User login
Official news magazine of the Society of Hospital Medicine
Copyright by Society of Hospital Medicine or related companies. All rights reserved. ISSN 1553-085X
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
div[contains(@class, 'pane-pub-article-hospitalist')]


C. difficile guidelines offer new possibilities
The American College of Gastroenterology has issued new guidelines on management of Clostridioides difficile infection that now include roles for fecal microbial transplant (FMT), combination testing, and bezlotoxumab.
The ACG’s previous guidelines on the diagnosis, management, and treatment of what was then still called Clostridium difficile were published in 2013. Since then, the organism’s name changed to Clostridioides difficile, and that’s just the beginning of the changes reflected in the scientific literature, wrote lead author Colleen R. Kelly, MD, of Brown University, Providence, R.I., and colleagues.
“Other developments include the increased recognition of diagnostic challenges in the era of nucleic acid amplification–based testing, new therapeutic options for treatment and prevention of recurrence, and increasing evidence to support fecal microbiota transplantation (FMT) in recurrent and severe infection,” the authors said.
The guidelines, published in the American Journal of Gastroenterology, include 23 graded recommendations addressing issues of prevention, diagnosis, treatment, prevention of recurrence, and guidance for special populations in the management of C. difficile infection (CDI).
New faces among familiar ones
In terms of diagnosis, the new guidelines recommend using both a highly sensitive testing modality and a highly specific one to help distinguish colonization from active infection. Specifically, the authors recommend that stool is first tested using a highly sensitive test, either nucleic acid amplification testing or glutamate dehydrogenase, followed by an enzyme immunoassays for its high specificity.
Changes to treatment recommendations include the initial use of oral vancomycin or oral fidaxomicin for cases of nonsevere CDI. Oral metronidazole may be considered for initial nonsevere CDI in low-risk patients, the authors noted. The evidence is strong for the continued recommendations of vancomycin (125 mg four times daily for 10 days) and fidaxomicin (200 mg twice daily for 10 days) for patients with severe CDI. For patients with fulminant CDI, the recommendations call for medical therapy including volume resuscitation and oral vancomycin, although combination therapy with parenteral metronidazole may be considered despite the very low quality of evidence.
A notable update to the guidelines is the recommendation of fecal microbiota transplant (FMT) for both severe and fulminant CDI cases that are resistant to antibiotics and to prevent recurrence in at-risk patients. Although the quality of evidence is ranked as low, the recommendation is strong, the authors wrote. “Beyond improved cure rates, FMT may result in decreased rates of CDI-related colectomy and sepsis and may offer survival benefit in this critically ill patient population.” However, most patients in studies of FMT required multiple treatments in combination with anti-CDI antibiotics.
Other recommendations to prevent recurrence include oral vancomycin prophylaxis during the subsequent use of systemic antibiotics in patients with a history of CDI. The guidelines also recommend bezlotoxumab for prevention of CDI recurrence in high-risk patients, and advise against discontinuing antisecretory therapy in CDI patients if there is an appropriate indication for use.
Based on the lack of quality evidence, the guidelines recommend against the use of probiotics for preventing CDI in patients being treated with antibiotics and for prevention of recurrent infection.
Special populations
For patients with inflammatory bowel disease, the guidelines recommend C. difficile testing when these individuals present with acute flares and diarrhea, and the use of vancomycin for treatment. In addition, the authors strongly recommended FMT for recurrent CDI in these patients. For pregnant, postpartum, and breastfeeding patients with CDI, the guidelines recommend vancomycin, and either vancomycin or fidaxomicin may be used for treating CDI in immunocompromised patients, the authors noted.
The updated guidelines are designed to complement those issued by the Infections Disease Society of America and Society of Healthcare Epidemiologists of America, the researchers noted.
Reflecting the research
The previous guidelines for C. difficile were issued in 2013, and much has changed since then in terms of epidemiology, diagnosis, treatment, and infection control, Sahil Khanna, MBBS, MS, of the Mayo Clinic, Rochester, Minn., said in an interview.
Notably, diagnostic testing has “made leaps and bounds” and new treatments have become available that were not included in earlier guidelines, said Dr. Khanna. In particular, the new guidelines are recommending a two-step diagnostic assay; “the diagnostic algorithm has changed, and hopefully that will help us change practice” to identify active infection more quickly and efficiently.
Another important update is the recommendation of fidaxomicin as an option for initial nonfulminant CDI as an alternative to vancomycin, Dr. Khanna said, noting that metronidazole remains an option for low-risk patients. An additional change is the advice to use a different treatment for a second recurrent infection rather than repeating the initial treatment.
The recommendation of bezlotoxumab for prevention of CDI recurrence in patients who are at high risk of recurrence is the first time this drug has appeared in major guidelines, Dr. Khanna observed.
The recommendation in support of fecal microbiota transplant is a key update to the management of CDI, including the guidance that the procedure can be repeated if necessary, he said.
Looking ahead, “Additional research is needed to fully understand the best testing algorithms for CDI,” Dr. Khanna explained. “More studies also are needed to show how FMT fully fits into the picture, and some current studies are looking at its potential earlier in the course of infection.”
The guidelines were developed in collaboration with the Practice Parameters Committee of the American College of Gastroenterology and received no outside funding. Dr. Kelly disclosed serving as a site investigator of a clinical trial for Finch Therapeutics and is an unpaid clinical advisory board member for OpenBiome. Dr. Khanna has coauthored previous guidelines on C. difficile. He disclosed consulting relationships with Finch, GlaxoSmithKline, Jetson, ProbioTech, and Shire/Takeda, as well as research support from Rebiotix, Seres, and Vedanta.
The American College of Gastroenterology has issued new guidelines on management of Clostridioides difficile infection that now include roles for fecal microbial transplant (FMT), combination testing, and bezlotoxumab.
The ACG’s previous guidelines on the diagnosis, management, and treatment of what was then still called Clostridium difficile were published in 2013. Since then, the organism’s name changed to Clostridioides difficile, and that’s just the beginning of the changes reflected in the scientific literature, wrote lead author Colleen R. Kelly, MD, of Brown University, Providence, R.I., and colleagues.
“Other developments include the increased recognition of diagnostic challenges in the era of nucleic acid amplification–based testing, new therapeutic options for treatment and prevention of recurrence, and increasing evidence to support fecal microbiota transplantation (FMT) in recurrent and severe infection,” the authors said.
The guidelines, published in the American Journal of Gastroenterology, include 23 graded recommendations addressing issues of prevention, diagnosis, treatment, prevention of recurrence, and guidance for special populations in the management of C. difficile infection (CDI).
New faces among familiar ones
In terms of diagnosis, the new guidelines recommend using both a highly sensitive testing modality and a highly specific one to help distinguish colonization from active infection. Specifically, the authors recommend that stool is first tested using a highly sensitive test, either nucleic acid amplification testing or glutamate dehydrogenase, followed by an enzyme immunoassays for its high specificity.
Changes to treatment recommendations include the initial use of oral vancomycin or oral fidaxomicin for cases of nonsevere CDI. Oral metronidazole may be considered for initial nonsevere CDI in low-risk patients, the authors noted. The evidence is strong for the continued recommendations of vancomycin (125 mg four times daily for 10 days) and fidaxomicin (200 mg twice daily for 10 days) for patients with severe CDI. For patients with fulminant CDI, the recommendations call for medical therapy including volume resuscitation and oral vancomycin, although combination therapy with parenteral metronidazole may be considered despite the very low quality of evidence.
A notable update to the guidelines is the recommendation of fecal microbiota transplant (FMT) for both severe and fulminant CDI cases that are resistant to antibiotics and to prevent recurrence in at-risk patients. Although the quality of evidence is ranked as low, the recommendation is strong, the authors wrote. “Beyond improved cure rates, FMT may result in decreased rates of CDI-related colectomy and sepsis and may offer survival benefit in this critically ill patient population.” However, most patients in studies of FMT required multiple treatments in combination with anti-CDI antibiotics.
Other recommendations to prevent recurrence include oral vancomycin prophylaxis during the subsequent use of systemic antibiotics in patients with a history of CDI. The guidelines also recommend bezlotoxumab for prevention of CDI recurrence in high-risk patients, and advise against discontinuing antisecretory therapy in CDI patients if there is an appropriate indication for use.
Based on the lack of quality evidence, the guidelines recommend against the use of probiotics for preventing CDI in patients being treated with antibiotics and for prevention of recurrent infection.
Special populations
For patients with inflammatory bowel disease, the guidelines recommend C. difficile testing when these individuals present with acute flares and diarrhea, and the use of vancomycin for treatment. In addition, the authors strongly recommended FMT for recurrent CDI in these patients. For pregnant, postpartum, and breastfeeding patients with CDI, the guidelines recommend vancomycin, and either vancomycin or fidaxomicin may be used for treating CDI in immunocompromised patients, the authors noted.
The updated guidelines are designed to complement those issued by the Infections Disease Society of America and Society of Healthcare Epidemiologists of America, the researchers noted.
Reflecting the research
The previous guidelines for C. difficile were issued in 2013, and much has changed since then in terms of epidemiology, diagnosis, treatment, and infection control, Sahil Khanna, MBBS, MS, of the Mayo Clinic, Rochester, Minn., said in an interview.
Notably, diagnostic testing has “made leaps and bounds” and new treatments have become available that were not included in earlier guidelines, said Dr. Khanna. In particular, the new guidelines are recommending a two-step diagnostic assay; “the diagnostic algorithm has changed, and hopefully that will help us change practice” to identify active infection more quickly and efficiently.
Another important update is the recommendation of fidaxomicin as an option for initial nonfulminant CDI as an alternative to vancomycin, Dr. Khanna said, noting that metronidazole remains an option for low-risk patients. An additional change is the advice to use a different treatment for a second recurrent infection rather than repeating the initial treatment.
The recommendation of bezlotoxumab for prevention of CDI recurrence in patients who are at high risk of recurrence is the first time this drug has appeared in major guidelines, Dr. Khanna observed.
The recommendation in support of fecal microbiota transplant is a key update to the management of CDI, including the guidance that the procedure can be repeated if necessary, he said.
Looking ahead, “Additional research is needed to fully understand the best testing algorithms for CDI,” Dr. Khanna explained. “More studies also are needed to show how FMT fully fits into the picture, and some current studies are looking at its potential earlier in the course of infection.”
The guidelines were developed in collaboration with the Practice Parameters Committee of the American College of Gastroenterology and received no outside funding. Dr. Kelly disclosed serving as a site investigator of a clinical trial for Finch Therapeutics and is an unpaid clinical advisory board member for OpenBiome. Dr. Khanna has coauthored previous guidelines on C. difficile. He disclosed consulting relationships with Finch, GlaxoSmithKline, Jetson, ProbioTech, and Shire/Takeda, as well as research support from Rebiotix, Seres, and Vedanta.
The American College of Gastroenterology has issued new guidelines on management of Clostridioides difficile infection that now include roles for fecal microbial transplant (FMT), combination testing, and bezlotoxumab.
The ACG’s previous guidelines on the diagnosis, management, and treatment of what was then still called Clostridium difficile were published in 2013. Since then, the organism’s name changed to Clostridioides difficile, and that’s just the beginning of the changes reflected in the scientific literature, wrote lead author Colleen R. Kelly, MD, of Brown University, Providence, R.I., and colleagues.
“Other developments include the increased recognition of diagnostic challenges in the era of nucleic acid amplification–based testing, new therapeutic options for treatment and prevention of recurrence, and increasing evidence to support fecal microbiota transplantation (FMT) in recurrent and severe infection,” the authors said.
The guidelines, published in the American Journal of Gastroenterology, include 23 graded recommendations addressing issues of prevention, diagnosis, treatment, prevention of recurrence, and guidance for special populations in the management of C. difficile infection (CDI).
New faces among familiar ones
In terms of diagnosis, the new guidelines recommend using both a highly sensitive testing modality and a highly specific one to help distinguish colonization from active infection. Specifically, the authors recommend that stool is first tested using a highly sensitive test, either nucleic acid amplification testing or glutamate dehydrogenase, followed by an enzyme immunoassays for its high specificity.
Changes to treatment recommendations include the initial use of oral vancomycin or oral fidaxomicin for cases of nonsevere CDI. Oral metronidazole may be considered for initial nonsevere CDI in low-risk patients, the authors noted. The evidence is strong for the continued recommendations of vancomycin (125 mg four times daily for 10 days) and fidaxomicin (200 mg twice daily for 10 days) for patients with severe CDI. For patients with fulminant CDI, the recommendations call for medical therapy including volume resuscitation and oral vancomycin, although combination therapy with parenteral metronidazole may be considered despite the very low quality of evidence.
A notable update to the guidelines is the recommendation of fecal microbiota transplant (FMT) for both severe and fulminant CDI cases that are resistant to antibiotics and to prevent recurrence in at-risk patients. Although the quality of evidence is ranked as low, the recommendation is strong, the authors wrote. “Beyond improved cure rates, FMT may result in decreased rates of CDI-related colectomy and sepsis and may offer survival benefit in this critically ill patient population.” However, most patients in studies of FMT required multiple treatments in combination with anti-CDI antibiotics.
Other recommendations to prevent recurrence include oral vancomycin prophylaxis during the subsequent use of systemic antibiotics in patients with a history of CDI. The guidelines also recommend bezlotoxumab for prevention of CDI recurrence in high-risk patients, and advise against discontinuing antisecretory therapy in CDI patients if there is an appropriate indication for use.
Based on the lack of quality evidence, the guidelines recommend against the use of probiotics for preventing CDI in patients being treated with antibiotics and for prevention of recurrent infection.
Special populations
For patients with inflammatory bowel disease, the guidelines recommend C. difficile testing when these individuals present with acute flares and diarrhea, and the use of vancomycin for treatment. In addition, the authors strongly recommended FMT for recurrent CDI in these patients. For pregnant, postpartum, and breastfeeding patients with CDI, the guidelines recommend vancomycin, and either vancomycin or fidaxomicin may be used for treating CDI in immunocompromised patients, the authors noted.
The updated guidelines are designed to complement those issued by the Infections Disease Society of America and Society of Healthcare Epidemiologists of America, the researchers noted.
Reflecting the research
The previous guidelines for C. difficile were issued in 2013, and much has changed since then in terms of epidemiology, diagnosis, treatment, and infection control, Sahil Khanna, MBBS, MS, of the Mayo Clinic, Rochester, Minn., said in an interview.
Notably, diagnostic testing has “made leaps and bounds” and new treatments have become available that were not included in earlier guidelines, said Dr. Khanna. In particular, the new guidelines are recommending a two-step diagnostic assay; “the diagnostic algorithm has changed, and hopefully that will help us change practice” to identify active infection more quickly and efficiently.
Another important update is the recommendation of fidaxomicin as an option for initial nonfulminant CDI as an alternative to vancomycin, Dr. Khanna said, noting that metronidazole remains an option for low-risk patients. An additional change is the advice to use a different treatment for a second recurrent infection rather than repeating the initial treatment.
The recommendation of bezlotoxumab for prevention of CDI recurrence in patients who are at high risk of recurrence is the first time this drug has appeared in major guidelines, Dr. Khanna observed.
The recommendation in support of fecal microbiota transplant is a key update to the management of CDI, including the guidance that the procedure can be repeated if necessary, he said.
Looking ahead, “Additional research is needed to fully understand the best testing algorithms for CDI,” Dr. Khanna explained. “More studies also are needed to show how FMT fully fits into the picture, and some current studies are looking at its potential earlier in the course of infection.”
The guidelines were developed in collaboration with the Practice Parameters Committee of the American College of Gastroenterology and received no outside funding. Dr. Kelly disclosed serving as a site investigator of a clinical trial for Finch Therapeutics and is an unpaid clinical advisory board member for OpenBiome. Dr. Khanna has coauthored previous guidelines on C. difficile. He disclosed consulting relationships with Finch, GlaxoSmithKline, Jetson, ProbioTech, and Shire/Takeda, as well as research support from Rebiotix, Seres, and Vedanta.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
FIND: A framework for success as a first-year hospitalist
Congratulations! You’re about to start your first year as a hospitalist, and in many cases your first real job. Hospital medicine is an incredibly rewarding subspecialty, but the progression from resident to attending physician can be daunting. To facilitate this transition, we present FIND (Familiarity, Identity, Network, and Direction) – a novel, sequential framework for success as a first-year hospitalist. For each component, we provide a narrative overview and a summary bullet point for quick reference.
Familiarity
- Lay the foundation: Learn the ins and outs of your job, EMR, and team.
Familiarize yourself with your surroundings. Know where your patients are located, where you can document, where to find equipment for procedures, and how to reach information technology. Proactively set up the electronic medical record on your home computer and phone. Make sure to review your responsibilities, including your call schedule, your shifts, your assigned patient panel, when you can leave campus, and how people should contact you. Also, others should know your expectations of them, especially if you are working with trainees.
Maintain a file with all of your orientation materials, including phone numbers and emails of key personnel. Know who your people are – who can access your calendar, who you can call with a clinical question or to escalate care, who can assist you with billing, and who helps with the throughput of your patients in the hospital. Take time to review your benefits, including parental leave, insurance coverage, retirement planning, vacation time, and ancillary services like laundry for your white coat. Familiarizing yourself with these basics will provide comfort and lay the foundation for your first year.
Identity
- Perform self-reflection: Overcome imposter syndrome and invest in hobbies.
One of the fundamental realizations that will occur with your first hospitalist job is that you are the attending. You walk in with a vision of your first job; be prepared to be surprised. You have earned the privilege of deciding on patient plans, and you are no longer obligated to staff with a senior physician. This is both empowering and terrifying. In a way, it may oddly remind you of intern year. A new hospital, new EMR, new colleagues, and imposter syndrome will trick you into doubting your decisions.
How to battle it? Positive thinking. You do know the basics of inpatient medicine and you do have a support system to cheer you on. As part of imposter syndrome, you may feel pressured to focus solely on work. Yet, your first job as a hospitalist is finally an amazing opportunity to focus on you. What hobbies have you been neglecting: cooking, photography, reading, more time with family, a new pet? You have the power to schedule your off-weeks. Are you interested in academics? Reserve a portion of your time off to explore scholarship opportunities at your institution. Your first job as a hospitalist is a chance to develop your identity, both as a physician and as an individual.
Network
- Engage your support system: Communicate with nursing, administration, colleagues.
Networking, or building a web of mutually beneficial professional relationships, is imperative for long-term career success. Hospitalists should focus on developing their network across multiple departments, such as nursing, subspecialties, medical education, and hospital administration. Curating a broad network will increase your visibility within your organization, showcase your unique services, and demonstrate your value.
To make networking encounters impactful, express interest, actively listen, ask relevant questions, and seek areas of mutual benefit. It’s equally important to cultivate these new relationships after the initial encounter and to demonstrate how your skill set will aid colleagues in achieving their professional goals. Over time, as you establish your niche, deliberate networking with those who share similar interests can lead to a wealth of new experiences and opportunities. Intentionally mastering networking early in your career provides insight into different aspects of the hospital system, new perspectives on ideas, and access to valuable guidance from other professionals. Engaging in networking to establish your support system is an essential step towards success as a first-year hospitalist.
Direction
- Visualize your path: Find a mentor and develop a mission statement and career plan.
Once you’re familiar with your work environment, confident in your identity, and acquainted with your support network, you’re ready for the final step – direction. Hospital medicine offers many professional avenues and clarifying your career path is challenging when attempted alone. A mentor is the necessary catalyst to find direction and purpose.
Selecting and engaging with a mentor will bolster your professional advancement, academic productivity, and most importantly, career satisfaction.1 At its best, mentorship is a symbiotic relationship. Your mentor should inspire you, challenge you, and support your growth and emotional well-being. In turn, as the mentee, you should be proactive, establish expectations, and take responsibility for maintaining communication to ensure a successful relationship. As your career takes shape over time, you may require a mentorship team to fulfill your unique needs.
When you’ve established a relationship with your mentor, take time to develop 1-year and 5-year plans. Your 1-year plan should focus on a few “quick wins,” often projects or opportunities at your home institution. Small victories in your first year will boost your confidence, motivation, and sense of control. Your 5-year plan should delineate the steps necessary to make your first major career transition, such as from instructor to assistant professor. Working with your mentor to draft a career mission statement is a useful first step in this process. Beginning with the end in mind, will help you visualize your direction.2
We hope that the FIND framework will help you find your path to success as a first-year hospitalist.
Dr. Nelson is a hospitalist and instructor of medicine at Harvard Medical School and Beth Israel Deaconess Medical Center, both in Boston. Dr. Ashford is assistant professor and program director, department of internal medicine/pediatrics, at the University of Nebraska Medical Center, Omaha. Dr. Rawal is clinical assistant professor of medicine at the University of Pittsburgh Medical Center. Dr. Crecelius is assistant professor of clinical medicine at Indiana University, Indianapolis. This article is sponsored by the SHM Physicians in Training committee, which submits quarterly content to the Hospitalist on topics relevant to trainees and early -career hospitalists.
References
1. Zerzan JT et al. Making the most of mentors: a guide for mentees. Acad Med. 2009;84:140-4. doi: 10.1097/ACM.0b013e3181906e8f.
2. Covey F. The seven habits of highly effective people. 25th anniversary edition. New York: Simon and Schuster, 2013.
Congratulations! You’re about to start your first year as a hospitalist, and in many cases your first real job. Hospital medicine is an incredibly rewarding subspecialty, but the progression from resident to attending physician can be daunting. To facilitate this transition, we present FIND (Familiarity, Identity, Network, and Direction) – a novel, sequential framework for success as a first-year hospitalist. For each component, we provide a narrative overview and a summary bullet point for quick reference.
Familiarity
- Lay the foundation: Learn the ins and outs of your job, EMR, and team.
Familiarize yourself with your surroundings. Know where your patients are located, where you can document, where to find equipment for procedures, and how to reach information technology. Proactively set up the electronic medical record on your home computer and phone. Make sure to review your responsibilities, including your call schedule, your shifts, your assigned patient panel, when you can leave campus, and how people should contact you. Also, others should know your expectations of them, especially if you are working with trainees.
Maintain a file with all of your orientation materials, including phone numbers and emails of key personnel. Know who your people are – who can access your calendar, who you can call with a clinical question or to escalate care, who can assist you with billing, and who helps with the throughput of your patients in the hospital. Take time to review your benefits, including parental leave, insurance coverage, retirement planning, vacation time, and ancillary services like laundry for your white coat. Familiarizing yourself with these basics will provide comfort and lay the foundation for your first year.
Identity
- Perform self-reflection: Overcome imposter syndrome and invest in hobbies.
One of the fundamental realizations that will occur with your first hospitalist job is that you are the attending. You walk in with a vision of your first job; be prepared to be surprised. You have earned the privilege of deciding on patient plans, and you are no longer obligated to staff with a senior physician. This is both empowering and terrifying. In a way, it may oddly remind you of intern year. A new hospital, new EMR, new colleagues, and imposter syndrome will trick you into doubting your decisions.
How to battle it? Positive thinking. You do know the basics of inpatient medicine and you do have a support system to cheer you on. As part of imposter syndrome, you may feel pressured to focus solely on work. Yet, your first job as a hospitalist is finally an amazing opportunity to focus on you. What hobbies have you been neglecting: cooking, photography, reading, more time with family, a new pet? You have the power to schedule your off-weeks. Are you interested in academics? Reserve a portion of your time off to explore scholarship opportunities at your institution. Your first job as a hospitalist is a chance to develop your identity, both as a physician and as an individual.
Network
- Engage your support system: Communicate with nursing, administration, colleagues.
Networking, or building a web of mutually beneficial professional relationships, is imperative for long-term career success. Hospitalists should focus on developing their network across multiple departments, such as nursing, subspecialties, medical education, and hospital administration. Curating a broad network will increase your visibility within your organization, showcase your unique services, and demonstrate your value.
To make networking encounters impactful, express interest, actively listen, ask relevant questions, and seek areas of mutual benefit. It’s equally important to cultivate these new relationships after the initial encounter and to demonstrate how your skill set will aid colleagues in achieving their professional goals. Over time, as you establish your niche, deliberate networking with those who share similar interests can lead to a wealth of new experiences and opportunities. Intentionally mastering networking early in your career provides insight into different aspects of the hospital system, new perspectives on ideas, and access to valuable guidance from other professionals. Engaging in networking to establish your support system is an essential step towards success as a first-year hospitalist.
Direction
- Visualize your path: Find a mentor and develop a mission statement and career plan.
Once you’re familiar with your work environment, confident in your identity, and acquainted with your support network, you’re ready for the final step – direction. Hospital medicine offers many professional avenues and clarifying your career path is challenging when attempted alone. A mentor is the necessary catalyst to find direction and purpose.
Selecting and engaging with a mentor will bolster your professional advancement, academic productivity, and most importantly, career satisfaction.1 At its best, mentorship is a symbiotic relationship. Your mentor should inspire you, challenge you, and support your growth and emotional well-being. In turn, as the mentee, you should be proactive, establish expectations, and take responsibility for maintaining communication to ensure a successful relationship. As your career takes shape over time, you may require a mentorship team to fulfill your unique needs.
When you’ve established a relationship with your mentor, take time to develop 1-year and 5-year plans. Your 1-year plan should focus on a few “quick wins,” often projects or opportunities at your home institution. Small victories in your first year will boost your confidence, motivation, and sense of control. Your 5-year plan should delineate the steps necessary to make your first major career transition, such as from instructor to assistant professor. Working with your mentor to draft a career mission statement is a useful first step in this process. Beginning with the end in mind, will help you visualize your direction.2
We hope that the FIND framework will help you find your path to success as a first-year hospitalist.
Dr. Nelson is a hospitalist and instructor of medicine at Harvard Medical School and Beth Israel Deaconess Medical Center, both in Boston. Dr. Ashford is assistant professor and program director, department of internal medicine/pediatrics, at the University of Nebraska Medical Center, Omaha. Dr. Rawal is clinical assistant professor of medicine at the University of Pittsburgh Medical Center. Dr. Crecelius is assistant professor of clinical medicine at Indiana University, Indianapolis. This article is sponsored by the SHM Physicians in Training committee, which submits quarterly content to the Hospitalist on topics relevant to trainees and early -career hospitalists.
References
1. Zerzan JT et al. Making the most of mentors: a guide for mentees. Acad Med. 2009;84:140-4. doi: 10.1097/ACM.0b013e3181906e8f.
2. Covey F. The seven habits of highly effective people. 25th anniversary edition. New York: Simon and Schuster, 2013.
Congratulations! You’re about to start your first year as a hospitalist, and in many cases your first real job. Hospital medicine is an incredibly rewarding subspecialty, but the progression from resident to attending physician can be daunting. To facilitate this transition, we present FIND (Familiarity, Identity, Network, and Direction) – a novel, sequential framework for success as a first-year hospitalist. For each component, we provide a narrative overview and a summary bullet point for quick reference.
Familiarity
- Lay the foundation: Learn the ins and outs of your job, EMR, and team.
Familiarize yourself with your surroundings. Know where your patients are located, where you can document, where to find equipment for procedures, and how to reach information technology. Proactively set up the electronic medical record on your home computer and phone. Make sure to review your responsibilities, including your call schedule, your shifts, your assigned patient panel, when you can leave campus, and how people should contact you. Also, others should know your expectations of them, especially if you are working with trainees.
Maintain a file with all of your orientation materials, including phone numbers and emails of key personnel. Know who your people are – who can access your calendar, who you can call with a clinical question or to escalate care, who can assist you with billing, and who helps with the throughput of your patients in the hospital. Take time to review your benefits, including parental leave, insurance coverage, retirement planning, vacation time, and ancillary services like laundry for your white coat. Familiarizing yourself with these basics will provide comfort and lay the foundation for your first year.
Identity
- Perform self-reflection: Overcome imposter syndrome and invest in hobbies.
One of the fundamental realizations that will occur with your first hospitalist job is that you are the attending. You walk in with a vision of your first job; be prepared to be surprised. You have earned the privilege of deciding on patient plans, and you are no longer obligated to staff with a senior physician. This is both empowering and terrifying. In a way, it may oddly remind you of intern year. A new hospital, new EMR, new colleagues, and imposter syndrome will trick you into doubting your decisions.
How to battle it? Positive thinking. You do know the basics of inpatient medicine and you do have a support system to cheer you on. As part of imposter syndrome, you may feel pressured to focus solely on work. Yet, your first job as a hospitalist is finally an amazing opportunity to focus on you. What hobbies have you been neglecting: cooking, photography, reading, more time with family, a new pet? You have the power to schedule your off-weeks. Are you interested in academics? Reserve a portion of your time off to explore scholarship opportunities at your institution. Your first job as a hospitalist is a chance to develop your identity, both as a physician and as an individual.
Network
- Engage your support system: Communicate with nursing, administration, colleagues.
Networking, or building a web of mutually beneficial professional relationships, is imperative for long-term career success. Hospitalists should focus on developing their network across multiple departments, such as nursing, subspecialties, medical education, and hospital administration. Curating a broad network will increase your visibility within your organization, showcase your unique services, and demonstrate your value.
To make networking encounters impactful, express interest, actively listen, ask relevant questions, and seek areas of mutual benefit. It’s equally important to cultivate these new relationships after the initial encounter and to demonstrate how your skill set will aid colleagues in achieving their professional goals. Over time, as you establish your niche, deliberate networking with those who share similar interests can lead to a wealth of new experiences and opportunities. Intentionally mastering networking early in your career provides insight into different aspects of the hospital system, new perspectives on ideas, and access to valuable guidance from other professionals. Engaging in networking to establish your support system is an essential step towards success as a first-year hospitalist.
Direction
- Visualize your path: Find a mentor and develop a mission statement and career plan.
Once you’re familiar with your work environment, confident in your identity, and acquainted with your support network, you’re ready for the final step – direction. Hospital medicine offers many professional avenues and clarifying your career path is challenging when attempted alone. A mentor is the necessary catalyst to find direction and purpose.
Selecting and engaging with a mentor will bolster your professional advancement, academic productivity, and most importantly, career satisfaction.1 At its best, mentorship is a symbiotic relationship. Your mentor should inspire you, challenge you, and support your growth and emotional well-being. In turn, as the mentee, you should be proactive, establish expectations, and take responsibility for maintaining communication to ensure a successful relationship. As your career takes shape over time, you may require a mentorship team to fulfill your unique needs.
When you’ve established a relationship with your mentor, take time to develop 1-year and 5-year plans. Your 1-year plan should focus on a few “quick wins,” often projects or opportunities at your home institution. Small victories in your first year will boost your confidence, motivation, and sense of control. Your 5-year plan should delineate the steps necessary to make your first major career transition, such as from instructor to assistant professor. Working with your mentor to draft a career mission statement is a useful first step in this process. Beginning with the end in mind, will help you visualize your direction.2
We hope that the FIND framework will help you find your path to success as a first-year hospitalist.
Dr. Nelson is a hospitalist and instructor of medicine at Harvard Medical School and Beth Israel Deaconess Medical Center, both in Boston. Dr. Ashford is assistant professor and program director, department of internal medicine/pediatrics, at the University of Nebraska Medical Center, Omaha. Dr. Rawal is clinical assistant professor of medicine at the University of Pittsburgh Medical Center. Dr. Crecelius is assistant professor of clinical medicine at Indiana University, Indianapolis. This article is sponsored by the SHM Physicians in Training committee, which submits quarterly content to the Hospitalist on topics relevant to trainees and early -career hospitalists.
References
1. Zerzan JT et al. Making the most of mentors: a guide for mentees. Acad Med. 2009;84:140-4. doi: 10.1097/ACM.0b013e3181906e8f.
2. Covey F. The seven habits of highly effective people. 25th anniversary edition. New York: Simon and Schuster, 2013.
FDA to add myocarditis warning to mRNA COVID-19 vaccines
The Food and Drug Administration is adding a warning to mRNA COVID-19 vaccines’ fact sheets as medical experts continue to investigate cases of heart inflammation, which are rare but are more likely to occur in young men and teen boys.
Doran Fink, MD, PhD, deputy director of the FDA’s division of vaccines and related products applications, told a Centers for Disease Control and Prevention expert panel on June 23 that the FDA is finalizing language on a warning statement for health care providers, vaccine recipients, and parents or caregivers of teens.
The incidents are more likely to follow the second dose of the Pfizer or Moderna vaccine, with chest pain and other symptoms occurring within several days to a week, the warning will note.
“Based on limited follow-up, most cases appear to have been associated with resolution of symptoms, but limited information is available about potential long-term sequelae,” Dr. Fink said, describing the statement to the Advisory Committee on Immunization Practices, independent experts who advise the CDC.
“Symptoms suggestive of myocarditis or pericarditis should result in vaccine recipients seeking medical attention,” he said.
Benefits outweigh risks
Although no formal vote occurred after the meeting, the ACIP members delivered a strong endorsement for continuing to vaccinate 12- to 29-year-olds with the Pfizer and Moderna vaccines despite the warning.
“To me it’s clear, based on current information, that the benefits of vaccine clearly outweigh the risks,” said ACIP member Veronica McNally, president and CEO of the Franny Strong Foundation in Bloomfield, Mich., a sentiment echoed by other members.
As ACIP was meeting, leaders of the nation’s major physician, nurse, and public health associations issued a statement supporting continued vaccination: “The facts are clear: this is an extremely rare side effect, and only an exceedingly small number of people will experience it after vaccination.
“Importantly, for the young people who do, most cases are mild, and individuals recover often on their own or with minimal treatment. In addition, we know that myocarditis and pericarditis are much more common if you get COVID-19, and the risks to the heart from COVID-19 infection can be more severe.”
ACIP heard the evidence behind that claim. According to the Vaccine Safety Datalink, which contains data from more than 12 million medical records, myocarditis or pericarditis occurs in 12- to 39-year-olds at a rate of 8 per 1 million after the second Pfizer dose and 19.8 per 1 million after the second Moderna dose.
The CDC continues to investigate the link between the mRNA vaccines and heart inflammation, including any differences between the vaccines.
Most of the symptoms resolved quickly, said Tom Shimabukuro, deputy director of CDC’s Immunization Safety Office. Of 323 cases analyzed by the CDC, 309 were hospitalized, 295 were discharged, and 218, or 79%, had recovered from symptoms.
“Most postvaccine myocarditis has been responding to minimal treatment,” pediatric cardiologist Matthew Oster, MD, MPH, from Children’s Healthcare of Atlanta, told the panel.
COVID ‘risks are higher’
Overall, the CDC has reported 2,767 COVID-19 deaths among people aged 12-29 years, and there have been 4,018 reported cases of the COVID-linked inflammatory disorder MIS-C since the beginning of the pandemic.
That amounts to 1 MIS-C case in every 3,200 COVID infections – 36% of them among teens aged 12-20 years and 62% among children who are Hispanic or Black and non-Hispanic, according to a CDC presentation.
The CDC estimated that every 1 million second-dose COVID vaccines administered to 12- to 17-year-old boys could prevent 5,700 cases of COVID-19, 215 hospitalizations, 71 ICU admissions, and 2 deaths. There could also be 56-69 myocarditis cases.
The emergence of new variants in the United States and the skewed pattern of vaccination around the country also may increase the risk to unvaccinated young people, noted Grace Lee, MD, MPH, chair of the ACIP’s COVID-19 Vaccine Safety Technical Subgroup and a pediatric infectious disease physician at Stanford (Calif.) Children’s Health.
“If you’re in an area with low vaccination, the risks are higher,” she said. “The benefits [of the vaccine] are going to be far, far greater than any risk.”
Individuals, parents, and their clinicians should consider the full scope of risk when making decisions about vaccination, she said.
As the pandemic evolves, medical experts have to balance the known risks and benefits while they gather more information, said William Schaffner, MD, an infectious disease physician at Vanderbilt University, Nashville, Tenn., and medical director of the National Foundation for Infectious Diseases.
“The story is not over,” Dr. Schaffner said in an interview. “Clearly, we are still working in the face of a pandemic, so there’s urgency to continue vaccinating. But they would like to know more about the long-term consequences of the myocarditis.”
Booster possibilities
Meanwhile, ACIP began conversations on the parameters for a possible vaccine booster. For now, there are simply questions: Would a third vaccine help the immunocompromised gain protection? Should people get a different type of vaccine – mRNA versus adenovirus vector – for their booster? Most important, how long do antibodies last?
“Prior to going around giving everyone boosters, we really need to improve the overall vaccination coverage,” said Helen Keipp Talbot, MD, associate professor of medicine at Vanderbilt University. “That will protect everyone.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration is adding a warning to mRNA COVID-19 vaccines’ fact sheets as medical experts continue to investigate cases of heart inflammation, which are rare but are more likely to occur in young men and teen boys.
Doran Fink, MD, PhD, deputy director of the FDA’s division of vaccines and related products applications, told a Centers for Disease Control and Prevention expert panel on June 23 that the FDA is finalizing language on a warning statement for health care providers, vaccine recipients, and parents or caregivers of teens.
The incidents are more likely to follow the second dose of the Pfizer or Moderna vaccine, with chest pain and other symptoms occurring within several days to a week, the warning will note.
“Based on limited follow-up, most cases appear to have been associated with resolution of symptoms, but limited information is available about potential long-term sequelae,” Dr. Fink said, describing the statement to the Advisory Committee on Immunization Practices, independent experts who advise the CDC.
“Symptoms suggestive of myocarditis or pericarditis should result in vaccine recipients seeking medical attention,” he said.
Benefits outweigh risks
Although no formal vote occurred after the meeting, the ACIP members delivered a strong endorsement for continuing to vaccinate 12- to 29-year-olds with the Pfizer and Moderna vaccines despite the warning.
“To me it’s clear, based on current information, that the benefits of vaccine clearly outweigh the risks,” said ACIP member Veronica McNally, president and CEO of the Franny Strong Foundation in Bloomfield, Mich., a sentiment echoed by other members.
As ACIP was meeting, leaders of the nation’s major physician, nurse, and public health associations issued a statement supporting continued vaccination: “The facts are clear: this is an extremely rare side effect, and only an exceedingly small number of people will experience it after vaccination.
“Importantly, for the young people who do, most cases are mild, and individuals recover often on their own or with minimal treatment. In addition, we know that myocarditis and pericarditis are much more common if you get COVID-19, and the risks to the heart from COVID-19 infection can be more severe.”
ACIP heard the evidence behind that claim. According to the Vaccine Safety Datalink, which contains data from more than 12 million medical records, myocarditis or pericarditis occurs in 12- to 39-year-olds at a rate of 8 per 1 million after the second Pfizer dose and 19.8 per 1 million after the second Moderna dose.
The CDC continues to investigate the link between the mRNA vaccines and heart inflammation, including any differences between the vaccines.
Most of the symptoms resolved quickly, said Tom Shimabukuro, deputy director of CDC’s Immunization Safety Office. Of 323 cases analyzed by the CDC, 309 were hospitalized, 295 were discharged, and 218, or 79%, had recovered from symptoms.
“Most postvaccine myocarditis has been responding to minimal treatment,” pediatric cardiologist Matthew Oster, MD, MPH, from Children’s Healthcare of Atlanta, told the panel.
COVID ‘risks are higher’
Overall, the CDC has reported 2,767 COVID-19 deaths among people aged 12-29 years, and there have been 4,018 reported cases of the COVID-linked inflammatory disorder MIS-C since the beginning of the pandemic.
That amounts to 1 MIS-C case in every 3,200 COVID infections – 36% of them among teens aged 12-20 years and 62% among children who are Hispanic or Black and non-Hispanic, according to a CDC presentation.
The CDC estimated that every 1 million second-dose COVID vaccines administered to 12- to 17-year-old boys could prevent 5,700 cases of COVID-19, 215 hospitalizations, 71 ICU admissions, and 2 deaths. There could also be 56-69 myocarditis cases.
The emergence of new variants in the United States and the skewed pattern of vaccination around the country also may increase the risk to unvaccinated young people, noted Grace Lee, MD, MPH, chair of the ACIP’s COVID-19 Vaccine Safety Technical Subgroup and a pediatric infectious disease physician at Stanford (Calif.) Children’s Health.
“If you’re in an area with low vaccination, the risks are higher,” she said. “The benefits [of the vaccine] are going to be far, far greater than any risk.”
Individuals, parents, and their clinicians should consider the full scope of risk when making decisions about vaccination, she said.
As the pandemic evolves, medical experts have to balance the known risks and benefits while they gather more information, said William Schaffner, MD, an infectious disease physician at Vanderbilt University, Nashville, Tenn., and medical director of the National Foundation for Infectious Diseases.
“The story is not over,” Dr. Schaffner said in an interview. “Clearly, we are still working in the face of a pandemic, so there’s urgency to continue vaccinating. But they would like to know more about the long-term consequences of the myocarditis.”
Booster possibilities
Meanwhile, ACIP began conversations on the parameters for a possible vaccine booster. For now, there are simply questions: Would a third vaccine help the immunocompromised gain protection? Should people get a different type of vaccine – mRNA versus adenovirus vector – for their booster? Most important, how long do antibodies last?
“Prior to going around giving everyone boosters, we really need to improve the overall vaccination coverage,” said Helen Keipp Talbot, MD, associate professor of medicine at Vanderbilt University. “That will protect everyone.”
A version of this article first appeared on Medscape.com.
The Food and Drug Administration is adding a warning to mRNA COVID-19 vaccines’ fact sheets as medical experts continue to investigate cases of heart inflammation, which are rare but are more likely to occur in young men and teen boys.
Doran Fink, MD, PhD, deputy director of the FDA’s division of vaccines and related products applications, told a Centers for Disease Control and Prevention expert panel on June 23 that the FDA is finalizing language on a warning statement for health care providers, vaccine recipients, and parents or caregivers of teens.
The incidents are more likely to follow the second dose of the Pfizer or Moderna vaccine, with chest pain and other symptoms occurring within several days to a week, the warning will note.
“Based on limited follow-up, most cases appear to have been associated with resolution of symptoms, but limited information is available about potential long-term sequelae,” Dr. Fink said, describing the statement to the Advisory Committee on Immunization Practices, independent experts who advise the CDC.
“Symptoms suggestive of myocarditis or pericarditis should result in vaccine recipients seeking medical attention,” he said.
Benefits outweigh risks
Although no formal vote occurred after the meeting, the ACIP members delivered a strong endorsement for continuing to vaccinate 12- to 29-year-olds with the Pfizer and Moderna vaccines despite the warning.
“To me it’s clear, based on current information, that the benefits of vaccine clearly outweigh the risks,” said ACIP member Veronica McNally, president and CEO of the Franny Strong Foundation in Bloomfield, Mich., a sentiment echoed by other members.
As ACIP was meeting, leaders of the nation’s major physician, nurse, and public health associations issued a statement supporting continued vaccination: “The facts are clear: this is an extremely rare side effect, and only an exceedingly small number of people will experience it after vaccination.
“Importantly, for the young people who do, most cases are mild, and individuals recover often on their own or with minimal treatment. In addition, we know that myocarditis and pericarditis are much more common if you get COVID-19, and the risks to the heart from COVID-19 infection can be more severe.”
ACIP heard the evidence behind that claim. According to the Vaccine Safety Datalink, which contains data from more than 12 million medical records, myocarditis or pericarditis occurs in 12- to 39-year-olds at a rate of 8 per 1 million after the second Pfizer dose and 19.8 per 1 million after the second Moderna dose.
The CDC continues to investigate the link between the mRNA vaccines and heart inflammation, including any differences between the vaccines.
Most of the symptoms resolved quickly, said Tom Shimabukuro, deputy director of CDC’s Immunization Safety Office. Of 323 cases analyzed by the CDC, 309 were hospitalized, 295 were discharged, and 218, or 79%, had recovered from symptoms.
“Most postvaccine myocarditis has been responding to minimal treatment,” pediatric cardiologist Matthew Oster, MD, MPH, from Children’s Healthcare of Atlanta, told the panel.
COVID ‘risks are higher’
Overall, the CDC has reported 2,767 COVID-19 deaths among people aged 12-29 years, and there have been 4,018 reported cases of the COVID-linked inflammatory disorder MIS-C since the beginning of the pandemic.
That amounts to 1 MIS-C case in every 3,200 COVID infections – 36% of them among teens aged 12-20 years and 62% among children who are Hispanic or Black and non-Hispanic, according to a CDC presentation.
The CDC estimated that every 1 million second-dose COVID vaccines administered to 12- to 17-year-old boys could prevent 5,700 cases of COVID-19, 215 hospitalizations, 71 ICU admissions, and 2 deaths. There could also be 56-69 myocarditis cases.
The emergence of new variants in the United States and the skewed pattern of vaccination around the country also may increase the risk to unvaccinated young people, noted Grace Lee, MD, MPH, chair of the ACIP’s COVID-19 Vaccine Safety Technical Subgroup and a pediatric infectious disease physician at Stanford (Calif.) Children’s Health.
“If you’re in an area with low vaccination, the risks are higher,” she said. “The benefits [of the vaccine] are going to be far, far greater than any risk.”
Individuals, parents, and their clinicians should consider the full scope of risk when making decisions about vaccination, she said.
As the pandemic evolves, medical experts have to balance the known risks and benefits while they gather more information, said William Schaffner, MD, an infectious disease physician at Vanderbilt University, Nashville, Tenn., and medical director of the National Foundation for Infectious Diseases.
“The story is not over,” Dr. Schaffner said in an interview. “Clearly, we are still working in the face of a pandemic, so there’s urgency to continue vaccinating. But they would like to know more about the long-term consequences of the myocarditis.”
Booster possibilities
Meanwhile, ACIP began conversations on the parameters for a possible vaccine booster. For now, there are simply questions: Would a third vaccine help the immunocompromised gain protection? Should people get a different type of vaccine – mRNA versus adenovirus vector – for their booster? Most important, how long do antibodies last?
“Prior to going around giving everyone boosters, we really need to improve the overall vaccination coverage,” said Helen Keipp Talbot, MD, associate professor of medicine at Vanderbilt University. “That will protect everyone.”
A version of this article first appeared on Medscape.com.
Tofacitinib shows mortality benefit in patients with COVID-19 pneumonia
The Janus kinase inhibitor tofacitinib reduces the risk of both death and respiratory failure in hospitalized adults with COVID-19 pneumonia, a new Brazilian study has found.
“Whether the use of JAK inhibitors is superior or additive to other specific immunomodulatory therapies in patients hospitalized with COVID-19 remains to be determined,” Patrícia O. Guimarães, MD, PhD, of the Hospital Israelita Albert Einstein in São Paulo, and coauthors wrote. The study was published in the New England Journal of Medicine.
The results of previous trials that tested JAK inhibitors as therapies for COVID-19 have been mixed. The second iteration of the Adaptive COVID-19 Treatment Trial (ACTT-2) found that a combination treatment of baricitinib and the Food and Drug Administration–authorized remdesivir was superior to remdesivir alone, but ACTT-4 – which compared baricitinib plus remdesivir with dexamethasone plus remdesivir – was stopped for futility in April 2021.
To assess the efficacy and safety of tofacitinib as a potential treatment for COVID-19, the researchers launched a randomized, double-blind trial made up of 289 patients from 15 sites in Brazil. The Study of Tofacitinib in Hospitalized Patients with COVID-19 Pneumonia (STOP-COVID) split its participants into two groups: one (n = 144) received 10 mg of oral tofacitinib twice daily and the other (n = 145) received placebo. Treatment was to be administered for up to 14 days or until hospital discharge. The participants’ mean age was 56 years, and 34.9% were women.
Over 89% of participants received glucocorticoids during hospitalization, a significant increase, compared with ACTT-2’s 12%. Through 28 days, death or respiratory failure occurred in 18.1% of the tofacitinib group and in 29.0% of the placebo group (risk ratio, 0.63; 95% confidence interval, 0.41-0.97; P = .04). Death from any cause occurred in 2.8% of the tofacitinib group and 5.5% of the placebo group (hazard ratio, 0.49; 95% CI, 0.15-1.63). The median number of days that treatment was administered was 5 in the tofacitinib group and 6 in the placebo group, and the median duration of hospital and ICU stays were similar across groups.
On the eight-level National Institute of Allergy and Infectious Diseases ordinal scale of disease severity, the proportional odds of having a worse score with tofacitinib, compared with placebo, was 0.6 (95% CI, 0.36-1.00) at day 14 and 0.54 (95% CI, 0.27-1.06) at day 28. Adverse events occurred in 26.1% of the tofacitinib group and 22.5% of the placebo group, with serious adverse events occurring in 20 patients (14.1%) on tofacitinib and 17 patients (12%) on placebo. Patients on tofacitinib suffered from events like deep vein thrombosis, acute myocardial infarction, ventricular tachycardia, and myocarditis, each of which affected one person, while one placebo patient each suffered from hemorrhagic stroke and cardiogenic shock. The incidence of serious infection was 3.5% in the tofacitinib group and 4.2% in the placebo group.
Timing may be everything
“There is a lot of interest in repurposing a variety of disease-modifying antirheumatic drugs for the treatment of COVID-19, which includes JAK inhibitors,” Zachary S. Wallace, MD, of the rheumatology unit at Massachusetts General Hospital, Boston, said in an interview. “The ACTT-2 data was compelling; it did suggest perhaps a benefit associated with baricitinib for COVID. This study certainly is more compelling.”
“For many people, there is this hyperinflammatory response in COVID-19 that seems to drive a lot of the morbidity and mortality that we see,” he added. “I think we all hypothesize that some of our treatments may be beneficial there. The challenge that we face is figuring out when the best time is to administer these medicines, and whether they need to be administered as part of a cocktail of therapy.”
Along those lines, Dr. Wallace cited a recent study he coauthored in which rheumatoid arthritis patients who were on JAK inhibitors at baseline had worse COVID-19 severity. But he emphasized that, despite their differing findings, the two studies are not irreconcilable.
“What this might speak to is, the timing of your exposure may be really important,” he said. “At the time of your initial infection, you may need certain aspects of your immune system that a JAK inhibitor may interfere with. But when you initiate a JAK inhibitor, once that phase is complete and you’re in this hyperinflammatory phase, you may have more benefit to target and treat the intense inflammation that we observe in patients who have COVID.”
He also offered up another variable potentially in play: different JAK inhibitors having different targets among the JAK receptors. “It may be that targeting specific JAKs is more beneficial when it comes to treating the hyperinflammatory response of COVID-19.”
The trial was sponsored by Pfizer. Several authors acknowledged potential conflicts of interest, including receiving grants and personal fees from Pfizer and various other pharmaceutical companies.
The Janus kinase inhibitor tofacitinib reduces the risk of both death and respiratory failure in hospitalized adults with COVID-19 pneumonia, a new Brazilian study has found.
“Whether the use of JAK inhibitors is superior or additive to other specific immunomodulatory therapies in patients hospitalized with COVID-19 remains to be determined,” Patrícia O. Guimarães, MD, PhD, of the Hospital Israelita Albert Einstein in São Paulo, and coauthors wrote. The study was published in the New England Journal of Medicine.
The results of previous trials that tested JAK inhibitors as therapies for COVID-19 have been mixed. The second iteration of the Adaptive COVID-19 Treatment Trial (ACTT-2) found that a combination treatment of baricitinib and the Food and Drug Administration–authorized remdesivir was superior to remdesivir alone, but ACTT-4 – which compared baricitinib plus remdesivir with dexamethasone plus remdesivir – was stopped for futility in April 2021.
To assess the efficacy and safety of tofacitinib as a potential treatment for COVID-19, the researchers launched a randomized, double-blind trial made up of 289 patients from 15 sites in Brazil. The Study of Tofacitinib in Hospitalized Patients with COVID-19 Pneumonia (STOP-COVID) split its participants into two groups: one (n = 144) received 10 mg of oral tofacitinib twice daily and the other (n = 145) received placebo. Treatment was to be administered for up to 14 days or until hospital discharge. The participants’ mean age was 56 years, and 34.9% were women.
Over 89% of participants received glucocorticoids during hospitalization, a significant increase, compared with ACTT-2’s 12%. Through 28 days, death or respiratory failure occurred in 18.1% of the tofacitinib group and in 29.0% of the placebo group (risk ratio, 0.63; 95% confidence interval, 0.41-0.97; P = .04). Death from any cause occurred in 2.8% of the tofacitinib group and 5.5% of the placebo group (hazard ratio, 0.49; 95% CI, 0.15-1.63). The median number of days that treatment was administered was 5 in the tofacitinib group and 6 in the placebo group, and the median duration of hospital and ICU stays were similar across groups.
On the eight-level National Institute of Allergy and Infectious Diseases ordinal scale of disease severity, the proportional odds of having a worse score with tofacitinib, compared with placebo, was 0.6 (95% CI, 0.36-1.00) at day 14 and 0.54 (95% CI, 0.27-1.06) at day 28. Adverse events occurred in 26.1% of the tofacitinib group and 22.5% of the placebo group, with serious adverse events occurring in 20 patients (14.1%) on tofacitinib and 17 patients (12%) on placebo. Patients on tofacitinib suffered from events like deep vein thrombosis, acute myocardial infarction, ventricular tachycardia, and myocarditis, each of which affected one person, while one placebo patient each suffered from hemorrhagic stroke and cardiogenic shock. The incidence of serious infection was 3.5% in the tofacitinib group and 4.2% in the placebo group.
Timing may be everything
“There is a lot of interest in repurposing a variety of disease-modifying antirheumatic drugs for the treatment of COVID-19, which includes JAK inhibitors,” Zachary S. Wallace, MD, of the rheumatology unit at Massachusetts General Hospital, Boston, said in an interview. “The ACTT-2 data was compelling; it did suggest perhaps a benefit associated with baricitinib for COVID. This study certainly is more compelling.”
“For many people, there is this hyperinflammatory response in COVID-19 that seems to drive a lot of the morbidity and mortality that we see,” he added. “I think we all hypothesize that some of our treatments may be beneficial there. The challenge that we face is figuring out when the best time is to administer these medicines, and whether they need to be administered as part of a cocktail of therapy.”
Along those lines, Dr. Wallace cited a recent study he coauthored in which rheumatoid arthritis patients who were on JAK inhibitors at baseline had worse COVID-19 severity. But he emphasized that, despite their differing findings, the two studies are not irreconcilable.
“What this might speak to is, the timing of your exposure may be really important,” he said. “At the time of your initial infection, you may need certain aspects of your immune system that a JAK inhibitor may interfere with. But when you initiate a JAK inhibitor, once that phase is complete and you’re in this hyperinflammatory phase, you may have more benefit to target and treat the intense inflammation that we observe in patients who have COVID.”
He also offered up another variable potentially in play: different JAK inhibitors having different targets among the JAK receptors. “It may be that targeting specific JAKs is more beneficial when it comes to treating the hyperinflammatory response of COVID-19.”
The trial was sponsored by Pfizer. Several authors acknowledged potential conflicts of interest, including receiving grants and personal fees from Pfizer and various other pharmaceutical companies.
The Janus kinase inhibitor tofacitinib reduces the risk of both death and respiratory failure in hospitalized adults with COVID-19 pneumonia, a new Brazilian study has found.
“Whether the use of JAK inhibitors is superior or additive to other specific immunomodulatory therapies in patients hospitalized with COVID-19 remains to be determined,” Patrícia O. Guimarães, MD, PhD, of the Hospital Israelita Albert Einstein in São Paulo, and coauthors wrote. The study was published in the New England Journal of Medicine.
The results of previous trials that tested JAK inhibitors as therapies for COVID-19 have been mixed. The second iteration of the Adaptive COVID-19 Treatment Trial (ACTT-2) found that a combination treatment of baricitinib and the Food and Drug Administration–authorized remdesivir was superior to remdesivir alone, but ACTT-4 – which compared baricitinib plus remdesivir with dexamethasone plus remdesivir – was stopped for futility in April 2021.
To assess the efficacy and safety of tofacitinib as a potential treatment for COVID-19, the researchers launched a randomized, double-blind trial made up of 289 patients from 15 sites in Brazil. The Study of Tofacitinib in Hospitalized Patients with COVID-19 Pneumonia (STOP-COVID) split its participants into two groups: one (n = 144) received 10 mg of oral tofacitinib twice daily and the other (n = 145) received placebo. Treatment was to be administered for up to 14 days or until hospital discharge. The participants’ mean age was 56 years, and 34.9% were women.
Over 89% of participants received glucocorticoids during hospitalization, a significant increase, compared with ACTT-2’s 12%. Through 28 days, death or respiratory failure occurred in 18.1% of the tofacitinib group and in 29.0% of the placebo group (risk ratio, 0.63; 95% confidence interval, 0.41-0.97; P = .04). Death from any cause occurred in 2.8% of the tofacitinib group and 5.5% of the placebo group (hazard ratio, 0.49; 95% CI, 0.15-1.63). The median number of days that treatment was administered was 5 in the tofacitinib group and 6 in the placebo group, and the median duration of hospital and ICU stays were similar across groups.
On the eight-level National Institute of Allergy and Infectious Diseases ordinal scale of disease severity, the proportional odds of having a worse score with tofacitinib, compared with placebo, was 0.6 (95% CI, 0.36-1.00) at day 14 and 0.54 (95% CI, 0.27-1.06) at day 28. Adverse events occurred in 26.1% of the tofacitinib group and 22.5% of the placebo group, with serious adverse events occurring in 20 patients (14.1%) on tofacitinib and 17 patients (12%) on placebo. Patients on tofacitinib suffered from events like deep vein thrombosis, acute myocardial infarction, ventricular tachycardia, and myocarditis, each of which affected one person, while one placebo patient each suffered from hemorrhagic stroke and cardiogenic shock. The incidence of serious infection was 3.5% in the tofacitinib group and 4.2% in the placebo group.
Timing may be everything
“There is a lot of interest in repurposing a variety of disease-modifying antirheumatic drugs for the treatment of COVID-19, which includes JAK inhibitors,” Zachary S. Wallace, MD, of the rheumatology unit at Massachusetts General Hospital, Boston, said in an interview. “The ACTT-2 data was compelling; it did suggest perhaps a benefit associated with baricitinib for COVID. This study certainly is more compelling.”
“For many people, there is this hyperinflammatory response in COVID-19 that seems to drive a lot of the morbidity and mortality that we see,” he added. “I think we all hypothesize that some of our treatments may be beneficial there. The challenge that we face is figuring out when the best time is to administer these medicines, and whether they need to be administered as part of a cocktail of therapy.”
Along those lines, Dr. Wallace cited a recent study he coauthored in which rheumatoid arthritis patients who were on JAK inhibitors at baseline had worse COVID-19 severity. But he emphasized that, despite their differing findings, the two studies are not irreconcilable.
“What this might speak to is, the timing of your exposure may be really important,” he said. “At the time of your initial infection, you may need certain aspects of your immune system that a JAK inhibitor may interfere with. But when you initiate a JAK inhibitor, once that phase is complete and you’re in this hyperinflammatory phase, you may have more benefit to target and treat the intense inflammation that we observe in patients who have COVID.”
He also offered up another variable potentially in play: different JAK inhibitors having different targets among the JAK receptors. “It may be that targeting specific JAKs is more beneficial when it comes to treating the hyperinflammatory response of COVID-19.”
The trial was sponsored by Pfizer. Several authors acknowledged potential conflicts of interest, including receiving grants and personal fees from Pfizer and various other pharmaceutical companies.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Sotagliflozin use in T2D patients linked with posthospitalization benefits in analysis
The outcome measure –days alive and out of the hospital – may be a meaningful, patient-centered way of capturing disease burden, the researchers wrote in their paper, published in Annals of Internal Medicine.
“The question was: Can we keep patients alive and out of the hospital for any reason, accounting for the duration of each hospitalization?” author Michael Szarek, PhD, a visiting professor in the division of cardiology at the University of Colorado at Denver, Aurora, said in an interview.
“For every 100 days of follow-up, patients in the sotagliflozin group were alive and out of the hospital 3% more days in relative terms or 2.9 days in absolute terms than those in the placebo group (91.8 vs. 88.9 days),” the researchers reported in their analysis of data from the SOLOIST-WHF trial.
“If you translate that to over the course of a year, that’s more than 10 days,” said Dr. Szarek, who is also a faculty member of CPC Clinical Research, an academic research organization affiliated with the University of Colorado.
Most patients in both groups survived to the end of the study without hospitalization, according to the paper.
Sotagliflozin, a sodium-glucose cotransporter 1 and SGLT2 inhibitor, is not approved in the United States. In 2019, the Food and Drug Administration rejected sotagliflozin as an adjunct to insulin for the treatment of type 1 diabetes after members of an advisory committee expressed concerns about an increased risk for diabetic ketoacidosis with the drug.
Methods and results
To examine whether sotagliflozin increased days alive and out of the hospital in the SOLOIST-WHF trial, Dr. Szarek and colleagues analyzed data from this randomized, double-blind, placebo-controlled study. The trial’s primary results were published in the New England Journal of Medicine in January 2021. Researchers conducted SOLOIST-WHF at more than 300 sites in 32 countries. The trial included 1,222 patients with type 2 diabetes and reduced or preserved ejection fraction who were recently hospitalized for worsening heart failure.
In the new analysis the researchers looked at hospitalizations for any reason and the duration of hospital admissions after randomization. They analyzed days alive and out of the hospital using prespecified models.
Similar proportions of patients who received sotagliflozin and placebo were hospitalized at least once (38.5% vs. 41.4%) during a median follow-up of 9 months. Fewer patients who received sotagliflozin were hospitalized more than once (16.3% vs. 22.1%). In all, 64 patients in the sotagliflozin group and 76 patients in the placebo group died.
The reason for each hospitalization was unspecified, except for cases of heart failure, the authors noted. About 62% of hospitalizations during the trial were for reasons other than heart failure.
Outside expert cites similarities to initial trial
The results for days alive and out of the hospital are “not particularly surprising given the previous publication” of the trial’s primary results, but the new analysis provides a “different view of outcomes that might be clinically meaningful for patients,” commented Frank Brosius, MD, a professor of medicine at the University of Arizona, Tucson.
The SOLOIST-WHF trial indicated that doctors may be able to effectively treat patients with relatively new heart failure with sotagliflozin as long as patients are relatively stable, said Dr. Brosius, who coauthored an editorial in the New England Journal of Medicine that accompanied the initial results from the SOLOIST-WHF trial. It appears that previously reported benefits with regard to heart failure outcomes “showed up in these other indicators” in the secondary analysis.
Still, the effect sizes in the new analysis were relatively small and “probably more studies will be necessary” to examine these end points, he added.
SOLOIST-WHF was funded by Sanofi at initiation and by Lexicon Pharmaceuticals at completion. Dr. Szarek disclosed grants from Lexicon and grants and personal fees from Sanofi, as well as personal fees from other companies. His coauthors included employees of Lexicon and other researchers with financial ties to Lexicon and other pharmaceutical companies. Dr. Brosius disclosed personal fees from the American Diabetes Association and is a member of the Diabetic Kidney Disease Collaborative task force for the American Society of Nephrology that is broadly advocating the use of SGLT2 inhibitors by patients with diabetic kidney diseases. He also has participated in an advisory group for treatment of diabetic kidney disease for Gilead.
The outcome measure –days alive and out of the hospital – may be a meaningful, patient-centered way of capturing disease burden, the researchers wrote in their paper, published in Annals of Internal Medicine.
“The question was: Can we keep patients alive and out of the hospital for any reason, accounting for the duration of each hospitalization?” author Michael Szarek, PhD, a visiting professor in the division of cardiology at the University of Colorado at Denver, Aurora, said in an interview.
“For every 100 days of follow-up, patients in the sotagliflozin group were alive and out of the hospital 3% more days in relative terms or 2.9 days in absolute terms than those in the placebo group (91.8 vs. 88.9 days),” the researchers reported in their analysis of data from the SOLOIST-WHF trial.
“If you translate that to over the course of a year, that’s more than 10 days,” said Dr. Szarek, who is also a faculty member of CPC Clinical Research, an academic research organization affiliated with the University of Colorado.
Most patients in both groups survived to the end of the study without hospitalization, according to the paper.
Sotagliflozin, a sodium-glucose cotransporter 1 and SGLT2 inhibitor, is not approved in the United States. In 2019, the Food and Drug Administration rejected sotagliflozin as an adjunct to insulin for the treatment of type 1 diabetes after members of an advisory committee expressed concerns about an increased risk for diabetic ketoacidosis with the drug.
Methods and results
To examine whether sotagliflozin increased days alive and out of the hospital in the SOLOIST-WHF trial, Dr. Szarek and colleagues analyzed data from this randomized, double-blind, placebo-controlled study. The trial’s primary results were published in the New England Journal of Medicine in January 2021. Researchers conducted SOLOIST-WHF at more than 300 sites in 32 countries. The trial included 1,222 patients with type 2 diabetes and reduced or preserved ejection fraction who were recently hospitalized for worsening heart failure.
In the new analysis the researchers looked at hospitalizations for any reason and the duration of hospital admissions after randomization. They analyzed days alive and out of the hospital using prespecified models.
Similar proportions of patients who received sotagliflozin and placebo were hospitalized at least once (38.5% vs. 41.4%) during a median follow-up of 9 months. Fewer patients who received sotagliflozin were hospitalized more than once (16.3% vs. 22.1%). In all, 64 patients in the sotagliflozin group and 76 patients in the placebo group died.
The reason for each hospitalization was unspecified, except for cases of heart failure, the authors noted. About 62% of hospitalizations during the trial were for reasons other than heart failure.
Outside expert cites similarities to initial trial
The results for days alive and out of the hospital are “not particularly surprising given the previous publication” of the trial’s primary results, but the new analysis provides a “different view of outcomes that might be clinically meaningful for patients,” commented Frank Brosius, MD, a professor of medicine at the University of Arizona, Tucson.
The SOLOIST-WHF trial indicated that doctors may be able to effectively treat patients with relatively new heart failure with sotagliflozin as long as patients are relatively stable, said Dr. Brosius, who coauthored an editorial in the New England Journal of Medicine that accompanied the initial results from the SOLOIST-WHF trial. It appears that previously reported benefits with regard to heart failure outcomes “showed up in these other indicators” in the secondary analysis.
Still, the effect sizes in the new analysis were relatively small and “probably more studies will be necessary” to examine these end points, he added.
SOLOIST-WHF was funded by Sanofi at initiation and by Lexicon Pharmaceuticals at completion. Dr. Szarek disclosed grants from Lexicon and grants and personal fees from Sanofi, as well as personal fees from other companies. His coauthors included employees of Lexicon and other researchers with financial ties to Lexicon and other pharmaceutical companies. Dr. Brosius disclosed personal fees from the American Diabetes Association and is a member of the Diabetic Kidney Disease Collaborative task force for the American Society of Nephrology that is broadly advocating the use of SGLT2 inhibitors by patients with diabetic kidney diseases. He also has participated in an advisory group for treatment of diabetic kidney disease for Gilead.
The outcome measure –days alive and out of the hospital – may be a meaningful, patient-centered way of capturing disease burden, the researchers wrote in their paper, published in Annals of Internal Medicine.
“The question was: Can we keep patients alive and out of the hospital for any reason, accounting for the duration of each hospitalization?” author Michael Szarek, PhD, a visiting professor in the division of cardiology at the University of Colorado at Denver, Aurora, said in an interview.
“For every 100 days of follow-up, patients in the sotagliflozin group were alive and out of the hospital 3% more days in relative terms or 2.9 days in absolute terms than those in the placebo group (91.8 vs. 88.9 days),” the researchers reported in their analysis of data from the SOLOIST-WHF trial.
“If you translate that to over the course of a year, that’s more than 10 days,” said Dr. Szarek, who is also a faculty member of CPC Clinical Research, an academic research organization affiliated with the University of Colorado.
Most patients in both groups survived to the end of the study without hospitalization, according to the paper.
Sotagliflozin, a sodium-glucose cotransporter 1 and SGLT2 inhibitor, is not approved in the United States. In 2019, the Food and Drug Administration rejected sotagliflozin as an adjunct to insulin for the treatment of type 1 diabetes after members of an advisory committee expressed concerns about an increased risk for diabetic ketoacidosis with the drug.
Methods and results
To examine whether sotagliflozin increased days alive and out of the hospital in the SOLOIST-WHF trial, Dr. Szarek and colleagues analyzed data from this randomized, double-blind, placebo-controlled study. The trial’s primary results were published in the New England Journal of Medicine in January 2021. Researchers conducted SOLOIST-WHF at more than 300 sites in 32 countries. The trial included 1,222 patients with type 2 diabetes and reduced or preserved ejection fraction who were recently hospitalized for worsening heart failure.
In the new analysis the researchers looked at hospitalizations for any reason and the duration of hospital admissions after randomization. They analyzed days alive and out of the hospital using prespecified models.
Similar proportions of patients who received sotagliflozin and placebo were hospitalized at least once (38.5% vs. 41.4%) during a median follow-up of 9 months. Fewer patients who received sotagliflozin were hospitalized more than once (16.3% vs. 22.1%). In all, 64 patients in the sotagliflozin group and 76 patients in the placebo group died.
The reason for each hospitalization was unspecified, except for cases of heart failure, the authors noted. About 62% of hospitalizations during the trial were for reasons other than heart failure.
Outside expert cites similarities to initial trial
The results for days alive and out of the hospital are “not particularly surprising given the previous publication” of the trial’s primary results, but the new analysis provides a “different view of outcomes that might be clinically meaningful for patients,” commented Frank Brosius, MD, a professor of medicine at the University of Arizona, Tucson.
The SOLOIST-WHF trial indicated that doctors may be able to effectively treat patients with relatively new heart failure with sotagliflozin as long as patients are relatively stable, said Dr. Brosius, who coauthored an editorial in the New England Journal of Medicine that accompanied the initial results from the SOLOIST-WHF trial. It appears that previously reported benefits with regard to heart failure outcomes “showed up in these other indicators” in the secondary analysis.
Still, the effect sizes in the new analysis were relatively small and “probably more studies will be necessary” to examine these end points, he added.
SOLOIST-WHF was funded by Sanofi at initiation and by Lexicon Pharmaceuticals at completion. Dr. Szarek disclosed grants from Lexicon and grants and personal fees from Sanofi, as well as personal fees from other companies. His coauthors included employees of Lexicon and other researchers with financial ties to Lexicon and other pharmaceutical companies. Dr. Brosius disclosed personal fees from the American Diabetes Association and is a member of the Diabetic Kidney Disease Collaborative task force for the American Society of Nephrology that is broadly advocating the use of SGLT2 inhibitors by patients with diabetic kidney diseases. He also has participated in an advisory group for treatment of diabetic kidney disease for Gilead.
FROM ANNALS OF INTERNAL MEDICINE
New data on COVID-19’s cognitive fallout
Investigators found cognitive changes, depression, and PTSD in infected patients, both in the subacute phase and 10 months after hospital discharge.
“We showed that cognitive and behavioral alterations are associated with COVID-19 infection within 2 months from hospital discharge and that they partially persist in the post-COVID phase,” study investigator Elisa Canu, PhD, neuroimaging research unit, division of neuroscience, IRCCS San Raffaele Scientific Institute, Milan, told a press briefing.
The findings were presented at the annual congress of the European Academy of Neurology.
Executive dysfunction
Previous research suggests about 30% of COVID-19 survivors have cognitive disturbances and 30%-40% have psychopathological disorders including anxiety and depression, said Dr. Canu.
These disturbances have been associated with the severity of acute-phase respiratory symptoms, infection-triggered neuroinflammation, cerebrovascular alterations, and/or neurodegeneration.
However, it’s unclear whether these disturbances persist in the post-COVID phase.
To investigate, the researchers explored cognitive and psychopathological features in 49 patients with confirmed COVID-19 admitted to a hospital ED. They examined these factors at 2 months (subacute phase) and at 10 months (post-COVID phase).
Participants had an average age of 61 years (age range, 40-75 years) and 73% were men. Most had at least one cardiovascular risk factor such as hypertension (55%), smoking (22%), and dyslipidemia (18%).
At hospital admission, 71% had an abnormal neurologic exam, 59% had hypogeusia (reduced sense of taste), 45% hyposmia (reduced sense of smell), 39% headache, and 20% confusion or drowsiness. During hospitalization, 27% had noninvasive ventilation.
In addition to cognitive and neurologic assessments, participants underwent MRI 2 months after hospital discharge. Researchers obtained data on gray matter, white matter, and total brain volume.
At 2 months post discharge, 53% of patients presented with at least one cognitive deficit. Many deficits related to executive function including difficulty planning, attention, and problem solving (16%).
However, some participants had memory issues (6%) or visuospatial disturbances (6%). Almost a quarter (23%) presented with a combination of symptoms related to executive dysfunction.
Low oxygen tied to more cognitive deficits
More than one-third of patients experienced symptoms of depression (16%) or PTSD (18%).
Patients younger than 50 years had more executive dysfunction, with these symptoms affecting 75% of younger patients. “Our explanation for that is that younger people had a milder clinical profile regarding COVID, so they were cared for at home,” said Dr. Canu.
While in hospital, patients may be on “continued alert” and receive structured interventions for cognitive and behavioral issues, she said.
More severe respiratory symptoms at hospital admission were significantly associated with deficits during the subacute phase (P = .002 for information processing).
“Low levels of oxygen in the brain could lead to confusion, headache, and brain fog, and cause the cognitive disturbances that we see,” said Dr. Canu.
White-matter hyperintensities were linked to cognitive deficits during this phase (P < .001 for verbal memory and delayed recall).
“These white-matter lesions are probably preexisting due to cardiovascular risk factors that were present in our population and may have amplified the memory disturbances we saw,” commented Dr. Canu.
The investigators did not find a significant relationship between cognitive performance and brain volume. Dr. Canu noted that cognitive and psychopathological disturbances are linked. For instance, she said, a patient with PTSD or depression may also have problems with attention or memory.
In the post-COVID phase, cognitive symptoms were reduced from 53% to 36%; again, the most common deficit was combined executive dysfunction symptoms. Depression persisted in 15% of patients and PTSD in 18%.
“We still don’t know if these alterations are a consequence of the infection,” said Dr. Canu. “And we don’t know whether the deficits are reversible or are part of a neurodegenerative process.”
The researchers plan to follow these patients further. “We definitely need longer follow-up and bigger populations, if possible, to see if these cognitive and psychopathological disturbances can improve in some way,” said Dr. Canu.
The study results underline the need for neuropsychological and neurologic monitoring in COVID patients. Cognitive stimulation training and physical activity, preferably outdoors, could be beneficial, Dr. Canu added.
A version of this article first appeared on Medscape.com.
Investigators found cognitive changes, depression, and PTSD in infected patients, both in the subacute phase and 10 months after hospital discharge.
“We showed that cognitive and behavioral alterations are associated with COVID-19 infection within 2 months from hospital discharge and that they partially persist in the post-COVID phase,” study investigator Elisa Canu, PhD, neuroimaging research unit, division of neuroscience, IRCCS San Raffaele Scientific Institute, Milan, told a press briefing.
The findings were presented at the annual congress of the European Academy of Neurology.
Executive dysfunction
Previous research suggests about 30% of COVID-19 survivors have cognitive disturbances and 30%-40% have psychopathological disorders including anxiety and depression, said Dr. Canu.
These disturbances have been associated with the severity of acute-phase respiratory symptoms, infection-triggered neuroinflammation, cerebrovascular alterations, and/or neurodegeneration.
However, it’s unclear whether these disturbances persist in the post-COVID phase.
To investigate, the researchers explored cognitive and psychopathological features in 49 patients with confirmed COVID-19 admitted to a hospital ED. They examined these factors at 2 months (subacute phase) and at 10 months (post-COVID phase).
Participants had an average age of 61 years (age range, 40-75 years) and 73% were men. Most had at least one cardiovascular risk factor such as hypertension (55%), smoking (22%), and dyslipidemia (18%).
At hospital admission, 71% had an abnormal neurologic exam, 59% had hypogeusia (reduced sense of taste), 45% hyposmia (reduced sense of smell), 39% headache, and 20% confusion or drowsiness. During hospitalization, 27% had noninvasive ventilation.
In addition to cognitive and neurologic assessments, participants underwent MRI 2 months after hospital discharge. Researchers obtained data on gray matter, white matter, and total brain volume.
At 2 months post discharge, 53% of patients presented with at least one cognitive deficit. Many deficits related to executive function including difficulty planning, attention, and problem solving (16%).
However, some participants had memory issues (6%) or visuospatial disturbances (6%). Almost a quarter (23%) presented with a combination of symptoms related to executive dysfunction.
Low oxygen tied to more cognitive deficits
More than one-third of patients experienced symptoms of depression (16%) or PTSD (18%).
Patients younger than 50 years had more executive dysfunction, with these symptoms affecting 75% of younger patients. “Our explanation for that is that younger people had a milder clinical profile regarding COVID, so they were cared for at home,” said Dr. Canu.
While in hospital, patients may be on “continued alert” and receive structured interventions for cognitive and behavioral issues, she said.
More severe respiratory symptoms at hospital admission were significantly associated with deficits during the subacute phase (P = .002 for information processing).
“Low levels of oxygen in the brain could lead to confusion, headache, and brain fog, and cause the cognitive disturbances that we see,” said Dr. Canu.
White-matter hyperintensities were linked to cognitive deficits during this phase (P < .001 for verbal memory and delayed recall).
“These white-matter lesions are probably preexisting due to cardiovascular risk factors that were present in our population and may have amplified the memory disturbances we saw,” commented Dr. Canu.
The investigators did not find a significant relationship between cognitive performance and brain volume. Dr. Canu noted that cognitive and psychopathological disturbances are linked. For instance, she said, a patient with PTSD or depression may also have problems with attention or memory.
In the post-COVID phase, cognitive symptoms were reduced from 53% to 36%; again, the most common deficit was combined executive dysfunction symptoms. Depression persisted in 15% of patients and PTSD in 18%.
“We still don’t know if these alterations are a consequence of the infection,” said Dr. Canu. “And we don’t know whether the deficits are reversible or are part of a neurodegenerative process.”
The researchers plan to follow these patients further. “We definitely need longer follow-up and bigger populations, if possible, to see if these cognitive and psychopathological disturbances can improve in some way,” said Dr. Canu.
The study results underline the need for neuropsychological and neurologic monitoring in COVID patients. Cognitive stimulation training and physical activity, preferably outdoors, could be beneficial, Dr. Canu added.
A version of this article first appeared on Medscape.com.
Investigators found cognitive changes, depression, and PTSD in infected patients, both in the subacute phase and 10 months after hospital discharge.
“We showed that cognitive and behavioral alterations are associated with COVID-19 infection within 2 months from hospital discharge and that they partially persist in the post-COVID phase,” study investigator Elisa Canu, PhD, neuroimaging research unit, division of neuroscience, IRCCS San Raffaele Scientific Institute, Milan, told a press briefing.
The findings were presented at the annual congress of the European Academy of Neurology.
Executive dysfunction
Previous research suggests about 30% of COVID-19 survivors have cognitive disturbances and 30%-40% have psychopathological disorders including anxiety and depression, said Dr. Canu.
These disturbances have been associated with the severity of acute-phase respiratory symptoms, infection-triggered neuroinflammation, cerebrovascular alterations, and/or neurodegeneration.
However, it’s unclear whether these disturbances persist in the post-COVID phase.
To investigate, the researchers explored cognitive and psychopathological features in 49 patients with confirmed COVID-19 admitted to a hospital ED. They examined these factors at 2 months (subacute phase) and at 10 months (post-COVID phase).
Participants had an average age of 61 years (age range, 40-75 years) and 73% were men. Most had at least one cardiovascular risk factor such as hypertension (55%), smoking (22%), and dyslipidemia (18%).
At hospital admission, 71% had an abnormal neurologic exam, 59% had hypogeusia (reduced sense of taste), 45% hyposmia (reduced sense of smell), 39% headache, and 20% confusion or drowsiness. During hospitalization, 27% had noninvasive ventilation.
In addition to cognitive and neurologic assessments, participants underwent MRI 2 months after hospital discharge. Researchers obtained data on gray matter, white matter, and total brain volume.
At 2 months post discharge, 53% of patients presented with at least one cognitive deficit. Many deficits related to executive function including difficulty planning, attention, and problem solving (16%).
However, some participants had memory issues (6%) or visuospatial disturbances (6%). Almost a quarter (23%) presented with a combination of symptoms related to executive dysfunction.
Low oxygen tied to more cognitive deficits
More than one-third of patients experienced symptoms of depression (16%) or PTSD (18%).
Patients younger than 50 years had more executive dysfunction, with these symptoms affecting 75% of younger patients. “Our explanation for that is that younger people had a milder clinical profile regarding COVID, so they were cared for at home,” said Dr. Canu.
While in hospital, patients may be on “continued alert” and receive structured interventions for cognitive and behavioral issues, she said.
More severe respiratory symptoms at hospital admission were significantly associated with deficits during the subacute phase (P = .002 for information processing).
“Low levels of oxygen in the brain could lead to confusion, headache, and brain fog, and cause the cognitive disturbances that we see,” said Dr. Canu.
White-matter hyperintensities were linked to cognitive deficits during this phase (P < .001 for verbal memory and delayed recall).
“These white-matter lesions are probably preexisting due to cardiovascular risk factors that were present in our population and may have amplified the memory disturbances we saw,” commented Dr. Canu.
The investigators did not find a significant relationship between cognitive performance and brain volume. Dr. Canu noted that cognitive and psychopathological disturbances are linked. For instance, she said, a patient with PTSD or depression may also have problems with attention or memory.
In the post-COVID phase, cognitive symptoms were reduced from 53% to 36%; again, the most common deficit was combined executive dysfunction symptoms. Depression persisted in 15% of patients and PTSD in 18%.
“We still don’t know if these alterations are a consequence of the infection,” said Dr. Canu. “And we don’t know whether the deficits are reversible or are part of a neurodegenerative process.”
The researchers plan to follow these patients further. “We definitely need longer follow-up and bigger populations, if possible, to see if these cognitive and psychopathological disturbances can improve in some way,” said Dr. Canu.
The study results underline the need for neuropsychological and neurologic monitoring in COVID patients. Cognitive stimulation training and physical activity, preferably outdoors, could be beneficial, Dr. Canu added.
A version of this article first appeared on Medscape.com.
Fact or fiction? Intravascular contrast and acute kidney injury
Withholding contrast may be the greater risk
Case
A 73-year-old man with stage III chronic kidney disease (CKD) presents to the emergency department with acute left–upper quadrant pain. Serum creatinine is 2.1mg/dL (eGFR 30 mL/min). Noncontrast computed tomography of the abdomen identifies small bowel inflammation and extensive atherosclerosis. Acute mesenteric ischemia is suspected, but further characterization requires intravenous contrast–enhanced images. He and his family worry about the safety of IV contrast and ask to speak with you.
Introduction
Intravenous iodinated contrast material enhances tissue conspicuity in CT imaging and improves its diagnostic performance. Several case reports published in the 1950s suggested that IV administration of high-osmolality contrast provoked acute kidney injury. An ensuing series of studies associated contrast utilization with renal impairment and additional data extrapolated from cardiology arteriography studies further amplified these concerns.
Contrast media use is often cited as a leading cause of hospital-acquired acute kidney injury.1 The associated fear of causing renal impairment or provoking the need for dialysis frequently leads clinicians to forgo contrast-enhanced CT studies or settle for suboptimal noncontrast imaging even in situations where these tests are clearly indicated. The potential for inadequate imaging to contribute to incomplete, delayed, or incorrect diagnoses represents an ongoing patient safety issue.
A growing body of literature suggests the risks of contrast-associated acute kidney injury are overstated, implying the truer danger lies with inadequate imaging, not contrast media utilization. This review discusses the definitions, risks, and incidence of contrast-associated acute kidney injury, informed by these recent studies.
Overview of the data
Definitions of contrast-induced renal dysfunction vary in clinical studies and range from a creatinine rise of 0.5-1 mg per deciliter or a 25%-50% increase from baseline within 2-5 days following contrast administration. In 2012, the Kidney Disease Improving Global Outcomes working group proposed the term “contrast-associated acute kidney injury” (CA-AKI) and defined it as a plasma creatinine rise of 0.3 mg/dL within 48 hours of contrast exposure, a creatinine increase by a factor of 1.5 over baseline within 7 days of contrast administration, or a urinary volume less than 0.5 mg per kg of body weight within 6 hours of contrast exposure (AKI Network or “AKIN” criteria for CA-AKI).2 Owing in part to inconsistent definitions and partly because of multiple potential confounders, the true incidence of contrast-associated acute kidney injury is uncertain.
The pathogenesis of CA-AKI is incompletely understood, but proposed mechanisms include direct tubular cytotoxic effects; reductions in intrarenal blood flow from contrast material–provoked arteriolar vasoconstriction and contrast-induced increases in blood viscosity; and renal microvascular thrombosis.
Risk factors for CA-AKI overlap with those for acute kidney injury in general. These include CKD, concurrent nephrotoxic medication use, advancing age, diabetes, hemodynamic disturbances to include intravascular volume depletion, systemic illness, and rapid arterial delivery of a large contrast volume.
Current American College of Radiology guidelines state that intravenous isotonic crystalloid volume expansion prior to contrast administration may provide some renal protection, although randomized clinical trial results are inconsistent. The largest clinical trials of N-acetylcysteine showed rates of CA-AKI, need for dialysis, and mortality were no different than placebo. Studies of intravenous sodium bicarbonate show outcomes similar to normal saline.
Introduced in the 1950s and used until the early 2000s, the osmolality of high-osmolality contrast material (HOCM) is roughly five times that of blood (1551 mOsm/kg H2O).3 The early case reports first identifying concern for contrast-induced renal damage were of HOCM used in angiography and pyelography testing. Multiple follow up clinical studies measured creatinine levels before and after contrast administration and classified the percentage of patients whose creatinine level rose above an arbitrary definition of renal injury as having contrast-induced renal injury. These studies formed the basis of the now longstanding concerns about contrast-associated renal dysfunction. Importantly, very few of these HOCM studies included a control group.
Following multiple studies demonstrating an improved safety profile with a similar image quality, the Food and Drug Administration approved low-osmolality contrast (LOCM, 413-796mOsm/kg H2O) in 1985. Early adoption was slow because of its significantly higher cost and incomplete Medicare reimbursement. Prices fell following generic LOCM introduction in 1995 and in 2005 Medicare approved universal reimbursement, leading to widespread use. The FDA approved an iso-osmolality contrast material (290 mOsm/kg H2O) in the mid-1990s; its safety profile and image quality is similar to LOCM. Both LOCM and iso-osmolality contrast material are used in CTs today. Iso-osmolality contrast is more viscous than LOCM and is currently more expensive. Iso-osmolality and LOCM have similar rates of CA-AKI.
A clinical series published in 2008 examined serum creatinine level variation over 5 consecutive days in 30,000 predominantly hospitalized patients who did not receive intravenous contrast material. Investigators simulated contrast administration between days 1 and 2, then observed creatinine changes over the subsequent days. The incidence of acute kidney injury following the simulated contrast dose closely resembled the rates identified in earlier studies that associated contrast exposure with renal injury.4 These results suggested that changes in renal function commonly attributed to contrast exposure may be because of other, concurrent, clinical factors.
A 2013 study compared 8,826 patients with stable renal function who received a low-osmolality contrast-enhanced CT with 8,826 patients who underwent a noncontrast study.5 After 1:1 propensity matching, they found higher rates of CA-AKI (as defined by AKIN criteria) among only those with baseline eGFR less than 30 mL/min. There was a trend towards higher rates of CA-AKI among those with baseline eGFR of 30-44 mL/min, and no difference among the bulk of patients with normal or near normal baseline renal function.
Another large propensity score–matched study published in 2014 compared 6,254 patients who underwent a contrast-enhanced CT with 6,254 patients who underwent a nonenhanced CT.
Investigators stratified this predominantly inpatient cohort by baseline eGFR. Results demonstrated similar rates of AKI between contrast material and non–contrast material cohorts. They concluded that intravenous contrast administration did not significantly affect the risk of acute kidney injury, even in patients with impaired renal function. The authors noted that the difference in contrast-mediated nephrotoxic risk in patients with eGFRless than 30 between their study and the Davenport study could be explained by their use of a different definition of CA-AKI, differences in propensity score calculation, and by enrolling greater numbers of patients with impaired kidney function in their study.6
Finally, a large single-center study published in 2017 included 16,801 ED patients divided into three groups; patients who received a contrast-enhanced CT, patients who underwent a noncontrast CT study, and a set of patients who did not undergo any CT imaging. Patients with creatinine levels under .4 mg/dL or over 4 mg/dL were excluded from initial analysis.
Investigators stratified each patient group by serum creatinine and eGFR and utilized both traditional contrast-induced nephropathy (serum creatinine increase of .5 mg/dL or a 25% increase over baseline serum creatinine level at 48-72 hours) and AKIN criteria to evaluate for acute kidney injury. Propensity score analyses comparing the contrast-enhanced group and two control groups failed to identify any significant change in AKI incidence. The authors concluded that, in situations where contrast-enhanced CT is indicated to avoid missing or delaying potential diagnoses, the risks of diagnostic failure outweigh any potential risks of contrast induced renal injury.7
While these three studies utilized control groups and propensity score matching, they are retrospective in nature and unknown or omitted confounding variables could be present. Together, though, they contribute to a growing body of literature suggesting that the risk of contrast-associated AKI relates less to the contrast itself and more to concurrent clinical factors affecting kidney function. Ethical concerns have to date prevented the conduct of a randomized trial of IV contrast in CT scanning. Table 1 summarizes the findings of these three studies.
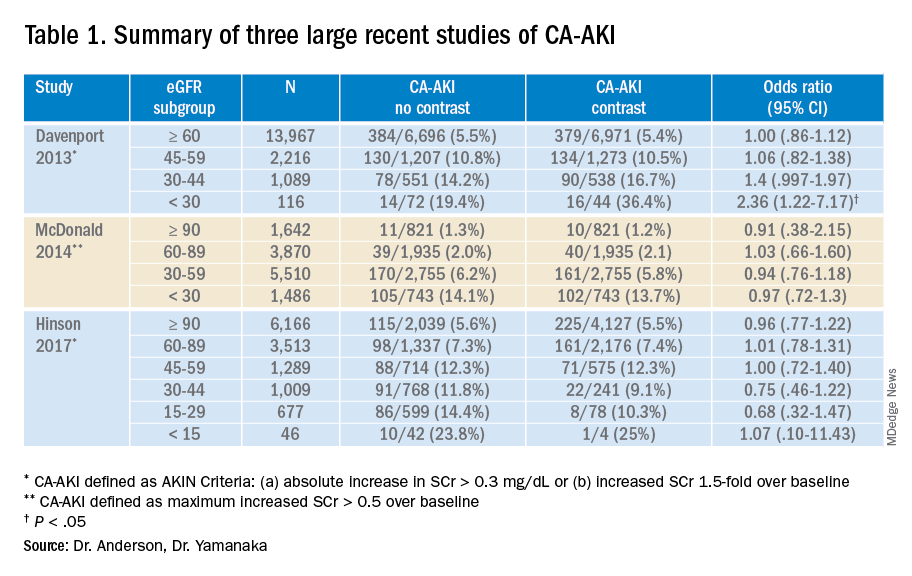
Application of the data to the case
The patient presented with abdominal pain potentially attributable to acute mesenteric ischemia, where a delayed or missed diagnosis can be potentially fatal. He was counseled about the comparatively small risk of CA-AKI with IV contrast and underwent contrast-enhanced CT scanning without incident. The diagnosis of acute mesenteric ischemia was confirmed, and he was referred for urgent laparotomy.
Bottom line
The absolute risk of CA-AKI varies according to baseline renal function and is not clearly linked to the receipt of IV contrast. The risks of withholding contrast may be greater than the risk of CA-AKI. Clinicians should counsel patients accordingly.
Dr. Anderson is national lead, VHA Hospital Medicine, and associate professor of medicine at the Minneapolis VA Health Care System. Dr. Yamanaka is a hospitalist at the Minneapolis VA Medical Center and an assistant professor of medicine at the University of Minnesota.
References
1. Nash K et al. Hospital-acquired renal insufficiency. Am J Kidney Dis. 2002;39(5):930-6. doi: 10.1053/ajkd.2002.32766.
2. Section 4: Contrast-induced AKI. Kidney Int Suppl. 2012;2(1):69-88. doi: 10.1038/kisup.2011.34.
3. Wilmot A et al. The adoption of low-osmolar contrast agents in the United States: Historical analysis of health policy and clinical practice. AJR Am J Roentgenol. 2012;199(5):1049-53. doi: 10.2214/AJR.11.8426.
4. Newhouse JH et al. Frequency of serum creatinine changes in the absence of iodinated contrast material: Implications for studies of contrast nephrotoxicity. AJR Am J Roentgenol. 2008;191(2):376-82. doi: 10.2214/AJR.07.3280.
5. Davenport MS et al. Contrast material-induced nephrotoxicity and intravenous low-osmolality iodinated contrast material: Risk stratification by using estimated glomerular filtration rate. Radiology. 2013;268(3):719-28. doi: 10.1148/radiol.13122276.
6. McDonald JS et al. Risk of intravenous contrast material-mediated acute kidney injury: A propensity score–matched study stratified by baseline-estimated glomerular filtration rate. Radiology. 2014;271(1):65-73. doi: 10.1148/radiol.13130775.
7. Hinson JS et al. Risk of acute kidney injury after intravenous contrast media administration. Ann Emerg Med. 2017;69(5):577-86. doi: 10.1016/j.annemergmed.2016.11.021.
Key points
- Early studies suggesting an association between IV contrast and AKI used an older formulation of contrast media not routinely used today. Importantly, these studies did not use control groups.
- Results from multiple recent large trials comparing IV contrast patients with controls suggest that AKI is not clearly linked to the receipt of IV contrast and that it varies according to baseline renal function.
- Randomized controlled trials of prophylactic normal saline or sodium bicarbonate to prevent CA-AKI show mixed results. Clinical trials comparing N-acetylcysteine with placebo showed no difference in the rates of AKI, dialysis initiation, or mortality.
Quiz
Which of the following is not clearly associated with acute kidney injury in hospitalized patients?
A. Decreased baseline glomerular filtration rate
B. Angiotensin-converting enzyme (ACE) inhibitor use
C. Hemodynamic instability
D. Intravenous contrast administration
Answer: D
While decreased baseline renal function, ACE inhibitors, and hemodynamic instability are known risk factors for hospital-associated renal injury, a growing body of literature suggests that intravenous contrast used in computed tomography studies does not precipitate acute kidney injury.
Further reading
McDonald JS et al. Frequency of acute kidney injury following intravenous contrast medium administration: a systematic review and meta-analysis. Radiology. 2013;267(1):119-128. doi: 10.1148/radiol.12121460.
McDonald RJ et al. Behind the numbers: Propensity score analysis – a primer for the diagnostic radiologist. Radiology. 2013;269(3):640-5. doi: 10.1148/radiol.13131465.
Luk L et al. Intravenous contrast-induced nephropathy – the rise and fall of a threatening idea. Adv Chronic Kidney Dis. 2017;24(3):169-75. doi: 10.1053/j.ackd.2017.03.001.
Mehran R et al. Contrast-associated acute kidney injury. N Engl J Med. 2019;380(22):2146-55. doi: 10.1056/NEJMra1805256.
Withholding contrast may be the greater risk
Withholding contrast may be the greater risk
Case
A 73-year-old man with stage III chronic kidney disease (CKD) presents to the emergency department with acute left–upper quadrant pain. Serum creatinine is 2.1mg/dL (eGFR 30 mL/min). Noncontrast computed tomography of the abdomen identifies small bowel inflammation and extensive atherosclerosis. Acute mesenteric ischemia is suspected, but further characterization requires intravenous contrast–enhanced images. He and his family worry about the safety of IV contrast and ask to speak with you.
Introduction
Intravenous iodinated contrast material enhances tissue conspicuity in CT imaging and improves its diagnostic performance. Several case reports published in the 1950s suggested that IV administration of high-osmolality contrast provoked acute kidney injury. An ensuing series of studies associated contrast utilization with renal impairment and additional data extrapolated from cardiology arteriography studies further amplified these concerns.
Contrast media use is often cited as a leading cause of hospital-acquired acute kidney injury.1 The associated fear of causing renal impairment or provoking the need for dialysis frequently leads clinicians to forgo contrast-enhanced CT studies or settle for suboptimal noncontrast imaging even in situations where these tests are clearly indicated. The potential for inadequate imaging to contribute to incomplete, delayed, or incorrect diagnoses represents an ongoing patient safety issue.
A growing body of literature suggests the risks of contrast-associated acute kidney injury are overstated, implying the truer danger lies with inadequate imaging, not contrast media utilization. This review discusses the definitions, risks, and incidence of contrast-associated acute kidney injury, informed by these recent studies.
Overview of the data
Definitions of contrast-induced renal dysfunction vary in clinical studies and range from a creatinine rise of 0.5-1 mg per deciliter or a 25%-50% increase from baseline within 2-5 days following contrast administration. In 2012, the Kidney Disease Improving Global Outcomes working group proposed the term “contrast-associated acute kidney injury” (CA-AKI) and defined it as a plasma creatinine rise of 0.3 mg/dL within 48 hours of contrast exposure, a creatinine increase by a factor of 1.5 over baseline within 7 days of contrast administration, or a urinary volume less than 0.5 mg per kg of body weight within 6 hours of contrast exposure (AKI Network or “AKIN” criteria for CA-AKI).2 Owing in part to inconsistent definitions and partly because of multiple potential confounders, the true incidence of contrast-associated acute kidney injury is uncertain.
The pathogenesis of CA-AKI is incompletely understood, but proposed mechanisms include direct tubular cytotoxic effects; reductions in intrarenal blood flow from contrast material–provoked arteriolar vasoconstriction and contrast-induced increases in blood viscosity; and renal microvascular thrombosis.
Risk factors for CA-AKI overlap with those for acute kidney injury in general. These include CKD, concurrent nephrotoxic medication use, advancing age, diabetes, hemodynamic disturbances to include intravascular volume depletion, systemic illness, and rapid arterial delivery of a large contrast volume.
Current American College of Radiology guidelines state that intravenous isotonic crystalloid volume expansion prior to contrast administration may provide some renal protection, although randomized clinical trial results are inconsistent. The largest clinical trials of N-acetylcysteine showed rates of CA-AKI, need for dialysis, and mortality were no different than placebo. Studies of intravenous sodium bicarbonate show outcomes similar to normal saline.
Introduced in the 1950s and used until the early 2000s, the osmolality of high-osmolality contrast material (HOCM) is roughly five times that of blood (1551 mOsm/kg H2O).3 The early case reports first identifying concern for contrast-induced renal damage were of HOCM used in angiography and pyelography testing. Multiple follow up clinical studies measured creatinine levels before and after contrast administration and classified the percentage of patients whose creatinine level rose above an arbitrary definition of renal injury as having contrast-induced renal injury. These studies formed the basis of the now longstanding concerns about contrast-associated renal dysfunction. Importantly, very few of these HOCM studies included a control group.
Following multiple studies demonstrating an improved safety profile with a similar image quality, the Food and Drug Administration approved low-osmolality contrast (LOCM, 413-796mOsm/kg H2O) in 1985. Early adoption was slow because of its significantly higher cost and incomplete Medicare reimbursement. Prices fell following generic LOCM introduction in 1995 and in 2005 Medicare approved universal reimbursement, leading to widespread use. The FDA approved an iso-osmolality contrast material (290 mOsm/kg H2O) in the mid-1990s; its safety profile and image quality is similar to LOCM. Both LOCM and iso-osmolality contrast material are used in CTs today. Iso-osmolality contrast is more viscous than LOCM and is currently more expensive. Iso-osmolality and LOCM have similar rates of CA-AKI.
A clinical series published in 2008 examined serum creatinine level variation over 5 consecutive days in 30,000 predominantly hospitalized patients who did not receive intravenous contrast material. Investigators simulated contrast administration between days 1 and 2, then observed creatinine changes over the subsequent days. The incidence of acute kidney injury following the simulated contrast dose closely resembled the rates identified in earlier studies that associated contrast exposure with renal injury.4 These results suggested that changes in renal function commonly attributed to contrast exposure may be because of other, concurrent, clinical factors.
A 2013 study compared 8,826 patients with stable renal function who received a low-osmolality contrast-enhanced CT with 8,826 patients who underwent a noncontrast study.5 After 1:1 propensity matching, they found higher rates of CA-AKI (as defined by AKIN criteria) among only those with baseline eGFR less than 30 mL/min. There was a trend towards higher rates of CA-AKI among those with baseline eGFR of 30-44 mL/min, and no difference among the bulk of patients with normal or near normal baseline renal function.
Another large propensity score–matched study published in 2014 compared 6,254 patients who underwent a contrast-enhanced CT with 6,254 patients who underwent a nonenhanced CT.
Investigators stratified this predominantly inpatient cohort by baseline eGFR. Results demonstrated similar rates of AKI between contrast material and non–contrast material cohorts. They concluded that intravenous contrast administration did not significantly affect the risk of acute kidney injury, even in patients with impaired renal function. The authors noted that the difference in contrast-mediated nephrotoxic risk in patients with eGFRless than 30 between their study and the Davenport study could be explained by their use of a different definition of CA-AKI, differences in propensity score calculation, and by enrolling greater numbers of patients with impaired kidney function in their study.6
Finally, a large single-center study published in 2017 included 16,801 ED patients divided into three groups; patients who received a contrast-enhanced CT, patients who underwent a noncontrast CT study, and a set of patients who did not undergo any CT imaging. Patients with creatinine levels under .4 mg/dL or over 4 mg/dL were excluded from initial analysis.
Investigators stratified each patient group by serum creatinine and eGFR and utilized both traditional contrast-induced nephropathy (serum creatinine increase of .5 mg/dL or a 25% increase over baseline serum creatinine level at 48-72 hours) and AKIN criteria to evaluate for acute kidney injury. Propensity score analyses comparing the contrast-enhanced group and two control groups failed to identify any significant change in AKI incidence. The authors concluded that, in situations where contrast-enhanced CT is indicated to avoid missing or delaying potential diagnoses, the risks of diagnostic failure outweigh any potential risks of contrast induced renal injury.7
While these three studies utilized control groups and propensity score matching, they are retrospective in nature and unknown or omitted confounding variables could be present. Together, though, they contribute to a growing body of literature suggesting that the risk of contrast-associated AKI relates less to the contrast itself and more to concurrent clinical factors affecting kidney function. Ethical concerns have to date prevented the conduct of a randomized trial of IV contrast in CT scanning. Table 1 summarizes the findings of these three studies.

Application of the data to the case
The patient presented with abdominal pain potentially attributable to acute mesenteric ischemia, where a delayed or missed diagnosis can be potentially fatal. He was counseled about the comparatively small risk of CA-AKI with IV contrast and underwent contrast-enhanced CT scanning without incident. The diagnosis of acute mesenteric ischemia was confirmed, and he was referred for urgent laparotomy.
Bottom line
The absolute risk of CA-AKI varies according to baseline renal function and is not clearly linked to the receipt of IV contrast. The risks of withholding contrast may be greater than the risk of CA-AKI. Clinicians should counsel patients accordingly.
Dr. Anderson is national lead, VHA Hospital Medicine, and associate professor of medicine at the Minneapolis VA Health Care System. Dr. Yamanaka is a hospitalist at the Minneapolis VA Medical Center and an assistant professor of medicine at the University of Minnesota.
References
1. Nash K et al. Hospital-acquired renal insufficiency. Am J Kidney Dis. 2002;39(5):930-6. doi: 10.1053/ajkd.2002.32766.
2. Section 4: Contrast-induced AKI. Kidney Int Suppl. 2012;2(1):69-88. doi: 10.1038/kisup.2011.34.
3. Wilmot A et al. The adoption of low-osmolar contrast agents in the United States: Historical analysis of health policy and clinical practice. AJR Am J Roentgenol. 2012;199(5):1049-53. doi: 10.2214/AJR.11.8426.
4. Newhouse JH et al. Frequency of serum creatinine changes in the absence of iodinated contrast material: Implications for studies of contrast nephrotoxicity. AJR Am J Roentgenol. 2008;191(2):376-82. doi: 10.2214/AJR.07.3280.
5. Davenport MS et al. Contrast material-induced nephrotoxicity and intravenous low-osmolality iodinated contrast material: Risk stratification by using estimated glomerular filtration rate. Radiology. 2013;268(3):719-28. doi: 10.1148/radiol.13122276.
6. McDonald JS et al. Risk of intravenous contrast material-mediated acute kidney injury: A propensity score–matched study stratified by baseline-estimated glomerular filtration rate. Radiology. 2014;271(1):65-73. doi: 10.1148/radiol.13130775.
7. Hinson JS et al. Risk of acute kidney injury after intravenous contrast media administration. Ann Emerg Med. 2017;69(5):577-86. doi: 10.1016/j.annemergmed.2016.11.021.
Key points
- Early studies suggesting an association between IV contrast and AKI used an older formulation of contrast media not routinely used today. Importantly, these studies did not use control groups.
- Results from multiple recent large trials comparing IV contrast patients with controls suggest that AKI is not clearly linked to the receipt of IV contrast and that it varies according to baseline renal function.
- Randomized controlled trials of prophylactic normal saline or sodium bicarbonate to prevent CA-AKI show mixed results. Clinical trials comparing N-acetylcysteine with placebo showed no difference in the rates of AKI, dialysis initiation, or mortality.
Quiz
Which of the following is not clearly associated with acute kidney injury in hospitalized patients?
A. Decreased baseline glomerular filtration rate
B. Angiotensin-converting enzyme (ACE) inhibitor use
C. Hemodynamic instability
D. Intravenous contrast administration
Answer: D
While decreased baseline renal function, ACE inhibitors, and hemodynamic instability are known risk factors for hospital-associated renal injury, a growing body of literature suggests that intravenous contrast used in computed tomography studies does not precipitate acute kidney injury.
Further reading
McDonald JS et al. Frequency of acute kidney injury following intravenous contrast medium administration: a systematic review and meta-analysis. Radiology. 2013;267(1):119-128. doi: 10.1148/radiol.12121460.
McDonald RJ et al. Behind the numbers: Propensity score analysis – a primer for the diagnostic radiologist. Radiology. 2013;269(3):640-5. doi: 10.1148/radiol.13131465.
Luk L et al. Intravenous contrast-induced nephropathy – the rise and fall of a threatening idea. Adv Chronic Kidney Dis. 2017;24(3):169-75. doi: 10.1053/j.ackd.2017.03.001.
Mehran R et al. Contrast-associated acute kidney injury. N Engl J Med. 2019;380(22):2146-55. doi: 10.1056/NEJMra1805256.
Case
A 73-year-old man with stage III chronic kidney disease (CKD) presents to the emergency department with acute left–upper quadrant pain. Serum creatinine is 2.1mg/dL (eGFR 30 mL/min). Noncontrast computed tomography of the abdomen identifies small bowel inflammation and extensive atherosclerosis. Acute mesenteric ischemia is suspected, but further characterization requires intravenous contrast–enhanced images. He and his family worry about the safety of IV contrast and ask to speak with you.
Introduction
Intravenous iodinated contrast material enhances tissue conspicuity in CT imaging and improves its diagnostic performance. Several case reports published in the 1950s suggested that IV administration of high-osmolality contrast provoked acute kidney injury. An ensuing series of studies associated contrast utilization with renal impairment and additional data extrapolated from cardiology arteriography studies further amplified these concerns.
Contrast media use is often cited as a leading cause of hospital-acquired acute kidney injury.1 The associated fear of causing renal impairment or provoking the need for dialysis frequently leads clinicians to forgo contrast-enhanced CT studies or settle for suboptimal noncontrast imaging even in situations where these tests are clearly indicated. The potential for inadequate imaging to contribute to incomplete, delayed, or incorrect diagnoses represents an ongoing patient safety issue.
A growing body of literature suggests the risks of contrast-associated acute kidney injury are overstated, implying the truer danger lies with inadequate imaging, not contrast media utilization. This review discusses the definitions, risks, and incidence of contrast-associated acute kidney injury, informed by these recent studies.
Overview of the data
Definitions of contrast-induced renal dysfunction vary in clinical studies and range from a creatinine rise of 0.5-1 mg per deciliter or a 25%-50% increase from baseline within 2-5 days following contrast administration. In 2012, the Kidney Disease Improving Global Outcomes working group proposed the term “contrast-associated acute kidney injury” (CA-AKI) and defined it as a plasma creatinine rise of 0.3 mg/dL within 48 hours of contrast exposure, a creatinine increase by a factor of 1.5 over baseline within 7 days of contrast administration, or a urinary volume less than 0.5 mg per kg of body weight within 6 hours of contrast exposure (AKI Network or “AKIN” criteria for CA-AKI).2 Owing in part to inconsistent definitions and partly because of multiple potential confounders, the true incidence of contrast-associated acute kidney injury is uncertain.
The pathogenesis of CA-AKI is incompletely understood, but proposed mechanisms include direct tubular cytotoxic effects; reductions in intrarenal blood flow from contrast material–provoked arteriolar vasoconstriction and contrast-induced increases in blood viscosity; and renal microvascular thrombosis.
Risk factors for CA-AKI overlap with those for acute kidney injury in general. These include CKD, concurrent nephrotoxic medication use, advancing age, diabetes, hemodynamic disturbances to include intravascular volume depletion, systemic illness, and rapid arterial delivery of a large contrast volume.
Current American College of Radiology guidelines state that intravenous isotonic crystalloid volume expansion prior to contrast administration may provide some renal protection, although randomized clinical trial results are inconsistent. The largest clinical trials of N-acetylcysteine showed rates of CA-AKI, need for dialysis, and mortality were no different than placebo. Studies of intravenous sodium bicarbonate show outcomes similar to normal saline.
Introduced in the 1950s and used until the early 2000s, the osmolality of high-osmolality contrast material (HOCM) is roughly five times that of blood (1551 mOsm/kg H2O).3 The early case reports first identifying concern for contrast-induced renal damage were of HOCM used in angiography and pyelography testing. Multiple follow up clinical studies measured creatinine levels before and after contrast administration and classified the percentage of patients whose creatinine level rose above an arbitrary definition of renal injury as having contrast-induced renal injury. These studies formed the basis of the now longstanding concerns about contrast-associated renal dysfunction. Importantly, very few of these HOCM studies included a control group.
Following multiple studies demonstrating an improved safety profile with a similar image quality, the Food and Drug Administration approved low-osmolality contrast (LOCM, 413-796mOsm/kg H2O) in 1985. Early adoption was slow because of its significantly higher cost and incomplete Medicare reimbursement. Prices fell following generic LOCM introduction in 1995 and in 2005 Medicare approved universal reimbursement, leading to widespread use. The FDA approved an iso-osmolality contrast material (290 mOsm/kg H2O) in the mid-1990s; its safety profile and image quality is similar to LOCM. Both LOCM and iso-osmolality contrast material are used in CTs today. Iso-osmolality contrast is more viscous than LOCM and is currently more expensive. Iso-osmolality and LOCM have similar rates of CA-AKI.
A clinical series published in 2008 examined serum creatinine level variation over 5 consecutive days in 30,000 predominantly hospitalized patients who did not receive intravenous contrast material. Investigators simulated contrast administration between days 1 and 2, then observed creatinine changes over the subsequent days. The incidence of acute kidney injury following the simulated contrast dose closely resembled the rates identified in earlier studies that associated contrast exposure with renal injury.4 These results suggested that changes in renal function commonly attributed to contrast exposure may be because of other, concurrent, clinical factors.
A 2013 study compared 8,826 patients with stable renal function who received a low-osmolality contrast-enhanced CT with 8,826 patients who underwent a noncontrast study.5 After 1:1 propensity matching, they found higher rates of CA-AKI (as defined by AKIN criteria) among only those with baseline eGFR less than 30 mL/min. There was a trend towards higher rates of CA-AKI among those with baseline eGFR of 30-44 mL/min, and no difference among the bulk of patients with normal or near normal baseline renal function.
Another large propensity score–matched study published in 2014 compared 6,254 patients who underwent a contrast-enhanced CT with 6,254 patients who underwent a nonenhanced CT.
Investigators stratified this predominantly inpatient cohort by baseline eGFR. Results demonstrated similar rates of AKI between contrast material and non–contrast material cohorts. They concluded that intravenous contrast administration did not significantly affect the risk of acute kidney injury, even in patients with impaired renal function. The authors noted that the difference in contrast-mediated nephrotoxic risk in patients with eGFRless than 30 between their study and the Davenport study could be explained by their use of a different definition of CA-AKI, differences in propensity score calculation, and by enrolling greater numbers of patients with impaired kidney function in their study.6
Finally, a large single-center study published in 2017 included 16,801 ED patients divided into three groups; patients who received a contrast-enhanced CT, patients who underwent a noncontrast CT study, and a set of patients who did not undergo any CT imaging. Patients with creatinine levels under .4 mg/dL or over 4 mg/dL were excluded from initial analysis.
Investigators stratified each patient group by serum creatinine and eGFR and utilized both traditional contrast-induced nephropathy (serum creatinine increase of .5 mg/dL or a 25% increase over baseline serum creatinine level at 48-72 hours) and AKIN criteria to evaluate for acute kidney injury. Propensity score analyses comparing the contrast-enhanced group and two control groups failed to identify any significant change in AKI incidence. The authors concluded that, in situations where contrast-enhanced CT is indicated to avoid missing or delaying potential diagnoses, the risks of diagnostic failure outweigh any potential risks of contrast induced renal injury.7
While these three studies utilized control groups and propensity score matching, they are retrospective in nature and unknown or omitted confounding variables could be present. Together, though, they contribute to a growing body of literature suggesting that the risk of contrast-associated AKI relates less to the contrast itself and more to concurrent clinical factors affecting kidney function. Ethical concerns have to date prevented the conduct of a randomized trial of IV contrast in CT scanning. Table 1 summarizes the findings of these three studies.

Application of the data to the case
The patient presented with abdominal pain potentially attributable to acute mesenteric ischemia, where a delayed or missed diagnosis can be potentially fatal. He was counseled about the comparatively small risk of CA-AKI with IV contrast and underwent contrast-enhanced CT scanning without incident. The diagnosis of acute mesenteric ischemia was confirmed, and he was referred for urgent laparotomy.
Bottom line
The absolute risk of CA-AKI varies according to baseline renal function and is not clearly linked to the receipt of IV contrast. The risks of withholding contrast may be greater than the risk of CA-AKI. Clinicians should counsel patients accordingly.
Dr. Anderson is national lead, VHA Hospital Medicine, and associate professor of medicine at the Minneapolis VA Health Care System. Dr. Yamanaka is a hospitalist at the Minneapolis VA Medical Center and an assistant professor of medicine at the University of Minnesota.
References
1. Nash K et al. Hospital-acquired renal insufficiency. Am J Kidney Dis. 2002;39(5):930-6. doi: 10.1053/ajkd.2002.32766.
2. Section 4: Contrast-induced AKI. Kidney Int Suppl. 2012;2(1):69-88. doi: 10.1038/kisup.2011.34.
3. Wilmot A et al. The adoption of low-osmolar contrast agents in the United States: Historical analysis of health policy and clinical practice. AJR Am J Roentgenol. 2012;199(5):1049-53. doi: 10.2214/AJR.11.8426.
4. Newhouse JH et al. Frequency of serum creatinine changes in the absence of iodinated contrast material: Implications for studies of contrast nephrotoxicity. AJR Am J Roentgenol. 2008;191(2):376-82. doi: 10.2214/AJR.07.3280.
5. Davenport MS et al. Contrast material-induced nephrotoxicity and intravenous low-osmolality iodinated contrast material: Risk stratification by using estimated glomerular filtration rate. Radiology. 2013;268(3):719-28. doi: 10.1148/radiol.13122276.
6. McDonald JS et al. Risk of intravenous contrast material-mediated acute kidney injury: A propensity score–matched study stratified by baseline-estimated glomerular filtration rate. Radiology. 2014;271(1):65-73. doi: 10.1148/radiol.13130775.
7. Hinson JS et al. Risk of acute kidney injury after intravenous contrast media administration. Ann Emerg Med. 2017;69(5):577-86. doi: 10.1016/j.annemergmed.2016.11.021.
Key points
- Early studies suggesting an association between IV contrast and AKI used an older formulation of contrast media not routinely used today. Importantly, these studies did not use control groups.
- Results from multiple recent large trials comparing IV contrast patients with controls suggest that AKI is not clearly linked to the receipt of IV contrast and that it varies according to baseline renal function.
- Randomized controlled trials of prophylactic normal saline or sodium bicarbonate to prevent CA-AKI show mixed results. Clinical trials comparing N-acetylcysteine with placebo showed no difference in the rates of AKI, dialysis initiation, or mortality.
Quiz
Which of the following is not clearly associated with acute kidney injury in hospitalized patients?
A. Decreased baseline glomerular filtration rate
B. Angiotensin-converting enzyme (ACE) inhibitor use
C. Hemodynamic instability
D. Intravenous contrast administration
Answer: D
While decreased baseline renal function, ACE inhibitors, and hemodynamic instability are known risk factors for hospital-associated renal injury, a growing body of literature suggests that intravenous contrast used in computed tomography studies does not precipitate acute kidney injury.
Further reading
McDonald JS et al. Frequency of acute kidney injury following intravenous contrast medium administration: a systematic review and meta-analysis. Radiology. 2013;267(1):119-128. doi: 10.1148/radiol.12121460.
McDonald RJ et al. Behind the numbers: Propensity score analysis – a primer for the diagnostic radiologist. Radiology. 2013;269(3):640-5. doi: 10.1148/radiol.13131465.
Luk L et al. Intravenous contrast-induced nephropathy – the rise and fall of a threatening idea. Adv Chronic Kidney Dis. 2017;24(3):169-75. doi: 10.1053/j.ackd.2017.03.001.
Mehran R et al. Contrast-associated acute kidney injury. N Engl J Med. 2019;380(22):2146-55. doi: 10.1056/NEJMra1805256.
High rates of work-related trauma, PTSD in intern physicians
Work-related posttraumatic stress disorder is three times higher in interns than the general population, new research shows.
Investigators assessed PTSD in more than 1,100 physicians at the end of their internship year and found that a little over half reported work-related trauma exposure, and of these, 20% screened positive for PTSD.
Overall, 10% of participants screened positive for PTSD by the end of the internship year, compared with a 12-month PTSD prevalence of 3.6% in the general population.
“Work-related trauma exposure and PTSD are common and underdiscussed phenomena among intern physicians,” lead author Mary Vance, MD, assistant professor of psychiatry, Uniformed Services University of the Health Sciences, Bethesda, Md., said in an interview.
“I urge medical educators and policy makers to include this topic in their discussions about physician well-being and to implement effective interventions to mitigate the impact of work-related trauma and PTSD among physician trainees,” she said.
The study was published online June 8 in JAMA Network Open.
Burnout, depression, suicide
“Burnout, depression, and suicide are increasingly recognized as occupational mental health hazards among health care professionals, including physicians,” Dr. Vance said.
“However, in my professional experience as a physician and educator, despite observing anecdotal evidence among my peers and trainees that this is also an issue,” she added.
This gap prompted her “to investigate rates of work-related trauma exposure and PTSD among physicians.”
The researchers sent emails to 4,350 individuals during academic year 2018-2019, 2 months prior to starting internships. Of these, 2,129 agreed to participate and 1,134 (58.6% female, 61.6% non-Hispanic White; mean age, 27.52) completed the study.
Prior to beginning internship, participants completed a baseline survey that assessed demographic characteristics as well as medical education and psychological and psychosocial factors.
Participants completed follow-up surveys sent by email at 3, 6, 9, and 12 months of the internship year. The surveys assessed stressful life events, concern over perceived medical errors in the past 3 months, and number of hours worked over the past week.
At month 12, current PTSD and symptoms of depression and anxiety were also assessed using the Primary Care PTSD Screen for DSM-5, the 9-item Patient Health Questionnaire, and the Generalized Anxiety Disorder 7-item scale, respectively.
Participants were asked to self-report whether they ever had an episode of depression and to complete the Risky Families Questionnaire to assess if they had experienced childhood abuse, neglect, and family conflict. Additionally, they completed an 11-item scale developed specifically for the study regarding recent stressful events.
‘Crucible’ year
A total of 56.4% of respondents reported work-related trauma exposure, and among these, 19.0% screened positive for PTSD. One-tenth (10.8%) of the entire sample screened positive for PTSD by the end of internship year, which is three times higher than the 12-month prevalence of PTSD in the general population (3.6%), the authors noted.
Trauma exposure differed by specialty, ranging from 43.1% in anesthesiology to 72.4% in emergency medicine. Of the respondents in internal medicine, surgery, and medicine/pediatrics, 56.6%, 63.3%, and 71%, respectively, reported work-related trauma exposure.
Work-related PTSD also differed by specialty, ranging from 7.5% in ob.gyn. to 30.0% in pediatrics. Of respondents in internal medicine and family practice, 23.9% and 25.9%, respectively, reported work-related PTSD.
Dr. Vance called the intern year “a crucible, during which newly minted doctors receive intensive on-the-job training at the front lines of patient care [and] work long hours in rapidly shifting environments, often caring for critically ill patients.”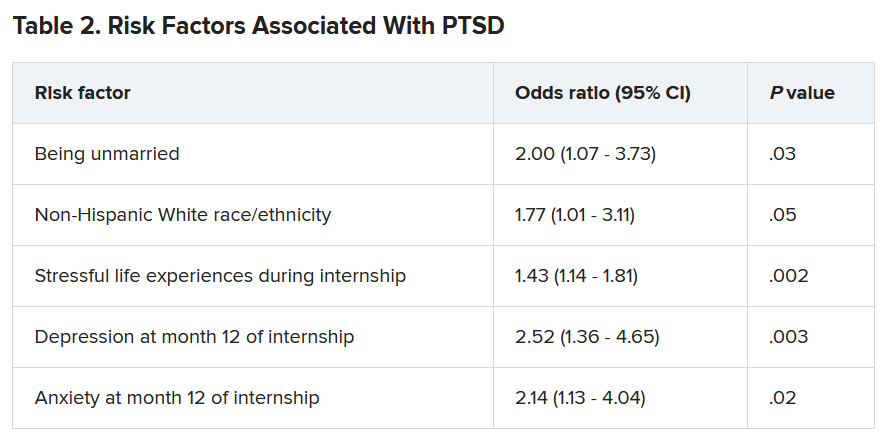
Work-related trauma exposure “is more likely to occur during this high-stress internship year than during the same year in the general population,” she said.
She noted that the “issue of workplace trauma and PTSD among health care workers became even more salient during the height of COVID,” adding that she expects it “to remain a pressure issue for healthcare workers in the post-COVID era.”
Call to action
Commenting on the study David A. Marcus, MD, chair, GME Physician Well-Being Committee, Northwell Health, New Hyde Park, N.Y., noted the study’s “relatively low response rate” is a “significant limitation” of the study.
An additional limitation is the lack of a baseline PTSD assessment, said Dr. Marcus, an assistant professor at Hofstra University, Hempstead, N.Y., who was not involved in the research.
Nevertheless, the “overall prevalence [of work-related PTSD] should serve as a call to action for physician leaders and for leaders in academic medicine,” he said.
Additionally, the study “reminds us that trauma-informed care should be an essential part of mental health support services provided to trainees and to physicians in general,” Dr. Marcus stated.
Also commenting on the study, Lotte N. Dyrbye, MD, professor of medicine and medical education, Mayo Clinic, Rochester, Minn., agreed.
“Organizational strategies should include system-level interventions to reduce the risk of frightening, horrible, or traumatic events from occurring in the workplace in the first place, as well as faculty development efforts to upskill teaching faculty in their ability to support trainees when such events do occur,” she said.
These approaches “should coincide with organizational efforts to support individual trainees by providing adequate time off after traumatic events, ensuring trainees can access affordable mental healthcare, and reducing other barriers to appropriate help-seeking, such as stigma, and efforts to build a culture of well-being,” suggested Dr. Dyrbye, who is codirector of the Mayo Clinic Program on Physician Wellbeing and was not involved in the study.
The study was supported by grants from the Blue Cross Blue Shield Foundation of Michigan and National Institutes of Health. Dr. Vance and coauthors, Dr. Marcus, and Dr. Dyrbye reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Work-related posttraumatic stress disorder is three times higher in interns than the general population, new research shows.
Investigators assessed PTSD in more than 1,100 physicians at the end of their internship year and found that a little over half reported work-related trauma exposure, and of these, 20% screened positive for PTSD.
Overall, 10% of participants screened positive for PTSD by the end of the internship year, compared with a 12-month PTSD prevalence of 3.6% in the general population.
“Work-related trauma exposure and PTSD are common and underdiscussed phenomena among intern physicians,” lead author Mary Vance, MD, assistant professor of psychiatry, Uniformed Services University of the Health Sciences, Bethesda, Md., said in an interview.
“I urge medical educators and policy makers to include this topic in their discussions about physician well-being and to implement effective interventions to mitigate the impact of work-related trauma and PTSD among physician trainees,” she said.
The study was published online June 8 in JAMA Network Open.
Burnout, depression, suicide
“Burnout, depression, and suicide are increasingly recognized as occupational mental health hazards among health care professionals, including physicians,” Dr. Vance said.
“However, in my professional experience as a physician and educator, despite observing anecdotal evidence among my peers and trainees that this is also an issue,” she added.
This gap prompted her “to investigate rates of work-related trauma exposure and PTSD among physicians.”
The researchers sent emails to 4,350 individuals during academic year 2018-2019, 2 months prior to starting internships. Of these, 2,129 agreed to participate and 1,134 (58.6% female, 61.6% non-Hispanic White; mean age, 27.52) completed the study.
Prior to beginning internship, participants completed a baseline survey that assessed demographic characteristics as well as medical education and psychological and psychosocial factors.
Participants completed follow-up surveys sent by email at 3, 6, 9, and 12 months of the internship year. The surveys assessed stressful life events, concern over perceived medical errors in the past 3 months, and number of hours worked over the past week.
At month 12, current PTSD and symptoms of depression and anxiety were also assessed using the Primary Care PTSD Screen for DSM-5, the 9-item Patient Health Questionnaire, and the Generalized Anxiety Disorder 7-item scale, respectively.
Participants were asked to self-report whether they ever had an episode of depression and to complete the Risky Families Questionnaire to assess if they had experienced childhood abuse, neglect, and family conflict. Additionally, they completed an 11-item scale developed specifically for the study regarding recent stressful events.
‘Crucible’ year
A total of 56.4% of respondents reported work-related trauma exposure, and among these, 19.0% screened positive for PTSD. One-tenth (10.8%) of the entire sample screened positive for PTSD by the end of internship year, which is three times higher than the 12-month prevalence of PTSD in the general population (3.6%), the authors noted.
Trauma exposure differed by specialty, ranging from 43.1% in anesthesiology to 72.4% in emergency medicine. Of the respondents in internal medicine, surgery, and medicine/pediatrics, 56.6%, 63.3%, and 71%, respectively, reported work-related trauma exposure.
Work-related PTSD also differed by specialty, ranging from 7.5% in ob.gyn. to 30.0% in pediatrics. Of respondents in internal medicine and family practice, 23.9% and 25.9%, respectively, reported work-related PTSD.
Dr. Vance called the intern year “a crucible, during which newly minted doctors receive intensive on-the-job training at the front lines of patient care [and] work long hours in rapidly shifting environments, often caring for critically ill patients.”
Work-related trauma exposure “is more likely to occur during this high-stress internship year than during the same year in the general population,” she said.
She noted that the “issue of workplace trauma and PTSD among health care workers became even more salient during the height of COVID,” adding that she expects it “to remain a pressure issue for healthcare workers in the post-COVID era.”
Call to action
Commenting on the study David A. Marcus, MD, chair, GME Physician Well-Being Committee, Northwell Health, New Hyde Park, N.Y., noted the study’s “relatively low response rate” is a “significant limitation” of the study.
An additional limitation is the lack of a baseline PTSD assessment, said Dr. Marcus, an assistant professor at Hofstra University, Hempstead, N.Y., who was not involved in the research.
Nevertheless, the “overall prevalence [of work-related PTSD] should serve as a call to action for physician leaders and for leaders in academic medicine,” he said.
Additionally, the study “reminds us that trauma-informed care should be an essential part of mental health support services provided to trainees and to physicians in general,” Dr. Marcus stated.
Also commenting on the study, Lotte N. Dyrbye, MD, professor of medicine and medical education, Mayo Clinic, Rochester, Minn., agreed.
“Organizational strategies should include system-level interventions to reduce the risk of frightening, horrible, or traumatic events from occurring in the workplace in the first place, as well as faculty development efforts to upskill teaching faculty in their ability to support trainees when such events do occur,” she said.
These approaches “should coincide with organizational efforts to support individual trainees by providing adequate time off after traumatic events, ensuring trainees can access affordable mental healthcare, and reducing other barriers to appropriate help-seeking, such as stigma, and efforts to build a culture of well-being,” suggested Dr. Dyrbye, who is codirector of the Mayo Clinic Program on Physician Wellbeing and was not involved in the study.
The study was supported by grants from the Blue Cross Blue Shield Foundation of Michigan and National Institutes of Health. Dr. Vance and coauthors, Dr. Marcus, and Dr. Dyrbye reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Work-related posttraumatic stress disorder is three times higher in interns than the general population, new research shows.
Investigators assessed PTSD in more than 1,100 physicians at the end of their internship year and found that a little over half reported work-related trauma exposure, and of these, 20% screened positive for PTSD.
Overall, 10% of participants screened positive for PTSD by the end of the internship year, compared with a 12-month PTSD prevalence of 3.6% in the general population.
“Work-related trauma exposure and PTSD are common and underdiscussed phenomena among intern physicians,” lead author Mary Vance, MD, assistant professor of psychiatry, Uniformed Services University of the Health Sciences, Bethesda, Md., said in an interview.
“I urge medical educators and policy makers to include this topic in their discussions about physician well-being and to implement effective interventions to mitigate the impact of work-related trauma and PTSD among physician trainees,” she said.
The study was published online June 8 in JAMA Network Open.
Burnout, depression, suicide
“Burnout, depression, and suicide are increasingly recognized as occupational mental health hazards among health care professionals, including physicians,” Dr. Vance said.
“However, in my professional experience as a physician and educator, despite observing anecdotal evidence among my peers and trainees that this is also an issue,” she added.
This gap prompted her “to investigate rates of work-related trauma exposure and PTSD among physicians.”
The researchers sent emails to 4,350 individuals during academic year 2018-2019, 2 months prior to starting internships. Of these, 2,129 agreed to participate and 1,134 (58.6% female, 61.6% non-Hispanic White; mean age, 27.52) completed the study.
Prior to beginning internship, participants completed a baseline survey that assessed demographic characteristics as well as medical education and psychological and psychosocial factors.
Participants completed follow-up surveys sent by email at 3, 6, 9, and 12 months of the internship year. The surveys assessed stressful life events, concern over perceived medical errors in the past 3 months, and number of hours worked over the past week.
At month 12, current PTSD and symptoms of depression and anxiety were also assessed using the Primary Care PTSD Screen for DSM-5, the 9-item Patient Health Questionnaire, and the Generalized Anxiety Disorder 7-item scale, respectively.
Participants were asked to self-report whether they ever had an episode of depression and to complete the Risky Families Questionnaire to assess if they had experienced childhood abuse, neglect, and family conflict. Additionally, they completed an 11-item scale developed specifically for the study regarding recent stressful events.
‘Crucible’ year
A total of 56.4% of respondents reported work-related trauma exposure, and among these, 19.0% screened positive for PTSD. One-tenth (10.8%) of the entire sample screened positive for PTSD by the end of internship year, which is three times higher than the 12-month prevalence of PTSD in the general population (3.6%), the authors noted.
Trauma exposure differed by specialty, ranging from 43.1% in anesthesiology to 72.4% in emergency medicine. Of the respondents in internal medicine, surgery, and medicine/pediatrics, 56.6%, 63.3%, and 71%, respectively, reported work-related trauma exposure.
Work-related PTSD also differed by specialty, ranging from 7.5% in ob.gyn. to 30.0% in pediatrics. Of respondents in internal medicine and family practice, 23.9% and 25.9%, respectively, reported work-related PTSD.
Dr. Vance called the intern year “a crucible, during which newly minted doctors receive intensive on-the-job training at the front lines of patient care [and] work long hours in rapidly shifting environments, often caring for critically ill patients.”
Work-related trauma exposure “is more likely to occur during this high-stress internship year than during the same year in the general population,” she said.
She noted that the “issue of workplace trauma and PTSD among health care workers became even more salient during the height of COVID,” adding that she expects it “to remain a pressure issue for healthcare workers in the post-COVID era.”
Call to action
Commenting on the study David A. Marcus, MD, chair, GME Physician Well-Being Committee, Northwell Health, New Hyde Park, N.Y., noted the study’s “relatively low response rate” is a “significant limitation” of the study.
An additional limitation is the lack of a baseline PTSD assessment, said Dr. Marcus, an assistant professor at Hofstra University, Hempstead, N.Y., who was not involved in the research.
Nevertheless, the “overall prevalence [of work-related PTSD] should serve as a call to action for physician leaders and for leaders in academic medicine,” he said.
Additionally, the study “reminds us that trauma-informed care should be an essential part of mental health support services provided to trainees and to physicians in general,” Dr. Marcus stated.
Also commenting on the study, Lotte N. Dyrbye, MD, professor of medicine and medical education, Mayo Clinic, Rochester, Minn., agreed.
“Organizational strategies should include system-level interventions to reduce the risk of frightening, horrible, or traumatic events from occurring in the workplace in the first place, as well as faculty development efforts to upskill teaching faculty in their ability to support trainees when such events do occur,” she said.
These approaches “should coincide with organizational efforts to support individual trainees by providing adequate time off after traumatic events, ensuring trainees can access affordable mental healthcare, and reducing other barriers to appropriate help-seeking, such as stigma, and efforts to build a culture of well-being,” suggested Dr. Dyrbye, who is codirector of the Mayo Clinic Program on Physician Wellbeing and was not involved in the study.
The study was supported by grants from the Blue Cross Blue Shield Foundation of Michigan and National Institutes of Health. Dr. Vance and coauthors, Dr. Marcus, and Dr. Dyrbye reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Reflections on 10 years of hospitalist productivity
Successful programs will recruit lifelong learners
The workload of individual hospitalists has long been a hot-button issue. In a 2013 survey of hospitalists, 40% felt workloads were unsafe on a monthly basis, and 22% reported ordering unnecessary testing or procedures because of time pressure.1 In a 2014 analysis of over 20,000 admissions to an academic hospital medicine service, increasing workload led to increased length of stay and cost per case.2 Although these studies suggest a “sweet spot” for hospitalist workload, many groups face constant pressure to increase revenue.
Over the past decade there has been a significant change in how hospital medicine programs are financed. In the 2010 State of Hospital Medicine (SoHM), the median financial support per physician hospitalist in adult hospital medicine groups (HMGs) was $98,253. By the 2020 SoHM, the financial support was $198,750, an increase of $100,497 in just 10 years. When this is combined with the explosive growth in the number of hospitalists, there is one inescapable conclusion – hospital medicine is expensive.
Over this same 10 years, net collections per hospitalist grew from $194,440 in 2010 to $216,779 in 2020, an increase of $22,339. The increase was caused by higher collections per encounter, not more encounters. Additionally, median compensation for adult/internal medicine hospitalists increased over the same period from $215,000 to $307,336, an increase of $92,336, or 43%. That is an increase of 3.7% per year, more than twice the rate of inflation or wage growth in the general economy over the same period. About 75% of this increase was funded by hospital support. It is clear – health care systems continue to find value in investing in hospitalists and hospital medicine programs.
With mounting costs for hospitals, there is pressure for the hospitalist model of care to change or for yearly billable encounters per hospitalist full-time equivalent to increase. Yet, the productivity of hospitalists, as measured by median billable encounters per year has remained flat. The 2010 SoHM listed median number of billable encounters per year for an internal medicine hospitalist as 2,230. In 2020, the number is 2,246 – a trivial 0.7% increase per decade, what amounts to a rounding error. There has been wiggle up and down over the years, but I suspect these are not trends but noise.
So the question is why. I think it is partly because hospital medicine leaders together with the leaders of their health care systems seem to be reaching an equilibrium. Productivity will always remain an expectation. This expectation will vary based on local circumstances. But for many HMGs, the days when productivity is pushed as the primary objective seem to be disappearing. Most hospital leaders seem to now understand that high productivity can be detrimental to other program goals.
But if productivity is flat, do 40% of hospitalists still feel they are providing unsafe care on a monthly basis? Without another study we don’t know, but here are some reasons why I’m hopeful. First, the hospitalist workforce is more experienced than 10 years ago and may be more efficient. Second, hospital medicine groups are larger and are therefore enabled to schedule more flexibly or enact jeopardy systems to level out workload on busy days. And lastly, hospitalists who feel they are providing unsafe care find greener pastures. The 2010 SoHM reported adult hospital medicine programs had a median 14.3% turnover rate. The 2020 SoHM turnover was 10.9%. While this is up from 2018 (7.4%) and 2016 (6.9%), the general trend is down.
Additionally, we all need to consider the possibility that there will be a disruptive innovation that will allow greater productivity for individual hospitalists while maintaining value. It is apparent the EHR is not yet that breakthrough. We all need to keep our eyes open, stay flexible, and be prepared to meet evolving demands on our programs.
We will see constant demands on hospitalists. But I’m hopeful that going forward expectations will increasingly shift away from simply working harder and seeing more patients, toward goals related to improving performance. Training programs generally produce excellent clinicians, but they often do not equip physicians to be excellent hospitalists. Successful hospital medicine programs will recruit lifelong learners and career hospitalists who are flexible and willing to innovate and adapt. The best programs will have structures in place to help excellent clinicians mature into the role of excellent hospitalists, and leaders that create and foster an environment of excellence.
Discover more 2020 SoHM Report data at www.hospitalmedicine.org/sohm.
Dr. Frederickson is medical director, hospital medicine and palliative care, at CHI Health, Omaha, Neb., and assistant professor at Creighton University, Omaha.
References
1. Michtalik HJ et al. Impact of Attending Physician Workload on Patient Care: A Survey of Hospitalists. JAMA Intern Med. 2013;173(5):375-7. doi: 10.1001/jamainternmed.2013.1864.
2. Elliott DJ et al. Effect of Hospitalist Workload on the Quality and Efficiency of Care. JAMA Intern Med. 2014;174(5):786-93. doi: 10.1001/jamainternmed.2014.300.
Successful programs will recruit lifelong learners
Successful programs will recruit lifelong learners
The workload of individual hospitalists has long been a hot-button issue. In a 2013 survey of hospitalists, 40% felt workloads were unsafe on a monthly basis, and 22% reported ordering unnecessary testing or procedures because of time pressure.1 In a 2014 analysis of over 20,000 admissions to an academic hospital medicine service, increasing workload led to increased length of stay and cost per case.2 Although these studies suggest a “sweet spot” for hospitalist workload, many groups face constant pressure to increase revenue.
Over the past decade there has been a significant change in how hospital medicine programs are financed. In the 2010 State of Hospital Medicine (SoHM), the median financial support per physician hospitalist in adult hospital medicine groups (HMGs) was $98,253. By the 2020 SoHM, the financial support was $198,750, an increase of $100,497 in just 10 years. When this is combined with the explosive growth in the number of hospitalists, there is one inescapable conclusion – hospital medicine is expensive.
Over this same 10 years, net collections per hospitalist grew from $194,440 in 2010 to $216,779 in 2020, an increase of $22,339. The increase was caused by higher collections per encounter, not more encounters. Additionally, median compensation for adult/internal medicine hospitalists increased over the same period from $215,000 to $307,336, an increase of $92,336, or 43%. That is an increase of 3.7% per year, more than twice the rate of inflation or wage growth in the general economy over the same period. About 75% of this increase was funded by hospital support. It is clear – health care systems continue to find value in investing in hospitalists and hospital medicine programs.
With mounting costs for hospitals, there is pressure for the hospitalist model of care to change or for yearly billable encounters per hospitalist full-time equivalent to increase. Yet, the productivity of hospitalists, as measured by median billable encounters per year has remained flat. The 2010 SoHM listed median number of billable encounters per year for an internal medicine hospitalist as 2,230. In 2020, the number is 2,246 – a trivial 0.7% increase per decade, what amounts to a rounding error. There has been wiggle up and down over the years, but I suspect these are not trends but noise.
So the question is why. I think it is partly because hospital medicine leaders together with the leaders of their health care systems seem to be reaching an equilibrium. Productivity will always remain an expectation. This expectation will vary based on local circumstances. But for many HMGs, the days when productivity is pushed as the primary objective seem to be disappearing. Most hospital leaders seem to now understand that high productivity can be detrimental to other program goals.
But if productivity is flat, do 40% of hospitalists still feel they are providing unsafe care on a monthly basis? Without another study we don’t know, but here are some reasons why I’m hopeful. First, the hospitalist workforce is more experienced than 10 years ago and may be more efficient. Second, hospital medicine groups are larger and are therefore enabled to schedule more flexibly or enact jeopardy systems to level out workload on busy days. And lastly, hospitalists who feel they are providing unsafe care find greener pastures. The 2010 SoHM reported adult hospital medicine programs had a median 14.3% turnover rate. The 2020 SoHM turnover was 10.9%. While this is up from 2018 (7.4%) and 2016 (6.9%), the general trend is down.
Additionally, we all need to consider the possibility that there will be a disruptive innovation that will allow greater productivity for individual hospitalists while maintaining value. It is apparent the EHR is not yet that breakthrough. We all need to keep our eyes open, stay flexible, and be prepared to meet evolving demands on our programs.
We will see constant demands on hospitalists. But I’m hopeful that going forward expectations will increasingly shift away from simply working harder and seeing more patients, toward goals related to improving performance. Training programs generally produce excellent clinicians, but they often do not equip physicians to be excellent hospitalists. Successful hospital medicine programs will recruit lifelong learners and career hospitalists who are flexible and willing to innovate and adapt. The best programs will have structures in place to help excellent clinicians mature into the role of excellent hospitalists, and leaders that create and foster an environment of excellence.
Discover more 2020 SoHM Report data at www.hospitalmedicine.org/sohm.
Dr. Frederickson is medical director, hospital medicine and palliative care, at CHI Health, Omaha, Neb., and assistant professor at Creighton University, Omaha.
References
1. Michtalik HJ et al. Impact of Attending Physician Workload on Patient Care: A Survey of Hospitalists. JAMA Intern Med. 2013;173(5):375-7. doi: 10.1001/jamainternmed.2013.1864.
2. Elliott DJ et al. Effect of Hospitalist Workload on the Quality and Efficiency of Care. JAMA Intern Med. 2014;174(5):786-93. doi: 10.1001/jamainternmed.2014.300.
The workload of individual hospitalists has long been a hot-button issue. In a 2013 survey of hospitalists, 40% felt workloads were unsafe on a monthly basis, and 22% reported ordering unnecessary testing or procedures because of time pressure.1 In a 2014 analysis of over 20,000 admissions to an academic hospital medicine service, increasing workload led to increased length of stay and cost per case.2 Although these studies suggest a “sweet spot” for hospitalist workload, many groups face constant pressure to increase revenue.
Over the past decade there has been a significant change in how hospital medicine programs are financed. In the 2010 State of Hospital Medicine (SoHM), the median financial support per physician hospitalist in adult hospital medicine groups (HMGs) was $98,253. By the 2020 SoHM, the financial support was $198,750, an increase of $100,497 in just 10 years. When this is combined with the explosive growth in the number of hospitalists, there is one inescapable conclusion – hospital medicine is expensive.
Over this same 10 years, net collections per hospitalist grew from $194,440 in 2010 to $216,779 in 2020, an increase of $22,339. The increase was caused by higher collections per encounter, not more encounters. Additionally, median compensation for adult/internal medicine hospitalists increased over the same period from $215,000 to $307,336, an increase of $92,336, or 43%. That is an increase of 3.7% per year, more than twice the rate of inflation or wage growth in the general economy over the same period. About 75% of this increase was funded by hospital support. It is clear – health care systems continue to find value in investing in hospitalists and hospital medicine programs.
With mounting costs for hospitals, there is pressure for the hospitalist model of care to change or for yearly billable encounters per hospitalist full-time equivalent to increase. Yet, the productivity of hospitalists, as measured by median billable encounters per year has remained flat. The 2010 SoHM listed median number of billable encounters per year for an internal medicine hospitalist as 2,230. In 2020, the number is 2,246 – a trivial 0.7% increase per decade, what amounts to a rounding error. There has been wiggle up and down over the years, but I suspect these are not trends but noise.
So the question is why. I think it is partly because hospital medicine leaders together with the leaders of their health care systems seem to be reaching an equilibrium. Productivity will always remain an expectation. This expectation will vary based on local circumstances. But for many HMGs, the days when productivity is pushed as the primary objective seem to be disappearing. Most hospital leaders seem to now understand that high productivity can be detrimental to other program goals.
But if productivity is flat, do 40% of hospitalists still feel they are providing unsafe care on a monthly basis? Without another study we don’t know, but here are some reasons why I’m hopeful. First, the hospitalist workforce is more experienced than 10 years ago and may be more efficient. Second, hospital medicine groups are larger and are therefore enabled to schedule more flexibly or enact jeopardy systems to level out workload on busy days. And lastly, hospitalists who feel they are providing unsafe care find greener pastures. The 2010 SoHM reported adult hospital medicine programs had a median 14.3% turnover rate. The 2020 SoHM turnover was 10.9%. While this is up from 2018 (7.4%) and 2016 (6.9%), the general trend is down.
Additionally, we all need to consider the possibility that there will be a disruptive innovation that will allow greater productivity for individual hospitalists while maintaining value. It is apparent the EHR is not yet that breakthrough. We all need to keep our eyes open, stay flexible, and be prepared to meet evolving demands on our programs.
We will see constant demands on hospitalists. But I’m hopeful that going forward expectations will increasingly shift away from simply working harder and seeing more patients, toward goals related to improving performance. Training programs generally produce excellent clinicians, but they often do not equip physicians to be excellent hospitalists. Successful hospital medicine programs will recruit lifelong learners and career hospitalists who are flexible and willing to innovate and adapt. The best programs will have structures in place to help excellent clinicians mature into the role of excellent hospitalists, and leaders that create and foster an environment of excellence.
Discover more 2020 SoHM Report data at www.hospitalmedicine.org/sohm.
Dr. Frederickson is medical director, hospital medicine and palliative care, at CHI Health, Omaha, Neb., and assistant professor at Creighton University, Omaha.
References
1. Michtalik HJ et al. Impact of Attending Physician Workload on Patient Care: A Survey of Hospitalists. JAMA Intern Med. 2013;173(5):375-7. doi: 10.1001/jamainternmed.2013.1864.
2. Elliott DJ et al. Effect of Hospitalist Workload on the Quality and Efficiency of Care. JAMA Intern Med. 2014;174(5):786-93. doi: 10.1001/jamainternmed.2014.300.
Safety-net burden linked with poorer inpatient cirrhosis outcomes
Patients with cirrhosis treated at hospitals with the highest safety-net burden, defined by their proportion of Medicaid or uninsured patients, had a 5% higher mortality rate than patients who were treated at hospitals with the lowest burden, according to a study of over 300,000 patients.
The study, which was published in the Journal of Clinical Gastroenterology, analyzed inpatient data from the National Inpatient Sample (NIS) database focusing on a 4-year time span between 2012 and 2016. The hospitals were categorized by safety-net burden, which was defined as having either a high, medium, or low number of uninsured patients or patients with Medicaid.
This is the first-known study to evaluate the impact of a hospital’s safety-net burden on hospitalization outcomes in cirrhosis patients, wrote authors Robert J. Wong, MD, MS, of Stanford (Calif.) University and Grishma Hirode, MAS, of the University of Toronto. Previous studies have shown that safety-net hospitals, especially those with a high safety-net burden, have poorer patient outcomes. These hospitals also serve a patient population that is at high risk for chronic liver disease and cirrhosis.
The new analysis included 322,944 individual hospitalizations of patients with cirrhosis. Of these, 57.8% were male, 63.7% were White, 9.9% were Black, and 15.6% were Hispanic. In terms of safety-net burden, 107,446 hospitalizations were at high-burden hospitals, 103,508 were at medium-burden hospitals, and 111,990 hospitalizations were at low-burden hospitals.
Overall, cirrhosis-related hospitalizations in hospitals with the highest burden were found to have significantly greater odds of in-hospital mortality than the lowest tertile hospitals (odds ratio, 1.05, P = .044). The patients were also younger (mean age, 56.7 years vs. 59.8 years in low-burden hospitals). They also had a higher proportion of male patients, minority patients, Hispanic patients, and patients with Medicaid or no insurance.
The odds of hospitalization in the highest tertile hospitals were found to be significantly higher, compared with the middle and lowest tertiles for Blacks and Hispanics, compared with Whites (OR 1.26 and OR 1.63, respectively). Black patients (OR, 1.26; 95%CI, 1.17-1.35; P < .001) and Hispanic patients (OR, 1.63; 95% CI, 1.50-1.78; P< .001) were more likely to be admitted for care at high-burden hospitals (26% to 54%). In-hospital mortality rates among all hospitalizations were 5.95% and the rate did not significantly differ by hospital burden status.
“Despite adjusting for safety-net burden, our study continued to demonstrate ethnic disparities in in-hospital mortality among cirrhosis-related hospitalizations,” the researchers wrote. Overall, the odds of in-hospital mortality were 27% higher in Black patients as compared with White patients.
However, significantly lower mortality was observed in Hispanic patients as compared with White patients (4.9% vs. 6.0%, P < .001), but why this occurred was not entirely clear. “Hispanic patients may be more likely to have NASH [nonalcoholic steatohepatitis]-related cirrhosis, which generally has a slower disease progression, compared with [hepatitis C virus] or alcoholic cirrhosis. As such, it is likely that NASH-cirrhosis Hispanic patients had less severe disease at presentation,” the researchers wrote.
Study design has limitations, but shows concerning trends
The study findings were limited by several factors including the inability to show causality based on the observational study design and cross-sectional nature of the database, the researchers said. The NIS database records individual hospitalizations, not individual patient data which means that it may include repeat hospitalizations from the same patient. In addition, the study was limited by a lack of data on outpatient cirrhosis outcomes and non–liver-related comorbidities.
However, the finding that ethnic minorities with cirrhosis were significantly more likely to be hospitalized in high safety-net hospitals than White patients is concerning, and more research is needed, they said.
“These observations highlight that, while disparities in resources and health care delivery inherent to safety-net health systems may partly explain and provide opportunities to improve cirrhosis hospitalization care, they alone do not explain all of the ethnic disparities in cirrhosis outcomes observed,” they concluded.
The current study was important to conduct at this time because rates of cirrhosis are on the rise, Michael Volk, MD, of Loma Linda (Calif.) University Health, said in an interview. “Millions of patients receive care in safety-net hospitals across the country.”
Dr. Volk said that he was not surprised by the overall outcomes. “Unfortunately, I expected that patient outcomes would be worse at safety-net hospitals than wealthier hospitals. However, I was surprised that Blacks had higher in-hospital mortality than Whites, even after adjusting for the hospital.”
Dr. Volk echoed the study’s stated limitation of the lack of data to address disparities.
“Additional research is needed to determine whether the higher in-hospital mortality among Blacks is related to biological differences such as differential rates of disease progression, or social differences such as access to outpatient care,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Volk had no relevant financial conflicts to disclose.
Patients with cirrhosis treated at hospitals with the highest safety-net burden, defined by their proportion of Medicaid or uninsured patients, had a 5% higher mortality rate than patients who were treated at hospitals with the lowest burden, according to a study of over 300,000 patients.
The study, which was published in the Journal of Clinical Gastroenterology, analyzed inpatient data from the National Inpatient Sample (NIS) database focusing on a 4-year time span between 2012 and 2016. The hospitals were categorized by safety-net burden, which was defined as having either a high, medium, or low number of uninsured patients or patients with Medicaid.
This is the first-known study to evaluate the impact of a hospital’s safety-net burden on hospitalization outcomes in cirrhosis patients, wrote authors Robert J. Wong, MD, MS, of Stanford (Calif.) University and Grishma Hirode, MAS, of the University of Toronto. Previous studies have shown that safety-net hospitals, especially those with a high safety-net burden, have poorer patient outcomes. These hospitals also serve a patient population that is at high risk for chronic liver disease and cirrhosis.
The new analysis included 322,944 individual hospitalizations of patients with cirrhosis. Of these, 57.8% were male, 63.7% were White, 9.9% were Black, and 15.6% were Hispanic. In terms of safety-net burden, 107,446 hospitalizations were at high-burden hospitals, 103,508 were at medium-burden hospitals, and 111,990 hospitalizations were at low-burden hospitals.
Overall, cirrhosis-related hospitalizations in hospitals with the highest burden were found to have significantly greater odds of in-hospital mortality than the lowest tertile hospitals (odds ratio, 1.05, P = .044). The patients were also younger (mean age, 56.7 years vs. 59.8 years in low-burden hospitals). They also had a higher proportion of male patients, minority patients, Hispanic patients, and patients with Medicaid or no insurance.
The odds of hospitalization in the highest tertile hospitals were found to be significantly higher, compared with the middle and lowest tertiles for Blacks and Hispanics, compared with Whites (OR 1.26 and OR 1.63, respectively). Black patients (OR, 1.26; 95%CI, 1.17-1.35; P < .001) and Hispanic patients (OR, 1.63; 95% CI, 1.50-1.78; P< .001) were more likely to be admitted for care at high-burden hospitals (26% to 54%). In-hospital mortality rates among all hospitalizations were 5.95% and the rate did not significantly differ by hospital burden status.
“Despite adjusting for safety-net burden, our study continued to demonstrate ethnic disparities in in-hospital mortality among cirrhosis-related hospitalizations,” the researchers wrote. Overall, the odds of in-hospital mortality were 27% higher in Black patients as compared with White patients.
However, significantly lower mortality was observed in Hispanic patients as compared with White patients (4.9% vs. 6.0%, P < .001), but why this occurred was not entirely clear. “Hispanic patients may be more likely to have NASH [nonalcoholic steatohepatitis]-related cirrhosis, which generally has a slower disease progression, compared with [hepatitis C virus] or alcoholic cirrhosis. As such, it is likely that NASH-cirrhosis Hispanic patients had less severe disease at presentation,” the researchers wrote.
Study design has limitations, but shows concerning trends
The study findings were limited by several factors including the inability to show causality based on the observational study design and cross-sectional nature of the database, the researchers said. The NIS database records individual hospitalizations, not individual patient data which means that it may include repeat hospitalizations from the same patient. In addition, the study was limited by a lack of data on outpatient cirrhosis outcomes and non–liver-related comorbidities.
However, the finding that ethnic minorities with cirrhosis were significantly more likely to be hospitalized in high safety-net hospitals than White patients is concerning, and more research is needed, they said.
“These observations highlight that, while disparities in resources and health care delivery inherent to safety-net health systems may partly explain and provide opportunities to improve cirrhosis hospitalization care, they alone do not explain all of the ethnic disparities in cirrhosis outcomes observed,” they concluded.
The current study was important to conduct at this time because rates of cirrhosis are on the rise, Michael Volk, MD, of Loma Linda (Calif.) University Health, said in an interview. “Millions of patients receive care in safety-net hospitals across the country.”
Dr. Volk said that he was not surprised by the overall outcomes. “Unfortunately, I expected that patient outcomes would be worse at safety-net hospitals than wealthier hospitals. However, I was surprised that Blacks had higher in-hospital mortality than Whites, even after adjusting for the hospital.”
Dr. Volk echoed the study’s stated limitation of the lack of data to address disparities.
“Additional research is needed to determine whether the higher in-hospital mortality among Blacks is related to biological differences such as differential rates of disease progression, or social differences such as access to outpatient care,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Volk had no relevant financial conflicts to disclose.
Patients with cirrhosis treated at hospitals with the highest safety-net burden, defined by their proportion of Medicaid or uninsured patients, had a 5% higher mortality rate than patients who were treated at hospitals with the lowest burden, according to a study of over 300,000 patients.
The study, which was published in the Journal of Clinical Gastroenterology, analyzed inpatient data from the National Inpatient Sample (NIS) database focusing on a 4-year time span between 2012 and 2016. The hospitals were categorized by safety-net burden, which was defined as having either a high, medium, or low number of uninsured patients or patients with Medicaid.
This is the first-known study to evaluate the impact of a hospital’s safety-net burden on hospitalization outcomes in cirrhosis patients, wrote authors Robert J. Wong, MD, MS, of Stanford (Calif.) University and Grishma Hirode, MAS, of the University of Toronto. Previous studies have shown that safety-net hospitals, especially those with a high safety-net burden, have poorer patient outcomes. These hospitals also serve a patient population that is at high risk for chronic liver disease and cirrhosis.
The new analysis included 322,944 individual hospitalizations of patients with cirrhosis. Of these, 57.8% were male, 63.7% were White, 9.9% were Black, and 15.6% were Hispanic. In terms of safety-net burden, 107,446 hospitalizations were at high-burden hospitals, 103,508 were at medium-burden hospitals, and 111,990 hospitalizations were at low-burden hospitals.
Overall, cirrhosis-related hospitalizations in hospitals with the highest burden were found to have significantly greater odds of in-hospital mortality than the lowest tertile hospitals (odds ratio, 1.05, P = .044). The patients were also younger (mean age, 56.7 years vs. 59.8 years in low-burden hospitals). They also had a higher proportion of male patients, minority patients, Hispanic patients, and patients with Medicaid or no insurance.
The odds of hospitalization in the highest tertile hospitals were found to be significantly higher, compared with the middle and lowest tertiles for Blacks and Hispanics, compared with Whites (OR 1.26 and OR 1.63, respectively). Black patients (OR, 1.26; 95%CI, 1.17-1.35; P < .001) and Hispanic patients (OR, 1.63; 95% CI, 1.50-1.78; P< .001) were more likely to be admitted for care at high-burden hospitals (26% to 54%). In-hospital mortality rates among all hospitalizations were 5.95% and the rate did not significantly differ by hospital burden status.
“Despite adjusting for safety-net burden, our study continued to demonstrate ethnic disparities in in-hospital mortality among cirrhosis-related hospitalizations,” the researchers wrote. Overall, the odds of in-hospital mortality were 27% higher in Black patients as compared with White patients.
However, significantly lower mortality was observed in Hispanic patients as compared with White patients (4.9% vs. 6.0%, P < .001), but why this occurred was not entirely clear. “Hispanic patients may be more likely to have NASH [nonalcoholic steatohepatitis]-related cirrhosis, which generally has a slower disease progression, compared with [hepatitis C virus] or alcoholic cirrhosis. As such, it is likely that NASH-cirrhosis Hispanic patients had less severe disease at presentation,” the researchers wrote.
Study design has limitations, but shows concerning trends
The study findings were limited by several factors including the inability to show causality based on the observational study design and cross-sectional nature of the database, the researchers said. The NIS database records individual hospitalizations, not individual patient data which means that it may include repeat hospitalizations from the same patient. In addition, the study was limited by a lack of data on outpatient cirrhosis outcomes and non–liver-related comorbidities.
However, the finding that ethnic minorities with cirrhosis were significantly more likely to be hospitalized in high safety-net hospitals than White patients is concerning, and more research is needed, they said.
“These observations highlight that, while disparities in resources and health care delivery inherent to safety-net health systems may partly explain and provide opportunities to improve cirrhosis hospitalization care, they alone do not explain all of the ethnic disparities in cirrhosis outcomes observed,” they concluded.
The current study was important to conduct at this time because rates of cirrhosis are on the rise, Michael Volk, MD, of Loma Linda (Calif.) University Health, said in an interview. “Millions of patients receive care in safety-net hospitals across the country.”
Dr. Volk said that he was not surprised by the overall outcomes. “Unfortunately, I expected that patient outcomes would be worse at safety-net hospitals than wealthier hospitals. However, I was surprised that Blacks had higher in-hospital mortality than Whites, even after adjusting for the hospital.”
Dr. Volk echoed the study’s stated limitation of the lack of data to address disparities.
“Additional research is needed to determine whether the higher in-hospital mortality among Blacks is related to biological differences such as differential rates of disease progression, or social differences such as access to outpatient care,” he said.
The study received no outside funding. The researchers had no financial conflicts to disclose. Dr. Volk had no relevant financial conflicts to disclose.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY




















