User login
Why pregnant people were left behind while vaccines moved at ‘warp speed’ to help the masses
Kia Slade was 7 months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of COVID-19 pneumonia had left Ms. Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Ms. Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Ms. Slade wouldn’t meet him until July. She was on a ventilator in a medically-induced coma for 8 weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Ms. Slade was vaccinated against COVID-19.
“I wish I had gotten the vaccine earlier,” said Ms. Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Ms. Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, 2022, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with COVID-19 and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people – who have special health needs and risks – were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of COVID-19 vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people – and the months-long delay in recommending that they be immunized – helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of COVID infection,” said Jane Van Dis, MD, an ob.gyn. at the University of Rochester (N.Y.) Medical Center who has treated many patients like Ms. Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New technology, uncertain risks
At the time COVID-19 vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Jacqueline Miller, MD, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Dr. Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Dr. Offit noted that vaccines have a strong record of safety in pregnancy and he sees no reason to have excluded pregnant people. None of the vaccines currently in use – including the chickenpox and rubella vaccines, which contain live viruses – have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Dr. Offit, the coinventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drug makers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Dr. Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Dr. Offit, a member of a committee that advises the Food and Drug Administration on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, 2021, after the first real-world vaccine studies had been published. The CDC followed suit in August of 2021.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Kelli Burroughs, MD, the department chair of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the COVID-19 vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, because of increased risks of hospitalization, ICU admission, and mechanical ventilation, Dr. Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe COVID-19 infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Ms. Slade were already at higher risk of maternal and infant mortality before the pandemic, because of higher underlying risks, unequal access to health care, and other factors. COVID-19 has magnified those risks, said Dr. Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Ms. Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with COVID-19 shots.
“If there had been data out there saying the COVID shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Ms. Slade, who has had type 2 diabetes for 12 years.
Working at warp speed
Government scientists at the NIH were concerned about the risk of COVID-19 to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Larry Corey, MD, a leader of the COVID-19 Prevention Network, which coordinated COVID-19 vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Dr. Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the COVID-19 vaccines, including pregnant people might have meant having to hit pause on those trials, Dr. Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Dr. Corey said. Making COVID-19 vaccines available within a year “saved hundreds of thousands of lives.”
The first data on COVID-19 vaccine safety in pregnancy was published in April of 2021 when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that COVID-19 vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Brenna Hughes, MD, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ COVID-19 expert group, agrees that adding pregnant people to large-scale COVID-19 vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with COVID is hard,” Dr. Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies – called developmental and reproductive toxicity studies – before testing vaccines in pregnant people. Although these studies are essential, they take 5-6 months, and weren’t completed until late 2020, around the time the first COVID-19 vaccines were authorized for adults, said Emily Erbelding, MD, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of the NIH.
Pregnancy studies “were an afterthought,” said Irina Burd, MD, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Dr. Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Dr. Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Dr. Hughes said. “But we should be protecting patients through research, not from research.”
Recovering physically and emotionally
Ms. Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Ms. Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, because of a breathing tube in her throat.
Today, Ms. Slade is a strong advocate of COVID-19 vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Ms. Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea, and even vomiting when she eats or drinks. Ms. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency department when the pain is unbearable. “Most days, I’m just miserable,” she said.
Her family suffered as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Ms. Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Ms. Slade said she knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Ms. Slade said, “and she said, ‘You know, I almost had to bury you.’ ”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Kia Slade was 7 months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of COVID-19 pneumonia had left Ms. Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Ms. Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Ms. Slade wouldn’t meet him until July. She was on a ventilator in a medically-induced coma for 8 weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Ms. Slade was vaccinated against COVID-19.
“I wish I had gotten the vaccine earlier,” said Ms. Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Ms. Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, 2022, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with COVID-19 and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people – who have special health needs and risks – were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of COVID-19 vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people – and the months-long delay in recommending that they be immunized – helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of COVID infection,” said Jane Van Dis, MD, an ob.gyn. at the University of Rochester (N.Y.) Medical Center who has treated many patients like Ms. Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New technology, uncertain risks
At the time COVID-19 vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Jacqueline Miller, MD, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Dr. Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Dr. Offit noted that vaccines have a strong record of safety in pregnancy and he sees no reason to have excluded pregnant people. None of the vaccines currently in use – including the chickenpox and rubella vaccines, which contain live viruses – have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Dr. Offit, the coinventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drug makers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Dr. Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Dr. Offit, a member of a committee that advises the Food and Drug Administration on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, 2021, after the first real-world vaccine studies had been published. The CDC followed suit in August of 2021.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Kelli Burroughs, MD, the department chair of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the COVID-19 vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, because of increased risks of hospitalization, ICU admission, and mechanical ventilation, Dr. Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe COVID-19 infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Ms. Slade were already at higher risk of maternal and infant mortality before the pandemic, because of higher underlying risks, unequal access to health care, and other factors. COVID-19 has magnified those risks, said Dr. Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Ms. Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with COVID-19 shots.
“If there had been data out there saying the COVID shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Ms. Slade, who has had type 2 diabetes for 12 years.
Working at warp speed
Government scientists at the NIH were concerned about the risk of COVID-19 to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Larry Corey, MD, a leader of the COVID-19 Prevention Network, which coordinated COVID-19 vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Dr. Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the COVID-19 vaccines, including pregnant people might have meant having to hit pause on those trials, Dr. Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Dr. Corey said. Making COVID-19 vaccines available within a year “saved hundreds of thousands of lives.”
The first data on COVID-19 vaccine safety in pregnancy was published in April of 2021 when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that COVID-19 vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Brenna Hughes, MD, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ COVID-19 expert group, agrees that adding pregnant people to large-scale COVID-19 vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with COVID is hard,” Dr. Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies – called developmental and reproductive toxicity studies – before testing vaccines in pregnant people. Although these studies are essential, they take 5-6 months, and weren’t completed until late 2020, around the time the first COVID-19 vaccines were authorized for adults, said Emily Erbelding, MD, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of the NIH.
Pregnancy studies “were an afterthought,” said Irina Burd, MD, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Dr. Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Dr. Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Dr. Hughes said. “But we should be protecting patients through research, not from research.”
Recovering physically and emotionally
Ms. Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Ms. Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, because of a breathing tube in her throat.
Today, Ms. Slade is a strong advocate of COVID-19 vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Ms. Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea, and even vomiting when she eats or drinks. Ms. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency department when the pain is unbearable. “Most days, I’m just miserable,” she said.
Her family suffered as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Ms. Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Ms. Slade said she knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Ms. Slade said, “and she said, ‘You know, I almost had to bury you.’ ”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Kia Slade was 7 months pregnant, unvaccinated, and fighting for breath, her oxygen levels plummeting, when her son came into the world last May.
A severe case of COVID-19 pneumonia had left Ms. Slade delirious. When the intensive care team tried to place an oxygen mask on her face, she snatched it away, she recalled. Her baby’s heart rate began to drop.
Ms. Slade’s doctor performed an emergency cesarean section at her bedside in the intensive care unit, delivering baby Tristan 10 weeks early. He weighed just 2 pounds, 14 ounces, about half the size of small full-term baby.
But Ms. Slade wouldn’t meet him until July. She was on a ventilator in a medically-induced coma for 8 weeks, and she developed a serious infection and blood clot while unconscious. It was only after a perilous 2½ months in the hospital, during which her heart stopped twice, that Ms. Slade was vaccinated against COVID-19.
“I wish I had gotten the vaccine earlier,” said Ms. Slade, 42, who remains too sick to return to work as a special education teacher in Baltimore. Doctors “kept pushing me to get vaccinated, but there just wasn’t enough information out there for me to do it.”
A year ago, there was little to no vaccine safety data for pregnant people like Ms. Slade, because they had been excluded from clinical trials run by Pfizer, Moderna, and other vaccine makers.
Lacking data, health experts were unsure and divided about how to advise expectant parents. Although U.S. health officials permitted pregnant people to be vaccinated, the World Health Organization in January 2021 actually discouraged them from doing so; it later reversed that recommendation.
The uncertainty led many women to delay vaccination, and only about two-thirds of the pregnant people who have been tracked by the Centers for Disease Control and Prevention were fully vaccinated as of Feb. 5, 2022, leaving many expectant moms at a high risk of infection and life-threatening complications.
More than 29,000 pregnant people have been hospitalized with COVID-19 and 274 have died, according to the CDC.
“There were surely women who were hospitalized because there wasn’t information available to them,” said Paul A. Offit, MD, director of the Vaccine Education Center at the Children’s Hospital of Philadelphia.
Vaccine developers say that pregnant people – who have special health needs and risks – were excluded from clinical trials to protect them from potential side effects of novel technologies, including the Pfizer and Moderna mRNA vaccines and formulations made with cold viruses, such as the Johnson & Johnson vaccine.
But a KHN analysis also shows that pregnant people were left behind because including them in vaccine studies would have complicated and potentially delayed the delivery of COVID-19 vaccines to the broader population.
A growing number of women’s health researchers and advocates say that excluding pregnant people – and the months-long delay in recommending that they be immunized – helped fuel widespread vaccine hesitancy in this vulnerable group.
“Women and their unborn fetuses are dying of COVID infection,” said Jane Van Dis, MD, an ob.gyn. at the University of Rochester (N.Y.) Medical Center who has treated many patients like Ms. Slade. “Our failure as a society to vaccinate women in pregnancy will be remembered by the children and families who lost their mothers to this disease.”
New technology, uncertain risks
At the time COVID-19 vaccines were being developed, scientists had very little experience using mRNA vaccines in pregnant women, said Jacqueline Miller, MD, a senior vice president involved in vaccine research at Moderna.
“When you study anything in pregnant women, you have two patients, the mom and the unborn child,” Dr. Miller said. “Until we had more safety data on the platform, it wasn’t something we wanted to undertake.”
But Dr. Offit noted that vaccines have a strong record of safety in pregnancy and he sees no reason to have excluded pregnant people. None of the vaccines currently in use – including the chickenpox and rubella vaccines, which contain live viruses – have been shown to harm fetuses, he said. Doctors routinely recommend that pregnant people receive pertussis and flu vaccinations.
Dr. Offit, the coinventor of a rotavirus vaccine, said that some concerns about vaccines stem from commercial, not medical, interests. Drug makers don’t want to risk that their product will be blamed for any problems occurring in pregnant people, even if coincidental, he said.
“These companies don’t want bad news,” Dr. Offit said.
In the United States, health officials typically would have told expectant mothers not to take a vaccine that was untested during pregnancy, said Dr. Offit, a member of a committee that advises the Food and Drug Administration on vaccines.
Due to the urgency of the pandemic, health agencies instead permitted pregnant people to make up their own minds about vaccines without recommending them.
Women’s medical associations were also hampered by the lack of data. Neither the American College of Obstetricians and Gynecologists nor the Society for Maternal-Fetal Medicine actively encouraged pregnant people to be vaccinated until July 30, 2021, after the first real-world vaccine studies had been published. The CDC followed suit in August of 2021.
“If we had had this data in the beginning, we would have been able to vaccinate more women,” said Kelli Burroughs, MD, the department chair of obstetrics and gynecology at Memorial Hermann Sugar Land Hospital near Houston.
Yet anti-vaccine groups wasted no time in scaring pregnant people, flooding social media with misinformation about impaired fertility and harm to the fetus.
In the first few months after the COVID-19 vaccines were approved, some doctors were ambivalent about recommending them, and some still advise pregnant patients against vaccination.
An estimated 67% of pregnant people today are fully vaccinated, compared with about 89% of people 65 and older, another high-risk group, and 65% of Americans overall. Vaccination rates are lower among minorities, with 65% of expectant Hispanic mothers and 53% of pregnant African Americans fully vaccinated, according to the CDC.
Vaccination is especially important during pregnancy, because of increased risks of hospitalization, ICU admission, and mechanical ventilation, Dr. Burroughs said. A study released in February from the National Institutes of Health found that pregnant people with a moderate to severe COVID-19 infection also were more likely to have a C-section, deliver preterm, or develop a postpartum hemorrhage.
Black moms such as Ms. Slade were already at higher risk of maternal and infant mortality before the pandemic, because of higher underlying risks, unequal access to health care, and other factors. COVID-19 has magnified those risks, said Dr. Burroughs, who has persuaded reluctant patients by revealing that she had a healthy pregnancy and child after being vaccinated.
Ms. Slade said she has never opposed vaccines and had no hesitation about receiving other vaccines while pregnant. But she said she “just wasn’t comfortable” with COVID-19 shots.
“If there had been data out there saying the COVID shot was safe, and that nothing would happen to my baby and there was no risk of birth defects, I would have taken it,” said Ms. Slade, who has had type 2 diabetes for 12 years.
Working at warp speed
Government scientists at the NIH were concerned about the risk of COVID-19 to pregnant people from the very beginning and knew that expectant moms needed vaccines as much or more than anyone else, said Larry Corey, MD, a leader of the COVID-19 Prevention Network, which coordinated COVID-19 vaccine trials for the federal government.
But including pregnant volunteers in the larger vaccine trials could have led to interruptions and delays, Dr. Corey said. Researchers would have had to enroll thousands of pregnant volunteers to achieve statistically robust results that weren’t due to chance, he said.
Pregnancy can bring on a wide range of complications: gestational diabetes, hypertension, anemia, bleeding, blood clots, or problems with the placenta, for example. Up to 20% of people who know they’re pregnant miscarry. Because researchers would have been obliged to investigate any medical problem to make sure it wasn’t caused by one of the COVID-19 vaccines, including pregnant people might have meant having to hit pause on those trials, Dr. Corey said.
With death tolls from the pandemic mounting, “we had a mission to do this as quickly and as thoroughly as possible,” Dr. Corey said. Making COVID-19 vaccines available within a year “saved hundreds of thousands of lives.”
The first data on COVID-19 vaccine safety in pregnancy was published in April of 2021 when the CDC released an analysis of nearly 36,000 vaccinated pregnant people who had enrolled in a registry called V-safe, which allows users to log the dates of their vaccinations and any subsequent symptoms.
Later research showed that COVID-19 vaccines weren’t associated with increased risk of miscarriage or premature delivery.
Brenna Hughes, MD, a maternal-fetal medicine specialist and member of the American College of Obstetricians and Gynecologists’ COVID-19 expert group, agrees that adding pregnant people to large-scale COVID-19 vaccine and drug trials may have been impractical. But researchers could have launched parallel trials of pregnant women, once early studies showed the vaccines were safe in humans, she said.
“Would it have been hard? Everything with COVID is hard,” Dr. Hughes said. “But it would have been feasible.”
The FDA requires that researchers perform additional animal studies – called developmental and reproductive toxicity studies – before testing vaccines in pregnant people. Although these studies are essential, they take 5-6 months, and weren’t completed until late 2020, around the time the first COVID-19 vaccines were authorized for adults, said Emily Erbelding, MD, director of microbiology and infectious diseases at the National Institute of Allergy and Infectious Diseases, part of the NIH.
Pregnancy studies “were an afterthought,” said Irina Burd, MD, director of Johns Hopkins’ Integrated Research Center for Fetal Medicine and a professor of gynecology and obstetrics. “They should have been done sooner.”
The NIH is conducting a study of pregnant and postpartum people who decided on their own to be vaccinated, Dr. Erbelding said. The study is due to be completed by July 2023.
Janssen and Moderna are also conducting studies in pregnant people, both due to be completed in 2024.
Pfizer scientists encountered problems when they initiated a clinical trial, which would have randomly assigned pregnant people to receive either a vaccine or placebo. Once vaccines were widely available, many patients weren’t willing to take a chance on being unvaccinated until after delivery.
Pfizer has stopped recruiting patients and has not said whether it will publicly report any data from the trial.
Dr. Hughes said vaccine developers need to include pregnant people from the very beginning.
“There is this notion of protecting pregnant people from research,” Dr. Hughes said. “But we should be protecting patients through research, not from research.”
Recovering physically and emotionally
Ms. Slade still regrets being deprived of time with her children while she fought the disease.
Being on a ventilator kept her from spending those early weeks with her newborn, or from seeing her 9-year-old daughter, Zoe.
Even when Ms. Slade was finally able to see her son, she wasn’t able to tell him she loved him or sing a lullaby, or even talk at all, because of a breathing tube in her throat.
Today, Ms. Slade is a strong advocate of COVID-19 vaccinations, urging her friends and family to get their shots to avoid suffering the way she has.
Ms. Slade had to relearn to walk after being bedridden for weeks. Her many weeks on a ventilator may have contributed to her stomach paralysis, which often causes intense pain, nausea, and even vomiting when she eats or drinks. Ms. Slade weighs 50 pounds less today than before she became pregnant and has resorted to going to the emergency department when the pain is unbearable. “Most days, I’m just miserable,” she said.
Her family suffered as well. Like many babies born prematurely, Tristan, now nearly 9 months old and crawling, receives physical therapy to strengthen his muscles. At 15 pounds, Tristan is largely healthy, although his doctor said he has symptoms of asthma.
Ms. Slade said she would like to attend family counseling with Zoe, who rarely complains and tends to keep her feelings to herself. Ms. Slade said she knows her illness must have been terrifying for her little girl.
“The other day she was talking to me,” Ms. Slade said, “and she said, ‘You know, I almost had to bury you.’ ”
KHN (Kaiser Health News) is a national newsroom that produces in-depth journalism about health issues. Together with Policy Analysis and Polling, KHN is one of the three major operating programs at KFF (Kaiser Family Foundation). KFF is an endowed nonprofit organization providing information on health issues to the nation.
Epidural may lower odds of severe maternal birth complications
Use of neuraxial analgesia for vaginal delivery is associated with a 14% decreased risk for severe maternal morbidity, in part from a reduction in postpartum hemorrhage, new research shows.
The findings indicate that increasing the use of epidural or combined spinal-epidural analgesia may improve maternal health outcomes, especially for Hispanic, Black, and uninsured women who are less likely than White women to receive these interventions, according to the researchers, who published their findings online in JAMA Network Open.
About 80% of non-Hispanic White women receive neuraxial analgesia during labor in the United States, compared with 70% of non-Hispanic Black women and 65% of Hispanic women, according to birth certificate data. Among women without health insurance, half receive epidurals.
Programs that inform pregnant women about epidural use, expand Medicaid, and provide in-house obstetric anesthesia teams “may improve patient participation in clinical decision making and access to care,” study author Guohua Li, MD, DrPH, of Columbia University, New York, said in a statement about the research.
Earlier data from France showed that neuraxial analgesia is associated with reduced risk for severe postpartum hemorrhage. To examine the association between labor neuraxial analgesia and severe maternal morbidity in the United States, Dr. Li and colleagues analyzed more than 575,000 vaginal deliveries in New York hospitals between 2010 and 2017; about half (47.4%) of the women received epidurals during labor.
The researchers focused on severe maternal morbidity, including 16 complications, such as heart failure and sepsis, and five procedures, including hysterectomy and ventilation.
They also considered patient characteristics and comorbidities and hospital-related factors to identify patients who were at higher risk for injury or death.
Severe maternal morbidity occurred in 1.3% of the women. Of the 7,712 women with severe morbidity, more than one in three (35.6%) experienced postpartum hemorrhage.
The overall incidence of severe maternal morbidity was 1.3% among women who received an epidural injection and 1.4% among those who did not. In a weighted analysis, the adjusted odds ratio of severe maternal morbidity associated with epidurals was 0.86 (95% confidence interval, 0.82-0.90).
The study is limited by its observational design and does not prove causation, the authors acknowledged.
“Labor neuraxial analgesia may facilitate early evaluation and management of the third stage of labor to avoid escalation of postpartum hemorrhaging into grave complications and death,” study author Jean Guglielminotti, MD, PhD, an anesthesiologist at Columbia University, said in a statement.
Concerning trends
The Department of Health & Human Services has labeled severe maternal morbidity a public health priority. Recent data from the Centers for Disease Control and Prevention show an increase in maternal mortality rates and worsening disparities by race and ethnicity.
According to the CDC, 861 women died of maternal causes in 2020, up from 754 in 2019. The rate of maternal mortality increased from 20.1 to 23.8 deaths per 100,000 live births.
For Black women, however, the maternal mortality rate was far higher: 55.3 deaths per 100,000 live births – nearly triple the figure of 19.1 per 100,000 for White women. Between 2019 and 2020, the mortality rate increased significantly for Black and Hispanic women, but not White mothers.
Researchers affiliated with University of Toronto and the Hospital for Sick Children agreed in an accompanying editorial that more access to neuraxial labor analgesia for vaginal delivery might improve maternal health outcomes and “may be a strategy well worth pursuing in public health policy.”
The intervention is relatively safe and can “alleviate discomfort and distress,” they wrote.
Neuraxial anesthesia in surgical procedures has been shown to decrease the risk for complications like deep vein thrombosis, pulmonary embolus, transfusion requirements, and kidney failure, said editorialists Evelina Pankiv, MD; Alan Yang, MSc; and Kazuyoshi Aoyama, MD, PhD.
Benefits potentially could stem from improving blood flow, mitigating hypercoagulation, or reducing surgical stress response. But there are rare risks to consider as well, including hemorrhage, infection, and neurologic injury, they added.
Guglielminotti disclosed grants from the National Institute on Minority Health and Health Disparities. Dr. Aoyama reported receiving grants from the Perioperative Services Facilitator Grant Program and Hospital for Sick Children. The other authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of neuraxial analgesia for vaginal delivery is associated with a 14% decreased risk for severe maternal morbidity, in part from a reduction in postpartum hemorrhage, new research shows.
The findings indicate that increasing the use of epidural or combined spinal-epidural analgesia may improve maternal health outcomes, especially for Hispanic, Black, and uninsured women who are less likely than White women to receive these interventions, according to the researchers, who published their findings online in JAMA Network Open.
About 80% of non-Hispanic White women receive neuraxial analgesia during labor in the United States, compared with 70% of non-Hispanic Black women and 65% of Hispanic women, according to birth certificate data. Among women without health insurance, half receive epidurals.
Programs that inform pregnant women about epidural use, expand Medicaid, and provide in-house obstetric anesthesia teams “may improve patient participation in clinical decision making and access to care,” study author Guohua Li, MD, DrPH, of Columbia University, New York, said in a statement about the research.
Earlier data from France showed that neuraxial analgesia is associated with reduced risk for severe postpartum hemorrhage. To examine the association between labor neuraxial analgesia and severe maternal morbidity in the United States, Dr. Li and colleagues analyzed more than 575,000 vaginal deliveries in New York hospitals between 2010 and 2017; about half (47.4%) of the women received epidurals during labor.
The researchers focused on severe maternal morbidity, including 16 complications, such as heart failure and sepsis, and five procedures, including hysterectomy and ventilation.
They also considered patient characteristics and comorbidities and hospital-related factors to identify patients who were at higher risk for injury or death.
Severe maternal morbidity occurred in 1.3% of the women. Of the 7,712 women with severe morbidity, more than one in three (35.6%) experienced postpartum hemorrhage.
The overall incidence of severe maternal morbidity was 1.3% among women who received an epidural injection and 1.4% among those who did not. In a weighted analysis, the adjusted odds ratio of severe maternal morbidity associated with epidurals was 0.86 (95% confidence interval, 0.82-0.90).
The study is limited by its observational design and does not prove causation, the authors acknowledged.
“Labor neuraxial analgesia may facilitate early evaluation and management of the third stage of labor to avoid escalation of postpartum hemorrhaging into grave complications and death,” study author Jean Guglielminotti, MD, PhD, an anesthesiologist at Columbia University, said in a statement.
Concerning trends
The Department of Health & Human Services has labeled severe maternal morbidity a public health priority. Recent data from the Centers for Disease Control and Prevention show an increase in maternal mortality rates and worsening disparities by race and ethnicity.
According to the CDC, 861 women died of maternal causes in 2020, up from 754 in 2019. The rate of maternal mortality increased from 20.1 to 23.8 deaths per 100,000 live births.
For Black women, however, the maternal mortality rate was far higher: 55.3 deaths per 100,000 live births – nearly triple the figure of 19.1 per 100,000 for White women. Between 2019 and 2020, the mortality rate increased significantly for Black and Hispanic women, but not White mothers.
Researchers affiliated with University of Toronto and the Hospital for Sick Children agreed in an accompanying editorial that more access to neuraxial labor analgesia for vaginal delivery might improve maternal health outcomes and “may be a strategy well worth pursuing in public health policy.”
The intervention is relatively safe and can “alleviate discomfort and distress,” they wrote.
Neuraxial anesthesia in surgical procedures has been shown to decrease the risk for complications like deep vein thrombosis, pulmonary embolus, transfusion requirements, and kidney failure, said editorialists Evelina Pankiv, MD; Alan Yang, MSc; and Kazuyoshi Aoyama, MD, PhD.
Benefits potentially could stem from improving blood flow, mitigating hypercoagulation, or reducing surgical stress response. But there are rare risks to consider as well, including hemorrhage, infection, and neurologic injury, they added.
Guglielminotti disclosed grants from the National Institute on Minority Health and Health Disparities. Dr. Aoyama reported receiving grants from the Perioperative Services Facilitator Grant Program and Hospital for Sick Children. The other authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Use of neuraxial analgesia for vaginal delivery is associated with a 14% decreased risk for severe maternal morbidity, in part from a reduction in postpartum hemorrhage, new research shows.
The findings indicate that increasing the use of epidural or combined spinal-epidural analgesia may improve maternal health outcomes, especially for Hispanic, Black, and uninsured women who are less likely than White women to receive these interventions, according to the researchers, who published their findings online in JAMA Network Open.
About 80% of non-Hispanic White women receive neuraxial analgesia during labor in the United States, compared with 70% of non-Hispanic Black women and 65% of Hispanic women, according to birth certificate data. Among women without health insurance, half receive epidurals.
Programs that inform pregnant women about epidural use, expand Medicaid, and provide in-house obstetric anesthesia teams “may improve patient participation in clinical decision making and access to care,” study author Guohua Li, MD, DrPH, of Columbia University, New York, said in a statement about the research.
Earlier data from France showed that neuraxial analgesia is associated with reduced risk for severe postpartum hemorrhage. To examine the association between labor neuraxial analgesia and severe maternal morbidity in the United States, Dr. Li and colleagues analyzed more than 575,000 vaginal deliveries in New York hospitals between 2010 and 2017; about half (47.4%) of the women received epidurals during labor.
The researchers focused on severe maternal morbidity, including 16 complications, such as heart failure and sepsis, and five procedures, including hysterectomy and ventilation.
They also considered patient characteristics and comorbidities and hospital-related factors to identify patients who were at higher risk for injury or death.
Severe maternal morbidity occurred in 1.3% of the women. Of the 7,712 women with severe morbidity, more than one in three (35.6%) experienced postpartum hemorrhage.
The overall incidence of severe maternal morbidity was 1.3% among women who received an epidural injection and 1.4% among those who did not. In a weighted analysis, the adjusted odds ratio of severe maternal morbidity associated with epidurals was 0.86 (95% confidence interval, 0.82-0.90).
The study is limited by its observational design and does not prove causation, the authors acknowledged.
“Labor neuraxial analgesia may facilitate early evaluation and management of the third stage of labor to avoid escalation of postpartum hemorrhaging into grave complications and death,” study author Jean Guglielminotti, MD, PhD, an anesthesiologist at Columbia University, said in a statement.
Concerning trends
The Department of Health & Human Services has labeled severe maternal morbidity a public health priority. Recent data from the Centers for Disease Control and Prevention show an increase in maternal mortality rates and worsening disparities by race and ethnicity.
According to the CDC, 861 women died of maternal causes in 2020, up from 754 in 2019. The rate of maternal mortality increased from 20.1 to 23.8 deaths per 100,000 live births.
For Black women, however, the maternal mortality rate was far higher: 55.3 deaths per 100,000 live births – nearly triple the figure of 19.1 per 100,000 for White women. Between 2019 and 2020, the mortality rate increased significantly for Black and Hispanic women, but not White mothers.
Researchers affiliated with University of Toronto and the Hospital for Sick Children agreed in an accompanying editorial that more access to neuraxial labor analgesia for vaginal delivery might improve maternal health outcomes and “may be a strategy well worth pursuing in public health policy.”
The intervention is relatively safe and can “alleviate discomfort and distress,” they wrote.
Neuraxial anesthesia in surgical procedures has been shown to decrease the risk for complications like deep vein thrombosis, pulmonary embolus, transfusion requirements, and kidney failure, said editorialists Evelina Pankiv, MD; Alan Yang, MSc; and Kazuyoshi Aoyama, MD, PhD.
Benefits potentially could stem from improving blood flow, mitigating hypercoagulation, or reducing surgical stress response. But there are rare risks to consider as well, including hemorrhage, infection, and neurologic injury, they added.
Guglielminotti disclosed grants from the National Institute on Minority Health and Health Disparities. Dr. Aoyama reported receiving grants from the Perioperative Services Facilitator Grant Program and Hospital for Sick Children. The other authors disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Stress and infertility – is it a proven cause and effect?
“Just relax, stop thinking about it and, more than likely, it will happen.” If ever there was a controversial subject in medicine, especially in reproduction, the relationship between stress and infertility would be high on the list. Who among us has not overheard or even personally shared with an infertility patient that they should try and reduce their stress to improve fertility? The theory is certainly not new. Hippocrates, back in the 5th century B.C., was one of the first to associate a woman’s psychological state with her reproductive potential. His contention was that a physical sign of psychological stress in women (which scholars later dubbed “hysteria”) could result in sterility. In medieval times, a German abbess and mystic named Hildegard of Bingen posited women suffering from melancholy – a condition that we today might call depression – were infertile as a result.
The deeper meaning behind the flippant advice to relax is implicit blame; that is, a woman interprets the link of stress and infertility as a declaration that she is sabotaging reproduction. Not only is this assumption flawed, but it does further damage to a woman’s emotional fragility. To provide the presumption of stress affecting reproduction, a recent survey of over 5,000 infertility patients found, remarkably, 98% considered emotional stress as either a cause or a contributor to infertility, and 31% believed stress was a cause of miscarriage, although racial differences existed (J Assist Reprod Genet. 2021 Apr;38[4]:877-87). This relationship was mostly seen in women who used complementary and alternative medicine, Black women, and those who frequented Internet search engines. Whereas women who had a professional degree, had more infertility insurance coverage, and were nonreligious were less likely to attribute stress to infertility. Intriguingly, the more engaged the physicians, the less patients linked stress with infertility, while the contrary also applied.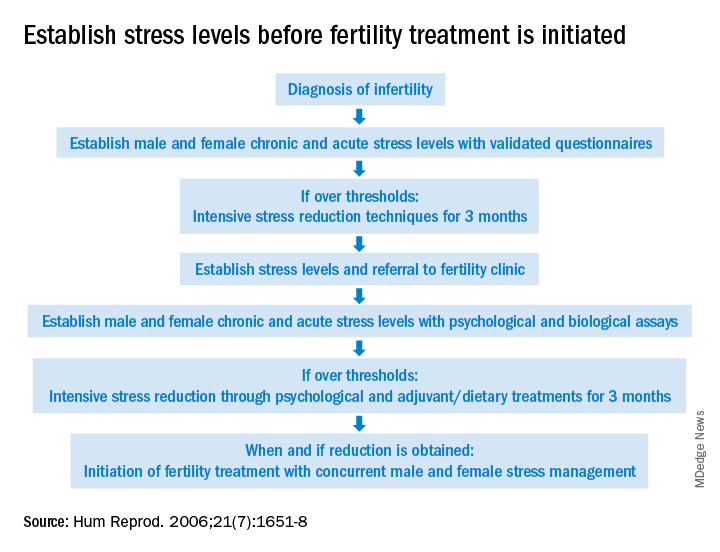
The power of stress can be exemplified by the pathophysiology of amenorrhea. Functional hypothalamic amenorrhea is the most common cause of the female athlete triad of secondary amenorrhea in women of childbearing age. It is a reversible disorder caused by stress related to weight loss, excessive exercise and/or traumatic mental experiences (Endocrines. 2021;2:203-11). Stress of infertility has also been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities (J Psychosom Obstet Gynaecol. 1993;14[Suppl]:45-52).
A definitive link between stress and infertility is evasive because of the lack of controlled, prospective longitudinal studies and the challenge of reducing variables in the analysis. The question remains which developed initially – the stress or the infertility? Infertility treatment is a physical, emotional, and financial investment. Stress and the duration of infertility are correlative. The additive factor is that poor insurance coverage for costly fertility treatment can not only heighten stress but, concurrently, subject the patient to the risk of exploitation driven by desperation whereby they accept unproven “add-ons” offered with assisted reproductive technologies (ART).
Both acute and chronic stress affect the number of oocytes retrieved and fertilized with ART as well as live birth delivery and birth weights (Fertil Steril. 2001;76:675-87). Men are also affected by stress, which is manifested by decreased libido and impaired semen, further compromised as the duration of infertility continues. The gut-derived hormone ghrelin appears to play a role with stress and reproduction (Endocr Rev. 2017;38:432-67).
As the relationship between stress and infertility is far from proven, there are conflicting study results. Two meta-analyses failed to show any association between stress and the outcomes of ART cycles (Hum Reprod. 2011;26:2763-76; BMJ. 2011;342:d223). In contrast, a recent study suggested stress during infertility treatment was contributed by the variables of low spousal support, financial constraints, and social coercion in the early years of marriage (J Hum Reprod Sci. 2018;11:172-9). Emotional distress was found to be three times greater in women whose families had unrealistic expectations from treatments.
Fortunately, psychotherapy during the ART cycle has demonstrated a benefit in outcomes. Domar revealed psychological support and cognitive behavior therapy resulted in higher pregnancy rates than in the control group (Fertil Steril. 2000;73:805-12). Another recent study appears to support stress reduction improving reproductive potential (Dialogues Clin Neurosci. 2018;20[1]:41-7).
Given the evidence provided in this article, it behooves infertility clinics to address baseline (chronic) stress and acute stress (because of infertility) prior to initiating treatment (see Figure). While the definitive answer addressing the impact of stress on reproduction remains unknown, we may share with our patients a definition in which they may find enlightenment, “Stress is trying to control an event in which one is incapable.”
Dr. Mark P Trolice is director of Fertility CARE: The IVF Center in Winter Park, Fla., and associate professor of obstetrics and gynecology at the University of Central Florida, Orlando.
“Just relax, stop thinking about it and, more than likely, it will happen.” If ever there was a controversial subject in medicine, especially in reproduction, the relationship between stress and infertility would be high on the list. Who among us has not overheard or even personally shared with an infertility patient that they should try and reduce their stress to improve fertility? The theory is certainly not new. Hippocrates, back in the 5th century B.C., was one of the first to associate a woman’s psychological state with her reproductive potential. His contention was that a physical sign of psychological stress in women (which scholars later dubbed “hysteria”) could result in sterility. In medieval times, a German abbess and mystic named Hildegard of Bingen posited women suffering from melancholy – a condition that we today might call depression – were infertile as a result.
The deeper meaning behind the flippant advice to relax is implicit blame; that is, a woman interprets the link of stress and infertility as a declaration that she is sabotaging reproduction. Not only is this assumption flawed, but it does further damage to a woman’s emotional fragility. To provide the presumption of stress affecting reproduction, a recent survey of over 5,000 infertility patients found, remarkably, 98% considered emotional stress as either a cause or a contributor to infertility, and 31% believed stress was a cause of miscarriage, although racial differences existed (J Assist Reprod Genet. 2021 Apr;38[4]:877-87). This relationship was mostly seen in women who used complementary and alternative medicine, Black women, and those who frequented Internet search engines. Whereas women who had a professional degree, had more infertility insurance coverage, and were nonreligious were less likely to attribute stress to infertility. Intriguingly, the more engaged the physicians, the less patients linked stress with infertility, while the contrary also applied.
The power of stress can be exemplified by the pathophysiology of amenorrhea. Functional hypothalamic amenorrhea is the most common cause of the female athlete triad of secondary amenorrhea in women of childbearing age. It is a reversible disorder caused by stress related to weight loss, excessive exercise and/or traumatic mental experiences (Endocrines. 2021;2:203-11). Stress of infertility has also been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities (J Psychosom Obstet Gynaecol. 1993;14[Suppl]:45-52).
A definitive link between stress and infertility is evasive because of the lack of controlled, prospective longitudinal studies and the challenge of reducing variables in the analysis. The question remains which developed initially – the stress or the infertility? Infertility treatment is a physical, emotional, and financial investment. Stress and the duration of infertility are correlative. The additive factor is that poor insurance coverage for costly fertility treatment can not only heighten stress but, concurrently, subject the patient to the risk of exploitation driven by desperation whereby they accept unproven “add-ons” offered with assisted reproductive technologies (ART).
Both acute and chronic stress affect the number of oocytes retrieved and fertilized with ART as well as live birth delivery and birth weights (Fertil Steril. 2001;76:675-87). Men are also affected by stress, which is manifested by decreased libido and impaired semen, further compromised as the duration of infertility continues. The gut-derived hormone ghrelin appears to play a role with stress and reproduction (Endocr Rev. 2017;38:432-67).
As the relationship between stress and infertility is far from proven, there are conflicting study results. Two meta-analyses failed to show any association between stress and the outcomes of ART cycles (Hum Reprod. 2011;26:2763-76; BMJ. 2011;342:d223). In contrast, a recent study suggested stress during infertility treatment was contributed by the variables of low spousal support, financial constraints, and social coercion in the early years of marriage (J Hum Reprod Sci. 2018;11:172-9). Emotional distress was found to be three times greater in women whose families had unrealistic expectations from treatments.
Fortunately, psychotherapy during the ART cycle has demonstrated a benefit in outcomes. Domar revealed psychological support and cognitive behavior therapy resulted in higher pregnancy rates than in the control group (Fertil Steril. 2000;73:805-12). Another recent study appears to support stress reduction improving reproductive potential (Dialogues Clin Neurosci. 2018;20[1]:41-7).
Given the evidence provided in this article, it behooves infertility clinics to address baseline (chronic) stress and acute stress (because of infertility) prior to initiating treatment (see Figure). While the definitive answer addressing the impact of stress on reproduction remains unknown, we may share with our patients a definition in which they may find enlightenment, “Stress is trying to control an event in which one is incapable.”
Dr. Mark P Trolice is director of Fertility CARE: The IVF Center in Winter Park, Fla., and associate professor of obstetrics and gynecology at the University of Central Florida, Orlando.
“Just relax, stop thinking about it and, more than likely, it will happen.” If ever there was a controversial subject in medicine, especially in reproduction, the relationship between stress and infertility would be high on the list. Who among us has not overheard or even personally shared with an infertility patient that they should try and reduce their stress to improve fertility? The theory is certainly not new. Hippocrates, back in the 5th century B.C., was one of the first to associate a woman’s psychological state with her reproductive potential. His contention was that a physical sign of psychological stress in women (which scholars later dubbed “hysteria”) could result in sterility. In medieval times, a German abbess and mystic named Hildegard of Bingen posited women suffering from melancholy – a condition that we today might call depression – were infertile as a result.
The deeper meaning behind the flippant advice to relax is implicit blame; that is, a woman interprets the link of stress and infertility as a declaration that she is sabotaging reproduction. Not only is this assumption flawed, but it does further damage to a woman’s emotional fragility. To provide the presumption of stress affecting reproduction, a recent survey of over 5,000 infertility patients found, remarkably, 98% considered emotional stress as either a cause or a contributor to infertility, and 31% believed stress was a cause of miscarriage, although racial differences existed (J Assist Reprod Genet. 2021 Apr;38[4]:877-87). This relationship was mostly seen in women who used complementary and alternative medicine, Black women, and those who frequented Internet search engines. Whereas women who had a professional degree, had more infertility insurance coverage, and were nonreligious were less likely to attribute stress to infertility. Intriguingly, the more engaged the physicians, the less patients linked stress with infertility, while the contrary also applied.
The power of stress can be exemplified by the pathophysiology of amenorrhea. Functional hypothalamic amenorrhea is the most common cause of the female athlete triad of secondary amenorrhea in women of childbearing age. It is a reversible disorder caused by stress related to weight loss, excessive exercise and/or traumatic mental experiences (Endocrines. 2021;2:203-11). Stress of infertility has also been demonstrated to be equivalent to a diagnosis of cancer and other major medical morbidities (J Psychosom Obstet Gynaecol. 1993;14[Suppl]:45-52).
A definitive link between stress and infertility is evasive because of the lack of controlled, prospective longitudinal studies and the challenge of reducing variables in the analysis. The question remains which developed initially – the stress or the infertility? Infertility treatment is a physical, emotional, and financial investment. Stress and the duration of infertility are correlative. The additive factor is that poor insurance coverage for costly fertility treatment can not only heighten stress but, concurrently, subject the patient to the risk of exploitation driven by desperation whereby they accept unproven “add-ons” offered with assisted reproductive technologies (ART).
Both acute and chronic stress affect the number of oocytes retrieved and fertilized with ART as well as live birth delivery and birth weights (Fertil Steril. 2001;76:675-87). Men are also affected by stress, which is manifested by decreased libido and impaired semen, further compromised as the duration of infertility continues. The gut-derived hormone ghrelin appears to play a role with stress and reproduction (Endocr Rev. 2017;38:432-67).
As the relationship between stress and infertility is far from proven, there are conflicting study results. Two meta-analyses failed to show any association between stress and the outcomes of ART cycles (Hum Reprod. 2011;26:2763-76; BMJ. 2011;342:d223). In contrast, a recent study suggested stress during infertility treatment was contributed by the variables of low spousal support, financial constraints, and social coercion in the early years of marriage (J Hum Reprod Sci. 2018;11:172-9). Emotional distress was found to be three times greater in women whose families had unrealistic expectations from treatments.
Fortunately, psychotherapy during the ART cycle has demonstrated a benefit in outcomes. Domar revealed psychological support and cognitive behavior therapy resulted in higher pregnancy rates than in the control group (Fertil Steril. 2000;73:805-12). Another recent study appears to support stress reduction improving reproductive potential (Dialogues Clin Neurosci. 2018;20[1]:41-7).
Given the evidence provided in this article, it behooves infertility clinics to address baseline (chronic) stress and acute stress (because of infertility) prior to initiating treatment (see Figure). While the definitive answer addressing the impact of stress on reproduction remains unknown, we may share with our patients a definition in which they may find enlightenment, “Stress is trying to control an event in which one is incapable.”
Dr. Mark P Trolice is director of Fertility CARE: The IVF Center in Winter Park, Fla., and associate professor of obstetrics and gynecology at the University of Central Florida, Orlando.
More than half of U.S. women enter pregnancy at higher CVD risk
Less than half of women in the United States enter pregnancy in favorable cardiovascular health, new research suggests.
In 2019, among women aged 20 to 44 years with live births in the United States, only 40.2% were in favorable cardiovascular health prior to pregnancy, defined as normal weight, no diabetes, and no hypertension.
Although all regions and states showed a decline in prepregnancy favorable cardiometabolic health, there were significant differences among geographic regions in the country, the authors report. “These data reveal critical deficiencies and geographic disparities in prepregnancy cardiometabolic health,” they conclude.
“One of the things that we know in the U.S. is that the maternal mortality rate has been increasing, and there are significant differences at the state level in both adverse maternal outcomes, such as maternal mortality, as well as adverse pregnancy outcomes,” corresponding author Sadiya S. Khan, MD, MS, FACC, Northwestern University Feinberg School of Medicine, Chicago, told this news organization.
“These outcomes are often related to health factors that predate pregnancy,” Dr. Khan explained, “and the processes that begin at the very, very beginning of conception are informed by health factors prior to pregnancy, in particular cardiometabolic factors like body mass index or obesity, high blood pressure, and diabetes.”
The results were published online on Feb. 14 in a special “Go Red for Women” spotlight issue of Circulation.
Cardiometabolic health factors
Using maternal birth records from live births in the Centers for Disease Control and Prevention Natality Database between 2016 and 2019, the authors analyzed data on 14,174,625 women with live births aged 20 to 44 years. The majority (81.4%) were 20 to 34 years of age, 22.7% were Hispanic or Latina, and 52.7% were non-Hispanic White.
Favorable cardiometabolic health was defined as a BMI of 18 to 24.9 kg/m2, absence of diabetes, and absence of hypertension.
Although all regions and states experienced a decline in favorable cardiometabolic health during the study period of 2016 to 2019, with a drop overall of 3.2% – from 43.5 to 40.2 per 100 live births – it was especially true of the South and Midwest regions.
In 2019, favorable prepregnancy cardiometabolic health was lowest in the South (38.1%) and Midwest (38.8%) and highest in the West (42.2%) and Northeast (43.6%).
State by state, the lowest prevalence of favorable cardiometabolic health was found in Mississippi, at 31.2%, and highest in Utah, at 47.2%.
They also found a correlation between favorable cardiometabolic health and state-level percentages of high-school education or less and enrollment in Medicaid in 2019.
Similar to what has been seen with cardiovascular disease, “we observe that the states with the lowest prevalence of favorable cardiometabolic health were in the Southeast United States,” said Dr. Khan, “and similar geographic variation was observed with some more patterns in education and Medicaid coverage for birth, and these were used as proxies for socioeconomic status in those areas.”
Although Dr. Khan notes that the relationships cannot be determined to be causal from this analysis, she said that “it does suggest that upstream social determinants of health are important determinants of cardiometabolic health.”
Socioeconomic intervention
Dr. Khan noted that policies at the federal and state level can identify ways to “ensure that individuals who are thinking about pregnancy have access to health care and have access to resources, too, from a broad range of health determinants, including housing stability, food security, as well as access to health care be optimized prior to pregnancy.”
The authors note that this analysis may actually overestimate the prevalence of favorable cardiometabolic health, and data on cholesterol, diet, a distinction between type 1 and type 2 diabetes, and physical activity were not available.
Only individuals with live births were included, which could result in the elimination of a potentially high-risk group; however, late pregnancy losses represent less than 0.3% of all pregnancies, they say.
The authors conclude that “future research is needed to equitably improve health prior to pregnancy and quantify the potential benefits in cardiovascular disease outcomes for birthing individuals and their offspring.”
This work was supported by grants from the National Heart, Lung, and Blood Institute and American Heart Association Transformational Project Award awarded to Sadiya S. Khan.
A version of this article first appeared on Medscape.com.
Less than half of women in the United States enter pregnancy in favorable cardiovascular health, new research suggests.
In 2019, among women aged 20 to 44 years with live births in the United States, only 40.2% were in favorable cardiovascular health prior to pregnancy, defined as normal weight, no diabetes, and no hypertension.
Although all regions and states showed a decline in prepregnancy favorable cardiometabolic health, there were significant differences among geographic regions in the country, the authors report. “These data reveal critical deficiencies and geographic disparities in prepregnancy cardiometabolic health,” they conclude.
“One of the things that we know in the U.S. is that the maternal mortality rate has been increasing, and there are significant differences at the state level in both adverse maternal outcomes, such as maternal mortality, as well as adverse pregnancy outcomes,” corresponding author Sadiya S. Khan, MD, MS, FACC, Northwestern University Feinberg School of Medicine, Chicago, told this news organization.
“These outcomes are often related to health factors that predate pregnancy,” Dr. Khan explained, “and the processes that begin at the very, very beginning of conception are informed by health factors prior to pregnancy, in particular cardiometabolic factors like body mass index or obesity, high blood pressure, and diabetes.”
The results were published online on Feb. 14 in a special “Go Red for Women” spotlight issue of Circulation.
Cardiometabolic health factors
Using maternal birth records from live births in the Centers for Disease Control and Prevention Natality Database between 2016 and 2019, the authors analyzed data on 14,174,625 women with live births aged 20 to 44 years. The majority (81.4%) were 20 to 34 years of age, 22.7% were Hispanic or Latina, and 52.7% were non-Hispanic White.
Favorable cardiometabolic health was defined as a BMI of 18 to 24.9 kg/m2, absence of diabetes, and absence of hypertension.
Although all regions and states experienced a decline in favorable cardiometabolic health during the study period of 2016 to 2019, with a drop overall of 3.2% – from 43.5 to 40.2 per 100 live births – it was especially true of the South and Midwest regions.
In 2019, favorable prepregnancy cardiometabolic health was lowest in the South (38.1%) and Midwest (38.8%) and highest in the West (42.2%) and Northeast (43.6%).
State by state, the lowest prevalence of favorable cardiometabolic health was found in Mississippi, at 31.2%, and highest in Utah, at 47.2%.
They also found a correlation between favorable cardiometabolic health and state-level percentages of high-school education or less and enrollment in Medicaid in 2019.
Similar to what has been seen with cardiovascular disease, “we observe that the states with the lowest prevalence of favorable cardiometabolic health were in the Southeast United States,” said Dr. Khan, “and similar geographic variation was observed with some more patterns in education and Medicaid coverage for birth, and these were used as proxies for socioeconomic status in those areas.”
Although Dr. Khan notes that the relationships cannot be determined to be causal from this analysis, she said that “it does suggest that upstream social determinants of health are important determinants of cardiometabolic health.”
Socioeconomic intervention
Dr. Khan noted that policies at the federal and state level can identify ways to “ensure that individuals who are thinking about pregnancy have access to health care and have access to resources, too, from a broad range of health determinants, including housing stability, food security, as well as access to health care be optimized prior to pregnancy.”
The authors note that this analysis may actually overestimate the prevalence of favorable cardiometabolic health, and data on cholesterol, diet, a distinction between type 1 and type 2 diabetes, and physical activity were not available.
Only individuals with live births were included, which could result in the elimination of a potentially high-risk group; however, late pregnancy losses represent less than 0.3% of all pregnancies, they say.
The authors conclude that “future research is needed to equitably improve health prior to pregnancy and quantify the potential benefits in cardiovascular disease outcomes for birthing individuals and their offspring.”
This work was supported by grants from the National Heart, Lung, and Blood Institute and American Heart Association Transformational Project Award awarded to Sadiya S. Khan.
A version of this article first appeared on Medscape.com.
Less than half of women in the United States enter pregnancy in favorable cardiovascular health, new research suggests.
In 2019, among women aged 20 to 44 years with live births in the United States, only 40.2% were in favorable cardiovascular health prior to pregnancy, defined as normal weight, no diabetes, and no hypertension.
Although all regions and states showed a decline in prepregnancy favorable cardiometabolic health, there were significant differences among geographic regions in the country, the authors report. “These data reveal critical deficiencies and geographic disparities in prepregnancy cardiometabolic health,” they conclude.
“One of the things that we know in the U.S. is that the maternal mortality rate has been increasing, and there are significant differences at the state level in both adverse maternal outcomes, such as maternal mortality, as well as adverse pregnancy outcomes,” corresponding author Sadiya S. Khan, MD, MS, FACC, Northwestern University Feinberg School of Medicine, Chicago, told this news organization.
“These outcomes are often related to health factors that predate pregnancy,” Dr. Khan explained, “and the processes that begin at the very, very beginning of conception are informed by health factors prior to pregnancy, in particular cardiometabolic factors like body mass index or obesity, high blood pressure, and diabetes.”
The results were published online on Feb. 14 in a special “Go Red for Women” spotlight issue of Circulation.
Cardiometabolic health factors
Using maternal birth records from live births in the Centers for Disease Control and Prevention Natality Database between 2016 and 2019, the authors analyzed data on 14,174,625 women with live births aged 20 to 44 years. The majority (81.4%) were 20 to 34 years of age, 22.7% were Hispanic or Latina, and 52.7% were non-Hispanic White.
Favorable cardiometabolic health was defined as a BMI of 18 to 24.9 kg/m2, absence of diabetes, and absence of hypertension.
Although all regions and states experienced a decline in favorable cardiometabolic health during the study period of 2016 to 2019, with a drop overall of 3.2% – from 43.5 to 40.2 per 100 live births – it was especially true of the South and Midwest regions.
In 2019, favorable prepregnancy cardiometabolic health was lowest in the South (38.1%) and Midwest (38.8%) and highest in the West (42.2%) and Northeast (43.6%).
State by state, the lowest prevalence of favorable cardiometabolic health was found in Mississippi, at 31.2%, and highest in Utah, at 47.2%.
They also found a correlation between favorable cardiometabolic health and state-level percentages of high-school education or less and enrollment in Medicaid in 2019.
Similar to what has been seen with cardiovascular disease, “we observe that the states with the lowest prevalence of favorable cardiometabolic health were in the Southeast United States,” said Dr. Khan, “and similar geographic variation was observed with some more patterns in education and Medicaid coverage for birth, and these were used as proxies for socioeconomic status in those areas.”
Although Dr. Khan notes that the relationships cannot be determined to be causal from this analysis, she said that “it does suggest that upstream social determinants of health are important determinants of cardiometabolic health.”
Socioeconomic intervention
Dr. Khan noted that policies at the federal and state level can identify ways to “ensure that individuals who are thinking about pregnancy have access to health care and have access to resources, too, from a broad range of health determinants, including housing stability, food security, as well as access to health care be optimized prior to pregnancy.”
The authors note that this analysis may actually overestimate the prevalence of favorable cardiometabolic health, and data on cholesterol, diet, a distinction between type 1 and type 2 diabetes, and physical activity were not available.
Only individuals with live births were included, which could result in the elimination of a potentially high-risk group; however, late pregnancy losses represent less than 0.3% of all pregnancies, they say.
The authors conclude that “future research is needed to equitably improve health prior to pregnancy and quantify the potential benefits in cardiovascular disease outcomes for birthing individuals and their offspring.”
This work was supported by grants from the National Heart, Lung, and Blood Institute and American Heart Association Transformational Project Award awarded to Sadiya S. Khan.
A version of this article first appeared on Medscape.com.
More than half of U.S. abortions now done with pills: Report
according to a report from the Guttmacher Institute, a research group that supports abortion rights.
A survey of abortion providers showed that 54% of all U.S. abortions were done with medication in 2020, marking the first time the proportion of medication abortions topped 50%, Guttmacher said.
In 2017, the last time such a survey was done, 39% of abortions were performed by medication, Guttmacher said. The organization said 24% of abortions were done with medication in 2011 and 6% in 2001, the year after the FDA approved the pills.
The 54% estimate is based on early findings, Guttmacher said in a news release. It said that “final estimates will be released in late 2022 and the proportion for medication abortion use is not expected to fall below 50%.”
Rachel Jones, PhD, a Guttmacher researcher, said the higher use of abortion pills may be linked to increases in telemedicine because of the COVID-19 pandemic and the FDA’s decision last year to allow the mailing of abortion pills to patients, the Associated Press reported. Those changes mean women can now consult with a doctor online, receive the pills by mail, and complete the abortion at home.
Abortion pills are recommended for the first 10 weeks of pregnancy, though research shows they can be safe in some cases after 10 weeks, Guttmacher said. Patients take the pill mifepristone, which blocks a hormone needed for pregnancy to continue, and a few days later take the pill misoprostol, which causes cramping that empties the womb.
“The introduction and availability of medication abortion has proven to be a game changer in expanding abortion care in the United States, and it will likely be an even more important option for people to obtain an abortion as many states continue to pass legislation to bar or restrict abortion access,” Guttmacher said in the news release.
Arizona, Arkansas, and Texas have banned the mailing of abortion pills. Similar bans were approved in Montana, Oklahoma, and South Dakota but were blocked in the courts, Guttmacher said.
Sixteen state legislatures have proposed bans or restrictions on medication-induced abortion this year, while 32 states require this type of abortion to be prescribed by doctors.
In Texas, orders for abortion pills increased sharply after the state legislature approved a highly restrictive abortion law, Politico reported, citing a study in The Journal of the American Medical Association.
Orders went up 1,180% in the first week after the Texas law took effect in September, researchers said. Orders dipped somewhat in later weeks but remained 175% higher than before the Texas law took effect.
A version of this article first appeared on WebMD.com.
according to a report from the Guttmacher Institute, a research group that supports abortion rights.
A survey of abortion providers showed that 54% of all U.S. abortions were done with medication in 2020, marking the first time the proportion of medication abortions topped 50%, Guttmacher said.
In 2017, the last time such a survey was done, 39% of abortions were performed by medication, Guttmacher said. The organization said 24% of abortions were done with medication in 2011 and 6% in 2001, the year after the FDA approved the pills.
The 54% estimate is based on early findings, Guttmacher said in a news release. It said that “final estimates will be released in late 2022 and the proportion for medication abortion use is not expected to fall below 50%.”
Rachel Jones, PhD, a Guttmacher researcher, said the higher use of abortion pills may be linked to increases in telemedicine because of the COVID-19 pandemic and the FDA’s decision last year to allow the mailing of abortion pills to patients, the Associated Press reported. Those changes mean women can now consult with a doctor online, receive the pills by mail, and complete the abortion at home.
Abortion pills are recommended for the first 10 weeks of pregnancy, though research shows they can be safe in some cases after 10 weeks, Guttmacher said. Patients take the pill mifepristone, which blocks a hormone needed for pregnancy to continue, and a few days later take the pill misoprostol, which causes cramping that empties the womb.
“The introduction and availability of medication abortion has proven to be a game changer in expanding abortion care in the United States, and it will likely be an even more important option for people to obtain an abortion as many states continue to pass legislation to bar or restrict abortion access,” Guttmacher said in the news release.
Arizona, Arkansas, and Texas have banned the mailing of abortion pills. Similar bans were approved in Montana, Oklahoma, and South Dakota but were blocked in the courts, Guttmacher said.
Sixteen state legislatures have proposed bans or restrictions on medication-induced abortion this year, while 32 states require this type of abortion to be prescribed by doctors.
In Texas, orders for abortion pills increased sharply after the state legislature approved a highly restrictive abortion law, Politico reported, citing a study in The Journal of the American Medical Association.
Orders went up 1,180% in the first week after the Texas law took effect in September, researchers said. Orders dipped somewhat in later weeks but remained 175% higher than before the Texas law took effect.
A version of this article first appeared on WebMD.com.
according to a report from the Guttmacher Institute, a research group that supports abortion rights.
A survey of abortion providers showed that 54% of all U.S. abortions were done with medication in 2020, marking the first time the proportion of medication abortions topped 50%, Guttmacher said.
In 2017, the last time such a survey was done, 39% of abortions were performed by medication, Guttmacher said. The organization said 24% of abortions were done with medication in 2011 and 6% in 2001, the year after the FDA approved the pills.
The 54% estimate is based on early findings, Guttmacher said in a news release. It said that “final estimates will be released in late 2022 and the proportion for medication abortion use is not expected to fall below 50%.”
Rachel Jones, PhD, a Guttmacher researcher, said the higher use of abortion pills may be linked to increases in telemedicine because of the COVID-19 pandemic and the FDA’s decision last year to allow the mailing of abortion pills to patients, the Associated Press reported. Those changes mean women can now consult with a doctor online, receive the pills by mail, and complete the abortion at home.
Abortion pills are recommended for the first 10 weeks of pregnancy, though research shows they can be safe in some cases after 10 weeks, Guttmacher said. Patients take the pill mifepristone, which blocks a hormone needed for pregnancy to continue, and a few days later take the pill misoprostol, which causes cramping that empties the womb.
“The introduction and availability of medication abortion has proven to be a game changer in expanding abortion care in the United States, and it will likely be an even more important option for people to obtain an abortion as many states continue to pass legislation to bar or restrict abortion access,” Guttmacher said in the news release.
Arizona, Arkansas, and Texas have banned the mailing of abortion pills. Similar bans were approved in Montana, Oklahoma, and South Dakota but were blocked in the courts, Guttmacher said.
Sixteen state legislatures have proposed bans or restrictions on medication-induced abortion this year, while 32 states require this type of abortion to be prescribed by doctors.
In Texas, orders for abortion pills increased sharply after the state legislature approved a highly restrictive abortion law, Politico reported, citing a study in The Journal of the American Medical Association.
Orders went up 1,180% in the first week after the Texas law took effect in September, researchers said. Orders dipped somewhat in later weeks but remained 175% higher than before the Texas law took effect.
A version of this article first appeared on WebMD.com.
Genetic mutation tied to moms’ perception of inadequate milk supply
The perception of an inadequate milk supply can contribute to a new mother’s decision to cease breastfeeding prematurely, but identifying at-risk women is difficult. Now a new study suggests that, for some women, this impression, known as perceived inadequacy of milk supply (PIMS) appears to have a basis in genetics.
Steven D. Hicks, MD, PhD, associate professor of pediatrics at Penn State University, Hershey, and colleagues found that variations in the MFGE8 gene was associated with perceived breast milk supply.
“Genotyping one lactogenic gene aided identification of mothers at risk for PIMS. If validated in a larger cohort, such an approach could be used to identify mothers who may benefit from increased lactation support.” Dr. Hicks’ group wrote in Breastfeeding Medicine.
A simple 24-hour polymerase chain reaction test around the time of delivery could identify vulnerable mothers, who could receive targeted lactation support including galactagogue supplementation and early scheduling of “nurse and pump” routines.
“Also, if a woman is found to have PIMS, she would definitely want to avoid going on oral contraceptives after delivery as these can affect milk supply,” Dr. Hicks said in an interview. “That would be like a double hit in a woman already at risk.“
In other measures, she would meet once a week with a lactation consultant for the first 6 months to make sure good breastfeeding practices as to frequency and duration of placing the baby on the breast are being maintained, Dr. Hicks continued. “She needs to make sure her supply isn’t going backwards because once it starts to dry up, it’s really hard to turn the ship around.”
Dietary interventions to promote lactation may also help.
The study
In the context of a larger study on breast milk factors protecting children from developing allergies, the prospective observational analysis collected data on 221 breastfeeding mothers aged 19-42 years (mean age, 29 years) for 12 months.
All mothers had initially planned to nurse their babies for at least 6 months but some stopped well short of their goal.
“At least a third told us they gave up because they felt their milk supply had run out,” Dr. Hicks said in an interview.
Using salivas swabs, the investigators assessed 18 genes secreted from mammary tissue and previously linked in cows to secretory function for single-nucleotide polymorphisms in 88 age-, race-, and parity-matched women. Of these, 45 had PIMS and 43 had perceived adequate milk supply (PAMS).
Among differences between the two groups:
- PIMS mothers breastfed exclusively for a shorter period than PAMS mothers: 7 ± 12 weeks versus 22 ± 19 weeks(P = .001)
- At 1 month after delivery, their reported milk production was lower: 17.6 ± 13.3 oz/day versus 27.0 ± 12.2 oz/day (P = .0001). This shortfall persisted at 4 months: 16.0 ± 14.1 oz/day versus 27.3 ± 14.9 oz/day (P < .0010.
- Between birth and 1 month, infants of PIMS mothers also showed a smaller average increase in weight-for-length z score between birth and 1 month: 0.74 ± 1.4 versus 1.4 ± 1.5 (P = .038).
Furthermore, maternal genotype for the rs2271714 variant within MFGE8 was associated with PIMS status (P = .009; adjusted P = .09; likelihood ratio, 9.33), and duration of exclusive breastfeeding (P = .009; adjusted P = .09; chi square, 9.39).
Adding the MFGE8 genotype to a model including maternal age, parity, previous breastfeeding duration, body mass index, education, and depression status significantly increased predictive accuracy for PIMS status, the authors noted.
“This is a very exciting observation made by Dr. Hicks and his team, and the first study to identify a genetic variant associated with perceived insufficient milk supply,” said Shannon L. Kelleher, PhD, professor of biological and nutritional sciences at University of Massachusetts, Lowell.
“It provides compelling evidence that insufficient milk supply is very common and supports the urgent need to identify the risk factors and develop interventions to improve breastfeeding success,” added Dr. Kelleher, who was not involved in the study.
“If genetic variants such as the one identified in MFGE8 are confirmed to be common in mothers with insufficient milk supply, the development of lactation-focused genetic tests to identify women at risk for low milk supply would revolutionize our ability to support breastfeeding mothers and help them achieve their breastfeeding goals.”
Although 83% of new mothers initiate breastfeeding, only 57% are still breastfeeding at 6 months and only 35% breastfeed until 12 months, according to the Centers for Disease Control and Prevention’s Breastfeeding Report Card.
This study was funded by grants to Dr. Hicks from the Gerber Foundation and the Center for Research on Women and Newborns Foundation. The authors disclosed no competing interests. Dr. Kelleher reported no conflicts of interest relevant to her comments.
The perception of an inadequate milk supply can contribute to a new mother’s decision to cease breastfeeding prematurely, but identifying at-risk women is difficult. Now a new study suggests that, for some women, this impression, known as perceived inadequacy of milk supply (PIMS) appears to have a basis in genetics.
Steven D. Hicks, MD, PhD, associate professor of pediatrics at Penn State University, Hershey, and colleagues found that variations in the MFGE8 gene was associated with perceived breast milk supply.
“Genotyping one lactogenic gene aided identification of mothers at risk for PIMS. If validated in a larger cohort, such an approach could be used to identify mothers who may benefit from increased lactation support.” Dr. Hicks’ group wrote in Breastfeeding Medicine.
A simple 24-hour polymerase chain reaction test around the time of delivery could identify vulnerable mothers, who could receive targeted lactation support including galactagogue supplementation and early scheduling of “nurse and pump” routines.
“Also, if a woman is found to have PIMS, she would definitely want to avoid going on oral contraceptives after delivery as these can affect milk supply,” Dr. Hicks said in an interview. “That would be like a double hit in a woman already at risk.“
In other measures, she would meet once a week with a lactation consultant for the first 6 months to make sure good breastfeeding practices as to frequency and duration of placing the baby on the breast are being maintained, Dr. Hicks continued. “She needs to make sure her supply isn’t going backwards because once it starts to dry up, it’s really hard to turn the ship around.”
Dietary interventions to promote lactation may also help.
The study
In the context of a larger study on breast milk factors protecting children from developing allergies, the prospective observational analysis collected data on 221 breastfeeding mothers aged 19-42 years (mean age, 29 years) for 12 months.
All mothers had initially planned to nurse their babies for at least 6 months but some stopped well short of their goal.
“At least a third told us they gave up because they felt their milk supply had run out,” Dr. Hicks said in an interview.
Using salivas swabs, the investigators assessed 18 genes secreted from mammary tissue and previously linked in cows to secretory function for single-nucleotide polymorphisms in 88 age-, race-, and parity-matched women. Of these, 45 had PIMS and 43 had perceived adequate milk supply (PAMS).
Among differences between the two groups:
- PIMS mothers breastfed exclusively for a shorter period than PAMS mothers: 7 ± 12 weeks versus 22 ± 19 weeks(P = .001)
- At 1 month after delivery, their reported milk production was lower: 17.6 ± 13.3 oz/day versus 27.0 ± 12.2 oz/day (P = .0001). This shortfall persisted at 4 months: 16.0 ± 14.1 oz/day versus 27.3 ± 14.9 oz/day (P < .0010.
- Between birth and 1 month, infants of PIMS mothers also showed a smaller average increase in weight-for-length z score between birth and 1 month: 0.74 ± 1.4 versus 1.4 ± 1.5 (P = .038).
Furthermore, maternal genotype for the rs2271714 variant within MFGE8 was associated with PIMS status (P = .009; adjusted P = .09; likelihood ratio, 9.33), and duration of exclusive breastfeeding (P = .009; adjusted P = .09; chi square, 9.39).
Adding the MFGE8 genotype to a model including maternal age, parity, previous breastfeeding duration, body mass index, education, and depression status significantly increased predictive accuracy for PIMS status, the authors noted.
“This is a very exciting observation made by Dr. Hicks and his team, and the first study to identify a genetic variant associated with perceived insufficient milk supply,” said Shannon L. Kelleher, PhD, professor of biological and nutritional sciences at University of Massachusetts, Lowell.
“It provides compelling evidence that insufficient milk supply is very common and supports the urgent need to identify the risk factors and develop interventions to improve breastfeeding success,” added Dr. Kelleher, who was not involved in the study.
“If genetic variants such as the one identified in MFGE8 are confirmed to be common in mothers with insufficient milk supply, the development of lactation-focused genetic tests to identify women at risk for low milk supply would revolutionize our ability to support breastfeeding mothers and help them achieve their breastfeeding goals.”
Although 83% of new mothers initiate breastfeeding, only 57% are still breastfeeding at 6 months and only 35% breastfeed until 12 months, according to the Centers for Disease Control and Prevention’s Breastfeeding Report Card.
This study was funded by grants to Dr. Hicks from the Gerber Foundation and the Center for Research on Women and Newborns Foundation. The authors disclosed no competing interests. Dr. Kelleher reported no conflicts of interest relevant to her comments.
The perception of an inadequate milk supply can contribute to a new mother’s decision to cease breastfeeding prematurely, but identifying at-risk women is difficult. Now a new study suggests that, for some women, this impression, known as perceived inadequacy of milk supply (PIMS) appears to have a basis in genetics.
Steven D. Hicks, MD, PhD, associate professor of pediatrics at Penn State University, Hershey, and colleagues found that variations in the MFGE8 gene was associated with perceived breast milk supply.
“Genotyping one lactogenic gene aided identification of mothers at risk for PIMS. If validated in a larger cohort, such an approach could be used to identify mothers who may benefit from increased lactation support.” Dr. Hicks’ group wrote in Breastfeeding Medicine.
A simple 24-hour polymerase chain reaction test around the time of delivery could identify vulnerable mothers, who could receive targeted lactation support including galactagogue supplementation and early scheduling of “nurse and pump” routines.
“Also, if a woman is found to have PIMS, she would definitely want to avoid going on oral contraceptives after delivery as these can affect milk supply,” Dr. Hicks said in an interview. “That would be like a double hit in a woman already at risk.“
In other measures, she would meet once a week with a lactation consultant for the first 6 months to make sure good breastfeeding practices as to frequency and duration of placing the baby on the breast are being maintained, Dr. Hicks continued. “She needs to make sure her supply isn’t going backwards because once it starts to dry up, it’s really hard to turn the ship around.”
Dietary interventions to promote lactation may also help.
The study
In the context of a larger study on breast milk factors protecting children from developing allergies, the prospective observational analysis collected data on 221 breastfeeding mothers aged 19-42 years (mean age, 29 years) for 12 months.
All mothers had initially planned to nurse their babies for at least 6 months but some stopped well short of their goal.
“At least a third told us they gave up because they felt their milk supply had run out,” Dr. Hicks said in an interview.
Using salivas swabs, the investigators assessed 18 genes secreted from mammary tissue and previously linked in cows to secretory function for single-nucleotide polymorphisms in 88 age-, race-, and parity-matched women. Of these, 45 had PIMS and 43 had perceived adequate milk supply (PAMS).
Among differences between the two groups:
- PIMS mothers breastfed exclusively for a shorter period than PAMS mothers: 7 ± 12 weeks versus 22 ± 19 weeks(P = .001)
- At 1 month after delivery, their reported milk production was lower: 17.6 ± 13.3 oz/day versus 27.0 ± 12.2 oz/day (P = .0001). This shortfall persisted at 4 months: 16.0 ± 14.1 oz/day versus 27.3 ± 14.9 oz/day (P < .0010.
- Between birth and 1 month, infants of PIMS mothers also showed a smaller average increase in weight-for-length z score between birth and 1 month: 0.74 ± 1.4 versus 1.4 ± 1.5 (P = .038).
Furthermore, maternal genotype for the rs2271714 variant within MFGE8 was associated with PIMS status (P = .009; adjusted P = .09; likelihood ratio, 9.33), and duration of exclusive breastfeeding (P = .009; adjusted P = .09; chi square, 9.39).
Adding the MFGE8 genotype to a model including maternal age, parity, previous breastfeeding duration, body mass index, education, and depression status significantly increased predictive accuracy for PIMS status, the authors noted.
“This is a very exciting observation made by Dr. Hicks and his team, and the first study to identify a genetic variant associated with perceived insufficient milk supply,” said Shannon L. Kelleher, PhD, professor of biological and nutritional sciences at University of Massachusetts, Lowell.
“It provides compelling evidence that insufficient milk supply is very common and supports the urgent need to identify the risk factors and develop interventions to improve breastfeeding success,” added Dr. Kelleher, who was not involved in the study.
“If genetic variants such as the one identified in MFGE8 are confirmed to be common in mothers with insufficient milk supply, the development of lactation-focused genetic tests to identify women at risk for low milk supply would revolutionize our ability to support breastfeeding mothers and help them achieve their breastfeeding goals.”
Although 83% of new mothers initiate breastfeeding, only 57% are still breastfeeding at 6 months and only 35% breastfeed until 12 months, according to the Centers for Disease Control and Prevention’s Breastfeeding Report Card.
This study was funded by grants to Dr. Hicks from the Gerber Foundation and the Center for Research on Women and Newborns Foundation. The authors disclosed no competing interests. Dr. Kelleher reported no conflicts of interest relevant to her comments.
FROM BREASTFEEDING MEDICINE
Variants of nine breast cancer genes associated with severe disease
Breast cancer risk genes are generally associated with triple-negative and high-grade disease, but differ substantially in their associated pathology, a large case-control analysis shows.
The findings have potential implications for genetic testing, risk prediction, variant classification, and screening, the authors noted.
To assess links between pathogenic germline variants in nine major breast cancer (BC) susceptibility genes and pathological features of nonmetastasized breast tumors, investigators from the Breast Cancer Association Consortium compared 42,680 women with breast cancer and 42,387 population-matched controls from the BRIDGES large-scale sequencing study.
They looked specifically at features relevant to prognosis and distinct therapeutic options, including tumor subtype, morphology, size, stage, and lymph node involvement, and found substantial differences in tumor subtype distribution by gene.
“Pathogenic [protein-truncating variants and pathogenic missense variants] in these ... they wrote, noting that carriers of rare genetic variants in the nine genes comprised more than 27% of women diagnosed at age 40 or younger with triple-negative disease (driven mainly by BRCA1) and about 16% of those diagnosed with hormone receptor (HR)–positive, HER2-negative high-grade disease.
All together, the nine genes were associated with 14.4% of tumors in women aged 40 years and younger, but less than 4% in women over age 60 years, and among younger women, the prevalence of variants combined was higher in those with triple-negative and HR-positive, HER2-negative disease versus other subtypes, they said.
The findings were published online on Jan. 27, 2022, in JAMA Oncology.
Study subjects were women aged 18-79 years who were sampled, independently of family history, from 38 international population- or hospital-based studies conducted between 1991 and 2016.
The genes assessed included ATM, BARD1, BRCA1, BRCA2, CHEK2, PALB2, RAD51C, RAD51D, and TP53.
Substantial heterogeneity was observed in the distribution of intrinsic subtypes by gene. For example, RAD51C, RAD51D, and BARD1 variants were associated mainly with triple-negative disease (odds ratios, 6.19, 6.19, and 10.05 respectively), and CHEK2 variants were associated with all subtypes (with ORs ranging from 2.21-3.17) except for triple-negative disease, the authors found.
“For ATM variants, the association was strongest for [HR-positive, HER2-negative]high-grade subtype (OR, 4.99). BRCA1 was associated with increased risk of all subtypes, but the ORs varied widely, being highest for triple-negative disease (OR, 55.32),” they wrote.
BRCA2 and PALB2 variants were also associated with triple-negative disease, TP53 variants were most strongly associated with HR-positive, HER2-negative and HR-negative, HER2-positive subtypes, and tumors occurring in pathogenic variant carriers were of higher grade, they added, noting that for most genes and subtypes, a decline in ORs was observed with increasing age.
All genes except CHEK2 were more strongly associated with high-grade disease.
These and other findings from the study can “inform guidelines for eligibility for gene panel sequencing and breast cancer surveillance in the general population,” the authors explained, adding that tumor characteristics can also be used to determine whether variants of uncertain significance are likely to be pathogenic.
The data should therefore “improve the precision of variant classification algorithms and extend them to a larger set of genes,” they noted.
“This case-control study suggests that rare variants in BC susceptibility genes display marked heterogeneity with respect to tumor phenotype, but also similarities between genes that are consistent with known biological functions. This present study provides detailed quantification of subtype-specific breast cancer risks; these can potentially improve risk prediction models and breast cancer prevention strategies,” they concluded.
More specifically, women found to be carrying variants in these genes may be offered enhanced screening, including by MRI, risk-reducing surgery, chemoprevention, and genetic counseling, corresponding author Nasim Mavaddat, MBBS, PhD, explained in an interview.
“Understanding tumor pathology associated with the genes can inform breast cancer risk prediction models, and management and treatment of women harboring pathogenic variants,” she noted.
The findings “enhance our understanding of breast cancer biology and aid efforts to classify whether genetic variants of uncertain significance are also likely to be pathogenic,” added Dr. Mavaddat, a senior research associate at the University of Cambridge (England).
She also said the investigators are taking steps to include the results in “CanRisk,” a cancer risk prediction model used by genetic counselors and other clinicians, and are “exploring whether variants in these genes are associated with contralateral breast cancer risk and survival after a breast cancer.”
As most participants were European, analyses should be extended to women of other racial and ethnic groups, she added.
The BRIDGES panel sequencing was supported by the European Union Horizon 2020 research and innovation program BRIDGES and the Wellcome Trust. The Breast Cancer Association Consortium is also funded by the European Union Horizon 2020 research and innovation program, the PERSPECTIVE I&I project, which is funded by the Government of Canada, and the Confluence project, which is funded with intramural funds from the National Cancer Institute. Dr. Mavaddat reported grants from the University of Cambridge, European Union Horizon 2020, Wellcome Trust, Genome Canada, Canadian Institutes of Health Research, and National Cancer Institute during the conduct of the study.
Breast cancer risk genes are generally associated with triple-negative and high-grade disease, but differ substantially in their associated pathology, a large case-control analysis shows.
The findings have potential implications for genetic testing, risk prediction, variant classification, and screening, the authors noted.
To assess links between pathogenic germline variants in nine major breast cancer (BC) susceptibility genes and pathological features of nonmetastasized breast tumors, investigators from the Breast Cancer Association Consortium compared 42,680 women with breast cancer and 42,387 population-matched controls from the BRIDGES large-scale sequencing study.
They looked specifically at features relevant to prognosis and distinct therapeutic options, including tumor subtype, morphology, size, stage, and lymph node involvement, and found substantial differences in tumor subtype distribution by gene.
“Pathogenic [protein-truncating variants and pathogenic missense variants] in these ... they wrote, noting that carriers of rare genetic variants in the nine genes comprised more than 27% of women diagnosed at age 40 or younger with triple-negative disease (driven mainly by BRCA1) and about 16% of those diagnosed with hormone receptor (HR)–positive, HER2-negative high-grade disease.
All together, the nine genes were associated with 14.4% of tumors in women aged 40 years and younger, but less than 4% in women over age 60 years, and among younger women, the prevalence of variants combined was higher in those with triple-negative and HR-positive, HER2-negative disease versus other subtypes, they said.
The findings were published online on Jan. 27, 2022, in JAMA Oncology.
Study subjects were women aged 18-79 years who were sampled, independently of family history, from 38 international population- or hospital-based studies conducted between 1991 and 2016.
The genes assessed included ATM, BARD1, BRCA1, BRCA2, CHEK2, PALB2, RAD51C, RAD51D, and TP53.
Substantial heterogeneity was observed in the distribution of intrinsic subtypes by gene. For example, RAD51C, RAD51D, and BARD1 variants were associated mainly with triple-negative disease (odds ratios, 6.19, 6.19, and 10.05 respectively), and CHEK2 variants were associated with all subtypes (with ORs ranging from 2.21-3.17) except for triple-negative disease, the authors found.
“For ATM variants, the association was strongest for [HR-positive, HER2-negative]high-grade subtype (OR, 4.99). BRCA1 was associated with increased risk of all subtypes, but the ORs varied widely, being highest for triple-negative disease (OR, 55.32),” they wrote.
BRCA2 and PALB2 variants were also associated with triple-negative disease, TP53 variants were most strongly associated with HR-positive, HER2-negative and HR-negative, HER2-positive subtypes, and tumors occurring in pathogenic variant carriers were of higher grade, they added, noting that for most genes and subtypes, a decline in ORs was observed with increasing age.
All genes except CHEK2 were more strongly associated with high-grade disease.
These and other findings from the study can “inform guidelines for eligibility for gene panel sequencing and breast cancer surveillance in the general population,” the authors explained, adding that tumor characteristics can also be used to determine whether variants of uncertain significance are likely to be pathogenic.
The data should therefore “improve the precision of variant classification algorithms and extend them to a larger set of genes,” they noted.
“This case-control study suggests that rare variants in BC susceptibility genes display marked heterogeneity with respect to tumor phenotype, but also similarities between genes that are consistent with known biological functions. This present study provides detailed quantification of subtype-specific breast cancer risks; these can potentially improve risk prediction models and breast cancer prevention strategies,” they concluded.
More specifically, women found to be carrying variants in these genes may be offered enhanced screening, including by MRI, risk-reducing surgery, chemoprevention, and genetic counseling, corresponding author Nasim Mavaddat, MBBS, PhD, explained in an interview.
“Understanding tumor pathology associated with the genes can inform breast cancer risk prediction models, and management and treatment of women harboring pathogenic variants,” she noted.
The findings “enhance our understanding of breast cancer biology and aid efforts to classify whether genetic variants of uncertain significance are also likely to be pathogenic,” added Dr. Mavaddat, a senior research associate at the University of Cambridge (England).
She also said the investigators are taking steps to include the results in “CanRisk,” a cancer risk prediction model used by genetic counselors and other clinicians, and are “exploring whether variants in these genes are associated with contralateral breast cancer risk and survival after a breast cancer.”
As most participants were European, analyses should be extended to women of other racial and ethnic groups, she added.
The BRIDGES panel sequencing was supported by the European Union Horizon 2020 research and innovation program BRIDGES and the Wellcome Trust. The Breast Cancer Association Consortium is also funded by the European Union Horizon 2020 research and innovation program, the PERSPECTIVE I&I project, which is funded by the Government of Canada, and the Confluence project, which is funded with intramural funds from the National Cancer Institute. Dr. Mavaddat reported grants from the University of Cambridge, European Union Horizon 2020, Wellcome Trust, Genome Canada, Canadian Institutes of Health Research, and National Cancer Institute during the conduct of the study.
Breast cancer risk genes are generally associated with triple-negative and high-grade disease, but differ substantially in their associated pathology, a large case-control analysis shows.
The findings have potential implications for genetic testing, risk prediction, variant classification, and screening, the authors noted.
To assess links between pathogenic germline variants in nine major breast cancer (BC) susceptibility genes and pathological features of nonmetastasized breast tumors, investigators from the Breast Cancer Association Consortium compared 42,680 women with breast cancer and 42,387 population-matched controls from the BRIDGES large-scale sequencing study.
They looked specifically at features relevant to prognosis and distinct therapeutic options, including tumor subtype, morphology, size, stage, and lymph node involvement, and found substantial differences in tumor subtype distribution by gene.
“Pathogenic [protein-truncating variants and pathogenic missense variants] in these ... they wrote, noting that carriers of rare genetic variants in the nine genes comprised more than 27% of women diagnosed at age 40 or younger with triple-negative disease (driven mainly by BRCA1) and about 16% of those diagnosed with hormone receptor (HR)–positive, HER2-negative high-grade disease.
All together, the nine genes were associated with 14.4% of tumors in women aged 40 years and younger, but less than 4% in women over age 60 years, and among younger women, the prevalence of variants combined was higher in those with triple-negative and HR-positive, HER2-negative disease versus other subtypes, they said.
The findings were published online on Jan. 27, 2022, in JAMA Oncology.
Study subjects were women aged 18-79 years who were sampled, independently of family history, from 38 international population- or hospital-based studies conducted between 1991 and 2016.
The genes assessed included ATM, BARD1, BRCA1, BRCA2, CHEK2, PALB2, RAD51C, RAD51D, and TP53.
Substantial heterogeneity was observed in the distribution of intrinsic subtypes by gene. For example, RAD51C, RAD51D, and BARD1 variants were associated mainly with triple-negative disease (odds ratios, 6.19, 6.19, and 10.05 respectively), and CHEK2 variants were associated with all subtypes (with ORs ranging from 2.21-3.17) except for triple-negative disease, the authors found.
“For ATM variants, the association was strongest for [HR-positive, HER2-negative]high-grade subtype (OR, 4.99). BRCA1 was associated with increased risk of all subtypes, but the ORs varied widely, being highest for triple-negative disease (OR, 55.32),” they wrote.
BRCA2 and PALB2 variants were also associated with triple-negative disease, TP53 variants were most strongly associated with HR-positive, HER2-negative and HR-negative, HER2-positive subtypes, and tumors occurring in pathogenic variant carriers were of higher grade, they added, noting that for most genes and subtypes, a decline in ORs was observed with increasing age.
All genes except CHEK2 were more strongly associated with high-grade disease.
These and other findings from the study can “inform guidelines for eligibility for gene panel sequencing and breast cancer surveillance in the general population,” the authors explained, adding that tumor characteristics can also be used to determine whether variants of uncertain significance are likely to be pathogenic.
The data should therefore “improve the precision of variant classification algorithms and extend them to a larger set of genes,” they noted.
“This case-control study suggests that rare variants in BC susceptibility genes display marked heterogeneity with respect to tumor phenotype, but also similarities between genes that are consistent with known biological functions. This present study provides detailed quantification of subtype-specific breast cancer risks; these can potentially improve risk prediction models and breast cancer prevention strategies,” they concluded.
More specifically, women found to be carrying variants in these genes may be offered enhanced screening, including by MRI, risk-reducing surgery, chemoprevention, and genetic counseling, corresponding author Nasim Mavaddat, MBBS, PhD, explained in an interview.
“Understanding tumor pathology associated with the genes can inform breast cancer risk prediction models, and management and treatment of women harboring pathogenic variants,” she noted.
The findings “enhance our understanding of breast cancer biology and aid efforts to classify whether genetic variants of uncertain significance are also likely to be pathogenic,” added Dr. Mavaddat, a senior research associate at the University of Cambridge (England).
She also said the investigators are taking steps to include the results in “CanRisk,” a cancer risk prediction model used by genetic counselors and other clinicians, and are “exploring whether variants in these genes are associated with contralateral breast cancer risk and survival after a breast cancer.”
As most participants were European, analyses should be extended to women of other racial and ethnic groups, she added.
The BRIDGES panel sequencing was supported by the European Union Horizon 2020 research and innovation program BRIDGES and the Wellcome Trust. The Breast Cancer Association Consortium is also funded by the European Union Horizon 2020 research and innovation program, the PERSPECTIVE I&I project, which is funded by the Government of Canada, and the Confluence project, which is funded with intramural funds from the National Cancer Institute. Dr. Mavaddat reported grants from the University of Cambridge, European Union Horizon 2020, Wellcome Trust, Genome Canada, Canadian Institutes of Health Research, and National Cancer Institute during the conduct of the study.
FROM JAMA ONCOLOGY
Poor trial representation tied to worse breast cancer survival
Women with early-stage breast cancer who are poorly represented in clinical trials have worse survival than their well-represented peers, according to a real-world analysis.
The study shows that more than half of women with early breast cancer are not well represented in clinical trials because of age, comorbidities, or race, yet they receive therapies based on the results of these trials.
“The most interesting finding is that patients with comorbidities resulting in lab abnormalities that would typically exclude them from receiving medication on a trial are still frequently receiving these medications and have an almost threefold higher mortality,” Gabrielle Rocque, MD, with the division of hematology and oncology, University of Alabama at Birmingham, told this news organization.
“We need to do a deeper dive to better understand what is driving this mortality difference and test specific medications in patients with these conditions to understand the optimal treatment for this population,” Dr. Rocque added.
The study was published Feb. 1 in JCO Oncology Practice.
Many patient groups are not well represented in clinical trials, including patients of color, older patients, and those with comorbidities, and it remains unclear how treatment outcomes may differ among these patients, compared with those who are well represented in trials.
To investigate, Dr. Rocque and colleagues looked at 11,770 women diagnosed with stage I-III breast cancer between 2005 and 2015 in the American Society of Clinical Oncology CancerLinQ database.
White women between 45 and 69 years of age with no comorbid conditions were considered well represented and made up 48% of the cohort.
Non-White women and/or those younger than 45 years or older than 70 were considered under represented and made up 45% of the cohort. The unrepresented group (7%) included women with comorbidities – such as liver disease, renal insufficiency, thrombocytopenia, anemia, or uncontrolled diabetes – or concurrent cancer.
The majority of the women received a high-intensity chemotherapy regimen, including 58% of unrepresented, 66% of underrepresented, and 63% of well-represented patients.
Compared with well-represented women, unrepresented women had a higher risk of death at 5 years (adjusted hazard ratio, 2.71; 95% confidence interval, 2.08-3.52).
Overall, the team found no significant increase in the risk of death at 5 years in underrepresented vs. well-represented women (aHR, 1.19; 95% CI, 0.98-1.45). However, that risk varied with age. Among underrepresented women, those aged 70 and older had more than a twofold higher risk of 5-year mortality (aHR, 2.21), while those younger than 45 had a lower risk of 5-year mortality (aHR, 0.63), compared with those aged 45-69 years.
For three cancer subtypes, unrepresented patients had a greater than twofold higher risk of 5-year mortality, compared with well-represented patients (aHR, 2.50 for HER2-positive disease; aHR, 2.54 for HR-positive/HER2-negative disease; and aHR, 2.75 for triple-negative disease).
Underrepresented patients with HR-positive/HER2-negative disease had a 38% increased risk of 5-year mortality, compared with their well-represented peers (aHR, 1.38). However, there were no significant differences in 5-year mortality for underrepresented vs. well-represented patients with HER2-positive or triple-negative subtypes.
Risky business?
This analysis shows that unrepresented populations receive common treatment regimens at a similar rate as well-represented patients, the researchers note.
“By excluding patients with differing clinical conditions from trials but including them in the population to which drugs can be disseminated, one runs the risk of inadvertently causing injury,” the authors caution.
“To inform the practice of evidence-based medicine in an equitable manner, our findings support a need to both expand clinical trial inclusion criteria and report on clinical trial outcomes by clinical and demographic characteristics,” Dr. Rocque and colleagues conclude.
Charles Shapiro, MD, professor of medicine, hematology, and medical oncology, Icahn School of Medicine at Mount Sinai, New York, is not surprised by the findings of this study.
“We know that clinical trials are too restrictive and include only a selected population largely without comorbidities, but in the real world, people have comorbidities,” Dr. Shapiro, who was not involved in the research, told this news organization.
The study “starkly illustrates” the poorer survival of populations not represented in clinical trials.
“It could be that we need to change clinical trials, maybe ask fewer questions or maybe ask more important questions and loosen the eligibility up, because in the real world, there are people with comorbidities and people who are over 70,” Dr. Shapiro stated.
Are strides being made to change that? “Not really,” Dr. Shapiro said in an interview.
The study was supported by grants from the Robert Wood Johnson Foundation and the American Cancer Society. Dr. Rocque has served as a consultant or advisor for Pfizer; has received research funding from Carevive Systems, Genentech, and Pfizer; and has received travel, accommodations, and expenses from Carevive. Dr. Shapiro has financial relationships with UptoDate, 2nd MD, and Anthenum.
A version of this article first appeared on Medscape.com.
Women with early-stage breast cancer who are poorly represented in clinical trials have worse survival than their well-represented peers, according to a real-world analysis.
The study shows that more than half of women with early breast cancer are not well represented in clinical trials because of age, comorbidities, or race, yet they receive therapies based on the results of these trials.
“The most interesting finding is that patients with comorbidities resulting in lab abnormalities that would typically exclude them from receiving medication on a trial are still frequently receiving these medications and have an almost threefold higher mortality,” Gabrielle Rocque, MD, with the division of hematology and oncology, University of Alabama at Birmingham, told this news organization.
“We need to do a deeper dive to better understand what is driving this mortality difference and test specific medications in patients with these conditions to understand the optimal treatment for this population,” Dr. Rocque added.
The study was published Feb. 1 in JCO Oncology Practice.
Many patient groups are not well represented in clinical trials, including patients of color, older patients, and those with comorbidities, and it remains unclear how treatment outcomes may differ among these patients, compared with those who are well represented in trials.
To investigate, Dr. Rocque and colleagues looked at 11,770 women diagnosed with stage I-III breast cancer between 2005 and 2015 in the American Society of Clinical Oncology CancerLinQ database.
White women between 45 and 69 years of age with no comorbid conditions were considered well represented and made up 48% of the cohort.
Non-White women and/or those younger than 45 years or older than 70 were considered under represented and made up 45% of the cohort. The unrepresented group (7%) included women with comorbidities – such as liver disease, renal insufficiency, thrombocytopenia, anemia, or uncontrolled diabetes – or concurrent cancer.
The majority of the women received a high-intensity chemotherapy regimen, including 58% of unrepresented, 66% of underrepresented, and 63% of well-represented patients.
Compared with well-represented women, unrepresented women had a higher risk of death at 5 years (adjusted hazard ratio, 2.71; 95% confidence interval, 2.08-3.52).
Overall, the team found no significant increase in the risk of death at 5 years in underrepresented vs. well-represented women (aHR, 1.19; 95% CI, 0.98-1.45). However, that risk varied with age. Among underrepresented women, those aged 70 and older had more than a twofold higher risk of 5-year mortality (aHR, 2.21), while those younger than 45 had a lower risk of 5-year mortality (aHR, 0.63), compared with those aged 45-69 years.
For three cancer subtypes, unrepresented patients had a greater than twofold higher risk of 5-year mortality, compared with well-represented patients (aHR, 2.50 for HER2-positive disease; aHR, 2.54 for HR-positive/HER2-negative disease; and aHR, 2.75 for triple-negative disease).
Underrepresented patients with HR-positive/HER2-negative disease had a 38% increased risk of 5-year mortality, compared with their well-represented peers (aHR, 1.38). However, there were no significant differences in 5-year mortality for underrepresented vs. well-represented patients with HER2-positive or triple-negative subtypes.
Risky business?
This analysis shows that unrepresented populations receive common treatment regimens at a similar rate as well-represented patients, the researchers note.
“By excluding patients with differing clinical conditions from trials but including them in the population to which drugs can be disseminated, one runs the risk of inadvertently causing injury,” the authors caution.
“To inform the practice of evidence-based medicine in an equitable manner, our findings support a need to both expand clinical trial inclusion criteria and report on clinical trial outcomes by clinical and demographic characteristics,” Dr. Rocque and colleagues conclude.
Charles Shapiro, MD, professor of medicine, hematology, and medical oncology, Icahn School of Medicine at Mount Sinai, New York, is not surprised by the findings of this study.
“We know that clinical trials are too restrictive and include only a selected population largely without comorbidities, but in the real world, people have comorbidities,” Dr. Shapiro, who was not involved in the research, told this news organization.
The study “starkly illustrates” the poorer survival of populations not represented in clinical trials.
“It could be that we need to change clinical trials, maybe ask fewer questions or maybe ask more important questions and loosen the eligibility up, because in the real world, there are people with comorbidities and people who are over 70,” Dr. Shapiro stated.
Are strides being made to change that? “Not really,” Dr. Shapiro said in an interview.
The study was supported by grants from the Robert Wood Johnson Foundation and the American Cancer Society. Dr. Rocque has served as a consultant or advisor for Pfizer; has received research funding from Carevive Systems, Genentech, and Pfizer; and has received travel, accommodations, and expenses from Carevive. Dr. Shapiro has financial relationships with UptoDate, 2nd MD, and Anthenum.
A version of this article first appeared on Medscape.com.
Women with early-stage breast cancer who are poorly represented in clinical trials have worse survival than their well-represented peers, according to a real-world analysis.
The study shows that more than half of women with early breast cancer are not well represented in clinical trials because of age, comorbidities, or race, yet they receive therapies based on the results of these trials.
“The most interesting finding is that patients with comorbidities resulting in lab abnormalities that would typically exclude them from receiving medication on a trial are still frequently receiving these medications and have an almost threefold higher mortality,” Gabrielle Rocque, MD, with the division of hematology and oncology, University of Alabama at Birmingham, told this news organization.
“We need to do a deeper dive to better understand what is driving this mortality difference and test specific medications in patients with these conditions to understand the optimal treatment for this population,” Dr. Rocque added.
The study was published Feb. 1 in JCO Oncology Practice.
Many patient groups are not well represented in clinical trials, including patients of color, older patients, and those with comorbidities, and it remains unclear how treatment outcomes may differ among these patients, compared with those who are well represented in trials.
To investigate, Dr. Rocque and colleagues looked at 11,770 women diagnosed with stage I-III breast cancer between 2005 and 2015 in the American Society of Clinical Oncology CancerLinQ database.
White women between 45 and 69 years of age with no comorbid conditions were considered well represented and made up 48% of the cohort.
Non-White women and/or those younger than 45 years or older than 70 were considered under represented and made up 45% of the cohort. The unrepresented group (7%) included women with comorbidities – such as liver disease, renal insufficiency, thrombocytopenia, anemia, or uncontrolled diabetes – or concurrent cancer.
The majority of the women received a high-intensity chemotherapy regimen, including 58% of unrepresented, 66% of underrepresented, and 63% of well-represented patients.
Compared with well-represented women, unrepresented women had a higher risk of death at 5 years (adjusted hazard ratio, 2.71; 95% confidence interval, 2.08-3.52).
Overall, the team found no significant increase in the risk of death at 5 years in underrepresented vs. well-represented women (aHR, 1.19; 95% CI, 0.98-1.45). However, that risk varied with age. Among underrepresented women, those aged 70 and older had more than a twofold higher risk of 5-year mortality (aHR, 2.21), while those younger than 45 had a lower risk of 5-year mortality (aHR, 0.63), compared with those aged 45-69 years.
For three cancer subtypes, unrepresented patients had a greater than twofold higher risk of 5-year mortality, compared with well-represented patients (aHR, 2.50 for HER2-positive disease; aHR, 2.54 for HR-positive/HER2-negative disease; and aHR, 2.75 for triple-negative disease).
Underrepresented patients with HR-positive/HER2-negative disease had a 38% increased risk of 5-year mortality, compared with their well-represented peers (aHR, 1.38). However, there were no significant differences in 5-year mortality for underrepresented vs. well-represented patients with HER2-positive or triple-negative subtypes.
Risky business?
This analysis shows that unrepresented populations receive common treatment regimens at a similar rate as well-represented patients, the researchers note.
“By excluding patients with differing clinical conditions from trials but including them in the population to which drugs can be disseminated, one runs the risk of inadvertently causing injury,” the authors caution.
“To inform the practice of evidence-based medicine in an equitable manner, our findings support a need to both expand clinical trial inclusion criteria and report on clinical trial outcomes by clinical and demographic characteristics,” Dr. Rocque and colleagues conclude.
Charles Shapiro, MD, professor of medicine, hematology, and medical oncology, Icahn School of Medicine at Mount Sinai, New York, is not surprised by the findings of this study.
“We know that clinical trials are too restrictive and include only a selected population largely without comorbidities, but in the real world, people have comorbidities,” Dr. Shapiro, who was not involved in the research, told this news organization.
The study “starkly illustrates” the poorer survival of populations not represented in clinical trials.
“It could be that we need to change clinical trials, maybe ask fewer questions or maybe ask more important questions and loosen the eligibility up, because in the real world, there are people with comorbidities and people who are over 70,” Dr. Shapiro stated.
Are strides being made to change that? “Not really,” Dr. Shapiro said in an interview.
The study was supported by grants from the Robert Wood Johnson Foundation and the American Cancer Society. Dr. Rocque has served as a consultant or advisor for Pfizer; has received research funding from Carevive Systems, Genentech, and Pfizer; and has received travel, accommodations, and expenses from Carevive. Dr. Shapiro has financial relationships with UptoDate, 2nd MD, and Anthenum.
A version of this article first appeared on Medscape.com.
FROM JCO ONCOLOGY PRACTICE
Group prenatal care had mixed effect on preterm birth disparities
Group prenatal care did not reduce preterm birth among low-income patients compared to patients receiving individual prenatal care, but the different care models did shrink racial disparities in low-birth-weight and preterm births, according to a study presented Feb. 3 at the annual meeting sponsored by the Society for Maternal-Fetal Medicine. Patients who attended more group prenatal care visits also had better outcomes, reported Amy Crockett, MD, professor of ob.gyn. and director of the Maternal-Fetal Medicine Fellowship Program at the University of South Carolina in Greenville.
Though the results did not show group prenatal care to be the “silver bullet” they might have hoped for in reducing preterm birth and low birth weight among Black women, the study did show group prenatal care to have some clinical effect, Dr. Crockett suggested.
Ilina Pluym, MD, assistant professor of maternal-fetal medicine at the University of California Los Angeles, said that preterm birth and low birth weight are complex problems that cannot be solved by a single fix.
“In terms of the racial disparities, it is difficult to prove that 9 months of good access to prenatal care will undo the years of limited health care and life stressors that, as a whole, possibly contribute more to the overall risk profile of the patient,” Dr. Pluym said. “But as providers, we want to intervene and help in the portion that we can, and optimizing prenatal care is one tangible thing we can try.”
The racial disparities that exist in preterm birth, low birth weight, and infant and maternal mortality are not caused by biologic or genetic factors, Dr. Crockett told attendees.
”Rather, they are the result of long-standing systemic racism and discrimination deeply embedded in the culture of the United States,” she said. “To achieve racial equity, we need reform at the societal level.” But she noted that it might be possible for individual practices to play a role as well, which led her to design a study comparing outcomes from group versus individual prenatal care.
The group care model used for the study is called Centering Pregnancy, in which 8-10 pregnant patients who are due in the same month attend 10 two-hour prenatal care sessions together. Dr. Crockett noted that other research has identified potential reductions in preterm birth with group care models, but some are underpowered while others are observational and limited by confounding, selection bias, or small sample sizes.
The researchers aimed to recruit 3,160 patients to ensure an adequately powered study after estimated losses to follow-up, but they were able to recruit only 2,350 patients, resulting in potentially underpowered results. All the patients were receiving care at a single obstetric practice for a medically low-risk singleton pregnancy of 20 or fewer weeks gestation. A total of 1,176 patients were randomly assigned to attend group prenatal care appointments, and 1,174 patients were assigned to individual prenatal care.
The patients in both groups were an average age of 25 with a prepregnancy body mass index of 29. A similar proportion in both the group and individual care were married (24%) patients and had a government payer (96%-97%). Approximately 41%were Black patients, 37% were White patients, and 21% were Hispanic patients in both groups. Rates of chronic hypertension, smoking, vaginal infection in pregnancy, parity, cervix length, and use of cerclage were similar in both groups.
Gestational age outcomes were available for 1,099 group care patients (94%) and 1,120 individual care patients (95%), and birth weight outcomes were available for 1,028 group care patients (87%) and 1,044 individual care patients (89%).
In the individual care group, 8.7% of the patients had a preterm birth, compared to 10.4% who attended group prenatal care, but the findings did not reach significance (odds ratio [OR], 1.22; 95% confidence interval [CI]: 0.92-1.63). The difference between the 9.6% of group care patients and 8.9% of individual care patients who had babies with low birth weights were not significantly different.
While no significant difference in preterm birth occurred between the groups, the secondary outcome looking at racial disparities yielded one statistically significant result. Black women receiving individual prenatal care were twice as likely as were White women to have an infant with a low birth weight (OR, 2.1; 95% CI: 1.29-3.5). Among those receiving group care, however, the 12.5% of Black women and 8.9% of White women whose infants had a low birth weight were not significantly different.
There was a trend toward narrower disparities in preterm birth in the group versus individual care groups. Among those receiving group care, 11.4% of Black women and 10.8% of White women had a preterm birth, compared to 10.2% of Black women and 7.7% of White women in the individual care group.
Then the researchers compared the groups with regard to compliance while adjusting for baseline differences. “We saw decreasing rates of preterm birth for Black patients relative to White patients, particularly after attending five or more sessions in the group care arm with the rates of preterm birth narrowing and the disparity becoming nonsignificant with more exposure to group care,” Dr. Crockett said. “In individual care, the rate of preterm birth remained the highest for Black women regardless of the number of visits attended.”
The idea of trying group prenatal care is appealing, Dr. Pluym said in an interview, though both models have their advantages.
”The strength of individual care is the focus on the patient exam and individual patient questions,” Dr. Pluym said. “The strengths of group prenatal care are the consolidation of the patient education aspect of prenatal care that is uniform for all patients, the sense of community patients feel, and the opportunity to hear other patients questions that you may not have thought of. It sounds like, anecdotally, patients and providers really found the group sessions valuable and they helped dampen implicit bias and build relationships.”
One potential limitation of this randomized controlled trial is that the providers were the same for both group and individual care, potentially causing some confounding, Dr. Pluym noted. But the study does have one clear clinical message, she said.
“My take away is more prenatal care is better – individual or group,” Dr. Pluym said. “Every clinic should evaluate their own structure and see if group care would be feasible for their patient population. There may be a benefit globally, but is it is not the ‘silver bullet,’ as they said, for lowering preterm birth or growth restriction.”
The research was funded by the Institute of Child Health and Development. The study researchers and Dr. Pluym reported no disclosures.
Group prenatal care did not reduce preterm birth among low-income patients compared to patients receiving individual prenatal care, but the different care models did shrink racial disparities in low-birth-weight and preterm births, according to a study presented Feb. 3 at the annual meeting sponsored by the Society for Maternal-Fetal Medicine. Patients who attended more group prenatal care visits also had better outcomes, reported Amy Crockett, MD, professor of ob.gyn. and director of the Maternal-Fetal Medicine Fellowship Program at the University of South Carolina in Greenville.
Though the results did not show group prenatal care to be the “silver bullet” they might have hoped for in reducing preterm birth and low birth weight among Black women, the study did show group prenatal care to have some clinical effect, Dr. Crockett suggested.
Ilina Pluym, MD, assistant professor of maternal-fetal medicine at the University of California Los Angeles, said that preterm birth and low birth weight are complex problems that cannot be solved by a single fix.
“In terms of the racial disparities, it is difficult to prove that 9 months of good access to prenatal care will undo the years of limited health care and life stressors that, as a whole, possibly contribute more to the overall risk profile of the patient,” Dr. Pluym said. “But as providers, we want to intervene and help in the portion that we can, and optimizing prenatal care is one tangible thing we can try.”
The racial disparities that exist in preterm birth, low birth weight, and infant and maternal mortality are not caused by biologic or genetic factors, Dr. Crockett told attendees.
”Rather, they are the result of long-standing systemic racism and discrimination deeply embedded in the culture of the United States,” she said. “To achieve racial equity, we need reform at the societal level.” But she noted that it might be possible for individual practices to play a role as well, which led her to design a study comparing outcomes from group versus individual prenatal care.
The group care model used for the study is called Centering Pregnancy, in which 8-10 pregnant patients who are due in the same month attend 10 two-hour prenatal care sessions together. Dr. Crockett noted that other research has identified potential reductions in preterm birth with group care models, but some are underpowered while others are observational and limited by confounding, selection bias, or small sample sizes.
The researchers aimed to recruit 3,160 patients to ensure an adequately powered study after estimated losses to follow-up, but they were able to recruit only 2,350 patients, resulting in potentially underpowered results. All the patients were receiving care at a single obstetric practice for a medically low-risk singleton pregnancy of 20 or fewer weeks gestation. A total of 1,176 patients were randomly assigned to attend group prenatal care appointments, and 1,174 patients were assigned to individual prenatal care.
The patients in both groups were an average age of 25 with a prepregnancy body mass index of 29. A similar proportion in both the group and individual care were married (24%) patients and had a government payer (96%-97%). Approximately 41%were Black patients, 37% were White patients, and 21% were Hispanic patients in both groups. Rates of chronic hypertension, smoking, vaginal infection in pregnancy, parity, cervix length, and use of cerclage were similar in both groups.
Gestational age outcomes were available for 1,099 group care patients (94%) and 1,120 individual care patients (95%), and birth weight outcomes were available for 1,028 group care patients (87%) and 1,044 individual care patients (89%).
In the individual care group, 8.7% of the patients had a preterm birth, compared to 10.4% who attended group prenatal care, but the findings did not reach significance (odds ratio [OR], 1.22; 95% confidence interval [CI]: 0.92-1.63). The difference between the 9.6% of group care patients and 8.9% of individual care patients who had babies with low birth weights were not significantly different.
While no significant difference in preterm birth occurred between the groups, the secondary outcome looking at racial disparities yielded one statistically significant result. Black women receiving individual prenatal care were twice as likely as were White women to have an infant with a low birth weight (OR, 2.1; 95% CI: 1.29-3.5). Among those receiving group care, however, the 12.5% of Black women and 8.9% of White women whose infants had a low birth weight were not significantly different.
There was a trend toward narrower disparities in preterm birth in the group versus individual care groups. Among those receiving group care, 11.4% of Black women and 10.8% of White women had a preterm birth, compared to 10.2% of Black women and 7.7% of White women in the individual care group.
Then the researchers compared the groups with regard to compliance while adjusting for baseline differences. “We saw decreasing rates of preterm birth for Black patients relative to White patients, particularly after attending five or more sessions in the group care arm with the rates of preterm birth narrowing and the disparity becoming nonsignificant with more exposure to group care,” Dr. Crockett said. “In individual care, the rate of preterm birth remained the highest for Black women regardless of the number of visits attended.”
The idea of trying group prenatal care is appealing, Dr. Pluym said in an interview, though both models have their advantages.
”The strength of individual care is the focus on the patient exam and individual patient questions,” Dr. Pluym said. “The strengths of group prenatal care are the consolidation of the patient education aspect of prenatal care that is uniform for all patients, the sense of community patients feel, and the opportunity to hear other patients questions that you may not have thought of. It sounds like, anecdotally, patients and providers really found the group sessions valuable and they helped dampen implicit bias and build relationships.”
One potential limitation of this randomized controlled trial is that the providers were the same for both group and individual care, potentially causing some confounding, Dr. Pluym noted. But the study does have one clear clinical message, she said.
“My take away is more prenatal care is better – individual or group,” Dr. Pluym said. “Every clinic should evaluate their own structure and see if group care would be feasible for their patient population. There may be a benefit globally, but is it is not the ‘silver bullet,’ as they said, for lowering preterm birth or growth restriction.”
The research was funded by the Institute of Child Health and Development. The study researchers and Dr. Pluym reported no disclosures.
Group prenatal care did not reduce preterm birth among low-income patients compared to patients receiving individual prenatal care, but the different care models did shrink racial disparities in low-birth-weight and preterm births, according to a study presented Feb. 3 at the annual meeting sponsored by the Society for Maternal-Fetal Medicine. Patients who attended more group prenatal care visits also had better outcomes, reported Amy Crockett, MD, professor of ob.gyn. and director of the Maternal-Fetal Medicine Fellowship Program at the University of South Carolina in Greenville.
Though the results did not show group prenatal care to be the “silver bullet” they might have hoped for in reducing preterm birth and low birth weight among Black women, the study did show group prenatal care to have some clinical effect, Dr. Crockett suggested.
Ilina Pluym, MD, assistant professor of maternal-fetal medicine at the University of California Los Angeles, said that preterm birth and low birth weight are complex problems that cannot be solved by a single fix.
“In terms of the racial disparities, it is difficult to prove that 9 months of good access to prenatal care will undo the years of limited health care and life stressors that, as a whole, possibly contribute more to the overall risk profile of the patient,” Dr. Pluym said. “But as providers, we want to intervene and help in the portion that we can, and optimizing prenatal care is one tangible thing we can try.”
The racial disparities that exist in preterm birth, low birth weight, and infant and maternal mortality are not caused by biologic or genetic factors, Dr. Crockett told attendees.
”Rather, they are the result of long-standing systemic racism and discrimination deeply embedded in the culture of the United States,” she said. “To achieve racial equity, we need reform at the societal level.” But she noted that it might be possible for individual practices to play a role as well, which led her to design a study comparing outcomes from group versus individual prenatal care.
The group care model used for the study is called Centering Pregnancy, in which 8-10 pregnant patients who are due in the same month attend 10 two-hour prenatal care sessions together. Dr. Crockett noted that other research has identified potential reductions in preterm birth with group care models, but some are underpowered while others are observational and limited by confounding, selection bias, or small sample sizes.
The researchers aimed to recruit 3,160 patients to ensure an adequately powered study after estimated losses to follow-up, but they were able to recruit only 2,350 patients, resulting in potentially underpowered results. All the patients were receiving care at a single obstetric practice for a medically low-risk singleton pregnancy of 20 or fewer weeks gestation. A total of 1,176 patients were randomly assigned to attend group prenatal care appointments, and 1,174 patients were assigned to individual prenatal care.
The patients in both groups were an average age of 25 with a prepregnancy body mass index of 29. A similar proportion in both the group and individual care were married (24%) patients and had a government payer (96%-97%). Approximately 41%were Black patients, 37% were White patients, and 21% were Hispanic patients in both groups. Rates of chronic hypertension, smoking, vaginal infection in pregnancy, parity, cervix length, and use of cerclage were similar in both groups.
Gestational age outcomes were available for 1,099 group care patients (94%) and 1,120 individual care patients (95%), and birth weight outcomes were available for 1,028 group care patients (87%) and 1,044 individual care patients (89%).
In the individual care group, 8.7% of the patients had a preterm birth, compared to 10.4% who attended group prenatal care, but the findings did not reach significance (odds ratio [OR], 1.22; 95% confidence interval [CI]: 0.92-1.63). The difference between the 9.6% of group care patients and 8.9% of individual care patients who had babies with low birth weights were not significantly different.
While no significant difference in preterm birth occurred between the groups, the secondary outcome looking at racial disparities yielded one statistically significant result. Black women receiving individual prenatal care were twice as likely as were White women to have an infant with a low birth weight (OR, 2.1; 95% CI: 1.29-3.5). Among those receiving group care, however, the 12.5% of Black women and 8.9% of White women whose infants had a low birth weight were not significantly different.
There was a trend toward narrower disparities in preterm birth in the group versus individual care groups. Among those receiving group care, 11.4% of Black women and 10.8% of White women had a preterm birth, compared to 10.2% of Black women and 7.7% of White women in the individual care group.
Then the researchers compared the groups with regard to compliance while adjusting for baseline differences. “We saw decreasing rates of preterm birth for Black patients relative to White patients, particularly after attending five or more sessions in the group care arm with the rates of preterm birth narrowing and the disparity becoming nonsignificant with more exposure to group care,” Dr. Crockett said. “In individual care, the rate of preterm birth remained the highest for Black women regardless of the number of visits attended.”
The idea of trying group prenatal care is appealing, Dr. Pluym said in an interview, though both models have their advantages.
”The strength of individual care is the focus on the patient exam and individual patient questions,” Dr. Pluym said. “The strengths of group prenatal care are the consolidation of the patient education aspect of prenatal care that is uniform for all patients, the sense of community patients feel, and the opportunity to hear other patients questions that you may not have thought of. It sounds like, anecdotally, patients and providers really found the group sessions valuable and they helped dampen implicit bias and build relationships.”
One potential limitation of this randomized controlled trial is that the providers were the same for both group and individual care, potentially causing some confounding, Dr. Pluym noted. But the study does have one clear clinical message, she said.
“My take away is more prenatal care is better – individual or group,” Dr. Pluym said. “Every clinic should evaluate their own structure and see if group care would be feasible for their patient population. There may be a benefit globally, but is it is not the ‘silver bullet,’ as they said, for lowering preterm birth or growth restriction.”
The research was funded by the Institute of Child Health and Development. The study researchers and Dr. Pluym reported no disclosures.
FROM THE PREGNANCY MEETING
Breastfeeding disparities further exacerbated by pending legislation
From the American Medical Association to the Centers for Disease Control and Prevention, health equity is the topic de jour. But how do you get health professionals, lawmakers, lactation providers, and the community on the same page, especially when it comes to addressing breastfeeding disparities?
It depends on who you ask.
In Georgia, a 2018 lawsuit challenging a State Legislature Bill directed toward lactation providers sits on the desk of a trial court judge, with a decision due any day now. The bill requires these providers to be licensed in order to continue to practice and receive compensation, a move that not only threatens the health of mothers and infants, but also jeopardizes a key component of Healthy People 2030: improving breastfeeding initiation, duration, and exclusivity among African American women. A similar bill is in Committee in the New York State Legislature.
“If the Act takes effect, it will force an estimated 800 different practitioners out of business and leave only 162 International Board Certified Lactation Counselors (IBCLCs) for the whole state,” Jaimie Cavanaugh, an attorney at the Institute for Justice and plaintiff coattorney said in an interview.
Ms. Cavanaugh also said that geographical data for the 162 IBCLCs demonstrate that they primarily work in urban vs. rural areas, and mostly in formal settings, factors that will further exacerbate disparities and limit access to much needed resources.
Bridging the breastfeeding divide
While overall breastfeeding initiation rates in the United States have steadily increased over the past decade from 72% to roughly 84%, only a quarter of infants are exclusively breastfed through 6 months, a rate well below the Healthy People 2030 goal of 42.4% (and American Academy of Pediatrics recommendations). Comparatively, breastfeeding initiation (75.8%) and exclusivity (17.2%) rates among African-American women are considerably lower.
The effects are great: Breastfed infants have lower risks for asthma, obesity, and type 1 diabetes, while mothers who breastfeed have lower risks for hypertension, type 2 diabetes, and gynecological cancers. Notably, most of these conditions disproportionately affect African Americans, compared with Whites and other ethnicities.
A key to changing these disparities appears to lie with the type of health care provided as well as the ease by which mothers can access it.
For example, findings of a small cross-sectional study published Jan. 31 in the Journal of Racial and Ethnic Health Disparities highlight the importance of a broad umbrella of support for African American mothers’ feeding choices. Not only does this umbrella include medical professionals and IBCLCs, but also certified lactation counselors (CLCs), peer counselors trained under the National Special Supplemental Nutrition Program for Women, Infants, and Children, partners, family, and the community at-large.
“We thought we were doing it right,” Lydia Furman, MD, lead study author and pediatric specialist at University Hospitals Rainbow Babies & Children’s Ahuja Center for Women & Children in Cleveland, told this news organization. “We have a WIC peer helper, an African American IBCLC and an African American CLC, and a breastfeeding support group twice a week but nobody was using these resources.”
One of the most important findings of the study – which aimed to understand factors driving breastfeeding practices and identify supports – was that women want help when they need it. “It doesn’t mean that you can’t have resources that are available during the day, but it means that a patient support group at 11 a.m. on Tuesday doesn’t help at all if you need it Monday at 2 a.m.” Dr. Furman said.
Take TaNeeka Davis, a 34-year-old mother of three residing outside of Atlanta whose personal experience mimics those of the women in Dr. Furman’s study. “I did breastfeed my first child; when I was in the hospital. I saw lactation one time and he latched perfectly but when I left the hospital, I couldn’t get him to latch anymore,” she said.
Ms. Davis explained that she was told that she would have to wait 2-3 weeks before she was able to meet again with a lactation specialist, so she found herself supplementing with formula, and eventually seeking nontraditional help.
“The traditional medical model does not allow for me to be able to reach out and talk to my doctor immediately, does not allow me to be like, ‘Hey, can you call me back in the next 15-20 minutes or an hour because my baby’s very fussy,’ ” Ms. Davis said. “I don’t have that kind of support.”
It takes a village
A 2017 Cochrane review reinforces the value of providing women with predictable, tailored, and multifaceted breastfeeding support offered by professional or lay/peer people or a combination of both.
This model is embodied in ROSE, a nonprofit organization dedicated to eliminating breastfeeding disparities and barriers experienced by mothers of color, including inadequate medical or family support, lack of shared decision-making, recognition of financial or psychological challenges, and historical antecedents. Many of these women’s ancestors were forced to wet-nurse slave masters’ children instead of breastfeeding their own children.
One of several national organizations solely dedicated to this issue, ROSE offers a variety of services and resources ranging from lactation counseling and peer support training programs to training for health professionals (for example, pediatricians, nurses) that serve communities of color. A companion arm (ROBE, Reaching Our Brothers Everywhere) aims to bring men into the fold through breastfeeding education and peer-to-peer connection. All of these services are provided in a judgment-free, culturally sensitive environment.
“We need to look not only into maternal health issues ... but also offer support to people who are working in the birthing community,” cofounder Mary N. Jackson, a CLC, WIC lactation consultant, and former president of the Georgia Breastfeeding Coalition said in an interview.
“We have Morehouse pediatricians coming to us just to talk to moms on how they can support them in the community. We have training – Community Transformers – where we talk to moms regardless of their social backgrounds; they’re working in the community helping other moms with breastfeeding, or moms will call them (with) their questions,” Ms. Jackson explained. Ms. Davis is now one of these women.
“Having the women of ROSE support me ... was such a game-changer,” she said. “Sometimes that support that you need, that is helpful, is peer-to-peer,” she noted, adding that ROSE does a lot more than fill in the gaps medically, but also psychologically.”
More pillars, less judgment
TaNeeka Davis pointed out that removing a pillar in the community like ROSE and other grassroots support outside of traditional models will likely have the opposite effect that lawmakers and the lobbyists fighting for certification and licensing aim to achieve, especially if other states adopt the same approach.
“The disparities are going to get even greater, you are going to see bigger gaps, less women even initiating breastfeeding. Why start something that you can’t finish? You can’t tell me that making laws that limit the amount of help we are able to get when it comes to breastfeeding will not have a detrimental effect – health effects – later in life,” she said.
Neither Ms. Jackson nor Ms. Davis believe that medical professionals should be replaced but rather that adjunctive, community-based help is integral for bridging the breastfeeding divide.
As clinicians, “we have to go beyond not judging to trying to figure out where people are, to meet your patients where they are,” said Dr. Furman. “It’s like the difference between cultural competence and cultural humility, which is more of an ongoing process.
Dr. Furman and Ms. Davis report no relevant financial relationships. Ms. Cavanaugh is the coattorney on the lawsuit. Ms. Jackson is employed by ROSE.
*This story was updated on Feb. 11, 2022.
From the American Medical Association to the Centers for Disease Control and Prevention, health equity is the topic de jour. But how do you get health professionals, lawmakers, lactation providers, and the community on the same page, especially when it comes to addressing breastfeeding disparities?
It depends on who you ask.
In Georgia, a 2018 lawsuit challenging a State Legislature Bill directed toward lactation providers sits on the desk of a trial court judge, with a decision due any day now. The bill requires these providers to be licensed in order to continue to practice and receive compensation, a move that not only threatens the health of mothers and infants, but also jeopardizes a key component of Healthy People 2030: improving breastfeeding initiation, duration, and exclusivity among African American women. A similar bill is in Committee in the New York State Legislature.
“If the Act takes effect, it will force an estimated 800 different practitioners out of business and leave only 162 International Board Certified Lactation Counselors (IBCLCs) for the whole state,” Jaimie Cavanaugh, an attorney at the Institute for Justice and plaintiff coattorney said in an interview.
Ms. Cavanaugh also said that geographical data for the 162 IBCLCs demonstrate that they primarily work in urban vs. rural areas, and mostly in formal settings, factors that will further exacerbate disparities and limit access to much needed resources.
Bridging the breastfeeding divide
While overall breastfeeding initiation rates in the United States have steadily increased over the past decade from 72% to roughly 84%, only a quarter of infants are exclusively breastfed through 6 months, a rate well below the Healthy People 2030 goal of 42.4% (and American Academy of Pediatrics recommendations). Comparatively, breastfeeding initiation (75.8%) and exclusivity (17.2%) rates among African-American women are considerably lower.
The effects are great: Breastfed infants have lower risks for asthma, obesity, and type 1 diabetes, while mothers who breastfeed have lower risks for hypertension, type 2 diabetes, and gynecological cancers. Notably, most of these conditions disproportionately affect African Americans, compared with Whites and other ethnicities.
A key to changing these disparities appears to lie with the type of health care provided as well as the ease by which mothers can access it.
For example, findings of a small cross-sectional study published Jan. 31 in the Journal of Racial and Ethnic Health Disparities highlight the importance of a broad umbrella of support for African American mothers’ feeding choices. Not only does this umbrella include medical professionals and IBCLCs, but also certified lactation counselors (CLCs), peer counselors trained under the National Special Supplemental Nutrition Program for Women, Infants, and Children, partners, family, and the community at-large.
“We thought we were doing it right,” Lydia Furman, MD, lead study author and pediatric specialist at University Hospitals Rainbow Babies & Children’s Ahuja Center for Women & Children in Cleveland, told this news organization. “We have a WIC peer helper, an African American IBCLC and an African American CLC, and a breastfeeding support group twice a week but nobody was using these resources.”
One of the most important findings of the study – which aimed to understand factors driving breastfeeding practices and identify supports – was that women want help when they need it. “It doesn’t mean that you can’t have resources that are available during the day, but it means that a patient support group at 11 a.m. on Tuesday doesn’t help at all if you need it Monday at 2 a.m.” Dr. Furman said.
Take TaNeeka Davis, a 34-year-old mother of three residing outside of Atlanta whose personal experience mimics those of the women in Dr. Furman’s study. “I did breastfeed my first child; when I was in the hospital. I saw lactation one time and he latched perfectly but when I left the hospital, I couldn’t get him to latch anymore,” she said.
Ms. Davis explained that she was told that she would have to wait 2-3 weeks before she was able to meet again with a lactation specialist, so she found herself supplementing with formula, and eventually seeking nontraditional help.
“The traditional medical model does not allow for me to be able to reach out and talk to my doctor immediately, does not allow me to be like, ‘Hey, can you call me back in the next 15-20 minutes or an hour because my baby’s very fussy,’ ” Ms. Davis said. “I don’t have that kind of support.”
It takes a village
A 2017 Cochrane review reinforces the value of providing women with predictable, tailored, and multifaceted breastfeeding support offered by professional or lay/peer people or a combination of both.
This model is embodied in ROSE, a nonprofit organization dedicated to eliminating breastfeeding disparities and barriers experienced by mothers of color, including inadequate medical or family support, lack of shared decision-making, recognition of financial or psychological challenges, and historical antecedents. Many of these women’s ancestors were forced to wet-nurse slave masters’ children instead of breastfeeding their own children.
One of several national organizations solely dedicated to this issue, ROSE offers a variety of services and resources ranging from lactation counseling and peer support training programs to training for health professionals (for example, pediatricians, nurses) that serve communities of color. A companion arm (ROBE, Reaching Our Brothers Everywhere) aims to bring men into the fold through breastfeeding education and peer-to-peer connection. All of these services are provided in a judgment-free, culturally sensitive environment.
“We need to look not only into maternal health issues ... but also offer support to people who are working in the birthing community,” cofounder Mary N. Jackson, a CLC, WIC lactation consultant, and former president of the Georgia Breastfeeding Coalition said in an interview.
“We have Morehouse pediatricians coming to us just to talk to moms on how they can support them in the community. We have training – Community Transformers – where we talk to moms regardless of their social backgrounds; they’re working in the community helping other moms with breastfeeding, or moms will call them (with) their questions,” Ms. Jackson explained. Ms. Davis is now one of these women.
“Having the women of ROSE support me ... was such a game-changer,” she said. “Sometimes that support that you need, that is helpful, is peer-to-peer,” she noted, adding that ROSE does a lot more than fill in the gaps medically, but also psychologically.”
More pillars, less judgment
TaNeeka Davis pointed out that removing a pillar in the community like ROSE and other grassroots support outside of traditional models will likely have the opposite effect that lawmakers and the lobbyists fighting for certification and licensing aim to achieve, especially if other states adopt the same approach.
“The disparities are going to get even greater, you are going to see bigger gaps, less women even initiating breastfeeding. Why start something that you can’t finish? You can’t tell me that making laws that limit the amount of help we are able to get when it comes to breastfeeding will not have a detrimental effect – health effects – later in life,” she said.
Neither Ms. Jackson nor Ms. Davis believe that medical professionals should be replaced but rather that adjunctive, community-based help is integral for bridging the breastfeeding divide.
As clinicians, “we have to go beyond not judging to trying to figure out where people are, to meet your patients where they are,” said Dr. Furman. “It’s like the difference between cultural competence and cultural humility, which is more of an ongoing process.
Dr. Furman and Ms. Davis report no relevant financial relationships. Ms. Cavanaugh is the coattorney on the lawsuit. Ms. Jackson is employed by ROSE.
*This story was updated on Feb. 11, 2022.
From the American Medical Association to the Centers for Disease Control and Prevention, health equity is the topic de jour. But how do you get health professionals, lawmakers, lactation providers, and the community on the same page, especially when it comes to addressing breastfeeding disparities?
It depends on who you ask.
In Georgia, a 2018 lawsuit challenging a State Legislature Bill directed toward lactation providers sits on the desk of a trial court judge, with a decision due any day now. The bill requires these providers to be licensed in order to continue to practice and receive compensation, a move that not only threatens the health of mothers and infants, but also jeopardizes a key component of Healthy People 2030: improving breastfeeding initiation, duration, and exclusivity among African American women. A similar bill is in Committee in the New York State Legislature.
“If the Act takes effect, it will force an estimated 800 different practitioners out of business and leave only 162 International Board Certified Lactation Counselors (IBCLCs) for the whole state,” Jaimie Cavanaugh, an attorney at the Institute for Justice and plaintiff coattorney said in an interview.
Ms. Cavanaugh also said that geographical data for the 162 IBCLCs demonstrate that they primarily work in urban vs. rural areas, and mostly in formal settings, factors that will further exacerbate disparities and limit access to much needed resources.
Bridging the breastfeeding divide
While overall breastfeeding initiation rates in the United States have steadily increased over the past decade from 72% to roughly 84%, only a quarter of infants are exclusively breastfed through 6 months, a rate well below the Healthy People 2030 goal of 42.4% (and American Academy of Pediatrics recommendations). Comparatively, breastfeeding initiation (75.8%) and exclusivity (17.2%) rates among African-American women are considerably lower.
The effects are great: Breastfed infants have lower risks for asthma, obesity, and type 1 diabetes, while mothers who breastfeed have lower risks for hypertension, type 2 diabetes, and gynecological cancers. Notably, most of these conditions disproportionately affect African Americans, compared with Whites and other ethnicities.
A key to changing these disparities appears to lie with the type of health care provided as well as the ease by which mothers can access it.
For example, findings of a small cross-sectional study published Jan. 31 in the Journal of Racial and Ethnic Health Disparities highlight the importance of a broad umbrella of support for African American mothers’ feeding choices. Not only does this umbrella include medical professionals and IBCLCs, but also certified lactation counselors (CLCs), peer counselors trained under the National Special Supplemental Nutrition Program for Women, Infants, and Children, partners, family, and the community at-large.
“We thought we were doing it right,” Lydia Furman, MD, lead study author and pediatric specialist at University Hospitals Rainbow Babies & Children’s Ahuja Center for Women & Children in Cleveland, told this news organization. “We have a WIC peer helper, an African American IBCLC and an African American CLC, and a breastfeeding support group twice a week but nobody was using these resources.”
One of the most important findings of the study – which aimed to understand factors driving breastfeeding practices and identify supports – was that women want help when they need it. “It doesn’t mean that you can’t have resources that are available during the day, but it means that a patient support group at 11 a.m. on Tuesday doesn’t help at all if you need it Monday at 2 a.m.” Dr. Furman said.
Take TaNeeka Davis, a 34-year-old mother of three residing outside of Atlanta whose personal experience mimics those of the women in Dr. Furman’s study. “I did breastfeed my first child; when I was in the hospital. I saw lactation one time and he latched perfectly but when I left the hospital, I couldn’t get him to latch anymore,” she said.
Ms. Davis explained that she was told that she would have to wait 2-3 weeks before she was able to meet again with a lactation specialist, so she found herself supplementing with formula, and eventually seeking nontraditional help.
“The traditional medical model does not allow for me to be able to reach out and talk to my doctor immediately, does not allow me to be like, ‘Hey, can you call me back in the next 15-20 minutes or an hour because my baby’s very fussy,’ ” Ms. Davis said. “I don’t have that kind of support.”
It takes a village
A 2017 Cochrane review reinforces the value of providing women with predictable, tailored, and multifaceted breastfeeding support offered by professional or lay/peer people or a combination of both.
This model is embodied in ROSE, a nonprofit organization dedicated to eliminating breastfeeding disparities and barriers experienced by mothers of color, including inadequate medical or family support, lack of shared decision-making, recognition of financial or psychological challenges, and historical antecedents. Many of these women’s ancestors were forced to wet-nurse slave masters’ children instead of breastfeeding their own children.
One of several national organizations solely dedicated to this issue, ROSE offers a variety of services and resources ranging from lactation counseling and peer support training programs to training for health professionals (for example, pediatricians, nurses) that serve communities of color. A companion arm (ROBE, Reaching Our Brothers Everywhere) aims to bring men into the fold through breastfeeding education and peer-to-peer connection. All of these services are provided in a judgment-free, culturally sensitive environment.
“We need to look not only into maternal health issues ... but also offer support to people who are working in the birthing community,” cofounder Mary N. Jackson, a CLC, WIC lactation consultant, and former president of the Georgia Breastfeeding Coalition said in an interview.
“We have Morehouse pediatricians coming to us just to talk to moms on how they can support them in the community. We have training – Community Transformers – where we talk to moms regardless of their social backgrounds; they’re working in the community helping other moms with breastfeeding, or moms will call them (with) their questions,” Ms. Jackson explained. Ms. Davis is now one of these women.
“Having the women of ROSE support me ... was such a game-changer,” she said. “Sometimes that support that you need, that is helpful, is peer-to-peer,” she noted, adding that ROSE does a lot more than fill in the gaps medically, but also psychologically.”
More pillars, less judgment
TaNeeka Davis pointed out that removing a pillar in the community like ROSE and other grassroots support outside of traditional models will likely have the opposite effect that lawmakers and the lobbyists fighting for certification and licensing aim to achieve, especially if other states adopt the same approach.
“The disparities are going to get even greater, you are going to see bigger gaps, less women even initiating breastfeeding. Why start something that you can’t finish? You can’t tell me that making laws that limit the amount of help we are able to get when it comes to breastfeeding will not have a detrimental effect – health effects – later in life,” she said.
Neither Ms. Jackson nor Ms. Davis believe that medical professionals should be replaced but rather that adjunctive, community-based help is integral for bridging the breastfeeding divide.
As clinicians, “we have to go beyond not judging to trying to figure out where people are, to meet your patients where they are,” said Dr. Furman. “It’s like the difference between cultural competence and cultural humility, which is more of an ongoing process.
Dr. Furman and Ms. Davis report no relevant financial relationships. Ms. Cavanaugh is the coattorney on the lawsuit. Ms. Jackson is employed by ROSE.
*This story was updated on Feb. 11, 2022.









