User login
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
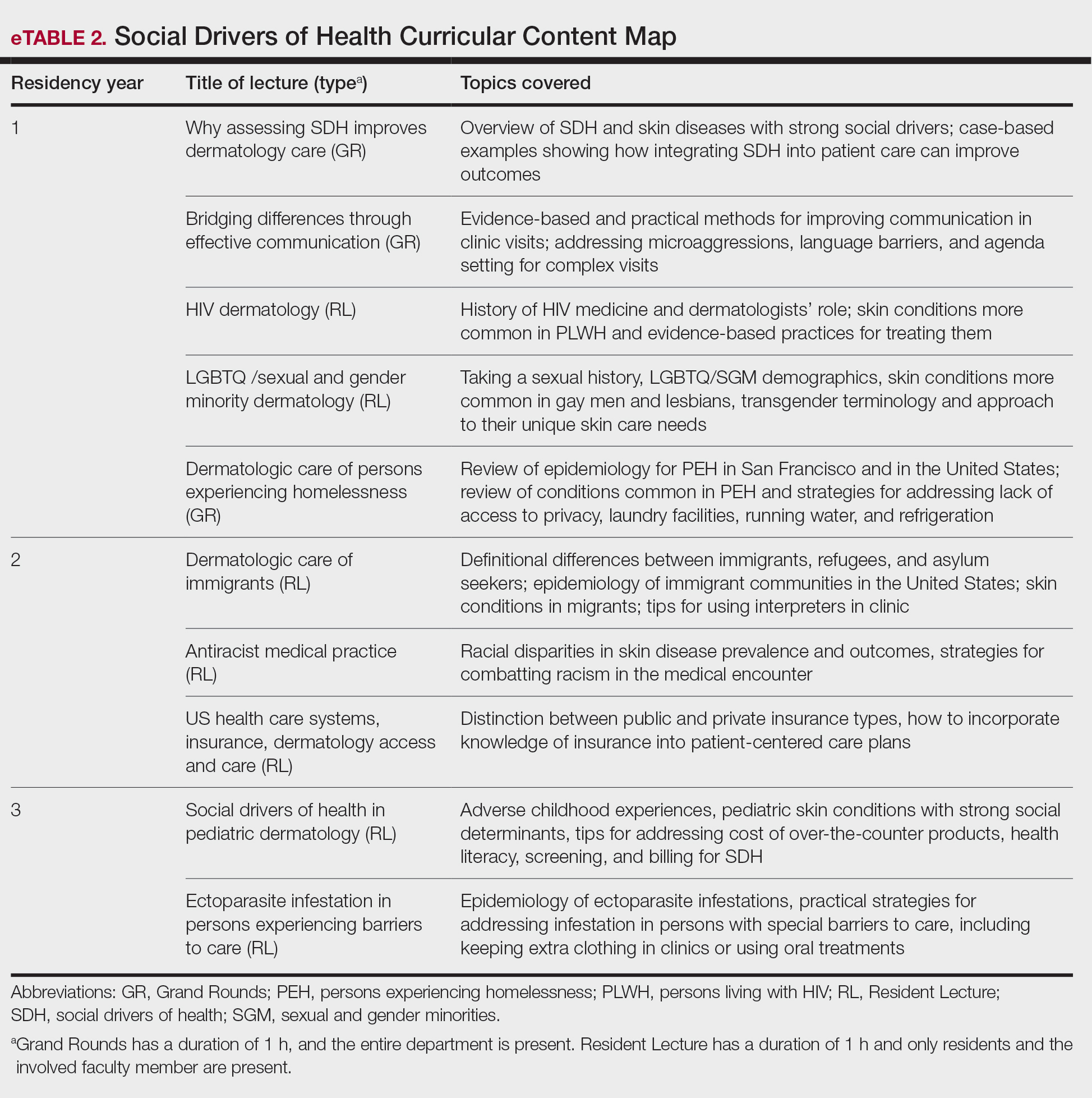
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.

To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Practice Points
- Integrating a formal curriculum on social drivers of health, including didactics on structural racism, cultural humility, communication, and practical strategies, can help residents learn to routinely assess social needs and develop feasible patient-centered care plans.
- Classroom learning paired with experiential rotations and community engagement in safety-net hospitals can help to build empathy, practical skills, and comfort managing real-world social barriers.
- Creation of trainee leadership roles and dedicated program support (eg, departmental funding) can sustain curriculum improvements, foster advocacy skills, and diversify the workforce pipeline.
Results From the First Annual Association of Professors of Dermatology Program Directors Survey
Educational organizations across several specialties, including internal medicine and obstetrics and gynecology, have formal surveys1; however, the field of dermatology has been without one. This study aimed to establish a formal survey for dermatology program directors (PDs) and clinician-educators. Because the Accreditation Council for Graduate Medical Education (ACGME) and American Board of Dermatology surveys do not capture all metrics relevant to dermatology residency educators, an annual survey for our specialty may be helpful to compare dermatology-specific data among programs. Responses could provide context and perspective to faculty and residents who respond to the ACGME annual survey, as our Association of Professors of Dermatology (APD) survey asks more in-depth questions, such as how often didactics occur and who leads them. Resident commute time and faculty demographics and training also are covered. Current ad hoc surveys disseminated through listserves of various medical associations contain overlapping questions and reflect relatively low response rates; dermatology PDs may benefit from a survey with a high response rate to which they can contribute future questions and topics that reflect recent trends and current needs in graduate medical education. As future surveys are administered, the results can be captured in a centralized database accessible by dermatology PDs.
Methods
A survey of PDs from 141 ACGME-accredited dermatology residency programs was conducted by the Residency Program Director Steering Committee of the APD from November 2022 to January 2023 using a prevalidated questionnaire. Personalized survey links were created and sent individually to each PD’s email listed in the ACGME accreditation data system. All survey responses were captured anonymously, with a number assigned to keep de-identified responses separate and organized. The survey consisted of 137 survey questions addressing topics that included program characteristics, PD demographics, the impact of the COVID-19 pandemic on clinical rotation and educational conferences, available resident resources, quality improvement, clinical and didactic instruction, research content, diversity and inclusion, wellness, professionalism, evaluation systems, and graduate outcomes.
Data were collected using Qualtrics survey tools. After removing duplicate and incomplete surveys, data were analyzed using Qualtrics reports and Microsoft Excel for data plotting, averages, and range calculations.
Results
One hundred forty-one personalized survey links were created and sent individually to each program’s filed email obtained from the APD listserv. Fifty-three responses were recorded after removing duplicate or incomplete surveys (38% [53/141] response rate). As of May 2023, there were 144 ACGME-accredited dermatology residency programs due to 3 newly accredited programs in 2022-2023 academic year, which were not included in our survey population.
Program Characteristics—Forty-four respondents (83%) were from a university-based program. Fifty respondents (94%) were from programs that were ACGME accredited prior to 2020, while 3 programs (6%) were American Osteopathic Association accredited prior to singular accreditation. Seventy-one percent (38/53) of respondents had 1 or more associate PDs.
PD Demographics—Eighty-seven percent (45/52) of PDs who responded to the survey graduated from a US allopathic medical school (MD), 10% (5/52) graduated from a US osteopathic medical school (DO), and 4% (2/52) graduated from an international medical school. Seventy-four percent (35/47) of respondents were White, 17% (8/47) were Asian, and 2% (1/47) were Black or African American; this data was not provided for 4 respondents. Forty-eight percent (23/48) of PDs identified as cisgender man, 48% (23/48) identified as cisgender woman, and 4% (2/48) preferred not to answer. Eighty-one percent (38/47) of PDs identified as heterosexual or straight, 15% (7/47) identified as gay or lesbian, and 4% (2/47) preferred not to answer.
Impact of COVID-19 Pandemic on Residency Training—Due to the COVID-19 pandemic, 88% (45/51) of respondents incorporated telemedicine into the resident clinical rotation schedule. Moving forward, 75% (38/51) of respondents indicated that their programs plan to continue to incorporate telemedicine into the rotation schedule. Based on 50 responses, the average of educational conferences that became virtual at the start of the COVID-19 pandemic was 87%; based on 46 responses, the percentage of educational conferences that will remain virtual moving forward is 46%, while 90% (46/51) of respondents indicated that their programs plan to use virtual conferences in some capacity moving forward. Seventy-three percent (37/51) of respondents indicated that they plan to use virtual interviews as part of residency recruitment moving forward.
Available Resources—Twenty-four percent (11/46) of respondents indicated that residents in their program do not get protected time or time off for CORE examinations. Seventy-five percent (33/44) of PDs said their program provides funding for residents to participate in board review courses. The chief residents at 63% (31/49) of programs receive additional compensation, and 69% (34/49) provide additional administrative time to chief residents. Seventy-one percent (24/34) of PDs reported their programs have scribes for attendings, and 12% (4/34) have scribes for residents. Support staff help residents with callbacks and in-basket messages according to 76% (35/46) of respondents. The majority (98% [45/46]) of PDs indicated that residents follow-up on results and messages from patients seen in resident clinics, and 43% (20/46) of programs have residents follow-up with patients seen in faculty clinics. Only 15% (7/46) of PDs responded they have schedules with residents dedicated to handle these tasks. According to respondents, 33% (17/52) have residents who are required to travel more than 25 miles to distant clinical sites. Of them, 35% (6/17) provide accommodations.
Quality Improvement—Seventy-one percent (35/49) of respondents indicated their department has a quality improvement/patient safety team or committee, and 94% (33/35) of these teams include residents. A lecture series on quality improvement and patient safety is offered at 67% (33/49) of the respondents’ programs, while morbidity and mortality conferences are offered in 73% (36/49).
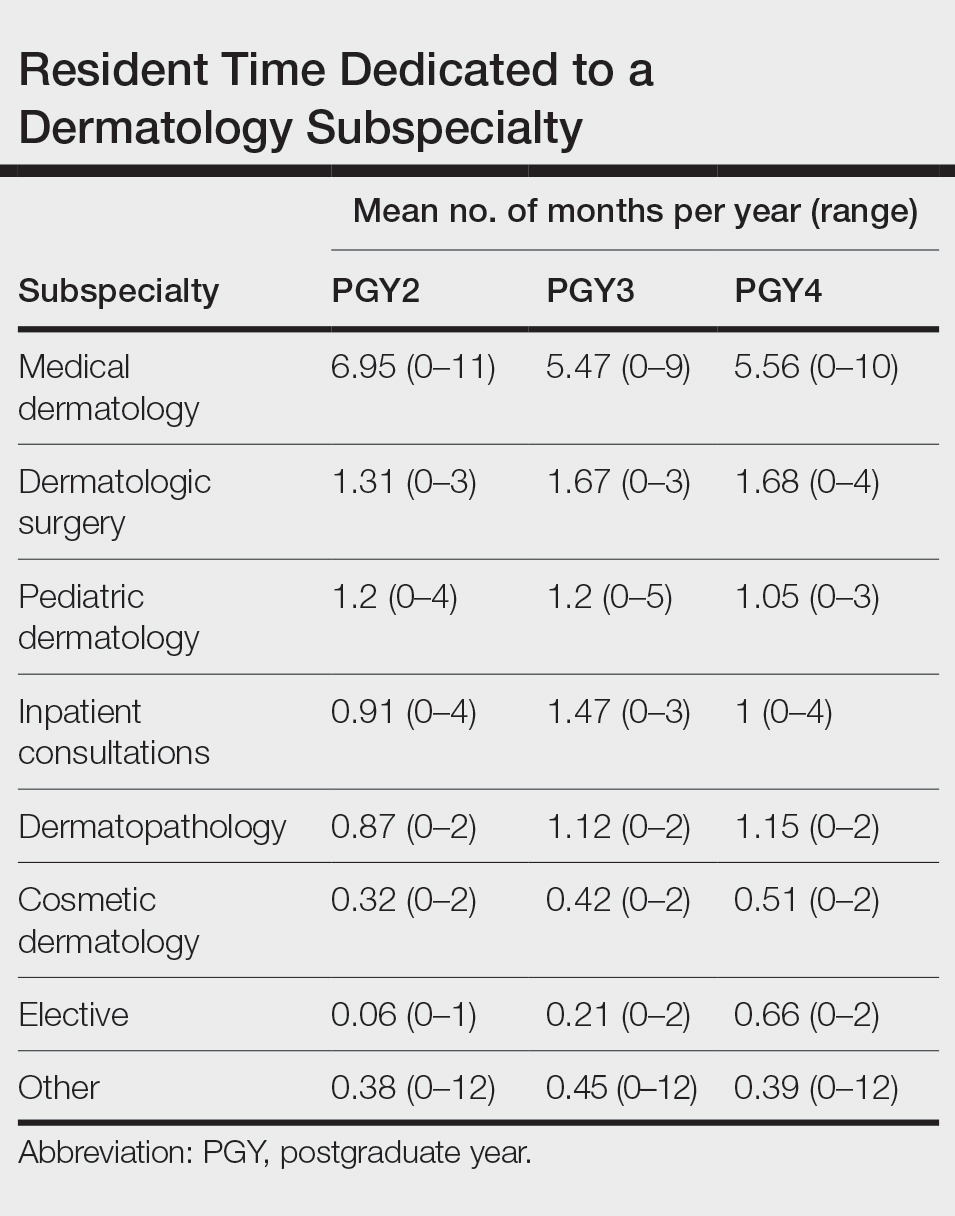
Clinical Instruction—Our survey asked PDs how many months each residency year spends on a certain rotational service. Based on 46 respondents, the average number of months dedicated to medical dermatology is 7, 5, and 6 months for postgraduate year (PGY) 2, PGY3, and PGY4, respectively. The average number of months spent in other subspecialties is provided in the Table. On average, PGY2 residents spend 8 half-days per week seeing patients in clinic, while PGY3 and PGY4 residents see patients for 7 half-days. The median and mean number of patients staffed by a single attending per hour in teaching clinics are 6 and 5.88, respectively. Respondents indicated that residents participate in the following specialty clinics: pediatric dermatology (96% [44/46]), laser/cosmetic (87% [40/44]), high-risk skin cancer (ie, immunosuppressed/transplant patient)(65% [30/44]), pigmented lesion/melanoma (52% [24/44]), connective tissue disease (52% [24/44]), teledermatology (50% [23/44]), free clinic for homeless and/or indigent populations (48% [22/44]), contact dermatitis (43% [20/44]), skin of color (43% [20/44]), oncodermatology (41% [19/44]), and bullous disease (33% [15/44]).
Additionally, in 87% (40/46) of programs, residents participate in a dedicated inpatient consultation service. Most respondents (98% [45/46]) responded that they utilize in-person consultations with a teledermatology supplement. Fifteen percent (7/46) utilize virtual teledermatology (live video-based consultations), and 57% (26/46) utilize asynchronous teledermatology (picture-based consultations). All respondents (n=46) indicated that 0% to 25% of patient encounters involving residents are teledermatology visits. Thirty-three percent (6/18) of programs have a global health special training track, 56% (10/18) have a Specialty Training and Advanced Research/Physician-Scientist Research Training track, 28% (5/18) have a diversity training track, and 50% (9/18) have a clinician educator training track.
Didactic Instruction—Five programs have a full day per week dedicated to didactics, while 36 programs have at least one half-day per week for didactics. On average, didactics in 57% (26/46) of programs are led by faculty alone, while 43% (20/46) are led at least in part by residents or fellows.
Research Content—Fifty percent (23/46) of programs have a specific research requirement for residents beyond general ACGME requirements, and 35% (16/46) require residents to participate in a longitudinal research project over the course of residency. There is a dedicated research coordinator for resident support at 63% (29/46) of programs. Dedicated biostatistics research support is available for resident projects at 42% (19/45) of programs. Additionally, at 42% (19/45) of programs, there is a dedicated faculty member for oversight of resident research.

Diversity, Equity, and Inclusion—Seventy-three percent (29/40) of programs have special diversity, equity, and inclusion programs or meetings specific to residency, 60% (24/40) have residency initiatives, and 55% (22/40) have a residency diversity committee. Eighty-six percent (42/49) of respondents strongly agreed that their current residents represent diverse ethnic and racial backgrounds (ie, >15% are not White). eTable 1 shows PD responses to this statement, which were stratified based on self-identified race. eTable 2 shows PD responses to the statement, “Our current residents represent an inclusion of gender/sexual orientation,” which were stratified based on self-identified gender identity/sexual orientation. Lastly, eTable 3 highlights the percentage of residents with an MD and DO degree, stratified based on PD degree.

Wellness—Forty-eight percent (20/42) of respondents indicated they are under stress and do not always have as much energy as before becoming a PD but do not feel burned out. Thirty-one percent (13/42) indicated they have 1 or more symptoms of burnout, such as emotional exhaustion. Eighty-six percent (36/42) are satisfied with their jobs overall (43% agree and 43% strongly agree [18/42 each]).
Evaluation System—Seventy-five percent (33/44) of programs deliver evaluations of residents by faculty online, 86% (38/44) of programs have PDs discuss evaluations in-person, and 20% (9/44) of programs have faculty evaluators discuss evaluations in-person. Seventy-seven percent (34/44) of programs have formal faculty-resident mentor-mentee programs. Clinical competency committee chair positions are filled by PDs, assistant PDs, or core faculty members 47%, 38%, and 16% of the time, respectively.
Graduation Outcomes of PGY4 Residents—About 28% (55/199) of graduating residents applied to a fellowship position, with the majority (15% [29/55]) matching into Mohs micrographic surgery and dermatologic oncology (MSDO) fellowships. Approximately 5% (9/199) and 4% (7/199) of graduates matched into dermatopathology and pediatric dermatology, respectively. The remaining 5% (10/199) of graduating residents applied to a fellowship but did not match. The majority (45% [91/199]) of residency graduates entered private practice after graduation. Approximately 21% (42/199) of graduating residents chose an academic practice with 17% (33/199), 2% (4/199), and 2% (3/199) of those positions being full-time, part-time, and adjunct, respectively.
Comment
The first annual APD survey is a novel data source and provides opportunities for areas of discussion and investigation. Evaluating the similarities and differences among dermatology residency programs across the United States can strengthen individual programs through collaboration and provide areas of cohesion among programs.
Diversity of PDs—An important area of discussion is diversity and PD demographics. Although DO students make up 1 in 4 US graduating medical students, they are not interviewed or ranked as often as MD students.2 Diversity in PD race and ethnicity may be worthy of investigation in future studies, as match rates and recruitment of diverse medical school applicants may be impacted by these demographics.
Continued Use of Telemedicine in Training—Since 2020, the benefits of virtual residency recruitment have been debated among PDs across all medical specialties. Points in favor of virtual interviews include cost savings for programs and especially for applicants, as well as time efficiency, reduced burden of travel, and reduced carbon footprint. A problem posed by virtual interviews is that candidates are unable to fully learn institutional cultures and social environments of the programs.3 Likewise, telehealth was an important means of clinical teaching for residents during the height of the COVID-19 pandemic, with benefits that included cost-effectiveness and reduction of disparities in access to dermatologic care.4 Seventy-five percent (38/51) of PDs indicated that their program plans to include telemedicine in resident clinical rotation moving forward.
Resources Available—Our survey showed that resources available for residents, delivery of lectures and program time allocated to didactics, protected academic or study time for residents, and allocation of program time for CORE examinations are highly variable across programs. This could inspire future studies to be done to determine the differences in success of the resident on CORE examinations and in digesting material.
Postgraduate Career Plans and Fellowship Matches—Residents of programs that have a home MSDO fellowship are more likely to successfully match into a MSDO fellowship.5 Based on this survey, approximately 28% of graduating residents applied to a fellowship position, with 15%, 5%, and 3% matching into desired MSDO, dermatopathology, and pediatric dermatology fellowships, respectively. Additional studies are needed to determine advantages and disadvantages that lead to residents reaching their career goals.
Limitations—Limitations of this study include a small sample size that may not adequately represent all ACGME-accredited dermatology residency programs and selection bias toward respondents who are more likely to participate in survey-based research.
Conclusion
The APD plans to continue to administer this survey on an annual basis, with updates to the content and questions based on input from PDs. This survey will continue to provide valuable information to drive collaboration among residency programs and optimize the learning experience for residents. Our hope is that the response rate will increase in coming years, allowing us to draw more generalizable conclusions. Nonetheless, the survey data allow individual dermatology residency programs to compare their specific characteristics to other programs.
- Maciejko L, Cope A, Mara K, et al. A national survey of obstetrics and gynecology emergency training and deficits in office emergency preparation [A53]. Obstet Gynecol. 2022;139:16S. doi:10.1097/01.AOG.0000826548.05758.26
- Lavertue SM, Terry R. A comparison of surgical subspecialty match rates in 2022 in the United States. Cureus. 2023;15:E37178. doi:10.7759/cureus.37178
- Domingo A, Rdesinski RE, Stenson A, et al. Virtual residency interviews: applicant perceptions regarding virtual interview effectiveness, advantages, and barriers. J Grad Med Educ. 2022;14:224-228. doi:10.4300/JGME-D-21-00675.1
- Rustad AM, Lio PA. Pandemic pressure: teledermatology and health care disparities. J Patient Exp. 2021;8:2374373521996982. doi:10.1177/2374373521996982
- Rickstrew J, Rajpara A, Hocker TLH. Dermatology residency program influences chance of successful surgery fellowship match. Dermatol Surg. 2021;47:1040-1042. doi:10.1097/DSS.0000000000002859
Educational organizations across several specialties, including internal medicine and obstetrics and gynecology, have formal surveys1; however, the field of dermatology has been without one. This study aimed to establish a formal survey for dermatology program directors (PDs) and clinician-educators. Because the Accreditation Council for Graduate Medical Education (ACGME) and American Board of Dermatology surveys do not capture all metrics relevant to dermatology residency educators, an annual survey for our specialty may be helpful to compare dermatology-specific data among programs. Responses could provide context and perspective to faculty and residents who respond to the ACGME annual survey, as our Association of Professors of Dermatology (APD) survey asks more in-depth questions, such as how often didactics occur and who leads them. Resident commute time and faculty demographics and training also are covered. Current ad hoc surveys disseminated through listserves of various medical associations contain overlapping questions and reflect relatively low response rates; dermatology PDs may benefit from a survey with a high response rate to which they can contribute future questions and topics that reflect recent trends and current needs in graduate medical education. As future surveys are administered, the results can be captured in a centralized database accessible by dermatology PDs.
Methods
A survey of PDs from 141 ACGME-accredited dermatology residency programs was conducted by the Residency Program Director Steering Committee of the APD from November 2022 to January 2023 using a prevalidated questionnaire. Personalized survey links were created and sent individually to each PD’s email listed in the ACGME accreditation data system. All survey responses were captured anonymously, with a number assigned to keep de-identified responses separate and organized. The survey consisted of 137 survey questions addressing topics that included program characteristics, PD demographics, the impact of the COVID-19 pandemic on clinical rotation and educational conferences, available resident resources, quality improvement, clinical and didactic instruction, research content, diversity and inclusion, wellness, professionalism, evaluation systems, and graduate outcomes.
Data were collected using Qualtrics survey tools. After removing duplicate and incomplete surveys, data were analyzed using Qualtrics reports and Microsoft Excel for data plotting, averages, and range calculations.
Results
One hundred forty-one personalized survey links were created and sent individually to each program’s filed email obtained from the APD listserv. Fifty-three responses were recorded after removing duplicate or incomplete surveys (38% [53/141] response rate). As of May 2023, there were 144 ACGME-accredited dermatology residency programs due to 3 newly accredited programs in 2022-2023 academic year, which were not included in our survey population.
Program Characteristics—Forty-four respondents (83%) were from a university-based program. Fifty respondents (94%) were from programs that were ACGME accredited prior to 2020, while 3 programs (6%) were American Osteopathic Association accredited prior to singular accreditation. Seventy-one percent (38/53) of respondents had 1 or more associate PDs.
PD Demographics—Eighty-seven percent (45/52) of PDs who responded to the survey graduated from a US allopathic medical school (MD), 10% (5/52) graduated from a US osteopathic medical school (DO), and 4% (2/52) graduated from an international medical school. Seventy-four percent (35/47) of respondents were White, 17% (8/47) were Asian, and 2% (1/47) were Black or African American; this data was not provided for 4 respondents. Forty-eight percent (23/48) of PDs identified as cisgender man, 48% (23/48) identified as cisgender woman, and 4% (2/48) preferred not to answer. Eighty-one percent (38/47) of PDs identified as heterosexual or straight, 15% (7/47) identified as gay or lesbian, and 4% (2/47) preferred not to answer.
Impact of COVID-19 Pandemic on Residency Training—Due to the COVID-19 pandemic, 88% (45/51) of respondents incorporated telemedicine into the resident clinical rotation schedule. Moving forward, 75% (38/51) of respondents indicated that their programs plan to continue to incorporate telemedicine into the rotation schedule. Based on 50 responses, the average of educational conferences that became virtual at the start of the COVID-19 pandemic was 87%; based on 46 responses, the percentage of educational conferences that will remain virtual moving forward is 46%, while 90% (46/51) of respondents indicated that their programs plan to use virtual conferences in some capacity moving forward. Seventy-three percent (37/51) of respondents indicated that they plan to use virtual interviews as part of residency recruitment moving forward.
Available Resources—Twenty-four percent (11/46) of respondents indicated that residents in their program do not get protected time or time off for CORE examinations. Seventy-five percent (33/44) of PDs said their program provides funding for residents to participate in board review courses. The chief residents at 63% (31/49) of programs receive additional compensation, and 69% (34/49) provide additional administrative time to chief residents. Seventy-one percent (24/34) of PDs reported their programs have scribes for attendings, and 12% (4/34) have scribes for residents. Support staff help residents with callbacks and in-basket messages according to 76% (35/46) of respondents. The majority (98% [45/46]) of PDs indicated that residents follow-up on results and messages from patients seen in resident clinics, and 43% (20/46) of programs have residents follow-up with patients seen in faculty clinics. Only 15% (7/46) of PDs responded they have schedules with residents dedicated to handle these tasks. According to respondents, 33% (17/52) have residents who are required to travel more than 25 miles to distant clinical sites. Of them, 35% (6/17) provide accommodations.
Quality Improvement—Seventy-one percent (35/49) of respondents indicated their department has a quality improvement/patient safety team or committee, and 94% (33/35) of these teams include residents. A lecture series on quality improvement and patient safety is offered at 67% (33/49) of the respondents’ programs, while morbidity and mortality conferences are offered in 73% (36/49).
Clinical Instruction—Our survey asked PDs how many months each residency year spends on a certain rotational service. Based on 46 respondents, the average number of months dedicated to medical dermatology is 7, 5, and 6 months for postgraduate year (PGY) 2, PGY3, and PGY4, respectively. The average number of months spent in other subspecialties is provided in the Table. On average, PGY2 residents spend 8 half-days per week seeing patients in clinic, while PGY3 and PGY4 residents see patients for 7 half-days. The median and mean number of patients staffed by a single attending per hour in teaching clinics are 6 and 5.88, respectively. Respondents indicated that residents participate in the following specialty clinics: pediatric dermatology (96% [44/46]), laser/cosmetic (87% [40/44]), high-risk skin cancer (ie, immunosuppressed/transplant patient)(65% [30/44]), pigmented lesion/melanoma (52% [24/44]), connective tissue disease (52% [24/44]), teledermatology (50% [23/44]), free clinic for homeless and/or indigent populations (48% [22/44]), contact dermatitis (43% [20/44]), skin of color (43% [20/44]), oncodermatology (41% [19/44]), and bullous disease (33% [15/44]).

Additionally, in 87% (40/46) of programs, residents participate in a dedicated inpatient consultation service. Most respondents (98% [45/46]) responded that they utilize in-person consultations with a teledermatology supplement. Fifteen percent (7/46) utilize virtual teledermatology (live video-based consultations), and 57% (26/46) utilize asynchronous teledermatology (picture-based consultations). All respondents (n=46) indicated that 0% to 25% of patient encounters involving residents are teledermatology visits. Thirty-three percent (6/18) of programs have a global health special training track, 56% (10/18) have a Specialty Training and Advanced Research/Physician-Scientist Research Training track, 28% (5/18) have a diversity training track, and 50% (9/18) have a clinician educator training track.
Didactic Instruction—Five programs have a full day per week dedicated to didactics, while 36 programs have at least one half-day per week for didactics. On average, didactics in 57% (26/46) of programs are led by faculty alone, while 43% (20/46) are led at least in part by residents or fellows.

Research Content—Fifty percent (23/46) of programs have a specific research requirement for residents beyond general ACGME requirements, and 35% (16/46) require residents to participate in a longitudinal research project over the course of residency. There is a dedicated research coordinator for resident support at 63% (29/46) of programs. Dedicated biostatistics research support is available for resident projects at 42% (19/45) of programs. Additionally, at 42% (19/45) of programs, there is a dedicated faculty member for oversight of resident research.

Diversity, Equity, and Inclusion—Seventy-three percent (29/40) of programs have special diversity, equity, and inclusion programs or meetings specific to residency, 60% (24/40) have residency initiatives, and 55% (22/40) have a residency diversity committee. Eighty-six percent (42/49) of respondents strongly agreed that their current residents represent diverse ethnic and racial backgrounds (ie, >15% are not White). eTable 1 shows PD responses to this statement, which were stratified based on self-identified race. eTable 2 shows PD responses to the statement, “Our current residents represent an inclusion of gender/sexual orientation,” which were stratified based on self-identified gender identity/sexual orientation. Lastly, eTable 3 highlights the percentage of residents with an MD and DO degree, stratified based on PD degree.

Wellness—Forty-eight percent (20/42) of respondents indicated they are under stress and do not always have as much energy as before becoming a PD but do not feel burned out. Thirty-one percent (13/42) indicated they have 1 or more symptoms of burnout, such as emotional exhaustion. Eighty-six percent (36/42) are satisfied with their jobs overall (43% agree and 43% strongly agree [18/42 each]).
Evaluation System—Seventy-five percent (33/44) of programs deliver evaluations of residents by faculty online, 86% (38/44) of programs have PDs discuss evaluations in-person, and 20% (9/44) of programs have faculty evaluators discuss evaluations in-person. Seventy-seven percent (34/44) of programs have formal faculty-resident mentor-mentee programs. Clinical competency committee chair positions are filled by PDs, assistant PDs, or core faculty members 47%, 38%, and 16% of the time, respectively.
Graduation Outcomes of PGY4 Residents—About 28% (55/199) of graduating residents applied to a fellowship position, with the majority (15% [29/55]) matching into Mohs micrographic surgery and dermatologic oncology (MSDO) fellowships. Approximately 5% (9/199) and 4% (7/199) of graduates matched into dermatopathology and pediatric dermatology, respectively. The remaining 5% (10/199) of graduating residents applied to a fellowship but did not match. The majority (45% [91/199]) of residency graduates entered private practice after graduation. Approximately 21% (42/199) of graduating residents chose an academic practice with 17% (33/199), 2% (4/199), and 2% (3/199) of those positions being full-time, part-time, and adjunct, respectively.
Comment
The first annual APD survey is a novel data source and provides opportunities for areas of discussion and investigation. Evaluating the similarities and differences among dermatology residency programs across the United States can strengthen individual programs through collaboration and provide areas of cohesion among programs.
Diversity of PDs—An important area of discussion is diversity and PD demographics. Although DO students make up 1 in 4 US graduating medical students, they are not interviewed or ranked as often as MD students.2 Diversity in PD race and ethnicity may be worthy of investigation in future studies, as match rates and recruitment of diverse medical school applicants may be impacted by these demographics.
Continued Use of Telemedicine in Training—Since 2020, the benefits of virtual residency recruitment have been debated among PDs across all medical specialties. Points in favor of virtual interviews include cost savings for programs and especially for applicants, as well as time efficiency, reduced burden of travel, and reduced carbon footprint. A problem posed by virtual interviews is that candidates are unable to fully learn institutional cultures and social environments of the programs.3 Likewise, telehealth was an important means of clinical teaching for residents during the height of the COVID-19 pandemic, with benefits that included cost-effectiveness and reduction of disparities in access to dermatologic care.4 Seventy-five percent (38/51) of PDs indicated that their program plans to include telemedicine in resident clinical rotation moving forward.
Resources Available—Our survey showed that resources available for residents, delivery of lectures and program time allocated to didactics, protected academic or study time for residents, and allocation of program time for CORE examinations are highly variable across programs. This could inspire future studies to be done to determine the differences in success of the resident on CORE examinations and in digesting material.
Postgraduate Career Plans and Fellowship Matches—Residents of programs that have a home MSDO fellowship are more likely to successfully match into a MSDO fellowship.5 Based on this survey, approximately 28% of graduating residents applied to a fellowship position, with 15%, 5%, and 3% matching into desired MSDO, dermatopathology, and pediatric dermatology fellowships, respectively. Additional studies are needed to determine advantages and disadvantages that lead to residents reaching their career goals.
Limitations—Limitations of this study include a small sample size that may not adequately represent all ACGME-accredited dermatology residency programs and selection bias toward respondents who are more likely to participate in survey-based research.
Conclusion
The APD plans to continue to administer this survey on an annual basis, with updates to the content and questions based on input from PDs. This survey will continue to provide valuable information to drive collaboration among residency programs and optimize the learning experience for residents. Our hope is that the response rate will increase in coming years, allowing us to draw more generalizable conclusions. Nonetheless, the survey data allow individual dermatology residency programs to compare their specific characteristics to other programs.
Educational organizations across several specialties, including internal medicine and obstetrics and gynecology, have formal surveys1; however, the field of dermatology has been without one. This study aimed to establish a formal survey for dermatology program directors (PDs) and clinician-educators. Because the Accreditation Council for Graduate Medical Education (ACGME) and American Board of Dermatology surveys do not capture all metrics relevant to dermatology residency educators, an annual survey for our specialty may be helpful to compare dermatology-specific data among programs. Responses could provide context and perspective to faculty and residents who respond to the ACGME annual survey, as our Association of Professors of Dermatology (APD) survey asks more in-depth questions, such as how often didactics occur and who leads them. Resident commute time and faculty demographics and training also are covered. Current ad hoc surveys disseminated through listserves of various medical associations contain overlapping questions and reflect relatively low response rates; dermatology PDs may benefit from a survey with a high response rate to which they can contribute future questions and topics that reflect recent trends and current needs in graduate medical education. As future surveys are administered, the results can be captured in a centralized database accessible by dermatology PDs.
Methods
A survey of PDs from 141 ACGME-accredited dermatology residency programs was conducted by the Residency Program Director Steering Committee of the APD from November 2022 to January 2023 using a prevalidated questionnaire. Personalized survey links were created and sent individually to each PD’s email listed in the ACGME accreditation data system. All survey responses were captured anonymously, with a number assigned to keep de-identified responses separate and organized. The survey consisted of 137 survey questions addressing topics that included program characteristics, PD demographics, the impact of the COVID-19 pandemic on clinical rotation and educational conferences, available resident resources, quality improvement, clinical and didactic instruction, research content, diversity and inclusion, wellness, professionalism, evaluation systems, and graduate outcomes.
Data were collected using Qualtrics survey tools. After removing duplicate and incomplete surveys, data were analyzed using Qualtrics reports and Microsoft Excel for data plotting, averages, and range calculations.
Results
One hundred forty-one personalized survey links were created and sent individually to each program’s filed email obtained from the APD listserv. Fifty-three responses were recorded after removing duplicate or incomplete surveys (38% [53/141] response rate). As of May 2023, there were 144 ACGME-accredited dermatology residency programs due to 3 newly accredited programs in 2022-2023 academic year, which were not included in our survey population.
Program Characteristics—Forty-four respondents (83%) were from a university-based program. Fifty respondents (94%) were from programs that were ACGME accredited prior to 2020, while 3 programs (6%) were American Osteopathic Association accredited prior to singular accreditation. Seventy-one percent (38/53) of respondents had 1 or more associate PDs.
PD Demographics—Eighty-seven percent (45/52) of PDs who responded to the survey graduated from a US allopathic medical school (MD), 10% (5/52) graduated from a US osteopathic medical school (DO), and 4% (2/52) graduated from an international medical school. Seventy-four percent (35/47) of respondents were White, 17% (8/47) were Asian, and 2% (1/47) were Black or African American; this data was not provided for 4 respondents. Forty-eight percent (23/48) of PDs identified as cisgender man, 48% (23/48) identified as cisgender woman, and 4% (2/48) preferred not to answer. Eighty-one percent (38/47) of PDs identified as heterosexual or straight, 15% (7/47) identified as gay or lesbian, and 4% (2/47) preferred not to answer.
Impact of COVID-19 Pandemic on Residency Training—Due to the COVID-19 pandemic, 88% (45/51) of respondents incorporated telemedicine into the resident clinical rotation schedule. Moving forward, 75% (38/51) of respondents indicated that their programs plan to continue to incorporate telemedicine into the rotation schedule. Based on 50 responses, the average of educational conferences that became virtual at the start of the COVID-19 pandemic was 87%; based on 46 responses, the percentage of educational conferences that will remain virtual moving forward is 46%, while 90% (46/51) of respondents indicated that their programs plan to use virtual conferences in some capacity moving forward. Seventy-three percent (37/51) of respondents indicated that they plan to use virtual interviews as part of residency recruitment moving forward.
Available Resources—Twenty-four percent (11/46) of respondents indicated that residents in their program do not get protected time or time off for CORE examinations. Seventy-five percent (33/44) of PDs said their program provides funding for residents to participate in board review courses. The chief residents at 63% (31/49) of programs receive additional compensation, and 69% (34/49) provide additional administrative time to chief residents. Seventy-one percent (24/34) of PDs reported their programs have scribes for attendings, and 12% (4/34) have scribes for residents. Support staff help residents with callbacks and in-basket messages according to 76% (35/46) of respondents. The majority (98% [45/46]) of PDs indicated that residents follow-up on results and messages from patients seen in resident clinics, and 43% (20/46) of programs have residents follow-up with patients seen in faculty clinics. Only 15% (7/46) of PDs responded they have schedules with residents dedicated to handle these tasks. According to respondents, 33% (17/52) have residents who are required to travel more than 25 miles to distant clinical sites. Of them, 35% (6/17) provide accommodations.
Quality Improvement—Seventy-one percent (35/49) of respondents indicated their department has a quality improvement/patient safety team or committee, and 94% (33/35) of these teams include residents. A lecture series on quality improvement and patient safety is offered at 67% (33/49) of the respondents’ programs, while morbidity and mortality conferences are offered in 73% (36/49).
Clinical Instruction—Our survey asked PDs how many months each residency year spends on a certain rotational service. Based on 46 respondents, the average number of months dedicated to medical dermatology is 7, 5, and 6 months for postgraduate year (PGY) 2, PGY3, and PGY4, respectively. The average number of months spent in other subspecialties is provided in the Table. On average, PGY2 residents spend 8 half-days per week seeing patients in clinic, while PGY3 and PGY4 residents see patients for 7 half-days. The median and mean number of patients staffed by a single attending per hour in teaching clinics are 6 and 5.88, respectively. Respondents indicated that residents participate in the following specialty clinics: pediatric dermatology (96% [44/46]), laser/cosmetic (87% [40/44]), high-risk skin cancer (ie, immunosuppressed/transplant patient)(65% [30/44]), pigmented lesion/melanoma (52% [24/44]), connective tissue disease (52% [24/44]), teledermatology (50% [23/44]), free clinic for homeless and/or indigent populations (48% [22/44]), contact dermatitis (43% [20/44]), skin of color (43% [20/44]), oncodermatology (41% [19/44]), and bullous disease (33% [15/44]).

Additionally, in 87% (40/46) of programs, residents participate in a dedicated inpatient consultation service. Most respondents (98% [45/46]) responded that they utilize in-person consultations with a teledermatology supplement. Fifteen percent (7/46) utilize virtual teledermatology (live video-based consultations), and 57% (26/46) utilize asynchronous teledermatology (picture-based consultations). All respondents (n=46) indicated that 0% to 25% of patient encounters involving residents are teledermatology visits. Thirty-three percent (6/18) of programs have a global health special training track, 56% (10/18) have a Specialty Training and Advanced Research/Physician-Scientist Research Training track, 28% (5/18) have a diversity training track, and 50% (9/18) have a clinician educator training track.
Didactic Instruction—Five programs have a full day per week dedicated to didactics, while 36 programs have at least one half-day per week for didactics. On average, didactics in 57% (26/46) of programs are led by faculty alone, while 43% (20/46) are led at least in part by residents or fellows.

Research Content—Fifty percent (23/46) of programs have a specific research requirement for residents beyond general ACGME requirements, and 35% (16/46) require residents to participate in a longitudinal research project over the course of residency. There is a dedicated research coordinator for resident support at 63% (29/46) of programs. Dedicated biostatistics research support is available for resident projects at 42% (19/45) of programs. Additionally, at 42% (19/45) of programs, there is a dedicated faculty member for oversight of resident research.

Diversity, Equity, and Inclusion—Seventy-three percent (29/40) of programs have special diversity, equity, and inclusion programs or meetings specific to residency, 60% (24/40) have residency initiatives, and 55% (22/40) have a residency diversity committee. Eighty-six percent (42/49) of respondents strongly agreed that their current residents represent diverse ethnic and racial backgrounds (ie, >15% are not White). eTable 1 shows PD responses to this statement, which were stratified based on self-identified race. eTable 2 shows PD responses to the statement, “Our current residents represent an inclusion of gender/sexual orientation,” which were stratified based on self-identified gender identity/sexual orientation. Lastly, eTable 3 highlights the percentage of residents with an MD and DO degree, stratified based on PD degree.

Wellness—Forty-eight percent (20/42) of respondents indicated they are under stress and do not always have as much energy as before becoming a PD but do not feel burned out. Thirty-one percent (13/42) indicated they have 1 or more symptoms of burnout, such as emotional exhaustion. Eighty-six percent (36/42) are satisfied with their jobs overall (43% agree and 43% strongly agree [18/42 each]).
Evaluation System—Seventy-five percent (33/44) of programs deliver evaluations of residents by faculty online, 86% (38/44) of programs have PDs discuss evaluations in-person, and 20% (9/44) of programs have faculty evaluators discuss evaluations in-person. Seventy-seven percent (34/44) of programs have formal faculty-resident mentor-mentee programs. Clinical competency committee chair positions are filled by PDs, assistant PDs, or core faculty members 47%, 38%, and 16% of the time, respectively.
Graduation Outcomes of PGY4 Residents—About 28% (55/199) of graduating residents applied to a fellowship position, with the majority (15% [29/55]) matching into Mohs micrographic surgery and dermatologic oncology (MSDO) fellowships. Approximately 5% (9/199) and 4% (7/199) of graduates matched into dermatopathology and pediatric dermatology, respectively. The remaining 5% (10/199) of graduating residents applied to a fellowship but did not match. The majority (45% [91/199]) of residency graduates entered private practice after graduation. Approximately 21% (42/199) of graduating residents chose an academic practice with 17% (33/199), 2% (4/199), and 2% (3/199) of those positions being full-time, part-time, and adjunct, respectively.
Comment
The first annual APD survey is a novel data source and provides opportunities for areas of discussion and investigation. Evaluating the similarities and differences among dermatology residency programs across the United States can strengthen individual programs through collaboration and provide areas of cohesion among programs.
Diversity of PDs—An important area of discussion is diversity and PD demographics. Although DO students make up 1 in 4 US graduating medical students, they are not interviewed or ranked as often as MD students.2 Diversity in PD race and ethnicity may be worthy of investigation in future studies, as match rates and recruitment of diverse medical school applicants may be impacted by these demographics.
Continued Use of Telemedicine in Training—Since 2020, the benefits of virtual residency recruitment have been debated among PDs across all medical specialties. Points in favor of virtual interviews include cost savings for programs and especially for applicants, as well as time efficiency, reduced burden of travel, and reduced carbon footprint. A problem posed by virtual interviews is that candidates are unable to fully learn institutional cultures and social environments of the programs.3 Likewise, telehealth was an important means of clinical teaching for residents during the height of the COVID-19 pandemic, with benefits that included cost-effectiveness and reduction of disparities in access to dermatologic care.4 Seventy-five percent (38/51) of PDs indicated that their program plans to include telemedicine in resident clinical rotation moving forward.
Resources Available—Our survey showed that resources available for residents, delivery of lectures and program time allocated to didactics, protected academic or study time for residents, and allocation of program time for CORE examinations are highly variable across programs. This could inspire future studies to be done to determine the differences in success of the resident on CORE examinations and in digesting material.
Postgraduate Career Plans and Fellowship Matches—Residents of programs that have a home MSDO fellowship are more likely to successfully match into a MSDO fellowship.5 Based on this survey, approximately 28% of graduating residents applied to a fellowship position, with 15%, 5%, and 3% matching into desired MSDO, dermatopathology, and pediatric dermatology fellowships, respectively. Additional studies are needed to determine advantages and disadvantages that lead to residents reaching their career goals.
Limitations—Limitations of this study include a small sample size that may not adequately represent all ACGME-accredited dermatology residency programs and selection bias toward respondents who are more likely to participate in survey-based research.
Conclusion
The APD plans to continue to administer this survey on an annual basis, with updates to the content and questions based on input from PDs. This survey will continue to provide valuable information to drive collaboration among residency programs and optimize the learning experience for residents. Our hope is that the response rate will increase in coming years, allowing us to draw more generalizable conclusions. Nonetheless, the survey data allow individual dermatology residency programs to compare their specific characteristics to other programs.
- Maciejko L, Cope A, Mara K, et al. A national survey of obstetrics and gynecology emergency training and deficits in office emergency preparation [A53]. Obstet Gynecol. 2022;139:16S. doi:10.1097/01.AOG.0000826548.05758.26
- Lavertue SM, Terry R. A comparison of surgical subspecialty match rates in 2022 in the United States. Cureus. 2023;15:E37178. doi:10.7759/cureus.37178
- Domingo A, Rdesinski RE, Stenson A, et al. Virtual residency interviews: applicant perceptions regarding virtual interview effectiveness, advantages, and barriers. J Grad Med Educ. 2022;14:224-228. doi:10.4300/JGME-D-21-00675.1
- Rustad AM, Lio PA. Pandemic pressure: teledermatology and health care disparities. J Patient Exp. 2021;8:2374373521996982. doi:10.1177/2374373521996982
- Rickstrew J, Rajpara A, Hocker TLH. Dermatology residency program influences chance of successful surgery fellowship match. Dermatol Surg. 2021;47:1040-1042. doi:10.1097/DSS.0000000000002859
- Maciejko L, Cope A, Mara K, et al. A national survey of obstetrics and gynecology emergency training and deficits in office emergency preparation [A53]. Obstet Gynecol. 2022;139:16S. doi:10.1097/01.AOG.0000826548.05758.26
- Lavertue SM, Terry R. A comparison of surgical subspecialty match rates in 2022 in the United States. Cureus. 2023;15:E37178. doi:10.7759/cureus.37178
- Domingo A, Rdesinski RE, Stenson A, et al. Virtual residency interviews: applicant perceptions regarding virtual interview effectiveness, advantages, and barriers. J Grad Med Educ. 2022;14:224-228. doi:10.4300/JGME-D-21-00675.1
- Rustad AM, Lio PA. Pandemic pressure: teledermatology and health care disparities. J Patient Exp. 2021;8:2374373521996982. doi:10.1177/2374373521996982
- Rickstrew J, Rajpara A, Hocker TLH. Dermatology residency program influences chance of successful surgery fellowship match. Dermatol Surg. 2021;47:1040-1042. doi:10.1097/DSS.0000000000002859
Practice Points
- The first annual Association of Professors of Dermatology program directors survey allows faculty to compare their programs to other dermatology residency programs across the United States.
- The results should inspire opportunities for growth, improvement, and collaboration among dermatology residency programs.
Focal Palmoplantar Keratoderma and Gingival Keratosis Caused by a KRT16 Mutation
To the Editor:
Focal palmoplantar keratoderma and gingival keratosis (FPGK)(Online Mendelian Inheritance in Man [OMIM] 148730) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma and gingival hyperkeratosis presenting as leukokeratosis. Focal palmoplantar keratoderma and gingival keratosis was first defined by Gorlin1 in 1976. Since then, only a few cases have been reported, but no causative mutations have been identified.2
Focal pressure-related palmoplantar keratoderma (PPK) and oral hyperkeratosis also are seen in pachyonychia congenita (PC)(OMIM 167200, 615726, 615728, 167210), a rare autosomal-dominant disorder of keratinization characterized by PPK and nail dystrophy. Patients with PC often present with plantar pain; more variable features include oral leukokeratosis, follicular hyperkeratosis, pilosebaceous and epidermal inclusion cysts, hoarseness, hyperhidrosis, and natal teeth. Pachyonychia congenita is caused by mutation in keratin genes KRT6A, KRT6B, KRT16, or KRT17.
Focal palmoplantar keratoderma and gingival keratosis as well as PC are distinct from other forms of PPK with gingival involvement such as
Despite the common features of FPGK and PC, they are considered distinct disorders due to absence of nail changes in FPGK and no prior evidence of a common genetic cause. We present a patient with familial FPGK found by whole exome sequencing to be caused by a mutation in KRT16.
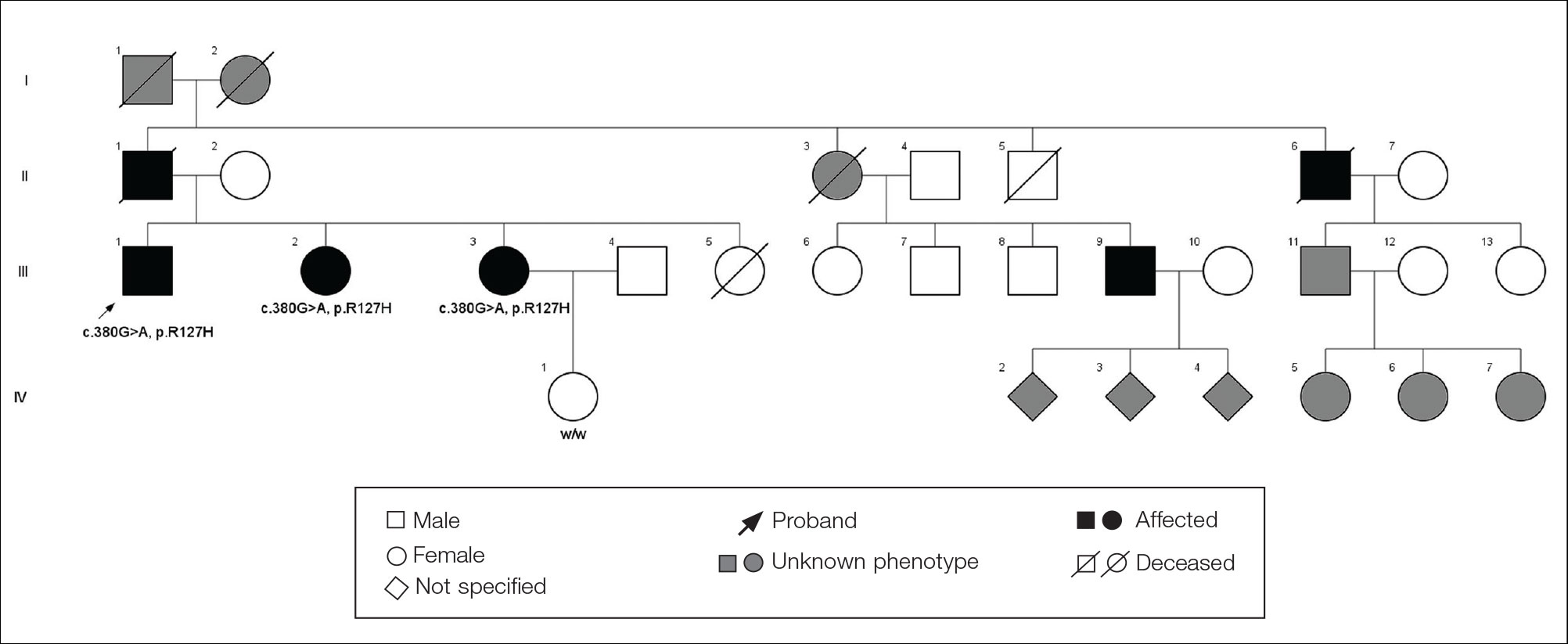
The proband was a 57-year-old man born to unrelated parents (Figure 1). He had no skin problems at birth, and his development was normal. He had painful focal keratoderma since childhood that were most prominent at pressure points on the soles and toes (Figure 2A), in addition to gingival hyperkeratosis and oral leukokeratosis (Figure 2B). He had no associated abnormalities of the skin, hair, or teeth and no nail findings (Figure 2C). He reported that his father and 2 of his 3 sisters were affected with similar symptoms. A punch biopsy of the right fifth toe was consistent with verrucous epidermal hyperplasia with perinuclear keratinization in the spinous layer (Figure 3A). A gingival biopsy showed perinuclear eosinophilic globules and basophilic stranding in the cytoplasm (Figure 3B). His older sister had more severe and painful focal keratoderma of the soles, punctate keratoderma of the palms, gingival hyperkeratosis, and leukokeratosis of the tongue.
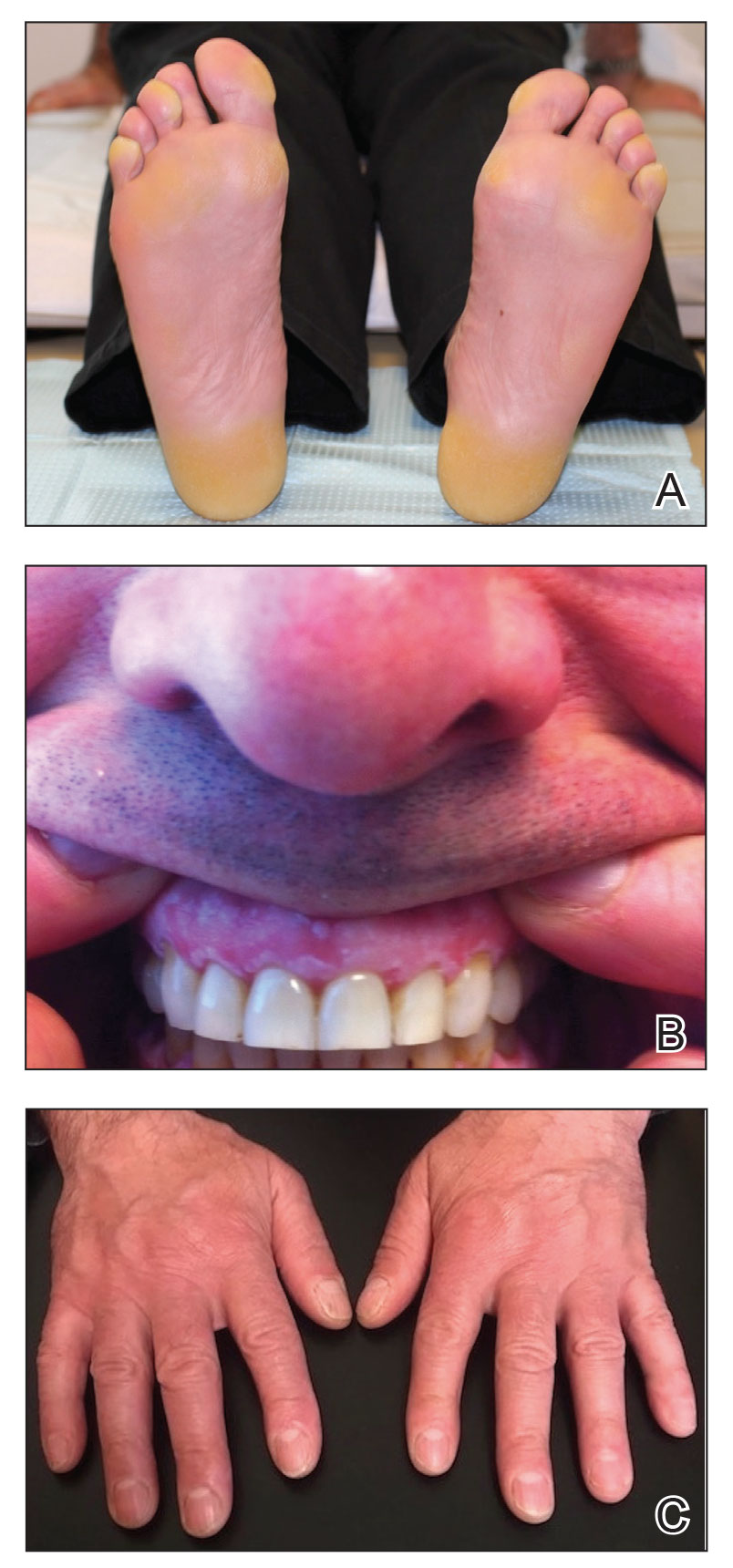
Whole exome sequencing of the proband revealed a heterozygous missense mutation in KRT16 (c.380G>A, p.R127H, rs57424749). Sanger sequencing confirmed this mutation and showed that it was heterozygous in both of his affected sisters and absent in his unaffected niece (Figure 1). The patient was treated with topical and systemic retinoids, keratolytics, and mechanical removal to moderate effect, with noted improvement in the appearance and associated pain of the plantar keratoderma.
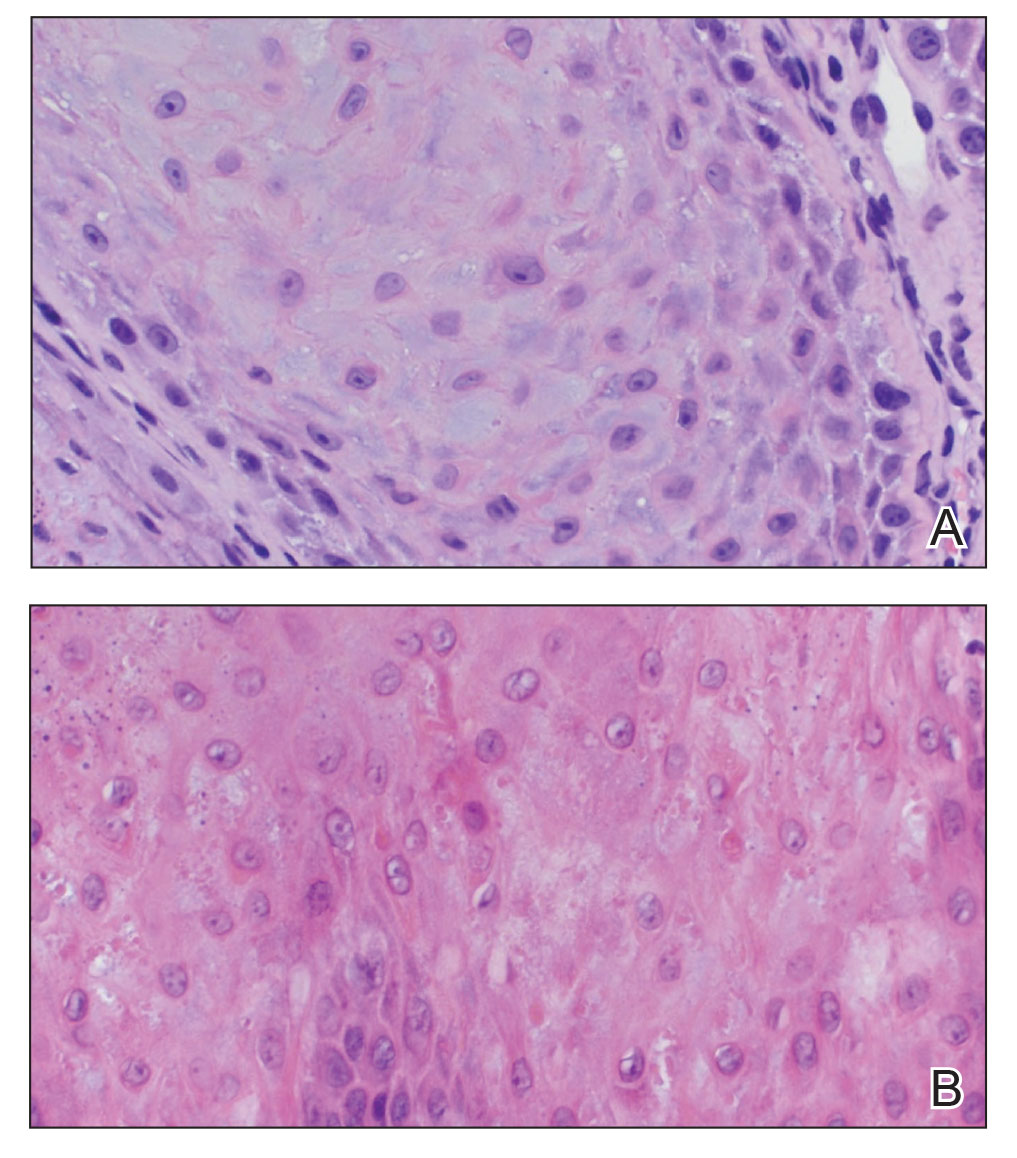
Phenotypic heterogeneity is common in PC, though PC due to KRT6A mutations demonstrates more severe nail disease with oral lesions, cysts, and follicular hyperkeratosis, while PC caused by KRT16 mutations generally presents with more extensive and painful PPK.4KRT16 mutations affecting p.R127 are frequent causes of PC, and genotype-phenotype correlations have been observed. Individuals with p.R127P mutations exhibit more severe disease with earlier age of onset, more extensive nail involvement and oral leukokeratosis, and greater impact on daily quality of life than in individuals with p.R127C mutations.5 Cases of PC with KRT16 p.R127S and p.R127G mutations also have been observed. The KRT16 c.380G>A, p.R127H mutation we documented has been reported in one kindred with PC who presented with PPK, oral leukokeratosis, toenail thickening, and pilosebaceous and follicular hyperkeratosis.6
Although patients with FPGK lack the thickening of fingernails and/or toenails considered a defining feature of PC, the disorders otherwise are phenotypically similar, suggesting the possibility of common pathogenesis. One linkage study of familial FPGK excluded genetic intervals containing type I and type II keratins but was limited to a single small kindred.2 This study and our data together suggest that, similar to PC, there are multiple genes in which mutations cause FPGK.
Murine Krt16 knockouts show distinct phenotypes depending on the mouse strain in which they are propagated, ranging from perinatal lethality to differences in the severity of oral and PPK lesions.7 These observations provide evidence that additional genetic variants contribute to Krt16 phenotypes in mice and suggest the same could be true for humans.
We propose that some cases of FPGK are due to mutations in KRT16 and thus share a genetic pathogenesis with PC, underscoring the utility of whole exome sequencing in providing genetic diagnoses for disorders that are genetically and clinically heterogeneous. Further biologic investigation of phenotypes caused by KRT16 mutation may reveal respective contributions of additional genetic variation and environmental effects to the variable clinical presentations.
- Gorlin RJ. Focal palmoplantar and marginal gingival hyperkeratosis—a syndrome. Birth Defects Orig Artic Ser. 1976;12:239-242.
- Kolde G, Hennies HC, Bethke G, et al. Focal palmoplantar and gingival keratosis: a distinct palmoplantar ectodermal dysplasia with epidermolytic alterations but lack of mutations in known keratins. J Am Acad Dermatol. 2005;52(3 pt 1):403-409.
- Duchatelet S, Hovnanian A. Olmsted syndrome: clinical, molecular and therapeutic aspects. Orphanet J Rare Dis. 2015;10:33.
- Spaunhurst KM, Hogendorf AM, Smith FJ, et al. Pachyonychia congenita patients with mutations in KRT6A have more extensive disease compared with patients who have mutations in KRT16. Br J Dermatol. 2012;166:875-878.
- Fu T, Leachman SA, Wilson NJ, et al. Genotype-phenotype correlations among pachyonychia congenita patients with K16 mutations. J Invest Dermatol. 2011;131:1025-1028.
- Wilson NJ, O’Toole EA, Milstone LM, et al. The molecular genetic analysis of the expanding pachyonychia congenita case collection. Br J Dermatol. 2014;171:343-355.
- Zieman A, Coulombe PA. The keratin 16 null phenotype is modestly impacted by genetic strain background in mice. Exp Dermatol. 2018;27:672-674.
To the Editor:
Focal palmoplantar keratoderma and gingival keratosis (FPGK)(Online Mendelian Inheritance in Man [OMIM] 148730) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma and gingival hyperkeratosis presenting as leukokeratosis. Focal palmoplantar keratoderma and gingival keratosis was first defined by Gorlin1 in 1976. Since then, only a few cases have been reported, but no causative mutations have been identified.2
Focal pressure-related palmoplantar keratoderma (PPK) and oral hyperkeratosis also are seen in pachyonychia congenita (PC)(OMIM 167200, 615726, 615728, 167210), a rare autosomal-dominant disorder of keratinization characterized by PPK and nail dystrophy. Patients with PC often present with plantar pain; more variable features include oral leukokeratosis, follicular hyperkeratosis, pilosebaceous and epidermal inclusion cysts, hoarseness, hyperhidrosis, and natal teeth. Pachyonychia congenita is caused by mutation in keratin genes KRT6A, KRT6B, KRT16, or KRT17.
Focal palmoplantar keratoderma and gingival keratosis as well as PC are distinct from other forms of PPK with gingival involvement such as
Despite the common features of FPGK and PC, they are considered distinct disorders due to absence of nail changes in FPGK and no prior evidence of a common genetic cause. We present a patient with familial FPGK found by whole exome sequencing to be caused by a mutation in KRT16.

The proband was a 57-year-old man born to unrelated parents (Figure 1). He had no skin problems at birth, and his development was normal. He had painful focal keratoderma since childhood that were most prominent at pressure points on the soles and toes (Figure 2A), in addition to gingival hyperkeratosis and oral leukokeratosis (Figure 2B). He had no associated abnormalities of the skin, hair, or teeth and no nail findings (Figure 2C). He reported that his father and 2 of his 3 sisters were affected with similar symptoms. A punch biopsy of the right fifth toe was consistent with verrucous epidermal hyperplasia with perinuclear keratinization in the spinous layer (Figure 3A). A gingival biopsy showed perinuclear eosinophilic globules and basophilic stranding in the cytoplasm (Figure 3B). His older sister had more severe and painful focal keratoderma of the soles, punctate keratoderma of the palms, gingival hyperkeratosis, and leukokeratosis of the tongue.

Whole exome sequencing of the proband revealed a heterozygous missense mutation in KRT16 (c.380G>A, p.R127H, rs57424749). Sanger sequencing confirmed this mutation and showed that it was heterozygous in both of his affected sisters and absent in his unaffected niece (Figure 1). The patient was treated with topical and systemic retinoids, keratolytics, and mechanical removal to moderate effect, with noted improvement in the appearance and associated pain of the plantar keratoderma.

Phenotypic heterogeneity is common in PC, though PC due to KRT6A mutations demonstrates more severe nail disease with oral lesions, cysts, and follicular hyperkeratosis, while PC caused by KRT16 mutations generally presents with more extensive and painful PPK.4KRT16 mutations affecting p.R127 are frequent causes of PC, and genotype-phenotype correlations have been observed. Individuals with p.R127P mutations exhibit more severe disease with earlier age of onset, more extensive nail involvement and oral leukokeratosis, and greater impact on daily quality of life than in individuals with p.R127C mutations.5 Cases of PC with KRT16 p.R127S and p.R127G mutations also have been observed. The KRT16 c.380G>A, p.R127H mutation we documented has been reported in one kindred with PC who presented with PPK, oral leukokeratosis, toenail thickening, and pilosebaceous and follicular hyperkeratosis.6
Although patients with FPGK lack the thickening of fingernails and/or toenails considered a defining feature of PC, the disorders otherwise are phenotypically similar, suggesting the possibility of common pathogenesis. One linkage study of familial FPGK excluded genetic intervals containing type I and type II keratins but was limited to a single small kindred.2 This study and our data together suggest that, similar to PC, there are multiple genes in which mutations cause FPGK.
Murine Krt16 knockouts show distinct phenotypes depending on the mouse strain in which they are propagated, ranging from perinatal lethality to differences in the severity of oral and PPK lesions.7 These observations provide evidence that additional genetic variants contribute to Krt16 phenotypes in mice and suggest the same could be true for humans.
We propose that some cases of FPGK are due to mutations in KRT16 and thus share a genetic pathogenesis with PC, underscoring the utility of whole exome sequencing in providing genetic diagnoses for disorders that are genetically and clinically heterogeneous. Further biologic investigation of phenotypes caused by KRT16 mutation may reveal respective contributions of additional genetic variation and environmental effects to the variable clinical presentations.
To the Editor:
Focal palmoplantar keratoderma and gingival keratosis (FPGK)(Online Mendelian Inheritance in Man [OMIM] 148730) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma and gingival hyperkeratosis presenting as leukokeratosis. Focal palmoplantar keratoderma and gingival keratosis was first defined by Gorlin1 in 1976. Since then, only a few cases have been reported, but no causative mutations have been identified.2
Focal pressure-related palmoplantar keratoderma (PPK) and oral hyperkeratosis also are seen in pachyonychia congenita (PC)(OMIM 167200, 615726, 615728, 167210), a rare autosomal-dominant disorder of keratinization characterized by PPK and nail dystrophy. Patients with PC often present with plantar pain; more variable features include oral leukokeratosis, follicular hyperkeratosis, pilosebaceous and epidermal inclusion cysts, hoarseness, hyperhidrosis, and natal teeth. Pachyonychia congenita is caused by mutation in keratin genes KRT6A, KRT6B, KRT16, or KRT17.
Focal palmoplantar keratoderma and gingival keratosis as well as PC are distinct from other forms of PPK with gingival involvement such as
Despite the common features of FPGK and PC, they are considered distinct disorders due to absence of nail changes in FPGK and no prior evidence of a common genetic cause. We present a patient with familial FPGK found by whole exome sequencing to be caused by a mutation in KRT16.

The proband was a 57-year-old man born to unrelated parents (Figure 1). He had no skin problems at birth, and his development was normal. He had painful focal keratoderma since childhood that were most prominent at pressure points on the soles and toes (Figure 2A), in addition to gingival hyperkeratosis and oral leukokeratosis (Figure 2B). He had no associated abnormalities of the skin, hair, or teeth and no nail findings (Figure 2C). He reported that his father and 2 of his 3 sisters were affected with similar symptoms. A punch biopsy of the right fifth toe was consistent with verrucous epidermal hyperplasia with perinuclear keratinization in the spinous layer (Figure 3A). A gingival biopsy showed perinuclear eosinophilic globules and basophilic stranding in the cytoplasm (Figure 3B). His older sister had more severe and painful focal keratoderma of the soles, punctate keratoderma of the palms, gingival hyperkeratosis, and leukokeratosis of the tongue.

Whole exome sequencing of the proband revealed a heterozygous missense mutation in KRT16 (c.380G>A, p.R127H, rs57424749). Sanger sequencing confirmed this mutation and showed that it was heterozygous in both of his affected sisters and absent in his unaffected niece (Figure 1). The patient was treated with topical and systemic retinoids, keratolytics, and mechanical removal to moderate effect, with noted improvement in the appearance and associated pain of the plantar keratoderma.

Phenotypic heterogeneity is common in PC, though PC due to KRT6A mutations demonstrates more severe nail disease with oral lesions, cysts, and follicular hyperkeratosis, while PC caused by KRT16 mutations generally presents with more extensive and painful PPK.4KRT16 mutations affecting p.R127 are frequent causes of PC, and genotype-phenotype correlations have been observed. Individuals with p.R127P mutations exhibit more severe disease with earlier age of onset, more extensive nail involvement and oral leukokeratosis, and greater impact on daily quality of life than in individuals with p.R127C mutations.5 Cases of PC with KRT16 p.R127S and p.R127G mutations also have been observed. The KRT16 c.380G>A, p.R127H mutation we documented has been reported in one kindred with PC who presented with PPK, oral leukokeratosis, toenail thickening, and pilosebaceous and follicular hyperkeratosis.6
Although patients with FPGK lack the thickening of fingernails and/or toenails considered a defining feature of PC, the disorders otherwise are phenotypically similar, suggesting the possibility of common pathogenesis. One linkage study of familial FPGK excluded genetic intervals containing type I and type II keratins but was limited to a single small kindred.2 This study and our data together suggest that, similar to PC, there are multiple genes in which mutations cause FPGK.
Murine Krt16 knockouts show distinct phenotypes depending on the mouse strain in which they are propagated, ranging from perinatal lethality to differences in the severity of oral and PPK lesions.7 These observations provide evidence that additional genetic variants contribute to Krt16 phenotypes in mice and suggest the same could be true for humans.
We propose that some cases of FPGK are due to mutations in KRT16 and thus share a genetic pathogenesis with PC, underscoring the utility of whole exome sequencing in providing genetic diagnoses for disorders that are genetically and clinically heterogeneous. Further biologic investigation of phenotypes caused by KRT16 mutation may reveal respective contributions of additional genetic variation and environmental effects to the variable clinical presentations.
- Gorlin RJ. Focal palmoplantar and marginal gingival hyperkeratosis—a syndrome. Birth Defects Orig Artic Ser. 1976;12:239-242.
- Kolde G, Hennies HC, Bethke G, et al. Focal palmoplantar and gingival keratosis: a distinct palmoplantar ectodermal dysplasia with epidermolytic alterations but lack of mutations in known keratins. J Am Acad Dermatol. 2005;52(3 pt 1):403-409.
- Duchatelet S, Hovnanian A. Olmsted syndrome: clinical, molecular and therapeutic aspects. Orphanet J Rare Dis. 2015;10:33.
- Spaunhurst KM, Hogendorf AM, Smith FJ, et al. Pachyonychia congenita patients with mutations in KRT6A have more extensive disease compared with patients who have mutations in KRT16. Br J Dermatol. 2012;166:875-878.
- Fu T, Leachman SA, Wilson NJ, et al. Genotype-phenotype correlations among pachyonychia congenita patients with K16 mutations. J Invest Dermatol. 2011;131:1025-1028.
- Wilson NJ, O’Toole EA, Milstone LM, et al. The molecular genetic analysis of the expanding pachyonychia congenita case collection. Br J Dermatol. 2014;171:343-355.
- Zieman A, Coulombe PA. The keratin 16 null phenotype is modestly impacted by genetic strain background in mice. Exp Dermatol. 2018;27:672-674.
- Gorlin RJ. Focal palmoplantar and marginal gingival hyperkeratosis—a syndrome. Birth Defects Orig Artic Ser. 1976;12:239-242.
- Kolde G, Hennies HC, Bethke G, et al. Focal palmoplantar and gingival keratosis: a distinct palmoplantar ectodermal dysplasia with epidermolytic alterations but lack of mutations in known keratins. J Am Acad Dermatol. 2005;52(3 pt 1):403-409.
- Duchatelet S, Hovnanian A. Olmsted syndrome: clinical, molecular and therapeutic aspects. Orphanet J Rare Dis. 2015;10:33.
- Spaunhurst KM, Hogendorf AM, Smith FJ, et al. Pachyonychia congenita patients with mutations in KRT6A have more extensive disease compared with patients who have mutations in KRT16. Br J Dermatol. 2012;166:875-878.
- Fu T, Leachman SA, Wilson NJ, et al. Genotype-phenotype correlations among pachyonychia congenita patients with K16 mutations. J Invest Dermatol. 2011;131:1025-1028.
- Wilson NJ, O’Toole EA, Milstone LM, et al. The molecular genetic analysis of the expanding pachyonychia congenita case collection. Br J Dermatol. 2014;171:343-355.
- Zieman A, Coulombe PA. The keratin 16 null phenotype is modestly impacted by genetic strain background in mice. Exp Dermatol. 2018;27:672-674.
Practice Points
- Focal palmoplantar keratoderma and gingival keratosis (FPGK) is a rare autosomal-dominant syndrome featuring focal, pressure-related, painful palmoplantar keratoderma (PPK) and gingival hyperkeratosis presenting as leukokeratosis.
- Focal pressure-related PPK and oral hyperkeratosis also are seen in pachyonychia congenita (PC), which is caused by mutations in keratin genes and is distinguished from FPGK by characteristic nail changes.
- A shared causative gene suggests that FPGK should be considered part of the PC spectrum.