User login
Doug Brunk is a San Diego-based award-winning reporter who began covering health care in 1991. Before joining the company, he wrote for the health sciences division of Columbia University and was an associate editor at Contemporary Long Term Care magazine when it won a Jesse H. Neal Award. His work has been syndicated by the Los Angeles Times and he is the author of two books related to the University of Kentucky Wildcats men's basketball program. Doug has a master’s degree in magazine journalism from the S.I. Newhouse School of Public Communications at Syracuse University. Follow him on Twitter @dougbrunk.
Treatment options for lentigo maligna far from perfect
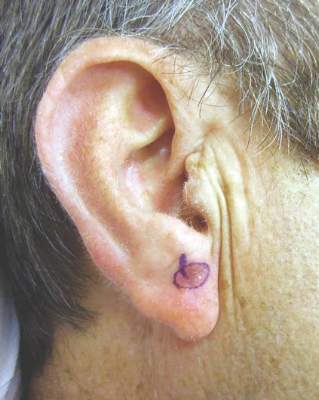
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
PARK CITY, UTAH – Dr. Glen M. Bowen has been working to improve the surgical treatment of lentigo maligna ever since he joined the Huntsman Cancer Institute at the University of Utah 16 years ago. A retrospective review from Memorial Sloan-Kettering Cancer Center found that on average, 7.1-mm margins are required to remove lentigo maligna (LM) (J. Am. Acad. Dermatol. 2008;58[1]:142-8).
“If you have an LM with a 10-mm diameter to begin with, 7.1-mm margins give you a final surgical diameter of 24.2 mm,” Dr. Bowen said at the annual meeting of the Pacific Dermatologic Association. “These are very morbid surgeries in cosmetically sensitive areas for a relatively low-risk tumor.”
The risk of LM progressing to an invasive melanoma is not known, but is estimated to range between 5% and 33%. Of 2,016 patients treated for LM at the Huntsman Cancer Institute, 522 have been treated with neo-adjuvant topical imiquimod 5% cream followed by a conservative staged excision with 2-mm margins with a recurrence rate of 2.3% during median follow-up of 5 years. Of their recurrences, about 20% recurred with invasion. All recurrences to date have been less than 1 mm in depth (stage IA), which has an estimated mortality risk of 5% at 5 years. “A 5% mortality rate of the 20% that recur with invasion of the 2.3% that recur after surgery yields a mortality risk of 0.023%,” Dr. Bowen said. “Due to the very low risk of actually dying from a recurrent LM, very large and morbid surgical defects strike me as a punishment that doesn’t fit the crime in terms of the cost-benefit ratio.”
He and his associates at the Huntsman Cancer Institute have observed some deaths in patients who presented with LM melanoma (invasive melanoma) but have not observed a single death in patients who presented with LM in situ that subsequently recurred. For these reasons, Dr. Bowen favors pretreating LM with imiquimod 5% cream followed by a conservative staged excision, a process that substantially decreases the size of the surgical defects.
His current treatment protocol involves a five-step process that begins with removing all of the visible lentigo maligna to rule out invasion, since 16% of LMs referred to him have harbored invasion when removed and are upgraded from stage 0 to IA. “I am not going to use a topical cream on an invasive melanoma,” he said. “After an excisional biopsy with minimal margins, I usually close the defect with a purse-string suture because it avoids removing standing cones and consequently enlarging the treatment area.”
Second, he traces a template of the LM border on transparent plastic and places a tiny tattoo in the center of the biopsy site to enable pinpoint placement of the template at the time of surgery.
Third, he treats the site with imiquimod 5% cream Monday through Friday for 2-3 months and sees the patient monthly for dosage adjustments when needed. The fourth step involves enabling the site to recover for 2-6 months to allow for resolution of the inflammatory infiltrate. The final step involves re-excising around the original template with 2-mm margins for confirmation with the use of a negative control taken from an equally sun-exposed site taken some distance away from the LM. “Caucasians will have atypical junctional melanocytic hyperplasia (AJMH), which must be subtracted out as background,” he said. “Otherwise, if you hold a non–sun-exposed site as your standard for a negative margin, you will never stop cutting.”
Dr. Bowen likes to use frozen radial sections with routine staining with H and E and immunostaining with MART-1 (Melan-A) and SOX-10. Processing takes 2 hours, he continued, “so I put in relaxing sutures, which will stretch out nicely over 2 hours so I can usually close the defects primarily.”
In Dr. Bowen’s opinion, topical imiquimod as monotherapy for LM is not safe, since about 30% of patients treated with imiquimod will still harbor residual LM. “In our dataset, about 70% have no residual LM, 20% have residual LM in the center but negative perimeter margins, and 10% have LM touching a perimeter margin and require a second stage,” he said. “Taken together, 90% of patients pretreated with imiquimod will be cleared in one stage of surgery with 2 mm margins.”
Making the distinction between LM and AJMH common to chronically sun-damaged skin is no easy task. Dr. Bowen cited a concordance study between dermatopathologists interpreting staged excisional margins on permanent sections for LM where the concordance was only moderate at best. In this study, the use of a negative control improved the concordance rate on “difficult” cases from 46% to 76%; P = .001 (Arch Dermatol. 2003 May;139(5):595-604). “What we really need is a molecular marker that will tell us if a melanocyte is malignant or not,” he said. “All we have now are immunostains that tell you if it’s melanocyte but nothing more.” He went on to say that in multivariate analysis in two studies of the histologic features of LM, the only feature that consistently predicted the difference between LM and AJMH was the melanocyte density and its ratio to the negative control (Dermatol Surg. 2011;37(5):657-63 and J. Plast. Reconstr. Aesthet. Surg. 2014;67(10):1322-32). “The MART-1 immunostain is extremely sensitive, but it makes the slide somewhat muddy, so it’s hard to do an accurate cell count,” he said. “For that reason, we also use a SOX-10 immunostain which is very specific but not as sensitive. I believe that the truth lies somewhere in between those two immunostains in light of a positive control from our lab and a negative control from the patient.”
He concluded that the neoadjuvant use of imiquimod followed by a conservative staged excision “allows me to clear 90% of LM with a 2 mm margin with a recurrence rate of 2.3% in patients with a mean follow-up of 5-years or greater.”
Dr. Bowen reported having no financial disclosures.
EXPERT ANALYSIS from PDA 2015
Biologic Treatment for Psoriasis Found to Have CVD Benefits
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
EXPERT ANALYSIS AT PDA 2015
Biologic treatment for psoriasis found to have CVD benefits
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
PARK CITY, UTAH – Evidence is mounting that the use of biologic therapies is associated with certain cardiovascular benefits in patients with psoriasis.
However, until the strength of the evidence becomes more robust, Dr. Jashin J. Wu does not recommend that patients are prescribed tumor necrosis factor (TNF) inhibitors specifically to reduce the risk of cardiovascular disease. “If systemic therapy is a consideration in the setting of CVD [cardiovascular disease] risk, it would appear that TNF inhibitors and methotrexate offer the best evidence of benefit,” Dr. Wu, director of the psoriasis clinic and director of dermatology research in the department of dermatology at Kaiser Permanente Los Angeles Medical Center, said at the annual meeting of the Pacific Dermatologic Association.
The National Psoriasis Foundation recently published a consensus statement about the potential impact of current therapies on cardiovascular disease, including the risk of diabetes (J Am Acad Dermatol. 2014;70[1]:168-77). In a retrospective study of 121,280 patients with rheumatoid arthritis (RA) or psoriasis, drug regimens were categorized into four mutually exclusive groups: TNF inhibitors with or without other disease-modifying antirheumatic drugs (DMARDs), methotrexate without TNF inhibitors or hydroxychloroquine, hydroxychloroquine without TNF inhibitors or methotrexate; and other nonbiologic DMARDs without TNF inhibitors, methotrexate or hydroxychloroquine (referent group). The researchers found that adjusted Cox proportional hazards for the risk of diabetes were lower among those on TNF inhibitors (hazard ratio, 0.62; 95% confidence interval, 0.42-0.91) and hydroxychloroquine (HR, 0.54; 95% CI, 0.36-0.80), compared with those on methotrexate (HR, 0.77, 95% CI, 0.53-1.13) (JAMA. 2011 Jun 22;305[24]:2525-31).
Increased carotid intimal media thickness (IMT) is also an independent predictor of CVD. In a prospective, observational study, 20 psoriasis patients received a 12-week treatment of TNF inhibitors (Ann Rheum Dis. 2011 Apr;70[4]:705-6). After 12 weeks, 9 patients continued on their TNF-inhibitor regimen (group 1), while 11 discontinued treatment because of financial constraints (group 2). Another 20 psoriasis patients who did not receive biologic therapy served as controls (group 3). The researchers measured IMT by carotid ultrasound at baseline, week 12 and at 2 years. They found that between baseline and 2 years, IMT decreased from 0.70 mm to 0.63 mm in group 1, from 0.71 to 0.67 in group 2, and increased from 0.79 to 0.82 in group 3. “This seems to suggest that the use of TNF inhibitors may improve atherosclerosis,” said Dr. Wu, who is currently running seven clinical trials of psoriasis patients. Similar findings were seen in a larger study that examined 224 patients with psoriatic arthritis and followed them for a mean of 55 months: 124 on TNF inhibitors, 104 on DMARDs, and 305 matched controls (Arterioscler Thromb Vasc Biol. 2011 Mar;31[3]:705-120).
In a separate study of 16 psoriasis patients treated with TNF inhibitors, researchers used ultrasound to measure IMT of carotid and brachial arteries after 6 months of therapy (J Am Acad Dermatol. 2013 Oct;69[4]:523-9). At baseline, all 16 patients had an IMT greater than normal. In those without initial calcified atherosclerotic plaques, 13 of 16 had a significant decrease of IMT (P = .0002). In those with initial calcified atherosclerotic plaques, 3 of 16 patients had a nonsignificant increase in IMT.
Increased arterial stiffness is another independent predictor of CVD. In a controlled study of 55 patients with psoriatic arthritis, RA, or ankylosing spondylitis, researchers used aortic pulse-wave velocity to measure aortic stiffness. Almost 35% of the patients (36) received TNF inhibitors while the remaining 19 did not receive treatment (Am J Hypertens. 2012 Jun;25[6]:644-50). After 1 year of follow-up, use of TNF inhibitors was associated with a significant improvement in aortic pulse-wave velocity (P = .02) and reduced carotid IMT progression (P = .04).
Other evidence suggests that systemic therapy with TNF inhibitors is associated with the reduction of cardiovascular events, including myocardial infarction. In a study led by Dr. Wu, 8,845 Kaiser Permanente patients with psoriasis were evaluated between Jan. 1, 2004, and Nov. 30, 2010, and placed into one of three groups: treatment with a TNF inhibitor, treatment with oral therapy or phototherapy, or treatment with a topical agent (referent group) (Arch Dermatol. 2012 Nov;148[11]:1244-50). On multivariate analysis, the investigators found that the use of TNF inhibitors was associated with a significant reduction in the rate of incident MI (HR, 0.50; P = .003), compared with the use of topical agents.
Dr. Wu went on to note that C-reactive protein (CRP) is a predictor of CVD such as MI, peripheral arterial disease, and sudden cardiac death. In a retrospective cohort study, Dr. Wu and his associates evaluated Kaiser Permanente patients with psoriasis, psoriatic arthritis, or RA who were treated with a combination of TNF plus methotrexate, or with methotrexate alone from Jan. 1, 2002, to July 31, 2011 (J. Am. Acad. Dermatol. 2015;72[5]:917-9). Only patients in the combination therapy group had a clinically and statistically significant decrease in CRP, compared with baseline (a mean decrease of 5.18 mg/dL). No significant changes between the two groups were observed in terms of BMI, blood pressure, fasting glucose, hemoglobin A1C, total cholesterol, LDL cholesterol, HDL cholesterol, triglycerides, or alanine aminotransferase.
HDL cholesterol composition and function have also been studied in psoriasis patients. In one trial, HDL cholesterol was isolated from 15 psoriasis patients at baseline and after effective topical and/or systemic psoriasis therapy, and from 15 healthy controls matched for age and sex (J. Invest. Dermatol. 2014;134[3]:635-42). The researchers found that HDL cholesterol from psoriasis patients showed a significantly impaired capability to mobilize cholesterol from macrophages. However, psoriasis therapy recovered HDL cholesterol composition and function, but had no effect on serum HDL cholesterol levels.
A better understanding of the cardiovascular effects of biologic therapy should be gleaned from the Vascular Inflammation in Psoriasis Trial, which includes $3.8 million in funding from the National Institutes of Health, to prospectively study the effect of therapy on biomarkers and on positron emission tomography with 18fluorodeoxyglucose (FDG-PET)/CT scan. The 1-year study just completed enrolling patients into one of three treatment groups: adalimumab, UVB phototherapy, or placebo. Biomarkers such as CRP, lipids, and imaging will be assessed at baseline, weeks 4, 8, 12, and then every 12 weeks. “The theory is that adalimumab, but not UVB phototherapy, will improve biomarkers and reduce vascular inflammation as measured by FDG-PET/CT scan,” Dr. Wu said. “Aggressive systemic therapy may alter the natural history of cardiovascular disease.”
Current recommendations from the National Psoriasis Foundation call for an assessment of blood pressure, pulse, and body mass index every 2 years, and an assessment of fasting blood glucose and lipid levels every 5 years, or every 2 years if the patient has additional risk factors.
Dr. Wu reported that he has received research and consulting fees from numerous pharmaceutical companies, including AbbVie and Amgen. These go to his employer.
EXPERT ANALYSIS AT PDA 2015
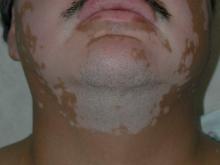
Top 10 treatments for vitiligo
PARK CITY, UTAH – At the annual meeting of the Pacific Dermatologic Association, Dr. Sancy A. Leachman offered a top 10 list of new agents and technologies for the treatment of vitiligo.
No. 10: Ultraviolet A1 (UVA1) phototherapy
Dr. Harvey Lui at the University of British Columbia in Vancouver is leading a phase II trial to evaluate the potential for UVA1 to induce repigmentation within vitiligo patches and to assess the side effect profile of the treatment. “I think it might work,” said Dr. Leachman, professor and chair of dermatology at Oregon Health & Science University (OHSU), Portland.
No. 9: Ginkgo biloba
The use of ginko biloba 40-60 mg 2-3 times per day, 10 minutes before a meal, was mentioned in a Cochrane Review of vitiligo treatments published on Feb. 24, 2015. “I think I’m going to give this a try in people who have failed other treatments and see if I can get some response,” Dr. Leachman said.
No. 8: Red light
Dr. Lui is leading a randomized phase II trial of low-intensity and high-intensity red light versus no treatment for vitiligo patches. Treatments will be given twice weekly for 10 weeks, with follow-up assessments at 4, 8, and 12 weeks post treatment.
No. 7: Micrografting
A novel suction blister device known as the CelluTome epidermal harvesting system uses heat and slight vacuum pressure to harvest healthy epidermal skin tissue without damaging the donor site. Dr. Leachman characterized the technology as “semiautomating the process of suction graft transplantation.”
No. 6: The ReCell device
Manufactured by Avita Medical, this investigational autologous cell harvesting device is used after CO2 abrasion and enables clinicians to create regenerative epithelial suspension with a small sample of the patient’s skin. A phase IV trial in the Netherlands is underway to assess the efficacy and safety of autologous epidermal cell suspension grafting with the ReCell device after CO2 laser abrasion, compared with CO2 laser abrasion alone and no treatment, in patients with piebaldism and stable vitiligo.
No. 5: Topical Photocil
In a pilot study sponsored by Applied Biology, researchers are enrolling patients with vitiligo to assess the safety and efficacy of Photocil. The primary outcome measure is the Vitiligo Area Severity Index (VASI). “When this cream is activated by sunlight, it degrades into narrow-band and UVB light, so you can put a topical cream on that will administer narrow-band UVB only in that spot,” said Dr. Leachman, who is also director of OHSU’s Knight Melanoma Research Program. “That’s amazing to me.”
No. 4: Afamelanotide
This is an analogue of a melanocyte-stimulating hormone. A randomized study conducted at two academic medical centers found that the combination of afamelanotide implant and narrow-band UVB phototherapy resulted in statistically superior and faster repigmentation, compared with narrow-band UVB monotherapy (JAMA Dermatol. 2015 Jan;151(1):42-50).
No. 3: Abatacept (Orencia)
This is a soluble fusion protein consisting of human cytotoxic T-lymphocyte–associated antigen 4 (CTLA4), which prevents T-cell activation. A phase I trial is underway at Brigham and Women’s Hospital in Boston to determine if weekly self-injections of the agent lead to clinical improvements of vitiligo lesions. The primary outcome measure is change in repigmentation with abatacept therapy based on the VASI score.
No. 2. Simvastatin
The notion of its use is based on STAT1 inhibition reducing interferon-gamma–dependent activation of CD8-positive T cells, according to Dr. Leachman. The concept has been successful in a mouse model, and a study in humans was recently completed by Dr. John Harris at the University of Massachusetts, Worcester. “What we have is the ability to apply an existing drug (Simvastatin) to the process and see if it works,” she said. “Wouldn’t it be cool if we could give a statin and improve vitiligo?”
No 1: Tofacitinib
This is a Janus kinase inhibitor commonly used for rheumatoid arthritis. According to Dr. Leachman, Janus kinase inhibition prevents STAT activation, “which prevents [interferon]-gamma production, which reduces activation of CD8-positive T cells via CXCL10 binding to CXCR3,” she said. A case report demonstrating its efficacy in a 53-year-old patient was recently published in JAMA Dermatology by Dr. Brett A. King and Dr. Brittany Craiglow, dermatologists at Yale School of Medicine, New Haven, Conn. “I’m hopeful that this [agent] will be made into a topical cream because these drugs do have substantial side effects,” Dr. Leachman said.
Dr. Leachman disclosed that she is a member of the medical and scientific advisory board for Myriad Genetics Laboratory. She has also participated in an advisory board meeting for Castle Biosciences and has participated in the DecisionDx registry.
PARK CITY, UTAH – At the annual meeting of the Pacific Dermatologic Association, Dr. Sancy A. Leachman offered a top 10 list of new agents and technologies for the treatment of vitiligo.
No. 10: Ultraviolet A1 (UVA1) phototherapy
Dr. Harvey Lui at the University of British Columbia in Vancouver is leading a phase II trial to evaluate the potential for UVA1 to induce repigmentation within vitiligo patches and to assess the side effect profile of the treatment. “I think it might work,” said Dr. Leachman, professor and chair of dermatology at Oregon Health & Science University (OHSU), Portland.
No. 9: Ginkgo biloba
The use of ginko biloba 40-60 mg 2-3 times per day, 10 minutes before a meal, was mentioned in a Cochrane Review of vitiligo treatments published on Feb. 24, 2015. “I think I’m going to give this a try in people who have failed other treatments and see if I can get some response,” Dr. Leachman said.
No. 8: Red light
Dr. Lui is leading a randomized phase II trial of low-intensity and high-intensity red light versus no treatment for vitiligo patches. Treatments will be given twice weekly for 10 weeks, with follow-up assessments at 4, 8, and 12 weeks post treatment.
No. 7: Micrografting
A novel suction blister device known as the CelluTome epidermal harvesting system uses heat and slight vacuum pressure to harvest healthy epidermal skin tissue without damaging the donor site. Dr. Leachman characterized the technology as “semiautomating the process of suction graft transplantation.”
No. 6: The ReCell device
Manufactured by Avita Medical, this investigational autologous cell harvesting device is used after CO2 abrasion and enables clinicians to create regenerative epithelial suspension with a small sample of the patient’s skin. A phase IV trial in the Netherlands is underway to assess the efficacy and safety of autologous epidermal cell suspension grafting with the ReCell device after CO2 laser abrasion, compared with CO2 laser abrasion alone and no treatment, in patients with piebaldism and stable vitiligo.
No. 5: Topical Photocil
In a pilot study sponsored by Applied Biology, researchers are enrolling patients with vitiligo to assess the safety and efficacy of Photocil. The primary outcome measure is the Vitiligo Area Severity Index (VASI). “When this cream is activated by sunlight, it degrades into narrow-band and UVB light, so you can put a topical cream on that will administer narrow-band UVB only in that spot,” said Dr. Leachman, who is also director of OHSU’s Knight Melanoma Research Program. “That’s amazing to me.”
No. 4: Afamelanotide
This is an analogue of a melanocyte-stimulating hormone. A randomized study conducted at two academic medical centers found that the combination of afamelanotide implant and narrow-band UVB phototherapy resulted in statistically superior and faster repigmentation, compared with narrow-band UVB monotherapy (JAMA Dermatol. 2015 Jan;151(1):42-50).
No. 3: Abatacept (Orencia)
This is a soluble fusion protein consisting of human cytotoxic T-lymphocyte–associated antigen 4 (CTLA4), which prevents T-cell activation. A phase I trial is underway at Brigham and Women’s Hospital in Boston to determine if weekly self-injections of the agent lead to clinical improvements of vitiligo lesions. The primary outcome measure is change in repigmentation with abatacept therapy based on the VASI score.
No. 2. Simvastatin
The notion of its use is based on STAT1 inhibition reducing interferon-gamma–dependent activation of CD8-positive T cells, according to Dr. Leachman. The concept has been successful in a mouse model, and a study in humans was recently completed by Dr. John Harris at the University of Massachusetts, Worcester. “What we have is the ability to apply an existing drug (Simvastatin) to the process and see if it works,” she said. “Wouldn’t it be cool if we could give a statin and improve vitiligo?”
No 1: Tofacitinib
This is a Janus kinase inhibitor commonly used for rheumatoid arthritis. According to Dr. Leachman, Janus kinase inhibition prevents STAT activation, “which prevents [interferon]-gamma production, which reduces activation of CD8-positive T cells via CXCL10 binding to CXCR3,” she said. A case report demonstrating its efficacy in a 53-year-old patient was recently published in JAMA Dermatology by Dr. Brett A. King and Dr. Brittany Craiglow, dermatologists at Yale School of Medicine, New Haven, Conn. “I’m hopeful that this [agent] will be made into a topical cream because these drugs do have substantial side effects,” Dr. Leachman said.
Dr. Leachman disclosed that she is a member of the medical and scientific advisory board for Myriad Genetics Laboratory. She has also participated in an advisory board meeting for Castle Biosciences and has participated in the DecisionDx registry.
PARK CITY, UTAH – At the annual meeting of the Pacific Dermatologic Association, Dr. Sancy A. Leachman offered a top 10 list of new agents and technologies for the treatment of vitiligo.
No. 10: Ultraviolet A1 (UVA1) phototherapy
Dr. Harvey Lui at the University of British Columbia in Vancouver is leading a phase II trial to evaluate the potential for UVA1 to induce repigmentation within vitiligo patches and to assess the side effect profile of the treatment. “I think it might work,” said Dr. Leachman, professor and chair of dermatology at Oregon Health & Science University (OHSU), Portland.
No. 9: Ginkgo biloba
The use of ginko biloba 40-60 mg 2-3 times per day, 10 minutes before a meal, was mentioned in a Cochrane Review of vitiligo treatments published on Feb. 24, 2015. “I think I’m going to give this a try in people who have failed other treatments and see if I can get some response,” Dr. Leachman said.
No. 8: Red light
Dr. Lui is leading a randomized phase II trial of low-intensity and high-intensity red light versus no treatment for vitiligo patches. Treatments will be given twice weekly for 10 weeks, with follow-up assessments at 4, 8, and 12 weeks post treatment.
No. 7: Micrografting
A novel suction blister device known as the CelluTome epidermal harvesting system uses heat and slight vacuum pressure to harvest healthy epidermal skin tissue without damaging the donor site. Dr. Leachman characterized the technology as “semiautomating the process of suction graft transplantation.”
No. 6: The ReCell device
Manufactured by Avita Medical, this investigational autologous cell harvesting device is used after CO2 abrasion and enables clinicians to create regenerative epithelial suspension with a small sample of the patient’s skin. A phase IV trial in the Netherlands is underway to assess the efficacy and safety of autologous epidermal cell suspension grafting with the ReCell device after CO2 laser abrasion, compared with CO2 laser abrasion alone and no treatment, in patients with piebaldism and stable vitiligo.
No. 5: Topical Photocil
In a pilot study sponsored by Applied Biology, researchers are enrolling patients with vitiligo to assess the safety and efficacy of Photocil. The primary outcome measure is the Vitiligo Area Severity Index (VASI). “When this cream is activated by sunlight, it degrades into narrow-band and UVB light, so you can put a topical cream on that will administer narrow-band UVB only in that spot,” said Dr. Leachman, who is also director of OHSU’s Knight Melanoma Research Program. “That’s amazing to me.”
No. 4: Afamelanotide
This is an analogue of a melanocyte-stimulating hormone. A randomized study conducted at two academic medical centers found that the combination of afamelanotide implant and narrow-band UVB phototherapy resulted in statistically superior and faster repigmentation, compared with narrow-band UVB monotherapy (JAMA Dermatol. 2015 Jan;151(1):42-50).
No. 3: Abatacept (Orencia)
This is a soluble fusion protein consisting of human cytotoxic T-lymphocyte–associated antigen 4 (CTLA4), which prevents T-cell activation. A phase I trial is underway at Brigham and Women’s Hospital in Boston to determine if weekly self-injections of the agent lead to clinical improvements of vitiligo lesions. The primary outcome measure is change in repigmentation with abatacept therapy based on the VASI score.
No. 2. Simvastatin
The notion of its use is based on STAT1 inhibition reducing interferon-gamma–dependent activation of CD8-positive T cells, according to Dr. Leachman. The concept has been successful in a mouse model, and a study in humans was recently completed by Dr. John Harris at the University of Massachusetts, Worcester. “What we have is the ability to apply an existing drug (Simvastatin) to the process and see if it works,” she said. “Wouldn’t it be cool if we could give a statin and improve vitiligo?”
No 1: Tofacitinib
This is a Janus kinase inhibitor commonly used for rheumatoid arthritis. According to Dr. Leachman, Janus kinase inhibition prevents STAT activation, “which prevents [interferon]-gamma production, which reduces activation of CD8-positive T cells via CXCL10 binding to CXCR3,” she said. A case report demonstrating its efficacy in a 53-year-old patient was recently published in JAMA Dermatology by Dr. Brett A. King and Dr. Brittany Craiglow, dermatologists at Yale School of Medicine, New Haven, Conn. “I’m hopeful that this [agent] will be made into a topical cream because these drugs do have substantial side effects,” Dr. Leachman said.
Dr. Leachman disclosed that she is a member of the medical and scientific advisory board for Myriad Genetics Laboratory. She has also participated in an advisory board meeting for Castle Biosciences and has participated in the DecisionDx registry.
EXPERT ANALYSIS AT PDA 2015
Health care transition practices continue to evolve
It’s been nearly 13 years since the American Academy of Pediatrics, the American Academy of Family Physicians, and the American College of Physicians released its Consensus Policy Statement on Health Care Transitions for Young Adults With Special Needs and 4 years since the AAP, AAFP, ACP Clinical Report: Supporting Health Care Transition (HCT) from Adolescence to Adulthood in the Medical Home was published, but formal adoption of suggested approaches has remained minimal.
A recent survey of 158 members of the Childhood Arthritis and Rheumatology Research Alliance in North America found that fewer than 10% of respondents were familiar with the consensus statement and the HCT clinical report, only 8% have a formal written transition policy, while 42% use an informal approach (J. Rheumatol. 2014;41:768-79). Respondents ranked fragmented adult health care and lack of time to provide sufficient services as the two major barriers to transition.
“Until recently, there hasn’t been a lot of focus on transition in fellowship training, and I don’t know how well the consensus policy statement and HCT clinical report are publicized in residency or fellowship training,” one of the study authors, Dr. Tova Ronis, said in an interview. “There’s definitely a role for more education about the policy statement and HCT clinical report.” As a result of inconsistent transition practices, some patients fall through the cracks in the transition from pediatric to adult rheumatology care. Transition can be difficult and even patients who receive transition care can have challenges, said Dr. Ronis of the pediatric rheumatology division at Children’s National Health System, Washington. She spoke of one young patient with severe systemic disease who was referred to ongoing adult care with an expert in the field. The patient “went to the adult office but didn’t like it there; she didn’t feel like she was being listened to and didn’t feel like the new doctor knew her well. She would come back to us, go back and forth, so her care became very fragmented. We communicated with the adult doctor but there wasn’t one person making the plan [of care].” In addition, other clinicians who had been providing subspecialty care and had not transitioned her to adult practice “were still at the children’s hospital, so when she experienced health problems or pain, she would come to the pediatric emergency department,” Dr. Ronis recalled. “She hadn’t identified the new institution as her medical home, so her care became very fragmented during that transition process. She actually missed some therapy because she didn’t like the new office, so wasn’t seen anywhere for a while.”
Dr. Paul T. Jensen said that most clinicians “have a gut feeling” that the current health care transition landscape has limitations, despite the availability of such tools as the Got Transition website, the Transition Readiness Assessment Questionnaire (TRAQ), as well as studies of novel approaches such as having a social worker serve as a transition coordinator(Pediatr. Rheumatol. 2015;13:17 [doi:10.1186/s12969-015-0013-0]) or having clinicians from adult practices see patients at a pediatric institution (Pediatr. Pulmonol. 2013;48:658-65). “I think that most, if not all, pediatric providers, including pediatric rheumatologists, worry about their patients as they get older, especially those who have a decreased maturity level,” said Dr. Jensen, who is a fellow in both adult and pediatric rheumatology at the Ohio State University Wexner Medical Center and Nationwide Children’s Hospital, Columbus. “But I think there’s a large lack of knowledge about what resources are available.”
Choosing transition practices that work for you
Dr. Patience White, a pediatric and adult rheumatologist who in 2011 coauthored the HCT Clinical Report, is familiar with such sentiment. She says that the topic of transition has “come to the forefront because the pediatric rheumatology field has grown. On top of that, many of our patients have grown up and moved on to other aspects of adulthood, so now pediatric providers will need to prepare to ‘let them go to adult health care.’ The perfect storm is happening.” Dr. White codirects the Got Transition Center for Health Care Transition Improvement, a federally funded program located at the National Alliance to Advance Adolescent Health. One of the center’s goals is to expand the use of the Six Core Elements of Health Care Transition that were developed to help make the 2011 Health Care Transitions Clinical Report actionable. These include practice quality improvement strategies such as establishing a transition policy, tracking progress, offering transition readiness assessments, creating a transition plan, transferring to an adult provider, and assessing the youth’s and family’s experience with the transition. The original Six Core Elements of Health Care Transition were tested in learning collaboratives across the United States and have been updated with input from the learning collaboratives and national transition experts into three separate packages of tools and evaluation approaches for transitioning youth to adult health care providers, transitioning to an adult approach to health care without changing providers, and integrating young adults into adult health care. For example, recommendations when transitioning youth to adult health care providers include developing a transition policy/statement “with input from youth and families that describes the practice’s approach to transition, including privacy and consent information,” as well as conducting regular transition readiness assessments, beginning at age 14 years, “to identify and discuss with youth and parent/caregiver their needs and goals in self-care.” Other tools available from the Got Transition website include a set of frequently asked transition questions developed by and for youth/young adults, as well as many transition resources.
While she acknowledged it may not be feasible for pediatric and adult rheumatologists to implement all Six Core Elements in their practice, Dr. White suggests providers might try implementing at least one or two and, for pediatric rheumatologists, start the discussion of the transition process when their patients reach the age of 13 years as recommended by the HCT clinical report. “Everybody says it’s hard, and I agree, changing practice processes to improve health care is hard work,” she said. “We know preparation for the youth and family needs to happen. We know transfer and communication has to occur, and that the accepting adult provider has to be informed and comfortable to make the transition process a success. It is about choosing one or two Six Core Element tools that can work for you, your practice, and your patients to help them through the transition process. If you’re an adult provider, you might choose to offer a welcome letter to my practice and a self-care assessment for the new young adult patients joining the practice so the adult health care provider can understand how proficient the young adults are with respect to their self-care skills and knowledge of the adult health care system.”
Disease-specific toolkits coming in 2016
Dr. White is also a consultant to the Pediatric to Adult Health Care Transitions Workgroup, an effort of the ACP Council of Subspecialty Societies to address gaps in care that exist with the transition of pediatric patients into adult health care. According to an ACP document describing the effort, the goal of workgroup is to identify “common tools/templates for a ‘general’ transition process, and subspecialty societies will utilize the templates to develop customized toolkits for specific diseases/conditions. The toolkits will then be collected and broadly disseminated,” with release targeted for May 2016 at the ACP national meeting. Dr. Stacy P. Ardoin, who is leading the rheumatology subspecialty group on behalf of the American College of Rheumatology, said the fact that this effort was initiated by the ACP “is great, because historically most of the interest in transition has bubbled up from the pediatric side. I think that adult providers are realizing more and more that patients who come to them with complex childhood onset conditions are challenging to manage, and that we really need to partner together to make things the best for our patients.”
Dr. Ardoin, a rheumatologist in the department of internal medicine at the Ohio State University Wexner Medical Center and in the department of pediatrics at Nationwide Children’s Hospital, characterized the final product as “not so much a definite protocol that everybody has to follow, but more just providing recommendations to the practitioner saying, ‘these are the things that we recommend and here are some tools to accomplish it.’ There’s such a lack of standardization for anything with transition despite the fact that we have consensus statements that have been put out in the past. Perhaps part of the problem is that they were too vague or not considered individually for each specialty. Hopefully, having each specialty take this on and look at their unique needs in the transition process will make people more willing to participate.”
Preparing patients to become health care self-advocates
Not all physicians are convinced that more recommendations will help clinicians navigate the intricacies of health care transition, however. “If children come to you as a physician at a young age (most of the kids with arthritis) or very ill (most of the children with systemic lupus erythematosus), good care over time represents a significant bonding experience,” said Dr. Thomas J. A. Lehman, chief of the division of pediatric rheumatology at the Hospital for Special Surgery in New York. “The children come to see you as a significant other in their life. If they then are suddenly ‘cast out’ because they reach a certain age it doesn’t go down any better than it would if a significant relative ‘cast them out.’ The doctors like to think they are transitioning the patients. The patients who have bonded feel they are being rejected. I don’t have an answer to this problem. However, none of the past HCT policy statements adequately recognize this aspect.”
For her part, Dr. White, who is also professor of medicine and pediatrics at George Washington University, Washington, said that in some published studies pediatric providers acknowledged that they are reluctant to transition patients to adult health care providers “and can overprotect their young patients as some parents do. This approach can result in the young adult being a poor self-advocate in the adult health care environment and not receiving developmentally appropriate health care. If a pediatric patient and family have not been informed of when they can no longer come to the pediatric rheumatology practice and have not discussed a transition to adult health care plan, they often assume they can stay with their pediatric rheumatologist forever.”
In addition, Dr. White continued, “some pediatric rheumatologists might say, ‘adult providers don’t know how to care for pediatric rheumatology [patients].’ Well, that may due to the adult providers not having received training in specific pediatric onset conditions. Communication between the pediatric and adult providers is key to a good transition experience for everyone. Trying to quickly build a new relationship with a new young adult patient to keep them engaged in their care and who has been used to the same pediatric provider for years is challenging, and the young adult and the adult provider need the pediatric provider’s support and availability to be a consultant to support the adult provider in the areas where they have little disease-specific experience.”
Dr. Jensen makes it a point to talk to his patients about health care transition in early adolescence, helps them set goals, and encourages them to “take more ownership in late adolescence for his or her own health: making their own appointments for follow-up visits, giving time to the patient in the room alone, and asking patients to maybe monitor medication adherence but not to lay the medications out for the patient in the morning,” he said. “When patients are given that opportunity, they may mature.”
Dr. Ronis, who is implementing a health care transition program at Children’s National Health System, thinks about health care transition as “a time where there’s risk for potentially worsening disease and negative outcomes due to perceived fragmentation of care. The more prepared we are and the more we prepare our patients to do this successfully, the better outcomes our patients will have.”
The Got Transition Center for Health Care Transition Improvement is funded by the Health Resources and Services Administration. The physicians interviewed for this story reported having no financial disclosures.
It’s been nearly 13 years since the American Academy of Pediatrics, the American Academy of Family Physicians, and the American College of Physicians released its Consensus Policy Statement on Health Care Transitions for Young Adults With Special Needs and 4 years since the AAP, AAFP, ACP Clinical Report: Supporting Health Care Transition (HCT) from Adolescence to Adulthood in the Medical Home was published, but formal adoption of suggested approaches has remained minimal.
A recent survey of 158 members of the Childhood Arthritis and Rheumatology Research Alliance in North America found that fewer than 10% of respondents were familiar with the consensus statement and the HCT clinical report, only 8% have a formal written transition policy, while 42% use an informal approach (J. Rheumatol. 2014;41:768-79). Respondents ranked fragmented adult health care and lack of time to provide sufficient services as the two major barriers to transition.
“Until recently, there hasn’t been a lot of focus on transition in fellowship training, and I don’t know how well the consensus policy statement and HCT clinical report are publicized in residency or fellowship training,” one of the study authors, Dr. Tova Ronis, said in an interview. “There’s definitely a role for more education about the policy statement and HCT clinical report.” As a result of inconsistent transition practices, some patients fall through the cracks in the transition from pediatric to adult rheumatology care. Transition can be difficult and even patients who receive transition care can have challenges, said Dr. Ronis of the pediatric rheumatology division at Children’s National Health System, Washington. She spoke of one young patient with severe systemic disease who was referred to ongoing adult care with an expert in the field. The patient “went to the adult office but didn’t like it there; she didn’t feel like she was being listened to and didn’t feel like the new doctor knew her well. She would come back to us, go back and forth, so her care became very fragmented. We communicated with the adult doctor but there wasn’t one person making the plan [of care].” In addition, other clinicians who had been providing subspecialty care and had not transitioned her to adult practice “were still at the children’s hospital, so when she experienced health problems or pain, she would come to the pediatric emergency department,” Dr. Ronis recalled. “She hadn’t identified the new institution as her medical home, so her care became very fragmented during that transition process. She actually missed some therapy because she didn’t like the new office, so wasn’t seen anywhere for a while.”
Dr. Paul T. Jensen said that most clinicians “have a gut feeling” that the current health care transition landscape has limitations, despite the availability of such tools as the Got Transition website, the Transition Readiness Assessment Questionnaire (TRAQ), as well as studies of novel approaches such as having a social worker serve as a transition coordinator(Pediatr. Rheumatol. 2015;13:17 [doi:10.1186/s12969-015-0013-0]) or having clinicians from adult practices see patients at a pediatric institution (Pediatr. Pulmonol. 2013;48:658-65). “I think that most, if not all, pediatric providers, including pediatric rheumatologists, worry about their patients as they get older, especially those who have a decreased maturity level,” said Dr. Jensen, who is a fellow in both adult and pediatric rheumatology at the Ohio State University Wexner Medical Center and Nationwide Children’s Hospital, Columbus. “But I think there’s a large lack of knowledge about what resources are available.”
Choosing transition practices that work for you
Dr. Patience White, a pediatric and adult rheumatologist who in 2011 coauthored the HCT Clinical Report, is familiar with such sentiment. She says that the topic of transition has “come to the forefront because the pediatric rheumatology field has grown. On top of that, many of our patients have grown up and moved on to other aspects of adulthood, so now pediatric providers will need to prepare to ‘let them go to adult health care.’ The perfect storm is happening.” Dr. White codirects the Got Transition Center for Health Care Transition Improvement, a federally funded program located at the National Alliance to Advance Adolescent Health. One of the center’s goals is to expand the use of the Six Core Elements of Health Care Transition that were developed to help make the 2011 Health Care Transitions Clinical Report actionable. These include practice quality improvement strategies such as establishing a transition policy, tracking progress, offering transition readiness assessments, creating a transition plan, transferring to an adult provider, and assessing the youth’s and family’s experience with the transition. The original Six Core Elements of Health Care Transition were tested in learning collaboratives across the United States and have been updated with input from the learning collaboratives and national transition experts into three separate packages of tools and evaluation approaches for transitioning youth to adult health care providers, transitioning to an adult approach to health care without changing providers, and integrating young adults into adult health care. For example, recommendations when transitioning youth to adult health care providers include developing a transition policy/statement “with input from youth and families that describes the practice’s approach to transition, including privacy and consent information,” as well as conducting regular transition readiness assessments, beginning at age 14 years, “to identify and discuss with youth and parent/caregiver their needs and goals in self-care.” Other tools available from the Got Transition website include a set of frequently asked transition questions developed by and for youth/young adults, as well as many transition resources.
While she acknowledged it may not be feasible for pediatric and adult rheumatologists to implement all Six Core Elements in their practice, Dr. White suggests providers might try implementing at least one or two and, for pediatric rheumatologists, start the discussion of the transition process when their patients reach the age of 13 years as recommended by the HCT clinical report. “Everybody says it’s hard, and I agree, changing practice processes to improve health care is hard work,” she said. “We know preparation for the youth and family needs to happen. We know transfer and communication has to occur, and that the accepting adult provider has to be informed and comfortable to make the transition process a success. It is about choosing one or two Six Core Element tools that can work for you, your practice, and your patients to help them through the transition process. If you’re an adult provider, you might choose to offer a welcome letter to my practice and a self-care assessment for the new young adult patients joining the practice so the adult health care provider can understand how proficient the young adults are with respect to their self-care skills and knowledge of the adult health care system.”
Disease-specific toolkits coming in 2016
Dr. White is also a consultant to the Pediatric to Adult Health Care Transitions Workgroup, an effort of the ACP Council of Subspecialty Societies to address gaps in care that exist with the transition of pediatric patients into adult health care. According to an ACP document describing the effort, the goal of workgroup is to identify “common tools/templates for a ‘general’ transition process, and subspecialty societies will utilize the templates to develop customized toolkits for specific diseases/conditions. The toolkits will then be collected and broadly disseminated,” with release targeted for May 2016 at the ACP national meeting. Dr. Stacy P. Ardoin, who is leading the rheumatology subspecialty group on behalf of the American College of Rheumatology, said the fact that this effort was initiated by the ACP “is great, because historically most of the interest in transition has bubbled up from the pediatric side. I think that adult providers are realizing more and more that patients who come to them with complex childhood onset conditions are challenging to manage, and that we really need to partner together to make things the best for our patients.”
Dr. Ardoin, a rheumatologist in the department of internal medicine at the Ohio State University Wexner Medical Center and in the department of pediatrics at Nationwide Children’s Hospital, characterized the final product as “not so much a definite protocol that everybody has to follow, but more just providing recommendations to the practitioner saying, ‘these are the things that we recommend and here are some tools to accomplish it.’ There’s such a lack of standardization for anything with transition despite the fact that we have consensus statements that have been put out in the past. Perhaps part of the problem is that they were too vague or not considered individually for each specialty. Hopefully, having each specialty take this on and look at their unique needs in the transition process will make people more willing to participate.”
Preparing patients to become health care self-advocates
Not all physicians are convinced that more recommendations will help clinicians navigate the intricacies of health care transition, however. “If children come to you as a physician at a young age (most of the kids with arthritis) or very ill (most of the children with systemic lupus erythematosus), good care over time represents a significant bonding experience,” said Dr. Thomas J. A. Lehman, chief of the division of pediatric rheumatology at the Hospital for Special Surgery in New York. “The children come to see you as a significant other in their life. If they then are suddenly ‘cast out’ because they reach a certain age it doesn’t go down any better than it would if a significant relative ‘cast them out.’ The doctors like to think they are transitioning the patients. The patients who have bonded feel they are being rejected. I don’t have an answer to this problem. However, none of the past HCT policy statements adequately recognize this aspect.”
For her part, Dr. White, who is also professor of medicine and pediatrics at George Washington University, Washington, said that in some published studies pediatric providers acknowledged that they are reluctant to transition patients to adult health care providers “and can overprotect their young patients as some parents do. This approach can result in the young adult being a poor self-advocate in the adult health care environment and not receiving developmentally appropriate health care. If a pediatric patient and family have not been informed of when they can no longer come to the pediatric rheumatology practice and have not discussed a transition to adult health care plan, they often assume they can stay with their pediatric rheumatologist forever.”
In addition, Dr. White continued, “some pediatric rheumatologists might say, ‘adult providers don’t know how to care for pediatric rheumatology [patients].’ Well, that may due to the adult providers not having received training in specific pediatric onset conditions. Communication between the pediatric and adult providers is key to a good transition experience for everyone. Trying to quickly build a new relationship with a new young adult patient to keep them engaged in their care and who has been used to the same pediatric provider for years is challenging, and the young adult and the adult provider need the pediatric provider’s support and availability to be a consultant to support the adult provider in the areas where they have little disease-specific experience.”
Dr. Jensen makes it a point to talk to his patients about health care transition in early adolescence, helps them set goals, and encourages them to “take more ownership in late adolescence for his or her own health: making their own appointments for follow-up visits, giving time to the patient in the room alone, and asking patients to maybe monitor medication adherence but not to lay the medications out for the patient in the morning,” he said. “When patients are given that opportunity, they may mature.”
Dr. Ronis, who is implementing a health care transition program at Children’s National Health System, thinks about health care transition as “a time where there’s risk for potentially worsening disease and negative outcomes due to perceived fragmentation of care. The more prepared we are and the more we prepare our patients to do this successfully, the better outcomes our patients will have.”
The Got Transition Center for Health Care Transition Improvement is funded by the Health Resources and Services Administration. The physicians interviewed for this story reported having no financial disclosures.
It’s been nearly 13 years since the American Academy of Pediatrics, the American Academy of Family Physicians, and the American College of Physicians released its Consensus Policy Statement on Health Care Transitions for Young Adults With Special Needs and 4 years since the AAP, AAFP, ACP Clinical Report: Supporting Health Care Transition (HCT) from Adolescence to Adulthood in the Medical Home was published, but formal adoption of suggested approaches has remained minimal.
A recent survey of 158 members of the Childhood Arthritis and Rheumatology Research Alliance in North America found that fewer than 10% of respondents were familiar with the consensus statement and the HCT clinical report, only 8% have a formal written transition policy, while 42% use an informal approach (J. Rheumatol. 2014;41:768-79). Respondents ranked fragmented adult health care and lack of time to provide sufficient services as the two major barriers to transition.
“Until recently, there hasn’t been a lot of focus on transition in fellowship training, and I don’t know how well the consensus policy statement and HCT clinical report are publicized in residency or fellowship training,” one of the study authors, Dr. Tova Ronis, said in an interview. “There’s definitely a role for more education about the policy statement and HCT clinical report.” As a result of inconsistent transition practices, some patients fall through the cracks in the transition from pediatric to adult rheumatology care. Transition can be difficult and even patients who receive transition care can have challenges, said Dr. Ronis of the pediatric rheumatology division at Children’s National Health System, Washington. She spoke of one young patient with severe systemic disease who was referred to ongoing adult care with an expert in the field. The patient “went to the adult office but didn’t like it there; she didn’t feel like she was being listened to and didn’t feel like the new doctor knew her well. She would come back to us, go back and forth, so her care became very fragmented. We communicated with the adult doctor but there wasn’t one person making the plan [of care].” In addition, other clinicians who had been providing subspecialty care and had not transitioned her to adult practice “were still at the children’s hospital, so when she experienced health problems or pain, she would come to the pediatric emergency department,” Dr. Ronis recalled. “She hadn’t identified the new institution as her medical home, so her care became very fragmented during that transition process. She actually missed some therapy because she didn’t like the new office, so wasn’t seen anywhere for a while.”
Dr. Paul T. Jensen said that most clinicians “have a gut feeling” that the current health care transition landscape has limitations, despite the availability of such tools as the Got Transition website, the Transition Readiness Assessment Questionnaire (TRAQ), as well as studies of novel approaches such as having a social worker serve as a transition coordinator(Pediatr. Rheumatol. 2015;13:17 [doi:10.1186/s12969-015-0013-0]) or having clinicians from adult practices see patients at a pediatric institution (Pediatr. Pulmonol. 2013;48:658-65). “I think that most, if not all, pediatric providers, including pediatric rheumatologists, worry about their patients as they get older, especially those who have a decreased maturity level,” said Dr. Jensen, who is a fellow in both adult and pediatric rheumatology at the Ohio State University Wexner Medical Center and Nationwide Children’s Hospital, Columbus. “But I think there’s a large lack of knowledge about what resources are available.”
Choosing transition practices that work for you
Dr. Patience White, a pediatric and adult rheumatologist who in 2011 coauthored the HCT Clinical Report, is familiar with such sentiment. She says that the topic of transition has “come to the forefront because the pediatric rheumatology field has grown. On top of that, many of our patients have grown up and moved on to other aspects of adulthood, so now pediatric providers will need to prepare to ‘let them go to adult health care.’ The perfect storm is happening.” Dr. White codirects the Got Transition Center for Health Care Transition Improvement, a federally funded program located at the National Alliance to Advance Adolescent Health. One of the center’s goals is to expand the use of the Six Core Elements of Health Care Transition that were developed to help make the 2011 Health Care Transitions Clinical Report actionable. These include practice quality improvement strategies such as establishing a transition policy, tracking progress, offering transition readiness assessments, creating a transition plan, transferring to an adult provider, and assessing the youth’s and family’s experience with the transition. The original Six Core Elements of Health Care Transition were tested in learning collaboratives across the United States and have been updated with input from the learning collaboratives and national transition experts into three separate packages of tools and evaluation approaches for transitioning youth to adult health care providers, transitioning to an adult approach to health care without changing providers, and integrating young adults into adult health care. For example, recommendations when transitioning youth to adult health care providers include developing a transition policy/statement “with input from youth and families that describes the practice’s approach to transition, including privacy and consent information,” as well as conducting regular transition readiness assessments, beginning at age 14 years, “to identify and discuss with youth and parent/caregiver their needs and goals in self-care.” Other tools available from the Got Transition website include a set of frequently asked transition questions developed by and for youth/young adults, as well as many transition resources.
While she acknowledged it may not be feasible for pediatric and adult rheumatologists to implement all Six Core Elements in their practice, Dr. White suggests providers might try implementing at least one or two and, for pediatric rheumatologists, start the discussion of the transition process when their patients reach the age of 13 years as recommended by the HCT clinical report. “Everybody says it’s hard, and I agree, changing practice processes to improve health care is hard work,” she said. “We know preparation for the youth and family needs to happen. We know transfer and communication has to occur, and that the accepting adult provider has to be informed and comfortable to make the transition process a success. It is about choosing one or two Six Core Element tools that can work for you, your practice, and your patients to help them through the transition process. If you’re an adult provider, you might choose to offer a welcome letter to my practice and a self-care assessment for the new young adult patients joining the practice so the adult health care provider can understand how proficient the young adults are with respect to their self-care skills and knowledge of the adult health care system.”
Disease-specific toolkits coming in 2016
Dr. White is also a consultant to the Pediatric to Adult Health Care Transitions Workgroup, an effort of the ACP Council of Subspecialty Societies to address gaps in care that exist with the transition of pediatric patients into adult health care. According to an ACP document describing the effort, the goal of workgroup is to identify “common tools/templates for a ‘general’ transition process, and subspecialty societies will utilize the templates to develop customized toolkits for specific diseases/conditions. The toolkits will then be collected and broadly disseminated,” with release targeted for May 2016 at the ACP national meeting. Dr. Stacy P. Ardoin, who is leading the rheumatology subspecialty group on behalf of the American College of Rheumatology, said the fact that this effort was initiated by the ACP “is great, because historically most of the interest in transition has bubbled up from the pediatric side. I think that adult providers are realizing more and more that patients who come to them with complex childhood onset conditions are challenging to manage, and that we really need to partner together to make things the best for our patients.”
Dr. Ardoin, a rheumatologist in the department of internal medicine at the Ohio State University Wexner Medical Center and in the department of pediatrics at Nationwide Children’s Hospital, characterized the final product as “not so much a definite protocol that everybody has to follow, but more just providing recommendations to the practitioner saying, ‘these are the things that we recommend and here are some tools to accomplish it.’ There’s such a lack of standardization for anything with transition despite the fact that we have consensus statements that have been put out in the past. Perhaps part of the problem is that they were too vague or not considered individually for each specialty. Hopefully, having each specialty take this on and look at their unique needs in the transition process will make people more willing to participate.”
Preparing patients to become health care self-advocates
Not all physicians are convinced that more recommendations will help clinicians navigate the intricacies of health care transition, however. “If children come to you as a physician at a young age (most of the kids with arthritis) or very ill (most of the children with systemic lupus erythematosus), good care over time represents a significant bonding experience,” said Dr. Thomas J. A. Lehman, chief of the division of pediatric rheumatology at the Hospital for Special Surgery in New York. “The children come to see you as a significant other in their life. If they then are suddenly ‘cast out’ because they reach a certain age it doesn’t go down any better than it would if a significant relative ‘cast them out.’ The doctors like to think they are transitioning the patients. The patients who have bonded feel they are being rejected. I don’t have an answer to this problem. However, none of the past HCT policy statements adequately recognize this aspect.”
For her part, Dr. White, who is also professor of medicine and pediatrics at George Washington University, Washington, said that in some published studies pediatric providers acknowledged that they are reluctant to transition patients to adult health care providers “and can overprotect their young patients as some parents do. This approach can result in the young adult being a poor self-advocate in the adult health care environment and not receiving developmentally appropriate health care. If a pediatric patient and family have not been informed of when they can no longer come to the pediatric rheumatology practice and have not discussed a transition to adult health care plan, they often assume they can stay with their pediatric rheumatologist forever.”
In addition, Dr. White continued, “some pediatric rheumatologists might say, ‘adult providers don’t know how to care for pediatric rheumatology [patients].’ Well, that may due to the adult providers not having received training in specific pediatric onset conditions. Communication between the pediatric and adult providers is key to a good transition experience for everyone. Trying to quickly build a new relationship with a new young adult patient to keep them engaged in their care and who has been used to the same pediatric provider for years is challenging, and the young adult and the adult provider need the pediatric provider’s support and availability to be a consultant to support the adult provider in the areas where they have little disease-specific experience.”
Dr. Jensen makes it a point to talk to his patients about health care transition in early adolescence, helps them set goals, and encourages them to “take more ownership in late adolescence for his or her own health: making their own appointments for follow-up visits, giving time to the patient in the room alone, and asking patients to maybe monitor medication adherence but not to lay the medications out for the patient in the morning,” he said. “When patients are given that opportunity, they may mature.”
Dr. Ronis, who is implementing a health care transition program at Children’s National Health System, thinks about health care transition as “a time where there’s risk for potentially worsening disease and negative outcomes due to perceived fragmentation of care. The more prepared we are and the more we prepare our patients to do this successfully, the better outcomes our patients will have.”
The Got Transition Center for Health Care Transition Improvement is funded by the Health Resources and Services Administration. The physicians interviewed for this story reported having no financial disclosures.
Mindfulness intervention helps veterans with PTSD
Veterans with PTSD who participated in a mindfulness-based stress reduction program experienced greater decrease in symptom severity, compared with those who participated in present-centered group therapy, according to a randomized, controlled trial. However, the magnitude of the average improvement was considered modest.
“The quality of scientific evidence supporting the efficacy of mindfulness-based interventions has recently been criticized,” researchers led by Melissa A. Polusny, Ph.D., reported on Aug. 4. “This study improves on shortcomings of previous trials by comparing mindfulness-based stress reduction with an active, credible control condition, taking steps to ensure treatment fidelity, and using both patient-reported and blinded clinician ratings of PTSD outcomes.”
From March 2012 to December 2013, Dr. Polusny of the Minneapolis VA Medical Center and her associates randomly assigned 116 veterans with PTSD to one of two treatment groups: 58 to mindfulness-based stress reduction therapy (MBSR) and 58 to patient-centered group therapy (PCT), each delivered in nine weekly group sessions (JAMA. 2015 Aug 4;314(5):456-65.). The MBSR sessions focused on teaching patients to attend to their thoughts, emotions, and sensations with an attitude of nonjudgment, kindness, and curiosity. The PCT sessions focused on teaching patients to tap into their existing skills and strengths to cope effectively with current stressors that might be exacerbated by PTSD symptoms.
The primary outcome was change in PTSD severity over time as measured by the PTSD Checklist (PCL) at weeks 3, 6, 9, and 17 (the 2-month follow-up). Possible PCL scores ranged from 17 to 85, with higher scores indicating more severe PTSD symptoms. Secondary outcomes included diagnosis and symptom severity of PTSD based on the Clinician-Administered PTSD Scale (CAPS) at baseline, week 9, and week 17, as well as improvements in depressive symptoms, quality of life, and mindfulness.
The mean age of study participants was 59 years, and 84% were white. Between baseline and week 9, PCL scores among patients in the MBSR group improved from 63.6 to 55.7, while scores among patients in the PCT group improved from 58.8 to 55.8, which translated into a between-group difference of 4.95 (P = .002). Between baseline and week 17, PCL scores among patients in the MBSR group improved from 63.6 to 54.4, while scores among patients in the PCT group improved from 58.8 to 56, which translated into a between-group difference of 6.44 (P less than .001).
As for secondary outcomes, patients in the MBSR group were more likely to demonstrate significant improvement in self-reported PTSD symptom severity at week 17, compared with their counterparts in the PCT group (48.9% vs. 28.1%, respectively, for a between-group difference of 20.9%; P = .03). However, they were no more likely to have loss of PTSD diagnosis (53.3% vs. 47.3%, respectively, for a between-group difference of 6%; P = .55).
Using a 10-point or greater reduction on the CAPS as a benchmark, the researchers found that both groups were similar in the percentage of participants showing clinically significant improvement in interview-rated PTSD symptom severity at 2-month follow-up (66.7% among patients in the MBSR group vs. 54.5% in the PCT group, for a between-group difference of 12.1%; P = .22). In addition, similar percentages of participants reported clinically significant improvement in depressive symptoms on the PHQ-9 (27.7% among patients in the MBSR group, vs. 22.8% in the PCT group, for a between-group difference of 4.9%; P = .57).
“Findings from the present study suggest that veterans who received mindfulness-based stress reduction therapy reported significant improvement in mindfulness skills after treatment, while there appeared to be little change in mindfulness skills reported by veterans who received present-centered group therapy,” the researchers wrote. “Moreover, findings suggest that greater reductions in PTSD symptom severity were associated with changes in mindfulness over the course of treatment. Improvements in quality of life made during treatment appeared to be maintained through the 2-month follow-up for participants receiving mindfulness-based stress reduction therapy, but reports of quality of life appeared to return to baseline levels for present-centered group therapy participants during this same follow-up period. Taken together, these findings suggest that mindfulness-based stress reduction may provide veterans with internal tools for promoting self-management of PTSD symptoms and quality of life.”
They acknowledged certain limitations of the study, including the fact that even though groups were structurally equivalent in number of weekly sessions, “therapist training and qualifications, and group format, present-centered group therapy may not have fully accounted for all nonspecific factors present in mindfulness-based stress reduction (e.g., therapist expectations) and was unequal in duration of sessions.”
The study was supported by the Minneapolis VA Health Care System and a grant from the Department of Veterans Affairs. The researchers reported having no financial disclosures.
Military personnel are at particularly high risk of PTSD because deployments to combat zones increase the risk of exposure to trauma. The effects of PTSD on military and civilian patients, their families, and society in general can be profound. Posttraumatic stress disorder is associated with increased risks of suicide, depression, substance use disorders, intimate partner violence, unemployment, and persistently low quality of life. In addition, trauma and PTSD are associated with a higher risk of other health problems, including coronary artery disease, arthritis, asthma, gastrointestinal symptoms, and all-cause mortality. There are also spiritual and moral dimensions to experiencing or committing acts of trauma, which can endure across the life span.
Although the results reported by Dr. Polusny and her associates are promising, the short duration of follow-up calls into question whether the effects of MBSR persist over time; thus, additional studies of MBSR and other mindfulness-based interventions for PTSD are warranted.
Group interventions such as PCT and MBSR could expand the availability of therapies for PTSD. Given the large number of individuals with PTSD, not all of whom will opt for or benefit sufficiently from existing approaches, additional treatments suitable for broad implementation are needed. If additional studies confirm that MBSR is efficacious for PTSD, it may represent a cost-effective approach to care. Mindfulness-based stress reduction can be led by facilitators who are not psychotherapists, which could expand the availability of PTSD practitioners and services. Other non–trauma focused approaches to PTSD include forms of meditation for which there is initial support among military personnel.
Another potential approach for improving patient outcomes is to involve family members in treatment, as recommended in a recent report of PTSD therapy by the Institute of Medicine (“Treatment for Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment,” Washington: The National Academies Press, 2014). For mindfulness-based and other skills-based approaches, inclusion of family members in groups or homework practices might help support the patients consistently practice the new skills in their everyday lives through encouragement and reminders that are available more frequently and organically than would be the case through weekly sessions with the instructor. Additionally, if family members are also learning more effective ways of coping with stress and the family is working together to support the patient, these efforts might lead to additional gains over time or shifts in family relational dynamics in support of a more robust recovery.
Dr. David J. Kearney and Tracy L. Simpson, Ph.D., are with the VA Puget Sound Health Care System, Seattle. Neither author reported having financial conflicts. Their remarks were condensed from an accompanying editorial (JAMA. 2015 Aug 4;314(5):453-5.).
Military personnel are at particularly high risk of PTSD because deployments to combat zones increase the risk of exposure to trauma. The effects of PTSD on military and civilian patients, their families, and society in general can be profound. Posttraumatic stress disorder is associated with increased risks of suicide, depression, substance use disorders, intimate partner violence, unemployment, and persistently low quality of life. In addition, trauma and PTSD are associated with a higher risk of other health problems, including coronary artery disease, arthritis, asthma, gastrointestinal symptoms, and all-cause mortality. There are also spiritual and moral dimensions to experiencing or committing acts of trauma, which can endure across the life span.
Although the results reported by Dr. Polusny and her associates are promising, the short duration of follow-up calls into question whether the effects of MBSR persist over time; thus, additional studies of MBSR and other mindfulness-based interventions for PTSD are warranted.
Group interventions such as PCT and MBSR could expand the availability of therapies for PTSD. Given the large number of individuals with PTSD, not all of whom will opt for or benefit sufficiently from existing approaches, additional treatments suitable for broad implementation are needed. If additional studies confirm that MBSR is efficacious for PTSD, it may represent a cost-effective approach to care. Mindfulness-based stress reduction can be led by facilitators who are not psychotherapists, which could expand the availability of PTSD practitioners and services. Other non–trauma focused approaches to PTSD include forms of meditation for which there is initial support among military personnel.
Another potential approach for improving patient outcomes is to involve family members in treatment, as recommended in a recent report of PTSD therapy by the Institute of Medicine (“Treatment for Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment,” Washington: The National Academies Press, 2014). For mindfulness-based and other skills-based approaches, inclusion of family members in groups or homework practices might help support the patients consistently practice the new skills in their everyday lives through encouragement and reminders that are available more frequently and organically than would be the case through weekly sessions with the instructor. Additionally, if family members are also learning more effective ways of coping with stress and the family is working together to support the patient, these efforts might lead to additional gains over time or shifts in family relational dynamics in support of a more robust recovery.
Dr. David J. Kearney and Tracy L. Simpson, Ph.D., are with the VA Puget Sound Health Care System, Seattle. Neither author reported having financial conflicts. Their remarks were condensed from an accompanying editorial (JAMA. 2015 Aug 4;314(5):453-5.).
Military personnel are at particularly high risk of PTSD because deployments to combat zones increase the risk of exposure to trauma. The effects of PTSD on military and civilian patients, their families, and society in general can be profound. Posttraumatic stress disorder is associated with increased risks of suicide, depression, substance use disorders, intimate partner violence, unemployment, and persistently low quality of life. In addition, trauma and PTSD are associated with a higher risk of other health problems, including coronary artery disease, arthritis, asthma, gastrointestinal symptoms, and all-cause mortality. There are also spiritual and moral dimensions to experiencing or committing acts of trauma, which can endure across the life span.
Although the results reported by Dr. Polusny and her associates are promising, the short duration of follow-up calls into question whether the effects of MBSR persist over time; thus, additional studies of MBSR and other mindfulness-based interventions for PTSD are warranted.
Group interventions such as PCT and MBSR could expand the availability of therapies for PTSD. Given the large number of individuals with PTSD, not all of whom will opt for or benefit sufficiently from existing approaches, additional treatments suitable for broad implementation are needed. If additional studies confirm that MBSR is efficacious for PTSD, it may represent a cost-effective approach to care. Mindfulness-based stress reduction can be led by facilitators who are not psychotherapists, which could expand the availability of PTSD practitioners and services. Other non–trauma focused approaches to PTSD include forms of meditation for which there is initial support among military personnel.
Another potential approach for improving patient outcomes is to involve family members in treatment, as recommended in a recent report of PTSD therapy by the Institute of Medicine (“Treatment for Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment,” Washington: The National Academies Press, 2014). For mindfulness-based and other skills-based approaches, inclusion of family members in groups or homework practices might help support the patients consistently practice the new skills in their everyday lives through encouragement and reminders that are available more frequently and organically than would be the case through weekly sessions with the instructor. Additionally, if family members are also learning more effective ways of coping with stress and the family is working together to support the patient, these efforts might lead to additional gains over time or shifts in family relational dynamics in support of a more robust recovery.
Dr. David J. Kearney and Tracy L. Simpson, Ph.D., are with the VA Puget Sound Health Care System, Seattle. Neither author reported having financial conflicts. Their remarks were condensed from an accompanying editorial (JAMA. 2015 Aug 4;314(5):453-5.).
Veterans with PTSD who participated in a mindfulness-based stress reduction program experienced greater decrease in symptom severity, compared with those who participated in present-centered group therapy, according to a randomized, controlled trial. However, the magnitude of the average improvement was considered modest.
“The quality of scientific evidence supporting the efficacy of mindfulness-based interventions has recently been criticized,” researchers led by Melissa A. Polusny, Ph.D., reported on Aug. 4. “This study improves on shortcomings of previous trials by comparing mindfulness-based stress reduction with an active, credible control condition, taking steps to ensure treatment fidelity, and using both patient-reported and blinded clinician ratings of PTSD outcomes.”
From March 2012 to December 2013, Dr. Polusny of the Minneapolis VA Medical Center and her associates randomly assigned 116 veterans with PTSD to one of two treatment groups: 58 to mindfulness-based stress reduction therapy (MBSR) and 58 to patient-centered group therapy (PCT), each delivered in nine weekly group sessions (JAMA. 2015 Aug 4;314(5):456-65.). The MBSR sessions focused on teaching patients to attend to their thoughts, emotions, and sensations with an attitude of nonjudgment, kindness, and curiosity. The PCT sessions focused on teaching patients to tap into their existing skills and strengths to cope effectively with current stressors that might be exacerbated by PTSD symptoms.
The primary outcome was change in PTSD severity over time as measured by the PTSD Checklist (PCL) at weeks 3, 6, 9, and 17 (the 2-month follow-up). Possible PCL scores ranged from 17 to 85, with higher scores indicating more severe PTSD symptoms. Secondary outcomes included diagnosis and symptom severity of PTSD based on the Clinician-Administered PTSD Scale (CAPS) at baseline, week 9, and week 17, as well as improvements in depressive symptoms, quality of life, and mindfulness.
The mean age of study participants was 59 years, and 84% were white. Between baseline and week 9, PCL scores among patients in the MBSR group improved from 63.6 to 55.7, while scores among patients in the PCT group improved from 58.8 to 55.8, which translated into a between-group difference of 4.95 (P = .002). Between baseline and week 17, PCL scores among patients in the MBSR group improved from 63.6 to 54.4, while scores among patients in the PCT group improved from 58.8 to 56, which translated into a between-group difference of 6.44 (P less than .001).
As for secondary outcomes, patients in the MBSR group were more likely to demonstrate significant improvement in self-reported PTSD symptom severity at week 17, compared with their counterparts in the PCT group (48.9% vs. 28.1%, respectively, for a between-group difference of 20.9%; P = .03). However, they were no more likely to have loss of PTSD diagnosis (53.3% vs. 47.3%, respectively, for a between-group difference of 6%; P = .55).
Using a 10-point or greater reduction on the CAPS as a benchmark, the researchers found that both groups were similar in the percentage of participants showing clinically significant improvement in interview-rated PTSD symptom severity at 2-month follow-up (66.7% among patients in the MBSR group vs. 54.5% in the PCT group, for a between-group difference of 12.1%; P = .22). In addition, similar percentages of participants reported clinically significant improvement in depressive symptoms on the PHQ-9 (27.7% among patients in the MBSR group, vs. 22.8% in the PCT group, for a between-group difference of 4.9%; P = .57).
“Findings from the present study suggest that veterans who received mindfulness-based stress reduction therapy reported significant improvement in mindfulness skills after treatment, while there appeared to be little change in mindfulness skills reported by veterans who received present-centered group therapy,” the researchers wrote. “Moreover, findings suggest that greater reductions in PTSD symptom severity were associated with changes in mindfulness over the course of treatment. Improvements in quality of life made during treatment appeared to be maintained through the 2-month follow-up for participants receiving mindfulness-based stress reduction therapy, but reports of quality of life appeared to return to baseline levels for present-centered group therapy participants during this same follow-up period. Taken together, these findings suggest that mindfulness-based stress reduction may provide veterans with internal tools for promoting self-management of PTSD symptoms and quality of life.”
They acknowledged certain limitations of the study, including the fact that even though groups were structurally equivalent in number of weekly sessions, “therapist training and qualifications, and group format, present-centered group therapy may not have fully accounted for all nonspecific factors present in mindfulness-based stress reduction (e.g., therapist expectations) and was unequal in duration of sessions.”
The study was supported by the Minneapolis VA Health Care System and a grant from the Department of Veterans Affairs. The researchers reported having no financial disclosures.
Veterans with PTSD who participated in a mindfulness-based stress reduction program experienced greater decrease in symptom severity, compared with those who participated in present-centered group therapy, according to a randomized, controlled trial. However, the magnitude of the average improvement was considered modest.
“The quality of scientific evidence supporting the efficacy of mindfulness-based interventions has recently been criticized,” researchers led by Melissa A. Polusny, Ph.D., reported on Aug. 4. “This study improves on shortcomings of previous trials by comparing mindfulness-based stress reduction with an active, credible control condition, taking steps to ensure treatment fidelity, and using both patient-reported and blinded clinician ratings of PTSD outcomes.”
From March 2012 to December 2013, Dr. Polusny of the Minneapolis VA Medical Center and her associates randomly assigned 116 veterans with PTSD to one of two treatment groups: 58 to mindfulness-based stress reduction therapy (MBSR) and 58 to patient-centered group therapy (PCT), each delivered in nine weekly group sessions (JAMA. 2015 Aug 4;314(5):456-65.). The MBSR sessions focused on teaching patients to attend to their thoughts, emotions, and sensations with an attitude of nonjudgment, kindness, and curiosity. The PCT sessions focused on teaching patients to tap into their existing skills and strengths to cope effectively with current stressors that might be exacerbated by PTSD symptoms.
The primary outcome was change in PTSD severity over time as measured by the PTSD Checklist (PCL) at weeks 3, 6, 9, and 17 (the 2-month follow-up). Possible PCL scores ranged from 17 to 85, with higher scores indicating more severe PTSD symptoms. Secondary outcomes included diagnosis and symptom severity of PTSD based on the Clinician-Administered PTSD Scale (CAPS) at baseline, week 9, and week 17, as well as improvements in depressive symptoms, quality of life, and mindfulness.
The mean age of study participants was 59 years, and 84% were white. Between baseline and week 9, PCL scores among patients in the MBSR group improved from 63.6 to 55.7, while scores among patients in the PCT group improved from 58.8 to 55.8, which translated into a between-group difference of 4.95 (P = .002). Between baseline and week 17, PCL scores among patients in the MBSR group improved from 63.6 to 54.4, while scores among patients in the PCT group improved from 58.8 to 56, which translated into a between-group difference of 6.44 (P less than .001).
As for secondary outcomes, patients in the MBSR group were more likely to demonstrate significant improvement in self-reported PTSD symptom severity at week 17, compared with their counterparts in the PCT group (48.9% vs. 28.1%, respectively, for a between-group difference of 20.9%; P = .03). However, they were no more likely to have loss of PTSD diagnosis (53.3% vs. 47.3%, respectively, for a between-group difference of 6%; P = .55).
Using a 10-point or greater reduction on the CAPS as a benchmark, the researchers found that both groups were similar in the percentage of participants showing clinically significant improvement in interview-rated PTSD symptom severity at 2-month follow-up (66.7% among patients in the MBSR group vs. 54.5% in the PCT group, for a between-group difference of 12.1%; P = .22). In addition, similar percentages of participants reported clinically significant improvement in depressive symptoms on the PHQ-9 (27.7% among patients in the MBSR group, vs. 22.8% in the PCT group, for a between-group difference of 4.9%; P = .57).
“Findings from the present study suggest that veterans who received mindfulness-based stress reduction therapy reported significant improvement in mindfulness skills after treatment, while there appeared to be little change in mindfulness skills reported by veterans who received present-centered group therapy,” the researchers wrote. “Moreover, findings suggest that greater reductions in PTSD symptom severity were associated with changes in mindfulness over the course of treatment. Improvements in quality of life made during treatment appeared to be maintained through the 2-month follow-up for participants receiving mindfulness-based stress reduction therapy, but reports of quality of life appeared to return to baseline levels for present-centered group therapy participants during this same follow-up period. Taken together, these findings suggest that mindfulness-based stress reduction may provide veterans with internal tools for promoting self-management of PTSD symptoms and quality of life.”
They acknowledged certain limitations of the study, including the fact that even though groups were structurally equivalent in number of weekly sessions, “therapist training and qualifications, and group format, present-centered group therapy may not have fully accounted for all nonspecific factors present in mindfulness-based stress reduction (e.g., therapist expectations) and was unequal in duration of sessions.”
The study was supported by the Minneapolis VA Health Care System and a grant from the Department of Veterans Affairs. The researchers reported having no financial disclosures.
FROM JAMA
Key clinical point: Mindfulness-based stress reduction helped lessen PTSD severity in veterans.
Major finding: Between baseline and week 17, PTSD Checklist scores among patients in the MBSR group improved from 63.6 to 54.4, while scores among patients in the PCT group improved from 58.8 to 56.0, which translated into a between-group difference of 6.44 (P less than .001).
Data source: A randomized, controlled trial in which 116 veterans with PTSD were assigned to one of two treatment groups: 58 to mindfulness-based stress reduction therapy and 58 to patient-centered group therapy, each delivered in nine weekly group sessions.
Disclosures: The study was supported by the Minneapolis VA Health Care System and a grant from the Department of Veterans Affairs. The researchers reported having no financial disclosures.
Targeting H. pylori may help prevent stomach cancer
Eradicating Helicobacter pylori bacteria with a short course of antibiotics and proton pump inhibitors helped reduce the risk of gastric cancer in otherwise healthy and asymptomatic H. pylori–positive adults by 34%, according to a review published online July 22.
However, because all but one of the six studies included in the analysis were conducted in Asian populations, it is not known whether the results would be the same in Western populations.
“Gastric cancer is the third most common cause of cancer death worldwide,” wrote the researchers, who are members of the Cochrane Upper Gastrointestinal and Pancreatic Diseases Group. “Individuals infected with H. pylori have a higher likelihood of developing gastric cancer than individuals who are not infected. Eradication of H. pylori in healthy asymptomatic individuals in the general population may reduce the incidence of gastric cancer, but the magnitude of this effect is unclear.”
For the review, the researchers searched the Cochrane Central Register of Controlled Trials and other major databases for randomized, controlled trials that compared at least 1 week of H. pylori therapy with placebo or no treatment in preventing subsequent development of gastric cancer in otherwise healthy and asymptomatic H. pylori–positive adults (Cochrane Database Syst. Rev. 2015 [doi:10.1002/14651858.CD005583.pub2]. To be included, trials had to follow up participants for at least 2 years and needed to have at least two participants with gastric cancer as an outcome. The researchers defined gastric cancer as any gastric adenocarcinoma, including intestinal (differentiated) or diffuse (undifferentiated) type, with or without specified histology.
In all, six studies with a total of 6,497 participants met eligibility criteria. The studies primarily used a combination of antibiotics and proton pump inhibitors. Five of the six studies were conducted in Asian populations. Of 3,294 participants assigned to eradication therapy, 51 (1.6%) subsequently developed gastric cancer, compared with 76 (2.4%) of 3,203 participants who received placebo or no treatment. “There was no statistically significant heterogeneity between individual trial results,” the researchers wrote. “Therefore there was a small, but statistically significant benefit of H. pylori eradication therapy in preventing gastric cancer in healthy asymptomatic infected individuals (risk ratio, 0.66).”
They pointed out that the effect of H. pylori eradication on preventing death from gastric cancer, compared with placebo or no treatment “was uncertain due to wide confidence intervals” and there was “no evidence of an effect on all-cause mortality. Adverse events data were poorly reported.”
The researchers acknowledged certain limitations of the review, including that fact that only three of the trials were considered to be at low risk of bias. In addition, the design of some trials made it “difficult to ascertain whether the significant reduction in the risk of subsequent gastric cancer was due to H. pylori eradication therapy alone, or to the antioxidants or vitamins that were coadministered in some of the trials.”
In terms of implication for clinical practice, “the findings of this systematic review and meta-analysis add to the increasing evidence that eradicating H. pylori in the general population has the potential to prevent gastric cancer,” they concluded. “International guidelines for the management of H. pylori infection may change as a result.”
The researchers reported having no relevant financial conflicts.
Eradicating Helicobacter pylori bacteria with a short course of antibiotics and proton pump inhibitors helped reduce the risk of gastric cancer in otherwise healthy and asymptomatic H. pylori–positive adults by 34%, according to a review published online July 22.
However, because all but one of the six studies included in the analysis were conducted in Asian populations, it is not known whether the results would be the same in Western populations.
“Gastric cancer is the third most common cause of cancer death worldwide,” wrote the researchers, who are members of the Cochrane Upper Gastrointestinal and Pancreatic Diseases Group. “Individuals infected with H. pylori have a higher likelihood of developing gastric cancer than individuals who are not infected. Eradication of H. pylori in healthy asymptomatic individuals in the general population may reduce the incidence of gastric cancer, but the magnitude of this effect is unclear.”
For the review, the researchers searched the Cochrane Central Register of Controlled Trials and other major databases for randomized, controlled trials that compared at least 1 week of H. pylori therapy with placebo or no treatment in preventing subsequent development of gastric cancer in otherwise healthy and asymptomatic H. pylori–positive adults (Cochrane Database Syst. Rev. 2015 [doi:10.1002/14651858.CD005583.pub2]. To be included, trials had to follow up participants for at least 2 years and needed to have at least two participants with gastric cancer as an outcome. The researchers defined gastric cancer as any gastric adenocarcinoma, including intestinal (differentiated) or diffuse (undifferentiated) type, with or without specified histology.
In all, six studies with a total of 6,497 participants met eligibility criteria. The studies primarily used a combination of antibiotics and proton pump inhibitors. Five of the six studies were conducted in Asian populations. Of 3,294 participants assigned to eradication therapy, 51 (1.6%) subsequently developed gastric cancer, compared with 76 (2.4%) of 3,203 participants who received placebo or no treatment. “There was no statistically significant heterogeneity between individual trial results,” the researchers wrote. “Therefore there was a small, but statistically significant benefit of H. pylori eradication therapy in preventing gastric cancer in healthy asymptomatic infected individuals (risk ratio, 0.66).”
They pointed out that the effect of H. pylori eradication on preventing death from gastric cancer, compared with placebo or no treatment “was uncertain due to wide confidence intervals” and there was “no evidence of an effect on all-cause mortality. Adverse events data were poorly reported.”
The researchers acknowledged certain limitations of the review, including that fact that only three of the trials were considered to be at low risk of bias. In addition, the design of some trials made it “difficult to ascertain whether the significant reduction in the risk of subsequent gastric cancer was due to H. pylori eradication therapy alone, or to the antioxidants or vitamins that were coadministered in some of the trials.”
In terms of implication for clinical practice, “the findings of this systematic review and meta-analysis add to the increasing evidence that eradicating H. pylori in the general population has the potential to prevent gastric cancer,” they concluded. “International guidelines for the management of H. pylori infection may change as a result.”
The researchers reported having no relevant financial conflicts.
Eradicating Helicobacter pylori bacteria with a short course of antibiotics and proton pump inhibitors helped reduce the risk of gastric cancer in otherwise healthy and asymptomatic H. pylori–positive adults by 34%, according to a review published online July 22.
However, because all but one of the six studies included in the analysis were conducted in Asian populations, it is not known whether the results would be the same in Western populations.
“Gastric cancer is the third most common cause of cancer death worldwide,” wrote the researchers, who are members of the Cochrane Upper Gastrointestinal and Pancreatic Diseases Group. “Individuals infected with H. pylori have a higher likelihood of developing gastric cancer than individuals who are not infected. Eradication of H. pylori in healthy asymptomatic individuals in the general population may reduce the incidence of gastric cancer, but the magnitude of this effect is unclear.”
For the review, the researchers searched the Cochrane Central Register of Controlled Trials and other major databases for randomized, controlled trials that compared at least 1 week of H. pylori therapy with placebo or no treatment in preventing subsequent development of gastric cancer in otherwise healthy and asymptomatic H. pylori–positive adults (Cochrane Database Syst. Rev. 2015 [doi:10.1002/14651858.CD005583.pub2]. To be included, trials had to follow up participants for at least 2 years and needed to have at least two participants with gastric cancer as an outcome. The researchers defined gastric cancer as any gastric adenocarcinoma, including intestinal (differentiated) or diffuse (undifferentiated) type, with or without specified histology.
In all, six studies with a total of 6,497 participants met eligibility criteria. The studies primarily used a combination of antibiotics and proton pump inhibitors. Five of the six studies were conducted in Asian populations. Of 3,294 participants assigned to eradication therapy, 51 (1.6%) subsequently developed gastric cancer, compared with 76 (2.4%) of 3,203 participants who received placebo or no treatment. “There was no statistically significant heterogeneity between individual trial results,” the researchers wrote. “Therefore there was a small, but statistically significant benefit of H. pylori eradication therapy in preventing gastric cancer in healthy asymptomatic infected individuals (risk ratio, 0.66).”
They pointed out that the effect of H. pylori eradication on preventing death from gastric cancer, compared with placebo or no treatment “was uncertain due to wide confidence intervals” and there was “no evidence of an effect on all-cause mortality. Adverse events data were poorly reported.”
The researchers acknowledged certain limitations of the review, including that fact that only three of the trials were considered to be at low risk of bias. In addition, the design of some trials made it “difficult to ascertain whether the significant reduction in the risk of subsequent gastric cancer was due to H. pylori eradication therapy alone, or to the antioxidants or vitamins that were coadministered in some of the trials.”
In terms of implication for clinical practice, “the findings of this systematic review and meta-analysis add to the increasing evidence that eradicating H. pylori in the general population has the potential to prevent gastric cancer,” they concluded. “International guidelines for the management of H. pylori infection may change as a result.”
The researchers reported having no relevant financial conflicts.
FROM COCHRANE DATABASE OF SYSTEMATIC REVIEWS
Key clinical point: Treating H. pylori may help prevent stomach cancer.
Major finding: Of 3,294 participants assigned to H. pylori eradication therapy, 51 (1.6%) subsequently developed gastric cancer, compared with 76 (2.4%) of 3,203 participants who received placebo or no treatment.
Data source: A review of six randomized, controlled trials that compared at least 1 week of H. pylori therapy with placebo or no treatment in preventing subsequent development of gastric cancer in 6,497 otherwise healthy and asymptomatic H. pylori–positive adults.
Disclosures: The researchers reported having no relevant financial conflicts.
Bills Emerge to Increase Birth Control Access
There’s been a flurry of legislative activity on Capitol Hill and at state houses around the country aimed at improving access to contraception. But there are still deep divides on the best way to achieve that access.
At the federal level, there are two competing bills that address access to contraceptives.
The first – the Allowing Greater Access to Safe and Effective Contraceptive Act (S. 1438) – introduced in May by Sen. Cory Gardner (R-Colo.) and Sen. Kelly Ayotte (R-N.H.), would incentivize manufacturers of “routine-use contraceptives” to file an application with the Food and Drug Administration to switch their products from prescription to over the counter (OTC). The bill would allow for priority review for these products and waive the usual FDA filing fee.
The legislation would also repeal the Affordable Care Act’s restriction on the use of health, medical, and flexible savings accounts to purchase OTC drugs without a prescription.
Despite the fact that the bill is aimed at encouraging the switch of contraceptives to OTC products, it’s been criticized by some reproductive health advocates for not adequately addressing the potential for increased costs if women purchase OTC contraceptives not covered by insurance.
Senate Democrats responded with their own birth control legislation. The Affordability Is Access Act (S. 1532), introduced in June by Sen. Patty Murray (D-Wash.), aims to build on the no-cost coverage for contraception in the Affordable Care Act by ensuring that if the FDA approves oral contraceptives for OTC use, they will be covered without cost sharing even though they are not prescription products.
Affordability at issue
The American Congress of Obstetricians and Gynecologists is backing Sen. Murray’s bill.
In an interview, Dr. Mark S. DeFrancesco, ACOG president, said he is pleased to see proposals from both sides of the aisle emerge on this topic because ACOG has long supported the concept of OTC sale of oral contraceptives. But the GOP-backed proposal does not address the issue of copays and insurance coverage, said Dr. DeFrancesco, who is a managing partner at Westwood Women’s Health in Waterbury, Conn.
“An unintended consequence could be, ironically, that it may decrease access for women because if contraceptives are reclassified as nonformulary drugs by going over the counter, can insurance companies say they’re not going to cover them as they don’t cover aspirin, for instance?” Dr. DeFrancesco said. “That’s our concern: that well-intended legislation could have a backfire effect.”
Missed opportunity for screening?
Not everyone favors moving contraceptives over the counter. Dr. W. David Hager, who practices at Baptist Health Medical Group Women’s Care in Lexington, Ky., expressed concern that providing OTC access to contraceptives would hinder the physician-patient relationship and impact screening efforts.
“One of the big advantages for me as a practicing ob.gyn. is to have face-to-face conversations, to be able to discuss risks and benefits, and to counsel young patients about the choices that they are making, to make sure they are making educated, informed choices,” Dr. Hager said.
A shift to OTC access for contraceptives could potentially decrease the uptake of human papillomavirus (HPV) vaccination, said Dr. Hager, who in the early 2000s served on the FDA’s Advisory Committee for Reproductive Health Drugs.
“We’re already at less than 50% implementation with Gardasil, and this may further decrease that,” he said. “It may delay Pap test screening and HPV screening if women choose after age 21 to just purchase over-the-counter contraceptives. It may delay well-woman screening, where women come in just for a wellness exam. And, in my opinion, it eliminates the potential for discussion of any contraindications to birth control pills. Granted, those are not many, but there are some patients who are not ideal candidates for oral contraceptives. I think it potentially takes a cohort of the highest-risk women and removes them from that potential screening and counseling session.”
Dr. DeFrancesco said he knows from experience that patients will come to see their ob.gyn. regularly just because it’s the right thing to do.
“There could be a small percentage of people who say ‘now I really don’t have to see the doctor, because that’s the only reason I was going,’ ” he said. “The good news about more people being covered with insurance is that at least people are more empowered to go to the physician’s office or the provider’s office to get care.”
States take the lead
While the federal bills are drawing attention, they are no closer to becoming law. But it’s a different story at the state level.
In Washington, the D.C. Council approved a bill that requires health plans to authorize the dispensing of up to 12 months of a woman’s prescription for contraception at one time. The legislation was signed by D.C. Mayor Muriel Bowser (D) in June but won’t apply to health plans until Jan. 1, 2017.
In Oregon, lawmakers enacted similar legislation (House Bill 3343) that requires health insurers to pay for a 12-month supply of the birth control pill, patch, or the ring at one time. The new law goes into effect on Jan. 1, 2016. The Oregon Medical Association, which supported the bill, said that the ability for women to get a 12-month supply of birth control at one time would ease administrative burdens for medical office staff, patients, and pharmacists by lessening refill requests.
In July, Oregon Gov. Kate Brown (D) also approved a related law House Bill 2879, which permits pharmacists in Oregon to prescribe hormonal contraceptive patches and self-administered oral contraceptives. The State Board of Pharmacy will develop rules for how the new system will work, but the law specifies that pharmacists must complete a training program for prescribing hormonal contraceptives, and it requires patients to use a self-screening risk assessment tool before they can receive a prescription. For minors, the law requires evidence of a previous prescription for hormonal contraceptives. The law goes into effect on Jan. 1, 2016.
In California, expanded pharmacist scope of practice legislation known as SB 493 was signed into law by Gov. Jerry Brown (D) on Oct. 1, 2013, and it became effective on Jan. 1, 2014. One of the law’s provisions enables pharmacists to furnish self-administered contraceptives according to standards developed by the California State Board of Pharmacy and the Medical Board of California.
However, this provision is still in the administrative rule-making process, said Sarah McBane, Pharm.D., president of the California Pharmacists Association. According to the original legislation, patients will be required to use a self-screening tool to identify risk factors.
There’s been a flurry of legislative activity on Capitol Hill and at state houses around the country aimed at improving access to contraception. But there are still deep divides on the best way to achieve that access.
At the federal level, there are two competing bills that address access to contraceptives.
The first – the Allowing Greater Access to Safe and Effective Contraceptive Act (S. 1438) – introduced in May by Sen. Cory Gardner (R-Colo.) and Sen. Kelly Ayotte (R-N.H.), would incentivize manufacturers of “routine-use contraceptives” to file an application with the Food and Drug Administration to switch their products from prescription to over the counter (OTC). The bill would allow for priority review for these products and waive the usual FDA filing fee.
The legislation would also repeal the Affordable Care Act’s restriction on the use of health, medical, and flexible savings accounts to purchase OTC drugs without a prescription.
Despite the fact that the bill is aimed at encouraging the switch of contraceptives to OTC products, it’s been criticized by some reproductive health advocates for not adequately addressing the potential for increased costs if women purchase OTC contraceptives not covered by insurance.
Senate Democrats responded with their own birth control legislation. The Affordability Is Access Act (S. 1532), introduced in June by Sen. Patty Murray (D-Wash.), aims to build on the no-cost coverage for contraception in the Affordable Care Act by ensuring that if the FDA approves oral contraceptives for OTC use, they will be covered without cost sharing even though they are not prescription products.
Affordability at issue
The American Congress of Obstetricians and Gynecologists is backing Sen. Murray’s bill.
In an interview, Dr. Mark S. DeFrancesco, ACOG president, said he is pleased to see proposals from both sides of the aisle emerge on this topic because ACOG has long supported the concept of OTC sale of oral contraceptives. But the GOP-backed proposal does not address the issue of copays and insurance coverage, said Dr. DeFrancesco, who is a managing partner at Westwood Women’s Health in Waterbury, Conn.
“An unintended consequence could be, ironically, that it may decrease access for women because if contraceptives are reclassified as nonformulary drugs by going over the counter, can insurance companies say they’re not going to cover them as they don’t cover aspirin, for instance?” Dr. DeFrancesco said. “That’s our concern: that well-intended legislation could have a backfire effect.”
Missed opportunity for screening?
Not everyone favors moving contraceptives over the counter. Dr. W. David Hager, who practices at Baptist Health Medical Group Women’s Care in Lexington, Ky., expressed concern that providing OTC access to contraceptives would hinder the physician-patient relationship and impact screening efforts.
“One of the big advantages for me as a practicing ob.gyn. is to have face-to-face conversations, to be able to discuss risks and benefits, and to counsel young patients about the choices that they are making, to make sure they are making educated, informed choices,” Dr. Hager said.
A shift to OTC access for contraceptives could potentially decrease the uptake of human papillomavirus (HPV) vaccination, said Dr. Hager, who in the early 2000s served on the FDA’s Advisory Committee for Reproductive Health Drugs.
“We’re already at less than 50% implementation with Gardasil, and this may further decrease that,” he said. “It may delay Pap test screening and HPV screening if women choose after age 21 to just purchase over-the-counter contraceptives. It may delay well-woman screening, where women come in just for a wellness exam. And, in my opinion, it eliminates the potential for discussion of any contraindications to birth control pills. Granted, those are not many, but there are some patients who are not ideal candidates for oral contraceptives. I think it potentially takes a cohort of the highest-risk women and removes them from that potential screening and counseling session.”
Dr. DeFrancesco said he knows from experience that patients will come to see their ob.gyn. regularly just because it’s the right thing to do.
“There could be a small percentage of people who say ‘now I really don’t have to see the doctor, because that’s the only reason I was going,’ ” he said. “The good news about more people being covered with insurance is that at least people are more empowered to go to the physician’s office or the provider’s office to get care.”
States take the lead
While the federal bills are drawing attention, they are no closer to becoming law. But it’s a different story at the state level.
In Washington, the D.C. Council approved a bill that requires health plans to authorize the dispensing of up to 12 months of a woman’s prescription for contraception at one time. The legislation was signed by D.C. Mayor Muriel Bowser (D) in June but won’t apply to health plans until Jan. 1, 2017.
In Oregon, lawmakers enacted similar legislation (House Bill 3343) that requires health insurers to pay for a 12-month supply of the birth control pill, patch, or the ring at one time. The new law goes into effect on Jan. 1, 2016. The Oregon Medical Association, which supported the bill, said that the ability for women to get a 12-month supply of birth control at one time would ease administrative burdens for medical office staff, patients, and pharmacists by lessening refill requests.
In July, Oregon Gov. Kate Brown (D) also approved a related law House Bill 2879, which permits pharmacists in Oregon to prescribe hormonal contraceptive patches and self-administered oral contraceptives. The State Board of Pharmacy will develop rules for how the new system will work, but the law specifies that pharmacists must complete a training program for prescribing hormonal contraceptives, and it requires patients to use a self-screening risk assessment tool before they can receive a prescription. For minors, the law requires evidence of a previous prescription for hormonal contraceptives. The law goes into effect on Jan. 1, 2016.
In California, expanded pharmacist scope of practice legislation known as SB 493 was signed into law by Gov. Jerry Brown (D) on Oct. 1, 2013, and it became effective on Jan. 1, 2014. One of the law’s provisions enables pharmacists to furnish self-administered contraceptives according to standards developed by the California State Board of Pharmacy and the Medical Board of California.
However, this provision is still in the administrative rule-making process, said Sarah McBane, Pharm.D., president of the California Pharmacists Association. According to the original legislation, patients will be required to use a self-screening tool to identify risk factors.
There’s been a flurry of legislative activity on Capitol Hill and at state houses around the country aimed at improving access to contraception. But there are still deep divides on the best way to achieve that access.
At the federal level, there are two competing bills that address access to contraceptives.
The first – the Allowing Greater Access to Safe and Effective Contraceptive Act (S. 1438) – introduced in May by Sen. Cory Gardner (R-Colo.) and Sen. Kelly Ayotte (R-N.H.), would incentivize manufacturers of “routine-use contraceptives” to file an application with the Food and Drug Administration to switch their products from prescription to over the counter (OTC). The bill would allow for priority review for these products and waive the usual FDA filing fee.
The legislation would also repeal the Affordable Care Act’s restriction on the use of health, medical, and flexible savings accounts to purchase OTC drugs without a prescription.
Despite the fact that the bill is aimed at encouraging the switch of contraceptives to OTC products, it’s been criticized by some reproductive health advocates for not adequately addressing the potential for increased costs if women purchase OTC contraceptives not covered by insurance.
Senate Democrats responded with their own birth control legislation. The Affordability Is Access Act (S. 1532), introduced in June by Sen. Patty Murray (D-Wash.), aims to build on the no-cost coverage for contraception in the Affordable Care Act by ensuring that if the FDA approves oral contraceptives for OTC use, they will be covered without cost sharing even though they are not prescription products.
Affordability at issue
The American Congress of Obstetricians and Gynecologists is backing Sen. Murray’s bill.
In an interview, Dr. Mark S. DeFrancesco, ACOG president, said he is pleased to see proposals from both sides of the aisle emerge on this topic because ACOG has long supported the concept of OTC sale of oral contraceptives. But the GOP-backed proposal does not address the issue of copays and insurance coverage, said Dr. DeFrancesco, who is a managing partner at Westwood Women’s Health in Waterbury, Conn.
“An unintended consequence could be, ironically, that it may decrease access for women because if contraceptives are reclassified as nonformulary drugs by going over the counter, can insurance companies say they’re not going to cover them as they don’t cover aspirin, for instance?” Dr. DeFrancesco said. “That’s our concern: that well-intended legislation could have a backfire effect.”
Missed opportunity for screening?
Not everyone favors moving contraceptives over the counter. Dr. W. David Hager, who practices at Baptist Health Medical Group Women’s Care in Lexington, Ky., expressed concern that providing OTC access to contraceptives would hinder the physician-patient relationship and impact screening efforts.
“One of the big advantages for me as a practicing ob.gyn. is to have face-to-face conversations, to be able to discuss risks and benefits, and to counsel young patients about the choices that they are making, to make sure they are making educated, informed choices,” Dr. Hager said.
A shift to OTC access for contraceptives could potentially decrease the uptake of human papillomavirus (HPV) vaccination, said Dr. Hager, who in the early 2000s served on the FDA’s Advisory Committee for Reproductive Health Drugs.
“We’re already at less than 50% implementation with Gardasil, and this may further decrease that,” he said. “It may delay Pap test screening and HPV screening if women choose after age 21 to just purchase over-the-counter contraceptives. It may delay well-woman screening, where women come in just for a wellness exam. And, in my opinion, it eliminates the potential for discussion of any contraindications to birth control pills. Granted, those are not many, but there are some patients who are not ideal candidates for oral contraceptives. I think it potentially takes a cohort of the highest-risk women and removes them from that potential screening and counseling session.”
Dr. DeFrancesco said he knows from experience that patients will come to see their ob.gyn. regularly just because it’s the right thing to do.
“There could be a small percentage of people who say ‘now I really don’t have to see the doctor, because that’s the only reason I was going,’ ” he said. “The good news about more people being covered with insurance is that at least people are more empowered to go to the physician’s office or the provider’s office to get care.”
States take the lead
While the federal bills are drawing attention, they are no closer to becoming law. But it’s a different story at the state level.
In Washington, the D.C. Council approved a bill that requires health plans to authorize the dispensing of up to 12 months of a woman’s prescription for contraception at one time. The legislation was signed by D.C. Mayor Muriel Bowser (D) in June but won’t apply to health plans until Jan. 1, 2017.
In Oregon, lawmakers enacted similar legislation (House Bill 3343) that requires health insurers to pay for a 12-month supply of the birth control pill, patch, or the ring at one time. The new law goes into effect on Jan. 1, 2016. The Oregon Medical Association, which supported the bill, said that the ability for women to get a 12-month supply of birth control at one time would ease administrative burdens for medical office staff, patients, and pharmacists by lessening refill requests.
In July, Oregon Gov. Kate Brown (D) also approved a related law House Bill 2879, which permits pharmacists in Oregon to prescribe hormonal contraceptive patches and self-administered oral contraceptives. The State Board of Pharmacy will develop rules for how the new system will work, but the law specifies that pharmacists must complete a training program for prescribing hormonal contraceptives, and it requires patients to use a self-screening risk assessment tool before they can receive a prescription. For minors, the law requires evidence of a previous prescription for hormonal contraceptives. The law goes into effect on Jan. 1, 2016.
In California, expanded pharmacist scope of practice legislation known as SB 493 was signed into law by Gov. Jerry Brown (D) on Oct. 1, 2013, and it became effective on Jan. 1, 2014. One of the law’s provisions enables pharmacists to furnish self-administered contraceptives according to standards developed by the California State Board of Pharmacy and the Medical Board of California.
However, this provision is still in the administrative rule-making process, said Sarah McBane, Pharm.D., president of the California Pharmacists Association. According to the original legislation, patients will be required to use a self-screening tool to identify risk factors.
Bills emerge to increase birth control access
There’s been a flurry of legislative activity on Capitol Hill and at state houses around the country aimed at improving access to contraception. But there are still deep divides on the best way to achieve that access.
At the federal level, there are two competing bills that address access to contraceptives.
The first – the Allowing Greater Access to Safe and Effective Contraceptive Act (S. 1438) – introduced in May by Sen. Cory Gardner (R-Colo.) and Sen. Kelly Ayotte (R-N.H.), would incentivize manufacturers of “routine-use contraceptives” to file an application with the Food and Drug Administration to switch their products from prescription to over the counter (OTC). The bill would allow for priority review for these products and waive the usual FDA filing fee.
The legislation would also repeal the Affordable Care Act’s restriction on the use of health, medical, and flexible savings accounts to purchase OTC drugs without a prescription.
Despite the fact that the bill is aimed at encouraging the switch of contraceptives to OTC products, it’s been criticized by some reproductive health advocates for not adequately addressing the potential for increased costs if women purchase OTC contraceptives not covered by insurance.
Senate Democrats responded with their own birth control legislation. The Affordability Is Access Act (S. 1532), introduced in June by Sen. Patty Murray (D-Wash.), aims to build on the no-cost coverage for contraception in the Affordable Care Act by ensuring that if the FDA approves oral contraceptives for OTC use, they will be covered without cost sharing even though they are not prescription products.
Affordability at issue
The American Congress of Obstetricians and Gynecologists is backing Sen. Murray’s bill.
In an interview, Dr. Mark S. DeFrancesco, ACOG president, said he is pleased to see proposals from both sides of the aisle emerge on this topic because ACOG has long supported the concept of OTC sale of oral contraceptives. But the GOP-backed proposal does not address the issue of copays and insurance coverage, said Dr. DeFrancesco, who is a managing partner at Westwood Women’s Health in Waterbury, Conn.
“An unintended consequence could be, ironically, that it may decrease access for women because if contraceptives are reclassified as nonformulary drugs by going over the counter, can insurance companies say they’re not going to cover them as they don’t cover aspirin, for instance?” Dr. DeFrancesco said. “That’s our concern: that well-intended legislation could have a backfire effect.”
Missed opportunity for screening?
Not everyone favors moving contraceptives over the counter. Dr. W. David Hager, who practices at Baptist Health Medical Group Women’s Care in Lexington, Ky., expressed concern that providing OTC access to contraceptives would hinder the physician-patient relationship and impact screening efforts.
“One of the big advantages for me as a practicing ob.gyn. is to have face-to-face conversations, to be able to discuss risks and benefits, and to counsel young patients about the choices that they are making, to make sure they are making educated, informed choices,” Dr. Hager said.
A shift to OTC access for contraceptives could potentially decrease the uptake of human papillomavirus (HPV) vaccination, said Dr. Hager, who in the early 2000s served on the FDA’s Advisory Committee for Reproductive Health Drugs.
“We’re already at less than 50% implementation with Gardasil, and this may further decrease that,” he said. “It may delay Pap test screening and HPV screening if women choose after age 21 to just purchase over-the-counter contraceptives. It may delay well-woman screening, where women come in just for a wellness exam. And, in my opinion, it eliminates the potential for discussion of any contraindications to birth control pills. Granted, those are not many, but there are some patients who are not ideal candidates for oral contraceptives. I think it potentially takes a cohort of the highest-risk women and removes them from that potential screening and counseling session.”
Dr. DeFrancesco said he knows from experience that patients will come to see their ob.gyn. regularly just because it’s the right thing to do.
“There could be a small percentage of people who say ‘now I really don’t have to see the doctor, because that’s the only reason I was going,’ ” he said. “The good news about more people being covered with insurance is that at least people are more empowered to go to the physician’s office or the provider’s office to get care.”
States take the lead
While the federal bills are drawing attention, they are no closer to becoming law. But it’s a different story at the state level.
In Washington, the D.C. Council approved a bill that requires health plans to authorize the dispensing of up to 12 months of a woman’s prescription for contraception at one time. The legislation was signed by D.C. Mayor Muriel Bowser (D) in June but won’t apply to health plans until Jan. 1, 2017.
In Oregon, lawmakers enacted similar legislation (House Bill 3343) that requires health insurers to pay for a 12-month supply of the birth control pill, patch, or the ring at one time. The new law goes into effect on Jan. 1, 2016. The Oregon Medical Association, which supported the bill, said that the ability for women to get a 12-month supply of birth control at one time would ease administrative burdens for medical office staff, patients, and pharmacists by lessening refill requests.
In July, Oregon Gov. Kate Brown (D) also approved a related law House Bill 2879, which permits pharmacists in Oregon to prescribe hormonal contraceptive patches and self-administered oral contraceptives. The State Board of Pharmacy will develop rules for how the new system will work, but the law specifies that pharmacists must complete a training program for prescribing hormonal contraceptives, and it requires patients to use a self-screening risk assessment tool before they can receive a prescription. For minors, the law requires evidence of a previous prescription for hormonal contraceptives. The law goes into effect on Jan. 1, 2016.
In California, expanded pharmacist scope of practice legislation known as SB 493 was signed into law by Gov. Jerry Brown (D) on Oct. 1, 2013, and it became effective on Jan. 1, 2014. One of the law’s provisions enables pharmacists to furnish self-administered contraceptives according to standards developed by the California State Board of Pharmacy and the Medical Board of California.
However, this provision is still in the administrative rule-making process, said Sarah McBane, Pharm.D., president of the California Pharmacists Association. According to the original legislation, patients will be required to use a self-screening tool to identify risk factors.
There’s been a flurry of legislative activity on Capitol Hill and at state houses around the country aimed at improving access to contraception. But there are still deep divides on the best way to achieve that access.
At the federal level, there are two competing bills that address access to contraceptives.
The first – the Allowing Greater Access to Safe and Effective Contraceptive Act (S. 1438) – introduced in May by Sen. Cory Gardner (R-Colo.) and Sen. Kelly Ayotte (R-N.H.), would incentivize manufacturers of “routine-use contraceptives” to file an application with the Food and Drug Administration to switch their products from prescription to over the counter (OTC). The bill would allow for priority review for these products and waive the usual FDA filing fee.
The legislation would also repeal the Affordable Care Act’s restriction on the use of health, medical, and flexible savings accounts to purchase OTC drugs without a prescription.
Despite the fact that the bill is aimed at encouraging the switch of contraceptives to OTC products, it’s been criticized by some reproductive health advocates for not adequately addressing the potential for increased costs if women purchase OTC contraceptives not covered by insurance.
Senate Democrats responded with their own birth control legislation. The Affordability Is Access Act (S. 1532), introduced in June by Sen. Patty Murray (D-Wash.), aims to build on the no-cost coverage for contraception in the Affordable Care Act by ensuring that if the FDA approves oral contraceptives for OTC use, they will be covered without cost sharing even though they are not prescription products.
Affordability at issue
The American Congress of Obstetricians and Gynecologists is backing Sen. Murray’s bill.
In an interview, Dr. Mark S. DeFrancesco, ACOG president, said he is pleased to see proposals from both sides of the aisle emerge on this topic because ACOG has long supported the concept of OTC sale of oral contraceptives. But the GOP-backed proposal does not address the issue of copays and insurance coverage, said Dr. DeFrancesco, who is a managing partner at Westwood Women’s Health in Waterbury, Conn.
“An unintended consequence could be, ironically, that it may decrease access for women because if contraceptives are reclassified as nonformulary drugs by going over the counter, can insurance companies say they’re not going to cover them as they don’t cover aspirin, for instance?” Dr. DeFrancesco said. “That’s our concern: that well-intended legislation could have a backfire effect.”
Missed opportunity for screening?
Not everyone favors moving contraceptives over the counter. Dr. W. David Hager, who practices at Baptist Health Medical Group Women’s Care in Lexington, Ky., expressed concern that providing OTC access to contraceptives would hinder the physician-patient relationship and impact screening efforts.
“One of the big advantages for me as a practicing ob.gyn. is to have face-to-face conversations, to be able to discuss risks and benefits, and to counsel young patients about the choices that they are making, to make sure they are making educated, informed choices,” Dr. Hager said.
A shift to OTC access for contraceptives could potentially decrease the uptake of human papillomavirus (HPV) vaccination, said Dr. Hager, who in the early 2000s served on the FDA’s Advisory Committee for Reproductive Health Drugs.
“We’re already at less than 50% implementation with Gardasil, and this may further decrease that,” he said. “It may delay Pap test screening and HPV screening if women choose after age 21 to just purchase over-the-counter contraceptives. It may delay well-woman screening, where women come in just for a wellness exam. And, in my opinion, it eliminates the potential for discussion of any contraindications to birth control pills. Granted, those are not many, but there are some patients who are not ideal candidates for oral contraceptives. I think it potentially takes a cohort of the highest-risk women and removes them from that potential screening and counseling session.”
Dr. DeFrancesco said he knows from experience that patients will come to see their ob.gyn. regularly just because it’s the right thing to do.
“There could be a small percentage of people who say ‘now I really don’t have to see the doctor, because that’s the only reason I was going,’ ” he said. “The good news about more people being covered with insurance is that at least people are more empowered to go to the physician’s office or the provider’s office to get care.”
States take the lead
While the federal bills are drawing attention, they are no closer to becoming law. But it’s a different story at the state level.
In Washington, the D.C. Council approved a bill that requires health plans to authorize the dispensing of up to 12 months of a woman’s prescription for contraception at one time. The legislation was signed by D.C. Mayor Muriel Bowser (D) in June but won’t apply to health plans until Jan. 1, 2017.
In Oregon, lawmakers enacted similar legislation (House Bill 3343) that requires health insurers to pay for a 12-month supply of the birth control pill, patch, or the ring at one time. The new law goes into effect on Jan. 1, 2016. The Oregon Medical Association, which supported the bill, said that the ability for women to get a 12-month supply of birth control at one time would ease administrative burdens for medical office staff, patients, and pharmacists by lessening refill requests.
In July, Oregon Gov. Kate Brown (D) also approved a related law House Bill 2879, which permits pharmacists in Oregon to prescribe hormonal contraceptive patches and self-administered oral contraceptives. The State Board of Pharmacy will develop rules for how the new system will work, but the law specifies that pharmacists must complete a training program for prescribing hormonal contraceptives, and it requires patients to use a self-screening risk assessment tool before they can receive a prescription. For minors, the law requires evidence of a previous prescription for hormonal contraceptives. The law goes into effect on Jan. 1, 2016.
In California, expanded pharmacist scope of practice legislation known as SB 493 was signed into law by Gov. Jerry Brown (D) on Oct. 1, 2013, and it became effective on Jan. 1, 2014. One of the law’s provisions enables pharmacists to furnish self-administered contraceptives according to standards developed by the California State Board of Pharmacy and the Medical Board of California.
However, this provision is still in the administrative rule-making process, said Sarah McBane, Pharm.D., president of the California Pharmacists Association. According to the original legislation, patients will be required to use a self-screening tool to identify risk factors.
There’s been a flurry of legislative activity on Capitol Hill and at state houses around the country aimed at improving access to contraception. But there are still deep divides on the best way to achieve that access.
At the federal level, there are two competing bills that address access to contraceptives.
The first – the Allowing Greater Access to Safe and Effective Contraceptive Act (S. 1438) – introduced in May by Sen. Cory Gardner (R-Colo.) and Sen. Kelly Ayotte (R-N.H.), would incentivize manufacturers of “routine-use contraceptives” to file an application with the Food and Drug Administration to switch their products from prescription to over the counter (OTC). The bill would allow for priority review for these products and waive the usual FDA filing fee.
The legislation would also repeal the Affordable Care Act’s restriction on the use of health, medical, and flexible savings accounts to purchase OTC drugs without a prescription.
Despite the fact that the bill is aimed at encouraging the switch of contraceptives to OTC products, it’s been criticized by some reproductive health advocates for not adequately addressing the potential for increased costs if women purchase OTC contraceptives not covered by insurance.
Senate Democrats responded with their own birth control legislation. The Affordability Is Access Act (S. 1532), introduced in June by Sen. Patty Murray (D-Wash.), aims to build on the no-cost coverage for contraception in the Affordable Care Act by ensuring that if the FDA approves oral contraceptives for OTC use, they will be covered without cost sharing even though they are not prescription products.
Affordability at issue
The American Congress of Obstetricians and Gynecologists is backing Sen. Murray’s bill.
In an interview, Dr. Mark S. DeFrancesco, ACOG president, said he is pleased to see proposals from both sides of the aisle emerge on this topic because ACOG has long supported the concept of OTC sale of oral contraceptives. But the GOP-backed proposal does not address the issue of copays and insurance coverage, said Dr. DeFrancesco, who is a managing partner at Westwood Women’s Health in Waterbury, Conn.
“An unintended consequence could be, ironically, that it may decrease access for women because if contraceptives are reclassified as nonformulary drugs by going over the counter, can insurance companies say they’re not going to cover them as they don’t cover aspirin, for instance?” Dr. DeFrancesco said. “That’s our concern: that well-intended legislation could have a backfire effect.”
Missed opportunity for screening?
Not everyone favors moving contraceptives over the counter. Dr. W. David Hager, who practices at Baptist Health Medical Group Women’s Care in Lexington, Ky., expressed concern that providing OTC access to contraceptives would hinder the physician-patient relationship and impact screening efforts.
“One of the big advantages for me as a practicing ob.gyn. is to have face-to-face conversations, to be able to discuss risks and benefits, and to counsel young patients about the choices that they are making, to make sure they are making educated, informed choices,” Dr. Hager said.
A shift to OTC access for contraceptives could potentially decrease the uptake of human papillomavirus (HPV) vaccination, said Dr. Hager, who in the early 2000s served on the FDA’s Advisory Committee for Reproductive Health Drugs.
“We’re already at less than 50% implementation with Gardasil, and this may further decrease that,” he said. “It may delay Pap test screening and HPV screening if women choose after age 21 to just purchase over-the-counter contraceptives. It may delay well-woman screening, where women come in just for a wellness exam. And, in my opinion, it eliminates the potential for discussion of any contraindications to birth control pills. Granted, those are not many, but there are some patients who are not ideal candidates for oral contraceptives. I think it potentially takes a cohort of the highest-risk women and removes them from that potential screening and counseling session.”
Dr. DeFrancesco said he knows from experience that patients will come to see their ob.gyn. regularly just because it’s the right thing to do.
“There could be a small percentage of people who say ‘now I really don’t have to see the doctor, because that’s the only reason I was going,’ ” he said. “The good news about more people being covered with insurance is that at least people are more empowered to go to the physician’s office or the provider’s office to get care.”
States take the lead
While the federal bills are drawing attention, they are no closer to becoming law. But it’s a different story at the state level.
In Washington, the D.C. Council approved a bill that requires health plans to authorize the dispensing of up to 12 months of a woman’s prescription for contraception at one time. The legislation was signed by D.C. Mayor Muriel Bowser (D) in June but won’t apply to health plans until Jan. 1, 2017.
In Oregon, lawmakers enacted similar legislation (House Bill 3343) that requires health insurers to pay for a 12-month supply of the birth control pill, patch, or the ring at one time. The new law goes into effect on Jan. 1, 2016. The Oregon Medical Association, which supported the bill, said that the ability for women to get a 12-month supply of birth control at one time would ease administrative burdens for medical office staff, patients, and pharmacists by lessening refill requests.
In July, Oregon Gov. Kate Brown (D) also approved a related law House Bill 2879, which permits pharmacists in Oregon to prescribe hormonal contraceptive patches and self-administered oral contraceptives. The State Board of Pharmacy will develop rules for how the new system will work, but the law specifies that pharmacists must complete a training program for prescribing hormonal contraceptives, and it requires patients to use a self-screening risk assessment tool before they can receive a prescription. For minors, the law requires evidence of a previous prescription for hormonal contraceptives. The law goes into effect on Jan. 1, 2016.
In California, expanded pharmacist scope of practice legislation known as SB 493 was signed into law by Gov. Jerry Brown (D) on Oct. 1, 2013, and it became effective on Jan. 1, 2014. One of the law’s provisions enables pharmacists to furnish self-administered contraceptives according to standards developed by the California State Board of Pharmacy and the Medical Board of California.
However, this provision is still in the administrative rule-making process, said Sarah McBane, Pharm.D., president of the California Pharmacists Association. According to the original legislation, patients will be required to use a self-screening tool to identify risk factors.
Routine screening sufficient for detecting occult cancer in patients with VTE
TORONTO – The prevalence of occult cancer is low in patients with a first unprovoked venous thromboembolism, according to results from a multicenter, randomized study presented at the International Society on Thrombosis and Haemostasis congress.
In addition, routine screening with the addition of a comprehensive CT scan of the abdomen and pelvis was no better than routine screening alone in detecting occult cancer in this population.
Those are key findings that Dr. Marc Carrier of the University of Ottawa presented from the Screening for Occult Malignancy in Patients with Idiopathic Venous Thromboembolism (SOME) trial, a multicenter, open-label, randomized controlled trial that compared the efficacy of conventional screening with or without comprehensive CT of the abdomen/pelvis for detecting occult cancers in patients with unprovoked venous thromboembolism (VTE). The results of this study were published the same day as his presentation in the New England Journal of Medicine.
“It has been described that up to 10% of patients with unprovoked VTE are diagnosed with cancer in the year following their VTE diagnosis,” Dr. Carrier said. “Therefore, it’s appealing for clinicians to screen these patients for occult cancer but it has led to a lot of great diversity in practices. Some clinicians prefer to use a limited screening strategy that would include a history, physical examination, routine blood tests, and a chest X-ray. Other clinicians prefer to use the limited screening strategy in combination with additional tests. That could be CT of the abdomen and pelvis, ultrasound, or tumor marker, or [computed axial tomography] scan. It’s hard for a physician to know what to use.”
For the SOME trial, a total of 854 patients with unprovoked VTE were randomized to two groups: 431 to limited occult cancer screening (basic blood work, chest X-ray, and breast/cervical/prostate cancer screening) and 423 to limited screening in combination with a comprehensive CT of the abdomen/pelvis. The comprehensive CT included a virtual colonoscopy and gastroscopy, a biphasic enhanced CT, a parenchymal pancreatogram, and a uniphasic enhanced CT of distended bladder. The primary outcome was confirmed cancer that was missed by the screening strategy and detected by the end of the 1-year follow-up period.
Dr. Carrier reported that 33 patients (3.9%) had a new diagnosis of cancer in the interval between randomization and 1-year follow-up: 14 in the limited-screening group and 19 in the limited-screening-plus-CT group, a difference that was not statistically significant (P = .28). In addition, the number of occult cancers missed by the end of the 1-year follow-up period was similar between the two groups: four in the limited-screening group and five in the limited-screening-plus-CT group.
He and his associates also found no significant differences between the limited-screening group and the limited-screening-plus-CT group in the rate of detection of early cancers (0.23% vs. 0.71%, respectively; P = .37), in overall mortality (1.4% vs. 1.2%; P > 0.99), or in cancer-related mortality (1.4% vs. 0.95%; P = .75).
“Occult cancers are not nearly as common as we thought they were, which is reassuring for clinicians and patients because then we don’t have to do a lot of investigations to try and find them, and often scare patients and expose them to radiation and additional procedures,” Dr. Carrier said in an interview. “Limited screening alone, which is what is recommended in Canada and in the United States for age- and gender-specific screening, is more than reasonable for these patients.”
The SOME trial was funded by the Heart and Stroke Foundation of Canada. Dr. Carrier had no relevant financial conflicts to disclose.
Therese Borden contributed to this article.
TORONTO – The prevalence of occult cancer is low in patients with a first unprovoked venous thromboembolism, according to results from a multicenter, randomized study presented at the International Society on Thrombosis and Haemostasis congress.
In addition, routine screening with the addition of a comprehensive CT scan of the abdomen and pelvis was no better than routine screening alone in detecting occult cancer in this population.
Those are key findings that Dr. Marc Carrier of the University of Ottawa presented from the Screening for Occult Malignancy in Patients with Idiopathic Venous Thromboembolism (SOME) trial, a multicenter, open-label, randomized controlled trial that compared the efficacy of conventional screening with or without comprehensive CT of the abdomen/pelvis for detecting occult cancers in patients with unprovoked venous thromboembolism (VTE). The results of this study were published the same day as his presentation in the New England Journal of Medicine.
“It has been described that up to 10% of patients with unprovoked VTE are diagnosed with cancer in the year following their VTE diagnosis,” Dr. Carrier said. “Therefore, it’s appealing for clinicians to screen these patients for occult cancer but it has led to a lot of great diversity in practices. Some clinicians prefer to use a limited screening strategy that would include a history, physical examination, routine blood tests, and a chest X-ray. Other clinicians prefer to use the limited screening strategy in combination with additional tests. That could be CT of the abdomen and pelvis, ultrasound, or tumor marker, or [computed axial tomography] scan. It’s hard for a physician to know what to use.”
For the SOME trial, a total of 854 patients with unprovoked VTE were randomized to two groups: 431 to limited occult cancer screening (basic blood work, chest X-ray, and breast/cervical/prostate cancer screening) and 423 to limited screening in combination with a comprehensive CT of the abdomen/pelvis. The comprehensive CT included a virtual colonoscopy and gastroscopy, a biphasic enhanced CT, a parenchymal pancreatogram, and a uniphasic enhanced CT of distended bladder. The primary outcome was confirmed cancer that was missed by the screening strategy and detected by the end of the 1-year follow-up period.
Dr. Carrier reported that 33 patients (3.9%) had a new diagnosis of cancer in the interval between randomization and 1-year follow-up: 14 in the limited-screening group and 19 in the limited-screening-plus-CT group, a difference that was not statistically significant (P = .28). In addition, the number of occult cancers missed by the end of the 1-year follow-up period was similar between the two groups: four in the limited-screening group and five in the limited-screening-plus-CT group.
He and his associates also found no significant differences between the limited-screening group and the limited-screening-plus-CT group in the rate of detection of early cancers (0.23% vs. 0.71%, respectively; P = .37), in overall mortality (1.4% vs. 1.2%; P > 0.99), or in cancer-related mortality (1.4% vs. 0.95%; P = .75).
“Occult cancers are not nearly as common as we thought they were, which is reassuring for clinicians and patients because then we don’t have to do a lot of investigations to try and find them, and often scare patients and expose them to radiation and additional procedures,” Dr. Carrier said in an interview. “Limited screening alone, which is what is recommended in Canada and in the United States for age- and gender-specific screening, is more than reasonable for these patients.”
The SOME trial was funded by the Heart and Stroke Foundation of Canada. Dr. Carrier had no relevant financial conflicts to disclose.
Therese Borden contributed to this article.
TORONTO – The prevalence of occult cancer is low in patients with a first unprovoked venous thromboembolism, according to results from a multicenter, randomized study presented at the International Society on Thrombosis and Haemostasis congress.
In addition, routine screening with the addition of a comprehensive CT scan of the abdomen and pelvis was no better than routine screening alone in detecting occult cancer in this population.
Those are key findings that Dr. Marc Carrier of the University of Ottawa presented from the Screening for Occult Malignancy in Patients with Idiopathic Venous Thromboembolism (SOME) trial, a multicenter, open-label, randomized controlled trial that compared the efficacy of conventional screening with or without comprehensive CT of the abdomen/pelvis for detecting occult cancers in patients with unprovoked venous thromboembolism (VTE). The results of this study were published the same day as his presentation in the New England Journal of Medicine.
“It has been described that up to 10% of patients with unprovoked VTE are diagnosed with cancer in the year following their VTE diagnosis,” Dr. Carrier said. “Therefore, it’s appealing for clinicians to screen these patients for occult cancer but it has led to a lot of great diversity in practices. Some clinicians prefer to use a limited screening strategy that would include a history, physical examination, routine blood tests, and a chest X-ray. Other clinicians prefer to use the limited screening strategy in combination with additional tests. That could be CT of the abdomen and pelvis, ultrasound, or tumor marker, or [computed axial tomography] scan. It’s hard for a physician to know what to use.”
For the SOME trial, a total of 854 patients with unprovoked VTE were randomized to two groups: 431 to limited occult cancer screening (basic blood work, chest X-ray, and breast/cervical/prostate cancer screening) and 423 to limited screening in combination with a comprehensive CT of the abdomen/pelvis. The comprehensive CT included a virtual colonoscopy and gastroscopy, a biphasic enhanced CT, a parenchymal pancreatogram, and a uniphasic enhanced CT of distended bladder. The primary outcome was confirmed cancer that was missed by the screening strategy and detected by the end of the 1-year follow-up period.
Dr. Carrier reported that 33 patients (3.9%) had a new diagnosis of cancer in the interval between randomization and 1-year follow-up: 14 in the limited-screening group and 19 in the limited-screening-plus-CT group, a difference that was not statistically significant (P = .28). In addition, the number of occult cancers missed by the end of the 1-year follow-up period was similar between the two groups: four in the limited-screening group and five in the limited-screening-plus-CT group.
He and his associates also found no significant differences between the limited-screening group and the limited-screening-plus-CT group in the rate of detection of early cancers (0.23% vs. 0.71%, respectively; P = .37), in overall mortality (1.4% vs. 1.2%; P > 0.99), or in cancer-related mortality (1.4% vs. 0.95%; P = .75).
“Occult cancers are not nearly as common as we thought they were, which is reassuring for clinicians and patients because then we don’t have to do a lot of investigations to try and find them, and often scare patients and expose them to radiation and additional procedures,” Dr. Carrier said in an interview. “Limited screening alone, which is what is recommended in Canada and in the United States for age- and gender-specific screening, is more than reasonable for these patients.”
The SOME trial was funded by the Heart and Stroke Foundation of Canada. Dr. Carrier had no relevant financial conflicts to disclose.
Therese Borden contributed to this article.
AT THE 2015 ISTH CONGRESS
Key clinical point: Occult cancers in patients with a first unprovoked VTE are not nearly as common as previously thought, and limited screening for such cancers is appropriate.
Major finding: There were no significant differences between the limited-screening group and the limited-screening-plus-CT group in the rate of detection of early cancers (0.23% vs. 0.71%); in overall mortality (1.4% vs. 1.2%), or in cancer-related mortality (1.4% vs. 0.95%).
Data source: A multicenter, open-label, randomized controlled trial of 854 patients with unprovoked VTE.
Disclosures: The trial was funded by the Heart and Stroke Foundation of Canada. Dr. Carrier reported having no financial disclosures.