User login
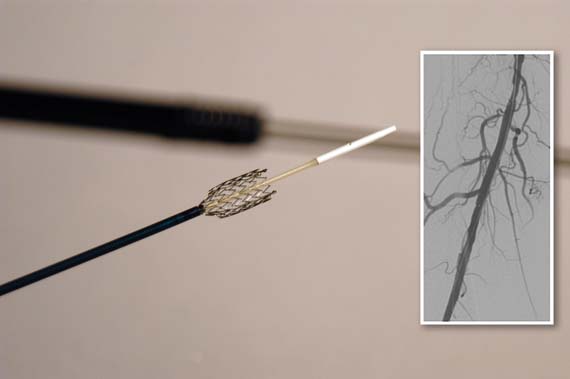
Bevacizumab plus chemo extends survival in metastatic cervical cancer
Adding bevacizumab to chemotherapy extended survival by about 4 months beyond the survival achieved with chemotherapy alone in chemotherapy-naïve patients with persistent, recurrent, or metastatic cervical cancer.
The findings seen at the interim analysis of the GOG(Gynecology Oncology Group) 240 study (Paclitaxel and Cisplatin or Topotecan With or Without Bevacizumab for Treating Patients With Stage IV-B, Recurrent, or Persistent Cervical Cancer) prompted the trial’s data safety monitoring committee to announce the study had reached its primary endpoint – a statistically significant increase in overall survival for patients given the regimen that included bevacizumab (Avastin). The interim analysis took place after 271 patients had died.
"The findings in this clinical trial are important because they are likely to change clinical practice and provide an opportunity to improve outcome in patients with recurrent cervical cancer who have previously had very limited treatment options," GOG study chair Dr. Krishnansu S. Tewari and also of University of California, Irvine Medical Center, Orange, said in a statement announcing the trial results.
The trial was designed to answer two questions: Whether topotecan in combination with paclitaxel was superior to cisplatin and paclitaxel in combination, and whether the addition of bevacizumab to either regimen improved overall survival.
The study comprised 452 patients in the United States and Spain with advanced, recurrent, or persistent cervical cancer that was not curable with standard treatment. The trial participants were randomized to four treatment arms: cisplatin 50 mg/m2 plus paclitaxel 135-175 mg/m2, with and without bevacizumab 15 mg/kg; and topotecan 0.75 mg/m2 d1-3 plus paclitaxel 175 mg/ m2 d1, with and without bevacizumab 15 mg/kg. The treatment cycles were repeated every 21 days until there was disease progression, unacceptable toxicity, or a complete response. In all, 225 received the bevacizumab-containing regimens and 227 received the standard regimens.
In an analysis conducted in 2012, it was determined that topotecan plus paclitaxel was not superior to the standard therapy of cisplatin plus paclitaxel and investigators, and patients were notified of the finding.
In the more recent analysis, those who received bevacizumab were 29% less likely to die than were those who did not get the drug – a significant difference (hazard ratio, 0.71). The median survival was 17 months vs. 13 months. The response rates were 48% for the bevacizumab regimens and 26% for the standard regimens.
Patients who received bevacizumab also experienced more grade 3 and 4 side effects, including bleeding (5% vs. 1%), thrombosis/embolism (9% vs. 2%), and gastrointestinal fistula (9% vs. 2%).
More than 12,000 women will be diagnosed with cervical cancer in the United States in 2013 and more than 4,000 women will die of the disease, according to estimates.
Although the full data have yet to be made public, the interim results alone are enough to change clinical practice.
There have been no real advances in cervical cancer since it was seen that cisplatin with radiation improves survival, and that was more than a decade ago. What we have now is evidence not just of a statistically significant difference in survival, but a clinically meaningful difference as well. Based on these results, I believe that this treatment approach will become the standard of care. These data are adequate for implementing this protocol now.
Careful patient selection will be critical when implementing bevicizumab regimens, as there was a high proportion of potentially serious toxicities. I would not use this regimen for patients who already have breathing problems, blood clots or fistulas. But for the average patient without these issues, who has good functional status, I would think the benefits far outweigh the risks. Bevicizumab does appear to add a very meaningful opportunity to achieve extended survival.
Further, a high dosage of bevacizumab was used in this trial. In lung and ovarian cancer, half this dose has been shown to be effective. I don’t know if cutting the dose would be effective in these patients, or, if it was, if that would reduce some of the toxicity. But I think this option could be explored, as bevicizumab is quite expensive, being able to halve the dose would translate into a real cost savings.
I hope third-party payers will quickly be made aware of these data, and that they will agree to pay for this regimen based on these very important results.
Dr. Maurie Markman is national director of medical oncology at the Cancer Treatment Centers of America. Dr. Markman has, in the past, served on an advisory board for Genentech but is not currently associated with the company. He was not involved with the GOG study.
Although the full data have yet to be made public, the interim results alone are enough to change clinical practice.
There have been no real advances in cervical cancer since it was seen that cisplatin with radiation improves survival, and that was more than a decade ago. What we have now is evidence not just of a statistically significant difference in survival, but a clinically meaningful difference as well. Based on these results, I believe that this treatment approach will become the standard of care. These data are adequate for implementing this protocol now.
Careful patient selection will be critical when implementing bevicizumab regimens, as there was a high proportion of potentially serious toxicities. I would not use this regimen for patients who already have breathing problems, blood clots or fistulas. But for the average patient without these issues, who has good functional status, I would think the benefits far outweigh the risks. Bevicizumab does appear to add a very meaningful opportunity to achieve extended survival.
Further, a high dosage of bevacizumab was used in this trial. In lung and ovarian cancer, half this dose has been shown to be effective. I don’t know if cutting the dose would be effective in these patients, or, if it was, if that would reduce some of the toxicity. But I think this option could be explored, as bevicizumab is quite expensive, being able to halve the dose would translate into a real cost savings.
I hope third-party payers will quickly be made aware of these data, and that they will agree to pay for this regimen based on these very important results.
Dr. Maurie Markman is national director of medical oncology at the Cancer Treatment Centers of America. Dr. Markman has, in the past, served on an advisory board for Genentech but is not currently associated with the company. He was not involved with the GOG study.
Although the full data have yet to be made public, the interim results alone are enough to change clinical practice.
There have been no real advances in cervical cancer since it was seen that cisplatin with radiation improves survival, and that was more than a decade ago. What we have now is evidence not just of a statistically significant difference in survival, but a clinically meaningful difference as well. Based on these results, I believe that this treatment approach will become the standard of care. These data are adequate for implementing this protocol now.
Careful patient selection will be critical when implementing bevicizumab regimens, as there was a high proportion of potentially serious toxicities. I would not use this regimen for patients who already have breathing problems, blood clots or fistulas. But for the average patient without these issues, who has good functional status, I would think the benefits far outweigh the risks. Bevicizumab does appear to add a very meaningful opportunity to achieve extended survival.
Further, a high dosage of bevacizumab was used in this trial. In lung and ovarian cancer, half this dose has been shown to be effective. I don’t know if cutting the dose would be effective in these patients, or, if it was, if that would reduce some of the toxicity. But I think this option could be explored, as bevicizumab is quite expensive, being able to halve the dose would translate into a real cost savings.
I hope third-party payers will quickly be made aware of these data, and that they will agree to pay for this regimen based on these very important results.
Dr. Maurie Markman is national director of medical oncology at the Cancer Treatment Centers of America. Dr. Markman has, in the past, served on an advisory board for Genentech but is not currently associated with the company. He was not involved with the GOG study.
Adding bevacizumab to chemotherapy extended survival by about 4 months beyond the survival achieved with chemotherapy alone in chemotherapy-naïve patients with persistent, recurrent, or metastatic cervical cancer.
The findings seen at the interim analysis of the GOG(Gynecology Oncology Group) 240 study (Paclitaxel and Cisplatin or Topotecan With or Without Bevacizumab for Treating Patients With Stage IV-B, Recurrent, or Persistent Cervical Cancer) prompted the trial’s data safety monitoring committee to announce the study had reached its primary endpoint – a statistically significant increase in overall survival for patients given the regimen that included bevacizumab (Avastin). The interim analysis took place after 271 patients had died.
"The findings in this clinical trial are important because they are likely to change clinical practice and provide an opportunity to improve outcome in patients with recurrent cervical cancer who have previously had very limited treatment options," GOG study chair Dr. Krishnansu S. Tewari and also of University of California, Irvine Medical Center, Orange, said in a statement announcing the trial results.
The trial was designed to answer two questions: Whether topotecan in combination with paclitaxel was superior to cisplatin and paclitaxel in combination, and whether the addition of bevacizumab to either regimen improved overall survival.
The study comprised 452 patients in the United States and Spain with advanced, recurrent, or persistent cervical cancer that was not curable with standard treatment. The trial participants were randomized to four treatment arms: cisplatin 50 mg/m2 plus paclitaxel 135-175 mg/m2, with and without bevacizumab 15 mg/kg; and topotecan 0.75 mg/m2 d1-3 plus paclitaxel 175 mg/ m2 d1, with and without bevacizumab 15 mg/kg. The treatment cycles were repeated every 21 days until there was disease progression, unacceptable toxicity, or a complete response. In all, 225 received the bevacizumab-containing regimens and 227 received the standard regimens.
In an analysis conducted in 2012, it was determined that topotecan plus paclitaxel was not superior to the standard therapy of cisplatin plus paclitaxel and investigators, and patients were notified of the finding.
In the more recent analysis, those who received bevacizumab were 29% less likely to die than were those who did not get the drug – a significant difference (hazard ratio, 0.71). The median survival was 17 months vs. 13 months. The response rates were 48% for the bevacizumab regimens and 26% for the standard regimens.
Patients who received bevacizumab also experienced more grade 3 and 4 side effects, including bleeding (5% vs. 1%), thrombosis/embolism (9% vs. 2%), and gastrointestinal fistula (9% vs. 2%).
More than 12,000 women will be diagnosed with cervical cancer in the United States in 2013 and more than 4,000 women will die of the disease, according to estimates.
Adding bevacizumab to chemotherapy extended survival by about 4 months beyond the survival achieved with chemotherapy alone in chemotherapy-naïve patients with persistent, recurrent, or metastatic cervical cancer.
The findings seen at the interim analysis of the GOG(Gynecology Oncology Group) 240 study (Paclitaxel and Cisplatin or Topotecan With or Without Bevacizumab for Treating Patients With Stage IV-B, Recurrent, or Persistent Cervical Cancer) prompted the trial’s data safety monitoring committee to announce the study had reached its primary endpoint – a statistically significant increase in overall survival for patients given the regimen that included bevacizumab (Avastin). The interim analysis took place after 271 patients had died.
"The findings in this clinical trial are important because they are likely to change clinical practice and provide an opportunity to improve outcome in patients with recurrent cervical cancer who have previously had very limited treatment options," GOG study chair Dr. Krishnansu S. Tewari and also of University of California, Irvine Medical Center, Orange, said in a statement announcing the trial results.
The trial was designed to answer two questions: Whether topotecan in combination with paclitaxel was superior to cisplatin and paclitaxel in combination, and whether the addition of bevacizumab to either regimen improved overall survival.
The study comprised 452 patients in the United States and Spain with advanced, recurrent, or persistent cervical cancer that was not curable with standard treatment. The trial participants were randomized to four treatment arms: cisplatin 50 mg/m2 plus paclitaxel 135-175 mg/m2, with and without bevacizumab 15 mg/kg; and topotecan 0.75 mg/m2 d1-3 plus paclitaxel 175 mg/ m2 d1, with and without bevacizumab 15 mg/kg. The treatment cycles were repeated every 21 days until there was disease progression, unacceptable toxicity, or a complete response. In all, 225 received the bevacizumab-containing regimens and 227 received the standard regimens.
In an analysis conducted in 2012, it was determined that topotecan plus paclitaxel was not superior to the standard therapy of cisplatin plus paclitaxel and investigators, and patients were notified of the finding.
In the more recent analysis, those who received bevacizumab were 29% less likely to die than were those who did not get the drug – a significant difference (hazard ratio, 0.71). The median survival was 17 months vs. 13 months. The response rates were 48% for the bevacizumab regimens and 26% for the standard regimens.
Patients who received bevacizumab also experienced more grade 3 and 4 side effects, including bleeding (5% vs. 1%), thrombosis/embolism (9% vs. 2%), and gastrointestinal fistula (9% vs. 2%).
More than 12,000 women will be diagnosed with cervical cancer in the United States in 2013 and more than 4,000 women will die of the disease, according to estimates.
Major Finding: Patients with recurrent, persistent, or metastatic cervical cancer who were randomized to a chemotherapy regimen including bevacizumab lived a median of 17 months and those in a regimen without bevacizumab lived a median of 13 months.
Data Source: The GOG240 trial randomized 452 patients to four treatment protocols, two of which contained bevicizumab.
Disclosures: The GOG240 study was funded by the National Cancer Institute and Genentech Inc., the maker of bevacizumab.
Job stress not linked with cancer risk
Job stress was not directly associated with the risks of lung, breast, colorectal, or prostate cancer in a large retrospective study.
"Work-related psychosocial stress is unlikely to be an important risk factor for these cancers," wrote Katriina Heikkilä, Ph.D., and her coauthors in the Feb. 7 issue of the British Medical Journal (BMJ 2013;346b:f165 [doi: 10.1136/bmj.f165]).
"Thus, though reducing work stress would undoubtedly improve the psychological and physical well-being of the working individuals as well as the working population, it is unlikely to have an important impact on cancer burden at a population level."
The study does not completely exonerate the role of stress in predisposing to cancer, said Dr. Heikkilä of the Finnish Institute of Occupational Health, Helsinki.
"For example, in a meta-analysis of different types of stress and risk of breast cancer, stress from adverse life events was consistently associated with an increased risk (Breast Cancer Res. 2011;13:208). ... In a French study, people with brain cancer were more likely to report adverse life events than controls without cancer, but there was no clear evidence for a difference in terms of stress at work between these groups (J. Neurooncol. 2011;103:307-16)."
The review included 116,056 subjects who had participated in 10 different trials conducted in Denmark, Finland, and Sweden. The mean follow-up in these studies was 12 years, but follow-up ranged from 5-23 years.
This long span is a particular strength of the review, the authors noted, "because most cancers have a latent period of years or even decades. If a true association between job strain and incident cancer existed because the physiological stress response has a role in cancer promotion or progression (for example, via the regulation of the inflammatory pathways), the follow-up periods in our analyses should have been long enough to detect such an association."
At baseline, the subjects were a mean of 38 years old. Most (62%) were of normal weight. A quarter of the subjects smoked tobacco at baseline, while 10% were classified as heavy drinkers, defined as consuming at least 15 drinks per week for women and 22 drinks per week for men.
Psychological stress at work was defined as a combination of high work demand and low control at the workplace. The results were examined as stress or no stress, and in quartiles of high-stress job (high demand/low control); active job (high demand/high control); passive job (low demand/low control); and low-strain job (low demand/high control). This was measured with a Likert-type scale and ascertained by questions taken from two validated stress questionnaires. The investigators also controlled for age, gender, socioeconomic position, body mass index, smoking, and alcohol intake.
The overall rate of cancer was 5% (5,765). The largest proportion of these cancers was breast cancer (0.9% of the study cohort; 1,010), followed by prostate cancer (0.7%; 865), colorectal cancer (0.5%; 522), and lung cancer (0.3%; 374).
In the binary analysis of job stress/no job stress, there was no significant association with overall cancer risk (hazard ratio, 0.97).
Nor were there significant associations between job stress and colorectal cancer (HR, 1.16), breast cancer (HR, 0.97), lung cancer (HR, 1.17), or prostate cancer (HR, 0.86). "There was also no clear evidence for an association between the categories of job strain and the risk of cancer," the authors wrote.
The study "suggests that many of the previously reported associations ... between work-related stress and risk of cancer could have been influenced by chance, low power in some studies, different covariate adjustment, or residual confounding from possible unmeasured common causes of work stress and cancer. Such common causes could include shift work (for which there is some evidence of an association with risk of breast cancer) or other sources of stress, perhaps combined with one another."
The study was sponsored by several research consortiums and federal agencies in Scandinavia. None of the authors had any financial disclosures.
Job stress was not directly associated with the risks of lung, breast, colorectal, or prostate cancer in a large retrospective study.
"Work-related psychosocial stress is unlikely to be an important risk factor for these cancers," wrote Katriina Heikkilä, Ph.D., and her coauthors in the Feb. 7 issue of the British Medical Journal (BMJ 2013;346b:f165 [doi: 10.1136/bmj.f165]).
"Thus, though reducing work stress would undoubtedly improve the psychological and physical well-being of the working individuals as well as the working population, it is unlikely to have an important impact on cancer burden at a population level."
The study does not completely exonerate the role of stress in predisposing to cancer, said Dr. Heikkilä of the Finnish Institute of Occupational Health, Helsinki.
"For example, in a meta-analysis of different types of stress and risk of breast cancer, stress from adverse life events was consistently associated with an increased risk (Breast Cancer Res. 2011;13:208). ... In a French study, people with brain cancer were more likely to report adverse life events than controls without cancer, but there was no clear evidence for a difference in terms of stress at work between these groups (J. Neurooncol. 2011;103:307-16)."
The review included 116,056 subjects who had participated in 10 different trials conducted in Denmark, Finland, and Sweden. The mean follow-up in these studies was 12 years, but follow-up ranged from 5-23 years.
This long span is a particular strength of the review, the authors noted, "because most cancers have a latent period of years or even decades. If a true association between job strain and incident cancer existed because the physiological stress response has a role in cancer promotion or progression (for example, via the regulation of the inflammatory pathways), the follow-up periods in our analyses should have been long enough to detect such an association."
At baseline, the subjects were a mean of 38 years old. Most (62%) were of normal weight. A quarter of the subjects smoked tobacco at baseline, while 10% were classified as heavy drinkers, defined as consuming at least 15 drinks per week for women and 22 drinks per week for men.
Psychological stress at work was defined as a combination of high work demand and low control at the workplace. The results were examined as stress or no stress, and in quartiles of high-stress job (high demand/low control); active job (high demand/high control); passive job (low demand/low control); and low-strain job (low demand/high control). This was measured with a Likert-type scale and ascertained by questions taken from two validated stress questionnaires. The investigators also controlled for age, gender, socioeconomic position, body mass index, smoking, and alcohol intake.
The overall rate of cancer was 5% (5,765). The largest proportion of these cancers was breast cancer (0.9% of the study cohort; 1,010), followed by prostate cancer (0.7%; 865), colorectal cancer (0.5%; 522), and lung cancer (0.3%; 374).
In the binary analysis of job stress/no job stress, there was no significant association with overall cancer risk (hazard ratio, 0.97).
Nor were there significant associations between job stress and colorectal cancer (HR, 1.16), breast cancer (HR, 0.97), lung cancer (HR, 1.17), or prostate cancer (HR, 0.86). "There was also no clear evidence for an association between the categories of job strain and the risk of cancer," the authors wrote.
The study "suggests that many of the previously reported associations ... between work-related stress and risk of cancer could have been influenced by chance, low power in some studies, different covariate adjustment, or residual confounding from possible unmeasured common causes of work stress and cancer. Such common causes could include shift work (for which there is some evidence of an association with risk of breast cancer) or other sources of stress, perhaps combined with one another."
The study was sponsored by several research consortiums and federal agencies in Scandinavia. None of the authors had any financial disclosures.
Job stress was not directly associated with the risks of lung, breast, colorectal, or prostate cancer in a large retrospective study.
"Work-related psychosocial stress is unlikely to be an important risk factor for these cancers," wrote Katriina Heikkilä, Ph.D., and her coauthors in the Feb. 7 issue of the British Medical Journal (BMJ 2013;346b:f165 [doi: 10.1136/bmj.f165]).
"Thus, though reducing work stress would undoubtedly improve the psychological and physical well-being of the working individuals as well as the working population, it is unlikely to have an important impact on cancer burden at a population level."
The study does not completely exonerate the role of stress in predisposing to cancer, said Dr. Heikkilä of the Finnish Institute of Occupational Health, Helsinki.
"For example, in a meta-analysis of different types of stress and risk of breast cancer, stress from adverse life events was consistently associated with an increased risk (Breast Cancer Res. 2011;13:208). ... In a French study, people with brain cancer were more likely to report adverse life events than controls without cancer, but there was no clear evidence for a difference in terms of stress at work between these groups (J. Neurooncol. 2011;103:307-16)."
The review included 116,056 subjects who had participated in 10 different trials conducted in Denmark, Finland, and Sweden. The mean follow-up in these studies was 12 years, but follow-up ranged from 5-23 years.
This long span is a particular strength of the review, the authors noted, "because most cancers have a latent period of years or even decades. If a true association between job strain and incident cancer existed because the physiological stress response has a role in cancer promotion or progression (for example, via the regulation of the inflammatory pathways), the follow-up periods in our analyses should have been long enough to detect such an association."
At baseline, the subjects were a mean of 38 years old. Most (62%) were of normal weight. A quarter of the subjects smoked tobacco at baseline, while 10% were classified as heavy drinkers, defined as consuming at least 15 drinks per week for women and 22 drinks per week for men.
Psychological stress at work was defined as a combination of high work demand and low control at the workplace. The results were examined as stress or no stress, and in quartiles of high-stress job (high demand/low control); active job (high demand/high control); passive job (low demand/low control); and low-strain job (low demand/high control). This was measured with a Likert-type scale and ascertained by questions taken from two validated stress questionnaires. The investigators also controlled for age, gender, socioeconomic position, body mass index, smoking, and alcohol intake.
The overall rate of cancer was 5% (5,765). The largest proportion of these cancers was breast cancer (0.9% of the study cohort; 1,010), followed by prostate cancer (0.7%; 865), colorectal cancer (0.5%; 522), and lung cancer (0.3%; 374).
In the binary analysis of job stress/no job stress, there was no significant association with overall cancer risk (hazard ratio, 0.97).
Nor were there significant associations between job stress and colorectal cancer (HR, 1.16), breast cancer (HR, 0.97), lung cancer (HR, 1.17), or prostate cancer (HR, 0.86). "There was also no clear evidence for an association between the categories of job strain and the risk of cancer," the authors wrote.
The study "suggests that many of the previously reported associations ... between work-related stress and risk of cancer could have been influenced by chance, low power in some studies, different covariate adjustment, or residual confounding from possible unmeasured common causes of work stress and cancer. Such common causes could include shift work (for which there is some evidence of an association with risk of breast cancer) or other sources of stress, perhaps combined with one another."
The study was sponsored by several research consortiums and federal agencies in Scandinavia. None of the authors had any financial disclosures.
FROM THE BRITISH MEDICAL JOURNAL
Major Finding: Job stress was not significantly associated with colorectal cancer (HR, 1.16), breast cancer (HR, 0.97), lung cancer (HR, 1.17), or prostate cancer (HR, 0.86).
Data Source: A retrospective cohort study of data from 10 large Scandinavian studies comprising more than 116,000 subjects.
Disclosures: The study was sponsored by several research consortiums and agencies in Scandinavia. None of the authors had any financial disclosures.
Sertraline, transcranial stimulation combination works well for depression
Transcranial direct current stimulation and sertraline together improve symptoms of depression significantly more effectively than either treatment alone.
A randomized controlled trial suggests that the treatments potentiate each other, Dr. Andre R. Brunoni and his colleagues wrote in the Feb. 6 online issue of JAMA Psychiatry (formerly Archives of General Psychiatry).
"The findings that the combined treatment was associated with a faster, greater response could indicate that each intervention has a distinct but additive mechanism of action," wrote Dr. Brunoni of the Clinical Research Center, University Hospital, São Paolo, Brazil, and his coauthors.
Transcranial direct current stimulation (tDCS) is a technique in which weak and direct electrical currents are applied to the brain through electrodes that are placed on the scalp. The antidepressant effects of tDCS "are based on the finding that the left dorsolateral prefrontal cortex is hypoactive in depression, and, therefore, anodal tDCS would be able to restore prefrontal activity by increasing activity in this area."
In light of the association between major depressive disorder and not only lateralized cortical dorsolateral prefrontal lobe dysfunction but also limbic subcortical dysfunction, Dr. Brunoni and his colleagues hypothesized that tDCS could act primarily in cortical activation, whereas selective serotonin reuptake inhibitors would act primarily on the down-regulation of limbic hyperactivity.
"In fact, a recent systematic review compared neuroimaging findings from psychological vs. pharmacological interventions, suggesting that the former were related to top-down (frontal activation) effects, whereas the latter were associated with bottom-up effects," they wrote.
The 6-week trial enrolled 120 patients with unipolar nonpsychotic major depressive disorder. They were randomized to four intervention groups: sertraline with sham tDCS, tDCS with placebo medication, placebo only (sham tDCS and placebo medication), and active tDCS with sertraline 50 mg/day. The tDCS applications consisted of ten 30-minute treatments in the first 10 days, followed by 2 additional sessions per week for the remainder of the study. The primary outcome was the Montgomery-Åsberg Depression Ratings Scale (MADRS) (JAMA Psychiatry 2013 Feb. 6 [doi: 10.1001/2013.jamapsychiatry.32]).
The combination treatment was significantly more effective than placebo, with a mean difference of 11.5 points in MADRS scores. The combination therapy also was significantly more effective than tDCS only (mean difference 5.9 points) and sertraline only (mean difference 8.5 points). No difference was found between the sertraline and tDCS only groups. These findings suggest that the two treatments exert a synergistic effect, the authors said.
The combination treatment also was associated with a quick response. At 2 weeks, there was already a significant MADRS score improvement in these patients (–48.5%), compared with placebo (–30%), sertraline only (–29%), and tDCS only (–34%).
There were five episodes of hypomania and two of clinical mania. Most of these (5) occurred in the combined treatment group.
The study was funded by the São Paulo Research Foundation. None of the investigators reported any financial disclosures.
Transcranial direct current stimulation is an experimental protocol that’s part of a new family of noninvasive neuromodulatory therapies – very exciting putative treatments for psychiatric disease, and for depression in particular.
tDCS is unique in that it is very safe; the machines are very easy to use and inexpensive to make. In fact, they run off of one 9-volt battery. This type of treatment would have a lot of practical use if it proved to be clinically useful.
This study examines the interplay between this new electrical treatment mode and the traditional neurotransmitter treatment mode. The results suggest that the value of medication and device is strengthened when they are used in combination.
This was a sizable study, which is good. It was blinded as well, which is easy to do with this device, since the only sensation is a brief tingling in the scalp that lasts only a few minutes.
One of the study’s big limitations is the low dose of sertraline used – 50 mg is considered a minimally effective dose. The drug effect size did not significantly differ from placebo, telling us that this was indeed a subtherapeutic dose. So this study is not really a fair test of whether the device works better than the medication. But combining them appeared to boost the benefit of even this low dose of antidepressant.
The recent approval of trigeminal nerve stimulation as a treatment for depression paves the way for these kinds of noninvasive device treatments. But right now, TNS is similar in effect size to antidepressant medications. What we really need are more effective and safer treatments. This study suggests that combining devices and drugs may be a way of moving toward that; it highlights the expansion of neuromodulation treatments and points toward an important new research avenue.
Dr. Sarah H. Lisanby is the Lawrence C. Katz Professor, and chair of psychiatry and behavioral sciences at Duke University, Durham, N.C. She had no financial disclosures.
Dr. Andre R. Brunoni, psychiatry, JAMA Psychiatry
Transcranial direct current stimulation is an experimental protocol that’s part of a new family of noninvasive neuromodulatory therapies – very exciting putative treatments for psychiatric disease, and for depression in particular.
tDCS is unique in that it is very safe; the machines are very easy to use and inexpensive to make. In fact, they run off of one 9-volt battery. This type of treatment would have a lot of practical use if it proved to be clinically useful.
This study examines the interplay between this new electrical treatment mode and the traditional neurotransmitter treatment mode. The results suggest that the value of medication and device is strengthened when they are used in combination.
This was a sizable study, which is good. It was blinded as well, which is easy to do with this device, since the only sensation is a brief tingling in the scalp that lasts only a few minutes.
One of the study’s big limitations is the low dose of sertraline used – 50 mg is considered a minimally effective dose. The drug effect size did not significantly differ from placebo, telling us that this was indeed a subtherapeutic dose. So this study is not really a fair test of whether the device works better than the medication. But combining them appeared to boost the benefit of even this low dose of antidepressant.
The recent approval of trigeminal nerve stimulation as a treatment for depression paves the way for these kinds of noninvasive device treatments. But right now, TNS is similar in effect size to antidepressant medications. What we really need are more effective and safer treatments. This study suggests that combining devices and drugs may be a way of moving toward that; it highlights the expansion of neuromodulation treatments and points toward an important new research avenue.
Dr. Sarah H. Lisanby is the Lawrence C. Katz Professor, and chair of psychiatry and behavioral sciences at Duke University, Durham, N.C. She had no financial disclosures.
Transcranial direct current stimulation is an experimental protocol that’s part of a new family of noninvasive neuromodulatory therapies – very exciting putative treatments for psychiatric disease, and for depression in particular.
tDCS is unique in that it is very safe; the machines are very easy to use and inexpensive to make. In fact, they run off of one 9-volt battery. This type of treatment would have a lot of practical use if it proved to be clinically useful.
This study examines the interplay between this new electrical treatment mode and the traditional neurotransmitter treatment mode. The results suggest that the value of medication and device is strengthened when they are used in combination.
This was a sizable study, which is good. It was blinded as well, which is easy to do with this device, since the only sensation is a brief tingling in the scalp that lasts only a few minutes.
One of the study’s big limitations is the low dose of sertraline used – 50 mg is considered a minimally effective dose. The drug effect size did not significantly differ from placebo, telling us that this was indeed a subtherapeutic dose. So this study is not really a fair test of whether the device works better than the medication. But combining them appeared to boost the benefit of even this low dose of antidepressant.
The recent approval of trigeminal nerve stimulation as a treatment for depression paves the way for these kinds of noninvasive device treatments. But right now, TNS is similar in effect size to antidepressant medications. What we really need are more effective and safer treatments. This study suggests that combining devices and drugs may be a way of moving toward that; it highlights the expansion of neuromodulation treatments and points toward an important new research avenue.
Dr. Sarah H. Lisanby is the Lawrence C. Katz Professor, and chair of psychiatry and behavioral sciences at Duke University, Durham, N.C. She had no financial disclosures.
Transcranial direct current stimulation and sertraline together improve symptoms of depression significantly more effectively than either treatment alone.
A randomized controlled trial suggests that the treatments potentiate each other, Dr. Andre R. Brunoni and his colleagues wrote in the Feb. 6 online issue of JAMA Psychiatry (formerly Archives of General Psychiatry).
"The findings that the combined treatment was associated with a faster, greater response could indicate that each intervention has a distinct but additive mechanism of action," wrote Dr. Brunoni of the Clinical Research Center, University Hospital, São Paolo, Brazil, and his coauthors.
Transcranial direct current stimulation (tDCS) is a technique in which weak and direct electrical currents are applied to the brain through electrodes that are placed on the scalp. The antidepressant effects of tDCS "are based on the finding that the left dorsolateral prefrontal cortex is hypoactive in depression, and, therefore, anodal tDCS would be able to restore prefrontal activity by increasing activity in this area."
In light of the association between major depressive disorder and not only lateralized cortical dorsolateral prefrontal lobe dysfunction but also limbic subcortical dysfunction, Dr. Brunoni and his colleagues hypothesized that tDCS could act primarily in cortical activation, whereas selective serotonin reuptake inhibitors would act primarily on the down-regulation of limbic hyperactivity.
"In fact, a recent systematic review compared neuroimaging findings from psychological vs. pharmacological interventions, suggesting that the former were related to top-down (frontal activation) effects, whereas the latter were associated with bottom-up effects," they wrote.
The 6-week trial enrolled 120 patients with unipolar nonpsychotic major depressive disorder. They were randomized to four intervention groups: sertraline with sham tDCS, tDCS with placebo medication, placebo only (sham tDCS and placebo medication), and active tDCS with sertraline 50 mg/day. The tDCS applications consisted of ten 30-minute treatments in the first 10 days, followed by 2 additional sessions per week for the remainder of the study. The primary outcome was the Montgomery-Åsberg Depression Ratings Scale (MADRS) (JAMA Psychiatry 2013 Feb. 6 [doi: 10.1001/2013.jamapsychiatry.32]).
The combination treatment was significantly more effective than placebo, with a mean difference of 11.5 points in MADRS scores. The combination therapy also was significantly more effective than tDCS only (mean difference 5.9 points) and sertraline only (mean difference 8.5 points). No difference was found between the sertraline and tDCS only groups. These findings suggest that the two treatments exert a synergistic effect, the authors said.
The combination treatment also was associated with a quick response. At 2 weeks, there was already a significant MADRS score improvement in these patients (–48.5%), compared with placebo (–30%), sertraline only (–29%), and tDCS only (–34%).
There were five episodes of hypomania and two of clinical mania. Most of these (5) occurred in the combined treatment group.
The study was funded by the São Paulo Research Foundation. None of the investigators reported any financial disclosures.
Transcranial direct current stimulation and sertraline together improve symptoms of depression significantly more effectively than either treatment alone.
A randomized controlled trial suggests that the treatments potentiate each other, Dr. Andre R. Brunoni and his colleagues wrote in the Feb. 6 online issue of JAMA Psychiatry (formerly Archives of General Psychiatry).
"The findings that the combined treatment was associated with a faster, greater response could indicate that each intervention has a distinct but additive mechanism of action," wrote Dr. Brunoni of the Clinical Research Center, University Hospital, São Paolo, Brazil, and his coauthors.
Transcranial direct current stimulation (tDCS) is a technique in which weak and direct electrical currents are applied to the brain through electrodes that are placed on the scalp. The antidepressant effects of tDCS "are based on the finding that the left dorsolateral prefrontal cortex is hypoactive in depression, and, therefore, anodal tDCS would be able to restore prefrontal activity by increasing activity in this area."
In light of the association between major depressive disorder and not only lateralized cortical dorsolateral prefrontal lobe dysfunction but also limbic subcortical dysfunction, Dr. Brunoni and his colleagues hypothesized that tDCS could act primarily in cortical activation, whereas selective serotonin reuptake inhibitors would act primarily on the down-regulation of limbic hyperactivity.
"In fact, a recent systematic review compared neuroimaging findings from psychological vs. pharmacological interventions, suggesting that the former were related to top-down (frontal activation) effects, whereas the latter were associated with bottom-up effects," they wrote.
The 6-week trial enrolled 120 patients with unipolar nonpsychotic major depressive disorder. They were randomized to four intervention groups: sertraline with sham tDCS, tDCS with placebo medication, placebo only (sham tDCS and placebo medication), and active tDCS with sertraline 50 mg/day. The tDCS applications consisted of ten 30-minute treatments in the first 10 days, followed by 2 additional sessions per week for the remainder of the study. The primary outcome was the Montgomery-Åsberg Depression Ratings Scale (MADRS) (JAMA Psychiatry 2013 Feb. 6 [doi: 10.1001/2013.jamapsychiatry.32]).
The combination treatment was significantly more effective than placebo, with a mean difference of 11.5 points in MADRS scores. The combination therapy also was significantly more effective than tDCS only (mean difference 5.9 points) and sertraline only (mean difference 8.5 points). No difference was found between the sertraline and tDCS only groups. These findings suggest that the two treatments exert a synergistic effect, the authors said.
The combination treatment also was associated with a quick response. At 2 weeks, there was already a significant MADRS score improvement in these patients (–48.5%), compared with placebo (–30%), sertraline only (–29%), and tDCS only (–34%).
There were five episodes of hypomania and two of clinical mania. Most of these (5) occurred in the combined treatment group.
The study was funded by the São Paulo Research Foundation. None of the investigators reported any financial disclosures.
Dr. Andre R. Brunoni, psychiatry, JAMA Psychiatry
Dr. Andre R. Brunoni, psychiatry, JAMA Psychiatry
FROM JAMA PSYCHIATRY
Major Finding: In patients with major depressive disorder, the combination of sertraline and transcranial direct current stimulation was more effective than placebo (mean difference in MADRS scores, 11.5 points; P less than .001), and proved more effective – and faster – than either treatment alone.
Data Source: A randomized controlled trial of 120 patients.
Disclosures: The study was funded by the São Paulo Research Foundation. None of the investigators reported any financial disclosures.
Multiple births from ART continue to decline, report finds
The number of multiple births from assisted reproductive technology continues to decline, with no more than 1% of women undergoing the procedures delivering triplets or higher orders of multiples in 2011.
In fact, in the highest-risk group of women aged 42 years and older, there were no higher-order multiple births at all that year, according to David Ball, Ph.D., president of the Society for Assisted Reproductive Technologies (SART).
"We are working toward an extinction of these pregnancies," especially in older mothers, Dr Ball said in an interview. "This report shows that we’re making progress."
The group has released its latest data, providing a picture of assisted reproductive technology (ART) results in 2011. Among 379 clinics, there were 154,412 ART cycles, resulting in the birth of 59,446 babies.
The continued decline in higher-order multiple births is a key finding, and one that confirms clinical improvements in a number of areas, Dr Ball said in an interview.
"Physicians who create the fertility treatment plans are getting much more sophisticated in being able to identify which plans will give the best egg quality in each individual patient. As a result, we’re starting out with good-quality eggs, which lead to good-quality embryos in the lab. There have also been improvements in the culture techniques, so we’re growing healthier embryos. And the final piece is that we’re getting better able to identify which embryo in the lab is the most likely to implant," said Dr. Ball.
And because clinics are growing better embryos, many women are choosing elective single-embryo transfer – another factor in keeping the multiple rates down.
In 2004, the first year SART recorded these numbers, 1% or fewer of women in each age group elected to have just a single embryo transferred; up to 6% of pregnancies were triplet or more.
By 2010, the single embryo transfer rate had risen to about 10% in women younger than 35 years. In 2011, the percentage of single embryo transfers was close to 12% for women aged 35 years or younger; 11%-31% of pregnancies were twins, and less than 1% were higher-order multiples.
"Single embryo transfer is being more accepted by professionals as well as patients," Dr Ball said. "But the decision always has to be balanced between maximizing the change of pregnancy and minimizing the chance of a multiple birth."
Frozen transfer rates from nondonor oocytes are also up from the 24,743 performed in 2010 – another good sign. In 2011, there were 28,166 frozen embryo transfers. The rates of live birth were similar to those in 2010, ranging from 39% for women younger than 35 years to 24% for those aged 41-42 years, and 16% for those older than 42 years.
"Hopefully, this means women and their physicians are confident they can get another good chance with frozen embryos, and this helps them become more comfortable with the idea of a single embryo transfer," Dr. Ball said.
Overall, women younger than 35 years continued to have the best results, the report indicated, including:
• 40% of cycles resulting in live births.
• 46% of transfers resulting in live births.
• 36% implantation rate.
• 31% of live births with twins.
Pregnancy results were inversely related to age. For those aged 41-42 years:
• 12% of cycles resulted in live births.
• 17% of transfers resulted in live birth.
• 9% implantation rate.
• 15% of live births with twins.
SART has also created an online registry that allows easy access to all of the outcomes data from 2004 to 2011.
It also allows patients and physicians to examine outcomes in each SART member fertility center. A "hover" feature facilitates patient understanding by providing explanations of technical terminology. Patients can search for a facility by distance by entering their zip code as well.
No disclosures were given.
David Ball, Ph.D., Society for Assisted Reproductive Technologies, SART, embryo, pregnancy
The number of multiple births from assisted reproductive technology continues to decline, with no more than 1% of women undergoing the procedures delivering triplets or higher orders of multiples in 2011.
In fact, in the highest-risk group of women aged 42 years and older, there were no higher-order multiple births at all that year, according to David Ball, Ph.D., president of the Society for Assisted Reproductive Technologies (SART).
"We are working toward an extinction of these pregnancies," especially in older mothers, Dr Ball said in an interview. "This report shows that we’re making progress."
The group has released its latest data, providing a picture of assisted reproductive technology (ART) results in 2011. Among 379 clinics, there were 154,412 ART cycles, resulting in the birth of 59,446 babies.
The continued decline in higher-order multiple births is a key finding, and one that confirms clinical improvements in a number of areas, Dr Ball said in an interview.
"Physicians who create the fertility treatment plans are getting much more sophisticated in being able to identify which plans will give the best egg quality in each individual patient. As a result, we’re starting out with good-quality eggs, which lead to good-quality embryos in the lab. There have also been improvements in the culture techniques, so we’re growing healthier embryos. And the final piece is that we’re getting better able to identify which embryo in the lab is the most likely to implant," said Dr. Ball.
And because clinics are growing better embryos, many women are choosing elective single-embryo transfer – another factor in keeping the multiple rates down.
In 2004, the first year SART recorded these numbers, 1% or fewer of women in each age group elected to have just a single embryo transferred; up to 6% of pregnancies were triplet or more.
By 2010, the single embryo transfer rate had risen to about 10% in women younger than 35 years. In 2011, the percentage of single embryo transfers was close to 12% for women aged 35 years or younger; 11%-31% of pregnancies were twins, and less than 1% were higher-order multiples.
"Single embryo transfer is being more accepted by professionals as well as patients," Dr Ball said. "But the decision always has to be balanced between maximizing the change of pregnancy and minimizing the chance of a multiple birth."
Frozen transfer rates from nondonor oocytes are also up from the 24,743 performed in 2010 – another good sign. In 2011, there were 28,166 frozen embryo transfers. The rates of live birth were similar to those in 2010, ranging from 39% for women younger than 35 years to 24% for those aged 41-42 years, and 16% for those older than 42 years.
"Hopefully, this means women and their physicians are confident they can get another good chance with frozen embryos, and this helps them become more comfortable with the idea of a single embryo transfer," Dr. Ball said.
Overall, women younger than 35 years continued to have the best results, the report indicated, including:
• 40% of cycles resulting in live births.
• 46% of transfers resulting in live births.
• 36% implantation rate.
• 31% of live births with twins.
Pregnancy results were inversely related to age. For those aged 41-42 years:
• 12% of cycles resulted in live births.
• 17% of transfers resulted in live birth.
• 9% implantation rate.
• 15% of live births with twins.
SART has also created an online registry that allows easy access to all of the outcomes data from 2004 to 2011.
It also allows patients and physicians to examine outcomes in each SART member fertility center. A "hover" feature facilitates patient understanding by providing explanations of technical terminology. Patients can search for a facility by distance by entering their zip code as well.
No disclosures were given.
The number of multiple births from assisted reproductive technology continues to decline, with no more than 1% of women undergoing the procedures delivering triplets or higher orders of multiples in 2011.
In fact, in the highest-risk group of women aged 42 years and older, there were no higher-order multiple births at all that year, according to David Ball, Ph.D., president of the Society for Assisted Reproductive Technologies (SART).
"We are working toward an extinction of these pregnancies," especially in older mothers, Dr Ball said in an interview. "This report shows that we’re making progress."
The group has released its latest data, providing a picture of assisted reproductive technology (ART) results in 2011. Among 379 clinics, there were 154,412 ART cycles, resulting in the birth of 59,446 babies.
The continued decline in higher-order multiple births is a key finding, and one that confirms clinical improvements in a number of areas, Dr Ball said in an interview.
"Physicians who create the fertility treatment plans are getting much more sophisticated in being able to identify which plans will give the best egg quality in each individual patient. As a result, we’re starting out with good-quality eggs, which lead to good-quality embryos in the lab. There have also been improvements in the culture techniques, so we’re growing healthier embryos. And the final piece is that we’re getting better able to identify which embryo in the lab is the most likely to implant," said Dr. Ball.
And because clinics are growing better embryos, many women are choosing elective single-embryo transfer – another factor in keeping the multiple rates down.
In 2004, the first year SART recorded these numbers, 1% or fewer of women in each age group elected to have just a single embryo transferred; up to 6% of pregnancies were triplet or more.
By 2010, the single embryo transfer rate had risen to about 10% in women younger than 35 years. In 2011, the percentage of single embryo transfers was close to 12% for women aged 35 years or younger; 11%-31% of pregnancies were twins, and less than 1% were higher-order multiples.
"Single embryo transfer is being more accepted by professionals as well as patients," Dr Ball said. "But the decision always has to be balanced between maximizing the change of pregnancy and minimizing the chance of a multiple birth."
Frozen transfer rates from nondonor oocytes are also up from the 24,743 performed in 2010 – another good sign. In 2011, there were 28,166 frozen embryo transfers. The rates of live birth were similar to those in 2010, ranging from 39% for women younger than 35 years to 24% for those aged 41-42 years, and 16% for those older than 42 years.
"Hopefully, this means women and their physicians are confident they can get another good chance with frozen embryos, and this helps them become more comfortable with the idea of a single embryo transfer," Dr. Ball said.
Overall, women younger than 35 years continued to have the best results, the report indicated, including:
• 40% of cycles resulting in live births.
• 46% of transfers resulting in live births.
• 36% implantation rate.
• 31% of live births with twins.
Pregnancy results were inversely related to age. For those aged 41-42 years:
• 12% of cycles resulted in live births.
• 17% of transfers resulted in live birth.
• 9% implantation rate.
• 15% of live births with twins.
SART has also created an online registry that allows easy access to all of the outcomes data from 2004 to 2011.
It also allows patients and physicians to examine outcomes in each SART member fertility center. A "hover" feature facilitates patient understanding by providing explanations of technical terminology. Patients can search for a facility by distance by entering their zip code as well.
No disclosures were given.
David Ball, Ph.D., Society for Assisted Reproductive Technologies, SART, embryo, pregnancy
David Ball, Ph.D., Society for Assisted Reproductive Technologies, SART, embryo, pregnancy
Major Finding: In the highest-risk group of women aged 42 years and older, there were no higher-order multiple births (triplets and higher) in 2011.
Data Source: Among 379 clinics, there were 154,412 ART cycles, resulting in the birth of 59,446 babies.
Disclosures: None was given.
Depressed stroke survivors at increased risk of death
Patients who become depressed after having had a stroke are four times more likely to die than those who have not had a stroke and who are not depressed.
Results of a large 21-year follow-up study stress the importance of depression screening in poststroke care, according to Dr. Amytis Towfighi, who will present her data in March at the annual meeting of the American Academy of Neurology in San Diego.
"Our research highlights the importance of screening for and treating depression in people who have experienced a stroke," said Dr. Towfighi, chair of the neurology department at the Rancho Los Amigos National Rehabilitation Center in Downey, Calif. "Given how common depression is after stroke and the potential consequences of having depression, looking for signs and symptoms and addressing them may be key."
Her study cohort comprised 10,550 people aged 25-74 years who participated in the 1971-1975 National Health and Nutrition Examination Survey. They were interviewed again in 1982-1984 and then followed through 1992. Dr. Towfighi and her colleagues examined the risk of death in four groups: Patients who had a stroke but no depression (73); those who had a stroke and depression (48); those who had neither stroke nor depression (8,138); and those who did not have a stroke but did have depression (2,291).
Subjects with both stroke and depression were about four times more likely to die than those with neither condition in the unadjusted (hazard ratio, 4.06) and adjusted (HR 3.69) analyses.
The multivariate analysis adjusted for age, gender, race, education and income levels, and marital status.
The factors linking stroke, depression, and mortality aren’t fully understood, Dr. Towfighi said in an interview. "However, there are both behavioral and physiological explanations. Behaviorally, individuals who are depressed may be less likely to live healthy lifestyles (abstaining from smoking, exercising regularly, and eating healthy). In addition, they may be less likely to take their medications and go to health care practitioners for routine follow-up. Physiologic processes such as changes in platelet function could also play a role."
In this study, depression did not seem to be simply a marker of having had a more severe stroke with lasting disability. "While some studies have revealed a link between severity of stroke and depression, others have not. A recent study also showed that transient ischemic attack is associated with depression. Since individuals with TIA do not exhibit any lasting symptoms, this would make the association between depression and stroke severity less likely," she said.
Checking on a stroke survivor’s psychological well-being can be a fast and easy step to include in stroke follow-up, Dr. Towfighi said. "There are simple tools for quickly screening for depression in a busy ambulatory care setting, including the Patient Health Questionnaires 2 and 9."
Depression is common after stroke and is treatable, she added. "It is important for patients, family members, and health care practitioners to be aware of the signs and symptoms of depression so that it can be identified and treated promptly."
Dr. Towfighi had no financial disclosures.
depression screening in poststroke care, according to Dr. Amytis Towfighi, American Academy of Neurology, neurology,
Patients who become depressed after having had a stroke are four times more likely to die than those who have not had a stroke and who are not depressed.
Results of a large 21-year follow-up study stress the importance of depression screening in poststroke care, according to Dr. Amytis Towfighi, who will present her data in March at the annual meeting of the American Academy of Neurology in San Diego.
"Our research highlights the importance of screening for and treating depression in people who have experienced a stroke," said Dr. Towfighi, chair of the neurology department at the Rancho Los Amigos National Rehabilitation Center in Downey, Calif. "Given how common depression is after stroke and the potential consequences of having depression, looking for signs and symptoms and addressing them may be key."
Her study cohort comprised 10,550 people aged 25-74 years who participated in the 1971-1975 National Health and Nutrition Examination Survey. They were interviewed again in 1982-1984 and then followed through 1992. Dr. Towfighi and her colleagues examined the risk of death in four groups: Patients who had a stroke but no depression (73); those who had a stroke and depression (48); those who had neither stroke nor depression (8,138); and those who did not have a stroke but did have depression (2,291).
Subjects with both stroke and depression were about four times more likely to die than those with neither condition in the unadjusted (hazard ratio, 4.06) and adjusted (HR 3.69) analyses.
The multivariate analysis adjusted for age, gender, race, education and income levels, and marital status.
The factors linking stroke, depression, and mortality aren’t fully understood, Dr. Towfighi said in an interview. "However, there are both behavioral and physiological explanations. Behaviorally, individuals who are depressed may be less likely to live healthy lifestyles (abstaining from smoking, exercising regularly, and eating healthy). In addition, they may be less likely to take their medications and go to health care practitioners for routine follow-up. Physiologic processes such as changes in platelet function could also play a role."
In this study, depression did not seem to be simply a marker of having had a more severe stroke with lasting disability. "While some studies have revealed a link between severity of stroke and depression, others have not. A recent study also showed that transient ischemic attack is associated with depression. Since individuals with TIA do not exhibit any lasting symptoms, this would make the association between depression and stroke severity less likely," she said.
Checking on a stroke survivor’s psychological well-being can be a fast and easy step to include in stroke follow-up, Dr. Towfighi said. "There are simple tools for quickly screening for depression in a busy ambulatory care setting, including the Patient Health Questionnaires 2 and 9."
Depression is common after stroke and is treatable, she added. "It is important for patients, family members, and health care practitioners to be aware of the signs and symptoms of depression so that it can be identified and treated promptly."
Dr. Towfighi had no financial disclosures.
Patients who become depressed after having had a stroke are four times more likely to die than those who have not had a stroke and who are not depressed.
Results of a large 21-year follow-up study stress the importance of depression screening in poststroke care, according to Dr. Amytis Towfighi, who will present her data in March at the annual meeting of the American Academy of Neurology in San Diego.
"Our research highlights the importance of screening for and treating depression in people who have experienced a stroke," said Dr. Towfighi, chair of the neurology department at the Rancho Los Amigos National Rehabilitation Center in Downey, Calif. "Given how common depression is after stroke and the potential consequences of having depression, looking for signs and symptoms and addressing them may be key."
Her study cohort comprised 10,550 people aged 25-74 years who participated in the 1971-1975 National Health and Nutrition Examination Survey. They were interviewed again in 1982-1984 and then followed through 1992. Dr. Towfighi and her colleagues examined the risk of death in four groups: Patients who had a stroke but no depression (73); those who had a stroke and depression (48); those who had neither stroke nor depression (8,138); and those who did not have a stroke but did have depression (2,291).
Subjects with both stroke and depression were about four times more likely to die than those with neither condition in the unadjusted (hazard ratio, 4.06) and adjusted (HR 3.69) analyses.
The multivariate analysis adjusted for age, gender, race, education and income levels, and marital status.
The factors linking stroke, depression, and mortality aren’t fully understood, Dr. Towfighi said in an interview. "However, there are both behavioral and physiological explanations. Behaviorally, individuals who are depressed may be less likely to live healthy lifestyles (abstaining from smoking, exercising regularly, and eating healthy). In addition, they may be less likely to take their medications and go to health care practitioners for routine follow-up. Physiologic processes such as changes in platelet function could also play a role."
In this study, depression did not seem to be simply a marker of having had a more severe stroke with lasting disability. "While some studies have revealed a link between severity of stroke and depression, others have not. A recent study also showed that transient ischemic attack is associated with depression. Since individuals with TIA do not exhibit any lasting symptoms, this would make the association between depression and stroke severity less likely," she said.
Checking on a stroke survivor’s psychological well-being can be a fast and easy step to include in stroke follow-up, Dr. Towfighi said. "There are simple tools for quickly screening for depression in a busy ambulatory care setting, including the Patient Health Questionnaires 2 and 9."
Depression is common after stroke and is treatable, she added. "It is important for patients, family members, and health care practitioners to be aware of the signs and symptoms of depression so that it can be identified and treated promptly."
Dr. Towfighi had no financial disclosures.
depression screening in poststroke care, according to Dr. Amytis Towfighi, American Academy of Neurology, neurology,
depression screening in poststroke care, according to Dr. Amytis Towfighi, American Academy of Neurology, neurology,
Major Finding: Individuals who had stroke and depression had a higher risk of dying of stroke in unadjusted (hazard ratio, 4.06) and adjusted (HR, 3.69) analyses.
Data Source: A cohort of 10,550 people who participated in the National Health and Nutrition Examination Survey I survey.
Disclosures: Dr. Towfighi had no financial disclosures.
Medicare needs more data before covering amyloid scans
BALTIMORE – A panel of Medicare advisers has refused to endorse the use of a brain scan that detects the presence of beta-amyloid plaques – a procedure experts say could play a key role in clinical management and Alzheimer’s disease drug research.
Members of MEDCAC (the Medicare Evidence Development and Coverage Advisory Committee) said they need considerably more data before they can recommend that the Centers for Medicare and Medicaid Services cover the procedure.
"We are all eager to decrease the burden of Alzheimer’s, but the real question, which the committee worked hard to answer today, is, ‘How will this test help us to do that?’ " Dr. Rita Redberg, committee chair, said in an interview. "The question we have to keep in mind is whether this test leads to better patient outcomes, or even to better diagnosis. It’s promising but at this time, we don’t have short- or long-term outcomes for the role amyloid scans can play in the diagnosis or management of people with Alzheimer’s."
The refusal was a "big disappointment" to the Alzheimer’s community, said Maria Carrillo, Ph.D., senior director of medical and scientific relations for the Alzheimer’s Association. However, she said in an interview, the association isn’t standing still on this issue.
"We will be recommending to the Centers for Medicare and Medicaid Services that they do consider coverage of beta-amyloid imaging according to the recommendations we made in the appropriate use criteria" on Jan. 28.
The criteria identified three patient groups in which the procedure could be appropriately used: patients with persistent or progressive, unexplained mild cognitive impairment; patients with an established dementia syndrome but an atypical presentation or clinical course; and patients whose dementia began at an unusually young age.
"We certainly understand that this requires development of an evidence base, and we do recommend that as well," Dr. Carrillo said. "But we think there is adequate evidence for the limited use we presented. We will be continuing the studies, but it will take a significant amount of time" to gather the follow-up data the panel requested.
The panel made its decision after hearing several hours of public comment, including statements from physicians who described patients’ experience with the procedure.
Dr. Howard M. Fillit of Mt. Sinai Medical Center, New York, used case reports to describe the impacts of both a negative and positive scan. He discussed an 80-year-old executive with a lifestyle of alcohol and sleeping pill consumption, travel, jet lag, and no exercise, who complained of memory problems. "I diagnosed amnestic MCI [mild cognitive impairment] and started him on acetylcholinesterase inhibitors."
The man had an amyloid PET scan, and "much to my surprise," it came back negative, Dr. Fillit said. Three months after cutting back on alcohol and his sleep medication, modifying travel, and increasing exercise, the patient was cognitively normal. Dr. Fillit discontinued the acetylcholinesterase inhibitors. "He remained in the business he loved, and didn’t have to resign from life."
Conversely, he said, a symptomatic female patient who could not afford the scan has had to leave her job without disability benefits. "She is now being forced to wait in limbo for the test of time."
Most committee members understood this kind of anecdotal benefit. But reimbursement approval demands data that directly links the test to patient outcomes, including improvements in disease management and quality of life.
Those data are scarce, said Dr. Steven Pearson, director of the Institute for Clinical and Economic Review. The agency recently published a report on Alzheimer’s diagnostic tests, aimed at generating the evidence necessary to determine insurance coverage. Only 1 of 15 amyloid PET studies directly examined how the scan might affect patient outcomes.
That industry-sponsored study examined how the scan would have affected clinical decision making for 229 patients evaluated for cognitive complaints, said Dr. Pearson (Alzheimer Dis. Assoc. Disord. 2013 Jan. 14 [doi:10.1097/WAD.0b013e318279d02a]). The patients were selected by memory disorder specialists. Clinicians in the study reviewed the initial symptoms and clinical findings and gave a working diagnosis and management plan both before and after a scan.
The scan resulted in a change of diagnosis for 55% of the cases, and clinicians reported an average increase of 22% in their diagnostic confidence. After seeing the results, 87% of physicians said they would change at least one thing in their clinical management plan.
Of the 86 patients initially diagnosed with probable Alzheimer’s, 12% had a negative scan, effectively ruling out the disorder. Half of these patients who were on Alzheimer’s medications then discontinued them.
In 22% of the positive-scan cases, physicians reported that they would change their recommendations on activities of daily living, like driving, working, and handling finances. For 16%, they would recommend enrolling in a clinical trial.
But, "these are records of intended management ... This is not the same as having hard data on physicians’ actual actions," Dr. Pearson pointed out.
Not all patient benefits can be quantified in a research study, said William Thies, Ph.D., medical director of the Alzheimer’s Association. Though it may seem intangible, the "value of knowing" can save patients and families time, money, and grief.
"It’s quite clear that many Alzheimer’s patients had a very prolonged period in which their diagnosis was in question. But when they look for answers, they’re given all sorts of vague diagnoses and tortured for many years until they finally learn they have Alzheimer’s. A test that helps us identify those people early can ease their anxiety, eliminate a long expensive period of diagnostic procedures, and result in profound benefits with regard to long-term disability insurance and planning for their future. They have an ability to come to some kind of closure and move on with the important issues they need to address."
But panel members said they needed more hard facts.
"I have no doubt that it detects amyloid, just as it’s intended to do," said Dr. Jeffrey W. Cozzens of the Southern Illinois University of Medicine, Carbondale. "And I’d like to see Medicare cover it for patients in clinical trials. But I need more real – not theoretical – data in order to fully support it."
None of the MEDCAC panel members, nor any other clinicians mentioned in this article, had any financial disclosures.
BALTIMORE – A panel of Medicare advisers has refused to endorse the use of a brain scan that detects the presence of beta-amyloid plaques – a procedure experts say could play a key role in clinical management and Alzheimer’s disease drug research.
Members of MEDCAC (the Medicare Evidence Development and Coverage Advisory Committee) said they need considerably more data before they can recommend that the Centers for Medicare and Medicaid Services cover the procedure.
"We are all eager to decrease the burden of Alzheimer’s, but the real question, which the committee worked hard to answer today, is, ‘How will this test help us to do that?’ " Dr. Rita Redberg, committee chair, said in an interview. "The question we have to keep in mind is whether this test leads to better patient outcomes, or even to better diagnosis. It’s promising but at this time, we don’t have short- or long-term outcomes for the role amyloid scans can play in the diagnosis or management of people with Alzheimer’s."
The refusal was a "big disappointment" to the Alzheimer’s community, said Maria Carrillo, Ph.D., senior director of medical and scientific relations for the Alzheimer’s Association. However, she said in an interview, the association isn’t standing still on this issue.
"We will be recommending to the Centers for Medicare and Medicaid Services that they do consider coverage of beta-amyloid imaging according to the recommendations we made in the appropriate use criteria" on Jan. 28.
The criteria identified three patient groups in which the procedure could be appropriately used: patients with persistent or progressive, unexplained mild cognitive impairment; patients with an established dementia syndrome but an atypical presentation or clinical course; and patients whose dementia began at an unusually young age.
"We certainly understand that this requires development of an evidence base, and we do recommend that as well," Dr. Carrillo said. "But we think there is adequate evidence for the limited use we presented. We will be continuing the studies, but it will take a significant amount of time" to gather the follow-up data the panel requested.
The panel made its decision after hearing several hours of public comment, including statements from physicians who described patients’ experience with the procedure.
Dr. Howard M. Fillit of Mt. Sinai Medical Center, New York, used case reports to describe the impacts of both a negative and positive scan. He discussed an 80-year-old executive with a lifestyle of alcohol and sleeping pill consumption, travel, jet lag, and no exercise, who complained of memory problems. "I diagnosed amnestic MCI [mild cognitive impairment] and started him on acetylcholinesterase inhibitors."
The man had an amyloid PET scan, and "much to my surprise," it came back negative, Dr. Fillit said. Three months after cutting back on alcohol and his sleep medication, modifying travel, and increasing exercise, the patient was cognitively normal. Dr. Fillit discontinued the acetylcholinesterase inhibitors. "He remained in the business he loved, and didn’t have to resign from life."
Conversely, he said, a symptomatic female patient who could not afford the scan has had to leave her job without disability benefits. "She is now being forced to wait in limbo for the test of time."
Most committee members understood this kind of anecdotal benefit. But reimbursement approval demands data that directly links the test to patient outcomes, including improvements in disease management and quality of life.
Those data are scarce, said Dr. Steven Pearson, director of the Institute for Clinical and Economic Review. The agency recently published a report on Alzheimer’s diagnostic tests, aimed at generating the evidence necessary to determine insurance coverage. Only 1 of 15 amyloid PET studies directly examined how the scan might affect patient outcomes.
That industry-sponsored study examined how the scan would have affected clinical decision making for 229 patients evaluated for cognitive complaints, said Dr. Pearson (Alzheimer Dis. Assoc. Disord. 2013 Jan. 14 [doi:10.1097/WAD.0b013e318279d02a]). The patients were selected by memory disorder specialists. Clinicians in the study reviewed the initial symptoms and clinical findings and gave a working diagnosis and management plan both before and after a scan.
The scan resulted in a change of diagnosis for 55% of the cases, and clinicians reported an average increase of 22% in their diagnostic confidence. After seeing the results, 87% of physicians said they would change at least one thing in their clinical management plan.
Of the 86 patients initially diagnosed with probable Alzheimer’s, 12% had a negative scan, effectively ruling out the disorder. Half of these patients who were on Alzheimer’s medications then discontinued them.
In 22% of the positive-scan cases, physicians reported that they would change their recommendations on activities of daily living, like driving, working, and handling finances. For 16%, they would recommend enrolling in a clinical trial.
But, "these are records of intended management ... This is not the same as having hard data on physicians’ actual actions," Dr. Pearson pointed out.
Not all patient benefits can be quantified in a research study, said William Thies, Ph.D., medical director of the Alzheimer’s Association. Though it may seem intangible, the "value of knowing" can save patients and families time, money, and grief.
"It’s quite clear that many Alzheimer’s patients had a very prolonged period in which their diagnosis was in question. But when they look for answers, they’re given all sorts of vague diagnoses and tortured for many years until they finally learn they have Alzheimer’s. A test that helps us identify those people early can ease their anxiety, eliminate a long expensive period of diagnostic procedures, and result in profound benefits with regard to long-term disability insurance and planning for their future. They have an ability to come to some kind of closure and move on with the important issues they need to address."
But panel members said they needed more hard facts.
"I have no doubt that it detects amyloid, just as it’s intended to do," said Dr. Jeffrey W. Cozzens of the Southern Illinois University of Medicine, Carbondale. "And I’d like to see Medicare cover it for patients in clinical trials. But I need more real – not theoretical – data in order to fully support it."
None of the MEDCAC panel members, nor any other clinicians mentioned in this article, had any financial disclosures.
BALTIMORE – A panel of Medicare advisers has refused to endorse the use of a brain scan that detects the presence of beta-amyloid plaques – a procedure experts say could play a key role in clinical management and Alzheimer’s disease drug research.
Members of MEDCAC (the Medicare Evidence Development and Coverage Advisory Committee) said they need considerably more data before they can recommend that the Centers for Medicare and Medicaid Services cover the procedure.
"We are all eager to decrease the burden of Alzheimer’s, but the real question, which the committee worked hard to answer today, is, ‘How will this test help us to do that?’ " Dr. Rita Redberg, committee chair, said in an interview. "The question we have to keep in mind is whether this test leads to better patient outcomes, or even to better diagnosis. It’s promising but at this time, we don’t have short- or long-term outcomes for the role amyloid scans can play in the diagnosis or management of people with Alzheimer’s."
The refusal was a "big disappointment" to the Alzheimer’s community, said Maria Carrillo, Ph.D., senior director of medical and scientific relations for the Alzheimer’s Association. However, she said in an interview, the association isn’t standing still on this issue.
"We will be recommending to the Centers for Medicare and Medicaid Services that they do consider coverage of beta-amyloid imaging according to the recommendations we made in the appropriate use criteria" on Jan. 28.
The criteria identified three patient groups in which the procedure could be appropriately used: patients with persistent or progressive, unexplained mild cognitive impairment; patients with an established dementia syndrome but an atypical presentation or clinical course; and patients whose dementia began at an unusually young age.
"We certainly understand that this requires development of an evidence base, and we do recommend that as well," Dr. Carrillo said. "But we think there is adequate evidence for the limited use we presented. We will be continuing the studies, but it will take a significant amount of time" to gather the follow-up data the panel requested.
The panel made its decision after hearing several hours of public comment, including statements from physicians who described patients’ experience with the procedure.
Dr. Howard M. Fillit of Mt. Sinai Medical Center, New York, used case reports to describe the impacts of both a negative and positive scan. He discussed an 80-year-old executive with a lifestyle of alcohol and sleeping pill consumption, travel, jet lag, and no exercise, who complained of memory problems. "I diagnosed amnestic MCI [mild cognitive impairment] and started him on acetylcholinesterase inhibitors."
The man had an amyloid PET scan, and "much to my surprise," it came back negative, Dr. Fillit said. Three months after cutting back on alcohol and his sleep medication, modifying travel, and increasing exercise, the patient was cognitively normal. Dr. Fillit discontinued the acetylcholinesterase inhibitors. "He remained in the business he loved, and didn’t have to resign from life."
Conversely, he said, a symptomatic female patient who could not afford the scan has had to leave her job without disability benefits. "She is now being forced to wait in limbo for the test of time."
Most committee members understood this kind of anecdotal benefit. But reimbursement approval demands data that directly links the test to patient outcomes, including improvements in disease management and quality of life.
Those data are scarce, said Dr. Steven Pearson, director of the Institute for Clinical and Economic Review. The agency recently published a report on Alzheimer’s diagnostic tests, aimed at generating the evidence necessary to determine insurance coverage. Only 1 of 15 amyloid PET studies directly examined how the scan might affect patient outcomes.
That industry-sponsored study examined how the scan would have affected clinical decision making for 229 patients evaluated for cognitive complaints, said Dr. Pearson (Alzheimer Dis. Assoc. Disord. 2013 Jan. 14 [doi:10.1097/WAD.0b013e318279d02a]). The patients were selected by memory disorder specialists. Clinicians in the study reviewed the initial symptoms and clinical findings and gave a working diagnosis and management plan both before and after a scan.
The scan resulted in a change of diagnosis for 55% of the cases, and clinicians reported an average increase of 22% in their diagnostic confidence. After seeing the results, 87% of physicians said they would change at least one thing in their clinical management plan.
Of the 86 patients initially diagnosed with probable Alzheimer’s, 12% had a negative scan, effectively ruling out the disorder. Half of these patients who were on Alzheimer’s medications then discontinued them.
In 22% of the positive-scan cases, physicians reported that they would change their recommendations on activities of daily living, like driving, working, and handling finances. For 16%, they would recommend enrolling in a clinical trial.
But, "these are records of intended management ... This is not the same as having hard data on physicians’ actual actions," Dr. Pearson pointed out.
Not all patient benefits can be quantified in a research study, said William Thies, Ph.D., medical director of the Alzheimer’s Association. Though it may seem intangible, the "value of knowing" can save patients and families time, money, and grief.
"It’s quite clear that many Alzheimer’s patients had a very prolonged period in which their diagnosis was in question. But when they look for answers, they’re given all sorts of vague diagnoses and tortured for many years until they finally learn they have Alzheimer’s. A test that helps us identify those people early can ease their anxiety, eliminate a long expensive period of diagnostic procedures, and result in profound benefits with regard to long-term disability insurance and planning for their future. They have an ability to come to some kind of closure and move on with the important issues they need to address."
But panel members said they needed more hard facts.
"I have no doubt that it detects amyloid, just as it’s intended to do," said Dr. Jeffrey W. Cozzens of the Southern Illinois University of Medicine, Carbondale. "And I’d like to see Medicare cover it for patients in clinical trials. But I need more real – not theoretical – data in order to fully support it."
None of the MEDCAC panel members, nor any other clinicians mentioned in this article, had any financial disclosures.
AT A MEETING OF MEDCAC
Amyloid PET imaging criteria for Alzheimer's outlined in report
New appropriate-use criteria for PET imaging with beta-amyloid agents stress that a positive scan cannot on its own indicate a diagnosis of Alzheimer’s disease. But in conjunction with a thorough clinical evaluation – including cognitive testing – the scan can add some degree of certainty to ruling Alzheimer’s in or out, according to the Amyloid Imaging Task Force of the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging.
Once a patient’s amyloid status is known, a patient and physician can focus on appropriate care. That can mean getting on the right medications, pursuing other testing, and even effectively planning a patient’s future, task force members Dr. Keith A. Johnson of Harvard Medical School, Boston, and his coauthors wrote in the paper establishing the criteria, which was published online Jan. 28 in the Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association and in the Journal of Nuclear Medicine (doi:10.1016/j.jalz.2013.01.002).
"The ultimate goal was to determine whether there is evidence that using amyloid PET leads to clinically meaningful improvement in outcomes or is useful in medical or personal decision making," the task force wrote.
When used appropriately, amyloid plaque PET imaging benefits both patients and physicians, whether the results are positive or negative, said Dr. Richard J. Caselli, a professor of neurology at the Mayo Clinic, Scottsdale, Ariz.
"This represents an important step forward," in Alzheimer’s clinical management," Dr. Caselli said in an interview. "Now it is a question of convincing third-party payers, including not only CMS [Centers for Medicare and Medicaid Services] but private insurers as well, of its necessity in appropriately selected patients."
CMS is convening its Medicare Evidence Development & Coverage Advisory Committee on Jan. 30 to vote on whether there is adequate evidence to determine if PET imaging of brain beta-amyloid changes health outcomes.
The task force consisted of experts in Alzheimer’s dementia and nuclear imaging. The team reviewed 80 studies that examined amyloid imaging, its diagnostic accuracy, ability to predict disease, potential implications on treatment planning, and even its possible impact on the psychological, financial, and social well-being of the patient and family.
The task force detailed three situations that warrant testing and – just as importantly – seven in which it is not indicated.
Clinical situations in which to test
The first indication is testing a patient who has persistent or progressive unexplained mild cognitive impairment. A positive finding would increase the certainty of an early Alzheimer’s pathology. A negative finding could exclude Alzheimer’s pathology at that point in time, so that the clinical investigation could consider other possible causes.
"Patients with unusually slowly progressive MCI are not common, but nonetheless pose significant diagnostic challenges that result in repeated testing over the years," said Dr. Caselli, who was not a member of the task force. "A supportive amyloid scan in such patients could reduce such duplicative testing."
The second indication would be testing a patient who satisfied the core clinical criteria for possible Alzheimer’s, but had an atypical presentation. This would include patients with an unusual course, such as sudden onset or episodic cognitive problems, or a comorbid condition that could confuse the clinical picture, like cerebrovascular disease. The indication notes that amyloid imaging can’t differentiate between Alzheimer’s disease and Lewy body pathology.
"Certainly, this may be another diagnostic asset in the evaluation of patients with unusual clinical features, but it should be recognized that Alzheimer’s disease itself has documented variants including visual, aphasic, and dysexecutive – all of which are recognized in the new diagnostic criteria – and apraxic, which was omitted in the new diagnostic criteria," said Dr. Caselli, who serves as associate director and clinical core director of the Alzheimer’s Disease Center at the Mayo Clinic in Arizona. "The decision to utilize amyloid PET in such cases will depend on the clinician’s own level of comfort but may be less important for those with more experience in these atypical presentations. What it should not do is prevent the appropriate reassessment of patients with an established diagnosis and course whose symptoms suddenly change [because of] a potential new disease process such as subdural hematoma or stroke."
The third indication for an amyloid PET scan is to determine pathology in patients with young-onset dementia – usually someone in the age range of 50-65 years, or sometimes even younger. For these patients, more detailed pathologic information is critical to decision making around issues like work, driving, lifestyle, and long-term planning.
"The presence or absence of AD [Alzheimer’s disease] pathology in this circumstance is frequently a critical component of the initial differential diagnosis," the document noted, "and it is well known from postmortem studies that clinical diagnosis based on history and examination is often wrong with regard to the presence of AD pathology."
This is where amyloid imaging could be the most clinically helpful, Dr. Caselli said.
"In my opinion, the use of amyloid PET will be most important for this indication. Younger patients are employed, have families they are supporting, and usually present early in their course when psychiatric pathology may be suspected. Imagine the 52-year-old who receives a poor job review and states he is unable to understand the new software or recall a client’s name, and seeks medical attention with the implication of disability hanging in the balance. Such a patient will not appear physically ill and is not generally suspected to have Alzheimer’s disease, but when current clinical methods indicate mild cognitive impairment, an amyloid PET scan could solidify the organicity of the diagnosis."
Clinical situations in which not to test
The document’s seven contraindications spell out scenarios in which amyloid imaging is unhelpful. These include:
• Patients who meet both the clinical criteria for Alzheimer’s and the typical age of onset. The potential benefit of detecting amyloid pathology probably would not alter patient management.
• Determining dementia severity. There’s no indication that amyloid burden correlates with symptom severity.
• Relying on reasons based solely on family history or positive apolipoprotein E–epsilon 4 status.
• Investigating a clinically unconfirmed cognitive complaint.
• Using this imaging instead of genotyping patients who have a suspected autosomal mutation.
• Making disease predictions in cognitively normal older adults
• Testing for any nonmedical use, such as for medicolegal, disability, insurance, or employment-related issues.
Patients who undergo the imaging procedure need to understand its benefits and its limitations, the task force said. Amyloid plaques don’t always mean Alzheimer’s disease is present. For example, as age increases, so does the likelihood of amyloid deposition, even in cognitively normal people. "Another major caveat is that a positive amyloid scan can also be seen in not only AD, but also in other medical conditions," such as Lewy body dementia and amyloid angiopathy, the document noted. The scan can’t differentiate these, and it is not useful when evaluating patients with frontotemporal dementia.
But amyloid imaging does confer a number of benefits when used appropriately, according to the task force. Whether the result is positive or negative, it furthers clinical decision-making in appropriately selected patients. It can guide medication management by getting patients on the right drugs faster and keeping them off those that won’t help. The results can shape other diagnostic investigations in the same way by letting patients avoid unnecessary tests or proceed to more informative ones.
There is much benefit in "the value of knowing" amyloid PET scan status, the document noted. About 25% of dementia cases can have reversible causes, like depression or vitamin B12 deficiency. And U.S. surveys have shown that most Americans would prefer to know if their symptoms could be caused by Alzheimer’s so that they can make independent decisions about the future.
But "what such polls often fail to mention are the number of people who would suffer psychological harm if they received ‘bad news’ from such testing," Dr. Caselli cautioned. "In another poll we have been conducting, more than 10% of patients stated they would ‘seriously consider suicide’ if they found there was biomarker evidence of preclinical Alzheimer’s disease. This is a powerful diagnostic modality, but one in which implications of results are not fully understood. What we offer our patients therefore must be limited to the scenarios the committee has described, at least until we have much more experience and understanding of the implications and reliability of test results."
Dr. Johnson and some of the other task force members reported multiple relationships with drug companies. Dr. Caselli has no financial disclosures.
New appropriate-use criteria for PET imaging with beta-amyloid agents stress that a positive scan cannot on its own indicate a diagnosis of Alzheimer’s disease. But in conjunction with a thorough clinical evaluation – including cognitive testing – the scan can add some degree of certainty to ruling Alzheimer’s in or out, according to the Amyloid Imaging Task Force of the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging.
Once a patient’s amyloid status is known, a patient and physician can focus on appropriate care. That can mean getting on the right medications, pursuing other testing, and even effectively planning a patient’s future, task force members Dr. Keith A. Johnson of Harvard Medical School, Boston, and his coauthors wrote in the paper establishing the criteria, which was published online Jan. 28 in the Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association and in the Journal of Nuclear Medicine (doi:10.1016/j.jalz.2013.01.002).
"The ultimate goal was to determine whether there is evidence that using amyloid PET leads to clinically meaningful improvement in outcomes or is useful in medical or personal decision making," the task force wrote.
When used appropriately, amyloid plaque PET imaging benefits both patients and physicians, whether the results are positive or negative, said Dr. Richard J. Caselli, a professor of neurology at the Mayo Clinic, Scottsdale, Ariz.
"This represents an important step forward," in Alzheimer’s clinical management," Dr. Caselli said in an interview. "Now it is a question of convincing third-party payers, including not only CMS [Centers for Medicare and Medicaid Services] but private insurers as well, of its necessity in appropriately selected patients."
CMS is convening its Medicare Evidence Development & Coverage Advisory Committee on Jan. 30 to vote on whether there is adequate evidence to determine if PET imaging of brain beta-amyloid changes health outcomes.
The task force consisted of experts in Alzheimer’s dementia and nuclear imaging. The team reviewed 80 studies that examined amyloid imaging, its diagnostic accuracy, ability to predict disease, potential implications on treatment planning, and even its possible impact on the psychological, financial, and social well-being of the patient and family.
The task force detailed three situations that warrant testing and – just as importantly – seven in which it is not indicated.
Clinical situations in which to test
The first indication is testing a patient who has persistent or progressive unexplained mild cognitive impairment. A positive finding would increase the certainty of an early Alzheimer’s pathology. A negative finding could exclude Alzheimer’s pathology at that point in time, so that the clinical investigation could consider other possible causes.
"Patients with unusually slowly progressive MCI are not common, but nonetheless pose significant diagnostic challenges that result in repeated testing over the years," said Dr. Caselli, who was not a member of the task force. "A supportive amyloid scan in such patients could reduce such duplicative testing."
The second indication would be testing a patient who satisfied the core clinical criteria for possible Alzheimer’s, but had an atypical presentation. This would include patients with an unusual course, such as sudden onset or episodic cognitive problems, or a comorbid condition that could confuse the clinical picture, like cerebrovascular disease. The indication notes that amyloid imaging can’t differentiate between Alzheimer’s disease and Lewy body pathology.
"Certainly, this may be another diagnostic asset in the evaluation of patients with unusual clinical features, but it should be recognized that Alzheimer’s disease itself has documented variants including visual, aphasic, and dysexecutive – all of which are recognized in the new diagnostic criteria – and apraxic, which was omitted in the new diagnostic criteria," said Dr. Caselli, who serves as associate director and clinical core director of the Alzheimer’s Disease Center at the Mayo Clinic in Arizona. "The decision to utilize amyloid PET in such cases will depend on the clinician’s own level of comfort but may be less important for those with more experience in these atypical presentations. What it should not do is prevent the appropriate reassessment of patients with an established diagnosis and course whose symptoms suddenly change [because of] a potential new disease process such as subdural hematoma or stroke."
The third indication for an amyloid PET scan is to determine pathology in patients with young-onset dementia – usually someone in the age range of 50-65 years, or sometimes even younger. For these patients, more detailed pathologic information is critical to decision making around issues like work, driving, lifestyle, and long-term planning.
"The presence or absence of AD [Alzheimer’s disease] pathology in this circumstance is frequently a critical component of the initial differential diagnosis," the document noted, "and it is well known from postmortem studies that clinical diagnosis based on history and examination is often wrong with regard to the presence of AD pathology."
This is where amyloid imaging could be the most clinically helpful, Dr. Caselli said.
"In my opinion, the use of amyloid PET will be most important for this indication. Younger patients are employed, have families they are supporting, and usually present early in their course when psychiatric pathology may be suspected. Imagine the 52-year-old who receives a poor job review and states he is unable to understand the new software or recall a client’s name, and seeks medical attention with the implication of disability hanging in the balance. Such a patient will not appear physically ill and is not generally suspected to have Alzheimer’s disease, but when current clinical methods indicate mild cognitive impairment, an amyloid PET scan could solidify the organicity of the diagnosis."
Clinical situations in which not to test
The document’s seven contraindications spell out scenarios in which amyloid imaging is unhelpful. These include:
• Patients who meet both the clinical criteria for Alzheimer’s and the typical age of onset. The potential benefit of detecting amyloid pathology probably would not alter patient management.
• Determining dementia severity. There’s no indication that amyloid burden correlates with symptom severity.
• Relying on reasons based solely on family history or positive apolipoprotein E–epsilon 4 status.
• Investigating a clinically unconfirmed cognitive complaint.
• Using this imaging instead of genotyping patients who have a suspected autosomal mutation.
• Making disease predictions in cognitively normal older adults
• Testing for any nonmedical use, such as for medicolegal, disability, insurance, or employment-related issues.
Patients who undergo the imaging procedure need to understand its benefits and its limitations, the task force said. Amyloid plaques don’t always mean Alzheimer’s disease is present. For example, as age increases, so does the likelihood of amyloid deposition, even in cognitively normal people. "Another major caveat is that a positive amyloid scan can also be seen in not only AD, but also in other medical conditions," such as Lewy body dementia and amyloid angiopathy, the document noted. The scan can’t differentiate these, and it is not useful when evaluating patients with frontotemporal dementia.
But amyloid imaging does confer a number of benefits when used appropriately, according to the task force. Whether the result is positive or negative, it furthers clinical decision-making in appropriately selected patients. It can guide medication management by getting patients on the right drugs faster and keeping them off those that won’t help. The results can shape other diagnostic investigations in the same way by letting patients avoid unnecessary tests or proceed to more informative ones.
There is much benefit in "the value of knowing" amyloid PET scan status, the document noted. About 25% of dementia cases can have reversible causes, like depression or vitamin B12 deficiency. And U.S. surveys have shown that most Americans would prefer to know if their symptoms could be caused by Alzheimer’s so that they can make independent decisions about the future.
But "what such polls often fail to mention are the number of people who would suffer psychological harm if they received ‘bad news’ from such testing," Dr. Caselli cautioned. "In another poll we have been conducting, more than 10% of patients stated they would ‘seriously consider suicide’ if they found there was biomarker evidence of preclinical Alzheimer’s disease. This is a powerful diagnostic modality, but one in which implications of results are not fully understood. What we offer our patients therefore must be limited to the scenarios the committee has described, at least until we have much more experience and understanding of the implications and reliability of test results."
Dr. Johnson and some of the other task force members reported multiple relationships with drug companies. Dr. Caselli has no financial disclosures.
New appropriate-use criteria for PET imaging with beta-amyloid agents stress that a positive scan cannot on its own indicate a diagnosis of Alzheimer’s disease. But in conjunction with a thorough clinical evaluation – including cognitive testing – the scan can add some degree of certainty to ruling Alzheimer’s in or out, according to the Amyloid Imaging Task Force of the Alzheimer’s Association and the Society of Nuclear Medicine and Molecular Imaging.
Once a patient’s amyloid status is known, a patient and physician can focus on appropriate care. That can mean getting on the right medications, pursuing other testing, and even effectively planning a patient’s future, task force members Dr. Keith A. Johnson of Harvard Medical School, Boston, and his coauthors wrote in the paper establishing the criteria, which was published online Jan. 28 in the Alzheimer’s & Dementia: The Journal of the Alzheimer’s Association and in the Journal of Nuclear Medicine (doi:10.1016/j.jalz.2013.01.002).
"The ultimate goal was to determine whether there is evidence that using amyloid PET leads to clinically meaningful improvement in outcomes or is useful in medical or personal decision making," the task force wrote.
When used appropriately, amyloid plaque PET imaging benefits both patients and physicians, whether the results are positive or negative, said Dr. Richard J. Caselli, a professor of neurology at the Mayo Clinic, Scottsdale, Ariz.
"This represents an important step forward," in Alzheimer’s clinical management," Dr. Caselli said in an interview. "Now it is a question of convincing third-party payers, including not only CMS [Centers for Medicare and Medicaid Services] but private insurers as well, of its necessity in appropriately selected patients."
CMS is convening its Medicare Evidence Development & Coverage Advisory Committee on Jan. 30 to vote on whether there is adequate evidence to determine if PET imaging of brain beta-amyloid changes health outcomes.
The task force consisted of experts in Alzheimer’s dementia and nuclear imaging. The team reviewed 80 studies that examined amyloid imaging, its diagnostic accuracy, ability to predict disease, potential implications on treatment planning, and even its possible impact on the psychological, financial, and social well-being of the patient and family.
The task force detailed three situations that warrant testing and – just as importantly – seven in which it is not indicated.
Clinical situations in which to test
The first indication is testing a patient who has persistent or progressive unexplained mild cognitive impairment. A positive finding would increase the certainty of an early Alzheimer’s pathology. A negative finding could exclude Alzheimer’s pathology at that point in time, so that the clinical investigation could consider other possible causes.
"Patients with unusually slowly progressive MCI are not common, but nonetheless pose significant diagnostic challenges that result in repeated testing over the years," said Dr. Caselli, who was not a member of the task force. "A supportive amyloid scan in such patients could reduce such duplicative testing."
The second indication would be testing a patient who satisfied the core clinical criteria for possible Alzheimer’s, but had an atypical presentation. This would include patients with an unusual course, such as sudden onset or episodic cognitive problems, or a comorbid condition that could confuse the clinical picture, like cerebrovascular disease. The indication notes that amyloid imaging can’t differentiate between Alzheimer’s disease and Lewy body pathology.
"Certainly, this may be another diagnostic asset in the evaluation of patients with unusual clinical features, but it should be recognized that Alzheimer’s disease itself has documented variants including visual, aphasic, and dysexecutive – all of which are recognized in the new diagnostic criteria – and apraxic, which was omitted in the new diagnostic criteria," said Dr. Caselli, who serves as associate director and clinical core director of the Alzheimer’s Disease Center at the Mayo Clinic in Arizona. "The decision to utilize amyloid PET in such cases will depend on the clinician’s own level of comfort but may be less important for those with more experience in these atypical presentations. What it should not do is prevent the appropriate reassessment of patients with an established diagnosis and course whose symptoms suddenly change [because of] a potential new disease process such as subdural hematoma or stroke."
The third indication for an amyloid PET scan is to determine pathology in patients with young-onset dementia – usually someone in the age range of 50-65 years, or sometimes even younger. For these patients, more detailed pathologic information is critical to decision making around issues like work, driving, lifestyle, and long-term planning.
"The presence or absence of AD [Alzheimer’s disease] pathology in this circumstance is frequently a critical component of the initial differential diagnosis," the document noted, "and it is well known from postmortem studies that clinical diagnosis based on history and examination is often wrong with regard to the presence of AD pathology."
This is where amyloid imaging could be the most clinically helpful, Dr. Caselli said.
"In my opinion, the use of amyloid PET will be most important for this indication. Younger patients are employed, have families they are supporting, and usually present early in their course when psychiatric pathology may be suspected. Imagine the 52-year-old who receives a poor job review and states he is unable to understand the new software or recall a client’s name, and seeks medical attention with the implication of disability hanging in the balance. Such a patient will not appear physically ill and is not generally suspected to have Alzheimer’s disease, but when current clinical methods indicate mild cognitive impairment, an amyloid PET scan could solidify the organicity of the diagnosis."
Clinical situations in which not to test
The document’s seven contraindications spell out scenarios in which amyloid imaging is unhelpful. These include:
• Patients who meet both the clinical criteria for Alzheimer’s and the typical age of onset. The potential benefit of detecting amyloid pathology probably would not alter patient management.
• Determining dementia severity. There’s no indication that amyloid burden correlates with symptom severity.
• Relying on reasons based solely on family history or positive apolipoprotein E–epsilon 4 status.
• Investigating a clinically unconfirmed cognitive complaint.
• Using this imaging instead of genotyping patients who have a suspected autosomal mutation.
• Making disease predictions in cognitively normal older adults
• Testing for any nonmedical use, such as for medicolegal, disability, insurance, or employment-related issues.
Patients who undergo the imaging procedure need to understand its benefits and its limitations, the task force said. Amyloid plaques don’t always mean Alzheimer’s disease is present. For example, as age increases, so does the likelihood of amyloid deposition, even in cognitively normal people. "Another major caveat is that a positive amyloid scan can also be seen in not only AD, but also in other medical conditions," such as Lewy body dementia and amyloid angiopathy, the document noted. The scan can’t differentiate these, and it is not useful when evaluating patients with frontotemporal dementia.
But amyloid imaging does confer a number of benefits when used appropriately, according to the task force. Whether the result is positive or negative, it furthers clinical decision-making in appropriately selected patients. It can guide medication management by getting patients on the right drugs faster and keeping them off those that won’t help. The results can shape other diagnostic investigations in the same way by letting patients avoid unnecessary tests or proceed to more informative ones.
There is much benefit in "the value of knowing" amyloid PET scan status, the document noted. About 25% of dementia cases can have reversible causes, like depression or vitamin B12 deficiency. And U.S. surveys have shown that most Americans would prefer to know if their symptoms could be caused by Alzheimer’s so that they can make independent decisions about the future.
But "what such polls often fail to mention are the number of people who would suffer psychological harm if they received ‘bad news’ from such testing," Dr. Caselli cautioned. "In another poll we have been conducting, more than 10% of patients stated they would ‘seriously consider suicide’ if they found there was biomarker evidence of preclinical Alzheimer’s disease. This is a powerful diagnostic modality, but one in which implications of results are not fully understood. What we offer our patients therefore must be limited to the scenarios the committee has described, at least until we have much more experience and understanding of the implications and reliability of test results."
Dr. Johnson and some of the other task force members reported multiple relationships with drug companies. Dr. Caselli has no financial disclosures.
Migraine with aura confers high CV risk in women
Migraine with aura was a large risk factor for cardiovascular events among women who participated in the Women’s Health Study.
Although the physiologic connection isn’t yet clear, the finding is, according to Dr. Tobias Kurth, who is the primary investigator in the study that examined the association. He will present the full results of the study in March at the annual meeting of American Academy of Neurology in San Diego.
"Migraine with aura was the second-largest factor contributing to the risk of heart attack and stroke," said Dr. Kurth of the French National Institute of Health and Medical Research in Bordeaux. "It was ahead of diabetes, smoking, body mass index, and a family history of heart disease."
Dr. Kurth, also of Brigham and Women’s Hospital in Boston, used the Women’s Health Study to examine how migraine with aura fits in with other well-known risk factors for cardiovascular disease in women. The 15-year study was originally designed to examine the impact of low-dose aspirin and vitamin E for the primary prevention of cardiovascular disease and cancer. It enrolled nearly 40,000 women who were aged 45 years or older when they were entered in 1993, he said in an interview.
The cohort for Dr. Kurth’s study comprised 27,860 women who provided baseline blood samples and were without major cardiovascular disease at baseline. Overall, 1,435 had migraine with aura.
During the entire study, there were 1,030 cases of heart attack, stroke, or cardiovascular death. A systolic blood pressure of 180 mm Hg or higher was the strongest risk factor, with an incidence of 9.8 per 1,000 women per year. That was an expected association, Dr. Kurth said.
What he did not expect was to see was an incidence of 7.9 per 1,000 women per year in women who had migraine with aura. This exceeded the rates of cardiovascular disease in all other risk groups, including diabetes (7.1), family history of prior myocardial infarction (5.4), current smoking (5.4), and having a body mass index of 35 kg/m2 or more (5.3).
The study doesn’t address any underlying physiologic connections. There are some theories, however.
"These involve vascular dysfunction," Dr. Kurth said. "People with migraines may have a different vascular reactivity, which might lead to some kind of a shared mechanism between migraine and cardiovascular disease. Inflammation, which is seen in both migraine and cardiovascular disease, might play a role. And genetic factors could be contributing as well."
Recognition of these patients is important, he said. "Patients who have migraine with aura could also potentially develop cardiovascular disease. This should be carefully assessed, because, unlike some of the other risk factors, migraine is not really modifiable."
Patients who have migraine with aura might especially benefit from lifestyle modification to further reduce their risk of heart disease. "People with migraine with aura can reduce their risk in the same ways others can, such as [by] not smoking, keeping blood pressure low and weight down, and exercising."
The National Institutes of Health funded the study. Dr. Kurth had no financial disclosures.
Migraine with aura was a large risk factor for cardiovascular events among women who participated in the Women’s Health Study.
Although the physiologic connection isn’t yet clear, the finding is, according to Dr. Tobias Kurth, who is the primary investigator in the study that examined the association. He will present the full results of the study in March at the annual meeting of American Academy of Neurology in San Diego.
"Migraine with aura was the second-largest factor contributing to the risk of heart attack and stroke," said Dr. Kurth of the French National Institute of Health and Medical Research in Bordeaux. "It was ahead of diabetes, smoking, body mass index, and a family history of heart disease."
Dr. Kurth, also of Brigham and Women’s Hospital in Boston, used the Women’s Health Study to examine how migraine with aura fits in with other well-known risk factors for cardiovascular disease in women. The 15-year study was originally designed to examine the impact of low-dose aspirin and vitamin E for the primary prevention of cardiovascular disease and cancer. It enrolled nearly 40,000 women who were aged 45 years or older when they were entered in 1993, he said in an interview.
The cohort for Dr. Kurth’s study comprised 27,860 women who provided baseline blood samples and were without major cardiovascular disease at baseline. Overall, 1,435 had migraine with aura.
During the entire study, there were 1,030 cases of heart attack, stroke, or cardiovascular death. A systolic blood pressure of 180 mm Hg or higher was the strongest risk factor, with an incidence of 9.8 per 1,000 women per year. That was an expected association, Dr. Kurth said.
What he did not expect was to see was an incidence of 7.9 per 1,000 women per year in women who had migraine with aura. This exceeded the rates of cardiovascular disease in all other risk groups, including diabetes (7.1), family history of prior myocardial infarction (5.4), current smoking (5.4), and having a body mass index of 35 kg/m2 or more (5.3).
The study doesn’t address any underlying physiologic connections. There are some theories, however.
"These involve vascular dysfunction," Dr. Kurth said. "People with migraines may have a different vascular reactivity, which might lead to some kind of a shared mechanism between migraine and cardiovascular disease. Inflammation, which is seen in both migraine and cardiovascular disease, might play a role. And genetic factors could be contributing as well."
Recognition of these patients is important, he said. "Patients who have migraine with aura could also potentially develop cardiovascular disease. This should be carefully assessed, because, unlike some of the other risk factors, migraine is not really modifiable."
Patients who have migraine with aura might especially benefit from lifestyle modification to further reduce their risk of heart disease. "People with migraine with aura can reduce their risk in the same ways others can, such as [by] not smoking, keeping blood pressure low and weight down, and exercising."
The National Institutes of Health funded the study. Dr. Kurth had no financial disclosures.
Migraine with aura was a large risk factor for cardiovascular events among women who participated in the Women’s Health Study.
Although the physiologic connection isn’t yet clear, the finding is, according to Dr. Tobias Kurth, who is the primary investigator in the study that examined the association. He will present the full results of the study in March at the annual meeting of American Academy of Neurology in San Diego.
"Migraine with aura was the second-largest factor contributing to the risk of heart attack and stroke," said Dr. Kurth of the French National Institute of Health and Medical Research in Bordeaux. "It was ahead of diabetes, smoking, body mass index, and a family history of heart disease."
Dr. Kurth, also of Brigham and Women’s Hospital in Boston, used the Women’s Health Study to examine how migraine with aura fits in with other well-known risk factors for cardiovascular disease in women. The 15-year study was originally designed to examine the impact of low-dose aspirin and vitamin E for the primary prevention of cardiovascular disease and cancer. It enrolled nearly 40,000 women who were aged 45 years or older when they were entered in 1993, he said in an interview.
The cohort for Dr. Kurth’s study comprised 27,860 women who provided baseline blood samples and were without major cardiovascular disease at baseline. Overall, 1,435 had migraine with aura.
During the entire study, there were 1,030 cases of heart attack, stroke, or cardiovascular death. A systolic blood pressure of 180 mm Hg or higher was the strongest risk factor, with an incidence of 9.8 per 1,000 women per year. That was an expected association, Dr. Kurth said.
What he did not expect was to see was an incidence of 7.9 per 1,000 women per year in women who had migraine with aura. This exceeded the rates of cardiovascular disease in all other risk groups, including diabetes (7.1), family history of prior myocardial infarction (5.4), current smoking (5.4), and having a body mass index of 35 kg/m2 or more (5.3).
The study doesn’t address any underlying physiologic connections. There are some theories, however.
"These involve vascular dysfunction," Dr. Kurth said. "People with migraines may have a different vascular reactivity, which might lead to some kind of a shared mechanism between migraine and cardiovascular disease. Inflammation, which is seen in both migraine and cardiovascular disease, might play a role. And genetic factors could be contributing as well."
Recognition of these patients is important, he said. "Patients who have migraine with aura could also potentially develop cardiovascular disease. This should be carefully assessed, because, unlike some of the other risk factors, migraine is not really modifiable."
Patients who have migraine with aura might especially benefit from lifestyle modification to further reduce their risk of heart disease. "People with migraine with aura can reduce their risk in the same ways others can, such as [by] not smoking, keeping blood pressure low and weight down, and exercising."
The National Institutes of Health funded the study. Dr. Kurth had no financial disclosures.
Major Finding: Cardiovascular events occurred with an incidence of 7.9 per 1,000 women per year among women who had migraine with aura, second only to hypertension of 180 mm Hg or greater (9.8 per 1,000 women per year.
Data Source: The findings were extracted from 27,860 women who were enrolled in the Women’s Health Study, a prospective cohort study.
Disclosures: The National Institutes of Health funded the study. Dr. Kurth had no financial disclosures.
New strain of norovirus wreaks havoc
A new strain of norovirus, identified last March in Australia, has swept through the United States, causing more than half of the 2012 outbreaks.
GII.4 Sydney is implicated in 53% of the outbreaks that occurred from September to December, Leslie Barclay and her colleagues reported in the Jan. 24 issue of the Morbidity and Mortality Weekly Report (2013;62:55).
The other outbreaks were caused by 10 different GI and GII genotypes, including GII.4 New Orleans, said Ms. Barclay of the National Calicivirus Laboratory at the Centers for Disease Control and Prevention, and her coauthors. However, she noted, the GII.4 Sydney virus appears to have replaced the New Orleans strain as the predominant pathogen.
GII.4 Sydney was implicated in 19% of the September norovirus outbreaks; 46% of the October outbreaks; and 58% of outbreaks in both November and December, Ms. Barclay and her coinvestigators said.
The report noted that 51% of the outbreaks were direct, person-to-person transmission; 20% were foodborne; and 1% waterborne. For the other outbreaks, the mode of transmission remains unknown.
The sudden appearance of a new norovirus strain isn’t unusual or unexpected, Dr. Aron J. Hall said in an interview.
"This group of viruses is constantly evolving. New strains develop all the time," said Dr. Hall, an epidemiologist with CDC’s Division of Viral Diseases. "This is following the same pattern we’ve seen for the past 10 years, with a new strain evolving every 2-3 years."
Since the norovirus season doesn’t normally peak until January, it’s too soon to say whether GII.4 Sydney is more virulent, either in the number or severity of cases it causes.
"Every time a new strain emerges, there’s a potential for increased disease activity, numbers of outbreaks, and severity. But sometimes they simply replace the predominant strain with no increases. We have to wait until we’ve tallied all the numbers before we can get an assessment of this season’s magnitude and impact," he said.
GII.4 Sydney was first identified in Australia, and rapidly moved to Japan, the Netherlands, and the United Kingdom, according to a Jan. 4 report (Eurosurv. 2013;18:1)
In early December and late November, the ProMed disease reporting system reported a 64% increase in confirmed norovirus lab reports in England and Wales, and increased deaths in elderly Japanese patients. Molecular assays pinpointed the cause as the new variant of norovirus, the Eurosurveillance researchers noted.
The virus is highly transmissible; exposure to as few as 18 viral particles can cause illness, according to a presentation by Jan Vinjé, Ph.D., head of the National Calicivirus Laboratory. And since about 30% of cases are asymptomatic, the transmission rate can be extremely high.
"These are the two characteristics that explain why norovirus is such a serious public health challenge," Dr. Hall said. "It takes a very tiny amount of exposure to make someone sick, and someone who is sick is shedding billions of viral particles. They can shed virus without any symptoms, so they don’t know they have it and there’s no way to advise them about how to avoid spreading it."
The illness is characterized by vomiting and/or diarrhea with abdominal cramps and low-grade fever. Most patients recover within 12-72 hours, but about 10% need hospitalization and fluid therapy. Severe illness and/or death are most common among the elderly and people with chronic illnesses.
The United States sees about 21 million cases annually; 800 will result in death. A norovirus outbreak can overwhelm local medical resources; the virus causes nearly 2 million outpatient visits and 400,000 emergency department visits each year. The combined annual cost reaches $2 billion, according to the CDC.
As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
GII.4 Sydney, Leslie Barclay, Morbidity and Mortality Weekly Report, GII.4 New Orleans, National Calicivirus Laboratory at the Centers for Disease Control and Prevention, Dr. Aron J. Hall, CDC’s Division of Viral Diseases,
A new strain of norovirus, identified last March in Australia, has swept through the United States, causing more than half of the 2012 outbreaks.
GII.4 Sydney is implicated in 53% of the outbreaks that occurred from September to December, Leslie Barclay and her colleagues reported in the Jan. 24 issue of the Morbidity and Mortality Weekly Report (2013;62:55).
The other outbreaks were caused by 10 different GI and GII genotypes, including GII.4 New Orleans, said Ms. Barclay of the National Calicivirus Laboratory at the Centers for Disease Control and Prevention, and her coauthors. However, she noted, the GII.4 Sydney virus appears to have replaced the New Orleans strain as the predominant pathogen.
GII.4 Sydney was implicated in 19% of the September norovirus outbreaks; 46% of the October outbreaks; and 58% of outbreaks in both November and December, Ms. Barclay and her coinvestigators said.
The report noted that 51% of the outbreaks were direct, person-to-person transmission; 20% were foodborne; and 1% waterborne. For the other outbreaks, the mode of transmission remains unknown.
The sudden appearance of a new norovirus strain isn’t unusual or unexpected, Dr. Aron J. Hall said in an interview.
"This group of viruses is constantly evolving. New strains develop all the time," said Dr. Hall, an epidemiologist with CDC’s Division of Viral Diseases. "This is following the same pattern we’ve seen for the past 10 years, with a new strain evolving every 2-3 years."
Since the norovirus season doesn’t normally peak until January, it’s too soon to say whether GII.4 Sydney is more virulent, either in the number or severity of cases it causes.
"Every time a new strain emerges, there’s a potential for increased disease activity, numbers of outbreaks, and severity. But sometimes they simply replace the predominant strain with no increases. We have to wait until we’ve tallied all the numbers before we can get an assessment of this season’s magnitude and impact," he said.
GII.4 Sydney was first identified in Australia, and rapidly moved to Japan, the Netherlands, and the United Kingdom, according to a Jan. 4 report (Eurosurv. 2013;18:1)
In early December and late November, the ProMed disease reporting system reported a 64% increase in confirmed norovirus lab reports in England and Wales, and increased deaths in elderly Japanese patients. Molecular assays pinpointed the cause as the new variant of norovirus, the Eurosurveillance researchers noted.
The virus is highly transmissible; exposure to as few as 18 viral particles can cause illness, according to a presentation by Jan Vinjé, Ph.D., head of the National Calicivirus Laboratory. And since about 30% of cases are asymptomatic, the transmission rate can be extremely high.
"These are the two characteristics that explain why norovirus is such a serious public health challenge," Dr. Hall said. "It takes a very tiny amount of exposure to make someone sick, and someone who is sick is shedding billions of viral particles. They can shed virus without any symptoms, so they don’t know they have it and there’s no way to advise them about how to avoid spreading it."
The illness is characterized by vomiting and/or diarrhea with abdominal cramps and low-grade fever. Most patients recover within 12-72 hours, but about 10% need hospitalization and fluid therapy. Severe illness and/or death are most common among the elderly and people with chronic illnesses.
The United States sees about 21 million cases annually; 800 will result in death. A norovirus outbreak can overwhelm local medical resources; the virus causes nearly 2 million outpatient visits and 400,000 emergency department visits each year. The combined annual cost reaches $2 billion, according to the CDC.
As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
A new strain of norovirus, identified last March in Australia, has swept through the United States, causing more than half of the 2012 outbreaks.
GII.4 Sydney is implicated in 53% of the outbreaks that occurred from September to December, Leslie Barclay and her colleagues reported in the Jan. 24 issue of the Morbidity and Mortality Weekly Report (2013;62:55).
The other outbreaks were caused by 10 different GI and GII genotypes, including GII.4 New Orleans, said Ms. Barclay of the National Calicivirus Laboratory at the Centers for Disease Control and Prevention, and her coauthors. However, she noted, the GII.4 Sydney virus appears to have replaced the New Orleans strain as the predominant pathogen.
GII.4 Sydney was implicated in 19% of the September norovirus outbreaks; 46% of the October outbreaks; and 58% of outbreaks in both November and December, Ms. Barclay and her coinvestigators said.
The report noted that 51% of the outbreaks were direct, person-to-person transmission; 20% were foodborne; and 1% waterborne. For the other outbreaks, the mode of transmission remains unknown.
The sudden appearance of a new norovirus strain isn’t unusual or unexpected, Dr. Aron J. Hall said in an interview.
"This group of viruses is constantly evolving. New strains develop all the time," said Dr. Hall, an epidemiologist with CDC’s Division of Viral Diseases. "This is following the same pattern we’ve seen for the past 10 years, with a new strain evolving every 2-3 years."
Since the norovirus season doesn’t normally peak until January, it’s too soon to say whether GII.4 Sydney is more virulent, either in the number or severity of cases it causes.
"Every time a new strain emerges, there’s a potential for increased disease activity, numbers of outbreaks, and severity. But sometimes they simply replace the predominant strain with no increases. We have to wait until we’ve tallied all the numbers before we can get an assessment of this season’s magnitude and impact," he said.
GII.4 Sydney was first identified in Australia, and rapidly moved to Japan, the Netherlands, and the United Kingdom, according to a Jan. 4 report (Eurosurv. 2013;18:1)
In early December and late November, the ProMed disease reporting system reported a 64% increase in confirmed norovirus lab reports in England and Wales, and increased deaths in elderly Japanese patients. Molecular assays pinpointed the cause as the new variant of norovirus, the Eurosurveillance researchers noted.
The virus is highly transmissible; exposure to as few as 18 viral particles can cause illness, according to a presentation by Jan Vinjé, Ph.D., head of the National Calicivirus Laboratory. And since about 30% of cases are asymptomatic, the transmission rate can be extremely high.
"These are the two characteristics that explain why norovirus is such a serious public health challenge," Dr. Hall said. "It takes a very tiny amount of exposure to make someone sick, and someone who is sick is shedding billions of viral particles. They can shed virus without any symptoms, so they don’t know they have it and there’s no way to advise them about how to avoid spreading it."
The illness is characterized by vomiting and/or diarrhea with abdominal cramps and low-grade fever. Most patients recover within 12-72 hours, but about 10% need hospitalization and fluid therapy. Severe illness and/or death are most common among the elderly and people with chronic illnesses.
The United States sees about 21 million cases annually; 800 will result in death. A norovirus outbreak can overwhelm local medical resources; the virus causes nearly 2 million outpatient visits and 400,000 emergency department visits each year. The combined annual cost reaches $2 billion, according to the CDC.
As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
GII.4 Sydney, Leslie Barclay, Morbidity and Mortality Weekly Report, GII.4 New Orleans, National Calicivirus Laboratory at the Centers for Disease Control and Prevention, Dr. Aron J. Hall, CDC’s Division of Viral Diseases,
GII.4 Sydney, Leslie Barclay, Morbidity and Mortality Weekly Report, GII.4 New Orleans, National Calicivirus Laboratory at the Centers for Disease Control and Prevention, Dr. Aron J. Hall, CDC’s Division of Viral Diseases,
FROM THE MORBIDITY AND MORTALITY WEEKLY REPORT
Major Finding: A new strain of norovirus caused 53% of outbreaks in the United States from September-December 2012.
Data Source: These statistics were obtained from the Centers for Disease Control and Prevention’s National Calicivirus Laboratory
Disclosures: As federal employees, Dr. Hall and Ms. Barclay have no financial disclosures.
Paclitaxel-eluting stent approved for PAD
The Food and Drug Administration has approved a new paclitaxel-eluting stent indciated for the treatment of peripheral artery disease.
The Zilver PTX Drug-Eluting Peripheral Stent, which is manufactured by Cook Medical of Bloomington, Ind., is the first drug-eluting stent to win approval for this indication.
The approval was based on findings from both a randomized controlled trial and on a registry study, which together comprised more than 1,200 patients.
According to an FDA press statement, the studies indicate that treatment with the stent "is at least as safe as treatment with percutaneous transluminal angioplasty (PTA) and significantly more effective."
The randomized trial included a total of 479 patients who had a single stenotic lesion less than 140 mm in one or both of the femoropopliteal arteries.
The patients were randomized to the paclitaxel-eluting stent or to PTA. If the transluminal procedure failed, then the patients received either the paclitaxel-eluting stent or a bare-metal stent.
After 12 months, 83% of the arteries treated with the drug-eluting stent were still open, compared with 33% of those in the PTA group.
In those patients who had the stent placed after a failed PTA, 90% of arteries were open, compared with 73% in those who got the bare-metal stent.
In October 2011, the FDA's Circulatory System Devices Panel voted 11 to 0 that the benefits of the Zilver PTX stent outweighed its risks as a treatment for patients with symptomatic atherosclerotic stenosis of the femoropopliteal arteries on the basis of that trial.
This past October, the 3-year results of the study were presented at the Vascular Interventional Advances 2012 meeting in Las Vegas. The results showed that there was a 70.7% primary patency seen for the paclitaxel-eluting stent, compared with 49.1% for PTA and bare-metal stents.
The registry study followed 767 patients for 24 months. These patients had a maximum of four stents placed; the stents could be utilized to treat a single lesion or to treat multiple lesions.
At 12 months, the fracture rate was 1.5%; fractures were not associated with any clinical problems. The rate of stent thrombosis was 2.8% at 12 months and 3.5% at 24 months.
"The clinical study demonstrated that the [the paclitaxel-eluting stent] is more effective than the use of balloon angioplasty for the treatment of symptomatic peripheral artery disease in above-the-knee femoropopliteal artery," Christy Foreman, director of the Office of Device Evaluation at the FDA's Center for Devices and Radiological Health, said in the statement.
"This approval expands the treatment options for patients suffering from symptomatic peripheral artery disease," she added.
In both studies, the most common major adverse event was restenosis requiring additional treatment to reestablish adequate flow in the artery.
The device is contraindicated in patients with stenoses that cannot be dilated to permit passage of the catheter or proper placement of the stent.
It is also contraindicated in patients who cannot receive recommended drug therapy due to bleeding disorders, or women who are pregnant, breastfeeding, or planning to become pregnant in the next 5 years.
The FDA will now require the manufacturer to conduct a 5-year postapproval study of 900 patients to further evaluate the stent's safety and efficacy.
The banging noise you hear emanating from your radiologic imaging area is not the MRI machine, but nails being pounded into the coffin of the fem-pop bypass graft operation. Incremental but significant progress has been made in treating SFA occlusive disease over the last decade from POBA to tools that facilitate crossing/reentry of CTOs to bare-metal/covered stenting to DES. The Zilver PTX trial and registry have resulted in commercial approval of a long enough, large enough self-expanding stent to be useful in peripheral interventions.
| Dr. Brian Rubin |
Treated lesions included ISRs as well as primary atherosclerotic lesions, and data from both trials showed near-identical 12 and 24 month results, with Zilver PTX resulting in primary patency rates 15-20% higher than results with bare-metal stenting alone. As of mid-November 2012, the complete FDA 57-page PDF of both studies is available online and should be required reading While most surgeons would claim their 12 month fem-AK pop bypass patency to be better than the 83% primary patency reported for Zilver PTX, multiple recent published reports suggest otherwise.
A number of important issues remain unresolved including: 1. how much will each stent cost? (the grapevine has it that the pricing will be surprisingly low) 2. am I really limited to a maximum of 14 cm treated length per leg? (probably not since paclitaxel levels peaked at less than 30 minutes, were gone within a few hours and were very low anyway 3. what antiplatelet therapy is required? (still to be determined- ASA alone is probably adequate) and 4. how do I treat smaller diameter vessels? (Zilver PTX is only approved in 6-8 mm diameters). Although 3-year data have recently been reported, the long-term outcomes after DES remain unknown at this time.
While fem-pop bypass may still find limited utility and needs to remain in the surgical armamentarium, the commercial approval of Zilver PTX stents has nibbled away again at the difference in outcomes between endovascular and open surgical therapy for SFA occlusive disease.
If other studies including long-term outcome reports confirm these initial salutary results, the fem-pop graft will soon follow the utilization curve of open aortic surgery and become an endangered operation.
Dr. Brian Rubin is a professor of the department of surgery at the Washington University School of Medicine, St. Louis.
The banging noise you hear emanating from your radiologic imaging area is not the MRI machine, but nails being pounded into the coffin of the fem-pop bypass graft operation. Incremental but significant progress has been made in treating SFA occlusive disease over the last decade from POBA to tools that facilitate crossing/reentry of CTOs to bare-metal/covered stenting to DES. The Zilver PTX trial and registry have resulted in commercial approval of a long enough, large enough self-expanding stent to be useful in peripheral interventions.
| Dr. Brian Rubin |
Treated lesions included ISRs as well as primary atherosclerotic lesions, and data from both trials showed near-identical 12 and 24 month results, with Zilver PTX resulting in primary patency rates 15-20% higher than results with bare-metal stenting alone. As of mid-November 2012, the complete FDA 57-page PDF of both studies is available online and should be required reading While most surgeons would claim their 12 month fem-AK pop bypass patency to be better than the 83% primary patency reported for Zilver PTX, multiple recent published reports suggest otherwise.
A number of important issues remain unresolved including: 1. how much will each stent cost? (the grapevine has it that the pricing will be surprisingly low) 2. am I really limited to a maximum of 14 cm treated length per leg? (probably not since paclitaxel levels peaked at less than 30 minutes, were gone within a few hours and were very low anyway 3. what antiplatelet therapy is required? (still to be determined- ASA alone is probably adequate) and 4. how do I treat smaller diameter vessels? (Zilver PTX is only approved in 6-8 mm diameters). Although 3-year data have recently been reported, the long-term outcomes after DES remain unknown at this time.
While fem-pop bypass may still find limited utility and needs to remain in the surgical armamentarium, the commercial approval of Zilver PTX stents has nibbled away again at the difference in outcomes between endovascular and open surgical therapy for SFA occlusive disease.
If other studies including long-term outcome reports confirm these initial salutary results, the fem-pop graft will soon follow the utilization curve of open aortic surgery and become an endangered operation.
Dr. Brian Rubin is a professor of the department of surgery at the Washington University School of Medicine, St. Louis.
The banging noise you hear emanating from your radiologic imaging area is not the MRI machine, but nails being pounded into the coffin of the fem-pop bypass graft operation. Incremental but significant progress has been made in treating SFA occlusive disease over the last decade from POBA to tools that facilitate crossing/reentry of CTOs to bare-metal/covered stenting to DES. The Zilver PTX trial and registry have resulted in commercial approval of a long enough, large enough self-expanding stent to be useful in peripheral interventions.
| Dr. Brian Rubin |
Treated lesions included ISRs as well as primary atherosclerotic lesions, and data from both trials showed near-identical 12 and 24 month results, with Zilver PTX resulting in primary patency rates 15-20% higher than results with bare-metal stenting alone. As of mid-November 2012, the complete FDA 57-page PDF of both studies is available online and should be required reading While most surgeons would claim their 12 month fem-AK pop bypass patency to be better than the 83% primary patency reported for Zilver PTX, multiple recent published reports suggest otherwise.
A number of important issues remain unresolved including: 1. how much will each stent cost? (the grapevine has it that the pricing will be surprisingly low) 2. am I really limited to a maximum of 14 cm treated length per leg? (probably not since paclitaxel levels peaked at less than 30 minutes, were gone within a few hours and were very low anyway 3. what antiplatelet therapy is required? (still to be determined- ASA alone is probably adequate) and 4. how do I treat smaller diameter vessels? (Zilver PTX is only approved in 6-8 mm diameters). Although 3-year data have recently been reported, the long-term outcomes after DES remain unknown at this time.
While fem-pop bypass may still find limited utility and needs to remain in the surgical armamentarium, the commercial approval of Zilver PTX stents has nibbled away again at the difference in outcomes between endovascular and open surgical therapy for SFA occlusive disease.
If other studies including long-term outcome reports confirm these initial salutary results, the fem-pop graft will soon follow the utilization curve of open aortic surgery and become an endangered operation.
Dr. Brian Rubin is a professor of the department of surgery at the Washington University School of Medicine, St. Louis.
The Food and Drug Administration has approved a new paclitaxel-eluting stent indciated for the treatment of peripheral artery disease.
The Zilver PTX Drug-Eluting Peripheral Stent, which is manufactured by Cook Medical of Bloomington, Ind., is the first drug-eluting stent to win approval for this indication.
The approval was based on findings from both a randomized controlled trial and on a registry study, which together comprised more than 1,200 patients.
According to an FDA press statement, the studies indicate that treatment with the stent "is at least as safe as treatment with percutaneous transluminal angioplasty (PTA) and significantly more effective."
The randomized trial included a total of 479 patients who had a single stenotic lesion less than 140 mm in one or both of the femoropopliteal arteries.
The patients were randomized to the paclitaxel-eluting stent or to PTA. If the transluminal procedure failed, then the patients received either the paclitaxel-eluting stent or a bare-metal stent.
After 12 months, 83% of the arteries treated with the drug-eluting stent were still open, compared with 33% of those in the PTA group.
In those patients who had the stent placed after a failed PTA, 90% of arteries were open, compared with 73% in those who got the bare-metal stent.
In October 2011, the FDA's Circulatory System Devices Panel voted 11 to 0 that the benefits of the Zilver PTX stent outweighed its risks as a treatment for patients with symptomatic atherosclerotic stenosis of the femoropopliteal arteries on the basis of that trial.
This past October, the 3-year results of the study were presented at the Vascular Interventional Advances 2012 meeting in Las Vegas. The results showed that there was a 70.7% primary patency seen for the paclitaxel-eluting stent, compared with 49.1% for PTA and bare-metal stents.
The registry study followed 767 patients for 24 months. These patients had a maximum of four stents placed; the stents could be utilized to treat a single lesion or to treat multiple lesions.
At 12 months, the fracture rate was 1.5%; fractures were not associated with any clinical problems. The rate of stent thrombosis was 2.8% at 12 months and 3.5% at 24 months.
"The clinical study demonstrated that the [the paclitaxel-eluting stent] is more effective than the use of balloon angioplasty for the treatment of symptomatic peripheral artery disease in above-the-knee femoropopliteal artery," Christy Foreman, director of the Office of Device Evaluation at the FDA's Center for Devices and Radiological Health, said in the statement.
"This approval expands the treatment options for patients suffering from symptomatic peripheral artery disease," she added.
In both studies, the most common major adverse event was restenosis requiring additional treatment to reestablish adequate flow in the artery.
The device is contraindicated in patients with stenoses that cannot be dilated to permit passage of the catheter or proper placement of the stent.
It is also contraindicated in patients who cannot receive recommended drug therapy due to bleeding disorders, or women who are pregnant, breastfeeding, or planning to become pregnant in the next 5 years.
The FDA will now require the manufacturer to conduct a 5-year postapproval study of 900 patients to further evaluate the stent's safety and efficacy.
The Food and Drug Administration has approved a new paclitaxel-eluting stent indciated for the treatment of peripheral artery disease.
The Zilver PTX Drug-Eluting Peripheral Stent, which is manufactured by Cook Medical of Bloomington, Ind., is the first drug-eluting stent to win approval for this indication.
The approval was based on findings from both a randomized controlled trial and on a registry study, which together comprised more than 1,200 patients.
According to an FDA press statement, the studies indicate that treatment with the stent "is at least as safe as treatment with percutaneous transluminal angioplasty (PTA) and significantly more effective."
The randomized trial included a total of 479 patients who had a single stenotic lesion less than 140 mm in one or both of the femoropopliteal arteries.
The patients were randomized to the paclitaxel-eluting stent or to PTA. If the transluminal procedure failed, then the patients received either the paclitaxel-eluting stent or a bare-metal stent.
After 12 months, 83% of the arteries treated with the drug-eluting stent were still open, compared with 33% of those in the PTA group.
In those patients who had the stent placed after a failed PTA, 90% of arteries were open, compared with 73% in those who got the bare-metal stent.
In October 2011, the FDA's Circulatory System Devices Panel voted 11 to 0 that the benefits of the Zilver PTX stent outweighed its risks as a treatment for patients with symptomatic atherosclerotic stenosis of the femoropopliteal arteries on the basis of that trial.
This past October, the 3-year results of the study were presented at the Vascular Interventional Advances 2012 meeting in Las Vegas. The results showed that there was a 70.7% primary patency seen for the paclitaxel-eluting stent, compared with 49.1% for PTA and bare-metal stents.
The registry study followed 767 patients for 24 months. These patients had a maximum of four stents placed; the stents could be utilized to treat a single lesion or to treat multiple lesions.
At 12 months, the fracture rate was 1.5%; fractures were not associated with any clinical problems. The rate of stent thrombosis was 2.8% at 12 months and 3.5% at 24 months.
"The clinical study demonstrated that the [the paclitaxel-eluting stent] is more effective than the use of balloon angioplasty for the treatment of symptomatic peripheral artery disease in above-the-knee femoropopliteal artery," Christy Foreman, director of the Office of Device Evaluation at the FDA's Center for Devices and Radiological Health, said in the statement.
"This approval expands the treatment options for patients suffering from symptomatic peripheral artery disease," she added.
In both studies, the most common major adverse event was restenosis requiring additional treatment to reestablish adequate flow in the artery.
The device is contraindicated in patients with stenoses that cannot be dilated to permit passage of the catheter or proper placement of the stent.
It is also contraindicated in patients who cannot receive recommended drug therapy due to bleeding disorders, or women who are pregnant, breastfeeding, or planning to become pregnant in the next 5 years.
The FDA will now require the manufacturer to conduct a 5-year postapproval study of 900 patients to further evaluate the stent's safety and efficacy.