User login
VIDEO: More gonadotropin, fewer live births
SAN FRANCISCO– The higher the dose of gonadotropin for ovarian stimulation, the lower the live birth rate, a retrospective study of 541,967 cycles of assisted reproductive technology showed.
The inverse correlation was less evident in older patients than in younger patients and in those who might be expected have a more normal response, Dr. Valerie L. Baker and her associates reported at the annual meeting of the American Society for Reproductive Medicine.
The findings were consistent regardless of the number of oocytes retrieved or the woman’s age, said Dr. Baker, chief of the division of reproductive endocrinology and infertility at Stanford (Calif.) University.
In a video interview, she talked about the study and why it has made her think twice about using very high doses of gonadotropins.
Dr. Baker reported financial associations with Good Start Genetics, Ovuline, Roche, the Society for Assisted Reproductive Technologies, and Teva Pharmaceuticals.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO– The higher the dose of gonadotropin for ovarian stimulation, the lower the live birth rate, a retrospective study of 541,967 cycles of assisted reproductive technology showed.
The inverse correlation was less evident in older patients than in younger patients and in those who might be expected have a more normal response, Dr. Valerie L. Baker and her associates reported at the annual meeting of the American Society for Reproductive Medicine.
The findings were consistent regardless of the number of oocytes retrieved or the woman’s age, said Dr. Baker, chief of the division of reproductive endocrinology and infertility at Stanford (Calif.) University.
In a video interview, she talked about the study and why it has made her think twice about using very high doses of gonadotropins.
Dr. Baker reported financial associations with Good Start Genetics, Ovuline, Roche, the Society for Assisted Reproductive Technologies, and Teva Pharmaceuticals.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN FRANCISCO– The higher the dose of gonadotropin for ovarian stimulation, the lower the live birth rate, a retrospective study of 541,967 cycles of assisted reproductive technology showed.
The inverse correlation was less evident in older patients than in younger patients and in those who might be expected have a more normal response, Dr. Valerie L. Baker and her associates reported at the annual meeting of the American Society for Reproductive Medicine.
The findings were consistent regardless of the number of oocytes retrieved or the woman’s age, said Dr. Baker, chief of the division of reproductive endocrinology and infertility at Stanford (Calif.) University.
In a video interview, she talked about the study and why it has made her think twice about using very high doses of gonadotropins.
Dr. Baker reported financial associations with Good Start Genetics, Ovuline, Roche, the Society for Assisted Reproductive Technologies, and Teva Pharmaceuticals.
On Twitter @sherryboschert
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT 2014 ASRM
VIDEO: Ovarian function is exciting possible CV risk marker
HONOLULU– A woman’s ovarian “clock” is dissociated from her general health, according to widely accepted thinking – but a new study has suggested the opposite.
That study showed greater cardiovascular risk in women with markers of greater ovarian aging. Dr. Marcelle I. Cedars of the University of California, San Francisco, and her associates presented the results at the annual meeting of the American Society for Reproductive Medicine, where the study won an award.
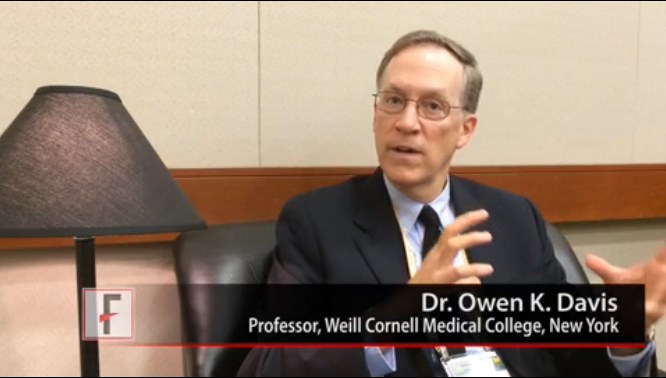
In a video interview, the society’s vice president, Dr. Owen K. Davis, described the potential significance of the study.
Women with cardiovascular disease tend to get diagnosed later and have worse outcomes, compared with men. If markers of ovarian function can help identify cardiovascular risk earlier in some women, the women may be candidates for more intensive surveillance, said Dr. Davis, professor of ob.gyn. and reproductive medicine at Weill Cornell Medical College, New York.
Dr. Davis reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
HONOLULU– A woman’s ovarian “clock” is dissociated from her general health, according to widely accepted thinking – but a new study has suggested the opposite.
That study showed greater cardiovascular risk in women with markers of greater ovarian aging. Dr. Marcelle I. Cedars of the University of California, San Francisco, and her associates presented the results at the annual meeting of the American Society for Reproductive Medicine, where the study won an award.
In a video interview, the society’s vice president, Dr. Owen K. Davis, described the potential significance of the study.
Women with cardiovascular disease tend to get diagnosed later and have worse outcomes, compared with men. If markers of ovarian function can help identify cardiovascular risk earlier in some women, the women may be candidates for more intensive surveillance, said Dr. Davis, professor of ob.gyn. and reproductive medicine at Weill Cornell Medical College, New York.
Dr. Davis reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
HONOLULU– A woman’s ovarian “clock” is dissociated from her general health, according to widely accepted thinking – but a new study has suggested the opposite.
That study showed greater cardiovascular risk in women with markers of greater ovarian aging. Dr. Marcelle I. Cedars of the University of California, San Francisco, and her associates presented the results at the annual meeting of the American Society for Reproductive Medicine, where the study won an award.
In a video interview, the society’s vice president, Dr. Owen K. Davis, described the potential significance of the study.
Women with cardiovascular disease tend to get diagnosed later and have worse outcomes, compared with men. If markers of ovarian function can help identify cardiovascular risk earlier in some women, the women may be candidates for more intensive surveillance, said Dr. Davis, professor of ob.gyn. and reproductive medicine at Weill Cornell Medical College, New York.
Dr. Davis reported having no financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @sherryboschert
EXPERT ANALYSIS FROM ASRM 2014
VIDEO: How U.S. health providers can contain Ebola
AUSTIN, TEX. – Why would travel restrictions make the Ebola epidemic worse in West Africa and expand its spread to U.S. shores? What can American intensive care units do to prepare, even if they aren’t designated Ebola centers? And are there lessons medical professionals and policymakers can apply from the successes and failures of the AIDS epidemic 30 years ago?
In a video interview at the annual meeting of the American College of Chest Physicians, Dr. Lewis Rubinson offered answers to those questions and perspectives on the current American response to the Ebola outbreak. Dr. Rubinson recently returned from treating more than 300 Ebola patients in Sierra Leone as a consulting physician for the World Health Organization.
“Honestly, I think if we don’t get it under control in the next few months in West Africa, there will be sporadic cases coming back [to the United States] for as long as we can think of,” cautioned Dr. Rubinson, director of the R. Adams Cowley Trauma Shock Center at the University of Maryland, Baltimore.
On Twitter @whitneymcknight
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AUSTIN, TEX. – Why would travel restrictions make the Ebola epidemic worse in West Africa and expand its spread to U.S. shores? What can American intensive care units do to prepare, even if they aren’t designated Ebola centers? And are there lessons medical professionals and policymakers can apply from the successes and failures of the AIDS epidemic 30 years ago?
In a video interview at the annual meeting of the American College of Chest Physicians, Dr. Lewis Rubinson offered answers to those questions and perspectives on the current American response to the Ebola outbreak. Dr. Rubinson recently returned from treating more than 300 Ebola patients in Sierra Leone as a consulting physician for the World Health Organization.
“Honestly, I think if we don’t get it under control in the next few months in West Africa, there will be sporadic cases coming back [to the United States] for as long as we can think of,” cautioned Dr. Rubinson, director of the R. Adams Cowley Trauma Shock Center at the University of Maryland, Baltimore.
On Twitter @whitneymcknight
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AUSTIN, TEX. – Why would travel restrictions make the Ebola epidemic worse in West Africa and expand its spread to U.S. shores? What can American intensive care units do to prepare, even if they aren’t designated Ebola centers? And are there lessons medical professionals and policymakers can apply from the successes and failures of the AIDS epidemic 30 years ago?
In a video interview at the annual meeting of the American College of Chest Physicians, Dr. Lewis Rubinson offered answers to those questions and perspectives on the current American response to the Ebola outbreak. Dr. Rubinson recently returned from treating more than 300 Ebola patients in Sierra Leone as a consulting physician for the World Health Organization.
“Honestly, I think if we don’t get it under control in the next few months in West Africa, there will be sporadic cases coming back [to the United States] for as long as we can think of,” cautioned Dr. Rubinson, director of the R. Adams Cowley Trauma Shock Center at the University of Maryland, Baltimore.
On Twitter @whitneymcknight
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT CHEST 2014
Baystate Medical Center's Unit-Based, Multidisciplinary Rounding Enhances Inpatient Care
The hospitalist-led Broder Service empowers all care-team members to focus on patient quality, satisfaction. Get an up-close look at the service with our 6-minute feature video:
The hospitalist-led Broder Service empowers all care-team members to focus on patient quality, satisfaction. Get an up-close look at the service with our 6-minute feature video:
The hospitalist-led Broder Service empowers all care-team members to focus on patient quality, satisfaction. Get an up-close look at the service with our 6-minute feature video:
VIDEO: Experts call for tighter regulation of glucose monitoring devices
WASHINGTON – Congress should back better regulation of glucose-monitoring technologies and expanded access to continuous glucose monitoring for patients with either type 1 or insulin-dependent type 2 diabetes, according to two endocrinology organizations.
At a consensus conference on glucose monitoring, hosted by the American Association of Clinical Endocrinologists and the American College of Endocrinology, advocates pushed for federal legislation that would increase the role endocrinology experts play in setting policy and reimbursement for glucose monitoring.
Inaccurate glucose level readings can have devastating effects, ranging from seizures to coma or even death. Self-test glucose monitoring kits are currently considered the standard of care for people with type 1 diabetes or with insulin-dependent type-2 diabetes.
However, the reliability of the kits has come into question over the years – although knowing which kits are defective is difficult, endocrinology experts noted. That’s in part because adverse event reporting by manufacturers is voluntary, and any data that are collected are not well reviewed.
“What this impressed upon me is the inherent unfairness in the playing field right now,” said Dr. Daniel Einhorn, past president of the AACE and medical director for the Scripps Whittier Diabetes Institute in La Jolla, Calif. “It’s possible to be a company that is less ethical and yet still make it in the market once cleared [by the FDA], because it’s hard to remove it.”
Abbott, Dexcom, Johnson and Johnson, Medtronic, Merck, and several other medical manufacturers supported the conference. Dr. Einhorn disclosed financial relationships with Janssen, Lilly, and Sanofi.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
WASHINGTON – Congress should back better regulation of glucose-monitoring technologies and expanded access to continuous glucose monitoring for patients with either type 1 or insulin-dependent type 2 diabetes, according to two endocrinology organizations.
At a consensus conference on glucose monitoring, hosted by the American Association of Clinical Endocrinologists and the American College of Endocrinology, advocates pushed for federal legislation that would increase the role endocrinology experts play in setting policy and reimbursement for glucose monitoring.
Inaccurate glucose level readings can have devastating effects, ranging from seizures to coma or even death. Self-test glucose monitoring kits are currently considered the standard of care for people with type 1 diabetes or with insulin-dependent type-2 diabetes.
However, the reliability of the kits has come into question over the years – although knowing which kits are defective is difficult, endocrinology experts noted. That’s in part because adverse event reporting by manufacturers is voluntary, and any data that are collected are not well reviewed.
“What this impressed upon me is the inherent unfairness in the playing field right now,” said Dr. Daniel Einhorn, past president of the AACE and medical director for the Scripps Whittier Diabetes Institute in La Jolla, Calif. “It’s possible to be a company that is less ethical and yet still make it in the market once cleared [by the FDA], because it’s hard to remove it.”
Abbott, Dexcom, Johnson and Johnson, Medtronic, Merck, and several other medical manufacturers supported the conference. Dr. Einhorn disclosed financial relationships with Janssen, Lilly, and Sanofi.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
WASHINGTON – Congress should back better regulation of glucose-monitoring technologies and expanded access to continuous glucose monitoring for patients with either type 1 or insulin-dependent type 2 diabetes, according to two endocrinology organizations.
At a consensus conference on glucose monitoring, hosted by the American Association of Clinical Endocrinologists and the American College of Endocrinology, advocates pushed for federal legislation that would increase the role endocrinology experts play in setting policy and reimbursement for glucose monitoring.
Inaccurate glucose level readings can have devastating effects, ranging from seizures to coma or even death. Self-test glucose monitoring kits are currently considered the standard of care for people with type 1 diabetes or with insulin-dependent type-2 diabetes.
However, the reliability of the kits has come into question over the years – although knowing which kits are defective is difficult, endocrinology experts noted. That’s in part because adverse event reporting by manufacturers is voluntary, and any data that are collected are not well reviewed.
“What this impressed upon me is the inherent unfairness in the playing field right now,” said Dr. Daniel Einhorn, past president of the AACE and medical director for the Scripps Whittier Diabetes Institute in La Jolla, Calif. “It’s possible to be a company that is less ethical and yet still make it in the market once cleared [by the FDA], because it’s hard to remove it.”
Abbott, Dexcom, Johnson and Johnson, Medtronic, Merck, and several other medical manufacturers supported the conference. Dr. Einhorn disclosed financial relationships with Janssen, Lilly, and Sanofi.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
On Twitter @whitneymcknight
AT AN AACE/ACE CONFERENCE
VIDEO: A practical protocol for monitoring discoid lupus
NEWPORT BEACH, CALIF.– Patients with discoid lupus should be checked routinely for systemic disease, but complete autoantibody studies aren’t necessary for those patients with a prior work-up and a documented negative antinuclear antibodies, according to Dr. Ruth Ann Vleugels.
“The reason why we are doing these investigations is because between 5% and up to about 20% of our discoid lupus patients can develop systemic disease,” said Dr. Vleugels of Brigham and Women’s Hospital in Boston, Mass. Some patients don’t develop symptoms until years later, she noted.
In an interview at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar, Dr. Vleugels shared her protocol for managing discoid lupus patients. She had no financial conflicts to disclose. SDEF and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEWPORT BEACH, CALIF.– Patients with discoid lupus should be checked routinely for systemic disease, but complete autoantibody studies aren’t necessary for those patients with a prior work-up and a documented negative antinuclear antibodies, according to Dr. Ruth Ann Vleugels.
“The reason why we are doing these investigations is because between 5% and up to about 20% of our discoid lupus patients can develop systemic disease,” said Dr. Vleugels of Brigham and Women’s Hospital in Boston, Mass. Some patients don’t develop symptoms until years later, she noted.
In an interview at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar, Dr. Vleugels shared her protocol for managing discoid lupus patients. She had no financial conflicts to disclose. SDEF and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
NEWPORT BEACH, CALIF.– Patients with discoid lupus should be checked routinely for systemic disease, but complete autoantibody studies aren’t necessary for those patients with a prior work-up and a documented negative antinuclear antibodies, according to Dr. Ruth Ann Vleugels.
“The reason why we are doing these investigations is because between 5% and up to about 20% of our discoid lupus patients can develop systemic disease,” said Dr. Vleugels of Brigham and Women’s Hospital in Boston, Mass. Some patients don’t develop symptoms until years later, she noted.
In an interview at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar, Dr. Vleugels shared her protocol for managing discoid lupus patients. She had no financial conflicts to disclose. SDEF and this news organization are owned by Frontline Medical Communications.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
VIDEO: Lung-MAP charts new ground in trial design
MADRID – Evaluating one new oncology drug is hard enough, but a novel collaborative clinical trial is taking on the task of testing five experimental drugs at once.
The Lung-MAP trial is using genomic profiling to direct patients to one of five simultaneous sub-studies that best fits their tumor’s genomic profile.
The treatments are for second-line, recurrent squamous cell lung cancer, an area of tremendous unmet need, according to Dr. Roy Herbst, chair of the Lung-MAP steering committee and chief of medical oncology at the Yale Cancer Center, New Haven, Conn.
The novel design of the phase II/III trial should speed up assessment and approval of investigational drugs, and improve the likelihood that patients from the most highly academic to the most rural community sites will receive a targeted therapy.
“It’s really open throughout the country and that makes me very proud that we’re bringing molecular profiling and new drugs to patients of all resource levels,” Dr. Herbst said.
Watch our interview with Dr. Herbst at the annual congress of the European Society for Clinical Oncology, where he spoke about Lung-MAP in a special session on new clinical trial designs.
LungMAP is sponsored by the Southwest Oncology Group with collaboration from several public and private partners including the National Cancer Institute.
Groups interested in participating in Lung-MAP can contact Lung-MAP protocol coordinator Crystal Miwa at [email protected] or Dr. Herbst at [email protected].
Details on the protocol are also available at clinicaltrials.gov or the Lung-MAP website.
Dr. Herbst reported serving on the science advisory board of Biothera, DiaTech Oncology, Koltan, and N of One.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MADRID – Evaluating one new oncology drug is hard enough, but a novel collaborative clinical trial is taking on the task of testing five experimental drugs at once.
The Lung-MAP trial is using genomic profiling to direct patients to one of five simultaneous sub-studies that best fits their tumor’s genomic profile.
The treatments are for second-line, recurrent squamous cell lung cancer, an area of tremendous unmet need, according to Dr. Roy Herbst, chair of the Lung-MAP steering committee and chief of medical oncology at the Yale Cancer Center, New Haven, Conn.
The novel design of the phase II/III trial should speed up assessment and approval of investigational drugs, and improve the likelihood that patients from the most highly academic to the most rural community sites will receive a targeted therapy.
“It’s really open throughout the country and that makes me very proud that we’re bringing molecular profiling and new drugs to patients of all resource levels,” Dr. Herbst said.
Watch our interview with Dr. Herbst at the annual congress of the European Society for Clinical Oncology, where he spoke about Lung-MAP in a special session on new clinical trial designs.
LungMAP is sponsored by the Southwest Oncology Group with collaboration from several public and private partners including the National Cancer Institute.
Groups interested in participating in Lung-MAP can contact Lung-MAP protocol coordinator Crystal Miwa at [email protected] or Dr. Herbst at [email protected].
Details on the protocol are also available at clinicaltrials.gov or the Lung-MAP website.
Dr. Herbst reported serving on the science advisory board of Biothera, DiaTech Oncology, Koltan, and N of One.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
MADRID – Evaluating one new oncology drug is hard enough, but a novel collaborative clinical trial is taking on the task of testing five experimental drugs at once.
The Lung-MAP trial is using genomic profiling to direct patients to one of five simultaneous sub-studies that best fits their tumor’s genomic profile.
The treatments are for second-line, recurrent squamous cell lung cancer, an area of tremendous unmet need, according to Dr. Roy Herbst, chair of the Lung-MAP steering committee and chief of medical oncology at the Yale Cancer Center, New Haven, Conn.
The novel design of the phase II/III trial should speed up assessment and approval of investigational drugs, and improve the likelihood that patients from the most highly academic to the most rural community sites will receive a targeted therapy.
“It’s really open throughout the country and that makes me very proud that we’re bringing molecular profiling and new drugs to patients of all resource levels,” Dr. Herbst said.
Watch our interview with Dr. Herbst at the annual congress of the European Society for Clinical Oncology, where he spoke about Lung-MAP in a special session on new clinical trial designs.
LungMAP is sponsored by the Southwest Oncology Group with collaboration from several public and private partners including the National Cancer Institute.
Groups interested in participating in Lung-MAP can contact Lung-MAP protocol coordinator Crystal Miwa at [email protected] or Dr. Herbst at [email protected].
Details on the protocol are also available at clinicaltrials.gov or the Lung-MAP website.
Dr. Herbst reported serving on the science advisory board of Biothera, DiaTech Oncology, Koltan, and N of One.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT ESMO 2014
Female sexual dysfunction: Is pharmacologic treatment a future reality or just wishful thinking?
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: Texting initiatives improve patient engagement
SAN DIEGO – Texting can provide a powerful connection with patients and an effective medium to motivate them to adopt healthy behaviors.
Given the pervasiveness of texting, “we know that this is reaching the audience we want to reach,” explained Dr. Colleen Kraft, health network medical director at Cincinnati Children’s Medical Center. “We know that it’s a way to get messages out there, and it’s a way to be able to ascertain feedback, because people can text you back.”
In a video interview at the annual meeting of the American Academy of Pediatrics, Dr. Kraft discusses why texting is so effective at engaging patients, and how texting initiatives could help reshape health management and improve patient outcomes.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN DIEGO – Texting can provide a powerful connection with patients and an effective medium to motivate them to adopt healthy behaviors.
Given the pervasiveness of texting, “we know that this is reaching the audience we want to reach,” explained Dr. Colleen Kraft, health network medical director at Cincinnati Children’s Medical Center. “We know that it’s a way to get messages out there, and it’s a way to be able to ascertain feedback, because people can text you back.”
In a video interview at the annual meeting of the American Academy of Pediatrics, Dr. Kraft discusses why texting is so effective at engaging patients, and how texting initiatives could help reshape health management and improve patient outcomes.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN DIEGO – Texting can provide a powerful connection with patients and an effective medium to motivate them to adopt healthy behaviors.
Given the pervasiveness of texting, “we know that this is reaching the audience we want to reach,” explained Dr. Colleen Kraft, health network medical director at Cincinnati Children’s Medical Center. “We know that it’s a way to get messages out there, and it’s a way to be able to ascertain feedback, because people can text you back.”
In a video interview at the annual meeting of the American Academy of Pediatrics, Dr. Kraft discusses why texting is so effective at engaging patients, and how texting initiatives could help reshape health management and improve patient outcomes.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT THE AAP NATIONAL CONFERENCE