User login
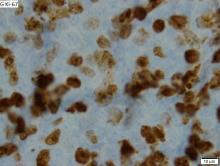
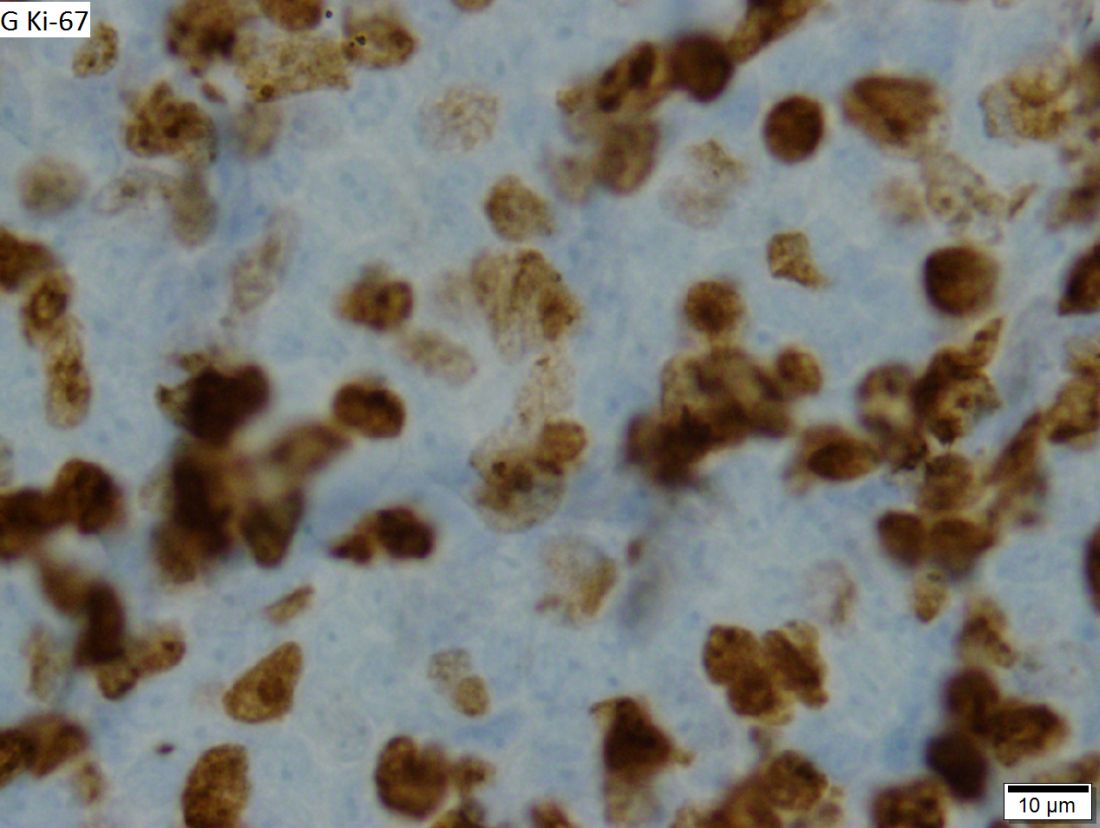
RIT consolidation may be an option for unfit MCL patients
For older, less fit patients with mantle cell lymphoma (MCL) who may not be able to withstand the rigors of autologous stem cell transplants (ASCT), induction chemotherapy followed by radioimmunotherapy (RIT) consolidation with ibritumomab tiuxetan (Zevalin) was associated with good response rates and promising progression-free and overall survival rates, according to results of a phase 2 prospective study.
RIT consolidation improved the complete response rate following first-line therapy from 41% to 91%, reported Wojciech Jurczak, MD, PhD, from the department of hematology at the Uniwersytet Jagiellonski in Krakow, Poland, and colleagues.
In the patients who received RIT following first-line induction, median progression-free survival was 3.3 years, and median overall survival was 6.5 years.
“The achieved responses are durable. Although, several novel agents and targeted therapies alone or in combination are currently being studied and developed in both the upfront and relapsed settings, RIT constitutes a valid and underused option especially in the first-line setting,” they wrote in a study published in Leukemia & Lymphoma.
The investigators enrolled 46 patients with clinical stage III to IV MCL who were either ineligible for, or unwilling to undergo, ASCT. The cohort included 34 patients with newly diagnosed advanced MCL and 12 with chemo-sensitive MCL in first relapse.
Patients were assigned to induction with six cycles of chemotherapy, with or without rituximab. Patients then underwent consolidation with RIT if they had confirmed reductions of the maximal lymph node diameter below 3 cm, their longest spleen measurement was below 15 cm, and bone marrow infiltration was less than 20%.
The chemotherapy regimens included either CVP (cyclophosphamide, vincristine and prednisone), CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), FC (fludarabine and cyclophosphamide), or FCM (FC plus mitoxantrone). Additionally, 27 of the 46 patients received rituximab, which was not considered the standard of care in Poland when the study began in 2005 and was delivered based on availability.
Of the 34 patients who received first-line chemotherapy, 20 received FC or FCM (with or without rituximab), and 14 received CHOP or CVP (with or without rituximab). In this group, 14 patients (41%) had a complete response, and 20 (95%) had a partial response. Of the 12 patients treated after first relapse, two (17%) had a complete response and 10 (83%) had partial response after induction.
RIT consolidation was performed 3-5 weeks after the last chemotherapy cycle. Patients with cytopenias after chemotherapy could wait an additional 3 weeks, during which they would receive a bridging dose of rituximab at the standard 375 mg/m2 dose. The patients received two doses of rituximab 250 mg/m2 administered 7 days then 24 hours prior to intravenous injection of 90Y-labeled ibritumomab tiuxetan. The radiation doses delivered were 0.4 mCi/kg for patients with normal platelet counts and 0.3 mCi/kg for those with platelet counts from 100,000 to 150,000 cells/mm3. The maximum dose was 32.0 mCi.
The longest follow-up was out to slightly more than 8 years.
For the patients who received RIT after first-line induction, the complete response rate was 91%, and the partial response rate was 9%, compared with 41% complete response and 59% partial response after induction. In this group, the median progression-free survival was 3.3 years, and the median overall survival was 6.5 years.
For the patients who received RIT consolidation after first relapse and second chemotherapy regimen, the complete response rate was 75% and the partial response rate was 25%, compared with 17% and 83% at the end of second induction therapy. In this group, the median progression-free survival was 1.8 years (P less than .05, compared with patients treated after first-line responses), and the median overall survival was 2.2 years (P less than .05).
At 8 years of follow-up, 30% of patients who received RIT consolidation following first-line therapy were alive.
Adverse events included cytopenias in the majority of patients (77%), which were grade 1 or 2 in severity in 43% and grade 3 or 4 in 34%. Grade 3 or 4 thrombocytopenia and leukopenia occurred more frequently in patients treated with fludarabine-based regimens, and the thrombocytopenias in these patients lasted longer and required more platelet transfusions than those in CHOP- or CVP-treated patients. Two patients who underwent RIT following FCM induction died from prolonged thrombocytopenia, resulting in hemorrhagic strokes.
Among all patients, 22 patients developed infections following RIT consolidation. Five patients, all of whom had received fludarabine, required hospitalization for the treatment of the infections. There were no infection-related deaths, however.
Five patients developed the myelodysplastic syndrome, with a median onset time of 26 months. Of these patients, four had received fludarabine, and one had undergone a prior ASCT.
The trial was sponsored by Schering AG. Dr. Jurczak reported speakers bureau participation and research funding from multiple companies, not including Schering AG.
SOURCE: Jurczak W et al. Leuk Lymphoma. 2019 Apr 9. doi: 10.1080/10428194.2019.1602261.
For older, less fit patients with mantle cell lymphoma (MCL) who may not be able to withstand the rigors of autologous stem cell transplants (ASCT), induction chemotherapy followed by radioimmunotherapy (RIT) consolidation with ibritumomab tiuxetan (Zevalin) was associated with good response rates and promising progression-free and overall survival rates, according to results of a phase 2 prospective study.
RIT consolidation improved the complete response rate following first-line therapy from 41% to 91%, reported Wojciech Jurczak, MD, PhD, from the department of hematology at the Uniwersytet Jagiellonski in Krakow, Poland, and colleagues.
In the patients who received RIT following first-line induction, median progression-free survival was 3.3 years, and median overall survival was 6.5 years.
“The achieved responses are durable. Although, several novel agents and targeted therapies alone or in combination are currently being studied and developed in both the upfront and relapsed settings, RIT constitutes a valid and underused option especially in the first-line setting,” they wrote in a study published in Leukemia & Lymphoma.
The investigators enrolled 46 patients with clinical stage III to IV MCL who were either ineligible for, or unwilling to undergo, ASCT. The cohort included 34 patients with newly diagnosed advanced MCL and 12 with chemo-sensitive MCL in first relapse.
Patients were assigned to induction with six cycles of chemotherapy, with or without rituximab. Patients then underwent consolidation with RIT if they had confirmed reductions of the maximal lymph node diameter below 3 cm, their longest spleen measurement was below 15 cm, and bone marrow infiltration was less than 20%.
The chemotherapy regimens included either CVP (cyclophosphamide, vincristine and prednisone), CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), FC (fludarabine and cyclophosphamide), or FCM (FC plus mitoxantrone). Additionally, 27 of the 46 patients received rituximab, which was not considered the standard of care in Poland when the study began in 2005 and was delivered based on availability.
Of the 34 patients who received first-line chemotherapy, 20 received FC or FCM (with or without rituximab), and 14 received CHOP or CVP (with or without rituximab). In this group, 14 patients (41%) had a complete response, and 20 (95%) had a partial response. Of the 12 patients treated after first relapse, two (17%) had a complete response and 10 (83%) had partial response after induction.
RIT consolidation was performed 3-5 weeks after the last chemotherapy cycle. Patients with cytopenias after chemotherapy could wait an additional 3 weeks, during which they would receive a bridging dose of rituximab at the standard 375 mg/m2 dose. The patients received two doses of rituximab 250 mg/m2 administered 7 days then 24 hours prior to intravenous injection of 90Y-labeled ibritumomab tiuxetan. The radiation doses delivered were 0.4 mCi/kg for patients with normal platelet counts and 0.3 mCi/kg for those with platelet counts from 100,000 to 150,000 cells/mm3. The maximum dose was 32.0 mCi.
The longest follow-up was out to slightly more than 8 years.
For the patients who received RIT after first-line induction, the complete response rate was 91%, and the partial response rate was 9%, compared with 41% complete response and 59% partial response after induction. In this group, the median progression-free survival was 3.3 years, and the median overall survival was 6.5 years.
For the patients who received RIT consolidation after first relapse and second chemotherapy regimen, the complete response rate was 75% and the partial response rate was 25%, compared with 17% and 83% at the end of second induction therapy. In this group, the median progression-free survival was 1.8 years (P less than .05, compared with patients treated after first-line responses), and the median overall survival was 2.2 years (P less than .05).
At 8 years of follow-up, 30% of patients who received RIT consolidation following first-line therapy were alive.
Adverse events included cytopenias in the majority of patients (77%), which were grade 1 or 2 in severity in 43% and grade 3 or 4 in 34%. Grade 3 or 4 thrombocytopenia and leukopenia occurred more frequently in patients treated with fludarabine-based regimens, and the thrombocytopenias in these patients lasted longer and required more platelet transfusions than those in CHOP- or CVP-treated patients. Two patients who underwent RIT following FCM induction died from prolonged thrombocytopenia, resulting in hemorrhagic strokes.
Among all patients, 22 patients developed infections following RIT consolidation. Five patients, all of whom had received fludarabine, required hospitalization for the treatment of the infections. There were no infection-related deaths, however.
Five patients developed the myelodysplastic syndrome, with a median onset time of 26 months. Of these patients, four had received fludarabine, and one had undergone a prior ASCT.
The trial was sponsored by Schering AG. Dr. Jurczak reported speakers bureau participation and research funding from multiple companies, not including Schering AG.
SOURCE: Jurczak W et al. Leuk Lymphoma. 2019 Apr 9. doi: 10.1080/10428194.2019.1602261.
For older, less fit patients with mantle cell lymphoma (MCL) who may not be able to withstand the rigors of autologous stem cell transplants (ASCT), induction chemotherapy followed by radioimmunotherapy (RIT) consolidation with ibritumomab tiuxetan (Zevalin) was associated with good response rates and promising progression-free and overall survival rates, according to results of a phase 2 prospective study.
RIT consolidation improved the complete response rate following first-line therapy from 41% to 91%, reported Wojciech Jurczak, MD, PhD, from the department of hematology at the Uniwersytet Jagiellonski in Krakow, Poland, and colleagues.
In the patients who received RIT following first-line induction, median progression-free survival was 3.3 years, and median overall survival was 6.5 years.
“The achieved responses are durable. Although, several novel agents and targeted therapies alone or in combination are currently being studied and developed in both the upfront and relapsed settings, RIT constitutes a valid and underused option especially in the first-line setting,” they wrote in a study published in Leukemia & Lymphoma.
The investigators enrolled 46 patients with clinical stage III to IV MCL who were either ineligible for, or unwilling to undergo, ASCT. The cohort included 34 patients with newly diagnosed advanced MCL and 12 with chemo-sensitive MCL in first relapse.
Patients were assigned to induction with six cycles of chemotherapy, with or without rituximab. Patients then underwent consolidation with RIT if they had confirmed reductions of the maximal lymph node diameter below 3 cm, their longest spleen measurement was below 15 cm, and bone marrow infiltration was less than 20%.
The chemotherapy regimens included either CVP (cyclophosphamide, vincristine and prednisone), CHOP (cyclophosphamide, doxorubicin, vincristine, and prednisone), FC (fludarabine and cyclophosphamide), or FCM (FC plus mitoxantrone). Additionally, 27 of the 46 patients received rituximab, which was not considered the standard of care in Poland when the study began in 2005 and was delivered based on availability.
Of the 34 patients who received first-line chemotherapy, 20 received FC or FCM (with or without rituximab), and 14 received CHOP or CVP (with or without rituximab). In this group, 14 patients (41%) had a complete response, and 20 (95%) had a partial response. Of the 12 patients treated after first relapse, two (17%) had a complete response and 10 (83%) had partial response after induction.
RIT consolidation was performed 3-5 weeks after the last chemotherapy cycle. Patients with cytopenias after chemotherapy could wait an additional 3 weeks, during which they would receive a bridging dose of rituximab at the standard 375 mg/m2 dose. The patients received two doses of rituximab 250 mg/m2 administered 7 days then 24 hours prior to intravenous injection of 90Y-labeled ibritumomab tiuxetan. The radiation doses delivered were 0.4 mCi/kg for patients with normal platelet counts and 0.3 mCi/kg for those with platelet counts from 100,000 to 150,000 cells/mm3. The maximum dose was 32.0 mCi.
The longest follow-up was out to slightly more than 8 years.
For the patients who received RIT after first-line induction, the complete response rate was 91%, and the partial response rate was 9%, compared with 41% complete response and 59% partial response after induction. In this group, the median progression-free survival was 3.3 years, and the median overall survival was 6.5 years.
For the patients who received RIT consolidation after first relapse and second chemotherapy regimen, the complete response rate was 75% and the partial response rate was 25%, compared with 17% and 83% at the end of second induction therapy. In this group, the median progression-free survival was 1.8 years (P less than .05, compared with patients treated after first-line responses), and the median overall survival was 2.2 years (P less than .05).
At 8 years of follow-up, 30% of patients who received RIT consolidation following first-line therapy were alive.
Adverse events included cytopenias in the majority of patients (77%), which were grade 1 or 2 in severity in 43% and grade 3 or 4 in 34%. Grade 3 or 4 thrombocytopenia and leukopenia occurred more frequently in patients treated with fludarabine-based regimens, and the thrombocytopenias in these patients lasted longer and required more platelet transfusions than those in CHOP- or CVP-treated patients. Two patients who underwent RIT following FCM induction died from prolonged thrombocytopenia, resulting in hemorrhagic strokes.
Among all patients, 22 patients developed infections following RIT consolidation. Five patients, all of whom had received fludarabine, required hospitalization for the treatment of the infections. There were no infection-related deaths, however.
Five patients developed the myelodysplastic syndrome, with a median onset time of 26 months. Of these patients, four had received fludarabine, and one had undergone a prior ASCT.
The trial was sponsored by Schering AG. Dr. Jurczak reported speakers bureau participation and research funding from multiple companies, not including Schering AG.
SOURCE: Jurczak W et al. Leuk Lymphoma. 2019 Apr 9. doi: 10.1080/10428194.2019.1602261.
FROM LEUKEMIA & LYMPHOMA
Low LDL cholesterol may increase women’s risk of hemorrhagic stroke
, according to research published in Neurology.
“Women with very low LDL cholesterol or low triglycerides should be monitored by their doctors for other stroke risk factors that can be modified, like high blood pressure and smoking, in order to reduce their risk of hemorrhagic stroke,” said Pamela M. Rist, ScD, instructor in epidemiology at Harvard Medical School, Boston. “Additional research is needed to determine how to lower the risk of hemorrhagic stroke in women with very low LDL and low triglycerides.”
Several meta-analyses have indicated that LDL cholesterol levels are inversely associated with the risk of hemorrhagic stroke. Because lipid-lowering treatments are used to prevent cardiovascular disease, this potential association has implications for clinical practice. Most of the studies included in these meta-analyses had low numbers of events among women, which prevented researchers from stratifying their results by sex. Because women are at greater risk of stroke than men, Dr. Rist and her colleagues sought to evaluate the association between lipid levels and risk of hemorrhagic stroke.
An analysis of the Women’s Health Study
The investigators examined data from the Women’s Health Study, a randomized, double-blind, placebo-controlled trial of low-dose aspirin and vitamin E for the primary prevention of cardiovascular disease and cancer among female American health professionals aged 45 years or older. The study ended in March 2004, but follow-up is ongoing. At regular intervals, the women complete a questionnaire about disease outcomes, including stroke. Some participants agreed to provide a fasting venous blood sample before randomization. With the subjects’ permission, a committee of physicians examined medical records for women who reported a stroke on a follow-up questionnaire.
Dr. Rist and her colleagues analyzed 27,937 samples for levels of LDL cholesterol, HDL cholesterol, total cholesterol, and triglycerides. They assigned each sample to one of five cholesterol level categories that were based on Adult Treatment Panel III guidelines. Cox proportional hazards models enabled the researchers to calculate the hazard ratio of incident hemorrhagic stroke events. They adjusted their results for covariates such as age, smoking status, menopausal status, body mass index, and alcohol consumption.
A U-shaped association
Women in the lowest category of LDL cholesterol level (less than 70 mg/dL) were younger, less likely to have a history of hypertension, and less likely to use cholesterol-lowering drugs than women in the reference group (100.0-129.9 mg/dL). Women with the lowest LDL cholesterol level were more likely to consume alcohol, have a normal weight, engage in physical activity, and be premenopausal than women in the reference group. The investigators confirmed 137 incident hemorrhagic stroke events during a mean 19.3 years of follow-up.
After data adjustment, the researchers found that women with the lowest level of LDL cholesterol had 2.17 times the risk of hemorrhagic stroke, compared with participants in the reference group. They found a trend toward increased risk among women with an LDL cholesterol level of 160 mg/dL or higher, but the result was not statistically significant. The highest risk for intracerebral hemorrhage (ICH) was among women with an LDL cholesterol level of less than 70 mg/dL (relative risk, 2.32), followed by women with a level of 160 mg/dL or higher (RR, 1.71).
In addition, after multivariable adjustment, women in the lowest quartile of triglycerides (less than or equal to 74 mg/dL for fasting and less than or equal to 85 mg/dL for nonfasting) had a significantly increased risk of hemorrhagic stroke, compared with women in the highest quartile (RR, 2.00). Low triglyceride levels were associated with an increased risk of subarachnoid hemorrhage, but not with an increased risk of ICH. Neither HDL cholesterol nor total cholesterol was associated with risk of hemorrhagic stroke, the researchers wrote.
Mechanism of increased risk unclear
The researchers do not yet know how low triglyceride and LDL cholesterol levels increase the risk of hemorrhagic stroke. One hypothesis is that low cholesterol promotes necrosis of the arterial medial layer’s smooth muscle cells. This impaired endothelium might be more susceptible to microaneurysms, which are common in patients with ICH, said the researchers.
The prospective design and the large sample size were two of the study’s strengths, but the study had important weaknesses as well, the researchers wrote. For example, few women were premenopausal at baseline, so the investigators could not evaluate whether menopausal status modifies the association between lipid levels and risk of hemorrhagic stroke. In addition, lipid levels were measured only at baseline, which prevented an analysis of whether change in lipid levels over time modifies the risk of hemorrhagic stroke.
Dr. Rist reported receiving a grant from the National Institutes of Health.
SOURCE: Rist PM et al. Neurology. 2019 April 10. doi: 10.1212/WNL.0000000000007454.
, according to research published in Neurology.
“Women with very low LDL cholesterol or low triglycerides should be monitored by their doctors for other stroke risk factors that can be modified, like high blood pressure and smoking, in order to reduce their risk of hemorrhagic stroke,” said Pamela M. Rist, ScD, instructor in epidemiology at Harvard Medical School, Boston. “Additional research is needed to determine how to lower the risk of hemorrhagic stroke in women with very low LDL and low triglycerides.”
Several meta-analyses have indicated that LDL cholesterol levels are inversely associated with the risk of hemorrhagic stroke. Because lipid-lowering treatments are used to prevent cardiovascular disease, this potential association has implications for clinical practice. Most of the studies included in these meta-analyses had low numbers of events among women, which prevented researchers from stratifying their results by sex. Because women are at greater risk of stroke than men, Dr. Rist and her colleagues sought to evaluate the association between lipid levels and risk of hemorrhagic stroke.
An analysis of the Women’s Health Study
The investigators examined data from the Women’s Health Study, a randomized, double-blind, placebo-controlled trial of low-dose aspirin and vitamin E for the primary prevention of cardiovascular disease and cancer among female American health professionals aged 45 years or older. The study ended in March 2004, but follow-up is ongoing. At regular intervals, the women complete a questionnaire about disease outcomes, including stroke. Some participants agreed to provide a fasting venous blood sample before randomization. With the subjects’ permission, a committee of physicians examined medical records for women who reported a stroke on a follow-up questionnaire.
Dr. Rist and her colleagues analyzed 27,937 samples for levels of LDL cholesterol, HDL cholesterol, total cholesterol, and triglycerides. They assigned each sample to one of five cholesterol level categories that were based on Adult Treatment Panel III guidelines. Cox proportional hazards models enabled the researchers to calculate the hazard ratio of incident hemorrhagic stroke events. They adjusted their results for covariates such as age, smoking status, menopausal status, body mass index, and alcohol consumption.
A U-shaped association
Women in the lowest category of LDL cholesterol level (less than 70 mg/dL) were younger, less likely to have a history of hypertension, and less likely to use cholesterol-lowering drugs than women in the reference group (100.0-129.9 mg/dL). Women with the lowest LDL cholesterol level were more likely to consume alcohol, have a normal weight, engage in physical activity, and be premenopausal than women in the reference group. The investigators confirmed 137 incident hemorrhagic stroke events during a mean 19.3 years of follow-up.
After data adjustment, the researchers found that women with the lowest level of LDL cholesterol had 2.17 times the risk of hemorrhagic stroke, compared with participants in the reference group. They found a trend toward increased risk among women with an LDL cholesterol level of 160 mg/dL or higher, but the result was not statistically significant. The highest risk for intracerebral hemorrhage (ICH) was among women with an LDL cholesterol level of less than 70 mg/dL (relative risk, 2.32), followed by women with a level of 160 mg/dL or higher (RR, 1.71).
In addition, after multivariable adjustment, women in the lowest quartile of triglycerides (less than or equal to 74 mg/dL for fasting and less than or equal to 85 mg/dL for nonfasting) had a significantly increased risk of hemorrhagic stroke, compared with women in the highest quartile (RR, 2.00). Low triglyceride levels were associated with an increased risk of subarachnoid hemorrhage, but not with an increased risk of ICH. Neither HDL cholesterol nor total cholesterol was associated with risk of hemorrhagic stroke, the researchers wrote.
Mechanism of increased risk unclear
The researchers do not yet know how low triglyceride and LDL cholesterol levels increase the risk of hemorrhagic stroke. One hypothesis is that low cholesterol promotes necrosis of the arterial medial layer’s smooth muscle cells. This impaired endothelium might be more susceptible to microaneurysms, which are common in patients with ICH, said the researchers.
The prospective design and the large sample size were two of the study’s strengths, but the study had important weaknesses as well, the researchers wrote. For example, few women were premenopausal at baseline, so the investigators could not evaluate whether menopausal status modifies the association between lipid levels and risk of hemorrhagic stroke. In addition, lipid levels were measured only at baseline, which prevented an analysis of whether change in lipid levels over time modifies the risk of hemorrhagic stroke.
Dr. Rist reported receiving a grant from the National Institutes of Health.
SOURCE: Rist PM et al. Neurology. 2019 April 10. doi: 10.1212/WNL.0000000000007454.
, according to research published in Neurology.
“Women with very low LDL cholesterol or low triglycerides should be monitored by their doctors for other stroke risk factors that can be modified, like high blood pressure and smoking, in order to reduce their risk of hemorrhagic stroke,” said Pamela M. Rist, ScD, instructor in epidemiology at Harvard Medical School, Boston. “Additional research is needed to determine how to lower the risk of hemorrhagic stroke in women with very low LDL and low triglycerides.”
Several meta-analyses have indicated that LDL cholesterol levels are inversely associated with the risk of hemorrhagic stroke. Because lipid-lowering treatments are used to prevent cardiovascular disease, this potential association has implications for clinical practice. Most of the studies included in these meta-analyses had low numbers of events among women, which prevented researchers from stratifying their results by sex. Because women are at greater risk of stroke than men, Dr. Rist and her colleagues sought to evaluate the association between lipid levels and risk of hemorrhagic stroke.
An analysis of the Women’s Health Study
The investigators examined data from the Women’s Health Study, a randomized, double-blind, placebo-controlled trial of low-dose aspirin and vitamin E for the primary prevention of cardiovascular disease and cancer among female American health professionals aged 45 years or older. The study ended in March 2004, but follow-up is ongoing. At regular intervals, the women complete a questionnaire about disease outcomes, including stroke. Some participants agreed to provide a fasting venous blood sample before randomization. With the subjects’ permission, a committee of physicians examined medical records for women who reported a stroke on a follow-up questionnaire.
Dr. Rist and her colleagues analyzed 27,937 samples for levels of LDL cholesterol, HDL cholesterol, total cholesterol, and triglycerides. They assigned each sample to one of five cholesterol level categories that were based on Adult Treatment Panel III guidelines. Cox proportional hazards models enabled the researchers to calculate the hazard ratio of incident hemorrhagic stroke events. They adjusted their results for covariates such as age, smoking status, menopausal status, body mass index, and alcohol consumption.
A U-shaped association
Women in the lowest category of LDL cholesterol level (less than 70 mg/dL) were younger, less likely to have a history of hypertension, and less likely to use cholesterol-lowering drugs than women in the reference group (100.0-129.9 mg/dL). Women with the lowest LDL cholesterol level were more likely to consume alcohol, have a normal weight, engage in physical activity, and be premenopausal than women in the reference group. The investigators confirmed 137 incident hemorrhagic stroke events during a mean 19.3 years of follow-up.
After data adjustment, the researchers found that women with the lowest level of LDL cholesterol had 2.17 times the risk of hemorrhagic stroke, compared with participants in the reference group. They found a trend toward increased risk among women with an LDL cholesterol level of 160 mg/dL or higher, but the result was not statistically significant. The highest risk for intracerebral hemorrhage (ICH) was among women with an LDL cholesterol level of less than 70 mg/dL (relative risk, 2.32), followed by women with a level of 160 mg/dL or higher (RR, 1.71).
In addition, after multivariable adjustment, women in the lowest quartile of triglycerides (less than or equal to 74 mg/dL for fasting and less than or equal to 85 mg/dL for nonfasting) had a significantly increased risk of hemorrhagic stroke, compared with women in the highest quartile (RR, 2.00). Low triglyceride levels were associated with an increased risk of subarachnoid hemorrhage, but not with an increased risk of ICH. Neither HDL cholesterol nor total cholesterol was associated with risk of hemorrhagic stroke, the researchers wrote.
Mechanism of increased risk unclear
The researchers do not yet know how low triglyceride and LDL cholesterol levels increase the risk of hemorrhagic stroke. One hypothesis is that low cholesterol promotes necrosis of the arterial medial layer’s smooth muscle cells. This impaired endothelium might be more susceptible to microaneurysms, which are common in patients with ICH, said the researchers.
The prospective design and the large sample size were two of the study’s strengths, but the study had important weaknesses as well, the researchers wrote. For example, few women were premenopausal at baseline, so the investigators could not evaluate whether menopausal status modifies the association between lipid levels and risk of hemorrhagic stroke. In addition, lipid levels were measured only at baseline, which prevented an analysis of whether change in lipid levels over time modifies the risk of hemorrhagic stroke.
Dr. Rist reported receiving a grant from the National Institutes of Health.
SOURCE: Rist PM et al. Neurology. 2019 April 10. doi: 10.1212/WNL.0000000000007454.
FROM NEUROLOGY
BACE-1 inhibition worsens cognition in patients with prodromal Alzheimer’s disease
More bad news for Alzheimer’s research. Two more BACE inhibitors fall far short of the finish line.
Declining cognitive scores, falls, suicidal ideation, and liver enzyme abnormalities were all seen in clinical trials.
The news doesn’t bode well for the therapeutic target of BACE (beta-site APP cleaving enzyme) inhibition. BACE is one of the enzymes that trims the amyloid precursor protein (APP). Inhibiting it does reduce the amount of toxic amyloid-beta in cerebrospinal fluid, and amyloid plaque in the brain. But none of these molecules has shown clinical benefit in dementia patients, whether their disease is mild, or moderate or – now – prodromal. And it is now apparent that inhibiting BACE also produces serious off-target problems.
“BACE-1 inhibition certainly seemed to have a sound rationale assuming the basis for amyloid’s role in Alzheimer’s disease pathogenesis is a gain of toxicity,” Richard J. Caselli, MD, of the Mayo Clinic, Rochester, Minn., said in an interview. “That APP is important for Alzheimer’s pathogenesis still seems clear but whether amyloid-beta toxicity is the driving force is no longer clear. Further, interruption of the APP system disrupts more than amyloid-beta peptide, possibly explaining the adverse cognitive effects of BACE-1 inhibition shown exhibited now by three different BACE-1 inhibitors.”
Verubecestat
Researchers got their first dose of bad news regarding verubecestat at the 2017 Clinical Trials in Alzheimer’s Disease meeting. There, Michael F. Egan, MD, Merck’s associate vice president of clinical neuroscience, discussed the molecule’s failure to slow cognitive decline in patients with mild to moderate Alzheimer’s disease. There was plenty of biomarker evidence that the drug did block amyloid-beta production, but there also was a plethora of concerning adverse events, Dr. Egan said in an interview.
However, verubecestat still was being pursued in patients with prodromal Alzheimer’s disease. In February, Merck stopped the trial after a futility analysis and announced that the company was terminating studies of verubecestat in that population as well. In the April 11 issue of the New England Journal of Medicine, Dr. Egan and his colleagues report the full extent of verubecestat’s failure in prodromal patients, and the accompanying adverse events.
At the time of termination, 1,454 patients had been enrolled. Of these, 485 received 12 mg/day, 484 received 40 mg/day, and 485 received placebo. About half of each group completed 104 weeks of treatment in the study, which was designed to extend up to 5 years.
The primary outcome was change in the Clinical Dementia Rating Scale–Sum of Boxes score (CDR-SB). Seven secondary outcomes examined other cognitive and functional end points, along with changes in hippocampal volume on MRI and amyloid burden as determined in PET imaging.
Not only did verubecestat fail to slow cognitive decline, it appeared to exacerbate it. The mean change on the CDR-SB was 1.65 in the 12-mg group, 2.02 in the 40-mg group, and 1.58 in the placebo group, favoring placebo.
“In an exploratory analysis according to time point, scores on the CDR-SB were also higher [signifying more impairment of cognition and daily functioning] in the 40-mg group than in the placebo group at 13, 26, and 52 weeks ... suggesting but not confirming the possibility of worse performance at these earlier time points in the high-dose verubecestat group,” the investigators said.
Verubecestat also was associated with more conversions to Alzheimer’s disease. Per 100 patient-years, the Alzheimer’s disease event rates were 24.5 in the 12-mg group, 25.5 in the 40-mg group, and 19.3 in the placebo group. Compared with placebo, those taking 12-mg doses were 30% more likely to develop Alzheimer’s disease and those taking 40-mg doses were 38% more likely. The findings suggest that “verubecestat may have accelerated the progression to diagnosis of dementia due to Alzheimer’s disease,” the investigators said.
The negative impact of verubecestat was apparent quite early in the study. “In exploratory analyses, both dose levels of verubecestat were associated with poorer outcomes on the [Composite Cognition Score-3 Domain] and the ADAS-Cog [Alzheimer’s Disease Assessment Scale–Cognitive Subscale] measures of cognition that, relative to placebo, appeared worse at week 13 and did not appear to progress thereafter.”
Results of the secondary end points, including the ADAS-Cog and the Mini-Mental State Exam, also indicated that verubecestat may have worsened cognitive performance.
Imaging outcomes were positive, however. Hippocampal volume was 6,448 mL in the 12-mg group, 6,469 mL in the 40-mg group, and 6,435 mL in the placebo group. Brain amyloid increased in the placebo group, as expected, and decreased in the active groups. The small group of patients who underwent cerebrospinal fluid sampling showed reductions of more than 60% in amyloid-beta and soluble APP-beta associated with verubecestat. These results show that the molecule was indeed hitting its intended target, but that doing so was not clinically beneficial.
Adverse events were more common in the verubecestat groups. These included rash, dermatitis, urticaria, sleep disturbance, weight loss, and cough. Hair coloring changed in 2.5% of patients in the 12-mg group and 5% of the 40-mg group, but in none of the subjects taking placebo.
Patients taking verubecestat were more likely to sustain falls and injuries and to express suicidal ideation.
The results of this trial differ from the study of verubecestat in mild to moderate Alzheimer’s disease, the investigators noted. Those patients did not decline cognitively as did those with prodromal disease.
“Patients at an earlier stage of the disease may be more sensitive to the effects of substantial BACE-1 inhibition, perhaps because of a role of BACE-1 in normal synaptic function. It is also possible that the effects of verubecestat are due to inhibition of BACE-2,” they said.
Atabecestat
In a research letter in the same issue of the New England Journal of Medicine (2019 Apr 11;380:1483-5), David Henley, MD, senior director of Janssen’s Alzheimer’s clinical development core, released similarly negative results from an interim analysis of EARLY (Efficacy and Safety Study of Atabecestat in Participants Who Are Asymptomatic at Risk for Developing Alzheimer’s Dementia) trial, a randomized study of the BACE-1 inhibitor candidate, atabecestat.
The phase 2 trial enrolled 557 patients with prodromal Alzheimer’s disease. The primary cognitive end point was change from baseline in the Preclinical Alzheimer’s Cognitive Composite (PACC) score.
This trial was discontinued in May 2018 because of liver-related adverse events, although safety follow-up continues. The research letter did not disclose details of the hepatic events, but a company press release from May 2018 referred to them in a general sense.
“Elevations of liver enzymes, which were serious in nature, have been observed in some study participants who received the Janssen BACE inhibitor, atabecestat. After a thorough evaluation of all available liver safety data from our studies, Janssen has concluded that the benefit-risk ratio is no longer favorable to continue development of atabecestat for people who have late-onset preclinical stage Alzheimer’s disease.”
Patients in EARLY were randomized to 5 mg, 25 mg, or placebo. As in the verubecestat trial, those randomized to placebo did better. The mean changes from baseline in the PACC score were −1.44 in the 25-mg group, −0.58 in the 5-mg group, and −0.32 in the placebo group.
“At month 6, the difference between the 25-mg group and the placebo group was −1.12 and the difference between the 5-mg group and the placebo group was −0.26, favoring placebo over the higher dose,” the authors said.
This theme reemerged in a secondary end point, the Repeatable Battery for the Assessment of Neuropsychological Status. The 25-mg group declined 3.58 points more than placebo, and the 5-mg group, 1.43 points more.
Adverse events were more common in the active groups and included depression, effects on sleep and dreams, and anxiety.
“The differences in cognitive performance between the groups are of uncertain clinical significance; however, given similar findings favoring placebo over BACE-1 inhibitors in other trial, we are communicating this potential signal of worsening cognitive function in the treated groups,” Dr. Henley said.
SOURCE: Egan MF et al. N Engl J Med. 2019 Apr 11;380:1408-20.
“Some trials fail because the experimental treatment proves to be no different than a control or standard intervention,” David Knopman, MD, wrote in an accompanying editorial (N Engl J Med. 2019 Apr 11;380:1476-8). “Others fail because of unacceptable side effects. In this issue of the Journal, an article by Egan et al. and a letter to the editor by Henley et al. (N Engl J Med. 2019 Apr 11;380:1483-5) describe a third reason for failure – a treatment worsens the target symptoms.
Certainly, beta-site amyloid precursor protein-cleaving enzyme 1 (BACE-1) inhibition makes sense when viewed in the light of the current understanding of Alzheimer’s disease neuropathology. The amyloid cascade hypothesis holds that toxic amyloid-beta fragments accumulate in the brain, form dense neuritic plaques, and lead to neuronal death and cognitive decline.
“The model is rooted in the inseparability of Alzheimer’s disease from abundant amyloid-beta pathologic features,” Dr. Knopman wrote. But, “Over the past 2 decades, the amyloid-beta–lowering strategy has been put to the test in trials of antiamyloid antibodies, none of which have been successful.”
Therefore, hitting amyloid at the source – the transmembrane cleavage domain – seemed important and, potentially, effective. But three BACE inhibitors (verubecestat, atabecestat, and lanabecestat) have shown similarly negative cognitive effects. “Together, these results suggest that preserved BACE-1 activity may be critical to normal synaptic functions. These observations place a limitation on how amyloid-beta lowering can be accomplished.”
It is possible that decreasing the level of BACE inhibition might ameliorate off-target effects and neuronal compromise but still be enough to reduce the generation of toxic amyloid-beta fragments, Dr. Knopman said. But, “Adjustments in the dose to a narrow window of BACE-1 inhibition would be difficult to accomplish in a clinical trial until there are peripheral biomarkers that reflect the activity of the agent in the brain.”
Thus far, most of the studied antiamyloid drugs have indeed reduced amyloid-beta levels, but none of those reductions affected cognition. A rethinking of amyloid-beta’s place in dementia progression may be in order.
“The dissociation between amyloid-beta lowering and cognitive benefits with both BACE-1 inhibition and antiamyloid antibody therapy is troubling. To be blunt, amyloid-beta lowering seems to be an ineffective approach, and it is time to focus on other targets to move therapeutics for Alzheimer’s disease forward.”
Dr. Knopman is a clinical neurologist and Alzheimer’s researcher at the Mayo Clinic, Rochester, Minn.
“Some trials fail because the experimental treatment proves to be no different than a control or standard intervention,” David Knopman, MD, wrote in an accompanying editorial (N Engl J Med. 2019 Apr 11;380:1476-8). “Others fail because of unacceptable side effects. In this issue of the Journal, an article by Egan et al. and a letter to the editor by Henley et al. (N Engl J Med. 2019 Apr 11;380:1483-5) describe a third reason for failure – a treatment worsens the target symptoms.
Certainly, beta-site amyloid precursor protein-cleaving enzyme 1 (BACE-1) inhibition makes sense when viewed in the light of the current understanding of Alzheimer’s disease neuropathology. The amyloid cascade hypothesis holds that toxic amyloid-beta fragments accumulate in the brain, form dense neuritic plaques, and lead to neuronal death and cognitive decline.
“The model is rooted in the inseparability of Alzheimer’s disease from abundant amyloid-beta pathologic features,” Dr. Knopman wrote. But, “Over the past 2 decades, the amyloid-beta–lowering strategy has been put to the test in trials of antiamyloid antibodies, none of which have been successful.”
Therefore, hitting amyloid at the source – the transmembrane cleavage domain – seemed important and, potentially, effective. But three BACE inhibitors (verubecestat, atabecestat, and lanabecestat) have shown similarly negative cognitive effects. “Together, these results suggest that preserved BACE-1 activity may be critical to normal synaptic functions. These observations place a limitation on how amyloid-beta lowering can be accomplished.”
It is possible that decreasing the level of BACE inhibition might ameliorate off-target effects and neuronal compromise but still be enough to reduce the generation of toxic amyloid-beta fragments, Dr. Knopman said. But, “Adjustments in the dose to a narrow window of BACE-1 inhibition would be difficult to accomplish in a clinical trial until there are peripheral biomarkers that reflect the activity of the agent in the brain.”
Thus far, most of the studied antiamyloid drugs have indeed reduced amyloid-beta levels, but none of those reductions affected cognition. A rethinking of amyloid-beta’s place in dementia progression may be in order.
“The dissociation between amyloid-beta lowering and cognitive benefits with both BACE-1 inhibition and antiamyloid antibody therapy is troubling. To be blunt, amyloid-beta lowering seems to be an ineffective approach, and it is time to focus on other targets to move therapeutics for Alzheimer’s disease forward.”
Dr. Knopman is a clinical neurologist and Alzheimer’s researcher at the Mayo Clinic, Rochester, Minn.
“Some trials fail because the experimental treatment proves to be no different than a control or standard intervention,” David Knopman, MD, wrote in an accompanying editorial (N Engl J Med. 2019 Apr 11;380:1476-8). “Others fail because of unacceptable side effects. In this issue of the Journal, an article by Egan et al. and a letter to the editor by Henley et al. (N Engl J Med. 2019 Apr 11;380:1483-5) describe a third reason for failure – a treatment worsens the target symptoms.
Certainly, beta-site amyloid precursor protein-cleaving enzyme 1 (BACE-1) inhibition makes sense when viewed in the light of the current understanding of Alzheimer’s disease neuropathology. The amyloid cascade hypothesis holds that toxic amyloid-beta fragments accumulate in the brain, form dense neuritic plaques, and lead to neuronal death and cognitive decline.
“The model is rooted in the inseparability of Alzheimer’s disease from abundant amyloid-beta pathologic features,” Dr. Knopman wrote. But, “Over the past 2 decades, the amyloid-beta–lowering strategy has been put to the test in trials of antiamyloid antibodies, none of which have been successful.”
Therefore, hitting amyloid at the source – the transmembrane cleavage domain – seemed important and, potentially, effective. But three BACE inhibitors (verubecestat, atabecestat, and lanabecestat) have shown similarly negative cognitive effects. “Together, these results suggest that preserved BACE-1 activity may be critical to normal synaptic functions. These observations place a limitation on how amyloid-beta lowering can be accomplished.”
It is possible that decreasing the level of BACE inhibition might ameliorate off-target effects and neuronal compromise but still be enough to reduce the generation of toxic amyloid-beta fragments, Dr. Knopman said. But, “Adjustments in the dose to a narrow window of BACE-1 inhibition would be difficult to accomplish in a clinical trial until there are peripheral biomarkers that reflect the activity of the agent in the brain.”
Thus far, most of the studied antiamyloid drugs have indeed reduced amyloid-beta levels, but none of those reductions affected cognition. A rethinking of amyloid-beta’s place in dementia progression may be in order.
“The dissociation between amyloid-beta lowering and cognitive benefits with both BACE-1 inhibition and antiamyloid antibody therapy is troubling. To be blunt, amyloid-beta lowering seems to be an ineffective approach, and it is time to focus on other targets to move therapeutics for Alzheimer’s disease forward.”
Dr. Knopman is a clinical neurologist and Alzheimer’s researcher at the Mayo Clinic, Rochester, Minn.
More bad news for Alzheimer’s research. Two more BACE inhibitors fall far short of the finish line.
Declining cognitive scores, falls, suicidal ideation, and liver enzyme abnormalities were all seen in clinical trials.
The news doesn’t bode well for the therapeutic target of BACE (beta-site APP cleaving enzyme) inhibition. BACE is one of the enzymes that trims the amyloid precursor protein (APP). Inhibiting it does reduce the amount of toxic amyloid-beta in cerebrospinal fluid, and amyloid plaque in the brain. But none of these molecules has shown clinical benefit in dementia patients, whether their disease is mild, or moderate or – now – prodromal. And it is now apparent that inhibiting BACE also produces serious off-target problems.
“BACE-1 inhibition certainly seemed to have a sound rationale assuming the basis for amyloid’s role in Alzheimer’s disease pathogenesis is a gain of toxicity,” Richard J. Caselli, MD, of the Mayo Clinic, Rochester, Minn., said in an interview. “That APP is important for Alzheimer’s pathogenesis still seems clear but whether amyloid-beta toxicity is the driving force is no longer clear. Further, interruption of the APP system disrupts more than amyloid-beta peptide, possibly explaining the adverse cognitive effects of BACE-1 inhibition shown exhibited now by three different BACE-1 inhibitors.”
Verubecestat
Researchers got their first dose of bad news regarding verubecestat at the 2017 Clinical Trials in Alzheimer’s Disease meeting. There, Michael F. Egan, MD, Merck’s associate vice president of clinical neuroscience, discussed the molecule’s failure to slow cognitive decline in patients with mild to moderate Alzheimer’s disease. There was plenty of biomarker evidence that the drug did block amyloid-beta production, but there also was a plethora of concerning adverse events, Dr. Egan said in an interview.
However, verubecestat still was being pursued in patients with prodromal Alzheimer’s disease. In February, Merck stopped the trial after a futility analysis and announced that the company was terminating studies of verubecestat in that population as well. In the April 11 issue of the New England Journal of Medicine, Dr. Egan and his colleagues report the full extent of verubecestat’s failure in prodromal patients, and the accompanying adverse events.
At the time of termination, 1,454 patients had been enrolled. Of these, 485 received 12 mg/day, 484 received 40 mg/day, and 485 received placebo. About half of each group completed 104 weeks of treatment in the study, which was designed to extend up to 5 years.
The primary outcome was change in the Clinical Dementia Rating Scale–Sum of Boxes score (CDR-SB). Seven secondary outcomes examined other cognitive and functional end points, along with changes in hippocampal volume on MRI and amyloid burden as determined in PET imaging.
Not only did verubecestat fail to slow cognitive decline, it appeared to exacerbate it. The mean change on the CDR-SB was 1.65 in the 12-mg group, 2.02 in the 40-mg group, and 1.58 in the placebo group, favoring placebo.
“In an exploratory analysis according to time point, scores on the CDR-SB were also higher [signifying more impairment of cognition and daily functioning] in the 40-mg group than in the placebo group at 13, 26, and 52 weeks ... suggesting but not confirming the possibility of worse performance at these earlier time points in the high-dose verubecestat group,” the investigators said.
Verubecestat also was associated with more conversions to Alzheimer’s disease. Per 100 patient-years, the Alzheimer’s disease event rates were 24.5 in the 12-mg group, 25.5 in the 40-mg group, and 19.3 in the placebo group. Compared with placebo, those taking 12-mg doses were 30% more likely to develop Alzheimer’s disease and those taking 40-mg doses were 38% more likely. The findings suggest that “verubecestat may have accelerated the progression to diagnosis of dementia due to Alzheimer’s disease,” the investigators said.
The negative impact of verubecestat was apparent quite early in the study. “In exploratory analyses, both dose levels of verubecestat were associated with poorer outcomes on the [Composite Cognition Score-3 Domain] and the ADAS-Cog [Alzheimer’s Disease Assessment Scale–Cognitive Subscale] measures of cognition that, relative to placebo, appeared worse at week 13 and did not appear to progress thereafter.”
Results of the secondary end points, including the ADAS-Cog and the Mini-Mental State Exam, also indicated that verubecestat may have worsened cognitive performance.
Imaging outcomes were positive, however. Hippocampal volume was 6,448 mL in the 12-mg group, 6,469 mL in the 40-mg group, and 6,435 mL in the placebo group. Brain amyloid increased in the placebo group, as expected, and decreased in the active groups. The small group of patients who underwent cerebrospinal fluid sampling showed reductions of more than 60% in amyloid-beta and soluble APP-beta associated with verubecestat. These results show that the molecule was indeed hitting its intended target, but that doing so was not clinically beneficial.
Adverse events were more common in the verubecestat groups. These included rash, dermatitis, urticaria, sleep disturbance, weight loss, and cough. Hair coloring changed in 2.5% of patients in the 12-mg group and 5% of the 40-mg group, but in none of the subjects taking placebo.
Patients taking verubecestat were more likely to sustain falls and injuries and to express suicidal ideation.
The results of this trial differ from the study of verubecestat in mild to moderate Alzheimer’s disease, the investigators noted. Those patients did not decline cognitively as did those with prodromal disease.
“Patients at an earlier stage of the disease may be more sensitive to the effects of substantial BACE-1 inhibition, perhaps because of a role of BACE-1 in normal synaptic function. It is also possible that the effects of verubecestat are due to inhibition of BACE-2,” they said.
Atabecestat
In a research letter in the same issue of the New England Journal of Medicine (2019 Apr 11;380:1483-5), David Henley, MD, senior director of Janssen’s Alzheimer’s clinical development core, released similarly negative results from an interim analysis of EARLY (Efficacy and Safety Study of Atabecestat in Participants Who Are Asymptomatic at Risk for Developing Alzheimer’s Dementia) trial, a randomized study of the BACE-1 inhibitor candidate, atabecestat.
The phase 2 trial enrolled 557 patients with prodromal Alzheimer’s disease. The primary cognitive end point was change from baseline in the Preclinical Alzheimer’s Cognitive Composite (PACC) score.
This trial was discontinued in May 2018 because of liver-related adverse events, although safety follow-up continues. The research letter did not disclose details of the hepatic events, but a company press release from May 2018 referred to them in a general sense.
“Elevations of liver enzymes, which were serious in nature, have been observed in some study participants who received the Janssen BACE inhibitor, atabecestat. After a thorough evaluation of all available liver safety data from our studies, Janssen has concluded that the benefit-risk ratio is no longer favorable to continue development of atabecestat for people who have late-onset preclinical stage Alzheimer’s disease.”
Patients in EARLY were randomized to 5 mg, 25 mg, or placebo. As in the verubecestat trial, those randomized to placebo did better. The mean changes from baseline in the PACC score were −1.44 in the 25-mg group, −0.58 in the 5-mg group, and −0.32 in the placebo group.
“At month 6, the difference between the 25-mg group and the placebo group was −1.12 and the difference between the 5-mg group and the placebo group was −0.26, favoring placebo over the higher dose,” the authors said.
This theme reemerged in a secondary end point, the Repeatable Battery for the Assessment of Neuropsychological Status. The 25-mg group declined 3.58 points more than placebo, and the 5-mg group, 1.43 points more.
Adverse events were more common in the active groups and included depression, effects on sleep and dreams, and anxiety.
“The differences in cognitive performance between the groups are of uncertain clinical significance; however, given similar findings favoring placebo over BACE-1 inhibitors in other trial, we are communicating this potential signal of worsening cognitive function in the treated groups,” Dr. Henley said.
SOURCE: Egan MF et al. N Engl J Med. 2019 Apr 11;380:1408-20.
More bad news for Alzheimer’s research. Two more BACE inhibitors fall far short of the finish line.
Declining cognitive scores, falls, suicidal ideation, and liver enzyme abnormalities were all seen in clinical trials.
The news doesn’t bode well for the therapeutic target of BACE (beta-site APP cleaving enzyme) inhibition. BACE is one of the enzymes that trims the amyloid precursor protein (APP). Inhibiting it does reduce the amount of toxic amyloid-beta in cerebrospinal fluid, and amyloid plaque in the brain. But none of these molecules has shown clinical benefit in dementia patients, whether their disease is mild, or moderate or – now – prodromal. And it is now apparent that inhibiting BACE also produces serious off-target problems.
“BACE-1 inhibition certainly seemed to have a sound rationale assuming the basis for amyloid’s role in Alzheimer’s disease pathogenesis is a gain of toxicity,” Richard J. Caselli, MD, of the Mayo Clinic, Rochester, Minn., said in an interview. “That APP is important for Alzheimer’s pathogenesis still seems clear but whether amyloid-beta toxicity is the driving force is no longer clear. Further, interruption of the APP system disrupts more than amyloid-beta peptide, possibly explaining the adverse cognitive effects of BACE-1 inhibition shown exhibited now by three different BACE-1 inhibitors.”
Verubecestat
Researchers got their first dose of bad news regarding verubecestat at the 2017 Clinical Trials in Alzheimer’s Disease meeting. There, Michael F. Egan, MD, Merck’s associate vice president of clinical neuroscience, discussed the molecule’s failure to slow cognitive decline in patients with mild to moderate Alzheimer’s disease. There was plenty of biomarker evidence that the drug did block amyloid-beta production, but there also was a plethora of concerning adverse events, Dr. Egan said in an interview.
However, verubecestat still was being pursued in patients with prodromal Alzheimer’s disease. In February, Merck stopped the trial after a futility analysis and announced that the company was terminating studies of verubecestat in that population as well. In the April 11 issue of the New England Journal of Medicine, Dr. Egan and his colleagues report the full extent of verubecestat’s failure in prodromal patients, and the accompanying adverse events.
At the time of termination, 1,454 patients had been enrolled. Of these, 485 received 12 mg/day, 484 received 40 mg/day, and 485 received placebo. About half of each group completed 104 weeks of treatment in the study, which was designed to extend up to 5 years.
The primary outcome was change in the Clinical Dementia Rating Scale–Sum of Boxes score (CDR-SB). Seven secondary outcomes examined other cognitive and functional end points, along with changes in hippocampal volume on MRI and amyloid burden as determined in PET imaging.
Not only did verubecestat fail to slow cognitive decline, it appeared to exacerbate it. The mean change on the CDR-SB was 1.65 in the 12-mg group, 2.02 in the 40-mg group, and 1.58 in the placebo group, favoring placebo.
“In an exploratory analysis according to time point, scores on the CDR-SB were also higher [signifying more impairment of cognition and daily functioning] in the 40-mg group than in the placebo group at 13, 26, and 52 weeks ... suggesting but not confirming the possibility of worse performance at these earlier time points in the high-dose verubecestat group,” the investigators said.
Verubecestat also was associated with more conversions to Alzheimer’s disease. Per 100 patient-years, the Alzheimer’s disease event rates were 24.5 in the 12-mg group, 25.5 in the 40-mg group, and 19.3 in the placebo group. Compared with placebo, those taking 12-mg doses were 30% more likely to develop Alzheimer’s disease and those taking 40-mg doses were 38% more likely. The findings suggest that “verubecestat may have accelerated the progression to diagnosis of dementia due to Alzheimer’s disease,” the investigators said.
The negative impact of verubecestat was apparent quite early in the study. “In exploratory analyses, both dose levels of verubecestat were associated with poorer outcomes on the [Composite Cognition Score-3 Domain] and the ADAS-Cog [Alzheimer’s Disease Assessment Scale–Cognitive Subscale] measures of cognition that, relative to placebo, appeared worse at week 13 and did not appear to progress thereafter.”
Results of the secondary end points, including the ADAS-Cog and the Mini-Mental State Exam, also indicated that verubecestat may have worsened cognitive performance.
Imaging outcomes were positive, however. Hippocampal volume was 6,448 mL in the 12-mg group, 6,469 mL in the 40-mg group, and 6,435 mL in the placebo group. Brain amyloid increased in the placebo group, as expected, and decreased in the active groups. The small group of patients who underwent cerebrospinal fluid sampling showed reductions of more than 60% in amyloid-beta and soluble APP-beta associated with verubecestat. These results show that the molecule was indeed hitting its intended target, but that doing so was not clinically beneficial.
Adverse events were more common in the verubecestat groups. These included rash, dermatitis, urticaria, sleep disturbance, weight loss, and cough. Hair coloring changed in 2.5% of patients in the 12-mg group and 5% of the 40-mg group, but in none of the subjects taking placebo.
Patients taking verubecestat were more likely to sustain falls and injuries and to express suicidal ideation.
The results of this trial differ from the study of verubecestat in mild to moderate Alzheimer’s disease, the investigators noted. Those patients did not decline cognitively as did those with prodromal disease.
“Patients at an earlier stage of the disease may be more sensitive to the effects of substantial BACE-1 inhibition, perhaps because of a role of BACE-1 in normal synaptic function. It is also possible that the effects of verubecestat are due to inhibition of BACE-2,” they said.
Atabecestat
In a research letter in the same issue of the New England Journal of Medicine (2019 Apr 11;380:1483-5), David Henley, MD, senior director of Janssen’s Alzheimer’s clinical development core, released similarly negative results from an interim analysis of EARLY (Efficacy and Safety Study of Atabecestat in Participants Who Are Asymptomatic at Risk for Developing Alzheimer’s Dementia) trial, a randomized study of the BACE-1 inhibitor candidate, atabecestat.
The phase 2 trial enrolled 557 patients with prodromal Alzheimer’s disease. The primary cognitive end point was change from baseline in the Preclinical Alzheimer’s Cognitive Composite (PACC) score.
This trial was discontinued in May 2018 because of liver-related adverse events, although safety follow-up continues. The research letter did not disclose details of the hepatic events, but a company press release from May 2018 referred to them in a general sense.
“Elevations of liver enzymes, which were serious in nature, have been observed in some study participants who received the Janssen BACE inhibitor, atabecestat. After a thorough evaluation of all available liver safety data from our studies, Janssen has concluded that the benefit-risk ratio is no longer favorable to continue development of atabecestat for people who have late-onset preclinical stage Alzheimer’s disease.”
Patients in EARLY were randomized to 5 mg, 25 mg, or placebo. As in the verubecestat trial, those randomized to placebo did better. The mean changes from baseline in the PACC score were −1.44 in the 25-mg group, −0.58 in the 5-mg group, and −0.32 in the placebo group.
“At month 6, the difference between the 25-mg group and the placebo group was −1.12 and the difference between the 5-mg group and the placebo group was −0.26, favoring placebo over the higher dose,” the authors said.
This theme reemerged in a secondary end point, the Repeatable Battery for the Assessment of Neuropsychological Status. The 25-mg group declined 3.58 points more than placebo, and the 5-mg group, 1.43 points more.
Adverse events were more common in the active groups and included depression, effects on sleep and dreams, and anxiety.
“The differences in cognitive performance between the groups are of uncertain clinical significance; however, given similar findings favoring placebo over BACE-1 inhibitors in other trial, we are communicating this potential signal of worsening cognitive function in the treated groups,” Dr. Henley said.
SOURCE: Egan MF et al. N Engl J Med. 2019 Apr 11;380:1408-20.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Bendamustine/rituximab combo proves viable for comorbid CLL
A combination of bendamustine and rituximab generated an 88% overall response rate and 96% overall survival rate at 2 years among patients with chronic lymphocytic leukemia (CLL) in a study of 83 patients aged 53-83 years.
Although combined fludarabine, cyclophosphamide, and rituximab has demonstrated success in younger patients with CLL, this therapy is often considered too aggressive for the majority of CLL patients, who tend to be older and have multiple comorbidities, wrote Martin Špacek, MD, of Charles University and General University Hospital in Prague and his colleagues.
The alternative treatment combination of bendamustine and rituximab (BR) has not been well studied in patients with comorbidities, they said.
In a study published in Leukemia Research, the researchers enrolled 83 previously untreated adults with progressive CLL. The average age of the participants was 71 years, and 61% were men. The median creatinine clearance for the study population was 65 mL/min, and all patients had comorbidities, defined as scores greater than 6 on the Cumulative Illness Rating Scale (CIRS).
All patients were prescribed 90 mg/m2 bendamustine on days 1 and 2 combined with 375 mg/m2 rituximab on day 0 of the first course, and 500 mg/m2 rituximab on day 1 during subsequent courses every 28 days for a maximum of six cycles.
The overall response rate to BR was 88.0%, with a complete response rate of 20.5%. At 2 years, progression-free survival and overall survival rates were 69.9% and 96.2%, respectively.
A total of 51 patients (61.4%) experienced at least one grade 3 or 4 adverse event. The most common hematologic effects were neutropenia (40 patients), thrombocytopenia (14 patients), and anemia (8 patients). The most common nonhematologic effects were grade 3– or grade 4–level infections in 12 patients. Six patients developed severe skin rash.
Additionally, one patient developed sepsis during treatment and died after the first course of therapy.
“Age and CIRS failed to predict any severe toxicities or BR dose reduction,” the researchers noted.
The findings support data from previous studies and represent the largest study of CLL patients with significant comorbidities to be treated with BR, the researchers said.
More prospective research is needed, but the results demonstrate that “chemoimmunotherapy with BR is an effective therapeutic option with manageable toxicity for the initial treatment of CLL patients with significant comorbidities,” the investigators wrote.
The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
SOURCE: Spacek M et al. Leuk Res. 2019;79:17-21.
A combination of bendamustine and rituximab generated an 88% overall response rate and 96% overall survival rate at 2 years among patients with chronic lymphocytic leukemia (CLL) in a study of 83 patients aged 53-83 years.
Although combined fludarabine, cyclophosphamide, and rituximab has demonstrated success in younger patients with CLL, this therapy is often considered too aggressive for the majority of CLL patients, who tend to be older and have multiple comorbidities, wrote Martin Špacek, MD, of Charles University and General University Hospital in Prague and his colleagues.
The alternative treatment combination of bendamustine and rituximab (BR) has not been well studied in patients with comorbidities, they said.
In a study published in Leukemia Research, the researchers enrolled 83 previously untreated adults with progressive CLL. The average age of the participants was 71 years, and 61% were men. The median creatinine clearance for the study population was 65 mL/min, and all patients had comorbidities, defined as scores greater than 6 on the Cumulative Illness Rating Scale (CIRS).
All patients were prescribed 90 mg/m2 bendamustine on days 1 and 2 combined with 375 mg/m2 rituximab on day 0 of the first course, and 500 mg/m2 rituximab on day 1 during subsequent courses every 28 days for a maximum of six cycles.
The overall response rate to BR was 88.0%, with a complete response rate of 20.5%. At 2 years, progression-free survival and overall survival rates were 69.9% and 96.2%, respectively.
A total of 51 patients (61.4%) experienced at least one grade 3 or 4 adverse event. The most common hematologic effects were neutropenia (40 patients), thrombocytopenia (14 patients), and anemia (8 patients). The most common nonhematologic effects were grade 3– or grade 4–level infections in 12 patients. Six patients developed severe skin rash.
Additionally, one patient developed sepsis during treatment and died after the first course of therapy.
“Age and CIRS failed to predict any severe toxicities or BR dose reduction,” the researchers noted.
The findings support data from previous studies and represent the largest study of CLL patients with significant comorbidities to be treated with BR, the researchers said.
More prospective research is needed, but the results demonstrate that “chemoimmunotherapy with BR is an effective therapeutic option with manageable toxicity for the initial treatment of CLL patients with significant comorbidities,” the investigators wrote.
The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
SOURCE: Spacek M et al. Leuk Res. 2019;79:17-21.
A combination of bendamustine and rituximab generated an 88% overall response rate and 96% overall survival rate at 2 years among patients with chronic lymphocytic leukemia (CLL) in a study of 83 patients aged 53-83 years.
Although combined fludarabine, cyclophosphamide, and rituximab has demonstrated success in younger patients with CLL, this therapy is often considered too aggressive for the majority of CLL patients, who tend to be older and have multiple comorbidities, wrote Martin Špacek, MD, of Charles University and General University Hospital in Prague and his colleagues.
The alternative treatment combination of bendamustine and rituximab (BR) has not been well studied in patients with comorbidities, they said.
In a study published in Leukemia Research, the researchers enrolled 83 previously untreated adults with progressive CLL. The average age of the participants was 71 years, and 61% were men. The median creatinine clearance for the study population was 65 mL/min, and all patients had comorbidities, defined as scores greater than 6 on the Cumulative Illness Rating Scale (CIRS).
All patients were prescribed 90 mg/m2 bendamustine on days 1 and 2 combined with 375 mg/m2 rituximab on day 0 of the first course, and 500 mg/m2 rituximab on day 1 during subsequent courses every 28 days for a maximum of six cycles.
The overall response rate to BR was 88.0%, with a complete response rate of 20.5%. At 2 years, progression-free survival and overall survival rates were 69.9% and 96.2%, respectively.
A total of 51 patients (61.4%) experienced at least one grade 3 or 4 adverse event. The most common hematologic effects were neutropenia (40 patients), thrombocytopenia (14 patients), and anemia (8 patients). The most common nonhematologic effects were grade 3– or grade 4–level infections in 12 patients. Six patients developed severe skin rash.
Additionally, one patient developed sepsis during treatment and died after the first course of therapy.
“Age and CIRS failed to predict any severe toxicities or BR dose reduction,” the researchers noted.
The findings support data from previous studies and represent the largest study of CLL patients with significant comorbidities to be treated with BR, the researchers said.
More prospective research is needed, but the results demonstrate that “chemoimmunotherapy with BR is an effective therapeutic option with manageable toxicity for the initial treatment of CLL patients with significant comorbidities,” the investigators wrote.
The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
SOURCE: Spacek M et al. Leuk Res. 2019;79:17-21.
FROM LEUKEMIA RESEARCH
Key clinical point:
Major finding: The overall response rate for the combination therapy was 88.0%; complete response was 20.5%.
Study details: A prospective, observational study of 83 patients with chronic lymphocytic leukemia.
Disclosures: The study was supported by the Ministry of Health, Czech Republic, the Charles University Progres program, and the Czech CLL Study Group. Researchers reported honoraria and travel grants from Mundipharma and Roche.
Source: Spacek M et al. Leuk Res. 2019;79:17-21.
In situ vaccination produced responses in indolent NHL
A three-pronged treatment approach can produce responses in indolent non-Hodgkin lymphoma (iNHL), according to research published in Nature Medicine.
The approach – “in situ vaccination (ISV)” – involves intratumoral injections of Fms-like tyrosine kinase 3 ligand (Flt3L), local radiotherapy, and intratumoral injections of a TLR3 agonist (poly-ICLC).
ISV produced responses in patients with iNHL, prompting regression of tumors that were directly targeted with ISV, as well as untreated tumors.
In preclinical experiments, ISV induced tumor regression in mice but also overcame resistance to PD1 inhibition. This result led researchers to initiate a trial testing ISV in combination with pembrolizumab in patients with lymphoma and solid tumors.
“We discovered why some tumors do not respond to PD1 blockade: insufficient dendritic cells (DCs) and cross-presentation,” lead study author Joshua Brody, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview. “We developed a treatment, in situ vaccination (ISV), which brings DCs to the tumor, loads them with tumor antigens, and activates the DCs.”
Specifically, the researchers found that injecting Flt3L into a tumor recruits intratumoral DCs, local radiotherapy loads the DCs with tumor-associated antigens, and poly-ICLC activates DCs. This approach produced responses in mouse models of lymphoma and patients with iNHL.
Preclinical results
Dr. Brody and his colleagues tested ISV in A20 tumor-bearing mice. The mice received intratumoral injections of Flt3L, followed by local radiotherapy and poly-ICLC.
Tumor regression occurred within days of radiotherapy. About 40% of mice experienced tumor-free survival of at least 3 months, although most tumors recurred within 4 weeks of ISV administration.
However, the researchers observed increased PD1 and PD-L1 expression in ISV-treated mice, so the team theorized that an anti-PD1 monoclonal antibody (RMP1-14) could improve the efficacy of ISV.
The researchers found that ISV plus RMP1-14 delayed tumor growth when compared with ISV alone, and the rate of durable remissions increased from about 40% to about 80%.
Clinical results
Dr. Brody and his colleagues also tested ISV in a clinical trial. That trial included 11 iNHL patients – 9 with follicular lymphoma, 1 with marginal zone lymphoma, and 1 with small lymphocytic lymphoma.
The patients received nine daily injections of Flt3L (25 mcg/kg) into a target lesion, then two doses of radiation (2 Gy) to the same lesion, and eight intratumoral injections of poly-ICLC (2 mg).
“We ... have observed dramatic clinical responses; i.e., we administer ISV at one tumor site, and tumors throughout the body regress,” Dr. Brody said.
At the target lesion, there were two complete responses, six partial responses, and three cases of stable disease. At nontarget lesions, there was one complete response, two partial responses, six cases of stable disease, and two cases of progression.
ISV was considered well tolerated. One patient had grade 2 fever, three had grade 1 fever, and nine had grade 1 flu-like symptoms. Two patients did not have any adverse events.
This research was supported by Merck, Celldex Therapeutics, Oncovir, and Genentech. The authors reported relationships with Acerta Pharma, Bristol Myers Squibb, Genentech, Gilead Sciences, Seattle Genetics, Pharmacyclics, Celgene, Celldex Therapeutics, and Oncovir.
SOURCE: Hammerich L et al. Nat Med. 2019 Apr 8. doi: 10.1038/s41591-019-0410-x.
A three-pronged treatment approach can produce responses in indolent non-Hodgkin lymphoma (iNHL), according to research published in Nature Medicine.
The approach – “in situ vaccination (ISV)” – involves intratumoral injections of Fms-like tyrosine kinase 3 ligand (Flt3L), local radiotherapy, and intratumoral injections of a TLR3 agonist (poly-ICLC).
ISV produced responses in patients with iNHL, prompting regression of tumors that were directly targeted with ISV, as well as untreated tumors.
In preclinical experiments, ISV induced tumor regression in mice but also overcame resistance to PD1 inhibition. This result led researchers to initiate a trial testing ISV in combination with pembrolizumab in patients with lymphoma and solid tumors.
“We discovered why some tumors do not respond to PD1 blockade: insufficient dendritic cells (DCs) and cross-presentation,” lead study author Joshua Brody, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview. “We developed a treatment, in situ vaccination (ISV), which brings DCs to the tumor, loads them with tumor antigens, and activates the DCs.”
Specifically, the researchers found that injecting Flt3L into a tumor recruits intratumoral DCs, local radiotherapy loads the DCs with tumor-associated antigens, and poly-ICLC activates DCs. This approach produced responses in mouse models of lymphoma and patients with iNHL.
Preclinical results
Dr. Brody and his colleagues tested ISV in A20 tumor-bearing mice. The mice received intratumoral injections of Flt3L, followed by local radiotherapy and poly-ICLC.
Tumor regression occurred within days of radiotherapy. About 40% of mice experienced tumor-free survival of at least 3 months, although most tumors recurred within 4 weeks of ISV administration.
However, the researchers observed increased PD1 and PD-L1 expression in ISV-treated mice, so the team theorized that an anti-PD1 monoclonal antibody (RMP1-14) could improve the efficacy of ISV.
The researchers found that ISV plus RMP1-14 delayed tumor growth when compared with ISV alone, and the rate of durable remissions increased from about 40% to about 80%.
Clinical results
Dr. Brody and his colleagues also tested ISV in a clinical trial. That trial included 11 iNHL patients – 9 with follicular lymphoma, 1 with marginal zone lymphoma, and 1 with small lymphocytic lymphoma.
The patients received nine daily injections of Flt3L (25 mcg/kg) into a target lesion, then two doses of radiation (2 Gy) to the same lesion, and eight intratumoral injections of poly-ICLC (2 mg).
“We ... have observed dramatic clinical responses; i.e., we administer ISV at one tumor site, and tumors throughout the body regress,” Dr. Brody said.
At the target lesion, there were two complete responses, six partial responses, and three cases of stable disease. At nontarget lesions, there was one complete response, two partial responses, six cases of stable disease, and two cases of progression.
ISV was considered well tolerated. One patient had grade 2 fever, three had grade 1 fever, and nine had grade 1 flu-like symptoms. Two patients did not have any adverse events.
This research was supported by Merck, Celldex Therapeutics, Oncovir, and Genentech. The authors reported relationships with Acerta Pharma, Bristol Myers Squibb, Genentech, Gilead Sciences, Seattle Genetics, Pharmacyclics, Celgene, Celldex Therapeutics, and Oncovir.
SOURCE: Hammerich L et al. Nat Med. 2019 Apr 8. doi: 10.1038/s41591-019-0410-x.
A three-pronged treatment approach can produce responses in indolent non-Hodgkin lymphoma (iNHL), according to research published in Nature Medicine.
The approach – “in situ vaccination (ISV)” – involves intratumoral injections of Fms-like tyrosine kinase 3 ligand (Flt3L), local radiotherapy, and intratumoral injections of a TLR3 agonist (poly-ICLC).
ISV produced responses in patients with iNHL, prompting regression of tumors that were directly targeted with ISV, as well as untreated tumors.
In preclinical experiments, ISV induced tumor regression in mice but also overcame resistance to PD1 inhibition. This result led researchers to initiate a trial testing ISV in combination with pembrolizumab in patients with lymphoma and solid tumors.
“We discovered why some tumors do not respond to PD1 blockade: insufficient dendritic cells (DCs) and cross-presentation,” lead study author Joshua Brody, MD, of the Icahn School of Medicine at Mount Sinai, New York, said in an interview. “We developed a treatment, in situ vaccination (ISV), which brings DCs to the tumor, loads them with tumor antigens, and activates the DCs.”
Specifically, the researchers found that injecting Flt3L into a tumor recruits intratumoral DCs, local radiotherapy loads the DCs with tumor-associated antigens, and poly-ICLC activates DCs. This approach produced responses in mouse models of lymphoma and patients with iNHL.
Preclinical results
Dr. Brody and his colleagues tested ISV in A20 tumor-bearing mice. The mice received intratumoral injections of Flt3L, followed by local radiotherapy and poly-ICLC.
Tumor regression occurred within days of radiotherapy. About 40% of mice experienced tumor-free survival of at least 3 months, although most tumors recurred within 4 weeks of ISV administration.
However, the researchers observed increased PD1 and PD-L1 expression in ISV-treated mice, so the team theorized that an anti-PD1 monoclonal antibody (RMP1-14) could improve the efficacy of ISV.
The researchers found that ISV plus RMP1-14 delayed tumor growth when compared with ISV alone, and the rate of durable remissions increased from about 40% to about 80%.
Clinical results
Dr. Brody and his colleagues also tested ISV in a clinical trial. That trial included 11 iNHL patients – 9 with follicular lymphoma, 1 with marginal zone lymphoma, and 1 with small lymphocytic lymphoma.
The patients received nine daily injections of Flt3L (25 mcg/kg) into a target lesion, then two doses of radiation (2 Gy) to the same lesion, and eight intratumoral injections of poly-ICLC (2 mg).
“We ... have observed dramatic clinical responses; i.e., we administer ISV at one tumor site, and tumors throughout the body regress,” Dr. Brody said.
At the target lesion, there were two complete responses, six partial responses, and three cases of stable disease. At nontarget lesions, there was one complete response, two partial responses, six cases of stable disease, and two cases of progression.
ISV was considered well tolerated. One patient had grade 2 fever, three had grade 1 fever, and nine had grade 1 flu-like symptoms. Two patients did not have any adverse events.
This research was supported by Merck, Celldex Therapeutics, Oncovir, and Genentech. The authors reported relationships with Acerta Pharma, Bristol Myers Squibb, Genentech, Gilead Sciences, Seattle Genetics, Pharmacyclics, Celgene, Celldex Therapeutics, and Oncovir.
SOURCE: Hammerich L et al. Nat Med. 2019 Apr 8. doi: 10.1038/s41591-019-0410-x.
FROM NATURE MEDICINE
DA-EPOCH-R appears more toxic than standard R-CHOP in DLBCL
The use of dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R) as upfront treatment in patients with diffuse large B-cell lymphoma (DLBCL) showed greater toxicity and did not improve progression-free survival versus standard rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), according to results from a phase 3 trial.
“Less favorable outcomes for patients with recurrent DLBCL prompted efforts to improve first-line approaches and biomarkers to identify high-risk patients,” wrote Nancy L. Bartlett, MD, of Washington University, St. Louis, and her colleagues wrote in the Journal of Clinical Oncology.
The Alliance/CALGB 50303 study included 491 patients with DLBCL who were randomized in a 1:1 fashion to receive DA-EPOCH-R or R-CHOP every 21 days for a total of six cycles. Dosing for the DA-EPOCH-R regimen was determined using absolute neutrophil and platelet counts.
The primary endpoint measured was progression-free survival (PFS); secondary endpoints included safety, overall survival (OS), and response rate.
After a median follow-up of 5.2 years, the researchers found no significant difference in PFS between the study arms (DA-EPOCH-R hazard ratio, 0.93; 95% confidence interval, 0.68-1.27; P = .65). Additionally, there was no significant difference in OS (HR, 1.09; 95% CI, 0.75-1.59; P = .64).
The overall response rate was 88.0% in the R-CHOP arm versus 86.7% in the DA-EPOCH-R arm (P = .67).
With respect to safety, grade 3 or 4 adverse events were more frequently seen in the DA-EPOCH-R group than in the R-CHOP group (P less than .001). These toxicities included febrile neutropenia, infections, neuropathy, and mucositis.
The researchers did see significantly improved PFS in the DA-EPOCH-R arm in post hoc subset analyses of patients with International Prognostic Index (IPI) 3-5, but the subset analysis “was unplanned and not powered” and the significance “must be tempered in light of multiple comparisons.”
“We now understand DLBCL is even more heterogeneous than appreciated when this trial was designed,” the researchers wrote. “Therefore, the National Clinical Trials Network is planning a precision medicine approach to identify molecular subsets of DLBCL and determine if specific chemotherapy platforms and/or targeted agents offer differential benefit.”
The study was supported by the National Cancer Institute. The authors reported financial relationships with Bristol-Myers Squibb, Celgene, Janssen, Jazz Pharmaceuticals, Morphosys, and other companies.
SOURCE: Bartlett NL et al. J Clin Oncol. 2019 Apr 2. doi: 10.1200/JCO.18.01994.
The use of dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R) as upfront treatment in patients with diffuse large B-cell lymphoma (DLBCL) showed greater toxicity and did not improve progression-free survival versus standard rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), according to results from a phase 3 trial.
“Less favorable outcomes for patients with recurrent DLBCL prompted efforts to improve first-line approaches and biomarkers to identify high-risk patients,” wrote Nancy L. Bartlett, MD, of Washington University, St. Louis, and her colleagues wrote in the Journal of Clinical Oncology.
The Alliance/CALGB 50303 study included 491 patients with DLBCL who were randomized in a 1:1 fashion to receive DA-EPOCH-R or R-CHOP every 21 days for a total of six cycles. Dosing for the DA-EPOCH-R regimen was determined using absolute neutrophil and platelet counts.
The primary endpoint measured was progression-free survival (PFS); secondary endpoints included safety, overall survival (OS), and response rate.
After a median follow-up of 5.2 years, the researchers found no significant difference in PFS between the study arms (DA-EPOCH-R hazard ratio, 0.93; 95% confidence interval, 0.68-1.27; P = .65). Additionally, there was no significant difference in OS (HR, 1.09; 95% CI, 0.75-1.59; P = .64).
The overall response rate was 88.0% in the R-CHOP arm versus 86.7% in the DA-EPOCH-R arm (P = .67).
With respect to safety, grade 3 or 4 adverse events were more frequently seen in the DA-EPOCH-R group than in the R-CHOP group (P less than .001). These toxicities included febrile neutropenia, infections, neuropathy, and mucositis.
The researchers did see significantly improved PFS in the DA-EPOCH-R arm in post hoc subset analyses of patients with International Prognostic Index (IPI) 3-5, but the subset analysis “was unplanned and not powered” and the significance “must be tempered in light of multiple comparisons.”
“We now understand DLBCL is even more heterogeneous than appreciated when this trial was designed,” the researchers wrote. “Therefore, the National Clinical Trials Network is planning a precision medicine approach to identify molecular subsets of DLBCL and determine if specific chemotherapy platforms and/or targeted agents offer differential benefit.”
The study was supported by the National Cancer Institute. The authors reported financial relationships with Bristol-Myers Squibb, Celgene, Janssen, Jazz Pharmaceuticals, Morphosys, and other companies.
SOURCE: Bartlett NL et al. J Clin Oncol. 2019 Apr 2. doi: 10.1200/JCO.18.01994.
The use of dose-adjusted etoposide, prednisone, vincristine, cyclophosphamide, doxorubicin, and rituximab (DA-EPOCH-R) as upfront treatment in patients with diffuse large B-cell lymphoma (DLBCL) showed greater toxicity and did not improve progression-free survival versus standard rituximab, cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP), according to results from a phase 3 trial.
“Less favorable outcomes for patients with recurrent DLBCL prompted efforts to improve first-line approaches and biomarkers to identify high-risk patients,” wrote Nancy L. Bartlett, MD, of Washington University, St. Louis, and her colleagues wrote in the Journal of Clinical Oncology.
The Alliance/CALGB 50303 study included 491 patients with DLBCL who were randomized in a 1:1 fashion to receive DA-EPOCH-R or R-CHOP every 21 days for a total of six cycles. Dosing for the DA-EPOCH-R regimen was determined using absolute neutrophil and platelet counts.
The primary endpoint measured was progression-free survival (PFS); secondary endpoints included safety, overall survival (OS), and response rate.
After a median follow-up of 5.2 years, the researchers found no significant difference in PFS between the study arms (DA-EPOCH-R hazard ratio, 0.93; 95% confidence interval, 0.68-1.27; P = .65). Additionally, there was no significant difference in OS (HR, 1.09; 95% CI, 0.75-1.59; P = .64).
The overall response rate was 88.0% in the R-CHOP arm versus 86.7% in the DA-EPOCH-R arm (P = .67).
With respect to safety, grade 3 or 4 adverse events were more frequently seen in the DA-EPOCH-R group than in the R-CHOP group (P less than .001). These toxicities included febrile neutropenia, infections, neuropathy, and mucositis.
The researchers did see significantly improved PFS in the DA-EPOCH-R arm in post hoc subset analyses of patients with International Prognostic Index (IPI) 3-5, but the subset analysis “was unplanned and not powered” and the significance “must be tempered in light of multiple comparisons.”
“We now understand DLBCL is even more heterogeneous than appreciated when this trial was designed,” the researchers wrote. “Therefore, the National Clinical Trials Network is planning a precision medicine approach to identify molecular subsets of DLBCL and determine if specific chemotherapy platforms and/or targeted agents offer differential benefit.”
The study was supported by the National Cancer Institute. The authors reported financial relationships with Bristol-Myers Squibb, Celgene, Janssen, Jazz Pharmaceuticals, Morphosys, and other companies.
SOURCE: Bartlett NL et al. J Clin Oncol. 2019 Apr 2. doi: 10.1200/JCO.18.01994.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Polatuzumab outperforms pinatuzumab in non-Hodgkin lymphoma
Favorable results from a phase 2 trial have prompted further development of polatuzumab vedotin in non-Hodgkin lymphoma.
In the ROMULUS trial, polatuzumab vedotin plus rituximab (R-pola) produced more durable responses than did pinatuzumab vedotin plus rituximab (R-pina) in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) or follicular lymphoma (FL).
Researchers also observed a more favorable benefit-risk profile with R-pola.
Franck Morschhauser, MD, of Centre Hospitalier Régional Universitaire de Lille, France, and his colleagues described these findings in the Lancet Haematology.
The ROMULUS trial included 81 DLBCL patients and 42 FL patients. They were randomized to receive R-pola or R-pina (rituximab at 375 mg/m2 plus either antibody-drug conjugate at 2.4 mg/kg) every 21 days until disease progression or unacceptable toxicity for up to 1 year.
Among DLBCL patients, the median age was 69 years in the R-pina arm and 68 years in the R-pola arm. Among FL patients, the median age was 59 years in the R-pina arm and 67 years in the R-pola arm.
Seventy-six percent of DLBCL patients randomized to R-pina were refractory to their last treatment, as were 80% of DLBCL patients assigned to R-pola, 52% of FL patients assigned to R-pina, and 35% of FL patients assigned to R-pola.
The median number of prior systemic therapies was three in the R-pina DLBCL arm, the R-pola DLBCL arm, and the R-pina FL arm. The median number of prior therapies was two in the R-pola FL arm.
Response and survival
Among the DLBCL patients, R-pina produced an objective response rate (ORR) of 60% and a complete response (CR) rate of 26%. R-pola produced an ORR of 54% and a CR rate of 21%. The median duration of response was 6.2 months in the R-pina arm and 13.4 months in the R-pola arm.
The median progression-free survival in the DLBCL cohort was 5.4 months for the R-pina arm and 5.6 months for the R-pola arm. The median overall survival was 16.5 months and 20.1 months, respectively.
In the FL cohort, R-pina produced an ORR of 62% and a CR rate of 5%. R-pola produced an ORR of 70% and a CR rate of 45%. The median duration of response was 6.5 months in the R-pina arm and 9.4 months in the R-pola arm.
The median progression-free survival in the FL cohort was 12.7 months for the R-pina arm and 15.3 months for the R-pola arm. The 2-year overall survival rate was 90.5% and 87.8%, respectively. The median overall survival was not reached in either arm.
“Patients treated with R-pola tended to have longer durations of response than those receiving R-pina (particularly those with relapsed or refractory diffuse large B-cell lymphoma), and the results for R-pola compared favorably with other novel antilymphoma agents,” Dr. Morschhauser and his colleagues wrote.
Safety
Among DLBCL patients, serious adverse events (AEs) occurred in 50.0% of those in the R-pina arm and 35.9% of those in the R-pola arm. Among FL patients, serious AEs occurred in 28.6% of those the R-pina arm and 35.0% of those in the R-pola arm.
Ten grade 5 AEs occurred in nine DLBCL patients who received R-pina (21.4%). These events included two cases of sepsis, influenza and pneumonia in the same patient, general physical health deterioration including one death attributed to disease progression, and one case each of Clostridium difficile sepsis, respiratory failure, urosepsis, and sudden death.
There was one grade 5 AE in a FL patient who received R-pola. The 84-year-old patient died of pulmonary congestion 64 days after the last of 12 cycles of treatment.
There were no fatal AEs in the other arms.
“These findings make pola a promising novel candidate for further clinical evaluation in combination regimens in treatment-refractory patients and also in a first-line setting in B-cell non-Hodgkin lymphoma,” Dr. Morschhauser and his colleagues wrote.
Polatuzumab vedotin was chosen by the study funder for further development in non-Hodgkin lymphoma, partly because of longer durations of response, compared with pinatuzumab vedotin.
Polatuzumab vedotin is currently under investigation in the phase 3 POLARIX study. The drug is being combined with rituximab, cyclophosphamide, doxorubicin, and prednisone and compared to rituximab-cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in patients with DLBCL.
The ROMULUS study was funded by F Hoffmann-La Roche. The study authors reported relationships with Roche and other companies.
SOURCE: Morschhauser F et al. Lancet Haematol. 2019 Mar 29. doi: 10.1016/S2352-3026(19)30026-2.
Favorable results from a phase 2 trial have prompted further development of polatuzumab vedotin in non-Hodgkin lymphoma.
In the ROMULUS trial, polatuzumab vedotin plus rituximab (R-pola) produced more durable responses than did pinatuzumab vedotin plus rituximab (R-pina) in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) or follicular lymphoma (FL).
Researchers also observed a more favorable benefit-risk profile with R-pola.
Franck Morschhauser, MD, of Centre Hospitalier Régional Universitaire de Lille, France, and his colleagues described these findings in the Lancet Haematology.
The ROMULUS trial included 81 DLBCL patients and 42 FL patients. They were randomized to receive R-pola or R-pina (rituximab at 375 mg/m2 plus either antibody-drug conjugate at 2.4 mg/kg) every 21 days until disease progression or unacceptable toxicity for up to 1 year.
Among DLBCL patients, the median age was 69 years in the R-pina arm and 68 years in the R-pola arm. Among FL patients, the median age was 59 years in the R-pina arm and 67 years in the R-pola arm.
Seventy-six percent of DLBCL patients randomized to R-pina were refractory to their last treatment, as were 80% of DLBCL patients assigned to R-pola, 52% of FL patients assigned to R-pina, and 35% of FL patients assigned to R-pola.
The median number of prior systemic therapies was three in the R-pina DLBCL arm, the R-pola DLBCL arm, and the R-pina FL arm. The median number of prior therapies was two in the R-pola FL arm.
Response and survival
Among the DLBCL patients, R-pina produced an objective response rate (ORR) of 60% and a complete response (CR) rate of 26%. R-pola produced an ORR of 54% and a CR rate of 21%. The median duration of response was 6.2 months in the R-pina arm and 13.4 months in the R-pola arm.
The median progression-free survival in the DLBCL cohort was 5.4 months for the R-pina arm and 5.6 months for the R-pola arm. The median overall survival was 16.5 months and 20.1 months, respectively.
In the FL cohort, R-pina produced an ORR of 62% and a CR rate of 5%. R-pola produced an ORR of 70% and a CR rate of 45%. The median duration of response was 6.5 months in the R-pina arm and 9.4 months in the R-pola arm.
The median progression-free survival in the FL cohort was 12.7 months for the R-pina arm and 15.3 months for the R-pola arm. The 2-year overall survival rate was 90.5% and 87.8%, respectively. The median overall survival was not reached in either arm.
“Patients treated with R-pola tended to have longer durations of response than those receiving R-pina (particularly those with relapsed or refractory diffuse large B-cell lymphoma), and the results for R-pola compared favorably with other novel antilymphoma agents,” Dr. Morschhauser and his colleagues wrote.
Safety
Among DLBCL patients, serious adverse events (AEs) occurred in 50.0% of those in the R-pina arm and 35.9% of those in the R-pola arm. Among FL patients, serious AEs occurred in 28.6% of those the R-pina arm and 35.0% of those in the R-pola arm.
Ten grade 5 AEs occurred in nine DLBCL patients who received R-pina (21.4%). These events included two cases of sepsis, influenza and pneumonia in the same patient, general physical health deterioration including one death attributed to disease progression, and one case each of Clostridium difficile sepsis, respiratory failure, urosepsis, and sudden death.
There was one grade 5 AE in a FL patient who received R-pola. The 84-year-old patient died of pulmonary congestion 64 days after the last of 12 cycles of treatment.
There were no fatal AEs in the other arms.
“These findings make pola a promising novel candidate for further clinical evaluation in combination regimens in treatment-refractory patients and also in a first-line setting in B-cell non-Hodgkin lymphoma,” Dr. Morschhauser and his colleagues wrote.
Polatuzumab vedotin was chosen by the study funder for further development in non-Hodgkin lymphoma, partly because of longer durations of response, compared with pinatuzumab vedotin.
Polatuzumab vedotin is currently under investigation in the phase 3 POLARIX study. The drug is being combined with rituximab, cyclophosphamide, doxorubicin, and prednisone and compared to rituximab-cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in patients with DLBCL.
The ROMULUS study was funded by F Hoffmann-La Roche. The study authors reported relationships with Roche and other companies.
SOURCE: Morschhauser F et al. Lancet Haematol. 2019 Mar 29. doi: 10.1016/S2352-3026(19)30026-2.
Favorable results from a phase 2 trial have prompted further development of polatuzumab vedotin in non-Hodgkin lymphoma.
In the ROMULUS trial, polatuzumab vedotin plus rituximab (R-pola) produced more durable responses than did pinatuzumab vedotin plus rituximab (R-pina) in patients with relapsed or refractory diffuse large B-cell lymphoma (DLBCL) or follicular lymphoma (FL).
Researchers also observed a more favorable benefit-risk profile with R-pola.
Franck Morschhauser, MD, of Centre Hospitalier Régional Universitaire de Lille, France, and his colleagues described these findings in the Lancet Haematology.
The ROMULUS trial included 81 DLBCL patients and 42 FL patients. They were randomized to receive R-pola or R-pina (rituximab at 375 mg/m2 plus either antibody-drug conjugate at 2.4 mg/kg) every 21 days until disease progression or unacceptable toxicity for up to 1 year.
Among DLBCL patients, the median age was 69 years in the R-pina arm and 68 years in the R-pola arm. Among FL patients, the median age was 59 years in the R-pina arm and 67 years in the R-pola arm.
Seventy-six percent of DLBCL patients randomized to R-pina were refractory to their last treatment, as were 80% of DLBCL patients assigned to R-pola, 52% of FL patients assigned to R-pina, and 35% of FL patients assigned to R-pola.
The median number of prior systemic therapies was three in the R-pina DLBCL arm, the R-pola DLBCL arm, and the R-pina FL arm. The median number of prior therapies was two in the R-pola FL arm.
Response and survival
Among the DLBCL patients, R-pina produced an objective response rate (ORR) of 60% and a complete response (CR) rate of 26%. R-pola produced an ORR of 54% and a CR rate of 21%. The median duration of response was 6.2 months in the R-pina arm and 13.4 months in the R-pola arm.
The median progression-free survival in the DLBCL cohort was 5.4 months for the R-pina arm and 5.6 months for the R-pola arm. The median overall survival was 16.5 months and 20.1 months, respectively.
In the FL cohort, R-pina produced an ORR of 62% and a CR rate of 5%. R-pola produced an ORR of 70% and a CR rate of 45%. The median duration of response was 6.5 months in the R-pina arm and 9.4 months in the R-pola arm.
The median progression-free survival in the FL cohort was 12.7 months for the R-pina arm and 15.3 months for the R-pola arm. The 2-year overall survival rate was 90.5% and 87.8%, respectively. The median overall survival was not reached in either arm.
“Patients treated with R-pola tended to have longer durations of response than those receiving R-pina (particularly those with relapsed or refractory diffuse large B-cell lymphoma), and the results for R-pola compared favorably with other novel antilymphoma agents,” Dr. Morschhauser and his colleagues wrote.
Safety
Among DLBCL patients, serious adverse events (AEs) occurred in 50.0% of those in the R-pina arm and 35.9% of those in the R-pola arm. Among FL patients, serious AEs occurred in 28.6% of those the R-pina arm and 35.0% of those in the R-pola arm.
Ten grade 5 AEs occurred in nine DLBCL patients who received R-pina (21.4%). These events included two cases of sepsis, influenza and pneumonia in the same patient, general physical health deterioration including one death attributed to disease progression, and one case each of Clostridium difficile sepsis, respiratory failure, urosepsis, and sudden death.
There was one grade 5 AE in a FL patient who received R-pola. The 84-year-old patient died of pulmonary congestion 64 days after the last of 12 cycles of treatment.
There were no fatal AEs in the other arms.
“These findings make pola a promising novel candidate for further clinical evaluation in combination regimens in treatment-refractory patients and also in a first-line setting in B-cell non-Hodgkin lymphoma,” Dr. Morschhauser and his colleagues wrote.
Polatuzumab vedotin was chosen by the study funder for further development in non-Hodgkin lymphoma, partly because of longer durations of response, compared with pinatuzumab vedotin.
Polatuzumab vedotin is currently under investigation in the phase 3 POLARIX study. The drug is being combined with rituximab, cyclophosphamide, doxorubicin, and prednisone and compared to rituximab-cyclophosphamide, doxorubicin, vincristine, and prednisone (R-CHOP) in patients with DLBCL.
The ROMULUS study was funded by F Hoffmann-La Roche. The study authors reported relationships with Roche and other companies.
SOURCE: Morschhauser F et al. Lancet Haematol. 2019 Mar 29. doi: 10.1016/S2352-3026(19)30026-2.
FROM LANCET HAEMATOLOGY
Early data support R-BAC for post-BTKi mantle cell lymphoma
GLASGOW – Patients with relapsed or refractory mantle cell lymphoma (MCL) who experience disease progression on a Bruton’s tyrosine kinase inhibitor (BTKi) may respond best to a combination of rituximab, bendamustine, and cytarabine (R-BAC), based on early results from an ongoing retrospective study.
Findings from the study, which were presented at the annual meeting of the British Society for Haematology, showed that R-BAC after BTKi failure had an overall response rate (ORR) of 90.5%.
This is a “remarkable response rate” according to the investigators, who cited previously reported response rates for other treatments ranging from 29% to 53%.
Treatment of relapsed/refractory MCL patients in the post-BTKi setting is an area of unmet clinical need, said senior author Simon Rule, MD, of the University of Plymouth, England. He noted that there is currently no consensus regarding best treatment strategy for this patient population.
Dr. Rule said that he and his colleagues have collected data on 30 patients so far, of which 22 were included in this early data release.
All patients received R-BAC between 2016 and 2018 at treatment centers in Italy and the United Kingdom. Treatment consisted of rituximab (375 mg/m2 or 500 mg) on day 1, bendamustine 70 mg/m2 on days 1 and 2, and cytarabine 500 mg/m2 on days 1 through 3, given in a 28-day cycle.
Patients received R-BAC immediately after BTKi failure. Data were drawn from hospital records.
Analysis showed that the median patient age was 65 years, with a range from 43 to 79 years. Most patients were men (81.8%), 55.0% were high risk based on the Mantle Cell Lymphoma International Prognostic Index, and 22.7% had blastoid morphology.
Patients had a median of two prior systemic therapies, with a range from one to six lines. First-line therapies included rituximab in combination with HDAC (high-dose cytarabine containing regimen), CHOP, CVP, or ibrutinib. Nine patients (42.9%) had allogeneic stem cell transplantation (ASCT) after induction treatment.
For BTKi therapy, most patients received ibrutinib (n = 18), while the remainder received acalabrutinib, tirabrutinib or M7583. Most patients discontinued BTKi therapy because of disease progression (90.9%); two patients stopped because of a lack of response (9.1%).
The median number of R-BAC cycles received was four. Two patients started with attenuated doses and seven patients reduced doses after the first cycle. More than 70% of patients completed R-BAC treatment.
The estimated median progression-free survival was 7.3 months and estimated median overall survival was 11.2 months.
Although the investigators reported a complete response rate of 57.1%, they noted that this figure “may be exaggerated” because of a lack of bone marrow biopsy; however, they suggested that the overall response rate (90.5%) “should be accurate.”
During the course of treatment, 31.8% of patients required inpatient admission, 22.7% developed neutropenic fever, and 77.8% required transfusion support. No treatment-related deaths occurred.
“This population, enriched for patients with high risk features, showed remarkable response rates to R-BAC,” the investigators wrote. “The treatment had acceptable toxicity, maintained efficacy at attenuated doses, and was used successfully as a bridge to ASCT in over 20% of patients.”
The investigators suggested that R-BAC should be considered a new standard of care in the United Kingdom for bendamustine-naive patients who are unable to be enrolled in clinical trials. “The high response rate makes it particularly appealing for patients considered candidates for consolidation ASCT,” they wrote.
In an interview, Dr. Rule added perspective to these findings.
“There’s been an obsession with venetoclax, that that’s the answer, but it really isn’t,” Dr. Rule said. “So people are looking for a new drug. I guess what I do differently to most people is I use CHOP frontline rather than bendamustine. To me, that’s the best way of sequencing the therapies, whereas if you use [bendamustine and rituximab] up front, which a lot of people do, particularly in the [United] States, your R-BAC might not be so effective.”
However, Dr. Rule said that first-line therapies appear to have minimal impact on R-BAC efficacy. “Even if you’ve had bendamustine, even if you’ve had high-dose cytarabine, even if you’ve had an allogeneic stem cell transplant, [R-BAC] still works,” he said.
Where patients have issues with tolerability, Dr. Rule noted that dose reductions are possible without sacrificing efficacy.
He offered an example of such a scenario. “My oldest patient was about 80 with blastoid disease, relapsing,” Dr. Rule said. “After ibrutinib, I gave him just a single dose of bendamustine at 70 mg, a single dose of cytarabine at 500 mg, just 1 day, and he had that six times, probably 3 weeks apart. He’s been in complete remission for over a year.”
With data on 30 patients collected, Dr. Rule said that he and his colleagues plan to present more extensive findings at the European Hematology Association Congress, held June 13-16 in Amsterdam.
The investigators reported having no conflicts of interest.
GLASGOW – Patients with relapsed or refractory mantle cell lymphoma (MCL) who experience disease progression on a Bruton’s tyrosine kinase inhibitor (BTKi) may respond best to a combination of rituximab, bendamustine, and cytarabine (R-BAC), based on early results from an ongoing retrospective study.
Findings from the study, which were presented at the annual meeting of the British Society for Haematology, showed that R-BAC after BTKi failure had an overall response rate (ORR) of 90.5%.
This is a “remarkable response rate” according to the investigators, who cited previously reported response rates for other treatments ranging from 29% to 53%.
Treatment of relapsed/refractory MCL patients in the post-BTKi setting is an area of unmet clinical need, said senior author Simon Rule, MD, of the University of Plymouth, England. He noted that there is currently no consensus regarding best treatment strategy for this patient population.
Dr. Rule said that he and his colleagues have collected data on 30 patients so far, of which 22 were included in this early data release.
All patients received R-BAC between 2016 and 2018 at treatment centers in Italy and the United Kingdom. Treatment consisted of rituximab (375 mg/m2 or 500 mg) on day 1, bendamustine 70 mg/m2 on days 1 and 2, and cytarabine 500 mg/m2 on days 1 through 3, given in a 28-day cycle.
Patients received R-BAC immediately after BTKi failure. Data were drawn from hospital records.
Analysis showed that the median patient age was 65 years, with a range from 43 to 79 years. Most patients were men (81.8%), 55.0% were high risk based on the Mantle Cell Lymphoma International Prognostic Index, and 22.7% had blastoid morphology.
Patients had a median of two prior systemic therapies, with a range from one to six lines. First-line therapies included rituximab in combination with HDAC (high-dose cytarabine containing regimen), CHOP, CVP, or ibrutinib. Nine patients (42.9%) had allogeneic stem cell transplantation (ASCT) after induction treatment.
For BTKi therapy, most patients received ibrutinib (n = 18), while the remainder received acalabrutinib, tirabrutinib or M7583. Most patients discontinued BTKi therapy because of disease progression (90.9%); two patients stopped because of a lack of response (9.1%).
The median number of R-BAC cycles received was four. Two patients started with attenuated doses and seven patients reduced doses after the first cycle. More than 70% of patients completed R-BAC treatment.
The estimated median progression-free survival was 7.3 months and estimated median overall survival was 11.2 months.
Although the investigators reported a complete response rate of 57.1%, they noted that this figure “may be exaggerated” because of a lack of bone marrow biopsy; however, they suggested that the overall response rate (90.5%) “should be accurate.”
During the course of treatment, 31.8% of patients required inpatient admission, 22.7% developed neutropenic fever, and 77.8% required transfusion support. No treatment-related deaths occurred.
“This population, enriched for patients with high risk features, showed remarkable response rates to R-BAC,” the investigators wrote. “The treatment had acceptable toxicity, maintained efficacy at attenuated doses, and was used successfully as a bridge to ASCT in over 20% of patients.”
The investigators suggested that R-BAC should be considered a new standard of care in the United Kingdom for bendamustine-naive patients who are unable to be enrolled in clinical trials. “The high response rate makes it particularly appealing for patients considered candidates for consolidation ASCT,” they wrote.
In an interview, Dr. Rule added perspective to these findings.
“There’s been an obsession with venetoclax, that that’s the answer, but it really isn’t,” Dr. Rule said. “So people are looking for a new drug. I guess what I do differently to most people is I use CHOP frontline rather than bendamustine. To me, that’s the best way of sequencing the therapies, whereas if you use [bendamustine and rituximab] up front, which a lot of people do, particularly in the [United] States, your R-BAC might not be so effective.”
However, Dr. Rule said that first-line therapies appear to have minimal impact on R-BAC efficacy. “Even if you’ve had bendamustine, even if you’ve had high-dose cytarabine, even if you’ve had an allogeneic stem cell transplant, [R-BAC] still works,” he said.
Where patients have issues with tolerability, Dr. Rule noted that dose reductions are possible without sacrificing efficacy.
He offered an example of such a scenario. “My oldest patient was about 80 with blastoid disease, relapsing,” Dr. Rule said. “After ibrutinib, I gave him just a single dose of bendamustine at 70 mg, a single dose of cytarabine at 500 mg, just 1 day, and he had that six times, probably 3 weeks apart. He’s been in complete remission for over a year.”
With data on 30 patients collected, Dr. Rule said that he and his colleagues plan to present more extensive findings at the European Hematology Association Congress, held June 13-16 in Amsterdam.
The investigators reported having no conflicts of interest.
GLASGOW – Patients with relapsed or refractory mantle cell lymphoma (MCL) who experience disease progression on a Bruton’s tyrosine kinase inhibitor (BTKi) may respond best to a combination of rituximab, bendamustine, and cytarabine (R-BAC), based on early results from an ongoing retrospective study.
Findings from the study, which were presented at the annual meeting of the British Society for Haematology, showed that R-BAC after BTKi failure had an overall response rate (ORR) of 90.5%.
This is a “remarkable response rate” according to the investigators, who cited previously reported response rates for other treatments ranging from 29% to 53%.
Treatment of relapsed/refractory MCL patients in the post-BTKi setting is an area of unmet clinical need, said senior author Simon Rule, MD, of the University of Plymouth, England. He noted that there is currently no consensus regarding best treatment strategy for this patient population.
Dr. Rule said that he and his colleagues have collected data on 30 patients so far, of which 22 were included in this early data release.
All patients received R-BAC between 2016 and 2018 at treatment centers in Italy and the United Kingdom. Treatment consisted of rituximab (375 mg/m2 or 500 mg) on day 1, bendamustine 70 mg/m2 on days 1 and 2, and cytarabine 500 mg/m2 on days 1 through 3, given in a 28-day cycle.
Patients received R-BAC immediately after BTKi failure. Data were drawn from hospital records.
Analysis showed that the median patient age was 65 years, with a range from 43 to 79 years. Most patients were men (81.8%), 55.0% were high risk based on the Mantle Cell Lymphoma International Prognostic Index, and 22.7% had blastoid morphology.
Patients had a median of two prior systemic therapies, with a range from one to six lines. First-line therapies included rituximab in combination with HDAC (high-dose cytarabine containing regimen), CHOP, CVP, or ibrutinib. Nine patients (42.9%) had allogeneic stem cell transplantation (ASCT) after induction treatment.
For BTKi therapy, most patients received ibrutinib (n = 18), while the remainder received acalabrutinib, tirabrutinib or M7583. Most patients discontinued BTKi therapy because of disease progression (90.9%); two patients stopped because of a lack of response (9.1%).
The median number of R-BAC cycles received was four. Two patients started with attenuated doses and seven patients reduced doses after the first cycle. More than 70% of patients completed R-BAC treatment.
The estimated median progression-free survival was 7.3 months and estimated median overall survival was 11.2 months.
Although the investigators reported a complete response rate of 57.1%, they noted that this figure “may be exaggerated” because of a lack of bone marrow biopsy; however, they suggested that the overall response rate (90.5%) “should be accurate.”
During the course of treatment, 31.8% of patients required inpatient admission, 22.7% developed neutropenic fever, and 77.8% required transfusion support. No treatment-related deaths occurred.
“This population, enriched for patients with high risk features, showed remarkable response rates to R-BAC,” the investigators wrote. “The treatment had acceptable toxicity, maintained efficacy at attenuated doses, and was used successfully as a bridge to ASCT in over 20% of patients.”
The investigators suggested that R-BAC should be considered a new standard of care in the United Kingdom for bendamustine-naive patients who are unable to be enrolled in clinical trials. “The high response rate makes it particularly appealing for patients considered candidates for consolidation ASCT,” they wrote.
In an interview, Dr. Rule added perspective to these findings.
“There’s been an obsession with venetoclax, that that’s the answer, but it really isn’t,” Dr. Rule said. “So people are looking for a new drug. I guess what I do differently to most people is I use CHOP frontline rather than bendamustine. To me, that’s the best way of sequencing the therapies, whereas if you use [bendamustine and rituximab] up front, which a lot of people do, particularly in the [United] States, your R-BAC might not be so effective.”
However, Dr. Rule said that first-line therapies appear to have minimal impact on R-BAC efficacy. “Even if you’ve had bendamustine, even if you’ve had high-dose cytarabine, even if you’ve had an allogeneic stem cell transplant, [R-BAC] still works,” he said.
Where patients have issues with tolerability, Dr. Rule noted that dose reductions are possible without sacrificing efficacy.
He offered an example of such a scenario. “My oldest patient was about 80 with blastoid disease, relapsing,” Dr. Rule said. “After ibrutinib, I gave him just a single dose of bendamustine at 70 mg, a single dose of cytarabine at 500 mg, just 1 day, and he had that six times, probably 3 weeks apart. He’s been in complete remission for over a year.”
With data on 30 patients collected, Dr. Rule said that he and his colleagues plan to present more extensive findings at the European Hematology Association Congress, held June 13-16 in Amsterdam.
The investigators reported having no conflicts of interest.
REPORTING FROM BSH 2019
Polyglutamine diseases are rare, but not the mutations that cause them
Polyglutamine diseases are a group of hereditary neurodegenerative disorders caused by mutations in which a trinucleotide repeat expands pathologically on a disease-associated gene. The diseases are rare, with the most common among them – Huntington disease – affecting between 10 and 14 per 100,000 people in Western countries, where prevalence is highest.
In polyglutamine diseases, which include the spinocerebellar ataxias, dentatorubral-pallidoluysian atrophy, and spinal bulbar muscular atrophy, higher CAG (cytosine-adenine-guanine) repeat numbers are associated with greater disease severity, faster progression, or earlier age at onset.
In research published April 1 in JAMA Neurology, investigators report that more than one-tenth of the European population carries CAG expansions that fall short of the repeats needed to cause any of 9 polyglutamine diseases – but are enough to put them at risk of having children who develop one. A smaller number of people – about 1% – carry enough CAG repeats to cause one of the diseases late in life.
For their research, Sarah L. Gardiner, MD, of Leiden (the Netherlands) University, and her colleagues looked at polyglutamine expansion variants for nine diseases in samples from 14,196 adults (56% of whom were women) from the Netherlands, Scotland, and Ireland. The samples were taken from five population-based cohort studies conducted between 1997 and 2012, and all subjects were without a history of polyglutamine disease or major depression.
Of these, 10.7% had a CAG repeat number on a disease-associated gene that was in the intermediate range, defined as a number of repeats that cannot cause disease but for which “expansion into the fully pathological range has been observed on intergenerational transmission,” Dr. Gardiner and her colleagues wrote. And some 1.3% of subjects were found to have CAG repeats within the disease-causing range, “mostly in the lower range associated with elderly onset.”
The investigators found no differences in sex, age, or body mass index between individuals with CAG repeat numbers within the pathological range and individuals with CAG repeat numbers within the normal or intermediate range.
Whether carriers of immediate or lower-range pathological CAG repeats went on to develop disease could not be measured, as follow-up data were not available. Another limitation of the study, the investigators acknowledged, was that the genotyping method used “did not allow us to determine the presence of trinucleotide interruptions,” which can affect disease penetrance.
“A late age at onset, a reduced penetrance, or the presence of interruptions could all explain the asymptomatic status of our carriers of intermediate and pathological polyglutamine disease–associated alleles at the time of assessment,” Dr. Gardiner and her colleagues wrote.
This study was funded by the European Union and Dutch government agencies; one of the population-based cohort studies from which the study sample was taken received some support from Bristol-Myers-Squibb. One of Dr. Gardiner’s coauthors, Raymund A. C. Roos, MD, PhD, disclosed being an adviser for UniQure, a gene-therapy firm, and no other conflicts of interest were reported.
SOURCE: Gardiner et al. JAMA Neurol. 2019 Apr 1. doi: 10.001/jamaneurol.2019.042.
Gardiner et al. describe the results of an appraisal of polyglutamine expansion variants in more than 14,000 individuals from the Netherlands, Scotland, and Ireland. Given the relative rarity of polyglutamine repeat disease, the first question that comes to mind is why were so many individuals identified with repeats in the pathogenic range? Based on our understanding of disease prevalence, it is unlikely that each of these individuals will become affected; therefore, this work suggests a reduced penetrance of these mutations. The findings are illustrative of a growing theme in human disease genetics: There are a very large number of apparently healthy individuals in the general population who carry mutations associated with various diseases. The phenomenon of reduced penetrance, where mutations cause disease in some but not all carriers, overlaps and arguably may be the same as that of variable expressivity, where the same mutation can lead to very different disease outcomes in different individuals. It is extremely likely that second-generation sequencing and population-scale screening will continue to reveal similar themes. We continue to appreciate the increasing complexity of the human genome and its relationship to disease, even those diseases we thought of previously as simple “single-gene” disorders.
Monia B. Hammer, PhD, and Andrew B. Singleton, PhD, are with the National Institute on Aging, National Institutes of Health, Bethesda, Md. Dr. Hammer and Dr. Singleton report no financial conflicts of interest related to their editorial.
Gardiner et al. describe the results of an appraisal of polyglutamine expansion variants in more than 14,000 individuals from the Netherlands, Scotland, and Ireland. Given the relative rarity of polyglutamine repeat disease, the first question that comes to mind is why were so many individuals identified with repeats in the pathogenic range? Based on our understanding of disease prevalence, it is unlikely that each of these individuals will become affected; therefore, this work suggests a reduced penetrance of these mutations. The findings are illustrative of a growing theme in human disease genetics: There are a very large number of apparently healthy individuals in the general population who carry mutations associated with various diseases. The phenomenon of reduced penetrance, where mutations cause disease in some but not all carriers, overlaps and arguably may be the same as that of variable expressivity, where the same mutation can lead to very different disease outcomes in different individuals. It is extremely likely that second-generation sequencing and population-scale screening will continue to reveal similar themes. We continue to appreciate the increasing complexity of the human genome and its relationship to disease, even those diseases we thought of previously as simple “single-gene” disorders.
Monia B. Hammer, PhD, and Andrew B. Singleton, PhD, are with the National Institute on Aging, National Institutes of Health, Bethesda, Md. Dr. Hammer and Dr. Singleton report no financial conflicts of interest related to their editorial.
Gardiner et al. describe the results of an appraisal of polyglutamine expansion variants in more than 14,000 individuals from the Netherlands, Scotland, and Ireland. Given the relative rarity of polyglutamine repeat disease, the first question that comes to mind is why were so many individuals identified with repeats in the pathogenic range? Based on our understanding of disease prevalence, it is unlikely that each of these individuals will become affected; therefore, this work suggests a reduced penetrance of these mutations. The findings are illustrative of a growing theme in human disease genetics: There are a very large number of apparently healthy individuals in the general population who carry mutations associated with various diseases. The phenomenon of reduced penetrance, where mutations cause disease in some but not all carriers, overlaps and arguably may be the same as that of variable expressivity, where the same mutation can lead to very different disease outcomes in different individuals. It is extremely likely that second-generation sequencing and population-scale screening will continue to reveal similar themes. We continue to appreciate the increasing complexity of the human genome and its relationship to disease, even those diseases we thought of previously as simple “single-gene” disorders.
Monia B. Hammer, PhD, and Andrew B. Singleton, PhD, are with the National Institute on Aging, National Institutes of Health, Bethesda, Md. Dr. Hammer and Dr. Singleton report no financial conflicts of interest related to their editorial.
Polyglutamine diseases are a group of hereditary neurodegenerative disorders caused by mutations in which a trinucleotide repeat expands pathologically on a disease-associated gene. The diseases are rare, with the most common among them – Huntington disease – affecting between 10 and 14 per 100,000 people in Western countries, where prevalence is highest.
In polyglutamine diseases, which include the spinocerebellar ataxias, dentatorubral-pallidoluysian atrophy, and spinal bulbar muscular atrophy, higher CAG (cytosine-adenine-guanine) repeat numbers are associated with greater disease severity, faster progression, or earlier age at onset.
In research published April 1 in JAMA Neurology, investigators report that more than one-tenth of the European population carries CAG expansions that fall short of the repeats needed to cause any of 9 polyglutamine diseases – but are enough to put them at risk of having children who develop one. A smaller number of people – about 1% – carry enough CAG repeats to cause one of the diseases late in life.
For their research, Sarah L. Gardiner, MD, of Leiden (the Netherlands) University, and her colleagues looked at polyglutamine expansion variants for nine diseases in samples from 14,196 adults (56% of whom were women) from the Netherlands, Scotland, and Ireland. The samples were taken from five population-based cohort studies conducted between 1997 and 2012, and all subjects were without a history of polyglutamine disease or major depression.
Of these, 10.7% had a CAG repeat number on a disease-associated gene that was in the intermediate range, defined as a number of repeats that cannot cause disease but for which “expansion into the fully pathological range has been observed on intergenerational transmission,” Dr. Gardiner and her colleagues wrote. And some 1.3% of subjects were found to have CAG repeats within the disease-causing range, “mostly in the lower range associated with elderly onset.”
The investigators found no differences in sex, age, or body mass index between individuals with CAG repeat numbers within the pathological range and individuals with CAG repeat numbers within the normal or intermediate range.
Whether carriers of immediate or lower-range pathological CAG repeats went on to develop disease could not be measured, as follow-up data were not available. Another limitation of the study, the investigators acknowledged, was that the genotyping method used “did not allow us to determine the presence of trinucleotide interruptions,” which can affect disease penetrance.
“A late age at onset, a reduced penetrance, or the presence of interruptions could all explain the asymptomatic status of our carriers of intermediate and pathological polyglutamine disease–associated alleles at the time of assessment,” Dr. Gardiner and her colleagues wrote.
This study was funded by the European Union and Dutch government agencies; one of the population-based cohort studies from which the study sample was taken received some support from Bristol-Myers-Squibb. One of Dr. Gardiner’s coauthors, Raymund A. C. Roos, MD, PhD, disclosed being an adviser for UniQure, a gene-therapy firm, and no other conflicts of interest were reported.
SOURCE: Gardiner et al. JAMA Neurol. 2019 Apr 1. doi: 10.001/jamaneurol.2019.042.
Polyglutamine diseases are a group of hereditary neurodegenerative disorders caused by mutations in which a trinucleotide repeat expands pathologically on a disease-associated gene. The diseases are rare, with the most common among them – Huntington disease – affecting between 10 and 14 per 100,000 people in Western countries, where prevalence is highest.
In polyglutamine diseases, which include the spinocerebellar ataxias, dentatorubral-pallidoluysian atrophy, and spinal bulbar muscular atrophy, higher CAG (cytosine-adenine-guanine) repeat numbers are associated with greater disease severity, faster progression, or earlier age at onset.
In research published April 1 in JAMA Neurology, investigators report that more than one-tenth of the European population carries CAG expansions that fall short of the repeats needed to cause any of 9 polyglutamine diseases – but are enough to put them at risk of having children who develop one. A smaller number of people – about 1% – carry enough CAG repeats to cause one of the diseases late in life.
For their research, Sarah L. Gardiner, MD, of Leiden (the Netherlands) University, and her colleagues looked at polyglutamine expansion variants for nine diseases in samples from 14,196 adults (56% of whom were women) from the Netherlands, Scotland, and Ireland. The samples were taken from five population-based cohort studies conducted between 1997 and 2012, and all subjects were without a history of polyglutamine disease or major depression.
Of these, 10.7% had a CAG repeat number on a disease-associated gene that was in the intermediate range, defined as a number of repeats that cannot cause disease but for which “expansion into the fully pathological range has been observed on intergenerational transmission,” Dr. Gardiner and her colleagues wrote. And some 1.3% of subjects were found to have CAG repeats within the disease-causing range, “mostly in the lower range associated with elderly onset.”
The investigators found no differences in sex, age, or body mass index between individuals with CAG repeat numbers within the pathological range and individuals with CAG repeat numbers within the normal or intermediate range.
Whether carriers of immediate or lower-range pathological CAG repeats went on to develop disease could not be measured, as follow-up data were not available. Another limitation of the study, the investigators acknowledged, was that the genotyping method used “did not allow us to determine the presence of trinucleotide interruptions,” which can affect disease penetrance.
“A late age at onset, a reduced penetrance, or the presence of interruptions could all explain the asymptomatic status of our carriers of intermediate and pathological polyglutamine disease–associated alleles at the time of assessment,” Dr. Gardiner and her colleagues wrote.
This study was funded by the European Union and Dutch government agencies; one of the population-based cohort studies from which the study sample was taken received some support from Bristol-Myers-Squibb. One of Dr. Gardiner’s coauthors, Raymund A. C. Roos, MD, PhD, disclosed being an adviser for UniQure, a gene-therapy firm, and no other conflicts of interest were reported.
SOURCE: Gardiner et al. JAMA Neurol. 2019 Apr 1. doi: 10.001/jamaneurol.2019.042.
FROM JAMA NEUROLOGY
Marcela Romero-Reyes, DDS, PhD, Comments on Peripheral and Central Headache Challenges
###
Dr. Rapoport: Do you commonly see patients who present with symptoms of both central and peripheral symptoms in practice?
Dr. Romero-Reyes: Yes, I see patients that present with temporomandibular disorders (TMD) and headache comorbidity, as well as patients with migraine, tension-type headache, and cervicogenic headache with myofascial pain.
Dr. Rapoport: Why do you think this condition is so challenging to treat?
Dr. Romero-Reyes: I think this is because of the lack of understanding and awareness that in addition to the multifactorial nature of headache disorders, other types of disorders that are not neurovascular in origin may influence trigeminovascular nociception, and these types of non-neurovascular disorders involve the skill and knowledge of other expertise.
Headaches receiving inputs from extracranial structures such as in TMD (temporomandibular joint [TMJ] and muscles of mastication) and/or cervical structures (cervical spine, cervical muscles) require multidisciplinary evaluation and management. In these cases, the management should involve a neurologist specialized in headache disorders, a dentist trained in TMD and orofacial pain disorders, and a physical therapist with special training in craniofacial and cervical Therapeutics. Multidisciplinary communication is key for successful management.
Another reason is that myofascial pain (MFP) is often overlooked in patients with headache disorders. In my experience, patients with episodic and chronic migraine, episodic and chronic tension-type headache, cervicogenic headache, and patients presenting TMD and headache comorbidity can present trigger points in the craniofacial and cervical muscles, an indication of MFP. It has been reported that these patients present a higher disability impact. The presence of MFP may be contributing to the activation of the trigeminovascular system and therefore facilitate, exacerbate, and perpetuate headache symptomatology and may accelerate the progression to a more chronic form of the disorder.
Dr. Rapoport: In your opinion, is this considered a controversial topic? Why or why not?
Dr. Romero-Reyes: Yes, I think it is necessary to clarify that tenderness in the back of the head or of neck muscles present in headache patients does not necessarily imply that it is due to a nerve compression. This could also be caused by local myalgia but more commonly, from latent or active myofascial trigger points present in the muscles of the area being palpated, or by referred pain beyond the area of the muscle being palpated. Suboccipital muscles (in the occiput area) are not the only muscle group that is associated with headache and neck pain symptomatology. For example, the trapezius muscle, which is an overlooked source of tension- type and cervicogenic headache, can present trigger points that can refer pain to the shoulder, neck, head, face and the eye. In addition, other craniofacial and cervical muscles such as the sternocleidomastoid (SCM) and temporalis muscles have been shown to be associated with headache symptomatology in the migraineur, as well as the chronic tension-type headache patient. Other muscles that also refer to the craniofacial area and can elicit headache and neck pain symptomatology include the masseter, occipitofrontalis, splenius capitis, splenius cervicis, semispinalis capitis, semispinalis cervicis and multifidi (cervical). The presence of trigger points in these muscles do not support or warrant the need to be removed or managed with non-conservative approaches.
Myofascial trigger points can result from muscle injury and overload, parafunctional activity, and poor head and neck posture. MFP is characterized by a regional pain and presence of localized tender areas (trigger points) in muscle, fascia or tendons that reproduce pain when palpated, and produce a pattern of regional pain spreading along the muscle palpated, or beyond the location boundary of the muscle palpated. It has been shown by microdyalisis that inflammatory mediators and neuropeptides are present in the area of an active trigger point. In addition, an increase of electromyography activity has been shown in trigger points in patients with chronic tension-type headache when compared with controls.
The importance of an evaluation by a skilled clinician in the craniofacial and cervical area to verify the source of pain is critical. The patient may be reporting pain in one area, but the source of the pain is in another area, and this is typical symptomatology present when there are active trigger points. In addition, an assessment of any contributing factors arising from the cervical spine (eg, poor posture) and craniofacial area (eg, TMD) that may exacerbate headache symptomatology is vital to proper diagnosis.
In my experience, patients with migraine, tension-type headache, cervicogenic headache, and TMD and headache comorbidity present MFP perpetuating headache symptomatology. MFP is not managed by surgical interventions. This perpetuating factor can be managed effectively with conservative measures. The plan is tailored for each patient’s needs. In general, the plan of management may include trigger point injections in the muscle with anesthetics, dry needling, and a physical therapy plan that may include education regarding habits and posture, exercises and physical therapy modalities, which are crucial to relieve pain and increase function. In cases of TMD and headache comorbidity, an occlusal appliance (stabilization appliance) can be included if necessary. We should also consider behavioral therapies (especially EMG biofeedback training) and some oral anti-inflammatories or muscle relaxants in the beginning of management, together with the plan of management mentioned above.
With these approaches to manage the MFP component in headache patients, I have been able to see that in migraineurs with MFP, the frequency and severity of the attacks decrease significantly. The patient may still experience migraine attacks, but feel happy to have the possibility to reduce medication intake and be in more control of their pain. In patients with tension-type headache, I have seen this even more dramatically.
This is telling us that headache pathophysiology involves a “conversation” between the peripheral and central nervous system, which influence each other. Peripheral nociceptive input coming from extracranial structures can induce trigeminovascular activation and therefore exacerbate a headache disorder and vice versa. Chronic myofascial pain may be the result of central sensitization due to the protracted peripheral nociceptive input (eg, poor posture, neck strain, parafunctional activity), therefore perpetuating the headache disorder even more.
Dr. Rapoport: Do you have any other comments about the article Treatment Challenges When Headache Has Central and Peripheral Involvement that you would like to share with our readers?
Dr. Romero-Reyes: It is simplistic to say migraine is either a peripheral or a central disorder, or that symptoms are either peripheral or central. Beyond thinking about migraine pain, migraine is fundamentally a brain (central) disorder. Its associated symptoms (nausea, phonophobia, photophobia) tell us this. Migraine headache is complex, and most likely the result of central mechanisms that can be influenced by peripheral inputs from the craniofacial and cervical region.
Embarking on surgical interventions for the management of headache disorders warrants a caution since it is still an experimental research question and the need of such therapies should be evaluated against conservative management. We are in a very exciting and hopeful time for migraine management. New evidence-based options from biological agents, such as anti-calcitonin gene-related peptide (CGRP) therapies, to non-pharmacological approaches, such as neuromodulation, can be offered to the patients. If the patient is experiencing pain in the neck area or other craniofacial area, it is recommended to have a thorough evaluation by a physical therapist with special training in cervical and craniofacial therapeutics and/or a dentist trained in TMD and orofacial pain disorders to work in consultation with a neurologist to elaborate a personalized management plan. Do not overlook the contribution of myofascial pain (trigger points) as well as TMD in the symptomatology of headache disorders. Few patients need to undergo surgical measures of peripheral nerves and muscles for improvement. An exhaustive evaluation must be undertaken first.
Resources for patients:
AHS
https://americanheadachesociety.org/
https://americanheadachesociety.org/wp-content/uploads/2018/06/Choosing-Wisely-Flyer.pdf
AAOP
https://aaop.clubexpress.com/content.aspx?sl=1152088466
PTBCTT
###
Dr. Rapoport: Do you commonly see patients who present with symptoms of both central and peripheral symptoms in practice?
Dr. Romero-Reyes: Yes, I see patients that present with temporomandibular disorders (TMD) and headache comorbidity, as well as patients with migraine, tension-type headache, and cervicogenic headache with myofascial pain.
Dr. Rapoport: Why do you think this condition is so challenging to treat?
Dr. Romero-Reyes: I think this is because of the lack of understanding and awareness that in addition to the multifactorial nature of headache disorders, other types of disorders that are not neurovascular in origin may influence trigeminovascular nociception, and these types of non-neurovascular disorders involve the skill and knowledge of other expertise.
Headaches receiving inputs from extracranial structures such as in TMD (temporomandibular joint [TMJ] and muscles of mastication) and/or cervical structures (cervical spine, cervical muscles) require multidisciplinary evaluation and management. In these cases, the management should involve a neurologist specialized in headache disorders, a dentist trained in TMD and orofacial pain disorders, and a physical therapist with special training in craniofacial and cervical Therapeutics. Multidisciplinary communication is key for successful management.
Another reason is that myofascial pain (MFP) is often overlooked in patients with headache disorders. In my experience, patients with episodic and chronic migraine, episodic and chronic tension-type headache, cervicogenic headache, and patients presenting TMD and headache comorbidity can present trigger points in the craniofacial and cervical muscles, an indication of MFP. It has been reported that these patients present a higher disability impact. The presence of MFP may be contributing to the activation of the trigeminovascular system and therefore facilitate, exacerbate, and perpetuate headache symptomatology and may accelerate the progression to a more chronic form of the disorder.
Dr. Rapoport: In your opinion, is this considered a controversial topic? Why or why not?
Dr. Romero-Reyes: Yes, I think it is necessary to clarify that tenderness in the back of the head or of neck muscles present in headache patients does not necessarily imply that it is due to a nerve compression. This could also be caused by local myalgia but more commonly, from latent or active myofascial trigger points present in the muscles of the area being palpated, or by referred pain beyond the area of the muscle being palpated. Suboccipital muscles (in the occiput area) are not the only muscle group that is associated with headache and neck pain symptomatology. For example, the trapezius muscle, which is an overlooked source of tension- type and cervicogenic headache, can present trigger points that can refer pain to the shoulder, neck, head, face and the eye. In addition, other craniofacial and cervical muscles such as the sternocleidomastoid (SCM) and temporalis muscles have been shown to be associated with headache symptomatology in the migraineur, as well as the chronic tension-type headache patient. Other muscles that also refer to the craniofacial area and can elicit headache and neck pain symptomatology include the masseter, occipitofrontalis, splenius capitis, splenius cervicis, semispinalis capitis, semispinalis cervicis and multifidi (cervical). The presence of trigger points in these muscles do not support or warrant the need to be removed or managed with non-conservative approaches.
Myofascial trigger points can result from muscle injury and overload, parafunctional activity, and poor head and neck posture. MFP is characterized by a regional pain and presence of localized tender areas (trigger points) in muscle, fascia or tendons that reproduce pain when palpated, and produce a pattern of regional pain spreading along the muscle palpated, or beyond the location boundary of the muscle palpated. It has been shown by microdyalisis that inflammatory mediators and neuropeptides are present in the area of an active trigger point. In addition, an increase of electromyography activity has been shown in trigger points in patients with chronic tension-type headache when compared with controls.
The importance of an evaluation by a skilled clinician in the craniofacial and cervical area to verify the source of pain is critical. The patient may be reporting pain in one area, but the source of the pain is in another area, and this is typical symptomatology present when there are active trigger points. In addition, an assessment of any contributing factors arising from the cervical spine (eg, poor posture) and craniofacial area (eg, TMD) that may exacerbate headache symptomatology is vital to proper diagnosis.
In my experience, patients with migraine, tension-type headache, cervicogenic headache, and TMD and headache comorbidity present MFP perpetuating headache symptomatology. MFP is not managed by surgical interventions. This perpetuating factor can be managed effectively with conservative measures. The plan is tailored for each patient’s needs. In general, the plan of management may include trigger point injections in the muscle with anesthetics, dry needling, and a physical therapy plan that may include education regarding habits and posture, exercises and physical therapy modalities, which are crucial to relieve pain and increase function. In cases of TMD and headache comorbidity, an occlusal appliance (stabilization appliance) can be included if necessary. We should also consider behavioral therapies (especially EMG biofeedback training) and some oral anti-inflammatories or muscle relaxants in the beginning of management, together with the plan of management mentioned above.
With these approaches to manage the MFP component in headache patients, I have been able to see that in migraineurs with MFP, the frequency and severity of the attacks decrease significantly. The patient may still experience migraine attacks, but feel happy to have the possibility to reduce medication intake and be in more control of their pain. In patients with tension-type headache, I have seen this even more dramatically.
This is telling us that headache pathophysiology involves a “conversation” between the peripheral and central nervous system, which influence each other. Peripheral nociceptive input coming from extracranial structures can induce trigeminovascular activation and therefore exacerbate a headache disorder and vice versa. Chronic myofascial pain may be the result of central sensitization due to the protracted peripheral nociceptive input (eg, poor posture, neck strain, parafunctional activity), therefore perpetuating the headache disorder even more.
Dr. Rapoport: Do you have any other comments about the article Treatment Challenges When Headache Has Central and Peripheral Involvement that you would like to share with our readers?
Dr. Romero-Reyes: It is simplistic to say migraine is either a peripheral or a central disorder, or that symptoms are either peripheral or central. Beyond thinking about migraine pain, migraine is fundamentally a brain (central) disorder. Its associated symptoms (nausea, phonophobia, photophobia) tell us this. Migraine headache is complex, and most likely the result of central mechanisms that can be influenced by peripheral inputs from the craniofacial and cervical region.
Embarking on surgical interventions for the management of headache disorders warrants a caution since it is still an experimental research question and the need of such therapies should be evaluated against conservative management. We are in a very exciting and hopeful time for migraine management. New evidence-based options from biological agents, such as anti-calcitonin gene-related peptide (CGRP) therapies, to non-pharmacological approaches, such as neuromodulation, can be offered to the patients. If the patient is experiencing pain in the neck area or other craniofacial area, it is recommended to have a thorough evaluation by a physical therapist with special training in cervical and craniofacial therapeutics and/or a dentist trained in TMD and orofacial pain disorders to work in consultation with a neurologist to elaborate a personalized management plan. Do not overlook the contribution of myofascial pain (trigger points) as well as TMD in the symptomatology of headache disorders. Few patients need to undergo surgical measures of peripheral nerves and muscles for improvement. An exhaustive evaluation must be undertaken first.
Resources for patients:
AHS
https://americanheadachesociety.org/
https://americanheadachesociety.org/wp-content/uploads/2018/06/Choosing-Wisely-Flyer.pdf
AAOP
https://aaop.clubexpress.com/content.aspx?sl=1152088466
PTBCTT
###
Dr. Rapoport: Do you commonly see patients who present with symptoms of both central and peripheral symptoms in practice?
Dr. Romero-Reyes: Yes, I see patients that present with temporomandibular disorders (TMD) and headache comorbidity, as well as patients with migraine, tension-type headache, and cervicogenic headache with myofascial pain.
Dr. Rapoport: Why do you think this condition is so challenging to treat?
Dr. Romero-Reyes: I think this is because of the lack of understanding and awareness that in addition to the multifactorial nature of headache disorders, other types of disorders that are not neurovascular in origin may influence trigeminovascular nociception, and these types of non-neurovascular disorders involve the skill and knowledge of other expertise.
Headaches receiving inputs from extracranial structures such as in TMD (temporomandibular joint [TMJ] and muscles of mastication) and/or cervical structures (cervical spine, cervical muscles) require multidisciplinary evaluation and management. In these cases, the management should involve a neurologist specialized in headache disorders, a dentist trained in TMD and orofacial pain disorders, and a physical therapist with special training in craniofacial and cervical Therapeutics. Multidisciplinary communication is key for successful management.
Another reason is that myofascial pain (MFP) is often overlooked in patients with headache disorders. In my experience, patients with episodic and chronic migraine, episodic and chronic tension-type headache, cervicogenic headache, and patients presenting TMD and headache comorbidity can present trigger points in the craniofacial and cervical muscles, an indication of MFP. It has been reported that these patients present a higher disability impact. The presence of MFP may be contributing to the activation of the trigeminovascular system and therefore facilitate, exacerbate, and perpetuate headache symptomatology and may accelerate the progression to a more chronic form of the disorder.
Dr. Rapoport: In your opinion, is this considered a controversial topic? Why or why not?
Dr. Romero-Reyes: Yes, I think it is necessary to clarify that tenderness in the back of the head or of neck muscles present in headache patients does not necessarily imply that it is due to a nerve compression. This could also be caused by local myalgia but more commonly, from latent or active myofascial trigger points present in the muscles of the area being palpated, or by referred pain beyond the area of the muscle being palpated. Suboccipital muscles (in the occiput area) are not the only muscle group that is associated with headache and neck pain symptomatology. For example, the trapezius muscle, which is an overlooked source of tension- type and cervicogenic headache, can present trigger points that can refer pain to the shoulder, neck, head, face and the eye. In addition, other craniofacial and cervical muscles such as the sternocleidomastoid (SCM) and temporalis muscles have been shown to be associated with headache symptomatology in the migraineur, as well as the chronic tension-type headache patient. Other muscles that also refer to the craniofacial area and can elicit headache and neck pain symptomatology include the masseter, occipitofrontalis, splenius capitis, splenius cervicis, semispinalis capitis, semispinalis cervicis and multifidi (cervical). The presence of trigger points in these muscles do not support or warrant the need to be removed or managed with non-conservative approaches.
Myofascial trigger points can result from muscle injury and overload, parafunctional activity, and poor head and neck posture. MFP is characterized by a regional pain and presence of localized tender areas (trigger points) in muscle, fascia or tendons that reproduce pain when palpated, and produce a pattern of regional pain spreading along the muscle palpated, or beyond the location boundary of the muscle palpated. It has been shown by microdyalisis that inflammatory mediators and neuropeptides are present in the area of an active trigger point. In addition, an increase of electromyography activity has been shown in trigger points in patients with chronic tension-type headache when compared with controls.
The importance of an evaluation by a skilled clinician in the craniofacial and cervical area to verify the source of pain is critical. The patient may be reporting pain in one area, but the source of the pain is in another area, and this is typical symptomatology present when there are active trigger points. In addition, an assessment of any contributing factors arising from the cervical spine (eg, poor posture) and craniofacial area (eg, TMD) that may exacerbate headache symptomatology is vital to proper diagnosis.
In my experience, patients with migraine, tension-type headache, cervicogenic headache, and TMD and headache comorbidity present MFP perpetuating headache symptomatology. MFP is not managed by surgical interventions. This perpetuating factor can be managed effectively with conservative measures. The plan is tailored for each patient’s needs. In general, the plan of management may include trigger point injections in the muscle with anesthetics, dry needling, and a physical therapy plan that may include education regarding habits and posture, exercises and physical therapy modalities, which are crucial to relieve pain and increase function. In cases of TMD and headache comorbidity, an occlusal appliance (stabilization appliance) can be included if necessary. We should also consider behavioral therapies (especially EMG biofeedback training) and some oral anti-inflammatories or muscle relaxants in the beginning of management, together with the plan of management mentioned above.
With these approaches to manage the MFP component in headache patients, I have been able to see that in migraineurs with MFP, the frequency and severity of the attacks decrease significantly. The patient may still experience migraine attacks, but feel happy to have the possibility to reduce medication intake and be in more control of their pain. In patients with tension-type headache, I have seen this even more dramatically.
This is telling us that headache pathophysiology involves a “conversation” between the peripheral and central nervous system, which influence each other. Peripheral nociceptive input coming from extracranial structures can induce trigeminovascular activation and therefore exacerbate a headache disorder and vice versa. Chronic myofascial pain may be the result of central sensitization due to the protracted peripheral nociceptive input (eg, poor posture, neck strain, parafunctional activity), therefore perpetuating the headache disorder even more.
Dr. Rapoport: Do you have any other comments about the article Treatment Challenges When Headache Has Central and Peripheral Involvement that you would like to share with our readers?
Dr. Romero-Reyes: It is simplistic to say migraine is either a peripheral or a central disorder, or that symptoms are either peripheral or central. Beyond thinking about migraine pain, migraine is fundamentally a brain (central) disorder. Its associated symptoms (nausea, phonophobia, photophobia) tell us this. Migraine headache is complex, and most likely the result of central mechanisms that can be influenced by peripheral inputs from the craniofacial and cervical region.
Embarking on surgical interventions for the management of headache disorders warrants a caution since it is still an experimental research question and the need of such therapies should be evaluated against conservative management. We are in a very exciting and hopeful time for migraine management. New evidence-based options from biological agents, such as anti-calcitonin gene-related peptide (CGRP) therapies, to non-pharmacological approaches, such as neuromodulation, can be offered to the patients. If the patient is experiencing pain in the neck area or other craniofacial area, it is recommended to have a thorough evaluation by a physical therapist with special training in cervical and craniofacial therapeutics and/or a dentist trained in TMD and orofacial pain disorders to work in consultation with a neurologist to elaborate a personalized management plan. Do not overlook the contribution of myofascial pain (trigger points) as well as TMD in the symptomatology of headache disorders. Few patients need to undergo surgical measures of peripheral nerves and muscles for improvement. An exhaustive evaluation must be undertaken first.
Resources for patients:
AHS
https://americanheadachesociety.org/
https://americanheadachesociety.org/wp-content/uploads/2018/06/Choosing-Wisely-Flyer.pdf
AAOP
https://aaop.clubexpress.com/content.aspx?sl=1152088466
PTBCTT