User login
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
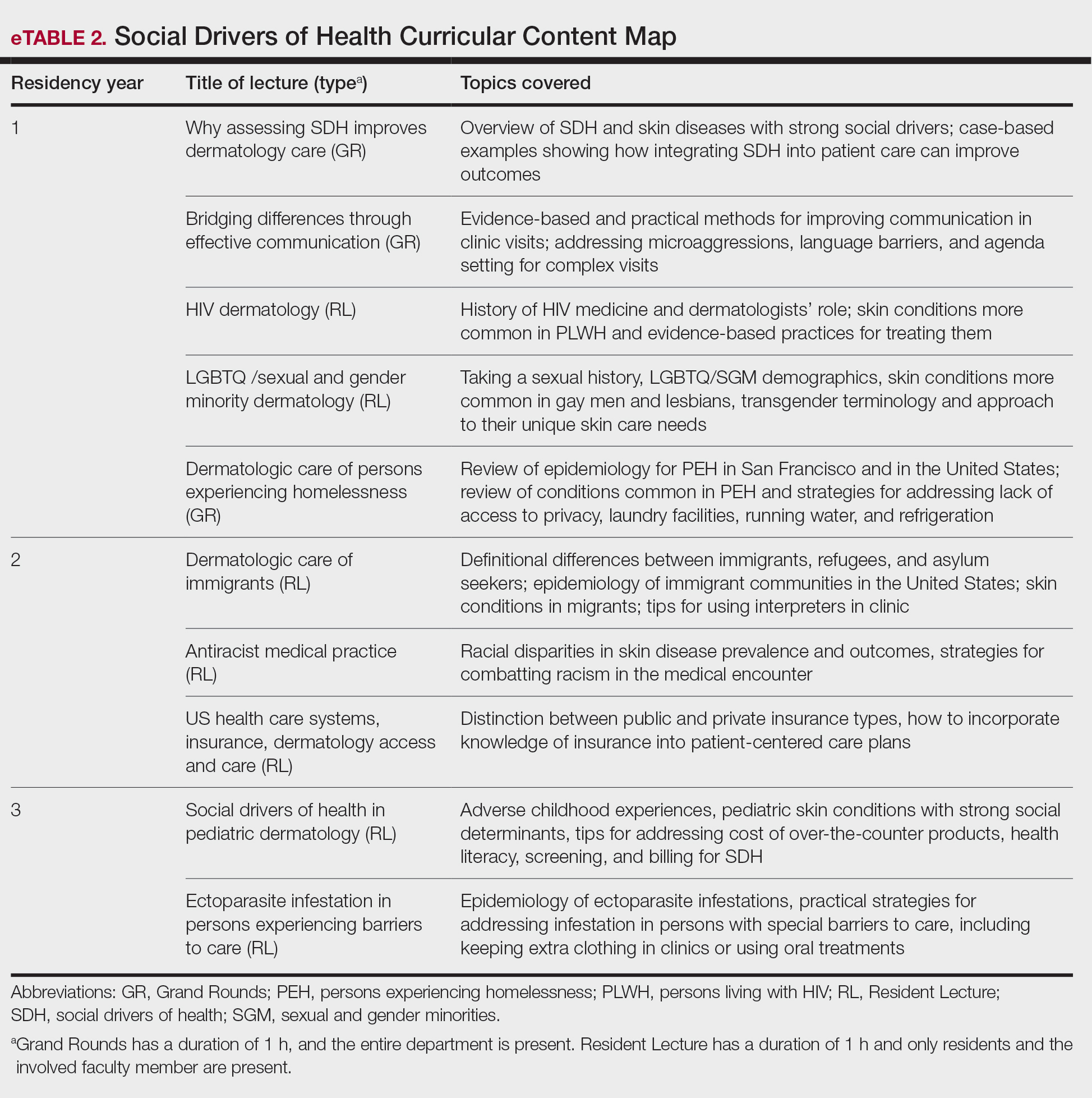
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.

To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
Social drivers of health (SDH) describe the conditions in which an individual is born, grows, lives, works, and ages—all of which collectively influence their health. Examples of SDH include employment status, literacy level, education level, housing status, food access, income level, and social cohesion. Social drivers of health are critical catalysts to attaining health equity. Effectively applying an understanding of how SDH affect the care of all patients is an essential competency for physicians practicing in the modern era of rising income inequality and housing instability and increasing racial, ethnic, language, religious, and cultural diversity in the United States; however, in dermatology residency, this skill set often is developed by the hidden curriculum (ie, the informal curriculum that is based on what patient scenarios a resident happens to face) rather than one represented by formal educational objectives.2
Adding to this challenge of limited formal curricula is that caring for minoritized, marginalized, and other populations facing specific barriers can evoke feelings of frustration, helplessness, and even anger. These feelings can test the limits of a physician’s identity as a healer, leading to burnout and self-protective attitudes such as distancing (emotionally, physically, or both) from these patients.3 This is particularly relevant given that the majority (76%-79% each year from 2007-2019) of medical student matriculants come from families with incomes in the top 2 quintiles nationwide, and fewer than 6% come from the lowest quintile earners.4,5 These data indicate that most trainees have not experienced (and may even have a hard time imagining) the degree of economic and housing instability faced by many of their low-income patients, the care of whom disproportionately falls to large academic medical centers, which sponsor dermatology training programs.6 Many trainees may feel uncomfortable communicating across the broad range of racial, socioeconomic, linguistic, and cultural differences they encounter during training and in practice. Structured opportunities to provide care in a supervised supportive environment combined with didactics that emphasize practical, evidence-based strategies can build empathy, improve attitudes toward patients from diverse backgrounds, and strengthen self-efficacy in challenging scenarios.3
In the past decade, there has been a push toward integrating our understanding of SDH into formal medical training.7 Other specialty training programs—including psychiatry,8 internal medicine,9 pediatrics,10 and family medicine11—have incorporated these elements into their curricula and competency evaluations. In dermatology, as in other specialties, making and implementing effective, patient-centered care plans requires attention to the various social and structural drivers that may influence outcomes. Dermatologists therefore should be educated about SDH during their training programs and empowered to address the ways they affect patient care.
At the University of California San Francisco (UCSF)(San Francisco, California), our dermatology trainees care for patients in several hospital systems citywide, including a tertiary academic medical center with multiple locations, a county hospital, and a Veterans Affairs medical center. Given the diversity of patient populations across our training sites—including many racially and ethnically minoritized individuals, immigrants, patients with limited English proficiency, people experiencing homelessness, and sexual and gender diverse individuals—we identified a critical opportunity to enhance our training through formal didactics and hands-on experiences that integrate SDH into existing curricula and strengthen trainees’ ability to provide high-quality care to all patients.
Implementing an SDH Curriculum
In May 2020, UCSF dermatology faculty with an interest in SDH collaborated with departmental educational leadership to develop a formal SDH curriculum centered around 8 core learning objectives for residents (eTable 1). To achieve these objectives, we organized a 3-year didactic and experiential curriculum consisting of lectures (eTable 2), grand rounds sessions, journal clubs, and community engagement opportunities. Residents also spend 7 months during their training rotating at San Francisco’s city and county hospital (Zuckerberg San Francisco General Hospital [San Francisco, California]) where all faculty are members of the core SDH curriculum development team and where residents can put into practice many of the skills learned in formal didactics to develop patient-centered care plans for low-income patients, approximately 40% of whom have limited English proficiency.


To further center the importance of SDH and health equity in our training program, we developed a Health Equity Chief leadership role for senior dermatology residents. Each year, 2 to 4 residents volunteer for and serve in this role, wherein they work with core faculty to review and improve SDH curriculum elements. They also work to enhance community engagement opportunities for residents (eg, pathway programs aimed at diversifying the dermatology workforce by introducing historically excluded local high school and college students to dermatology as a career path) and improve dermatology trainees’ awareness of the history and health needs of the specific communities we serve in San Francisco. They also are prepared to become leaders in the field of health equity and to improve the care of diverse patient populations after residency. Our faculty curriculum leaders meet quarterly with our Health Equity Chiefs to review their individual and collective goals and strategize ways to improve learner and community engagement. Departmental funds are made available to support these efforts.
Leadership at our safety-net county hospital also developed a patient navigator position to improve our ability to care for patients with the most complex medical conditions and social needs. This role is held by a medical student taking a funded gap year and incorporates aspects of social work (eg, identifying barriers to care and connecting patients with resources such as transportation), quality improvement, and clinical research.12
Assessing Residents’ Experience of a New SDH Curriculum
Prior to curriculum implementation, we surveyed graduating UCSF dermatology residents in June 2020 to assess their familiarity with SDH and the social and medical needs of various populations facing barriers to care, their comfort level with specific challenging clinical situations, and their desire for additional training. Responses were measured using a 5-part Likert scale, with additional options for free-text response. After initiating the SDH curriculum in July 2020, we sent the same survey each year to all senior residents immediately prior to their graduation, offering a small financial incentive ($15 cash gift card) to those who completed the survey. We obtained UCSF Institutional Review Board approval to utilize these survey data to better understand and to enhance residents’ experience of the SDH curriculum.
All 8 residents invited in 2020 completed the survey assessing curriculum efficacy (100% response rate). For the 2023 and 2024 classes, data were analyzed in aggregate (n=14), with a 50% response rate. After implementation of the SDH curriculum, there was improvement in learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (SD, 0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically learners’ awareness of challenges faced by every patient population, from a mean (SD) of 3.12 (0.66) to 4.52 (0.69)(P<.05). Learners were more comfortable handling hypothetical clinical scenarios requiring them to identify and address specific SDH after vs before implementation of the curriculum (mean [SD], 3.5 [1.06] before vs 4.0 [1.16] after)(P>.05), though this difference was not statistically significant. Finally, many respondents expressed appreciation that our curriculum improved their ability to care for patients in complex social circumstances. Residents suggested in the free-text responses that learning more about the historical underpinnings of health disparities, opportunities for grassroots activism, and how to provide more culturally competent care of Native American populations could improve our curriculum.
Implications for Dermatology Training
Our survey results indicate that a formal SDH curriculum can improve dermatology residents’ ability to care for populations with complex social needs. We advocate for implementing SDH curricula into dermatology training programs nationwide, as has been recommended by others.13,14 We also propose that structural competency should eventually be a key dermatologic competency as determined by the Accreditation Council for Graduate Medical Education, in line with the American Medical Association’s recommendation that structural competency is a learned skill required to end health inequity.15 The Accreditation Council for Graduate Medical Education specialty program requirements currently are being revised; interested individuals can engage in this process by submitting this suggestion for public comment (https://www.acgme.org/programs-and-institutions/programs/review-and-comment/).
Limitations of a survey include the relatively small sample size (7-8 per year) and variable response rates. In addition, we did not survey each class of residents at the beginning and end of their training; our comparisons therefore were limited by comparing different individuals with distinct backgrounds and experiences. Furthermore, we acknowledge that the experience of developing this curriculum in San Francisco may be distinct from other communities, where access to dermatologic care may vary according to both the availability of public health insurance and the treatments covered by public insurers. In San Francisco, insurance coverage is near universal, such that residents in our training program regularly care for undocumented immigrants, persons experiencing homelessness, and other populations that might find it challenging to present to dermatology clinics in other settings nationwide.
Final Thoughts
Future directions of our curriculum include exploration of novel curriculum delivery methods (including a problem-based curriculum approach and other more experiential didactics), increased opportunities for community engagement, greater focus on advocacy with an emphasis on broader social and structural policies and their downstream effects, and focusing more specifically on the history and needs of specific low-income San Francisco neighborhoods and diverse patient populations.
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
- US Department of Health and Human Services. Health Equity in Healthy People 2030. Accessed January 7, 2025. https://odphp.healtwh.gov/healthypeople/priority-areas/health-equity-healthy-people-2030
- Axelson DJ, Stull MJ, Coates WC. Social determinants of health: a missing link in emergency medicine training. AEM Educ Train. 2018;2:66-68. doi:10.1002/aet2.10056
- Brenner AM, Guerrero APS, Beresin EV, et al. Teaching medical students and residents about homelessness: complex, evidence-based, and imperative. Acad Psychiatry. 2016;40:572-575. doi:10.1007/s40596-016-0571-6
- Youngclaus J, Roskovensky L. An Updated Look at the Economic Diversity of U.S. Medical Students. American Association of Medical Colleges Analysis in Brief. 2018;18. https://www.aamc.org/media/9596/download?attachment
- Shahriar AA, Puram VV, Miller JM, et al. Socioeconomic diversity of the matriculating US medical student body by race, ethnicity, and sex, 2017-2019. JAMA Netw Open. 2022;5:E222621. doi:10.1001/jamanetworkopen.2022.2621
- Williams JC, Maxey AE, Wei ML, et al. A cross-sectional analysis of Medicaid acceptance among US dermatology residency training programs. J Am Acad Dermatol. 2022;86:453-455. doi:10.1016/j.jaad.2021.09.046
- Daniel H, Bornstein S, Kane G; Health and Public Policy Committee of the American College of Physicians. Addressing social determinants to improve patient care and promote health equity: an American College of Physicians position paper. Ann Intern Med. 2018;168:577-578. doi:10.2105/AJPH
- Hansen H, Kline N, Braslow J, et al. From cultural to structural competency—training psychiatry residents to act on social determinants of health and institutional racism. JAMA Psychiatry. 2018;75:117-118. doi:10.1001/jamapsychiatry.2017.3894
- Schmidt S, Higgins S, George M, et al. An experiential resident module for understanding social determinants of health at an academic safety-net hospital. MedEdPORTAL. 2017;26:10647. doi:10.15766/mep_2374-8265.10647
- Hoffman BD, Rose J, Best D, et al. The community pediatrics training initiative project planning tool: a practical approach to community-based advocacy. MedEdPORTAL. 2017;13:10630.
- Chrisman-Khawam L, Abdullah N, Dhoopar A. Teaching health-care trainees empathy and homelessness IQ through service learning, reflective practice, and altruistic attribution. Int J Psychiatry Med. 2017;52:245-254. doi:10.1177/0091217417730288
- Sanchez-Anguiano ME, Klufas D, Amerson E. Screening for cardiometabolic risk factors in patients with psoriasis and hidradenitis suppurativa: a pilot study in a safety net population. J Am Acad Dermatol. 2024;91:1269-1272. doi:10.1016/j.jaad.2024.07.1518
- Riley C, Vasquez R, Pritchett EN. Equipping dermatologists to address structural and social drivers of inequities—structural competency. JAMA Dermatol. 2024;160:1037-1038. doi:10.1001/jamadermatol.2024.2351
- Crawl-Bey A, Pritchett EN, Riley C. 54338 Structural competency in dermatology: a pilot curriculum for equipping residents to address structural factors that contribute to health inequity. J Am Acad Dermatol. 2024;91(3 suppl):AB318. doi:10.1016/j.jaad.2024.07.1264
- Smith TM. New competency focus involves structural factors in health. American Medical Association. December 18, 202. Accessed March 23, 2026. https://www.ama-assn.org/education/changemeded-initiative/new-competency-focus-involves-structural-factors-health
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Social Drivers of Health Curriculum for Dermatology Residents: the UCSF Experience
Practice Points
- Integrating a formal curriculum on social drivers of health, including didactics on structural racism, cultural humility, communication, and practical strategies, can help residents learn to routinely assess social needs and develop feasible patient-centered care plans.
- Classroom learning paired with experiential rotations and community engagement in safety-net hospitals can help to build empathy, practical skills, and comfort managing real-world social barriers.
- Creation of trainee leadership roles and dedicated program support (eg, departmental funding) can sustain curriculum improvements, foster advocacy skills, and diversify the workforce pipeline.
Inpatient Management of Hidradenitis Suppurativa: A Delphi Consensus Study
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition that affects approximately 0.1% of the US population.1,2 Severe disease or HS flares can lead patients to seek care through the emergency department (ED), with some requiring inpatient admission. 3 Inpatient hospitalization of patients with HS has increased over the last 2 decades, and patients with HS utilize emergency and inpatient care more frequently than those with other dermatologic conditions.4,5 Minority patients and those of lower socioeconomic status are more likely to present to the ED for HS management due to limited access to care and other existing comorbid conditions. 4 In a 2022 study of the Nationwide Readmissions Database, the authors looked at hospital readmission rates of patients with HS compared with those with heart failure—both patient populations with chronic debilitating conditions. Results indicated that the hospital readmission rates for patients with HS surpassed those of patients with heart failure for that year, highlighting the need for improved inpatient management of HS.6
Patients with HS present to the ED with severe pain, fever, wound care, or the need for surgical intervention. The ED and inpatient hospital setting are locations in which physicians may not be as familiar with the diagnosis or treatment of HS, specifically flares or severe disease. 7 The inpatient care setting provides access to certain resources that can be challenging to obtain in the outpatient clinical setting, such as social workers and pain specialists, but also can prove challenging in obtaining other resources for HS management, such as advanced medical therapies. Given the increase in hospital- based care for HS and lack of widespread inpatient access to dermatology and HS experts, consensus recommendations for management of HS in the acute hospital setting would be beneficial. In our study, we sought to generate a collection of expert consensus statements providers can refer to when managing patients with HS in the inpatient setting.
Methods
The study team at the Wake Forest University School of Medicine (Winston-Salem, North Carolina)(M.N., R.P., L.C.S.) developed an initial set of consensus statements based on current published HS treatment guidelines,8,9 publications on management of inpatient HS,3 published supportive care guidelines for Stevens-Johnson syndrome, 10 and personal clinical experience in managing inpatient HS, which resulted in 50 statements organized into the following categories: overall care, wound care, genital care, pain management, infection control, medical management, surgical management, nutrition, and transitional care guidelines. This study was approved by the Wake Forest University institutional review board (IRB00084257).
Participant Recruitment—Dermatologists were identified for participation in the study based on membership in the Society of Dermatology Hospitalists and the Hidradenitis Suppurativa Foundation or authorship of publications relevant to HS or inpatient dermatology. Dermatologists from larger academic institutions with HS specialty clinics and inpatient dermatology services also were identified. Participants were invited via email and could suggest other experts for inclusion. A total of 31 dermatologists were invited to participate in the study, with 26 agreeing to participate. All participating dermatologists were practicing in the United States.
Delphi Study—In the first round of the Delphi study, the participants were sent an online survey via REDCap in which they were asked to rank the appropriateness of each of the proposed 50 guideline statements on a scale of 1 (very inappropriate) to 9 (very appropriate). Participants also were able to provide commentary and feedback on each of the statements. Survey results were analyzed using the RAND/ UCLA Appropriateness Method.11 For each statement, the median rating for appropriateness, interpercentile range (IPR), IPR adjusted for symmetry, and disagreement index (DI) were calculated (DI=IPR/IPR adjusted for symmetry). The 30th and 70th percentiles were used in the DI calculation as the upper and lower limits, respectively. A median rating for appropriateness of 1.0 to 3.9 was considered “inappropriate,” 4.0 to 6.9 was considered “uncertain appropriateness,” and 7.0 to 9.0 was “appropriate.” A DI value greater than or equal to 1 indicated a lack of consensus regarding the appropriateness of the statement. Following each round, participants received a copy of their responses along with the group median rank of each statement. Statements that did not reach consensus in the first Delphi round were revised based on feedback received by the participants, and a second survey with 14 statements was sent via REDCap 2 weeks later. The RAND/UCLA Appropriateness Method also was applied to this second Delphi round. After the second survey, participants received a copy of anonymized comments regarding the consensus statements and were allowed to provide additional final commentary to be included in the discussion of these recommendations.
Results
Twenty-six dermatologists completed the first-round survey, and 24 participants completed the second-round survey. All participants self-identified as having expertise in either HS (n=22 [85%]) or inpatient dermatology (n=17 [65%]), and 13 (50%) participants self-identified as experts in both HS and inpatient dermatology. All participants, except 1, were affiliated with an academic health system with inpatient dermatology services. The average length of time in practice as a dermatologist was 10 years (median, 9 years [range, 3–27 years]).
Of the 50 initial proposed consensus statements, 26 (52%) achieved consensus after the first round; 21 statements revealed DI calculations that did not achieve consensus. Two statements achieved consensus but received median ratings for appropriateness, indicating uncertain appropriateness; because of this, 1 statement was removed and 1 was revised based on participant feedback, resulting in 13 revised statements (eTable 1). Controversial topics in the consensus process included obtaining wound cultures and meaningful culture data interpretation, use of specific biologic medications in the inpatient setting, and use of intravenous ertapenem. Participant responses to these topics are discussed in detail below. Of these secondround statements, all achieved consensus. The final set of consensus statements can be found in eTable 2.
Comment
Our Delphi consensus study combined the expertise of both dermatologists who care for patients with HS and those with inpatient dermatology experience to produce a set of recommendations for the management of HS in the hospital care setting. A strength of this study is inclusion of many national leaders in both HS and inpatient dermatology, with some participants having developed the previously published HS treatment guidelines and others having participated in inpatient dermatology Delphi studies.8-10 The expertise is further strengthened by the geographically diverse institutional representation within the United States.
The final consensus recommendations included 40 statements covering a range of patient care issues, including use of appropriate inpatient subspecialists (care team), supportive care measures (wound care, pain control, genital care), disease-oriented treatment (medical management, surgical management), inpatient complications (infection control, nutrition), and successful transition back to outpatient management (transitional care). These recommendations are meant to serve as a resource for providers to consider when taking care of inpatient HS flares, recognizing that the complexity and individual circumstances of each patient are unique.
Delphi Consensus Recommendations Compared to Prior Guidelines—Several recommendations in the current study align with the previously published North American clinical management guidelines for HS.8,9 Our recommendations agree with prior guidelines on the importance of disease staging and pain assessment using validated assessment tools as well as screening for HS comorbidities. There also is agreement in the potential benefit of involving pain specialists in the development of a comprehensive pain management plan. The inpatient care setting provides a unique opportunity to engage multiple specialists and collaborate on patient care in a timely manner. Our recommendations regarding surgical care also align with established guidelines in recommending incision and drainage as an acute bedside procedure best utilized for symptom relief in inflamed abscesses and relegating most other surgical management to the outpatient setting. Wound care recommendations also are similar, with our expert participants agreeing on individualizing dressing choices based on wound characteristics. A benefit of inpatient wound care is access to skilled nursing for dressing changes and potentially improved access to more sophisticated dressing materials. Our recommendations differ from the prior guidelines in our focus on severe HS, HS flares, and HS complications, which constitute the majority of inpatient disease management. We provide additional guidance on management of secondary infections, perianal fistulous disease, and importantly transitional care to optimize discharge planning.
Differing Opinions in Our Analysis—Despite the success of our Delphi consensus process, there were some differing opinions regarding certain aspects of inpatient HS management, which is to be expected given the lack of strong evidence-based research to support some of the recommended practices. There were differing opinions on the utility of wound culture data, with some participants feeling culture data could help with antibiotic susceptibility and resistance patterns, while others felt wound cultures represent bacterial colonization or biofilm formation.
Initial consensus statements in the first Delphi round were created for individual biologic medications but did not achieve consensus, and feedback on the use of biologics in the inpatient environment was mixed, largely due to logistic and insurance issues. Many participants felt biologic medication cost, difficulty obtaining inpatient reimbursement, health care resource utilization, and availability of biologics in different hospital systems prevented recommending the use of specific biologics during hospitalization. The one exception was in the case of a hospitalized patient who was already receiving infliximab for HS: there was consensus on ensuring the patient dosing was maximized, if appropriate, to 10 mg/kg.12 Ertapenem use also was controversial, with some participants using it as a bridge therapy to either outpatient biologic use or surgery, while others felt it was onerous and difficult to establish reliable access to secure intravenous administration and regular dosing once the patient left the inpatient setting.13 Others said they have experienced objections from infectious disease colleagues on the use of intravenous antibiotics, citing antibiotic stewardship concerns.
Patient Care in the Inpatient Setting—Prior literature suggests patients admitted as inpatients for HS tend to be of lower socioeconomic status and are admitted to larger urban teaching hospitals.14,15 Patients with lower socioeconomic status have increased difficulty accessing health care resources; therefore, inpatient admission serves as an opportunity to provide a holistic HS assessment and coordinate resources for chronic outpatient management.
Study Limitations—This Delphi consensus study has some limitations. The existing literature on inpatient management of HS is limited, challenging our ability to assess the extent to which these published recommendations are already being implemented. Additionally, the study included HS and inpatient dermatology experts from the United States, which means the recommendations may not be generalizable to other countries. Most participants practiced dermatology at large tertiary care academic medical centers, which may limit the ability to implement recommendations in all US inpatient care settings such as small community-based hospitals; however, many of the supportive care guidelines such as pain control, wound care, nutritional support, and social work should be achievable in most inpatient care settings.
Conclusion
Given the increase in inpatient and ED health care utilization for HS, there is an urgent need for expert consensus recommendations on inpatient management of this unique patient population, which requires complex multidisciplinary care. Our recommendations are a resource for providers to utilize and potentially improve the standard of care we provide these patients.
Acknowledgment—We thank the Wake Forest University Clinical and Translational Science Institute (Winston- Salem, North Carolina) for providing statistical help.
- Garg A, Kirby JS, Lavian J, et al. Sex- and age-adjusted population analysis of prevalence estimates for hidradenitis suppurativa in the United States. JAMA Dermatol. 2017;153:760-764.
- Ingram JR. The epidemiology of hidradenitis suppurativa. Br J Dermatol. 2020;183:990-998. doi:10.1111/bjd.19435
- Charrow A, Savage KT, Flood K, et al. Hidradenitis suppurativa for the dermatologic hospitalist. Cutis. 2019;104:276-280.
- Anzaldi L, Perkins JA, Byrd AS, et al. Characterizing inpatient hospitalizations for hidradenitis suppurativa in the United States. J Am Acad Dermatol. 2020;82:510-513. doi:10.1016/j.jaad.2019.09.019
- Khalsa A, Liu G, Kirby JS. Increased utilization of emergency department and inpatient care by patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73:609-614. doi:10.1016/j.jaad.2015.06.053
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the nationwide readmissions database. J Am Acad Dermatol. 2022;87:188-192. doi:10.1016/j.jaad.2021.06.894
- Kirby JS, Miller JJ, Adams DR, et al. Health care utilization patterns and costs for patients with hidradenitis suppurativa. JAMA Dermatol. 2014;150:937-944. doi:10.1001/jamadermatol.2014.691
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part I: diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81:76-90. doi:10.1016/j .jaad.2019.02.067
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi:10.1016/j.jaad.2019.02.068
- Seminario-Vidal L, Kroshinsky D, Malachowski SJ, et al. Society of Dermatology Hospitalists supportive care guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in adults. J Am Acad Dermatol. 2020;82:1553-1567. doi:10.1016/j .jaad.2020.02.066
- Fitch K, Bernstein SJ, Burnand B, et al. The RAND/UCLA Appropriateness Method: User’s Manual. Rand; 2001.
- Oskardmay AN, Miles JA, Sayed CJ. Determining the optimal dose of infliximab for treatment of hidradenitis suppurativa. J Am Acad Dermatol. 2019;81:702-708. doi:10.1016/j.jaad.2019.05.022
- Join-Lambert O, Coignard-Biehler H, Jais JP, et al. Efficacy of ertapenem in severe hidradenitis suppurativa: a pilot study in a cohort of 30 consecutive patients. J Antimicrob Chemother. 2016;71:513-520. doi:10.1093/jac/dkv361
- Khanna R, Whang KA, Huang AH, et al. Inpatient burden of hidradenitis suppurativa in the United States: analysis of the 2016 National Inpatient Sample. J Dermatolog Treat. 2022;33:1150-1152. doi:10.1080/09 546634.2020.1773380
- Patel A, Patel A, Solanki D, et al. Hidradenitis suppurativa in the United States: insights from the national inpatient sample (2008-2017) on contemporary trends in demographics, hospitalization rates, chronic comorbid conditions, and mortality. Cureus. 2022;14:E24755. doi:10.7759/cureus.24755
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition that affects approximately 0.1% of the US population.1,2 Severe disease or HS flares can lead patients to seek care through the emergency department (ED), with some requiring inpatient admission. 3 Inpatient hospitalization of patients with HS has increased over the last 2 decades, and patients with HS utilize emergency and inpatient care more frequently than those with other dermatologic conditions.4,5 Minority patients and those of lower socioeconomic status are more likely to present to the ED for HS management due to limited access to care and other existing comorbid conditions. 4 In a 2022 study of the Nationwide Readmissions Database, the authors looked at hospital readmission rates of patients with HS compared with those with heart failure—both patient populations with chronic debilitating conditions. Results indicated that the hospital readmission rates for patients with HS surpassed those of patients with heart failure for that year, highlighting the need for improved inpatient management of HS.6
Patients with HS present to the ED with severe pain, fever, wound care, or the need for surgical intervention. The ED and inpatient hospital setting are locations in which physicians may not be as familiar with the diagnosis or treatment of HS, specifically flares or severe disease. 7 The inpatient care setting provides access to certain resources that can be challenging to obtain in the outpatient clinical setting, such as social workers and pain specialists, but also can prove challenging in obtaining other resources for HS management, such as advanced medical therapies. Given the increase in hospital- based care for HS and lack of widespread inpatient access to dermatology and HS experts, consensus recommendations for management of HS in the acute hospital setting would be beneficial. In our study, we sought to generate a collection of expert consensus statements providers can refer to when managing patients with HS in the inpatient setting.
Methods
The study team at the Wake Forest University School of Medicine (Winston-Salem, North Carolina)(M.N., R.P., L.C.S.) developed an initial set of consensus statements based on current published HS treatment guidelines,8,9 publications on management of inpatient HS,3 published supportive care guidelines for Stevens-Johnson syndrome, 10 and personal clinical experience in managing inpatient HS, which resulted in 50 statements organized into the following categories: overall care, wound care, genital care, pain management, infection control, medical management, surgical management, nutrition, and transitional care guidelines. This study was approved by the Wake Forest University institutional review board (IRB00084257).
Participant Recruitment—Dermatologists were identified for participation in the study based on membership in the Society of Dermatology Hospitalists and the Hidradenitis Suppurativa Foundation or authorship of publications relevant to HS or inpatient dermatology. Dermatologists from larger academic institutions with HS specialty clinics and inpatient dermatology services also were identified. Participants were invited via email and could suggest other experts for inclusion. A total of 31 dermatologists were invited to participate in the study, with 26 agreeing to participate. All participating dermatologists were practicing in the United States.
Delphi Study—In the first round of the Delphi study, the participants were sent an online survey via REDCap in which they were asked to rank the appropriateness of each of the proposed 50 guideline statements on a scale of 1 (very inappropriate) to 9 (very appropriate). Participants also were able to provide commentary and feedback on each of the statements. Survey results were analyzed using the RAND/ UCLA Appropriateness Method.11 For each statement, the median rating for appropriateness, interpercentile range (IPR), IPR adjusted for symmetry, and disagreement index (DI) were calculated (DI=IPR/IPR adjusted for symmetry). The 30th and 70th percentiles were used in the DI calculation as the upper and lower limits, respectively. A median rating for appropriateness of 1.0 to 3.9 was considered “inappropriate,” 4.0 to 6.9 was considered “uncertain appropriateness,” and 7.0 to 9.0 was “appropriate.” A DI value greater than or equal to 1 indicated a lack of consensus regarding the appropriateness of the statement. Following each round, participants received a copy of their responses along with the group median rank of each statement. Statements that did not reach consensus in the first Delphi round were revised based on feedback received by the participants, and a second survey with 14 statements was sent via REDCap 2 weeks later. The RAND/UCLA Appropriateness Method also was applied to this second Delphi round. After the second survey, participants received a copy of anonymized comments regarding the consensus statements and were allowed to provide additional final commentary to be included in the discussion of these recommendations.
Results
Twenty-six dermatologists completed the first-round survey, and 24 participants completed the second-round survey. All participants self-identified as having expertise in either HS (n=22 [85%]) or inpatient dermatology (n=17 [65%]), and 13 (50%) participants self-identified as experts in both HS and inpatient dermatology. All participants, except 1, were affiliated with an academic health system with inpatient dermatology services. The average length of time in practice as a dermatologist was 10 years (median, 9 years [range, 3–27 years]).
Of the 50 initial proposed consensus statements, 26 (52%) achieved consensus after the first round; 21 statements revealed DI calculations that did not achieve consensus. Two statements achieved consensus but received median ratings for appropriateness, indicating uncertain appropriateness; because of this, 1 statement was removed and 1 was revised based on participant feedback, resulting in 13 revised statements (eTable 1). Controversial topics in the consensus process included obtaining wound cultures and meaningful culture data interpretation, use of specific biologic medications in the inpatient setting, and use of intravenous ertapenem. Participant responses to these topics are discussed in detail below. Of these secondround statements, all achieved consensus. The final set of consensus statements can be found in eTable 2.
Comment
Our Delphi consensus study combined the expertise of both dermatologists who care for patients with HS and those with inpatient dermatology experience to produce a set of recommendations for the management of HS in the hospital care setting. A strength of this study is inclusion of many national leaders in both HS and inpatient dermatology, with some participants having developed the previously published HS treatment guidelines and others having participated in inpatient dermatology Delphi studies.8-10 The expertise is further strengthened by the geographically diverse institutional representation within the United States.
The final consensus recommendations included 40 statements covering a range of patient care issues, including use of appropriate inpatient subspecialists (care team), supportive care measures (wound care, pain control, genital care), disease-oriented treatment (medical management, surgical management), inpatient complications (infection control, nutrition), and successful transition back to outpatient management (transitional care). These recommendations are meant to serve as a resource for providers to consider when taking care of inpatient HS flares, recognizing that the complexity and individual circumstances of each patient are unique.
Delphi Consensus Recommendations Compared to Prior Guidelines—Several recommendations in the current study align with the previously published North American clinical management guidelines for HS.8,9 Our recommendations agree with prior guidelines on the importance of disease staging and pain assessment using validated assessment tools as well as screening for HS comorbidities. There also is agreement in the potential benefit of involving pain specialists in the development of a comprehensive pain management plan. The inpatient care setting provides a unique opportunity to engage multiple specialists and collaborate on patient care in a timely manner. Our recommendations regarding surgical care also align with established guidelines in recommending incision and drainage as an acute bedside procedure best utilized for symptom relief in inflamed abscesses and relegating most other surgical management to the outpatient setting. Wound care recommendations also are similar, with our expert participants agreeing on individualizing dressing choices based on wound characteristics. A benefit of inpatient wound care is access to skilled nursing for dressing changes and potentially improved access to more sophisticated dressing materials. Our recommendations differ from the prior guidelines in our focus on severe HS, HS flares, and HS complications, which constitute the majority of inpatient disease management. We provide additional guidance on management of secondary infections, perianal fistulous disease, and importantly transitional care to optimize discharge planning.
Differing Opinions in Our Analysis—Despite the success of our Delphi consensus process, there were some differing opinions regarding certain aspects of inpatient HS management, which is to be expected given the lack of strong evidence-based research to support some of the recommended practices. There were differing opinions on the utility of wound culture data, with some participants feeling culture data could help with antibiotic susceptibility and resistance patterns, while others felt wound cultures represent bacterial colonization or biofilm formation.
Initial consensus statements in the first Delphi round were created for individual biologic medications but did not achieve consensus, and feedback on the use of biologics in the inpatient environment was mixed, largely due to logistic and insurance issues. Many participants felt biologic medication cost, difficulty obtaining inpatient reimbursement, health care resource utilization, and availability of biologics in different hospital systems prevented recommending the use of specific biologics during hospitalization. The one exception was in the case of a hospitalized patient who was already receiving infliximab for HS: there was consensus on ensuring the patient dosing was maximized, if appropriate, to 10 mg/kg.12 Ertapenem use also was controversial, with some participants using it as a bridge therapy to either outpatient biologic use or surgery, while others felt it was onerous and difficult to establish reliable access to secure intravenous administration and regular dosing once the patient left the inpatient setting.13 Others said they have experienced objections from infectious disease colleagues on the use of intravenous antibiotics, citing antibiotic stewardship concerns.
Patient Care in the Inpatient Setting—Prior literature suggests patients admitted as inpatients for HS tend to be of lower socioeconomic status and are admitted to larger urban teaching hospitals.14,15 Patients with lower socioeconomic status have increased difficulty accessing health care resources; therefore, inpatient admission serves as an opportunity to provide a holistic HS assessment and coordinate resources for chronic outpatient management.
Study Limitations—This Delphi consensus study has some limitations. The existing literature on inpatient management of HS is limited, challenging our ability to assess the extent to which these published recommendations are already being implemented. Additionally, the study included HS and inpatient dermatology experts from the United States, which means the recommendations may not be generalizable to other countries. Most participants practiced dermatology at large tertiary care academic medical centers, which may limit the ability to implement recommendations in all US inpatient care settings such as small community-based hospitals; however, many of the supportive care guidelines such as pain control, wound care, nutritional support, and social work should be achievable in most inpatient care settings.
Conclusion
Given the increase in inpatient and ED health care utilization for HS, there is an urgent need for expert consensus recommendations on inpatient management of this unique patient population, which requires complex multidisciplinary care. Our recommendations are a resource for providers to utilize and potentially improve the standard of care we provide these patients.
Acknowledgment—We thank the Wake Forest University Clinical and Translational Science Institute (Winston- Salem, North Carolina) for providing statistical help.
Hidradenitis suppurativa (HS) is a chronic inflammatory skin condition that affects approximately 0.1% of the US population.1,2 Severe disease or HS flares can lead patients to seek care through the emergency department (ED), with some requiring inpatient admission. 3 Inpatient hospitalization of patients with HS has increased over the last 2 decades, and patients with HS utilize emergency and inpatient care more frequently than those with other dermatologic conditions.4,5 Minority patients and those of lower socioeconomic status are more likely to present to the ED for HS management due to limited access to care and other existing comorbid conditions. 4 In a 2022 study of the Nationwide Readmissions Database, the authors looked at hospital readmission rates of patients with HS compared with those with heart failure—both patient populations with chronic debilitating conditions. Results indicated that the hospital readmission rates for patients with HS surpassed those of patients with heart failure for that year, highlighting the need for improved inpatient management of HS.6
Patients with HS present to the ED with severe pain, fever, wound care, or the need for surgical intervention. The ED and inpatient hospital setting are locations in which physicians may not be as familiar with the diagnosis or treatment of HS, specifically flares or severe disease. 7 The inpatient care setting provides access to certain resources that can be challenging to obtain in the outpatient clinical setting, such as social workers and pain specialists, but also can prove challenging in obtaining other resources for HS management, such as advanced medical therapies. Given the increase in hospital- based care for HS and lack of widespread inpatient access to dermatology and HS experts, consensus recommendations for management of HS in the acute hospital setting would be beneficial. In our study, we sought to generate a collection of expert consensus statements providers can refer to when managing patients with HS in the inpatient setting.
Methods
The study team at the Wake Forest University School of Medicine (Winston-Salem, North Carolina)(M.N., R.P., L.C.S.) developed an initial set of consensus statements based on current published HS treatment guidelines,8,9 publications on management of inpatient HS,3 published supportive care guidelines for Stevens-Johnson syndrome, 10 and personal clinical experience in managing inpatient HS, which resulted in 50 statements organized into the following categories: overall care, wound care, genital care, pain management, infection control, medical management, surgical management, nutrition, and transitional care guidelines. This study was approved by the Wake Forest University institutional review board (IRB00084257).
Participant Recruitment—Dermatologists were identified for participation in the study based on membership in the Society of Dermatology Hospitalists and the Hidradenitis Suppurativa Foundation or authorship of publications relevant to HS or inpatient dermatology. Dermatologists from larger academic institutions with HS specialty clinics and inpatient dermatology services also were identified. Participants were invited via email and could suggest other experts for inclusion. A total of 31 dermatologists were invited to participate in the study, with 26 agreeing to participate. All participating dermatologists were practicing in the United States.
Delphi Study—In the first round of the Delphi study, the participants were sent an online survey via REDCap in which they were asked to rank the appropriateness of each of the proposed 50 guideline statements on a scale of 1 (very inappropriate) to 9 (very appropriate). Participants also were able to provide commentary and feedback on each of the statements. Survey results were analyzed using the RAND/ UCLA Appropriateness Method.11 For each statement, the median rating for appropriateness, interpercentile range (IPR), IPR adjusted for symmetry, and disagreement index (DI) were calculated (DI=IPR/IPR adjusted for symmetry). The 30th and 70th percentiles were used in the DI calculation as the upper and lower limits, respectively. A median rating for appropriateness of 1.0 to 3.9 was considered “inappropriate,” 4.0 to 6.9 was considered “uncertain appropriateness,” and 7.0 to 9.0 was “appropriate.” A DI value greater than or equal to 1 indicated a lack of consensus regarding the appropriateness of the statement. Following each round, participants received a copy of their responses along with the group median rank of each statement. Statements that did not reach consensus in the first Delphi round were revised based on feedback received by the participants, and a second survey with 14 statements was sent via REDCap 2 weeks later. The RAND/UCLA Appropriateness Method also was applied to this second Delphi round. After the second survey, participants received a copy of anonymized comments regarding the consensus statements and were allowed to provide additional final commentary to be included in the discussion of these recommendations.
Results
Twenty-six dermatologists completed the first-round survey, and 24 participants completed the second-round survey. All participants self-identified as having expertise in either HS (n=22 [85%]) or inpatient dermatology (n=17 [65%]), and 13 (50%) participants self-identified as experts in both HS and inpatient dermatology. All participants, except 1, were affiliated with an academic health system with inpatient dermatology services. The average length of time in practice as a dermatologist was 10 years (median, 9 years [range, 3–27 years]).
Of the 50 initial proposed consensus statements, 26 (52%) achieved consensus after the first round; 21 statements revealed DI calculations that did not achieve consensus. Two statements achieved consensus but received median ratings for appropriateness, indicating uncertain appropriateness; because of this, 1 statement was removed and 1 was revised based on participant feedback, resulting in 13 revised statements (eTable 1). Controversial topics in the consensus process included obtaining wound cultures and meaningful culture data interpretation, use of specific biologic medications in the inpatient setting, and use of intravenous ertapenem. Participant responses to these topics are discussed in detail below. Of these secondround statements, all achieved consensus. The final set of consensus statements can be found in eTable 2.
Comment
Our Delphi consensus study combined the expertise of both dermatologists who care for patients with HS and those with inpatient dermatology experience to produce a set of recommendations for the management of HS in the hospital care setting. A strength of this study is inclusion of many national leaders in both HS and inpatient dermatology, with some participants having developed the previously published HS treatment guidelines and others having participated in inpatient dermatology Delphi studies.8-10 The expertise is further strengthened by the geographically diverse institutional representation within the United States.
The final consensus recommendations included 40 statements covering a range of patient care issues, including use of appropriate inpatient subspecialists (care team), supportive care measures (wound care, pain control, genital care), disease-oriented treatment (medical management, surgical management), inpatient complications (infection control, nutrition), and successful transition back to outpatient management (transitional care). These recommendations are meant to serve as a resource for providers to consider when taking care of inpatient HS flares, recognizing that the complexity and individual circumstances of each patient are unique.
Delphi Consensus Recommendations Compared to Prior Guidelines—Several recommendations in the current study align with the previously published North American clinical management guidelines for HS.8,9 Our recommendations agree with prior guidelines on the importance of disease staging and pain assessment using validated assessment tools as well as screening for HS comorbidities. There also is agreement in the potential benefit of involving pain specialists in the development of a comprehensive pain management plan. The inpatient care setting provides a unique opportunity to engage multiple specialists and collaborate on patient care in a timely manner. Our recommendations regarding surgical care also align with established guidelines in recommending incision and drainage as an acute bedside procedure best utilized for symptom relief in inflamed abscesses and relegating most other surgical management to the outpatient setting. Wound care recommendations also are similar, with our expert participants agreeing on individualizing dressing choices based on wound characteristics. A benefit of inpatient wound care is access to skilled nursing for dressing changes and potentially improved access to more sophisticated dressing materials. Our recommendations differ from the prior guidelines in our focus on severe HS, HS flares, and HS complications, which constitute the majority of inpatient disease management. We provide additional guidance on management of secondary infections, perianal fistulous disease, and importantly transitional care to optimize discharge planning.
Differing Opinions in Our Analysis—Despite the success of our Delphi consensus process, there were some differing opinions regarding certain aspects of inpatient HS management, which is to be expected given the lack of strong evidence-based research to support some of the recommended practices. There were differing opinions on the utility of wound culture data, with some participants feeling culture data could help with antibiotic susceptibility and resistance patterns, while others felt wound cultures represent bacterial colonization or biofilm formation.
Initial consensus statements in the first Delphi round were created for individual biologic medications but did not achieve consensus, and feedback on the use of biologics in the inpatient environment was mixed, largely due to logistic and insurance issues. Many participants felt biologic medication cost, difficulty obtaining inpatient reimbursement, health care resource utilization, and availability of biologics in different hospital systems prevented recommending the use of specific biologics during hospitalization. The one exception was in the case of a hospitalized patient who was already receiving infliximab for HS: there was consensus on ensuring the patient dosing was maximized, if appropriate, to 10 mg/kg.12 Ertapenem use also was controversial, with some participants using it as a bridge therapy to either outpatient biologic use or surgery, while others felt it was onerous and difficult to establish reliable access to secure intravenous administration and regular dosing once the patient left the inpatient setting.13 Others said they have experienced objections from infectious disease colleagues on the use of intravenous antibiotics, citing antibiotic stewardship concerns.
Patient Care in the Inpatient Setting—Prior literature suggests patients admitted as inpatients for HS tend to be of lower socioeconomic status and are admitted to larger urban teaching hospitals.14,15 Patients with lower socioeconomic status have increased difficulty accessing health care resources; therefore, inpatient admission serves as an opportunity to provide a holistic HS assessment and coordinate resources for chronic outpatient management.
Study Limitations—This Delphi consensus study has some limitations. The existing literature on inpatient management of HS is limited, challenging our ability to assess the extent to which these published recommendations are already being implemented. Additionally, the study included HS and inpatient dermatology experts from the United States, which means the recommendations may not be generalizable to other countries. Most participants practiced dermatology at large tertiary care academic medical centers, which may limit the ability to implement recommendations in all US inpatient care settings such as small community-based hospitals; however, many of the supportive care guidelines such as pain control, wound care, nutritional support, and social work should be achievable in most inpatient care settings.
Conclusion
Given the increase in inpatient and ED health care utilization for HS, there is an urgent need for expert consensus recommendations on inpatient management of this unique patient population, which requires complex multidisciplinary care. Our recommendations are a resource for providers to utilize and potentially improve the standard of care we provide these patients.
Acknowledgment—We thank the Wake Forest University Clinical and Translational Science Institute (Winston- Salem, North Carolina) for providing statistical help.
- Garg A, Kirby JS, Lavian J, et al. Sex- and age-adjusted population analysis of prevalence estimates for hidradenitis suppurativa in the United States. JAMA Dermatol. 2017;153:760-764.
- Ingram JR. The epidemiology of hidradenitis suppurativa. Br J Dermatol. 2020;183:990-998. doi:10.1111/bjd.19435
- Charrow A, Savage KT, Flood K, et al. Hidradenitis suppurativa for the dermatologic hospitalist. Cutis. 2019;104:276-280.
- Anzaldi L, Perkins JA, Byrd AS, et al. Characterizing inpatient hospitalizations for hidradenitis suppurativa in the United States. J Am Acad Dermatol. 2020;82:510-513. doi:10.1016/j.jaad.2019.09.019
- Khalsa A, Liu G, Kirby JS. Increased utilization of emergency department and inpatient care by patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73:609-614. doi:10.1016/j.jaad.2015.06.053
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the nationwide readmissions database. J Am Acad Dermatol. 2022;87:188-192. doi:10.1016/j.jaad.2021.06.894
- Kirby JS, Miller JJ, Adams DR, et al. Health care utilization patterns and costs for patients with hidradenitis suppurativa. JAMA Dermatol. 2014;150:937-944. doi:10.1001/jamadermatol.2014.691
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part I: diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81:76-90. doi:10.1016/j .jaad.2019.02.067
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi:10.1016/j.jaad.2019.02.068
- Seminario-Vidal L, Kroshinsky D, Malachowski SJ, et al. Society of Dermatology Hospitalists supportive care guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in adults. J Am Acad Dermatol. 2020;82:1553-1567. doi:10.1016/j .jaad.2020.02.066
- Fitch K, Bernstein SJ, Burnand B, et al. The RAND/UCLA Appropriateness Method: User’s Manual. Rand; 2001.
- Oskardmay AN, Miles JA, Sayed CJ. Determining the optimal dose of infliximab for treatment of hidradenitis suppurativa. J Am Acad Dermatol. 2019;81:702-708. doi:10.1016/j.jaad.2019.05.022
- Join-Lambert O, Coignard-Biehler H, Jais JP, et al. Efficacy of ertapenem in severe hidradenitis suppurativa: a pilot study in a cohort of 30 consecutive patients. J Antimicrob Chemother. 2016;71:513-520. doi:10.1093/jac/dkv361
- Khanna R, Whang KA, Huang AH, et al. Inpatient burden of hidradenitis suppurativa in the United States: analysis of the 2016 National Inpatient Sample. J Dermatolog Treat. 2022;33:1150-1152. doi:10.1080/09 546634.2020.1773380
- Patel A, Patel A, Solanki D, et al. Hidradenitis suppurativa in the United States: insights from the national inpatient sample (2008-2017) on contemporary trends in demographics, hospitalization rates, chronic comorbid conditions, and mortality. Cureus. 2022;14:E24755. doi:10.7759/cureus.24755
- Garg A, Kirby JS, Lavian J, et al. Sex- and age-adjusted population analysis of prevalence estimates for hidradenitis suppurativa in the United States. JAMA Dermatol. 2017;153:760-764.
- Ingram JR. The epidemiology of hidradenitis suppurativa. Br J Dermatol. 2020;183:990-998. doi:10.1111/bjd.19435
- Charrow A, Savage KT, Flood K, et al. Hidradenitis suppurativa for the dermatologic hospitalist. Cutis. 2019;104:276-280.
- Anzaldi L, Perkins JA, Byrd AS, et al. Characterizing inpatient hospitalizations for hidradenitis suppurativa in the United States. J Am Acad Dermatol. 2020;82:510-513. doi:10.1016/j.jaad.2019.09.019
- Khalsa A, Liu G, Kirby JS. Increased utilization of emergency department and inpatient care by patients with hidradenitis suppurativa. J Am Acad Dermatol. 2015;73:609-614. doi:10.1016/j.jaad.2015.06.053
- Edigin E, Kaul S, Eseaton PO, et al. At 180 days hidradenitis suppurativa readmission rate is comparable to heart failure: analysis of the nationwide readmissions database. J Am Acad Dermatol. 2022;87:188-192. doi:10.1016/j.jaad.2021.06.894
- Kirby JS, Miller JJ, Adams DR, et al. Health care utilization patterns and costs for patients with hidradenitis suppurativa. JAMA Dermatol. 2014;150:937-944. doi:10.1001/jamadermatol.2014.691
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part I: diagnosis, evaluation, and the use of complementary and procedural management. J Am Acad Dermatol. 2019;81:76-90. doi:10.1016/j .jaad.2019.02.067
- Alikhan A, Sayed C, Alavi A, et al. North American clinical management guidelines for hidradenitis suppurativa: a publication from the United States and Canadian Hidradenitis Suppurativa Foundations: part II: topical, intralesional, and systemic medical management. J Am Acad Dermatol. 2019;81:91-101. doi:10.1016/j.jaad.2019.02.068
- Seminario-Vidal L, Kroshinsky D, Malachowski SJ, et al. Society of Dermatology Hospitalists supportive care guidelines for the management of Stevens-Johnson syndrome/toxic epidermal necrolysis in adults. J Am Acad Dermatol. 2020;82:1553-1567. doi:10.1016/j .jaad.2020.02.066
- Fitch K, Bernstein SJ, Burnand B, et al. The RAND/UCLA Appropriateness Method: User’s Manual. Rand; 2001.
- Oskardmay AN, Miles JA, Sayed CJ. Determining the optimal dose of infliximab for treatment of hidradenitis suppurativa. J Am Acad Dermatol. 2019;81:702-708. doi:10.1016/j.jaad.2019.05.022
- Join-Lambert O, Coignard-Biehler H, Jais JP, et al. Efficacy of ertapenem in severe hidradenitis suppurativa: a pilot study in a cohort of 30 consecutive patients. J Antimicrob Chemother. 2016;71:513-520. doi:10.1093/jac/dkv361
- Khanna R, Whang KA, Huang AH, et al. Inpatient burden of hidradenitis suppurativa in the United States: analysis of the 2016 National Inpatient Sample. J Dermatolog Treat. 2022;33:1150-1152. doi:10.1080/09 546634.2020.1773380
- Patel A, Patel A, Solanki D, et al. Hidradenitis suppurativa in the United States: insights from the national inpatient sample (2008-2017) on contemporary trends in demographics, hospitalization rates, chronic comorbid conditions, and mortality. Cureus. 2022;14:E24755. doi:10.7759/cureus.24755
Practice Points
- Given the increase in hospital-based care for hidradenitis suppurativa (HS) and the lack of widespread inpatient access to dermatology and HS experts, consensus recommendations for management of HS in the acute hospital setting would be beneficial.
- Our Delphi study yielded 40 statements that reached consensus covering a range of patient care issues (eg, appropriate inpatient subspecialists [care team]), supportive care measures (wound care, pain control, genital care), disease-oriented treatment (medical management, surgical management), inpatient complications (infection control, nutrition), and successful transition to outpatient management (transitional care).
- These recommendations serve as an important resource for providers caring for inpatients with HS and represent a successful collaboration between inpatient dermatology and HS experts.
What Neglected Tropical Diseases Teach Us About Stigma
Neglected tropical diseases (NTDs) are a group of 20 diseases that typically are chronic and cause long-term disability, which negatively impacts work productivity, child survival, and school performance and attendance with adverse effect on future earnings.1 Data from the 2013 Global Burden of Disease study revealed that half of the world’s NTDs occur in poor populations living in wealthy countries.2 Neglected tropical diseases with skin manifestations include parasitic infections (eg, American trypanosomiasis, African trypanosomiasis, dracunculiasis, echinococcosis, foodborne trematodiases, leishmaniasis, lymphatic filariasis, onchocerciasis, scabies and other ectoparasites, schistosomiasis, soil-transmitted helminths, taeniasis/cysticercosis), bacterial infections (eg, Buruli ulcer, leprosy, yaws), fungal infections (eg, mycetoma, chromoblastomycosis, deep mycoses), and viral infections (eg, dengue, chikungunya). Rabies and snakebite envenomization involve the skin through inoculation. Within the larger group of NTDs, the World Health Organization has identified “skin NTDs” as a subgroup of NTDs that present primarily with changes in the skin.3 In the absence of early diagnosis and treatment of these diseases, chronic and lifelong disfigurement, disability, stigma, and socioeconomic losses ensue.
The Department of Health of the Government of Western Australia stated:
Stigma is a mark of disgrace that sets a person apart from others. When a person is labeled by their illness they are no longer seen as an individual but as part of a stereotyped group. Negative attitudes and beliefs toward this group create prejudice which leads to negative actions and discrimination.4
Stigma associated with skin NTDs exemplifies how skin diseases can have enduring impact on individuals.5 For example, scarring from inactive cutaneous leishmaniasis carries heavy psychosocial burden. Young women reported that facial scarring from cutaneous leishmaniasis led to marriage rejections.6 Some even reported extreme suicidal ideations.7 Recently, major depressive disorder associated with scarring from inactive cutaneous leishmaniasis has been recognized as a notable contributor to disease burden from cutaneous leishmaniasis.8
Lymphatic filariasis is a major cause of leg and scrotal lymphedema worldwide. Even when the condition is treated, lymphedema often persists due to chronic irreversible lymphatic damage. A systematic review of 18 stigma studies in lymphatic filariasis found common themes related to the deleterious consequences of stigma on social relationships; work and education opportunities; health outcomes from reduced treatment-seeking behavior; and mental health, including anxiety, depression, and suicidal tendencies.9 In one subdistrict in India, implementation of a community-based lymphedema management program that consisted of teaching hygiene and limb care for more than 20,000 lymphedema patients and performing community outreach activities (eg, street plays, radio programs, informational brochures) to teach people about lymphatic filariasis and lymphedema care was associated with community members being accepting of patients and an improvement in their understanding of disease etiology.10
Skin involvement from onchocerciasis infection (onchocercal skin disease) is another condition associated with notable stigma.9 Through the African Programme for Onchocerciasis Control, annual mass drug administration of ivermectin in onchocerciasis-endemic communities has reduced the rate of onchocercal skin disease in these communities. In looking at perception of stigma in onchocercal skin diseases before community-directed ivermectin therapy and 7 to 10 years after, avoidance of people with onchocercal skin disease decreased from 32.7% to 4.3%. There also was an improvement in relationships between healthy people and those with onchocercal skin disease.11
One of the most stigmatizing conditions is leprosy, often referred to as Hansen disease to give credit to the person who discovered that leprosy was caused by Mycobacterium leprae and not from sin, being cursed, or genetic inheritance. Even with this knowledge, stigma persists that can lead to family abandonment and social isolation, which further impacts afflicted individuals’ willingness to seek care, thus leading to disease progression. More recently, there has been research looking at interventions to reduce the stigma that individuals afflicted with leprosy face. In a study from Indonesia where individuals with leprosy were randomized to counseling, socioeconomic development, or contact between community members and affected people, all interventions were associated with a reduction in stigma.12 A rights-based counseling module integrated individual, family, and group forms of counseling and consisted of 5 sessions that focused on medical knowledge of leprosy and rights of individuals with leprosy, along with elements of cognitive behavioral therapy. Socioeconomic development involved opportunities for business training, creation of community groups through which microfinance services were administered, and other assistance to improve livelihood. Informed by evidence from the field of human immunodeficiency virus and mental health that co
Although steps are being taken to address the psychosocial burden of skin NTDs, there is still much work to be done. From the public health lens that largely governs the policies and approaches toward addressing NTDs, the focus often is on interrupting and eliminating disease transmission. Morbidity management, including reduction in stigma and functional impairment, is not always the priority. It is in this space that dermatologists are uniquely positioned to advocate for management approaches that address the morbidity associated with skin NTDs. We have an intimate understanding of how impactful skin diseases can be, even if they are not commonly fatal. Globally, skin diseases are the fourth leading cause of nonfatal disease burden,14 yet dermatology lacks effective evidence-based interventions for reducing stigma in our patients with visible chronic diseases.15
Every day, we see firsthand how skin diseases affect not only our patients but also their families, friends, and caregivers. Although we may not see skin NTDs on a regular basis in our clinics, we can understand almost intuitively how devastating skin NTDs could be on individuals, families, and communities. For patients with skin NTDs, receiving medical therapy is only one component of treatment. In addition to optimizing early diagnosis and treatment, interventions taken to educate families and communities affected by skin NTDs are vitally important. Stigma reduction is possible, as we have seen from the aforementioned interventions used in communities with lymphatic filariasis, onchocerciasis, and leprosy. We call upon our fellow dermatologists to take interest in creating, evaluating, and promoting interventions that address stigma in skin NTDs; it is critical in achieving and maintaining health and well-being for our patients.
- Neglected tropical diseases. World Health Organization website. https://www.who.int/neglected_diseases/diseases/en/. Accessed September 10, 2019.
- Hotez PJ, Damania A, Naghavi M. Blue Marble Health and the Global Burden of Disease Study 2013. PLoS Negl Trop Dis. 2016;10:E0004744.
- Skin NTDs. World Health Organization website. https://www.who.int/neglected_diseases/skin-ntds/en/. Accessed September 10, 2019.
- Government of Western Australia Department of Health. Stigma, discrimination and mental illness. February 2009. http://www.health.wa.gov.au/docreg/Education/Population/Health_Problems/Mental_Illness/Mentalhealth_stigma_fact.pdf. Accessed September 10, 2019.
- Hotez PJ. Stigma: the stealth weapon of the NTD. PLoS Negl Trop Dis. 2008;2:E230.
- Bennis I, Belaid L, De Brouwere V, et al. “The mosquitoes that destroy your face.” social impact of cutaneous leishmaniasis in Southeastern Morocco, a qualitative study. PLoS One. 2017;12:E0189906.
- Bennis I, Thys S, Filali H, et al. Psychosocial impact of scars due to cutaneous leishmaniasis on high school students in Errachidia province, Morocco. Infect Dis Poverty. 2017;6:46.
- Bailey F, Mondragon-Shem K, Haines LR, et al. Cutaneous leishmaniasis and co-morbid major depressive disorder: a systematic review with burden estimates. PLoS Negl Trop Dis. 2019;13:E0007092.
- Hofstraat K, van Brakel WH. Social stigma towards neglected tropical diseases: a systematic review. Int Health. 2016;8(suppl 1):I53-I70.
- Cassidy T, Worrell CM, Little K, et al. Experiences of a community-based lymphedema management program for lymphatic filariasis in Odisha State, India: an analysis of focus group discussions with patients, families, community members and program volunteers. PLoS Negl Trop Dis. 2016;10:E0004424.
- Tchounkeu YF, Onyeneho NG, Wanji S, et al. Changes in stigma and discrimination of onchocerciasis in Africa. Trans R Soc Trop Med Hyg. 2012;106:340-347.
- Dadun D, Van Brakel WH, Peters RMH, et al. Impact of socio-economic development, contact and peer counselling on stigma against persons affected by leprosy in Cirebon, Indonesia—a randomised controlled trial. Lepr Rev. 2017;88:2-22.
- Kumar A, Lambert S, Lockwood DNJ. Picturing health: a new face for leprosy. Lancet. 2019;393:629-638.
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134:1527-1534.
- Topp J, Andrees V, Weinberger NA, et al. Strategies to reduce stigma related to visible chronic skin diseases: a systematic review [published online June 8, 2019]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.15734.
Neglected tropical diseases (NTDs) are a group of 20 diseases that typically are chronic and cause long-term disability, which negatively impacts work productivity, child survival, and school performance and attendance with adverse effect on future earnings.1 Data from the 2013 Global Burden of Disease study revealed that half of the world’s NTDs occur in poor populations living in wealthy countries.2 Neglected tropical diseases with skin manifestations include parasitic infections (eg, American trypanosomiasis, African trypanosomiasis, dracunculiasis, echinococcosis, foodborne trematodiases, leishmaniasis, lymphatic filariasis, onchocerciasis, scabies and other ectoparasites, schistosomiasis, soil-transmitted helminths, taeniasis/cysticercosis), bacterial infections (eg, Buruli ulcer, leprosy, yaws), fungal infections (eg, mycetoma, chromoblastomycosis, deep mycoses), and viral infections (eg, dengue, chikungunya). Rabies and snakebite envenomization involve the skin through inoculation. Within the larger group of NTDs, the World Health Organization has identified “skin NTDs” as a subgroup of NTDs that present primarily with changes in the skin.3 In the absence of early diagnosis and treatment of these diseases, chronic and lifelong disfigurement, disability, stigma, and socioeconomic losses ensue.
The Department of Health of the Government of Western Australia stated:
Stigma is a mark of disgrace that sets a person apart from others. When a person is labeled by their illness they are no longer seen as an individual but as part of a stereotyped group. Negative attitudes and beliefs toward this group create prejudice which leads to negative actions and discrimination.4
Stigma associated with skin NTDs exemplifies how skin diseases can have enduring impact on individuals.5 For example, scarring from inactive cutaneous leishmaniasis carries heavy psychosocial burden. Young women reported that facial scarring from cutaneous leishmaniasis led to marriage rejections.6 Some even reported extreme suicidal ideations.7 Recently, major depressive disorder associated with scarring from inactive cutaneous leishmaniasis has been recognized as a notable contributor to disease burden from cutaneous leishmaniasis.8
Lymphatic filariasis is a major cause of leg and scrotal lymphedema worldwide. Even when the condition is treated, lymphedema often persists due to chronic irreversible lymphatic damage. A systematic review of 18 stigma studies in lymphatic filariasis found common themes related to the deleterious consequences of stigma on social relationships; work and education opportunities; health outcomes from reduced treatment-seeking behavior; and mental health, including anxiety, depression, and suicidal tendencies.9 In one subdistrict in India, implementation of a community-based lymphedema management program that consisted of teaching hygiene and limb care for more than 20,000 lymphedema patients and performing community outreach activities (eg, street plays, radio programs, informational brochures) to teach people about lymphatic filariasis and lymphedema care was associated with community members being accepting of patients and an improvement in their understanding of disease etiology.10
Skin involvement from onchocerciasis infection (onchocercal skin disease) is another condition associated with notable stigma.9 Through the African Programme for Onchocerciasis Control, annual mass drug administration of ivermectin in onchocerciasis-endemic communities has reduced the rate of onchocercal skin disease in these communities. In looking at perception of stigma in onchocercal skin diseases before community-directed ivermectin therapy and 7 to 10 years after, avoidance of people with onchocercal skin disease decreased from 32.7% to 4.3%. There also was an improvement in relationships between healthy people and those with onchocercal skin disease.11
One of the most stigmatizing conditions is leprosy, often referred to as Hansen disease to give credit to the person who discovered that leprosy was caused by Mycobacterium leprae and not from sin, being cursed, or genetic inheritance. Even with this knowledge, stigma persists that can lead to family abandonment and social isolation, which further impacts afflicted individuals’ willingness to seek care, thus leading to disease progression. More recently, there has been research looking at interventions to reduce the stigma that individuals afflicted with leprosy face. In a study from Indonesia where individuals with leprosy were randomized to counseling, socioeconomic development, or contact between community members and affected people, all interventions were associated with a reduction in stigma.12 A rights-based counseling module integrated individual, family, and group forms of counseling and consisted of 5 sessions that focused on medical knowledge of leprosy and rights of individuals with leprosy, along with elements of cognitive behavioral therapy. Socioeconomic development involved opportunities for business training, creation of community groups through which microfinance services were administered, and other assistance to improve livelihood. Informed by evidence from the field of human immunodeficiency virus and mental health that co
Although steps are being taken to address the psychosocial burden of skin NTDs, there is still much work to be done. From the public health lens that largely governs the policies and approaches toward addressing NTDs, the focus often is on interrupting and eliminating disease transmission. Morbidity management, including reduction in stigma and functional impairment, is not always the priority. It is in this space that dermatologists are uniquely positioned to advocate for management approaches that address the morbidity associated with skin NTDs. We have an intimate understanding of how impactful skin diseases can be, even if they are not commonly fatal. Globally, skin diseases are the fourth leading cause of nonfatal disease burden,14 yet dermatology lacks effective evidence-based interventions for reducing stigma in our patients with visible chronic diseases.15
Every day, we see firsthand how skin diseases affect not only our patients but also their families, friends, and caregivers. Although we may not see skin NTDs on a regular basis in our clinics, we can understand almost intuitively how devastating skin NTDs could be on individuals, families, and communities. For patients with skin NTDs, receiving medical therapy is only one component of treatment. In addition to optimizing early diagnosis and treatment, interventions taken to educate families and communities affected by skin NTDs are vitally important. Stigma reduction is possible, as we have seen from the aforementioned interventions used in communities with lymphatic filariasis, onchocerciasis, and leprosy. We call upon our fellow dermatologists to take interest in creating, evaluating, and promoting interventions that address stigma in skin NTDs; it is critical in achieving and maintaining health and well-being for our patients.
Neglected tropical diseases (NTDs) are a group of 20 diseases that typically are chronic and cause long-term disability, which negatively impacts work productivity, child survival, and school performance and attendance with adverse effect on future earnings.1 Data from the 2013 Global Burden of Disease study revealed that half of the world’s NTDs occur in poor populations living in wealthy countries.2 Neglected tropical diseases with skin manifestations include parasitic infections (eg, American trypanosomiasis, African trypanosomiasis, dracunculiasis, echinococcosis, foodborne trematodiases, leishmaniasis, lymphatic filariasis, onchocerciasis, scabies and other ectoparasites, schistosomiasis, soil-transmitted helminths, taeniasis/cysticercosis), bacterial infections (eg, Buruli ulcer, leprosy, yaws), fungal infections (eg, mycetoma, chromoblastomycosis, deep mycoses), and viral infections (eg, dengue, chikungunya). Rabies and snakebite envenomization involve the skin through inoculation. Within the larger group of NTDs, the World Health Organization has identified “skin NTDs” as a subgroup of NTDs that present primarily with changes in the skin.3 In the absence of early diagnosis and treatment of these diseases, chronic and lifelong disfigurement, disability, stigma, and socioeconomic losses ensue.
The Department of Health of the Government of Western Australia stated:
Stigma is a mark of disgrace that sets a person apart from others. When a person is labeled by their illness they are no longer seen as an individual but as part of a stereotyped group. Negative attitudes and beliefs toward this group create prejudice which leads to negative actions and discrimination.4
Stigma associated with skin NTDs exemplifies how skin diseases can have enduring impact on individuals.5 For example, scarring from inactive cutaneous leishmaniasis carries heavy psychosocial burden. Young women reported that facial scarring from cutaneous leishmaniasis led to marriage rejections.6 Some even reported extreme suicidal ideations.7 Recently, major depressive disorder associated with scarring from inactive cutaneous leishmaniasis has been recognized as a notable contributor to disease burden from cutaneous leishmaniasis.8
Lymphatic filariasis is a major cause of leg and scrotal lymphedema worldwide. Even when the condition is treated, lymphedema often persists due to chronic irreversible lymphatic damage. A systematic review of 18 stigma studies in lymphatic filariasis found common themes related to the deleterious consequences of stigma on social relationships; work and education opportunities; health outcomes from reduced treatment-seeking behavior; and mental health, including anxiety, depression, and suicidal tendencies.9 In one subdistrict in India, implementation of a community-based lymphedema management program that consisted of teaching hygiene and limb care for more than 20,000 lymphedema patients and performing community outreach activities (eg, street plays, radio programs, informational brochures) to teach people about lymphatic filariasis and lymphedema care was associated with community members being accepting of patients and an improvement in their understanding of disease etiology.10
Skin involvement from onchocerciasis infection (onchocercal skin disease) is another condition associated with notable stigma.9 Through the African Programme for Onchocerciasis Control, annual mass drug administration of ivermectin in onchocerciasis-endemic communities has reduced the rate of onchocercal skin disease in these communities. In looking at perception of stigma in onchocercal skin diseases before community-directed ivermectin therapy and 7 to 10 years after, avoidance of people with onchocercal skin disease decreased from 32.7% to 4.3%. There also was an improvement in relationships between healthy people and those with onchocercal skin disease.11
One of the most stigmatizing conditions is leprosy, often referred to as Hansen disease to give credit to the person who discovered that leprosy was caused by Mycobacterium leprae and not from sin, being cursed, or genetic inheritance. Even with this knowledge, stigma persists that can lead to family abandonment and social isolation, which further impacts afflicted individuals’ willingness to seek care, thus leading to disease progression. More recently, there has been research looking at interventions to reduce the stigma that individuals afflicted with leprosy face. In a study from Indonesia where individuals with leprosy were randomized to counseling, socioeconomic development, or contact between community members and affected people, all interventions were associated with a reduction in stigma.12 A rights-based counseling module integrated individual, family, and group forms of counseling and consisted of 5 sessions that focused on medical knowledge of leprosy and rights of individuals with leprosy, along with elements of cognitive behavioral therapy. Socioeconomic development involved opportunities for business training, creation of community groups through which microfinance services were administered, and other assistance to improve livelihood. Informed by evidence from the field of human immunodeficiency virus and mental health that co
Although steps are being taken to address the psychosocial burden of skin NTDs, there is still much work to be done. From the public health lens that largely governs the policies and approaches toward addressing NTDs, the focus often is on interrupting and eliminating disease transmission. Morbidity management, including reduction in stigma and functional impairment, is not always the priority. It is in this space that dermatologists are uniquely positioned to advocate for management approaches that address the morbidity associated with skin NTDs. We have an intimate understanding of how impactful skin diseases can be, even if they are not commonly fatal. Globally, skin diseases are the fourth leading cause of nonfatal disease burden,14 yet dermatology lacks effective evidence-based interventions for reducing stigma in our patients with visible chronic diseases.15
Every day, we see firsthand how skin diseases affect not only our patients but also their families, friends, and caregivers. Although we may not see skin NTDs on a regular basis in our clinics, we can understand almost intuitively how devastating skin NTDs could be on individuals, families, and communities. For patients with skin NTDs, receiving medical therapy is only one component of treatment. In addition to optimizing early diagnosis and treatment, interventions taken to educate families and communities affected by skin NTDs are vitally important. Stigma reduction is possible, as we have seen from the aforementioned interventions used in communities with lymphatic filariasis, onchocerciasis, and leprosy. We call upon our fellow dermatologists to take interest in creating, evaluating, and promoting interventions that address stigma in skin NTDs; it is critical in achieving and maintaining health and well-being for our patients.
- Neglected tropical diseases. World Health Organization website. https://www.who.int/neglected_diseases/diseases/en/. Accessed September 10, 2019.
- Hotez PJ, Damania A, Naghavi M. Blue Marble Health and the Global Burden of Disease Study 2013. PLoS Negl Trop Dis. 2016;10:E0004744.
- Skin NTDs. World Health Organization website. https://www.who.int/neglected_diseases/skin-ntds/en/. Accessed September 10, 2019.
- Government of Western Australia Department of Health. Stigma, discrimination and mental illness. February 2009. http://www.health.wa.gov.au/docreg/Education/Population/Health_Problems/Mental_Illness/Mentalhealth_stigma_fact.pdf. Accessed September 10, 2019.
- Hotez PJ. Stigma: the stealth weapon of the NTD. PLoS Negl Trop Dis. 2008;2:E230.
- Bennis I, Belaid L, De Brouwere V, et al. “The mosquitoes that destroy your face.” social impact of cutaneous leishmaniasis in Southeastern Morocco, a qualitative study. PLoS One. 2017;12:E0189906.
- Bennis I, Thys S, Filali H, et al. Psychosocial impact of scars due to cutaneous leishmaniasis on high school students in Errachidia province, Morocco. Infect Dis Poverty. 2017;6:46.
- Bailey F, Mondragon-Shem K, Haines LR, et al. Cutaneous leishmaniasis and co-morbid major depressive disorder: a systematic review with burden estimates. PLoS Negl Trop Dis. 2019;13:E0007092.
- Hofstraat K, van Brakel WH. Social stigma towards neglected tropical diseases: a systematic review. Int Health. 2016;8(suppl 1):I53-I70.
- Cassidy T, Worrell CM, Little K, et al. Experiences of a community-based lymphedema management program for lymphatic filariasis in Odisha State, India: an analysis of focus group discussions with patients, families, community members and program volunteers. PLoS Negl Trop Dis. 2016;10:E0004424.
- Tchounkeu YF, Onyeneho NG, Wanji S, et al. Changes in stigma and discrimination of onchocerciasis in Africa. Trans R Soc Trop Med Hyg. 2012;106:340-347.
- Dadun D, Van Brakel WH, Peters RMH, et al. Impact of socio-economic development, contact and peer counselling on stigma against persons affected by leprosy in Cirebon, Indonesia—a randomised controlled trial. Lepr Rev. 2017;88:2-22.
- Kumar A, Lambert S, Lockwood DNJ. Picturing health: a new face for leprosy. Lancet. 2019;393:629-638.
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134:1527-1534.
- Topp J, Andrees V, Weinberger NA, et al. Strategies to reduce stigma related to visible chronic skin diseases: a systematic review [published online June 8, 2019]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.15734.
- Neglected tropical diseases. World Health Organization website. https://www.who.int/neglected_diseases/diseases/en/. Accessed September 10, 2019.
- Hotez PJ, Damania A, Naghavi M. Blue Marble Health and the Global Burden of Disease Study 2013. PLoS Negl Trop Dis. 2016;10:E0004744.
- Skin NTDs. World Health Organization website. https://www.who.int/neglected_diseases/skin-ntds/en/. Accessed September 10, 2019.
- Government of Western Australia Department of Health. Stigma, discrimination and mental illness. February 2009. http://www.health.wa.gov.au/docreg/Education/Population/Health_Problems/Mental_Illness/Mentalhealth_stigma_fact.pdf. Accessed September 10, 2019.
- Hotez PJ. Stigma: the stealth weapon of the NTD. PLoS Negl Trop Dis. 2008;2:E230.
- Bennis I, Belaid L, De Brouwere V, et al. “The mosquitoes that destroy your face.” social impact of cutaneous leishmaniasis in Southeastern Morocco, a qualitative study. PLoS One. 2017;12:E0189906.
- Bennis I, Thys S, Filali H, et al. Psychosocial impact of scars due to cutaneous leishmaniasis on high school students in Errachidia province, Morocco. Infect Dis Poverty. 2017;6:46.
- Bailey F, Mondragon-Shem K, Haines LR, et al. Cutaneous leishmaniasis and co-morbid major depressive disorder: a systematic review with burden estimates. PLoS Negl Trop Dis. 2019;13:E0007092.
- Hofstraat K, van Brakel WH. Social stigma towards neglected tropical diseases: a systematic review. Int Health. 2016;8(suppl 1):I53-I70.
- Cassidy T, Worrell CM, Little K, et al. Experiences of a community-based lymphedema management program for lymphatic filariasis in Odisha State, India: an analysis of focus group discussions with patients, families, community members and program volunteers. PLoS Negl Trop Dis. 2016;10:E0004424.
- Tchounkeu YF, Onyeneho NG, Wanji S, et al. Changes in stigma and discrimination of onchocerciasis in Africa. Trans R Soc Trop Med Hyg. 2012;106:340-347.
- Dadun D, Van Brakel WH, Peters RMH, et al. Impact of socio-economic development, contact and peer counselling on stigma against persons affected by leprosy in Cirebon, Indonesia—a randomised controlled trial. Lepr Rev. 2017;88:2-22.
- Kumar A, Lambert S, Lockwood DNJ. Picturing health: a new face for leprosy. Lancet. 2019;393:629-638.
- Hay RJ, Johns NE, Williams HC, et al. The global burden of skin disease in 2010: an analysis of the prevalence and impact of skin conditions. J Invest Dermatol. 2014;134:1527-1534.
- Topp J, Andrees V, Weinberger NA, et al. Strategies to reduce stigma related to visible chronic skin diseases: a systematic review [published online June 8, 2019]. J Eur Acad Dermatol Venereol. doi:10.1111/jdv.15734.