User login
Lipids may improve with treatment of juvenile idiopathic arthritis
Successful antirheumatic treatment improved the abnormal lipid levels associated with juvenile idiopathic arthritis in a small, prospective study of 58 children with the disease.
The children’s cholesterol levels and ratios – as well as C-reactive protein (CRP) levels – improved significantly over an 18-month period of treatment that controlled the disease.
"These results strongly suggest that lipid profiles are associated with disease activity and that effective antirheumatic therapy can reverse, at least partially, the adverse lipid profiles in patients with juvenile idiopathic arthritis," Dr. Chang-Ching Shen and colleagues wrote (Semin. Arthritis Rheum. 2013 Jan. 3 [doi:10.1016/j.semarthrit.2012.10.002]).
This is the first longitudinal study to report the association of disease activity with serum lipid profiles in juvenile idiopathic arthritis (JIA) patients, said Dr. Shen of the Chang Gung Memorial Hospital and Chang Gung University College of Medicine, Taoyuan, Taiwan, and coauthors.
After 18 months of treatment with antirheumatic drugs, the investigators compared lipid profiles in 58 children treated for newly diagnosed JIA and stratified them into inactive (31) and active (27) disease status.
The patients’ mean age was 13 years, and the mean disease duration was about 5 months. More than 90% were taking NSAIDs. Methotrexate was the most common antirheumatic drug given (58% of inactive patients, 67% of active patients), followed by azathioprine (10% and 19%, respectively), sulfasalazine (16% and 11%), etanercept (13% and 11%), and prednisolone (52% and 63%).
Over the 18-month study, there were significant changes in lipid levels and atherogenic measures in the inactive disease group but not in the active disease group. High-density lipoprotein (HDL) cholesterol level increased by 5.5 mg/dL in the inactive group but decreased by 1.15 mg/dL in the active group. Total cholesterol increased 7.5 mg/dL in the inactive group and decreased 5.5 mg/dL in the active group. These changes correlated with changes in CRP, which decreased 14 mg/L in the inactive group and increased about 1 mg/L in the active group.
The investigators then compared the JIA patients with a matched set of 232 controls from the Nutrition and Health Survey in Taiwan, a national population-based study evaluating cardiovascular risk factors, including dyslipidemia, in Taiwanese people aged 4 years and older. The active disease group had significantly lower levels of HDL and total cholesterol than did control group patients. Levels in the control and inactive disease groups were similar. There were no between-group differences in the LDL/HDL ratio or in the ratio of total cholesterol/HDL cholesterol.
"Without using any lipid-modifying drugs, the aberrant lipid profiles were improved by a control of inflammatory status, which may in turn reduce the risk of cardiovascular disease in JIA," the investigators noted. "The results can provide a useful clue for further immunopathogenesis studies."
The currently available version of the report, a corrected proof, did not note study sponsors or any financial disclosures for any of the authors.
Successful antirheumatic treatment improved the abnormal lipid levels associated with juvenile idiopathic arthritis in a small, prospective study of 58 children with the disease.
The children’s cholesterol levels and ratios – as well as C-reactive protein (CRP) levels – improved significantly over an 18-month period of treatment that controlled the disease.
"These results strongly suggest that lipid profiles are associated with disease activity and that effective antirheumatic therapy can reverse, at least partially, the adverse lipid profiles in patients with juvenile idiopathic arthritis," Dr. Chang-Ching Shen and colleagues wrote (Semin. Arthritis Rheum. 2013 Jan. 3 [doi:10.1016/j.semarthrit.2012.10.002]).
This is the first longitudinal study to report the association of disease activity with serum lipid profiles in juvenile idiopathic arthritis (JIA) patients, said Dr. Shen of the Chang Gung Memorial Hospital and Chang Gung University College of Medicine, Taoyuan, Taiwan, and coauthors.
After 18 months of treatment with antirheumatic drugs, the investigators compared lipid profiles in 58 children treated for newly diagnosed JIA and stratified them into inactive (31) and active (27) disease status.
The patients’ mean age was 13 years, and the mean disease duration was about 5 months. More than 90% were taking NSAIDs. Methotrexate was the most common antirheumatic drug given (58% of inactive patients, 67% of active patients), followed by azathioprine (10% and 19%, respectively), sulfasalazine (16% and 11%), etanercept (13% and 11%), and prednisolone (52% and 63%).
Over the 18-month study, there were significant changes in lipid levels and atherogenic measures in the inactive disease group but not in the active disease group. High-density lipoprotein (HDL) cholesterol level increased by 5.5 mg/dL in the inactive group but decreased by 1.15 mg/dL in the active group. Total cholesterol increased 7.5 mg/dL in the inactive group and decreased 5.5 mg/dL in the active group. These changes correlated with changes in CRP, which decreased 14 mg/L in the inactive group and increased about 1 mg/L in the active group.
The investigators then compared the JIA patients with a matched set of 232 controls from the Nutrition and Health Survey in Taiwan, a national population-based study evaluating cardiovascular risk factors, including dyslipidemia, in Taiwanese people aged 4 years and older. The active disease group had significantly lower levels of HDL and total cholesterol than did control group patients. Levels in the control and inactive disease groups were similar. There were no between-group differences in the LDL/HDL ratio or in the ratio of total cholesterol/HDL cholesterol.
"Without using any lipid-modifying drugs, the aberrant lipid profiles were improved by a control of inflammatory status, which may in turn reduce the risk of cardiovascular disease in JIA," the investigators noted. "The results can provide a useful clue for further immunopathogenesis studies."
The currently available version of the report, a corrected proof, did not note study sponsors or any financial disclosures for any of the authors.
Successful antirheumatic treatment improved the abnormal lipid levels associated with juvenile idiopathic arthritis in a small, prospective study of 58 children with the disease.
The children’s cholesterol levels and ratios – as well as C-reactive protein (CRP) levels – improved significantly over an 18-month period of treatment that controlled the disease.
"These results strongly suggest that lipid profiles are associated with disease activity and that effective antirheumatic therapy can reverse, at least partially, the adverse lipid profiles in patients with juvenile idiopathic arthritis," Dr. Chang-Ching Shen and colleagues wrote (Semin. Arthritis Rheum. 2013 Jan. 3 [doi:10.1016/j.semarthrit.2012.10.002]).
This is the first longitudinal study to report the association of disease activity with serum lipid profiles in juvenile idiopathic arthritis (JIA) patients, said Dr. Shen of the Chang Gung Memorial Hospital and Chang Gung University College of Medicine, Taoyuan, Taiwan, and coauthors.
After 18 months of treatment with antirheumatic drugs, the investigators compared lipid profiles in 58 children treated for newly diagnosed JIA and stratified them into inactive (31) and active (27) disease status.
The patients’ mean age was 13 years, and the mean disease duration was about 5 months. More than 90% were taking NSAIDs. Methotrexate was the most common antirheumatic drug given (58% of inactive patients, 67% of active patients), followed by azathioprine (10% and 19%, respectively), sulfasalazine (16% and 11%), etanercept (13% and 11%), and prednisolone (52% and 63%).
Over the 18-month study, there were significant changes in lipid levels and atherogenic measures in the inactive disease group but not in the active disease group. High-density lipoprotein (HDL) cholesterol level increased by 5.5 mg/dL in the inactive group but decreased by 1.15 mg/dL in the active group. Total cholesterol increased 7.5 mg/dL in the inactive group and decreased 5.5 mg/dL in the active group. These changes correlated with changes in CRP, which decreased 14 mg/L in the inactive group and increased about 1 mg/L in the active group.
The investigators then compared the JIA patients with a matched set of 232 controls from the Nutrition and Health Survey in Taiwan, a national population-based study evaluating cardiovascular risk factors, including dyslipidemia, in Taiwanese people aged 4 years and older. The active disease group had significantly lower levels of HDL and total cholesterol than did control group patients. Levels in the control and inactive disease groups were similar. There were no between-group differences in the LDL/HDL ratio or in the ratio of total cholesterol/HDL cholesterol.
"Without using any lipid-modifying drugs, the aberrant lipid profiles were improved by a control of inflammatory status, which may in turn reduce the risk of cardiovascular disease in JIA," the investigators noted. "The results can provide a useful clue for further immunopathogenesis studies."
The currently available version of the report, a corrected proof, did not note study sponsors or any financial disclosures for any of the authors.
FROM SEMINARS IN ARTHRITIS AND RHEUMATISM
Major Finding: Disease inactivity after 18 months of treatment with antirheumatic drugs was associated with an increase of 5.5 mg/dL in HDL cholesterol, an increase of 7.5 mg/dL in total cholesterol, and a decrease of 14 mg/L in CRP levels, compared with no significant changes in patients who continued to have active disease.
Data Source: A prospective study of 31 children with inactive juvenile idiopathic arthritis, 27 with active disease, and 232 matched controls.
Disclosures: The paper did not note the study sponsor or contain any financial disclosure statement.
Influenza deaths in kids still on the rise
Influenza in the United States continued to strike hard at children, with 9 additional pediatric deaths last week, bringing the total for this season to 29.
The week of Jan. 6-12 was also hard on adults aged 65 and older. For this group, the influenza-related hospitalization rate was 82/100,000 population – strikingly higher than the national rate of 19/100,000 population, Dr. Thomas Frieden, director of the Centers for Disease Control and Prevention, said in a press briefing.
"We are about halfway through this flu season, and it’s shaping up to be worse than average and particularly hard on the elderly" Dr. Frieden said. "Influenza always takes the heaviest toll on seniors, especially when H3N2 is the prevalent strain, as it is this year." Surveillance methods are available online.
Thirty states and New York City reported high levels of flu activity during the week ending on Jan. 12 – an increase from 24 states the week before. However, there is some small suggestion that the season, which started about a month earlier than usual, may be peaking, Dr. Frieden noted. Nationwide, the rate of influenza-like illness was 4.3% - a slight decreased from 4.8% last week. Rates are still rising in some western states, including California. A map of the outbreak is available online.
Dr. Frieden cautioned that the decreased infection rate won’t yet affect rates of hospitalizations, complications, and deaths. "We expect to see these in a phased response [to infection rates]. We expect to see the numbers of hospitalizations and deaths rise further in the next week or so, as people develop complications from the illness."
Some spot shortages of vaccine occurred last week. "We continue to hear reports of shortages. Some patients have not been able to find vaccine and some providers have not been able to order."
The five vaccine manufacturing companies initially projected a national supply of about 135 million doses, of which 129 million have already been distributed. The companies have agreed to make an additional 10 million doses available, much of which will be directed at the western states, where infections are still on the uptick. The total of 145 million doses is about double the amount of vaccine available several years ago.
"There is still vaccine out there," Dr. Frieden said." Some formulations might be temporarily out of stock, but providers who want to should be able to order more."
Providers can track vaccine availability at http://preventinfluenza.org/ivats/ivats_12_13.pdf. Patients can find vaccine near their home by entering their ZIP code on the www.preventinfluenza.org home page.
"Although it’s not perfect, vaccination is the best tool we have to protect from the flu," he said. "There is still time to vaccinate, especially in states where most of the season is still to come."
Although there’s still plenty of antiviral medication, the Food and Drug Administration is taking steps to avoid any problems. There are some spot shortages of oral suspension oseltamivir for children aged younger than 1 year. Pharmacists can compound a substitute using the 30-mg, 40-mg, and even 75-mg capsules, said FDA Commissioner Margaret Hamburg. Directions are included on the oseltamavir packaging insert.
The FDA has also approved the distribution of 2 million older – but still effective – 75-mg oseltamivir capsules. The medication is not expired, but is packaged in older material, with an insert that doesn’t include the compounding instructions, Ms. Hamburg said during the briefing.
Dr. Frieden reiterated the importance of early antiviral treatment.
"In studies of very sick patients, death rates were cut by 75% or more by prompt use. For high-risk patients it can mean the difference between mild diseases and the intensive care unit or death."
High-risk groups include the elderly, children, anyone with a compromised immune system, children with neurologic or developmental disorders, and those with asthma, diabetes, or heart disease
"Even if a rapid flu test is negative for a high-risk patient, they should be treated in the first 48 hours of developing symptoms, because the rapid flu test does give a number of false negatives," Dr Frieden said.
Older and high-risk adults should especially avoid being around children who may be infected. "This is not a good time to babysit the grandchildren," he said.
Influenza in the United States continued to strike hard at children, with 9 additional pediatric deaths last week, bringing the total for this season to 29.
The week of Jan. 6-12 was also hard on adults aged 65 and older. For this group, the influenza-related hospitalization rate was 82/100,000 population – strikingly higher than the national rate of 19/100,000 population, Dr. Thomas Frieden, director of the Centers for Disease Control and Prevention, said in a press briefing.
"We are about halfway through this flu season, and it’s shaping up to be worse than average and particularly hard on the elderly" Dr. Frieden said. "Influenza always takes the heaviest toll on seniors, especially when H3N2 is the prevalent strain, as it is this year." Surveillance methods are available online.
Thirty states and New York City reported high levels of flu activity during the week ending on Jan. 12 – an increase from 24 states the week before. However, there is some small suggestion that the season, which started about a month earlier than usual, may be peaking, Dr. Frieden noted. Nationwide, the rate of influenza-like illness was 4.3% - a slight decreased from 4.8% last week. Rates are still rising in some western states, including California. A map of the outbreak is available online.
Dr. Frieden cautioned that the decreased infection rate won’t yet affect rates of hospitalizations, complications, and deaths. "We expect to see these in a phased response [to infection rates]. We expect to see the numbers of hospitalizations and deaths rise further in the next week or so, as people develop complications from the illness."
Some spot shortages of vaccine occurred last week. "We continue to hear reports of shortages. Some patients have not been able to find vaccine and some providers have not been able to order."
The five vaccine manufacturing companies initially projected a national supply of about 135 million doses, of which 129 million have already been distributed. The companies have agreed to make an additional 10 million doses available, much of which will be directed at the western states, where infections are still on the uptick. The total of 145 million doses is about double the amount of vaccine available several years ago.
"There is still vaccine out there," Dr. Frieden said." Some formulations might be temporarily out of stock, but providers who want to should be able to order more."
Providers can track vaccine availability at http://preventinfluenza.org/ivats/ivats_12_13.pdf. Patients can find vaccine near their home by entering their ZIP code on the www.preventinfluenza.org home page.
"Although it’s not perfect, vaccination is the best tool we have to protect from the flu," he said. "There is still time to vaccinate, especially in states where most of the season is still to come."
Although there’s still plenty of antiviral medication, the Food and Drug Administration is taking steps to avoid any problems. There are some spot shortages of oral suspension oseltamivir for children aged younger than 1 year. Pharmacists can compound a substitute using the 30-mg, 40-mg, and even 75-mg capsules, said FDA Commissioner Margaret Hamburg. Directions are included on the oseltamavir packaging insert.
The FDA has also approved the distribution of 2 million older – but still effective – 75-mg oseltamivir capsules. The medication is not expired, but is packaged in older material, with an insert that doesn’t include the compounding instructions, Ms. Hamburg said during the briefing.
Dr. Frieden reiterated the importance of early antiviral treatment.
"In studies of very sick patients, death rates were cut by 75% or more by prompt use. For high-risk patients it can mean the difference between mild diseases and the intensive care unit or death."
High-risk groups include the elderly, children, anyone with a compromised immune system, children with neurologic or developmental disorders, and those with asthma, diabetes, or heart disease
"Even if a rapid flu test is negative for a high-risk patient, they should be treated in the first 48 hours of developing symptoms, because the rapid flu test does give a number of false negatives," Dr Frieden said.
Older and high-risk adults should especially avoid being around children who may be infected. "This is not a good time to babysit the grandchildren," he said.
Influenza in the United States continued to strike hard at children, with 9 additional pediatric deaths last week, bringing the total for this season to 29.
The week of Jan. 6-12 was also hard on adults aged 65 and older. For this group, the influenza-related hospitalization rate was 82/100,000 population – strikingly higher than the national rate of 19/100,000 population, Dr. Thomas Frieden, director of the Centers for Disease Control and Prevention, said in a press briefing.
"We are about halfway through this flu season, and it’s shaping up to be worse than average and particularly hard on the elderly" Dr. Frieden said. "Influenza always takes the heaviest toll on seniors, especially when H3N2 is the prevalent strain, as it is this year." Surveillance methods are available online.
Thirty states and New York City reported high levels of flu activity during the week ending on Jan. 12 – an increase from 24 states the week before. However, there is some small suggestion that the season, which started about a month earlier than usual, may be peaking, Dr. Frieden noted. Nationwide, the rate of influenza-like illness was 4.3% - a slight decreased from 4.8% last week. Rates are still rising in some western states, including California. A map of the outbreak is available online.
Dr. Frieden cautioned that the decreased infection rate won’t yet affect rates of hospitalizations, complications, and deaths. "We expect to see these in a phased response [to infection rates]. We expect to see the numbers of hospitalizations and deaths rise further in the next week or so, as people develop complications from the illness."
Some spot shortages of vaccine occurred last week. "We continue to hear reports of shortages. Some patients have not been able to find vaccine and some providers have not been able to order."
The five vaccine manufacturing companies initially projected a national supply of about 135 million doses, of which 129 million have already been distributed. The companies have agreed to make an additional 10 million doses available, much of which will be directed at the western states, where infections are still on the uptick. The total of 145 million doses is about double the amount of vaccine available several years ago.
"There is still vaccine out there," Dr. Frieden said." Some formulations might be temporarily out of stock, but providers who want to should be able to order more."
Providers can track vaccine availability at http://preventinfluenza.org/ivats/ivats_12_13.pdf. Patients can find vaccine near their home by entering their ZIP code on the www.preventinfluenza.org home page.
"Although it’s not perfect, vaccination is the best tool we have to protect from the flu," he said. "There is still time to vaccinate, especially in states where most of the season is still to come."
Although there’s still plenty of antiviral medication, the Food and Drug Administration is taking steps to avoid any problems. There are some spot shortages of oral suspension oseltamivir for children aged younger than 1 year. Pharmacists can compound a substitute using the 30-mg, 40-mg, and even 75-mg capsules, said FDA Commissioner Margaret Hamburg. Directions are included on the oseltamavir packaging insert.
The FDA has also approved the distribution of 2 million older – but still effective – 75-mg oseltamivir capsules. The medication is not expired, but is packaged in older material, with an insert that doesn’t include the compounding instructions, Ms. Hamburg said during the briefing.
Dr. Frieden reiterated the importance of early antiviral treatment.
"In studies of very sick patients, death rates were cut by 75% or more by prompt use. For high-risk patients it can mean the difference between mild diseases and the intensive care unit or death."
High-risk groups include the elderly, children, anyone with a compromised immune system, children with neurologic or developmental disorders, and those with asthma, diabetes, or heart disease
"Even if a rapid flu test is negative for a high-risk patient, they should be treated in the first 48 hours of developing symptoms, because the rapid flu test does give a number of false negatives," Dr Frieden said.
Older and high-risk adults should especially avoid being around children who may be infected. "This is not a good time to babysit the grandchildren," he said.
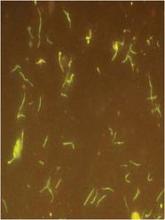
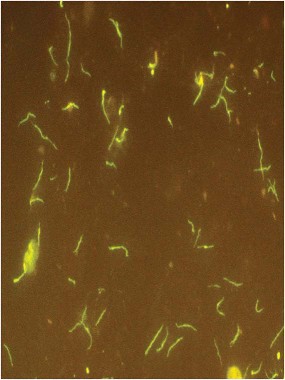
Newly described tick-borne infection found in New England
At least 24 people in the northeast United States have been infected with a newly described, as-yet-unnamed, tick-borne illness.
Borrelia miyamotoi is a spirochete related to Borrelia burgdorferi, the microorganism that causes Lyme disease, and is transmitted by the same four tick species that carry B. burgdorferi. In a case series of 875 patients, B. miyamotoi was present in 1% of healthy people who lived in a Lyme-endemic area, 3% of those with suspected Lyme, and 21% of those with a warm-weather viral-like illness in the late spring or summer, who attended a Lyme screening clinic.
Eighteen of the cases plus an additional one are described in two reports published in the Jan. 17 issue of the New England Journal of Medicine. These occurred in Connecticut, New York, and Rhode Island; one was in New Jersey. Since these cases were described, five additional patients have been confirmed, according to a spokesperson for Imugen, the Norwood, Mass., laboratory that performed immunoassays on some of the patients. Those were in Massachusetts, New Jersey, Connecticut, and New York.
There is reason to believe that patients can be coinfected with both B. miyamotoi and B. burgdorferi, and that ticks can harbor both simultaneously, said Dr. Peter Krause, an epidemiologist at Yale School of Public Health, New Haven, Conn., and primary author of one of the papers (N. Engl. J. Med. 2013 Jan. 17 [doi:10.1056/NEJMc1215469]).
But because the spirochetes are genetically similar, the illnesses they cause respond to the same antibiotic regimen, Dr. Krause said in an interview.
B. miyamotoi was first identified in Japan in 1995. In 2001, another Yale epidemiologist, Durland Fish, Ph.D., found it in 2.5% of tick nymphs collected in Maryland. In 2011, Dr. Krause and Dr. Fish together identified the first human cases – a series of 46 patients in Russia.
Dr. Krause reviewed serum samples collected from 1990 to 2010: 584 from healthy subjects who lived in Lyme-endemic areas and participated in a serotyping survey; 277 from the group with suspected Lyme disease; and 14 from those who presented at the Lyme clinic.
Four of the 18 positive subjects had reported the typical erythema migrans skin lesion. Two others had an atypical smaller-sized lesion – less than 5 cm in diameter. The rest had no reported rash.
Three patients were actually negative for B. miyamotoi during their acute illness, but showed seroconversion during their recovery. This indicates that B. miyamotoi probably did not cause the initial illness, suggesting coinfection with B. burgdorferi.
One of these patients was quite ill, according to the report. This patient had no skin lesion and no evidence of human granulocytic anaplasmosis coinfection. He showed a fever of more than 39 degrees Celsius, headache, neck stiffness, fatigue, myalgia, arthralgia, abdominal pain, cough, sore throat, and right inguinal lymphadenopathy. This patient recovered after a 14-day course of doxycycline; all of the other symptomatic patients treated with standard doxycycline or amoxicillin treatment also recovered.
None of the cases in Dr. Krause’s cohort showed any serious neurologic symptoms. However, the single patient reported from New Jersey – an immunocompromised 80-year-old women – did.
This woman was treated in 2011, Dr. Joseph L. Gugliotta and colleagues reported. She had experienced 4 months of declining mental status, including increasing confusion; withdrawal from family interactions; episodes of staying in bed; unstable gait; hearing problems; and a 30-pound weight loss and decreased appetite. She also had a recent diagnosis of depression, wrote Dr. Gugliotta, an infectious disease specialist in Flemington, N.J. (N. Engl. J. Med. 2013 Jan. 17 [doi:10.1056/NEJMoa1209039]).
The patient had been treated for non-Hodgkin’s lymphoma during 2005, and was still taking rituximab every 6 months.
She did not report having any recent tick bites, and had no typical Lyme rash. However, she had been treated for Lyme disease twice in the past, in 2006 and 2007, when she did present with the rash. She lived in a rural setting in a Lyme-endemic area.
The initial exam showed no new oncologic activity and no structural brain problems. A lumbar puncture showed pleocytosis. However, "an extraordinary density of morphologically distinct spirochetes" later identified as B. miyamotoi were found in CSF sediment, the authors noted.
The patient did not respond well to an initial infusion of ceftriaxone, developing a fever and low systolic blood pressure. She then received a 30-day course of 24 million units intravenous penicillin G. Her symptoms began to improve after 5 days of treatment. By the end of the regimen, she had returned to normal, Dr. Gugliotta and his coauthors said.
Any patient who develops an otherwise-unexplained viral-like illness in the spring or summer should be considered at high risk for one – or both – of the Borrelia illnesses, Dr. Krause said. A relapsing fever should be especially concerning. He stressed that the emblematic "bull’s-eye" rash may or may not occur.
Clinically, the B. miyamotoi spirochetes are more likely to be seen in blood smears than are their burgdorferi cousins. But B. miyamotoi can only be completely distinguished by serotyping or polymerase chain reaction, Dr. Krause said. These lab tests are not widely available now, but should be by early summer, he added.
Since effective treatment does not depend on positive identification, it’s not clinically necessary to determine the infective agent. But accurate tracking of the organism is important for epidemiologic data, and Dr. Krause encouraged physicians to pinpoint the cause of any tick-borne disease.
B. miyamotoi is certainly more widespread than the current case series suggest, Dr. Fish said in an interview. Because it’s been isolated in Japan, Germany, Russia, and now the United States, and in all of the tick species that carry Lyme, B. miyamotoi can be considered endemic in all northern hemisphere regions that harbor any of the four Lyme disease–causing tick species.
In fact, Dr. Fish said, many presumed cases of Lyme are probably due either to B. miyamotoi alone or to coinfection by both species.
"We saw it in about 2%," of ticks analyzed at the Yale lab, he said. "It appears to be widely distributed and I think it’s going to be seen more and more. This disease could actually be occurring as frequently as Lyme."
The 875-patient case series was supported by grants from the National Institutes of Health, the Gordon and Llura Gund Foundation, the Howard Hughes Medical Institute, and the G. Harold and Leila Y. Mathers Foundation. Dr. Gugliotta’s study was supported in part by grants from the National Institute of Health, the Evelyn Lilly Lutz Foundation and by a gift from Gordon and Lulie Gund. Neither Dr. Gugliotta, Dr. Krause, nor Dr. Fish had any financial disclosures.
At least 24 people in the northeast United States have been infected with a newly described, as-yet-unnamed, tick-borne illness.
Borrelia miyamotoi is a spirochete related to Borrelia burgdorferi, the microorganism that causes Lyme disease, and is transmitted by the same four tick species that carry B. burgdorferi. In a case series of 875 patients, B. miyamotoi was present in 1% of healthy people who lived in a Lyme-endemic area, 3% of those with suspected Lyme, and 21% of those with a warm-weather viral-like illness in the late spring or summer, who attended a Lyme screening clinic.
Eighteen of the cases plus an additional one are described in two reports published in the Jan. 17 issue of the New England Journal of Medicine. These occurred in Connecticut, New York, and Rhode Island; one was in New Jersey. Since these cases were described, five additional patients have been confirmed, according to a spokesperson for Imugen, the Norwood, Mass., laboratory that performed immunoassays on some of the patients. Those were in Massachusetts, New Jersey, Connecticut, and New York.
There is reason to believe that patients can be coinfected with both B. miyamotoi and B. burgdorferi, and that ticks can harbor both simultaneously, said Dr. Peter Krause, an epidemiologist at Yale School of Public Health, New Haven, Conn., and primary author of one of the papers (N. Engl. J. Med. 2013 Jan. 17 [doi:10.1056/NEJMc1215469]).
But because the spirochetes are genetically similar, the illnesses they cause respond to the same antibiotic regimen, Dr. Krause said in an interview.
B. miyamotoi was first identified in Japan in 1995. In 2001, another Yale epidemiologist, Durland Fish, Ph.D., found it in 2.5% of tick nymphs collected in Maryland. In 2011, Dr. Krause and Dr. Fish together identified the first human cases – a series of 46 patients in Russia.
Dr. Krause reviewed serum samples collected from 1990 to 2010: 584 from healthy subjects who lived in Lyme-endemic areas and participated in a serotyping survey; 277 from the group with suspected Lyme disease; and 14 from those who presented at the Lyme clinic.
Four of the 18 positive subjects had reported the typical erythema migrans skin lesion. Two others had an atypical smaller-sized lesion – less than 5 cm in diameter. The rest had no reported rash.
Three patients were actually negative for B. miyamotoi during their acute illness, but showed seroconversion during their recovery. This indicates that B. miyamotoi probably did not cause the initial illness, suggesting coinfection with B. burgdorferi.
One of these patients was quite ill, according to the report. This patient had no skin lesion and no evidence of human granulocytic anaplasmosis coinfection. He showed a fever of more than 39 degrees Celsius, headache, neck stiffness, fatigue, myalgia, arthralgia, abdominal pain, cough, sore throat, and right inguinal lymphadenopathy. This patient recovered after a 14-day course of doxycycline; all of the other symptomatic patients treated with standard doxycycline or amoxicillin treatment also recovered.
None of the cases in Dr. Krause’s cohort showed any serious neurologic symptoms. However, the single patient reported from New Jersey – an immunocompromised 80-year-old women – did.
This woman was treated in 2011, Dr. Joseph L. Gugliotta and colleagues reported. She had experienced 4 months of declining mental status, including increasing confusion; withdrawal from family interactions; episodes of staying in bed; unstable gait; hearing problems; and a 30-pound weight loss and decreased appetite. She also had a recent diagnosis of depression, wrote Dr. Gugliotta, an infectious disease specialist in Flemington, N.J. (N. Engl. J. Med. 2013 Jan. 17 [doi:10.1056/NEJMoa1209039]).
The patient had been treated for non-Hodgkin’s lymphoma during 2005, and was still taking rituximab every 6 months.
She did not report having any recent tick bites, and had no typical Lyme rash. However, she had been treated for Lyme disease twice in the past, in 2006 and 2007, when she did present with the rash. She lived in a rural setting in a Lyme-endemic area.
The initial exam showed no new oncologic activity and no structural brain problems. A lumbar puncture showed pleocytosis. However, "an extraordinary density of morphologically distinct spirochetes" later identified as B. miyamotoi were found in CSF sediment, the authors noted.
The patient did not respond well to an initial infusion of ceftriaxone, developing a fever and low systolic blood pressure. She then received a 30-day course of 24 million units intravenous penicillin G. Her symptoms began to improve after 5 days of treatment. By the end of the regimen, she had returned to normal, Dr. Gugliotta and his coauthors said.
Any patient who develops an otherwise-unexplained viral-like illness in the spring or summer should be considered at high risk for one – or both – of the Borrelia illnesses, Dr. Krause said. A relapsing fever should be especially concerning. He stressed that the emblematic "bull’s-eye" rash may or may not occur.
Clinically, the B. miyamotoi spirochetes are more likely to be seen in blood smears than are their burgdorferi cousins. But B. miyamotoi can only be completely distinguished by serotyping or polymerase chain reaction, Dr. Krause said. These lab tests are not widely available now, but should be by early summer, he added.
Since effective treatment does not depend on positive identification, it’s not clinically necessary to determine the infective agent. But accurate tracking of the organism is important for epidemiologic data, and Dr. Krause encouraged physicians to pinpoint the cause of any tick-borne disease.
B. miyamotoi is certainly more widespread than the current case series suggest, Dr. Fish said in an interview. Because it’s been isolated in Japan, Germany, Russia, and now the United States, and in all of the tick species that carry Lyme, B. miyamotoi can be considered endemic in all northern hemisphere regions that harbor any of the four Lyme disease–causing tick species.
In fact, Dr. Fish said, many presumed cases of Lyme are probably due either to B. miyamotoi alone or to coinfection by both species.
"We saw it in about 2%," of ticks analyzed at the Yale lab, he said. "It appears to be widely distributed and I think it’s going to be seen more and more. This disease could actually be occurring as frequently as Lyme."
The 875-patient case series was supported by grants from the National Institutes of Health, the Gordon and Llura Gund Foundation, the Howard Hughes Medical Institute, and the G. Harold and Leila Y. Mathers Foundation. Dr. Gugliotta’s study was supported in part by grants from the National Institute of Health, the Evelyn Lilly Lutz Foundation and by a gift from Gordon and Lulie Gund. Neither Dr. Gugliotta, Dr. Krause, nor Dr. Fish had any financial disclosures.
At least 24 people in the northeast United States have been infected with a newly described, as-yet-unnamed, tick-borne illness.
Borrelia miyamotoi is a spirochete related to Borrelia burgdorferi, the microorganism that causes Lyme disease, and is transmitted by the same four tick species that carry B. burgdorferi. In a case series of 875 patients, B. miyamotoi was present in 1% of healthy people who lived in a Lyme-endemic area, 3% of those with suspected Lyme, and 21% of those with a warm-weather viral-like illness in the late spring or summer, who attended a Lyme screening clinic.
Eighteen of the cases plus an additional one are described in two reports published in the Jan. 17 issue of the New England Journal of Medicine. These occurred in Connecticut, New York, and Rhode Island; one was in New Jersey. Since these cases were described, five additional patients have been confirmed, according to a spokesperson for Imugen, the Norwood, Mass., laboratory that performed immunoassays on some of the patients. Those were in Massachusetts, New Jersey, Connecticut, and New York.
There is reason to believe that patients can be coinfected with both B. miyamotoi and B. burgdorferi, and that ticks can harbor both simultaneously, said Dr. Peter Krause, an epidemiologist at Yale School of Public Health, New Haven, Conn., and primary author of one of the papers (N. Engl. J. Med. 2013 Jan. 17 [doi:10.1056/NEJMc1215469]).
But because the spirochetes are genetically similar, the illnesses they cause respond to the same antibiotic regimen, Dr. Krause said in an interview.
B. miyamotoi was first identified in Japan in 1995. In 2001, another Yale epidemiologist, Durland Fish, Ph.D., found it in 2.5% of tick nymphs collected in Maryland. In 2011, Dr. Krause and Dr. Fish together identified the first human cases – a series of 46 patients in Russia.
Dr. Krause reviewed serum samples collected from 1990 to 2010: 584 from healthy subjects who lived in Lyme-endemic areas and participated in a serotyping survey; 277 from the group with suspected Lyme disease; and 14 from those who presented at the Lyme clinic.
Four of the 18 positive subjects had reported the typical erythema migrans skin lesion. Two others had an atypical smaller-sized lesion – less than 5 cm in diameter. The rest had no reported rash.
Three patients were actually negative for B. miyamotoi during their acute illness, but showed seroconversion during their recovery. This indicates that B. miyamotoi probably did not cause the initial illness, suggesting coinfection with B. burgdorferi.
One of these patients was quite ill, according to the report. This patient had no skin lesion and no evidence of human granulocytic anaplasmosis coinfection. He showed a fever of more than 39 degrees Celsius, headache, neck stiffness, fatigue, myalgia, arthralgia, abdominal pain, cough, sore throat, and right inguinal lymphadenopathy. This patient recovered after a 14-day course of doxycycline; all of the other symptomatic patients treated with standard doxycycline or amoxicillin treatment also recovered.
None of the cases in Dr. Krause’s cohort showed any serious neurologic symptoms. However, the single patient reported from New Jersey – an immunocompromised 80-year-old women – did.
This woman was treated in 2011, Dr. Joseph L. Gugliotta and colleagues reported. She had experienced 4 months of declining mental status, including increasing confusion; withdrawal from family interactions; episodes of staying in bed; unstable gait; hearing problems; and a 30-pound weight loss and decreased appetite. She also had a recent diagnosis of depression, wrote Dr. Gugliotta, an infectious disease specialist in Flemington, N.J. (N. Engl. J. Med. 2013 Jan. 17 [doi:10.1056/NEJMoa1209039]).
The patient had been treated for non-Hodgkin’s lymphoma during 2005, and was still taking rituximab every 6 months.
She did not report having any recent tick bites, and had no typical Lyme rash. However, she had been treated for Lyme disease twice in the past, in 2006 and 2007, when she did present with the rash. She lived in a rural setting in a Lyme-endemic area.
The initial exam showed no new oncologic activity and no structural brain problems. A lumbar puncture showed pleocytosis. However, "an extraordinary density of morphologically distinct spirochetes" later identified as B. miyamotoi were found in CSF sediment, the authors noted.
The patient did not respond well to an initial infusion of ceftriaxone, developing a fever and low systolic blood pressure. She then received a 30-day course of 24 million units intravenous penicillin G. Her symptoms began to improve after 5 days of treatment. By the end of the regimen, she had returned to normal, Dr. Gugliotta and his coauthors said.
Any patient who develops an otherwise-unexplained viral-like illness in the spring or summer should be considered at high risk for one – or both – of the Borrelia illnesses, Dr. Krause said. A relapsing fever should be especially concerning. He stressed that the emblematic "bull’s-eye" rash may or may not occur.
Clinically, the B. miyamotoi spirochetes are more likely to be seen in blood smears than are their burgdorferi cousins. But B. miyamotoi can only be completely distinguished by serotyping or polymerase chain reaction, Dr. Krause said. These lab tests are not widely available now, but should be by early summer, he added.
Since effective treatment does not depend on positive identification, it’s not clinically necessary to determine the infective agent. But accurate tracking of the organism is important for epidemiologic data, and Dr. Krause encouraged physicians to pinpoint the cause of any tick-borne disease.
B. miyamotoi is certainly more widespread than the current case series suggest, Dr. Fish said in an interview. Because it’s been isolated in Japan, Germany, Russia, and now the United States, and in all of the tick species that carry Lyme, B. miyamotoi can be considered endemic in all northern hemisphere regions that harbor any of the four Lyme disease–causing tick species.
In fact, Dr. Fish said, many presumed cases of Lyme are probably due either to B. miyamotoi alone or to coinfection by both species.
"We saw it in about 2%," of ticks analyzed at the Yale lab, he said. "It appears to be widely distributed and I think it’s going to be seen more and more. This disease could actually be occurring as frequently as Lyme."
The 875-patient case series was supported by grants from the National Institutes of Health, the Gordon and Llura Gund Foundation, the Howard Hughes Medical Institute, and the G. Harold and Leila Y. Mathers Foundation. Dr. Gugliotta’s study was supported in part by grants from the National Institute of Health, the Evelyn Lilly Lutz Foundation and by a gift from Gordon and Lulie Gund. Neither Dr. Gugliotta, Dr. Krause, nor Dr. Fish had any financial disclosures.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: A newly described illness, similar to Lyme disease and caused by a related organism, has been reported in 24 people in New England and New Jersey.
Data Source: A review of serum samples from a single case report and from data supplied by a laboratory that can test for the organism.
Disclosures: The 875-patient case series was supported by grants from the National Institutes of Health, the Gordon and Llura Gund Foundation, the Howard Hughes Medical Institute, and the G. Harold and Leila Y. Mathers Foundation. Dr. Gugliotta’s study was supported in part by grants from the National Institute of Health, the Evelyn Lilly Lutz Foundation and by a gift from Gordon and Lulie Gund. Neither Dr. Gugliotta, Dr. Krause, nor Dr. Fish had any financial disclosures.
Feds announce $11 million Alzheimer's research grant
Alzheimer’s research will get a sizable boost this year, with an $11 million grant from the federal government and the promise of up to $55 million by 2018.
The National Institute on Aging allocated the funds to further the Alzheimer’s Disease Cooperative Study (ADCS), a long-term project that many researchers hope could result in more effective treatments – and perhaps even a cure for the disease.
"The ADCS is a key initiative in the federal program to discover, develop, and test new Alzheimer’s treatments and diagnostic tools," Dr. Richard J. Hodes, director of the National Institute on Aging, wrote in a press statement. "Over the years, it has proved invaluable in advancing our understanding about the disease and how to conduct research in this challenging area. I am particularly excited that this round of studies will use what we have learned by testing interventions presymptomatically, as early as we can in the development of the disease, where we now think the best hope lies for keeping Alzheimer’s at bay."
ADCS is a consortium of academic medical centers and clinics set up by NIH in 1991 to collaborate on the development of Alzheimer’s treatments and diagnostic tools. This newest round of studies will test drug and exercise interventions in people in the early stages of the disease, evaluate a medication to reduce agitation in people with Alzheimer’s dementia, and search for a way to speed testing of drugs in clinical trials. The grant makes possible four new studies:
• The Anti-Amyloid Treatment in Asymptomatic Alzheimer’s disease (A4) secondary prevention trial will test an amyloid-clearing drug in the presymptomatic stage of the disease. The study population will consist of 1,000 symptom-free older volunteers who have had positron emission tomography brain images confirming abnormal levels of amyloid. The 3-year trial will probably test a single agent, but the specifics have yet to be announced. However, it’s likely to be a monoclonal antibody, several of which have already shown some activity against beta-amyloid brain plaques, according to principal investigator Dr. Reisa A. Sperling, who is the director of the Center for Alzheimer’s Research and Treatment and professor of neurology at Harvard Medical School, Boston.
• The year-long Exercise MCI Trial seeks to determine how exercise could impact the progression of mild cognitive impairment. Sedentary patients with MCI will be randomized to one of two exercise programs: gentle stretching or vigorous aerobic workouts. Endpoints include changes in cognition, cerebrospinal fluid biomarkers, and brain imaging.
• The Prazosin Treatment for Disruptive Agitation in Alzheimer’s Disease will determine whether prazosin is well tolerated in frail, elderly people as a treatment for agitation. Dr. Elaine R. Peskind, the Friends of Alzheimer’s Research Professor of Psychiatry and Behavioral Sciences at the University of Washington in Seattle, is heading the project, which seeks 120 patients with Alzheimer’s who experience at least two disruptive episodes per week. Patients will be randomized to the drug or placebo for 24 weeks. Endpoints are changes on the Clinical Global Impression of Change and Neuropsychiatric Inventory total score. In 2009, Dr. Peskind and her team conducted a small placebo-controlled trial of 22 Alzheimer’s patients with agitation/aggression. Findings from that study showed that the drug improved behavioral symptoms in patients.
• The CSF Pharmacodynamic Trial will examine how some drugs in development affect levels of beta-amyloid and tau levels over time. The results will help determine whether a drug is a fit candidate for further investigation and a clinical trial.
The announcement of a continuing federal commitment is a valuable contribution to the Alzheimer’s fight, but much more is needed, said William H. Thies, Ph.D., chief medical and scientific officer of the Alzheimer’s Association, Chicago.
"The studies announced today are significant steps in the fight to better treat and eventually prevent Alzheimer’s disease," Dr. Thies said in an interview. "However, because of the chronic underfunding of Alzheimer’s research, there are many more equally valid and necessary investigations that are not happening. The Alzheimer’s Association believes that if we are to more quickly unravel the important questions about Alzheimer’s disease cause, risk, treatment, and prevention, we must make a greater commitment to research."
The first-ever U.S. National Plan to Address Alzheimer’s Disease was unveiled in May 2012, and now this plan must be effectively implemented, he added. "An additional $100 million has been recommended for 2013 for Alzheimer’s research, and now must be confirmed."
Dr. Hodes, Dr. Sperling, Dr. Peskind, and Dr. Thies did not report any financial conflicts of interest.
The National Institute on Aging, Alzheimer’s Disease Cooperative Study, ADCS, effective treatments, Dr. Richard J. Hodes, Exercise MCI Trial
Alzheimer’s research will get a sizable boost this year, with an $11 million grant from the federal government and the promise of up to $55 million by 2018.
The National Institute on Aging allocated the funds to further the Alzheimer’s Disease Cooperative Study (ADCS), a long-term project that many researchers hope could result in more effective treatments – and perhaps even a cure for the disease.
"The ADCS is a key initiative in the federal program to discover, develop, and test new Alzheimer’s treatments and diagnostic tools," Dr. Richard J. Hodes, director of the National Institute on Aging, wrote in a press statement. "Over the years, it has proved invaluable in advancing our understanding about the disease and how to conduct research in this challenging area. I am particularly excited that this round of studies will use what we have learned by testing interventions presymptomatically, as early as we can in the development of the disease, where we now think the best hope lies for keeping Alzheimer’s at bay."
ADCS is a consortium of academic medical centers and clinics set up by NIH in 1991 to collaborate on the development of Alzheimer’s treatments and diagnostic tools. This newest round of studies will test drug and exercise interventions in people in the early stages of the disease, evaluate a medication to reduce agitation in people with Alzheimer’s dementia, and search for a way to speed testing of drugs in clinical trials. The grant makes possible four new studies:
• The Anti-Amyloid Treatment in Asymptomatic Alzheimer’s disease (A4) secondary prevention trial will test an amyloid-clearing drug in the presymptomatic stage of the disease. The study population will consist of 1,000 symptom-free older volunteers who have had positron emission tomography brain images confirming abnormal levels of amyloid. The 3-year trial will probably test a single agent, but the specifics have yet to be announced. However, it’s likely to be a monoclonal antibody, several of which have already shown some activity against beta-amyloid brain plaques, according to principal investigator Dr. Reisa A. Sperling, who is the director of the Center for Alzheimer’s Research and Treatment and professor of neurology at Harvard Medical School, Boston.
• The year-long Exercise MCI Trial seeks to determine how exercise could impact the progression of mild cognitive impairment. Sedentary patients with MCI will be randomized to one of two exercise programs: gentle stretching or vigorous aerobic workouts. Endpoints include changes in cognition, cerebrospinal fluid biomarkers, and brain imaging.
• The Prazosin Treatment for Disruptive Agitation in Alzheimer’s Disease will determine whether prazosin is well tolerated in frail, elderly people as a treatment for agitation. Dr. Elaine R. Peskind, the Friends of Alzheimer’s Research Professor of Psychiatry and Behavioral Sciences at the University of Washington in Seattle, is heading the project, which seeks 120 patients with Alzheimer’s who experience at least two disruptive episodes per week. Patients will be randomized to the drug or placebo for 24 weeks. Endpoints are changes on the Clinical Global Impression of Change and Neuropsychiatric Inventory total score. In 2009, Dr. Peskind and her team conducted a small placebo-controlled trial of 22 Alzheimer’s patients with agitation/aggression. Findings from that study showed that the drug improved behavioral symptoms in patients.
• The CSF Pharmacodynamic Trial will examine how some drugs in development affect levels of beta-amyloid and tau levels over time. The results will help determine whether a drug is a fit candidate for further investigation and a clinical trial.
The announcement of a continuing federal commitment is a valuable contribution to the Alzheimer’s fight, but much more is needed, said William H. Thies, Ph.D., chief medical and scientific officer of the Alzheimer’s Association, Chicago.
"The studies announced today are significant steps in the fight to better treat and eventually prevent Alzheimer’s disease," Dr. Thies said in an interview. "However, because of the chronic underfunding of Alzheimer’s research, there are many more equally valid and necessary investigations that are not happening. The Alzheimer’s Association believes that if we are to more quickly unravel the important questions about Alzheimer’s disease cause, risk, treatment, and prevention, we must make a greater commitment to research."
The first-ever U.S. National Plan to Address Alzheimer’s Disease was unveiled in May 2012, and now this plan must be effectively implemented, he added. "An additional $100 million has been recommended for 2013 for Alzheimer’s research, and now must be confirmed."
Dr. Hodes, Dr. Sperling, Dr. Peskind, and Dr. Thies did not report any financial conflicts of interest.
Alzheimer’s research will get a sizable boost this year, with an $11 million grant from the federal government and the promise of up to $55 million by 2018.
The National Institute on Aging allocated the funds to further the Alzheimer’s Disease Cooperative Study (ADCS), a long-term project that many researchers hope could result in more effective treatments – and perhaps even a cure for the disease.
"The ADCS is a key initiative in the federal program to discover, develop, and test new Alzheimer’s treatments and diagnostic tools," Dr. Richard J. Hodes, director of the National Institute on Aging, wrote in a press statement. "Over the years, it has proved invaluable in advancing our understanding about the disease and how to conduct research in this challenging area. I am particularly excited that this round of studies will use what we have learned by testing interventions presymptomatically, as early as we can in the development of the disease, where we now think the best hope lies for keeping Alzheimer’s at bay."
ADCS is a consortium of academic medical centers and clinics set up by NIH in 1991 to collaborate on the development of Alzheimer’s treatments and diagnostic tools. This newest round of studies will test drug and exercise interventions in people in the early stages of the disease, evaluate a medication to reduce agitation in people with Alzheimer’s dementia, and search for a way to speed testing of drugs in clinical trials. The grant makes possible four new studies:
• The Anti-Amyloid Treatment in Asymptomatic Alzheimer’s disease (A4) secondary prevention trial will test an amyloid-clearing drug in the presymptomatic stage of the disease. The study population will consist of 1,000 symptom-free older volunteers who have had positron emission tomography brain images confirming abnormal levels of amyloid. The 3-year trial will probably test a single agent, but the specifics have yet to be announced. However, it’s likely to be a monoclonal antibody, several of which have already shown some activity against beta-amyloid brain plaques, according to principal investigator Dr. Reisa A. Sperling, who is the director of the Center for Alzheimer’s Research and Treatment and professor of neurology at Harvard Medical School, Boston.
• The year-long Exercise MCI Trial seeks to determine how exercise could impact the progression of mild cognitive impairment. Sedentary patients with MCI will be randomized to one of two exercise programs: gentle stretching or vigorous aerobic workouts. Endpoints include changes in cognition, cerebrospinal fluid biomarkers, and brain imaging.
• The Prazosin Treatment for Disruptive Agitation in Alzheimer’s Disease will determine whether prazosin is well tolerated in frail, elderly people as a treatment for agitation. Dr. Elaine R. Peskind, the Friends of Alzheimer’s Research Professor of Psychiatry and Behavioral Sciences at the University of Washington in Seattle, is heading the project, which seeks 120 patients with Alzheimer’s who experience at least two disruptive episodes per week. Patients will be randomized to the drug or placebo for 24 weeks. Endpoints are changes on the Clinical Global Impression of Change and Neuropsychiatric Inventory total score. In 2009, Dr. Peskind and her team conducted a small placebo-controlled trial of 22 Alzheimer’s patients with agitation/aggression. Findings from that study showed that the drug improved behavioral symptoms in patients.
• The CSF Pharmacodynamic Trial will examine how some drugs in development affect levels of beta-amyloid and tau levels over time. The results will help determine whether a drug is a fit candidate for further investigation and a clinical trial.
The announcement of a continuing federal commitment is a valuable contribution to the Alzheimer’s fight, but much more is needed, said William H. Thies, Ph.D., chief medical and scientific officer of the Alzheimer’s Association, Chicago.
"The studies announced today are significant steps in the fight to better treat and eventually prevent Alzheimer’s disease," Dr. Thies said in an interview. "However, because of the chronic underfunding of Alzheimer’s research, there are many more equally valid and necessary investigations that are not happening. The Alzheimer’s Association believes that if we are to more quickly unravel the important questions about Alzheimer’s disease cause, risk, treatment, and prevention, we must make a greater commitment to research."
The first-ever U.S. National Plan to Address Alzheimer’s Disease was unveiled in May 2012, and now this plan must be effectively implemented, he added. "An additional $100 million has been recommended for 2013 for Alzheimer’s research, and now must be confirmed."
Dr. Hodes, Dr. Sperling, Dr. Peskind, and Dr. Thies did not report any financial conflicts of interest.
The National Institute on Aging, Alzheimer’s Disease Cooperative Study, ADCS, effective treatments, Dr. Richard J. Hodes, Exercise MCI Trial
The National Institute on Aging, Alzheimer’s Disease Cooperative Study, ADCS, effective treatments, Dr. Richard J. Hodes, Exercise MCI Trial
Scoring tool targets hospitalized patients needing acid suppressants
A scoring system based on demographics and comorbidities predicted which hospitalized patients were at risk for nosocomial gastrointestinal bleeding.
Used as a guideline for administering acid suppressors, the system found that treating fewer than 100 high-risk patients would prevent one case of bleeding, Dr. Shoshana Herzig and her colleagues wrote in the January online issue of General Internal Medicine (2013 [doi:10.1007/s11606-012-2296-x]).
"This scoring system allows identification of subsets of patients in whom the risk of nosocomial gastrointestinal bleeding may be higher enough to warrant the use of prophylactic acid-suppressive medication, in the absence of other indicators for use," said Dr. Herzig of Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, and her coauthors.
The model "allows for risk stratification of patients using readily available information, and can be used to guide more selective use of acid-suppressive medication in patients outside of the ICU," the investigators wrote. The investigators reviewed about 76,000 records of noncritically ill patients hospitalized for more than 3 days, from 2004 through 2007. Nosocomial gastrointestinal bleeding occurred in about 3% of the group.
The authors constructed the risk score by identifying several factors that could potentially affect bleeding, including age, sex, comorbid conditions, admission service, and the receipt of certain medications during hospitalization.
A multivariate analysis identified those factors that significantly correlated with gastrointestinal bleeding. These included:
• Age more than 60 years (odds ratio, 2.2).
• Male sex (OR, 1.6).
• Liver disease (OR, 2.1).
• Acute renal failure (OR, 1.9).
• Admission to a medical service (OR, 2.7).
• Prophylactic anticoagulation (OR, 1.7).
• Coagulotherapy without antiplatelet medication (OR, 2.6).
• Coagulotherapy with one antiplatelet agent (OR, 3.2).
• Coagulotherapy with dual antiplatelet agents (OR, 3.3).
Based on these factors, they constructed a points-based risk scoring system that significantly correlated with the incidence of gastrointestinal bleeding. Patients in the lowest-score quartile were at the least risk of bleeding (less than 0.3%), while those in the highest quartile had the highest risk (1.5%).
The number needed to treat (NNT) to prevent one bleed increased as the risk score increased. The NNT was 500 in patients whose score was at least 6, 179 in those whose score was at least 8, 95 in those whose score was at least 10 (moderate risk), and 48 in those whose score was at least 12 (high risk).
The patients’ mean age was 56 years, although the range was very wide (18-107 years); 40% were male. Most patients received an acid-suppressing medication (58%). Proton pump inhibitors were most commonly used (81%); 29% received a histamine-2 receptor antagonist.
"With further validation at other medical centers, this scoring system may help clinicians individualize the decision to prescribe acid-suppressive medication as prophylaxis," the authors said.
The study was funded by grants from the National Institute on Aging and the National Center for Research Resources. None of the authors reported any financial conflicts.
A scoring system based on demographics and comorbidities predicted which hospitalized patients were at risk for nosocomial gastrointestinal bleeding.
Used as a guideline for administering acid suppressors, the system found that treating fewer than 100 high-risk patients would prevent one case of bleeding, Dr. Shoshana Herzig and her colleagues wrote in the January online issue of General Internal Medicine (2013 [doi:10.1007/s11606-012-2296-x]).
"This scoring system allows identification of subsets of patients in whom the risk of nosocomial gastrointestinal bleeding may be higher enough to warrant the use of prophylactic acid-suppressive medication, in the absence of other indicators for use," said Dr. Herzig of Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, and her coauthors.
The model "allows for risk stratification of patients using readily available information, and can be used to guide more selective use of acid-suppressive medication in patients outside of the ICU," the investigators wrote. The investigators reviewed about 76,000 records of noncritically ill patients hospitalized for more than 3 days, from 2004 through 2007. Nosocomial gastrointestinal bleeding occurred in about 3% of the group.
The authors constructed the risk score by identifying several factors that could potentially affect bleeding, including age, sex, comorbid conditions, admission service, and the receipt of certain medications during hospitalization.
A multivariate analysis identified those factors that significantly correlated with gastrointestinal bleeding. These included:
• Age more than 60 years (odds ratio, 2.2).
• Male sex (OR, 1.6).
• Liver disease (OR, 2.1).
• Acute renal failure (OR, 1.9).
• Admission to a medical service (OR, 2.7).
• Prophylactic anticoagulation (OR, 1.7).
• Coagulotherapy without antiplatelet medication (OR, 2.6).
• Coagulotherapy with one antiplatelet agent (OR, 3.2).
• Coagulotherapy with dual antiplatelet agents (OR, 3.3).
Based on these factors, they constructed a points-based risk scoring system that significantly correlated with the incidence of gastrointestinal bleeding. Patients in the lowest-score quartile were at the least risk of bleeding (less than 0.3%), while those in the highest quartile had the highest risk (1.5%).
The number needed to treat (NNT) to prevent one bleed increased as the risk score increased. The NNT was 500 in patients whose score was at least 6, 179 in those whose score was at least 8, 95 in those whose score was at least 10 (moderate risk), and 48 in those whose score was at least 12 (high risk).
The patients’ mean age was 56 years, although the range was very wide (18-107 years); 40% were male. Most patients received an acid-suppressing medication (58%). Proton pump inhibitors were most commonly used (81%); 29% received a histamine-2 receptor antagonist.
"With further validation at other medical centers, this scoring system may help clinicians individualize the decision to prescribe acid-suppressive medication as prophylaxis," the authors said.
The study was funded by grants from the National Institute on Aging and the National Center for Research Resources. None of the authors reported any financial conflicts.
A scoring system based on demographics and comorbidities predicted which hospitalized patients were at risk for nosocomial gastrointestinal bleeding.
Used as a guideline for administering acid suppressors, the system found that treating fewer than 100 high-risk patients would prevent one case of bleeding, Dr. Shoshana Herzig and her colleagues wrote in the January online issue of General Internal Medicine (2013 [doi:10.1007/s11606-012-2296-x]).
"This scoring system allows identification of subsets of patients in whom the risk of nosocomial gastrointestinal bleeding may be higher enough to warrant the use of prophylactic acid-suppressive medication, in the absence of other indicators for use," said Dr. Herzig of Beth Israel Deaconess Medical Center and Harvard Medical School, Boston, and her coauthors.
The model "allows for risk stratification of patients using readily available information, and can be used to guide more selective use of acid-suppressive medication in patients outside of the ICU," the investigators wrote. The investigators reviewed about 76,000 records of noncritically ill patients hospitalized for more than 3 days, from 2004 through 2007. Nosocomial gastrointestinal bleeding occurred in about 3% of the group.
The authors constructed the risk score by identifying several factors that could potentially affect bleeding, including age, sex, comorbid conditions, admission service, and the receipt of certain medications during hospitalization.
A multivariate analysis identified those factors that significantly correlated with gastrointestinal bleeding. These included:
• Age more than 60 years (odds ratio, 2.2).
• Male sex (OR, 1.6).
• Liver disease (OR, 2.1).
• Acute renal failure (OR, 1.9).
• Admission to a medical service (OR, 2.7).
• Prophylactic anticoagulation (OR, 1.7).
• Coagulotherapy without antiplatelet medication (OR, 2.6).
• Coagulotherapy with one antiplatelet agent (OR, 3.2).
• Coagulotherapy with dual antiplatelet agents (OR, 3.3).
Based on these factors, they constructed a points-based risk scoring system that significantly correlated with the incidence of gastrointestinal bleeding. Patients in the lowest-score quartile were at the least risk of bleeding (less than 0.3%), while those in the highest quartile had the highest risk (1.5%).
The number needed to treat (NNT) to prevent one bleed increased as the risk score increased. The NNT was 500 in patients whose score was at least 6, 179 in those whose score was at least 8, 95 in those whose score was at least 10 (moderate risk), and 48 in those whose score was at least 12 (high risk).
The patients’ mean age was 56 years, although the range was very wide (18-107 years); 40% were male. Most patients received an acid-suppressing medication (58%). Proton pump inhibitors were most commonly used (81%); 29% received a histamine-2 receptor antagonist.
"With further validation at other medical centers, this scoring system may help clinicians individualize the decision to prescribe acid-suppressive medication as prophylaxis," the authors said.
The study was funded by grants from the National Institute on Aging and the National Center for Research Resources. None of the authors reported any financial conflicts.
FROM GENERAL INTERNAL MEDICINE
Major Finding: Acid suppressants would prevent one nosocomial gastrointestinal bleed for every 48 high-risk patients and every 95 moderate-risk patients treated, a risk-scoring system showed.
Data Source: A retrospective chart study of more than 76,000 patients.
Disclosures: The study was funded by grants from the National Institute on Aging and the National Center for Research Resources. None of the authors reported having any financial conflicts.
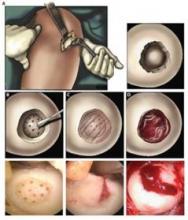
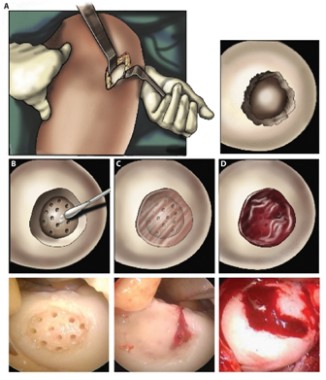
Hydrogel repaired joint defects, sped healing
A light-cured hydrogel combined with microfracture encouraged cartilage repair in knee joints and was associated with reduced pain, a small pilot trial has found.
When applied to a debrided defect in a knee joint, the gel seeped into subchondral microfracture, trapping blood released by microfracture and forming a clot that filled most of the bone defect. Compared with clots formed solely by microfracture, the gel-clot was larger in volume, suggesting that it "may be able to retain more proteins and cells in the defect space ... further augmenting the biological factors that can promote new tissue growth," Blanka Sharma, Ph.D., of Johns Hopkins University, Baltimore, and her colleagues wrote Jan. 9 in Science Translational Medicine.
The authors employed the light-polymerized gel with microfracture in 15 patients with a 2-4 cm2 symptomatic cartilage defect on the medial femoral condyle; three patients with similar defects underwent standard microfracture surgery. The study compared healing, function, and pain over a 6-month postoperative period (Sci. Trans. Med. 2013;5:167ra6).
At 3 months after surgery, MRI scanning in the investigational group showed a combination of clot material, hydrogel, and synovial fluid in the defect. By 6 months, new tissue filled an average of 86% of the defect in the investigational group, compared with an average defect fill of 64% in the microfracture-only group – a significant difference.
Most patients in the investigational group evaluated by MRI (12 of 14) had more than a 75% defect filling, compared with 1 of the 3 control patients.
Most patients who received the hydrogel also experienced significant reductions in pain frequency and severity, with the biggest improvements occurring in the first 3 months after surgery. One patient in this group continued to have pain at 6 months. The probable cause of pain was a misalignment in the treated knee, since the defect was more than 75% filled with new tissue, the authors said.
Control patients experienced similar significant reductions in pain frequency and severity during the first 3 months after surgery. By 6 months, one patient in the group continued to experience improvements in pain; this person "also had a high defect fill (84%), whereas the other two patients had fill values less than 75%," the authors noted.
At 6 months, pain frequency scores were not significantly different between the two groups. There were similar findings in pain severity scores. Knee function improved similarly in both groups.
The study was funded by the National Institutes of Health and the Arthritis Foundation. None of the authors had any financial disclosures.
A light-cured hydrogel combined with microfracture encouraged cartilage repair in knee joints and was associated with reduced pain, a small pilot trial has found.
When applied to a debrided defect in a knee joint, the gel seeped into subchondral microfracture, trapping blood released by microfracture and forming a clot that filled most of the bone defect. Compared with clots formed solely by microfracture, the gel-clot was larger in volume, suggesting that it "may be able to retain more proteins and cells in the defect space ... further augmenting the biological factors that can promote new tissue growth," Blanka Sharma, Ph.D., of Johns Hopkins University, Baltimore, and her colleagues wrote Jan. 9 in Science Translational Medicine.
The authors employed the light-polymerized gel with microfracture in 15 patients with a 2-4 cm2 symptomatic cartilage defect on the medial femoral condyle; three patients with similar defects underwent standard microfracture surgery. The study compared healing, function, and pain over a 6-month postoperative period (Sci. Trans. Med. 2013;5:167ra6).
At 3 months after surgery, MRI scanning in the investigational group showed a combination of clot material, hydrogel, and synovial fluid in the defect. By 6 months, new tissue filled an average of 86% of the defect in the investigational group, compared with an average defect fill of 64% in the microfracture-only group – a significant difference.
Most patients in the investigational group evaluated by MRI (12 of 14) had more than a 75% defect filling, compared with 1 of the 3 control patients.
Most patients who received the hydrogel also experienced significant reductions in pain frequency and severity, with the biggest improvements occurring in the first 3 months after surgery. One patient in this group continued to have pain at 6 months. The probable cause of pain was a misalignment in the treated knee, since the defect was more than 75% filled with new tissue, the authors said.
Control patients experienced similar significant reductions in pain frequency and severity during the first 3 months after surgery. By 6 months, one patient in the group continued to experience improvements in pain; this person "also had a high defect fill (84%), whereas the other two patients had fill values less than 75%," the authors noted.
At 6 months, pain frequency scores were not significantly different between the two groups. There were similar findings in pain severity scores. Knee function improved similarly in both groups.
The study was funded by the National Institutes of Health and the Arthritis Foundation. None of the authors had any financial disclosures.
A light-cured hydrogel combined with microfracture encouraged cartilage repair in knee joints and was associated with reduced pain, a small pilot trial has found.
When applied to a debrided defect in a knee joint, the gel seeped into subchondral microfracture, trapping blood released by microfracture and forming a clot that filled most of the bone defect. Compared with clots formed solely by microfracture, the gel-clot was larger in volume, suggesting that it "may be able to retain more proteins and cells in the defect space ... further augmenting the biological factors that can promote new tissue growth," Blanka Sharma, Ph.D., of Johns Hopkins University, Baltimore, and her colleagues wrote Jan. 9 in Science Translational Medicine.
The authors employed the light-polymerized gel with microfracture in 15 patients with a 2-4 cm2 symptomatic cartilage defect on the medial femoral condyle; three patients with similar defects underwent standard microfracture surgery. The study compared healing, function, and pain over a 6-month postoperative period (Sci. Trans. Med. 2013;5:167ra6).
At 3 months after surgery, MRI scanning in the investigational group showed a combination of clot material, hydrogel, and synovial fluid in the defect. By 6 months, new tissue filled an average of 86% of the defect in the investigational group, compared with an average defect fill of 64% in the microfracture-only group – a significant difference.
Most patients in the investigational group evaluated by MRI (12 of 14) had more than a 75% defect filling, compared with 1 of the 3 control patients.
Most patients who received the hydrogel also experienced significant reductions in pain frequency and severity, with the biggest improvements occurring in the first 3 months after surgery. One patient in this group continued to have pain at 6 months. The probable cause of pain was a misalignment in the treated knee, since the defect was more than 75% filled with new tissue, the authors said.
Control patients experienced similar significant reductions in pain frequency and severity during the first 3 months after surgery. By 6 months, one patient in the group continued to experience improvements in pain; this person "also had a high defect fill (84%), whereas the other two patients had fill values less than 75%," the authors noted.
At 6 months, pain frequency scores were not significantly different between the two groups. There were similar findings in pain severity scores. Knee function improved similarly in both groups.
The study was funded by the National Institutes of Health and the Arthritis Foundation. None of the authors had any financial disclosures.
FROM SCIENCE TRANSLATIONAL MEDICINE
Major Finding: After 6 months, joint defects treated with microfracture plus a photo-polymerized hydrogel were 86% filled, compared with a 64% fill of defects treated with microfracture alone.
Data Source: A pilot study of 18 patients, 15 of whom received the combination treatment.
Disclosures: None of the authors had any financial disclosures. The National Institutes of Health and the Arthritis Foundation funded the study.
Some psychiatric disorders hamper ability to get pregnant
Some psychiatric disorders appear to be associated with lower fecundity in both men and women, suggesting that natural selection attempts to discourage the perpetuation of genetic variants associated with them.
Instead, new mutations could be one reason that the disorders continue to exist, Robert A. Power and his colleagues wrote in the January issue of JAMA Psychiatry (formerly Archives of General Psychiatry).
The authors also found that psychiatric disorders affected men’s fecundity more than women’s. "This sex-specific effect suggests that psychiatric morbidity impairs interest or ability to find suitable mating partners or inhibits biological fertility to a greater extent in men," wrote Mr. Power of Kings College, London.
The study data were extracted from two of Sweden’s population registries – the Multi-Generation Register and the Swedish Hospital Discharge Register. More than 2.3 million people born from 1950-1970 were cross-linked by individual patient identification numbers, which allowed the researchers to trace not only patients but also their siblings. At the time of the analysis, no patient was younger than 40 years (JAMA Psychiatry 2013;70:22-30).
The authors tracked fecundity in about 177,000 patients who had schizophrenia, autism, bipolar disorder, anorexia nervosa, or substance abuse. Rates were compared with fecundity in 261,000 siblings. The researchers then compared these rates with those found in the general population.
About 19,000 patients had schizophrenia. They had significantly fewer children than the general population’s (fecundity rate [FR] 0.23 for men and 0.47 for women). In a univariate analysis, sisters of the patients had a significantly higher fecundity rate (FR, 1.02), but this difference disappeared once comorbidities were factored into the analysis. Brothers also had significantly decreased fecundity (FR, 0.97).
"Our results suggest a strong selection pressure to remove genetic variants associated with schizophrenia from the population," the authors said. "This is further evidence for the role of recent or de novo mutations in the genetic susceptibility to schizophrenia that has neither reached the frequency of nor existed long enough to be removed from the population."
Autism was present in 2,947 patients; these had 4,471 siblings. Fecundity was significantly lower in both men and women (FR, 0.25 and 0.48, respectively). Among the siblings, brothers also had fewer children (FR, 0.94). Among sisters, the rate was not significantly different than the general population.
"Individuals with autism showed the greatest reduction in fecundity among all examined disorders. This was not unexpected because previous investigations have shown that few individuals with autism ever married or had children.
"We propose that rare highly deleterious variants and sexually antagonistic polymorphisms may contribute to the genetic disposition to autism. The similarity to schizophrenia is notable because it has been proposed that the autistic and psychotic spectrums reflect two extremes of social cognition."
Bipolar disorder was present in 14,439 patients, among whom were 22,986 siblings. Fecundity was significantly lower than the general population in both men (FR, 0.75) and women (FR, 0.85). While brothers had similar rates to that of the general population, sisters had significantly more children (FR, 1.03). But incorporating comorbidities into the analysis changed the significance for both patients and sisters, with the patient rate increasing to just below that of the general population (FR, 0.94), and the sisters’ rate increased rate no longer being significant (FR, 0.95).
"It has been suggested that the introduction of lithium as a treatment for bipolar disorder has led to improved functioning and, as a result, greater fecundity in those populations where treatment is available."
There were 81,295 patients with depression, among who were 119,645 siblings. While men with depression had significantly lower rates (FR, 0.93), women with the disorder were not significantly different than the general population. Siblings had significantly more children than the general population (FR, brothers 1.01, sisters 1.04) – a difference that was unchanged by factoring in comorbidities. The addition of comorbidities to the analysis did not change the decreased fecundity rate for male patients, but actually increased the rate for female patients (FR, 1.03).
"Notably, depression was an exception to the five other studied disorders ... genes associated with depression seems to be maintained in the population by balancing selection because the cost to affected individuals is roughly equal to the benefit to their siblings. If this is the case, it would be the first strong evidence for balancing selection in a psychiatric disorder."
There were 3,275 patients with anorexia, who had a total of 5,172 siblings. Both men and women with the disorder had significantly reduced fecundity (0.54 and 0.58, respectively). In the sibling group, there were no significant differences for either brothers or sisters. None of the findings changed when comorbidities were factored in.
"Our calculations suggest that anorexia is under weaker negative selection relative to schizophrenia and autism," the authors said.
Substance abuse was present in 55,933 patients, who had a total of 81,592 siblings. The fecundity rate was significantly lower in both men and women (FR, 0.78 and 0.93, respectively). Siblings had significantly more children than the general population (FR, 1.03 for brothers and 1.05 for sisters).
"Our findings suggest that this increased fecundity in siblings almost entirely accounts for the cost to affected individuals, with only a slight decrease in the frequency of these individuals’ genes predicted each generation. Considering that most drugs are a new environmental exposure when seen from an evolutionary perspective, it is possible that there has been insufficient time for selection to act on risk alleles. ... It has also been suggested that substance abuse is associated with risk-taking behavior in both sexes, including sexual risk taking."
The study was funded by the Medical Research Council of the United Kingdom. One of the coauthors reported having received consulting fees and honoraria from GlaxoSmithKline and Lundbeck.
Some psychiatric disorders appear to be associated with lower fecundity in both men and women, suggesting that natural selection attempts to discourage the perpetuation of genetic variants associated with them.
Instead, new mutations could be one reason that the disorders continue to exist, Robert A. Power and his colleagues wrote in the January issue of JAMA Psychiatry (formerly Archives of General Psychiatry).
The authors also found that psychiatric disorders affected men’s fecundity more than women’s. "This sex-specific effect suggests that psychiatric morbidity impairs interest or ability to find suitable mating partners or inhibits biological fertility to a greater extent in men," wrote Mr. Power of Kings College, London.
The study data were extracted from two of Sweden’s population registries – the Multi-Generation Register and the Swedish Hospital Discharge Register. More than 2.3 million people born from 1950-1970 were cross-linked by individual patient identification numbers, which allowed the researchers to trace not only patients but also their siblings. At the time of the analysis, no patient was younger than 40 years (JAMA Psychiatry 2013;70:22-30).
The authors tracked fecundity in about 177,000 patients who had schizophrenia, autism, bipolar disorder, anorexia nervosa, or substance abuse. Rates were compared with fecundity in 261,000 siblings. The researchers then compared these rates with those found in the general population.
About 19,000 patients had schizophrenia. They had significantly fewer children than the general population’s (fecundity rate [FR] 0.23 for men and 0.47 for women). In a univariate analysis, sisters of the patients had a significantly higher fecundity rate (FR, 1.02), but this difference disappeared once comorbidities were factored into the analysis. Brothers also had significantly decreased fecundity (FR, 0.97).
"Our results suggest a strong selection pressure to remove genetic variants associated with schizophrenia from the population," the authors said. "This is further evidence for the role of recent or de novo mutations in the genetic susceptibility to schizophrenia that has neither reached the frequency of nor existed long enough to be removed from the population."
Autism was present in 2,947 patients; these had 4,471 siblings. Fecundity was significantly lower in both men and women (FR, 0.25 and 0.48, respectively). Among the siblings, brothers also had fewer children (FR, 0.94). Among sisters, the rate was not significantly different than the general population.
"Individuals with autism showed the greatest reduction in fecundity among all examined disorders. This was not unexpected because previous investigations have shown that few individuals with autism ever married or had children.
"We propose that rare highly deleterious variants and sexually antagonistic polymorphisms may contribute to the genetic disposition to autism. The similarity to schizophrenia is notable because it has been proposed that the autistic and psychotic spectrums reflect two extremes of social cognition."
Bipolar disorder was present in 14,439 patients, among whom were 22,986 siblings. Fecundity was significantly lower than the general population in both men (FR, 0.75) and women (FR, 0.85). While brothers had similar rates to that of the general population, sisters had significantly more children (FR, 1.03). But incorporating comorbidities into the analysis changed the significance for both patients and sisters, with the patient rate increasing to just below that of the general population (FR, 0.94), and the sisters’ rate increased rate no longer being significant (FR, 0.95).
"It has been suggested that the introduction of lithium as a treatment for bipolar disorder has led to improved functioning and, as a result, greater fecundity in those populations where treatment is available."
There were 81,295 patients with depression, among who were 119,645 siblings. While men with depression had significantly lower rates (FR, 0.93), women with the disorder were not significantly different than the general population. Siblings had significantly more children than the general population (FR, brothers 1.01, sisters 1.04) – a difference that was unchanged by factoring in comorbidities. The addition of comorbidities to the analysis did not change the decreased fecundity rate for male patients, but actually increased the rate for female patients (FR, 1.03).
"Notably, depression was an exception to the five other studied disorders ... genes associated with depression seems to be maintained in the population by balancing selection because the cost to affected individuals is roughly equal to the benefit to their siblings. If this is the case, it would be the first strong evidence for balancing selection in a psychiatric disorder."
There were 3,275 patients with anorexia, who had a total of 5,172 siblings. Both men and women with the disorder had significantly reduced fecundity (0.54 and 0.58, respectively). In the sibling group, there were no significant differences for either brothers or sisters. None of the findings changed when comorbidities were factored in.
"Our calculations suggest that anorexia is under weaker negative selection relative to schizophrenia and autism," the authors said.
Substance abuse was present in 55,933 patients, who had a total of 81,592 siblings. The fecundity rate was significantly lower in both men and women (FR, 0.78 and 0.93, respectively). Siblings had significantly more children than the general population (FR, 1.03 for brothers and 1.05 for sisters).
"Our findings suggest that this increased fecundity in siblings almost entirely accounts for the cost to affected individuals, with only a slight decrease in the frequency of these individuals’ genes predicted each generation. Considering that most drugs are a new environmental exposure when seen from an evolutionary perspective, it is possible that there has been insufficient time for selection to act on risk alleles. ... It has also been suggested that substance abuse is associated with risk-taking behavior in both sexes, including sexual risk taking."
The study was funded by the Medical Research Council of the United Kingdom. One of the coauthors reported having received consulting fees and honoraria from GlaxoSmithKline and Lundbeck.
Some psychiatric disorders appear to be associated with lower fecundity in both men and women, suggesting that natural selection attempts to discourage the perpetuation of genetic variants associated with them.
Instead, new mutations could be one reason that the disorders continue to exist, Robert A. Power and his colleagues wrote in the January issue of JAMA Psychiatry (formerly Archives of General Psychiatry).
The authors also found that psychiatric disorders affected men’s fecundity more than women’s. "This sex-specific effect suggests that psychiatric morbidity impairs interest or ability to find suitable mating partners or inhibits biological fertility to a greater extent in men," wrote Mr. Power of Kings College, London.
The study data were extracted from two of Sweden’s population registries – the Multi-Generation Register and the Swedish Hospital Discharge Register. More than 2.3 million people born from 1950-1970 were cross-linked by individual patient identification numbers, which allowed the researchers to trace not only patients but also their siblings. At the time of the analysis, no patient was younger than 40 years (JAMA Psychiatry 2013;70:22-30).
The authors tracked fecundity in about 177,000 patients who had schizophrenia, autism, bipolar disorder, anorexia nervosa, or substance abuse. Rates were compared with fecundity in 261,000 siblings. The researchers then compared these rates with those found in the general population.
About 19,000 patients had schizophrenia. They had significantly fewer children than the general population’s (fecundity rate [FR] 0.23 for men and 0.47 for women). In a univariate analysis, sisters of the patients had a significantly higher fecundity rate (FR, 1.02), but this difference disappeared once comorbidities were factored into the analysis. Brothers also had significantly decreased fecundity (FR, 0.97).
"Our results suggest a strong selection pressure to remove genetic variants associated with schizophrenia from the population," the authors said. "This is further evidence for the role of recent or de novo mutations in the genetic susceptibility to schizophrenia that has neither reached the frequency of nor existed long enough to be removed from the population."
Autism was present in 2,947 patients; these had 4,471 siblings. Fecundity was significantly lower in both men and women (FR, 0.25 and 0.48, respectively). Among the siblings, brothers also had fewer children (FR, 0.94). Among sisters, the rate was not significantly different than the general population.
"Individuals with autism showed the greatest reduction in fecundity among all examined disorders. This was not unexpected because previous investigations have shown that few individuals with autism ever married or had children.
"We propose that rare highly deleterious variants and sexually antagonistic polymorphisms may contribute to the genetic disposition to autism. The similarity to schizophrenia is notable because it has been proposed that the autistic and psychotic spectrums reflect two extremes of social cognition."
Bipolar disorder was present in 14,439 patients, among whom were 22,986 siblings. Fecundity was significantly lower than the general population in both men (FR, 0.75) and women (FR, 0.85). While brothers had similar rates to that of the general population, sisters had significantly more children (FR, 1.03). But incorporating comorbidities into the analysis changed the significance for both patients and sisters, with the patient rate increasing to just below that of the general population (FR, 0.94), and the sisters’ rate increased rate no longer being significant (FR, 0.95).
"It has been suggested that the introduction of lithium as a treatment for bipolar disorder has led to improved functioning and, as a result, greater fecundity in those populations where treatment is available."
There were 81,295 patients with depression, among who were 119,645 siblings. While men with depression had significantly lower rates (FR, 0.93), women with the disorder were not significantly different than the general population. Siblings had significantly more children than the general population (FR, brothers 1.01, sisters 1.04) – a difference that was unchanged by factoring in comorbidities. The addition of comorbidities to the analysis did not change the decreased fecundity rate for male patients, but actually increased the rate for female patients (FR, 1.03).
"Notably, depression was an exception to the five other studied disorders ... genes associated with depression seems to be maintained in the population by balancing selection because the cost to affected individuals is roughly equal to the benefit to their siblings. If this is the case, it would be the first strong evidence for balancing selection in a psychiatric disorder."
There were 3,275 patients with anorexia, who had a total of 5,172 siblings. Both men and women with the disorder had significantly reduced fecundity (0.54 and 0.58, respectively). In the sibling group, there were no significant differences for either brothers or sisters. None of the findings changed when comorbidities were factored in.
"Our calculations suggest that anorexia is under weaker negative selection relative to schizophrenia and autism," the authors said.
Substance abuse was present in 55,933 patients, who had a total of 81,592 siblings. The fecundity rate was significantly lower in both men and women (FR, 0.78 and 0.93, respectively). Siblings had significantly more children than the general population (FR, 1.03 for brothers and 1.05 for sisters).
"Our findings suggest that this increased fecundity in siblings almost entirely accounts for the cost to affected individuals, with only a slight decrease in the frequency of these individuals’ genes predicted each generation. Considering that most drugs are a new environmental exposure when seen from an evolutionary perspective, it is possible that there has been insufficient time for selection to act on risk alleles. ... It has also been suggested that substance abuse is associated with risk-taking behavior in both sexes, including sexual risk taking."
The study was funded by the Medical Research Council of the United Kingdom. One of the coauthors reported having received consulting fees and honoraria from GlaxoSmithKline and Lundbeck.
FROM JAMA PSYCHIATRY
Major Finding: Fecundity rates appear lower in patients with some psychiatric disorders, ranging from 25%-95% that of the general population.
Data Source: The population registry-based study included about 177,000 patients in Sweden with schizophrenia, autism, bipolar disorder, depression, anorexia, and substance abuse.
Disclosures: The study was funded by the Medical Research Council of the United Kingdom. One of the coauthors reported having received consulting fees and honoraria from GlaxoSmithKline and Lundbeck.
FDA approves bedaquiline for resistant tuberculosis
The Food and Drug Administration has granted an accelerated approval for bedaquiline – the first antituberculosis drug to target the key energy enzyme, adenosine triphosphate.
Because of its novel method of action, bedaquiline fills a previously unmet need, FDA officials said.
"Multidrug-resistant tuberculosis (MDR-TB) poses a serious health threat throughout the world, and [bedaquiline] provides much-needed treatment for patients who have don’t have other therapeutic options available," Dr. Edward Cox, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research, said in a statement. "Because the drug also carries some significant risks, doctors should make sure they use it appropriately and only in patients who don’t have other treatment options."
Bedaquiline carries a boxed warning about its potential effect on heart rhythm; it has been associated with QT-segment prolongation, which could lead to a potentially fatal dysrhythmia. According to materials submitted to the FDA Anti-Infective Drugs Advisory Committee, 26 patients taking the drug died during the two pivotal phase II studies, compared with four who took placebo. Three deaths were cardiac related, including one from hypertension, one from cardiac arrest associated with pneumonia, one from congestive heart failure, according to the materials submitted by manufacturer Janssen Therapeutics.
The two studies upon which FDA based its approval involved almost 400 patients, all of whom had pulmonary MDR-TB. The first compared a standard, four-drug regimen plus placebo to the same regimen plus 400 mg bedaquiline daily for 2 weeks, followed by 200 mg three times a week for 22 weeks.
At 24 weeks, 78% of those treated with bedaquiline had a culture conversion, compared with 58% of those on placebo, a significant difference. Treatment failure occurred in 23% of the active group and 43% of the placebo group.
The second study was an open-label trial of 233 previously treated patients who still had culture-positive MDR-TB. They received the same dosing regimen of bedaquiline combined with an individualized background regimen. At 24 weeks, sputum conversion was 80%, with negative cultures appearing in a mean of 57 days.
The accelerated approval program allows provisional approval to drugs showing a positive treatment effect for serious disease, based on data from a surrogate endpoint likely to predict clinical benefit – in this case, sputum conversion. Bedaquiline will be available while Janssen conducts confirmatory phase III trials.
Since bedaquiline seems safe and effective for a rare disease without a satisfactory alternative treatment, the FDA also granted it a fast-track designation, priority review, and an orphan drug designation.
The drug will be marketed under the trade name Sirturo.
The Food and Drug Administration has granted an accelerated approval for bedaquiline – the first antituberculosis drug to target the key energy enzyme, adenosine triphosphate.
Because of its novel method of action, bedaquiline fills a previously unmet need, FDA officials said.
"Multidrug-resistant tuberculosis (MDR-TB) poses a serious health threat throughout the world, and [bedaquiline] provides much-needed treatment for patients who have don’t have other therapeutic options available," Dr. Edward Cox, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research, said in a statement. "Because the drug also carries some significant risks, doctors should make sure they use it appropriately and only in patients who don’t have other treatment options."
Bedaquiline carries a boxed warning about its potential effect on heart rhythm; it has been associated with QT-segment prolongation, which could lead to a potentially fatal dysrhythmia. According to materials submitted to the FDA Anti-Infective Drugs Advisory Committee, 26 patients taking the drug died during the two pivotal phase II studies, compared with four who took placebo. Three deaths were cardiac related, including one from hypertension, one from cardiac arrest associated with pneumonia, one from congestive heart failure, according to the materials submitted by manufacturer Janssen Therapeutics.
The two studies upon which FDA based its approval involved almost 400 patients, all of whom had pulmonary MDR-TB. The first compared a standard, four-drug regimen plus placebo to the same regimen plus 400 mg bedaquiline daily for 2 weeks, followed by 200 mg three times a week for 22 weeks.
At 24 weeks, 78% of those treated with bedaquiline had a culture conversion, compared with 58% of those on placebo, a significant difference. Treatment failure occurred in 23% of the active group and 43% of the placebo group.
The second study was an open-label trial of 233 previously treated patients who still had culture-positive MDR-TB. They received the same dosing regimen of bedaquiline combined with an individualized background regimen. At 24 weeks, sputum conversion was 80%, with negative cultures appearing in a mean of 57 days.
The accelerated approval program allows provisional approval to drugs showing a positive treatment effect for serious disease, based on data from a surrogate endpoint likely to predict clinical benefit – in this case, sputum conversion. Bedaquiline will be available while Janssen conducts confirmatory phase III trials.
Since bedaquiline seems safe and effective for a rare disease without a satisfactory alternative treatment, the FDA also granted it a fast-track designation, priority review, and an orphan drug designation.
The drug will be marketed under the trade name Sirturo.
The Food and Drug Administration has granted an accelerated approval for bedaquiline – the first antituberculosis drug to target the key energy enzyme, adenosine triphosphate.
Because of its novel method of action, bedaquiline fills a previously unmet need, FDA officials said.
"Multidrug-resistant tuberculosis (MDR-TB) poses a serious health threat throughout the world, and [bedaquiline] provides much-needed treatment for patients who have don’t have other therapeutic options available," Dr. Edward Cox, director of the Office of Antimicrobial Products in the FDA’s Center for Drug Evaluation and Research, said in a statement. "Because the drug also carries some significant risks, doctors should make sure they use it appropriately and only in patients who don’t have other treatment options."
Bedaquiline carries a boxed warning about its potential effect on heart rhythm; it has been associated with QT-segment prolongation, which could lead to a potentially fatal dysrhythmia. According to materials submitted to the FDA Anti-Infective Drugs Advisory Committee, 26 patients taking the drug died during the two pivotal phase II studies, compared with four who took placebo. Three deaths were cardiac related, including one from hypertension, one from cardiac arrest associated with pneumonia, one from congestive heart failure, according to the materials submitted by manufacturer Janssen Therapeutics.
The two studies upon which FDA based its approval involved almost 400 patients, all of whom had pulmonary MDR-TB. The first compared a standard, four-drug regimen plus placebo to the same regimen plus 400 mg bedaquiline daily for 2 weeks, followed by 200 mg three times a week for 22 weeks.
At 24 weeks, 78% of those treated with bedaquiline had a culture conversion, compared with 58% of those on placebo, a significant difference. Treatment failure occurred in 23% of the active group and 43% of the placebo group.
The second study was an open-label trial of 233 previously treated patients who still had culture-positive MDR-TB. They received the same dosing regimen of bedaquiline combined with an individualized background regimen. At 24 weeks, sputum conversion was 80%, with negative cultures appearing in a mean of 57 days.
The accelerated approval program allows provisional approval to drugs showing a positive treatment effect for serious disease, based on data from a surrogate endpoint likely to predict clinical benefit – in this case, sputum conversion. Bedaquiline will be available while Janssen conducts confirmatory phase III trials.
Since bedaquiline seems safe and effective for a rare disease without a satisfactory alternative treatment, the FDA also granted it a fast-track designation, priority review, and an orphan drug designation.
The drug will be marketed under the trade name Sirturo.
Overweight pregnant women can safely gain under 25 pounds
Although overweight or obese women should gain no more than 25 pounds in a singleton pregnancy, there is no harm in their gaining less, as long as the fetus is growing as it should, according to the American College of Obstetricians and Gynecologists.
There is no medical need to increase weight gain just to meet the goals the Institute of Medicine recommended in 2009, ACOG said.
"For an obese pregnant woman who is gaining less weight than recommended, but who has an appropriately growing fetus, no evidence exists that encouraging increased weight to conform with the updated IOM guidelines will improve maternal or fetal outcomes," ACOG’s Committee on Obstetric Practice wrote in the January issue of Obstetrics and Gynecology (2013;121:210-2). The committee made a similar statement about overweight pregnant women.
In examining the IOM recommendations, ACOG reviewed the extant literature on maternal and fetal outcomes in overweight and obese women. The 2009 weight gain recommendations have not been universally embraced, the group said, because many clinicians feel they aren’t strict enough for overweight or obese women.
"The [recommendations] have met with controversial reactions from some physicians who believe that the weight gain targets are too high," the committee wrote. "Also, these perceived high weight gain targets do not address concerns regarding postpartum weight retention. In addition, concerns have been raised that the guidelines do not differentiate degrees of obesity, especially for morbidly obese women."
Setting weight goals for any pregnancy means balancing risks and benefits for two patients simultaneously, the committee noted. Obese mothers are more likely to develop gestational diabetes and preeclampsia. They have more cesarean births, and with those, a greater risk of wound infection and venous thromboembolism.
The infants of these mothers are more likely to have birth defects, including a doubling of the risk for neural tube defects. Stillbirth is more common, as is fetal macrosomia, a prime driver of an increased risk for cesarean section and its attendant maternal risks.
After reviewing both the IOM recommendations and the current literature, ACOG made several of its own recommendations for managing overweight or obese pregnant patients (Obstet. Gynecol. 2013;121:213-7).
In the preconceptional period:
• Include information about the risks obesity poses to both mother and infant, and encourage the patient to lose weight before becoming pregnant.
• Offer a nutritional assessment and exercise prescription.
During pregnancy:
• Assess height, weight, and body mass index at the first prenatal visit, and explain the IOM weight gain recommendations. Review them at each visit.
• Continue with nutritional counseling and exercise.
• For patients who have undergone bariatric surgery, prescribe additional iron, vitamin B12, folate, vitamin D, and calcium.
During labor and delivery:
• Get an early anesthesia consult, as both epidurals and general anesthesia are uniquely challenging in obese patients.
• For patients undergoing a cesarean, consider a higher-than-usual dose of prophylactic antibiotics to ward off wound infections.
• Assess each patient for thromboembolism potential; use pneumatic compression, unfractionated heparin, or low-molecular-weight heparin as indicated.
• Consider suture closure of the subcutaneous fat layer after a cesarean, as this could help reduce the risk of postoperative wound disruption.
Post partum:
• Encourage the patient to lose weight before attempting another pregnancy – enlist the help of a specialist.
All ACOG committee members are required to follow the college’s guidelines for relationships with the health care industry, according to the ACOG website.
Although overweight or obese women should gain no more than 25 pounds in a singleton pregnancy, there is no harm in their gaining less, as long as the fetus is growing as it should, according to the American College of Obstetricians and Gynecologists.
There is no medical need to increase weight gain just to meet the goals the Institute of Medicine recommended in 2009, ACOG said.
"For an obese pregnant woman who is gaining less weight than recommended, but who has an appropriately growing fetus, no evidence exists that encouraging increased weight to conform with the updated IOM guidelines will improve maternal or fetal outcomes," ACOG’s Committee on Obstetric Practice wrote in the January issue of Obstetrics and Gynecology (2013;121:210-2). The committee made a similar statement about overweight pregnant women.
In examining the IOM recommendations, ACOG reviewed the extant literature on maternal and fetal outcomes in overweight and obese women. The 2009 weight gain recommendations have not been universally embraced, the group said, because many clinicians feel they aren’t strict enough for overweight or obese women.
"The [recommendations] have met with controversial reactions from some physicians who believe that the weight gain targets are too high," the committee wrote. "Also, these perceived high weight gain targets do not address concerns regarding postpartum weight retention. In addition, concerns have been raised that the guidelines do not differentiate degrees of obesity, especially for morbidly obese women."
Setting weight goals for any pregnancy means balancing risks and benefits for two patients simultaneously, the committee noted. Obese mothers are more likely to develop gestational diabetes and preeclampsia. They have more cesarean births, and with those, a greater risk of wound infection and venous thromboembolism.
The infants of these mothers are more likely to have birth defects, including a doubling of the risk for neural tube defects. Stillbirth is more common, as is fetal macrosomia, a prime driver of an increased risk for cesarean section and its attendant maternal risks.
After reviewing both the IOM recommendations and the current literature, ACOG made several of its own recommendations for managing overweight or obese pregnant patients (Obstet. Gynecol. 2013;121:213-7).
In the preconceptional period:
• Include information about the risks obesity poses to both mother and infant, and encourage the patient to lose weight before becoming pregnant.
• Offer a nutritional assessment and exercise prescription.
During pregnancy:
• Assess height, weight, and body mass index at the first prenatal visit, and explain the IOM weight gain recommendations. Review them at each visit.
• Continue with nutritional counseling and exercise.
• For patients who have undergone bariatric surgery, prescribe additional iron, vitamin B12, folate, vitamin D, and calcium.
During labor and delivery:
• Get an early anesthesia consult, as both epidurals and general anesthesia are uniquely challenging in obese patients.
• For patients undergoing a cesarean, consider a higher-than-usual dose of prophylactic antibiotics to ward off wound infections.
• Assess each patient for thromboembolism potential; use pneumatic compression, unfractionated heparin, or low-molecular-weight heparin as indicated.
• Consider suture closure of the subcutaneous fat layer after a cesarean, as this could help reduce the risk of postoperative wound disruption.
Post partum:
• Encourage the patient to lose weight before attempting another pregnancy – enlist the help of a specialist.
All ACOG committee members are required to follow the college’s guidelines for relationships with the health care industry, according to the ACOG website.
Although overweight or obese women should gain no more than 25 pounds in a singleton pregnancy, there is no harm in their gaining less, as long as the fetus is growing as it should, according to the American College of Obstetricians and Gynecologists.
There is no medical need to increase weight gain just to meet the goals the Institute of Medicine recommended in 2009, ACOG said.
"For an obese pregnant woman who is gaining less weight than recommended, but who has an appropriately growing fetus, no evidence exists that encouraging increased weight to conform with the updated IOM guidelines will improve maternal or fetal outcomes," ACOG’s Committee on Obstetric Practice wrote in the January issue of Obstetrics and Gynecology (2013;121:210-2). The committee made a similar statement about overweight pregnant women.
In examining the IOM recommendations, ACOG reviewed the extant literature on maternal and fetal outcomes in overweight and obese women. The 2009 weight gain recommendations have not been universally embraced, the group said, because many clinicians feel they aren’t strict enough for overweight or obese women.
"The [recommendations] have met with controversial reactions from some physicians who believe that the weight gain targets are too high," the committee wrote. "Also, these perceived high weight gain targets do not address concerns regarding postpartum weight retention. In addition, concerns have been raised that the guidelines do not differentiate degrees of obesity, especially for morbidly obese women."
Setting weight goals for any pregnancy means balancing risks and benefits for two patients simultaneously, the committee noted. Obese mothers are more likely to develop gestational diabetes and preeclampsia. They have more cesarean births, and with those, a greater risk of wound infection and venous thromboembolism.
The infants of these mothers are more likely to have birth defects, including a doubling of the risk for neural tube defects. Stillbirth is more common, as is fetal macrosomia, a prime driver of an increased risk for cesarean section and its attendant maternal risks.
After reviewing both the IOM recommendations and the current literature, ACOG made several of its own recommendations for managing overweight or obese pregnant patients (Obstet. Gynecol. 2013;121:213-7).
In the preconceptional period:
• Include information about the risks obesity poses to both mother and infant, and encourage the patient to lose weight before becoming pregnant.
• Offer a nutritional assessment and exercise prescription.
During pregnancy:
• Assess height, weight, and body mass index at the first prenatal visit, and explain the IOM weight gain recommendations. Review them at each visit.
• Continue with nutritional counseling and exercise.
• For patients who have undergone bariatric surgery, prescribe additional iron, vitamin B12, folate, vitamin D, and calcium.
During labor and delivery:
• Get an early anesthesia consult, as both epidurals and general anesthesia are uniquely challenging in obese patients.
• For patients undergoing a cesarean, consider a higher-than-usual dose of prophylactic antibiotics to ward off wound infections.
• Assess each patient for thromboembolism potential; use pneumatic compression, unfractionated heparin, or low-molecular-weight heparin as indicated.
• Consider suture closure of the subcutaneous fat layer after a cesarean, as this could help reduce the risk of postoperative wound disruption.
Post partum:
• Encourage the patient to lose weight before attempting another pregnancy – enlist the help of a specialist.
All ACOG committee members are required to follow the college’s guidelines for relationships with the health care industry, according to the ACOG website.
FROM OBSTETRICS AND GYNECOLOGY
'Chemobrain' starts before chemotherapy in breast cancer study
SAN ANTONIO – The muddled thinking that sometimes affects breast cancer patients is manifested by decreased activity in a brain region that plays a key role in working memory, according to results of a functional imaging study.
"Chemobrain," as it’s sometimes known, appears even before chemotherapy starts, suggesting that more may be at play than a cognitive reaction to the medications, Bernadine Cimprich, Ph.D., reported at the annual San Antonio Breast Cancer Symposium.
The findings of her functional imaging study show a strong correlation between fatigue and decreased activation in the left inferior frontal gyrus.
There’s no question that chemotherapy agents can have cognitive effects, said Dr. Cimprich, an associate professor emeritus of nursing at the University of Michigan, Ann Arbor. "But even before treatment, we saw reduced function in the regions needed to perform this task."
Her prospective comparative study comprised 69 women with localized (stage 0-III) breast cancer, and 32 age-matched healthy controls. The patients were 24-34 days post surgery, but had not yet received either chemotherapy (29) or radiotherapy (37). All of the women reported their levels of fatigue.
Before and after treatment, the patients performed a verbal test of working memory while undergoing functional magnetic resonance brain imaging both before and after treatments. Each test had several difficulty levels. Patients also self-reported fatigue at both time points.
The patients were an average of 51 years old. Half of the chemotherapy group and 95% of the radiotherapy group had undergone a breast-conserving surgical procedure. The other half of the chemotherapy group had mastectomies.
The subjects performed the Verbal Working Memory Task during scanning. Following the scan, they completed the Attentional Function Index and the Functional Assessment of Cancer Therapy-Fatigue. The memory test involved three levels of difficulty, from low to high demand.
Compared with the radiotherapy and control groups, the chemotherapy group reported more fatigue at both time points, and performed significantly more poorly on the cognitive test at both time points (P less than .05). Greater fatigue in the chemotherapy group was positively associated with and correlated with poorer cognitive performance; the difference was significant in the post-treatment period (P = .03).
The radiotherapy group performed significantly better than the chemotherapy group, and significantly worse than the control group. Fatigue scores also fell between those of the chemotherapy group and the control group.
Imaging showed a positive correlation between poor cognitive performance and decreased activity in the left anterior frontal inferior gyrus. The score differences in the chemotherapy group "were mainly due to lower pretreatment activation in an area of the prefrontal cortex supporting working memory, the anatomical left inferior frontal gyrus, at the higher task demand," Dr. Cimprich said at a press briefing.
The level of inactivation in the region also significantly predicted the severity of fatigue in both treatment groups (P less than .01). The post-treatment imaging, conducted about 5 months after the baseline assessment, showed no differences in brain activation. However, those who had the lowest activation also had the highest post-treatment fatigue, she added.
"Women who were not able to activate this region suffered significantly greater fatigue after treatment, regardless of whether they received chemotherapy or radiotherapy," Dr. Cimprich said.
Dr. Cimprich had no financial disclosures. The study was supported by the National Institutes of Health and the National Institute of Nursing Research.
SAN ANTONIO – The muddled thinking that sometimes affects breast cancer patients is manifested by decreased activity in a brain region that plays a key role in working memory, according to results of a functional imaging study.
"Chemobrain," as it’s sometimes known, appears even before chemotherapy starts, suggesting that more may be at play than a cognitive reaction to the medications, Bernadine Cimprich, Ph.D., reported at the annual San Antonio Breast Cancer Symposium.
The findings of her functional imaging study show a strong correlation between fatigue and decreased activation in the left inferior frontal gyrus.
There’s no question that chemotherapy agents can have cognitive effects, said Dr. Cimprich, an associate professor emeritus of nursing at the University of Michigan, Ann Arbor. "But even before treatment, we saw reduced function in the regions needed to perform this task."
Her prospective comparative study comprised 69 women with localized (stage 0-III) breast cancer, and 32 age-matched healthy controls. The patients were 24-34 days post surgery, but had not yet received either chemotherapy (29) or radiotherapy (37). All of the women reported their levels of fatigue.
Before and after treatment, the patients performed a verbal test of working memory while undergoing functional magnetic resonance brain imaging both before and after treatments. Each test had several difficulty levels. Patients also self-reported fatigue at both time points.
The patients were an average of 51 years old. Half of the chemotherapy group and 95% of the radiotherapy group had undergone a breast-conserving surgical procedure. The other half of the chemotherapy group had mastectomies.
The subjects performed the Verbal Working Memory Task during scanning. Following the scan, they completed the Attentional Function Index and the Functional Assessment of Cancer Therapy-Fatigue. The memory test involved three levels of difficulty, from low to high demand.
Compared with the radiotherapy and control groups, the chemotherapy group reported more fatigue at both time points, and performed significantly more poorly on the cognitive test at both time points (P less than .05). Greater fatigue in the chemotherapy group was positively associated with and correlated with poorer cognitive performance; the difference was significant in the post-treatment period (P = .03).
The radiotherapy group performed significantly better than the chemotherapy group, and significantly worse than the control group. Fatigue scores also fell between those of the chemotherapy group and the control group.
Imaging showed a positive correlation between poor cognitive performance and decreased activity in the left anterior frontal inferior gyrus. The score differences in the chemotherapy group "were mainly due to lower pretreatment activation in an area of the prefrontal cortex supporting working memory, the anatomical left inferior frontal gyrus, at the higher task demand," Dr. Cimprich said at a press briefing.
The level of inactivation in the region also significantly predicted the severity of fatigue in both treatment groups (P less than .01). The post-treatment imaging, conducted about 5 months after the baseline assessment, showed no differences in brain activation. However, those who had the lowest activation also had the highest post-treatment fatigue, she added.
"Women who were not able to activate this region suffered significantly greater fatigue after treatment, regardless of whether they received chemotherapy or radiotherapy," Dr. Cimprich said.
Dr. Cimprich had no financial disclosures. The study was supported by the National Institutes of Health and the National Institute of Nursing Research.
SAN ANTONIO – The muddled thinking that sometimes affects breast cancer patients is manifested by decreased activity in a brain region that plays a key role in working memory, according to results of a functional imaging study.
"Chemobrain," as it’s sometimes known, appears even before chemotherapy starts, suggesting that more may be at play than a cognitive reaction to the medications, Bernadine Cimprich, Ph.D., reported at the annual San Antonio Breast Cancer Symposium.
The findings of her functional imaging study show a strong correlation between fatigue and decreased activation in the left inferior frontal gyrus.
There’s no question that chemotherapy agents can have cognitive effects, said Dr. Cimprich, an associate professor emeritus of nursing at the University of Michigan, Ann Arbor. "But even before treatment, we saw reduced function in the regions needed to perform this task."
Her prospective comparative study comprised 69 women with localized (stage 0-III) breast cancer, and 32 age-matched healthy controls. The patients were 24-34 days post surgery, but had not yet received either chemotherapy (29) or radiotherapy (37). All of the women reported their levels of fatigue.
Before and after treatment, the patients performed a verbal test of working memory while undergoing functional magnetic resonance brain imaging both before and after treatments. Each test had several difficulty levels. Patients also self-reported fatigue at both time points.
The patients were an average of 51 years old. Half of the chemotherapy group and 95% of the radiotherapy group had undergone a breast-conserving surgical procedure. The other half of the chemotherapy group had mastectomies.
The subjects performed the Verbal Working Memory Task during scanning. Following the scan, they completed the Attentional Function Index and the Functional Assessment of Cancer Therapy-Fatigue. The memory test involved three levels of difficulty, from low to high demand.
Compared with the radiotherapy and control groups, the chemotherapy group reported more fatigue at both time points, and performed significantly more poorly on the cognitive test at both time points (P less than .05). Greater fatigue in the chemotherapy group was positively associated with and correlated with poorer cognitive performance; the difference was significant in the post-treatment period (P = .03).
The radiotherapy group performed significantly better than the chemotherapy group, and significantly worse than the control group. Fatigue scores also fell between those of the chemotherapy group and the control group.
Imaging showed a positive correlation between poor cognitive performance and decreased activity in the left anterior frontal inferior gyrus. The score differences in the chemotherapy group "were mainly due to lower pretreatment activation in an area of the prefrontal cortex supporting working memory, the anatomical left inferior frontal gyrus, at the higher task demand," Dr. Cimprich said at a press briefing.
The level of inactivation in the region also significantly predicted the severity of fatigue in both treatment groups (P less than .01). The post-treatment imaging, conducted about 5 months after the baseline assessment, showed no differences in brain activation. However, those who had the lowest activation also had the highest post-treatment fatigue, she added.
"Women who were not able to activate this region suffered significantly greater fatigue after treatment, regardless of whether they received chemotherapy or radiotherapy," Dr. Cimprich said.
Dr. Cimprich had no financial disclosures. The study was supported by the National Institutes of Health and the National Institute of Nursing Research.
AT THE ANNUAL SAN ANTONIO BREAST CANCER SYMPOSIUM
Major Finding: Women scheduled to undergo chemotherapy after surgery for breast cancer were significantly more likely to show low brain activation in a task of working memory than other groups studied (P less than .05).
Data Source: A prospective, comparative study of 69 patients and 32 matched controls.
Disclosures: Dr. Cimprich had no financial disclosures. The study was supported by the National Institutes of Health and the National Institute of Nursing Research.