User login
CDC updates guidance on opioid prescribing in adults
The Centers for Disease Control and Prevention (CDC) recently published updated guidelines on prescribing opioids for pain that stress the need for a flexible and individual approach to pain management.1 New recommendations emphasize the use of nonopioid therapies whenever appropriate, support consideration of opioid therapy for patients with acute pain when the benefits are expected to outweigh the risks, and urge clinicians to work with patients receiving opioid therapy to determine whether it should be continued or tapered.
This revision to the agency’s 2016 guidelines is aimed at primary care clinicians who prescribe opioids to adult outpatients for treatment of pain. The recommendations are not meant for patients with sickle-cell disease or cancer-related pain, or those receiving palliative and end-of-life care.
Why an update was needed. In 2021, more than 107,000 Americans died of a drug overdose.2 Although prescription opioids caused only about 16% of these deaths, they account for a population death rate of 4:100,000—which, despite national efforts, has not changed much since 2013.3,4
Following publication of the CDC’s 2016 guidelines on prescribing opioids for chronic pain,5 there was a decline in opioid prescribing but not in related deaths. Furthermore, there appeared to have been some negative effects of reduced prescribing, including untreated and undertreated pain, and rapid tapering or sudden discontinuation of opioids in chronic users, causing withdrawal symptoms and psychological distress in these patients. To address these issues, the CDC published the new guideline in 2022.1
Categories of pain. The guideline panel classified pain into 3 categories: acute pain (duration of < 1 month), subacute pain (duration of 1-3 months), and chronic pain (duration of > 3 months).
When to prescribe opioids. The guidelines recommend a new approach to deciding whether to prescribe opioid therapy. In most cases, nonopioid options—such as nonsteroidal anti-inflammatory drugs (NSAIDs) and exercise—should be tried first, since they are as effective as opioids for many types of acute, subacute, and chronic pain. Opioids should be considered if these options fail and the potential benefits outweigh the risks. In moderate-to-severe acute pain, opioids are an option if NSAIDs are unlikely to be effective or are contraindicated.1
How to prescribe opioids. Before prescribing opioids, clinicians should discuss with the patient the known risks and benefits and offer an accompanying prescription for naloxone. Opioids should be prescribed at the lowest effective dose and for a time period limited to the expected duration of the pain. When starting opioids, immediate-release opioids should be prescribed instead of extended-release or long-acting opioids.1
Precautionary measures. Clinicians should review the patient’s history of controlled substance prescriptions via their state’s prescription drug monitoring program and consider the use of toxicology testing to determine whether the patient is receiving high-risk opioid dosages or combinations. Clinicians should be especially cautious about prescribing opioids and benzodiazepines concurrently.1
Continue or stop opioid treatment? A new recommendation advises clinicians to individually assess the benefits and risks of continuing therapy for patients who have been receiving opioids chronically. Whenever the decision is made to stop or reduce treatment, remember that opioid therapy should not be stopped abruptly or reduced quickly. The guideline panel suggests tapering by 10% per month.1
Finally, patients with opioid use disorder should be offered or referred for treatment with medications. Detoxification alone, without medication, is not recommended.1
1. Dowell D, Ragan KR, Jones CM, et al. CDC clinical practice guideline for prescribing opioids for pain—United States, 2022. MMWR Recomm Rep. 2022;71:1-95. doi: 10.15585/mmwr.rr7103a1
2. CDC. US overdose deaths in 2021 increased half as much as in 2020—but are still up 15%. Published May 11, 2022. Accessed January 25, 2023. www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/202205.htm
3. CDC. SUDORS Dashboard: fatal overdose data. Updated December 8, 2022. Accessed January 25, 2023. www.cdc.gov/drugoverdose/fatal/dashboard/index.html
4. Mattson CL, Tanz LJ, Quinn K, et al. Trends and geographic patterns in drug and synthetic opioid overdose deaths—United States, 2013–2019. MMWR Morb Mortal Wkly Rep. 2021;70:202-207. doi: 10.15585/mmwr.mm7006a4
5. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49. doi: 10.15585/mmwr.rr6501e1:26987082
The Centers for Disease Control and Prevention (CDC) recently published updated guidelines on prescribing opioids for pain that stress the need for a flexible and individual approach to pain management.1 New recommendations emphasize the use of nonopioid therapies whenever appropriate, support consideration of opioid therapy for patients with acute pain when the benefits are expected to outweigh the risks, and urge clinicians to work with patients receiving opioid therapy to determine whether it should be continued or tapered.
This revision to the agency’s 2016 guidelines is aimed at primary care clinicians who prescribe opioids to adult outpatients for treatment of pain. The recommendations are not meant for patients with sickle-cell disease or cancer-related pain, or those receiving palliative and end-of-life care.
Why an update was needed. In 2021, more than 107,000 Americans died of a drug overdose.2 Although prescription opioids caused only about 16% of these deaths, they account for a population death rate of 4:100,000—which, despite national efforts, has not changed much since 2013.3,4
Following publication of the CDC’s 2016 guidelines on prescribing opioids for chronic pain,5 there was a decline in opioid prescribing but not in related deaths. Furthermore, there appeared to have been some negative effects of reduced prescribing, including untreated and undertreated pain, and rapid tapering or sudden discontinuation of opioids in chronic users, causing withdrawal symptoms and psychological distress in these patients. To address these issues, the CDC published the new guideline in 2022.1
Categories of pain. The guideline panel classified pain into 3 categories: acute pain (duration of < 1 month), subacute pain (duration of 1-3 months), and chronic pain (duration of > 3 months).
When to prescribe opioids. The guidelines recommend a new approach to deciding whether to prescribe opioid therapy. In most cases, nonopioid options—such as nonsteroidal anti-inflammatory drugs (NSAIDs) and exercise—should be tried first, since they are as effective as opioids for many types of acute, subacute, and chronic pain. Opioids should be considered if these options fail and the potential benefits outweigh the risks. In moderate-to-severe acute pain, opioids are an option if NSAIDs are unlikely to be effective or are contraindicated.1
How to prescribe opioids. Before prescribing opioids, clinicians should discuss with the patient the known risks and benefits and offer an accompanying prescription for naloxone. Opioids should be prescribed at the lowest effective dose and for a time period limited to the expected duration of the pain. When starting opioids, immediate-release opioids should be prescribed instead of extended-release or long-acting opioids.1
Precautionary measures. Clinicians should review the patient’s history of controlled substance prescriptions via their state’s prescription drug monitoring program and consider the use of toxicology testing to determine whether the patient is receiving high-risk opioid dosages or combinations. Clinicians should be especially cautious about prescribing opioids and benzodiazepines concurrently.1
Continue or stop opioid treatment? A new recommendation advises clinicians to individually assess the benefits and risks of continuing therapy for patients who have been receiving opioids chronically. Whenever the decision is made to stop or reduce treatment, remember that opioid therapy should not be stopped abruptly or reduced quickly. The guideline panel suggests tapering by 10% per month.1
Finally, patients with opioid use disorder should be offered or referred for treatment with medications. Detoxification alone, without medication, is not recommended.1
The Centers for Disease Control and Prevention (CDC) recently published updated guidelines on prescribing opioids for pain that stress the need for a flexible and individual approach to pain management.1 New recommendations emphasize the use of nonopioid therapies whenever appropriate, support consideration of opioid therapy for patients with acute pain when the benefits are expected to outweigh the risks, and urge clinicians to work with patients receiving opioid therapy to determine whether it should be continued or tapered.
This revision to the agency’s 2016 guidelines is aimed at primary care clinicians who prescribe opioids to adult outpatients for treatment of pain. The recommendations are not meant for patients with sickle-cell disease or cancer-related pain, or those receiving palliative and end-of-life care.
Why an update was needed. In 2021, more than 107,000 Americans died of a drug overdose.2 Although prescription opioids caused only about 16% of these deaths, they account for a population death rate of 4:100,000—which, despite national efforts, has not changed much since 2013.3,4
Following publication of the CDC’s 2016 guidelines on prescribing opioids for chronic pain,5 there was a decline in opioid prescribing but not in related deaths. Furthermore, there appeared to have been some negative effects of reduced prescribing, including untreated and undertreated pain, and rapid tapering or sudden discontinuation of opioids in chronic users, causing withdrawal symptoms and psychological distress in these patients. To address these issues, the CDC published the new guideline in 2022.1
Categories of pain. The guideline panel classified pain into 3 categories: acute pain (duration of < 1 month), subacute pain (duration of 1-3 months), and chronic pain (duration of > 3 months).
When to prescribe opioids. The guidelines recommend a new approach to deciding whether to prescribe opioid therapy. In most cases, nonopioid options—such as nonsteroidal anti-inflammatory drugs (NSAIDs) and exercise—should be tried first, since they are as effective as opioids for many types of acute, subacute, and chronic pain. Opioids should be considered if these options fail and the potential benefits outweigh the risks. In moderate-to-severe acute pain, opioids are an option if NSAIDs are unlikely to be effective or are contraindicated.1
How to prescribe opioids. Before prescribing opioids, clinicians should discuss with the patient the known risks and benefits and offer an accompanying prescription for naloxone. Opioids should be prescribed at the lowest effective dose and for a time period limited to the expected duration of the pain. When starting opioids, immediate-release opioids should be prescribed instead of extended-release or long-acting opioids.1
Precautionary measures. Clinicians should review the patient’s history of controlled substance prescriptions via their state’s prescription drug monitoring program and consider the use of toxicology testing to determine whether the patient is receiving high-risk opioid dosages or combinations. Clinicians should be especially cautious about prescribing opioids and benzodiazepines concurrently.1
Continue or stop opioid treatment? A new recommendation advises clinicians to individually assess the benefits and risks of continuing therapy for patients who have been receiving opioids chronically. Whenever the decision is made to stop or reduce treatment, remember that opioid therapy should not be stopped abruptly or reduced quickly. The guideline panel suggests tapering by 10% per month.1
Finally, patients with opioid use disorder should be offered or referred for treatment with medications. Detoxification alone, without medication, is not recommended.1
1. Dowell D, Ragan KR, Jones CM, et al. CDC clinical practice guideline for prescribing opioids for pain—United States, 2022. MMWR Recomm Rep. 2022;71:1-95. doi: 10.15585/mmwr.rr7103a1
2. CDC. US overdose deaths in 2021 increased half as much as in 2020—but are still up 15%. Published May 11, 2022. Accessed January 25, 2023. www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/202205.htm
3. CDC. SUDORS Dashboard: fatal overdose data. Updated December 8, 2022. Accessed January 25, 2023. www.cdc.gov/drugoverdose/fatal/dashboard/index.html
4. Mattson CL, Tanz LJ, Quinn K, et al. Trends and geographic patterns in drug and synthetic opioid overdose deaths—United States, 2013–2019. MMWR Morb Mortal Wkly Rep. 2021;70:202-207. doi: 10.15585/mmwr.mm7006a4
5. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49. doi: 10.15585/mmwr.rr6501e1:26987082
1. Dowell D, Ragan KR, Jones CM, et al. CDC clinical practice guideline for prescribing opioids for pain—United States, 2022. MMWR Recomm Rep. 2022;71:1-95. doi: 10.15585/mmwr.rr7103a1
2. CDC. US overdose deaths in 2021 increased half as much as in 2020—but are still up 15%. Published May 11, 2022. Accessed January 25, 2023. www.cdc.gov/nchs/pressroom/nchs_press_releases/2022/202205.htm
3. CDC. SUDORS Dashboard: fatal overdose data. Updated December 8, 2022. Accessed January 25, 2023. www.cdc.gov/drugoverdose/fatal/dashboard/index.html
4. Mattson CL, Tanz LJ, Quinn K, et al. Trends and geographic patterns in drug and synthetic opioid overdose deaths—United States, 2013–2019. MMWR Morb Mortal Wkly Rep. 2021;70:202-207. doi: 10.15585/mmwr.mm7006a4
5. Dowell D, Haegerich TM, Chou R. CDC guideline for prescribing opioids for chronic pain—United States, 2016. MMWR Recomm Rep. 2016;65:1-49. doi: 10.15585/mmwr.rr6501e1:26987082
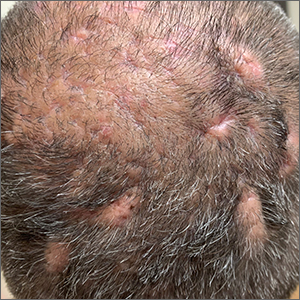
Scalp ridges

The gyrate or cerebriform pattern of inflammatory, often pus-filled, subcutaneous tracts of the scalp pointed to a diagnosis of dissecting cellulitis. This patient did not have the fluctuant tracts frequently seen in more active disease but did have the scarring and alopecia common with this disorder.
Dissecting cellulitis is similar to acne and hidradenitis suppurativa in that it starts with follicular plugging. This plugging leads to inflammation, dilation and rupture of the follicle, and purulent sinus tract formation. The sinus tracts of the scalp can be extensive. Dissecting cellulitis is most common in 18- to 40-year-olds and more common in Black individuals.1 When it occurs in conjunction with cystic acne and hidradenitis suppurativa, it is known as the follicular occlusion triad syndrome.
While oral antibiotics are an option for the treatment of dissecting cellulitis, oral isotretinoin is the first-line approach. Tumor necrosis factor alfa inhibitors have also been used with success, according to case reports.1
Given that this patient had a small area of current inflammation, he was started on oral doxycycline 100 mg twice daily for 2 months. He was scheduled for a follow-up appointment in 3 months to reassess his progress and to explore treatment with isotretinoin if the condition worsened or did not improve.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Federico A, Rossi A, Caro G, et al. Are dissecting cellulitis and hidradenitis suppurativa different diseases? Clin Dermatol. 2021;39:496-499. doi: 10.1016/j.clindermatol.2021.01.002

The gyrate or cerebriform pattern of inflammatory, often pus-filled, subcutaneous tracts of the scalp pointed to a diagnosis of dissecting cellulitis. This patient did not have the fluctuant tracts frequently seen in more active disease but did have the scarring and alopecia common with this disorder.
Dissecting cellulitis is similar to acne and hidradenitis suppurativa in that it starts with follicular plugging. This plugging leads to inflammation, dilation and rupture of the follicle, and purulent sinus tract formation. The sinus tracts of the scalp can be extensive. Dissecting cellulitis is most common in 18- to 40-year-olds and more common in Black individuals.1 When it occurs in conjunction with cystic acne and hidradenitis suppurativa, it is known as the follicular occlusion triad syndrome.
While oral antibiotics are an option for the treatment of dissecting cellulitis, oral isotretinoin is the first-line approach. Tumor necrosis factor alfa inhibitors have also been used with success, according to case reports.1
Given that this patient had a small area of current inflammation, he was started on oral doxycycline 100 mg twice daily for 2 months. He was scheduled for a follow-up appointment in 3 months to reassess his progress and to explore treatment with isotretinoin if the condition worsened or did not improve.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

The gyrate or cerebriform pattern of inflammatory, often pus-filled, subcutaneous tracts of the scalp pointed to a diagnosis of dissecting cellulitis. This patient did not have the fluctuant tracts frequently seen in more active disease but did have the scarring and alopecia common with this disorder.
Dissecting cellulitis is similar to acne and hidradenitis suppurativa in that it starts with follicular plugging. This plugging leads to inflammation, dilation and rupture of the follicle, and purulent sinus tract formation. The sinus tracts of the scalp can be extensive. Dissecting cellulitis is most common in 18- to 40-year-olds and more common in Black individuals.1 When it occurs in conjunction with cystic acne and hidradenitis suppurativa, it is known as the follicular occlusion triad syndrome.
While oral antibiotics are an option for the treatment of dissecting cellulitis, oral isotretinoin is the first-line approach. Tumor necrosis factor alfa inhibitors have also been used with success, according to case reports.1
Given that this patient had a small area of current inflammation, he was started on oral doxycycline 100 mg twice daily for 2 months. He was scheduled for a follow-up appointment in 3 months to reassess his progress and to explore treatment with isotretinoin if the condition worsened or did not improve.
Photo and text courtesy of Daniel Stulberg, MD, FAAFP, Professor and Chair, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Federico A, Rossi A, Caro G, et al. Are dissecting cellulitis and hidradenitis suppurativa different diseases? Clin Dermatol. 2021;39:496-499. doi: 10.1016/j.clindermatol.2021.01.002
1. Federico A, Rossi A, Caro G, et al. Are dissecting cellulitis and hidradenitis suppurativa different diseases? Clin Dermatol. 2021;39:496-499. doi: 10.1016/j.clindermatol.2021.01.002
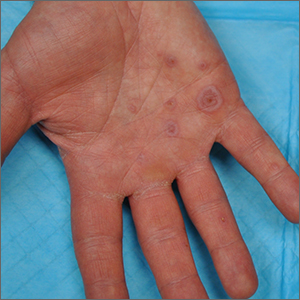
Palmar rash
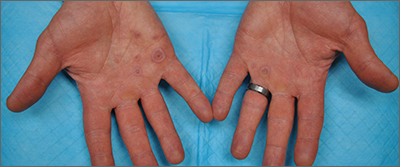
This patient’s targetoid and tingling skin lesions, in association with herpes simplex virus (HSV) infection, are a classic presentation of erythema multiforme (EM).
EM is an acute, self-limited, immune-mediated process that most commonly arises in a symmetrical pattern on acral surfaces. These lesions may be accompanied by eruptions on oral, anogenital, or ocular mucosa. EM is classified into 2 subtypes: major and minor. EM major refers to EM with significant mucosal involvement on at least 2 mucosal sites; it may also manifest with a prodrome of fevers, arthralgias, and malaise. EM minor is used to classify EM with minimal mucosal involvement.1
The term “multiforme” denotes the varied dermatologic changes, including macules, papules, and targetoid lesions with 3 identifiable zones, which are pathognomonic for EM. The classic 3 zones consist of an inner dusky, vesicular, or necrotic center; a middle elevated edematous surrounding ring; and an outer ring of macular erythema. Patients may also present with an atypical macular target lesion, characterized by fewer than 3 zones with an ill-defined border between the zones. The lesions may be asymptomatic, or patients may describe an itchy or burning sensation.
The differential diagnosis of EM includes urticaria, fixed drug eruption, subacute lupus erythematosus, Kawasaki disease, erythema annulare centrifugum, vasculitis, and Stevens-Johnson syndrome.
Infections with HSV types 1 or 2 are the leading cause of EM and are thought to involve a cell-mediated immune process directed against viral antigens in skin.2 Other infectious causes include cytomegalovirus, Epstein-Barr virus, influenza virus, and—rarely—newer strains of coronavirus.3 Pharmacologic reactions are the cause in a small percentage of patients, and may involve nonsteroidal anti-inflammatory drugs, antibiotics, sulfonamides, antiepileptics, and tumor necrosis factor-alpha inhibitors. Studies also link the development of EM to primary malignancy, autoimmune disease, and immunizations.1
The treatment of EM is dependent on the clinical course and severity of the disease. If a causative agent is identified, it should be discontinued (if a drug) or treated (if an infection). Topical antiseptic mouthwashes, antihistamines, and topical corticosteroids can be used to relieve cutaneous discomfort. Biologics and immunosuppressants can be used with patients who have severe symptoms or functional impairment. Patients who have recurrences associated with HSV should be given antiviral prophylaxis for 6 months consisting of oral acyclovir 10 mg/kg/d, valacyclovir 500 to 1000 mg/d, or famciclovir 250 mg twice daily.1
Given the recurrent nature of this patient’s disease, and its association with HSV outbreaks, he was prescribed prophylactic valacyclovir 1000 mg/d orally for 6 months to reduce HSV outbreaks and hopefully prevent future EM episodes.
Photo courtesy of Cyrelle F. Finan, MD. Text courtesy of Lynn Midani, BS, University of New Mexico School of Medicine, and Cyrelle F. Finan, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Trayes KP, Love G, Studdiford JS. Erythema multiforme: recognition and management. Am Fam Physician. 2019;100:82-88.
2. Hafsi W, Badri T. Erythema multiforme. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022. Updated August 1, 2022. Accessed December 15, 2022. www.ncbi.nlm.nih.gov/books/NBK470259/
3. Bennardo L, Nisticò SP, Dastoli S, et al. Erythema multiforme and COVID-19: what do we know? Medicina. 2021;57:828. https://doi.org/10.3390/medicina57080828

This patient’s targetoid and tingling skin lesions, in association with herpes simplex virus (HSV) infection, are a classic presentation of erythema multiforme (EM).
EM is an acute, self-limited, immune-mediated process that most commonly arises in a symmetrical pattern on acral surfaces. These lesions may be accompanied by eruptions on oral, anogenital, or ocular mucosa. EM is classified into 2 subtypes: major and minor. EM major refers to EM with significant mucosal involvement on at least 2 mucosal sites; it may also manifest with a prodrome of fevers, arthralgias, and malaise. EM minor is used to classify EM with minimal mucosal involvement.1
The term “multiforme” denotes the varied dermatologic changes, including macules, papules, and targetoid lesions with 3 identifiable zones, which are pathognomonic for EM. The classic 3 zones consist of an inner dusky, vesicular, or necrotic center; a middle elevated edematous surrounding ring; and an outer ring of macular erythema. Patients may also present with an atypical macular target lesion, characterized by fewer than 3 zones with an ill-defined border between the zones. The lesions may be asymptomatic, or patients may describe an itchy or burning sensation.
The differential diagnosis of EM includes urticaria, fixed drug eruption, subacute lupus erythematosus, Kawasaki disease, erythema annulare centrifugum, vasculitis, and Stevens-Johnson syndrome.
Infections with HSV types 1 or 2 are the leading cause of EM and are thought to involve a cell-mediated immune process directed against viral antigens in skin.2 Other infectious causes include cytomegalovirus, Epstein-Barr virus, influenza virus, and—rarely—newer strains of coronavirus.3 Pharmacologic reactions are the cause in a small percentage of patients, and may involve nonsteroidal anti-inflammatory drugs, antibiotics, sulfonamides, antiepileptics, and tumor necrosis factor-alpha inhibitors. Studies also link the development of EM to primary malignancy, autoimmune disease, and immunizations.1
The treatment of EM is dependent on the clinical course and severity of the disease. If a causative agent is identified, it should be discontinued (if a drug) or treated (if an infection). Topical antiseptic mouthwashes, antihistamines, and topical corticosteroids can be used to relieve cutaneous discomfort. Biologics and immunosuppressants can be used with patients who have severe symptoms or functional impairment. Patients who have recurrences associated with HSV should be given antiviral prophylaxis for 6 months consisting of oral acyclovir 10 mg/kg/d, valacyclovir 500 to 1000 mg/d, or famciclovir 250 mg twice daily.1
Given the recurrent nature of this patient’s disease, and its association with HSV outbreaks, he was prescribed prophylactic valacyclovir 1000 mg/d orally for 6 months to reduce HSV outbreaks and hopefully prevent future EM episodes.
Photo courtesy of Cyrelle F. Finan, MD. Text courtesy of Lynn Midani, BS, University of New Mexico School of Medicine, and Cyrelle F. Finan, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.

This patient’s targetoid and tingling skin lesions, in association with herpes simplex virus (HSV) infection, are a classic presentation of erythema multiforme (EM).
EM is an acute, self-limited, immune-mediated process that most commonly arises in a symmetrical pattern on acral surfaces. These lesions may be accompanied by eruptions on oral, anogenital, or ocular mucosa. EM is classified into 2 subtypes: major and minor. EM major refers to EM with significant mucosal involvement on at least 2 mucosal sites; it may also manifest with a prodrome of fevers, arthralgias, and malaise. EM minor is used to classify EM with minimal mucosal involvement.1
The term “multiforme” denotes the varied dermatologic changes, including macules, papules, and targetoid lesions with 3 identifiable zones, which are pathognomonic for EM. The classic 3 zones consist of an inner dusky, vesicular, or necrotic center; a middle elevated edematous surrounding ring; and an outer ring of macular erythema. Patients may also present with an atypical macular target lesion, characterized by fewer than 3 zones with an ill-defined border between the zones. The lesions may be asymptomatic, or patients may describe an itchy or burning sensation.
The differential diagnosis of EM includes urticaria, fixed drug eruption, subacute lupus erythematosus, Kawasaki disease, erythema annulare centrifugum, vasculitis, and Stevens-Johnson syndrome.
Infections with HSV types 1 or 2 are the leading cause of EM and are thought to involve a cell-mediated immune process directed against viral antigens in skin.2 Other infectious causes include cytomegalovirus, Epstein-Barr virus, influenza virus, and—rarely—newer strains of coronavirus.3 Pharmacologic reactions are the cause in a small percentage of patients, and may involve nonsteroidal anti-inflammatory drugs, antibiotics, sulfonamides, antiepileptics, and tumor necrosis factor-alpha inhibitors. Studies also link the development of EM to primary malignancy, autoimmune disease, and immunizations.1
The treatment of EM is dependent on the clinical course and severity of the disease. If a causative agent is identified, it should be discontinued (if a drug) or treated (if an infection). Topical antiseptic mouthwashes, antihistamines, and topical corticosteroids can be used to relieve cutaneous discomfort. Biologics and immunosuppressants can be used with patients who have severe symptoms or functional impairment. Patients who have recurrences associated with HSV should be given antiviral prophylaxis for 6 months consisting of oral acyclovir 10 mg/kg/d, valacyclovir 500 to 1000 mg/d, or famciclovir 250 mg twice daily.1
Given the recurrent nature of this patient’s disease, and its association with HSV outbreaks, he was prescribed prophylactic valacyclovir 1000 mg/d orally for 6 months to reduce HSV outbreaks and hopefully prevent future EM episodes.
Photo courtesy of Cyrelle F. Finan, MD. Text courtesy of Lynn Midani, BS, University of New Mexico School of Medicine, and Cyrelle F. Finan, MD, Department of Dermatology, and Daniel Stulberg, MD, FAAFP, Department of Family and Community Medicine, Western Michigan University Homer Stryker, MD School of Medicine, Kalamazoo.
1. Trayes KP, Love G, Studdiford JS. Erythema multiforme: recognition and management. Am Fam Physician. 2019;100:82-88.
2. Hafsi W, Badri T. Erythema multiforme. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022. Updated August 1, 2022. Accessed December 15, 2022. www.ncbi.nlm.nih.gov/books/NBK470259/
3. Bennardo L, Nisticò SP, Dastoli S, et al. Erythema multiforme and COVID-19: what do we know? Medicina. 2021;57:828. https://doi.org/10.3390/medicina57080828
1. Trayes KP, Love G, Studdiford JS. Erythema multiforme: recognition and management. Am Fam Physician. 2019;100:82-88.
2. Hafsi W, Badri T. Erythema multiforme. StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2022. Updated August 1, 2022. Accessed December 15, 2022. www.ncbi.nlm.nih.gov/books/NBK470259/
3. Bennardo L, Nisticò SP, Dastoli S, et al. Erythema multiforme and COVID-19: what do we know? Medicina. 2021;57:828. https://doi.org/10.3390/medicina57080828
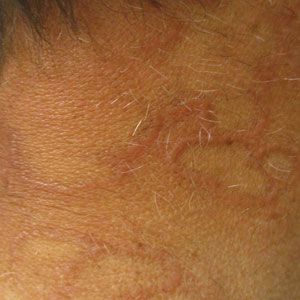
Annular Plaques Overlying Hyperpigmented Telangiectatic Patches on the Neck
The Diagnosis: Annular Elastolytic Giant Cell Granuloma
Histologic examination of the shave biopsies showed a granulomatous infiltrate of small lymphocytes, histiocytes, and multinucleated giant cells. The giant cells have abundant eosinophilic cytoplasm, with several also containing fragments of basophilic elastic fibers (elastophagocytosis)(Figure). Additionally, the granulomas revealed no signs of necrosis, making an infectious source unlikely, and examination under polarized light was negative for foreign material. These clinical and histologic findings were diagnostic for annular elastolytic giant cell granuloma (AEGCG).
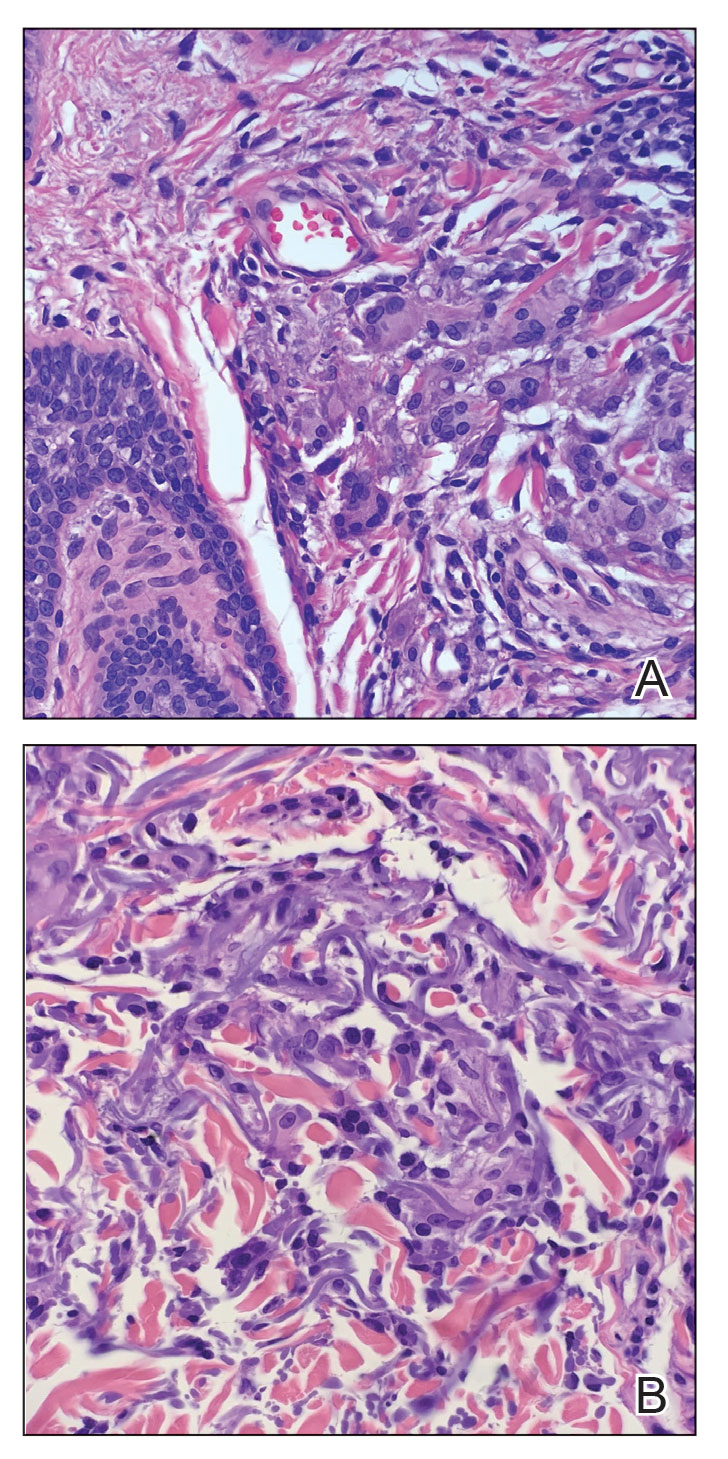
Annular elastolytic giant cell granuloma is a rare chronic inflammatory disorder that classically presents on sun-exposed skin as annular plaques with elevated borders and atrophic centers.1-4 Histologically, AEGCG is characterized by diffuse granulomatous infiltrates composed of multinucleated giant cells, histiocytes, and lymphocytes in the dermis, along with phagocytosis of elastic fibers by multinucleated giant cells.5 The underlying etiology and pathogenesis of AEGCG remains unknown.6
Annular elastolytic giant cell granuloma commonly affects females aged 35 to 75 years; however, cases have been reported in the male and pediatric patient populations.1,2 Documented cases are known to last from 1 month to 10 years.7,8 Although the mechanisms underlying the development of AEGCG remain to be elucidated, studies have determined that the skin disorder is associated with sarcoidosis, molluscum contagiosum, amyloidosis, diabetes mellitus, and cutaneous T-cell lymphoma.9 Diabetes mellitus is the most common comorbidity associated with AEGCG, and it is theorized that diabetes contributes to the increased incidence of AEGCG in this population by inducing damage to elastic fibers in the skin.10 One study that examined 50 cases of AEGCG found that 38 patients had serum glucose levels evaluated, with 8 cases being subsequently diagnosed with diabetes mellitus and 6 cases with apparent impaired glucose tolerance, indicating that 37% of the sample population with AEGCG who were evaluated for metabolic disease were found to have definitive or latent type 2 diabetes mellitus.11 Although AEGCG is a rare disorder, a substantial number of patients diagnosed with AEGCG also have diabetes mellitus, making it important to consider screening all patients with AEGCG for diabetes given the ease and widely available resources to check glucose levels.
Actinic granuloma, granuloma annulare, atypical facial necrobiosis lipoidica, granuloma multiforme, secondary syphilis, tinea corporis, and erythema annulare centrifugum most commonly are included in the differential diagnosis with AEGCG; histopathology is the key determinant in discerning between these conditions.12 Our patient presented with typical annular plaques overlying hyperpigmented telangiectatic patches. With known type 2 diabetes mellitus and the clinical findings, granuloma annulare, erythema annulare centrifugum, and AEGCG remained high on the differential.
No standard of care exists for AEGCG due to its rare nature and tendency to spontaneously resolve. The most common first-line treatment includes topical and intralesional steroids, topical pimecrolimus, and the use of sunscreen and other sun-protective measures. UV radiation, specifically UVA, has been determined to be a causal factor for AEGCG by changing the antigenicity of elastic fibers and producing an immune response in individuals with fair skin.13 Further, resistant cases of AEGCG successfully have been treated with cyclosporine, systemic steroids, antimalarials, dapsone, and oral retinoids.14,15 Some studies reported partial regression or full resolution with topical tretinoin; adalimumab; clobetasol ointment; or a combination of corticosteroids, antihistamines, and hydroxychloroquine.2 Only 1 case series using sulfasalazine reported worsening symptoms after treatment initiation.16 Our patient deferred systemic medications and was treated with 4 weeks of topical triamcinolone followed by 4 weeks of topical tacrolimus with minimal improvement. At the time of diagnosis, our patient also was encouraged to use sun-protective measures. At 6-month follow-up, the lesions remained stable, and the decision was made to continue with photoprotection.
- Mistry AM, Patel R, Mistry M, et al. Annular elastolytic giant cell granuloma. Cureus. 2020;12:E11456. doi:10.7759/cureus.11456
- Chen WT, Hsiao PF, Wu YH. Spectrum and clinical variants of giant cell elastolytic granuloma. Int J Dermatol. 2017;56:738-745. doi:10.1111/ijd.13502
- Raposo I, Mota F, Lobo I, et al. Annular elastolytic giant cell granuloma: a “visible” diagnosis. Dermatol Online J. 2017;23:13030/qt9rq3j927
- Klemke CD, Siebold D, Dippel E, et al. Generalised annular elastolytic giant cell granuloma. Dermatology. 2003;207:420-422. doi:10.1159/000074132
- Hassan R, Arunprasath P, Padmavathy L, et al. Annular elastolytic giant cell granuloma in association with Hashimoto’s thyroiditis. Indian Dermatol Online J. 2016;7:107-110. doi:10.4103/2229-5178.178087
- Kaya Erdog˘ an H, Arık D, Acer E, et al. Clinicopathological features of annular elastolytic giant cell granuloma patients. Turkish J Dermatol. 2018;12:85-89.
- Can B, Kavala M, Türkog˘ lu Z, et al. Successful treatment of annular elastolytic giant cell granuloma with hydroxychloroquine. Int J Dermatol. 2013;52:509-511. doi:10.1111 /j.1365-4632.2011.04941.x
- Arora S, Malik A, Patil C, et al. Annular elastolytic giant cell granuloma: a report of 10 cases. Indian Dermatol Online J. 2015;6(suppl 1):S17-S20. doi:10.4103/2229-5178.171055
- Doulaveri G, Tsagroni E, Giannadaki M, et al. Annular elastolytic giant cell granuloma in a 70-year-old woman. Int J Dermatol. 2003;42:290-291. doi:10.1046/j.1365-4362.2003.01767.x
- Marmon S, O’Reilly KE, Fischer M, et al. Papular variant of annular elastolytic giant-cell granuloma. Dermatol Online J. 2012;18:23.
- Aso Y, Izaki S, Teraki Y. Annular elastolytic giant cell granuloma associated with diabetes mellitus: a case report and review of the Japanese literature. Clin Exp Dermatol. 2011;36:917-919. doi:10.1111 /j.1365-2230.2011.04094.x
- Liu X, Zhang W, Liu Y, et al. A case of annular elastolytic giant cell granuloma associated with syphilis. Case Rep Dermatol. 2018; 10:158-161. doi:10.1159/000489910
- Gutiérrez-González E, Pereiro M Jr, Toribio J. Elastolytic actinic giant cell granuloma. Dermatol Clin. 2015;33:331-341. doi:10.1016/j.det.2015.03.002
- de Oliveira FL, de Barros Silveira LK, Machado Ade M, et al. Hybrid clinical and histopathological pattern in annular lesions: an overlap between annular elastolytic giant cell granuloma and granuloma annulare? Case Rep Dermatol Med. 2012;2012:102915. doi:10.1155/2012/102915
- Wagenseller A, Larocca C, Vashi NA. Treatment of annular elastolytic giant cell granuloma with topical tretinoin. J Drugs Dermatol. 2017;16:699-700.
- Yang YW, Lehrer MD, Mangold AR, et al. Treatment of granuloma annulare and related granulomatous diseases with sulphasalazine: a series of 16 cases. J Eur Acad Dermatol Venereol. 2021;35:211-215. doi:10.1111/jdv.16356
The Diagnosis: Annular Elastolytic Giant Cell Granuloma
Histologic examination of the shave biopsies showed a granulomatous infiltrate of small lymphocytes, histiocytes, and multinucleated giant cells. The giant cells have abundant eosinophilic cytoplasm, with several also containing fragments of basophilic elastic fibers (elastophagocytosis)(Figure). Additionally, the granulomas revealed no signs of necrosis, making an infectious source unlikely, and examination under polarized light was negative for foreign material. These clinical and histologic findings were diagnostic for annular elastolytic giant cell granuloma (AEGCG).

Annular elastolytic giant cell granuloma is a rare chronic inflammatory disorder that classically presents on sun-exposed skin as annular plaques with elevated borders and atrophic centers.1-4 Histologically, AEGCG is characterized by diffuse granulomatous infiltrates composed of multinucleated giant cells, histiocytes, and lymphocytes in the dermis, along with phagocytosis of elastic fibers by multinucleated giant cells.5 The underlying etiology and pathogenesis of AEGCG remains unknown.6
Annular elastolytic giant cell granuloma commonly affects females aged 35 to 75 years; however, cases have been reported in the male and pediatric patient populations.1,2 Documented cases are known to last from 1 month to 10 years.7,8 Although the mechanisms underlying the development of AEGCG remain to be elucidated, studies have determined that the skin disorder is associated with sarcoidosis, molluscum contagiosum, amyloidosis, diabetes mellitus, and cutaneous T-cell lymphoma.9 Diabetes mellitus is the most common comorbidity associated with AEGCG, and it is theorized that diabetes contributes to the increased incidence of AEGCG in this population by inducing damage to elastic fibers in the skin.10 One study that examined 50 cases of AEGCG found that 38 patients had serum glucose levels evaluated, with 8 cases being subsequently diagnosed with diabetes mellitus and 6 cases with apparent impaired glucose tolerance, indicating that 37% of the sample population with AEGCG who were evaluated for metabolic disease were found to have definitive or latent type 2 diabetes mellitus.11 Although AEGCG is a rare disorder, a substantial number of patients diagnosed with AEGCG also have diabetes mellitus, making it important to consider screening all patients with AEGCG for diabetes given the ease and widely available resources to check glucose levels.
Actinic granuloma, granuloma annulare, atypical facial necrobiosis lipoidica, granuloma multiforme, secondary syphilis, tinea corporis, and erythema annulare centrifugum most commonly are included in the differential diagnosis with AEGCG; histopathology is the key determinant in discerning between these conditions.12 Our patient presented with typical annular plaques overlying hyperpigmented telangiectatic patches. With known type 2 diabetes mellitus and the clinical findings, granuloma annulare, erythema annulare centrifugum, and AEGCG remained high on the differential.
No standard of care exists for AEGCG due to its rare nature and tendency to spontaneously resolve. The most common first-line treatment includes topical and intralesional steroids, topical pimecrolimus, and the use of sunscreen and other sun-protective measures. UV radiation, specifically UVA, has been determined to be a causal factor for AEGCG by changing the antigenicity of elastic fibers and producing an immune response in individuals with fair skin.13 Further, resistant cases of AEGCG successfully have been treated with cyclosporine, systemic steroids, antimalarials, dapsone, and oral retinoids.14,15 Some studies reported partial regression or full resolution with topical tretinoin; adalimumab; clobetasol ointment; or a combination of corticosteroids, antihistamines, and hydroxychloroquine.2 Only 1 case series using sulfasalazine reported worsening symptoms after treatment initiation.16 Our patient deferred systemic medications and was treated with 4 weeks of topical triamcinolone followed by 4 weeks of topical tacrolimus with minimal improvement. At the time of diagnosis, our patient also was encouraged to use sun-protective measures. At 6-month follow-up, the lesions remained stable, and the decision was made to continue with photoprotection.
The Diagnosis: Annular Elastolytic Giant Cell Granuloma
Histologic examination of the shave biopsies showed a granulomatous infiltrate of small lymphocytes, histiocytes, and multinucleated giant cells. The giant cells have abundant eosinophilic cytoplasm, with several also containing fragments of basophilic elastic fibers (elastophagocytosis)(Figure). Additionally, the granulomas revealed no signs of necrosis, making an infectious source unlikely, and examination under polarized light was negative for foreign material. These clinical and histologic findings were diagnostic for annular elastolytic giant cell granuloma (AEGCG).

Annular elastolytic giant cell granuloma is a rare chronic inflammatory disorder that classically presents on sun-exposed skin as annular plaques with elevated borders and atrophic centers.1-4 Histologically, AEGCG is characterized by diffuse granulomatous infiltrates composed of multinucleated giant cells, histiocytes, and lymphocytes in the dermis, along with phagocytosis of elastic fibers by multinucleated giant cells.5 The underlying etiology and pathogenesis of AEGCG remains unknown.6
Annular elastolytic giant cell granuloma commonly affects females aged 35 to 75 years; however, cases have been reported in the male and pediatric patient populations.1,2 Documented cases are known to last from 1 month to 10 years.7,8 Although the mechanisms underlying the development of AEGCG remain to be elucidated, studies have determined that the skin disorder is associated with sarcoidosis, molluscum contagiosum, amyloidosis, diabetes mellitus, and cutaneous T-cell lymphoma.9 Diabetes mellitus is the most common comorbidity associated with AEGCG, and it is theorized that diabetes contributes to the increased incidence of AEGCG in this population by inducing damage to elastic fibers in the skin.10 One study that examined 50 cases of AEGCG found that 38 patients had serum glucose levels evaluated, with 8 cases being subsequently diagnosed with diabetes mellitus and 6 cases with apparent impaired glucose tolerance, indicating that 37% of the sample population with AEGCG who were evaluated for metabolic disease were found to have definitive or latent type 2 diabetes mellitus.11 Although AEGCG is a rare disorder, a substantial number of patients diagnosed with AEGCG also have diabetes mellitus, making it important to consider screening all patients with AEGCG for diabetes given the ease and widely available resources to check glucose levels.
Actinic granuloma, granuloma annulare, atypical facial necrobiosis lipoidica, granuloma multiforme, secondary syphilis, tinea corporis, and erythema annulare centrifugum most commonly are included in the differential diagnosis with AEGCG; histopathology is the key determinant in discerning between these conditions.12 Our patient presented with typical annular plaques overlying hyperpigmented telangiectatic patches. With known type 2 diabetes mellitus and the clinical findings, granuloma annulare, erythema annulare centrifugum, and AEGCG remained high on the differential.
No standard of care exists for AEGCG due to its rare nature and tendency to spontaneously resolve. The most common first-line treatment includes topical and intralesional steroids, topical pimecrolimus, and the use of sunscreen and other sun-protective measures. UV radiation, specifically UVA, has been determined to be a causal factor for AEGCG by changing the antigenicity of elastic fibers and producing an immune response in individuals with fair skin.13 Further, resistant cases of AEGCG successfully have been treated with cyclosporine, systemic steroids, antimalarials, dapsone, and oral retinoids.14,15 Some studies reported partial regression or full resolution with topical tretinoin; adalimumab; clobetasol ointment; or a combination of corticosteroids, antihistamines, and hydroxychloroquine.2 Only 1 case series using sulfasalazine reported worsening symptoms after treatment initiation.16 Our patient deferred systemic medications and was treated with 4 weeks of topical triamcinolone followed by 4 weeks of topical tacrolimus with minimal improvement. At the time of diagnosis, our patient also was encouraged to use sun-protective measures. At 6-month follow-up, the lesions remained stable, and the decision was made to continue with photoprotection.
- Mistry AM, Patel R, Mistry M, et al. Annular elastolytic giant cell granuloma. Cureus. 2020;12:E11456. doi:10.7759/cureus.11456
- Chen WT, Hsiao PF, Wu YH. Spectrum and clinical variants of giant cell elastolytic granuloma. Int J Dermatol. 2017;56:738-745. doi:10.1111/ijd.13502
- Raposo I, Mota F, Lobo I, et al. Annular elastolytic giant cell granuloma: a “visible” diagnosis. Dermatol Online J. 2017;23:13030/qt9rq3j927
- Klemke CD, Siebold D, Dippel E, et al. Generalised annular elastolytic giant cell granuloma. Dermatology. 2003;207:420-422. doi:10.1159/000074132
- Hassan R, Arunprasath P, Padmavathy L, et al. Annular elastolytic giant cell granuloma in association with Hashimoto’s thyroiditis. Indian Dermatol Online J. 2016;7:107-110. doi:10.4103/2229-5178.178087
- Kaya Erdog˘ an H, Arık D, Acer E, et al. Clinicopathological features of annular elastolytic giant cell granuloma patients. Turkish J Dermatol. 2018;12:85-89.
- Can B, Kavala M, Türkog˘ lu Z, et al. Successful treatment of annular elastolytic giant cell granuloma with hydroxychloroquine. Int J Dermatol. 2013;52:509-511. doi:10.1111 /j.1365-4632.2011.04941.x
- Arora S, Malik A, Patil C, et al. Annular elastolytic giant cell granuloma: a report of 10 cases. Indian Dermatol Online J. 2015;6(suppl 1):S17-S20. doi:10.4103/2229-5178.171055
- Doulaveri G, Tsagroni E, Giannadaki M, et al. Annular elastolytic giant cell granuloma in a 70-year-old woman. Int J Dermatol. 2003;42:290-291. doi:10.1046/j.1365-4362.2003.01767.x
- Marmon S, O’Reilly KE, Fischer M, et al. Papular variant of annular elastolytic giant-cell granuloma. Dermatol Online J. 2012;18:23.
- Aso Y, Izaki S, Teraki Y. Annular elastolytic giant cell granuloma associated with diabetes mellitus: a case report and review of the Japanese literature. Clin Exp Dermatol. 2011;36:917-919. doi:10.1111 /j.1365-2230.2011.04094.x
- Liu X, Zhang W, Liu Y, et al. A case of annular elastolytic giant cell granuloma associated with syphilis. Case Rep Dermatol. 2018; 10:158-161. doi:10.1159/000489910
- Gutiérrez-González E, Pereiro M Jr, Toribio J. Elastolytic actinic giant cell granuloma. Dermatol Clin. 2015;33:331-341. doi:10.1016/j.det.2015.03.002
- de Oliveira FL, de Barros Silveira LK, Machado Ade M, et al. Hybrid clinical and histopathological pattern in annular lesions: an overlap between annular elastolytic giant cell granuloma and granuloma annulare? Case Rep Dermatol Med. 2012;2012:102915. doi:10.1155/2012/102915
- Wagenseller A, Larocca C, Vashi NA. Treatment of annular elastolytic giant cell granuloma with topical tretinoin. J Drugs Dermatol. 2017;16:699-700.
- Yang YW, Lehrer MD, Mangold AR, et al. Treatment of granuloma annulare and related granulomatous diseases with sulphasalazine: a series of 16 cases. J Eur Acad Dermatol Venereol. 2021;35:211-215. doi:10.1111/jdv.16356
- Mistry AM, Patel R, Mistry M, et al. Annular elastolytic giant cell granuloma. Cureus. 2020;12:E11456. doi:10.7759/cureus.11456
- Chen WT, Hsiao PF, Wu YH. Spectrum and clinical variants of giant cell elastolytic granuloma. Int J Dermatol. 2017;56:738-745. doi:10.1111/ijd.13502
- Raposo I, Mota F, Lobo I, et al. Annular elastolytic giant cell granuloma: a “visible” diagnosis. Dermatol Online J. 2017;23:13030/qt9rq3j927
- Klemke CD, Siebold D, Dippel E, et al. Generalised annular elastolytic giant cell granuloma. Dermatology. 2003;207:420-422. doi:10.1159/000074132
- Hassan R, Arunprasath P, Padmavathy L, et al. Annular elastolytic giant cell granuloma in association with Hashimoto’s thyroiditis. Indian Dermatol Online J. 2016;7:107-110. doi:10.4103/2229-5178.178087
- Kaya Erdog˘ an H, Arık D, Acer E, et al. Clinicopathological features of annular elastolytic giant cell granuloma patients. Turkish J Dermatol. 2018;12:85-89.
- Can B, Kavala M, Türkog˘ lu Z, et al. Successful treatment of annular elastolytic giant cell granuloma with hydroxychloroquine. Int J Dermatol. 2013;52:509-511. doi:10.1111 /j.1365-4632.2011.04941.x
- Arora S, Malik A, Patil C, et al. Annular elastolytic giant cell granuloma: a report of 10 cases. Indian Dermatol Online J. 2015;6(suppl 1):S17-S20. doi:10.4103/2229-5178.171055
- Doulaveri G, Tsagroni E, Giannadaki M, et al. Annular elastolytic giant cell granuloma in a 70-year-old woman. Int J Dermatol. 2003;42:290-291. doi:10.1046/j.1365-4362.2003.01767.x
- Marmon S, O’Reilly KE, Fischer M, et al. Papular variant of annular elastolytic giant-cell granuloma. Dermatol Online J. 2012;18:23.
- Aso Y, Izaki S, Teraki Y. Annular elastolytic giant cell granuloma associated with diabetes mellitus: a case report and review of the Japanese literature. Clin Exp Dermatol. 2011;36:917-919. doi:10.1111 /j.1365-2230.2011.04094.x
- Liu X, Zhang W, Liu Y, et al. A case of annular elastolytic giant cell granuloma associated with syphilis. Case Rep Dermatol. 2018; 10:158-161. doi:10.1159/000489910
- Gutiérrez-González E, Pereiro M Jr, Toribio J. Elastolytic actinic giant cell granuloma. Dermatol Clin. 2015;33:331-341. doi:10.1016/j.det.2015.03.002
- de Oliveira FL, de Barros Silveira LK, Machado Ade M, et al. Hybrid clinical and histopathological pattern in annular lesions: an overlap between annular elastolytic giant cell granuloma and granuloma annulare? Case Rep Dermatol Med. 2012;2012:102915. doi:10.1155/2012/102915
- Wagenseller A, Larocca C, Vashi NA. Treatment of annular elastolytic giant cell granuloma with topical tretinoin. J Drugs Dermatol. 2017;16:699-700.
- Yang YW, Lehrer MD, Mangold AR, et al. Treatment of granuloma annulare and related granulomatous diseases with sulphasalazine: a series of 16 cases. J Eur Acad Dermatol Venereol. 2021;35:211-215. doi:10.1111/jdv.16356
A 58-year-old man with a history of type 2 diabetes mellitus, nephrolithiasis, hypovitaminosis D, and hypercholesterolemia presented to our dermatology clinic for a follow-up total-body skin examination after a prior diagnosis of basal cell carcinoma on the vertex of the scalp. Physical examination revealed extensive photodamage and annular plaques overlying hyperpigmented telangiectatic patches on the dorsal portion of the neck. The eruption persisted for 1 year and failed to improve with clotrimazole cream. His medications included simvastatin, metformin, chlorthalidone, vitamin D, and tamsulosin. Two shave biopsies from the posterior neck were performed.

Commentary: Early Diagnosis of PsA, February 2023
Appropriate assessment of MSK symptoms and signs by dermatologists may lead to more appropriate referral to rheumatologists. MSUS is being increasingly explored for early identification of PsA. A handheld, chip-based ultrasound device (HHUD) is a novel promising instrument that can be easily implemented in clinical practice. In a prospective study including 140 patients with psoriasis who presented to dermatologists with arthralgia. Grobelski and colleagues screened for PsA using medical history, CE, and the German Psoriasis Arthritis Diagnostic PsA screening questionnaire (GEPARD) paired with MSUS examination of up to three painful joints by trained dermatologists. Nineteen patients (13.6%) were diagnosed with PsA by rheumatologists. Interestingly, in 45 of the 46 patients the preliminary diagnosis of PsA was revised to "no PsA" after MSUS. The addition of MSUS changed the sensitivity and specificity of early PsA screening strategy from 88.2% and 54.4% to 70.6% and 90.4%, respectively. The positive predictive value increased to 56.5% from 25.4% after MSUS. Thus, the use of a quick MSUS using HHUD may lead to more accurate referral to rheumatologists. The challenge is seamless integration of MSUS into busy dermatology practices.
The goal of PsA treatment is to achieve a state of remission or low disease activity. Criteria for minimal disease activity (MDA) have been established. Achieving MDA leads to better health-related quality of life (HRQOL), as well as less joint damage. In a prospective cohort study that included 240 patients with newly diagnosed disease-modifying antirheumatic drug-naive PsA, Snoeck Henkemans and colleagues demonstrate that failure to achieve MDA in the first year after the diagnosis of PsA was associated with worse HRQOL and health status, functional impairment, fatigue, pain, and higher anxiety and depression. Compared with patients who achieved sustained MDA in the first year after diagnosis, those who did not achieve MDA had higher scores for pain, fatigue, and functional ability and higher anxiety and depression during follow-up, which persisted despite treatment intensification. Thus, implementation of treat-to-target strategies with the aim of achieving sustained MDA within 1 year of diagnosis is likely to have better long-term benefits in this lifelong disease.
Another study emphasized the need for early treatment to improve long-term outcomes. In a post hoc analysis of two phase 3 trials including 1554 patients with PsA who received 300-mg or 150-mg secukinumab with or without a loading dose, Mease and colleagues showed that high baseline radiographic damage reduced the likelihood of achieving MDA.
Overall, these studies indicate that early diagnosis and treatment prior to developing joint damage with the aim to achieve sustained MDA within a year will lead to better long-term outcome for patients with PsA.
Appropriate assessment of MSK symptoms and signs by dermatologists may lead to more appropriate referral to rheumatologists. MSUS is being increasingly explored for early identification of PsA. A handheld, chip-based ultrasound device (HHUD) is a novel promising instrument that can be easily implemented in clinical practice. In a prospective study including 140 patients with psoriasis who presented to dermatologists with arthralgia. Grobelski and colleagues screened for PsA using medical history, CE, and the German Psoriasis Arthritis Diagnostic PsA screening questionnaire (GEPARD) paired with MSUS examination of up to three painful joints by trained dermatologists. Nineteen patients (13.6%) were diagnosed with PsA by rheumatologists. Interestingly, in 45 of the 46 patients the preliminary diagnosis of PsA was revised to "no PsA" after MSUS. The addition of MSUS changed the sensitivity and specificity of early PsA screening strategy from 88.2% and 54.4% to 70.6% and 90.4%, respectively. The positive predictive value increased to 56.5% from 25.4% after MSUS. Thus, the use of a quick MSUS using HHUD may lead to more accurate referral to rheumatologists. The challenge is seamless integration of MSUS into busy dermatology practices.
The goal of PsA treatment is to achieve a state of remission or low disease activity. Criteria for minimal disease activity (MDA) have been established. Achieving MDA leads to better health-related quality of life (HRQOL), as well as less joint damage. In a prospective cohort study that included 240 patients with newly diagnosed disease-modifying antirheumatic drug-naive PsA, Snoeck Henkemans and colleagues demonstrate that failure to achieve MDA in the first year after the diagnosis of PsA was associated with worse HRQOL and health status, functional impairment, fatigue, pain, and higher anxiety and depression. Compared with patients who achieved sustained MDA in the first year after diagnosis, those who did not achieve MDA had higher scores for pain, fatigue, and functional ability and higher anxiety and depression during follow-up, which persisted despite treatment intensification. Thus, implementation of treat-to-target strategies with the aim of achieving sustained MDA within 1 year of diagnosis is likely to have better long-term benefits in this lifelong disease.
Another study emphasized the need for early treatment to improve long-term outcomes. In a post hoc analysis of two phase 3 trials including 1554 patients with PsA who received 300-mg or 150-mg secukinumab with or without a loading dose, Mease and colleagues showed that high baseline radiographic damage reduced the likelihood of achieving MDA.
Overall, these studies indicate that early diagnosis and treatment prior to developing joint damage with the aim to achieve sustained MDA within a year will lead to better long-term outcome for patients with PsA.
Appropriate assessment of MSK symptoms and signs by dermatologists may lead to more appropriate referral to rheumatologists. MSUS is being increasingly explored for early identification of PsA. A handheld, chip-based ultrasound device (HHUD) is a novel promising instrument that can be easily implemented in clinical practice. In a prospective study including 140 patients with psoriasis who presented to dermatologists with arthralgia. Grobelski and colleagues screened for PsA using medical history, CE, and the German Psoriasis Arthritis Diagnostic PsA screening questionnaire (GEPARD) paired with MSUS examination of up to three painful joints by trained dermatologists. Nineteen patients (13.6%) were diagnosed with PsA by rheumatologists. Interestingly, in 45 of the 46 patients the preliminary diagnosis of PsA was revised to "no PsA" after MSUS. The addition of MSUS changed the sensitivity and specificity of early PsA screening strategy from 88.2% and 54.4% to 70.6% and 90.4%, respectively. The positive predictive value increased to 56.5% from 25.4% after MSUS. Thus, the use of a quick MSUS using HHUD may lead to more accurate referral to rheumatologists. The challenge is seamless integration of MSUS into busy dermatology practices.
The goal of PsA treatment is to achieve a state of remission or low disease activity. Criteria for minimal disease activity (MDA) have been established. Achieving MDA leads to better health-related quality of life (HRQOL), as well as less joint damage. In a prospective cohort study that included 240 patients with newly diagnosed disease-modifying antirheumatic drug-naive PsA, Snoeck Henkemans and colleagues demonstrate that failure to achieve MDA in the first year after the diagnosis of PsA was associated with worse HRQOL and health status, functional impairment, fatigue, pain, and higher anxiety and depression. Compared with patients who achieved sustained MDA in the first year after diagnosis, those who did not achieve MDA had higher scores for pain, fatigue, and functional ability and higher anxiety and depression during follow-up, which persisted despite treatment intensification. Thus, implementation of treat-to-target strategies with the aim of achieving sustained MDA within 1 year of diagnosis is likely to have better long-term benefits in this lifelong disease.
Another study emphasized the need for early treatment to improve long-term outcomes. In a post hoc analysis of two phase 3 trials including 1554 patients with PsA who received 300-mg or 150-mg secukinumab with or without a loading dose, Mease and colleagues showed that high baseline radiographic damage reduced the likelihood of achieving MDA.
Overall, these studies indicate that early diagnosis and treatment prior to developing joint damage with the aim to achieve sustained MDA within a year will lead to better long-term outcome for patients with PsA.
14-year-old boy • aching midsternal pain following a basketball injury • worsening pain with direct pressure and when the patient sneezed • Dx?
THE CASE
A 14-year-old boy sought care at our clinic for persistent chest pain after being hit in the chest with a teammate’s shoulder during a basketball game 3 weeks earlier. He had aching midsternal chest pain that worsened with direct pressure and when he sneezed, twisted, or bent forward. There was no bruising or swelling.
On examination, the patient demonstrated normal perfusion and normal work of breathing. He had focal tenderness with palpation at the manubrium with no noticeable step-off, and mild tenderness at the adjacent costochondral junctions and over his pectoral muscles. His sternal pain along the proximal sternum was reproducible with a weighted wall push-up. Although the patient maintained full range of motion in his upper extremities, he did have sternal pain with flexion, abduction, and external rotation of the bilateral upper extremities against resistance. Anteroposterior (AP) and lateral chest radiographs were unremarkable.
THE DIAGNOSIS
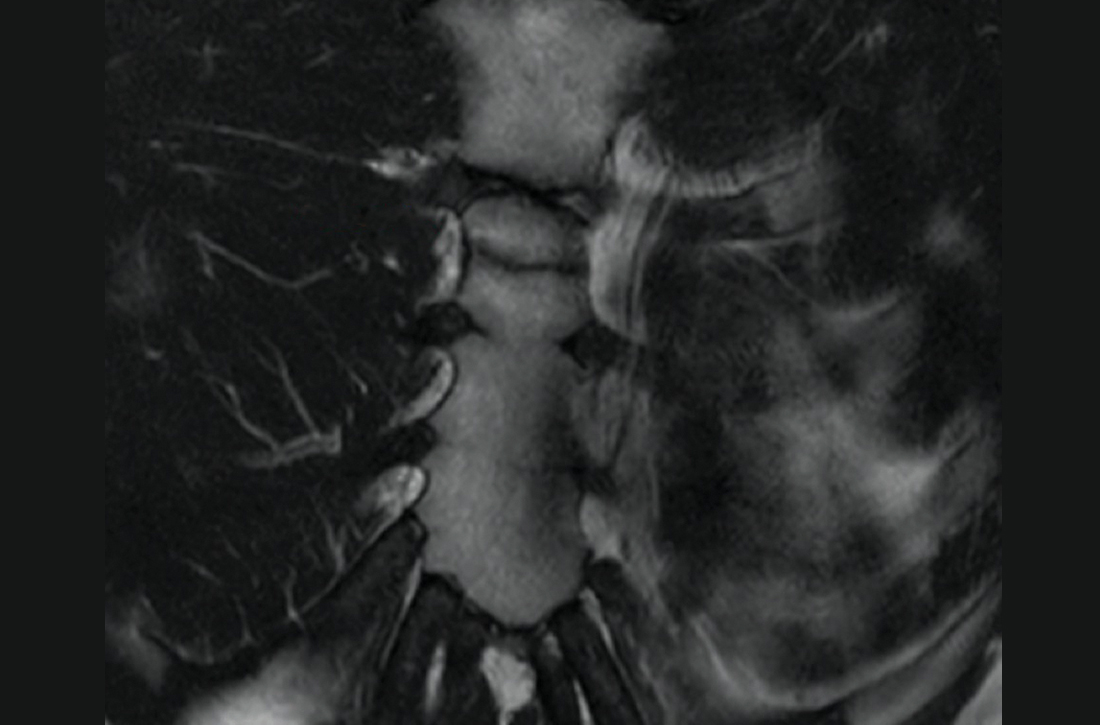
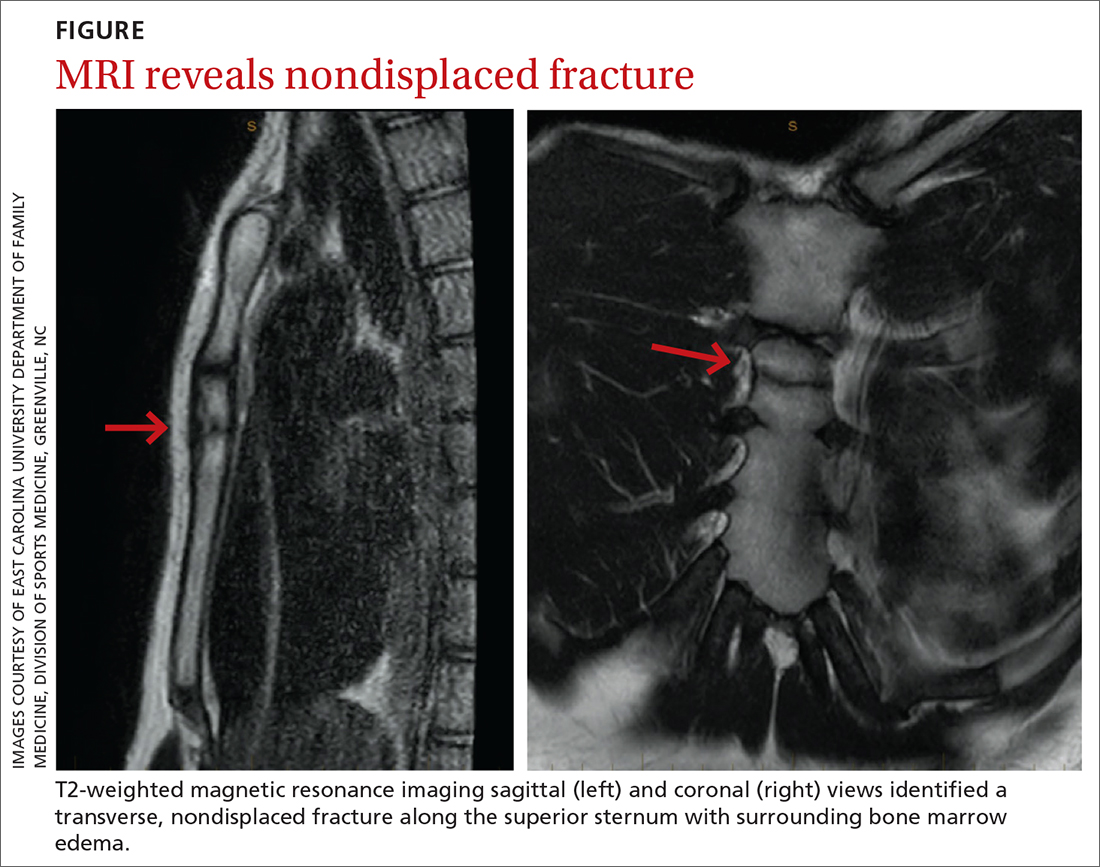
The unremarkable chest radiographs prompted further investigation with a diagnostic ultrasound, which revealed a small cortical defect with overlying anechoic fluid collection in the area of focal tenderness. T2-weighted magnetic resonance imaging (MRI) of the chest was performed; it revealed a transverse, nondisplaced fracture of the superior body of the sternum with surrounding bone marrow edema (FIGURE).
DISCUSSION
Fractures of the sternum comprise < 1% of traumatic fractures and have a low mortality rate (0.7%).1,2 The rarity of these fractures is attributed to the ribs’ elastic recoil, which protects the chest wall from anterior forces.1,3 These fractures are even more unusual in children due to the increased elasticity of their chest walls.4-6 Thus, it takes a significant amount of force for a child’s sternum to fracture.
While isolated sternum fractures can occur, two-thirds of sternum fractures are nonisolated and are associated with injuries to surrounding structures (including the heart, lungs, and vasculature) or fractures of the ribs and spine.2,3 Most often, these injuries are caused by significant blunt trauma to the anterior chest, rapid deceleration, or flexion-compression injury.2,3 They are typically transverse and localized, with 70% of fractures occurring in the mid-body and 17.6% at the manubriosternal joint.1,3,6
Athletes with a sternal fracture typically present as our patient did, with a history of blunt force trauma to the chest and with pain and tenderness over the anterior midline of the chest that increases with respiration or movement.1 A physical examination that includes chest palpation and auscultation of the heart and lungs must be performed to rule out damage to intrathoracic structures and assess the patient’s cardiac and pulmonary stability. An electrocardiogram should be performed to confirm that there are no cardiovascular complications.3,4
Initial imaging should include AP and lateral chest radiographs because any displacement will occur in the sagittal plane.1,2,4-6 If the radiograph shows no clear pathology, follow up with computed tomography, ultrasound, MRI, or technetium bone scans to gain additional information.1 Diagnosis of sternal fractures is especially difficult in children due to the presence of ossification centers for bone growth, which may be misinterpreted as a sternal fracture in the absence of a proper understanding of sternal development.5,6 On ultrasound, sternal fractures appear as a sharp step-off in the cortex, whereas in the absence of fracture, there is no cortical step-off and the cartilaginous plate between ossification centers appears in line with the cortex.7
Continue to: A self-limiting injury that requires proper pain control
A self-limiting injury that requires proper pain control
Isolated sternal fractures are typically self-limiting with a good prognosis.2 These injuries are managed supportively with rest, ice, and analgesics1; proper pain control is crucial to prevent respiratory compromise.8
Complete recovery for most patients occurs in 10 to 12 weeks.9 Recovery periods longer than 12 weeks are associated with nonisolated sternal fractures that are complicated by soft-tissue injury, injuries to the chest wall (such as sternoclavicular joint dislocation, usually from a fall on the shoulder), or fracture nonunion.1,2,5
Anterior sternoclavicular joint dislocations and stable posterior dislocations are managed with closed reduction and immobilization in a figure-of-eight brace.1 Operative management is reserved for patients with displaced fractures, sternal deformity, chest wall instability, respiratory insufficiency, uncontrolled pain, or fracture nonunion.1,3,8
A return-to-play protocol can begin once the patient is asymptomatic.1 The timeframe for a full return to play can vary from 6 weeks to 6 months, depending on the severity of the fracture.1 This process is guided by how quickly the symptoms resolve and by radiographic stability.9
Our patient was followed every 3 to 4 weeks and started physical therapy 6 weeks after his injury occurred. He was held from play for 10 weeks and gradually returned to play; he returned to full-contact activity after tolerating a practice without pain.
THE TAKEAWAY
Children typically have greater chest wall elasticity, and thus, it is unusual for them to sustain a sternal fracture. Diagnosis in children is complicated by the presence of ossification centers for bone growth on imaging. In this case, the fracture was first noticed on ultrasound and confirmed with MRI. Since these fractures can be associated with damage to surrounding structures, additional injuries should be considered when evaluating a patient with a sternum fracture.
CORRESPONDENCE
Catherine Romaine, East Carolina University, Brody School of Medicine, 600 Moye Boulevard, Greenville, NC 27834; [email protected]
1. Alent J, Narducci DM, Moran B, et al. Sternal injuries in sport: a review of the literature. Sports Med. 2018;48:2715-2724. doi: 10.1007/s40279-018-0990-5
2. Khoriati A-A, Rajakulasingam R, Shah R. Sternal fractures and their management. J Emerg Trauma Shock. 2013;6:113-116. doi: 10.4103/0974-2700.110763
3. Athanassiadi K, Gerazounis M, Moustardas M, et al. Sternal fractures: retrospective analysis of 100 cases. World J Surg. 2002;26:1243-1246. doi: 10.1007/s00268-002-6511-5
4. Ferguson LP, Wilkinson AG, Beattie TF. Fracture of the sternum in children. Emerg Med J. 2003;20:518-520. doi: 10.1136/emj.20.6.518
5. Ramgopal S, Shaffiey SA, Conti KA. Pediatric sternal fractures from a Level 1 trauma center. J Pediatr Surg. 2019;54:1628-1631. doi: 10.1016/j.jpedsurg.2018.08.040
6. Sesia SB, Prüfer F, Mayr J. Sternal fracture in children: diagnosis by ultrasonography. European J Pediatr Surg Rep. 2017;5:e39-e42. doi: 10.1055/s-0037-1606197
7. Nickson C, Rippey J. Ultrasonography of sternal fractures. Australas J Ultrasound Med. 2011;14:6-11. doi: 10.1002/j.2205-0140.2011.tb00131.x
8. Bauman ZM, Yanala U, Waibel BH, et al. Sternal fixation for isolated traumatic sternal fractures improves pain and upper extremity range of motion. Eur J Trauma Emerg Surg. 2022;48:225-230. doi: 10.1007/s00068-020-01568-x
9. Culp B, Hurbanek JG, Novak J, et al. Acute traumatic sternum fracture in a female college hockey player. Orthopedics. 2010;33:683. doi: 10.3928/01477447-20100722-17
THE CASE
A 14-year-old boy sought care at our clinic for persistent chest pain after being hit in the chest with a teammate’s shoulder during a basketball game 3 weeks earlier. He had aching midsternal chest pain that worsened with direct pressure and when he sneezed, twisted, or bent forward. There was no bruising or swelling.
On examination, the patient demonstrated normal perfusion and normal work of breathing. He had focal tenderness with palpation at the manubrium with no noticeable step-off, and mild tenderness at the adjacent costochondral junctions and over his pectoral muscles. His sternal pain along the proximal sternum was reproducible with a weighted wall push-up. Although the patient maintained full range of motion in his upper extremities, he did have sternal pain with flexion, abduction, and external rotation of the bilateral upper extremities against resistance. Anteroposterior (AP) and lateral chest radiographs were unremarkable.
THE DIAGNOSIS
The unremarkable chest radiographs prompted further investigation with a diagnostic ultrasound, which revealed a small cortical defect with overlying anechoic fluid collection in the area of focal tenderness. T2-weighted magnetic resonance imaging (MRI) of the chest was performed; it revealed a transverse, nondisplaced fracture of the superior body of the sternum with surrounding bone marrow edema (FIGURE).

DISCUSSION
Fractures of the sternum comprise < 1% of traumatic fractures and have a low mortality rate (0.7%).1,2 The rarity of these fractures is attributed to the ribs’ elastic recoil, which protects the chest wall from anterior forces.1,3 These fractures are even more unusual in children due to the increased elasticity of their chest walls.4-6 Thus, it takes a significant amount of force for a child’s sternum to fracture.
While isolated sternum fractures can occur, two-thirds of sternum fractures are nonisolated and are associated with injuries to surrounding structures (including the heart, lungs, and vasculature) or fractures of the ribs and spine.2,3 Most often, these injuries are caused by significant blunt trauma to the anterior chest, rapid deceleration, or flexion-compression injury.2,3 They are typically transverse and localized, with 70% of fractures occurring in the mid-body and 17.6% at the manubriosternal joint.1,3,6
Athletes with a sternal fracture typically present as our patient did, with a history of blunt force trauma to the chest and with pain and tenderness over the anterior midline of the chest that increases with respiration or movement.1 A physical examination that includes chest palpation and auscultation of the heart and lungs must be performed to rule out damage to intrathoracic structures and assess the patient’s cardiac and pulmonary stability. An electrocardiogram should be performed to confirm that there are no cardiovascular complications.3,4
Initial imaging should include AP and lateral chest radiographs because any displacement will occur in the sagittal plane.1,2,4-6 If the radiograph shows no clear pathology, follow up with computed tomography, ultrasound, MRI, or technetium bone scans to gain additional information.1 Diagnosis of sternal fractures is especially difficult in children due to the presence of ossification centers for bone growth, which may be misinterpreted as a sternal fracture in the absence of a proper understanding of sternal development.5,6 On ultrasound, sternal fractures appear as a sharp step-off in the cortex, whereas in the absence of fracture, there is no cortical step-off and the cartilaginous plate between ossification centers appears in line with the cortex.7
Continue to: A self-limiting injury that requires proper pain control
A self-limiting injury that requires proper pain control
Isolated sternal fractures are typically self-limiting with a good prognosis.2 These injuries are managed supportively with rest, ice, and analgesics1; proper pain control is crucial to prevent respiratory compromise.8
Complete recovery for most patients occurs in 10 to 12 weeks.9 Recovery periods longer than 12 weeks are associated with nonisolated sternal fractures that are complicated by soft-tissue injury, injuries to the chest wall (such as sternoclavicular joint dislocation, usually from a fall on the shoulder), or fracture nonunion.1,2,5
Anterior sternoclavicular joint dislocations and stable posterior dislocations are managed with closed reduction and immobilization in a figure-of-eight brace.1 Operative management is reserved for patients with displaced fractures, sternal deformity, chest wall instability, respiratory insufficiency, uncontrolled pain, or fracture nonunion.1,3,8
A return-to-play protocol can begin once the patient is asymptomatic.1 The timeframe for a full return to play can vary from 6 weeks to 6 months, depending on the severity of the fracture.1 This process is guided by how quickly the symptoms resolve and by radiographic stability.9
Our patient was followed every 3 to 4 weeks and started physical therapy 6 weeks after his injury occurred. He was held from play for 10 weeks and gradually returned to play; he returned to full-contact activity after tolerating a practice without pain.
THE TAKEAWAY
Children typically have greater chest wall elasticity, and thus, it is unusual for them to sustain a sternal fracture. Diagnosis in children is complicated by the presence of ossification centers for bone growth on imaging. In this case, the fracture was first noticed on ultrasound and confirmed with MRI. Since these fractures can be associated with damage to surrounding structures, additional injuries should be considered when evaluating a patient with a sternum fracture.
CORRESPONDENCE
Catherine Romaine, East Carolina University, Brody School of Medicine, 600 Moye Boulevard, Greenville, NC 27834; [email protected]
THE CASE
A 14-year-old boy sought care at our clinic for persistent chest pain after being hit in the chest with a teammate’s shoulder during a basketball game 3 weeks earlier. He had aching midsternal chest pain that worsened with direct pressure and when he sneezed, twisted, or bent forward. There was no bruising or swelling.
On examination, the patient demonstrated normal perfusion and normal work of breathing. He had focal tenderness with palpation at the manubrium with no noticeable step-off, and mild tenderness at the adjacent costochondral junctions and over his pectoral muscles. His sternal pain along the proximal sternum was reproducible with a weighted wall push-up. Although the patient maintained full range of motion in his upper extremities, he did have sternal pain with flexion, abduction, and external rotation of the bilateral upper extremities against resistance. Anteroposterior (AP) and lateral chest radiographs were unremarkable.
THE DIAGNOSIS
The unremarkable chest radiographs prompted further investigation with a diagnostic ultrasound, which revealed a small cortical defect with overlying anechoic fluid collection in the area of focal tenderness. T2-weighted magnetic resonance imaging (MRI) of the chest was performed; it revealed a transverse, nondisplaced fracture of the superior body of the sternum with surrounding bone marrow edema (FIGURE).

DISCUSSION
Fractures of the sternum comprise < 1% of traumatic fractures and have a low mortality rate (0.7%).1,2 The rarity of these fractures is attributed to the ribs’ elastic recoil, which protects the chest wall from anterior forces.1,3 These fractures are even more unusual in children due to the increased elasticity of their chest walls.4-6 Thus, it takes a significant amount of force for a child’s sternum to fracture.
While isolated sternum fractures can occur, two-thirds of sternum fractures are nonisolated and are associated with injuries to surrounding structures (including the heart, lungs, and vasculature) or fractures of the ribs and spine.2,3 Most often, these injuries are caused by significant blunt trauma to the anterior chest, rapid deceleration, or flexion-compression injury.2,3 They are typically transverse and localized, with 70% of fractures occurring in the mid-body and 17.6% at the manubriosternal joint.1,3,6
Athletes with a sternal fracture typically present as our patient did, with a history of blunt force trauma to the chest and with pain and tenderness over the anterior midline of the chest that increases with respiration or movement.1 A physical examination that includes chest palpation and auscultation of the heart and lungs must be performed to rule out damage to intrathoracic structures and assess the patient’s cardiac and pulmonary stability. An electrocardiogram should be performed to confirm that there are no cardiovascular complications.3,4
Initial imaging should include AP and lateral chest radiographs because any displacement will occur in the sagittal plane.1,2,4-6 If the radiograph shows no clear pathology, follow up with computed tomography, ultrasound, MRI, or technetium bone scans to gain additional information.1 Diagnosis of sternal fractures is especially difficult in children due to the presence of ossification centers for bone growth, which may be misinterpreted as a sternal fracture in the absence of a proper understanding of sternal development.5,6 On ultrasound, sternal fractures appear as a sharp step-off in the cortex, whereas in the absence of fracture, there is no cortical step-off and the cartilaginous plate between ossification centers appears in line with the cortex.7
Continue to: A self-limiting injury that requires proper pain control
A self-limiting injury that requires proper pain control
Isolated sternal fractures are typically self-limiting with a good prognosis.2 These injuries are managed supportively with rest, ice, and analgesics1; proper pain control is crucial to prevent respiratory compromise.8
Complete recovery for most patients occurs in 10 to 12 weeks.9 Recovery periods longer than 12 weeks are associated with nonisolated sternal fractures that are complicated by soft-tissue injury, injuries to the chest wall (such as sternoclavicular joint dislocation, usually from a fall on the shoulder), or fracture nonunion.1,2,5
Anterior sternoclavicular joint dislocations and stable posterior dislocations are managed with closed reduction and immobilization in a figure-of-eight brace.1 Operative management is reserved for patients with displaced fractures, sternal deformity, chest wall instability, respiratory insufficiency, uncontrolled pain, or fracture nonunion.1,3,8
A return-to-play protocol can begin once the patient is asymptomatic.1 The timeframe for a full return to play can vary from 6 weeks to 6 months, depending on the severity of the fracture.1 This process is guided by how quickly the symptoms resolve and by radiographic stability.9
Our patient was followed every 3 to 4 weeks and started physical therapy 6 weeks after his injury occurred. He was held from play for 10 weeks and gradually returned to play; he returned to full-contact activity after tolerating a practice without pain.
THE TAKEAWAY
Children typically have greater chest wall elasticity, and thus, it is unusual for them to sustain a sternal fracture. Diagnosis in children is complicated by the presence of ossification centers for bone growth on imaging. In this case, the fracture was first noticed on ultrasound and confirmed with MRI. Since these fractures can be associated with damage to surrounding structures, additional injuries should be considered when evaluating a patient with a sternum fracture.
CORRESPONDENCE
Catherine Romaine, East Carolina University, Brody School of Medicine, 600 Moye Boulevard, Greenville, NC 27834; [email protected]
1. Alent J, Narducci DM, Moran B, et al. Sternal injuries in sport: a review of the literature. Sports Med. 2018;48:2715-2724. doi: 10.1007/s40279-018-0990-5
2. Khoriati A-A, Rajakulasingam R, Shah R. Sternal fractures and their management. J Emerg Trauma Shock. 2013;6:113-116. doi: 10.4103/0974-2700.110763
3. Athanassiadi K, Gerazounis M, Moustardas M, et al. Sternal fractures: retrospective analysis of 100 cases. World J Surg. 2002;26:1243-1246. doi: 10.1007/s00268-002-6511-5
4. Ferguson LP, Wilkinson AG, Beattie TF. Fracture of the sternum in children. Emerg Med J. 2003;20:518-520. doi: 10.1136/emj.20.6.518
5. Ramgopal S, Shaffiey SA, Conti KA. Pediatric sternal fractures from a Level 1 trauma center. J Pediatr Surg. 2019;54:1628-1631. doi: 10.1016/j.jpedsurg.2018.08.040
6. Sesia SB, Prüfer F, Mayr J. Sternal fracture in children: diagnosis by ultrasonography. European J Pediatr Surg Rep. 2017;5:e39-e42. doi: 10.1055/s-0037-1606197
7. Nickson C, Rippey J. Ultrasonography of sternal fractures. Australas J Ultrasound Med. 2011;14:6-11. doi: 10.1002/j.2205-0140.2011.tb00131.x
8. Bauman ZM, Yanala U, Waibel BH, et al. Sternal fixation for isolated traumatic sternal fractures improves pain and upper extremity range of motion. Eur J Trauma Emerg Surg. 2022;48:225-230. doi: 10.1007/s00068-020-01568-x
9. Culp B, Hurbanek JG, Novak J, et al. Acute traumatic sternum fracture in a female college hockey player. Orthopedics. 2010;33:683. doi: 10.3928/01477447-20100722-17
1. Alent J, Narducci DM, Moran B, et al. Sternal injuries in sport: a review of the literature. Sports Med. 2018;48:2715-2724. doi: 10.1007/s40279-018-0990-5
2. Khoriati A-A, Rajakulasingam R, Shah R. Sternal fractures and their management. J Emerg Trauma Shock. 2013;6:113-116. doi: 10.4103/0974-2700.110763
3. Athanassiadi K, Gerazounis M, Moustardas M, et al. Sternal fractures: retrospective analysis of 100 cases. World J Surg. 2002;26:1243-1246. doi: 10.1007/s00268-002-6511-5
4. Ferguson LP, Wilkinson AG, Beattie TF. Fracture of the sternum in children. Emerg Med J. 2003;20:518-520. doi: 10.1136/emj.20.6.518
5. Ramgopal S, Shaffiey SA, Conti KA. Pediatric sternal fractures from a Level 1 trauma center. J Pediatr Surg. 2019;54:1628-1631. doi: 10.1016/j.jpedsurg.2018.08.040
6. Sesia SB, Prüfer F, Mayr J. Sternal fracture in children: diagnosis by ultrasonography. European J Pediatr Surg Rep. 2017;5:e39-e42. doi: 10.1055/s-0037-1606197
7. Nickson C, Rippey J. Ultrasonography of sternal fractures. Australas J Ultrasound Med. 2011;14:6-11. doi: 10.1002/j.2205-0140.2011.tb00131.x
8. Bauman ZM, Yanala U, Waibel BH, et al. Sternal fixation for isolated traumatic sternal fractures improves pain and upper extremity range of motion. Eur J Trauma Emerg Surg. 2022;48:225-230. doi: 10.1007/s00068-020-01568-x
9. Culp B, Hurbanek JG, Novak J, et al. Acute traumatic sternum fracture in a female college hockey player. Orthopedics. 2010;33:683. doi: 10.3928/01477447-20100722-17
Consider this tool to reduce antibiotic-associated adverse events in patients with sepsis
ILLUSTRATIVE CASE
A 52-year-old woman presents to the emergency department complaining of dysuria and a fever. Her work-up yields a diagnosis of sepsis secondary to pyelonephritis and bacteremia. She is admitted and started on broad-spectrum antimicrobial therapy. The patient’s symptoms improve significantly over the next 48 hours of treatment. When should antibiotic therapy be discontinued to reduce the patient’s risk for antibiotic-associated AEs and to optimize antimicrobial stewardship?
Antimicrobial resistance is a growing public health risk associated with considerable morbidity and mortality, extended hospitalization, and increased medical expenditures.2-4 Antibiotic stewardship is vital in curbing antimicrobial resistance. The predictive biomarker PCT has emerged as both a diagnostic and prognostic agent for numerous infectious diseases. It has recently received much attention as an adjunct to clinical judgment for discontinuation of antibiotic therapy in hospitalized patients with lower respiratory tract infections and/or sepsis.5-11 Indeed, use of PCT guidance in these patients has resulted in decreased AEs, as well as an enhanced survival benefit.5-15
The utility of PCT-guided early discontinuation of antibiotics had yet to be studied in an expanded population of hospitalized patients with sepsis—especially with regard to AEs associated with multidrug-resistant organisms (MDROs) and Clostridioides difficile (formerly Clostridium difficile). The Surviving Sepsis Campaign’s 2021 international guidelines support the use of PCT in conjunction with clinical evaluation for shortening the duration of antibiotic therapy (“weak recommendation, low quality of evidence”).16 They also suggest daily reassessment for de-escalation of antibiotic use (“weak recommendation, very low quality of evidence”) as a possible way to decrease MDROs and AEs but state that more and better trials are needed.15
STUDY SUMMARY
PCT-guided intervention reduced infection-associated AEs
This pragmatic, real-world, multicenter, randomized clinical trial evaluated the use of PCT-guided early discontinuation of antibiotic therapy in patients with sepsis, in hopes of decreasing infection-associated AEs related to prolonged antibiotic exposure.1 The trial took place in 7 hospitals in Athens, Greece, with 266 patients randomized to the PCT-guided intervention or the standard of care (SOC)—the 2016 international guidelines for the management of sepsis and septic shock from the Surviving Sepsis campaign.17 Study participants had sepsis, as defined by a sequential organ failure assessment (SOFA) score ≥ 2, and infections that included pneumonia, pyelonephritis, or bacteremia.16 Pregnancy, lactation, HIV infection with a low CD4 count, neutropenia, cystic fibrosis, and viral, parasitic, or tuberculosis infections were exclusion criteria. Of note, all patients were managed on general medical wards and not in intensive care units.
Serum PCT samples were collected at baseline and then at Day 5 of therapy. Discontinuation of antibiotic therapy in the PCT trial arm occurred once PCT levels were ≤ 0.5 mcg/L or were reduced by at least 80%. If PCT levels did not meet one of these criteria, the lab test would be repeated daily and antibiotic therapy would continue until the rule was met. Neither patients nor investigators were blinded to the treatment assignments, but investigators in the SOC arm were kept unaware of Day 5 PCT results. In the PCT arm, 71% of participants met Day 5 criteria for stopping antibiotics, and a retrospective analysis indicated that a near-identical 70% in the SOC arm also would have met the same criteria.
The assessment of stool colonization with either C difficile or MDROs was done by stool cultures at baseline and on Days 7, 28, and 180.
The primary outcome of infection-associated AEs, which was evaluated at 180 days, was defined as new cases of C difficile or MDRO infection, or death associated with baseline infection with either C difficile or an MDRO. Of the 133 participants allocated to each trial arm, 8 patients in the intervention group and 2 in the SOC group withdrew consent prior to treatment in the intervention group, with the remaining 125 and 131 participants, respectively, completing the interventions and not lost to follow-up.
Continue to: In an intention-to-treat analysis...
In an intention-to-treat analysis, 9 participants (7.2%; 95% CI, 3.8%-13.1%) in the PCT group compared with 20 participants (15.3%; 95% CI, 10.1%-22.4%) in the SOC group experienced the primary outcome of an antibiotic-associated AE at 180 days, resulting in a hazard ratio (HR) of 0.45 (95% CI, 0.2-0.98).
Secondary outcomes also favored the PCT arm regarding 28-day mortality (19 vs 37 patients; HR = 0.51; 95% CI, 0.29-0.89), median length of antibiotic treatment (5 days in the PCT group and 10 days in the SOC group; P < .001), and median hospitalization cost (24% greater in the SOC group; P = .05). Results for 180-day mortality were 30.4% in the PCT arm and 38.2% in the SOC arm (HR = 0.71; 95% CI, 0.42-1.19), thereby not achieving statistical significance.
WHAT'S NEW
An effective tool in reducing AEs in patients with sepsis
In this multicenter trial, PCT proved successful as a clinical decision tool for discontinuing antibiotic therapy and decreasing infection-associated AEs in patients with sepsis.
Caveats
A promising approach but its superiority is uncertain
The confidence interval for the AE hazard ratio was very wide, but significant, suggesting greater uncertainty and less precision in the chance of obtaining improved outcomes with PCT-guided intervention. However, these data also clarify that outcomes should (at least) not be worse with PCT-directed therapy.
CHALLENGES TO IMPLEMENTATION
Assay limitations and potential resistance to a new decision tool
The primary challenge to implementation is likely the availability of the PCT assay and the immediacy of turnaround time to enable physicians to make daily decisions regarding antibiotic therapy de-escalation. Additionally, as with any new knowledge, local culture and physician buy-in may limit implementation of this ever-more-valuable patient care tool.
1. Kyriazopoulou E, Liaskou-Antoniou L, Adamis G, et al. Procalcitonin to reduce long-term infection-associated adverse events in sepsis: a randomized trial. Am J Respir Crit Care Med. 2021;203:202-210. doi: 10.1164/rccm.202004-1201OC
2. European Centre for Disease Prevention and Control. US CDC report on antibiotic resistance threats in the United States, 2013. ECDC comment. September 18, 2013. Accessed December 29, 2022. www.ecdc.europa.eu/en/news-events/us-cdc-report-antibiotic-resistance-threats-united-states-2013
3. Peters L, Olson L, Khu DTK, et al. Multiple antibiotic resistance as a risk factor for mortality and prolonged hospital stay: a cohort study among neonatal intensive care patients with hospital-acquired infections caused by gram-negative bacteria in Vietnam. PloS One. 2019;14:e0215666. doi: 10.1371/journal.pone.0215666
4. Cosgrove SE. The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis. 2006;42(suppl 2):S82-S89. doi: 10.1086/499406
5. Schuetz P, Beishuizen A, Broyles M, et al. Procalcitonin (PCT)-guided antibiotic stewardship: an international experts consensus on optimized clinical use. Clin Chem Lab Med. 2019;57:1308-1318. doi: 10.1515/cclm-2018-1181
6. Schuetz P, Christ-Crain M, Thomann R, et al; ProHOSP Study Group. Effect of procalcitonin-based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302:1059-1066. doi: 10.1001/jama.2009.1297
7. Bouadma L, Luyt CE, Tubach F, et al; PRORATA trial group. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375:463-474. doi: 10.1016/S0140-6736(09)61879-1
8. Christ-Crain M, Jaccard-Stolz D, Bingisser R, et al. Effect of procalcitonin-guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster-randomised, single-blinded intervention trial. Lancet. 2004;363:600-607. doi: 10.1016/S0140-6736(04)15591-8
9. Christ-Crain M, Stolz D, Bingisser R, et al. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84-93. doi: 10.1164/rccm.200512-1922OC
10. de Jong E, van Oers JA, Beishuizen A, et al. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis. 2016;16:819-827. doi: 10.1016/S1473-3099(16)00053-0
11. Nobre V, Harbarth S, Graf JD, et al. Use of procalcitonin to shorten antibiotic treatment duration in septic patients: a randomized trial. Am J Respir Crit Care Med. 2008;177:498-505. doi: 10.1164/rccm.200708-1238OC
12. Schuetz P, Wirz Y, Sager R, et al. Effect of procalcitonin-guided antibiotic treatment on mortality in acute respiratory infections: a patient level meta-analysis. Lancet Infect Dis. 2018;18:95-107. doi: 10.1016/S1473-3099(17)30592-3
13. Schuetz P, Chiappa V, Briel M, et al. Procalcitonin algorithms for antibiotic therapy decisions: a systematic review of randomized controlled trials and recommendations for clinical algorithms. Arch Intern Med. 2011;171:1322-1331. doi: 10.1001/archin ternmed.2011.318
14. Wirz Y, Meier MA, Bouadma L, et al. Effect of procalcitonin-guided antibiotic treatment on clinical outcomes in intensive care unit patients with infection and sepsis patients: a patient-level meta-analysis of randomized trials. Crit Care. 2018;22:191. doi: 10.1186/s13054-018-2125-7
15. Elnajdy D, El-Dahiyat F. Antibiotics duration guided by biomarkers in hospitalized adult patients; a systematic review and meta-analysis. Infect Dis (Lond). 2022;54:387-402. doi: 10.1080/23744235.2022.2037701
16. Evans L, Rhodes A, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021;49:e1063-e1143. doi: 10.1097/CCM.0000000000005337
17. Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 2017;43:304-377. doi: 10.1007/s00134-017-4683-6
ILLUSTRATIVE CASE
A 52-year-old woman presents to the emergency department complaining of dysuria and a fever. Her work-up yields a diagnosis of sepsis secondary to pyelonephritis and bacteremia. She is admitted and started on broad-spectrum antimicrobial therapy. The patient’s symptoms improve significantly over the next 48 hours of treatment. When should antibiotic therapy be discontinued to reduce the patient’s risk for antibiotic-associated AEs and to optimize antimicrobial stewardship?
Antimicrobial resistance is a growing public health risk associated with considerable morbidity and mortality, extended hospitalization, and increased medical expenditures.2-4 Antibiotic stewardship is vital in curbing antimicrobial resistance. The predictive biomarker PCT has emerged as both a diagnostic and prognostic agent for numerous infectious diseases. It has recently received much attention as an adjunct to clinical judgment for discontinuation of antibiotic therapy in hospitalized patients with lower respiratory tract infections and/or sepsis.5-11 Indeed, use of PCT guidance in these patients has resulted in decreased AEs, as well as an enhanced survival benefit.5-15
The utility of PCT-guided early discontinuation of antibiotics had yet to be studied in an expanded population of hospitalized patients with sepsis—especially with regard to AEs associated with multidrug-resistant organisms (MDROs) and Clostridioides difficile (formerly Clostridium difficile). The Surviving Sepsis Campaign’s 2021 international guidelines support the use of PCT in conjunction with clinical evaluation for shortening the duration of antibiotic therapy (“weak recommendation, low quality of evidence”).16 They also suggest daily reassessment for de-escalation of antibiotic use (“weak recommendation, very low quality of evidence”) as a possible way to decrease MDROs and AEs but state that more and better trials are needed.15
STUDY SUMMARY
PCT-guided intervention reduced infection-associated AEs
This pragmatic, real-world, multicenter, randomized clinical trial evaluated the use of PCT-guided early discontinuation of antibiotic therapy in patients with sepsis, in hopes of decreasing infection-associated AEs related to prolonged antibiotic exposure.1 The trial took place in 7 hospitals in Athens, Greece, with 266 patients randomized to the PCT-guided intervention or the standard of care (SOC)—the 2016 international guidelines for the management of sepsis and septic shock from the Surviving Sepsis campaign.17 Study participants had sepsis, as defined by a sequential organ failure assessment (SOFA) score ≥ 2, and infections that included pneumonia, pyelonephritis, or bacteremia.16 Pregnancy, lactation, HIV infection with a low CD4 count, neutropenia, cystic fibrosis, and viral, parasitic, or tuberculosis infections were exclusion criteria. Of note, all patients were managed on general medical wards and not in intensive care units.
Serum PCT samples were collected at baseline and then at Day 5 of therapy. Discontinuation of antibiotic therapy in the PCT trial arm occurred once PCT levels were ≤ 0.5 mcg/L or were reduced by at least 80%. If PCT levels did not meet one of these criteria, the lab test would be repeated daily and antibiotic therapy would continue until the rule was met. Neither patients nor investigators were blinded to the treatment assignments, but investigators in the SOC arm were kept unaware of Day 5 PCT results. In the PCT arm, 71% of participants met Day 5 criteria for stopping antibiotics, and a retrospective analysis indicated that a near-identical 70% in the SOC arm also would have met the same criteria.
The assessment of stool colonization with either C difficile or MDROs was done by stool cultures at baseline and on Days 7, 28, and 180.
The primary outcome of infection-associated AEs, which was evaluated at 180 days, was defined as new cases of C difficile or MDRO infection, or death associated with baseline infection with either C difficile or an MDRO. Of the 133 participants allocated to each trial arm, 8 patients in the intervention group and 2 in the SOC group withdrew consent prior to treatment in the intervention group, with the remaining 125 and 131 participants, respectively, completing the interventions and not lost to follow-up.
Continue to: In an intention-to-treat analysis...
In an intention-to-treat analysis, 9 participants (7.2%; 95% CI, 3.8%-13.1%) in the PCT group compared with 20 participants (15.3%; 95% CI, 10.1%-22.4%) in the SOC group experienced the primary outcome of an antibiotic-associated AE at 180 days, resulting in a hazard ratio (HR) of 0.45 (95% CI, 0.2-0.98).
Secondary outcomes also favored the PCT arm regarding 28-day mortality (19 vs 37 patients; HR = 0.51; 95% CI, 0.29-0.89), median length of antibiotic treatment (5 days in the PCT group and 10 days in the SOC group; P < .001), and median hospitalization cost (24% greater in the SOC group; P = .05). Results for 180-day mortality were 30.4% in the PCT arm and 38.2% in the SOC arm (HR = 0.71; 95% CI, 0.42-1.19), thereby not achieving statistical significance.
WHAT'S NEW
An effective tool in reducing AEs in patients with sepsis
In this multicenter trial, PCT proved successful as a clinical decision tool for discontinuing antibiotic therapy and decreasing infection-associated AEs in patients with sepsis.
Caveats
A promising approach but its superiority is uncertain
The confidence interval for the AE hazard ratio was very wide, but significant, suggesting greater uncertainty and less precision in the chance of obtaining improved outcomes with PCT-guided intervention. However, these data also clarify that outcomes should (at least) not be worse with PCT-directed therapy.
CHALLENGES TO IMPLEMENTATION
Assay limitations and potential resistance to a new decision tool
The primary challenge to implementation is likely the availability of the PCT assay and the immediacy of turnaround time to enable physicians to make daily decisions regarding antibiotic therapy de-escalation. Additionally, as with any new knowledge, local culture and physician buy-in may limit implementation of this ever-more-valuable patient care tool.
ILLUSTRATIVE CASE
A 52-year-old woman presents to the emergency department complaining of dysuria and a fever. Her work-up yields a diagnosis of sepsis secondary to pyelonephritis and bacteremia. She is admitted and started on broad-spectrum antimicrobial therapy. The patient’s symptoms improve significantly over the next 48 hours of treatment. When should antibiotic therapy be discontinued to reduce the patient’s risk for antibiotic-associated AEs and to optimize antimicrobial stewardship?
Antimicrobial resistance is a growing public health risk associated with considerable morbidity and mortality, extended hospitalization, and increased medical expenditures.2-4 Antibiotic stewardship is vital in curbing antimicrobial resistance. The predictive biomarker PCT has emerged as both a diagnostic and prognostic agent for numerous infectious diseases. It has recently received much attention as an adjunct to clinical judgment for discontinuation of antibiotic therapy in hospitalized patients with lower respiratory tract infections and/or sepsis.5-11 Indeed, use of PCT guidance in these patients has resulted in decreased AEs, as well as an enhanced survival benefit.5-15
The utility of PCT-guided early discontinuation of antibiotics had yet to be studied in an expanded population of hospitalized patients with sepsis—especially with regard to AEs associated with multidrug-resistant organisms (MDROs) and Clostridioides difficile (formerly Clostridium difficile). The Surviving Sepsis Campaign’s 2021 international guidelines support the use of PCT in conjunction with clinical evaluation for shortening the duration of antibiotic therapy (“weak recommendation, low quality of evidence”).16 They also suggest daily reassessment for de-escalation of antibiotic use (“weak recommendation, very low quality of evidence”) as a possible way to decrease MDROs and AEs but state that more and better trials are needed.15
STUDY SUMMARY
PCT-guided intervention reduced infection-associated AEs
This pragmatic, real-world, multicenter, randomized clinical trial evaluated the use of PCT-guided early discontinuation of antibiotic therapy in patients with sepsis, in hopes of decreasing infection-associated AEs related to prolonged antibiotic exposure.1 The trial took place in 7 hospitals in Athens, Greece, with 266 patients randomized to the PCT-guided intervention or the standard of care (SOC)—the 2016 international guidelines for the management of sepsis and septic shock from the Surviving Sepsis campaign.17 Study participants had sepsis, as defined by a sequential organ failure assessment (SOFA) score ≥ 2, and infections that included pneumonia, pyelonephritis, or bacteremia.16 Pregnancy, lactation, HIV infection with a low CD4 count, neutropenia, cystic fibrosis, and viral, parasitic, or tuberculosis infections were exclusion criteria. Of note, all patients were managed on general medical wards and not in intensive care units.
Serum PCT samples were collected at baseline and then at Day 5 of therapy. Discontinuation of antibiotic therapy in the PCT trial arm occurred once PCT levels were ≤ 0.5 mcg/L or were reduced by at least 80%. If PCT levels did not meet one of these criteria, the lab test would be repeated daily and antibiotic therapy would continue until the rule was met. Neither patients nor investigators were blinded to the treatment assignments, but investigators in the SOC arm were kept unaware of Day 5 PCT results. In the PCT arm, 71% of participants met Day 5 criteria for stopping antibiotics, and a retrospective analysis indicated that a near-identical 70% in the SOC arm also would have met the same criteria.
The assessment of stool colonization with either C difficile or MDROs was done by stool cultures at baseline and on Days 7, 28, and 180.
The primary outcome of infection-associated AEs, which was evaluated at 180 days, was defined as new cases of C difficile or MDRO infection, or death associated with baseline infection with either C difficile or an MDRO. Of the 133 participants allocated to each trial arm, 8 patients in the intervention group and 2 in the SOC group withdrew consent prior to treatment in the intervention group, with the remaining 125 and 131 participants, respectively, completing the interventions and not lost to follow-up.
Continue to: In an intention-to-treat analysis...
In an intention-to-treat analysis, 9 participants (7.2%; 95% CI, 3.8%-13.1%) in the PCT group compared with 20 participants (15.3%; 95% CI, 10.1%-22.4%) in the SOC group experienced the primary outcome of an antibiotic-associated AE at 180 days, resulting in a hazard ratio (HR) of 0.45 (95% CI, 0.2-0.98).
Secondary outcomes also favored the PCT arm regarding 28-day mortality (19 vs 37 patients; HR = 0.51; 95% CI, 0.29-0.89), median length of antibiotic treatment (5 days in the PCT group and 10 days in the SOC group; P < .001), and median hospitalization cost (24% greater in the SOC group; P = .05). Results for 180-day mortality were 30.4% in the PCT arm and 38.2% in the SOC arm (HR = 0.71; 95% CI, 0.42-1.19), thereby not achieving statistical significance.
WHAT'S NEW
An effective tool in reducing AEs in patients with sepsis
In this multicenter trial, PCT proved successful as a clinical decision tool for discontinuing antibiotic therapy and decreasing infection-associated AEs in patients with sepsis.
Caveats
A promising approach but its superiority is uncertain
The confidence interval for the AE hazard ratio was very wide, but significant, suggesting greater uncertainty and less precision in the chance of obtaining improved outcomes with PCT-guided intervention. However, these data also clarify that outcomes should (at least) not be worse with PCT-directed therapy.
CHALLENGES TO IMPLEMENTATION
Assay limitations and potential resistance to a new decision tool
The primary challenge to implementation is likely the availability of the PCT assay and the immediacy of turnaround time to enable physicians to make daily decisions regarding antibiotic therapy de-escalation. Additionally, as with any new knowledge, local culture and physician buy-in may limit implementation of this ever-more-valuable patient care tool.
1. Kyriazopoulou E, Liaskou-Antoniou L, Adamis G, et al. Procalcitonin to reduce long-term infection-associated adverse events in sepsis: a randomized trial. Am J Respir Crit Care Med. 2021;203:202-210. doi: 10.1164/rccm.202004-1201OC
2. European Centre for Disease Prevention and Control. US CDC report on antibiotic resistance threats in the United States, 2013. ECDC comment. September 18, 2013. Accessed December 29, 2022. www.ecdc.europa.eu/en/news-events/us-cdc-report-antibiotic-resistance-threats-united-states-2013
3. Peters L, Olson L, Khu DTK, et al. Multiple antibiotic resistance as a risk factor for mortality and prolonged hospital stay: a cohort study among neonatal intensive care patients with hospital-acquired infections caused by gram-negative bacteria in Vietnam. PloS One. 2019;14:e0215666. doi: 10.1371/journal.pone.0215666
4. Cosgrove SE. The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis. 2006;42(suppl 2):S82-S89. doi: 10.1086/499406
5. Schuetz P, Beishuizen A, Broyles M, et al. Procalcitonin (PCT)-guided antibiotic stewardship: an international experts consensus on optimized clinical use. Clin Chem Lab Med. 2019;57:1308-1318. doi: 10.1515/cclm-2018-1181
6. Schuetz P, Christ-Crain M, Thomann R, et al; ProHOSP Study Group. Effect of procalcitonin-based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302:1059-1066. doi: 10.1001/jama.2009.1297
7. Bouadma L, Luyt CE, Tubach F, et al; PRORATA trial group. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375:463-474. doi: 10.1016/S0140-6736(09)61879-1
8. Christ-Crain M, Jaccard-Stolz D, Bingisser R, et al. Effect of procalcitonin-guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster-randomised, single-blinded intervention trial. Lancet. 2004;363:600-607. doi: 10.1016/S0140-6736(04)15591-8
9. Christ-Crain M, Stolz D, Bingisser R, et al. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84-93. doi: 10.1164/rccm.200512-1922OC
10. de Jong E, van Oers JA, Beishuizen A, et al. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis. 2016;16:819-827. doi: 10.1016/S1473-3099(16)00053-0
11. Nobre V, Harbarth S, Graf JD, et al. Use of procalcitonin to shorten antibiotic treatment duration in septic patients: a randomized trial. Am J Respir Crit Care Med. 2008;177:498-505. doi: 10.1164/rccm.200708-1238OC
12. Schuetz P, Wirz Y, Sager R, et al. Effect of procalcitonin-guided antibiotic treatment on mortality in acute respiratory infections: a patient level meta-analysis. Lancet Infect Dis. 2018;18:95-107. doi: 10.1016/S1473-3099(17)30592-3
13. Schuetz P, Chiappa V, Briel M, et al. Procalcitonin algorithms for antibiotic therapy decisions: a systematic review of randomized controlled trials and recommendations for clinical algorithms. Arch Intern Med. 2011;171:1322-1331. doi: 10.1001/archin ternmed.2011.318
14. Wirz Y, Meier MA, Bouadma L, et al. Effect of procalcitonin-guided antibiotic treatment on clinical outcomes in intensive care unit patients with infection and sepsis patients: a patient-level meta-analysis of randomized trials. Crit Care. 2018;22:191. doi: 10.1186/s13054-018-2125-7
15. Elnajdy D, El-Dahiyat F. Antibiotics duration guided by biomarkers in hospitalized adult patients; a systematic review and meta-analysis. Infect Dis (Lond). 2022;54:387-402. doi: 10.1080/23744235.2022.2037701
16. Evans L, Rhodes A, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021;49:e1063-e1143. doi: 10.1097/CCM.0000000000005337
17. Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 2017;43:304-377. doi: 10.1007/s00134-017-4683-6
1. Kyriazopoulou E, Liaskou-Antoniou L, Adamis G, et al. Procalcitonin to reduce long-term infection-associated adverse events in sepsis: a randomized trial. Am J Respir Crit Care Med. 2021;203:202-210. doi: 10.1164/rccm.202004-1201OC
2. European Centre for Disease Prevention and Control. US CDC report on antibiotic resistance threats in the United States, 2013. ECDC comment. September 18, 2013. Accessed December 29, 2022. www.ecdc.europa.eu/en/news-events/us-cdc-report-antibiotic-resistance-threats-united-states-2013
3. Peters L, Olson L, Khu DTK, et al. Multiple antibiotic resistance as a risk factor for mortality and prolonged hospital stay: a cohort study among neonatal intensive care patients with hospital-acquired infections caused by gram-negative bacteria in Vietnam. PloS One. 2019;14:e0215666. doi: 10.1371/journal.pone.0215666
4. Cosgrove SE. The relationship between antimicrobial resistance and patient outcomes: mortality, length of hospital stay, and health care costs. Clin Infect Dis. 2006;42(suppl 2):S82-S89. doi: 10.1086/499406
5. Schuetz P, Beishuizen A, Broyles M, et al. Procalcitonin (PCT)-guided antibiotic stewardship: an international experts consensus on optimized clinical use. Clin Chem Lab Med. 2019;57:1308-1318. doi: 10.1515/cclm-2018-1181
6. Schuetz P, Christ-Crain M, Thomann R, et al; ProHOSP Study Group. Effect of procalcitonin-based guidelines vs standard guidelines on antibiotic use in lower respiratory tract infections: the ProHOSP randomized controlled trial. JAMA. 2009;302:1059-1066. doi: 10.1001/jama.2009.1297
7. Bouadma L, Luyt CE, Tubach F, et al; PRORATA trial group. Use of procalcitonin to reduce patients’ exposure to antibiotics in intensive care units (PRORATA trial): a multicentre randomised controlled trial. Lancet. 2010;375:463-474. doi: 10.1016/S0140-6736(09)61879-1
8. Christ-Crain M, Jaccard-Stolz D, Bingisser R, et al. Effect of procalcitonin-guided treatment on antibiotic use and outcome in lower respiratory tract infections: cluster-randomised, single-blinded intervention trial. Lancet. 2004;363:600-607. doi: 10.1016/S0140-6736(04)15591-8
9. Christ-Crain M, Stolz D, Bingisser R, et al. Procalcitonin guidance of antibiotic therapy in community-acquired pneumonia: a randomized trial. Am J Respir Crit Care Med. 2006;174:84-93. doi: 10.1164/rccm.200512-1922OC
10. de Jong E, van Oers JA, Beishuizen A, et al. Efficacy and safety of procalcitonin guidance in reducing the duration of antibiotic treatment in critically ill patients: a randomised, controlled, open-label trial. Lancet Infect Dis. 2016;16:819-827. doi: 10.1016/S1473-3099(16)00053-0
11. Nobre V, Harbarth S, Graf JD, et al. Use of procalcitonin to shorten antibiotic treatment duration in septic patients: a randomized trial. Am J Respir Crit Care Med. 2008;177:498-505. doi: 10.1164/rccm.200708-1238OC
12. Schuetz P, Wirz Y, Sager R, et al. Effect of procalcitonin-guided antibiotic treatment on mortality in acute respiratory infections: a patient level meta-analysis. Lancet Infect Dis. 2018;18:95-107. doi: 10.1016/S1473-3099(17)30592-3
13. Schuetz P, Chiappa V, Briel M, et al. Procalcitonin algorithms for antibiotic therapy decisions: a systematic review of randomized controlled trials and recommendations for clinical algorithms. Arch Intern Med. 2011;171:1322-1331. doi: 10.1001/archin ternmed.2011.318
14. Wirz Y, Meier MA, Bouadma L, et al. Effect of procalcitonin-guided antibiotic treatment on clinical outcomes in intensive care unit patients with infection and sepsis patients: a patient-level meta-analysis of randomized trials. Crit Care. 2018;22:191. doi: 10.1186/s13054-018-2125-7
15. Elnajdy D, El-Dahiyat F. Antibiotics duration guided by biomarkers in hospitalized adult patients; a systematic review and meta-analysis. Infect Dis (Lond). 2022;54:387-402. doi: 10.1080/23744235.2022.2037701
16. Evans L, Rhodes A, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock 2021. Crit Care Med. 2021;49:e1063-e1143. doi: 10.1097/CCM.0000000000005337
17. Rhodes A, Evans LE, Alhazzani W, et al. Surviving Sepsis Campaign: international guidelines for management of sepsis and septic shock: 2016. Intensive Care Med. 2017;43:304-377. doi: 10.1007/s00134-017-4683-6
PRACTICE CHANGER
For patients hospitalized with sepsis, consider procalcitonin (PCT)-guided early discontinuation of antibiotic therapy for fewer infection-associated adverse events (AEs).
STRENGTH OF RECOMMENDATION
Kyriazopoulou E, Liaskou-Antoniou L, Adamis G, et al. Procalcitonin to reduce long-term infection-associated adverse events in sepsis. A randomized trial. Am J Respir Crit Care Med. 2021;203:202-210. doi: 10.1164/rccm.202004-1201OC
Does regular walking improve lipid levels in adults?
Evidence summary
Walking’s impact on cholesterol levels is modest, inconsistent
A 2022 systematic review and meta-analysis of 21 studies (n = 1129) evaluated the effects of walking on lipids and lipoproteins in women older than 18 years who were overweight or obese and were not taking any lipid-lowering medications. Median TC was 206 mg/dL and median LDL was 126 mg/dL.1
The primary outcome found that walking decreased TC and LDL levels independent of diet and weight loss. Twenty studies reported on TC and showed that walking significantly decreased TC levels compared to the control groups (raw mean difference [RMD] = 6.7 mg/dL; 95% CI, 0.4-12.9; P = .04). Fifteen studies examined LDL and showed a significant decrease in LDL levels with walking compared to control groups (RMD = 7.4 mg/dL; 95% CI, 0.3-14.5; P = .04). However, the small magnitude of the changes may have little clinical impact.1
There were no significant changes in the walking groups compared to the control groups for triglycerides (17 studies; RMD = 2.2 mg/dL; 95% CI, –8.4 to 12.8; P = .68) or high-density lipoprotein (HDL) (18 studies; RMD = 1.5 mg/dL; 95% CI, –0.4 to 3.3; P = .12). Included studies were required to be controlled but were otherwise not described. The overall risk for bias was determined to be low.1
A 2020 RCT (n = 22) assessed the effects of a walking intervention on cholesterol and cardiovascular disease (CVD) risk in individuals ages 40 to 65 years with moderate CVD risk but without diabetes or CVD.2 Moderate CVD risk was defined as a 2% to 5% 10-year risk for a CVD event using the European HeartScore, which incorporates age, sex, blood pressure, lipid levels, and smoking status3; however, study participants were not required to have hyperlipidemia. Participants were enrolled in a 12-week, nurse-led intervention of moderate-paced walking for 30 to 45 minutes 5 times weekly.
Individuals in the intervention group had significant decreases in average TC levels from baseline to follow-up (244.6 mg/dL vs 213.7 mg/dL; P = .001). As a result, participants’ average 10-year CVD risk was significantly reduced from moderate risk to low risk (2.6% vs 1.8%; P = 038) and was significantly lower in the intervention group than in the control group at follow-up (1.8% vs 3.1%; P = .019). No blinding was used, and the use of lipid-lowering medications was not reported, which could have impacted the results.2
A 2008 RCT (n = 67) examined the effect of a home-based walking program (12 weeks of brisk walking, at least 30 min/d and at least 5 d/wk, with at least 300 kcal burned per walk) vs a sedentary control group in men ages 45 to 65 years with hyperlipidemia (TC > 240 mg/dL and/or TC/HDL-C ratio ≥ 6) who were not receiving lipid-lowering medication. There were no significant changes from baseline to follow-up in the walking group compared to the control group in TC (adjusted mean difference [AMD] = –9.3 mg/dL; 95% CI, –22.8 to 4.64; P = .19), HDL-C (AMD = 2.7 mg/dL; 95% CI, –0.4 to 5.4; P = .07) or triglycerides (AMD = –26.6 mg/dL; 95% CI, –56.7 to 2.7; P = .07).4
A 2002 RCT (n = 111) of sedentary men and women (BMI, 25-35; ages, 40-65 years) with dyslipidemia (LDL of 130-190 mg/dL, or HDL < 40 mg/dL for men or < 45 mg/dL for women) examined the impact of various physical activity levels for 8 months when compared to a control group observed for 6 months. The group assigned to low-amount, moderate-intensity physical activity walked an equivalent of 12 miles per week.5
Continue to: In this group...
In this group, there was a significant decrease in average triglyceride concentrations from baseline to follow-up (mean ± standard error = 196.8 ± 30.5 mg/dL vs 145.2 ± 16.0 mg/dL; P < .001). Significance of the change compared with changes in the control group was not reported, although triglycerides in the control group increased from baseline to follow-up (132.1 ± 11.0 vs 155.8 ± 14.9 mg/dL). There were no significant changes from baseline to follow-up in TC (194 ± 4.8 vs 197.9 ± 5.4 mg/dL), LDL (122.7 ± 4.0 vs 127.8 ± 4.1 mg/dL), or HDL (42.0 ± 1.9 vs 43.1 ± 2.5 mg/dL); P values of pre-post changes and comparison to control group were not reported.5
Recommendations from others
The Physical Activity Guidelines for Americans, published by the Department of Health and Human Services and updated in 2018, cite adherence to the published guidelines as a protective factor against high LDL and total lipids in both adults and children.6 The guidelines for adults recommend 150 to 300 minutes of moderate-intensity or 75 to 150 minutes of vigorous-intensity aerobic exercise per week, as well as muscle-strengthening activities of moderate or greater intensity 2 or more days per week. Brisk walking is included as an example of a moderate-intensity activity. These same guidelines are cited and endorsed by the American College of Sports Medicine and the American Heart Association.7,8
Editor’s takeaway
The lipid reductions achieved from walking—if any—are minimal. By themselves, these small reductions will not accomplish our lipid-lowering goals. However, cholesterol goals are primarily disease oriented. This evidence does not directly inform us of important patient-oriented outcomes, such as morbidity, mortality, and vitality.
1. Ballard AM, Davis A, Wong B, et al. The effects of exclusive walking on lipids and lipoproteins in women with overweight and obesity: a systematic review and meta-analysis. Am J Health Promot. 2022;36:328-339. doi: 10.1177/08901171211048135
2. Akgöz AD, Gözüm S. Effectiveness of a nurse-led physical activity intervention to decrease cardiovascular disease risk in middle-aged adults: a pilot randomized controlled study. J Vasc Nurs. 2020;38:140-148. doi: 10.1016/j.jvn.2020.05.002
3. European Association of Preventive Cardiology. HeartScore. Accessed December 23, 2022. www.heartscore.org/en_GB
4. Coghill N, Cooper AR. The effect of a home-based walking program on risk factors for coronary heart disease in hypercholesterolaemic men: a randomized controlled trial. Prev Med. 2008; 46:545-551. doi: 10.1016/j.ypmed.2008.01.002
5. Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med. 2002;347:1483-1492. doi: 10.1056/NEJMoa020194
6. US Department of Health and Human Services. Physical Activity Guidelines for Americans, 2nd edition. Washington, DC: US Department of Health and Human Services; 2018. Accessed December 23, 2022. https://health.gov/sites/default/files/2019-09/Physical_Activity_Guidelines_2nd_edition.pdf
7. American Heart Association. Recommendations for physical activity in adults and kids. Accessed December 23, 2022. www.heart.org/en/healthy-living/fitness/fitness-basics/aha-recs-for-physical-activity-in-adults
8. American College of Sports Medicine. Trending topic: physical activity guidelines. Accessed December 23, 2022. www.acsm.org/education-resources/trending-topics-resources/physical-activity-guidelines
Evidence summary
Walking’s impact on cholesterol levels is modest, inconsistent
A 2022 systematic review and meta-analysis of 21 studies (n = 1129) evaluated the effects of walking on lipids and lipoproteins in women older than 18 years who were overweight or obese and were not taking any lipid-lowering medications. Median TC was 206 mg/dL and median LDL was 126 mg/dL.1
The primary outcome found that walking decreased TC and LDL levels independent of diet and weight loss. Twenty studies reported on TC and showed that walking significantly decreased TC levels compared to the control groups (raw mean difference [RMD] = 6.7 mg/dL; 95% CI, 0.4-12.9; P = .04). Fifteen studies examined LDL and showed a significant decrease in LDL levels with walking compared to control groups (RMD = 7.4 mg/dL; 95% CI, 0.3-14.5; P = .04). However, the small magnitude of the changes may have little clinical impact.1
There were no significant changes in the walking groups compared to the control groups for triglycerides (17 studies; RMD = 2.2 mg/dL; 95% CI, –8.4 to 12.8; P = .68) or high-density lipoprotein (HDL) (18 studies; RMD = 1.5 mg/dL; 95% CI, –0.4 to 3.3; P = .12). Included studies were required to be controlled but were otherwise not described. The overall risk for bias was determined to be low.1
A 2020 RCT (n = 22) assessed the effects of a walking intervention on cholesterol and cardiovascular disease (CVD) risk in individuals ages 40 to 65 years with moderate CVD risk but without diabetes or CVD.2 Moderate CVD risk was defined as a 2% to 5% 10-year risk for a CVD event using the European HeartScore, which incorporates age, sex, blood pressure, lipid levels, and smoking status3; however, study participants were not required to have hyperlipidemia. Participants were enrolled in a 12-week, nurse-led intervention of moderate-paced walking for 30 to 45 minutes 5 times weekly.
Individuals in the intervention group had significant decreases in average TC levels from baseline to follow-up (244.6 mg/dL vs 213.7 mg/dL; P = .001). As a result, participants’ average 10-year CVD risk was significantly reduced from moderate risk to low risk (2.6% vs 1.8%; P = 038) and was significantly lower in the intervention group than in the control group at follow-up (1.8% vs 3.1%; P = .019). No blinding was used, and the use of lipid-lowering medications was not reported, which could have impacted the results.2
A 2008 RCT (n = 67) examined the effect of a home-based walking program (12 weeks of brisk walking, at least 30 min/d and at least 5 d/wk, with at least 300 kcal burned per walk) vs a sedentary control group in men ages 45 to 65 years with hyperlipidemia (TC > 240 mg/dL and/or TC/HDL-C ratio ≥ 6) who were not receiving lipid-lowering medication. There were no significant changes from baseline to follow-up in the walking group compared to the control group in TC (adjusted mean difference [AMD] = –9.3 mg/dL; 95% CI, –22.8 to 4.64; P = .19), HDL-C (AMD = 2.7 mg/dL; 95% CI, –0.4 to 5.4; P = .07) or triglycerides (AMD = –26.6 mg/dL; 95% CI, –56.7 to 2.7; P = .07).4
A 2002 RCT (n = 111) of sedentary men and women (BMI, 25-35; ages, 40-65 years) with dyslipidemia (LDL of 130-190 mg/dL, or HDL < 40 mg/dL for men or < 45 mg/dL for women) examined the impact of various physical activity levels for 8 months when compared to a control group observed for 6 months. The group assigned to low-amount, moderate-intensity physical activity walked an equivalent of 12 miles per week.5
Continue to: In this group...
In this group, there was a significant decrease in average triglyceride concentrations from baseline to follow-up (mean ± standard error = 196.8 ± 30.5 mg/dL vs 145.2 ± 16.0 mg/dL; P < .001). Significance of the change compared with changes in the control group was not reported, although triglycerides in the control group increased from baseline to follow-up (132.1 ± 11.0 vs 155.8 ± 14.9 mg/dL). There were no significant changes from baseline to follow-up in TC (194 ± 4.8 vs 197.9 ± 5.4 mg/dL), LDL (122.7 ± 4.0 vs 127.8 ± 4.1 mg/dL), or HDL (42.0 ± 1.9 vs 43.1 ± 2.5 mg/dL); P values of pre-post changes and comparison to control group were not reported.5
Recommendations from others
The Physical Activity Guidelines for Americans, published by the Department of Health and Human Services and updated in 2018, cite adherence to the published guidelines as a protective factor against high LDL and total lipids in both adults and children.6 The guidelines for adults recommend 150 to 300 minutes of moderate-intensity or 75 to 150 minutes of vigorous-intensity aerobic exercise per week, as well as muscle-strengthening activities of moderate or greater intensity 2 or more days per week. Brisk walking is included as an example of a moderate-intensity activity. These same guidelines are cited and endorsed by the American College of Sports Medicine and the American Heart Association.7,8
Editor’s takeaway
The lipid reductions achieved from walking—if any—are minimal. By themselves, these small reductions will not accomplish our lipid-lowering goals. However, cholesterol goals are primarily disease oriented. This evidence does not directly inform us of important patient-oriented outcomes, such as morbidity, mortality, and vitality.
Evidence summary
Walking’s impact on cholesterol levels is modest, inconsistent
A 2022 systematic review and meta-analysis of 21 studies (n = 1129) evaluated the effects of walking on lipids and lipoproteins in women older than 18 years who were overweight or obese and were not taking any lipid-lowering medications. Median TC was 206 mg/dL and median LDL was 126 mg/dL.1
The primary outcome found that walking decreased TC and LDL levels independent of diet and weight loss. Twenty studies reported on TC and showed that walking significantly decreased TC levels compared to the control groups (raw mean difference [RMD] = 6.7 mg/dL; 95% CI, 0.4-12.9; P = .04). Fifteen studies examined LDL and showed a significant decrease in LDL levels with walking compared to control groups (RMD = 7.4 mg/dL; 95% CI, 0.3-14.5; P = .04). However, the small magnitude of the changes may have little clinical impact.1
There were no significant changes in the walking groups compared to the control groups for triglycerides (17 studies; RMD = 2.2 mg/dL; 95% CI, –8.4 to 12.8; P = .68) or high-density lipoprotein (HDL) (18 studies; RMD = 1.5 mg/dL; 95% CI, –0.4 to 3.3; P = .12). Included studies were required to be controlled but were otherwise not described. The overall risk for bias was determined to be low.1
A 2020 RCT (n = 22) assessed the effects of a walking intervention on cholesterol and cardiovascular disease (CVD) risk in individuals ages 40 to 65 years with moderate CVD risk but without diabetes or CVD.2 Moderate CVD risk was defined as a 2% to 5% 10-year risk for a CVD event using the European HeartScore, which incorporates age, sex, blood pressure, lipid levels, and smoking status3; however, study participants were not required to have hyperlipidemia. Participants were enrolled in a 12-week, nurse-led intervention of moderate-paced walking for 30 to 45 minutes 5 times weekly.
Individuals in the intervention group had significant decreases in average TC levels from baseline to follow-up (244.6 mg/dL vs 213.7 mg/dL; P = .001). As a result, participants’ average 10-year CVD risk was significantly reduced from moderate risk to low risk (2.6% vs 1.8%; P = 038) and was significantly lower in the intervention group than in the control group at follow-up (1.8% vs 3.1%; P = .019). No blinding was used, and the use of lipid-lowering medications was not reported, which could have impacted the results.2
A 2008 RCT (n = 67) examined the effect of a home-based walking program (12 weeks of brisk walking, at least 30 min/d and at least 5 d/wk, with at least 300 kcal burned per walk) vs a sedentary control group in men ages 45 to 65 years with hyperlipidemia (TC > 240 mg/dL and/or TC/HDL-C ratio ≥ 6) who were not receiving lipid-lowering medication. There were no significant changes from baseline to follow-up in the walking group compared to the control group in TC (adjusted mean difference [AMD] = –9.3 mg/dL; 95% CI, –22.8 to 4.64; P = .19), HDL-C (AMD = 2.7 mg/dL; 95% CI, –0.4 to 5.4; P = .07) or triglycerides (AMD = –26.6 mg/dL; 95% CI, –56.7 to 2.7; P = .07).4
A 2002 RCT (n = 111) of sedentary men and women (BMI, 25-35; ages, 40-65 years) with dyslipidemia (LDL of 130-190 mg/dL, or HDL < 40 mg/dL for men or < 45 mg/dL for women) examined the impact of various physical activity levels for 8 months when compared to a control group observed for 6 months. The group assigned to low-amount, moderate-intensity physical activity walked an equivalent of 12 miles per week.5
Continue to: In this group...
In this group, there was a significant decrease in average triglyceride concentrations from baseline to follow-up (mean ± standard error = 196.8 ± 30.5 mg/dL vs 145.2 ± 16.0 mg/dL; P < .001). Significance of the change compared with changes in the control group was not reported, although triglycerides in the control group increased from baseline to follow-up (132.1 ± 11.0 vs 155.8 ± 14.9 mg/dL). There were no significant changes from baseline to follow-up in TC (194 ± 4.8 vs 197.9 ± 5.4 mg/dL), LDL (122.7 ± 4.0 vs 127.8 ± 4.1 mg/dL), or HDL (42.0 ± 1.9 vs 43.1 ± 2.5 mg/dL); P values of pre-post changes and comparison to control group were not reported.5
Recommendations from others
The Physical Activity Guidelines for Americans, published by the Department of Health and Human Services and updated in 2018, cite adherence to the published guidelines as a protective factor against high LDL and total lipids in both adults and children.6 The guidelines for adults recommend 150 to 300 minutes of moderate-intensity or 75 to 150 minutes of vigorous-intensity aerobic exercise per week, as well as muscle-strengthening activities of moderate or greater intensity 2 or more days per week. Brisk walking is included as an example of a moderate-intensity activity. These same guidelines are cited and endorsed by the American College of Sports Medicine and the American Heart Association.7,8
Editor’s takeaway
The lipid reductions achieved from walking—if any—are minimal. By themselves, these small reductions will not accomplish our lipid-lowering goals. However, cholesterol goals are primarily disease oriented. This evidence does not directly inform us of important patient-oriented outcomes, such as morbidity, mortality, and vitality.
1. Ballard AM, Davis A, Wong B, et al. The effects of exclusive walking on lipids and lipoproteins in women with overweight and obesity: a systematic review and meta-analysis. Am J Health Promot. 2022;36:328-339. doi: 10.1177/08901171211048135
2. Akgöz AD, Gözüm S. Effectiveness of a nurse-led physical activity intervention to decrease cardiovascular disease risk in middle-aged adults: a pilot randomized controlled study. J Vasc Nurs. 2020;38:140-148. doi: 10.1016/j.jvn.2020.05.002
3. European Association of Preventive Cardiology. HeartScore. Accessed December 23, 2022. www.heartscore.org/en_GB
4. Coghill N, Cooper AR. The effect of a home-based walking program on risk factors for coronary heart disease in hypercholesterolaemic men: a randomized controlled trial. Prev Med. 2008; 46:545-551. doi: 10.1016/j.ypmed.2008.01.002
5. Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med. 2002;347:1483-1492. doi: 10.1056/NEJMoa020194
6. US Department of Health and Human Services. Physical Activity Guidelines for Americans, 2nd edition. Washington, DC: US Department of Health and Human Services; 2018. Accessed December 23, 2022. https://health.gov/sites/default/files/2019-09/Physical_Activity_Guidelines_2nd_edition.pdf
7. American Heart Association. Recommendations for physical activity in adults and kids. Accessed December 23, 2022. www.heart.org/en/healthy-living/fitness/fitness-basics/aha-recs-for-physical-activity-in-adults
8. American College of Sports Medicine. Trending topic: physical activity guidelines. Accessed December 23, 2022. www.acsm.org/education-resources/trending-topics-resources/physical-activity-guidelines
1. Ballard AM, Davis A, Wong B, et al. The effects of exclusive walking on lipids and lipoproteins in women with overweight and obesity: a systematic review and meta-analysis. Am J Health Promot. 2022;36:328-339. doi: 10.1177/08901171211048135
2. Akgöz AD, Gözüm S. Effectiveness of a nurse-led physical activity intervention to decrease cardiovascular disease risk in middle-aged adults: a pilot randomized controlled study. J Vasc Nurs. 2020;38:140-148. doi: 10.1016/j.jvn.2020.05.002
3. European Association of Preventive Cardiology. HeartScore. Accessed December 23, 2022. www.heartscore.org/en_GB
4. Coghill N, Cooper AR. The effect of a home-based walking program on risk factors for coronary heart disease in hypercholesterolaemic men: a randomized controlled trial. Prev Med. 2008; 46:545-551. doi: 10.1016/j.ypmed.2008.01.002
5. Kraus WE, Houmard JA, Duscha BD, et al. Effects of the amount and intensity of exercise on plasma lipoproteins. N Engl J Med. 2002;347:1483-1492. doi: 10.1056/NEJMoa020194
6. US Department of Health and Human Services. Physical Activity Guidelines for Americans, 2nd edition. Washington, DC: US Department of Health and Human Services; 2018. Accessed December 23, 2022. https://health.gov/sites/default/files/2019-09/Physical_Activity_Guidelines_2nd_edition.pdf
7. American Heart Association. Recommendations for physical activity in adults and kids. Accessed December 23, 2022. www.heart.org/en/healthy-living/fitness/fitness-basics/aha-recs-for-physical-activity-in-adults
8. American College of Sports Medicine. Trending topic: physical activity guidelines. Accessed December 23, 2022. www.acsm.org/education-resources/trending-topics-resources/physical-activity-guidelines
EVIDENCE-BASED ANSWER:
Minimally. Regular moderate- intensity walking for a period of 4 or more weeks minimally decreased total cholesterol (TC) and low-density lipoprotein (LDL) levels by about 7 mg/dL in women with overweight or obesity (strength of recommendation [SOR]: C, systematic review and meta-analysis on disease-oriented evidence). For adults ages 40 to 65 years, regular walking for 3 or more months inconsistently affected cholesterol and triglyceride levels (SOR: C, based on 3 randomized controlled trials [RCTs] with disease-oriented evidence).
Best Practices in HR+/HER2- Breast Cancer
Two out of three patients diagnosed with breast cancer are found to have the HR+/HER2- subtype, and over time, at least one third of patients with this subtype develop metastatic disease. But recent advances in the treatment of both early and metastatic HR+/HER2- disease have resulted in significant improvement in outcomes.
In this panel ReCAP, Drs Kathy Miller from Indiana University and Alexandra Thomas from Wake Forest University review how progress in treatment has affected outcomes across all stages of HR+/HER2- breast cancer.
They report that the use of trastuzumab deruxtecan in the metastatic setting improves overall survival. They also discuss how adjuvant use of abemaciclib in early-stage breast cancer improves disease-free survival, as evidenced by the latest updates of the monarchE trial, now providing 4 years of follow-up.
--
Ballvé-Lantero Professor of Oncology, Indiana University School of Medicine, Indianapolis, Indiana
Kathy D. Miller, MD, has disclosed the following relevant financial relationships:
Serve on independent Data Monitoring Committees for ongoing trials for: Merck; Genentech/Roche; AstraZeneca; Celcuity
Two out of three patients diagnosed with breast cancer are found to have the HR+/HER2- subtype, and over time, at least one third of patients with this subtype develop metastatic disease. But recent advances in the treatment of both early and metastatic HR+/HER2- disease have resulted in significant improvement in outcomes.
In this panel ReCAP, Drs Kathy Miller from Indiana University and Alexandra Thomas from Wake Forest University review how progress in treatment has affected outcomes across all stages of HR+/HER2- breast cancer.
They report that the use of trastuzumab deruxtecan in the metastatic setting improves overall survival. They also discuss how adjuvant use of abemaciclib in early-stage breast cancer improves disease-free survival, as evidenced by the latest updates of the monarchE trial, now providing 4 years of follow-up.
--
Ballvé-Lantero Professor of Oncology, Indiana University School of Medicine, Indianapolis, Indiana
Kathy D. Miller, MD, has disclosed the following relevant financial relationships:
Serve on independent Data Monitoring Committees for ongoing trials for: Merck; Genentech/Roche; AstraZeneca; Celcuity
Two out of three patients diagnosed with breast cancer are found to have the HR+/HER2- subtype, and over time, at least one third of patients with this subtype develop metastatic disease. But recent advances in the treatment of both early and metastatic HR+/HER2- disease have resulted in significant improvement in outcomes.
In this panel ReCAP, Drs Kathy Miller from Indiana University and Alexandra Thomas from Wake Forest University review how progress in treatment has affected outcomes across all stages of HR+/HER2- breast cancer.
They report that the use of trastuzumab deruxtecan in the metastatic setting improves overall survival. They also discuss how adjuvant use of abemaciclib in early-stage breast cancer improves disease-free survival, as evidenced by the latest updates of the monarchE trial, now providing 4 years of follow-up.
--
Ballvé-Lantero Professor of Oncology, Indiana University School of Medicine, Indianapolis, Indiana
Kathy D. Miller, MD, has disclosed the following relevant financial relationships:
Serve on independent Data Monitoring Committees for ongoing trials for: Merck; Genentech/Roche; AstraZeneca; Celcuity

Commentary: HER2+-targeted therapy, ovarian suppression, and adjuvant therapy in breast cancer, February 2023
The elevated risk for recurrence in young women with HR+ early breast cancer highlights the importance of aggressive endocrine therapy in the majority of patients in this population. Examples of approaches to maximize endocrine therapy benefit include the addition of ovarian suppression to either tamoxifen or an aromatase inhibitor (AI) as well as an extended duration of adjuvant endocrine therapy.4,5 Among 3047 premenopausal women included in SOFT study, at 12 years follow-up, the addition of ovarian function suppression (OFS) to tamoxifen significantly improved disease-free survival (DFS) compared with tamoxifen alone (hazard ratio 0.82; P = .03) with a more pronounced DFS benefit with exemestane plus OFS compared with tamoxifen (hazard ratio 0.69) (Francis et al). In the HER2- subgroup, those who received prior chemotherapy had 12-year OFS rates of 78.8% with tamoxifen, 81.1% with tamoxifen plus OFS and 84.4% with exemestane plus OFS. Furthermore, in the HER2- subgroup, women younger than 35 years had absolute improvements in 12-year OS of 9.1% with tamoxifen plus OFS and 16.5% with exemestane plus OFS compared with tamoxifen. These updated results provide further support for OFS added to tamoxifen or an AI (with more benefit seen with an AI) in the treatment of HR+ early breast cancer in young women who are at high risk for recurrence. Longer follow-up will be important to better define the treatment effect considering recurrence patterns for this subtype of breast cancer.
Various guidelines recommend the use of adjuvant bisphosphonates for postmenopausal patients with early breast cancer on the basis of disease-free and bone metastasis-free survival benefits.6 A regimen of zolendronic acid every 6 months for 3 years is commonly used in clinical practice. A substudy of ABCSG-12, including 725 premenopausal patients with HR+ early breast cancer on ovarian suppression randomly assigned to receive tamoxifen or anastrozole with or without zolendronic acid every 6 months, investigated the effect of shorter duration of bisphosphonate therapy on breast cancer outcomes (Beltran-Bless et al). After a median follow-up of 96 months, there was no statistically significant difference in DFS (hazard ratio 0.88; log-rank P = .642) or OS (stratified hazard ratio 1.16; log-rank P = .796) between patients who received ≤6 or ≥7 infusions. Rates of adverse events were increased in the patients who received ≥7 or ≤6 infusions (arthralgia, 20.1% vs 12.4%; nausea, 12.8% vs 7.3%; bone pain, 41.6% vs 34.9%). Modifications to adjuvant breast cancer regimens that can provide more ease for patients with less toxicity while maintaining efficacy are greatly desired to simultaneously support quality of life and disease outcomes.
Additional References
- Swain SM, Baselga J, Kim SB, et al; for the CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med. 2015;372:724-734. Doi: 10.1056/NEJMoa1413513
- Miles D, Ciruelos E, Schneeweiss A, et al; for the PERUSE investigators. Final results from the PERUSE study of first-line pertuzumab plus trastuzumab plus a taxane for HER2-positive locally recurrent or metastatic breast cancer, with a multivariable approach to guide prognostication. Ann Oncol. 2021;32:1245-1255. Doi: 10.1016/j.annonc.2021.06.024
- Hua X, Bi X-W, Zhao J-L, et al; for the South China Breast Cancer Group (SCBCG). Trastuzumab plus endocrine therapy or chemotherapy as first-line treatment for patients with hormone receptor-positive and HER2-positive metastatic breast cancer (SYSUCC-002). Clin Cancer Res. 2022;28:637-645. Doi: 10.1158/1078-0432.CCR-21-3435
- Kim H-A, Lee JW, Nam SJ, et al; for the Korean Breast Cancer Study Group. Adding ovarian suppression to tamoxifen for premenopausal breast Cancer: a randomized phase III trial. J Clin Oncol. 2020;38:434-443. Doi: 10.1200/JCO.19.00126
- Davies C, Pan H, Godwin J, et al; for the Adjuvant Tamoxifen: Longer Against Shorter (ATLAS) Collaborative Group. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. Doi: 10.1016/S0140-6736(12)61963-1
- Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Adjuvant bisphosphonate treatment in early breast cancer: meta-analyses of individual patient data from randomised trials. Lancet. 2015;386:1353-1361. Doi: 10.1016/S0140-6736(15)60908-4
The elevated risk for recurrence in young women with HR+ early breast cancer highlights the importance of aggressive endocrine therapy in the majority of patients in this population. Examples of approaches to maximize endocrine therapy benefit include the addition of ovarian suppression to either tamoxifen or an aromatase inhibitor (AI) as well as an extended duration of adjuvant endocrine therapy.4,5 Among 3047 premenopausal women included in SOFT study, at 12 years follow-up, the addition of ovarian function suppression (OFS) to tamoxifen significantly improved disease-free survival (DFS) compared with tamoxifen alone (hazard ratio 0.82; P = .03) with a more pronounced DFS benefit with exemestane plus OFS compared with tamoxifen (hazard ratio 0.69) (Francis et al). In the HER2- subgroup, those who received prior chemotherapy had 12-year OFS rates of 78.8% with tamoxifen, 81.1% with tamoxifen plus OFS and 84.4% with exemestane plus OFS. Furthermore, in the HER2- subgroup, women younger than 35 years had absolute improvements in 12-year OS of 9.1% with tamoxifen plus OFS and 16.5% with exemestane plus OFS compared with tamoxifen. These updated results provide further support for OFS added to tamoxifen or an AI (with more benefit seen with an AI) in the treatment of HR+ early breast cancer in young women who are at high risk for recurrence. Longer follow-up will be important to better define the treatment effect considering recurrence patterns for this subtype of breast cancer.
Various guidelines recommend the use of adjuvant bisphosphonates for postmenopausal patients with early breast cancer on the basis of disease-free and bone metastasis-free survival benefits.6 A regimen of zolendronic acid every 6 months for 3 years is commonly used in clinical practice. A substudy of ABCSG-12, including 725 premenopausal patients with HR+ early breast cancer on ovarian suppression randomly assigned to receive tamoxifen or anastrozole with or without zolendronic acid every 6 months, investigated the effect of shorter duration of bisphosphonate therapy on breast cancer outcomes (Beltran-Bless et al). After a median follow-up of 96 months, there was no statistically significant difference in DFS (hazard ratio 0.88; log-rank P = .642) or OS (stratified hazard ratio 1.16; log-rank P = .796) between patients who received ≤6 or ≥7 infusions. Rates of adverse events were increased in the patients who received ≥7 or ≤6 infusions (arthralgia, 20.1% vs 12.4%; nausea, 12.8% vs 7.3%; bone pain, 41.6% vs 34.9%). Modifications to adjuvant breast cancer regimens that can provide more ease for patients with less toxicity while maintaining efficacy are greatly desired to simultaneously support quality of life and disease outcomes.
Additional References
- Swain SM, Baselga J, Kim SB, et al; for the CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med. 2015;372:724-734. Doi: 10.1056/NEJMoa1413513
- Miles D, Ciruelos E, Schneeweiss A, et al; for the PERUSE investigators. Final results from the PERUSE study of first-line pertuzumab plus trastuzumab plus a taxane for HER2-positive locally recurrent or metastatic breast cancer, with a multivariable approach to guide prognostication. Ann Oncol. 2021;32:1245-1255. Doi: 10.1016/j.annonc.2021.06.024
- Hua X, Bi X-W, Zhao J-L, et al; for the South China Breast Cancer Group (SCBCG). Trastuzumab plus endocrine therapy or chemotherapy as first-line treatment for patients with hormone receptor-positive and HER2-positive metastatic breast cancer (SYSUCC-002). Clin Cancer Res. 2022;28:637-645. Doi: 10.1158/1078-0432.CCR-21-3435
- Kim H-A, Lee JW, Nam SJ, et al; for the Korean Breast Cancer Study Group. Adding ovarian suppression to tamoxifen for premenopausal breast Cancer: a randomized phase III trial. J Clin Oncol. 2020;38:434-443. Doi: 10.1200/JCO.19.00126
- Davies C, Pan H, Godwin J, et al; for the Adjuvant Tamoxifen: Longer Against Shorter (ATLAS) Collaborative Group. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. Doi: 10.1016/S0140-6736(12)61963-1
- Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Adjuvant bisphosphonate treatment in early breast cancer: meta-analyses of individual patient data from randomised trials. Lancet. 2015;386:1353-1361. Doi: 10.1016/S0140-6736(15)60908-4
The elevated risk for recurrence in young women with HR+ early breast cancer highlights the importance of aggressive endocrine therapy in the majority of patients in this population. Examples of approaches to maximize endocrine therapy benefit include the addition of ovarian suppression to either tamoxifen or an aromatase inhibitor (AI) as well as an extended duration of adjuvant endocrine therapy.4,5 Among 3047 premenopausal women included in SOFT study, at 12 years follow-up, the addition of ovarian function suppression (OFS) to tamoxifen significantly improved disease-free survival (DFS) compared with tamoxifen alone (hazard ratio 0.82; P = .03) with a more pronounced DFS benefit with exemestane plus OFS compared with tamoxifen (hazard ratio 0.69) (Francis et al). In the HER2- subgroup, those who received prior chemotherapy had 12-year OFS rates of 78.8% with tamoxifen, 81.1% with tamoxifen plus OFS and 84.4% with exemestane plus OFS. Furthermore, in the HER2- subgroup, women younger than 35 years had absolute improvements in 12-year OS of 9.1% with tamoxifen plus OFS and 16.5% with exemestane plus OFS compared with tamoxifen. These updated results provide further support for OFS added to tamoxifen or an AI (with more benefit seen with an AI) in the treatment of HR+ early breast cancer in young women who are at high risk for recurrence. Longer follow-up will be important to better define the treatment effect considering recurrence patterns for this subtype of breast cancer.
Various guidelines recommend the use of adjuvant bisphosphonates for postmenopausal patients with early breast cancer on the basis of disease-free and bone metastasis-free survival benefits.6 A regimen of zolendronic acid every 6 months for 3 years is commonly used in clinical practice. A substudy of ABCSG-12, including 725 premenopausal patients with HR+ early breast cancer on ovarian suppression randomly assigned to receive tamoxifen or anastrozole with or without zolendronic acid every 6 months, investigated the effect of shorter duration of bisphosphonate therapy on breast cancer outcomes (Beltran-Bless et al). After a median follow-up of 96 months, there was no statistically significant difference in DFS (hazard ratio 0.88; log-rank P = .642) or OS (stratified hazard ratio 1.16; log-rank P = .796) between patients who received ≤6 or ≥7 infusions. Rates of adverse events were increased in the patients who received ≥7 or ≤6 infusions (arthralgia, 20.1% vs 12.4%; nausea, 12.8% vs 7.3%; bone pain, 41.6% vs 34.9%). Modifications to adjuvant breast cancer regimens that can provide more ease for patients with less toxicity while maintaining efficacy are greatly desired to simultaneously support quality of life and disease outcomes.
Additional References
- Swain SM, Baselga J, Kim SB, et al; for the CLEOPATRA Study Group. Pertuzumab, trastuzumab, and docetaxel in HER2-positive metastatic breast cancer. N Engl J Med. 2015;372:724-734. Doi: 10.1056/NEJMoa1413513
- Miles D, Ciruelos E, Schneeweiss A, et al; for the PERUSE investigators. Final results from the PERUSE study of first-line pertuzumab plus trastuzumab plus a taxane for HER2-positive locally recurrent or metastatic breast cancer, with a multivariable approach to guide prognostication. Ann Oncol. 2021;32:1245-1255. Doi: 10.1016/j.annonc.2021.06.024
- Hua X, Bi X-W, Zhao J-L, et al; for the South China Breast Cancer Group (SCBCG). Trastuzumab plus endocrine therapy or chemotherapy as first-line treatment for patients with hormone receptor-positive and HER2-positive metastatic breast cancer (SYSUCC-002). Clin Cancer Res. 2022;28:637-645. Doi: 10.1158/1078-0432.CCR-21-3435
- Kim H-A, Lee JW, Nam SJ, et al; for the Korean Breast Cancer Study Group. Adding ovarian suppression to tamoxifen for premenopausal breast Cancer: a randomized phase III trial. J Clin Oncol. 2020;38:434-443. Doi: 10.1200/JCO.19.00126
- Davies C, Pan H, Godwin J, et al; for the Adjuvant Tamoxifen: Longer Against Shorter (ATLAS) Collaborative Group. Long-term effects of continuing adjuvant tamoxifen to 10 years versus stopping at 5 years after diagnosis of oestrogen receptor-positive breast cancer: ATLAS, a randomised trial. Lancet. 2013;381:805-816. Doi: 10.1016/S0140-6736(12)61963-1
- Early Breast Cancer Trialists' Collaborative Group (EBCTCG). Adjuvant bisphosphonate treatment in early breast cancer: meta-analyses of individual patient data from randomised trials. Lancet. 2015;386:1353-1361. Doi: 10.1016/S0140-6736(15)60908-4