User login
‘Best Hospitals for Maternity’ offers national perspective
“The Best Hospitals for Maternity” looked at data for 2019 and covered uncomplicated but not high-risk pregnancies. “All families deserve to be informed on how hospitals perform on key indicators of quality, which is why U.S. News has compiled and published a trove of maternal health data from hospitals across the country,” Ben Harder, managing editor and chief of health analysis at U.S. News, said in a written statement.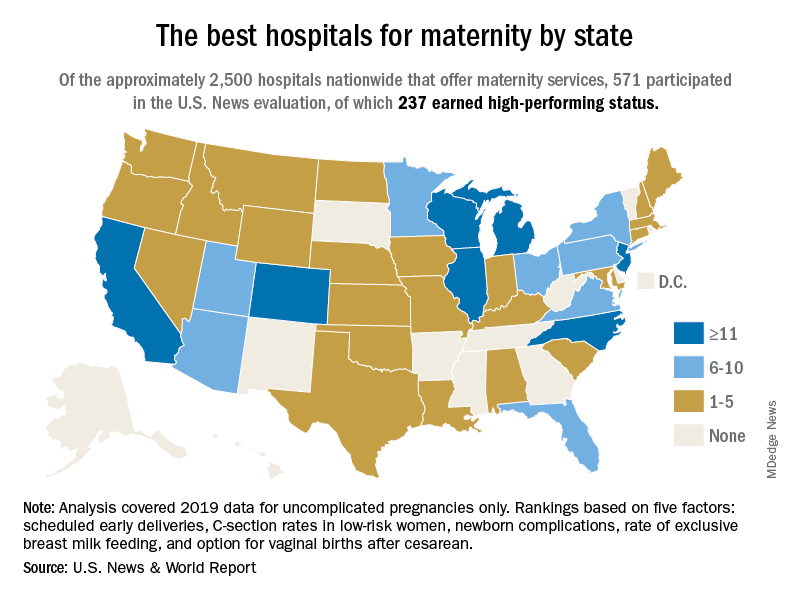
The 237 best performers were selected from an overall pool of 571 hospitals that participated in the analysis, representing every state except Alaska, Arkansas, and Vermont, U.S. News said, noting that about 2,700 hospitals in the United States offer maternity services.
California has the highest number of best performers, 33, followed by Illinois and New Jersey, both with 15. Colorado is home to 14 best-performing hospitals, Michigan has 12, and North Carolina and Wisconsin each have 12. “Hospitals that performed well had fewer newborn complications, fewer early deliveries and fewer C-sections, compared to other hospitals across the nation,” Mr. Harder said.
The composite score constructed by U.S. News involved five quality measures: nulliparous, term, singleton, and vertex cesarean delivery rates; early elective delivery rates; unexpected newborn complications rates; routine vaginal birth after cesarean (VBAC) delivery availability; and exclusive breast milk–feeding rates. The composite score averaged 80.0 for high performers and 54.9 for the other participating hospitals, U.S. News reported.
Averages for the high performers on each of the five measures looked like this:
- C-section rate, 21.1%.
- Early elective delivery rate, 1.3%.
- Overall unexpected newborn complication rates, 2.38%
- Exclusive breast milk–feeding rate, 58.6%.
- VBAC availability, 92.4%.
Data for four measures were collected from the hospitals via online survey over a 4-month window that began on April 29, 2021. Rates of early elective delivery came from the Centers for Medicare & Medicaid Services Care Compare.
“The Best Hospitals for Maternity” looked at data for 2019 and covered uncomplicated but not high-risk pregnancies. “All families deserve to be informed on how hospitals perform on key indicators of quality, which is why U.S. News has compiled and published a trove of maternal health data from hospitals across the country,” Ben Harder, managing editor and chief of health analysis at U.S. News, said in a written statement.
The 237 best performers were selected from an overall pool of 571 hospitals that participated in the analysis, representing every state except Alaska, Arkansas, and Vermont, U.S. News said, noting that about 2,700 hospitals in the United States offer maternity services.
California has the highest number of best performers, 33, followed by Illinois and New Jersey, both with 15. Colorado is home to 14 best-performing hospitals, Michigan has 12, and North Carolina and Wisconsin each have 12. “Hospitals that performed well had fewer newborn complications, fewer early deliveries and fewer C-sections, compared to other hospitals across the nation,” Mr. Harder said.
The composite score constructed by U.S. News involved five quality measures: nulliparous, term, singleton, and vertex cesarean delivery rates; early elective delivery rates; unexpected newborn complications rates; routine vaginal birth after cesarean (VBAC) delivery availability; and exclusive breast milk–feeding rates. The composite score averaged 80.0 for high performers and 54.9 for the other participating hospitals, U.S. News reported.
Averages for the high performers on each of the five measures looked like this:
- C-section rate, 21.1%.
- Early elective delivery rate, 1.3%.
- Overall unexpected newborn complication rates, 2.38%
- Exclusive breast milk–feeding rate, 58.6%.
- VBAC availability, 92.4%.
Data for four measures were collected from the hospitals via online survey over a 4-month window that began on April 29, 2021. Rates of early elective delivery came from the Centers for Medicare & Medicaid Services Care Compare.
“The Best Hospitals for Maternity” looked at data for 2019 and covered uncomplicated but not high-risk pregnancies. “All families deserve to be informed on how hospitals perform on key indicators of quality, which is why U.S. News has compiled and published a trove of maternal health data from hospitals across the country,” Ben Harder, managing editor and chief of health analysis at U.S. News, said in a written statement.
The 237 best performers were selected from an overall pool of 571 hospitals that participated in the analysis, representing every state except Alaska, Arkansas, and Vermont, U.S. News said, noting that about 2,700 hospitals in the United States offer maternity services.
California has the highest number of best performers, 33, followed by Illinois and New Jersey, both with 15. Colorado is home to 14 best-performing hospitals, Michigan has 12, and North Carolina and Wisconsin each have 12. “Hospitals that performed well had fewer newborn complications, fewer early deliveries and fewer C-sections, compared to other hospitals across the nation,” Mr. Harder said.
The composite score constructed by U.S. News involved five quality measures: nulliparous, term, singleton, and vertex cesarean delivery rates; early elective delivery rates; unexpected newborn complications rates; routine vaginal birth after cesarean (VBAC) delivery availability; and exclusive breast milk–feeding rates. The composite score averaged 80.0 for high performers and 54.9 for the other participating hospitals, U.S. News reported.
Averages for the high performers on each of the five measures looked like this:
- C-section rate, 21.1%.
- Early elective delivery rate, 1.3%.
- Overall unexpected newborn complication rates, 2.38%
- Exclusive breast milk–feeding rate, 58.6%.
- VBAC availability, 92.4%.
Data for four measures were collected from the hospitals via online survey over a 4-month window that began on April 29, 2021. Rates of early elective delivery came from the Centers for Medicare & Medicaid Services Care Compare.
Large analysis confirms safety of nipple-sparing mastectomy
A new analysis of over 22,000 mastectomy patients confirms what smaller studies have indicated: Patients who undergo nipple-sparing mastectomy have overall and disease-free survival similar to that of those who receive a total mastectomy.
When nipple-sparing mastectomy was introduced, many experts felt uneasy about opting for the less invasive procedure, recalled Rosa Hwang, MD, associate medical director for breast surgery at MD Anderson Cancer Center in Houston. “The concern was leaving all this skin,” said Dr. Hwang. “Are you going to leave cancer behind” and increase the risk of local recurrence?
Over the past 2 decades, the number of patients undergoing nipple-sparing mastectomy increased and, in turn, studies began to demonstrate the safety of the procedure.
However, large analyses evaluating long-term outcomes – namely, overall survival and breast cancer-specific survival – of nipple-sparing mastectomy were still lacking.
The latest study, published online Nov. 20 in Annals of Surgical Oncology, compared the long-term prognosis and survival benefits of nipple-sparing to total mastectomy in thousands of women. The analysis, which pulled data from the SEER cancer database, included 5,765 patients who underwent the nipple-sparing procedure and 17,289 patients who had a total mastectomy.
The authors found that overall survival and breast cancer–specific survival were similar for women undergoing nipple-sparing mastectomy and those receiving a total mastectomy. In fact, over the long-term, the nipple-sparing group slightly edged out the total mastectomy group in overall survival (94.61% vs. 93% at 5 years and 86.34% vs. 83.48% at 10 years, respectively) and in breast cancer-specific survival rates (96.16% vs. 95.74% at 5 years, and 92.2% vs. 91.37% at 10 years). The differences, however, were not significant.
The study also found that certain subgroups – including White women, women over age 46, those with a median household income of $70,000 or more, hormone receptor-positive, and HER2 negative – had significantly better overall survival rate with the nipple-sparing procedure (P < .05). However, the authors noted, the survival advantage in the nipple-sparing group did not extend to breast cancer–specific survival.
Dr. Hwang, who was not involved in the current analysis, said the significant overall survival result in the subgroup analysis was surprising because “there’s no biological reason why one would expect that to be true.”
Given that the subgroups did not demonstrate better breast cancer–specific survival, Dr. Hwang believes the overall survival finding may have more to do with comorbidities, which the study did not account for, than type of mastectomy.
When choosing who is eligible for a nipple-sparing mastectomy, “We’re more selective,” Dr. Hwang said. For instance, patients with uncontrolled diabetes or who smoke are unlikely to be candidates. “So, I think it’s possible that medical comorbidities and medical conditions between these groups [were] different.”
According to the authors, coding inconsistencies represent another possible weakness of the study. From 1998 to 2010, “the term ‘nipple-sparing mastectomy’ was coded as a [total mastectomy] with the ‘subcutaneous mastectomy’ code.” It’s possible that some patients receiving the nipple-sparing procedure before 2011 were not appropriately coded in the current study.
Moving forward, a large prospective study that includes comorbidities would be helpful, but overall the study helps validate that “nipple-sparing mastectomy is a safe operation for selected patients,” Dr. Hwang said.
A version of this article first appeared on Medscape.com.
A new analysis of over 22,000 mastectomy patients confirms what smaller studies have indicated: Patients who undergo nipple-sparing mastectomy have overall and disease-free survival similar to that of those who receive a total mastectomy.
When nipple-sparing mastectomy was introduced, many experts felt uneasy about opting for the less invasive procedure, recalled Rosa Hwang, MD, associate medical director for breast surgery at MD Anderson Cancer Center in Houston. “The concern was leaving all this skin,” said Dr. Hwang. “Are you going to leave cancer behind” and increase the risk of local recurrence?
Over the past 2 decades, the number of patients undergoing nipple-sparing mastectomy increased and, in turn, studies began to demonstrate the safety of the procedure.
However, large analyses evaluating long-term outcomes – namely, overall survival and breast cancer-specific survival – of nipple-sparing mastectomy were still lacking.
The latest study, published online Nov. 20 in Annals of Surgical Oncology, compared the long-term prognosis and survival benefits of nipple-sparing to total mastectomy in thousands of women. The analysis, which pulled data from the SEER cancer database, included 5,765 patients who underwent the nipple-sparing procedure and 17,289 patients who had a total mastectomy.
The authors found that overall survival and breast cancer–specific survival were similar for women undergoing nipple-sparing mastectomy and those receiving a total mastectomy. In fact, over the long-term, the nipple-sparing group slightly edged out the total mastectomy group in overall survival (94.61% vs. 93% at 5 years and 86.34% vs. 83.48% at 10 years, respectively) and in breast cancer-specific survival rates (96.16% vs. 95.74% at 5 years, and 92.2% vs. 91.37% at 10 years). The differences, however, were not significant.
The study also found that certain subgroups – including White women, women over age 46, those with a median household income of $70,000 or more, hormone receptor-positive, and HER2 negative – had significantly better overall survival rate with the nipple-sparing procedure (P < .05). However, the authors noted, the survival advantage in the nipple-sparing group did not extend to breast cancer–specific survival.
Dr. Hwang, who was not involved in the current analysis, said the significant overall survival result in the subgroup analysis was surprising because “there’s no biological reason why one would expect that to be true.”
Given that the subgroups did not demonstrate better breast cancer–specific survival, Dr. Hwang believes the overall survival finding may have more to do with comorbidities, which the study did not account for, than type of mastectomy.
When choosing who is eligible for a nipple-sparing mastectomy, “We’re more selective,” Dr. Hwang said. For instance, patients with uncontrolled diabetes or who smoke are unlikely to be candidates. “So, I think it’s possible that medical comorbidities and medical conditions between these groups [were] different.”
According to the authors, coding inconsistencies represent another possible weakness of the study. From 1998 to 2010, “the term ‘nipple-sparing mastectomy’ was coded as a [total mastectomy] with the ‘subcutaneous mastectomy’ code.” It’s possible that some patients receiving the nipple-sparing procedure before 2011 were not appropriately coded in the current study.
Moving forward, a large prospective study that includes comorbidities would be helpful, but overall the study helps validate that “nipple-sparing mastectomy is a safe operation for selected patients,” Dr. Hwang said.
A version of this article first appeared on Medscape.com.
A new analysis of over 22,000 mastectomy patients confirms what smaller studies have indicated: Patients who undergo nipple-sparing mastectomy have overall and disease-free survival similar to that of those who receive a total mastectomy.
When nipple-sparing mastectomy was introduced, many experts felt uneasy about opting for the less invasive procedure, recalled Rosa Hwang, MD, associate medical director for breast surgery at MD Anderson Cancer Center in Houston. “The concern was leaving all this skin,” said Dr. Hwang. “Are you going to leave cancer behind” and increase the risk of local recurrence?
Over the past 2 decades, the number of patients undergoing nipple-sparing mastectomy increased and, in turn, studies began to demonstrate the safety of the procedure.
However, large analyses evaluating long-term outcomes – namely, overall survival and breast cancer-specific survival – of nipple-sparing mastectomy were still lacking.
The latest study, published online Nov. 20 in Annals of Surgical Oncology, compared the long-term prognosis and survival benefits of nipple-sparing to total mastectomy in thousands of women. The analysis, which pulled data from the SEER cancer database, included 5,765 patients who underwent the nipple-sparing procedure and 17,289 patients who had a total mastectomy.
The authors found that overall survival and breast cancer–specific survival were similar for women undergoing nipple-sparing mastectomy and those receiving a total mastectomy. In fact, over the long-term, the nipple-sparing group slightly edged out the total mastectomy group in overall survival (94.61% vs. 93% at 5 years and 86.34% vs. 83.48% at 10 years, respectively) and in breast cancer-specific survival rates (96.16% vs. 95.74% at 5 years, and 92.2% vs. 91.37% at 10 years). The differences, however, were not significant.
The study also found that certain subgroups – including White women, women over age 46, those with a median household income of $70,000 or more, hormone receptor-positive, and HER2 negative – had significantly better overall survival rate with the nipple-sparing procedure (P < .05). However, the authors noted, the survival advantage in the nipple-sparing group did not extend to breast cancer–specific survival.
Dr. Hwang, who was not involved in the current analysis, said the significant overall survival result in the subgroup analysis was surprising because “there’s no biological reason why one would expect that to be true.”
Given that the subgroups did not demonstrate better breast cancer–specific survival, Dr. Hwang believes the overall survival finding may have more to do with comorbidities, which the study did not account for, than type of mastectomy.
When choosing who is eligible for a nipple-sparing mastectomy, “We’re more selective,” Dr. Hwang said. For instance, patients with uncontrolled diabetes or who smoke are unlikely to be candidates. “So, I think it’s possible that medical comorbidities and medical conditions between these groups [were] different.”
According to the authors, coding inconsistencies represent another possible weakness of the study. From 1998 to 2010, “the term ‘nipple-sparing mastectomy’ was coded as a [total mastectomy] with the ‘subcutaneous mastectomy’ code.” It’s possible that some patients receiving the nipple-sparing procedure before 2011 were not appropriately coded in the current study.
Moving forward, a large prospective study that includes comorbidities would be helpful, but overall the study helps validate that “nipple-sparing mastectomy is a safe operation for selected patients,” Dr. Hwang said.
A version of this article first appeared on Medscape.com.
The gender pay gap, care economy, and mental health
According to an analysis by the Pew Research Center and a report by the National Women’s Law Center, women were earning approximately $0.83-$0.84 for every $1.00 earned by their male counterparts in 2020. Accordingly, women would need to work an additional 42 days to receive compensation for earnings by men during that year. Moreover, these gaps exist with respect to race inequalities. For example, Black and Latinx women who are working full-time were reported to earn approximately $0.64 and $0.57, respectively, for every $1.00 compared with their white, non-Hispanic male counterparts. Striking, isn’t it?
The gender pay gap also affects physicians. A 2021 Medscape survey found that male physicians earn 35% more than female physicians. The biggest gap seems to be between male and female specialists, with men earning $376,000 and women $283,000.
Gender inequality and COVID-19
In addition to workplace responsibilities, women are more likely to take on unpaid positions in the informal care economy – examples of these tasks include cleaning, grocery shopping, and child care. In fact, the COVID-19 pandemic has increased the burden of unpaid care work among women, which often incurs a significant impact on their participation in the paid economy.
A study in the United States evaluating the impact of gender inequality during COVID-19 suggested that the rise in unemployment among women during this time may be related to decreased occupational flexibility. Accordingly, the closure of schools and caregiving facilities has translated into increased responsibilities as the informal caregiver, and a decreased ability to fulfill work obligations. Consequently, women may be overwhelmed and unable to maintain their employment status, are limited in their work opportunities, and/or are furloughed or passed over for promotions.
Gendered pay gaps affect mental health
A study by Platt and colleagues investigated the relationship between gendered wage gaps and gendered disparities in depression and anxiety disorders. Researchers found that females with a lower income compared with their matched male counterparts were more likely to experience depression and generalized anxiety disorders (i.e., they were 2.4 times more likely to experience depression and 4 times more likely to experience anxiety), while women who earned more than men did not report a significant difference in depression there were reduced gaps in the prevalence of anxiety disorders. As such, it has been suggested that wage gap inequalities are a contributing factor to gendered mental health disparities.
Reduced pay is not only a signifier of reduced returns on human capital. It may also have implications for one’s role in the care economy (e.g., greater time allocation as a result of reduced return), and may result in a higher likelihood for relocation as it relates to a partner’s work, overqualification for a position, inflexible work schedules, and reduced work autonomy.
Wage inequalities may act as a proxy for workplace inequalities such as promotions, prestigious projects, limited upward mobility, and internalized negative workplace experiences, all of which may contribute to increased sleep loss, stress, and related mental health stressors.
One might say, “A few cents, so what?” We should encourage conversations around the gender pay gap and develop strategies to combat this economic and social disparity.
Ms. Lui completed an HBSc global health specialist degree at the University of Toronto, where she is now an MSc candidate. She has received income from Braxia Scientific Corp. A version of this article first appeared on Medscape.com.
According to an analysis by the Pew Research Center and a report by the National Women’s Law Center, women were earning approximately $0.83-$0.84 for every $1.00 earned by their male counterparts in 2020. Accordingly, women would need to work an additional 42 days to receive compensation for earnings by men during that year. Moreover, these gaps exist with respect to race inequalities. For example, Black and Latinx women who are working full-time were reported to earn approximately $0.64 and $0.57, respectively, for every $1.00 compared with their white, non-Hispanic male counterparts. Striking, isn’t it?
The gender pay gap also affects physicians. A 2021 Medscape survey found that male physicians earn 35% more than female physicians. The biggest gap seems to be between male and female specialists, with men earning $376,000 and women $283,000.
Gender inequality and COVID-19
In addition to workplace responsibilities, women are more likely to take on unpaid positions in the informal care economy – examples of these tasks include cleaning, grocery shopping, and child care. In fact, the COVID-19 pandemic has increased the burden of unpaid care work among women, which often incurs a significant impact on their participation in the paid economy.
A study in the United States evaluating the impact of gender inequality during COVID-19 suggested that the rise in unemployment among women during this time may be related to decreased occupational flexibility. Accordingly, the closure of schools and caregiving facilities has translated into increased responsibilities as the informal caregiver, and a decreased ability to fulfill work obligations. Consequently, women may be overwhelmed and unable to maintain their employment status, are limited in their work opportunities, and/or are furloughed or passed over for promotions.
Gendered pay gaps affect mental health
A study by Platt and colleagues investigated the relationship between gendered wage gaps and gendered disparities in depression and anxiety disorders. Researchers found that females with a lower income compared with their matched male counterparts were more likely to experience depression and generalized anxiety disorders (i.e., they were 2.4 times more likely to experience depression and 4 times more likely to experience anxiety), while women who earned more than men did not report a significant difference in depression there were reduced gaps in the prevalence of anxiety disorders. As such, it has been suggested that wage gap inequalities are a contributing factor to gendered mental health disparities.
Reduced pay is not only a signifier of reduced returns on human capital. It may also have implications for one’s role in the care economy (e.g., greater time allocation as a result of reduced return), and may result in a higher likelihood for relocation as it relates to a partner’s work, overqualification for a position, inflexible work schedules, and reduced work autonomy.
Wage inequalities may act as a proxy for workplace inequalities such as promotions, prestigious projects, limited upward mobility, and internalized negative workplace experiences, all of which may contribute to increased sleep loss, stress, and related mental health stressors.
One might say, “A few cents, so what?” We should encourage conversations around the gender pay gap and develop strategies to combat this economic and social disparity.
Ms. Lui completed an HBSc global health specialist degree at the University of Toronto, where she is now an MSc candidate. She has received income from Braxia Scientific Corp. A version of this article first appeared on Medscape.com.
According to an analysis by the Pew Research Center and a report by the National Women’s Law Center, women were earning approximately $0.83-$0.84 for every $1.00 earned by their male counterparts in 2020. Accordingly, women would need to work an additional 42 days to receive compensation for earnings by men during that year. Moreover, these gaps exist with respect to race inequalities. For example, Black and Latinx women who are working full-time were reported to earn approximately $0.64 and $0.57, respectively, for every $1.00 compared with their white, non-Hispanic male counterparts. Striking, isn’t it?
The gender pay gap also affects physicians. A 2021 Medscape survey found that male physicians earn 35% more than female physicians. The biggest gap seems to be between male and female specialists, with men earning $376,000 and women $283,000.
Gender inequality and COVID-19
In addition to workplace responsibilities, women are more likely to take on unpaid positions in the informal care economy – examples of these tasks include cleaning, grocery shopping, and child care. In fact, the COVID-19 pandemic has increased the burden of unpaid care work among women, which often incurs a significant impact on their participation in the paid economy.
A study in the United States evaluating the impact of gender inequality during COVID-19 suggested that the rise in unemployment among women during this time may be related to decreased occupational flexibility. Accordingly, the closure of schools and caregiving facilities has translated into increased responsibilities as the informal caregiver, and a decreased ability to fulfill work obligations. Consequently, women may be overwhelmed and unable to maintain their employment status, are limited in their work opportunities, and/or are furloughed or passed over for promotions.
Gendered pay gaps affect mental health
A study by Platt and colleagues investigated the relationship between gendered wage gaps and gendered disparities in depression and anxiety disorders. Researchers found that females with a lower income compared with their matched male counterparts were more likely to experience depression and generalized anxiety disorders (i.e., they were 2.4 times more likely to experience depression and 4 times more likely to experience anxiety), while women who earned more than men did not report a significant difference in depression there were reduced gaps in the prevalence of anxiety disorders. As such, it has been suggested that wage gap inequalities are a contributing factor to gendered mental health disparities.
Reduced pay is not only a signifier of reduced returns on human capital. It may also have implications for one’s role in the care economy (e.g., greater time allocation as a result of reduced return), and may result in a higher likelihood for relocation as it relates to a partner’s work, overqualification for a position, inflexible work schedules, and reduced work autonomy.
Wage inequalities may act as a proxy for workplace inequalities such as promotions, prestigious projects, limited upward mobility, and internalized negative workplace experiences, all of which may contribute to increased sleep loss, stress, and related mental health stressors.
One might say, “A few cents, so what?” We should encourage conversations around the gender pay gap and develop strategies to combat this economic and social disparity.
Ms. Lui completed an HBSc global health specialist degree at the University of Toronto, where she is now an MSc candidate. She has received income from Braxia Scientific Corp. A version of this article first appeared on Medscape.com.
HPV vaccines reduce cervical cancer rates in young females
Two different studies have found that, provided young females are immunized with the human papilloma virus (HPV) vaccine at a young enough age, both the incidence of and mortality from cervical cancer can be dramatically curtailed, data from the United Kingdom and to a lesser extent, the United States indicate.
In the U.K. study, published online in The Lancet, researchers showed that the national vaccination program against HPV, initiated in England in 2008, has all but eradicated cervical cancer and cervical intraepithelial neoplasia (CIN3) in young girls who received the vaccine at the age of 12 and 13 years (school year 8) prior to their sexual debut.
In this age group, cervical cancer rates were 87% lower than rates among previously nonvaccinated generations, while CIN3 rates were reduced by 97%, as researchers report. “It’s been incredible to see the impact of HPV vaccination, and now we can prove it prevented hundreds of women from developing cancer in England,” senior author Peter Sasieni, MD, King’s College London, said in a statement. “To see the real-life impact of the vaccine has been truly rewarding,” he added.
“This study provides the first direct evidence of the impact of the UK HPV vaccination campaign on cervical cancer incidence, showing a large reduction in cervical cancer rates in vaccinated cohorts,” Kate Soldan, MD, UK Health Security Agency, London, said in the same statement.
“This represents an important step forward in cervical cancer prevention, and we hope that these new results encourage uptake as the success of the vaccination programme relies not only on the efficacy of the vaccine but also the proportion of the population vaccinated,” she added.
Vanessa Saliba, MD, a consultant epidemiologist for the UK Health Security Agency, agreed, adding that “these remarkable findings confirm that the HPV vaccine saves lives by dramatically reducing cervical cancer rates among women.”
“This reminds us that vaccines are one of the most important tools we have to help us live longer, healthier lives,” she reemphasized.
British HPV program
When initiated in 2008, the national HPV vaccination program used the bivalent, Cervarix vaccine against HPV 16 and 18. As researchers noted, these two HPV types are responsible for 70%-80% of all cervical cancers in England.
However, in 2012, the program switched to the quadrivalent HPV vaccine (Gardasil) which is also effective against two additional HPV types, 6 and 11, both of which cause genital warts. The program also originally recommended the three-dose regimen for both HPV vaccines.
Now, only two doses of the vaccine are given to girls under the age of 15 even though it has been shown that a single dose of the HPV vaccine provides good protection against persistent infection, with efficacy rates that are similar to that of three doses, as the authors point out.
Among the cohort eligible for vaccination at 12 or 13 years of age, 89% received at least one dose of the HPV vaccine while 85% of the same age group received all three shots.
Cancer registry
Data from a population-based cancer registry was used to estimate the early effect of the bivalent HPV program on the incidence of cervical cancer and CIN3 in England between January 2006 and June 2019. During the study interval, there were 27,946 diagnoses of cervical cancer and 318,058 diagnoses of CIN3, lead author Milena Falcaro, MD, King’s College London, and colleagues report. Participants were then analyzed separately according to their age at the time of vaccination and the incidence rates calculated for both cervical cancer and CIN3 in the three separate groups.
For slightly older girls who received the vaccine between 14 and 16 years of age (school year 10-11), cervical cancer was reduced by 62% while CIN3 rates were reduced by 75%. For those who received the vaccine between 16 and 18 years of age (school year 12-13), cervical cancer rates were reduced by 34% while CIN3 rates were reduced by 39%, study authors add.
Indeed, the authors estimate that by June 2019 there were approximately 450 fewer cases of cervical cancer and 17,200 fewer cases of CIN3 than would otherwise have been expected in the vaccinated population in England.
The authors acknowledge that cervical cancer is rare in young women and vaccinated populations are still young. For example, the youngest recipients would have been immunized at the age of 12 in 2008 and would still be only 23 years old in 2019 when the study ended.
Thus, the authors emphasize that, because the vaccinated populations are still young, it’s too early to assess the full effect of HPV vaccination on cervical cancer rates.
Asked to comment on the study, Maurice Markman, MD, president, Medicine and Science Cancer Treatment Centers of America, pointed out that results from the British study are very similar to those from a Swedish study assessing the effect of the quadrivalent vaccine alone.
“You can put any superlatives you want in here, but these are stunningly positive results,” Dr. Markman said in an interview. As an oncologist who has been treating cervical cancer for 40 years – particularly advanced cervical cancer – “I can tell you this is one of the most devastating diseases to women, and the ability to eliminate this cancer with something as simple as a vaccine is the goal of cancer therapy, and it’s been remarkably successful,” he stressed.
Editorial commentary
Commenting on the findings, editorialists Maggie Cruickshank, MD, University of Aberdeen (Scotland), and Mihaela Grigore, MD, University of Medicine and Pharmacy, Lasi, Romania, point out that published reports evaluating the effect of HPV vaccination on cervical cancer rates have been scarce until now.
“The most important issue, besides the availability of the vaccine ... is the education of the population to accept vaccination because a high rate of immunization is a key element of success,” they emphasize. “Even in a wealthy country such as England with free access to HPV immunization, uptake has not reached the 90% vaccination target of girls aged 15 years set by the WHO [World Health Organization],” the editorialists add.
Dr. Cruickshank and Dr. Grigore also suggest that the effect HPV vaccination is having on cervical cancer rates as shown in this study should also stimulate vaccination programs in low- and middle-income countries where cervical cancer is a far greater public health issue than it is in countries with established systems of vaccination and screening.
HPV vaccination in the United States
The HPV vaccination program is similarly reducing the incidence of and mortality from cervical cancer among younger women in the United States who are most likely to have received the vaccine. As reported by lead author, Justin Barnes, MD, Washington University, St. Louis, the incidence of cervical cancer dropped by 37.7% from 2001 through 2005 to 2010 through 2017 in girls and young women between 15 and 24 years of age.
The U.S. study was published online in JAMA Pediatrics.
“HPV vaccine coverage in the U.S. has improved over the last few years although it was quite poor for many years,” senior author of the U.K. study, Peter Sasieni, MD, King’s College London, said in an interview. “Thus, one would anticipate a lower impact on the population in the U.S., because vaccine uptake, particularly in those aged 11-14 years was so much lower than it was in the U.K.,” he noted.
SEER databases
National age-adjusted cervical cancer incidence and mortality data from January 2001 through December 2017 for women and girls between 15 and 39 years of age were obtained from the combined Surveillance, Epidemiology, and End Results as well as the National Program of Cancer Registries databases. Mortality data was obtained from the National Center for Health Statistics.
Investigators then compared percentage changes in the incidence of and mortality from cervical cancer from January 2001 through December 2005 during the prevaccination years to that observed between January 2010 through December 2017 during the postvaccination years. They also compared incidence and mortality rates in three different cohorts: females between 15 and 24 years of age, those between 25 and 29 years of age, and those between 30 and 39 years of age.
“The older two groups were included as comparison, given their low vaccination rates,” the authors explained. Results showed that, during the same study interval from 2001 through 2005 to 2010 through 2017, the incidence of cervical cancer dropped by only 16.1% in women between 25 and 29 years of age and by only 8% for women between 30 and 39 years of age, the investigators report.
Reductions in mortality from cervical cancer were only strikingly so in the youngest age group of females between 15 and 24 years of age, among whom there was a 43.3% reduction in mortality from 2001-2005 to 2010-2017, as Dr. Barnes and colleagues note.
This pattern changed substantially in women between the ages of 25 and 29, among whom there was a 4.3% increase in mortality from cervical cancer during the same study interval and a small, 4.7% reduction among women between 30 and 39 years of age, investigators add. In actual numbers, mortality rates from cervical cancer were very low at only 0.6 per 100,000 in females between 15 and 24 years of age.
This compared to a mortality rate of 0.57 per 100,000 in women between 25 and 29 years of age and 1.89 per 100,000 in the oldest age group. “These nationwide data showed decreased cervical cancer incidence and mortality among women and girls aged 15-24 years after HPV vaccine introduction,” Dr. Barnes notes.
“Thus, the current study adds to knowledge by quantitatively comparing changes in cervical cancer incidence by age-based vaccine eligibility and providing suggestive evidence for vaccine-associated decreases in cervical cancer mortality,” investigators add.
However, as the authors also point out, while the reduction in mortality from cervical cancer associated with HPV vaccination may translate to older age groups as HPV-vaccinated cohorts age, “the number of deaths and hence the number of potentially averted deaths in young women and girls was small,” they caution, “and efforts to further improve vaccination uptake remain important.”
None of the authors or the editorialists had any conflicts of interest to declare.
Two different studies have found that, provided young females are immunized with the human papilloma virus (HPV) vaccine at a young enough age, both the incidence of and mortality from cervical cancer can be dramatically curtailed, data from the United Kingdom and to a lesser extent, the United States indicate.
In the U.K. study, published online in The Lancet, researchers showed that the national vaccination program against HPV, initiated in England in 2008, has all but eradicated cervical cancer and cervical intraepithelial neoplasia (CIN3) in young girls who received the vaccine at the age of 12 and 13 years (school year 8) prior to their sexual debut.
In this age group, cervical cancer rates were 87% lower than rates among previously nonvaccinated generations, while CIN3 rates were reduced by 97%, as researchers report. “It’s been incredible to see the impact of HPV vaccination, and now we can prove it prevented hundreds of women from developing cancer in England,” senior author Peter Sasieni, MD, King’s College London, said in a statement. “To see the real-life impact of the vaccine has been truly rewarding,” he added.
“This study provides the first direct evidence of the impact of the UK HPV vaccination campaign on cervical cancer incidence, showing a large reduction in cervical cancer rates in vaccinated cohorts,” Kate Soldan, MD, UK Health Security Agency, London, said in the same statement.
“This represents an important step forward in cervical cancer prevention, and we hope that these new results encourage uptake as the success of the vaccination programme relies not only on the efficacy of the vaccine but also the proportion of the population vaccinated,” she added.
Vanessa Saliba, MD, a consultant epidemiologist for the UK Health Security Agency, agreed, adding that “these remarkable findings confirm that the HPV vaccine saves lives by dramatically reducing cervical cancer rates among women.”
“This reminds us that vaccines are one of the most important tools we have to help us live longer, healthier lives,” she reemphasized.
British HPV program
When initiated in 2008, the national HPV vaccination program used the bivalent, Cervarix vaccine against HPV 16 and 18. As researchers noted, these two HPV types are responsible for 70%-80% of all cervical cancers in England.
However, in 2012, the program switched to the quadrivalent HPV vaccine (Gardasil) which is also effective against two additional HPV types, 6 and 11, both of which cause genital warts. The program also originally recommended the three-dose regimen for both HPV vaccines.
Now, only two doses of the vaccine are given to girls under the age of 15 even though it has been shown that a single dose of the HPV vaccine provides good protection against persistent infection, with efficacy rates that are similar to that of three doses, as the authors point out.
Among the cohort eligible for vaccination at 12 or 13 years of age, 89% received at least one dose of the HPV vaccine while 85% of the same age group received all three shots.
Cancer registry
Data from a population-based cancer registry was used to estimate the early effect of the bivalent HPV program on the incidence of cervical cancer and CIN3 in England between January 2006 and June 2019. During the study interval, there were 27,946 diagnoses of cervical cancer and 318,058 diagnoses of CIN3, lead author Milena Falcaro, MD, King’s College London, and colleagues report. Participants were then analyzed separately according to their age at the time of vaccination and the incidence rates calculated for both cervical cancer and CIN3 in the three separate groups.
For slightly older girls who received the vaccine between 14 and 16 years of age (school year 10-11), cervical cancer was reduced by 62% while CIN3 rates were reduced by 75%. For those who received the vaccine between 16 and 18 years of age (school year 12-13), cervical cancer rates were reduced by 34% while CIN3 rates were reduced by 39%, study authors add.
Indeed, the authors estimate that by June 2019 there were approximately 450 fewer cases of cervical cancer and 17,200 fewer cases of CIN3 than would otherwise have been expected in the vaccinated population in England.
The authors acknowledge that cervical cancer is rare in young women and vaccinated populations are still young. For example, the youngest recipients would have been immunized at the age of 12 in 2008 and would still be only 23 years old in 2019 when the study ended.
Thus, the authors emphasize that, because the vaccinated populations are still young, it’s too early to assess the full effect of HPV vaccination on cervical cancer rates.
Asked to comment on the study, Maurice Markman, MD, president, Medicine and Science Cancer Treatment Centers of America, pointed out that results from the British study are very similar to those from a Swedish study assessing the effect of the quadrivalent vaccine alone.
“You can put any superlatives you want in here, but these are stunningly positive results,” Dr. Markman said in an interview. As an oncologist who has been treating cervical cancer for 40 years – particularly advanced cervical cancer – “I can tell you this is one of the most devastating diseases to women, and the ability to eliminate this cancer with something as simple as a vaccine is the goal of cancer therapy, and it’s been remarkably successful,” he stressed.
Editorial commentary
Commenting on the findings, editorialists Maggie Cruickshank, MD, University of Aberdeen (Scotland), and Mihaela Grigore, MD, University of Medicine and Pharmacy, Lasi, Romania, point out that published reports evaluating the effect of HPV vaccination on cervical cancer rates have been scarce until now.
“The most important issue, besides the availability of the vaccine ... is the education of the population to accept vaccination because a high rate of immunization is a key element of success,” they emphasize. “Even in a wealthy country such as England with free access to HPV immunization, uptake has not reached the 90% vaccination target of girls aged 15 years set by the WHO [World Health Organization],” the editorialists add.
Dr. Cruickshank and Dr. Grigore also suggest that the effect HPV vaccination is having on cervical cancer rates as shown in this study should also stimulate vaccination programs in low- and middle-income countries where cervical cancer is a far greater public health issue than it is in countries with established systems of vaccination and screening.
HPV vaccination in the United States
The HPV vaccination program is similarly reducing the incidence of and mortality from cervical cancer among younger women in the United States who are most likely to have received the vaccine. As reported by lead author, Justin Barnes, MD, Washington University, St. Louis, the incidence of cervical cancer dropped by 37.7% from 2001 through 2005 to 2010 through 2017 in girls and young women between 15 and 24 years of age.
The U.S. study was published online in JAMA Pediatrics.
“HPV vaccine coverage in the U.S. has improved over the last few years although it was quite poor for many years,” senior author of the U.K. study, Peter Sasieni, MD, King’s College London, said in an interview. “Thus, one would anticipate a lower impact on the population in the U.S., because vaccine uptake, particularly in those aged 11-14 years was so much lower than it was in the U.K.,” he noted.
SEER databases
National age-adjusted cervical cancer incidence and mortality data from January 2001 through December 2017 for women and girls between 15 and 39 years of age were obtained from the combined Surveillance, Epidemiology, and End Results as well as the National Program of Cancer Registries databases. Mortality data was obtained from the National Center for Health Statistics.
Investigators then compared percentage changes in the incidence of and mortality from cervical cancer from January 2001 through December 2005 during the prevaccination years to that observed between January 2010 through December 2017 during the postvaccination years. They also compared incidence and mortality rates in three different cohorts: females between 15 and 24 years of age, those between 25 and 29 years of age, and those between 30 and 39 years of age.
“The older two groups were included as comparison, given their low vaccination rates,” the authors explained. Results showed that, during the same study interval from 2001 through 2005 to 2010 through 2017, the incidence of cervical cancer dropped by only 16.1% in women between 25 and 29 years of age and by only 8% for women between 30 and 39 years of age, the investigators report.
Reductions in mortality from cervical cancer were only strikingly so in the youngest age group of females between 15 and 24 years of age, among whom there was a 43.3% reduction in mortality from 2001-2005 to 2010-2017, as Dr. Barnes and colleagues note.
This pattern changed substantially in women between the ages of 25 and 29, among whom there was a 4.3% increase in mortality from cervical cancer during the same study interval and a small, 4.7% reduction among women between 30 and 39 years of age, investigators add. In actual numbers, mortality rates from cervical cancer were very low at only 0.6 per 100,000 in females between 15 and 24 years of age.
This compared to a mortality rate of 0.57 per 100,000 in women between 25 and 29 years of age and 1.89 per 100,000 in the oldest age group. “These nationwide data showed decreased cervical cancer incidence and mortality among women and girls aged 15-24 years after HPV vaccine introduction,” Dr. Barnes notes.
“Thus, the current study adds to knowledge by quantitatively comparing changes in cervical cancer incidence by age-based vaccine eligibility and providing suggestive evidence for vaccine-associated decreases in cervical cancer mortality,” investigators add.
However, as the authors also point out, while the reduction in mortality from cervical cancer associated with HPV vaccination may translate to older age groups as HPV-vaccinated cohorts age, “the number of deaths and hence the number of potentially averted deaths in young women and girls was small,” they caution, “and efforts to further improve vaccination uptake remain important.”
None of the authors or the editorialists had any conflicts of interest to declare.
Two different studies have found that, provided young females are immunized with the human papilloma virus (HPV) vaccine at a young enough age, both the incidence of and mortality from cervical cancer can be dramatically curtailed, data from the United Kingdom and to a lesser extent, the United States indicate.
In the U.K. study, published online in The Lancet, researchers showed that the national vaccination program against HPV, initiated in England in 2008, has all but eradicated cervical cancer and cervical intraepithelial neoplasia (CIN3) in young girls who received the vaccine at the age of 12 and 13 years (school year 8) prior to their sexual debut.
In this age group, cervical cancer rates were 87% lower than rates among previously nonvaccinated generations, while CIN3 rates were reduced by 97%, as researchers report. “It’s been incredible to see the impact of HPV vaccination, and now we can prove it prevented hundreds of women from developing cancer in England,” senior author Peter Sasieni, MD, King’s College London, said in a statement. “To see the real-life impact of the vaccine has been truly rewarding,” he added.
“This study provides the first direct evidence of the impact of the UK HPV vaccination campaign on cervical cancer incidence, showing a large reduction in cervical cancer rates in vaccinated cohorts,” Kate Soldan, MD, UK Health Security Agency, London, said in the same statement.
“This represents an important step forward in cervical cancer prevention, and we hope that these new results encourage uptake as the success of the vaccination programme relies not only on the efficacy of the vaccine but also the proportion of the population vaccinated,” she added.
Vanessa Saliba, MD, a consultant epidemiologist for the UK Health Security Agency, agreed, adding that “these remarkable findings confirm that the HPV vaccine saves lives by dramatically reducing cervical cancer rates among women.”
“This reminds us that vaccines are one of the most important tools we have to help us live longer, healthier lives,” she reemphasized.
British HPV program
When initiated in 2008, the national HPV vaccination program used the bivalent, Cervarix vaccine against HPV 16 and 18. As researchers noted, these two HPV types are responsible for 70%-80% of all cervical cancers in England.
However, in 2012, the program switched to the quadrivalent HPV vaccine (Gardasil) which is also effective against two additional HPV types, 6 and 11, both of which cause genital warts. The program also originally recommended the three-dose regimen for both HPV vaccines.
Now, only two doses of the vaccine are given to girls under the age of 15 even though it has been shown that a single dose of the HPV vaccine provides good protection against persistent infection, with efficacy rates that are similar to that of three doses, as the authors point out.
Among the cohort eligible for vaccination at 12 or 13 years of age, 89% received at least one dose of the HPV vaccine while 85% of the same age group received all three shots.
Cancer registry
Data from a population-based cancer registry was used to estimate the early effect of the bivalent HPV program on the incidence of cervical cancer and CIN3 in England between January 2006 and June 2019. During the study interval, there were 27,946 diagnoses of cervical cancer and 318,058 diagnoses of CIN3, lead author Milena Falcaro, MD, King’s College London, and colleagues report. Participants were then analyzed separately according to their age at the time of vaccination and the incidence rates calculated for both cervical cancer and CIN3 in the three separate groups.
For slightly older girls who received the vaccine between 14 and 16 years of age (school year 10-11), cervical cancer was reduced by 62% while CIN3 rates were reduced by 75%. For those who received the vaccine between 16 and 18 years of age (school year 12-13), cervical cancer rates were reduced by 34% while CIN3 rates were reduced by 39%, study authors add.
Indeed, the authors estimate that by June 2019 there were approximately 450 fewer cases of cervical cancer and 17,200 fewer cases of CIN3 than would otherwise have been expected in the vaccinated population in England.
The authors acknowledge that cervical cancer is rare in young women and vaccinated populations are still young. For example, the youngest recipients would have been immunized at the age of 12 in 2008 and would still be only 23 years old in 2019 when the study ended.
Thus, the authors emphasize that, because the vaccinated populations are still young, it’s too early to assess the full effect of HPV vaccination on cervical cancer rates.
Asked to comment on the study, Maurice Markman, MD, president, Medicine and Science Cancer Treatment Centers of America, pointed out that results from the British study are very similar to those from a Swedish study assessing the effect of the quadrivalent vaccine alone.
“You can put any superlatives you want in here, but these are stunningly positive results,” Dr. Markman said in an interview. As an oncologist who has been treating cervical cancer for 40 years – particularly advanced cervical cancer – “I can tell you this is one of the most devastating diseases to women, and the ability to eliminate this cancer with something as simple as a vaccine is the goal of cancer therapy, and it’s been remarkably successful,” he stressed.
Editorial commentary
Commenting on the findings, editorialists Maggie Cruickshank, MD, University of Aberdeen (Scotland), and Mihaela Grigore, MD, University of Medicine and Pharmacy, Lasi, Romania, point out that published reports evaluating the effect of HPV vaccination on cervical cancer rates have been scarce until now.
“The most important issue, besides the availability of the vaccine ... is the education of the population to accept vaccination because a high rate of immunization is a key element of success,” they emphasize. “Even in a wealthy country such as England with free access to HPV immunization, uptake has not reached the 90% vaccination target of girls aged 15 years set by the WHO [World Health Organization],” the editorialists add.
Dr. Cruickshank and Dr. Grigore also suggest that the effect HPV vaccination is having on cervical cancer rates as shown in this study should also stimulate vaccination programs in low- and middle-income countries where cervical cancer is a far greater public health issue than it is in countries with established systems of vaccination and screening.
HPV vaccination in the United States
The HPV vaccination program is similarly reducing the incidence of and mortality from cervical cancer among younger women in the United States who are most likely to have received the vaccine. As reported by lead author, Justin Barnes, MD, Washington University, St. Louis, the incidence of cervical cancer dropped by 37.7% from 2001 through 2005 to 2010 through 2017 in girls and young women between 15 and 24 years of age.
The U.S. study was published online in JAMA Pediatrics.
“HPV vaccine coverage in the U.S. has improved over the last few years although it was quite poor for many years,” senior author of the U.K. study, Peter Sasieni, MD, King’s College London, said in an interview. “Thus, one would anticipate a lower impact on the population in the U.S., because vaccine uptake, particularly in those aged 11-14 years was so much lower than it was in the U.K.,” he noted.
SEER databases
National age-adjusted cervical cancer incidence and mortality data from January 2001 through December 2017 for women and girls between 15 and 39 years of age were obtained from the combined Surveillance, Epidemiology, and End Results as well as the National Program of Cancer Registries databases. Mortality data was obtained from the National Center for Health Statistics.
Investigators then compared percentage changes in the incidence of and mortality from cervical cancer from January 2001 through December 2005 during the prevaccination years to that observed between January 2010 through December 2017 during the postvaccination years. They also compared incidence and mortality rates in three different cohorts: females between 15 and 24 years of age, those between 25 and 29 years of age, and those between 30 and 39 years of age.
“The older two groups were included as comparison, given their low vaccination rates,” the authors explained. Results showed that, during the same study interval from 2001 through 2005 to 2010 through 2017, the incidence of cervical cancer dropped by only 16.1% in women between 25 and 29 years of age and by only 8% for women between 30 and 39 years of age, the investigators report.
Reductions in mortality from cervical cancer were only strikingly so in the youngest age group of females between 15 and 24 years of age, among whom there was a 43.3% reduction in mortality from 2001-2005 to 2010-2017, as Dr. Barnes and colleagues note.
This pattern changed substantially in women between the ages of 25 and 29, among whom there was a 4.3% increase in mortality from cervical cancer during the same study interval and a small, 4.7% reduction among women between 30 and 39 years of age, investigators add. In actual numbers, mortality rates from cervical cancer were very low at only 0.6 per 100,000 in females between 15 and 24 years of age.
This compared to a mortality rate of 0.57 per 100,000 in women between 25 and 29 years of age and 1.89 per 100,000 in the oldest age group. “These nationwide data showed decreased cervical cancer incidence and mortality among women and girls aged 15-24 years after HPV vaccine introduction,” Dr. Barnes notes.
“Thus, the current study adds to knowledge by quantitatively comparing changes in cervical cancer incidence by age-based vaccine eligibility and providing suggestive evidence for vaccine-associated decreases in cervical cancer mortality,” investigators add.
However, as the authors also point out, while the reduction in mortality from cervical cancer associated with HPV vaccination may translate to older age groups as HPV-vaccinated cohorts age, “the number of deaths and hence the number of potentially averted deaths in young women and girls was small,” they caution, “and efforts to further improve vaccination uptake remain important.”
None of the authors or the editorialists had any conflicts of interest to declare.
Supreme Court receptive to case that could overturn Roe v. Wade
The justices heard from lawyers arguing for and against a 2018 Mississippi law that, with few exceptions, bans abortion after 15 weeks, claiming that a fetus is viable outside the womb at that age. The Supreme Court’s 1973 Roe v. Wade decision and legal rulings in the decades since, including the 1992 decision in Planned Parenthood v. Casey, have said that abortion should be available to the point of viability – established as about 23 weeks.
The court also ruled in Casey that state laws could not present an “undue burden” on a woman’s ability to obtain an abortion.
The Mississippi attorney general did not initially seek to overturn Roe and Casey, but later argued in Dobbs v. Jackson Women’s Health Organization that both cases were erroneously decided and should be completely thrown out.
“It is an egregiously wrong decision that has inflicted tremendous damage on our country and will continue to do so and take innumerable human lives unless and until this court overrules it,” said Scott G. Stewart, Mississippi’s solicitor general.
When it accepted the Mississippi case, the Supreme Court did not agree to weigh in on overturning Roe or Casey, but the justices’ leanings were evident during the hearing, and it is possible they would throw out those landmark cases.
Justice Clarence Thomas asked repeatedly for the law’s challengers to point out where the right to an abortion was written in the Constitution, as did Justice Samuel Alito.
“If we were talking about the Second Amendment, I know exactly what we’re talking about, if we’re talking about the Fourth Amendment, I know what we’re talking about, because it’s written, it’s there,” said Justice Thomas. “What specifically is the right here that we’re talking about?” he asked U.S. Solicitor General Elizabeth Prelogar.
She said the right to abortion was embedded in the 14th amendment’s guarantee of the pursuit of liberty.
“If this Court renounces the liberty interest recognized in Roe and reaffirmed in Casey, it would be an unprecedented contraction of individual rights,” and a departure from court doctrine of upholding precedent, known as stare decisis, she said.
Chief Justice John Roberts seemed to be against throwing out either of the landmark abortion cases, but instead wanted to focus on whether the 15 weeks was a reasonable time point. But he seemed to be alone in honing-in on that issue.
“Roberts seem desperate for some limiting principle that isn’t reversing Roe, and none of the other conservative justices are biting,” tweeted Mary Ziegler, a historian who has written about abortion.
But justices Neil Gorsuch, Amy Coney Barrett, and Brett Kavanaugh all appeared to be receptive to the idea that the prior precedent set by Roe and Casey could be overturned.
Neil Katyal, the former U.S. acting solicitor general and a Supreme Court lawyer, tweeted during the arguments that he saw “nothing so far sympathetic to the challengers. And a lot that has been very hostile.”
He cautioned that questions during oral arguments “often are just trying to understand a lawyer’s position,” adding, “But the tea leaves here are ominous.”
If Roe v. Wade is overturned, 22 states have laws already on the books that could be used to restrict abortion, according to the Guttmacher Institute. Almost all abortions would be banned in 12 states that have so-called “trigger” laws: Arkansas, Idaho, Kentucky, Louisiana, Mississippi, Missouri, North Dakota, Oklahoma, South Dakota, Tennessee, Texas, and Utah.
Seventeen states have abortion restrictions that have been unenforced or blocked by courts that would go back into effect if Roe is nullified. An additional seven states have laws that intend to restrict abortion in the absence of Roe and four states have passed constitutional amendments to specifically not protect the right to abortion.
Guttmacher reports that 15 states and the District of Columbia have passed laws that protect the right to abortion.
Jackson Women’s Health – the state’s sole abortion provider – sued to block the Mississippi law soon after it passed. A federal judge ruled against the state and that decision was upheld by the U.S. Fifth Circuit Court of Appeals, which also issued a permanent injunction against the law. The Supreme Court in May 2021 agreed to take Mississippi’s appeal.
Earlier in November, the Supreme Court heard arguments in two cases challenging a restrictive Texas law, Whole Woman’s Health v. Jackson and U.S. v. Texas. The justices seemed receptive to the idea that the law, SB 8, was unconstitutional. But the court did not grant a request by the Biden administration to halt the law while the challenges made their way through the courts.
A version of this article first appeared on WebMD.com.
The justices heard from lawyers arguing for and against a 2018 Mississippi law that, with few exceptions, bans abortion after 15 weeks, claiming that a fetus is viable outside the womb at that age. The Supreme Court’s 1973 Roe v. Wade decision and legal rulings in the decades since, including the 1992 decision in Planned Parenthood v. Casey, have said that abortion should be available to the point of viability – established as about 23 weeks.
The court also ruled in Casey that state laws could not present an “undue burden” on a woman’s ability to obtain an abortion.
The Mississippi attorney general did not initially seek to overturn Roe and Casey, but later argued in Dobbs v. Jackson Women’s Health Organization that both cases were erroneously decided and should be completely thrown out.
“It is an egregiously wrong decision that has inflicted tremendous damage on our country and will continue to do so and take innumerable human lives unless and until this court overrules it,” said Scott G. Stewart, Mississippi’s solicitor general.
When it accepted the Mississippi case, the Supreme Court did not agree to weigh in on overturning Roe or Casey, but the justices’ leanings were evident during the hearing, and it is possible they would throw out those landmark cases.
Justice Clarence Thomas asked repeatedly for the law’s challengers to point out where the right to an abortion was written in the Constitution, as did Justice Samuel Alito.
“If we were talking about the Second Amendment, I know exactly what we’re talking about, if we’re talking about the Fourth Amendment, I know what we’re talking about, because it’s written, it’s there,” said Justice Thomas. “What specifically is the right here that we’re talking about?” he asked U.S. Solicitor General Elizabeth Prelogar.
She said the right to abortion was embedded in the 14th amendment’s guarantee of the pursuit of liberty.
“If this Court renounces the liberty interest recognized in Roe and reaffirmed in Casey, it would be an unprecedented contraction of individual rights,” and a departure from court doctrine of upholding precedent, known as stare decisis, she said.
Chief Justice John Roberts seemed to be against throwing out either of the landmark abortion cases, but instead wanted to focus on whether the 15 weeks was a reasonable time point. But he seemed to be alone in honing-in on that issue.
“Roberts seem desperate for some limiting principle that isn’t reversing Roe, and none of the other conservative justices are biting,” tweeted Mary Ziegler, a historian who has written about abortion.
But justices Neil Gorsuch, Amy Coney Barrett, and Brett Kavanaugh all appeared to be receptive to the idea that the prior precedent set by Roe and Casey could be overturned.
Neil Katyal, the former U.S. acting solicitor general and a Supreme Court lawyer, tweeted during the arguments that he saw “nothing so far sympathetic to the challengers. And a lot that has been very hostile.”
He cautioned that questions during oral arguments “often are just trying to understand a lawyer’s position,” adding, “But the tea leaves here are ominous.”
If Roe v. Wade is overturned, 22 states have laws already on the books that could be used to restrict abortion, according to the Guttmacher Institute. Almost all abortions would be banned in 12 states that have so-called “trigger” laws: Arkansas, Idaho, Kentucky, Louisiana, Mississippi, Missouri, North Dakota, Oklahoma, South Dakota, Tennessee, Texas, and Utah.
Seventeen states have abortion restrictions that have been unenforced or blocked by courts that would go back into effect if Roe is nullified. An additional seven states have laws that intend to restrict abortion in the absence of Roe and four states have passed constitutional amendments to specifically not protect the right to abortion.
Guttmacher reports that 15 states and the District of Columbia have passed laws that protect the right to abortion.
Jackson Women’s Health – the state’s sole abortion provider – sued to block the Mississippi law soon after it passed. A federal judge ruled against the state and that decision was upheld by the U.S. Fifth Circuit Court of Appeals, which also issued a permanent injunction against the law. The Supreme Court in May 2021 agreed to take Mississippi’s appeal.
Earlier in November, the Supreme Court heard arguments in two cases challenging a restrictive Texas law, Whole Woman’s Health v. Jackson and U.S. v. Texas. The justices seemed receptive to the idea that the law, SB 8, was unconstitutional. But the court did not grant a request by the Biden administration to halt the law while the challenges made their way through the courts.
A version of this article first appeared on WebMD.com.
The justices heard from lawyers arguing for and against a 2018 Mississippi law that, with few exceptions, bans abortion after 15 weeks, claiming that a fetus is viable outside the womb at that age. The Supreme Court’s 1973 Roe v. Wade decision and legal rulings in the decades since, including the 1992 decision in Planned Parenthood v. Casey, have said that abortion should be available to the point of viability – established as about 23 weeks.
The court also ruled in Casey that state laws could not present an “undue burden” on a woman’s ability to obtain an abortion.
The Mississippi attorney general did not initially seek to overturn Roe and Casey, but later argued in Dobbs v. Jackson Women’s Health Organization that both cases were erroneously decided and should be completely thrown out.
“It is an egregiously wrong decision that has inflicted tremendous damage on our country and will continue to do so and take innumerable human lives unless and until this court overrules it,” said Scott G. Stewart, Mississippi’s solicitor general.
When it accepted the Mississippi case, the Supreme Court did not agree to weigh in on overturning Roe or Casey, but the justices’ leanings were evident during the hearing, and it is possible they would throw out those landmark cases.
Justice Clarence Thomas asked repeatedly for the law’s challengers to point out where the right to an abortion was written in the Constitution, as did Justice Samuel Alito.
“If we were talking about the Second Amendment, I know exactly what we’re talking about, if we’re talking about the Fourth Amendment, I know what we’re talking about, because it’s written, it’s there,” said Justice Thomas. “What specifically is the right here that we’re talking about?” he asked U.S. Solicitor General Elizabeth Prelogar.
She said the right to abortion was embedded in the 14th amendment’s guarantee of the pursuit of liberty.
“If this Court renounces the liberty interest recognized in Roe and reaffirmed in Casey, it would be an unprecedented contraction of individual rights,” and a departure from court doctrine of upholding precedent, known as stare decisis, she said.
Chief Justice John Roberts seemed to be against throwing out either of the landmark abortion cases, but instead wanted to focus on whether the 15 weeks was a reasonable time point. But he seemed to be alone in honing-in on that issue.
“Roberts seem desperate for some limiting principle that isn’t reversing Roe, and none of the other conservative justices are biting,” tweeted Mary Ziegler, a historian who has written about abortion.
But justices Neil Gorsuch, Amy Coney Barrett, and Brett Kavanaugh all appeared to be receptive to the idea that the prior precedent set by Roe and Casey could be overturned.
Neil Katyal, the former U.S. acting solicitor general and a Supreme Court lawyer, tweeted during the arguments that he saw “nothing so far sympathetic to the challengers. And a lot that has been very hostile.”
He cautioned that questions during oral arguments “often are just trying to understand a lawyer’s position,” adding, “But the tea leaves here are ominous.”
If Roe v. Wade is overturned, 22 states have laws already on the books that could be used to restrict abortion, according to the Guttmacher Institute. Almost all abortions would be banned in 12 states that have so-called “trigger” laws: Arkansas, Idaho, Kentucky, Louisiana, Mississippi, Missouri, North Dakota, Oklahoma, South Dakota, Tennessee, Texas, and Utah.
Seventeen states have abortion restrictions that have been unenforced or blocked by courts that would go back into effect if Roe is nullified. An additional seven states have laws that intend to restrict abortion in the absence of Roe and four states have passed constitutional amendments to specifically not protect the right to abortion.
Guttmacher reports that 15 states and the District of Columbia have passed laws that protect the right to abortion.
Jackson Women’s Health – the state’s sole abortion provider – sued to block the Mississippi law soon after it passed. A federal judge ruled against the state and that decision was upheld by the U.S. Fifth Circuit Court of Appeals, which also issued a permanent injunction against the law. The Supreme Court in May 2021 agreed to take Mississippi’s appeal.
Earlier in November, the Supreme Court heard arguments in two cases challenging a restrictive Texas law, Whole Woman’s Health v. Jackson and U.S. v. Texas. The justices seemed receptive to the idea that the law, SB 8, was unconstitutional. But the court did not grant a request by the Biden administration to halt the law while the challenges made their way through the courts.
A version of this article first appeared on WebMD.com.
IUDs may increase background enhancement on breast MRI
Intrauterine contraceptive devices (IUDs) have been linked to increased background enhancement on breast MRI, according to research presented at the Radiological Society of North America 2021 annual meeting.
About 10.4% of women 15-49 years of age who use contraception have an IUD or contraceptive implant, according to the Centers for Disease Control and Prevention. Unlike oral or transdermal hormonal contraceptives and hormone replacement therapy, levonorgestrel-releasing IUDs release a small amount of the hormone directly into the uterus and are thought to have a much more localized effect, Luisa Huck, MD, the lead author of the study, said in an interview.
But women with IUDs have long reported adverse effects associated with other hormonal medication. “In the past, some women reported depression, headaches, sleep disorders, and panic attacks,” noted Dr. Huck, a radiology resident at RWTH Aachen University in Germany.
Christiane Kuhl, MD, chief of the department of radiology at RWTH Aachen University and senior author of the research, had also observed that women with hormonal IUDs often have increased background parenchymal enhancement (BPE) on contrast-enhanced MRI. BPE “has been established as a sensitive marker of hormonal stimulation of breast,” the study authors wrote, and previous studies have shown that women using hormonal medications have higher BPE on breast MRIs.
To better understand whether IUDs can increase BPE, Dr. Huck and colleagues used the hospital database to search for premenopausal women who had undergone breast MRIs for screening between January 2014 and July 2020. To be included, women had to have had at least two scans: one with and one without an IUD in place, with the scan conducted at least 4 weeks after IUD placement or removal. All women in the study had no history of breast cancer or hormone or antihormone intake.
The study involved 48 women with an average age of 45 years and a median of 27 months between the two scans. Forty-six of the women had the Mirena levonorgestrel-releasing IUD and two had the Jaydess IUD. To account for hormone variations between patients, the researchers used each patient as their own reference point. To control for age-related effects, 25 women had their first MRI without an IUD and their second scan with an IUD in place. The second group of 23 women underwent their first MRI with an IUD and had it removed before the second scan.
Hormonal effects on breast enhancement are very complex, and hormonal stimulation is not always predictably correlated with changes on MRI imaging.
For 23 women in the study, background enhancement was higher on scans with the IUD than without (P < .001). For 24 women, there was no change in BPE with or without an IUD, and one woman had lower BPE with an IUD than without.
“It is very interesting and relevant to practice to consider that the presence of an intrauterine device would have potential impact on the enhancement we see in the breast on MRI imaging,” Samantha Heller, MD, PhD, associate professor of radiology at New York University, said in an interview.
However, the study used BPE as a measure for hormonal shifts, and “hormonal effects on breast enhancement are very complex, and hormonal stimulation is not always predictably correlated with changes on MRI imaging,” she noted. BPE on MRI can fluctuate, so testing actual hormone levels in patients with elevated BPE could be helpful to identify hormonal shifts, she added. It is also important to understand why half of the women in the study showed no variation in BPE, she said.
The study findings are not very surprising, considering that it is known that low levels of progesterone from IUDs circulate in the blood stream, Frances Casey, MD, MPH, associate professor in the department of obstetrics and gynecology at Virginia Commonwealth University in Richmond, said in an interview. They do not suggest that there should be any changes to IUD guidelines, she added.
However, “the study findings raise the question as to whether IUD status should be documented as a matter of course prior to performing breast MRI,” said Dr. Heller. “It is standard to document the timing of a woman’s menstrual cycle, as well as to note any hormone suppression or replacement therapy. This is in part so that the radiologist may understand the etiology of any observed variation in background enhancement,” she explained.
Although increased enhancement on MRI has sometimes been linked to higher chances of recommendations for additional imaging or biopsies, she noted, “more work would be needed to understand the impact – if any – of an IUD on breast MRI recommendations due to enhancement changes.”
Dr. Huck, Dr. Heller, and Dr. Casey disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Intrauterine contraceptive devices (IUDs) have been linked to increased background enhancement on breast MRI, according to research presented at the Radiological Society of North America 2021 annual meeting.
About 10.4% of women 15-49 years of age who use contraception have an IUD or contraceptive implant, according to the Centers for Disease Control and Prevention. Unlike oral or transdermal hormonal contraceptives and hormone replacement therapy, levonorgestrel-releasing IUDs release a small amount of the hormone directly into the uterus and are thought to have a much more localized effect, Luisa Huck, MD, the lead author of the study, said in an interview.
But women with IUDs have long reported adverse effects associated with other hormonal medication. “In the past, some women reported depression, headaches, sleep disorders, and panic attacks,” noted Dr. Huck, a radiology resident at RWTH Aachen University in Germany.
Christiane Kuhl, MD, chief of the department of radiology at RWTH Aachen University and senior author of the research, had also observed that women with hormonal IUDs often have increased background parenchymal enhancement (BPE) on contrast-enhanced MRI. BPE “has been established as a sensitive marker of hormonal stimulation of breast,” the study authors wrote, and previous studies have shown that women using hormonal medications have higher BPE on breast MRIs.
To better understand whether IUDs can increase BPE, Dr. Huck and colleagues used the hospital database to search for premenopausal women who had undergone breast MRIs for screening between January 2014 and July 2020. To be included, women had to have had at least two scans: one with and one without an IUD in place, with the scan conducted at least 4 weeks after IUD placement or removal. All women in the study had no history of breast cancer or hormone or antihormone intake.
The study involved 48 women with an average age of 45 years and a median of 27 months between the two scans. Forty-six of the women had the Mirena levonorgestrel-releasing IUD and two had the Jaydess IUD. To account for hormone variations between patients, the researchers used each patient as their own reference point. To control for age-related effects, 25 women had their first MRI without an IUD and their second scan with an IUD in place. The second group of 23 women underwent their first MRI with an IUD and had it removed before the second scan.
Hormonal effects on breast enhancement are very complex, and hormonal stimulation is not always predictably correlated with changes on MRI imaging.
For 23 women in the study, background enhancement was higher on scans with the IUD than without (P < .001). For 24 women, there was no change in BPE with or without an IUD, and one woman had lower BPE with an IUD than without.
“It is very interesting and relevant to practice to consider that the presence of an intrauterine device would have potential impact on the enhancement we see in the breast on MRI imaging,” Samantha Heller, MD, PhD, associate professor of radiology at New York University, said in an interview.
However, the study used BPE as a measure for hormonal shifts, and “hormonal effects on breast enhancement are very complex, and hormonal stimulation is not always predictably correlated with changes on MRI imaging,” she noted. BPE on MRI can fluctuate, so testing actual hormone levels in patients with elevated BPE could be helpful to identify hormonal shifts, she added. It is also important to understand why half of the women in the study showed no variation in BPE, she said.
The study findings are not very surprising, considering that it is known that low levels of progesterone from IUDs circulate in the blood stream, Frances Casey, MD, MPH, associate professor in the department of obstetrics and gynecology at Virginia Commonwealth University in Richmond, said in an interview. They do not suggest that there should be any changes to IUD guidelines, she added.
However, “the study findings raise the question as to whether IUD status should be documented as a matter of course prior to performing breast MRI,” said Dr. Heller. “It is standard to document the timing of a woman’s menstrual cycle, as well as to note any hormone suppression or replacement therapy. This is in part so that the radiologist may understand the etiology of any observed variation in background enhancement,” she explained.
Although increased enhancement on MRI has sometimes been linked to higher chances of recommendations for additional imaging or biopsies, she noted, “more work would be needed to understand the impact – if any – of an IUD on breast MRI recommendations due to enhancement changes.”
Dr. Huck, Dr. Heller, and Dr. Casey disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Intrauterine contraceptive devices (IUDs) have been linked to increased background enhancement on breast MRI, according to research presented at the Radiological Society of North America 2021 annual meeting.
About 10.4% of women 15-49 years of age who use contraception have an IUD or contraceptive implant, according to the Centers for Disease Control and Prevention. Unlike oral or transdermal hormonal contraceptives and hormone replacement therapy, levonorgestrel-releasing IUDs release a small amount of the hormone directly into the uterus and are thought to have a much more localized effect, Luisa Huck, MD, the lead author of the study, said in an interview.
But women with IUDs have long reported adverse effects associated with other hormonal medication. “In the past, some women reported depression, headaches, sleep disorders, and panic attacks,” noted Dr. Huck, a radiology resident at RWTH Aachen University in Germany.
Christiane Kuhl, MD, chief of the department of radiology at RWTH Aachen University and senior author of the research, had also observed that women with hormonal IUDs often have increased background parenchymal enhancement (BPE) on contrast-enhanced MRI. BPE “has been established as a sensitive marker of hormonal stimulation of breast,” the study authors wrote, and previous studies have shown that women using hormonal medications have higher BPE on breast MRIs.
To better understand whether IUDs can increase BPE, Dr. Huck and colleagues used the hospital database to search for premenopausal women who had undergone breast MRIs for screening between January 2014 and July 2020. To be included, women had to have had at least two scans: one with and one without an IUD in place, with the scan conducted at least 4 weeks after IUD placement or removal. All women in the study had no history of breast cancer or hormone or antihormone intake.
The study involved 48 women with an average age of 45 years and a median of 27 months between the two scans. Forty-six of the women had the Mirena levonorgestrel-releasing IUD and two had the Jaydess IUD. To account for hormone variations between patients, the researchers used each patient as their own reference point. To control for age-related effects, 25 women had their first MRI without an IUD and their second scan with an IUD in place. The second group of 23 women underwent their first MRI with an IUD and had it removed before the second scan.
Hormonal effects on breast enhancement are very complex, and hormonal stimulation is not always predictably correlated with changes on MRI imaging.
For 23 women in the study, background enhancement was higher on scans with the IUD than without (P < .001). For 24 women, there was no change in BPE with or without an IUD, and one woman had lower BPE with an IUD than without.
“It is very interesting and relevant to practice to consider that the presence of an intrauterine device would have potential impact on the enhancement we see in the breast on MRI imaging,” Samantha Heller, MD, PhD, associate professor of radiology at New York University, said in an interview.
However, the study used BPE as a measure for hormonal shifts, and “hormonal effects on breast enhancement are very complex, and hormonal stimulation is not always predictably correlated with changes on MRI imaging,” she noted. BPE on MRI can fluctuate, so testing actual hormone levels in patients with elevated BPE could be helpful to identify hormonal shifts, she added. It is also important to understand why half of the women in the study showed no variation in BPE, she said.
The study findings are not very surprising, considering that it is known that low levels of progesterone from IUDs circulate in the blood stream, Frances Casey, MD, MPH, associate professor in the department of obstetrics and gynecology at Virginia Commonwealth University in Richmond, said in an interview. They do not suggest that there should be any changes to IUD guidelines, she added.
However, “the study findings raise the question as to whether IUD status should be documented as a matter of course prior to performing breast MRI,” said Dr. Heller. “It is standard to document the timing of a woman’s menstrual cycle, as well as to note any hormone suppression or replacement therapy. This is in part so that the radiologist may understand the etiology of any observed variation in background enhancement,” she explained.
Although increased enhancement on MRI has sometimes been linked to higher chances of recommendations for additional imaging or biopsies, she noted, “more work would be needed to understand the impact – if any – of an IUD on breast MRI recommendations due to enhancement changes.”
Dr. Huck, Dr. Heller, and Dr. Casey disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Sickle cell raises risk for stillbirth
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
Both sickle cell trait and sickle cell disease were significantly associated with an increased risk of stillbirth, based on data from more than 50,000 women.
Pregnant women with sickle cell disease (SCD) are at increased risk of complications, including stillbirth, but many women with the disease in the United States lack access to specialty care, Silvia P. Canelón, PhD, of the University of Pennsylvania, Philadelphia, and colleagues wrote. Sickle cell trait (SCT), defined as one abnormal allele of the hemoglobin gene, is not considered a disease state because many carriers are asymptomatic, and therefore even less likely to be assessed for potential complications. “However, it is possible for people with SCT to experience sickling of red blood cells under severe hypoxia, dehydration, and hyperthermia. This condition can lead to severe medical complications for sickle cell carriers, including fetal loss, splenic infarction, exercise-related sudden death, and others,” they noted.
In a study published in JAMA Network Open, the researchers reviewed data from 63,334 deliveries in 50,560 women between Jan. 1, 2010, and Aug. 15, 2017, at four quaternary academic medical centers in Pennsylvania. Of these, 1,904 had SCT but not SCD, and 164 had SCD. The mean age of the women was 29.5 years, and approximately 56% were single at the time of delivery. A majority (87%) of the study population was Rhesus-factor positive, 47.0% were Black or African American, 33.7% were White, and 45.2% had ABO blood type O.
Risk factors for stillbirth used in the analysis included SCD, numbers of pain crises and blood transfusions before delivery, delivery episode (to represent parity), history of cesarean delivery, multiple gestation, age, marital status, race and ethnicity, ABO blood type, Rhesus factor, and year of delivery.
Overall, the prevalence of stillbirth in women with SCT was 1.1%, compared with 0.8% in the general study population, and was significantly associated with increased risk of stillbirth after controlling for multiple risk factors. The adjusted odds ratio was 8.94 for stillbirth risk in women with SCT, compared with women without SCT (P = .045), although the risk was greater among women with SCD, compared with those without SCD (aOR, 26.40).
“In addition, the stratified analysis found Black or African American patients with SCD to be at higher risk of stillbirth, compared with Black or African American patients without SCD (aOR, 3.59),” but no significant association was noted between stillbirth and SCT, the researchers wrote. Stillbirth rates were 1.1% in Black or African American women overall, 2.7% in those with SCD, and 1.0% in those with SCT. Overall, multiple gestation was associated with an increased risk of stillbirth (aOR, 4.68), while a history of cesarean delivery and being married at the time of delivery were associated with decreased risk (aOR, 0.44 and 0.72, respectively).
The lack of association between stillbirth and SCT in Black or African American patients supports some previous research, but contradicts other studies, the researchers wrote. “Ultimately, it may be impossible to disentangle the risks due to the disease and those due to disparities associated with the disease that have resulted from longstanding inequity and stigma,” they said. The findings also suggest that biological mechanisms of SCT may contribute to severe clinical complications, and therefore “invite a more critical examination of the assumption that SCT is not a disease state.”
The study findings were limited by several factors including the lack of assessment of SCT independent of other comorbidities, such as hypertension, preeclampsia, diabetes, and obesity, and by the use of billing codes that could misclassify patients, the researchers noted.
However, the results support some findings from previous studies of the potential health complications for pregnant SCT patients. The large study population highlights the need to identify women’s SCT status during obstetric care, and to provide both pregnancy guidance for SCT patients and systemic support of comprehensive care for SCD and SCT patients, they concluded.
Disparities may drive stillbirth in sickle cell trait women
“There is a paucity of research evaluating sickle cell trait and the risk of adverse pregnancy outcomes such as stillbirth,” Iris Krishna, MD, of Emory University, Atlanta, said in an interview. “Prior studies evaluating the risk of stillbirth have yielded mixed results, and an increased risk of stillbirth in women with sickle cell trait has not been established. This study is unique in that it attempts to address how racial inequities and health disparities may contribute to risk of stillbirth in women with sickle cell trait.”
Although the study findings suggest an increased risk of stillbirth in women with sickle cell trait, an analysis stratified for Black or African American patients showed no association, Dr. Krishna said. “The prevalence of stillbirth was noted to be 1% among Black or African American patients with sickle cell trait compared to the prevalence of stillbirth of 1.1% among Black or African American women with no sickle cell trait or disease. Although, sickle cell trait or sickle cell disease can be found in any racial or ethnic group, it disproportionately affects Black or African Americans, with a sickle cell trait carrier rate of approximately 1 in 10. The mixed findings in this study amongst racial/ethnic groups further suggest that there is more research needed before an association between stillbirth and sickle cell trait can be supported.”
As for clinical implications, “it is well established that for women with sickle cell trait there is an increased risk of urinary tract infections in pregnancy,” said Dr. Krishna. “Women with sickle cell trait should have a urine culture performed at their first prenatal visit and each trimester. At this time, studies evaluating risk of stillbirth in women with sickle cell trait have yielded conflicting results, and current consensus is that women with sickle cell trait are not at increased risk. In comparison, women with sickle cell disease are at increased risk for stillbirth and adverse pregnancy outcomes. Women with sickle cell disease should be followed closely during pregnancy and fetal surveillance implemented at 32 weeks, if not sooner, to reduce risk of stillbirth.
“Prior studies evaluating risk of stillbirth in women with sickle cell trait consist of retrospective cohorts with small study populations,” Dr. Krishna added. Notably, the current study was limited by the inability to adjust for comorbidities including diabetes, hypertension, and obesity, that are not only associated with an increased risk for stillbirth, but also disproportionately common among Black women.
“More studies are needed evaluating the relationship between these comorbidities as well as studies specifically evaluating how race affects care and pregnancy outcomes,” Dr. Krisha emphasized.
The study was funded by the University of Pennsylvania department of biostatistics, epidemiology, and informatics. Lead author Dr. Canelón disclosed grants from the Centers for Disease Control and Prevention, Clinical and Translational Science Awards, and grants from the National Institutes of Health outside the submitted work. Dr. Krishna had no financial conflicts to disclose, but serves on the editorial advisory board of Ob.Gyn News.
FROM JAMA NETWORK OPEN
FDA approves imaging drug for detecting ovarian cancer lesions
The new drug “is designed to improve the ability to locate additional ovarian cancerous tissue that is normally difficult to detect during surgery,” according to the agency.
Pafolacianine, administered via intravenous injection prior to surgery, is the first FDA-approved tumor-targeted fluorescent agent for ovarian cancer.
In a press statement, drug inventor Philip Low, PhD, of Purdue University in West Lafayette, Ind., said the agent causes ovarian cancer lesions to “light up like stars against a night sky.”
Improving detection of ovarian cancer lesions is critical given that ovarian cancer is one of the “deadliest of all female reproductive system cancers,” according to the American Cancer Society. The organization estimates that there will be more than 21,000 new cases and more than 13,000 deaths in 2021.
Currently, surgeons use preoperative imaging as well as visual inspection of tumors under normal light and examination by touch to identify ovarian cancer lesions.
Pafolacianine offers a new tool to enhance surgeons’ ability “to identify deadly ovarian tumors that may otherwise go undetected,” Alex Gorovets, MD, deputy director of the office of specialty medicine in the FDA’s Center for Drug Evaluation and Research, said in a press statement.
Ovarian cancer often causes the body to overproduce the folate receptor protein in cell membranes. Pafolacianine, employed with a near-infrared fluorescence imaging system cleared by the FDA for use alongside the drug, binds to and illuminates these proteins under fluorescent light, “boosting surgeons’ ability to identify the cancerous tissue,” the agency in a statement.
The safety and effectiveness of pafolacianine was evaluated in a randomized, multi-center, open-label study of women diagnosed with ovarian cancer or with high clinical suspicion of ovarian cancer. Of the 134 women undergoing surgery who received a dose of pafolacianine and were evaluated under both normal and fluorescent light, 26.9% had at least one cancerous lesion detected that was not observed by standard visual or tactile inspection.
The most common side effects of pafolacianine were infusion-related reactions, including nausea, vomiting, abdominal pain, flushing, dyspepsia, chest discomfort, itching, and hypersensitivity.
Pafolacianine may cause fetal harm when administered to a pregnant woman. The use of folate, folic acid, or folate-containing supplements should be avoided within 48 hours before administration of pafolacianine.
The FDA also cautioned about the possible risk of image interpretation errors, including false negatives and false positives, with the use of the new drug and near-infrared fluorescence imaging system.
The FDA previously granted pafolacianine orphan-drug, priority, and fast track designations.
A version of this article first appeared on Medscape.com.
The new drug “is designed to improve the ability to locate additional ovarian cancerous tissue that is normally difficult to detect during surgery,” according to the agency.
Pafolacianine, administered via intravenous injection prior to surgery, is the first FDA-approved tumor-targeted fluorescent agent for ovarian cancer.
In a press statement, drug inventor Philip Low, PhD, of Purdue University in West Lafayette, Ind., said the agent causes ovarian cancer lesions to “light up like stars against a night sky.”
Improving detection of ovarian cancer lesions is critical given that ovarian cancer is one of the “deadliest of all female reproductive system cancers,” according to the American Cancer Society. The organization estimates that there will be more than 21,000 new cases and more than 13,000 deaths in 2021.
Currently, surgeons use preoperative imaging as well as visual inspection of tumors under normal light and examination by touch to identify ovarian cancer lesions.
Pafolacianine offers a new tool to enhance surgeons’ ability “to identify deadly ovarian tumors that may otherwise go undetected,” Alex Gorovets, MD, deputy director of the office of specialty medicine in the FDA’s Center for Drug Evaluation and Research, said in a press statement.
Ovarian cancer often causes the body to overproduce the folate receptor protein in cell membranes. Pafolacianine, employed with a near-infrared fluorescence imaging system cleared by the FDA for use alongside the drug, binds to and illuminates these proteins under fluorescent light, “boosting surgeons’ ability to identify the cancerous tissue,” the agency in a statement.
The safety and effectiveness of pafolacianine was evaluated in a randomized, multi-center, open-label study of women diagnosed with ovarian cancer or with high clinical suspicion of ovarian cancer. Of the 134 women undergoing surgery who received a dose of pafolacianine and were evaluated under both normal and fluorescent light, 26.9% had at least one cancerous lesion detected that was not observed by standard visual or tactile inspection.
The most common side effects of pafolacianine were infusion-related reactions, including nausea, vomiting, abdominal pain, flushing, dyspepsia, chest discomfort, itching, and hypersensitivity.
Pafolacianine may cause fetal harm when administered to a pregnant woman. The use of folate, folic acid, or folate-containing supplements should be avoided within 48 hours before administration of pafolacianine.
The FDA also cautioned about the possible risk of image interpretation errors, including false negatives and false positives, with the use of the new drug and near-infrared fluorescence imaging system.
The FDA previously granted pafolacianine orphan-drug, priority, and fast track designations.
A version of this article first appeared on Medscape.com.
The new drug “is designed to improve the ability to locate additional ovarian cancerous tissue that is normally difficult to detect during surgery,” according to the agency.
Pafolacianine, administered via intravenous injection prior to surgery, is the first FDA-approved tumor-targeted fluorescent agent for ovarian cancer.
In a press statement, drug inventor Philip Low, PhD, of Purdue University in West Lafayette, Ind., said the agent causes ovarian cancer lesions to “light up like stars against a night sky.”
Improving detection of ovarian cancer lesions is critical given that ovarian cancer is one of the “deadliest of all female reproductive system cancers,” according to the American Cancer Society. The organization estimates that there will be more than 21,000 new cases and more than 13,000 deaths in 2021.
Currently, surgeons use preoperative imaging as well as visual inspection of tumors under normal light and examination by touch to identify ovarian cancer lesions.
Pafolacianine offers a new tool to enhance surgeons’ ability “to identify deadly ovarian tumors that may otherwise go undetected,” Alex Gorovets, MD, deputy director of the office of specialty medicine in the FDA’s Center for Drug Evaluation and Research, said in a press statement.
Ovarian cancer often causes the body to overproduce the folate receptor protein in cell membranes. Pafolacianine, employed with a near-infrared fluorescence imaging system cleared by the FDA for use alongside the drug, binds to and illuminates these proteins under fluorescent light, “boosting surgeons’ ability to identify the cancerous tissue,” the agency in a statement.
The safety and effectiveness of pafolacianine was evaluated in a randomized, multi-center, open-label study of women diagnosed with ovarian cancer or with high clinical suspicion of ovarian cancer. Of the 134 women undergoing surgery who received a dose of pafolacianine and were evaluated under both normal and fluorescent light, 26.9% had at least one cancerous lesion detected that was not observed by standard visual or tactile inspection.
The most common side effects of pafolacianine were infusion-related reactions, including nausea, vomiting, abdominal pain, flushing, dyspepsia, chest discomfort, itching, and hypersensitivity.
Pafolacianine may cause fetal harm when administered to a pregnant woman. The use of folate, folic acid, or folate-containing supplements should be avoided within 48 hours before administration of pafolacianine.
The FDA also cautioned about the possible risk of image interpretation errors, including false negatives and false positives, with the use of the new drug and near-infrared fluorescence imaging system.
The FDA previously granted pafolacianine orphan-drug, priority, and fast track designations.
A version of this article first appeared on Medscape.com.
Congress OKs Veterans Affairs Expansive New Maternal Care Program
It’s called the Momnibus—the Black Maternal Health Momnibus Act of 2021 (HR 959) with 12 bills addressing “every dimension of the maternal health crisis in America.” The first bill in the Momnibus to pass Congress is the Protecting Moms Who Served act, which sets up a $15 million maternal care program within the US Department of Veterans Affairs (VA). “There has never been a comprehensive evaluation of how our nation’s growing maternal mortality crisis is impacting our women veterans, even though they may be at higher risk due to their service,” said Sen. Tammy Duckworth (D-IL), a co-sponsor of the Momnibus. The bill has passed Congress and awaits President Biden’s signature.
Rep. Lauren Underwood (D-IL) along with Rep. Alma Adams (D- NC-12), Sen. Cory Booker D-NJ), and members of the Black Maternal Health Caucus reintroduced the bill (first introduced last year). According to Rep. Underwood, the act would codify and strengthen the VA maternity care coordination programs. It also will require the US Government Accountability Office to report the deaths of pregnant and postpartum veterans and to focus on any racial or ethnic disparities. The bill passed overwhelmingly, 414 to 9 and awaits President Biden’s signature.
The Momnibus’s cute name represents a very serious purpose. “Maternal mortality has historically been used as a key indicator of the health of a population,” say researchers from National Vital Statistics Reports. But American mothers are dying at the highest rate in the developed world, and the numbers have been rising dramatically. Between 1987, when the Centers for Disease Control and Prevention (CDC) launched the Pregnancy Mortality Surveillance System in 2017, the latest year for available data, the number of reported pregnancy-related deaths in the United States rose steadily from 7.2 deaths per 100,000 live births to 17.3 per 100,000.
The maternal morbidity crisis is particularly stark among certain groups of women. Black women are acutely at risk, dying at 3 to 4 times the rate of White women (41.7 deaths per 100,000 live births), and one-third higher than the next highest risk group, Native American women (28.3 deaths per 100,000 live births).
But just how accurate have the data been? The study published in National Vital Statistics Report found that using a checkbox for “cause of death” specifying maternal death identified more than triple the number of maternal deaths. Without the checkbox item, maternal mortality rates in 2015 and 2016 would have been reported as 8.7 deaths per 100,000 live births, compared with 8.9 in 2002. With the checkbox, the rate would be reported as 20.9 per 100,000 live births in 2015 and 21.8/100,000 in 2016.
The CDC states that the reasons for the rising numbers are unclear; advances in identification have improved over time, for one. But by and large, the women are dying of preventable causes, such as hypertension, diabetes mellitus, and chronic heart disease. Nearly 60% of maternal deaths are deemed preventable.
Black and other minority women, though, may be dying of biases. Researchers from Beth Israel and Harvard cite studies that have found racial and ethnic disparities in obstetric care delivery. Non-Hispanic Blacks women, Hispanic women, and Asian women, for instance, have lower odds of labor induction when compared with that of White women. The odds of receiving an episiotomy are lower in non-Hispanic Black and Hispanic women. The Listening to Mothers survey III found that 24% of participants perceived discrimination during birth hospitalization, predominantly among Black or Hispanic women and uninsured women.
A maternal health equity advocacy group, 4Kira4Moms, was founded by the husband of Kira Johnson who died of hemorrhage following a routine scheduled cesarean section. In the recovery room, her catheter began turning pink with blood. For 10 hours, her husband said, he and her family begged the medical staff for help but were told his wife was not a priority. Thus, the Momnibus also contains the Kira Johnson Act, which will establish funding for community-based groups to provide Black pregnant women with more support.
Among other changes, the Momnibus will:
- Make critical investments in social determinants of health that influence maternal health outcomes, such as housing, transportation, and nutrition;
- Provide funding to community-based organizations that are working to improve maternal health outcomes and promote equity;
- Comprehensively study the unique maternal health risks facing pregnant and postpartum veterans and support VA maternity care coordination programs;
- Support mothers with mental health conditions and substance use disorders; and
- Promote innovative payment models to incentivize high-quality maternity care and nonclinical perinatal support
A variety of recent bills in Congress address maternal health. The Mothers and Offspring Mortality and Morbidity Awareness (MOMMA) Act, for instance, also would specifically address maternal health disparities by improving data collection and reporting, improving maternal care, and advancing respectful, equitable care. It also would extend Medicaid and the Children’s Health Insurance Program coverage. Katie Shea Barrett, MPH, executive director of March for Moms, a coalition of families, health care practitioners, policy makers, and partners advocating for mothers’ and families’ health, notes in an essay for thehill.com that Medicaid coverage ends about 60 days postpartum, although half of the maternal deaths happen between 42 days and 1 year postpartum.
She writes: “[W]e have to directly address the disproportionate impact of maternal mortality on women of color by training providers in offering care that is culturally competent and free of implicit bias. Health systems must be aware and respectful of cultural norms when providing care and be mindful of buying into stereotypes based on race, ethnicity, and even underlying medical conditions like diabetes, which often lead to perceived discrimination and perpetuate systems of injustice.”
In April, Vice President Kamala Harris called for sweeping action to curb racial inequities in pregnancy and childbirth. In an email Q&A with STAT, she said, “With every day that goes by and every woman who dies, the need for action grows more urgent.”
It’s called the Momnibus—the Black Maternal Health Momnibus Act of 2021 (HR 959) with 12 bills addressing “every dimension of the maternal health crisis in America.” The first bill in the Momnibus to pass Congress is the Protecting Moms Who Served act, which sets up a $15 million maternal care program within the US Department of Veterans Affairs (VA). “There has never been a comprehensive evaluation of how our nation’s growing maternal mortality crisis is impacting our women veterans, even though they may be at higher risk due to their service,” said Sen. Tammy Duckworth (D-IL), a co-sponsor of the Momnibus. The bill has passed Congress and awaits President Biden’s signature.
Rep. Lauren Underwood (D-IL) along with Rep. Alma Adams (D- NC-12), Sen. Cory Booker D-NJ), and members of the Black Maternal Health Caucus reintroduced the bill (first introduced last year). According to Rep. Underwood, the act would codify and strengthen the VA maternity care coordination programs. It also will require the US Government Accountability Office to report the deaths of pregnant and postpartum veterans and to focus on any racial or ethnic disparities. The bill passed overwhelmingly, 414 to 9 and awaits President Biden’s signature.
The Momnibus’s cute name represents a very serious purpose. “Maternal mortality has historically been used as a key indicator of the health of a population,” say researchers from National Vital Statistics Reports. But American mothers are dying at the highest rate in the developed world, and the numbers have been rising dramatically. Between 1987, when the Centers for Disease Control and Prevention (CDC) launched the Pregnancy Mortality Surveillance System in 2017, the latest year for available data, the number of reported pregnancy-related deaths in the United States rose steadily from 7.2 deaths per 100,000 live births to 17.3 per 100,000.
The maternal morbidity crisis is particularly stark among certain groups of women. Black women are acutely at risk, dying at 3 to 4 times the rate of White women (41.7 deaths per 100,000 live births), and one-third higher than the next highest risk group, Native American women (28.3 deaths per 100,000 live births).
But just how accurate have the data been? The study published in National Vital Statistics Report found that using a checkbox for “cause of death” specifying maternal death identified more than triple the number of maternal deaths. Without the checkbox item, maternal mortality rates in 2015 and 2016 would have been reported as 8.7 deaths per 100,000 live births, compared with 8.9 in 2002. With the checkbox, the rate would be reported as 20.9 per 100,000 live births in 2015 and 21.8/100,000 in 2016.
The CDC states that the reasons for the rising numbers are unclear; advances in identification have improved over time, for one. But by and large, the women are dying of preventable causes, such as hypertension, diabetes mellitus, and chronic heart disease. Nearly 60% of maternal deaths are deemed preventable.
Black and other minority women, though, may be dying of biases. Researchers from Beth Israel and Harvard cite studies that have found racial and ethnic disparities in obstetric care delivery. Non-Hispanic Blacks women, Hispanic women, and Asian women, for instance, have lower odds of labor induction when compared with that of White women. The odds of receiving an episiotomy are lower in non-Hispanic Black and Hispanic women. The Listening to Mothers survey III found that 24% of participants perceived discrimination during birth hospitalization, predominantly among Black or Hispanic women and uninsured women.
A maternal health equity advocacy group, 4Kira4Moms, was founded by the husband of Kira Johnson who died of hemorrhage following a routine scheduled cesarean section. In the recovery room, her catheter began turning pink with blood. For 10 hours, her husband said, he and her family begged the medical staff for help but were told his wife was not a priority. Thus, the Momnibus also contains the Kira Johnson Act, which will establish funding for community-based groups to provide Black pregnant women with more support.
Among other changes, the Momnibus will:
- Make critical investments in social determinants of health that influence maternal health outcomes, such as housing, transportation, and nutrition;
- Provide funding to community-based organizations that are working to improve maternal health outcomes and promote equity;
- Comprehensively study the unique maternal health risks facing pregnant and postpartum veterans and support VA maternity care coordination programs;
- Support mothers with mental health conditions and substance use disorders; and
- Promote innovative payment models to incentivize high-quality maternity care and nonclinical perinatal support
A variety of recent bills in Congress address maternal health. The Mothers and Offspring Mortality and Morbidity Awareness (MOMMA) Act, for instance, also would specifically address maternal health disparities by improving data collection and reporting, improving maternal care, and advancing respectful, equitable care. It also would extend Medicaid and the Children’s Health Insurance Program coverage. Katie Shea Barrett, MPH, executive director of March for Moms, a coalition of families, health care practitioners, policy makers, and partners advocating for mothers’ and families’ health, notes in an essay for thehill.com that Medicaid coverage ends about 60 days postpartum, although half of the maternal deaths happen between 42 days and 1 year postpartum.
She writes: “[W]e have to directly address the disproportionate impact of maternal mortality on women of color by training providers in offering care that is culturally competent and free of implicit bias. Health systems must be aware and respectful of cultural norms when providing care and be mindful of buying into stereotypes based on race, ethnicity, and even underlying medical conditions like diabetes, which often lead to perceived discrimination and perpetuate systems of injustice.”
In April, Vice President Kamala Harris called for sweeping action to curb racial inequities in pregnancy and childbirth. In an email Q&A with STAT, she said, “With every day that goes by and every woman who dies, the need for action grows more urgent.”
It’s called the Momnibus—the Black Maternal Health Momnibus Act of 2021 (HR 959) with 12 bills addressing “every dimension of the maternal health crisis in America.” The first bill in the Momnibus to pass Congress is the Protecting Moms Who Served act, which sets up a $15 million maternal care program within the US Department of Veterans Affairs (VA). “There has never been a comprehensive evaluation of how our nation’s growing maternal mortality crisis is impacting our women veterans, even though they may be at higher risk due to their service,” said Sen. Tammy Duckworth (D-IL), a co-sponsor of the Momnibus. The bill has passed Congress and awaits President Biden’s signature.
Rep. Lauren Underwood (D-IL) along with Rep. Alma Adams (D- NC-12), Sen. Cory Booker D-NJ), and members of the Black Maternal Health Caucus reintroduced the bill (first introduced last year). According to Rep. Underwood, the act would codify and strengthen the VA maternity care coordination programs. It also will require the US Government Accountability Office to report the deaths of pregnant and postpartum veterans and to focus on any racial or ethnic disparities. The bill passed overwhelmingly, 414 to 9 and awaits President Biden’s signature.
The Momnibus’s cute name represents a very serious purpose. “Maternal mortality has historically been used as a key indicator of the health of a population,” say researchers from National Vital Statistics Reports. But American mothers are dying at the highest rate in the developed world, and the numbers have been rising dramatically. Between 1987, when the Centers for Disease Control and Prevention (CDC) launched the Pregnancy Mortality Surveillance System in 2017, the latest year for available data, the number of reported pregnancy-related deaths in the United States rose steadily from 7.2 deaths per 100,000 live births to 17.3 per 100,000.
The maternal morbidity crisis is particularly stark among certain groups of women. Black women are acutely at risk, dying at 3 to 4 times the rate of White women (41.7 deaths per 100,000 live births), and one-third higher than the next highest risk group, Native American women (28.3 deaths per 100,000 live births).
But just how accurate have the data been? The study published in National Vital Statistics Report found that using a checkbox for “cause of death” specifying maternal death identified more than triple the number of maternal deaths. Without the checkbox item, maternal mortality rates in 2015 and 2016 would have been reported as 8.7 deaths per 100,000 live births, compared with 8.9 in 2002. With the checkbox, the rate would be reported as 20.9 per 100,000 live births in 2015 and 21.8/100,000 in 2016.
The CDC states that the reasons for the rising numbers are unclear; advances in identification have improved over time, for one. But by and large, the women are dying of preventable causes, such as hypertension, diabetes mellitus, and chronic heart disease. Nearly 60% of maternal deaths are deemed preventable.
Black and other minority women, though, may be dying of biases. Researchers from Beth Israel and Harvard cite studies that have found racial and ethnic disparities in obstetric care delivery. Non-Hispanic Blacks women, Hispanic women, and Asian women, for instance, have lower odds of labor induction when compared with that of White women. The odds of receiving an episiotomy are lower in non-Hispanic Black and Hispanic women. The Listening to Mothers survey III found that 24% of participants perceived discrimination during birth hospitalization, predominantly among Black or Hispanic women and uninsured women.
A maternal health equity advocacy group, 4Kira4Moms, was founded by the husband of Kira Johnson who died of hemorrhage following a routine scheduled cesarean section. In the recovery room, her catheter began turning pink with blood. For 10 hours, her husband said, he and her family begged the medical staff for help but were told his wife was not a priority. Thus, the Momnibus also contains the Kira Johnson Act, which will establish funding for community-based groups to provide Black pregnant women with more support.
Among other changes, the Momnibus will:
- Make critical investments in social determinants of health that influence maternal health outcomes, such as housing, transportation, and nutrition;
- Provide funding to community-based organizations that are working to improve maternal health outcomes and promote equity;
- Comprehensively study the unique maternal health risks facing pregnant and postpartum veterans and support VA maternity care coordination programs;
- Support mothers with mental health conditions and substance use disorders; and
- Promote innovative payment models to incentivize high-quality maternity care and nonclinical perinatal support
A variety of recent bills in Congress address maternal health. The Mothers and Offspring Mortality and Morbidity Awareness (MOMMA) Act, for instance, also would specifically address maternal health disparities by improving data collection and reporting, improving maternal care, and advancing respectful, equitable care. It also would extend Medicaid and the Children’s Health Insurance Program coverage. Katie Shea Barrett, MPH, executive director of March for Moms, a coalition of families, health care practitioners, policy makers, and partners advocating for mothers’ and families’ health, notes in an essay for thehill.com that Medicaid coverage ends about 60 days postpartum, although half of the maternal deaths happen between 42 days and 1 year postpartum.
She writes: “[W]e have to directly address the disproportionate impact of maternal mortality on women of color by training providers in offering care that is culturally competent and free of implicit bias. Health systems must be aware and respectful of cultural norms when providing care and be mindful of buying into stereotypes based on race, ethnicity, and even underlying medical conditions like diabetes, which often lead to perceived discrimination and perpetuate systems of injustice.”
In April, Vice President Kamala Harris called for sweeping action to curb racial inequities in pregnancy and childbirth. In an email Q&A with STAT, she said, “With every day that goes by and every woman who dies, the need for action grows more urgent.”
Big drop in U.S. cervical cancer rates, mortality in younger women
The analysis adds to a growing body of evidence demonstrating vaccine-associated changes in cervical cancer incidence and mortality.
Previous data from the United Kingdom, published earlier in November, showed that cervical cancer rates were 87% lower among girls who received the HPV vaccine compared to previously unvaccinated generations. Based on the analysis, the authors concluded that the UK’s HPV immunization program “almost eliminated cervical cancer” in women born since September 1995.
The latest study, published Nov. 29 in JAMA Pediatrics , reports a 38% drop in cervical cancer incidence and a 43% decline in mortality among young women and girls after HPV vaccination was introduced in the United States.
“These results are encouraging,” Peter Sasieni, MD, of King’s College London, and senior author on the U.K. study, told this news organization in an email.
The difference in incidence rates between the U.K. and U.S. studies, Dr. Sasieni explained, is likely due to HPV vaccine coverage not expanding as significantly in the United States as it has in the United Kingdom, and “thus one would anticipate a lower impact on the population in the U.S.”
In the U.S. analysis, Justin Barnes, MD, a radiation oncology resident at Washington University, St. Louis, and colleagues examined cervical cancer incidence between January 2001 and December 2017 using Surveillance, Epidemiology, and End Results and National Program of Cancer Registries data as well as mortality data from the National Center for Health Statistics.
Dr. Barnes and colleagues then compared changes in cervical cancer incidence and mortality between prevaccination years (January 2001 to December 2005) and postvaccination years (January 2010 to December 2017) among three age cohorts – 15-24 years, 25-29 years, and 30-39 years.
“The older 2 groups were included as comparison, given their low vaccination rates,” Dr. Barnes and colleagues explained.
Results show that between the prevaccination and postvaccination periods, the incidence of cervical cancer dropped by 38% in the youngest cohort and by only 16% in the middle-aged group and 8% in the oldest cohort.
Women and girls in the youngest group saw a striking drop in mortality: a 43% decline, which translated to a mortality rate of 0.6 per 100,000.
On the other hand, the authors report a 4.7% decline in mortality in the oldest group and a 4.3% increase in mortality in the middle-aged group – translating to a mortality rate of 1.89 per 100,000 and 0.57 per 100,000, respectively.
Overall, “these nationwide data showed decreased cervical cancer incidence and mortality among women and girls aged 15-24 years after HPV vaccine introduction,” Dr. Barnes and colleagues wrote. The changes in cervical cancer incidence and mortality observed in the youngest age group “were greater than changes in those aged 25 to 29 years and 30 to 39 years, suggesting possible associations with HPV vaccination.”
This analysis lines up with previous evidence from U.S. epidemiologic data, which “have shown decreased cervical cancer incidence after vaccine implementation in women and girls aged 15 to 24 years but not older women.”
Although “the number of deaths and hence the number of potentially averted deaths in young women and girls was small,” the study adds to the current literature by “providing suggestive evidence for vaccine-associated decreases in cervical cancer mortality,” investigators concluded.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The analysis adds to a growing body of evidence demonstrating vaccine-associated changes in cervical cancer incidence and mortality.
Previous data from the United Kingdom, published earlier in November, showed that cervical cancer rates were 87% lower among girls who received the HPV vaccine compared to previously unvaccinated generations. Based on the analysis, the authors concluded that the UK’s HPV immunization program “almost eliminated cervical cancer” in women born since September 1995.
The latest study, published Nov. 29 in JAMA Pediatrics , reports a 38% drop in cervical cancer incidence and a 43% decline in mortality among young women and girls after HPV vaccination was introduced in the United States.
“These results are encouraging,” Peter Sasieni, MD, of King’s College London, and senior author on the U.K. study, told this news organization in an email.
The difference in incidence rates between the U.K. and U.S. studies, Dr. Sasieni explained, is likely due to HPV vaccine coverage not expanding as significantly in the United States as it has in the United Kingdom, and “thus one would anticipate a lower impact on the population in the U.S.”
In the U.S. analysis, Justin Barnes, MD, a radiation oncology resident at Washington University, St. Louis, and colleagues examined cervical cancer incidence between January 2001 and December 2017 using Surveillance, Epidemiology, and End Results and National Program of Cancer Registries data as well as mortality data from the National Center for Health Statistics.
Dr. Barnes and colleagues then compared changes in cervical cancer incidence and mortality between prevaccination years (January 2001 to December 2005) and postvaccination years (January 2010 to December 2017) among three age cohorts – 15-24 years, 25-29 years, and 30-39 years.
“The older 2 groups were included as comparison, given their low vaccination rates,” Dr. Barnes and colleagues explained.
Results show that between the prevaccination and postvaccination periods, the incidence of cervical cancer dropped by 38% in the youngest cohort and by only 16% in the middle-aged group and 8% in the oldest cohort.
Women and girls in the youngest group saw a striking drop in mortality: a 43% decline, which translated to a mortality rate of 0.6 per 100,000.
On the other hand, the authors report a 4.7% decline in mortality in the oldest group and a 4.3% increase in mortality in the middle-aged group – translating to a mortality rate of 1.89 per 100,000 and 0.57 per 100,000, respectively.
Overall, “these nationwide data showed decreased cervical cancer incidence and mortality among women and girls aged 15-24 years after HPV vaccine introduction,” Dr. Barnes and colleagues wrote. The changes in cervical cancer incidence and mortality observed in the youngest age group “were greater than changes in those aged 25 to 29 years and 30 to 39 years, suggesting possible associations with HPV vaccination.”
This analysis lines up with previous evidence from U.S. epidemiologic data, which “have shown decreased cervical cancer incidence after vaccine implementation in women and girls aged 15 to 24 years but not older women.”
Although “the number of deaths and hence the number of potentially averted deaths in young women and girls was small,” the study adds to the current literature by “providing suggestive evidence for vaccine-associated decreases in cervical cancer mortality,” investigators concluded.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
The analysis adds to a growing body of evidence demonstrating vaccine-associated changes in cervical cancer incidence and mortality.
Previous data from the United Kingdom, published earlier in November, showed that cervical cancer rates were 87% lower among girls who received the HPV vaccine compared to previously unvaccinated generations. Based on the analysis, the authors concluded that the UK’s HPV immunization program “almost eliminated cervical cancer” in women born since September 1995.
The latest study, published Nov. 29 in JAMA Pediatrics , reports a 38% drop in cervical cancer incidence and a 43% decline in mortality among young women and girls after HPV vaccination was introduced in the United States.
“These results are encouraging,” Peter Sasieni, MD, of King’s College London, and senior author on the U.K. study, told this news organization in an email.
The difference in incidence rates between the U.K. and U.S. studies, Dr. Sasieni explained, is likely due to HPV vaccine coverage not expanding as significantly in the United States as it has in the United Kingdom, and “thus one would anticipate a lower impact on the population in the U.S.”
In the U.S. analysis, Justin Barnes, MD, a radiation oncology resident at Washington University, St. Louis, and colleagues examined cervical cancer incidence between January 2001 and December 2017 using Surveillance, Epidemiology, and End Results and National Program of Cancer Registries data as well as mortality data from the National Center for Health Statistics.
Dr. Barnes and colleagues then compared changes in cervical cancer incidence and mortality between prevaccination years (January 2001 to December 2005) and postvaccination years (January 2010 to December 2017) among three age cohorts – 15-24 years, 25-29 years, and 30-39 years.
“The older 2 groups were included as comparison, given their low vaccination rates,” Dr. Barnes and colleagues explained.
Results show that between the prevaccination and postvaccination periods, the incidence of cervical cancer dropped by 38% in the youngest cohort and by only 16% in the middle-aged group and 8% in the oldest cohort.
Women and girls in the youngest group saw a striking drop in mortality: a 43% decline, which translated to a mortality rate of 0.6 per 100,000.
On the other hand, the authors report a 4.7% decline in mortality in the oldest group and a 4.3% increase in mortality in the middle-aged group – translating to a mortality rate of 1.89 per 100,000 and 0.57 per 100,000, respectively.
Overall, “these nationwide data showed decreased cervical cancer incidence and mortality among women and girls aged 15-24 years after HPV vaccine introduction,” Dr. Barnes and colleagues wrote. The changes in cervical cancer incidence and mortality observed in the youngest age group “were greater than changes in those aged 25 to 29 years and 30 to 39 years, suggesting possible associations with HPV vaccination.”
This analysis lines up with previous evidence from U.S. epidemiologic data, which “have shown decreased cervical cancer incidence after vaccine implementation in women and girls aged 15 to 24 years but not older women.”
Although “the number of deaths and hence the number of potentially averted deaths in young women and girls was small,” the study adds to the current literature by “providing suggestive evidence for vaccine-associated decreases in cervical cancer mortality,” investigators concluded.
The authors have disclosed no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM JAMA PEDIATRICS

