User login
FDA approves venetoclax/obinutuzumab combo for CLL
The Food and Drug Administration has approved the combination of venetoclax (Venclexta) plus obinutuzumab (Gazyva) for patients with previously untreated chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma.
The approval provides a chemotherapy-free, fixed duration treatment. The FDA based the approval on the results of the phase 3 CLL14 trial, which will be presented at the 2019 annual meeting of the American Society of Clinical Oncology.
Researchers randomized 432 patients to either a 12-month duration of venetoclax with a 6-month duration of obinutuzumab or to a 6-month duration of obinutuzumab plus chlorambucil and another 6-month duration of chlorambucil.
The newly approved combination reduced the risk of disease progression or death (progression-free survival as assessed by an independent review committee) by 67%, compared with obinutuzumab/chlorambucil (hazard ratio, 0.33; P less than .0001).
Venetoclax/obinutuzumab also had a higher rate of minimal residual disease negativity in bone marrow and peripheral blood, compared to the other combination, according to Genentech.
The most common adverse reactions of any grade reported for venetoclax/obinutuzumab were neutropenia, diarrhea, fatigue, nausea, anemia, and upper respiratory tract infection.
The Food and Drug Administration has approved the combination of venetoclax (Venclexta) plus obinutuzumab (Gazyva) for patients with previously untreated chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma.
The approval provides a chemotherapy-free, fixed duration treatment. The FDA based the approval on the results of the phase 3 CLL14 trial, which will be presented at the 2019 annual meeting of the American Society of Clinical Oncology.
Researchers randomized 432 patients to either a 12-month duration of venetoclax with a 6-month duration of obinutuzumab or to a 6-month duration of obinutuzumab plus chlorambucil and another 6-month duration of chlorambucil.
The newly approved combination reduced the risk of disease progression or death (progression-free survival as assessed by an independent review committee) by 67%, compared with obinutuzumab/chlorambucil (hazard ratio, 0.33; P less than .0001).
Venetoclax/obinutuzumab also had a higher rate of minimal residual disease negativity in bone marrow and peripheral blood, compared to the other combination, according to Genentech.
The most common adverse reactions of any grade reported for venetoclax/obinutuzumab were neutropenia, diarrhea, fatigue, nausea, anemia, and upper respiratory tract infection.
The Food and Drug Administration has approved the combination of venetoclax (Venclexta) plus obinutuzumab (Gazyva) for patients with previously untreated chronic lymphocytic leukemia (CLL) or small lymphocytic lymphoma.
The approval provides a chemotherapy-free, fixed duration treatment. The FDA based the approval on the results of the phase 3 CLL14 trial, which will be presented at the 2019 annual meeting of the American Society of Clinical Oncology.
Researchers randomized 432 patients to either a 12-month duration of venetoclax with a 6-month duration of obinutuzumab or to a 6-month duration of obinutuzumab plus chlorambucil and another 6-month duration of chlorambucil.
The newly approved combination reduced the risk of disease progression or death (progression-free survival as assessed by an independent review committee) by 67%, compared with obinutuzumab/chlorambucil (hazard ratio, 0.33; P less than .0001).
Venetoclax/obinutuzumab also had a higher rate of minimal residual disease negativity in bone marrow and peripheral blood, compared to the other combination, according to Genentech.
The most common adverse reactions of any grade reported for venetoclax/obinutuzumab were neutropenia, diarrhea, fatigue, nausea, anemia, and upper respiratory tract infection.
Atypical case of cutaneous MCL mimics SPTCL
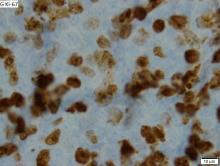
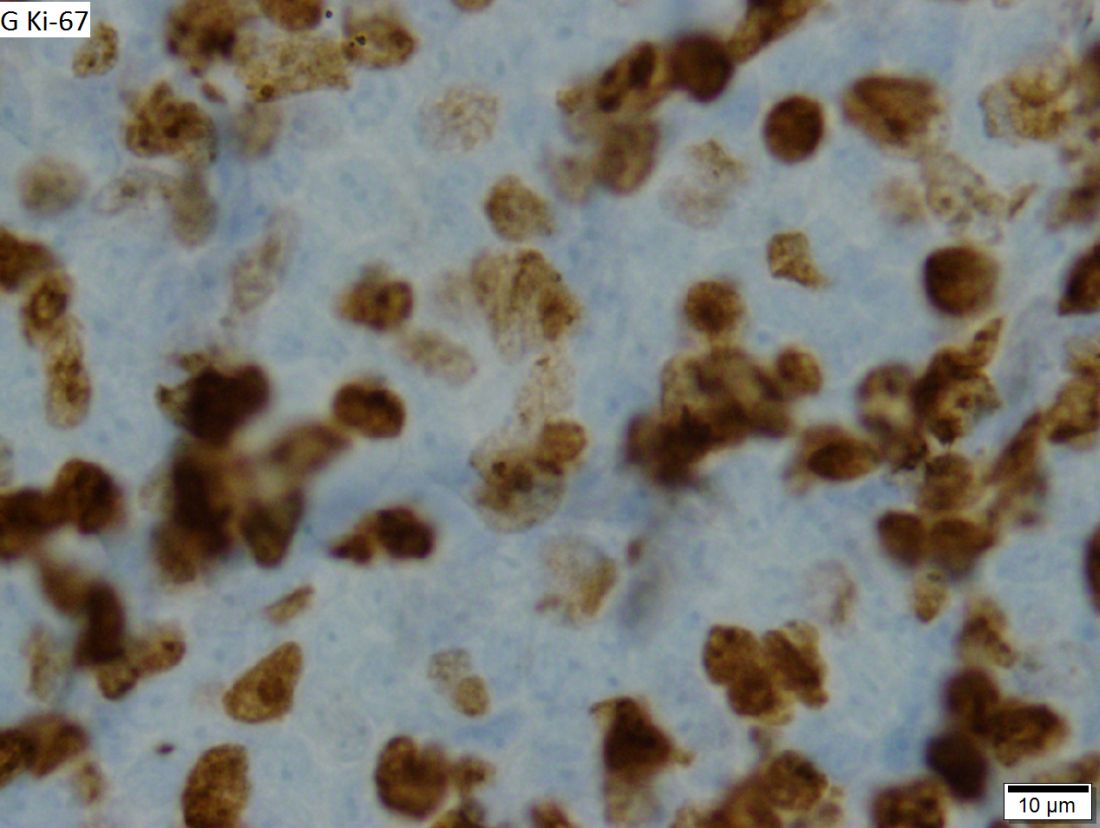
An atypical case of cutaneous mantle cell lymphoma (MCL) with histomorphological features mimicking subcutaneous panniculitis-like T-cell lymphoma (SPTCL) highlights a “potential pitfall,” according to investigators.
This unusual case stresses the importance of molecular cytogenetics and/or immunohistochemistry for panniculitis-type lymphomas, reported lead author Caroline Laggis, MD of the University of Utah, Salt Lake City, and colleagues.
“While morphologic features of SPTCL, specifically rimming of adipocytes by neoplastic lymphoid cells, have been documented in other types of lymphomas, this case is exceptional in that the morphologic features of SPTCL are showed in secondary cutaneous involvement by MCL,” the investigators wrote. Their report is in Journal of Cutaneous Pathology.
The patient was a 69-year-old man who presented with 2-year history of night sweats and fever of unknown origin, and, closer to presentation, weight loss and tender bumps under the skin of his pelvic region.
Subsequent computed tomography and excisional lymph node biopsy led to a diagnosis of MCL, with a Mantle Cell Lymphoma International Prognostic Index of 5, suggesting aggressive, intermediate-risk disease. Further imaging showed involvement of the nasopharynx, and cervical and mediastinal lymph nodes.
Bendamustine and rituximab chemotherapy was given unremarkably until the final cycle, at which point the patient presented with tender subcutaneous nodules on his lower legs. Histopathology from punch biopsies revealed “a dense infiltrate of monomorphic, mitotically active lymphoid cells with infiltration between the deep dermal collagen and the adipocytes in subcutaneous fat,” the investigators wrote, noting that the infiltrative cells were blastoid and 70% expressed cyclin D1, supporting cutaneous involvement of his systemic MCL.
Treatment was switched to ibrutinib and selinexor via a clinical trial, which led to temporary improvement of leg lesions; when the lesions returned, biopsy was performed with the same histopathological result. Lenalidomide and rituximab were started, but without success, and disease spread to the central nervous system.
Another biopsy of the skin lesions again supported cutaneous MCL, with tumor cells rimming individual adipocytes.
Because of this atypical morphology, fluorescence in situ hybridization (FISH) was conducted, revealing t(11;14)(q13:32) positivity, thereby “confirming the diagnosis of cutaneous involvement by systemic MCL,” the investigators wrote.
Genomic sequencing revealed abnormalities of “ataxia-telangiectasia mutated, mechanistic target of rapamycin kinase (mTOR), BCL6 corepressor, and FAS-associated factor 1, as well as the expected mutation in IGH-CCND1, leading to cyclin D1 upregulation.”
Subsequent treatment was unsuccessful, and the patient died from his disease.
“The complex and central role that mTOR plays in adipose homeostasis may link our tumor to its preference to the adipose tissue, although further investigation is warranted regarding specific genomic alterations in lymphomas and the implications these mutations have in the involvement of tumor cells with cutaneous and adipose environments,” the investigators wrote.
The investigators did not report conflicts of interest.
SOURCE: Laggis C et al. 2019 Apr 8. doi:10.1111/cup.13471.
An atypical case of cutaneous mantle cell lymphoma (MCL) with histomorphological features mimicking subcutaneous panniculitis-like T-cell lymphoma (SPTCL) highlights a “potential pitfall,” according to investigators.
This unusual case stresses the importance of molecular cytogenetics and/or immunohistochemistry for panniculitis-type lymphomas, reported lead author Caroline Laggis, MD of the University of Utah, Salt Lake City, and colleagues.
“While morphologic features of SPTCL, specifically rimming of adipocytes by neoplastic lymphoid cells, have been documented in other types of lymphomas, this case is exceptional in that the morphologic features of SPTCL are showed in secondary cutaneous involvement by MCL,” the investigators wrote. Their report is in Journal of Cutaneous Pathology.
The patient was a 69-year-old man who presented with 2-year history of night sweats and fever of unknown origin, and, closer to presentation, weight loss and tender bumps under the skin of his pelvic region.
Subsequent computed tomography and excisional lymph node biopsy led to a diagnosis of MCL, with a Mantle Cell Lymphoma International Prognostic Index of 5, suggesting aggressive, intermediate-risk disease. Further imaging showed involvement of the nasopharynx, and cervical and mediastinal lymph nodes.
Bendamustine and rituximab chemotherapy was given unremarkably until the final cycle, at which point the patient presented with tender subcutaneous nodules on his lower legs. Histopathology from punch biopsies revealed “a dense infiltrate of monomorphic, mitotically active lymphoid cells with infiltration between the deep dermal collagen and the adipocytes in subcutaneous fat,” the investigators wrote, noting that the infiltrative cells were blastoid and 70% expressed cyclin D1, supporting cutaneous involvement of his systemic MCL.
Treatment was switched to ibrutinib and selinexor via a clinical trial, which led to temporary improvement of leg lesions; when the lesions returned, biopsy was performed with the same histopathological result. Lenalidomide and rituximab were started, but without success, and disease spread to the central nervous system.
Another biopsy of the skin lesions again supported cutaneous MCL, with tumor cells rimming individual adipocytes.
Because of this atypical morphology, fluorescence in situ hybridization (FISH) was conducted, revealing t(11;14)(q13:32) positivity, thereby “confirming the diagnosis of cutaneous involvement by systemic MCL,” the investigators wrote.
Genomic sequencing revealed abnormalities of “ataxia-telangiectasia mutated, mechanistic target of rapamycin kinase (mTOR), BCL6 corepressor, and FAS-associated factor 1, as well as the expected mutation in IGH-CCND1, leading to cyclin D1 upregulation.”
Subsequent treatment was unsuccessful, and the patient died from his disease.
“The complex and central role that mTOR plays in adipose homeostasis may link our tumor to its preference to the adipose tissue, although further investigation is warranted regarding specific genomic alterations in lymphomas and the implications these mutations have in the involvement of tumor cells with cutaneous and adipose environments,” the investigators wrote.
The investigators did not report conflicts of interest.
SOURCE: Laggis C et al. 2019 Apr 8. doi:10.1111/cup.13471.
An atypical case of cutaneous mantle cell lymphoma (MCL) with histomorphological features mimicking subcutaneous panniculitis-like T-cell lymphoma (SPTCL) highlights a “potential pitfall,” according to investigators.
This unusual case stresses the importance of molecular cytogenetics and/or immunohistochemistry for panniculitis-type lymphomas, reported lead author Caroline Laggis, MD of the University of Utah, Salt Lake City, and colleagues.
“While morphologic features of SPTCL, specifically rimming of adipocytes by neoplastic lymphoid cells, have been documented in other types of lymphomas, this case is exceptional in that the morphologic features of SPTCL are showed in secondary cutaneous involvement by MCL,” the investigators wrote. Their report is in Journal of Cutaneous Pathology.
The patient was a 69-year-old man who presented with 2-year history of night sweats and fever of unknown origin, and, closer to presentation, weight loss and tender bumps under the skin of his pelvic region.
Subsequent computed tomography and excisional lymph node biopsy led to a diagnosis of MCL, with a Mantle Cell Lymphoma International Prognostic Index of 5, suggesting aggressive, intermediate-risk disease. Further imaging showed involvement of the nasopharynx, and cervical and mediastinal lymph nodes.
Bendamustine and rituximab chemotherapy was given unremarkably until the final cycle, at which point the patient presented with tender subcutaneous nodules on his lower legs. Histopathology from punch biopsies revealed “a dense infiltrate of monomorphic, mitotically active lymphoid cells with infiltration between the deep dermal collagen and the adipocytes in subcutaneous fat,” the investigators wrote, noting that the infiltrative cells were blastoid and 70% expressed cyclin D1, supporting cutaneous involvement of his systemic MCL.
Treatment was switched to ibrutinib and selinexor via a clinical trial, which led to temporary improvement of leg lesions; when the lesions returned, biopsy was performed with the same histopathological result. Lenalidomide and rituximab were started, but without success, and disease spread to the central nervous system.
Another biopsy of the skin lesions again supported cutaneous MCL, with tumor cells rimming individual adipocytes.
Because of this atypical morphology, fluorescence in situ hybridization (FISH) was conducted, revealing t(11;14)(q13:32) positivity, thereby “confirming the diagnosis of cutaneous involvement by systemic MCL,” the investigators wrote.
Genomic sequencing revealed abnormalities of “ataxia-telangiectasia mutated, mechanistic target of rapamycin kinase (mTOR), BCL6 corepressor, and FAS-associated factor 1, as well as the expected mutation in IGH-CCND1, leading to cyclin D1 upregulation.”
Subsequent treatment was unsuccessful, and the patient died from his disease.
“The complex and central role that mTOR plays in adipose homeostasis may link our tumor to its preference to the adipose tissue, although further investigation is warranted regarding specific genomic alterations in lymphomas and the implications these mutations have in the involvement of tumor cells with cutaneous and adipose environments,” the investigators wrote.
The investigators did not report conflicts of interest.
SOURCE: Laggis C et al. 2019 Apr 8. doi:10.1111/cup.13471.
FROM JOURNAL OF CUTANEOUS PATHOLOGY
Inhibitor may overcome ibrutinib resistance in MCL
Investigators have identified a mechanism of ibrutinib resistance in mantle cell lymphoma (MCL) and showed that a small molecule can overcome that resistance in vitro and in vivo.
The team found that ibrutinib-resistant MCL cells rely on oxidative phosphorylation (OXPHOS) and glutaminolysis to survive.
Targeting the OXPHOS pathway with a small molecule, IACS-010759, inhibited the proliferation of ibrutinib-resistant cells in vitro.
IACS-010759 also decreased tumor volume and improved survival in mouse models of ibrutinib-resistant MCL and double-hit B-cell lymphoma.
Now, IACS-10759 is being tested in phase 1 trials of lymphoma and solid tumors (NCT03291938) as well as acute myeloid leukemia (NCT02882321).
Liang Zhang, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, and his colleagues conducted the preclinical research and described their findings in Science Translational Medicine.
The investigators sequenced samples from MCL patients with ibrutinib-sensitive and -resistant disease and found that “glutamine-fueled OXPHOS appears to be a prominent energy metabolism pathway in ibrutinib-resistant MCL cells.”
This finding prompted the team to test IACS-010759, an inhibitor of ETC complex I, in ibrutinib-resistant MCL. They theorized that the inhibitor would be effective because, during OXPHOS, electrons are transferred from electron donors to acceptors through the ETC in redox reactions that release energy to form ATP, and OXPHOS generates ATP to meet requirements for cell growth.
In experiments, IACS-010759 inhibited the proliferation of two ibrutinib-resistant MCL cell lines, Z-138 and Maver-1, in a dose-dependent manner.
The investigators also tested IACS-010759 in two mouse models of ibrutinib-resistant MCL. In both models, mice treated with IACS-010759 had a significant reduction in tumor volume, compared with controls. In one model, IACS-010759 extended survival by a median of 11 days.
Finally, the team tested IACS-010759 in a model of ibrutinib-resistant, double-hit (MYC and BCL-2) B-cell lymphoma with central nervous system involvement. Again, IACS-010759 significantly inhibited tumor growth. Compared to ibrutinib and vehicle control, IACS-010759 provided a median survival benefit of more than 20 days.
There were no toxicities associated with IACS-010759 treatment, according to the investigators.
This research was supported by the MD Anderson B Cell Lymphoma Moon Shot Project, Gary Rogers Foundation, Kinder Foundation, Cullen Foundation, Cancer Prevention Research Institute of Texas, and the National Institutes of Health. Most investigators reported having no competing interests, but two reported a patent (WO/2015/130790).
SOURCE: Zhang L et al. Sci Transl Med. 2019 May 8. doi: 10.1126/scitranslmed.aau1167.
Investigators have identified a mechanism of ibrutinib resistance in mantle cell lymphoma (MCL) and showed that a small molecule can overcome that resistance in vitro and in vivo.
The team found that ibrutinib-resistant MCL cells rely on oxidative phosphorylation (OXPHOS) and glutaminolysis to survive.
Targeting the OXPHOS pathway with a small molecule, IACS-010759, inhibited the proliferation of ibrutinib-resistant cells in vitro.
IACS-010759 also decreased tumor volume and improved survival in mouse models of ibrutinib-resistant MCL and double-hit B-cell lymphoma.
Now, IACS-10759 is being tested in phase 1 trials of lymphoma and solid tumors (NCT03291938) as well as acute myeloid leukemia (NCT02882321).
Liang Zhang, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, and his colleagues conducted the preclinical research and described their findings in Science Translational Medicine.
The investigators sequenced samples from MCL patients with ibrutinib-sensitive and -resistant disease and found that “glutamine-fueled OXPHOS appears to be a prominent energy metabolism pathway in ibrutinib-resistant MCL cells.”
This finding prompted the team to test IACS-010759, an inhibitor of ETC complex I, in ibrutinib-resistant MCL. They theorized that the inhibitor would be effective because, during OXPHOS, electrons are transferred from electron donors to acceptors through the ETC in redox reactions that release energy to form ATP, and OXPHOS generates ATP to meet requirements for cell growth.
In experiments, IACS-010759 inhibited the proliferation of two ibrutinib-resistant MCL cell lines, Z-138 and Maver-1, in a dose-dependent manner.
The investigators also tested IACS-010759 in two mouse models of ibrutinib-resistant MCL. In both models, mice treated with IACS-010759 had a significant reduction in tumor volume, compared with controls. In one model, IACS-010759 extended survival by a median of 11 days.
Finally, the team tested IACS-010759 in a model of ibrutinib-resistant, double-hit (MYC and BCL-2) B-cell lymphoma with central nervous system involvement. Again, IACS-010759 significantly inhibited tumor growth. Compared to ibrutinib and vehicle control, IACS-010759 provided a median survival benefit of more than 20 days.
There were no toxicities associated with IACS-010759 treatment, according to the investigators.
This research was supported by the MD Anderson B Cell Lymphoma Moon Shot Project, Gary Rogers Foundation, Kinder Foundation, Cullen Foundation, Cancer Prevention Research Institute of Texas, and the National Institutes of Health. Most investigators reported having no competing interests, but two reported a patent (WO/2015/130790).
SOURCE: Zhang L et al. Sci Transl Med. 2019 May 8. doi: 10.1126/scitranslmed.aau1167.
Investigators have identified a mechanism of ibrutinib resistance in mantle cell lymphoma (MCL) and showed that a small molecule can overcome that resistance in vitro and in vivo.
The team found that ibrutinib-resistant MCL cells rely on oxidative phosphorylation (OXPHOS) and glutaminolysis to survive.
Targeting the OXPHOS pathway with a small molecule, IACS-010759, inhibited the proliferation of ibrutinib-resistant cells in vitro.
IACS-010759 also decreased tumor volume and improved survival in mouse models of ibrutinib-resistant MCL and double-hit B-cell lymphoma.
Now, IACS-10759 is being tested in phase 1 trials of lymphoma and solid tumors (NCT03291938) as well as acute myeloid leukemia (NCT02882321).
Liang Zhang, MD, PhD, of the University of Texas MD Anderson Cancer Center in Houston, and his colleagues conducted the preclinical research and described their findings in Science Translational Medicine.
The investigators sequenced samples from MCL patients with ibrutinib-sensitive and -resistant disease and found that “glutamine-fueled OXPHOS appears to be a prominent energy metabolism pathway in ibrutinib-resistant MCL cells.”
This finding prompted the team to test IACS-010759, an inhibitor of ETC complex I, in ibrutinib-resistant MCL. They theorized that the inhibitor would be effective because, during OXPHOS, electrons are transferred from electron donors to acceptors through the ETC in redox reactions that release energy to form ATP, and OXPHOS generates ATP to meet requirements for cell growth.
In experiments, IACS-010759 inhibited the proliferation of two ibrutinib-resistant MCL cell lines, Z-138 and Maver-1, in a dose-dependent manner.
The investigators also tested IACS-010759 in two mouse models of ibrutinib-resistant MCL. In both models, mice treated with IACS-010759 had a significant reduction in tumor volume, compared with controls. In one model, IACS-010759 extended survival by a median of 11 days.
Finally, the team tested IACS-010759 in a model of ibrutinib-resistant, double-hit (MYC and BCL-2) B-cell lymphoma with central nervous system involvement. Again, IACS-010759 significantly inhibited tumor growth. Compared to ibrutinib and vehicle control, IACS-010759 provided a median survival benefit of more than 20 days.
There were no toxicities associated with IACS-010759 treatment, according to the investigators.
This research was supported by the MD Anderson B Cell Lymphoma Moon Shot Project, Gary Rogers Foundation, Kinder Foundation, Cullen Foundation, Cancer Prevention Research Institute of Texas, and the National Institutes of Health. Most investigators reported having no competing interests, but two reported a patent (WO/2015/130790).
SOURCE: Zhang L et al. Sci Transl Med. 2019 May 8. doi: 10.1126/scitranslmed.aau1167.
FROM SCIENCE TRANSLATIONAL MEDICINE
BTK inhibitor reduces MS enhancing lesions
. However, there was no difference between the 25-mg once daily, 75-mg once daily, 75-mg twice daily, and placebo-treated groups in Expanded Disability Status Scale scores, according to a double-blind, randomized, phase 2 trial published in the New England Journal of Medicine (2019 May 10. doi: 10.1056/NEJMoa1901981).
We first reported on the results of this trial when they were presented at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis. Find our coverage at the link below.
. However, there was no difference between the 25-mg once daily, 75-mg once daily, 75-mg twice daily, and placebo-treated groups in Expanded Disability Status Scale scores, according to a double-blind, randomized, phase 2 trial published in the New England Journal of Medicine (2019 May 10. doi: 10.1056/NEJMoa1901981).
We first reported on the results of this trial when they were presented at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis. Find our coverage at the link below.
. However, there was no difference between the 25-mg once daily, 75-mg once daily, 75-mg twice daily, and placebo-treated groups in Expanded Disability Status Scale scores, according to a double-blind, randomized, phase 2 trial published in the New England Journal of Medicine (2019 May 10. doi: 10.1056/NEJMoa1901981).
We first reported on the results of this trial when they were presented at the annual congress of the European Committee for Treatment and Research in Multiple Sclerosis. Find our coverage at the link below.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Interview with Stephen Krieger, MD, on the topographical model of multiple sclerosis
We interviewed Dr. Stephen Krieger to discuss his research in the implementation of the topographical model of Multiple Sclerosis.
What is the concept behind the topographical model of multiple sclerosis (MS)?
DR. KRIEGER: MS is an incredibly heterogeneous, and in many ways, unpredictable disease. Some people with MS will have a relapsing course, others will take a progressive course of disease, and many will have a disease course that spans both a relapsing phase and a progressive phase.
We have traditionally divided MS into phenotypes like relapsing-remitting MS (RRMS), secondary-progressive MS (SPMS), or primary-progressive MS (PPMS), and these phenotypes have been foundational in our field and used to define clinical trial cohorts and outcomes. They also have been used for the approval of our medicines. In practice, however, it sometimes can be difficult to know precisely what kind of MS, what phenotypes of MS, that an individual patient has.
The topographical model, which was proposed four years ago, tries to unify our concepts of MS in a way that spans across those phenotypes and animates the disease course in a more dynamic way to bridge from one phenotype to another.
As an individual patient, for example, develops clinically isolated syndrome (CIS) or first attack, and then RRMS, and then later SPMS, that gets depicted in a dynamic visualization through the topographical model. The model also makes use of the idea that where an MS lesion is in the central nervous system (CNS) defines the clinical symptoms that it causes.
This is something that we have long known, and the art of localization in neurology has existed for at least a couple hundred years. But we have not used that in the way we have depicted MS clinical course in recent decades. The topographical model tries to bring this idea of mapping an individual patient’s disease topography back into the clinical picture.
In the topographical model, the lesions are shown as different topographical peaks, via the hills and valleys of areas of MS damage across different regions of the CNS [image]. They are compensated for by reserve, by the ability that the nervous system has to compensate and to keep a disease process from crossing the clinical threshold and causing symptoms. What the topographical model displays is that patients with MS lose reserve as time passes.
We know that there is brain atrophy, brain stem atrophy, spinal cord atrophy, and retinal nerve fiber layer thinning in this disease. The topographical model takes the concept that MS causes a loss of tissue across the CNS and applies it to where the lesions are in the CNS. The coming together of those two things brings about the clinical picture unmasking the deficit from those lesions over time. The short version is a depiction of disease course in MS. The way it looks has been likened to a leaking swimming pool, where there is a shallow end and a deep end, and as reserve drains over time more and more of that subclinical disease becomes unmasked.
In the Laitman article (2018), you applied the model to real patients. What were the main findings from that study?
DR. KRIEGER: Until now, the topographical model has been conceptual with a visual depiction, and I think it has been important as an educational tool and an aid to help shape our thinking about MS in a unified way.
The Laitman et al research, is the first time we have applied the concepts of the model to individual patients to confirm whether we could map individual patients’ MS histories in the topographical model and see if we could depict their clinical course this way. We found that we could.
One of the most important points that the topographical model makes is the idea that as progression occurs and reserve is lost, there is an unmasking of underlying disease. Meaning, all of the signs and symptoms that a patient has had during their relapse when they were accumulating lesions should be re-revealed or recapitulated when reserve is lost and progression occurs.
To confirm this, we mapped ten patients in the topographical model. We characterized their signs and symptoms of relapses during the relapsing phase and we found that the vast majority of these symptoms had redeclared themselves at the time that these patients developed SPMS. Furthermore, those symptoms were continuing to worsen in their pattern; that is in the pattern of their disease topography as the years have continued to pass since they developed SPMS.
This was the first empirical study in real patients to show that the principles of the topographical model held true. This recapitulation hypothesis of symptoms in progressive disease was borne out, and that can help to lay the groundwork for future empirical studies to see how this model can be used as a predictive tool.
How does this new theory of MS disease progression better inform treatment decisions than the disease course theories that currently exist?
DR. KRIEGER: We have had the clinical phenotypes for 20 years and it has been very helpful to us in the development of treatments that we have shown are effective for RRMS and in more recent years for PPMS. What we don’t really have is a way of personalizing and predicting the individual person’s disease trajectory.
Although we have prognostic factors that we know are important, such as age and MRI disease burden, there is still great uncertainty of the clinical course in the individual patient. If the topographical model can be further empirically validated using real world data, that could help us to predict what is going to happen to an individual patient. That can help us to make better treatment decisions for them because it could inform our treatment decisions in a more personalized way.
Is there any other recent research that supports these concepts?
DR. KRIEGER: We talk a lot about the need for biomarkers in MS to help us predict disease course and the topographical model makes the case that lesion location is a crucial biomarker. That is, the patient that has lesions in the spinal cord and the brain stem is more likely to have progressive signs and symptoms referable to those lesions.
A separate piece of work recently done by Keegan and colleagues that was published in Multiple Sclerosis Journal, looked at their own cohort of patients that had at least one critically located lesion, typically in the high cervical spinal cord or the lower brain stem, as being the crucial driver of the development of motor dysfunction and progressive disability.
In an editorial I wrote with my colleague, Fred Lublin, called “Location, location, location,” we point out that this is in some ways the best data in support of the concept of the topographical model that I have seen. It outlines a framework or a methodology where the importance of lesion location in defining the clinical picture and the risk of progression for an individual patient can be studied.
We interviewed Dr. Stephen Krieger to discuss his research in the implementation of the topographical model of Multiple Sclerosis.
What is the concept behind the topographical model of multiple sclerosis (MS)?
DR. KRIEGER: MS is an incredibly heterogeneous, and in many ways, unpredictable disease. Some people with MS will have a relapsing course, others will take a progressive course of disease, and many will have a disease course that spans both a relapsing phase and a progressive phase.
We have traditionally divided MS into phenotypes like relapsing-remitting MS (RRMS), secondary-progressive MS (SPMS), or primary-progressive MS (PPMS), and these phenotypes have been foundational in our field and used to define clinical trial cohorts and outcomes. They also have been used for the approval of our medicines. In practice, however, it sometimes can be difficult to know precisely what kind of MS, what phenotypes of MS, that an individual patient has.
The topographical model, which was proposed four years ago, tries to unify our concepts of MS in a way that spans across those phenotypes and animates the disease course in a more dynamic way to bridge from one phenotype to another.
As an individual patient, for example, develops clinically isolated syndrome (CIS) or first attack, and then RRMS, and then later SPMS, that gets depicted in a dynamic visualization through the topographical model. The model also makes use of the idea that where an MS lesion is in the central nervous system (CNS) defines the clinical symptoms that it causes.
This is something that we have long known, and the art of localization in neurology has existed for at least a couple hundred years. But we have not used that in the way we have depicted MS clinical course in recent decades. The topographical model tries to bring this idea of mapping an individual patient’s disease topography back into the clinical picture.
In the topographical model, the lesions are shown as different topographical peaks, via the hills and valleys of areas of MS damage across different regions of the CNS [image]. They are compensated for by reserve, by the ability that the nervous system has to compensate and to keep a disease process from crossing the clinical threshold and causing symptoms. What the topographical model displays is that patients with MS lose reserve as time passes.
We know that there is brain atrophy, brain stem atrophy, spinal cord atrophy, and retinal nerve fiber layer thinning in this disease. The topographical model takes the concept that MS causes a loss of tissue across the CNS and applies it to where the lesions are in the CNS. The coming together of those two things brings about the clinical picture unmasking the deficit from those lesions over time. The short version is a depiction of disease course in MS. The way it looks has been likened to a leaking swimming pool, where there is a shallow end and a deep end, and as reserve drains over time more and more of that subclinical disease becomes unmasked.
In the Laitman article (2018), you applied the model to real patients. What were the main findings from that study?
DR. KRIEGER: Until now, the topographical model has been conceptual with a visual depiction, and I think it has been important as an educational tool and an aid to help shape our thinking about MS in a unified way.
The Laitman et al research, is the first time we have applied the concepts of the model to individual patients to confirm whether we could map individual patients’ MS histories in the topographical model and see if we could depict their clinical course this way. We found that we could.
One of the most important points that the topographical model makes is the idea that as progression occurs and reserve is lost, there is an unmasking of underlying disease. Meaning, all of the signs and symptoms that a patient has had during their relapse when they were accumulating lesions should be re-revealed or recapitulated when reserve is lost and progression occurs.
To confirm this, we mapped ten patients in the topographical model. We characterized their signs and symptoms of relapses during the relapsing phase and we found that the vast majority of these symptoms had redeclared themselves at the time that these patients developed SPMS. Furthermore, those symptoms were continuing to worsen in their pattern; that is in the pattern of their disease topography as the years have continued to pass since they developed SPMS.
This was the first empirical study in real patients to show that the principles of the topographical model held true. This recapitulation hypothesis of symptoms in progressive disease was borne out, and that can help to lay the groundwork for future empirical studies to see how this model can be used as a predictive tool.
How does this new theory of MS disease progression better inform treatment decisions than the disease course theories that currently exist?
DR. KRIEGER: We have had the clinical phenotypes for 20 years and it has been very helpful to us in the development of treatments that we have shown are effective for RRMS and in more recent years for PPMS. What we don’t really have is a way of personalizing and predicting the individual person’s disease trajectory.
Although we have prognostic factors that we know are important, such as age and MRI disease burden, there is still great uncertainty of the clinical course in the individual patient. If the topographical model can be further empirically validated using real world data, that could help us to predict what is going to happen to an individual patient. That can help us to make better treatment decisions for them because it could inform our treatment decisions in a more personalized way.
Is there any other recent research that supports these concepts?
DR. KRIEGER: We talk a lot about the need for biomarkers in MS to help us predict disease course and the topographical model makes the case that lesion location is a crucial biomarker. That is, the patient that has lesions in the spinal cord and the brain stem is more likely to have progressive signs and symptoms referable to those lesions.
A separate piece of work recently done by Keegan and colleagues that was published in Multiple Sclerosis Journal, looked at their own cohort of patients that had at least one critically located lesion, typically in the high cervical spinal cord or the lower brain stem, as being the crucial driver of the development of motor dysfunction and progressive disability.
In an editorial I wrote with my colleague, Fred Lublin, called “Location, location, location,” we point out that this is in some ways the best data in support of the concept of the topographical model that I have seen. It outlines a framework or a methodology where the importance of lesion location in defining the clinical picture and the risk of progression for an individual patient can be studied.
We interviewed Dr. Stephen Krieger to discuss his research in the implementation of the topographical model of Multiple Sclerosis.
What is the concept behind the topographical model of multiple sclerosis (MS)?
DR. KRIEGER: MS is an incredibly heterogeneous, and in many ways, unpredictable disease. Some people with MS will have a relapsing course, others will take a progressive course of disease, and many will have a disease course that spans both a relapsing phase and a progressive phase.
We have traditionally divided MS into phenotypes like relapsing-remitting MS (RRMS), secondary-progressive MS (SPMS), or primary-progressive MS (PPMS), and these phenotypes have been foundational in our field and used to define clinical trial cohorts and outcomes. They also have been used for the approval of our medicines. In practice, however, it sometimes can be difficult to know precisely what kind of MS, what phenotypes of MS, that an individual patient has.
The topographical model, which was proposed four years ago, tries to unify our concepts of MS in a way that spans across those phenotypes and animates the disease course in a more dynamic way to bridge from one phenotype to another.
As an individual patient, for example, develops clinically isolated syndrome (CIS) or first attack, and then RRMS, and then later SPMS, that gets depicted in a dynamic visualization through the topographical model. The model also makes use of the idea that where an MS lesion is in the central nervous system (CNS) defines the clinical symptoms that it causes.
This is something that we have long known, and the art of localization in neurology has existed for at least a couple hundred years. But we have not used that in the way we have depicted MS clinical course in recent decades. The topographical model tries to bring this idea of mapping an individual patient’s disease topography back into the clinical picture.
In the topographical model, the lesions are shown as different topographical peaks, via the hills and valleys of areas of MS damage across different regions of the CNS [image]. They are compensated for by reserve, by the ability that the nervous system has to compensate and to keep a disease process from crossing the clinical threshold and causing symptoms. What the topographical model displays is that patients with MS lose reserve as time passes.
We know that there is brain atrophy, brain stem atrophy, spinal cord atrophy, and retinal nerve fiber layer thinning in this disease. The topographical model takes the concept that MS causes a loss of tissue across the CNS and applies it to where the lesions are in the CNS. The coming together of those two things brings about the clinical picture unmasking the deficit from those lesions over time. The short version is a depiction of disease course in MS. The way it looks has been likened to a leaking swimming pool, where there is a shallow end and a deep end, and as reserve drains over time more and more of that subclinical disease becomes unmasked.
In the Laitman article (2018), you applied the model to real patients. What were the main findings from that study?
DR. KRIEGER: Until now, the topographical model has been conceptual with a visual depiction, and I think it has been important as an educational tool and an aid to help shape our thinking about MS in a unified way.
The Laitman et al research, is the first time we have applied the concepts of the model to individual patients to confirm whether we could map individual patients’ MS histories in the topographical model and see if we could depict their clinical course this way. We found that we could.
One of the most important points that the topographical model makes is the idea that as progression occurs and reserve is lost, there is an unmasking of underlying disease. Meaning, all of the signs and symptoms that a patient has had during their relapse when they were accumulating lesions should be re-revealed or recapitulated when reserve is lost and progression occurs.
To confirm this, we mapped ten patients in the topographical model. We characterized their signs and symptoms of relapses during the relapsing phase and we found that the vast majority of these symptoms had redeclared themselves at the time that these patients developed SPMS. Furthermore, those symptoms were continuing to worsen in their pattern; that is in the pattern of their disease topography as the years have continued to pass since they developed SPMS.
This was the first empirical study in real patients to show that the principles of the topographical model held true. This recapitulation hypothesis of symptoms in progressive disease was borne out, and that can help to lay the groundwork for future empirical studies to see how this model can be used as a predictive tool.
How does this new theory of MS disease progression better inform treatment decisions than the disease course theories that currently exist?
DR. KRIEGER: We have had the clinical phenotypes for 20 years and it has been very helpful to us in the development of treatments that we have shown are effective for RRMS and in more recent years for PPMS. What we don’t really have is a way of personalizing and predicting the individual person’s disease trajectory.
Although we have prognostic factors that we know are important, such as age and MRI disease burden, there is still great uncertainty of the clinical course in the individual patient. If the topographical model can be further empirically validated using real world data, that could help us to predict what is going to happen to an individual patient. That can help us to make better treatment decisions for them because it could inform our treatment decisions in a more personalized way.
Is there any other recent research that supports these concepts?
DR. KRIEGER: We talk a lot about the need for biomarkers in MS to help us predict disease course and the topographical model makes the case that lesion location is a crucial biomarker. That is, the patient that has lesions in the spinal cord and the brain stem is more likely to have progressive signs and symptoms referable to those lesions.
A separate piece of work recently done by Keegan and colleagues that was published in Multiple Sclerosis Journal, looked at their own cohort of patients that had at least one critically located lesion, typically in the high cervical spinal cord or the lower brain stem, as being the crucial driver of the development of motor dysfunction and progressive disability.
In an editorial I wrote with my colleague, Fred Lublin, called “Location, location, location,” we point out that this is in some ways the best data in support of the concept of the topographical model that I have seen. It outlines a framework or a methodology where the importance of lesion location in defining the clinical picture and the risk of progression for an individual patient can be studied.
Apremilast and Phototherapy for Treatment of Psoriasis in a Patient With Human Immunodeficiency Virus
To the Editor:
A 50-year old man with Fitzpatrick skin type IV, human immunodeficiency virus (HIV), fatty liver disease, and moderate psoriasis (10% body surface area [BSA] affected) currently treated with clobetasol spray and calcitriol ointment presented with persistent psoriatic lesions on the trunk, arms, legs, and buttocks. His CD4 count was 460 and his HIV RNA count was 48 copies/mL on polymerase chain reaction 2 months prior to the current presentation. He had been undergoing phototherapy 3 times weekly for the last 5 months for treatment of psoriasis.
At the current presentation, he was started on an apremilast starter pack with the dosage titrated from 10 mg to 30 mg over the course of 1 week. He was maintained on a dose of 30 mg twice daily after 1 week and continued clobetasol spray, calcitriol ointment, and phototherapy 3 times weekly with the intent to reduce the frequency after adequate control of psoriasis was achieved. After 3 months of treatment, the affected BSA was 0%. He continued apremilast, and phototherapy was reduced to once weekly. Phototherapy was discontinued after 7 months of concomitant treatment with apremilast after clearance was maintained. It was reinitiated twice weekly after a mild flare (3% BSA affected). After 20 total months of treatment, the patient was no longer able to afford apremilast treatment and presented with a severe psoriasis flare (40% BSA affected). He was switched to acitretin with a plan to apply for apremilast financial assistance programs.
Psoriasis treatment in the HIV population poses a challenge given the immunosuppressed state of these patients, the risk of reactivation of latent infections, and the refractory nature of psoriasis in the setting of HIV. Two of the authors (S.P.R. and J.J.W.) previously reported a case of moderate to severe psoriasis in a patient with HIV and hepatitis C who demonstrated treatment success with apremilast until it was discontinued due to financial implications.1 Currently, apremilast is not widely used to treat psoriasis in the HIV population. The National Psoriasis Foundation 2010 guidelines recommended UV light therapy for treatment of moderate to severe psoriasis in HIV-positive patients, with oral retinoids as the second-line treatment.2 There remains a need for updated guidelines on the use of systemic agents for psoriasis treatment in the HIV population.
Apremilast, a phosphodiesterase 4 inhibitor, is an oral therapy that restores the balance of proinflammatory and anti-inflammatory cytokines by inhibiting inflammatory cytokine (eg, tumor necrosis factor α, IFN-γ, IL-2, IL-12, IL-23) secretion and stimulating anti-inflammatory cytokine (eg, IL-6, IL-10) production. In 2015, the phase 3 ESTEEM 13 and ESTEEM 24 trials demonstrated the efficacy of apremilast 30 mg twice daily for treatment of psoriasis. In both trials, the psoriasis area and severity index 75 response rate at week 16 was significantly
Use of other systemic agents such as tumor necrosis factor α inhibitors and ustekinumab has been reported in HIV-positive patients.5-7 There is no current data on IL-17 and IL-23 inhibitors. Acitretin generally is recommended as a second-line agent in HIV patients given its lack of immunosuppression2; however, methotrexate and cyclosporine should be avoided given the risk of opportunistic infections.8
Apremilast is a promising therapy with a favorable safety profile that should be considered as an adjuvant treatment to first-line agents such as phototherapy in HIV-positive patients. Apremilast has been successfully used in an HIV patient with a concomitant chronic hepatitis C infection.1 Systemic medications such as apremilast should be managed in coordination with infectious disease specialists with close monitoring of CD4 levels and viral loads as well as prophylactic agents.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:e481-e482.
- Menon K, Van Voorhees AS, Bebo BF Jr, et al. Psoriasis in patients with HIV infection: from the medical board of the National Psoriasis Foundation [published online July 31, 2009]. J Am Acad Dermatol. 2010;62:291-299.
- Papp K, Reich K, Leonardi CL, et al. Apremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized, controlled trial (Efficacy and Safety Trial Evaluating the Effects of Apremilast in Psoriasis [ESTEEM] 1). J Am Acad Dermatol. 2015;73:37-49.
- Paul C, Cather J, Gooderham M, et al. Efficacy and safety of apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate-to-severe plaque psoriasis over 52 weeks: a phase III, randomized controlled trial (ESTEEM 2). Br J Dermatol. 2015;173:1387-1399.
- Lindsey SF, Weiss J, Lee ES, et al. Treatment of severe psoriasis and psoriatic arthritis with adalimumab in an HIV-positive patient. J Drugs Dermatol. 2014;13:869-871.
- Saeki H, Ito T, Hayashi M, et al. Successful treatment of ustekinumab in a severe psoriasis patient with human immunodeficiency virus infection. J Eur Acad Dermatol Venereol. 2015;29:1653-1655.
- Paparizos V, Rallis E, Kirsten L, et al. Ustekinumab for the treatment of HIV psoriasis. J Dermatolog Treat. 2012;23:398-399.
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections [published online July 11, 2018]. J Am Acad Dermatol. 2019;80:43-53.
To the Editor:
A 50-year old man with Fitzpatrick skin type IV, human immunodeficiency virus (HIV), fatty liver disease, and moderate psoriasis (10% body surface area [BSA] affected) currently treated with clobetasol spray and calcitriol ointment presented with persistent psoriatic lesions on the trunk, arms, legs, and buttocks. His CD4 count was 460 and his HIV RNA count was 48 copies/mL on polymerase chain reaction 2 months prior to the current presentation. He had been undergoing phototherapy 3 times weekly for the last 5 months for treatment of psoriasis.
At the current presentation, he was started on an apremilast starter pack with the dosage titrated from 10 mg to 30 mg over the course of 1 week. He was maintained on a dose of 30 mg twice daily after 1 week and continued clobetasol spray, calcitriol ointment, and phototherapy 3 times weekly with the intent to reduce the frequency after adequate control of psoriasis was achieved. After 3 months of treatment, the affected BSA was 0%. He continued apremilast, and phototherapy was reduced to once weekly. Phototherapy was discontinued after 7 months of concomitant treatment with apremilast after clearance was maintained. It was reinitiated twice weekly after a mild flare (3% BSA affected). After 20 total months of treatment, the patient was no longer able to afford apremilast treatment and presented with a severe psoriasis flare (40% BSA affected). He was switched to acitretin with a plan to apply for apremilast financial assistance programs.
Psoriasis treatment in the HIV population poses a challenge given the immunosuppressed state of these patients, the risk of reactivation of latent infections, and the refractory nature of psoriasis in the setting of HIV. Two of the authors (S.P.R. and J.J.W.) previously reported a case of moderate to severe psoriasis in a patient with HIV and hepatitis C who demonstrated treatment success with apremilast until it was discontinued due to financial implications.1 Currently, apremilast is not widely used to treat psoriasis in the HIV population. The National Psoriasis Foundation 2010 guidelines recommended UV light therapy for treatment of moderate to severe psoriasis in HIV-positive patients, with oral retinoids as the second-line treatment.2 There remains a need for updated guidelines on the use of systemic agents for psoriasis treatment in the HIV population.
Apremilast, a phosphodiesterase 4 inhibitor, is an oral therapy that restores the balance of proinflammatory and anti-inflammatory cytokines by inhibiting inflammatory cytokine (eg, tumor necrosis factor α, IFN-γ, IL-2, IL-12, IL-23) secretion and stimulating anti-inflammatory cytokine (eg, IL-6, IL-10) production. In 2015, the phase 3 ESTEEM 13 and ESTEEM 24 trials demonstrated the efficacy of apremilast 30 mg twice daily for treatment of psoriasis. In both trials, the psoriasis area and severity index 75 response rate at week 16 was significantly
Use of other systemic agents such as tumor necrosis factor α inhibitors and ustekinumab has been reported in HIV-positive patients.5-7 There is no current data on IL-17 and IL-23 inhibitors. Acitretin generally is recommended as a second-line agent in HIV patients given its lack of immunosuppression2; however, methotrexate and cyclosporine should be avoided given the risk of opportunistic infections.8
Apremilast is a promising therapy with a favorable safety profile that should be considered as an adjuvant treatment to first-line agents such as phototherapy in HIV-positive patients. Apremilast has been successfully used in an HIV patient with a concomitant chronic hepatitis C infection.1 Systemic medications such as apremilast should be managed in coordination with infectious disease specialists with close monitoring of CD4 levels and viral loads as well as prophylactic agents.
To the Editor:
A 50-year old man with Fitzpatrick skin type IV, human immunodeficiency virus (HIV), fatty liver disease, and moderate psoriasis (10% body surface area [BSA] affected) currently treated with clobetasol spray and calcitriol ointment presented with persistent psoriatic lesions on the trunk, arms, legs, and buttocks. His CD4 count was 460 and his HIV RNA count was 48 copies/mL on polymerase chain reaction 2 months prior to the current presentation. He had been undergoing phototherapy 3 times weekly for the last 5 months for treatment of psoriasis.
At the current presentation, he was started on an apremilast starter pack with the dosage titrated from 10 mg to 30 mg over the course of 1 week. He was maintained on a dose of 30 mg twice daily after 1 week and continued clobetasol spray, calcitriol ointment, and phototherapy 3 times weekly with the intent to reduce the frequency after adequate control of psoriasis was achieved. After 3 months of treatment, the affected BSA was 0%. He continued apremilast, and phototherapy was reduced to once weekly. Phototherapy was discontinued after 7 months of concomitant treatment with apremilast after clearance was maintained. It was reinitiated twice weekly after a mild flare (3% BSA affected). After 20 total months of treatment, the patient was no longer able to afford apremilast treatment and presented with a severe psoriasis flare (40% BSA affected). He was switched to acitretin with a plan to apply for apremilast financial assistance programs.
Psoriasis treatment in the HIV population poses a challenge given the immunosuppressed state of these patients, the risk of reactivation of latent infections, and the refractory nature of psoriasis in the setting of HIV. Two of the authors (S.P.R. and J.J.W.) previously reported a case of moderate to severe psoriasis in a patient with HIV and hepatitis C who demonstrated treatment success with apremilast until it was discontinued due to financial implications.1 Currently, apremilast is not widely used to treat psoriasis in the HIV population. The National Psoriasis Foundation 2010 guidelines recommended UV light therapy for treatment of moderate to severe psoriasis in HIV-positive patients, with oral retinoids as the second-line treatment.2 There remains a need for updated guidelines on the use of systemic agents for psoriasis treatment in the HIV population.
Apremilast, a phosphodiesterase 4 inhibitor, is an oral therapy that restores the balance of proinflammatory and anti-inflammatory cytokines by inhibiting inflammatory cytokine (eg, tumor necrosis factor α, IFN-γ, IL-2, IL-12, IL-23) secretion and stimulating anti-inflammatory cytokine (eg, IL-6, IL-10) production. In 2015, the phase 3 ESTEEM 13 and ESTEEM 24 trials demonstrated the efficacy of apremilast 30 mg twice daily for treatment of psoriasis. In both trials, the psoriasis area and severity index 75 response rate at week 16 was significantly
Use of other systemic agents such as tumor necrosis factor α inhibitors and ustekinumab has been reported in HIV-positive patients.5-7 There is no current data on IL-17 and IL-23 inhibitors. Acitretin generally is recommended as a second-line agent in HIV patients given its lack of immunosuppression2; however, methotrexate and cyclosporine should be avoided given the risk of opportunistic infections.8
Apremilast is a promising therapy with a favorable safety profile that should be considered as an adjuvant treatment to first-line agents such as phototherapy in HIV-positive patients. Apremilast has been successfully used in an HIV patient with a concomitant chronic hepatitis C infection.1 Systemic medications such as apremilast should be managed in coordination with infectious disease specialists with close monitoring of CD4 levels and viral loads as well as prophylactic agents.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:e481-e482.
- Menon K, Van Voorhees AS, Bebo BF Jr, et al. Psoriasis in patients with HIV infection: from the medical board of the National Psoriasis Foundation [published online July 31, 2009]. J Am Acad Dermatol. 2010;62:291-299.
- Papp K, Reich K, Leonardi CL, et al. Apremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized, controlled trial (Efficacy and Safety Trial Evaluating the Effects of Apremilast in Psoriasis [ESTEEM] 1). J Am Acad Dermatol. 2015;73:37-49.
- Paul C, Cather J, Gooderham M, et al. Efficacy and safety of apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate-to-severe plaque psoriasis over 52 weeks: a phase III, randomized controlled trial (ESTEEM 2). Br J Dermatol. 2015;173:1387-1399.
- Lindsey SF, Weiss J, Lee ES, et al. Treatment of severe psoriasis and psoriatic arthritis with adalimumab in an HIV-positive patient. J Drugs Dermatol. 2014;13:869-871.
- Saeki H, Ito T, Hayashi M, et al. Successful treatment of ustekinumab in a severe psoriasis patient with human immunodeficiency virus infection. J Eur Acad Dermatol Venereol. 2015;29:1653-1655.
- Paparizos V, Rallis E, Kirsten L, et al. Ustekinumab for the treatment of HIV psoriasis. J Dermatolog Treat. 2012;23:398-399.
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections [published online July 11, 2018]. J Am Acad Dermatol. 2019;80:43-53.
- Reddy SP, Shah VV, Wu JJ. Apremilast for a psoriasis patient with HIV and hepatitis C. J Eur Acad Dermatol Venereol. 2017;31:e481-e482.
- Menon K, Van Voorhees AS, Bebo BF Jr, et al. Psoriasis in patients with HIV infection: from the medical board of the National Psoriasis Foundation [published online July 31, 2009]. J Am Acad Dermatol. 2010;62:291-299.
- Papp K, Reich K, Leonardi CL, et al. Apremilast, an oral phosphodiesterase 4 (PDE4) inhibitor, in patients with moderate to severe plaque psoriasis: results of a phase III, randomized, controlled trial (Efficacy and Safety Trial Evaluating the Effects of Apremilast in Psoriasis [ESTEEM] 1). J Am Acad Dermatol. 2015;73:37-49.
- Paul C, Cather J, Gooderham M, et al. Efficacy and safety of apremilast, an oral phosphodiesterase 4 inhibitor, in patients with moderate-to-severe plaque psoriasis over 52 weeks: a phase III, randomized controlled trial (ESTEEM 2). Br J Dermatol. 2015;173:1387-1399.
- Lindsey SF, Weiss J, Lee ES, et al. Treatment of severe psoriasis and psoriatic arthritis with adalimumab in an HIV-positive patient. J Drugs Dermatol. 2014;13:869-871.
- Saeki H, Ito T, Hayashi M, et al. Successful treatment of ustekinumab in a severe psoriasis patient with human immunodeficiency virus infection. J Eur Acad Dermatol Venereol. 2015;29:1653-1655.
- Paparizos V, Rallis E, Kirsten L, et al. Ustekinumab for the treatment of HIV psoriasis. J Dermatolog Treat. 2012;23:398-399.
- Kaushik SB, Lebwohl MG. Psoriasis: which therapy for which patient: focus on special populations and chronic infections [published online July 11, 2018]. J Am Acad Dermatol. 2019;80:43-53.
Practice Point
- Apremilast may be considered as a first-line therapy in the human immunodeficiency virus population due to decreased immunosuppression.
Mantle Cell Lymphoma Roundtable Discussion
Mantle cell lymphoma (MCL) is a rare, often aggressive form of non-Hodgkin lymphoma that develops when the body makes abnormal B-cells, and it is typically diagnosed at a later stage of disease. In this video series, Dr. Andre Goy sits down with Drs. Matthew Matasar and Peter Martin to discuss diagnosis, treatment, and unmet needs in MCL.

This video roundtable was produced by the Custom Programs division. The faculty received modest honoraria from Custom Programs for participating in this roundtable.
The faculty was solely responsible for the content presented.
Disclosures
Dr. Goy is on the speaker’s bureau and reports grant/research support from Acerta, Genentech, Kite/Gilead, Janssen, Pharmacyclics, and Takeda, and stocks/shares with COTA.
Dr. Matasar reports stock and other ownership interests with Merck; receiving honoraria from Bayer, Genentech, GlaxoSmithKline, Janssen, Pharmacyclics, Roche, and Seattle Genetics; consulting or advisory roles with Bayer, Genentech, Daiichi Sankyo, Juno Therapeutics, Merck, Roche, Rocket Medical, Seattle Genetics, and Teva; and research funding from Bayer, Genentech, GlaxoSmithKline, Janssen, Pharmacyclics, Roche, Rocket Medical, and Seattle Genetics.
Dr. Martin reports consulting for AstraZeneca, Bayer, Celgene, and Janssen.
Mantle cell lymphoma (MCL) is a rare, often aggressive form of non-Hodgkin lymphoma that develops when the body makes abnormal B-cells, and it is typically diagnosed at a later stage of disease. In this video series, Dr. Andre Goy sits down with Drs. Matthew Matasar and Peter Martin to discuss diagnosis, treatment, and unmet needs in MCL.

This video roundtable was produced by the Custom Programs division. The faculty received modest honoraria from Custom Programs for participating in this roundtable.
The faculty was solely responsible for the content presented.
Disclosures
Dr. Goy is on the speaker’s bureau and reports grant/research support from Acerta, Genentech, Kite/Gilead, Janssen, Pharmacyclics, and Takeda, and stocks/shares with COTA.
Dr. Matasar reports stock and other ownership interests with Merck; receiving honoraria from Bayer, Genentech, GlaxoSmithKline, Janssen, Pharmacyclics, Roche, and Seattle Genetics; consulting or advisory roles with Bayer, Genentech, Daiichi Sankyo, Juno Therapeutics, Merck, Roche, Rocket Medical, Seattle Genetics, and Teva; and research funding from Bayer, Genentech, GlaxoSmithKline, Janssen, Pharmacyclics, Roche, Rocket Medical, and Seattle Genetics.
Dr. Martin reports consulting for AstraZeneca, Bayer, Celgene, and Janssen.
Mantle cell lymphoma (MCL) is a rare, often aggressive form of non-Hodgkin lymphoma that develops when the body makes abnormal B-cells, and it is typically diagnosed at a later stage of disease. In this video series, Dr. Andre Goy sits down with Drs. Matthew Matasar and Peter Martin to discuss diagnosis, treatment, and unmet needs in MCL.

This video roundtable was produced by the Custom Programs division. The faculty received modest honoraria from Custom Programs for participating in this roundtable.
The faculty was solely responsible for the content presented.
Disclosures
Dr. Goy is on the speaker’s bureau and reports grant/research support from Acerta, Genentech, Kite/Gilead, Janssen, Pharmacyclics, and Takeda, and stocks/shares with COTA.
Dr. Matasar reports stock and other ownership interests with Merck; receiving honoraria from Bayer, Genentech, GlaxoSmithKline, Janssen, Pharmacyclics, Roche, and Seattle Genetics; consulting or advisory roles with Bayer, Genentech, Daiichi Sankyo, Juno Therapeutics, Merck, Roche, Rocket Medical, Seattle Genetics, and Teva; and research funding from Bayer, Genentech, GlaxoSmithKline, Janssen, Pharmacyclics, Roche, Rocket Medical, and Seattle Genetics.
Dr. Martin reports consulting for AstraZeneca, Bayer, Celgene, and Janssen.
CSF and plasma biomarkers predict survival in sporadic Creutzfeldt-Jakob disease
, according to research published online ahead of print May 6 in JAMA Neurology. Levels of total tau in plasma and CSF are correlated, and plasma total tau is associated with survival time. These findings suggest that plasma total tau level could be a valid biomarker that guides clinical care for patients with sporadic Creutzfeldt-Jakob disease, said the investigators.
The accurate prediction of disease duration can assist clinicians and caregivers in clinical management, as well as influence the design of clinical trials. Previous studies have found that baseline protein levels in CSF and plasma are associated with disease duration in patients with sporadic Creutzfeldt-Jakob disease. To replicate these findings, Adam M. Staffaroni, PhD, of the department of neurology at the University of California, San Francisco, and colleagues conducted a longitudinal cohort study.
Evaluating fluid and nonfluid biomarkers
Dr. Staffaroni and colleagues recruited 193 participants with probable or definite sporadic Creutzfeldt-Jakob disease who had codon 129 genotyping and were referred to the UCSF Memory and Aging Center from March 2004 to January 2018. All participants underwent cognitive testing, informant measures, a neurologic examination, and CSF and blood sample collection. The researchers excluded from analysis five participants who had been placed on life-extending treatments. Participants were evaluated until death or censored at the time of statistical analysis.
Dr. Staffaroni and colleagues examined the following nonfluid biomarkers of survival: sex, age, codon 129 genotype, Barthel Index, and Medical Research Council (MRC) Prion Disease Rating Scale. In addition, they examined total tau level, phosphorylated tau level, total tau:phosphorylated tau ratio, neurofilament light (NfL) level, beta-amyloid 42 level, neuron-specific enolase level, 14-3-3 test result, and real-time quaking-induced conversion test in CSF as fluid biomarkers of survival. Finally, Dr. Staffaroni’s group analyzed total tau level, NfL level, and glial fibrillary acidic protein level in plasma as additional fluid biomarkers of survival.
The researchers fitted Cox proportional hazard models with time to event as the outcome. They log-transformed fluid biomarkers and ran models with and without nonfluid biomarkers of survival.
Plasma total tau was associated with survival
In all, 188 patients were included in the analysis. The population’s mean age was 63.8 years. Approximately 45% of participants were women. The diagnosis of sporadic Creutzfeldt-Jakob disease was pathologically confirmed for 78.2% of participants and probable for 21.8% of participants.
Dr. Staffaroni’s group observed strong correlations between plasma and CSF NfL concentrations and between plasma and CSF total tau concentrations. CSF total tau and CSF NfL concentrations were also correlated.
Among the nonfluid biomarkers, Barthel Index, MRC Scale, and codon 129 genotype were significantly associated with survival time. Lower level of function at baseline predicted a faster disease course.
Among the fluid biomarkers, greater levels of plasma total tau and NfL levels at baseline were associated with shorter survival. After the investigators controlled for Barthel Index and codon 129 genotype, the association of plasma total tau level with survival time remained significant. Plasma total tau level and Barthel Index (hazard ratio, 0.98) each independently predicted survival. Dr. Staffaroni and colleagues found that the hazard ratios for all CSF biomarkers were in the expected direction, and that those for total tau level, total tau:phosphorylated tau ratio, NfL level, and neuron-specific enolase level were statistically significant. Furthermore, positive results for 14-3-3 protein, neuron-specific enolase level, and total tau level were associated with a shorter time until death. Like the plasma biomarkers, CSF total tau level remained associated with survival after the investigators controlled for Barthel Index and codon 129 genotype. The same was true of CSF total tau:phosphorylated tau ratio, neuron-specific enolase level, and 14-3-3 result.
Plasma tau could be a diagnostic biomarker
“The hazard ratio associated with plasma total tau level was more than 40% higher than other fluid biomarkers of interest,” said the authors. “These findings further bolster the value of blood-based biomarkers, based on their minimally invasive and relatively inexpensive nature, and build on prior studies that suggested patients with sporadic Creutzfeldt-Jakob disease and controls can be discriminated with relatively high accuracy using blood-based assays.” When Dr. Staffaroni and colleagues modeled baseline functional status and plasma total tau levels together, they found that both were independent predictors of survival time. “This [finding] suggests that clinical measures and plasma total tau level could be combined to further improve prediction accuracy.”
Among the study’s limitations was its comparatively small subsample of patients for whom all plasma and CSF biomarkers were available. Disease duration and survival were longer in the study population than in the literature. “Another limitation is that the plasma biomarkers in this study, one of which showed great promise for predicting survival, were assayed using a research protocol,” said the researchers. “Widespread clinical use of these biomarkers will require well-validated commercial assays, development of which is underway.”
Before these biomarkers can be used in the clinic, these results will need to be replicated, and neurologists will need to develop consensus cutoffs for the biomarker levels. The researchers did not analyze plasma tau level as a diagnostic biomarker, but future studies should examine this potential, Dr. Staffaroni and colleagues concluded.
Grants from the National Institute on Aging and the National Institute of Allergy and Infectious Diseases supported the study.
SOURCE: Staffaroni AM et al. JAMA Neurol. 2019 May 6. doi: 10.1001/jamaneurol.2019.1071.
, according to research published online ahead of print May 6 in JAMA Neurology. Levels of total tau in plasma and CSF are correlated, and plasma total tau is associated with survival time. These findings suggest that plasma total tau level could be a valid biomarker that guides clinical care for patients with sporadic Creutzfeldt-Jakob disease, said the investigators.
The accurate prediction of disease duration can assist clinicians and caregivers in clinical management, as well as influence the design of clinical trials. Previous studies have found that baseline protein levels in CSF and plasma are associated with disease duration in patients with sporadic Creutzfeldt-Jakob disease. To replicate these findings, Adam M. Staffaroni, PhD, of the department of neurology at the University of California, San Francisco, and colleagues conducted a longitudinal cohort study.
Evaluating fluid and nonfluid biomarkers
Dr. Staffaroni and colleagues recruited 193 participants with probable or definite sporadic Creutzfeldt-Jakob disease who had codon 129 genotyping and were referred to the UCSF Memory and Aging Center from March 2004 to January 2018. All participants underwent cognitive testing, informant measures, a neurologic examination, and CSF and blood sample collection. The researchers excluded from analysis five participants who had been placed on life-extending treatments. Participants were evaluated until death or censored at the time of statistical analysis.
Dr. Staffaroni and colleagues examined the following nonfluid biomarkers of survival: sex, age, codon 129 genotype, Barthel Index, and Medical Research Council (MRC) Prion Disease Rating Scale. In addition, they examined total tau level, phosphorylated tau level, total tau:phosphorylated tau ratio, neurofilament light (NfL) level, beta-amyloid 42 level, neuron-specific enolase level, 14-3-3 test result, and real-time quaking-induced conversion test in CSF as fluid biomarkers of survival. Finally, Dr. Staffaroni’s group analyzed total tau level, NfL level, and glial fibrillary acidic protein level in plasma as additional fluid biomarkers of survival.
The researchers fitted Cox proportional hazard models with time to event as the outcome. They log-transformed fluid biomarkers and ran models with and without nonfluid biomarkers of survival.
Plasma total tau was associated with survival
In all, 188 patients were included in the analysis. The population’s mean age was 63.8 years. Approximately 45% of participants were women. The diagnosis of sporadic Creutzfeldt-Jakob disease was pathologically confirmed for 78.2% of participants and probable for 21.8% of participants.
Dr. Staffaroni’s group observed strong correlations between plasma and CSF NfL concentrations and between plasma and CSF total tau concentrations. CSF total tau and CSF NfL concentrations were also correlated.
Among the nonfluid biomarkers, Barthel Index, MRC Scale, and codon 129 genotype were significantly associated with survival time. Lower level of function at baseline predicted a faster disease course.
Among the fluid biomarkers, greater levels of plasma total tau and NfL levels at baseline were associated with shorter survival. After the investigators controlled for Barthel Index and codon 129 genotype, the association of plasma total tau level with survival time remained significant. Plasma total tau level and Barthel Index (hazard ratio, 0.98) each independently predicted survival. Dr. Staffaroni and colleagues found that the hazard ratios for all CSF biomarkers were in the expected direction, and that those for total tau level, total tau:phosphorylated tau ratio, NfL level, and neuron-specific enolase level were statistically significant. Furthermore, positive results for 14-3-3 protein, neuron-specific enolase level, and total tau level were associated with a shorter time until death. Like the plasma biomarkers, CSF total tau level remained associated with survival after the investigators controlled for Barthel Index and codon 129 genotype. The same was true of CSF total tau:phosphorylated tau ratio, neuron-specific enolase level, and 14-3-3 result.
Plasma tau could be a diagnostic biomarker
“The hazard ratio associated with plasma total tau level was more than 40% higher than other fluid biomarkers of interest,” said the authors. “These findings further bolster the value of blood-based biomarkers, based on their minimally invasive and relatively inexpensive nature, and build on prior studies that suggested patients with sporadic Creutzfeldt-Jakob disease and controls can be discriminated with relatively high accuracy using blood-based assays.” When Dr. Staffaroni and colleagues modeled baseline functional status and plasma total tau levels together, they found that both were independent predictors of survival time. “This [finding] suggests that clinical measures and plasma total tau level could be combined to further improve prediction accuracy.”
Among the study’s limitations was its comparatively small subsample of patients for whom all plasma and CSF biomarkers were available. Disease duration and survival were longer in the study population than in the literature. “Another limitation is that the plasma biomarkers in this study, one of which showed great promise for predicting survival, were assayed using a research protocol,” said the researchers. “Widespread clinical use of these biomarkers will require well-validated commercial assays, development of which is underway.”
Before these biomarkers can be used in the clinic, these results will need to be replicated, and neurologists will need to develop consensus cutoffs for the biomarker levels. The researchers did not analyze plasma tau level as a diagnostic biomarker, but future studies should examine this potential, Dr. Staffaroni and colleagues concluded.
Grants from the National Institute on Aging and the National Institute of Allergy and Infectious Diseases supported the study.
SOURCE: Staffaroni AM et al. JAMA Neurol. 2019 May 6. doi: 10.1001/jamaneurol.2019.1071.
, according to research published online ahead of print May 6 in JAMA Neurology. Levels of total tau in plasma and CSF are correlated, and plasma total tau is associated with survival time. These findings suggest that plasma total tau level could be a valid biomarker that guides clinical care for patients with sporadic Creutzfeldt-Jakob disease, said the investigators.
The accurate prediction of disease duration can assist clinicians and caregivers in clinical management, as well as influence the design of clinical trials. Previous studies have found that baseline protein levels in CSF and plasma are associated with disease duration in patients with sporadic Creutzfeldt-Jakob disease. To replicate these findings, Adam M. Staffaroni, PhD, of the department of neurology at the University of California, San Francisco, and colleagues conducted a longitudinal cohort study.
Evaluating fluid and nonfluid biomarkers
Dr. Staffaroni and colleagues recruited 193 participants with probable or definite sporadic Creutzfeldt-Jakob disease who had codon 129 genotyping and were referred to the UCSF Memory and Aging Center from March 2004 to January 2018. All participants underwent cognitive testing, informant measures, a neurologic examination, and CSF and blood sample collection. The researchers excluded from analysis five participants who had been placed on life-extending treatments. Participants were evaluated until death or censored at the time of statistical analysis.
Dr. Staffaroni and colleagues examined the following nonfluid biomarkers of survival: sex, age, codon 129 genotype, Barthel Index, and Medical Research Council (MRC) Prion Disease Rating Scale. In addition, they examined total tau level, phosphorylated tau level, total tau:phosphorylated tau ratio, neurofilament light (NfL) level, beta-amyloid 42 level, neuron-specific enolase level, 14-3-3 test result, and real-time quaking-induced conversion test in CSF as fluid biomarkers of survival. Finally, Dr. Staffaroni’s group analyzed total tau level, NfL level, and glial fibrillary acidic protein level in plasma as additional fluid biomarkers of survival.
The researchers fitted Cox proportional hazard models with time to event as the outcome. They log-transformed fluid biomarkers and ran models with and without nonfluid biomarkers of survival.
Plasma total tau was associated with survival
In all, 188 patients were included in the analysis. The population’s mean age was 63.8 years. Approximately 45% of participants were women. The diagnosis of sporadic Creutzfeldt-Jakob disease was pathologically confirmed for 78.2% of participants and probable for 21.8% of participants.
Dr. Staffaroni’s group observed strong correlations between plasma and CSF NfL concentrations and between plasma and CSF total tau concentrations. CSF total tau and CSF NfL concentrations were also correlated.
Among the nonfluid biomarkers, Barthel Index, MRC Scale, and codon 129 genotype were significantly associated with survival time. Lower level of function at baseline predicted a faster disease course.
Among the fluid biomarkers, greater levels of plasma total tau and NfL levels at baseline were associated with shorter survival. After the investigators controlled for Barthel Index and codon 129 genotype, the association of plasma total tau level with survival time remained significant. Plasma total tau level and Barthel Index (hazard ratio, 0.98) each independently predicted survival. Dr. Staffaroni and colleagues found that the hazard ratios for all CSF biomarkers were in the expected direction, and that those for total tau level, total tau:phosphorylated tau ratio, NfL level, and neuron-specific enolase level were statistically significant. Furthermore, positive results for 14-3-3 protein, neuron-specific enolase level, and total tau level were associated with a shorter time until death. Like the plasma biomarkers, CSF total tau level remained associated with survival after the investigators controlled for Barthel Index and codon 129 genotype. The same was true of CSF total tau:phosphorylated tau ratio, neuron-specific enolase level, and 14-3-3 result.
Plasma tau could be a diagnostic biomarker
“The hazard ratio associated with plasma total tau level was more than 40% higher than other fluid biomarkers of interest,” said the authors. “These findings further bolster the value of blood-based biomarkers, based on their minimally invasive and relatively inexpensive nature, and build on prior studies that suggested patients with sporadic Creutzfeldt-Jakob disease and controls can be discriminated with relatively high accuracy using blood-based assays.” When Dr. Staffaroni and colleagues modeled baseline functional status and plasma total tau levels together, they found that both were independent predictors of survival time. “This [finding] suggests that clinical measures and plasma total tau level could be combined to further improve prediction accuracy.”
Among the study’s limitations was its comparatively small subsample of patients for whom all plasma and CSF biomarkers were available. Disease duration and survival were longer in the study population than in the literature. “Another limitation is that the plasma biomarkers in this study, one of which showed great promise for predicting survival, were assayed using a research protocol,” said the researchers. “Widespread clinical use of these biomarkers will require well-validated commercial assays, development of which is underway.”
Before these biomarkers can be used in the clinic, these results will need to be replicated, and neurologists will need to develop consensus cutoffs for the biomarker levels. The researchers did not analyze plasma tau level as a diagnostic biomarker, but future studies should examine this potential, Dr. Staffaroni and colleagues concluded.
Grants from the National Institute on Aging and the National Institute of Allergy and Infectious Diseases supported the study.
SOURCE: Staffaroni AM et al. JAMA Neurol. 2019 May 6. doi: 10.1001/jamaneurol.2019.1071.
FROM JAMA NEUROLOGY
Key clinical point: Invasive and minimally invasive biomarkers are associated with survival in sporadic Creutzfeldt-Jakob disease.
Major finding: The hazard ratio for plasma total tau level was more than 40% larger than any other biomarker.
Study details: A longitudinal study of 188 participants with sporadic Creutzfeldt-Jakob disease.
Disclosures: Grants from the National Institute on Aging and the National Institute of Allergy and Infectious Diseases supported the study.
Source: Staffaroni AM et al. JAMA Neurol. 2019 May 6. doi: 10.1001/jamaneurol.2019.1071.
What do patients want in a migraine preventive?
, according to the results of a study published in Headache. When offered hypothetical preventive migraine medicines with a wide array of attributes, patients leaned toward those with a reduction in migraine days and an avoidance of weight gain, according to an analysis of responses to a discrete-choice experiment survey.
“We found that respondents had a significant willingness to pay for medicines with higher efficacy and less-severe adverse events,” wrote Carol Mansfield, PhD, of RTI Health Solutions in North Carolina, and coauthors.
To evaluate patient preferences for theoretical migraine medicine, the researchers conducted a discrete-choice experiment via a web-based survey. Respondents met eligibility criteria if they were adults aged 18 years or older who self-reported 6 or more migraine days per month and completed the survey in full. They were asked to choose between options defined by six attributes: reduction in headache days per month, frequency of limitations with physical activities, cognition problems, weight gain, how the medicine is taken, and monthly out-of-pocket cost.
Of the 300 respondents included in the analysis, 72% indicated that migraines make physical activities difficult all or most of the time, and 81% had taken a prescription migraine preventive in the last 6 months. Respondents reported, on average, approximately 16 headache days per month. Among noncost attributes, respondents valued a change from a 10% reduction in migraine days to a 50% reduction more highly than avoiding the worst levels of adverse events – defined as memory problems and 10% weight gain – but were willing to trade off efficacy for less-severe adverse events. Avoiding memory problems was more important than avoiding thinking problems. Avoiding a 10% weight gain was more important than avoiding thinking and memory problems. Respondents preferred a once-monthly injection or daily pill to twice-monthly injections. Respondents, on average, were willing to pay $116 per month for an improvement from 10% to 50% in reduced headache days (95% confidence interval [CI], $91-$141) and $43 for an improvement from 10% to 25% (95% CI, $34-$53). They were also willing to pay $84 per month to avoid a 10% weight gain (95% CI, $64-$103), $59 per month to avoid memory problems (95% CI, $42-$76), and $32 per month to avoid thinking problems (95% CI, $18-$46).
The coauthors acknowledged their study’s limitations, including all migraine diagnoses being self-reported and the study sample not necessarily being representative of patients with migraine overall. In addition, though the potential medicinal attributes used were prominent in clinical literature and focus groups, they could choose only a limited amount and so their analysis “did not address other attributes that may be important to patients.”
Given their findings, the researchers recommended that “clinicians should work with patients to select treatments that meet each patient’s needs.”
Amgen and Novartis funded the study. The authors reported numerous conflicts of interest, including receiving grants, consulting fees, and royalties from pharmaceutical companies and organizations. During the study, three of the authors were employed at RTI Health Solutions, a non-for-profit organization that conducts research with pharmaceutical companies such as the study’s sponsor.
SOURCE: Mansfield C et al. Headache. 2019 May;59(5):715-26. doi: 10.1111/head.13498.
, according to the results of a study published in Headache. When offered hypothetical preventive migraine medicines with a wide array of attributes, patients leaned toward those with a reduction in migraine days and an avoidance of weight gain, according to an analysis of responses to a discrete-choice experiment survey.
“We found that respondents had a significant willingness to pay for medicines with higher efficacy and less-severe adverse events,” wrote Carol Mansfield, PhD, of RTI Health Solutions in North Carolina, and coauthors.
To evaluate patient preferences for theoretical migraine medicine, the researchers conducted a discrete-choice experiment via a web-based survey. Respondents met eligibility criteria if they were adults aged 18 years or older who self-reported 6 or more migraine days per month and completed the survey in full. They were asked to choose between options defined by six attributes: reduction in headache days per month, frequency of limitations with physical activities, cognition problems, weight gain, how the medicine is taken, and monthly out-of-pocket cost.
Of the 300 respondents included in the analysis, 72% indicated that migraines make physical activities difficult all or most of the time, and 81% had taken a prescription migraine preventive in the last 6 months. Respondents reported, on average, approximately 16 headache days per month. Among noncost attributes, respondents valued a change from a 10% reduction in migraine days to a 50% reduction more highly than avoiding the worst levels of adverse events – defined as memory problems and 10% weight gain – but were willing to trade off efficacy for less-severe adverse events. Avoiding memory problems was more important than avoiding thinking problems. Avoiding a 10% weight gain was more important than avoiding thinking and memory problems. Respondents preferred a once-monthly injection or daily pill to twice-monthly injections. Respondents, on average, were willing to pay $116 per month for an improvement from 10% to 50% in reduced headache days (95% confidence interval [CI], $91-$141) and $43 for an improvement from 10% to 25% (95% CI, $34-$53). They were also willing to pay $84 per month to avoid a 10% weight gain (95% CI, $64-$103), $59 per month to avoid memory problems (95% CI, $42-$76), and $32 per month to avoid thinking problems (95% CI, $18-$46).
The coauthors acknowledged their study’s limitations, including all migraine diagnoses being self-reported and the study sample not necessarily being representative of patients with migraine overall. In addition, though the potential medicinal attributes used were prominent in clinical literature and focus groups, they could choose only a limited amount and so their analysis “did not address other attributes that may be important to patients.”
Given their findings, the researchers recommended that “clinicians should work with patients to select treatments that meet each patient’s needs.”
Amgen and Novartis funded the study. The authors reported numerous conflicts of interest, including receiving grants, consulting fees, and royalties from pharmaceutical companies and organizations. During the study, three of the authors were employed at RTI Health Solutions, a non-for-profit organization that conducts research with pharmaceutical companies such as the study’s sponsor.
SOURCE: Mansfield C et al. Headache. 2019 May;59(5):715-26. doi: 10.1111/head.13498.
, according to the results of a study published in Headache. When offered hypothetical preventive migraine medicines with a wide array of attributes, patients leaned toward those with a reduction in migraine days and an avoidance of weight gain, according to an analysis of responses to a discrete-choice experiment survey.
“We found that respondents had a significant willingness to pay for medicines with higher efficacy and less-severe adverse events,” wrote Carol Mansfield, PhD, of RTI Health Solutions in North Carolina, and coauthors.
To evaluate patient preferences for theoretical migraine medicine, the researchers conducted a discrete-choice experiment via a web-based survey. Respondents met eligibility criteria if they were adults aged 18 years or older who self-reported 6 or more migraine days per month and completed the survey in full. They were asked to choose between options defined by six attributes: reduction in headache days per month, frequency of limitations with physical activities, cognition problems, weight gain, how the medicine is taken, and monthly out-of-pocket cost.
Of the 300 respondents included in the analysis, 72% indicated that migraines make physical activities difficult all or most of the time, and 81% had taken a prescription migraine preventive in the last 6 months. Respondents reported, on average, approximately 16 headache days per month. Among noncost attributes, respondents valued a change from a 10% reduction in migraine days to a 50% reduction more highly than avoiding the worst levels of adverse events – defined as memory problems and 10% weight gain – but were willing to trade off efficacy for less-severe adverse events. Avoiding memory problems was more important than avoiding thinking problems. Avoiding a 10% weight gain was more important than avoiding thinking and memory problems. Respondents preferred a once-monthly injection or daily pill to twice-monthly injections. Respondents, on average, were willing to pay $116 per month for an improvement from 10% to 50% in reduced headache days (95% confidence interval [CI], $91-$141) and $43 for an improvement from 10% to 25% (95% CI, $34-$53). They were also willing to pay $84 per month to avoid a 10% weight gain (95% CI, $64-$103), $59 per month to avoid memory problems (95% CI, $42-$76), and $32 per month to avoid thinking problems (95% CI, $18-$46).
The coauthors acknowledged their study’s limitations, including all migraine diagnoses being self-reported and the study sample not necessarily being representative of patients with migraine overall. In addition, though the potential medicinal attributes used were prominent in clinical literature and focus groups, they could choose only a limited amount and so their analysis “did not address other attributes that may be important to patients.”
Given their findings, the researchers recommended that “clinicians should work with patients to select treatments that meet each patient’s needs.”
Amgen and Novartis funded the study. The authors reported numerous conflicts of interest, including receiving grants, consulting fees, and royalties from pharmaceutical companies and organizations. During the study, three of the authors were employed at RTI Health Solutions, a non-for-profit organization that conducts research with pharmaceutical companies such as the study’s sponsor.
SOURCE: Mansfield C et al. Headache. 2019 May;59(5):715-26. doi: 10.1111/head.13498.
FROM HEADACHE
Cluster headache is associated with increased suicidality
Short- and long-term cluster headache disease burden, as well as depressive symptoms, contributes to suicidality, according to research published online Cephalalgia. Development of treatments that reduce the headache-related burden and prevent future bouts could reduce suicidality, said the researchers.
Although cluster headache has been called the “suicide headache,” few studies have examined suicidality in patients with cluster headache. Research by Rozen et al. found that the rate of suicidal attempt among patients was similar to that among the general population. The results have not been replicated, however, and the investigators did not examine whether suicidality varied according to the phases of the disorder.
A prospective, multicenter study
Mi Ji Lee, MD, PhD, clinical assistant professor of neurology at Samsung Medical Center in Seoul, South Korea, and colleagues conducted a prospective study to investigate the suicidality associated with cluster headache and the factors associated with increased suicidality in that disorder. The researchers enrolled 193 consecutive patients with cluster headache between September 2016 and August 2018 at 15 hospitals. They examined the patients and used the Patient Health Questionnaire–9 (PHQ-9) and the General Anxiety Disorder–7 item scale (GAD-7) screening tools. During the ictal and interictal phases, the researchers asked the patients whether they had had passive suicidal ideation, active suicidal ideation, suicidal planning, or suicidal attempt. Dr. Ji Lee and colleagues performed univariable and multivariable logistic regression analyses to evaluate the factors associated with high ictal suicidality, which was defined as two or more positive responses during the ictal phase. Participants were followed up during the between-bout phase.
The researchers excluded 18 patients from analysis because they were between bouts at enrollment. The mean age of the remaining 175 patients was 38.4 years. Mean age at onset was 29.9 years. About 85% of the patients were male. The diagnosis was definite cluster headache for 87.4% of the sample and probable cluster headache for 12.6%. In addition, 88% of the population had episodic cluster headache.
Suicidal ideation increased during the ictal phase
During the ictal phase, 64.2% of participants reported passive suicidal ideation, and 35.8% reported active suicidal ideation. Furthermore, 5.8% of patients had a suicidal plan, and 2.3% attempted suicide. In the interictal phase, 4.0% of patients reported passive suicidal ideation, and 3.5% reported active suicidal ideation. Interictal suicidal planning was reported by 2.9% of participants, and 1.2% of participants attempted suicide interictally. The results were similar between patients with definite and probable cluster headache.
The ictal phase increased the odds of passive suicidal ideation (odds ratio [OR], 42.46), active suicidal ideation (OR, 15.55), suicidal planning (OR, 2.06), and suicidal attempt (OR, 2.02), compared with the interictal phase. The differences in suicidal planning and suicidal attempt between the ictal and interictal phases, however, were not statistically significant.
Longer disease duration, higher attack intensity, higher Headache Impact Test–6 (HIT-6) score, GAD-7 score, and PHQ-9 score were associated with high ictal suicidality. Disease duration, HIT-6, and PHQ-9 remained significantly associated with high ictal suicidality in the multivariate analysis. Younger age at onset, longer disease duration, total number of lifetime bouts, and higher GAD-7 and PHQ-9 scores were significantly associated with interictal suicidality in the univariable analysis. The total number of lifetime bouts and the PHQ-9 scores remained significant in the multivariable analysis.
In all, 54 patients were followed up between bouts. None reported passive suicidal ideation, 1.9% reported active suicidal ideation, 1.9% reported suicidal planning, and none reported suicidal attempt. Compared with the between-bouts period, the ictal phase was associated with significantly higher odds of active suicidal ideation (OR, 37.32) and nonsignificantly increased suicidal planning (OR, 3.20).
Patients need a disease-modifying treatment
Taken together, the study results underscore the importance of proper management of cluster headache to reduce its burden, said the authors. “Given that greater headache-related impact was independently associated with ictal suicidality, an intensive treatment to reduce the headache-related impact might be beneficial to prevent suicide in cluster headache patients,” they said. In addition to reducing headache-related impact and headache intensity, “a disease-modifying treatment to prevent further bouts is warranted to decrease suicidality in cluster headache patients.”
Although patients with cluster headache had increased suicidality in the ictal and interictal phases, they had lower suicidality between bouts, compared with the general population. This result suggests that patients remain mentally healthy when the bouts are over, and that “a strategy to shorten the length of bout is warranted,” said Dr. Ji Lee and colleagues. Furthermore, the fact that suicidality did not differ significantly between patients with definite cluster headache and those with probable cluster headache “prompts clinicians for an increased identification and intensive treatment strategy for probable cluster headache.”
The current study is the first prospective investigation of suicidality in the various phases of cluster headache, according to the investigators. It nevertheless has several limitations. The prevalence of chronic cluster headache was low in the study population, and not all patients presented for follow-up during the period between bouts. In addition, the data were obtained from recall, and consequently may be less accurate than those gained from prospective recording. Finally, Dr. Ji Lee and colleagues did not gather information on personality disorders, insomnia, substance abuse, or addiction, even though these factors can influence suicidality in patients with chronic pain.
The investigators reported no conflicts of interest related to their research. The study was supported by a grant from the Korean Neurological Association.
SOURCE: Ji Lee M et al. Cephalalgia. 2019 Apr 24. doi: 10.1177/0333102419845660.
Short- and long-term cluster headache disease burden, as well as depressive symptoms, contributes to suicidality, according to research published online Cephalalgia. Development of treatments that reduce the headache-related burden and prevent future bouts could reduce suicidality, said the researchers.
Although cluster headache has been called the “suicide headache,” few studies have examined suicidality in patients with cluster headache. Research by Rozen et al. found that the rate of suicidal attempt among patients was similar to that among the general population. The results have not been replicated, however, and the investigators did not examine whether suicidality varied according to the phases of the disorder.
A prospective, multicenter study
Mi Ji Lee, MD, PhD, clinical assistant professor of neurology at Samsung Medical Center in Seoul, South Korea, and colleagues conducted a prospective study to investigate the suicidality associated with cluster headache and the factors associated with increased suicidality in that disorder. The researchers enrolled 193 consecutive patients with cluster headache between September 2016 and August 2018 at 15 hospitals. They examined the patients and used the Patient Health Questionnaire–9 (PHQ-9) and the General Anxiety Disorder–7 item scale (GAD-7) screening tools. During the ictal and interictal phases, the researchers asked the patients whether they had had passive suicidal ideation, active suicidal ideation, suicidal planning, or suicidal attempt. Dr. Ji Lee and colleagues performed univariable and multivariable logistic regression analyses to evaluate the factors associated with high ictal suicidality, which was defined as two or more positive responses during the ictal phase. Participants were followed up during the between-bout phase.
The researchers excluded 18 patients from analysis because they were between bouts at enrollment. The mean age of the remaining 175 patients was 38.4 years. Mean age at onset was 29.9 years. About 85% of the patients were male. The diagnosis was definite cluster headache for 87.4% of the sample and probable cluster headache for 12.6%. In addition, 88% of the population had episodic cluster headache.
Suicidal ideation increased during the ictal phase
During the ictal phase, 64.2% of participants reported passive suicidal ideation, and 35.8% reported active suicidal ideation. Furthermore, 5.8% of patients had a suicidal plan, and 2.3% attempted suicide. In the interictal phase, 4.0% of patients reported passive suicidal ideation, and 3.5% reported active suicidal ideation. Interictal suicidal planning was reported by 2.9% of participants, and 1.2% of participants attempted suicide interictally. The results were similar between patients with definite and probable cluster headache.
The ictal phase increased the odds of passive suicidal ideation (odds ratio [OR], 42.46), active suicidal ideation (OR, 15.55), suicidal planning (OR, 2.06), and suicidal attempt (OR, 2.02), compared with the interictal phase. The differences in suicidal planning and suicidal attempt between the ictal and interictal phases, however, were not statistically significant.
Longer disease duration, higher attack intensity, higher Headache Impact Test–6 (HIT-6) score, GAD-7 score, and PHQ-9 score were associated with high ictal suicidality. Disease duration, HIT-6, and PHQ-9 remained significantly associated with high ictal suicidality in the multivariate analysis. Younger age at onset, longer disease duration, total number of lifetime bouts, and higher GAD-7 and PHQ-9 scores were significantly associated with interictal suicidality in the univariable analysis. The total number of lifetime bouts and the PHQ-9 scores remained significant in the multivariable analysis.
In all, 54 patients were followed up between bouts. None reported passive suicidal ideation, 1.9% reported active suicidal ideation, 1.9% reported suicidal planning, and none reported suicidal attempt. Compared with the between-bouts period, the ictal phase was associated with significantly higher odds of active suicidal ideation (OR, 37.32) and nonsignificantly increased suicidal planning (OR, 3.20).
Patients need a disease-modifying treatment
Taken together, the study results underscore the importance of proper management of cluster headache to reduce its burden, said the authors. “Given that greater headache-related impact was independently associated with ictal suicidality, an intensive treatment to reduce the headache-related impact might be beneficial to prevent suicide in cluster headache patients,” they said. In addition to reducing headache-related impact and headache intensity, “a disease-modifying treatment to prevent further bouts is warranted to decrease suicidality in cluster headache patients.”
Although patients with cluster headache had increased suicidality in the ictal and interictal phases, they had lower suicidality between bouts, compared with the general population. This result suggests that patients remain mentally healthy when the bouts are over, and that “a strategy to shorten the length of bout is warranted,” said Dr. Ji Lee and colleagues. Furthermore, the fact that suicidality did not differ significantly between patients with definite cluster headache and those with probable cluster headache “prompts clinicians for an increased identification and intensive treatment strategy for probable cluster headache.”
The current study is the first prospective investigation of suicidality in the various phases of cluster headache, according to the investigators. It nevertheless has several limitations. The prevalence of chronic cluster headache was low in the study population, and not all patients presented for follow-up during the period between bouts. In addition, the data were obtained from recall, and consequently may be less accurate than those gained from prospective recording. Finally, Dr. Ji Lee and colleagues did not gather information on personality disorders, insomnia, substance abuse, or addiction, even though these factors can influence suicidality in patients with chronic pain.
The investigators reported no conflicts of interest related to their research. The study was supported by a grant from the Korean Neurological Association.
SOURCE: Ji Lee M et al. Cephalalgia. 2019 Apr 24. doi: 10.1177/0333102419845660.
Short- and long-term cluster headache disease burden, as well as depressive symptoms, contributes to suicidality, according to research published online Cephalalgia. Development of treatments that reduce the headache-related burden and prevent future bouts could reduce suicidality, said the researchers.
Although cluster headache has been called the “suicide headache,” few studies have examined suicidality in patients with cluster headache. Research by Rozen et al. found that the rate of suicidal attempt among patients was similar to that among the general population. The results have not been replicated, however, and the investigators did not examine whether suicidality varied according to the phases of the disorder.
A prospective, multicenter study
Mi Ji Lee, MD, PhD, clinical assistant professor of neurology at Samsung Medical Center in Seoul, South Korea, and colleagues conducted a prospective study to investigate the suicidality associated with cluster headache and the factors associated with increased suicidality in that disorder. The researchers enrolled 193 consecutive patients with cluster headache between September 2016 and August 2018 at 15 hospitals. They examined the patients and used the Patient Health Questionnaire–9 (PHQ-9) and the General Anxiety Disorder–7 item scale (GAD-7) screening tools. During the ictal and interictal phases, the researchers asked the patients whether they had had passive suicidal ideation, active suicidal ideation, suicidal planning, or suicidal attempt. Dr. Ji Lee and colleagues performed univariable and multivariable logistic regression analyses to evaluate the factors associated with high ictal suicidality, which was defined as two or more positive responses during the ictal phase. Participants were followed up during the between-bout phase.
The researchers excluded 18 patients from analysis because they were between bouts at enrollment. The mean age of the remaining 175 patients was 38.4 years. Mean age at onset was 29.9 years. About 85% of the patients were male. The diagnosis was definite cluster headache for 87.4% of the sample and probable cluster headache for 12.6%. In addition, 88% of the population had episodic cluster headache.
Suicidal ideation increased during the ictal phase
During the ictal phase, 64.2% of participants reported passive suicidal ideation, and 35.8% reported active suicidal ideation. Furthermore, 5.8% of patients had a suicidal plan, and 2.3% attempted suicide. In the interictal phase, 4.0% of patients reported passive suicidal ideation, and 3.5% reported active suicidal ideation. Interictal suicidal planning was reported by 2.9% of participants, and 1.2% of participants attempted suicide interictally. The results were similar between patients with definite and probable cluster headache.
The ictal phase increased the odds of passive suicidal ideation (odds ratio [OR], 42.46), active suicidal ideation (OR, 15.55), suicidal planning (OR, 2.06), and suicidal attempt (OR, 2.02), compared with the interictal phase. The differences in suicidal planning and suicidal attempt between the ictal and interictal phases, however, were not statistically significant.
Longer disease duration, higher attack intensity, higher Headache Impact Test–6 (HIT-6) score, GAD-7 score, and PHQ-9 score were associated with high ictal suicidality. Disease duration, HIT-6, and PHQ-9 remained significantly associated with high ictal suicidality in the multivariate analysis. Younger age at onset, longer disease duration, total number of lifetime bouts, and higher GAD-7 and PHQ-9 scores were significantly associated with interictal suicidality in the univariable analysis. The total number of lifetime bouts and the PHQ-9 scores remained significant in the multivariable analysis.
In all, 54 patients were followed up between bouts. None reported passive suicidal ideation, 1.9% reported active suicidal ideation, 1.9% reported suicidal planning, and none reported suicidal attempt. Compared with the between-bouts period, the ictal phase was associated with significantly higher odds of active suicidal ideation (OR, 37.32) and nonsignificantly increased suicidal planning (OR, 3.20).
Patients need a disease-modifying treatment
Taken together, the study results underscore the importance of proper management of cluster headache to reduce its burden, said the authors. “Given that greater headache-related impact was independently associated with ictal suicidality, an intensive treatment to reduce the headache-related impact might be beneficial to prevent suicide in cluster headache patients,” they said. In addition to reducing headache-related impact and headache intensity, “a disease-modifying treatment to prevent further bouts is warranted to decrease suicidality in cluster headache patients.”
Although patients with cluster headache had increased suicidality in the ictal and interictal phases, they had lower suicidality between bouts, compared with the general population. This result suggests that patients remain mentally healthy when the bouts are over, and that “a strategy to shorten the length of bout is warranted,” said Dr. Ji Lee and colleagues. Furthermore, the fact that suicidality did not differ significantly between patients with definite cluster headache and those with probable cluster headache “prompts clinicians for an increased identification and intensive treatment strategy for probable cluster headache.”
The current study is the first prospective investigation of suicidality in the various phases of cluster headache, according to the investigators. It nevertheless has several limitations. The prevalence of chronic cluster headache was low in the study population, and not all patients presented for follow-up during the period between bouts. In addition, the data were obtained from recall, and consequently may be less accurate than those gained from prospective recording. Finally, Dr. Ji Lee and colleagues did not gather information on personality disorders, insomnia, substance abuse, or addiction, even though these factors can influence suicidality in patients with chronic pain.
The investigators reported no conflicts of interest related to their research. The study was supported by a grant from the Korean Neurological Association.
SOURCE: Ji Lee M et al. Cephalalgia. 2019 Apr 24. doi: 10.1177/0333102419845660.
FROM CEPHALAGIA
Key clinical point: Cluster headache is associated with increased suicidality during attacks and within the active period.
Major finding: Cluster headache attacks increased the risk of active suicidal ideation (odds ratio, 15.55).
Study details: A prospective, multicenter study of 175 patients with cluster headache.
Disclosures: The study was supported by a grant from the Korean Neurological Association.
Source: Ji Lee M et al. Cephalalgia. 2019 Apr 24. doi: 10.1177/0333102419845660.