User login
CTAD: Revived Alzheimer’s symptomatic drug set to test as donepezil combo
BARCELONA – RVT-101, a novel small molecule that increases acetylcholine release, is poised for what its developers say could be its pivotal trial for U.S. approval.
The MINDSET study will examine the effect of the 5-HT6 receptor antagonist RVT-101, which will be given in conjunction with donepezil.
As an acetylcholine booster, RVT-101 would not be a disease-modifying therapy. And like other symptomatic drugs, it would not alter the trajectory of Alzheimer’s disease.
Launched last month, MINDSET will be conducted in North America, South America, Europe, and Asia. The study investigators want to enroll least 1,000 patients with mild to moderate Alzheimer’s disease who are on stable doses of donepezil, Dr. Lawrence Friedhoff said at the Clinical Trials on Alzheimer’s Disease conference.
Axovant Sciences’ RVT-101, formerly developed as SB742457 by GlaxoSmithKline, has been investigated in 13 studies of both monotherapy and adjunctive therapy in more than 2,000 patients with Alzheimer’s, said Dr. Friedhoff, chief development officer at Axovant Sciences. As monotherapy, it was ineffective, conferring no significant benefits on either cognition or function.
The earlier study upon which MINDSET is modeled was a modestly successful phase IIb dose-ranging trial that comprised 684 patients. At 48 weeks, the combination of 35 mg SB742457 (RVT-101) and donepezil conferred about a 2-point benefit on the Alzheimer’s Disease Assessment Scale–Cognitive (ADAS-cog) and the Alzheimer’s Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) over donepezil alone. Neither the 35-mg dose nor the 15-mg dose, which was also tested, conferred any benefit on the Clinical Dementia Rating Scale–Sum of Boxes (CDR-SB) test.
“Regulators have told us that if our results are similar, then this study will be suitable for submission for approval,” Dr. Friedhoff said. “We have a reasonably high level of confidence that it will also be effective in this trial.”
The 24-week MINDSET will randomize patients to their donepezil dosage plus either placebo or 35 mg RVT-101 daily. Coprimary endpoints are the ADAS-cog and the ADCS-ADL. The CDR-SB is not an endpoint in MINDSET, which Dr. Friedhoff noted was designed to maximize the chance of success. A 12-month open-label extension study focusing on long-term safety will follow the randomized trial.
GSK ceased development of RVT-101 in 2010 without public explanation. However, a recent review of several of the GSK studies suggests that it simply didn’t meet the studies’ success criteria.
Axovant was formed in 2014 to give RVT-101 a new shot as an add-on treatment. The combination makes sense, Dr. Friedhoff said.
“It increases acetylcholine in the brain. We already know this is an effective way to treat Alzheimer’s, because our acetylcholinesterase inhibitors achieve this in a different way, by inhibiting the breakdown of acetylcholine,” Dr. Friedhoff explained. “It makes sense that they should work well together. It doesn’t help much to increase the release if it’s broken down quickly.”
RVT-101 has an attractive safety profile, Dr. Friedhoff said. It had a low rate of gastrointestinal side effects (nausea 2%, diarrhea 5%) and was associated with very few falls in prior studies (about 3%). That probably is because the 5-HT6 receptor is located only in the central nervous system; there is no peripheral binding, so cholinergic toxicities to other systems are limited, he noted.
That relatively benign side effect profile is probably reflected in the study’s large completer rate, Dr. Friedhoff said. The placebo and 35-mg dose had virtually identical completer rates of 88% and 89%, respectively. The dropout rates were low and similar in both groups.
Axovant also aims to investigate RVT-101 in patients with Lewy body dementia, Dr. Friedhoff added. The company expects to launch a phase IIb clinical study early next year.
Axovant Sciences is sponsoring the MINDSET study. Dr. Friedhoff is an employee of the company.
[email protected]
On Twitter @Alz_Gal
BARCELONA – RVT-101, a novel small molecule that increases acetylcholine release, is poised for what its developers say could be its pivotal trial for U.S. approval.
The MINDSET study will examine the effect of the 5-HT6 receptor antagonist RVT-101, which will be given in conjunction with donepezil.
As an acetylcholine booster, RVT-101 would not be a disease-modifying therapy. And like other symptomatic drugs, it would not alter the trajectory of Alzheimer’s disease.
Launched last month, MINDSET will be conducted in North America, South America, Europe, and Asia. The study investigators want to enroll least 1,000 patients with mild to moderate Alzheimer’s disease who are on stable doses of donepezil, Dr. Lawrence Friedhoff said at the Clinical Trials on Alzheimer’s Disease conference.
Axovant Sciences’ RVT-101, formerly developed as SB742457 by GlaxoSmithKline, has been investigated in 13 studies of both monotherapy and adjunctive therapy in more than 2,000 patients with Alzheimer’s, said Dr. Friedhoff, chief development officer at Axovant Sciences. As monotherapy, it was ineffective, conferring no significant benefits on either cognition or function.
The earlier study upon which MINDSET is modeled was a modestly successful phase IIb dose-ranging trial that comprised 684 patients. At 48 weeks, the combination of 35 mg SB742457 (RVT-101) and donepezil conferred about a 2-point benefit on the Alzheimer’s Disease Assessment Scale–Cognitive (ADAS-cog) and the Alzheimer’s Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) over donepezil alone. Neither the 35-mg dose nor the 15-mg dose, which was also tested, conferred any benefit on the Clinical Dementia Rating Scale–Sum of Boxes (CDR-SB) test.
“Regulators have told us that if our results are similar, then this study will be suitable for submission for approval,” Dr. Friedhoff said. “We have a reasonably high level of confidence that it will also be effective in this trial.”
The 24-week MINDSET will randomize patients to their donepezil dosage plus either placebo or 35 mg RVT-101 daily. Coprimary endpoints are the ADAS-cog and the ADCS-ADL. The CDR-SB is not an endpoint in MINDSET, which Dr. Friedhoff noted was designed to maximize the chance of success. A 12-month open-label extension study focusing on long-term safety will follow the randomized trial.
GSK ceased development of RVT-101 in 2010 without public explanation. However, a recent review of several of the GSK studies suggests that it simply didn’t meet the studies’ success criteria.
Axovant was formed in 2014 to give RVT-101 a new shot as an add-on treatment. The combination makes sense, Dr. Friedhoff said.
“It increases acetylcholine in the brain. We already know this is an effective way to treat Alzheimer’s, because our acetylcholinesterase inhibitors achieve this in a different way, by inhibiting the breakdown of acetylcholine,” Dr. Friedhoff explained. “It makes sense that they should work well together. It doesn’t help much to increase the release if it’s broken down quickly.”
RVT-101 has an attractive safety profile, Dr. Friedhoff said. It had a low rate of gastrointestinal side effects (nausea 2%, diarrhea 5%) and was associated with very few falls in prior studies (about 3%). That probably is because the 5-HT6 receptor is located only in the central nervous system; there is no peripheral binding, so cholinergic toxicities to other systems are limited, he noted.
That relatively benign side effect profile is probably reflected in the study’s large completer rate, Dr. Friedhoff said. The placebo and 35-mg dose had virtually identical completer rates of 88% and 89%, respectively. The dropout rates were low and similar in both groups.
Axovant also aims to investigate RVT-101 in patients with Lewy body dementia, Dr. Friedhoff added. The company expects to launch a phase IIb clinical study early next year.
Axovant Sciences is sponsoring the MINDSET study. Dr. Friedhoff is an employee of the company.
[email protected]
On Twitter @Alz_Gal
BARCELONA – RVT-101, a novel small molecule that increases acetylcholine release, is poised for what its developers say could be its pivotal trial for U.S. approval.
The MINDSET study will examine the effect of the 5-HT6 receptor antagonist RVT-101, which will be given in conjunction with donepezil.
As an acetylcholine booster, RVT-101 would not be a disease-modifying therapy. And like other symptomatic drugs, it would not alter the trajectory of Alzheimer’s disease.
Launched last month, MINDSET will be conducted in North America, South America, Europe, and Asia. The study investigators want to enroll least 1,000 patients with mild to moderate Alzheimer’s disease who are on stable doses of donepezil, Dr. Lawrence Friedhoff said at the Clinical Trials on Alzheimer’s Disease conference.
Axovant Sciences’ RVT-101, formerly developed as SB742457 by GlaxoSmithKline, has been investigated in 13 studies of both monotherapy and adjunctive therapy in more than 2,000 patients with Alzheimer’s, said Dr. Friedhoff, chief development officer at Axovant Sciences. As monotherapy, it was ineffective, conferring no significant benefits on either cognition or function.
The earlier study upon which MINDSET is modeled was a modestly successful phase IIb dose-ranging trial that comprised 684 patients. At 48 weeks, the combination of 35 mg SB742457 (RVT-101) and donepezil conferred about a 2-point benefit on the Alzheimer’s Disease Assessment Scale–Cognitive (ADAS-cog) and the Alzheimer’s Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) over donepezil alone. Neither the 35-mg dose nor the 15-mg dose, which was also tested, conferred any benefit on the Clinical Dementia Rating Scale–Sum of Boxes (CDR-SB) test.
“Regulators have told us that if our results are similar, then this study will be suitable for submission for approval,” Dr. Friedhoff said. “We have a reasonably high level of confidence that it will also be effective in this trial.”
The 24-week MINDSET will randomize patients to their donepezil dosage plus either placebo or 35 mg RVT-101 daily. Coprimary endpoints are the ADAS-cog and the ADCS-ADL. The CDR-SB is not an endpoint in MINDSET, which Dr. Friedhoff noted was designed to maximize the chance of success. A 12-month open-label extension study focusing on long-term safety will follow the randomized trial.
GSK ceased development of RVT-101 in 2010 without public explanation. However, a recent review of several of the GSK studies suggests that it simply didn’t meet the studies’ success criteria.
Axovant was formed in 2014 to give RVT-101 a new shot as an add-on treatment. The combination makes sense, Dr. Friedhoff said.
“It increases acetylcholine in the brain. We already know this is an effective way to treat Alzheimer’s, because our acetylcholinesterase inhibitors achieve this in a different way, by inhibiting the breakdown of acetylcholine,” Dr. Friedhoff explained. “It makes sense that they should work well together. It doesn’t help much to increase the release if it’s broken down quickly.”
RVT-101 has an attractive safety profile, Dr. Friedhoff said. It had a low rate of gastrointestinal side effects (nausea 2%, diarrhea 5%) and was associated with very few falls in prior studies (about 3%). That probably is because the 5-HT6 receptor is located only in the central nervous system; there is no peripheral binding, so cholinergic toxicities to other systems are limited, he noted.
That relatively benign side effect profile is probably reflected in the study’s large completer rate, Dr. Friedhoff said. The placebo and 35-mg dose had virtually identical completer rates of 88% and 89%, respectively. The dropout rates were low and similar in both groups.
Axovant also aims to investigate RVT-101 in patients with Lewy body dementia, Dr. Friedhoff added. The company expects to launch a phase IIb clinical study early next year.
Axovant Sciences is sponsoring the MINDSET study. Dr. Friedhoff is an employee of the company.
[email protected]
On Twitter @Alz_Gal
AT CTAD
Key clinical point: RVT-101, a 5-HT6 antagonist, will be explored as a symptomatic treatment for Alzheimer’s disease in combination with donepezil.
Major finding: In its most successful past trial, 35 mg RVT-101 given with background donepezil conferred about a 2-point benefit on cognitive and functional outcomes over donepezil alone.
Data source: MINDSET, which randomized 1,000 patients with mild to moderate Alzheimer’s disease to their background donepezil dosage plus either placebo or 35 mg RVT-101.
Disclosures: Axovant Sciences is sponsoring the MINDSET study. Dr. Friedhoff is an employee of the company.
Omega-3 plus physical, mental training prevented cognitive decline for 3 years
BARCELONA – A combination of cognitive and physical training plus omega-3 fatty acid supplements boosted cognitive performance significantly in a group of people with subjective memory complaints.
Among those with more serious memory issues, the combination worked even better, preventing any decline at all over the 3-year study period, Dr. Bruno Vellas said at the Clinical Trials on Alzheimer’s Disease conference.
The program was also especially effective in subjects who were homozygous for the high-risk e4 allele of the apolipoprotein E gene, said Dr. Vellas, chief of the Alzheimer’s Disease Clinical and Research Center at the University Hospital Center in Toulouse, France.
The findings must be confirmed in larger studies, he said, but they strengthen the growing body of evidence supporting nonpharmacologic Alzheimer’s prevention tactics.
“This confirms the data from FINGER. In FINGER we had some improvement, but here we are able to show that we can prevent decline, which is maybe even more important, especially for high-risk patients,” Dr. Vellas said.
The Multidomain Alzheimer Preventive Trial (MAPT) comprised 1,680 people who had reported subjective memory complaints to their primary care physician. It examined the effect of a comprehensive cognitive, nutrition, and physical training program, with or without omega-3 fatty acid supplementation. In addition to the cognitive outcome, MAPT used functional PET imaging to examine brain glucose metabolism at baseline and 6 and 12 months, and amyloid plaque burden at 12-18 months.
Cognitive training focused on reasoning and memory training. Its main objective was to teach participants how to use adaptive strategies in solving everyday problems – for example, using mnemonics to remember a grocery list.
The physical training component involved at least 150 minutes of moderate exercise each week; walking 30 minutes/day was the most frequently recommended. Participants also received an individually designed at-home exercise plan that was reviewed and updated every 6 months.
Nutritional counseling was based on the French National Nutrition and Health Program. Eight key guidelines on healthy diet were discussed.
These interventions were all offered in small groups, during 12 2-hour sessions in the first 2 months. There were brief individual interviews every 6 months, and intensive booster sessions after 1 year and 2 years.
Each supplement capsule contained a specially compounded combination of 400 mg docosahexaenoic acid (DHA) and 112.5 mg eicosapentaenoic acid (EPA). It was taken twice a day.
The trial consisted of four arms: omega-3 supplements (DHA) alone; placebo capsules alone; training without DHA supplements; and both training and DHA
The mean age of the study participants was 75 years. While the mean Mini Mental State Exam score was 28, indicating no cognitive impairment, some subjects did dip down into the mild cognitive impairment range. About 40% had a Clinical Dementia Rating sum of boxes (CDR) score of 0.5 (very mild Alzheimer’s); the rest had a 0 score. No one had frank dementia. About 23% were positive for APOE e4.
Compliance with the program was very good, Dr. Vellas said: a mean of 71% for the training programs and 84% for the supplements over the 3 years.
In both the intent-to-treat and per-protocol analyses, the training program and the combination of training and DHA were significantly more effective than either DHA or placebo.
Those taking DHA alone or placebo declined similarly from baseline. Those in the training program (with or without DHA) experienced a significant uptick in cognitive score during the first year. During the second year that gain fell, but it still remained significantly above baseline in both groups.
Dr. Vellas also examined the composite cognitive endpoint in the subgroup with a 0.5 score on the CDR scale. Those taking placebo or DHA-only experienced no improvement and fell significantly below baseline by the end of the study. Those who were in the training program only experienced a boost in scores in the first 6 months, which rapidly fell off. By 2 years, the scores were significantly below baseline, although not as depressed as those in the placebo and DHA-only groups.
The best outcome occurred in the training program plus DHA group. Their test scores remained completely stable over the entire period, suggesting that the combination intervention prevented cognitive decline.
It conferred the biggest benefit on those who were APOE e4-positive. Their scores rose significantly by 12 months and, although they did begin to decline, they remained significantly improved through the study’s end. The training program alone also improved the cognitive score by 12 months, but that dipped back to baseline by the end of the study. Those taking placebo or DHA alone experienced significant score declines.
Amyloid imaging was not performed at baseline, but was done 12-18 months into the study. At that point, the combination intervention group had the lowest percentage of amyloid-positive subjects (22%). The rate of amyloid positivity was 34% in the training program alone; 46% in the DHA group alone; and 51% in the placebo group.
It’s not possible to determine whether the intervention altered the trajectory of amyloid deposition, Dr. Vellas said; that will be explored in a different study.
PET imaging showed striking between-group differences in glucose metabolism. Those in the training-alone group experienced a transient metabolic increase in the right and left temporal regions, which, compared with placebo, was significant at 6 months but not at 12 months.
In the DHA-only group, there was no difference at 6 months, compared with placebo. At 12 months, there was significantly more activity in the left supplementary motor area.
The combination group showed significant increases in metabolism in a number of regions, including the right hemisphere cluster (insula/putamen/amygdala/superior and middle temporal region); the left middle temporal pole fusiform gyrus and anterior temporal region; the right middle and inferior frontal region; and the right rolandic opercula.
The European Commission funded the study. Dr. Vellas had no financial disclosures.
BARCELONA – A combination of cognitive and physical training plus omega-3 fatty acid supplements boosted cognitive performance significantly in a group of people with subjective memory complaints.
Among those with more serious memory issues, the combination worked even better, preventing any decline at all over the 3-year study period, Dr. Bruno Vellas said at the Clinical Trials on Alzheimer’s Disease conference.
The program was also especially effective in subjects who were homozygous for the high-risk e4 allele of the apolipoprotein E gene, said Dr. Vellas, chief of the Alzheimer’s Disease Clinical and Research Center at the University Hospital Center in Toulouse, France.
The findings must be confirmed in larger studies, he said, but they strengthen the growing body of evidence supporting nonpharmacologic Alzheimer’s prevention tactics.
“This confirms the data from FINGER. In FINGER we had some improvement, but here we are able to show that we can prevent decline, which is maybe even more important, especially for high-risk patients,” Dr. Vellas said.
The Multidomain Alzheimer Preventive Trial (MAPT) comprised 1,680 people who had reported subjective memory complaints to their primary care physician. It examined the effect of a comprehensive cognitive, nutrition, and physical training program, with or without omega-3 fatty acid supplementation. In addition to the cognitive outcome, MAPT used functional PET imaging to examine brain glucose metabolism at baseline and 6 and 12 months, and amyloid plaque burden at 12-18 months.
Cognitive training focused on reasoning and memory training. Its main objective was to teach participants how to use adaptive strategies in solving everyday problems – for example, using mnemonics to remember a grocery list.
The physical training component involved at least 150 minutes of moderate exercise each week; walking 30 minutes/day was the most frequently recommended. Participants also received an individually designed at-home exercise plan that was reviewed and updated every 6 months.
Nutritional counseling was based on the French National Nutrition and Health Program. Eight key guidelines on healthy diet were discussed.
These interventions were all offered in small groups, during 12 2-hour sessions in the first 2 months. There were brief individual interviews every 6 months, and intensive booster sessions after 1 year and 2 years.
Each supplement capsule contained a specially compounded combination of 400 mg docosahexaenoic acid (DHA) and 112.5 mg eicosapentaenoic acid (EPA). It was taken twice a day.
The trial consisted of four arms: omega-3 supplements (DHA) alone; placebo capsules alone; training without DHA supplements; and both training and DHA
The mean age of the study participants was 75 years. While the mean Mini Mental State Exam score was 28, indicating no cognitive impairment, some subjects did dip down into the mild cognitive impairment range. About 40% had a Clinical Dementia Rating sum of boxes (CDR) score of 0.5 (very mild Alzheimer’s); the rest had a 0 score. No one had frank dementia. About 23% were positive for APOE e4.
Compliance with the program was very good, Dr. Vellas said: a mean of 71% for the training programs and 84% for the supplements over the 3 years.
In both the intent-to-treat and per-protocol analyses, the training program and the combination of training and DHA were significantly more effective than either DHA or placebo.
Those taking DHA alone or placebo declined similarly from baseline. Those in the training program (with or without DHA) experienced a significant uptick in cognitive score during the first year. During the second year that gain fell, but it still remained significantly above baseline in both groups.
Dr. Vellas also examined the composite cognitive endpoint in the subgroup with a 0.5 score on the CDR scale. Those taking placebo or DHA-only experienced no improvement and fell significantly below baseline by the end of the study. Those who were in the training program only experienced a boost in scores in the first 6 months, which rapidly fell off. By 2 years, the scores were significantly below baseline, although not as depressed as those in the placebo and DHA-only groups.
The best outcome occurred in the training program plus DHA group. Their test scores remained completely stable over the entire period, suggesting that the combination intervention prevented cognitive decline.
It conferred the biggest benefit on those who were APOE e4-positive. Their scores rose significantly by 12 months and, although they did begin to decline, they remained significantly improved through the study’s end. The training program alone also improved the cognitive score by 12 months, but that dipped back to baseline by the end of the study. Those taking placebo or DHA alone experienced significant score declines.
Amyloid imaging was not performed at baseline, but was done 12-18 months into the study. At that point, the combination intervention group had the lowest percentage of amyloid-positive subjects (22%). The rate of amyloid positivity was 34% in the training program alone; 46% in the DHA group alone; and 51% in the placebo group.
It’s not possible to determine whether the intervention altered the trajectory of amyloid deposition, Dr. Vellas said; that will be explored in a different study.
PET imaging showed striking between-group differences in glucose metabolism. Those in the training-alone group experienced a transient metabolic increase in the right and left temporal regions, which, compared with placebo, was significant at 6 months but not at 12 months.
In the DHA-only group, there was no difference at 6 months, compared with placebo. At 12 months, there was significantly more activity in the left supplementary motor area.
The combination group showed significant increases in metabolism in a number of regions, including the right hemisphere cluster (insula/putamen/amygdala/superior and middle temporal region); the left middle temporal pole fusiform gyrus and anterior temporal region; the right middle and inferior frontal region; and the right rolandic opercula.
The European Commission funded the study. Dr. Vellas had no financial disclosures.
BARCELONA – A combination of cognitive and physical training plus omega-3 fatty acid supplements boosted cognitive performance significantly in a group of people with subjective memory complaints.
Among those with more serious memory issues, the combination worked even better, preventing any decline at all over the 3-year study period, Dr. Bruno Vellas said at the Clinical Trials on Alzheimer’s Disease conference.
The program was also especially effective in subjects who were homozygous for the high-risk e4 allele of the apolipoprotein E gene, said Dr. Vellas, chief of the Alzheimer’s Disease Clinical and Research Center at the University Hospital Center in Toulouse, France.
The findings must be confirmed in larger studies, he said, but they strengthen the growing body of evidence supporting nonpharmacologic Alzheimer’s prevention tactics.
“This confirms the data from FINGER. In FINGER we had some improvement, but here we are able to show that we can prevent decline, which is maybe even more important, especially for high-risk patients,” Dr. Vellas said.
The Multidomain Alzheimer Preventive Trial (MAPT) comprised 1,680 people who had reported subjective memory complaints to their primary care physician. It examined the effect of a comprehensive cognitive, nutrition, and physical training program, with or without omega-3 fatty acid supplementation. In addition to the cognitive outcome, MAPT used functional PET imaging to examine brain glucose metabolism at baseline and 6 and 12 months, and amyloid plaque burden at 12-18 months.
Cognitive training focused on reasoning and memory training. Its main objective was to teach participants how to use adaptive strategies in solving everyday problems – for example, using mnemonics to remember a grocery list.
The physical training component involved at least 150 minutes of moderate exercise each week; walking 30 minutes/day was the most frequently recommended. Participants also received an individually designed at-home exercise plan that was reviewed and updated every 6 months.
Nutritional counseling was based on the French National Nutrition and Health Program. Eight key guidelines on healthy diet were discussed.
These interventions were all offered in small groups, during 12 2-hour sessions in the first 2 months. There were brief individual interviews every 6 months, and intensive booster sessions after 1 year and 2 years.
Each supplement capsule contained a specially compounded combination of 400 mg docosahexaenoic acid (DHA) and 112.5 mg eicosapentaenoic acid (EPA). It was taken twice a day.
The trial consisted of four arms: omega-3 supplements (DHA) alone; placebo capsules alone; training without DHA supplements; and both training and DHA
The mean age of the study participants was 75 years. While the mean Mini Mental State Exam score was 28, indicating no cognitive impairment, some subjects did dip down into the mild cognitive impairment range. About 40% had a Clinical Dementia Rating sum of boxes (CDR) score of 0.5 (very mild Alzheimer’s); the rest had a 0 score. No one had frank dementia. About 23% were positive for APOE e4.
Compliance with the program was very good, Dr. Vellas said: a mean of 71% for the training programs and 84% for the supplements over the 3 years.
In both the intent-to-treat and per-protocol analyses, the training program and the combination of training and DHA were significantly more effective than either DHA or placebo.
Those taking DHA alone or placebo declined similarly from baseline. Those in the training program (with or without DHA) experienced a significant uptick in cognitive score during the first year. During the second year that gain fell, but it still remained significantly above baseline in both groups.
Dr. Vellas also examined the composite cognitive endpoint in the subgroup with a 0.5 score on the CDR scale. Those taking placebo or DHA-only experienced no improvement and fell significantly below baseline by the end of the study. Those who were in the training program only experienced a boost in scores in the first 6 months, which rapidly fell off. By 2 years, the scores were significantly below baseline, although not as depressed as those in the placebo and DHA-only groups.
The best outcome occurred in the training program plus DHA group. Their test scores remained completely stable over the entire period, suggesting that the combination intervention prevented cognitive decline.
It conferred the biggest benefit on those who were APOE e4-positive. Their scores rose significantly by 12 months and, although they did begin to decline, they remained significantly improved through the study’s end. The training program alone also improved the cognitive score by 12 months, but that dipped back to baseline by the end of the study. Those taking placebo or DHA alone experienced significant score declines.
Amyloid imaging was not performed at baseline, but was done 12-18 months into the study. At that point, the combination intervention group had the lowest percentage of amyloid-positive subjects (22%). The rate of amyloid positivity was 34% in the training program alone; 46% in the DHA group alone; and 51% in the placebo group.
It’s not possible to determine whether the intervention altered the trajectory of amyloid deposition, Dr. Vellas said; that will be explored in a different study.
PET imaging showed striking between-group differences in glucose metabolism. Those in the training-alone group experienced a transient metabolic increase in the right and left temporal regions, which, compared with placebo, was significant at 6 months but not at 12 months.
In the DHA-only group, there was no difference at 6 months, compared with placebo. At 12 months, there was significantly more activity in the left supplementary motor area.
The combination group showed significant increases in metabolism in a number of regions, including the right hemisphere cluster (insula/putamen/amygdala/superior and middle temporal region); the left middle temporal pole fusiform gyrus and anterior temporal region; the right middle and inferior frontal region; and the right rolandic opercula.
The European Commission funded the study. Dr. Vellas had no financial disclosures.
AT CTAD
Key clinical point: Exercise, nutrition, and mental training combined with omega-3 supplements stopped cognitive decline.
Major finding: The combination of omega-3 supplementation and a comprehensive training program prevented any decline in cognitive scores during 3 years.
Data source: The Multidomain Alzheimer Preventive Trial randomized 1,680 subjects to four treatment arms.
Disclosures: The European Commission funded the study. Dr. Vellas had no financial disclosures.
High-risk patients fared best in lifestyle intervention trial for cognitive decline
BARCELONA – New, unpublished data from the FINGER multimodal lifestyle intervention study indicates that targeting nutrition, exercise, and metabolic and cardiovascular risk factors boosted overall cognitive performance, memory, and executive function to the greatest extent in elderly people at risk of cognitive decline who carried the apolipoprotein E–epsilon 4 allele.
“The findings were especially clear in changes on the comprehensive neuropsychological test battery,” which was the study’s primary endpoint, Dr. Miia Kivipelto of the Karolinska Institute, Stockholm, said at the Clinical Trials on Alzheimer’s Disease conference. “This is a very effective intervention that we can recommend, especially for people with this genetic risk factor.”
The randomized, controlled FINGER (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability) trial was a proof-of-concept study designed to show how a multimodal lifestyle intervention might not only slow or prevent cognitive decline, but help improve cognition among patients who are already experiencing decline. It enrolled 1,260 participants aged 60-77 years who were cognitively normal at baseline but at risk for decline based on their CAIDE (Cardiovascular Risk Factors, Aging, and Incidence of Dementia) risk score.
The cohort was randomized to a control program of 2 years of regular health counseling with their physician, or to the intervention, which consisted of:
• Dietary counseling with recommendations to consume increased amounts of fruit, vegetables, whole grains, lean protein, and healthy fats.
• Progressive aerobic exercise and weight training, conducted by physical therapists, several times each week.
• Cognitive training several times a week with a computer program that targeted executive processes, working memory, episodic memory, and mental speed.
• Managing metabolic and cardiovascular risk factors, including blood pressure, weight, and body mass index. This was addressed in group sessions and with visits to participants’ own physicians.
The subjects had a mean age of 69 years at baseline. About 70% reported physical activity at least twice a week. About half reported eating fish at least twice a week, and 60% reported a daily intake of vegetables. Most (65%) had hypertension and hypercholesterolemia (70%). Another 13% had diabetes, and 5% had a prior heart attack.
The primary endpoint was change on an extended version of the neuropsychological test battery, which was conducted at baseline and at months 12 and 24. Secondary endpoints were changes on the individual components of executive function, memory, and processing speed.
By the end of the study, subjects in the intervention group experienced a significant, 25% greater improvement on the overall score than did those in the control group. Improvements on the secondary measures were significant for the intervention group as well: 150% better than the control group in processing speed, 83% better in executive functioning, and 40% better in short-term memory.
The risk of cognitive decline increased by 30% in the control group by the study’s end, whereas subjects in the intervention group experienced no increased risk. The Lancet published the study’s main results earlier this year (Lancet. 2015;385[9984]:2255-63).
The new data that Dr. Kivipelto described showed that people with at least one apolipoprotein E (APOE)–epsilon 4 allele reaped a significantly greater benefit than did those without the high-risk allele. In the intervention group, carriers had an estimated mean change z-score on the overall neuropsychological test battery that was significantly higher than in noncarriers (0.17 vs. 0.10, respectively; P = .045), whereas the difference between carriers and noncarriers in the control group was not statistically significant (0.22 vs. 0.19; P = .28). Carriers in the intervention group also tended to benefit more than did noncarriers in the individual domains.
Dr. Kivipelto explored how the program might be working on a molecular level. In an analysis of 756 subjects, including 244 APOE–epsilon 4 carriers, she found attenuated telomere shortening in carriers in the intervention group, but not in carriers assigned to the control group or any of the noncarriers. This attenuation was also more pronounced in younger subjects, Dr. Kivipelto added.
She also presented some preliminary data on how the intervention improved function and quality of life. “There was some decline after 2 years in the control group, but the intervention group remained stable,” Dr. Kivipelto said. “There was also significant improvement in general health, mental function, and social function in the intervention group.”
General daily function was good for the entire cohort at baseline, but by the end of the study, significant differences had emerged, she said. “We were surprised to see that after 2 years, the control group actually had a 50% increased risk for at least one new difficulty with activities of daily living, and for those with no difficulties at baseline, the increased risk was even stronger, 76%.”
The program was not associated with any serious adverse events, or any adverse event at all other than musculoskeletal soreness from exercise activities. It also appeared to be practical and made a lasting impact, which was a gratifying finding, Dr. Kivipelto said. At the end of the study, intervention group participants had decreased their body mass index by about 0.8 kg/m2, which was significantly more than for control group subjects. Most reported that they were still eating fish and exercising at least twice a week and eating vegetables every day.
“FINGER is the first long-term trial to show that a multidomain intervention like this one can maintain and improve cognitive decline,” she said. “It important that we’ve also seen the program is feasible, has no obvious side effects, and that it’s not limited to cognitive domains. It also has a positive impact on function and quality of life.”
Dr. Kivipelto had no financial disclosures. The study was supported by grants from the Academy of Finland’s Responding to Public Health Challenges Research Programme, La Carita Foundation, the Alzheimer’s Association, the Alzheimer’s Research and Prevention Foundation, the Juho Vainio Foundation, Novo Nordisk Foundation, the Finnish Social Insurance Institution, the Finnish Ministry of Education and Culture Research, Salama Bint Hamdan Al Nahyan Foundation, the Axa Research Fund, and various University Hospitals in Finland.
*This story was updated on 11/6/2015.
BARCELONA – New, unpublished data from the FINGER multimodal lifestyle intervention study indicates that targeting nutrition, exercise, and metabolic and cardiovascular risk factors boosted overall cognitive performance, memory, and executive function to the greatest extent in elderly people at risk of cognitive decline who carried the apolipoprotein E–epsilon 4 allele.
“The findings were especially clear in changes on the comprehensive neuropsychological test battery,” which was the study’s primary endpoint, Dr. Miia Kivipelto of the Karolinska Institute, Stockholm, said at the Clinical Trials on Alzheimer’s Disease conference. “This is a very effective intervention that we can recommend, especially for people with this genetic risk factor.”
The randomized, controlled FINGER (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability) trial was a proof-of-concept study designed to show how a multimodal lifestyle intervention might not only slow or prevent cognitive decline, but help improve cognition among patients who are already experiencing decline. It enrolled 1,260 participants aged 60-77 years who were cognitively normal at baseline but at risk for decline based on their CAIDE (Cardiovascular Risk Factors, Aging, and Incidence of Dementia) risk score.
The cohort was randomized to a control program of 2 years of regular health counseling with their physician, or to the intervention, which consisted of:
• Dietary counseling with recommendations to consume increased amounts of fruit, vegetables, whole grains, lean protein, and healthy fats.
• Progressive aerobic exercise and weight training, conducted by physical therapists, several times each week.
• Cognitive training several times a week with a computer program that targeted executive processes, working memory, episodic memory, and mental speed.
• Managing metabolic and cardiovascular risk factors, including blood pressure, weight, and body mass index. This was addressed in group sessions and with visits to participants’ own physicians.
The subjects had a mean age of 69 years at baseline. About 70% reported physical activity at least twice a week. About half reported eating fish at least twice a week, and 60% reported a daily intake of vegetables. Most (65%) had hypertension and hypercholesterolemia (70%). Another 13% had diabetes, and 5% had a prior heart attack.
The primary endpoint was change on an extended version of the neuropsychological test battery, which was conducted at baseline and at months 12 and 24. Secondary endpoints were changes on the individual components of executive function, memory, and processing speed.
By the end of the study, subjects in the intervention group experienced a significant, 25% greater improvement on the overall score than did those in the control group. Improvements on the secondary measures were significant for the intervention group as well: 150% better than the control group in processing speed, 83% better in executive functioning, and 40% better in short-term memory.
The risk of cognitive decline increased by 30% in the control group by the study’s end, whereas subjects in the intervention group experienced no increased risk. The Lancet published the study’s main results earlier this year (Lancet. 2015;385[9984]:2255-63).
The new data that Dr. Kivipelto described showed that people with at least one apolipoprotein E (APOE)–epsilon 4 allele reaped a significantly greater benefit than did those without the high-risk allele. In the intervention group, carriers had an estimated mean change z-score on the overall neuropsychological test battery that was significantly higher than in noncarriers (0.17 vs. 0.10, respectively; P = .045), whereas the difference between carriers and noncarriers in the control group was not statistically significant (0.22 vs. 0.19; P = .28). Carriers in the intervention group also tended to benefit more than did noncarriers in the individual domains.
Dr. Kivipelto explored how the program might be working on a molecular level. In an analysis of 756 subjects, including 244 APOE–epsilon 4 carriers, she found attenuated telomere shortening in carriers in the intervention group, but not in carriers assigned to the control group or any of the noncarriers. This attenuation was also more pronounced in younger subjects, Dr. Kivipelto added.
She also presented some preliminary data on how the intervention improved function and quality of life. “There was some decline after 2 years in the control group, but the intervention group remained stable,” Dr. Kivipelto said. “There was also significant improvement in general health, mental function, and social function in the intervention group.”
General daily function was good for the entire cohort at baseline, but by the end of the study, significant differences had emerged, she said. “We were surprised to see that after 2 years, the control group actually had a 50% increased risk for at least one new difficulty with activities of daily living, and for those with no difficulties at baseline, the increased risk was even stronger, 76%.”
The program was not associated with any serious adverse events, or any adverse event at all other than musculoskeletal soreness from exercise activities. It also appeared to be practical and made a lasting impact, which was a gratifying finding, Dr. Kivipelto said. At the end of the study, intervention group participants had decreased their body mass index by about 0.8 kg/m2, which was significantly more than for control group subjects. Most reported that they were still eating fish and exercising at least twice a week and eating vegetables every day.
“FINGER is the first long-term trial to show that a multidomain intervention like this one can maintain and improve cognitive decline,” she said. “It important that we’ve also seen the program is feasible, has no obvious side effects, and that it’s not limited to cognitive domains. It also has a positive impact on function and quality of life.”
Dr. Kivipelto had no financial disclosures. The study was supported by grants from the Academy of Finland’s Responding to Public Health Challenges Research Programme, La Carita Foundation, the Alzheimer’s Association, the Alzheimer’s Research and Prevention Foundation, the Juho Vainio Foundation, Novo Nordisk Foundation, the Finnish Social Insurance Institution, the Finnish Ministry of Education and Culture Research, Salama Bint Hamdan Al Nahyan Foundation, the Axa Research Fund, and various University Hospitals in Finland.
*This story was updated on 11/6/2015.
BARCELONA – New, unpublished data from the FINGER multimodal lifestyle intervention study indicates that targeting nutrition, exercise, and metabolic and cardiovascular risk factors boosted overall cognitive performance, memory, and executive function to the greatest extent in elderly people at risk of cognitive decline who carried the apolipoprotein E–epsilon 4 allele.
“The findings were especially clear in changes on the comprehensive neuropsychological test battery,” which was the study’s primary endpoint, Dr. Miia Kivipelto of the Karolinska Institute, Stockholm, said at the Clinical Trials on Alzheimer’s Disease conference. “This is a very effective intervention that we can recommend, especially for people with this genetic risk factor.”
The randomized, controlled FINGER (Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability) trial was a proof-of-concept study designed to show how a multimodal lifestyle intervention might not only slow or prevent cognitive decline, but help improve cognition among patients who are already experiencing decline. It enrolled 1,260 participants aged 60-77 years who were cognitively normal at baseline but at risk for decline based on their CAIDE (Cardiovascular Risk Factors, Aging, and Incidence of Dementia) risk score.
The cohort was randomized to a control program of 2 years of regular health counseling with their physician, or to the intervention, which consisted of:
• Dietary counseling with recommendations to consume increased amounts of fruit, vegetables, whole grains, lean protein, and healthy fats.
• Progressive aerobic exercise and weight training, conducted by physical therapists, several times each week.
• Cognitive training several times a week with a computer program that targeted executive processes, working memory, episodic memory, and mental speed.
• Managing metabolic and cardiovascular risk factors, including blood pressure, weight, and body mass index. This was addressed in group sessions and with visits to participants’ own physicians.
The subjects had a mean age of 69 years at baseline. About 70% reported physical activity at least twice a week. About half reported eating fish at least twice a week, and 60% reported a daily intake of vegetables. Most (65%) had hypertension and hypercholesterolemia (70%). Another 13% had diabetes, and 5% had a prior heart attack.
The primary endpoint was change on an extended version of the neuropsychological test battery, which was conducted at baseline and at months 12 and 24. Secondary endpoints were changes on the individual components of executive function, memory, and processing speed.
By the end of the study, subjects in the intervention group experienced a significant, 25% greater improvement on the overall score than did those in the control group. Improvements on the secondary measures were significant for the intervention group as well: 150% better than the control group in processing speed, 83% better in executive functioning, and 40% better in short-term memory.
The risk of cognitive decline increased by 30% in the control group by the study’s end, whereas subjects in the intervention group experienced no increased risk. The Lancet published the study’s main results earlier this year (Lancet. 2015;385[9984]:2255-63).
The new data that Dr. Kivipelto described showed that people with at least one apolipoprotein E (APOE)–epsilon 4 allele reaped a significantly greater benefit than did those without the high-risk allele. In the intervention group, carriers had an estimated mean change z-score on the overall neuropsychological test battery that was significantly higher than in noncarriers (0.17 vs. 0.10, respectively; P = .045), whereas the difference between carriers and noncarriers in the control group was not statistically significant (0.22 vs. 0.19; P = .28). Carriers in the intervention group also tended to benefit more than did noncarriers in the individual domains.
Dr. Kivipelto explored how the program might be working on a molecular level. In an analysis of 756 subjects, including 244 APOE–epsilon 4 carriers, she found attenuated telomere shortening in carriers in the intervention group, but not in carriers assigned to the control group or any of the noncarriers. This attenuation was also more pronounced in younger subjects, Dr. Kivipelto added.
She also presented some preliminary data on how the intervention improved function and quality of life. “There was some decline after 2 years in the control group, but the intervention group remained stable,” Dr. Kivipelto said. “There was also significant improvement in general health, mental function, and social function in the intervention group.”
General daily function was good for the entire cohort at baseline, but by the end of the study, significant differences had emerged, she said. “We were surprised to see that after 2 years, the control group actually had a 50% increased risk for at least one new difficulty with activities of daily living, and for those with no difficulties at baseline, the increased risk was even stronger, 76%.”
The program was not associated with any serious adverse events, or any adverse event at all other than musculoskeletal soreness from exercise activities. It also appeared to be practical and made a lasting impact, which was a gratifying finding, Dr. Kivipelto said. At the end of the study, intervention group participants had decreased their body mass index by about 0.8 kg/m2, which was significantly more than for control group subjects. Most reported that they were still eating fish and exercising at least twice a week and eating vegetables every day.
“FINGER is the first long-term trial to show that a multidomain intervention like this one can maintain and improve cognitive decline,” she said. “It important that we’ve also seen the program is feasible, has no obvious side effects, and that it’s not limited to cognitive domains. It also has a positive impact on function and quality of life.”
Dr. Kivipelto had no financial disclosures. The study was supported by grants from the Academy of Finland’s Responding to Public Health Challenges Research Programme, La Carita Foundation, the Alzheimer’s Association, the Alzheimer’s Research and Prevention Foundation, the Juho Vainio Foundation, Novo Nordisk Foundation, the Finnish Social Insurance Institution, the Finnish Ministry of Education and Culture Research, Salama Bint Hamdan Al Nahyan Foundation, the Axa Research Fund, and various University Hospitals in Finland.
*This story was updated on 11/6/2015.
AT CTAD
Key clinical point: Targeting nutrition, exercise, and cardiovascular risk factors improved cognition in elderly people at risk of cognitive decline.
Major finding: Subjects in the intervention group experienced a significant 25% greater improvement on the overall neuropsychological test battery score than did those in the control group, with benefits in processing speed, memory, and executive function.
Data source: FINGER randomized 1,260 participants to a control program of 2 years of regular health counseling with their physician or to a multimodal lifestyle intervention.
Disclosures: Dr. Kivipelto had no financial disclosures. The study was supported by grants from the Academy of Finland’s Responding to Public Health Challenges Research Programme, La Carita Foundation, the Alzheimer’s Association, the Alzheimer’s Research and Prevention Foundation, the Juho Vainio Foundation, Novo Nordisk Foundation, the Finnish Social Insurance Institution, the Finnish Ministry of Education and Culture Research, Salama Bint Hamdan Al Nahyan Foundation, the Axa Research Fund, and various University Hospitals in Finland.
Guidelines: Follow low-risk benign thyroid nodules conservatively
LAKE BUENA VISTA, FLA. – Observation is a reasonable option for managing patients with benign thyroid nodules that display initial low-suspicion ultrasound patterns.
Presented at the International Thyroid Congress, the American Thyroid Association’s new guidelines on the management of thyroid nodules offer specific ultrasound patterns that help distinguish benign nodules from developing malignancy (Thyroid. 2015 Oct. doi: 10.1089/thy.2015.0020]).
They reflect new data obtained from studies conducted in the years since 2009, when the last guidelines were issued, Dr. Susan Mandel said at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
“We now know that about 20% of cytologically benign thyroid nodules will grow,” said Dr. Mandel of the University of Pennsylvania, Philadelphia. But very few of those will become malignant. According to a newly published study of 553 patients, 200 must be followed to identify one cancer that would have been missed (Thyroid. 2015 Oct 25;10:1115-20).
The new guidelines reflect findings like these and attempt to balance sensible follow-up while avoiding overtreatment, Dr. Mandel said. Diagnosed thyroid cancers are on the rise in the United States, from about 37,000 in 2009 to about 63,000 last year. But this is not because the cancer itself is more prevalent. More likely, it is the result of the increasing use of sonography or other imaging studies, which allows for earlier diagnosis and treatment. These technologies have a downside as well, leading to overdiagnosis and overtreatment of benign nodules that, most likely, would never undergo malignant transformation, Dr. Mandel said.
Nodules are most likely to grow in younger patients and those with longer follow-up, “which makes sense, as thyroid nodules grow very slowly.” Predominately solid nodules are also more likely to grow than are cystic nodules. Size at baseline is a controversial topic. There is some evidence that subcentimeter nodules are more likely to grow, while larger nodules are less likely.
Worrisome initial ultrasound patterns, however, should throw up a red flag even if the nodule is cytologically negative at baseline, Dr. Mandel said. These patterns are:
• Marked hypoechogeniticy.
• Microcalcifications.
• Irregular, microlobulated margins.
• Taller-than-wide shape.
• Incomplete or peripheral calcification.
These nodules should undergo a repeat ultrasound and a repeat ultrasound-guided fine needle biopsy within 6-12 months of the initial assessment, the guidelines recommend.
For nodules of any state, if (1) there is sonographic evidence of growth (20% increase in at least 2 nodule dimensions with a minimal increase of 2 mm or (2) more than 50% change in volume) or (3) development of new suspicious sonographic features, there should be an additional fine-needle biopsy. “Although the risk of malignancy after two benign cytology results is virtually zero, routine repeat biopsy is not a viable or cost-effective option because of the low false negative rate,” the document notes.
Small nodules (less than 1 cm) that have very low-suspicion ultrasound patterns (including spongiform nodules) don’t require routine follow-up sonograms. Nor do nodules larger than 5 mm without high-suspicion findings.
The guidelines also offer recommendations on treatment. There is no need to offer routine therapy with thyroid stimulating hormone (TSH) for benign nodules. “Though modest responses to therapy can be detected, the potential harm outweighs benefit for most patients,” the document states.
If benign nodules grow, surgery could be an option if they become large (greater than 4 cm), or cause compression or structural symptoms – or if there is clinical concern, although the document does not specify exactly what that could be.
Patients with growing nodules that are benign after a second biopsy should be regularly monitored. Most asymptomatic nodules demonstrating modest growth should be followed without intervention,” it says.
Recurrent benign cystic thyroid nodules may be treated surgically or with ethanol ablation, but asymptotic cystic nodules may be followed conservatively There are no data to guide the use of thyroid hormone treatment for benign nodules, even if they do grow.
The guidelines also offer recommendations for initial serum and molecular studies, and treatment for follicular and differentiated thyroid cancers.
Dr. Mandel had no financial disclosures.
LAKE BUENA VISTA, FLA. – Observation is a reasonable option for managing patients with benign thyroid nodules that display initial low-suspicion ultrasound patterns.
Presented at the International Thyroid Congress, the American Thyroid Association’s new guidelines on the management of thyroid nodules offer specific ultrasound patterns that help distinguish benign nodules from developing malignancy (Thyroid. 2015 Oct. doi: 10.1089/thy.2015.0020]).
They reflect new data obtained from studies conducted in the years since 2009, when the last guidelines were issued, Dr. Susan Mandel said at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
“We now know that about 20% of cytologically benign thyroid nodules will grow,” said Dr. Mandel of the University of Pennsylvania, Philadelphia. But very few of those will become malignant. According to a newly published study of 553 patients, 200 must be followed to identify one cancer that would have been missed (Thyroid. 2015 Oct 25;10:1115-20).
The new guidelines reflect findings like these and attempt to balance sensible follow-up while avoiding overtreatment, Dr. Mandel said. Diagnosed thyroid cancers are on the rise in the United States, from about 37,000 in 2009 to about 63,000 last year. But this is not because the cancer itself is more prevalent. More likely, it is the result of the increasing use of sonography or other imaging studies, which allows for earlier diagnosis and treatment. These technologies have a downside as well, leading to overdiagnosis and overtreatment of benign nodules that, most likely, would never undergo malignant transformation, Dr. Mandel said.
Nodules are most likely to grow in younger patients and those with longer follow-up, “which makes sense, as thyroid nodules grow very slowly.” Predominately solid nodules are also more likely to grow than are cystic nodules. Size at baseline is a controversial topic. There is some evidence that subcentimeter nodules are more likely to grow, while larger nodules are less likely.
Worrisome initial ultrasound patterns, however, should throw up a red flag even if the nodule is cytologically negative at baseline, Dr. Mandel said. These patterns are:
• Marked hypoechogeniticy.
• Microcalcifications.
• Irregular, microlobulated margins.
• Taller-than-wide shape.
• Incomplete or peripheral calcification.
These nodules should undergo a repeat ultrasound and a repeat ultrasound-guided fine needle biopsy within 6-12 months of the initial assessment, the guidelines recommend.
For nodules of any state, if (1) there is sonographic evidence of growth (20% increase in at least 2 nodule dimensions with a minimal increase of 2 mm or (2) more than 50% change in volume) or (3) development of new suspicious sonographic features, there should be an additional fine-needle biopsy. “Although the risk of malignancy after two benign cytology results is virtually zero, routine repeat biopsy is not a viable or cost-effective option because of the low false negative rate,” the document notes.
Small nodules (less than 1 cm) that have very low-suspicion ultrasound patterns (including spongiform nodules) don’t require routine follow-up sonograms. Nor do nodules larger than 5 mm without high-suspicion findings.
The guidelines also offer recommendations on treatment. There is no need to offer routine therapy with thyroid stimulating hormone (TSH) for benign nodules. “Though modest responses to therapy can be detected, the potential harm outweighs benefit for most patients,” the document states.
If benign nodules grow, surgery could be an option if they become large (greater than 4 cm), or cause compression or structural symptoms – or if there is clinical concern, although the document does not specify exactly what that could be.
Patients with growing nodules that are benign after a second biopsy should be regularly monitored. Most asymptomatic nodules demonstrating modest growth should be followed without intervention,” it says.
Recurrent benign cystic thyroid nodules may be treated surgically or with ethanol ablation, but asymptotic cystic nodules may be followed conservatively There are no data to guide the use of thyroid hormone treatment for benign nodules, even if they do grow.
The guidelines also offer recommendations for initial serum and molecular studies, and treatment for follicular and differentiated thyroid cancers.
Dr. Mandel had no financial disclosures.
LAKE BUENA VISTA, FLA. – Observation is a reasonable option for managing patients with benign thyroid nodules that display initial low-suspicion ultrasound patterns.
Presented at the International Thyroid Congress, the American Thyroid Association’s new guidelines on the management of thyroid nodules offer specific ultrasound patterns that help distinguish benign nodules from developing malignancy (Thyroid. 2015 Oct. doi: 10.1089/thy.2015.0020]).
They reflect new data obtained from studies conducted in the years since 2009, when the last guidelines were issued, Dr. Susan Mandel said at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
“We now know that about 20% of cytologically benign thyroid nodules will grow,” said Dr. Mandel of the University of Pennsylvania, Philadelphia. But very few of those will become malignant. According to a newly published study of 553 patients, 200 must be followed to identify one cancer that would have been missed (Thyroid. 2015 Oct 25;10:1115-20).
The new guidelines reflect findings like these and attempt to balance sensible follow-up while avoiding overtreatment, Dr. Mandel said. Diagnosed thyroid cancers are on the rise in the United States, from about 37,000 in 2009 to about 63,000 last year. But this is not because the cancer itself is more prevalent. More likely, it is the result of the increasing use of sonography or other imaging studies, which allows for earlier diagnosis and treatment. These technologies have a downside as well, leading to overdiagnosis and overtreatment of benign nodules that, most likely, would never undergo malignant transformation, Dr. Mandel said.
Nodules are most likely to grow in younger patients and those with longer follow-up, “which makes sense, as thyroid nodules grow very slowly.” Predominately solid nodules are also more likely to grow than are cystic nodules. Size at baseline is a controversial topic. There is some evidence that subcentimeter nodules are more likely to grow, while larger nodules are less likely.
Worrisome initial ultrasound patterns, however, should throw up a red flag even if the nodule is cytologically negative at baseline, Dr. Mandel said. These patterns are:
• Marked hypoechogeniticy.
• Microcalcifications.
• Irregular, microlobulated margins.
• Taller-than-wide shape.
• Incomplete or peripheral calcification.
These nodules should undergo a repeat ultrasound and a repeat ultrasound-guided fine needle biopsy within 6-12 months of the initial assessment, the guidelines recommend.
For nodules of any state, if (1) there is sonographic evidence of growth (20% increase in at least 2 nodule dimensions with a minimal increase of 2 mm or (2) more than 50% change in volume) or (3) development of new suspicious sonographic features, there should be an additional fine-needle biopsy. “Although the risk of malignancy after two benign cytology results is virtually zero, routine repeat biopsy is not a viable or cost-effective option because of the low false negative rate,” the document notes.
Small nodules (less than 1 cm) that have very low-suspicion ultrasound patterns (including spongiform nodules) don’t require routine follow-up sonograms. Nor do nodules larger than 5 mm without high-suspicion findings.
The guidelines also offer recommendations on treatment. There is no need to offer routine therapy with thyroid stimulating hormone (TSH) for benign nodules. “Though modest responses to therapy can be detected, the potential harm outweighs benefit for most patients,” the document states.
If benign nodules grow, surgery could be an option if they become large (greater than 4 cm), or cause compression or structural symptoms – or if there is clinical concern, although the document does not specify exactly what that could be.
Patients with growing nodules that are benign after a second biopsy should be regularly monitored. Most asymptomatic nodules demonstrating modest growth should be followed without intervention,” it says.
Recurrent benign cystic thyroid nodules may be treated surgically or with ethanol ablation, but asymptotic cystic nodules may be followed conservatively There are no data to guide the use of thyroid hormone treatment for benign nodules, even if they do grow.
The guidelines also offer recommendations for initial serum and molecular studies, and treatment for follicular and differentiated thyroid cancers.
Dr. Mandel had no financial disclosures.
EXPERT ANALYSIS FROM ITC 2015
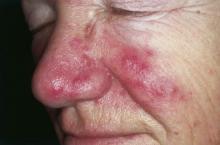
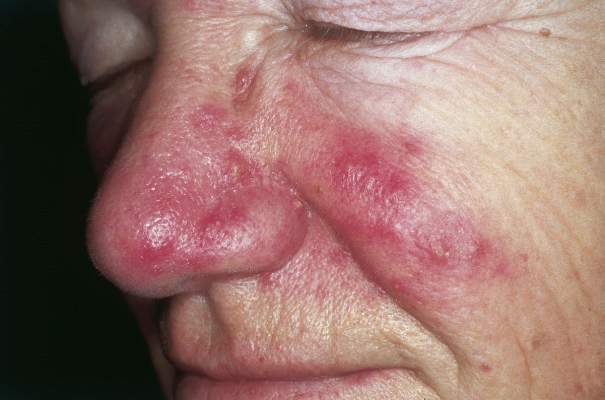
Inflammatory loop drives rosacea
NEW YORK – Rosacea seems to represent a Möbius strip of an overly enthusiastic immune response, with chemicals that recruit neutrophils, which, in turn, activate even more immunomodulators.
Researchers have been pointing the finger at trigger-happy neutrophils for more than a decade, Dr. David E. Cohen said at the summer meeting of the American Academy of Dermatology. But this group of cells is really more of an accomplice than a mastermind.
“Neutrophils were once the alpha and omega of rosacea,” said Dr. Cohen, the Charles C. and Dorothea E. Harris Professor of Dermatology, New York University. “But what are they doing on the skin and why are they there? They’re doing exactly what they’re supposed to do – defending against invasion by dumping out a whole poisonous soup of chemicals,” designed to protect and defend against microbes.
In the chronic disease of rosacea, however, these frontline soldiers are constantly recruited, even when there’s no invader to fight. They continue to carry out their preprogrammed attacks, releasing proteins that activate enzymes that stimulate immunomodulators that, in turn, call more neutrophils to the battlefield. Now, new research suggests that cathelicidins – antimicrobial peptides that are especially present in epithelial cells – are giving neutrophils their marching orders.
“Cathelicidins sit fully engaged on the skin and, the minute there’s a breach, they are turned on as the first defense,” Dr. Cohen said. “We need that kind of help right away – we can’t afford to wait around” for the second wave of defenders: neutrophils and lymphocytes.
This command seems to be bidirectional, though. Neutrophils release matrix metalloproteases (MMPs), enzymes critical for wound healing. MMPs remove dead extracellular matrix and microbes, prepare the wound bed for angiogenesis, and stimulate epidermal cell migration. But these benefits also exacerbate the things that make rosacea such a tough problem. By dissolving tissue, MMPs ramp up inflammation. By stimulating angiogenesis, they contribute to vasodilation, erythema, and telangiectasias.
And, Dr. Cohen noted, MMPs also just happen to recruit more neutrophils, which are capable of activating triptych enzymes, which turn on cathelicidins. “Now, a loop is being theorized,” he said. “The recruitment tools are kept on and this keeps cycling and cycling allowing the neutrophils to stay in the skin” for years.
The exaggerated presence of MMPs is likely the very reason that the tetracycline class of antibiotics can be so effective during rosacea exacerbations, Dr. Cohen said. The drugs aren’t necessarily fighting bacteria – instead, their anti-MMP properties function as a temporary break in this inflammatory loop. “They interfere with MMP, and you can’t turn on cathelicidin without MMP. If you lock MMP down, you can break the cycle because there is no neutrophil recruitment.”
Interestingly, he said, antibiotics that target MMP appear to suppress the enzymes at subantimicrobial doses. “This opens the possibility that we might be able to treat with these drugs and not exert antibiotic effects,” lessening the problems associated with indiscriminate antibiotic usage.
Inflammatory overdrive makes rosacea skin hypersensitive to any kind of inflammatory trigger, said Dr. Hilary Baldwin, medical director of the Acne Treatment and Research Center in Morristown, N.J. At the meeting, Dr. Baldwin discussed several new topical medications aimed at breaking the cycle:
• 1% Ivermectin: People with rosacea can harbor up to six times more demodex mites than can people without the disorder. The explanation behind this relationship is unclear: Is rosacea skin somehow more hospitable to the mite, or is an excess of mites a direct causative factor? Whatever the interaction, topical ivermectin breaks the link as both an antimicrobial and an anti-inflammatory agent. Ivermectin directly inhibits cellular and humoral immune response, and regulates tumor necrosis factor–alpha and interleukins 1-beta and 10.
Two randomized, double-blind vehicle-controlled studies evaluating 1% ivermectin cream in patients with papulopustular rosacea found that almost 80% of rosacea patients were clear or nearly clear on the Investigator’s Global Assessment (IGA) scale at 12 weeks – significantly more than those who were using a vehicle placebo. Inflammatory lesions significantly decreased and adverse events were similar to those associated with the vehicle (J Drugs Dermatol. 2014 Mar;13[3]:316-23). Another study found that ivermectin was significantly more effective than metronidazole, she said.
• Azelaic acid: A phase III study examined azelaic acid foam in almost 1,000 patients with moderate to severe papulopustular rosacea. IGA was clear or near-clear in 32% of the active group at 12 weeks, and 23.5% of the vehicle control group – a significant difference. There were significantly fewer inflammatory lesions among those in the active treatment group as well. Drug-related cutaneous adverse events were significantly more common among those treated with azelaic acid (7% vs. about 4%) and were mostly pain, pruritus, and dryness at application site (Cutis. 2015 Jul;96[1]:54-61).
• Oxymetazoline and brimonidine: These two alpha-adrenergic receptor agonists are under investigation for their ability to decrease erythema. Both cause a transient vasoconstriction. Two studies of patients with moderate or severe erythema of rosacea found that a topical brimonidine gel formulation significantly decreased erythema for up to 12 hours after application, compared with a vehicle gel (J Drugs Dermatol. 2013 Jun 1;12[6]:650-6). However, there have been several case reports of severe rebound, with redness not only returning, but returning at a more severe grade (J Am Acad Dermatol. 2014 Feb;70[2]:e37-8).
• Isotretinoin: New investigations are looking at topical retinoids for rosacea. They do appear effective – but not only for the reason they work in acne. While they decrease the size of sebaceous glands and the amount of sebum produced, they seem to exert their benefit through an anti-inflammatory pathway, as well. Isotretinoin significantly decreases toll-like receptor-2 expression on monocytes.
Isotretinoin has been shown to be more effective than doxycycline in clearing skin (J Dtsch Dermatol Ges. 2010 Jul;8[7]:505-15) but has also seemed helpful in preventing and slowing phyma development and improving ocular disease.
Both Dr. Cohen and Dr. Baldwin disclosed relationships with multiple pharmaceutical companies.
NEW YORK – Rosacea seems to represent a Möbius strip of an overly enthusiastic immune response, with chemicals that recruit neutrophils, which, in turn, activate even more immunomodulators.
Researchers have been pointing the finger at trigger-happy neutrophils for more than a decade, Dr. David E. Cohen said at the summer meeting of the American Academy of Dermatology. But this group of cells is really more of an accomplice than a mastermind.
“Neutrophils were once the alpha and omega of rosacea,” said Dr. Cohen, the Charles C. and Dorothea E. Harris Professor of Dermatology, New York University. “But what are they doing on the skin and why are they there? They’re doing exactly what they’re supposed to do – defending against invasion by dumping out a whole poisonous soup of chemicals,” designed to protect and defend against microbes.
In the chronic disease of rosacea, however, these frontline soldiers are constantly recruited, even when there’s no invader to fight. They continue to carry out their preprogrammed attacks, releasing proteins that activate enzymes that stimulate immunomodulators that, in turn, call more neutrophils to the battlefield. Now, new research suggests that cathelicidins – antimicrobial peptides that are especially present in epithelial cells – are giving neutrophils their marching orders.
“Cathelicidins sit fully engaged on the skin and, the minute there’s a breach, they are turned on as the first defense,” Dr. Cohen said. “We need that kind of help right away – we can’t afford to wait around” for the second wave of defenders: neutrophils and lymphocytes.
This command seems to be bidirectional, though. Neutrophils release matrix metalloproteases (MMPs), enzymes critical for wound healing. MMPs remove dead extracellular matrix and microbes, prepare the wound bed for angiogenesis, and stimulate epidermal cell migration. But these benefits also exacerbate the things that make rosacea such a tough problem. By dissolving tissue, MMPs ramp up inflammation. By stimulating angiogenesis, they contribute to vasodilation, erythema, and telangiectasias.
And, Dr. Cohen noted, MMPs also just happen to recruit more neutrophils, which are capable of activating triptych enzymes, which turn on cathelicidins. “Now, a loop is being theorized,” he said. “The recruitment tools are kept on and this keeps cycling and cycling allowing the neutrophils to stay in the skin” for years.
The exaggerated presence of MMPs is likely the very reason that the tetracycline class of antibiotics can be so effective during rosacea exacerbations, Dr. Cohen said. The drugs aren’t necessarily fighting bacteria – instead, their anti-MMP properties function as a temporary break in this inflammatory loop. “They interfere with MMP, and you can’t turn on cathelicidin without MMP. If you lock MMP down, you can break the cycle because there is no neutrophil recruitment.”
Interestingly, he said, antibiotics that target MMP appear to suppress the enzymes at subantimicrobial doses. “This opens the possibility that we might be able to treat with these drugs and not exert antibiotic effects,” lessening the problems associated with indiscriminate antibiotic usage.
Inflammatory overdrive makes rosacea skin hypersensitive to any kind of inflammatory trigger, said Dr. Hilary Baldwin, medical director of the Acne Treatment and Research Center in Morristown, N.J. At the meeting, Dr. Baldwin discussed several new topical medications aimed at breaking the cycle:
• 1% Ivermectin: People with rosacea can harbor up to six times more demodex mites than can people without the disorder. The explanation behind this relationship is unclear: Is rosacea skin somehow more hospitable to the mite, or is an excess of mites a direct causative factor? Whatever the interaction, topical ivermectin breaks the link as both an antimicrobial and an anti-inflammatory agent. Ivermectin directly inhibits cellular and humoral immune response, and regulates tumor necrosis factor–alpha and interleukins 1-beta and 10.
Two randomized, double-blind vehicle-controlled studies evaluating 1% ivermectin cream in patients with papulopustular rosacea found that almost 80% of rosacea patients were clear or nearly clear on the Investigator’s Global Assessment (IGA) scale at 12 weeks – significantly more than those who were using a vehicle placebo. Inflammatory lesions significantly decreased and adverse events were similar to those associated with the vehicle (J Drugs Dermatol. 2014 Mar;13[3]:316-23). Another study found that ivermectin was significantly more effective than metronidazole, she said.
• Azelaic acid: A phase III study examined azelaic acid foam in almost 1,000 patients with moderate to severe papulopustular rosacea. IGA was clear or near-clear in 32% of the active group at 12 weeks, and 23.5% of the vehicle control group – a significant difference. There were significantly fewer inflammatory lesions among those in the active treatment group as well. Drug-related cutaneous adverse events were significantly more common among those treated with azelaic acid (7% vs. about 4%) and were mostly pain, pruritus, and dryness at application site (Cutis. 2015 Jul;96[1]:54-61).
• Oxymetazoline and brimonidine: These two alpha-adrenergic receptor agonists are under investigation for their ability to decrease erythema. Both cause a transient vasoconstriction. Two studies of patients with moderate or severe erythema of rosacea found that a topical brimonidine gel formulation significantly decreased erythema for up to 12 hours after application, compared with a vehicle gel (J Drugs Dermatol. 2013 Jun 1;12[6]:650-6). However, there have been several case reports of severe rebound, with redness not only returning, but returning at a more severe grade (J Am Acad Dermatol. 2014 Feb;70[2]:e37-8).
• Isotretinoin: New investigations are looking at topical retinoids for rosacea. They do appear effective – but not only for the reason they work in acne. While they decrease the size of sebaceous glands and the amount of sebum produced, they seem to exert their benefit through an anti-inflammatory pathway, as well. Isotretinoin significantly decreases toll-like receptor-2 expression on monocytes.
Isotretinoin has been shown to be more effective than doxycycline in clearing skin (J Dtsch Dermatol Ges. 2010 Jul;8[7]:505-15) but has also seemed helpful in preventing and slowing phyma development and improving ocular disease.
Both Dr. Cohen and Dr. Baldwin disclosed relationships with multiple pharmaceutical companies.
NEW YORK – Rosacea seems to represent a Möbius strip of an overly enthusiastic immune response, with chemicals that recruit neutrophils, which, in turn, activate even more immunomodulators.
Researchers have been pointing the finger at trigger-happy neutrophils for more than a decade, Dr. David E. Cohen said at the summer meeting of the American Academy of Dermatology. But this group of cells is really more of an accomplice than a mastermind.
“Neutrophils were once the alpha and omega of rosacea,” said Dr. Cohen, the Charles C. and Dorothea E. Harris Professor of Dermatology, New York University. “But what are they doing on the skin and why are they there? They’re doing exactly what they’re supposed to do – defending against invasion by dumping out a whole poisonous soup of chemicals,” designed to protect and defend against microbes.
In the chronic disease of rosacea, however, these frontline soldiers are constantly recruited, even when there’s no invader to fight. They continue to carry out their preprogrammed attacks, releasing proteins that activate enzymes that stimulate immunomodulators that, in turn, call more neutrophils to the battlefield. Now, new research suggests that cathelicidins – antimicrobial peptides that are especially present in epithelial cells – are giving neutrophils their marching orders.
“Cathelicidins sit fully engaged on the skin and, the minute there’s a breach, they are turned on as the first defense,” Dr. Cohen said. “We need that kind of help right away – we can’t afford to wait around” for the second wave of defenders: neutrophils and lymphocytes.
This command seems to be bidirectional, though. Neutrophils release matrix metalloproteases (MMPs), enzymes critical for wound healing. MMPs remove dead extracellular matrix and microbes, prepare the wound bed for angiogenesis, and stimulate epidermal cell migration. But these benefits also exacerbate the things that make rosacea such a tough problem. By dissolving tissue, MMPs ramp up inflammation. By stimulating angiogenesis, they contribute to vasodilation, erythema, and telangiectasias.
And, Dr. Cohen noted, MMPs also just happen to recruit more neutrophils, which are capable of activating triptych enzymes, which turn on cathelicidins. “Now, a loop is being theorized,” he said. “The recruitment tools are kept on and this keeps cycling and cycling allowing the neutrophils to stay in the skin” for years.
The exaggerated presence of MMPs is likely the very reason that the tetracycline class of antibiotics can be so effective during rosacea exacerbations, Dr. Cohen said. The drugs aren’t necessarily fighting bacteria – instead, their anti-MMP properties function as a temporary break in this inflammatory loop. “They interfere with MMP, and you can’t turn on cathelicidin without MMP. If you lock MMP down, you can break the cycle because there is no neutrophil recruitment.”
Interestingly, he said, antibiotics that target MMP appear to suppress the enzymes at subantimicrobial doses. “This opens the possibility that we might be able to treat with these drugs and not exert antibiotic effects,” lessening the problems associated with indiscriminate antibiotic usage.
Inflammatory overdrive makes rosacea skin hypersensitive to any kind of inflammatory trigger, said Dr. Hilary Baldwin, medical director of the Acne Treatment and Research Center in Morristown, N.J. At the meeting, Dr. Baldwin discussed several new topical medications aimed at breaking the cycle:
• 1% Ivermectin: People with rosacea can harbor up to six times more demodex mites than can people without the disorder. The explanation behind this relationship is unclear: Is rosacea skin somehow more hospitable to the mite, or is an excess of mites a direct causative factor? Whatever the interaction, topical ivermectin breaks the link as both an antimicrobial and an anti-inflammatory agent. Ivermectin directly inhibits cellular and humoral immune response, and regulates tumor necrosis factor–alpha and interleukins 1-beta and 10.
Two randomized, double-blind vehicle-controlled studies evaluating 1% ivermectin cream in patients with papulopustular rosacea found that almost 80% of rosacea patients were clear or nearly clear on the Investigator’s Global Assessment (IGA) scale at 12 weeks – significantly more than those who were using a vehicle placebo. Inflammatory lesions significantly decreased and adverse events were similar to those associated with the vehicle (J Drugs Dermatol. 2014 Mar;13[3]:316-23). Another study found that ivermectin was significantly more effective than metronidazole, she said.
• Azelaic acid: A phase III study examined azelaic acid foam in almost 1,000 patients with moderate to severe papulopustular rosacea. IGA was clear or near-clear in 32% of the active group at 12 weeks, and 23.5% of the vehicle control group – a significant difference. There were significantly fewer inflammatory lesions among those in the active treatment group as well. Drug-related cutaneous adverse events were significantly more common among those treated with azelaic acid (7% vs. about 4%) and were mostly pain, pruritus, and dryness at application site (Cutis. 2015 Jul;96[1]:54-61).
• Oxymetazoline and brimonidine: These two alpha-adrenergic receptor agonists are under investigation for their ability to decrease erythema. Both cause a transient vasoconstriction. Two studies of patients with moderate or severe erythema of rosacea found that a topical brimonidine gel formulation significantly decreased erythema for up to 12 hours after application, compared with a vehicle gel (J Drugs Dermatol. 2013 Jun 1;12[6]:650-6). However, there have been several case reports of severe rebound, with redness not only returning, but returning at a more severe grade (J Am Acad Dermatol. 2014 Feb;70[2]:e37-8).
• Isotretinoin: New investigations are looking at topical retinoids for rosacea. They do appear effective – but not only for the reason they work in acne. While they decrease the size of sebaceous glands and the amount of sebum produced, they seem to exert their benefit through an anti-inflammatory pathway, as well. Isotretinoin significantly decreases toll-like receptor-2 expression on monocytes.
Isotretinoin has been shown to be more effective than doxycycline in clearing skin (J Dtsch Dermatol Ges. 2010 Jul;8[7]:505-15) but has also seemed helpful in preventing and slowing phyma development and improving ocular disease.
Both Dr. Cohen and Dr. Baldwin disclosed relationships with multiple pharmaceutical companies.
EXPERT ANALYSIS FROM THE AAD SUMMER ACADEMY
High free thyroxine may increase risk of sudden cardiac death
LAKE BUENA VISTA, FLA. – High levels of free thyroxine (T4) can double the risk of sudden cardiac death, a large cohort study has determined.
Even people with free T4 in the upper range of normal may face an increased risk – up to 4% over 10 years, Dr. Layal Chaker said at the International Thyroid Conference.
The association remained significant, even when researchers controlled for independent cardiovascular risk factors, including hypertension and hyperlipidemia, said Dr. Chaker of Erasmus Medical Center, Rotterdam, the Netherlands.
The findings were based on a 9-year analysis of about 10,000 people included in the Rotterdam Elderly Study, conducted from 1990 to 2008. That study followed 15,000 people from middle age to old age, assessing cardiovascular and neurological diseases and their relationship to aging.
Dr. Chaker’s study comprised a subset of those who were without frank cardiovascular disease at baseline. She defined sudden cardiac death as a natural death from cardiac causes, heralded by an abrupt loss of consciousness within an hour of the onset of acute symptoms. Unwitnessed deaths were also included in the analysis. All of the death records were reviewed by two clinicians and a senior cardiologist.
The cohort comprised 10,318 subjects who had measurements of thyroid stimulating hormone (TSH) and free thyroxine (free T4); the mean age was 65 years.
Dr. Chaker stratified the group into tertiles based on the levels of these biomarkers. She conducted a multivariate analysis that controlled for age, sex, pulse, hypertension, cholesterol levels, diabetes, body mass index, smoking, and QT interval.
There were 261 sudden cardiac deaths by the end of follow-up.
There was no significant relationship between any level of TSH and sudden cardiac death. However, when she assessed the deaths by tertiles of free T4, she found a significant 40% increase in the risk among those whose levels ranged from 1.29 to 4 ng/L. The absolute 10-year risk rose from 1% at the lowest tertile to 7% in the highest.
Dr. Chaker then included only patients whose free T4 levels were in the euthyroid range of 0.85-1.95 ng/L. Among these, the risk of sudden cardiac death increased as free T4 increased (hazard ratio [HR], 2.25 for the highest level). The absolute 10-year risk rose from 1% at the lowest euthyroid level to 4% at 1.95 ng/L.
The reason for this finding isn’t completely clear, although other studies have shown a relationship between cardiac problems and thyroid function, she said.
“There may be some hemodynamic abnormalities that go along with even subclinical hyperthyroidism. High free T4 also has been associated with atrial fibrillation; both subclinical hyper- and hypothyroidism are associated with a prolongation of the QT interval.”
The meeting was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society. Dr. Chaker had no financial disclosures.
On Twitter @Alz_Gal
LAKE BUENA VISTA, FLA. – High levels of free thyroxine (T4) can double the risk of sudden cardiac death, a large cohort study has determined.
Even people with free T4 in the upper range of normal may face an increased risk – up to 4% over 10 years, Dr. Layal Chaker said at the International Thyroid Conference.
The association remained significant, even when researchers controlled for independent cardiovascular risk factors, including hypertension and hyperlipidemia, said Dr. Chaker of Erasmus Medical Center, Rotterdam, the Netherlands.
The findings were based on a 9-year analysis of about 10,000 people included in the Rotterdam Elderly Study, conducted from 1990 to 2008. That study followed 15,000 people from middle age to old age, assessing cardiovascular and neurological diseases and their relationship to aging.
Dr. Chaker’s study comprised a subset of those who were without frank cardiovascular disease at baseline. She defined sudden cardiac death as a natural death from cardiac causes, heralded by an abrupt loss of consciousness within an hour of the onset of acute symptoms. Unwitnessed deaths were also included in the analysis. All of the death records were reviewed by two clinicians and a senior cardiologist.
The cohort comprised 10,318 subjects who had measurements of thyroid stimulating hormone (TSH) and free thyroxine (free T4); the mean age was 65 years.
Dr. Chaker stratified the group into tertiles based on the levels of these biomarkers. She conducted a multivariate analysis that controlled for age, sex, pulse, hypertension, cholesterol levels, diabetes, body mass index, smoking, and QT interval.
There were 261 sudden cardiac deaths by the end of follow-up.
There was no significant relationship between any level of TSH and sudden cardiac death. However, when she assessed the deaths by tertiles of free T4, she found a significant 40% increase in the risk among those whose levels ranged from 1.29 to 4 ng/L. The absolute 10-year risk rose from 1% at the lowest tertile to 7% in the highest.
Dr. Chaker then included only patients whose free T4 levels were in the euthyroid range of 0.85-1.95 ng/L. Among these, the risk of sudden cardiac death increased as free T4 increased (hazard ratio [HR], 2.25 for the highest level). The absolute 10-year risk rose from 1% at the lowest euthyroid level to 4% at 1.95 ng/L.
The reason for this finding isn’t completely clear, although other studies have shown a relationship between cardiac problems and thyroid function, she said.
“There may be some hemodynamic abnormalities that go along with even subclinical hyperthyroidism. High free T4 also has been associated with atrial fibrillation; both subclinical hyper- and hypothyroidism are associated with a prolongation of the QT interval.”
The meeting was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society. Dr. Chaker had no financial disclosures.
On Twitter @Alz_Gal
LAKE BUENA VISTA, FLA. – High levels of free thyroxine (T4) can double the risk of sudden cardiac death, a large cohort study has determined.
Even people with free T4 in the upper range of normal may face an increased risk – up to 4% over 10 years, Dr. Layal Chaker said at the International Thyroid Conference.
The association remained significant, even when researchers controlled for independent cardiovascular risk factors, including hypertension and hyperlipidemia, said Dr. Chaker of Erasmus Medical Center, Rotterdam, the Netherlands.
The findings were based on a 9-year analysis of about 10,000 people included in the Rotterdam Elderly Study, conducted from 1990 to 2008. That study followed 15,000 people from middle age to old age, assessing cardiovascular and neurological diseases and their relationship to aging.
Dr. Chaker’s study comprised a subset of those who were without frank cardiovascular disease at baseline. She defined sudden cardiac death as a natural death from cardiac causes, heralded by an abrupt loss of consciousness within an hour of the onset of acute symptoms. Unwitnessed deaths were also included in the analysis. All of the death records were reviewed by two clinicians and a senior cardiologist.
The cohort comprised 10,318 subjects who had measurements of thyroid stimulating hormone (TSH) and free thyroxine (free T4); the mean age was 65 years.
Dr. Chaker stratified the group into tertiles based on the levels of these biomarkers. She conducted a multivariate analysis that controlled for age, sex, pulse, hypertension, cholesterol levels, diabetes, body mass index, smoking, and QT interval.
There were 261 sudden cardiac deaths by the end of follow-up.
There was no significant relationship between any level of TSH and sudden cardiac death. However, when she assessed the deaths by tertiles of free T4, she found a significant 40% increase in the risk among those whose levels ranged from 1.29 to 4 ng/L. The absolute 10-year risk rose from 1% at the lowest tertile to 7% in the highest.
Dr. Chaker then included only patients whose free T4 levels were in the euthyroid range of 0.85-1.95 ng/L. Among these, the risk of sudden cardiac death increased as free T4 increased (hazard ratio [HR], 2.25 for the highest level). The absolute 10-year risk rose from 1% at the lowest euthyroid level to 4% at 1.95 ng/L.
The reason for this finding isn’t completely clear, although other studies have shown a relationship between cardiac problems and thyroid function, she said.
“There may be some hemodynamic abnormalities that go along with even subclinical hyperthyroidism. High free T4 also has been associated with atrial fibrillation; both subclinical hyper- and hypothyroidism are associated with a prolongation of the QT interval.”
The meeting was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society. Dr. Chaker had no financial disclosures.
On Twitter @Alz_Gal
AT ITC 2015
Key clinical point: Even subclinical hyperthyroidism may increase the risk of sudden cardiac death.
Major finding: High free thyroxine in euthyroid patients doubled the risk of sudden cardiac death.
Data source: A longitudinal cohort study comprising 10,318 subjects.
Disclosures: Dr. Chaker had no financial disclosures.
ITC: Study provides first evidence of paclitaxel benefit for anaplastic thyroid cancer
LAKE BUENA VISTA, FLA. – Weekly infusions of paclitaxel delayed progression in some patients with the very aggressive anaplastic thyroid cancer, a small prospective study determined.
The drug was most effective as adjuvant therapy for patients who had already undergone chemotherapy plus resection of the primary tumor, Dr. Naoyoshi Onoda reported in a poster session at the International Thyroid Conference. They survived for a median of 1 year (112-788 days) – an impressive feat considering that most patients with anaplastic thyroid cancer die within 6 months of diagnosis.
This finding suggests a place for paclitaxel as a standardized therapy for such patients, said Dr. Onoda of Osaka (Japan) City University. “We have objective data supporting standardized chemotherapy for the first time in the world.”
Anaplastic thyroid cancer is a very rare – but very aggressive – disease; there is no standardized treatment option. Dr. Onoda and his colleagues conducted a national prospective open-label study of weekly paclitaxel infusions in 56 patients with the malignancy.
The cohort was a median of 71 years old. All had stage IV disease: 10 were grade A, 18 grade B, 24 grade C, and four grade X. They received 80 mg/m2 infusions once a week. The median number of cycles was 2, although it ranged from 0-23 cycles.
Almost everyone (98%) experienced adverse events; the most common was anemia (77%). About a quarter (28%) experienced adverse events of at least grade 3, but there were no serious events and no deaths related to the study drug.
The objective response rate and the clinical benefit rate were 23% and 79%. The agent was not curative; at the last follow-up, no patient had achieved a complete response, and 43 of 56 in the study had died of their disease. “Overall, the median time to progression was only 47 days, and median overall survival just 227 days. That is so very short. But it’s a little bit longer than we had been seeing rates from reported cases,” he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
The study was sponsored by the Prospective Clinical Study Committee of the Anaplastic Thyroid Carcinoma Research Consortium of Japan (ATCCJ). Dr. Onoda had no financial disclosures.
LAKE BUENA VISTA, FLA. – Weekly infusions of paclitaxel delayed progression in some patients with the very aggressive anaplastic thyroid cancer, a small prospective study determined.
The drug was most effective as adjuvant therapy for patients who had already undergone chemotherapy plus resection of the primary tumor, Dr. Naoyoshi Onoda reported in a poster session at the International Thyroid Conference. They survived for a median of 1 year (112-788 days) – an impressive feat considering that most patients with anaplastic thyroid cancer die within 6 months of diagnosis.
This finding suggests a place for paclitaxel as a standardized therapy for such patients, said Dr. Onoda of Osaka (Japan) City University. “We have objective data supporting standardized chemotherapy for the first time in the world.”
Anaplastic thyroid cancer is a very rare – but very aggressive – disease; there is no standardized treatment option. Dr. Onoda and his colleagues conducted a national prospective open-label study of weekly paclitaxel infusions in 56 patients with the malignancy.
The cohort was a median of 71 years old. All had stage IV disease: 10 were grade A, 18 grade B, 24 grade C, and four grade X. They received 80 mg/m2 infusions once a week. The median number of cycles was 2, although it ranged from 0-23 cycles.
Almost everyone (98%) experienced adverse events; the most common was anemia (77%). About a quarter (28%) experienced adverse events of at least grade 3, but there were no serious events and no deaths related to the study drug.
The objective response rate and the clinical benefit rate were 23% and 79%. The agent was not curative; at the last follow-up, no patient had achieved a complete response, and 43 of 56 in the study had died of their disease. “Overall, the median time to progression was only 47 days, and median overall survival just 227 days. That is so very short. But it’s a little bit longer than we had been seeing rates from reported cases,” he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
The study was sponsored by the Prospective Clinical Study Committee of the Anaplastic Thyroid Carcinoma Research Consortium of Japan (ATCCJ). Dr. Onoda had no financial disclosures.
LAKE BUENA VISTA, FLA. – Weekly infusions of paclitaxel delayed progression in some patients with the very aggressive anaplastic thyroid cancer, a small prospective study determined.
The drug was most effective as adjuvant therapy for patients who had already undergone chemotherapy plus resection of the primary tumor, Dr. Naoyoshi Onoda reported in a poster session at the International Thyroid Conference. They survived for a median of 1 year (112-788 days) – an impressive feat considering that most patients with anaplastic thyroid cancer die within 6 months of diagnosis.
This finding suggests a place for paclitaxel as a standardized therapy for such patients, said Dr. Onoda of Osaka (Japan) City University. “We have objective data supporting standardized chemotherapy for the first time in the world.”
Anaplastic thyroid cancer is a very rare – but very aggressive – disease; there is no standardized treatment option. Dr. Onoda and his colleagues conducted a national prospective open-label study of weekly paclitaxel infusions in 56 patients with the malignancy.
The cohort was a median of 71 years old. All had stage IV disease: 10 were grade A, 18 grade B, 24 grade C, and four grade X. They received 80 mg/m2 infusions once a week. The median number of cycles was 2, although it ranged from 0-23 cycles.
Almost everyone (98%) experienced adverse events; the most common was anemia (77%). About a quarter (28%) experienced adverse events of at least grade 3, but there were no serious events and no deaths related to the study drug.
The objective response rate and the clinical benefit rate were 23% and 79%. The agent was not curative; at the last follow-up, no patient had achieved a complete response, and 43 of 56 in the study had died of their disease. “Overall, the median time to progression was only 47 days, and median overall survival just 227 days. That is so very short. But it’s a little bit longer than we had been seeing rates from reported cases,” he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
The study was sponsored by the Prospective Clinical Study Committee of the Anaplastic Thyroid Carcinoma Research Consortium of Japan (ATCCJ). Dr. Onoda had no financial disclosures.
AT ITC 2015
Key clinical point: Paclitaxel may have use as a standardized treatment in anaplastic thyroid cancer.
Major finding: Weekly paclitaxel extended survival to a median of 1 year as adjuvant therapy for patients with resected anaplastic thyroid cancer.
Data source: The prospective open-label study involved 56 patients.
Disclosures: The study was sponsored by the Prospective Clinical Study Committee of the Anaplastic Thyroid Carcinoma Research Consortium of Japan (ATCCJ). Dr. Onoda had no financial disclosures.
ITC: Levothyroxine may improve obstetric outcomes in women with low thyroid function
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
LAKE BUENA VISTA, FLA. – Normalizing maternal thyroid function early in pregnancy may improve obstetric outcomes for women with subclinical hypothyroidism.
A high level of thyroid-stimulating hormone (TSH) was associated with increased stillbirth in women who didn’t receive prenatal levothyroxine, Peter N. Taylor, Ph.D., reported at the International Thyroid Congress.
And women who were untreated for low free thyroxine (T4) were significantly more likely to need an early cesarean section than treated women, suggesting that levothyroxine could be reducing conditions leading to maternal-fetal distress.
“The good thing is that many physicians are already treating with levothyroxine anyway,” said Dr. Taylor of Cardiff (Wales) University. “There are no obvious adverse obstetric events associated with it. It seems relatively benign, and the accepted dose of 150 mcg per day seems adequate.”
He reported data from a subgroup analysis of the large Controlled Antenatal Thyroid Screening Study (N Eng J Med. 2012;366:493-501). That trial enrolled more than 21,000 mother-child pairs, and examined the effect of maternal levothyroxine treatment on the child’s IQ at 3 years. Among the children of women with hypothyroidism, the mean IQ scores were 99.2 and 100 in the treated and control groups – not significantly different. The proportions of children with an IQ of less than 85 were 12% in the treated group and 14% in the control group, also not significantly different.
Dr. Taylor’s cohort involved about 14,000 mother-child pairs from this study, all of whom were from Wales. Using national health care databases as well as study data, he was able to examine obstetric outcomes in women with low thyroid function (664) who received or did not receive levothyroxine early between 11 and 16 weeks’ gestation.
About half of the hypothyroid women (351) had TSH levels above 3.5 mU/L. The remainder had free T4 levels below 10.9 pmol/L. Each of these groups was randomized to levothyroxine treatment or placebo. The rest of the cohort had normal thyroid status.
The study’s primary endpoint was the rate of stillbirth. Secondary endpoints were cesarean section rate (both overall and early); gestational age at birth; and macrosomia.
Among the euthyroid group, the rate of stillbirth was 0.34%, similar to the national background rate. There were no stillbirths among women with high TSH who were treated. Three (1.68%) occurred in the untreated group. The TSH levels in those women before treatment were 3.63 mU/L, 3.66 mU/L, and 4.58 mU/L – not dramatically high, Dr. Taylor noted. “But they could have risen later in pregnancy as stress on the thyroid increased.”
After adjusting for maternal age, weight, parity, birth year, and smoking, stillbirths were five times more likely among the untreated women than the treated women. However, Dr. Taylor cautioned, “This is a very small number of events. But it is quite seductive to think that stillbirths could be prevented by levothyroxine.”
There were no significant associations of high TSH with macrosomia or gestational age.
Untreated low free T4 was not associated with stillbirth. However, Dr. Taylor said, it was very strongly associated with early C-section.
The overall C-section rate was similar between untreated and treated women (28%). But 5.6% of the untreated women had an early C-section, compared with none of the treated women. This hints strongly at a protective effect of levothyroxine, Dr. Taylor said. Early C-sections – between 26 and 32 weeks – are medically driven rather than driven by patient choice. This finding of fewer early interventions among treated women suggests that levothyroxine is exerting some protective effect, especially given the finding that infants of untreated mothers actually tended to be about 133 g lighter at birth.
“We would speculate this is probably due to a decrease in preeclampsia and gestational hypertension, which are more common among women with hypothyroidism.”
Infants of untreated mothers also were born slightly earlier – about half a week, he said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
Dr. Taylor had no financial disclosures.
AT ITC 2015
Key clinical point: Levothyroxine may improve obstetric outcomes in women with low thyroid function.
Major finding: Stillbirths were five times more likely among women who were untreated for high levels of thyroid-stimulating hormone during pregnancy.
Data source: The analysis comprised about 14,000 mother-child pairs.
Disclosures: Dr. Taylor had no financial disclosures.
Ipilimumab approved as adjuvant treatment for resected metastatic melanoma
Ipilimumab is now approved as an adjuvant treatment for patients who have undergone a complete resection of metastatic cutaneous melanoma.
The approval is based on data from a large study showing that ipilimumab, marketed as Yervoy by Bristol-Myers Squibb, improved recurrence-free survival, compared with placebo.
“Today’s approval of Yervoy extends its use to patients who are at high risk of developing recurrence of melanoma after surgery,” Dr. Richard Pazdur, director of the office of hematology and oncology products in Food and Drug Administration’s Center for Drug Evaluation and Research, said in the FDA statement announcing the approval on Oct. 28. “This new use of the drug in earlier stages of the disease builds on our understanding of the immune system’s interaction with cancer.”
Ipilimumab, a monoclonal antibody that blocks cytotoxic T-lymphocyte antigen 4 (CTLA-4), was initially approved in 2011 for the treatment of unresectable or metastatic melanoma.
The new indication is for the adjuvant treatment of patients “with cutaneous melanoma with pathologic involvement of regional lymph nodes of more than 1 mm who have undergone complete resection, including total lymphadenectomy.”
In the study of 951 patients, with resected Stage IIIA, IIIB, and IIIC cutaneous melanoma, the median recurrence-free survival was 26 months in the ipilimumab group vs. 17 months in the placebo group; 3-year recurrence-free survival was 46.5% vs. 34.8% (Lancet Oncol. 2015; 16[5]:522-30). The data on overall survival have not yet been analyzed.
The recommended dose and schedule for ipilimumab for adjuvant treatment is 10 mg/kg administered intravenously over 90 minutes every 3 weeks for four doses, followed by 10 mg/kg every 12 weeks for up to 3 years. Doses are omitted, not delayed, if toxicity occurs.
In the study, 52% of those treated with ipilimumab had to discontinue the drug because of adverse reactions. In addition, 41% experienced grade 3-5 immune mediated adverse reactions, including enterocolitis (16%), hepatitis (11%), endocrinopathy (8%), dermatitis (4%), and neuropathy (1.7%).
There were five treatment-related deaths in the study, from immune-mediated adverse reactions, which were enterocolitis (three), Guillain-Barré syndrome (one) and myocarditis (one).The prescribing information includes a boxed warning about the risk of immune-mediated adverse reactions.
In a statement, FDA officials cautioned physicians to carefully monitor patients taking the drug for signs of enterocolitis, dermatitis, neuropathy, and endocrinopathy. Liver function, adrenocorticotropic hormone level, and thyroid function should be measured at baseline and before each dose.
Among the most common reactions were rash, pruritus, diarrhea, nausea, colitis, vomiting, weight loss, fatigue, pyrexia, headache, decreased appetite, and insomnia.
CTLA-4 “may play a role in slowing down or turning off the body’s immune system, and affects its ability to fight off cancerous cells. Yervoy may work by allowing the body’s immune system to recognize, target and attack cells in melanoma tumors,” according to the FDA statement.
Serious adverse events associated with ipilimumab should be reported to the FDA’s MedWatch program or at 800-332-1088.
Ipilimumab is now approved as an adjuvant treatment for patients who have undergone a complete resection of metastatic cutaneous melanoma.
The approval is based on data from a large study showing that ipilimumab, marketed as Yervoy by Bristol-Myers Squibb, improved recurrence-free survival, compared with placebo.
“Today’s approval of Yervoy extends its use to patients who are at high risk of developing recurrence of melanoma after surgery,” Dr. Richard Pazdur, director of the office of hematology and oncology products in Food and Drug Administration’s Center for Drug Evaluation and Research, said in the FDA statement announcing the approval on Oct. 28. “This new use of the drug in earlier stages of the disease builds on our understanding of the immune system’s interaction with cancer.”
Ipilimumab, a monoclonal antibody that blocks cytotoxic T-lymphocyte antigen 4 (CTLA-4), was initially approved in 2011 for the treatment of unresectable or metastatic melanoma.
The new indication is for the adjuvant treatment of patients “with cutaneous melanoma with pathologic involvement of regional lymph nodes of more than 1 mm who have undergone complete resection, including total lymphadenectomy.”
In the study of 951 patients, with resected Stage IIIA, IIIB, and IIIC cutaneous melanoma, the median recurrence-free survival was 26 months in the ipilimumab group vs. 17 months in the placebo group; 3-year recurrence-free survival was 46.5% vs. 34.8% (Lancet Oncol. 2015; 16[5]:522-30). The data on overall survival have not yet been analyzed.
The recommended dose and schedule for ipilimumab for adjuvant treatment is 10 mg/kg administered intravenously over 90 minutes every 3 weeks for four doses, followed by 10 mg/kg every 12 weeks for up to 3 years. Doses are omitted, not delayed, if toxicity occurs.
In the study, 52% of those treated with ipilimumab had to discontinue the drug because of adverse reactions. In addition, 41% experienced grade 3-5 immune mediated adverse reactions, including enterocolitis (16%), hepatitis (11%), endocrinopathy (8%), dermatitis (4%), and neuropathy (1.7%).
There were five treatment-related deaths in the study, from immune-mediated adverse reactions, which were enterocolitis (three), Guillain-Barré syndrome (one) and myocarditis (one).The prescribing information includes a boxed warning about the risk of immune-mediated adverse reactions.
In a statement, FDA officials cautioned physicians to carefully monitor patients taking the drug for signs of enterocolitis, dermatitis, neuropathy, and endocrinopathy. Liver function, adrenocorticotropic hormone level, and thyroid function should be measured at baseline and before each dose.
Among the most common reactions were rash, pruritus, diarrhea, nausea, colitis, vomiting, weight loss, fatigue, pyrexia, headache, decreased appetite, and insomnia.
CTLA-4 “may play a role in slowing down or turning off the body’s immune system, and affects its ability to fight off cancerous cells. Yervoy may work by allowing the body’s immune system to recognize, target and attack cells in melanoma tumors,” according to the FDA statement.
Serious adverse events associated with ipilimumab should be reported to the FDA’s MedWatch program or at 800-332-1088.
Ipilimumab is now approved as an adjuvant treatment for patients who have undergone a complete resection of metastatic cutaneous melanoma.
The approval is based on data from a large study showing that ipilimumab, marketed as Yervoy by Bristol-Myers Squibb, improved recurrence-free survival, compared with placebo.
“Today’s approval of Yervoy extends its use to patients who are at high risk of developing recurrence of melanoma after surgery,” Dr. Richard Pazdur, director of the office of hematology and oncology products in Food and Drug Administration’s Center for Drug Evaluation and Research, said in the FDA statement announcing the approval on Oct. 28. “This new use of the drug in earlier stages of the disease builds on our understanding of the immune system’s interaction with cancer.”
Ipilimumab, a monoclonal antibody that blocks cytotoxic T-lymphocyte antigen 4 (CTLA-4), was initially approved in 2011 for the treatment of unresectable or metastatic melanoma.
The new indication is for the adjuvant treatment of patients “with cutaneous melanoma with pathologic involvement of regional lymph nodes of more than 1 mm who have undergone complete resection, including total lymphadenectomy.”
In the study of 951 patients, with resected Stage IIIA, IIIB, and IIIC cutaneous melanoma, the median recurrence-free survival was 26 months in the ipilimumab group vs. 17 months in the placebo group; 3-year recurrence-free survival was 46.5% vs. 34.8% (Lancet Oncol. 2015; 16[5]:522-30). The data on overall survival have not yet been analyzed.
The recommended dose and schedule for ipilimumab for adjuvant treatment is 10 mg/kg administered intravenously over 90 minutes every 3 weeks for four doses, followed by 10 mg/kg every 12 weeks for up to 3 years. Doses are omitted, not delayed, if toxicity occurs.
In the study, 52% of those treated with ipilimumab had to discontinue the drug because of adverse reactions. In addition, 41% experienced grade 3-5 immune mediated adverse reactions, including enterocolitis (16%), hepatitis (11%), endocrinopathy (8%), dermatitis (4%), and neuropathy (1.7%).
There were five treatment-related deaths in the study, from immune-mediated adverse reactions, which were enterocolitis (three), Guillain-Barré syndrome (one) and myocarditis (one).The prescribing information includes a boxed warning about the risk of immune-mediated adverse reactions.
In a statement, FDA officials cautioned physicians to carefully monitor patients taking the drug for signs of enterocolitis, dermatitis, neuropathy, and endocrinopathy. Liver function, adrenocorticotropic hormone level, and thyroid function should be measured at baseline and before each dose.
Among the most common reactions were rash, pruritus, diarrhea, nausea, colitis, vomiting, weight loss, fatigue, pyrexia, headache, decreased appetite, and insomnia.
CTLA-4 “may play a role in slowing down or turning off the body’s immune system, and affects its ability to fight off cancerous cells. Yervoy may work by allowing the body’s immune system to recognize, target and attack cells in melanoma tumors,” according to the FDA statement.
Serious adverse events associated with ipilimumab should be reported to the FDA’s MedWatch program or at 800-332-1088.
Trial to prove utility of amyloid imaging seeks physicians for referrals
A major study aimed at proving the clinical utility of amyloid imaging in diagnosing dementia is now recruiting participating physicians.
The IDEAS (Imaging Dementia – Evidence for Amyloid Scanning) Study, sponsored by the Alzheimer’s Association and managed by the American College of Radiology, seeks to enroll 18,488 patients aged 65 years and older with progressive, unexplained mild cognitive impairment or dementia of uncertain cause. The study will evaluate how knowing a patient’s amyloid status might affect clinical decision-making and health care utilization over 12 months. Patients will begin to enter the study early next year.
The scope of the $100 million study is unprecedented, said Maria Carrillo, Ph.D., chief science officer of the Alzheimer’s Association and cochair of the study.
“The Alzheimer’s Association is leading the IDEAS Study because we understand the importance of early and accurate diagnosis and management of dementia for individuals with the disease and their families,” she said in a statement. “There has never been a study of this scale to evaluate the clinical value of brain amyloid imaging in diagnosing and caring for those affected by Alzheimer’s.”
Physicians who participate will team with radiologists and nuclear medicine physicians at PET facilities; everyone will consent to fulfilling study training, data, and timeline requirements. Study referrals must come from dementia specialists who are registered as participating in the trial. Once accepted, a patient will undergo one amyloid PET scan and be followed for 12 months. The scans will be paid for under the CMS Coverage with Evidence Development policy.
The study will collect data in two areas:
• How the scan affects patient management in the first 90 days after the procedure, comparing actual postscan management to the prescan intended management. Outcomes here include the use of Alzheimer’s drugs, other medications, and counseling about safety and future planning.
• How the scan affects 12-month medical outcomes in both patients and controls. This analysis will be based on Medicare claims data; the primary objective is to learn whether amyloid imaging is associated with any significant reductions in outcomes such as hospitalization and emergency department visits. Health resource utilization will also be examined.
By examining these data, the study aims to show that amyloid imaging early in the disease process will improve both clinical and financial outcomes. If this can be established, the goal will be for the Centers for Medicare & Medicaid Services to reverse its 2013 decision to not cover the scans, except for patients who are enrolled in clinical trials.
At that time, CMS agreed that amyloid imaging is useful for excluding Alzheimer’s in some clinically difficult diagnoses and that it is a very good tool for ensuring that amyloid-positive patients are actually being enrolled in studies. But, officials said, there is little extant evidence that being able to identify amyloid plaques in the brain of a cognitively impaired patient can do anything to alter the trajectory of illness, either clinically of financially. It’s never been linked to improvements in any clinical outcomes – either avoidance of expensive or futile testing, reduced hospitalizations, improved quality of life, or delay of disease progression. And since there are no disease-modifying therapies, amyloid imaging has never been proven to contribute to treatment decisions.
Referring physician eligibility
Participating physicians must be dementia specialists: trained and board certified in neurology, psychiatry, or geriatric medicine and devoting at least 25% of patient time to the evaluation and care of adults with acquired cognitive impairment or dementia. Each dementia specialist will be included in a contractual agreement with the American College of Radiology, which will ensure eligibility and facilitate payment for submitted data. Each referring physician’s practice will need to obtain approval of an Institutional Review Board to participate in the study. The IDEAS team has contracted with Schulman Associates to provide central Institutional Review Board (IRB) services, but local IRB approval is also acceptable.
Patient eligibility
Patients must have either mild cognitive impairment or dementia of unknown etiology, meeting the Alzheimer’s Association’s appropriate use criteria for amyloid imaging. They must undergo laboratory tests (complete blood count, comprehensive metabolic panel, thyroid function tests, and vitamin B12 level) and structural brain imaging (CT or MRI). Information about other diagnostic tests completed prior to referral (e.g., lumbar puncture; fluorodeoxyglucose, or FDG-PET) will be collected, but these tests are not required.
Times have changed.
When I was a medical student in the early 1980s, the prevailing philosophy seemed to be that Alzheimer’s disease was diagnosed on the basis of negative tests, and that less-common causes of dementia, such as Pick’s disease, were clinically indistinguishable and identifiable only at autopsy. With greater neuropsychological sophistication and a wave of newly trained behavioral neurologists, Alzheimer’s disease was clinically identifiable as a distinct syndrome that was readily distinguished in most cases from other forms of dementia.
Technological breakthroughs have now added biomarkers to the diagnostic armamentarium. Despite the considerable progress that has been made in understanding the neurobiology of Alzheimer’s disease and the refinements in its diagnosis, therapy has lagged woefully. Modest symptomatic measures are available, but no one would argue that the current state is even close to satisfactory.

|
Dr. Richard J. Caselli |
Molecular imaging, such as amyloid PET and more recently tau PET, can image specific pathology and very obviously can aid in the diagnosis of Alzheimer’s disease. Amyloid PET is not perfect, though. Patients can have neurofibrillary tau tangle–only Alzheimer’s with a negative scan, and normal elderly controls with positive scans may fail to exhibit signs of dementia prior to their ultimate demise. A patient suspected of having Alzheimer’s has limited therapeutic options, and because they are all symptom based, they vary little based on whether the true diagnosis is Alzheimer’s, frontotemporal dementia, or Lewy body dementia.
New therapeutic agents being tested in research trials are a different matter and clearly need to be administered to a pathologically appropriate substrate, which molecular imaging can clarify. Because of the clinical limitations, however, CMS [Centers for Medicare & Medicaid Services] elected to withhold reimbursement of amyloid PET – a decision that has frustrated dementia specialists and families seeking definite evidence of a progressive, incurable, fatal disorder.
The IDEAS study is our chance to demonstrate what many suspect: that molecular imaging can provide more definite answers, and that such answers are most needed in diagnostically challenging cases.
Since amyloid PET will be reimbursed under the terms of this trial, our field has a golden opportunity to prove the clinical value of amyloid PET. We are all encouraged to contribute to its completion and hope for its success.
Dr. Richard J. Caselli is a professor of neurology at the Mayo Clinic in Scottsdale, Ariz., and clinical core director of the Arizona Alzheimer’s Disease Center, Phoenix. He has no relevant disclosures.
This commentary was updated October 29, 2015.
Times have changed.
When I was a medical student in the early 1980s, the prevailing philosophy seemed to be that Alzheimer’s disease was diagnosed on the basis of negative tests, and that less-common causes of dementia, such as Pick’s disease, were clinically indistinguishable and identifiable only at autopsy. With greater neuropsychological sophistication and a wave of newly trained behavioral neurologists, Alzheimer’s disease was clinically identifiable as a distinct syndrome that was readily distinguished in most cases from other forms of dementia.
Technological breakthroughs have now added biomarkers to the diagnostic armamentarium. Despite the considerable progress that has been made in understanding the neurobiology of Alzheimer’s disease and the refinements in its diagnosis, therapy has lagged woefully. Modest symptomatic measures are available, but no one would argue that the current state is even close to satisfactory.

|
Dr. Richard J. Caselli |
Molecular imaging, such as amyloid PET and more recently tau PET, can image specific pathology and very obviously can aid in the diagnosis of Alzheimer’s disease. Amyloid PET is not perfect, though. Patients can have neurofibrillary tau tangle–only Alzheimer’s with a negative scan, and normal elderly controls with positive scans may fail to exhibit signs of dementia prior to their ultimate demise. A patient suspected of having Alzheimer’s has limited therapeutic options, and because they are all symptom based, they vary little based on whether the true diagnosis is Alzheimer’s, frontotemporal dementia, or Lewy body dementia.
New therapeutic agents being tested in research trials are a different matter and clearly need to be administered to a pathologically appropriate substrate, which molecular imaging can clarify. Because of the clinical limitations, however, CMS [Centers for Medicare & Medicaid Services] elected to withhold reimbursement of amyloid PET – a decision that has frustrated dementia specialists and families seeking definite evidence of a progressive, incurable, fatal disorder.
The IDEAS study is our chance to demonstrate what many suspect: that molecular imaging can provide more definite answers, and that such answers are most needed in diagnostically challenging cases.
Since amyloid PET will be reimbursed under the terms of this trial, our field has a golden opportunity to prove the clinical value of amyloid PET. We are all encouraged to contribute to its completion and hope for its success.
Dr. Richard J. Caselli is a professor of neurology at the Mayo Clinic in Scottsdale, Ariz., and clinical core director of the Arizona Alzheimer’s Disease Center, Phoenix. He has no relevant disclosures.
This commentary was updated October 29, 2015.
Times have changed.
When I was a medical student in the early 1980s, the prevailing philosophy seemed to be that Alzheimer’s disease was diagnosed on the basis of negative tests, and that less-common causes of dementia, such as Pick’s disease, were clinically indistinguishable and identifiable only at autopsy. With greater neuropsychological sophistication and a wave of newly trained behavioral neurologists, Alzheimer’s disease was clinically identifiable as a distinct syndrome that was readily distinguished in most cases from other forms of dementia.
Technological breakthroughs have now added biomarkers to the diagnostic armamentarium. Despite the considerable progress that has been made in understanding the neurobiology of Alzheimer’s disease and the refinements in its diagnosis, therapy has lagged woefully. Modest symptomatic measures are available, but no one would argue that the current state is even close to satisfactory.

|
Dr. Richard J. Caselli |
Molecular imaging, such as amyloid PET and more recently tau PET, can image specific pathology and very obviously can aid in the diagnosis of Alzheimer’s disease. Amyloid PET is not perfect, though. Patients can have neurofibrillary tau tangle–only Alzheimer’s with a negative scan, and normal elderly controls with positive scans may fail to exhibit signs of dementia prior to their ultimate demise. A patient suspected of having Alzheimer’s has limited therapeutic options, and because they are all symptom based, they vary little based on whether the true diagnosis is Alzheimer’s, frontotemporal dementia, or Lewy body dementia.
New therapeutic agents being tested in research trials are a different matter and clearly need to be administered to a pathologically appropriate substrate, which molecular imaging can clarify. Because of the clinical limitations, however, CMS [Centers for Medicare & Medicaid Services] elected to withhold reimbursement of amyloid PET – a decision that has frustrated dementia specialists and families seeking definite evidence of a progressive, incurable, fatal disorder.
The IDEAS study is our chance to demonstrate what many suspect: that molecular imaging can provide more definite answers, and that such answers are most needed in diagnostically challenging cases.
Since amyloid PET will be reimbursed under the terms of this trial, our field has a golden opportunity to prove the clinical value of amyloid PET. We are all encouraged to contribute to its completion and hope for its success.
Dr. Richard J. Caselli is a professor of neurology at the Mayo Clinic in Scottsdale, Ariz., and clinical core director of the Arizona Alzheimer’s Disease Center, Phoenix. He has no relevant disclosures.
This commentary was updated October 29, 2015.
A major study aimed at proving the clinical utility of amyloid imaging in diagnosing dementia is now recruiting participating physicians.
The IDEAS (Imaging Dementia – Evidence for Amyloid Scanning) Study, sponsored by the Alzheimer’s Association and managed by the American College of Radiology, seeks to enroll 18,488 patients aged 65 years and older with progressive, unexplained mild cognitive impairment or dementia of uncertain cause. The study will evaluate how knowing a patient’s amyloid status might affect clinical decision-making and health care utilization over 12 months. Patients will begin to enter the study early next year.
The scope of the $100 million study is unprecedented, said Maria Carrillo, Ph.D., chief science officer of the Alzheimer’s Association and cochair of the study.
“The Alzheimer’s Association is leading the IDEAS Study because we understand the importance of early and accurate diagnosis and management of dementia for individuals with the disease and their families,” she said in a statement. “There has never been a study of this scale to evaluate the clinical value of brain amyloid imaging in diagnosing and caring for those affected by Alzheimer’s.”
Physicians who participate will team with radiologists and nuclear medicine physicians at PET facilities; everyone will consent to fulfilling study training, data, and timeline requirements. Study referrals must come from dementia specialists who are registered as participating in the trial. Once accepted, a patient will undergo one amyloid PET scan and be followed for 12 months. The scans will be paid for under the CMS Coverage with Evidence Development policy.
The study will collect data in two areas:
• How the scan affects patient management in the first 90 days after the procedure, comparing actual postscan management to the prescan intended management. Outcomes here include the use of Alzheimer’s drugs, other medications, and counseling about safety and future planning.
• How the scan affects 12-month medical outcomes in both patients and controls. This analysis will be based on Medicare claims data; the primary objective is to learn whether amyloid imaging is associated with any significant reductions in outcomes such as hospitalization and emergency department visits. Health resource utilization will also be examined.
By examining these data, the study aims to show that amyloid imaging early in the disease process will improve both clinical and financial outcomes. If this can be established, the goal will be for the Centers for Medicare & Medicaid Services to reverse its 2013 decision to not cover the scans, except for patients who are enrolled in clinical trials.
At that time, CMS agreed that amyloid imaging is useful for excluding Alzheimer’s in some clinically difficult diagnoses and that it is a very good tool for ensuring that amyloid-positive patients are actually being enrolled in studies. But, officials said, there is little extant evidence that being able to identify amyloid plaques in the brain of a cognitively impaired patient can do anything to alter the trajectory of illness, either clinically of financially. It’s never been linked to improvements in any clinical outcomes – either avoidance of expensive or futile testing, reduced hospitalizations, improved quality of life, or delay of disease progression. And since there are no disease-modifying therapies, amyloid imaging has never been proven to contribute to treatment decisions.
Referring physician eligibility
Participating physicians must be dementia specialists: trained and board certified in neurology, psychiatry, or geriatric medicine and devoting at least 25% of patient time to the evaluation and care of adults with acquired cognitive impairment or dementia. Each dementia specialist will be included in a contractual agreement with the American College of Radiology, which will ensure eligibility and facilitate payment for submitted data. Each referring physician’s practice will need to obtain approval of an Institutional Review Board to participate in the study. The IDEAS team has contracted with Schulman Associates to provide central Institutional Review Board (IRB) services, but local IRB approval is also acceptable.
Patient eligibility
Patients must have either mild cognitive impairment or dementia of unknown etiology, meeting the Alzheimer’s Association’s appropriate use criteria for amyloid imaging. They must undergo laboratory tests (complete blood count, comprehensive metabolic panel, thyroid function tests, and vitamin B12 level) and structural brain imaging (CT or MRI). Information about other diagnostic tests completed prior to referral (e.g., lumbar puncture; fluorodeoxyglucose, or FDG-PET) will be collected, but these tests are not required.
A major study aimed at proving the clinical utility of amyloid imaging in diagnosing dementia is now recruiting participating physicians.
The IDEAS (Imaging Dementia – Evidence for Amyloid Scanning) Study, sponsored by the Alzheimer’s Association and managed by the American College of Radiology, seeks to enroll 18,488 patients aged 65 years and older with progressive, unexplained mild cognitive impairment or dementia of uncertain cause. The study will evaluate how knowing a patient’s amyloid status might affect clinical decision-making and health care utilization over 12 months. Patients will begin to enter the study early next year.
The scope of the $100 million study is unprecedented, said Maria Carrillo, Ph.D., chief science officer of the Alzheimer’s Association and cochair of the study.
“The Alzheimer’s Association is leading the IDEAS Study because we understand the importance of early and accurate diagnosis and management of dementia for individuals with the disease and their families,” she said in a statement. “There has never been a study of this scale to evaluate the clinical value of brain amyloid imaging in diagnosing and caring for those affected by Alzheimer’s.”
Physicians who participate will team with radiologists and nuclear medicine physicians at PET facilities; everyone will consent to fulfilling study training, data, and timeline requirements. Study referrals must come from dementia specialists who are registered as participating in the trial. Once accepted, a patient will undergo one amyloid PET scan and be followed for 12 months. The scans will be paid for under the CMS Coverage with Evidence Development policy.
The study will collect data in two areas:
• How the scan affects patient management in the first 90 days after the procedure, comparing actual postscan management to the prescan intended management. Outcomes here include the use of Alzheimer’s drugs, other medications, and counseling about safety and future planning.
• How the scan affects 12-month medical outcomes in both patients and controls. This analysis will be based on Medicare claims data; the primary objective is to learn whether amyloid imaging is associated with any significant reductions in outcomes such as hospitalization and emergency department visits. Health resource utilization will also be examined.
By examining these data, the study aims to show that amyloid imaging early in the disease process will improve both clinical and financial outcomes. If this can be established, the goal will be for the Centers for Medicare & Medicaid Services to reverse its 2013 decision to not cover the scans, except for patients who are enrolled in clinical trials.
At that time, CMS agreed that amyloid imaging is useful for excluding Alzheimer’s in some clinically difficult diagnoses and that it is a very good tool for ensuring that amyloid-positive patients are actually being enrolled in studies. But, officials said, there is little extant evidence that being able to identify amyloid plaques in the brain of a cognitively impaired patient can do anything to alter the trajectory of illness, either clinically of financially. It’s never been linked to improvements in any clinical outcomes – either avoidance of expensive or futile testing, reduced hospitalizations, improved quality of life, or delay of disease progression. And since there are no disease-modifying therapies, amyloid imaging has never been proven to contribute to treatment decisions.
Referring physician eligibility
Participating physicians must be dementia specialists: trained and board certified in neurology, psychiatry, or geriatric medicine and devoting at least 25% of patient time to the evaluation and care of adults with acquired cognitive impairment or dementia. Each dementia specialist will be included in a contractual agreement with the American College of Radiology, which will ensure eligibility and facilitate payment for submitted data. Each referring physician’s practice will need to obtain approval of an Institutional Review Board to participate in the study. The IDEAS team has contracted with Schulman Associates to provide central Institutional Review Board (IRB) services, but local IRB approval is also acceptable.
Patient eligibility
Patients must have either mild cognitive impairment or dementia of unknown etiology, meeting the Alzheimer’s Association’s appropriate use criteria for amyloid imaging. They must undergo laboratory tests (complete blood count, comprehensive metabolic panel, thyroid function tests, and vitamin B12 level) and structural brain imaging (CT or MRI). Information about other diagnostic tests completed prior to referral (e.g., lumbar puncture; fluorodeoxyglucose, or FDG-PET) will be collected, but these tests are not required.