User login
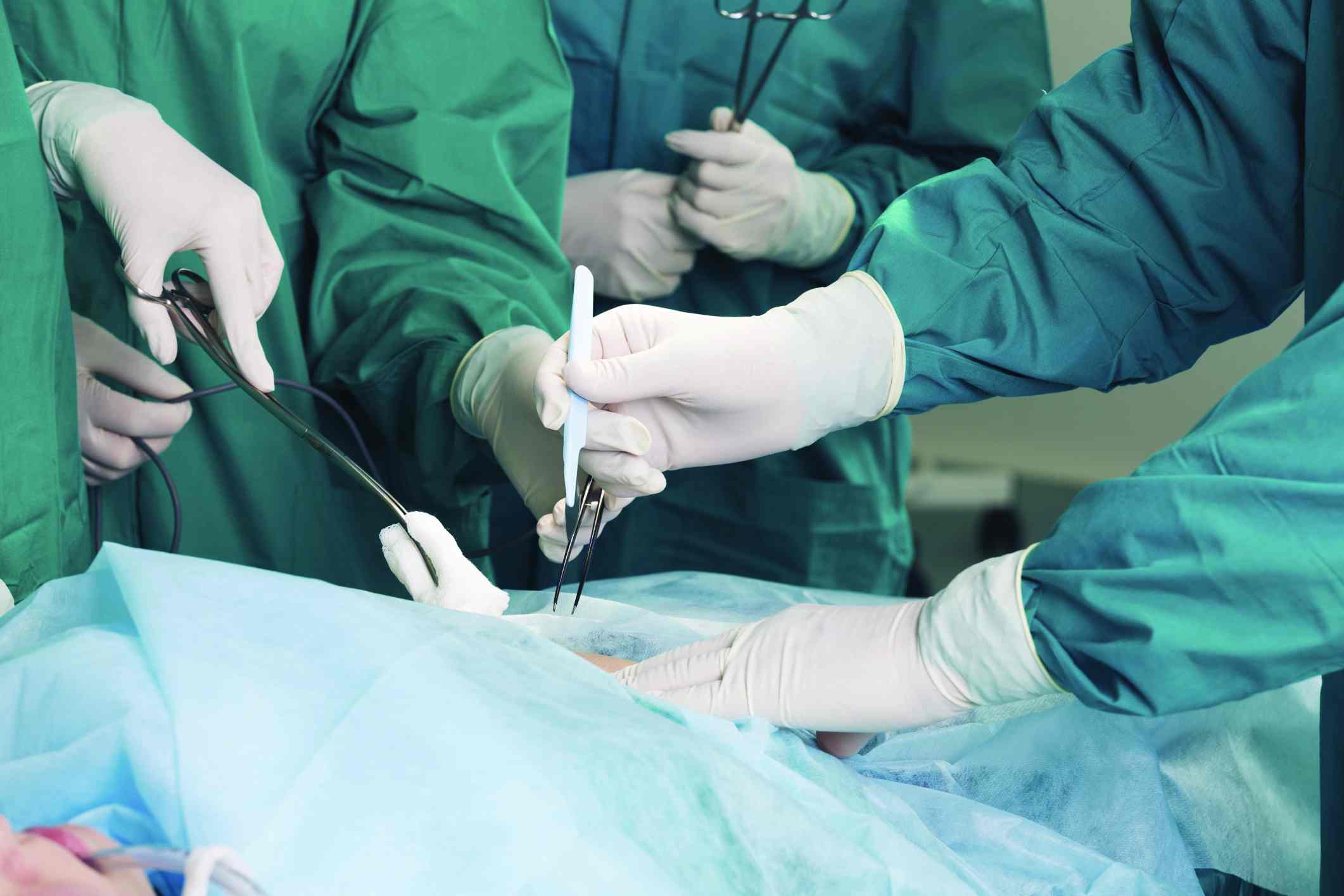
Laparoscopic TAP blocks offer quick, easy, postoperative analgesia
LAS VEGAS – A laparoscopic transversus abdominis plane (TAP) block is easy to learn, quick to perform, and provides effective postoperative analgesia for women undergoing minimally invasive gynecologic surgeries.
TAP blocks are usually done by an anesthesiologist under ultrasound guidance, Dr. Shanti Mohling said at a meeting sponsored by AAGL. But they can also be performed quite efficiently during a laparoscopic procedure using anatomic landmarks.
“All you need to perform this is a knowledge of the anatomy, a beveled needle with tubing, and two syringes with 20-30 cc of analgesic,” said Dr. Mohling of the University of Tennessee, Chattanooga. “It takes about 3 minutes and it’s very time saving, compared to having an anesthesiologist do this with ultrasound guidance.”
She presented a video of her technique, which relies on identification of the lumbar triangle of Petit to deliver a large volume of anesthetic into the transversus abdominis plane – the neurovascular space between the internal oblique and transversus abdominis muscles. The area is of critical importance to pain sensation, she said. “This plane carries the afferent nociceptor nerves for T7-L1, including the ilioinguinal and iliohypogastric nerves.”
The efficacy of a laparoscopically administered TAP block during laparoscopic gynecologic surgery has been demonstrated in several studies, she noted. One – a review of 61 cases of total laparoscopic hysterectomy - found associations between the use of the TAP black and reduced length of stay and lower opioid consumption (Aust N Z J Obstet Gynaecol. 2011 Dec;51[6]:544-7.).
The video described her technique, beginning with identifying the triangle of Petit. “The triangle of Petit is the area formed between the iliac crest inferiorly, the latissimus dorsi posteriorly, and the external oblique anteriorly. Within this triangle, we find perfect access to the transversus abdominis plane.”
Once the triangle is identified, a beveled needle is advanced slowly, “as to appreciate the ‘double pop sensation’ of the needle as it passes the fascia of the external and internal oblique muscles. Laparoscopically, this can be observed,” Dr. Mohling said. “If the needle can be seen just beneath the peritoneum, then it has penetrated too far.”
Once into the space, which can easily accommodate a large volume of fluid, she delivers the anesthetic into the neurovascular plane. “Importantly, the injectate must be of sufficient volume to effectively spread throughout the neurovascular plane. Typically this requires 20-30 cc on each side,” she said.
Long-acting agents like bupivacaine or ropivacaine are preferable. Dr. Mohling uses a solution of 10 cc liposomal bupivacaine; 10 cc 0.25% bupivacaine; and 10 cc normal saline.
As the anesthetic is injected, it’s important to track it visually to assure correct placement, she noted. “The delivery can be noted laparoscopically by watching a bulge spreading beneath the transversus abdominis fascia.”
The potential for complications is low, but these include intraperitoneal injection; abdominal wall or bowel hematoma; transient femoral nerve palsy; and local anesthetic toxicity.
Dr. Mohling is in the process of conducting a randomized controlled trial of 100 women undergoing planned laparoscopic or robotic hysterectomy. They will be assigned to either TAP block with the liposomal bupivacaine solution or to traditional preincisional local anesthetic with bupivacaine alone.
“I believe we should all add this technique to our practice,” she said. “It’s easy and there is an increasing body of evidence supporting these blocks.”
Dr. Mohling reported having no financial disclosures.
LAS VEGAS – A laparoscopic transversus abdominis plane (TAP) block is easy to learn, quick to perform, and provides effective postoperative analgesia for women undergoing minimally invasive gynecologic surgeries.
TAP blocks are usually done by an anesthesiologist under ultrasound guidance, Dr. Shanti Mohling said at a meeting sponsored by AAGL. But they can also be performed quite efficiently during a laparoscopic procedure using anatomic landmarks.
“All you need to perform this is a knowledge of the anatomy, a beveled needle with tubing, and two syringes with 20-30 cc of analgesic,” said Dr. Mohling of the University of Tennessee, Chattanooga. “It takes about 3 minutes and it’s very time saving, compared to having an anesthesiologist do this with ultrasound guidance.”
She presented a video of her technique, which relies on identification of the lumbar triangle of Petit to deliver a large volume of anesthetic into the transversus abdominis plane – the neurovascular space between the internal oblique and transversus abdominis muscles. The area is of critical importance to pain sensation, she said. “This plane carries the afferent nociceptor nerves for T7-L1, including the ilioinguinal and iliohypogastric nerves.”
The efficacy of a laparoscopically administered TAP block during laparoscopic gynecologic surgery has been demonstrated in several studies, she noted. One – a review of 61 cases of total laparoscopic hysterectomy - found associations between the use of the TAP black and reduced length of stay and lower opioid consumption (Aust N Z J Obstet Gynaecol. 2011 Dec;51[6]:544-7.).
The video described her technique, beginning with identifying the triangle of Petit. “The triangle of Petit is the area formed between the iliac crest inferiorly, the latissimus dorsi posteriorly, and the external oblique anteriorly. Within this triangle, we find perfect access to the transversus abdominis plane.”
Once the triangle is identified, a beveled needle is advanced slowly, “as to appreciate the ‘double pop sensation’ of the needle as it passes the fascia of the external and internal oblique muscles. Laparoscopically, this can be observed,” Dr. Mohling said. “If the needle can be seen just beneath the peritoneum, then it has penetrated too far.”
Once into the space, which can easily accommodate a large volume of fluid, she delivers the anesthetic into the neurovascular plane. “Importantly, the injectate must be of sufficient volume to effectively spread throughout the neurovascular plane. Typically this requires 20-30 cc on each side,” she said.
Long-acting agents like bupivacaine or ropivacaine are preferable. Dr. Mohling uses a solution of 10 cc liposomal bupivacaine; 10 cc 0.25% bupivacaine; and 10 cc normal saline.
As the anesthetic is injected, it’s important to track it visually to assure correct placement, she noted. “The delivery can be noted laparoscopically by watching a bulge spreading beneath the transversus abdominis fascia.”
The potential for complications is low, but these include intraperitoneal injection; abdominal wall or bowel hematoma; transient femoral nerve palsy; and local anesthetic toxicity.
Dr. Mohling is in the process of conducting a randomized controlled trial of 100 women undergoing planned laparoscopic or robotic hysterectomy. They will be assigned to either TAP block with the liposomal bupivacaine solution or to traditional preincisional local anesthetic with bupivacaine alone.
“I believe we should all add this technique to our practice,” she said. “It’s easy and there is an increasing body of evidence supporting these blocks.”
Dr. Mohling reported having no financial disclosures.
LAS VEGAS – A laparoscopic transversus abdominis plane (TAP) block is easy to learn, quick to perform, and provides effective postoperative analgesia for women undergoing minimally invasive gynecologic surgeries.
TAP blocks are usually done by an anesthesiologist under ultrasound guidance, Dr. Shanti Mohling said at a meeting sponsored by AAGL. But they can also be performed quite efficiently during a laparoscopic procedure using anatomic landmarks.
“All you need to perform this is a knowledge of the anatomy, a beveled needle with tubing, and two syringes with 20-30 cc of analgesic,” said Dr. Mohling of the University of Tennessee, Chattanooga. “It takes about 3 minutes and it’s very time saving, compared to having an anesthesiologist do this with ultrasound guidance.”
She presented a video of her technique, which relies on identification of the lumbar triangle of Petit to deliver a large volume of anesthetic into the transversus abdominis plane – the neurovascular space between the internal oblique and transversus abdominis muscles. The area is of critical importance to pain sensation, she said. “This plane carries the afferent nociceptor nerves for T7-L1, including the ilioinguinal and iliohypogastric nerves.”
The efficacy of a laparoscopically administered TAP block during laparoscopic gynecologic surgery has been demonstrated in several studies, she noted. One – a review of 61 cases of total laparoscopic hysterectomy - found associations between the use of the TAP black and reduced length of stay and lower opioid consumption (Aust N Z J Obstet Gynaecol. 2011 Dec;51[6]:544-7.).
The video described her technique, beginning with identifying the triangle of Petit. “The triangle of Petit is the area formed between the iliac crest inferiorly, the latissimus dorsi posteriorly, and the external oblique anteriorly. Within this triangle, we find perfect access to the transversus abdominis plane.”
Once the triangle is identified, a beveled needle is advanced slowly, “as to appreciate the ‘double pop sensation’ of the needle as it passes the fascia of the external and internal oblique muscles. Laparoscopically, this can be observed,” Dr. Mohling said. “If the needle can be seen just beneath the peritoneum, then it has penetrated too far.”
Once into the space, which can easily accommodate a large volume of fluid, she delivers the anesthetic into the neurovascular plane. “Importantly, the injectate must be of sufficient volume to effectively spread throughout the neurovascular plane. Typically this requires 20-30 cc on each side,” she said.
Long-acting agents like bupivacaine or ropivacaine are preferable. Dr. Mohling uses a solution of 10 cc liposomal bupivacaine; 10 cc 0.25% bupivacaine; and 10 cc normal saline.
As the anesthetic is injected, it’s important to track it visually to assure correct placement, she noted. “The delivery can be noted laparoscopically by watching a bulge spreading beneath the transversus abdominis fascia.”
The potential for complications is low, but these include intraperitoneal injection; abdominal wall or bowel hematoma; transient femoral nerve palsy; and local anesthetic toxicity.
Dr. Mohling is in the process of conducting a randomized controlled trial of 100 women undergoing planned laparoscopic or robotic hysterectomy. They will be assigned to either TAP block with the liposomal bupivacaine solution or to traditional preincisional local anesthetic with bupivacaine alone.
“I believe we should all add this technique to our practice,” she said. “It’s easy and there is an increasing body of evidence supporting these blocks.”
Dr. Mohling reported having no financial disclosures.
EXPERT ANALYSIS FROM THE AAGL GLOBAL CONGRESS
FDA gives nod to rapid-infusion bendamustine for CLL
The Food and Drug Administration has approved a new rapid-infusion form of bendamustine hydrochloride for patients with chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma.
The new formulation of bendamustine (Bendeka) is a 50-mL liquid designed for 10-minute infusion. It was granted orphan drug status for both the leukemia and lymphoma indications.
Bendeka is approved as primary therapy for chronic lymphocytic leukemia (CLL) and also for indolent B-cell non-Hodgkin lymphoma (NHL) that has progressed during or within 6 months of treatment with rituximab or a rituximab-containing regimen.
According to the prescribing information, the recommended dosing regimen for CLL is 100 mg/m2 infused intravenously over 10 minutes on days 1 and 2 of a 28-day cycle, up to six cycles. For NHL, the regimen calls for 120 mg/m2 infused intravenously over 10 minutes on days 1 and 2 of a 21-day cycle, up to eight cycles.
The most common hematologic adverse reactions are lymphopenia, anemia, leukopenia, thrombocytopenia, and neutropenia. Bendamustine has been associated with severe – and even fatal – myelosuppression.
Bendeka is manufactured by Teva Pharmaceutical Industries and succeeds Teva’s previously approved form of bendamustine, Treanda, approved in 2008. “Since 2008, Treanda has played a valuable role in the treatment of patients with CLL or indolent B-cell NHL that has progressed,” Paul Rittman, Teva Oncology’s vice president and general manager, said in a press statement. Treanda’s orphan drug exclusivity status for the NHL indication was set to expire in October, and its pediatric exclusivity status for that indication, next April. Its CLL exclusivity status expired in September.
Teva purchased the new formulation from Eagle Pharmaceuticals last February. The deal was closed with an upfront payment of $30 million, and potential for up to $90 million in additional payments, as well as double-digit royalties on net sales.
At the time of purchase, Eagle had secured orphan drug designations for Bendeka in both CLL and NHL and had submitted the New Drug Application under priority review. Bendeka may be eligible for a 7-year exclusivity status.
The drug is scheduled to be available during the first quarter of 2016.
The Food and Drug Administration has approved a new rapid-infusion form of bendamustine hydrochloride for patients with chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma.
The new formulation of bendamustine (Bendeka) is a 50-mL liquid designed for 10-minute infusion. It was granted orphan drug status for both the leukemia and lymphoma indications.
Bendeka is approved as primary therapy for chronic lymphocytic leukemia (CLL) and also for indolent B-cell non-Hodgkin lymphoma (NHL) that has progressed during or within 6 months of treatment with rituximab or a rituximab-containing regimen.
According to the prescribing information, the recommended dosing regimen for CLL is 100 mg/m2 infused intravenously over 10 minutes on days 1 and 2 of a 28-day cycle, up to six cycles. For NHL, the regimen calls for 120 mg/m2 infused intravenously over 10 minutes on days 1 and 2 of a 21-day cycle, up to eight cycles.
The most common hematologic adverse reactions are lymphopenia, anemia, leukopenia, thrombocytopenia, and neutropenia. Bendamustine has been associated with severe – and even fatal – myelosuppression.
Bendeka is manufactured by Teva Pharmaceutical Industries and succeeds Teva’s previously approved form of bendamustine, Treanda, approved in 2008. “Since 2008, Treanda has played a valuable role in the treatment of patients with CLL or indolent B-cell NHL that has progressed,” Paul Rittman, Teva Oncology’s vice president and general manager, said in a press statement. Treanda’s orphan drug exclusivity status for the NHL indication was set to expire in October, and its pediatric exclusivity status for that indication, next April. Its CLL exclusivity status expired in September.
Teva purchased the new formulation from Eagle Pharmaceuticals last February. The deal was closed with an upfront payment of $30 million, and potential for up to $90 million in additional payments, as well as double-digit royalties on net sales.
At the time of purchase, Eagle had secured orphan drug designations for Bendeka in both CLL and NHL and had submitted the New Drug Application under priority review. Bendeka may be eligible for a 7-year exclusivity status.
The drug is scheduled to be available during the first quarter of 2016.
The Food and Drug Administration has approved a new rapid-infusion form of bendamustine hydrochloride for patients with chronic lymphocytic leukemia and indolent B-cell non-Hodgkin lymphoma.
The new formulation of bendamustine (Bendeka) is a 50-mL liquid designed for 10-minute infusion. It was granted orphan drug status for both the leukemia and lymphoma indications.
Bendeka is approved as primary therapy for chronic lymphocytic leukemia (CLL) and also for indolent B-cell non-Hodgkin lymphoma (NHL) that has progressed during or within 6 months of treatment with rituximab or a rituximab-containing regimen.
According to the prescribing information, the recommended dosing regimen for CLL is 100 mg/m2 infused intravenously over 10 minutes on days 1 and 2 of a 28-day cycle, up to six cycles. For NHL, the regimen calls for 120 mg/m2 infused intravenously over 10 minutes on days 1 and 2 of a 21-day cycle, up to eight cycles.
The most common hematologic adverse reactions are lymphopenia, anemia, leukopenia, thrombocytopenia, and neutropenia. Bendamustine has been associated with severe – and even fatal – myelosuppression.
Bendeka is manufactured by Teva Pharmaceutical Industries and succeeds Teva’s previously approved form of bendamustine, Treanda, approved in 2008. “Since 2008, Treanda has played a valuable role in the treatment of patients with CLL or indolent B-cell NHL that has progressed,” Paul Rittman, Teva Oncology’s vice president and general manager, said in a press statement. Treanda’s orphan drug exclusivity status for the NHL indication was set to expire in October, and its pediatric exclusivity status for that indication, next April. Its CLL exclusivity status expired in September.
Teva purchased the new formulation from Eagle Pharmaceuticals last February. The deal was closed with an upfront payment of $30 million, and potential for up to $90 million in additional payments, as well as double-digit royalties on net sales.
At the time of purchase, Eagle had secured orphan drug designations for Bendeka in both CLL and NHL and had submitted the New Drug Application under priority review. Bendeka may be eligible for a 7-year exclusivity status.
The drug is scheduled to be available during the first quarter of 2016.
GeneMatch designed to speed up Alzheimer’s trial recruitment
A new Alzheimer’s research program aims to accelerate the recruitment of people interested in volunteering for Alzheimer’s studies based on genetic risk factors.
GeneMatch, created and administered by the Banner Alzheimer’s Institute, Phoenix, will be a key part of the research toward finding an intervention to slow or stop the development of Alzheimer’s disease in people who carry the high-risk variant of the apolipoprotein E gene (APOE). By creating a large pool of cognitively healthy subjects who are already stratified by APOE status, GeneMatch can accelerate enrollment in studies targeting those with the highest-risk allele, APOE epsilon-4 (APOE4).
“Research studies in healthy people at different levels of genetic risk for Alzheimer’s promise to further clarify the earliest biological changes associated with the disease, clarify what genetic risk disclosure means to people in the new era of Alzheimer’s prevention trials, and find effective treatments to end Alzheimer’s as quickly as possible,” said Dr. Eric Reiman, executive director at Banner and codirector of the Alzheimer’s Prevention Initiative, of which GeneMatch is a part. “GeneMatch is intended to provide an unprecedented resource of genetically characterized research volunteers to do just that.”
Because at least one APOE4 allele is carried by about 14% in the general population, recruiting a sufficient number of these subjects for studies can take years. Having a pool of prescreened subjects should make the process much easier, much quicker, and much less expensive, according to Dr. Pierre N. Tariot, director of the institute.
For example, Dr. Tariot said during a teleconference, the upcoming Alzheimer’s Prevention Initiative APOE4 Trialneeds about 1,300 cognitively normal people who carry two copies of the APOE4 allele. That genetic combination – the most aggressive risk factor for late-onset Alzheimer’s – occurs in less than 2% of the general population.
“We would need to genotype tens of thousands of people to end up with that many participants,” Dr. Tariot said. GeneMatch should lower the screening number from tens of thousands to thousands, he said.
In fact, the API APOE4 Trial, which will test both the anti-amyloid antibody crenezumab and a beta-secretase inhibitor in homozygous APOE4 carriers, will be the first one to use GeneMatch along with traditional recruitment methods, Dr. Tariot said. Other Alzheimer’s studies with a genetic component may also take advantage of the subject pool because it will be a shared resource.
The GeneMatch program grew from the challenge researchers faced in recruiting enough subjects for the API APOE4 trial, Dr. Reiman said.
“We decided to conduct these screening tests in a way to benefit the entire AD research community,” he said. “We hope this shared resource will help researchers clarify the earliest brain changes, any protective factors for APOE4, and contribute to a growing number of prevention trials and find something that will work to delay or stop Alzheimer’s onset within the next 10 years. We really think this will dramatically shift how researches conduct trials, reducing the number of volunteers we need to recruit and saving time and money.”
“Recruiting to prevention trials based on genetic risk provides a major advantage since risk can be roughly quantified and major subgroups responses identified,” said Dr. Richard J. Caselli, when asked to comment on the new program. “Biomarker evidence is another approach, but one can question how presymptomatic someone with a head full of amyloid really is, especially over the usual recruitment ages. GeneMatch seems to be the first mass genetic screening and registry program in the U.S. and should accelerate recruitment into trials substantially,” said Dr. Caselli, professor of neurology at the Mayo Clinic, Scottsdale, Ariz., and associate director and clinical core director of the clinic’s Alzheimer’s Disease Center.
GeneMatch seeks to enroll tens of thousands of people who are aged 55-75 years, and cognitively normal. It’s open to anyone with or without a family history of Alzheimer’s. After completing a brief online educational module, people will be asked to consent to the protocol. Those who agree will receive a cheek swab kit in the mail; samples will be genotyped for APOE status.
GeneMatch, however, won’t disclose that information to participants. They would learn it only if they enrolled in a study that requires or allows disclosure of that information, said Dr. Jason Karlawish of the University of Pennsylvania, Philadelphia.
“The decision to learn the APOE genotype is a personal one and has to reflect the ethics of informed consent among people who had adequate time to consider their decision,” he said during the teleconference. “We respect the fact that some people want to know this information, and some do not. Our goal for the API APOE4 study is to capture into this study those who want to know.”
GeneMatch is not only an important research tool, Dr. Tariot said, but a way that anyone can take part in the search for an Alzheimer’s treatment.
“This process changes the role people can play in research. Families of patients often feel helpless after a diagnosis, but GeneMatch can give them the opportunity to take action, to be part of the movement to end this before another generation is lost.”
A new Alzheimer’s research program aims to accelerate the recruitment of people interested in volunteering for Alzheimer’s studies based on genetic risk factors.
GeneMatch, created and administered by the Banner Alzheimer’s Institute, Phoenix, will be a key part of the research toward finding an intervention to slow or stop the development of Alzheimer’s disease in people who carry the high-risk variant of the apolipoprotein E gene (APOE). By creating a large pool of cognitively healthy subjects who are already stratified by APOE status, GeneMatch can accelerate enrollment in studies targeting those with the highest-risk allele, APOE epsilon-4 (APOE4).
“Research studies in healthy people at different levels of genetic risk for Alzheimer’s promise to further clarify the earliest biological changes associated with the disease, clarify what genetic risk disclosure means to people in the new era of Alzheimer’s prevention trials, and find effective treatments to end Alzheimer’s as quickly as possible,” said Dr. Eric Reiman, executive director at Banner and codirector of the Alzheimer’s Prevention Initiative, of which GeneMatch is a part. “GeneMatch is intended to provide an unprecedented resource of genetically characterized research volunteers to do just that.”
Because at least one APOE4 allele is carried by about 14% in the general population, recruiting a sufficient number of these subjects for studies can take years. Having a pool of prescreened subjects should make the process much easier, much quicker, and much less expensive, according to Dr. Pierre N. Tariot, director of the institute.
For example, Dr. Tariot said during a teleconference, the upcoming Alzheimer’s Prevention Initiative APOE4 Trialneeds about 1,300 cognitively normal people who carry two copies of the APOE4 allele. That genetic combination – the most aggressive risk factor for late-onset Alzheimer’s – occurs in less than 2% of the general population.
“We would need to genotype tens of thousands of people to end up with that many participants,” Dr. Tariot said. GeneMatch should lower the screening number from tens of thousands to thousands, he said.
In fact, the API APOE4 Trial, which will test both the anti-amyloid antibody crenezumab and a beta-secretase inhibitor in homozygous APOE4 carriers, will be the first one to use GeneMatch along with traditional recruitment methods, Dr. Tariot said. Other Alzheimer’s studies with a genetic component may also take advantage of the subject pool because it will be a shared resource.
The GeneMatch program grew from the challenge researchers faced in recruiting enough subjects for the API APOE4 trial, Dr. Reiman said.
“We decided to conduct these screening tests in a way to benefit the entire AD research community,” he said. “We hope this shared resource will help researchers clarify the earliest brain changes, any protective factors for APOE4, and contribute to a growing number of prevention trials and find something that will work to delay or stop Alzheimer’s onset within the next 10 years. We really think this will dramatically shift how researches conduct trials, reducing the number of volunteers we need to recruit and saving time and money.”
“Recruiting to prevention trials based on genetic risk provides a major advantage since risk can be roughly quantified and major subgroups responses identified,” said Dr. Richard J. Caselli, when asked to comment on the new program. “Biomarker evidence is another approach, but one can question how presymptomatic someone with a head full of amyloid really is, especially over the usual recruitment ages. GeneMatch seems to be the first mass genetic screening and registry program in the U.S. and should accelerate recruitment into trials substantially,” said Dr. Caselli, professor of neurology at the Mayo Clinic, Scottsdale, Ariz., and associate director and clinical core director of the clinic’s Alzheimer’s Disease Center.
GeneMatch seeks to enroll tens of thousands of people who are aged 55-75 years, and cognitively normal. It’s open to anyone with or without a family history of Alzheimer’s. After completing a brief online educational module, people will be asked to consent to the protocol. Those who agree will receive a cheek swab kit in the mail; samples will be genotyped for APOE status.
GeneMatch, however, won’t disclose that information to participants. They would learn it only if they enrolled in a study that requires or allows disclosure of that information, said Dr. Jason Karlawish of the University of Pennsylvania, Philadelphia.
“The decision to learn the APOE genotype is a personal one and has to reflect the ethics of informed consent among people who had adequate time to consider their decision,” he said during the teleconference. “We respect the fact that some people want to know this information, and some do not. Our goal for the API APOE4 study is to capture into this study those who want to know.”
GeneMatch is not only an important research tool, Dr. Tariot said, but a way that anyone can take part in the search for an Alzheimer’s treatment.
“This process changes the role people can play in research. Families of patients often feel helpless after a diagnosis, but GeneMatch can give them the opportunity to take action, to be part of the movement to end this before another generation is lost.”
A new Alzheimer’s research program aims to accelerate the recruitment of people interested in volunteering for Alzheimer’s studies based on genetic risk factors.
GeneMatch, created and administered by the Banner Alzheimer’s Institute, Phoenix, will be a key part of the research toward finding an intervention to slow or stop the development of Alzheimer’s disease in people who carry the high-risk variant of the apolipoprotein E gene (APOE). By creating a large pool of cognitively healthy subjects who are already stratified by APOE status, GeneMatch can accelerate enrollment in studies targeting those with the highest-risk allele, APOE epsilon-4 (APOE4).
“Research studies in healthy people at different levels of genetic risk for Alzheimer’s promise to further clarify the earliest biological changes associated with the disease, clarify what genetic risk disclosure means to people in the new era of Alzheimer’s prevention trials, and find effective treatments to end Alzheimer’s as quickly as possible,” said Dr. Eric Reiman, executive director at Banner and codirector of the Alzheimer’s Prevention Initiative, of which GeneMatch is a part. “GeneMatch is intended to provide an unprecedented resource of genetically characterized research volunteers to do just that.”
Because at least one APOE4 allele is carried by about 14% in the general population, recruiting a sufficient number of these subjects for studies can take years. Having a pool of prescreened subjects should make the process much easier, much quicker, and much less expensive, according to Dr. Pierre N. Tariot, director of the institute.
For example, Dr. Tariot said during a teleconference, the upcoming Alzheimer’s Prevention Initiative APOE4 Trialneeds about 1,300 cognitively normal people who carry two copies of the APOE4 allele. That genetic combination – the most aggressive risk factor for late-onset Alzheimer’s – occurs in less than 2% of the general population.
“We would need to genotype tens of thousands of people to end up with that many participants,” Dr. Tariot said. GeneMatch should lower the screening number from tens of thousands to thousands, he said.
In fact, the API APOE4 Trial, which will test both the anti-amyloid antibody crenezumab and a beta-secretase inhibitor in homozygous APOE4 carriers, will be the first one to use GeneMatch along with traditional recruitment methods, Dr. Tariot said. Other Alzheimer’s studies with a genetic component may also take advantage of the subject pool because it will be a shared resource.
The GeneMatch program grew from the challenge researchers faced in recruiting enough subjects for the API APOE4 trial, Dr. Reiman said.
“We decided to conduct these screening tests in a way to benefit the entire AD research community,” he said. “We hope this shared resource will help researchers clarify the earliest brain changes, any protective factors for APOE4, and contribute to a growing number of prevention trials and find something that will work to delay or stop Alzheimer’s onset within the next 10 years. We really think this will dramatically shift how researches conduct trials, reducing the number of volunteers we need to recruit and saving time and money.”
“Recruiting to prevention trials based on genetic risk provides a major advantage since risk can be roughly quantified and major subgroups responses identified,” said Dr. Richard J. Caselli, when asked to comment on the new program. “Biomarker evidence is another approach, but one can question how presymptomatic someone with a head full of amyloid really is, especially over the usual recruitment ages. GeneMatch seems to be the first mass genetic screening and registry program in the U.S. and should accelerate recruitment into trials substantially,” said Dr. Caselli, professor of neurology at the Mayo Clinic, Scottsdale, Ariz., and associate director and clinical core director of the clinic’s Alzheimer’s Disease Center.
GeneMatch seeks to enroll tens of thousands of people who are aged 55-75 years, and cognitively normal. It’s open to anyone with or without a family history of Alzheimer’s. After completing a brief online educational module, people will be asked to consent to the protocol. Those who agree will receive a cheek swab kit in the mail; samples will be genotyped for APOE status.
GeneMatch, however, won’t disclose that information to participants. They would learn it only if they enrolled in a study that requires or allows disclosure of that information, said Dr. Jason Karlawish of the University of Pennsylvania, Philadelphia.
“The decision to learn the APOE genotype is a personal one and has to reflect the ethics of informed consent among people who had adequate time to consider their decision,” he said during the teleconference. “We respect the fact that some people want to know this information, and some do not. Our goal for the API APOE4 study is to capture into this study those who want to know.”
GeneMatch is not only an important research tool, Dr. Tariot said, but a way that anyone can take part in the search for an Alzheimer’s treatment.
“This process changes the role people can play in research. Families of patients often feel helpless after a diagnosis, but GeneMatch can give them the opportunity to take action, to be part of the movement to end this before another generation is lost.”
New analyses of data keep hope alive for Alzheimer’s drug gantenerumab
BARCELONA – A failed phase III clinical trial for the investigational antiamyloid antibody gantenerumab in patients with Alzheimer’s disease may have had better results with a higher dose aimed at a more targeted population, new post hoc analyses of the trial’s data suggest.
The subanalyses of the now-defunct SCARLET ROAD study indicate that patients who had a more rapid progression of disease and took gantenerumab 225 mg received some cognitive benefit, especially in those who had the highest plasma concentrations.
Biomarker results also shifted toward the positive, according to data presented at the Clinical Trials in Alzheimer’s Disease conference. Amyloid PET imaging showed significantly greater plaque reduction in the brains of those with the highest gantenerumab exposure. Cerebrospinal fluid tests found that the antibody significantly decreased the level of phosphorylated tau. This, said Dr. Tania Nikolcheva, is the first evidence that attacking amyloid brain plaques can have a downstream effect on tau, which is now considered to be the major player in Alzheimer’s cognitive decline.
“We demonstrated that gantenerumab shows a consistent effect on the intended target as well as on downstream biomarkers,” said Dr. Nikolcheva of Hoffman-LaRoche, which is developing the drug. “These seem to be exposure- and dose-dependent, which supports following this trial with tests of higher dosages.”
In December 2014, Roche’s SCARLET ROAD study was halted after a 2-year interim analysis found that neither the 105 mg nor 225 mg dose conferred a significant cognitive benefit relative to placebo. But the antibody’s story is still unfolding: The drug is still being investigated in another Roche-sponsored trial of patients with mild disease called MARGUERITE ROAD.
Gantenerumab’s most highly anticipated test, however, will be in the Dominantly Inherited Alzheimer Network Trial(DIAN-TU). This study is comparing gantenerumab to another antiamyloid antibody, solanezumab, and to placebo. A crucial investigation with the potential of proving the amyloid cascade hypothesis, DIAN-TU is enrolling people who are cognitively normal at baseline but are virtually guaranteed to develop early-onset Alzheimer’s because they carry a mutation in the presenilin or amyloid precursor protein gene. The hope is that the drugs will prevent or at least delay the onset of cognitive symptoms by preventing or slowing amyloid plaque formation.
Roche didn’t release any clinical or biomarker data when it ended SCARLET ROAD a year ago. During the Alzheimer’s Association International Conference (AAIC) in July, the company disclosed that there were no significant differences after 2 years of treatment between placebo and either drug dose in the primary cognitive endpoint, the Clinical Dementia Rating–sum of boxes (CDR-sb), or in any secondary endpoints: the Alzheimer’s Disease Assessment Scale–cognitive subscale 13 (ADAS-cog13), the Mini-Mental State Examination (MMSE), and the Functional Activities Questionnaire (FAQ).
Rate of disease progression proved important
However, there were signs that the 225-mg dose of gantenerumab was at least somewhat effective in the subgroup of patients who seemed destined to have more rapidly progressing disease, Roche’s Dr. Robert Lasser said at AAIC. The benefit was obscured in the overall analysis, he said, because patient progression rates varied so highly – about a third progressed rapidly, a third slowly, and a third didn’t progress at all over the 2-year period examined in the interim analysis.
While the dropout rates were similar in the three treatment arms, data presented at CTAD showed that patients in the placebo group who dropped out were progressing four times faster on the MMSE and three times faster on the ADAS-cog13 than did those who remained in the study. Dropping out probably reflected their desire to abandon a noneffective therapy and seek symptomatic treatment, which was not allowed in the study protocol, said Dr. Juergen Dukart of Roche.
Conversely, patients with rapidly progressing disease in the active groups may have responded well to the treatment, making them more likely to stay in the trial. The disproportionate retention combined with variable progression could have masked the real treatment effect, he said.
Drug exposure in rapidly progressing disease
The trial-ending interim analysis of 315 subjects didn’t include data on the 190 who had dropped out by 2 years. But outcomes presented at CTAD did include data on those subjects, and significant treatment differences appeared, especially in light of drug exposure levels. Of the fast progressers, those with higher plasma gantenerumab concentrations had less clinical decline than did those with lower drug exposure or those taking placebo, said Dr. Sylvie Retout of Roche. Among fast progressers, the ADAS-cog13 increased (indicating worsening) by 6 points in the placebo group; by 5 points in the low-exposure group; by 4 points in the medium-exposure group; and by 2 points in the high-exposure group. Similar exposure trends among fast progressers were seen on MMSE scores, but not on CDR-sb scores.
The story was different among patients who progressed slowly, Dr. Retout said. “Among slow progressers, no exposure-response relationship could be observed, indicating that the slow disease progression over the 2-year treatment period was probably limiting the detection of a drug effect,” she said.
Finding the right trial population
The problem of progression variability, dropout rates, and potentially skewed clinical outcomes is a tough nut for Alzheimer’s drug trials, Dr. Lasser said at the CTAD conference.
“This is likely to continue to surprise us if we still have this 25%-30% in our trials who don’t decline over the course of treatment. There really aren’t any big differences at baseline that stand out so that you could create the inclusion criteria to minimize this population of patients.”
Amyloid imaging, which does at least allow recruitment of a pure Alzheimer’s cohort, was not widely available when the SCARLET ROAD cohort was recruited. And although amyloid imaging is a great tool for predicting conversion to dementia from mild cognitive impairment, it’s not terribly useful as a tool for predicting progression.
Factoring baseline cerebrospinal fluid tau levels into the equation didn’t improve prediction, Dr. Lasser noted. Apolipoprotein E (APOE) allele status was similarly unhelpful.
“We didn’t find very much difference among the allele groups; there was no consistent pattern, and the between-group differences were small. So we’re beginning to hypothesize that while APOE is an excellent risk marker before cognitive decline develops, once it does start, allele status may not be as crucial in determining the patient’s eventual outcome.”
The only hints of progression speed at baseline were in the two functional measures of CDR-sb and FAQ. Fast progressers showed significantly more impairment on both of these at baseline than did slow progressers.
“Progression seems to be more likely in those who have functional decline already present as part of their prodromal presentation,” Dr. Lasser said. “At baseline, the most meaningful difference was not the MMSE, but the CDR-sb. This indicates that functional impairment may be a critical factor to look at when you’re enrolling subjects.”
On Twitter @Alz_Gal
BARCELONA – A failed phase III clinical trial for the investigational antiamyloid antibody gantenerumab in patients with Alzheimer’s disease may have had better results with a higher dose aimed at a more targeted population, new post hoc analyses of the trial’s data suggest.
The subanalyses of the now-defunct SCARLET ROAD study indicate that patients who had a more rapid progression of disease and took gantenerumab 225 mg received some cognitive benefit, especially in those who had the highest plasma concentrations.
Biomarker results also shifted toward the positive, according to data presented at the Clinical Trials in Alzheimer’s Disease conference. Amyloid PET imaging showed significantly greater plaque reduction in the brains of those with the highest gantenerumab exposure. Cerebrospinal fluid tests found that the antibody significantly decreased the level of phosphorylated tau. This, said Dr. Tania Nikolcheva, is the first evidence that attacking amyloid brain plaques can have a downstream effect on tau, which is now considered to be the major player in Alzheimer’s cognitive decline.
“We demonstrated that gantenerumab shows a consistent effect on the intended target as well as on downstream biomarkers,” said Dr. Nikolcheva of Hoffman-LaRoche, which is developing the drug. “These seem to be exposure- and dose-dependent, which supports following this trial with tests of higher dosages.”
In December 2014, Roche’s SCARLET ROAD study was halted after a 2-year interim analysis found that neither the 105 mg nor 225 mg dose conferred a significant cognitive benefit relative to placebo. But the antibody’s story is still unfolding: The drug is still being investigated in another Roche-sponsored trial of patients with mild disease called MARGUERITE ROAD.
Gantenerumab’s most highly anticipated test, however, will be in the Dominantly Inherited Alzheimer Network Trial(DIAN-TU). This study is comparing gantenerumab to another antiamyloid antibody, solanezumab, and to placebo. A crucial investigation with the potential of proving the amyloid cascade hypothesis, DIAN-TU is enrolling people who are cognitively normal at baseline but are virtually guaranteed to develop early-onset Alzheimer’s because they carry a mutation in the presenilin or amyloid precursor protein gene. The hope is that the drugs will prevent or at least delay the onset of cognitive symptoms by preventing or slowing amyloid plaque formation.
Roche didn’t release any clinical or biomarker data when it ended SCARLET ROAD a year ago. During the Alzheimer’s Association International Conference (AAIC) in July, the company disclosed that there were no significant differences after 2 years of treatment between placebo and either drug dose in the primary cognitive endpoint, the Clinical Dementia Rating–sum of boxes (CDR-sb), or in any secondary endpoints: the Alzheimer’s Disease Assessment Scale–cognitive subscale 13 (ADAS-cog13), the Mini-Mental State Examination (MMSE), and the Functional Activities Questionnaire (FAQ).
Rate of disease progression proved important
However, there were signs that the 225-mg dose of gantenerumab was at least somewhat effective in the subgroup of patients who seemed destined to have more rapidly progressing disease, Roche’s Dr. Robert Lasser said at AAIC. The benefit was obscured in the overall analysis, he said, because patient progression rates varied so highly – about a third progressed rapidly, a third slowly, and a third didn’t progress at all over the 2-year period examined in the interim analysis.
While the dropout rates were similar in the three treatment arms, data presented at CTAD showed that patients in the placebo group who dropped out were progressing four times faster on the MMSE and three times faster on the ADAS-cog13 than did those who remained in the study. Dropping out probably reflected their desire to abandon a noneffective therapy and seek symptomatic treatment, which was not allowed in the study protocol, said Dr. Juergen Dukart of Roche.
Conversely, patients with rapidly progressing disease in the active groups may have responded well to the treatment, making them more likely to stay in the trial. The disproportionate retention combined with variable progression could have masked the real treatment effect, he said.
Drug exposure in rapidly progressing disease
The trial-ending interim analysis of 315 subjects didn’t include data on the 190 who had dropped out by 2 years. But outcomes presented at CTAD did include data on those subjects, and significant treatment differences appeared, especially in light of drug exposure levels. Of the fast progressers, those with higher plasma gantenerumab concentrations had less clinical decline than did those with lower drug exposure or those taking placebo, said Dr. Sylvie Retout of Roche. Among fast progressers, the ADAS-cog13 increased (indicating worsening) by 6 points in the placebo group; by 5 points in the low-exposure group; by 4 points in the medium-exposure group; and by 2 points in the high-exposure group. Similar exposure trends among fast progressers were seen on MMSE scores, but not on CDR-sb scores.
The story was different among patients who progressed slowly, Dr. Retout said. “Among slow progressers, no exposure-response relationship could be observed, indicating that the slow disease progression over the 2-year treatment period was probably limiting the detection of a drug effect,” she said.
Finding the right trial population
The problem of progression variability, dropout rates, and potentially skewed clinical outcomes is a tough nut for Alzheimer’s drug trials, Dr. Lasser said at the CTAD conference.
“This is likely to continue to surprise us if we still have this 25%-30% in our trials who don’t decline over the course of treatment. There really aren’t any big differences at baseline that stand out so that you could create the inclusion criteria to minimize this population of patients.”
Amyloid imaging, which does at least allow recruitment of a pure Alzheimer’s cohort, was not widely available when the SCARLET ROAD cohort was recruited. And although amyloid imaging is a great tool for predicting conversion to dementia from mild cognitive impairment, it’s not terribly useful as a tool for predicting progression.
Factoring baseline cerebrospinal fluid tau levels into the equation didn’t improve prediction, Dr. Lasser noted. Apolipoprotein E (APOE) allele status was similarly unhelpful.
“We didn’t find very much difference among the allele groups; there was no consistent pattern, and the between-group differences were small. So we’re beginning to hypothesize that while APOE is an excellent risk marker before cognitive decline develops, once it does start, allele status may not be as crucial in determining the patient’s eventual outcome.”
The only hints of progression speed at baseline were in the two functional measures of CDR-sb and FAQ. Fast progressers showed significantly more impairment on both of these at baseline than did slow progressers.
“Progression seems to be more likely in those who have functional decline already present as part of their prodromal presentation,” Dr. Lasser said. “At baseline, the most meaningful difference was not the MMSE, but the CDR-sb. This indicates that functional impairment may be a critical factor to look at when you’re enrolling subjects.”
On Twitter @Alz_Gal
BARCELONA – A failed phase III clinical trial for the investigational antiamyloid antibody gantenerumab in patients with Alzheimer’s disease may have had better results with a higher dose aimed at a more targeted population, new post hoc analyses of the trial’s data suggest.
The subanalyses of the now-defunct SCARLET ROAD study indicate that patients who had a more rapid progression of disease and took gantenerumab 225 mg received some cognitive benefit, especially in those who had the highest plasma concentrations.
Biomarker results also shifted toward the positive, according to data presented at the Clinical Trials in Alzheimer’s Disease conference. Amyloid PET imaging showed significantly greater plaque reduction in the brains of those with the highest gantenerumab exposure. Cerebrospinal fluid tests found that the antibody significantly decreased the level of phosphorylated tau. This, said Dr. Tania Nikolcheva, is the first evidence that attacking amyloid brain plaques can have a downstream effect on tau, which is now considered to be the major player in Alzheimer’s cognitive decline.
“We demonstrated that gantenerumab shows a consistent effect on the intended target as well as on downstream biomarkers,” said Dr. Nikolcheva of Hoffman-LaRoche, which is developing the drug. “These seem to be exposure- and dose-dependent, which supports following this trial with tests of higher dosages.”
In December 2014, Roche’s SCARLET ROAD study was halted after a 2-year interim analysis found that neither the 105 mg nor 225 mg dose conferred a significant cognitive benefit relative to placebo. But the antibody’s story is still unfolding: The drug is still being investigated in another Roche-sponsored trial of patients with mild disease called MARGUERITE ROAD.
Gantenerumab’s most highly anticipated test, however, will be in the Dominantly Inherited Alzheimer Network Trial(DIAN-TU). This study is comparing gantenerumab to another antiamyloid antibody, solanezumab, and to placebo. A crucial investigation with the potential of proving the amyloid cascade hypothesis, DIAN-TU is enrolling people who are cognitively normal at baseline but are virtually guaranteed to develop early-onset Alzheimer’s because they carry a mutation in the presenilin or amyloid precursor protein gene. The hope is that the drugs will prevent or at least delay the onset of cognitive symptoms by preventing or slowing amyloid plaque formation.
Roche didn’t release any clinical or biomarker data when it ended SCARLET ROAD a year ago. During the Alzheimer’s Association International Conference (AAIC) in July, the company disclosed that there were no significant differences after 2 years of treatment between placebo and either drug dose in the primary cognitive endpoint, the Clinical Dementia Rating–sum of boxes (CDR-sb), or in any secondary endpoints: the Alzheimer’s Disease Assessment Scale–cognitive subscale 13 (ADAS-cog13), the Mini-Mental State Examination (MMSE), and the Functional Activities Questionnaire (FAQ).
Rate of disease progression proved important
However, there were signs that the 225-mg dose of gantenerumab was at least somewhat effective in the subgroup of patients who seemed destined to have more rapidly progressing disease, Roche’s Dr. Robert Lasser said at AAIC. The benefit was obscured in the overall analysis, he said, because patient progression rates varied so highly – about a third progressed rapidly, a third slowly, and a third didn’t progress at all over the 2-year period examined in the interim analysis.
While the dropout rates were similar in the three treatment arms, data presented at CTAD showed that patients in the placebo group who dropped out were progressing four times faster on the MMSE and three times faster on the ADAS-cog13 than did those who remained in the study. Dropping out probably reflected their desire to abandon a noneffective therapy and seek symptomatic treatment, which was not allowed in the study protocol, said Dr. Juergen Dukart of Roche.
Conversely, patients with rapidly progressing disease in the active groups may have responded well to the treatment, making them more likely to stay in the trial. The disproportionate retention combined with variable progression could have masked the real treatment effect, he said.
Drug exposure in rapidly progressing disease
The trial-ending interim analysis of 315 subjects didn’t include data on the 190 who had dropped out by 2 years. But outcomes presented at CTAD did include data on those subjects, and significant treatment differences appeared, especially in light of drug exposure levels. Of the fast progressers, those with higher plasma gantenerumab concentrations had less clinical decline than did those with lower drug exposure or those taking placebo, said Dr. Sylvie Retout of Roche. Among fast progressers, the ADAS-cog13 increased (indicating worsening) by 6 points in the placebo group; by 5 points in the low-exposure group; by 4 points in the medium-exposure group; and by 2 points in the high-exposure group. Similar exposure trends among fast progressers were seen on MMSE scores, but not on CDR-sb scores.
The story was different among patients who progressed slowly, Dr. Retout said. “Among slow progressers, no exposure-response relationship could be observed, indicating that the slow disease progression over the 2-year treatment period was probably limiting the detection of a drug effect,” she said.
Finding the right trial population
The problem of progression variability, dropout rates, and potentially skewed clinical outcomes is a tough nut for Alzheimer’s drug trials, Dr. Lasser said at the CTAD conference.
“This is likely to continue to surprise us if we still have this 25%-30% in our trials who don’t decline over the course of treatment. There really aren’t any big differences at baseline that stand out so that you could create the inclusion criteria to minimize this population of patients.”
Amyloid imaging, which does at least allow recruitment of a pure Alzheimer’s cohort, was not widely available when the SCARLET ROAD cohort was recruited. And although amyloid imaging is a great tool for predicting conversion to dementia from mild cognitive impairment, it’s not terribly useful as a tool for predicting progression.
Factoring baseline cerebrospinal fluid tau levels into the equation didn’t improve prediction, Dr. Lasser noted. Apolipoprotein E (APOE) allele status was similarly unhelpful.
“We didn’t find very much difference among the allele groups; there was no consistent pattern, and the between-group differences were small. So we’re beginning to hypothesize that while APOE is an excellent risk marker before cognitive decline develops, once it does start, allele status may not be as crucial in determining the patient’s eventual outcome.”
The only hints of progression speed at baseline were in the two functional measures of CDR-sb and FAQ. Fast progressers showed significantly more impairment on both of these at baseline than did slow progressers.
“Progression seems to be more likely in those who have functional decline already present as part of their prodromal presentation,” Dr. Lasser said. “At baseline, the most meaningful difference was not the MMSE, but the CDR-sb. This indicates that functional impairment may be a critical factor to look at when you’re enrolling subjects.”
On Twitter @Alz_Gal
AT CTAD
Intrauterine palpator plus 3-D ultrasound boost metroplasty success
LAS VEGAS – The combination of three-dimensional ultrasound and a graduated intrauterine palpator significantly increased the rate of complete metroplasties in a group of women with unexplained infertility or repeat miscarriage.
The instrument, which is marked in millimeters, allows the surgeon to more accurately gauge how much of a uterine septum is removed during the procedure, as well as approximate the shape of what is left. Combined with transvaginal ultrasound visualization, the technique makes a complete, but not overly aggressive, resection more likely, Dr. Attilio Di Spiezio Sardo said at the meeting sponsored by the AAGL.
“One of man’s dilemmas in everyday life is knowing when to stop,” said Dr. Sardo of the University of Naples “Federico II,” Italy. “The same applies to hysteroscopic metroplasties. We still don’t know where to end the incision in order to avoid complications, significant bleeding, or perforation, and in order to avoid abnormal anatomical results.”
The new data he presented builds on his 2009 work, which used 3-D transvaginal ultrasound as the basis for a new subclassification system for uterine anomalies to be used during in-office hysteroscopy.
The aim of the current study was to assess whether the addition of a 5 French graduated intrauterine palpator could improve the accuracy of hysteroscopic metroplasty, compared with that obtained without the instrument. Anatomic results were assessed by 3-D transvaginal ultrasound and second-look hysteroscopy and classified as complete (residual septum less than 5 mm), suboptimal (residual septum 5 mm-10 mm), or incomplete (residual septum greater than 10 mm).
All procedures were performed with the same initial technique under conscious sedation with a 5 mm hysteroscope and miniaturized 5 French instruments. First, the surgeon used a bipolar electrode to remove three-quarters of the septum. Blunt scissors were then used to refine the septal base.
In the intervention group, however, the intrauterine palpator was used to measure the portion of the remaining septum. The metroplasty was stopped when the intrauterine palpator showed that the resected septum corresponded to the presurgical ultrasonographic measures, and had a fundal notch of 1 cm. In the control group, the procedure was stopped when the tubal ostia were clearly visible on the same line and/or hemorrhage from small myometrial vessels of the fundus occurred.
The mean procedural time was similar in the palpator and control groups (12.6 minutes vs. 11.7 minutes). There was one postsurgical intrauterine adhesion in each group.
There were significantly more complete resections in the palpator group than the control group (71% vs. 41%), although the number of suboptimal resections was similar (28% vs. 20%). There were 12 incomplete resections, all of which were in the control group. There was no correlation between septal length and the completeness of resection, Dr. Sardo added.
The combination of 3-D transvaginal ultrasound and a graduated palpator to physically explore the intrauterine space should help improve outcomes in what can be a frustrating procedure, he said.
“I used to think my metroplasties were perfect, and then I reviewed the videos and ultrasounds and sometimes saw that part of the septum was still there,” Dr. Sardo said. “After endometrial ablation, I think hysteroscopic metroplasty is one of the most frustrating problems” for a gynecologic surgeon.
Dr. Sardo reported having no financial disclosures.
LAS VEGAS – The combination of three-dimensional ultrasound and a graduated intrauterine palpator significantly increased the rate of complete metroplasties in a group of women with unexplained infertility or repeat miscarriage.
The instrument, which is marked in millimeters, allows the surgeon to more accurately gauge how much of a uterine septum is removed during the procedure, as well as approximate the shape of what is left. Combined with transvaginal ultrasound visualization, the technique makes a complete, but not overly aggressive, resection more likely, Dr. Attilio Di Spiezio Sardo said at the meeting sponsored by the AAGL.
“One of man’s dilemmas in everyday life is knowing when to stop,” said Dr. Sardo of the University of Naples “Federico II,” Italy. “The same applies to hysteroscopic metroplasties. We still don’t know where to end the incision in order to avoid complications, significant bleeding, or perforation, and in order to avoid abnormal anatomical results.”
The new data he presented builds on his 2009 work, which used 3-D transvaginal ultrasound as the basis for a new subclassification system for uterine anomalies to be used during in-office hysteroscopy.
The aim of the current study was to assess whether the addition of a 5 French graduated intrauterine palpator could improve the accuracy of hysteroscopic metroplasty, compared with that obtained without the instrument. Anatomic results were assessed by 3-D transvaginal ultrasound and second-look hysteroscopy and classified as complete (residual septum less than 5 mm), suboptimal (residual septum 5 mm-10 mm), or incomplete (residual septum greater than 10 mm).
All procedures were performed with the same initial technique under conscious sedation with a 5 mm hysteroscope and miniaturized 5 French instruments. First, the surgeon used a bipolar electrode to remove three-quarters of the septum. Blunt scissors were then used to refine the septal base.
In the intervention group, however, the intrauterine palpator was used to measure the portion of the remaining septum. The metroplasty was stopped when the intrauterine palpator showed that the resected septum corresponded to the presurgical ultrasonographic measures, and had a fundal notch of 1 cm. In the control group, the procedure was stopped when the tubal ostia were clearly visible on the same line and/or hemorrhage from small myometrial vessels of the fundus occurred.
The mean procedural time was similar in the palpator and control groups (12.6 minutes vs. 11.7 minutes). There was one postsurgical intrauterine adhesion in each group.
There were significantly more complete resections in the palpator group than the control group (71% vs. 41%), although the number of suboptimal resections was similar (28% vs. 20%). There were 12 incomplete resections, all of which were in the control group. There was no correlation between septal length and the completeness of resection, Dr. Sardo added.
The combination of 3-D transvaginal ultrasound and a graduated palpator to physically explore the intrauterine space should help improve outcomes in what can be a frustrating procedure, he said.
“I used to think my metroplasties were perfect, and then I reviewed the videos and ultrasounds and sometimes saw that part of the septum was still there,” Dr. Sardo said. “After endometrial ablation, I think hysteroscopic metroplasty is one of the most frustrating problems” for a gynecologic surgeon.
Dr. Sardo reported having no financial disclosures.
LAS VEGAS – The combination of three-dimensional ultrasound and a graduated intrauterine palpator significantly increased the rate of complete metroplasties in a group of women with unexplained infertility or repeat miscarriage.
The instrument, which is marked in millimeters, allows the surgeon to more accurately gauge how much of a uterine septum is removed during the procedure, as well as approximate the shape of what is left. Combined with transvaginal ultrasound visualization, the technique makes a complete, but not overly aggressive, resection more likely, Dr. Attilio Di Spiezio Sardo said at the meeting sponsored by the AAGL.
“One of man’s dilemmas in everyday life is knowing when to stop,” said Dr. Sardo of the University of Naples “Federico II,” Italy. “The same applies to hysteroscopic metroplasties. We still don’t know where to end the incision in order to avoid complications, significant bleeding, or perforation, and in order to avoid abnormal anatomical results.”
The new data he presented builds on his 2009 work, which used 3-D transvaginal ultrasound as the basis for a new subclassification system for uterine anomalies to be used during in-office hysteroscopy.
The aim of the current study was to assess whether the addition of a 5 French graduated intrauterine palpator could improve the accuracy of hysteroscopic metroplasty, compared with that obtained without the instrument. Anatomic results were assessed by 3-D transvaginal ultrasound and second-look hysteroscopy and classified as complete (residual septum less than 5 mm), suboptimal (residual septum 5 mm-10 mm), or incomplete (residual septum greater than 10 mm).
All procedures were performed with the same initial technique under conscious sedation with a 5 mm hysteroscope and miniaturized 5 French instruments. First, the surgeon used a bipolar electrode to remove three-quarters of the septum. Blunt scissors were then used to refine the septal base.
In the intervention group, however, the intrauterine palpator was used to measure the portion of the remaining septum. The metroplasty was stopped when the intrauterine palpator showed that the resected septum corresponded to the presurgical ultrasonographic measures, and had a fundal notch of 1 cm. In the control group, the procedure was stopped when the tubal ostia were clearly visible on the same line and/or hemorrhage from small myometrial vessels of the fundus occurred.
The mean procedural time was similar in the palpator and control groups (12.6 minutes vs. 11.7 minutes). There was one postsurgical intrauterine adhesion in each group.
There were significantly more complete resections in the palpator group than the control group (71% vs. 41%), although the number of suboptimal resections was similar (28% vs. 20%). There were 12 incomplete resections, all of which were in the control group. There was no correlation between septal length and the completeness of resection, Dr. Sardo added.
The combination of 3-D transvaginal ultrasound and a graduated palpator to physically explore the intrauterine space should help improve outcomes in what can be a frustrating procedure, he said.
“I used to think my metroplasties were perfect, and then I reviewed the videos and ultrasounds and sometimes saw that part of the septum was still there,” Dr. Sardo said. “After endometrial ablation, I think hysteroscopic metroplasty is one of the most frustrating problems” for a gynecologic surgeon.
Dr. Sardo reported having no financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Key clinical point: Using a novel, graduated palpator along with transvaginal ultrasound increased the likelihood of a successful metroplasty.
Major finding: A successful procedure occurred in 71% of the intervention group and 41% of the control group.
Data source: A randomized study of 90 women.
Disclosures: Dr. Sardo reported having no financial disclosures.
CTAD: Anti-tau vaccine demonstrated safety, immunogenicity in phase I study
BARCELONA – An investigational anti-tau vaccine has passed its first big clinical milestone with a successful phase I trial of 30 patients with mild to moderate Alzheimer’s disease.
The vaccine, AADvac-1, was safe and induced a robust immune response in the study. Patients who received the vaccine generally showed stable cognition over 6 months, although their trajectories were variable and the follow-up was short, Dr. Matej Ondrus said at the Clinical Trials in Alzheimer’s Disease conference.
The results in all three areas examined – safety, immunogenicity, and cognition – were good enough to propel AADvac-1 into aphase II study, scheduled for launch in early 2016, said Dr. Ondrus, medical director of Axon Neuroscience, the vaccine’s developer.
Dr. Ondrus refrained from overemphasizing the importance of such early cognitive findings. “We must stress that the variability was high and that this trial was not powered to show cognition. However, we did observe that cognition remained stable,” he said.
AADvac-1 is an active vaccine that targets neurofibrillary tau tangles. According to the company, it induces antibodies against a pathological conformation in the microtubule binding domain of diseased tau protein. An emerging body of evidence suggests that tau aggregation, rather than amyloid plaque formation, is directly linked to cognitive decline in Alzheimer’s. Only one other anti-tau vaccine, (ACI-35), is being investigated.
Patients in the phase I trial were randomized at four Austrian sites to either placebo (n = 6) or to the active vaccine (n = 24), in six monthly infusions. After 3 months of treatment, patients in the placebo arm crossed over to the active vaccine. Two patients dropped out, leaving 28 to finish the study. Of these, 25 have entered the 18-month follow-up study.
At baseline, patients were a mean age of 68 years and had a mean score of 20 on the Mini-Mental State Exam. Most (19) were taking an acetylcholinesterase inhibitor, including 11 in conjunction with memantine. One was on memantine monotherapy.
Infusion reactions were common, occurring in 43% of patients; 98% of these were mild.
There were five serious adverse events, all of them different. Two patients dropped out because of a serious adverse event, but only one – a viral infection followed by epileptic seizure – was judged as possibly related to the active vaccine.
There were three abnormal findings on brain imaging, Dr. Ondrus said. Two of these were expected brain changes for the study population and not reported as adverse events. The third finding was of two new microbleeds in a patient who had already experienced microbleeds at baseline. These were mild and not accompanied by any clinical symptoms, but nonetheless, were reported as an adverse event.
There were no differences seen on electrocardiogram and no physical manifestations of cardiovascular effects, he added.
AADvac-1 induced a robust immune response in 29 patients, which was seen after the second infusion and continued to mount thereafter. One patient reached maximum titer after two infusions; the rest reached it after six infusions. The average geometric mean titer after infusion six was 1:52,000.
The main cognitive endpoint was the Alzheimer’s Disease Assessment Scale–Cognitive Subscale. Dr. Ondrus showed data indicating that the vaccine was associated with a stabilizing of cognition, although the company did not conduct a statistical analysis of the data. He also noted that the company is looking for associations between cognitive results and individual antibody titer levels.
Only four patients contributed cerebrospinal fluid at baseline and study’s end, he added. Those samples are currently being analyzed.
On Twitter @alz_gal
BARCELONA – An investigational anti-tau vaccine has passed its first big clinical milestone with a successful phase I trial of 30 patients with mild to moderate Alzheimer’s disease.
The vaccine, AADvac-1, was safe and induced a robust immune response in the study. Patients who received the vaccine generally showed stable cognition over 6 months, although their trajectories were variable and the follow-up was short, Dr. Matej Ondrus said at the Clinical Trials in Alzheimer’s Disease conference.
The results in all three areas examined – safety, immunogenicity, and cognition – were good enough to propel AADvac-1 into aphase II study, scheduled for launch in early 2016, said Dr. Ondrus, medical director of Axon Neuroscience, the vaccine’s developer.
Dr. Ondrus refrained from overemphasizing the importance of such early cognitive findings. “We must stress that the variability was high and that this trial was not powered to show cognition. However, we did observe that cognition remained stable,” he said.
AADvac-1 is an active vaccine that targets neurofibrillary tau tangles. According to the company, it induces antibodies against a pathological conformation in the microtubule binding domain of diseased tau protein. An emerging body of evidence suggests that tau aggregation, rather than amyloid plaque formation, is directly linked to cognitive decline in Alzheimer’s. Only one other anti-tau vaccine, (ACI-35), is being investigated.
Patients in the phase I trial were randomized at four Austrian sites to either placebo (n = 6) or to the active vaccine (n = 24), in six monthly infusions. After 3 months of treatment, patients in the placebo arm crossed over to the active vaccine. Two patients dropped out, leaving 28 to finish the study. Of these, 25 have entered the 18-month follow-up study.
At baseline, patients were a mean age of 68 years and had a mean score of 20 on the Mini-Mental State Exam. Most (19) were taking an acetylcholinesterase inhibitor, including 11 in conjunction with memantine. One was on memantine monotherapy.
Infusion reactions were common, occurring in 43% of patients; 98% of these were mild.
There were five serious adverse events, all of them different. Two patients dropped out because of a serious adverse event, but only one – a viral infection followed by epileptic seizure – was judged as possibly related to the active vaccine.
There were three abnormal findings on brain imaging, Dr. Ondrus said. Two of these were expected brain changes for the study population and not reported as adverse events. The third finding was of two new microbleeds in a patient who had already experienced microbleeds at baseline. These were mild and not accompanied by any clinical symptoms, but nonetheless, were reported as an adverse event.
There were no differences seen on electrocardiogram and no physical manifestations of cardiovascular effects, he added.
AADvac-1 induced a robust immune response in 29 patients, which was seen after the second infusion and continued to mount thereafter. One patient reached maximum titer after two infusions; the rest reached it after six infusions. The average geometric mean titer after infusion six was 1:52,000.
The main cognitive endpoint was the Alzheimer’s Disease Assessment Scale–Cognitive Subscale. Dr. Ondrus showed data indicating that the vaccine was associated with a stabilizing of cognition, although the company did not conduct a statistical analysis of the data. He also noted that the company is looking for associations between cognitive results and individual antibody titer levels.
Only four patients contributed cerebrospinal fluid at baseline and study’s end, he added. Those samples are currently being analyzed.
On Twitter @alz_gal
BARCELONA – An investigational anti-tau vaccine has passed its first big clinical milestone with a successful phase I trial of 30 patients with mild to moderate Alzheimer’s disease.
The vaccine, AADvac-1, was safe and induced a robust immune response in the study. Patients who received the vaccine generally showed stable cognition over 6 months, although their trajectories were variable and the follow-up was short, Dr. Matej Ondrus said at the Clinical Trials in Alzheimer’s Disease conference.
The results in all three areas examined – safety, immunogenicity, and cognition – were good enough to propel AADvac-1 into aphase II study, scheduled for launch in early 2016, said Dr. Ondrus, medical director of Axon Neuroscience, the vaccine’s developer.
Dr. Ondrus refrained from overemphasizing the importance of such early cognitive findings. “We must stress that the variability was high and that this trial was not powered to show cognition. However, we did observe that cognition remained stable,” he said.
AADvac-1 is an active vaccine that targets neurofibrillary tau tangles. According to the company, it induces antibodies against a pathological conformation in the microtubule binding domain of diseased tau protein. An emerging body of evidence suggests that tau aggregation, rather than amyloid plaque formation, is directly linked to cognitive decline in Alzheimer’s. Only one other anti-tau vaccine, (ACI-35), is being investigated.
Patients in the phase I trial were randomized at four Austrian sites to either placebo (n = 6) or to the active vaccine (n = 24), in six monthly infusions. After 3 months of treatment, patients in the placebo arm crossed over to the active vaccine. Two patients dropped out, leaving 28 to finish the study. Of these, 25 have entered the 18-month follow-up study.
At baseline, patients were a mean age of 68 years and had a mean score of 20 on the Mini-Mental State Exam. Most (19) were taking an acetylcholinesterase inhibitor, including 11 in conjunction with memantine. One was on memantine monotherapy.
Infusion reactions were common, occurring in 43% of patients; 98% of these were mild.
There were five serious adverse events, all of them different. Two patients dropped out because of a serious adverse event, but only one – a viral infection followed by epileptic seizure – was judged as possibly related to the active vaccine.
There were three abnormal findings on brain imaging, Dr. Ondrus said. Two of these were expected brain changes for the study population and not reported as adverse events. The third finding was of two new microbleeds in a patient who had already experienced microbleeds at baseline. These were mild and not accompanied by any clinical symptoms, but nonetheless, were reported as an adverse event.
There were no differences seen on electrocardiogram and no physical manifestations of cardiovascular effects, he added.
AADvac-1 induced a robust immune response in 29 patients, which was seen after the second infusion and continued to mount thereafter. One patient reached maximum titer after two infusions; the rest reached it after six infusions. The average geometric mean titer after infusion six was 1:52,000.
The main cognitive endpoint was the Alzheimer’s Disease Assessment Scale–Cognitive Subscale. Dr. Ondrus showed data indicating that the vaccine was associated with a stabilizing of cognition, although the company did not conduct a statistical analysis of the data. He also noted that the company is looking for associations between cognitive results and individual antibody titer levels.
Only four patients contributed cerebrospinal fluid at baseline and study’s end, he added. Those samples are currently being analyzed.
On Twitter @alz_gal
AT CTAD
Key clinical point: An anti-tau vaccine has been judged safe and will advance to phase II testing.
Major finding: AADvac-1 induced a robust immune response in 29 of 30 patients, and was associated with stable cognition over 6 months.
Data source: A placebo-controlled crossover trial of 30 patients with mild-moderate Alzheimer’s disease.
Disclosures: Axon Neuroscience is developing the vaccine and sponsored the study. Dr. Ondrus is the company’s medical director.
Treatment options for adenomyosis supported by limited high-quality data
LAS VEGAS – Levonorgestrel and hysterectomy are probably the best treatments for adenomyosis – or at least the most well-supported medical and surgical options.
There are other therapies, Dr. Jason Abbott said at a meeting sponsored by AAGL, but almost every treatment has limited high-quality data.
Hysterectomy cures the problem of pain and uterine bleeding completely with no serious adverse effects. But it’s invasive and irreversible, said Dr. Abbott of the Royal Hospital for Women, Australia. And many women, no matter how bad their symptoms or where they are in their reproductive life, simply don’t want to give up their uterus.
“Hysterectomy, as a therapy for adenomyosis, is beautiful,” Dr. Abbott said. “We love it because it cures the condition – but it’s an end game, for sure.”
Intrauterine levonorgestrel systems are probably the best-supported medical therapy, he said.
“The levonorgestrel intrauterine system does work and there are a number of randomized controlled trials comparing this to hysterectomy,” Dr. Abbott said. “It controls adenomyosis very well, and if you look at iron and anemia outcomes a year after treatment initiation, there is no difference – so that is a very good outcome.”
Levonorgestrel is also fully reversible – a must for women who want to conceive in the future. And it’s cheap, especially compared to surgery.
“Even if you allow for the 18%-20% who won’t like it and have it removed, then there’s a pretty good chance you’ll still come up on the positive side compared to hysterectomy, even if you have to repeat the 5-year course two, three, or four times,” he said.
While there are no economic data comparing it directly to hysterectomy, conclusions can be extrapolated from studies that compared endometrial ablation and the surgery.
“You need a 40% failure of ablation for the levonorgestrel to be less economically effective, because then you are getting into having two procedures,” he said. “With levonorgestrel, we have a much lower 20% failure rate and you’re already starting with a much lower cost. So I would say this is entirely reasonable, and a very good place to start. We should continue to offer this to our patients.”
Dr. Abbott noted other treatment options as well:
• Danazol
“This has been around since the 1970s and it works. But women absolutely hate it. They don’t want to look or sound like a man,” Dr. Abbott said. “It’s fallen pretty much by the wayside and [is] only used when we are absolutely desperate.”
There are, however, data suggesting that contraceptive devices loaded with danazol have much less systemic absorption and can be effective without the androgenic effects. A 2010 case series of 35 infertile women found that both a cervical ring and intrauterine device effected endometrial atrophy but did not inhibit ovulation; 13% of those using the IUD and 66% of those using the ring were able to conceive.
• Gonadotropin-releasing hormone agonists
“These are used for a lot of issues with endometriosis, but there’s only one randomized controlled trial on adenomyosis, which compared it with aromatase inhibitors,” Dr. Abbott said. “Both were effective in reducing the symptoms. Once you induce amenorrhea, you don’t have pain with periods.”
There are also studies comparing GnRH agonists with oral contraceptives in adenomyosis. “They do work equally well, but the problem is these are only short-term studies [6-24 months]. Once you stop, your symptoms do come back, and women could have 10-15 years before menopause stops the problem. So this is a short-term solution to a long-term problem,” he said.
The GnRH agonist side effect profile can be problematic, he added.
• Oral contraceptives
“The pill is fantastic and we all use it to control abnormal bleeding. It’s cheap, and it’s been around forever. However, a lot of women now don’t want to use any hormones in any way, shape, or form. There are also no randomized data for its use in adenomyosis,” Dr. Abbott said. “I think pragmatically, yes, it can give very good symptom control and if there’s no direct evidence against it, what’s the worst that can happen? They don’t like it and then you take them off. I think it’s a perfectly reasonable option.”
• Ablation
There are no randomized data supporting endometrial ablation in adenomyosis. “What we’ve got are the randomized data in studies of abnormal uterine bleeding, which suggest a 25% failure rate,” he said. “I think it’s a reasonable procedure for adenomyosis.”
• Resection
Surgery for adenomyosis is very difficult to perform and carries some not inconsiderable risks. “You’re removing a big chunk of myometrium and you’re never sure if you’re getting it all anyway, so there’s a big chance of persistence and recurrence,” Dr. Abbott said. There’s also the chance that the surgery will introduce abnormal tissue into unaffected myometrium, he added.
And while there aren’t a lot of data out there on obstetric outcomes, the current literature suggests a 2% rate of uterine rupture even before labor and delivery. “But it doesn’t remove the uterus, so that’s something to consider,” he said. “It is an option that’s out there.”
Dr. Abbott reported that he has consulted with Bayer on the Mirena levonorgestrel intrauterine system.
LAS VEGAS – Levonorgestrel and hysterectomy are probably the best treatments for adenomyosis – or at least the most well-supported medical and surgical options.
There are other therapies, Dr. Jason Abbott said at a meeting sponsored by AAGL, but almost every treatment has limited high-quality data.
Hysterectomy cures the problem of pain and uterine bleeding completely with no serious adverse effects. But it’s invasive and irreversible, said Dr. Abbott of the Royal Hospital for Women, Australia. And many women, no matter how bad their symptoms or where they are in their reproductive life, simply don’t want to give up their uterus.
“Hysterectomy, as a therapy for adenomyosis, is beautiful,” Dr. Abbott said. “We love it because it cures the condition – but it’s an end game, for sure.”
Intrauterine levonorgestrel systems are probably the best-supported medical therapy, he said.
“The levonorgestrel intrauterine system does work and there are a number of randomized controlled trials comparing this to hysterectomy,” Dr. Abbott said. “It controls adenomyosis very well, and if you look at iron and anemia outcomes a year after treatment initiation, there is no difference – so that is a very good outcome.”
Levonorgestrel is also fully reversible – a must for women who want to conceive in the future. And it’s cheap, especially compared to surgery.
“Even if you allow for the 18%-20% who won’t like it and have it removed, then there’s a pretty good chance you’ll still come up on the positive side compared to hysterectomy, even if you have to repeat the 5-year course two, three, or four times,” he said.
While there are no economic data comparing it directly to hysterectomy, conclusions can be extrapolated from studies that compared endometrial ablation and the surgery.
“You need a 40% failure of ablation for the levonorgestrel to be less economically effective, because then you are getting into having two procedures,” he said. “With levonorgestrel, we have a much lower 20% failure rate and you’re already starting with a much lower cost. So I would say this is entirely reasonable, and a very good place to start. We should continue to offer this to our patients.”
Dr. Abbott noted other treatment options as well:
• Danazol
“This has been around since the 1970s and it works. But women absolutely hate it. They don’t want to look or sound like a man,” Dr. Abbott said. “It’s fallen pretty much by the wayside and [is] only used when we are absolutely desperate.”
There are, however, data suggesting that contraceptive devices loaded with danazol have much less systemic absorption and can be effective without the androgenic effects. A 2010 case series of 35 infertile women found that both a cervical ring and intrauterine device effected endometrial atrophy but did not inhibit ovulation; 13% of those using the IUD and 66% of those using the ring were able to conceive.
• Gonadotropin-releasing hormone agonists
“These are used for a lot of issues with endometriosis, but there’s only one randomized controlled trial on adenomyosis, which compared it with aromatase inhibitors,” Dr. Abbott said. “Both were effective in reducing the symptoms. Once you induce amenorrhea, you don’t have pain with periods.”
There are also studies comparing GnRH agonists with oral contraceptives in adenomyosis. “They do work equally well, but the problem is these are only short-term studies [6-24 months]. Once you stop, your symptoms do come back, and women could have 10-15 years before menopause stops the problem. So this is a short-term solution to a long-term problem,” he said.
The GnRH agonist side effect profile can be problematic, he added.
• Oral contraceptives
“The pill is fantastic and we all use it to control abnormal bleeding. It’s cheap, and it’s been around forever. However, a lot of women now don’t want to use any hormones in any way, shape, or form. There are also no randomized data for its use in adenomyosis,” Dr. Abbott said. “I think pragmatically, yes, it can give very good symptom control and if there’s no direct evidence against it, what’s the worst that can happen? They don’t like it and then you take them off. I think it’s a perfectly reasonable option.”
• Ablation
There are no randomized data supporting endometrial ablation in adenomyosis. “What we’ve got are the randomized data in studies of abnormal uterine bleeding, which suggest a 25% failure rate,” he said. “I think it’s a reasonable procedure for adenomyosis.”
• Resection
Surgery for adenomyosis is very difficult to perform and carries some not inconsiderable risks. “You’re removing a big chunk of myometrium and you’re never sure if you’re getting it all anyway, so there’s a big chance of persistence and recurrence,” Dr. Abbott said. There’s also the chance that the surgery will introduce abnormal tissue into unaffected myometrium, he added.
And while there aren’t a lot of data out there on obstetric outcomes, the current literature suggests a 2% rate of uterine rupture even before labor and delivery. “But it doesn’t remove the uterus, so that’s something to consider,” he said. “It is an option that’s out there.”
Dr. Abbott reported that he has consulted with Bayer on the Mirena levonorgestrel intrauterine system.
LAS VEGAS – Levonorgestrel and hysterectomy are probably the best treatments for adenomyosis – or at least the most well-supported medical and surgical options.
There are other therapies, Dr. Jason Abbott said at a meeting sponsored by AAGL, but almost every treatment has limited high-quality data.
Hysterectomy cures the problem of pain and uterine bleeding completely with no serious adverse effects. But it’s invasive and irreversible, said Dr. Abbott of the Royal Hospital for Women, Australia. And many women, no matter how bad their symptoms or where they are in their reproductive life, simply don’t want to give up their uterus.
“Hysterectomy, as a therapy for adenomyosis, is beautiful,” Dr. Abbott said. “We love it because it cures the condition – but it’s an end game, for sure.”
Intrauterine levonorgestrel systems are probably the best-supported medical therapy, he said.
“The levonorgestrel intrauterine system does work and there are a number of randomized controlled trials comparing this to hysterectomy,” Dr. Abbott said. “It controls adenomyosis very well, and if you look at iron and anemia outcomes a year after treatment initiation, there is no difference – so that is a very good outcome.”
Levonorgestrel is also fully reversible – a must for women who want to conceive in the future. And it’s cheap, especially compared to surgery.
“Even if you allow for the 18%-20% who won’t like it and have it removed, then there’s a pretty good chance you’ll still come up on the positive side compared to hysterectomy, even if you have to repeat the 5-year course two, three, or four times,” he said.
While there are no economic data comparing it directly to hysterectomy, conclusions can be extrapolated from studies that compared endometrial ablation and the surgery.
“You need a 40% failure of ablation for the levonorgestrel to be less economically effective, because then you are getting into having two procedures,” he said. “With levonorgestrel, we have a much lower 20% failure rate and you’re already starting with a much lower cost. So I would say this is entirely reasonable, and a very good place to start. We should continue to offer this to our patients.”
Dr. Abbott noted other treatment options as well:
• Danazol
“This has been around since the 1970s and it works. But women absolutely hate it. They don’t want to look or sound like a man,” Dr. Abbott said. “It’s fallen pretty much by the wayside and [is] only used when we are absolutely desperate.”
There are, however, data suggesting that contraceptive devices loaded with danazol have much less systemic absorption and can be effective without the androgenic effects. A 2010 case series of 35 infertile women found that both a cervical ring and intrauterine device effected endometrial atrophy but did not inhibit ovulation; 13% of those using the IUD and 66% of those using the ring were able to conceive.
• Gonadotropin-releasing hormone agonists
“These are used for a lot of issues with endometriosis, but there’s only one randomized controlled trial on adenomyosis, which compared it with aromatase inhibitors,” Dr. Abbott said. “Both were effective in reducing the symptoms. Once you induce amenorrhea, you don’t have pain with periods.”
There are also studies comparing GnRH agonists with oral contraceptives in adenomyosis. “They do work equally well, but the problem is these are only short-term studies [6-24 months]. Once you stop, your symptoms do come back, and women could have 10-15 years before menopause stops the problem. So this is a short-term solution to a long-term problem,” he said.
The GnRH agonist side effect profile can be problematic, he added.
• Oral contraceptives
“The pill is fantastic and we all use it to control abnormal bleeding. It’s cheap, and it’s been around forever. However, a lot of women now don’t want to use any hormones in any way, shape, or form. There are also no randomized data for its use in adenomyosis,” Dr. Abbott said. “I think pragmatically, yes, it can give very good symptom control and if there’s no direct evidence against it, what’s the worst that can happen? They don’t like it and then you take them off. I think it’s a perfectly reasonable option.”
• Ablation
There are no randomized data supporting endometrial ablation in adenomyosis. “What we’ve got are the randomized data in studies of abnormal uterine bleeding, which suggest a 25% failure rate,” he said. “I think it’s a reasonable procedure for adenomyosis.”
• Resection
Surgery for adenomyosis is very difficult to perform and carries some not inconsiderable risks. “You’re removing a big chunk of myometrium and you’re never sure if you’re getting it all anyway, so there’s a big chance of persistence and recurrence,” Dr. Abbott said. There’s also the chance that the surgery will introduce abnormal tissue into unaffected myometrium, he added.
And while there aren’t a lot of data out there on obstetric outcomes, the current literature suggests a 2% rate of uterine rupture even before labor and delivery. “But it doesn’t remove the uterus, so that’s something to consider,” he said. “It is an option that’s out there.”
Dr. Abbott reported that he has consulted with Bayer on the Mirena levonorgestrel intrauterine system.
EXPERT ANALYSIS FROM THE AAGL GLOBAL CONGRESS
Adhesion barriers linked to complications in myomectomy, hysterectomy
LAS VEGAS – Adhesion barriers may cause more problems than they prevent when employed in myomectomy and hysterectomy.
Ileus, fever, hematomas, and even small bowel obstruction occurred significantly more often when the barriers were used in conjunction with those procedures – especially when performed as open surgeries, Dr. François Closon reported at the AAGL Global Congress. And although their use remains relatively small among these patients, it’s been steadily rising since 2003, he said.
“Adhesion barriers are also being used about eight times more often in myomectomies than in hysterectomies, and this seems to be going up every year,” Dr. Closon of McGill University, Montreal, said at the meeting sponsored by AAGL.
His retrospective study drew on the U.S. Healthcare Cost and Utilization Project Nationwide Inpatient Sample, and examined the perioperative outcomes of adhesion barriers among nearly 475,000 women who underwent myomectomy or hysterectomy during 2003-2011. Of these, 9,000 (about 2%) had an adhesion barrier applied during the surgery. There were no significant baseline differences between the groups, except for age. Women in the myomectomy group were younger than those in the hysterectomy group (mean 36 years vs. 46 years). Age, however, was not associated with the use of an adhesion barrier.
Dr. Closon found a significant linear increase in the use of barriers in both procedures over the study period. In hysterectomy, barrier use rose slightly, but significantly, from 0.37% in 2003 to 2% in 2011. The increase was much greater for use in myomectomy, which rose from 3% to 7% during the same period.
Ileus was significantly more common among myomectomies that used the barrier (4% vs. 3%; OR, 1.49). The difference was more pronounced with hysterectomies (5% vs. 2.5%; OR, 1.97).
Small bowel obstruction was rare, but significantly more common in hysterectomies that employed adhesion barriers (0.2% vs. 0.4%; OR, 1.89).
Fever was also significantly more common in both procedures when the barrier was used (myomectomy 4.4% vs. 3%; OR, 1.43; hysterectomy 2.5% vs. 1.6%; OR, 1.65).
“The odds of intra-abdominal hematoma, postoperative pain, and a length of stay of 3 or more days were also significantly higher when an adhesion barrier was applied,” Dr. Closon said.
He reported having no financial disclosures.
LAS VEGAS – Adhesion barriers may cause more problems than they prevent when employed in myomectomy and hysterectomy.
Ileus, fever, hematomas, and even small bowel obstruction occurred significantly more often when the barriers were used in conjunction with those procedures – especially when performed as open surgeries, Dr. François Closon reported at the AAGL Global Congress. And although their use remains relatively small among these patients, it’s been steadily rising since 2003, he said.
“Adhesion barriers are also being used about eight times more often in myomectomies than in hysterectomies, and this seems to be going up every year,” Dr. Closon of McGill University, Montreal, said at the meeting sponsored by AAGL.
His retrospective study drew on the U.S. Healthcare Cost and Utilization Project Nationwide Inpatient Sample, and examined the perioperative outcomes of adhesion barriers among nearly 475,000 women who underwent myomectomy or hysterectomy during 2003-2011. Of these, 9,000 (about 2%) had an adhesion barrier applied during the surgery. There were no significant baseline differences between the groups, except for age. Women in the myomectomy group were younger than those in the hysterectomy group (mean 36 years vs. 46 years). Age, however, was not associated with the use of an adhesion barrier.
Dr. Closon found a significant linear increase in the use of barriers in both procedures over the study period. In hysterectomy, barrier use rose slightly, but significantly, from 0.37% in 2003 to 2% in 2011. The increase was much greater for use in myomectomy, which rose from 3% to 7% during the same period.
Ileus was significantly more common among myomectomies that used the barrier (4% vs. 3%; OR, 1.49). The difference was more pronounced with hysterectomies (5% vs. 2.5%; OR, 1.97).
Small bowel obstruction was rare, but significantly more common in hysterectomies that employed adhesion barriers (0.2% vs. 0.4%; OR, 1.89).
Fever was also significantly more common in both procedures when the barrier was used (myomectomy 4.4% vs. 3%; OR, 1.43; hysterectomy 2.5% vs. 1.6%; OR, 1.65).
“The odds of intra-abdominal hematoma, postoperative pain, and a length of stay of 3 or more days were also significantly higher when an adhesion barrier was applied,” Dr. Closon said.
He reported having no financial disclosures.
LAS VEGAS – Adhesion barriers may cause more problems than they prevent when employed in myomectomy and hysterectomy.
Ileus, fever, hematomas, and even small bowel obstruction occurred significantly more often when the barriers were used in conjunction with those procedures – especially when performed as open surgeries, Dr. François Closon reported at the AAGL Global Congress. And although their use remains relatively small among these patients, it’s been steadily rising since 2003, he said.
“Adhesion barriers are also being used about eight times more often in myomectomies than in hysterectomies, and this seems to be going up every year,” Dr. Closon of McGill University, Montreal, said at the meeting sponsored by AAGL.
His retrospective study drew on the U.S. Healthcare Cost and Utilization Project Nationwide Inpatient Sample, and examined the perioperative outcomes of adhesion barriers among nearly 475,000 women who underwent myomectomy or hysterectomy during 2003-2011. Of these, 9,000 (about 2%) had an adhesion barrier applied during the surgery. There were no significant baseline differences between the groups, except for age. Women in the myomectomy group were younger than those in the hysterectomy group (mean 36 years vs. 46 years). Age, however, was not associated with the use of an adhesion barrier.
Dr. Closon found a significant linear increase in the use of barriers in both procedures over the study period. In hysterectomy, barrier use rose slightly, but significantly, from 0.37% in 2003 to 2% in 2011. The increase was much greater for use in myomectomy, which rose from 3% to 7% during the same period.
Ileus was significantly more common among myomectomies that used the barrier (4% vs. 3%; OR, 1.49). The difference was more pronounced with hysterectomies (5% vs. 2.5%; OR, 1.97).
Small bowel obstruction was rare, but significantly more common in hysterectomies that employed adhesion barriers (0.2% vs. 0.4%; OR, 1.89).
Fever was also significantly more common in both procedures when the barrier was used (myomectomy 4.4% vs. 3%; OR, 1.43; hysterectomy 2.5% vs. 1.6%; OR, 1.65).
“The odds of intra-abdominal hematoma, postoperative pain, and a length of stay of 3 or more days were also significantly higher when an adhesion barrier was applied,” Dr. Closon said.
He reported having no financial disclosures.
AT THE AAGL GLOBAL CONGRESS
Key clinical point: Adhesion barriers are associated with perioperative complications in myomectomy and hysterectomy.
Major finding: Ileus was significantly more common in hysterectomies that used an adhesion barrier (5% vs. 2.5%; odds ratio, 1.97).
Data source: The retrospective database study comprised 9,000 women.
Disclosures: Dr. Closon reported having no financial disclosures.
CTAD: New aducanumab subanalysis bolsters phase III trials in very mild Alzheimer’s
BARCELONA – A new look at aducanumab’s phase I study data has reinforced the developer’s confidence that its newly launched phase III trials are targeting the right group: patients with very early Alzheimer’s disease.
A subgroup analysis of patients in the phase IPRIME study with the mildest disease showed that the antibody effectively cleared amyloid plaques and that three of the four doses tested effected at least some cognitive improvement at 54 weeks, Dr. Vissia Viglietta said at the Clinical Trials on Alzheimer’s Disease conference.
The parsed cognitive data look slightly better in this very mild group than they did in the overall analysis, especially those associated with the 6-mg/kg dose, which failed to confer significant benefit in either of the study’s two cognitive measures, the Mini–Mental State Exam (MMSE) and Clinical Dementia Rating scale-sum of boxes (CDR-SB). In the subgroup analysis, however, the 6-mg/kg dose significantly slowed decline as measured by the CDR-SB. It still had no significant effect on the MMSE, compared with placebo.
The subanalysis should be viewed with the same caution as the overall PRIME data, said Dr. Viglietta, director of medical research at Biogen, which is developing the drug. The phase I study was designed to prove safety, not efficacy. The numbers in each dosing group were small, and the numbers in the subanalysis were even smaller.
“The confidence intervals were also quite large, and it was obviously not powered to detect clinical endpoints,” which were investigated in an exploratory analysis that was not prespecified, Dr. Viglietta said. But the numbers provide some assurance that the phase III trials – dubbed EMERGE and ENGAGE – are targeting the right population. “We hope to be able to confirm both the safety and efficacy we saw in PRIME’s early Alzheimer’s population.”
The subanalysis comprised 92 of the original 166-patient cohort. They had a mean age of 72 years. About 70% had prodromal disease, and the rest had mild disease. Importantly, all patients in the PRIME study had confirmed amyloid pathology as measured by PET amyloid imaging, making it the first antiamyloid drug trial comprising only amyloid-positive patients. The phase III studies will also employ baseline PET amyloid imaging in patient selection.
By 26 weeks, aducanumab at the 3-, 6-, and 10-mg/kg doses was associated with significant clearance of amyloid plaque; the effect was sustained at 54 weeks. The 1-mg/kg dose had no significant effect on plaque. Amyloid was removed throughout the entire brain, with no one region driving the change, Dr. Viglietta noted.
All doses except 1 mg/kg significantly slowed decline on the CDR-SB. On the MMSE, the 3-mg/kg and 10-mg/kg doses conferred significant benefit. The 1-mg/kg and 6-mg/kg doses did not.
As in the overall PRIME analysis, amyloid-related imaging abnormalities (ARIA) were the most common adverse event. ARIA can be classified as hemorrhagic (ARIA-H) or edematous (ARIA-E); all of the 15 cases were ARIA-E. None of these were symptomatic; all were revealed during routine imaging studies. The incidence was dose dependent: There were no cases in the 1-mg/kg group, but it occurred in 11% of the 3-mg/kg group, 29% of the 6-mg/kg group, and 44% of the 10-mg/kg group.
ARIA-E was also more prevalent and seemed to be more serious among patients who carried the apolipoprotein E epsilon-4 (APOE4) allele. Twelve of the 15 cases occurred in APOE4 carriers. Eight patients with ARIA-E discontinued treatment (seven in the 10-mg/kg group and one in the 6-mg/kg group). Of these, six were APOE4 positive. The rest of the patients continued treatment either at the same or a reduced dose.
“ARIA-E was monitorable and manageable, and the majority of patients did not have a recurrence when they continued their treatment,” Dr. Viglietta said.
The two identical phase III trials, EMERGE and ENGAGE, are beginning to get underway. In September, Biogen announced that the first patient had been recruited. In all, the studies aim to enroll 2,700 patients with early Alzheimer’s. They will be conducted in more than 20 countries, according to the company website.
In addition to a positive PET amyloid imaging study for either trial, participants must have a CDR global score of 0.5, an MMSE score of 24-30; and a Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) score of 85 or lower.
Each study will randomize patients to placebo or two active doses, depending on APOE4 status. APOE4 carriers will be randomized to either 3- or 6-mg/kg doses and noncarriers to either 6- or 10-mg/kg doses. The 3.5-year studies will have an active randomization phase of 18 months, followed by a 24-month extension study.
The primary endpoint is change from baseline on the CDR-SB. Secondary endpoints are change on the MMSE, the Alzheimer’s Disease Assessment Study cognitive scale (ADAS-cog), and the Alzheimer’s Disease Cooperative Study activities of daily living and mild cognitive impairment scales (ADCS ADL, MCI). A subset of patients will undergo additional PET amyloid imaging as a tertiary endpoint.
Biogen is actively seeking patients for these studies. A recruitment website walks patients through some questions to identify those who might initially qualify.
BARCELONA – A new look at aducanumab’s phase I study data has reinforced the developer’s confidence that its newly launched phase III trials are targeting the right group: patients with very early Alzheimer’s disease.
A subgroup analysis of patients in the phase IPRIME study with the mildest disease showed that the antibody effectively cleared amyloid plaques and that three of the four doses tested effected at least some cognitive improvement at 54 weeks, Dr. Vissia Viglietta said at the Clinical Trials on Alzheimer’s Disease conference.
The parsed cognitive data look slightly better in this very mild group than they did in the overall analysis, especially those associated with the 6-mg/kg dose, which failed to confer significant benefit in either of the study’s two cognitive measures, the Mini–Mental State Exam (MMSE) and Clinical Dementia Rating scale-sum of boxes (CDR-SB). In the subgroup analysis, however, the 6-mg/kg dose significantly slowed decline as measured by the CDR-SB. It still had no significant effect on the MMSE, compared with placebo.
The subanalysis should be viewed with the same caution as the overall PRIME data, said Dr. Viglietta, director of medical research at Biogen, which is developing the drug. The phase I study was designed to prove safety, not efficacy. The numbers in each dosing group were small, and the numbers in the subanalysis were even smaller.
“The confidence intervals were also quite large, and it was obviously not powered to detect clinical endpoints,” which were investigated in an exploratory analysis that was not prespecified, Dr. Viglietta said. But the numbers provide some assurance that the phase III trials – dubbed EMERGE and ENGAGE – are targeting the right population. “We hope to be able to confirm both the safety and efficacy we saw in PRIME’s early Alzheimer’s population.”
The subanalysis comprised 92 of the original 166-patient cohort. They had a mean age of 72 years. About 70% had prodromal disease, and the rest had mild disease. Importantly, all patients in the PRIME study had confirmed amyloid pathology as measured by PET amyloid imaging, making it the first antiamyloid drug trial comprising only amyloid-positive patients. The phase III studies will also employ baseline PET amyloid imaging in patient selection.
By 26 weeks, aducanumab at the 3-, 6-, and 10-mg/kg doses was associated with significant clearance of amyloid plaque; the effect was sustained at 54 weeks. The 1-mg/kg dose had no significant effect on plaque. Amyloid was removed throughout the entire brain, with no one region driving the change, Dr. Viglietta noted.
All doses except 1 mg/kg significantly slowed decline on the CDR-SB. On the MMSE, the 3-mg/kg and 10-mg/kg doses conferred significant benefit. The 1-mg/kg and 6-mg/kg doses did not.
As in the overall PRIME analysis, amyloid-related imaging abnormalities (ARIA) were the most common adverse event. ARIA can be classified as hemorrhagic (ARIA-H) or edematous (ARIA-E); all of the 15 cases were ARIA-E. None of these were symptomatic; all were revealed during routine imaging studies. The incidence was dose dependent: There were no cases in the 1-mg/kg group, but it occurred in 11% of the 3-mg/kg group, 29% of the 6-mg/kg group, and 44% of the 10-mg/kg group.
ARIA-E was also more prevalent and seemed to be more serious among patients who carried the apolipoprotein E epsilon-4 (APOE4) allele. Twelve of the 15 cases occurred in APOE4 carriers. Eight patients with ARIA-E discontinued treatment (seven in the 10-mg/kg group and one in the 6-mg/kg group). Of these, six were APOE4 positive. The rest of the patients continued treatment either at the same or a reduced dose.
“ARIA-E was monitorable and manageable, and the majority of patients did not have a recurrence when they continued their treatment,” Dr. Viglietta said.
The two identical phase III trials, EMERGE and ENGAGE, are beginning to get underway. In September, Biogen announced that the first patient had been recruited. In all, the studies aim to enroll 2,700 patients with early Alzheimer’s. They will be conducted in more than 20 countries, according to the company website.
In addition to a positive PET amyloid imaging study for either trial, participants must have a CDR global score of 0.5, an MMSE score of 24-30; and a Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) score of 85 or lower.
Each study will randomize patients to placebo or two active doses, depending on APOE4 status. APOE4 carriers will be randomized to either 3- or 6-mg/kg doses and noncarriers to either 6- or 10-mg/kg doses. The 3.5-year studies will have an active randomization phase of 18 months, followed by a 24-month extension study.
The primary endpoint is change from baseline on the CDR-SB. Secondary endpoints are change on the MMSE, the Alzheimer’s Disease Assessment Study cognitive scale (ADAS-cog), and the Alzheimer’s Disease Cooperative Study activities of daily living and mild cognitive impairment scales (ADCS ADL, MCI). A subset of patients will undergo additional PET amyloid imaging as a tertiary endpoint.
Biogen is actively seeking patients for these studies. A recruitment website walks patients through some questions to identify those who might initially qualify.
BARCELONA – A new look at aducanumab’s phase I study data has reinforced the developer’s confidence that its newly launched phase III trials are targeting the right group: patients with very early Alzheimer’s disease.
A subgroup analysis of patients in the phase IPRIME study with the mildest disease showed that the antibody effectively cleared amyloid plaques and that three of the four doses tested effected at least some cognitive improvement at 54 weeks, Dr. Vissia Viglietta said at the Clinical Trials on Alzheimer’s Disease conference.
The parsed cognitive data look slightly better in this very mild group than they did in the overall analysis, especially those associated with the 6-mg/kg dose, which failed to confer significant benefit in either of the study’s two cognitive measures, the Mini–Mental State Exam (MMSE) and Clinical Dementia Rating scale-sum of boxes (CDR-SB). In the subgroup analysis, however, the 6-mg/kg dose significantly slowed decline as measured by the CDR-SB. It still had no significant effect on the MMSE, compared with placebo.
The subanalysis should be viewed with the same caution as the overall PRIME data, said Dr. Viglietta, director of medical research at Biogen, which is developing the drug. The phase I study was designed to prove safety, not efficacy. The numbers in each dosing group were small, and the numbers in the subanalysis were even smaller.
“The confidence intervals were also quite large, and it was obviously not powered to detect clinical endpoints,” which were investigated in an exploratory analysis that was not prespecified, Dr. Viglietta said. But the numbers provide some assurance that the phase III trials – dubbed EMERGE and ENGAGE – are targeting the right population. “We hope to be able to confirm both the safety and efficacy we saw in PRIME’s early Alzheimer’s population.”
The subanalysis comprised 92 of the original 166-patient cohort. They had a mean age of 72 years. About 70% had prodromal disease, and the rest had mild disease. Importantly, all patients in the PRIME study had confirmed amyloid pathology as measured by PET amyloid imaging, making it the first antiamyloid drug trial comprising only amyloid-positive patients. The phase III studies will also employ baseline PET amyloid imaging in patient selection.
By 26 weeks, aducanumab at the 3-, 6-, and 10-mg/kg doses was associated with significant clearance of amyloid plaque; the effect was sustained at 54 weeks. The 1-mg/kg dose had no significant effect on plaque. Amyloid was removed throughout the entire brain, with no one region driving the change, Dr. Viglietta noted.
All doses except 1 mg/kg significantly slowed decline on the CDR-SB. On the MMSE, the 3-mg/kg and 10-mg/kg doses conferred significant benefit. The 1-mg/kg and 6-mg/kg doses did not.
As in the overall PRIME analysis, amyloid-related imaging abnormalities (ARIA) were the most common adverse event. ARIA can be classified as hemorrhagic (ARIA-H) or edematous (ARIA-E); all of the 15 cases were ARIA-E. None of these were symptomatic; all were revealed during routine imaging studies. The incidence was dose dependent: There were no cases in the 1-mg/kg group, but it occurred in 11% of the 3-mg/kg group, 29% of the 6-mg/kg group, and 44% of the 10-mg/kg group.
ARIA-E was also more prevalent and seemed to be more serious among patients who carried the apolipoprotein E epsilon-4 (APOE4) allele. Twelve of the 15 cases occurred in APOE4 carriers. Eight patients with ARIA-E discontinued treatment (seven in the 10-mg/kg group and one in the 6-mg/kg group). Of these, six were APOE4 positive. The rest of the patients continued treatment either at the same or a reduced dose.
“ARIA-E was monitorable and manageable, and the majority of patients did not have a recurrence when they continued their treatment,” Dr. Viglietta said.
The two identical phase III trials, EMERGE and ENGAGE, are beginning to get underway. In September, Biogen announced that the first patient had been recruited. In all, the studies aim to enroll 2,700 patients with early Alzheimer’s. They will be conducted in more than 20 countries, according to the company website.
In addition to a positive PET amyloid imaging study for either trial, participants must have a CDR global score of 0.5, an MMSE score of 24-30; and a Repeatable Battery for the Assessment of Neuropsychological Status (RBANS) score of 85 or lower.
Each study will randomize patients to placebo or two active doses, depending on APOE4 status. APOE4 carriers will be randomized to either 3- or 6-mg/kg doses and noncarriers to either 6- or 10-mg/kg doses. The 3.5-year studies will have an active randomization phase of 18 months, followed by a 24-month extension study.
The primary endpoint is change from baseline on the CDR-SB. Secondary endpoints are change on the MMSE, the Alzheimer’s Disease Assessment Study cognitive scale (ADAS-cog), and the Alzheimer’s Disease Cooperative Study activities of daily living and mild cognitive impairment scales (ADCS ADL, MCI). A subset of patients will undergo additional PET amyloid imaging as a tertiary endpoint.
Biogen is actively seeking patients for these studies. A recruitment website walks patients through some questions to identify those who might initially qualify.
AT CTAD
Key clinical point: A subanalysis of patients with very mild Alzheimer’s who received aducanumab in the PRIME trial provides some assurance that phase III trials of the drug that are currently enrolling patients are targeting the right population.
Major finding: All doses except 1 mg/kg significantly slowed decline on the CDR-SB at 54 weeks. On the MMSE, the 3-mg/kg and 10-mg/kg doses conferred significant benefit. The 1-mg/kg and 6-mg/kg doses did not.
Data source: A subanalysis of 92 patients in the 166-patient PRIME trial.
Disclosures: The presenter is an employee of Biogen, which sponsored the PRIME trial and is sponsoring the EMERGE and ENGAGE trials.
CTAD: Sigma-1 receptor agonist passes muster in small Alzheimer’s trial
BARCELONA – A novel small molecule that may help clear misfolded proteins imparted some significant cognitive benefits to Alzheimer’s patients after 5 weeks, even with a mid-study washout period in which patients stopped taking the drug, according to the results of a small, open-label, uncontrolled, phase IIa crossover trial.
The investigational drug, called Anavex 2-73, also was associated with significant improvements in the P300 electroencephalogram wave, a measure of nerve conduction associated with cognitive processing, Dr. Stephen Macfarlane said at the Clinical Trials on Alzheimer’s disease conference.
The study was small – just 32 patients with mild to moderate Alzheimer’s disease – and not powered for cognitive endpoints, said Dr. Macfarlane, director of Aged Psychiatry for Alfred Health, Victoria, Australia. Nevertheless, its clinical findings add an intriguing cachet to the study’s successful pharmacokinetic data, he said.
The results were encouraging enough for the manufacturer, Anavex Life Sciences to lengthen its preplanned 26-week extension trial to 52 weeks and move ahead with phase III planning, Dr. Macfarlane said.
Anavex 2-73 is an agonist of the sigma-1 receptor but also activates the muscarinic receptor. The sigma-1 receptoris found on neurons and glia in many areas of the central nervous system. It modulates a number of processes implicated in neurodegenerative diseases, including glutamate and calcium activity, reaction to oxidative stress, and mitochondrial function. There is also some evidence that sigma-1 receptor activation can induce neuronal regrowth and functional recovery after stroke. The sigma-1 receptor also appears to play a role in helping cells clear misfolded proteins – a pathway that makes it an attractive drug target in Alzheimer’s disease, as well as other neurodegenerative diseases with aberrant proteins, such as Parkinson’s and Huntington’s diseases.
In preclinical testing, Anavex 2-73 showed an additional cognitive property, Dr. Macfarlane said. “It seems to have a cognitive-enhancing effect, not only on transgenic mice, but on wild-type mice as well. When the [Alzheimer’s model] mice were fed it, they did significantly better on the water maze test. But when wild-type mice consumed it, they did better as well. This does suggest that it can enhance cognition.”
The study that Dr. Macfarlane presented at the conference randomized patients to either oral or intravenous treatment for 12 days, followed by a 12-day washout period, then crossed to the opposite administration for 12 days. At the end of 5 weeks, all patients were invited to continue with oral treatment out to 52 weeks. All of the 30 patients who finished the 5-week study elected to participate.
Patients were a mean of 71 years old. The median Mini-Mental State Examination (MMSE) score was 20.5. Most (78%) were taking a stable dose of acetylcholinesterase inhibitor.
The primary endpoints were safety, tolerability, and pharmacokinetics. The exploratory cognitive measures included electroencephalogram/event-related potential, MMSE, and the Cogstate battery. Function was assessed with the Alzheimer’s Disease Cooperative Study activities of daily living scale (ADCS-ADL) in patients who had completed at least 12 weeks of the follow-up study.
There were no safety concerns, Dr. Macfarlane said. While 94% of the subjects did have an adverse event, only one was considered serious. This was a case of delirium that developed in a patient who had several risk factors for the disorder, including a prior episode; it was not considered related to the study drug.
Dizziness was the most common adverse event (20 incidents in 15 patients), followed by headache (16 in 10 patients). Most (94%) occurred in the first week of treatment. All incidents were considered mild or moderate.
At the end of 5 weeks, the drug was associated with a median MMSE score increase of 1.5 points. While this change was not statistically significant, improvements on five of the six Cogstate battery measures were significant, with the biggest boost seen in the one-back test of working memory.
Dr. Macfarlane compared the magnitude of improvement to that which has been seen in a large Australian prospective cohort study, AIBL (Australian Imaging, Biomarkers & Lifestyle Flagship Study of Ageing). The Anavex 2-73 cohort made significant gains relative to the AIBL cohort taking acetylcholinesterase inhibitors at the same time period.
“Our group improved by 1.5 standard deviations across 5 weeks [compared with AIBL subjects], which included that 12-day period with no exposure to the study drug,” Dr. Macfarlane said. “This was quite an unexpected finding but potentially an important one.”
Anavex 2-73 was also associated with significant changes on EEG measures of event-related potential, particularly the P300 wave, which measures decision-making speed. The drug closed 80% of the gap between patients and historical controls in P300 amplitude. It also closed 85% of the gap in task accuracy and 66% of that in reaction time. Finally, patients taking Anavex 2-73 exceeded control performance on false-alarm reactions by 4%.
Patients who have completed 12 weeks of treatment in the 52-week extension trial experienced a mean improvement of 3.2 points on the ADCS-ADL measure. The difference, while not statistically significant, is encouraging at this point in treatment, Dr. Macfarlane said.
Dr. Macfarlane has no financial interest in Anavex 2-73, and has no financial disclosures.
BARCELONA – A novel small molecule that may help clear misfolded proteins imparted some significant cognitive benefits to Alzheimer’s patients after 5 weeks, even with a mid-study washout period in which patients stopped taking the drug, according to the results of a small, open-label, uncontrolled, phase IIa crossover trial.
The investigational drug, called Anavex 2-73, also was associated with significant improvements in the P300 electroencephalogram wave, a measure of nerve conduction associated with cognitive processing, Dr. Stephen Macfarlane said at the Clinical Trials on Alzheimer’s disease conference.
The study was small – just 32 patients with mild to moderate Alzheimer’s disease – and not powered for cognitive endpoints, said Dr. Macfarlane, director of Aged Psychiatry for Alfred Health, Victoria, Australia. Nevertheless, its clinical findings add an intriguing cachet to the study’s successful pharmacokinetic data, he said.
The results were encouraging enough for the manufacturer, Anavex Life Sciences to lengthen its preplanned 26-week extension trial to 52 weeks and move ahead with phase III planning, Dr. Macfarlane said.
Anavex 2-73 is an agonist of the sigma-1 receptor but also activates the muscarinic receptor. The sigma-1 receptoris found on neurons and glia in many areas of the central nervous system. It modulates a number of processes implicated in neurodegenerative diseases, including glutamate and calcium activity, reaction to oxidative stress, and mitochondrial function. There is also some evidence that sigma-1 receptor activation can induce neuronal regrowth and functional recovery after stroke. The sigma-1 receptor also appears to play a role in helping cells clear misfolded proteins – a pathway that makes it an attractive drug target in Alzheimer’s disease, as well as other neurodegenerative diseases with aberrant proteins, such as Parkinson’s and Huntington’s diseases.
In preclinical testing, Anavex 2-73 showed an additional cognitive property, Dr. Macfarlane said. “It seems to have a cognitive-enhancing effect, not only on transgenic mice, but on wild-type mice as well. When the [Alzheimer’s model] mice were fed it, they did significantly better on the water maze test. But when wild-type mice consumed it, they did better as well. This does suggest that it can enhance cognition.”
The study that Dr. Macfarlane presented at the conference randomized patients to either oral or intravenous treatment for 12 days, followed by a 12-day washout period, then crossed to the opposite administration for 12 days. At the end of 5 weeks, all patients were invited to continue with oral treatment out to 52 weeks. All of the 30 patients who finished the 5-week study elected to participate.
Patients were a mean of 71 years old. The median Mini-Mental State Examination (MMSE) score was 20.5. Most (78%) were taking a stable dose of acetylcholinesterase inhibitor.
The primary endpoints were safety, tolerability, and pharmacokinetics. The exploratory cognitive measures included electroencephalogram/event-related potential, MMSE, and the Cogstate battery. Function was assessed with the Alzheimer’s Disease Cooperative Study activities of daily living scale (ADCS-ADL) in patients who had completed at least 12 weeks of the follow-up study.
There were no safety concerns, Dr. Macfarlane said. While 94% of the subjects did have an adverse event, only one was considered serious. This was a case of delirium that developed in a patient who had several risk factors for the disorder, including a prior episode; it was not considered related to the study drug.
Dizziness was the most common adverse event (20 incidents in 15 patients), followed by headache (16 in 10 patients). Most (94%) occurred in the first week of treatment. All incidents were considered mild or moderate.
At the end of 5 weeks, the drug was associated with a median MMSE score increase of 1.5 points. While this change was not statistically significant, improvements on five of the six Cogstate battery measures were significant, with the biggest boost seen in the one-back test of working memory.
Dr. Macfarlane compared the magnitude of improvement to that which has been seen in a large Australian prospective cohort study, AIBL (Australian Imaging, Biomarkers & Lifestyle Flagship Study of Ageing). The Anavex 2-73 cohort made significant gains relative to the AIBL cohort taking acetylcholinesterase inhibitors at the same time period.
“Our group improved by 1.5 standard deviations across 5 weeks [compared with AIBL subjects], which included that 12-day period with no exposure to the study drug,” Dr. Macfarlane said. “This was quite an unexpected finding but potentially an important one.”
Anavex 2-73 was also associated with significant changes on EEG measures of event-related potential, particularly the P300 wave, which measures decision-making speed. The drug closed 80% of the gap between patients and historical controls in P300 amplitude. It also closed 85% of the gap in task accuracy and 66% of that in reaction time. Finally, patients taking Anavex 2-73 exceeded control performance on false-alarm reactions by 4%.
Patients who have completed 12 weeks of treatment in the 52-week extension trial experienced a mean improvement of 3.2 points on the ADCS-ADL measure. The difference, while not statistically significant, is encouraging at this point in treatment, Dr. Macfarlane said.
Dr. Macfarlane has no financial interest in Anavex 2-73, and has no financial disclosures.
BARCELONA – A novel small molecule that may help clear misfolded proteins imparted some significant cognitive benefits to Alzheimer’s patients after 5 weeks, even with a mid-study washout period in which patients stopped taking the drug, according to the results of a small, open-label, uncontrolled, phase IIa crossover trial.
The investigational drug, called Anavex 2-73, also was associated with significant improvements in the P300 electroencephalogram wave, a measure of nerve conduction associated with cognitive processing, Dr. Stephen Macfarlane said at the Clinical Trials on Alzheimer’s disease conference.
The study was small – just 32 patients with mild to moderate Alzheimer’s disease – and not powered for cognitive endpoints, said Dr. Macfarlane, director of Aged Psychiatry for Alfred Health, Victoria, Australia. Nevertheless, its clinical findings add an intriguing cachet to the study’s successful pharmacokinetic data, he said.
The results were encouraging enough for the manufacturer, Anavex Life Sciences to lengthen its preplanned 26-week extension trial to 52 weeks and move ahead with phase III planning, Dr. Macfarlane said.
Anavex 2-73 is an agonist of the sigma-1 receptor but also activates the muscarinic receptor. The sigma-1 receptoris found on neurons and glia in many areas of the central nervous system. It modulates a number of processes implicated in neurodegenerative diseases, including glutamate and calcium activity, reaction to oxidative stress, and mitochondrial function. There is also some evidence that sigma-1 receptor activation can induce neuronal regrowth and functional recovery after stroke. The sigma-1 receptor also appears to play a role in helping cells clear misfolded proteins – a pathway that makes it an attractive drug target in Alzheimer’s disease, as well as other neurodegenerative diseases with aberrant proteins, such as Parkinson’s and Huntington’s diseases.
In preclinical testing, Anavex 2-73 showed an additional cognitive property, Dr. Macfarlane said. “It seems to have a cognitive-enhancing effect, not only on transgenic mice, but on wild-type mice as well. When the [Alzheimer’s model] mice were fed it, they did significantly better on the water maze test. But when wild-type mice consumed it, they did better as well. This does suggest that it can enhance cognition.”
The study that Dr. Macfarlane presented at the conference randomized patients to either oral or intravenous treatment for 12 days, followed by a 12-day washout period, then crossed to the opposite administration for 12 days. At the end of 5 weeks, all patients were invited to continue with oral treatment out to 52 weeks. All of the 30 patients who finished the 5-week study elected to participate.
Patients were a mean of 71 years old. The median Mini-Mental State Examination (MMSE) score was 20.5. Most (78%) were taking a stable dose of acetylcholinesterase inhibitor.
The primary endpoints were safety, tolerability, and pharmacokinetics. The exploratory cognitive measures included electroencephalogram/event-related potential, MMSE, and the Cogstate battery. Function was assessed with the Alzheimer’s Disease Cooperative Study activities of daily living scale (ADCS-ADL) in patients who had completed at least 12 weeks of the follow-up study.
There were no safety concerns, Dr. Macfarlane said. While 94% of the subjects did have an adverse event, only one was considered serious. This was a case of delirium that developed in a patient who had several risk factors for the disorder, including a prior episode; it was not considered related to the study drug.
Dizziness was the most common adverse event (20 incidents in 15 patients), followed by headache (16 in 10 patients). Most (94%) occurred in the first week of treatment. All incidents were considered mild or moderate.
At the end of 5 weeks, the drug was associated with a median MMSE score increase of 1.5 points. While this change was not statistically significant, improvements on five of the six Cogstate battery measures were significant, with the biggest boost seen in the one-back test of working memory.
Dr. Macfarlane compared the magnitude of improvement to that which has been seen in a large Australian prospective cohort study, AIBL (Australian Imaging, Biomarkers & Lifestyle Flagship Study of Ageing). The Anavex 2-73 cohort made significant gains relative to the AIBL cohort taking acetylcholinesterase inhibitors at the same time period.
“Our group improved by 1.5 standard deviations across 5 weeks [compared with AIBL subjects], which included that 12-day period with no exposure to the study drug,” Dr. Macfarlane said. “This was quite an unexpected finding but potentially an important one.”
Anavex 2-73 was also associated with significant changes on EEG measures of event-related potential, particularly the P300 wave, which measures decision-making speed. The drug closed 80% of the gap between patients and historical controls in P300 amplitude. It also closed 85% of the gap in task accuracy and 66% of that in reaction time. Finally, patients taking Anavex 2-73 exceeded control performance on false-alarm reactions by 4%.
Patients who have completed 12 weeks of treatment in the 52-week extension trial experienced a mean improvement of 3.2 points on the ADCS-ADL measure. The difference, while not statistically significant, is encouraging at this point in treatment, Dr. Macfarlane said.
Dr. Macfarlane has no financial interest in Anavex 2-73, and has no financial disclosures.
AT CTAD
Key clinical point: A novel agonist of the sigma-1 receptor improved cognition in an early study of patients with mild to moderate Alzheimer’s disease.
Major finding: Anavex 2-73 was associated with a 1.5-point increase on the MMSE and significant improvements in five of six domains on the Cogstate battery.
Data source: An open-label, uncontrolled, phase IIa crossover trial of 32 patients with mild to moderate Alzheimer’s disease.
Disclosures: Dr. Macfarlane has no financial interest in Anavex 2-73 and has no financial disclosures.