User login
Official Newspaper of the American College of Surgeons
Parents seek easily understood public reporting of cardiac outcome measures
HOUSTON – Parents of children with congenital heart disease cite survival statistics, surgeon-specific experience, and complication rates as the three most important congenital heart surgery outcome measures to report publicly, results from a large survey show.
“Recently, an increasing demand for the public reporting of pediatric heart surgery outcomes has led to the development of several different reporting schemes, including a hospital star rating system and procedure-specific mortality data tables for the Society of Thoracic Surgeons benchmark operations,” study investigator Mallory L. Irons, MD, said during a press briefing at the annual meeting of the Society of Thoracic Surgeons. “However, despite the availability of these reporting schemes, there remain unanswered questions about the optimal format and content of public reporting for pediatric heart surgery outcomes.”
“These procedures have been determined to represent more homogenous patient groups,” she said. “How data [are] presented may be just as important as the data itself. Noticeably absent from these frameworks is parent input regarding the information that may be necessary to make an informed choice about their child’s care.
“Failure to consider this perspective may contribute to suboptimal reporting and misunderstanding of the data presented. The goal of the current study was to identify parent preferences regarding the content of pediatric heart surgery outcomes reports, specifically with respect to the type of data that is included as well as the optimal format for presentation of this data.”
She and her associates developed a 43-question survey for 1,862 parents of children born with heart defects. The parents were invited to participate through member lists from patient advocacy groups Mended Little Hearts, the Pediatric Congenital Heart Association, and Sisters by Heart, and from a cohort of patients who underwent surgical correction for an STS benchmark procedure at the Children’s Hospital of Philadelphia after Jan. 1, 2007.
Of the 1,862 parents, 1,281 (69%) provided complete responses for inclusion in the final analysis. The mean age of respondents was 37 years, 92% were mothers of children with congenital heart disease, and 92% were white. “Most reported household incomes in excess of $65,000 per year, but lower income households were also significantly represented,” Dr. Irons added. More than half of the children (57%) were diagnosed with CHD prenatally, 63% underwent an initial repair in the neonatal period, and 60% of families were referred to a cardiac surgical center by a physician, while 23% were transferred from their birth hospital.
When the respondents were asked to rank categories of outcome measures or other types of information to include in an optimal public reporting scheme on a scale of 1 (most important) to 8 (least important), they identified survival statistics, surgeons’ experience with congenital heart surgery, and complication rates as the three most important. These preferences were stable across differences in education levels, household incomes, and race or ethnicity, Dr. Irons said.
Presented with three display formats for hospital-specific mortality rates, most parents (89%) identified a numerical procedure-based approach as the best format, and more than half (60%) identified the hospital star rating system as the worst potential format to display mortality data. These preferences also were stable across differences in education levels, household incomes, and race or ethnicity.
Dr. Irons acknowledged certain limitations of the study, including its retrospective design, and the fact that it lacked input from parents who do not have Internet access. “Similarly, the availability of the survey in English only prevents non-native English speakers from contributing,” she said. “Finally, while we have identified what types of information parents want, we must acknowledge that small case numbers limit the presentation of certain types of data, such as surgeon-specific outcomes, in a statistically meaningful way.”
In her opinion, an optimal reporting system requires a common database in which to collect and analyze data, a robust methodology for risk adjustment, and a way to compare performance across a field that is inherently heterogeneous.
“The ideal public reporting system should be valid as well as easily understood by all stakeholders,” Dr. Irons concluded. “We must recognize that just because parents want certain data, [it] does not mean that we can provide it in a meaningful, statistically valid way. Perhaps the most important takeaway from our study is the importance of involving parents and other stakeholders in the design and planning of methodologies for public reporting of congenital heart surgery outcomes. Ultimately, the optimal platform will represent a melding of what parents want, and what is statistically meaningful and valid.”
Jeffrey P. Jacobs, MD, a pediatric heart surgeon at Johns Hopkins University, Baltimore, who also chairs the STS Workforce on National Databases, characterized the study as “an extremely important paper that examines the format and structure of how it might be best to share information about cardiac surgical outcomes with patients and their families.” He said the STS “has studied multiple different strategies, and currently we use a method where we report outcomes using a categorical system of star ratings and augment that with detailed specific data with point estimates for confidence intervals. We continue to study and explore better ways to share this information with our patients and with their families.”
Dr. Irons reported having no financial disclosures.
HOUSTON – Parents of children with congenital heart disease cite survival statistics, surgeon-specific experience, and complication rates as the three most important congenital heart surgery outcome measures to report publicly, results from a large survey show.
“Recently, an increasing demand for the public reporting of pediatric heart surgery outcomes has led to the development of several different reporting schemes, including a hospital star rating system and procedure-specific mortality data tables for the Society of Thoracic Surgeons benchmark operations,” study investigator Mallory L. Irons, MD, said during a press briefing at the annual meeting of the Society of Thoracic Surgeons. “However, despite the availability of these reporting schemes, there remain unanswered questions about the optimal format and content of public reporting for pediatric heart surgery outcomes.”
“These procedures have been determined to represent more homogenous patient groups,” she said. “How data [are] presented may be just as important as the data itself. Noticeably absent from these frameworks is parent input regarding the information that may be necessary to make an informed choice about their child’s care.
“Failure to consider this perspective may contribute to suboptimal reporting and misunderstanding of the data presented. The goal of the current study was to identify parent preferences regarding the content of pediatric heart surgery outcomes reports, specifically with respect to the type of data that is included as well as the optimal format for presentation of this data.”
She and her associates developed a 43-question survey for 1,862 parents of children born with heart defects. The parents were invited to participate through member lists from patient advocacy groups Mended Little Hearts, the Pediatric Congenital Heart Association, and Sisters by Heart, and from a cohort of patients who underwent surgical correction for an STS benchmark procedure at the Children’s Hospital of Philadelphia after Jan. 1, 2007.
Of the 1,862 parents, 1,281 (69%) provided complete responses for inclusion in the final analysis. The mean age of respondents was 37 years, 92% were mothers of children with congenital heart disease, and 92% were white. “Most reported household incomes in excess of $65,000 per year, but lower income households were also significantly represented,” Dr. Irons added. More than half of the children (57%) were diagnosed with CHD prenatally, 63% underwent an initial repair in the neonatal period, and 60% of families were referred to a cardiac surgical center by a physician, while 23% were transferred from their birth hospital.
When the respondents were asked to rank categories of outcome measures or other types of information to include in an optimal public reporting scheme on a scale of 1 (most important) to 8 (least important), they identified survival statistics, surgeons’ experience with congenital heart surgery, and complication rates as the three most important. These preferences were stable across differences in education levels, household incomes, and race or ethnicity, Dr. Irons said.
Presented with three display formats for hospital-specific mortality rates, most parents (89%) identified a numerical procedure-based approach as the best format, and more than half (60%) identified the hospital star rating system as the worst potential format to display mortality data. These preferences also were stable across differences in education levels, household incomes, and race or ethnicity.
Dr. Irons acknowledged certain limitations of the study, including its retrospective design, and the fact that it lacked input from parents who do not have Internet access. “Similarly, the availability of the survey in English only prevents non-native English speakers from contributing,” she said. “Finally, while we have identified what types of information parents want, we must acknowledge that small case numbers limit the presentation of certain types of data, such as surgeon-specific outcomes, in a statistically meaningful way.”
In her opinion, an optimal reporting system requires a common database in which to collect and analyze data, a robust methodology for risk adjustment, and a way to compare performance across a field that is inherently heterogeneous.
“The ideal public reporting system should be valid as well as easily understood by all stakeholders,” Dr. Irons concluded. “We must recognize that just because parents want certain data, [it] does not mean that we can provide it in a meaningful, statistically valid way. Perhaps the most important takeaway from our study is the importance of involving parents and other stakeholders in the design and planning of methodologies for public reporting of congenital heart surgery outcomes. Ultimately, the optimal platform will represent a melding of what parents want, and what is statistically meaningful and valid.”
Jeffrey P. Jacobs, MD, a pediatric heart surgeon at Johns Hopkins University, Baltimore, who also chairs the STS Workforce on National Databases, characterized the study as “an extremely important paper that examines the format and structure of how it might be best to share information about cardiac surgical outcomes with patients and their families.” He said the STS “has studied multiple different strategies, and currently we use a method where we report outcomes using a categorical system of star ratings and augment that with detailed specific data with point estimates for confidence intervals. We continue to study and explore better ways to share this information with our patients and with their families.”
Dr. Irons reported having no financial disclosures.
HOUSTON – Parents of children with congenital heart disease cite survival statistics, surgeon-specific experience, and complication rates as the three most important congenital heart surgery outcome measures to report publicly, results from a large survey show.
“Recently, an increasing demand for the public reporting of pediatric heart surgery outcomes has led to the development of several different reporting schemes, including a hospital star rating system and procedure-specific mortality data tables for the Society of Thoracic Surgeons benchmark operations,” study investigator Mallory L. Irons, MD, said during a press briefing at the annual meeting of the Society of Thoracic Surgeons. “However, despite the availability of these reporting schemes, there remain unanswered questions about the optimal format and content of public reporting for pediatric heart surgery outcomes.”
“These procedures have been determined to represent more homogenous patient groups,” she said. “How data [are] presented may be just as important as the data itself. Noticeably absent from these frameworks is parent input regarding the information that may be necessary to make an informed choice about their child’s care.
“Failure to consider this perspective may contribute to suboptimal reporting and misunderstanding of the data presented. The goal of the current study was to identify parent preferences regarding the content of pediatric heart surgery outcomes reports, specifically with respect to the type of data that is included as well as the optimal format for presentation of this data.”
She and her associates developed a 43-question survey for 1,862 parents of children born with heart defects. The parents were invited to participate through member lists from patient advocacy groups Mended Little Hearts, the Pediatric Congenital Heart Association, and Sisters by Heart, and from a cohort of patients who underwent surgical correction for an STS benchmark procedure at the Children’s Hospital of Philadelphia after Jan. 1, 2007.
Of the 1,862 parents, 1,281 (69%) provided complete responses for inclusion in the final analysis. The mean age of respondents was 37 years, 92% were mothers of children with congenital heart disease, and 92% were white. “Most reported household incomes in excess of $65,000 per year, but lower income households were also significantly represented,” Dr. Irons added. More than half of the children (57%) were diagnosed with CHD prenatally, 63% underwent an initial repair in the neonatal period, and 60% of families were referred to a cardiac surgical center by a physician, while 23% were transferred from their birth hospital.
When the respondents were asked to rank categories of outcome measures or other types of information to include in an optimal public reporting scheme on a scale of 1 (most important) to 8 (least important), they identified survival statistics, surgeons’ experience with congenital heart surgery, and complication rates as the three most important. These preferences were stable across differences in education levels, household incomes, and race or ethnicity, Dr. Irons said.
Presented with three display formats for hospital-specific mortality rates, most parents (89%) identified a numerical procedure-based approach as the best format, and more than half (60%) identified the hospital star rating system as the worst potential format to display mortality data. These preferences also were stable across differences in education levels, household incomes, and race or ethnicity.
Dr. Irons acknowledged certain limitations of the study, including its retrospective design, and the fact that it lacked input from parents who do not have Internet access. “Similarly, the availability of the survey in English only prevents non-native English speakers from contributing,” she said. “Finally, while we have identified what types of information parents want, we must acknowledge that small case numbers limit the presentation of certain types of data, such as surgeon-specific outcomes, in a statistically meaningful way.”
In her opinion, an optimal reporting system requires a common database in which to collect and analyze data, a robust methodology for risk adjustment, and a way to compare performance across a field that is inherently heterogeneous.
“The ideal public reporting system should be valid as well as easily understood by all stakeholders,” Dr. Irons concluded. “We must recognize that just because parents want certain data, [it] does not mean that we can provide it in a meaningful, statistically valid way. Perhaps the most important takeaway from our study is the importance of involving parents and other stakeholders in the design and planning of methodologies for public reporting of congenital heart surgery outcomes. Ultimately, the optimal platform will represent a melding of what parents want, and what is statistically meaningful and valid.”
Jeffrey P. Jacobs, MD, a pediatric heart surgeon at Johns Hopkins University, Baltimore, who also chairs the STS Workforce on National Databases, characterized the study as “an extremely important paper that examines the format and structure of how it might be best to share information about cardiac surgical outcomes with patients and their families.” He said the STS “has studied multiple different strategies, and currently we use a method where we report outcomes using a categorical system of star ratings and augment that with detailed specific data with point estimates for confidence intervals. We continue to study and explore better ways to share this information with our patients and with their families.”
Dr. Irons reported having no financial disclosures.
AT THE STS ANNUAL MEETING
Key clinical point:
Major finding: When parents of children with congenital heart disease were asked to rank categories of outcome measures or other types of information to include in an optimal public reporting scheme, they identified survival statistics, surgeons’ experience with congenital heart surgery, and complication rates as the three most important
Data source: A retrospective survey of 1,862 parents of children born with heart defects.
Disclosures: Dr. Irons reported having no financial disclosures.
Triclosan sutures halve surgical site infections in children
The use of triclosan-impregnated sutures reduced by half the incidence of surgical site infections in children, a large randomized study has determined.
Overall, the antibiotic-treated sutures cut the number of these infections by 52%, but they were particularly effective in reducing the risk of deep surgical site infections (SSIs), Marjo Renko, MD, wrote (Lancet Infect Dis. 2017;17[1]:50-7).
The study was conducted in clean wounds in healthy children and in a center that already had a very low rate of surgical site infections (just 5%) – showing that improvement is possible even in optimal care settings, wrote Dr. Renko, of the University of Oulu, Finland, and her colleagues.
“This randomized, controlled study shows that even in low-risk settings, where other prophylactic measures are available to use, triclosan-containing sutures effectively prevented the occurrence of SSIs in children,” the team wrote.
The study cohort comprised 1,633 children aged 7-17 who underwent surgery at a single Finnish hospital from 2010-2014. Most were there for planned surgery (87%); the remainder had emergency surgery. The most common surgical site was musculoskeletal (40%), followed by abdominal wall surgery (about 25%), and urogenital surgery (about 13%). The rest were intraabdominal or procedures on the nervous system, chest, and skin or subcutaneous tissue.
The children were randomized to either plain or triclosan-impregnated sutures. The primary outcome was the occurrence of a superficial or deep surgical site infection, based on Centers for Disease Control and Prevention criteria. The procedures were performed by 69 surgeons.
In a modified intent-to-treat analysis, a surgical site infection occurred in 3% of the triclosan-suture group (20 children) and in 5% of the control suture group (42 children). In the control group, these infections were most often of chest incisions (15%), followed by skin incisions (10%) and nervous system, intraabdominal, and musculoskeletal incisions (8% each). In the triclosan group, the most common site of infection was skin (10%), followed by musculoskeletal (4%), nervous system (2%), and urogenital and abdominal wall incisions (1% each).
Compared with control sutures, triclosan sutures reduced the overall risk of a surgical site infection by 52% (relative risk, 0.48; 95% confidence interval, 0.28-0.80). The number needed to treat to avoid one infection was 36.
The sutures were significantly more effective in reducing deep infections than superficial infections. Superficial infections occurred in 2% of the triclosan group (17) and 4% of the control group (28) – a risk reduction of 39% (RR, 0.61; 95% CI, 0.34-1.09) Deep infections occurred in less than 1% of the triclosan group (3) and 2% of the control group (14) – a risk reduction of 79% (RR, 0.21’ CI, 0.07-0.66).
Infections were associated with an increased incidence of wound dehiscence in the control group (6% vs. 4%), the need for additional antimicrobial agents (7% vs. 2%), and wound revisions (2% vs. less than 1%). Children in the control group also had more outpatient visits (8% vs. 4%) and were more often readmitted because of their infection (2% vs. 1%).
The authors noted that triclosan, in the setting of increased household use, “has raised concerns about the toxic effects of the drug on the human body. Observational studies have reported associations between triclosan exposures and altered thyroid hormone levels, body mass index, and waist circumference.”
Two Norwegian studies found that the drug was associated with inhalation allergies and seasonal allergies.
“Because of the agent’s suspected toxicity and to prevent further development of resistant bacteria, use of triclosan should be restricted and reserved only for medical procedures with adequate evidence,” they noted. However, “SSIs cause much morbidity and mortality after surgical procedures, and economic evaluations recommend the use of triclosan-containing material.”
Dr. Renko received grants from the Alma and K.A. Snellman Foundation, the Finnish Medical Foundation, and the Foundation for Pediatric Research.
[email protected]
On Twitter @Alz_Gal
The study by Dr. Marjo Renko and her colleagues is impressive in its sheer numbers, if not so much in its findings, Felix J. Hüttner, MD, and Markus K. Diener, MD, wrote in an accompanying editorial (Lancet Infect Dis. 2017;17[1]:3-4).
“We congratulate the authors on successfully doing a pragmatic, large-scale trial in a difficult setting; randomized controlled trials in children are known to pose specific challenges to researchers. However, the monocenter design raises some concerns about the generalizability of the results.”
Single-center trials can overestimate treatment effects, the colleagues noted. Dr. Renko’s conclusions don’t line up with their own metaanalysis of triclosan-containing sutures for abdominal wall closure. In it, three single-center trials found in favor of the triclosan sutures, but two multicenter trials did not.
The variation in infection rates in each type of surgery is a clue to the difficulty of a one-size-fits-all intervention like the treated sutures. “The differences between the intervention group and the control group vary widely by surgery type – for example, 0% versus 15% for thoracic surgery, compared with 1% versus 1% for surgery of the urinary system and genitals. Thus, triclosan-containing sutures might only be beneficial for specific types of operations and in our opinion, it cannot be concluded that triclosan-containing sutures reduce surgical site infections in all of these indications. Future trials should focus at individual types of pediatric surgery to evaluate a potential beneficial effect.”
Dr. Hüttner and Dr. Diener are surgeons at the University of Heidelberg, Germany. Dr. Hüttner had no financial disclosures. Dr. Diener has received grants from Johnson & Johnson Medical Limited.
The study by Dr. Marjo Renko and her colleagues is impressive in its sheer numbers, if not so much in its findings, Felix J. Hüttner, MD, and Markus K. Diener, MD, wrote in an accompanying editorial (Lancet Infect Dis. 2017;17[1]:3-4).
“We congratulate the authors on successfully doing a pragmatic, large-scale trial in a difficult setting; randomized controlled trials in children are known to pose specific challenges to researchers. However, the monocenter design raises some concerns about the generalizability of the results.”
Single-center trials can overestimate treatment effects, the colleagues noted. Dr. Renko’s conclusions don’t line up with their own metaanalysis of triclosan-containing sutures for abdominal wall closure. In it, three single-center trials found in favor of the triclosan sutures, but two multicenter trials did not.
The variation in infection rates in each type of surgery is a clue to the difficulty of a one-size-fits-all intervention like the treated sutures. “The differences between the intervention group and the control group vary widely by surgery type – for example, 0% versus 15% for thoracic surgery, compared with 1% versus 1% for surgery of the urinary system and genitals. Thus, triclosan-containing sutures might only be beneficial for specific types of operations and in our opinion, it cannot be concluded that triclosan-containing sutures reduce surgical site infections in all of these indications. Future trials should focus at individual types of pediatric surgery to evaluate a potential beneficial effect.”
Dr. Hüttner and Dr. Diener are surgeons at the University of Heidelberg, Germany. Dr. Hüttner had no financial disclosures. Dr. Diener has received grants from Johnson & Johnson Medical Limited.
The study by Dr. Marjo Renko and her colleagues is impressive in its sheer numbers, if not so much in its findings, Felix J. Hüttner, MD, and Markus K. Diener, MD, wrote in an accompanying editorial (Lancet Infect Dis. 2017;17[1]:3-4).
“We congratulate the authors on successfully doing a pragmatic, large-scale trial in a difficult setting; randomized controlled trials in children are known to pose specific challenges to researchers. However, the monocenter design raises some concerns about the generalizability of the results.”
Single-center trials can overestimate treatment effects, the colleagues noted. Dr. Renko’s conclusions don’t line up with their own metaanalysis of triclosan-containing sutures for abdominal wall closure. In it, three single-center trials found in favor of the triclosan sutures, but two multicenter trials did not.
The variation in infection rates in each type of surgery is a clue to the difficulty of a one-size-fits-all intervention like the treated sutures. “The differences between the intervention group and the control group vary widely by surgery type – for example, 0% versus 15% for thoracic surgery, compared with 1% versus 1% for surgery of the urinary system and genitals. Thus, triclosan-containing sutures might only be beneficial for specific types of operations and in our opinion, it cannot be concluded that triclosan-containing sutures reduce surgical site infections in all of these indications. Future trials should focus at individual types of pediatric surgery to evaluate a potential beneficial effect.”
Dr. Hüttner and Dr. Diener are surgeons at the University of Heidelberg, Germany. Dr. Hüttner had no financial disclosures. Dr. Diener has received grants from Johnson & Johnson Medical Limited.
The use of triclosan-impregnated sutures reduced by half the incidence of surgical site infections in children, a large randomized study has determined.
Overall, the antibiotic-treated sutures cut the number of these infections by 52%, but they were particularly effective in reducing the risk of deep surgical site infections (SSIs), Marjo Renko, MD, wrote (Lancet Infect Dis. 2017;17[1]:50-7).
The study was conducted in clean wounds in healthy children and in a center that already had a very low rate of surgical site infections (just 5%) – showing that improvement is possible even in optimal care settings, wrote Dr. Renko, of the University of Oulu, Finland, and her colleagues.
“This randomized, controlled study shows that even in low-risk settings, where other prophylactic measures are available to use, triclosan-containing sutures effectively prevented the occurrence of SSIs in children,” the team wrote.
The study cohort comprised 1,633 children aged 7-17 who underwent surgery at a single Finnish hospital from 2010-2014. Most were there for planned surgery (87%); the remainder had emergency surgery. The most common surgical site was musculoskeletal (40%), followed by abdominal wall surgery (about 25%), and urogenital surgery (about 13%). The rest were intraabdominal or procedures on the nervous system, chest, and skin or subcutaneous tissue.
The children were randomized to either plain or triclosan-impregnated sutures. The primary outcome was the occurrence of a superficial or deep surgical site infection, based on Centers for Disease Control and Prevention criteria. The procedures were performed by 69 surgeons.
In a modified intent-to-treat analysis, a surgical site infection occurred in 3% of the triclosan-suture group (20 children) and in 5% of the control suture group (42 children). In the control group, these infections were most often of chest incisions (15%), followed by skin incisions (10%) and nervous system, intraabdominal, and musculoskeletal incisions (8% each). In the triclosan group, the most common site of infection was skin (10%), followed by musculoskeletal (4%), nervous system (2%), and urogenital and abdominal wall incisions (1% each).
Compared with control sutures, triclosan sutures reduced the overall risk of a surgical site infection by 52% (relative risk, 0.48; 95% confidence interval, 0.28-0.80). The number needed to treat to avoid one infection was 36.
The sutures were significantly more effective in reducing deep infections than superficial infections. Superficial infections occurred in 2% of the triclosan group (17) and 4% of the control group (28) – a risk reduction of 39% (RR, 0.61; 95% CI, 0.34-1.09) Deep infections occurred in less than 1% of the triclosan group (3) and 2% of the control group (14) – a risk reduction of 79% (RR, 0.21’ CI, 0.07-0.66).
Infections were associated with an increased incidence of wound dehiscence in the control group (6% vs. 4%), the need for additional antimicrobial agents (7% vs. 2%), and wound revisions (2% vs. less than 1%). Children in the control group also had more outpatient visits (8% vs. 4%) and were more often readmitted because of their infection (2% vs. 1%).
The authors noted that triclosan, in the setting of increased household use, “has raised concerns about the toxic effects of the drug on the human body. Observational studies have reported associations between triclosan exposures and altered thyroid hormone levels, body mass index, and waist circumference.”
Two Norwegian studies found that the drug was associated with inhalation allergies and seasonal allergies.
“Because of the agent’s suspected toxicity and to prevent further development of resistant bacteria, use of triclosan should be restricted and reserved only for medical procedures with adequate evidence,” they noted. However, “SSIs cause much morbidity and mortality after surgical procedures, and economic evaluations recommend the use of triclosan-containing material.”
Dr. Renko received grants from the Alma and K.A. Snellman Foundation, the Finnish Medical Foundation, and the Foundation for Pediatric Research.
[email protected]
On Twitter @Alz_Gal
The use of triclosan-impregnated sutures reduced by half the incidence of surgical site infections in children, a large randomized study has determined.
Overall, the antibiotic-treated sutures cut the number of these infections by 52%, but they were particularly effective in reducing the risk of deep surgical site infections (SSIs), Marjo Renko, MD, wrote (Lancet Infect Dis. 2017;17[1]:50-7).
The study was conducted in clean wounds in healthy children and in a center that already had a very low rate of surgical site infections (just 5%) – showing that improvement is possible even in optimal care settings, wrote Dr. Renko, of the University of Oulu, Finland, and her colleagues.
“This randomized, controlled study shows that even in low-risk settings, where other prophylactic measures are available to use, triclosan-containing sutures effectively prevented the occurrence of SSIs in children,” the team wrote.
The study cohort comprised 1,633 children aged 7-17 who underwent surgery at a single Finnish hospital from 2010-2014. Most were there for planned surgery (87%); the remainder had emergency surgery. The most common surgical site was musculoskeletal (40%), followed by abdominal wall surgery (about 25%), and urogenital surgery (about 13%). The rest were intraabdominal or procedures on the nervous system, chest, and skin or subcutaneous tissue.
The children were randomized to either plain or triclosan-impregnated sutures. The primary outcome was the occurrence of a superficial or deep surgical site infection, based on Centers for Disease Control and Prevention criteria. The procedures were performed by 69 surgeons.
In a modified intent-to-treat analysis, a surgical site infection occurred in 3% of the triclosan-suture group (20 children) and in 5% of the control suture group (42 children). In the control group, these infections were most often of chest incisions (15%), followed by skin incisions (10%) and nervous system, intraabdominal, and musculoskeletal incisions (8% each). In the triclosan group, the most common site of infection was skin (10%), followed by musculoskeletal (4%), nervous system (2%), and urogenital and abdominal wall incisions (1% each).
Compared with control sutures, triclosan sutures reduced the overall risk of a surgical site infection by 52% (relative risk, 0.48; 95% confidence interval, 0.28-0.80). The number needed to treat to avoid one infection was 36.
The sutures were significantly more effective in reducing deep infections than superficial infections. Superficial infections occurred in 2% of the triclosan group (17) and 4% of the control group (28) – a risk reduction of 39% (RR, 0.61; 95% CI, 0.34-1.09) Deep infections occurred in less than 1% of the triclosan group (3) and 2% of the control group (14) – a risk reduction of 79% (RR, 0.21’ CI, 0.07-0.66).
Infections were associated with an increased incidence of wound dehiscence in the control group (6% vs. 4%), the need for additional antimicrobial agents (7% vs. 2%), and wound revisions (2% vs. less than 1%). Children in the control group also had more outpatient visits (8% vs. 4%) and were more often readmitted because of their infection (2% vs. 1%).
The authors noted that triclosan, in the setting of increased household use, “has raised concerns about the toxic effects of the drug on the human body. Observational studies have reported associations between triclosan exposures and altered thyroid hormone levels, body mass index, and waist circumference.”
Two Norwegian studies found that the drug was associated with inhalation allergies and seasonal allergies.
“Because of the agent’s suspected toxicity and to prevent further development of resistant bacteria, use of triclosan should be restricted and reserved only for medical procedures with adequate evidence,” they noted. However, “SSIs cause much morbidity and mortality after surgical procedures, and economic evaluations recommend the use of triclosan-containing material.”
Dr. Renko received grants from the Alma and K.A. Snellman Foundation, the Finnish Medical Foundation, and the Foundation for Pediatric Research.
[email protected]
On Twitter @Alz_Gal
FROM LANCET INFECTIOUS DISEASES
Key clinical point:
Major finding: Overall, the sutures were associated with a 52% decrease in SSIs.
Data source: The study randomized 1,633 children undergoing surgery to the triclosan sutures or to a control suture.
Disclosures: Dr. Renko received grants from the Alma and K.A. Snellman Foundation, the Finnish Medical Foundation, and the Foundation for Pediatric Research.
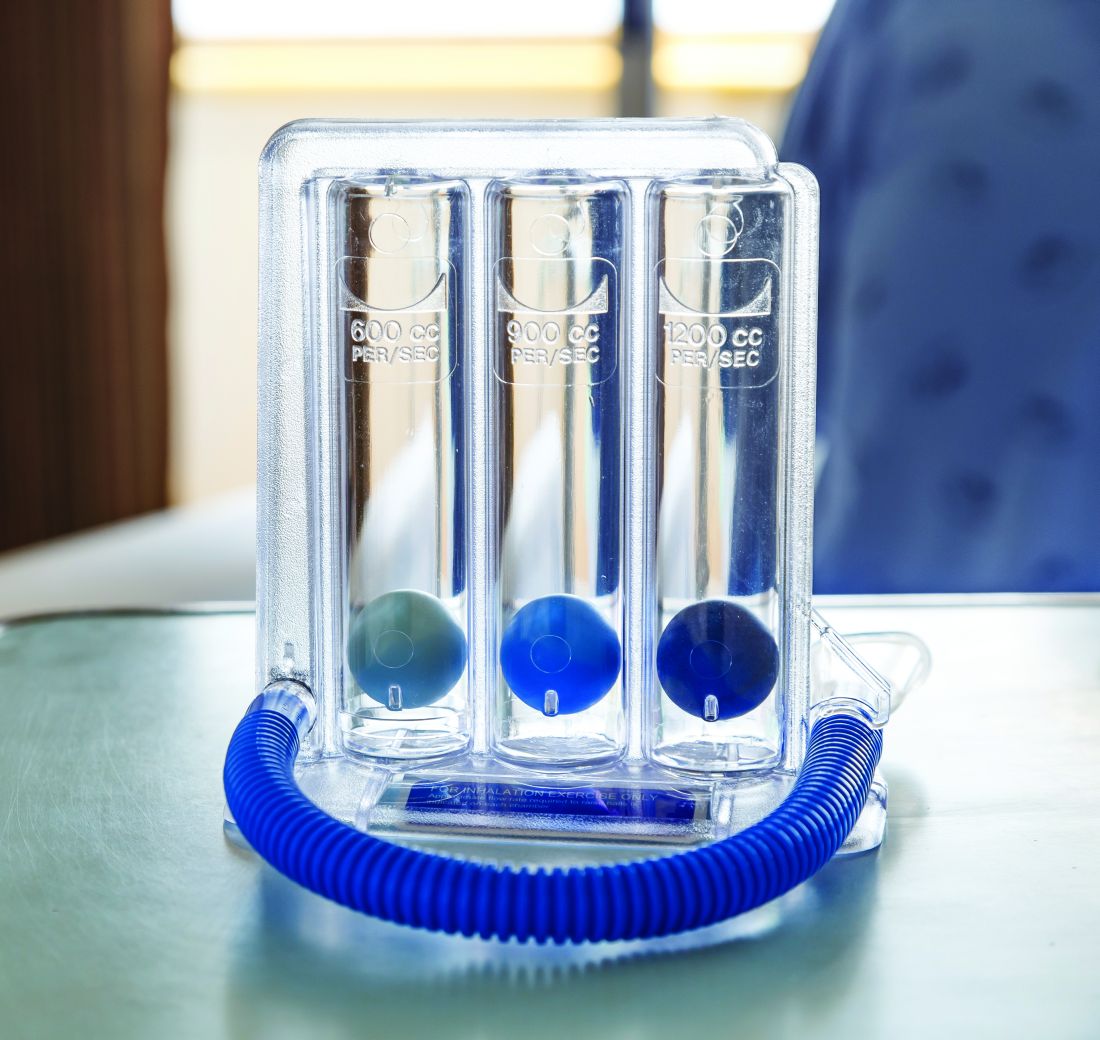
Postop incentive spirometry had minimal impact on hypoxemia in bariatric surgery patients
The effect of incentive spirometry (IS) on postoperative hypoxemia in bariatric surgery patients was found to be insignificant, according to a randomized cohort study published in JAMA Surgery.
“At present, postoperative IS is considered the standard of care and is incorporated into standardized bariatric surgery recovery protocols,” wrote the authors of the study, led by Haddon Pantel, MD, of the Lahey Hospital and Medical Center in Burlington, Mass. “However, despite the ubiquitous use of IS in the postoperative period, data on its efficacy are conflicting, and high-quality evidence is lacking.” (JAMA Surg. doi:10.1001/jamasurg.2016.4981)
A total of 224 patients were evenly randomized into one of two cohorts; one cohort received no postoperative IS and acted as the control, while the other received postoperative IS. Patients from each of these cohorts were followed up at 6, 12, and 24 hours to measure SaO2 levels as a sign of hypoxemia, which was defined as a level of under 92%.
No significant differences were observed between the two cohorts at any of the three follow-up periods in terms of SaO2 levels. At 6 hours, hypoxemia incidence rates were 11.9% in the control group and 10.4% in the IS group (P = .72). At the 12-hour follow-up, the control group registered a 5.4% incidence rate, compared with 8.2% for those receiving postoperative IS (P = .40). And finally, at 24-hour follow-up, the control group had a 3.7% rate of hypoxemia, while those in the IS cohort had a 4.6% rate (P = .73). In addition, there were no significant differences observed in the average SaO2 levels between the two cohorts (P = .99, P = .40, and P = .69 at 6, 12, and 24 hours, respectively) nor was there a significantly higher rate of pulmonary complications in one cohort versus the other (P = .24).
The authors concluded, “With health care moving toward a more evidence-based, economically driven, and environmentally sustainable field, this study adds evidence to the concept that IS should not be universally used in all patients undergoing surgery and does not appear to be necessary in elective bariatric surgical procedures.”
The study was funded by Lahey Hospital and Medical Center’s department of general surgery; the authors reported no relevant financial disclosures.
The effect of incentive spirometry (IS) on postoperative hypoxemia in bariatric surgery patients was found to be insignificant, according to a randomized cohort study published in JAMA Surgery.
“At present, postoperative IS is considered the standard of care and is incorporated into standardized bariatric surgery recovery protocols,” wrote the authors of the study, led by Haddon Pantel, MD, of the Lahey Hospital and Medical Center in Burlington, Mass. “However, despite the ubiquitous use of IS in the postoperative period, data on its efficacy are conflicting, and high-quality evidence is lacking.” (JAMA Surg. doi:10.1001/jamasurg.2016.4981)
A total of 224 patients were evenly randomized into one of two cohorts; one cohort received no postoperative IS and acted as the control, while the other received postoperative IS. Patients from each of these cohorts were followed up at 6, 12, and 24 hours to measure SaO2 levels as a sign of hypoxemia, which was defined as a level of under 92%.
No significant differences were observed between the two cohorts at any of the three follow-up periods in terms of SaO2 levels. At 6 hours, hypoxemia incidence rates were 11.9% in the control group and 10.4% in the IS group (P = .72). At the 12-hour follow-up, the control group registered a 5.4% incidence rate, compared with 8.2% for those receiving postoperative IS (P = .40). And finally, at 24-hour follow-up, the control group had a 3.7% rate of hypoxemia, while those in the IS cohort had a 4.6% rate (P = .73). In addition, there were no significant differences observed in the average SaO2 levels between the two cohorts (P = .99, P = .40, and P = .69 at 6, 12, and 24 hours, respectively) nor was there a significantly higher rate of pulmonary complications in one cohort versus the other (P = .24).
The authors concluded, “With health care moving toward a more evidence-based, economically driven, and environmentally sustainable field, this study adds evidence to the concept that IS should not be universally used in all patients undergoing surgery and does not appear to be necessary in elective bariatric surgical procedures.”
The study was funded by Lahey Hospital and Medical Center’s department of general surgery; the authors reported no relevant financial disclosures.
The effect of incentive spirometry (IS) on postoperative hypoxemia in bariatric surgery patients was found to be insignificant, according to a randomized cohort study published in JAMA Surgery.
“At present, postoperative IS is considered the standard of care and is incorporated into standardized bariatric surgery recovery protocols,” wrote the authors of the study, led by Haddon Pantel, MD, of the Lahey Hospital and Medical Center in Burlington, Mass. “However, despite the ubiquitous use of IS in the postoperative period, data on its efficacy are conflicting, and high-quality evidence is lacking.” (JAMA Surg. doi:10.1001/jamasurg.2016.4981)
A total of 224 patients were evenly randomized into one of two cohorts; one cohort received no postoperative IS and acted as the control, while the other received postoperative IS. Patients from each of these cohorts were followed up at 6, 12, and 24 hours to measure SaO2 levels as a sign of hypoxemia, which was defined as a level of under 92%.
No significant differences were observed between the two cohorts at any of the three follow-up periods in terms of SaO2 levels. At 6 hours, hypoxemia incidence rates were 11.9% in the control group and 10.4% in the IS group (P = .72). At the 12-hour follow-up, the control group registered a 5.4% incidence rate, compared with 8.2% for those receiving postoperative IS (P = .40). And finally, at 24-hour follow-up, the control group had a 3.7% rate of hypoxemia, while those in the IS cohort had a 4.6% rate (P = .73). In addition, there were no significant differences observed in the average SaO2 levels between the two cohorts (P = .99, P = .40, and P = .69 at 6, 12, and 24 hours, respectively) nor was there a significantly higher rate of pulmonary complications in one cohort versus the other (P = .24).
The authors concluded, “With health care moving toward a more evidence-based, economically driven, and environmentally sustainable field, this study adds evidence to the concept that IS should not be universally used in all patients undergoing surgery and does not appear to be necessary in elective bariatric surgical procedures.”
The study was funded by Lahey Hospital and Medical Center’s department of general surgery; the authors reported no relevant financial disclosures.
FROM JAMA SURGERY
Key clinical point:
Major finding: No significant difference in hypoxemia frequency was found between postoperative IS and control cohorts at 6, 12, and 24-hour follow-ups (P = .72, .40, and .73, respectively).
Data source: A randomized, noninferiority cohort study of 224 bariatric surgery patients during May 2015 through June 2016.
Disclosures: Study funded by Lahey Hospital and Medical Center; authors reported no relevant financial disclosures.
Endoscopic resection alone sufficed in many T1 colorectal cancers
Patients with T1 colorectal cancer might not benefit from additional surgery after endoscopic resection unless they have positive or indeterminate resection margins or high-risk histology, according to a retrospective, population-based study of 1,315 patients.
After a median follow-up of 6.6 years, the rates of colorectal cancer (CRC) recurrence were 6.2% in patients who underwent endoscopic resection only and 6.4% in patients who also had additional surgery (P = .9), reported Tim D.G. Belderbos, MD, of University Medical Center Utrecht (the Netherlands). Rates of local recurrence also were similar between these groups (4.1% and 3.7%, P = .3), he and his associates reported in the March issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2016.08.041).
Among high-risk patients, however, the rates of overall recurrence were 14% with endoscopic resection only and 7% with endoscopic resection plus additional surgery (P = .06), and the rates of local recurrence were 12% and 1%, respectively (P = .004). “Based on our study, we recommend performing additional surgery after initial endoscopic resection in cases of high-risk T1 CRC, determined by high-risk histology and/or positive resection margins,” the researchers concluded. Invasive CRCs confined to the colonic submucosa (T1 CRC) present a treatment dilemma – they are usually cured by complete endoscopic resection, but up to 13% involve lymph node metastases and need additional surgery, the investigators noted. To identify predictors of recurrence and metastasis, they studied all patients diagnosed with T1 CRC in the Southeast Netherlands from 1995 through 2011. A total of 370 patients (28%) underwent endoscopic resection only, 220 (17%) underwent endoscopic resection with additional surgery, and 725 (55%) had an initial surgical resection.
Surgery after endoscopic resection was more likely when patients had positive or doubtful resection margins (P less than .001), and this link remained significant after high-risk histology, tumor location, time period, age, sex, and comorbidities were controlled for. Endoscopic resection plus surgery did not reduce the risk of recurrence, compared with endoscopic resection only (P = .3), after the investigators accounted for age, sex, year of procedure, tumor location, and margin characteristics. Initial surgery was associated with significantly lower rates of overall and local recurrence, compared with endoscopic resection only, but the differences also lost significance in the multivariable analysis (P = .2).
Only the presence of positive resection margins significantly predicted recurrence among patients undergoing endoscopic resection (hazard ratio, 6.9; 95% confidence interval, 2.3-20.9). Positive or doubtful resection margins also predicted recurrence after initial surgery, with hazard ratios of 13.2 and 3.4, respectively. High-risk histology – that is, poor differentiation, deep submucosal invasion, or lymphangioinvasion – was significantly associated with lymph node metastasis (OR, 2.2; 95% CI, 1.3-3.7; P less than .002), but not with recurrence after resection margins were accounted for. This might result from missing histology data or the fact that patients with high-risk histology tended to undergo surgical rather than endoscopic resection, the researchers said.
They noted several other study limitations, including a lack of details about lesions and procedures. Also, endoscopic submucosal resection was not practiced in the Netherlands during the study period, they said.
The investigators did not report funding sources and had no disclosures.
Patients with T1 colorectal cancer might not benefit from additional surgery after endoscopic resection unless they have positive or indeterminate resection margins or high-risk histology, according to a retrospective, population-based study of 1,315 patients.
After a median follow-up of 6.6 years, the rates of colorectal cancer (CRC) recurrence were 6.2% in patients who underwent endoscopic resection only and 6.4% in patients who also had additional surgery (P = .9), reported Tim D.G. Belderbos, MD, of University Medical Center Utrecht (the Netherlands). Rates of local recurrence also were similar between these groups (4.1% and 3.7%, P = .3), he and his associates reported in the March issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2016.08.041).
Among high-risk patients, however, the rates of overall recurrence were 14% with endoscopic resection only and 7% with endoscopic resection plus additional surgery (P = .06), and the rates of local recurrence were 12% and 1%, respectively (P = .004). “Based on our study, we recommend performing additional surgery after initial endoscopic resection in cases of high-risk T1 CRC, determined by high-risk histology and/or positive resection margins,” the researchers concluded. Invasive CRCs confined to the colonic submucosa (T1 CRC) present a treatment dilemma – they are usually cured by complete endoscopic resection, but up to 13% involve lymph node metastases and need additional surgery, the investigators noted. To identify predictors of recurrence and metastasis, they studied all patients diagnosed with T1 CRC in the Southeast Netherlands from 1995 through 2011. A total of 370 patients (28%) underwent endoscopic resection only, 220 (17%) underwent endoscopic resection with additional surgery, and 725 (55%) had an initial surgical resection.
Surgery after endoscopic resection was more likely when patients had positive or doubtful resection margins (P less than .001), and this link remained significant after high-risk histology, tumor location, time period, age, sex, and comorbidities were controlled for. Endoscopic resection plus surgery did not reduce the risk of recurrence, compared with endoscopic resection only (P = .3), after the investigators accounted for age, sex, year of procedure, tumor location, and margin characteristics. Initial surgery was associated with significantly lower rates of overall and local recurrence, compared with endoscopic resection only, but the differences also lost significance in the multivariable analysis (P = .2).
Only the presence of positive resection margins significantly predicted recurrence among patients undergoing endoscopic resection (hazard ratio, 6.9; 95% confidence interval, 2.3-20.9). Positive or doubtful resection margins also predicted recurrence after initial surgery, with hazard ratios of 13.2 and 3.4, respectively. High-risk histology – that is, poor differentiation, deep submucosal invasion, or lymphangioinvasion – was significantly associated with lymph node metastasis (OR, 2.2; 95% CI, 1.3-3.7; P less than .002), but not with recurrence after resection margins were accounted for. This might result from missing histology data or the fact that patients with high-risk histology tended to undergo surgical rather than endoscopic resection, the researchers said.
They noted several other study limitations, including a lack of details about lesions and procedures. Also, endoscopic submucosal resection was not practiced in the Netherlands during the study period, they said.
The investigators did not report funding sources and had no disclosures.
Patients with T1 colorectal cancer might not benefit from additional surgery after endoscopic resection unless they have positive or indeterminate resection margins or high-risk histology, according to a retrospective, population-based study of 1,315 patients.
After a median follow-up of 6.6 years, the rates of colorectal cancer (CRC) recurrence were 6.2% in patients who underwent endoscopic resection only and 6.4% in patients who also had additional surgery (P = .9), reported Tim D.G. Belderbos, MD, of University Medical Center Utrecht (the Netherlands). Rates of local recurrence also were similar between these groups (4.1% and 3.7%, P = .3), he and his associates reported in the March issue of Clinical Gastroenterology and Hepatology (doi: 10.1016/j.cgh.2016.08.041).
Among high-risk patients, however, the rates of overall recurrence were 14% with endoscopic resection only and 7% with endoscopic resection plus additional surgery (P = .06), and the rates of local recurrence were 12% and 1%, respectively (P = .004). “Based on our study, we recommend performing additional surgery after initial endoscopic resection in cases of high-risk T1 CRC, determined by high-risk histology and/or positive resection margins,” the researchers concluded. Invasive CRCs confined to the colonic submucosa (T1 CRC) present a treatment dilemma – they are usually cured by complete endoscopic resection, but up to 13% involve lymph node metastases and need additional surgery, the investigators noted. To identify predictors of recurrence and metastasis, they studied all patients diagnosed with T1 CRC in the Southeast Netherlands from 1995 through 2011. A total of 370 patients (28%) underwent endoscopic resection only, 220 (17%) underwent endoscopic resection with additional surgery, and 725 (55%) had an initial surgical resection.
Surgery after endoscopic resection was more likely when patients had positive or doubtful resection margins (P less than .001), and this link remained significant after high-risk histology, tumor location, time period, age, sex, and comorbidities were controlled for. Endoscopic resection plus surgery did not reduce the risk of recurrence, compared with endoscopic resection only (P = .3), after the investigators accounted for age, sex, year of procedure, tumor location, and margin characteristics. Initial surgery was associated with significantly lower rates of overall and local recurrence, compared with endoscopic resection only, but the differences also lost significance in the multivariable analysis (P = .2).
Only the presence of positive resection margins significantly predicted recurrence among patients undergoing endoscopic resection (hazard ratio, 6.9; 95% confidence interval, 2.3-20.9). Positive or doubtful resection margins also predicted recurrence after initial surgery, with hazard ratios of 13.2 and 3.4, respectively. High-risk histology – that is, poor differentiation, deep submucosal invasion, or lymphangioinvasion – was significantly associated with lymph node metastasis (OR, 2.2; 95% CI, 1.3-3.7; P less than .002), but not with recurrence after resection margins were accounted for. This might result from missing histology data or the fact that patients with high-risk histology tended to undergo surgical rather than endoscopic resection, the researchers said.
They noted several other study limitations, including a lack of details about lesions and procedures. Also, endoscopic submucosal resection was not practiced in the Netherlands during the study period, they said.
The investigators did not report funding sources and had no disclosures.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point. Patients with T1 colorectal cancer might not benefit from additional surgery after endoscopic resection unless they have positive or indeterminate resection margins or high-risk histology.
Major finding: After a median follow-up of 6.6 years, rates of CRC recurrence were 6.2% in patients who underwent endoscopic resection only, and 6.4% in patients who also had additional surgery (P = .9). Among high-risk patients, these rates were 14% and 7%, respectively (P = .06).
Data source: A retrospective population-based study of 1,315 patients who underwent endoscopic or surgical resection of T1 colorectal cancer.
Disclosures: The investigators did not report funding sources and had no disclosures.
COBRA trial takes the long view of absorbable biosynthetic mesh outcomes
Absorbable, biosynthetic surgical mesh used to repair ventral hernia defects may be a good alternative to biologic and permanent synthetic mesh products both in terms of long-term durability and cost, according to a longitudinal cohort study.
The results of the COBRA (Complex Open Bioabsorbable Reconstruction of the Abdominal Wall) study published in the January issue of Annals of Surgery represent the longest follow-up of patients in whom this product was used. Lead author of the study, Michael J. Rosen, MD, professor of surgery at Case Western Reserve University, Cleveland, and his colleagues wrote: “Absorbable synthetic mesh has the prospective advantages of a reduced cost, minimal constraints in manufacturing alternative sizes (lengths, widths, and thicknesses), informed consent in certain religious or cultural groups, and ability to be iterative in generational improvements in mesh constructs based on outcome studies, compared with allogeneic or xenogenic mesh.”
Contaminated wounds were present in 77% of participants. About one-fourth of patients had concomitant procedures for fistula takedown; a quarter of the cohort also required the removal of infected, previously placed mesh. More than a fifth of patients required a concomitant repair of both a midline and parastomal hernia; the mean size for the hernia defects was 137 cm2, and the average width was 9 cm.
Placement of the biosynthetic mesh was left to the discretion of the surgeon, but 90% chose retrorectus placement. Primary fascial closure using a single unit of the material was successful in all patients, 68 of whom required concomitant component separation; 21 of these had an external oblique release. Another 50 of these had transversus abdominis release.
At 24 months, when 84% of patients completed follow-up, 17% were found to have a hernia recurrence. Intraperitoneal placement of the material was found to significantly increase the risk of hernia recurrence (P less than or equal to .04). Infections at the surgical site were associated with a higher risk of recurrence (P less than .01). Patient-reported physical and mental quality-of-life scores at 24 months improved significantly from baseline (P less than .05), showing sustained improvement at 6 and 12 months post procedure.
While more studies are needed, the COBRA findings suggest absorbable, biosynthetic mesh compares favorably with biologic mesh when it comes to recurrence. “The Repair of Infected and Contaminated Hernias (RICH) trial is the only long-term, multicentered, prospective trial to evaluate biologic mesh in CDC [Centers for Disease Control and Prevention] class II to IV wounds. The RICH trial reported 66% surgical site occurrence and 28% hernia recurrence after 2 years’ follow-up in patients who underwent ventral hernia repair with a non–cross-linked porcine dermis,” the investigators noted.
Cost is another area in which bioabsorbable synthetic mesh compares favorably with the biologics, not only in terms of savings from fewer recurrences but also the cost of the mesh itself. Biologic mesh can cost $10,000 or more while synthetics run about a quarter of that (Clin Colon Rectal Surg. 2014 Dec; 27[4]:140-8).
In conclusion, the investigators commented that despite the lack of a control group and random assignment in the study, the results “should not be underestimated” when considering alternatives to biologic and costlier, permanent synthetic meshes.
The study was funded by W.L. Gore. The authors had no relevant financial disclosures.
Absorbable, biosynthetic surgical mesh used to repair ventral hernia defects may be a good alternative to biologic and permanent synthetic mesh products both in terms of long-term durability and cost, according to a longitudinal cohort study.
The results of the COBRA (Complex Open Bioabsorbable Reconstruction of the Abdominal Wall) study published in the January issue of Annals of Surgery represent the longest follow-up of patients in whom this product was used. Lead author of the study, Michael J. Rosen, MD, professor of surgery at Case Western Reserve University, Cleveland, and his colleagues wrote: “Absorbable synthetic mesh has the prospective advantages of a reduced cost, minimal constraints in manufacturing alternative sizes (lengths, widths, and thicknesses), informed consent in certain religious or cultural groups, and ability to be iterative in generational improvements in mesh constructs based on outcome studies, compared with allogeneic or xenogenic mesh.”
Contaminated wounds were present in 77% of participants. About one-fourth of patients had concomitant procedures for fistula takedown; a quarter of the cohort also required the removal of infected, previously placed mesh. More than a fifth of patients required a concomitant repair of both a midline and parastomal hernia; the mean size for the hernia defects was 137 cm2, and the average width was 9 cm.
Placement of the biosynthetic mesh was left to the discretion of the surgeon, but 90% chose retrorectus placement. Primary fascial closure using a single unit of the material was successful in all patients, 68 of whom required concomitant component separation; 21 of these had an external oblique release. Another 50 of these had transversus abdominis release.
At 24 months, when 84% of patients completed follow-up, 17% were found to have a hernia recurrence. Intraperitoneal placement of the material was found to significantly increase the risk of hernia recurrence (P less than or equal to .04). Infections at the surgical site were associated with a higher risk of recurrence (P less than .01). Patient-reported physical and mental quality-of-life scores at 24 months improved significantly from baseline (P less than .05), showing sustained improvement at 6 and 12 months post procedure.
While more studies are needed, the COBRA findings suggest absorbable, biosynthetic mesh compares favorably with biologic mesh when it comes to recurrence. “The Repair of Infected and Contaminated Hernias (RICH) trial is the only long-term, multicentered, prospective trial to evaluate biologic mesh in CDC [Centers for Disease Control and Prevention] class II to IV wounds. The RICH trial reported 66% surgical site occurrence and 28% hernia recurrence after 2 years’ follow-up in patients who underwent ventral hernia repair with a non–cross-linked porcine dermis,” the investigators noted.
Cost is another area in which bioabsorbable synthetic mesh compares favorably with the biologics, not only in terms of savings from fewer recurrences but also the cost of the mesh itself. Biologic mesh can cost $10,000 or more while synthetics run about a quarter of that (Clin Colon Rectal Surg. 2014 Dec; 27[4]:140-8).
In conclusion, the investigators commented that despite the lack of a control group and random assignment in the study, the results “should not be underestimated” when considering alternatives to biologic and costlier, permanent synthetic meshes.
The study was funded by W.L. Gore. The authors had no relevant financial disclosures.
Absorbable, biosynthetic surgical mesh used to repair ventral hernia defects may be a good alternative to biologic and permanent synthetic mesh products both in terms of long-term durability and cost, according to a longitudinal cohort study.
The results of the COBRA (Complex Open Bioabsorbable Reconstruction of the Abdominal Wall) study published in the January issue of Annals of Surgery represent the longest follow-up of patients in whom this product was used. Lead author of the study, Michael J. Rosen, MD, professor of surgery at Case Western Reserve University, Cleveland, and his colleagues wrote: “Absorbable synthetic mesh has the prospective advantages of a reduced cost, minimal constraints in manufacturing alternative sizes (lengths, widths, and thicknesses), informed consent in certain religious or cultural groups, and ability to be iterative in generational improvements in mesh constructs based on outcome studies, compared with allogeneic or xenogenic mesh.”
Contaminated wounds were present in 77% of participants. About one-fourth of patients had concomitant procedures for fistula takedown; a quarter of the cohort also required the removal of infected, previously placed mesh. More than a fifth of patients required a concomitant repair of both a midline and parastomal hernia; the mean size for the hernia defects was 137 cm2, and the average width was 9 cm.
Placement of the biosynthetic mesh was left to the discretion of the surgeon, but 90% chose retrorectus placement. Primary fascial closure using a single unit of the material was successful in all patients, 68 of whom required concomitant component separation; 21 of these had an external oblique release. Another 50 of these had transversus abdominis release.
At 24 months, when 84% of patients completed follow-up, 17% were found to have a hernia recurrence. Intraperitoneal placement of the material was found to significantly increase the risk of hernia recurrence (P less than or equal to .04). Infections at the surgical site were associated with a higher risk of recurrence (P less than .01). Patient-reported physical and mental quality-of-life scores at 24 months improved significantly from baseline (P less than .05), showing sustained improvement at 6 and 12 months post procedure.
While more studies are needed, the COBRA findings suggest absorbable, biosynthetic mesh compares favorably with biologic mesh when it comes to recurrence. “The Repair of Infected and Contaminated Hernias (RICH) trial is the only long-term, multicentered, prospective trial to evaluate biologic mesh in CDC [Centers for Disease Control and Prevention] class II to IV wounds. The RICH trial reported 66% surgical site occurrence and 28% hernia recurrence after 2 years’ follow-up in patients who underwent ventral hernia repair with a non–cross-linked porcine dermis,” the investigators noted.
Cost is another area in which bioabsorbable synthetic mesh compares favorably with the biologics, not only in terms of savings from fewer recurrences but also the cost of the mesh itself. Biologic mesh can cost $10,000 or more while synthetics run about a quarter of that (Clin Colon Rectal Surg. 2014 Dec; 27[4]:140-8).
In conclusion, the investigators commented that despite the lack of a control group and random assignment in the study, the results “should not be underestimated” when considering alternatives to biologic and costlier, permanent synthetic meshes.
The study was funded by W.L. Gore. The authors had no relevant financial disclosures.
Key clinical point:
Major finding: At 24 months, 17% of patients were found to have a hernia recurrence.
Data source: An international, multisite, prospective, intention-to-treat cohort analysis of 104 patients with contaminated or noncontaminated hernia defects of at least 9 cm2 in size.
Disclosures: The study was funded by W.L. Gore. The authors had no relevant financial disclosures.
Fresh Press: ACS Surgery News January issue now online
The January issue of ACS Surgery News is available on the website. This month’s issue features a special report on burnout. A new paradigm of burnout is emerging: The roots of the problem may be institutional. Addressing physician burnout must begin with recognition of the challenge and a commitment to change from the top levels of management, according to a study by Tait D. Shanafelt, MD, and John Noseworthy, MD, of the Mayo Clinic.
Don’t miss our annual Meet the Editorial Advisory Board feature. This year, we welcome seven new members: Joshua A. Broghammer, MD, FACS; Samer G. Mattar, MD, FACS; Arden M. Morris, MD, FACS; Rudolfo J. Oviedo, MD, FACS; Kevin M. Reavis, MD, FACS; Michael D. Sarap, MD, FACS; and Gary Timmerman, MD, FACS. On behalf of the editors and our readers, we sincerely thank our members who have finished their term. These colleagues have given of their time and expertise for the benefit of their fellow surgeons. They have earned our admiration and gratitude.
The January issue of ACS Surgery News is available on the website. This month’s issue features a special report on burnout. A new paradigm of burnout is emerging: The roots of the problem may be institutional. Addressing physician burnout must begin with recognition of the challenge and a commitment to change from the top levels of management, according to a study by Tait D. Shanafelt, MD, and John Noseworthy, MD, of the Mayo Clinic.
Don’t miss our annual Meet the Editorial Advisory Board feature. This year, we welcome seven new members: Joshua A. Broghammer, MD, FACS; Samer G. Mattar, MD, FACS; Arden M. Morris, MD, FACS; Rudolfo J. Oviedo, MD, FACS; Kevin M. Reavis, MD, FACS; Michael D. Sarap, MD, FACS; and Gary Timmerman, MD, FACS. On behalf of the editors and our readers, we sincerely thank our members who have finished their term. These colleagues have given of their time and expertise for the benefit of their fellow surgeons. They have earned our admiration and gratitude.
The January issue of ACS Surgery News is available on the website. This month’s issue features a special report on burnout. A new paradigm of burnout is emerging: The roots of the problem may be institutional. Addressing physician burnout must begin with recognition of the challenge and a commitment to change from the top levels of management, according to a study by Tait D. Shanafelt, MD, and John Noseworthy, MD, of the Mayo Clinic.
Don’t miss our annual Meet the Editorial Advisory Board feature. This year, we welcome seven new members: Joshua A. Broghammer, MD, FACS; Samer G. Mattar, MD, FACS; Arden M. Morris, MD, FACS; Rudolfo J. Oviedo, MD, FACS; Kevin M. Reavis, MD, FACS; Michael D. Sarap, MD, FACS; and Gary Timmerman, MD, FACS. On behalf of the editors and our readers, we sincerely thank our members who have finished their term. These colleagues have given of their time and expertise for the benefit of their fellow surgeons. They have earned our admiration and gratitude.
Adjuvant chemotherapy overused in young patients with colon cancer
Adjuvant chemotherapy may be overused among younger patients with colon cancer, without clear evidence of survival benefit over surgery alone, according to a report in JAMA Surgery.
Using data from 3,143 patients with histologically confirmed primary colon adenocarcinoma in the U.S. Department of Defense’s Central Cancer Registry and Military Heath System medical claims databases, researchers compared overall survival in those who underwent surgery and adjuvant chemotherapy to those who underwent surgery alone.
They found patients aged 18-49 years were up to eight times more likely to receive postoperative systemic chemotherapy across all tumor stages compared to patients aged 65-75 years. The odds ratios ranged from 7.98 for stage I tumors to 2.30 for stage III tumors (JAMA Surgery 2017, Jan 25. doi:10.1001/jamasurg.2016.5050).
“Furthermore, young and middle-aged adults were 2.5 times more likely to receive multiagent chemotherapy regimens and most patients with information on chemotherapy regimens underwent multiagent regimens, suggesting a tendency toward more intense treatments,” wrote Janna Manjelievskaia, MPH, of Walter Reed National Military Medical Center, and coauthors.*
However, they found that there was no significant difference in survival between those who had surgery and chemotherapy compared to those who had surgery alone, across age groups and tumor stage.
They did note greater overall survival among middle-aged patients with stage I and stage IV disease who were treated with surgery alone, compared to their older counterparts. Younger patients with stage III disease who received surgery alone also had slightly better survival than did older patients.
“The study suggests that more use of chemotherapy in younger patients did not result in additional survival benefits,” the authors wrote.
While national guidelines advise that selected patients with stage II disease – those with inadequately sampled nodes, T3 lesions or poorly differentiated histology – can be considered for adjuvant chemotherapy, the authors argued there is no solid evidence for the effectiveness of chemotherapy in these patients.
“Patients with cancer who receive chemotherapy are vulnerable to its toxicity and adverse effects and may have reduced quality of life,” they wrote. “As a result, patients may undergo decreased physical, functional, emotional, and social well-being, although these changes might be mitigated over time.”
Given the additional economic and financial cost of adjuvant chemotherapy, the authors called for further research to evaluate the appropriate use of chemotherapy in colon cancer.
The John P. Murtha Cancer Center, Walter Reed National Military Medical Center, and the National Cancer Institute supported the study. No conflicts of interest were declared.
* This story was updated on 2/6/2107
The study by Manjelievskaia et al. is a call for action, and invites contemplation and in-depth study. Appropriate treatment is vital for a patient’s survival, but excess treatment may increase complications and is a poor stewardship of health care funds.
Further investigation of the discrepancies in stage II would be worthwhile, and additional research on the age discrepancies in stage I disease would not only be interesting but also mandatory. Colorectal cancer tumor boards frequently concentrate on the complex care of rectal cancer and metastatic colon cancer. This is also a clear call for improved oversight of chemotherapy for colon cancer.
Tonia M. Young-Fadok, MD, is at the Mayo Clinic, Phoenix, Ariz. These comments are exerpts from an accompanying editorial (JAMA Surg. 2017, Jan 25. doi: 10.1001/jamasurg.2016.5051). No conflicts of interest were declared.
The study by Manjelievskaia et al. is a call for action, and invites contemplation and in-depth study. Appropriate treatment is vital for a patient’s survival, but excess treatment may increase complications and is a poor stewardship of health care funds.
Further investigation of the discrepancies in stage II would be worthwhile, and additional research on the age discrepancies in stage I disease would not only be interesting but also mandatory. Colorectal cancer tumor boards frequently concentrate on the complex care of rectal cancer and metastatic colon cancer. This is also a clear call for improved oversight of chemotherapy for colon cancer.
Tonia M. Young-Fadok, MD, is at the Mayo Clinic, Phoenix, Ariz. These comments are exerpts from an accompanying editorial (JAMA Surg. 2017, Jan 25. doi: 10.1001/jamasurg.2016.5051). No conflicts of interest were declared.
The study by Manjelievskaia et al. is a call for action, and invites contemplation and in-depth study. Appropriate treatment is vital for a patient’s survival, but excess treatment may increase complications and is a poor stewardship of health care funds.
Further investigation of the discrepancies in stage II would be worthwhile, and additional research on the age discrepancies in stage I disease would not only be interesting but also mandatory. Colorectal cancer tumor boards frequently concentrate on the complex care of rectal cancer and metastatic colon cancer. This is also a clear call for improved oversight of chemotherapy for colon cancer.
Tonia M. Young-Fadok, MD, is at the Mayo Clinic, Phoenix, Ariz. These comments are exerpts from an accompanying editorial (JAMA Surg. 2017, Jan 25. doi: 10.1001/jamasurg.2016.5051). No conflicts of interest were declared.
Adjuvant chemotherapy may be overused among younger patients with colon cancer, without clear evidence of survival benefit over surgery alone, according to a report in JAMA Surgery.
Using data from 3,143 patients with histologically confirmed primary colon adenocarcinoma in the U.S. Department of Defense’s Central Cancer Registry and Military Heath System medical claims databases, researchers compared overall survival in those who underwent surgery and adjuvant chemotherapy to those who underwent surgery alone.
They found patients aged 18-49 years were up to eight times more likely to receive postoperative systemic chemotherapy across all tumor stages compared to patients aged 65-75 years. The odds ratios ranged from 7.98 for stage I tumors to 2.30 for stage III tumors (JAMA Surgery 2017, Jan 25. doi:10.1001/jamasurg.2016.5050).
“Furthermore, young and middle-aged adults were 2.5 times more likely to receive multiagent chemotherapy regimens and most patients with information on chemotherapy regimens underwent multiagent regimens, suggesting a tendency toward more intense treatments,” wrote Janna Manjelievskaia, MPH, of Walter Reed National Military Medical Center, and coauthors.*
However, they found that there was no significant difference in survival between those who had surgery and chemotherapy compared to those who had surgery alone, across age groups and tumor stage.
They did note greater overall survival among middle-aged patients with stage I and stage IV disease who were treated with surgery alone, compared to their older counterparts. Younger patients with stage III disease who received surgery alone also had slightly better survival than did older patients.
“The study suggests that more use of chemotherapy in younger patients did not result in additional survival benefits,” the authors wrote.
While national guidelines advise that selected patients with stage II disease – those with inadequately sampled nodes, T3 lesions or poorly differentiated histology – can be considered for adjuvant chemotherapy, the authors argued there is no solid evidence for the effectiveness of chemotherapy in these patients.
“Patients with cancer who receive chemotherapy are vulnerable to its toxicity and adverse effects and may have reduced quality of life,” they wrote. “As a result, patients may undergo decreased physical, functional, emotional, and social well-being, although these changes might be mitigated over time.”
Given the additional economic and financial cost of adjuvant chemotherapy, the authors called for further research to evaluate the appropriate use of chemotherapy in colon cancer.
The John P. Murtha Cancer Center, Walter Reed National Military Medical Center, and the National Cancer Institute supported the study. No conflicts of interest were declared.
* This story was updated on 2/6/2107
Adjuvant chemotherapy may be overused among younger patients with colon cancer, without clear evidence of survival benefit over surgery alone, according to a report in JAMA Surgery.
Using data from 3,143 patients with histologically confirmed primary colon adenocarcinoma in the U.S. Department of Defense’s Central Cancer Registry and Military Heath System medical claims databases, researchers compared overall survival in those who underwent surgery and adjuvant chemotherapy to those who underwent surgery alone.
They found patients aged 18-49 years were up to eight times more likely to receive postoperative systemic chemotherapy across all tumor stages compared to patients aged 65-75 years. The odds ratios ranged from 7.98 for stage I tumors to 2.30 for stage III tumors (JAMA Surgery 2017, Jan 25. doi:10.1001/jamasurg.2016.5050).
“Furthermore, young and middle-aged adults were 2.5 times more likely to receive multiagent chemotherapy regimens and most patients with information on chemotherapy regimens underwent multiagent regimens, suggesting a tendency toward more intense treatments,” wrote Janna Manjelievskaia, MPH, of Walter Reed National Military Medical Center, and coauthors.*
However, they found that there was no significant difference in survival between those who had surgery and chemotherapy compared to those who had surgery alone, across age groups and tumor stage.
They did note greater overall survival among middle-aged patients with stage I and stage IV disease who were treated with surgery alone, compared to their older counterparts. Younger patients with stage III disease who received surgery alone also had slightly better survival than did older patients.
“The study suggests that more use of chemotherapy in younger patients did not result in additional survival benefits,” the authors wrote.
While national guidelines advise that selected patients with stage II disease – those with inadequately sampled nodes, T3 lesions or poorly differentiated histology – can be considered for adjuvant chemotherapy, the authors argued there is no solid evidence for the effectiveness of chemotherapy in these patients.
“Patients with cancer who receive chemotherapy are vulnerable to its toxicity and adverse effects and may have reduced quality of life,” they wrote. “As a result, patients may undergo decreased physical, functional, emotional, and social well-being, although these changes might be mitigated over time.”
Given the additional economic and financial cost of adjuvant chemotherapy, the authors called for further research to evaluate the appropriate use of chemotherapy in colon cancer.
The John P. Murtha Cancer Center, Walter Reed National Military Medical Center, and the National Cancer Institute supported the study. No conflicts of interest were declared.
* This story was updated on 2/6/2107
FROM JAMA SURGERY
Key clinical point: Adjuvant chemotherapy may be overused among younger patients with colon cancer, without clear evidence of a survival benefit over surgery alone.
Major finding: Younger patients with colon cancer are between two and eight times more likely to have adjuvant chemotherapy in addition to surgery compared to older patients with colon cancer.
Data source: A cohort study of 3,143 patients with histologically confirmed primary colon adenocarcinoma.
Disclosures: The John P. Murtha Cancer Center, Walter Reed National Military Medical Center, and the National Cancer Institute supported the study. No conflicts of interest were declared.
An alternative device for ESRD patients with central venous obstruction
CHICAGO – Catheter dependence is often the final option available for hemodialysis patients who have exhausted upper extremity access because of central venous obstruction. But an alternative device that combines a standard expanded polytetrafluoroethylene (ePTFE) arterial graft component with an entirely internalized central venous catheter component may provide an additional option that can help avoid catheters in selected patients, according to pooled results reported at a symposium on vascular surgery sponsored by Northwestern University.
Virginia L. Wong, MD, of University Hospitals Cleveland Medical Center, reported on her group’s and others’ experience using the Hemodialysis Reliable Outflow (HeRO) graft (Merit Medical) to gain access to the superior vena cava (SVC), thus allowing for further upper extremity access options. The device has its limitations in patients with CVO, Dr. Wong noted, “but it can be an important tool for the dedicated access surgeon who is likely to be referred the most complicated patients who have run out of just about every other option.”
The Food and Drug Administration approved the HeRO graft for CVO in 2008, but a recent pooled analysis (Eur J Vasc Endovasc Surg. 2015;50[1]:108-13), which showed a 1-year primary patency rate of 22% and a secondary patency rate of 60%, may provide clarity on how the device can be used to treat CVO in end-stage renal disease (ESRD) patients when the care team desires an alternative to femoral arteriovenous graft, Dr. Wong said. “The 1-year primary patency rate overall was not very good, but with aggressive thrombectomy programs the 1-year patency rate was decent,” she said.
The pooled analysis involved eight series from 2009 to 2015, but the largest series, which involved 164 patients, reported primary and secondary patency rates of 48.8% and 90.8%, respectively (Eur J Vasc Endovasc Surg. 2012;44[1]:93-9). “Patency for these alternative accesses may not be quite what we can achieve with standard upper-extremity access,” Dr. Wong said, “but these patients do not have the standard access as an option.”
Dr. Wong explained where the HeRO fits into the existing vascular practice. “The current data suggest that we should try to exhaust all traditional upper extremity access options before considering anything else, but the HeRO could be considered as an acceptable option for suitable patients,” she said. However, to achieve those outcomes, “you need to have an aggressive thrombectomy program.”
HeRO may be an option for salvage of an existing arm access, plagued by recalcitrant CVO, while still preserving the femoral sites and for future hemodialysis access and/or renal transplantation, Dr. Wong said.
The HeRO also has been used in alternative configurations, taking advantage of axillary or subclavian routes to the SVC when both internal jugular veins are occluded. Dr. Wong has used the femoral route to the inferior vena cava (IVC) for salvaging the femoral AV graft in which iliofemoral venous outflow has been compromised.
Anatomically, the patient must be able to accept a large-bore (19-Fr) access catheter into the central vein. Physiologically, the patient must be able to maintain patency of the long, low-resistance HeRO circuit, which can be up to 50 cm in length, she said. The protocol at Dr. Wong’s institution recommends an inflow arterial diameter of at least 3 mm, along with a left ventricular ejection fraction of 20% or greater and a minimum systolic blood pressure of 100 mm Hg for HeRO on the right side, and possibly higher when coming from the left.
Chronic hypotension is a frequent disqualifier, although some of these patients may benefit from midodrine hydrochloride, she said. In any event, a review of medications and consultation with nephrology and the dialysis unit are mandatory elements of patient screening. “I usually request hemodialysis run sheets from the last three sessions to see what systolic blood pressure excursion is like over the course of treatment,” she said.
The basic principles of hemo-access care are important when considering the HeRO for CVO patients, Dr. Wong said. These include site/side preservation, catheter avoidance and “not to burn any bridges” for future access. “Individualization of care and careful patient selection are probably the best bets if you’re just starting out,” she said. “Choose good patients before resorting to HeRO as the last option for a fairly marginal candidate.”
Dr. Wong had no relevant financial relationships to disclose.
CHICAGO – Catheter dependence is often the final option available for hemodialysis patients who have exhausted upper extremity access because of central venous obstruction. But an alternative device that combines a standard expanded polytetrafluoroethylene (ePTFE) arterial graft component with an entirely internalized central venous catheter component may provide an additional option that can help avoid catheters in selected patients, according to pooled results reported at a symposium on vascular surgery sponsored by Northwestern University.
Virginia L. Wong, MD, of University Hospitals Cleveland Medical Center, reported on her group’s and others’ experience using the Hemodialysis Reliable Outflow (HeRO) graft (Merit Medical) to gain access to the superior vena cava (SVC), thus allowing for further upper extremity access options. The device has its limitations in patients with CVO, Dr. Wong noted, “but it can be an important tool for the dedicated access surgeon who is likely to be referred the most complicated patients who have run out of just about every other option.”
The Food and Drug Administration approved the HeRO graft for CVO in 2008, but a recent pooled analysis (Eur J Vasc Endovasc Surg. 2015;50[1]:108-13), which showed a 1-year primary patency rate of 22% and a secondary patency rate of 60%, may provide clarity on how the device can be used to treat CVO in end-stage renal disease (ESRD) patients when the care team desires an alternative to femoral arteriovenous graft, Dr. Wong said. “The 1-year primary patency rate overall was not very good, but with aggressive thrombectomy programs the 1-year patency rate was decent,” she said.
The pooled analysis involved eight series from 2009 to 2015, but the largest series, which involved 164 patients, reported primary and secondary patency rates of 48.8% and 90.8%, respectively (Eur J Vasc Endovasc Surg. 2012;44[1]:93-9). “Patency for these alternative accesses may not be quite what we can achieve with standard upper-extremity access,” Dr. Wong said, “but these patients do not have the standard access as an option.”
Dr. Wong explained where the HeRO fits into the existing vascular practice. “The current data suggest that we should try to exhaust all traditional upper extremity access options before considering anything else, but the HeRO could be considered as an acceptable option for suitable patients,” she said. However, to achieve those outcomes, “you need to have an aggressive thrombectomy program.”
HeRO may be an option for salvage of an existing arm access, plagued by recalcitrant CVO, while still preserving the femoral sites and for future hemodialysis access and/or renal transplantation, Dr. Wong said.
The HeRO also has been used in alternative configurations, taking advantage of axillary or subclavian routes to the SVC when both internal jugular veins are occluded. Dr. Wong has used the femoral route to the inferior vena cava (IVC) for salvaging the femoral AV graft in which iliofemoral venous outflow has been compromised.
Anatomically, the patient must be able to accept a large-bore (19-Fr) access catheter into the central vein. Physiologically, the patient must be able to maintain patency of the long, low-resistance HeRO circuit, which can be up to 50 cm in length, she said. The protocol at Dr. Wong’s institution recommends an inflow arterial diameter of at least 3 mm, along with a left ventricular ejection fraction of 20% or greater and a minimum systolic blood pressure of 100 mm Hg for HeRO on the right side, and possibly higher when coming from the left.
Chronic hypotension is a frequent disqualifier, although some of these patients may benefit from midodrine hydrochloride, she said. In any event, a review of medications and consultation with nephrology and the dialysis unit are mandatory elements of patient screening. “I usually request hemodialysis run sheets from the last three sessions to see what systolic blood pressure excursion is like over the course of treatment,” she said.
The basic principles of hemo-access care are important when considering the HeRO for CVO patients, Dr. Wong said. These include site/side preservation, catheter avoidance and “not to burn any bridges” for future access. “Individualization of care and careful patient selection are probably the best bets if you’re just starting out,” she said. “Choose good patients before resorting to HeRO as the last option for a fairly marginal candidate.”
Dr. Wong had no relevant financial relationships to disclose.
CHICAGO – Catheter dependence is often the final option available for hemodialysis patients who have exhausted upper extremity access because of central venous obstruction. But an alternative device that combines a standard expanded polytetrafluoroethylene (ePTFE) arterial graft component with an entirely internalized central venous catheter component may provide an additional option that can help avoid catheters in selected patients, according to pooled results reported at a symposium on vascular surgery sponsored by Northwestern University.
Virginia L. Wong, MD, of University Hospitals Cleveland Medical Center, reported on her group’s and others’ experience using the Hemodialysis Reliable Outflow (HeRO) graft (Merit Medical) to gain access to the superior vena cava (SVC), thus allowing for further upper extremity access options. The device has its limitations in patients with CVO, Dr. Wong noted, “but it can be an important tool for the dedicated access surgeon who is likely to be referred the most complicated patients who have run out of just about every other option.”
The Food and Drug Administration approved the HeRO graft for CVO in 2008, but a recent pooled analysis (Eur J Vasc Endovasc Surg. 2015;50[1]:108-13), which showed a 1-year primary patency rate of 22% and a secondary patency rate of 60%, may provide clarity on how the device can be used to treat CVO in end-stage renal disease (ESRD) patients when the care team desires an alternative to femoral arteriovenous graft, Dr. Wong said. “The 1-year primary patency rate overall was not very good, but with aggressive thrombectomy programs the 1-year patency rate was decent,” she said.
The pooled analysis involved eight series from 2009 to 2015, but the largest series, which involved 164 patients, reported primary and secondary patency rates of 48.8% and 90.8%, respectively (Eur J Vasc Endovasc Surg. 2012;44[1]:93-9). “Patency for these alternative accesses may not be quite what we can achieve with standard upper-extremity access,” Dr. Wong said, “but these patients do not have the standard access as an option.”
Dr. Wong explained where the HeRO fits into the existing vascular practice. “The current data suggest that we should try to exhaust all traditional upper extremity access options before considering anything else, but the HeRO could be considered as an acceptable option for suitable patients,” she said. However, to achieve those outcomes, “you need to have an aggressive thrombectomy program.”
HeRO may be an option for salvage of an existing arm access, plagued by recalcitrant CVO, while still preserving the femoral sites and for future hemodialysis access and/or renal transplantation, Dr. Wong said.
The HeRO also has been used in alternative configurations, taking advantage of axillary or subclavian routes to the SVC when both internal jugular veins are occluded. Dr. Wong has used the femoral route to the inferior vena cava (IVC) for salvaging the femoral AV graft in which iliofemoral venous outflow has been compromised.
Anatomically, the patient must be able to accept a large-bore (19-Fr) access catheter into the central vein. Physiologically, the patient must be able to maintain patency of the long, low-resistance HeRO circuit, which can be up to 50 cm in length, she said. The protocol at Dr. Wong’s institution recommends an inflow arterial diameter of at least 3 mm, along with a left ventricular ejection fraction of 20% or greater and a minimum systolic blood pressure of 100 mm Hg for HeRO on the right side, and possibly higher when coming from the left.
Chronic hypotension is a frequent disqualifier, although some of these patients may benefit from midodrine hydrochloride, she said. In any event, a review of medications and consultation with nephrology and the dialysis unit are mandatory elements of patient screening. “I usually request hemodialysis run sheets from the last three sessions to see what systolic blood pressure excursion is like over the course of treatment,” she said.
The basic principles of hemo-access care are important when considering the HeRO for CVO patients, Dr. Wong said. These include site/side preservation, catheter avoidance and “not to burn any bridges” for future access. “Individualization of care and careful patient selection are probably the best bets if you’re just starting out,” she said. “Choose good patients before resorting to HeRO as the last option for a fairly marginal candidate.”
Dr. Wong had no relevant financial relationships to disclose.
Key clinical point: Combined graft-catheter device may preserve femoral access for hemodialysis for patients with central venous obstruction.
Major finding: One-year primary potency rate was 22% and secondary patency rate 60% for device recipients.
Data source: Literature review, including pooled results from eight studies involving 408 subjects.
Disclosures: Dr. Wong reported having no financial disclosures.
Now is time to embrace emerging PAD interventions
CHICAGO – Bioresorbable scaffolds, new drugs, adjuvant interventions, and stem and progenitor cell therapy will change how vascular surgeons treat peripheral artery disease in the next 5 years, so they must embrace these emerging treatments or run the risk of being displaced by other specialists, according to a presentation at a symposium on vascular surgery sponsored by Northwestern University.
“Vascular surgeons must position their practices to be the nexus for the evaluation and treatment of the patient and proactively engage in the critical trials of these new technologies,” said Patrick J. Geraghty, MD, of Washington University, St. Louis. “If our specialty fails to adapt to new treatment options, we risk getting sidelined as critical limb ischemia (CLI) treatment moves into a multimodality model.”
Dr. Geraghty focused on several future directions for PAD treatment: improved drug-eluting stents (DES) for superficial femoral artery disease; drug-coated balloons and modified DES for infrapopliteal disease; biologic modifiers for claudication and CLI; and bioresorbable, drug-eluting scaffolds for infrainguinal interventions.
“You’re not simply a plumber anymore; you’re a biological response modifier,” Dr. Geraghty said, explaining that biologic response modification technologies are the logical successor where standard surgical and endovascular techniques have either fallen short (as in early patency loss due to restenosis) or failed to offer effective alternatives (as in no-option advanced CLI patients). “And that takes many of us out of our comfort zone,” he said.
Dr. Geraghty noted the VIBRANT trial (J Vasc Surg. 2013;58[2]:386-95) and similar studies of non–drug eluting constructs identified early restenosis as the primary culprit in endovascular patency loss. “If you could reduce those early patency losses, you’d have an admirable primary patency rate for these complex lesions,” he said. “We’re able to reconstruct a vessel lumen. The question is, how to best maintain it?”
To answer that, Dr. Geraghty noted that the SIROCCO II trial (J Vasc Interv Radiol. 2005;16[3]:331-8) failed to show an advantage for a sirolimus-eluting stent over bare nitinol stent for superficial femoral artery (SFA) disease, but the subsequent Zilver PTX trial showed the benefits of paclitaxel-eluting stents over 5 years (Circulation. 2016;133[15]:1472-83).
He noted that drug-coated balloons (DCBs) trials have yielded mixed results in infrapopliteal intervention. Most notably, the multicenter In.Pact DEEP trial (Circulation. 2015;131[5]:495-502) failed to show treatment efficacy, Dr. Geraghty said. “The In.Pact DEEP results sharply contrasted with the positive data from trials of similar DCBs in the SFA” (N Engl J Med. 2015;373[2]:145-53).
With regard to DES for infrapopliteal disease, Dr. Geraghty noted the promise of positive results of the ACHILLES (J Am Coll Cardiol. 2012;60[22]:2290-5) and DESTINY (J Vasc Surg. 2012;55[2]:390-9) trials, along with the modest structural changes needed to convert from coronary to proximal tibial applications.
Bioresorbable vascular scaffolds (BVS) for CLI have also made recent advances. “It has been a slow road, but I’m happy that industry has pursued this aggressively,” Dr. Geraghty said. He pointed out that the ESPRIT I trial of bioresorbable everolimus-eluting vascular scaffolds in PAD involving the external iliac artery and SFA reported restenosis rates of 12.1% and 16.1% at 1 and 2 years, respectively (JACC Cardiovasc Interv. 2016;9[11]:1178-87). A trial of the Absorb BVS (Abbott) for short infrapopliteal lesions showed primary patency rates of 96% and 85% at 1 and 2 years, he said (JACC Cardiovasc Interv. 2016;9[7]:715-24).
“Vascular surgeons should be tracking BVS technology closely,” Dr. Geraghty said. “It achieves multiple desirable goals: immediate scaffolding for luminal restoration; mitigation of the restenotic stimulus via stent resorption; drug delivery for inhibition of restenosis; and the prospect of simpler re-interventions.”
Stem/progenitor cell therapies may also provide new solutions for no-option vasculature. One trial that showed “promising trends,” Dr. Geraghty said, is the RESTORE-CLI study of bone marrow aspiration (Mol Ther. 2012;20[6]:1280-6). “This trial reported a trend toward improved time to failure and reduced amputation-free survival, but did not meet its primary endpoint,” he said. “Likewise, the recently presented Biomet MOBILE data failed to meet its primary endpoint, but showed favorable trends in some treatment subgroups” (J Vasc Surg. 2011;54[6]:1650-8).
Dr. Geraghty noted that trial design in this field may need to change directions. “Look at the Delphi consensus matrices for the WIfI (Wound, Ischemia, foot Infection) Threatened limb Classification System (J Vasc Surg. 2014;59[1]:220-34). These show that complex wounds bear a significant risk of amputation, perhaps unmitigated by successful revascularization.” In addition, he called amputation-free survival “a rather blunt instrument” for evaluating how therapies impact limb outcomes and said it can confound the analysis of their effectiveness.
“Instead of confining the progenitor-cell therapies to no-option CLI trials, I’m eager to also see them investigated for treatment of claudication,” Dr. Geraghty said. “Can cell-based therapies possibly displace endovascular interventions as the first-line, least-harmful option for claudication?”
Dr. Geraghty also touched on intra/extravascular adjuvant therapies: antithrombin nanoparticles; inhibitory nanoparticles and polymeric wraps; and adventitial drug delivery techniques, among others.
“It’s critically important for vascular surgeons to position themselves for continued success in CLI treatment,” he said. “That involves aggressive practice branding, active trial participation, critical analysis of new technologies, and adoption of new, even disruptive, treatment modalities that show patient benefit.”
Dr. Geraghty disclosed stock ownership in Pulse Therapeutics; consultant fees from Bard Peripheral Vascular, Boston Scientific, Intact Vascular, Bard/Lutonix and Spectranetics; and serving as principal investigator for trials by Cook Medical, Bard/Lutonix, and Intact Vascular, with fees going to Washington University Medical School.
CHICAGO – Bioresorbable scaffolds, new drugs, adjuvant interventions, and stem and progenitor cell therapy will change how vascular surgeons treat peripheral artery disease in the next 5 years, so they must embrace these emerging treatments or run the risk of being displaced by other specialists, according to a presentation at a symposium on vascular surgery sponsored by Northwestern University.
“Vascular surgeons must position their practices to be the nexus for the evaluation and treatment of the patient and proactively engage in the critical trials of these new technologies,” said Patrick J. Geraghty, MD, of Washington University, St. Louis. “If our specialty fails to adapt to new treatment options, we risk getting sidelined as critical limb ischemia (CLI) treatment moves into a multimodality model.”
Dr. Geraghty focused on several future directions for PAD treatment: improved drug-eluting stents (DES) for superficial femoral artery disease; drug-coated balloons and modified DES for infrapopliteal disease; biologic modifiers for claudication and CLI; and bioresorbable, drug-eluting scaffolds for infrainguinal interventions.
“You’re not simply a plumber anymore; you’re a biological response modifier,” Dr. Geraghty said, explaining that biologic response modification technologies are the logical successor where standard surgical and endovascular techniques have either fallen short (as in early patency loss due to restenosis) or failed to offer effective alternatives (as in no-option advanced CLI patients). “And that takes many of us out of our comfort zone,” he said.
Dr. Geraghty noted the VIBRANT trial (J Vasc Surg. 2013;58[2]:386-95) and similar studies of non–drug eluting constructs identified early restenosis as the primary culprit in endovascular patency loss. “If you could reduce those early patency losses, you’d have an admirable primary patency rate for these complex lesions,” he said. “We’re able to reconstruct a vessel lumen. The question is, how to best maintain it?”
To answer that, Dr. Geraghty noted that the SIROCCO II trial (J Vasc Interv Radiol. 2005;16[3]:331-8) failed to show an advantage for a sirolimus-eluting stent over bare nitinol stent for superficial femoral artery (SFA) disease, but the subsequent Zilver PTX trial showed the benefits of paclitaxel-eluting stents over 5 years (Circulation. 2016;133[15]:1472-83).
He noted that drug-coated balloons (DCBs) trials have yielded mixed results in infrapopliteal intervention. Most notably, the multicenter In.Pact DEEP trial (Circulation. 2015;131[5]:495-502) failed to show treatment efficacy, Dr. Geraghty said. “The In.Pact DEEP results sharply contrasted with the positive data from trials of similar DCBs in the SFA” (N Engl J Med. 2015;373[2]:145-53).
With regard to DES for infrapopliteal disease, Dr. Geraghty noted the promise of positive results of the ACHILLES (J Am Coll Cardiol. 2012;60[22]:2290-5) and DESTINY (J Vasc Surg. 2012;55[2]:390-9) trials, along with the modest structural changes needed to convert from coronary to proximal tibial applications.
Bioresorbable vascular scaffolds (BVS) for CLI have also made recent advances. “It has been a slow road, but I’m happy that industry has pursued this aggressively,” Dr. Geraghty said. He pointed out that the ESPRIT I trial of bioresorbable everolimus-eluting vascular scaffolds in PAD involving the external iliac artery and SFA reported restenosis rates of 12.1% and 16.1% at 1 and 2 years, respectively (JACC Cardiovasc Interv. 2016;9[11]:1178-87). A trial of the Absorb BVS (Abbott) for short infrapopliteal lesions showed primary patency rates of 96% and 85% at 1 and 2 years, he said (JACC Cardiovasc Interv. 2016;9[7]:715-24).
“Vascular surgeons should be tracking BVS technology closely,” Dr. Geraghty said. “It achieves multiple desirable goals: immediate scaffolding for luminal restoration; mitigation of the restenotic stimulus via stent resorption; drug delivery for inhibition of restenosis; and the prospect of simpler re-interventions.”
Stem/progenitor cell therapies may also provide new solutions for no-option vasculature. One trial that showed “promising trends,” Dr. Geraghty said, is the RESTORE-CLI study of bone marrow aspiration (Mol Ther. 2012;20[6]:1280-6). “This trial reported a trend toward improved time to failure and reduced amputation-free survival, but did not meet its primary endpoint,” he said. “Likewise, the recently presented Biomet MOBILE data failed to meet its primary endpoint, but showed favorable trends in some treatment subgroups” (J Vasc Surg. 2011;54[6]:1650-8).
Dr. Geraghty noted that trial design in this field may need to change directions. “Look at the Delphi consensus matrices for the WIfI (Wound, Ischemia, foot Infection) Threatened limb Classification System (J Vasc Surg. 2014;59[1]:220-34). These show that complex wounds bear a significant risk of amputation, perhaps unmitigated by successful revascularization.” In addition, he called amputation-free survival “a rather blunt instrument” for evaluating how therapies impact limb outcomes and said it can confound the analysis of their effectiveness.
“Instead of confining the progenitor-cell therapies to no-option CLI trials, I’m eager to also see them investigated for treatment of claudication,” Dr. Geraghty said. “Can cell-based therapies possibly displace endovascular interventions as the first-line, least-harmful option for claudication?”
Dr. Geraghty also touched on intra/extravascular adjuvant therapies: antithrombin nanoparticles; inhibitory nanoparticles and polymeric wraps; and adventitial drug delivery techniques, among others.
“It’s critically important for vascular surgeons to position themselves for continued success in CLI treatment,” he said. “That involves aggressive practice branding, active trial participation, critical analysis of new technologies, and adoption of new, even disruptive, treatment modalities that show patient benefit.”
Dr. Geraghty disclosed stock ownership in Pulse Therapeutics; consultant fees from Bard Peripheral Vascular, Boston Scientific, Intact Vascular, Bard/Lutonix and Spectranetics; and serving as principal investigator for trials by Cook Medical, Bard/Lutonix, and Intact Vascular, with fees going to Washington University Medical School.
CHICAGO – Bioresorbable scaffolds, new drugs, adjuvant interventions, and stem and progenitor cell therapy will change how vascular surgeons treat peripheral artery disease in the next 5 years, so they must embrace these emerging treatments or run the risk of being displaced by other specialists, according to a presentation at a symposium on vascular surgery sponsored by Northwestern University.
“Vascular surgeons must position their practices to be the nexus for the evaluation and treatment of the patient and proactively engage in the critical trials of these new technologies,” said Patrick J. Geraghty, MD, of Washington University, St. Louis. “If our specialty fails to adapt to new treatment options, we risk getting sidelined as critical limb ischemia (CLI) treatment moves into a multimodality model.”
Dr. Geraghty focused on several future directions for PAD treatment: improved drug-eluting stents (DES) for superficial femoral artery disease; drug-coated balloons and modified DES for infrapopliteal disease; biologic modifiers for claudication and CLI; and bioresorbable, drug-eluting scaffolds for infrainguinal interventions.
“You’re not simply a plumber anymore; you’re a biological response modifier,” Dr. Geraghty said, explaining that biologic response modification technologies are the logical successor where standard surgical and endovascular techniques have either fallen short (as in early patency loss due to restenosis) or failed to offer effective alternatives (as in no-option advanced CLI patients). “And that takes many of us out of our comfort zone,” he said.
Dr. Geraghty noted the VIBRANT trial (J Vasc Surg. 2013;58[2]:386-95) and similar studies of non–drug eluting constructs identified early restenosis as the primary culprit in endovascular patency loss. “If you could reduce those early patency losses, you’d have an admirable primary patency rate for these complex lesions,” he said. “We’re able to reconstruct a vessel lumen. The question is, how to best maintain it?”
To answer that, Dr. Geraghty noted that the SIROCCO II trial (J Vasc Interv Radiol. 2005;16[3]:331-8) failed to show an advantage for a sirolimus-eluting stent over bare nitinol stent for superficial femoral artery (SFA) disease, but the subsequent Zilver PTX trial showed the benefits of paclitaxel-eluting stents over 5 years (Circulation. 2016;133[15]:1472-83).
He noted that drug-coated balloons (DCBs) trials have yielded mixed results in infrapopliteal intervention. Most notably, the multicenter In.Pact DEEP trial (Circulation. 2015;131[5]:495-502) failed to show treatment efficacy, Dr. Geraghty said. “The In.Pact DEEP results sharply contrasted with the positive data from trials of similar DCBs in the SFA” (N Engl J Med. 2015;373[2]:145-53).
With regard to DES for infrapopliteal disease, Dr. Geraghty noted the promise of positive results of the ACHILLES (J Am Coll Cardiol. 2012;60[22]:2290-5) and DESTINY (J Vasc Surg. 2012;55[2]:390-9) trials, along with the modest structural changes needed to convert from coronary to proximal tibial applications.
Bioresorbable vascular scaffolds (BVS) for CLI have also made recent advances. “It has been a slow road, but I’m happy that industry has pursued this aggressively,” Dr. Geraghty said. He pointed out that the ESPRIT I trial of bioresorbable everolimus-eluting vascular scaffolds in PAD involving the external iliac artery and SFA reported restenosis rates of 12.1% and 16.1% at 1 and 2 years, respectively (JACC Cardiovasc Interv. 2016;9[11]:1178-87). A trial of the Absorb BVS (Abbott) for short infrapopliteal lesions showed primary patency rates of 96% and 85% at 1 and 2 years, he said (JACC Cardiovasc Interv. 2016;9[7]:715-24).
“Vascular surgeons should be tracking BVS technology closely,” Dr. Geraghty said. “It achieves multiple desirable goals: immediate scaffolding for luminal restoration; mitigation of the restenotic stimulus via stent resorption; drug delivery for inhibition of restenosis; and the prospect of simpler re-interventions.”
Stem/progenitor cell therapies may also provide new solutions for no-option vasculature. One trial that showed “promising trends,” Dr. Geraghty said, is the RESTORE-CLI study of bone marrow aspiration (Mol Ther. 2012;20[6]:1280-6). “This trial reported a trend toward improved time to failure and reduced amputation-free survival, but did not meet its primary endpoint,” he said. “Likewise, the recently presented Biomet MOBILE data failed to meet its primary endpoint, but showed favorable trends in some treatment subgroups” (J Vasc Surg. 2011;54[6]:1650-8).
Dr. Geraghty noted that trial design in this field may need to change directions. “Look at the Delphi consensus matrices for the WIfI (Wound, Ischemia, foot Infection) Threatened limb Classification System (J Vasc Surg. 2014;59[1]:220-34). These show that complex wounds bear a significant risk of amputation, perhaps unmitigated by successful revascularization.” In addition, he called amputation-free survival “a rather blunt instrument” for evaluating how therapies impact limb outcomes and said it can confound the analysis of their effectiveness.
“Instead of confining the progenitor-cell therapies to no-option CLI trials, I’m eager to also see them investigated for treatment of claudication,” Dr. Geraghty said. “Can cell-based therapies possibly displace endovascular interventions as the first-line, least-harmful option for claudication?”
Dr. Geraghty also touched on intra/extravascular adjuvant therapies: antithrombin nanoparticles; inhibitory nanoparticles and polymeric wraps; and adventitial drug delivery techniques, among others.
“It’s critically important for vascular surgeons to position themselves for continued success in CLI treatment,” he said. “That involves aggressive practice branding, active trial participation, critical analysis of new technologies, and adoption of new, even disruptive, treatment modalities that show patient benefit.”
Dr. Geraghty disclosed stock ownership in Pulse Therapeutics; consultant fees from Bard Peripheral Vascular, Boston Scientific, Intact Vascular, Bard/Lutonix and Spectranetics; and serving as principal investigator for trials by Cook Medical, Bard/Lutonix, and Intact Vascular, with fees going to Washington University Medical School.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Key clinical point: Emerging treatments for lower-extremity interventions range from improved drug-eluting stents for the superficial femoral artery and infrapopliteal disease to bioresorbable, drug-eluting scaffolds for infrainguinal interventions.
Major finding: The future of minimally invasive revascularization hinges on reliably reopening stenosed or occluded arteries, maintaining vessel patency and using therapies to stimulate arteriogenesis or angiogenesis without reintervention.
Data source: Review of literature.
Disclosures: Dr. Geraghty disclosed stock ownership in Pulse Therapeutics; consultant fees from Bard Peripheral Vascular, Boston Scientific, Intact Vascular, Bard/Lutonix and Spectranetics; and serving as principal investigator for trials by Cook Medical, Bard/Lutonix, and Intact Vascular, with fees going to Washington University Medical School.
Incompatible Type A plasma found safe for initial resuscitation of trauma patients
HOLLYWOOD, FLA. – Incompatible Type A plasma appears to be a safe and effective part of an initial resuscitation protocol for trauma patients who need a massive transfusion.
There were no increases in morbidity, mortality, or transfusion-related acute lung injury among 120 patients who received Type A plasma, compared with those who got compatible plasma, Bryan C. Morse, MD, said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Type AB blood products are preferred for initial transfusions for trauma patients with unknown blood type. While type AB blood products are universally acceptable to patients, they are also in short supply. In an attempt to mitigate this shortage, some trauma centers are relying on anecdotal data, much drawn from real-life combat experience dating from World War II to present times, suggesting that Type A plasma is safe for initial resuscitation protocols. But the body of data from well-constructed trials is small, said Dr. Morse of Emory University, Atlanta. Thus, EAST sponsored this retrospective registry study, which examined outcomes in 1,536 trauma patients who received plasma transfusions as part of a massive transfusion protocol from 2012 to 2016.
The primary endpoints were overall morbidity, and mortality at four time points: 6 and 24 hours, and 7 and 28 days. Eight trauma centers contributed data to the study.
The group was largely male (75%) with a mean age of 37 years. Patients were seriously injured, with a mean Injury Severity Score (ISS) of 25. About 60% suffered from blunt-force trauma. Among the entire group, 120 (8%) received incompatible type A plasma.
About 28% of patients (434) experienced an adverse event. These were numerically but not significantly more common among the incompatible A plasma group (35% vs. 28%; P = .14). Events included acute respiratory distress syndrome (6% vs. 7.6%), thromboembolism (9% vs. 7%), pneumonia (19% vs. 15%), and acute kidney injury (8% each).
There were two cases of transfusion-related acute lung injury, both of which occurred in the compatible type A group.
Mortality was similar at every time point: 6 hours (16% vs. 15%), 24 hours (25% vs. 22%), 7 days (35% vs. 32%), and 28 days (38% vs. 35%).
A multivariate regression model controlled for treatment center, ISS, units of packed red cells given by 4 hours, mechanism of injury, Type A plasma incompatibility, and age.
In the morbidity analysis, only ISS and units of red blood cells at 4 hours were associated with a significant increase in risk (odd ratio 1.02). Incompatible Type A plasma did not significantly increase the risk of morbidity.
In the mortality analysis, units of red cells, ISS, and age were significantly associated with increased risk. Again, incompatible Type A plasma did not significantly increase the risk of death.
Dr. Morse had no financial declaration.
[email protected]
On Twitter @Alz_Gal
HOLLYWOOD, FLA. – Incompatible Type A plasma appears to be a safe and effective part of an initial resuscitation protocol for trauma patients who need a massive transfusion.
There were no increases in morbidity, mortality, or transfusion-related acute lung injury among 120 patients who received Type A plasma, compared with those who got compatible plasma, Bryan C. Morse, MD, said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Type AB blood products are preferred for initial transfusions for trauma patients with unknown blood type. While type AB blood products are universally acceptable to patients, they are also in short supply. In an attempt to mitigate this shortage, some trauma centers are relying on anecdotal data, much drawn from real-life combat experience dating from World War II to present times, suggesting that Type A plasma is safe for initial resuscitation protocols. But the body of data from well-constructed trials is small, said Dr. Morse of Emory University, Atlanta. Thus, EAST sponsored this retrospective registry study, which examined outcomes in 1,536 trauma patients who received plasma transfusions as part of a massive transfusion protocol from 2012 to 2016.
The primary endpoints were overall morbidity, and mortality at four time points: 6 and 24 hours, and 7 and 28 days. Eight trauma centers contributed data to the study.
The group was largely male (75%) with a mean age of 37 years. Patients were seriously injured, with a mean Injury Severity Score (ISS) of 25. About 60% suffered from blunt-force trauma. Among the entire group, 120 (8%) received incompatible type A plasma.
About 28% of patients (434) experienced an adverse event. These were numerically but not significantly more common among the incompatible A plasma group (35% vs. 28%; P = .14). Events included acute respiratory distress syndrome (6% vs. 7.6%), thromboembolism (9% vs. 7%), pneumonia (19% vs. 15%), and acute kidney injury (8% each).
There were two cases of transfusion-related acute lung injury, both of which occurred in the compatible type A group.
Mortality was similar at every time point: 6 hours (16% vs. 15%), 24 hours (25% vs. 22%), 7 days (35% vs. 32%), and 28 days (38% vs. 35%).
A multivariate regression model controlled for treatment center, ISS, units of packed red cells given by 4 hours, mechanism of injury, Type A plasma incompatibility, and age.
In the morbidity analysis, only ISS and units of red blood cells at 4 hours were associated with a significant increase in risk (odd ratio 1.02). Incompatible Type A plasma did not significantly increase the risk of morbidity.
In the mortality analysis, units of red cells, ISS, and age were significantly associated with increased risk. Again, incompatible Type A plasma did not significantly increase the risk of death.
Dr. Morse had no financial declaration.
[email protected]
On Twitter @Alz_Gal
HOLLYWOOD, FLA. – Incompatible Type A plasma appears to be a safe and effective part of an initial resuscitation protocol for trauma patients who need a massive transfusion.
There were no increases in morbidity, mortality, or transfusion-related acute lung injury among 120 patients who received Type A plasma, compared with those who got compatible plasma, Bryan C. Morse, MD, said at the annual scientific assembly of the Eastern Association for the Surgery of Trauma.
Type AB blood products are preferred for initial transfusions for trauma patients with unknown blood type. While type AB blood products are universally acceptable to patients, they are also in short supply. In an attempt to mitigate this shortage, some trauma centers are relying on anecdotal data, much drawn from real-life combat experience dating from World War II to present times, suggesting that Type A plasma is safe for initial resuscitation protocols. But the body of data from well-constructed trials is small, said Dr. Morse of Emory University, Atlanta. Thus, EAST sponsored this retrospective registry study, which examined outcomes in 1,536 trauma patients who received plasma transfusions as part of a massive transfusion protocol from 2012 to 2016.
The primary endpoints were overall morbidity, and mortality at four time points: 6 and 24 hours, and 7 and 28 days. Eight trauma centers contributed data to the study.
The group was largely male (75%) with a mean age of 37 years. Patients were seriously injured, with a mean Injury Severity Score (ISS) of 25. About 60% suffered from blunt-force trauma. Among the entire group, 120 (8%) received incompatible type A plasma.
About 28% of patients (434) experienced an adverse event. These were numerically but not significantly more common among the incompatible A plasma group (35% vs. 28%; P = .14). Events included acute respiratory distress syndrome (6% vs. 7.6%), thromboembolism (9% vs. 7%), pneumonia (19% vs. 15%), and acute kidney injury (8% each).
There were two cases of transfusion-related acute lung injury, both of which occurred in the compatible type A group.
Mortality was similar at every time point: 6 hours (16% vs. 15%), 24 hours (25% vs. 22%), 7 days (35% vs. 32%), and 28 days (38% vs. 35%).
A multivariate regression model controlled for treatment center, ISS, units of packed red cells given by 4 hours, mechanism of injury, Type A plasma incompatibility, and age.
In the morbidity analysis, only ISS and units of red blood cells at 4 hours were associated with a significant increase in risk (odd ratio 1.02). Incompatible Type A plasma did not significantly increase the risk of morbidity.
In the mortality analysis, units of red cells, ISS, and age were significantly associated with increased risk. Again, incompatible Type A plasma did not significantly increase the risk of death.
Dr. Morse had no financial declaration.
[email protected]
On Twitter @Alz_Gal
AT THE EAST SCIENTIFIC ASSEMBLY
Key clinical point:
Major finding: Adverse events were not significantly more common among the incompatible A plasma group (35% vs. 28%; P = .14).
Data source: The retrospective study comprised 1,536 patients.
Disclosures: Dr. Morse had no financial disclosures.