User login
TCT: Early intervention cut mortality in severe, asymptomatic AS
SAN FRANCISCO - Early valve replacement may be in the best interest of asymptomatic patients with severe aortic stenosis, possibly halving their 5-year risk of death, based on data from the CURRENT AS registry study.
Compared to watchful waiting, early surgical intervention also reduced by 81% the risk of hospitalization for heart failure, Dr. Tomohiko Taniguchi said at the Transcatheter Cardiovascular Therapeutics annual meeting. The study was simultaneously published in the Journal of the American College of Cardiology (Am Coll Cardiol. 2015. doi: 10.1016/j.jacc.2015.10.001).
Observation has been the byword for asymptomatic patients with severe aortic stenosis (AS). The American College of Cardiology recommends a conservative approach to the asymptomatic AS patient, but acknowledges the disorder inevitably progresses in nearly all patients. In the ACC’s 2014 treatment guidelines, survival during the asymptomatic phase is similar to that of age-matched controls when patients are carefully followed.
But the CURRENT AS registry results suggest that “the long-term outcome of asymptomatic patients with severe aortic stenosis was dismal when they were managed conservatively in real clinical practice,” Dr. Taniguchi said during a press briefing at the meeting, which was sponsored by the Cardiovascular Research Foundation.
“If you’re watching and waiting, and you wait for sudden death, then that is a problem,” said Dr. Ajay J. Kirtane, who moderated the briefing. Early intervention “potentially changes the game because we do have a less-invasive procedure we can offer – transcatheter aortic valve replacement (AVR),” said Dr. Kirtane of New York-Presbyterian Hospital.
In the CURRENT AS study, severe AS was considered a peak aortic jet velocity over 4.0 m/s, or a mean aortic pressure gradient greater than 40 mm Hg, or an aortic valve area less than 1.0 cm2. The registry includes 3,815 patients; Dr. Taniguchi of Kyoto University reported outcomes for a propensity-score matched cohort of 582 patients, 291 in the initial AVR group and 291 in the conservatively managed group. There was no treatment randomization; treatment decisions were made at the clinical level.
Patients in the matched cohort were in their early 70s; in 80%, the AS etiology was degenerative. The mean aortic pressure gradient was 54 mm Hg in the early intervention group and 45 mm Hg in the watchful waiting group. In 79% of the early intervention group and in 54% of the watchful waiting group, the mean aortic pressure gradient was below 40 mm Hg.
Among the patients who underwent AVR despite being asymptomatic, most (63%) had at least one surgical indication, including severe AS (41%), left ventricular dysfunction (7%), rapid hemodynamic progression (11%), or active infective endocarditis (0.3%). Other cardiac surgery indications were present in 8%. Many of the conservatively managed patients (41%) did eventually require AVR, Dr. Taniguchi noted.
By the end of the 5-year follow up period, 26% of the conservative therapy group and 15% of the early AVR group had died – a significant difference (hazard ratio, 0.64; P = .02).
The coprimary endpoint of heart failure hospitalization was also significantly more common among the conservatively treated group (19.9% vs. 3.8%; HR, 0.19; P less than .001).
The early intervention group also did significantly better on several secondary clinical outcomes, including cardiovascular death (10% vs. 18.6%), aortic valve-related death (5.3% vs. 13.5%), sudden death (3.6% vs. 5.8%), and emerging symptoms (3.2% vs. 46.3%).
Dr. Taniguchi had no financial disclosures. The study was sponsored by Kyoto University.
The findings make a good case for carefully considering which patients might benefit more from early intervention than from close observation. They also suggest a place for less invasive valve replacement rather than watchful waiting in some asymptomatic patients.

|
Dr. Jeffrey Popma |
I think this finding is fabulous, and raises the question of whether the ventricle cares even if the patient doesn’t care. Should we be intervening earlier with transcatheter aortic valve replacement? A less-invasive therapy early on may have benefits.
Dr. Jeffrey J. Popma is professor of medicine at Harvard Medical School and director of interventional cardiology at the Beth Israel Deaconess Medical Center, both in Boston.
The findings make a good case for carefully considering which patients might benefit more from early intervention than from close observation. They also suggest a place for less invasive valve replacement rather than watchful waiting in some asymptomatic patients.

|
Dr. Jeffrey Popma |
I think this finding is fabulous, and raises the question of whether the ventricle cares even if the patient doesn’t care. Should we be intervening earlier with transcatheter aortic valve replacement? A less-invasive therapy early on may have benefits.
Dr. Jeffrey J. Popma is professor of medicine at Harvard Medical School and director of interventional cardiology at the Beth Israel Deaconess Medical Center, both in Boston.
The findings make a good case for carefully considering which patients might benefit more from early intervention than from close observation. They also suggest a place for less invasive valve replacement rather than watchful waiting in some asymptomatic patients.

|
Dr. Jeffrey Popma |
I think this finding is fabulous, and raises the question of whether the ventricle cares even if the patient doesn’t care. Should we be intervening earlier with transcatheter aortic valve replacement? A less-invasive therapy early on may have benefits.
Dr. Jeffrey J. Popma is professor of medicine at Harvard Medical School and director of interventional cardiology at the Beth Israel Deaconess Medical Center, both in Boston.
SAN FRANCISCO - Early valve replacement may be in the best interest of asymptomatic patients with severe aortic stenosis, possibly halving their 5-year risk of death, based on data from the CURRENT AS registry study.
Compared to watchful waiting, early surgical intervention also reduced by 81% the risk of hospitalization for heart failure, Dr. Tomohiko Taniguchi said at the Transcatheter Cardiovascular Therapeutics annual meeting. The study was simultaneously published in the Journal of the American College of Cardiology (Am Coll Cardiol. 2015. doi: 10.1016/j.jacc.2015.10.001).
Observation has been the byword for asymptomatic patients with severe aortic stenosis (AS). The American College of Cardiology recommends a conservative approach to the asymptomatic AS patient, but acknowledges the disorder inevitably progresses in nearly all patients. In the ACC’s 2014 treatment guidelines, survival during the asymptomatic phase is similar to that of age-matched controls when patients are carefully followed.
But the CURRENT AS registry results suggest that “the long-term outcome of asymptomatic patients with severe aortic stenosis was dismal when they were managed conservatively in real clinical practice,” Dr. Taniguchi said during a press briefing at the meeting, which was sponsored by the Cardiovascular Research Foundation.
“If you’re watching and waiting, and you wait for sudden death, then that is a problem,” said Dr. Ajay J. Kirtane, who moderated the briefing. Early intervention “potentially changes the game because we do have a less-invasive procedure we can offer – transcatheter aortic valve replacement (AVR),” said Dr. Kirtane of New York-Presbyterian Hospital.
In the CURRENT AS study, severe AS was considered a peak aortic jet velocity over 4.0 m/s, or a mean aortic pressure gradient greater than 40 mm Hg, or an aortic valve area less than 1.0 cm2. The registry includes 3,815 patients; Dr. Taniguchi of Kyoto University reported outcomes for a propensity-score matched cohort of 582 patients, 291 in the initial AVR group and 291 in the conservatively managed group. There was no treatment randomization; treatment decisions were made at the clinical level.
Patients in the matched cohort were in their early 70s; in 80%, the AS etiology was degenerative. The mean aortic pressure gradient was 54 mm Hg in the early intervention group and 45 mm Hg in the watchful waiting group. In 79% of the early intervention group and in 54% of the watchful waiting group, the mean aortic pressure gradient was below 40 mm Hg.
Among the patients who underwent AVR despite being asymptomatic, most (63%) had at least one surgical indication, including severe AS (41%), left ventricular dysfunction (7%), rapid hemodynamic progression (11%), or active infective endocarditis (0.3%). Other cardiac surgery indications were present in 8%. Many of the conservatively managed patients (41%) did eventually require AVR, Dr. Taniguchi noted.
By the end of the 5-year follow up period, 26% of the conservative therapy group and 15% of the early AVR group had died – a significant difference (hazard ratio, 0.64; P = .02).
The coprimary endpoint of heart failure hospitalization was also significantly more common among the conservatively treated group (19.9% vs. 3.8%; HR, 0.19; P less than .001).
The early intervention group also did significantly better on several secondary clinical outcomes, including cardiovascular death (10% vs. 18.6%), aortic valve-related death (5.3% vs. 13.5%), sudden death (3.6% vs. 5.8%), and emerging symptoms (3.2% vs. 46.3%).
Dr. Taniguchi had no financial disclosures. The study was sponsored by Kyoto University.
SAN FRANCISCO - Early valve replacement may be in the best interest of asymptomatic patients with severe aortic stenosis, possibly halving their 5-year risk of death, based on data from the CURRENT AS registry study.
Compared to watchful waiting, early surgical intervention also reduced by 81% the risk of hospitalization for heart failure, Dr. Tomohiko Taniguchi said at the Transcatheter Cardiovascular Therapeutics annual meeting. The study was simultaneously published in the Journal of the American College of Cardiology (Am Coll Cardiol. 2015. doi: 10.1016/j.jacc.2015.10.001).
Observation has been the byword for asymptomatic patients with severe aortic stenosis (AS). The American College of Cardiology recommends a conservative approach to the asymptomatic AS patient, but acknowledges the disorder inevitably progresses in nearly all patients. In the ACC’s 2014 treatment guidelines, survival during the asymptomatic phase is similar to that of age-matched controls when patients are carefully followed.
But the CURRENT AS registry results suggest that “the long-term outcome of asymptomatic patients with severe aortic stenosis was dismal when they were managed conservatively in real clinical practice,” Dr. Taniguchi said during a press briefing at the meeting, which was sponsored by the Cardiovascular Research Foundation.
“If you’re watching and waiting, and you wait for sudden death, then that is a problem,” said Dr. Ajay J. Kirtane, who moderated the briefing. Early intervention “potentially changes the game because we do have a less-invasive procedure we can offer – transcatheter aortic valve replacement (AVR),” said Dr. Kirtane of New York-Presbyterian Hospital.
In the CURRENT AS study, severe AS was considered a peak aortic jet velocity over 4.0 m/s, or a mean aortic pressure gradient greater than 40 mm Hg, or an aortic valve area less than 1.0 cm2. The registry includes 3,815 patients; Dr. Taniguchi of Kyoto University reported outcomes for a propensity-score matched cohort of 582 patients, 291 in the initial AVR group and 291 in the conservatively managed group. There was no treatment randomization; treatment decisions were made at the clinical level.
Patients in the matched cohort were in their early 70s; in 80%, the AS etiology was degenerative. The mean aortic pressure gradient was 54 mm Hg in the early intervention group and 45 mm Hg in the watchful waiting group. In 79% of the early intervention group and in 54% of the watchful waiting group, the mean aortic pressure gradient was below 40 mm Hg.
Among the patients who underwent AVR despite being asymptomatic, most (63%) had at least one surgical indication, including severe AS (41%), left ventricular dysfunction (7%), rapid hemodynamic progression (11%), or active infective endocarditis (0.3%). Other cardiac surgery indications were present in 8%. Many of the conservatively managed patients (41%) did eventually require AVR, Dr. Taniguchi noted.
By the end of the 5-year follow up period, 26% of the conservative therapy group and 15% of the early AVR group had died – a significant difference (hazard ratio, 0.64; P = .02).
The coprimary endpoint of heart failure hospitalization was also significantly more common among the conservatively treated group (19.9% vs. 3.8%; HR, 0.19; P less than .001).
The early intervention group also did significantly better on several secondary clinical outcomes, including cardiovascular death (10% vs. 18.6%), aortic valve-related death (5.3% vs. 13.5%), sudden death (3.6% vs. 5.8%), and emerging symptoms (3.2% vs. 46.3%).
Dr. Taniguchi had no financial disclosures. The study was sponsored by Kyoto University.
TCT: FORMA system tested in severe tricuspid regurgitation
The investigational FORMA system seems safe and may be effective in patients with NYHA Class III/IV heart failure and severe tricuspid valve regurgitation, based on 13 first-in-human cases.
A Canadian surgical team employed the FORMA system (Edwards Lifesciences) as compassionate use therapy for a set of patients with inoperable tricuspid regurgitation. The device was successfully deployed in 12 of the 13 patients, according to data presented at the Transcatheter Cardiovascular Therapeutics annual meeting. There were no deaths or major clinical complications in any of the patients.
A report on seven of these patients was simultaneously published in the Journal of the American College of Cardiology. All of the patients had severe tricuspid regurgitation and heart failure; before surgery, six had a New York Heart Association (NYHA) Functional Classification of III/IV. By 30 days after the procedure, all had improved to NYHA II, wrote Dr. Francisco Campelo-Parada of the Quebec Heart and Lung Institute, the paper’s primary author. Peripheral edema declined and all patients experienced functional improvement, as well.
According to Edwards Lifesciences, the FORMA device uses a foam-filled polymer balloon spacer to reduce tricuspid regurgitation by occupying the regurgitant orifice area and providing a surface for the coaptation of the valve’s native leaflets. Implantation is performed via the left axillary vein.
Patients in the series were a mean of 76 years old. All had severe tricuspid regurgitation. The mean maximal vena contracta was 15.5 mm.
Six had coronary artery disease and five had previously undergone open heart surgery. Additionally, two had previously undergone mitral valve surgery and two had undergone aortic valve surgery. Pulmonary hypertension was present in five. Five patients also had persistent atrial fibrillation. Six had renal insufficiency, with one patient on dialysis. The baseline furosemide dose was 80 mg/day.
All procedures were performed under general sedation and fluoroscopic guidance, with postprocedural positioning checked by cardiac-CT and/or a chest x-ray. The mean postop stay was 4 days.
Tricuspid regurgitation was reduced by at least 1 degree in all patients during the operation; four patients had an immediate 2-degree reduction, reclassifying their regurgitation as mild. Two experienced new-onset atrial fibrillation, and one had several episodes of nonsustained ventricular tachycardia that was managed with beta-blockers.
At the first clinical follow-up 30 days after surgery, all but one patient had an improvement to Class II NYHA status.
Two patients were able to reduce their diuretic dosage; there were no other medication changes. Peripheral edema declined in the entire cohort. Tricuspid regurgitation was graded as moderate in all patients.
There were also associated improvements in quality of life, based on scores on the Kansas City Cardiomyopathy Questionnaire, which increased from 59 before surgery to 86 after surgery. Exercise capacity as measured by the 6-Minute Walk Test improved from 297 meters to 326 meters.
The authors suggested that the 15-mm spacer used in the FORMA device was not well-matched with the mean 15.5-mm vena contracta size in the cohort. Better outcomes might be possible if a larger spacer were available.
“Despite good device positioning, complete coaptation was not achieved, resulting in significant residual degree of postprocedural tricuspid regurgitation,” they said. “Also, the very advanced stage of the disease in most patients may have played a role in the mild reduction at 30 days.”
Despite the rather mild, 1-degree improvement, patients did make considerable improvements in heart failure and functional status. Therefore, the team recommended further study for FORMA, with an eye toward optimizing patient selection.
“Specific criteria for quantifying right ventricular dysfunction and pulmonary hypertension, along with novel quantitative echocardiographic imaging criteria may be required,” they said. “It is conceivable that larger than the currently available spacer sizes may be required to improve echocardiographic results in patients with large noncoaptation defects and vena contracta.”
Dr. Campelo-Parada had no financial disclosures with regard to the device.
The investigational FORMA system seems safe and may be effective in patients with NYHA Class III/IV heart failure and severe tricuspid valve regurgitation, based on 13 first-in-human cases.
A Canadian surgical team employed the FORMA system (Edwards Lifesciences) as compassionate use therapy for a set of patients with inoperable tricuspid regurgitation. The device was successfully deployed in 12 of the 13 patients, according to data presented at the Transcatheter Cardiovascular Therapeutics annual meeting. There were no deaths or major clinical complications in any of the patients.
A report on seven of these patients was simultaneously published in the Journal of the American College of Cardiology. All of the patients had severe tricuspid regurgitation and heart failure; before surgery, six had a New York Heart Association (NYHA) Functional Classification of III/IV. By 30 days after the procedure, all had improved to NYHA II, wrote Dr. Francisco Campelo-Parada of the Quebec Heart and Lung Institute, the paper’s primary author. Peripheral edema declined and all patients experienced functional improvement, as well.
According to Edwards Lifesciences, the FORMA device uses a foam-filled polymer balloon spacer to reduce tricuspid regurgitation by occupying the regurgitant orifice area and providing a surface for the coaptation of the valve’s native leaflets. Implantation is performed via the left axillary vein.
Patients in the series were a mean of 76 years old. All had severe tricuspid regurgitation. The mean maximal vena contracta was 15.5 mm.
Six had coronary artery disease and five had previously undergone open heart surgery. Additionally, two had previously undergone mitral valve surgery and two had undergone aortic valve surgery. Pulmonary hypertension was present in five. Five patients also had persistent atrial fibrillation. Six had renal insufficiency, with one patient on dialysis. The baseline furosemide dose was 80 mg/day.
All procedures were performed under general sedation and fluoroscopic guidance, with postprocedural positioning checked by cardiac-CT and/or a chest x-ray. The mean postop stay was 4 days.
Tricuspid regurgitation was reduced by at least 1 degree in all patients during the operation; four patients had an immediate 2-degree reduction, reclassifying their regurgitation as mild. Two experienced new-onset atrial fibrillation, and one had several episodes of nonsustained ventricular tachycardia that was managed with beta-blockers.
At the first clinical follow-up 30 days after surgery, all but one patient had an improvement to Class II NYHA status.
Two patients were able to reduce their diuretic dosage; there were no other medication changes. Peripheral edema declined in the entire cohort. Tricuspid regurgitation was graded as moderate in all patients.
There were also associated improvements in quality of life, based on scores on the Kansas City Cardiomyopathy Questionnaire, which increased from 59 before surgery to 86 after surgery. Exercise capacity as measured by the 6-Minute Walk Test improved from 297 meters to 326 meters.
The authors suggested that the 15-mm spacer used in the FORMA device was not well-matched with the mean 15.5-mm vena contracta size in the cohort. Better outcomes might be possible if a larger spacer were available.
“Despite good device positioning, complete coaptation was not achieved, resulting in significant residual degree of postprocedural tricuspid regurgitation,” they said. “Also, the very advanced stage of the disease in most patients may have played a role in the mild reduction at 30 days.”
Despite the rather mild, 1-degree improvement, patients did make considerable improvements in heart failure and functional status. Therefore, the team recommended further study for FORMA, with an eye toward optimizing patient selection.
“Specific criteria for quantifying right ventricular dysfunction and pulmonary hypertension, along with novel quantitative echocardiographic imaging criteria may be required,” they said. “It is conceivable that larger than the currently available spacer sizes may be required to improve echocardiographic results in patients with large noncoaptation defects and vena contracta.”
Dr. Campelo-Parada had no financial disclosures with regard to the device.
The investigational FORMA system seems safe and may be effective in patients with NYHA Class III/IV heart failure and severe tricuspid valve regurgitation, based on 13 first-in-human cases.
A Canadian surgical team employed the FORMA system (Edwards Lifesciences) as compassionate use therapy for a set of patients with inoperable tricuspid regurgitation. The device was successfully deployed in 12 of the 13 patients, according to data presented at the Transcatheter Cardiovascular Therapeutics annual meeting. There were no deaths or major clinical complications in any of the patients.
A report on seven of these patients was simultaneously published in the Journal of the American College of Cardiology. All of the patients had severe tricuspid regurgitation and heart failure; before surgery, six had a New York Heart Association (NYHA) Functional Classification of III/IV. By 30 days after the procedure, all had improved to NYHA II, wrote Dr. Francisco Campelo-Parada of the Quebec Heart and Lung Institute, the paper’s primary author. Peripheral edema declined and all patients experienced functional improvement, as well.
According to Edwards Lifesciences, the FORMA device uses a foam-filled polymer balloon spacer to reduce tricuspid regurgitation by occupying the regurgitant orifice area and providing a surface for the coaptation of the valve’s native leaflets. Implantation is performed via the left axillary vein.
Patients in the series were a mean of 76 years old. All had severe tricuspid regurgitation. The mean maximal vena contracta was 15.5 mm.
Six had coronary artery disease and five had previously undergone open heart surgery. Additionally, two had previously undergone mitral valve surgery and two had undergone aortic valve surgery. Pulmonary hypertension was present in five. Five patients also had persistent atrial fibrillation. Six had renal insufficiency, with one patient on dialysis. The baseline furosemide dose was 80 mg/day.
All procedures were performed under general sedation and fluoroscopic guidance, with postprocedural positioning checked by cardiac-CT and/or a chest x-ray. The mean postop stay was 4 days.
Tricuspid regurgitation was reduced by at least 1 degree in all patients during the operation; four patients had an immediate 2-degree reduction, reclassifying their regurgitation as mild. Two experienced new-onset atrial fibrillation, and one had several episodes of nonsustained ventricular tachycardia that was managed with beta-blockers.
At the first clinical follow-up 30 days after surgery, all but one patient had an improvement to Class II NYHA status.
Two patients were able to reduce their diuretic dosage; there were no other medication changes. Peripheral edema declined in the entire cohort. Tricuspid regurgitation was graded as moderate in all patients.
There were also associated improvements in quality of life, based on scores on the Kansas City Cardiomyopathy Questionnaire, which increased from 59 before surgery to 86 after surgery. Exercise capacity as measured by the 6-Minute Walk Test improved from 297 meters to 326 meters.
The authors suggested that the 15-mm spacer used in the FORMA device was not well-matched with the mean 15.5-mm vena contracta size in the cohort. Better outcomes might be possible if a larger spacer were available.
“Despite good device positioning, complete coaptation was not achieved, resulting in significant residual degree of postprocedural tricuspid regurgitation,” they said. “Also, the very advanced stage of the disease in most patients may have played a role in the mild reduction at 30 days.”
Despite the rather mild, 1-degree improvement, patients did make considerable improvements in heart failure and functional status. Therefore, the team recommended further study for FORMA, with an eye toward optimizing patient selection.
“Specific criteria for quantifying right ventricular dysfunction and pulmonary hypertension, along with novel quantitative echocardiographic imaging criteria may be required,” they said. “It is conceivable that larger than the currently available spacer sizes may be required to improve echocardiographic results in patients with large noncoaptation defects and vena contracta.”
Dr. Campelo-Parada had no financial disclosures with regard to the device.
FROM TCT 2015
Key clinical point: The investigational FORMA system seems safe and may be effective in patients with NYHA Class III/IV heart failure and severe tricuspid valve regurgitation.
Major finding: The improved heart failure from NYHA Class III/IV to Class II in six of seven patients with severe tricuspid valve regurgitation.
Data source: The device has been used in 13 patients thus far, under compassionate use allowance.
Disclosures: Edwards Lifesciences manufactures and is investigating the device. Dr. Campelo-Parada had no disclosures.
RFA, ethanol ablation equally effective for thyroid nodules
LAKE BUENA VISTA, FLA. – Ethanol ablation is just as effective as radiofrequency ablation for cystic thyroid nodules, resulting in similar volume reduction and similarly improving symptomatic and cosmetic outcomes at 6 months.
Radiofrequnecy ablation (RFA) did have a slight edge over ethanol injection (EA) in therapeutic response, Dr. Hye Sun Park said at the International Thyroid Conference. But because ethanol ablation is easier and less expensive, she recommended that it be considered as first-line therapy for these lesions.
Dr. Park of the University of Ulsan, Asan Medical Center in Seoul, South Korea, reported a trial of 46 patients, mean age 50 years old, with benign cystic thyroid nodules who were randomized to the two treatments. Patients randomized to ethanol, however, had significantly larger-volume lesions (14.7 vs. 8.6 mL), and symptom scores. Cosmetic scores and nodule vascularity were similar.
RFA was performed with an 18-gauge monopolar, internally cooled electrode with a 1-cm active tip, using the moving shot technique. Patients undergoing EA first had fluid removed from the nodules using a 16-gauge needle. Ablation consisted of an injection of 99% ethanol into the cystic space, which was removed after 2 minutes.
The primary outcome was nodule volume at 6 months. Secondary outcomes were postprocedural pain and complications, Dr. Park said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
At follow-up, the volume reductions were similar in RFA and EA (87.5 vs. 82.4 ml). The therapeutic success rate was 100% for patients who had RFA and 92% for those who had EA. Two patients in the EA group had major bleeding from the nodule, which interrupted the procedure; they later had a successful RFA. There were no complications in the RFA group.
There was only one major complication during follow-up: One patient who received EA complained of voice change, which resolved spontaneously by 2 months after ablation.
Dr. Park had no financial disclosures.
LAKE BUENA VISTA, FLA. – Ethanol ablation is just as effective as radiofrequency ablation for cystic thyroid nodules, resulting in similar volume reduction and similarly improving symptomatic and cosmetic outcomes at 6 months.
Radiofrequnecy ablation (RFA) did have a slight edge over ethanol injection (EA) in therapeutic response, Dr. Hye Sun Park said at the International Thyroid Conference. But because ethanol ablation is easier and less expensive, she recommended that it be considered as first-line therapy for these lesions.
Dr. Park of the University of Ulsan, Asan Medical Center in Seoul, South Korea, reported a trial of 46 patients, mean age 50 years old, with benign cystic thyroid nodules who were randomized to the two treatments. Patients randomized to ethanol, however, had significantly larger-volume lesions (14.7 vs. 8.6 mL), and symptom scores. Cosmetic scores and nodule vascularity were similar.
RFA was performed with an 18-gauge monopolar, internally cooled electrode with a 1-cm active tip, using the moving shot technique. Patients undergoing EA first had fluid removed from the nodules using a 16-gauge needle. Ablation consisted of an injection of 99% ethanol into the cystic space, which was removed after 2 minutes.
The primary outcome was nodule volume at 6 months. Secondary outcomes were postprocedural pain and complications, Dr. Park said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
At follow-up, the volume reductions were similar in RFA and EA (87.5 vs. 82.4 ml). The therapeutic success rate was 100% for patients who had RFA and 92% for those who had EA. Two patients in the EA group had major bleeding from the nodule, which interrupted the procedure; they later had a successful RFA. There were no complications in the RFA group.
There was only one major complication during follow-up: One patient who received EA complained of voice change, which resolved spontaneously by 2 months after ablation.
Dr. Park had no financial disclosures.
LAKE BUENA VISTA, FLA. – Ethanol ablation is just as effective as radiofrequency ablation for cystic thyroid nodules, resulting in similar volume reduction and similarly improving symptomatic and cosmetic outcomes at 6 months.
Radiofrequnecy ablation (RFA) did have a slight edge over ethanol injection (EA) in therapeutic response, Dr. Hye Sun Park said at the International Thyroid Conference. But because ethanol ablation is easier and less expensive, she recommended that it be considered as first-line therapy for these lesions.
Dr. Park of the University of Ulsan, Asan Medical Center in Seoul, South Korea, reported a trial of 46 patients, mean age 50 years old, with benign cystic thyroid nodules who were randomized to the two treatments. Patients randomized to ethanol, however, had significantly larger-volume lesions (14.7 vs. 8.6 mL), and symptom scores. Cosmetic scores and nodule vascularity were similar.
RFA was performed with an 18-gauge monopolar, internally cooled electrode with a 1-cm active tip, using the moving shot technique. Patients undergoing EA first had fluid removed from the nodules using a 16-gauge needle. Ablation consisted of an injection of 99% ethanol into the cystic space, which was removed after 2 minutes.
The primary outcome was nodule volume at 6 months. Secondary outcomes were postprocedural pain and complications, Dr. Park said at the meeting held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
At follow-up, the volume reductions were similar in RFA and EA (87.5 vs. 82.4 ml). The therapeutic success rate was 100% for patients who had RFA and 92% for those who had EA. Two patients in the EA group had major bleeding from the nodule, which interrupted the procedure; they later had a successful RFA. There were no complications in the RFA group.
There was only one major complication during follow-up: One patient who received EA complained of voice change, which resolved spontaneously by 2 months after ablation.
Dr. Park had no financial disclosures.
AT ITC 2015
Key clinical point: Radiofrequency ablation and ethanol ablation were similarly effective in treating cystic thyroid nodules.
Major finding: At 6 months, radiofrequency ablation and ethanol ablation achieved similar reductions in the volume of benign cystic thyroid nodules (87.5 vs. 82.4 mL).
Data source: The randomized study comprised 46 patients.
Disclosures: Dr Hye Sun Park had no financial disclosures.
Thyroglobulin can’t predict pazopanib response in differentiated thyroid cancer
Lake BUENA VISTA, FLA. – Pazopanib is an effective therapy for advanced thyroid cancer, but at present, there seems to be no way to predict which patients will respond to it.
An investigation into the predictive value of thyroglobulin found that the level during treatment did change, but it did so in parallel with response; there was no way to use the protein to parse out which patients would do well, Dr. Keith Bible said at the International Thyroid Congress.
“We had hoped that it might be a predictor as early as 4 weeks, so we could assign patients into categories of response and perhaps stop treatment earlier,” said Dr. Bible of the Mayo Clinic, Rochester, Minn. “Unfortunately, there was no way to do that.”
Pazopanib (Votrient) is a tyrosine kinase inhibitor of vascular endothelial growth factor receptors. It is approved for advanced soft tissue sarcoma and advanced renal cell carcinoma. The Mayo Clinic Consortium has been investigating pazopanib in phase II trials for advanced differentiated and medullary thyroid cancers. In a 2010 report, it was shown to induce at least a partial response in about half of the patients who received it (Lancet Oncol. 2010 Oct;11[10]:962-72).
Last year, it was also shown to be effective in advanced medullary thyroid cancer. Five of 35 patients attained partial Response Evaluation Criteria In Solid Tumors (RECIST) responses (14%) (J Clin Endocrinol Metab. 2014 May;99[5]:1687-93).
The drug has not been successful in treating advanced anaplastic thyroid cancer, however.
Because of pazopanib’s proclivity to induce sometimes-severe hypertension, investigators were hoping for some way to stratify potential responders. Thyroglobulin levels could be one marker, Dr. Bible said at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
He and the consortium investigators examined how thyroglobulin levels correlated with RECIST scores in 60 patients with metastatic differentiated thyroid cancers. The patients received a median of 10 cycles, but the range was wide (1-53 cycles). Most of them (92%) had already received systemic therapy, including a tyrosine kinase inhibitor and/or radioactive iodine.
The most common side effect was hypertension, which occurred in 75% patients, and was severe in 23%. Of those with a severe reaction, 53% required a new prescription for an antihypertensive medication. No one left the study or required a pazopanib dose reduction because of a blood pressure elevation, however. “We responded with a aggressive treatment, but it was a prominent issue,” Dr. Bible said.
Other adverse events were fatigue (83%; 8% severe); decrease in neutrophils (47%; 8% severe); diarrhea (78%; 7% severe); hand-foot syndrome (17%; 7% severe); and elevations of liver enzymes (53%; 6% severe). There were no deaths related to the study drug.
Partial RECIST responses occurred in 22 patients (37%). Thyroglobulin change did not differ by response after cycle 1, although its nadir was lower among patients who attained a partial response than among those who maintained disease stability (–87% vs. –69%).
“There was this correlation of nadir with maximum RECIST response, but this occurred in parallel with the response, so it was not capable of providing a prediction of response,” Dr. Bible said.
Prior therapy also was not a response predictor, he added.
Genomic profiling was available for 16 patients; of these, 11 had mutations of BRAF, p53, JAK3 or HRAS. Thyroglobulin change and response to treatment was not significantly correlated with any of these mutations. Nor did it correlate with any type of prior tumor therapy.
The finding that pazopanib can benefit patients previously treated with a kinase inhibitor is an interesting one, Dr. Bible noted.
“Most kinase inhibitors are very promiscuous – they work on a number of pathways and have a footprint which is very messy. Most of them seem to have some activity in differentiated thyroid cancer, but we are still struggling to understand how that footprint varies. In theory they are all targeting VEGF receptors, but it’s striking that we can go from one kinase inhibitor to the next and still get a response.”
Dr. Bible had no financial declarations.
On Twitter @Alz_Gal
Lake BUENA VISTA, FLA. – Pazopanib is an effective therapy for advanced thyroid cancer, but at present, there seems to be no way to predict which patients will respond to it.
An investigation into the predictive value of thyroglobulin found that the level during treatment did change, but it did so in parallel with response; there was no way to use the protein to parse out which patients would do well, Dr. Keith Bible said at the International Thyroid Congress.
“We had hoped that it might be a predictor as early as 4 weeks, so we could assign patients into categories of response and perhaps stop treatment earlier,” said Dr. Bible of the Mayo Clinic, Rochester, Minn. “Unfortunately, there was no way to do that.”
Pazopanib (Votrient) is a tyrosine kinase inhibitor of vascular endothelial growth factor receptors. It is approved for advanced soft tissue sarcoma and advanced renal cell carcinoma. The Mayo Clinic Consortium has been investigating pazopanib in phase II trials for advanced differentiated and medullary thyroid cancers. In a 2010 report, it was shown to induce at least a partial response in about half of the patients who received it (Lancet Oncol. 2010 Oct;11[10]:962-72).
Last year, it was also shown to be effective in advanced medullary thyroid cancer. Five of 35 patients attained partial Response Evaluation Criteria In Solid Tumors (RECIST) responses (14%) (J Clin Endocrinol Metab. 2014 May;99[5]:1687-93).
The drug has not been successful in treating advanced anaplastic thyroid cancer, however.
Because of pazopanib’s proclivity to induce sometimes-severe hypertension, investigators were hoping for some way to stratify potential responders. Thyroglobulin levels could be one marker, Dr. Bible said at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
He and the consortium investigators examined how thyroglobulin levels correlated with RECIST scores in 60 patients with metastatic differentiated thyroid cancers. The patients received a median of 10 cycles, but the range was wide (1-53 cycles). Most of them (92%) had already received systemic therapy, including a tyrosine kinase inhibitor and/or radioactive iodine.
The most common side effect was hypertension, which occurred in 75% patients, and was severe in 23%. Of those with a severe reaction, 53% required a new prescription for an antihypertensive medication. No one left the study or required a pazopanib dose reduction because of a blood pressure elevation, however. “We responded with a aggressive treatment, but it was a prominent issue,” Dr. Bible said.
Other adverse events were fatigue (83%; 8% severe); decrease in neutrophils (47%; 8% severe); diarrhea (78%; 7% severe); hand-foot syndrome (17%; 7% severe); and elevations of liver enzymes (53%; 6% severe). There were no deaths related to the study drug.
Partial RECIST responses occurred in 22 patients (37%). Thyroglobulin change did not differ by response after cycle 1, although its nadir was lower among patients who attained a partial response than among those who maintained disease stability (–87% vs. –69%).
“There was this correlation of nadir with maximum RECIST response, but this occurred in parallel with the response, so it was not capable of providing a prediction of response,” Dr. Bible said.
Prior therapy also was not a response predictor, he added.
Genomic profiling was available for 16 patients; of these, 11 had mutations of BRAF, p53, JAK3 or HRAS. Thyroglobulin change and response to treatment was not significantly correlated with any of these mutations. Nor did it correlate with any type of prior tumor therapy.
The finding that pazopanib can benefit patients previously treated with a kinase inhibitor is an interesting one, Dr. Bible noted.
“Most kinase inhibitors are very promiscuous – they work on a number of pathways and have a footprint which is very messy. Most of them seem to have some activity in differentiated thyroid cancer, but we are still struggling to understand how that footprint varies. In theory they are all targeting VEGF receptors, but it’s striking that we can go from one kinase inhibitor to the next and still get a response.”
Dr. Bible had no financial declarations.
On Twitter @Alz_Gal
Lake BUENA VISTA, FLA. – Pazopanib is an effective therapy for advanced thyroid cancer, but at present, there seems to be no way to predict which patients will respond to it.
An investigation into the predictive value of thyroglobulin found that the level during treatment did change, but it did so in parallel with response; there was no way to use the protein to parse out which patients would do well, Dr. Keith Bible said at the International Thyroid Congress.
“We had hoped that it might be a predictor as early as 4 weeks, so we could assign patients into categories of response and perhaps stop treatment earlier,” said Dr. Bible of the Mayo Clinic, Rochester, Minn. “Unfortunately, there was no way to do that.”
Pazopanib (Votrient) is a tyrosine kinase inhibitor of vascular endothelial growth factor receptors. It is approved for advanced soft tissue sarcoma and advanced renal cell carcinoma. The Mayo Clinic Consortium has been investigating pazopanib in phase II trials for advanced differentiated and medullary thyroid cancers. In a 2010 report, it was shown to induce at least a partial response in about half of the patients who received it (Lancet Oncol. 2010 Oct;11[10]:962-72).
Last year, it was also shown to be effective in advanced medullary thyroid cancer. Five of 35 patients attained partial Response Evaluation Criteria In Solid Tumors (RECIST) responses (14%) (J Clin Endocrinol Metab. 2014 May;99[5]:1687-93).
The drug has not been successful in treating advanced anaplastic thyroid cancer, however.
Because of pazopanib’s proclivity to induce sometimes-severe hypertension, investigators were hoping for some way to stratify potential responders. Thyroglobulin levels could be one marker, Dr. Bible said at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association, European Thyroid Association, and Latin American Thyroid Society.
He and the consortium investigators examined how thyroglobulin levels correlated with RECIST scores in 60 patients with metastatic differentiated thyroid cancers. The patients received a median of 10 cycles, but the range was wide (1-53 cycles). Most of them (92%) had already received systemic therapy, including a tyrosine kinase inhibitor and/or radioactive iodine.
The most common side effect was hypertension, which occurred in 75% patients, and was severe in 23%. Of those with a severe reaction, 53% required a new prescription for an antihypertensive medication. No one left the study or required a pazopanib dose reduction because of a blood pressure elevation, however. “We responded with a aggressive treatment, but it was a prominent issue,” Dr. Bible said.
Other adverse events were fatigue (83%; 8% severe); decrease in neutrophils (47%; 8% severe); diarrhea (78%; 7% severe); hand-foot syndrome (17%; 7% severe); and elevations of liver enzymes (53%; 6% severe). There were no deaths related to the study drug.
Partial RECIST responses occurred in 22 patients (37%). Thyroglobulin change did not differ by response after cycle 1, although its nadir was lower among patients who attained a partial response than among those who maintained disease stability (–87% vs. –69%).
“There was this correlation of nadir with maximum RECIST response, but this occurred in parallel with the response, so it was not capable of providing a prediction of response,” Dr. Bible said.
Prior therapy also was not a response predictor, he added.
Genomic profiling was available for 16 patients; of these, 11 had mutations of BRAF, p53, JAK3 or HRAS. Thyroglobulin change and response to treatment was not significantly correlated with any of these mutations. Nor did it correlate with any type of prior tumor therapy.
The finding that pazopanib can benefit patients previously treated with a kinase inhibitor is an interesting one, Dr. Bible noted.
“Most kinase inhibitors are very promiscuous – they work on a number of pathways and have a footprint which is very messy. Most of them seem to have some activity in differentiated thyroid cancer, but we are still struggling to understand how that footprint varies. In theory they are all targeting VEGF receptors, but it’s striking that we can go from one kinase inhibitor to the next and still get a response.”
Dr. Bible had no financial declarations.
On Twitter @Alz_Gal
AT ITC 2015
Key clinical point: Falling thyroglobulin levels cannot predict early response to pazopanib in differentiated thyroid cancers.
Major finding: There was a 37% response rate for patients taking pazopanib for differentiated thyroid cancer, but thyroglobulin decline was not an early predictor of response.
Data source: A prospective study of 60 patients.
Disclosures: Dr. Bible had no financial disclosures.
Cabozantinib boosts survival for some patients with RET+ medullary thyroid cancer
LAKE BUENA VISTA, Fla. – The tyrosine kinase inhibitor cabozantinib significantly extended overall survival for patients with RET-M918T–positive medullary thyroid cancer.
The placebo-controlled EXAM trial did not show any significant differences in overall survival among the entire group, Dr. Steven Sherman said at the International Thyroid Congress. But in a subgroup analysis, those with RET-M918T mutations who received the drug had the best outcomes by far, with a mean overall survival 2 years longer than that of similar patients who received placebo (44 vs. 19 months; HR 0.60; P = .026).
Cabozantinib (COMETRIQ) delivers a three-way punch to thyroid tumors, said Dr. Sherman of MD Anderson Cancer Center, Houston. It inhibits tyrosine kinase, vascular endothelial growth factor receptors, and mutant RET. But EXAM does not provide any clues about the exact mechanism by which it subdued this particular class of tumors.
“We may well be targeting mutant RET as well as tyrosine kinase and VEGF-R. But the other possibility is that these RET tumors are highly dependent on VEGF for angiogenesis and that inhibiting that is the key element. The data from this study don’t allow us to speculate on which is the most important,” said Dr. Sherman.
The 2-year trial randomized 330 patients with progressive medullary thyroid cancer to cabozantinib (140 mg per day) or placebo. If patients taking placebo progressed, they were not allowed to cross over to cabozantimib. The primary endpoint was progression-free survival (PFS); results were published in 2013 in the Journal of Clinical Oncology (doi: 10.1200/JCO.2012.48.4659).
Dr. Sherman presented the final results on overall survival at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
Almost half of the cohort had received prior therapy, and 21%, a prior tyrosine kinase inhibitor. About half had RET mutations; 12% were RET negative, and genomic status was unknown in the remainder. M918T was the predominant RET mutation (74%). The main sites of metastasis were lymph nodes, liver, lung, and bone.
PFS was significantly better in those taking the study drug (11 vs. 4 months; HR 0.28; P equal to or less than .01). Significant PFS benefits were seen regardless of age, prior treatment, and RET status.
In the overall survival analysis, however, cabozantinib conferred no significant benefit over placebo across the entire cohort (26.6 vs 21 months; HR 0.85; P = .24).
RET mutation profiles were available for 65% of patients. The drug did not confer significant overall survival benefit to all those with RET mutations (31.6 vs. 24.8 months; HR 0.79; P = .24). However, those with the M918T mutation who took cabozantinib survived significantly longer than did those who took placebo.
A small number of patients (16) had RAS mutations, and those who took cabozantinib did have better overall survival than did those who took placebo, although the difference was not statistically significant.
Adverse events were more common in the cabozantinib arm and included pneumonia (4.2%), pulmonary embolism (3.3%), mucosal inflammation (2.8%), hypocalcemia (2.8%), hypertension, dysphagia, dehydration, and lung abscess (2.3% each).
The EXAM study was sponsored by Exelixis. Dr. Sherman had no financial disclosures.
On Twitter @Alz_Gal
LAKE BUENA VISTA, Fla. – The tyrosine kinase inhibitor cabozantinib significantly extended overall survival for patients with RET-M918T–positive medullary thyroid cancer.
The placebo-controlled EXAM trial did not show any significant differences in overall survival among the entire group, Dr. Steven Sherman said at the International Thyroid Congress. But in a subgroup analysis, those with RET-M918T mutations who received the drug had the best outcomes by far, with a mean overall survival 2 years longer than that of similar patients who received placebo (44 vs. 19 months; HR 0.60; P = .026).
Cabozantinib (COMETRIQ) delivers a three-way punch to thyroid tumors, said Dr. Sherman of MD Anderson Cancer Center, Houston. It inhibits tyrosine kinase, vascular endothelial growth factor receptors, and mutant RET. But EXAM does not provide any clues about the exact mechanism by which it subdued this particular class of tumors.
“We may well be targeting mutant RET as well as tyrosine kinase and VEGF-R. But the other possibility is that these RET tumors are highly dependent on VEGF for angiogenesis and that inhibiting that is the key element. The data from this study don’t allow us to speculate on which is the most important,” said Dr. Sherman.
The 2-year trial randomized 330 patients with progressive medullary thyroid cancer to cabozantinib (140 mg per day) or placebo. If patients taking placebo progressed, they were not allowed to cross over to cabozantimib. The primary endpoint was progression-free survival (PFS); results were published in 2013 in the Journal of Clinical Oncology (doi: 10.1200/JCO.2012.48.4659).
Dr. Sherman presented the final results on overall survival at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
Almost half of the cohort had received prior therapy, and 21%, a prior tyrosine kinase inhibitor. About half had RET mutations; 12% were RET negative, and genomic status was unknown in the remainder. M918T was the predominant RET mutation (74%). The main sites of metastasis were lymph nodes, liver, lung, and bone.
PFS was significantly better in those taking the study drug (11 vs. 4 months; HR 0.28; P equal to or less than .01). Significant PFS benefits were seen regardless of age, prior treatment, and RET status.
In the overall survival analysis, however, cabozantinib conferred no significant benefit over placebo across the entire cohort (26.6 vs 21 months; HR 0.85; P = .24).
RET mutation profiles were available for 65% of patients. The drug did not confer significant overall survival benefit to all those with RET mutations (31.6 vs. 24.8 months; HR 0.79; P = .24). However, those with the M918T mutation who took cabozantinib survived significantly longer than did those who took placebo.
A small number of patients (16) had RAS mutations, and those who took cabozantinib did have better overall survival than did those who took placebo, although the difference was not statistically significant.
Adverse events were more common in the cabozantinib arm and included pneumonia (4.2%), pulmonary embolism (3.3%), mucosal inflammation (2.8%), hypocalcemia (2.8%), hypertension, dysphagia, dehydration, and lung abscess (2.3% each).
The EXAM study was sponsored by Exelixis. Dr. Sherman had no financial disclosures.
On Twitter @Alz_Gal
LAKE BUENA VISTA, Fla. – The tyrosine kinase inhibitor cabozantinib significantly extended overall survival for patients with RET-M918T–positive medullary thyroid cancer.
The placebo-controlled EXAM trial did not show any significant differences in overall survival among the entire group, Dr. Steven Sherman said at the International Thyroid Congress. But in a subgroup analysis, those with RET-M918T mutations who received the drug had the best outcomes by far, with a mean overall survival 2 years longer than that of similar patients who received placebo (44 vs. 19 months; HR 0.60; P = .026).
Cabozantinib (COMETRIQ) delivers a three-way punch to thyroid tumors, said Dr. Sherman of MD Anderson Cancer Center, Houston. It inhibits tyrosine kinase, vascular endothelial growth factor receptors, and mutant RET. But EXAM does not provide any clues about the exact mechanism by which it subdued this particular class of tumors.
“We may well be targeting mutant RET as well as tyrosine kinase and VEGF-R. But the other possibility is that these RET tumors are highly dependent on VEGF for angiogenesis and that inhibiting that is the key element. The data from this study don’t allow us to speculate on which is the most important,” said Dr. Sherman.
The 2-year trial randomized 330 patients with progressive medullary thyroid cancer to cabozantinib (140 mg per day) or placebo. If patients taking placebo progressed, they were not allowed to cross over to cabozantimib. The primary endpoint was progression-free survival (PFS); results were published in 2013 in the Journal of Clinical Oncology (doi: 10.1200/JCO.2012.48.4659).
Dr. Sherman presented the final results on overall survival at the meeting, which was held by the American Thyroid Association, Asia-Oceania Thyroid Association , European Thyroid Association, and Latin American Thyroid Society.
Almost half of the cohort had received prior therapy, and 21%, a prior tyrosine kinase inhibitor. About half had RET mutations; 12% were RET negative, and genomic status was unknown in the remainder. M918T was the predominant RET mutation (74%). The main sites of metastasis were lymph nodes, liver, lung, and bone.
PFS was significantly better in those taking the study drug (11 vs. 4 months; HR 0.28; P equal to or less than .01). Significant PFS benefits were seen regardless of age, prior treatment, and RET status.
In the overall survival analysis, however, cabozantinib conferred no significant benefit over placebo across the entire cohort (26.6 vs 21 months; HR 0.85; P = .24).
RET mutation profiles were available for 65% of patients. The drug did not confer significant overall survival benefit to all those with RET mutations (31.6 vs. 24.8 months; HR 0.79; P = .24). However, those with the M918T mutation who took cabozantinib survived significantly longer than did those who took placebo.
A small number of patients (16) had RAS mutations, and those who took cabozantinib did have better overall survival than did those who took placebo, although the difference was not statistically significant.
Adverse events were more common in the cabozantinib arm and included pneumonia (4.2%), pulmonary embolism (3.3%), mucosal inflammation (2.8%), hypocalcemia (2.8%), hypertension, dysphagia, dehydration, and lung abscess (2.3% each).
The EXAM study was sponsored by Exelixis. Dr. Sherman had no financial disclosures.
On Twitter @Alz_Gal
AT ITC 2015
Key clinical point: Cabozantinib improved overall survival in RET-M918T–positive progressive medullary thyroid cancers.
Major finding: Compared to placebo, cabozantinib conferred a 2-year overall survival advantage upon patients with RET-M918T–positive medullary thyroid tumors.
Data source: The EXAM study, which randomized 330 patients to cabozantinib or placebo for 24 months.
Disclosures: Exelixis sponsored the study. Dr. Sherman had no financial disclosures.
Ocrelizumab decreases relapses, brain lesion development in relapsing-remitting MS
Ocrelizumab reduced both relapses and disability progression to a significantly greater extent than did interferon beta-1a in patients with relapsing-remitting multiple sclerosis in two large phase III trials.
The selective B-cell–targeting monoclonal antibody cut the annualized relapse rate by almost half when compared against interferon beta-1a and significantly reduced the risk of confirmed disability progression at both 12 and 24 weeks, Dr. Stephen Hauser reported at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis in Barcelona.
Ocrelizumab, a humanized monoclonal antibody that selectively targets CD20-positive B cells, succeeded on secondary outcomes as well, reducing T1 gadolinium-enhancing lesions by more than 90%, and the emergence or enhancement of T2 hyperintense lesions by 80%, said Dr. Hauser, chair of neurology at the University of California, San Francisco.
The data are from two identically designed phase III trials conducted by Genentech. OPERA 1 and OPERA 2 enrolled 1,656 patients with relapsing-remitting multiple sclerosis (RRMS). They were randomized to either 600-mg ocrelizumab IV infusions every 24 weeks plus placebo subcutaneous injections three times weekly or to 44-mcg subcutaneous injections of interferon beta-1a (Rebif) thrice weekly plus placebo IV infusions every 24 weeks. Both studies were conducted for 96 weeks.
The patients were a typical RRMS cohort: young (mean 37 years) and mostly women (66%). The mean baseline score on the Extended Disability Status Scale was 2.77 and the mean time since diagnosis about 4 years. Patients had about 1.5 relapses in the first and second year before entering the studies; about one-third (595) were naive to treatment.
Compared with interferon beta-1a, ocrelizumab reduced the annualized relapse rate by 46% in OPERA 1 and 47% in OPERA 2. It also posted 43% and 37% risk reductions in confirmed disability progression sustained for both 12 and 24 weeks.
On imaging measures, ocrelizumab bested interferon beta-1a as well. It conferred a 94% and 95% reduction in the total number of T1 gadolinium-enhancing lesions and a 77% and 83% reduction in the total number of new and/or enlarging hyperintense T2 lesions. An exploratory analysis suggested that the drug reduced the rate of whole brain volume, compared with interferon beta-1a.
A pooled safety analysis comprised 83% of the entire study population. Adverse events were similar in both treatment groups overall. Infusion reactions were significantly more common with ocrelizumab than with interferon beta-1a (34% vs. 9.7%). There were similar rates of serious adverse events, including serious infections (6.9% and 8.7%), and there were no cases of progressive multifocal leukoencephalopathy.
Long-term safety investigations are still ongoing, Genentech’s Dr. Peter Chin said in an interview. Ocrelizumab was originally developed by Roche and Biogen for rheumatoid arthritis. But in 2010, the companies halted development of the drug when an interim safety analysis found a significantly increased incidence of serious infections.
While Genentech will keep monitoring for those problems, they may be less likely to develop in MS patients than in patients with RA, said Dr. Chin, principal medical director for Genentech-Roche Global Neuroscience Development.
“Patients with RRMS are typically younger and, overall, healthier, than those with RA, without many of the comorbidities that predispose them to opportunistic infections,” he said. “They also tend to be taking concurrent immunosuppressive drugs, which increases their risk in a way we wouldn’t see in most MS patients.”
Dr. Hauser noted that he has received financial support from F. Hoffmann-La Roche, Symbiotix, and Bionure.
Ocrelizumab reduced both relapses and disability progression to a significantly greater extent than did interferon beta-1a in patients with relapsing-remitting multiple sclerosis in two large phase III trials.
The selective B-cell–targeting monoclonal antibody cut the annualized relapse rate by almost half when compared against interferon beta-1a and significantly reduced the risk of confirmed disability progression at both 12 and 24 weeks, Dr. Stephen Hauser reported at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis in Barcelona.
Ocrelizumab, a humanized monoclonal antibody that selectively targets CD20-positive B cells, succeeded on secondary outcomes as well, reducing T1 gadolinium-enhancing lesions by more than 90%, and the emergence or enhancement of T2 hyperintense lesions by 80%, said Dr. Hauser, chair of neurology at the University of California, San Francisco.
The data are from two identically designed phase III trials conducted by Genentech. OPERA 1 and OPERA 2 enrolled 1,656 patients with relapsing-remitting multiple sclerosis (RRMS). They were randomized to either 600-mg ocrelizumab IV infusions every 24 weeks plus placebo subcutaneous injections three times weekly or to 44-mcg subcutaneous injections of interferon beta-1a (Rebif) thrice weekly plus placebo IV infusions every 24 weeks. Both studies were conducted for 96 weeks.
The patients were a typical RRMS cohort: young (mean 37 years) and mostly women (66%). The mean baseline score on the Extended Disability Status Scale was 2.77 and the mean time since diagnosis about 4 years. Patients had about 1.5 relapses in the first and second year before entering the studies; about one-third (595) were naive to treatment.
Compared with interferon beta-1a, ocrelizumab reduced the annualized relapse rate by 46% in OPERA 1 and 47% in OPERA 2. It also posted 43% and 37% risk reductions in confirmed disability progression sustained for both 12 and 24 weeks.
On imaging measures, ocrelizumab bested interferon beta-1a as well. It conferred a 94% and 95% reduction in the total number of T1 gadolinium-enhancing lesions and a 77% and 83% reduction in the total number of new and/or enlarging hyperintense T2 lesions. An exploratory analysis suggested that the drug reduced the rate of whole brain volume, compared with interferon beta-1a.
A pooled safety analysis comprised 83% of the entire study population. Adverse events were similar in both treatment groups overall. Infusion reactions were significantly more common with ocrelizumab than with interferon beta-1a (34% vs. 9.7%). There were similar rates of serious adverse events, including serious infections (6.9% and 8.7%), and there were no cases of progressive multifocal leukoencephalopathy.
Long-term safety investigations are still ongoing, Genentech’s Dr. Peter Chin said in an interview. Ocrelizumab was originally developed by Roche and Biogen for rheumatoid arthritis. But in 2010, the companies halted development of the drug when an interim safety analysis found a significantly increased incidence of serious infections.
While Genentech will keep monitoring for those problems, they may be less likely to develop in MS patients than in patients with RA, said Dr. Chin, principal medical director for Genentech-Roche Global Neuroscience Development.
“Patients with RRMS are typically younger and, overall, healthier, than those with RA, without many of the comorbidities that predispose them to opportunistic infections,” he said. “They also tend to be taking concurrent immunosuppressive drugs, which increases their risk in a way we wouldn’t see in most MS patients.”
Dr. Hauser noted that he has received financial support from F. Hoffmann-La Roche, Symbiotix, and Bionure.
Ocrelizumab reduced both relapses and disability progression to a significantly greater extent than did interferon beta-1a in patients with relapsing-remitting multiple sclerosis in two large phase III trials.
The selective B-cell–targeting monoclonal antibody cut the annualized relapse rate by almost half when compared against interferon beta-1a and significantly reduced the risk of confirmed disability progression at both 12 and 24 weeks, Dr. Stephen Hauser reported at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis in Barcelona.
Ocrelizumab, a humanized monoclonal antibody that selectively targets CD20-positive B cells, succeeded on secondary outcomes as well, reducing T1 gadolinium-enhancing lesions by more than 90%, and the emergence or enhancement of T2 hyperintense lesions by 80%, said Dr. Hauser, chair of neurology at the University of California, San Francisco.
The data are from two identically designed phase III trials conducted by Genentech. OPERA 1 and OPERA 2 enrolled 1,656 patients with relapsing-remitting multiple sclerosis (RRMS). They were randomized to either 600-mg ocrelizumab IV infusions every 24 weeks plus placebo subcutaneous injections three times weekly or to 44-mcg subcutaneous injections of interferon beta-1a (Rebif) thrice weekly plus placebo IV infusions every 24 weeks. Both studies were conducted for 96 weeks.
The patients were a typical RRMS cohort: young (mean 37 years) and mostly women (66%). The mean baseline score on the Extended Disability Status Scale was 2.77 and the mean time since diagnosis about 4 years. Patients had about 1.5 relapses in the first and second year before entering the studies; about one-third (595) were naive to treatment.
Compared with interferon beta-1a, ocrelizumab reduced the annualized relapse rate by 46% in OPERA 1 and 47% in OPERA 2. It also posted 43% and 37% risk reductions in confirmed disability progression sustained for both 12 and 24 weeks.
On imaging measures, ocrelizumab bested interferon beta-1a as well. It conferred a 94% and 95% reduction in the total number of T1 gadolinium-enhancing lesions and a 77% and 83% reduction in the total number of new and/or enlarging hyperintense T2 lesions. An exploratory analysis suggested that the drug reduced the rate of whole brain volume, compared with interferon beta-1a.
A pooled safety analysis comprised 83% of the entire study population. Adverse events were similar in both treatment groups overall. Infusion reactions were significantly more common with ocrelizumab than with interferon beta-1a (34% vs. 9.7%). There were similar rates of serious adverse events, including serious infections (6.9% and 8.7%), and there were no cases of progressive multifocal leukoencephalopathy.
Long-term safety investigations are still ongoing, Genentech’s Dr. Peter Chin said in an interview. Ocrelizumab was originally developed by Roche and Biogen for rheumatoid arthritis. But in 2010, the companies halted development of the drug when an interim safety analysis found a significantly increased incidence of serious infections.
While Genentech will keep monitoring for those problems, they may be less likely to develop in MS patients than in patients with RA, said Dr. Chin, principal medical director for Genentech-Roche Global Neuroscience Development.
“Patients with RRMS are typically younger and, overall, healthier, than those with RA, without many of the comorbidities that predispose them to opportunistic infections,” he said. “They also tend to be taking concurrent immunosuppressive drugs, which increases their risk in a way we wouldn’t see in most MS patients.”
Dr. Hauser noted that he has received financial support from F. Hoffmann-La Roche, Symbiotix, and Bionure.
FROM ECTRIMS 2015
Key clinical point: Ocrelizumab decreased relapses and new lesions in relapsing remitting MS.
Major finding: Compared with interferon beta-1a, ocrelizumab nearly halved annualized relapse rates and decreased new lesions by 80%.
Data source: OPERA 1 and 2 randomized 1,656 patients to either ocrelizumab or interferon beta-1a.
Disclosures: Genentech (a subsidiary of Roche) sponsored the studies. Dr. Hauser noted that he has received financial support from F. Hoffmann-La Roche, Symbiotix, and Bionure.
For subacute STEMI, thrombectomy adds no benefit to PCI
The addition of thrombectomy to percutaneous coronary intervention didn’t improve cardiac flow or clinical endpoints any more than PCI alone in patients with a subacute ST-elevation myocardial infarction.
Thrombosis aspiration decreased microvascular obstruction less than 1% more than did PCI alone – 2.5% vs. 3% of the left ventricular mass, Dr. Steffan Desch said at the Transcatheter Cardiovascular Therapeutics annual meeting. There were also no significant differences in infarct size, myocardial salvage, or left ventricular ejection fraction, he said at the meeting sponsored by the Cardiovascular Research Foundation.
“Routine manual thrombectomy and aspiration didn’t show any significant reduction in microvascular obstruction on imaging,” said Dr. Desch of University Heart Center, Lübeck, Germany. “This finding was supported by a variety of secondary endpoints, none of them significantly beneficial.”
The study, which was simultaneously published online (JACC Cardiovasc Interv. 2015. doi: 10.1016/j.jcin.2015.09.010), gives the first firm evidence that clot removal is not particularly helpful to patients who present late after symptom onset. With a mean development time of 28 hours, thrombi in this cohort were more mature, with higher fibrin content than the typically soft material seen in patients with acute ST-elevation myocardial infarction (STEMI). When the clot becomes denser and more organized, it is likely less suitable for aspiration, he said.
The cohort comprised 152 patients who presented in a 12- to 48-hour window after onset of symptoms. They were randomized to either standard PCI or to clot aspiration followed by PCI. The primary outcome was the extent of major vessel occlusion on magnetic resonance imaging conducted 1-4 days after the intervention. Secondary outcomes included infarct size, myocardial salvage, and left ventricular volume and ejection fraction.
Patients were a mean of 66 years old, with typical baseline characteristics. Most were men; hypertension was common (about 70%). About half had signs of ongoing ischemia at admission. The door-to-balloon time was 78 minutes in the combination therapy group and 62 minutes in the PCI-only group. Most (62.5%) had a complete occlusion of the culprit vessel.
In addition to providing no benefit in microvascular occlusion, aspiration did not significantly improve TIMI flow grade above that achieved with PCI only. After the intervention, 78% of those in the thrombectomy group and 69% of those in the PCI group achieved a TIMI flow grade 3. Nor was there a significant difference in myocardial blush grade (70% vs. 65%). When troponin T values were used to assess enzymatic infarct size, they were similar in both groups at 24 and 48 hours.
Clinical outcomes were similar as well. All-cause mortality was 3% in the aspiration group and 5% in the PCI-only group; cardiovascular death occurred in 3% and 4%, respectively. There were no reinfarctions and no stent thromboses. One stroke occurred in a patient who underwent PCI alone.
Dr. Desch noted in the published article that the study took all comers, rather than selecting for specific patient characteristics. Therefore, he said “it is possible that thrombus aspiration might only be advantageous in specific subsets of patients such as those with large thrombus burden, total occlusion or reduced flow.”
The study was funded by a research grant from Medtronic. Dr. Desch reported grant/research support from Medtronic.
The addition of thrombectomy to percutaneous coronary intervention didn’t improve cardiac flow or clinical endpoints any more than PCI alone in patients with a subacute ST-elevation myocardial infarction.
Thrombosis aspiration decreased microvascular obstruction less than 1% more than did PCI alone – 2.5% vs. 3% of the left ventricular mass, Dr. Steffan Desch said at the Transcatheter Cardiovascular Therapeutics annual meeting. There were also no significant differences in infarct size, myocardial salvage, or left ventricular ejection fraction, he said at the meeting sponsored by the Cardiovascular Research Foundation.
“Routine manual thrombectomy and aspiration didn’t show any significant reduction in microvascular obstruction on imaging,” said Dr. Desch of University Heart Center, Lübeck, Germany. “This finding was supported by a variety of secondary endpoints, none of them significantly beneficial.”
The study, which was simultaneously published online (JACC Cardiovasc Interv. 2015. doi: 10.1016/j.jcin.2015.09.010), gives the first firm evidence that clot removal is not particularly helpful to patients who present late after symptom onset. With a mean development time of 28 hours, thrombi in this cohort were more mature, with higher fibrin content than the typically soft material seen in patients with acute ST-elevation myocardial infarction (STEMI). When the clot becomes denser and more organized, it is likely less suitable for aspiration, he said.
The cohort comprised 152 patients who presented in a 12- to 48-hour window after onset of symptoms. They were randomized to either standard PCI or to clot aspiration followed by PCI. The primary outcome was the extent of major vessel occlusion on magnetic resonance imaging conducted 1-4 days after the intervention. Secondary outcomes included infarct size, myocardial salvage, and left ventricular volume and ejection fraction.
Patients were a mean of 66 years old, with typical baseline characteristics. Most were men; hypertension was common (about 70%). About half had signs of ongoing ischemia at admission. The door-to-balloon time was 78 minutes in the combination therapy group and 62 minutes in the PCI-only group. Most (62.5%) had a complete occlusion of the culprit vessel.
In addition to providing no benefit in microvascular occlusion, aspiration did not significantly improve TIMI flow grade above that achieved with PCI only. After the intervention, 78% of those in the thrombectomy group and 69% of those in the PCI group achieved a TIMI flow grade 3. Nor was there a significant difference in myocardial blush grade (70% vs. 65%). When troponin T values were used to assess enzymatic infarct size, they were similar in both groups at 24 and 48 hours.
Clinical outcomes were similar as well. All-cause mortality was 3% in the aspiration group and 5% in the PCI-only group; cardiovascular death occurred in 3% and 4%, respectively. There were no reinfarctions and no stent thromboses. One stroke occurred in a patient who underwent PCI alone.
Dr. Desch noted in the published article that the study took all comers, rather than selecting for specific patient characteristics. Therefore, he said “it is possible that thrombus aspiration might only be advantageous in specific subsets of patients such as those with large thrombus burden, total occlusion or reduced flow.”
The study was funded by a research grant from Medtronic. Dr. Desch reported grant/research support from Medtronic.
The addition of thrombectomy to percutaneous coronary intervention didn’t improve cardiac flow or clinical endpoints any more than PCI alone in patients with a subacute ST-elevation myocardial infarction.
Thrombosis aspiration decreased microvascular obstruction less than 1% more than did PCI alone – 2.5% vs. 3% of the left ventricular mass, Dr. Steffan Desch said at the Transcatheter Cardiovascular Therapeutics annual meeting. There were also no significant differences in infarct size, myocardial salvage, or left ventricular ejection fraction, he said at the meeting sponsored by the Cardiovascular Research Foundation.
“Routine manual thrombectomy and aspiration didn’t show any significant reduction in microvascular obstruction on imaging,” said Dr. Desch of University Heart Center, Lübeck, Germany. “This finding was supported by a variety of secondary endpoints, none of them significantly beneficial.”
The study, which was simultaneously published online (JACC Cardiovasc Interv. 2015. doi: 10.1016/j.jcin.2015.09.010), gives the first firm evidence that clot removal is not particularly helpful to patients who present late after symptom onset. With a mean development time of 28 hours, thrombi in this cohort were more mature, with higher fibrin content than the typically soft material seen in patients with acute ST-elevation myocardial infarction (STEMI). When the clot becomes denser and more organized, it is likely less suitable for aspiration, he said.
The cohort comprised 152 patients who presented in a 12- to 48-hour window after onset of symptoms. They were randomized to either standard PCI or to clot aspiration followed by PCI. The primary outcome was the extent of major vessel occlusion on magnetic resonance imaging conducted 1-4 days after the intervention. Secondary outcomes included infarct size, myocardial salvage, and left ventricular volume and ejection fraction.
Patients were a mean of 66 years old, with typical baseline characteristics. Most were men; hypertension was common (about 70%). About half had signs of ongoing ischemia at admission. The door-to-balloon time was 78 minutes in the combination therapy group and 62 minutes in the PCI-only group. Most (62.5%) had a complete occlusion of the culprit vessel.
In addition to providing no benefit in microvascular occlusion, aspiration did not significantly improve TIMI flow grade above that achieved with PCI only. After the intervention, 78% of those in the thrombectomy group and 69% of those in the PCI group achieved a TIMI flow grade 3. Nor was there a significant difference in myocardial blush grade (70% vs. 65%). When troponin T values were used to assess enzymatic infarct size, they were similar in both groups at 24 and 48 hours.
Clinical outcomes were similar as well. All-cause mortality was 3% in the aspiration group and 5% in the PCI-only group; cardiovascular death occurred in 3% and 4%, respectively. There were no reinfarctions and no stent thromboses. One stroke occurred in a patient who underwent PCI alone.
Dr. Desch noted in the published article that the study took all comers, rather than selecting for specific patient characteristics. Therefore, he said “it is possible that thrombus aspiration might only be advantageous in specific subsets of patients such as those with large thrombus burden, total occlusion or reduced flow.”
The study was funded by a research grant from Medtronic. Dr. Desch reported grant/research support from Medtronic.
AT TCT 2015
Key clinical point: Thrombus aspiration doesn’t reduce microvascular obstruction in subacute STEMI patients undergoing PCI late after symptom onset.
Major finding: Thrombosis aspiration improved microvascular obstruction less than 1% more than did PCI alone – 2.5% vs. 3% of the left ventricular mass – and conferred no other indications of clinical benefit.
Data source: The study randomized 152 patients with late-presentation STEMI to PCI alone or to thrombectomy plus PCI.
Disclosures: The study was funded by a research grant from Medtronic. Dr. Desch reported grant/research support from Medtronic.
TCT: Routine thrombectomy with PCI raises stroke risk 66% in STEMI patients
Routine manual thrombectomy during percutaneous coronary intervention offers no significant long-term benefit over PCI alone for patients with acute ST-segment–elevation myocardial infarction.
In fact, thrombectomy was associated with a 66% increase in stroke risk in the year following a heart attack, Dr. Sanjit Jolly reported at the Transcatheter Cardiovascular Therapeutics annual meeting.
A 1-year analysis of the TOTAL trial confirmed the study’s recently published 1-month findings: Routine thrombectomy isn’t any better than PCI alone in the clinical outcomes of cardiovascular death, myocardial infarction, cardiogenic shock, or heart failure.
As it was at 1 month, however, stroke at 1 year was significantly more common; it occurred in 60 patients (1.2%) in the thrombectomy group and 36 (0.7%) in the PCI-alone group, for an increase in risk of 66% (P = .015).
The findings were simultaneously published online (Lancet 2015 Oct 13 doi: 10.1016/ S0140-6736[15]00448-1).
“Based on these endpoints, manual thrombectomy can no longer be recommended as a primary strategy in these patients,” Dr. Jolly said at the meeting, which was sponsored by the Cardiovascular Research Foundation.
Despite the unequivocal findings, it’s unclear when clinicians will completely embrace TOTAL’s results, noted Dr. Jolly of McMaster University, Hamilton, Ont.
“Unfortunately, practice has not changed with these findings. It often takes time for new evidence to get translated into the clinic, although some of my colleagues have told me that the results are giving them pause, causing them to use less thrombus aspiration. But interventional cardiologists are very visually driven. If we see something, we want to take it out. So it may take some time for these to be adopted,” he said.
The Thromwbectomy With PCI Versus PCI Alone in Patients With STEMI (TOTAL) trial randomized 10,723 patients to percutaneous coronary intervention with or without routine manual thrombectomy. The primary outcome was a composite of death from cardiovascular causes, recurrent myocardial infarction, cardiogenic shock, or New York Heart Association class IV heart failure within 180 days. The key safety outcome was stroke within 30 days.
The TOTAL results showed no difference in the primary outcome between thrombectomy and PCI patients (6.9% vs. 7%) (N Engl J Med. 2015 Apr 9;372[15]:1389-98).
Rates of cardiovascular death were similar (3% in each group) as were those for a combination of the primary outcome plus stent thrombosis or revascularization (10% in each group). Patients who had a thrombectomy were twice as likely to have a stroke within 30 days, although the absolute numbers were small (0.7% vs. 0.3%; HR, 2.06).
The 1-year follow-up study provided important perspective about the long-term risks and benefits of the two strategies.
There was no difference in the rate of the composite endpoint, which occurred in 7.8% of each group. Cardiovascular death occurred in 3.6% of the thrombectomy group and 3.8% of the PCI-alone group. There were similar rates of recurrent heart attack (2.5% vs. 2.3%), cardiogenic shock (1.9% vs. 2.1%), and class IV heart failure (2.1% vs. 1.9%).
The finding of significantly elevated stroke risk at 30 days was also present at 1 year, occurring in 1.2% of the thrombectomy patients and 0.7% of the PCI-alone patients (HR, 1.66; P = .015). The risk of a combination of stroke or transient ischemic attack was also increased significantly, by 65%, occurring in 1.4% and 0.9%, respectively (HR, 1.65; P = .008).
To further confirm the findings, Dr. Jolly also presented a meta-analysis of all-cause mortality in 20,352 patients involved in 16 studies comparing PCI plus thrombectomy with PCI alone. The meta-analysis also found that routine thrombectomy conferred no mortality benefit over PCI.
It did, however, confirm a 43% increased risk of stroke in PCI plus thrombectomy vs. PCI alone, at 0.9% and 0.6% (P = .03).
TOTAL was funded by the Canadian Institutes of Health Research, the Canadian Network and Centre for Trials Internationally, and Medtronic. Dr. Jolly received grants from Medtronic during the study.
On Twitter @Alz_Gal
Routine manual thrombectomy during percutaneous coronary intervention offers no significant long-term benefit over PCI alone for patients with acute ST-segment–elevation myocardial infarction.
In fact, thrombectomy was associated with a 66% increase in stroke risk in the year following a heart attack, Dr. Sanjit Jolly reported at the Transcatheter Cardiovascular Therapeutics annual meeting.
A 1-year analysis of the TOTAL trial confirmed the study’s recently published 1-month findings: Routine thrombectomy isn’t any better than PCI alone in the clinical outcomes of cardiovascular death, myocardial infarction, cardiogenic shock, or heart failure.
As it was at 1 month, however, stroke at 1 year was significantly more common; it occurred in 60 patients (1.2%) in the thrombectomy group and 36 (0.7%) in the PCI-alone group, for an increase in risk of 66% (P = .015).
The findings were simultaneously published online (Lancet 2015 Oct 13 doi: 10.1016/ S0140-6736[15]00448-1).
“Based on these endpoints, manual thrombectomy can no longer be recommended as a primary strategy in these patients,” Dr. Jolly said at the meeting, which was sponsored by the Cardiovascular Research Foundation.
Despite the unequivocal findings, it’s unclear when clinicians will completely embrace TOTAL’s results, noted Dr. Jolly of McMaster University, Hamilton, Ont.
“Unfortunately, practice has not changed with these findings. It often takes time for new evidence to get translated into the clinic, although some of my colleagues have told me that the results are giving them pause, causing them to use less thrombus aspiration. But interventional cardiologists are very visually driven. If we see something, we want to take it out. So it may take some time for these to be adopted,” he said.
The Thromwbectomy With PCI Versus PCI Alone in Patients With STEMI (TOTAL) trial randomized 10,723 patients to percutaneous coronary intervention with or without routine manual thrombectomy. The primary outcome was a composite of death from cardiovascular causes, recurrent myocardial infarction, cardiogenic shock, or New York Heart Association class IV heart failure within 180 days. The key safety outcome was stroke within 30 days.
The TOTAL results showed no difference in the primary outcome between thrombectomy and PCI patients (6.9% vs. 7%) (N Engl J Med. 2015 Apr 9;372[15]:1389-98).
Rates of cardiovascular death were similar (3% in each group) as were those for a combination of the primary outcome plus stent thrombosis or revascularization (10% in each group). Patients who had a thrombectomy were twice as likely to have a stroke within 30 days, although the absolute numbers were small (0.7% vs. 0.3%; HR, 2.06).
The 1-year follow-up study provided important perspective about the long-term risks and benefits of the two strategies.
There was no difference in the rate of the composite endpoint, which occurred in 7.8% of each group. Cardiovascular death occurred in 3.6% of the thrombectomy group and 3.8% of the PCI-alone group. There were similar rates of recurrent heart attack (2.5% vs. 2.3%), cardiogenic shock (1.9% vs. 2.1%), and class IV heart failure (2.1% vs. 1.9%).
The finding of significantly elevated stroke risk at 30 days was also present at 1 year, occurring in 1.2% of the thrombectomy patients and 0.7% of the PCI-alone patients (HR, 1.66; P = .015). The risk of a combination of stroke or transient ischemic attack was also increased significantly, by 65%, occurring in 1.4% and 0.9%, respectively (HR, 1.65; P = .008).
To further confirm the findings, Dr. Jolly also presented a meta-analysis of all-cause mortality in 20,352 patients involved in 16 studies comparing PCI plus thrombectomy with PCI alone. The meta-analysis also found that routine thrombectomy conferred no mortality benefit over PCI.
It did, however, confirm a 43% increased risk of stroke in PCI plus thrombectomy vs. PCI alone, at 0.9% and 0.6% (P = .03).
TOTAL was funded by the Canadian Institutes of Health Research, the Canadian Network and Centre for Trials Internationally, and Medtronic. Dr. Jolly received grants from Medtronic during the study.
On Twitter @Alz_Gal
Routine manual thrombectomy during percutaneous coronary intervention offers no significant long-term benefit over PCI alone for patients with acute ST-segment–elevation myocardial infarction.
In fact, thrombectomy was associated with a 66% increase in stroke risk in the year following a heart attack, Dr. Sanjit Jolly reported at the Transcatheter Cardiovascular Therapeutics annual meeting.
A 1-year analysis of the TOTAL trial confirmed the study’s recently published 1-month findings: Routine thrombectomy isn’t any better than PCI alone in the clinical outcomes of cardiovascular death, myocardial infarction, cardiogenic shock, or heart failure.
As it was at 1 month, however, stroke at 1 year was significantly more common; it occurred in 60 patients (1.2%) in the thrombectomy group and 36 (0.7%) in the PCI-alone group, for an increase in risk of 66% (P = .015).
The findings were simultaneously published online (Lancet 2015 Oct 13 doi: 10.1016/ S0140-6736[15]00448-1).
“Based on these endpoints, manual thrombectomy can no longer be recommended as a primary strategy in these patients,” Dr. Jolly said at the meeting, which was sponsored by the Cardiovascular Research Foundation.
Despite the unequivocal findings, it’s unclear when clinicians will completely embrace TOTAL’s results, noted Dr. Jolly of McMaster University, Hamilton, Ont.
“Unfortunately, practice has not changed with these findings. It often takes time for new evidence to get translated into the clinic, although some of my colleagues have told me that the results are giving them pause, causing them to use less thrombus aspiration. But interventional cardiologists are very visually driven. If we see something, we want to take it out. So it may take some time for these to be adopted,” he said.
The Thromwbectomy With PCI Versus PCI Alone in Patients With STEMI (TOTAL) trial randomized 10,723 patients to percutaneous coronary intervention with or without routine manual thrombectomy. The primary outcome was a composite of death from cardiovascular causes, recurrent myocardial infarction, cardiogenic shock, or New York Heart Association class IV heart failure within 180 days. The key safety outcome was stroke within 30 days.
The TOTAL results showed no difference in the primary outcome between thrombectomy and PCI patients (6.9% vs. 7%) (N Engl J Med. 2015 Apr 9;372[15]:1389-98).
Rates of cardiovascular death were similar (3% in each group) as were those for a combination of the primary outcome plus stent thrombosis or revascularization (10% in each group). Patients who had a thrombectomy were twice as likely to have a stroke within 30 days, although the absolute numbers were small (0.7% vs. 0.3%; HR, 2.06).
The 1-year follow-up study provided important perspective about the long-term risks and benefits of the two strategies.
There was no difference in the rate of the composite endpoint, which occurred in 7.8% of each group. Cardiovascular death occurred in 3.6% of the thrombectomy group and 3.8% of the PCI-alone group. There were similar rates of recurrent heart attack (2.5% vs. 2.3%), cardiogenic shock (1.9% vs. 2.1%), and class IV heart failure (2.1% vs. 1.9%).
The finding of significantly elevated stroke risk at 30 days was also present at 1 year, occurring in 1.2% of the thrombectomy patients and 0.7% of the PCI-alone patients (HR, 1.66; P = .015). The risk of a combination of stroke or transient ischemic attack was also increased significantly, by 65%, occurring in 1.4% and 0.9%, respectively (HR, 1.65; P = .008).
To further confirm the findings, Dr. Jolly also presented a meta-analysis of all-cause mortality in 20,352 patients involved in 16 studies comparing PCI plus thrombectomy with PCI alone. The meta-analysis also found that routine thrombectomy conferred no mortality benefit over PCI.
It did, however, confirm a 43% increased risk of stroke in PCI plus thrombectomy vs. PCI alone, at 0.9% and 0.6% (P = .03).
TOTAL was funded by the Canadian Institutes of Health Research, the Canadian Network and Centre for Trials Internationally, and Medtronic. Dr. Jolly received grants from Medtronic during the study.
On Twitter @Alz_Gal
AT TCT 2015
Key clinical point: Adding routine thrombectomy to PCI doesn’t improve outcomes; in fact, it increases the risk of stroke by 66% at 1 year.
Major finding: At 1 year, thrombectomy didn’t improve cardiovascular outcomes over PCI alone for patients with STEMI; in fact, it increased the risk of stroke by 66%.
Data source: The TOTAL trial randomizing 10,732 patients with STEMI to either PCI plus manual thrombectomy or PCI alone.
Disclosures: TOTAL was funded by the Canadian Institutes of Health Research, the Canadian Network and Centre for Trials Internationally, and Medtronic. Dr. Jolly received grants from Medtronic during the study.
Ocrelizumab posts first-ever positive outcome data for primary progressive MS
The selective B-cell–targeting monoclonal antibody ocrelizumab became the first drug to improve outcomes in primary progressive multiple sclerosis based on results from the randomized, placebo-controlled, phase III ORATORIO trial.
After 24 weeks of treatment, ocrelizumab reduced the risk of progression in clinical disability by 25%, compared with placebo. Over 2 years, the antibody also reduced the volume of hyperintense T2 lesions by 3.4%, whereas patients taking placebo experienced a 7% increase. The rate of whole brain volume loss was also significantly reduced.
The findings confer the first really good news in how to treat patients with this very difficult condition, said Dr. Xavier Montalbán, who presented the results at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis, held in Barcelona.
“We’ve been looking for something positive for these patients for years and have never found this,” said Dr. Montalbán, director of clinical neuroimmunology at Vall d’Hebron University Hospital, Barcelona. “We can discuss the magnitude of the effect, but I have to say I am very, very happy with this.”
The results are certainly encouraging, said Dr. Peter Calabresi, director of the Johns Hopkins Multiple Sclerosis Center and the division of neuroimmunology at The Johns Hopkins Hospital, Baltimore, although he tempered his enthusiasm a bit regarding patient selection and safety. In the entire cohort of 732 patients, 13 malignancies occurred over 3 years and occurred more than twice as often in the ocrelizumab arm than in the placebo arm (2.3% vs. 0.8%). These included four breast cancers in the active arm and none in the placebo arm.
“The data in general look very strong but with a possible safety signal related to cancers and deaths,” Dr. Calabresi said in an interview. “I’d need to see more detail in this regard, but overall I am very happy to see something work for primary progressive MS, where we have no approved drugs, so this is very important in that regard.
“As others have pointed out, though, the study was designed to target young people with inflammation (active MRIs) in which cases the drug works well. But many of our people with progressive MS are older and have no inflammation, so I’m guessing there is no effect in that group.”
Ocrelizumab, a humanized monoclonal antibody, selectively targets CD20-positive B cells. Since not all B cells are positive for the marker, ocrelizumab leaves intact the patient’s immune memory, as well as the ability to create more B cells. Its success bolsters the emerging idea that B cells may be even more important than T cells in the pathophysiology of MS.
ORATORIO was a 2:1 randomization of 732 patients with primary progressive MS to infusions of either 600 mg ocrelizumab or placebo every 24 weeks for 120 months.
Most patients finished the trial (80% active, 66% placebo). The withdrawals in the placebo arm were primarily due to lack of efficacy. Patients were a mean of 44 years old, with a mean 6.5 years since symptom onset. Most (88%) were treatment-naive. The Expanded Disability Status Score (EDSS) at baseline was 4.7 in both groups. At baseline, about a quarter of patients had gadolinium-enhancing lesions (mean number, 1) with a mean T2 lesion volume of 11 cm3, and their mean whole-brain volume was 1,467 cm3. All patients had either elevated immunoglobulin in cerebrospinal fluid or oligoclonal bands.
The primary endpoint of ORATORIO was 12-week confirmed disability progression (CDP). Secondary endpoints included 24-week CDP; timed 25-foot walk; and T2 lesion and whole brain volume.
By 120 weeks, patients taking the study drug experienced a mean 24% reduction in the risk of CDP (hazard ratio, 0.76; P = .0321). The treatment curves began to separate at week 24, and the differences in the risk of CDP were statistically significant at every evaluation. The benefit has been maintained in a safety evaluation that is extending treatment to 216 weeks in a subset of patients.
*Ocrelizumab significantly slowed the worsening of patients’ 25-foot walking time by 29% at 120 weeks when compared against placebo. Walking times increased by 39% from baseline on ocrelizumab, compared with a 55% rise for those on placebo (P = .0404).
On the imaging endpoints, the drug was associated with a highly significant difference in T2 lesion volume, decreasing the load by 3.4%, while the placebo arm experienced an increase of 7.4% from baseline (P = .0001).
Those taking the drug also experienced a small but significantly lower amount of whole-brain volume loss (–0.9% vs. –1.1%; P = .02).
Infusion reactions were most common with the first dose (27% active vs. 7% placebo). One patient taking the study drug withdrew at that time from a serious reaction, but the remainder were mild to moderate in severity. Reactions decreased over time, and all remained at the mild-moderate level.
Overall adverse events occurred at similar rates between those taking the drug and those taking placebo (95% vs. 90%). Infections were similar (70% vs. 68%) overall, although upper respiratory infections were more common among the active group (11% vs. 6%).
There were five deaths: one in the placebo arm (a motor vehicle accident) and four in the active arm (pulmonary embolism, pneumonia, pancreatic cancer, and pneumonia aspiration). These, as well as the malignancy findings, are still being evaluated, Dr. Montalbán said.
Based on the positive results, Genentech will be submitting the drug for approval in both the United States and Europe in the first quarter of 2016.
Dr. Montalbán disclosed ties with Genentech – the trial’s sponsor – and other pharmaceutical companies.
*Correction, 10/14/15: The timed 25-foot walk test results were previously misstated.
The selective B-cell–targeting monoclonal antibody ocrelizumab became the first drug to improve outcomes in primary progressive multiple sclerosis based on results from the randomized, placebo-controlled, phase III ORATORIO trial.
After 24 weeks of treatment, ocrelizumab reduced the risk of progression in clinical disability by 25%, compared with placebo. Over 2 years, the antibody also reduced the volume of hyperintense T2 lesions by 3.4%, whereas patients taking placebo experienced a 7% increase. The rate of whole brain volume loss was also significantly reduced.
The findings confer the first really good news in how to treat patients with this very difficult condition, said Dr. Xavier Montalbán, who presented the results at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis, held in Barcelona.
“We’ve been looking for something positive for these patients for years and have never found this,” said Dr. Montalbán, director of clinical neuroimmunology at Vall d’Hebron University Hospital, Barcelona. “We can discuss the magnitude of the effect, but I have to say I am very, very happy with this.”
The results are certainly encouraging, said Dr. Peter Calabresi, director of the Johns Hopkins Multiple Sclerosis Center and the division of neuroimmunology at The Johns Hopkins Hospital, Baltimore, although he tempered his enthusiasm a bit regarding patient selection and safety. In the entire cohort of 732 patients, 13 malignancies occurred over 3 years and occurred more than twice as often in the ocrelizumab arm than in the placebo arm (2.3% vs. 0.8%). These included four breast cancers in the active arm and none in the placebo arm.
“The data in general look very strong but with a possible safety signal related to cancers and deaths,” Dr. Calabresi said in an interview. “I’d need to see more detail in this regard, but overall I am very happy to see something work for primary progressive MS, where we have no approved drugs, so this is very important in that regard.
“As others have pointed out, though, the study was designed to target young people with inflammation (active MRIs) in which cases the drug works well. But many of our people with progressive MS are older and have no inflammation, so I’m guessing there is no effect in that group.”
Ocrelizumab, a humanized monoclonal antibody, selectively targets CD20-positive B cells. Since not all B cells are positive for the marker, ocrelizumab leaves intact the patient’s immune memory, as well as the ability to create more B cells. Its success bolsters the emerging idea that B cells may be even more important than T cells in the pathophysiology of MS.
ORATORIO was a 2:1 randomization of 732 patients with primary progressive MS to infusions of either 600 mg ocrelizumab or placebo every 24 weeks for 120 months.
Most patients finished the trial (80% active, 66% placebo). The withdrawals in the placebo arm were primarily due to lack of efficacy. Patients were a mean of 44 years old, with a mean 6.5 years since symptom onset. Most (88%) were treatment-naive. The Expanded Disability Status Score (EDSS) at baseline was 4.7 in both groups. At baseline, about a quarter of patients had gadolinium-enhancing lesions (mean number, 1) with a mean T2 lesion volume of 11 cm3, and their mean whole-brain volume was 1,467 cm3. All patients had either elevated immunoglobulin in cerebrospinal fluid or oligoclonal bands.
The primary endpoint of ORATORIO was 12-week confirmed disability progression (CDP). Secondary endpoints included 24-week CDP; timed 25-foot walk; and T2 lesion and whole brain volume.
By 120 weeks, patients taking the study drug experienced a mean 24% reduction in the risk of CDP (hazard ratio, 0.76; P = .0321). The treatment curves began to separate at week 24, and the differences in the risk of CDP were statistically significant at every evaluation. The benefit has been maintained in a safety evaluation that is extending treatment to 216 weeks in a subset of patients.
*Ocrelizumab significantly slowed the worsening of patients’ 25-foot walking time by 29% at 120 weeks when compared against placebo. Walking times increased by 39% from baseline on ocrelizumab, compared with a 55% rise for those on placebo (P = .0404).
On the imaging endpoints, the drug was associated with a highly significant difference in T2 lesion volume, decreasing the load by 3.4%, while the placebo arm experienced an increase of 7.4% from baseline (P = .0001).
Those taking the drug also experienced a small but significantly lower amount of whole-brain volume loss (–0.9% vs. –1.1%; P = .02).
Infusion reactions were most common with the first dose (27% active vs. 7% placebo). One patient taking the study drug withdrew at that time from a serious reaction, but the remainder were mild to moderate in severity. Reactions decreased over time, and all remained at the mild-moderate level.
Overall adverse events occurred at similar rates between those taking the drug and those taking placebo (95% vs. 90%). Infections were similar (70% vs. 68%) overall, although upper respiratory infections were more common among the active group (11% vs. 6%).
There were five deaths: one in the placebo arm (a motor vehicle accident) and four in the active arm (pulmonary embolism, pneumonia, pancreatic cancer, and pneumonia aspiration). These, as well as the malignancy findings, are still being evaluated, Dr. Montalbán said.
Based on the positive results, Genentech will be submitting the drug for approval in both the United States and Europe in the first quarter of 2016.
Dr. Montalbán disclosed ties with Genentech – the trial’s sponsor – and other pharmaceutical companies.
*Correction, 10/14/15: The timed 25-foot walk test results were previously misstated.
The selective B-cell–targeting monoclonal antibody ocrelizumab became the first drug to improve outcomes in primary progressive multiple sclerosis based on results from the randomized, placebo-controlled, phase III ORATORIO trial.
After 24 weeks of treatment, ocrelizumab reduced the risk of progression in clinical disability by 25%, compared with placebo. Over 2 years, the antibody also reduced the volume of hyperintense T2 lesions by 3.4%, whereas patients taking placebo experienced a 7% increase. The rate of whole brain volume loss was also significantly reduced.
The findings confer the first really good news in how to treat patients with this very difficult condition, said Dr. Xavier Montalbán, who presented the results at the annual meeting of the European Committee for Treatment and Research in Multiple Sclerosis, held in Barcelona.
“We’ve been looking for something positive for these patients for years and have never found this,” said Dr. Montalbán, director of clinical neuroimmunology at Vall d’Hebron University Hospital, Barcelona. “We can discuss the magnitude of the effect, but I have to say I am very, very happy with this.”
The results are certainly encouraging, said Dr. Peter Calabresi, director of the Johns Hopkins Multiple Sclerosis Center and the division of neuroimmunology at The Johns Hopkins Hospital, Baltimore, although he tempered his enthusiasm a bit regarding patient selection and safety. In the entire cohort of 732 patients, 13 malignancies occurred over 3 years and occurred more than twice as often in the ocrelizumab arm than in the placebo arm (2.3% vs. 0.8%). These included four breast cancers in the active arm and none in the placebo arm.
“The data in general look very strong but with a possible safety signal related to cancers and deaths,” Dr. Calabresi said in an interview. “I’d need to see more detail in this regard, but overall I am very happy to see something work for primary progressive MS, where we have no approved drugs, so this is very important in that regard.
“As others have pointed out, though, the study was designed to target young people with inflammation (active MRIs) in which cases the drug works well. But many of our people with progressive MS are older and have no inflammation, so I’m guessing there is no effect in that group.”
Ocrelizumab, a humanized monoclonal antibody, selectively targets CD20-positive B cells. Since not all B cells are positive for the marker, ocrelizumab leaves intact the patient’s immune memory, as well as the ability to create more B cells. Its success bolsters the emerging idea that B cells may be even more important than T cells in the pathophysiology of MS.
ORATORIO was a 2:1 randomization of 732 patients with primary progressive MS to infusions of either 600 mg ocrelizumab or placebo every 24 weeks for 120 months.
Most patients finished the trial (80% active, 66% placebo). The withdrawals in the placebo arm were primarily due to lack of efficacy. Patients were a mean of 44 years old, with a mean 6.5 years since symptom onset. Most (88%) were treatment-naive. The Expanded Disability Status Score (EDSS) at baseline was 4.7 in both groups. At baseline, about a quarter of patients had gadolinium-enhancing lesions (mean number, 1) with a mean T2 lesion volume of 11 cm3, and their mean whole-brain volume was 1,467 cm3. All patients had either elevated immunoglobulin in cerebrospinal fluid or oligoclonal bands.
The primary endpoint of ORATORIO was 12-week confirmed disability progression (CDP). Secondary endpoints included 24-week CDP; timed 25-foot walk; and T2 lesion and whole brain volume.
By 120 weeks, patients taking the study drug experienced a mean 24% reduction in the risk of CDP (hazard ratio, 0.76; P = .0321). The treatment curves began to separate at week 24, and the differences in the risk of CDP were statistically significant at every evaluation. The benefit has been maintained in a safety evaluation that is extending treatment to 216 weeks in a subset of patients.
*Ocrelizumab significantly slowed the worsening of patients’ 25-foot walking time by 29% at 120 weeks when compared against placebo. Walking times increased by 39% from baseline on ocrelizumab, compared with a 55% rise for those on placebo (P = .0404).
On the imaging endpoints, the drug was associated with a highly significant difference in T2 lesion volume, decreasing the load by 3.4%, while the placebo arm experienced an increase of 7.4% from baseline (P = .0001).
Those taking the drug also experienced a small but significantly lower amount of whole-brain volume loss (–0.9% vs. –1.1%; P = .02).
Infusion reactions were most common with the first dose (27% active vs. 7% placebo). One patient taking the study drug withdrew at that time from a serious reaction, but the remainder were mild to moderate in severity. Reactions decreased over time, and all remained at the mild-moderate level.
Overall adverse events occurred at similar rates between those taking the drug and those taking placebo (95% vs. 90%). Infections were similar (70% vs. 68%) overall, although upper respiratory infections were more common among the active group (11% vs. 6%).
There were five deaths: one in the placebo arm (a motor vehicle accident) and four in the active arm (pulmonary embolism, pneumonia, pancreatic cancer, and pneumonia aspiration). These, as well as the malignancy findings, are still being evaluated, Dr. Montalbán said.
Based on the positive results, Genentech will be submitting the drug for approval in both the United States and Europe in the first quarter of 2016.
Dr. Montalbán disclosed ties with Genentech – the trial’s sponsor – and other pharmaceutical companies.
*Correction, 10/14/15: The timed 25-foot walk test results were previously misstated.
FROM ECTRIMS 2015
Key clinical point: The humanized monoclonal antibody ocrelizumab is the first drug to post positive results for patients with primary progressive multiple sclerosis.
Major finding: Compared with placebo, ocrelizumab reduced the risk of progression in clinical disability for at least 24 weeks by 25%.
Data source: ORATORIO randomized 732 patients to placebo or 600 mg ocrelizumab infusions every 24 weeks.
Disclosures: Dr. Montalbán disclosed ties with Genentech – the sponsor of the trial – and other pharmaceutical companies.
EASD: Liraglutide lowers HbA1c when added to insulin in longstanding type 2 diabetes
STOCKHOLM – Adding liraglutide to multiple daily insulin injections helped stabilize blood glucose in patients with type 2 diabetes, while also resulting in weight loss and reduction of daily insulin doses.
All this was accomplished without an increase in the risk of hypoglycemia, Dr. Marcus Lind said at the annual meeting of the European Association for the Study of Diabetes.
A randomized, placebo-controlled study showed that the approach was successful in patients with long-standing disease, said Dr. Lind of the University of Gothenburg, Sweden. “We used to think that incretin-based therapies were most helpful in early type 2 diabetes. This seems to confirm that they are effective during the entire disease process.”
The soon-to-be-published 24-week study examined the addition of liraglutide in 124 patients with type 2 diabetes of long duration (a mean of 17 years). They were overweight, with a mean body mass index of 33.5 (about 218 pounds). At baseline, their mean hemoglobin A1c was 9%; they were taking a mean of 105 units of insulin each day in about four injections.
The study’s primary endpoint was change in HbA1c at 24 weeks. This declined significantly more among those taking liraglutide than those taking a placebo (–1.6% vs. –0.4%). “This is a difference of 1.1%, which is very clinically significant,” Dr. Lind said.
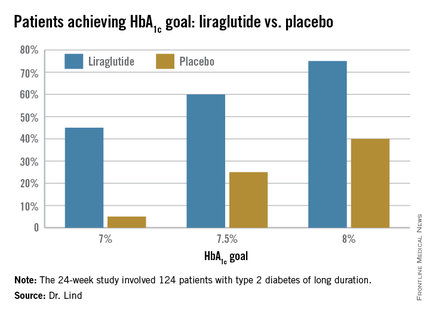
Dr. Lind assigned individualized HbA1c goals to patients, with the cutpoints of 7%, 7.5%, and 8%. Significantly more patients taking liraglutide were able to achieve those goals (see chart).
They also lost about 8 pounds more weight than did the placebo patients, and experienced a greater decrease in systolic blood pressure (–5.5 mm Hg more than placebo). There was no effect on diastolic blood pressure or lipids.
There were no severe hypoglycemic events in either group, nor any between-group difference in nonsevere events.
There were three serious adverse events among three patients taking liraglutide, and eight among four patients taking placebo; none of these were pancreatitis or pancreatic cancer.
Patients taking the study drug experienced significantly more nausea, especially at the beginning of the study, compared to the end of the study (22% vs. 5%).
The study was investigator initiated but received financial support from Novo Nordisk, which provided the drug. Dr. Lind disclosed financial relationships with numerous pharmaceutical companies, including honoraria from Novo Nordisk.
STOCKHOLM – Adding liraglutide to multiple daily insulin injections helped stabilize blood glucose in patients with type 2 diabetes, while also resulting in weight loss and reduction of daily insulin doses.
All this was accomplished without an increase in the risk of hypoglycemia, Dr. Marcus Lind said at the annual meeting of the European Association for the Study of Diabetes.
A randomized, placebo-controlled study showed that the approach was successful in patients with long-standing disease, said Dr. Lind of the University of Gothenburg, Sweden. “We used to think that incretin-based therapies were most helpful in early type 2 diabetes. This seems to confirm that they are effective during the entire disease process.”
The soon-to-be-published 24-week study examined the addition of liraglutide in 124 patients with type 2 diabetes of long duration (a mean of 17 years). They were overweight, with a mean body mass index of 33.5 (about 218 pounds). At baseline, their mean hemoglobin A1c was 9%; they were taking a mean of 105 units of insulin each day in about four injections.
The study’s primary endpoint was change in HbA1c at 24 weeks. This declined significantly more among those taking liraglutide than those taking a placebo (–1.6% vs. –0.4%). “This is a difference of 1.1%, which is very clinically significant,” Dr. Lind said.

Dr. Lind assigned individualized HbA1c goals to patients, with the cutpoints of 7%, 7.5%, and 8%. Significantly more patients taking liraglutide were able to achieve those goals (see chart).
They also lost about 8 pounds more weight than did the placebo patients, and experienced a greater decrease in systolic blood pressure (–5.5 mm Hg more than placebo). There was no effect on diastolic blood pressure or lipids.
There were no severe hypoglycemic events in either group, nor any between-group difference in nonsevere events.
There were three serious adverse events among three patients taking liraglutide, and eight among four patients taking placebo; none of these were pancreatitis or pancreatic cancer.
Patients taking the study drug experienced significantly more nausea, especially at the beginning of the study, compared to the end of the study (22% vs. 5%).
The study was investigator initiated but received financial support from Novo Nordisk, which provided the drug. Dr. Lind disclosed financial relationships with numerous pharmaceutical companies, including honoraria from Novo Nordisk.
STOCKHOLM – Adding liraglutide to multiple daily insulin injections helped stabilize blood glucose in patients with type 2 diabetes, while also resulting in weight loss and reduction of daily insulin doses.
All this was accomplished without an increase in the risk of hypoglycemia, Dr. Marcus Lind said at the annual meeting of the European Association for the Study of Diabetes.
A randomized, placebo-controlled study showed that the approach was successful in patients with long-standing disease, said Dr. Lind of the University of Gothenburg, Sweden. “We used to think that incretin-based therapies were most helpful in early type 2 diabetes. This seems to confirm that they are effective during the entire disease process.”
The soon-to-be-published 24-week study examined the addition of liraglutide in 124 patients with type 2 diabetes of long duration (a mean of 17 years). They were overweight, with a mean body mass index of 33.5 (about 218 pounds). At baseline, their mean hemoglobin A1c was 9%; they were taking a mean of 105 units of insulin each day in about four injections.
The study’s primary endpoint was change in HbA1c at 24 weeks. This declined significantly more among those taking liraglutide than those taking a placebo (–1.6% vs. –0.4%). “This is a difference of 1.1%, which is very clinically significant,” Dr. Lind said.

Dr. Lind assigned individualized HbA1c goals to patients, with the cutpoints of 7%, 7.5%, and 8%. Significantly more patients taking liraglutide were able to achieve those goals (see chart).
They also lost about 8 pounds more weight than did the placebo patients, and experienced a greater decrease in systolic blood pressure (–5.5 mm Hg more than placebo). There was no effect on diastolic blood pressure or lipids.
There were no severe hypoglycemic events in either group, nor any between-group difference in nonsevere events.
There were three serious adverse events among three patients taking liraglutide, and eight among four patients taking placebo; none of these were pancreatitis or pancreatic cancer.
Patients taking the study drug experienced significantly more nausea, especially at the beginning of the study, compared to the end of the study (22% vs. 5%).
The study was investigator initiated but received financial support from Novo Nordisk, which provided the drug. Dr. Lind disclosed financial relationships with numerous pharmaceutical companies, including honoraria from Novo Nordisk.
AT EASD 2015
Key clinical point: Liraglutide added to multiple daily insulin injections lowered HbA1c significantly more than did placebo in patients with type 2 diabetes.
Major finding: HbA1c at 24 weeks declined significantly more among those taking liraglutide than those taking a placebo (–1.6% vs. –0.4%).
Data source: A randomized, placebo-controlled study of 124 patients.
Disclosures: The study was investigator initiated but received financial support from Novo Nordisk, which provided the drug. Dr. Lind disclosed financial relationships with numerous pharmaceutical companies, including honoraria from Novo Nordisk.