User login
Amyloid brain imaging changed clinical management in 60% of MCI and dementia patients
.
Diagnoses changed from Alzheimer’s disease to non–Alzheimer’s disease in 25% of 11,409 patients and from non–Alzheimer’s disease to Alzheimer’s disease in 10.5%, reported Gil Rabinovici, MD, and his colleagues. The use of Alzheimer’s disease drugs doubled in amyloid-positive MCI patients, and increased by a third in amyloid-positive dementia patients. Physicians involved in the study said the scans provided key clinical information in 82% of cases with post-scan management changes.
Scans also benefited amyloid-negative patients. Before the scan, 71% of these carried an Alzheimer’s disease diagnosis; afterward, just 10% did, opening the way for an accurate diagnosis and more effective treatment.
The study was powered to detect a 30% or greater change in the MCI and dementia groups. The 60% change emphasize how useful amyloid PET scans could be in clinical practice, Dr. Rabinovici, the study’s lead author and principal investigator, said in a press statement.
“We are impressed by the magnitude of these results, which make it clear that amyloid PET imaging can have a major impact on how we diagnose and care for patients with Alzheimer’s disease and other forms of cognitive decline,” said Dr. Rabinovici of the University of California, San Francisco.
Alzheimer’s Association leaders were similarly pleased.
“These results present highly credible, large-scale evidence that amyloid PET imaging can be a powerful tool to improve the accuracy of Alzheimer’s diagnosis and lead to better medical management, especially in difficult-to-diagnose cases,” said Maria C. Carrillo, PhD, chief science officer of the Alzheimer’s Association and a coauthor of the study. “It is important that amyloid PET imaging be more broadly accessible to those who need it.”
Next steps
Ultimately, investigators hope the nationwide-wide, open-label study will prove the clinical value of amyloid PET scanning and convince the Centers for Medicare & Medicaid Services to make the test a fully covered service for those who meet the appropriate use criteria set forth by the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging.
IDEAS’ second goal – showing that the scans improve health outcomes – is scheduled for 2020. These data are a key component of the CMS decision, but they might be a tough sell, Clifford R. Jack Jr., MD, and Ronald C. Petersen, MD, PhD, wrote in an accompanying editorial. Dr. Jack and Dr. Petersen are affiliated with the Mayo Clinic in Rochester, Minn.
“For CMS to cover the cost of amyloid PET, it must be demonstrated that the result of a scan has an effect on patient outcomes, not just patient care processes – and, without a disease-modifying therapy available, that might be a challenge,” they wrote.
IDEAS is a funding collaboration of the CMS, the Alzheimer’s Association, Avid Radiopharmaceuticals/Eli Lilly, General Electric Healthcare, Piramal Imaging, and the American College of Radiology. Dr. Rabinovici had no financial disclosures.
SOURCE: Rabinovici GD et al. JAMA. 2019 Apr 2. doi: 10.1001/jama.2019.2000.
Current clinical practice does not routinely include biomarkers, and if given a choice, most patients would prefer brain imaging to spinal fluid-based testing, so IDEAS may be making imaging-based biomarker characterization a real possibility in the future.
Richard J. Caselli, MD, is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center. He made these comments in an interview.
Current clinical practice does not routinely include biomarkers, and if given a choice, most patients would prefer brain imaging to spinal fluid-based testing, so IDEAS may be making imaging-based biomarker characterization a real possibility in the future.
Richard J. Caselli, MD, is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center. He made these comments in an interview.
Current clinical practice does not routinely include biomarkers, and if given a choice, most patients would prefer brain imaging to spinal fluid-based testing, so IDEAS may be making imaging-based biomarker characterization a real possibility in the future.
Richard J. Caselli, MD, is professor of neurology at the Mayo Clinic Arizona in Scottsdale and associate director and clinical core director of the Arizona Alzheimer’s Disease Center. He made these comments in an interview.
.
Diagnoses changed from Alzheimer’s disease to non–Alzheimer’s disease in 25% of 11,409 patients and from non–Alzheimer’s disease to Alzheimer’s disease in 10.5%, reported Gil Rabinovici, MD, and his colleagues. The use of Alzheimer’s disease drugs doubled in amyloid-positive MCI patients, and increased by a third in amyloid-positive dementia patients. Physicians involved in the study said the scans provided key clinical information in 82% of cases with post-scan management changes.
Scans also benefited amyloid-negative patients. Before the scan, 71% of these carried an Alzheimer’s disease diagnosis; afterward, just 10% did, opening the way for an accurate diagnosis and more effective treatment.
The study was powered to detect a 30% or greater change in the MCI and dementia groups. The 60% change emphasize how useful amyloid PET scans could be in clinical practice, Dr. Rabinovici, the study’s lead author and principal investigator, said in a press statement.
“We are impressed by the magnitude of these results, which make it clear that amyloid PET imaging can have a major impact on how we diagnose and care for patients with Alzheimer’s disease and other forms of cognitive decline,” said Dr. Rabinovici of the University of California, San Francisco.
Alzheimer’s Association leaders were similarly pleased.
“These results present highly credible, large-scale evidence that amyloid PET imaging can be a powerful tool to improve the accuracy of Alzheimer’s diagnosis and lead to better medical management, especially in difficult-to-diagnose cases,” said Maria C. Carrillo, PhD, chief science officer of the Alzheimer’s Association and a coauthor of the study. “It is important that amyloid PET imaging be more broadly accessible to those who need it.”
Next steps
Ultimately, investigators hope the nationwide-wide, open-label study will prove the clinical value of amyloid PET scanning and convince the Centers for Medicare & Medicaid Services to make the test a fully covered service for those who meet the appropriate use criteria set forth by the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging.
IDEAS’ second goal – showing that the scans improve health outcomes – is scheduled for 2020. These data are a key component of the CMS decision, but they might be a tough sell, Clifford R. Jack Jr., MD, and Ronald C. Petersen, MD, PhD, wrote in an accompanying editorial. Dr. Jack and Dr. Petersen are affiliated with the Mayo Clinic in Rochester, Minn.
“For CMS to cover the cost of amyloid PET, it must be demonstrated that the result of a scan has an effect on patient outcomes, not just patient care processes – and, without a disease-modifying therapy available, that might be a challenge,” they wrote.
IDEAS is a funding collaboration of the CMS, the Alzheimer’s Association, Avid Radiopharmaceuticals/Eli Lilly, General Electric Healthcare, Piramal Imaging, and the American College of Radiology. Dr. Rabinovici had no financial disclosures.
SOURCE: Rabinovici GD et al. JAMA. 2019 Apr 2. doi: 10.1001/jama.2019.2000.
.
Diagnoses changed from Alzheimer’s disease to non–Alzheimer’s disease in 25% of 11,409 patients and from non–Alzheimer’s disease to Alzheimer’s disease in 10.5%, reported Gil Rabinovici, MD, and his colleagues. The use of Alzheimer’s disease drugs doubled in amyloid-positive MCI patients, and increased by a third in amyloid-positive dementia patients. Physicians involved in the study said the scans provided key clinical information in 82% of cases with post-scan management changes.
Scans also benefited amyloid-negative patients. Before the scan, 71% of these carried an Alzheimer’s disease diagnosis; afterward, just 10% did, opening the way for an accurate diagnosis and more effective treatment.
The study was powered to detect a 30% or greater change in the MCI and dementia groups. The 60% change emphasize how useful amyloid PET scans could be in clinical practice, Dr. Rabinovici, the study’s lead author and principal investigator, said in a press statement.
“We are impressed by the magnitude of these results, which make it clear that amyloid PET imaging can have a major impact on how we diagnose and care for patients with Alzheimer’s disease and other forms of cognitive decline,” said Dr. Rabinovici of the University of California, San Francisco.
Alzheimer’s Association leaders were similarly pleased.
“These results present highly credible, large-scale evidence that amyloid PET imaging can be a powerful tool to improve the accuracy of Alzheimer’s diagnosis and lead to better medical management, especially in difficult-to-diagnose cases,” said Maria C. Carrillo, PhD, chief science officer of the Alzheimer’s Association and a coauthor of the study. “It is important that amyloid PET imaging be more broadly accessible to those who need it.”
Next steps
Ultimately, investigators hope the nationwide-wide, open-label study will prove the clinical value of amyloid PET scanning and convince the Centers for Medicare & Medicaid Services to make the test a fully covered service for those who meet the appropriate use criteria set forth by the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging.
IDEAS’ second goal – showing that the scans improve health outcomes – is scheduled for 2020. These data are a key component of the CMS decision, but they might be a tough sell, Clifford R. Jack Jr., MD, and Ronald C. Petersen, MD, PhD, wrote in an accompanying editorial. Dr. Jack and Dr. Petersen are affiliated with the Mayo Clinic in Rochester, Minn.
“For CMS to cover the cost of amyloid PET, it must be demonstrated that the result of a scan has an effect on patient outcomes, not just patient care processes – and, without a disease-modifying therapy available, that might be a challenge,” they wrote.
IDEAS is a funding collaboration of the CMS, the Alzheimer’s Association, Avid Radiopharmaceuticals/Eli Lilly, General Electric Healthcare, Piramal Imaging, and the American College of Radiology. Dr. Rabinovici had no financial disclosures.
SOURCE: Rabinovici GD et al. JAMA. 2019 Apr 2. doi: 10.1001/jama.2019.2000.
FROM THE JOURNAL OF THE AMERICAN MEDICAL ASSOCIATION
FDA approves Mavenclad for treatment of relapsing MS
including relapsing/remitting and active secondary progressive disease.
The drug’s manufacturer, EMD Serono, said in a press release that cladribine is the first short-course oral therapy for such patients, and its use is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS. Cladribine is not recommended for use in patients with clinically isolated syndrome.
The agency’s decision is based on results from a clinical trial of 1,326 patients with relapsing MS who had experienced at least one relapse in the previous 12 months. Patients who received cladribine had significantly fewer relapses than did those who received placebo; the progression of disability was also significantly reduced in the cladribine group, compared with placebo, according to the FDA’s announcement.
The most common adverse events associated with cladribine include upper respiratory tract infections, headache, and decreased lymphocyte counts. In addition, the medication must be dispensed with a patient medication guide because the label includes a boxed warning for increased risk of malignancy and fetal harm. Other warnings include a risk for decreased lymphocyte count, hematologic toxicity and bone marrow suppression, and graft-versus-host-disease.
“We are committed to supporting the development of safe and effective treatments for patients with multiple sclerosis. The approval of Mavenclad represents an additional option for patients who have tried another treatment without success,” Billy Dunn, MD, director of the division of neurology products in the FDA’s Center for Drug Evaluation and Research, said in the announcement.
The approved dose of cladribine is 3.5 mg/kg body weight over 2 years, administered as one treatment course of 1.75 mg/kg per year, each consisting of 2 treatment weeks. Additional courses of cladribine are not to be administered because retreatment with cladribine during years 3 and 4 may further increase the risk of malignancy. The safety and efficacy of reinitiating cladribine more than 2 years after completing two treatment courses has not been studied, according to EMD Serono.
Cladribine is approved in more than 50 other countries and was approved for use in the European Union in August 2017.
including relapsing/remitting and active secondary progressive disease.
The drug’s manufacturer, EMD Serono, said in a press release that cladribine is the first short-course oral therapy for such patients, and its use is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS. Cladribine is not recommended for use in patients with clinically isolated syndrome.
The agency’s decision is based on results from a clinical trial of 1,326 patients with relapsing MS who had experienced at least one relapse in the previous 12 months. Patients who received cladribine had significantly fewer relapses than did those who received placebo; the progression of disability was also significantly reduced in the cladribine group, compared with placebo, according to the FDA’s announcement.
The most common adverse events associated with cladribine include upper respiratory tract infections, headache, and decreased lymphocyte counts. In addition, the medication must be dispensed with a patient medication guide because the label includes a boxed warning for increased risk of malignancy and fetal harm. Other warnings include a risk for decreased lymphocyte count, hematologic toxicity and bone marrow suppression, and graft-versus-host-disease.
“We are committed to supporting the development of safe and effective treatments for patients with multiple sclerosis. The approval of Mavenclad represents an additional option for patients who have tried another treatment without success,” Billy Dunn, MD, director of the division of neurology products in the FDA’s Center for Drug Evaluation and Research, said in the announcement.
The approved dose of cladribine is 3.5 mg/kg body weight over 2 years, administered as one treatment course of 1.75 mg/kg per year, each consisting of 2 treatment weeks. Additional courses of cladribine are not to be administered because retreatment with cladribine during years 3 and 4 may further increase the risk of malignancy. The safety and efficacy of reinitiating cladribine more than 2 years after completing two treatment courses has not been studied, according to EMD Serono.
Cladribine is approved in more than 50 other countries and was approved for use in the European Union in August 2017.
including relapsing/remitting and active secondary progressive disease.
The drug’s manufacturer, EMD Serono, said in a press release that cladribine is the first short-course oral therapy for such patients, and its use is generally recommended for patients who have had an inadequate response to, or are unable to tolerate, an alternate drug indicated for the treatment of MS. Cladribine is not recommended for use in patients with clinically isolated syndrome.
The agency’s decision is based on results from a clinical trial of 1,326 patients with relapsing MS who had experienced at least one relapse in the previous 12 months. Patients who received cladribine had significantly fewer relapses than did those who received placebo; the progression of disability was also significantly reduced in the cladribine group, compared with placebo, according to the FDA’s announcement.
The most common adverse events associated with cladribine include upper respiratory tract infections, headache, and decreased lymphocyte counts. In addition, the medication must be dispensed with a patient medication guide because the label includes a boxed warning for increased risk of malignancy and fetal harm. Other warnings include a risk for decreased lymphocyte count, hematologic toxicity and bone marrow suppression, and graft-versus-host-disease.
“We are committed to supporting the development of safe and effective treatments for patients with multiple sclerosis. The approval of Mavenclad represents an additional option for patients who have tried another treatment without success,” Billy Dunn, MD, director of the division of neurology products in the FDA’s Center for Drug Evaluation and Research, said in the announcement.
The approved dose of cladribine is 3.5 mg/kg body weight over 2 years, administered as one treatment course of 1.75 mg/kg per year, each consisting of 2 treatment weeks. Additional courses of cladribine are not to be administered because retreatment with cladribine during years 3 and 4 may further increase the risk of malignancy. The safety and efficacy of reinitiating cladribine more than 2 years after completing two treatment courses has not been studied, according to EMD Serono.
Cladribine is approved in more than 50 other countries and was approved for use in the European Union in August 2017.
Ibrutinib sustained responses in refractory CLL in long-term follow-up
Prolonged exposure to ibrutinib showed sustained progression-free and overall survival and had tolerable safety outcomes in patients with relapsed or refractory chronic lymphocytic leukemia, according to a post hoc analysis of the phase 3 RESONATE trial.
“This study ... provides further evidence for efficacy and safety with prolonged treatment across multiple high-risk genomic and clinical disease features and with increasing depth of response,” John C. Byrd, MD, of the Ohio State University, Columbus, and his colleagues wrote in Blood.
RESONATE included 391 high-risk patients with relapsed or refractory chronic lymphocytic leukemia (CLL). Study participants were randomized in a 1:1 fashion to receive ibrutinib 420 mg daily or ofatumumab (initial infusion of 300 mg followed by seven weekly infusions and four monthly infusions of 2,000 mg) for a maximum of 24 weeks. Drug therapy was continued until cancer progression or intolerable toxicity of either agent was seen.
“Primary analysis at median follow-up of 9.7 months demonstrated superiority of ibrutinib over ofatumumab in PFS [progression-free survival], OS [overall survival], and overall response,” the researchers wrote. “With extended follow-up of median 44 months, these same results persist; a plateau of PFS has not yet been reached in this long-term follow-up. We also observe very durable remissions among patients of all genomic groups, including those with del(17)(p13.1), del(11)(q22.3), or unmutated IgHV [immunoglobulin heavy chain gene], who are traditionally considered high-risk populations.”
After an extended follow-up (median, 44 months), the team found that the PFS benefit with ibrutinib was sustained, compared with ofatumumab (hazard ratio, 0.133; 95% confidence interval, 0.099-0.178; P less than .0001). The 3-year PFS rate was 59% for ibrutinib, compared with 3% for ofatumumab. Similar PFS benefits were seen among subgroups of high- and very high–risk patients, based on their scores on the International Prognostic Index for CLL.
The OS benefit was also sustained in those randomized to ibrutinib (HR, 0.591; 95% CI, 0.378-0.926; P = .0208). The continued OS benefit with ibrutinib versus ofatumumab continued even after a sensitivity analysis adjusted for crossover of patients to ibrutinib.
With respect to safety, adverse events of any grade were similar to previous reports of ibrutinib. In fact, the prevalence of adverse events (grade 3 or higher) decreased over time for participants that continued on ibrutinib.
“Multiple studies are ongoing to investigate ibrutinib earlier in the course of CLL therapy, including phase 3 studies of first-line ibrutinib [or ibrutinib combined with anti-CD20 therapy], compared with standard chemoimmunotherapy regimens,” they wrote.
The study was sponsored by Pharmacyclics and Janssen. The authors reported financial disclosures related to the sponsors and several other companies.
SOURCE: Byrd JC et al. Blood. 2019 Mar 6. doi: 10.1182/blood-2018-08-870238.
Prolonged exposure to ibrutinib showed sustained progression-free and overall survival and had tolerable safety outcomes in patients with relapsed or refractory chronic lymphocytic leukemia, according to a post hoc analysis of the phase 3 RESONATE trial.
“This study ... provides further evidence for efficacy and safety with prolonged treatment across multiple high-risk genomic and clinical disease features and with increasing depth of response,” John C. Byrd, MD, of the Ohio State University, Columbus, and his colleagues wrote in Blood.
RESONATE included 391 high-risk patients with relapsed or refractory chronic lymphocytic leukemia (CLL). Study participants were randomized in a 1:1 fashion to receive ibrutinib 420 mg daily or ofatumumab (initial infusion of 300 mg followed by seven weekly infusions and four monthly infusions of 2,000 mg) for a maximum of 24 weeks. Drug therapy was continued until cancer progression or intolerable toxicity of either agent was seen.
“Primary analysis at median follow-up of 9.7 months demonstrated superiority of ibrutinib over ofatumumab in PFS [progression-free survival], OS [overall survival], and overall response,” the researchers wrote. “With extended follow-up of median 44 months, these same results persist; a plateau of PFS has not yet been reached in this long-term follow-up. We also observe very durable remissions among patients of all genomic groups, including those with del(17)(p13.1), del(11)(q22.3), or unmutated IgHV [immunoglobulin heavy chain gene], who are traditionally considered high-risk populations.”
After an extended follow-up (median, 44 months), the team found that the PFS benefit with ibrutinib was sustained, compared with ofatumumab (hazard ratio, 0.133; 95% confidence interval, 0.099-0.178; P less than .0001). The 3-year PFS rate was 59% for ibrutinib, compared with 3% for ofatumumab. Similar PFS benefits were seen among subgroups of high- and very high–risk patients, based on their scores on the International Prognostic Index for CLL.
The OS benefit was also sustained in those randomized to ibrutinib (HR, 0.591; 95% CI, 0.378-0.926; P = .0208). The continued OS benefit with ibrutinib versus ofatumumab continued even after a sensitivity analysis adjusted for crossover of patients to ibrutinib.
With respect to safety, adverse events of any grade were similar to previous reports of ibrutinib. In fact, the prevalence of adverse events (grade 3 or higher) decreased over time for participants that continued on ibrutinib.
“Multiple studies are ongoing to investigate ibrutinib earlier in the course of CLL therapy, including phase 3 studies of first-line ibrutinib [or ibrutinib combined with anti-CD20 therapy], compared with standard chemoimmunotherapy regimens,” they wrote.
The study was sponsored by Pharmacyclics and Janssen. The authors reported financial disclosures related to the sponsors and several other companies.
SOURCE: Byrd JC et al. Blood. 2019 Mar 6. doi: 10.1182/blood-2018-08-870238.
Prolonged exposure to ibrutinib showed sustained progression-free and overall survival and had tolerable safety outcomes in patients with relapsed or refractory chronic lymphocytic leukemia, according to a post hoc analysis of the phase 3 RESONATE trial.
“This study ... provides further evidence for efficacy and safety with prolonged treatment across multiple high-risk genomic and clinical disease features and with increasing depth of response,” John C. Byrd, MD, of the Ohio State University, Columbus, and his colleagues wrote in Blood.
RESONATE included 391 high-risk patients with relapsed or refractory chronic lymphocytic leukemia (CLL). Study participants were randomized in a 1:1 fashion to receive ibrutinib 420 mg daily or ofatumumab (initial infusion of 300 mg followed by seven weekly infusions and four monthly infusions of 2,000 mg) for a maximum of 24 weeks. Drug therapy was continued until cancer progression or intolerable toxicity of either agent was seen.
“Primary analysis at median follow-up of 9.7 months demonstrated superiority of ibrutinib over ofatumumab in PFS [progression-free survival], OS [overall survival], and overall response,” the researchers wrote. “With extended follow-up of median 44 months, these same results persist; a plateau of PFS has not yet been reached in this long-term follow-up. We also observe very durable remissions among patients of all genomic groups, including those with del(17)(p13.1), del(11)(q22.3), or unmutated IgHV [immunoglobulin heavy chain gene], who are traditionally considered high-risk populations.”
After an extended follow-up (median, 44 months), the team found that the PFS benefit with ibrutinib was sustained, compared with ofatumumab (hazard ratio, 0.133; 95% confidence interval, 0.099-0.178; P less than .0001). The 3-year PFS rate was 59% for ibrutinib, compared with 3% for ofatumumab. Similar PFS benefits were seen among subgroups of high- and very high–risk patients, based on their scores on the International Prognostic Index for CLL.
The OS benefit was also sustained in those randomized to ibrutinib (HR, 0.591; 95% CI, 0.378-0.926; P = .0208). The continued OS benefit with ibrutinib versus ofatumumab continued even after a sensitivity analysis adjusted for crossover of patients to ibrutinib.
With respect to safety, adverse events of any grade were similar to previous reports of ibrutinib. In fact, the prevalence of adverse events (grade 3 or higher) decreased over time for participants that continued on ibrutinib.
“Multiple studies are ongoing to investigate ibrutinib earlier in the course of CLL therapy, including phase 3 studies of first-line ibrutinib [or ibrutinib combined with anti-CD20 therapy], compared with standard chemoimmunotherapy regimens,” they wrote.
The study was sponsored by Pharmacyclics and Janssen. The authors reported financial disclosures related to the sponsors and several other companies.
SOURCE: Byrd JC et al. Blood. 2019 Mar 6. doi: 10.1182/blood-2018-08-870238.
FROM BLOOD
Many EMS protocols for status epilepticus do not follow evidence-based guidelines
“Many protocols did not follow evidence-based guidelines and did not accurately define generalized convulsive status epilepticus,” said John P. Betjemann, MD, associate professor of neurology at the University of California, San Francisco, and his colleagues. They reported their findings in the March 26 issue of JAMA.
Generalized convulsive status epilepticus is a neurologic emergency, and trials published in 2001 and 2012 found that benzodiazepines are effective prehospital treatments for patients with generalized convulsive status epilepticus. These trials informed a 2016 evidence-based guideline that cites level A evidence for intramuscular midazolam, IV lorazepam, and IV diazepam as initial treatment options for adults.
To determine whether EMS system protocols follow these recommendations, the investigators reviewed treatment protocols from 33 EMS systems that cover the 58 counties in California. The researchers reviewed EMS system protocols between May and June 2018 to determine when they were last updated and whether they defined generalized convulsive status epilepticus according to the guideline (namely, 5 or more minutes of continuous seizure or two or more discrete seizures between which a patient has incomplete recovery of consciousness). They also determined whether the protocols included any of the three benzodiazepines in the guideline and, if so, at what dose and using which route of administration.
Protocols’ most recent revision dates ranged between 2007 and 2018. Twenty-seven protocols (81.8%) were revised after the second clinical trial was published in 2012, and 17 (51.5%) were revised after the 2016 guideline. Seven EMS system protocols (21.2%) defined generalized convulsive status epilepticus according to the guideline. Thirty-two protocols (97.0%) included intramuscular midazolam, 2 (6.1%) included IV lorazepam, and 5 (15.2%) included IV diazepam.
Although the protocols “appropriately emphasized” intramuscular midazolam, the protocol doses often were lower than those used in the trials or recommended in the guideline. In addition, most protocols listed IV and intraosseous midazolam as options, although these treatments were not studied in the trials nor recommended in the guideline. In all, six of the protocols (18.2%) recommended at least one medication by the route and dose suggested in the trials or in the guideline.
“Why EMS system protocols deviate from the evidence and how this affects patient outcomes deserves further study,” the authors said.
The researchers noted that they examined EMS protocols in only one state and that “protocols may not necessarily reflect what emergency medical technicians actually do in practice.” In addition, the researchers accessed the most recent protocols by consulting EMS system websites rather than by contacting each EMS system for its most up-to-date protocol.
The authors reported personal compensation from JAMA Neurology and from Continuum Audio unrelated to the present study, as well as grants from the National Institutes of Health.
SOURCE: Betjemann JP et al. JAMA. 2019 Mar 26.
“Many protocols did not follow evidence-based guidelines and did not accurately define generalized convulsive status epilepticus,” said John P. Betjemann, MD, associate professor of neurology at the University of California, San Francisco, and his colleagues. They reported their findings in the March 26 issue of JAMA.
Generalized convulsive status epilepticus is a neurologic emergency, and trials published in 2001 and 2012 found that benzodiazepines are effective prehospital treatments for patients with generalized convulsive status epilepticus. These trials informed a 2016 evidence-based guideline that cites level A evidence for intramuscular midazolam, IV lorazepam, and IV diazepam as initial treatment options for adults.
To determine whether EMS system protocols follow these recommendations, the investigators reviewed treatment protocols from 33 EMS systems that cover the 58 counties in California. The researchers reviewed EMS system protocols between May and June 2018 to determine when they were last updated and whether they defined generalized convulsive status epilepticus according to the guideline (namely, 5 or more minutes of continuous seizure or two or more discrete seizures between which a patient has incomplete recovery of consciousness). They also determined whether the protocols included any of the three benzodiazepines in the guideline and, if so, at what dose and using which route of administration.
Protocols’ most recent revision dates ranged between 2007 and 2018. Twenty-seven protocols (81.8%) were revised after the second clinical trial was published in 2012, and 17 (51.5%) were revised after the 2016 guideline. Seven EMS system protocols (21.2%) defined generalized convulsive status epilepticus according to the guideline. Thirty-two protocols (97.0%) included intramuscular midazolam, 2 (6.1%) included IV lorazepam, and 5 (15.2%) included IV diazepam.
Although the protocols “appropriately emphasized” intramuscular midazolam, the protocol doses often were lower than those used in the trials or recommended in the guideline. In addition, most protocols listed IV and intraosseous midazolam as options, although these treatments were not studied in the trials nor recommended in the guideline. In all, six of the protocols (18.2%) recommended at least one medication by the route and dose suggested in the trials or in the guideline.
“Why EMS system protocols deviate from the evidence and how this affects patient outcomes deserves further study,” the authors said.
The researchers noted that they examined EMS protocols in only one state and that “protocols may not necessarily reflect what emergency medical technicians actually do in practice.” In addition, the researchers accessed the most recent protocols by consulting EMS system websites rather than by contacting each EMS system for its most up-to-date protocol.
The authors reported personal compensation from JAMA Neurology and from Continuum Audio unrelated to the present study, as well as grants from the National Institutes of Health.
SOURCE: Betjemann JP et al. JAMA. 2019 Mar 26.
“Many protocols did not follow evidence-based guidelines and did not accurately define generalized convulsive status epilepticus,” said John P. Betjemann, MD, associate professor of neurology at the University of California, San Francisco, and his colleagues. They reported their findings in the March 26 issue of JAMA.
Generalized convulsive status epilepticus is a neurologic emergency, and trials published in 2001 and 2012 found that benzodiazepines are effective prehospital treatments for patients with generalized convulsive status epilepticus. These trials informed a 2016 evidence-based guideline that cites level A evidence for intramuscular midazolam, IV lorazepam, and IV diazepam as initial treatment options for adults.
To determine whether EMS system protocols follow these recommendations, the investigators reviewed treatment protocols from 33 EMS systems that cover the 58 counties in California. The researchers reviewed EMS system protocols between May and June 2018 to determine when they were last updated and whether they defined generalized convulsive status epilepticus according to the guideline (namely, 5 or more minutes of continuous seizure or two or more discrete seizures between which a patient has incomplete recovery of consciousness). They also determined whether the protocols included any of the three benzodiazepines in the guideline and, if so, at what dose and using which route of administration.
Protocols’ most recent revision dates ranged between 2007 and 2018. Twenty-seven protocols (81.8%) were revised after the second clinical trial was published in 2012, and 17 (51.5%) were revised after the 2016 guideline. Seven EMS system protocols (21.2%) defined generalized convulsive status epilepticus according to the guideline. Thirty-two protocols (97.0%) included intramuscular midazolam, 2 (6.1%) included IV lorazepam, and 5 (15.2%) included IV diazepam.
Although the protocols “appropriately emphasized” intramuscular midazolam, the protocol doses often were lower than those used in the trials or recommended in the guideline. In addition, most protocols listed IV and intraosseous midazolam as options, although these treatments were not studied in the trials nor recommended in the guideline. In all, six of the protocols (18.2%) recommended at least one medication by the route and dose suggested in the trials or in the guideline.
“Why EMS system protocols deviate from the evidence and how this affects patient outcomes deserves further study,” the authors said.
The researchers noted that they examined EMS protocols in only one state and that “protocols may not necessarily reflect what emergency medical technicians actually do in practice.” In addition, the researchers accessed the most recent protocols by consulting EMS system websites rather than by contacting each EMS system for its most up-to-date protocol.
The authors reported personal compensation from JAMA Neurology and from Continuum Audio unrelated to the present study, as well as grants from the National Institutes of Health.
SOURCE: Betjemann JP et al. JAMA. 2019 Mar 26.
FROM JAMA
Key clinical point: Many emergency medical services (EMS) system protocols may not follow evidence-based guidelines or accurately define generalized convulsive status epilepticus.
Major finding: In all, 18.2% of the protocols recommended at least one medication by the route and at the dose suggested in clinical trials or in an evidence-based guideline.
Study details: A review of treatment protocols from 33 EMS systems that cover the 58 counties in California.
Disclosures: The authors reported personal compensation from JAMA Neurology and Continuum Audio unrelated to the present study and grants from the National Institutes of Health.
Source: Betjemann JP et al. JAMA. 2019 March 26.
Interview with John Corboy, MD, on discontinuing disease modifying therapy in elderly patients with MS
Discontinuing
How would you characterize the prevalence of MS in the elderly?
DR. CORBOY: A recent large demographic study put together by the National MS Society found that there’s almost a million individuals diagnosed with MS over the course of the last 40 to 50 years. The largest population segment was those aged 55 to 64 years. People with MS aged 55 or older constituted 46% of all those with MS.
What disease-modifying therapies (DMTs) are approved by the FDA for the elderly?
DR. CORBOY: Of the drugs that have received FDA approval, most are for individuals over the age of 18 and there’s no specific age cutoff. However, there’s no data supporting DMT use in people over the age of 55 because they were excluded from the studies.
There’s one DMT, fingolimod, that was approved for use in patients under the age of 18; all others are approved for 18 and above. However, none of them are explicitly approved for people over the age of 55, because there is no data to support it.
What is the goal of your study, the DISCO MS trial?
DR. CORBOY: The DISCO MS trial will be the first randomized, controlled, blinded discontinuation trial in the MS space. The objective is to assess the benefit of DMTs in patients over the age of 55.
Part of the rationale for the trial is that prior subgroup analyses have shown that the vast majority of the benefit that we’ve been able to measure with all of these DMTs is seen in those who are under age 45.
A number of studies have examined existing databases and individuals who were either randomly or deliberately taken off of their medication as they age, including people who were felt to be stable with no recent relapses and no recent changes on their MRI brain. These studies reinforced that when discontinuing medications, the individuals who were much more likely to have recurrence of disease activity were younger patients.
Pathological studies clearly show the number of acutely inflamed plaques in the white matter is dramatically lower in autopsies of older vs younger patients. There are different changes in older patients, with lymphocytic nodules in the meninges, gray matter plaques related to these meningeal nodules, microglial activation, and smaller numbers of active, or mostly, inactive, white matter plaques. It’s been difficult to show any substantial benefit in slowing disability progression, much of which is felt to not be associated with acute inflammatory disease in the aging patient. All of these medicines, which can be thought of as anti-inflammatory medicines, are very beneficial when patients are young but less so as they age.
Would you describe the DISCO MS study design?
DR. CORBOY: Our study looks at individuals who are 55 and older who have not had a relapse for at least 5 years, and who’ve not had a change on their brain scan for at least 3 years.
Individuals will be randomized to either stay on the medication that they’re currently taking or discontinue that medicine. They will be followed then for 2 years. The primary outcome will be either a new relapse or a new scan change. The examining investigators are blinded to whether the patient is currently taking a MS disease modifying therapy.
Secondary outcomes include progression of disability as measured by confirmed change on the Extended Disability Status Scale (EDSS).
The enrollment goal is about 300 patients. There are presently 15 sites. The goal is to have the study completed in about 3 years. We’re presently over halfway through enrollment.
We also have a number of patient-reported outcomes because we’re particularly interested in the patient’s view of what’s going on in terms of how they feel. Understanding that dynamic will be extremely important.
We are including both patients with relapsing MS and progressive forms of MS, noting that they should have no relapse and no scan change at study entry.
What are the challenges with this study?
DR. CORBOY: One challenge is interpreting the information with the assumption that the hypothesis is validated. The hypothesis is that in a stable population of older patients that we can safely discontinue DMTs.
If that is found to be true, the question is how many people will be affected? We know that about 46% of people with MS are 55 and older, but there are not really good estimates of the number of individuals 55 and older who remain on a DMT and who are stable by the definition I just described.
It can be safely said, I think, that a substantial number of the individuals 55 and older are still on DMTs. If there’s almost a million people with MS and 46% are 55 and older, that means around 400,000 people with MS in the United States are aged 55 and older. If only half of those are on a DMT, that leaves 200,000. If only half of those are stable and could go off therapy, that would mean perhaps 100,000 people could discontinue DMTs in the United States. If all those assumptions are true, that would be a substantial savings in the health care burden of the United States from a relatively small population of individuals.
Beyond the cost, there are adverse events associated with using these medications. Older patients are more likely to be at risk of complications of MS DMTs. There also are doctor visits, blood monitoring, and other things that are done over time, and the inconvenience of taking a medicine on a routine basis if, indeed, it’s really not necessary because there is no benefit. Moreover, older individuals have other conditions (eg diabetes, hypertension, arrhythmias, cancer, etc) that may limit their ability to use medications due to risk. We’re very interested to see the outcome.
Discontinuing
How would you characterize the prevalence of MS in the elderly?
DR. CORBOY: A recent large demographic study put together by the National MS Society found that there’s almost a million individuals diagnosed with MS over the course of the last 40 to 50 years. The largest population segment was those aged 55 to 64 years. People with MS aged 55 or older constituted 46% of all those with MS.
What disease-modifying therapies (DMTs) are approved by the FDA for the elderly?
DR. CORBOY: Of the drugs that have received FDA approval, most are for individuals over the age of 18 and there’s no specific age cutoff. However, there’s no data supporting DMT use in people over the age of 55 because they were excluded from the studies.
There’s one DMT, fingolimod, that was approved for use in patients under the age of 18; all others are approved for 18 and above. However, none of them are explicitly approved for people over the age of 55, because there is no data to support it.
What is the goal of your study, the DISCO MS trial?
DR. CORBOY: The DISCO MS trial will be the first randomized, controlled, blinded discontinuation trial in the MS space. The objective is to assess the benefit of DMTs in patients over the age of 55.
Part of the rationale for the trial is that prior subgroup analyses have shown that the vast majority of the benefit that we’ve been able to measure with all of these DMTs is seen in those who are under age 45.
A number of studies have examined existing databases and individuals who were either randomly or deliberately taken off of their medication as they age, including people who were felt to be stable with no recent relapses and no recent changes on their MRI brain. These studies reinforced that when discontinuing medications, the individuals who were much more likely to have recurrence of disease activity were younger patients.
Pathological studies clearly show the number of acutely inflamed plaques in the white matter is dramatically lower in autopsies of older vs younger patients. There are different changes in older patients, with lymphocytic nodules in the meninges, gray matter plaques related to these meningeal nodules, microglial activation, and smaller numbers of active, or mostly, inactive, white matter plaques. It’s been difficult to show any substantial benefit in slowing disability progression, much of which is felt to not be associated with acute inflammatory disease in the aging patient. All of these medicines, which can be thought of as anti-inflammatory medicines, are very beneficial when patients are young but less so as they age.
Would you describe the DISCO MS study design?
DR. CORBOY: Our study looks at individuals who are 55 and older who have not had a relapse for at least 5 years, and who’ve not had a change on their brain scan for at least 3 years.
Individuals will be randomized to either stay on the medication that they’re currently taking or discontinue that medicine. They will be followed then for 2 years. The primary outcome will be either a new relapse or a new scan change. The examining investigators are blinded to whether the patient is currently taking a MS disease modifying therapy.
Secondary outcomes include progression of disability as measured by confirmed change on the Extended Disability Status Scale (EDSS).
The enrollment goal is about 300 patients. There are presently 15 sites. The goal is to have the study completed in about 3 years. We’re presently over halfway through enrollment.
We also have a number of patient-reported outcomes because we’re particularly interested in the patient’s view of what’s going on in terms of how they feel. Understanding that dynamic will be extremely important.
We are including both patients with relapsing MS and progressive forms of MS, noting that they should have no relapse and no scan change at study entry.
What are the challenges with this study?
DR. CORBOY: One challenge is interpreting the information with the assumption that the hypothesis is validated. The hypothesis is that in a stable population of older patients that we can safely discontinue DMTs.
If that is found to be true, the question is how many people will be affected? We know that about 46% of people with MS are 55 and older, but there are not really good estimates of the number of individuals 55 and older who remain on a DMT and who are stable by the definition I just described.
It can be safely said, I think, that a substantial number of the individuals 55 and older are still on DMTs. If there’s almost a million people with MS and 46% are 55 and older, that means around 400,000 people with MS in the United States are aged 55 and older. If only half of those are on a DMT, that leaves 200,000. If only half of those are stable and could go off therapy, that would mean perhaps 100,000 people could discontinue DMTs in the United States. If all those assumptions are true, that would be a substantial savings in the health care burden of the United States from a relatively small population of individuals.
Beyond the cost, there are adverse events associated with using these medications. Older patients are more likely to be at risk of complications of MS DMTs. There also are doctor visits, blood monitoring, and other things that are done over time, and the inconvenience of taking a medicine on a routine basis if, indeed, it’s really not necessary because there is no benefit. Moreover, older individuals have other conditions (eg diabetes, hypertension, arrhythmias, cancer, etc) that may limit their ability to use medications due to risk. We’re very interested to see the outcome.
Discontinuing
How would you characterize the prevalence of MS in the elderly?
DR. CORBOY: A recent large demographic study put together by the National MS Society found that there’s almost a million individuals diagnosed with MS over the course of the last 40 to 50 years. The largest population segment was those aged 55 to 64 years. People with MS aged 55 or older constituted 46% of all those with MS.
What disease-modifying therapies (DMTs) are approved by the FDA for the elderly?
DR. CORBOY: Of the drugs that have received FDA approval, most are for individuals over the age of 18 and there’s no specific age cutoff. However, there’s no data supporting DMT use in people over the age of 55 because they were excluded from the studies.
There’s one DMT, fingolimod, that was approved for use in patients under the age of 18; all others are approved for 18 and above. However, none of them are explicitly approved for people over the age of 55, because there is no data to support it.
What is the goal of your study, the DISCO MS trial?
DR. CORBOY: The DISCO MS trial will be the first randomized, controlled, blinded discontinuation trial in the MS space. The objective is to assess the benefit of DMTs in patients over the age of 55.
Part of the rationale for the trial is that prior subgroup analyses have shown that the vast majority of the benefit that we’ve been able to measure with all of these DMTs is seen in those who are under age 45.
A number of studies have examined existing databases and individuals who were either randomly or deliberately taken off of their medication as they age, including people who were felt to be stable with no recent relapses and no recent changes on their MRI brain. These studies reinforced that when discontinuing medications, the individuals who were much more likely to have recurrence of disease activity were younger patients.
Pathological studies clearly show the number of acutely inflamed plaques in the white matter is dramatically lower in autopsies of older vs younger patients. There are different changes in older patients, with lymphocytic nodules in the meninges, gray matter plaques related to these meningeal nodules, microglial activation, and smaller numbers of active, or mostly, inactive, white matter plaques. It’s been difficult to show any substantial benefit in slowing disability progression, much of which is felt to not be associated with acute inflammatory disease in the aging patient. All of these medicines, which can be thought of as anti-inflammatory medicines, are very beneficial when patients are young but less so as they age.
Would you describe the DISCO MS study design?
DR. CORBOY: Our study looks at individuals who are 55 and older who have not had a relapse for at least 5 years, and who’ve not had a change on their brain scan for at least 3 years.
Individuals will be randomized to either stay on the medication that they’re currently taking or discontinue that medicine. They will be followed then for 2 years. The primary outcome will be either a new relapse or a new scan change. The examining investigators are blinded to whether the patient is currently taking a MS disease modifying therapy.
Secondary outcomes include progression of disability as measured by confirmed change on the Extended Disability Status Scale (EDSS).
The enrollment goal is about 300 patients. There are presently 15 sites. The goal is to have the study completed in about 3 years. We’re presently over halfway through enrollment.
We also have a number of patient-reported outcomes because we’re particularly interested in the patient’s view of what’s going on in terms of how they feel. Understanding that dynamic will be extremely important.
We are including both patients with relapsing MS and progressive forms of MS, noting that they should have no relapse and no scan change at study entry.
What are the challenges with this study?
DR. CORBOY: One challenge is interpreting the information with the assumption that the hypothesis is validated. The hypothesis is that in a stable population of older patients that we can safely discontinue DMTs.
If that is found to be true, the question is how many people will be affected? We know that about 46% of people with MS are 55 and older, but there are not really good estimates of the number of individuals 55 and older who remain on a DMT and who are stable by the definition I just described.
It can be safely said, I think, that a substantial number of the individuals 55 and older are still on DMTs. If there’s almost a million people with MS and 46% are 55 and older, that means around 400,000 people with MS in the United States are aged 55 and older. If only half of those are on a DMT, that leaves 200,000. If only half of those are stable and could go off therapy, that would mean perhaps 100,000 people could discontinue DMTs in the United States. If all those assumptions are true, that would be a substantial savings in the health care burden of the United States from a relatively small population of individuals.
Beyond the cost, there are adverse events associated with using these medications. Older patients are more likely to be at risk of complications of MS DMTs. There also are doctor visits, blood monitoring, and other things that are done over time, and the inconvenience of taking a medicine on a routine basis if, indeed, it’s really not necessary because there is no benefit. Moreover, older individuals have other conditions (eg diabetes, hypertension, arrhythmias, cancer, etc) that may limit their ability to use medications due to risk. We’re very interested to see the outcome.
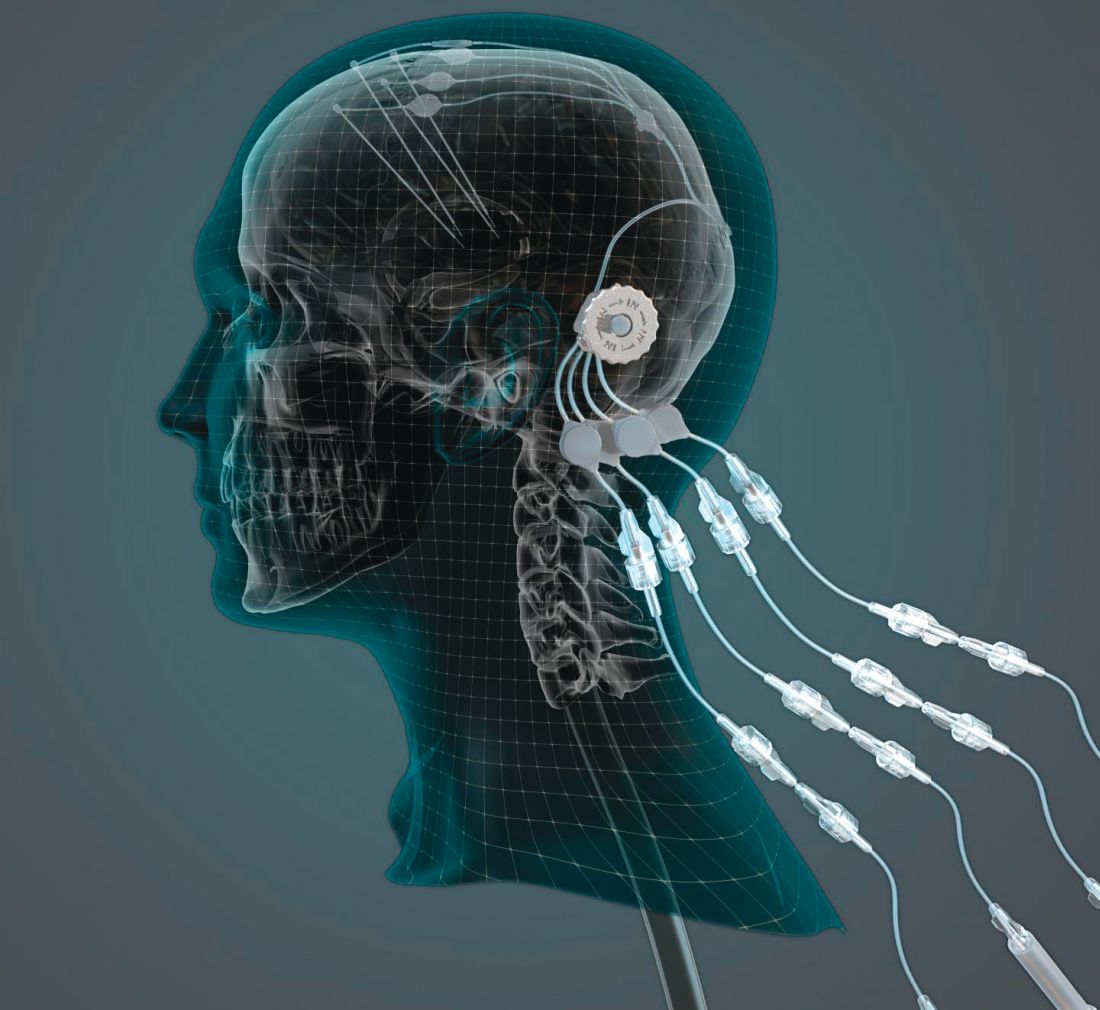
Can intraputamenal infusions of GDNF treat Parkinson’s disease?
researchers reported. The investigational therapy, delivered through a skull-mounted port, was well tolerated in a 40-week, randomized, controlled trial and a 40-week, open-label extension.
Neither study met its primary endpoint, but post hoc analyses suggest possible clinical benefits. In addition, PET imaging after the 40-week, randomized trial found significantly increased 18F-DOPA uptake in patients who received GDNF. The randomized trial was published in the March 2019 issue of Brain; data from the open-label extension were published online ahead of print Feb. 26, 2019, in the Journal of Parkinson’s Disease.
“The spatial and relative magnitude of the improvement in the brain scans is beyond anything seen previously in trials of surgically delivered growth-factor treatments for Parkinson’s [disease],” said principal investigator Alan L. Whone, MBChB, PhD, of the University of Bristol (England) and North Bristol National Health Service Trust. “This represents some of the most compelling evidence yet that we may have a means to possibly reawaken and restore the dopamine brain cells that are gradually destroyed in Parkinson’s [disease].”
Nevertheless, the trial did not confirm clinical benefits. The hypothesis that growth factors can benefit patients with Parkinson’s disease may be incorrect, the researchers acknowledged. It also is possible that the hypothesis is valid and that a trial with a higher GDNF dose, longer treatment duration, patients with an earlier disease stage, or different outcome measures would yield positive results. GDNF warrants further study, they wrote.
The findings could have implications for other neurologic disorders as well.
“This trial has shown that we can safely and repeatedly infuse drugs directly into patients’ brains over months or years. This is a significant breakthrough in our ability to treat neurologic conditions ... because most drugs that might work cannot cross from the bloodstream into the brain,” said Steven Gill, MB, MS. Mr. Gill, of the North Bristol NHS Trust and the U.K.-based engineering firm Renishaw, designed the convection-enhanced delivery system used in the studies.
A neurotrophic protein
GDNF has neurorestorative and neuroprotective effects in animal models of Parkinson’s disease. In open-label studies, continuous, low-rate intraputamenal administration of GDNF has shown signs of potential efficacy, but a placebo-controlled trial did not replicate clinical benefits. In the present studies, the researchers assessed intermittent GDNF administration using convection-enhanced delivery, which can achieve wider and more even distribution of GDNF, compared with the previous approach.
The researchers conducted a single-center, randomized, double-blind, placebo-controlled trial to study this novel administration approach. Patients were aged 35-75 years, had motor symptoms for at least 5 years, and had moderate disease severity in the off state (that is, Hoehn and Yahr stage 2-3 and Unified Parkinson’s Disease Rating Scale motor score–part III [UPDRS-III] of 25-45).
In a pilot stage of the trial, six patients were randomized 2:1 to receive GDNF (120 mcg per putamen) or placebo. In the primary stage, another 35 patients were randomized 1:1 to GDNF or placebo. The primary outcome was the percentage change from baseline to week 40 in the off-state UPDRS-III among patients from the primary stage of the trial. Further analyses included all 41 patients from the pilot and primary stages.
Patients in the primary analysis had a mean age of 56.4 years and mean disease duration of 10.9 years. About half were female.
Results on primary and secondary clinical endpoints did not significantly differ between the groups. Average off state UPDRS motor score decreased by 17.3 in the active treatment group, compared with 11.8 in the placebo group.
A post hoc analysis, however, found that nine patients (43%) in the active-treatment group had a large, clinically important motor improvement of 10 or more points in the off state, whereas no placebo patients did. These “10-point responders in the GDNF group are a potential focus of interest; however, as this is a post hoc finding we would not wish to overinterpret its meaning,” Dr. Whone and his colleagues wrote. Among patients who received GDNF, PET imaging demonstrated significantly increased 18F-DOPA uptake throughout the putamen, ranging from a 25% increase in the left anterior putamen to a 100% increase in both posterior putamena, whereas patients who received placebo did not have significantly increased uptake.
No drug-related serious adverse events were reported. “The majority of device-related adverse events were port site associated, most commonly local hypertrophic scarring or infections, amenable to antibiotics,” the investigators wrote. “The frequency of these declined during the trial as surgical and device handling experience improved.”
Open-label extension
By week 80, when all participants had received GDNF, both groups showed moderate to large improvement in symptoms, compared with baseline. From baseline to week 80, percentage change in UPDRS motor score in the off state did not significantly differ between patients who received GDNF for 80 weeks and patients who received placebo followed by GDNF (26.7% vs. 27.6%). Secondary endpoints also did not differ between the groups. Treatment compliance was 97.8%; no patients discontinued the study.
The trials were funded by Parkinson’s UK with support from the Cure Parkinson’s Trust and in association with the North Bristol NHS Trust. GDNF and additional resources and funding were provided by MedGenesis Therapeutix, which owns the license for GDNF and received funding from the Michael J. Fox Foundation for Parkinson’s Research. Renishaw manufactured the convection-enhanced delivery device on behalf of North Bristol NHS Trust. The Gatsby Foundation provided a 3T MRI scanner. Some study authors are employed by and have shares or share options with MedGenesis Therapeutix. Other authors are employees of Renishaw. Dr. Gill is Renishaw’s medical director and may have a future royalty share from the drug delivery system that he invented.
SOURCES: Whone AL et al. Brain. 2019 Feb 26. doi: 10.1093/brain/awz023; Whone AL et al. J Parkinsons Dis. 2019 Feb 26. doi: 10.3233/JPD-191576.
researchers reported. The investigational therapy, delivered through a skull-mounted port, was well tolerated in a 40-week, randomized, controlled trial and a 40-week, open-label extension.
Neither study met its primary endpoint, but post hoc analyses suggest possible clinical benefits. In addition, PET imaging after the 40-week, randomized trial found significantly increased 18F-DOPA uptake in patients who received GDNF. The randomized trial was published in the March 2019 issue of Brain; data from the open-label extension were published online ahead of print Feb. 26, 2019, in the Journal of Parkinson’s Disease.
“The spatial and relative magnitude of the improvement in the brain scans is beyond anything seen previously in trials of surgically delivered growth-factor treatments for Parkinson’s [disease],” said principal investigator Alan L. Whone, MBChB, PhD, of the University of Bristol (England) and North Bristol National Health Service Trust. “This represents some of the most compelling evidence yet that we may have a means to possibly reawaken and restore the dopamine brain cells that are gradually destroyed in Parkinson’s [disease].”
Nevertheless, the trial did not confirm clinical benefits. The hypothesis that growth factors can benefit patients with Parkinson’s disease may be incorrect, the researchers acknowledged. It also is possible that the hypothesis is valid and that a trial with a higher GDNF dose, longer treatment duration, patients with an earlier disease stage, or different outcome measures would yield positive results. GDNF warrants further study, they wrote.
The findings could have implications for other neurologic disorders as well.
“This trial has shown that we can safely and repeatedly infuse drugs directly into patients’ brains over months or years. This is a significant breakthrough in our ability to treat neurologic conditions ... because most drugs that might work cannot cross from the bloodstream into the brain,” said Steven Gill, MB, MS. Mr. Gill, of the North Bristol NHS Trust and the U.K.-based engineering firm Renishaw, designed the convection-enhanced delivery system used in the studies.
A neurotrophic protein
GDNF has neurorestorative and neuroprotective effects in animal models of Parkinson’s disease. In open-label studies, continuous, low-rate intraputamenal administration of GDNF has shown signs of potential efficacy, but a placebo-controlled trial did not replicate clinical benefits. In the present studies, the researchers assessed intermittent GDNF administration using convection-enhanced delivery, which can achieve wider and more even distribution of GDNF, compared with the previous approach.
The researchers conducted a single-center, randomized, double-blind, placebo-controlled trial to study this novel administration approach. Patients were aged 35-75 years, had motor symptoms for at least 5 years, and had moderate disease severity in the off state (that is, Hoehn and Yahr stage 2-3 and Unified Parkinson’s Disease Rating Scale motor score–part III [UPDRS-III] of 25-45).
In a pilot stage of the trial, six patients were randomized 2:1 to receive GDNF (120 mcg per putamen) or placebo. In the primary stage, another 35 patients were randomized 1:1 to GDNF or placebo. The primary outcome was the percentage change from baseline to week 40 in the off-state UPDRS-III among patients from the primary stage of the trial. Further analyses included all 41 patients from the pilot and primary stages.
Patients in the primary analysis had a mean age of 56.4 years and mean disease duration of 10.9 years. About half were female.
Results on primary and secondary clinical endpoints did not significantly differ between the groups. Average off state UPDRS motor score decreased by 17.3 in the active treatment group, compared with 11.8 in the placebo group.
A post hoc analysis, however, found that nine patients (43%) in the active-treatment group had a large, clinically important motor improvement of 10 or more points in the off state, whereas no placebo patients did. These “10-point responders in the GDNF group are a potential focus of interest; however, as this is a post hoc finding we would not wish to overinterpret its meaning,” Dr. Whone and his colleagues wrote. Among patients who received GDNF, PET imaging demonstrated significantly increased 18F-DOPA uptake throughout the putamen, ranging from a 25% increase in the left anterior putamen to a 100% increase in both posterior putamena, whereas patients who received placebo did not have significantly increased uptake.
No drug-related serious adverse events were reported. “The majority of device-related adverse events were port site associated, most commonly local hypertrophic scarring or infections, amenable to antibiotics,” the investigators wrote. “The frequency of these declined during the trial as surgical and device handling experience improved.”
Open-label extension
By week 80, when all participants had received GDNF, both groups showed moderate to large improvement in symptoms, compared with baseline. From baseline to week 80, percentage change in UPDRS motor score in the off state did not significantly differ between patients who received GDNF for 80 weeks and patients who received placebo followed by GDNF (26.7% vs. 27.6%). Secondary endpoints also did not differ between the groups. Treatment compliance was 97.8%; no patients discontinued the study.
The trials were funded by Parkinson’s UK with support from the Cure Parkinson’s Trust and in association with the North Bristol NHS Trust. GDNF and additional resources and funding were provided by MedGenesis Therapeutix, which owns the license for GDNF and received funding from the Michael J. Fox Foundation for Parkinson’s Research. Renishaw manufactured the convection-enhanced delivery device on behalf of North Bristol NHS Trust. The Gatsby Foundation provided a 3T MRI scanner. Some study authors are employed by and have shares or share options with MedGenesis Therapeutix. Other authors are employees of Renishaw. Dr. Gill is Renishaw’s medical director and may have a future royalty share from the drug delivery system that he invented.
SOURCES: Whone AL et al. Brain. 2019 Feb 26. doi: 10.1093/brain/awz023; Whone AL et al. J Parkinsons Dis. 2019 Feb 26. doi: 10.3233/JPD-191576.
researchers reported. The investigational therapy, delivered through a skull-mounted port, was well tolerated in a 40-week, randomized, controlled trial and a 40-week, open-label extension.
Neither study met its primary endpoint, but post hoc analyses suggest possible clinical benefits. In addition, PET imaging after the 40-week, randomized trial found significantly increased 18F-DOPA uptake in patients who received GDNF. The randomized trial was published in the March 2019 issue of Brain; data from the open-label extension were published online ahead of print Feb. 26, 2019, in the Journal of Parkinson’s Disease.
“The spatial and relative magnitude of the improvement in the brain scans is beyond anything seen previously in trials of surgically delivered growth-factor treatments for Parkinson’s [disease],” said principal investigator Alan L. Whone, MBChB, PhD, of the University of Bristol (England) and North Bristol National Health Service Trust. “This represents some of the most compelling evidence yet that we may have a means to possibly reawaken and restore the dopamine brain cells that are gradually destroyed in Parkinson’s [disease].”
Nevertheless, the trial did not confirm clinical benefits. The hypothesis that growth factors can benefit patients with Parkinson’s disease may be incorrect, the researchers acknowledged. It also is possible that the hypothesis is valid and that a trial with a higher GDNF dose, longer treatment duration, patients with an earlier disease stage, or different outcome measures would yield positive results. GDNF warrants further study, they wrote.
The findings could have implications for other neurologic disorders as well.
“This trial has shown that we can safely and repeatedly infuse drugs directly into patients’ brains over months or years. This is a significant breakthrough in our ability to treat neurologic conditions ... because most drugs that might work cannot cross from the bloodstream into the brain,” said Steven Gill, MB, MS. Mr. Gill, of the North Bristol NHS Trust and the U.K.-based engineering firm Renishaw, designed the convection-enhanced delivery system used in the studies.
A neurotrophic protein
GDNF has neurorestorative and neuroprotective effects in animal models of Parkinson’s disease. In open-label studies, continuous, low-rate intraputamenal administration of GDNF has shown signs of potential efficacy, but a placebo-controlled trial did not replicate clinical benefits. In the present studies, the researchers assessed intermittent GDNF administration using convection-enhanced delivery, which can achieve wider and more even distribution of GDNF, compared with the previous approach.
The researchers conducted a single-center, randomized, double-blind, placebo-controlled trial to study this novel administration approach. Patients were aged 35-75 years, had motor symptoms for at least 5 years, and had moderate disease severity in the off state (that is, Hoehn and Yahr stage 2-3 and Unified Parkinson’s Disease Rating Scale motor score–part III [UPDRS-III] of 25-45).
In a pilot stage of the trial, six patients were randomized 2:1 to receive GDNF (120 mcg per putamen) or placebo. In the primary stage, another 35 patients were randomized 1:1 to GDNF or placebo. The primary outcome was the percentage change from baseline to week 40 in the off-state UPDRS-III among patients from the primary stage of the trial. Further analyses included all 41 patients from the pilot and primary stages.
Patients in the primary analysis had a mean age of 56.4 years and mean disease duration of 10.9 years. About half were female.
Results on primary and secondary clinical endpoints did not significantly differ between the groups. Average off state UPDRS motor score decreased by 17.3 in the active treatment group, compared with 11.8 in the placebo group.
A post hoc analysis, however, found that nine patients (43%) in the active-treatment group had a large, clinically important motor improvement of 10 or more points in the off state, whereas no placebo patients did. These “10-point responders in the GDNF group are a potential focus of interest; however, as this is a post hoc finding we would not wish to overinterpret its meaning,” Dr. Whone and his colleagues wrote. Among patients who received GDNF, PET imaging demonstrated significantly increased 18F-DOPA uptake throughout the putamen, ranging from a 25% increase in the left anterior putamen to a 100% increase in both posterior putamena, whereas patients who received placebo did not have significantly increased uptake.
No drug-related serious adverse events were reported. “The majority of device-related adverse events were port site associated, most commonly local hypertrophic scarring or infections, amenable to antibiotics,” the investigators wrote. “The frequency of these declined during the trial as surgical and device handling experience improved.”
Open-label extension
By week 80, when all participants had received GDNF, both groups showed moderate to large improvement in symptoms, compared with baseline. From baseline to week 80, percentage change in UPDRS motor score in the off state did not significantly differ between patients who received GDNF for 80 weeks and patients who received placebo followed by GDNF (26.7% vs. 27.6%). Secondary endpoints also did not differ between the groups. Treatment compliance was 97.8%; no patients discontinued the study.
The trials were funded by Parkinson’s UK with support from the Cure Parkinson’s Trust and in association with the North Bristol NHS Trust. GDNF and additional resources and funding were provided by MedGenesis Therapeutix, which owns the license for GDNF and received funding from the Michael J. Fox Foundation for Parkinson’s Research. Renishaw manufactured the convection-enhanced delivery device on behalf of North Bristol NHS Trust. The Gatsby Foundation provided a 3T MRI scanner. Some study authors are employed by and have shares or share options with MedGenesis Therapeutix. Other authors are employees of Renishaw. Dr. Gill is Renishaw’s medical director and may have a future royalty share from the drug delivery system that he invented.
SOURCES: Whone AL et al. Brain. 2019 Feb 26. doi: 10.1093/brain/awz023; Whone AL et al. J Parkinsons Dis. 2019 Feb 26. doi: 10.3233/JPD-191576.
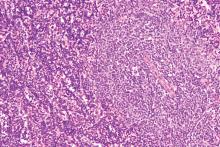
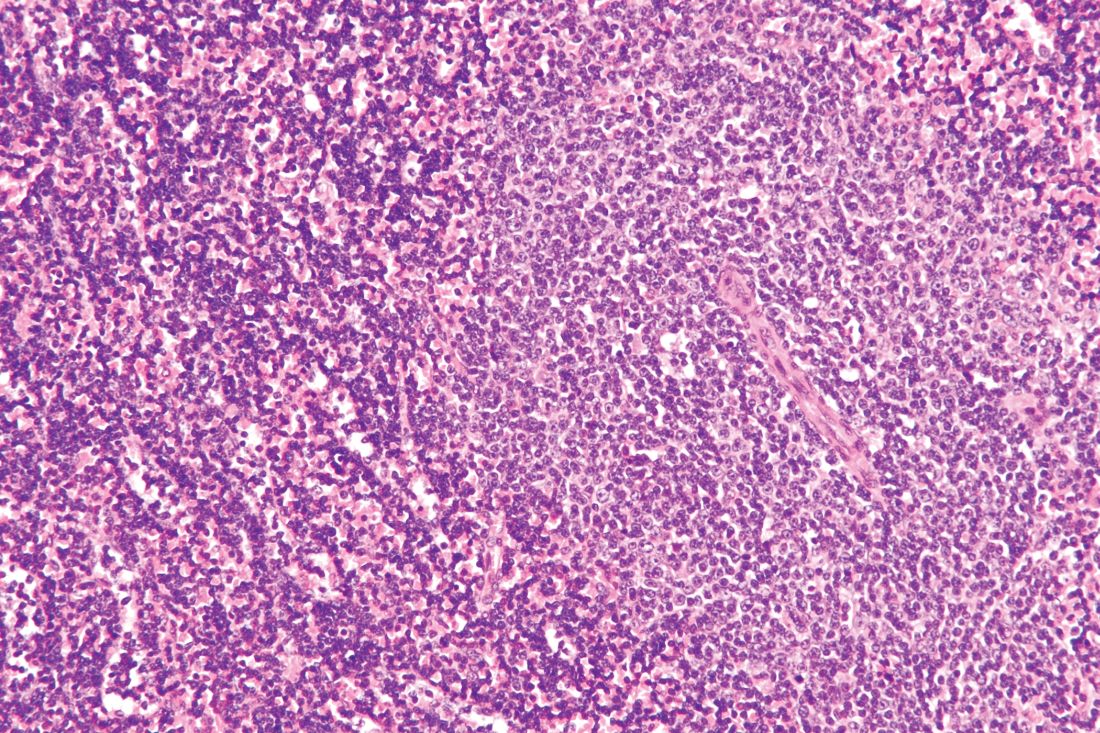
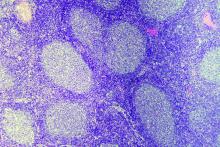
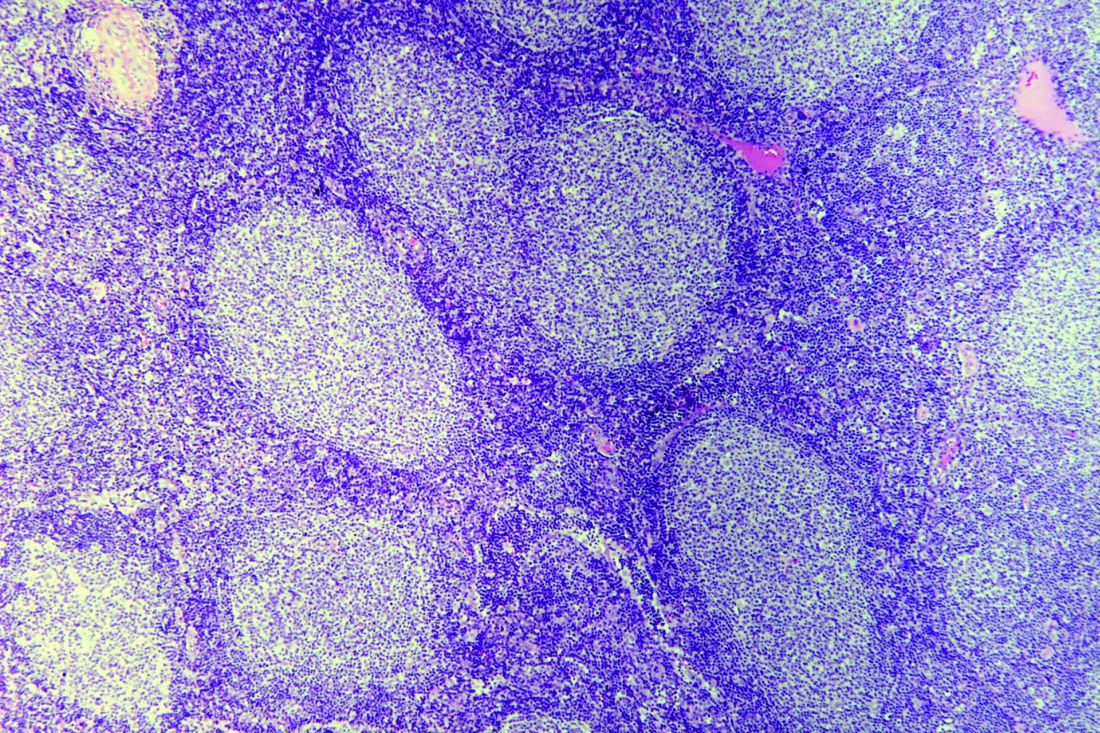
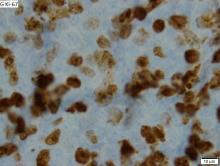
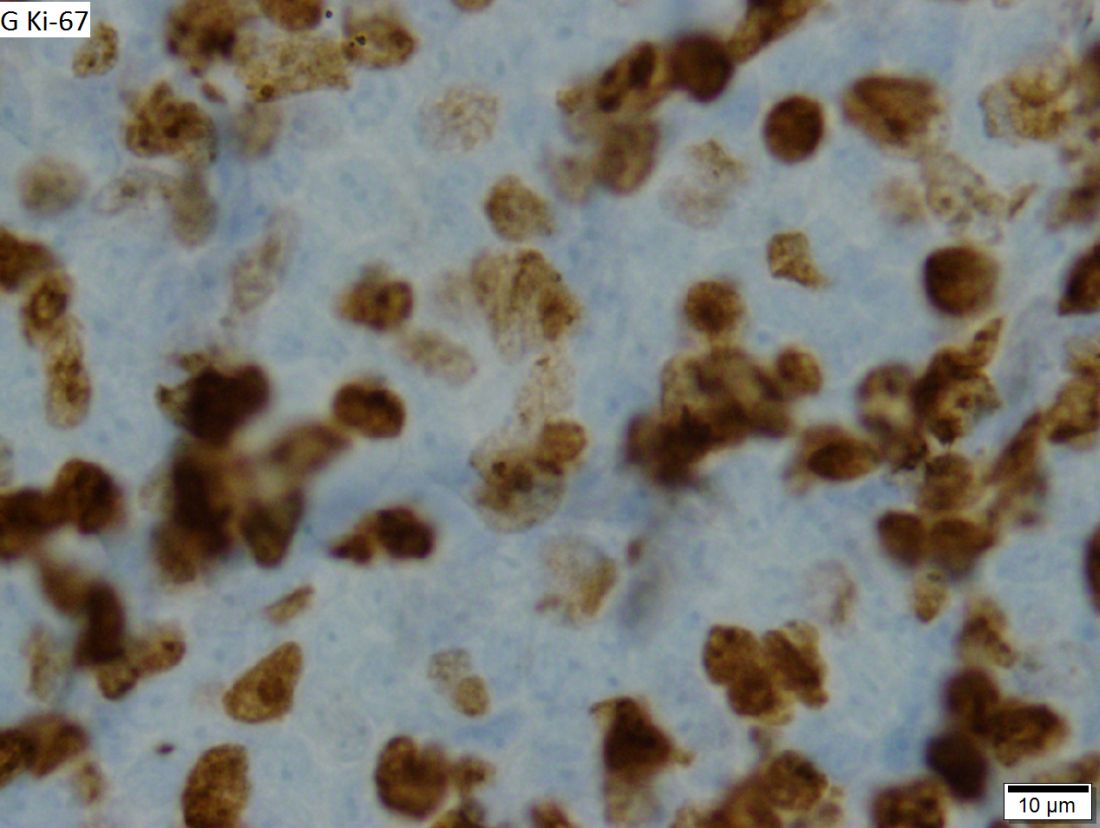
Rituximab boosts survival in primary CNS lymphoma
For patients with primary central nervous system lymphoma (PCNSL), adding rituximab to combination high-dose methotrexate and temozolomide significantly boosted the 5-year overall survival rate, according to a retrospective study.
The triplet combination could be a safe and effective first-line option for patients with PCNSL, particularly the frail and elderly, who may have issues with toxicity when receiving current standard care, reported lead author Cui Chen, MD, of Sun Yat-Sen University Cancer Center in Guangzhou, China, and his colleagues.
“An increasing number of studies and meta‐analyses have investigated the effect of rituximab in PCNSL, indicating that rituximab can robustly enhance the response rate and possibly improve survival,” the investigators wrote in Cancer Medicine. “However, data regarding the addition of rituximab to [methotrexate and temozolomide] for PCNSL are limited, and no study has directly compared the efficacy of [rituximab/high-dose methotrexate/temozolomide] to that of [high-dose methotrexate/temozolomide].”
The study involved 62 patients with untreated PCNSL who were diagnosed between 2005 and 2015. Out of the 62 patients, 32 received rituximab/high-dose methotrexate/temozolomide (RMT) and 30 received high-dose methotrexate/temozolomide (MT). Patients received up to eight cycles of therapy, with discontinuation upon disease progression or toxicity.
The results showed that patients treated with RMT had significantly better outcomes than those who received MT, first marked by objective response rates, which were 93.7% for RMT and 69.0% for MT.
Survival rates also showed the advantage of rituximab. For the RMT group, 2-year and 5-year progression-free survival rates were 81.3% and 53.3%, respectively, compared with 46.5% and 29.1% for patients receiving MT.
Most importantly, rituximab boosted overall survival to a significant and notable extent, with higher rates at 2 years (82.3% vs. 65.7%) and 5 years (82.3% vs. 50.0%).
Efficacy did not diminish safety, as no significant differences in toxicity were found between treatment types. The most common grade 3-4 toxicities were hematologic; most commonly, this entailed neutropenia, which occurred in about one-quarter of patients.
“Given its outstanding efficacy and favorable toxicity, we consider RMT to be a feasible and safe therapeutic approach as a first‐line treatment for PCNSL. Moreover, RMT is an ideal regimen for elderly patients and frail populations who may not tolerate [whole‐brain radiation therapy] or [autologous stem‐cell transplantation],” the researchers wrote.
The study was funded by the Natural Science Foundation of Guangdong Province. The researchers reported having no conflicts of interest.
SOURCE: Chen C et al. Cancer Med. 2019 Mar 1. doi: 10.1002/cam4.1906.
For patients with primary central nervous system lymphoma (PCNSL), adding rituximab to combination high-dose methotrexate and temozolomide significantly boosted the 5-year overall survival rate, according to a retrospective study.
The triplet combination could be a safe and effective first-line option for patients with PCNSL, particularly the frail and elderly, who may have issues with toxicity when receiving current standard care, reported lead author Cui Chen, MD, of Sun Yat-Sen University Cancer Center in Guangzhou, China, and his colleagues.
“An increasing number of studies and meta‐analyses have investigated the effect of rituximab in PCNSL, indicating that rituximab can robustly enhance the response rate and possibly improve survival,” the investigators wrote in Cancer Medicine. “However, data regarding the addition of rituximab to [methotrexate and temozolomide] for PCNSL are limited, and no study has directly compared the efficacy of [rituximab/high-dose methotrexate/temozolomide] to that of [high-dose methotrexate/temozolomide].”
The study involved 62 patients with untreated PCNSL who were diagnosed between 2005 and 2015. Out of the 62 patients, 32 received rituximab/high-dose methotrexate/temozolomide (RMT) and 30 received high-dose methotrexate/temozolomide (MT). Patients received up to eight cycles of therapy, with discontinuation upon disease progression or toxicity.
The results showed that patients treated with RMT had significantly better outcomes than those who received MT, first marked by objective response rates, which were 93.7% for RMT and 69.0% for MT.
Survival rates also showed the advantage of rituximab. For the RMT group, 2-year and 5-year progression-free survival rates were 81.3% and 53.3%, respectively, compared with 46.5% and 29.1% for patients receiving MT.
Most importantly, rituximab boosted overall survival to a significant and notable extent, with higher rates at 2 years (82.3% vs. 65.7%) and 5 years (82.3% vs. 50.0%).
Efficacy did not diminish safety, as no significant differences in toxicity were found between treatment types. The most common grade 3-4 toxicities were hematologic; most commonly, this entailed neutropenia, which occurred in about one-quarter of patients.
“Given its outstanding efficacy and favorable toxicity, we consider RMT to be a feasible and safe therapeutic approach as a first‐line treatment for PCNSL. Moreover, RMT is an ideal regimen for elderly patients and frail populations who may not tolerate [whole‐brain radiation therapy] or [autologous stem‐cell transplantation],” the researchers wrote.
The study was funded by the Natural Science Foundation of Guangdong Province. The researchers reported having no conflicts of interest.
SOURCE: Chen C et al. Cancer Med. 2019 Mar 1. doi: 10.1002/cam4.1906.
For patients with primary central nervous system lymphoma (PCNSL), adding rituximab to combination high-dose methotrexate and temozolomide significantly boosted the 5-year overall survival rate, according to a retrospective study.
The triplet combination could be a safe and effective first-line option for patients with PCNSL, particularly the frail and elderly, who may have issues with toxicity when receiving current standard care, reported lead author Cui Chen, MD, of Sun Yat-Sen University Cancer Center in Guangzhou, China, and his colleagues.
“An increasing number of studies and meta‐analyses have investigated the effect of rituximab in PCNSL, indicating that rituximab can robustly enhance the response rate and possibly improve survival,” the investigators wrote in Cancer Medicine. “However, data regarding the addition of rituximab to [methotrexate and temozolomide] for PCNSL are limited, and no study has directly compared the efficacy of [rituximab/high-dose methotrexate/temozolomide] to that of [high-dose methotrexate/temozolomide].”
The study involved 62 patients with untreated PCNSL who were diagnosed between 2005 and 2015. Out of the 62 patients, 32 received rituximab/high-dose methotrexate/temozolomide (RMT) and 30 received high-dose methotrexate/temozolomide (MT). Patients received up to eight cycles of therapy, with discontinuation upon disease progression or toxicity.
The results showed that patients treated with RMT had significantly better outcomes than those who received MT, first marked by objective response rates, which were 93.7% for RMT and 69.0% for MT.
Survival rates also showed the advantage of rituximab. For the RMT group, 2-year and 5-year progression-free survival rates were 81.3% and 53.3%, respectively, compared with 46.5% and 29.1% for patients receiving MT.
Most importantly, rituximab boosted overall survival to a significant and notable extent, with higher rates at 2 years (82.3% vs. 65.7%) and 5 years (82.3% vs. 50.0%).
Efficacy did not diminish safety, as no significant differences in toxicity were found between treatment types. The most common grade 3-4 toxicities were hematologic; most commonly, this entailed neutropenia, which occurred in about one-quarter of patients.
“Given its outstanding efficacy and favorable toxicity, we consider RMT to be a feasible and safe therapeutic approach as a first‐line treatment for PCNSL. Moreover, RMT is an ideal regimen for elderly patients and frail populations who may not tolerate [whole‐brain radiation therapy] or [autologous stem‐cell transplantation],” the researchers wrote.
The study was funded by the Natural Science Foundation of Guangdong Province. The researchers reported having no conflicts of interest.
SOURCE: Chen C et al. Cancer Med. 2019 Mar 1. doi: 10.1002/cam4.1906.
FROM CANCER MEDICINE
High survival in relapsed FL after primary radiotherapy
The prognosis post relapse following primary radiotherapy was found to be excellent for patients with localized follicular lymphoma (FL), according to a retrospective analysis.
But patients who experienced early relapse – at 1 year or less after diagnosis – had significantly worse survival.
While primary radiotherapy may be curative for localized FL, about 30%-50% of patients will relapse and optimal salvage therapy is not well defined, Michael S. Binkley, MD, of Stanford (Calif.) University, and his colleagues wrote in the International Journal of Radiation Oncology, Biology, Physics.
The researchers retrospectively studied 512 patients with localized FL using data from multiple centers under the direction of the International Lymphoma Radiation Oncology Group (ILROG). Clinical information was collected, including method of detection, age at relapse, location of recurrence, and response to salvage therapy.
The team defined disease recurrence as lymphoma nonresponsive to primary radiotherapy inside of the prescribed dose volume, which included having no radiographic or clinical response within 6 months of initial radiotherapy treatment.
With a median follow-up of 52 months, Dr. Binkley and his colleagues found that 29.1% of patients developed relapsed lymphoma at a median 23 months (range, 1-143 months) following primary radiotherapy.
The team reported that the 3-year overall survival rate was 91.4% for patients with lymphoma recurrence after primary radiotherapy. In total, 16 deaths occurred during follow-up: eight were lymphoma specific deaths, three were from other causes, and in five patients the cause was unknown.
Of the 149 cases of relapsed lymphoma, 93 were indolent. There were also three cases of FL grade 3B/not otherwise specified and 18 cases of diffuse large B-cell lymphoma. In 35 patients who relapsed, biopsies were not performed.
“The excellent prognosis observed for this relapse cohort emphasizes that primary radiation for localized follicular lymphoma is an excellent treatment option,” the researchers wrote.
When the researchers examined survival based on the time of relapse, they found that overall survival was “significantly worse” for patients who had relapsed 12 months or less after the date of diagnosis, at 88.7%, compared with all others at 95.8% (P = .01).
They found no difference in overall survival between patients who received immediate salvage treatment, compared with observation after relapse (P = .28). There was also no significant difference in survival between patients who relapsed in 1 year or less but underwent immediate treatment, compared to early relapsed patients who were observed (log-rank P = .34).
“[A]ny decision to offer treatment at time of relapse must be weighed with the risk of acute and late adverse effects. Greater than 60% of patients in our cohort with indolent recurrence who underwent salvage treatment received rituximab, likely contributing to the excellent outcomes,” the researchers wrote. “However, nearly 60% of patients with indolent recurrence who were observed did not have disease progression nor [did they] receive treatment within 3 years.”
The researchers reported having no conflicts of interest.
SOURCE: Binkley MS et al. Int J Radiat Oncol Biol Phys. 2019 Mar 8. doi: 10.1016/j.ijrobp.2019.03.004.
The prognosis post relapse following primary radiotherapy was found to be excellent for patients with localized follicular lymphoma (FL), according to a retrospective analysis.
But patients who experienced early relapse – at 1 year or less after diagnosis – had significantly worse survival.
While primary radiotherapy may be curative for localized FL, about 30%-50% of patients will relapse and optimal salvage therapy is not well defined, Michael S. Binkley, MD, of Stanford (Calif.) University, and his colleagues wrote in the International Journal of Radiation Oncology, Biology, Physics.
The researchers retrospectively studied 512 patients with localized FL using data from multiple centers under the direction of the International Lymphoma Radiation Oncology Group (ILROG). Clinical information was collected, including method of detection, age at relapse, location of recurrence, and response to salvage therapy.
The team defined disease recurrence as lymphoma nonresponsive to primary radiotherapy inside of the prescribed dose volume, which included having no radiographic or clinical response within 6 months of initial radiotherapy treatment.
With a median follow-up of 52 months, Dr. Binkley and his colleagues found that 29.1% of patients developed relapsed lymphoma at a median 23 months (range, 1-143 months) following primary radiotherapy.
The team reported that the 3-year overall survival rate was 91.4% for patients with lymphoma recurrence after primary radiotherapy. In total, 16 deaths occurred during follow-up: eight were lymphoma specific deaths, three were from other causes, and in five patients the cause was unknown.
Of the 149 cases of relapsed lymphoma, 93 were indolent. There were also three cases of FL grade 3B/not otherwise specified and 18 cases of diffuse large B-cell lymphoma. In 35 patients who relapsed, biopsies were not performed.
“The excellent prognosis observed for this relapse cohort emphasizes that primary radiation for localized follicular lymphoma is an excellent treatment option,” the researchers wrote.
When the researchers examined survival based on the time of relapse, they found that overall survival was “significantly worse” for patients who had relapsed 12 months or less after the date of diagnosis, at 88.7%, compared with all others at 95.8% (P = .01).
They found no difference in overall survival between patients who received immediate salvage treatment, compared with observation after relapse (P = .28). There was also no significant difference in survival between patients who relapsed in 1 year or less but underwent immediate treatment, compared to early relapsed patients who were observed (log-rank P = .34).
“[A]ny decision to offer treatment at time of relapse must be weighed with the risk of acute and late adverse effects. Greater than 60% of patients in our cohort with indolent recurrence who underwent salvage treatment received rituximab, likely contributing to the excellent outcomes,” the researchers wrote. “However, nearly 60% of patients with indolent recurrence who were observed did not have disease progression nor [did they] receive treatment within 3 years.”
The researchers reported having no conflicts of interest.
SOURCE: Binkley MS et al. Int J Radiat Oncol Biol Phys. 2019 Mar 8. doi: 10.1016/j.ijrobp.2019.03.004.
The prognosis post relapse following primary radiotherapy was found to be excellent for patients with localized follicular lymphoma (FL), according to a retrospective analysis.
But patients who experienced early relapse – at 1 year or less after diagnosis – had significantly worse survival.
While primary radiotherapy may be curative for localized FL, about 30%-50% of patients will relapse and optimal salvage therapy is not well defined, Michael S. Binkley, MD, of Stanford (Calif.) University, and his colleagues wrote in the International Journal of Radiation Oncology, Biology, Physics.
The researchers retrospectively studied 512 patients with localized FL using data from multiple centers under the direction of the International Lymphoma Radiation Oncology Group (ILROG). Clinical information was collected, including method of detection, age at relapse, location of recurrence, and response to salvage therapy.
The team defined disease recurrence as lymphoma nonresponsive to primary radiotherapy inside of the prescribed dose volume, which included having no radiographic or clinical response within 6 months of initial radiotherapy treatment.
With a median follow-up of 52 months, Dr. Binkley and his colleagues found that 29.1% of patients developed relapsed lymphoma at a median 23 months (range, 1-143 months) following primary radiotherapy.
The team reported that the 3-year overall survival rate was 91.4% for patients with lymphoma recurrence after primary radiotherapy. In total, 16 deaths occurred during follow-up: eight were lymphoma specific deaths, three were from other causes, and in five patients the cause was unknown.
Of the 149 cases of relapsed lymphoma, 93 were indolent. There were also three cases of FL grade 3B/not otherwise specified and 18 cases of diffuse large B-cell lymphoma. In 35 patients who relapsed, biopsies were not performed.
“The excellent prognosis observed for this relapse cohort emphasizes that primary radiation for localized follicular lymphoma is an excellent treatment option,” the researchers wrote.
When the researchers examined survival based on the time of relapse, they found that overall survival was “significantly worse” for patients who had relapsed 12 months or less after the date of diagnosis, at 88.7%, compared with all others at 95.8% (P = .01).
They found no difference in overall survival between patients who received immediate salvage treatment, compared with observation after relapse (P = .28). There was also no significant difference in survival between patients who relapsed in 1 year or less but underwent immediate treatment, compared to early relapsed patients who were observed (log-rank P = .34).
“[A]ny decision to offer treatment at time of relapse must be weighed with the risk of acute and late adverse effects. Greater than 60% of patients in our cohort with indolent recurrence who underwent salvage treatment received rituximab, likely contributing to the excellent outcomes,” the researchers wrote. “However, nearly 60% of patients with indolent recurrence who were observed did not have disease progression nor [did they] receive treatment within 3 years.”
The researchers reported having no conflicts of interest.
SOURCE: Binkley MS et al. Int J Radiat Oncol Biol Phys. 2019 Mar 8. doi: 10.1016/j.ijrobp.2019.03.004.
FROM THE INTERNATIONAL JOURNAL OF RADIATION ONCOLOGY, BIOLOGY, PHYSICS
MCL survival rates improve with novel agents
Survival outcomes for patients with mantle cell lymphoma (MCL) substantially improved from 1995 to 2013, particularly for those with advanced-stage tumors, according to a retrospective analysis.
The median overall survival for the study period was 52 months and 57 months in two cancer databases.
“Over the past 20 years, many novel agents and treatment regimens have been developed to treat MCL,” Shuangshuang Fu, PhD, of the University of Texas, Houston, and her colleagues wrote in Cancer Epidemiology.
The researchers retrospectively studied population-based data from two separate databases: the national Surveillance, Epidemiology and End Results (SEER) database and the Texas Cancer Registry (TCR). They identified all adult patients who received a new diagnosis of MCL between Jan. 1, 1995, and Dec. 31, 2013.
A total of 9,610 patients were included in the study: 7,555 patients from SEER and 2,055 from the TCR. The team collected data related to MCL diagnosis, mortality, and other variables, including age at diagnosis, marital status, sex, and tumor stage.
In total, 76.2% and 61.6% of patients from the SEER and TCR databases, respectively, had an advanced-stage tumor.
Dr. Fu and her colleagues found that all-cause mortality rates in both groups were significantly reduced from 1995 to 2013 (SEER, P less than .001; TCR, P = .03).
In addition, the team reported that the median overall survival time for all patients in the SEER database was 52 months, and it was 57 months for the TCR database.
“MCL patients with [an] advanced stage tumor benefitted most from the introduction of newly developed regimens,” they added.
The researchers acknowledged that a key limitation of the study was the inability to assess treatment regimen–specific survival, which could only be estimated with these data.
“The findings of our study further confirmed the impact of novel agents on improved survival over time that was shown in other studies,” they wrote.
The study was supported by grant funding from the Cancer Prevention Research Institute of Texas and the National Institutes of Health. The researchers reported having no conflicts of interest.
SOURCE: Fu S et al. Cancer Epidemiol. 2019 Feb;58:89-97.
Survival outcomes for patients with mantle cell lymphoma (MCL) substantially improved from 1995 to 2013, particularly for those with advanced-stage tumors, according to a retrospective analysis.
The median overall survival for the study period was 52 months and 57 months in two cancer databases.
“Over the past 20 years, many novel agents and treatment regimens have been developed to treat MCL,” Shuangshuang Fu, PhD, of the University of Texas, Houston, and her colleagues wrote in Cancer Epidemiology.
The researchers retrospectively studied population-based data from two separate databases: the national Surveillance, Epidemiology and End Results (SEER) database and the Texas Cancer Registry (TCR). They identified all adult patients who received a new diagnosis of MCL between Jan. 1, 1995, and Dec. 31, 2013.
A total of 9,610 patients were included in the study: 7,555 patients from SEER and 2,055 from the TCR. The team collected data related to MCL diagnosis, mortality, and other variables, including age at diagnosis, marital status, sex, and tumor stage.
In total, 76.2% and 61.6% of patients from the SEER and TCR databases, respectively, had an advanced-stage tumor.
Dr. Fu and her colleagues found that all-cause mortality rates in both groups were significantly reduced from 1995 to 2013 (SEER, P less than .001; TCR, P = .03).
In addition, the team reported that the median overall survival time for all patients in the SEER database was 52 months, and it was 57 months for the TCR database.
“MCL patients with [an] advanced stage tumor benefitted most from the introduction of newly developed regimens,” they added.
The researchers acknowledged that a key limitation of the study was the inability to assess treatment regimen–specific survival, which could only be estimated with these data.
“The findings of our study further confirmed the impact of novel agents on improved survival over time that was shown in other studies,” they wrote.
The study was supported by grant funding from the Cancer Prevention Research Institute of Texas and the National Institutes of Health. The researchers reported having no conflicts of interest.
SOURCE: Fu S et al. Cancer Epidemiol. 2019 Feb;58:89-97.
Survival outcomes for patients with mantle cell lymphoma (MCL) substantially improved from 1995 to 2013, particularly for those with advanced-stage tumors, according to a retrospective analysis.
The median overall survival for the study period was 52 months and 57 months in two cancer databases.
“Over the past 20 years, many novel agents and treatment regimens have been developed to treat MCL,” Shuangshuang Fu, PhD, of the University of Texas, Houston, and her colleagues wrote in Cancer Epidemiology.
The researchers retrospectively studied population-based data from two separate databases: the national Surveillance, Epidemiology and End Results (SEER) database and the Texas Cancer Registry (TCR). They identified all adult patients who received a new diagnosis of MCL between Jan. 1, 1995, and Dec. 31, 2013.
A total of 9,610 patients were included in the study: 7,555 patients from SEER and 2,055 from the TCR. The team collected data related to MCL diagnosis, mortality, and other variables, including age at diagnosis, marital status, sex, and tumor stage.
In total, 76.2% and 61.6% of patients from the SEER and TCR databases, respectively, had an advanced-stage tumor.
Dr. Fu and her colleagues found that all-cause mortality rates in both groups were significantly reduced from 1995 to 2013 (SEER, P less than .001; TCR, P = .03).
In addition, the team reported that the median overall survival time for all patients in the SEER database was 52 months, and it was 57 months for the TCR database.
“MCL patients with [an] advanced stage tumor benefitted most from the introduction of newly developed regimens,” they added.
The researchers acknowledged that a key limitation of the study was the inability to assess treatment regimen–specific survival, which could only be estimated with these data.
“The findings of our study further confirmed the impact of novel agents on improved survival over time that was shown in other studies,” they wrote.
The study was supported by grant funding from the Cancer Prevention Research Institute of Texas and the National Institutes of Health. The researchers reported having no conflicts of interest.
SOURCE: Fu S et al. Cancer Epidemiol. 2019 Feb;58:89-97.
FROM CANCER EPIDEMIOLOGY
Worse survival seen among black patients with MCL
Black non-Hispanic patients with mantle cell lymphoma (MCL) have a lower rate of 5-year overall survival, compared with white non-Hispanic and Hispanic patients, according to a retrospective analysis of more than 18,000 cases.
However, black patients were also most likely to receive treatment at an academic center, which was an independent predictor of better survival, reported Nikesh N. Shah, MD, of Emory University, Atlanta, and his colleagues. This finding suggests that even academic centers still need to focus on overcoming demographic disparities.
“Racial and socioeconomic differences have been reported in many malignancies and certain lymphomas; however, few studies report on disparities in MCL,” the investigators wrote in Clinical Lymphoma, Myeloma & Leukemia. “To our knowledge this is the first such study to assess racial and socioeconomic disparities in this disease.”
The investigators reviewed 18,120 patients with MCL diagnosed between 2004 and 2013; data were drawn from the National Cancer Database. The primary endpoint was overall survival from the time of diagnosis, with analyses conducted to assess various associations with race/ethnicity, facility type, clinical/tumor characteristics, cancer stage, insurance type, and other factors.
Results showed that Hispanic patients had the highest rate of overall survival, at 55.8%, followed by white patients, at 50.1%. Trailing behind these groups were black patients (46.8%) and patients of other races/ethnicities (46.0%).
Along with survival disparities, race/ethnicity was tied to certain clinical and treatment characteristics. Compared with white patients, black patients were more likely to experience B symptoms (28% vs. 25%) and have Medicaid or lack insurance (15% vs. 5%). Black and Hispanic patients were also less likely than white non-Hispanic patients to receive stem cell transplant (13% vs. 10% vs. 10%).
Although black patients were more likely than white patients to receive treatment at an academic center (51% vs. 38%), a factor independently associated with best survival among center types, whatever advantage provided apparently did not exceed disadvantages associated with race.
“We report inferior overall survival in black patients after accounting for socioeconomic status, as seen in other malignancies,” the investigators wrote. “Surprisingly, these patients were more likely to be treated at academic centers, which independently showed improved overall survival in multivariable analysis that controlled for age, disease stage, insurance status, and other socioeconomic factors.”
The researchers cited a number of steps that could help close the survival gap, including providing more comprehensive supportive care between physician visits and enrollment of patients from diverse racial background on clinical trials.
The study was funded by the National Institutes of Health. The researchers reported having no conflicts of interest.
SOURCE: Shah NN et al. Clin Lymphoma Myeloma Leuk. 2019 Mar 11. doi: 10.1016/j.clml.2019.03.006.
Black non-Hispanic patients with mantle cell lymphoma (MCL) have a lower rate of 5-year overall survival, compared with white non-Hispanic and Hispanic patients, according to a retrospective analysis of more than 18,000 cases.
However, black patients were also most likely to receive treatment at an academic center, which was an independent predictor of better survival, reported Nikesh N. Shah, MD, of Emory University, Atlanta, and his colleagues. This finding suggests that even academic centers still need to focus on overcoming demographic disparities.
“Racial and socioeconomic differences have been reported in many malignancies and certain lymphomas; however, few studies report on disparities in MCL,” the investigators wrote in Clinical Lymphoma, Myeloma & Leukemia. “To our knowledge this is the first such study to assess racial and socioeconomic disparities in this disease.”
The investigators reviewed 18,120 patients with MCL diagnosed between 2004 and 2013; data were drawn from the National Cancer Database. The primary endpoint was overall survival from the time of diagnosis, with analyses conducted to assess various associations with race/ethnicity, facility type, clinical/tumor characteristics, cancer stage, insurance type, and other factors.
Results showed that Hispanic patients had the highest rate of overall survival, at 55.8%, followed by white patients, at 50.1%. Trailing behind these groups were black patients (46.8%) and patients of other races/ethnicities (46.0%).
Along with survival disparities, race/ethnicity was tied to certain clinical and treatment characteristics. Compared with white patients, black patients were more likely to experience B symptoms (28% vs. 25%) and have Medicaid or lack insurance (15% vs. 5%). Black and Hispanic patients were also less likely than white non-Hispanic patients to receive stem cell transplant (13% vs. 10% vs. 10%).
Although black patients were more likely than white patients to receive treatment at an academic center (51% vs. 38%), a factor independently associated with best survival among center types, whatever advantage provided apparently did not exceed disadvantages associated with race.
“We report inferior overall survival in black patients after accounting for socioeconomic status, as seen in other malignancies,” the investigators wrote. “Surprisingly, these patients were more likely to be treated at academic centers, which independently showed improved overall survival in multivariable analysis that controlled for age, disease stage, insurance status, and other socioeconomic factors.”
The researchers cited a number of steps that could help close the survival gap, including providing more comprehensive supportive care between physician visits and enrollment of patients from diverse racial background on clinical trials.
The study was funded by the National Institutes of Health. The researchers reported having no conflicts of interest.
SOURCE: Shah NN et al. Clin Lymphoma Myeloma Leuk. 2019 Mar 11. doi: 10.1016/j.clml.2019.03.006.
Black non-Hispanic patients with mantle cell lymphoma (MCL) have a lower rate of 5-year overall survival, compared with white non-Hispanic and Hispanic patients, according to a retrospective analysis of more than 18,000 cases.
However, black patients were also most likely to receive treatment at an academic center, which was an independent predictor of better survival, reported Nikesh N. Shah, MD, of Emory University, Atlanta, and his colleagues. This finding suggests that even academic centers still need to focus on overcoming demographic disparities.
“Racial and socioeconomic differences have been reported in many malignancies and certain lymphomas; however, few studies report on disparities in MCL,” the investigators wrote in Clinical Lymphoma, Myeloma & Leukemia. “To our knowledge this is the first such study to assess racial and socioeconomic disparities in this disease.”
The investigators reviewed 18,120 patients with MCL diagnosed between 2004 and 2013; data were drawn from the National Cancer Database. The primary endpoint was overall survival from the time of diagnosis, with analyses conducted to assess various associations with race/ethnicity, facility type, clinical/tumor characteristics, cancer stage, insurance type, and other factors.
Results showed that Hispanic patients had the highest rate of overall survival, at 55.8%, followed by white patients, at 50.1%. Trailing behind these groups were black patients (46.8%) and patients of other races/ethnicities (46.0%).
Along with survival disparities, race/ethnicity was tied to certain clinical and treatment characteristics. Compared with white patients, black patients were more likely to experience B symptoms (28% vs. 25%) and have Medicaid or lack insurance (15% vs. 5%). Black and Hispanic patients were also less likely than white non-Hispanic patients to receive stem cell transplant (13% vs. 10% vs. 10%).
Although black patients were more likely than white patients to receive treatment at an academic center (51% vs. 38%), a factor independently associated with best survival among center types, whatever advantage provided apparently did not exceed disadvantages associated with race.
“We report inferior overall survival in black patients after accounting for socioeconomic status, as seen in other malignancies,” the investigators wrote. “Surprisingly, these patients were more likely to be treated at academic centers, which independently showed improved overall survival in multivariable analysis that controlled for age, disease stage, insurance status, and other socioeconomic factors.”
The researchers cited a number of steps that could help close the survival gap, including providing more comprehensive supportive care between physician visits and enrollment of patients from diverse racial background on clinical trials.
The study was funded by the National Institutes of Health. The researchers reported having no conflicts of interest.
SOURCE: Shah NN et al. Clin Lymphoma Myeloma Leuk. 2019 Mar 11. doi: 10.1016/j.clml.2019.03.006.
FROM CLINICAL LYMPHOMA, MYELOMA & LEUKEMIA