User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
nav[contains(@class, 'nav-ce-stack nav-ce-stack__large-screen')]
header[@id='header']
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]
Older adults with type 2 diabetes find weight loss, deprescribing benefits in GLP-1 agonists, small study suggests
Glucagon-like peptide 1 (GLP-1) agonists may help clinicians manage uncontrolled type 2 diabetes in some older patients without the need for additional glucose-controlling medications, according to a study presented Nov. 8 at the annual meeting of the Gerontological Society of America.
The study analyzed charts of 30 adults aged 65-84 years who were seen in clinic from January 2022 to February 2023 and were started on GLP-1 or GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonists. Participants had uncontrolled type 2 diabetes with initial A1c levels ranging from 9.6% to 12.6% and a body mass index between 27 and 48.2. The patients also received education about their conditions as well as counseling on diet and lifestyle modifications.
All participants experienced a reduction in A1c to a range of 5.8% to 7.7%, and a moderate reduction in BMI to between 23 and 39.8 within the year.
“The reduction in BMI that we saw in our patients even though they were still in the category of obesity produces a substantial benefit in the management [of type 2 diabetes],” because weight loss helps to control the condition, said Anna Pendrey, MD, assistant professor of clinical family medicine and geriatrics at Indiana University, Indianapolis, and sole author of the study.
In some cases, the addition of a GLP-1 agonist or GLP-1/GIP agonist allowed for clinicians to deprescribe other medications such as insulin and sulfonylureas, which can cause hypoglycemia in older adults, Dr. Pendrey said.
Approximately 11% of U.S. adults have type 2 diabetes, a percentage that is likely to grow given the prevalence of childhood obesity, according to the Centers for Disease Control and Prevention. Dr. Pendrey highlighted the increased incidence of newly diagnosed diabetes in individuals aged 65-79 years.
Previous studies have shown that GLP-1 agonists have the potential to aid in weight reduction, glucose control, and the prevention of major adverse cardiovascular events in these patients.
The new study is one of many post hoc analyses that mark another step forward in addressing the complex challenges associated with diabetes in older adults, according to Rodolfo Galindo, MD, director of the Comprehensive Diabetes Center at the University of Miami Health System in Florida.
“I believe this is important because unfortunately many of our older adults have both diabetes and obesity,” Dr. Galindo, who was not involved with the research, told this news organization. “You can induce remission of type 2 diabetes through weight loss that GLP-1s can cause.”
The treatment paradigm has shifted away from focusing only on lowering glucose levels as the primary means to prevent complications from diabetes, Dr. Galindo said.
Indeed, weight loss can modify diseases and prevent other complications associated with type 2 diabetes, Dr. Pendrey said.
“Weight loss and diabetes mellitus control also produces cardiovascular protection that is significant for patients with diabetes, so this group of patients in my opinion are the ones that benefit the most from GLP-1s,” she said.
Side effects of GLP-1 agonists can include nausea and vomiting, which could lead to dehydration. GLP-1s can also increase the risk for pancreatitis. For older adults, weight loss from the drug could cause sarcopenia, or loss of muscle mass, Dr. Galindo said.
“This is the reason why patients in treatment with GLP-1s have to be in close contact with their providers,” Dr. Pendrey said.
This study was independently supported. Dr. Pendrey and Dr. Galindo report no relevant conflicts.
A version of this article appeared on Medscape.com.
Glucagon-like peptide 1 (GLP-1) agonists may help clinicians manage uncontrolled type 2 diabetes in some older patients without the need for additional glucose-controlling medications, according to a study presented Nov. 8 at the annual meeting of the Gerontological Society of America.
The study analyzed charts of 30 adults aged 65-84 years who were seen in clinic from January 2022 to February 2023 and were started on GLP-1 or GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonists. Participants had uncontrolled type 2 diabetes with initial A1c levels ranging from 9.6% to 12.6% and a body mass index between 27 and 48.2. The patients also received education about their conditions as well as counseling on diet and lifestyle modifications.
All participants experienced a reduction in A1c to a range of 5.8% to 7.7%, and a moderate reduction in BMI to between 23 and 39.8 within the year.
“The reduction in BMI that we saw in our patients even though they were still in the category of obesity produces a substantial benefit in the management [of type 2 diabetes],” because weight loss helps to control the condition, said Anna Pendrey, MD, assistant professor of clinical family medicine and geriatrics at Indiana University, Indianapolis, and sole author of the study.
In some cases, the addition of a GLP-1 agonist or GLP-1/GIP agonist allowed for clinicians to deprescribe other medications such as insulin and sulfonylureas, which can cause hypoglycemia in older adults, Dr. Pendrey said.
Approximately 11% of U.S. adults have type 2 diabetes, a percentage that is likely to grow given the prevalence of childhood obesity, according to the Centers for Disease Control and Prevention. Dr. Pendrey highlighted the increased incidence of newly diagnosed diabetes in individuals aged 65-79 years.
Previous studies have shown that GLP-1 agonists have the potential to aid in weight reduction, glucose control, and the prevention of major adverse cardiovascular events in these patients.
The new study is one of many post hoc analyses that mark another step forward in addressing the complex challenges associated with diabetes in older adults, according to Rodolfo Galindo, MD, director of the Comprehensive Diabetes Center at the University of Miami Health System in Florida.
“I believe this is important because unfortunately many of our older adults have both diabetes and obesity,” Dr. Galindo, who was not involved with the research, told this news organization. “You can induce remission of type 2 diabetes through weight loss that GLP-1s can cause.”
The treatment paradigm has shifted away from focusing only on lowering glucose levels as the primary means to prevent complications from diabetes, Dr. Galindo said.
Indeed, weight loss can modify diseases and prevent other complications associated with type 2 diabetes, Dr. Pendrey said.
“Weight loss and diabetes mellitus control also produces cardiovascular protection that is significant for patients with diabetes, so this group of patients in my opinion are the ones that benefit the most from GLP-1s,” she said.
Side effects of GLP-1 agonists can include nausea and vomiting, which could lead to dehydration. GLP-1s can also increase the risk for pancreatitis. For older adults, weight loss from the drug could cause sarcopenia, or loss of muscle mass, Dr. Galindo said.
“This is the reason why patients in treatment with GLP-1s have to be in close contact with their providers,” Dr. Pendrey said.
This study was independently supported. Dr. Pendrey and Dr. Galindo report no relevant conflicts.
A version of this article appeared on Medscape.com.
Glucagon-like peptide 1 (GLP-1) agonists may help clinicians manage uncontrolled type 2 diabetes in some older patients without the need for additional glucose-controlling medications, according to a study presented Nov. 8 at the annual meeting of the Gerontological Society of America.
The study analyzed charts of 30 adults aged 65-84 years who were seen in clinic from January 2022 to February 2023 and were started on GLP-1 or GLP-1 and glucose-dependent insulinotropic polypeptide (GIP) agonists. Participants had uncontrolled type 2 diabetes with initial A1c levels ranging from 9.6% to 12.6% and a body mass index between 27 and 48.2. The patients also received education about their conditions as well as counseling on diet and lifestyle modifications.
All participants experienced a reduction in A1c to a range of 5.8% to 7.7%, and a moderate reduction in BMI to between 23 and 39.8 within the year.
“The reduction in BMI that we saw in our patients even though they were still in the category of obesity produces a substantial benefit in the management [of type 2 diabetes],” because weight loss helps to control the condition, said Anna Pendrey, MD, assistant professor of clinical family medicine and geriatrics at Indiana University, Indianapolis, and sole author of the study.
In some cases, the addition of a GLP-1 agonist or GLP-1/GIP agonist allowed for clinicians to deprescribe other medications such as insulin and sulfonylureas, which can cause hypoglycemia in older adults, Dr. Pendrey said.
Approximately 11% of U.S. adults have type 2 diabetes, a percentage that is likely to grow given the prevalence of childhood obesity, according to the Centers for Disease Control and Prevention. Dr. Pendrey highlighted the increased incidence of newly diagnosed diabetes in individuals aged 65-79 years.
Previous studies have shown that GLP-1 agonists have the potential to aid in weight reduction, glucose control, and the prevention of major adverse cardiovascular events in these patients.
The new study is one of many post hoc analyses that mark another step forward in addressing the complex challenges associated with diabetes in older adults, according to Rodolfo Galindo, MD, director of the Comprehensive Diabetes Center at the University of Miami Health System in Florida.
“I believe this is important because unfortunately many of our older adults have both diabetes and obesity,” Dr. Galindo, who was not involved with the research, told this news organization. “You can induce remission of type 2 diabetes through weight loss that GLP-1s can cause.”
The treatment paradigm has shifted away from focusing only on lowering glucose levels as the primary means to prevent complications from diabetes, Dr. Galindo said.
Indeed, weight loss can modify diseases and prevent other complications associated with type 2 diabetes, Dr. Pendrey said.
“Weight loss and diabetes mellitus control also produces cardiovascular protection that is significant for patients with diabetes, so this group of patients in my opinion are the ones that benefit the most from GLP-1s,” she said.
Side effects of GLP-1 agonists can include nausea and vomiting, which could lead to dehydration. GLP-1s can also increase the risk for pancreatitis. For older adults, weight loss from the drug could cause sarcopenia, or loss of muscle mass, Dr. Galindo said.
“This is the reason why patients in treatment with GLP-1s have to be in close contact with their providers,” Dr. Pendrey said.
This study was independently supported. Dr. Pendrey and Dr. Galindo report no relevant conflicts.
A version of this article appeared on Medscape.com.
Chronic diarrhea management: Be wary of false diarrhea
PARIS – said gastroenterologist Nassim Hammoudi, MD, PhD, of the Lariboisière Hospital in Paris, during France’s annual general medicine conference (JNMG 2023). He said that doctors need to understand the characteristics of chronic diarrhea and adapt its management accordingly. In his presentation, Dr. Hammoudi highlighted the clinical signs that should be considered.
Mechanisms of chronic diarrhea
Chronic diarrhea can result from different mechanisms, such as motility disorders related to accelerated intestinal transit, malabsorption, osmotic diarrhea, and secretory diarrhea, which are often interlinked. When an endoscopy is performed, it is recommended to conduct multilevel biopsies to detect microscopic colitis, which Dr. Hammoudi believes is “probably underdiagnosed.”
Diarrhea is defined as the passage of frequent stools (more than three a day), soft to liquid in consistency, and a daily weight exceeding 300 g. It is considered chronic when it persists for more than a month.
Identifying false diarrhea
Practitioners must first distinguish between genuine and false diarrhea, with the latter presenting in most consultations. “Thorough questioning is fundamental,” Dr. Hammoudi emphasized. It is essential to determine the daily stool count, the presence of nocturnal stools, and stool consistency. “A soft stool passed once a day is not diarrhea,” he said.
The most challenging form of false diarrhea to identify is what he called “constipated person’s diarrhea.” These patients, who are typically elderly, reside in care homes, and are bed-bound and taking morphine, have daily liquid stools but are actually constipated. “Taking antidiarrheal medications makes the situation worse,” said Dr. Hammoudi.
Another type of false diarrhea is tenesmus, in which patients feel like they have a full rectum, even though it is physiologically empty. The recurring urge to defecate results in mucus discharges that resemble diarrhea. Inflammatory rectal involvement could be the cause, necessitating a gastroenterology consultation.
Anal incontinence can also cause false diarrhea. It is more common in elderly people residing in care homes and in women in the postpartum period. This condition is difficult to manage and requires referral to a gastroenterologist.
Chronic diarrhea: Could cancer be the culprit?
After ruling out false diarrhea, clinicians should be vigilant for warning signs. The first question to consider, said Dr. Hammoudi, is whether the chronic diarrhea is associated with a lesion. Several criteria should prompt a colonoscopy, especially to search for colorectal cancer lesions:
- Age greater than 50 years
- Personal or family history of colorectal cancer
- Recent changes in bowel habits
- Rectal bleeding
- Nighttime stools
- Unexplained weight loss
- Iron-deficiency anemia
Obvious causes of chronic diarrhea should be prioritized in the management plan. Medications top the list, with more than 500 treatments – for example, ACE inhibitors, proton pump inhibitors (PPIs), antidiabetic drugs, colchicine, magnesium, laxatives – known to have diarrhea as a side effect.
Certain dietary habits can also exacerbate diarrhea, such as milk consumption in cases of lactose intolerance, or excessive sugar intake, which can lead to osmotic diarrhea.
IBS is often at play
Once these causes have been ruled out, several etiological pathways should be investigated. The first relates to motility issues, which are the most common diarrhea-related problem, said Dr. Hammoudi.
This type of diarrhea is linked to rapid intestinal transit time and is characterized by postprandial bowel movements (occurring shortly after a meal). Here, patients experience urgency and notice identifiable food debris in their stools. It tends to stop when fasting and can be treated effectively with antidiarrheals.
Irritable bowel syndrome (IBS) is the main cause of rapid intestinal transit diarrhea. It is defined as recurrent abdominal pain (at least 1 day/week) over a period of 3 months, associated with two of the following criteria: pain eases or worsens on passing feces, change in frequency of bowel movements, change in the consistency of stools.
Symptoms may come on suddenly, sometimes after taking antibiotics, and may result in misdiagnosis.
IBS medications treat the symptoms. Antispasmodics, such as trimebutine, phloroglucinol (Spasfon), or pinaverium bromide (Dicetel) are recommended, even there can be a placebo effect. The antidiarrheal medication loperamide (Imodium) can also be used. Probiotics may be beneficial, as an imbalanced intestinal microbiota is often implicated.
Dietary changes can also have a positive impact. Encouraging a diet rich in fruit and vegetables to enhance fiber intake is advised. A low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols) diet, targeting short-chain carbohydrates, can also be tried to identify foods to avoid, although it may be challenging to stick to.
Postinfectious IBS is a frequent cause of rapid intestinal transit diarrhea. It generally follows an episode of acute infectious diarrhea. “Symptoms may come on suddenly, sometimes after taking antibiotics, and may result in misdiagnosis,” said Dr. Hammoudi. This type of IBS often resolves spontaneously within 6 months.
Consider the possibility of SIBO
Another cause of rapid intestinal transit diarrhea is small intestinal bacterial overgrowth (SIBO). It is difficult to distinguish between IBS and SIBO. Often, affected patients are diabetic, overweight, or have had bowel surgery.
The only way to diagnose SIBO is by conducting a breath test to measure the production of hydrogen and methane by the microbiota after ingesting sugar. However, the test is difficult to access and not fully covered by social security plans in France, said Dr. Hammoudi.
In cases of suspected SIBO and severe symptoms, a 7- to 10-day course of antibiotics can be attempted to provide relief, although a diagnosis should be confirmed before considering this option, Dr. Hammoudi said.
Malabsorption diarrhea
Another major cause of chronic diarrhea is malabsorption, characterized by large, fatty stools that are difficult to flush. Despite a normal diet, this type of diarrhea is associated with weight loss and nutritional deficiencies.
Its diagnosis involves measuring fat in the stools (steatorrhea) and possibly testing fecal elastase, an enzyme produced by the pancreas that is involved in digestion.
The most important causes of malabsorption diarrhea are pancreatic insufficiency, celiac disease, and Crohn’s disease. Generally, any lesion in the small intestine can lead to malabsorption-related diarrhea.
Celiac disease, or gluten intolerance, is an autoimmune condition triggered by a reaction to gluten proteins. Several antibodies can be produced in the presence of gluten proteins. Diagnosis is confirmed by positive antitransglutaminase antibodies and a duodenal biopsy through esophagogastroduodenoscopy.
The only treatment for celiac disease is a lifelong gluten-free diet. Celiac disease is increasingly diagnosed in adults, said Dr. Hammoudi, and should be considered as a possibility. This condition must be distinguished from gluten sensitivity, which can cause digestive issues, possibly leading to rapid intestinal transit diarrhea. “The only treatment for celiac disease is a lifelong gluten-free diet,” Dr. Hammoudi added.
Crohn’s disease, a type of inflammatory bowel disease, affects the entire digestive tract, particularly the terminal small intestine, which promotes malabsorption. In ulcerative colitis, another IBD affecting the rectum, any associated rectal syndrome can result in false diarrhea with stools containing blood and mucus.
Osmotic diarrhea, on the other hand, is linked to the presence of highly osmotic agents in the digestive tract. This type of diarrhea is watery and short-lived, stopping once the agents are no longer absorbed. The main culprits are lactose (in cases of lactose intolerance) and laxatives.
Drug-induced microscopic colitis
Secretory diarrhea is characterized by excessive secretions by the digestive tract, leading to significant potassium loss. This type of diarrhea is not related to food intake and is resistant to fasting.
Major causes of secretory diarrhea include microscopic colitis, parasitic infections, and endocrine tumors. Between 10% and 15% of patients with chronic diarrhea and apparently normal colonoscopy have microscopic colitis.
Dr. Hammoudi advised specialists seeking to determine the cause of chronic diarrhea to routinely collect multilevel bowel biopsies during colonoscopies from macroscopically normal mucosa to rule out microscopic colitis.
Microscopic colitis is mainly linked to the use of medications like PPIs and NSAIDs. These drugs can induce malabsorption-related diarrhea by damaging the intestinal wall.
In addition to discontinuing the implicated medication, the treatment for microscopic colitis includes low-dose budesonide (multiple brands). Biologics used in IBD may also be considered in cases of recurrent colitis.
Finally, exudative enteropathy can be a distinct cause of chronic diarrhea. It is characterized by albumin leakage (Waldmann’s disease) and manifests with edema, malnutrition, and significant hypoalbuminemia.
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
PARIS – said gastroenterologist Nassim Hammoudi, MD, PhD, of the Lariboisière Hospital in Paris, during France’s annual general medicine conference (JNMG 2023). He said that doctors need to understand the characteristics of chronic diarrhea and adapt its management accordingly. In his presentation, Dr. Hammoudi highlighted the clinical signs that should be considered.
Mechanisms of chronic diarrhea
Chronic diarrhea can result from different mechanisms, such as motility disorders related to accelerated intestinal transit, malabsorption, osmotic diarrhea, and secretory diarrhea, which are often interlinked. When an endoscopy is performed, it is recommended to conduct multilevel biopsies to detect microscopic colitis, which Dr. Hammoudi believes is “probably underdiagnosed.”
Diarrhea is defined as the passage of frequent stools (more than three a day), soft to liquid in consistency, and a daily weight exceeding 300 g. It is considered chronic when it persists for more than a month.
Identifying false diarrhea
Practitioners must first distinguish between genuine and false diarrhea, with the latter presenting in most consultations. “Thorough questioning is fundamental,” Dr. Hammoudi emphasized. It is essential to determine the daily stool count, the presence of nocturnal stools, and stool consistency. “A soft stool passed once a day is not diarrhea,” he said.
The most challenging form of false diarrhea to identify is what he called “constipated person’s diarrhea.” These patients, who are typically elderly, reside in care homes, and are bed-bound and taking morphine, have daily liquid stools but are actually constipated. “Taking antidiarrheal medications makes the situation worse,” said Dr. Hammoudi.
Another type of false diarrhea is tenesmus, in which patients feel like they have a full rectum, even though it is physiologically empty. The recurring urge to defecate results in mucus discharges that resemble diarrhea. Inflammatory rectal involvement could be the cause, necessitating a gastroenterology consultation.
Anal incontinence can also cause false diarrhea. It is more common in elderly people residing in care homes and in women in the postpartum period. This condition is difficult to manage and requires referral to a gastroenterologist.
Chronic diarrhea: Could cancer be the culprit?
After ruling out false diarrhea, clinicians should be vigilant for warning signs. The first question to consider, said Dr. Hammoudi, is whether the chronic diarrhea is associated with a lesion. Several criteria should prompt a colonoscopy, especially to search for colorectal cancer lesions:
- Age greater than 50 years
- Personal or family history of colorectal cancer
- Recent changes in bowel habits
- Rectal bleeding
- Nighttime stools
- Unexplained weight loss
- Iron-deficiency anemia
Obvious causes of chronic diarrhea should be prioritized in the management plan. Medications top the list, with more than 500 treatments – for example, ACE inhibitors, proton pump inhibitors (PPIs), antidiabetic drugs, colchicine, magnesium, laxatives – known to have diarrhea as a side effect.
Certain dietary habits can also exacerbate diarrhea, such as milk consumption in cases of lactose intolerance, or excessive sugar intake, which can lead to osmotic diarrhea.
IBS is often at play
Once these causes have been ruled out, several etiological pathways should be investigated. The first relates to motility issues, which are the most common diarrhea-related problem, said Dr. Hammoudi.
This type of diarrhea is linked to rapid intestinal transit time and is characterized by postprandial bowel movements (occurring shortly after a meal). Here, patients experience urgency and notice identifiable food debris in their stools. It tends to stop when fasting and can be treated effectively with antidiarrheals.
Irritable bowel syndrome (IBS) is the main cause of rapid intestinal transit diarrhea. It is defined as recurrent abdominal pain (at least 1 day/week) over a period of 3 months, associated with two of the following criteria: pain eases or worsens on passing feces, change in frequency of bowel movements, change in the consistency of stools.
Symptoms may come on suddenly, sometimes after taking antibiotics, and may result in misdiagnosis.
IBS medications treat the symptoms. Antispasmodics, such as trimebutine, phloroglucinol (Spasfon), or pinaverium bromide (Dicetel) are recommended, even there can be a placebo effect. The antidiarrheal medication loperamide (Imodium) can also be used. Probiotics may be beneficial, as an imbalanced intestinal microbiota is often implicated.
Dietary changes can also have a positive impact. Encouraging a diet rich in fruit and vegetables to enhance fiber intake is advised. A low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols) diet, targeting short-chain carbohydrates, can also be tried to identify foods to avoid, although it may be challenging to stick to.
Postinfectious IBS is a frequent cause of rapid intestinal transit diarrhea. It generally follows an episode of acute infectious diarrhea. “Symptoms may come on suddenly, sometimes after taking antibiotics, and may result in misdiagnosis,” said Dr. Hammoudi. This type of IBS often resolves spontaneously within 6 months.
Consider the possibility of SIBO
Another cause of rapid intestinal transit diarrhea is small intestinal bacterial overgrowth (SIBO). It is difficult to distinguish between IBS and SIBO. Often, affected patients are diabetic, overweight, or have had bowel surgery.
The only way to diagnose SIBO is by conducting a breath test to measure the production of hydrogen and methane by the microbiota after ingesting sugar. However, the test is difficult to access and not fully covered by social security plans in France, said Dr. Hammoudi.
In cases of suspected SIBO and severe symptoms, a 7- to 10-day course of antibiotics can be attempted to provide relief, although a diagnosis should be confirmed before considering this option, Dr. Hammoudi said.
Malabsorption diarrhea
Another major cause of chronic diarrhea is malabsorption, characterized by large, fatty stools that are difficult to flush. Despite a normal diet, this type of diarrhea is associated with weight loss and nutritional deficiencies.
Its diagnosis involves measuring fat in the stools (steatorrhea) and possibly testing fecal elastase, an enzyme produced by the pancreas that is involved in digestion.
The most important causes of malabsorption diarrhea are pancreatic insufficiency, celiac disease, and Crohn’s disease. Generally, any lesion in the small intestine can lead to malabsorption-related diarrhea.
Celiac disease, or gluten intolerance, is an autoimmune condition triggered by a reaction to gluten proteins. Several antibodies can be produced in the presence of gluten proteins. Diagnosis is confirmed by positive antitransglutaminase antibodies and a duodenal biopsy through esophagogastroduodenoscopy.
The only treatment for celiac disease is a lifelong gluten-free diet. Celiac disease is increasingly diagnosed in adults, said Dr. Hammoudi, and should be considered as a possibility. This condition must be distinguished from gluten sensitivity, which can cause digestive issues, possibly leading to rapid intestinal transit diarrhea. “The only treatment for celiac disease is a lifelong gluten-free diet,” Dr. Hammoudi added.
Crohn’s disease, a type of inflammatory bowel disease, affects the entire digestive tract, particularly the terminal small intestine, which promotes malabsorption. In ulcerative colitis, another IBD affecting the rectum, any associated rectal syndrome can result in false diarrhea with stools containing blood and mucus.
Osmotic diarrhea, on the other hand, is linked to the presence of highly osmotic agents in the digestive tract. This type of diarrhea is watery and short-lived, stopping once the agents are no longer absorbed. The main culprits are lactose (in cases of lactose intolerance) and laxatives.
Drug-induced microscopic colitis
Secretory diarrhea is characterized by excessive secretions by the digestive tract, leading to significant potassium loss. This type of diarrhea is not related to food intake and is resistant to fasting.
Major causes of secretory diarrhea include microscopic colitis, parasitic infections, and endocrine tumors. Between 10% and 15% of patients with chronic diarrhea and apparently normal colonoscopy have microscopic colitis.
Dr. Hammoudi advised specialists seeking to determine the cause of chronic diarrhea to routinely collect multilevel bowel biopsies during colonoscopies from macroscopically normal mucosa to rule out microscopic colitis.
Microscopic colitis is mainly linked to the use of medications like PPIs and NSAIDs. These drugs can induce malabsorption-related diarrhea by damaging the intestinal wall.
In addition to discontinuing the implicated medication, the treatment for microscopic colitis includes low-dose budesonide (multiple brands). Biologics used in IBD may also be considered in cases of recurrent colitis.
Finally, exudative enteropathy can be a distinct cause of chronic diarrhea. It is characterized by albumin leakage (Waldmann’s disease) and manifests with edema, malnutrition, and significant hypoalbuminemia.
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
PARIS – said gastroenterologist Nassim Hammoudi, MD, PhD, of the Lariboisière Hospital in Paris, during France’s annual general medicine conference (JNMG 2023). He said that doctors need to understand the characteristics of chronic diarrhea and adapt its management accordingly. In his presentation, Dr. Hammoudi highlighted the clinical signs that should be considered.
Mechanisms of chronic diarrhea
Chronic diarrhea can result from different mechanisms, such as motility disorders related to accelerated intestinal transit, malabsorption, osmotic diarrhea, and secretory diarrhea, which are often interlinked. When an endoscopy is performed, it is recommended to conduct multilevel biopsies to detect microscopic colitis, which Dr. Hammoudi believes is “probably underdiagnosed.”
Diarrhea is defined as the passage of frequent stools (more than three a day), soft to liquid in consistency, and a daily weight exceeding 300 g. It is considered chronic when it persists for more than a month.
Identifying false diarrhea
Practitioners must first distinguish between genuine and false diarrhea, with the latter presenting in most consultations. “Thorough questioning is fundamental,” Dr. Hammoudi emphasized. It is essential to determine the daily stool count, the presence of nocturnal stools, and stool consistency. “A soft stool passed once a day is not diarrhea,” he said.
The most challenging form of false diarrhea to identify is what he called “constipated person’s diarrhea.” These patients, who are typically elderly, reside in care homes, and are bed-bound and taking morphine, have daily liquid stools but are actually constipated. “Taking antidiarrheal medications makes the situation worse,” said Dr. Hammoudi.
Another type of false diarrhea is tenesmus, in which patients feel like they have a full rectum, even though it is physiologically empty. The recurring urge to defecate results in mucus discharges that resemble diarrhea. Inflammatory rectal involvement could be the cause, necessitating a gastroenterology consultation.
Anal incontinence can also cause false diarrhea. It is more common in elderly people residing in care homes and in women in the postpartum period. This condition is difficult to manage and requires referral to a gastroenterologist.
Chronic diarrhea: Could cancer be the culprit?
After ruling out false diarrhea, clinicians should be vigilant for warning signs. The first question to consider, said Dr. Hammoudi, is whether the chronic diarrhea is associated with a lesion. Several criteria should prompt a colonoscopy, especially to search for colorectal cancer lesions:
- Age greater than 50 years
- Personal or family history of colorectal cancer
- Recent changes in bowel habits
- Rectal bleeding
- Nighttime stools
- Unexplained weight loss
- Iron-deficiency anemia
Obvious causes of chronic diarrhea should be prioritized in the management plan. Medications top the list, with more than 500 treatments – for example, ACE inhibitors, proton pump inhibitors (PPIs), antidiabetic drugs, colchicine, magnesium, laxatives – known to have diarrhea as a side effect.
Certain dietary habits can also exacerbate diarrhea, such as milk consumption in cases of lactose intolerance, or excessive sugar intake, which can lead to osmotic diarrhea.
IBS is often at play
Once these causes have been ruled out, several etiological pathways should be investigated. The first relates to motility issues, which are the most common diarrhea-related problem, said Dr. Hammoudi.
This type of diarrhea is linked to rapid intestinal transit time and is characterized by postprandial bowel movements (occurring shortly after a meal). Here, patients experience urgency and notice identifiable food debris in their stools. It tends to stop when fasting and can be treated effectively with antidiarrheals.
Irritable bowel syndrome (IBS) is the main cause of rapid intestinal transit diarrhea. It is defined as recurrent abdominal pain (at least 1 day/week) over a period of 3 months, associated with two of the following criteria: pain eases or worsens on passing feces, change in frequency of bowel movements, change in the consistency of stools.
Symptoms may come on suddenly, sometimes after taking antibiotics, and may result in misdiagnosis.
IBS medications treat the symptoms. Antispasmodics, such as trimebutine, phloroglucinol (Spasfon), or pinaverium bromide (Dicetel) are recommended, even there can be a placebo effect. The antidiarrheal medication loperamide (Imodium) can also be used. Probiotics may be beneficial, as an imbalanced intestinal microbiota is often implicated.
Dietary changes can also have a positive impact. Encouraging a diet rich in fruit and vegetables to enhance fiber intake is advised. A low FODMAP (fermentable oligosaccharides, disaccharides, monosaccharides, and polyols) diet, targeting short-chain carbohydrates, can also be tried to identify foods to avoid, although it may be challenging to stick to.
Postinfectious IBS is a frequent cause of rapid intestinal transit diarrhea. It generally follows an episode of acute infectious diarrhea. “Symptoms may come on suddenly, sometimes after taking antibiotics, and may result in misdiagnosis,” said Dr. Hammoudi. This type of IBS often resolves spontaneously within 6 months.
Consider the possibility of SIBO
Another cause of rapid intestinal transit diarrhea is small intestinal bacterial overgrowth (SIBO). It is difficult to distinguish between IBS and SIBO. Often, affected patients are diabetic, overweight, or have had bowel surgery.
The only way to diagnose SIBO is by conducting a breath test to measure the production of hydrogen and methane by the microbiota after ingesting sugar. However, the test is difficult to access and not fully covered by social security plans in France, said Dr. Hammoudi.
In cases of suspected SIBO and severe symptoms, a 7- to 10-day course of antibiotics can be attempted to provide relief, although a diagnosis should be confirmed before considering this option, Dr. Hammoudi said.
Malabsorption diarrhea
Another major cause of chronic diarrhea is malabsorption, characterized by large, fatty stools that are difficult to flush. Despite a normal diet, this type of diarrhea is associated with weight loss and nutritional deficiencies.
Its diagnosis involves measuring fat in the stools (steatorrhea) and possibly testing fecal elastase, an enzyme produced by the pancreas that is involved in digestion.
The most important causes of malabsorption diarrhea are pancreatic insufficiency, celiac disease, and Crohn’s disease. Generally, any lesion in the small intestine can lead to malabsorption-related diarrhea.
Celiac disease, or gluten intolerance, is an autoimmune condition triggered by a reaction to gluten proteins. Several antibodies can be produced in the presence of gluten proteins. Diagnosis is confirmed by positive antitransglutaminase antibodies and a duodenal biopsy through esophagogastroduodenoscopy.
The only treatment for celiac disease is a lifelong gluten-free diet. Celiac disease is increasingly diagnosed in adults, said Dr. Hammoudi, and should be considered as a possibility. This condition must be distinguished from gluten sensitivity, which can cause digestive issues, possibly leading to rapid intestinal transit diarrhea. “The only treatment for celiac disease is a lifelong gluten-free diet,” Dr. Hammoudi added.
Crohn’s disease, a type of inflammatory bowel disease, affects the entire digestive tract, particularly the terminal small intestine, which promotes malabsorption. In ulcerative colitis, another IBD affecting the rectum, any associated rectal syndrome can result in false diarrhea with stools containing blood and mucus.
Osmotic diarrhea, on the other hand, is linked to the presence of highly osmotic agents in the digestive tract. This type of diarrhea is watery and short-lived, stopping once the agents are no longer absorbed. The main culprits are lactose (in cases of lactose intolerance) and laxatives.
Drug-induced microscopic colitis
Secretory diarrhea is characterized by excessive secretions by the digestive tract, leading to significant potassium loss. This type of diarrhea is not related to food intake and is resistant to fasting.
Major causes of secretory diarrhea include microscopic colitis, parasitic infections, and endocrine tumors. Between 10% and 15% of patients with chronic diarrhea and apparently normal colonoscopy have microscopic colitis.
Dr. Hammoudi advised specialists seeking to determine the cause of chronic diarrhea to routinely collect multilevel bowel biopsies during colonoscopies from macroscopically normal mucosa to rule out microscopic colitis.
Microscopic colitis is mainly linked to the use of medications like PPIs and NSAIDs. These drugs can induce malabsorption-related diarrhea by damaging the intestinal wall.
In addition to discontinuing the implicated medication, the treatment for microscopic colitis includes low-dose budesonide (multiple brands). Biologics used in IBD may also be considered in cases of recurrent colitis.
Finally, exudative enteropathy can be a distinct cause of chronic diarrhea. It is characterized by albumin leakage (Waldmann’s disease) and manifests with edema, malnutrition, and significant hypoalbuminemia.
This article was translated from the Medscape French edition. A version of this article appeared on Medscape.com.
Weight-loss drugs improve liver measures, too
BOSTON – With the current demand for type 2 diabetes drugs that both improve glycemic control and help patients shed large amounts of weight, liver specialists have speculated that the metabolic benefits could also extend to the liver.
Spoiler alert: they do.
, reported Takamasa Ohki, MD, PhD, and colleagues from the department of gastroenterology at Mitsui Memorial Hospital in Tokyo.
“Body weight reduction within 8 weeks after administration of these agents was an independent factor [that] contributed to rapid improvement of ALT. Maintenance of body weight and T2DM after normalization of ALT was also very important,” they wrote in a scientific poster presented at the annual meeting of the American Association for the Study of Liver Diseases.
The biggest losers benefit most
Dr. Ohki and colleagues evaluated the effectiveness of SGLT-2 inhibitors and GLP-1 agonists as treatment of MAFLD for patients with T2DM.
They conducted a retrospective study of 233 patients who had both conditions and who received either of the drug classes at their institution from June 2010 through December 2021; the most recent follow-up was in December 2022. The primary endpoint of the study was normalization of ALT values.
A total of 54 patients had a 3% or greater reduction in body weight within 8 weeks of beginning treatment with their respective drugs. The researchers found that for 47 of these patients (87%), ALT values normalized; the 12-, 24-, and 36-month cumulative normalization rates were 61%, 73%, and 80%, respectively.
In contrast, among the 179 patients who did not lose weight as robustly or rapidly, 137 (76.5%) demonstrated normalization of ALT, with cumulative normalization rates of 41%, 59% and 69%, respectively (P < .01).
In multivariate analysis that controlled for age, sex, smoking, hypertension, dyslipidemia, weight reduction, and antidiabetes drug use, body weight reduction of at least 3% within 8 weeks of beginning treatment with either an SLT-2 or GLP-1 agent was associated independently with normalization of ALT, with a hazard ratio (HR) of 0.67 (P = .028).
Improvement of T2DM was an independent predictor for ALT normalization (HR, 0.64; P = .015).
Other factors contributing to ALT normalization included use of sulfonylurea (HR, 0.63; P < .01) and insulin (HR, 0.54; P < .01).
In all, 103 of the 184 patients with initial normalization of ALT values experienced a recurrence of ALT elevation during follow-up. In multivariate analysis, body weight gain and exacerbation of T2DM were independent factors for ALT reexacerbation (HR, 0.52 and 0.48, respectively; P < .01 for both comparisons).
Duration of effect uncertain
Philip A. Newsome, PhD, FRCPE, professor of experimental hepatology and honorary consultant hepatologist at the University of Birmingham, England, who was not involved in the study, has conducted research into the metabolic effects of SGLT-2 inhibitors and GLP-1 agonists. In an interview, he said that both drug classes are likely to have positive near-term effects on metabolic dysfunction–associated steatohepatitis (MASH, formerly NASH) through their effects on glucose control and reduction in associated comorbidities.
“The unknown question,” he added, is what will happen in the long term. “I think there are some uncertainties around what proportion of patients will essentially be downstaged or downgraded such that they don’t develop any other problem; I suspect that will be the case in very many patients. However, I suspect there will also be a large proportion that end up requiring additional therapy above and beyond weight loss,” said Dr. Newsome.
The investigators did not report a funding source for the study. Dr. Ohki and colleagues have disclosed no relevant financial relationships. Dr. Newsome has consulted on behalf of his institution with Novo Nordisk, BMS, Gilead, Pfizer, Poxel, and Intercept and has received a grant from Pharmaxis and Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
BOSTON – With the current demand for type 2 diabetes drugs that both improve glycemic control and help patients shed large amounts of weight, liver specialists have speculated that the metabolic benefits could also extend to the liver.
Spoiler alert: they do.
, reported Takamasa Ohki, MD, PhD, and colleagues from the department of gastroenterology at Mitsui Memorial Hospital in Tokyo.
“Body weight reduction within 8 weeks after administration of these agents was an independent factor [that] contributed to rapid improvement of ALT. Maintenance of body weight and T2DM after normalization of ALT was also very important,” they wrote in a scientific poster presented at the annual meeting of the American Association for the Study of Liver Diseases.
The biggest losers benefit most
Dr. Ohki and colleagues evaluated the effectiveness of SGLT-2 inhibitors and GLP-1 agonists as treatment of MAFLD for patients with T2DM.
They conducted a retrospective study of 233 patients who had both conditions and who received either of the drug classes at their institution from June 2010 through December 2021; the most recent follow-up was in December 2022. The primary endpoint of the study was normalization of ALT values.
A total of 54 patients had a 3% or greater reduction in body weight within 8 weeks of beginning treatment with their respective drugs. The researchers found that for 47 of these patients (87%), ALT values normalized; the 12-, 24-, and 36-month cumulative normalization rates were 61%, 73%, and 80%, respectively.
In contrast, among the 179 patients who did not lose weight as robustly or rapidly, 137 (76.5%) demonstrated normalization of ALT, with cumulative normalization rates of 41%, 59% and 69%, respectively (P < .01).
In multivariate analysis that controlled for age, sex, smoking, hypertension, dyslipidemia, weight reduction, and antidiabetes drug use, body weight reduction of at least 3% within 8 weeks of beginning treatment with either an SLT-2 or GLP-1 agent was associated independently with normalization of ALT, with a hazard ratio (HR) of 0.67 (P = .028).
Improvement of T2DM was an independent predictor for ALT normalization (HR, 0.64; P = .015).
Other factors contributing to ALT normalization included use of sulfonylurea (HR, 0.63; P < .01) and insulin (HR, 0.54; P < .01).
In all, 103 of the 184 patients with initial normalization of ALT values experienced a recurrence of ALT elevation during follow-up. In multivariate analysis, body weight gain and exacerbation of T2DM were independent factors for ALT reexacerbation (HR, 0.52 and 0.48, respectively; P < .01 for both comparisons).
Duration of effect uncertain
Philip A. Newsome, PhD, FRCPE, professor of experimental hepatology and honorary consultant hepatologist at the University of Birmingham, England, who was not involved in the study, has conducted research into the metabolic effects of SGLT-2 inhibitors and GLP-1 agonists. In an interview, he said that both drug classes are likely to have positive near-term effects on metabolic dysfunction–associated steatohepatitis (MASH, formerly NASH) through their effects on glucose control and reduction in associated comorbidities.
“The unknown question,” he added, is what will happen in the long term. “I think there are some uncertainties around what proportion of patients will essentially be downstaged or downgraded such that they don’t develop any other problem; I suspect that will be the case in very many patients. However, I suspect there will also be a large proportion that end up requiring additional therapy above and beyond weight loss,” said Dr. Newsome.
The investigators did not report a funding source for the study. Dr. Ohki and colleagues have disclosed no relevant financial relationships. Dr. Newsome has consulted on behalf of his institution with Novo Nordisk, BMS, Gilead, Pfizer, Poxel, and Intercept and has received a grant from Pharmaxis and Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
BOSTON – With the current demand for type 2 diabetes drugs that both improve glycemic control and help patients shed large amounts of weight, liver specialists have speculated that the metabolic benefits could also extend to the liver.
Spoiler alert: they do.
, reported Takamasa Ohki, MD, PhD, and colleagues from the department of gastroenterology at Mitsui Memorial Hospital in Tokyo.
“Body weight reduction within 8 weeks after administration of these agents was an independent factor [that] contributed to rapid improvement of ALT. Maintenance of body weight and T2DM after normalization of ALT was also very important,” they wrote in a scientific poster presented at the annual meeting of the American Association for the Study of Liver Diseases.
The biggest losers benefit most
Dr. Ohki and colleagues evaluated the effectiveness of SGLT-2 inhibitors and GLP-1 agonists as treatment of MAFLD for patients with T2DM.
They conducted a retrospective study of 233 patients who had both conditions and who received either of the drug classes at their institution from June 2010 through December 2021; the most recent follow-up was in December 2022. The primary endpoint of the study was normalization of ALT values.
A total of 54 patients had a 3% or greater reduction in body weight within 8 weeks of beginning treatment with their respective drugs. The researchers found that for 47 of these patients (87%), ALT values normalized; the 12-, 24-, and 36-month cumulative normalization rates were 61%, 73%, and 80%, respectively.
In contrast, among the 179 patients who did not lose weight as robustly or rapidly, 137 (76.5%) demonstrated normalization of ALT, with cumulative normalization rates of 41%, 59% and 69%, respectively (P < .01).
In multivariate analysis that controlled for age, sex, smoking, hypertension, dyslipidemia, weight reduction, and antidiabetes drug use, body weight reduction of at least 3% within 8 weeks of beginning treatment with either an SLT-2 or GLP-1 agent was associated independently with normalization of ALT, with a hazard ratio (HR) of 0.67 (P = .028).
Improvement of T2DM was an independent predictor for ALT normalization (HR, 0.64; P = .015).
Other factors contributing to ALT normalization included use of sulfonylurea (HR, 0.63; P < .01) and insulin (HR, 0.54; P < .01).
In all, 103 of the 184 patients with initial normalization of ALT values experienced a recurrence of ALT elevation during follow-up. In multivariate analysis, body weight gain and exacerbation of T2DM were independent factors for ALT reexacerbation (HR, 0.52 and 0.48, respectively; P < .01 for both comparisons).
Duration of effect uncertain
Philip A. Newsome, PhD, FRCPE, professor of experimental hepatology and honorary consultant hepatologist at the University of Birmingham, England, who was not involved in the study, has conducted research into the metabolic effects of SGLT-2 inhibitors and GLP-1 agonists. In an interview, he said that both drug classes are likely to have positive near-term effects on metabolic dysfunction–associated steatohepatitis (MASH, formerly NASH) through their effects on glucose control and reduction in associated comorbidities.
“The unknown question,” he added, is what will happen in the long term. “I think there are some uncertainties around what proportion of patients will essentially be downstaged or downgraded such that they don’t develop any other problem; I suspect that will be the case in very many patients. However, I suspect there will also be a large proportion that end up requiring additional therapy above and beyond weight loss,” said Dr. Newsome.
The investigators did not report a funding source for the study. Dr. Ohki and colleagues have disclosed no relevant financial relationships. Dr. Newsome has consulted on behalf of his institution with Novo Nordisk, BMS, Gilead, Pfizer, Poxel, and Intercept and has received a grant from Pharmaxis and Boehringer Ingelheim.
A version of this article appeared on Medscape.com.
AT THE LIVER MEETING
Lower-extremity lymphedema associated with more skin cancer risk
TOPLINE:
.
METHODOLOGY:
- In the retrospective cohort study, researchers reviewed reports at Mayo Clinic for all patients who had LE lymphedema, limiting the review to those who had an ICD code for lymphedema.
- 4,437 patients with the ICD code from 2000 to 2020 were compared with 4,437 matched controls.
- The records of patients with skin cancer diagnoses were reviewed manually to determine whether the skin cancer, its management, or both were a cause of lymphedema; cancers that caused secondary lymphedema were excluded.
- This is the first large-scale study evaluating the association between LE lymphedema and LE skin cancer.
TAKEAWAY:
- 211 patients (4.6%) in the LE lymphedema group had any ICD code for LE skin cancer, compared with 89 (2%) in the control group.
- Among those with LE lymphedema, the risk for skin cancer was 1.98 times greater compared with those without lymphedema (95% confidence interval, 1.43-2.74; P < .001). Cases included all types of skin cancer.
- Nineteen of 24 patients with unilateral LE lymphedema had a history of immunosuppression.
- In the group of 24 patients with unilateral LE lymphedema, the lymphedematous LE was more likely to have one or more skin cancers than were the unaffected LE (87.5% vs. 33.3%; P < .05), and skin cancer was 2.65 times more likely to develop on the affected LE than in the unaffected LE (95% CI, 1.17-5.99; P = .02).
IN PRACTICE:
“Our findings suggest the need for a relatively high degree of suspicion of skin cancer at sites with lymphedema,” senior author, Afsaneh Alavi, MD, professor of dermatology at the Mayo Clinic, said in a Mayo Clinic press release reporting the results.
SOURCE:
The study was conducted by researchers at the Mayo Clinic and Meharry Medical College, Nashville. It was published in the November 2023 Mayo Clinic Proceedings.
LIMITATIONS:
This was a single-center retrospective study, and patients with LE lymphedema may be overdiagnosed with LE skin cancer because they have a greater number of examinations.
DISCLOSURES:
Dr. Alavi reports having been a consultant for AbbVie, Boehringer Ingelheim, InflaRx, Novartis, and UCB SA and an investigator for Processa Pharmaceuticals and Boehringer Ingelheim. The other authors had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- In the retrospective cohort study, researchers reviewed reports at Mayo Clinic for all patients who had LE lymphedema, limiting the review to those who had an ICD code for lymphedema.
- 4,437 patients with the ICD code from 2000 to 2020 were compared with 4,437 matched controls.
- The records of patients with skin cancer diagnoses were reviewed manually to determine whether the skin cancer, its management, or both were a cause of lymphedema; cancers that caused secondary lymphedema were excluded.
- This is the first large-scale study evaluating the association between LE lymphedema and LE skin cancer.
TAKEAWAY:
- 211 patients (4.6%) in the LE lymphedema group had any ICD code for LE skin cancer, compared with 89 (2%) in the control group.
- Among those with LE lymphedema, the risk for skin cancer was 1.98 times greater compared with those without lymphedema (95% confidence interval, 1.43-2.74; P < .001). Cases included all types of skin cancer.
- Nineteen of 24 patients with unilateral LE lymphedema had a history of immunosuppression.
- In the group of 24 patients with unilateral LE lymphedema, the lymphedematous LE was more likely to have one or more skin cancers than were the unaffected LE (87.5% vs. 33.3%; P < .05), and skin cancer was 2.65 times more likely to develop on the affected LE than in the unaffected LE (95% CI, 1.17-5.99; P = .02).
IN PRACTICE:
“Our findings suggest the need for a relatively high degree of suspicion of skin cancer at sites with lymphedema,” senior author, Afsaneh Alavi, MD, professor of dermatology at the Mayo Clinic, said in a Mayo Clinic press release reporting the results.
SOURCE:
The study was conducted by researchers at the Mayo Clinic and Meharry Medical College, Nashville. It was published in the November 2023 Mayo Clinic Proceedings.
LIMITATIONS:
This was a single-center retrospective study, and patients with LE lymphedema may be overdiagnosed with LE skin cancer because they have a greater number of examinations.
DISCLOSURES:
Dr. Alavi reports having been a consultant for AbbVie, Boehringer Ingelheim, InflaRx, Novartis, and UCB SA and an investigator for Processa Pharmaceuticals and Boehringer Ingelheim. The other authors had no disclosures.
A version of this article first appeared on Medscape.com.
TOPLINE:
.
METHODOLOGY:
- In the retrospective cohort study, researchers reviewed reports at Mayo Clinic for all patients who had LE lymphedema, limiting the review to those who had an ICD code for lymphedema.
- 4,437 patients with the ICD code from 2000 to 2020 were compared with 4,437 matched controls.
- The records of patients with skin cancer diagnoses were reviewed manually to determine whether the skin cancer, its management, or both were a cause of lymphedema; cancers that caused secondary lymphedema were excluded.
- This is the first large-scale study evaluating the association between LE lymphedema and LE skin cancer.
TAKEAWAY:
- 211 patients (4.6%) in the LE lymphedema group had any ICD code for LE skin cancer, compared with 89 (2%) in the control group.
- Among those with LE lymphedema, the risk for skin cancer was 1.98 times greater compared with those without lymphedema (95% confidence interval, 1.43-2.74; P < .001). Cases included all types of skin cancer.
- Nineteen of 24 patients with unilateral LE lymphedema had a history of immunosuppression.
- In the group of 24 patients with unilateral LE lymphedema, the lymphedematous LE was more likely to have one or more skin cancers than were the unaffected LE (87.5% vs. 33.3%; P < .05), and skin cancer was 2.65 times more likely to develop on the affected LE than in the unaffected LE (95% CI, 1.17-5.99; P = .02).
IN PRACTICE:
“Our findings suggest the need for a relatively high degree of suspicion of skin cancer at sites with lymphedema,” senior author, Afsaneh Alavi, MD, professor of dermatology at the Mayo Clinic, said in a Mayo Clinic press release reporting the results.
SOURCE:
The study was conducted by researchers at the Mayo Clinic and Meharry Medical College, Nashville. It was published in the November 2023 Mayo Clinic Proceedings.
LIMITATIONS:
This was a single-center retrospective study, and patients with LE lymphedema may be overdiagnosed with LE skin cancer because they have a greater number of examinations.
DISCLOSURES:
Dr. Alavi reports having been a consultant for AbbVie, Boehringer Ingelheim, InflaRx, Novartis, and UCB SA and an investigator for Processa Pharmaceuticals and Boehringer Ingelheim. The other authors had no disclosures.
A version of this article first appeared on Medscape.com.
AI tool perfect in study of inflammatory diseases
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
Artificial intelligence can distinguish overlapping inflammatory conditions with total accuracy, according to a new study presented at the annual meeting of the American College of Rheumatology.
Texas pediatricians faced a conundrum during the pandemic. Endemic typhus, a flea-borne tropical infection common to the region, is nearly indistinguishable from multisystem inflammatory syndrome in children (MIS-C), a rare condition set in motion by SARS-CoV-2 infection. Children with either ailment had seemingly identical symptoms: fever, rash, gastrointestinal issues, and in need of swift treatment. A diagnosis of endemic typhus can take 4-6 days to confirm.
Tiphanie Vogel, MD, PhD, a pediatric rheumatologist at Texas Children’s Hospital, Houston, and colleagues sought to create a tool to hasten diagnosis and, ideally, treatment. To do so, they incorporated machine learning and clinical factors available within the first 6 hours of the onset of symptoms.
The team analyzed 49 demographic, clinical, and laboratory measures from the medical records of 133 children with MIS-C and 87 with endemic typhus. Using deep learning, they narrowed the model to 30 essential features that became the backbone of AI-MET, a two-phase clinical-decision support system.
Phase 1 uses 17 clinical factors and can be performed on paper. If a patient’s score in phase 1 is not determinative, clinicians proceed to phase 2, which uses an additional 13 weighted factors and machine learning.
In testing, the two-part tool classified each of the 220 test patients perfectly. And it diagnosed a second group of 111 patients with MIS-C with 99% (110/111) accuracy.
Of note, “that first step classifies [a patient] correctly half of the time,” Dr. Vogel said, so the second, AI phase of the tool was necessary for only half of cases. Dr. Vogel said that’s a good sign; it means that the tool is useful in settings where AI may not always be feasible, like in a busy ED.
Melissa Mizesko, MD, a pediatric rheumatologist at Driscoll Children’s Hospital in Corpus Christi, Tex., said that the new tool could help clinicians streamline care. When cases of MIS-C peaked in Texas, clinicians often would start sick children on doxycycline and treat for MIS-C at the same time, then wait to see whether the antibiotic brought the fever down.
“This [new tool] is helpful if you live in a part of the country that has typhus,” said Jane Burns, MD, director of the Kawasaki Disease Research Center at the University of California, San Diego, who helped develop a similar AI-based tool to distinguish MIS-C from Kawasaki disease. But she encouraged the researchers to expand their testing to include other conditions. Although the AI model Dr. Vogel’s group developed can pinpoint MIS-C or endemic typhus, what if a child has neither condition? “It’s not often you’re dealing with a diagnosis between just two specific diseases,” Dr. Burns said.
Dr. Vogel is also interested in making AI-MET more efficient. “This go-round we prioritized perfect accuracy,” she said. But 30 clinical factors, with 17 of them recorded and calculated by hand, is a lot. “Could we still get this to be very accurate, maybe not perfect, with less inputs?”
In addition to refining AI-MET, which Texas Children’s eventually hopes to make available to other institutions, Dr. Vogel and associates are also considering other use cases for AI. Lupus is one option. “Maybe with machine learning we could identify clues at diagnosis that would help recommend targeted treatment,” she said
Dr. Vogel disclosed potential conflicts of interest with Moderna, Novartis, Pfizer, and SOBI. Dr. Burns and Dr. Mizesko disclosed no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
FROM ACR 2023
Jury still out on whether green tea lowers colon cancer risk
Can green tea lower your risk of colorectal cancer? It depends on who – and what research – you believe.
Some researchers have found little or no significant risk from drinking the popular tea, while others point to a potential benefit. Now add two more studies – one that found no reduced risk and another that seems to strengthen the link between green tea and a lower risk of colon cancer.
Randomized controlled trials – where some people get randomly assigned to drink green tea and others do not – are considered the gold standard of medical research. Combine the findings from several of these trials, the thinking goes, and the findings get even stronger.
Combining random trials so far shows no advantage from green tea. But there may still be a benefit, said lead researcher Vishal Chandel, MD, who is affiliated with Suburban Community Hospital in Norristown, Pa. It could be that there are just not enough randomized controlled trials yet to show green tea has a protective effect.
“Many, many factors contribute to colorectal cancer, and one of them is diet. One thing which struck me was tea, because tea is something that people consume all over the world, and it has shown some stronger effects in Japan and in China,” said Dr. Chandel.
Comparing hundreds of people
Dr. Chandel and colleagues found three randomized controlled trials that looked at the link between green tea and colorectal cancer risk. Combined, the data included 451 people with colorectal cancer and 460 others without cancer who made up a control, or comparison, group.
They found green tea consumption did not reduce the risk of colorectal cancer in a statically significant way.
“There are only three randomized controlled trials from anywhere concerning green tea and colon cancer,” Dr. Chandel said. “We really need more. If we had 7, 8, or 10 … I’m very positive we will have a much stronger association to say that green tea can have a positive effect.”
Comparing thousands of people
Dr. Chandel and colleagues also performed another study where they looked at less rigorous evidence – 10 cohort studies and 15 prospective case-control studies. These studies included 198,488 cancer cases and 581,556 controls. This time, they found a stronger link between green tea and a reduced risk of colorectal cancer.
The “meta-analysis results indicate a lower tendency to develop colorectal cancer with green tea consumption, with reduced risk of colorectal cancer more pronounced in Asia than America or Europe,” the authors note. “Although there is insufficient epidemiological data to conclude at present that green tea can have protective effects in human beings.”
Dr. Chandel presented the findings of both studies in Vancouver at the American College of Gastroenterology annual scientific meeting.
Why green tea?
Dr. Chandel said he studied colorectal cancer because it is the third most diagnosed cancer worldwide, accounting for about 10% of all new cancer cases in 2020, according to the World Health Organization’s Global Cancer Observatory data. It is also a common cause of cancer death globally, second only to lung cancer.
Green tea contains high level of polyphenols known as catechins. The main catechin in green tea believed to provide cancer protective effects is epigallocatechin-3 gallate (EGCG). EGCG “has been shown in some studies to inhibit or prevent colon cancer,” Dr. Chandel said.
EGCG is present in higher amounts in green tea, compared with black or oolong tea, because green tea is made from unfermented, unoxidized tea leaves.
Difficult to read the tea leaves
These studies “add to the literature, which remains undefined regarding the role of green tea in reducing the risk of colorectal cancer,” Catherine Eng, MD, a spokesperson for the American Society of Clinical Oncology, said when asked to comment.
Although combining three trials did not reveal a significant benefit, looking at a greater number of studies did in some populations, said Dr. Eng, codirector of gastrointestinal oncology and chair of surgical and medical oncology at Vanderbilt-Ingram Cancer Center in Nashville.
“Potential benefit for green tea in reducing the risk of colorectal cancer was noted in the Asian cases but was not found to be statistically significant in the European or U.S. studies,” she said. “Currently, the role of dietary consumption of green tea on reducing the risk of colorectal cancer is not well established and requires further investigation.”
A version of this article appeared on WebMD.com.
Can green tea lower your risk of colorectal cancer? It depends on who – and what research – you believe.
Some researchers have found little or no significant risk from drinking the popular tea, while others point to a potential benefit. Now add two more studies – one that found no reduced risk and another that seems to strengthen the link between green tea and a lower risk of colon cancer.
Randomized controlled trials – where some people get randomly assigned to drink green tea and others do not – are considered the gold standard of medical research. Combine the findings from several of these trials, the thinking goes, and the findings get even stronger.
Combining random trials so far shows no advantage from green tea. But there may still be a benefit, said lead researcher Vishal Chandel, MD, who is affiliated with Suburban Community Hospital in Norristown, Pa. It could be that there are just not enough randomized controlled trials yet to show green tea has a protective effect.
“Many, many factors contribute to colorectal cancer, and one of them is diet. One thing which struck me was tea, because tea is something that people consume all over the world, and it has shown some stronger effects in Japan and in China,” said Dr. Chandel.
Comparing hundreds of people
Dr. Chandel and colleagues found three randomized controlled trials that looked at the link between green tea and colorectal cancer risk. Combined, the data included 451 people with colorectal cancer and 460 others without cancer who made up a control, or comparison, group.
They found green tea consumption did not reduce the risk of colorectal cancer in a statically significant way.
“There are only three randomized controlled trials from anywhere concerning green tea and colon cancer,” Dr. Chandel said. “We really need more. If we had 7, 8, or 10 … I’m very positive we will have a much stronger association to say that green tea can have a positive effect.”
Comparing thousands of people
Dr. Chandel and colleagues also performed another study where they looked at less rigorous evidence – 10 cohort studies and 15 prospective case-control studies. These studies included 198,488 cancer cases and 581,556 controls. This time, they found a stronger link between green tea and a reduced risk of colorectal cancer.
The “meta-analysis results indicate a lower tendency to develop colorectal cancer with green tea consumption, with reduced risk of colorectal cancer more pronounced in Asia than America or Europe,” the authors note. “Although there is insufficient epidemiological data to conclude at present that green tea can have protective effects in human beings.”
Dr. Chandel presented the findings of both studies in Vancouver at the American College of Gastroenterology annual scientific meeting.
Why green tea?
Dr. Chandel said he studied colorectal cancer because it is the third most diagnosed cancer worldwide, accounting for about 10% of all new cancer cases in 2020, according to the World Health Organization’s Global Cancer Observatory data. It is also a common cause of cancer death globally, second only to lung cancer.
Green tea contains high level of polyphenols known as catechins. The main catechin in green tea believed to provide cancer protective effects is epigallocatechin-3 gallate (EGCG). EGCG “has been shown in some studies to inhibit or prevent colon cancer,” Dr. Chandel said.
EGCG is present in higher amounts in green tea, compared with black or oolong tea, because green tea is made from unfermented, unoxidized tea leaves.
Difficult to read the tea leaves
These studies “add to the literature, which remains undefined regarding the role of green tea in reducing the risk of colorectal cancer,” Catherine Eng, MD, a spokesperson for the American Society of Clinical Oncology, said when asked to comment.
Although combining three trials did not reveal a significant benefit, looking at a greater number of studies did in some populations, said Dr. Eng, codirector of gastrointestinal oncology and chair of surgical and medical oncology at Vanderbilt-Ingram Cancer Center in Nashville.
“Potential benefit for green tea in reducing the risk of colorectal cancer was noted in the Asian cases but was not found to be statistically significant in the European or U.S. studies,” she said. “Currently, the role of dietary consumption of green tea on reducing the risk of colorectal cancer is not well established and requires further investigation.”
A version of this article appeared on WebMD.com.
Can green tea lower your risk of colorectal cancer? It depends on who – and what research – you believe.
Some researchers have found little or no significant risk from drinking the popular tea, while others point to a potential benefit. Now add two more studies – one that found no reduced risk and another that seems to strengthen the link between green tea and a lower risk of colon cancer.
Randomized controlled trials – where some people get randomly assigned to drink green tea and others do not – are considered the gold standard of medical research. Combine the findings from several of these trials, the thinking goes, and the findings get even stronger.
Combining random trials so far shows no advantage from green tea. But there may still be a benefit, said lead researcher Vishal Chandel, MD, who is affiliated with Suburban Community Hospital in Norristown, Pa. It could be that there are just not enough randomized controlled trials yet to show green tea has a protective effect.
“Many, many factors contribute to colorectal cancer, and one of them is diet. One thing which struck me was tea, because tea is something that people consume all over the world, and it has shown some stronger effects in Japan and in China,” said Dr. Chandel.
Comparing hundreds of people
Dr. Chandel and colleagues found three randomized controlled trials that looked at the link between green tea and colorectal cancer risk. Combined, the data included 451 people with colorectal cancer and 460 others without cancer who made up a control, or comparison, group.
They found green tea consumption did not reduce the risk of colorectal cancer in a statically significant way.
“There are only three randomized controlled trials from anywhere concerning green tea and colon cancer,” Dr. Chandel said. “We really need more. If we had 7, 8, or 10 … I’m very positive we will have a much stronger association to say that green tea can have a positive effect.”
Comparing thousands of people
Dr. Chandel and colleagues also performed another study where they looked at less rigorous evidence – 10 cohort studies and 15 prospective case-control studies. These studies included 198,488 cancer cases and 581,556 controls. This time, they found a stronger link between green tea and a reduced risk of colorectal cancer.
The “meta-analysis results indicate a lower tendency to develop colorectal cancer with green tea consumption, with reduced risk of colorectal cancer more pronounced in Asia than America or Europe,” the authors note. “Although there is insufficient epidemiological data to conclude at present that green tea can have protective effects in human beings.”
Dr. Chandel presented the findings of both studies in Vancouver at the American College of Gastroenterology annual scientific meeting.
Why green tea?
Dr. Chandel said he studied colorectal cancer because it is the third most diagnosed cancer worldwide, accounting for about 10% of all new cancer cases in 2020, according to the World Health Organization’s Global Cancer Observatory data. It is also a common cause of cancer death globally, second only to lung cancer.
Green tea contains high level of polyphenols known as catechins. The main catechin in green tea believed to provide cancer protective effects is epigallocatechin-3 gallate (EGCG). EGCG “has been shown in some studies to inhibit or prevent colon cancer,” Dr. Chandel said.
EGCG is present in higher amounts in green tea, compared with black or oolong tea, because green tea is made from unfermented, unoxidized tea leaves.
Difficult to read the tea leaves
These studies “add to the literature, which remains undefined regarding the role of green tea in reducing the risk of colorectal cancer,” Catherine Eng, MD, a spokesperson for the American Society of Clinical Oncology, said when asked to comment.
Although combining three trials did not reveal a significant benefit, looking at a greater number of studies did in some populations, said Dr. Eng, codirector of gastrointestinal oncology and chair of surgical and medical oncology at Vanderbilt-Ingram Cancer Center in Nashville.
“Potential benefit for green tea in reducing the risk of colorectal cancer was noted in the Asian cases but was not found to be statistically significant in the European or U.S. studies,” she said. “Currently, the role of dietary consumption of green tea on reducing the risk of colorectal cancer is not well established and requires further investigation.”
A version of this article appeared on WebMD.com.
Apixaban cuts stroke but ups bleeding in subclinical AFib: ARTESIA
in the ARTESIA study.
The results appear to contrast somewhat with the recently reported NOAH-AFNET 6 trial, which failed to show a reduction in stroke with the anticoagulant edoxaban versus placebo in a similar patient group, but that trial was stopped early and so was underpowered.
However, the lead investigators of both trials say the studies actually show consistent results – both found a lower rate of stroke than expected in this population, but the confidence intervals for stroke reduction with anticoagulation overlap, suggesting there is likely some effect, albeit less than that in clinical AFib.
The big question is whether the reduction in stroke with anticoagulation outweighs the increase in major bleeding.
A new meta-analysis of the two trials showed that “oral anticoagulation with edoxaban or apixaban reduces the risk of ischemic stroke by approximately one-third and increases major bleeding by roughly double.”
In absolute numbers, there were three fewer ischemic strokes per 1,000 patient-years with anticoagulation in the two trials combined, at the cost of seven more major bleeds.
The lead investigators of the two trials have somewhat different opinions on how these findings may translate into clinical practice.
Jeff Healey, MD, Population Health Research Institute, McMaster University, Hamilton, Ont., lead investigator of the ARTESIA trial, believes that the risks and benefits need to be assessed in individual patients, but there should be some patient groups that will benefit from anticoagulation treatment.
“In patients with pacemakers or implantable loop recorders with continuous monitoring, subclinical AF[ib] is detected in about one third of patients, so this is extremely common,” he said in an interview. “The question is whether this is just a normal feature of getting older or is this like AF[ib] that we see in the clinic which increases stroke risk, and I think we can conclude from ARTESIA that this subclinical AF[ib] is associated with an increased risk of stroke, although that is lower than the risk with clinical AF[ib], and that it can be reduced by anticoagulation.”
Until recently it hasn’t been possible to quantify the risk associated with subclinical AFib, he noted. “But now we have a rich dataset to use to see if we can tease out some specifics on this. Future analyses of this dataset will help define patients where the benefits outweigh the risks of bleeding. For now, I think we can look at the data in a qualitative way and consider the totality of risk factors in each patient – their bleeding risk, stroke risk, how much AF[ib] they have, and make a decision as to whether to give anticoagulation or not.”
But Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), lead investigator of the NOAH-AFNET 6 trial said: “Both trials showed the stroke rate is low in these patients – about 1% per year – and that anticoagulation can reduce it a bit further at the expense of increasing major bleeding. I don’t believe the AF[ib] episodes picked up on these devices constitute a sufficient stroke risk to warrant anticoagulation, given the bleeding risk.”
Dr. Kirchhof suggests an alternate approach of performing further traditional AFib monitoring on these patients.
“I think going forward in my practice, when we come across this device-detected AF[ib], we will do further investigations with an established method for detecting AF[ib] involving surface ECG monitoring – maybe a 3-day or 7-day Holter. If that shows AF[ib], then we will be on firm ground to start anticoagulation. If that doesn’t show AF[ib], we will probably not use anticoagulation.”
The ARTESIA trial and the meta-analysis of the two trials were both presented at the annual scientific sessions of the American Heart Association. Both studies were also simultaneously published online – ARTESIA in the New England Journal of Medicine and the meta-analysis in Circulation.
ARTESIA
For the ARTESIA study, 4012 patients with device-detected AFib and other clinical risk factors for stroke were randomly assigned to treatment with apixaban (5 mg twice daily) or aspirin (81 mg daily).
After a mean follow-up of 3.5 years, the primary endpoint – stroke or systemic embolism – occurred in 55 patients in the apixaban group (0.78% per patient-year), compared with 86 patients in the aspirin group (1.24% per patient-year), giving a hazard ratio of 0.63 (95% confidence interval, 0.45-0.88; P = .007).
“The risk of stroke or systemic embolism was lower by 37% with apixaban than with aspirin, and the risk of disabling or fatal stroke was lower by 49%,” Dr. Healey reported.
In the “on-treatment” population, the rate of major bleeding was 1.71% per patient-year in the apixaban group and 0.94% per patient-year in the aspirin group (HR, 1.80; 95% CI, 1.26-2.57; P = .001).
Fatal bleeding occurred in five patients in the apixaban group and eight patients in the aspirin group. Symptomatic intracranial hemorrhage occurred in 12 patients with apixaban and 15 patients with aspirin.
One of the main findings of the trial is the lower-than-expected risk of ischemic stroke in this population – about 1% per year in the aspirin group, which was reduced to 0.64% per year in the apixaban group.
The authors noted that “simply counting strokes as compared with bleeding events might suggest a neutral overall effect. With apixaban as compared with aspirin, 31 fewer cases of stroke or systemic embolism were seen in the intention-to-treat analysis, as compared with 39 more major bleeding events in the on-treatment analysis.”
However, they pointed out that strokes involve permanent loss of brain tissue, whereas major bleeding is usually reversible, with most patients having complete recovery, which was the case in this study.
“Thus, on the basis of the considerably greater severity of the stroke events prevented than the bleeding events caused, we believe that these findings favor consideration of the use of oral anticoagulation for patients with risk factors for stroke in whom subclinical atrial fibrillation develops,” they concluded.
First well-powered trial addressing this question
Discussing the ARTESIA trial at an AHA press conference, Christine Albert, MD, Cedars-Sinai Medical Center, Los Angeles, said: “I want to emphasize how important this trial is.”
She explained that current guidelines do not recommend any treatment for patients with device-detected AFib that is not shown on ECG, even though it is known this confers some excess risk of stroke.
“ARTESIA is the first well-powered, long-term trial looking at this question,” she said. “It found a clear reduction in the risk of stroke/systemic embolism with apixaban vs aspirin, but there was also a significant amount of bleeding – about an 80% increase. The question is whether the benefit on stroke is worth it given the bleeding risk.”
Dr. Albert highlighted the low absolute risk of stroke in this study population of around 1.2%, pointing out that even with the 37% relative reduction with anticoagulation, stroke is only reduced in absolute terms by 0.4%.
“We are going to have to take this back to committees and guidelines and look at the balance between the benefit on stroke and the increase in bleeding,” she concluded.
Noting that observational studies have shown that the duration of AFib impacts the risk of stroke, Dr. Albert suggested that patients with longer-duration AFib may benefit from anticoagulation to a greater extent; and given that the bleeding seen in ARTESIA was mainly GI bleeding, it might be possible to screen out patients at high risk of GI bleeding.
She also pointed out that a lot of patients discontinued anticoagulation treatment in both ARTESIA and NOAH-AFNET 6, showing that this is not an easy strategy for elderly patients.
In an editorial accompanying publication of the ARTESIA trial, Emma Svennberg, MD, Karolinska Institute, Stockholm, also concluded that, “going forward, we must balance the increased bleeding risks with the risk for disabling strokes,” and that “future substudies and meta-analyses may provide further insights regarding treatment benefits in specific subgroups.”
NOAH-AFNET 6: New subgroup analysis
The previously reported NOAH-AFNET 6 study randomly assigned 2,538 patients with subclinical AFib and additional risk factors for stroke to anticoagulation with edoxaban or placebo. The trial was stopped early, so it was underpowered – but it found no difference between groups in the incidence of the composite endpoint of stroke, systemic embolism, or death from cardiovascular causes or in the incidence of stroke, although there was higher risk of major bleeding.
Again, there was a low rate of stroke in this trial with just 49 strokes in total in the whole study. The NOAH-AFNET-6 investigators concluded that these patients should not receive anticoagulation because the risk of bleeding outweighed any potential benefits.
A new subanalysis of the 259 patients who had durations of subclinical AFib of 24 hours or longer in the NOAH-AFNET 6 trial was presented at the AHA meeting, and simultaneously published online in the European Heart Journal.
This showed that the rate of stroke also appeared low in patients with these long durations of subclinical AFib, and that there was no interaction between the duration of subclinical AFib and the efficacy and safety of oral anticoagulation.
But with such a low number of events in the study as a whole and in the long duration subclinical AFib subgroup (in which there were just two strokes in each treatment group), this analysis was unlikely to show a difference, Dr. Kirchhof commented.
The subgroup analysis did, however, show that patients experiencing subclinical AFib durations of 24 hours or more were more likely to develop clinical AFib over time than those with shorter durations, suggesting the need for regular ECGs in these patients.
Dr. Kirchhof said better methods are needed to detect patients with subclinical AFib at high risk of stroke. “I don’t think our clinical stroke risk factor scores such as CHA2DS2-VASc are sufficient to detect high-risk patients. Patients in both NOAH-AFNET 6 and ARTESIA had a median CHA2DS2-VASc score of 4, but they had a stroke rate of just 1% per year,” he noted.
The meta-analysis of the two trials showed that the results from both are consistent, with an overall reduction in ischemic stroke with oral anticoagulation (relative risk, 0.68). Oral anticoagulation also reduced a composite of cardiovascular death, all-cause stroke, peripheral arterial embolism, myocardial infarction, or pulmonary embolism (RR, 0.85).
There was no significant difference in cardiovascular death (RR, 0.95) or all-cause mortality (RR, 1.08), but anticoagulation significantly increased major bleeding (RR, 1.62).
Aspirin use complicates results
Dr. Healey said further analyses of the ARTESIA data will try to tease out the effect of concomitant aspirin use in the trial.
He explained that patients in this trial were allowed to take a single antiplatelet agent on top of study therapy.
“It is difficult to work out the exact use of antiplatelet therapy as it changed throughout the study,” he said. “About two-thirds were taking antiplatelet agents at the time of enrollment into the trial, but this decreased throughout the study. Many clinicians stopped open-label antiplatelet therapy during the trial when new evidence came out to suggest that there was no added benefit of adding aspirin on top of anticoagulants.
“We need to look carefully as to what impact that may have had,” Dr. Healey added. “We know from other studies that adding an antiplatelet on top of an anticoagulant doesn’t do much to thromboembolic events, but it approximately doubles the risk of major bleeding.”
In contrast, the NOAH-AFNET trial did not allow aspirin use in the anticoagulation group and aspirin was taken by around half the patients in the placebo group who had an indication for its use.
The authors of the meta-analysis pointed out that the omission of aspirin in nearly half of the control patients in NOAH-AFNET 6 and the early termination of the trial may have led to a slightly higher estimate for excess major bleeding with anticoagulation.
The ARTESIA study was supported by the Canadian Institutes for Health Research, the Bristol Myers Squibb-Pfizer Alliance, the Heart and Stroke Foundation of Canada, the Canadian Stroke Prevention Intervention Network, Hamilton Health Sciences, the Advancing Clinical Trials Network and the Population Health Research Institute. Dr. Healey reported research grants and speaking fees from BMS/Pfizer Alliance, Servier, Novartis, Boston Scientific, Medtronic; and acts as a consultant to Bayer, Servier and Boston Scientific. The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
in the ARTESIA study.
The results appear to contrast somewhat with the recently reported NOAH-AFNET 6 trial, which failed to show a reduction in stroke with the anticoagulant edoxaban versus placebo in a similar patient group, but that trial was stopped early and so was underpowered.
However, the lead investigators of both trials say the studies actually show consistent results – both found a lower rate of stroke than expected in this population, but the confidence intervals for stroke reduction with anticoagulation overlap, suggesting there is likely some effect, albeit less than that in clinical AFib.
The big question is whether the reduction in stroke with anticoagulation outweighs the increase in major bleeding.
A new meta-analysis of the two trials showed that “oral anticoagulation with edoxaban or apixaban reduces the risk of ischemic stroke by approximately one-third and increases major bleeding by roughly double.”
In absolute numbers, there were three fewer ischemic strokes per 1,000 patient-years with anticoagulation in the two trials combined, at the cost of seven more major bleeds.
The lead investigators of the two trials have somewhat different opinions on how these findings may translate into clinical practice.
Jeff Healey, MD, Population Health Research Institute, McMaster University, Hamilton, Ont., lead investigator of the ARTESIA trial, believes that the risks and benefits need to be assessed in individual patients, but there should be some patient groups that will benefit from anticoagulation treatment.
“In patients with pacemakers or implantable loop recorders with continuous monitoring, subclinical AF[ib] is detected in about one third of patients, so this is extremely common,” he said in an interview. “The question is whether this is just a normal feature of getting older or is this like AF[ib] that we see in the clinic which increases stroke risk, and I think we can conclude from ARTESIA that this subclinical AF[ib] is associated with an increased risk of stroke, although that is lower than the risk with clinical AF[ib], and that it can be reduced by anticoagulation.”
Until recently it hasn’t been possible to quantify the risk associated with subclinical AFib, he noted. “But now we have a rich dataset to use to see if we can tease out some specifics on this. Future analyses of this dataset will help define patients where the benefits outweigh the risks of bleeding. For now, I think we can look at the data in a qualitative way and consider the totality of risk factors in each patient – their bleeding risk, stroke risk, how much AF[ib] they have, and make a decision as to whether to give anticoagulation or not.”
But Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), lead investigator of the NOAH-AFNET 6 trial said: “Both trials showed the stroke rate is low in these patients – about 1% per year – and that anticoagulation can reduce it a bit further at the expense of increasing major bleeding. I don’t believe the AF[ib] episodes picked up on these devices constitute a sufficient stroke risk to warrant anticoagulation, given the bleeding risk.”
Dr. Kirchhof suggests an alternate approach of performing further traditional AFib monitoring on these patients.
“I think going forward in my practice, when we come across this device-detected AF[ib], we will do further investigations with an established method for detecting AF[ib] involving surface ECG monitoring – maybe a 3-day or 7-day Holter. If that shows AF[ib], then we will be on firm ground to start anticoagulation. If that doesn’t show AF[ib], we will probably not use anticoagulation.”
The ARTESIA trial and the meta-analysis of the two trials were both presented at the annual scientific sessions of the American Heart Association. Both studies were also simultaneously published online – ARTESIA in the New England Journal of Medicine and the meta-analysis in Circulation.
ARTESIA
For the ARTESIA study, 4012 patients with device-detected AFib and other clinical risk factors for stroke were randomly assigned to treatment with apixaban (5 mg twice daily) or aspirin (81 mg daily).
After a mean follow-up of 3.5 years, the primary endpoint – stroke or systemic embolism – occurred in 55 patients in the apixaban group (0.78% per patient-year), compared with 86 patients in the aspirin group (1.24% per patient-year), giving a hazard ratio of 0.63 (95% confidence interval, 0.45-0.88; P = .007).
“The risk of stroke or systemic embolism was lower by 37% with apixaban than with aspirin, and the risk of disabling or fatal stroke was lower by 49%,” Dr. Healey reported.
In the “on-treatment” population, the rate of major bleeding was 1.71% per patient-year in the apixaban group and 0.94% per patient-year in the aspirin group (HR, 1.80; 95% CI, 1.26-2.57; P = .001).
Fatal bleeding occurred in five patients in the apixaban group and eight patients in the aspirin group. Symptomatic intracranial hemorrhage occurred in 12 patients with apixaban and 15 patients with aspirin.
One of the main findings of the trial is the lower-than-expected risk of ischemic stroke in this population – about 1% per year in the aspirin group, which was reduced to 0.64% per year in the apixaban group.
The authors noted that “simply counting strokes as compared with bleeding events might suggest a neutral overall effect. With apixaban as compared with aspirin, 31 fewer cases of stroke or systemic embolism were seen in the intention-to-treat analysis, as compared with 39 more major bleeding events in the on-treatment analysis.”
However, they pointed out that strokes involve permanent loss of brain tissue, whereas major bleeding is usually reversible, with most patients having complete recovery, which was the case in this study.
“Thus, on the basis of the considerably greater severity of the stroke events prevented than the bleeding events caused, we believe that these findings favor consideration of the use of oral anticoagulation for patients with risk factors for stroke in whom subclinical atrial fibrillation develops,” they concluded.
First well-powered trial addressing this question
Discussing the ARTESIA trial at an AHA press conference, Christine Albert, MD, Cedars-Sinai Medical Center, Los Angeles, said: “I want to emphasize how important this trial is.”
She explained that current guidelines do not recommend any treatment for patients with device-detected AFib that is not shown on ECG, even though it is known this confers some excess risk of stroke.
“ARTESIA is the first well-powered, long-term trial looking at this question,” she said. “It found a clear reduction in the risk of stroke/systemic embolism with apixaban vs aspirin, but there was also a significant amount of bleeding – about an 80% increase. The question is whether the benefit on stroke is worth it given the bleeding risk.”
Dr. Albert highlighted the low absolute risk of stroke in this study population of around 1.2%, pointing out that even with the 37% relative reduction with anticoagulation, stroke is only reduced in absolute terms by 0.4%.
“We are going to have to take this back to committees and guidelines and look at the balance between the benefit on stroke and the increase in bleeding,” she concluded.
Noting that observational studies have shown that the duration of AFib impacts the risk of stroke, Dr. Albert suggested that patients with longer-duration AFib may benefit from anticoagulation to a greater extent; and given that the bleeding seen in ARTESIA was mainly GI bleeding, it might be possible to screen out patients at high risk of GI bleeding.
She also pointed out that a lot of patients discontinued anticoagulation treatment in both ARTESIA and NOAH-AFNET 6, showing that this is not an easy strategy for elderly patients.
In an editorial accompanying publication of the ARTESIA trial, Emma Svennberg, MD, Karolinska Institute, Stockholm, also concluded that, “going forward, we must balance the increased bleeding risks with the risk for disabling strokes,” and that “future substudies and meta-analyses may provide further insights regarding treatment benefits in specific subgroups.”
NOAH-AFNET 6: New subgroup analysis
The previously reported NOAH-AFNET 6 study randomly assigned 2,538 patients with subclinical AFib and additional risk factors for stroke to anticoagulation with edoxaban or placebo. The trial was stopped early, so it was underpowered – but it found no difference between groups in the incidence of the composite endpoint of stroke, systemic embolism, or death from cardiovascular causes or in the incidence of stroke, although there was higher risk of major bleeding.
Again, there was a low rate of stroke in this trial with just 49 strokes in total in the whole study. The NOAH-AFNET-6 investigators concluded that these patients should not receive anticoagulation because the risk of bleeding outweighed any potential benefits.
A new subanalysis of the 259 patients who had durations of subclinical AFib of 24 hours or longer in the NOAH-AFNET 6 trial was presented at the AHA meeting, and simultaneously published online in the European Heart Journal.
This showed that the rate of stroke also appeared low in patients with these long durations of subclinical AFib, and that there was no interaction between the duration of subclinical AFib and the efficacy and safety of oral anticoagulation.
But with such a low number of events in the study as a whole and in the long duration subclinical AFib subgroup (in which there were just two strokes in each treatment group), this analysis was unlikely to show a difference, Dr. Kirchhof commented.
The subgroup analysis did, however, show that patients experiencing subclinical AFib durations of 24 hours or more were more likely to develop clinical AFib over time than those with shorter durations, suggesting the need for regular ECGs in these patients.
Dr. Kirchhof said better methods are needed to detect patients with subclinical AFib at high risk of stroke. “I don’t think our clinical stroke risk factor scores such as CHA2DS2-VASc are sufficient to detect high-risk patients. Patients in both NOAH-AFNET 6 and ARTESIA had a median CHA2DS2-VASc score of 4, but they had a stroke rate of just 1% per year,” he noted.
The meta-analysis of the two trials showed that the results from both are consistent, with an overall reduction in ischemic stroke with oral anticoagulation (relative risk, 0.68). Oral anticoagulation also reduced a composite of cardiovascular death, all-cause stroke, peripheral arterial embolism, myocardial infarction, or pulmonary embolism (RR, 0.85).
There was no significant difference in cardiovascular death (RR, 0.95) or all-cause mortality (RR, 1.08), but anticoagulation significantly increased major bleeding (RR, 1.62).
Aspirin use complicates results
Dr. Healey said further analyses of the ARTESIA data will try to tease out the effect of concomitant aspirin use in the trial.
He explained that patients in this trial were allowed to take a single antiplatelet agent on top of study therapy.
“It is difficult to work out the exact use of antiplatelet therapy as it changed throughout the study,” he said. “About two-thirds were taking antiplatelet agents at the time of enrollment into the trial, but this decreased throughout the study. Many clinicians stopped open-label antiplatelet therapy during the trial when new evidence came out to suggest that there was no added benefit of adding aspirin on top of anticoagulants.
“We need to look carefully as to what impact that may have had,” Dr. Healey added. “We know from other studies that adding an antiplatelet on top of an anticoagulant doesn’t do much to thromboembolic events, but it approximately doubles the risk of major bleeding.”
In contrast, the NOAH-AFNET trial did not allow aspirin use in the anticoagulation group and aspirin was taken by around half the patients in the placebo group who had an indication for its use.
The authors of the meta-analysis pointed out that the omission of aspirin in nearly half of the control patients in NOAH-AFNET 6 and the early termination of the trial may have led to a slightly higher estimate for excess major bleeding with anticoagulation.
The ARTESIA study was supported by the Canadian Institutes for Health Research, the Bristol Myers Squibb-Pfizer Alliance, the Heart and Stroke Foundation of Canada, the Canadian Stroke Prevention Intervention Network, Hamilton Health Sciences, the Advancing Clinical Trials Network and the Population Health Research Institute. Dr. Healey reported research grants and speaking fees from BMS/Pfizer Alliance, Servier, Novartis, Boston Scientific, Medtronic; and acts as a consultant to Bayer, Servier and Boston Scientific. The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
in the ARTESIA study.
The results appear to contrast somewhat with the recently reported NOAH-AFNET 6 trial, which failed to show a reduction in stroke with the anticoagulant edoxaban versus placebo in a similar patient group, but that trial was stopped early and so was underpowered.
However, the lead investigators of both trials say the studies actually show consistent results – both found a lower rate of stroke than expected in this population, but the confidence intervals for stroke reduction with anticoagulation overlap, suggesting there is likely some effect, albeit less than that in clinical AFib.
The big question is whether the reduction in stroke with anticoagulation outweighs the increase in major bleeding.
A new meta-analysis of the two trials showed that “oral anticoagulation with edoxaban or apixaban reduces the risk of ischemic stroke by approximately one-third and increases major bleeding by roughly double.”
In absolute numbers, there were three fewer ischemic strokes per 1,000 patient-years with anticoagulation in the two trials combined, at the cost of seven more major bleeds.
The lead investigators of the two trials have somewhat different opinions on how these findings may translate into clinical practice.
Jeff Healey, MD, Population Health Research Institute, McMaster University, Hamilton, Ont., lead investigator of the ARTESIA trial, believes that the risks and benefits need to be assessed in individual patients, but there should be some patient groups that will benefit from anticoagulation treatment.
“In patients with pacemakers or implantable loop recorders with continuous monitoring, subclinical AF[ib] is detected in about one third of patients, so this is extremely common,” he said in an interview. “The question is whether this is just a normal feature of getting older or is this like AF[ib] that we see in the clinic which increases stroke risk, and I think we can conclude from ARTESIA that this subclinical AF[ib] is associated with an increased risk of stroke, although that is lower than the risk with clinical AF[ib], and that it can be reduced by anticoagulation.”
Until recently it hasn’t been possible to quantify the risk associated with subclinical AFib, he noted. “But now we have a rich dataset to use to see if we can tease out some specifics on this. Future analyses of this dataset will help define patients where the benefits outweigh the risks of bleeding. For now, I think we can look at the data in a qualitative way and consider the totality of risk factors in each patient – their bleeding risk, stroke risk, how much AF[ib] they have, and make a decision as to whether to give anticoagulation or not.”
But Paulus Kirchhof, MD, University Heart and Vascular Center Hamburg (Germany), lead investigator of the NOAH-AFNET 6 trial said: “Both trials showed the stroke rate is low in these patients – about 1% per year – and that anticoagulation can reduce it a bit further at the expense of increasing major bleeding. I don’t believe the AF[ib] episodes picked up on these devices constitute a sufficient stroke risk to warrant anticoagulation, given the bleeding risk.”
Dr. Kirchhof suggests an alternate approach of performing further traditional AFib monitoring on these patients.
“I think going forward in my practice, when we come across this device-detected AF[ib], we will do further investigations with an established method for detecting AF[ib] involving surface ECG monitoring – maybe a 3-day or 7-day Holter. If that shows AF[ib], then we will be on firm ground to start anticoagulation. If that doesn’t show AF[ib], we will probably not use anticoagulation.”
The ARTESIA trial and the meta-analysis of the two trials were both presented at the annual scientific sessions of the American Heart Association. Both studies were also simultaneously published online – ARTESIA in the New England Journal of Medicine and the meta-analysis in Circulation.
ARTESIA
For the ARTESIA study, 4012 patients with device-detected AFib and other clinical risk factors for stroke were randomly assigned to treatment with apixaban (5 mg twice daily) or aspirin (81 mg daily).
After a mean follow-up of 3.5 years, the primary endpoint – stroke or systemic embolism – occurred in 55 patients in the apixaban group (0.78% per patient-year), compared with 86 patients in the aspirin group (1.24% per patient-year), giving a hazard ratio of 0.63 (95% confidence interval, 0.45-0.88; P = .007).
“The risk of stroke or systemic embolism was lower by 37% with apixaban than with aspirin, and the risk of disabling or fatal stroke was lower by 49%,” Dr. Healey reported.
In the “on-treatment” population, the rate of major bleeding was 1.71% per patient-year in the apixaban group and 0.94% per patient-year in the aspirin group (HR, 1.80; 95% CI, 1.26-2.57; P = .001).
Fatal bleeding occurred in five patients in the apixaban group and eight patients in the aspirin group. Symptomatic intracranial hemorrhage occurred in 12 patients with apixaban and 15 patients with aspirin.
One of the main findings of the trial is the lower-than-expected risk of ischemic stroke in this population – about 1% per year in the aspirin group, which was reduced to 0.64% per year in the apixaban group.
The authors noted that “simply counting strokes as compared with bleeding events might suggest a neutral overall effect. With apixaban as compared with aspirin, 31 fewer cases of stroke or systemic embolism were seen in the intention-to-treat analysis, as compared with 39 more major bleeding events in the on-treatment analysis.”
However, they pointed out that strokes involve permanent loss of brain tissue, whereas major bleeding is usually reversible, with most patients having complete recovery, which was the case in this study.
“Thus, on the basis of the considerably greater severity of the stroke events prevented than the bleeding events caused, we believe that these findings favor consideration of the use of oral anticoagulation for patients with risk factors for stroke in whom subclinical atrial fibrillation develops,” they concluded.
First well-powered trial addressing this question
Discussing the ARTESIA trial at an AHA press conference, Christine Albert, MD, Cedars-Sinai Medical Center, Los Angeles, said: “I want to emphasize how important this trial is.”
She explained that current guidelines do not recommend any treatment for patients with device-detected AFib that is not shown on ECG, even though it is known this confers some excess risk of stroke.
“ARTESIA is the first well-powered, long-term trial looking at this question,” she said. “It found a clear reduction in the risk of stroke/systemic embolism with apixaban vs aspirin, but there was also a significant amount of bleeding – about an 80% increase. The question is whether the benefit on stroke is worth it given the bleeding risk.”
Dr. Albert highlighted the low absolute risk of stroke in this study population of around 1.2%, pointing out that even with the 37% relative reduction with anticoagulation, stroke is only reduced in absolute terms by 0.4%.
“We are going to have to take this back to committees and guidelines and look at the balance between the benefit on stroke and the increase in bleeding,” she concluded.
Noting that observational studies have shown that the duration of AFib impacts the risk of stroke, Dr. Albert suggested that patients with longer-duration AFib may benefit from anticoagulation to a greater extent; and given that the bleeding seen in ARTESIA was mainly GI bleeding, it might be possible to screen out patients at high risk of GI bleeding.
She also pointed out that a lot of patients discontinued anticoagulation treatment in both ARTESIA and NOAH-AFNET 6, showing that this is not an easy strategy for elderly patients.
In an editorial accompanying publication of the ARTESIA trial, Emma Svennberg, MD, Karolinska Institute, Stockholm, also concluded that, “going forward, we must balance the increased bleeding risks with the risk for disabling strokes,” and that “future substudies and meta-analyses may provide further insights regarding treatment benefits in specific subgroups.”
NOAH-AFNET 6: New subgroup analysis
The previously reported NOAH-AFNET 6 study randomly assigned 2,538 patients with subclinical AFib and additional risk factors for stroke to anticoagulation with edoxaban or placebo. The trial was stopped early, so it was underpowered – but it found no difference between groups in the incidence of the composite endpoint of stroke, systemic embolism, or death from cardiovascular causes or in the incidence of stroke, although there was higher risk of major bleeding.
Again, there was a low rate of stroke in this trial with just 49 strokes in total in the whole study. The NOAH-AFNET-6 investigators concluded that these patients should not receive anticoagulation because the risk of bleeding outweighed any potential benefits.
A new subanalysis of the 259 patients who had durations of subclinical AFib of 24 hours or longer in the NOAH-AFNET 6 trial was presented at the AHA meeting, and simultaneously published online in the European Heart Journal.
This showed that the rate of stroke also appeared low in patients with these long durations of subclinical AFib, and that there was no interaction between the duration of subclinical AFib and the efficacy and safety of oral anticoagulation.
But with such a low number of events in the study as a whole and in the long duration subclinical AFib subgroup (in which there were just two strokes in each treatment group), this analysis was unlikely to show a difference, Dr. Kirchhof commented.
The subgroup analysis did, however, show that patients experiencing subclinical AFib durations of 24 hours or more were more likely to develop clinical AFib over time than those with shorter durations, suggesting the need for regular ECGs in these patients.
Dr. Kirchhof said better methods are needed to detect patients with subclinical AFib at high risk of stroke. “I don’t think our clinical stroke risk factor scores such as CHA2DS2-VASc are sufficient to detect high-risk patients. Patients in both NOAH-AFNET 6 and ARTESIA had a median CHA2DS2-VASc score of 4, but they had a stroke rate of just 1% per year,” he noted.
The meta-analysis of the two trials showed that the results from both are consistent, with an overall reduction in ischemic stroke with oral anticoagulation (relative risk, 0.68). Oral anticoagulation also reduced a composite of cardiovascular death, all-cause stroke, peripheral arterial embolism, myocardial infarction, or pulmonary embolism (RR, 0.85).
There was no significant difference in cardiovascular death (RR, 0.95) or all-cause mortality (RR, 1.08), but anticoagulation significantly increased major bleeding (RR, 1.62).
Aspirin use complicates results
Dr. Healey said further analyses of the ARTESIA data will try to tease out the effect of concomitant aspirin use in the trial.
He explained that patients in this trial were allowed to take a single antiplatelet agent on top of study therapy.
“It is difficult to work out the exact use of antiplatelet therapy as it changed throughout the study,” he said. “About two-thirds were taking antiplatelet agents at the time of enrollment into the trial, but this decreased throughout the study. Many clinicians stopped open-label antiplatelet therapy during the trial when new evidence came out to suggest that there was no added benefit of adding aspirin on top of anticoagulants.
“We need to look carefully as to what impact that may have had,” Dr. Healey added. “We know from other studies that adding an antiplatelet on top of an anticoagulant doesn’t do much to thromboembolic events, but it approximately doubles the risk of major bleeding.”
In contrast, the NOAH-AFNET trial did not allow aspirin use in the anticoagulation group and aspirin was taken by around half the patients in the placebo group who had an indication for its use.
The authors of the meta-analysis pointed out that the omission of aspirin in nearly half of the control patients in NOAH-AFNET 6 and the early termination of the trial may have led to a slightly higher estimate for excess major bleeding with anticoagulation.
The ARTESIA study was supported by the Canadian Institutes for Health Research, the Bristol Myers Squibb-Pfizer Alliance, the Heart and Stroke Foundation of Canada, the Canadian Stroke Prevention Intervention Network, Hamilton Health Sciences, the Advancing Clinical Trials Network and the Population Health Research Institute. Dr. Healey reported research grants and speaking fees from BMS/Pfizer Alliance, Servier, Novartis, Boston Scientific, Medtronic; and acts as a consultant to Bayer, Servier and Boston Scientific. The NOAH-AFNET 6 trial was an investigator-initiated trial funded by the German Center for Cardiovascular Research and Daiichi Sankyo Europe. Dr. Kirchhof reported research support from several drug and device companies active in AFib. He is also listed as an inventor on two patents held by the University of Hamburg on AFib therapy and AFib markers.
A version of this article first appeared on Medscape.com.
FROM AHA 2023
Impressive bleeding profile with factor XI inhibitor in AFib: AZALEA
; the risk of stroke was moderate to high.
The trial was stopped earlier this year because of an “overwhelming” reduction in bleeding with abelacimab in comparison to rivaroxaban. Abelacimab is a monoclonal antibody given by subcutaneous injection once a month.
“Details of the bleeding results have now shown that the 150-mg dose of abelacimab, which is the dose being carried forward to phase 3 trials, was associated with a 67% reduction in major or clinically relevant nonmajor bleeding, the primary endpoint of the study.”
In addition, major bleeding was reduced by 74%, and major gastrointestinal bleeding was reduced by 93%.
“We are seeing really profound reductions in bleeding with this agent vs. a NOAC [novel oral anticoagulant],” lead AZALEA investigator Christian Ruff, MD, professor of medicine at Brigham and Women’s Hospital, Boston, said in an interview.
“Major bleeding – effectively the type of bleeding that results in hospitalization – is reduced by more than two-thirds, and major GI bleeding – which is the most common type of bleeding experienced by AF patients on anticoagulants – is almost eliminated. This gives us real hope that we have finally found an anticoagulant that is remarkably safe and will allow us to use anticoagulation in our most vulnerable patients,” he said.
Dr. Ruff presented the full results from the AZALEA trial at the American Heart Association scientific sessions.
He noted that AFib is one of the most common medical conditions in the world and that it confers an increased risk of stroke. Anticoagulants reduce this risk very effectively, and while the NOACS, such as apixaban and rivaroxaban, are safer than warfarin, significant bleeding still occurs, and “shockingly,” he said, between 30% and 60% of patients are not prescribed an anticoagulant or discontinue treatment because of bleeding concerns.
“Clearly, we need safer anticoagulants to protect these patients. Factor XI inhibitors, of which abelacimab is one, have emerged as the most promising agents, as they are thought to provide precision anticoagulation,” Dr. Ruff said.
He explained that factor XI appears to be involved in the formation of thrombus, which blocks arteries and causes strokes and myocardial infarction (thrombosis), but not in the healing process of blood vessels after injury (hemostasis). So, it is believed that inhibiting factor XI should reduce thrombotic events without causing excess bleeding.
AZALEA, which is the largest and longest trial of a factor XI inhibitor to date, enrolled 1,287 adults with AF who were at moderate to high risk of stroke.
They were randomly assigned to receive one of three treatments: oral rivaroxaban 20 mg daily; abelacimab 90 mg; or abelacimab 150 mg. Abelacimab was given monthly by injection.
Both doses of abelacimab inhibited factor XI almost completely; 97% inhibition was achieved with the 90-mg dose, and 99% inhibition was achieved with the 150-mg dose.
Results showed that after a median follow-up of 1.8 years, there was a clear reduction in all bleeding endpoints with both doses of abelacimab, compared with rivaroxaban.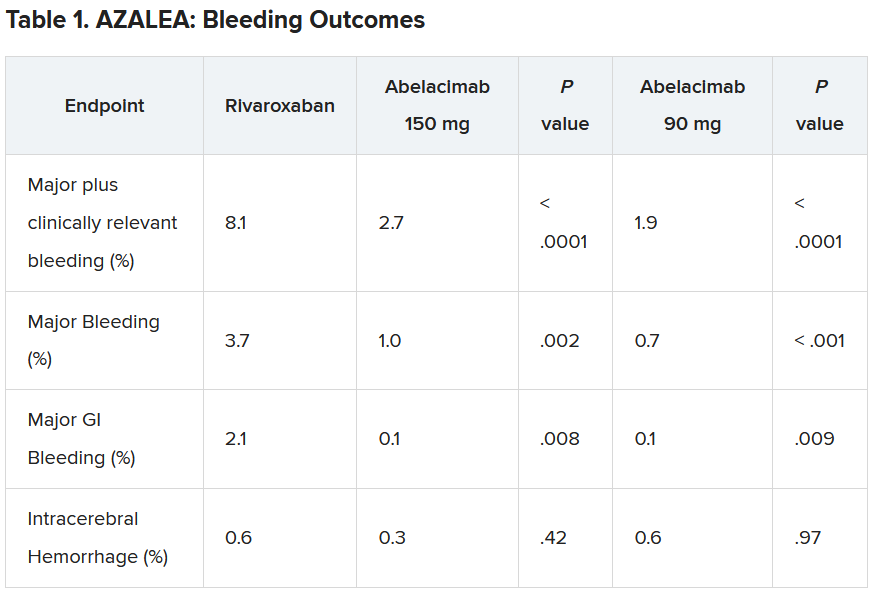
Dr. Ruff explained that the trial was powered to detect differences in bleeding, not stroke, but the investigators approached this in an exploratory way.
“As expected, the numbers were low, with just 25 strokes (23 ischemic strokes) across all three groups in the trial. So, because of this very low rate, we are really not able to compare how abelacimab compares with rivaroxaban in reducing stroke,” he commented.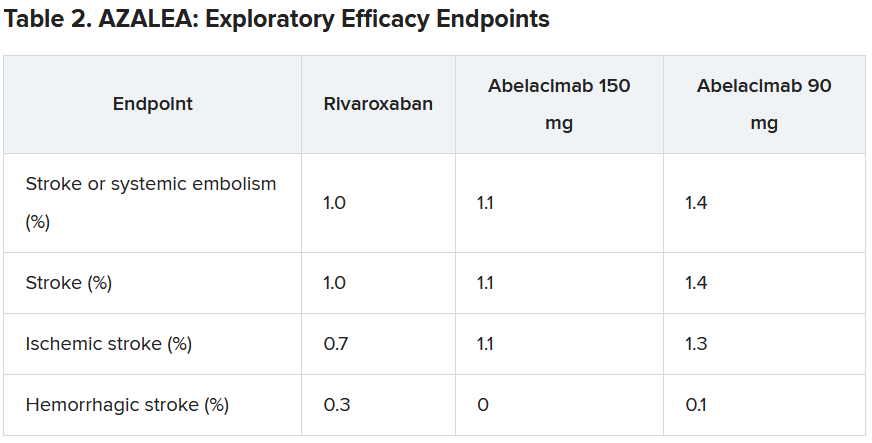
He did, however, suggest that the low stroke rate in the study was encouraging.
“If we look at the same population without anticoagulation, the stroke rate would be about 7% per year. And we see here in this trial that in all three arms, the stroke rate was just above 1% per year. I think this shows that all the patients in the trial were getting highly effective anticoagulation,” he said.
“But what this trial doesn’t answer – because the numbers are so low – is exactly how effective factor XI inhibition with abelacimab is, compared to NOACs in reducing stroke rates. That requires dedicated phase 3 trials.”
Dr. Ruff pointed out that there are some reassuring data from phase 2 trials in venous thromboembolism (VTE), in which the 150-mg dose of abelacimab was associated with an 80% reduction in VTE, compared with enoxaparin. “Historically in the development of anticoagulants, efficacy in VTE has translated into efficacy in stroke prevention, so that is very encouraging,” he commented.
“So, I think our results along with the VTE results are encouraging, but the precision regarding the relative efficacy compared to NOACs is still an open question that needs to be clarified in phase 3 trials,” he concluded.
Several phase 3 trials are now underway with abelacimab and two other small-molecule orally available factor XI inhibitors, milvexian (BMS/Janssen) and asundexian (Bayer).
The designated discussant of the AZALEA study at the AHA meeting, Manesh Patel. MD, Duke University, Durham, N.C., described the results as “an important step forward.”
“This trial, with the prior data in this field, show that factor XI inhibition as a target is biologically possible (studies showing > 95% inhibition), significantly less bleeding than NOACS. We await the phase 3 studies, but having significantly less bleeding and similar or less stroke would be a substantial step forward for the field,” he said.
John Alexander, MD, also from Duke University, said: “There were clinically important reductions in bleeding with both doses of abelacimab, compared with rivaroxaban. This is consistent to what we’ve seen with comparisons between other factor XI inhibitors and other factor Xa inhibitors.”
On the exploratory efficacy results, Dr. Alexander agreed with Dr. Ruff that it was not possible to get any idea of how abelacimab compared with rivaroxaban in reducing stroke. “The hazard ratio and confidence intervals comparing abelacimab and rivaroxaban include substantial lower rates, no difference, and substantially higher rates,” he noted.
“We need to wait for the results of phase 3 trials, with abelacimab and other factor XI inhibitors, to understand how well factor XI inhibition prevents stroke and systemic embolism in patients with atrial fibrillation,” Dr. Alexander added. “These trials are ongoing.”
Dr. Ruff concluded: “Assuming the data from ongoing phase 3 trials confirm the benefit of factor XI inhibitors for stroke prevention in people with AF, it will really be transformative for the field of cardiology.
“Our first mission in treating people with AF is to prevent stroke, and our ability to do this with a remarkably safe anticoagulant such as abelacimab would be an incredible advance,” he concluded.
Dr. Ruff receives research funding from Anthos for abelacimab trials, is on an AF executive committee for BMS/Janssen (milvexian), and has been on an advisory board for Bayer (asundexian). Dr. Patel has received grants from and acts as an advisor to Bayer and Janssen. Dr. Alexander receives research funding from Bayer.
A version of this article first appeared on Medscape.com.
; the risk of stroke was moderate to high.
The trial was stopped earlier this year because of an “overwhelming” reduction in bleeding with abelacimab in comparison to rivaroxaban. Abelacimab is a monoclonal antibody given by subcutaneous injection once a month.
“Details of the bleeding results have now shown that the 150-mg dose of abelacimab, which is the dose being carried forward to phase 3 trials, was associated with a 67% reduction in major or clinically relevant nonmajor bleeding, the primary endpoint of the study.”
In addition, major bleeding was reduced by 74%, and major gastrointestinal bleeding was reduced by 93%.
“We are seeing really profound reductions in bleeding with this agent vs. a NOAC [novel oral anticoagulant],” lead AZALEA investigator Christian Ruff, MD, professor of medicine at Brigham and Women’s Hospital, Boston, said in an interview.
“Major bleeding – effectively the type of bleeding that results in hospitalization – is reduced by more than two-thirds, and major GI bleeding – which is the most common type of bleeding experienced by AF patients on anticoagulants – is almost eliminated. This gives us real hope that we have finally found an anticoagulant that is remarkably safe and will allow us to use anticoagulation in our most vulnerable patients,” he said.
Dr. Ruff presented the full results from the AZALEA trial at the American Heart Association scientific sessions.
He noted that AFib is one of the most common medical conditions in the world and that it confers an increased risk of stroke. Anticoagulants reduce this risk very effectively, and while the NOACS, such as apixaban and rivaroxaban, are safer than warfarin, significant bleeding still occurs, and “shockingly,” he said, between 30% and 60% of patients are not prescribed an anticoagulant or discontinue treatment because of bleeding concerns.
“Clearly, we need safer anticoagulants to protect these patients. Factor XI inhibitors, of which abelacimab is one, have emerged as the most promising agents, as they are thought to provide precision anticoagulation,” Dr. Ruff said.
He explained that factor XI appears to be involved in the formation of thrombus, which blocks arteries and causes strokes and myocardial infarction (thrombosis), but not in the healing process of blood vessels after injury (hemostasis). So, it is believed that inhibiting factor XI should reduce thrombotic events without causing excess bleeding.
AZALEA, which is the largest and longest trial of a factor XI inhibitor to date, enrolled 1,287 adults with AF who were at moderate to high risk of stroke.
They were randomly assigned to receive one of three treatments: oral rivaroxaban 20 mg daily; abelacimab 90 mg; or abelacimab 150 mg. Abelacimab was given monthly by injection.
Both doses of abelacimab inhibited factor XI almost completely; 97% inhibition was achieved with the 90-mg dose, and 99% inhibition was achieved with the 150-mg dose.
Results showed that after a median follow-up of 1.8 years, there was a clear reduction in all bleeding endpoints with both doses of abelacimab, compared with rivaroxaban.
Dr. Ruff explained that the trial was powered to detect differences in bleeding, not stroke, but the investigators approached this in an exploratory way.
“As expected, the numbers were low, with just 25 strokes (23 ischemic strokes) across all three groups in the trial. So, because of this very low rate, we are really not able to compare how abelacimab compares with rivaroxaban in reducing stroke,” he commented.
He did, however, suggest that the low stroke rate in the study was encouraging.
“If we look at the same population without anticoagulation, the stroke rate would be about 7% per year. And we see here in this trial that in all three arms, the stroke rate was just above 1% per year. I think this shows that all the patients in the trial were getting highly effective anticoagulation,” he said.
“But what this trial doesn’t answer – because the numbers are so low – is exactly how effective factor XI inhibition with abelacimab is, compared to NOACs in reducing stroke rates. That requires dedicated phase 3 trials.”
Dr. Ruff pointed out that there are some reassuring data from phase 2 trials in venous thromboembolism (VTE), in which the 150-mg dose of abelacimab was associated with an 80% reduction in VTE, compared with enoxaparin. “Historically in the development of anticoagulants, efficacy in VTE has translated into efficacy in stroke prevention, so that is very encouraging,” he commented.
“So, I think our results along with the VTE results are encouraging, but the precision regarding the relative efficacy compared to NOACs is still an open question that needs to be clarified in phase 3 trials,” he concluded.
Several phase 3 trials are now underway with abelacimab and two other small-molecule orally available factor XI inhibitors, milvexian (BMS/Janssen) and asundexian (Bayer).
The designated discussant of the AZALEA study at the AHA meeting, Manesh Patel. MD, Duke University, Durham, N.C., described the results as “an important step forward.”
“This trial, with the prior data in this field, show that factor XI inhibition as a target is biologically possible (studies showing > 95% inhibition), significantly less bleeding than NOACS. We await the phase 3 studies, but having significantly less bleeding and similar or less stroke would be a substantial step forward for the field,” he said.
John Alexander, MD, also from Duke University, said: “There were clinically important reductions in bleeding with both doses of abelacimab, compared with rivaroxaban. This is consistent to what we’ve seen with comparisons between other factor XI inhibitors and other factor Xa inhibitors.”
On the exploratory efficacy results, Dr. Alexander agreed with Dr. Ruff that it was not possible to get any idea of how abelacimab compared with rivaroxaban in reducing stroke. “The hazard ratio and confidence intervals comparing abelacimab and rivaroxaban include substantial lower rates, no difference, and substantially higher rates,” he noted.
“We need to wait for the results of phase 3 trials, with abelacimab and other factor XI inhibitors, to understand how well factor XI inhibition prevents stroke and systemic embolism in patients with atrial fibrillation,” Dr. Alexander added. “These trials are ongoing.”
Dr. Ruff concluded: “Assuming the data from ongoing phase 3 trials confirm the benefit of factor XI inhibitors for stroke prevention in people with AF, it will really be transformative for the field of cardiology.
“Our first mission in treating people with AF is to prevent stroke, and our ability to do this with a remarkably safe anticoagulant such as abelacimab would be an incredible advance,” he concluded.
Dr. Ruff receives research funding from Anthos for abelacimab trials, is on an AF executive committee for BMS/Janssen (milvexian), and has been on an advisory board for Bayer (asundexian). Dr. Patel has received grants from and acts as an advisor to Bayer and Janssen. Dr. Alexander receives research funding from Bayer.
A version of this article first appeared on Medscape.com.
; the risk of stroke was moderate to high.
The trial was stopped earlier this year because of an “overwhelming” reduction in bleeding with abelacimab in comparison to rivaroxaban. Abelacimab is a monoclonal antibody given by subcutaneous injection once a month.
“Details of the bleeding results have now shown that the 150-mg dose of abelacimab, which is the dose being carried forward to phase 3 trials, was associated with a 67% reduction in major or clinically relevant nonmajor bleeding, the primary endpoint of the study.”
In addition, major bleeding was reduced by 74%, and major gastrointestinal bleeding was reduced by 93%.
“We are seeing really profound reductions in bleeding with this agent vs. a NOAC [novel oral anticoagulant],” lead AZALEA investigator Christian Ruff, MD, professor of medicine at Brigham and Women’s Hospital, Boston, said in an interview.
“Major bleeding – effectively the type of bleeding that results in hospitalization – is reduced by more than two-thirds, and major GI bleeding – which is the most common type of bleeding experienced by AF patients on anticoagulants – is almost eliminated. This gives us real hope that we have finally found an anticoagulant that is remarkably safe and will allow us to use anticoagulation in our most vulnerable patients,” he said.
Dr. Ruff presented the full results from the AZALEA trial at the American Heart Association scientific sessions.
He noted that AFib is one of the most common medical conditions in the world and that it confers an increased risk of stroke. Anticoagulants reduce this risk very effectively, and while the NOACS, such as apixaban and rivaroxaban, are safer than warfarin, significant bleeding still occurs, and “shockingly,” he said, between 30% and 60% of patients are not prescribed an anticoagulant or discontinue treatment because of bleeding concerns.
“Clearly, we need safer anticoagulants to protect these patients. Factor XI inhibitors, of which abelacimab is one, have emerged as the most promising agents, as they are thought to provide precision anticoagulation,” Dr. Ruff said.
He explained that factor XI appears to be involved in the formation of thrombus, which blocks arteries and causes strokes and myocardial infarction (thrombosis), but not in the healing process of blood vessels after injury (hemostasis). So, it is believed that inhibiting factor XI should reduce thrombotic events without causing excess bleeding.
AZALEA, which is the largest and longest trial of a factor XI inhibitor to date, enrolled 1,287 adults with AF who were at moderate to high risk of stroke.
They were randomly assigned to receive one of three treatments: oral rivaroxaban 20 mg daily; abelacimab 90 mg; or abelacimab 150 mg. Abelacimab was given monthly by injection.
Both doses of abelacimab inhibited factor XI almost completely; 97% inhibition was achieved with the 90-mg dose, and 99% inhibition was achieved with the 150-mg dose.
Results showed that after a median follow-up of 1.8 years, there was a clear reduction in all bleeding endpoints with both doses of abelacimab, compared with rivaroxaban.
Dr. Ruff explained that the trial was powered to detect differences in bleeding, not stroke, but the investigators approached this in an exploratory way.
“As expected, the numbers were low, with just 25 strokes (23 ischemic strokes) across all three groups in the trial. So, because of this very low rate, we are really not able to compare how abelacimab compares with rivaroxaban in reducing stroke,” he commented.
He did, however, suggest that the low stroke rate in the study was encouraging.
“If we look at the same population without anticoagulation, the stroke rate would be about 7% per year. And we see here in this trial that in all three arms, the stroke rate was just above 1% per year. I think this shows that all the patients in the trial were getting highly effective anticoagulation,” he said.
“But what this trial doesn’t answer – because the numbers are so low – is exactly how effective factor XI inhibition with abelacimab is, compared to NOACs in reducing stroke rates. That requires dedicated phase 3 trials.”
Dr. Ruff pointed out that there are some reassuring data from phase 2 trials in venous thromboembolism (VTE), in which the 150-mg dose of abelacimab was associated with an 80% reduction in VTE, compared with enoxaparin. “Historically in the development of anticoagulants, efficacy in VTE has translated into efficacy in stroke prevention, so that is very encouraging,” he commented.
“So, I think our results along with the VTE results are encouraging, but the precision regarding the relative efficacy compared to NOACs is still an open question that needs to be clarified in phase 3 trials,” he concluded.
Several phase 3 trials are now underway with abelacimab and two other small-molecule orally available factor XI inhibitors, milvexian (BMS/Janssen) and asundexian (Bayer).
The designated discussant of the AZALEA study at the AHA meeting, Manesh Patel. MD, Duke University, Durham, N.C., described the results as “an important step forward.”
“This trial, with the prior data in this field, show that factor XI inhibition as a target is biologically possible (studies showing > 95% inhibition), significantly less bleeding than NOACS. We await the phase 3 studies, but having significantly less bleeding and similar or less stroke would be a substantial step forward for the field,” he said.
John Alexander, MD, also from Duke University, said: “There were clinically important reductions in bleeding with both doses of abelacimab, compared with rivaroxaban. This is consistent to what we’ve seen with comparisons between other factor XI inhibitors and other factor Xa inhibitors.”
On the exploratory efficacy results, Dr. Alexander agreed with Dr. Ruff that it was not possible to get any idea of how abelacimab compared with rivaroxaban in reducing stroke. “The hazard ratio and confidence intervals comparing abelacimab and rivaroxaban include substantial lower rates, no difference, and substantially higher rates,” he noted.
“We need to wait for the results of phase 3 trials, with abelacimab and other factor XI inhibitors, to understand how well factor XI inhibition prevents stroke and systemic embolism in patients with atrial fibrillation,” Dr. Alexander added. “These trials are ongoing.”
Dr. Ruff concluded: “Assuming the data from ongoing phase 3 trials confirm the benefit of factor XI inhibitors for stroke prevention in people with AF, it will really be transformative for the field of cardiology.
“Our first mission in treating people with AF is to prevent stroke, and our ability to do this with a remarkably safe anticoagulant such as abelacimab would be an incredible advance,” he concluded.
Dr. Ruff receives research funding from Anthos for abelacimab trials, is on an AF executive committee for BMS/Janssen (milvexian), and has been on an advisory board for Bayer (asundexian). Dr. Patel has received grants from and acts as an advisor to Bayer and Janssen. Dr. Alexander receives research funding from Bayer.
A version of this article first appeared on Medscape.com.
FROM AHA 2023
Atrial fibrillation linked to dementia, especially when diagnosed before age 65 years
TOPLINE:
Adults with atrial fibrillation (AFib) are at increased risk for dementia, especially when AFib occurs before age 65 years, new research shows. Investigators note the findings highlight the importance of monitoring cognitive function in adults with AF.
METHODOLOGY:
- This prospective, population-based cohort study leveraged data from 433,746 UK Biobank participants (55% women), including 30,601 with AFib, who were followed for a median of 12.6 years
- Incident cases of dementia were determined through linkage from multiple databases.
- Cox proportional hazards models and propensity score matching were used to estimate the association between age at onset of AFib and incident dementia.
TAKEAWAY:
- During follow-up, new-onset dementia occurred in 5,898 participants (2,546 with Alzheimer’s disease [AD] and 1,211 with vascular dementia [VD]), of which, 1,031 had AFib (350 with AD; 320 with VD).
- Compared with participants without AFib, those with AFib had a 42% higher risk for all-cause dementia (adjusted hazard ratio, 1.42; P < .001) and more than double the risk for VD (aHR, 2.06; P < .001), but no significantly higher risk for AD.
- Younger age at AFib onset was associated with higher risks for all-cause dementia, AD and VD, with aHRs per 10-year decrease of 1.23, 1.27, and 1.35, respectively (P < .001 for all).
- After propensity score matching, AFib onset before age 65 years had the highest risk for all-cause dementia (aHR, 1.82; P < .001), followed by AF onset at age 65-74 years (aHR, 1.47; P < .001). Similar results were seen in AD and VD.
IN PRACTICE:
“The findings indicate that careful monitoring of cognitive function for patients with a younger [AFib] onset age, particularly those diagnosed with [AFib] before age 65 years, is important to attenuate the risk of subsequent dementia,” the authors write.
SOURCE:
The study, with first author Wenya Zhang, with the Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, was published online in JAMA Network Open.
LIMITATIONS:
Because the study was observational, a cause-effect relationship cannot be established. Despite the adjustment for many underlying confounders, residual unidentified confounders may still exist. The vast majority of participants were White. The analyses did not consider the potential impact of effective treatment of AFib on dementia risk.
DISCLOSURES:
The study had no commercial funding. The authors have declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Adults with atrial fibrillation (AFib) are at increased risk for dementia, especially when AFib occurs before age 65 years, new research shows. Investigators note the findings highlight the importance of monitoring cognitive function in adults with AF.
METHODOLOGY:
- This prospective, population-based cohort study leveraged data from 433,746 UK Biobank participants (55% women), including 30,601 with AFib, who were followed for a median of 12.6 years
- Incident cases of dementia were determined through linkage from multiple databases.
- Cox proportional hazards models and propensity score matching were used to estimate the association between age at onset of AFib and incident dementia.
TAKEAWAY:
- During follow-up, new-onset dementia occurred in 5,898 participants (2,546 with Alzheimer’s disease [AD] and 1,211 with vascular dementia [VD]), of which, 1,031 had AFib (350 with AD; 320 with VD).
- Compared with participants without AFib, those with AFib had a 42% higher risk for all-cause dementia (adjusted hazard ratio, 1.42; P < .001) and more than double the risk for VD (aHR, 2.06; P < .001), but no significantly higher risk for AD.
- Younger age at AFib onset was associated with higher risks for all-cause dementia, AD and VD, with aHRs per 10-year decrease of 1.23, 1.27, and 1.35, respectively (P < .001 for all).
- After propensity score matching, AFib onset before age 65 years had the highest risk for all-cause dementia (aHR, 1.82; P < .001), followed by AF onset at age 65-74 years (aHR, 1.47; P < .001). Similar results were seen in AD and VD.
IN PRACTICE:
“The findings indicate that careful monitoring of cognitive function for patients with a younger [AFib] onset age, particularly those diagnosed with [AFib] before age 65 years, is important to attenuate the risk of subsequent dementia,” the authors write.
SOURCE:
The study, with first author Wenya Zhang, with the Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, was published online in JAMA Network Open.
LIMITATIONS:
Because the study was observational, a cause-effect relationship cannot be established. Despite the adjustment for many underlying confounders, residual unidentified confounders may still exist. The vast majority of participants were White. The analyses did not consider the potential impact of effective treatment of AFib on dementia risk.
DISCLOSURES:
The study had no commercial funding. The authors have declared no conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Adults with atrial fibrillation (AFib) are at increased risk for dementia, especially when AFib occurs before age 65 years, new research shows. Investigators note the findings highlight the importance of monitoring cognitive function in adults with AF.
METHODOLOGY:
- This prospective, population-based cohort study leveraged data from 433,746 UK Biobank participants (55% women), including 30,601 with AFib, who were followed for a median of 12.6 years
- Incident cases of dementia were determined through linkage from multiple databases.
- Cox proportional hazards models and propensity score matching were used to estimate the association between age at onset of AFib and incident dementia.
TAKEAWAY:
- During follow-up, new-onset dementia occurred in 5,898 participants (2,546 with Alzheimer’s disease [AD] and 1,211 with vascular dementia [VD]), of which, 1,031 had AFib (350 with AD; 320 with VD).
- Compared with participants without AFib, those with AFib had a 42% higher risk for all-cause dementia (adjusted hazard ratio, 1.42; P < .001) and more than double the risk for VD (aHR, 2.06; P < .001), but no significantly higher risk for AD.
- Younger age at AFib onset was associated with higher risks for all-cause dementia, AD and VD, with aHRs per 10-year decrease of 1.23, 1.27, and 1.35, respectively (P < .001 for all).
- After propensity score matching, AFib onset before age 65 years had the highest risk for all-cause dementia (aHR, 1.82; P < .001), followed by AF onset at age 65-74 years (aHR, 1.47; P < .001). Similar results were seen in AD and VD.
IN PRACTICE:
“The findings indicate that careful monitoring of cognitive function for patients with a younger [AFib] onset age, particularly those diagnosed with [AFib] before age 65 years, is important to attenuate the risk of subsequent dementia,” the authors write.
SOURCE:
The study, with first author Wenya Zhang, with the Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, was published online in JAMA Network Open.
LIMITATIONS:
Because the study was observational, a cause-effect relationship cannot be established. Despite the adjustment for many underlying confounders, residual unidentified confounders may still exist. The vast majority of participants were White. The analyses did not consider the potential impact of effective treatment of AFib on dementia risk.
DISCLOSURES:
The study had no commercial funding. The authors have declared no conflicts of interest.
A version of this article appeared on Medscape.com.
Blood pressure lowering reduces dementia risk
Results of a trial using an intensive, 4-year program aimed at blood pressure lowering showed that intervention reduced not only blood pressure, but also significantly reduced the risk of total dementia over that period.
and cognitive impairment no dementia (CIND), a secondary outcome, was also significantly reduced by 16%.
“Blood pressure reduction is effective in reducing the risk of dementia in patients with hypertension,” concluded Jiang He, MD, PhD, professor of epidemiology and medicine and director of Tulane University’s Translational Science Institute, New Orleans. “This proven, effective intervention should be widely scaled up to reduce the global burden of dementia.”
He presented these results from the China Rural Hypertension Control Project (CRHCP) at the annual scientific sessions of the American Heart Association.
Target organ damage
Keith Ferdinand, MD, also from Tulane University, commented on the findings during a press conference at the meeting, noting that the result “opens our opportunity to recognize that the target organ damage of hypertension also now includes dementia.”
The researchers were able to “rigorously lower blood pressure from 157 to 127.6 in the intervention, 155 to 147 in the controls – 22 mg Hg – and if you look at the P values for all the various outcomes, they were very robust,” Dr. Ferdinand said.
Another interesting feature about the strategy used in this trial is that “this was true team-based care,” he pointed out. The trained interventionists in the study, called village doctors, collaborated with primary care physicians and initiated medications. “They stayed on a simple treatment protocol, and they were able to assist patients to ensure they had free medications, health coaching for lifestyle, home blood pressure measurement, and ensuring adherence.”
So, Dr. Ferdinand added, “one of the questions is whether this is a model we can use in other places around the globe, in places with low resources, and in the United States in disadvantaged populations.”
Public health priority
It’s estimated that the global number of those living with dementia will increase from 57.4 million in 2019 to 152.8 million by 2050, Dr. He said. “In the absence of curative treatment, the primary prevention of dementia through risk factor reduction, such as blood pressure lowering, becomes a public health priority.”
Previous randomized trials have lacked sample size and duration but have reported a nonsignificant reduction in dementia associated with antihypertensive treatment in patients with hypertension or a history of stroke, Dr. He noted.
This new trial aimed to test the effectiveness of intensive BP intervention to reduce the risk of all-cause dementia and cognitive impairment over a 48-month intervention period versus usual care.
It was an open-label, blinded-endpoint, cluster-randomized trial, and included 33,995 individual patients from 325 villages in China, aged 40 years and older, with untreated hypertension. The villages were randomly assigned to an intervention group or usual care, stratified by province, county, and township.
Patients were eligible if they had mean untreated systolic BP greater than 140 mm Hg and/or diastolic BP greater than 90 mm Hg or mean treated systolic BP of greater than 130 and/or diastolic greater than 80 mm Hg. Patients with a history of cardiovascular disease, chronic kidney disease, or diabetes and a mean systolic BP greater than 130 mm Hg and/or diastolic BP greater than 80 mm Hg from six measures on two different days were also eligible.
All were enrolled in the China New Rural Cooperative Medical Scheme, which covers 99% of rural residents for health care services, Dr. He noted.
The intervention was a simple stepped-care protocol for hypertension treatment, aimed at achieving a target systolic BP of less than 130 mm Hg and diastolic of less than 80 mm Hg.
Village doctors started and titrated antihypertensive treatment based on a protocol and were able to deliver discounted and free medications to patients. They also did health coaching on lifestyle modification and adherence to medication, and instructed patients on home BP monitoring.
Patients were provided training, supervision, and consultation by primary care physicians and hypertension specialists.
At the month 48 follow-up visit, the participants were assessed by neurologists who were blinded to randomization assignments. Neurologists did a variety of tests and assessments including collecting data on the patient’s medical and psychiatric history and risk factors for dementia, as well as neurologic assessment using the Mini-Mental State Examination, the Functional Activities Questionnaire, and the Quick Dementia Rating System.
The primary outcome was all-cause dementia, defined according to recommendations from the National Institute on Aging–Alzheimer’s Association work groups on diagnostic guidelines for Alzheimer’s disease.
Secondary outcomes included CIND, a composite outcome of dementia or CIND, and a composite of dementia or deaths.
The final diagnosis of all-cause dementia or CIND was made by an expert adjudication panel blinded to the intervention assignment.
At 48 months, 91.3% of patients completed the follow-up for clinical outcomes. Participants were an average of 63 years of age, 61% were female, and 23% had less than a primary school education, Dr. He noted.
The net group differences in systolic and diastolic BP reduction were 22 and 9.3 mm Hg, respectively (P < .0001).
Significant differences were also seen between the groups in the primary outcome of all-cause dementia, as well as secondary outcomes of CIND, dementia or cognitive impairment, or dementia or deaths.
Serious adverse events were more common in the usual care group, and there was no difference between groups in the occurrence of falls or syncope.
The effect was consistent across subgroups, Dr. He said, including age, sex, education, cigarette smoking, body mass index, systolic BP, and fasting plasma glucose at baseline.
First definitive evidence
Invited discussant for the trial, Daniel W. Jones, MD, University of Mississippi Medical Center, Jackson, and past president of the AHA, pointed out that previous results from CRHCP on cardiovascular outcomes, reported earlier in 2023 in The Lancet, showed that, similar to results of the large SPRINT trial, lowering systolic BP to a goal of less than 130 mm Hg reduced a composite endpoint of MI, stroke, heart failure requiring hospitalization, and cardiovascular disease death over the 36-month follow-up.
The SPRINT findings also suggested a possible reduction in dementia, Dr. Jones said.
Now, in these new CRHCP results, “there was a clear benefit for intensive BP control in reducing risk for dementia and cognitive dysfunction,” he said. “This is, importantly, the first definitive evidence of dementia risk reduction demonstrated in a randomized controlled clinical trial. This outcome supports observational data that shows a strong relationship between BP and dementia.”
Since it is the first of its kind though, replication of the results will be important, he noted.
The study also showed that the intervention, using minimally trained village doctors, sustained BP control for 48 months. “This model could be used in any setting with modifications, including in the United States,” Dr. Jones said.
The study was supported by the Ministry of Science and Technology of China; U.S. investigators did not receive financial support from this study. The researchers and Dr. Jones disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Results of a trial using an intensive, 4-year program aimed at blood pressure lowering showed that intervention reduced not only blood pressure, but also significantly reduced the risk of total dementia over that period.
and cognitive impairment no dementia (CIND), a secondary outcome, was also significantly reduced by 16%.
“Blood pressure reduction is effective in reducing the risk of dementia in patients with hypertension,” concluded Jiang He, MD, PhD, professor of epidemiology and medicine and director of Tulane University’s Translational Science Institute, New Orleans. “This proven, effective intervention should be widely scaled up to reduce the global burden of dementia.”
He presented these results from the China Rural Hypertension Control Project (CRHCP) at the annual scientific sessions of the American Heart Association.
Target organ damage
Keith Ferdinand, MD, also from Tulane University, commented on the findings during a press conference at the meeting, noting that the result “opens our opportunity to recognize that the target organ damage of hypertension also now includes dementia.”
The researchers were able to “rigorously lower blood pressure from 157 to 127.6 in the intervention, 155 to 147 in the controls – 22 mg Hg – and if you look at the P values for all the various outcomes, they were very robust,” Dr. Ferdinand said.
Another interesting feature about the strategy used in this trial is that “this was true team-based care,” he pointed out. The trained interventionists in the study, called village doctors, collaborated with primary care physicians and initiated medications. “They stayed on a simple treatment protocol, and they were able to assist patients to ensure they had free medications, health coaching for lifestyle, home blood pressure measurement, and ensuring adherence.”
So, Dr. Ferdinand added, “one of the questions is whether this is a model we can use in other places around the globe, in places with low resources, and in the United States in disadvantaged populations.”
Public health priority
It’s estimated that the global number of those living with dementia will increase from 57.4 million in 2019 to 152.8 million by 2050, Dr. He said. “In the absence of curative treatment, the primary prevention of dementia through risk factor reduction, such as blood pressure lowering, becomes a public health priority.”
Previous randomized trials have lacked sample size and duration but have reported a nonsignificant reduction in dementia associated with antihypertensive treatment in patients with hypertension or a history of stroke, Dr. He noted.
This new trial aimed to test the effectiveness of intensive BP intervention to reduce the risk of all-cause dementia and cognitive impairment over a 48-month intervention period versus usual care.
It was an open-label, blinded-endpoint, cluster-randomized trial, and included 33,995 individual patients from 325 villages in China, aged 40 years and older, with untreated hypertension. The villages were randomly assigned to an intervention group or usual care, stratified by province, county, and township.
Patients were eligible if they had mean untreated systolic BP greater than 140 mm Hg and/or diastolic BP greater than 90 mm Hg or mean treated systolic BP of greater than 130 and/or diastolic greater than 80 mm Hg. Patients with a history of cardiovascular disease, chronic kidney disease, or diabetes and a mean systolic BP greater than 130 mm Hg and/or diastolic BP greater than 80 mm Hg from six measures on two different days were also eligible.
All were enrolled in the China New Rural Cooperative Medical Scheme, which covers 99% of rural residents for health care services, Dr. He noted.
The intervention was a simple stepped-care protocol for hypertension treatment, aimed at achieving a target systolic BP of less than 130 mm Hg and diastolic of less than 80 mm Hg.
Village doctors started and titrated antihypertensive treatment based on a protocol and were able to deliver discounted and free medications to patients. They also did health coaching on lifestyle modification and adherence to medication, and instructed patients on home BP monitoring.
Patients were provided training, supervision, and consultation by primary care physicians and hypertension specialists.
At the month 48 follow-up visit, the participants were assessed by neurologists who were blinded to randomization assignments. Neurologists did a variety of tests and assessments including collecting data on the patient’s medical and psychiatric history and risk factors for dementia, as well as neurologic assessment using the Mini-Mental State Examination, the Functional Activities Questionnaire, and the Quick Dementia Rating System.
The primary outcome was all-cause dementia, defined according to recommendations from the National Institute on Aging–Alzheimer’s Association work groups on diagnostic guidelines for Alzheimer’s disease.
Secondary outcomes included CIND, a composite outcome of dementia or CIND, and a composite of dementia or deaths.
The final diagnosis of all-cause dementia or CIND was made by an expert adjudication panel blinded to the intervention assignment.
At 48 months, 91.3% of patients completed the follow-up for clinical outcomes. Participants were an average of 63 years of age, 61% were female, and 23% had less than a primary school education, Dr. He noted.
The net group differences in systolic and diastolic BP reduction were 22 and 9.3 mm Hg, respectively (P < .0001).
Significant differences were also seen between the groups in the primary outcome of all-cause dementia, as well as secondary outcomes of CIND, dementia or cognitive impairment, or dementia or deaths.
Serious adverse events were more common in the usual care group, and there was no difference between groups in the occurrence of falls or syncope.
The effect was consistent across subgroups, Dr. He said, including age, sex, education, cigarette smoking, body mass index, systolic BP, and fasting plasma glucose at baseline.
First definitive evidence
Invited discussant for the trial, Daniel W. Jones, MD, University of Mississippi Medical Center, Jackson, and past president of the AHA, pointed out that previous results from CRHCP on cardiovascular outcomes, reported earlier in 2023 in The Lancet, showed that, similar to results of the large SPRINT trial, lowering systolic BP to a goal of less than 130 mm Hg reduced a composite endpoint of MI, stroke, heart failure requiring hospitalization, and cardiovascular disease death over the 36-month follow-up.
The SPRINT findings also suggested a possible reduction in dementia, Dr. Jones said.
Now, in these new CRHCP results, “there was a clear benefit for intensive BP control in reducing risk for dementia and cognitive dysfunction,” he said. “This is, importantly, the first definitive evidence of dementia risk reduction demonstrated in a randomized controlled clinical trial. This outcome supports observational data that shows a strong relationship between BP and dementia.”
Since it is the first of its kind though, replication of the results will be important, he noted.
The study also showed that the intervention, using minimally trained village doctors, sustained BP control for 48 months. “This model could be used in any setting with modifications, including in the United States,” Dr. Jones said.
The study was supported by the Ministry of Science and Technology of China; U.S. investigators did not receive financial support from this study. The researchers and Dr. Jones disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
Results of a trial using an intensive, 4-year program aimed at blood pressure lowering showed that intervention reduced not only blood pressure, but also significantly reduced the risk of total dementia over that period.
and cognitive impairment no dementia (CIND), a secondary outcome, was also significantly reduced by 16%.
“Blood pressure reduction is effective in reducing the risk of dementia in patients with hypertension,” concluded Jiang He, MD, PhD, professor of epidemiology and medicine and director of Tulane University’s Translational Science Institute, New Orleans. “This proven, effective intervention should be widely scaled up to reduce the global burden of dementia.”
He presented these results from the China Rural Hypertension Control Project (CRHCP) at the annual scientific sessions of the American Heart Association.
Target organ damage
Keith Ferdinand, MD, also from Tulane University, commented on the findings during a press conference at the meeting, noting that the result “opens our opportunity to recognize that the target organ damage of hypertension also now includes dementia.”
The researchers were able to “rigorously lower blood pressure from 157 to 127.6 in the intervention, 155 to 147 in the controls – 22 mg Hg – and if you look at the P values for all the various outcomes, they were very robust,” Dr. Ferdinand said.
Another interesting feature about the strategy used in this trial is that “this was true team-based care,” he pointed out. The trained interventionists in the study, called village doctors, collaborated with primary care physicians and initiated medications. “They stayed on a simple treatment protocol, and they were able to assist patients to ensure they had free medications, health coaching for lifestyle, home blood pressure measurement, and ensuring adherence.”
So, Dr. Ferdinand added, “one of the questions is whether this is a model we can use in other places around the globe, in places with low resources, and in the United States in disadvantaged populations.”
Public health priority
It’s estimated that the global number of those living with dementia will increase from 57.4 million in 2019 to 152.8 million by 2050, Dr. He said. “In the absence of curative treatment, the primary prevention of dementia through risk factor reduction, such as blood pressure lowering, becomes a public health priority.”
Previous randomized trials have lacked sample size and duration but have reported a nonsignificant reduction in dementia associated with antihypertensive treatment in patients with hypertension or a history of stroke, Dr. He noted.
This new trial aimed to test the effectiveness of intensive BP intervention to reduce the risk of all-cause dementia and cognitive impairment over a 48-month intervention period versus usual care.
It was an open-label, blinded-endpoint, cluster-randomized trial, and included 33,995 individual patients from 325 villages in China, aged 40 years and older, with untreated hypertension. The villages were randomly assigned to an intervention group or usual care, stratified by province, county, and township.
Patients were eligible if they had mean untreated systolic BP greater than 140 mm Hg and/or diastolic BP greater than 90 mm Hg or mean treated systolic BP of greater than 130 and/or diastolic greater than 80 mm Hg. Patients with a history of cardiovascular disease, chronic kidney disease, or diabetes and a mean systolic BP greater than 130 mm Hg and/or diastolic BP greater than 80 mm Hg from six measures on two different days were also eligible.
All were enrolled in the China New Rural Cooperative Medical Scheme, which covers 99% of rural residents for health care services, Dr. He noted.
The intervention was a simple stepped-care protocol for hypertension treatment, aimed at achieving a target systolic BP of less than 130 mm Hg and diastolic of less than 80 mm Hg.
Village doctors started and titrated antihypertensive treatment based on a protocol and were able to deliver discounted and free medications to patients. They also did health coaching on lifestyle modification and adherence to medication, and instructed patients on home BP monitoring.
Patients were provided training, supervision, and consultation by primary care physicians and hypertension specialists.
At the month 48 follow-up visit, the participants were assessed by neurologists who were blinded to randomization assignments. Neurologists did a variety of tests and assessments including collecting data on the patient’s medical and psychiatric history and risk factors for dementia, as well as neurologic assessment using the Mini-Mental State Examination, the Functional Activities Questionnaire, and the Quick Dementia Rating System.
The primary outcome was all-cause dementia, defined according to recommendations from the National Institute on Aging–Alzheimer’s Association work groups on diagnostic guidelines for Alzheimer’s disease.
Secondary outcomes included CIND, a composite outcome of dementia or CIND, and a composite of dementia or deaths.
The final diagnosis of all-cause dementia or CIND was made by an expert adjudication panel blinded to the intervention assignment.
At 48 months, 91.3% of patients completed the follow-up for clinical outcomes. Participants were an average of 63 years of age, 61% were female, and 23% had less than a primary school education, Dr. He noted.
The net group differences in systolic and diastolic BP reduction were 22 and 9.3 mm Hg, respectively (P < .0001).
Significant differences were also seen between the groups in the primary outcome of all-cause dementia, as well as secondary outcomes of CIND, dementia or cognitive impairment, or dementia or deaths.
Serious adverse events were more common in the usual care group, and there was no difference between groups in the occurrence of falls or syncope.
The effect was consistent across subgroups, Dr. He said, including age, sex, education, cigarette smoking, body mass index, systolic BP, and fasting plasma glucose at baseline.
First definitive evidence
Invited discussant for the trial, Daniel W. Jones, MD, University of Mississippi Medical Center, Jackson, and past president of the AHA, pointed out that previous results from CRHCP on cardiovascular outcomes, reported earlier in 2023 in The Lancet, showed that, similar to results of the large SPRINT trial, lowering systolic BP to a goal of less than 130 mm Hg reduced a composite endpoint of MI, stroke, heart failure requiring hospitalization, and cardiovascular disease death over the 36-month follow-up.
The SPRINT findings also suggested a possible reduction in dementia, Dr. Jones said.
Now, in these new CRHCP results, “there was a clear benefit for intensive BP control in reducing risk for dementia and cognitive dysfunction,” he said. “This is, importantly, the first definitive evidence of dementia risk reduction demonstrated in a randomized controlled clinical trial. This outcome supports observational data that shows a strong relationship between BP and dementia.”
Since it is the first of its kind though, replication of the results will be important, he noted.
The study also showed that the intervention, using minimally trained village doctors, sustained BP control for 48 months. “This model could be used in any setting with modifications, including in the United States,” Dr. Jones said.
The study was supported by the Ministry of Science and Technology of China; U.S. investigators did not receive financial support from this study. The researchers and Dr. Jones disclosed no relevant financial relationships.
A version of this article appeared on Medscape.com.
FROM AHA 2023