User login
Official Newspaper of the American College of Surgeons
New auto-grafting techniques could advance wound healing
MIAMI – Pinch grafting can accelerate the healing of chronic, treatment-resistant wounds such as leg ulcers, while at the same time reducing morbidity to the donor skin site. A new epidermal harvesting device also is showing promise, as is a new tool that minces autologous skin grafts prior to application to promote wound healing.
These and other advances in wound healing were presented at the Orlando Dermatology Aesthetic and Clinical Conference. The pinch grafts and minced grafts each rely on the newly added skin to stimulate cytokines. Interestingly, there is evidence that grafts taken from hair-bearing donor sites could be superior for stimulating cytokines and accelerating wound healing, said Robert Kirsner, MD, PhD, of the University of Miami Health System.
Islands of regrowth
Physicians perform pinch grafting by taking small punches of skin from a donor site on the thigh, abdomen, or elsewhere, and then transferring the grafts to serve as islands of regrowth in a wound. Pinch grafting can be faster and less expensive than techniques typically performed in an operating room, such as meshed auto-grafting. In contrast, pinch grafting can be accomplished in an office setting “and patients can do quite well.” Dr. Kirsner said. In terms of outcomes, “our data is typical,” he added. “About 50% of refractory ulcers heal, 25% improve, and a percentage recur.”
Spreadable skin grafts
Another autologous grafting technique that can be performed at the bedside uses the Xpansion Micro-Autografting Kit, which minces autologous, split thickness skin grafts. “Then you apply them like peanut butter to bread,” Dr. Kirsner said.
The micro-autografts can help heal both acute and chronic wounds, including full thickness wounds from trauma, some burn wounds, diabetic foot ulcers, and venous ulcers, according to the manufacturer’s website.
Epidermal harvesting (without anesthesia)
Epidermal grafting can make sense because the epidermis regenerates. “You can lift off just the epidermis with heat or suction, “ Dr. Kirsner said. For the first time, he added, a new tool allows epidermal grafting without the need for anesthesia (Cellutome Epidermal Harvesting System). The device raises little microdomes of epidermis down to the basal layer, including basal keratinocytes and melanocytes, and a dermatologist can use a sterile dressing to transfer them to the wound. Confocal microscopy shows the dermoepidermal junction healing as early as within 2 days.
The epidermal harvesting was initially developed for pigment problems, such as piebaldism. (Dermatol Surg. 2017 Jan;43[1]:159-60). “We quickly realized it might have applicability for nonhealing wounds,” Dr. Kirsner said.
Deeper wound healing
A novel strategy for triggering deeper wound healing evolved from fractional laser technology, which remove columns of skin to generate healing. Instead, Rox Anderson, MD, of Massachusetts General Hospital, Boston, “envisioned pulling up microcolumns of full thickness epidermis, all the way to the fat, placing them into a wound, and the wound would heal with very little donor site morbidity,” Dr. Kirsner said.
This tool is coming out in spring of this year, he noted. It will resemble a fractional laser, “but now you have the skin available to place in another wound.” Prior animal studies revealed a healing benefit with very little scarring, he added.
Is hairier better?
Does the donor site matter? Dr. Kirsner asked. Although dermatologists typically graft skin from an abdomen or thigh, a hair-bearing site may be a better option because of the presence of pluripotent stem cells, according to a case report (Wounds. 2016 Apr;28[4]:109-11). J.D. Fox of the University of Miami, Dr. Kirsner, and their colleagues treated a large, chronic venous leg ulcer, almost 60 cm2, with punch grafts from a variety of donor sites.
“The side that got scalp punch grafts healed better, suggesting with skin taken from richly hairy area, you’ll get better results,” Dr. Kirsner said.
Another study supports this strategy (J Am Acad Dermatol. 2016 Nov;75[5]:1007-14). These researchers reported greater wound size reduction using grafts containing hair follicles versus nonhairy areas, again suggesting follicular stem cells play a role in better wound healing, Dr. Kirsner said. “This may be a better source of donor skin in the future.”
Dr. Kirsner is a consultant for Cardinal Health, Mölnlycke, Amniox, Organogenesis, Kerecis, Keretec, and KCI, an Acelity company.
MIAMI – Pinch grafting can accelerate the healing of chronic, treatment-resistant wounds such as leg ulcers, while at the same time reducing morbidity to the donor skin site. A new epidermal harvesting device also is showing promise, as is a new tool that minces autologous skin grafts prior to application to promote wound healing.
These and other advances in wound healing were presented at the Orlando Dermatology Aesthetic and Clinical Conference. The pinch grafts and minced grafts each rely on the newly added skin to stimulate cytokines. Interestingly, there is evidence that grafts taken from hair-bearing donor sites could be superior for stimulating cytokines and accelerating wound healing, said Robert Kirsner, MD, PhD, of the University of Miami Health System.
Islands of regrowth
Physicians perform pinch grafting by taking small punches of skin from a donor site on the thigh, abdomen, or elsewhere, and then transferring the grafts to serve as islands of regrowth in a wound. Pinch grafting can be faster and less expensive than techniques typically performed in an operating room, such as meshed auto-grafting. In contrast, pinch grafting can be accomplished in an office setting “and patients can do quite well.” Dr. Kirsner said. In terms of outcomes, “our data is typical,” he added. “About 50% of refractory ulcers heal, 25% improve, and a percentage recur.”
Spreadable skin grafts
Another autologous grafting technique that can be performed at the bedside uses the Xpansion Micro-Autografting Kit, which minces autologous, split thickness skin grafts. “Then you apply them like peanut butter to bread,” Dr. Kirsner said.
The micro-autografts can help heal both acute and chronic wounds, including full thickness wounds from trauma, some burn wounds, diabetic foot ulcers, and venous ulcers, according to the manufacturer’s website.
Epidermal harvesting (without anesthesia)
Epidermal grafting can make sense because the epidermis regenerates. “You can lift off just the epidermis with heat or suction, “ Dr. Kirsner said. For the first time, he added, a new tool allows epidermal grafting without the need for anesthesia (Cellutome Epidermal Harvesting System). The device raises little microdomes of epidermis down to the basal layer, including basal keratinocytes and melanocytes, and a dermatologist can use a sterile dressing to transfer them to the wound. Confocal microscopy shows the dermoepidermal junction healing as early as within 2 days.
The epidermal harvesting was initially developed for pigment problems, such as piebaldism. (Dermatol Surg. 2017 Jan;43[1]:159-60). “We quickly realized it might have applicability for nonhealing wounds,” Dr. Kirsner said.
Deeper wound healing
A novel strategy for triggering deeper wound healing evolved from fractional laser technology, which remove columns of skin to generate healing. Instead, Rox Anderson, MD, of Massachusetts General Hospital, Boston, “envisioned pulling up microcolumns of full thickness epidermis, all the way to the fat, placing them into a wound, and the wound would heal with very little donor site morbidity,” Dr. Kirsner said.
This tool is coming out in spring of this year, he noted. It will resemble a fractional laser, “but now you have the skin available to place in another wound.” Prior animal studies revealed a healing benefit with very little scarring, he added.
Is hairier better?
Does the donor site matter? Dr. Kirsner asked. Although dermatologists typically graft skin from an abdomen or thigh, a hair-bearing site may be a better option because of the presence of pluripotent stem cells, according to a case report (Wounds. 2016 Apr;28[4]:109-11). J.D. Fox of the University of Miami, Dr. Kirsner, and their colleagues treated a large, chronic venous leg ulcer, almost 60 cm2, with punch grafts from a variety of donor sites.
“The side that got scalp punch grafts healed better, suggesting with skin taken from richly hairy area, you’ll get better results,” Dr. Kirsner said.
Another study supports this strategy (J Am Acad Dermatol. 2016 Nov;75[5]:1007-14). These researchers reported greater wound size reduction using grafts containing hair follicles versus nonhairy areas, again suggesting follicular stem cells play a role in better wound healing, Dr. Kirsner said. “This may be a better source of donor skin in the future.”
Dr. Kirsner is a consultant for Cardinal Health, Mölnlycke, Amniox, Organogenesis, Kerecis, Keretec, and KCI, an Acelity company.
MIAMI – Pinch grafting can accelerate the healing of chronic, treatment-resistant wounds such as leg ulcers, while at the same time reducing morbidity to the donor skin site. A new epidermal harvesting device also is showing promise, as is a new tool that minces autologous skin grafts prior to application to promote wound healing.
These and other advances in wound healing were presented at the Orlando Dermatology Aesthetic and Clinical Conference. The pinch grafts and minced grafts each rely on the newly added skin to stimulate cytokines. Interestingly, there is evidence that grafts taken from hair-bearing donor sites could be superior for stimulating cytokines and accelerating wound healing, said Robert Kirsner, MD, PhD, of the University of Miami Health System.
Islands of regrowth
Physicians perform pinch grafting by taking small punches of skin from a donor site on the thigh, abdomen, or elsewhere, and then transferring the grafts to serve as islands of regrowth in a wound. Pinch grafting can be faster and less expensive than techniques typically performed in an operating room, such as meshed auto-grafting. In contrast, pinch grafting can be accomplished in an office setting “and patients can do quite well.” Dr. Kirsner said. In terms of outcomes, “our data is typical,” he added. “About 50% of refractory ulcers heal, 25% improve, and a percentage recur.”
Spreadable skin grafts
Another autologous grafting technique that can be performed at the bedside uses the Xpansion Micro-Autografting Kit, which minces autologous, split thickness skin grafts. “Then you apply them like peanut butter to bread,” Dr. Kirsner said.
The micro-autografts can help heal both acute and chronic wounds, including full thickness wounds from trauma, some burn wounds, diabetic foot ulcers, and venous ulcers, according to the manufacturer’s website.
Epidermal harvesting (without anesthesia)
Epidermal grafting can make sense because the epidermis regenerates. “You can lift off just the epidermis with heat or suction, “ Dr. Kirsner said. For the first time, he added, a new tool allows epidermal grafting without the need for anesthesia (Cellutome Epidermal Harvesting System). The device raises little microdomes of epidermis down to the basal layer, including basal keratinocytes and melanocytes, and a dermatologist can use a sterile dressing to transfer them to the wound. Confocal microscopy shows the dermoepidermal junction healing as early as within 2 days.
The epidermal harvesting was initially developed for pigment problems, such as piebaldism. (Dermatol Surg. 2017 Jan;43[1]:159-60). “We quickly realized it might have applicability for nonhealing wounds,” Dr. Kirsner said.
Deeper wound healing
A novel strategy for triggering deeper wound healing evolved from fractional laser technology, which remove columns of skin to generate healing. Instead, Rox Anderson, MD, of Massachusetts General Hospital, Boston, “envisioned pulling up microcolumns of full thickness epidermis, all the way to the fat, placing them into a wound, and the wound would heal with very little donor site morbidity,” Dr. Kirsner said.
This tool is coming out in spring of this year, he noted. It will resemble a fractional laser, “but now you have the skin available to place in another wound.” Prior animal studies revealed a healing benefit with very little scarring, he added.
Is hairier better?
Does the donor site matter? Dr. Kirsner asked. Although dermatologists typically graft skin from an abdomen or thigh, a hair-bearing site may be a better option because of the presence of pluripotent stem cells, according to a case report (Wounds. 2016 Apr;28[4]:109-11). J.D. Fox of the University of Miami, Dr. Kirsner, and their colleagues treated a large, chronic venous leg ulcer, almost 60 cm2, with punch grafts from a variety of donor sites.
“The side that got scalp punch grafts healed better, suggesting with skin taken from richly hairy area, you’ll get better results,” Dr. Kirsner said.
Another study supports this strategy (J Am Acad Dermatol. 2016 Nov;75[5]:1007-14). These researchers reported greater wound size reduction using grafts containing hair follicles versus nonhairy areas, again suggesting follicular stem cells play a role in better wound healing, Dr. Kirsner said. “This may be a better source of donor skin in the future.”
Dr. Kirsner is a consultant for Cardinal Health, Mölnlycke, Amniox, Organogenesis, Kerecis, Keretec, and KCI, an Acelity company.
Survival better with breast-conserving therapy for early cancers
AMSTERDAM – In real-life practice, women with early, localized breast cancer who underwent breast conserving therapy had better breast cancer–specific and overall survival compared with women who underwent mastectomy, according to investigators in the Netherlands.
Among nearly 130,000 patients treated over two different time periods, breast-conserving surgery and radiation (BCT) was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities – irrespective of either hormonal or human epidermal growth factor receptor 2 (HER2) status, reported Mirelle Lagendijk, MD, of Erasmus Medical Center Cancer Institute in Rotterdam, the Netherlands.
For patients 50 and younger, overall survival (OS), but not breast cancer–specific survival (BCSS), was superior with the more conservative approach.
“Breast conserving therapy in these identified subgroups seems to be the preferable treatment when both treatments are optional,” Dr. Lagendijk said at an annual congress sponsored by the European Cancer Organisation.
Although recent observational studies have shown survival with BCT to be at least equivalent for women with early stage disease, there is still a lack of sufficient data on BCSS, potential confounders such as systemic therapies and comorbidities, and on the relative effects of BCT or mastectomy on subgroups, she said.
The investigators drew data from the Netherlands Cancer Registry on 129,692 patients with early, primary invasive breast cancer without metastases other than to regional lymph nodes (T1-2NO-2MO).
They compared BCT to mastectomy for BCSS and OS in the population as a whole and in subgroups based on prognostic factors. They controlled for age, tumor and nodal stage, comorbidities, systemic therapy, hormone receptor and HER2 status, differentiation grade, morphology, year of treatment, axillary lymph node dissection, and contralateral breast cancer.
They divided patients into two treatment time periods. The older cohort consisted of 60,381 patients treated from 1999 through 2005, 48% of whom underwent mastectomy, with a median follow-up of 11.1 years, and 52% of whom had BCT, with a median follow-up of 12 years.
The more recent cohort consisted of 69,311 patients, 40% of whom had mastectomy with a median follow-up of 5.9 years, and 60% of whom had BCT with a median follow-up of 6.1 years.
In both time periods, deaths from all causes were lower among patients treated with BCT. In the older cohort, 13,960 of 28,968 patients (48.2%) who underwent mastectomy had died, compared with 8,915 of 31,413 patients (28.4%) who underwent BCT. In the more recent cohort, 5,504 of 27,731 (19.8%) of patients who had mastectomies had died, compared with 3,702 of 41,580 (8.9%) who underwent BCT.
“Irrespective of the time cohort and irrespective of the treatment, around 50% of the events were breast cancer related,” Dr. Lagendijk said.
BCSS was superior with BCT in each time cohort (log-rank P less than .001 for each). In the earlier cohort, BCT was significantly superior for BCSS across all disease stages; in the later cohort, it was significant for all but stages T1N1 and T1-2N2.
BCSS was superior for patients in all age categories in the early cohort, and for patients 50 and older in the later cohort.
“The final stratification performed for comorbidities present in the patients evaluated showed, surprisingly, that especially for those patients with comorbidity, there was significantly better breast cancer-specific survival when treated by breast conserving therapy as compared to a mastectomy,” Dr. Lagendijk said.
The investigators acknowledged that the study was limited by its retrospective design, potential confounding by severity, and the inability to show causal relationship between survival and treatment type.
Dutch health agencies sponsored the study. Dr. Lagendijk and Dr. Naredi reported no conflicts of interest.
AMSTERDAM – In real-life practice, women with early, localized breast cancer who underwent breast conserving therapy had better breast cancer–specific and overall survival compared with women who underwent mastectomy, according to investigators in the Netherlands.
Among nearly 130,000 patients treated over two different time periods, breast-conserving surgery and radiation (BCT) was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities – irrespective of either hormonal or human epidermal growth factor receptor 2 (HER2) status, reported Mirelle Lagendijk, MD, of Erasmus Medical Center Cancer Institute in Rotterdam, the Netherlands.
For patients 50 and younger, overall survival (OS), but not breast cancer–specific survival (BCSS), was superior with the more conservative approach.
“Breast conserving therapy in these identified subgroups seems to be the preferable treatment when both treatments are optional,” Dr. Lagendijk said at an annual congress sponsored by the European Cancer Organisation.
Although recent observational studies have shown survival with BCT to be at least equivalent for women with early stage disease, there is still a lack of sufficient data on BCSS, potential confounders such as systemic therapies and comorbidities, and on the relative effects of BCT or mastectomy on subgroups, she said.
The investigators drew data from the Netherlands Cancer Registry on 129,692 patients with early, primary invasive breast cancer without metastases other than to regional lymph nodes (T1-2NO-2MO).
They compared BCT to mastectomy for BCSS and OS in the population as a whole and in subgroups based on prognostic factors. They controlled for age, tumor and nodal stage, comorbidities, systemic therapy, hormone receptor and HER2 status, differentiation grade, morphology, year of treatment, axillary lymph node dissection, and contralateral breast cancer.
They divided patients into two treatment time periods. The older cohort consisted of 60,381 patients treated from 1999 through 2005, 48% of whom underwent mastectomy, with a median follow-up of 11.1 years, and 52% of whom had BCT, with a median follow-up of 12 years.
The more recent cohort consisted of 69,311 patients, 40% of whom had mastectomy with a median follow-up of 5.9 years, and 60% of whom had BCT with a median follow-up of 6.1 years.
In both time periods, deaths from all causes were lower among patients treated with BCT. In the older cohort, 13,960 of 28,968 patients (48.2%) who underwent mastectomy had died, compared with 8,915 of 31,413 patients (28.4%) who underwent BCT. In the more recent cohort, 5,504 of 27,731 (19.8%) of patients who had mastectomies had died, compared with 3,702 of 41,580 (8.9%) who underwent BCT.
“Irrespective of the time cohort and irrespective of the treatment, around 50% of the events were breast cancer related,” Dr. Lagendijk said.
BCSS was superior with BCT in each time cohort (log-rank P less than .001 for each). In the earlier cohort, BCT was significantly superior for BCSS across all disease stages; in the later cohort, it was significant for all but stages T1N1 and T1-2N2.
BCSS was superior for patients in all age categories in the early cohort, and for patients 50 and older in the later cohort.
“The final stratification performed for comorbidities present in the patients evaluated showed, surprisingly, that especially for those patients with comorbidity, there was significantly better breast cancer-specific survival when treated by breast conserving therapy as compared to a mastectomy,” Dr. Lagendijk said.
The investigators acknowledged that the study was limited by its retrospective design, potential confounding by severity, and the inability to show causal relationship between survival and treatment type.
Dutch health agencies sponsored the study. Dr. Lagendijk and Dr. Naredi reported no conflicts of interest.
AMSTERDAM – In real-life practice, women with early, localized breast cancer who underwent breast conserving therapy had better breast cancer–specific and overall survival compared with women who underwent mastectomy, according to investigators in the Netherlands.
Among nearly 130,000 patients treated over two different time periods, breast-conserving surgery and radiation (BCT) was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities – irrespective of either hormonal or human epidermal growth factor receptor 2 (HER2) status, reported Mirelle Lagendijk, MD, of Erasmus Medical Center Cancer Institute in Rotterdam, the Netherlands.
For patients 50 and younger, overall survival (OS), but not breast cancer–specific survival (BCSS), was superior with the more conservative approach.
“Breast conserving therapy in these identified subgroups seems to be the preferable treatment when both treatments are optional,” Dr. Lagendijk said at an annual congress sponsored by the European Cancer Organisation.
Although recent observational studies have shown survival with BCT to be at least equivalent for women with early stage disease, there is still a lack of sufficient data on BCSS, potential confounders such as systemic therapies and comorbidities, and on the relative effects of BCT or mastectomy on subgroups, she said.
The investigators drew data from the Netherlands Cancer Registry on 129,692 patients with early, primary invasive breast cancer without metastases other than to regional lymph nodes (T1-2NO-2MO).
They compared BCT to mastectomy for BCSS and OS in the population as a whole and in subgroups based on prognostic factors. They controlled for age, tumor and nodal stage, comorbidities, systemic therapy, hormone receptor and HER2 status, differentiation grade, morphology, year of treatment, axillary lymph node dissection, and contralateral breast cancer.
They divided patients into two treatment time periods. The older cohort consisted of 60,381 patients treated from 1999 through 2005, 48% of whom underwent mastectomy, with a median follow-up of 11.1 years, and 52% of whom had BCT, with a median follow-up of 12 years.
The more recent cohort consisted of 69,311 patients, 40% of whom had mastectomy with a median follow-up of 5.9 years, and 60% of whom had BCT with a median follow-up of 6.1 years.
In both time periods, deaths from all causes were lower among patients treated with BCT. In the older cohort, 13,960 of 28,968 patients (48.2%) who underwent mastectomy had died, compared with 8,915 of 31,413 patients (28.4%) who underwent BCT. In the more recent cohort, 5,504 of 27,731 (19.8%) of patients who had mastectomies had died, compared with 3,702 of 41,580 (8.9%) who underwent BCT.
“Irrespective of the time cohort and irrespective of the treatment, around 50% of the events were breast cancer related,” Dr. Lagendijk said.
BCSS was superior with BCT in each time cohort (log-rank P less than .001 for each). In the earlier cohort, BCT was significantly superior for BCSS across all disease stages; in the later cohort, it was significant for all but stages T1N1 and T1-2N2.
BCSS was superior for patients in all age categories in the early cohort, and for patients 50 and older in the later cohort.
“The final stratification performed for comorbidities present in the patients evaluated showed, surprisingly, that especially for those patients with comorbidity, there was significantly better breast cancer-specific survival when treated by breast conserving therapy as compared to a mastectomy,” Dr. Lagendijk said.
The investigators acknowledged that the study was limited by its retrospective design, potential confounding by severity, and the inability to show causal relationship between survival and treatment type.
Dutch health agencies sponsored the study. Dr. Lagendijk and Dr. Naredi reported no conflicts of interest.
AT ECCO2017
Key clinical point: Breast cancer–specific survival and overall survival were better among women who had breast-conserving therapy (BCT) compared with mastectomy.
Major finding: BCT was associated with superior survival for women older than 50, patients who did not receive adjuvant chemotherapy, and those with comorbidities.
Data source: Retrospective registry data study of 129,692 women treated for early breast cancer in the Netherlands during 1999-2005 and 2006-2012.
Disclosures: Dutch health agencies sponsored the study. Dr. Langendijk and Dr. Naredi reported no conflicts of interest.
From the Washington Office: Navigating MIPS in 2017
2017 is here and the new Merit-based Incentive Payment System (MIPS) is now in effect. MIPS has taken a number of steps to streamline reporting and make it easier to avoid penalties and achieve positive updates. However, over time penalties for non-participation or poor performance will grow. Therefore, it is critically important that all surgeons make a plan for how they can best participate in order to succeed. Knowing what options are available is vital to navigating the new reporting requirements and achieving the best possible financial outcome.
Background on MIPS and its components
MIPS began measuring performance in January 2017. The data reported in 2017 will be used to adjust payments in 2019. MIPS took the Physician Quality Reporting System (PQRS), the Value Based Modifier (VM), and the EHR Incentive Program commonly referred to as Meaningful Use (EHR-MU), added a new component that provides credit for Improvement Activities and combined them to derive a composite MIPS Final Score. The components of the Final Score are known as Quality, (formerly PQRS), Cost, (formerly VM), Advancing Care Information (ACI), (formerly EHR-MU), and Improvement Activities. The weights for the individual components of the final score for the first year of the MIPS program are represented in the chart above.
Though CMS has chosen not to provide any weight to the Cost component during the first year of the program, those who report Quality data will receive feedback reports on their performance in the Cost component.
2017: The transition year
The Centers for Medicare & Medicaid Services (CMS) designated 2017 as a transition year and has provided a clear pathway to avoid penalties. In addition, CMS has reduced the reporting requirements in 2017 for those who wish to fully participate in preparation for the future or those practices whose goal is the achievement of a positive payment update. It is important to note that the funds available for positive payment updates are derived from the penalties assessed on those who choose NOT to participate. Accordingly, by making it easier to avoid penalties in the first year, CMS has also reduced the amount of funds available for positive incentives.
Participating to avoid penalties
For 2017, CMS instituted options to allow surgeons to “Pick Your Pace” for participation in MIPS. Those who choose not to participate at any level will receive the full negative payment adjustment of 4% in 2019. However, it is noteworthy that a 4% negative payment adjustment is less than half of the negative adjustments associated with the PQRS, VM, and Meaningful Use programs in 2016.
To avoid the 4% penalty, CMS only requires that surgeons test their ability to report data in any of three reporting components, namely Quality, ACI or Improvement Activities. Information for the Cost component is derived automatically and has no reporting requirement. To avoid a penalty, surgeons must simply report one Quality measure for a single patient, attest to participating in an approved Improvement Activity for at least 90 consecutive days or complete the Base score requirements for ACI.
Participating to prepare for future success
Those who wish to attempt to achieve a higher score must report data for 50% of all patients seen (for ALL payors) for any consecutive 90 day period. Accordingly, one could begin as late as October 2, 2017. How data is reported depends upon the circumstances of an individual’s practice as there are multiple methods (electronic health record, registry, or qualified clinical data registry) for submitting data to CMS. It should be noted that data can also be submitted either on an individual basis or as a group.
Reporting pathway toward potentially receiving a positive payment update: Reporting for Quality
To receive the full potential Quality score, data must be submitted for 50% of all patients seen (for ALL payors) for any consecutive 90 day period on a minimum of 6 measures including one Outcome measure. Alternatively, one can choose to use a specialty measure set to report on 50% of all patients seen (for ALL payors) for any consecutive 90 day period. Those who do meet the reporting requirement and perform well on the measures will receive up to 60 points toward their MIPS Final Score. For those who intend to simply avoid penalties for the first year of the MIPS program, reporting a single measure for a single patient will earn the 3 points necessary to meet the threshold prescribed by CMS to avoid a penalty.
Reporting for ACI
The ACI component is worth 25 percent of the MIPS Final Score. The assessment for ACI is a composite score composed of two parts, a Base score and a Performance score. To receive credit for the ACI component 2017, one must have either 2014 or 2015 Edition CEHRT.
The Base score is an “all-or-nothing” threshold and accounts for 50 percent of the total score for the ACI component. Achievement of the Base score is required before any score can be accrued for the Performance portion. Achieving the Base score is also one of the options prescribed by CMS sufficient to avoid MIPS penalties in the first year and if the Base Score is achieved, one will not receive a penalty for 2017. The ACI measures are intended to ensure that certified EHRs are being used for core tasks such as providing patients with online access to their medical records, exchanging health information with patients and other providers, electronic prescribing and protecting sensitive patient health information.
Once all of the measures for the Base score have been met, clinicians are eligible to receive credit for performance on both a subset of the Base score measures and on a set of additional optional measures. Bonus points are also available by reporting certain Improvement Activities via a certified EHR.
Reporting for Improvement Activities
While the Improvement Activities (IA) is a new category, surgeons are familiar with many of the activities including maintenance of certification, use of the ACS Surgical Risk Calculator, participation in a QCDR and registry with their state’s prescription drug monitoring program. Each activity is assigned a point value of either 20 points (high value) or 10 points (medium value). The reporting requirement for the IA is fulfilled by simple attestation via either a registry, qualified clinical data registry, or a portal on the CMS website. To receive full credit, most surgeons must select and attest to having completed between two and four activities for a total of 40 points. Some surgeons in rural or small practices will only need to complete one high value or two medium value activities to achieve full credit. Those who fulfill the requirement will receive 15 points toward the MIPS Final Score. For those whose goal is simply to avoid a penalty in the first reporting year of MIPS, reporting a single activity for 90 days is enough to avoid any MIPS penalties for 2017
For those seeking further information, the ACS website (www.facs.org/qpp) has additional fact sheets and informational videos on the MIPS program.
Until next month …
Dr. Bailey is a pediatric surgeon, and Medical Director, Advocacy, for the Division of Advocacy and Health Policy in the ACS offices in Washington, D.C.
2017 is here and the new Merit-based Incentive Payment System (MIPS) is now in effect. MIPS has taken a number of steps to streamline reporting and make it easier to avoid penalties and achieve positive updates. However, over time penalties for non-participation or poor performance will grow. Therefore, it is critically important that all surgeons make a plan for how they can best participate in order to succeed. Knowing what options are available is vital to navigating the new reporting requirements and achieving the best possible financial outcome.
Background on MIPS and its components
MIPS began measuring performance in January 2017. The data reported in 2017 will be used to adjust payments in 2019. MIPS took the Physician Quality Reporting System (PQRS), the Value Based Modifier (VM), and the EHR Incentive Program commonly referred to as Meaningful Use (EHR-MU), added a new component that provides credit for Improvement Activities and combined them to derive a composite MIPS Final Score. The components of the Final Score are known as Quality, (formerly PQRS), Cost, (formerly VM), Advancing Care Information (ACI), (formerly EHR-MU), and Improvement Activities. The weights for the individual components of the final score for the first year of the MIPS program are represented in the chart above.
Though CMS has chosen not to provide any weight to the Cost component during the first year of the program, those who report Quality data will receive feedback reports on their performance in the Cost component.
2017: The transition year
The Centers for Medicare & Medicaid Services (CMS) designated 2017 as a transition year and has provided a clear pathway to avoid penalties. In addition, CMS has reduced the reporting requirements in 2017 for those who wish to fully participate in preparation for the future or those practices whose goal is the achievement of a positive payment update. It is important to note that the funds available for positive payment updates are derived from the penalties assessed on those who choose NOT to participate. Accordingly, by making it easier to avoid penalties in the first year, CMS has also reduced the amount of funds available for positive incentives.
Participating to avoid penalties
For 2017, CMS instituted options to allow surgeons to “Pick Your Pace” for participation in MIPS. Those who choose not to participate at any level will receive the full negative payment adjustment of 4% in 2019. However, it is noteworthy that a 4% negative payment adjustment is less than half of the negative adjustments associated with the PQRS, VM, and Meaningful Use programs in 2016.
To avoid the 4% penalty, CMS only requires that surgeons test their ability to report data in any of three reporting components, namely Quality, ACI or Improvement Activities. Information for the Cost component is derived automatically and has no reporting requirement. To avoid a penalty, surgeons must simply report one Quality measure for a single patient, attest to participating in an approved Improvement Activity for at least 90 consecutive days or complete the Base score requirements for ACI.
Participating to prepare for future success
Those who wish to attempt to achieve a higher score must report data for 50% of all patients seen (for ALL payors) for any consecutive 90 day period. Accordingly, one could begin as late as October 2, 2017. How data is reported depends upon the circumstances of an individual’s practice as there are multiple methods (electronic health record, registry, or qualified clinical data registry) for submitting data to CMS. It should be noted that data can also be submitted either on an individual basis or as a group.
Reporting pathway toward potentially receiving a positive payment update: Reporting for Quality
To receive the full potential Quality score, data must be submitted for 50% of all patients seen (for ALL payors) for any consecutive 90 day period on a minimum of 6 measures including one Outcome measure. Alternatively, one can choose to use a specialty measure set to report on 50% of all patients seen (for ALL payors) for any consecutive 90 day period. Those who do meet the reporting requirement and perform well on the measures will receive up to 60 points toward their MIPS Final Score. For those who intend to simply avoid penalties for the first year of the MIPS program, reporting a single measure for a single patient will earn the 3 points necessary to meet the threshold prescribed by CMS to avoid a penalty.
Reporting for ACI
The ACI component is worth 25 percent of the MIPS Final Score. The assessment for ACI is a composite score composed of two parts, a Base score and a Performance score. To receive credit for the ACI component 2017, one must have either 2014 or 2015 Edition CEHRT.
The Base score is an “all-or-nothing” threshold and accounts for 50 percent of the total score for the ACI component. Achievement of the Base score is required before any score can be accrued for the Performance portion. Achieving the Base score is also one of the options prescribed by CMS sufficient to avoid MIPS penalties in the first year and if the Base Score is achieved, one will not receive a penalty for 2017. The ACI measures are intended to ensure that certified EHRs are being used for core tasks such as providing patients with online access to their medical records, exchanging health information with patients and other providers, electronic prescribing and protecting sensitive patient health information.
Once all of the measures for the Base score have been met, clinicians are eligible to receive credit for performance on both a subset of the Base score measures and on a set of additional optional measures. Bonus points are also available by reporting certain Improvement Activities via a certified EHR.
Reporting for Improvement Activities
While the Improvement Activities (IA) is a new category, surgeons are familiar with many of the activities including maintenance of certification, use of the ACS Surgical Risk Calculator, participation in a QCDR and registry with their state’s prescription drug monitoring program. Each activity is assigned a point value of either 20 points (high value) or 10 points (medium value). The reporting requirement for the IA is fulfilled by simple attestation via either a registry, qualified clinical data registry, or a portal on the CMS website. To receive full credit, most surgeons must select and attest to having completed between two and four activities for a total of 40 points. Some surgeons in rural or small practices will only need to complete one high value or two medium value activities to achieve full credit. Those who fulfill the requirement will receive 15 points toward the MIPS Final Score. For those whose goal is simply to avoid a penalty in the first reporting year of MIPS, reporting a single activity for 90 days is enough to avoid any MIPS penalties for 2017
For those seeking further information, the ACS website (www.facs.org/qpp) has additional fact sheets and informational videos on the MIPS program.
Until next month …
Dr. Bailey is a pediatric surgeon, and Medical Director, Advocacy, for the Division of Advocacy and Health Policy in the ACS offices in Washington, D.C.
2017 is here and the new Merit-based Incentive Payment System (MIPS) is now in effect. MIPS has taken a number of steps to streamline reporting and make it easier to avoid penalties and achieve positive updates. However, over time penalties for non-participation or poor performance will grow. Therefore, it is critically important that all surgeons make a plan for how they can best participate in order to succeed. Knowing what options are available is vital to navigating the new reporting requirements and achieving the best possible financial outcome.
Background on MIPS and its components
MIPS began measuring performance in January 2017. The data reported in 2017 will be used to adjust payments in 2019. MIPS took the Physician Quality Reporting System (PQRS), the Value Based Modifier (VM), and the EHR Incentive Program commonly referred to as Meaningful Use (EHR-MU), added a new component that provides credit for Improvement Activities and combined them to derive a composite MIPS Final Score. The components of the Final Score are known as Quality, (formerly PQRS), Cost, (formerly VM), Advancing Care Information (ACI), (formerly EHR-MU), and Improvement Activities. The weights for the individual components of the final score for the first year of the MIPS program are represented in the chart above.
Though CMS has chosen not to provide any weight to the Cost component during the first year of the program, those who report Quality data will receive feedback reports on their performance in the Cost component.
2017: The transition year
The Centers for Medicare & Medicaid Services (CMS) designated 2017 as a transition year and has provided a clear pathway to avoid penalties. In addition, CMS has reduced the reporting requirements in 2017 for those who wish to fully participate in preparation for the future or those practices whose goal is the achievement of a positive payment update. It is important to note that the funds available for positive payment updates are derived from the penalties assessed on those who choose NOT to participate. Accordingly, by making it easier to avoid penalties in the first year, CMS has also reduced the amount of funds available for positive incentives.
Participating to avoid penalties
For 2017, CMS instituted options to allow surgeons to “Pick Your Pace” for participation in MIPS. Those who choose not to participate at any level will receive the full negative payment adjustment of 4% in 2019. However, it is noteworthy that a 4% negative payment adjustment is less than half of the negative adjustments associated with the PQRS, VM, and Meaningful Use programs in 2016.
To avoid the 4% penalty, CMS only requires that surgeons test their ability to report data in any of three reporting components, namely Quality, ACI or Improvement Activities. Information for the Cost component is derived automatically and has no reporting requirement. To avoid a penalty, surgeons must simply report one Quality measure for a single patient, attest to participating in an approved Improvement Activity for at least 90 consecutive days or complete the Base score requirements for ACI.
Participating to prepare for future success
Those who wish to attempt to achieve a higher score must report data for 50% of all patients seen (for ALL payors) for any consecutive 90 day period. Accordingly, one could begin as late as October 2, 2017. How data is reported depends upon the circumstances of an individual’s practice as there are multiple methods (electronic health record, registry, or qualified clinical data registry) for submitting data to CMS. It should be noted that data can also be submitted either on an individual basis or as a group.
Reporting pathway toward potentially receiving a positive payment update: Reporting for Quality
To receive the full potential Quality score, data must be submitted for 50% of all patients seen (for ALL payors) for any consecutive 90 day period on a minimum of 6 measures including one Outcome measure. Alternatively, one can choose to use a specialty measure set to report on 50% of all patients seen (for ALL payors) for any consecutive 90 day period. Those who do meet the reporting requirement and perform well on the measures will receive up to 60 points toward their MIPS Final Score. For those who intend to simply avoid penalties for the first year of the MIPS program, reporting a single measure for a single patient will earn the 3 points necessary to meet the threshold prescribed by CMS to avoid a penalty.
Reporting for ACI
The ACI component is worth 25 percent of the MIPS Final Score. The assessment for ACI is a composite score composed of two parts, a Base score and a Performance score. To receive credit for the ACI component 2017, one must have either 2014 or 2015 Edition CEHRT.
The Base score is an “all-or-nothing” threshold and accounts for 50 percent of the total score for the ACI component. Achievement of the Base score is required before any score can be accrued for the Performance portion. Achieving the Base score is also one of the options prescribed by CMS sufficient to avoid MIPS penalties in the first year and if the Base Score is achieved, one will not receive a penalty for 2017. The ACI measures are intended to ensure that certified EHRs are being used for core tasks such as providing patients with online access to their medical records, exchanging health information with patients and other providers, electronic prescribing and protecting sensitive patient health information.
Once all of the measures for the Base score have been met, clinicians are eligible to receive credit for performance on both a subset of the Base score measures and on a set of additional optional measures. Bonus points are also available by reporting certain Improvement Activities via a certified EHR.
Reporting for Improvement Activities
While the Improvement Activities (IA) is a new category, surgeons are familiar with many of the activities including maintenance of certification, use of the ACS Surgical Risk Calculator, participation in a QCDR and registry with their state’s prescription drug monitoring program. Each activity is assigned a point value of either 20 points (high value) or 10 points (medium value). The reporting requirement for the IA is fulfilled by simple attestation via either a registry, qualified clinical data registry, or a portal on the CMS website. To receive full credit, most surgeons must select and attest to having completed between two and four activities for a total of 40 points. Some surgeons in rural or small practices will only need to complete one high value or two medium value activities to achieve full credit. Those who fulfill the requirement will receive 15 points toward the MIPS Final Score. For those whose goal is simply to avoid a penalty in the first reporting year of MIPS, reporting a single activity for 90 days is enough to avoid any MIPS penalties for 2017
For those seeking further information, the ACS website (www.facs.org/qpp) has additional fact sheets and informational videos on the MIPS program.
Until next month …
Dr. Bailey is a pediatric surgeon, and Medical Director, Advocacy, for the Division of Advocacy and Health Policy in the ACS offices in Washington, D.C.
SAVR an option for elderly with aortic stenosis
HOUSTON – Surgical aortic valve replacement can be performed in intermediate-risk elderly patients with an operative mortality rate of 4.1%, which is better than expected, according to results from a large multicenter analysis. However, the rate of in-hospital stroke was 5.4% – twice what was expected.
“This is most likely secondary to neurologic assessment [that was conducted] for all patients postoperatively,” Vinod H. Thourani, MD, said at the annual meeting of the Society of Thoracic Surgeons.
The findings come from an in-depth analysis of SAVR outcomes in patients who participated in the Placement of Aortic Transcatheter Valves trial, known as PARTNER 2A. Conducted from December 2011 to November 2013, PARTNER 2A evaluated 2,032 medium-risk patients with aortic stenosis who were randomized to SAVR or transcatheter aortic valve replacement (TAVR) in 57 North American centers and found no significant difference in the 2-year rate of death or disabling stroke (N Engl J Med. 2016 Apr 28;3749[17]:1609-20).
Dr. Thourani’s analysis focused on the 937 patients who underwent SAVR. The main objectives were to describe operative mortality and hospital morbidities compared with STS benchmarks, describe time-related mortality and stroke including preoperative predictors for these outcomes, evaluate the effect of concomitant procedures on mortality and hospital morbidities, and evaluate longitudinal valve performance after SAVR.
The average age of these patients was 82 years, 45% were female, and their mean STS risk score was 5.8. In addition, 26% had prior coronary artery bypass surgery, 10% had a previous stroke, and 12% had previous pacemaker placement. Of the 30% of patients with chronic obstructive pulmonary disease, 9.6% were oxygen dependent going into the operating room, reported Dr. Thourani, one of the PARTNER 2A investigators, and a cardiothoracic surgeon at Emory University, Atlanta.
Most of the patients (85%) had a full sternotomy, while 15% had a mini sternotomy. Isolated AVR was done in 79% of patients, 15% of patients had AVR plus CABG, and 6% had AVR and other concomitant procedures. The mean coronary bypass time for isolated AVR was 98 minutes, and rose to a mean of 129 minutes when a concomitant procedure was added. The mean cross-clamp time was 69 minutes, and rose to a mean of 95 minutes when a concomitant procedure was added.
The investigators observed that all-cause operative mortality was 4.1%, which is lower than STS predicted-risk models. At the same time, mortality for AVR plus a concomitant procedure was 5%, followed by isolated AVR (4.2%) and AVR plus CABG plus a concomitant procedure (2.9%). The rate of in-hospital stroke was 5.4% and the rate of in-hospital deep sternal wound infection was 0.8%. At 2 years postoperatively, mortality was 17% among those who underwent isolated AVR, 18% among those who underwent AVR plus CABG, and 21% among those who underwent AVR plus a concomitant procedure, differences that did not reach statistical significance. The rate of stroke at 2 years also was similar between groups: 12% among those who underwent isolated AVR, 11% in those who underwent AVR plus a concomitant procedure, and 8.2% in those who underwent AVR plus CABG.
The main risk factor for early death after SAVR was longer procedure time (P less than .0001), while risk factors for later deaths included cachexia (P = .02), lower ejection fraction (P = .01), higher creatinine (P = .03), coronary artery disease (P = .03), and smaller prostheses (P = .01)
Dr. Thourani and his associates also found that 33% of patients had severe prosthesis-patient mismatch, yet they had survival rates similar to the rates of those without severe prosthesis-patient mismatch.
“From this adjudicated, prospectively collected data in the contemporary era, SAVR can be performed in intermediate-risk elderly patients with mortality commensurate with national benchmarks,” he concluded. “Continued surveillance of these patients remains extremely important.”
Dr. Thourani disclosed that he is a consultant for and has received research support from Edwards Lifesciences. Other authors of the study reported having numerous relevant financial disclosures.
This analysis of the surgical arm of the PARTNER 2A trial reveals respectable outcome for those so-called intermediaterisk patients with severe symptomatic aortic stenosis. The fact that mortality at 2 years was similar between the surgical and the catheter arm of the trial (upward of 17%), speaks of the multiple comorbidities present in these patients (N Engl J Med.
This analysis of the surgical arm of the PARTNER 2A trial reveals respectable outcome for those so-called intermediaterisk patients with severe symptomatic aortic stenosis. The fact that mortality at 2 years was similar between the surgical and the catheter arm of the trial (upward of 17%), speaks of the multiple comorbidities present in these patients (N Engl J Med.
This analysis of the surgical arm of the PARTNER 2A trial reveals respectable outcome for those so-called intermediaterisk patients with severe symptomatic aortic stenosis. The fact that mortality at 2 years was similar between the surgical and the catheter arm of the trial (upward of 17%), speaks of the multiple comorbidities present in these patients (N Engl J Med.
HOUSTON – Surgical aortic valve replacement can be performed in intermediate-risk elderly patients with an operative mortality rate of 4.1%, which is better than expected, according to results from a large multicenter analysis. However, the rate of in-hospital stroke was 5.4% – twice what was expected.
“This is most likely secondary to neurologic assessment [that was conducted] for all patients postoperatively,” Vinod H. Thourani, MD, said at the annual meeting of the Society of Thoracic Surgeons.
The findings come from an in-depth analysis of SAVR outcomes in patients who participated in the Placement of Aortic Transcatheter Valves trial, known as PARTNER 2A. Conducted from December 2011 to November 2013, PARTNER 2A evaluated 2,032 medium-risk patients with aortic stenosis who were randomized to SAVR or transcatheter aortic valve replacement (TAVR) in 57 North American centers and found no significant difference in the 2-year rate of death or disabling stroke (N Engl J Med. 2016 Apr 28;3749[17]:1609-20).
Dr. Thourani’s analysis focused on the 937 patients who underwent SAVR. The main objectives were to describe operative mortality and hospital morbidities compared with STS benchmarks, describe time-related mortality and stroke including preoperative predictors for these outcomes, evaluate the effect of concomitant procedures on mortality and hospital morbidities, and evaluate longitudinal valve performance after SAVR.
The average age of these patients was 82 years, 45% were female, and their mean STS risk score was 5.8. In addition, 26% had prior coronary artery bypass surgery, 10% had a previous stroke, and 12% had previous pacemaker placement. Of the 30% of patients with chronic obstructive pulmonary disease, 9.6% were oxygen dependent going into the operating room, reported Dr. Thourani, one of the PARTNER 2A investigators, and a cardiothoracic surgeon at Emory University, Atlanta.
Most of the patients (85%) had a full sternotomy, while 15% had a mini sternotomy. Isolated AVR was done in 79% of patients, 15% of patients had AVR plus CABG, and 6% had AVR and other concomitant procedures. The mean coronary bypass time for isolated AVR was 98 minutes, and rose to a mean of 129 minutes when a concomitant procedure was added. The mean cross-clamp time was 69 minutes, and rose to a mean of 95 minutes when a concomitant procedure was added.
The investigators observed that all-cause operative mortality was 4.1%, which is lower than STS predicted-risk models. At the same time, mortality for AVR plus a concomitant procedure was 5%, followed by isolated AVR (4.2%) and AVR plus CABG plus a concomitant procedure (2.9%). The rate of in-hospital stroke was 5.4% and the rate of in-hospital deep sternal wound infection was 0.8%. At 2 years postoperatively, mortality was 17% among those who underwent isolated AVR, 18% among those who underwent AVR plus CABG, and 21% among those who underwent AVR plus a concomitant procedure, differences that did not reach statistical significance. The rate of stroke at 2 years also was similar between groups: 12% among those who underwent isolated AVR, 11% in those who underwent AVR plus a concomitant procedure, and 8.2% in those who underwent AVR plus CABG.
The main risk factor for early death after SAVR was longer procedure time (P less than .0001), while risk factors for later deaths included cachexia (P = .02), lower ejection fraction (P = .01), higher creatinine (P = .03), coronary artery disease (P = .03), and smaller prostheses (P = .01)
Dr. Thourani and his associates also found that 33% of patients had severe prosthesis-patient mismatch, yet they had survival rates similar to the rates of those without severe prosthesis-patient mismatch.
“From this adjudicated, prospectively collected data in the contemporary era, SAVR can be performed in intermediate-risk elderly patients with mortality commensurate with national benchmarks,” he concluded. “Continued surveillance of these patients remains extremely important.”
Dr. Thourani disclosed that he is a consultant for and has received research support from Edwards Lifesciences. Other authors of the study reported having numerous relevant financial disclosures.
HOUSTON – Surgical aortic valve replacement can be performed in intermediate-risk elderly patients with an operative mortality rate of 4.1%, which is better than expected, according to results from a large multicenter analysis. However, the rate of in-hospital stroke was 5.4% – twice what was expected.
“This is most likely secondary to neurologic assessment [that was conducted] for all patients postoperatively,” Vinod H. Thourani, MD, said at the annual meeting of the Society of Thoracic Surgeons.
The findings come from an in-depth analysis of SAVR outcomes in patients who participated in the Placement of Aortic Transcatheter Valves trial, known as PARTNER 2A. Conducted from December 2011 to November 2013, PARTNER 2A evaluated 2,032 medium-risk patients with aortic stenosis who were randomized to SAVR or transcatheter aortic valve replacement (TAVR) in 57 North American centers and found no significant difference in the 2-year rate of death or disabling stroke (N Engl J Med. 2016 Apr 28;3749[17]:1609-20).
Dr. Thourani’s analysis focused on the 937 patients who underwent SAVR. The main objectives were to describe operative mortality and hospital morbidities compared with STS benchmarks, describe time-related mortality and stroke including preoperative predictors for these outcomes, evaluate the effect of concomitant procedures on mortality and hospital morbidities, and evaluate longitudinal valve performance after SAVR.
The average age of these patients was 82 years, 45% were female, and their mean STS risk score was 5.8. In addition, 26% had prior coronary artery bypass surgery, 10% had a previous stroke, and 12% had previous pacemaker placement. Of the 30% of patients with chronic obstructive pulmonary disease, 9.6% were oxygen dependent going into the operating room, reported Dr. Thourani, one of the PARTNER 2A investigators, and a cardiothoracic surgeon at Emory University, Atlanta.
Most of the patients (85%) had a full sternotomy, while 15% had a mini sternotomy. Isolated AVR was done in 79% of patients, 15% of patients had AVR plus CABG, and 6% had AVR and other concomitant procedures. The mean coronary bypass time for isolated AVR was 98 minutes, and rose to a mean of 129 minutes when a concomitant procedure was added. The mean cross-clamp time was 69 minutes, and rose to a mean of 95 minutes when a concomitant procedure was added.
The investigators observed that all-cause operative mortality was 4.1%, which is lower than STS predicted-risk models. At the same time, mortality for AVR plus a concomitant procedure was 5%, followed by isolated AVR (4.2%) and AVR plus CABG plus a concomitant procedure (2.9%). The rate of in-hospital stroke was 5.4% and the rate of in-hospital deep sternal wound infection was 0.8%. At 2 years postoperatively, mortality was 17% among those who underwent isolated AVR, 18% among those who underwent AVR plus CABG, and 21% among those who underwent AVR plus a concomitant procedure, differences that did not reach statistical significance. The rate of stroke at 2 years also was similar between groups: 12% among those who underwent isolated AVR, 11% in those who underwent AVR plus a concomitant procedure, and 8.2% in those who underwent AVR plus CABG.
The main risk factor for early death after SAVR was longer procedure time (P less than .0001), while risk factors for later deaths included cachexia (P = .02), lower ejection fraction (P = .01), higher creatinine (P = .03), coronary artery disease (P = .03), and smaller prostheses (P = .01)
Dr. Thourani and his associates also found that 33% of patients had severe prosthesis-patient mismatch, yet they had survival rates similar to the rates of those without severe prosthesis-patient mismatch.
“From this adjudicated, prospectively collected data in the contemporary era, SAVR can be performed in intermediate-risk elderly patients with mortality commensurate with national benchmarks,” he concluded. “Continued surveillance of these patients remains extremely important.”
Dr. Thourani disclosed that he is a consultant for and has received research support from Edwards Lifesciences. Other authors of the study reported having numerous relevant financial disclosures.
AT THE STS ANNUAL MEETING
Key clinical point:
Major finding: All-cause operative mortality was 4.1%, which is lower than STS predicted risk models.
Data source: A study of 937 medium-risk patients with aortic stenosis who were randomized to SAVR in the PARTNER 2A trial.
Disclosures: Dr. Thourani is a consultant for and has received research support from Edwards Lifesciences. Other authors of the study reported having numerous relevant financial disclosures.
Pairing vascular reconstruction, pancreatic cancer resection
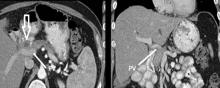
CHICAGO – More than 53,000 people will develop pancreatic ductal adenocarcinoma in the United States this year, and upwards of 41,000 will die from the disease, many of them with tumors considered unresectable because they involve adjacent vessels. However, researchers at the University of California, Irvine, have found that careful removal of the tumor around involved veins and arteries, even in borderline cases, can improve outcomes for these patients.
Roy M. Fujitani, MD, updated previously published data on a single-center study he coauthored in 2015 of 270 patients who had undergone a Whipple operation, 183 for pancreatic adenocarcinoma (J Vasc Surg. 2015;61:475-80) at a symposium on vascular surgery sponsored by Northwestern University.
Resection of pancreatic tumors without vascular involvement is fairly straightforward for surgical oncologists to perform, Dr. Fujitani said, but pancreatic tumors enter the borderline resectable category when preoperative CT scan shows portal vein abutment, for which vascular surgery should provide counsel and assist. However, even in some cases when preoperative CT scan shows unresectable, locally advanced pancreatic tumor with celiac artery encasement, neoadjuvant therapy may downstage the disease into the borderline category, he said.
“Patients with borderline resectable or stage II disease are those one should consider for reconstruction,” Dr. Fujitani said. Resectable findings of borderline disease include encasement of the portal vein, superior mesenteric vein and the confluence of the portal venous system (with suitable proximal and distal targets for reconstruction); and less-than-circumferential involvement of the common hepatic artery or right hepatic artery – but without involvement of the superior mesenteric artery or the celiac axis and “certainly not” the aorta. “This would account for about one-fourth of patients in high-volume centers as being able to receive concomitant vascular reconstruction,” Dr. Fujitani said.
In the UCI series, 60 patients with borderline lesions underwent vascular reconstruction. “As it turned out, there was no significant difference in survival between the reconstruction group and the nonreconstruction group,” Dr. Fujitani said, “but it’s important to note that these patients who had the reconstruction would never have been operated on if we were not able to do the reconstruction.” Thirty-day mortality was around 5% and 1-year survival around 70% in both groups, he said. However, at about 1.5 years the Kaplan-Meier survival curves between the two groups diverged, which Dr. Fujitani attributed to more advanced disease in the reconstruction group.
“We found lymph node status and tumor margins were most important in determining survival of these patients,” he said. “Gaining an R0 resection is the most important thing that determines favorable survivability.”
Dr. Fujitani also reviewed different techniques for vascular reconstruction, and while differences in complication rates or 1-, 2-, or 3-year survival were not statistically significant, he did note that mean survival with lateral venorrhaphy exceeded that of primary anastomosis and interposition graft – 21 months vs. 13 months vs. 4 months, suggesting the merits of a more aggressive approach to vascular resection and reconstruction.
“Improvement of survival outcomes may be achieved with concomitant advanced vascular reconstruction in carefully selected patients,” Dr. Fujitani said. “There are multiple options for vascular reconstruction for mesenteric portal venous and visceral arterial involvement using standard vascular surgical techniques.” He added that a dedicated team of experienced surgical oncologists and vascular surgeons for these reconstructions “is essential for successful outcomes.”
Dr. Fujitani had no relevant financial relationships to disclose.
CHICAGO – More than 53,000 people will develop pancreatic ductal adenocarcinoma in the United States this year, and upwards of 41,000 will die from the disease, many of them with tumors considered unresectable because they involve adjacent vessels. However, researchers at the University of California, Irvine, have found that careful removal of the tumor around involved veins and arteries, even in borderline cases, can improve outcomes for these patients.
Roy M. Fujitani, MD, updated previously published data on a single-center study he coauthored in 2015 of 270 patients who had undergone a Whipple operation, 183 for pancreatic adenocarcinoma (J Vasc Surg. 2015;61:475-80) at a symposium on vascular surgery sponsored by Northwestern University.
Resection of pancreatic tumors without vascular involvement is fairly straightforward for surgical oncologists to perform, Dr. Fujitani said, but pancreatic tumors enter the borderline resectable category when preoperative CT scan shows portal vein abutment, for which vascular surgery should provide counsel and assist. However, even in some cases when preoperative CT scan shows unresectable, locally advanced pancreatic tumor with celiac artery encasement, neoadjuvant therapy may downstage the disease into the borderline category, he said.
“Patients with borderline resectable or stage II disease are those one should consider for reconstruction,” Dr. Fujitani said. Resectable findings of borderline disease include encasement of the portal vein, superior mesenteric vein and the confluence of the portal venous system (with suitable proximal and distal targets for reconstruction); and less-than-circumferential involvement of the common hepatic artery or right hepatic artery – but without involvement of the superior mesenteric artery or the celiac axis and “certainly not” the aorta. “This would account for about one-fourth of patients in high-volume centers as being able to receive concomitant vascular reconstruction,” Dr. Fujitani said.
In the UCI series, 60 patients with borderline lesions underwent vascular reconstruction. “As it turned out, there was no significant difference in survival between the reconstruction group and the nonreconstruction group,” Dr. Fujitani said, “but it’s important to note that these patients who had the reconstruction would never have been operated on if we were not able to do the reconstruction.” Thirty-day mortality was around 5% and 1-year survival around 70% in both groups, he said. However, at about 1.5 years the Kaplan-Meier survival curves between the two groups diverged, which Dr. Fujitani attributed to more advanced disease in the reconstruction group.
“We found lymph node status and tumor margins were most important in determining survival of these patients,” he said. “Gaining an R0 resection is the most important thing that determines favorable survivability.”
Dr. Fujitani also reviewed different techniques for vascular reconstruction, and while differences in complication rates or 1-, 2-, or 3-year survival were not statistically significant, he did note that mean survival with lateral venorrhaphy exceeded that of primary anastomosis and interposition graft – 21 months vs. 13 months vs. 4 months, suggesting the merits of a more aggressive approach to vascular resection and reconstruction.
“Improvement of survival outcomes may be achieved with concomitant advanced vascular reconstruction in carefully selected patients,” Dr. Fujitani said. “There are multiple options for vascular reconstruction for mesenteric portal venous and visceral arterial involvement using standard vascular surgical techniques.” He added that a dedicated team of experienced surgical oncologists and vascular surgeons for these reconstructions “is essential for successful outcomes.”
Dr. Fujitani had no relevant financial relationships to disclose.
CHICAGO – More than 53,000 people will develop pancreatic ductal adenocarcinoma in the United States this year, and upwards of 41,000 will die from the disease, many of them with tumors considered unresectable because they involve adjacent vessels. However, researchers at the University of California, Irvine, have found that careful removal of the tumor around involved veins and arteries, even in borderline cases, can improve outcomes for these patients.
Roy M. Fujitani, MD, updated previously published data on a single-center study he coauthored in 2015 of 270 patients who had undergone a Whipple operation, 183 for pancreatic adenocarcinoma (J Vasc Surg. 2015;61:475-80) at a symposium on vascular surgery sponsored by Northwestern University.
Resection of pancreatic tumors without vascular involvement is fairly straightforward for surgical oncologists to perform, Dr. Fujitani said, but pancreatic tumors enter the borderline resectable category when preoperative CT scan shows portal vein abutment, for which vascular surgery should provide counsel and assist. However, even in some cases when preoperative CT scan shows unresectable, locally advanced pancreatic tumor with celiac artery encasement, neoadjuvant therapy may downstage the disease into the borderline category, he said.
“Patients with borderline resectable or stage II disease are those one should consider for reconstruction,” Dr. Fujitani said. Resectable findings of borderline disease include encasement of the portal vein, superior mesenteric vein and the confluence of the portal venous system (with suitable proximal and distal targets for reconstruction); and less-than-circumferential involvement of the common hepatic artery or right hepatic artery – but without involvement of the superior mesenteric artery or the celiac axis and “certainly not” the aorta. “This would account for about one-fourth of patients in high-volume centers as being able to receive concomitant vascular reconstruction,” Dr. Fujitani said.
In the UCI series, 60 patients with borderline lesions underwent vascular reconstruction. “As it turned out, there was no significant difference in survival between the reconstruction group and the nonreconstruction group,” Dr. Fujitani said, “but it’s important to note that these patients who had the reconstruction would never have been operated on if we were not able to do the reconstruction.” Thirty-day mortality was around 5% and 1-year survival around 70% in both groups, he said. However, at about 1.5 years the Kaplan-Meier survival curves between the two groups diverged, which Dr. Fujitani attributed to more advanced disease in the reconstruction group.
“We found lymph node status and tumor margins were most important in determining survival of these patients,” he said. “Gaining an R0 resection is the most important thing that determines favorable survivability.”
Dr. Fujitani also reviewed different techniques for vascular reconstruction, and while differences in complication rates or 1-, 2-, or 3-year survival were not statistically significant, he did note that mean survival with lateral venorrhaphy exceeded that of primary anastomosis and interposition graft – 21 months vs. 13 months vs. 4 months, suggesting the merits of a more aggressive approach to vascular resection and reconstruction.
“Improvement of survival outcomes may be achieved with concomitant advanced vascular reconstruction in carefully selected patients,” Dr. Fujitani said. “There are multiple options for vascular reconstruction for mesenteric portal venous and visceral arterial involvement using standard vascular surgical techniques.” He added that a dedicated team of experienced surgical oncologists and vascular surgeons for these reconstructions “is essential for successful outcomes.”
Dr. Fujitani had no relevant financial relationships to disclose.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Key clinical point: A more aggressive vascular resection and reconstruction in pancreatic cancer may improve outcomes and palliation in these patients.
Major finding: Mean survival with lateral venorrhaphy exceeded primary anastomosis and interposition graft (21 months vs. 13 months vs. 4 months).
Data source: Updated data of previously published single-center retrospective review of 183 patients who had Whipple procedure for pancreatic adenocarcinoma.
Disclosures: Dr. Fujitani reported having no financial disclosures.
VQI confirms improvements in vascular practice
CHICAGO – Five years after the Society for Vascular Surgery launched the Vascular Quality Initiative, participating centers are more likely to use chlorhexidine and have also cut their surgery times and reduced their transfusion rates, according to results presented at a symposium on vascular surgery sponsored by Northwestern University.
But more drastic have been the improvements once low-performing centers have made in these measures and others, Larry Kraiss, MD, of the University of Utah, Salt Lake City, said in reporting an update on VQI. “If you look at centers that had a big change in not using chlorhexidine to using chlorhexidine, the reduction of surgical site infections [SSI] in that subgroup was actually pretty significant,” said Dr. Kraiss, chair of the governing council of the SVS Patient Safety Organization, which oversees VQI.
These pivotal improvements came about after the VQI distributed what it calls COPI reports – for Center Opportunity Profile for Improvement – to participating centers. Currently, 379 centers in 46 states and Ontario participate in VQI, feeding data into 12 different vascular procedure registries ranging from peripheral vascular interventions to lower-extremity amputations. As of Nov. 1, 2016, 330,400 procedures had been submitted to VQI.
Dr. Kraiss called the COPI report the “workhorse” of the VQI. “It can give participating centers insight into what they can do to improve outcomes,” he said. It is one of three types of reports VQI provides. The others are benchmarking reports that show the masked ratings for all participating centers but confidentially highlight the rating of the individual center receiving the report; and reports for individual providers.
The most recent readout of the SSI COPI report compared measures in two periods: 2011-2012 and 2013-2014. In those periods, overall use of chlorhexidine rose from 66.6% to 81.2%; transfusion rates of more than 2 units fell from 14.4% to 11.5%; the share of procedures lasting 220 minutes or more fell from 50.2% to 47.7%; and SSI rate overall fell from 3.4% to 3.1%. While the change in SSI was not statistically significant, Dr. Kraiss said the 17 centers that had a large increase in chlorhexidine use did see statistically significant declines in SSI.
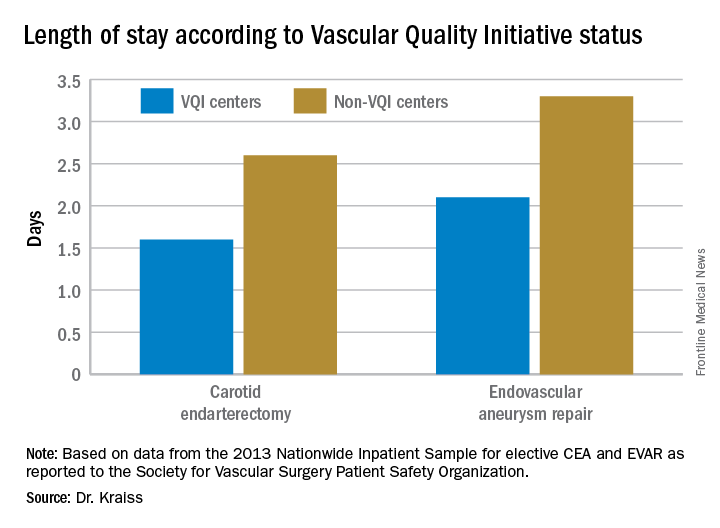
VQI also showed a 5-year survival rate of 79% of patients discharged with both statin and aspirin therapy vs. 61% for patients discharged without (J Vasc Surg. 2015;61[4]:1010-9). “This represents an opportunity to inform individual providers about how often they discharge patients on an aspirin and statin,” Dr. Kraiss said. Provider-targeted reports show how individual physicians rate in their region and nationwide.
VQI is more than a registry, Dr. Kraiss said; it’s also organized into 17 regional quality groups that provide surgeons a safe place to discuss VQI data and how to use that to encourage best practices. “There’s no risk of compromising or making the information identifiable,” he said. “It’s a matter of just getting together and trying to share best practices in a relatively informal environment, and hopefully through that drive quality improvement.
Other benefits of participating in VQI are that it can help surgeons comply with requirements for Medicare’s Merit-based Incentive Payment System (MIPS). VQI also offers opportunities to enroll in industry-sponsored clinical trials, which can help defray the cost of VQI participation, he said.
Dr. Kraiss had no relevant financial relationships to disclose.
CHICAGO – Five years after the Society for Vascular Surgery launched the Vascular Quality Initiative, participating centers are more likely to use chlorhexidine and have also cut their surgery times and reduced their transfusion rates, according to results presented at a symposium on vascular surgery sponsored by Northwestern University.
But more drastic have been the improvements once low-performing centers have made in these measures and others, Larry Kraiss, MD, of the University of Utah, Salt Lake City, said in reporting an update on VQI. “If you look at centers that had a big change in not using chlorhexidine to using chlorhexidine, the reduction of surgical site infections [SSI] in that subgroup was actually pretty significant,” said Dr. Kraiss, chair of the governing council of the SVS Patient Safety Organization, which oversees VQI.
These pivotal improvements came about after the VQI distributed what it calls COPI reports – for Center Opportunity Profile for Improvement – to participating centers. Currently, 379 centers in 46 states and Ontario participate in VQI, feeding data into 12 different vascular procedure registries ranging from peripheral vascular interventions to lower-extremity amputations. As of Nov. 1, 2016, 330,400 procedures had been submitted to VQI.
Dr. Kraiss called the COPI report the “workhorse” of the VQI. “It can give participating centers insight into what they can do to improve outcomes,” he said. It is one of three types of reports VQI provides. The others are benchmarking reports that show the masked ratings for all participating centers but confidentially highlight the rating of the individual center receiving the report; and reports for individual providers.
The most recent readout of the SSI COPI report compared measures in two periods: 2011-2012 and 2013-2014. In those periods, overall use of chlorhexidine rose from 66.6% to 81.2%; transfusion rates of more than 2 units fell from 14.4% to 11.5%; the share of procedures lasting 220 minutes or more fell from 50.2% to 47.7%; and SSI rate overall fell from 3.4% to 3.1%. While the change in SSI was not statistically significant, Dr. Kraiss said the 17 centers that had a large increase in chlorhexidine use did see statistically significant declines in SSI.

VQI also showed a 5-year survival rate of 79% of patients discharged with both statin and aspirin therapy vs. 61% for patients discharged without (J Vasc Surg. 2015;61[4]:1010-9). “This represents an opportunity to inform individual providers about how often they discharge patients on an aspirin and statin,” Dr. Kraiss said. Provider-targeted reports show how individual physicians rate in their region and nationwide.
VQI is more than a registry, Dr. Kraiss said; it’s also organized into 17 regional quality groups that provide surgeons a safe place to discuss VQI data and how to use that to encourage best practices. “There’s no risk of compromising or making the information identifiable,” he said. “It’s a matter of just getting together and trying to share best practices in a relatively informal environment, and hopefully through that drive quality improvement.
Other benefits of participating in VQI are that it can help surgeons comply with requirements for Medicare’s Merit-based Incentive Payment System (MIPS). VQI also offers opportunities to enroll in industry-sponsored clinical trials, which can help defray the cost of VQI participation, he said.
Dr. Kraiss had no relevant financial relationships to disclose.
CHICAGO – Five years after the Society for Vascular Surgery launched the Vascular Quality Initiative, participating centers are more likely to use chlorhexidine and have also cut their surgery times and reduced their transfusion rates, according to results presented at a symposium on vascular surgery sponsored by Northwestern University.
But more drastic have been the improvements once low-performing centers have made in these measures and others, Larry Kraiss, MD, of the University of Utah, Salt Lake City, said in reporting an update on VQI. “If you look at centers that had a big change in not using chlorhexidine to using chlorhexidine, the reduction of surgical site infections [SSI] in that subgroup was actually pretty significant,” said Dr. Kraiss, chair of the governing council of the SVS Patient Safety Organization, which oversees VQI.
These pivotal improvements came about after the VQI distributed what it calls COPI reports – for Center Opportunity Profile for Improvement – to participating centers. Currently, 379 centers in 46 states and Ontario participate in VQI, feeding data into 12 different vascular procedure registries ranging from peripheral vascular interventions to lower-extremity amputations. As of Nov. 1, 2016, 330,400 procedures had been submitted to VQI.
Dr. Kraiss called the COPI report the “workhorse” of the VQI. “It can give participating centers insight into what they can do to improve outcomes,” he said. It is one of three types of reports VQI provides. The others are benchmarking reports that show the masked ratings for all participating centers but confidentially highlight the rating of the individual center receiving the report; and reports for individual providers.
The most recent readout of the SSI COPI report compared measures in two periods: 2011-2012 and 2013-2014. In those periods, overall use of chlorhexidine rose from 66.6% to 81.2%; transfusion rates of more than 2 units fell from 14.4% to 11.5%; the share of procedures lasting 220 minutes or more fell from 50.2% to 47.7%; and SSI rate overall fell from 3.4% to 3.1%. While the change in SSI was not statistically significant, Dr. Kraiss said the 17 centers that had a large increase in chlorhexidine use did see statistically significant declines in SSI.

VQI also showed a 5-year survival rate of 79% of patients discharged with both statin and aspirin therapy vs. 61% for patients discharged without (J Vasc Surg. 2015;61[4]:1010-9). “This represents an opportunity to inform individual providers about how often they discharge patients on an aspirin and statin,” Dr. Kraiss said. Provider-targeted reports show how individual physicians rate in their region and nationwide.
VQI is more than a registry, Dr. Kraiss said; it’s also organized into 17 regional quality groups that provide surgeons a safe place to discuss VQI data and how to use that to encourage best practices. “There’s no risk of compromising or making the information identifiable,” he said. “It’s a matter of just getting together and trying to share best practices in a relatively informal environment, and hopefully through that drive quality improvement.
Other benefits of participating in VQI are that it can help surgeons comply with requirements for Medicare’s Merit-based Incentive Payment System (MIPS). VQI also offers opportunities to enroll in industry-sponsored clinical trials, which can help defray the cost of VQI participation, he said.
Dr. Kraiss had no relevant financial relationships to disclose.
AT THE NORTHWESTERN VASCULAR SYMPOSIUM
Key clinical point: The Vascular Quality Initiative (VQI) provides comparative outcomes data that centers and surgeons can use to improve quality.
Major finding: Hospital length of stay for carotid endarterectomy averages 1.6 days for VQI centers vs. 2.6 days for nonparticipating centers.
Data source: VQI database.
Disclosures: Dr. Kraiss reported having no financial disclosures.
Trump nominates Neil Gorsuch as 9th Supreme Court justice
President Donald Trump has chosen Neil Gorsuch, a conservative judge who presides over Denver’s 10th Circuit as his nominee for U.S. Supreme Court justice. A federal judge for 10 years, Judge Gorsuch is a long-time comrade of deceased Supreme Court Justice Antonin Scalia with a strong record of supporting religious freedom and less government control.
At a White House East Room ceremony on Jan. 31, Judge Gorsuch said he was honored and humbled by President’s Trump’s nomination and that he looked forward to answering questions during his Senate nomination hearing.
President Trump hailed Judge Gorsuch’s credentials as the “best he has ever seen,” and called him more than qualified to take the reins as the next Supreme Court justice.
“He has an extraordinary resume, as good as it gets,” President Trump said. “The qualifications of Judge Gorsuch are beyond dispute. He is a man of our country and a man who our country needs – and needs badly – to uphold the rule of law and the rule of justice.”
Judge Gorsuch, 49,was appointed to the 10th U.S. Circuit Court of Appeals in Denver, by President George W. Bush. His nomination was confirmed unanimously in the Senate. He holds a doctoral degree from Oxford University, (England), a law degree from Harvard Law School, Cambridge, Mass., and an undergraduate degree from Columbia University, New York. Judge Gorsuch began his legal career as a law clerk to Supreme Court justices Byron R. White and Anthony M. Kennedy, as well as to Judge David B. Sentelle of the U.S. Court of Appeals for the D.C. Circuit. Prior to his judicial appointment, Judge Gorsuch served as principal deputy associate attorney general at the Justice Department under George W. Bush.

A Senate confirmation hearing had not been announced at press time. Judge Gorsuch has pledged to work with both parties during the hearing to answer questions and alleviate any concerns.
“I look forward to speaking with members from both sides of the aisle,” he said. “I consider the United States Senate the greatest deliberative body in the world, and I respect the important role the Constitution affords it in the confirmation of our judges.”
Justice Antonin Scalia died suddenly on Feb. 13, 2016, leaving the high court with only eight members. Then-President Barack Obama nominated Judge Merrick Garland of the U.S. Court of Appeals for the District of Columbia Circuit to fill Scalia’s seat. However, the Republican-controlled Senate blocked Judge Garland from ever having a hearing and his nomination was never fully considered.
[email protected]
On Twitter @legal_med
President Donald Trump has chosen Neil Gorsuch, a conservative judge who presides over Denver’s 10th Circuit as his nominee for U.S. Supreme Court justice. A federal judge for 10 years, Judge Gorsuch is a long-time comrade of deceased Supreme Court Justice Antonin Scalia with a strong record of supporting religious freedom and less government control.
At a White House East Room ceremony on Jan. 31, Judge Gorsuch said he was honored and humbled by President’s Trump’s nomination and that he looked forward to answering questions during his Senate nomination hearing.
President Trump hailed Judge Gorsuch’s credentials as the “best he has ever seen,” and called him more than qualified to take the reins as the next Supreme Court justice.
“He has an extraordinary resume, as good as it gets,” President Trump said. “The qualifications of Judge Gorsuch are beyond dispute. He is a man of our country and a man who our country needs – and needs badly – to uphold the rule of law and the rule of justice.”
Judge Gorsuch, 49,was appointed to the 10th U.S. Circuit Court of Appeals in Denver, by President George W. Bush. His nomination was confirmed unanimously in the Senate. He holds a doctoral degree from Oxford University, (England), a law degree from Harvard Law School, Cambridge, Mass., and an undergraduate degree from Columbia University, New York. Judge Gorsuch began his legal career as a law clerk to Supreme Court justices Byron R. White and Anthony M. Kennedy, as well as to Judge David B. Sentelle of the U.S. Court of Appeals for the D.C. Circuit. Prior to his judicial appointment, Judge Gorsuch served as principal deputy associate attorney general at the Justice Department under George W. Bush.

A Senate confirmation hearing had not been announced at press time. Judge Gorsuch has pledged to work with both parties during the hearing to answer questions and alleviate any concerns.
“I look forward to speaking with members from both sides of the aisle,” he said. “I consider the United States Senate the greatest deliberative body in the world, and I respect the important role the Constitution affords it in the confirmation of our judges.”
Justice Antonin Scalia died suddenly on Feb. 13, 2016, leaving the high court with only eight members. Then-President Barack Obama nominated Judge Merrick Garland of the U.S. Court of Appeals for the District of Columbia Circuit to fill Scalia’s seat. However, the Republican-controlled Senate blocked Judge Garland from ever having a hearing and his nomination was never fully considered.
[email protected]
On Twitter @legal_med
President Donald Trump has chosen Neil Gorsuch, a conservative judge who presides over Denver’s 10th Circuit as his nominee for U.S. Supreme Court justice. A federal judge for 10 years, Judge Gorsuch is a long-time comrade of deceased Supreme Court Justice Antonin Scalia with a strong record of supporting religious freedom and less government control.
At a White House East Room ceremony on Jan. 31, Judge Gorsuch said he was honored and humbled by President’s Trump’s nomination and that he looked forward to answering questions during his Senate nomination hearing.
President Trump hailed Judge Gorsuch’s credentials as the “best he has ever seen,” and called him more than qualified to take the reins as the next Supreme Court justice.
“He has an extraordinary resume, as good as it gets,” President Trump said. “The qualifications of Judge Gorsuch are beyond dispute. He is a man of our country and a man who our country needs – and needs badly – to uphold the rule of law and the rule of justice.”
Judge Gorsuch, 49,was appointed to the 10th U.S. Circuit Court of Appeals in Denver, by President George W. Bush. His nomination was confirmed unanimously in the Senate. He holds a doctoral degree from Oxford University, (England), a law degree from Harvard Law School, Cambridge, Mass., and an undergraduate degree from Columbia University, New York. Judge Gorsuch began his legal career as a law clerk to Supreme Court justices Byron R. White and Anthony M. Kennedy, as well as to Judge David B. Sentelle of the U.S. Court of Appeals for the D.C. Circuit. Prior to his judicial appointment, Judge Gorsuch served as principal deputy associate attorney general at the Justice Department under George W. Bush.

A Senate confirmation hearing had not been announced at press time. Judge Gorsuch has pledged to work with both parties during the hearing to answer questions and alleviate any concerns.
“I look forward to speaking with members from both sides of the aisle,” he said. “I consider the United States Senate the greatest deliberative body in the world, and I respect the important role the Constitution affords it in the confirmation of our judges.”
Justice Antonin Scalia died suddenly on Feb. 13, 2016, leaving the high court with only eight members. Then-President Barack Obama nominated Judge Merrick Garland of the U.S. Court of Appeals for the District of Columbia Circuit to fill Scalia’s seat. However, the Republican-controlled Senate blocked Judge Garland from ever having a hearing and his nomination was never fully considered.
[email protected]
On Twitter @legal_med
Bursectomy provides no benefit over omentectomy for gastric cancers
SAN FRANCISCO – Bursectomy was not found to be superior to omentectomy for improving survival in patients with subserosal/serosal gastric cancer in a Japanese randomized phase III study.
The procedure – the dissection of the peritoneal lining covering the pancreas and anterior plane of the transverse mesocolon to prevent peritoneal metastasis – was common worldwide and considered standard in Japan from the 1950s until the mid-1990s, but was replaced by omentectomy following publication of reports questioning its value, according to Masanori Terashima, MD, of Shizuoka Cancer Center, Nagaizumi, Japan.
However, interest in bursectomy was rekindled when a 2012 noninferiority phase II study suggested that bursectomy may improve survival; the authors concluded that it should not be abandoned as a futile procedure until more definitive data could be obtained (Gastric Cancer. 2012;15[1]:42-8).
“So based on this result, [Japanese Clinical Oncology Group] conducted a large-scale randomized phase III trial [JCOG1001] to evaluate the efficacy of bursectomy. The objective of this study was to confirm the superiority of bursectomy for clinical T3 or clinical T4a gastric cancer patients in terms of overall survival,” Dr. Terashima said at the symposium, sponsored by ASCO, ASTRO, the American Gastroenterological Association, and Society of Surgical Oncology.
At a planned interim analysis when 522 patients had been enrolled, the Data and Safety Monitoring Committee approved continued enrollment. However, at a second interim analysis when 54% of expected events had been observed, the committee recommended early release of the survival results because “there was little possibility of demonstration of the superiority of bursectomy,” he said.
Overall 3-year survival was 83.3% among 601 patients in the bursectomy arm, compared with 86% in 600 patients in the nonbursectomy arm (hazard ratio, 1.07). The predictive probability of superiority of bursectomy was 12.7%.
Study subjects were adults aged 20-80 years (median of 65-66 years) with histologically proven adenocarcinoma of the stomach and clinical T3 or T4 disease. They enrolled from 57 institutions and were intraoperatively randomized to the bursectomy or nonbursectomy arm after confirmation of tumor stage. All received adjuvant chemotherapy with S-1 for 1 year for pathologic stage II/III disease.
Patients with bulky nodal metastases, Borrmann type 4 and 8 cm or larger Borrmann type 3 tumors were excluded, as their poor prognosis made them candidates for other clinical trials, Dr. Terashima said.
Patients’ background and operative procedures were well balanced between the arms, he noted.
For those in the bursectomy group, operation time was longer (median, 254 vs. 222 minutes), and blood loss was larger (330 vs. 230 mL), although the incidence of blood transfusion was not significantly different between the groups (4.5% vs. 4.8%).
The incidence of grade 3 or higher complications was slightly higher in the bursectomy arm (13.3% vs. 11.6%). This was due largely to the incidence of pancreatic fistulas, which was nearly double in the bursectomy arm (4.8% vs. 2.5% ).
Mortality was “quite low” in both groups (0.2 vs. 0.8%).
“There was no significant difference in overall survival between the arms. Bursectomy seemed to be a bit inferior to the nonbursectomy arm. Relapse-free survival also demonstrated no significant difference between the arms. Again, bursectomy seemed to be a bit worse than nonbursectomy,” he said.
The most common site of recurrence for all patient was the peritoneum, with 63 and 56 patients in the bursectomy and nonbursectomy arms, respectively, experiencing peritoneal recurrence.
“We could not detect any subgroup who may have a benefit from bursectomy,” Dr. Terashima said.
“Bursectomy is not recommended as a standard treatment for clinical T3 or clinical T4 gastric cancer, while complete omentectomy remains a part of standard procedure,” he concluded.
Dr. Terashima reported receiving honoraria, and/or research funding from Chugai Pharma, Eisai; Lilly, Otsuka, Taiho Pharmaceutical, Takeda, and Yakult Honsha.
SAN FRANCISCO – Bursectomy was not found to be superior to omentectomy for improving survival in patients with subserosal/serosal gastric cancer in a Japanese randomized phase III study.
The procedure – the dissection of the peritoneal lining covering the pancreas and anterior plane of the transverse mesocolon to prevent peritoneal metastasis – was common worldwide and considered standard in Japan from the 1950s until the mid-1990s, but was replaced by omentectomy following publication of reports questioning its value, according to Masanori Terashima, MD, of Shizuoka Cancer Center, Nagaizumi, Japan.
However, interest in bursectomy was rekindled when a 2012 noninferiority phase II study suggested that bursectomy may improve survival; the authors concluded that it should not be abandoned as a futile procedure until more definitive data could be obtained (Gastric Cancer. 2012;15[1]:42-8).
“So based on this result, [Japanese Clinical Oncology Group] conducted a large-scale randomized phase III trial [JCOG1001] to evaluate the efficacy of bursectomy. The objective of this study was to confirm the superiority of bursectomy for clinical T3 or clinical T4a gastric cancer patients in terms of overall survival,” Dr. Terashima said at the symposium, sponsored by ASCO, ASTRO, the American Gastroenterological Association, and Society of Surgical Oncology.
At a planned interim analysis when 522 patients had been enrolled, the Data and Safety Monitoring Committee approved continued enrollment. However, at a second interim analysis when 54% of expected events had been observed, the committee recommended early release of the survival results because “there was little possibility of demonstration of the superiority of bursectomy,” he said.
Overall 3-year survival was 83.3% among 601 patients in the bursectomy arm, compared with 86% in 600 patients in the nonbursectomy arm (hazard ratio, 1.07). The predictive probability of superiority of bursectomy was 12.7%.
Study subjects were adults aged 20-80 years (median of 65-66 years) with histologically proven adenocarcinoma of the stomach and clinical T3 or T4 disease. They enrolled from 57 institutions and were intraoperatively randomized to the bursectomy or nonbursectomy arm after confirmation of tumor stage. All received adjuvant chemotherapy with S-1 for 1 year for pathologic stage II/III disease.
Patients with bulky nodal metastases, Borrmann type 4 and 8 cm or larger Borrmann type 3 tumors were excluded, as their poor prognosis made them candidates for other clinical trials, Dr. Terashima said.
Patients’ background and operative procedures were well balanced between the arms, he noted.
For those in the bursectomy group, operation time was longer (median, 254 vs. 222 minutes), and blood loss was larger (330 vs. 230 mL), although the incidence of blood transfusion was not significantly different between the groups (4.5% vs. 4.8%).
The incidence of grade 3 or higher complications was slightly higher in the bursectomy arm (13.3% vs. 11.6%). This was due largely to the incidence of pancreatic fistulas, which was nearly double in the bursectomy arm (4.8% vs. 2.5% ).
Mortality was “quite low” in both groups (0.2 vs. 0.8%).
“There was no significant difference in overall survival between the arms. Bursectomy seemed to be a bit inferior to the nonbursectomy arm. Relapse-free survival also demonstrated no significant difference between the arms. Again, bursectomy seemed to be a bit worse than nonbursectomy,” he said.
The most common site of recurrence for all patient was the peritoneum, with 63 and 56 patients in the bursectomy and nonbursectomy arms, respectively, experiencing peritoneal recurrence.
“We could not detect any subgroup who may have a benefit from bursectomy,” Dr. Terashima said.
“Bursectomy is not recommended as a standard treatment for clinical T3 or clinical T4 gastric cancer, while complete omentectomy remains a part of standard procedure,” he concluded.
Dr. Terashima reported receiving honoraria, and/or research funding from Chugai Pharma, Eisai; Lilly, Otsuka, Taiho Pharmaceutical, Takeda, and Yakult Honsha.
SAN FRANCISCO – Bursectomy was not found to be superior to omentectomy for improving survival in patients with subserosal/serosal gastric cancer in a Japanese randomized phase III study.
The procedure – the dissection of the peritoneal lining covering the pancreas and anterior plane of the transverse mesocolon to prevent peritoneal metastasis – was common worldwide and considered standard in Japan from the 1950s until the mid-1990s, but was replaced by omentectomy following publication of reports questioning its value, according to Masanori Terashima, MD, of Shizuoka Cancer Center, Nagaizumi, Japan.
However, interest in bursectomy was rekindled when a 2012 noninferiority phase II study suggested that bursectomy may improve survival; the authors concluded that it should not be abandoned as a futile procedure until more definitive data could be obtained (Gastric Cancer. 2012;15[1]:42-8).
“So based on this result, [Japanese Clinical Oncology Group] conducted a large-scale randomized phase III trial [JCOG1001] to evaluate the efficacy of bursectomy. The objective of this study was to confirm the superiority of bursectomy for clinical T3 or clinical T4a gastric cancer patients in terms of overall survival,” Dr. Terashima said at the symposium, sponsored by ASCO, ASTRO, the American Gastroenterological Association, and Society of Surgical Oncology.
At a planned interim analysis when 522 patients had been enrolled, the Data and Safety Monitoring Committee approved continued enrollment. However, at a second interim analysis when 54% of expected events had been observed, the committee recommended early release of the survival results because “there was little possibility of demonstration of the superiority of bursectomy,” he said.
Overall 3-year survival was 83.3% among 601 patients in the bursectomy arm, compared with 86% in 600 patients in the nonbursectomy arm (hazard ratio, 1.07). The predictive probability of superiority of bursectomy was 12.7%.
Study subjects were adults aged 20-80 years (median of 65-66 years) with histologically proven adenocarcinoma of the stomach and clinical T3 or T4 disease. They enrolled from 57 institutions and were intraoperatively randomized to the bursectomy or nonbursectomy arm after confirmation of tumor stage. All received adjuvant chemotherapy with S-1 for 1 year for pathologic stage II/III disease.
Patients with bulky nodal metastases, Borrmann type 4 and 8 cm or larger Borrmann type 3 tumors were excluded, as their poor prognosis made them candidates for other clinical trials, Dr. Terashima said.
Patients’ background and operative procedures were well balanced between the arms, he noted.
For those in the bursectomy group, operation time was longer (median, 254 vs. 222 minutes), and blood loss was larger (330 vs. 230 mL), although the incidence of blood transfusion was not significantly different between the groups (4.5% vs. 4.8%).
The incidence of grade 3 or higher complications was slightly higher in the bursectomy arm (13.3% vs. 11.6%). This was due largely to the incidence of pancreatic fistulas, which was nearly double in the bursectomy arm (4.8% vs. 2.5% ).
Mortality was “quite low” in both groups (0.2 vs. 0.8%).
“There was no significant difference in overall survival between the arms. Bursectomy seemed to be a bit inferior to the nonbursectomy arm. Relapse-free survival also demonstrated no significant difference between the arms. Again, bursectomy seemed to be a bit worse than nonbursectomy,” he said.
The most common site of recurrence for all patient was the peritoneum, with 63 and 56 patients in the bursectomy and nonbursectomy arms, respectively, experiencing peritoneal recurrence.
“We could not detect any subgroup who may have a benefit from bursectomy,” Dr. Terashima said.
“Bursectomy is not recommended as a standard treatment for clinical T3 or clinical T4 gastric cancer, while complete omentectomy remains a part of standard procedure,” he concluded.
Dr. Terashima reported receiving honoraria, and/or research funding from Chugai Pharma, Eisai; Lilly, Otsuka, Taiho Pharmaceutical, Takeda, and Yakult Honsha.
AT THE 2017 GASTROINTESTINAL CANCERS SYMPOSIUM
Key clinical point:
Major finding: Overall 3-year survival was 83.3% and 86% in the bursectomy and nonbursectomy arms, respectively (hazard ratio, 1.07).
Data source: The randomized phase III JCOG1001 trial with more than 1,200 subjects.
Disclosures: Dr. Terashima reported receiving honoraria, and/or research funding from Chugai Pharma, Eisai; Lilly, Otsuka, Taiho Pharmaceutical, Takeda, and Yakult Honsha.
February Hot Threads in ACS Communities
Your colleagues already have a lot to say in 2017. Here are the top discussion threads in ACS Communities just prior to press time (communities in which the threads appear are listed in parentheses):
1. Care for the Vulnerable vs Cash for the Powerful... (General Surgery)
2. Car lease or buy (General Surgery)
3. Interval cholecystectomy (General Surgery)
4. Enemy of good is better (General Surgery)
5. Crushed meds and elixirs (Bariatric Surgery)
6. Unsuspected cancer on reduction mammoplasty (Breast Surgery)
7. UC with sigmoid tumor (Colon and Rectal Surgery)
8. Transverse colostomy problems (Colon and Rectal Surgery)
9. Complex anal fistula (Colon and Rectal Surgery)
10. Ethical dilemma (Trauma Surgery)
To join communities, log in to ACS Communities at http://acscommunities.facs.org/home, go to “Browse All Communities” near the top of any page, and click the blue “Join” button next to the community you’d like to join. If you have any questions, please send them to [email protected].
Your colleagues already have a lot to say in 2017. Here are the top discussion threads in ACS Communities just prior to press time (communities in which the threads appear are listed in parentheses):
1. Care for the Vulnerable vs Cash for the Powerful... (General Surgery)
2. Car lease or buy (General Surgery)
3. Interval cholecystectomy (General Surgery)
4. Enemy of good is better (General Surgery)
5. Crushed meds and elixirs (Bariatric Surgery)
6. Unsuspected cancer on reduction mammoplasty (Breast Surgery)
7. UC with sigmoid tumor (Colon and Rectal Surgery)
8. Transverse colostomy problems (Colon and Rectal Surgery)
9. Complex anal fistula (Colon and Rectal Surgery)
10. Ethical dilemma (Trauma Surgery)
To join communities, log in to ACS Communities at http://acscommunities.facs.org/home, go to “Browse All Communities” near the top of any page, and click the blue “Join” button next to the community you’d like to join. If you have any questions, please send them to [email protected].
Your colleagues already have a lot to say in 2017. Here are the top discussion threads in ACS Communities just prior to press time (communities in which the threads appear are listed in parentheses):
1. Care for the Vulnerable vs Cash for the Powerful... (General Surgery)
2. Car lease or buy (General Surgery)
3. Interval cholecystectomy (General Surgery)
4. Enemy of good is better (General Surgery)
5. Crushed meds and elixirs (Bariatric Surgery)
6. Unsuspected cancer on reduction mammoplasty (Breast Surgery)
7. UC with sigmoid tumor (Colon and Rectal Surgery)
8. Transverse colostomy problems (Colon and Rectal Surgery)
9. Complex anal fistula (Colon and Rectal Surgery)
10. Ethical dilemma (Trauma Surgery)
To join communities, log in to ACS Communities at http://acscommunities.facs.org/home, go to “Browse All Communities” near the top of any page, and click the blue “Join” button next to the community you’d like to join. If you have any questions, please send them to [email protected].
STICHES boosts CABG role in severe LV dysfunction
SNOWMASS, COLO. – Coronary artery bypass graft surgery in patients with severe ischemic left ventricular dysfunction is overdue for an upgrade in status in the American College of Cardiology/American Heart Association guidelines on the strength of the landmark STICH trial and its extended follow-up stage known as STICHES, according to Vinod H. Thourani, MD.
Currently, the guidelines give CABG in this large and growing population a class IIb recommendation, meaning it “might be considered.” This undervalues the study’s core lesson: “STICHES showed a clear survival benefit with CABG, so this most likely should become a class IIa recommendation,” Dr. Thourani said at the Annual Cardiovascular Conference at Snowmass.
He went on to describe how he applies the key study findings to individual patients.
At 5 and 10 years of follow-up, the probability of all-cause mortality was reduced by 14% and 16%, respectively, in the CABG group. The surgery provided on average an 18-month extension of life. The price paid for the CABG benefit was a 3.6% mortality rate at 30 days; however, this was overcome by the 2-year mark, at which point survival in the CABG group surpassed that in controls. Thereafter, the all-cause mortality gap between the two groups continued to widen for the duration of follow-up.
For the composite endpoint of all-cause mortality or cardiovascular hospitalization, the CABG group enjoyed a 26% relative risk reduction, compared with optimal medical management alone at 5 years, and a 28% reduction in risk at 10 years. The two study groups diverged in terms of risk of cardiovascular hospitalization after only 3 months.
CABG provided a reduction in the risk of cardiovascular death that was consistent across all ages. In contrast, the reduction in all-cause mortality was not, since a higher proportion of deaths in older patients came from cancer and other noncardiovascular causes (Circulation. 2016 Nov 1;134[18]:1314-24).
There have been no randomized, controlled trials of percutaneous coronary intervention in patients with heart failure.
“An interesting finding in STICHES was that medical therapy had a much higher 10-year all-cause mortality the younger the patient was. So CABG particularly benefits those who are at a younger age – in this study, age 60 or less. As you get older, say, at 80 years of age, I’m not sure there’s a huge benefit in all-cause mortality at that point,” said Dr. Thourani, professor of surgery and medicine, and codirector of the structural heart and valve center at Emory University in Atlanta.
In a STICH substudy, roughly half of participants underwent presurgical myocardial viability testing via single-photon emission CT and/or dobutamine echocardiography. The investigators found that the results didn’t predict mortality benefit for CABG (N Engl J Med. 2011 Apr 28;364[17]:1617-25).
More recently, however, other investigators have reported MRI to have prognostic value. For example, Belgian investigators showed that medical therapy in patients with ischemic heart failure and dysfunctional but viable myocardium on delayed-enhanced MRI was associated with a 4.56-fold increased likelihood of mortality during 3 years of follow-up, compared with complete revascularization via CABG (J Am Coll Cardiol. 2012 Feb 28;59[9]:825-35).
“This observation has been useful for me,” Dr. Thourani said. “My own personal practice is if I have good targets, I don’t do viability testing, but if I have really bad targets where I know I’m going to have a tough time sewing grafts, I try to get an MRI for viability testing.”
One important lesson of STICH is that all patients with heart failure and a low left ventricular ejection fraction should have a coronary angiogram, even if they are free of ischemia on noninvasive testing and have no angina. That’s because the patients enrolled in STICH had little or no angina, the surgeon continued.
These STICH-type patients will benefit greatly from a heart team assessment factoring in an individual’s Society of Thoracic Surgeons’ predicted risk score, based on age, comorbidities, and other factors. For example, if a patient’s STS risk score with CABG is 0.7%, that’s a strong argument for opting for the surgery, since the 30-day operative mortality in STICH was 3.6%. If, on the other hand, the STS score is greater than 7%, that’s a tougher call.
“I think it’s really important that a heart team assessment includes a noninvasive cardiologist as well as an interventional cardiologist and cardiac surgeon because I think interventionalists and cardiac surgeons sometimes get a little goofy in their assessment of these patients,” Dr. Thourani said.
Patients with a low ejection fraction and coronary artery disease who are deemed poor candidates for CABG should be evaluated for a mechanical circulatory support device or a heart transplant.
“I think that’s something we don’t think about enough, quite honestly,” he said.
Dr. Thourani reported serving as a consultant to Abbott Vascular, Edwards Lifesciences, and Gore, and receiving research grants from numerous companies.
SNOWMASS, COLO. – Coronary artery bypass graft surgery in patients with severe ischemic left ventricular dysfunction is overdue for an upgrade in status in the American College of Cardiology/American Heart Association guidelines on the strength of the landmark STICH trial and its extended follow-up stage known as STICHES, according to Vinod H. Thourani, MD.
Currently, the guidelines give CABG in this large and growing population a class IIb recommendation, meaning it “might be considered.” This undervalues the study’s core lesson: “STICHES showed a clear survival benefit with CABG, so this most likely should become a class IIa recommendation,” Dr. Thourani said at the Annual Cardiovascular Conference at Snowmass.
He went on to describe how he applies the key study findings to individual patients.
At 5 and 10 years of follow-up, the probability of all-cause mortality was reduced by 14% and 16%, respectively, in the CABG group. The surgery provided on average an 18-month extension of life. The price paid for the CABG benefit was a 3.6% mortality rate at 30 days; however, this was overcome by the 2-year mark, at which point survival in the CABG group surpassed that in controls. Thereafter, the all-cause mortality gap between the two groups continued to widen for the duration of follow-up.
For the composite endpoint of all-cause mortality or cardiovascular hospitalization, the CABG group enjoyed a 26% relative risk reduction, compared with optimal medical management alone at 5 years, and a 28% reduction in risk at 10 years. The two study groups diverged in terms of risk of cardiovascular hospitalization after only 3 months.
CABG provided a reduction in the risk of cardiovascular death that was consistent across all ages. In contrast, the reduction in all-cause mortality was not, since a higher proportion of deaths in older patients came from cancer and other noncardiovascular causes (Circulation. 2016 Nov 1;134[18]:1314-24).
There have been no randomized, controlled trials of percutaneous coronary intervention in patients with heart failure.
“An interesting finding in STICHES was that medical therapy had a much higher 10-year all-cause mortality the younger the patient was. So CABG particularly benefits those who are at a younger age – in this study, age 60 or less. As you get older, say, at 80 years of age, I’m not sure there’s a huge benefit in all-cause mortality at that point,” said Dr. Thourani, professor of surgery and medicine, and codirector of the structural heart and valve center at Emory University in Atlanta.
In a STICH substudy, roughly half of participants underwent presurgical myocardial viability testing via single-photon emission CT and/or dobutamine echocardiography. The investigators found that the results didn’t predict mortality benefit for CABG (N Engl J Med. 2011 Apr 28;364[17]:1617-25).
More recently, however, other investigators have reported MRI to have prognostic value. For example, Belgian investigators showed that medical therapy in patients with ischemic heart failure and dysfunctional but viable myocardium on delayed-enhanced MRI was associated with a 4.56-fold increased likelihood of mortality during 3 years of follow-up, compared with complete revascularization via CABG (J Am Coll Cardiol. 2012 Feb 28;59[9]:825-35).
“This observation has been useful for me,” Dr. Thourani said. “My own personal practice is if I have good targets, I don’t do viability testing, but if I have really bad targets where I know I’m going to have a tough time sewing grafts, I try to get an MRI for viability testing.”
One important lesson of STICH is that all patients with heart failure and a low left ventricular ejection fraction should have a coronary angiogram, even if they are free of ischemia on noninvasive testing and have no angina. That’s because the patients enrolled in STICH had little or no angina, the surgeon continued.
These STICH-type patients will benefit greatly from a heart team assessment factoring in an individual’s Society of Thoracic Surgeons’ predicted risk score, based on age, comorbidities, and other factors. For example, if a patient’s STS risk score with CABG is 0.7%, that’s a strong argument for opting for the surgery, since the 30-day operative mortality in STICH was 3.6%. If, on the other hand, the STS score is greater than 7%, that’s a tougher call.
“I think it’s really important that a heart team assessment includes a noninvasive cardiologist as well as an interventional cardiologist and cardiac surgeon because I think interventionalists and cardiac surgeons sometimes get a little goofy in their assessment of these patients,” Dr. Thourani said.
Patients with a low ejection fraction and coronary artery disease who are deemed poor candidates for CABG should be evaluated for a mechanical circulatory support device or a heart transplant.
“I think that’s something we don’t think about enough, quite honestly,” he said.
Dr. Thourani reported serving as a consultant to Abbott Vascular, Edwards Lifesciences, and Gore, and receiving research grants from numerous companies.
SNOWMASS, COLO. – Coronary artery bypass graft surgery in patients with severe ischemic left ventricular dysfunction is overdue for an upgrade in status in the American College of Cardiology/American Heart Association guidelines on the strength of the landmark STICH trial and its extended follow-up stage known as STICHES, according to Vinod H. Thourani, MD.
Currently, the guidelines give CABG in this large and growing population a class IIb recommendation, meaning it “might be considered.” This undervalues the study’s core lesson: “STICHES showed a clear survival benefit with CABG, so this most likely should become a class IIa recommendation,” Dr. Thourani said at the Annual Cardiovascular Conference at Snowmass.
He went on to describe how he applies the key study findings to individual patients.
At 5 and 10 years of follow-up, the probability of all-cause mortality was reduced by 14% and 16%, respectively, in the CABG group. The surgery provided on average an 18-month extension of life. The price paid for the CABG benefit was a 3.6% mortality rate at 30 days; however, this was overcome by the 2-year mark, at which point survival in the CABG group surpassed that in controls. Thereafter, the all-cause mortality gap between the two groups continued to widen for the duration of follow-up.
For the composite endpoint of all-cause mortality or cardiovascular hospitalization, the CABG group enjoyed a 26% relative risk reduction, compared with optimal medical management alone at 5 years, and a 28% reduction in risk at 10 years. The two study groups diverged in terms of risk of cardiovascular hospitalization after only 3 months.
CABG provided a reduction in the risk of cardiovascular death that was consistent across all ages. In contrast, the reduction in all-cause mortality was not, since a higher proportion of deaths in older patients came from cancer and other noncardiovascular causes (Circulation. 2016 Nov 1;134[18]:1314-24).
There have been no randomized, controlled trials of percutaneous coronary intervention in patients with heart failure.
“An interesting finding in STICHES was that medical therapy had a much higher 10-year all-cause mortality the younger the patient was. So CABG particularly benefits those who are at a younger age – in this study, age 60 or less. As you get older, say, at 80 years of age, I’m not sure there’s a huge benefit in all-cause mortality at that point,” said Dr. Thourani, professor of surgery and medicine, and codirector of the structural heart and valve center at Emory University in Atlanta.
In a STICH substudy, roughly half of participants underwent presurgical myocardial viability testing via single-photon emission CT and/or dobutamine echocardiography. The investigators found that the results didn’t predict mortality benefit for CABG (N Engl J Med. 2011 Apr 28;364[17]:1617-25).
More recently, however, other investigators have reported MRI to have prognostic value. For example, Belgian investigators showed that medical therapy in patients with ischemic heart failure and dysfunctional but viable myocardium on delayed-enhanced MRI was associated with a 4.56-fold increased likelihood of mortality during 3 years of follow-up, compared with complete revascularization via CABG (J Am Coll Cardiol. 2012 Feb 28;59[9]:825-35).
“This observation has been useful for me,” Dr. Thourani said. “My own personal practice is if I have good targets, I don’t do viability testing, but if I have really bad targets where I know I’m going to have a tough time sewing grafts, I try to get an MRI for viability testing.”
One important lesson of STICH is that all patients with heart failure and a low left ventricular ejection fraction should have a coronary angiogram, even if they are free of ischemia on noninvasive testing and have no angina. That’s because the patients enrolled in STICH had little or no angina, the surgeon continued.
These STICH-type patients will benefit greatly from a heart team assessment factoring in an individual’s Society of Thoracic Surgeons’ predicted risk score, based on age, comorbidities, and other factors. For example, if a patient’s STS risk score with CABG is 0.7%, that’s a strong argument for opting for the surgery, since the 30-day operative mortality in STICH was 3.6%. If, on the other hand, the STS score is greater than 7%, that’s a tougher call.
“I think it’s really important that a heart team assessment includes a noninvasive cardiologist as well as an interventional cardiologist and cardiac surgeon because I think interventionalists and cardiac surgeons sometimes get a little goofy in their assessment of these patients,” Dr. Thourani said.
Patients with a low ejection fraction and coronary artery disease who are deemed poor candidates for CABG should be evaluated for a mechanical circulatory support device or a heart transplant.
“I think that’s something we don’t think about enough, quite honestly,” he said.
Dr. Thourani reported serving as a consultant to Abbott Vascular, Edwards Lifesciences, and Gore, and receiving research grants from numerous companies.
EXPERT ANALYSIS AT THE CARDIOVASCULAR CONFERENCE AT SNOWMASS