User login
ACIP recommends meningococcal vaccine for at-risk infants
The quadrivalent meningococcal vaccine MenACWY-CRM, or Menveo, may now be given to infants aged 2-23 months who are considered at high risk for meningococcal disease or who travel to areas where it is hyperendemic or epidemic, according to the recommendations of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
High-risk children are those with component complement deficiency, HIV infection, or anatomic or functional asplenia. Children in an outbreak of a vaccine-preventable meningitis serogroup also should receive the series. About 5,000 infants per year fit into these categories.
The Advisory Committee on Immunization Practices (ACIP) approved the decision by a vote of 13-1, with one abstention. The decision aligns ACIP’s recommendation with the recently expanded indication for the vaccine, which protects against serotypes A, C, W-135, and Y. In August, the Food and Drug Administration approved the vaccine for infants and toddlers older than 2 months.
However, because of the extremely low incidence of meningococcal disease in children in the United States, the committee did not recommend it for all healthy children. "Very few children are considered at increased risk, so a routine recommendation would prevent just a very low number of cases," said Jessica MacNeil, an epidemiologist with the CDC.
The FDA licensing of MenACWY-CRM and ACIP’s recommendation give pediatricians a third option for infant meningococcal vaccination. HibMenCY-TT (MenHibrix) and MenACWY-D (Menactra) are also approved for some of these same indications. However, Ms. MacNeil noted, Menactra cannot be used in children with asplenia, and MenHibrix can’t be used for travel or as a booster dose.
The committee based its decision on three phase III safety and efficacy trials. These studies comprised nearly 11,000 infants – 9,000 of whom had the four-dose series and 2,000 of whom had a two-dose series, said Dr. Peter Dull of Novartis Vaccines.
The studies confirmed an average 90% efficacy against all four serotypes after the 12-month dose. By 40 months of age, efficacy had waned somewhat: 10% of infants had a seroresponse to type A; 34%, to type C; 76%, to type W-135; and 67%, to type Y. Of the strains included in the vaccine, types C, Y, and W-135 cause 75% of meningococcal disease in those aged 11 years and older.
Dr. Dull said that Novartis is following a cohort that received the vaccine as infants; next year, 60-month immunogenicity data will be available.
After adjustment for some confounders, the vaccine did not significantly interact with the immunogenic potential of other recommended childhood vaccines. Novartis studies also found it safe. Up to 60% of children had mild to moderate injection site reactions. The incidence of systemic reactions was no different from that for routine vaccines alone.
There were 11 serious adverse events possibly related to the vaccine among the 5,000 children included in the safety analyses. These included acute encephalomyelitis, cellulitis, complex partial seizure, epilepsy, febrile seizure, and Kawasaki disease. Ten deaths occurred, but were not considered vaccine related.
The recommended dosing schedule will be:
• Aged 2-6 months: Four doses at 2, 4, 6, and 12 months.
• Aged 7-23 months: Two doses with the second dose given in the second year of life.
• Aged 2-11 years: One or two doses.
Boosters should be given to those who remain at risk, beginning at 3 years after the primary series and then every 5 years thereafter.
The committee also agreed, by a vote of 14 with one abstention, to include MenACWY-CRM in the Vaccines for Children program.
One member who voted disclosed that her institution receives research funding from drug companies. None of the other voting members had any relevant financial disclosures.
The quadrivalent meningococcal vaccine MenACWY-CRM, or Menveo, may now be given to infants aged 2-23 months who are considered at high risk for meningococcal disease or who travel to areas where it is hyperendemic or epidemic, according to the recommendations of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
High-risk children are those with component complement deficiency, HIV infection, or anatomic or functional asplenia. Children in an outbreak of a vaccine-preventable meningitis serogroup also should receive the series. About 5,000 infants per year fit into these categories.
The Advisory Committee on Immunization Practices (ACIP) approved the decision by a vote of 13-1, with one abstention. The decision aligns ACIP’s recommendation with the recently expanded indication for the vaccine, which protects against serotypes A, C, W-135, and Y. In August, the Food and Drug Administration approved the vaccine for infants and toddlers older than 2 months.
However, because of the extremely low incidence of meningococcal disease in children in the United States, the committee did not recommend it for all healthy children. "Very few children are considered at increased risk, so a routine recommendation would prevent just a very low number of cases," said Jessica MacNeil, an epidemiologist with the CDC.
The FDA licensing of MenACWY-CRM and ACIP’s recommendation give pediatricians a third option for infant meningococcal vaccination. HibMenCY-TT (MenHibrix) and MenACWY-D (Menactra) are also approved for some of these same indications. However, Ms. MacNeil noted, Menactra cannot be used in children with asplenia, and MenHibrix can’t be used for travel or as a booster dose.
The committee based its decision on three phase III safety and efficacy trials. These studies comprised nearly 11,000 infants – 9,000 of whom had the four-dose series and 2,000 of whom had a two-dose series, said Dr. Peter Dull of Novartis Vaccines.
The studies confirmed an average 90% efficacy against all four serotypes after the 12-month dose. By 40 months of age, efficacy had waned somewhat: 10% of infants had a seroresponse to type A; 34%, to type C; 76%, to type W-135; and 67%, to type Y. Of the strains included in the vaccine, types C, Y, and W-135 cause 75% of meningococcal disease in those aged 11 years and older.
Dr. Dull said that Novartis is following a cohort that received the vaccine as infants; next year, 60-month immunogenicity data will be available.
After adjustment for some confounders, the vaccine did not significantly interact with the immunogenic potential of other recommended childhood vaccines. Novartis studies also found it safe. Up to 60% of children had mild to moderate injection site reactions. The incidence of systemic reactions was no different from that for routine vaccines alone.
There were 11 serious adverse events possibly related to the vaccine among the 5,000 children included in the safety analyses. These included acute encephalomyelitis, cellulitis, complex partial seizure, epilepsy, febrile seizure, and Kawasaki disease. Ten deaths occurred, but were not considered vaccine related.
The recommended dosing schedule will be:
• Aged 2-6 months: Four doses at 2, 4, 6, and 12 months.
• Aged 7-23 months: Two doses with the second dose given in the second year of life.
• Aged 2-11 years: One or two doses.
Boosters should be given to those who remain at risk, beginning at 3 years after the primary series and then every 5 years thereafter.
The committee also agreed, by a vote of 14 with one abstention, to include MenACWY-CRM in the Vaccines for Children program.
One member who voted disclosed that her institution receives research funding from drug companies. None of the other voting members had any relevant financial disclosures.
The quadrivalent meningococcal vaccine MenACWY-CRM, or Menveo, may now be given to infants aged 2-23 months who are considered at high risk for meningococcal disease or who travel to areas where it is hyperendemic or epidemic, according to the recommendations of the Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices.
High-risk children are those with component complement deficiency, HIV infection, or anatomic or functional asplenia. Children in an outbreak of a vaccine-preventable meningitis serogroup also should receive the series. About 5,000 infants per year fit into these categories.
The Advisory Committee on Immunization Practices (ACIP) approved the decision by a vote of 13-1, with one abstention. The decision aligns ACIP’s recommendation with the recently expanded indication for the vaccine, which protects against serotypes A, C, W-135, and Y. In August, the Food and Drug Administration approved the vaccine for infants and toddlers older than 2 months.
However, because of the extremely low incidence of meningococcal disease in children in the United States, the committee did not recommend it for all healthy children. "Very few children are considered at increased risk, so a routine recommendation would prevent just a very low number of cases," said Jessica MacNeil, an epidemiologist with the CDC.
The FDA licensing of MenACWY-CRM and ACIP’s recommendation give pediatricians a third option for infant meningococcal vaccination. HibMenCY-TT (MenHibrix) and MenACWY-D (Menactra) are also approved for some of these same indications. However, Ms. MacNeil noted, Menactra cannot be used in children with asplenia, and MenHibrix can’t be used for travel or as a booster dose.
The committee based its decision on three phase III safety and efficacy trials. These studies comprised nearly 11,000 infants – 9,000 of whom had the four-dose series and 2,000 of whom had a two-dose series, said Dr. Peter Dull of Novartis Vaccines.
The studies confirmed an average 90% efficacy against all four serotypes after the 12-month dose. By 40 months of age, efficacy had waned somewhat: 10% of infants had a seroresponse to type A; 34%, to type C; 76%, to type W-135; and 67%, to type Y. Of the strains included in the vaccine, types C, Y, and W-135 cause 75% of meningococcal disease in those aged 11 years and older.
Dr. Dull said that Novartis is following a cohort that received the vaccine as infants; next year, 60-month immunogenicity data will be available.
After adjustment for some confounders, the vaccine did not significantly interact with the immunogenic potential of other recommended childhood vaccines. Novartis studies also found it safe. Up to 60% of children had mild to moderate injection site reactions. The incidence of systemic reactions was no different from that for routine vaccines alone.
There were 11 serious adverse events possibly related to the vaccine among the 5,000 children included in the safety analyses. These included acute encephalomyelitis, cellulitis, complex partial seizure, epilepsy, febrile seizure, and Kawasaki disease. Ten deaths occurred, but were not considered vaccine related.
The recommended dosing schedule will be:
• Aged 2-6 months: Four doses at 2, 4, 6, and 12 months.
• Aged 7-23 months: Two doses with the second dose given in the second year of life.
• Aged 2-11 years: One or two doses.
Boosters should be given to those who remain at risk, beginning at 3 years after the primary series and then every 5 years thereafter.
The committee also agreed, by a vote of 14 with one abstention, to include MenACWY-CRM in the Vaccines for Children program.
One member who voted disclosed that her institution receives research funding from drug companies. None of the other voting members had any relevant financial disclosures.
AT ACIP
Colorectal surgeons post better rectal surgery outcomes than general surgeons
WASHINGTON – Although general surgeons perform the majority of rectal resections in the United States, their patients have more severe postoperative complications, longer lengths of stay, higher hospital costs, and higher mortality rates than do those of colorectal surgeons.
A review of more than 60,000 cases over a 6-year period found that patients who had been operated on by a general surgeon had a 42% increase in the risk of mortality. They stayed in the hospital an average 1 day longer than the patients of colorectal surgeons, and had bills more than $1,000 higher. Although their overall complication rate was lower, general surgeons’ patients tended to have more severe complications, including pulmonary complications and surgical site infections, Dr. Marc Casasanta said at the annual clinical congress of the American College of Surgeons.
Dr. Casasanta of the University of Texas, Houston, extracted his data from the Premier Perspective database, the largest inpatient database in the United States. His analysis included 60,412 nonemergent rectal resections performed from 2005 to 2011.
Most of these (74%) were performed by general surgeons, with colorectal surgeons performing the remainder. Adverse outcomes examined included length and cost of hospital admission, overall complications and severe complications, and mortality.
There were several significant differences in the patient populations. General surgeons had older patients (63 vs. 61 years), and their patients tended to be sicker, with less minor disease severity (31% vs. 35%) and more major (29% vs. 17%) and extreme disease severity (8% vs. 6%).
Colorectal surgeons treated significantly more patients with inflammatory bowel disease (10% vs. 4%). They worked exclusively in urban hospitals, compared with 87% of general surgeons. Half of colorectal surgeons worked in teaching hospitals versus 37% of general surgeons.
The mean overall length of stay was a little more than 8 days. The mean hospital cost was $19,093.The overall complication rate was 29%, and mortality was 1.5%.
Complications varied significantly by surgeon specialty. General surgeons had significantly higher rates of digestive complications (including anastomotic problems), as well as pulmonary complications, lower-extremity thromboembolism, shock, and surgical site infections, including intra-abdominal infections.
The univariate analysis identified several patient characteristics significantly associated with poor outcomes, including age older than 62 years (odds ratio, 1.43), female gender (OR, 1.23), major disease severity (OR, 4.7), extreme disease severity (OR, 23), and a diagnosis of inflammatory bowel disease (OR, 1.41).
Other significant risk factors were being treated in a rural hospital (OR 1.29), being treated in a teaching hospital (OR, 1.08), and having a general surgeon (OR, 1.16). Having a colorectal surgeon decreased the risk of a poor outcome by 14%.
In the multivariate analysis, patients of general surgeons were 13% less likely to have any complication and 20% less likely to have ileus. But these patients were also significantly more likely to have severe complications, including pulmonary problems (13%) and surgical site infections (11%).
The length of stay was about 1 day longer, and hospitals costs were $1,122 more expensive. Patients of general surgeons were 42% more likely to die.
Dr. Casasanta said he had no relevant financial disclosures. However, Dr. Eric Haas, the primary investigator, said he has been a consultant for Applied Medical; and has received consulting fees, research funding, and honoraria from Intuitive Surgical.
WASHINGTON – Although general surgeons perform the majority of rectal resections in the United States, their patients have more severe postoperative complications, longer lengths of stay, higher hospital costs, and higher mortality rates than do those of colorectal surgeons.
A review of more than 60,000 cases over a 6-year period found that patients who had been operated on by a general surgeon had a 42% increase in the risk of mortality. They stayed in the hospital an average 1 day longer than the patients of colorectal surgeons, and had bills more than $1,000 higher. Although their overall complication rate was lower, general surgeons’ patients tended to have more severe complications, including pulmonary complications and surgical site infections, Dr. Marc Casasanta said at the annual clinical congress of the American College of Surgeons.
Dr. Casasanta of the University of Texas, Houston, extracted his data from the Premier Perspective database, the largest inpatient database in the United States. His analysis included 60,412 nonemergent rectal resections performed from 2005 to 2011.
Most of these (74%) were performed by general surgeons, with colorectal surgeons performing the remainder. Adverse outcomes examined included length and cost of hospital admission, overall complications and severe complications, and mortality.
There were several significant differences in the patient populations. General surgeons had older patients (63 vs. 61 years), and their patients tended to be sicker, with less minor disease severity (31% vs. 35%) and more major (29% vs. 17%) and extreme disease severity (8% vs. 6%).
Colorectal surgeons treated significantly more patients with inflammatory bowel disease (10% vs. 4%). They worked exclusively in urban hospitals, compared with 87% of general surgeons. Half of colorectal surgeons worked in teaching hospitals versus 37% of general surgeons.
The mean overall length of stay was a little more than 8 days. The mean hospital cost was $19,093.The overall complication rate was 29%, and mortality was 1.5%.
Complications varied significantly by surgeon specialty. General surgeons had significantly higher rates of digestive complications (including anastomotic problems), as well as pulmonary complications, lower-extremity thromboembolism, shock, and surgical site infections, including intra-abdominal infections.
The univariate analysis identified several patient characteristics significantly associated with poor outcomes, including age older than 62 years (odds ratio, 1.43), female gender (OR, 1.23), major disease severity (OR, 4.7), extreme disease severity (OR, 23), and a diagnosis of inflammatory bowel disease (OR, 1.41).
Other significant risk factors were being treated in a rural hospital (OR 1.29), being treated in a teaching hospital (OR, 1.08), and having a general surgeon (OR, 1.16). Having a colorectal surgeon decreased the risk of a poor outcome by 14%.
In the multivariate analysis, patients of general surgeons were 13% less likely to have any complication and 20% less likely to have ileus. But these patients were also significantly more likely to have severe complications, including pulmonary problems (13%) and surgical site infections (11%).
The length of stay was about 1 day longer, and hospitals costs were $1,122 more expensive. Patients of general surgeons were 42% more likely to die.
Dr. Casasanta said he had no relevant financial disclosures. However, Dr. Eric Haas, the primary investigator, said he has been a consultant for Applied Medical; and has received consulting fees, research funding, and honoraria from Intuitive Surgical.
WASHINGTON – Although general surgeons perform the majority of rectal resections in the United States, their patients have more severe postoperative complications, longer lengths of stay, higher hospital costs, and higher mortality rates than do those of colorectal surgeons.
A review of more than 60,000 cases over a 6-year period found that patients who had been operated on by a general surgeon had a 42% increase in the risk of mortality. They stayed in the hospital an average 1 day longer than the patients of colorectal surgeons, and had bills more than $1,000 higher. Although their overall complication rate was lower, general surgeons’ patients tended to have more severe complications, including pulmonary complications and surgical site infections, Dr. Marc Casasanta said at the annual clinical congress of the American College of Surgeons.
Dr. Casasanta of the University of Texas, Houston, extracted his data from the Premier Perspective database, the largest inpatient database in the United States. His analysis included 60,412 nonemergent rectal resections performed from 2005 to 2011.
Most of these (74%) were performed by general surgeons, with colorectal surgeons performing the remainder. Adverse outcomes examined included length and cost of hospital admission, overall complications and severe complications, and mortality.
There were several significant differences in the patient populations. General surgeons had older patients (63 vs. 61 years), and their patients tended to be sicker, with less minor disease severity (31% vs. 35%) and more major (29% vs. 17%) and extreme disease severity (8% vs. 6%).
Colorectal surgeons treated significantly more patients with inflammatory bowel disease (10% vs. 4%). They worked exclusively in urban hospitals, compared with 87% of general surgeons. Half of colorectal surgeons worked in teaching hospitals versus 37% of general surgeons.
The mean overall length of stay was a little more than 8 days. The mean hospital cost was $19,093.The overall complication rate was 29%, and mortality was 1.5%.
Complications varied significantly by surgeon specialty. General surgeons had significantly higher rates of digestive complications (including anastomotic problems), as well as pulmonary complications, lower-extremity thromboembolism, shock, and surgical site infections, including intra-abdominal infections.
The univariate analysis identified several patient characteristics significantly associated with poor outcomes, including age older than 62 years (odds ratio, 1.43), female gender (OR, 1.23), major disease severity (OR, 4.7), extreme disease severity (OR, 23), and a diagnosis of inflammatory bowel disease (OR, 1.41).
Other significant risk factors were being treated in a rural hospital (OR 1.29), being treated in a teaching hospital (OR, 1.08), and having a general surgeon (OR, 1.16). Having a colorectal surgeon decreased the risk of a poor outcome by 14%.
In the multivariate analysis, patients of general surgeons were 13% less likely to have any complication and 20% less likely to have ileus. But these patients were also significantly more likely to have severe complications, including pulmonary problems (13%) and surgical site infections (11%).
The length of stay was about 1 day longer, and hospitals costs were $1,122 more expensive. Patients of general surgeons were 42% more likely to die.
Dr. Casasanta said he had no relevant financial disclosures. However, Dr. Eric Haas, the primary investigator, said he has been a consultant for Applied Medical; and has received consulting fees, research funding, and honoraria from Intuitive Surgical.
AT THE ACS CLINICAL CONGRESS
Major finding: Patients undergoing a rectal resection had more serious complications, and were 42% more likely to die if they had a general surgeon than if they had a colorectal surgeon.
Data source: The study included more than 60,000 surgeries performed over a 6-year period.
Disclosures: Dr. Marc Casasanta said he had no relevant financial disclosures. However, Dr. Eric Haas, the primary investigator, said he has been a consultant for Applied Medical, and has received consulting fees, research funding, and honoraria from Intuitive Surgical.
Sigmoidoscopy may not be enough for older patients
WASHINGTON – Colon cancer screening with sigmoidoscopy alone could miss up to 50% of colon polyps in older patients.
As people age, polyps seem to develop more and more proximally, Dr. Victor Tsirline said at the annual clinical congress of the American College of Surgeons. His review of more than 120,000 colonoscopies found that a flexible sigmoidoscopy alone could miss 44% of polyps in patients aged 60-69 years and 50% in those aged 70-79 years.
"We found that proximal colon polyps are more frequent with advanced age than previously considered," said Dr. Tsirline of Carolinas Medical Center, Charlotte, N.C. "So if this is true, what happens if we use sigmoidoscopy instead of colonoscopy? If we had, we would have missed 22,800 polyps, and 16,800 of those would have been adenomatous. In [patients 59 and younger] 32%-36% would be missed and in the older patients, 45%-50%."
Dr. Tsirline obtained his data from the Provation MD endoscopy transcription system. He obtained information on 120,365 colonoscopies that were performed from 2003 to 2011.
He cross-referenced this with CoPathPlus, a pathology reporting system. This allowed him to cross-reference polyp pathology (adenoma vs. hyperplasia) by computer algorithm. There was complete information available on 43,833 polyps.
Because of the large sample size, he set his level of statistical significance at P = less than 0.01.
The patients in the study were aged 20-90 years. Of the entire group of procedures, 53,492 colonoscopies (44%) identified polyps. Most studies (64%) found a single polyp; 25% found two, and 11% found three or more. A subset of the colonoscopies was only for average risk screening (44,806). Of these, 46% identified polyps.
Overall, 48% of polyps were adenomatous; 37% were hyperplastic. Pathology was not available for the remainder.
The polyps were fairly evenly distributed throughout the colon: rectum, 18%; sigmoid, 26%; descending, 14%; transverse, 16%; ascending, 15%; cecum, 11%.
However, when broken down by patient age, the distribution changed significantly. With every advancing decade of life, patients were:
• 22% less likely to have polyps in the rectum or sigmoid.
• 7% more likely to have polyps in the descending colon.
• 19% more likely to have polyps in the transverse colon.
• 30% more likely to have polyps in the ascending colon.
• 22% more likely to have polyps in the cecum.
All of these risks were statistically significant, and they held for both adenomatous and hyperplastic polyps.
The findings led Dr. Tsirline to conclude that flexible sigmoidoscopy should not be relied upon as an effective colon cancer screening method in patients older than 60 years. The U.S. Preventive Services Task Force states that sigmoidoscopy every 5 years combined with high-sensitivity fecal occult blood testing every 3 years is an adequate screening alternative.
"From this study, it’s pretty apparent that sigmoidoscopy should not be used for colon cancer screening in older patients," he said.
During a discussion, Dr. Tsirline fielded a question about screening the very elderly – patients in their 80s and 90s. The study group did include a small number of these patients, he said.
"I think the argument for not screening older individuals is based on the question of whether finding a colon cancer would change anything. Most people think the risks of screening and treatment would outweigh the benefits. Yes, you may find anything, but what are you going to do about it?"
Dr. Tsirline had no financial disclosures.
WASHINGTON – Colon cancer screening with sigmoidoscopy alone could miss up to 50% of colon polyps in older patients.
As people age, polyps seem to develop more and more proximally, Dr. Victor Tsirline said at the annual clinical congress of the American College of Surgeons. His review of more than 120,000 colonoscopies found that a flexible sigmoidoscopy alone could miss 44% of polyps in patients aged 60-69 years and 50% in those aged 70-79 years.
"We found that proximal colon polyps are more frequent with advanced age than previously considered," said Dr. Tsirline of Carolinas Medical Center, Charlotte, N.C. "So if this is true, what happens if we use sigmoidoscopy instead of colonoscopy? If we had, we would have missed 22,800 polyps, and 16,800 of those would have been adenomatous. In [patients 59 and younger] 32%-36% would be missed and in the older patients, 45%-50%."
Dr. Tsirline obtained his data from the Provation MD endoscopy transcription system. He obtained information on 120,365 colonoscopies that were performed from 2003 to 2011.
He cross-referenced this with CoPathPlus, a pathology reporting system. This allowed him to cross-reference polyp pathology (adenoma vs. hyperplasia) by computer algorithm. There was complete information available on 43,833 polyps.
Because of the large sample size, he set his level of statistical significance at P = less than 0.01.
The patients in the study were aged 20-90 years. Of the entire group of procedures, 53,492 colonoscopies (44%) identified polyps. Most studies (64%) found a single polyp; 25% found two, and 11% found three or more. A subset of the colonoscopies was only for average risk screening (44,806). Of these, 46% identified polyps.
Overall, 48% of polyps were adenomatous; 37% were hyperplastic. Pathology was not available for the remainder.
The polyps were fairly evenly distributed throughout the colon: rectum, 18%; sigmoid, 26%; descending, 14%; transverse, 16%; ascending, 15%; cecum, 11%.
However, when broken down by patient age, the distribution changed significantly. With every advancing decade of life, patients were:
• 22% less likely to have polyps in the rectum or sigmoid.
• 7% more likely to have polyps in the descending colon.
• 19% more likely to have polyps in the transverse colon.
• 30% more likely to have polyps in the ascending colon.
• 22% more likely to have polyps in the cecum.
All of these risks were statistically significant, and they held for both adenomatous and hyperplastic polyps.
The findings led Dr. Tsirline to conclude that flexible sigmoidoscopy should not be relied upon as an effective colon cancer screening method in patients older than 60 years. The U.S. Preventive Services Task Force states that sigmoidoscopy every 5 years combined with high-sensitivity fecal occult blood testing every 3 years is an adequate screening alternative.
"From this study, it’s pretty apparent that sigmoidoscopy should not be used for colon cancer screening in older patients," he said.
During a discussion, Dr. Tsirline fielded a question about screening the very elderly – patients in their 80s and 90s. The study group did include a small number of these patients, he said.
"I think the argument for not screening older individuals is based on the question of whether finding a colon cancer would change anything. Most people think the risks of screening and treatment would outweigh the benefits. Yes, you may find anything, but what are you going to do about it?"
Dr. Tsirline had no financial disclosures.
WASHINGTON – Colon cancer screening with sigmoidoscopy alone could miss up to 50% of colon polyps in older patients.
As people age, polyps seem to develop more and more proximally, Dr. Victor Tsirline said at the annual clinical congress of the American College of Surgeons. His review of more than 120,000 colonoscopies found that a flexible sigmoidoscopy alone could miss 44% of polyps in patients aged 60-69 years and 50% in those aged 70-79 years.
"We found that proximal colon polyps are more frequent with advanced age than previously considered," said Dr. Tsirline of Carolinas Medical Center, Charlotte, N.C. "So if this is true, what happens if we use sigmoidoscopy instead of colonoscopy? If we had, we would have missed 22,800 polyps, and 16,800 of those would have been adenomatous. In [patients 59 and younger] 32%-36% would be missed and in the older patients, 45%-50%."
Dr. Tsirline obtained his data from the Provation MD endoscopy transcription system. He obtained information on 120,365 colonoscopies that were performed from 2003 to 2011.
He cross-referenced this with CoPathPlus, a pathology reporting system. This allowed him to cross-reference polyp pathology (adenoma vs. hyperplasia) by computer algorithm. There was complete information available on 43,833 polyps.
Because of the large sample size, he set his level of statistical significance at P = less than 0.01.
The patients in the study were aged 20-90 years. Of the entire group of procedures, 53,492 colonoscopies (44%) identified polyps. Most studies (64%) found a single polyp; 25% found two, and 11% found three or more. A subset of the colonoscopies was only for average risk screening (44,806). Of these, 46% identified polyps.
Overall, 48% of polyps were adenomatous; 37% were hyperplastic. Pathology was not available for the remainder.
The polyps were fairly evenly distributed throughout the colon: rectum, 18%; sigmoid, 26%; descending, 14%; transverse, 16%; ascending, 15%; cecum, 11%.
However, when broken down by patient age, the distribution changed significantly. With every advancing decade of life, patients were:
• 22% less likely to have polyps in the rectum or sigmoid.
• 7% more likely to have polyps in the descending colon.
• 19% more likely to have polyps in the transverse colon.
• 30% more likely to have polyps in the ascending colon.
• 22% more likely to have polyps in the cecum.
All of these risks were statistically significant, and they held for both adenomatous and hyperplastic polyps.
The findings led Dr. Tsirline to conclude that flexible sigmoidoscopy should not be relied upon as an effective colon cancer screening method in patients older than 60 years. The U.S. Preventive Services Task Force states that sigmoidoscopy every 5 years combined with high-sensitivity fecal occult blood testing every 3 years is an adequate screening alternative.
"From this study, it’s pretty apparent that sigmoidoscopy should not be used for colon cancer screening in older patients," he said.
During a discussion, Dr. Tsirline fielded a question about screening the very elderly – patients in their 80s and 90s. The study group did include a small number of these patients, he said.
"I think the argument for not screening older individuals is based on the question of whether finding a colon cancer would change anything. Most people think the risks of screening and treatment would outweigh the benefits. Yes, you may find anything, but what are you going to do about it?"
Dr. Tsirline had no financial disclosures.
AT THE ACS CLINICAL CONGRESS
Major finding: Because people are 30% more likely to have polyps in the ascending colon and 22% more likely to have polyps in the cecum as they age, sigmoidoscopy may not be an adequate screening method.
Data source: Study of 120,000 colonoscopies.
Disclosures: Dr. Victor Tsirline had no financial disclosures.
In the hormone therapy fight, bioidenticals come out swinging
With seemingly ageless celebrities touting the benefits of bioidentical, compounded hormones, and the cloud of the Women’s Health Initiative still obscuring the view, both physicians and patients want to know the truth about natural hormone replacement.
But as in so many areas of life, the truth can be many things to many people, Dr. Jan L. Shifren shared at the annual meeting of the North American Menopause Society (NAMS).
"For a woman with bothersome menopausal symptoms and concerns about potential hormone therapy [HT] risks, ‘bioHT’ may mean a formulation of hormones with all of the benefits and no risks, as it’s ‘natural’ and contains no package insert with a black box warning," said Dr. Shifren, director of the menopause program at Massachusetts General Hospital, Boston. "For a bioHT practitioner, it may mean a way of helping menopausal women disenfranchised with the medical establishment, while providing a steady source of income. For a clinician or pharmacist practicing within the current standard of care, an FDA inspector, or a lawyer for a women with endometrial cancer associated with its use, bioHT may mean something quite different!"
Bioidentical hormones are typically plant-based compounds with a molecular structure identical to that of the corresponding human hormone. Dosing is individual and based on a woman’s salivary hormone levels. Prescriptions are pharmacist compounded, typically in a topical preparation, but sometimes in an oral tablet or injected pellet.
All of this may sound reasonable at first glance, Dr. Shifren said. But science and money just keep getting in the way of the celebrity endorsements.
"Hormone therapy should be guided by symptoms. There are no data to support the use of blood or saliva measurements to improve treatment efficacy," Dr. Shifren said. Serum hormone levels constantly fluctuate – not only within the menstrual cycle, but also within a single day, "so it’s impossible to ‘match’ any individual woman’s ‘ideal’ hormone levels."
Additionally, Dr. Shifren said, salivary levels don’t even correspond to serum levels.
Safety is one of the biggest problems with such compounds. Because their manufacture is in the hands of a single individual, there’s no quality oversight. Women can’t count on getting the correct prescribed dose of hormone – and sometimes not even close to it.
A recent study commissioned by MORE magazine examined the exact hormone content in 12 bioHT prescriptions, which were filled by 12 pharmacies. Two pharmacies were retail stores and 10 were online companies.
Flora Research Labs analyzed the medications with mass spectrometry. The values varied widely: 96%-260% of the prescribed estrogen dose and 60%-80% of the prescribed progesterone dose.
"None of these would have met FDA standards," Dr. Shifren said. "The purity, bioavailability, and dose-to-dose consistency of any given prescription is an unknown."
Concern about the safety of conventional HT frequently drives women to seek out what they consider a more natural alternative. Although much more data have emerged, the original furor over the Women’s Health Initiative still casts HT in a negative light for many, Dr. Shifren said.
For women in good health, who start combination estrogen/progesterone HT early in menopause, at the lowest effective dose, the increased risks of cardiovascular disease and cancer are negligible. A 2010 British study of 15,710 cases and almost 60,000 controls found that low-dose transdermal HT had no effect on stroke risk. High-dose patch estrogen increased the risk by 89%, and both low- and high-dose oral estrogen increased the risk by 69%; however, each of those increases translated to an absolute increase of only about 1 stroke per year (BMJ 2010;340:c2519 [doi: 10.1136/bmj.c2519]).
A 2008 French study of almost 81,000 women found no significant associations between breast cancer and route of HT administration. However, the study did conclude that the combination of estrogen and progesterone was probably the safest, with a risk ratio of 1.0. Estrogen plus dydrogesterone resulted in a nonsignificant risk increase (relative risk, 1.16). Estrogen in combination with other progestogens carried a significant 69% risk increase (Breast Cancer Res. Treat. 2008;107:103-11).
Finally, Dr. Shifren said, it’s just about impossible to take profit out of the picture. Compounding HT sales are reaching more than $2 billion per year now.
"Many practitioners who prescribe compounded hormones also sell them, or benefit financially from relationships with compounders. This poses a potential conflict of interest and violates standards of professional ethical conduct," she said.
Dr. Shifren provides research consulting for New England Research Institutes.
With seemingly ageless celebrities touting the benefits of bioidentical, compounded hormones, and the cloud of the Women’s Health Initiative still obscuring the view, both physicians and patients want to know the truth about natural hormone replacement.
But as in so many areas of life, the truth can be many things to many people, Dr. Jan L. Shifren shared at the annual meeting of the North American Menopause Society (NAMS).
"For a woman with bothersome menopausal symptoms and concerns about potential hormone therapy [HT] risks, ‘bioHT’ may mean a formulation of hormones with all of the benefits and no risks, as it’s ‘natural’ and contains no package insert with a black box warning," said Dr. Shifren, director of the menopause program at Massachusetts General Hospital, Boston. "For a bioHT practitioner, it may mean a way of helping menopausal women disenfranchised with the medical establishment, while providing a steady source of income. For a clinician or pharmacist practicing within the current standard of care, an FDA inspector, or a lawyer for a women with endometrial cancer associated with its use, bioHT may mean something quite different!"
Bioidentical hormones are typically plant-based compounds with a molecular structure identical to that of the corresponding human hormone. Dosing is individual and based on a woman’s salivary hormone levels. Prescriptions are pharmacist compounded, typically in a topical preparation, but sometimes in an oral tablet or injected pellet.
All of this may sound reasonable at first glance, Dr. Shifren said. But science and money just keep getting in the way of the celebrity endorsements.
"Hormone therapy should be guided by symptoms. There are no data to support the use of blood or saliva measurements to improve treatment efficacy," Dr. Shifren said. Serum hormone levels constantly fluctuate – not only within the menstrual cycle, but also within a single day, "so it’s impossible to ‘match’ any individual woman’s ‘ideal’ hormone levels."
Additionally, Dr. Shifren said, salivary levels don’t even correspond to serum levels.
Safety is one of the biggest problems with such compounds. Because their manufacture is in the hands of a single individual, there’s no quality oversight. Women can’t count on getting the correct prescribed dose of hormone – and sometimes not even close to it.
A recent study commissioned by MORE magazine examined the exact hormone content in 12 bioHT prescriptions, which were filled by 12 pharmacies. Two pharmacies were retail stores and 10 were online companies.
Flora Research Labs analyzed the medications with mass spectrometry. The values varied widely: 96%-260% of the prescribed estrogen dose and 60%-80% of the prescribed progesterone dose.
"None of these would have met FDA standards," Dr. Shifren said. "The purity, bioavailability, and dose-to-dose consistency of any given prescription is an unknown."
Concern about the safety of conventional HT frequently drives women to seek out what they consider a more natural alternative. Although much more data have emerged, the original furor over the Women’s Health Initiative still casts HT in a negative light for many, Dr. Shifren said.
For women in good health, who start combination estrogen/progesterone HT early in menopause, at the lowest effective dose, the increased risks of cardiovascular disease and cancer are negligible. A 2010 British study of 15,710 cases and almost 60,000 controls found that low-dose transdermal HT had no effect on stroke risk. High-dose patch estrogen increased the risk by 89%, and both low- and high-dose oral estrogen increased the risk by 69%; however, each of those increases translated to an absolute increase of only about 1 stroke per year (BMJ 2010;340:c2519 [doi: 10.1136/bmj.c2519]).
A 2008 French study of almost 81,000 women found no significant associations between breast cancer and route of HT administration. However, the study did conclude that the combination of estrogen and progesterone was probably the safest, with a risk ratio of 1.0. Estrogen plus dydrogesterone resulted in a nonsignificant risk increase (relative risk, 1.16). Estrogen in combination with other progestogens carried a significant 69% risk increase (Breast Cancer Res. Treat. 2008;107:103-11).
Finally, Dr. Shifren said, it’s just about impossible to take profit out of the picture. Compounding HT sales are reaching more than $2 billion per year now.
"Many practitioners who prescribe compounded hormones also sell them, or benefit financially from relationships with compounders. This poses a potential conflict of interest and violates standards of professional ethical conduct," she said.
Dr. Shifren provides research consulting for New England Research Institutes.
With seemingly ageless celebrities touting the benefits of bioidentical, compounded hormones, and the cloud of the Women’s Health Initiative still obscuring the view, both physicians and patients want to know the truth about natural hormone replacement.
But as in so many areas of life, the truth can be many things to many people, Dr. Jan L. Shifren shared at the annual meeting of the North American Menopause Society (NAMS).
"For a woman with bothersome menopausal symptoms and concerns about potential hormone therapy [HT] risks, ‘bioHT’ may mean a formulation of hormones with all of the benefits and no risks, as it’s ‘natural’ and contains no package insert with a black box warning," said Dr. Shifren, director of the menopause program at Massachusetts General Hospital, Boston. "For a bioHT practitioner, it may mean a way of helping menopausal women disenfranchised with the medical establishment, while providing a steady source of income. For a clinician or pharmacist practicing within the current standard of care, an FDA inspector, or a lawyer for a women with endometrial cancer associated with its use, bioHT may mean something quite different!"
Bioidentical hormones are typically plant-based compounds with a molecular structure identical to that of the corresponding human hormone. Dosing is individual and based on a woman’s salivary hormone levels. Prescriptions are pharmacist compounded, typically in a topical preparation, but sometimes in an oral tablet or injected pellet.
All of this may sound reasonable at first glance, Dr. Shifren said. But science and money just keep getting in the way of the celebrity endorsements.
"Hormone therapy should be guided by symptoms. There are no data to support the use of blood or saliva measurements to improve treatment efficacy," Dr. Shifren said. Serum hormone levels constantly fluctuate – not only within the menstrual cycle, but also within a single day, "so it’s impossible to ‘match’ any individual woman’s ‘ideal’ hormone levels."
Additionally, Dr. Shifren said, salivary levels don’t even correspond to serum levels.
Safety is one of the biggest problems with such compounds. Because their manufacture is in the hands of a single individual, there’s no quality oversight. Women can’t count on getting the correct prescribed dose of hormone – and sometimes not even close to it.
A recent study commissioned by MORE magazine examined the exact hormone content in 12 bioHT prescriptions, which were filled by 12 pharmacies. Two pharmacies were retail stores and 10 were online companies.
Flora Research Labs analyzed the medications with mass spectrometry. The values varied widely: 96%-260% of the prescribed estrogen dose and 60%-80% of the prescribed progesterone dose.
"None of these would have met FDA standards," Dr. Shifren said. "The purity, bioavailability, and dose-to-dose consistency of any given prescription is an unknown."
Concern about the safety of conventional HT frequently drives women to seek out what they consider a more natural alternative. Although much more data have emerged, the original furor over the Women’s Health Initiative still casts HT in a negative light for many, Dr. Shifren said.
For women in good health, who start combination estrogen/progesterone HT early in menopause, at the lowest effective dose, the increased risks of cardiovascular disease and cancer are negligible. A 2010 British study of 15,710 cases and almost 60,000 controls found that low-dose transdermal HT had no effect on stroke risk. High-dose patch estrogen increased the risk by 89%, and both low- and high-dose oral estrogen increased the risk by 69%; however, each of those increases translated to an absolute increase of only about 1 stroke per year (BMJ 2010;340:c2519 [doi: 10.1136/bmj.c2519]).
A 2008 French study of almost 81,000 women found no significant associations between breast cancer and route of HT administration. However, the study did conclude that the combination of estrogen and progesterone was probably the safest, with a risk ratio of 1.0. Estrogen plus dydrogesterone resulted in a nonsignificant risk increase (relative risk, 1.16). Estrogen in combination with other progestogens carried a significant 69% risk increase (Breast Cancer Res. Treat. 2008;107:103-11).
Finally, Dr. Shifren said, it’s just about impossible to take profit out of the picture. Compounding HT sales are reaching more than $2 billion per year now.
"Many practitioners who prescribe compounded hormones also sell them, or benefit financially from relationships with compounders. This poses a potential conflict of interest and violates standards of professional ethical conduct," she said.
Dr. Shifren provides research consulting for New England Research Institutes.
EXPERT ANALYSIS FROM THE NAMS 2013 ANNUAL MEETING
Early walking and eating improve outcomes for bowel surgery patients
WASHINGTON – A program to enhance recovery from bowel surgery significantly decreased the incidence rate of both postoperative ileus and 30-day readmission.
Early walking and postoperative alimentation, beginning with three times on postop day 1, drove the improved outcomes, Dr. Terrence Loftus reported at the annual clinical congress of the American College of Surgeons.
His 3-year retrospective study compared outcomes before and after the program was implemented. In addition to confirming the benefits, the study says something about how change becomes part of a systemwide culture, said Dr. Loftus, medical director of surgical services and clinical resources for Banner Health, Phoenix.
"When you see a before-and-after picture, it gives you the sense that change is like clicking on a light switch, and then everything starts happening. In reality, it’s not. It’s something that catches on over time. It’s a social process that gets passed along. It’s not Dr. Loftus coming in and showing a few slides and saying, ‘Okay, now this is what you have to do.’ It’s surgeons having conversations in the lounge and saying, ‘I tried this and it worked. You should try it too.’ "
In 2010, Banner Health System’s Surgery Clinical Consensus Group decided to tackle the system’s less-than-stellar rates of postoperative ileus and readmission among patients who had bowel surgery.
While there are a number of published enhanced-care pathways, Dr. Loftus found a wide variety of applications over Banner Health’s large medical system. "In some places, people didn’t even know about them, and in others they were using their own version," he said. "We needed a systemwide approach to recovery – something that would be useful over a large, heterogeneous health care system."
The Banner consensus group came up with the Bowel Surgery Strategic Initiative, a program implemented in 19 hospitals spread over six states, covered by 119 surgeons. The system was widely diversified, including everything from an 18-bed critical access unit to a large 600-bed level I trauma center.
"The surgeons ranged from solo private practice, to group practices, hospital employed, and locum tenens, so you can begin to understand the wide diversity of practices and environments we were trying to implement this in," Dr. Loftus said.
The first step was to develop a literature-based GI care pathway that could be widely implemented. The group’s first version had 80 steps – vastly too many.
The final program was based on just two drivers: early alimentation and early ambulation. Eating began with the earliest possible removal of the nasogastric tube (if present), 200-250 cc of oral intake on postoperative day 1, and a progressively advancing diet. Ambulation started with getting up and walking three times on postop day 1, with progressively increasing physical activity.
The team studied a 3-year period spanning 18 months on either side of the implementation date of July 2011. The analysis included 5,000 patients who had undergone elective small- or large-bowel surgery during that time period.
Dr. Loftus said there was a significant increase in both ambulation and alimentation. Before the program, the mean ambulation rate was 53%; that increased to 73% afterward. Alimentation increased from 51% to 71%. The composite of both drivers increased from 53% to 72%.
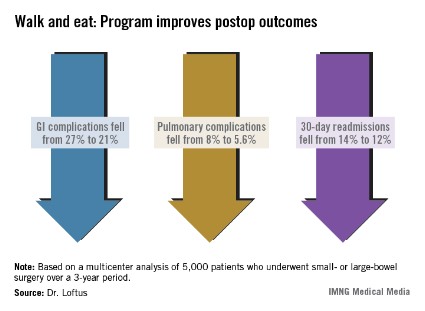
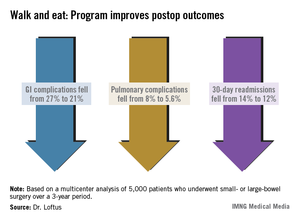
There were associated improvements in most of the clinical outcomes assessed. Overall, the complication rate decreased significantly, from 36% to 29%. Gastrointestinal complications fell from 27% to 21%, with the incidence of postoperative ileus declining from 27% to 21%. Pulmonary complications also decreased significantly, from 8% to 5.6%.
Thirty-day readmission declined from 14% to 12% – also a significant decrease. This was expected, Dr. Loftus said, because most of the readmissions were due to paralytic ileus.
Length of stay, however, was not significantly affected. There was no change in mortality.
The project also illuminated the importance of a facility’s own professional culture. Chart audits found that some facilities were only ambulating patients about a third of the time that it was ordered – even though nursing estimated the rate at 80% and surgeons at 70%.
Facilities that had particularly good postintervention outcomes were already ahead of the game before the project. "These already had a culture of activity. Nursing was getting patients up on day 1, and they set up their schedules in a way that says, ‘These patients will get up. We are going to do this no matter what.’ "
Dr. Loftus had no financial disclosures.
WASHINGTON – A program to enhance recovery from bowel surgery significantly decreased the incidence rate of both postoperative ileus and 30-day readmission.
Early walking and postoperative alimentation, beginning with three times on postop day 1, drove the improved outcomes, Dr. Terrence Loftus reported at the annual clinical congress of the American College of Surgeons.
His 3-year retrospective study compared outcomes before and after the program was implemented. In addition to confirming the benefits, the study says something about how change becomes part of a systemwide culture, said Dr. Loftus, medical director of surgical services and clinical resources for Banner Health, Phoenix.

"When you see a before-and-after picture, it gives you the sense that change is like clicking on a light switch, and then everything starts happening. In reality, it’s not. It’s something that catches on over time. It’s a social process that gets passed along. It’s not Dr. Loftus coming in and showing a few slides and saying, ‘Okay, now this is what you have to do.’ It’s surgeons having conversations in the lounge and saying, ‘I tried this and it worked. You should try it too.’ "
In 2010, Banner Health System’s Surgery Clinical Consensus Group decided to tackle the system’s less-than-stellar rates of postoperative ileus and readmission among patients who had bowel surgery.
While there are a number of published enhanced-care pathways, Dr. Loftus found a wide variety of applications over Banner Health’s large medical system. "In some places, people didn’t even know about them, and in others they were using their own version," he said. "We needed a systemwide approach to recovery – something that would be useful over a large, heterogeneous health care system."
The Banner consensus group came up with the Bowel Surgery Strategic Initiative, a program implemented in 19 hospitals spread over six states, covered by 119 surgeons. The system was widely diversified, including everything from an 18-bed critical access unit to a large 600-bed level I trauma center.
"The surgeons ranged from solo private practice, to group practices, hospital employed, and locum tenens, so you can begin to understand the wide diversity of practices and environments we were trying to implement this in," Dr. Loftus said.
The first step was to develop a literature-based GI care pathway that could be widely implemented. The group’s first version had 80 steps – vastly too many.
The final program was based on just two drivers: early alimentation and early ambulation. Eating began with the earliest possible removal of the nasogastric tube (if present), 200-250 cc of oral intake on postoperative day 1, and a progressively advancing diet. Ambulation started with getting up and walking three times on postop day 1, with progressively increasing physical activity.
The team studied a 3-year period spanning 18 months on either side of the implementation date of July 2011. The analysis included 5,000 patients who had undergone elective small- or large-bowel surgery during that time period.
Dr. Loftus said there was a significant increase in both ambulation and alimentation. Before the program, the mean ambulation rate was 53%; that increased to 73% afterward. Alimentation increased from 51% to 71%. The composite of both drivers increased from 53% to 72%.
There were associated improvements in most of the clinical outcomes assessed. Overall, the complication rate decreased significantly, from 36% to 29%. Gastrointestinal complications fell from 27% to 21%, with the incidence of postoperative ileus declining from 27% to 21%. Pulmonary complications also decreased significantly, from 8% to 5.6%.
Thirty-day readmission declined from 14% to 12% – also a significant decrease. This was expected, Dr. Loftus said, because most of the readmissions were due to paralytic ileus.
Length of stay, however, was not significantly affected. There was no change in mortality.
The project also illuminated the importance of a facility’s own professional culture. Chart audits found that some facilities were only ambulating patients about a third of the time that it was ordered – even though nursing estimated the rate at 80% and surgeons at 70%.
Facilities that had particularly good postintervention outcomes were already ahead of the game before the project. "These already had a culture of activity. Nursing was getting patients up on day 1, and they set up their schedules in a way that says, ‘These patients will get up. We are going to do this no matter what.’ "
Dr. Loftus had no financial disclosures.
WASHINGTON – A program to enhance recovery from bowel surgery significantly decreased the incidence rate of both postoperative ileus and 30-day readmission.
Early walking and postoperative alimentation, beginning with three times on postop day 1, drove the improved outcomes, Dr. Terrence Loftus reported at the annual clinical congress of the American College of Surgeons.
His 3-year retrospective study compared outcomes before and after the program was implemented. In addition to confirming the benefits, the study says something about how change becomes part of a systemwide culture, said Dr. Loftus, medical director of surgical services and clinical resources for Banner Health, Phoenix.

"When you see a before-and-after picture, it gives you the sense that change is like clicking on a light switch, and then everything starts happening. In reality, it’s not. It’s something that catches on over time. It’s a social process that gets passed along. It’s not Dr. Loftus coming in and showing a few slides and saying, ‘Okay, now this is what you have to do.’ It’s surgeons having conversations in the lounge and saying, ‘I tried this and it worked. You should try it too.’ "
In 2010, Banner Health System’s Surgery Clinical Consensus Group decided to tackle the system’s less-than-stellar rates of postoperative ileus and readmission among patients who had bowel surgery.
While there are a number of published enhanced-care pathways, Dr. Loftus found a wide variety of applications over Banner Health’s large medical system. "In some places, people didn’t even know about them, and in others they were using their own version," he said. "We needed a systemwide approach to recovery – something that would be useful over a large, heterogeneous health care system."
The Banner consensus group came up with the Bowel Surgery Strategic Initiative, a program implemented in 19 hospitals spread over six states, covered by 119 surgeons. The system was widely diversified, including everything from an 18-bed critical access unit to a large 600-bed level I trauma center.
"The surgeons ranged from solo private practice, to group practices, hospital employed, and locum tenens, so you can begin to understand the wide diversity of practices and environments we were trying to implement this in," Dr. Loftus said.
The first step was to develop a literature-based GI care pathway that could be widely implemented. The group’s first version had 80 steps – vastly too many.
The final program was based on just two drivers: early alimentation and early ambulation. Eating began with the earliest possible removal of the nasogastric tube (if present), 200-250 cc of oral intake on postoperative day 1, and a progressively advancing diet. Ambulation started with getting up and walking three times on postop day 1, with progressively increasing physical activity.
The team studied a 3-year period spanning 18 months on either side of the implementation date of July 2011. The analysis included 5,000 patients who had undergone elective small- or large-bowel surgery during that time period.
Dr. Loftus said there was a significant increase in both ambulation and alimentation. Before the program, the mean ambulation rate was 53%; that increased to 73% afterward. Alimentation increased from 51% to 71%. The composite of both drivers increased from 53% to 72%.
There were associated improvements in most of the clinical outcomes assessed. Overall, the complication rate decreased significantly, from 36% to 29%. Gastrointestinal complications fell from 27% to 21%, with the incidence of postoperative ileus declining from 27% to 21%. Pulmonary complications also decreased significantly, from 8% to 5.6%.
Thirty-day readmission declined from 14% to 12% – also a significant decrease. This was expected, Dr. Loftus said, because most of the readmissions were due to paralytic ileus.
Length of stay, however, was not significantly affected. There was no change in mortality.
The project also illuminated the importance of a facility’s own professional culture. Chart audits found that some facilities were only ambulating patients about a third of the time that it was ordered – even though nursing estimated the rate at 80% and surgeons at 70%.
Facilities that had particularly good postintervention outcomes were already ahead of the game before the project. "These already had a culture of activity. Nursing was getting patients up on day 1, and they set up their schedules in a way that says, ‘These patients will get up. We are going to do this no matter what.’ "
Dr. Loftus had no financial disclosures.
AT THE ACS CLINICAL CONGRESS
Major finding: A recovery program based on early ambulation and alimentation for patients with bowel surgery decreased readmission from 14% to 12%, and postoperative complications from 36% to 29%.
Data source: The retrospective study contained 5,000 patients who had elective small- or large-bowel surgery.
Disclosures: Dr. Loftus had no financial disclosures.
Alvimopan reduces ileus, cuts hospital time in bowel surgery
WASHINGTON – The nonopioid pain reliever alvimopan significantly decreased the incidence of postoperative ileus and shortened hospital stay among bowel surgery patients who took the drug as part of an existing accelerated recovery program.
Compared with patients who took placebo, those who took alvimopan left the hospital about 1 day sooner. In addition to the decrease in ileus, patients experienced a quicker return to normal bowel function, including time to first flatus and first bowel movement, Dr. Robert Moesinger said at the annual clinical congress of the American College of Surgeons.
"Alvimopan augmented the already improved outcomes we had seen with our validated recovery pathway, in both laparoscopic- and open-surgery patients," said Dr. Moesinger of Intermountain Healthcare, Salt Lake City. "Given the very diverse nature of our health care system, with multiple types of hospitals and surgeons, we feel these data are widely applicable and we are very comfortable recommending its routine use for patients having elective bowel surgery."
Dr. Moesinger and his colleagues conducted a randomized, placebo-controlled trial of alvimopan in 248 such patients. Those taking the study drug received 12 mg before surgery; after surgery, they received 12 mg twice a day until discharge.
The primary endpoint was postoperative length of stay. Secondary endpoints included the incidence of postoperative ileus, time to first flatus and first bowel movement, tolerance of solid food, total hospital and pharmacy costs, nasogastric tube reinsertion, 30-day readmission rates, and anastomotic leak.
The patients’ mean age was 61 years. There were no significant differences in any of their baseline demographics or clinical characteristics. Because of a statistical fluke, the placebo group did contain significantly more open-surgery patients than the alvimopan group (34% vs. 20%). The rest of the patients had laparoscopic surgery. Patients had an average of nine doses of the study drug.
The mean length of stay was 4 days in the alvimopan group and 5 in the placebo group – a significant difference.
There was significantly less postoperative ileus in the alvimopan group (2% vs. 10%). Significantly fewer of those taking the study drug needed a nasogastric tube reinserted (2% vs. 9%). The rates of 30-day readmission, reoperation, and anastomotic leak were similar in both groups. The mean time to first bowel movement was about 1 day sooner in the alvimopan group.
The median hospital cost was $10,832 for the alvimopan group and $11,924 for the placebo group – a significant difference. The median total pharmacy cost was $476 vs. $501; this difference was not significant.
Because Cubist Pharmaceuticals, which makes alvimopan, provided the study drug at no cost, the overall cost analysis cannot be considered complete, Dr. Moesinger noted. However, the pharmacy cost for a similar course of the drug would be $84. Figuring that cost into the total saved for each admission ($1,686) still yielded a net financial benefit of $930 per patient, he said.
Dr. Moesinger had no financial disclosures.
Alvimopan (Entereg, Cubist Pharmaceuticals) is an orally administered, peripherally acting mu-opioid receptor antagonist that does not cross the blood-brain barrier. It is designed to reverse opioid-induced changes in the gastrointestinal tract without adversely affecting opioid-induced analgesia.

|
| Dr. Brian E. Lacy |
Alvimopan, the first FDA-approved medication for the treatment of postoperative ileus (POI), was approved in May 2008 with a Risk Evaluation and Mitigation Strategy in place, due to concerns over an increased number of myocardial infarctions in one research study.
Postoperative ileus is the impairment of gastrointestinal motility after abdominal or pelvic surgery, and is an expected complication of major abdominal surgery. It can affect all segments of the gastrointestinal tract and may lead to symptoms of nausea, vomiting, bloating, distention, constipation, and inability to evacuate flatus. Several studies have demonstrated that POI delays hospital discharges, increases the rate of hospital readmission after abdominal surgery, and increases the risk of postoperative complications. As such, POI imposes a significant economic impact on the health care system.
The current study confirms earlier studies involving over 2,000 patients that led to the approval of alvimopan. The current study was a single-center, randomized, double-blind study comparing placebo to alvimopan. Patients randomized to alvimopan (12 mg preoperatively; 12 mg twice daily until discharge) were discharged from the hospital 1 day earlier, were less likely to have a nasogastric tube reinserted, and had a bowel movement 1 day earlier, compared with those patients receiving placebo. The authors reported a significant cost savings per patient.*
These findings are important given the absence of other FDA-approved medications for the treatment of POI and the lack of other effective treatments. Alvimopan is available only to patients who are in hospitals that are registered to use this medication under the EASE (Entereg Access Support and Education) program. This recent study should prompt nonparticipating hospitals to consider routine use of alvimopan in all patients undergoing elective and emergent surgeries at risk for development of POI.
Dr. Brian E. Lacy is a professor of medicine at the Geisel School of Medicine at Dartmouth, Hanover, N.H., and chief of the section of gastroenterology and hepatology at Dartmouth-Hitchcock Medical Center. He has no relevant conflicts of interest.
*CORRECTION 11/21/13: The original version of this story misstated the cost savings per patient.
Alvimopan (Entereg, Cubist Pharmaceuticals) is an orally administered, peripherally acting mu-opioid receptor antagonist that does not cross the blood-brain barrier. It is designed to reverse opioid-induced changes in the gastrointestinal tract without adversely affecting opioid-induced analgesia.

|
| Dr. Brian E. Lacy |
Alvimopan, the first FDA-approved medication for the treatment of postoperative ileus (POI), was approved in May 2008 with a Risk Evaluation and Mitigation Strategy in place, due to concerns over an increased number of myocardial infarctions in one research study.
Postoperative ileus is the impairment of gastrointestinal motility after abdominal or pelvic surgery, and is an expected complication of major abdominal surgery. It can affect all segments of the gastrointestinal tract and may lead to symptoms of nausea, vomiting, bloating, distention, constipation, and inability to evacuate flatus. Several studies have demonstrated that POI delays hospital discharges, increases the rate of hospital readmission after abdominal surgery, and increases the risk of postoperative complications. As such, POI imposes a significant economic impact on the health care system.
The current study confirms earlier studies involving over 2,000 patients that led to the approval of alvimopan. The current study was a single-center, randomized, double-blind study comparing placebo to alvimopan. Patients randomized to alvimopan (12 mg preoperatively; 12 mg twice daily until discharge) were discharged from the hospital 1 day earlier, were less likely to have a nasogastric tube reinserted, and had a bowel movement 1 day earlier, compared with those patients receiving placebo. The authors reported a significant cost savings per patient.*
These findings are important given the absence of other FDA-approved medications for the treatment of POI and the lack of other effective treatments. Alvimopan is available only to patients who are in hospitals that are registered to use this medication under the EASE (Entereg Access Support and Education) program. This recent study should prompt nonparticipating hospitals to consider routine use of alvimopan in all patients undergoing elective and emergent surgeries at risk for development of POI.
Dr. Brian E. Lacy is a professor of medicine at the Geisel School of Medicine at Dartmouth, Hanover, N.H., and chief of the section of gastroenterology and hepatology at Dartmouth-Hitchcock Medical Center. He has no relevant conflicts of interest.
*CORRECTION 11/21/13: The original version of this story misstated the cost savings per patient.
Alvimopan (Entereg, Cubist Pharmaceuticals) is an orally administered, peripherally acting mu-opioid receptor antagonist that does not cross the blood-brain barrier. It is designed to reverse opioid-induced changes in the gastrointestinal tract without adversely affecting opioid-induced analgesia.

|
| Dr. Brian E. Lacy |
Alvimopan, the first FDA-approved medication for the treatment of postoperative ileus (POI), was approved in May 2008 with a Risk Evaluation and Mitigation Strategy in place, due to concerns over an increased number of myocardial infarctions in one research study.
Postoperative ileus is the impairment of gastrointestinal motility after abdominal or pelvic surgery, and is an expected complication of major abdominal surgery. It can affect all segments of the gastrointestinal tract and may lead to symptoms of nausea, vomiting, bloating, distention, constipation, and inability to evacuate flatus. Several studies have demonstrated that POI delays hospital discharges, increases the rate of hospital readmission after abdominal surgery, and increases the risk of postoperative complications. As such, POI imposes a significant economic impact on the health care system.
The current study confirms earlier studies involving over 2,000 patients that led to the approval of alvimopan. The current study was a single-center, randomized, double-blind study comparing placebo to alvimopan. Patients randomized to alvimopan (12 mg preoperatively; 12 mg twice daily until discharge) were discharged from the hospital 1 day earlier, were less likely to have a nasogastric tube reinserted, and had a bowel movement 1 day earlier, compared with those patients receiving placebo. The authors reported a significant cost savings per patient.*
These findings are important given the absence of other FDA-approved medications for the treatment of POI and the lack of other effective treatments. Alvimopan is available only to patients who are in hospitals that are registered to use this medication under the EASE (Entereg Access Support and Education) program. This recent study should prompt nonparticipating hospitals to consider routine use of alvimopan in all patients undergoing elective and emergent surgeries at risk for development of POI.
Dr. Brian E. Lacy is a professor of medicine at the Geisel School of Medicine at Dartmouth, Hanover, N.H., and chief of the section of gastroenterology and hepatology at Dartmouth-Hitchcock Medical Center. He has no relevant conflicts of interest.
*CORRECTION 11/21/13: The original version of this story misstated the cost savings per patient.
WASHINGTON – The nonopioid pain reliever alvimopan significantly decreased the incidence of postoperative ileus and shortened hospital stay among bowel surgery patients who took the drug as part of an existing accelerated recovery program.
Compared with patients who took placebo, those who took alvimopan left the hospital about 1 day sooner. In addition to the decrease in ileus, patients experienced a quicker return to normal bowel function, including time to first flatus and first bowel movement, Dr. Robert Moesinger said at the annual clinical congress of the American College of Surgeons.
"Alvimopan augmented the already improved outcomes we had seen with our validated recovery pathway, in both laparoscopic- and open-surgery patients," said Dr. Moesinger of Intermountain Healthcare, Salt Lake City. "Given the very diverse nature of our health care system, with multiple types of hospitals and surgeons, we feel these data are widely applicable and we are very comfortable recommending its routine use for patients having elective bowel surgery."
Dr. Moesinger and his colleagues conducted a randomized, placebo-controlled trial of alvimopan in 248 such patients. Those taking the study drug received 12 mg before surgery; after surgery, they received 12 mg twice a day until discharge.
The primary endpoint was postoperative length of stay. Secondary endpoints included the incidence of postoperative ileus, time to first flatus and first bowel movement, tolerance of solid food, total hospital and pharmacy costs, nasogastric tube reinsertion, 30-day readmission rates, and anastomotic leak.
The patients’ mean age was 61 years. There were no significant differences in any of their baseline demographics or clinical characteristics. Because of a statistical fluke, the placebo group did contain significantly more open-surgery patients than the alvimopan group (34% vs. 20%). The rest of the patients had laparoscopic surgery. Patients had an average of nine doses of the study drug.
The mean length of stay was 4 days in the alvimopan group and 5 in the placebo group – a significant difference.
There was significantly less postoperative ileus in the alvimopan group (2% vs. 10%). Significantly fewer of those taking the study drug needed a nasogastric tube reinserted (2% vs. 9%). The rates of 30-day readmission, reoperation, and anastomotic leak were similar in both groups. The mean time to first bowel movement was about 1 day sooner in the alvimopan group.
The median hospital cost was $10,832 for the alvimopan group and $11,924 for the placebo group – a significant difference. The median total pharmacy cost was $476 vs. $501; this difference was not significant.
Because Cubist Pharmaceuticals, which makes alvimopan, provided the study drug at no cost, the overall cost analysis cannot be considered complete, Dr. Moesinger noted. However, the pharmacy cost for a similar course of the drug would be $84. Figuring that cost into the total saved for each admission ($1,686) still yielded a net financial benefit of $930 per patient, he said.
Dr. Moesinger had no financial disclosures.
WASHINGTON – The nonopioid pain reliever alvimopan significantly decreased the incidence of postoperative ileus and shortened hospital stay among bowel surgery patients who took the drug as part of an existing accelerated recovery program.
Compared with patients who took placebo, those who took alvimopan left the hospital about 1 day sooner. In addition to the decrease in ileus, patients experienced a quicker return to normal bowel function, including time to first flatus and first bowel movement, Dr. Robert Moesinger said at the annual clinical congress of the American College of Surgeons.
"Alvimopan augmented the already improved outcomes we had seen with our validated recovery pathway, in both laparoscopic- and open-surgery patients," said Dr. Moesinger of Intermountain Healthcare, Salt Lake City. "Given the very diverse nature of our health care system, with multiple types of hospitals and surgeons, we feel these data are widely applicable and we are very comfortable recommending its routine use for patients having elective bowel surgery."
Dr. Moesinger and his colleagues conducted a randomized, placebo-controlled trial of alvimopan in 248 such patients. Those taking the study drug received 12 mg before surgery; after surgery, they received 12 mg twice a day until discharge.
The primary endpoint was postoperative length of stay. Secondary endpoints included the incidence of postoperative ileus, time to first flatus and first bowel movement, tolerance of solid food, total hospital and pharmacy costs, nasogastric tube reinsertion, 30-day readmission rates, and anastomotic leak.
The patients’ mean age was 61 years. There were no significant differences in any of their baseline demographics or clinical characteristics. Because of a statistical fluke, the placebo group did contain significantly more open-surgery patients than the alvimopan group (34% vs. 20%). The rest of the patients had laparoscopic surgery. Patients had an average of nine doses of the study drug.
The mean length of stay was 4 days in the alvimopan group and 5 in the placebo group – a significant difference.
There was significantly less postoperative ileus in the alvimopan group (2% vs. 10%). Significantly fewer of those taking the study drug needed a nasogastric tube reinserted (2% vs. 9%). The rates of 30-day readmission, reoperation, and anastomotic leak were similar in both groups. The mean time to first bowel movement was about 1 day sooner in the alvimopan group.
The median hospital cost was $10,832 for the alvimopan group and $11,924 for the placebo group – a significant difference. The median total pharmacy cost was $476 vs. $501; this difference was not significant.
Because Cubist Pharmaceuticals, which makes alvimopan, provided the study drug at no cost, the overall cost analysis cannot be considered complete, Dr. Moesinger noted. However, the pharmacy cost for a similar course of the drug would be $84. Figuring that cost into the total saved for each admission ($1,686) still yielded a net financial benefit of $930 per patient, he said.
Dr. Moesinger had no financial disclosures.
AT THE ACS CLINICAL CONGRESS
Major finding: Compared with placebo, alvimopan given before and after elective bowel surgery reduced the length of stay by 1 day and significantly decreased the incidence of postoperative ileus (2% vs. 10%).
Data source: The randomized placebo-controlled trial comprised 248 patients.
Disclosures: Dr. Moesinger had no financial disclosures.
Surgeons as proficient as gastroenterologists at colonoscopy
WASHINGTON (IMNG) – A British study suggests that it is experience, not specialty, that makes the endoscopist.
The review of more than 2,000 colonoscopies performed in 1 year determined that gastroenterologists and surgeons had identical colonoscopy completion and adverse-event outcomes, Dr. Camille Yvon reported at the annual congress of the American College of Surgeons.
Dr. Yvon, of the Medway Maritime NHS Hospital in Gillingham, England, reported the results of an audit conducted at the Kent facility from March 2011 to March 2012. It was based on prospectively collected data and was designed to compare clinical outcomes among a group of 10 physicians who were performing endoscopies during that time. Included were five consultant colorectal surgeons, one senior trust grade surgeon, and four consultant gastroenterologists.
All of the clinicians were certified by the U.K. Joint Advisory Group on Endoscopy (JAG). The group requires clinicians to complete a mentored training period and to meet specific safety and quality standards, including the following:
• 90% completion rate for colonoscopies (entering the cecum).
• Adenoma detection rate more than 10%.
• Polyp recovery more than 90%.
• Good-quality bowel prep more than 90%.
• Colonoscopy perforation rate less than 1/1,000.
• Postpolypectomy bleeding requiring transfusion less than 1/100.
• Postpolypectomy perforation rate less than 1/500.
• Flexible sigmoidoscopy perforation rate less than 1/5,000.
To become JAG accredited, physicians must be supervised by an accredited physician while performing at least 200 procedures, 100 of which were in the past year; 75% must have been colonoscopies. Certification assessment is done by two different people on two different occasions.
The audit comprised 2,058 colonoscopies. The measured outcomes were number of colonoscopies per clinician, cecal intubation rate of at least 90%, perforation rate of less than 1/1,000, and a less than 1/100 rate of postpolypectomy bleeding requiring a transfusion.
Of the total number of colonoscopies, 1,237 were done by the surgeons and the rest by the gastroenterologists. Clinicians performed an average of 205 procedures per year (range, 105-448 per year). Gastroenterologists performed an average of 205 per year, while surgeons performed an average of 206.
The overall cecal intubation rate was 94%. The average rate for both surgeons and gastroenterologists was also 94%.
There were no perforations during the study period. There were two instances of severe postpolypectomy bleeding (0.09%). However, Dr. Yvon reported, the number was too small for any meaningful statistical analysis.
Dr. Yvon had no financial disclosures.
WASHINGTON (IMNG) – A British study suggests that it is experience, not specialty, that makes the endoscopist.
The review of more than 2,000 colonoscopies performed in 1 year determined that gastroenterologists and surgeons had identical colonoscopy completion and adverse-event outcomes, Dr. Camille Yvon reported at the annual congress of the American College of Surgeons.
Dr. Yvon, of the Medway Maritime NHS Hospital in Gillingham, England, reported the results of an audit conducted at the Kent facility from March 2011 to March 2012. It was based on prospectively collected data and was designed to compare clinical outcomes among a group of 10 physicians who were performing endoscopies during that time. Included were five consultant colorectal surgeons, one senior trust grade surgeon, and four consultant gastroenterologists.
All of the clinicians were certified by the U.K. Joint Advisory Group on Endoscopy (JAG). The group requires clinicians to complete a mentored training period and to meet specific safety and quality standards, including the following:
• 90% completion rate for colonoscopies (entering the cecum).
• Adenoma detection rate more than 10%.
• Polyp recovery more than 90%.
• Good-quality bowel prep more than 90%.
• Colonoscopy perforation rate less than 1/1,000.
• Postpolypectomy bleeding requiring transfusion less than 1/100.
• Postpolypectomy perforation rate less than 1/500.
• Flexible sigmoidoscopy perforation rate less than 1/5,000.
To become JAG accredited, physicians must be supervised by an accredited physician while performing at least 200 procedures, 100 of which were in the past year; 75% must have been colonoscopies. Certification assessment is done by two different people on two different occasions.
The audit comprised 2,058 colonoscopies. The measured outcomes were number of colonoscopies per clinician, cecal intubation rate of at least 90%, perforation rate of less than 1/1,000, and a less than 1/100 rate of postpolypectomy bleeding requiring a transfusion.
Of the total number of colonoscopies, 1,237 were done by the surgeons and the rest by the gastroenterologists. Clinicians performed an average of 205 procedures per year (range, 105-448 per year). Gastroenterologists performed an average of 205 per year, while surgeons performed an average of 206.
The overall cecal intubation rate was 94%. The average rate for both surgeons and gastroenterologists was also 94%.
There were no perforations during the study period. There were two instances of severe postpolypectomy bleeding (0.09%). However, Dr. Yvon reported, the number was too small for any meaningful statistical analysis.
Dr. Yvon had no financial disclosures.
WASHINGTON (IMNG) – A British study suggests that it is experience, not specialty, that makes the endoscopist.
The review of more than 2,000 colonoscopies performed in 1 year determined that gastroenterologists and surgeons had identical colonoscopy completion and adverse-event outcomes, Dr. Camille Yvon reported at the annual congress of the American College of Surgeons.
Dr. Yvon, of the Medway Maritime NHS Hospital in Gillingham, England, reported the results of an audit conducted at the Kent facility from March 2011 to March 2012. It was based on prospectively collected data and was designed to compare clinical outcomes among a group of 10 physicians who were performing endoscopies during that time. Included were five consultant colorectal surgeons, one senior trust grade surgeon, and four consultant gastroenterologists.
All of the clinicians were certified by the U.K. Joint Advisory Group on Endoscopy (JAG). The group requires clinicians to complete a mentored training period and to meet specific safety and quality standards, including the following:
• 90% completion rate for colonoscopies (entering the cecum).
• Adenoma detection rate more than 10%.
• Polyp recovery more than 90%.
• Good-quality bowel prep more than 90%.
• Colonoscopy perforation rate less than 1/1,000.
• Postpolypectomy bleeding requiring transfusion less than 1/100.
• Postpolypectomy perforation rate less than 1/500.
• Flexible sigmoidoscopy perforation rate less than 1/5,000.
To become JAG accredited, physicians must be supervised by an accredited physician while performing at least 200 procedures, 100 of which were in the past year; 75% must have been colonoscopies. Certification assessment is done by two different people on two different occasions.
The audit comprised 2,058 colonoscopies. The measured outcomes were number of colonoscopies per clinician, cecal intubation rate of at least 90%, perforation rate of less than 1/1,000, and a less than 1/100 rate of postpolypectomy bleeding requiring a transfusion.
Of the total number of colonoscopies, 1,237 were done by the surgeons and the rest by the gastroenterologists. Clinicians performed an average of 205 procedures per year (range, 105-448 per year). Gastroenterologists performed an average of 205 per year, while surgeons performed an average of 206.
The overall cecal intubation rate was 94%. The average rate for both surgeons and gastroenterologists was also 94%.
There were no perforations during the study period. There were two instances of severe postpolypectomy bleeding (0.09%). However, Dr. Yvon reported, the number was too small for any meaningful statistical analysis.
Dr. Yvon had no financial disclosures.
AT THE ACS CLINICAL CONGRESS
Major finding: Gastroenterologists and surgeons both completed an average of 94% of colonoscopies they performed during a 1-year audit period.
Data source: The audit examined outcomes of more than 2,000 colonoscopies.
Disclosures: Dr. Yvon had no financial disclosures.
Long-term data show laparoscopic Roux-en-Y still safer, cheaper than open
WASHINGTON – Fifteen years after having a Roux-en-Y gastric bypass, patients who had laparoscopic surgery were more likely to still be alive, without long-term complications and with a much smaller surgery-related medical bill than those who had open surgery.
Laparoscopic surgery conferred a 44% decrease in the chance of death from any cause, and a 20% decrease in the risk of having a long-term complication related to bariatric surgery, Dr. Anna Weiss reported at the annual clinical congress of the American College of Surgeons.
Her population-based study of 135,000 patients is one of the largest and longest of its kind, allowing it to pick up long-term outcomes trends that have not been detailed before, said Dr. Weiss of the University of California, San Diego.
The study used data from the California Office of Statewide Health Planning and Development. The database assigns every patient a unique identification number and tracks every hospital admission for that patient. Dr. Weiss looked at health outcomes for patients who underwent an open or laparoscopic Roux-en-Y gastric bypass anytime from 1995 to 2009.
The study’s primary outcome was all-cause mortality. Secondary outcomes included the incidence of gallbladder disease, cholecystectomy, marginal ulcer, and reoperation. It also examined the cost of the index surgery and subsequent costs related to the surgery.
She found a significant, time-bound association with the frequency of laparoscopic bypass. In 1995, less than 5% of the procedures were performed laparoscopically. By 2000, that had risen to about 20%. Thereafter, the frequency rose sharply. By 2009, just over 90% of Roux-en-Y procedures were minimally invasive.
About 80% of the patients were women; about 72% were white. They ranged widely in age, with more than 2,000 being 80 years or older at the time of surgery. But, Dr. Weiss said, the majority, about 70%, were aged 30-60 years.
Comorbidities were common and included prior gallbladder disease (20%), hypertension (51%), hyperlipidemia (31%), and diabetes (28%).
Most patients (67%) stayed in the hospital no more than 3 days. However, significantly more patients with laparoscopic surgery had these short stays (90% vs. 44% of the open-surgery group). Significantly more patients in the open-surgery group stayed 4-6 days (33% vs. 8%) and 7 or more days (22% vs. 2%).
Most of the patients had private insurance (80%). Medicare insured 10% and Medi-Cal, 6%. The remainder were self-pay. Significantly more laparoscopic procedures were performed at Centers of Excellence (76% vs. 59% of open procedures).
Overall, the rates of marginal ulcer and reoperations were low (1% each). However, Dr. Weiss noted, other outcomes were much more common, including gallbladder complications (22%) and cholecystectomies (13%). Long-term complications were significantly more common in the open group (10% vs. less than 5%).
By the end of the follow-up period, overall all-cause mortality was 11%. It was significantly less in the laparoscopic group than in the open group (about 5% vs. 20%).
Laparoscopic surgery carried a significantly smaller price tag than open surgery. The mean cost of the index operation was $56,170 for laparoscopic surgery, compared with a mean of $87,026 for open surgery. Over the entire follow-up period, patients who had open surgery racked up about $31,000 more in costs related directly to the procedure. However, Dr. Weiss noted, the standard deviation of lifetime costs ranged from $208,153 for an open procedure vs. $84,299 for a laparoscopic procedure.
Despite these differences, publicly insured patients were much more likely to have had open surgery. Patients with Medicare were significantly more likely to have an open than a laparoscopic procedure (15% vs. 6%), as were patients with Medi-Cal (7% vs. 4%). Patients with private insurance were significantly more likely to have laparoscopic surgery (86% vs. 74%).
In a multivariate analysis, several factors were significantly associated with outcome. Female gender reduced the risk of death by more than 30%. Younger age also portended better outcomes. Compared with patients in their 20s, those in their 50s were three times as likely to have died; those in their 60s, six times as likely, and those in their 70s, almost nine times as likely. The few patients who had surgery in their 80s were 10 times as likely to die as the youngest. However, several commentators pointed out during discussion that age-related all-cause mortality is not a surprising finding in a longitudinal study.
Insurance was also significantly related to outcome. Dr. Weiss used the self-pay group as her reference group. Compared with these patients, Medicare and Medi-Cal patients were twice as likely to have died by the study’s end. "Interestingly, they were no more likely to have long-term complications. However, when they did, these were more serious complications, possibly indicating that overall those with public insurance could have been a sicker group" at baseline.
Longer length of stay was also a significant predictor of mortality, with patients staying a week or longer five times more likely to have died by the end of follow-up than the reference group of those staying 4-6 days. "Interestingly, we also found that patients who stayed 3 days or less were 73% more likely to have died." This probably represented patients who had serious perioperative complications, she suggested.
Having either surgery at a Center of Excellence reduced the risk of mortality by 25%, she added.
Dr. Weiss had no financial disclosures.
WASHINGTON – Fifteen years after having a Roux-en-Y gastric bypass, patients who had laparoscopic surgery were more likely to still be alive, without long-term complications and with a much smaller surgery-related medical bill than those who had open surgery.
Laparoscopic surgery conferred a 44% decrease in the chance of death from any cause, and a 20% decrease in the risk of having a long-term complication related to bariatric surgery, Dr. Anna Weiss reported at the annual clinical congress of the American College of Surgeons.
Her population-based study of 135,000 patients is one of the largest and longest of its kind, allowing it to pick up long-term outcomes trends that have not been detailed before, said Dr. Weiss of the University of California, San Diego.
The study used data from the California Office of Statewide Health Planning and Development. The database assigns every patient a unique identification number and tracks every hospital admission for that patient. Dr. Weiss looked at health outcomes for patients who underwent an open or laparoscopic Roux-en-Y gastric bypass anytime from 1995 to 2009.
The study’s primary outcome was all-cause mortality. Secondary outcomes included the incidence of gallbladder disease, cholecystectomy, marginal ulcer, and reoperation. It also examined the cost of the index surgery and subsequent costs related to the surgery.
She found a significant, time-bound association with the frequency of laparoscopic bypass. In 1995, less than 5% of the procedures were performed laparoscopically. By 2000, that had risen to about 20%. Thereafter, the frequency rose sharply. By 2009, just over 90% of Roux-en-Y procedures were minimally invasive.
About 80% of the patients were women; about 72% were white. They ranged widely in age, with more than 2,000 being 80 years or older at the time of surgery. But, Dr. Weiss said, the majority, about 70%, were aged 30-60 years.
Comorbidities were common and included prior gallbladder disease (20%), hypertension (51%), hyperlipidemia (31%), and diabetes (28%).
Most patients (67%) stayed in the hospital no more than 3 days. However, significantly more patients with laparoscopic surgery had these short stays (90% vs. 44% of the open-surgery group). Significantly more patients in the open-surgery group stayed 4-6 days (33% vs. 8%) and 7 or more days (22% vs. 2%).
Most of the patients had private insurance (80%). Medicare insured 10% and Medi-Cal, 6%. The remainder were self-pay. Significantly more laparoscopic procedures were performed at Centers of Excellence (76% vs. 59% of open procedures).
Overall, the rates of marginal ulcer and reoperations were low (1% each). However, Dr. Weiss noted, other outcomes were much more common, including gallbladder complications (22%) and cholecystectomies (13%). Long-term complications were significantly more common in the open group (10% vs. less than 5%).
By the end of the follow-up period, overall all-cause mortality was 11%. It was significantly less in the laparoscopic group than in the open group (about 5% vs. 20%).
Laparoscopic surgery carried a significantly smaller price tag than open surgery. The mean cost of the index operation was $56,170 for laparoscopic surgery, compared with a mean of $87,026 for open surgery. Over the entire follow-up period, patients who had open surgery racked up about $31,000 more in costs related directly to the procedure. However, Dr. Weiss noted, the standard deviation of lifetime costs ranged from $208,153 for an open procedure vs. $84,299 for a laparoscopic procedure.
Despite these differences, publicly insured patients were much more likely to have had open surgery. Patients with Medicare were significantly more likely to have an open than a laparoscopic procedure (15% vs. 6%), as were patients with Medi-Cal (7% vs. 4%). Patients with private insurance were significantly more likely to have laparoscopic surgery (86% vs. 74%).
In a multivariate analysis, several factors were significantly associated with outcome. Female gender reduced the risk of death by more than 30%. Younger age also portended better outcomes. Compared with patients in their 20s, those in their 50s were three times as likely to have died; those in their 60s, six times as likely, and those in their 70s, almost nine times as likely. The few patients who had surgery in their 80s were 10 times as likely to die as the youngest. However, several commentators pointed out during discussion that age-related all-cause mortality is not a surprising finding in a longitudinal study.
Insurance was also significantly related to outcome. Dr. Weiss used the self-pay group as her reference group. Compared with these patients, Medicare and Medi-Cal patients were twice as likely to have died by the study’s end. "Interestingly, they were no more likely to have long-term complications. However, when they did, these were more serious complications, possibly indicating that overall those with public insurance could have been a sicker group" at baseline.
Longer length of stay was also a significant predictor of mortality, with patients staying a week or longer five times more likely to have died by the end of follow-up than the reference group of those staying 4-6 days. "Interestingly, we also found that patients who stayed 3 days or less were 73% more likely to have died." This probably represented patients who had serious perioperative complications, she suggested.
Having either surgery at a Center of Excellence reduced the risk of mortality by 25%, she added.
Dr. Weiss had no financial disclosures.
WASHINGTON – Fifteen years after having a Roux-en-Y gastric bypass, patients who had laparoscopic surgery were more likely to still be alive, without long-term complications and with a much smaller surgery-related medical bill than those who had open surgery.
Laparoscopic surgery conferred a 44% decrease in the chance of death from any cause, and a 20% decrease in the risk of having a long-term complication related to bariatric surgery, Dr. Anna Weiss reported at the annual clinical congress of the American College of Surgeons.
Her population-based study of 135,000 patients is one of the largest and longest of its kind, allowing it to pick up long-term outcomes trends that have not been detailed before, said Dr. Weiss of the University of California, San Diego.
The study used data from the California Office of Statewide Health Planning and Development. The database assigns every patient a unique identification number and tracks every hospital admission for that patient. Dr. Weiss looked at health outcomes for patients who underwent an open or laparoscopic Roux-en-Y gastric bypass anytime from 1995 to 2009.
The study’s primary outcome was all-cause mortality. Secondary outcomes included the incidence of gallbladder disease, cholecystectomy, marginal ulcer, and reoperation. It also examined the cost of the index surgery and subsequent costs related to the surgery.
She found a significant, time-bound association with the frequency of laparoscopic bypass. In 1995, less than 5% of the procedures were performed laparoscopically. By 2000, that had risen to about 20%. Thereafter, the frequency rose sharply. By 2009, just over 90% of Roux-en-Y procedures were minimally invasive.
About 80% of the patients were women; about 72% were white. They ranged widely in age, with more than 2,000 being 80 years or older at the time of surgery. But, Dr. Weiss said, the majority, about 70%, were aged 30-60 years.
Comorbidities were common and included prior gallbladder disease (20%), hypertension (51%), hyperlipidemia (31%), and diabetes (28%).
Most patients (67%) stayed in the hospital no more than 3 days. However, significantly more patients with laparoscopic surgery had these short stays (90% vs. 44% of the open-surgery group). Significantly more patients in the open-surgery group stayed 4-6 days (33% vs. 8%) and 7 or more days (22% vs. 2%).
Most of the patients had private insurance (80%). Medicare insured 10% and Medi-Cal, 6%. The remainder were self-pay. Significantly more laparoscopic procedures were performed at Centers of Excellence (76% vs. 59% of open procedures).
Overall, the rates of marginal ulcer and reoperations were low (1% each). However, Dr. Weiss noted, other outcomes were much more common, including gallbladder complications (22%) and cholecystectomies (13%). Long-term complications were significantly more common in the open group (10% vs. less than 5%).
By the end of the follow-up period, overall all-cause mortality was 11%. It was significantly less in the laparoscopic group than in the open group (about 5% vs. 20%).
Laparoscopic surgery carried a significantly smaller price tag than open surgery. The mean cost of the index operation was $56,170 for laparoscopic surgery, compared with a mean of $87,026 for open surgery. Over the entire follow-up period, patients who had open surgery racked up about $31,000 more in costs related directly to the procedure. However, Dr. Weiss noted, the standard deviation of lifetime costs ranged from $208,153 for an open procedure vs. $84,299 for a laparoscopic procedure.
Despite these differences, publicly insured patients were much more likely to have had open surgery. Patients with Medicare were significantly more likely to have an open than a laparoscopic procedure (15% vs. 6%), as were patients with Medi-Cal (7% vs. 4%). Patients with private insurance were significantly more likely to have laparoscopic surgery (86% vs. 74%).
In a multivariate analysis, several factors were significantly associated with outcome. Female gender reduced the risk of death by more than 30%. Younger age also portended better outcomes. Compared with patients in their 20s, those in their 50s were three times as likely to have died; those in their 60s, six times as likely, and those in their 70s, almost nine times as likely. The few patients who had surgery in their 80s were 10 times as likely to die as the youngest. However, several commentators pointed out during discussion that age-related all-cause mortality is not a surprising finding in a longitudinal study.
Insurance was also significantly related to outcome. Dr. Weiss used the self-pay group as her reference group. Compared with these patients, Medicare and Medi-Cal patients were twice as likely to have died by the study’s end. "Interestingly, they were no more likely to have long-term complications. However, when they did, these were more serious complications, possibly indicating that overall those with public insurance could have been a sicker group" at baseline.
Longer length of stay was also a significant predictor of mortality, with patients staying a week or longer five times more likely to have died by the end of follow-up than the reference group of those staying 4-6 days. "Interestingly, we also found that patients who stayed 3 days or less were 73% more likely to have died." This probably represented patients who had serious perioperative complications, she suggested.
Having either surgery at a Center of Excellence reduced the risk of mortality by 25%, she added.
Dr. Weiss had no financial disclosures.
AT THE ACS CLINICAL CONGRESS
Major finding: After up to 15 years of follow-up, patients who had laparoscopic gastric bypass were 44% less likely to have died and 20% less likely to have a long-term surgical complication than patients who had an open procedure.
Data source: The longitudinal study comprised data on 135,000 patients treated from 1995 to 2009.
Disclosures: Dr. Weiss had no financial disclosures.
Investigational biologic lessened symptoms in some patients with lupus
An investigational biologic that targets T cells in patients with systemic lupus erythematosus significantly lessened symptoms in a subset of patients who completed a 24-week randomized trial.
In the overall analysis, the drug – rigerimod (Lupuzor) – did not perform significantly better than did placebo. But a post hoc analysis of patients with a higher clinical disease score found better results, with a response rate of 84% vs. 46% who took placebo, Dr. Robert Zimmer and his colleagues reported in the November issue of the Annals of the Rheumatic Diseases (2013 [doi:10.1136/annrheumdis-2012-202460]).
The drug was well tolerated, with 40% of the treatment group experiencing at least one side effect, compared with 49% of the placebo group, wrote Dr. Zimmer, president and chief science officer of ImmuPharma, the French company that is developing rigerimod.
Rigerimod is a made from a small nuclear ribonucleoprotein. Studies suggest that it inhibits T-cell reactivity, thereby reducing proteinuria, vasculitis, and dermatitis. It also prevented the production of antibodies to DNA from a lupus-model mouse.
The phase II study comprised an intent-to-treat group 149 patients who had baseline total scores of 6 or more on the SLEDAI-2K (Systemic Lupus Erythematosus Disease Activity Index-2K). They were randomized to three arms: subcutaneous injections of 200 mcg rigerimod every 4 weeks (group 1), every 2 weeks (group 2), or placebo injections.
All patients were treated for 12 weeks and then followed for an additional 12 weeks.
Most (96%) were women; their mean age at enrollment was about 38 years. The mean disease duration was about 8 years. The mean SLEDAI-2K total score was 11, and the mean physician’s global assessment (PGA), slightly more than 1. More than 80% were taking corticosteroids; about half were taking concomitant antimalarials and a quarter, concomitant azathioprine. Less than 5% were taking methotrexate.
Responders were considered those who had a reduction of at least 4 points in their baseline SLEDAI-2K score, with no increase in the PGA score. Treatment failures were those who had a severe disease flare, needed a steroid increase of at least 80 mg prednisone equivalent, or received biologics. Patients could continue their existing, stable medication regimens during the study.
The dropout rate was 9% (13 patients) – 8% of group 1, 2% of group 2 and 16% of the placebo group.
By the end of the 12-week treatment period, the response rate was significantly better in group 1 than in the placebo group (53% vs. 36%). Response in group 2 was not significantly different from group 1(45%).
By the end of week 24, the response rate had increased similarly in the entire study population (group 1, 59%: group 2, 59%; placebo, 53%). These rates were not significantly different from one another.
The target group analysis was conducted after the study’s end. The investigators restricted this group to patients whose SLEDAI-2K clinical –not overall – score was at least 6. "Despite the fact that this population had not been defined at the beginning of the study, the changes in the inclusion criteria led us to carefully analyze this population [clinical SLEDAI score of 6 and above] as this population will be the phase III population," they noted.
At week 12 in this population, there were significantly more responders in group 1 than in the placebo group (62% vs. 39%). The difference between group 2 and placebo was not statistically significant.
By week 24, groups 1 and 2 had similar response rates (69% and 62%), but these were not significantly higher than placebo (56%), with p values of 0.11 for group 1 vs. placebo and 0.28 for group 2 vs. placebo.
The investigators did not break down response by symptom, but did say "The apparent clinical benefit observed f or patients who received 200 mcg Lupuzor every 4 weeks, compared with those who received placebo every 2 weeks, were mainly due to an improvement in articular and cutaneous symptoms (arthritis and rash) at week 12."
The incidence of adverse events was low and similar between the groups; 40% of patients in each active group experienced at least one, compared with 49% of patients in the placebo group. The most common were injection site reactions, which occurred in 6% of group 1, 10% of group 2, and 2% of the placebo group.
One patient died during the trial. The cause of death was pneumonia, which was not deemed related to the study drug. The investigators noted that this patient had been on immunosuppressant therapy before entering the trial. Two other patients also developed pneumonia. "Thus," the investigators said, "while there did not appear to be an increased incidence of serious infections with Lupuzor in this short study, longer-term studies are needed to further characterise the overall tolerability profile of Lupuzor."
Finally, the investigators suggested that the clinical SLEDAI score, rather than the overall score, is the best way to assess this drug. "Comparing the number of responders in the target population and those of the overall population it appears that there is no change. Exactly the same number of responders was recorded in the three groups in the overall population and the target population. This indicates that the commonly used inclusion criterion [SLEDAI score of 6 and above] creates a bias in the evaluation of the study results in falsely reducing the response rate and therefore statistical analysis."
ImmuPharma is developing the drug and conducted the study.
An investigational biologic that targets T cells in patients with systemic lupus erythematosus significantly lessened symptoms in a subset of patients who completed a 24-week randomized trial.
In the overall analysis, the drug – rigerimod (Lupuzor) – did not perform significantly better than did placebo. But a post hoc analysis of patients with a higher clinical disease score found better results, with a response rate of 84% vs. 46% who took placebo, Dr. Robert Zimmer and his colleagues reported in the November issue of the Annals of the Rheumatic Diseases (2013 [doi:10.1136/annrheumdis-2012-202460]).
The drug was well tolerated, with 40% of the treatment group experiencing at least one side effect, compared with 49% of the placebo group, wrote Dr. Zimmer, president and chief science officer of ImmuPharma, the French company that is developing rigerimod.
Rigerimod is a made from a small nuclear ribonucleoprotein. Studies suggest that it inhibits T-cell reactivity, thereby reducing proteinuria, vasculitis, and dermatitis. It also prevented the production of antibodies to DNA from a lupus-model mouse.
The phase II study comprised an intent-to-treat group 149 patients who had baseline total scores of 6 or more on the SLEDAI-2K (Systemic Lupus Erythematosus Disease Activity Index-2K). They were randomized to three arms: subcutaneous injections of 200 mcg rigerimod every 4 weeks (group 1), every 2 weeks (group 2), or placebo injections.
All patients were treated for 12 weeks and then followed for an additional 12 weeks.
Most (96%) were women; their mean age at enrollment was about 38 years. The mean disease duration was about 8 years. The mean SLEDAI-2K total score was 11, and the mean physician’s global assessment (PGA), slightly more than 1. More than 80% were taking corticosteroids; about half were taking concomitant antimalarials and a quarter, concomitant azathioprine. Less than 5% were taking methotrexate.
Responders were considered those who had a reduction of at least 4 points in their baseline SLEDAI-2K score, with no increase in the PGA score. Treatment failures were those who had a severe disease flare, needed a steroid increase of at least 80 mg prednisone equivalent, or received biologics. Patients could continue their existing, stable medication regimens during the study.
The dropout rate was 9% (13 patients) – 8% of group 1, 2% of group 2 and 16% of the placebo group.
By the end of the 12-week treatment period, the response rate was significantly better in group 1 than in the placebo group (53% vs. 36%). Response in group 2 was not significantly different from group 1(45%).
By the end of week 24, the response rate had increased similarly in the entire study population (group 1, 59%: group 2, 59%; placebo, 53%). These rates were not significantly different from one another.
The target group analysis was conducted after the study’s end. The investigators restricted this group to patients whose SLEDAI-2K clinical –not overall – score was at least 6. "Despite the fact that this population had not been defined at the beginning of the study, the changes in the inclusion criteria led us to carefully analyze this population [clinical SLEDAI score of 6 and above] as this population will be the phase III population," they noted.
At week 12 in this population, there were significantly more responders in group 1 than in the placebo group (62% vs. 39%). The difference between group 2 and placebo was not statistically significant.
By week 24, groups 1 and 2 had similar response rates (69% and 62%), but these were not significantly higher than placebo (56%), with p values of 0.11 for group 1 vs. placebo and 0.28 for group 2 vs. placebo.
The investigators did not break down response by symptom, but did say "The apparent clinical benefit observed f or patients who received 200 mcg Lupuzor every 4 weeks, compared with those who received placebo every 2 weeks, were mainly due to an improvement in articular and cutaneous symptoms (arthritis and rash) at week 12."
The incidence of adverse events was low and similar between the groups; 40% of patients in each active group experienced at least one, compared with 49% of patients in the placebo group. The most common were injection site reactions, which occurred in 6% of group 1, 10% of group 2, and 2% of the placebo group.
One patient died during the trial. The cause of death was pneumonia, which was not deemed related to the study drug. The investigators noted that this patient had been on immunosuppressant therapy before entering the trial. Two other patients also developed pneumonia. "Thus," the investigators said, "while there did not appear to be an increased incidence of serious infections with Lupuzor in this short study, longer-term studies are needed to further characterise the overall tolerability profile of Lupuzor."
Finally, the investigators suggested that the clinical SLEDAI score, rather than the overall score, is the best way to assess this drug. "Comparing the number of responders in the target population and those of the overall population it appears that there is no change. Exactly the same number of responders was recorded in the three groups in the overall population and the target population. This indicates that the commonly used inclusion criterion [SLEDAI score of 6 and above] creates a bias in the evaluation of the study results in falsely reducing the response rate and therefore statistical analysis."
ImmuPharma is developing the drug and conducted the study.
An investigational biologic that targets T cells in patients with systemic lupus erythematosus significantly lessened symptoms in a subset of patients who completed a 24-week randomized trial.
In the overall analysis, the drug – rigerimod (Lupuzor) – did not perform significantly better than did placebo. But a post hoc analysis of patients with a higher clinical disease score found better results, with a response rate of 84% vs. 46% who took placebo, Dr. Robert Zimmer and his colleagues reported in the November issue of the Annals of the Rheumatic Diseases (2013 [doi:10.1136/annrheumdis-2012-202460]).
The drug was well tolerated, with 40% of the treatment group experiencing at least one side effect, compared with 49% of the placebo group, wrote Dr. Zimmer, president and chief science officer of ImmuPharma, the French company that is developing rigerimod.
Rigerimod is a made from a small nuclear ribonucleoprotein. Studies suggest that it inhibits T-cell reactivity, thereby reducing proteinuria, vasculitis, and dermatitis. It also prevented the production of antibodies to DNA from a lupus-model mouse.
The phase II study comprised an intent-to-treat group 149 patients who had baseline total scores of 6 or more on the SLEDAI-2K (Systemic Lupus Erythematosus Disease Activity Index-2K). They were randomized to three arms: subcutaneous injections of 200 mcg rigerimod every 4 weeks (group 1), every 2 weeks (group 2), or placebo injections.
All patients were treated for 12 weeks and then followed for an additional 12 weeks.
Most (96%) were women; their mean age at enrollment was about 38 years. The mean disease duration was about 8 years. The mean SLEDAI-2K total score was 11, and the mean physician’s global assessment (PGA), slightly more than 1. More than 80% were taking corticosteroids; about half were taking concomitant antimalarials and a quarter, concomitant azathioprine. Less than 5% were taking methotrexate.
Responders were considered those who had a reduction of at least 4 points in their baseline SLEDAI-2K score, with no increase in the PGA score. Treatment failures were those who had a severe disease flare, needed a steroid increase of at least 80 mg prednisone equivalent, or received biologics. Patients could continue their existing, stable medication regimens during the study.
The dropout rate was 9% (13 patients) – 8% of group 1, 2% of group 2 and 16% of the placebo group.
By the end of the 12-week treatment period, the response rate was significantly better in group 1 than in the placebo group (53% vs. 36%). Response in group 2 was not significantly different from group 1(45%).
By the end of week 24, the response rate had increased similarly in the entire study population (group 1, 59%: group 2, 59%; placebo, 53%). These rates were not significantly different from one another.
The target group analysis was conducted after the study’s end. The investigators restricted this group to patients whose SLEDAI-2K clinical –not overall – score was at least 6. "Despite the fact that this population had not been defined at the beginning of the study, the changes in the inclusion criteria led us to carefully analyze this population [clinical SLEDAI score of 6 and above] as this population will be the phase III population," they noted.
At week 12 in this population, there were significantly more responders in group 1 than in the placebo group (62% vs. 39%). The difference between group 2 and placebo was not statistically significant.
By week 24, groups 1 and 2 had similar response rates (69% and 62%), but these were not significantly higher than placebo (56%), with p values of 0.11 for group 1 vs. placebo and 0.28 for group 2 vs. placebo.
The investigators did not break down response by symptom, but did say "The apparent clinical benefit observed f or patients who received 200 mcg Lupuzor every 4 weeks, compared with those who received placebo every 2 weeks, were mainly due to an improvement in articular and cutaneous symptoms (arthritis and rash) at week 12."
The incidence of adverse events was low and similar between the groups; 40% of patients in each active group experienced at least one, compared with 49% of patients in the placebo group. The most common were injection site reactions, which occurred in 6% of group 1, 10% of group 2, and 2% of the placebo group.
One patient died during the trial. The cause of death was pneumonia, which was not deemed related to the study drug. The investigators noted that this patient had been on immunosuppressant therapy before entering the trial. Two other patients also developed pneumonia. "Thus," the investigators said, "while there did not appear to be an increased incidence of serious infections with Lupuzor in this short study, longer-term studies are needed to further characterise the overall tolerability profile of Lupuzor."
Finally, the investigators suggested that the clinical SLEDAI score, rather than the overall score, is the best way to assess this drug. "Comparing the number of responders in the target population and those of the overall population it appears that there is no change. Exactly the same number of responders was recorded in the three groups in the overall population and the target population. This indicates that the commonly used inclusion criterion [SLEDAI score of 6 and above] creates a bias in the evaluation of the study results in falsely reducing the response rate and therefore statistical analysis."
ImmuPharma is developing the drug and conducted the study.
Major finding: The investigational biologic rigerimod was superior to placebo in patients with a higher lupus clinical disease activity score, with a response rate of 84% vs. 46%.
Data source: The 24-week randomized, phase IIb trial involved 149 patients.
Disclosures: ImmuPharma is developing rigerimod and conducted the study. Dr. Zimmer is the company’s president and chief scientific officer.
Gastric Bypass Induces Diabetes Remission in Obese Patients
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
WASHINGTON – Gastric bypass surgery resulted in significantly more weight loss and also improved measures of glycemic control significantly more than did other forms of bariatric surgery, Dr. John Morton reported at the annual clinical congress of the American College of Surgeons.
However, he noted, while remission did correlate with weight loss in patients who had gastric banding or sleeving, it appeared to be independent of weight loss in those who had the bypass surgery. While he didn’t speculate on the reasons for this finding, he did affirm his belief that gastric bypass is the best option for most obese patients with comorbid diabetes.
"I feel very comfortable recommending it" for these patients, said Dr. Morton of the Stanford (Calif.) University. "There are, of course, other clinical conditions to consider when deciding [among] bypass, banding, and sleeve, but if the only consideration is around diabetes, I’m 100% comfortable in recommending it for obese diabetics."
He presented the 1-year follow-up data on 1,792 obese patients who underwent bariatric surgery. Of these, 1,364 had a Roux-en-Y bypass; 264 had a sleeve gastrectomy; and 164 had adjustable gastric banding.
The patients were a mean of about 46 years old. Body mass index was statistically, but not clinically, different between the groups (bypass 47 kg/m2; band 44 kg/m2; sleeve 44 kg/m2). Waist circumference ranged from 51 to 53 inches. About 75% of the group was female and more than half, white.
Overall, about one-third of each group had type 2 diabetes. Most were taking only oral medications. About 5% took only insulin, and about a quarter took both oral agents and insulin. At baseline, the mean HbA1c was more than 7% in each group. The mean fasting insulin was 36 microU/mL in the bypass group, 28 microU/mL in the band group, and 32 microU/mL in the sleeve group.
By 12 months after surgery, patients with diabetes who had bypass had lost the most weight – a mean of 71% of their excess body weight, compared with 38% in the banding group and 50% in the sleeve group.
Those who had the bypass surgery also experienced the biggest change in their HbA1c – dropping almost 16% to a mean of 5.8%. Patients in the other two groups experienced a mean drop of 10%, resulting in HbA1c levels of right around 6%.
Fasting insulin levels also improved significantly more in the bypass group, falling from a baseline mean of 56 microU/mL to 7.8 microU/mL – a decrease of 68%. In the band group, the level fell from 28 microU/mL to 12 microU/mL – a 52% decrease. In the sleeve group, levels fell from 32 microU/mL to 10 microU/mL – a 61% decrease.
"Fasting insulin is also considered an independent marker of cardiac risk," Dr. Morton added, indicating that the risk of cardiovascular problems would fall along with insulin levels.
Blood glucose improved significantly more in the bypass group, falling from a baseline mean of 149 mg/dL to 101 mg/dL – a 22% change. In the band group, the level fell from 140 mg/dL to 125 mg/dL (8%) and in the sleeve group, from 130 mg/dL to 118 mg/dL (5.6%).
A multivariate analysis controlled for surgery type, sex, body mass index, race, age, and insurance status. Of these factors, surgery type was the strongest predictor, with bypass patients three times more likely to achieve that goal than those undergoing banding or sleeve placement.
Dr. Morton did not present his complication data. However, during the discussion period, he said the three procedures were similarly safe. The gastric sleeve group had a higher leak rate than did the other groups, but that remained less than 1%. Readmission rates were comparable, he said, but he did not provide that number.
"Any time you consider this, it has to be a risk/benefit analysis," he said. "It’s our philosophy that for obese patients with severe diabetes, we approach them first with the bypass because it has the most proven track record over time."
Dr. Morton has up to 7 years of data on some of the patients, and said he is now analyzing that. But when questioned about durability, he agreed that diabetes can recur in the rather common scenario of a patient regaining weight. "At the end of the day, though, what’s important is that the obese patient with diabetes gets treatment. All three surgeries demonstrated some improvement, and I believe that any surgery is better than no surgery at all."
Dr. Morton has received research support from Covidien.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
WASHINGTON – Gastric bypass surgery resulted in significantly more weight loss and also improved measures of glycemic control significantly more than did other forms of bariatric surgery, Dr. John Morton reported at the annual clinical congress of the American College of Surgeons.
However, he noted, while remission did correlate with weight loss in patients who had gastric banding or sleeving, it appeared to be independent of weight loss in those who had the bypass surgery. While he didn’t speculate on the reasons for this finding, he did affirm his belief that gastric bypass is the best option for most obese patients with comorbid diabetes.
"I feel very comfortable recommending it" for these patients, said Dr. Morton of the Stanford (Calif.) University. "There are, of course, other clinical conditions to consider when deciding [among] bypass, banding, and sleeve, but if the only consideration is around diabetes, I’m 100% comfortable in recommending it for obese diabetics."
He presented the 1-year follow-up data on 1,792 obese patients who underwent bariatric surgery. Of these, 1,364 had a Roux-en-Y bypass; 264 had a sleeve gastrectomy; and 164 had adjustable gastric banding.
The patients were a mean of about 46 years old. Body mass index was statistically, but not clinically, different between the groups (bypass 47 kg/m2; band 44 kg/m2; sleeve 44 kg/m2). Waist circumference ranged from 51 to 53 inches. About 75% of the group was female and more than half, white.
Overall, about one-third of each group had type 2 diabetes. Most were taking only oral medications. About 5% took only insulin, and about a quarter took both oral agents and insulin. At baseline, the mean HbA1c was more than 7% in each group. The mean fasting insulin was 36 microU/mL in the bypass group, 28 microU/mL in the band group, and 32 microU/mL in the sleeve group.
By 12 months after surgery, patients with diabetes who had bypass had lost the most weight – a mean of 71% of their excess body weight, compared with 38% in the banding group and 50% in the sleeve group.
Those who had the bypass surgery also experienced the biggest change in their HbA1c – dropping almost 16% to a mean of 5.8%. Patients in the other two groups experienced a mean drop of 10%, resulting in HbA1c levels of right around 6%.
Fasting insulin levels also improved significantly more in the bypass group, falling from a baseline mean of 56 microU/mL to 7.8 microU/mL – a decrease of 68%. In the band group, the level fell from 28 microU/mL to 12 microU/mL – a 52% decrease. In the sleeve group, levels fell from 32 microU/mL to 10 microU/mL – a 61% decrease.
"Fasting insulin is also considered an independent marker of cardiac risk," Dr. Morton added, indicating that the risk of cardiovascular problems would fall along with insulin levels.
Blood glucose improved significantly more in the bypass group, falling from a baseline mean of 149 mg/dL to 101 mg/dL – a 22% change. In the band group, the level fell from 140 mg/dL to 125 mg/dL (8%) and in the sleeve group, from 130 mg/dL to 118 mg/dL (5.6%).
A multivariate analysis controlled for surgery type, sex, body mass index, race, age, and insurance status. Of these factors, surgery type was the strongest predictor, with bypass patients three times more likely to achieve that goal than those undergoing banding or sleeve placement.
Dr. Morton did not present his complication data. However, during the discussion period, he said the three procedures were similarly safe. The gastric sleeve group had a higher leak rate than did the other groups, but that remained less than 1%. Readmission rates were comparable, he said, but he did not provide that number.
"Any time you consider this, it has to be a risk/benefit analysis," he said. "It’s our philosophy that for obese patients with severe diabetes, we approach them first with the bypass because it has the most proven track record over time."
Dr. Morton has up to 7 years of data on some of the patients, and said he is now analyzing that. But when questioned about durability, he agreed that diabetes can recur in the rather common scenario of a patient regaining weight. "At the end of the day, though, what’s important is that the obese patient with diabetes gets treatment. All three surgeries demonstrated some improvement, and I believe that any surgery is better than no surgery at all."
Dr. Morton has received research support from Covidien.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
Earn 0.25 hours AMA PRA Category 1 credit: Read this article, and click the link at the end to take the post-test.
WASHINGTON – Gastric bypass surgery resulted in significantly more weight loss and also improved measures of glycemic control significantly more than did other forms of bariatric surgery, Dr. John Morton reported at the annual clinical congress of the American College of Surgeons.
However, he noted, while remission did correlate with weight loss in patients who had gastric banding or sleeving, it appeared to be independent of weight loss in those who had the bypass surgery. While he didn’t speculate on the reasons for this finding, he did affirm his belief that gastric bypass is the best option for most obese patients with comorbid diabetes.
"I feel very comfortable recommending it" for these patients, said Dr. Morton of the Stanford (Calif.) University. "There are, of course, other clinical conditions to consider when deciding [among] bypass, banding, and sleeve, but if the only consideration is around diabetes, I’m 100% comfortable in recommending it for obese diabetics."
He presented the 1-year follow-up data on 1,792 obese patients who underwent bariatric surgery. Of these, 1,364 had a Roux-en-Y bypass; 264 had a sleeve gastrectomy; and 164 had adjustable gastric banding.
The patients were a mean of about 46 years old. Body mass index was statistically, but not clinically, different between the groups (bypass 47 kg/m2; band 44 kg/m2; sleeve 44 kg/m2). Waist circumference ranged from 51 to 53 inches. About 75% of the group was female and more than half, white.
Overall, about one-third of each group had type 2 diabetes. Most were taking only oral medications. About 5% took only insulin, and about a quarter took both oral agents and insulin. At baseline, the mean HbA1c was more than 7% in each group. The mean fasting insulin was 36 microU/mL in the bypass group, 28 microU/mL in the band group, and 32 microU/mL in the sleeve group.
By 12 months after surgery, patients with diabetes who had bypass had lost the most weight – a mean of 71% of their excess body weight, compared with 38% in the banding group and 50% in the sleeve group.
Those who had the bypass surgery also experienced the biggest change in their HbA1c – dropping almost 16% to a mean of 5.8%. Patients in the other two groups experienced a mean drop of 10%, resulting in HbA1c levels of right around 6%.
Fasting insulin levels also improved significantly more in the bypass group, falling from a baseline mean of 56 microU/mL to 7.8 microU/mL – a decrease of 68%. In the band group, the level fell from 28 microU/mL to 12 microU/mL – a 52% decrease. In the sleeve group, levels fell from 32 microU/mL to 10 microU/mL – a 61% decrease.
"Fasting insulin is also considered an independent marker of cardiac risk," Dr. Morton added, indicating that the risk of cardiovascular problems would fall along with insulin levels.
Blood glucose improved significantly more in the bypass group, falling from a baseline mean of 149 mg/dL to 101 mg/dL – a 22% change. In the band group, the level fell from 140 mg/dL to 125 mg/dL (8%) and in the sleeve group, from 130 mg/dL to 118 mg/dL (5.6%).
A multivariate analysis controlled for surgery type, sex, body mass index, race, age, and insurance status. Of these factors, surgery type was the strongest predictor, with bypass patients three times more likely to achieve that goal than those undergoing banding or sleeve placement.
Dr. Morton did not present his complication data. However, during the discussion period, he said the three procedures were similarly safe. The gastric sleeve group had a higher leak rate than did the other groups, but that remained less than 1%. Readmission rates were comparable, he said, but he did not provide that number.
"Any time you consider this, it has to be a risk/benefit analysis," he said. "It’s our philosophy that for obese patients with severe diabetes, we approach them first with the bypass because it has the most proven track record over time."
Dr. Morton has up to 7 years of data on some of the patients, and said he is now analyzing that. But when questioned about durability, he agreed that diabetes can recur in the rather common scenario of a patient regaining weight. "At the end of the day, though, what’s important is that the obese patient with diabetes gets treatment. All three surgeries demonstrated some improvement, and I believe that any surgery is better than no surgery at all."
Dr. Morton has received research support from Covidien.
To earn 0.25 hours AMA PRA Category 1 credit after reading this article, take the post-test here.
AT THE ACS CLINICAL CONGRESS