User login
-
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'main-prefix')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
div[contains(@class, 'view-medstat-quiz-listing-panes')]
div[contains(@class, 'pane-article-sidebar-latest-news')]


High and Low Body Mass Indices Promote Respiratory Symptoms
TOPLINE:
Individuals with either high or low body mass index (BMI) showed an increased risk for respiratory symptoms and diseases than those with BMI in the normal range.
METHODOLOGY:
- The researchers reviewed data from the National Health and Nutrition Examination Survey (NHANES) from 2003 to 2012; the study population included 12,719 adults older than 40 years with data on respiratory symptoms; 51% were female, and 53.3% were non-Hispanic White individuals.
- The study population was divided into quartiles based on BMI as follows: 3180 individuals with BMI of 13.2-24.9 kg/m2, 3175 with BMI of 24.9-28.4 kg/m2, 3180 with BMI of 28.4-32.5 kg/m2, and 3184 with BMI of 32.5-82.0 kg/m2.
- The study sought to assess the correlation between BMI and respiratory symptoms (cough, wheezing, and dyspnea), chronic obstructive pulmonary disease (COPD), and asthma in unadjusted and adjusted models based on sex, race, marital status, poverty-income ratio (PIR), education level, and smoking status.
TAKEAWAY:
- In a logistic regression and curve fitting analysis, BMI showed a U-shaped relationship with respiratory symptoms, asthma, and COPD, with increased risk in individuals with high or low BMI than those with BMIs in the middle quartiles.
- In a stratified analysis by race, the risk for cough was significantly higher among non-Hispanic Black individuals than other races (P < .0001), and a higher BMI was associated with an increased risk for COPD in non-Hispanic Black individuals (odds ratio, 1.053; P < .0001).
- The researchers found no significant impact of biological sex on the relationship between BMI and respiratory symptoms, COPD, or asthma.
- The results support previous studies showing that a BMI that is too low can be detrimental to health.
IN PRACTICE:
“These results suggest that the risk of small airway obstruction in underweight individuals deserves more attention and that excessive wasting may also affect the prognosis of patients with COPD,” the researchers wrote.
SOURCE:
The lead author on the study was Yuefeng Sun of Shandong University of Traditional Chinese Medicine, Jinan, China. The study was published online on January 10, 2024, in Scientific Reports.
LIMITATIONS:
The cross-sectional NHANES database prevented conclusions of causality, and potential confounding factors that were not accounted for could have affected the results.
DISCLOSURES:
The study was supported by the Shandong Province Taishan Scholar Project. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
Individuals with either high or low body mass index (BMI) showed an increased risk for respiratory symptoms and diseases than those with BMI in the normal range.
METHODOLOGY:
- The researchers reviewed data from the National Health and Nutrition Examination Survey (NHANES) from 2003 to 2012; the study population included 12,719 adults older than 40 years with data on respiratory symptoms; 51% were female, and 53.3% were non-Hispanic White individuals.
- The study population was divided into quartiles based on BMI as follows: 3180 individuals with BMI of 13.2-24.9 kg/m2, 3175 with BMI of 24.9-28.4 kg/m2, 3180 with BMI of 28.4-32.5 kg/m2, and 3184 with BMI of 32.5-82.0 kg/m2.
- The study sought to assess the correlation between BMI and respiratory symptoms (cough, wheezing, and dyspnea), chronic obstructive pulmonary disease (COPD), and asthma in unadjusted and adjusted models based on sex, race, marital status, poverty-income ratio (PIR), education level, and smoking status.
TAKEAWAY:
- In a logistic regression and curve fitting analysis, BMI showed a U-shaped relationship with respiratory symptoms, asthma, and COPD, with increased risk in individuals with high or low BMI than those with BMIs in the middle quartiles.
- In a stratified analysis by race, the risk for cough was significantly higher among non-Hispanic Black individuals than other races (P < .0001), and a higher BMI was associated with an increased risk for COPD in non-Hispanic Black individuals (odds ratio, 1.053; P < .0001).
- The researchers found no significant impact of biological sex on the relationship between BMI and respiratory symptoms, COPD, or asthma.
- The results support previous studies showing that a BMI that is too low can be detrimental to health.
IN PRACTICE:
“These results suggest that the risk of small airway obstruction in underweight individuals deserves more attention and that excessive wasting may also affect the prognosis of patients with COPD,” the researchers wrote.
SOURCE:
The lead author on the study was Yuefeng Sun of Shandong University of Traditional Chinese Medicine, Jinan, China. The study was published online on January 10, 2024, in Scientific Reports.
LIMITATIONS:
The cross-sectional NHANES database prevented conclusions of causality, and potential confounding factors that were not accounted for could have affected the results.
DISCLOSURES:
The study was supported by the Shandong Province Taishan Scholar Project. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
TOPLINE:
Individuals with either high or low body mass index (BMI) showed an increased risk for respiratory symptoms and diseases than those with BMI in the normal range.
METHODOLOGY:
- The researchers reviewed data from the National Health and Nutrition Examination Survey (NHANES) from 2003 to 2012; the study population included 12,719 adults older than 40 years with data on respiratory symptoms; 51% were female, and 53.3% were non-Hispanic White individuals.
- The study population was divided into quartiles based on BMI as follows: 3180 individuals with BMI of 13.2-24.9 kg/m2, 3175 with BMI of 24.9-28.4 kg/m2, 3180 with BMI of 28.4-32.5 kg/m2, and 3184 with BMI of 32.5-82.0 kg/m2.
- The study sought to assess the correlation between BMI and respiratory symptoms (cough, wheezing, and dyspnea), chronic obstructive pulmonary disease (COPD), and asthma in unadjusted and adjusted models based on sex, race, marital status, poverty-income ratio (PIR), education level, and smoking status.
TAKEAWAY:
- In a logistic regression and curve fitting analysis, BMI showed a U-shaped relationship with respiratory symptoms, asthma, and COPD, with increased risk in individuals with high or low BMI than those with BMIs in the middle quartiles.
- In a stratified analysis by race, the risk for cough was significantly higher among non-Hispanic Black individuals than other races (P < .0001), and a higher BMI was associated with an increased risk for COPD in non-Hispanic Black individuals (odds ratio, 1.053; P < .0001).
- The researchers found no significant impact of biological sex on the relationship between BMI and respiratory symptoms, COPD, or asthma.
- The results support previous studies showing that a BMI that is too low can be detrimental to health.
IN PRACTICE:
“These results suggest that the risk of small airway obstruction in underweight individuals deserves more attention and that excessive wasting may also affect the prognosis of patients with COPD,” the researchers wrote.
SOURCE:
The lead author on the study was Yuefeng Sun of Shandong University of Traditional Chinese Medicine, Jinan, China. The study was published online on January 10, 2024, in Scientific Reports.
LIMITATIONS:
The cross-sectional NHANES database prevented conclusions of causality, and potential confounding factors that were not accounted for could have affected the results.
DISCLOSURES:
The study was supported by the Shandong Province Taishan Scholar Project. The researchers had no financial conflicts to disclose.
A version of this article appeared on Medscape.com.
Researchers Uncover Nanoplastics in Water Bottles
Using an advanced microscopic technique, American researchers have detected 100,000 nanoplastic molecules per liter of water in plastic bottles. Because of their small size, these particles can enter the bloodstream, cells, and the brain, thus posing potential health risks. The study, recently published in the Proceedings of the National Academy of Sciences, raises concerns about the impact of these nanoparticles.
An Unknown Realm
Formed as plastics break down into increasingly small pieces, these particles are consumed by humans and other organisms, with unknown effects on health and ecosystems. Whereas macroplastics have been found in various organs, including the lungs and liver, the study marks a unique exploration into the world of nanoplastics.
Concerns about nanoplastic presence in humans intensified when a 2018 study revealed contamination signs in 93% of 259 examined bottles from nine countries.
The novelty of this research lies in its focus, using a refined spectrometry method, on the poorly understood world of nanoplastics, which derive from the decomposition of microplastics. For the first time, American researchers, including biophysicists and chemists, counted and identified these tiny particles in bottled water. On average, they found around 240,000 detectable plastic fragments per liter, which is 10-100 times more than previous estimates based on larger sizes.
Microplastics are defined as fragments ranging from 5 mm to 1 µm, whereas nanoplastics, particles < 1 µm, are measured in billionths of a meter.
In contrast to microplastics, nanoplastics are so small that they can traverse the intestines and lungs and move directly into the bloodstream, reaching organs such as the heart or brain or even the fetus via the placenta.
“This was previously an obscure, unexplored area. Toxicity studies could only speculate about what was in there,” said Beizhan Yan, PhD, coauthor of the study and environmental chemist at the Lamont–Doherty Earth Observatory of Columbia University, New York. “This study opens a window for us to observe a world we were not exposed to before.”
90% Nanoplastics Found
The new study employed a technique called stimulated Raman scattering microscopy, which was invented by study coauthor Wei Min, a biophysicist at Columbia. This method involves probing samples simultaneously with two lasers tuned to resonate specific molecules.
Researchers tested three bottled water brands that are popular in the United States, analyzing plastic particles up to 100 nm in size. They identified 110,000-370,000 plastic particles per liter. About 90% were nanoplastics — which are invisible by standard imaging techniques — and the rest were microplastics. The study also identified the seven plastics involved.
The most common is polyamide, a type of nylon, likely from plastic filters purportedly used to purify water before bottling. Next is polyethylene terephthalate, which is commonly used for water bottles and other food containers. Researchers also found other common plastics, including polystyrene, polyvinyl chloride, and methyl methacrylate, used in various industrial processes.
Not Size But Quantity
What’s more concerning is that the seven types of plastics accounted for only about 10% of all nanoparticles found in the samples. Researchers have no idea about the composition of the remaining 90%. If these are all nanoparticles, their number could reach tens of millions per liter, representing the complex composition of seemingly simple water samples, as noted by the authors.
Researchers now plan to expand beyond bottled water, exploring the vast realm of nanoplastics. They emphasize that, in terms of mass, nanoplastics are far smaller than microplastics, but “it’s not about size. It’s about the numbers as smaller things can easily penetrate us.”
The team aims to study tap water, which also contains microplastics but in much smaller proportions than bottled water.
This article was translated from the Medscape French edition.
Using an advanced microscopic technique, American researchers have detected 100,000 nanoplastic molecules per liter of water in plastic bottles. Because of their small size, these particles can enter the bloodstream, cells, and the brain, thus posing potential health risks. The study, recently published in the Proceedings of the National Academy of Sciences, raises concerns about the impact of these nanoparticles.
An Unknown Realm
Formed as plastics break down into increasingly small pieces, these particles are consumed by humans and other organisms, with unknown effects on health and ecosystems. Whereas macroplastics have been found in various organs, including the lungs and liver, the study marks a unique exploration into the world of nanoplastics.
Concerns about nanoplastic presence in humans intensified when a 2018 study revealed contamination signs in 93% of 259 examined bottles from nine countries.
The novelty of this research lies in its focus, using a refined spectrometry method, on the poorly understood world of nanoplastics, which derive from the decomposition of microplastics. For the first time, American researchers, including biophysicists and chemists, counted and identified these tiny particles in bottled water. On average, they found around 240,000 detectable plastic fragments per liter, which is 10-100 times more than previous estimates based on larger sizes.
Microplastics are defined as fragments ranging from 5 mm to 1 µm, whereas nanoplastics, particles < 1 µm, are measured in billionths of a meter.
In contrast to microplastics, nanoplastics are so small that they can traverse the intestines and lungs and move directly into the bloodstream, reaching organs such as the heart or brain or even the fetus via the placenta.
“This was previously an obscure, unexplored area. Toxicity studies could only speculate about what was in there,” said Beizhan Yan, PhD, coauthor of the study and environmental chemist at the Lamont–Doherty Earth Observatory of Columbia University, New York. “This study opens a window for us to observe a world we were not exposed to before.”
90% Nanoplastics Found
The new study employed a technique called stimulated Raman scattering microscopy, which was invented by study coauthor Wei Min, a biophysicist at Columbia. This method involves probing samples simultaneously with two lasers tuned to resonate specific molecules.
Researchers tested three bottled water brands that are popular in the United States, analyzing plastic particles up to 100 nm in size. They identified 110,000-370,000 plastic particles per liter. About 90% were nanoplastics — which are invisible by standard imaging techniques — and the rest were microplastics. The study also identified the seven plastics involved.
The most common is polyamide, a type of nylon, likely from plastic filters purportedly used to purify water before bottling. Next is polyethylene terephthalate, which is commonly used for water bottles and other food containers. Researchers also found other common plastics, including polystyrene, polyvinyl chloride, and methyl methacrylate, used in various industrial processes.
Not Size But Quantity
What’s more concerning is that the seven types of plastics accounted for only about 10% of all nanoparticles found in the samples. Researchers have no idea about the composition of the remaining 90%. If these are all nanoparticles, their number could reach tens of millions per liter, representing the complex composition of seemingly simple water samples, as noted by the authors.
Researchers now plan to expand beyond bottled water, exploring the vast realm of nanoplastics. They emphasize that, in terms of mass, nanoplastics are far smaller than microplastics, but “it’s not about size. It’s about the numbers as smaller things can easily penetrate us.”
The team aims to study tap water, which also contains microplastics but in much smaller proportions than bottled water.
This article was translated from the Medscape French edition.
Using an advanced microscopic technique, American researchers have detected 100,000 nanoplastic molecules per liter of water in plastic bottles. Because of their small size, these particles can enter the bloodstream, cells, and the brain, thus posing potential health risks. The study, recently published in the Proceedings of the National Academy of Sciences, raises concerns about the impact of these nanoparticles.
An Unknown Realm
Formed as plastics break down into increasingly small pieces, these particles are consumed by humans and other organisms, with unknown effects on health and ecosystems. Whereas macroplastics have been found in various organs, including the lungs and liver, the study marks a unique exploration into the world of nanoplastics.
Concerns about nanoplastic presence in humans intensified when a 2018 study revealed contamination signs in 93% of 259 examined bottles from nine countries.
The novelty of this research lies in its focus, using a refined spectrometry method, on the poorly understood world of nanoplastics, which derive from the decomposition of microplastics. For the first time, American researchers, including biophysicists and chemists, counted and identified these tiny particles in bottled water. On average, they found around 240,000 detectable plastic fragments per liter, which is 10-100 times more than previous estimates based on larger sizes.
Microplastics are defined as fragments ranging from 5 mm to 1 µm, whereas nanoplastics, particles < 1 µm, are measured in billionths of a meter.
In contrast to microplastics, nanoplastics are so small that they can traverse the intestines and lungs and move directly into the bloodstream, reaching organs such as the heart or brain or even the fetus via the placenta.
“This was previously an obscure, unexplored area. Toxicity studies could only speculate about what was in there,” said Beizhan Yan, PhD, coauthor of the study and environmental chemist at the Lamont–Doherty Earth Observatory of Columbia University, New York. “This study opens a window for us to observe a world we were not exposed to before.”
90% Nanoplastics Found
The new study employed a technique called stimulated Raman scattering microscopy, which was invented by study coauthor Wei Min, a biophysicist at Columbia. This method involves probing samples simultaneously with two lasers tuned to resonate specific molecules.
Researchers tested three bottled water brands that are popular in the United States, analyzing plastic particles up to 100 nm in size. They identified 110,000-370,000 plastic particles per liter. About 90% were nanoplastics — which are invisible by standard imaging techniques — and the rest were microplastics. The study also identified the seven plastics involved.
The most common is polyamide, a type of nylon, likely from plastic filters purportedly used to purify water before bottling. Next is polyethylene terephthalate, which is commonly used for water bottles and other food containers. Researchers also found other common plastics, including polystyrene, polyvinyl chloride, and methyl methacrylate, used in various industrial processes.
Not Size But Quantity
What’s more concerning is that the seven types of plastics accounted for only about 10% of all nanoparticles found in the samples. Researchers have no idea about the composition of the remaining 90%. If these are all nanoparticles, their number could reach tens of millions per liter, representing the complex composition of seemingly simple water samples, as noted by the authors.
Researchers now plan to expand beyond bottled water, exploring the vast realm of nanoplastics. They emphasize that, in terms of mass, nanoplastics are far smaller than microplastics, but “it’s not about size. It’s about the numbers as smaller things can easily penetrate us.”
The team aims to study tap water, which also contains microplastics but in much smaller proportions than bottled water.
This article was translated from the Medscape French edition.
FROM THE PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES
Why Are Women More Likely to Get Long COVID?
Annette Gillaspie, a nurse in a small Oregon hospital, hoped she would be back working with patients by now. She contracted COVID-19 on the job early in the pandemic and ended up with long COVID.
After recovering a bit, her fatigue and dizziness returned, and today she is still working a desk job. She has also experienced more severe menstrual periods than before she had COVID.
“Being a female with long COVID definitely does add to the roller-coaster effect of symptoms,” Ms. Gillaspie said.
reported by the Centers for Disease Control and Prevention (CDC). Researchers are trying to determine why, what causes the gender disparity, and how best to treat it.
Scientists are also starting to look at the impact of long COVID on female reproductive health, including menstruation, pregnancy, and menopause.
Sex differences are common in infection-associated illnesses, said Beth Pollack, MS, a research scientist specializing in long COVID in the Massachusetts Institute of Technology’s Department of Biological Engineering, Cambridge, Massachusetts. “It informs research priorities and the lens with which we understand long COVID.”
For example, reproductive health issues for women, such as puberty, pregnancy, and menopause, can alter the course of illness in a subset of women in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and postural orthostatic tachycardia syndrome (POTS), a condition that can cause dizziness and worse.
“This suggests that sex hormones may play key roles in immune responses to infections,” Ms. Pollack said.
ME/CFS and a Possible Link to Long COVID in Women
Some of the research into long COVID is being led by teams studying infection-associated chronic illnesses like ME/CFS.
The problem: Advocates say ME/CFS has been under-researched. Poorly understood for years, the condition is one of a handful of chronic illnesses linked to infections, including Lyme disease and now long COVID. Perhaps not coincidently, they are more likely to affect women.
Many of the research findings about long COVID mirror data that emerged in past ME/CFS research, said Jaime Seltzer, the scientific director at #MEAction, Santa Monica, California, an advocacy group. One point in particular: ME/CFS strikes women about twice as much as men, according to the CDC.
Ms. Seltzer said the response to long COVID could be much further ahead if the research community acknowledged the work done over the years on ME/CFS. Many of the potential biomarkers and risk factors emerging for long COVID were also suspected in ME/CFS, but not thoroughly studied, she said.
She also said not enough work has been done to unravel the links between gender and these chronic conditions.
“We’re stuck in this Groundhog Day situation,” she said. “There isn’t any research, so we can’t say anything definitively.”
Some New Research, Some New Clues
Scientists like Ms. Pollack are slowly making inroads. She was lead author on a 2023 review investigating the impact of long COVID on female reproductive health. The paper highlights long COVID links to ME/CFS, POTS, and Ehlers-Danlos syndrome (EDS), as well as a resulting laundry list of female reproductive health issues. The hope is physicians will examine how the menstrual cycle, pregnancy, and menopause affect symptoms and illness progression of long COVID.
The Tal Research group at MIT (where Ms. Pollack works) has also added long COVID to the list of infection-associated illnesses it studies. The lab is conducting a large study looking into both Lyme disease and long COVID. The goals are to identify biomarkers that can predict who will not recover and to advance available treatments.
Another MIT program, “SEXX + Immunity” holds seminars and networking sessions for scientists looking into the role of female and male biology in immune responses to infection.
Barriers to Progress Remain
On the clinical side, female patients with long COVID also have to deal with a historical bias that still lurks in medicine when it comes to women’s health, said Alba Azola, MD, an assistant professor of physical medicine at Johns Hopkins Medicine, Baltimore, Maryland.
Dr. Azola said she has discovered clinical descriptions of ME/CFE in the literature archives that describe it as “neurasthenia” and dismiss it as psychological.
Patients say that it is still happening, and while it may not be so blunt, “you can read between the lines,” Dr. Azola said.
Dr. Azola, who has worked with long COVID patients and is now seeing people with ME/CFS, said the symptoms of infection-associated chronic illness can mimic menopause, and many of her patients received that misdiagnosis. She recommends that doctors rule out long COVID for women with multiple symptoms before attributing symptoms to menopause.
Seeing that some long COVID patients were developing ME/CFS, staff at the Bateman Horne Center in Salt Lake City, Utah, set up a program for the condition in 2021. They were already treating patients with ME/CFS and what they call “multi-symptom chronic complex diseases.”
Jennifer Bell, a certified nurse practitioner at the center, said she has not seen any patients with ovarian failure but plenty with reproductive health issues.
“There definitely is a hormonal connection, but I don’t think there’s a good understanding about what is happening,” she said.
Most of her patients are female, and the more serious patients tend to go through a worsening of their symptoms in the week prior to getting a period, she said.
One thing Ms. Bell said she’s noticed in the past year is an increase in patients with EDS, which is also more common in women.
Like long COVID, many of the conditions traditionally treated at the center have no cure. But Ms. Bell said the center has developed an expertise in treating post-exertional malaise, a common symptom of long COVID, and keeps up with the literature for treatments to try, like the combination of guanfacine and the antioxidant N-acetyl cysteine to treat brain fog, an approach developed at Yale.
“It’s a very challenging illness to treat,” Ms. Bell said.
Since the emergence of long COVID, researchers have warned that symptoms vary so much from person to person that treatment will need to be targeted.
Ms. Pollack of MIT agrees and sees a big role for personalized medicine.
We need to “identify phenotypes within and across these overlapping and co-occurring illnesses so that we can identify the right therapeutics for each person,” she said.
As for Annette Gillaspie, she still hopes her long COVID will subside so she can get out from behind the desk and return to her normal nursing duties.
“I just got to a point where I realized I’m likely never going to be able to do my job,” she said. “It was incredibly heart breaking, but it’s the reality of long COVID, and I know I’m not the only one to have to step away from a job I loved.”
A version of this article appeared on Medscape.com.
Annette Gillaspie, a nurse in a small Oregon hospital, hoped she would be back working with patients by now. She contracted COVID-19 on the job early in the pandemic and ended up with long COVID.
After recovering a bit, her fatigue and dizziness returned, and today she is still working a desk job. She has also experienced more severe menstrual periods than before she had COVID.
“Being a female with long COVID definitely does add to the roller-coaster effect of symptoms,” Ms. Gillaspie said.
reported by the Centers for Disease Control and Prevention (CDC). Researchers are trying to determine why, what causes the gender disparity, and how best to treat it.
Scientists are also starting to look at the impact of long COVID on female reproductive health, including menstruation, pregnancy, and menopause.
Sex differences are common in infection-associated illnesses, said Beth Pollack, MS, a research scientist specializing in long COVID in the Massachusetts Institute of Technology’s Department of Biological Engineering, Cambridge, Massachusetts. “It informs research priorities and the lens with which we understand long COVID.”
For example, reproductive health issues for women, such as puberty, pregnancy, and menopause, can alter the course of illness in a subset of women in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and postural orthostatic tachycardia syndrome (POTS), a condition that can cause dizziness and worse.
“This suggests that sex hormones may play key roles in immune responses to infections,” Ms. Pollack said.
ME/CFS and a Possible Link to Long COVID in Women
Some of the research into long COVID is being led by teams studying infection-associated chronic illnesses like ME/CFS.
The problem: Advocates say ME/CFS has been under-researched. Poorly understood for years, the condition is one of a handful of chronic illnesses linked to infections, including Lyme disease and now long COVID. Perhaps not coincidently, they are more likely to affect women.
Many of the research findings about long COVID mirror data that emerged in past ME/CFS research, said Jaime Seltzer, the scientific director at #MEAction, Santa Monica, California, an advocacy group. One point in particular: ME/CFS strikes women about twice as much as men, according to the CDC.
Ms. Seltzer said the response to long COVID could be much further ahead if the research community acknowledged the work done over the years on ME/CFS. Many of the potential biomarkers and risk factors emerging for long COVID were also suspected in ME/CFS, but not thoroughly studied, she said.
She also said not enough work has been done to unravel the links between gender and these chronic conditions.
“We’re stuck in this Groundhog Day situation,” she said. “There isn’t any research, so we can’t say anything definitively.”
Some New Research, Some New Clues
Scientists like Ms. Pollack are slowly making inroads. She was lead author on a 2023 review investigating the impact of long COVID on female reproductive health. The paper highlights long COVID links to ME/CFS, POTS, and Ehlers-Danlos syndrome (EDS), as well as a resulting laundry list of female reproductive health issues. The hope is physicians will examine how the menstrual cycle, pregnancy, and menopause affect symptoms and illness progression of long COVID.
The Tal Research group at MIT (where Ms. Pollack works) has also added long COVID to the list of infection-associated illnesses it studies. The lab is conducting a large study looking into both Lyme disease and long COVID. The goals are to identify biomarkers that can predict who will not recover and to advance available treatments.
Another MIT program, “SEXX + Immunity” holds seminars and networking sessions for scientists looking into the role of female and male biology in immune responses to infection.
Barriers to Progress Remain
On the clinical side, female patients with long COVID also have to deal with a historical bias that still lurks in medicine when it comes to women’s health, said Alba Azola, MD, an assistant professor of physical medicine at Johns Hopkins Medicine, Baltimore, Maryland.
Dr. Azola said she has discovered clinical descriptions of ME/CFE in the literature archives that describe it as “neurasthenia” and dismiss it as psychological.
Patients say that it is still happening, and while it may not be so blunt, “you can read between the lines,” Dr. Azola said.
Dr. Azola, who has worked with long COVID patients and is now seeing people with ME/CFS, said the symptoms of infection-associated chronic illness can mimic menopause, and many of her patients received that misdiagnosis. She recommends that doctors rule out long COVID for women with multiple symptoms before attributing symptoms to menopause.
Seeing that some long COVID patients were developing ME/CFS, staff at the Bateman Horne Center in Salt Lake City, Utah, set up a program for the condition in 2021. They were already treating patients with ME/CFS and what they call “multi-symptom chronic complex diseases.”
Jennifer Bell, a certified nurse practitioner at the center, said she has not seen any patients with ovarian failure but plenty with reproductive health issues.
“There definitely is a hormonal connection, but I don’t think there’s a good understanding about what is happening,” she said.
Most of her patients are female, and the more serious patients tend to go through a worsening of their symptoms in the week prior to getting a period, she said.
One thing Ms. Bell said she’s noticed in the past year is an increase in patients with EDS, which is also more common in women.
Like long COVID, many of the conditions traditionally treated at the center have no cure. But Ms. Bell said the center has developed an expertise in treating post-exertional malaise, a common symptom of long COVID, and keeps up with the literature for treatments to try, like the combination of guanfacine and the antioxidant N-acetyl cysteine to treat brain fog, an approach developed at Yale.
“It’s a very challenging illness to treat,” Ms. Bell said.
Since the emergence of long COVID, researchers have warned that symptoms vary so much from person to person that treatment will need to be targeted.
Ms. Pollack of MIT agrees and sees a big role for personalized medicine.
We need to “identify phenotypes within and across these overlapping and co-occurring illnesses so that we can identify the right therapeutics for each person,” she said.
As for Annette Gillaspie, she still hopes her long COVID will subside so she can get out from behind the desk and return to her normal nursing duties.
“I just got to a point where I realized I’m likely never going to be able to do my job,” she said. “It was incredibly heart breaking, but it’s the reality of long COVID, and I know I’m not the only one to have to step away from a job I loved.”
A version of this article appeared on Medscape.com.
Annette Gillaspie, a nurse in a small Oregon hospital, hoped she would be back working with patients by now. She contracted COVID-19 on the job early in the pandemic and ended up with long COVID.
After recovering a bit, her fatigue and dizziness returned, and today she is still working a desk job. She has also experienced more severe menstrual periods than before she had COVID.
“Being a female with long COVID definitely does add to the roller-coaster effect of symptoms,” Ms. Gillaspie said.
reported by the Centers for Disease Control and Prevention (CDC). Researchers are trying to determine why, what causes the gender disparity, and how best to treat it.
Scientists are also starting to look at the impact of long COVID on female reproductive health, including menstruation, pregnancy, and menopause.
Sex differences are common in infection-associated illnesses, said Beth Pollack, MS, a research scientist specializing in long COVID in the Massachusetts Institute of Technology’s Department of Biological Engineering, Cambridge, Massachusetts. “It informs research priorities and the lens with which we understand long COVID.”
For example, reproductive health issues for women, such as puberty, pregnancy, and menopause, can alter the course of illness in a subset of women in myalgic encephalomyelitis/chronic fatigue syndrome (ME/CFS) and postural orthostatic tachycardia syndrome (POTS), a condition that can cause dizziness and worse.
“This suggests that sex hormones may play key roles in immune responses to infections,” Ms. Pollack said.
ME/CFS and a Possible Link to Long COVID in Women
Some of the research into long COVID is being led by teams studying infection-associated chronic illnesses like ME/CFS.
The problem: Advocates say ME/CFS has been under-researched. Poorly understood for years, the condition is one of a handful of chronic illnesses linked to infections, including Lyme disease and now long COVID. Perhaps not coincidently, they are more likely to affect women.
Many of the research findings about long COVID mirror data that emerged in past ME/CFS research, said Jaime Seltzer, the scientific director at #MEAction, Santa Monica, California, an advocacy group. One point in particular: ME/CFS strikes women about twice as much as men, according to the CDC.
Ms. Seltzer said the response to long COVID could be much further ahead if the research community acknowledged the work done over the years on ME/CFS. Many of the potential biomarkers and risk factors emerging for long COVID were also suspected in ME/CFS, but not thoroughly studied, she said.
She also said not enough work has been done to unravel the links between gender and these chronic conditions.
“We’re stuck in this Groundhog Day situation,” she said. “There isn’t any research, so we can’t say anything definitively.”
Some New Research, Some New Clues
Scientists like Ms. Pollack are slowly making inroads. She was lead author on a 2023 review investigating the impact of long COVID on female reproductive health. The paper highlights long COVID links to ME/CFS, POTS, and Ehlers-Danlos syndrome (EDS), as well as a resulting laundry list of female reproductive health issues. The hope is physicians will examine how the menstrual cycle, pregnancy, and menopause affect symptoms and illness progression of long COVID.
The Tal Research group at MIT (where Ms. Pollack works) has also added long COVID to the list of infection-associated illnesses it studies. The lab is conducting a large study looking into both Lyme disease and long COVID. The goals are to identify biomarkers that can predict who will not recover and to advance available treatments.
Another MIT program, “SEXX + Immunity” holds seminars and networking sessions for scientists looking into the role of female and male biology in immune responses to infection.
Barriers to Progress Remain
On the clinical side, female patients with long COVID also have to deal with a historical bias that still lurks in medicine when it comes to women’s health, said Alba Azola, MD, an assistant professor of physical medicine at Johns Hopkins Medicine, Baltimore, Maryland.
Dr. Azola said she has discovered clinical descriptions of ME/CFE in the literature archives that describe it as “neurasthenia” and dismiss it as psychological.
Patients say that it is still happening, and while it may not be so blunt, “you can read between the lines,” Dr. Azola said.
Dr. Azola, who has worked with long COVID patients and is now seeing people with ME/CFS, said the symptoms of infection-associated chronic illness can mimic menopause, and many of her patients received that misdiagnosis. She recommends that doctors rule out long COVID for women with multiple symptoms before attributing symptoms to menopause.
Seeing that some long COVID patients were developing ME/CFS, staff at the Bateman Horne Center in Salt Lake City, Utah, set up a program for the condition in 2021. They were already treating patients with ME/CFS and what they call “multi-symptom chronic complex diseases.”
Jennifer Bell, a certified nurse practitioner at the center, said she has not seen any patients with ovarian failure but plenty with reproductive health issues.
“There definitely is a hormonal connection, but I don’t think there’s a good understanding about what is happening,” she said.
Most of her patients are female, and the more serious patients tend to go through a worsening of their symptoms in the week prior to getting a period, she said.
One thing Ms. Bell said she’s noticed in the past year is an increase in patients with EDS, which is also more common in women.
Like long COVID, many of the conditions traditionally treated at the center have no cure. But Ms. Bell said the center has developed an expertise in treating post-exertional malaise, a common symptom of long COVID, and keeps up with the literature for treatments to try, like the combination of guanfacine and the antioxidant N-acetyl cysteine to treat brain fog, an approach developed at Yale.
“It’s a very challenging illness to treat,” Ms. Bell said.
Since the emergence of long COVID, researchers have warned that symptoms vary so much from person to person that treatment will need to be targeted.
Ms. Pollack of MIT agrees and sees a big role for personalized medicine.
We need to “identify phenotypes within and across these overlapping and co-occurring illnesses so that we can identify the right therapeutics for each person,” she said.
As for Annette Gillaspie, she still hopes her long COVID will subside so she can get out from behind the desk and return to her normal nursing duties.
“I just got to a point where I realized I’m likely never going to be able to do my job,” she said. “It was incredibly heart breaking, but it’s the reality of long COVID, and I know I’m not the only one to have to step away from a job I loved.”
A version of this article appeared on Medscape.com.
Smoking Associated With Increased Risk for Hair Loss Among Men
, according to a new study.
In addition, the odds of developing AGA are higher among those who smoke at least 10 cigarettes per day than among those who smoke less, the study authors found.
“Men who smoke are more likely to develop and experience progression of male pattern hair loss,” lead author Aditya Gupta, MD, PhD, professor of medicine at the University of Toronto, Toronto, and director of clinical research at Mediprobe Research Inc., London, Ontario, Canada, told this news organization.
“Our patients with male pattern baldness need to be educated about the negative effects of smoking, given that this condition can have a profound negative psychological impact on those who suffer from it,” he said.
The study was published online in the Journal of Cosmetic Dermatology.
Analyzing Smoking’s Effects
Smoking generally has been accepted as a risk factor for the development and progression of AGA or the most common form of hair loss. The research evidence on this association has been inconsistent, however, the authors wrote.
The investigators conducted a review and meta-analysis of eight observational studies to understand the links between smoking and AGA. Ever-smokers were defined as current and former smokers.
Overall, based on six studies, men who have ever smoked are 1.8 times more likely (P < .05) to develop AGA.
Based on two studies, men who smoke 10 or more cigarettes daily are about twice as likely (P < .05) to develop AGA than those who smoke up to 10 cigarettes per day.
Based on four studies, ever smoking is associated with 1.3 times higher odds of AGA progressing from mild (ie, Norwood-Hamilton stages I-III) to more severe (stages IV-VII) than among those who have never smoked.
Based on two studies, there’s no association between AGA progression and smoking intensity (as defined as smoking up to 20 cigarettes daily vs smoking 20 or more cigarettes per day).
“Though our pooled analysis found no significant association between smoking intensity and severity of male AGA, a positive correlation may exist and be detected through an analysis that is statistically better powered,” said Dr. Gupta.
The investigators noted the limitations of their analysis, such as its reliance on observational studies and its lack of data about nicotine levels, smoking intensity, and smoking cessation among study participants.
Additional studies are needed to better understand the links between smoking and hair loss, said Dr. Gupta, as well as the effects of smoking cessation.
Improving Practice and Research
Commenting on the findings for this news organization, Arash Babadjouni, MD, a dermatologist at Midwestern University, Glendale, Arizona, said, “Smoking is not only a preventable cause of significant systemic disease but also affects the follicular growth cycle and fiber pigmentation. The prevalence of hair loss and premature hair graying is higher in smokers than nonsmokers.”
Dr. Babadjouni, who wasn’t involved with this study, has researched the associations between smoking and hair loss and premature hair graying.
“Evidence of this association can be used to clinically promote smoking cessation and emphasize the consequences of smoking on hair,” he said. “Smoking status should be assessed in patients who are presenting to their dermatologist and physicians alike for evaluation of alopecia and premature hair graying.”
The study was conducted without outside funding, and the authors declared no conflicts of interest. Dr. Babadjouni reported no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to a new study.
In addition, the odds of developing AGA are higher among those who smoke at least 10 cigarettes per day than among those who smoke less, the study authors found.
“Men who smoke are more likely to develop and experience progression of male pattern hair loss,” lead author Aditya Gupta, MD, PhD, professor of medicine at the University of Toronto, Toronto, and director of clinical research at Mediprobe Research Inc., London, Ontario, Canada, told this news organization.
“Our patients with male pattern baldness need to be educated about the negative effects of smoking, given that this condition can have a profound negative psychological impact on those who suffer from it,” he said.
The study was published online in the Journal of Cosmetic Dermatology.
Analyzing Smoking’s Effects
Smoking generally has been accepted as a risk factor for the development and progression of AGA or the most common form of hair loss. The research evidence on this association has been inconsistent, however, the authors wrote.
The investigators conducted a review and meta-analysis of eight observational studies to understand the links between smoking and AGA. Ever-smokers were defined as current and former smokers.
Overall, based on six studies, men who have ever smoked are 1.8 times more likely (P < .05) to develop AGA.
Based on two studies, men who smoke 10 or more cigarettes daily are about twice as likely (P < .05) to develop AGA than those who smoke up to 10 cigarettes per day.
Based on four studies, ever smoking is associated with 1.3 times higher odds of AGA progressing from mild (ie, Norwood-Hamilton stages I-III) to more severe (stages IV-VII) than among those who have never smoked.
Based on two studies, there’s no association between AGA progression and smoking intensity (as defined as smoking up to 20 cigarettes daily vs smoking 20 or more cigarettes per day).
“Though our pooled analysis found no significant association between smoking intensity and severity of male AGA, a positive correlation may exist and be detected through an analysis that is statistically better powered,” said Dr. Gupta.
The investigators noted the limitations of their analysis, such as its reliance on observational studies and its lack of data about nicotine levels, smoking intensity, and smoking cessation among study participants.
Additional studies are needed to better understand the links between smoking and hair loss, said Dr. Gupta, as well as the effects of smoking cessation.
Improving Practice and Research
Commenting on the findings for this news organization, Arash Babadjouni, MD, a dermatologist at Midwestern University, Glendale, Arizona, said, “Smoking is not only a preventable cause of significant systemic disease but also affects the follicular growth cycle and fiber pigmentation. The prevalence of hair loss and premature hair graying is higher in smokers than nonsmokers.”
Dr. Babadjouni, who wasn’t involved with this study, has researched the associations between smoking and hair loss and premature hair graying.
“Evidence of this association can be used to clinically promote smoking cessation and emphasize the consequences of smoking on hair,” he said. “Smoking status should be assessed in patients who are presenting to their dermatologist and physicians alike for evaluation of alopecia and premature hair graying.”
The study was conducted without outside funding, and the authors declared no conflicts of interest. Dr. Babadjouni reported no relevant disclosures.
A version of this article appeared on Medscape.com.
, according to a new study.
In addition, the odds of developing AGA are higher among those who smoke at least 10 cigarettes per day than among those who smoke less, the study authors found.
“Men who smoke are more likely to develop and experience progression of male pattern hair loss,” lead author Aditya Gupta, MD, PhD, professor of medicine at the University of Toronto, Toronto, and director of clinical research at Mediprobe Research Inc., London, Ontario, Canada, told this news organization.
“Our patients with male pattern baldness need to be educated about the negative effects of smoking, given that this condition can have a profound negative psychological impact on those who suffer from it,” he said.
The study was published online in the Journal of Cosmetic Dermatology.
Analyzing Smoking’s Effects
Smoking generally has been accepted as a risk factor for the development and progression of AGA or the most common form of hair loss. The research evidence on this association has been inconsistent, however, the authors wrote.
The investigators conducted a review and meta-analysis of eight observational studies to understand the links between smoking and AGA. Ever-smokers were defined as current and former smokers.
Overall, based on six studies, men who have ever smoked are 1.8 times more likely (P < .05) to develop AGA.
Based on two studies, men who smoke 10 or more cigarettes daily are about twice as likely (P < .05) to develop AGA than those who smoke up to 10 cigarettes per day.
Based on four studies, ever smoking is associated with 1.3 times higher odds of AGA progressing from mild (ie, Norwood-Hamilton stages I-III) to more severe (stages IV-VII) than among those who have never smoked.
Based on two studies, there’s no association between AGA progression and smoking intensity (as defined as smoking up to 20 cigarettes daily vs smoking 20 or more cigarettes per day).
“Though our pooled analysis found no significant association between smoking intensity and severity of male AGA, a positive correlation may exist and be detected through an analysis that is statistically better powered,” said Dr. Gupta.
The investigators noted the limitations of their analysis, such as its reliance on observational studies and its lack of data about nicotine levels, smoking intensity, and smoking cessation among study participants.
Additional studies are needed to better understand the links between smoking and hair loss, said Dr. Gupta, as well as the effects of smoking cessation.
Improving Practice and Research
Commenting on the findings for this news organization, Arash Babadjouni, MD, a dermatologist at Midwestern University, Glendale, Arizona, said, “Smoking is not only a preventable cause of significant systemic disease but also affects the follicular growth cycle and fiber pigmentation. The prevalence of hair loss and premature hair graying is higher in smokers than nonsmokers.”
Dr. Babadjouni, who wasn’t involved with this study, has researched the associations between smoking and hair loss and premature hair graying.
“Evidence of this association can be used to clinically promote smoking cessation and emphasize the consequences of smoking on hair,” he said. “Smoking status should be assessed in patients who are presenting to their dermatologist and physicians alike for evaluation of alopecia and premature hair graying.”
The study was conducted without outside funding, and the authors declared no conflicts of interest. Dr. Babadjouni reported no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM THE JOURNAL OF COSMETIC DERMATOLOGY
How a Simple Urine Test Could Reveal Early-Stage Lung Cancer
Lung cancer is the deadliest cancer in the world, largely because so many patients are diagnosed late.
Screening more patients could help, yet screening rates remain critically low. In the United States, only about 6% of eligible people get screened , according to the American Lung Association. Contrast that with screening rates for breast, cervical, and colorectal cancer, which all top 70%.
But what if lung cancer detection was as simple as taking a puff on an inhaler and following up with a urine test?
, according to research published this month in Science Advances. If the sensors spot these proteins, they produce a signal in the urine that can be detected with a paper test strip.
“It’s a more complex version of a pregnancy test, but it’s very simple to use,” said Qian Zhong, PhD, an MIT researcher and co-lead author of the study.
Currently, the only recommended screening test for lung cancer is low-dose CT. But not everyone has easy access to screening facilities, said the other co-lead author Edward Tan, PhD, a former MIT postdoc and currently a scientist at the biotech company Prime Medicine, Cambridge, Massachusetts.
“Our focus is to provide an alternative for the early detection of lung cancer that does not rely on resource-intensive infrastructure,” said Dr. Tan. “Most developing countries don’t have such resources” — and residents in some parts of the United States don’t have easy access, either, he said.
How It Works
The sensors are polymer nanoparticles coated in DNA barcodes, short DNA sequences that are unique and easy to identify. The researchers engineered the particles to be targeted by protease enzymes linked to stage I lung adenocarcinoma. Upon contact, the proteases cleave off the barcodes, which make their way into the bloodstream and are excreted in urine. A test strip can detect them, revealing results about 20 minutes from the time it’s dipped.
The researchers tested this system in mice genetically engineered to develop human-like lung tumors. Using aerosol nebulizers, they delivered 20 sensors to mice with the equivalent of stage I or II cancer. Using a machine learning algorithm, they identified the four most accurate sensors. With 100% specificity, those four sensors exhibited sensitivity of 84.6%.
“One advantage of using inhalation is that it’s noninvasive, and another advantage is that it distributes across the lung quite homogeneously,” said Dr. Tan. The time from inhalation to detection is also relatively fast — in mice, the whole process took about 2 hours, and Dr. Zhong speculated that it would not be much longer in humans.
Other Applications and Challenges
An injectable version of this technology, also developed at MIT, has already been tested in a phase 1 clinical trial for diagnosing liver cancer and nonalcoholic steatohepatitis. The injection also works in tandem with a urine test, the researchers showed in 2021. According to Tan, his research group (led by Sangeeta Bhatia, MD, PhD) was the first to describe this type of technology to screen for diseases.
The lab is also working toward using inhalable sensors to distinguish between viral, bacterial, and fungal pneumonia. And the technology could also be used to diagnose other lung conditions like asthma and chronic obstructive pulmonary disease, Dr. Tan said.
The tech is certainly “innovative,” remarked Gaetano Rocco, MD, a thoracic surgeon and lung cancer researcher at Memorial Sloan Kettering Cancer Center, Basking Ridge, New Jersey, who was not involved in the study.
Still, challenges may arise when applying it to people. Many factors are involved in regulating fluid volume, potentially interfering with the ability to detect the compounds in the urine, Rocco said. Diet, hydration, drug interference, renal function, and some chronic diseases could all limit effectiveness.
Another challenge: Human cancer can be more heterogeneous (containing different kinds of cancer cells), so four sensors may not be enough, Zhong said. He and colleagues are beginning to analyze human biopsy samples to see whether the same sensors that worked in mice would also work in humans. If all goes well, they hope to do studies on humans or nonhuman primates.
A version of this article appeared on Medscape.com.
Lung cancer is the deadliest cancer in the world, largely because so many patients are diagnosed late.
Screening more patients could help, yet screening rates remain critically low. In the United States, only about 6% of eligible people get screened , according to the American Lung Association. Contrast that with screening rates for breast, cervical, and colorectal cancer, which all top 70%.
But what if lung cancer detection was as simple as taking a puff on an inhaler and following up with a urine test?
, according to research published this month in Science Advances. If the sensors spot these proteins, they produce a signal in the urine that can be detected with a paper test strip.
“It’s a more complex version of a pregnancy test, but it’s very simple to use,” said Qian Zhong, PhD, an MIT researcher and co-lead author of the study.
Currently, the only recommended screening test for lung cancer is low-dose CT. But not everyone has easy access to screening facilities, said the other co-lead author Edward Tan, PhD, a former MIT postdoc and currently a scientist at the biotech company Prime Medicine, Cambridge, Massachusetts.
“Our focus is to provide an alternative for the early detection of lung cancer that does not rely on resource-intensive infrastructure,” said Dr. Tan. “Most developing countries don’t have such resources” — and residents in some parts of the United States don’t have easy access, either, he said.
How It Works
The sensors are polymer nanoparticles coated in DNA barcodes, short DNA sequences that are unique and easy to identify. The researchers engineered the particles to be targeted by protease enzymes linked to stage I lung adenocarcinoma. Upon contact, the proteases cleave off the barcodes, which make their way into the bloodstream and are excreted in urine. A test strip can detect them, revealing results about 20 minutes from the time it’s dipped.
The researchers tested this system in mice genetically engineered to develop human-like lung tumors. Using aerosol nebulizers, they delivered 20 sensors to mice with the equivalent of stage I or II cancer. Using a machine learning algorithm, they identified the four most accurate sensors. With 100% specificity, those four sensors exhibited sensitivity of 84.6%.
“One advantage of using inhalation is that it’s noninvasive, and another advantage is that it distributes across the lung quite homogeneously,” said Dr. Tan. The time from inhalation to detection is also relatively fast — in mice, the whole process took about 2 hours, and Dr. Zhong speculated that it would not be much longer in humans.
Other Applications and Challenges
An injectable version of this technology, also developed at MIT, has already been tested in a phase 1 clinical trial for diagnosing liver cancer and nonalcoholic steatohepatitis. The injection also works in tandem with a urine test, the researchers showed in 2021. According to Tan, his research group (led by Sangeeta Bhatia, MD, PhD) was the first to describe this type of technology to screen for diseases.
The lab is also working toward using inhalable sensors to distinguish between viral, bacterial, and fungal pneumonia. And the technology could also be used to diagnose other lung conditions like asthma and chronic obstructive pulmonary disease, Dr. Tan said.
The tech is certainly “innovative,” remarked Gaetano Rocco, MD, a thoracic surgeon and lung cancer researcher at Memorial Sloan Kettering Cancer Center, Basking Ridge, New Jersey, who was not involved in the study.
Still, challenges may arise when applying it to people. Many factors are involved in regulating fluid volume, potentially interfering with the ability to detect the compounds in the urine, Rocco said. Diet, hydration, drug interference, renal function, and some chronic diseases could all limit effectiveness.
Another challenge: Human cancer can be more heterogeneous (containing different kinds of cancer cells), so four sensors may not be enough, Zhong said. He and colleagues are beginning to analyze human biopsy samples to see whether the same sensors that worked in mice would also work in humans. If all goes well, they hope to do studies on humans or nonhuman primates.
A version of this article appeared on Medscape.com.
Lung cancer is the deadliest cancer in the world, largely because so many patients are diagnosed late.
Screening more patients could help, yet screening rates remain critically low. In the United States, only about 6% of eligible people get screened , according to the American Lung Association. Contrast that with screening rates for breast, cervical, and colorectal cancer, which all top 70%.
But what if lung cancer detection was as simple as taking a puff on an inhaler and following up with a urine test?
, according to research published this month in Science Advances. If the sensors spot these proteins, they produce a signal in the urine that can be detected with a paper test strip.
“It’s a more complex version of a pregnancy test, but it’s very simple to use,” said Qian Zhong, PhD, an MIT researcher and co-lead author of the study.
Currently, the only recommended screening test for lung cancer is low-dose CT. But not everyone has easy access to screening facilities, said the other co-lead author Edward Tan, PhD, a former MIT postdoc and currently a scientist at the biotech company Prime Medicine, Cambridge, Massachusetts.
“Our focus is to provide an alternative for the early detection of lung cancer that does not rely on resource-intensive infrastructure,” said Dr. Tan. “Most developing countries don’t have such resources” — and residents in some parts of the United States don’t have easy access, either, he said.
How It Works
The sensors are polymer nanoparticles coated in DNA barcodes, short DNA sequences that are unique and easy to identify. The researchers engineered the particles to be targeted by protease enzymes linked to stage I lung adenocarcinoma. Upon contact, the proteases cleave off the barcodes, which make their way into the bloodstream and are excreted in urine. A test strip can detect them, revealing results about 20 minutes from the time it’s dipped.
The researchers tested this system in mice genetically engineered to develop human-like lung tumors. Using aerosol nebulizers, they delivered 20 sensors to mice with the equivalent of stage I or II cancer. Using a machine learning algorithm, they identified the four most accurate sensors. With 100% specificity, those four sensors exhibited sensitivity of 84.6%.
“One advantage of using inhalation is that it’s noninvasive, and another advantage is that it distributes across the lung quite homogeneously,” said Dr. Tan. The time from inhalation to detection is also relatively fast — in mice, the whole process took about 2 hours, and Dr. Zhong speculated that it would not be much longer in humans.
Other Applications and Challenges
An injectable version of this technology, also developed at MIT, has already been tested in a phase 1 clinical trial for diagnosing liver cancer and nonalcoholic steatohepatitis. The injection also works in tandem with a urine test, the researchers showed in 2021. According to Tan, his research group (led by Sangeeta Bhatia, MD, PhD) was the first to describe this type of technology to screen for diseases.
The lab is also working toward using inhalable sensors to distinguish between viral, bacterial, and fungal pneumonia. And the technology could also be used to diagnose other lung conditions like asthma and chronic obstructive pulmonary disease, Dr. Tan said.
The tech is certainly “innovative,” remarked Gaetano Rocco, MD, a thoracic surgeon and lung cancer researcher at Memorial Sloan Kettering Cancer Center, Basking Ridge, New Jersey, who was not involved in the study.
Still, challenges may arise when applying it to people. Many factors are involved in regulating fluid volume, potentially interfering with the ability to detect the compounds in the urine, Rocco said. Diet, hydration, drug interference, renal function, and some chronic diseases could all limit effectiveness.
Another challenge: Human cancer can be more heterogeneous (containing different kinds of cancer cells), so four sensors may not be enough, Zhong said. He and colleagues are beginning to analyze human biopsy samples to see whether the same sensors that worked in mice would also work in humans. If all goes well, they hope to do studies on humans or nonhuman primates.
A version of this article appeared on Medscape.com.
Radiation Oncologists Fight for Payment Reform Amid Cuts
The American Society for Radiation Oncology (ASTRO) recently announced its partnership with three other groups — the American College of Radiation Oncology, the American College of Radiology, and the American Society of Clinical Oncology — to change how the specialty is paid for services.
Over the past decade, radiation oncologists have seen a 23% drop in Medicare reimbursement for radiation therapy services, with more cuts to come, according to a press release from ASTRO.
Traditionally, Medicare has reimbursed on the basis of the fraction of radiation delivered. But with moves toward hypofractionated regimens, deescalated therapy, and other changes in the field, reimbursement has continued to dwindle.
The cuts have led to practice consolidation and closures that threaten patient access especially in rural and underserved areas, a spokesperson for the group told this news organization.
To reverse this trend, ASTRO recently proposed the Radiation Oncology Case Rate program, a legislative initiative to base reimbursements on patient volumes instead of fractions delivered.
ASTRO is currently drafting a congressional bill to change the current payment structure, which “has become untenable,” the spokesperson said.
A version of this article appeared on Medscape.com.
The American Society for Radiation Oncology (ASTRO) recently announced its partnership with three other groups — the American College of Radiation Oncology, the American College of Radiology, and the American Society of Clinical Oncology — to change how the specialty is paid for services.
Over the past decade, radiation oncologists have seen a 23% drop in Medicare reimbursement for radiation therapy services, with more cuts to come, according to a press release from ASTRO.
Traditionally, Medicare has reimbursed on the basis of the fraction of radiation delivered. But with moves toward hypofractionated regimens, deescalated therapy, and other changes in the field, reimbursement has continued to dwindle.
The cuts have led to practice consolidation and closures that threaten patient access especially in rural and underserved areas, a spokesperson for the group told this news organization.
To reverse this trend, ASTRO recently proposed the Radiation Oncology Case Rate program, a legislative initiative to base reimbursements on patient volumes instead of fractions delivered.
ASTRO is currently drafting a congressional bill to change the current payment structure, which “has become untenable,” the spokesperson said.
A version of this article appeared on Medscape.com.
The American Society for Radiation Oncology (ASTRO) recently announced its partnership with three other groups — the American College of Radiation Oncology, the American College of Radiology, and the American Society of Clinical Oncology — to change how the specialty is paid for services.
Over the past decade, radiation oncologists have seen a 23% drop in Medicare reimbursement for radiation therapy services, with more cuts to come, according to a press release from ASTRO.
Traditionally, Medicare has reimbursed on the basis of the fraction of radiation delivered. But with moves toward hypofractionated regimens, deescalated therapy, and other changes in the field, reimbursement has continued to dwindle.
The cuts have led to practice consolidation and closures that threaten patient access especially in rural and underserved areas, a spokesperson for the group told this news organization.
To reverse this trend, ASTRO recently proposed the Radiation Oncology Case Rate program, a legislative initiative to base reimbursements on patient volumes instead of fractions delivered.
ASTRO is currently drafting a congressional bill to change the current payment structure, which “has become untenable,” the spokesperson said.
A version of this article appeared on Medscape.com.
Flovent Brand Discontinuation Likely Smooth for Many, Difficult for Some
A recent alert posted on the Asthma and Allergy Foundation of America (AAFA) website blog announced, “Flovent HFA and Flovent Diskus Asthma Medicine Being Discontinued.” A further heading positioned next to images of the two red inhaler devices stated: “Generic versions of the same medicines and devices are available but you need to check your insurance.” While few, it is generally thought, will have trouble finding suitable alternatives, the warning captured the reality descending upon some individual asthma sufferers whose insurance coverage may need tweaking at the very least, or at worst may be lacking.
The AAFA blog included a GSK (GlaxoSmithKline) November 2023 statement to AAFA regarding the brand name FLOVENT discontinuation. It noted the launch of an authorized Flovent HFA (fluticasone propionate inhalation aerosol) generic in May 2022 and a planned (October 2023) launch of an authorized generic for Flovent Diskus (fluticasone propionate inhalation powder) as “part of our commitment to be ambitious for patients.” The GSK statement continues: “These GSK manufactured authorized generics will provide patients in the US with potentially lower cost alternatives of these medically important products. We recognize that patients have a number of options in the therapeutic area and therefore remain committed to ensuring the affordability of our medicines.”
GSK will continue to manufacture the authorized generics, but they will be distributed by Prasco LLC.
Medicaid Rebate Cap Removed
As a Forbes article on January 3, 2024, by Joshua Cohen (“New Medicaid Rebate Rule Causes Problems For Asthma Patients On Flovent”) points out, the Flovent January 1, 2024, discontinuation coincided with the removal of the Medicaid rebate cap (American Rescue Plan Medicaid Drug Rebate Program) targeting manufacturers who had previously raised medication prices at rates higher than the inflation rate. The Forbes story notes GoodRx data showing a 47% increase in Flovent price since 2014. The implication is that drug manufacturers could be forced to sell such a drug to Medicaid at a loss because of the rebate cap removal. An authorized generic introduced to the market at a lower price under a private label with no price history, however, would not be subject to the higher Medicaid rebates.
Motivation considerations aside, the fallout for patients may or may not include a lower cost alternative. The authorized generic versions of Flovent HFA and Flovent Diskus are identical to the branded products with respect to the drugs and the devices. The GSK statement expressed hope that most insurance plans will replace the brand name with the authorized generic. The possibility persists, however, that there may be some that do not — resulting in a need to find the right substitute and/or higher out-of-pocket costs.
“Even though some patients may experience some disruption initially in their prescriptions,” Diego J. Maselli, MD, professor and chief, division of pulmonary diseases and critical care, UT Health at San Antonio, Texas, said in an interview, “fortunately, there are quite a few alternatives, and we don’t anticipate significant problems. It will be a wrinkle for some of the patients with regard to coverage, but there are definitely many alternatives that can provide good enough treatment for them.”
Similar alternative inhalers?
The alternatives have their specific properties and qualities, but the vast majority of experts, Dr. Maselli said, consider them to be very similar.
For CAREMARK CVS, a major pharmaceutical benefits manager, the preferred Flovent substitute is Pulmicort Flexhaler, a dry-powder inhaler that contains budesonide rather than fluticasone. While Flovent HF is a metered dose inhaler with a propellant, the Pulmicort device contains budesonide as a dry powder and requires activation through inhalation, which can be problematic for young children, AAFA CEO Kenneth Mendez said in an interview. To address that issue, he said, CVS Caremark is covering the authorized fluticasone metered dose inhaler generic for children under 6 years old. “Those individuals 6 years and older with severe asthma who can’t breathe deeply enough to get the medicine into their lungs will have to work with their doctors to apply for a formulary exception. And that’s a complicated process,” Mr. Mendez observed. “And it can take some time,” he added.
Another key issue highlighted here, he emphasized, is “how complicated this system is.” The U.S. drug pricing ecosystem involves multiple manufacturers, pharmacy benefit managers, insurance companies and their various plans, and federal policies potentially creating situations that may reduce access to critical medicines for patients, Mr. Mendez said. “Some people will be scurrying and scrambling to try to get coverage. The scope of the impact is actually unknown, but we’re going to find out now. As a nonprofit, we monitor social media and we’re listening closely.”
AAFA’s further concern is the rising costs of asthma medications. “It’s the number one thing we hear about as a patient organization,” Mr. Mendez said. On January 9, 2024, AAFA issued a press release praising the previous day’s news item from the U.S. Senate Committee on Health, Education, Labor & Pensions (“Chairman Sanders, Baldwin, Luján, Markey Launch HELP Committee Investigation into Efforts by Pharmaceutical Companies to Manipulate the Price of Asthma Inhalers). In it, Senator Bernie Sanders pointed to the more than 12-fold higher cost in the United States compared with the United Kingdom for GSK’s inhaler combining fluticasone and a beta2 agonist. The Senate HELP Committee has sent letters to the CEOs of the four major inhaler manufacturers (AstraZeneca, Boehringer Ingelheim, GSK, and Teva), stating: “These prices force patients, especially the uninsured and underinsured, to ration doses or abandon their prescriptions altogether. The results are predictable and devastating.”
High costs of inhalers could lead to rationing
AAFA research, the AAFA press release states, confirms that when asthma medicine costs become a barrier to treatment, people with asthma ration or discontinue medication use. The release also includes Mr. Mendez’s plea for a broad national conversation. “We are hopeful the HELP Committee investigation will lead to a national conversation about asthma drug costs and produce action that breaks down barriers to affordable treatment for people with asthma. The bottom line is that cost drives access. We understand the barriers, now it is important to move toward solutions.”
AAFA’s blog advises that when an individual’s insurance plan does not cover the authorized generic and does not offer a formulary exception, other inhaler options include ArmonAir Digihaler and Arnuity Ellipta. Because these are not identical to the authorized generics, individuals should check with their doctors regarding available doses and inhaler types and, if necessary, request training on inhaler use.
“It is really important for people with asthma to continue their asthma control medicines, especially during respiratory illness season.” AAFA urges individuals with asthma who are currently Flovent users to check with their doctors or pharmacists about the best next steps for them.
A recent alert posted on the Asthma and Allergy Foundation of America (AAFA) website blog announced, “Flovent HFA and Flovent Diskus Asthma Medicine Being Discontinued.” A further heading positioned next to images of the two red inhaler devices stated: “Generic versions of the same medicines and devices are available but you need to check your insurance.” While few, it is generally thought, will have trouble finding suitable alternatives, the warning captured the reality descending upon some individual asthma sufferers whose insurance coverage may need tweaking at the very least, or at worst may be lacking.
The AAFA blog included a GSK (GlaxoSmithKline) November 2023 statement to AAFA regarding the brand name FLOVENT discontinuation. It noted the launch of an authorized Flovent HFA (fluticasone propionate inhalation aerosol) generic in May 2022 and a planned (October 2023) launch of an authorized generic for Flovent Diskus (fluticasone propionate inhalation powder) as “part of our commitment to be ambitious for patients.” The GSK statement continues: “These GSK manufactured authorized generics will provide patients in the US with potentially lower cost alternatives of these medically important products. We recognize that patients have a number of options in the therapeutic area and therefore remain committed to ensuring the affordability of our medicines.”
GSK will continue to manufacture the authorized generics, but they will be distributed by Prasco LLC.
Medicaid Rebate Cap Removed
As a Forbes article on January 3, 2024, by Joshua Cohen (“New Medicaid Rebate Rule Causes Problems For Asthma Patients On Flovent”) points out, the Flovent January 1, 2024, discontinuation coincided with the removal of the Medicaid rebate cap (American Rescue Plan Medicaid Drug Rebate Program) targeting manufacturers who had previously raised medication prices at rates higher than the inflation rate. The Forbes story notes GoodRx data showing a 47% increase in Flovent price since 2014. The implication is that drug manufacturers could be forced to sell such a drug to Medicaid at a loss because of the rebate cap removal. An authorized generic introduced to the market at a lower price under a private label with no price history, however, would not be subject to the higher Medicaid rebates.
Motivation considerations aside, the fallout for patients may or may not include a lower cost alternative. The authorized generic versions of Flovent HFA and Flovent Diskus are identical to the branded products with respect to the drugs and the devices. The GSK statement expressed hope that most insurance plans will replace the brand name with the authorized generic. The possibility persists, however, that there may be some that do not — resulting in a need to find the right substitute and/or higher out-of-pocket costs.
“Even though some patients may experience some disruption initially in their prescriptions,” Diego J. Maselli, MD, professor and chief, division of pulmonary diseases and critical care, UT Health at San Antonio, Texas, said in an interview, “fortunately, there are quite a few alternatives, and we don’t anticipate significant problems. It will be a wrinkle for some of the patients with regard to coverage, but there are definitely many alternatives that can provide good enough treatment for them.”
Similar alternative inhalers?
The alternatives have their specific properties and qualities, but the vast majority of experts, Dr. Maselli said, consider them to be very similar.
For CAREMARK CVS, a major pharmaceutical benefits manager, the preferred Flovent substitute is Pulmicort Flexhaler, a dry-powder inhaler that contains budesonide rather than fluticasone. While Flovent HF is a metered dose inhaler with a propellant, the Pulmicort device contains budesonide as a dry powder and requires activation through inhalation, which can be problematic for young children, AAFA CEO Kenneth Mendez said in an interview. To address that issue, he said, CVS Caremark is covering the authorized fluticasone metered dose inhaler generic for children under 6 years old. “Those individuals 6 years and older with severe asthma who can’t breathe deeply enough to get the medicine into their lungs will have to work with their doctors to apply for a formulary exception. And that’s a complicated process,” Mr. Mendez observed. “And it can take some time,” he added.
Another key issue highlighted here, he emphasized, is “how complicated this system is.” The U.S. drug pricing ecosystem involves multiple manufacturers, pharmacy benefit managers, insurance companies and their various plans, and federal policies potentially creating situations that may reduce access to critical medicines for patients, Mr. Mendez said. “Some people will be scurrying and scrambling to try to get coverage. The scope of the impact is actually unknown, but we’re going to find out now. As a nonprofit, we monitor social media and we’re listening closely.”
AAFA’s further concern is the rising costs of asthma medications. “It’s the number one thing we hear about as a patient organization,” Mr. Mendez said. On January 9, 2024, AAFA issued a press release praising the previous day’s news item from the U.S. Senate Committee on Health, Education, Labor & Pensions (“Chairman Sanders, Baldwin, Luján, Markey Launch HELP Committee Investigation into Efforts by Pharmaceutical Companies to Manipulate the Price of Asthma Inhalers). In it, Senator Bernie Sanders pointed to the more than 12-fold higher cost in the United States compared with the United Kingdom for GSK’s inhaler combining fluticasone and a beta2 agonist. The Senate HELP Committee has sent letters to the CEOs of the four major inhaler manufacturers (AstraZeneca, Boehringer Ingelheim, GSK, and Teva), stating: “These prices force patients, especially the uninsured and underinsured, to ration doses or abandon their prescriptions altogether. The results are predictable and devastating.”
High costs of inhalers could lead to rationing
AAFA research, the AAFA press release states, confirms that when asthma medicine costs become a barrier to treatment, people with asthma ration or discontinue medication use. The release also includes Mr. Mendez’s plea for a broad national conversation. “We are hopeful the HELP Committee investigation will lead to a national conversation about asthma drug costs and produce action that breaks down barriers to affordable treatment for people with asthma. The bottom line is that cost drives access. We understand the barriers, now it is important to move toward solutions.”
AAFA’s blog advises that when an individual’s insurance plan does not cover the authorized generic and does not offer a formulary exception, other inhaler options include ArmonAir Digihaler and Arnuity Ellipta. Because these are not identical to the authorized generics, individuals should check with their doctors regarding available doses and inhaler types and, if necessary, request training on inhaler use.
“It is really important for people with asthma to continue their asthma control medicines, especially during respiratory illness season.” AAFA urges individuals with asthma who are currently Flovent users to check with their doctors or pharmacists about the best next steps for them.
A recent alert posted on the Asthma and Allergy Foundation of America (AAFA) website blog announced, “Flovent HFA and Flovent Diskus Asthma Medicine Being Discontinued.” A further heading positioned next to images of the two red inhaler devices stated: “Generic versions of the same medicines and devices are available but you need to check your insurance.” While few, it is generally thought, will have trouble finding suitable alternatives, the warning captured the reality descending upon some individual asthma sufferers whose insurance coverage may need tweaking at the very least, or at worst may be lacking.
The AAFA blog included a GSK (GlaxoSmithKline) November 2023 statement to AAFA regarding the brand name FLOVENT discontinuation. It noted the launch of an authorized Flovent HFA (fluticasone propionate inhalation aerosol) generic in May 2022 and a planned (October 2023) launch of an authorized generic for Flovent Diskus (fluticasone propionate inhalation powder) as “part of our commitment to be ambitious for patients.” The GSK statement continues: “These GSK manufactured authorized generics will provide patients in the US with potentially lower cost alternatives of these medically important products. We recognize that patients have a number of options in the therapeutic area and therefore remain committed to ensuring the affordability of our medicines.”
GSK will continue to manufacture the authorized generics, but they will be distributed by Prasco LLC.
Medicaid Rebate Cap Removed
As a Forbes article on January 3, 2024, by Joshua Cohen (“New Medicaid Rebate Rule Causes Problems For Asthma Patients On Flovent”) points out, the Flovent January 1, 2024, discontinuation coincided with the removal of the Medicaid rebate cap (American Rescue Plan Medicaid Drug Rebate Program) targeting manufacturers who had previously raised medication prices at rates higher than the inflation rate. The Forbes story notes GoodRx data showing a 47% increase in Flovent price since 2014. The implication is that drug manufacturers could be forced to sell such a drug to Medicaid at a loss because of the rebate cap removal. An authorized generic introduced to the market at a lower price under a private label with no price history, however, would not be subject to the higher Medicaid rebates.
Motivation considerations aside, the fallout for patients may or may not include a lower cost alternative. The authorized generic versions of Flovent HFA and Flovent Diskus are identical to the branded products with respect to the drugs and the devices. The GSK statement expressed hope that most insurance plans will replace the brand name with the authorized generic. The possibility persists, however, that there may be some that do not — resulting in a need to find the right substitute and/or higher out-of-pocket costs.
“Even though some patients may experience some disruption initially in their prescriptions,” Diego J. Maselli, MD, professor and chief, division of pulmonary diseases and critical care, UT Health at San Antonio, Texas, said in an interview, “fortunately, there are quite a few alternatives, and we don’t anticipate significant problems. It will be a wrinkle for some of the patients with regard to coverage, but there are definitely many alternatives that can provide good enough treatment for them.”
Similar alternative inhalers?
The alternatives have their specific properties and qualities, but the vast majority of experts, Dr. Maselli said, consider them to be very similar.
For CAREMARK CVS, a major pharmaceutical benefits manager, the preferred Flovent substitute is Pulmicort Flexhaler, a dry-powder inhaler that contains budesonide rather than fluticasone. While Flovent HF is a metered dose inhaler with a propellant, the Pulmicort device contains budesonide as a dry powder and requires activation through inhalation, which can be problematic for young children, AAFA CEO Kenneth Mendez said in an interview. To address that issue, he said, CVS Caremark is covering the authorized fluticasone metered dose inhaler generic for children under 6 years old. “Those individuals 6 years and older with severe asthma who can’t breathe deeply enough to get the medicine into their lungs will have to work with their doctors to apply for a formulary exception. And that’s a complicated process,” Mr. Mendez observed. “And it can take some time,” he added.
Another key issue highlighted here, he emphasized, is “how complicated this system is.” The U.S. drug pricing ecosystem involves multiple manufacturers, pharmacy benefit managers, insurance companies and their various plans, and federal policies potentially creating situations that may reduce access to critical medicines for patients, Mr. Mendez said. “Some people will be scurrying and scrambling to try to get coverage. The scope of the impact is actually unknown, but we’re going to find out now. As a nonprofit, we monitor social media and we’re listening closely.”
AAFA’s further concern is the rising costs of asthma medications. “It’s the number one thing we hear about as a patient organization,” Mr. Mendez said. On January 9, 2024, AAFA issued a press release praising the previous day’s news item from the U.S. Senate Committee on Health, Education, Labor & Pensions (“Chairman Sanders, Baldwin, Luján, Markey Launch HELP Committee Investigation into Efforts by Pharmaceutical Companies to Manipulate the Price of Asthma Inhalers). In it, Senator Bernie Sanders pointed to the more than 12-fold higher cost in the United States compared with the United Kingdom for GSK’s inhaler combining fluticasone and a beta2 agonist. The Senate HELP Committee has sent letters to the CEOs of the four major inhaler manufacturers (AstraZeneca, Boehringer Ingelheim, GSK, and Teva), stating: “These prices force patients, especially the uninsured and underinsured, to ration doses or abandon their prescriptions altogether. The results are predictable and devastating.”
High costs of inhalers could lead to rationing
AAFA research, the AAFA press release states, confirms that when asthma medicine costs become a barrier to treatment, people with asthma ration or discontinue medication use. The release also includes Mr. Mendez’s plea for a broad national conversation. “We are hopeful the HELP Committee investigation will lead to a national conversation about asthma drug costs and produce action that breaks down barriers to affordable treatment for people with asthma. The bottom line is that cost drives access. We understand the barriers, now it is important to move toward solutions.”
AAFA’s blog advises that when an individual’s insurance plan does not cover the authorized generic and does not offer a formulary exception, other inhaler options include ArmonAir Digihaler and Arnuity Ellipta. Because these are not identical to the authorized generics, individuals should check with their doctors regarding available doses and inhaler types and, if necessary, request training on inhaler use.
“It is really important for people with asthma to continue their asthma control medicines, especially during respiratory illness season.” AAFA urges individuals with asthma who are currently Flovent users to check with their doctors or pharmacists about the best next steps for them.
New Federal Rule for Prior Authorizations a ‘Major Win’ for Patients, Doctors
Physicians groups on January 17 hailed a new federal rule requiring health insurers to streamline and disclose more information about their prior authorization processes, saying it will improve patient care and reduce doctors’ administrative burden.
Health insurers participating in federal programs, including Medicare Advantage and Medicaid, must now respond to expedited prior authorization requests within 72 hours and other requests within 7 days under the long-awaited final rule, released on January 17 by the Centers for Medicare & Medicaid Services (CMS).
Insurers also must include their reasons for denying a prior authorization request and will be required to publicly release data on denial and approval rates for medical treatment. They’ll also need to give patients more information about their decisions to deny care. Insurers must comply with some of the rule’s provisions by January 2026 and others by January 2027.
The final rule “is an important step forward” toward the Medical Group Management Association’s goal of reducing the overall volume of prior authorization requests, said Anders Gilberg, the group’s senior vice president for government affairs, in a statement.
“Only then will medical groups find meaningful reprieve from these onerous, ill-intentioned administrative requirements that dangerously impede patient care,” Mr. Gilberg said.
Health insurers have long lobbied against increased regulation of prior authorization, arguing that it’s needed to rein in healthcare costs and prevent unnecessary treatment.
“We appreciate CMS’s announcement of enforcement discretion that will permit plans to use one standard, rather than mixing and matching, to reduce costs and speed implementation,” said America’s Health Insurance Plans, an insurers’ lobbying group, in an unsigned statement. “However, we must remember that the CMS rule is only half the picture; the Office of the Coordinator for Health Information Technology (ONC) should swiftly require vendors to build electronic prior authorization capabilities into the electronic health record so that providers can do their part, or plans will build a bridge to nowhere.”
The rule comes as health insurers have increasingly been criticized for onerous and time-consuming prior authorization procedures that physicians say unfairly delay or deny the medical treatment that their patients need. With federal legislation to rein in prior authorization overuse at a standstill, 30 states have introduced their own bills to address the problem. Regulators and lawsuits also have called attention to insurers’ increasing use of artificial intelligence and algorithms to deny claims without human review.
“Family physicians know firsthand how prior authorizations divert valuable time and resources away from direct patient care. We also know that these types of administrative requirements are driving physicians away from the workforce and worsening physician shortages,” said Steven P. Furr, MD, president of the American Academy of Family Physicians, in a statement praising the new rule.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, called the final rule “ a major win” for patients and physicians, adding that its requirements for health insurers to integrate their prior authorization procedures into physicians’ electronic health records systems will also help make “the current time-consuming, manual workflow” more efficient.
A version of this article first appeared on Medscape.com.
Physicians groups on January 17 hailed a new federal rule requiring health insurers to streamline and disclose more information about their prior authorization processes, saying it will improve patient care and reduce doctors’ administrative burden.
Health insurers participating in federal programs, including Medicare Advantage and Medicaid, must now respond to expedited prior authorization requests within 72 hours and other requests within 7 days under the long-awaited final rule, released on January 17 by the Centers for Medicare & Medicaid Services (CMS).
Insurers also must include their reasons for denying a prior authorization request and will be required to publicly release data on denial and approval rates for medical treatment. They’ll also need to give patients more information about their decisions to deny care. Insurers must comply with some of the rule’s provisions by January 2026 and others by January 2027.
The final rule “is an important step forward” toward the Medical Group Management Association’s goal of reducing the overall volume of prior authorization requests, said Anders Gilberg, the group’s senior vice president for government affairs, in a statement.
“Only then will medical groups find meaningful reprieve from these onerous, ill-intentioned administrative requirements that dangerously impede patient care,” Mr. Gilberg said.
Health insurers have long lobbied against increased regulation of prior authorization, arguing that it’s needed to rein in healthcare costs and prevent unnecessary treatment.
“We appreciate CMS’s announcement of enforcement discretion that will permit plans to use one standard, rather than mixing and matching, to reduce costs and speed implementation,” said America’s Health Insurance Plans, an insurers’ lobbying group, in an unsigned statement. “However, we must remember that the CMS rule is only half the picture; the Office of the Coordinator for Health Information Technology (ONC) should swiftly require vendors to build electronic prior authorization capabilities into the electronic health record so that providers can do their part, or plans will build a bridge to nowhere.”
The rule comes as health insurers have increasingly been criticized for onerous and time-consuming prior authorization procedures that physicians say unfairly delay or deny the medical treatment that their patients need. With federal legislation to rein in prior authorization overuse at a standstill, 30 states have introduced their own bills to address the problem. Regulators and lawsuits also have called attention to insurers’ increasing use of artificial intelligence and algorithms to deny claims without human review.
“Family physicians know firsthand how prior authorizations divert valuable time and resources away from direct patient care. We also know that these types of administrative requirements are driving physicians away from the workforce and worsening physician shortages,” said Steven P. Furr, MD, president of the American Academy of Family Physicians, in a statement praising the new rule.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, called the final rule “ a major win” for patients and physicians, adding that its requirements for health insurers to integrate their prior authorization procedures into physicians’ electronic health records systems will also help make “the current time-consuming, manual workflow” more efficient.
A version of this article first appeared on Medscape.com.
Physicians groups on January 17 hailed a new federal rule requiring health insurers to streamline and disclose more information about their prior authorization processes, saying it will improve patient care and reduce doctors’ administrative burden.
Health insurers participating in federal programs, including Medicare Advantage and Medicaid, must now respond to expedited prior authorization requests within 72 hours and other requests within 7 days under the long-awaited final rule, released on January 17 by the Centers for Medicare & Medicaid Services (CMS).
Insurers also must include their reasons for denying a prior authorization request and will be required to publicly release data on denial and approval rates for medical treatment. They’ll also need to give patients more information about their decisions to deny care. Insurers must comply with some of the rule’s provisions by January 2026 and others by January 2027.
The final rule “is an important step forward” toward the Medical Group Management Association’s goal of reducing the overall volume of prior authorization requests, said Anders Gilberg, the group’s senior vice president for government affairs, in a statement.
“Only then will medical groups find meaningful reprieve from these onerous, ill-intentioned administrative requirements that dangerously impede patient care,” Mr. Gilberg said.
Health insurers have long lobbied against increased regulation of prior authorization, arguing that it’s needed to rein in healthcare costs and prevent unnecessary treatment.
“We appreciate CMS’s announcement of enforcement discretion that will permit plans to use one standard, rather than mixing and matching, to reduce costs and speed implementation,” said America’s Health Insurance Plans, an insurers’ lobbying group, in an unsigned statement. “However, we must remember that the CMS rule is only half the picture; the Office of the Coordinator for Health Information Technology (ONC) should swiftly require vendors to build electronic prior authorization capabilities into the electronic health record so that providers can do their part, or plans will build a bridge to nowhere.”
The rule comes as health insurers have increasingly been criticized for onerous and time-consuming prior authorization procedures that physicians say unfairly delay or deny the medical treatment that their patients need. With federal legislation to rein in prior authorization overuse at a standstill, 30 states have introduced their own bills to address the problem. Regulators and lawsuits also have called attention to insurers’ increasing use of artificial intelligence and algorithms to deny claims without human review.
“Family physicians know firsthand how prior authorizations divert valuable time and resources away from direct patient care. We also know that these types of administrative requirements are driving physicians away from the workforce and worsening physician shortages,” said Steven P. Furr, MD, president of the American Academy of Family Physicians, in a statement praising the new rule.
Jesse M. Ehrenfeld, MD, MPH, president of the American Medical Association, called the final rule “ a major win” for patients and physicians, adding that its requirements for health insurers to integrate their prior authorization procedures into physicians’ electronic health records systems will also help make “the current time-consuming, manual workflow” more efficient.
A version of this article first appeared on Medscape.com.
Continued Caution Needed Combining Nitrates With ED Drugs
New research supports continued caution in prescribing a phosphodiesterase-5 inhibitor (PDE5i) to treat erectile dysfunction (ED) in men with heart disease using nitrate medications.
In a large Swedish population study of men with stable coronary artery disease (CAD), the combined use of a PDE5i and nitrates was associated with a higher risk for cardiovascular (CV) morbidity and mortality.
“According to current recommendations, PDE5i are contraindicated in patients taking organic nitrates; however, in clinical practice, both are commonly prescribed, and concomitant use has increased,” first author Ylva Trolle Lagerros, MD, PhD, with Karolinska Institutet, Stockholm, Sweden, told this news organization.
and weigh the benefits of the medication against the possible increased risk for cardiovascular morbidity and mortality given by this combination,” Dr. Lagerros said.
The study was published online in the Journal of the American College of Cardiology (JACC).
The researchers used the Swedish Patient Register and the Prescribed Drug Register to assess the association between PDE5i treatment and CV outcomes in men with stable CAD treated with nitrate medication.
Among 55,777 men with a history of previous myocardial infarction (MI) or coronary revascularization who had filled at least two nitrate prescriptions (sublingual, oral, or both), 5710 also had at least two filled prescriptions of a PDE5i.
In multivariate-adjusted analysis, the combined use of PDE5i treatment with nitrates was associated with an increased relative risk for all studied outcomes, including all-cause mortality, CV and non-CV mortality, MI, heart failure, cardiac revascularization (hazard ratio), and major adverse cardiovascular events.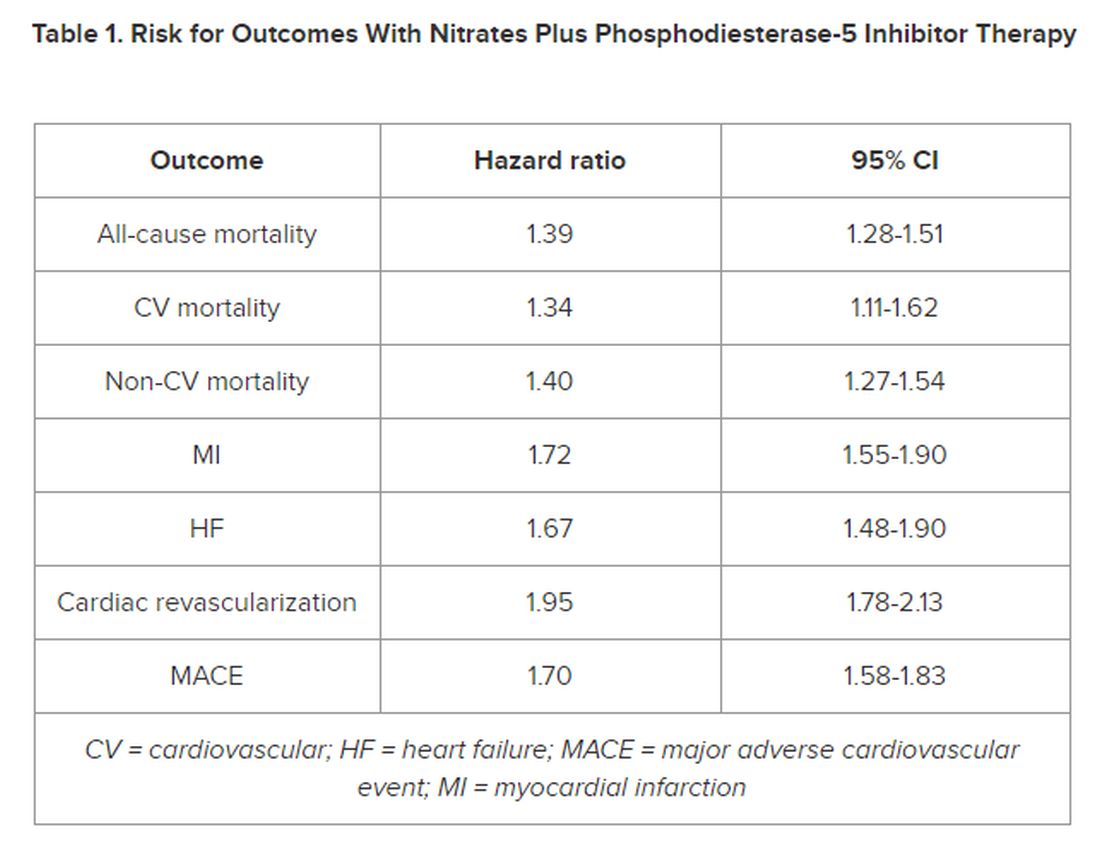
However, the number of events 28 days following a PDE5i prescription fill was “few, with lower incidence rates than in subjects taking nitrates only, indicating a low immediate risk for any event,” the authors noted in their article.
‘Common Bedfellows’
In a JACC editorial, Glenn N. Levine, MD, with Baylor College of Medicine, Houston, Texas, noted that, “ED and CAD are unfortunate, and all too common, bedfellows. But, as with most relationships, assuming proper precautions and care, they can coexist together for many years, perhaps even a lifetime.”
Dr. Levine noted that PDE5is are “reasonably safe” in most patients with stable CAD and only mild angina if not on chronic nitrate therapy. For those on chronic oral nitrate therapy, the use of PDE5is should continue to be regarded as “ill-advised at best and generally contraindicated.”
In some patients on oral nitrate therapy who want to use a PDE5i, particularly those who have undergone revascularization and have minimal or no angina, Dr. Levine said it may be reasonable to initiate a several-week trial of the nitrate therapy (or on a different class of antianginal therapy) and assess if the patient remains relatively angina-free.
In those patients with just rare exertional angina at generally higher levels of activity or those prescribed sublingual nitroglycerin “just in case,” it may be reasonable to prescribe PDE5i after a “clear and detailed” discussion with the patient of the risks for temporarily combining PDE5i and sublingual nitroglycerin.
Dr. Levine said these patients should be instructed not to take nitroglycerin within 24 hours of using a shorter-acting PDE5i and within 48 hours of using the longer-acting PDE5i tadalafil.
They should also be told to call 9-1-1 if angina develops during sexual intercourse and does not resolve upon cessation of such sexual activity, as well as to make medical personnel aware that they have recently used a PDE5i.
The study was funded by Region Stockholm, the Center for Innovative Medicine, and Karolinska Institutet. The researchers and editorial writer had declared no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
New research supports continued caution in prescribing a phosphodiesterase-5 inhibitor (PDE5i) to treat erectile dysfunction (ED) in men with heart disease using nitrate medications.
In a large Swedish population study of men with stable coronary artery disease (CAD), the combined use of a PDE5i and nitrates was associated with a higher risk for cardiovascular (CV) morbidity and mortality.
“According to current recommendations, PDE5i are contraindicated in patients taking organic nitrates; however, in clinical practice, both are commonly prescribed, and concomitant use has increased,” first author Ylva Trolle Lagerros, MD, PhD, with Karolinska Institutet, Stockholm, Sweden, told this news organization.
and weigh the benefits of the medication against the possible increased risk for cardiovascular morbidity and mortality given by this combination,” Dr. Lagerros said.
The study was published online in the Journal of the American College of Cardiology (JACC).
The researchers used the Swedish Patient Register and the Prescribed Drug Register to assess the association between PDE5i treatment and CV outcomes in men with stable CAD treated with nitrate medication.
Among 55,777 men with a history of previous myocardial infarction (MI) or coronary revascularization who had filled at least two nitrate prescriptions (sublingual, oral, or both), 5710 also had at least two filled prescriptions of a PDE5i.
In multivariate-adjusted analysis, the combined use of PDE5i treatment with nitrates was associated with an increased relative risk for all studied outcomes, including all-cause mortality, CV and non-CV mortality, MI, heart failure, cardiac revascularization (hazard ratio), and major adverse cardiovascular events.
However, the number of events 28 days following a PDE5i prescription fill was “few, with lower incidence rates than in subjects taking nitrates only, indicating a low immediate risk for any event,” the authors noted in their article.
‘Common Bedfellows’
In a JACC editorial, Glenn N. Levine, MD, with Baylor College of Medicine, Houston, Texas, noted that, “ED and CAD are unfortunate, and all too common, bedfellows. But, as with most relationships, assuming proper precautions and care, they can coexist together for many years, perhaps even a lifetime.”
Dr. Levine noted that PDE5is are “reasonably safe” in most patients with stable CAD and only mild angina if not on chronic nitrate therapy. For those on chronic oral nitrate therapy, the use of PDE5is should continue to be regarded as “ill-advised at best and generally contraindicated.”
In some patients on oral nitrate therapy who want to use a PDE5i, particularly those who have undergone revascularization and have minimal or no angina, Dr. Levine said it may be reasonable to initiate a several-week trial of the nitrate therapy (or on a different class of antianginal therapy) and assess if the patient remains relatively angina-free.
In those patients with just rare exertional angina at generally higher levels of activity or those prescribed sublingual nitroglycerin “just in case,” it may be reasonable to prescribe PDE5i after a “clear and detailed” discussion with the patient of the risks for temporarily combining PDE5i and sublingual nitroglycerin.
Dr. Levine said these patients should be instructed not to take nitroglycerin within 24 hours of using a shorter-acting PDE5i and within 48 hours of using the longer-acting PDE5i tadalafil.
They should also be told to call 9-1-1 if angina develops during sexual intercourse and does not resolve upon cessation of such sexual activity, as well as to make medical personnel aware that they have recently used a PDE5i.
The study was funded by Region Stockholm, the Center for Innovative Medicine, and Karolinska Institutet. The researchers and editorial writer had declared no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
New research supports continued caution in prescribing a phosphodiesterase-5 inhibitor (PDE5i) to treat erectile dysfunction (ED) in men with heart disease using nitrate medications.
In a large Swedish population study of men with stable coronary artery disease (CAD), the combined use of a PDE5i and nitrates was associated with a higher risk for cardiovascular (CV) morbidity and mortality.
“According to current recommendations, PDE5i are contraindicated in patients taking organic nitrates; however, in clinical practice, both are commonly prescribed, and concomitant use has increased,” first author Ylva Trolle Lagerros, MD, PhD, with Karolinska Institutet, Stockholm, Sweden, told this news organization.
and weigh the benefits of the medication against the possible increased risk for cardiovascular morbidity and mortality given by this combination,” Dr. Lagerros said.
The study was published online in the Journal of the American College of Cardiology (JACC).
The researchers used the Swedish Patient Register and the Prescribed Drug Register to assess the association between PDE5i treatment and CV outcomes in men with stable CAD treated with nitrate medication.
Among 55,777 men with a history of previous myocardial infarction (MI) or coronary revascularization who had filled at least two nitrate prescriptions (sublingual, oral, or both), 5710 also had at least two filled prescriptions of a PDE5i.
In multivariate-adjusted analysis, the combined use of PDE5i treatment with nitrates was associated with an increased relative risk for all studied outcomes, including all-cause mortality, CV and non-CV mortality, MI, heart failure, cardiac revascularization (hazard ratio), and major adverse cardiovascular events.
However, the number of events 28 days following a PDE5i prescription fill was “few, with lower incidence rates than in subjects taking nitrates only, indicating a low immediate risk for any event,” the authors noted in their article.
‘Common Bedfellows’
In a JACC editorial, Glenn N. Levine, MD, with Baylor College of Medicine, Houston, Texas, noted that, “ED and CAD are unfortunate, and all too common, bedfellows. But, as with most relationships, assuming proper precautions and care, they can coexist together for many years, perhaps even a lifetime.”
Dr. Levine noted that PDE5is are “reasonably safe” in most patients with stable CAD and only mild angina if not on chronic nitrate therapy. For those on chronic oral nitrate therapy, the use of PDE5is should continue to be regarded as “ill-advised at best and generally contraindicated.”
In some patients on oral nitrate therapy who want to use a PDE5i, particularly those who have undergone revascularization and have minimal or no angina, Dr. Levine said it may be reasonable to initiate a several-week trial of the nitrate therapy (or on a different class of antianginal therapy) and assess if the patient remains relatively angina-free.
In those patients with just rare exertional angina at generally higher levels of activity or those prescribed sublingual nitroglycerin “just in case,” it may be reasonable to prescribe PDE5i after a “clear and detailed” discussion with the patient of the risks for temporarily combining PDE5i and sublingual nitroglycerin.
Dr. Levine said these patients should be instructed not to take nitroglycerin within 24 hours of using a shorter-acting PDE5i and within 48 hours of using the longer-acting PDE5i tadalafil.
They should also be told to call 9-1-1 if angina develops during sexual intercourse and does not resolve upon cessation of such sexual activity, as well as to make medical personnel aware that they have recently used a PDE5i.
The study was funded by Region Stockholm, the Center for Innovative Medicine, and Karolinska Institutet. The researchers and editorial writer had declared no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
New Insights Into Mortality in Takotsubo Syndrome
TOPLINE:
Mortality in patients with takotsubo syndrome (TTS), sometimes called broken heart syndrome or stress-induced cardiomyopathy is substantially higher than that in the general population and comparable with that in patients having myocardial infarction (MI), results of a new case-control study showed. The rates of medication use are similar for TTS and MI, despite no current clinical trials or recommendations to guide such therapies, the authors noted.
METHODOLOGY:
- The study included 620 Scottish patients (mean age, 66 years; 91% women) with TTS, a potentially fatal condition that mimics MI, predominantly affects middle-aged women, and is often triggered by stress.
- The analysis also included two age-, sex-, and geographically matched control groups: Representative participants from the general Scottish population (1:4) and patients with acute MI (1:1).
- Using comprehensive national data sets, researchers extracted information for all three cohorts on prescribing of cardiovascular and noncardiovascular medications, including the duration of dispensing and causes of death, and clustered the major causes of death into 17 major groups.
- At a median follow-up of 5.5 years, there were 722 deaths (153 in patients with TTS, 195 in those with MI, and 374 in the general population cohort).
TAKEAWAY:
- and slightly lower than that in patients having MI (HR, 0.76; 95% CI, 0.62-0.94; P = .012), with cardiovascular causes, particularly heart failure, being the most strongly associated with TTS (HR, 2.47; 95% CI, 1.81-3.39; P < .0001 vs general population), followed by pulmonary causes. Noncardiovascular mortality was similar in TTS and MI.
- Prescription rates of cardiovascular and noncardiovascular medications were similar between patients with TTS and MI.
- The only cardiovascular therapy associated with lower mortality in patients with TTS was angiotensin-converting enzyme inhibitor or angiotensin receptor blocker therapy (P = .0056); in contrast, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers, antiplatelet agents, and statins were all associated with improved survival in patients with MI.
- Diuretics were associated with worse outcomes in both patients with TTS and MI, as was psychotropic therapy.
IN PRACTICE:
“These findings may help to lay the foundations for further exploration of potential mechanisms and treatments” for TTS, an “increasingly recognized and potentially fatal condition,” the authors concluded.
In an accompanying comment, Rodolfo Citro, MD, PHD, Cardiovascular and Thoracic Department, San Giovanni di Dio e Ruggi d’ Aragona University Hospital, Salerno, Italy, and colleagues said the authors should be commended for providing data on cardiovascular mortality “during one of the longest available follow-ups in TTS,” adding the study “suggests the importance of further research for more appropriate management of patients with acute and long-term TTS.”
SOURCE:
The research was led by Amelia E. Rudd, MSC, Aberdeen Cardiovascular and Diabetes Centre, University of Aberdeen and NHS Grampian, Aberdeen, Scotland. It was published online in the Journal of the American College of Cardiology.
LIMITATIONS:
Complete alignment of all variables related to clinical characteristics of patients with TTS and MI wasn’t feasible. During the study, TTS was still relatively unfamiliar to clinicians and underdiagnosed. As the study used a national data set of routinely collected data, not all desirable information was available, including indications of why drugs were prescribed or discontinued, which could have led to imprecise results. As the study used nonrandomized data, causality can’t be assumed.
DISCLOSURES:
Dr. Rudd had no relevant conflicts of interest. Study author Dana K. Dawson, Aberdeen Cardiovascular and Diabetes Centre, University of Aberdeen, Scotland, declared receiving the Chief Scientist Office Scotland award CGA-16-4 and the BHF Research Training Fellowship. Commentary authors had no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Mortality in patients with takotsubo syndrome (TTS), sometimes called broken heart syndrome or stress-induced cardiomyopathy is substantially higher than that in the general population and comparable with that in patients having myocardial infarction (MI), results of a new case-control study showed. The rates of medication use are similar for TTS and MI, despite no current clinical trials or recommendations to guide such therapies, the authors noted.
METHODOLOGY:
- The study included 620 Scottish patients (mean age, 66 years; 91% women) with TTS, a potentially fatal condition that mimics MI, predominantly affects middle-aged women, and is often triggered by stress.
- The analysis also included two age-, sex-, and geographically matched control groups: Representative participants from the general Scottish population (1:4) and patients with acute MI (1:1).
- Using comprehensive national data sets, researchers extracted information for all three cohorts on prescribing of cardiovascular and noncardiovascular medications, including the duration of dispensing and causes of death, and clustered the major causes of death into 17 major groups.
- At a median follow-up of 5.5 years, there were 722 deaths (153 in patients with TTS, 195 in those with MI, and 374 in the general population cohort).
TAKEAWAY:
- and slightly lower than that in patients having MI (HR, 0.76; 95% CI, 0.62-0.94; P = .012), with cardiovascular causes, particularly heart failure, being the most strongly associated with TTS (HR, 2.47; 95% CI, 1.81-3.39; P < .0001 vs general population), followed by pulmonary causes. Noncardiovascular mortality was similar in TTS and MI.
- Prescription rates of cardiovascular and noncardiovascular medications were similar between patients with TTS and MI.
- The only cardiovascular therapy associated with lower mortality in patients with TTS was angiotensin-converting enzyme inhibitor or angiotensin receptor blocker therapy (P = .0056); in contrast, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers, antiplatelet agents, and statins were all associated with improved survival in patients with MI.
- Diuretics were associated with worse outcomes in both patients with TTS and MI, as was psychotropic therapy.
IN PRACTICE:
“These findings may help to lay the foundations for further exploration of potential mechanisms and treatments” for TTS, an “increasingly recognized and potentially fatal condition,” the authors concluded.
In an accompanying comment, Rodolfo Citro, MD, PHD, Cardiovascular and Thoracic Department, San Giovanni di Dio e Ruggi d’ Aragona University Hospital, Salerno, Italy, and colleagues said the authors should be commended for providing data on cardiovascular mortality “during one of the longest available follow-ups in TTS,” adding the study “suggests the importance of further research for more appropriate management of patients with acute and long-term TTS.”
SOURCE:
The research was led by Amelia E. Rudd, MSC, Aberdeen Cardiovascular and Diabetes Centre, University of Aberdeen and NHS Grampian, Aberdeen, Scotland. It was published online in the Journal of the American College of Cardiology.
LIMITATIONS:
Complete alignment of all variables related to clinical characteristics of patients with TTS and MI wasn’t feasible. During the study, TTS was still relatively unfamiliar to clinicians and underdiagnosed. As the study used a national data set of routinely collected data, not all desirable information was available, including indications of why drugs were prescribed or discontinued, which could have led to imprecise results. As the study used nonrandomized data, causality can’t be assumed.
DISCLOSURES:
Dr. Rudd had no relevant conflicts of interest. Study author Dana K. Dawson, Aberdeen Cardiovascular and Diabetes Centre, University of Aberdeen, Scotland, declared receiving the Chief Scientist Office Scotland award CGA-16-4 and the BHF Research Training Fellowship. Commentary authors had no relevant conflicts of interest.
A version of this article appeared on Medscape.com.
TOPLINE:
Mortality in patients with takotsubo syndrome (TTS), sometimes called broken heart syndrome or stress-induced cardiomyopathy is substantially higher than that in the general population and comparable with that in patients having myocardial infarction (MI), results of a new case-control study showed. The rates of medication use are similar for TTS and MI, despite no current clinical trials or recommendations to guide such therapies, the authors noted.
METHODOLOGY:
- The study included 620 Scottish patients (mean age, 66 years; 91% women) with TTS, a potentially fatal condition that mimics MI, predominantly affects middle-aged women, and is often triggered by stress.
- The analysis also included two age-, sex-, and geographically matched control groups: Representative participants from the general Scottish population (1:4) and patients with acute MI (1:1).
- Using comprehensive national data sets, researchers extracted information for all three cohorts on prescribing of cardiovascular and noncardiovascular medications, including the duration of dispensing and causes of death, and clustered the major causes of death into 17 major groups.
- At a median follow-up of 5.5 years, there were 722 deaths (153 in patients with TTS, 195 in those with MI, and 374 in the general population cohort).
TAKEAWAY:
- and slightly lower than that in patients having MI (HR, 0.76; 95% CI, 0.62-0.94; P = .012), with cardiovascular causes, particularly heart failure, being the most strongly associated with TTS (HR, 2.47; 95% CI, 1.81-3.39; P < .0001 vs general population), followed by pulmonary causes. Noncardiovascular mortality was similar in TTS and MI.
- Prescription rates of cardiovascular and noncardiovascular medications were similar between patients with TTS and MI.
- The only cardiovascular therapy associated with lower mortality in patients with TTS was angiotensin-converting enzyme inhibitor or angiotensin receptor blocker therapy (P = .0056); in contrast, angiotensin-converting enzyme inhibitors, angiotensin receptor blockers, beta-blockers, antiplatelet agents, and statins were all associated with improved survival in patients with MI.
- Diuretics were associated with worse outcomes in both patients with TTS and MI, as was psychotropic therapy.
IN PRACTICE:
“These findings may help to lay the foundations for further exploration of potential mechanisms and treatments” for TTS, an “increasingly recognized and potentially fatal condition,” the authors concluded.
In an accompanying comment, Rodolfo Citro, MD, PHD, Cardiovascular and Thoracic Department, San Giovanni di Dio e Ruggi d’ Aragona University Hospital, Salerno, Italy, and colleagues said the authors should be commended for providing data on cardiovascular mortality “during one of the longest available follow-ups in TTS,” adding the study “suggests the importance of further research for more appropriate management of patients with acute and long-term TTS.”
SOURCE:
The research was led by Amelia E. Rudd, MSC, Aberdeen Cardiovascular and Diabetes Centre, University of Aberdeen and NHS Grampian, Aberdeen, Scotland. It was published online in the Journal of the American College of Cardiology.
LIMITATIONS:
Complete alignment of all variables related to clinical characteristics of patients with TTS and MI wasn’t feasible. During the study, TTS was still relatively unfamiliar to clinicians and underdiagnosed. As the study used a national data set of routinely collected data, not all desirable information was available, including indications of why drugs were prescribed or discontinued, which could have led to imprecise results. As the study used nonrandomized data, causality can’t be assumed.
DISCLOSURES:
Dr. Rudd had no relevant conflicts of interest. Study author Dana K. Dawson, Aberdeen Cardiovascular and Diabetes Centre, University of Aberdeen, Scotland, declared receiving the Chief Scientist Office Scotland award CGA-16-4 and the BHF Research Training Fellowship. Commentary authors had no relevant conflicts of interest.
A version of this article appeared on Medscape.com.