User login
Out-of-pocket costs for neurologic medications rise sharply
The out-of-pocket cost of multiple sclerosis (MS) treatments increased the most, with a 20-fold increase during that time. The average out-of-pocket cost for MS therapy was $15/month in 2004, compared with $309/month in 2016. Patients also had to pay more for brand name medications for peripheral neuropathy, dementia, and Parkinson’s disease, researchers said.
“Out-of-pocket costs vary widely both across and within conditions,” said study author Brian C. Callaghan, MD, an assistant professor of neurology at the University of Michigan in Ann Arbor, and research colleagues. “To minimize patient financial burden, neurologists require access to precise cost information when making treatment decisions.”
Prior studies have found that high drug costs “can create burdens such as medical debt, skipping food or other essentials, or even not taking drugs as often as necessary,” Dr. Callaghan said in a news release.
To assess how out-of-pocket costs affect patients with neurologic conditions, the investigators analyzed data from a large, privately insured health care claims database. They determined medication costs for patients with MS, peripheral neuropathy, epilepsy, dementia, and Parkinson’s disease who were seen by outpatient neurologists. They also compared costs for high-deductible and traditional plans and explored cumulative out-of-pocket costs during the first 2 years after diagnosis.
The analysis examined the five most commonly prescribed drugs by neurologists for each condition based on Medicare data. In addition, the researchers included in their analysis all approved MS medications, lacosamide as a brand name epilepsy drug, and venlafaxine, a peripheral neuropathy medication that transitioned from brand to generic.
In all, the study population included 105,355 patients with MS, 314,530 with peripheral neuropathy, 281,073 with epilepsy, 120,720 with dementia, and 90,801 with Parkinson’s disease.
In 2016, patients in high-deductible health plans had an average monthly out-of-pocket expense that was approximately twice that of patients not in those plans – $661 versus $246 among patients with MS, and $40 versus $18 among patients with epilepsy.
In the 2 years after diagnosis in 2012 or 2013, cumulative out-of-pocket costs for patients with MS were a mean of $2,238, but costs varied widely. Cumulative costs were no more than $90 for patients in the bottom 5% of expenses, whereas they exceeded $9,800 for patients in the top 5% of expenses. Among patients with epilepsy, cumulative out-of-pocket costs were $230 in the 2 years after diagnosis.
“In 2004, out-of-pocket costs were of such low magnitude that physicians could typically ignore these costs for most patients and not adversely affect the financial status of patients or their adherence to medications. However, by 2016, out-of-pockets costs have risen to the point where neurologists should consider out-of-pocket costs for most medications and for most patients,” Dr. Callaghan and colleagues wrote.
Ralph L. Sacco, MD, president of the American Academy of Neurology (AAN), said in a news release that the AAN has created a Neurology Drug Pricing Task Force and is advocating for better drug-pricing policies. “This study provides important information to help us better understand how these problems can directly affect our patients,” Dr. Sacco said.
“Everyone deserves affordable access to the medications that will be most beneficial, but if the drugs are too expensive, people may simply not take them, possibly leading to medical complications and higher costs later,” Dr. Sacco said.
The study was supported by the AAN. Several authors are supported by National Institutes of Health grants. Dr. Callaghan receives research support from Impeto Medical and performs consulting work.
SOURCE: Callaghan BC et al. Neurology. 2019 May 1. doi: 10.1212/WNL.0000000000007564.
The out-of-pocket cost of multiple sclerosis (MS) treatments increased the most, with a 20-fold increase during that time. The average out-of-pocket cost for MS therapy was $15/month in 2004, compared with $309/month in 2016. Patients also had to pay more for brand name medications for peripheral neuropathy, dementia, and Parkinson’s disease, researchers said.
“Out-of-pocket costs vary widely both across and within conditions,” said study author Brian C. Callaghan, MD, an assistant professor of neurology at the University of Michigan in Ann Arbor, and research colleagues. “To minimize patient financial burden, neurologists require access to precise cost information when making treatment decisions.”
Prior studies have found that high drug costs “can create burdens such as medical debt, skipping food or other essentials, or even not taking drugs as often as necessary,” Dr. Callaghan said in a news release.
To assess how out-of-pocket costs affect patients with neurologic conditions, the investigators analyzed data from a large, privately insured health care claims database. They determined medication costs for patients with MS, peripheral neuropathy, epilepsy, dementia, and Parkinson’s disease who were seen by outpatient neurologists. They also compared costs for high-deductible and traditional plans and explored cumulative out-of-pocket costs during the first 2 years after diagnosis.
The analysis examined the five most commonly prescribed drugs by neurologists for each condition based on Medicare data. In addition, the researchers included in their analysis all approved MS medications, lacosamide as a brand name epilepsy drug, and venlafaxine, a peripheral neuropathy medication that transitioned from brand to generic.
In all, the study population included 105,355 patients with MS, 314,530 with peripheral neuropathy, 281,073 with epilepsy, 120,720 with dementia, and 90,801 with Parkinson’s disease.
In 2016, patients in high-deductible health plans had an average monthly out-of-pocket expense that was approximately twice that of patients not in those plans – $661 versus $246 among patients with MS, and $40 versus $18 among patients with epilepsy.
In the 2 years after diagnosis in 2012 or 2013, cumulative out-of-pocket costs for patients with MS were a mean of $2,238, but costs varied widely. Cumulative costs were no more than $90 for patients in the bottom 5% of expenses, whereas they exceeded $9,800 for patients in the top 5% of expenses. Among patients with epilepsy, cumulative out-of-pocket costs were $230 in the 2 years after diagnosis.
“In 2004, out-of-pocket costs were of such low magnitude that physicians could typically ignore these costs for most patients and not adversely affect the financial status of patients or their adherence to medications. However, by 2016, out-of-pockets costs have risen to the point where neurologists should consider out-of-pocket costs for most medications and for most patients,” Dr. Callaghan and colleagues wrote.
Ralph L. Sacco, MD, president of the American Academy of Neurology (AAN), said in a news release that the AAN has created a Neurology Drug Pricing Task Force and is advocating for better drug-pricing policies. “This study provides important information to help us better understand how these problems can directly affect our patients,” Dr. Sacco said.
“Everyone deserves affordable access to the medications that will be most beneficial, but if the drugs are too expensive, people may simply not take them, possibly leading to medical complications and higher costs later,” Dr. Sacco said.
The study was supported by the AAN. Several authors are supported by National Institutes of Health grants. Dr. Callaghan receives research support from Impeto Medical and performs consulting work.
SOURCE: Callaghan BC et al. Neurology. 2019 May 1. doi: 10.1212/WNL.0000000000007564.
The out-of-pocket cost of multiple sclerosis (MS) treatments increased the most, with a 20-fold increase during that time. The average out-of-pocket cost for MS therapy was $15/month in 2004, compared with $309/month in 2016. Patients also had to pay more for brand name medications for peripheral neuropathy, dementia, and Parkinson’s disease, researchers said.
“Out-of-pocket costs vary widely both across and within conditions,” said study author Brian C. Callaghan, MD, an assistant professor of neurology at the University of Michigan in Ann Arbor, and research colleagues. “To minimize patient financial burden, neurologists require access to precise cost information when making treatment decisions.”
Prior studies have found that high drug costs “can create burdens such as medical debt, skipping food or other essentials, or even not taking drugs as often as necessary,” Dr. Callaghan said in a news release.
To assess how out-of-pocket costs affect patients with neurologic conditions, the investigators analyzed data from a large, privately insured health care claims database. They determined medication costs for patients with MS, peripheral neuropathy, epilepsy, dementia, and Parkinson’s disease who were seen by outpatient neurologists. They also compared costs for high-deductible and traditional plans and explored cumulative out-of-pocket costs during the first 2 years after diagnosis.
The analysis examined the five most commonly prescribed drugs by neurologists for each condition based on Medicare data. In addition, the researchers included in their analysis all approved MS medications, lacosamide as a brand name epilepsy drug, and venlafaxine, a peripheral neuropathy medication that transitioned from brand to generic.
In all, the study population included 105,355 patients with MS, 314,530 with peripheral neuropathy, 281,073 with epilepsy, 120,720 with dementia, and 90,801 with Parkinson’s disease.
In 2016, patients in high-deductible health plans had an average monthly out-of-pocket expense that was approximately twice that of patients not in those plans – $661 versus $246 among patients with MS, and $40 versus $18 among patients with epilepsy.
In the 2 years after diagnosis in 2012 or 2013, cumulative out-of-pocket costs for patients with MS were a mean of $2,238, but costs varied widely. Cumulative costs were no more than $90 for patients in the bottom 5% of expenses, whereas they exceeded $9,800 for patients in the top 5% of expenses. Among patients with epilepsy, cumulative out-of-pocket costs were $230 in the 2 years after diagnosis.
“In 2004, out-of-pocket costs were of such low magnitude that physicians could typically ignore these costs for most patients and not adversely affect the financial status of patients or their adherence to medications. However, by 2016, out-of-pockets costs have risen to the point where neurologists should consider out-of-pocket costs for most medications and for most patients,” Dr. Callaghan and colleagues wrote.
Ralph L. Sacco, MD, president of the American Academy of Neurology (AAN), said in a news release that the AAN has created a Neurology Drug Pricing Task Force and is advocating for better drug-pricing policies. “This study provides important information to help us better understand how these problems can directly affect our patients,” Dr. Sacco said.
“Everyone deserves affordable access to the medications that will be most beneficial, but if the drugs are too expensive, people may simply not take them, possibly leading to medical complications and higher costs later,” Dr. Sacco said.
The study was supported by the AAN. Several authors are supported by National Institutes of Health grants. Dr. Callaghan receives research support from Impeto Medical and performs consulting work.
SOURCE: Callaghan BC et al. Neurology. 2019 May 1. doi: 10.1212/WNL.0000000000007564.
FROM NEUROLOGY
Long-term antibiotic use may heighten stroke, CHD risk
, according to a study in the European Heart Journal.
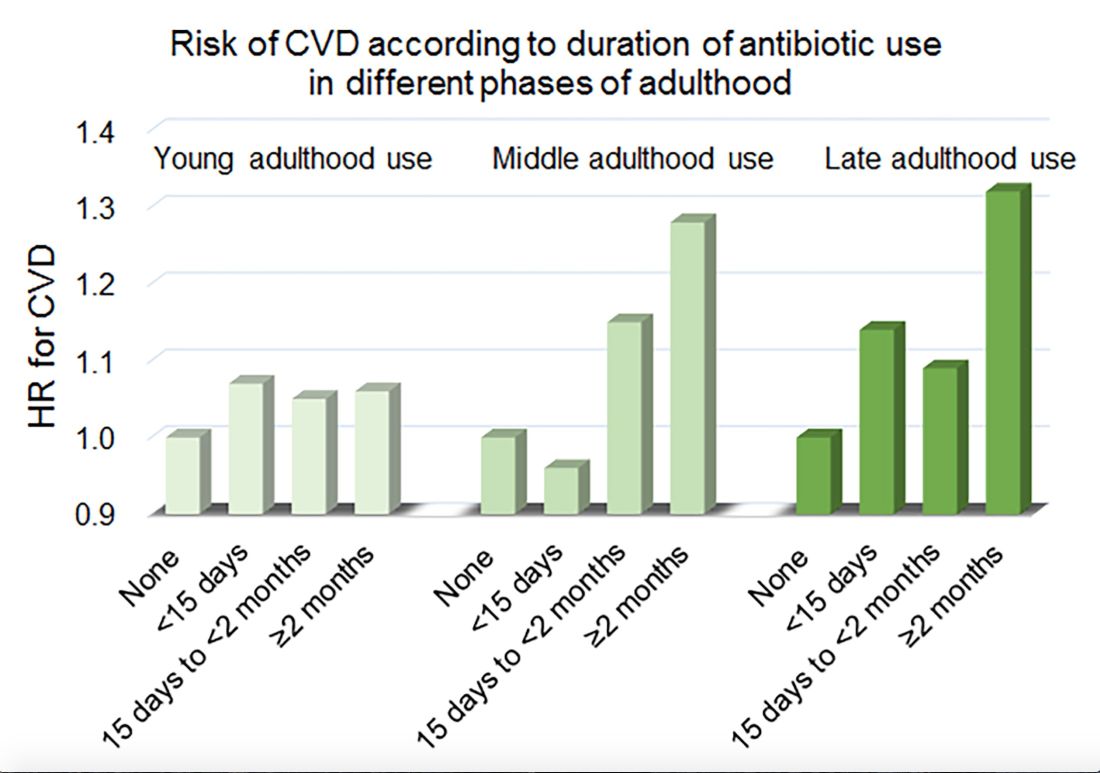
Women in the Nurses’ Health Study who used antibiotics for 2 or more months between ages 40 and 59 years or at age 60 years and older had a significantly increased risk of cardiovascular disease, compared with those who did not use antibiotics. Antibiotic use between 20 and 39 years old was not significantly related to cardiovascular disease.
Prior research has found that antibiotics may have long-lasting effects on gut microbiota and relate to cardiovascular disease risk.
“Antibiotic use is the most critical factor in altering the balance of microorganisms in the gut,” said lead investigator Lu Qi, MD, PhD, in a news release. “Previous studies have shown a link between alterations in the microbiotic environment of the gut and inflammation and narrowing of the blood vessels, stroke, and heart disease,” said Dr. Qi, who is the director of the Tulane University Obesity Research Center in New Orleans and an adjunct professor of nutrition at Harvard T.C. Chan School of Public Health in Boston.
To evaluate associations between life stage, antibiotic exposure, and subsequent cardiovascular disease, researchers analyzed data from 36,429 participants in the Nurses’ Health Study. The women were at least 60 years old and had no history of cardiovascular disease or cancer when they completed a 2004 questionnaire about antibiotic usage during young, middle, and late adulthood. The questionnaire asked participants to indicate the total time using antibiotics with eight categories ranging from none to 5 or more years.
The researchers defined incident cardiovascular disease as a composite endpoint of coronary heart disease (nonfatal myocardial infarction or fatal coronary heart disease) and stroke (nonfatal or fatal). They calculated person-years of follow-up from the questionnaire return date until date of cardiovascular disease diagnosis, death, or end of follow-up in 2012.
Women with longer duration of antibiotic use were more likely to use other medications and have unfavorable cardiovascular risk profiles, including family history of myocardial infarction and higher body mass index. Antibiotics most often were used to treat respiratory infections. During an average follow-up of 7.6 years, 1,056 participants developed cardiovascular disease.
In a multivariable model that adjusted for demographics, diet, lifestyle, reason for antibiotic use, medications, overweight status, and other factors, long-term antibiotic use – 2 months or more – in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Although antibiotic use was self-reported, which could lead to misclassification, the participants were health professionals, which may mitigate this limitation, the authors noted. Whether these findings apply to men and other populations requires further study, they said.
Because of the study’s observational design, the results “cannot show that antibiotics cause heart disease and stroke, only that there is a link between them,” Dr. Qi said. “It’s possible that women who reported more antibiotic use might be sicker in other ways that we were unable to measure, or there may be other factors that could affect the results that we have not been able take account of.”
“Our study suggests that antibiotics should be used only when they are absolutely needed,” he concluded. “Considering the potentially cumulative adverse effects, the shorter time of antibiotic use the better.”
The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
SOURCE: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
, according to a study in the European Heart Journal.

Women in the Nurses’ Health Study who used antibiotics for 2 or more months between ages 40 and 59 years or at age 60 years and older had a significantly increased risk of cardiovascular disease, compared with those who did not use antibiotics. Antibiotic use between 20 and 39 years old was not significantly related to cardiovascular disease.
Prior research has found that antibiotics may have long-lasting effects on gut microbiota and relate to cardiovascular disease risk.
“Antibiotic use is the most critical factor in altering the balance of microorganisms in the gut,” said lead investigator Lu Qi, MD, PhD, in a news release. “Previous studies have shown a link between alterations in the microbiotic environment of the gut and inflammation and narrowing of the blood vessels, stroke, and heart disease,” said Dr. Qi, who is the director of the Tulane University Obesity Research Center in New Orleans and an adjunct professor of nutrition at Harvard T.C. Chan School of Public Health in Boston.
To evaluate associations between life stage, antibiotic exposure, and subsequent cardiovascular disease, researchers analyzed data from 36,429 participants in the Nurses’ Health Study. The women were at least 60 years old and had no history of cardiovascular disease or cancer when they completed a 2004 questionnaire about antibiotic usage during young, middle, and late adulthood. The questionnaire asked participants to indicate the total time using antibiotics with eight categories ranging from none to 5 or more years.
The researchers defined incident cardiovascular disease as a composite endpoint of coronary heart disease (nonfatal myocardial infarction or fatal coronary heart disease) and stroke (nonfatal or fatal). They calculated person-years of follow-up from the questionnaire return date until date of cardiovascular disease diagnosis, death, or end of follow-up in 2012.
Women with longer duration of antibiotic use were more likely to use other medications and have unfavorable cardiovascular risk profiles, including family history of myocardial infarction and higher body mass index. Antibiotics most often were used to treat respiratory infections. During an average follow-up of 7.6 years, 1,056 participants developed cardiovascular disease.
In a multivariable model that adjusted for demographics, diet, lifestyle, reason for antibiotic use, medications, overweight status, and other factors, long-term antibiotic use – 2 months or more – in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Although antibiotic use was self-reported, which could lead to misclassification, the participants were health professionals, which may mitigate this limitation, the authors noted. Whether these findings apply to men and other populations requires further study, they said.
Because of the study’s observational design, the results “cannot show that antibiotics cause heart disease and stroke, only that there is a link between them,” Dr. Qi said. “It’s possible that women who reported more antibiotic use might be sicker in other ways that we were unable to measure, or there may be other factors that could affect the results that we have not been able take account of.”
“Our study suggests that antibiotics should be used only when they are absolutely needed,” he concluded. “Considering the potentially cumulative adverse effects, the shorter time of antibiotic use the better.”
The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
SOURCE: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
, according to a study in the European Heart Journal.

Women in the Nurses’ Health Study who used antibiotics for 2 or more months between ages 40 and 59 years or at age 60 years and older had a significantly increased risk of cardiovascular disease, compared with those who did not use antibiotics. Antibiotic use between 20 and 39 years old was not significantly related to cardiovascular disease.
Prior research has found that antibiotics may have long-lasting effects on gut microbiota and relate to cardiovascular disease risk.
“Antibiotic use is the most critical factor in altering the balance of microorganisms in the gut,” said lead investigator Lu Qi, MD, PhD, in a news release. “Previous studies have shown a link between alterations in the microbiotic environment of the gut and inflammation and narrowing of the blood vessels, stroke, and heart disease,” said Dr. Qi, who is the director of the Tulane University Obesity Research Center in New Orleans and an adjunct professor of nutrition at Harvard T.C. Chan School of Public Health in Boston.
To evaluate associations between life stage, antibiotic exposure, and subsequent cardiovascular disease, researchers analyzed data from 36,429 participants in the Nurses’ Health Study. The women were at least 60 years old and had no history of cardiovascular disease or cancer when they completed a 2004 questionnaire about antibiotic usage during young, middle, and late adulthood. The questionnaire asked participants to indicate the total time using antibiotics with eight categories ranging from none to 5 or more years.
The researchers defined incident cardiovascular disease as a composite endpoint of coronary heart disease (nonfatal myocardial infarction or fatal coronary heart disease) and stroke (nonfatal or fatal). They calculated person-years of follow-up from the questionnaire return date until date of cardiovascular disease diagnosis, death, or end of follow-up in 2012.
Women with longer duration of antibiotic use were more likely to use other medications and have unfavorable cardiovascular risk profiles, including family history of myocardial infarction and higher body mass index. Antibiotics most often were used to treat respiratory infections. During an average follow-up of 7.6 years, 1,056 participants developed cardiovascular disease.
In a multivariable model that adjusted for demographics, diet, lifestyle, reason for antibiotic use, medications, overweight status, and other factors, long-term antibiotic use – 2 months or more – in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Although antibiotic use was self-reported, which could lead to misclassification, the participants were health professionals, which may mitigate this limitation, the authors noted. Whether these findings apply to men and other populations requires further study, they said.
Because of the study’s observational design, the results “cannot show that antibiotics cause heart disease and stroke, only that there is a link between them,” Dr. Qi said. “It’s possible that women who reported more antibiotic use might be sicker in other ways that we were unable to measure, or there may be other factors that could affect the results that we have not been able take account of.”
“Our study suggests that antibiotics should be used only when they are absolutely needed,” he concluded. “Considering the potentially cumulative adverse effects, the shorter time of antibiotic use the better.”
The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
SOURCE: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
FROM THE EUROPEAN HEART JOURNAL
Key clinical point: Among middle-aged and older women, 2 or more months’ exposure to antibiotics is associated with an increased risk of coronary heart disease or stroke.
Major finding: Long-term antibiotic use in late adulthood was associated with significantly increased risk of cardiovascular disease (hazard ratio, 1.32), as was long-term antibiotic use in middle adulthood (HR, 1.28).
Study details: An analysis of data from nearly 36,500 women in the Nurses’ Health Study.
Disclosures: The study was supported by National Institutes of Health grants, the Boston Obesity Nutrition Research Center, and the United States–Israel Binational Science Foundation. One author received support from the Japan Society for the Promotion of Science. The authors had no conflicts of interest.
Source: Heianza Y et al. Eur Heart J. 2019 Apr 24. doi: 10.1093/eurheartj/ehz231.
Idelalisib shows long-term safety, efficacy for relapsed CLL
For patients with relapsed/refractory chronic lymphocytic leukemia (CLL), long-term treatment with the phosphoinositol 3-kinase inhibitor idelalisib appears safe and effective, according to investigators.
Final results from a phase 3 trial confirmed survival advantages when idelalisib is used in combination with rituximab, reported lead author Jeff P. Sharman, MD, of Willamette Valley Cancer Institute and Research Center in Springfield, Ore., and colleagues.
During follow-up, which exceeded 5 years in some patients, no new idelalisib-related adverse events were encountered, supporting the safety of long-term use, the investigators noted. The report is in the Journal of Clinical Oncology.
This study was “pivotal” for treating elderly patients with relapsed CLL, the investigators wrote, as these patients previously had few treatment options beyond supportive or palliative care.
Earlier results from the study showed that adding idelalisib to rituximab raised overall response rates from about 15.5% to 83.6% and median progression-free survival from 6.5 months to 19.4 months, resulting in “significantly better clinical outcomes compared with those seen with rituximab alone,” leading to approval by the Food and Drug Administration.
During the primary study, 110 patients received a combination of idelalisib and rituximab, while 108 patients received rituximab and placebo. The median patient age was 71 years, with a median of three lines of prior therapy. The present analysis focused on the 110 patients in the combination group who received at least one dose of idelalisib, whether or not they elected to participate in the extension phase.
After a median follow-up of 18 months, ranging from 0.3 months to 67.6 months, the overall response rate was 85.5% and the median progression-free survival was 20.3 months, both of which are similar to earlier findings. Median overall survival was 40.6 months.
With a median duration of exposure of 16.2 months, the safety analysis revealed no new idelalisib-related adverse events.
However, the investigators pointed out that prolonged therapy often led to diarrhea, which ultimately occurred in about half of patients (46.4%). Roughly equal amounts of patients experienced grade 2 (17.3%) or grade 3 or greater diarrhea (16.4%). In cases of grade 3 or greater diarrhea, steroid therapy was recommended, typically resulting in symptom resolution within 2 weeks; however, “there were insufficient numbers of patients to determine if steroid therapy affected the duration of symptoms,” the investigators wrote.
“The longer-term data presented here confirm the previously reported efficacy of targeting PI3K with idelalisib in patients with relapsed/refractory CLL and support the use of [idelalisib and rituximab] in this patient population with careful management of potential [adverse events],” they wrote.
Gilead Sciences funded the study. Dr. Sharman reported financial relationships with Gilead and other companies.
SOURCE: Sharman JP et al. J Clin Oncol. 2019 Apr 17. doi: 10.1200/JCO.18.01460.
For patients with relapsed/refractory chronic lymphocytic leukemia (CLL), long-term treatment with the phosphoinositol 3-kinase inhibitor idelalisib appears safe and effective, according to investigators.
Final results from a phase 3 trial confirmed survival advantages when idelalisib is used in combination with rituximab, reported lead author Jeff P. Sharman, MD, of Willamette Valley Cancer Institute and Research Center in Springfield, Ore., and colleagues.
During follow-up, which exceeded 5 years in some patients, no new idelalisib-related adverse events were encountered, supporting the safety of long-term use, the investigators noted. The report is in the Journal of Clinical Oncology.
This study was “pivotal” for treating elderly patients with relapsed CLL, the investigators wrote, as these patients previously had few treatment options beyond supportive or palliative care.
Earlier results from the study showed that adding idelalisib to rituximab raised overall response rates from about 15.5% to 83.6% and median progression-free survival from 6.5 months to 19.4 months, resulting in “significantly better clinical outcomes compared with those seen with rituximab alone,” leading to approval by the Food and Drug Administration.
During the primary study, 110 patients received a combination of idelalisib and rituximab, while 108 patients received rituximab and placebo. The median patient age was 71 years, with a median of three lines of prior therapy. The present analysis focused on the 110 patients in the combination group who received at least one dose of idelalisib, whether or not they elected to participate in the extension phase.
After a median follow-up of 18 months, ranging from 0.3 months to 67.6 months, the overall response rate was 85.5% and the median progression-free survival was 20.3 months, both of which are similar to earlier findings. Median overall survival was 40.6 months.
With a median duration of exposure of 16.2 months, the safety analysis revealed no new idelalisib-related adverse events.
However, the investigators pointed out that prolonged therapy often led to diarrhea, which ultimately occurred in about half of patients (46.4%). Roughly equal amounts of patients experienced grade 2 (17.3%) or grade 3 or greater diarrhea (16.4%). In cases of grade 3 or greater diarrhea, steroid therapy was recommended, typically resulting in symptom resolution within 2 weeks; however, “there were insufficient numbers of patients to determine if steroid therapy affected the duration of symptoms,” the investigators wrote.
“The longer-term data presented here confirm the previously reported efficacy of targeting PI3K with idelalisib in patients with relapsed/refractory CLL and support the use of [idelalisib and rituximab] in this patient population with careful management of potential [adverse events],” they wrote.
Gilead Sciences funded the study. Dr. Sharman reported financial relationships with Gilead and other companies.
SOURCE: Sharman JP et al. J Clin Oncol. 2019 Apr 17. doi: 10.1200/JCO.18.01460.
For patients with relapsed/refractory chronic lymphocytic leukemia (CLL), long-term treatment with the phosphoinositol 3-kinase inhibitor idelalisib appears safe and effective, according to investigators.
Final results from a phase 3 trial confirmed survival advantages when idelalisib is used in combination with rituximab, reported lead author Jeff P. Sharman, MD, of Willamette Valley Cancer Institute and Research Center in Springfield, Ore., and colleagues.
During follow-up, which exceeded 5 years in some patients, no new idelalisib-related adverse events were encountered, supporting the safety of long-term use, the investigators noted. The report is in the Journal of Clinical Oncology.
This study was “pivotal” for treating elderly patients with relapsed CLL, the investigators wrote, as these patients previously had few treatment options beyond supportive or palliative care.
Earlier results from the study showed that adding idelalisib to rituximab raised overall response rates from about 15.5% to 83.6% and median progression-free survival from 6.5 months to 19.4 months, resulting in “significantly better clinical outcomes compared with those seen with rituximab alone,” leading to approval by the Food and Drug Administration.
During the primary study, 110 patients received a combination of idelalisib and rituximab, while 108 patients received rituximab and placebo. The median patient age was 71 years, with a median of three lines of prior therapy. The present analysis focused on the 110 patients in the combination group who received at least one dose of idelalisib, whether or not they elected to participate in the extension phase.
After a median follow-up of 18 months, ranging from 0.3 months to 67.6 months, the overall response rate was 85.5% and the median progression-free survival was 20.3 months, both of which are similar to earlier findings. Median overall survival was 40.6 months.
With a median duration of exposure of 16.2 months, the safety analysis revealed no new idelalisib-related adverse events.
However, the investigators pointed out that prolonged therapy often led to diarrhea, which ultimately occurred in about half of patients (46.4%). Roughly equal amounts of patients experienced grade 2 (17.3%) or grade 3 or greater diarrhea (16.4%). In cases of grade 3 or greater diarrhea, steroid therapy was recommended, typically resulting in symptom resolution within 2 weeks; however, “there were insufficient numbers of patients to determine if steroid therapy affected the duration of symptoms,” the investigators wrote.
“The longer-term data presented here confirm the previously reported efficacy of targeting PI3K with idelalisib in patients with relapsed/refractory CLL and support the use of [idelalisib and rituximab] in this patient population with careful management of potential [adverse events],” they wrote.
Gilead Sciences funded the study. Dr. Sharman reported financial relationships with Gilead and other companies.
SOURCE: Sharman JP et al. J Clin Oncol. 2019 Apr 17. doi: 10.1200/JCO.18.01460.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Lessons from KEYNOTE-158 and the role of R-CHOP
In this edition of “How I will treat my next patient,” I take a look at two recent trials – one offers potential in previously-treated cervical cancer patients with poor prognosis and the other confirms the role of R-CHOP as the standard of care in diffuse large B-cell lymphoma.
Pembrolizumab in KEYNOTE-158
In an international phase 2 “basket trial,” Hyun Cheol Chung, MD, PhD, and colleagues used pembrolizumab 200 mg every 3 weeks in 98 previously treated patients with advanced cervical cancer. Almost 84% of o the patients had PD-L1 positive tumors (greater than 1%). The authors said that viral induction of malignancy leads to antigen production and upregulation of PD-1. Therefore, advanced cervical cancer patients would likely express PD-L1 on tumor cells and respond to immune checkpoint inhibitor therapy.
In this interim report, there were 12 responses (all in PD-L1 positive patients), with three complete responses. Median response duration had not been reached at median follow-up of 10.2 months. Seven of 12 responses were ongoing at 12 months. There were grade 3-4 adverse events in 12.2% of patients and no treatment-related deaths.
The study – “Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Cervical Cancer: Results From the Phase II KEYNOTE-158 Study” – was published in the Journal of Clinical Oncology (2019 April 3. doi: 10.1200/JCO.18.01265).
The encouraging results of pembrolizumab in this generally chemotherapy-refractory patient population were consistent with other small, early-phase studies investigating immune checkpoint inhibitors that led to the accelerated approval of pembrolizumab in previously treated PD-L1 advanced cervical cancer patients with progressive disease after chemotherapy.
What this means in practice
Although excitement should be tempered about an interim report of an organ-specific subset of a phase 2 international basket trial that was heavily populated by young PS 0-1 patients and generated an overall response rate of less than 15%, no conventional chemotherapy or biologic agent offers the potential of complete or prolonged response, and disease control rates of 30%.
Clinical trials should always be the first choice, but immune checkpoint inhibitors offer an attractive off-study option.
Among many single agents in National Comprehensive Cancer Network guidelines for recurrent advanced cervical cancer after first-line cisplatin-based chemotherapy, there is a reason why pembrolizumab is listed first. For patients with PD-L1 expressing tumors or MSI-H/dMMR tumors, I would use it.
Frontline therapy in DLBCL
In a large, randomized phase 3 trial, close to 500 stage III-IV patients with diffuse large B-cell lymphoma (DLBCL), including primary mediastinal B-cell lymphoma and intravascular large B-cell lymphoma, were assigned to receive either conventional R-CHOP chemotherapy or the more complex, more toxic DA-EPOCH-R regimen that appeared superior in single-institution studies and was feasible in multi-institutional phase 2 trials.
The study – “Dose-Adjusted EPOCH-R Compared With R-CHOP as Frontline Therapy for Diffuse Large B-Cell Lymphoma: Clinical Outcomes of the Phase III Intergroup Trial Alliance/CALGB 50303” – was published in the Journal of Clinical Oncology (2019 Apr 2. doi: 10.1200/JCO.18.01994).
In the study, progression-free survival and overall survival were no different for R-CHOP and DA-EPOCH-R, but – predictably – DA-EPOCH-R was more toxic and had more treatment discontinuations.
R-CHOP had better outcomes than expected. This suggests that patient-selection bias (more favorable histology, fewer high-risk subsets who required urgent therapy) may have been at work.
Further study of DA-EPOCH-R in higher IPI patients or in patients selected because of more adverse molecular features (DE phenotype, MYC+, double hit) is warranted given the poor outcomes with R-CHOP in high-risk patients, intriguing results in single institution trials of DA-EPOCH-R, and the underrepresentation of high-risk patients in the current study.
What this means in practice
Whether by virtue of the types of patients enrolled or because it is the best regimen in all DLBCL patients, R-CHOP remains the standard of care outside of a clinical trial.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers, and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I will treat my next patient,” I take a look at two recent trials – one offers potential in previously-treated cervical cancer patients with poor prognosis and the other confirms the role of R-CHOP as the standard of care in diffuse large B-cell lymphoma.
Pembrolizumab in KEYNOTE-158
In an international phase 2 “basket trial,” Hyun Cheol Chung, MD, PhD, and colleagues used pembrolizumab 200 mg every 3 weeks in 98 previously treated patients with advanced cervical cancer. Almost 84% of o the patients had PD-L1 positive tumors (greater than 1%). The authors said that viral induction of malignancy leads to antigen production and upregulation of PD-1. Therefore, advanced cervical cancer patients would likely express PD-L1 on tumor cells and respond to immune checkpoint inhibitor therapy.
In this interim report, there were 12 responses (all in PD-L1 positive patients), with three complete responses. Median response duration had not been reached at median follow-up of 10.2 months. Seven of 12 responses were ongoing at 12 months. There were grade 3-4 adverse events in 12.2% of patients and no treatment-related deaths.
The study – “Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Cervical Cancer: Results From the Phase II KEYNOTE-158 Study” – was published in the Journal of Clinical Oncology (2019 April 3. doi: 10.1200/JCO.18.01265).
The encouraging results of pembrolizumab in this generally chemotherapy-refractory patient population were consistent with other small, early-phase studies investigating immune checkpoint inhibitors that led to the accelerated approval of pembrolizumab in previously treated PD-L1 advanced cervical cancer patients with progressive disease after chemotherapy.
What this means in practice
Although excitement should be tempered about an interim report of an organ-specific subset of a phase 2 international basket trial that was heavily populated by young PS 0-1 patients and generated an overall response rate of less than 15%, no conventional chemotherapy or biologic agent offers the potential of complete or prolonged response, and disease control rates of 30%.
Clinical trials should always be the first choice, but immune checkpoint inhibitors offer an attractive off-study option.
Among many single agents in National Comprehensive Cancer Network guidelines for recurrent advanced cervical cancer after first-line cisplatin-based chemotherapy, there is a reason why pembrolizumab is listed first. For patients with PD-L1 expressing tumors or MSI-H/dMMR tumors, I would use it.
Frontline therapy in DLBCL
In a large, randomized phase 3 trial, close to 500 stage III-IV patients with diffuse large B-cell lymphoma (DLBCL), including primary mediastinal B-cell lymphoma and intravascular large B-cell lymphoma, were assigned to receive either conventional R-CHOP chemotherapy or the more complex, more toxic DA-EPOCH-R regimen that appeared superior in single-institution studies and was feasible in multi-institutional phase 2 trials.
The study – “Dose-Adjusted EPOCH-R Compared With R-CHOP as Frontline Therapy for Diffuse Large B-Cell Lymphoma: Clinical Outcomes of the Phase III Intergroup Trial Alliance/CALGB 50303” – was published in the Journal of Clinical Oncology (2019 Apr 2. doi: 10.1200/JCO.18.01994).
In the study, progression-free survival and overall survival were no different for R-CHOP and DA-EPOCH-R, but – predictably – DA-EPOCH-R was more toxic and had more treatment discontinuations.
R-CHOP had better outcomes than expected. This suggests that patient-selection bias (more favorable histology, fewer high-risk subsets who required urgent therapy) may have been at work.
Further study of DA-EPOCH-R in higher IPI patients or in patients selected because of more adverse molecular features (DE phenotype, MYC+, double hit) is warranted given the poor outcomes with R-CHOP in high-risk patients, intriguing results in single institution trials of DA-EPOCH-R, and the underrepresentation of high-risk patients in the current study.
What this means in practice
Whether by virtue of the types of patients enrolled or because it is the best regimen in all DLBCL patients, R-CHOP remains the standard of care outside of a clinical trial.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers, and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I will treat my next patient,” I take a look at two recent trials – one offers potential in previously-treated cervical cancer patients with poor prognosis and the other confirms the role of R-CHOP as the standard of care in diffuse large B-cell lymphoma.
Pembrolizumab in KEYNOTE-158
In an international phase 2 “basket trial,” Hyun Cheol Chung, MD, PhD, and colleagues used pembrolizumab 200 mg every 3 weeks in 98 previously treated patients with advanced cervical cancer. Almost 84% of o the patients had PD-L1 positive tumors (greater than 1%). The authors said that viral induction of malignancy leads to antigen production and upregulation of PD-1. Therefore, advanced cervical cancer patients would likely express PD-L1 on tumor cells and respond to immune checkpoint inhibitor therapy.
In this interim report, there were 12 responses (all in PD-L1 positive patients), with three complete responses. Median response duration had not been reached at median follow-up of 10.2 months. Seven of 12 responses were ongoing at 12 months. There were grade 3-4 adverse events in 12.2% of patients and no treatment-related deaths.
The study – “Efficacy and Safety of Pembrolizumab in Previously Treated Advanced Cervical Cancer: Results From the Phase II KEYNOTE-158 Study” – was published in the Journal of Clinical Oncology (2019 April 3. doi: 10.1200/JCO.18.01265).
The encouraging results of pembrolizumab in this generally chemotherapy-refractory patient population were consistent with other small, early-phase studies investigating immune checkpoint inhibitors that led to the accelerated approval of pembrolizumab in previously treated PD-L1 advanced cervical cancer patients with progressive disease after chemotherapy.
What this means in practice
Although excitement should be tempered about an interim report of an organ-specific subset of a phase 2 international basket trial that was heavily populated by young PS 0-1 patients and generated an overall response rate of less than 15%, no conventional chemotherapy or biologic agent offers the potential of complete or prolonged response, and disease control rates of 30%.
Clinical trials should always be the first choice, but immune checkpoint inhibitors offer an attractive off-study option.
Among many single agents in National Comprehensive Cancer Network guidelines for recurrent advanced cervical cancer after first-line cisplatin-based chemotherapy, there is a reason why pembrolizumab is listed first. For patients with PD-L1 expressing tumors or MSI-H/dMMR tumors, I would use it.
Frontline therapy in DLBCL
In a large, randomized phase 3 trial, close to 500 stage III-IV patients with diffuse large B-cell lymphoma (DLBCL), including primary mediastinal B-cell lymphoma and intravascular large B-cell lymphoma, were assigned to receive either conventional R-CHOP chemotherapy or the more complex, more toxic DA-EPOCH-R regimen that appeared superior in single-institution studies and was feasible in multi-institutional phase 2 trials.
The study – “Dose-Adjusted EPOCH-R Compared With R-CHOP as Frontline Therapy for Diffuse Large B-Cell Lymphoma: Clinical Outcomes of the Phase III Intergroup Trial Alliance/CALGB 50303” – was published in the Journal of Clinical Oncology (2019 Apr 2. doi: 10.1200/JCO.18.01994).
In the study, progression-free survival and overall survival were no different for R-CHOP and DA-EPOCH-R, but – predictably – DA-EPOCH-R was more toxic and had more treatment discontinuations.
R-CHOP had better outcomes than expected. This suggests that patient-selection bias (more favorable histology, fewer high-risk subsets who required urgent therapy) may have been at work.
Further study of DA-EPOCH-R in higher IPI patients or in patients selected because of more adverse molecular features (DE phenotype, MYC+, double hit) is warranted given the poor outcomes with R-CHOP in high-risk patients, intriguing results in single institution trials of DA-EPOCH-R, and the underrepresentation of high-risk patients in the current study.
What this means in practice
Whether by virtue of the types of patients enrolled or because it is the best regimen in all DLBCL patients, R-CHOP remains the standard of care outside of a clinical trial.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers, and in expanding access to clinical trials to medically underserved populations.
High-dose MTX-based chemo is well tolerated in older PCNSL patients
GLASGOW – Most older patients with primary central nervous system lymphoma (PCNSL) can tolerate high-dose methotrexate-based chemotherapy and achieve similar outcomes as younger and fitter patients, according to a retrospective analysis of 244 patients in the United Kingdom.
For older patients – at least 65 years old – who received methotrexate-based regimens, treatment-related mortality was 6.8%, which is comparable with rates seen in trials involving younger patients, reported lead author Edward Poynton, MD, of University College Hospital in London.
Specifically, Dr. Poynton cited the phase 2 IELSG32 trial, which had a treatment-related mortality rate of 6% among patients up to age 70 years. These patients were treated with the established protocol for younger patients: chemotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix) followed by autologous stem cell transplant or whole-brain radiotherapy.
Introducing Dr. Poynton’s presentation at the annual meeting of the British Society for Haematology, Simon Rule, MD, of the University of Plymouth (England), added historical context to the new findings.
“When I started in hematology ... [PCNSL] was a universally fatal disease, pretty much,” Dr. Rule said. “And then we had methotrexate, and it worked occasionally. And then we had a randomized trial, which was randomization of methotrexate plus or minus high-dose cytarabine, showing benefit.”
This combination became the benchmark against which subsequent randomized trials were measured; however, such high-intensity regimens have raised concerns about safety and efficacy in older patients, Dr. Rule said, noting that the present study serves to inform clinicians about real-world outcomes in this population.
The retrospective analysis reviewed 244 patients who were aged at least 65 years when histologically diagnosed with PCNSL at 14 U.K. tertiary centers between 2012 and 2017. All patients received first-line care of any kind, ranging from best supportive care to clinical trial therapy. Patients were grouped into three treatment cohorts divided by level of frailty. Analysis showed that these divisions correlated with age, renal function, Eastern Cooperative Oncology Group performance status, and treatment intensity.
The frail group received palliative treatment consisting of whole-brain radiotherapy, an oral alkylator, or best supportive care. The less-fit group received methotrexate in combination with rituximab, an oral alkylator, or both. The fit group was most intensively treated, receiving high-dose methotrexate and cytarabine – with or without rituximab – or MATRix.
The primary objective was overall response rate, while the secondary objectives were median overall survival and progression-free survival.
The analysis showed that 79% of patients (n = 193) received methotrexate-based therapy of some kind, with 61% receiving three or more cycles of therapy and 30% requiring dose reductions. The overall response rate was 63%.
Dr. Poynton noted that about two-thirds of patients who achieved a partial response in early assessment went on to achieve a complete response. Patients in the fit group more often responded than those who were less fit (87% vs. 65%; P = .01) and more often received consolidation therapy (42% vs. 23%; P = .01).
Fitness level was also associated with median overall survival, which was longest in the fit group at 42 months. The other two groups had dramatically shorter survival times: 8 months in the less-fit group and just 2 months in the frail group.
A closer look at the data revealed some patterns, Dr. Poynton said.
“What we see is that age at diagnosis is significantly correlated with progression-free survival but not with overall survival,” he said, noting that, in contrast, performance status was associated with both survival measures.
Methotrexate dose also impacted both survival measures. Patients who received 75% or more of their induction dose over the course of treatment had better median overall survival and progression-free survival than those who received less than 75%. Similarly, consolidation therapy improved both survival measures.
Patients aged older than 70 years who received intensive chemotherapy had a treatment-related mortality rate of 4.8%, which is lower than the overall treatment-related mortality, Dr. Poynton reported.
Considering the correlation between methotrexate dose and survival, Dr. Poynton suggested that “dose reductions should be carefully considered.”
He also noted that patients in the fit cohort who received intensive chemotherapy had comparable outcomes with younger patients in prospective trials, and yet 44% of patients older than 65 years in the real world who received high-dose methotrexate with cytarabine would have been ineligible for the IELSG32 trial.
“We’ve been able to identify this cohort of patients retrospectively,” Dr. Poynton said. “They definitely exist, and I think we need to work harder at how are going to identify these patients prospectively in the future, so we know which of our patients who are older can benefit from intensive chemotherapy and which patients won’t.”
Dr. Poynton reported having no relevant financial disclosures. His coinvestigators reported relationships with AbbVie, Merck, Takeda, Jazz Pharmaceuticals, and others.
GLASGOW – Most older patients with primary central nervous system lymphoma (PCNSL) can tolerate high-dose methotrexate-based chemotherapy and achieve similar outcomes as younger and fitter patients, according to a retrospective analysis of 244 patients in the United Kingdom.
For older patients – at least 65 years old – who received methotrexate-based regimens, treatment-related mortality was 6.8%, which is comparable with rates seen in trials involving younger patients, reported lead author Edward Poynton, MD, of University College Hospital in London.
Specifically, Dr. Poynton cited the phase 2 IELSG32 trial, which had a treatment-related mortality rate of 6% among patients up to age 70 years. These patients were treated with the established protocol for younger patients: chemotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix) followed by autologous stem cell transplant or whole-brain radiotherapy.
Introducing Dr. Poynton’s presentation at the annual meeting of the British Society for Haematology, Simon Rule, MD, of the University of Plymouth (England), added historical context to the new findings.
“When I started in hematology ... [PCNSL] was a universally fatal disease, pretty much,” Dr. Rule said. “And then we had methotrexate, and it worked occasionally. And then we had a randomized trial, which was randomization of methotrexate plus or minus high-dose cytarabine, showing benefit.”
This combination became the benchmark against which subsequent randomized trials were measured; however, such high-intensity regimens have raised concerns about safety and efficacy in older patients, Dr. Rule said, noting that the present study serves to inform clinicians about real-world outcomes in this population.
The retrospective analysis reviewed 244 patients who were aged at least 65 years when histologically diagnosed with PCNSL at 14 U.K. tertiary centers between 2012 and 2017. All patients received first-line care of any kind, ranging from best supportive care to clinical trial therapy. Patients were grouped into three treatment cohorts divided by level of frailty. Analysis showed that these divisions correlated with age, renal function, Eastern Cooperative Oncology Group performance status, and treatment intensity.
The frail group received palliative treatment consisting of whole-brain radiotherapy, an oral alkylator, or best supportive care. The less-fit group received methotrexate in combination with rituximab, an oral alkylator, or both. The fit group was most intensively treated, receiving high-dose methotrexate and cytarabine – with or without rituximab – or MATRix.
The primary objective was overall response rate, while the secondary objectives were median overall survival and progression-free survival.
The analysis showed that 79% of patients (n = 193) received methotrexate-based therapy of some kind, with 61% receiving three or more cycles of therapy and 30% requiring dose reductions. The overall response rate was 63%.
Dr. Poynton noted that about two-thirds of patients who achieved a partial response in early assessment went on to achieve a complete response. Patients in the fit group more often responded than those who were less fit (87% vs. 65%; P = .01) and more often received consolidation therapy (42% vs. 23%; P = .01).
Fitness level was also associated with median overall survival, which was longest in the fit group at 42 months. The other two groups had dramatically shorter survival times: 8 months in the less-fit group and just 2 months in the frail group.
A closer look at the data revealed some patterns, Dr. Poynton said.
“What we see is that age at diagnosis is significantly correlated with progression-free survival but not with overall survival,” he said, noting that, in contrast, performance status was associated with both survival measures.
Methotrexate dose also impacted both survival measures. Patients who received 75% or more of their induction dose over the course of treatment had better median overall survival and progression-free survival than those who received less than 75%. Similarly, consolidation therapy improved both survival measures.
Patients aged older than 70 years who received intensive chemotherapy had a treatment-related mortality rate of 4.8%, which is lower than the overall treatment-related mortality, Dr. Poynton reported.
Considering the correlation between methotrexate dose and survival, Dr. Poynton suggested that “dose reductions should be carefully considered.”
He also noted that patients in the fit cohort who received intensive chemotherapy had comparable outcomes with younger patients in prospective trials, and yet 44% of patients older than 65 years in the real world who received high-dose methotrexate with cytarabine would have been ineligible for the IELSG32 trial.
“We’ve been able to identify this cohort of patients retrospectively,” Dr. Poynton said. “They definitely exist, and I think we need to work harder at how are going to identify these patients prospectively in the future, so we know which of our patients who are older can benefit from intensive chemotherapy and which patients won’t.”
Dr. Poynton reported having no relevant financial disclosures. His coinvestigators reported relationships with AbbVie, Merck, Takeda, Jazz Pharmaceuticals, and others.
GLASGOW – Most older patients with primary central nervous system lymphoma (PCNSL) can tolerate high-dose methotrexate-based chemotherapy and achieve similar outcomes as younger and fitter patients, according to a retrospective analysis of 244 patients in the United Kingdom.
For older patients – at least 65 years old – who received methotrexate-based regimens, treatment-related mortality was 6.8%, which is comparable with rates seen in trials involving younger patients, reported lead author Edward Poynton, MD, of University College Hospital in London.
Specifically, Dr. Poynton cited the phase 2 IELSG32 trial, which had a treatment-related mortality rate of 6% among patients up to age 70 years. These patients were treated with the established protocol for younger patients: chemotherapy with methotrexate, cytarabine, thiotepa, and rituximab (MATRix) followed by autologous stem cell transplant or whole-brain radiotherapy.
Introducing Dr. Poynton’s presentation at the annual meeting of the British Society for Haematology, Simon Rule, MD, of the University of Plymouth (England), added historical context to the new findings.
“When I started in hematology ... [PCNSL] was a universally fatal disease, pretty much,” Dr. Rule said. “And then we had methotrexate, and it worked occasionally. And then we had a randomized trial, which was randomization of methotrexate plus or minus high-dose cytarabine, showing benefit.”
This combination became the benchmark against which subsequent randomized trials were measured; however, such high-intensity regimens have raised concerns about safety and efficacy in older patients, Dr. Rule said, noting that the present study serves to inform clinicians about real-world outcomes in this population.
The retrospective analysis reviewed 244 patients who were aged at least 65 years when histologically diagnosed with PCNSL at 14 U.K. tertiary centers between 2012 and 2017. All patients received first-line care of any kind, ranging from best supportive care to clinical trial therapy. Patients were grouped into three treatment cohorts divided by level of frailty. Analysis showed that these divisions correlated with age, renal function, Eastern Cooperative Oncology Group performance status, and treatment intensity.
The frail group received palliative treatment consisting of whole-brain radiotherapy, an oral alkylator, or best supportive care. The less-fit group received methotrexate in combination with rituximab, an oral alkylator, or both. The fit group was most intensively treated, receiving high-dose methotrexate and cytarabine – with or without rituximab – or MATRix.
The primary objective was overall response rate, while the secondary objectives were median overall survival and progression-free survival.
The analysis showed that 79% of patients (n = 193) received methotrexate-based therapy of some kind, with 61% receiving three or more cycles of therapy and 30% requiring dose reductions. The overall response rate was 63%.
Dr. Poynton noted that about two-thirds of patients who achieved a partial response in early assessment went on to achieve a complete response. Patients in the fit group more often responded than those who were less fit (87% vs. 65%; P = .01) and more often received consolidation therapy (42% vs. 23%; P = .01).
Fitness level was also associated with median overall survival, which was longest in the fit group at 42 months. The other two groups had dramatically shorter survival times: 8 months in the less-fit group and just 2 months in the frail group.
A closer look at the data revealed some patterns, Dr. Poynton said.
“What we see is that age at diagnosis is significantly correlated with progression-free survival but not with overall survival,” he said, noting that, in contrast, performance status was associated with both survival measures.
Methotrexate dose also impacted both survival measures. Patients who received 75% or more of their induction dose over the course of treatment had better median overall survival and progression-free survival than those who received less than 75%. Similarly, consolidation therapy improved both survival measures.
Patients aged older than 70 years who received intensive chemotherapy had a treatment-related mortality rate of 4.8%, which is lower than the overall treatment-related mortality, Dr. Poynton reported.
Considering the correlation between methotrexate dose and survival, Dr. Poynton suggested that “dose reductions should be carefully considered.”
He also noted that patients in the fit cohort who received intensive chemotherapy had comparable outcomes with younger patients in prospective trials, and yet 44% of patients older than 65 years in the real world who received high-dose methotrexate with cytarabine would have been ineligible for the IELSG32 trial.
“We’ve been able to identify this cohort of patients retrospectively,” Dr. Poynton said. “They definitely exist, and I think we need to work harder at how are going to identify these patients prospectively in the future, so we know which of our patients who are older can benefit from intensive chemotherapy and which patients won’t.”
Dr. Poynton reported having no relevant financial disclosures. His coinvestigators reported relationships with AbbVie, Merck, Takeda, Jazz Pharmaceuticals, and others.
REPORTING FROM BSH 2019
Monitoring, early intervention key to CAR T safety
GLASGOW – Constant patient monitoring and early intervention with tocilizumab and steroids are essential to the safe delivery of chimeric antigen receptor (CAR) T-cell therapy in patients with non-Hodgkin lymphoma (NHL), according to a leading expert.
As a clinical researcher at MD Anderson Cancer Center in Houston, Loretta Nastoupil, MD has played an active role in the evolution of CAR T-cell therapy, from early trials to ongoing development of treatment protocols. During a presentation at the annual meeting of the British Society for Haematology, Dr. Nastoupil discussed leading topics in CAR T-cell therapy, with an emphasis on safe delivery.
“[Toxicity] is something we don’t talk about as much as we should, partly because this therapy works and it’s really exciting,” Dr. Nastoupil said. “But the toxicity is not something that I minimize, and it’s very challenging. It’s led us to restructure our inpatient services. It’s led to a lot of sleepless nights. These patients can do very, very well, or they can do very, very poorly in terms of toxicity and I think the most important strategy is recognition and early intervention.”
Monitoring
Early recognition depends on close monitoring, Dr. Nastoupil said, which is carried out by highly trained nursing staff who follow therapy-specific decision algorithms.
“We have nurses that are on the front line,” Dr. Nastoupil said. “They’re the most important group. We have staff that round on [patients] daily, but the nurses are there 24 hours a day. We have a flow sheet where they grade cytokine release syndrome and neurotoxicity every 8 hours, or if there is an acute change in symptoms or toxicity, they’ll do it in real time.”
Dr. Nastoupil said that if these toxicities are detected, intervention is occurring sooner than it did with some of the first patients to receive CAR-T cell therapy.
“Initially there was a lot of fear surrounding anything that would abort the CAR-T cell therapy,” Dr. Nastoupil said. “There was concern that if you were trying to mitigate some of the toxicity you might have a negative impact on efficacy ... [W]ith the first iteration of studies, generally we were waiting until grade 3 or higher cytokine release syndrome before initiating either tocilizumab and/or steroids. As the studies evolved, it started to move into grade 2 toxicity that we started using therapy, mostly because we started to see that those patients were still responding.”
At MD Anderson, these earlier interventions have decreased severity of adverse events.
“It’s rare nowadays to have grade 3 or 4 cytokine release syndrome because we are generally introducing abortive therapy at grade 2,” Dr. Nastoupil said, citing increased use of steroids and tocilizumab.
Currently, no consensus exists for managing these events, partly because clinicians are still learning about best management practices.
“There will be a consensus on management,” Dr. Nastoupil said. “I think that’s needed. The problem is, it will probably evolve as we get more experience with managing these patients. I think there’s been a little hesitation to put something out on paper knowing that a year from now that might change.”
Grading toxicity
In contrast, Dr. Nastoupil said that a consensus has been reached for grading acute toxicity. Of note, fever is now considered an essential element of cytokine release syndrome.
“The first thing we see [with cytokine release syndrome] is fever, generally speaking,” Dr. Nastoupil said. “That will prompt a workup for infection because these patients are going to be neutropenic. And we initiate broad spectrum antimicrobials.”
She said that some patients treated with CAR T-cell therapy have had disseminated fungal infections, so clinicians need to be on the lookout for septic shock.
To assess neurotoxicity, the team at MD Anderson uses an objective scoring system, called “CARTOX.” This helps maintain consistency when facing broadly different neurological presentations.
“There’s such a wide ranging spectrum of patients that are undergoing neurotoxicity you can’t expect someone, even myself, to be consistent when you are trying to tease out how serious it is,” Dr. Nastoupil said.
With CARTOX, nurses can easily score patients and call clinicians with results. Still, this doesn’t eliminate difficulties inherent to managing neurotoxicity, particularly when it is severe.
“I’d say one of the areas that is still very challenging is when [patients with neurotoxicity] are no longer responding,” Dr. Nastoupil said. “You have to be very mindful of seizure activity. We’ve had a couple of patients with status [epilepticus]. You don’t see seizure activity physically, but when you do an EEG, you pick it up.”
Dr. Nastoupil added that most centers are now giving patients prophylactic levetiracetam (Keppra) to lower seizure risk.
Choosing therapy
When selecting between the two therapies currently approved by the Food and Drug Administration – tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta) – based on safety, Dr. Nastoupil said that rates of cytokine release syndrome appear similar, but neurotoxicity rates may differ.
“Cytokine release syndrome in my opinion is probably more similar than different in terms of grade 3 or higher because tocilizumab and steroids work quite well in aborting those toxicities,” Dr. Nastoupil said. “But neurotoxicity still sticks out in my mind as the most striking difference, where with axicabtagene you see more grade 3 or higher neurotoxicity, though very, very few deaths as a result of this. But it’s very challenging in terms of management.”
According to Dr. Nastoupil, comparisons between CAR T-cell therapies have been complicated by differences in clinical trial methodologies. However, she offered a general conclusion regarding efficacy.
“[W]hat I’ll tell you, at the end of the day, is [that existing CAR T-cell therapies] all seem to sort of settle out around 30%-40% in terms of durable responses,” Dr. Nastoupil said.
Dr. Nastoupil concluded her presentation with an overview and look to the future.
“I do think [CAR T-cell therapy] is transformative, particularly for our chemo refractory patients,” she said. “There is nothing else like it. The problem right now is that it is only durable in 40% of patients. So can we be better at selecting out patients that are more likely to respond? Does introducing this in earlier lines of therapy increase that fraction of patients that are potentially cured?”
Considering these questions, she said: “We need more patients. We need more data. We need longer follow-up to understand the nuances of this therapy.”
Dr. Nastoupil previously reported financial relationships with Celgene, Genentech, Gilead, Merck, Novartis, Spectrum, and TG Therapeutics.
GLASGOW – Constant patient monitoring and early intervention with tocilizumab and steroids are essential to the safe delivery of chimeric antigen receptor (CAR) T-cell therapy in patients with non-Hodgkin lymphoma (NHL), according to a leading expert.
As a clinical researcher at MD Anderson Cancer Center in Houston, Loretta Nastoupil, MD has played an active role in the evolution of CAR T-cell therapy, from early trials to ongoing development of treatment protocols. During a presentation at the annual meeting of the British Society for Haematology, Dr. Nastoupil discussed leading topics in CAR T-cell therapy, with an emphasis on safe delivery.
“[Toxicity] is something we don’t talk about as much as we should, partly because this therapy works and it’s really exciting,” Dr. Nastoupil said. “But the toxicity is not something that I minimize, and it’s very challenging. It’s led us to restructure our inpatient services. It’s led to a lot of sleepless nights. These patients can do very, very well, or they can do very, very poorly in terms of toxicity and I think the most important strategy is recognition and early intervention.”
Monitoring
Early recognition depends on close monitoring, Dr. Nastoupil said, which is carried out by highly trained nursing staff who follow therapy-specific decision algorithms.
“We have nurses that are on the front line,” Dr. Nastoupil said. “They’re the most important group. We have staff that round on [patients] daily, but the nurses are there 24 hours a day. We have a flow sheet where they grade cytokine release syndrome and neurotoxicity every 8 hours, or if there is an acute change in symptoms or toxicity, they’ll do it in real time.”
Dr. Nastoupil said that if these toxicities are detected, intervention is occurring sooner than it did with some of the first patients to receive CAR-T cell therapy.
“Initially there was a lot of fear surrounding anything that would abort the CAR-T cell therapy,” Dr. Nastoupil said. “There was concern that if you were trying to mitigate some of the toxicity you might have a negative impact on efficacy ... [W]ith the first iteration of studies, generally we were waiting until grade 3 or higher cytokine release syndrome before initiating either tocilizumab and/or steroids. As the studies evolved, it started to move into grade 2 toxicity that we started using therapy, mostly because we started to see that those patients were still responding.”
At MD Anderson, these earlier interventions have decreased severity of adverse events.
“It’s rare nowadays to have grade 3 or 4 cytokine release syndrome because we are generally introducing abortive therapy at grade 2,” Dr. Nastoupil said, citing increased use of steroids and tocilizumab.
Currently, no consensus exists for managing these events, partly because clinicians are still learning about best management practices.
“There will be a consensus on management,” Dr. Nastoupil said. “I think that’s needed. The problem is, it will probably evolve as we get more experience with managing these patients. I think there’s been a little hesitation to put something out on paper knowing that a year from now that might change.”
Grading toxicity
In contrast, Dr. Nastoupil said that a consensus has been reached for grading acute toxicity. Of note, fever is now considered an essential element of cytokine release syndrome.
“The first thing we see [with cytokine release syndrome] is fever, generally speaking,” Dr. Nastoupil said. “That will prompt a workup for infection because these patients are going to be neutropenic. And we initiate broad spectrum antimicrobials.”
She said that some patients treated with CAR T-cell therapy have had disseminated fungal infections, so clinicians need to be on the lookout for septic shock.
To assess neurotoxicity, the team at MD Anderson uses an objective scoring system, called “CARTOX.” This helps maintain consistency when facing broadly different neurological presentations.
“There’s such a wide ranging spectrum of patients that are undergoing neurotoxicity you can’t expect someone, even myself, to be consistent when you are trying to tease out how serious it is,” Dr. Nastoupil said.
With CARTOX, nurses can easily score patients and call clinicians with results. Still, this doesn’t eliminate difficulties inherent to managing neurotoxicity, particularly when it is severe.
“I’d say one of the areas that is still very challenging is when [patients with neurotoxicity] are no longer responding,” Dr. Nastoupil said. “You have to be very mindful of seizure activity. We’ve had a couple of patients with status [epilepticus]. You don’t see seizure activity physically, but when you do an EEG, you pick it up.”
Dr. Nastoupil added that most centers are now giving patients prophylactic levetiracetam (Keppra) to lower seizure risk.
Choosing therapy
When selecting between the two therapies currently approved by the Food and Drug Administration – tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta) – based on safety, Dr. Nastoupil said that rates of cytokine release syndrome appear similar, but neurotoxicity rates may differ.
“Cytokine release syndrome in my opinion is probably more similar than different in terms of grade 3 or higher because tocilizumab and steroids work quite well in aborting those toxicities,” Dr. Nastoupil said. “But neurotoxicity still sticks out in my mind as the most striking difference, where with axicabtagene you see more grade 3 or higher neurotoxicity, though very, very few deaths as a result of this. But it’s very challenging in terms of management.”
According to Dr. Nastoupil, comparisons between CAR T-cell therapies have been complicated by differences in clinical trial methodologies. However, she offered a general conclusion regarding efficacy.
“[W]hat I’ll tell you, at the end of the day, is [that existing CAR T-cell therapies] all seem to sort of settle out around 30%-40% in terms of durable responses,” Dr. Nastoupil said.
Dr. Nastoupil concluded her presentation with an overview and look to the future.
“I do think [CAR T-cell therapy] is transformative, particularly for our chemo refractory patients,” she said. “There is nothing else like it. The problem right now is that it is only durable in 40% of patients. So can we be better at selecting out patients that are more likely to respond? Does introducing this in earlier lines of therapy increase that fraction of patients that are potentially cured?”
Considering these questions, she said: “We need more patients. We need more data. We need longer follow-up to understand the nuances of this therapy.”
Dr. Nastoupil previously reported financial relationships with Celgene, Genentech, Gilead, Merck, Novartis, Spectrum, and TG Therapeutics.
GLASGOW – Constant patient monitoring and early intervention with tocilizumab and steroids are essential to the safe delivery of chimeric antigen receptor (CAR) T-cell therapy in patients with non-Hodgkin lymphoma (NHL), according to a leading expert.
As a clinical researcher at MD Anderson Cancer Center in Houston, Loretta Nastoupil, MD has played an active role in the evolution of CAR T-cell therapy, from early trials to ongoing development of treatment protocols. During a presentation at the annual meeting of the British Society for Haematology, Dr. Nastoupil discussed leading topics in CAR T-cell therapy, with an emphasis on safe delivery.
“[Toxicity] is something we don’t talk about as much as we should, partly because this therapy works and it’s really exciting,” Dr. Nastoupil said. “But the toxicity is not something that I minimize, and it’s very challenging. It’s led us to restructure our inpatient services. It’s led to a lot of sleepless nights. These patients can do very, very well, or they can do very, very poorly in terms of toxicity and I think the most important strategy is recognition and early intervention.”
Monitoring
Early recognition depends on close monitoring, Dr. Nastoupil said, which is carried out by highly trained nursing staff who follow therapy-specific decision algorithms.
“We have nurses that are on the front line,” Dr. Nastoupil said. “They’re the most important group. We have staff that round on [patients] daily, but the nurses are there 24 hours a day. We have a flow sheet where they grade cytokine release syndrome and neurotoxicity every 8 hours, or if there is an acute change in symptoms or toxicity, they’ll do it in real time.”
Dr. Nastoupil said that if these toxicities are detected, intervention is occurring sooner than it did with some of the first patients to receive CAR-T cell therapy.
“Initially there was a lot of fear surrounding anything that would abort the CAR-T cell therapy,” Dr. Nastoupil said. “There was concern that if you were trying to mitigate some of the toxicity you might have a negative impact on efficacy ... [W]ith the first iteration of studies, generally we were waiting until grade 3 or higher cytokine release syndrome before initiating either tocilizumab and/or steroids. As the studies evolved, it started to move into grade 2 toxicity that we started using therapy, mostly because we started to see that those patients were still responding.”
At MD Anderson, these earlier interventions have decreased severity of adverse events.
“It’s rare nowadays to have grade 3 or 4 cytokine release syndrome because we are generally introducing abortive therapy at grade 2,” Dr. Nastoupil said, citing increased use of steroids and tocilizumab.
Currently, no consensus exists for managing these events, partly because clinicians are still learning about best management practices.
“There will be a consensus on management,” Dr. Nastoupil said. “I think that’s needed. The problem is, it will probably evolve as we get more experience with managing these patients. I think there’s been a little hesitation to put something out on paper knowing that a year from now that might change.”
Grading toxicity
In contrast, Dr. Nastoupil said that a consensus has been reached for grading acute toxicity. Of note, fever is now considered an essential element of cytokine release syndrome.
“The first thing we see [with cytokine release syndrome] is fever, generally speaking,” Dr. Nastoupil said. “That will prompt a workup for infection because these patients are going to be neutropenic. And we initiate broad spectrum antimicrobials.”
She said that some patients treated with CAR T-cell therapy have had disseminated fungal infections, so clinicians need to be on the lookout for septic shock.
To assess neurotoxicity, the team at MD Anderson uses an objective scoring system, called “CARTOX.” This helps maintain consistency when facing broadly different neurological presentations.
“There’s such a wide ranging spectrum of patients that are undergoing neurotoxicity you can’t expect someone, even myself, to be consistent when you are trying to tease out how serious it is,” Dr. Nastoupil said.
With CARTOX, nurses can easily score patients and call clinicians with results. Still, this doesn’t eliminate difficulties inherent to managing neurotoxicity, particularly when it is severe.
“I’d say one of the areas that is still very challenging is when [patients with neurotoxicity] are no longer responding,” Dr. Nastoupil said. “You have to be very mindful of seizure activity. We’ve had a couple of patients with status [epilepticus]. You don’t see seizure activity physically, but when you do an EEG, you pick it up.”
Dr. Nastoupil added that most centers are now giving patients prophylactic levetiracetam (Keppra) to lower seizure risk.
Choosing therapy
When selecting between the two therapies currently approved by the Food and Drug Administration – tisagenlecleucel (Kymriah) and axicabtagene ciloleucel (Yescarta) – based on safety, Dr. Nastoupil said that rates of cytokine release syndrome appear similar, but neurotoxicity rates may differ.
“Cytokine release syndrome in my opinion is probably more similar than different in terms of grade 3 or higher because tocilizumab and steroids work quite well in aborting those toxicities,” Dr. Nastoupil said. “But neurotoxicity still sticks out in my mind as the most striking difference, where with axicabtagene you see more grade 3 or higher neurotoxicity, though very, very few deaths as a result of this. But it’s very challenging in terms of management.”
According to Dr. Nastoupil, comparisons between CAR T-cell therapies have been complicated by differences in clinical trial methodologies. However, she offered a general conclusion regarding efficacy.
“[W]hat I’ll tell you, at the end of the day, is [that existing CAR T-cell therapies] all seem to sort of settle out around 30%-40% in terms of durable responses,” Dr. Nastoupil said.
Dr. Nastoupil concluded her presentation with an overview and look to the future.
“I do think [CAR T-cell therapy] is transformative, particularly for our chemo refractory patients,” she said. “There is nothing else like it. The problem right now is that it is only durable in 40% of patients. So can we be better at selecting out patients that are more likely to respond? Does introducing this in earlier lines of therapy increase that fraction of patients that are potentially cured?”
Considering these questions, she said: “We need more patients. We need more data. We need longer follow-up to understand the nuances of this therapy.”
Dr. Nastoupil previously reported financial relationships with Celgene, Genentech, Gilead, Merck, Novartis, Spectrum, and TG Therapeutics.
EXPERT ANALYSIS FROM BSH 2019
TMS is associated with improved recollection in older adults
according to a small pilot study published online ahead of print April 17 in Neurology. Stimulation also increased functional MRI signals associated with recollection throughout the hippocampal-cortical network.
“Disruption and abnormal functioning of the hippocampal-cortical network, the region of the brain involved in memory formation, has been linked to age-related memory decline, so it’s exciting to see that, by targeting this region, magnetic stimulation may help improve memory in older adults,” said Joel L. Voss, PhD, of Northwestern University in Chicago. “These results may help us better understand how this network supports memory.”
Recollection is the type of memory most impaired during normal aging. Other types of memory, such as recognition, are relatively spared. Research indicates that multiple sessions of TMS improve recollection and hippocampal-cortical network function in young adults.
Dr. Voss and colleagues performed a pilot study to evaluate whether TMS could improve recollection in older adults. They enrolled 15 cognitively normal older adults (mean age, 72.46 years) into a sham-controlled, single-blind, counterbalanced experiment. Eleven subjects were women. Participants underwent fMRI while learning objects paired with scenes and locations. Investigators administered TMS to lateral parietal locations that were based on each participant’s fMRI connectivity with the hippocampus. Using a within-subjects crossover design, Dr. Voss’s group assessed participants’ recollection and recognition memory at baseline and 24 hours and also 1 week after five consecutive daily sessions of full-intensity stimulation, compared with low-intensity sham stimulation.
At baseline, participants had impaired recollection, but not impaired recognition, compared with a historical sample of younger adults. At 24 hours, TMS provided robust recollection improvement and weak recognition improvement, compared with sham. TMS improved recollection by 31.1% from baseline, and sham yielded a nonsignificant change of −3.1%. Recollection improvements after TMS were consistent across participants. Recognition changed by a nonsignificant 2.8% following TMS and by a nonsignificant −2.9% following sham.
The investigators also found a significant and consistent increase in fMRI recollection activity for the targeted hippocampal-cortical network, but not for the control frontal-parietal network. They observed no increased fMRI activity for recognition.
“These findings demonstrate a causal link between recollection and the hippocampal-cortical network in older adults,” said Dr. Voss. “While our small study examined age-related memory loss, it did not examine this stimulation in people with memory loss from more serious conditions such as mild cognitive impairment or Alzheimer’s disease.” Furthermore, the study was designed to test for neural and behavioral target engagement, but not clinical efficacy, he added.
The study’s limitations included its small sample size, its single-site design, and its lack of active control stimulation. Nevertheless, it identified specific and consistent effects of stimulation across participants that were consistent with previous findings in younger adults. “These findings motivate future studies to optimize the effectiveness of noninvasive stimulation for treatment of age-related memory impairment and to improve mechanistic understanding of the hippocampal-cortical networks that support episodic memory across the lifespan,” said Dr. Voss.
The National Institute on Aging, as well as the Northwestern University Cognitive Neurology and Alzheimer’s Disease Center, supported the study.
SOURCE: Nilakantan AS et al. Neurology. 2019 Apr 17 doi: 10.1212/WNL.0000000000007502.
according to a small pilot study published online ahead of print April 17 in Neurology. Stimulation also increased functional MRI signals associated with recollection throughout the hippocampal-cortical network.
“Disruption and abnormal functioning of the hippocampal-cortical network, the region of the brain involved in memory formation, has been linked to age-related memory decline, so it’s exciting to see that, by targeting this region, magnetic stimulation may help improve memory in older adults,” said Joel L. Voss, PhD, of Northwestern University in Chicago. “These results may help us better understand how this network supports memory.”
Recollection is the type of memory most impaired during normal aging. Other types of memory, such as recognition, are relatively spared. Research indicates that multiple sessions of TMS improve recollection and hippocampal-cortical network function in young adults.
Dr. Voss and colleagues performed a pilot study to evaluate whether TMS could improve recollection in older adults. They enrolled 15 cognitively normal older adults (mean age, 72.46 years) into a sham-controlled, single-blind, counterbalanced experiment. Eleven subjects were women. Participants underwent fMRI while learning objects paired with scenes and locations. Investigators administered TMS to lateral parietal locations that were based on each participant’s fMRI connectivity with the hippocampus. Using a within-subjects crossover design, Dr. Voss’s group assessed participants’ recollection and recognition memory at baseline and 24 hours and also 1 week after five consecutive daily sessions of full-intensity stimulation, compared with low-intensity sham stimulation.
At baseline, participants had impaired recollection, but not impaired recognition, compared with a historical sample of younger adults. At 24 hours, TMS provided robust recollection improvement and weak recognition improvement, compared with sham. TMS improved recollection by 31.1% from baseline, and sham yielded a nonsignificant change of −3.1%. Recollection improvements after TMS were consistent across participants. Recognition changed by a nonsignificant 2.8% following TMS and by a nonsignificant −2.9% following sham.
The investigators also found a significant and consistent increase in fMRI recollection activity for the targeted hippocampal-cortical network, but not for the control frontal-parietal network. They observed no increased fMRI activity for recognition.
“These findings demonstrate a causal link between recollection and the hippocampal-cortical network in older adults,” said Dr. Voss. “While our small study examined age-related memory loss, it did not examine this stimulation in people with memory loss from more serious conditions such as mild cognitive impairment or Alzheimer’s disease.” Furthermore, the study was designed to test for neural and behavioral target engagement, but not clinical efficacy, he added.
The study’s limitations included its small sample size, its single-site design, and its lack of active control stimulation. Nevertheless, it identified specific and consistent effects of stimulation across participants that were consistent with previous findings in younger adults. “These findings motivate future studies to optimize the effectiveness of noninvasive stimulation for treatment of age-related memory impairment and to improve mechanistic understanding of the hippocampal-cortical networks that support episodic memory across the lifespan,” said Dr. Voss.
The National Institute on Aging, as well as the Northwestern University Cognitive Neurology and Alzheimer’s Disease Center, supported the study.
SOURCE: Nilakantan AS et al. Neurology. 2019 Apr 17 doi: 10.1212/WNL.0000000000007502.
according to a small pilot study published online ahead of print April 17 in Neurology. Stimulation also increased functional MRI signals associated with recollection throughout the hippocampal-cortical network.
“Disruption and abnormal functioning of the hippocampal-cortical network, the region of the brain involved in memory formation, has been linked to age-related memory decline, so it’s exciting to see that, by targeting this region, magnetic stimulation may help improve memory in older adults,” said Joel L. Voss, PhD, of Northwestern University in Chicago. “These results may help us better understand how this network supports memory.”
Recollection is the type of memory most impaired during normal aging. Other types of memory, such as recognition, are relatively spared. Research indicates that multiple sessions of TMS improve recollection and hippocampal-cortical network function in young adults.
Dr. Voss and colleagues performed a pilot study to evaluate whether TMS could improve recollection in older adults. They enrolled 15 cognitively normal older adults (mean age, 72.46 years) into a sham-controlled, single-blind, counterbalanced experiment. Eleven subjects were women. Participants underwent fMRI while learning objects paired with scenes and locations. Investigators administered TMS to lateral parietal locations that were based on each participant’s fMRI connectivity with the hippocampus. Using a within-subjects crossover design, Dr. Voss’s group assessed participants’ recollection and recognition memory at baseline and 24 hours and also 1 week after five consecutive daily sessions of full-intensity stimulation, compared with low-intensity sham stimulation.
At baseline, participants had impaired recollection, but not impaired recognition, compared with a historical sample of younger adults. At 24 hours, TMS provided robust recollection improvement and weak recognition improvement, compared with sham. TMS improved recollection by 31.1% from baseline, and sham yielded a nonsignificant change of −3.1%. Recollection improvements after TMS were consistent across participants. Recognition changed by a nonsignificant 2.8% following TMS and by a nonsignificant −2.9% following sham.
The investigators also found a significant and consistent increase in fMRI recollection activity for the targeted hippocampal-cortical network, but not for the control frontal-parietal network. They observed no increased fMRI activity for recognition.
“These findings demonstrate a causal link between recollection and the hippocampal-cortical network in older adults,” said Dr. Voss. “While our small study examined age-related memory loss, it did not examine this stimulation in people with memory loss from more serious conditions such as mild cognitive impairment or Alzheimer’s disease.” Furthermore, the study was designed to test for neural and behavioral target engagement, but not clinical efficacy, he added.
The study’s limitations included its small sample size, its single-site design, and its lack of active control stimulation. Nevertheless, it identified specific and consistent effects of stimulation across participants that were consistent with previous findings in younger adults. “These findings motivate future studies to optimize the effectiveness of noninvasive stimulation for treatment of age-related memory impairment and to improve mechanistic understanding of the hippocampal-cortical networks that support episodic memory across the lifespan,” said Dr. Voss.
The National Institute on Aging, as well as the Northwestern University Cognitive Neurology and Alzheimer’s Disease Center, supported the study.
SOURCE: Nilakantan AS et al. Neurology. 2019 Apr 17 doi: 10.1212/WNL.0000000000007502.
FROM NEUROLOGY
Plasma levels of neurofilament light track neurodegeneration in MCI and Alzheimer’s disease
Plasma levels of the axonal protein also correlated with its presence in cerebrospinal fluid, and mirrored the changes in amyloid beta (Abeta) 42, Niklas Mattsson, MD, and colleagues wrote in JAMA Neurology.
“Taken together, these findings suggest that the neurofilament light level is a dynamic biomarker that changes throughout the course of Alzheimer’s disease and is sensitive to progressive neurodegeneration,” wrote Dr. Mattsson of Lund (Sweden) University and his coauthors. “This has important implications, given the unmet need for noninvasive blood-based methods to objectively track longitudinal neurodegeneration in Alzheimer’s disease.”
A blood-based biomarker of Alzheimer’s disease progression could open a new door for drug trials, the authors noted. Previously, NfL levels have only been available by lumbar puncture – a invasive and expensive test that many patients resist. If NfL plasma levels do reliably track dementia progression, the test could become a standard part of clinical trials, providing regular drug response data as the study progresses.
Neurofilaments are polypeptides that give structure to the neuronal cytoskeleton and regulate microtubule function. Injured cells release the protein very quickly. Neurofilaments are elevated in traumatic brain injury, multiple sclerosis, and some psychiatric illnesses. Neurofilament levels have even been used to predict neurologic recovery after cardiac arrest.
Dr. Mattsson and his team used data obtained over 11 years from 1,583 subjects enrolled in the Alzheimer’s Disease Neuroimaging Initiative study. The sample comprised three groups: cognitively unimpaired controls (401), patients with MCI (855), and patients with Alzheimer’s dementia (327). The investigators analyzed 4,326 samples.
In addition to the NfL measurements, they tracked Abeta and tau in cerebrospinal fluid (CSF), structural brain changes by 18fluorodeoxyglucose (FDG)–PET and MRI, and cognitive and functional performance. The primary outcome was NfL’s association with these changes. The team set the lower limit of NfL as 6.7 ng/L and the upper, 1,620 ng/L.
At baseline, only advancing age correlated with NfL levels. But it was significantly higher in patients with MCI (37.9 ng/L) and Alzheimer’s dementia (45.9 ng/L) than it was in the control subjects (32.1 ng/L). Over the years of follow-up, levels increased in all groups, but NfL increased more rapidly among patients with MCI and Alzheimer’s dementia than controls (2.7 vs. 2.4 ng/L per year). The difference was most pronounced when comparing levels in patients with Alzheimer’s dementia and MCI with controls. However, control subjects who were Abeta positive by CSF had greater NfL changes than Abeta-negative controls.
Baseline measures of CSF Abeta and tau, as well as hippocampal and ventricular volume and cortical thickness, also correlated with NfL levels, as did cognition and functional scores.
During follow-up, the diagnostic groups showed different NfL trajectories, which correlated strongly with the other measures.
In the control group, NfL increases correlated with lower FDG-PET measures, lower CSF Abeta, reduced hippocampal volume, and higher ventricular volume. Among patients with MCI, NfL increases correlated most strongly with hippocampal volume, temporal region, and cognition. Among patients with Alzheimer’s dementia, the NfL increase most strongly tracked cognitive decline,
When the investigators applied these findings to the A/T/N (amyloid, tau, neurodegeneration) classification system, NfL most often correlated with neurodegeneration, but not always. This might suggest that the neuronal damage occurred separately from Abeta changes. In all three groups, rapid NfL increases mirrored the rate of change in most other measures.
“In controls and patients with MCI and Alzheimer’s dementia, greater rates of NfL were associated with accelerated reduction in FDG-PET measures … expansion of ventricular volume,” and a reduction in cognitive and functional performance, Dr. Mattsson and his colleagues wrote.“ In addition, greater increases in NfL levels were associated with accelerated loss of hippocampal volume and entorhinal cortical thickness in controls and patients with MCI and with accelerated increases in total tau level, phosphorylated tau level, and white matter lesions in patients with MCI. In general, the concentrations of blood-based NfL appears to reflect the intensity of the neuronal injury.”
Dr Mattsson reported being a consultant for the Alzheimer’s Disease Neuroimaging Initiative.
SOURCE: Mattsson N et al. JAMA Neurol. 2019 Apr 22. doi: 10.1001/jamaneurol.2019.0765.
This is an impressive study that convincingly demonstrates the sensitivity of plasma neurofilament light (NfL) to disease progression in patients with mild cognitive impairment and dementia in the Alzheimer’s Disease Neuroimaging Initiative cohort.
It is known that NfL is sensitive to neuronal damage that can result from a variety of pathologies, not limited to Alzheimer’s disease, so it is not diagnostically specific. In the present study there was even overlap of values between the cognitively unimpaired and mild cognitive impairment groups at baseline as well, although the levels separated over time.
In the right setting, NfL might still be a clinically useful diagnostic marker, especially for mild-stage disease when other clinical measures such as mental status scores and structural brain scans are inconclusive, and further study seems warranted for such a possibility.
Its greatest utility, however, will likely be in clinical trials. It would have been of the greatest interest to know how NfL levels changed in the setting of demonstrated cerebral amyloid clearance by agents such as aducanumab that failed to halt dementia progression, or in the setting of BACE1 inhibitor-related worsening of cognition. Did NfL levels remain static in the former and rise in the latter? Looking ahead, it seems likely that this easily accessed biomarker will become an integral part of clinical trial design. Assuming cost is not overly burdensome, it may even find its way eventually into clinical practice.
Dr. Caselli is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
This is an impressive study that convincingly demonstrates the sensitivity of plasma neurofilament light (NfL) to disease progression in patients with mild cognitive impairment and dementia in the Alzheimer’s Disease Neuroimaging Initiative cohort.
It is known that NfL is sensitive to neuronal damage that can result from a variety of pathologies, not limited to Alzheimer’s disease, so it is not diagnostically specific. In the present study there was even overlap of values between the cognitively unimpaired and mild cognitive impairment groups at baseline as well, although the levels separated over time.
In the right setting, NfL might still be a clinically useful diagnostic marker, especially for mild-stage disease when other clinical measures such as mental status scores and structural brain scans are inconclusive, and further study seems warranted for such a possibility.
Its greatest utility, however, will likely be in clinical trials. It would have been of the greatest interest to know how NfL levels changed in the setting of demonstrated cerebral amyloid clearance by agents such as aducanumab that failed to halt dementia progression, or in the setting of BACE1 inhibitor-related worsening of cognition. Did NfL levels remain static in the former and rise in the latter? Looking ahead, it seems likely that this easily accessed biomarker will become an integral part of clinical trial design. Assuming cost is not overly burdensome, it may even find its way eventually into clinical practice.
Dr. Caselli is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
This is an impressive study that convincingly demonstrates the sensitivity of plasma neurofilament light (NfL) to disease progression in patients with mild cognitive impairment and dementia in the Alzheimer’s Disease Neuroimaging Initiative cohort.
It is known that NfL is sensitive to neuronal damage that can result from a variety of pathologies, not limited to Alzheimer’s disease, so it is not diagnostically specific. In the present study there was even overlap of values between the cognitively unimpaired and mild cognitive impairment groups at baseline as well, although the levels separated over time.
In the right setting, NfL might still be a clinically useful diagnostic marker, especially for mild-stage disease when other clinical measures such as mental status scores and structural brain scans are inconclusive, and further study seems warranted for such a possibility.
Its greatest utility, however, will likely be in clinical trials. It would have been of the greatest interest to know how NfL levels changed in the setting of demonstrated cerebral amyloid clearance by agents such as aducanumab that failed to halt dementia progression, or in the setting of BACE1 inhibitor-related worsening of cognition. Did NfL levels remain static in the former and rise in the latter? Looking ahead, it seems likely that this easily accessed biomarker will become an integral part of clinical trial design. Assuming cost is not overly burdensome, it may even find its way eventually into clinical practice.
Dr. Caselli is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center.
Plasma levels of the axonal protein also correlated with its presence in cerebrospinal fluid, and mirrored the changes in amyloid beta (Abeta) 42, Niklas Mattsson, MD, and colleagues wrote in JAMA Neurology.
“Taken together, these findings suggest that the neurofilament light level is a dynamic biomarker that changes throughout the course of Alzheimer’s disease and is sensitive to progressive neurodegeneration,” wrote Dr. Mattsson of Lund (Sweden) University and his coauthors. “This has important implications, given the unmet need for noninvasive blood-based methods to objectively track longitudinal neurodegeneration in Alzheimer’s disease.”
A blood-based biomarker of Alzheimer’s disease progression could open a new door for drug trials, the authors noted. Previously, NfL levels have only been available by lumbar puncture – a invasive and expensive test that many patients resist. If NfL plasma levels do reliably track dementia progression, the test could become a standard part of clinical trials, providing regular drug response data as the study progresses.
Neurofilaments are polypeptides that give structure to the neuronal cytoskeleton and regulate microtubule function. Injured cells release the protein very quickly. Neurofilaments are elevated in traumatic brain injury, multiple sclerosis, and some psychiatric illnesses. Neurofilament levels have even been used to predict neurologic recovery after cardiac arrest.
Dr. Mattsson and his team used data obtained over 11 years from 1,583 subjects enrolled in the Alzheimer’s Disease Neuroimaging Initiative study. The sample comprised three groups: cognitively unimpaired controls (401), patients with MCI (855), and patients with Alzheimer’s dementia (327). The investigators analyzed 4,326 samples.
In addition to the NfL measurements, they tracked Abeta and tau in cerebrospinal fluid (CSF), structural brain changes by 18fluorodeoxyglucose (FDG)–PET and MRI, and cognitive and functional performance. The primary outcome was NfL’s association with these changes. The team set the lower limit of NfL as 6.7 ng/L and the upper, 1,620 ng/L.
At baseline, only advancing age correlated with NfL levels. But it was significantly higher in patients with MCI (37.9 ng/L) and Alzheimer’s dementia (45.9 ng/L) than it was in the control subjects (32.1 ng/L). Over the years of follow-up, levels increased in all groups, but NfL increased more rapidly among patients with MCI and Alzheimer’s dementia than controls (2.7 vs. 2.4 ng/L per year). The difference was most pronounced when comparing levels in patients with Alzheimer’s dementia and MCI with controls. However, control subjects who were Abeta positive by CSF had greater NfL changes than Abeta-negative controls.
Baseline measures of CSF Abeta and tau, as well as hippocampal and ventricular volume and cortical thickness, also correlated with NfL levels, as did cognition and functional scores.
During follow-up, the diagnostic groups showed different NfL trajectories, which correlated strongly with the other measures.
In the control group, NfL increases correlated with lower FDG-PET measures, lower CSF Abeta, reduced hippocampal volume, and higher ventricular volume. Among patients with MCI, NfL increases correlated most strongly with hippocampal volume, temporal region, and cognition. Among patients with Alzheimer’s dementia, the NfL increase most strongly tracked cognitive decline,
When the investigators applied these findings to the A/T/N (amyloid, tau, neurodegeneration) classification system, NfL most often correlated with neurodegeneration, but not always. This might suggest that the neuronal damage occurred separately from Abeta changes. In all three groups, rapid NfL increases mirrored the rate of change in most other measures.
“In controls and patients with MCI and Alzheimer’s dementia, greater rates of NfL were associated with accelerated reduction in FDG-PET measures … expansion of ventricular volume,” and a reduction in cognitive and functional performance, Dr. Mattsson and his colleagues wrote.“ In addition, greater increases in NfL levels were associated with accelerated loss of hippocampal volume and entorhinal cortical thickness in controls and patients with MCI and with accelerated increases in total tau level, phosphorylated tau level, and white matter lesions in patients with MCI. In general, the concentrations of blood-based NfL appears to reflect the intensity of the neuronal injury.”
Dr Mattsson reported being a consultant for the Alzheimer’s Disease Neuroimaging Initiative.
SOURCE: Mattsson N et al. JAMA Neurol. 2019 Apr 22. doi: 10.1001/jamaneurol.2019.0765.
Plasma levels of the axonal protein also correlated with its presence in cerebrospinal fluid, and mirrored the changes in amyloid beta (Abeta) 42, Niklas Mattsson, MD, and colleagues wrote in JAMA Neurology.
“Taken together, these findings suggest that the neurofilament light level is a dynamic biomarker that changes throughout the course of Alzheimer’s disease and is sensitive to progressive neurodegeneration,” wrote Dr. Mattsson of Lund (Sweden) University and his coauthors. “This has important implications, given the unmet need for noninvasive blood-based methods to objectively track longitudinal neurodegeneration in Alzheimer’s disease.”
A blood-based biomarker of Alzheimer’s disease progression could open a new door for drug trials, the authors noted. Previously, NfL levels have only been available by lumbar puncture – a invasive and expensive test that many patients resist. If NfL plasma levels do reliably track dementia progression, the test could become a standard part of clinical trials, providing regular drug response data as the study progresses.
Neurofilaments are polypeptides that give structure to the neuronal cytoskeleton and regulate microtubule function. Injured cells release the protein very quickly. Neurofilaments are elevated in traumatic brain injury, multiple sclerosis, and some psychiatric illnesses. Neurofilament levels have even been used to predict neurologic recovery after cardiac arrest.
Dr. Mattsson and his team used data obtained over 11 years from 1,583 subjects enrolled in the Alzheimer’s Disease Neuroimaging Initiative study. The sample comprised three groups: cognitively unimpaired controls (401), patients with MCI (855), and patients with Alzheimer’s dementia (327). The investigators analyzed 4,326 samples.
In addition to the NfL measurements, they tracked Abeta and tau in cerebrospinal fluid (CSF), structural brain changes by 18fluorodeoxyglucose (FDG)–PET and MRI, and cognitive and functional performance. The primary outcome was NfL’s association with these changes. The team set the lower limit of NfL as 6.7 ng/L and the upper, 1,620 ng/L.
At baseline, only advancing age correlated with NfL levels. But it was significantly higher in patients with MCI (37.9 ng/L) and Alzheimer’s dementia (45.9 ng/L) than it was in the control subjects (32.1 ng/L). Over the years of follow-up, levels increased in all groups, but NfL increased more rapidly among patients with MCI and Alzheimer’s dementia than controls (2.7 vs. 2.4 ng/L per year). The difference was most pronounced when comparing levels in patients with Alzheimer’s dementia and MCI with controls. However, control subjects who were Abeta positive by CSF had greater NfL changes than Abeta-negative controls.
Baseline measures of CSF Abeta and tau, as well as hippocampal and ventricular volume and cortical thickness, also correlated with NfL levels, as did cognition and functional scores.
During follow-up, the diagnostic groups showed different NfL trajectories, which correlated strongly with the other measures.
In the control group, NfL increases correlated with lower FDG-PET measures, lower CSF Abeta, reduced hippocampal volume, and higher ventricular volume. Among patients with MCI, NfL increases correlated most strongly with hippocampal volume, temporal region, and cognition. Among patients with Alzheimer’s dementia, the NfL increase most strongly tracked cognitive decline,
When the investigators applied these findings to the A/T/N (amyloid, tau, neurodegeneration) classification system, NfL most often correlated with neurodegeneration, but not always. This might suggest that the neuronal damage occurred separately from Abeta changes. In all three groups, rapid NfL increases mirrored the rate of change in most other measures.
“In controls and patients with MCI and Alzheimer’s dementia, greater rates of NfL were associated with accelerated reduction in FDG-PET measures … expansion of ventricular volume,” and a reduction in cognitive and functional performance, Dr. Mattsson and his colleagues wrote.“ In addition, greater increases in NfL levels were associated with accelerated loss of hippocampal volume and entorhinal cortical thickness in controls and patients with MCI and with accelerated increases in total tau level, phosphorylated tau level, and white matter lesions in patients with MCI. In general, the concentrations of blood-based NfL appears to reflect the intensity of the neuronal injury.”
Dr Mattsson reported being a consultant for the Alzheimer’s Disease Neuroimaging Initiative.
SOURCE: Mattsson N et al. JAMA Neurol. 2019 Apr 22. doi: 10.1001/jamaneurol.2019.0765.
FROM JAMA NEUROLOGY
Role of Diet in Treating Skin Conditions



Can immune checkpoint inhibitors treat PML?
investigators reported in the New England Journal of Medicine.
Three research teams described 10 cases in which patients with PML received pembrolizumab or nivolumab.
In one study, researchers administered pembrolizumab to eight adults with PML. Five patients had clinical improvement or stabilization, whereas 3 patients did not. Among the patients with clinical improvement, treatment led to reduced JC viral load in cerebrospinal fluid (CSF) and increased CD4+ and CD8+ anti–JC virus activity in vitro. Among patients without clinical improvement, treatment did not meaningfully change viral load or antiviral cellular immune response.
In a separate letter, researchers in Germany described an additional patient with PML who had clinical stabilization and no disease progression on MRI after treatment with pembrolizumab.
In another letter, researchers in France described a patient with PML whose condition improved after treatment with nivolumab.
“Do pembrolizumab and nivolumab fit the bill for treatment of PML? The current reports are encouraging but suggest that the presence of JC virus–specific T cells in the blood is a prerequisite for their use,” said Igor J. Koralnik, MD, of the department of neurological sciences at Rush University Medical Center in Chicago, in an accompanying editorial. “A controlled trial may be needed to determine whether immune checkpoint inhibitors are indeed able to keep JC virus in check in patients with PML.”
Reinvigorating T cells
Both monoclonal antibodies target programmed cell death protein 1 (PD-1), which inhibits T-cell proliferation and cytokine production when it binds its associated ligand, Dr. Koralnik said. Pembrolizumab and nivolumab block this inhibition and have been used to spur T-cell activity against tumors in patients with cancer.
PML, an often fatal brain infection caused by the JC virus in patients with immunosuppression, has no specific treatment. Management hinges on “recovery of the immune system, either by treating the underlying cause of immunosuppression or by discontinuing the use of immunosuppressive medications,” said Dr. Koralnik.
Pembrolizumab
Prior studies have found that PD-1 expression is elevated on T lymphocytes of patients with PML. To determine whether PD-1 blockade with pembrolizumab reinvigorates anti–JC virus immune activity in patients with PML, Irene Cortese, MD, of the National Institutes of Health’s Neuroimmunology Clinic and her research colleagues administered pembrolizumab at a dose of 2 mg/kg of body weight every 4-6 weeks to eight adults with PML. The patients received 1-3 doses, and each patient had a different underlying condition.
In all patients, treatment induced down-regulation of PD-1 expression on lymphocytes in CSF and peripheral blood, and five of the eight patients had clinical stabilization or improvement. Of the other three patients who did not improve, one had stabilized prior to treatment and remained stable. The other two patients died from PML.
Additional reports
Separately, Sebastian Rauer, MD, of Albert Ludwigs University in Freiburg, Germany, and his colleagues reported that a patient with PML whose symptoms culminated in mutism in February 2018 began speaking again after receiving five infusions of pembrolizumab over 10 weeks. “In addition, the size and number of lesions on MRI decreased, and JCV was no longer detectable in CSF,” Dr. Rauer and his colleagues wrote. “The patient has remained stable as of the end of March 2019, with persistent but abating psychomotor slowing, aphasia, and disorientation.”
Finally, Ondine Walter, of Toulouse (France) University Hospital and colleagues described the case of a 60-year-old woman with PML who received nivolumab on a compassionate-use basis. Two weeks after treatment, JC viral load in CSF and blood had decreased. “Starting 8 weeks after the initiation of nivolumab therapy, the patient’s neurologic symptoms and signs stabilized, and subsequently she showed improved alertness, and the ptosis and hemiplegia abated.”
Reason for caution
Prior studies, however, give reasons for caution when considering the potential use of immune checkpoint inhibitors to treat PML, Dr. Koralnik noted. In one case, a patient developed an inflammatory form of PML known as immune reconstitution inflammatory syndrome after receiving nivolumab (J Neurovirol. 2019 March 12. doi: 10.1007/s13365-019-00738-x). In addition, researchers have reported a case of PML that occurred after 1 year of nivolumab treatment, and four cases of PML related to nivolumab have been reported in pharmacovigilance databases (Emerg Infect Dis. 2018;24:1594-6). The cost and safety profiles of the medications also may be considerations, Dr. Koralnik said.
The study by Dr. Cortese and colleagues was funded by the National Institutes of Health, and the authors had no relevant disclosures. Some of the research letter authors disclosed grants and personal fees from pharmaceutical companies.
SOURCES: Cortese I et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMoa1815039; Rauer S et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMc1817193; Walter O et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMc1816198; Koralnik IJ. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMe1904140.
investigators reported in the New England Journal of Medicine.
Three research teams described 10 cases in which patients with PML received pembrolizumab or nivolumab.
In one study, researchers administered pembrolizumab to eight adults with PML. Five patients had clinical improvement or stabilization, whereas 3 patients did not. Among the patients with clinical improvement, treatment led to reduced JC viral load in cerebrospinal fluid (CSF) and increased CD4+ and CD8+ anti–JC virus activity in vitro. Among patients without clinical improvement, treatment did not meaningfully change viral load or antiviral cellular immune response.
In a separate letter, researchers in Germany described an additional patient with PML who had clinical stabilization and no disease progression on MRI after treatment with pembrolizumab.
In another letter, researchers in France described a patient with PML whose condition improved after treatment with nivolumab.
“Do pembrolizumab and nivolumab fit the bill for treatment of PML? The current reports are encouraging but suggest that the presence of JC virus–specific T cells in the blood is a prerequisite for their use,” said Igor J. Koralnik, MD, of the department of neurological sciences at Rush University Medical Center in Chicago, in an accompanying editorial. “A controlled trial may be needed to determine whether immune checkpoint inhibitors are indeed able to keep JC virus in check in patients with PML.”
Reinvigorating T cells
Both monoclonal antibodies target programmed cell death protein 1 (PD-1), which inhibits T-cell proliferation and cytokine production when it binds its associated ligand, Dr. Koralnik said. Pembrolizumab and nivolumab block this inhibition and have been used to spur T-cell activity against tumors in patients with cancer.
PML, an often fatal brain infection caused by the JC virus in patients with immunosuppression, has no specific treatment. Management hinges on “recovery of the immune system, either by treating the underlying cause of immunosuppression or by discontinuing the use of immunosuppressive medications,” said Dr. Koralnik.
Pembrolizumab
Prior studies have found that PD-1 expression is elevated on T lymphocytes of patients with PML. To determine whether PD-1 blockade with pembrolizumab reinvigorates anti–JC virus immune activity in patients with PML, Irene Cortese, MD, of the National Institutes of Health’s Neuroimmunology Clinic and her research colleagues administered pembrolizumab at a dose of 2 mg/kg of body weight every 4-6 weeks to eight adults with PML. The patients received 1-3 doses, and each patient had a different underlying condition.
In all patients, treatment induced down-regulation of PD-1 expression on lymphocytes in CSF and peripheral blood, and five of the eight patients had clinical stabilization or improvement. Of the other three patients who did not improve, one had stabilized prior to treatment and remained stable. The other two patients died from PML.
Additional reports
Separately, Sebastian Rauer, MD, of Albert Ludwigs University in Freiburg, Germany, and his colleagues reported that a patient with PML whose symptoms culminated in mutism in February 2018 began speaking again after receiving five infusions of pembrolizumab over 10 weeks. “In addition, the size and number of lesions on MRI decreased, and JCV was no longer detectable in CSF,” Dr. Rauer and his colleagues wrote. “The patient has remained stable as of the end of March 2019, with persistent but abating psychomotor slowing, aphasia, and disorientation.”
Finally, Ondine Walter, of Toulouse (France) University Hospital and colleagues described the case of a 60-year-old woman with PML who received nivolumab on a compassionate-use basis. Two weeks after treatment, JC viral load in CSF and blood had decreased. “Starting 8 weeks after the initiation of nivolumab therapy, the patient’s neurologic symptoms and signs stabilized, and subsequently she showed improved alertness, and the ptosis and hemiplegia abated.”
Reason for caution
Prior studies, however, give reasons for caution when considering the potential use of immune checkpoint inhibitors to treat PML, Dr. Koralnik noted. In one case, a patient developed an inflammatory form of PML known as immune reconstitution inflammatory syndrome after receiving nivolumab (J Neurovirol. 2019 March 12. doi: 10.1007/s13365-019-00738-x). In addition, researchers have reported a case of PML that occurred after 1 year of nivolumab treatment, and four cases of PML related to nivolumab have been reported in pharmacovigilance databases (Emerg Infect Dis. 2018;24:1594-6). The cost and safety profiles of the medications also may be considerations, Dr. Koralnik said.
The study by Dr. Cortese and colleagues was funded by the National Institutes of Health, and the authors had no relevant disclosures. Some of the research letter authors disclosed grants and personal fees from pharmaceutical companies.
SOURCES: Cortese I et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMoa1815039; Rauer S et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMc1817193; Walter O et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMc1816198; Koralnik IJ. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMe1904140.
investigators reported in the New England Journal of Medicine.
Three research teams described 10 cases in which patients with PML received pembrolizumab or nivolumab.
In one study, researchers administered pembrolizumab to eight adults with PML. Five patients had clinical improvement or stabilization, whereas 3 patients did not. Among the patients with clinical improvement, treatment led to reduced JC viral load in cerebrospinal fluid (CSF) and increased CD4+ and CD8+ anti–JC virus activity in vitro. Among patients without clinical improvement, treatment did not meaningfully change viral load or antiviral cellular immune response.
In a separate letter, researchers in Germany described an additional patient with PML who had clinical stabilization and no disease progression on MRI after treatment with pembrolizumab.
In another letter, researchers in France described a patient with PML whose condition improved after treatment with nivolumab.
“Do pembrolizumab and nivolumab fit the bill for treatment of PML? The current reports are encouraging but suggest that the presence of JC virus–specific T cells in the blood is a prerequisite for their use,” said Igor J. Koralnik, MD, of the department of neurological sciences at Rush University Medical Center in Chicago, in an accompanying editorial. “A controlled trial may be needed to determine whether immune checkpoint inhibitors are indeed able to keep JC virus in check in patients with PML.”
Reinvigorating T cells
Both monoclonal antibodies target programmed cell death protein 1 (PD-1), which inhibits T-cell proliferation and cytokine production when it binds its associated ligand, Dr. Koralnik said. Pembrolizumab and nivolumab block this inhibition and have been used to spur T-cell activity against tumors in patients with cancer.
PML, an often fatal brain infection caused by the JC virus in patients with immunosuppression, has no specific treatment. Management hinges on “recovery of the immune system, either by treating the underlying cause of immunosuppression or by discontinuing the use of immunosuppressive medications,” said Dr. Koralnik.
Pembrolizumab
Prior studies have found that PD-1 expression is elevated on T lymphocytes of patients with PML. To determine whether PD-1 blockade with pembrolizumab reinvigorates anti–JC virus immune activity in patients with PML, Irene Cortese, MD, of the National Institutes of Health’s Neuroimmunology Clinic and her research colleagues administered pembrolizumab at a dose of 2 mg/kg of body weight every 4-6 weeks to eight adults with PML. The patients received 1-3 doses, and each patient had a different underlying condition.
In all patients, treatment induced down-regulation of PD-1 expression on lymphocytes in CSF and peripheral blood, and five of the eight patients had clinical stabilization or improvement. Of the other three patients who did not improve, one had stabilized prior to treatment and remained stable. The other two patients died from PML.
Additional reports
Separately, Sebastian Rauer, MD, of Albert Ludwigs University in Freiburg, Germany, and his colleagues reported that a patient with PML whose symptoms culminated in mutism in February 2018 began speaking again after receiving five infusions of pembrolizumab over 10 weeks. “In addition, the size and number of lesions on MRI decreased, and JCV was no longer detectable in CSF,” Dr. Rauer and his colleagues wrote. “The patient has remained stable as of the end of March 2019, with persistent but abating psychomotor slowing, aphasia, and disorientation.”
Finally, Ondine Walter, of Toulouse (France) University Hospital and colleagues described the case of a 60-year-old woman with PML who received nivolumab on a compassionate-use basis. Two weeks after treatment, JC viral load in CSF and blood had decreased. “Starting 8 weeks after the initiation of nivolumab therapy, the patient’s neurologic symptoms and signs stabilized, and subsequently she showed improved alertness, and the ptosis and hemiplegia abated.”
Reason for caution
Prior studies, however, give reasons for caution when considering the potential use of immune checkpoint inhibitors to treat PML, Dr. Koralnik noted. In one case, a patient developed an inflammatory form of PML known as immune reconstitution inflammatory syndrome after receiving nivolumab (J Neurovirol. 2019 March 12. doi: 10.1007/s13365-019-00738-x). In addition, researchers have reported a case of PML that occurred after 1 year of nivolumab treatment, and four cases of PML related to nivolumab have been reported in pharmacovigilance databases (Emerg Infect Dis. 2018;24:1594-6). The cost and safety profiles of the medications also may be considerations, Dr. Koralnik said.
The study by Dr. Cortese and colleagues was funded by the National Institutes of Health, and the authors had no relevant disclosures. Some of the research letter authors disclosed grants and personal fees from pharmaceutical companies.
SOURCES: Cortese I et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMoa1815039; Rauer S et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMc1817193; Walter O et al. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMc1816198; Koralnik IJ. N Engl J Med. 2019 Apr 10. doi: 10.1056/NEJMe1904140.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE







