User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
U.S. reports record-breaking 1.35 million new COVID cases in a day
The United States reported 1.35 million new COVID-19 cases on Jan. 10, logging the highest daily total for any country in the world during the pandemic.
The United States set the previous record of 1 million cases on Jan. 3. (A large number of cases are reported on Mondays, since many states don’t provide updates over the weekend, according to Reuters.)
Still, the 7-day average for new cases has surpassed 700,000, tripling in 2 weeks as the contagious Omicron variant continues to spread across the country.
The daily record of new cases came a day after the United States crossed the grim milestone of 60 million COVID-19 cases during the pandemic, according to the latest data from Johns Hopkins University. More than 11 million new cases were reported in the past 28 days, with 5 million reported since Jan. 2.
Globally, more than 310 million cases have been reported, resulting in nearly 5.5 million COVID-19 deaths. Almost 40 million cases have been confirmed worldwide during the past month, with the United States accounting for 28% of those.
Texas became the second state to report more than 5 million cases since the pandemic began, behind California’s total of 6 million cases. Florida has reported more than 4.6 million, while New York has reported more than 4.1 million.
The United States has also hit an all-time high for hospitalizations, with nearly 146,000 COVID-19 patients in hospitals across the country, according to the latest data from the U.S. Department of Health and Human Services. The previous record was 142,000 hospitalizations in January 2021.
Jan. 11’s hospitalizations are more than twice as many as 2 weeks ago, according to CNN. About 78% of inpatient beds are in use nationwide, and 21% are being used for COVID-19 patients.
Deaths are averaging about 1,700 per day, Reuters reported, which is up from 1,400 in recent days but not much higher than earlier this winter. The peak average was 3,400 daily deaths in mid-January 2021.
The surging numbers of cases and hospitalizations across the country are straining hospitals. On Jan. 10, Virginia Gov. Ralph Northam declared a state of emergency after the number of intensive care unit hospitalizations more than doubled since Dec. 1, CNN reported. The order allows hospitals to expand bed capacity, use telehealth options, and be more flexible with staffing.
Texas is hiring at least 2,700 medical staff to help with the surge, CNN reported, and Kentucky has mobilized the National Guard to provide support.
“Omicron continues to burn through the commonwealth, growing at levels we have never seen before. Omicron is significantly more contagious than even the Delta variant,” Kentucky Gov. Andy Beshear said during a news briefing Jan. 10.
Kentucky reported its highest weekly total of cases last week and has its highest rate of positive tests, at 26%. Mr. Beshear said the state is down to 134 available adult ICU beds.
“If it spreads at the rate we are seeing, it is certainly going to fill up our hospitals,” he said.
A version of this article first appeared on WebMD.com.
The United States reported 1.35 million new COVID-19 cases on Jan. 10, logging the highest daily total for any country in the world during the pandemic.
The United States set the previous record of 1 million cases on Jan. 3. (A large number of cases are reported on Mondays, since many states don’t provide updates over the weekend, according to Reuters.)
Still, the 7-day average for new cases has surpassed 700,000, tripling in 2 weeks as the contagious Omicron variant continues to spread across the country.
The daily record of new cases came a day after the United States crossed the grim milestone of 60 million COVID-19 cases during the pandemic, according to the latest data from Johns Hopkins University. More than 11 million new cases were reported in the past 28 days, with 5 million reported since Jan. 2.
Globally, more than 310 million cases have been reported, resulting in nearly 5.5 million COVID-19 deaths. Almost 40 million cases have been confirmed worldwide during the past month, with the United States accounting for 28% of those.
Texas became the second state to report more than 5 million cases since the pandemic began, behind California’s total of 6 million cases. Florida has reported more than 4.6 million, while New York has reported more than 4.1 million.
The United States has also hit an all-time high for hospitalizations, with nearly 146,000 COVID-19 patients in hospitals across the country, according to the latest data from the U.S. Department of Health and Human Services. The previous record was 142,000 hospitalizations in January 2021.
Jan. 11’s hospitalizations are more than twice as many as 2 weeks ago, according to CNN. About 78% of inpatient beds are in use nationwide, and 21% are being used for COVID-19 patients.
Deaths are averaging about 1,700 per day, Reuters reported, which is up from 1,400 in recent days but not much higher than earlier this winter. The peak average was 3,400 daily deaths in mid-January 2021.
The surging numbers of cases and hospitalizations across the country are straining hospitals. On Jan. 10, Virginia Gov. Ralph Northam declared a state of emergency after the number of intensive care unit hospitalizations more than doubled since Dec. 1, CNN reported. The order allows hospitals to expand bed capacity, use telehealth options, and be more flexible with staffing.
Texas is hiring at least 2,700 medical staff to help with the surge, CNN reported, and Kentucky has mobilized the National Guard to provide support.
“Omicron continues to burn through the commonwealth, growing at levels we have never seen before. Omicron is significantly more contagious than even the Delta variant,” Kentucky Gov. Andy Beshear said during a news briefing Jan. 10.
Kentucky reported its highest weekly total of cases last week and has its highest rate of positive tests, at 26%. Mr. Beshear said the state is down to 134 available adult ICU beds.
“If it spreads at the rate we are seeing, it is certainly going to fill up our hospitals,” he said.
A version of this article first appeared on WebMD.com.
The United States reported 1.35 million new COVID-19 cases on Jan. 10, logging the highest daily total for any country in the world during the pandemic.
The United States set the previous record of 1 million cases on Jan. 3. (A large number of cases are reported on Mondays, since many states don’t provide updates over the weekend, according to Reuters.)
Still, the 7-day average for new cases has surpassed 700,000, tripling in 2 weeks as the contagious Omicron variant continues to spread across the country.
The daily record of new cases came a day after the United States crossed the grim milestone of 60 million COVID-19 cases during the pandemic, according to the latest data from Johns Hopkins University. More than 11 million new cases were reported in the past 28 days, with 5 million reported since Jan. 2.
Globally, more than 310 million cases have been reported, resulting in nearly 5.5 million COVID-19 deaths. Almost 40 million cases have been confirmed worldwide during the past month, with the United States accounting for 28% of those.
Texas became the second state to report more than 5 million cases since the pandemic began, behind California’s total of 6 million cases. Florida has reported more than 4.6 million, while New York has reported more than 4.1 million.
The United States has also hit an all-time high for hospitalizations, with nearly 146,000 COVID-19 patients in hospitals across the country, according to the latest data from the U.S. Department of Health and Human Services. The previous record was 142,000 hospitalizations in January 2021.
Jan. 11’s hospitalizations are more than twice as many as 2 weeks ago, according to CNN. About 78% of inpatient beds are in use nationwide, and 21% are being used for COVID-19 patients.
Deaths are averaging about 1,700 per day, Reuters reported, which is up from 1,400 in recent days but not much higher than earlier this winter. The peak average was 3,400 daily deaths in mid-January 2021.
The surging numbers of cases and hospitalizations across the country are straining hospitals. On Jan. 10, Virginia Gov. Ralph Northam declared a state of emergency after the number of intensive care unit hospitalizations more than doubled since Dec. 1, CNN reported. The order allows hospitals to expand bed capacity, use telehealth options, and be more flexible with staffing.
Texas is hiring at least 2,700 medical staff to help with the surge, CNN reported, and Kentucky has mobilized the National Guard to provide support.
“Omicron continues to burn through the commonwealth, growing at levels we have never seen before. Omicron is significantly more contagious than even the Delta variant,” Kentucky Gov. Andy Beshear said during a news briefing Jan. 10.
Kentucky reported its highest weekly total of cases last week and has its highest rate of positive tests, at 26%. Mr. Beshear said the state is down to 134 available adult ICU beds.
“If it spreads at the rate we are seeing, it is certainly going to fill up our hospitals,” he said.
A version of this article first appeared on WebMD.com.
What if the National Guard Can’t Help?
What if the National Guard Can’t Help?
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
In early January, Ohio not only set a state record for COVID-19 hospitalizations—it had the fourth highest rate in the country, with 6,747 hospitalized coronavirus patients on January 10, a 40% increase over the previous 21 days. Most were unvaccinated. To help overwhelmed hospitals cope, Ohio Gov. Mike DeWine turned to the National Guard. Unfortunately, nearly half of the Ohio National Guard also were unvaccinated.
By US Department of Defense (DoD) directive, National Guard members must have a COVID-19 vaccination to be deployed on hospital missions. Thus, in COVID hotspots across the nation, governors are on the horns of a dilemma. They want and need to deploy the National Guard to give medical and nonclinical support but aren’t sure whether they will be able to or, indeed, whether they should.
So far, vaccinated teams are already on the ground in a number of states. In Indiana, where hospitalizations jumped 50% over 2 weeks in December, the National Guard sent 6-person teams, all fully vaccinated. In New Hampshire, 70 guards are being deployed to help hospitals with food service, clerical work, and other nonmedical functions. New York Governor Kathy Hochul has deployed guard members for help to ease the strain on nursing homes. Massachusetts Governor Charlie Baker has activated up to 500 guard members; some will be supporting 55 acute care hospital and 12 ambulance services. In Maine, where cases have peaked, Governor Janet Mills activated guard members to support nursing facilities and administer monoclonal antibodies. The Louisiana National Guard has administered more than 542,000 COVID-19 tests and 206,300 vaccines. As many as 1,000 Maryland Air and Army National Guardsmen are being activated to help with testing and other missions.
However, as in Ohio, other states are facing problematic scenarios. For instance, about 40% of the more than 20,000 Texas National Guard are refusing to get vaccinated, challenging the Biden Administration vaccine requirement for all military.
And a court showdown over federal vaccine mandates, started by Governor Kevin Stitt of Oklahoma and joined by the Republican governors of Wyoming, Iowa, Alaska, Nebraska, and Mississippi, came to a head in December. Last November, Stitt asked Defense Secretary Lloyd Austin to exempt Oklahoma’s National Guard from the vaccine mandate. He claimed the requirement violated the personal freedoms of many Oklahomans and could cause them to “potentially sacrifice their personal beliefs.” But in a memo to the Joint Chiefs chairmen, the service secretaries and the head of the National Guard Bureau, Austin wrote that Pentagon funds could not be used to pay for duties performed under Title 32 for members of the Guard who do not comply with the military’s vaccine requirement. (Title 32 refers to Guard operations under state orders.) Austin also said National Guard members must be vaccinated to participate in drills, training, and other duty conducted under Title 32.
Stitt, maintaining that he is commander in chief of the Oklahoma National Guard as long as it operates under Title 32 orders, put out his own memo stipulating that no Guard member was required to get vaccinated. He also ordered Brig. Gen. Thomas Mancino, newly appointed commander of the Oklahoma National Guard, to not enforce the mandate. Subsequently, Mancino issued a statement pointing out that current state law is limited in protecting troops who opt out of the shot. Moreover, if the Guard were called up under federal orders, he said, he would enforce the mandate. Training events, schools, and mobilizations were going to “eventually force you out of that safe harbor,” he wrote, “…This is reality.”
In late December, a federal judge denied Oklahoma’s motion to enjoin the mandate. The Oklahoma Attorney General’s office responded, “We will not be surprised if the President’s vaccine mandate actually reduces the nation’s military readiness instead of promoting it.”
In a press briefing, Pentagon press secretary John Kirby said, “The Secretary has the authorities he needs to require this vaccine across the force, including the National Guard. …[E]ven when they’re in a Title 32 status.” He added, “It is a lawful order for National Guardsmen to receive the COVID vaccine. It’s a lawful order, and refusing to do that, absent of an improved exemption, puts them in the same potential [position] as active-duty members who refuse the vaccine.” That could mean, for instance, loss of pay and membership in the National Guard.
A core rationale for the mandate, according to Secretary Austin, is the need for military readiness—meaning Guard members must be healthy and fit for duty. And that extends to being healthy and fit for missions like transporting at-risk patients. Ohio National Guard Adjutant General Major General John Harris Jr. said, “I would never put a soldier or airman in harm’s way without the best protection we could put on them—body armor, helmets. And this medical readiness is the exact same thing. We’re putting folks into harm’s way.” He has moved the deadline from the Pentagon’s June 30 date to March 31—a move that boosted the vaccination rate from 53% to 56% in one week.
Ohio Governor DeWine has expressed frustration that almost half of the Ohio Army National Guard personnel can’t be deployed on this mission because they’re unvaccinated. “In some of our testing places, 40 to 50% of the people are testing positive,” he said. “So this is a high-risk operation. You need to be protected. The best way for you to be protected is to get the vaccination.”
As of December 2021, according to the National Guard Bureau, the National Guard as a whole was 66% fully vaccinated. The percentages vary according to service; for instance, nearly 90% of airmen have been vaccinated, compared with only 40% of Army Guardsmen. Among the states challenging the mandate, the vaccinated rates have been moving upward: In Alaska, about 92% of the Air National Guard have been vaccinated—leaving roughly 11,000 troops who had not met the December 2 deadline. In Iowa, as of Nov. 30, 91% of Air National Guard and 80% of Army National Guard members had been vaccinated, but about 9,000 soldiers had been directed to get the vaccination or risk disciplinary action. Almost 2,200 of the more than 2,800-strong Wyoming National Guard (77%) have received at least 1 dose. Nebraska Air National Guard’s force of 1,000 was 94% fully vaccinated as of December 1. (Maj Scott Ingalsbe, public affairs officer, said, “Vaccinations are tied to individual medical readiness. They provide service members with the best protection available so they can perform missions across the globe.”).
In most states, Army National Guard members have until June 30, 2022, to comply. “Our soldiers …have until [the DoD’s deadline], and some of them are just going to wait close to the deadline,” John Goheen of the National Guard Association of the United States said in a discussion on NPR. “That’s human nature.”
Earlier this month, Texas Governor Greg Abbott told National Guard members they can ignore the Pentagon’s COVID-19 vaccine mandate: “President Biden is not your commander-in-chief.” He has also sued the Biden administration over the requirement.
In the meantime, the hospitals at breaking point must hope for the best and take as much help as they can get.
What if the National Guard Can’t Help?
What if the National Guard Can’t Help?
Pig heart successfully transplanted to man
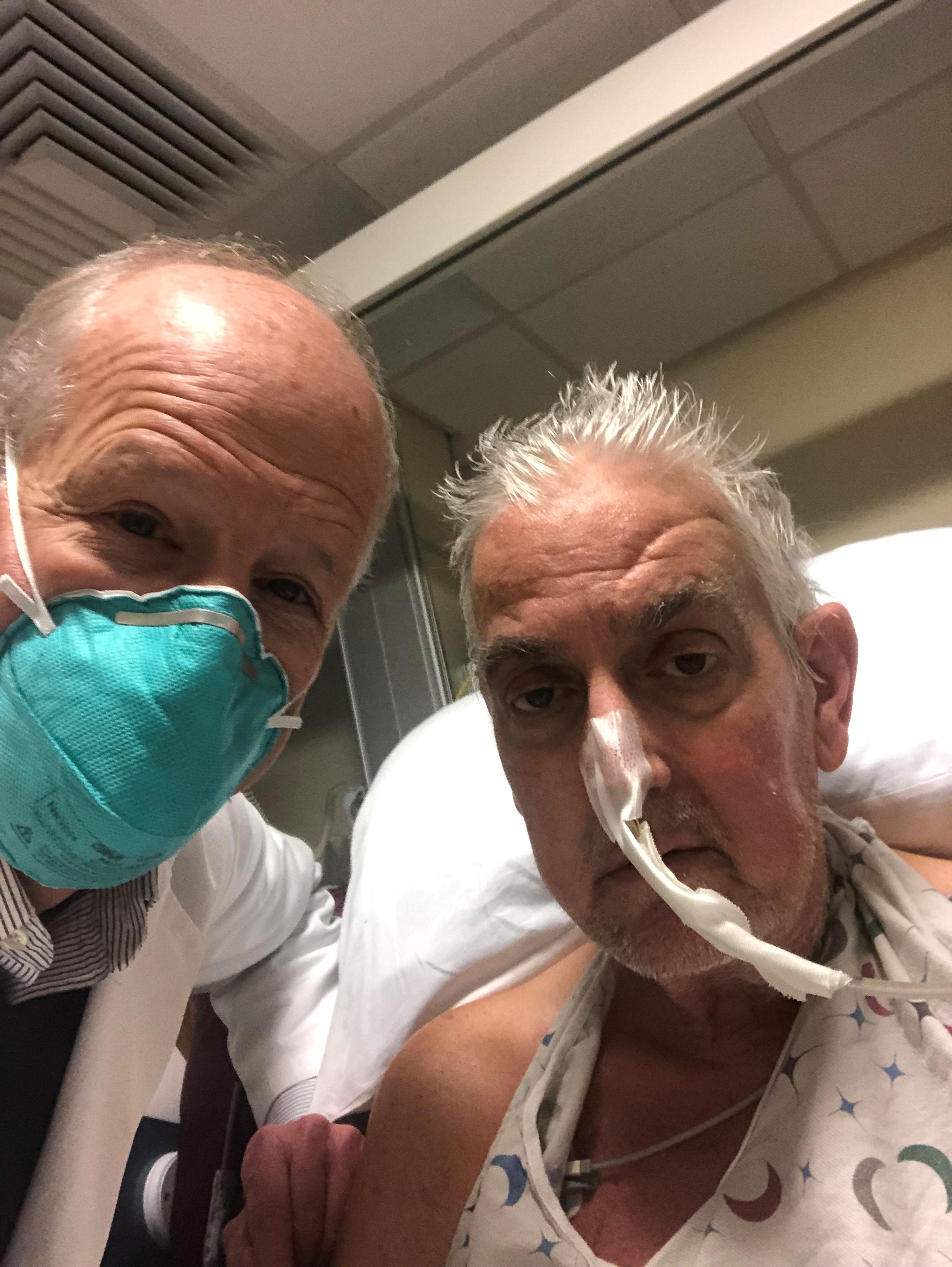
A genetically modified pig heart has been successfully transplanted into a 57-year-old man who had no other treatment options but is “doing well” 3 days after the procedure, officials at the University of Maryland Medical Center (UMMC), Baltimore, announced Jan. 10.
“This organ transplant demonstrated for the first time that a genetically modified animal heart can function like a human heart without immediate rejection by the body,” they said.
Three genes associated with antibody-mediated rejection had been knocked out in the pig supplying the transplanted heart, and six human genes associated with immune acceptance of the organ had been inserted into the pig’s genome, notes a UMMC press release.
“Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue, which totaled 10 unique gene edits made in the donor pig,” the release states.
The patient, Maryland resident David Bennett, had required mechanical circulatory support to stay alive but was rejected for standard heart transplantation at UMMC and other centers. He was ineligible for an implanted ventricular assist device due to ventricular arrhythmias.
Mr. Bennett “is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits,” the announcement says.
“We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” notes a quote from Bartley P. Griffith, MD, the UMMC surgeon who performed the procedure.
The pig supplying the heart was provided to the center by Revivicor (Blacksburg, Virginia), a regenerative medicine company. An experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Massachusetts) was also used, in addition to standard immunosuppressants.
A version of this article first appeared on Medscape.com.
A genetically modified pig heart has been successfully transplanted into a 57-year-old man who had no other treatment options but is “doing well” 3 days after the procedure, officials at the University of Maryland Medical Center (UMMC), Baltimore, announced Jan. 10.
“This organ transplant demonstrated for the first time that a genetically modified animal heart can function like a human heart without immediate rejection by the body,” they said.
Three genes associated with antibody-mediated rejection had been knocked out in the pig supplying the transplanted heart, and six human genes associated with immune acceptance of the organ had been inserted into the pig’s genome, notes a UMMC press release.
“Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue, which totaled 10 unique gene edits made in the donor pig,” the release states.
The patient, Maryland resident David Bennett, had required mechanical circulatory support to stay alive but was rejected for standard heart transplantation at UMMC and other centers. He was ineligible for an implanted ventricular assist device due to ventricular arrhythmias.
Mr. Bennett “is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits,” the announcement says.
“We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” notes a quote from Bartley P. Griffith, MD, the UMMC surgeon who performed the procedure.
The pig supplying the heart was provided to the center by Revivicor (Blacksburg, Virginia), a regenerative medicine company. An experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Massachusetts) was also used, in addition to standard immunosuppressants.
A version of this article first appeared on Medscape.com.
A genetically modified pig heart has been successfully transplanted into a 57-year-old man who had no other treatment options but is “doing well” 3 days after the procedure, officials at the University of Maryland Medical Center (UMMC), Baltimore, announced Jan. 10.
“This organ transplant demonstrated for the first time that a genetically modified animal heart can function like a human heart without immediate rejection by the body,” they said.
Three genes associated with antibody-mediated rejection had been knocked out in the pig supplying the transplanted heart, and six human genes associated with immune acceptance of the organ had been inserted into the pig’s genome, notes a UMMC press release.
“Lastly, one additional gene in the pig was knocked out to prevent excessive growth of the pig heart tissue, which totaled 10 unique gene edits made in the donor pig,” the release states.
The patient, Maryland resident David Bennett, had required mechanical circulatory support to stay alive but was rejected for standard heart transplantation at UMMC and other centers. He was ineligible for an implanted ventricular assist device due to ventricular arrhythmias.
Mr. Bennett “is being carefully monitored over the next days and weeks to determine whether the transplant provides lifesaving benefits,” the announcement says.
“We are proceeding cautiously, but we are also optimistic that this first-in-the-world surgery will provide an important new option for patients in the future,” notes a quote from Bartley P. Griffith, MD, the UMMC surgeon who performed the procedure.
The pig supplying the heart was provided to the center by Revivicor (Blacksburg, Virginia), a regenerative medicine company. An experimental antirejection medication (Kiniksa Pharmaceuticals; Lexington, Massachusetts) was also used, in addition to standard immunosuppressants.
A version of this article first appeared on Medscape.com.
Is outpatient care as safe as inpatient for TIA, minor stroke?
In a meta-analysis of more than 200,000 patients with TIA or mIS, risk for subsequent stroke within 90 days was 2.1% for those treated in a TIA clinic versus 2.8% for patients treated in inpatient settings, which was not significantly different. The risk for patients treated in an emergency department was higher, at 3.5%.
“The message is that if you do the correct risk stratification and then triage patients based on their risk profile, you can safely discharge and have a timely follow-up for the patients who have low risk for a subsequent event,” said coinvestigator Ramin Zand, MD, vascular neurologist and stroke attending physician at Geisinger Health System, Danville, Pennsylvania.
The findings were published online Jan. 5 in JAMA Network Open.
Higher risk in EDs
There is currently no consensus on the care protocol for patients with TIA or mIS, and the rate at which these patients are hospitalized varies by region, hospital, and practitioner, the investigators noted.
Previous studies have indicated that outpatient management of certain individuals with TIA can be safe and cost-effective.
The current researchers searched for retrospective and prospective studies of adult patients that provided information about ischemic stroke after TIA or mIS. Studies that used time- and tissue-based definitions of TIA were included, as well as studies that used various definitions of mIS.
The investigators examined care provided at TIA clinics, inpatient settings (such as medical-surgical units, stroke units, or observation units), EDs, and unspecified settings. Their main aim was to compare outcomes between TIA clinics and inpatient settings.
In all, 226,683 patients (recruited between 1981 and 2018) from 71 studies were included in the meta-analysis. The studies examined 101 cohorts, 24 of which were studied prospectively. Among the 5,636 patients who received care in TIA clinics, the mean age was 65.7 years, and 50.8% of this group were men. Among the 130,139 inpatients, the mean age was 78.3 years, and 61.6% of the group were women.
Results showed no significant difference in risk for subsequent stroke between patients treated in the inpatient and outpatient settings.
Among patients treated in a TIA clinic, risk for subsequent stroke following a TIA or mIS was 0.3% within 2 days, 1.0% within 7 days, 1.3% within 30 days, and 2.1% within 90 days. Among those treated as inpatients, risk for subsequent stroke was 0.5% within 2 days, 1.2% within 7 days, 1.6% within 30 days, and 2.8% within 90 days.
Risk for subsequent stroke was higher among patients treated in the ED and in unspecified settings. At the EDs, the risk was 1.9% within 2 days, 3.4% within 7 days, 3.5% within 30 days, and 3.5% within 90 days. Among those treated in unspecified settings, the risk was 2.2% within 2 days, 3.4% within 7 days, 4.2% within 30 days, and 6.0% within 90 days.
Patients treated in the ED also had a significantly higher risk for subsequent stroke at 2 and 7 days, compared with those treated in inpatient settings and a significantly higher risk for subsequent stroke at 2, 7, and 90 days, compared with those treated in TIA clinics.
‘Most comprehensive look’
“This is the most comprehensive look at all the studies to try and answer this research question,” said Dr. Zand. The results were similar to what was expected, he added.
The infrastructure and resources differed among the sites at which the various studies were conducted, and the investigators adjusted for these differences as much as possible, Dr. Zand noted. A certain amount of selection bias may remain, but it does not affect the overall conclusion, he added.
“Timely outpatient care among low-risk TIA patients is both feasible and safe,” he said.
Dr. Zand noted that the findings have implications not only for patient management but also for the management of the health system. “It’s not feasible nor desirable to admit all the TIA patients, especially with the lessons that we learned from COVID, the burden on the health systems, and the fact that many hospitals are operating at full capacity right now,” he said.
The recommendation is to hospitalize high-risk patients and provide outpatient evaluation and workup to low-risk patients, he added. “This is exactly what we saw in this study,” Dr. Zand said.
Selection bias?
Commenting on the research, Louis R. Caplan, MD, professor of neurology at Harvard Medical School, Boston, noted that evaluation of patients with TIA or mIS “can be done very well as an outpatient” if clinicians have experienced personnel, the outpatient facilities to do the studies necessary, and criteria in place for deciding who to admit or not admit.
However, the decision on whether to choose an inpatient or outpatient approach for a particular patient is complicated, said Dr. Caplan, who was not involved with the research.
Clinicians must consider factors such as whether the patient is mobile, has a car, or has a significant other. The patient’s symptoms and past illnesses also influence the decision, he added.
Dr. Caplan noted that in the meta-analysis, far fewer patients were seen in the TIA clinics than were seen in the inpatient setting. In addition, none of the studies used uniform criteria to determine which patients should undergo workup as outpatients and which as inpatients. “There was a lot of selection bias that may have had nothing to do with how sick the person was,” Dr. Caplan said.
In addition, few hospitals in the United States have an outpatient TIA clinic, he noted. Most of the studies of TIA clinics that the researchers examined were conducted in Europe. “It’s easier to do [that] in Europe because of their socialized medicine,” said Dr. Caplan.
But TIA clinics should be more widespread in the U.S., he added. “Insurance companies should be willing to pay for comparable facilities, inpatient and outpatient,” he said.
The study was conducted without external funding. Dr. Zand reported no relevant financial relationships. Dr. Caplan was an investigator for TIAregistry.org, which analyzed the outcomes of treatment in TIA clinics in Europe.
A version of this article first appeared on Medscape.com.
In a meta-analysis of more than 200,000 patients with TIA or mIS, risk for subsequent stroke within 90 days was 2.1% for those treated in a TIA clinic versus 2.8% for patients treated in inpatient settings, which was not significantly different. The risk for patients treated in an emergency department was higher, at 3.5%.
“The message is that if you do the correct risk stratification and then triage patients based on their risk profile, you can safely discharge and have a timely follow-up for the patients who have low risk for a subsequent event,” said coinvestigator Ramin Zand, MD, vascular neurologist and stroke attending physician at Geisinger Health System, Danville, Pennsylvania.
The findings were published online Jan. 5 in JAMA Network Open.
Higher risk in EDs
There is currently no consensus on the care protocol for patients with TIA or mIS, and the rate at which these patients are hospitalized varies by region, hospital, and practitioner, the investigators noted.
Previous studies have indicated that outpatient management of certain individuals with TIA can be safe and cost-effective.
The current researchers searched for retrospective and prospective studies of adult patients that provided information about ischemic stroke after TIA or mIS. Studies that used time- and tissue-based definitions of TIA were included, as well as studies that used various definitions of mIS.
The investigators examined care provided at TIA clinics, inpatient settings (such as medical-surgical units, stroke units, or observation units), EDs, and unspecified settings. Their main aim was to compare outcomes between TIA clinics and inpatient settings.
In all, 226,683 patients (recruited between 1981 and 2018) from 71 studies were included in the meta-analysis. The studies examined 101 cohorts, 24 of which were studied prospectively. Among the 5,636 patients who received care in TIA clinics, the mean age was 65.7 years, and 50.8% of this group were men. Among the 130,139 inpatients, the mean age was 78.3 years, and 61.6% of the group were women.
Results showed no significant difference in risk for subsequent stroke between patients treated in the inpatient and outpatient settings.
Among patients treated in a TIA clinic, risk for subsequent stroke following a TIA or mIS was 0.3% within 2 days, 1.0% within 7 days, 1.3% within 30 days, and 2.1% within 90 days. Among those treated as inpatients, risk for subsequent stroke was 0.5% within 2 days, 1.2% within 7 days, 1.6% within 30 days, and 2.8% within 90 days.
Risk for subsequent stroke was higher among patients treated in the ED and in unspecified settings. At the EDs, the risk was 1.9% within 2 days, 3.4% within 7 days, 3.5% within 30 days, and 3.5% within 90 days. Among those treated in unspecified settings, the risk was 2.2% within 2 days, 3.4% within 7 days, 4.2% within 30 days, and 6.0% within 90 days.
Patients treated in the ED also had a significantly higher risk for subsequent stroke at 2 and 7 days, compared with those treated in inpatient settings and a significantly higher risk for subsequent stroke at 2, 7, and 90 days, compared with those treated in TIA clinics.
‘Most comprehensive look’
“This is the most comprehensive look at all the studies to try and answer this research question,” said Dr. Zand. The results were similar to what was expected, he added.
The infrastructure and resources differed among the sites at which the various studies were conducted, and the investigators adjusted for these differences as much as possible, Dr. Zand noted. A certain amount of selection bias may remain, but it does not affect the overall conclusion, he added.
“Timely outpatient care among low-risk TIA patients is both feasible and safe,” he said.
Dr. Zand noted that the findings have implications not only for patient management but also for the management of the health system. “It’s not feasible nor desirable to admit all the TIA patients, especially with the lessons that we learned from COVID, the burden on the health systems, and the fact that many hospitals are operating at full capacity right now,” he said.
The recommendation is to hospitalize high-risk patients and provide outpatient evaluation and workup to low-risk patients, he added. “This is exactly what we saw in this study,” Dr. Zand said.
Selection bias?
Commenting on the research, Louis R. Caplan, MD, professor of neurology at Harvard Medical School, Boston, noted that evaluation of patients with TIA or mIS “can be done very well as an outpatient” if clinicians have experienced personnel, the outpatient facilities to do the studies necessary, and criteria in place for deciding who to admit or not admit.
However, the decision on whether to choose an inpatient or outpatient approach for a particular patient is complicated, said Dr. Caplan, who was not involved with the research.
Clinicians must consider factors such as whether the patient is mobile, has a car, or has a significant other. The patient’s symptoms and past illnesses also influence the decision, he added.
Dr. Caplan noted that in the meta-analysis, far fewer patients were seen in the TIA clinics than were seen in the inpatient setting. In addition, none of the studies used uniform criteria to determine which patients should undergo workup as outpatients and which as inpatients. “There was a lot of selection bias that may have had nothing to do with how sick the person was,” Dr. Caplan said.
In addition, few hospitals in the United States have an outpatient TIA clinic, he noted. Most of the studies of TIA clinics that the researchers examined were conducted in Europe. “It’s easier to do [that] in Europe because of their socialized medicine,” said Dr. Caplan.
But TIA clinics should be more widespread in the U.S., he added. “Insurance companies should be willing to pay for comparable facilities, inpatient and outpatient,” he said.
The study was conducted without external funding. Dr. Zand reported no relevant financial relationships. Dr. Caplan was an investigator for TIAregistry.org, which analyzed the outcomes of treatment in TIA clinics in Europe.
A version of this article first appeared on Medscape.com.
In a meta-analysis of more than 200,000 patients with TIA or mIS, risk for subsequent stroke within 90 days was 2.1% for those treated in a TIA clinic versus 2.8% for patients treated in inpatient settings, which was not significantly different. The risk for patients treated in an emergency department was higher, at 3.5%.
“The message is that if you do the correct risk stratification and then triage patients based on their risk profile, you can safely discharge and have a timely follow-up for the patients who have low risk for a subsequent event,” said coinvestigator Ramin Zand, MD, vascular neurologist and stroke attending physician at Geisinger Health System, Danville, Pennsylvania.
The findings were published online Jan. 5 in JAMA Network Open.
Higher risk in EDs
There is currently no consensus on the care protocol for patients with TIA or mIS, and the rate at which these patients are hospitalized varies by region, hospital, and practitioner, the investigators noted.
Previous studies have indicated that outpatient management of certain individuals with TIA can be safe and cost-effective.
The current researchers searched for retrospective and prospective studies of adult patients that provided information about ischemic stroke after TIA or mIS. Studies that used time- and tissue-based definitions of TIA were included, as well as studies that used various definitions of mIS.
The investigators examined care provided at TIA clinics, inpatient settings (such as medical-surgical units, stroke units, or observation units), EDs, and unspecified settings. Their main aim was to compare outcomes between TIA clinics and inpatient settings.
In all, 226,683 patients (recruited between 1981 and 2018) from 71 studies were included in the meta-analysis. The studies examined 101 cohorts, 24 of which were studied prospectively. Among the 5,636 patients who received care in TIA clinics, the mean age was 65.7 years, and 50.8% of this group were men. Among the 130,139 inpatients, the mean age was 78.3 years, and 61.6% of the group were women.
Results showed no significant difference in risk for subsequent stroke between patients treated in the inpatient and outpatient settings.
Among patients treated in a TIA clinic, risk for subsequent stroke following a TIA or mIS was 0.3% within 2 days, 1.0% within 7 days, 1.3% within 30 days, and 2.1% within 90 days. Among those treated as inpatients, risk for subsequent stroke was 0.5% within 2 days, 1.2% within 7 days, 1.6% within 30 days, and 2.8% within 90 days.
Risk for subsequent stroke was higher among patients treated in the ED and in unspecified settings. At the EDs, the risk was 1.9% within 2 days, 3.4% within 7 days, 3.5% within 30 days, and 3.5% within 90 days. Among those treated in unspecified settings, the risk was 2.2% within 2 days, 3.4% within 7 days, 4.2% within 30 days, and 6.0% within 90 days.
Patients treated in the ED also had a significantly higher risk for subsequent stroke at 2 and 7 days, compared with those treated in inpatient settings and a significantly higher risk for subsequent stroke at 2, 7, and 90 days, compared with those treated in TIA clinics.
‘Most comprehensive look’
“This is the most comprehensive look at all the studies to try and answer this research question,” said Dr. Zand. The results were similar to what was expected, he added.
The infrastructure and resources differed among the sites at which the various studies were conducted, and the investigators adjusted for these differences as much as possible, Dr. Zand noted. A certain amount of selection bias may remain, but it does not affect the overall conclusion, he added.
“Timely outpatient care among low-risk TIA patients is both feasible and safe,” he said.
Dr. Zand noted that the findings have implications not only for patient management but also for the management of the health system. “It’s not feasible nor desirable to admit all the TIA patients, especially with the lessons that we learned from COVID, the burden on the health systems, and the fact that many hospitals are operating at full capacity right now,” he said.
The recommendation is to hospitalize high-risk patients and provide outpatient evaluation and workup to low-risk patients, he added. “This is exactly what we saw in this study,” Dr. Zand said.
Selection bias?
Commenting on the research, Louis R. Caplan, MD, professor of neurology at Harvard Medical School, Boston, noted that evaluation of patients with TIA or mIS “can be done very well as an outpatient” if clinicians have experienced personnel, the outpatient facilities to do the studies necessary, and criteria in place for deciding who to admit or not admit.
However, the decision on whether to choose an inpatient or outpatient approach for a particular patient is complicated, said Dr. Caplan, who was not involved with the research.
Clinicians must consider factors such as whether the patient is mobile, has a car, or has a significant other. The patient’s symptoms and past illnesses also influence the decision, he added.
Dr. Caplan noted that in the meta-analysis, far fewer patients were seen in the TIA clinics than were seen in the inpatient setting. In addition, none of the studies used uniform criteria to determine which patients should undergo workup as outpatients and which as inpatients. “There was a lot of selection bias that may have had nothing to do with how sick the person was,” Dr. Caplan said.
In addition, few hospitals in the United States have an outpatient TIA clinic, he noted. Most of the studies of TIA clinics that the researchers examined were conducted in Europe. “It’s easier to do [that] in Europe because of their socialized medicine,” said Dr. Caplan.
But TIA clinics should be more widespread in the U.S., he added. “Insurance companies should be willing to pay for comparable facilities, inpatient and outpatient,” he said.
The study was conducted without external funding. Dr. Zand reported no relevant financial relationships. Dr. Caplan was an investigator for TIAregistry.org, which analyzed the outcomes of treatment in TIA clinics in Europe.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
COVID-vaccine myocarditis: Rare, mild, and usually in young men
The risk of myocarditis after immunization with mRNA-based vaccines against SARS-CoV-2 raised concerns when it came to light in early 2021. But as report after report showed such cases to be rare and usually mild and self-limited, focus has turned to the “how and why.”
The mechanism linking the BNT162b2 (Pfizer-BioNTech) and especially mRNA-1273 (Moderna) vaccines to the occurrence of myocarditis is unclear for now, but one potential driver may be tied to a peculiarity that became apparent early: It occurs overwhelmingly in younger males, from 16 to perhaps 40 or 50 years of age. Excess risk has not been consistently seen among women, girls, and older men.
That observation has led to speculation that higher testosterone levels in adolescent boys and young men may somehow promote the adverse vaccine effect, whereas greater levels of estrogen among girls and women in the same age range may be cardioprotective.
Unlikely, brief, and ‘benign’
“Most of the myocarditis is benign, by which I mean that maybe the patients are admitted due to chest pain, but without reduction in ventricular function,” Enrico Ammirati, MD, PhD, a myocarditis expert at De Gasperis Cardio Center and Transplant Center, Niguarda Hospital, Milan, said in an interview.
In a Nov. 14 address on this topic at the annual scientific sessions of the American Heart Association, Dror Mevorach, MD, described the typical case presentation as “mild” and one that clears in fairly short order based on resolution of “clinical symptoms, inflammatory markers and troponin decline, EKG normalization, echo normalization, and a relatively short length of hospital stay.”
Dr. Mevorach, of Hadassah Hebrew University Medical Center, Jerusalem, subsequently published the findings in a report in the New England Journal of Medicine that described 136 confirmed myocarditis cases among more than 5 million people in Israel immunized with the Pfizer-BioNTech vaccine. Myocarditis was considered “mild” in 129 cases, or 95%.
And the risk is tiny, compared with myocarditis from infection by SARS-CoV-2, not to mention the possibility of nasty clinical COVID-19 complications such as pneumonia and pulmonary embolism, Dr. Mevorach observed.
Many other reports agree that the incidence is minimal, especially given the rewards of vaccination. In a separate NEJM publication in September 2021 – from Noam Barda, MD, Clalit (Israel) Research Institute, and colleagues on 1.7 million people in that country, about half unvaccinated and half given the Pfizer-BioNTech vaccine – there were an estimated 2.7 cases of myocarditis per 100,000 vaccinated persons. There were also 11 cases of myocarditis per 100,000 persons who were positive for SARS-CoV-2 infection.
And in a recent case series of vaccinated people aged 16 or older, the myocarditis rate after a first or second Pfizer-BioNTech or Moderna injection was estimated at 1 or fewer per 100,000. The corresponding estimate was 4 such cases per 100,000 after a positive SARS-CoV-2 test among the same population, notes a report published Dec.14, 2021, in Nature Medicine.
In general, “the risk of any kind of cardiac injury is vastly lower with a vaccine than it is with the actual viral infection,” Leslie T. Cooper Jr., MD, a myocarditis expert and clinical trialist at the Mayo Clinic, Jacksonville, Fla., said in an interview. With the mRNA-based vaccines, “we do not have any conceivable danger signal that would outweigh the benefit of vaccination.”
Males of a certain age
Evidence that such myocarditis predominates in young adult men and adolescent boys, especially following a second vaccine dose, is remarkably consistent.
The risk was elevated only among mRNA-based vaccine recipients who were younger than 40 in the recent Nature Medicine analysis. Among that group, estimates after a second dose numbered fewer than 1 case per 100,000 for Pfizer-BioNTech and 1.5 per 100,000 for Moderna.
In a third analysis from Israel – also in NEJM, from Guy Witberg, MD, Rabin Medical Center, Petah Tikva, and colleagues, based on 2.5 million people aged 16 and older with at least one Pfizer-BioNTech injection – 2.1 cases per 100,000 were estimated overall, but the number rose to 10.7 per 100,000 among those aged 16-29 years.
In Dr. Mevorach’s NEJM report, estimates after a second Pfizer-BioNTech vaccine dose were 1 per 26,000 males versus 1 in 218,000 females, compared with 1 myocarditis case in 10,857 persons among “the general unvaccinated population.”
Most recipients of a first vaccine dose were younger than 50, and 16- to 29-year-olds accounted for most who completed two doses, noted Dr. Mevorach. Younger males bore the brunt of any myocarditis: the estimated prevalence after a second dose among males aged 16-19 was 1 per 6,637, compared with 1 per 99,853 females in the same age range, the group reported.
In the BMJ report, based on about 5 million people 12 years of age or older in Denmark, the estimated rates of myocarditis or pericarditis associated with Moderna immunization were 2 per 100,000 among women but 6.3 per 100,000 for men. The incidence and sex difference was much lower among those getting the Pfizer-BioNTech vaccine: 1.3 per 100,000 and 1.5 per 100,000 in women and men, respectively.
Sex hormones may be key
The predominance of vaccine-associated myocarditis among adolescent and young adult males is probably more about the myocarditis itself than the vaccines, observed Biykem Bozkurt, MD, PhD, who has been studying COVID-related myocarditis at Baylor College of Medicine, Houston.
Male sex historically is associated in both epidemiologic studies and experimental models with a greater propensity for most any form of myocarditis, Dr. Bozkurt said in an interview. Given that males aged 16-19 or so appear to be at highest risk of myocarditis as a complication of SARS-CoV-2 vaccination, the mechanism may well be related to sex hormones.
“Therefore, testosterone is implicated as a player in their higher risk of inflammation and injury and lack of adaptive response in terms of healing, and in terms of prevention of injury,” Dr. Bozkurt said. For its part, estrogen inhibits proinflammatory processes and, in particular, “blunts cell-mediated immune responses.”
“We don’t know the mechanism, but a theory that attributes a protective role to estrogen, or a risk associated with testosterone, is reasonable. It makes sense, at least based on epidemiological data,” Dr. Ammirati agreed. Still, “we do not have any direct evidence in human beings.”
Sex-associated differences in experimental myocarditis have been reported in the journals for at least 70 years, but “the testosterone literature and the estrogen literature have not been evaluated in detail in vaccine-associated myocarditis,” Dr. Cooper said.
Most myocarditis in the laboratory is viral, Dr. Cooper observed, and “the links between testosterone, viruses, and inflammation have been pretty well worked out, I would say, if you’re a mouse. If you’re a human, I think it’s still a bit uncertain.”
Were it to apply in humans, greater testosterone levels might independently promote myocarditis, “and if estrogen is cardioprotective, it would be another mechanism,” Dr. Cooper said. “That would translate to slight male predominance in most kinds of myocarditis.”
In males, compared with females, “the heart can be more vulnerable to events such as arrhythmias or to immune-mediated phenomena. So, probably there is also higher vulnerability to myocarditis in men,” Dr. Ammirati noted.
Male predominance in vaccine-related myocarditis is provocative, so it’s worth considering whether testosterone is part of the mechanism as well as the possibility of estrogen cardioprotection, Dr. Ammirati said. But given limitations of the animal models, “we don’t really have robust data to support any part of that.”
Although myocarditis is in some way immune mediated, “and hormones can modulate the response,” the mechanism has to be more than just sex hormones, he said. “They probably cannot explain the specificity for the heart. It’s not a systemic response, it’s an organ-specific response.”
Modulation of immune responses
Details about the immune processes underlying mRNA-vaccine myocarditis, hormone modulated or not, have been elusive. The complication doesn’t resemble serum sickness, nor does it seem to be a reaction to infection by other cardiotropic viruses, such as coxsackie virus B, a cause of viral myocarditis, Dr. Bozkurt said. The latter had been a compelling possibility because such hypersensitivity to smallpox vaccination is well recognized.
“We don’t know the mechanism, that’s the short answer. But there are many hypotheses,” she said. One candidate widely proposed in the literature: autoantibodies driven by molecular mimicry between the SARS-CoV-2 spike protein targeted by the mRNA vaccines and a structurally similar myocardial protein, possibly alpha-myosin, noted Dr. Bozkurt and colleagues in a recent publication.
But elevations in specific “antiheart antibodies” have not been documented in recipients of the two mRNA-based vaccines, said Dr. Cooper. “So, I would say that – although molecular mimicry is a well-established mechanism of, for example, rheumatic carditis after a streptococcal A infection – that has not been demonstrated yet for COVID-19 mRNA vaccination–related myocarditis.”
“We probably won’t know, ever, with a huge level of certainty, the exact mechanisms,” Dr. Cooper added. There is no animal model for vaccine-induced myocarditis, and “We’re still talking very, very small numbers of patients. The vast majority of them recover,” and so don’t generally provide mechanistic clues.
Prospects for younger children
Vaccination against SARS-CoV-2 has now been authorized by the Centers for Disease Control and Prevention for kids as young as 5-11 years, using the Pfizer-BioNTech vaccine. Experience so far suggests the immunization is safe in that age group with negligible risk of myocarditis or other complications. But with prospects of possible authorization in children younger than 5, should myocarditis be a concern for them?
Probably not, if the complication is driven primarily by sex hormones, Dr. Cooper proposed. “One would predict that before puberty you would have a lower – much, much lower – rate of myocarditis in males than you would in the 16- to 19-year-old range, and that it would be roughly equal to females.” Dr. Ammirati and Dr. Bozkurt largely agreed.
It remains to be seen whether the vaccine-related myocarditis risk applies to children younger than 12, “but I doubt it. I think it’s going to be puberty-related,” Dr. Bozkurt said. Still, “I don’t want to hypothesize without data.”
A version of this article first appeared on Medscape.com.
The risk of myocarditis after immunization with mRNA-based vaccines against SARS-CoV-2 raised concerns when it came to light in early 2021. But as report after report showed such cases to be rare and usually mild and self-limited, focus has turned to the “how and why.”
The mechanism linking the BNT162b2 (Pfizer-BioNTech) and especially mRNA-1273 (Moderna) vaccines to the occurrence of myocarditis is unclear for now, but one potential driver may be tied to a peculiarity that became apparent early: It occurs overwhelmingly in younger males, from 16 to perhaps 40 or 50 years of age. Excess risk has not been consistently seen among women, girls, and older men.
That observation has led to speculation that higher testosterone levels in adolescent boys and young men may somehow promote the adverse vaccine effect, whereas greater levels of estrogen among girls and women in the same age range may be cardioprotective.
Unlikely, brief, and ‘benign’
“Most of the myocarditis is benign, by which I mean that maybe the patients are admitted due to chest pain, but without reduction in ventricular function,” Enrico Ammirati, MD, PhD, a myocarditis expert at De Gasperis Cardio Center and Transplant Center, Niguarda Hospital, Milan, said in an interview.
In a Nov. 14 address on this topic at the annual scientific sessions of the American Heart Association, Dror Mevorach, MD, described the typical case presentation as “mild” and one that clears in fairly short order based on resolution of “clinical symptoms, inflammatory markers and troponin decline, EKG normalization, echo normalization, and a relatively short length of hospital stay.”
Dr. Mevorach, of Hadassah Hebrew University Medical Center, Jerusalem, subsequently published the findings in a report in the New England Journal of Medicine that described 136 confirmed myocarditis cases among more than 5 million people in Israel immunized with the Pfizer-BioNTech vaccine. Myocarditis was considered “mild” in 129 cases, or 95%.
And the risk is tiny, compared with myocarditis from infection by SARS-CoV-2, not to mention the possibility of nasty clinical COVID-19 complications such as pneumonia and pulmonary embolism, Dr. Mevorach observed.
Many other reports agree that the incidence is minimal, especially given the rewards of vaccination. In a separate NEJM publication in September 2021 – from Noam Barda, MD, Clalit (Israel) Research Institute, and colleagues on 1.7 million people in that country, about half unvaccinated and half given the Pfizer-BioNTech vaccine – there were an estimated 2.7 cases of myocarditis per 100,000 vaccinated persons. There were also 11 cases of myocarditis per 100,000 persons who were positive for SARS-CoV-2 infection.
And in a recent case series of vaccinated people aged 16 or older, the myocarditis rate after a first or second Pfizer-BioNTech or Moderna injection was estimated at 1 or fewer per 100,000. The corresponding estimate was 4 such cases per 100,000 after a positive SARS-CoV-2 test among the same population, notes a report published Dec.14, 2021, in Nature Medicine.
In general, “the risk of any kind of cardiac injury is vastly lower with a vaccine than it is with the actual viral infection,” Leslie T. Cooper Jr., MD, a myocarditis expert and clinical trialist at the Mayo Clinic, Jacksonville, Fla., said in an interview. With the mRNA-based vaccines, “we do not have any conceivable danger signal that would outweigh the benefit of vaccination.”
Males of a certain age
Evidence that such myocarditis predominates in young adult men and adolescent boys, especially following a second vaccine dose, is remarkably consistent.
The risk was elevated only among mRNA-based vaccine recipients who were younger than 40 in the recent Nature Medicine analysis. Among that group, estimates after a second dose numbered fewer than 1 case per 100,000 for Pfizer-BioNTech and 1.5 per 100,000 for Moderna.
In a third analysis from Israel – also in NEJM, from Guy Witberg, MD, Rabin Medical Center, Petah Tikva, and colleagues, based on 2.5 million people aged 16 and older with at least one Pfizer-BioNTech injection – 2.1 cases per 100,000 were estimated overall, but the number rose to 10.7 per 100,000 among those aged 16-29 years.
In Dr. Mevorach’s NEJM report, estimates after a second Pfizer-BioNTech vaccine dose were 1 per 26,000 males versus 1 in 218,000 females, compared with 1 myocarditis case in 10,857 persons among “the general unvaccinated population.”
Most recipients of a first vaccine dose were younger than 50, and 16- to 29-year-olds accounted for most who completed two doses, noted Dr. Mevorach. Younger males bore the brunt of any myocarditis: the estimated prevalence after a second dose among males aged 16-19 was 1 per 6,637, compared with 1 per 99,853 females in the same age range, the group reported.
In the BMJ report, based on about 5 million people 12 years of age or older in Denmark, the estimated rates of myocarditis or pericarditis associated with Moderna immunization were 2 per 100,000 among women but 6.3 per 100,000 for men. The incidence and sex difference was much lower among those getting the Pfizer-BioNTech vaccine: 1.3 per 100,000 and 1.5 per 100,000 in women and men, respectively.
Sex hormones may be key
The predominance of vaccine-associated myocarditis among adolescent and young adult males is probably more about the myocarditis itself than the vaccines, observed Biykem Bozkurt, MD, PhD, who has been studying COVID-related myocarditis at Baylor College of Medicine, Houston.
Male sex historically is associated in both epidemiologic studies and experimental models with a greater propensity for most any form of myocarditis, Dr. Bozkurt said in an interview. Given that males aged 16-19 or so appear to be at highest risk of myocarditis as a complication of SARS-CoV-2 vaccination, the mechanism may well be related to sex hormones.
“Therefore, testosterone is implicated as a player in their higher risk of inflammation and injury and lack of adaptive response in terms of healing, and in terms of prevention of injury,” Dr. Bozkurt said. For its part, estrogen inhibits proinflammatory processes and, in particular, “blunts cell-mediated immune responses.”
“We don’t know the mechanism, but a theory that attributes a protective role to estrogen, or a risk associated with testosterone, is reasonable. It makes sense, at least based on epidemiological data,” Dr. Ammirati agreed. Still, “we do not have any direct evidence in human beings.”
Sex-associated differences in experimental myocarditis have been reported in the journals for at least 70 years, but “the testosterone literature and the estrogen literature have not been evaluated in detail in vaccine-associated myocarditis,” Dr. Cooper said.
Most myocarditis in the laboratory is viral, Dr. Cooper observed, and “the links between testosterone, viruses, and inflammation have been pretty well worked out, I would say, if you’re a mouse. If you’re a human, I think it’s still a bit uncertain.”
Were it to apply in humans, greater testosterone levels might independently promote myocarditis, “and if estrogen is cardioprotective, it would be another mechanism,” Dr. Cooper said. “That would translate to slight male predominance in most kinds of myocarditis.”
In males, compared with females, “the heart can be more vulnerable to events such as arrhythmias or to immune-mediated phenomena. So, probably there is also higher vulnerability to myocarditis in men,” Dr. Ammirati noted.
Male predominance in vaccine-related myocarditis is provocative, so it’s worth considering whether testosterone is part of the mechanism as well as the possibility of estrogen cardioprotection, Dr. Ammirati said. But given limitations of the animal models, “we don’t really have robust data to support any part of that.”
Although myocarditis is in some way immune mediated, “and hormones can modulate the response,” the mechanism has to be more than just sex hormones, he said. “They probably cannot explain the specificity for the heart. It’s not a systemic response, it’s an organ-specific response.”
Modulation of immune responses
Details about the immune processes underlying mRNA-vaccine myocarditis, hormone modulated or not, have been elusive. The complication doesn’t resemble serum sickness, nor does it seem to be a reaction to infection by other cardiotropic viruses, such as coxsackie virus B, a cause of viral myocarditis, Dr. Bozkurt said. The latter had been a compelling possibility because such hypersensitivity to smallpox vaccination is well recognized.
“We don’t know the mechanism, that’s the short answer. But there are many hypotheses,” she said. One candidate widely proposed in the literature: autoantibodies driven by molecular mimicry between the SARS-CoV-2 spike protein targeted by the mRNA vaccines and a structurally similar myocardial protein, possibly alpha-myosin, noted Dr. Bozkurt and colleagues in a recent publication.
But elevations in specific “antiheart antibodies” have not been documented in recipients of the two mRNA-based vaccines, said Dr. Cooper. “So, I would say that – although molecular mimicry is a well-established mechanism of, for example, rheumatic carditis after a streptococcal A infection – that has not been demonstrated yet for COVID-19 mRNA vaccination–related myocarditis.”
“We probably won’t know, ever, with a huge level of certainty, the exact mechanisms,” Dr. Cooper added. There is no animal model for vaccine-induced myocarditis, and “We’re still talking very, very small numbers of patients. The vast majority of them recover,” and so don’t generally provide mechanistic clues.
Prospects for younger children
Vaccination against SARS-CoV-2 has now been authorized by the Centers for Disease Control and Prevention for kids as young as 5-11 years, using the Pfizer-BioNTech vaccine. Experience so far suggests the immunization is safe in that age group with negligible risk of myocarditis or other complications. But with prospects of possible authorization in children younger than 5, should myocarditis be a concern for them?
Probably not, if the complication is driven primarily by sex hormones, Dr. Cooper proposed. “One would predict that before puberty you would have a lower – much, much lower – rate of myocarditis in males than you would in the 16- to 19-year-old range, and that it would be roughly equal to females.” Dr. Ammirati and Dr. Bozkurt largely agreed.
It remains to be seen whether the vaccine-related myocarditis risk applies to children younger than 12, “but I doubt it. I think it’s going to be puberty-related,” Dr. Bozkurt said. Still, “I don’t want to hypothesize without data.”
A version of this article first appeared on Medscape.com.
The risk of myocarditis after immunization with mRNA-based vaccines against SARS-CoV-2 raised concerns when it came to light in early 2021. But as report after report showed such cases to be rare and usually mild and self-limited, focus has turned to the “how and why.”
The mechanism linking the BNT162b2 (Pfizer-BioNTech) and especially mRNA-1273 (Moderna) vaccines to the occurrence of myocarditis is unclear for now, but one potential driver may be tied to a peculiarity that became apparent early: It occurs overwhelmingly in younger males, from 16 to perhaps 40 or 50 years of age. Excess risk has not been consistently seen among women, girls, and older men.
That observation has led to speculation that higher testosterone levels in adolescent boys and young men may somehow promote the adverse vaccine effect, whereas greater levels of estrogen among girls and women in the same age range may be cardioprotective.
Unlikely, brief, and ‘benign’
“Most of the myocarditis is benign, by which I mean that maybe the patients are admitted due to chest pain, but without reduction in ventricular function,” Enrico Ammirati, MD, PhD, a myocarditis expert at De Gasperis Cardio Center and Transplant Center, Niguarda Hospital, Milan, said in an interview.
In a Nov. 14 address on this topic at the annual scientific sessions of the American Heart Association, Dror Mevorach, MD, described the typical case presentation as “mild” and one that clears in fairly short order based on resolution of “clinical symptoms, inflammatory markers and troponin decline, EKG normalization, echo normalization, and a relatively short length of hospital stay.”
Dr. Mevorach, of Hadassah Hebrew University Medical Center, Jerusalem, subsequently published the findings in a report in the New England Journal of Medicine that described 136 confirmed myocarditis cases among more than 5 million people in Israel immunized with the Pfizer-BioNTech vaccine. Myocarditis was considered “mild” in 129 cases, or 95%.
And the risk is tiny, compared with myocarditis from infection by SARS-CoV-2, not to mention the possibility of nasty clinical COVID-19 complications such as pneumonia and pulmonary embolism, Dr. Mevorach observed.
Many other reports agree that the incidence is minimal, especially given the rewards of vaccination. In a separate NEJM publication in September 2021 – from Noam Barda, MD, Clalit (Israel) Research Institute, and colleagues on 1.7 million people in that country, about half unvaccinated and half given the Pfizer-BioNTech vaccine – there were an estimated 2.7 cases of myocarditis per 100,000 vaccinated persons. There were also 11 cases of myocarditis per 100,000 persons who were positive for SARS-CoV-2 infection.
And in a recent case series of vaccinated people aged 16 or older, the myocarditis rate after a first or second Pfizer-BioNTech or Moderna injection was estimated at 1 or fewer per 100,000. The corresponding estimate was 4 such cases per 100,000 after a positive SARS-CoV-2 test among the same population, notes a report published Dec.14, 2021, in Nature Medicine.
In general, “the risk of any kind of cardiac injury is vastly lower with a vaccine than it is with the actual viral infection,” Leslie T. Cooper Jr., MD, a myocarditis expert and clinical trialist at the Mayo Clinic, Jacksonville, Fla., said in an interview. With the mRNA-based vaccines, “we do not have any conceivable danger signal that would outweigh the benefit of vaccination.”
Males of a certain age
Evidence that such myocarditis predominates in young adult men and adolescent boys, especially following a second vaccine dose, is remarkably consistent.
The risk was elevated only among mRNA-based vaccine recipients who were younger than 40 in the recent Nature Medicine analysis. Among that group, estimates after a second dose numbered fewer than 1 case per 100,000 for Pfizer-BioNTech and 1.5 per 100,000 for Moderna.
In a third analysis from Israel – also in NEJM, from Guy Witberg, MD, Rabin Medical Center, Petah Tikva, and colleagues, based on 2.5 million people aged 16 and older with at least one Pfizer-BioNTech injection – 2.1 cases per 100,000 were estimated overall, but the number rose to 10.7 per 100,000 among those aged 16-29 years.
In Dr. Mevorach’s NEJM report, estimates after a second Pfizer-BioNTech vaccine dose were 1 per 26,000 males versus 1 in 218,000 females, compared with 1 myocarditis case in 10,857 persons among “the general unvaccinated population.”
Most recipients of a first vaccine dose were younger than 50, and 16- to 29-year-olds accounted for most who completed two doses, noted Dr. Mevorach. Younger males bore the brunt of any myocarditis: the estimated prevalence after a second dose among males aged 16-19 was 1 per 6,637, compared with 1 per 99,853 females in the same age range, the group reported.
In the BMJ report, based on about 5 million people 12 years of age or older in Denmark, the estimated rates of myocarditis or pericarditis associated with Moderna immunization were 2 per 100,000 among women but 6.3 per 100,000 for men. The incidence and sex difference was much lower among those getting the Pfizer-BioNTech vaccine: 1.3 per 100,000 and 1.5 per 100,000 in women and men, respectively.
Sex hormones may be key
The predominance of vaccine-associated myocarditis among adolescent and young adult males is probably more about the myocarditis itself than the vaccines, observed Biykem Bozkurt, MD, PhD, who has been studying COVID-related myocarditis at Baylor College of Medicine, Houston.
Male sex historically is associated in both epidemiologic studies and experimental models with a greater propensity for most any form of myocarditis, Dr. Bozkurt said in an interview. Given that males aged 16-19 or so appear to be at highest risk of myocarditis as a complication of SARS-CoV-2 vaccination, the mechanism may well be related to sex hormones.
“Therefore, testosterone is implicated as a player in their higher risk of inflammation and injury and lack of adaptive response in terms of healing, and in terms of prevention of injury,” Dr. Bozkurt said. For its part, estrogen inhibits proinflammatory processes and, in particular, “blunts cell-mediated immune responses.”
“We don’t know the mechanism, but a theory that attributes a protective role to estrogen, or a risk associated with testosterone, is reasonable. It makes sense, at least based on epidemiological data,” Dr. Ammirati agreed. Still, “we do not have any direct evidence in human beings.”
Sex-associated differences in experimental myocarditis have been reported in the journals for at least 70 years, but “the testosterone literature and the estrogen literature have not been evaluated in detail in vaccine-associated myocarditis,” Dr. Cooper said.
Most myocarditis in the laboratory is viral, Dr. Cooper observed, and “the links between testosterone, viruses, and inflammation have been pretty well worked out, I would say, if you’re a mouse. If you’re a human, I think it’s still a bit uncertain.”
Were it to apply in humans, greater testosterone levels might independently promote myocarditis, “and if estrogen is cardioprotective, it would be another mechanism,” Dr. Cooper said. “That would translate to slight male predominance in most kinds of myocarditis.”
In males, compared with females, “the heart can be more vulnerable to events such as arrhythmias or to immune-mediated phenomena. So, probably there is also higher vulnerability to myocarditis in men,” Dr. Ammirati noted.
Male predominance in vaccine-related myocarditis is provocative, so it’s worth considering whether testosterone is part of the mechanism as well as the possibility of estrogen cardioprotection, Dr. Ammirati said. But given limitations of the animal models, “we don’t really have robust data to support any part of that.”
Although myocarditis is in some way immune mediated, “and hormones can modulate the response,” the mechanism has to be more than just sex hormones, he said. “They probably cannot explain the specificity for the heart. It’s not a systemic response, it’s an organ-specific response.”
Modulation of immune responses
Details about the immune processes underlying mRNA-vaccine myocarditis, hormone modulated or not, have been elusive. The complication doesn’t resemble serum sickness, nor does it seem to be a reaction to infection by other cardiotropic viruses, such as coxsackie virus B, a cause of viral myocarditis, Dr. Bozkurt said. The latter had been a compelling possibility because such hypersensitivity to smallpox vaccination is well recognized.
“We don’t know the mechanism, that’s the short answer. But there are many hypotheses,” she said. One candidate widely proposed in the literature: autoantibodies driven by molecular mimicry between the SARS-CoV-2 spike protein targeted by the mRNA vaccines and a structurally similar myocardial protein, possibly alpha-myosin, noted Dr. Bozkurt and colleagues in a recent publication.
But elevations in specific “antiheart antibodies” have not been documented in recipients of the two mRNA-based vaccines, said Dr. Cooper. “So, I would say that – although molecular mimicry is a well-established mechanism of, for example, rheumatic carditis after a streptococcal A infection – that has not been demonstrated yet for COVID-19 mRNA vaccination–related myocarditis.”
“We probably won’t know, ever, with a huge level of certainty, the exact mechanisms,” Dr. Cooper added. There is no animal model for vaccine-induced myocarditis, and “We’re still talking very, very small numbers of patients. The vast majority of them recover,” and so don’t generally provide mechanistic clues.
Prospects for younger children
Vaccination against SARS-CoV-2 has now been authorized by the Centers for Disease Control and Prevention for kids as young as 5-11 years, using the Pfizer-BioNTech vaccine. Experience so far suggests the immunization is safe in that age group with negligible risk of myocarditis or other complications. But with prospects of possible authorization in children younger than 5, should myocarditis be a concern for them?
Probably not, if the complication is driven primarily by sex hormones, Dr. Cooper proposed. “One would predict that before puberty you would have a lower – much, much lower – rate of myocarditis in males than you would in the 16- to 19-year-old range, and that it would be roughly equal to females.” Dr. Ammirati and Dr. Bozkurt largely agreed.
It remains to be seen whether the vaccine-related myocarditis risk applies to children younger than 12, “but I doubt it. I think it’s going to be puberty-related,” Dr. Bozkurt said. Still, “I don’t want to hypothesize without data.”
A version of this article first appeared on Medscape.com.
AHA advice for diabetes patients to stay heart healthy
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
A new document from the American Heart Association summarizes the latest research on cardiovascular risk factor management in type 2 diabetes, including medications, lifestyle, and social determinants of health.
Despite the availability of effective therapies for improving cardiovascular risk, in the United States fewer than one in five people with type 2 diabetes and without known cardiovascular disease meet control targets for a combination of A1c, blood pressure, LDL cholesterol, and nonsmoking status.
That proportion drops to less than 1 in 10 if body mass index less than 30 kg/m2 is included among the targets, and even less than that among individuals with established atherosclerotic cardiovascular disease, Joshua J. Joseph, MD, and colleagues point out in their paper, published online Jan. 10 in Circulation.
“This new scientific statement is an urgent call to action to follow the latest evidence-based approaches and to develop new best practices to advance type 2 diabetes treatment and care and reduce cardiovascular disease risk,” wrote Dr. Joseph, assistant professor of medicine in the division of endocrinology, diabetes, and metabolism at The Ohio State University, Columbus, Ohio, and coauthors.
The statement is not a guideline but an expert analysis that may inform future clinical practice guidelines, according to a press release from the AHA.
The new statement reviews evidence through June 2020 for lifestyle management of diabetes and weight, glycemic targets and control, blood pressure management, lipid management, antithrombotic therapy, and screening for cardiovascular and renal complications, including imaging. It also discusses the clinical implications of recent cardiovascular outcomes trials of newer glucose-lowering medications.
However, Dr. Joseph and colleagues point out, clinical care and treatment account for just 10%-20% of modifiable contributors to health outcomes. The other 80%-90% relate to social determinants of health, including health-related behaviors, socioeconomic factors, environmental factors, and racism.
“If we are to continue to advance the management of cardiovascular risk factors, we must also address the [social determinants of health] in the delivery of health care,” they noted.
Overall, they advise a patient-centered approach, meaning “reframing our clinical encounters to think about patients as people who live in families, communities, and societies that must be considered in their cardiovascular risk management.”
“People with [type 2 diabetes] face numerous barriers to health including access to care and equitable care, which must be considered when developing individualized care plans with our patients,” Dr. Joseph said in the AHA press release.
Lifestyle, medications for lowering A1c, BP, lipids
For lifestyle management, the authors say, “culturally appropriate recommendations through diabetes self-management education and support and medical nutrition therapy are key to meeting individualized goals for behavioral change and diabetes self-management.”
The document summarizes recommendations from other professional societies regarding glycemic targets and glucose lowering medications, i.e., target A1c levels of either < 7% or < 6.5% for the majority, with adjustments based on individual factors, such as life expectancy. It advises on use of metformin as first-line therapy followed by a sodium-glucose cotransporter-2 inhibitor or a glucagon-like peptide-1 agonist for those with established cardiovascular disease or risk factors.
“Cost may be a barrier to taking some [type 2 diabetes] medications as prescribed; however, many of these medications are now more commonly covered by more health insurance plans,” Dr. Joseph said.
“Another barrier is recognition by patients that these newer [type 2 diabetes] medications are also effective in reducing the risk of heart disease, stroke, heart failure, and kidney disease.”
Blood pressure treatment guidelines differ between those of the AHA/American College of Cardiology (ACC) and the American Diabetes Association (ADA), most notably that the AHA/ACC guidelines advise a general target of < 130/80 mm Hg, whereas ADA advises < 140/90 mm Hg or < 130/80 mm Hg for those with high risk if it can be safely achieved.
The decision should be “patient-centered with shared decision-making,” Dr. Joseph and colleagues advised.
For lipid-lowering, the document cites the 2018 ACC/AHA cholesterol guidelines, which include advising statins as first-line therapy for both primary and secondary prevention in diabetes, with highest intensity statins used in those at highest risk. But again, treatment should be individualized, and other agents should be used for patients in whom statins don’t work or aren’t tolerated.
And while use of antiplatelets – that is, aspirin – is well established as secondary prevention in type 2 diabetes, given new data suggesting that the risk for major bleeding could outweigh the benefits for primary prevention, “the relative benefits of antithrombotic approaches need to be weighed carefully against risks using a patient-centered approach,” the authors advised.
Among the many imaging tests available to facilitate cardiovascular risk stratification in type 2 diabetes, coronary artery calcification (CAC) CT screening is one of the few with sufficient data to support routine use in selected patients. The National Lipid Association, for example, recommends escalation to high-intensity statin for CAC > 100.
“One avenue to continue to address and advance diabetes management is through breaking down the four walls of the clinic or hospital through community engagement, clinic-to-community connections, and academic-community-government partnerships that may help address and support modifiable lifestyle behaviors such as physical activity, nutrition, smoking cessation and stress management,” Dr. Joseph concluded.
The AHA receives funding primarily from individuals. Foundations and corporations, including pharmaceutical, device manufacturers, and other companies, also make donations and fund AHA programs and events. The AHA’s strict policies prevent these relationships from influencing the science content. Revenues from pharmaceutical and biotech companies, device manufacturers, and health insurance providers and the AHA’s financial information are available on the association’s website. Dr. Joseph has disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
At-risk Americans become eligible for fourth COVID shot this week
The Centers for Disease Control and Prevention endorsed a third dose of the Pfizer or Moderna vaccines for moderately and severely immunocompromised people on Aug. 13, which is considered part of their first immunization series rather than a booster shot.
In October, the CDC said moderately and severely immunocompromised people could receive a booster shot, or a fourth dose of the vaccine , 6 months after their third dose.
But the CDC last week shortened the timeline to 5 months for a booster shot of the Pfizer or Moderna vaccines. That means immunocompromised people could begin signing up for a fourth shot later this week, the New York Times reported.
About 2.7% of U.S. adults, or about 7 million adults, are considered immunocompromised, according to the CDC. They’re more likely to contract severe COVID-19, have a higher risk for long COVID, have lower antibody levels after vaccination, and develop serious breakthrough infections. About 40% of hospitalized breakthrough cases are among immunocompromised people.
According to CDC guidance, people are considered to be “moderately or severely immunocompromised” if they have:
- Active cancer treatment for tumors or cancers of the blood
- Had an organ transplant and are taking medicine to suppress the immune system
- Had a stem cell transplant in the last 2 years and are taking medicine to suppress the immune system
- Advanced or untreated HIV infection
- Moderate or severe primary immunodeficiency, such as DiGeorge syndrome or Wiskott-Aldrich syndrome
- Active treatment with high-dose corticosteroids or other drugs that suppress the immune response
So far, only moderately and severely immunocompromised Americans are eligible for a fourth shot. Israel has begun offering fourth doses to high-risk groups, including older adults, but the Biden administration hasn’t yet said whether the United States will follow, the Times reported.
Overall, the focus remains on getting third shots to Americans who are eligible for boosters, Rochelle Walensky, MD, the CDC director, told reporters Jan. 7. U.S. officials will remain in touch with Israel to follow their data on fourth shots.
“We will be following our own data carefully as well, to see how these boosters are working in terms of waning effectiveness, not just for infection but, importantly, for severe disease,” she said.
A version of this article first appeared on WebMD.com .
The Centers for Disease Control and Prevention endorsed a third dose of the Pfizer or Moderna vaccines for moderately and severely immunocompromised people on Aug. 13, which is considered part of their first immunization series rather than a booster shot.
In October, the CDC said moderately and severely immunocompromised people could receive a booster shot, or a fourth dose of the vaccine , 6 months after their third dose.
But the CDC last week shortened the timeline to 5 months for a booster shot of the Pfizer or Moderna vaccines. That means immunocompromised people could begin signing up for a fourth shot later this week, the New York Times reported.
About 2.7% of U.S. adults, or about 7 million adults, are considered immunocompromised, according to the CDC. They’re more likely to contract severe COVID-19, have a higher risk for long COVID, have lower antibody levels after vaccination, and develop serious breakthrough infections. About 40% of hospitalized breakthrough cases are among immunocompromised people.
According to CDC guidance, people are considered to be “moderately or severely immunocompromised” if they have:
- Active cancer treatment for tumors or cancers of the blood
- Had an organ transplant and are taking medicine to suppress the immune system
- Had a stem cell transplant in the last 2 years and are taking medicine to suppress the immune system
- Advanced or untreated HIV infection
- Moderate or severe primary immunodeficiency, such as DiGeorge syndrome or Wiskott-Aldrich syndrome
- Active treatment with high-dose corticosteroids or other drugs that suppress the immune response
So far, only moderately and severely immunocompromised Americans are eligible for a fourth shot. Israel has begun offering fourth doses to high-risk groups, including older adults, but the Biden administration hasn’t yet said whether the United States will follow, the Times reported.
Overall, the focus remains on getting third shots to Americans who are eligible for boosters, Rochelle Walensky, MD, the CDC director, told reporters Jan. 7. U.S. officials will remain in touch with Israel to follow their data on fourth shots.
“We will be following our own data carefully as well, to see how these boosters are working in terms of waning effectiveness, not just for infection but, importantly, for severe disease,” she said.
A version of this article first appeared on WebMD.com .
The Centers for Disease Control and Prevention endorsed a third dose of the Pfizer or Moderna vaccines for moderately and severely immunocompromised people on Aug. 13, which is considered part of their first immunization series rather than a booster shot.
In October, the CDC said moderately and severely immunocompromised people could receive a booster shot, or a fourth dose of the vaccine , 6 months after their third dose.
But the CDC last week shortened the timeline to 5 months for a booster shot of the Pfizer or Moderna vaccines. That means immunocompromised people could begin signing up for a fourth shot later this week, the New York Times reported.
About 2.7% of U.S. adults, or about 7 million adults, are considered immunocompromised, according to the CDC. They’re more likely to contract severe COVID-19, have a higher risk for long COVID, have lower antibody levels after vaccination, and develop serious breakthrough infections. About 40% of hospitalized breakthrough cases are among immunocompromised people.
According to CDC guidance, people are considered to be “moderately or severely immunocompromised” if they have:
- Active cancer treatment for tumors or cancers of the blood
- Had an organ transplant and are taking medicine to suppress the immune system
- Had a stem cell transplant in the last 2 years and are taking medicine to suppress the immune system
- Advanced or untreated HIV infection
- Moderate or severe primary immunodeficiency, such as DiGeorge syndrome or Wiskott-Aldrich syndrome
- Active treatment with high-dose corticosteroids or other drugs that suppress the immune response
So far, only moderately and severely immunocompromised Americans are eligible for a fourth shot. Israel has begun offering fourth doses to high-risk groups, including older adults, but the Biden administration hasn’t yet said whether the United States will follow, the Times reported.
Overall, the focus remains on getting third shots to Americans who are eligible for boosters, Rochelle Walensky, MD, the CDC director, told reporters Jan. 7. U.S. officials will remain in touch with Israel to follow their data on fourth shots.
“We will be following our own data carefully as well, to see how these boosters are working in terms of waning effectiveness, not just for infection but, importantly, for severe disease,” she said.
A version of this article first appeared on WebMD.com .
Lack of high school education vaccine hesitancy predictor
Lack of a high school education is a predictor of whether a person will be resistant to getting the COVID-19 vaccine, a new study shows.
Researchers from the University of North Carolina looked at vaccination rates in 3,142 counties in the U.S. They compared them to population characteristics based on the CDC Social Vulnerability Index.
They found that more than half of the unvaccinated adults in the U.S. with strong vaccine hesitancy had a high school education or less. Vaccine hesitancy was defined as refusal to be vaccinated even if the COVID-19 vaccine was available.
The other main predictor for vaccine hesitancy was concern about vaccine availability and distribution, the researchers said.
“Our study suggests that low education levels are a major contributor to vaccine hesitancy and ultimately vaccination levels,” the authors wrote. The study was published in the American Journal of Infection Control. “Specifically, low vaccination levels were found in communities with a less educated population and with more concern about vaccine uptake capacity, suggesting that education is an ongoing challenge.”
“Our findings suggest that policy makers and community leaders should tailor vaccine information and efforts to those with limited education and specifically address knowledge concerns that are prevalent and likely more modifiable.”
The study was based on data gathered months ago. It says that as of May 9, 2021, 34.7% of the U.S. population was fully vaccinated and that 8% reported a strong unwillingness to get vaccinated.
At press time, the Centers for Disease Control and Prevention’s COVID Data Tracker showed that 62.5% of the U.S. population was fully vaccinated.
According to the study, other consistent characteristics of people who are vaccine hesitant are that they belong to a racial minority, are 65 or older, live in a household with children 18 or younger, or are unemployed.
When asked why they were vaccine hesitant, people gave these reasons: Lack of trust in COVID-19 vaccines (55%), concerns about side effects (48%), and lack of trust in government (46%).
Lack of access to vaccines, often cited in previous studies about resistance to other vaccines, was not cited as a reason for not getting the COVID-19 vaccine.
“COVID-19 vaccine hesitancy is a public health threat,” the researchers concluded. “Since education levels are not easily modifiable, our results suggest that policymakers would be best served by closing knowledge gaps to overcome negative perceptions of the vaccine through tailored interventions.”
A version of this article first appeared on WebMD.com.
Lack of a high school education is a predictor of whether a person will be resistant to getting the COVID-19 vaccine, a new study shows.
Researchers from the University of North Carolina looked at vaccination rates in 3,142 counties in the U.S. They compared them to population characteristics based on the CDC Social Vulnerability Index.
They found that more than half of the unvaccinated adults in the U.S. with strong vaccine hesitancy had a high school education or less. Vaccine hesitancy was defined as refusal to be vaccinated even if the COVID-19 vaccine was available.
The other main predictor for vaccine hesitancy was concern about vaccine availability and distribution, the researchers said.
“Our study suggests that low education levels are a major contributor to vaccine hesitancy and ultimately vaccination levels,” the authors wrote. The study was published in the American Journal of Infection Control. “Specifically, low vaccination levels were found in communities with a less educated population and with more concern about vaccine uptake capacity, suggesting that education is an ongoing challenge.”
“Our findings suggest that policy makers and community leaders should tailor vaccine information and efforts to those with limited education and specifically address knowledge concerns that are prevalent and likely more modifiable.”
The study was based on data gathered months ago. It says that as of May 9, 2021, 34.7% of the U.S. population was fully vaccinated and that 8% reported a strong unwillingness to get vaccinated.
At press time, the Centers for Disease Control and Prevention’s COVID Data Tracker showed that 62.5% of the U.S. population was fully vaccinated.
According to the study, other consistent characteristics of people who are vaccine hesitant are that they belong to a racial minority, are 65 or older, live in a household with children 18 or younger, or are unemployed.
When asked why they were vaccine hesitant, people gave these reasons: Lack of trust in COVID-19 vaccines (55%), concerns about side effects (48%), and lack of trust in government (46%).
Lack of access to vaccines, often cited in previous studies about resistance to other vaccines, was not cited as a reason for not getting the COVID-19 vaccine.
“COVID-19 vaccine hesitancy is a public health threat,” the researchers concluded. “Since education levels are not easily modifiable, our results suggest that policymakers would be best served by closing knowledge gaps to overcome negative perceptions of the vaccine through tailored interventions.”
A version of this article first appeared on WebMD.com.
Lack of a high school education is a predictor of whether a person will be resistant to getting the COVID-19 vaccine, a new study shows.
Researchers from the University of North Carolina looked at vaccination rates in 3,142 counties in the U.S. They compared them to population characteristics based on the CDC Social Vulnerability Index.
They found that more than half of the unvaccinated adults in the U.S. with strong vaccine hesitancy had a high school education or less. Vaccine hesitancy was defined as refusal to be vaccinated even if the COVID-19 vaccine was available.
The other main predictor for vaccine hesitancy was concern about vaccine availability and distribution, the researchers said.
“Our study suggests that low education levels are a major contributor to vaccine hesitancy and ultimately vaccination levels,” the authors wrote. The study was published in the American Journal of Infection Control. “Specifically, low vaccination levels were found in communities with a less educated population and with more concern about vaccine uptake capacity, suggesting that education is an ongoing challenge.”
“Our findings suggest that policy makers and community leaders should tailor vaccine information and efforts to those with limited education and specifically address knowledge concerns that are prevalent and likely more modifiable.”
The study was based on data gathered months ago. It says that as of May 9, 2021, 34.7% of the U.S. population was fully vaccinated and that 8% reported a strong unwillingness to get vaccinated.
At press time, the Centers for Disease Control and Prevention’s COVID Data Tracker showed that 62.5% of the U.S. population was fully vaccinated.
According to the study, other consistent characteristics of people who are vaccine hesitant are that they belong to a racial minority, are 65 or older, live in a household with children 18 or younger, or are unemployed.
When asked why they were vaccine hesitant, people gave these reasons: Lack of trust in COVID-19 vaccines (55%), concerns about side effects (48%), and lack of trust in government (46%).
Lack of access to vaccines, often cited in previous studies about resistance to other vaccines, was not cited as a reason for not getting the COVID-19 vaccine.
“COVID-19 vaccine hesitancy is a public health threat,” the researchers concluded. “Since education levels are not easily modifiable, our results suggest that policymakers would be best served by closing knowledge gaps to overcome negative perceptions of the vaccine through tailored interventions.”
A version of this article first appeared on WebMD.com.
FROM THE AMERICAN JOURNAL OF INFECTION CONTROL
COVID-19 linked to increased diabetes risk in youth
SARS-CoV-2 infection was associated with an increased risk for diabetes among youth, whereas other acute respiratory infections were not, new data from the U.S. Centers for Disease Control and Prevention indicate.
The results from two large U.S. health claims databases were published in an early release in the CDC’s Morbidity and Mortality Weekly Report by Catherine E. Barrett, PhD, and colleagues of the CDC’s COVID-19 Emergency Response Team and Division of Diabetes Translation.
Clinicians should monitor individuals younger than 18 years in the months following a SARS-CoV-2 infection for new diabetes onset, they advise.
The findings, which are supported by independent studies in adults, “underscore the importance of COVID-19 prevention among all age groups, including vaccination for all eligible children and adolescents, and chronic disease prevention and treatment,” Dr. Barrett and colleagues say.
Diabetes type couldn’t be reliably distinguished from the databases, which is noted as an important study limitation.
“SARS-CoV-2 infection might lead to type 1 or type 2 diabetes through complex and differing mechanisms,” they say.
Emerging evidence began to suggest, in mid-2020, that COVID-19 may trigger the onset of diabetes in healthy people. A new global registry was subsequently established to collect data on patients with COVID-19–related diabetes, called the CoviDiab registry.
Not clear if diabetes after COVID-19 is transient or permanent
From one of the databases used in the new study, known as IQVIA, 80,893 individuals aged younger than 18 years diagnosed with COVID-19 during March 2020 to February 26, 2021, were compared with age- and sex-matched people during that period who did not have COVID-19 and to prepandemic groups with and without a diagnosis of acute respiratory illness during March 1, 2017, to February 26, 2018.
From the second database, HealthVerity, 439,439 youth diagnosed with COVID-19 during March 1, 2020, to June 28, 2021, were compared with age- and sex-matched youth without COVID-19. Here, there was no prepandemic comparison group.
Diabetes diagnoses were coded in 0.08% with COVID-19 vs. 0.03% without COVID-19 in IQVIA and in 0.25% vs. 0.19% in HealthVerity.
Thus, new diabetes diagnoses were 166% and 31% more likely to occur in those with COVID-19 in IQVIA and HealthVerity, respectively. And in IQVIA, those with COVID-19 were 116% more likely to develop diabetes than were those with prepandemic acute respiratory illnesses. Those differences were all significant, whereas non–SARS-CoV-2 respiratory infections were not associated with diabetes, Dr. Barrett and colleagues say.
In both databases, diabetic ketoacidosis (DKA) was more common at diabetes onset among those with, vs. without, COVID-19: 48.5% vs. 13.6% in IQVIA and 40.2% vs. 29.7% in HealthVerity. In IQVIA, 22.0% with prepandemic acute respiratory illness presented with DKA.
Dr. Barrett and colleagues offer several potential explanations for the observed association between COVID-19 and diabetes, including a direct attack on pancreatic beta cells expressing angiotensin-converting enzyme 2 receptors, or via stress hyperglycemia resulting from cytokine storm and alterations in glucose metabolism.
Another possibility is the precipitation to diabetes from prediabetes; the latter is a condition present in one in five U.S. adolescents.
Steroid treatment during hospitalization might have led to transient hyperglycemia, but only 1.5% to 2.2% of diabetes codes were for drug- or chemical-induced diabetes. The majority were for type 1 or 2.
Alternatively, pandemic-associated weight gain might have also contributed to risks for both severe COVID-19 and type 2 diabetes.
“Although this study can provide information on the risk for diabetes following SARS-CoV-2 infection, additional data are needed to understand underlying pathogenic mechanisms, either those caused by SARS-CoV-2 infection itself or resulting from treatments, and whether a COVID-19–associated diabetes diagnosis is transient or leads to a chronic condition,” Dr. Barrett and colleagues conclude.
A version of this article first appeared on Medscape.com.
SARS-CoV-2 infection was associated with an increased risk for diabetes among youth, whereas other acute respiratory infections were not, new data from the U.S. Centers for Disease Control and Prevention indicate.
The results from two large U.S. health claims databases were published in an early release in the CDC’s Morbidity and Mortality Weekly Report by Catherine E. Barrett, PhD, and colleagues of the CDC’s COVID-19 Emergency Response Team and Division of Diabetes Translation.
Clinicians should monitor individuals younger than 18 years in the months following a SARS-CoV-2 infection for new diabetes onset, they advise.
The findings, which are supported by independent studies in adults, “underscore the importance of COVID-19 prevention among all age groups, including vaccination for all eligible children and adolescents, and chronic disease prevention and treatment,” Dr. Barrett and colleagues say.
Diabetes type couldn’t be reliably distinguished from the databases, which is noted as an important study limitation.
“SARS-CoV-2 infection might lead to type 1 or type 2 diabetes through complex and differing mechanisms,” they say.
Emerging evidence began to suggest, in mid-2020, that COVID-19 may trigger the onset of diabetes in healthy people. A new global registry was subsequently established to collect data on patients with COVID-19–related diabetes, called the CoviDiab registry.
Not clear if diabetes after COVID-19 is transient or permanent
From one of the databases used in the new study, known as IQVIA, 80,893 individuals aged younger than 18 years diagnosed with COVID-19 during March 2020 to February 26, 2021, were compared with age- and sex-matched people during that period who did not have COVID-19 and to prepandemic groups with and without a diagnosis of acute respiratory illness during March 1, 2017, to February 26, 2018.
From the second database, HealthVerity, 439,439 youth diagnosed with COVID-19 during March 1, 2020, to June 28, 2021, were compared with age- and sex-matched youth without COVID-19. Here, there was no prepandemic comparison group.
Diabetes diagnoses were coded in 0.08% with COVID-19 vs. 0.03% without COVID-19 in IQVIA and in 0.25% vs. 0.19% in HealthVerity.
Thus, new diabetes diagnoses were 166% and 31% more likely to occur in those with COVID-19 in IQVIA and HealthVerity, respectively. And in IQVIA, those with COVID-19 were 116% more likely to develop diabetes than were those with prepandemic acute respiratory illnesses. Those differences were all significant, whereas non–SARS-CoV-2 respiratory infections were not associated with diabetes, Dr. Barrett and colleagues say.
In both databases, diabetic ketoacidosis (DKA) was more common at diabetes onset among those with, vs. without, COVID-19: 48.5% vs. 13.6% in IQVIA and 40.2% vs. 29.7% in HealthVerity. In IQVIA, 22.0% with prepandemic acute respiratory illness presented with DKA.
Dr. Barrett and colleagues offer several potential explanations for the observed association between COVID-19 and diabetes, including a direct attack on pancreatic beta cells expressing angiotensin-converting enzyme 2 receptors, or via stress hyperglycemia resulting from cytokine storm and alterations in glucose metabolism.
Another possibility is the precipitation to diabetes from prediabetes; the latter is a condition present in one in five U.S. adolescents.
Steroid treatment during hospitalization might have led to transient hyperglycemia, but only 1.5% to 2.2% of diabetes codes were for drug- or chemical-induced diabetes. The majority were for type 1 or 2.
Alternatively, pandemic-associated weight gain might have also contributed to risks for both severe COVID-19 and type 2 diabetes.
“Although this study can provide information on the risk for diabetes following SARS-CoV-2 infection, additional data are needed to understand underlying pathogenic mechanisms, either those caused by SARS-CoV-2 infection itself or resulting from treatments, and whether a COVID-19–associated diabetes diagnosis is transient or leads to a chronic condition,” Dr. Barrett and colleagues conclude.
A version of this article first appeared on Medscape.com.
SARS-CoV-2 infection was associated with an increased risk for diabetes among youth, whereas other acute respiratory infections were not, new data from the U.S. Centers for Disease Control and Prevention indicate.
The results from two large U.S. health claims databases were published in an early release in the CDC’s Morbidity and Mortality Weekly Report by Catherine E. Barrett, PhD, and colleagues of the CDC’s COVID-19 Emergency Response Team and Division of Diabetes Translation.
Clinicians should monitor individuals younger than 18 years in the months following a SARS-CoV-2 infection for new diabetes onset, they advise.
The findings, which are supported by independent studies in adults, “underscore the importance of COVID-19 prevention among all age groups, including vaccination for all eligible children and adolescents, and chronic disease prevention and treatment,” Dr. Barrett and colleagues say.
Diabetes type couldn’t be reliably distinguished from the databases, which is noted as an important study limitation.
“SARS-CoV-2 infection might lead to type 1 or type 2 diabetes through complex and differing mechanisms,” they say.
Emerging evidence began to suggest, in mid-2020, that COVID-19 may trigger the onset of diabetes in healthy people. A new global registry was subsequently established to collect data on patients with COVID-19–related diabetes, called the CoviDiab registry.
Not clear if diabetes after COVID-19 is transient or permanent
From one of the databases used in the new study, known as IQVIA, 80,893 individuals aged younger than 18 years diagnosed with COVID-19 during March 2020 to February 26, 2021, were compared with age- and sex-matched people during that period who did not have COVID-19 and to prepandemic groups with and without a diagnosis of acute respiratory illness during March 1, 2017, to February 26, 2018.
From the second database, HealthVerity, 439,439 youth diagnosed with COVID-19 during March 1, 2020, to June 28, 2021, were compared with age- and sex-matched youth without COVID-19. Here, there was no prepandemic comparison group.
Diabetes diagnoses were coded in 0.08% with COVID-19 vs. 0.03% without COVID-19 in IQVIA and in 0.25% vs. 0.19% in HealthVerity.
Thus, new diabetes diagnoses were 166% and 31% more likely to occur in those with COVID-19 in IQVIA and HealthVerity, respectively. And in IQVIA, those with COVID-19 were 116% more likely to develop diabetes than were those with prepandemic acute respiratory illnesses. Those differences were all significant, whereas non–SARS-CoV-2 respiratory infections were not associated with diabetes, Dr. Barrett and colleagues say.
In both databases, diabetic ketoacidosis (DKA) was more common at diabetes onset among those with, vs. without, COVID-19: 48.5% vs. 13.6% in IQVIA and 40.2% vs. 29.7% in HealthVerity. In IQVIA, 22.0% with prepandemic acute respiratory illness presented with DKA.
Dr. Barrett and colleagues offer several potential explanations for the observed association between COVID-19 and diabetes, including a direct attack on pancreatic beta cells expressing angiotensin-converting enzyme 2 receptors, or via stress hyperglycemia resulting from cytokine storm and alterations in glucose metabolism.
Another possibility is the precipitation to diabetes from prediabetes; the latter is a condition present in one in five U.S. adolescents.
Steroid treatment during hospitalization might have led to transient hyperglycemia, but only 1.5% to 2.2% of diabetes codes were for drug- or chemical-induced diabetes. The majority were for type 1 or 2.
Alternatively, pandemic-associated weight gain might have also contributed to risks for both severe COVID-19 and type 2 diabetes.
“Although this study can provide information on the risk for diabetes following SARS-CoV-2 infection, additional data are needed to understand underlying pathogenic mechanisms, either those caused by SARS-CoV-2 infection itself or resulting from treatments, and whether a COVID-19–associated diabetes diagnosis is transient or leads to a chronic condition,” Dr. Barrett and colleagues conclude.
A version of this article first appeared on Medscape.com.
FROM MMWR
As pandemic regs expire, states get tougher on telehealth: report
Among the most important restrictions that have been reinstated in some states are those barring requirements for insurers to cover telehealth and regulations that prohibit telehealth visits across state lines, unless the physician is licensed in both states.
“Only three states – Arizona, Florida, and Indiana – allow all health care providers to easily practice telehealth across state lines,” says a news release on the think tanks’ report. “Forty-seven others have arbitrary barriers in place that limit patients’ access to specialists and available appointments based purely on residency.”
“Once the [state-based] public health emergency declarations started to end or executive orders were withdrawn, many of the new flexibilities for providers, insurers, and patients were lost overnight,” Vittorio Nastasi, a policy analyst at Reason Foundation and a co-author of the report, says in the news release. “States need to adopt a number of telehealth reforms to provide their residents better access to this safe and effective virtual care.”
On a positive note, the report says, most states have removed the requirement that a patient must first see a provider in person before they can use telehealth services. The exceptions are Tennessee, Alaska, and West Virginia, which require an in-person visit before certain telehealth services can be provided.
In addition, 20 states allow nurse practitioners to conduct telehealth visits without being under the supervision of a physician. Prior to the pandemic, some states allowed only doctors to use telehealth, the report says, but, during the COVID crisis, “the acute shortage of providers in many counties adds to the need for more kinds of providers to be able to use it.”
A number of states place restrictions on the telehealth modalities that can be utilized. Under the definition by the American Telemedicine Association, telehealth includes audio-video visits, remote patient monitoring, and “store and forward” telemedicine, which entails collecting clinical information and sending it to another site for evaluation. The latter method is particularly useful for consultations with specialists, the report notes.
Coverage mandates and payment parity
The report also examines other parameters of telehealth regulations in each state, including whether they have telehealth coverage mandates and whether they require physicians to be paid the same amount for similar types of in-person and telehealth visits.
The report views insurance mandates as beneficial, but not if they require coverage of all virtual services. While telehealth can be a game changer for post-stroke care and for other “treatment-intensive conditions,” the report says, the evidence of better outcomes for other conditions treated through telehealth is far less certain. Therefore, it advises states to “protect flexibility so that new innovative models can emerge.”
Ateev Mehrotra, MD, a professor at Harvard Medical School who studies telehealth, agrees that it offers more value in some clinical situations than in others. “High value is improving quality or outcomes at a reasonable cost,” he told this news organization. “If a telemedicine visit for stroke can save a person’s life and prevent disability, let’s pay for it. A telemedicine visit for a cold may not be necessary. Mom’s chicken soup is fine.”
A little over half of the states still require payment parity, according to the report. While these regulations are intended to promote the use of telehealth, the authors note, they can increase the growth of health care costs. Moreover, they argue, it’s hard to defend equal payments for virtual visits when the overhead required to deliver them – such as office rental, utility, and labor costs – is much lower than that for in-person visits. Also, it makes no sense for health systems to charge facility fees for telehealth visits when these visits can be initiated from anywhere, they say.
Dr. Mehrotra concurs with this view. “If you see someone in your office, your fee includes all the overhead for your office, and it’s a substantial cost,” he says. “For many procedures, it’s more than half of the cost. If you have a telemedicine visit and you’re at home, why would you pay the same amount? The visit may take the same amount of time, but all the money that goes for overhead is not accounted for.”
Telemedicine across state lines
The report’s contention about the difficulty of conducting telehealth encounters across most state lines seems to be at odds with the growth in the Interstate Medical Licensure Compact, which makes it easier for physicians in one compact member state to get licensed in others. Currently, 35 states belong to the compact, Joe Knickrehm, vice president of communications for the Federation of State Medical Boards, told this news organization.
In addition, he says, “12 state boards issue a special purpose license, telemedicine license or certificate, or license to practice medicine across state lines to allow for the practice of telemedicine.”
The catch, Dr. Mehrotra says, is that, despite the streamlining of license applications in compact member states, the fees charged by the state boards are still very high – a point that the report also makes. “If I want to have broad scope of practice, I’d have to pay thousands of dollars to many states. The license fees start to add up. Also, I have to keep track of each state’s CME requirements, which are all different. Keeping up with all of that is an administration burden, and it’s a pain.”
Mr. Knickrehm contends that obtaining multiple licenses via the compact “is generally less expensive for physicians than the cost of requesting transcripts, fingerprints, and other necessary paperwork each time they apply for licensure in a new state. Physicians are seeing the benefits of an expedited process that allows them to begin practicing more quickly [in other states].”
Dr. Mehrotra says he has seen the same retrenchment in state telehealth regulations that the report references. However, he says, “CMS [the Centers for Medicare & Medicaid Services] has signaled that at least through 2022 and maybe into 2023, they’ll continue their extensions of telemedicine [pandemic regulations].” After that, Congress would have to decide whether to make the changes permanent.
“Right now, it’s hard for me to see how a payer is going to pull back on telehealth, unless there’s ample evidence of overuse of telehealth,” he argues. “With the public and providers liking telehealth, it’s hard to say on theoretical grounds that we should stop using it. That’s why Medicare and others have extended it and why Congress will too.”
A version of this article first appeared on Medscape.com.
Among the most important restrictions that have been reinstated in some states are those barring requirements for insurers to cover telehealth and regulations that prohibit telehealth visits across state lines, unless the physician is licensed in both states.
“Only three states – Arizona, Florida, and Indiana – allow all health care providers to easily practice telehealth across state lines,” says a news release on the think tanks’ report. “Forty-seven others have arbitrary barriers in place that limit patients’ access to specialists and available appointments based purely on residency.”
“Once the [state-based] public health emergency declarations started to end or executive orders were withdrawn, many of the new flexibilities for providers, insurers, and patients were lost overnight,” Vittorio Nastasi, a policy analyst at Reason Foundation and a co-author of the report, says in the news release. “States need to adopt a number of telehealth reforms to provide their residents better access to this safe and effective virtual care.”
On a positive note, the report says, most states have removed the requirement that a patient must first see a provider in person before they can use telehealth services. The exceptions are Tennessee, Alaska, and West Virginia, which require an in-person visit before certain telehealth services can be provided.
In addition, 20 states allow nurse practitioners to conduct telehealth visits without being under the supervision of a physician. Prior to the pandemic, some states allowed only doctors to use telehealth, the report says, but, during the COVID crisis, “the acute shortage of providers in many counties adds to the need for more kinds of providers to be able to use it.”
A number of states place restrictions on the telehealth modalities that can be utilized. Under the definition by the American Telemedicine Association, telehealth includes audio-video visits, remote patient monitoring, and “store and forward” telemedicine, which entails collecting clinical information and sending it to another site for evaluation. The latter method is particularly useful for consultations with specialists, the report notes.
Coverage mandates and payment parity
The report also examines other parameters of telehealth regulations in each state, including whether they have telehealth coverage mandates and whether they require physicians to be paid the same amount for similar types of in-person and telehealth visits.
The report views insurance mandates as beneficial, but not if they require coverage of all virtual services. While telehealth can be a game changer for post-stroke care and for other “treatment-intensive conditions,” the report says, the evidence of better outcomes for other conditions treated through telehealth is far less certain. Therefore, it advises states to “protect flexibility so that new innovative models can emerge.”
Ateev Mehrotra, MD, a professor at Harvard Medical School who studies telehealth, agrees that it offers more value in some clinical situations than in others. “High value is improving quality or outcomes at a reasonable cost,” he told this news organization. “If a telemedicine visit for stroke can save a person’s life and prevent disability, let’s pay for it. A telemedicine visit for a cold may not be necessary. Mom’s chicken soup is fine.”
A little over half of the states still require payment parity, according to the report. While these regulations are intended to promote the use of telehealth, the authors note, they can increase the growth of health care costs. Moreover, they argue, it’s hard to defend equal payments for virtual visits when the overhead required to deliver them – such as office rental, utility, and labor costs – is much lower than that for in-person visits. Also, it makes no sense for health systems to charge facility fees for telehealth visits when these visits can be initiated from anywhere, they say.
Dr. Mehrotra concurs with this view. “If you see someone in your office, your fee includes all the overhead for your office, and it’s a substantial cost,” he says. “For many procedures, it’s more than half of the cost. If you have a telemedicine visit and you’re at home, why would you pay the same amount? The visit may take the same amount of time, but all the money that goes for overhead is not accounted for.”
Telemedicine across state lines
The report’s contention about the difficulty of conducting telehealth encounters across most state lines seems to be at odds with the growth in the Interstate Medical Licensure Compact, which makes it easier for physicians in one compact member state to get licensed in others. Currently, 35 states belong to the compact, Joe Knickrehm, vice president of communications for the Federation of State Medical Boards, told this news organization.
In addition, he says, “12 state boards issue a special purpose license, telemedicine license or certificate, or license to practice medicine across state lines to allow for the practice of telemedicine.”
The catch, Dr. Mehrotra says, is that, despite the streamlining of license applications in compact member states, the fees charged by the state boards are still very high – a point that the report also makes. “If I want to have broad scope of practice, I’d have to pay thousands of dollars to many states. The license fees start to add up. Also, I have to keep track of each state’s CME requirements, which are all different. Keeping up with all of that is an administration burden, and it’s a pain.”
Mr. Knickrehm contends that obtaining multiple licenses via the compact “is generally less expensive for physicians than the cost of requesting transcripts, fingerprints, and other necessary paperwork each time they apply for licensure in a new state. Physicians are seeing the benefits of an expedited process that allows them to begin practicing more quickly [in other states].”
Dr. Mehrotra says he has seen the same retrenchment in state telehealth regulations that the report references. However, he says, “CMS [the Centers for Medicare & Medicaid Services] has signaled that at least through 2022 and maybe into 2023, they’ll continue their extensions of telemedicine [pandemic regulations].” After that, Congress would have to decide whether to make the changes permanent.
“Right now, it’s hard for me to see how a payer is going to pull back on telehealth, unless there’s ample evidence of overuse of telehealth,” he argues. “With the public and providers liking telehealth, it’s hard to say on theoretical grounds that we should stop using it. That’s why Medicare and others have extended it and why Congress will too.”
A version of this article first appeared on Medscape.com.
Among the most important restrictions that have been reinstated in some states are those barring requirements for insurers to cover telehealth and regulations that prohibit telehealth visits across state lines, unless the physician is licensed in both states.
“Only three states – Arizona, Florida, and Indiana – allow all health care providers to easily practice telehealth across state lines,” says a news release on the think tanks’ report. “Forty-seven others have arbitrary barriers in place that limit patients’ access to specialists and available appointments based purely on residency.”
“Once the [state-based] public health emergency declarations started to end or executive orders were withdrawn, many of the new flexibilities for providers, insurers, and patients were lost overnight,” Vittorio Nastasi, a policy analyst at Reason Foundation and a co-author of the report, says in the news release. “States need to adopt a number of telehealth reforms to provide their residents better access to this safe and effective virtual care.”
On a positive note, the report says, most states have removed the requirement that a patient must first see a provider in person before they can use telehealth services. The exceptions are Tennessee, Alaska, and West Virginia, which require an in-person visit before certain telehealth services can be provided.
In addition, 20 states allow nurse practitioners to conduct telehealth visits without being under the supervision of a physician. Prior to the pandemic, some states allowed only doctors to use telehealth, the report says, but, during the COVID crisis, “the acute shortage of providers in many counties adds to the need for more kinds of providers to be able to use it.”
A number of states place restrictions on the telehealth modalities that can be utilized. Under the definition by the American Telemedicine Association, telehealth includes audio-video visits, remote patient monitoring, and “store and forward” telemedicine, which entails collecting clinical information and sending it to another site for evaluation. The latter method is particularly useful for consultations with specialists, the report notes.
Coverage mandates and payment parity
The report also examines other parameters of telehealth regulations in each state, including whether they have telehealth coverage mandates and whether they require physicians to be paid the same amount for similar types of in-person and telehealth visits.
The report views insurance mandates as beneficial, but not if they require coverage of all virtual services. While telehealth can be a game changer for post-stroke care and for other “treatment-intensive conditions,” the report says, the evidence of better outcomes for other conditions treated through telehealth is far less certain. Therefore, it advises states to “protect flexibility so that new innovative models can emerge.”
Ateev Mehrotra, MD, a professor at Harvard Medical School who studies telehealth, agrees that it offers more value in some clinical situations than in others. “High value is improving quality or outcomes at a reasonable cost,” he told this news organization. “If a telemedicine visit for stroke can save a person’s life and prevent disability, let’s pay for it. A telemedicine visit for a cold may not be necessary. Mom’s chicken soup is fine.”
A little over half of the states still require payment parity, according to the report. While these regulations are intended to promote the use of telehealth, the authors note, they can increase the growth of health care costs. Moreover, they argue, it’s hard to defend equal payments for virtual visits when the overhead required to deliver them – such as office rental, utility, and labor costs – is much lower than that for in-person visits. Also, it makes no sense for health systems to charge facility fees for telehealth visits when these visits can be initiated from anywhere, they say.
Dr. Mehrotra concurs with this view. “If you see someone in your office, your fee includes all the overhead for your office, and it’s a substantial cost,” he says. “For many procedures, it’s more than half of the cost. If you have a telemedicine visit and you’re at home, why would you pay the same amount? The visit may take the same amount of time, but all the money that goes for overhead is not accounted for.”
Telemedicine across state lines
The report’s contention about the difficulty of conducting telehealth encounters across most state lines seems to be at odds with the growth in the Interstate Medical Licensure Compact, which makes it easier for physicians in one compact member state to get licensed in others. Currently, 35 states belong to the compact, Joe Knickrehm, vice president of communications for the Federation of State Medical Boards, told this news organization.
In addition, he says, “12 state boards issue a special purpose license, telemedicine license or certificate, or license to practice medicine across state lines to allow for the practice of telemedicine.”
The catch, Dr. Mehrotra says, is that, despite the streamlining of license applications in compact member states, the fees charged by the state boards are still very high – a point that the report also makes. “If I want to have broad scope of practice, I’d have to pay thousands of dollars to many states. The license fees start to add up. Also, I have to keep track of each state’s CME requirements, which are all different. Keeping up with all of that is an administration burden, and it’s a pain.”
Mr. Knickrehm contends that obtaining multiple licenses via the compact “is generally less expensive for physicians than the cost of requesting transcripts, fingerprints, and other necessary paperwork each time they apply for licensure in a new state. Physicians are seeing the benefits of an expedited process that allows them to begin practicing more quickly [in other states].”
Dr. Mehrotra says he has seen the same retrenchment in state telehealth regulations that the report references. However, he says, “CMS [the Centers for Medicare & Medicaid Services] has signaled that at least through 2022 and maybe into 2023, they’ll continue their extensions of telemedicine [pandemic regulations].” After that, Congress would have to decide whether to make the changes permanent.
“Right now, it’s hard for me to see how a payer is going to pull back on telehealth, unless there’s ample evidence of overuse of telehealth,” he argues. “With the public and providers liking telehealth, it’s hard to say on theoretical grounds that we should stop using it. That’s why Medicare and others have extended it and why Congress will too.”
A version of this article first appeared on Medscape.com.