arise that can evade at least some of the immunity provided by vaccination. That’s why boosters are built into the COVID vaccination program.
While it’s not clear why some long COVID patients see improvements in their symptoms after being vaccinated or boosted and others do not, Dr. Al-Aly said there’s little evidence vaccines can make long COVID worse. “There are some reports out there that some people with long COVID, when they got a vaccine or booster, their symptoms got worse. You’ll read anecdotes on this side,” he said, adding that efforts to see if this is really happening have been inconclusive.
“The general consensus is that vaccines really save lives,” Dr. Al-Aly said. “Getting vaccinated, even if you are a long COVID patient, is better than not getting vaccinated.”
A version of this article first appeared on WebMD.com.
FROM NATURE MEDICINE
Florida doc dies by suicide after allegedly drugging and raping patients
according to a police statement.
A week later, a Collier County Sheriff’s deputy found Dr. Salata’s body near his Naples home with a gunshot wound to the head, according to police. The medical examiner later ruled it a suicide.
Dr. Salata co-owned Pura Vida Medical Spa in Naples with his wife Jill Salata, a certified family nurse practitioner. They specialized in cosmetic treatment and surgery.
Naples police said that they arrested Dr. Salata after two female patients accused the doctor of allegedly drugging and raping them while they were still unconscious.
Both victims described being given nitrous oxide, also called laughing gas, for sedation and pain from the cosmetic procedure. The first victim, age 51, said Dr. Salata prescribed alprazolam (Xanax) to take before the procedure and then also gave her nitrous oxide and tequila, causing her to black out, according to NBC2 News.
The second victim, age 72, told police that as the nitrous oxide was wearing off, she found Dr. Salata performing sexual intercourse. The victim felt shocked after the sedation subsided about what had taken place, contacted police, and submitted to a sexual assault examination, according to the police statement.
At Dr. Salata’s November 22 hearing before Judge Michael Provost, a prosecutor asked the judge whether Dr. Salata should surrender his firearms; Provost reportedly dismissed the idea.
“It is disappointing and frustrating that Dr. Salata has escaped justice,” said one victim’s attorney, Adam Horowitz, in a blog post. “Yet, we are relieved that no other women will be assaulted by Dr. Salata again. It took tremendous courage for my client to tell her truth. She was ready to hold him accountable in court.”
Horowitz says he plans to file a civil lawsuit on behalf of his client against Dr. Salata’s estate. The Naples police are continuing their investigation into the victims’ cases, which now includes a third woman, said spokesman Lt. Bryan McGinn.
Meanwhile, the Pura Vida Medical Spa has closed permanently and its website has been deleted. One reviewer named Soul F. wrote on the spa’s Yelp page: “And now may God have mercy on this rapist’s soul. Amen.”
A version of this article first appeared on Medscape.com.
according to a police statement.
A week later, a Collier County Sheriff’s deputy found Dr. Salata’s body near his Naples home with a gunshot wound to the head, according to police. The medical examiner later ruled it a suicide.
Dr. Salata co-owned Pura Vida Medical Spa in Naples with his wife Jill Salata, a certified family nurse practitioner. They specialized in cosmetic treatment and surgery.
Naples police said that they arrested Dr. Salata after two female patients accused the doctor of allegedly drugging and raping them while they were still unconscious.
Both victims described being given nitrous oxide, also called laughing gas, for sedation and pain from the cosmetic procedure. The first victim, age 51, said Dr. Salata prescribed alprazolam (Xanax) to take before the procedure and then also gave her nitrous oxide and tequila, causing her to black out, according to NBC2 News.
The second victim, age 72, told police that as the nitrous oxide was wearing off, she found Dr. Salata performing sexual intercourse. The victim felt shocked after the sedation subsided about what had taken place, contacted police, and submitted to a sexual assault examination, according to the police statement.
At Dr. Salata’s November 22 hearing before Judge Michael Provost, a prosecutor asked the judge whether Dr. Salata should surrender his firearms; Provost reportedly dismissed the idea.
“It is disappointing and frustrating that Dr. Salata has escaped justice,” said one victim’s attorney, Adam Horowitz, in a blog post. “Yet, we are relieved that no other women will be assaulted by Dr. Salata again. It took tremendous courage for my client to tell her truth. She was ready to hold him accountable in court.”
Horowitz says he plans to file a civil lawsuit on behalf of his client against Dr. Salata’s estate. The Naples police are continuing their investigation into the victims’ cases, which now includes a third woman, said spokesman Lt. Bryan McGinn.
Meanwhile, the Pura Vida Medical Spa has closed permanently and its website has been deleted. One reviewer named Soul F. wrote on the spa’s Yelp page: “And now may God have mercy on this rapist’s soul. Amen.”
A version of this article first appeared on Medscape.com.
according to a police statement.
A week later, a Collier County Sheriff’s deputy found Dr. Salata’s body near his Naples home with a gunshot wound to the head, according to police. The medical examiner later ruled it a suicide.
Dr. Salata co-owned Pura Vida Medical Spa in Naples with his wife Jill Salata, a certified family nurse practitioner. They specialized in cosmetic treatment and surgery.
Naples police said that they arrested Dr. Salata after two female patients accused the doctor of allegedly drugging and raping them while they were still unconscious.
Both victims described being given nitrous oxide, also called laughing gas, for sedation and pain from the cosmetic procedure. The first victim, age 51, said Dr. Salata prescribed alprazolam (Xanax) to take before the procedure and then also gave her nitrous oxide and tequila, causing her to black out, according to NBC2 News.
The second victim, age 72, told police that as the nitrous oxide was wearing off, she found Dr. Salata performing sexual intercourse. The victim felt shocked after the sedation subsided about what had taken place, contacted police, and submitted to a sexual assault examination, according to the police statement.
At Dr. Salata’s November 22 hearing before Judge Michael Provost, a prosecutor asked the judge whether Dr. Salata should surrender his firearms; Provost reportedly dismissed the idea.
“It is disappointing and frustrating that Dr. Salata has escaped justice,” said one victim’s attorney, Adam Horowitz, in a blog post. “Yet, we are relieved that no other women will be assaulted by Dr. Salata again. It took tremendous courage for my client to tell her truth. She was ready to hold him accountable in court.”
Horowitz says he plans to file a civil lawsuit on behalf of his client against Dr. Salata’s estate. The Naples police are continuing their investigation into the victims’ cases, which now includes a third woman, said spokesman Lt. Bryan McGinn.
Meanwhile, the Pura Vida Medical Spa has closed permanently and its website has been deleted. One reviewer named Soul F. wrote on the spa’s Yelp page: “And now may God have mercy on this rapist’s soul. Amen.”
A version of this article first appeared on Medscape.com.
Infant BCG vaccine protects only those under age 5 years
Bacillus Calmette-Guérin (BCG) vaccines are given to more than 100 million children every year, but there is considerable debate regarding the effectiveness of BCG vaccination in preventing tuberculosis and death, particularly among older children and adults.
The most extensive study ever conducted on the efficacy of the BCG vaccine for protection against tuberculosis, stratified by age and history of previous tuberculosis, was published in September 2022 in The Lancet Global Health. The study, which comprises a systematic review and meta-analysis, analyzed individual-level data from 26 case-contact cohort studies published over the past 20 years. The studies included data from 70,000 participants. The primary outcome was a composite of prevalent (diagnosed at or within 90 days of baseline) and incident (diagnosed more than 90 days after baseline) tuberculosis in contacts exposed to tuberculosis. Secondary outcomes were pulmonary tuberculosis, extrapulmonary tuberculosis, and mortality.
Participants were characterized as having been exposed to tuberculosis if they were reported to have been a close contact (either living in the same household or having substantial interaction outside the household) of a person with microbiologically or radiologically diagnosed pulmonary tuberculosis. Previous tuberculosis was defined as a positive interferon-gamma (IFN-gamma) release assay or tuberculin skin test, also known as PPD or Mantoux test.
Most studies included in the analysis were conducted in the past 10 years in countries with a high tuberculosis burden. Those countries included India, South Africa, China, Vietnam, Indonesia, Uganda, the Gambia, and Brazil.
Primary outcomes
The study’s main findings included the following:
- The overall effectiveness of BCG vaccination against all forms of tuberculosis was 18% (adjusted odds ratio, 0.82; 95% CI, 0.74-0.91).
- Stratified by age, BCG vaccination only significantly protected against all tuberculosis in children younger than 5 years (aOR, 0.63; 95% CI, 0.49-0.81).
- There was no protective effect among those whose previous tests for tuberculosis were negative unless they were younger than 5 years (aOR, 0.54; 95% CI, 0.32-0.90).
- Among contacts who had a positive tuberculin skin test or IFN-gamma release assay, BCG vaccination significantly protected against tuberculosis among all participants (aOR, 0.81; 95% CI, 0.69-0.96), participants younger than 5 years (aOR, 0.68; 95% CI, 0.47-0.97), and participants aged 5-9 years (aOR, 0.62; 95% CI, 0.38-0.99).
- BCG vaccination was protective against pulmonary tuberculosis (19% effectiveness), but this effect was only seen in children younger than 3 years (42% effectiveness) when stratified by age.
- Protection against all tuberculosis and pulmonary tuberculosis was greater among female participants than male participants.
“This is a definitive BCG protection study because it involves a significant number of individuals evaluated using this meta-analysis. Protection is clearly lost with age. From as early as age 5, no protective effect can be observed. Protection, including against pulmonary tuberculosis, can be observed up to 3 years of age,” stated study author Julio Croda, MD, PhD, chair of the Brazilian Society of Tropical Medicine.
Dr. Croda emphasized that the findings from their study indicate that BCG vaccine protects against pulmonary tuberculosis and that those results differ from results of some previous studies.
“Every physician believes the BCG vaccine protects against serious forms of tuberculosis up to age 5. That fact is not surprising at all,” Dr. Croda remarked. “However, the fact that it protects against pulmonary tuberculosis, especially in children younger than 3, was surprising. In medical practice, we did not believe in this protection.”
Currently, 1.2% of new tuberculosis cases in Brazil occur among those younger than 5. Nevertheless, these cases represent 40.1% of new diagnoses recorded among those younger than 15, highlighting the importance of protection for this age group. An increase in extrapulmonary tuberculosis cases was recently observed in patients younger than 5.
Isabella Ballalai, MD, PhD, is deputy chair of the Brazilian Society of Immunizations. Although she did not participate in this study, she commented on its findings. “All publications are welcome; they help us think,” she explained. She emphasized that the BCG vaccine is not optimal. “There are studies indicating 80% efficacy and others indicating 0%. So, what we can look at is decades of effectiveness in practice.”
Dr. Ballalai explained that the BCG vaccine could keep severe forms of tuberculosis, meningitis, and miliary tuberculosis at bay. She shared her experience of caring for several patients with tuberculous meningitis shortly after she had graduated. “Today, thanks to the BCG vaccine, we don’t see it anymore.” However, she pointed out that the vaccine›s efficacy and effectiveness against pulmonary tuberculosis are low and that pulmonary tuberculosis remains the most significant problem among adults.
Dr. Ballalai also emphasized a few shortcomings of the study. “One is the definition of ‘vaccinated’ and ‘unvaccinated,’ which was based on the presence or absence of a mark on the arm. Today, we know that the absence of a mark does not indicate that the child has not been vaccinated, nor that the vaccine has not been effective. Therefore, several vaccinated participants may have been included amongst the unvaccinated participants.”
The authors emphasized that the definition of “vaccinated” and “unvaccinated” was based on a scar and on vaccination records, and they recognized that participants who did not have a scar on their arm could have been misclassified. Regardless, it is still considered a sensitive indicator. “Few vaccinated children from various settings do not show a scar years after vaccine administration,” they stated in their article.
Adults unprotected
Dr. Ballalai also shared her concerns regarding the lack of protection for older individuals. “We know those older than 60 are at greater risk for complications of tuberculosis. Individuals in this age group naturally have a lower immunity, and they usually have comorbidities. From this study, I can only conclude what was already expected: that adults who received a BCG vaccine as infants are not clear of pulmonary tuberculosis.”
Dr. Croda agreed that it was already evident that the BCG vaccine administered at birth did not provide protection for adults. “In the past, even with 80%-90% vaccine coverage, there were numerous tuberculosis cases in adults in Brazil.”
Are boosters needed?
The authors concluded that immunoprotection needs to be boosted in older populations, as vaccination at birth is ineffective for adolescents and adults. They have also discussed whether children older than 10 years and adults could benefit from a booster shot.
Dr. Croda emphasized that there is no indication for this, because there are no data regarding protection with a booster dose during adulthood. However, he cited a study conducted in South Africa in which the BCG vaccine was compared with another vaccine, and another study, which is being conducted in India, is assessing whether a BCG booster offers protection against pulmonary tuberculosis. “There are few studies. Perhaps the revaccination of more vulnerable groups could be of interest, but additional studies are needed first.”
Dr. Croda intends to assess revaccination in those deprived of liberty, in which the incidence of tuberculosis is very high. From 2015 to 2021, many new cases were recorded in this population in Brazil. The number rose from 5,860 to 6,773 during that period.
“However, BCG revaccination carries a significant risk of patients presenting with serious adverse events,” Dr. Ballalai pointed out. He noted that several years ago, to extend protection, Brazil adopted a booster program for persons aged 10 years or older, but the program was discontinued owing to the numerous adverse events reported and the absence of evidence of benefit from increased protection against tuberculosis.
“The adult groups at greater risk for severe tuberculosis manifestations normally presented with an underlying disease, particularly in immunocompromised patient groups. The [administration of the] BCG [vaccine] is contraindicated for those who are immunocompromised. And, for the older population, we do not have data on [vaccine] safety,” she emphasized.
Nonspecific immune protection
One of the study’s secondary outcomes regarded mortality. Four studies in the meta-analysis followed up tuberculosis contacts for death. In these studies, which evaluated 20,000 participants, BCG vaccination was shown to be significantly protective against death for participants younger than 15 years.
However, the authors urged caution in interpreting these data. They emphasized that they were unable to identify specific mechanisms by which BCG vaccination might have reduced mortality, and there are possible study biases that could have led to an overestimation of mortality benefit. Moreover, given the observational nature of the included studies, vaccinated children might have had higher socioeconomic status and greater access to health care, and they may have been more likely to have received other vaccinations, compared with children who did not receive BCG vaccines.
Nevertheless, previous experimental and observational studies have found that BCG vaccination might provide nonspecific or off-target immune protection against an array of other pathogens.
“In small studies conducted in Africa, those younger than 5 were protected not only against tuberculosis but also against other respiratory diseases,” Dr. Croda affirmed. “However, these are small studies, and for now, there is no recommendation for using BCG vaccination to prevent other respiratory infections.”
A long-awaited, critical study on the impact of the BCG vaccine on COVID-19, in which Brazilian researchers participated, will be published in the New England Journal of Medicine.
New vaccines needed
The BCG vaccine is one of the oldest vaccines, but there are still several crucial unanswered questions about its use.
Previously published studies that examined the protective effect of BCG vaccination only considered low-burden settings and the historical literature before 1950. These studies need updating, but doing so has not been a simple task. To answer their questions, individual-level participant data for a prespecified list of variables, including the characteristics of the exposed participant (contact), the index case, and the environment, were requested from authors of all eligible studies.
Much of the data used in the published research were found through discussions with authors and experts in the field, as well as through data deposited in data storage repositories, conference abstracts, dissertations, and even direct requests to the authors. “The Pan-American Health Organization helped with this data collection and contacting some authors,” said Dr. Croda.
With the new data, the authors confirmed that infant BCG vaccination, although important to young children who are at high risk for tuberculosis, does not prevent adult-type cavitary tuberculosis and is therefore insufficient to impede the tuberculosis epidemic. “Novel vaccines are urgently needed,” they concluded.
“We need to develop novel, more effective vaccines, which, when administered during infancy, would ensure lifelong protection,” Dr. Croda added.
Dr. Croda and Dr. Ballalai reported no relevant financial relationships.
This article was translated from the Medscape Portuguese edition. A version of this article appeared on Medscape.com.
Bacillus Calmette-Guérin (BCG) vaccines are given to more than 100 million children every year, but there is considerable debate regarding the effectiveness of BCG vaccination in preventing tuberculosis and death, particularly among older children and adults.
The most extensive study ever conducted on the efficacy of the BCG vaccine for protection against tuberculosis, stratified by age and history of previous tuberculosis, was published in September 2022 in The Lancet Global Health. The study, which comprises a systematic review and meta-analysis, analyzed individual-level data from 26 case-contact cohort studies published over the past 20 years. The studies included data from 70,000 participants. The primary outcome was a composite of prevalent (diagnosed at or within 90 days of baseline) and incident (diagnosed more than 90 days after baseline) tuberculosis in contacts exposed to tuberculosis. Secondary outcomes were pulmonary tuberculosis, extrapulmonary tuberculosis, and mortality.
Participants were characterized as having been exposed to tuberculosis if they were reported to have been a close contact (either living in the same household or having substantial interaction outside the household) of a person with microbiologically or radiologically diagnosed pulmonary tuberculosis. Previous tuberculosis was defined as a positive interferon-gamma (IFN-gamma) release assay or tuberculin skin test, also known as PPD or Mantoux test.
Most studies included in the analysis were conducted in the past 10 years in countries with a high tuberculosis burden. Those countries included India, South Africa, China, Vietnam, Indonesia, Uganda, the Gambia, and Brazil.
Primary outcomes
The study’s main findings included the following:
- The overall effectiveness of BCG vaccination against all forms of tuberculosis was 18% (adjusted odds ratio, 0.82; 95% CI, 0.74-0.91).
- Stratified by age, BCG vaccination only significantly protected against all tuberculosis in children younger than 5 years (aOR, 0.63; 95% CI, 0.49-0.81).
- There was no protective effect among those whose previous tests for tuberculosis were negative unless they were younger than 5 years (aOR, 0.54; 95% CI, 0.32-0.90).
- Among contacts who had a positive tuberculin skin test or IFN-gamma release assay, BCG vaccination significantly protected against tuberculosis among all participants (aOR, 0.81; 95% CI, 0.69-0.96), participants younger than 5 years (aOR, 0.68; 95% CI, 0.47-0.97), and participants aged 5-9 years (aOR, 0.62; 95% CI, 0.38-0.99).
- BCG vaccination was protective against pulmonary tuberculosis (19% effectiveness), but this effect was only seen in children younger than 3 years (42% effectiveness) when stratified by age.
- Protection against all tuberculosis and pulmonary tuberculosis was greater among female participants than male participants.
“This is a definitive BCG protection study because it involves a significant number of individuals evaluated using this meta-analysis. Protection is clearly lost with age. From as early as age 5, no protective effect can be observed. Protection, including against pulmonary tuberculosis, can be observed up to 3 years of age,” stated study author Julio Croda, MD, PhD, chair of the Brazilian Society of Tropical Medicine.
Dr. Croda emphasized that the findings from their study indicate that BCG vaccine protects against pulmonary tuberculosis and that those results differ from results of some previous studies.
“Every physician believes the BCG vaccine protects against serious forms of tuberculosis up to age 5. That fact is not surprising at all,” Dr. Croda remarked. “However, the fact that it protects against pulmonary tuberculosis, especially in children younger than 3, was surprising. In medical practice, we did not believe in this protection.”
Currently, 1.2% of new tuberculosis cases in Brazil occur among those younger than 5. Nevertheless, these cases represent 40.1% of new diagnoses recorded among those younger than 15, highlighting the importance of protection for this age group. An increase in extrapulmonary tuberculosis cases was recently observed in patients younger than 5.
Isabella Ballalai, MD, PhD, is deputy chair of the Brazilian Society of Immunizations. Although she did not participate in this study, she commented on its findings. “All publications are welcome; they help us think,” she explained. She emphasized that the BCG vaccine is not optimal. “There are studies indicating 80% efficacy and others indicating 0%. So, what we can look at is decades of effectiveness in practice.”
Dr. Ballalai explained that the BCG vaccine could keep severe forms of tuberculosis, meningitis, and miliary tuberculosis at bay. She shared her experience of caring for several patients with tuberculous meningitis shortly after she had graduated. “Today, thanks to the BCG vaccine, we don’t see it anymore.” However, she pointed out that the vaccine›s efficacy and effectiveness against pulmonary tuberculosis are low and that pulmonary tuberculosis remains the most significant problem among adults.
Dr. Ballalai also emphasized a few shortcomings of the study. “One is the definition of ‘vaccinated’ and ‘unvaccinated,’ which was based on the presence or absence of a mark on the arm. Today, we know that the absence of a mark does not indicate that the child has not been vaccinated, nor that the vaccine has not been effective. Therefore, several vaccinated participants may have been included amongst the unvaccinated participants.”
The authors emphasized that the definition of “vaccinated” and “unvaccinated” was based on a scar and on vaccination records, and they recognized that participants who did not have a scar on their arm could have been misclassified. Regardless, it is still considered a sensitive indicator. “Few vaccinated children from various settings do not show a scar years after vaccine administration,” they stated in their article.
Adults unprotected
Dr. Ballalai also shared her concerns regarding the lack of protection for older individuals. “We know those older than 60 are at greater risk for complications of tuberculosis. Individuals in this age group naturally have a lower immunity, and they usually have comorbidities. From this study, I can only conclude what was already expected: that adults who received a BCG vaccine as infants are not clear of pulmonary tuberculosis.”
Dr. Croda agreed that it was already evident that the BCG vaccine administered at birth did not provide protection for adults. “In the past, even with 80%-90% vaccine coverage, there were numerous tuberculosis cases in adults in Brazil.”
Are boosters needed?
The authors concluded that immunoprotection needs to be boosted in older populations, as vaccination at birth is ineffective for adolescents and adults. They have also discussed whether children older than 10 years and adults could benefit from a booster shot.
Dr. Croda emphasized that there is no indication for this, because there are no data regarding protection with a booster dose during adulthood. However, he cited a study conducted in South Africa in which the BCG vaccine was compared with another vaccine, and another study, which is being conducted in India, is assessing whether a BCG booster offers protection against pulmonary tuberculosis. “There are few studies. Perhaps the revaccination of more vulnerable groups could be of interest, but additional studies are needed first.”
Dr. Croda intends to assess revaccination in those deprived of liberty, in which the incidence of tuberculosis is very high. From 2015 to 2021, many new cases were recorded in this population in Brazil. The number rose from 5,860 to 6,773 during that period.
“However, BCG revaccination carries a significant risk of patients presenting with serious adverse events,” Dr. Ballalai pointed out. He noted that several years ago, to extend protection, Brazil adopted a booster program for persons aged 10 years or older, but the program was discontinued owing to the numerous adverse events reported and the absence of evidence of benefit from increased protection against tuberculosis.
“The adult groups at greater risk for severe tuberculosis manifestations normally presented with an underlying disease, particularly in immunocompromised patient groups. The [administration of the] BCG [vaccine] is contraindicated for those who are immunocompromised. And, for the older population, we do not have data on [vaccine] safety,” she emphasized.
Nonspecific immune protection
One of the study’s secondary outcomes regarded mortality. Four studies in the meta-analysis followed up tuberculosis contacts for death. In these studies, which evaluated 20,000 participants, BCG vaccination was shown to be significantly protective against death for participants younger than 15 years.
However, the authors urged caution in interpreting these data. They emphasized that they were unable to identify specific mechanisms by which BCG vaccination might have reduced mortality, and there are possible study biases that could have led to an overestimation of mortality benefit. Moreover, given the observational nature of the included studies, vaccinated children might have had higher socioeconomic status and greater access to health care, and they may have been more likely to have received other vaccinations, compared with children who did not receive BCG vaccines.
Nevertheless, previous experimental and observational studies have found that BCG vaccination might provide nonspecific or off-target immune protection against an array of other pathogens.
“In small studies conducted in Africa, those younger than 5 were protected not only against tuberculosis but also against other respiratory diseases,” Dr. Croda affirmed. “However, these are small studies, and for now, there is no recommendation for using BCG vaccination to prevent other respiratory infections.”
A long-awaited, critical study on the impact of the BCG vaccine on COVID-19, in which Brazilian researchers participated, will be published in the New England Journal of Medicine.
New vaccines needed
The BCG vaccine is one of the oldest vaccines, but there are still several crucial unanswered questions about its use.
Previously published studies that examined the protective effect of BCG vaccination only considered low-burden settings and the historical literature before 1950. These studies need updating, but doing so has not been a simple task. To answer their questions, individual-level participant data for a prespecified list of variables, including the characteristics of the exposed participant (contact), the index case, and the environment, were requested from authors of all eligible studies.
Much of the data used in the published research were found through discussions with authors and experts in the field, as well as through data deposited in data storage repositories, conference abstracts, dissertations, and even direct requests to the authors. “The Pan-American Health Organization helped with this data collection and contacting some authors,” said Dr. Croda.
With the new data, the authors confirmed that infant BCG vaccination, although important to young children who are at high risk for tuberculosis, does not prevent adult-type cavitary tuberculosis and is therefore insufficient to impede the tuberculosis epidemic. “Novel vaccines are urgently needed,” they concluded.
“We need to develop novel, more effective vaccines, which, when administered during infancy, would ensure lifelong protection,” Dr. Croda added.
Dr. Croda and Dr. Ballalai reported no relevant financial relationships.
This article was translated from the Medscape Portuguese edition. A version of this article appeared on Medscape.com.
Bacillus Calmette-Guérin (BCG) vaccines are given to more than 100 million children every year, but there is considerable debate regarding the effectiveness of BCG vaccination in preventing tuberculosis and death, particularly among older children and adults.
The most extensive study ever conducted on the efficacy of the BCG vaccine for protection against tuberculosis, stratified by age and history of previous tuberculosis, was published in September 2022 in The Lancet Global Health. The study, which comprises a systematic review and meta-analysis, analyzed individual-level data from 26 case-contact cohort studies published over the past 20 years. The studies included data from 70,000 participants. The primary outcome was a composite of prevalent (diagnosed at or within 90 days of baseline) and incident (diagnosed more than 90 days after baseline) tuberculosis in contacts exposed to tuberculosis. Secondary outcomes were pulmonary tuberculosis, extrapulmonary tuberculosis, and mortality.
Participants were characterized as having been exposed to tuberculosis if they were reported to have been a close contact (either living in the same household or having substantial interaction outside the household) of a person with microbiologically or radiologically diagnosed pulmonary tuberculosis. Previous tuberculosis was defined as a positive interferon-gamma (IFN-gamma) release assay or tuberculin skin test, also known as PPD or Mantoux test.
Most studies included in the analysis were conducted in the past 10 years in countries with a high tuberculosis burden. Those countries included India, South Africa, China, Vietnam, Indonesia, Uganda, the Gambia, and Brazil.
Primary outcomes
The study’s main findings included the following:
- The overall effectiveness of BCG vaccination against all forms of tuberculosis was 18% (adjusted odds ratio, 0.82; 95% CI, 0.74-0.91).
- Stratified by age, BCG vaccination only significantly protected against all tuberculosis in children younger than 5 years (aOR, 0.63; 95% CI, 0.49-0.81).
- There was no protective effect among those whose previous tests for tuberculosis were negative unless they were younger than 5 years (aOR, 0.54; 95% CI, 0.32-0.90).
- Among contacts who had a positive tuberculin skin test or IFN-gamma release assay, BCG vaccination significantly protected against tuberculosis among all participants (aOR, 0.81; 95% CI, 0.69-0.96), participants younger than 5 years (aOR, 0.68; 95% CI, 0.47-0.97), and participants aged 5-9 years (aOR, 0.62; 95% CI, 0.38-0.99).
- BCG vaccination was protective against pulmonary tuberculosis (19% effectiveness), but this effect was only seen in children younger than 3 years (42% effectiveness) when stratified by age.
- Protection against all tuberculosis and pulmonary tuberculosis was greater among female participants than male participants.
“This is a definitive BCG protection study because it involves a significant number of individuals evaluated using this meta-analysis. Protection is clearly lost with age. From as early as age 5, no protective effect can be observed. Protection, including against pulmonary tuberculosis, can be observed up to 3 years of age,” stated study author Julio Croda, MD, PhD, chair of the Brazilian Society of Tropical Medicine.
Dr. Croda emphasized that the findings from their study indicate that BCG vaccine protects against pulmonary tuberculosis and that those results differ from results of some previous studies.
“Every physician believes the BCG vaccine protects against serious forms of tuberculosis up to age 5. That fact is not surprising at all,” Dr. Croda remarked. “However, the fact that it protects against pulmonary tuberculosis, especially in children younger than 3, was surprising. In medical practice, we did not believe in this protection.”
Currently, 1.2% of new tuberculosis cases in Brazil occur among those younger than 5. Nevertheless, these cases represent 40.1% of new diagnoses recorded among those younger than 15, highlighting the importance of protection for this age group. An increase in extrapulmonary tuberculosis cases was recently observed in patients younger than 5.
Isabella Ballalai, MD, PhD, is deputy chair of the Brazilian Society of Immunizations. Although she did not participate in this study, she commented on its findings. “All publications are welcome; they help us think,” she explained. She emphasized that the BCG vaccine is not optimal. “There are studies indicating 80% efficacy and others indicating 0%. So, what we can look at is decades of effectiveness in practice.”
Dr. Ballalai explained that the BCG vaccine could keep severe forms of tuberculosis, meningitis, and miliary tuberculosis at bay. She shared her experience of caring for several patients with tuberculous meningitis shortly after she had graduated. “Today, thanks to the BCG vaccine, we don’t see it anymore.” However, she pointed out that the vaccine›s efficacy and effectiveness against pulmonary tuberculosis are low and that pulmonary tuberculosis remains the most significant problem among adults.
Dr. Ballalai also emphasized a few shortcomings of the study. “One is the definition of ‘vaccinated’ and ‘unvaccinated,’ which was based on the presence or absence of a mark on the arm. Today, we know that the absence of a mark does not indicate that the child has not been vaccinated, nor that the vaccine has not been effective. Therefore, several vaccinated participants may have been included amongst the unvaccinated participants.”
The authors emphasized that the definition of “vaccinated” and “unvaccinated” was based on a scar and on vaccination records, and they recognized that participants who did not have a scar on their arm could have been misclassified. Regardless, it is still considered a sensitive indicator. “Few vaccinated children from various settings do not show a scar years after vaccine administration,” they stated in their article.
Adults unprotected
Dr. Ballalai also shared her concerns regarding the lack of protection for older individuals. “We know those older than 60 are at greater risk for complications of tuberculosis. Individuals in this age group naturally have a lower immunity, and they usually have comorbidities. From this study, I can only conclude what was already expected: that adults who received a BCG vaccine as infants are not clear of pulmonary tuberculosis.”
Dr. Croda agreed that it was already evident that the BCG vaccine administered at birth did not provide protection for adults. “In the past, even with 80%-90% vaccine coverage, there were numerous tuberculosis cases in adults in Brazil.”
Are boosters needed?
The authors concluded that immunoprotection needs to be boosted in older populations, as vaccination at birth is ineffective for adolescents and adults. They have also discussed whether children older than 10 years and adults could benefit from a booster shot.
Dr. Croda emphasized that there is no indication for this, because there are no data regarding protection with a booster dose during adulthood. However, he cited a study conducted in South Africa in which the BCG vaccine was compared with another vaccine, and another study, which is being conducted in India, is assessing whether a BCG booster offers protection against pulmonary tuberculosis. “There are few studies. Perhaps the revaccination of more vulnerable groups could be of interest, but additional studies are needed first.”
Dr. Croda intends to assess revaccination in those deprived of liberty, in which the incidence of tuberculosis is very high. From 2015 to 2021, many new cases were recorded in this population in Brazil. The number rose from 5,860 to 6,773 during that period.
“However, BCG revaccination carries a significant risk of patients presenting with serious adverse events,” Dr. Ballalai pointed out. He noted that several years ago, to extend protection, Brazil adopted a booster program for persons aged 10 years or older, but the program was discontinued owing to the numerous adverse events reported and the absence of evidence of benefit from increased protection against tuberculosis.
“The adult groups at greater risk for severe tuberculosis manifestations normally presented with an underlying disease, particularly in immunocompromised patient groups. The [administration of the] BCG [vaccine] is contraindicated for those who are immunocompromised. And, for the older population, we do not have data on [vaccine] safety,” she emphasized.
Nonspecific immune protection
One of the study’s secondary outcomes regarded mortality. Four studies in the meta-analysis followed up tuberculosis contacts for death. In these studies, which evaluated 20,000 participants, BCG vaccination was shown to be significantly protective against death for participants younger than 15 years.
However, the authors urged caution in interpreting these data. They emphasized that they were unable to identify specific mechanisms by which BCG vaccination might have reduced mortality, and there are possible study biases that could have led to an overestimation of mortality benefit. Moreover, given the observational nature of the included studies, vaccinated children might have had higher socioeconomic status and greater access to health care, and they may have been more likely to have received other vaccinations, compared with children who did not receive BCG vaccines.
Nevertheless, previous experimental and observational studies have found that BCG vaccination might provide nonspecific or off-target immune protection against an array of other pathogens.
“In small studies conducted in Africa, those younger than 5 were protected not only against tuberculosis but also against other respiratory diseases,” Dr. Croda affirmed. “However, these are small studies, and for now, there is no recommendation for using BCG vaccination to prevent other respiratory infections.”
A long-awaited, critical study on the impact of the BCG vaccine on COVID-19, in which Brazilian researchers participated, will be published in the New England Journal of Medicine.
New vaccines needed
The BCG vaccine is one of the oldest vaccines, but there are still several crucial unanswered questions about its use.
Previously published studies that examined the protective effect of BCG vaccination only considered low-burden settings and the historical literature before 1950. These studies need updating, but doing so has not been a simple task. To answer their questions, individual-level participant data for a prespecified list of variables, including the characteristics of the exposed participant (contact), the index case, and the environment, were requested from authors of all eligible studies.
Much of the data used in the published research were found through discussions with authors and experts in the field, as well as through data deposited in data storage repositories, conference abstracts, dissertations, and even direct requests to the authors. “The Pan-American Health Organization helped with this data collection and contacting some authors,” said Dr. Croda.
With the new data, the authors confirmed that infant BCG vaccination, although important to young children who are at high risk for tuberculosis, does not prevent adult-type cavitary tuberculosis and is therefore insufficient to impede the tuberculosis epidemic. “Novel vaccines are urgently needed,” they concluded.
“We need to develop novel, more effective vaccines, which, when administered during infancy, would ensure lifelong protection,” Dr. Croda added.
Dr. Croda and Dr. Ballalai reported no relevant financial relationships.
This article was translated from the Medscape Portuguese edition. A version of this article appeared on Medscape.com.
FROM THE LANCET GLOBAL HEALTH
FDA pulls U.S. authorization for Eli Lilly’s COVID drug bebtelovimab
the Food and Drug Administration said, citing it is not expected to neutralize the dominant BQ.1 and BQ.1.1 subvariants of Omicron.
The announcement on Nov. 30 takes away authorization from the last COVID-19 monoclonal antibody treatment, leaving Pfizer’s antiviral drug Paxlovid, Merck’s Lagevrio, and Gilead Sciences’ Veklury as treatments for the disease, besides convalescent plasma for some patients.
AstraZeneca’s monoclonal antibody Evusheld is also authorized for protection against COVID-19 infection in some people.
Eli Lilly and its authorized distributors have paused commercial distribution of the monoclonal antibody until further notice from the agency, while the U.S. government has also paused fulfillment of any pending requests under its scheme to help uninsured and underinsured Americans access the drug.
The drug, which was discovered by Abcellera and commercialized by Eli Lilly, received an authorization from the FDA in February.
BQ.1 and BQ.1.1 have become the dominant strains in the United States after a steady increase in prevalence over the last 2 months, surpassing Omicron’s BA.5 subvariant, which had driven cases earlier in the year.
The subvariants accounted for around 57% of the cases nationally, as per government data last week.
Reuters Health Information © 2022
the Food and Drug Administration said, citing it is not expected to neutralize the dominant BQ.1 and BQ.1.1 subvariants of Omicron.
The announcement on Nov. 30 takes away authorization from the last COVID-19 monoclonal antibody treatment, leaving Pfizer’s antiviral drug Paxlovid, Merck’s Lagevrio, and Gilead Sciences’ Veklury as treatments for the disease, besides convalescent plasma for some patients.
AstraZeneca’s monoclonal antibody Evusheld is also authorized for protection against COVID-19 infection in some people.
Eli Lilly and its authorized distributors have paused commercial distribution of the monoclonal antibody until further notice from the agency, while the U.S. government has also paused fulfillment of any pending requests under its scheme to help uninsured and underinsured Americans access the drug.
The drug, which was discovered by Abcellera and commercialized by Eli Lilly, received an authorization from the FDA in February.
BQ.1 and BQ.1.1 have become the dominant strains in the United States after a steady increase in prevalence over the last 2 months, surpassing Omicron’s BA.5 subvariant, which had driven cases earlier in the year.
The subvariants accounted for around 57% of the cases nationally, as per government data last week.
Reuters Health Information © 2022
the Food and Drug Administration said, citing it is not expected to neutralize the dominant BQ.1 and BQ.1.1 subvariants of Omicron.
The announcement on Nov. 30 takes away authorization from the last COVID-19 monoclonal antibody treatment, leaving Pfizer’s antiviral drug Paxlovid, Merck’s Lagevrio, and Gilead Sciences’ Veklury as treatments for the disease, besides convalescent plasma for some patients.
AstraZeneca’s monoclonal antibody Evusheld is also authorized for protection against COVID-19 infection in some people.
Eli Lilly and its authorized distributors have paused commercial distribution of the monoclonal antibody until further notice from the agency, while the U.S. government has also paused fulfillment of any pending requests under its scheme to help uninsured and underinsured Americans access the drug.
The drug, which was discovered by Abcellera and commercialized by Eli Lilly, received an authorization from the FDA in February.
BQ.1 and BQ.1.1 have become the dominant strains in the United States after a steady increase in prevalence over the last 2 months, surpassing Omicron’s BA.5 subvariant, which had driven cases earlier in the year.
The subvariants accounted for around 57% of the cases nationally, as per government data last week.
Reuters Health Information © 2022
RSV surge stuns parents and strains providers, but doctors offer help
RSV cases peaked in mid-November, according to the latest Centers for Disease Control and Prevention data, with RSV-associated hospitalizations in the United States among patients 0-4 years having maxed out five times higher than they were at the same time in 2021. These surges strained providers and left parents scrambling for care. Fortunately, pediatric hospitalizations appear to be subsiding.
In interviews, the parents of the child who had a severe case of RSV reflected on their son’s bout with the illness, and doctors described challenges to dealing with the surge in RSV cases this season. The physicians also offered advice on how recognize and respond to future cases of the virus.
Sebastian Witt’s story
“I didn’t even know what RSV was,” said Malte Witt, whose son, Sebastian, 2, was recently hospitalized for RSV in Denver.
Mr. Witt and his wife, Emily Witt, both 32, thought they were dealing with a typical cold until Sebastian’s condition dramatically deteriorated about 36 hours after symptom onset.
“He basically just slumped over and collapsed, coughing uncontrollably,” Mr. Witt said in an interview. “He couldn’t catch his breath.”
The Witts rushed Sebastian to the ED at Children’s Hospital Colorado, expecting to see a doctor immediately. Instead, they spent the night in an overcrowded waiting room alongside many other families in the same situation.
“There was no room for anyone to sit anywhere,” Mr. Witt said. “There were people sitting on the floor. I counted maybe six children hooked up to oxygen when we walked in.”
After waiting approximately 45 minutes, a nurse checked Sebastian’s oxygen saturation. The readings were 79%-83%. This range is significantly below thresholds for supplemental oxygen described by most pediatric guidelines, which range from 90 to 94%.
The nurse connected Sebastian to bottled oxygen in the waiting room, and a recheck 4 hours later showed that his oxygen saturation had improved.
But the improvement didn’t last.
“At roughly hour 10 in the waiting room – it was 4 in the morning – you could tell that Seb was exhausted, really not acting like himself,” Mr. Witt said. “We thought maybe it’s just late at night, he hasn’t really slept. But then Emily noticed that his oxygen tank had run out.”
Mr. Witt told a nurse, and after another check revealed low oxygen saturation, Sebastian was finally admitted.
Early RSV surge strains pediatric providers
With RSV-associated hospitalizations peaking at 48 per 100,000 children, Colorado has been among the states hardest hit by the virus. New Mexico – where hospitalizations peaked at 56.4 per 100,000 children – comes in second. Even in states like California, where hospitalization rates have been almost 10-fold lower than New Mexico, pediatric providers have been stretched to their limits.
“Many hospitals are really being overwhelmed with admissions for RSV, both routine RSV – relatively mild hospitalizations with bronchiolitis – as well as kids in the ICU with more severe cases,” said Dean Blumberg, MD, chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, said in an interview.
Dr. Blumberg believes the severity of the 2022-2023 RSV season is likely COVID related.
“All community-associated respiratory viral infections are out of whack because of the pandemic, and all the masking and social distancing that was occurring,” he said.
This may also explain why older kids are coming down with more severe cases of RSV.
“Some children are getting RSV for the first time as older children,” Dr. Blumberg said, noting that, historically, most children were infected in the first 2 years of life. “There are reports of children 3 or 4 years of age being admitted with their first episode of RSV because of the [COVID] pandemic.”
This year’s RSV season is also notable for arriving early, potentially catching the community off guard, according to Jennifer D. Kusma, MD, a primary care pediatrician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
“People who should have been protected often weren’t protected yet,” Dr. Kusma said in an interview.
Treatments new, old, and unproven
On Nov. 17, in the midst of the RSV surge, the American Academy of Pediatrics issued updated guidance for palivizumab, an RSV-targeting monoclonal antibody labeled for children at risk of severe RSV, including those with pre-existing lung or heart conditions, and infants with a history of premature birth (less than or equal to 35 weeks’ gestational age).
“If RSV disease activity persists at high levels in a given region through the fall and winter, the AAP supports providing more than five consecutive doses of palivizumab to eligible children,” the update stated.
Insurance companies appear to be responding in kind, covering additional doses for children in need.
“[Payers] have agreed that, if [palivizumab] needs to be given for an additional month or 2 or 3, then they’re making a commitment that they’ll reimburse hospitals for providing that,” Dr. Blumberg said.
For ineligible patients, such as Sebastian, who was born prematurely at 36 weeks – 1 week shy of the label requirement – treatment relies upon supportive care with oxygen and IV fluids.
At home, parents are left with simpler options.
Dr. Blumberg and Dr. Kusma recommended keeping children hydrated, maintaining humidified air, and using saline nose drops with bulb suction to clear mucus.
In the Witts’ experience, that last step may be easier said than done.
“Every time a nurse would walk into the room, Sebastian would yell: ‘Go away, doctor! I don’t want snot sucker!’” Mr. Witt said.
“If you over snot-suck, that’s really uncomfortable for the kid, and really hard for you,” Ms. Witt said. “And it doesn’t make much of a difference. It’s just very hard to find a middle ground, where you’re helping and keeping them comfortable.”
Some parents are turning to novel strategies, such as nebulized hypertonic saline, currently marketed on Amazon for children with RSV.
Although the AAP offers a weak recommendation for nebulized hypertonic saline in children hospitalized more than 72 hours, they advise against it in the emergency setting, citing inconsistent findings in clinical trials.
To any parents tempted by thousands of positive Amazon reviews, Dr. Blumberg said, “I wouldn’t waste my money on that.”
Dr. Kusma agreed.
“[Nebulized hypertonic saline] can be irritating,” she said. “It’s saltwater, essentially. If a parent is in the position where they’re worried about their child’s breathing to the point that they think they need to use it, I would err on the side of calling your pediatrician and being seen.”
Going in, coming home
Dr. Kusma said parents should seek medical attention if a child is breathing faster and working harder to get air. Increased work of breathing is characterized by pulling of the skin at the notch where the throat meets the chest bone (tracheal tugging), and flattening of the belly that makes the ribcage more prominent.
Mr. Witt saw these signs in Sebastian. He knew they were significant, because a friend who is a nurse had previously shown him some examples of children who exhibited these symptoms online.
“That’s how I knew that things were actually really dangerous,” Mr. Witt said. “Had she not shown me those videos a month and a half before this happened, I don’t know that we would have hit the alarm bell as quickly as we did.”
After spending their second night and the following day in a cramped preoperative room converted to manage overflow from the emergency department, Sebastian’s condition improved, and he was discharged. The Witts are relieved to be home, but frustrations from their ordeal remain, especially considering the estimated $5,000 in out-of-pocket costs they expect to pay.
“How is this our health care system?” Ms. Witt asked. “This is unbelievable.”
An optimistic outlook
RSV seasons typically demonstrate a clear peak, followed by a decline through the rest of the season, suggesting better times lie ahead; however, this season has been anything but typical.
“I’m hopeful that it will just go away and stay away,” Dr. Kusma said, citing this trend. “But I can’t know for sure.”
To anxious parents, Dr. Blumberg offered an optimistic view of RSV seasons to come.
“There’s hope,” he said. “There are vaccines that are being developed that are very close to FDA approval. So, it’s possible that this time next year, we might have widespread RSV vaccination available for children so that we don’t have to go through this nightmare again.”
Dr. Blumberg and Dr. Kusma disclosed no relevant conflicts of interest.
RSV cases peaked in mid-November, according to the latest Centers for Disease Control and Prevention data, with RSV-associated hospitalizations in the United States among patients 0-4 years having maxed out five times higher than they were at the same time in 2021. These surges strained providers and left parents scrambling for care. Fortunately, pediatric hospitalizations appear to be subsiding.
In interviews, the parents of the child who had a severe case of RSV reflected on their son’s bout with the illness, and doctors described challenges to dealing with the surge in RSV cases this season. The physicians also offered advice on how recognize and respond to future cases of the virus.
Sebastian Witt’s story
“I didn’t even know what RSV was,” said Malte Witt, whose son, Sebastian, 2, was recently hospitalized for RSV in Denver.
Mr. Witt and his wife, Emily Witt, both 32, thought they were dealing with a typical cold until Sebastian’s condition dramatically deteriorated about 36 hours after symptom onset.
“He basically just slumped over and collapsed, coughing uncontrollably,” Mr. Witt said in an interview. “He couldn’t catch his breath.”
The Witts rushed Sebastian to the ED at Children’s Hospital Colorado, expecting to see a doctor immediately. Instead, they spent the night in an overcrowded waiting room alongside many other families in the same situation.
“There was no room for anyone to sit anywhere,” Mr. Witt said. “There were people sitting on the floor. I counted maybe six children hooked up to oxygen when we walked in.”
After waiting approximately 45 minutes, a nurse checked Sebastian’s oxygen saturation. The readings were 79%-83%. This range is significantly below thresholds for supplemental oxygen described by most pediatric guidelines, which range from 90 to 94%.
The nurse connected Sebastian to bottled oxygen in the waiting room, and a recheck 4 hours later showed that his oxygen saturation had improved.
But the improvement didn’t last.
“At roughly hour 10 in the waiting room – it was 4 in the morning – you could tell that Seb was exhausted, really not acting like himself,” Mr. Witt said. “We thought maybe it’s just late at night, he hasn’t really slept. But then Emily noticed that his oxygen tank had run out.”
Mr. Witt told a nurse, and after another check revealed low oxygen saturation, Sebastian was finally admitted.
Early RSV surge strains pediatric providers
With RSV-associated hospitalizations peaking at 48 per 100,000 children, Colorado has been among the states hardest hit by the virus. New Mexico – where hospitalizations peaked at 56.4 per 100,000 children – comes in second. Even in states like California, where hospitalization rates have been almost 10-fold lower than New Mexico, pediatric providers have been stretched to their limits.
“Many hospitals are really being overwhelmed with admissions for RSV, both routine RSV – relatively mild hospitalizations with bronchiolitis – as well as kids in the ICU with more severe cases,” said Dean Blumberg, MD, chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, said in an interview.
Dr. Blumberg believes the severity of the 2022-2023 RSV season is likely COVID related.
“All community-associated respiratory viral infections are out of whack because of the pandemic, and all the masking and social distancing that was occurring,” he said.
This may also explain why older kids are coming down with more severe cases of RSV.
“Some children are getting RSV for the first time as older children,” Dr. Blumberg said, noting that, historically, most children were infected in the first 2 years of life. “There are reports of children 3 or 4 years of age being admitted with their first episode of RSV because of the [COVID] pandemic.”
This year’s RSV season is also notable for arriving early, potentially catching the community off guard, according to Jennifer D. Kusma, MD, a primary care pediatrician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
“People who should have been protected often weren’t protected yet,” Dr. Kusma said in an interview.
Treatments new, old, and unproven
On Nov. 17, in the midst of the RSV surge, the American Academy of Pediatrics issued updated guidance for palivizumab, an RSV-targeting monoclonal antibody labeled for children at risk of severe RSV, including those with pre-existing lung or heart conditions, and infants with a history of premature birth (less than or equal to 35 weeks’ gestational age).
“If RSV disease activity persists at high levels in a given region through the fall and winter, the AAP supports providing more than five consecutive doses of palivizumab to eligible children,” the update stated.
Insurance companies appear to be responding in kind, covering additional doses for children in need.
“[Payers] have agreed that, if [palivizumab] needs to be given for an additional month or 2 or 3, then they’re making a commitment that they’ll reimburse hospitals for providing that,” Dr. Blumberg said.
For ineligible patients, such as Sebastian, who was born prematurely at 36 weeks – 1 week shy of the label requirement – treatment relies upon supportive care with oxygen and IV fluids.
At home, parents are left with simpler options.
Dr. Blumberg and Dr. Kusma recommended keeping children hydrated, maintaining humidified air, and using saline nose drops with bulb suction to clear mucus.
In the Witts’ experience, that last step may be easier said than done.
“Every time a nurse would walk into the room, Sebastian would yell: ‘Go away, doctor! I don’t want snot sucker!’” Mr. Witt said.
“If you over snot-suck, that’s really uncomfortable for the kid, and really hard for you,” Ms. Witt said. “And it doesn’t make much of a difference. It’s just very hard to find a middle ground, where you’re helping and keeping them comfortable.”
Some parents are turning to novel strategies, such as nebulized hypertonic saline, currently marketed on Amazon for children with RSV.
Although the AAP offers a weak recommendation for nebulized hypertonic saline in children hospitalized more than 72 hours, they advise against it in the emergency setting, citing inconsistent findings in clinical trials.
To any parents tempted by thousands of positive Amazon reviews, Dr. Blumberg said, “I wouldn’t waste my money on that.”
Dr. Kusma agreed.
“[Nebulized hypertonic saline] can be irritating,” she said. “It’s saltwater, essentially. If a parent is in the position where they’re worried about their child’s breathing to the point that they think they need to use it, I would err on the side of calling your pediatrician and being seen.”
Going in, coming home
Dr. Kusma said parents should seek medical attention if a child is breathing faster and working harder to get air. Increased work of breathing is characterized by pulling of the skin at the notch where the throat meets the chest bone (tracheal tugging), and flattening of the belly that makes the ribcage more prominent.
Mr. Witt saw these signs in Sebastian. He knew they were significant, because a friend who is a nurse had previously shown him some examples of children who exhibited these symptoms online.
“That’s how I knew that things were actually really dangerous,” Mr. Witt said. “Had she not shown me those videos a month and a half before this happened, I don’t know that we would have hit the alarm bell as quickly as we did.”
After spending their second night and the following day in a cramped preoperative room converted to manage overflow from the emergency department, Sebastian’s condition improved, and he was discharged. The Witts are relieved to be home, but frustrations from their ordeal remain, especially considering the estimated $5,000 in out-of-pocket costs they expect to pay.
“How is this our health care system?” Ms. Witt asked. “This is unbelievable.”
An optimistic outlook
RSV seasons typically demonstrate a clear peak, followed by a decline through the rest of the season, suggesting better times lie ahead; however, this season has been anything but typical.
“I’m hopeful that it will just go away and stay away,” Dr. Kusma said, citing this trend. “But I can’t know for sure.”
To anxious parents, Dr. Blumberg offered an optimistic view of RSV seasons to come.
“There’s hope,” he said. “There are vaccines that are being developed that are very close to FDA approval. So, it’s possible that this time next year, we might have widespread RSV vaccination available for children so that we don’t have to go through this nightmare again.”
Dr. Blumberg and Dr. Kusma disclosed no relevant conflicts of interest.
RSV cases peaked in mid-November, according to the latest Centers for Disease Control and Prevention data, with RSV-associated hospitalizations in the United States among patients 0-4 years having maxed out five times higher than they were at the same time in 2021. These surges strained providers and left parents scrambling for care. Fortunately, pediatric hospitalizations appear to be subsiding.
In interviews, the parents of the child who had a severe case of RSV reflected on their son’s bout with the illness, and doctors described challenges to dealing with the surge in RSV cases this season. The physicians also offered advice on how recognize and respond to future cases of the virus.
Sebastian Witt’s story
“I didn’t even know what RSV was,” said Malte Witt, whose son, Sebastian, 2, was recently hospitalized for RSV in Denver.
Mr. Witt and his wife, Emily Witt, both 32, thought they were dealing with a typical cold until Sebastian’s condition dramatically deteriorated about 36 hours after symptom onset.
“He basically just slumped over and collapsed, coughing uncontrollably,” Mr. Witt said in an interview. “He couldn’t catch his breath.”
The Witts rushed Sebastian to the ED at Children’s Hospital Colorado, expecting to see a doctor immediately. Instead, they spent the night in an overcrowded waiting room alongside many other families in the same situation.
“There was no room for anyone to sit anywhere,” Mr. Witt said. “There were people sitting on the floor. I counted maybe six children hooked up to oxygen when we walked in.”
After waiting approximately 45 minutes, a nurse checked Sebastian’s oxygen saturation. The readings were 79%-83%. This range is significantly below thresholds for supplemental oxygen described by most pediatric guidelines, which range from 90 to 94%.
The nurse connected Sebastian to bottled oxygen in the waiting room, and a recheck 4 hours later showed that his oxygen saturation had improved.
But the improvement didn’t last.
“At roughly hour 10 in the waiting room – it was 4 in the morning – you could tell that Seb was exhausted, really not acting like himself,” Mr. Witt said. “We thought maybe it’s just late at night, he hasn’t really slept. But then Emily noticed that his oxygen tank had run out.”
Mr. Witt told a nurse, and after another check revealed low oxygen saturation, Sebastian was finally admitted.
Early RSV surge strains pediatric providers
With RSV-associated hospitalizations peaking at 48 per 100,000 children, Colorado has been among the states hardest hit by the virus. New Mexico – where hospitalizations peaked at 56.4 per 100,000 children – comes in second. Even in states like California, where hospitalization rates have been almost 10-fold lower than New Mexico, pediatric providers have been stretched to their limits.
“Many hospitals are really being overwhelmed with admissions for RSV, both routine RSV – relatively mild hospitalizations with bronchiolitis – as well as kids in the ICU with more severe cases,” said Dean Blumberg, MD, chief of the division of pediatric infectious diseases at UC Davis Health, Sacramento, said in an interview.
Dr. Blumberg believes the severity of the 2022-2023 RSV season is likely COVID related.
“All community-associated respiratory viral infections are out of whack because of the pandemic, and all the masking and social distancing that was occurring,” he said.
This may also explain why older kids are coming down with more severe cases of RSV.
“Some children are getting RSV for the first time as older children,” Dr. Blumberg said, noting that, historically, most children were infected in the first 2 years of life. “There are reports of children 3 or 4 years of age being admitted with their first episode of RSV because of the [COVID] pandemic.”
This year’s RSV season is also notable for arriving early, potentially catching the community off guard, according to Jennifer D. Kusma, MD, a primary care pediatrician at Ann & Robert H. Lurie Children’s Hospital of Chicago.
“People who should have been protected often weren’t protected yet,” Dr. Kusma said in an interview.
Treatments new, old, and unproven
On Nov. 17, in the midst of the RSV surge, the American Academy of Pediatrics issued updated guidance for palivizumab, an RSV-targeting monoclonal antibody labeled for children at risk of severe RSV, including those with pre-existing lung or heart conditions, and infants with a history of premature birth (less than or equal to 35 weeks’ gestational age).
“If RSV disease activity persists at high levels in a given region through the fall and winter, the AAP supports providing more than five consecutive doses of palivizumab to eligible children,” the update stated.
Insurance companies appear to be responding in kind, covering additional doses for children in need.
“[Payers] have agreed that, if [palivizumab] needs to be given for an additional month or 2 or 3, then they’re making a commitment that they’ll reimburse hospitals for providing that,” Dr. Blumberg said.
For ineligible patients, such as Sebastian, who was born prematurely at 36 weeks – 1 week shy of the label requirement – treatment relies upon supportive care with oxygen and IV fluids.
At home, parents are left with simpler options.
Dr. Blumberg and Dr. Kusma recommended keeping children hydrated, maintaining humidified air, and using saline nose drops with bulb suction to clear mucus.
In the Witts’ experience, that last step may be easier said than done.
“Every time a nurse would walk into the room, Sebastian would yell: ‘Go away, doctor! I don’t want snot sucker!’” Mr. Witt said.
“If you over snot-suck, that’s really uncomfortable for the kid, and really hard for you,” Ms. Witt said. “And it doesn’t make much of a difference. It’s just very hard to find a middle ground, where you’re helping and keeping them comfortable.”
Some parents are turning to novel strategies, such as nebulized hypertonic saline, currently marketed on Amazon for children with RSV.
Although the AAP offers a weak recommendation for nebulized hypertonic saline in children hospitalized more than 72 hours, they advise against it in the emergency setting, citing inconsistent findings in clinical trials.
To any parents tempted by thousands of positive Amazon reviews, Dr. Blumberg said, “I wouldn’t waste my money on that.”
Dr. Kusma agreed.
“[Nebulized hypertonic saline] can be irritating,” she said. “It’s saltwater, essentially. If a parent is in the position where they’re worried about their child’s breathing to the point that they think they need to use it, I would err on the side of calling your pediatrician and being seen.”
Going in, coming home
Dr. Kusma said parents should seek medical attention if a child is breathing faster and working harder to get air. Increased work of breathing is characterized by pulling of the skin at the notch where the throat meets the chest bone (tracheal tugging), and flattening of the belly that makes the ribcage more prominent.
Mr. Witt saw these signs in Sebastian. He knew they were significant, because a friend who is a nurse had previously shown him some examples of children who exhibited these symptoms online.
“That’s how I knew that things were actually really dangerous,” Mr. Witt said. “Had she not shown me those videos a month and a half before this happened, I don’t know that we would have hit the alarm bell as quickly as we did.”
After spending their second night and the following day in a cramped preoperative room converted to manage overflow from the emergency department, Sebastian’s condition improved, and he was discharged. The Witts are relieved to be home, but frustrations from their ordeal remain, especially considering the estimated $5,000 in out-of-pocket costs they expect to pay.
“How is this our health care system?” Ms. Witt asked. “This is unbelievable.”
An optimistic outlook
RSV seasons typically demonstrate a clear peak, followed by a decline through the rest of the season, suggesting better times lie ahead; however, this season has been anything but typical.
“I’m hopeful that it will just go away and stay away,” Dr. Kusma said, citing this trend. “But I can’t know for sure.”
To anxious parents, Dr. Blumberg offered an optimistic view of RSV seasons to come.
“There’s hope,” he said. “There are vaccines that are being developed that are very close to FDA approval. So, it’s possible that this time next year, we might have widespread RSV vaccination available for children so that we don’t have to go through this nightmare again.”
Dr. Blumberg and Dr. Kusma disclosed no relevant conflicts of interest.