User login
AVAHO
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]


Excess cancer deaths predicted as care is disrupted by COVID-19
The majority of patients who have cancer or are suspected of having cancer are not accessing healthcare services in the United Kingdom or the United States because of the COVID-19 pandemic, the first report of its kind estimates.
As a result, there will be an excess of deaths among patients who have cancer and multiple comorbidities in both countries during the current coronavirus emergency, the report warns.
The authors calculate that there will be 6,270 excess deaths among cancer patients 1 year from now in England and 33,890 excess deaths among cancer patients in the United States. (In the United States, the estimated excess number of deaths applies only to patients older than 40 years, they note.)
“The recorded underlying cause of these excess deaths may be cancer, COVID-19, or comorbidity (such as myocardial infarction),” Alvina Lai, PhD, University College London, United Kingdom, and colleagues observe.
“Our data have highlighted how cancer patients with multimorbidity are a particularly at-risk group during the current pandemic,” they emphasize.
The study was published on ResearchGate as a preprint and has not undergone peer review.
Commenting on the study on the UK Science Media Center, several experts emphasized the lack of peer review, noting that interpretation of these data needs to be further refined on the basis of that input. One expert suggested that there are “substantial uncertainties that this paper does not adequately communicate.” But others argued that this topic was important enough to warrant early release of the data.
Chris Bunce, PhD, University of Birmingham, United Kingdom, said this study represents “a highly valuable contribution.”
“It is universally accepted that early diagnosis and treatment and adherence to treatment regimens saves lives,” he pointed out.
“Therefore, these COVID-19-related impacts will cost lives,” Bunce said.
“And if this information is to influence cancer care and guide policy during the COVID-19 crisis, then it is important that the findings are disseminated and discussed immediately, warranting their release ahead of peer view,” he added.
In a Medscape UK commentary, oncologist Karol Sikora, MD, PhD, argues that “restarting cancer services can’t come soon enough.”
“Resonably Argued Numerical Estimate”
“It’s well known that there have been considerable changes in the provision of health care for many conditions, including cancers, as a result of all the measures to deal with the COVID-19 crisis,” said Kevin McConway, PhD, professor emeritus of applied statistics, the Open University, Milton Keynes, United Kingdom.
“It seems inevitable that there will be increased deaths in cancer patients if they are infected with the virus or because of changes in the health services available to them, and quite possibly also from socio-economic effects of the responses to the crisis,” he continued.
“This study is the first that I have seen that produces a reasonably argued numerical estimate of the number of excess deaths of people with cancer arising from these factors in the UK and the USA,” he added.
Declines in Urgent Referrals and Chemo Attendance
For the study, the team used DATA-CAN, the UK National Health Data Research Hub for Cancer, to assess weekly returns for urgent cancer referrals for early diagnosis and also chemotherapy attendances for hospitals in Leeds, London, and Northern Ireland going back to 2018.
The data revealed that there have been major declines in chemotherapy attendances. There has been, on average, a 60% decrease from prepandemic levels in eight hospitals in the three regions that were assessed.
Urgent cancer referrals have dropped by an average of 76% compared to prepandemic levels in the three regions.
On the conservative assumption that the COVID-19 pandemic will only affect patients with newly diagnosed cancer (incident cases), the researchers estimate that the proportion of the population affected by the emergency (PAE) is 40% and that the relative impact of the emergency (RIE) is 1.5.
PAE is a summary measure of exposure to the adverse health consequences of the emergency; RIE is a summary measure of the combined impact on mortality of infection, health service change, physical distancing, and economic downturn, the authors explain.
Comorbidities Common
“Comorbidities were common in people with cancer,” the study authors note. For example, more than one quarter of the study population had at least one comorbidity; more than 14% had two.
For incident cancers, the number of excess deaths steadily increased in conjunction with an increase in the number of comorbidities, such that more than 80% of deaths occurred in patients with one or more comorbidities.
“When considering both prevalent and incident cancers together with a COVID-19 PAE of 40%, we estimated 17,991 excess deaths at a RIE of 1.5; 78.1% of these deaths occur in patients with ≥1 comorbidities,” the authors report.
“The excess risk of death in people living with cancer during the COVID-19 emergency may be due not only to COVID-19 infection, but also to the unintended health consequences of changes in health service provision, the physical or psychological effects of social distancing, and economic upheaval,” they state.
“This is the first study demonstrating profound recent changes in cancer care delivery in multiple centers,” the authors observe.
Lai has disclosed no relevant financial relationships. Several coauthors have various relationships with industry, as listed in their article. The commentators have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The majority of patients who have cancer or are suspected of having cancer are not accessing healthcare services in the United Kingdom or the United States because of the COVID-19 pandemic, the first report of its kind estimates.
As a result, there will be an excess of deaths among patients who have cancer and multiple comorbidities in both countries during the current coronavirus emergency, the report warns.
The authors calculate that there will be 6,270 excess deaths among cancer patients 1 year from now in England and 33,890 excess deaths among cancer patients in the United States. (In the United States, the estimated excess number of deaths applies only to patients older than 40 years, they note.)
“The recorded underlying cause of these excess deaths may be cancer, COVID-19, or comorbidity (such as myocardial infarction),” Alvina Lai, PhD, University College London, United Kingdom, and colleagues observe.
“Our data have highlighted how cancer patients with multimorbidity are a particularly at-risk group during the current pandemic,” they emphasize.
The study was published on ResearchGate as a preprint and has not undergone peer review.
Commenting on the study on the UK Science Media Center, several experts emphasized the lack of peer review, noting that interpretation of these data needs to be further refined on the basis of that input. One expert suggested that there are “substantial uncertainties that this paper does not adequately communicate.” But others argued that this topic was important enough to warrant early release of the data.
Chris Bunce, PhD, University of Birmingham, United Kingdom, said this study represents “a highly valuable contribution.”
“It is universally accepted that early diagnosis and treatment and adherence to treatment regimens saves lives,” he pointed out.
“Therefore, these COVID-19-related impacts will cost lives,” Bunce said.
“And if this information is to influence cancer care and guide policy during the COVID-19 crisis, then it is important that the findings are disseminated and discussed immediately, warranting their release ahead of peer view,” he added.
In a Medscape UK commentary, oncologist Karol Sikora, MD, PhD, argues that “restarting cancer services can’t come soon enough.”
“Resonably Argued Numerical Estimate”
“It’s well known that there have been considerable changes in the provision of health care for many conditions, including cancers, as a result of all the measures to deal with the COVID-19 crisis,” said Kevin McConway, PhD, professor emeritus of applied statistics, the Open University, Milton Keynes, United Kingdom.
“It seems inevitable that there will be increased deaths in cancer patients if they are infected with the virus or because of changes in the health services available to them, and quite possibly also from socio-economic effects of the responses to the crisis,” he continued.
“This study is the first that I have seen that produces a reasonably argued numerical estimate of the number of excess deaths of people with cancer arising from these factors in the UK and the USA,” he added.
Declines in Urgent Referrals and Chemo Attendance
For the study, the team used DATA-CAN, the UK National Health Data Research Hub for Cancer, to assess weekly returns for urgent cancer referrals for early diagnosis and also chemotherapy attendances for hospitals in Leeds, London, and Northern Ireland going back to 2018.
The data revealed that there have been major declines in chemotherapy attendances. There has been, on average, a 60% decrease from prepandemic levels in eight hospitals in the three regions that were assessed.
Urgent cancer referrals have dropped by an average of 76% compared to prepandemic levels in the three regions.
On the conservative assumption that the COVID-19 pandemic will only affect patients with newly diagnosed cancer (incident cases), the researchers estimate that the proportion of the population affected by the emergency (PAE) is 40% and that the relative impact of the emergency (RIE) is 1.5.
PAE is a summary measure of exposure to the adverse health consequences of the emergency; RIE is a summary measure of the combined impact on mortality of infection, health service change, physical distancing, and economic downturn, the authors explain.
Comorbidities Common
“Comorbidities were common in people with cancer,” the study authors note. For example, more than one quarter of the study population had at least one comorbidity; more than 14% had two.
For incident cancers, the number of excess deaths steadily increased in conjunction with an increase in the number of comorbidities, such that more than 80% of deaths occurred in patients with one or more comorbidities.
“When considering both prevalent and incident cancers together with a COVID-19 PAE of 40%, we estimated 17,991 excess deaths at a RIE of 1.5; 78.1% of these deaths occur in patients with ≥1 comorbidities,” the authors report.
“The excess risk of death in people living with cancer during the COVID-19 emergency may be due not only to COVID-19 infection, but also to the unintended health consequences of changes in health service provision, the physical or psychological effects of social distancing, and economic upheaval,” they state.
“This is the first study demonstrating profound recent changes in cancer care delivery in multiple centers,” the authors observe.
Lai has disclosed no relevant financial relationships. Several coauthors have various relationships with industry, as listed in their article. The commentators have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
The majority of patients who have cancer or are suspected of having cancer are not accessing healthcare services in the United Kingdom or the United States because of the COVID-19 pandemic, the first report of its kind estimates.
As a result, there will be an excess of deaths among patients who have cancer and multiple comorbidities in both countries during the current coronavirus emergency, the report warns.
The authors calculate that there will be 6,270 excess deaths among cancer patients 1 year from now in England and 33,890 excess deaths among cancer patients in the United States. (In the United States, the estimated excess number of deaths applies only to patients older than 40 years, they note.)
“The recorded underlying cause of these excess deaths may be cancer, COVID-19, or comorbidity (such as myocardial infarction),” Alvina Lai, PhD, University College London, United Kingdom, and colleagues observe.
“Our data have highlighted how cancer patients with multimorbidity are a particularly at-risk group during the current pandemic,” they emphasize.
The study was published on ResearchGate as a preprint and has not undergone peer review.
Commenting on the study on the UK Science Media Center, several experts emphasized the lack of peer review, noting that interpretation of these data needs to be further refined on the basis of that input. One expert suggested that there are “substantial uncertainties that this paper does not adequately communicate.” But others argued that this topic was important enough to warrant early release of the data.
Chris Bunce, PhD, University of Birmingham, United Kingdom, said this study represents “a highly valuable contribution.”
“It is universally accepted that early diagnosis and treatment and adherence to treatment regimens saves lives,” he pointed out.
“Therefore, these COVID-19-related impacts will cost lives,” Bunce said.
“And if this information is to influence cancer care and guide policy during the COVID-19 crisis, then it is important that the findings are disseminated and discussed immediately, warranting their release ahead of peer view,” he added.
In a Medscape UK commentary, oncologist Karol Sikora, MD, PhD, argues that “restarting cancer services can’t come soon enough.”
“Resonably Argued Numerical Estimate”
“It’s well known that there have been considerable changes in the provision of health care for many conditions, including cancers, as a result of all the measures to deal with the COVID-19 crisis,” said Kevin McConway, PhD, professor emeritus of applied statistics, the Open University, Milton Keynes, United Kingdom.
“It seems inevitable that there will be increased deaths in cancer patients if they are infected with the virus or because of changes in the health services available to them, and quite possibly also from socio-economic effects of the responses to the crisis,” he continued.
“This study is the first that I have seen that produces a reasonably argued numerical estimate of the number of excess deaths of people with cancer arising from these factors in the UK and the USA,” he added.
Declines in Urgent Referrals and Chemo Attendance
For the study, the team used DATA-CAN, the UK National Health Data Research Hub for Cancer, to assess weekly returns for urgent cancer referrals for early diagnosis and also chemotherapy attendances for hospitals in Leeds, London, and Northern Ireland going back to 2018.
The data revealed that there have been major declines in chemotherapy attendances. There has been, on average, a 60% decrease from prepandemic levels in eight hospitals in the three regions that were assessed.
Urgent cancer referrals have dropped by an average of 76% compared to prepandemic levels in the three regions.
On the conservative assumption that the COVID-19 pandemic will only affect patients with newly diagnosed cancer (incident cases), the researchers estimate that the proportion of the population affected by the emergency (PAE) is 40% and that the relative impact of the emergency (RIE) is 1.5.
PAE is a summary measure of exposure to the adverse health consequences of the emergency; RIE is a summary measure of the combined impact on mortality of infection, health service change, physical distancing, and economic downturn, the authors explain.
Comorbidities Common
“Comorbidities were common in people with cancer,” the study authors note. For example, more than one quarter of the study population had at least one comorbidity; more than 14% had two.
For incident cancers, the number of excess deaths steadily increased in conjunction with an increase in the number of comorbidities, such that more than 80% of deaths occurred in patients with one or more comorbidities.
“When considering both prevalent and incident cancers together with a COVID-19 PAE of 40%, we estimated 17,991 excess deaths at a RIE of 1.5; 78.1% of these deaths occur in patients with ≥1 comorbidities,” the authors report.
“The excess risk of death in people living with cancer during the COVID-19 emergency may be due not only to COVID-19 infection, but also to the unintended health consequences of changes in health service provision, the physical or psychological effects of social distancing, and economic upheaval,” they state.
“This is the first study demonstrating profound recent changes in cancer care delivery in multiple centers,” the authors observe.
Lai has disclosed no relevant financial relationships. Several coauthors have various relationships with industry, as listed in their article. The commentators have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Antitumor treatment may increase risk of severe events in COVID-19 patients
Cancer patients who received antitumor treatment within 14 days of COVID-19 diagnosis had an increased risk of severe events, according to data from three hospitals in Wuhan.
Patients with patchy consolidation at hospital admission also had an increased risk of severe events, defined as ICU admission, mechanical ventilation, or death.
However, these findings are limited by the small number of patients studied and the retrospective nature of the analysis, according to researchers.
Li Zhang, MD, PhD, of Tongji Hospital in Wuhan, China, presented this research at the AACR virtual meeting I. Some of the data were previously published in Annals of Oncology.
The researchers studied 28 patients with cancer among 1,276 patients with COVID-19 treated at three hospitals in Wuhan. The most common cancer types were lung (n = 7), esophageal (n = 4), and breast (n = 3). Patients had other gastrointestinal, gynecologic, genitourinary, and head and neck cancers as well.
The patients’ median age was 65 years (range, 56-70 years), 60.9% were men, 35.7% had stage IV cancer, and 28.6% had hospital-acquired COVID-19. Antitumor treatments included chemotherapy (n = 22), surgery (n = 21), radiotherapy (n = 21), targeted therapy (n = 5), and immune checkpoint inhibitors (n = 2).
COVID-19 treatment
Most patients (n = 22) received oxygen as their only respiratory intervention, although 10 received mechanical ventilation.
For systemic therapy, patients received antibiotic treatment (n = 23), corticosteroids (n = 15), intravenous immunoglobulin (n = 10), and tocilizumab (n = 1).
Antiviral treatments included umifenovir (n = 14), lopinavir/ritonavir (n = 10), ganciclovir (n = 9), ribavirin (n = 1), or a combination of antiviral drugs (n = 9).
“No cancer patients were enrolled in clinical trials, so no one received hydroxychloroquine or remdesivir,” Dr. Zhang noted.
Outcomes
In all, 15 patients (53.6%) had severe events. The median time from COVID-19 diagnosis to severe events was 7 days (range, 5-15 days).
A total of eight patients (28.6%) died – three with lung cancer, two with prostate cancer, one with liver cancer, one with rectal cancer, and one with testicular cancer.
Causes of death were acute respiratory distress syndrome (n = 5), septic shock (n = 1), suspected pulmonary embolism (n = 1), and acute myocardial infarction (n = 1).
By April 4, 14 patients had been discharged from the hospital, and 6 were still hospitalized. The median duration of hospitalization was 18.4 days for discharged patients and 29.4 days for patients still in hospital.
Follow-up CT scans showed improvement in 13 patients, no changes in 5 patients, and deterioration in 6 patients.
Factors associated with severe events
In a multivariable analysis, receiving antitumor treatment within 14 days of COVID-19 diagnosis was associated with severe events (hazard ratio, 4.079; P = .037).
However, only seven patients received antitumor treatments within 14 days of COVID-19 diagnosis – three chemotherapy, two targeted therapy, one radiotherapy, and one immune checkpoint inhibitor. Five of these seven patients had severe events.
Another factor associated with severe events in multivariable analysis was patchy consolidation on CT scan at admission (HR, 5.438; P = .01). Age and gender were not significantly associated with severe events.
Immune checkpoint inhibitors
Dr. Zhang and colleagues also analyzed a second group of cancer patients and their family members to determine if patients on immune checkpoint inhibitors have an increased risk of COVID-19.
This group included 124 cancer patients treated with immune checkpoint inhibitors for at least 2 months. The patients had a median age of 59 years (range, 54-65 years), and 61.8% were men. Most patients (95.2%) had stage IV cancer, and the most common cancers were lung (54.0%), esophageal (18.6%), and head and neck (10.7%).
In this group, only one cancer patient developed COVID-19 (via nosocomial infection). In another case, a patient’s spouse developed COVID-19, but the patient did not.
Dr. Zhang said this “limited information did not suggest cancer patients treated with immune checkpoint inhibitors were more vulnerable to COVID infection.”
Dr. Zhang and colleagues reported no conflicts of interest. This research was funded by the National Natural Science Foundation of China and Huazhong University of Science and Technology COVID-19 Rapid Response Call China.
SOURCE: Zhang L et al. Ann Oncol. 2020 Mar 26. doi: 10.1016/j.annonc.2020.03.296.
Cancer patients who received antitumor treatment within 14 days of COVID-19 diagnosis had an increased risk of severe events, according to data from three hospitals in Wuhan.
Patients with patchy consolidation at hospital admission also had an increased risk of severe events, defined as ICU admission, mechanical ventilation, or death.
However, these findings are limited by the small number of patients studied and the retrospective nature of the analysis, according to researchers.
Li Zhang, MD, PhD, of Tongji Hospital in Wuhan, China, presented this research at the AACR virtual meeting I. Some of the data were previously published in Annals of Oncology.
The researchers studied 28 patients with cancer among 1,276 patients with COVID-19 treated at three hospitals in Wuhan. The most common cancer types were lung (n = 7), esophageal (n = 4), and breast (n = 3). Patients had other gastrointestinal, gynecologic, genitourinary, and head and neck cancers as well.
The patients’ median age was 65 years (range, 56-70 years), 60.9% were men, 35.7% had stage IV cancer, and 28.6% had hospital-acquired COVID-19. Antitumor treatments included chemotherapy (n = 22), surgery (n = 21), radiotherapy (n = 21), targeted therapy (n = 5), and immune checkpoint inhibitors (n = 2).
COVID-19 treatment
Most patients (n = 22) received oxygen as their only respiratory intervention, although 10 received mechanical ventilation.
For systemic therapy, patients received antibiotic treatment (n = 23), corticosteroids (n = 15), intravenous immunoglobulin (n = 10), and tocilizumab (n = 1).
Antiviral treatments included umifenovir (n = 14), lopinavir/ritonavir (n = 10), ganciclovir (n = 9), ribavirin (n = 1), or a combination of antiviral drugs (n = 9).
“No cancer patients were enrolled in clinical trials, so no one received hydroxychloroquine or remdesivir,” Dr. Zhang noted.
Outcomes
In all, 15 patients (53.6%) had severe events. The median time from COVID-19 diagnosis to severe events was 7 days (range, 5-15 days).
A total of eight patients (28.6%) died – three with lung cancer, two with prostate cancer, one with liver cancer, one with rectal cancer, and one with testicular cancer.
Causes of death were acute respiratory distress syndrome (n = 5), septic shock (n = 1), suspected pulmonary embolism (n = 1), and acute myocardial infarction (n = 1).
By April 4, 14 patients had been discharged from the hospital, and 6 were still hospitalized. The median duration of hospitalization was 18.4 days for discharged patients and 29.4 days for patients still in hospital.
Follow-up CT scans showed improvement in 13 patients, no changes in 5 patients, and deterioration in 6 patients.
Factors associated with severe events
In a multivariable analysis, receiving antitumor treatment within 14 days of COVID-19 diagnosis was associated with severe events (hazard ratio, 4.079; P = .037).
However, only seven patients received antitumor treatments within 14 days of COVID-19 diagnosis – three chemotherapy, two targeted therapy, one radiotherapy, and one immune checkpoint inhibitor. Five of these seven patients had severe events.
Another factor associated with severe events in multivariable analysis was patchy consolidation on CT scan at admission (HR, 5.438; P = .01). Age and gender were not significantly associated with severe events.
Immune checkpoint inhibitors
Dr. Zhang and colleagues also analyzed a second group of cancer patients and their family members to determine if patients on immune checkpoint inhibitors have an increased risk of COVID-19.
This group included 124 cancer patients treated with immune checkpoint inhibitors for at least 2 months. The patients had a median age of 59 years (range, 54-65 years), and 61.8% were men. Most patients (95.2%) had stage IV cancer, and the most common cancers were lung (54.0%), esophageal (18.6%), and head and neck (10.7%).
In this group, only one cancer patient developed COVID-19 (via nosocomial infection). In another case, a patient’s spouse developed COVID-19, but the patient did not.
Dr. Zhang said this “limited information did not suggest cancer patients treated with immune checkpoint inhibitors were more vulnerable to COVID infection.”
Dr. Zhang and colleagues reported no conflicts of interest. This research was funded by the National Natural Science Foundation of China and Huazhong University of Science and Technology COVID-19 Rapid Response Call China.
SOURCE: Zhang L et al. Ann Oncol. 2020 Mar 26. doi: 10.1016/j.annonc.2020.03.296.
Cancer patients who received antitumor treatment within 14 days of COVID-19 diagnosis had an increased risk of severe events, according to data from three hospitals in Wuhan.
Patients with patchy consolidation at hospital admission also had an increased risk of severe events, defined as ICU admission, mechanical ventilation, or death.
However, these findings are limited by the small number of patients studied and the retrospective nature of the analysis, according to researchers.
Li Zhang, MD, PhD, of Tongji Hospital in Wuhan, China, presented this research at the AACR virtual meeting I. Some of the data were previously published in Annals of Oncology.
The researchers studied 28 patients with cancer among 1,276 patients with COVID-19 treated at three hospitals in Wuhan. The most common cancer types were lung (n = 7), esophageal (n = 4), and breast (n = 3). Patients had other gastrointestinal, gynecologic, genitourinary, and head and neck cancers as well.
The patients’ median age was 65 years (range, 56-70 years), 60.9% were men, 35.7% had stage IV cancer, and 28.6% had hospital-acquired COVID-19. Antitumor treatments included chemotherapy (n = 22), surgery (n = 21), radiotherapy (n = 21), targeted therapy (n = 5), and immune checkpoint inhibitors (n = 2).
COVID-19 treatment
Most patients (n = 22) received oxygen as their only respiratory intervention, although 10 received mechanical ventilation.
For systemic therapy, patients received antibiotic treatment (n = 23), corticosteroids (n = 15), intravenous immunoglobulin (n = 10), and tocilizumab (n = 1).
Antiviral treatments included umifenovir (n = 14), lopinavir/ritonavir (n = 10), ganciclovir (n = 9), ribavirin (n = 1), or a combination of antiviral drugs (n = 9).
“No cancer patients were enrolled in clinical trials, so no one received hydroxychloroquine or remdesivir,” Dr. Zhang noted.
Outcomes
In all, 15 patients (53.6%) had severe events. The median time from COVID-19 diagnosis to severe events was 7 days (range, 5-15 days).
A total of eight patients (28.6%) died – three with lung cancer, two with prostate cancer, one with liver cancer, one with rectal cancer, and one with testicular cancer.
Causes of death were acute respiratory distress syndrome (n = 5), septic shock (n = 1), suspected pulmonary embolism (n = 1), and acute myocardial infarction (n = 1).
By April 4, 14 patients had been discharged from the hospital, and 6 were still hospitalized. The median duration of hospitalization was 18.4 days for discharged patients and 29.4 days for patients still in hospital.
Follow-up CT scans showed improvement in 13 patients, no changes in 5 patients, and deterioration in 6 patients.
Factors associated with severe events
In a multivariable analysis, receiving antitumor treatment within 14 days of COVID-19 diagnosis was associated with severe events (hazard ratio, 4.079; P = .037).
However, only seven patients received antitumor treatments within 14 days of COVID-19 diagnosis – three chemotherapy, two targeted therapy, one radiotherapy, and one immune checkpoint inhibitor. Five of these seven patients had severe events.
Another factor associated with severe events in multivariable analysis was patchy consolidation on CT scan at admission (HR, 5.438; P = .01). Age and gender were not significantly associated with severe events.
Immune checkpoint inhibitors
Dr. Zhang and colleagues also analyzed a second group of cancer patients and their family members to determine if patients on immune checkpoint inhibitors have an increased risk of COVID-19.
This group included 124 cancer patients treated with immune checkpoint inhibitors for at least 2 months. The patients had a median age of 59 years (range, 54-65 years), and 61.8% were men. Most patients (95.2%) had stage IV cancer, and the most common cancers were lung (54.0%), esophageal (18.6%), and head and neck (10.7%).
In this group, only one cancer patient developed COVID-19 (via nosocomial infection). In another case, a patient’s spouse developed COVID-19, but the patient did not.
Dr. Zhang said this “limited information did not suggest cancer patients treated with immune checkpoint inhibitors were more vulnerable to COVID infection.”
Dr. Zhang and colleagues reported no conflicts of interest. This research was funded by the National Natural Science Foundation of China and Huazhong University of Science and Technology COVID-19 Rapid Response Call China.
SOURCE: Zhang L et al. Ann Oncol. 2020 Mar 26. doi: 10.1016/j.annonc.2020.03.296.
FROM AACR 2020
Call for Immuno-oncology and Immunotherapy Manuscripts
Federal Practitioner is inviting hematology and oncology health care providers and researchers to contribute to the November 2020 special issue on immuno-oncology. The special issue is produced in cooperation with the Association of VA Hematology/Oncology (AVAHO). The journal is especially interested in new research, case studies, review articles, and patient care program descriptions.
Interested authors can send a brief 2 to 3 sentence abstract to [email protected] by May 29, 2020, or submit a completed article directly into Editorial Manager, a web-based manuscript submission and review system. The updated and complete submission guidelines, including details about the style and format, can be found here:
http://www.mdedge.com/fedprac/
All manuscripts submitted to Federal Practitioner for both special and regular issues will be subject to peer review. Peer reviews are conducted in a double-blind fashion, and the reviewers are asked to comment on the manuscript’s importance, accuracy, relevance, clarity, timeliness, balance, and reference citation. Final decisions on all submitted manuscripts are made by the Editorial Advisory Association Hematology/Oncology special issue advisory board.
Federal Practitioner is inviting hematology and oncology health care providers and researchers to contribute to the November 2020 special issue on immuno-oncology. The special issue is produced in cooperation with the Association of VA Hematology/Oncology (AVAHO). The journal is especially interested in new research, case studies, review articles, and patient care program descriptions.
Interested authors can send a brief 2 to 3 sentence abstract to [email protected] by May 29, 2020, or submit a completed article directly into Editorial Manager, a web-based manuscript submission and review system. The updated and complete submission guidelines, including details about the style and format, can be found here:
http://www.mdedge.com/fedprac/
All manuscripts submitted to Federal Practitioner for both special and regular issues will be subject to peer review. Peer reviews are conducted in a double-blind fashion, and the reviewers are asked to comment on the manuscript’s importance, accuracy, relevance, clarity, timeliness, balance, and reference citation. Final decisions on all submitted manuscripts are made by the Editorial Advisory Association Hematology/Oncology special issue advisory board.
Federal Practitioner is inviting hematology and oncology health care providers and researchers to contribute to the November 2020 special issue on immuno-oncology. The special issue is produced in cooperation with the Association of VA Hematology/Oncology (AVAHO). The journal is especially interested in new research, case studies, review articles, and patient care program descriptions.
Interested authors can send a brief 2 to 3 sentence abstract to [email protected] by May 29, 2020, or submit a completed article directly into Editorial Manager, a web-based manuscript submission and review system. The updated and complete submission guidelines, including details about the style and format, can be found here:
http://www.mdedge.com/fedprac/
All manuscripts submitted to Federal Practitioner for both special and regular issues will be subject to peer review. Peer reviews are conducted in a double-blind fashion, and the reviewers are asked to comment on the manuscript’s importance, accuracy, relevance, clarity, timeliness, balance, and reference citation. Final decisions on all submitted manuscripts are made by the Editorial Advisory Association Hematology/Oncology special issue advisory board.
Increased risk of lung cancer with COPD, even in never smokers
an observational cohort study has shown.
Patients with COPD who had never smoked had more than double the risk of developing lung cancer (with an adjusted hazard ratio [HR] of 2.67), compared to individuals without COPD who had never smoked.
This was slightly higher than the increased risk seen in individuals who had smoked but who did not have COPD. This group had an almost double the risk of developing lung cancer (adjusted HR, 1.97), again compared to never smokers, the investigators added.
The highest risk of lung cancer was in patients who had COPD and who had smoked; this group had a sixfold risk of developing lung cancer (adjusted HR, 6.19) compared with never smokers without COPD, they note.
“COPD was a strong independent risk factor for lung cancer incidence in never smokers,” conclude the authors, led by Hye Yun Park, MD, Samsung Medical Center, Seoul, South Korea.
“Future studies should evaluate whether COPD patients are candidates for lung cancer screening, irrespective of smoking status,” they suggest.
The study was published March 10 in the journal Thorax.
It was based on an analysis of data from the National Health Insurance (NHS) Service National Sample Cohort between January 2002 and December 2013.
“We included all men and women, 40 to 84 years of age, who underwent at least one health screening examination provided by the NHS during the study period,” Park and colleagues explain.
Overall, the cohort included 338,548 men and women. Participants were followed-up for a median of 7 years.
Over the study interval, 1834 participants developed lung cancer.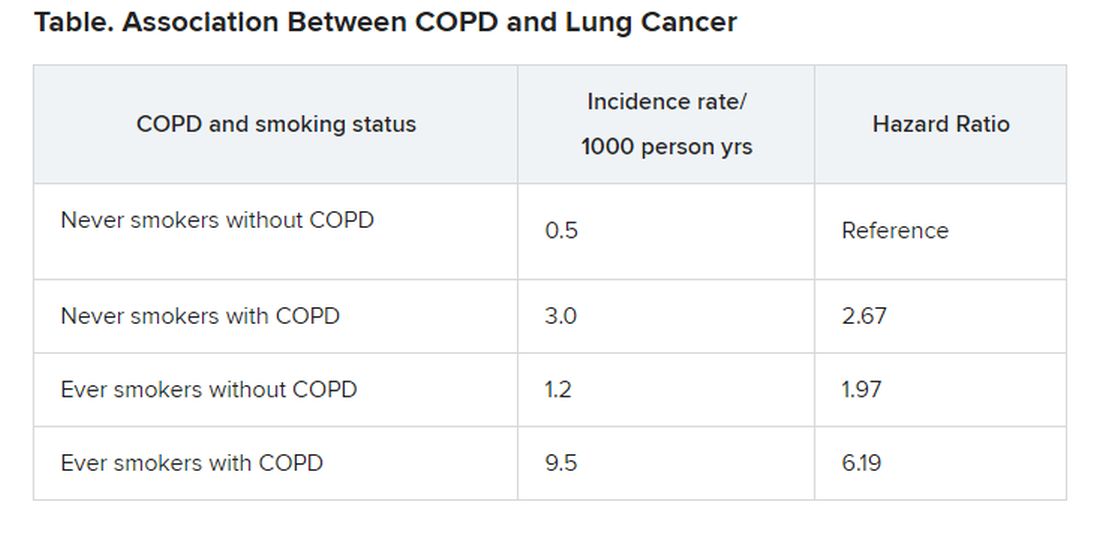
“The risk of disease [lung cancer] in never smokers with COPD was higher than that in ever smokers without COPD,” the investigators observe.
“Given that poor lung function in COPD is often a barrier to optimal lung cancer treatment due to increased risk of treatment-related morbidities, our study suggests that early detection of lung cancer in COPD patients may reduce the risk of treatment complications,” the authors write.
The study was supported by the National Research Foundation of Korea. The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
an observational cohort study has shown.
Patients with COPD who had never smoked had more than double the risk of developing lung cancer (with an adjusted hazard ratio [HR] of 2.67), compared to individuals without COPD who had never smoked.
This was slightly higher than the increased risk seen in individuals who had smoked but who did not have COPD. This group had an almost double the risk of developing lung cancer (adjusted HR, 1.97), again compared to never smokers, the investigators added.
The highest risk of lung cancer was in patients who had COPD and who had smoked; this group had a sixfold risk of developing lung cancer (adjusted HR, 6.19) compared with never smokers without COPD, they note.
“COPD was a strong independent risk factor for lung cancer incidence in never smokers,” conclude the authors, led by Hye Yun Park, MD, Samsung Medical Center, Seoul, South Korea.
“Future studies should evaluate whether COPD patients are candidates for lung cancer screening, irrespective of smoking status,” they suggest.
The study was published March 10 in the journal Thorax.
It was based on an analysis of data from the National Health Insurance (NHS) Service National Sample Cohort between January 2002 and December 2013.
“We included all men and women, 40 to 84 years of age, who underwent at least one health screening examination provided by the NHS during the study period,” Park and colleagues explain.
Overall, the cohort included 338,548 men and women. Participants were followed-up for a median of 7 years.
Over the study interval, 1834 participants developed lung cancer.
“The risk of disease [lung cancer] in never smokers with COPD was higher than that in ever smokers without COPD,” the investigators observe.
“Given that poor lung function in COPD is often a barrier to optimal lung cancer treatment due to increased risk of treatment-related morbidities, our study suggests that early detection of lung cancer in COPD patients may reduce the risk of treatment complications,” the authors write.
The study was supported by the National Research Foundation of Korea. The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
an observational cohort study has shown.
Patients with COPD who had never smoked had more than double the risk of developing lung cancer (with an adjusted hazard ratio [HR] of 2.67), compared to individuals without COPD who had never smoked.
This was slightly higher than the increased risk seen in individuals who had smoked but who did not have COPD. This group had an almost double the risk of developing lung cancer (adjusted HR, 1.97), again compared to never smokers, the investigators added.
The highest risk of lung cancer was in patients who had COPD and who had smoked; this group had a sixfold risk of developing lung cancer (adjusted HR, 6.19) compared with never smokers without COPD, they note.
“COPD was a strong independent risk factor for lung cancer incidence in never smokers,” conclude the authors, led by Hye Yun Park, MD, Samsung Medical Center, Seoul, South Korea.
“Future studies should evaluate whether COPD patients are candidates for lung cancer screening, irrespective of smoking status,” they suggest.
The study was published March 10 in the journal Thorax.
It was based on an analysis of data from the National Health Insurance (NHS) Service National Sample Cohort between January 2002 and December 2013.
“We included all men and women, 40 to 84 years of age, who underwent at least one health screening examination provided by the NHS during the study period,” Park and colleagues explain.
Overall, the cohort included 338,548 men and women. Participants were followed-up for a median of 7 years.
Over the study interval, 1834 participants developed lung cancer.
“The risk of disease [lung cancer] in never smokers with COPD was higher than that in ever smokers without COPD,” the investigators observe.
“Given that poor lung function in COPD is often a barrier to optimal lung cancer treatment due to increased risk of treatment-related morbidities, our study suggests that early detection of lung cancer in COPD patients may reduce the risk of treatment complications,” the authors write.
The study was supported by the National Research Foundation of Korea. The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
ASCO panel outlines cancer care challenges during COVID-19 pandemic
The COVID-19 pandemic continues to exact a heavy price on cancer patients, cancer care, and clinical trials, an expert panel reported during a presscast.
“Limited data available thus far are sobering: In Italy, about 20% of COVID-related deaths occurred in people with cancer, and, in China, COVID-19 patients who had cancer were about five times more likely than others to die or be placed on a ventilator in an intensive care unit,” said Howard A “Skip” Burris, MD, president of the American Society of Clinical Oncology and president and CEO of the Sarah Cannon Cancer Institute in Nashville, Tenn.
“We also have little evidence on returning COVID-19 patients with cancer. Physicians have to rely on limited data, anecdotal reports, and their own professional expertise” regarding the extent of increased risk to cancer patients with COVID-19, whether to interrupt or modify treatment, and the effects of cancer on recovery from COVID-19 infection, Dr. Burris said during the ASCO-sponsored online presscast.
Care of COVID-free patients
For cancer patients without COVID-19, the picture is equally dim, with the prospect of delayed surgery, chemotherapy, or screening; shortages of medications and equipment needed for critical care; the shift to telemedicine that may increase patient anxiety; and the potential loss of access to innovative therapies through clinical trials, Dr. Burris said.
“We’re concerned that some hospitals have effectively deemed all cancer surgeries to be elective, requiring them to be postponed. For patients with fast-moving or hard-to-treat cancer, this delay may be devastating,” he said.
Dr. Burris also cited concerns about delayed cancer diagnosis. “In a typical month, roughly 150,000 Americans are diagnosed with cancer. But right now, routine screening visits are postponed, and patients with pain or other warning signs may put off a doctor’s visit because of social distancing,” he said.
The pandemic has also exacerbated shortages of sedatives and opioid analgesics required for intubation and mechanical ventilation of patients.
Trials halted or slowed
Dr. Burris also briefly discussed results of a new survey, which were posted online ahead of publication in JCO Oncology Practice. The survey showed that, of 14 academic and 18 community-based cancer programs, 59.4% reported halting screening and/or enrollment for at least some clinical trials and suspending research-based clinical visits except for those where cancer treatment was delivered.
“Half of respondents reported ceasing research-only blood and/or tissue collections,” the authors of the article reported.
“Trial interruptions are devastating news for thousands of patients; in many cases, clinical trials are the best or only appropriate option for care,” Dr. Burris said.
The article authors, led by David Waterhouse, MD, of Oncology Hematology Care in Cincinnati, pointed to a silver lining in the pandemic cloud in the form of opportunities to improve clinical trials going forward.
“Nearly all respondents (90.3%) identified telehealth visits for participants as a potential improvement to clinical trial conduct, and more than three-quarters (77.4%) indicated that remote patient review of symptoms held similar potential,” the authors wrote.
Other potential improvements included remote site visits from trial sponsors and/or contract research organizations, more efficient study enrollment through secure electronic platforms, direct shipment of oral drugs to patients, remote assessments of adverse events, and streamlined data collection.
Lessons from the front lines
Another member of the presscast panel, Melissa Dillmon, MD, of the Harbin Clinic Cancer Center in Rome, Georgia, described the experience of community oncologists during the pandemic.
Her community, located in northeastern Georgia, experienced a COVID-19 outbreak in early March linked to services at two large churches. Community public health authorities issued a shelter-in-place order before the state government issued stay-at-home guidelines and shuttered all but essential business, some of which were allowed by state order to reopen as of April 24.
Dr. Dillmon’s center began screening patients for COVID-19 symptoms at the door, limited visitors or companions, instituted virtual visits and tumor boards, and set up a cancer treatment triage system that would allow essential surgeries to proceed and most infusions to continue, while delaying the start of chemotherapy when possible.
“We have encouraged patients to continue on treatment, especially if treatment is being given with curative intent, or if the cancer is responding well already to treatment,” she said.
The center, located in a community with a high prevalence of comorbidities and high incidence of lung cancer, has seen a sharp decline in colonoscopies, mammograms, and lung scans as patient shelter in place.
“We have great concerns about patients missing their screening lung scans, as this program has already proven to be finding earlier lung cancers that are curable,” Dr. Dillmon said.
A view from Washington state
Another panel member, Gary Lyman, MD, of the Fred Hutchinson Cancer Research Center in Seattle, described the response by the state of Washington, the initial epicenter of the COVID-19 outbreak in the United States.
Following identification of infections in hospitalized patients and at a nursing home in Kirkland, Washington, “our response, which began in early March and progressed through the second and third week in March at the state level, was to restrict large gatherings; progressively, schools were closed; larger businesses closed; and, by March 23, a stay-at-home policy was implemented, and all nonessential businesses were closed,” Dr. Lyman said.
“We believe, based on what has happened since that time, that this has considerably flattened the curve,” he continued.
Lessons from the Washington experience include the need to plan for a long-term disruption or alteration of cancer care, expand COVID-19 testing to all patients coming into hospitals or major clinics, institute aggressive supportive care measures, prepare for subsequent waves of infection, collect and share data, and, for remote or rural areas, identify lifelines to needed resources, Dr. Lyman said.
ASCO resources
Also speaking at the presscast, Jonathan Marron, MD, of Boston Children’s Hospital and Harvard Medical School, Boston, outlined ASCO’s guidance on allocation of scarce resources during the COVID-19 pandemic.
Richard L. Schilsky, MD, ASCO chief medical officer and executive vice president, outlined community-wide collaborations, data initiatives, and online resources for both clinicians and patients.
The COVID-19 pandemic continues to exact a heavy price on cancer patients, cancer care, and clinical trials, an expert panel reported during a presscast.
“Limited data available thus far are sobering: In Italy, about 20% of COVID-related deaths occurred in people with cancer, and, in China, COVID-19 patients who had cancer were about five times more likely than others to die or be placed on a ventilator in an intensive care unit,” said Howard A “Skip” Burris, MD, president of the American Society of Clinical Oncology and president and CEO of the Sarah Cannon Cancer Institute in Nashville, Tenn.
“We also have little evidence on returning COVID-19 patients with cancer. Physicians have to rely on limited data, anecdotal reports, and their own professional expertise” regarding the extent of increased risk to cancer patients with COVID-19, whether to interrupt or modify treatment, and the effects of cancer on recovery from COVID-19 infection, Dr. Burris said during the ASCO-sponsored online presscast.
Care of COVID-free patients
For cancer patients without COVID-19, the picture is equally dim, with the prospect of delayed surgery, chemotherapy, or screening; shortages of medications and equipment needed for critical care; the shift to telemedicine that may increase patient anxiety; and the potential loss of access to innovative therapies through clinical trials, Dr. Burris said.
“We’re concerned that some hospitals have effectively deemed all cancer surgeries to be elective, requiring them to be postponed. For patients with fast-moving or hard-to-treat cancer, this delay may be devastating,” he said.
Dr. Burris also cited concerns about delayed cancer diagnosis. “In a typical month, roughly 150,000 Americans are diagnosed with cancer. But right now, routine screening visits are postponed, and patients with pain or other warning signs may put off a doctor’s visit because of social distancing,” he said.
The pandemic has also exacerbated shortages of sedatives and opioid analgesics required for intubation and mechanical ventilation of patients.
Trials halted or slowed
Dr. Burris also briefly discussed results of a new survey, which were posted online ahead of publication in JCO Oncology Practice. The survey showed that, of 14 academic and 18 community-based cancer programs, 59.4% reported halting screening and/or enrollment for at least some clinical trials and suspending research-based clinical visits except for those where cancer treatment was delivered.
“Half of respondents reported ceasing research-only blood and/or tissue collections,” the authors of the article reported.
“Trial interruptions are devastating news for thousands of patients; in many cases, clinical trials are the best or only appropriate option for care,” Dr. Burris said.
The article authors, led by David Waterhouse, MD, of Oncology Hematology Care in Cincinnati, pointed to a silver lining in the pandemic cloud in the form of opportunities to improve clinical trials going forward.
“Nearly all respondents (90.3%) identified telehealth visits for participants as a potential improvement to clinical trial conduct, and more than three-quarters (77.4%) indicated that remote patient review of symptoms held similar potential,” the authors wrote.
Other potential improvements included remote site visits from trial sponsors and/or contract research organizations, more efficient study enrollment through secure electronic platforms, direct shipment of oral drugs to patients, remote assessments of adverse events, and streamlined data collection.
Lessons from the front lines
Another member of the presscast panel, Melissa Dillmon, MD, of the Harbin Clinic Cancer Center in Rome, Georgia, described the experience of community oncologists during the pandemic.
Her community, located in northeastern Georgia, experienced a COVID-19 outbreak in early March linked to services at two large churches. Community public health authorities issued a shelter-in-place order before the state government issued stay-at-home guidelines and shuttered all but essential business, some of which were allowed by state order to reopen as of April 24.
Dr. Dillmon’s center began screening patients for COVID-19 symptoms at the door, limited visitors or companions, instituted virtual visits and tumor boards, and set up a cancer treatment triage system that would allow essential surgeries to proceed and most infusions to continue, while delaying the start of chemotherapy when possible.
“We have encouraged patients to continue on treatment, especially if treatment is being given with curative intent, or if the cancer is responding well already to treatment,” she said.
The center, located in a community with a high prevalence of comorbidities and high incidence of lung cancer, has seen a sharp decline in colonoscopies, mammograms, and lung scans as patient shelter in place.
“We have great concerns about patients missing their screening lung scans, as this program has already proven to be finding earlier lung cancers that are curable,” Dr. Dillmon said.
A view from Washington state
Another panel member, Gary Lyman, MD, of the Fred Hutchinson Cancer Research Center in Seattle, described the response by the state of Washington, the initial epicenter of the COVID-19 outbreak in the United States.
Following identification of infections in hospitalized patients and at a nursing home in Kirkland, Washington, “our response, which began in early March and progressed through the second and third week in March at the state level, was to restrict large gatherings; progressively, schools were closed; larger businesses closed; and, by March 23, a stay-at-home policy was implemented, and all nonessential businesses were closed,” Dr. Lyman said.
“We believe, based on what has happened since that time, that this has considerably flattened the curve,” he continued.
Lessons from the Washington experience include the need to plan for a long-term disruption or alteration of cancer care, expand COVID-19 testing to all patients coming into hospitals or major clinics, institute aggressive supportive care measures, prepare for subsequent waves of infection, collect and share data, and, for remote or rural areas, identify lifelines to needed resources, Dr. Lyman said.
ASCO resources
Also speaking at the presscast, Jonathan Marron, MD, of Boston Children’s Hospital and Harvard Medical School, Boston, outlined ASCO’s guidance on allocation of scarce resources during the COVID-19 pandemic.
Richard L. Schilsky, MD, ASCO chief medical officer and executive vice president, outlined community-wide collaborations, data initiatives, and online resources for both clinicians and patients.
The COVID-19 pandemic continues to exact a heavy price on cancer patients, cancer care, and clinical trials, an expert panel reported during a presscast.
“Limited data available thus far are sobering: In Italy, about 20% of COVID-related deaths occurred in people with cancer, and, in China, COVID-19 patients who had cancer were about five times more likely than others to die or be placed on a ventilator in an intensive care unit,” said Howard A “Skip” Burris, MD, president of the American Society of Clinical Oncology and president and CEO of the Sarah Cannon Cancer Institute in Nashville, Tenn.
“We also have little evidence on returning COVID-19 patients with cancer. Physicians have to rely on limited data, anecdotal reports, and their own professional expertise” regarding the extent of increased risk to cancer patients with COVID-19, whether to interrupt or modify treatment, and the effects of cancer on recovery from COVID-19 infection, Dr. Burris said during the ASCO-sponsored online presscast.
Care of COVID-free patients
For cancer patients without COVID-19, the picture is equally dim, with the prospect of delayed surgery, chemotherapy, or screening; shortages of medications and equipment needed for critical care; the shift to telemedicine that may increase patient anxiety; and the potential loss of access to innovative therapies through clinical trials, Dr. Burris said.
“We’re concerned that some hospitals have effectively deemed all cancer surgeries to be elective, requiring them to be postponed. For patients with fast-moving or hard-to-treat cancer, this delay may be devastating,” he said.
Dr. Burris also cited concerns about delayed cancer diagnosis. “In a typical month, roughly 150,000 Americans are diagnosed with cancer. But right now, routine screening visits are postponed, and patients with pain or other warning signs may put off a doctor’s visit because of social distancing,” he said.
The pandemic has also exacerbated shortages of sedatives and opioid analgesics required for intubation and mechanical ventilation of patients.
Trials halted or slowed
Dr. Burris also briefly discussed results of a new survey, which were posted online ahead of publication in JCO Oncology Practice. The survey showed that, of 14 academic and 18 community-based cancer programs, 59.4% reported halting screening and/or enrollment for at least some clinical trials and suspending research-based clinical visits except for those where cancer treatment was delivered.
“Half of respondents reported ceasing research-only blood and/or tissue collections,” the authors of the article reported.
“Trial interruptions are devastating news for thousands of patients; in many cases, clinical trials are the best or only appropriate option for care,” Dr. Burris said.
The article authors, led by David Waterhouse, MD, of Oncology Hematology Care in Cincinnati, pointed to a silver lining in the pandemic cloud in the form of opportunities to improve clinical trials going forward.
“Nearly all respondents (90.3%) identified telehealth visits for participants as a potential improvement to clinical trial conduct, and more than three-quarters (77.4%) indicated that remote patient review of symptoms held similar potential,” the authors wrote.
Other potential improvements included remote site visits from trial sponsors and/or contract research organizations, more efficient study enrollment through secure electronic platforms, direct shipment of oral drugs to patients, remote assessments of adverse events, and streamlined data collection.
Lessons from the front lines
Another member of the presscast panel, Melissa Dillmon, MD, of the Harbin Clinic Cancer Center in Rome, Georgia, described the experience of community oncologists during the pandemic.
Her community, located in northeastern Georgia, experienced a COVID-19 outbreak in early March linked to services at two large churches. Community public health authorities issued a shelter-in-place order before the state government issued stay-at-home guidelines and shuttered all but essential business, some of which were allowed by state order to reopen as of April 24.
Dr. Dillmon’s center began screening patients for COVID-19 symptoms at the door, limited visitors or companions, instituted virtual visits and tumor boards, and set up a cancer treatment triage system that would allow essential surgeries to proceed and most infusions to continue, while delaying the start of chemotherapy when possible.
“We have encouraged patients to continue on treatment, especially if treatment is being given with curative intent, or if the cancer is responding well already to treatment,” she said.
The center, located in a community with a high prevalence of comorbidities and high incidence of lung cancer, has seen a sharp decline in colonoscopies, mammograms, and lung scans as patient shelter in place.
“We have great concerns about patients missing their screening lung scans, as this program has already proven to be finding earlier lung cancers that are curable,” Dr. Dillmon said.
A view from Washington state
Another panel member, Gary Lyman, MD, of the Fred Hutchinson Cancer Research Center in Seattle, described the response by the state of Washington, the initial epicenter of the COVID-19 outbreak in the United States.
Following identification of infections in hospitalized patients and at a nursing home in Kirkland, Washington, “our response, which began in early March and progressed through the second and third week in March at the state level, was to restrict large gatherings; progressively, schools were closed; larger businesses closed; and, by March 23, a stay-at-home policy was implemented, and all nonessential businesses were closed,” Dr. Lyman said.
“We believe, based on what has happened since that time, that this has considerably flattened the curve,” he continued.
Lessons from the Washington experience include the need to plan for a long-term disruption or alteration of cancer care, expand COVID-19 testing to all patients coming into hospitals or major clinics, institute aggressive supportive care measures, prepare for subsequent waves of infection, collect and share data, and, for remote or rural areas, identify lifelines to needed resources, Dr. Lyman said.
ASCO resources
Also speaking at the presscast, Jonathan Marron, MD, of Boston Children’s Hospital and Harvard Medical School, Boston, outlined ASCO’s guidance on allocation of scarce resources during the COVID-19 pandemic.
Richard L. Schilsky, MD, ASCO chief medical officer and executive vice president, outlined community-wide collaborations, data initiatives, and online resources for both clinicians and patients.
Global registry collects data on pediatric cancer patients with COVID-19
A week after its launch, a new online registry has information on more than 2 dozen cases of pediatric cancer patients with COVID-19.
The registry, created by St. Jude Children’s Research Hospital in Memphis, Tenn., and the International Society of Paediatric Oncology, is the first global COVID-19 registry for children with cancer.
Clinicians enter cases through an online form, then complete 30- and 60-day follow-up reports via email. St. Jude compiles the data and releases regularly updated summaries, including the number of cases by country and by treatment. Eventually, researchers might be able to apply for access to the raw data for their own projects.
It’s all free of charge, said Carlos Rodriguez-Galindo, MD, chair of the department of global pediatric medicine at St. Jude.
The registry is hosted on a website called “The Global COVID-19 Observatory and Resource Center for Childhood Cancer.” In addition to the registry, the website has a resource library and a discussion forum where clinicians can exchange information.
Other COVID-19 cancer registries have launched recently as well, including registries created by the COVID-19 and Cancer Consortium and the American Society of Clinical Oncology. The idea is to compile and disseminate best practices and other information quickly amid concerns that immunosuppressed cancer patients might be especially vulnerable.
So far, that doesn’t seem to be the case for children. Their relative protection from the disease and serious complications seems to hold even when they have cancer, Dr. Rodriguez-Galindo said.
“When we talk with the people in China” the number of COVID-19 cases in children with cancer is “very small,” he said. There are a couple of reports from Europe finding the same thing, and the severity of COVID-19 also “seems to be lower than you would expect,” he added.
The new registry will help better define the situation, according to Dr. Rodriguez-Galindo.
St. Jude is working with European countries that have their own national pediatric cancer COVID-19 registries to share information. St. Jude’s ties with lower- and middle-income countries, established via the department of global pediatric medicine, should help populate the global registry as well.
Furthermore, international surveys are being planned to gauge the impact of COVID-19 on children with cancer and their access to care.
A week after its launch, a new online registry has information on more than 2 dozen cases of pediatric cancer patients with COVID-19.
The registry, created by St. Jude Children’s Research Hospital in Memphis, Tenn., and the International Society of Paediatric Oncology, is the first global COVID-19 registry for children with cancer.
Clinicians enter cases through an online form, then complete 30- and 60-day follow-up reports via email. St. Jude compiles the data and releases regularly updated summaries, including the number of cases by country and by treatment. Eventually, researchers might be able to apply for access to the raw data for their own projects.
It’s all free of charge, said Carlos Rodriguez-Galindo, MD, chair of the department of global pediatric medicine at St. Jude.
The registry is hosted on a website called “The Global COVID-19 Observatory and Resource Center for Childhood Cancer.” In addition to the registry, the website has a resource library and a discussion forum where clinicians can exchange information.
Other COVID-19 cancer registries have launched recently as well, including registries created by the COVID-19 and Cancer Consortium and the American Society of Clinical Oncology. The idea is to compile and disseminate best practices and other information quickly amid concerns that immunosuppressed cancer patients might be especially vulnerable.
So far, that doesn’t seem to be the case for children. Their relative protection from the disease and serious complications seems to hold even when they have cancer, Dr. Rodriguez-Galindo said.
“When we talk with the people in China” the number of COVID-19 cases in children with cancer is “very small,” he said. There are a couple of reports from Europe finding the same thing, and the severity of COVID-19 also “seems to be lower than you would expect,” he added.
The new registry will help better define the situation, according to Dr. Rodriguez-Galindo.
St. Jude is working with European countries that have their own national pediatric cancer COVID-19 registries to share information. St. Jude’s ties with lower- and middle-income countries, established via the department of global pediatric medicine, should help populate the global registry as well.
Furthermore, international surveys are being planned to gauge the impact of COVID-19 on children with cancer and their access to care.
A week after its launch, a new online registry has information on more than 2 dozen cases of pediatric cancer patients with COVID-19.
The registry, created by St. Jude Children’s Research Hospital in Memphis, Tenn., and the International Society of Paediatric Oncology, is the first global COVID-19 registry for children with cancer.
Clinicians enter cases through an online form, then complete 30- and 60-day follow-up reports via email. St. Jude compiles the data and releases regularly updated summaries, including the number of cases by country and by treatment. Eventually, researchers might be able to apply for access to the raw data for their own projects.
It’s all free of charge, said Carlos Rodriguez-Galindo, MD, chair of the department of global pediatric medicine at St. Jude.
The registry is hosted on a website called “The Global COVID-19 Observatory and Resource Center for Childhood Cancer.” In addition to the registry, the website has a resource library and a discussion forum where clinicians can exchange information.
Other COVID-19 cancer registries have launched recently as well, including registries created by the COVID-19 and Cancer Consortium and the American Society of Clinical Oncology. The idea is to compile and disseminate best practices and other information quickly amid concerns that immunosuppressed cancer patients might be especially vulnerable.
So far, that doesn’t seem to be the case for children. Their relative protection from the disease and serious complications seems to hold even when they have cancer, Dr. Rodriguez-Galindo said.
“When we talk with the people in China” the number of COVID-19 cases in children with cancer is “very small,” he said. There are a couple of reports from Europe finding the same thing, and the severity of COVID-19 also “seems to be lower than you would expect,” he added.
The new registry will help better define the situation, according to Dr. Rodriguez-Galindo.
St. Jude is working with European countries that have their own national pediatric cancer COVID-19 registries to share information. St. Jude’s ties with lower- and middle-income countries, established via the department of global pediatric medicine, should help populate the global registry as well.
Furthermore, international surveys are being planned to gauge the impact of COVID-19 on children with cancer and their access to care.
Six million childhood cancer deaths could be prevented over the next 30 years
Unless global investments are made to improve care worldwide, 11.1 million children will die from cancer over the next 30 years; 9.3 million of them (84%) will be in low- and lower-middle–income countries, according to a report in Lancet Oncology.
The report suggests that one in two new cases of childhood cancer are undiagnosed in low- and middle-income countries. If that trend continues, the number of children with cancer who are never diagnosed over the next 3 decades will exceed the number of those who are diagnosed.
Childhood cancer “is not complex, expensive, difficult to diagnose, or complicated to treat,” yet there’s a “worldwide inequity and a bleak picture for children with cancer” in low-income and middle-income countries, according to the report authors. The authors are 44 oncologists, pediatricians, and global health experts from around the world, led by Rifat Atun, MD, a professor of global health systems at Harvard University in Boston.
“For too long, there has been a widespread misconception that caring for children with cancer in low- and middle-income countries is expensive, unattainable, and inappropriate because of competing health priorities. Nothing could be further from the truth,” Dr. Atun said in a statement.
Dr. Atun and colleagues argued that the burden of childhood cancer “could be vastly reduced with new funding to scale up cost-effective interventions.” In fact, the authors estimated that scaling up interventions could prevent 6.2 million childhood cancer deaths between 2020 and 2050.
The reduction in deaths would translate to 318.4 million life-years gained, which would, in turn, translate to a global lifetime productivity gain of $2,580 billion, four times greater than the cumulative cost of $594 billion. This would mean a net return of $3 for every $1 spent.
Potential funders include governments, professional organizations, philanthropic groups, and industry, according to the authors. They also laid out the following six-pronged framework on how to proceed:
- Include childhood cancer in universal health coverage.
- Develop national cancer control plans for low-income and middle-income countries.
- End out-of-pocket costs for childhood cancer.
- Establish national and regional cancer networks to increase access to care.
- Expand population-based cancer registries to include children.
- Invest in research and innovations in low-income and middle-income countries.
“Success will be attained through political leadership, global solidarity, collective action, inclusive participation of all major stakeholders, and alignment of national and global efforts to expand access to effective and sustainable care for children with cancer,” the authors wrote.
No funding sources were reported. The authors didn’t have any disclosures.
SOURCE: Atun R et al. Lancet Oncol. 2020 Apr;21(4):e185-224.
Unless global investments are made to improve care worldwide, 11.1 million children will die from cancer over the next 30 years; 9.3 million of them (84%) will be in low- and lower-middle–income countries, according to a report in Lancet Oncology.
The report suggests that one in two new cases of childhood cancer are undiagnosed in low- and middle-income countries. If that trend continues, the number of children with cancer who are never diagnosed over the next 3 decades will exceed the number of those who are diagnosed.
Childhood cancer “is not complex, expensive, difficult to diagnose, or complicated to treat,” yet there’s a “worldwide inequity and a bleak picture for children with cancer” in low-income and middle-income countries, according to the report authors. The authors are 44 oncologists, pediatricians, and global health experts from around the world, led by Rifat Atun, MD, a professor of global health systems at Harvard University in Boston.
“For too long, there has been a widespread misconception that caring for children with cancer in low- and middle-income countries is expensive, unattainable, and inappropriate because of competing health priorities. Nothing could be further from the truth,” Dr. Atun said in a statement.
Dr. Atun and colleagues argued that the burden of childhood cancer “could be vastly reduced with new funding to scale up cost-effective interventions.” In fact, the authors estimated that scaling up interventions could prevent 6.2 million childhood cancer deaths between 2020 and 2050.
The reduction in deaths would translate to 318.4 million life-years gained, which would, in turn, translate to a global lifetime productivity gain of $2,580 billion, four times greater than the cumulative cost of $594 billion. This would mean a net return of $3 for every $1 spent.
Potential funders include governments, professional organizations, philanthropic groups, and industry, according to the authors. They also laid out the following six-pronged framework on how to proceed:
- Include childhood cancer in universal health coverage.
- Develop national cancer control plans for low-income and middle-income countries.
- End out-of-pocket costs for childhood cancer.
- Establish national and regional cancer networks to increase access to care.
- Expand population-based cancer registries to include children.
- Invest in research and innovations in low-income and middle-income countries.
“Success will be attained through political leadership, global solidarity, collective action, inclusive participation of all major stakeholders, and alignment of national and global efforts to expand access to effective and sustainable care for children with cancer,” the authors wrote.
No funding sources were reported. The authors didn’t have any disclosures.
SOURCE: Atun R et al. Lancet Oncol. 2020 Apr;21(4):e185-224.
Unless global investments are made to improve care worldwide, 11.1 million children will die from cancer over the next 30 years; 9.3 million of them (84%) will be in low- and lower-middle–income countries, according to a report in Lancet Oncology.
The report suggests that one in two new cases of childhood cancer are undiagnosed in low- and middle-income countries. If that trend continues, the number of children with cancer who are never diagnosed over the next 3 decades will exceed the number of those who are diagnosed.
Childhood cancer “is not complex, expensive, difficult to diagnose, or complicated to treat,” yet there’s a “worldwide inequity and a bleak picture for children with cancer” in low-income and middle-income countries, according to the report authors. The authors are 44 oncologists, pediatricians, and global health experts from around the world, led by Rifat Atun, MD, a professor of global health systems at Harvard University in Boston.
“For too long, there has been a widespread misconception that caring for children with cancer in low- and middle-income countries is expensive, unattainable, and inappropriate because of competing health priorities. Nothing could be further from the truth,” Dr. Atun said in a statement.
Dr. Atun and colleagues argued that the burden of childhood cancer “could be vastly reduced with new funding to scale up cost-effective interventions.” In fact, the authors estimated that scaling up interventions could prevent 6.2 million childhood cancer deaths between 2020 and 2050.
The reduction in deaths would translate to 318.4 million life-years gained, which would, in turn, translate to a global lifetime productivity gain of $2,580 billion, four times greater than the cumulative cost of $594 billion. This would mean a net return of $3 for every $1 spent.
Potential funders include governments, professional organizations, philanthropic groups, and industry, according to the authors. They also laid out the following six-pronged framework on how to proceed:
- Include childhood cancer in universal health coverage.
- Develop national cancer control plans for low-income and middle-income countries.
- End out-of-pocket costs for childhood cancer.
- Establish national and regional cancer networks to increase access to care.
- Expand population-based cancer registries to include children.
- Invest in research and innovations in low-income and middle-income countries.
“Success will be attained through political leadership, global solidarity, collective action, inclusive participation of all major stakeholders, and alignment of national and global efforts to expand access to effective and sustainable care for children with cancer,” the authors wrote.
No funding sources were reported. The authors didn’t have any disclosures.
SOURCE: Atun R et al. Lancet Oncol. 2020 Apr;21(4):e185-224.
FROM LANCET ONCOLOGY
Will coronavirus restrictions lead to more advanced cancers?
My pathology lab once faced a daily flood of colon polyps, pap smears, and prostate biopsies. Suddenly, our work has dried up. The coronavirus pandemic has cleared out operating rooms and clinics across the country. Endoscopy and radiology suites have gone dark.
Pathology is largely driven by mass screening programs, and the machinery of screening has grinded to a halt during the COVID-19 pandemic. The American Cancer Society currently recommends that “no one should go to a health care facility for routine cancer screening at this time.”
But malignancies are still growing and spreading even though a great deal of medical care is on hold. The most urgent cancer care is still taking place; the risks of delaying treatment for patients with advanced or symptomatic cancer are obvious—these tumors can cause severe pain and life-threatening complications.
But that leaves us with a more complex and uncomfortable question: Will the pause in screening ultimately leave patients with tiny, asymptomatic cancers or precursor lesions worse off? What will a delay mean for those with ductal carcinoma in situ or small breast cancers? What’s the long-term effect of all those dysplastic nevi and early melanoma left unexcised by dermatologists? Perhaps more troubling, what about the spreading kidney cancer that may have turned up as an incidental finding on a CT scan?
COVID-19: A natural experiment
For many years, we’ve been dealing with the other side of the screening question: overdiagnosing and treating cancers that would probably never harm the patient. Overdiagnosis has been on a decades-long rise due to organized screening like PSA testing and mammography, as well as through ad hoc detection from heavier use of medical imaging. All of these have been disrupted by the pandemic.
Because the correlation between medical interventions and cancer overdiagnosis is clear, we can safely assume that overdiagnosis will decline during the pandemic. But what will be the net effect? Early detection of cancer undoubtedly saves some lives, but how many and at what cost has been a seemingly intractable debate.
Until now.
The coronavirus outbreak will be a natural experiment like no other. Economists and epidemiologists love to study “natural experiments” – systemic shocks that shed light on a complex phenomenon.
The unexpected nationwide delay in screening will undoubtedly inform the debate on overdiagnosis. For one, we can learn whether less intensive screening leads to more advanced cancers. Because screening will probably return to normal at different times across the country, we can almost simulate a randomized trial. Will this transformative data be a silver lining to this awful time?
The pressure to ‘fight’
The pandemic has also raised a question about cancer screening that goes beyond data: Why has the loud epidemic of coronavirus so thoroughly trumped cancer’s silent one? To me, the necessary urgency of our coronavirus response stands in stark contrast to the overly aggressive public health messaging used for cancer screening.
The tools used to fight the coronavirus epidemic have been forceful. We’re all diligently washing our hands and staying inside. We’re making sacrifices in our jobs and personal lives to stop the virus’ spread.
Cancer screening has similarly been touted as dogma – an urgent public health intervention that only a fool would turn down. The American Cancer Society once ran an infamous advertisement suggesting that if you decline mammography, you “need more than your breasts examined.” Even today, well-intentioned organizations run cancer screening drives pushing people to pledge to “get screened now.” It is no surprise, then, that I have had patients and family members confide in me that they feel guilty about not pursuing all of their recommended screening tests. The thought of anyone feeling like they caused their own cancer appalls me.
This pressure extends into the clinic. In many practices, primary care doctors are evaluated based on how many patients “comply” with screening recommendations. There seems to be a relentless drive to reach 100% screening penetration. These oversimplified tactics run counter to the shared decision making and informed consent we profess to value in medicine.
The tricky thing about cancer screening is that because most people will never develop the cancer being screened for, we know that most people can also never be helped by it. This doesn’t make screening useless, just as washing your hands can help even if it doesn’t guarantee that you won’t catch coronavirus. We know that some individuals benefit, which we detect at the population level. Overdiagnosis arises in the same way, as a phenomenon detected within populations and not individuals. These aspects of screening are what has led to cancer being viewed as a “societal disease” requiring a uniform response – 100% screening compliance.
Metaphors of war
These assumptions fall apart now that we are facing a real societal disease, an infectious disease outbreak. Coronavirus has made us reflect on what actions individuals should take in order to protect others. But cancer is not a contagion. When we decide whether and how to screen, we make intimate decisions affecting primarily ourselves and our family – not society at large.
Countless articles have been written about the use of metaphor in cancer, perhaps most famously by essayist and breast cancer patient Susan Sontag. Sontag and others have been critical of the rampant use of war metaphors in the cancer community. Wars invoke sacrifice, duty, and suffering. The “battle” against coronavirus really puts the “war on cancer” in perspective. These pandemic weeks have terrified me. I have been willing to do anything to protect myself and others. They’ve also exhausted me. We can’t be at war forever.
When this current war ends, will the “war on cancer” resume unchanged? Screening will no doubt begin again, hopefully improved by data from the coronavirus natural experiment. But I wonder whether we will tolerate the same kinds of public health messages – and whether we should – having now experienced an infectious disease outbreak where our actions as individuals really do have an impact on the health of others.
After feeling helpless, besieged, and even guilt-ridden during the pandemic, I think many people would appreciate regaining a sense of control over other aspects of their health. Cancer screening can save lives, but it’s a choice we should make for ourselves based on an understanding of the trade-offs and our own preferences. When screening restarts, I hope its paternalistic dogma can be replaced by nuanced, empowering tactics more appropriate for peacetime.
Benjamin Mazer, MD, MBA, is an anatomic and clinical pathology resident at Yale with interests in diagnostic surgical pathology, laboratory management, and evidence-based medicine.
This article first appeared on Medscape.com.
My pathology lab once faced a daily flood of colon polyps, pap smears, and prostate biopsies. Suddenly, our work has dried up. The coronavirus pandemic has cleared out operating rooms and clinics across the country. Endoscopy and radiology suites have gone dark.
Pathology is largely driven by mass screening programs, and the machinery of screening has grinded to a halt during the COVID-19 pandemic. The American Cancer Society currently recommends that “no one should go to a health care facility for routine cancer screening at this time.”
But malignancies are still growing and spreading even though a great deal of medical care is on hold. The most urgent cancer care is still taking place; the risks of delaying treatment for patients with advanced or symptomatic cancer are obvious—these tumors can cause severe pain and life-threatening complications.
But that leaves us with a more complex and uncomfortable question: Will the pause in screening ultimately leave patients with tiny, asymptomatic cancers or precursor lesions worse off? What will a delay mean for those with ductal carcinoma in situ or small breast cancers? What’s the long-term effect of all those dysplastic nevi and early melanoma left unexcised by dermatologists? Perhaps more troubling, what about the spreading kidney cancer that may have turned up as an incidental finding on a CT scan?
COVID-19: A natural experiment
For many years, we’ve been dealing with the other side of the screening question: overdiagnosing and treating cancers that would probably never harm the patient. Overdiagnosis has been on a decades-long rise due to organized screening like PSA testing and mammography, as well as through ad hoc detection from heavier use of medical imaging. All of these have been disrupted by the pandemic.
Because the correlation between medical interventions and cancer overdiagnosis is clear, we can safely assume that overdiagnosis will decline during the pandemic. But what will be the net effect? Early detection of cancer undoubtedly saves some lives, but how many and at what cost has been a seemingly intractable debate.
Until now.
The coronavirus outbreak will be a natural experiment like no other. Economists and epidemiologists love to study “natural experiments” – systemic shocks that shed light on a complex phenomenon.
The unexpected nationwide delay in screening will undoubtedly inform the debate on overdiagnosis. For one, we can learn whether less intensive screening leads to more advanced cancers. Because screening will probably return to normal at different times across the country, we can almost simulate a randomized trial. Will this transformative data be a silver lining to this awful time?
The pressure to ‘fight’
The pandemic has also raised a question about cancer screening that goes beyond data: Why has the loud epidemic of coronavirus so thoroughly trumped cancer’s silent one? To me, the necessary urgency of our coronavirus response stands in stark contrast to the overly aggressive public health messaging used for cancer screening.
The tools used to fight the coronavirus epidemic have been forceful. We’re all diligently washing our hands and staying inside. We’re making sacrifices in our jobs and personal lives to stop the virus’ spread.
Cancer screening has similarly been touted as dogma – an urgent public health intervention that only a fool would turn down. The American Cancer Society once ran an infamous advertisement suggesting that if you decline mammography, you “need more than your breasts examined.” Even today, well-intentioned organizations run cancer screening drives pushing people to pledge to “get screened now.” It is no surprise, then, that I have had patients and family members confide in me that they feel guilty about not pursuing all of their recommended screening tests. The thought of anyone feeling like they caused their own cancer appalls me.
This pressure extends into the clinic. In many practices, primary care doctors are evaluated based on how many patients “comply” with screening recommendations. There seems to be a relentless drive to reach 100% screening penetration. These oversimplified tactics run counter to the shared decision making and informed consent we profess to value in medicine.
The tricky thing about cancer screening is that because most people will never develop the cancer being screened for, we know that most people can also never be helped by it. This doesn’t make screening useless, just as washing your hands can help even if it doesn’t guarantee that you won’t catch coronavirus. We know that some individuals benefit, which we detect at the population level. Overdiagnosis arises in the same way, as a phenomenon detected within populations and not individuals. These aspects of screening are what has led to cancer being viewed as a “societal disease” requiring a uniform response – 100% screening compliance.
Metaphors of war
These assumptions fall apart now that we are facing a real societal disease, an infectious disease outbreak. Coronavirus has made us reflect on what actions individuals should take in order to protect others. But cancer is not a contagion. When we decide whether and how to screen, we make intimate decisions affecting primarily ourselves and our family – not society at large.
Countless articles have been written about the use of metaphor in cancer, perhaps most famously by essayist and breast cancer patient Susan Sontag. Sontag and others have been critical of the rampant use of war metaphors in the cancer community. Wars invoke sacrifice, duty, and suffering. The “battle” against coronavirus really puts the “war on cancer” in perspective. These pandemic weeks have terrified me. I have been willing to do anything to protect myself and others. They’ve also exhausted me. We can’t be at war forever.
When this current war ends, will the “war on cancer” resume unchanged? Screening will no doubt begin again, hopefully improved by data from the coronavirus natural experiment. But I wonder whether we will tolerate the same kinds of public health messages – and whether we should – having now experienced an infectious disease outbreak where our actions as individuals really do have an impact on the health of others.
After feeling helpless, besieged, and even guilt-ridden during the pandemic, I think many people would appreciate regaining a sense of control over other aspects of their health. Cancer screening can save lives, but it’s a choice we should make for ourselves based on an understanding of the trade-offs and our own preferences. When screening restarts, I hope its paternalistic dogma can be replaced by nuanced, empowering tactics more appropriate for peacetime.
Benjamin Mazer, MD, MBA, is an anatomic and clinical pathology resident at Yale with interests in diagnostic surgical pathology, laboratory management, and evidence-based medicine.
This article first appeared on Medscape.com.
My pathology lab once faced a daily flood of colon polyps, pap smears, and prostate biopsies. Suddenly, our work has dried up. The coronavirus pandemic has cleared out operating rooms and clinics across the country. Endoscopy and radiology suites have gone dark.
Pathology is largely driven by mass screening programs, and the machinery of screening has grinded to a halt during the COVID-19 pandemic. The American Cancer Society currently recommends that “no one should go to a health care facility for routine cancer screening at this time.”
But malignancies are still growing and spreading even though a great deal of medical care is on hold. The most urgent cancer care is still taking place; the risks of delaying treatment for patients with advanced or symptomatic cancer are obvious—these tumors can cause severe pain and life-threatening complications.
But that leaves us with a more complex and uncomfortable question: Will the pause in screening ultimately leave patients with tiny, asymptomatic cancers or precursor lesions worse off? What will a delay mean for those with ductal carcinoma in situ or small breast cancers? What’s the long-term effect of all those dysplastic nevi and early melanoma left unexcised by dermatologists? Perhaps more troubling, what about the spreading kidney cancer that may have turned up as an incidental finding on a CT scan?
COVID-19: A natural experiment
For many years, we’ve been dealing with the other side of the screening question: overdiagnosing and treating cancers that would probably never harm the patient. Overdiagnosis has been on a decades-long rise due to organized screening like PSA testing and mammography, as well as through ad hoc detection from heavier use of medical imaging. All of these have been disrupted by the pandemic.
Because the correlation between medical interventions and cancer overdiagnosis is clear, we can safely assume that overdiagnosis will decline during the pandemic. But what will be the net effect? Early detection of cancer undoubtedly saves some lives, but how many and at what cost has been a seemingly intractable debate.
Until now.
The coronavirus outbreak will be a natural experiment like no other. Economists and epidemiologists love to study “natural experiments” – systemic shocks that shed light on a complex phenomenon.
The unexpected nationwide delay in screening will undoubtedly inform the debate on overdiagnosis. For one, we can learn whether less intensive screening leads to more advanced cancers. Because screening will probably return to normal at different times across the country, we can almost simulate a randomized trial. Will this transformative data be a silver lining to this awful time?
The pressure to ‘fight’
The pandemic has also raised a question about cancer screening that goes beyond data: Why has the loud epidemic of coronavirus so thoroughly trumped cancer’s silent one? To me, the necessary urgency of our coronavirus response stands in stark contrast to the overly aggressive public health messaging used for cancer screening.
The tools used to fight the coronavirus epidemic have been forceful. We’re all diligently washing our hands and staying inside. We’re making sacrifices in our jobs and personal lives to stop the virus’ spread.
Cancer screening has similarly been touted as dogma – an urgent public health intervention that only a fool would turn down. The American Cancer Society once ran an infamous advertisement suggesting that if you decline mammography, you “need more than your breasts examined.” Even today, well-intentioned organizations run cancer screening drives pushing people to pledge to “get screened now.” It is no surprise, then, that I have had patients and family members confide in me that they feel guilty about not pursuing all of their recommended screening tests. The thought of anyone feeling like they caused their own cancer appalls me.
This pressure extends into the clinic. In many practices, primary care doctors are evaluated based on how many patients “comply” with screening recommendations. There seems to be a relentless drive to reach 100% screening penetration. These oversimplified tactics run counter to the shared decision making and informed consent we profess to value in medicine.
The tricky thing about cancer screening is that because most people will never develop the cancer being screened for, we know that most people can also never be helped by it. This doesn’t make screening useless, just as washing your hands can help even if it doesn’t guarantee that you won’t catch coronavirus. We know that some individuals benefit, which we detect at the population level. Overdiagnosis arises in the same way, as a phenomenon detected within populations and not individuals. These aspects of screening are what has led to cancer being viewed as a “societal disease” requiring a uniform response – 100% screening compliance.
Metaphors of war
These assumptions fall apart now that we are facing a real societal disease, an infectious disease outbreak. Coronavirus has made us reflect on what actions individuals should take in order to protect others. But cancer is not a contagion. When we decide whether and how to screen, we make intimate decisions affecting primarily ourselves and our family – not society at large.
Countless articles have been written about the use of metaphor in cancer, perhaps most famously by essayist and breast cancer patient Susan Sontag. Sontag and others have been critical of the rampant use of war metaphors in the cancer community. Wars invoke sacrifice, duty, and suffering. The “battle” against coronavirus really puts the “war on cancer” in perspective. These pandemic weeks have terrified me. I have been willing to do anything to protect myself and others. They’ve also exhausted me. We can’t be at war forever.
When this current war ends, will the “war on cancer” resume unchanged? Screening will no doubt begin again, hopefully improved by data from the coronavirus natural experiment. But I wonder whether we will tolerate the same kinds of public health messages – and whether we should – having now experienced an infectious disease outbreak where our actions as individuals really do have an impact on the health of others.
After feeling helpless, besieged, and even guilt-ridden during the pandemic, I think many people would appreciate regaining a sense of control over other aspects of their health. Cancer screening can save lives, but it’s a choice we should make for ourselves based on an understanding of the trade-offs and our own preferences. When screening restarts, I hope its paternalistic dogma can be replaced by nuanced, empowering tactics more appropriate for peacetime.
Benjamin Mazer, MD, MBA, is an anatomic and clinical pathology resident at Yale with interests in diagnostic surgical pathology, laboratory management, and evidence-based medicine.
This article first appeared on Medscape.com.
European cancer centers restructure care in the era of COVID-19
Delivering cancer care during the COVID-19 pandemic has proved particularly challenging, as minimizing the risk of infection must be balanced with maintaining optimal outcomes.
Healthcare systems and oncologists have had to reorganize standard oncologic care in order to protect vulnerable patients from exposure to COVID-19 as well as deal with pandemic-related issues of equipment and staffing shortages.
A new article now describes how seven cancer centers in Europe rapidly reorganized their oncologic services and are tackling this crisis, as well as offering guidance to other institutions.
This was a major undertaking, to work out a system where patients can still get care but in a safer manner, explained coauthor Emile Voest, MD, medical director of the Netherlands Cancer Institute in Amsterdam.
“Decisions needed to be taken based on availability of personnel, protective materials, and urgencies,” he told Medscape Medical News. “Because every country had its own speed of development of the COVID pandemic, there were different scenarios in all institutions, but all with a common factor of key expertise on how to de-escalate in a safe manner.”
The article was published April 16 in Nature Medicine.
The Netherlands Cancer Institute (the Netherlands), Karolinska Institute (Sweden), Institute Gustave Roussy (France), Cambridge Cancer Center (United Kingdom), Istituto Nazionale dei Tumori di Milano (Italy), German Cancer Research Center (Germany), and Vall d’Hebron Institute of Oncology (Spain) have been working closely together in a legal entity since 2014, and have created ‘Cancer Core Europe’ (CCE). The goal is to “maximize coherence and critical mass in cancer research,” the authors note.
The consortium represents roughly 60,000 patients with newly diagnosed cancer, delivers approximately 300,000 treatment courses, and conducts about 1.2 million consultations annually, with more than 1,500 ongoing clinical trials. In a joint effort, the centers collected, translated, and compared the guidelines that had been put in place to treat patients with cancer during the COVID-19 pandemic.
Cancer treatment is multidisciplinary and involves many specialties including surgery, radiology, pathology, radiation oncology, and medical oncology. Coordinating care among disciplines is a very complex process, Voest noted.
“Changing treatment also means that you need to reconsider capacities and requirements,” he said. “Hospitals have installed crisis teams that were very good at coordinating these efforts.”
Restructuring care
Cancer care had to be reorganized on multiple levels, and the CCE centers looked at several aspects that needed to be accounted for, to ensure continuity in cancer care.
“The biggest challenge for the NHS and other healthcare systems is the surge of patients requiring oxygen and/or intensive care, and the nature and infectiousness of the virus,” said coauthor Carlos Caldas, MD, FMedSci, professor of cancer medicine at the University of Cambridge, United Kingdom. “In hospitals that are mostly run close to capacity, and where all kinds of patients are treated, this has created major resource and logistical problems.”
For regular clinical activities, the institutions with dedicated cancer centers (German Cancer Research Center, Institute Gustave Roussy, Istituto Nazionale dei Tumori di Milano, and Netherlands Cancer Institute) have attempted to stay COVID-19 free. This policy would in turn help ensure that sufficient clinical and intensive-care capacity could be reserved for critical cancer surgeries or management of treatment-related side effects, and allow hospitals outside of the CCE to transfer patients with cancer to these centers. The general hospitals can then focus on caring for patients with COVID-19, as well as other illnesses/injuries that require inpatient care.
As the CCE centers located within general hospitals (Cambridge Cancer Center, Vall d’Hebron Institute of Oncology and Karolinska Institute) have to admit patients with suspected and positive cases of COVID-19, being “COVID-19 free” was never a realistic or pursued goal.
The authors note that it is the responsibility of all healthcare professionals to ensure patients are not exposed to COVID-19, and this has meant minimizing hospital visits and person-to-person contact. For example, whenever possible, consultations take place via telephone calls or over the Internet, and nonurgent appointments that would require a patient’s physical presence at the clinic have been postponed. Visitors are also not permitted to accompany patients when admitted to the hospital or during procedures.
Standard-of-care treatment regimens have been adapted across all centers to minimize the number of hospital visits and hospitalizations and prevent “anticancer treatment-induced” complications of COVID-19.
To minimize visits and hospitalizations, strategies include converting intravenous treatments to oral or subcutaneous regimens when possible; switching from cytotoxic chemotherapy to a less-toxic approach to minimize the risk of complications requiring hospitalization; or to pause therapies when possible (stable disease reached or better). In addition, nonemergency surgeries have been postponed or replaced by radiotherapy.
To prevent anticancer treatment-induced complications of COVID-19, most centers use the paradigm that the added benefit for tumor control should be weighed against the potential risk for COVID-19–related morbidity and mortality. To prevent or reduce the risk of neutropenia and lymphopenia, for example, all centers have suggested a de-escalation of cytotoxic chemotherapy or targeted treatment strategies, or to forgo second or subsequent lines of palliative treatments if response rates from up-front therapy are low.
Some of these changes may be here to stay, noted Caldas. “One of the positive messages that comes out of this is that, clearly, care can be delivered in a safe and compassionate manner without requiring as many hospital visits as in the pre-COVID-19 era,” he said. “In the future, we will take heed of the COVID-19 experience to improve delivery of cancer care.”
Capacity of facilities
Many healthcare systems have become overwhelmed as the pandemic has intensified, thus making it necessary to prioritize. To prepare for this possibility, CCE centers have established protocols to categorize and prioritize patients for systemic treatment or surgery. While the protocols vary by center, they are comparable with one another as they prioritize on the basis of anticipated treatment outcome, the authors note.
The guidelines in CCE centers unanimously recommend that neoadjuvant therapies and curative surgeries be the top priority, for the times when operating room and/or ICU capacity is limited. As an alternative, neoadjuvant systemic treatments may be initiated or extended to postpone surgery, and other nonsurgical interventions can be considered.
In addition, some centers agree that certain elective surgeries can be safely delayed if backed by scientific evidence. As an example, an 11-week deferment of surgery may be acceptable for patients with rectal cancer after downstaging.
Cancer centers may also need to upscale and downscale quickly, depending on how the pandemic evolves, and many have already outlined scenarios to prepare for increasing or decreasing their capacity using phased approaches.
The Netherlands Cancer Institute, for example, has defined four phases of increasing severity; in Germany, capacity planning has been coordinated among 18 hospitals and the federal ministry of health, in order to prevent shortages of cancer services.
“We note that the optimal downscaling strategies depend on country- and center-specific capacities and preferences,” they write. “Therefore, it is difficult to propose a common schedule, and it will be most effective if hospitals outline their own phase-specific downscaling strategies based on the prioritization schemes and practical handles discussed above.”
Future research
Better strategies will be needed to reduce the impact of COVID-19 in cancer care, and four research priorities were identified to allow for evidence-based adjustments of cancer care protocols while the pandemic continues:
- Collect real-world data about the effects of adjustment and de-escalation of treatment regimens on outcomes
- Determine the incidence of COVID-19 in both the general population and among patients with cancer who have received systemic therapies, with large-scale serological testing
- Develop an epidemiological model that will allow estimates of the cumulative incidence of COVID-19 for a patient with cancer, within a specific time frame
- Determine COVID-19 related morbidity and mortality in patients with cancer who have been treated with systemic therapies and/or granulocyte colony-stimulating factor (G-CSF). Several projects are currently underway, such as the UK Coronavirus Cancer Monitoring Project.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Delivering cancer care during the COVID-19 pandemic has proved particularly challenging, as minimizing the risk of infection must be balanced with maintaining optimal outcomes.
Healthcare systems and oncologists have had to reorganize standard oncologic care in order to protect vulnerable patients from exposure to COVID-19 as well as deal with pandemic-related issues of equipment and staffing shortages.
A new article now describes how seven cancer centers in Europe rapidly reorganized their oncologic services and are tackling this crisis, as well as offering guidance to other institutions.
This was a major undertaking, to work out a system where patients can still get care but in a safer manner, explained coauthor Emile Voest, MD, medical director of the Netherlands Cancer Institute in Amsterdam.
“Decisions needed to be taken based on availability of personnel, protective materials, and urgencies,” he told Medscape Medical News. “Because every country had its own speed of development of the COVID pandemic, there were different scenarios in all institutions, but all with a common factor of key expertise on how to de-escalate in a safe manner.”
The article was published April 16 in Nature Medicine.
The Netherlands Cancer Institute (the Netherlands), Karolinska Institute (Sweden), Institute Gustave Roussy (France), Cambridge Cancer Center (United Kingdom), Istituto Nazionale dei Tumori di Milano (Italy), German Cancer Research Center (Germany), and Vall d’Hebron Institute of Oncology (Spain) have been working closely together in a legal entity since 2014, and have created ‘Cancer Core Europe’ (CCE). The goal is to “maximize coherence and critical mass in cancer research,” the authors note.
The consortium represents roughly 60,000 patients with newly diagnosed cancer, delivers approximately 300,000 treatment courses, and conducts about 1.2 million consultations annually, with more than 1,500 ongoing clinical trials. In a joint effort, the centers collected, translated, and compared the guidelines that had been put in place to treat patients with cancer during the COVID-19 pandemic.
Cancer treatment is multidisciplinary and involves many specialties including surgery, radiology, pathology, radiation oncology, and medical oncology. Coordinating care among disciplines is a very complex process, Voest noted.
“Changing treatment also means that you need to reconsider capacities and requirements,” he said. “Hospitals have installed crisis teams that were very good at coordinating these efforts.”
Restructuring care
Cancer care had to be reorganized on multiple levels, and the CCE centers looked at several aspects that needed to be accounted for, to ensure continuity in cancer care.
“The biggest challenge for the NHS and other healthcare systems is the surge of patients requiring oxygen and/or intensive care, and the nature and infectiousness of the virus,” said coauthor Carlos Caldas, MD, FMedSci, professor of cancer medicine at the University of Cambridge, United Kingdom. “In hospitals that are mostly run close to capacity, and where all kinds of patients are treated, this has created major resource and logistical problems.”
For regular clinical activities, the institutions with dedicated cancer centers (German Cancer Research Center, Institute Gustave Roussy, Istituto Nazionale dei Tumori di Milano, and Netherlands Cancer Institute) have attempted to stay COVID-19 free. This policy would in turn help ensure that sufficient clinical and intensive-care capacity could be reserved for critical cancer surgeries or management of treatment-related side effects, and allow hospitals outside of the CCE to transfer patients with cancer to these centers. The general hospitals can then focus on caring for patients with COVID-19, as well as other illnesses/injuries that require inpatient care.
As the CCE centers located within general hospitals (Cambridge Cancer Center, Vall d’Hebron Institute of Oncology and Karolinska Institute) have to admit patients with suspected and positive cases of COVID-19, being “COVID-19 free” was never a realistic or pursued goal.
The authors note that it is the responsibility of all healthcare professionals to ensure patients are not exposed to COVID-19, and this has meant minimizing hospital visits and person-to-person contact. For example, whenever possible, consultations take place via telephone calls or over the Internet, and nonurgent appointments that would require a patient’s physical presence at the clinic have been postponed. Visitors are also not permitted to accompany patients when admitted to the hospital or during procedures.
Standard-of-care treatment regimens have been adapted across all centers to minimize the number of hospital visits and hospitalizations and prevent “anticancer treatment-induced” complications of COVID-19.
To minimize visits and hospitalizations, strategies include converting intravenous treatments to oral or subcutaneous regimens when possible; switching from cytotoxic chemotherapy to a less-toxic approach to minimize the risk of complications requiring hospitalization; or to pause therapies when possible (stable disease reached or better). In addition, nonemergency surgeries have been postponed or replaced by radiotherapy.
To prevent anticancer treatment-induced complications of COVID-19, most centers use the paradigm that the added benefit for tumor control should be weighed against the potential risk for COVID-19–related morbidity and mortality. To prevent or reduce the risk of neutropenia and lymphopenia, for example, all centers have suggested a de-escalation of cytotoxic chemotherapy or targeted treatment strategies, or to forgo second or subsequent lines of palliative treatments if response rates from up-front therapy are low.
Some of these changes may be here to stay, noted Caldas. “One of the positive messages that comes out of this is that, clearly, care can be delivered in a safe and compassionate manner without requiring as many hospital visits as in the pre-COVID-19 era,” he said. “In the future, we will take heed of the COVID-19 experience to improve delivery of cancer care.”
Capacity of facilities
Many healthcare systems have become overwhelmed as the pandemic has intensified, thus making it necessary to prioritize. To prepare for this possibility, CCE centers have established protocols to categorize and prioritize patients for systemic treatment or surgery. While the protocols vary by center, they are comparable with one another as they prioritize on the basis of anticipated treatment outcome, the authors note.
The guidelines in CCE centers unanimously recommend that neoadjuvant therapies and curative surgeries be the top priority, for the times when operating room and/or ICU capacity is limited. As an alternative, neoadjuvant systemic treatments may be initiated or extended to postpone surgery, and other nonsurgical interventions can be considered.
In addition, some centers agree that certain elective surgeries can be safely delayed if backed by scientific evidence. As an example, an 11-week deferment of surgery may be acceptable for patients with rectal cancer after downstaging.
Cancer centers may also need to upscale and downscale quickly, depending on how the pandemic evolves, and many have already outlined scenarios to prepare for increasing or decreasing their capacity using phased approaches.
The Netherlands Cancer Institute, for example, has defined four phases of increasing severity; in Germany, capacity planning has been coordinated among 18 hospitals and the federal ministry of health, in order to prevent shortages of cancer services.
“We note that the optimal downscaling strategies depend on country- and center-specific capacities and preferences,” they write. “Therefore, it is difficult to propose a common schedule, and it will be most effective if hospitals outline their own phase-specific downscaling strategies based on the prioritization schemes and practical handles discussed above.”
Future research
Better strategies will be needed to reduce the impact of COVID-19 in cancer care, and four research priorities were identified to allow for evidence-based adjustments of cancer care protocols while the pandemic continues:
- Collect real-world data about the effects of adjustment and de-escalation of treatment regimens on outcomes
- Determine the incidence of COVID-19 in both the general population and among patients with cancer who have received systemic therapies, with large-scale serological testing
- Develop an epidemiological model that will allow estimates of the cumulative incidence of COVID-19 for a patient with cancer, within a specific time frame
- Determine COVID-19 related morbidity and mortality in patients with cancer who have been treated with systemic therapies and/or granulocyte colony-stimulating factor (G-CSF). Several projects are currently underway, such as the UK Coronavirus Cancer Monitoring Project.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Delivering cancer care during the COVID-19 pandemic has proved particularly challenging, as minimizing the risk of infection must be balanced with maintaining optimal outcomes.
Healthcare systems and oncologists have had to reorganize standard oncologic care in order to protect vulnerable patients from exposure to COVID-19 as well as deal with pandemic-related issues of equipment and staffing shortages.
A new article now describes how seven cancer centers in Europe rapidly reorganized their oncologic services and are tackling this crisis, as well as offering guidance to other institutions.
This was a major undertaking, to work out a system where patients can still get care but in a safer manner, explained coauthor Emile Voest, MD, medical director of the Netherlands Cancer Institute in Amsterdam.
“Decisions needed to be taken based on availability of personnel, protective materials, and urgencies,” he told Medscape Medical News. “Because every country had its own speed of development of the COVID pandemic, there were different scenarios in all institutions, but all with a common factor of key expertise on how to de-escalate in a safe manner.”
The article was published April 16 in Nature Medicine.
The Netherlands Cancer Institute (the Netherlands), Karolinska Institute (Sweden), Institute Gustave Roussy (France), Cambridge Cancer Center (United Kingdom), Istituto Nazionale dei Tumori di Milano (Italy), German Cancer Research Center (Germany), and Vall d’Hebron Institute of Oncology (Spain) have been working closely together in a legal entity since 2014, and have created ‘Cancer Core Europe’ (CCE). The goal is to “maximize coherence and critical mass in cancer research,” the authors note.
The consortium represents roughly 60,000 patients with newly diagnosed cancer, delivers approximately 300,000 treatment courses, and conducts about 1.2 million consultations annually, with more than 1,500 ongoing clinical trials. In a joint effort, the centers collected, translated, and compared the guidelines that had been put in place to treat patients with cancer during the COVID-19 pandemic.
Cancer treatment is multidisciplinary and involves many specialties including surgery, radiology, pathology, radiation oncology, and medical oncology. Coordinating care among disciplines is a very complex process, Voest noted.
“Changing treatment also means that you need to reconsider capacities and requirements,” he said. “Hospitals have installed crisis teams that were very good at coordinating these efforts.”
Restructuring care
Cancer care had to be reorganized on multiple levels, and the CCE centers looked at several aspects that needed to be accounted for, to ensure continuity in cancer care.
“The biggest challenge for the NHS and other healthcare systems is the surge of patients requiring oxygen and/or intensive care, and the nature and infectiousness of the virus,” said coauthor Carlos Caldas, MD, FMedSci, professor of cancer medicine at the University of Cambridge, United Kingdom. “In hospitals that are mostly run close to capacity, and where all kinds of patients are treated, this has created major resource and logistical problems.”
For regular clinical activities, the institutions with dedicated cancer centers (German Cancer Research Center, Institute Gustave Roussy, Istituto Nazionale dei Tumori di Milano, and Netherlands Cancer Institute) have attempted to stay COVID-19 free. This policy would in turn help ensure that sufficient clinical and intensive-care capacity could be reserved for critical cancer surgeries or management of treatment-related side effects, and allow hospitals outside of the CCE to transfer patients with cancer to these centers. The general hospitals can then focus on caring for patients with COVID-19, as well as other illnesses/injuries that require inpatient care.
As the CCE centers located within general hospitals (Cambridge Cancer Center, Vall d’Hebron Institute of Oncology and Karolinska Institute) have to admit patients with suspected and positive cases of COVID-19, being “COVID-19 free” was never a realistic or pursued goal.
The authors note that it is the responsibility of all healthcare professionals to ensure patients are not exposed to COVID-19, and this has meant minimizing hospital visits and person-to-person contact. For example, whenever possible, consultations take place via telephone calls or over the Internet, and nonurgent appointments that would require a patient’s physical presence at the clinic have been postponed. Visitors are also not permitted to accompany patients when admitted to the hospital or during procedures.
Standard-of-care treatment regimens have been adapted across all centers to minimize the number of hospital visits and hospitalizations and prevent “anticancer treatment-induced” complications of COVID-19.
To minimize visits and hospitalizations, strategies include converting intravenous treatments to oral or subcutaneous regimens when possible; switching from cytotoxic chemotherapy to a less-toxic approach to minimize the risk of complications requiring hospitalization; or to pause therapies when possible (stable disease reached or better). In addition, nonemergency surgeries have been postponed or replaced by radiotherapy.
To prevent anticancer treatment-induced complications of COVID-19, most centers use the paradigm that the added benefit for tumor control should be weighed against the potential risk for COVID-19–related morbidity and mortality. To prevent or reduce the risk of neutropenia and lymphopenia, for example, all centers have suggested a de-escalation of cytotoxic chemotherapy or targeted treatment strategies, or to forgo second or subsequent lines of palliative treatments if response rates from up-front therapy are low.
Some of these changes may be here to stay, noted Caldas. “One of the positive messages that comes out of this is that, clearly, care can be delivered in a safe and compassionate manner without requiring as many hospital visits as in the pre-COVID-19 era,” he said. “In the future, we will take heed of the COVID-19 experience to improve delivery of cancer care.”
Capacity of facilities
Many healthcare systems have become overwhelmed as the pandemic has intensified, thus making it necessary to prioritize. To prepare for this possibility, CCE centers have established protocols to categorize and prioritize patients for systemic treatment or surgery. While the protocols vary by center, they are comparable with one another as they prioritize on the basis of anticipated treatment outcome, the authors note.
The guidelines in CCE centers unanimously recommend that neoadjuvant therapies and curative surgeries be the top priority, for the times when operating room and/or ICU capacity is limited. As an alternative, neoadjuvant systemic treatments may be initiated or extended to postpone surgery, and other nonsurgical interventions can be considered.
In addition, some centers agree that certain elective surgeries can be safely delayed if backed by scientific evidence. As an example, an 11-week deferment of surgery may be acceptable for patients with rectal cancer after downstaging.
Cancer centers may also need to upscale and downscale quickly, depending on how the pandemic evolves, and many have already outlined scenarios to prepare for increasing or decreasing their capacity using phased approaches.
The Netherlands Cancer Institute, for example, has defined four phases of increasing severity; in Germany, capacity planning has been coordinated among 18 hospitals and the federal ministry of health, in order to prevent shortages of cancer services.
“We note that the optimal downscaling strategies depend on country- and center-specific capacities and preferences,” they write. “Therefore, it is difficult to propose a common schedule, and it will be most effective if hospitals outline their own phase-specific downscaling strategies based on the prioritization schemes and practical handles discussed above.”
Future research
Better strategies will be needed to reduce the impact of COVID-19 in cancer care, and four research priorities were identified to allow for evidence-based adjustments of cancer care protocols while the pandemic continues:
- Collect real-world data about the effects of adjustment and de-escalation of treatment regimens on outcomes
- Determine the incidence of COVID-19 in both the general population and among patients with cancer who have received systemic therapies, with large-scale serological testing
- Develop an epidemiological model that will allow estimates of the cumulative incidence of COVID-19 for a patient with cancer, within a specific time frame
- Determine COVID-19 related morbidity and mortality in patients with cancer who have been treated with systemic therapies and/or granulocyte colony-stimulating factor (G-CSF). Several projects are currently underway, such as the UK Coronavirus Cancer Monitoring Project.
The authors have disclosed no relevant financial relationships.
This article first appeared on Medscape.com.
Prioritize lung cancer patients for COVID-19 testing, physicians recommend
Lung cancer patients should be prioritized for COVID-19 testing, according to an editorial published in Annals of Oncology.
In fact, treatment recommendations should call for baseline COVID-19 testing for all patients with lung cancer, Antonio Passaro, MD, PhD, of the European Institute of Oncology in Milan, Italy, and colleagues argued in the editorial.
“While all types of malignancies seem to be associated with high COVID-19 prevalence, morbidity, and mortality, lung cancer represents a specific scenario of cumulative risk factors for COVID-19 complications,” the authors wrote.
“[Lung cancer patients] are at a uniquely escalated risk of complications from COVID-19 due to the common features of smoking history, respiratory and cardiac disease, advanced age, and often predisposing risks from treatment, such as lung surgery and immunosuppressive chemotherapy,” said Howard (Jack) West, MD, of City of Hope Comprehensive Cancer Center in Duarte, Calif., who was not involved in the editorial.
“They also routinely experience a cough as well as chest imaging that may overlap between their underlying lung cancer, possible side effects of treatment, and potential COVID-19, leading to troubling ambiguity that can only be addressed by proactive and widespread testing of patients with lung cancer at the earliest opportunity and as a very high priority,” Dr. West added.
Dr. Passaro and colleagues’ editorial outlined these and other issues that suggest a need to prioritize testing in lung cancer patients.
Disease characteristics, treatment, and imaging
Lung cancer patients may have “defective pulmonary architecture,” such as mechanical obstruction from a tumor or previous lung surgery, that predisposes them to infection and can increase the risk of cytokine release. This is a concern because massive cytokine release during SARS-CoV-2 infection “has been postulated to be the major step in leading to the development of ARDS [acute respiratory distress syndrome],” Dr. Passaro and colleagues wrote.
The authors also argued that similar clinical symptoms among lung cancer patients and those with COVID-19 – such as cough, fever, and dyspnea – underscore the need for an accurate screening model to allow for early COVID-19 detection and potentially improve outcomes.
Similarly, lung cancer patients and COVID-19 patients may have overlapping findings on imaging. The radiologic effects of some common treatments for lung cancer can lead to the same kind of ground glass opacities and other findings seen in COVID-19 patients. Therefore, the authors predict an increase in “COVID-19-suspicious imaging, even in the absence of new symptoms” in the coming weeks.
Another issue to consider is the frequent use of corticosteroids in cancer patients. Corticosteroids may be harmful when used for COVID-19–related acute respiratory distress syndrome and could mask early symptoms of infection. Therefore, routine COVID-19 testing in patients receiving steroids may be warranted, according to Dr. Passaro and colleagues.
In addition, immunosuppression associated with cancer treatment “may impose specific consideration on the schedule and dose of cytotoxic chemotherapy for lung cancer patients in epidemic areas,” the authors wrote.
Increasing awareness: A registry and guidelines
“In the era of COVID-19, the optimal management of patients with lung cancer remains unknown, and the oncology community should have increased awareness to prevent the emergence of an increase in cancer-related and infectious mortality,” Dr. Passaro and colleagues wrote.
To that end, a novel global registry (TERAVOLT) has been launched and is collecting data worldwide with an aim of developing a tailored risk assessment strategy for lung cancer patients. The authors noted that developing international consensus with respect to COVID-19 testing in lung cancer is essential for achieving that goal.
The European Society for Medical Oncology recently released guidelines for treating lung cancer patients during the COVID-19 pandemic, but those guidelines do not include recommendations on COVID-19 testing.
“Baseline SARS-CoV-2 testing for all patients affected by lung cancer should be recommended,” Dr. Passaro and colleagues wrote. “In addition, for those patients with a negative swab test and new ground glass opacities detected on CT scan, with or without new respiratory symptoms, bronchoscopy should be considered to increase testing sensitivity.”
This work was partially supported by the Italian Ministry of Health. The authors reported having no relevant conflicts of interest. Dr. West is a regular correspondent for Medscape, which is owned by the same parent company as MDedge.
SOURCE: Passaro A et al. Annals of Oncology. doi: 10.1016/j.annonc.2020.04.002.
Lung cancer patients should be prioritized for COVID-19 testing, according to an editorial published in Annals of Oncology.
In fact, treatment recommendations should call for baseline COVID-19 testing for all patients with lung cancer, Antonio Passaro, MD, PhD, of the European Institute of Oncology in Milan, Italy, and colleagues argued in the editorial.
“While all types of malignancies seem to be associated with high COVID-19 prevalence, morbidity, and mortality, lung cancer represents a specific scenario of cumulative risk factors for COVID-19 complications,” the authors wrote.
“[Lung cancer patients] are at a uniquely escalated risk of complications from COVID-19 due to the common features of smoking history, respiratory and cardiac disease, advanced age, and often predisposing risks from treatment, such as lung surgery and immunosuppressive chemotherapy,” said Howard (Jack) West, MD, of City of Hope Comprehensive Cancer Center in Duarte, Calif., who was not involved in the editorial.
“They also routinely experience a cough as well as chest imaging that may overlap between their underlying lung cancer, possible side effects of treatment, and potential COVID-19, leading to troubling ambiguity that can only be addressed by proactive and widespread testing of patients with lung cancer at the earliest opportunity and as a very high priority,” Dr. West added.
Dr. Passaro and colleagues’ editorial outlined these and other issues that suggest a need to prioritize testing in lung cancer patients.
Disease characteristics, treatment, and imaging
Lung cancer patients may have “defective pulmonary architecture,” such as mechanical obstruction from a tumor or previous lung surgery, that predisposes them to infection and can increase the risk of cytokine release. This is a concern because massive cytokine release during SARS-CoV-2 infection “has been postulated to be the major step in leading to the development of ARDS [acute respiratory distress syndrome],” Dr. Passaro and colleagues wrote.
The authors also argued that similar clinical symptoms among lung cancer patients and those with COVID-19 – such as cough, fever, and dyspnea – underscore the need for an accurate screening model to allow for early COVID-19 detection and potentially improve outcomes.
Similarly, lung cancer patients and COVID-19 patients may have overlapping findings on imaging. The radiologic effects of some common treatments for lung cancer can lead to the same kind of ground glass opacities and other findings seen in COVID-19 patients. Therefore, the authors predict an increase in “COVID-19-suspicious imaging, even in the absence of new symptoms” in the coming weeks.
Another issue to consider is the frequent use of corticosteroids in cancer patients. Corticosteroids may be harmful when used for COVID-19–related acute respiratory distress syndrome and could mask early symptoms of infection. Therefore, routine COVID-19 testing in patients receiving steroids may be warranted, according to Dr. Passaro and colleagues.
In addition, immunosuppression associated with cancer treatment “may impose specific consideration on the schedule and dose of cytotoxic chemotherapy for lung cancer patients in epidemic areas,” the authors wrote.
Increasing awareness: A registry and guidelines
“In the era of COVID-19, the optimal management of patients with lung cancer remains unknown, and the oncology community should have increased awareness to prevent the emergence of an increase in cancer-related and infectious mortality,” Dr. Passaro and colleagues wrote.
To that end, a novel global registry (TERAVOLT) has been launched and is collecting data worldwide with an aim of developing a tailored risk assessment strategy for lung cancer patients. The authors noted that developing international consensus with respect to COVID-19 testing in lung cancer is essential for achieving that goal.
The European Society for Medical Oncology recently released guidelines for treating lung cancer patients during the COVID-19 pandemic, but those guidelines do not include recommendations on COVID-19 testing.
“Baseline SARS-CoV-2 testing for all patients affected by lung cancer should be recommended,” Dr. Passaro and colleagues wrote. “In addition, for those patients with a negative swab test and new ground glass opacities detected on CT scan, with or without new respiratory symptoms, bronchoscopy should be considered to increase testing sensitivity.”
This work was partially supported by the Italian Ministry of Health. The authors reported having no relevant conflicts of interest. Dr. West is a regular correspondent for Medscape, which is owned by the same parent company as MDedge.
SOURCE: Passaro A et al. Annals of Oncology. doi: 10.1016/j.annonc.2020.04.002.
Lung cancer patients should be prioritized for COVID-19 testing, according to an editorial published in Annals of Oncology.
In fact, treatment recommendations should call for baseline COVID-19 testing for all patients with lung cancer, Antonio Passaro, MD, PhD, of the European Institute of Oncology in Milan, Italy, and colleagues argued in the editorial.
“While all types of malignancies seem to be associated with high COVID-19 prevalence, morbidity, and mortality, lung cancer represents a specific scenario of cumulative risk factors for COVID-19 complications,” the authors wrote.
“[Lung cancer patients] are at a uniquely escalated risk of complications from COVID-19 due to the common features of smoking history, respiratory and cardiac disease, advanced age, and often predisposing risks from treatment, such as lung surgery and immunosuppressive chemotherapy,” said Howard (Jack) West, MD, of City of Hope Comprehensive Cancer Center in Duarte, Calif., who was not involved in the editorial.
“They also routinely experience a cough as well as chest imaging that may overlap between their underlying lung cancer, possible side effects of treatment, and potential COVID-19, leading to troubling ambiguity that can only be addressed by proactive and widespread testing of patients with lung cancer at the earliest opportunity and as a very high priority,” Dr. West added.
Dr. Passaro and colleagues’ editorial outlined these and other issues that suggest a need to prioritize testing in lung cancer patients.
Disease characteristics, treatment, and imaging
Lung cancer patients may have “defective pulmonary architecture,” such as mechanical obstruction from a tumor or previous lung surgery, that predisposes them to infection and can increase the risk of cytokine release. This is a concern because massive cytokine release during SARS-CoV-2 infection “has been postulated to be the major step in leading to the development of ARDS [acute respiratory distress syndrome],” Dr. Passaro and colleagues wrote.
The authors also argued that similar clinical symptoms among lung cancer patients and those with COVID-19 – such as cough, fever, and dyspnea – underscore the need for an accurate screening model to allow for early COVID-19 detection and potentially improve outcomes.
Similarly, lung cancer patients and COVID-19 patients may have overlapping findings on imaging. The radiologic effects of some common treatments for lung cancer can lead to the same kind of ground glass opacities and other findings seen in COVID-19 patients. Therefore, the authors predict an increase in “COVID-19-suspicious imaging, even in the absence of new symptoms” in the coming weeks.
Another issue to consider is the frequent use of corticosteroids in cancer patients. Corticosteroids may be harmful when used for COVID-19–related acute respiratory distress syndrome and could mask early symptoms of infection. Therefore, routine COVID-19 testing in patients receiving steroids may be warranted, according to Dr. Passaro and colleagues.
In addition, immunosuppression associated with cancer treatment “may impose specific consideration on the schedule and dose of cytotoxic chemotherapy for lung cancer patients in epidemic areas,” the authors wrote.
Increasing awareness: A registry and guidelines
“In the era of COVID-19, the optimal management of patients with lung cancer remains unknown, and the oncology community should have increased awareness to prevent the emergence of an increase in cancer-related and infectious mortality,” Dr. Passaro and colleagues wrote.
To that end, a novel global registry (TERAVOLT) has been launched and is collecting data worldwide with an aim of developing a tailored risk assessment strategy for lung cancer patients. The authors noted that developing international consensus with respect to COVID-19 testing in lung cancer is essential for achieving that goal.
The European Society for Medical Oncology recently released guidelines for treating lung cancer patients during the COVID-19 pandemic, but those guidelines do not include recommendations on COVID-19 testing.
“Baseline SARS-CoV-2 testing for all patients affected by lung cancer should be recommended,” Dr. Passaro and colleagues wrote. “In addition, for those patients with a negative swab test and new ground glass opacities detected on CT scan, with or without new respiratory symptoms, bronchoscopy should be considered to increase testing sensitivity.”
This work was partially supported by the Italian Ministry of Health. The authors reported having no relevant conflicts of interest. Dr. West is a regular correspondent for Medscape, which is owned by the same parent company as MDedge.
SOURCE: Passaro A et al. Annals of Oncology. doi: 10.1016/j.annonc.2020.04.002.
FROM ANNALS OF ONCOLOGY

